User login
C. difficile Infections Hit All-Time High
Clostridium difficile infections have reached an all-time high in the United States, and 94% of these infections initiate with medical care, based on data from the Centers for Disease Control and Prevention. C. difficile–related deaths increased from 3,000 in 1999-2000 to 14,000 in 2006-2007, according to the CDC.
The data were published as a CDC Vital Signs report and were presented in a telebriefing on March 6.
C. difficile is "a formidable opponent," and a patient safety issue everywhere that medical care is provided, said Dr. Clifford McDonald, a CDC epidemiologist and the lead author of the report. CDC’s data show that 25% of C. difficile infections first appear in hospitalized patients, while 75% occur either in nursing home residents or in people recently treated in doctors’ offices or clinics. People most at risk are those who take antibiotics and receive care in an outpatient setting.
In general, the risk of developing C. difficile increases with age; although half of C. difficile infections occur in those younger than 65 years, 90% of C. difficile-related deaths occur in those aged 65 years and older, said Dr. McDonald.
He said that clinicians can help reduce C. difficile infections by following six steps:
• Prescribe antibiotics judiciously.
• Be proactive about testing patients for C. difficile if they develop diarrhea while taking antibiotics.
• Isolate patients with C. difficile.
• Wear gloves and gowns when treating C. difficile patients, even for short visits.
• Clean surfaces in exam and treatment rooms with bleach or other spore-killing products.
• When a patient transfers to another facility, notify the medical team about a C. difficile infection.
Also, be sure to order the appropriate cultures to determine whether antibiotics are really needed, Dr. McDonald suggested, and watch for signs that signal C. difficile. "Antibiotic-associated diarrhea is very common," but C. difficile accounts for only about one-third of that, he said.
However, certain clues suggest C. difficile, including more than three unformed stools in 24 hours, fever, abdominal pain, diarrhea that continues once an antibiotic has been discontinued, or diarrhea that began only once an antibiotic was discontinued, he said.
If someone has been on antibiotics, think about C. difficile early and get them tested, whether they are patients in inpatient or outpatient facilities, Dr. McDonald emphasized.
To determine the current prevalence of C. difficile, CDC researchers reviewed data from their Emerging Infections Program, which conducted population-based surveillance from eight geographic areas, and the National Healthcare Safety Network (NHSN). In 2010, a total of 10,342 cases of C. difficile infection were identified via the Emerging Infections Program in 2010, and a total of 42,157 incident laboratory-identified CDI events were reported via the NHSN.
On a positive note, early results from state-led programs in Illinois, Massachusetts, and New York showed that hospital collaboration can reduce C. difficile infections, Dr. McDonald said. The 71 hospitals in these states that participated in C. difficile–prevention programs reduced infection rates by 20% over 21 months. "These promising results follow similar efforts in England, a nation that dropped C. difficile infections by more than 50% during a recent 3-year period," the CDC researchers said in the full report (MMWR 2012;61:1-6).
For additional information about tracking HAIs infections, contact the Emerging Infections Program or the NHSN.
Dr. McDonald had no financial conflicts to disclose.
The recent alarm by the Centers for Disease Control and Prevention which reports on the increasing incidence and burden of Clostridium difficile infections (CDI) in the United States is drawing attention to a phenomenon which is already well- known to gastroenterologists and infectious Ddisease physicians worldwide. In fact, the sweeping changes in the epidemiology of CDI – with reports of increasing rates, outbreaks, and elevated morbidity and mortality since 2002 originally in parts of Canada and the United States – has now been described in almost every developed country which keeps statistics on this infection.
The only fact which is even more alarming in the CDC’s report is that there has not been any “leveling off” or abatement in this health care–associated complication in the U.S. Although GI and ID physicians, hospitalists, and many other health care providers around the globe have been aware of the disquieting rise in CDI since 2002, the hoped-for stabilization or reduction in its incidence due to aggressive infection prevention and control (IPC) techniques, as has been seen in the U.K., has not materialized in the U.S. and central Canada.
Why is it so difficult to control CDI? Because it requires the accomplishment of multiple simultaneous aggressive IPC maneuvers – all of which must be done correctly -– in order to overcome this infection on an institutional level:
- Appropriate antimicrobial use and stewardship- Rapid testing, isolation, and treatment of any patient with diarrhea who has recently received antibiotics, while awaiting test results
- Rigorous hand-washing and appropriate use of personal protective equipment when in contact with suspected or confirmed cases
- Meticulous and frequent environmental cleaning with sporicidal agents which are tolerable to patients and personnel
- Attention to cleaning and disinfection of the innumerable shared pieces of medical equipment, like blood pressure cuffs, thermometers, bladder scanners, and the like
- High level of scrutiny for quickly detecting and treating recurrences, which occur in 15%-40% of CDI patients
- Real-time surveillance to detect outbreaks and effect heightened measures, when necessary.
The “new CDI” has demonstrated itself to be an unforgiving infection in health care facilities. Any lapse in one or more of the above IPC interventions is enough to cause a rise in the incidence or complication rate.
It is unclear exactly why the “new CDI” is behaving as it does. Certainly, the appearance of a new hyper-virulent strain with additional fluoroquinolone resistance (on top of C. difficile’s “usual” multi-drug resistance), a mutation in the toxin regulator gene, and a seemingly greater propensity to sporulate (and thus resist disinfection) appears to be the event which has coincided with the re-emergence of this disease during the past 10 years.
However, not all of the clinical aspects of this “new CDI” can be explained by these findings alone. For instance, the rapid progression from diarrhea to fulminant colitis in the elderly and the immunocompromised, the high recurrence rate (compared with historical controls) and the elevated morbidity (i.e., colectomy and need for intensive care) and mortality remain unnerving manifestations without a real explanation.
The number of scientific publications describing the new epidemiology, pathogenesis, and prophylactic or treatment options has risen impressively over the past several years in order to gain an understanding of this relentless bacterium.
Medical journals are not the only place for CDI-related news, however. The internet and social media sites are rife with stories of personal tragedies from this affliction. Typical stories like a healthy grandfather undergoing uncomplicated elective surgery but then dying from antibiotic-induced CDI during or soon after his hospitalization are too commonly detailed in surveillance data and in personal blogs.
Sites dedicated to and run by patients who have suffered from CDI compete with other sites established by the all-too-commonly depressed individuals who have had to undergo a therapeutic colectomy or who are suffering multiple recurrences of this disease and are searching for centers offering a fecal transplant.
There is no doubt that the true tragedy of CDI, a health care complication with an attributable mortality of almost 15% in the frail elderly over 85 years of age, is the fact that it is more likely to kill than the primary cause of hospitalization (such as a pneumonia, cellulitis, or hip fracture) in this population.
At a time when CDI is clearly overcoming our ability to control it in many parts of the world, it remains puzzling why the U.S. and many European countries do not yet have a true, real-time local and national CDI surveillance network for tracking the number of cases.
Using hospital discharge data or disease coding has been shown to be inaccurate and too late to be of immediate use. Real-time local, state, and national CDI surveillance is essential in telling us where we are, where we are going, and what we have to do to control this affliction. There is clearly a battle going on between health care providers and CDI in many countries, including the U.S. and Canada. We need to use as many tools and tricks as possible to gain control. This is one war we cannot afford to lose.
MARK MILLER, M.D., FRCPC, is Chief, Infectious Diseases, and Head, Infection Prevention and Control Unit, Jewish General Hospital, Montreal.
The recent alarm by the Centers for Disease Control and Prevention which reports on the increasing incidence and burden of Clostridium difficile infections (CDI) in the United States is drawing attention to a phenomenon which is already well- known to gastroenterologists and infectious Ddisease physicians worldwide. In fact, the sweeping changes in the epidemiology of CDI – with reports of increasing rates, outbreaks, and elevated morbidity and mortality since 2002 originally in parts of Canada and the United States – has now been described in almost every developed country which keeps statistics on this infection.
The only fact which is even more alarming in the CDC’s report is that there has not been any “leveling off” or abatement in this health care–associated complication in the U.S. Although GI and ID physicians, hospitalists, and many other health care providers around the globe have been aware of the disquieting rise in CDI since 2002, the hoped-for stabilization or reduction in its incidence due to aggressive infection prevention and control (IPC) techniques, as has been seen in the U.K., has not materialized in the U.S. and central Canada.
Why is it so difficult to control CDI? Because it requires the accomplishment of multiple simultaneous aggressive IPC maneuvers – all of which must be done correctly -– in order to overcome this infection on an institutional level:
- Appropriate antimicrobial use and stewardship- Rapid testing, isolation, and treatment of any patient with diarrhea who has recently received antibiotics, while awaiting test results
- Rigorous hand-washing and appropriate use of personal protective equipment when in contact with suspected or confirmed cases
- Meticulous and frequent environmental cleaning with sporicidal agents which are tolerable to patients and personnel
- Attention to cleaning and disinfection of the innumerable shared pieces of medical equipment, like blood pressure cuffs, thermometers, bladder scanners, and the like
- High level of scrutiny for quickly detecting and treating recurrences, which occur in 15%-40% of CDI patients
- Real-time surveillance to detect outbreaks and effect heightened measures, when necessary.
The “new CDI” has demonstrated itself to be an unforgiving infection in health care facilities. Any lapse in one or more of the above IPC interventions is enough to cause a rise in the incidence or complication rate.
It is unclear exactly why the “new CDI” is behaving as it does. Certainly, the appearance of a new hyper-virulent strain with additional fluoroquinolone resistance (on top of C. difficile’s “usual” multi-drug resistance), a mutation in the toxin regulator gene, and a seemingly greater propensity to sporulate (and thus resist disinfection) appears to be the event which has coincided with the re-emergence of this disease during the past 10 years.
However, not all of the clinical aspects of this “new CDI” can be explained by these findings alone. For instance, the rapid progression from diarrhea to fulminant colitis in the elderly and the immunocompromised, the high recurrence rate (compared with historical controls) and the elevated morbidity (i.e., colectomy and need for intensive care) and mortality remain unnerving manifestations without a real explanation.
The number of scientific publications describing the new epidemiology, pathogenesis, and prophylactic or treatment options has risen impressively over the past several years in order to gain an understanding of this relentless bacterium.
Medical journals are not the only place for CDI-related news, however. The internet and social media sites are rife with stories of personal tragedies from this affliction. Typical stories like a healthy grandfather undergoing uncomplicated elective surgery but then dying from antibiotic-induced CDI during or soon after his hospitalization are too commonly detailed in surveillance data and in personal blogs.
Sites dedicated to and run by patients who have suffered from CDI compete with other sites established by the all-too-commonly depressed individuals who have had to undergo a therapeutic colectomy or who are suffering multiple recurrences of this disease and are searching for centers offering a fecal transplant.
There is no doubt that the true tragedy of CDI, a health care complication with an attributable mortality of almost 15% in the frail elderly over 85 years of age, is the fact that it is more likely to kill than the primary cause of hospitalization (such as a pneumonia, cellulitis, or hip fracture) in this population.
At a time when CDI is clearly overcoming our ability to control it in many parts of the world, it remains puzzling why the U.S. and many European countries do not yet have a true, real-time local and national CDI surveillance network for tracking the number of cases.
Using hospital discharge data or disease coding has been shown to be inaccurate and too late to be of immediate use. Real-time local, state, and national CDI surveillance is essential in telling us where we are, where we are going, and what we have to do to control this affliction. There is clearly a battle going on between health care providers and CDI in many countries, including the U.S. and Canada. We need to use as many tools and tricks as possible to gain control. This is one war we cannot afford to lose.
MARK MILLER, M.D., FRCPC, is Chief, Infectious Diseases, and Head, Infection Prevention and Control Unit, Jewish General Hospital, Montreal.
The recent alarm by the Centers for Disease Control and Prevention which reports on the increasing incidence and burden of Clostridium difficile infections (CDI) in the United States is drawing attention to a phenomenon which is already well- known to gastroenterologists and infectious Ddisease physicians worldwide. In fact, the sweeping changes in the epidemiology of CDI – with reports of increasing rates, outbreaks, and elevated morbidity and mortality since 2002 originally in parts of Canada and the United States – has now been described in almost every developed country which keeps statistics on this infection.
The only fact which is even more alarming in the CDC’s report is that there has not been any “leveling off” or abatement in this health care–associated complication in the U.S. Although GI and ID physicians, hospitalists, and many other health care providers around the globe have been aware of the disquieting rise in CDI since 2002, the hoped-for stabilization or reduction in its incidence due to aggressive infection prevention and control (IPC) techniques, as has been seen in the U.K., has not materialized in the U.S. and central Canada.
Why is it so difficult to control CDI? Because it requires the accomplishment of multiple simultaneous aggressive IPC maneuvers – all of which must be done correctly -– in order to overcome this infection on an institutional level:
- Appropriate antimicrobial use and stewardship- Rapid testing, isolation, and treatment of any patient with diarrhea who has recently received antibiotics, while awaiting test results
- Rigorous hand-washing and appropriate use of personal protective equipment when in contact with suspected or confirmed cases
- Meticulous and frequent environmental cleaning with sporicidal agents which are tolerable to patients and personnel
- Attention to cleaning and disinfection of the innumerable shared pieces of medical equipment, like blood pressure cuffs, thermometers, bladder scanners, and the like
- High level of scrutiny for quickly detecting and treating recurrences, which occur in 15%-40% of CDI patients
- Real-time surveillance to detect outbreaks and effect heightened measures, when necessary.
The “new CDI” has demonstrated itself to be an unforgiving infection in health care facilities. Any lapse in one or more of the above IPC interventions is enough to cause a rise in the incidence or complication rate.
It is unclear exactly why the “new CDI” is behaving as it does. Certainly, the appearance of a new hyper-virulent strain with additional fluoroquinolone resistance (on top of C. difficile’s “usual” multi-drug resistance), a mutation in the toxin regulator gene, and a seemingly greater propensity to sporulate (and thus resist disinfection) appears to be the event which has coincided with the re-emergence of this disease during the past 10 years.
However, not all of the clinical aspects of this “new CDI” can be explained by these findings alone. For instance, the rapid progression from diarrhea to fulminant colitis in the elderly and the immunocompromised, the high recurrence rate (compared with historical controls) and the elevated morbidity (i.e., colectomy and need for intensive care) and mortality remain unnerving manifestations without a real explanation.
The number of scientific publications describing the new epidemiology, pathogenesis, and prophylactic or treatment options has risen impressively over the past several years in order to gain an understanding of this relentless bacterium.
Medical journals are not the only place for CDI-related news, however. The internet and social media sites are rife with stories of personal tragedies from this affliction. Typical stories like a healthy grandfather undergoing uncomplicated elective surgery but then dying from antibiotic-induced CDI during or soon after his hospitalization are too commonly detailed in surveillance data and in personal blogs.
Sites dedicated to and run by patients who have suffered from CDI compete with other sites established by the all-too-commonly depressed individuals who have had to undergo a therapeutic colectomy or who are suffering multiple recurrences of this disease and are searching for centers offering a fecal transplant.
There is no doubt that the true tragedy of CDI, a health care complication with an attributable mortality of almost 15% in the frail elderly over 85 years of age, is the fact that it is more likely to kill than the primary cause of hospitalization (such as a pneumonia, cellulitis, or hip fracture) in this population.
At a time when CDI is clearly overcoming our ability to control it in many parts of the world, it remains puzzling why the U.S. and many European countries do not yet have a true, real-time local and national CDI surveillance network for tracking the number of cases.
Using hospital discharge data or disease coding has been shown to be inaccurate and too late to be of immediate use. Real-time local, state, and national CDI surveillance is essential in telling us where we are, where we are going, and what we have to do to control this affliction. There is clearly a battle going on between health care providers and CDI in many countries, including the U.S. and Canada. We need to use as many tools and tricks as possible to gain control. This is one war we cannot afford to lose.
MARK MILLER, M.D., FRCPC, is Chief, Infectious Diseases, and Head, Infection Prevention and Control Unit, Jewish General Hospital, Montreal.
Clostridium difficile infections have reached an all-time high in the United States, and 94% of these infections initiate with medical care, based on data from the Centers for Disease Control and Prevention. C. difficile–related deaths increased from 3,000 in 1999-2000 to 14,000 in 2006-2007, according to the CDC.
The data were published as a CDC Vital Signs report and were presented in a telebriefing on March 6.
C. difficile is "a formidable opponent," and a patient safety issue everywhere that medical care is provided, said Dr. Clifford McDonald, a CDC epidemiologist and the lead author of the report. CDC’s data show that 25% of C. difficile infections first appear in hospitalized patients, while 75% occur either in nursing home residents or in people recently treated in doctors’ offices or clinics. People most at risk are those who take antibiotics and receive care in an outpatient setting.
In general, the risk of developing C. difficile increases with age; although half of C. difficile infections occur in those younger than 65 years, 90% of C. difficile-related deaths occur in those aged 65 years and older, said Dr. McDonald.
He said that clinicians can help reduce C. difficile infections by following six steps:
• Prescribe antibiotics judiciously.
• Be proactive about testing patients for C. difficile if they develop diarrhea while taking antibiotics.
• Isolate patients with C. difficile.
• Wear gloves and gowns when treating C. difficile patients, even for short visits.
• Clean surfaces in exam and treatment rooms with bleach or other spore-killing products.
• When a patient transfers to another facility, notify the medical team about a C. difficile infection.
Also, be sure to order the appropriate cultures to determine whether antibiotics are really needed, Dr. McDonald suggested, and watch for signs that signal C. difficile. "Antibiotic-associated diarrhea is very common," but C. difficile accounts for only about one-third of that, he said.
However, certain clues suggest C. difficile, including more than three unformed stools in 24 hours, fever, abdominal pain, diarrhea that continues once an antibiotic has been discontinued, or diarrhea that began only once an antibiotic was discontinued, he said.
If someone has been on antibiotics, think about C. difficile early and get them tested, whether they are patients in inpatient or outpatient facilities, Dr. McDonald emphasized.
To determine the current prevalence of C. difficile, CDC researchers reviewed data from their Emerging Infections Program, which conducted population-based surveillance from eight geographic areas, and the National Healthcare Safety Network (NHSN). In 2010, a total of 10,342 cases of C. difficile infection were identified via the Emerging Infections Program in 2010, and a total of 42,157 incident laboratory-identified CDI events were reported via the NHSN.
On a positive note, early results from state-led programs in Illinois, Massachusetts, and New York showed that hospital collaboration can reduce C. difficile infections, Dr. McDonald said. The 71 hospitals in these states that participated in C. difficile–prevention programs reduced infection rates by 20% over 21 months. "These promising results follow similar efforts in England, a nation that dropped C. difficile infections by more than 50% during a recent 3-year period," the CDC researchers said in the full report (MMWR 2012;61:1-6).
For additional information about tracking HAIs infections, contact the Emerging Infections Program or the NHSN.
Dr. McDonald had no financial conflicts to disclose.
Clostridium difficile infections have reached an all-time high in the United States, and 94% of these infections initiate with medical care, based on data from the Centers for Disease Control and Prevention. C. difficile–related deaths increased from 3,000 in 1999-2000 to 14,000 in 2006-2007, according to the CDC.
The data were published as a CDC Vital Signs report and were presented in a telebriefing on March 6.
C. difficile is "a formidable opponent," and a patient safety issue everywhere that medical care is provided, said Dr. Clifford McDonald, a CDC epidemiologist and the lead author of the report. CDC’s data show that 25% of C. difficile infections first appear in hospitalized patients, while 75% occur either in nursing home residents or in people recently treated in doctors’ offices or clinics. People most at risk are those who take antibiotics and receive care in an outpatient setting.
In general, the risk of developing C. difficile increases with age; although half of C. difficile infections occur in those younger than 65 years, 90% of C. difficile-related deaths occur in those aged 65 years and older, said Dr. McDonald.
He said that clinicians can help reduce C. difficile infections by following six steps:
• Prescribe antibiotics judiciously.
• Be proactive about testing patients for C. difficile if they develop diarrhea while taking antibiotics.
• Isolate patients with C. difficile.
• Wear gloves and gowns when treating C. difficile patients, even for short visits.
• Clean surfaces in exam and treatment rooms with bleach or other spore-killing products.
• When a patient transfers to another facility, notify the medical team about a C. difficile infection.
Also, be sure to order the appropriate cultures to determine whether antibiotics are really needed, Dr. McDonald suggested, and watch for signs that signal C. difficile. "Antibiotic-associated diarrhea is very common," but C. difficile accounts for only about one-third of that, he said.
However, certain clues suggest C. difficile, including more than three unformed stools in 24 hours, fever, abdominal pain, diarrhea that continues once an antibiotic has been discontinued, or diarrhea that began only once an antibiotic was discontinued, he said.
If someone has been on antibiotics, think about C. difficile early and get them tested, whether they are patients in inpatient or outpatient facilities, Dr. McDonald emphasized.
To determine the current prevalence of C. difficile, CDC researchers reviewed data from their Emerging Infections Program, which conducted population-based surveillance from eight geographic areas, and the National Healthcare Safety Network (NHSN). In 2010, a total of 10,342 cases of C. difficile infection were identified via the Emerging Infections Program in 2010, and a total of 42,157 incident laboratory-identified CDI events were reported via the NHSN.
On a positive note, early results from state-led programs in Illinois, Massachusetts, and New York showed that hospital collaboration can reduce C. difficile infections, Dr. McDonald said. The 71 hospitals in these states that participated in C. difficile–prevention programs reduced infection rates by 20% over 21 months. "These promising results follow similar efforts in England, a nation that dropped C. difficile infections by more than 50% during a recent 3-year period," the CDC researchers said in the full report (MMWR 2012;61:1-6).
For additional information about tracking HAIs infections, contact the Emerging Infections Program or the NHSN.
Dr. McDonald had no financial conflicts to disclose.
Major Finding: C. difficile-related deaths increased from 3,000 in 1999-2000 to 14,000 in 2006-2007.
Data Source: The data were taken from the Centers for Disease Control and Prevention’s Emerging Infections Program and the National Healthcare Safety Network (NHSN).
Disclosures: Dr. McDonald had no financial conflicts to disclose.
Different Strokes for Different Folks
A 35‐year‐old woman presented to her primary care physician complaining of left post‐auricular pain, swelling, and redness. She described the pain as 8 out of 10, constant, sharp, and nonradiating. She denied fever or chills. A presumptive diagnosis of cellulitis led to a prescription for oral trimethoprim‐sulfamethoxazole. Left facial swelling worsened despite 4 days of antibiotics, so she came to the emergency department.
Noninfectious causes of this woman's symptoms include trauma, or an inflammatory condition such as polychondritis. Key infectious considerations are mastoiditis or a mastoid abscess. Herpes zoster with involvement of the pinna and auditory canal may also present with pain and redness. In the absence of findings suggestive of an infection arising from the auditory canal, cellulitis is a reasonable consideration. With the growing incidence of community‐acquired methicillin‐resistant Staphylococcus aureus infections, an agent effective against this pathogen such as trimethoprim‐sulfamethoxazole may be used, usually in combination with an antibiotic that provides more reliable coverage for group A streptococcus.
Her past medical history included poorly controlled type II diabetes mellitus and asthma. She reported no previous surgical history. Her current medications were insulin, albuterol inhaler, and trimethoprim‐sulfamethoxazole, although she had a history of noncompliance with her insulin. She was married with 1 child and was unemployed. She smoked 1 pack of cigarettes daily, drank up to 6 beers daily, and denied use of illicit drugs.
Her history of diabetes increases her risk of malignant otitis externa. Both diabetes and excess alcohol consumption are risk factors for herpes zoster. Smoking has been shown to increase the risk of otitis media and carriage by S. pneumoniae, a common pathogen in ear infections.
She was ill‐appearing and in moderate respiratory distress. Her temperature was 39C, blood pressure 149/93 mmHg, pulse 95 beats per minute, respiratory rate of 26 times per minute, with an oxygen saturation of 96% while breathing ambient air. She had swelling of the left side of the face extending to the left forehead and lateral neck. Examination of the external ear and auditory canal were unremarkable. The swelling had no associated erythema, tenderness, or lymphadenopathy. She had no oropharyngeal or nasal ulcers present. Her pupils were equal, round, and reactive to light and accommodation with normal sclera. Her trachea was midline; thyroid exam was normal. The heart sounds included normal S1 and S2 without murmurs, rubs, or gallops. Her lung exam was remarkable for inspiratory stridor. The abdominal examination revealed no distention, tenderness, organomegaly, or masses. Cranial nerve testing revealed a left‐sided central seventh nerve palsy along with decreased visual acuity of the left eye. Strength, sensation, and deep tendon reflexes were normal.
While there are many causes of facial nerve palsy, distinguishing between a peripheral palsy (which causes paralysis of the entire ipsilateral side of the face) and a central palsy (which spares the musculature of the forehead) is important. The most common type of peripheral facial nerve palsy is Bell's palsy. Infections such as meningitis or tumors of the central nervous system can cause central facial nerve or other cranial nerve palsy. Important infections to consider in this case would be viral such as herpes zoster or simplex, or atypical bacteria such as Mycoplasma and Rickettsia, which may explain the neurologic but not all of the other clinical findings in this case. It is also critical to determine whether she has an isolated seventh cranial nerve palsy or if other cranial nerves are involved such as may occur with basilar meningitis, which has a myriad of infectious and noninfectious causes. The decreased visual acuity may be a result of corneal dryness and abrasions from inability to close the eye but may also represent optic nerve problems, so detailed ophthalmologic exam is essential. Her ill appearance coupled with facial and neck swelling leads me to at least consider Lemierre's syndrome with central nervous system involvement. Finally, facial swelling and the inspiratory stridor may represent angioedema, although one‐sided involvement of the face would be unusual.
The results of initial laboratory testing were as follows: sodium, 138 mmol/L; potassium, 3.4 mmol/L; chloride, 109 mmol/L; bicarbonate, 14 mmol/L; blood urea nitrogen level, 19 mg/dL; creatinine, 1.1 mg/dL; white cell count, 23,510/mm3; differential, 90% neutrophils, 1% bands, 7% lymphocytes, 2% monocytes; hemoglobin level, 12.5 g/dL; platelet count, 566,000/mm3; hemoglobin A1c, 11%; albumin, 1.6 g/dL; total protein, 6.2 g/dL; total bilirubin, 0.8 mg/dL; alkaline phosphatase, 103 U/L; alanine aminotransferase level, 14 U/L; international normalized ratio of 1.2; partial thromboplastin time, 29 seconds (normal value, 2434 seconds); erythrocyte sedimentation rate, 121 mm/hr; creatine kinase, 561 U/L (normal value 25190). Arterial blood gas measurements with the patient breathing 50% oxygen revealed a pH of 7.34, a partial pressure of carbon dioxide of 28 mmHg, and a partial pressure of oxygen of 228 mmHg.
I am concerned that this patient has sepsis, likely due to an infectious trigger. With her clinical presentation localized to the head and neck, her history of diabetes, and the accelerated sedimentation rate, malignant otitis externa would explain many of her findings. Empiric anti‐infective therapy directed toward Pseudomonas aeruginosa should be initiated, and imaging of the head and ear should be undertaken.
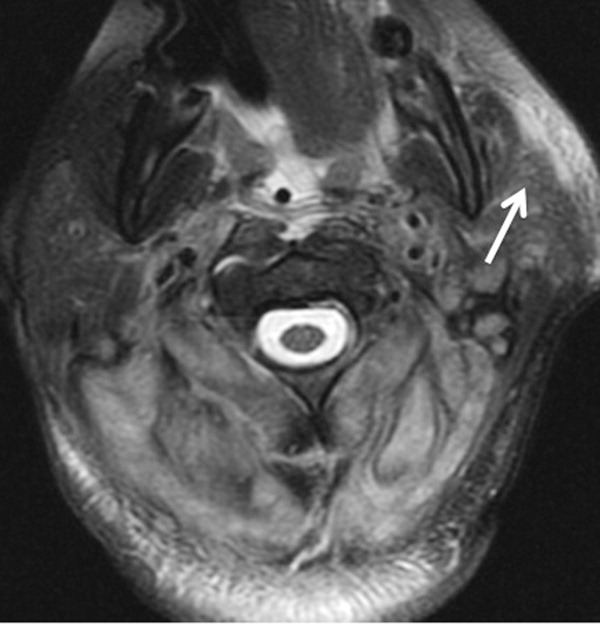
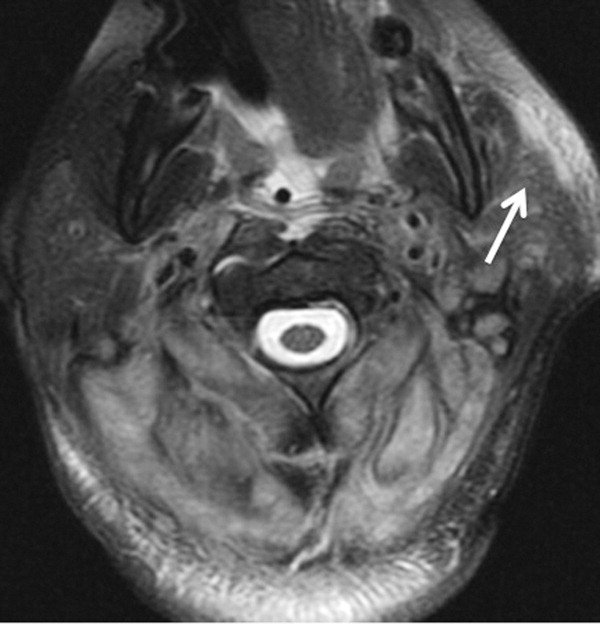
The patient required intubation due to increased respiratory distress and stridor. Her physicians used intravenous vancomycin, clindamycin, and piperacillin/tazobactam to treat presumed cellulitis. Her abnormal neurologic exam led to magnetic resonance (MR) imaging and MR angiography of her neck and brain, which showed evidence of multiple regions of ischemia in the left occipital and inferior parietal distributions, as well as bilateral cerebellar distributions and enhancement of the parotid gland and mastoid air cells (Figure 1). A cerebral angiogram revealed irregularity and caliber reduction in multiple cervical and intracranial arteries, associated with intraluminal thrombi within the left intracranial vertebral artery, consistent with either vasculitis or infectious angioinvasion (Figure 2).

The angioinvasive nature of the findings on imaging leads me to suspect fungal infection. The patient's history of diabetes mellitus and acidosis are risk factors for mucormycosis. Aspergillus and Fusarium may also be angioinvasive but would be much more likely in neutropenic or severely immunocompromised patients. S. aureus may cause septic emboli mimicking angioinvasion but should be readily detected in conventional blood cultures. At this point, I would empirically begin amphotericin B; tissue, however, is needed for definitive diagnosis and a surgical consult should be requested.
After reviewing her imaging studies, an investigation for vasculitis and hypercoagulable states including antinuclear antibody, anti‐deoxyribonucleic acid, anti‐Smith antibody, anti‐SSA antibody level, anti‐SSB level, antineutrophil cytoplasmic antibody, activated protein C resistance level, factor VIII level, human immunodeficiency virus antibody, homocysteine level, cardiolipin antibody testing, lupus anticoagulant, prothrombin 20210 mutation, and protein C level was done, and all tests were normal. Protein S level was slightly low at 64% (normal value 65%140%). Given the enlarged parotid gland and the enhancement of the left parotid bed on magnetic resonance imaging, she underwent a parotid biopsy that revealed sialadenitis.
Systemic vasculitides can result in tissue damage, mediated by the release of endogenous cellular contents from dying cells, known as damage‐associated molecular patterns, sufficient to cause systemic inflammatory response syndrome (SIRS). This patient presented with acute symptoms but has negative laboratory studies for autoantibodies. The parotid biopsy also did not reveal evidence of vasculitis. All these findings make the diagnosis of vasculitis much less likely.
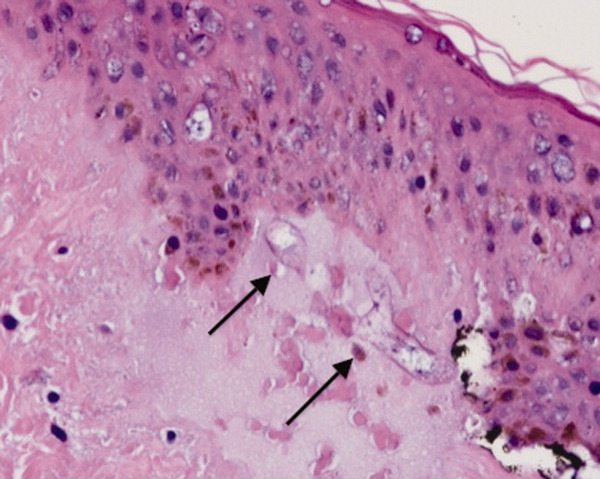
She remained in the medical intensive care unit on mechanical ventilation, with minimal symptomatic improvement. On hospital day 10, the patient developed necrosis of the left external ear. A punch biopsy of the necrotic area of her left pinna was performed; the pathology report read: Sections of punch biopsy of skin show an unremarkable epidermis. There is dermal necrosis involving the stroma and adnexal structures. Intravascular thrombi within the deep dermis are seen. Within superficial dermis there are broad, elongated, nonseptated hyaline structures reminiscent of Mucor. Special stains (periodic acid‐Schiff stain and Grocott Gomori methenamine silver stain [GMS]) performed with appropriately reactive controls fail to highlight these structures (Figure 3). The infectious disease team reviewed the pathology slides with the pathologist. As there was inconclusive evidence for zygomycosis, ie, only a few hyaline structures which failed to stain with GMS stain, the consultants recommended no change in the patient's management.
The gross and microscopic evidence of necrosis and areas of intravascular thrombi are nonspecific but compatible with a fungal infection in a patient with risk factors for zygomycosis. The GMS stain is a very sensitive stain for fungal structures, so a negative stain in this case is surprising, but additional testing such as immunohistochemistry should be pursued to confirm or refute this diagnosis. While Rhizopus species can be contaminants, the laboratory finding of these organisms in specimens from patients with risk factors for zygomycosis should not be ignored.
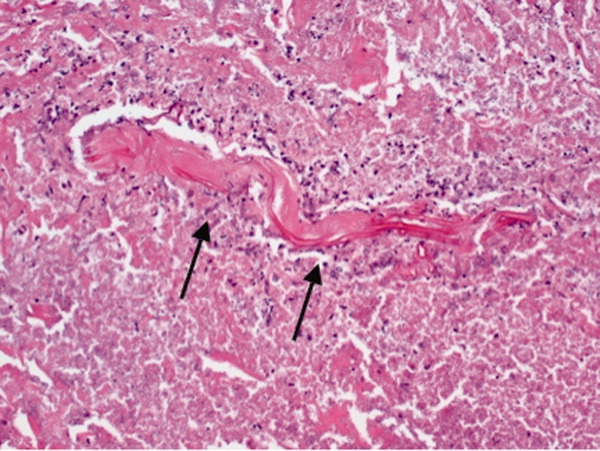
On hospital day 12, the patient was noted to have increased facial swelling. A computed tomographic (CT) angiogram of the neck revealed necrosis of the anterior and posterior paraspinal muscles from the skull base to C34, marked swelling of the left parotid gland, and left inferior parieto‐occipital enhancing lesion. An incisional parotid biopsy was performed. Special stains were positive for broad‐based fungal hyphae consistent with mucormycosis (Figure 4).
Given these findings, the patient should be started on amphotericin B immediately. Medical therapy alone generally does not suffice, and aggressive surgical debridement combined with intravenous antifungal therapy results in better outcomes. The longer the duration of symptoms and the greater the progression of disease, the less favorable the prognosis.
The patient was started on amphotericin B lipid complex and micafungin. However, after 16 days of therapy, repeat imaging of the neck showed worsening necrosis of the neck muscles. At this time, she underwent extensive debridement of face and neck, and posaconazole was added. After prolonged hospitalization, she was discharged to a rehabilitation facility on posaconazole. She resided in a nursing facility for 6 months. One year after her hospitalization, she is living at home and is able to ambulate independently, but requires feeding through a percutaneous endoscopic gastrostomy (PEG) tube because she remains dysphagic.
COMMENTARY
Infections caused by the ubiquitous fungi of the class Zygomycetes typically take 1 of 5 forms: rhinocerebral, pulmonary, gastrointestinal, disseminated, and cutaneous. The presentation varies widely, ranging from plaques, skin swelling, pustules, cellulitis, blisters, nodules, ulcerations, and ecthyma gangrenosum‐like lesions to deeper infections such as necrotizing fasciitis, osteomyelitis, and disseminated infection.1 Infections typically occur in immunocompromised hosts, including transplant recipients and patients with hematologic malignancy, but also occur in patients with diabetes mellitus, intravenous drug users, and patients on deferoxamine therapy.2 Deferoxamine and other iron‐binding therapy is thought to predispose to zygomycetes infections because of improved iron uptake of the fungal species and, thus, stimulation of growth.3 Pulmonary and rhinocerebral infections are the most common clinically encountered forms, and 44% of cutaneous infections are complicated by deep extension or dissemination.4
The articles cited above describe the more typical presentations of this rare disease. However, this patient had an unusual presentation, as parotid involvement due to zygomycosis has only been described once previously.5 Her inflammatory vasculitis and ensuing strokes from involvement of the carotid artery are recognized complications of zygomycosis, and in 1 case series of 41 patients with rhinocerebral mucormycosis, carotid involvement was seen in 31% of patients.6 After the punch biopsy of the patient's pinna showing nonseptated hyphae reminiscent of Mucor, why did her physicians delay administering amphotericin?
There are 2 likely possibilities: anchoring bias or error in medical decision‐making due to inaccurate probability estimates. Anchoring bias describes a heuristic where the initial diagnosis or gestalt biases the physician's process for assigning a final diagnosis.7, 8 This bias creates cognitive errors by limiting creativity in diagnosis. In this case, the infectious disease team carefully weighed the information obtained from the first biopsy. Given their low pretest estimate of this virtually unreported presentation of a rare disease, they decided to evaluate further without beginning antifungal therapy. Of note, there were few hyaline structures, and those structures lacked uptake of GMS. Since they considered the diagnosis yet rejected the diagnosis due to insufficient evidence, it is unlikely that anchoring bias played a role.
Was there an error in medical decision‐making? The physicians in this case faced a very common medical dilemma: whether or not to start a toxic medication empirically or wait for diagnostic confirmation prior to treatment.9 To solve this dilemma, one can apply decision analysis. Moskowitz et al described 5 phases of medical decision analysis by which a probabilistic right answer to clinical scenarios can be deduced mathematically.10 To solve this problem, probabilities must be assigned to the risk of giving a drug to a patient without the disease versus the risk of not giving a drug to a patient with the disease. For example, amphotericin deoxycholate causes acute renal failure in 30% to 47% of patients. Newer formulations of amphotericin, such as liposomal amphotericin and lipid complex, result in lower rates of nephrotoxicity (27% vs 47%). The risk of not giving amphotericin to a patient with zygomycosis is death. Even in patients treated with amphotericin, the mortality rate has been shown to be 66%, and up to 100% in those with strokes related to zygomycosis.2, 6, 11 Simply looking at these probabilities, decision analysis would favor empiric treatment.
The physicians caring for this patient did not have the luxury of retrospective speculation. After looking at all of the data, the equivocal skin biopsy and rare clinical presentation, the question to ask would change: What is the risk of giving amphotericin empirically to someone who, based on available information, has a very low probability of having zygomycosis? When phrased in this manner, there is a 47% chance of nephrotoxicity with amphotericin versus the very small probability that you have diagnosed a case of zygomycosis that has only been described once in the literature. Mathematically andmore importantlyclinically, this question becomes more difficult to answer. However, no value can be placed on the possibility of death in suspected zygomycosis, and the risk of short‐term amphotericin use is much less than that of a course of treatment. As such, empiric therapy should always be given.
Physicians are not mathematicians, and dynamic clinical scenarios are not so easily made into static math problems. Disease presentations evolve over time towards a diagnosable clinical pattern, as was the case with this patient. Two days after the aforementioned biopsy, she worsened and in less time than it would have taken to isolate zygomycosis from the first biopsy, a second biopsy revealed the typical nonseptated hyphae demarcated with the GMS stain. Even appropriate diagnostic testing, thoughtful interpretation, and avoidance of certain cognitive errors can result in incorrect diagnoses and delayed treatment. It is monitoring the progression of disease and collecting additional data that allows physicians to mold a diagnosis and create a treatment plan.
The primary treatment of zygomycosis should include amphotericin. However, there are limited data to support combination therapy with an echinocandin in severe cases, as in this patient.12 Posaconazole is not recommended for monotherapy as an initial therapy, but there is data for its use as salvage therapy in zygomycosis.13 This case highlights the difficulties that physicians face in the diagnosis and treatment of rare diseases. Cerebral infarction in a hematologic malignancy, uncontrolled diabetes, or iron chelation therapy could be the initial presentation of rhinocerebral zygomycosis. There truly are different strokes for different folks. Recognizing this and similar presentations may lead to a more rapid diagnosis and treatment of zygomycosis.
TEACHING POINTS
-
Zygomycosis has a wide range of clinical presentations ranging from skin lesions to deep tissue infections. As it is an angioinvasive organism, it can also present as cerebral infarcts and brain abscesses.
-
Zygomycosis infections should be suspected in patients with uncontrolled diabetes, hematologic or oncologic malignancies, and patients on iron chelation therapy with a potentially compatible clinical picture.
-
If zygomycosis infection is suspected, rapid histologic diagnosis should be attempted. However, as histologic diagnosis can take time, empiric therapy with amphotericin should always be administered.
-
Amphotericin remains the primary medical therapy for this disease; however, there is limited emerging evidence to suggest that echinocandins can be used in combination with amphotericin for improved treatment of severe rhinocerebral zygomyocosis. Posaconazole has a role as salvage therapy in zygomycosis, but should not be used as the sole primary treatment.
The approach to clinical conundrums by an expert clinician is revealed through the presentation of an actual patient's case in an approach typical of a morning report. Similarly to patient care, sequential pieces of information are provided to the clinician, who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
This icon represents the patient's case. Each paragraph that follows represents the discussant's thoughts.
Acknowledgements
The authors are indebted to Dr Glenn Roberson at the Department of Radiology, University of Alabama at Birmingham, for providing the radiographic images; to Dr Aleodor Andea at the Department of Pathology, University of Alabama at Birmingham, for providing the pathology images; and to Dr. Crysten Brinkley at the Department of Neurology at the University of Alabama at Birmingham for her assistance with this case presentation.
Disclosure: Nothing to report.
- ,,,.Mucormycosis: emerging prominence of cutaneous infections.Clin Infect Dis.1994;19:67–76.
- ,,,.Zygomycosis in the 1990s in a tertiary‐care cancer center.Clin Infect Dis.2000;30:851–856.
- ,,, et al.Mucormycosis during deferoxamine therapy is a siderophore‐mediated infection. In vitro and in vivo animal studies.J Clin Invest.1993;91:1979–1986.
- ,,, et al.Epidemiology and outcome of zygomycosis: a review of 929 reported cases.Clin Infect Dis.2005;41:634–653.
- ,,,,,.Cutaneous mucormycosis of the head and neck with parotid gland involvement: first report of a case.Ear Nose Throat J.2004;83:282–286.
- ,,,,,.A successful combined endovascular and surgical treatment of a cranial base mucormycosis with an associated internal carotid artery pseudoaneurysm.Neurosurgery.2009;65:733–740.
- ,.Judgment under uncertainty: heuristics and biases.Science.1974;185:1124–1131.
- ,,,,.Clinical problem‐solving. Anchors away.N Engl J Med.2007;356:504–509.
- ,,,.Clinical problem‐solving. Empirically incorrect.N Engl J Med.2006;354:509–514.
- ,,.Dealing with uncertainty, risks, and tradeoffs in clinical decisions. A cognitive science approach.Ann Intern Med.1988;108:435–449.
- ,,.Fatal strokes in patients with rhino‐orbito‐cerebral mucormycosis and associated vasculopathy.Scand J Infect Dis.2004;36:643–648.
- ,,, et al.Combination polyene‐caspofungin treatment of rhino‐orbital‐cerebral mucormycosis.Clin Infect Dis.2008;47:364–371.
- ,,,,.Posaconazole is effective as salvage therapy in zygomycosis: a retrospective summary of 91 cases.Clin Infect Dis.2006;42:e61–e65.
A 35‐year‐old woman presented to her primary care physician complaining of left post‐auricular pain, swelling, and redness. She described the pain as 8 out of 10, constant, sharp, and nonradiating. She denied fever or chills. A presumptive diagnosis of cellulitis led to a prescription for oral trimethoprim‐sulfamethoxazole. Left facial swelling worsened despite 4 days of antibiotics, so she came to the emergency department.
Noninfectious causes of this woman's symptoms include trauma, or an inflammatory condition such as polychondritis. Key infectious considerations are mastoiditis or a mastoid abscess. Herpes zoster with involvement of the pinna and auditory canal may also present with pain and redness. In the absence of findings suggestive of an infection arising from the auditory canal, cellulitis is a reasonable consideration. With the growing incidence of community‐acquired methicillin‐resistant Staphylococcus aureus infections, an agent effective against this pathogen such as trimethoprim‐sulfamethoxazole may be used, usually in combination with an antibiotic that provides more reliable coverage for group A streptococcus.
Her past medical history included poorly controlled type II diabetes mellitus and asthma. She reported no previous surgical history. Her current medications were insulin, albuterol inhaler, and trimethoprim‐sulfamethoxazole, although she had a history of noncompliance with her insulin. She was married with 1 child and was unemployed. She smoked 1 pack of cigarettes daily, drank up to 6 beers daily, and denied use of illicit drugs.
Her history of diabetes increases her risk of malignant otitis externa. Both diabetes and excess alcohol consumption are risk factors for herpes zoster. Smoking has been shown to increase the risk of otitis media and carriage by S. pneumoniae, a common pathogen in ear infections.
She was ill‐appearing and in moderate respiratory distress. Her temperature was 39C, blood pressure 149/93 mmHg, pulse 95 beats per minute, respiratory rate of 26 times per minute, with an oxygen saturation of 96% while breathing ambient air. She had swelling of the left side of the face extending to the left forehead and lateral neck. Examination of the external ear and auditory canal were unremarkable. The swelling had no associated erythema, tenderness, or lymphadenopathy. She had no oropharyngeal or nasal ulcers present. Her pupils were equal, round, and reactive to light and accommodation with normal sclera. Her trachea was midline; thyroid exam was normal. The heart sounds included normal S1 and S2 without murmurs, rubs, or gallops. Her lung exam was remarkable for inspiratory stridor. The abdominal examination revealed no distention, tenderness, organomegaly, or masses. Cranial nerve testing revealed a left‐sided central seventh nerve palsy along with decreased visual acuity of the left eye. Strength, sensation, and deep tendon reflexes were normal.
While there are many causes of facial nerve palsy, distinguishing between a peripheral palsy (which causes paralysis of the entire ipsilateral side of the face) and a central palsy (which spares the musculature of the forehead) is important. The most common type of peripheral facial nerve palsy is Bell's palsy. Infections such as meningitis or tumors of the central nervous system can cause central facial nerve or other cranial nerve palsy. Important infections to consider in this case would be viral such as herpes zoster or simplex, or atypical bacteria such as Mycoplasma and Rickettsia, which may explain the neurologic but not all of the other clinical findings in this case. It is also critical to determine whether she has an isolated seventh cranial nerve palsy or if other cranial nerves are involved such as may occur with basilar meningitis, which has a myriad of infectious and noninfectious causes. The decreased visual acuity may be a result of corneal dryness and abrasions from inability to close the eye but may also represent optic nerve problems, so detailed ophthalmologic exam is essential. Her ill appearance coupled with facial and neck swelling leads me to at least consider Lemierre's syndrome with central nervous system involvement. Finally, facial swelling and the inspiratory stridor may represent angioedema, although one‐sided involvement of the face would be unusual.
The results of initial laboratory testing were as follows: sodium, 138 mmol/L; potassium, 3.4 mmol/L; chloride, 109 mmol/L; bicarbonate, 14 mmol/L; blood urea nitrogen level, 19 mg/dL; creatinine, 1.1 mg/dL; white cell count, 23,510/mm3; differential, 90% neutrophils, 1% bands, 7% lymphocytes, 2% monocytes; hemoglobin level, 12.5 g/dL; platelet count, 566,000/mm3; hemoglobin A1c, 11%; albumin, 1.6 g/dL; total protein, 6.2 g/dL; total bilirubin, 0.8 mg/dL; alkaline phosphatase, 103 U/L; alanine aminotransferase level, 14 U/L; international normalized ratio of 1.2; partial thromboplastin time, 29 seconds (normal value, 2434 seconds); erythrocyte sedimentation rate, 121 mm/hr; creatine kinase, 561 U/L (normal value 25190). Arterial blood gas measurements with the patient breathing 50% oxygen revealed a pH of 7.34, a partial pressure of carbon dioxide of 28 mmHg, and a partial pressure of oxygen of 228 mmHg.
I am concerned that this patient has sepsis, likely due to an infectious trigger. With her clinical presentation localized to the head and neck, her history of diabetes, and the accelerated sedimentation rate, malignant otitis externa would explain many of her findings. Empiric anti‐infective therapy directed toward Pseudomonas aeruginosa should be initiated, and imaging of the head and ear should be undertaken.
The patient required intubation due to increased respiratory distress and stridor. Her physicians used intravenous vancomycin, clindamycin, and piperacillin/tazobactam to treat presumed cellulitis. Her abnormal neurologic exam led to magnetic resonance (MR) imaging and MR angiography of her neck and brain, which showed evidence of multiple regions of ischemia in the left occipital and inferior parietal distributions, as well as bilateral cerebellar distributions and enhancement of the parotid gland and mastoid air cells (Figure 1). A cerebral angiogram revealed irregularity and caliber reduction in multiple cervical and intracranial arteries, associated with intraluminal thrombi within the left intracranial vertebral artery, consistent with either vasculitis or infectious angioinvasion (Figure 2).


The angioinvasive nature of the findings on imaging leads me to suspect fungal infection. The patient's history of diabetes mellitus and acidosis are risk factors for mucormycosis. Aspergillus and Fusarium may also be angioinvasive but would be much more likely in neutropenic or severely immunocompromised patients. S. aureus may cause septic emboli mimicking angioinvasion but should be readily detected in conventional blood cultures. At this point, I would empirically begin amphotericin B; tissue, however, is needed for definitive diagnosis and a surgical consult should be requested.
After reviewing her imaging studies, an investigation for vasculitis and hypercoagulable states including antinuclear antibody, anti‐deoxyribonucleic acid, anti‐Smith antibody, anti‐SSA antibody level, anti‐SSB level, antineutrophil cytoplasmic antibody, activated protein C resistance level, factor VIII level, human immunodeficiency virus antibody, homocysteine level, cardiolipin antibody testing, lupus anticoagulant, prothrombin 20210 mutation, and protein C level was done, and all tests were normal. Protein S level was slightly low at 64% (normal value 65%140%). Given the enlarged parotid gland and the enhancement of the left parotid bed on magnetic resonance imaging, she underwent a parotid biopsy that revealed sialadenitis.
Systemic vasculitides can result in tissue damage, mediated by the release of endogenous cellular contents from dying cells, known as damage‐associated molecular patterns, sufficient to cause systemic inflammatory response syndrome (SIRS). This patient presented with acute symptoms but has negative laboratory studies for autoantibodies. The parotid biopsy also did not reveal evidence of vasculitis. All these findings make the diagnosis of vasculitis much less likely.
She remained in the medical intensive care unit on mechanical ventilation, with minimal symptomatic improvement. On hospital day 10, the patient developed necrosis of the left external ear. A punch biopsy of the necrotic area of her left pinna was performed; the pathology report read: Sections of punch biopsy of skin show an unremarkable epidermis. There is dermal necrosis involving the stroma and adnexal structures. Intravascular thrombi within the deep dermis are seen. Within superficial dermis there are broad, elongated, nonseptated hyaline structures reminiscent of Mucor. Special stains (periodic acid‐Schiff stain and Grocott Gomori methenamine silver stain [GMS]) performed with appropriately reactive controls fail to highlight these structures (Figure 3). The infectious disease team reviewed the pathology slides with the pathologist. As there was inconclusive evidence for zygomycosis, ie, only a few hyaline structures which failed to stain with GMS stain, the consultants recommended no change in the patient's management.

The gross and microscopic evidence of necrosis and areas of intravascular thrombi are nonspecific but compatible with a fungal infection in a patient with risk factors for zygomycosis. The GMS stain is a very sensitive stain for fungal structures, so a negative stain in this case is surprising, but additional testing such as immunohistochemistry should be pursued to confirm or refute this diagnosis. While Rhizopus species can be contaminants, the laboratory finding of these organisms in specimens from patients with risk factors for zygomycosis should not be ignored.
On hospital day 12, the patient was noted to have increased facial swelling. A computed tomographic (CT) angiogram of the neck revealed necrosis of the anterior and posterior paraspinal muscles from the skull base to C34, marked swelling of the left parotid gland, and left inferior parieto‐occipital enhancing lesion. An incisional parotid biopsy was performed. Special stains were positive for broad‐based fungal hyphae consistent with mucormycosis (Figure 4).

Given these findings, the patient should be started on amphotericin B immediately. Medical therapy alone generally does not suffice, and aggressive surgical debridement combined with intravenous antifungal therapy results in better outcomes. The longer the duration of symptoms and the greater the progression of disease, the less favorable the prognosis.
The patient was started on amphotericin B lipid complex and micafungin. However, after 16 days of therapy, repeat imaging of the neck showed worsening necrosis of the neck muscles. At this time, she underwent extensive debridement of face and neck, and posaconazole was added. After prolonged hospitalization, she was discharged to a rehabilitation facility on posaconazole. She resided in a nursing facility for 6 months. One year after her hospitalization, she is living at home and is able to ambulate independently, but requires feeding through a percutaneous endoscopic gastrostomy (PEG) tube because she remains dysphagic.
COMMENTARY
Infections caused by the ubiquitous fungi of the class Zygomycetes typically take 1 of 5 forms: rhinocerebral, pulmonary, gastrointestinal, disseminated, and cutaneous. The presentation varies widely, ranging from plaques, skin swelling, pustules, cellulitis, blisters, nodules, ulcerations, and ecthyma gangrenosum‐like lesions to deeper infections such as necrotizing fasciitis, osteomyelitis, and disseminated infection.1 Infections typically occur in immunocompromised hosts, including transplant recipients and patients with hematologic malignancy, but also occur in patients with diabetes mellitus, intravenous drug users, and patients on deferoxamine therapy.2 Deferoxamine and other iron‐binding therapy is thought to predispose to zygomycetes infections because of improved iron uptake of the fungal species and, thus, stimulation of growth.3 Pulmonary and rhinocerebral infections are the most common clinically encountered forms, and 44% of cutaneous infections are complicated by deep extension or dissemination.4
The articles cited above describe the more typical presentations of this rare disease. However, this patient had an unusual presentation, as parotid involvement due to zygomycosis has only been described once previously.5 Her inflammatory vasculitis and ensuing strokes from involvement of the carotid artery are recognized complications of zygomycosis, and in 1 case series of 41 patients with rhinocerebral mucormycosis, carotid involvement was seen in 31% of patients.6 After the punch biopsy of the patient's pinna showing nonseptated hyphae reminiscent of Mucor, why did her physicians delay administering amphotericin?
There are 2 likely possibilities: anchoring bias or error in medical decision‐making due to inaccurate probability estimates. Anchoring bias describes a heuristic where the initial diagnosis or gestalt biases the physician's process for assigning a final diagnosis.7, 8 This bias creates cognitive errors by limiting creativity in diagnosis. In this case, the infectious disease team carefully weighed the information obtained from the first biopsy. Given their low pretest estimate of this virtually unreported presentation of a rare disease, they decided to evaluate further without beginning antifungal therapy. Of note, there were few hyaline structures, and those structures lacked uptake of GMS. Since they considered the diagnosis yet rejected the diagnosis due to insufficient evidence, it is unlikely that anchoring bias played a role.
Was there an error in medical decision‐making? The physicians in this case faced a very common medical dilemma: whether or not to start a toxic medication empirically or wait for diagnostic confirmation prior to treatment.9 To solve this dilemma, one can apply decision analysis. Moskowitz et al described 5 phases of medical decision analysis by which a probabilistic right answer to clinical scenarios can be deduced mathematically.10 To solve this problem, probabilities must be assigned to the risk of giving a drug to a patient without the disease versus the risk of not giving a drug to a patient with the disease. For example, amphotericin deoxycholate causes acute renal failure in 30% to 47% of patients. Newer formulations of amphotericin, such as liposomal amphotericin and lipid complex, result in lower rates of nephrotoxicity (27% vs 47%). The risk of not giving amphotericin to a patient with zygomycosis is death. Even in patients treated with amphotericin, the mortality rate has been shown to be 66%, and up to 100% in those with strokes related to zygomycosis.2, 6, 11 Simply looking at these probabilities, decision analysis would favor empiric treatment.
The physicians caring for this patient did not have the luxury of retrospective speculation. After looking at all of the data, the equivocal skin biopsy and rare clinical presentation, the question to ask would change: What is the risk of giving amphotericin empirically to someone who, based on available information, has a very low probability of having zygomycosis? When phrased in this manner, there is a 47% chance of nephrotoxicity with amphotericin versus the very small probability that you have diagnosed a case of zygomycosis that has only been described once in the literature. Mathematically andmore importantlyclinically, this question becomes more difficult to answer. However, no value can be placed on the possibility of death in suspected zygomycosis, and the risk of short‐term amphotericin use is much less than that of a course of treatment. As such, empiric therapy should always be given.
Physicians are not mathematicians, and dynamic clinical scenarios are not so easily made into static math problems. Disease presentations evolve over time towards a diagnosable clinical pattern, as was the case with this patient. Two days after the aforementioned biopsy, she worsened and in less time than it would have taken to isolate zygomycosis from the first biopsy, a second biopsy revealed the typical nonseptated hyphae demarcated with the GMS stain. Even appropriate diagnostic testing, thoughtful interpretation, and avoidance of certain cognitive errors can result in incorrect diagnoses and delayed treatment. It is monitoring the progression of disease and collecting additional data that allows physicians to mold a diagnosis and create a treatment plan.
The primary treatment of zygomycosis should include amphotericin. However, there are limited data to support combination therapy with an echinocandin in severe cases, as in this patient.12 Posaconazole is not recommended for monotherapy as an initial therapy, but there is data for its use as salvage therapy in zygomycosis.13 This case highlights the difficulties that physicians face in the diagnosis and treatment of rare diseases. Cerebral infarction in a hematologic malignancy, uncontrolled diabetes, or iron chelation therapy could be the initial presentation of rhinocerebral zygomycosis. There truly are different strokes for different folks. Recognizing this and similar presentations may lead to a more rapid diagnosis and treatment of zygomycosis.
TEACHING POINTS
-
Zygomycosis has a wide range of clinical presentations ranging from skin lesions to deep tissue infections. As it is an angioinvasive organism, it can also present as cerebral infarcts and brain abscesses.
-
Zygomycosis infections should be suspected in patients with uncontrolled diabetes, hematologic or oncologic malignancies, and patients on iron chelation therapy with a potentially compatible clinical picture.
-
If zygomycosis infection is suspected, rapid histologic diagnosis should be attempted. However, as histologic diagnosis can take time, empiric therapy with amphotericin should always be administered.
-
Amphotericin remains the primary medical therapy for this disease; however, there is limited emerging evidence to suggest that echinocandins can be used in combination with amphotericin for improved treatment of severe rhinocerebral zygomyocosis. Posaconazole has a role as salvage therapy in zygomycosis, but should not be used as the sole primary treatment.
The approach to clinical conundrums by an expert clinician is revealed through the presentation of an actual patient's case in an approach typical of a morning report. Similarly to patient care, sequential pieces of information are provided to the clinician, who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
This icon represents the patient's case. Each paragraph that follows represents the discussant's thoughts.
Acknowledgements
The authors are indebted to Dr Glenn Roberson at the Department of Radiology, University of Alabama at Birmingham, for providing the radiographic images; to Dr Aleodor Andea at the Department of Pathology, University of Alabama at Birmingham, for providing the pathology images; and to Dr. Crysten Brinkley at the Department of Neurology at the University of Alabama at Birmingham for her assistance with this case presentation.
Disclosure: Nothing to report.
A 35‐year‐old woman presented to her primary care physician complaining of left post‐auricular pain, swelling, and redness. She described the pain as 8 out of 10, constant, sharp, and nonradiating. She denied fever or chills. A presumptive diagnosis of cellulitis led to a prescription for oral trimethoprim‐sulfamethoxazole. Left facial swelling worsened despite 4 days of antibiotics, so she came to the emergency department.
Noninfectious causes of this woman's symptoms include trauma, or an inflammatory condition such as polychondritis. Key infectious considerations are mastoiditis or a mastoid abscess. Herpes zoster with involvement of the pinna and auditory canal may also present with pain and redness. In the absence of findings suggestive of an infection arising from the auditory canal, cellulitis is a reasonable consideration. With the growing incidence of community‐acquired methicillin‐resistant Staphylococcus aureus infections, an agent effective against this pathogen such as trimethoprim‐sulfamethoxazole may be used, usually in combination with an antibiotic that provides more reliable coverage for group A streptococcus.
Her past medical history included poorly controlled type II diabetes mellitus and asthma. She reported no previous surgical history. Her current medications were insulin, albuterol inhaler, and trimethoprim‐sulfamethoxazole, although she had a history of noncompliance with her insulin. She was married with 1 child and was unemployed. She smoked 1 pack of cigarettes daily, drank up to 6 beers daily, and denied use of illicit drugs.
Her history of diabetes increases her risk of malignant otitis externa. Both diabetes and excess alcohol consumption are risk factors for herpes zoster. Smoking has been shown to increase the risk of otitis media and carriage by S. pneumoniae, a common pathogen in ear infections.
She was ill‐appearing and in moderate respiratory distress. Her temperature was 39C, blood pressure 149/93 mmHg, pulse 95 beats per minute, respiratory rate of 26 times per minute, with an oxygen saturation of 96% while breathing ambient air. She had swelling of the left side of the face extending to the left forehead and lateral neck. Examination of the external ear and auditory canal were unremarkable. The swelling had no associated erythema, tenderness, or lymphadenopathy. She had no oropharyngeal or nasal ulcers present. Her pupils were equal, round, and reactive to light and accommodation with normal sclera. Her trachea was midline; thyroid exam was normal. The heart sounds included normal S1 and S2 without murmurs, rubs, or gallops. Her lung exam was remarkable for inspiratory stridor. The abdominal examination revealed no distention, tenderness, organomegaly, or masses. Cranial nerve testing revealed a left‐sided central seventh nerve palsy along with decreased visual acuity of the left eye. Strength, sensation, and deep tendon reflexes were normal.
While there are many causes of facial nerve palsy, distinguishing between a peripheral palsy (which causes paralysis of the entire ipsilateral side of the face) and a central palsy (which spares the musculature of the forehead) is important. The most common type of peripheral facial nerve palsy is Bell's palsy. Infections such as meningitis or tumors of the central nervous system can cause central facial nerve or other cranial nerve palsy. Important infections to consider in this case would be viral such as herpes zoster or simplex, or atypical bacteria such as Mycoplasma and Rickettsia, which may explain the neurologic but not all of the other clinical findings in this case. It is also critical to determine whether she has an isolated seventh cranial nerve palsy or if other cranial nerves are involved such as may occur with basilar meningitis, which has a myriad of infectious and noninfectious causes. The decreased visual acuity may be a result of corneal dryness and abrasions from inability to close the eye but may also represent optic nerve problems, so detailed ophthalmologic exam is essential. Her ill appearance coupled with facial and neck swelling leads me to at least consider Lemierre's syndrome with central nervous system involvement. Finally, facial swelling and the inspiratory stridor may represent angioedema, although one‐sided involvement of the face would be unusual.
The results of initial laboratory testing were as follows: sodium, 138 mmol/L; potassium, 3.4 mmol/L; chloride, 109 mmol/L; bicarbonate, 14 mmol/L; blood urea nitrogen level, 19 mg/dL; creatinine, 1.1 mg/dL; white cell count, 23,510/mm3; differential, 90% neutrophils, 1% bands, 7% lymphocytes, 2% monocytes; hemoglobin level, 12.5 g/dL; platelet count, 566,000/mm3; hemoglobin A1c, 11%; albumin, 1.6 g/dL; total protein, 6.2 g/dL; total bilirubin, 0.8 mg/dL; alkaline phosphatase, 103 U/L; alanine aminotransferase level, 14 U/L; international normalized ratio of 1.2; partial thromboplastin time, 29 seconds (normal value, 2434 seconds); erythrocyte sedimentation rate, 121 mm/hr; creatine kinase, 561 U/L (normal value 25190). Arterial blood gas measurements with the patient breathing 50% oxygen revealed a pH of 7.34, a partial pressure of carbon dioxide of 28 mmHg, and a partial pressure of oxygen of 228 mmHg.
I am concerned that this patient has sepsis, likely due to an infectious trigger. With her clinical presentation localized to the head and neck, her history of diabetes, and the accelerated sedimentation rate, malignant otitis externa would explain many of her findings. Empiric anti‐infective therapy directed toward Pseudomonas aeruginosa should be initiated, and imaging of the head and ear should be undertaken.
The patient required intubation due to increased respiratory distress and stridor. Her physicians used intravenous vancomycin, clindamycin, and piperacillin/tazobactam to treat presumed cellulitis. Her abnormal neurologic exam led to magnetic resonance (MR) imaging and MR angiography of her neck and brain, which showed evidence of multiple regions of ischemia in the left occipital and inferior parietal distributions, as well as bilateral cerebellar distributions and enhancement of the parotid gland and mastoid air cells (Figure 1). A cerebral angiogram revealed irregularity and caliber reduction in multiple cervical and intracranial arteries, associated with intraluminal thrombi within the left intracranial vertebral artery, consistent with either vasculitis or infectious angioinvasion (Figure 2).


The angioinvasive nature of the findings on imaging leads me to suspect fungal infection. The patient's history of diabetes mellitus and acidosis are risk factors for mucormycosis. Aspergillus and Fusarium may also be angioinvasive but would be much more likely in neutropenic or severely immunocompromised patients. S. aureus may cause septic emboli mimicking angioinvasion but should be readily detected in conventional blood cultures. At this point, I would empirically begin amphotericin B; tissue, however, is needed for definitive diagnosis and a surgical consult should be requested.
After reviewing her imaging studies, an investigation for vasculitis and hypercoagulable states including antinuclear antibody, anti‐deoxyribonucleic acid, anti‐Smith antibody, anti‐SSA antibody level, anti‐SSB level, antineutrophil cytoplasmic antibody, activated protein C resistance level, factor VIII level, human immunodeficiency virus antibody, homocysteine level, cardiolipin antibody testing, lupus anticoagulant, prothrombin 20210 mutation, and protein C level was done, and all tests were normal. Protein S level was slightly low at 64% (normal value 65%140%). Given the enlarged parotid gland and the enhancement of the left parotid bed on magnetic resonance imaging, she underwent a parotid biopsy that revealed sialadenitis.
Systemic vasculitides can result in tissue damage, mediated by the release of endogenous cellular contents from dying cells, known as damage‐associated molecular patterns, sufficient to cause systemic inflammatory response syndrome (SIRS). This patient presented with acute symptoms but has negative laboratory studies for autoantibodies. The parotid biopsy also did not reveal evidence of vasculitis. All these findings make the diagnosis of vasculitis much less likely.
She remained in the medical intensive care unit on mechanical ventilation, with minimal symptomatic improvement. On hospital day 10, the patient developed necrosis of the left external ear. A punch biopsy of the necrotic area of her left pinna was performed; the pathology report read: Sections of punch biopsy of skin show an unremarkable epidermis. There is dermal necrosis involving the stroma and adnexal structures. Intravascular thrombi within the deep dermis are seen. Within superficial dermis there are broad, elongated, nonseptated hyaline structures reminiscent of Mucor. Special stains (periodic acid‐Schiff stain and Grocott Gomori methenamine silver stain [GMS]) performed with appropriately reactive controls fail to highlight these structures (Figure 3). The infectious disease team reviewed the pathology slides with the pathologist. As there was inconclusive evidence for zygomycosis, ie, only a few hyaline structures which failed to stain with GMS stain, the consultants recommended no change in the patient's management.

The gross and microscopic evidence of necrosis and areas of intravascular thrombi are nonspecific but compatible with a fungal infection in a patient with risk factors for zygomycosis. The GMS stain is a very sensitive stain for fungal structures, so a negative stain in this case is surprising, but additional testing such as immunohistochemistry should be pursued to confirm or refute this diagnosis. While Rhizopus species can be contaminants, the laboratory finding of these organisms in specimens from patients with risk factors for zygomycosis should not be ignored.
On hospital day 12, the patient was noted to have increased facial swelling. A computed tomographic (CT) angiogram of the neck revealed necrosis of the anterior and posterior paraspinal muscles from the skull base to C34, marked swelling of the left parotid gland, and left inferior parieto‐occipital enhancing lesion. An incisional parotid biopsy was performed. Special stains were positive for broad‐based fungal hyphae consistent with mucormycosis (Figure 4).

Given these findings, the patient should be started on amphotericin B immediately. Medical therapy alone generally does not suffice, and aggressive surgical debridement combined with intravenous antifungal therapy results in better outcomes. The longer the duration of symptoms and the greater the progression of disease, the less favorable the prognosis.
The patient was started on amphotericin B lipid complex and micafungin. However, after 16 days of therapy, repeat imaging of the neck showed worsening necrosis of the neck muscles. At this time, she underwent extensive debridement of face and neck, and posaconazole was added. After prolonged hospitalization, she was discharged to a rehabilitation facility on posaconazole. She resided in a nursing facility for 6 months. One year after her hospitalization, she is living at home and is able to ambulate independently, but requires feeding through a percutaneous endoscopic gastrostomy (PEG) tube because she remains dysphagic.
COMMENTARY
Infections caused by the ubiquitous fungi of the class Zygomycetes typically take 1 of 5 forms: rhinocerebral, pulmonary, gastrointestinal, disseminated, and cutaneous. The presentation varies widely, ranging from plaques, skin swelling, pustules, cellulitis, blisters, nodules, ulcerations, and ecthyma gangrenosum‐like lesions to deeper infections such as necrotizing fasciitis, osteomyelitis, and disseminated infection.1 Infections typically occur in immunocompromised hosts, including transplant recipients and patients with hematologic malignancy, but also occur in patients with diabetes mellitus, intravenous drug users, and patients on deferoxamine therapy.2 Deferoxamine and other iron‐binding therapy is thought to predispose to zygomycetes infections because of improved iron uptake of the fungal species and, thus, stimulation of growth.3 Pulmonary and rhinocerebral infections are the most common clinically encountered forms, and 44% of cutaneous infections are complicated by deep extension or dissemination.4
The articles cited above describe the more typical presentations of this rare disease. However, this patient had an unusual presentation, as parotid involvement due to zygomycosis has only been described once previously.5 Her inflammatory vasculitis and ensuing strokes from involvement of the carotid artery are recognized complications of zygomycosis, and in 1 case series of 41 patients with rhinocerebral mucormycosis, carotid involvement was seen in 31% of patients.6 After the punch biopsy of the patient's pinna showing nonseptated hyphae reminiscent of Mucor, why did her physicians delay administering amphotericin?
There are 2 likely possibilities: anchoring bias or error in medical decision‐making due to inaccurate probability estimates. Anchoring bias describes a heuristic where the initial diagnosis or gestalt biases the physician's process for assigning a final diagnosis.7, 8 This bias creates cognitive errors by limiting creativity in diagnosis. In this case, the infectious disease team carefully weighed the information obtained from the first biopsy. Given their low pretest estimate of this virtually unreported presentation of a rare disease, they decided to evaluate further without beginning antifungal therapy. Of note, there were few hyaline structures, and those structures lacked uptake of GMS. Since they considered the diagnosis yet rejected the diagnosis due to insufficient evidence, it is unlikely that anchoring bias played a role.
Was there an error in medical decision‐making? The physicians in this case faced a very common medical dilemma: whether or not to start a toxic medication empirically or wait for diagnostic confirmation prior to treatment.9 To solve this dilemma, one can apply decision analysis. Moskowitz et al described 5 phases of medical decision analysis by which a probabilistic right answer to clinical scenarios can be deduced mathematically.10 To solve this problem, probabilities must be assigned to the risk of giving a drug to a patient without the disease versus the risk of not giving a drug to a patient with the disease. For example, amphotericin deoxycholate causes acute renal failure in 30% to 47% of patients. Newer formulations of amphotericin, such as liposomal amphotericin and lipid complex, result in lower rates of nephrotoxicity (27% vs 47%). The risk of not giving amphotericin to a patient with zygomycosis is death. Even in patients treated with amphotericin, the mortality rate has been shown to be 66%, and up to 100% in those with strokes related to zygomycosis.2, 6, 11 Simply looking at these probabilities, decision analysis would favor empiric treatment.
The physicians caring for this patient did not have the luxury of retrospective speculation. After looking at all of the data, the equivocal skin biopsy and rare clinical presentation, the question to ask would change: What is the risk of giving amphotericin empirically to someone who, based on available information, has a very low probability of having zygomycosis? When phrased in this manner, there is a 47% chance of nephrotoxicity with amphotericin versus the very small probability that you have diagnosed a case of zygomycosis that has only been described once in the literature. Mathematically andmore importantlyclinically, this question becomes more difficult to answer. However, no value can be placed on the possibility of death in suspected zygomycosis, and the risk of short‐term amphotericin use is much less than that of a course of treatment. As such, empiric therapy should always be given.
Physicians are not mathematicians, and dynamic clinical scenarios are not so easily made into static math problems. Disease presentations evolve over time towards a diagnosable clinical pattern, as was the case with this patient. Two days after the aforementioned biopsy, she worsened and in less time than it would have taken to isolate zygomycosis from the first biopsy, a second biopsy revealed the typical nonseptated hyphae demarcated with the GMS stain. Even appropriate diagnostic testing, thoughtful interpretation, and avoidance of certain cognitive errors can result in incorrect diagnoses and delayed treatment. It is monitoring the progression of disease and collecting additional data that allows physicians to mold a diagnosis and create a treatment plan.
The primary treatment of zygomycosis should include amphotericin. However, there are limited data to support combination therapy with an echinocandin in severe cases, as in this patient.12 Posaconazole is not recommended for monotherapy as an initial therapy, but there is data for its use as salvage therapy in zygomycosis.13 This case highlights the difficulties that physicians face in the diagnosis and treatment of rare diseases. Cerebral infarction in a hematologic malignancy, uncontrolled diabetes, or iron chelation therapy could be the initial presentation of rhinocerebral zygomycosis. There truly are different strokes for different folks. Recognizing this and similar presentations may lead to a more rapid diagnosis and treatment of zygomycosis.
TEACHING POINTS
-
Zygomycosis has a wide range of clinical presentations ranging from skin lesions to deep tissue infections. As it is an angioinvasive organism, it can also present as cerebral infarcts and brain abscesses.
-
Zygomycosis infections should be suspected in patients with uncontrolled diabetes, hematologic or oncologic malignancies, and patients on iron chelation therapy with a potentially compatible clinical picture.
-
If zygomycosis infection is suspected, rapid histologic diagnosis should be attempted. However, as histologic diagnosis can take time, empiric therapy with amphotericin should always be administered.
-
Amphotericin remains the primary medical therapy for this disease; however, there is limited emerging evidence to suggest that echinocandins can be used in combination with amphotericin for improved treatment of severe rhinocerebral zygomyocosis. Posaconazole has a role as salvage therapy in zygomycosis, but should not be used as the sole primary treatment.
The approach to clinical conundrums by an expert clinician is revealed through the presentation of an actual patient's case in an approach typical of a morning report. Similarly to patient care, sequential pieces of information are provided to the clinician, who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
This icon represents the patient's case. Each paragraph that follows represents the discussant's thoughts.
Acknowledgements
The authors are indebted to Dr Glenn Roberson at the Department of Radiology, University of Alabama at Birmingham, for providing the radiographic images; to Dr Aleodor Andea at the Department of Pathology, University of Alabama at Birmingham, for providing the pathology images; and to Dr. Crysten Brinkley at the Department of Neurology at the University of Alabama at Birmingham for her assistance with this case presentation.
Disclosure: Nothing to report.
- ,,,.Mucormycosis: emerging prominence of cutaneous infections.Clin Infect Dis.1994;19:67–76.
- ,,,.Zygomycosis in the 1990s in a tertiary‐care cancer center.Clin Infect Dis.2000;30:851–856.
- ,,, et al.Mucormycosis during deferoxamine therapy is a siderophore‐mediated infection. In vitro and in vivo animal studies.J Clin Invest.1993;91:1979–1986.
- ,,, et al.Epidemiology and outcome of zygomycosis: a review of 929 reported cases.Clin Infect Dis.2005;41:634–653.
- ,,,,,.Cutaneous mucormycosis of the head and neck with parotid gland involvement: first report of a case.Ear Nose Throat J.2004;83:282–286.
- ,,,,,.A successful combined endovascular and surgical treatment of a cranial base mucormycosis with an associated internal carotid artery pseudoaneurysm.Neurosurgery.2009;65:733–740.
- ,.Judgment under uncertainty: heuristics and biases.Science.1974;185:1124–1131.
- ,,,,.Clinical problem‐solving. Anchors away.N Engl J Med.2007;356:504–509.
- ,,,.Clinical problem‐solving. Empirically incorrect.N Engl J Med.2006;354:509–514.
- ,,.Dealing with uncertainty, risks, and tradeoffs in clinical decisions. A cognitive science approach.Ann Intern Med.1988;108:435–449.
- ,,.Fatal strokes in patients with rhino‐orbito‐cerebral mucormycosis and associated vasculopathy.Scand J Infect Dis.2004;36:643–648.
- ,,, et al.Combination polyene‐caspofungin treatment of rhino‐orbital‐cerebral mucormycosis.Clin Infect Dis.2008;47:364–371.
- ,,,,.Posaconazole is effective as salvage therapy in zygomycosis: a retrospective summary of 91 cases.Clin Infect Dis.2006;42:e61–e65.
- ,,,.Mucormycosis: emerging prominence of cutaneous infections.Clin Infect Dis.1994;19:67–76.
- ,,,.Zygomycosis in the 1990s in a tertiary‐care cancer center.Clin Infect Dis.2000;30:851–856.
- ,,, et al.Mucormycosis during deferoxamine therapy is a siderophore‐mediated infection. In vitro and in vivo animal studies.J Clin Invest.1993;91:1979–1986.
- ,,, et al.Epidemiology and outcome of zygomycosis: a review of 929 reported cases.Clin Infect Dis.2005;41:634–653.
- ,,,,,.Cutaneous mucormycosis of the head and neck with parotid gland involvement: first report of a case.Ear Nose Throat J.2004;83:282–286.
- ,,,,,.A successful combined endovascular and surgical treatment of a cranial base mucormycosis with an associated internal carotid artery pseudoaneurysm.Neurosurgery.2009;65:733–740.
- ,.Judgment under uncertainty: heuristics and biases.Science.1974;185:1124–1131.
- ,,,,.Clinical problem‐solving. Anchors away.N Engl J Med.2007;356:504–509.
- ,,,.Clinical problem‐solving. Empirically incorrect.N Engl J Med.2006;354:509–514.
- ,,.Dealing with uncertainty, risks, and tradeoffs in clinical decisions. A cognitive science approach.Ann Intern Med.1988;108:435–449.
- ,,.Fatal strokes in patients with rhino‐orbito‐cerebral mucormycosis and associated vasculopathy.Scand J Infect Dis.2004;36:643–648.
- ,,, et al.Combination polyene‐caspofungin treatment of rhino‐orbital‐cerebral mucormycosis.Clin Infect Dis.2008;47:364–371.
- ,,,,.Posaconazole is effective as salvage therapy in zygomycosis: a retrospective summary of 91 cases.Clin Infect Dis.2006;42:e61–e65.
ONLINE EXCLUSIVE: Listen to a senior research scholar Mary Gatta, PhD, discuss why gender pay gaps continue to exist
Click here to listen to Dr. Gatta
Click here to listen to Dr. Gatta
Click here to listen to Dr. Gatta
ONLINE EXCLUSIVE: Listen to Bob Wachter, Norm Orenstein, and Michael Pistoria preview HM12 keynote addresses
Click here to listen to Dr. Wachter
Click here to listen to Dr. Pistoria
Click here to listen to Norm Orenstein
Click here to listen to Dr. Wachter
Click here to listen to Dr. Pistoria
Click here to listen to Norm Orenstein
Click here to listen to Dr. Wachter
Click here to listen to Dr. Pistoria
Click here to listen to Norm Orenstein
ONLINE EXCLUSIVE: Hospitalists turned C-Suiters tell their secrets
Click here to listen to Dr. Sperring
Click here to listen to Dr. Sperring
Click here to listen to Dr. Sperring
Dabigatran
To the Editor: I read with interest the review of dabigatran (Pradaxa) by Drs. Wartak and Bartholomew, which provides an excellent overview of this new oral anticoagulant.1
This article does not mention clearly two key points about the guidelines for using dabigatran, which are different in the United States than in the other 75 countries where it has been approved.1 First, the RE-LY trial2,3 excluded patients with a creatinine clearance rate less than 30 mL/min/1.73 m2, a common situation in the elderly. Second, in contrast to other countries, the US Food and Drug Administration (FDA) approved dabigatran for patients with a creatinine clearance rate of 15 to 30 mL/min/1.73 m2, although at a lower dose.3 No dose adjustment is suggested in patients with less severe (mild or moderate) renal impairment.3 This may lead to potential misuse and problems. In fact, lethal side effects have been reported in France by Legrand et al.4 Furthermore, a report is in press on dabigatran-associated acute renal failure,5 and recently the German publication Die Zeit reported 50 deaths from bleeding in patients with atrial fibrillation treated with dabigatran.6
Therefore, despite suggestions that dabigatran does not require monitoring of its effects during treatment,1,3 renal, hematologic, and hepatic variables should be monitored before and after initiation of dabigatran5 until more experience is gained with this new drug, and especially in the elderly and those with chronic kidney disease that is stage 4 (estimated glomerular filtration rate 15–29 mL/min/1.73 m2) or stage 5 (< 15 mL/min/1.73 m2).
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Connolly SJ, Ezekowitz MD, Yusuf S, et al., RE-LY Steering Committee and Investigators. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361:1139–1151.
- Pradaxa (prescribing information). Ridgefield, CT. Boehringer Ingelheim Pharmaceuticals, Inc, 2011.
- Legrand M, Mateo J, Aribaud A, et al. The use of dabigatran in elderly patients. Arch Intern Med 2011; 171:1285–1286.
- Pazmiño PA. Dabigatran-associated acute renal failure (DAARF). El Paso Physician 2011. In press.
- Kaiser C. Pradaxa bleeding deaths raise concern. MedPageToday. www.medpagetoday.com/Cardiology/Strokes/29411. Accessed February 9, 2012.
To the Editor: I read with interest the review of dabigatran (Pradaxa) by Drs. Wartak and Bartholomew, which provides an excellent overview of this new oral anticoagulant.1
This article does not mention clearly two key points about the guidelines for using dabigatran, which are different in the United States than in the other 75 countries where it has been approved.1 First, the RE-LY trial2,3 excluded patients with a creatinine clearance rate less than 30 mL/min/1.73 m2, a common situation in the elderly. Second, in contrast to other countries, the US Food and Drug Administration (FDA) approved dabigatran for patients with a creatinine clearance rate of 15 to 30 mL/min/1.73 m2, although at a lower dose.3 No dose adjustment is suggested in patients with less severe (mild or moderate) renal impairment.3 This may lead to potential misuse and problems. In fact, lethal side effects have been reported in France by Legrand et al.4 Furthermore, a report is in press on dabigatran-associated acute renal failure,5 and recently the German publication Die Zeit reported 50 deaths from bleeding in patients with atrial fibrillation treated with dabigatran.6
Therefore, despite suggestions that dabigatran does not require monitoring of its effects during treatment,1,3 renal, hematologic, and hepatic variables should be monitored before and after initiation of dabigatran5 until more experience is gained with this new drug, and especially in the elderly and those with chronic kidney disease that is stage 4 (estimated glomerular filtration rate 15–29 mL/min/1.73 m2) or stage 5 (< 15 mL/min/1.73 m2).
To the Editor: I read with interest the review of dabigatran (Pradaxa) by Drs. Wartak and Bartholomew, which provides an excellent overview of this new oral anticoagulant.1
This article does not mention clearly two key points about the guidelines for using dabigatran, which are different in the United States than in the other 75 countries where it has been approved.1 First, the RE-LY trial2,3 excluded patients with a creatinine clearance rate less than 30 mL/min/1.73 m2, a common situation in the elderly. Second, in contrast to other countries, the US Food and Drug Administration (FDA) approved dabigatran for patients with a creatinine clearance rate of 15 to 30 mL/min/1.73 m2, although at a lower dose.3 No dose adjustment is suggested in patients with less severe (mild or moderate) renal impairment.3 This may lead to potential misuse and problems. In fact, lethal side effects have been reported in France by Legrand et al.4 Furthermore, a report is in press on dabigatran-associated acute renal failure,5 and recently the German publication Die Zeit reported 50 deaths from bleeding in patients with atrial fibrillation treated with dabigatran.6
Therefore, despite suggestions that dabigatran does not require monitoring of its effects during treatment,1,3 renal, hematologic, and hepatic variables should be monitored before and after initiation of dabigatran5 until more experience is gained with this new drug, and especially in the elderly and those with chronic kidney disease that is stage 4 (estimated glomerular filtration rate 15–29 mL/min/1.73 m2) or stage 5 (< 15 mL/min/1.73 m2).
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Connolly SJ, Ezekowitz MD, Yusuf S, et al., RE-LY Steering Committee and Investigators. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361:1139–1151.
- Pradaxa (prescribing information). Ridgefield, CT. Boehringer Ingelheim Pharmaceuticals, Inc, 2011.
- Legrand M, Mateo J, Aribaud A, et al. The use of dabigatran in elderly patients. Arch Intern Med 2011; 171:1285–1286.
- Pazmiño PA. Dabigatran-associated acute renal failure (DAARF). El Paso Physician 2011. In press.
- Kaiser C. Pradaxa bleeding deaths raise concern. MedPageToday. www.medpagetoday.com/Cardiology/Strokes/29411. Accessed February 9, 2012.
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Connolly SJ, Ezekowitz MD, Yusuf S, et al., RE-LY Steering Committee and Investigators. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361:1139–1151.
- Pradaxa (prescribing information). Ridgefield, CT. Boehringer Ingelheim Pharmaceuticals, Inc, 2011.
- Legrand M, Mateo J, Aribaud A, et al. The use of dabigatran in elderly patients. Arch Intern Med 2011; 171:1285–1286.
- Pazmiño PA. Dabigatran-associated acute renal failure (DAARF). El Paso Physician 2011. In press.
- Kaiser C. Pradaxa bleeding deaths raise concern. MedPageToday. www.medpagetoday.com/Cardiology/Strokes/29411. Accessed February 9, 2012.
In reply: Dabigatran
In Reply: Dabigatran has gained significant popularity in the United States. From its approval in October 2010 and through August 2011, approximately 1.1 million prescriptions for it were dispensed, and 371,000 patients received it from US outpatient retail pharmacies.1 We appreciate the letters from Drs. Pazmiño and Hirsch and believe there are reasons to be vigilant when using dabigatran.
In response to the letter from Dr. Pazmiño, we agree with his concerns and have covered them in our review. We would like to emphasize that our review was intended to help US clinicians understand this new drug and was not restricted to the RE-LY trial. The limitations of the trials of dabigatran to date (including the lack of patients with renal impairment in the RE-LY trial) have been mentioned in many articles, including ours. Please see the section TOPICS OF FUTURE INTEREST.2
The FDA did not recommend any dose adjustment for patients with moderate renal impairment (creatinine clearance 30–50 mL/min), as it was convinced that the 150-mg dose had a superior risk-benefit profile, even for patients with a higher risk of bleeding, compared with the 110-mg dose.3 It is hard for us to comment on the specific reasoning behind the FDA’s approval for using 75 mg of dabigatran in patients with creatinine clearance between 15 and 30 mL/min. However, we know this was based on pharmacokinetic and pharmacodynamic modeling and not on efficacy and safety data.3 With respect to dosing and monitoring, we did stress this point in our article, stating that the use of dabigatran obviates the need for routine laboratory monitoring. However, one may measure the drug’s activity in certain situations (suspected overdose, bleeding, need for emergency surgery, impaired renal function, pregnancy, and obesity, and in children). Please see the section DOES DABIGATRAN NEED MONITORING? CAN IT EVEN BE MONITORED? in our review.2
Dr. Pazmiño suggests renal, hematologic and hepatic variables should be monitored before and after starting dabigatran. We agree that renal function should be monitored and have covered this point. Please see the section WHO SHOULD NOT RECEIVE DABIGATRAN.2 Hematologic and hepatic variables can be monitored if a clinician decides to do so, but this is not limited specifically to dabigatran. Also, to clarify, dabigatran is not approved for those with stage 5 chronic kidney disease. And we share his concern about the lack of experience with this new drug, and we included a word of caution in the section ADVANTAGES AND DISADVANTAGES OF DABIGATRAN.2
We agree with Dr. Hirsch’s concerns about recombinant factor VIIa. We are not recommending its use as a routine practice but as an available option. Our article was a global review on dabigatran, and our aim was to cover the best available evidence and treatment options in a comprehensive way. However, in response to Dr. Hirsch’s comments, the systematic review by Yank et al4 drew its data from 16 randomized controlled trials but excluded patients on anticoagulants (except for those in a few observational studies), and factor VIIa was compared with placebo.4 So these findings are not applicable to patients with dabigatran-related bleeding, and to draw any definite conclusion would not be correct. If recombinant factor VIIa has failed to show a benefit in terms of a lower mortality rate, we could also point out that there was no mortality benefit seen in reversing warfarin anticoagulation in patients with acute intracranial hemorrhage with the use of vitamin K, fresh-frozen plasma, or prothrombin complex concentrate.5 This should not lead one to stop using these treatments.
Clinicians are well accustomed to managing warfarin- or heparin-related bleeding using specific antidotes. It is very important to understand the mechanism of action of dabigatran, and to realize that there is no antidote. Recombinant factor VIIa is a potent hemostatic agent, and there are many published case reports and case series highlighting its efficacy in preventing bleeding.6–12 It is used when all other options are exhausted. It is never a routine practice: it is always a last resort a clinician takes to prevent catastrophic bleeding. We believe economic concerns are very important, but it will be difficult to extrapolate a specific benchmark while treating for an individual case. At present, it seems unlikely that a randomized trial of recombinant factor VIIa will be conducted, and guidance is to be based on available animal studies and clinical anecdotes. A recent review on reversing anticoagulation therapy13 proposes treating major bleeding complications of direct thrombin inhibitors with activated prothrombin complex and recombinant factor VIIa.13
We acknowledge that serious, even fatal bleeding events have been reported with dabigatran. The FDA is evaluating postmarketing reports and is also using an active surveillance system to compare new users of dabigatran and warfarin with respect to the likelihood of their being hospitalized for bleeding.1 With time and experience, we will learn more.
Finally, as with any new drug, the absence of data on long-term safety and efficacy is an important issue and should be considered when prescribing this new medication.
- US Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): drug safety communication—safety review of post-marketing reports of serious bleeding events. http://www.fda.gov/Safety/MedWatch/Safety-Information/SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed February 8, 2012.
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Beasley BN, Unger EF, Temple R. Anticoagulant options—why the FDA approved a higher but not a lower dose of dabigatran. N Engl J Med 2011; 364:1788–1790.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Bechtel BF, Nunez TC, Lyon JA, Cotton BA, Barrett TW. Treatments for reversing warfarin anticoagulation in patients with acute intracranial hemorrhage: a structured literature review. Int J Emerg Med 2011; 4:40.
- Warren O, Mandal K, Hadjianastassiou V, et al. Recombinant activated factor VII in cardiac surgery: a systematic review. Ann Thorac Surg 2007; 83:707–714.
- Chapman AJ, Blount AL, Davis AT, Hooker RL. Recombinant factor VIIa (NovoSeven RT) use in high risk cardiac surgery. Eur J Cardiothorac Surg 2011; 40:1314–1318; discussion 1318–1319.
- Vavra KA, Lutz MF, Smythe MA. Recombinant factor VIIa to manage major bleeding from newer parenteral anticoagulants. Ann Pharmacother 2010; 44:718–726.
- Ilyas C, Beyer GM, Dutton RP, Scalea TM, Hess JR. Recombinant factor VIIa for warfarin-associated intracranial bleeding. J Clin Anesth 2008; 20:276–279.
- Brody DL, Aiyagari V, Shackleford AM, Diringer MN. Use of recombinant factor VIIa in patients with warfarin-associated intracranial hemorrhage. Neurocrit Care 2005; 2:263–267.
- Nagle EL, Tsu LV, Dager WE. Bivalirudin for anticoagulation during hypothermic cardiopulmonary bypass and recombinant factor VIIa for iatrogenic coagulopathy. Ann Pharmacother 2011; 45:e47.
- Kobayashi T, Nakabayashi M, Yoshioka A, Maeda M, Ikenoue T. Recombinant activated factor VII (rFVIIa/NovoSeven(®)) in the management of severe postpartum haemorrhage: initial report of a multicentre case series in Japan. Int J Hematol 2011; 95:57–63.
- Ghanny S, Warkentin TE, Crowther MA. Reversing anticoagulant
therapy. Curr Drug Discov Technol 2011; Oct 21 (epub ahead of print).
In Reply: Dabigatran has gained significant popularity in the United States. From its approval in October 2010 and through August 2011, approximately 1.1 million prescriptions for it were dispensed, and 371,000 patients received it from US outpatient retail pharmacies.1 We appreciate the letters from Drs. Pazmiño and Hirsch and believe there are reasons to be vigilant when using dabigatran.
In response to the letter from Dr. Pazmiño, we agree with his concerns and have covered them in our review. We would like to emphasize that our review was intended to help US clinicians understand this new drug and was not restricted to the RE-LY trial. The limitations of the trials of dabigatran to date (including the lack of patients with renal impairment in the RE-LY trial) have been mentioned in many articles, including ours. Please see the section TOPICS OF FUTURE INTEREST.2
The FDA did not recommend any dose adjustment for patients with moderate renal impairment (creatinine clearance 30–50 mL/min), as it was convinced that the 150-mg dose had a superior risk-benefit profile, even for patients with a higher risk of bleeding, compared with the 110-mg dose.3 It is hard for us to comment on the specific reasoning behind the FDA’s approval for using 75 mg of dabigatran in patients with creatinine clearance between 15 and 30 mL/min. However, we know this was based on pharmacokinetic and pharmacodynamic modeling and not on efficacy and safety data.3 With respect to dosing and monitoring, we did stress this point in our article, stating that the use of dabigatran obviates the need for routine laboratory monitoring. However, one may measure the drug’s activity in certain situations (suspected overdose, bleeding, need for emergency surgery, impaired renal function, pregnancy, and obesity, and in children). Please see the section DOES DABIGATRAN NEED MONITORING? CAN IT EVEN BE MONITORED? in our review.2
Dr. Pazmiño suggests renal, hematologic and hepatic variables should be monitored before and after starting dabigatran. We agree that renal function should be monitored and have covered this point. Please see the section WHO SHOULD NOT RECEIVE DABIGATRAN.2 Hematologic and hepatic variables can be monitored if a clinician decides to do so, but this is not limited specifically to dabigatran. Also, to clarify, dabigatran is not approved for those with stage 5 chronic kidney disease. And we share his concern about the lack of experience with this new drug, and we included a word of caution in the section ADVANTAGES AND DISADVANTAGES OF DABIGATRAN.2
We agree with Dr. Hirsch’s concerns about recombinant factor VIIa. We are not recommending its use as a routine practice but as an available option. Our article was a global review on dabigatran, and our aim was to cover the best available evidence and treatment options in a comprehensive way. However, in response to Dr. Hirsch’s comments, the systematic review by Yank et al4 drew its data from 16 randomized controlled trials but excluded patients on anticoagulants (except for those in a few observational studies), and factor VIIa was compared with placebo.4 So these findings are not applicable to patients with dabigatran-related bleeding, and to draw any definite conclusion would not be correct. If recombinant factor VIIa has failed to show a benefit in terms of a lower mortality rate, we could also point out that there was no mortality benefit seen in reversing warfarin anticoagulation in patients with acute intracranial hemorrhage with the use of vitamin K, fresh-frozen plasma, or prothrombin complex concentrate.5 This should not lead one to stop using these treatments.
Clinicians are well accustomed to managing warfarin- or heparin-related bleeding using specific antidotes. It is very important to understand the mechanism of action of dabigatran, and to realize that there is no antidote. Recombinant factor VIIa is a potent hemostatic agent, and there are many published case reports and case series highlighting its efficacy in preventing bleeding.6–12 It is used when all other options are exhausted. It is never a routine practice: it is always a last resort a clinician takes to prevent catastrophic bleeding. We believe economic concerns are very important, but it will be difficult to extrapolate a specific benchmark while treating for an individual case. At present, it seems unlikely that a randomized trial of recombinant factor VIIa will be conducted, and guidance is to be based on available animal studies and clinical anecdotes. A recent review on reversing anticoagulation therapy13 proposes treating major bleeding complications of direct thrombin inhibitors with activated prothrombin complex and recombinant factor VIIa.13
We acknowledge that serious, even fatal bleeding events have been reported with dabigatran. The FDA is evaluating postmarketing reports and is also using an active surveillance system to compare new users of dabigatran and warfarin with respect to the likelihood of their being hospitalized for bleeding.1 With time and experience, we will learn more.
Finally, as with any new drug, the absence of data on long-term safety and efficacy is an important issue and should be considered when prescribing this new medication.
In Reply: Dabigatran has gained significant popularity in the United States. From its approval in October 2010 and through August 2011, approximately 1.1 million prescriptions for it were dispensed, and 371,000 patients received it from US outpatient retail pharmacies.1 We appreciate the letters from Drs. Pazmiño and Hirsch and believe there are reasons to be vigilant when using dabigatran.
In response to the letter from Dr. Pazmiño, we agree with his concerns and have covered them in our review. We would like to emphasize that our review was intended to help US clinicians understand this new drug and was not restricted to the RE-LY trial. The limitations of the trials of dabigatran to date (including the lack of patients with renal impairment in the RE-LY trial) have been mentioned in many articles, including ours. Please see the section TOPICS OF FUTURE INTEREST.2
The FDA did not recommend any dose adjustment for patients with moderate renal impairment (creatinine clearance 30–50 mL/min), as it was convinced that the 150-mg dose had a superior risk-benefit profile, even for patients with a higher risk of bleeding, compared with the 110-mg dose.3 It is hard for us to comment on the specific reasoning behind the FDA’s approval for using 75 mg of dabigatran in patients with creatinine clearance between 15 and 30 mL/min. However, we know this was based on pharmacokinetic and pharmacodynamic modeling and not on efficacy and safety data.3 With respect to dosing and monitoring, we did stress this point in our article, stating that the use of dabigatran obviates the need for routine laboratory monitoring. However, one may measure the drug’s activity in certain situations (suspected overdose, bleeding, need for emergency surgery, impaired renal function, pregnancy, and obesity, and in children). Please see the section DOES DABIGATRAN NEED MONITORING? CAN IT EVEN BE MONITORED? in our review.2
Dr. Pazmiño suggests renal, hematologic and hepatic variables should be monitored before and after starting dabigatran. We agree that renal function should be monitored and have covered this point. Please see the section WHO SHOULD NOT RECEIVE DABIGATRAN.2 Hematologic and hepatic variables can be monitored if a clinician decides to do so, but this is not limited specifically to dabigatran. Also, to clarify, dabigatran is not approved for those with stage 5 chronic kidney disease. And we share his concern about the lack of experience with this new drug, and we included a word of caution in the section ADVANTAGES AND DISADVANTAGES OF DABIGATRAN.2
We agree with Dr. Hirsch’s concerns about recombinant factor VIIa. We are not recommending its use as a routine practice but as an available option. Our article was a global review on dabigatran, and our aim was to cover the best available evidence and treatment options in a comprehensive way. However, in response to Dr. Hirsch’s comments, the systematic review by Yank et al4 drew its data from 16 randomized controlled trials but excluded patients on anticoagulants (except for those in a few observational studies), and factor VIIa was compared with placebo.4 So these findings are not applicable to patients with dabigatran-related bleeding, and to draw any definite conclusion would not be correct. If recombinant factor VIIa has failed to show a benefit in terms of a lower mortality rate, we could also point out that there was no mortality benefit seen in reversing warfarin anticoagulation in patients with acute intracranial hemorrhage with the use of vitamin K, fresh-frozen plasma, or prothrombin complex concentrate.5 This should not lead one to stop using these treatments.
Clinicians are well accustomed to managing warfarin- or heparin-related bleeding using specific antidotes. It is very important to understand the mechanism of action of dabigatran, and to realize that there is no antidote. Recombinant factor VIIa is a potent hemostatic agent, and there are many published case reports and case series highlighting its efficacy in preventing bleeding.6–12 It is used when all other options are exhausted. It is never a routine practice: it is always a last resort a clinician takes to prevent catastrophic bleeding. We believe economic concerns are very important, but it will be difficult to extrapolate a specific benchmark while treating for an individual case. At present, it seems unlikely that a randomized trial of recombinant factor VIIa will be conducted, and guidance is to be based on available animal studies and clinical anecdotes. A recent review on reversing anticoagulation therapy13 proposes treating major bleeding complications of direct thrombin inhibitors with activated prothrombin complex and recombinant factor VIIa.13
We acknowledge that serious, even fatal bleeding events have been reported with dabigatran. The FDA is evaluating postmarketing reports and is also using an active surveillance system to compare new users of dabigatran and warfarin with respect to the likelihood of their being hospitalized for bleeding.1 With time and experience, we will learn more.
Finally, as with any new drug, the absence of data on long-term safety and efficacy is an important issue and should be considered when prescribing this new medication.
- US Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): drug safety communication—safety review of post-marketing reports of serious bleeding events. http://www.fda.gov/Safety/MedWatch/Safety-Information/SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed February 8, 2012.
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Beasley BN, Unger EF, Temple R. Anticoagulant options—why the FDA approved a higher but not a lower dose of dabigatran. N Engl J Med 2011; 364:1788–1790.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Bechtel BF, Nunez TC, Lyon JA, Cotton BA, Barrett TW. Treatments for reversing warfarin anticoagulation in patients with acute intracranial hemorrhage: a structured literature review. Int J Emerg Med 2011; 4:40.
- Warren O, Mandal K, Hadjianastassiou V, et al. Recombinant activated factor VII in cardiac surgery: a systematic review. Ann Thorac Surg 2007; 83:707–714.
- Chapman AJ, Blount AL, Davis AT, Hooker RL. Recombinant factor VIIa (NovoSeven RT) use in high risk cardiac surgery. Eur J Cardiothorac Surg 2011; 40:1314–1318; discussion 1318–1319.
- Vavra KA, Lutz MF, Smythe MA. Recombinant factor VIIa to manage major bleeding from newer parenteral anticoagulants. Ann Pharmacother 2010; 44:718–726.
- Ilyas C, Beyer GM, Dutton RP, Scalea TM, Hess JR. Recombinant factor VIIa for warfarin-associated intracranial bleeding. J Clin Anesth 2008; 20:276–279.
- Brody DL, Aiyagari V, Shackleford AM, Diringer MN. Use of recombinant factor VIIa in patients with warfarin-associated intracranial hemorrhage. Neurocrit Care 2005; 2:263–267.
- Nagle EL, Tsu LV, Dager WE. Bivalirudin for anticoagulation during hypothermic cardiopulmonary bypass and recombinant factor VIIa for iatrogenic coagulopathy. Ann Pharmacother 2011; 45:e47.
- Kobayashi T, Nakabayashi M, Yoshioka A, Maeda M, Ikenoue T. Recombinant activated factor VII (rFVIIa/NovoSeven(®)) in the management of severe postpartum haemorrhage: initial report of a multicentre case series in Japan. Int J Hematol 2011; 95:57–63.
- Ghanny S, Warkentin TE, Crowther MA. Reversing anticoagulant
therapy. Curr Drug Discov Technol 2011; Oct 21 (epub ahead of print).
- US Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): drug safety communication—safety review of post-marketing reports of serious bleeding events. http://www.fda.gov/Safety/MedWatch/Safety-Information/SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed February 8, 2012.
- Wartak SA, Bartholomew JR. Dabigatran: will it change clinical practice? Cleve Clin J Med 2011; 78:657–664.
- Beasley BN, Unger EF, Temple R. Anticoagulant options—why the FDA approved a higher but not a lower dose of dabigatran. N Engl J Med 2011; 364:1788–1790.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Bechtel BF, Nunez TC, Lyon JA, Cotton BA, Barrett TW. Treatments for reversing warfarin anticoagulation in patients with acute intracranial hemorrhage: a structured literature review. Int J Emerg Med 2011; 4:40.
- Warren O, Mandal K, Hadjianastassiou V, et al. Recombinant activated factor VII in cardiac surgery: a systematic review. Ann Thorac Surg 2007; 83:707–714.
- Chapman AJ, Blount AL, Davis AT, Hooker RL. Recombinant factor VIIa (NovoSeven RT) use in high risk cardiac surgery. Eur J Cardiothorac Surg 2011; 40:1314–1318; discussion 1318–1319.
- Vavra KA, Lutz MF, Smythe MA. Recombinant factor VIIa to manage major bleeding from newer parenteral anticoagulants. Ann Pharmacother 2010; 44:718–726.
- Ilyas C, Beyer GM, Dutton RP, Scalea TM, Hess JR. Recombinant factor VIIa for warfarin-associated intracranial bleeding. J Clin Anesth 2008; 20:276–279.
- Brody DL, Aiyagari V, Shackleford AM, Diringer MN. Use of recombinant factor VIIa in patients with warfarin-associated intracranial hemorrhage. Neurocrit Care 2005; 2:263–267.
- Nagle EL, Tsu LV, Dager WE. Bivalirudin for anticoagulation during hypothermic cardiopulmonary bypass and recombinant factor VIIa for iatrogenic coagulopathy. Ann Pharmacother 2011; 45:e47.
- Kobayashi T, Nakabayashi M, Yoshioka A, Maeda M, Ikenoue T. Recombinant activated factor VII (rFVIIa/NovoSeven(®)) in the management of severe postpartum haemorrhage: initial report of a multicentre case series in Japan. Int J Hematol 2011; 95:57–63.
- Ghanny S, Warkentin TE, Crowther MA. Reversing anticoagulant
therapy. Curr Drug Discov Technol 2011; Oct 21 (epub ahead of print).
Report reveals detailed data of ruxolitinib in MF
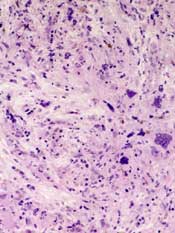
Ruxolitinib can ease the symptoms of myelofibrosis and improve patient survival, according to results of the COMFORT-I study.
Detailed data from this phase 3 trial, which led to the US approval of ruxolitinib in November, appear in the March 1 edition of The New England Journal of Medicine. Some of these data were also presented at the recent ASH meeting.
“The phase 1/2 clinical trial showed that ruxolitinib improves quality of life for many patients with myelofibrosis, and now this phase 3 study indicates that the drug extends survival in a patient population that has lacked effective treatments,” said principal investigator Srdan Verstovsek, MD, PhD, of The University of Texas MD Anderson Cancer Center.
The trial, which was funded by the maker of ruxolitinib, enrolled 309 patients from 89 US centers. The researchers randomized patients to receive ruxolitinib (n=155) or placebo (n=154) orally twice daily.
Patients recorded their symptoms over the course of the 24-week study using an electronic diary. And the investigators monitored patients’ spleen volumes via MRI. The primary endpoint of the study was a 35% reduction in spleen volume.
Most patients on placebo experienced progressive splenomegaly and a worsening of myelofibrosis-related symptoms. For this reason, 38 patients in the placebo arm withdrew from the study, and 111 patients crossed over to the ruxolitinib arm.
The median time to cross over was 41 weeks. Only 2 patients remained on placebo at the time of analysis.
Reduction in splenomegaly
“It quickly became apparent who was getting the placebo and who was getting the drug,” said study author Jason Gotlib, MD, of Stanford University Medical Center.
Dr Gotlib noted that the spleen volume in patients receiving the drug began to decrease within 1 to 2 weeks. In fact, 41.9% of treated patients experienced at least 35% shrinkage in their spleen volume. And the spleen remained smaller in 67% of those responders for 48 weeks or longer.
In comparison, 0.7% of patients in the placebo group experienced spleen reduction by 35%. At week 24, the ruxolitinib group had an average reduction in spleen volume of 31.6%, while the placebo group experienced an average increase in spleen volume of 8.1%.
Symptom improvement
Every night, patients completed the Myelofibrosis Symptom Assessment Form, an electronic diary. They evaluated the intensity of night sweats, itching, abdominal discomfort, pain under the ribs on the left side, a feeling of fullness, muscle and bone pain, and inactivity.
In the treated group, 45.9% of patients reported a reduction of 50% or more in their total symptom score over 24 weeks, compared to 5.3% in the placebo group. In addition, treated patients gained weight, while patients in the placebo group lost weight.
Improvements usually occurred within the first 4 weeks of treatment and were not limited to patients who also experienced reduction in spleen size, Dr Verstovsek said.
Adverse events and other drawbacks
Nonhematologic adverse events occurred at similar rates in both treatment arms. And roughly 11% of patients in each arm withdrew from the study due to adverse events.
However, anemia and thrombocytopenia were more common among patients who received ruxolitinib. One patient discontinued the drug due to anemia and one due to thrombocytopenia.
Other shortcomings of the drug are that it failed to reverse bone marrow damage and did not have a lasting effect on splenomegaly. Although many patients maintained a smaller spleen volume for at least 48 weeks while on ruxolitinib, their spleens began to enlarge again if they stopped taking the drug.
“Ruxolitinib doesn’t cure the disease,” Dr Gotlib said. “But the degree of benefit is clinically meaningful and substantial and allows many patients to re-engage in their daily activities.”
Mysteries of survival and mechanism
At a median follow-up of 51 weeks, there had been 13 deaths (8.4%) in the ruxolitinib group, compared with 24 (15.7%) in the placebo group. This represents a nearly 50% reduction in mortality, but Dr Gotlib indicated that the long-term implications of these data are not clear.
Another puzzler is how, exactly, ruxolitinib works. The drug is an inhibitor of JAK1 and JAK2, but patients responded to the drug whether or not they had the JAK2V617F mutation.
“Ruxolitinib is inhibiting overactive JAK/STAT intracellular signaling pathway no matter what,” Dr Verstovsek said. ![]()

Ruxolitinib can ease the symptoms of myelofibrosis and improve patient survival, according to results of the COMFORT-I study.
Detailed data from this phase 3 trial, which led to the US approval of ruxolitinib in November, appear in the March 1 edition of The New England Journal of Medicine. Some of these data were also presented at the recent ASH meeting.
“The phase 1/2 clinical trial showed that ruxolitinib improves quality of life for many patients with myelofibrosis, and now this phase 3 study indicates that the drug extends survival in a patient population that has lacked effective treatments,” said principal investigator Srdan Verstovsek, MD, PhD, of The University of Texas MD Anderson Cancer Center.
The trial, which was funded by the maker of ruxolitinib, enrolled 309 patients from 89 US centers. The researchers randomized patients to receive ruxolitinib (n=155) or placebo (n=154) orally twice daily.
Patients recorded their symptoms over the course of the 24-week study using an electronic diary. And the investigators monitored patients’ spleen volumes via MRI. The primary endpoint of the study was a 35% reduction in spleen volume.
Most patients on placebo experienced progressive splenomegaly and a worsening of myelofibrosis-related symptoms. For this reason, 38 patients in the placebo arm withdrew from the study, and 111 patients crossed over to the ruxolitinib arm.
The median time to cross over was 41 weeks. Only 2 patients remained on placebo at the time of analysis.
Reduction in splenomegaly
“It quickly became apparent who was getting the placebo and who was getting the drug,” said study author Jason Gotlib, MD, of Stanford University Medical Center.
Dr Gotlib noted that the spleen volume in patients receiving the drug began to decrease within 1 to 2 weeks. In fact, 41.9% of treated patients experienced at least 35% shrinkage in their spleen volume. And the spleen remained smaller in 67% of those responders for 48 weeks or longer.
In comparison, 0.7% of patients in the placebo group experienced spleen reduction by 35%. At week 24, the ruxolitinib group had an average reduction in spleen volume of 31.6%, while the placebo group experienced an average increase in spleen volume of 8.1%.
Symptom improvement
Every night, patients completed the Myelofibrosis Symptom Assessment Form, an electronic diary. They evaluated the intensity of night sweats, itching, abdominal discomfort, pain under the ribs on the left side, a feeling of fullness, muscle and bone pain, and inactivity.
In the treated group, 45.9% of patients reported a reduction of 50% or more in their total symptom score over 24 weeks, compared to 5.3% in the placebo group. In addition, treated patients gained weight, while patients in the placebo group lost weight.
Improvements usually occurred within the first 4 weeks of treatment and were not limited to patients who also experienced reduction in spleen size, Dr Verstovsek said.
Adverse events and other drawbacks
Nonhematologic adverse events occurred at similar rates in both treatment arms. And roughly 11% of patients in each arm withdrew from the study due to adverse events.
However, anemia and thrombocytopenia were more common among patients who received ruxolitinib. One patient discontinued the drug due to anemia and one due to thrombocytopenia.
Other shortcomings of the drug are that it failed to reverse bone marrow damage and did not have a lasting effect on splenomegaly. Although many patients maintained a smaller spleen volume for at least 48 weeks while on ruxolitinib, their spleens began to enlarge again if they stopped taking the drug.
“Ruxolitinib doesn’t cure the disease,” Dr Gotlib said. “But the degree of benefit is clinically meaningful and substantial and allows many patients to re-engage in their daily activities.”
Mysteries of survival and mechanism
At a median follow-up of 51 weeks, there had been 13 deaths (8.4%) in the ruxolitinib group, compared with 24 (15.7%) in the placebo group. This represents a nearly 50% reduction in mortality, but Dr Gotlib indicated that the long-term implications of these data are not clear.
Another puzzler is how, exactly, ruxolitinib works. The drug is an inhibitor of JAK1 and JAK2, but patients responded to the drug whether or not they had the JAK2V617F mutation.
“Ruxolitinib is inhibiting overactive JAK/STAT intracellular signaling pathway no matter what,” Dr Verstovsek said. ![]()

Ruxolitinib can ease the symptoms of myelofibrosis and improve patient survival, according to results of the COMFORT-I study.
Detailed data from this phase 3 trial, which led to the US approval of ruxolitinib in November, appear in the March 1 edition of The New England Journal of Medicine. Some of these data were also presented at the recent ASH meeting.
“The phase 1/2 clinical trial showed that ruxolitinib improves quality of life for many patients with myelofibrosis, and now this phase 3 study indicates that the drug extends survival in a patient population that has lacked effective treatments,” said principal investigator Srdan Verstovsek, MD, PhD, of The University of Texas MD Anderson Cancer Center.
The trial, which was funded by the maker of ruxolitinib, enrolled 309 patients from 89 US centers. The researchers randomized patients to receive ruxolitinib (n=155) or placebo (n=154) orally twice daily.
Patients recorded their symptoms over the course of the 24-week study using an electronic diary. And the investigators monitored patients’ spleen volumes via MRI. The primary endpoint of the study was a 35% reduction in spleen volume.
Most patients on placebo experienced progressive splenomegaly and a worsening of myelofibrosis-related symptoms. For this reason, 38 patients in the placebo arm withdrew from the study, and 111 patients crossed over to the ruxolitinib arm.
The median time to cross over was 41 weeks. Only 2 patients remained on placebo at the time of analysis.
Reduction in splenomegaly
“It quickly became apparent who was getting the placebo and who was getting the drug,” said study author Jason Gotlib, MD, of Stanford University Medical Center.
Dr Gotlib noted that the spleen volume in patients receiving the drug began to decrease within 1 to 2 weeks. In fact, 41.9% of treated patients experienced at least 35% shrinkage in their spleen volume. And the spleen remained smaller in 67% of those responders for 48 weeks or longer.
In comparison, 0.7% of patients in the placebo group experienced spleen reduction by 35%. At week 24, the ruxolitinib group had an average reduction in spleen volume of 31.6%, while the placebo group experienced an average increase in spleen volume of 8.1%.
Symptom improvement
Every night, patients completed the Myelofibrosis Symptom Assessment Form, an electronic diary. They evaluated the intensity of night sweats, itching, abdominal discomfort, pain under the ribs on the left side, a feeling of fullness, muscle and bone pain, and inactivity.
In the treated group, 45.9% of patients reported a reduction of 50% or more in their total symptom score over 24 weeks, compared to 5.3% in the placebo group. In addition, treated patients gained weight, while patients in the placebo group lost weight.
Improvements usually occurred within the first 4 weeks of treatment and were not limited to patients who also experienced reduction in spleen size, Dr Verstovsek said.
Adverse events and other drawbacks
Nonhematologic adverse events occurred at similar rates in both treatment arms. And roughly 11% of patients in each arm withdrew from the study due to adverse events.
However, anemia and thrombocytopenia were more common among patients who received ruxolitinib. One patient discontinued the drug due to anemia and one due to thrombocytopenia.
Other shortcomings of the drug are that it failed to reverse bone marrow damage and did not have a lasting effect on splenomegaly. Although many patients maintained a smaller spleen volume for at least 48 weeks while on ruxolitinib, their spleens began to enlarge again if they stopped taking the drug.
“Ruxolitinib doesn’t cure the disease,” Dr Gotlib said. “But the degree of benefit is clinically meaningful and substantial and allows many patients to re-engage in their daily activities.”
Mysteries of survival and mechanism
At a median follow-up of 51 weeks, there had been 13 deaths (8.4%) in the ruxolitinib group, compared with 24 (15.7%) in the placebo group. This represents a nearly 50% reduction in mortality, but Dr Gotlib indicated that the long-term implications of these data are not clear.
Another puzzler is how, exactly, ruxolitinib works. The drug is an inhibitor of JAK1 and JAK2, but patients responded to the drug whether or not they had the JAK2V617F mutation.
“Ruxolitinib is inhibiting overactive JAK/STAT intracellular signaling pathway no matter what,” Dr Verstovsek said. ![]()
Protease inhibitors: Silver bullets for chronic hepatitis C infection?
The treatment of hepatitis c virus (HCV) infection is on the brink of major changes with the recent approval of the first direct-acting antiviral agents, the protease inhibitors boceprevir (Victrelis) and telaprevir (Incivek).
Both drugs were approved by the US Food and Drug Administration (FDA) Advisory Panel for Chronic Hepatitis C in May 2011 and are believed to significantly improve treatment outcomes for patients with HCV genotype 1 infection.
A MAJOR PUBLIC HEALTH PROBLEM
HCV infection is a major public health problem. Nearly 4 million people in the United States are infected.6,7 Most patients with acute HCV infection become chronically infected, and up to 25% eventually develop cirrhosis and its complications, making HCV infection the leading indication for liver transplantation.8–10
Chronic HCV infection has a large global impact, with 180 million people affected across all economic and social groups.11 The highest prevalence of HCV has been reported in Egypt (14%), in part due to the use of inadequately sterilized needles in mass programs to treat endemic schistosomiasis. In developed countries, hepatocellular carcinoma associated with HCV has the fastest growing cancer-related death rate.12
CURRENTLY, FEWER THAN 50% OF PATIENTS ARE CURED
The goal of HCV treatment is to eradicate the virus. However, most infected patients (especially in the United States and Europe) are infected with HCV genotype 1, which is the most difficult genotype to treat.
Successful treatment of HCV is defined as achieving a sustained virologic response—ie, the absence of detectable HCV RNA in the serum 24 weeks after completion of therapy. Once a sustained virologic response is achieved, lifetime “cure” of HCV infection is expected in more than 99% of patients.13
The current standard therapy for HCV, pegylated interferon plus ribavirin for 48 weeks, is effective in only 40% to 50% of patients with genotype 1 infection.14 Therefore, assessing predictors of response before starting treatment can help select patients who are most likely to benefit from therapy.
Viral factors associated with a sustained virologic response include HCV genotypes other than genotype 1 and a low baseline viral load.
Beneficial patient-related factors include younger age, nonblack ethnicity, low body weight (≤ 75 kg), low body mass index, absence of insulin resistance, and absence of advanced fibrosis or cirrhosis.
More recently, a single-nucleotide polymorphism near the interleukin 28B (IL28B) gene, coding for interferon lambda 3, was found to be associated with a twofold difference in the rates of sustained virologic response: patients with the favorable genotype CC were two times more likely to achieve a sustained virologic response than patients with the CT or TT genotypes.15–17
PROTEASE INHIBITORS: MECHANISM OF ACTION
NS3/4A protease inhibitors rely on the principle of end-product inhibition, in which the cleavage product of the protease (a peptide) acts to inhibit the enzyme activity; this is why they are called peptidomimetics. The active site of the NS3/4A protease is a shallow groove composed of three highly conserved amino acid residues, which may explain why protease inhibitors display high antiviral efficacy but pose a low barrier to the development of resistance.20
Protease inhibitors are prone to resistance
The development of viral resistance to protease inhibitors has been a major drawback to their use in patients with chronic HCV infection.21
HCV is a highly variable virus with many genetically distinct but closely related quasispecies circulating in the blood at any given time. Drug-resistant, mutated variants preexist within the patient’s quasispecies, but only in small quantities because of their lesser replication fitness compared with the wild-type virus.22 When direct-acting antiviral therapy is started, the quantity of the wild-type virus decreases and the mutated virus gains replication fitness. Using protease inhibitors as monotherapy selects resistant viral populations rapidly within a few days or weeks.
HCV subtypes 1a and 1b may have different resistance profiles. With genotype 1a, some resistance-associated amino acid substitutions require only one nucleotide change, but with genotype 1b, two nucleotide changes are needed, making resistance less frequent in patients with HCV genotype 1b.23
BOCEPREVIR
Boceprevir is a specific inhibitor of the HCV viral protease NS3/4A.
In phase 3 clinical trials, boceprevir 800 mg three times a day was used with pegylated interferon alfa-2b (PegIntron) 1.5 μg/kg/week and ribavirin (Rebetol) 600 to 1,400 mg daily according to body weight.
Before patients started taking boceprevir, they went through a 4-week lead-in phase, during which they received pegylated interferon and ribavirin. This schedule appeared to reduce the incidence of viral breakthrough in phase 2 trials, and it produced higher rates of sustained virologic response and lower relapse rates compared with triple therapy without a lead-in phase.
Rapid virologic response was defined as undetectable HCV RNA at week 4 of boceprevir therapy (week 8 of the whole regimen).
Boceprevir in previously untreated patients with HCV genotype 1: The SPRINT-2 trial
The Serine Protease Inhibitor Therapy 2 (SPRINT-2) trial1 included more than 1,000 previously untreated adults with HCV genotype 1 infection (938 nonblack patients and 159 black patients; two other nonblack patients did not receive any study drug and were not included in the analysis). In this double-blind trial, patients were randomized into three groups:
- The control group received the standard of care with pegylated interferon and ribavirin for 48 weeks
- The response-guided therapy group received boceprevir plus pegylated interferon and ribavirin for 24 weeks after the 4-week lead-in phase; if HCV RNA was undetectable from week 8 to week 24, treatment was considered complete, but if HCV RNA was detectable at any point from week 8 to week 24, pegylated interferon and ribavirin were continued for a total of 48 weeks.
- The fixed-duration therapy group received boceprevir, pegylated interferon, and ribavirin for 44 weeks after the lead-in period.
The rate of relapse was 8% and 9% in the boceprevir groups vs 23% in the control group. Patients in the boceprevir groups who had a decrease in HCV RNA of less than 1 log10 during the lead-in phase were found to have a significantly higher rate of boceprevirresistant variants than those who achieved a decrease of HCV RNA of 1 log10 or more.
Boceprevir in previously treated patients with HCV genotype 1: The RESPOND-2 trial
The Retreatment With HCV Serine Protease Inhibitor Boceprevir and PegIntron/Rebetol 2) (RESPOND-2) trial2 was designed to assess the efficacy of combined boceprevir, pegylated interferon, and ribavirin for repeat treatment of patients with HCV genotype 1. These patients had previously undergone standard treatment and had a reduction of 2 log10 or more in HCV RNA after 12 weeks of therapy but with detectable HCV RNA during the therapy period or had had a relapse (defined as undetectable HCV RNA at the end of a previous course of therapy with HCV RNA positivity thereafter). Importantly, null-responders (those who had a reduction of less than 2 log10 in HCV RNA after 12 weeks of therapy) were excluded from this trial.
After a lead-in period of interferon-ribavirin treatment for 4 weeks, 403 patients were assigned to one of three treatment groups:
- Pegylated interferon and ribavirin for 44 weeks (the control group)
- Boceprevir, pegylated interferon, and ribavirin in a response-guided regimen
- Boceprevir, pegylated interferon, and ribavirin for 44 weeks (the fixed-duration group).
Sustained virologic response was achieved in only 21% of patients in the control group. Adding boceprevir increased the rate to 59% in the response-guided therapy group and to 67% in the fixed-duration group. Previous relapsers had better rates than partial responders (69%–75% vs 40%–52%).
Importantly, patients who had a poor response to pegylated interferon and ribavirin during the lead-in phase (defined as having less than a 1-log decrease in the virus before starting boceprevir) had significantly lower rates of sustained virologic response and higher rates of resistance-associated virus variants.
Side effects of boceprevir
Overall, boceprevir is well tolerated. The most common side effects of triple therapy are those usually seen with pegylated interferon and ribavirin, such as flulike symptoms and fatigue (Table 2). However, anemia was more frequent in the boceprevir groups in both SPRINT-2 and RESPOND-2 (45%–50% compared with 20%–29% in the control groups). Erythropoietin was allowed in these studies and was used in about 40% of patients.
The other common side effect associated with boceprevir was dysgeusia (alteration of taste). Dysgeusia was reported by approximately 40% of patients; however, most dysgeusia events were mild to moderate in intensity and did not lead to treatment cessation.
In the SPRINT-2 trial,1 the study drugs had to be discontinued in 12% to 16% of patients in the boceprevir groups because of adverse events, which was similar to the rate (16%) in the control group. Erythropoietin was allowed in this trial, and it was used in 43% of patients in the boceprevir groups compared with 24% in the control group, with discontinuation owing to anemia occurring in 2% and 1% of cases, respectively.
TELAPREVIR
Telaprevir, the other protease NS3/4A inhibitor, has also shown efficacy over current standard therapy in phase 3 clinical trials. It was used in a dose of 750 mg three times a day with pegylated interferon alfa-2a (Pegasys) 180 μg per week and ribavirin (Copegus) 1,000 to 1,200 mg daily according to body weight. A lead-in phase with pegylated interferon and ribavirin was not applied with telaprevir, as it was in the boceprevir trials. Extended rapid virologic response was defined as an undetectable HCV RNA at weeks 4 and 12 of therapy.
Telaprevir in previously untreated patients with HCV genotype 1
The ADVANCE study3 was a double-blind randomized trial assessing the efficacy and safety of telaprevir in combination with pegylated interferon and ribavirin in more than 1,000 previously untreated patients. The three treatment groups received:
- Telaprevir, pegylated interferon, and ribavirin for 8 weeks, followed by pegylated interferon and ribavirin alone for 16 weeks in patients who achieved an extended rapid virologic response (total duration of 24 weeks) or 40 weeks in patients who did not (total duration of 48 weeks)
- Telaprevir, pegylated interferon, and ribavirin for 12 weeks, followed by pegylated interferon-ribavirin alone for 12 (total of 24 weeks) or 36 weeks (total of 48 weeks) according to extended rapid virologic response
- Standard care with pegylated interferon and ribavirin for 48 weeks.
The rate of sustained virologic response was 69% in the group that received telaprevir for 8 weeks and 75% in the group that received it for 12 weeks compared with 44% in the control group (P < .0001 for both) (Table 2). Patients infected with HCV genotype 1b had a higher sustained virologic response rate (79%) than those infected with HCV genotype 1a (71%).
Sustained virologic response rates were lower in black patients and patients with bridging fibrosis or cirrhosis, but were still significantly higher in the telaprevir groups than in the control group. The results of this subset analysis were limited by small numbers of patients in each category.
In total, 57% of those who received telaprevir for 8 weeks and 58% of those who received it for 12 weeks achieved an extended rapid virologic response and were able to cut the duration of their therapy in half (from 48 weeks to 24 weeks).
The relapse rates were 9% in the telaprevir groups and 28% in the control group.
The rate of virologic failure was lower in patients who received triple therapy than in those who received interferon-ribavirin alone (8% in the group that got telaprevir for 12 weeks and 13% in the group that got it for 8 weeks, vs 32% in the control group). The failure rate was also lower in patients with HCV genotype 1b infection than in those with genotype 1a.
The ILLUMINATE study4 (Illustrating the Effects of Combination Therapy With Telaprevir) investigated whether longer duration of treatment than that given in the ADVANCE trial increased the rate of sustained virologic response. Previously untreated patients received telaprevir, interferon, and ribavirin for 12 weeks, and those who achieved an extended rapid virologic response were randomized at week 20 to continue interferonribavirin treatment for 24 or 48 weeks of total treatment.
The sustained virologic response rates in patients who achieved an extended rapid virologic response were 92% in the group that received pegylated interferon and ribavirin for 12 weeks, and 88% in those who received it for 48 weeks. Thus, the results of this study support the use of response-guided therapy for telaprevir-based regimens.
Telaprevir in previously treated patients with HCV genotype 1: The REALIZE trial
In this phase 3 placebo-controlled trial,5 622 patients with prior relapse, partial response, or null response were randomly allocated into one of three groups:
- Telaprevir for 12 weeks plus pegylated interferon and ribavirin for 48 weeks
- Lead-in for 4 weeks followed by 12 weeks of triple therapy and another 32 weeks of pegylated interferon and ribavirin
- Pegylated interferon and ribavirin for 48 weeks (the control group).
The overall sustained virologic response rates were 66% and 64%, respectively, in the telaprevir groups vs 17% in the control group (P < .0001). The sustained virologic response rates in the telaprevir groups were 83% to 88% in prior relapsers, 54% to 59% in partial responders, and 29% to 33% in null-responders. Of note, patients did not benefit from the lead-in phase.
This was the only trial to investigate the response to triple therapy in null-responders, a group in which treatment has been considered hopeless. A response rate of approximately 31% was encouraging, especially if we compare it with the 5% response rate achieved with the current standard of care with pegylated interferon and ribavirin.
Telaprevir side effects
As with boceprevir-based triple therapy, the most common adverse events were related to pegylated interferon (Table 2).
Nearly 50% of patients who receive telaprevir develop a skin rash that is primarily eczematous, can be managed with topical steroids, and usually resolves when telaprevir is discontinued. Severe rashes occurred in 3% to 6% of patients in the ADVANCE trial,3 and three suspected cases of Stevens-Johnson syndrome have been reported to the FDA.
Other side effects that were more frequent with telaprevir included pruritus, nausea, diarrhea, and anemia. On average, the hemoglobin level decreased by an additional 1 g/dL in the telaprevir treatment groups compared with the groups that received only pegylated interferon-ribavirin. Erythropoietin use was not allowed in the phase 3 telaprevir studies, and anemia was managed by ribavirin dose reduction.
In the ADVANCE trial,3 study drugs were discontinued owing to adverse events in 7% to 8% of the patients in the telaprevir groups compared with 4% in the control group. In the ILLUMINATE trial,4 17% of patients had to permanently discontinue all study drugs due to adverse events.
FDA-APPROVED TREATMENT REGIMENS FOR BOCEPREVIR AND TELAPREVIR
For treatment algorithms, see the eFigures that accompany this article online.
Boceprevir in previously untreated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 28—Stop all treatment if HCV RNA was undetectable at weeks 8 and 24
- Week 36—Measure HCV RNA; stop boceprevir
- Week 48—Stop all treatment (eFigure 1).
Boceprevir in previously treated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 36—if HCV RNA was not detectable at week 8, stop all treatment now; if HCV RNA was detectable at week 8, stop boceprevir now but continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 2).
Telaprevir in previously untreated patients and prior relapsers
- Week 0—start telaprevir, pegylated interferon, and ribavirin
- Week 4—measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL
- Week 12—Stop telaprevir; measure HCV RNA; stop all treatment if HCV RNA is more than 1,000 IU/mL
- Week 24—Stop pegylated interferon and ribavirin if HCV RNA was undetectable at week 12; measure HCV RNA and stop treatment if it is detectable; otherwise, continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 3).
Telaprevir in patients who previously achieved a partial or null response
- Week 0—Start telaprevir, pegylated interferon, and ribavirin
- Week 4—Measure HCV RNA; stop treatment if it is more than 1,000 IU/mL
- Week 12—Measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL; if less than 1,000 IU/mL then stop telaprevir but continue pegylated interferon and ribavirin
- Week 24—Measure HCV RNA; stop treatment if HCV RNA is detectable
- Week 48—Stop all treatment (eFigure 4).
Drug interactions with boceprevir and telaprevir
Both boceprevir and telaprevir inhibit cytochrome P450 3A (CYP3A) and thus are contraindicated in combination with drugs highly dependent on CYP3A for clearance and with drugs for which elevated plasma concentrations are associated with serious adverse events, such as atorvastatin (Lipitor), simvastatin (Zocor), sildenafil (Viagra), midazolam (Versed), and St. John’s wort. Giving potent inducers of CYP3A with boceprevir or telaprevir may lead to lower exposure and loss of efficacy of both protease inhibitors.
EMERGING THERAPIES FOR HCV
Thanks to a better understanding of the biology of HCV infection, the effort to develop new therapeutic agents started to focus on targeting specific steps of the viral life cycle, including attachment, entry into cells, replication, and release.24
Currently, more than 50 clinical trials are evaluating new direct-acting antivirals to treat HCV infection.25 Monoclonal and polyclonal antibodies that target the molecular process involved in HCV attachment and entry are being developed.26 The nonstructural protein NS5B (RNA polymerase) is intimately involved in viral replication and represents a promising target.27 Several nucleosides and nonnucleoside protease inhibitors have already entered clinical trials.
The low fidelity of the HCV replication machinery leads to a very high mutation rate, thus enabling the virus to quickly develop mutations that resist agents targeting viral enzymes.28 Therefore, a novel approach is to target host cofactors that are essential for HCV replication. An intriguing study by Lanford et al29 demonstrated that antagonizing microRNA-122 (the most abundant microRNA in the liver and an essential cofactor for viral RNA replication) by the oligonucleotide SPC3649 caused marked and prolonged reduction of HCV viremia in chronically infected chimpanzees.29
Although we are still in the early stages of drug development, the future holds great promise for newer drugs to improve the sustained virologic response, shorten the duration of treatment, improve tolerability with interferon-sparing regimens, and decrease viral resistance.
FUTURE PERSPECTIVES
With the introduction of the first direct-acting antiviral medications for HCV (boceprevir and telaprevir), 2011 will be marked as the year that changed hepatitis C treatment for the better. Triple therapy with pegylated interferon, ribavirin, and either boceprevir or telaprevir has the potential for increasing the rate of sustained virologic response to around 70% in previously untreated patients and 65% in previously treated patients who are infected with HCV genotype 1. The IL28B polymorphisms appear to play a role in the rate of sustained virologic response achieved with triple therapy, with preliminary data showing a better response rate in patients who have the CC genotype.17
These drugs will add up to $50,000 to the cost of treating hepatitis C virus infection, depending on the drug used and the length of treatment. However, they may be well worth it if they prevent liver failure and the need for transplantation.
Many questions remain, such as how to use these new regimens to treat special patient populations—for example, those with a recurrence of HCV infection after liver transplantation, those co-infected with HCV and human immunodeficiency virus, and those infected with HCV genotypes other than genotype 1.
Other direct-acting antiviral agents that specifically target the replication cycle of HCV are currently in clinical development. In fact, the future has already started with the release of the Interferon-Free Regimen for the Management of HCV (INFORM-1) study results.30 This was the first trial to evaluate an interferon-free regimen for patients with chronic HCV infection using two direct-acting antiviral drugs (the protease inhibitor danoprevir and the polymerase inhibitor RG7128), with promising results.
- Poordad F, McCone J, Bacon BR, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1195–1206.
- Bacon BR, Gordon SC, Lawitz E, et al. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1207–1217.
- Jacobson IM, McHutchison JG, Dusheiko G, et al; for the ADVANCE Study Team. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med 2011; 364:2405–2416.
- Sherman KE, Flamm SL, Afdhal NH, et al; for the ILLUMINATE Study Team. Response-guided telaprevir combination treatment for hepatitis C virus infection. N Engl J Med 2011; 365:1014–1024.
- Zeuzem S, Andreone P, Pol S, et al; for the REALIZE Study Team. Telaprevir for retreatment of HCV infection. N Engl J Med 2011; 364:2417–2428.
- Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 2006; 144:705–714.
- Mitchell AE, Colvin HM, Palmer Beasley R. Institute of Medicine recommendations for the prevention and control of hepatitis B and C. Hepatology 2010; 51:729–733.
- Kim WR. The burden of hepatitis C in the United States. Hepatology 2002; 36:S30–S34.
- Marcellin P, Asselah T, Boyer N. Fibrosis and disease progression in hepatitis C. Hepatology 2002; 36:S47–S56.
- Seeff LB. Natural history of chronic hepatitis C. Hepatology 2002; 36:S35–S46.
- Lavanchy D. The global burden of hepatitis C. Liver Int 2009; 29(suppl 1):74–81.
- National Institutes of Health Consensus Development Conference Statement: Management of hepatitis C: 2002—June 10–12, 2002. Hepatology 2002; 36:S3–S20.
- Pearlman BL, Traub N. Sustained virologic response to antiviral therapy for chronic hepatitis C virus infection: a cure and so much more. Clin Infect Dis 2011; 52:889–900.
- Hoofnagle JH, Seeff LB. Peginterferon and ribavirin for chronic hepatitis C. N Engl J Med 2006; 355:2444–2451.
- Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature 2009; 461:399–401.
- Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet 2009; 41:1100–1104.
- Thompson AJ, Muir AJ, Sulkowski MS, et al. Interleukin-28B polymorphism improves viral kinetics and is the strongest pretreatment predictor of sustained virologic response in genotype 1 hepatitis C virus. Gastroenterology 2010; 139:120–129.e118.
- Nielsen SU, Bassendine MF, Burt AD, Bevitt DJ, Toms GL. Characterization of the genome and structural proteins of hepatitis C virus resolved from infected human liver. J Gen Virol 2004; 85:1497–1507.
- Penin F, Dubuisson J, Rey FA, Moradpour D, Pawlotsky JM. Structural biology of hepatitis C virus. Hepatology 2004; 39:5–19.
- Nelson DR. The role of triple therapy with protease inhibitors in hepatitis C virus genotype 1 naive patients. Liver Int 2011; 31(suppl 1):53–57.
- Pawlotsky JM. Treatment failure and resistance with direct-acting antiviral drugs against hepatitis C virus. Hepatology 2011; 53:1742–1751.
- Monto A, Schooley RT, Lai JC, et al. Lessons from HIV therapy applied to viral hepatitis therapy: summary of a workshop. Am J Gastroenterol 2010; 105:989–1004.
- McCown MF, Rajyaguru S, Kular S, Cammack N, Najera I. GT-1a or GT-1b subtype-specific resistance profiles for hepatitis C virus inhibitors telaprevir and HCV-796. Antimicrob Agents Chemother 2009; 53:2129–2132.
- Cholongitas E, Papatheodoridis GV. Review article: novel therapeutic options for chronic hepatitis C. Aliment Pharmacol Ther 2008; 27:866–884.
- Naggie S, Patel K, McHutchison J. Hepatitis C virus directly acting antivirals: current developments with NS3/4A HCV serine protease inhibitors. J Antimicrob Chemother 2010; 65:2063–2069.
- Mir HM, Birerdinc A, Younossi ZM. Monoclonal and polyclonal antibodies against the HCV envelope proteins. Clin Liver Dis 2009; 13:477–486.
- Birerdinc A, Younossi ZM. Emerging therapies for hepatitis C virus. Expert Opin Emerg Drugs 2010; 15:535–544.
- Khattab MA. Targeting host factors: a novel rationale for the management of hepatitis C virus. World J Gastroenterol 2009; 15:3472–3479.
- Lanford RE, Hildebrandt-Eriksen ES, Petri A, et al. Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science 2010; 327:198–201.
- Gane EJ, Roberts SK, Stedman CA, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet 2010; 376:1467–1475.
The treatment of hepatitis c virus (HCV) infection is on the brink of major changes with the recent approval of the first direct-acting antiviral agents, the protease inhibitors boceprevir (Victrelis) and telaprevir (Incivek).
Both drugs were approved by the US Food and Drug Administration (FDA) Advisory Panel for Chronic Hepatitis C in May 2011 and are believed to significantly improve treatment outcomes for patients with HCV genotype 1 infection.
A MAJOR PUBLIC HEALTH PROBLEM
HCV infection is a major public health problem. Nearly 4 million people in the United States are infected.6,7 Most patients with acute HCV infection become chronically infected, and up to 25% eventually develop cirrhosis and its complications, making HCV infection the leading indication for liver transplantation.8–10
Chronic HCV infection has a large global impact, with 180 million people affected across all economic and social groups.11 The highest prevalence of HCV has been reported in Egypt (14%), in part due to the use of inadequately sterilized needles in mass programs to treat endemic schistosomiasis. In developed countries, hepatocellular carcinoma associated with HCV has the fastest growing cancer-related death rate.12
CURRENTLY, FEWER THAN 50% OF PATIENTS ARE CURED
The goal of HCV treatment is to eradicate the virus. However, most infected patients (especially in the United States and Europe) are infected with HCV genotype 1, which is the most difficult genotype to treat.
Successful treatment of HCV is defined as achieving a sustained virologic response—ie, the absence of detectable HCV RNA in the serum 24 weeks after completion of therapy. Once a sustained virologic response is achieved, lifetime “cure” of HCV infection is expected in more than 99% of patients.13
The current standard therapy for HCV, pegylated interferon plus ribavirin for 48 weeks, is effective in only 40% to 50% of patients with genotype 1 infection.14 Therefore, assessing predictors of response before starting treatment can help select patients who are most likely to benefit from therapy.
Viral factors associated with a sustained virologic response include HCV genotypes other than genotype 1 and a low baseline viral load.
Beneficial patient-related factors include younger age, nonblack ethnicity, low body weight (≤ 75 kg), low body mass index, absence of insulin resistance, and absence of advanced fibrosis or cirrhosis.
More recently, a single-nucleotide polymorphism near the interleukin 28B (IL28B) gene, coding for interferon lambda 3, was found to be associated with a twofold difference in the rates of sustained virologic response: patients with the favorable genotype CC were two times more likely to achieve a sustained virologic response than patients with the CT or TT genotypes.15–17
PROTEASE INHIBITORS: MECHANISM OF ACTION
NS3/4A protease inhibitors rely on the principle of end-product inhibition, in which the cleavage product of the protease (a peptide) acts to inhibit the enzyme activity; this is why they are called peptidomimetics. The active site of the NS3/4A protease is a shallow groove composed of three highly conserved amino acid residues, which may explain why protease inhibitors display high antiviral efficacy but pose a low barrier to the development of resistance.20
Protease inhibitors are prone to resistance
The development of viral resistance to protease inhibitors has been a major drawback to their use in patients with chronic HCV infection.21
HCV is a highly variable virus with many genetically distinct but closely related quasispecies circulating in the blood at any given time. Drug-resistant, mutated variants preexist within the patient’s quasispecies, but only in small quantities because of their lesser replication fitness compared with the wild-type virus.22 When direct-acting antiviral therapy is started, the quantity of the wild-type virus decreases and the mutated virus gains replication fitness. Using protease inhibitors as monotherapy selects resistant viral populations rapidly within a few days or weeks.
HCV subtypes 1a and 1b may have different resistance profiles. With genotype 1a, some resistance-associated amino acid substitutions require only one nucleotide change, but with genotype 1b, two nucleotide changes are needed, making resistance less frequent in patients with HCV genotype 1b.23
BOCEPREVIR
Boceprevir is a specific inhibitor of the HCV viral protease NS3/4A.
In phase 3 clinical trials, boceprevir 800 mg three times a day was used with pegylated interferon alfa-2b (PegIntron) 1.5 μg/kg/week and ribavirin (Rebetol) 600 to 1,400 mg daily according to body weight.
Before patients started taking boceprevir, they went through a 4-week lead-in phase, during which they received pegylated interferon and ribavirin. This schedule appeared to reduce the incidence of viral breakthrough in phase 2 trials, and it produced higher rates of sustained virologic response and lower relapse rates compared with triple therapy without a lead-in phase.
Rapid virologic response was defined as undetectable HCV RNA at week 4 of boceprevir therapy (week 8 of the whole regimen).
Boceprevir in previously untreated patients with HCV genotype 1: The SPRINT-2 trial
The Serine Protease Inhibitor Therapy 2 (SPRINT-2) trial1 included more than 1,000 previously untreated adults with HCV genotype 1 infection (938 nonblack patients and 159 black patients; two other nonblack patients did not receive any study drug and were not included in the analysis). In this double-blind trial, patients were randomized into three groups:
- The control group received the standard of care with pegylated interferon and ribavirin for 48 weeks
- The response-guided therapy group received boceprevir plus pegylated interferon and ribavirin for 24 weeks after the 4-week lead-in phase; if HCV RNA was undetectable from week 8 to week 24, treatment was considered complete, but if HCV RNA was detectable at any point from week 8 to week 24, pegylated interferon and ribavirin were continued for a total of 48 weeks.
- The fixed-duration therapy group received boceprevir, pegylated interferon, and ribavirin for 44 weeks after the lead-in period.
The rate of relapse was 8% and 9% in the boceprevir groups vs 23% in the control group. Patients in the boceprevir groups who had a decrease in HCV RNA of less than 1 log10 during the lead-in phase were found to have a significantly higher rate of boceprevirresistant variants than those who achieved a decrease of HCV RNA of 1 log10 or more.
Boceprevir in previously treated patients with HCV genotype 1: The RESPOND-2 trial
The Retreatment With HCV Serine Protease Inhibitor Boceprevir and PegIntron/Rebetol 2) (RESPOND-2) trial2 was designed to assess the efficacy of combined boceprevir, pegylated interferon, and ribavirin for repeat treatment of patients with HCV genotype 1. These patients had previously undergone standard treatment and had a reduction of 2 log10 or more in HCV RNA after 12 weeks of therapy but with detectable HCV RNA during the therapy period or had had a relapse (defined as undetectable HCV RNA at the end of a previous course of therapy with HCV RNA positivity thereafter). Importantly, null-responders (those who had a reduction of less than 2 log10 in HCV RNA after 12 weeks of therapy) were excluded from this trial.
After a lead-in period of interferon-ribavirin treatment for 4 weeks, 403 patients were assigned to one of three treatment groups:
- Pegylated interferon and ribavirin for 44 weeks (the control group)
- Boceprevir, pegylated interferon, and ribavirin in a response-guided regimen
- Boceprevir, pegylated interferon, and ribavirin for 44 weeks (the fixed-duration group).
Sustained virologic response was achieved in only 21% of patients in the control group. Adding boceprevir increased the rate to 59% in the response-guided therapy group and to 67% in the fixed-duration group. Previous relapsers had better rates than partial responders (69%–75% vs 40%–52%).
Importantly, patients who had a poor response to pegylated interferon and ribavirin during the lead-in phase (defined as having less than a 1-log decrease in the virus before starting boceprevir) had significantly lower rates of sustained virologic response and higher rates of resistance-associated virus variants.
Side effects of boceprevir
Overall, boceprevir is well tolerated. The most common side effects of triple therapy are those usually seen with pegylated interferon and ribavirin, such as flulike symptoms and fatigue (Table 2). However, anemia was more frequent in the boceprevir groups in both SPRINT-2 and RESPOND-2 (45%–50% compared with 20%–29% in the control groups). Erythropoietin was allowed in these studies and was used in about 40% of patients.
The other common side effect associated with boceprevir was dysgeusia (alteration of taste). Dysgeusia was reported by approximately 40% of patients; however, most dysgeusia events were mild to moderate in intensity and did not lead to treatment cessation.
In the SPRINT-2 trial,1 the study drugs had to be discontinued in 12% to 16% of patients in the boceprevir groups because of adverse events, which was similar to the rate (16%) in the control group. Erythropoietin was allowed in this trial, and it was used in 43% of patients in the boceprevir groups compared with 24% in the control group, with discontinuation owing to anemia occurring in 2% and 1% of cases, respectively.
TELAPREVIR
Telaprevir, the other protease NS3/4A inhibitor, has also shown efficacy over current standard therapy in phase 3 clinical trials. It was used in a dose of 750 mg three times a day with pegylated interferon alfa-2a (Pegasys) 180 μg per week and ribavirin (Copegus) 1,000 to 1,200 mg daily according to body weight. A lead-in phase with pegylated interferon and ribavirin was not applied with telaprevir, as it was in the boceprevir trials. Extended rapid virologic response was defined as an undetectable HCV RNA at weeks 4 and 12 of therapy.
Telaprevir in previously untreated patients with HCV genotype 1
The ADVANCE study3 was a double-blind randomized trial assessing the efficacy and safety of telaprevir in combination with pegylated interferon and ribavirin in more than 1,000 previously untreated patients. The three treatment groups received:
- Telaprevir, pegylated interferon, and ribavirin for 8 weeks, followed by pegylated interferon and ribavirin alone for 16 weeks in patients who achieved an extended rapid virologic response (total duration of 24 weeks) or 40 weeks in patients who did not (total duration of 48 weeks)
- Telaprevir, pegylated interferon, and ribavirin for 12 weeks, followed by pegylated interferon-ribavirin alone for 12 (total of 24 weeks) or 36 weeks (total of 48 weeks) according to extended rapid virologic response
- Standard care with pegylated interferon and ribavirin for 48 weeks.
The rate of sustained virologic response was 69% in the group that received telaprevir for 8 weeks and 75% in the group that received it for 12 weeks compared with 44% in the control group (P < .0001 for both) (Table 2). Patients infected with HCV genotype 1b had a higher sustained virologic response rate (79%) than those infected with HCV genotype 1a (71%).
Sustained virologic response rates were lower in black patients and patients with bridging fibrosis or cirrhosis, but were still significantly higher in the telaprevir groups than in the control group. The results of this subset analysis were limited by small numbers of patients in each category.
In total, 57% of those who received telaprevir for 8 weeks and 58% of those who received it for 12 weeks achieved an extended rapid virologic response and were able to cut the duration of their therapy in half (from 48 weeks to 24 weeks).
The relapse rates were 9% in the telaprevir groups and 28% in the control group.
The rate of virologic failure was lower in patients who received triple therapy than in those who received interferon-ribavirin alone (8% in the group that got telaprevir for 12 weeks and 13% in the group that got it for 8 weeks, vs 32% in the control group). The failure rate was also lower in patients with HCV genotype 1b infection than in those with genotype 1a.
The ILLUMINATE study4 (Illustrating the Effects of Combination Therapy With Telaprevir) investigated whether longer duration of treatment than that given in the ADVANCE trial increased the rate of sustained virologic response. Previously untreated patients received telaprevir, interferon, and ribavirin for 12 weeks, and those who achieved an extended rapid virologic response were randomized at week 20 to continue interferonribavirin treatment for 24 or 48 weeks of total treatment.
The sustained virologic response rates in patients who achieved an extended rapid virologic response were 92% in the group that received pegylated interferon and ribavirin for 12 weeks, and 88% in those who received it for 48 weeks. Thus, the results of this study support the use of response-guided therapy for telaprevir-based regimens.
Telaprevir in previously treated patients with HCV genotype 1: The REALIZE trial
In this phase 3 placebo-controlled trial,5 622 patients with prior relapse, partial response, or null response were randomly allocated into one of three groups:
- Telaprevir for 12 weeks plus pegylated interferon and ribavirin for 48 weeks
- Lead-in for 4 weeks followed by 12 weeks of triple therapy and another 32 weeks of pegylated interferon and ribavirin
- Pegylated interferon and ribavirin for 48 weeks (the control group).
The overall sustained virologic response rates were 66% and 64%, respectively, in the telaprevir groups vs 17% in the control group (P < .0001). The sustained virologic response rates in the telaprevir groups were 83% to 88% in prior relapsers, 54% to 59% in partial responders, and 29% to 33% in null-responders. Of note, patients did not benefit from the lead-in phase.
This was the only trial to investigate the response to triple therapy in null-responders, a group in which treatment has been considered hopeless. A response rate of approximately 31% was encouraging, especially if we compare it with the 5% response rate achieved with the current standard of care with pegylated interferon and ribavirin.
Telaprevir side effects
As with boceprevir-based triple therapy, the most common adverse events were related to pegylated interferon (Table 2).
Nearly 50% of patients who receive telaprevir develop a skin rash that is primarily eczematous, can be managed with topical steroids, and usually resolves when telaprevir is discontinued. Severe rashes occurred in 3% to 6% of patients in the ADVANCE trial,3 and three suspected cases of Stevens-Johnson syndrome have been reported to the FDA.
Other side effects that were more frequent with telaprevir included pruritus, nausea, diarrhea, and anemia. On average, the hemoglobin level decreased by an additional 1 g/dL in the telaprevir treatment groups compared with the groups that received only pegylated interferon-ribavirin. Erythropoietin use was not allowed in the phase 3 telaprevir studies, and anemia was managed by ribavirin dose reduction.
In the ADVANCE trial,3 study drugs were discontinued owing to adverse events in 7% to 8% of the patients in the telaprevir groups compared with 4% in the control group. In the ILLUMINATE trial,4 17% of patients had to permanently discontinue all study drugs due to adverse events.
FDA-APPROVED TREATMENT REGIMENS FOR BOCEPREVIR AND TELAPREVIR
For treatment algorithms, see the eFigures that accompany this article online.
Boceprevir in previously untreated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 28—Stop all treatment if HCV RNA was undetectable at weeks 8 and 24
- Week 36—Measure HCV RNA; stop boceprevir
- Week 48—Stop all treatment (eFigure 1).
Boceprevir in previously treated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 36—if HCV RNA was not detectable at week 8, stop all treatment now; if HCV RNA was detectable at week 8, stop boceprevir now but continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 2).
Telaprevir in previously untreated patients and prior relapsers
- Week 0—start telaprevir, pegylated interferon, and ribavirin
- Week 4—measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL
- Week 12—Stop telaprevir; measure HCV RNA; stop all treatment if HCV RNA is more than 1,000 IU/mL
- Week 24—Stop pegylated interferon and ribavirin if HCV RNA was undetectable at week 12; measure HCV RNA and stop treatment if it is detectable; otherwise, continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 3).
Telaprevir in patients who previously achieved a partial or null response
- Week 0—Start telaprevir, pegylated interferon, and ribavirin
- Week 4—Measure HCV RNA; stop treatment if it is more than 1,000 IU/mL
- Week 12—Measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL; if less than 1,000 IU/mL then stop telaprevir but continue pegylated interferon and ribavirin
- Week 24—Measure HCV RNA; stop treatment if HCV RNA is detectable
- Week 48—Stop all treatment (eFigure 4).
Drug interactions with boceprevir and telaprevir
Both boceprevir and telaprevir inhibit cytochrome P450 3A (CYP3A) and thus are contraindicated in combination with drugs highly dependent on CYP3A for clearance and with drugs for which elevated plasma concentrations are associated with serious adverse events, such as atorvastatin (Lipitor), simvastatin (Zocor), sildenafil (Viagra), midazolam (Versed), and St. John’s wort. Giving potent inducers of CYP3A with boceprevir or telaprevir may lead to lower exposure and loss of efficacy of both protease inhibitors.
EMERGING THERAPIES FOR HCV
Thanks to a better understanding of the biology of HCV infection, the effort to develop new therapeutic agents started to focus on targeting specific steps of the viral life cycle, including attachment, entry into cells, replication, and release.24
Currently, more than 50 clinical trials are evaluating new direct-acting antivirals to treat HCV infection.25 Monoclonal and polyclonal antibodies that target the molecular process involved in HCV attachment and entry are being developed.26 The nonstructural protein NS5B (RNA polymerase) is intimately involved in viral replication and represents a promising target.27 Several nucleosides and nonnucleoside protease inhibitors have already entered clinical trials.
The low fidelity of the HCV replication machinery leads to a very high mutation rate, thus enabling the virus to quickly develop mutations that resist agents targeting viral enzymes.28 Therefore, a novel approach is to target host cofactors that are essential for HCV replication. An intriguing study by Lanford et al29 demonstrated that antagonizing microRNA-122 (the most abundant microRNA in the liver and an essential cofactor for viral RNA replication) by the oligonucleotide SPC3649 caused marked and prolonged reduction of HCV viremia in chronically infected chimpanzees.29
Although we are still in the early stages of drug development, the future holds great promise for newer drugs to improve the sustained virologic response, shorten the duration of treatment, improve tolerability with interferon-sparing regimens, and decrease viral resistance.
FUTURE PERSPECTIVES
With the introduction of the first direct-acting antiviral medications for HCV (boceprevir and telaprevir), 2011 will be marked as the year that changed hepatitis C treatment for the better. Triple therapy with pegylated interferon, ribavirin, and either boceprevir or telaprevir has the potential for increasing the rate of sustained virologic response to around 70% in previously untreated patients and 65% in previously treated patients who are infected with HCV genotype 1. The IL28B polymorphisms appear to play a role in the rate of sustained virologic response achieved with triple therapy, with preliminary data showing a better response rate in patients who have the CC genotype.17
These drugs will add up to $50,000 to the cost of treating hepatitis C virus infection, depending on the drug used and the length of treatment. However, they may be well worth it if they prevent liver failure and the need for transplantation.
Many questions remain, such as how to use these new regimens to treat special patient populations—for example, those with a recurrence of HCV infection after liver transplantation, those co-infected with HCV and human immunodeficiency virus, and those infected with HCV genotypes other than genotype 1.
Other direct-acting antiviral agents that specifically target the replication cycle of HCV are currently in clinical development. In fact, the future has already started with the release of the Interferon-Free Regimen for the Management of HCV (INFORM-1) study results.30 This was the first trial to evaluate an interferon-free regimen for patients with chronic HCV infection using two direct-acting antiviral drugs (the protease inhibitor danoprevir and the polymerase inhibitor RG7128), with promising results.
The treatment of hepatitis c virus (HCV) infection is on the brink of major changes with the recent approval of the first direct-acting antiviral agents, the protease inhibitors boceprevir (Victrelis) and telaprevir (Incivek).
Both drugs were approved by the US Food and Drug Administration (FDA) Advisory Panel for Chronic Hepatitis C in May 2011 and are believed to significantly improve treatment outcomes for patients with HCV genotype 1 infection.
A MAJOR PUBLIC HEALTH PROBLEM
HCV infection is a major public health problem. Nearly 4 million people in the United States are infected.6,7 Most patients with acute HCV infection become chronically infected, and up to 25% eventually develop cirrhosis and its complications, making HCV infection the leading indication for liver transplantation.8–10
Chronic HCV infection has a large global impact, with 180 million people affected across all economic and social groups.11 The highest prevalence of HCV has been reported in Egypt (14%), in part due to the use of inadequately sterilized needles in mass programs to treat endemic schistosomiasis. In developed countries, hepatocellular carcinoma associated with HCV has the fastest growing cancer-related death rate.12
CURRENTLY, FEWER THAN 50% OF PATIENTS ARE CURED
The goal of HCV treatment is to eradicate the virus. However, most infected patients (especially in the United States and Europe) are infected with HCV genotype 1, which is the most difficult genotype to treat.
Successful treatment of HCV is defined as achieving a sustained virologic response—ie, the absence of detectable HCV RNA in the serum 24 weeks after completion of therapy. Once a sustained virologic response is achieved, lifetime “cure” of HCV infection is expected in more than 99% of patients.13
The current standard therapy for HCV, pegylated interferon plus ribavirin for 48 weeks, is effective in only 40% to 50% of patients with genotype 1 infection.14 Therefore, assessing predictors of response before starting treatment can help select patients who are most likely to benefit from therapy.
Viral factors associated with a sustained virologic response include HCV genotypes other than genotype 1 and a low baseline viral load.
Beneficial patient-related factors include younger age, nonblack ethnicity, low body weight (≤ 75 kg), low body mass index, absence of insulin resistance, and absence of advanced fibrosis or cirrhosis.
More recently, a single-nucleotide polymorphism near the interleukin 28B (IL28B) gene, coding for interferon lambda 3, was found to be associated with a twofold difference in the rates of sustained virologic response: patients with the favorable genotype CC were two times more likely to achieve a sustained virologic response than patients with the CT or TT genotypes.15–17
PROTEASE INHIBITORS: MECHANISM OF ACTION
NS3/4A protease inhibitors rely on the principle of end-product inhibition, in which the cleavage product of the protease (a peptide) acts to inhibit the enzyme activity; this is why they are called peptidomimetics. The active site of the NS3/4A protease is a shallow groove composed of three highly conserved amino acid residues, which may explain why protease inhibitors display high antiviral efficacy but pose a low barrier to the development of resistance.20
Protease inhibitors are prone to resistance
The development of viral resistance to protease inhibitors has been a major drawback to their use in patients with chronic HCV infection.21
HCV is a highly variable virus with many genetically distinct but closely related quasispecies circulating in the blood at any given time. Drug-resistant, mutated variants preexist within the patient’s quasispecies, but only in small quantities because of their lesser replication fitness compared with the wild-type virus.22 When direct-acting antiviral therapy is started, the quantity of the wild-type virus decreases and the mutated virus gains replication fitness. Using protease inhibitors as monotherapy selects resistant viral populations rapidly within a few days or weeks.
HCV subtypes 1a and 1b may have different resistance profiles. With genotype 1a, some resistance-associated amino acid substitutions require only one nucleotide change, but with genotype 1b, two nucleotide changes are needed, making resistance less frequent in patients with HCV genotype 1b.23
BOCEPREVIR
Boceprevir is a specific inhibitor of the HCV viral protease NS3/4A.
In phase 3 clinical trials, boceprevir 800 mg three times a day was used with pegylated interferon alfa-2b (PegIntron) 1.5 μg/kg/week and ribavirin (Rebetol) 600 to 1,400 mg daily according to body weight.
Before patients started taking boceprevir, they went through a 4-week lead-in phase, during which they received pegylated interferon and ribavirin. This schedule appeared to reduce the incidence of viral breakthrough in phase 2 trials, and it produced higher rates of sustained virologic response and lower relapse rates compared with triple therapy without a lead-in phase.
Rapid virologic response was defined as undetectable HCV RNA at week 4 of boceprevir therapy (week 8 of the whole regimen).
Boceprevir in previously untreated patients with HCV genotype 1: The SPRINT-2 trial
The Serine Protease Inhibitor Therapy 2 (SPRINT-2) trial1 included more than 1,000 previously untreated adults with HCV genotype 1 infection (938 nonblack patients and 159 black patients; two other nonblack patients did not receive any study drug and were not included in the analysis). In this double-blind trial, patients were randomized into three groups:
- The control group received the standard of care with pegylated interferon and ribavirin for 48 weeks
- The response-guided therapy group received boceprevir plus pegylated interferon and ribavirin for 24 weeks after the 4-week lead-in phase; if HCV RNA was undetectable from week 8 to week 24, treatment was considered complete, but if HCV RNA was detectable at any point from week 8 to week 24, pegylated interferon and ribavirin were continued for a total of 48 weeks.
- The fixed-duration therapy group received boceprevir, pegylated interferon, and ribavirin for 44 weeks after the lead-in period.
The rate of relapse was 8% and 9% in the boceprevir groups vs 23% in the control group. Patients in the boceprevir groups who had a decrease in HCV RNA of less than 1 log10 during the lead-in phase were found to have a significantly higher rate of boceprevirresistant variants than those who achieved a decrease of HCV RNA of 1 log10 or more.
Boceprevir in previously treated patients with HCV genotype 1: The RESPOND-2 trial
The Retreatment With HCV Serine Protease Inhibitor Boceprevir and PegIntron/Rebetol 2) (RESPOND-2) trial2 was designed to assess the efficacy of combined boceprevir, pegylated interferon, and ribavirin for repeat treatment of patients with HCV genotype 1. These patients had previously undergone standard treatment and had a reduction of 2 log10 or more in HCV RNA after 12 weeks of therapy but with detectable HCV RNA during the therapy period or had had a relapse (defined as undetectable HCV RNA at the end of a previous course of therapy with HCV RNA positivity thereafter). Importantly, null-responders (those who had a reduction of less than 2 log10 in HCV RNA after 12 weeks of therapy) were excluded from this trial.
After a lead-in period of interferon-ribavirin treatment for 4 weeks, 403 patients were assigned to one of three treatment groups:
- Pegylated interferon and ribavirin for 44 weeks (the control group)
- Boceprevir, pegylated interferon, and ribavirin in a response-guided regimen
- Boceprevir, pegylated interferon, and ribavirin for 44 weeks (the fixed-duration group).
Sustained virologic response was achieved in only 21% of patients in the control group. Adding boceprevir increased the rate to 59% in the response-guided therapy group and to 67% in the fixed-duration group. Previous relapsers had better rates than partial responders (69%–75% vs 40%–52%).
Importantly, patients who had a poor response to pegylated interferon and ribavirin during the lead-in phase (defined as having less than a 1-log decrease in the virus before starting boceprevir) had significantly lower rates of sustained virologic response and higher rates of resistance-associated virus variants.
Side effects of boceprevir
Overall, boceprevir is well tolerated. The most common side effects of triple therapy are those usually seen with pegylated interferon and ribavirin, such as flulike symptoms and fatigue (Table 2). However, anemia was more frequent in the boceprevir groups in both SPRINT-2 and RESPOND-2 (45%–50% compared with 20%–29% in the control groups). Erythropoietin was allowed in these studies and was used in about 40% of patients.
The other common side effect associated with boceprevir was dysgeusia (alteration of taste). Dysgeusia was reported by approximately 40% of patients; however, most dysgeusia events were mild to moderate in intensity and did not lead to treatment cessation.
In the SPRINT-2 trial,1 the study drugs had to be discontinued in 12% to 16% of patients in the boceprevir groups because of adverse events, which was similar to the rate (16%) in the control group. Erythropoietin was allowed in this trial, and it was used in 43% of patients in the boceprevir groups compared with 24% in the control group, with discontinuation owing to anemia occurring in 2% and 1% of cases, respectively.
TELAPREVIR
Telaprevir, the other protease NS3/4A inhibitor, has also shown efficacy over current standard therapy in phase 3 clinical trials. It was used in a dose of 750 mg three times a day with pegylated interferon alfa-2a (Pegasys) 180 μg per week and ribavirin (Copegus) 1,000 to 1,200 mg daily according to body weight. A lead-in phase with pegylated interferon and ribavirin was not applied with telaprevir, as it was in the boceprevir trials. Extended rapid virologic response was defined as an undetectable HCV RNA at weeks 4 and 12 of therapy.
Telaprevir in previously untreated patients with HCV genotype 1
The ADVANCE study3 was a double-blind randomized trial assessing the efficacy and safety of telaprevir in combination with pegylated interferon and ribavirin in more than 1,000 previously untreated patients. The three treatment groups received:
- Telaprevir, pegylated interferon, and ribavirin for 8 weeks, followed by pegylated interferon and ribavirin alone for 16 weeks in patients who achieved an extended rapid virologic response (total duration of 24 weeks) or 40 weeks in patients who did not (total duration of 48 weeks)
- Telaprevir, pegylated interferon, and ribavirin for 12 weeks, followed by pegylated interferon-ribavirin alone for 12 (total of 24 weeks) or 36 weeks (total of 48 weeks) according to extended rapid virologic response
- Standard care with pegylated interferon and ribavirin for 48 weeks.
The rate of sustained virologic response was 69% in the group that received telaprevir for 8 weeks and 75% in the group that received it for 12 weeks compared with 44% in the control group (P < .0001 for both) (Table 2). Patients infected with HCV genotype 1b had a higher sustained virologic response rate (79%) than those infected with HCV genotype 1a (71%).
Sustained virologic response rates were lower in black patients and patients with bridging fibrosis or cirrhosis, but were still significantly higher in the telaprevir groups than in the control group. The results of this subset analysis were limited by small numbers of patients in each category.
In total, 57% of those who received telaprevir for 8 weeks and 58% of those who received it for 12 weeks achieved an extended rapid virologic response and were able to cut the duration of their therapy in half (from 48 weeks to 24 weeks).
The relapse rates were 9% in the telaprevir groups and 28% in the control group.
The rate of virologic failure was lower in patients who received triple therapy than in those who received interferon-ribavirin alone (8% in the group that got telaprevir for 12 weeks and 13% in the group that got it for 8 weeks, vs 32% in the control group). The failure rate was also lower in patients with HCV genotype 1b infection than in those with genotype 1a.
The ILLUMINATE study4 (Illustrating the Effects of Combination Therapy With Telaprevir) investigated whether longer duration of treatment than that given in the ADVANCE trial increased the rate of sustained virologic response. Previously untreated patients received telaprevir, interferon, and ribavirin for 12 weeks, and those who achieved an extended rapid virologic response were randomized at week 20 to continue interferonribavirin treatment for 24 or 48 weeks of total treatment.
The sustained virologic response rates in patients who achieved an extended rapid virologic response were 92% in the group that received pegylated interferon and ribavirin for 12 weeks, and 88% in those who received it for 48 weeks. Thus, the results of this study support the use of response-guided therapy for telaprevir-based regimens.
Telaprevir in previously treated patients with HCV genotype 1: The REALIZE trial
In this phase 3 placebo-controlled trial,5 622 patients with prior relapse, partial response, or null response were randomly allocated into one of three groups:
- Telaprevir for 12 weeks plus pegylated interferon and ribavirin for 48 weeks
- Lead-in for 4 weeks followed by 12 weeks of triple therapy and another 32 weeks of pegylated interferon and ribavirin
- Pegylated interferon and ribavirin for 48 weeks (the control group).
The overall sustained virologic response rates were 66% and 64%, respectively, in the telaprevir groups vs 17% in the control group (P < .0001). The sustained virologic response rates in the telaprevir groups were 83% to 88% in prior relapsers, 54% to 59% in partial responders, and 29% to 33% in null-responders. Of note, patients did not benefit from the lead-in phase.
This was the only trial to investigate the response to triple therapy in null-responders, a group in which treatment has been considered hopeless. A response rate of approximately 31% was encouraging, especially if we compare it with the 5% response rate achieved with the current standard of care with pegylated interferon and ribavirin.
Telaprevir side effects
As with boceprevir-based triple therapy, the most common adverse events were related to pegylated interferon (Table 2).
Nearly 50% of patients who receive telaprevir develop a skin rash that is primarily eczematous, can be managed with topical steroids, and usually resolves when telaprevir is discontinued. Severe rashes occurred in 3% to 6% of patients in the ADVANCE trial,3 and three suspected cases of Stevens-Johnson syndrome have been reported to the FDA.
Other side effects that were more frequent with telaprevir included pruritus, nausea, diarrhea, and anemia. On average, the hemoglobin level decreased by an additional 1 g/dL in the telaprevir treatment groups compared with the groups that received only pegylated interferon-ribavirin. Erythropoietin use was not allowed in the phase 3 telaprevir studies, and anemia was managed by ribavirin dose reduction.
In the ADVANCE trial,3 study drugs were discontinued owing to adverse events in 7% to 8% of the patients in the telaprevir groups compared with 4% in the control group. In the ILLUMINATE trial,4 17% of patients had to permanently discontinue all study drugs due to adverse events.
FDA-APPROVED TREATMENT REGIMENS FOR BOCEPREVIR AND TELAPREVIR
For treatment algorithms, see the eFigures that accompany this article online.
Boceprevir in previously untreated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 28—Stop all treatment if HCV RNA was undetectable at weeks 8 and 24
- Week 36—Measure HCV RNA; stop boceprevir
- Week 48—Stop all treatment (eFigure 1).
Boceprevir in previously treated patients
- Week 0—Start pegylated interferon and ribavirin
- Week 4—Add boceprevir
- Week 8—Measure HCV RNA
- Week 12—Measure HCV RNA; stop treatment if it is more than 100 IU/mL
- Week 24—Measure HCV RNA; stop treatment if it is detectable
- Week 36—if HCV RNA was not detectable at week 8, stop all treatment now; if HCV RNA was detectable at week 8, stop boceprevir now but continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 2).
Telaprevir in previously untreated patients and prior relapsers
- Week 0—start telaprevir, pegylated interferon, and ribavirin
- Week 4—measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL
- Week 12—Stop telaprevir; measure HCV RNA; stop all treatment if HCV RNA is more than 1,000 IU/mL
- Week 24—Stop pegylated interferon and ribavirin if HCV RNA was undetectable at week 12; measure HCV RNA and stop treatment if it is detectable; otherwise, continue pegylated interferon and ribavirin
- Week 48—Stop all treatment (eFigure 3).
Telaprevir in patients who previously achieved a partial or null response
- Week 0—Start telaprevir, pegylated interferon, and ribavirin
- Week 4—Measure HCV RNA; stop treatment if it is more than 1,000 IU/mL
- Week 12—Measure HCV RNA; stop all treatment if it is more than 1,000 IU/mL; if less than 1,000 IU/mL then stop telaprevir but continue pegylated interferon and ribavirin
- Week 24—Measure HCV RNA; stop treatment if HCV RNA is detectable
- Week 48—Stop all treatment (eFigure 4).
Drug interactions with boceprevir and telaprevir
Both boceprevir and telaprevir inhibit cytochrome P450 3A (CYP3A) and thus are contraindicated in combination with drugs highly dependent on CYP3A for clearance and with drugs for which elevated plasma concentrations are associated with serious adverse events, such as atorvastatin (Lipitor), simvastatin (Zocor), sildenafil (Viagra), midazolam (Versed), and St. John’s wort. Giving potent inducers of CYP3A with boceprevir or telaprevir may lead to lower exposure and loss of efficacy of both protease inhibitors.
EMERGING THERAPIES FOR HCV
Thanks to a better understanding of the biology of HCV infection, the effort to develop new therapeutic agents started to focus on targeting specific steps of the viral life cycle, including attachment, entry into cells, replication, and release.24
Currently, more than 50 clinical trials are evaluating new direct-acting antivirals to treat HCV infection.25 Monoclonal and polyclonal antibodies that target the molecular process involved in HCV attachment and entry are being developed.26 The nonstructural protein NS5B (RNA polymerase) is intimately involved in viral replication and represents a promising target.27 Several nucleosides and nonnucleoside protease inhibitors have already entered clinical trials.
The low fidelity of the HCV replication machinery leads to a very high mutation rate, thus enabling the virus to quickly develop mutations that resist agents targeting viral enzymes.28 Therefore, a novel approach is to target host cofactors that are essential for HCV replication. An intriguing study by Lanford et al29 demonstrated that antagonizing microRNA-122 (the most abundant microRNA in the liver and an essential cofactor for viral RNA replication) by the oligonucleotide SPC3649 caused marked and prolonged reduction of HCV viremia in chronically infected chimpanzees.29
Although we are still in the early stages of drug development, the future holds great promise for newer drugs to improve the sustained virologic response, shorten the duration of treatment, improve tolerability with interferon-sparing regimens, and decrease viral resistance.
FUTURE PERSPECTIVES
With the introduction of the first direct-acting antiviral medications for HCV (boceprevir and telaprevir), 2011 will be marked as the year that changed hepatitis C treatment for the better. Triple therapy with pegylated interferon, ribavirin, and either boceprevir or telaprevir has the potential for increasing the rate of sustained virologic response to around 70% in previously untreated patients and 65% in previously treated patients who are infected with HCV genotype 1. The IL28B polymorphisms appear to play a role in the rate of sustained virologic response achieved with triple therapy, with preliminary data showing a better response rate in patients who have the CC genotype.17
These drugs will add up to $50,000 to the cost of treating hepatitis C virus infection, depending on the drug used and the length of treatment. However, they may be well worth it if they prevent liver failure and the need for transplantation.
Many questions remain, such as how to use these new regimens to treat special patient populations—for example, those with a recurrence of HCV infection after liver transplantation, those co-infected with HCV and human immunodeficiency virus, and those infected with HCV genotypes other than genotype 1.
Other direct-acting antiviral agents that specifically target the replication cycle of HCV are currently in clinical development. In fact, the future has already started with the release of the Interferon-Free Regimen for the Management of HCV (INFORM-1) study results.30 This was the first trial to evaluate an interferon-free regimen for patients with chronic HCV infection using two direct-acting antiviral drugs (the protease inhibitor danoprevir and the polymerase inhibitor RG7128), with promising results.
- Poordad F, McCone J, Bacon BR, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1195–1206.
- Bacon BR, Gordon SC, Lawitz E, et al. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1207–1217.
- Jacobson IM, McHutchison JG, Dusheiko G, et al; for the ADVANCE Study Team. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med 2011; 364:2405–2416.
- Sherman KE, Flamm SL, Afdhal NH, et al; for the ILLUMINATE Study Team. Response-guided telaprevir combination treatment for hepatitis C virus infection. N Engl J Med 2011; 365:1014–1024.
- Zeuzem S, Andreone P, Pol S, et al; for the REALIZE Study Team. Telaprevir for retreatment of HCV infection. N Engl J Med 2011; 364:2417–2428.
- Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 2006; 144:705–714.
- Mitchell AE, Colvin HM, Palmer Beasley R. Institute of Medicine recommendations for the prevention and control of hepatitis B and C. Hepatology 2010; 51:729–733.
- Kim WR. The burden of hepatitis C in the United States. Hepatology 2002; 36:S30–S34.
- Marcellin P, Asselah T, Boyer N. Fibrosis and disease progression in hepatitis C. Hepatology 2002; 36:S47–S56.
- Seeff LB. Natural history of chronic hepatitis C. Hepatology 2002; 36:S35–S46.
- Lavanchy D. The global burden of hepatitis C. Liver Int 2009; 29(suppl 1):74–81.
- National Institutes of Health Consensus Development Conference Statement: Management of hepatitis C: 2002—June 10–12, 2002. Hepatology 2002; 36:S3–S20.
- Pearlman BL, Traub N. Sustained virologic response to antiviral therapy for chronic hepatitis C virus infection: a cure and so much more. Clin Infect Dis 2011; 52:889–900.
- Hoofnagle JH, Seeff LB. Peginterferon and ribavirin for chronic hepatitis C. N Engl J Med 2006; 355:2444–2451.
- Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature 2009; 461:399–401.
- Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet 2009; 41:1100–1104.
- Thompson AJ, Muir AJ, Sulkowski MS, et al. Interleukin-28B polymorphism improves viral kinetics and is the strongest pretreatment predictor of sustained virologic response in genotype 1 hepatitis C virus. Gastroenterology 2010; 139:120–129.e118.
- Nielsen SU, Bassendine MF, Burt AD, Bevitt DJ, Toms GL. Characterization of the genome and structural proteins of hepatitis C virus resolved from infected human liver. J Gen Virol 2004; 85:1497–1507.
- Penin F, Dubuisson J, Rey FA, Moradpour D, Pawlotsky JM. Structural biology of hepatitis C virus. Hepatology 2004; 39:5–19.
- Nelson DR. The role of triple therapy with protease inhibitors in hepatitis C virus genotype 1 naive patients. Liver Int 2011; 31(suppl 1):53–57.
- Pawlotsky JM. Treatment failure and resistance with direct-acting antiviral drugs against hepatitis C virus. Hepatology 2011; 53:1742–1751.
- Monto A, Schooley RT, Lai JC, et al. Lessons from HIV therapy applied to viral hepatitis therapy: summary of a workshop. Am J Gastroenterol 2010; 105:989–1004.
- McCown MF, Rajyaguru S, Kular S, Cammack N, Najera I. GT-1a or GT-1b subtype-specific resistance profiles for hepatitis C virus inhibitors telaprevir and HCV-796. Antimicrob Agents Chemother 2009; 53:2129–2132.
- Cholongitas E, Papatheodoridis GV. Review article: novel therapeutic options for chronic hepatitis C. Aliment Pharmacol Ther 2008; 27:866–884.
- Naggie S, Patel K, McHutchison J. Hepatitis C virus directly acting antivirals: current developments with NS3/4A HCV serine protease inhibitors. J Antimicrob Chemother 2010; 65:2063–2069.
- Mir HM, Birerdinc A, Younossi ZM. Monoclonal and polyclonal antibodies against the HCV envelope proteins. Clin Liver Dis 2009; 13:477–486.
- Birerdinc A, Younossi ZM. Emerging therapies for hepatitis C virus. Expert Opin Emerg Drugs 2010; 15:535–544.
- Khattab MA. Targeting host factors: a novel rationale for the management of hepatitis C virus. World J Gastroenterol 2009; 15:3472–3479.
- Lanford RE, Hildebrandt-Eriksen ES, Petri A, et al. Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science 2010; 327:198–201.
- Gane EJ, Roberts SK, Stedman CA, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet 2010; 376:1467–1475.
- Poordad F, McCone J, Bacon BR, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1195–1206.
- Bacon BR, Gordon SC, Lawitz E, et al. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med 2011; 364:1207–1217.
- Jacobson IM, McHutchison JG, Dusheiko G, et al; for the ADVANCE Study Team. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med 2011; 364:2405–2416.
- Sherman KE, Flamm SL, Afdhal NH, et al; for the ILLUMINATE Study Team. Response-guided telaprevir combination treatment for hepatitis C virus infection. N Engl J Med 2011; 365:1014–1024.
- Zeuzem S, Andreone P, Pol S, et al; for the REALIZE Study Team. Telaprevir for retreatment of HCV infection. N Engl J Med 2011; 364:2417–2428.
- Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 2006; 144:705–714.
- Mitchell AE, Colvin HM, Palmer Beasley R. Institute of Medicine recommendations for the prevention and control of hepatitis B and C. Hepatology 2010; 51:729–733.
- Kim WR. The burden of hepatitis C in the United States. Hepatology 2002; 36:S30–S34.
- Marcellin P, Asselah T, Boyer N. Fibrosis and disease progression in hepatitis C. Hepatology 2002; 36:S47–S56.
- Seeff LB. Natural history of chronic hepatitis C. Hepatology 2002; 36:S35–S46.
- Lavanchy D. The global burden of hepatitis C. Liver Int 2009; 29(suppl 1):74–81.
- National Institutes of Health Consensus Development Conference Statement: Management of hepatitis C: 2002—June 10–12, 2002. Hepatology 2002; 36:S3–S20.
- Pearlman BL, Traub N. Sustained virologic response to antiviral therapy for chronic hepatitis C virus infection: a cure and so much more. Clin Infect Dis 2011; 52:889–900.
- Hoofnagle JH, Seeff LB. Peginterferon and ribavirin for chronic hepatitis C. N Engl J Med 2006; 355:2444–2451.
- Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature 2009; 461:399–401.
- Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet 2009; 41:1100–1104.
- Thompson AJ, Muir AJ, Sulkowski MS, et al. Interleukin-28B polymorphism improves viral kinetics and is the strongest pretreatment predictor of sustained virologic response in genotype 1 hepatitis C virus. Gastroenterology 2010; 139:120–129.e118.
- Nielsen SU, Bassendine MF, Burt AD, Bevitt DJ, Toms GL. Characterization of the genome and structural proteins of hepatitis C virus resolved from infected human liver. J Gen Virol 2004; 85:1497–1507.
- Penin F, Dubuisson J, Rey FA, Moradpour D, Pawlotsky JM. Structural biology of hepatitis C virus. Hepatology 2004; 39:5–19.
- Nelson DR. The role of triple therapy with protease inhibitors in hepatitis C virus genotype 1 naive patients. Liver Int 2011; 31(suppl 1):53–57.
- Pawlotsky JM. Treatment failure and resistance with direct-acting antiviral drugs against hepatitis C virus. Hepatology 2011; 53:1742–1751.
- Monto A, Schooley RT, Lai JC, et al. Lessons from HIV therapy applied to viral hepatitis therapy: summary of a workshop. Am J Gastroenterol 2010; 105:989–1004.
- McCown MF, Rajyaguru S, Kular S, Cammack N, Najera I. GT-1a or GT-1b subtype-specific resistance profiles for hepatitis C virus inhibitors telaprevir and HCV-796. Antimicrob Agents Chemother 2009; 53:2129–2132.
- Cholongitas E, Papatheodoridis GV. Review article: novel therapeutic options for chronic hepatitis C. Aliment Pharmacol Ther 2008; 27:866–884.
- Naggie S, Patel K, McHutchison J. Hepatitis C virus directly acting antivirals: current developments with NS3/4A HCV serine protease inhibitors. J Antimicrob Chemother 2010; 65:2063–2069.
- Mir HM, Birerdinc A, Younossi ZM. Monoclonal and polyclonal antibodies against the HCV envelope proteins. Clin Liver Dis 2009; 13:477–486.
- Birerdinc A, Younossi ZM. Emerging therapies for hepatitis C virus. Expert Opin Emerg Drugs 2010; 15:535–544.
- Khattab MA. Targeting host factors: a novel rationale for the management of hepatitis C virus. World J Gastroenterol 2009; 15:3472–3479.
- Lanford RE, Hildebrandt-Eriksen ES, Petri A, et al. Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science 2010; 327:198–201.
- Gane EJ, Roberts SK, Stedman CA, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet 2010; 376:1467–1475.
KEY POINTS
- Standard care with the combination of pegylated interferon and ribavirin produces a sustained virologic response in about 40% of patients infected with HCV genotype 1, the most prevalent genotype in North America.
- New phase 3 trials showed that the addition of an oral protease inhibitor (boceprevir or telaprevir) increased the sustained virologic response rates to 70% in patients infected with HCV genotype 1.
- Boceprevir and telaprevir must be used in combination with pegylated interferon and ribavirin; they should not be used as monotherapy because of concern about the development of drug-resistant mutations.
- The main side effects of boceprevir were anemia and dysgeusia. Adverse events associated with telaprevir included rash, pruritus, anemia, and diarrhea.
Update on contraceptive options: A case-based discussion
Contraceptive counseling is both an art and a science. The role of the health care provider is to determine the patient’s medical eligibility and match her preferences and lifestyle to an appropriate method for both contraceptive and potentially noncontraceptive benefits, while minimizing the risk of unintended pregnancy.
Women throughout the range of reproductive years need appropriate counseling and education on hormones, the menstrual cycle, and the efficacy of contraception as part of their routine gynecologic evaluation. Issues of access to birth control, cost, possible side effects, and actual effectiveness of methods are important to discuss.
In this paper we will discuss common clinical practice case scenarios to illustrate contraceptive counseling and management, including:
- Perimenopausal women
- Women with thrombophilia
- Women who contemplate becoming pregnant in the future
- Women with psychiatric illness
- Women with hypertension.
HALF OF ALL PREGNANCIES ARE UNPLANNED
Although many contraceptive options are available, 48% of all pregnancies in the United States are unintended.1 In 2009, the national teen birth rate was 39.1 births per 1,000 girls and women age 15 to 19 years, which was 37% lower than in 1991.2 Still, African American and Hispanic teenagers living in southern states have disproportionately higher rates.
The rate of unintended pregnancy is a little lower at the older end of the reproductive age range, but still high: 35% of all pregnancies in women over 40 years old are also unintended.2
To find out why these numbers are so high, in 2007 the US Centers for Disease Control and Prevention (CDC) conducted a survey3 that included 8,000 women reporting unintended pregnancy who had not used contraception. Of these, 39% were married. Surprisingly, more than one-third of women said they did not know they could get pregnant when they did.3
WHAT’S NEW IN CONTRACEPTION?
The “pill” was approved by the US Food and Drug Administration (FDA) more than 50 years ago, and it is still the most commonly used contraceptive method (followed by surgical sterilization). Enovid, the pill formulated by Dr. John Rock and Dr. Gregory Pincus in the 1950s, contained 150 μg of mestranol (equivalent to 90 μg of ethinyl estradiol) and 9.85 mg of norethynodrel, a very potent progestin. Our current oral contraceptive pills contain much lower hormone doses and have fewer androgenic side effects.4
In May 2010, the CDC and the World Health Organization (WHO) updated their safety guidelines for all hormonal contraceptives and the use of these agents in patients with various medical and family histories. They ranked contraceptive methods from those with no restriction to those with unacceptable risk to their use. This document can be accessed at www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm.5
New developments in oral contraceptives are notably in the 19-nortestosterone derivatives, the family that includes the second-generation progestogens already available such as norgestimate (contained in Ortho-Cyclen) and norethindrone (contained in Loestrin). A newer progestin, dienogest, is available in a preparation that also contains estradiol valerate (Natazia). Drospirenone, which is similar to spironolactone, is contained in Yaz, Yasmin, and newer products that also contain levomefolate calcium (Beyaz, Safyral).
LoLoestrin Fe, which contains active pills containing 10 μg of ethinyl estradiol and 1 mg of norethindrone and placebo pills with 75 mg of ferrous fumarate, was recently approved by the FDA and offers an ultra-low dose of estrogen.
Depot medroxyprogesterone acetate now comes in a 104-mg suspension for subcutaneous injection every 3 months; it is called depo-subQ provera 104. Standard medroxyprogesterone acetate 150 mg for intramuscular injection every 3 months (Depo-Provera) is still available and has gone generic. The newer product offers the advantages of lower dose and less weight-gain. Also, it allows capable and willing patients to self-administer their contraceptives. However, it is more expensive—$ 104 per injection for a patient without insurance at Cleveland Clinic, compared with $46 for Depo-Provera and $10 for the generic intramuscular preparation for a patient with insurance.
A new option for emergency contraception, ulipristal (ella) is a progesterone antagonist-agonist available only by prescription. Taken in a single oral dose of 30 μg, it is effective for up to 120 hours after unprotected intercourse. It joins Plan B (levonorgestrel 1.5 mg in a single dose) and Next Choice (two doses of levonorgestrel 0.75 mg each), which are available over-the-counter for women age 17 years or older, and by prescription for those 16 years and younger, for use up to 72 hours after unprotected intercourse.
CASE 1: CONTRACEPTION IN PERIMENOPAUSE
A 48-year-old attorney who has had two children complains of irregular menstrual cycles and of occasional hot flashes at night that wake her from sleep. She keeps a menstrual calendar; it shows her last menstrual period was 3 months ago. She took oral contraceptives for 15 years before she had her first child. She is using condoms intermittently for contraception. Her body mass index is normal at 24 kg/m2, and she does not smoke. How do you counsel her?
A variety of hormonal options
This healthy perimenopausal woman has a variety of hormonal contraception options that would have the added benefit of regulating her menstrual cycle or suppressing it altogether. These include the levonorgestrel intrauterine system (Mirena IUS), various injectable products (such as Depo-Provera or the newer depo-subQ provera 104), contraceptive pills, the Ortho Evra contraceptive patch, and the vaginal contraceptive ring (NuvaRing). Of these, low-dose birth control pills may be the best option, as they would help with cycle control, offer contraception, and better regulate hormonal fluctuations to reduce her hot flashes.
Hormonal contraception can safely be used in women in their 30s and 40s, and often until menopause if the benefit outweighs the risk.
An estradiol valerate-dienogest oral contraceptive with a quadriphasic dosing schedule (Natazia) has been studied in women up to age 50. Although it was approved in 2010 in the United States, this pill has been used in Europe since the 1990s. The 26 active pills contain tapering doses of the active drugs, with the aim of mimicking the natural menstrual cycle, similar to triphasic pills. Estradiol valerate is a bioidentical estrogen, as it is rapidly metabolized to estradiol (E2), which is identical to 17-beta estradiol and estrone (E3) produced by the ovary. A dose of 2 mg of estradiol valerate is equivalent to 10 μg of ethinyl estradiol, which is the estrogen component in most other oral contraceptives. Low-dose pills by definition contain less than 50 μg of ethinyl estradiol. Dienogest, the progesterone component, has a 17-cyanomethyl group that accounts for its strongly progestogenic and weakly antiandrogenic properties.
All oral hormonal contraceptives can increase triglycerides by inducing the CYP450 system in the liver. However, in clinical trials, estradiol valerate-dienogest also caused other changes in lipid metabolism, such as a nonsignificant increase in high-density lipoprotein cholesterol and a slight reduction in low-density lipoprotein cholesterol and lipoprotein(a) compared with ethinyl estradiol-levonorgestrel preparations.6
It is important to advise patients that, compared with users of other oral contraceptives, estradiol valerate-dienogest users may experience fewer days of menstrual bleeding and more cycles without withdrawal bleeding. This product can therefore be an effective alternative for women with menorrhagia.
All classes of hormonal contraception carry a similar risk of side effects, such as headache, breast tenderness, nausea, irregular bleeding, and mood changes. Some women have no side effects.
CASE 2: THROMBOPHILIA
A 39-year-old woman with a body mass index of 31 kg/m2 (obese) has a history of protein S deficiency with active lower-extremity deep vein thrombosis, for which she is taking warfarin (Coumadin). She experiences menorrhagia and dysmenorrhea due to intramural fibroids and possible adenomyosis seen on transvaginal ultrasonography and confirmed by magnetic resonance imaging. Hysteroscopy reveals no polyps or submucosal fibroids. An endometrial biopsy is negative for malignancy.
She desires contraception. How do you counsel her?
Estrogens are contraindicated—except, perhaps, in select cases
This patient has many reasons for heavy bleeding. She is on warfarin, which effectively inhibits synthesis of vitamin K-dependent coagulation factor. She also has fibroids and adenomyosis. The latter is a difficult condition to control, as the location of the intramuscular glands makes treatments such as ablation, dilation and curettage, and oral agents ineffective.
All estrogen-containing formulations (pills, ring, patch) are contraindicated in women with acute venous thromboembolism (VTE) and known thrombophilia. A newer agent approved for treating menorrhagia (not for contraception), tranexamic acid (Lysteda), also carries a contraindication for patients with thrombophilia or history of VTE; however, the evidence for the latter is controversial.7
The updated CDC guidelines for the use of hormonal contraceptives state that patients who receive anticoagulation for at least 3 months and who have no history of VTE or a low risk of recurrent VTE (no evidence of active cancer, no known thrombophilia) may use estrogen-containing contraceptives in select cases (category 3—theoretical risk outweighs benefits, but not an absolute contraindication).5 Although this is not common clinical practice, select patients may benefit from menstrual cycle control while receiving anticoagulation. However, other contraceptive alternatives are preferred if possible.
Progestin-only treatments such as the Mirena IUS (if the fibroids do not distort the uterine cavity) and the etonogestrel implant (Implanon) are nonsurgical options that may reduce menorrhagia and are safer alternatives for patients with thrombophilia.
The Paragard (copper) intrauterine device would provide nonhormonal contraception without diminishing menorrhagia. Obviously, barrier methods (which are less effective than hormonal contraception) can be suggested for contraception alone. A viable option for women finished with childbearing is hysterectomy, which provides contraceptive benefit and definitive treatment of menorrhagia due to adenomyosis.
Laboratory screening for VTE is not required before starting estrogen-containing contraceptives. However, one should take a detailed history and inquire about VTE events or a family history of recurrent VTE.
VTE rates among reproductive-age women are 4 to 5 per 10,000 women per year.8 The rate of VTE in oral contraceptive users is estimated as 9 to 10 per 10,000 women per year.9 However, rates of VTE associated with pregnancy and postpartum states are exponentially greater. Although recent studies have shown some discrepancy in rates of VTE across different classes of progestins,10,11 the absolute risk of VTE with hormonal contraceptives is very low.
In December 2011, an FDA panel voted 15 to 11 that the benefits of drospirenone-containing contraceptives (eg, Yaz, Yasmin, Beyaz, Safyral), such as preventing pregnancy, outweigh the potential risk. However, product labeling may change in the future to more accurately reflect the risk-benefit ratio. Stay tuned for better-designed trials to further assess VTE risk across progestins.
Health care providers should engage patients in an informed discussion about all risks and benefits of hormonal contraceptives and note this risk of VTE is higher in gravid women.
CASE 3: FUTURE FERTILITY
A 30-year-old surgical resident who has never been pregnant comes for her annual examination. She currently desires birth control but would like to be pregnant 1 to 2 years from now. She has no history of significant medical illness. Her body mass index is 23 kg/m2, and she takes no medications. How do you counsel her?
Many options; also consider folic acid
Effective counseling leads to patient-centered decision-making for all treatments and procedures. Contraceptive counseling should elicit the patient’s perspective about hormonal methods and educate her on efficacy, proper use, and common adverse effects.
Contraception should fit the patient’s lifestyle. Questions as simple as “Are you a good pill-taker?” or “Are you comfortable with injections?” will help you and the patient assess what will work effectively and will maintain good adherence.
Deciding on a contraceptive option that is cost-effective is crucial, particularly for many young women or adolescents. Many oral contraceptives are widely available as generic formulations for less than $10 per month. Although generic drugs are not required to be 100% bioequivalent to their brand-name counterparts, they can provide a more economical option. For a complete guide to different hormonal contraceptive formulations, we suggest Choosing a birth control method, available on the Web site of the Association of Reproductive Health Professionals at www.arhp.org/upload-Docs/choosingqrg.pdf.12
As discussed earlier, half of all pregnancies are unplanned, and so women of childbearing age should be ingesting 400 μg of folic acid daily. Debate exists as to whether Americans who eat a balanced diet need a multivitamin.13 However, there is no debate about folic acid, which is proven to prevent neural tube defects. Newer formulations of ethinyl estradiol-drospirenone (Beyaz, Safyral) now contain an active form of folic acid (levomefolate calcium 451 mg in each pill). For the above patient who needs contraception and is willing to take birth control, the addition of folic acid provides an essential element in preconception counseling.
Regardless of the current contraceptive choice, patients who actively desire pregnancy should take a prenatal vitamin that contains folic acid and iron.
In addition to combined oral contraceptives, other options for this patient include medroxyprogesterone acetate (intramuscular or subcutaneous), NuvaRing, or intrauterine devices. The Ortho Evra patch is also an option for this patient. However, since 2008 the patch has carried an FDA warning that the risk of VTE is twice as high with this product than with oral contraceptives that contain 30 μg of ethinyl estradiol plus levonorgestrel.14 Postmarketing data did not show any higher risk of VTE in patch users compared with oral contraceptive users less than 40 years of age, however.15
CASE 4: PSYCHIATRIC ILLNESS
A 21-year-old woman who has bipolar II disorder comes to your office for her annual gynecologic evaluation. She has one sexual partner and desires oral contraceptive pills. Lithium treatment has failed for her, but her condition is stable on carbamazepine (Tegretol). She asks if it is true that women can still get pregnant while on the birth control pill. How do you counsel her?
Possible interactions with psychiatric drugs
Like the woman in case 3, this patient has many options, including estrogen-containing pills, the vaginal ring, the patch, injectable contraceptives, and intrauterine devices.
Certain antiepileptic, antipsychotic, or headache medications such as carbamezapine, phenytoin (Dilantin), oxcarbazepine (Trileptal), and topiramate (Topamax) decrease levels of hormonal contraceptives by induction of the CYP450 enzymes. Conversely, it is suggested that lamotrigine (Lamictal) levels decrease by up to 49% while patients concomitantly take oral contraceptive pills, which can induce seizure activity.16 Also, antibiotics such as rifampin (Rifadin) and even herbs such as St. John’s Wort can decrease the effectiveness of hormonal contraceptives by increasing their metabolism.
On the positive side, depot medroxyprogesterone acetate raises the seizure threshold by a mechanism attributed to high levels of progestins and is a better option for epileptic patients. A bulletin of the American College of Gynecologists addresses the paucity of data on hormonal treatments in depressed patients. However, some evidence points to slight improvement of depressive symptoms after 1 year in patients who took Depo-Provera compared with those who discontinued the drug.17
The Pearl index, a measure of contraceptive efficacy
We refer to the Pearl index when answering our patients’ questions about contraceptive efficacy. The Pearl index is defined as the number of unintended pregnancies per 100 women per year. The typical (or actual) effectiveness for each contraceptive method is quoted rather than the theoretical (perfect-use) efficacy.
We suggest simplifying this discussion with patients. For example, for every 100 women using male condoms for contraception, 15 women have unintended pregnancies per year. With hormonal contraceptives (pill, patch, or ring), for every 100 women there are 8 per year with unintended pregnancy, 3 of 100 with Depo-Provera, and less than 1 in 100 using intrauterine devices or female or male sterilization.18
Efficacy decreases (and the failure rate increases) with frequency of intercourse, irregular menstrual cycles, missed pills, improper dosing, and drug-drug interactions as described above.
CASE 5: HYPERTENSION
A 33-year-old woman who has been pregnant twice experienced preeclampsia in her last pregnancy, and now her blood pressure is consistently approximately 140/90 mm Hg on multiple office visits and ambulatory monitoring. She desires contraception. How do you counsel her?
Avoid estrogen-containing products
According to the WHO and CDC guidelines,5 women with controlled or uncontrolled hypertension should not be offered combined oral contraceptives, the patch, or the ring (category 3—theoretical or proven risks outweigh the benefits, and category 4 for systolic blood pressure greater than 160 mm Hg or diastolic blood pressure greater than 100 mm Hg).
The progesterone-only pill (“mini pill”), medroxyprogesterone acetate (intramuscular or subcutaneous), Mirena IUS, the copper intrauterine device, and the etonogestrel implant are all safer options.
A small subset of patients develop elevated blood pressure after starting hormonal contraceptives. Estrogen-containing hormones can increase the liver’s output of angiotensinogen, which is a renin substrate that activates the renin-angiotensin-aldosterone system. If this becomes clinically apparent, these patients should refrain from estrogen-containing products and use progestin-only formulations as a safer alternative.
Patients with isolated elevated hypertriglyceridemia should avoid oral contraceptives. However, the patch, the ring, and progestin-only methods may be acceptable.
Diabetic patients with microvascular complications of retinopathy or nephropathy and any patient with macrovascular disease (stroke, cardiovascular disease) should not be offered estrogen-containing contraception.
- Finer LB, Henshaw SK. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspect Sex Reprod Health 2006; 38:90–96.
- Centers for Disease Control and Prevention. Vital signs: teenage pregnancy—United States 1991–2009. MMWR 2011; 60:414–420. www.cdc.gov/mmwr/preview/mmwrhtml/mm6013a5.htm. Accessed January 10, 2012.
- Nettleman MD, Chung H, Brewer J, Ayoola A, Reed PL. Reasons for unprotected intercourse: analysis of the PRAMS survey. Contraception 2007; 75:361–366.
- Nelson A. New low-dose, extended-cycle pills with levonorgestrel and ethinyl estradiol: an evolutionary step on birth control. Int J Womens Health 2010; 2:99–106.
- US Centers for Disease Control and Prevention. Appendix L. Summary of classifications for hormonal contraceptive methods and intrauterine devices. MMWR 2010; 59( RR04):76–81. www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm. Accessed January 10, 2012.
- Wiegratz L, Lee JH, Kutchera E, et al. Effect of dienogest-containing oral contraceptives in lipid metabolism. Contraception 2002; 65:223–229.
- Sundström A, Seaman H, Kieler H, Alfredsson L. The risk of venous thromboembolism associated with the use of tranexamic acid and other drugs used to treat menorrhagia: a case-control study using the General Practice Research Database. Br J Obstet Gynecol 2009; 116:91–97.
- Heinemann L, Dinger JC. Range of published estimates of venous thromboembolism incidence in young women. Contraception 2007; 75:328–336.
- Dinger JC, Heinemann LA, Kühl-Habich D. The safety of a drospirenone-containing oral contraceptive: final results from the European Active Surveillance Study on oral contraceptives based on 142,475 women-years of observation. Contraception 2007; 75:344–354.
- Parkin L, Sharples K, Hernandez RK, Jick SS. Risk of venous thromboembolism in users of oral contraceptives containing drospirenone or levonorgestrel: nested case-control study based on UK General Practice Research Database. BMJ 2011; 342:d2139.
- Jick SS, Hernandez RK. Risk of non-fatal venous thromboembolism in women using oral contraceptives containing drospirenone compared with women using oral contraceptives containing levonorgestrel: case-control study using United States claims data. BMJ 2011; 342:d2151.
- Association of Reproductive Health Professionals (ARHP). Choosing a birth control method. www.arhp.org/uploadDocs/choosingqrg.pdf. Accessed January 10, 2012.
- Caballero B. Should healthy people take a multivitamin? Cleve Clin J Med 2010; 77:656–657.
- US Food and Drug Administration. Ortho Evra questions and answers (1/18/2008). www.fda.gov/Drugs/DrugSafety/Postmarket-DrugSafetyInformationforPatientsandProviders/ucm110403.htm. Accessed January 10, 2012.
- Jick SS, Hagberg KW, Hernandez RK, Kaye JA. Postmarketing study of ORTHO EVRA and levonorgestrel oral contraceptives containing hormonal contraceptives with 30 mcg of ethinyl estradiol in relation to nonfatal venous thromboembolism. Contraception 2010; 81:16–21.
- Sabers A, Ohman I, Christensen J, Tomson T. Oral contraceptives reduce lamotrigine plasma levels. Neurology 2003; 61:570–571.
- Westhoff C, Truman C, Kalmuss D, et al; Depressive symptoms and Depo-Provera. Contraception 1998; 57:237–240.
- Trusell Wynn LL. Reducing unintended pregnancy in the United States. Contraception 2008; 77:1–5.
Contraceptive counseling is both an art and a science. The role of the health care provider is to determine the patient’s medical eligibility and match her preferences and lifestyle to an appropriate method for both contraceptive and potentially noncontraceptive benefits, while minimizing the risk of unintended pregnancy.
Women throughout the range of reproductive years need appropriate counseling and education on hormones, the menstrual cycle, and the efficacy of contraception as part of their routine gynecologic evaluation. Issues of access to birth control, cost, possible side effects, and actual effectiveness of methods are important to discuss.
In this paper we will discuss common clinical practice case scenarios to illustrate contraceptive counseling and management, including:
- Perimenopausal women
- Women with thrombophilia
- Women who contemplate becoming pregnant in the future
- Women with psychiatric illness
- Women with hypertension.
HALF OF ALL PREGNANCIES ARE UNPLANNED
Although many contraceptive options are available, 48% of all pregnancies in the United States are unintended.1 In 2009, the national teen birth rate was 39.1 births per 1,000 girls and women age 15 to 19 years, which was 37% lower than in 1991.2 Still, African American and Hispanic teenagers living in southern states have disproportionately higher rates.
The rate of unintended pregnancy is a little lower at the older end of the reproductive age range, but still high: 35% of all pregnancies in women over 40 years old are also unintended.2
To find out why these numbers are so high, in 2007 the US Centers for Disease Control and Prevention (CDC) conducted a survey3 that included 8,000 women reporting unintended pregnancy who had not used contraception. Of these, 39% were married. Surprisingly, more than one-third of women said they did not know they could get pregnant when they did.3
WHAT’S NEW IN CONTRACEPTION?
The “pill” was approved by the US Food and Drug Administration (FDA) more than 50 years ago, and it is still the most commonly used contraceptive method (followed by surgical sterilization). Enovid, the pill formulated by Dr. John Rock and Dr. Gregory Pincus in the 1950s, contained 150 μg of mestranol (equivalent to 90 μg of ethinyl estradiol) and 9.85 mg of norethynodrel, a very potent progestin. Our current oral contraceptive pills contain much lower hormone doses and have fewer androgenic side effects.4
In May 2010, the CDC and the World Health Organization (WHO) updated their safety guidelines for all hormonal contraceptives and the use of these agents in patients with various medical and family histories. They ranked contraceptive methods from those with no restriction to those with unacceptable risk to their use. This document can be accessed at www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm.5
New developments in oral contraceptives are notably in the 19-nortestosterone derivatives, the family that includes the second-generation progestogens already available such as norgestimate (contained in Ortho-Cyclen) and norethindrone (contained in Loestrin). A newer progestin, dienogest, is available in a preparation that also contains estradiol valerate (Natazia). Drospirenone, which is similar to spironolactone, is contained in Yaz, Yasmin, and newer products that also contain levomefolate calcium (Beyaz, Safyral).
LoLoestrin Fe, which contains active pills containing 10 μg of ethinyl estradiol and 1 mg of norethindrone and placebo pills with 75 mg of ferrous fumarate, was recently approved by the FDA and offers an ultra-low dose of estrogen.
Depot medroxyprogesterone acetate now comes in a 104-mg suspension for subcutaneous injection every 3 months; it is called depo-subQ provera 104. Standard medroxyprogesterone acetate 150 mg for intramuscular injection every 3 months (Depo-Provera) is still available and has gone generic. The newer product offers the advantages of lower dose and less weight-gain. Also, it allows capable and willing patients to self-administer their contraceptives. However, it is more expensive—$ 104 per injection for a patient without insurance at Cleveland Clinic, compared with $46 for Depo-Provera and $10 for the generic intramuscular preparation for a patient with insurance.
A new option for emergency contraception, ulipristal (ella) is a progesterone antagonist-agonist available only by prescription. Taken in a single oral dose of 30 μg, it is effective for up to 120 hours after unprotected intercourse. It joins Plan B (levonorgestrel 1.5 mg in a single dose) and Next Choice (two doses of levonorgestrel 0.75 mg each), which are available over-the-counter for women age 17 years or older, and by prescription for those 16 years and younger, for use up to 72 hours after unprotected intercourse.
CASE 1: CONTRACEPTION IN PERIMENOPAUSE
A 48-year-old attorney who has had two children complains of irregular menstrual cycles and of occasional hot flashes at night that wake her from sleep. She keeps a menstrual calendar; it shows her last menstrual period was 3 months ago. She took oral contraceptives for 15 years before she had her first child. She is using condoms intermittently for contraception. Her body mass index is normal at 24 kg/m2, and she does not smoke. How do you counsel her?
A variety of hormonal options
This healthy perimenopausal woman has a variety of hormonal contraception options that would have the added benefit of regulating her menstrual cycle or suppressing it altogether. These include the levonorgestrel intrauterine system (Mirena IUS), various injectable products (such as Depo-Provera or the newer depo-subQ provera 104), contraceptive pills, the Ortho Evra contraceptive patch, and the vaginal contraceptive ring (NuvaRing). Of these, low-dose birth control pills may be the best option, as they would help with cycle control, offer contraception, and better regulate hormonal fluctuations to reduce her hot flashes.
Hormonal contraception can safely be used in women in their 30s and 40s, and often until menopause if the benefit outweighs the risk.
An estradiol valerate-dienogest oral contraceptive with a quadriphasic dosing schedule (Natazia) has been studied in women up to age 50. Although it was approved in 2010 in the United States, this pill has been used in Europe since the 1990s. The 26 active pills contain tapering doses of the active drugs, with the aim of mimicking the natural menstrual cycle, similar to triphasic pills. Estradiol valerate is a bioidentical estrogen, as it is rapidly metabolized to estradiol (E2), which is identical to 17-beta estradiol and estrone (E3) produced by the ovary. A dose of 2 mg of estradiol valerate is equivalent to 10 μg of ethinyl estradiol, which is the estrogen component in most other oral contraceptives. Low-dose pills by definition contain less than 50 μg of ethinyl estradiol. Dienogest, the progesterone component, has a 17-cyanomethyl group that accounts for its strongly progestogenic and weakly antiandrogenic properties.
All oral hormonal contraceptives can increase triglycerides by inducing the CYP450 system in the liver. However, in clinical trials, estradiol valerate-dienogest also caused other changes in lipid metabolism, such as a nonsignificant increase in high-density lipoprotein cholesterol and a slight reduction in low-density lipoprotein cholesterol and lipoprotein(a) compared with ethinyl estradiol-levonorgestrel preparations.6
It is important to advise patients that, compared with users of other oral contraceptives, estradiol valerate-dienogest users may experience fewer days of menstrual bleeding and more cycles without withdrawal bleeding. This product can therefore be an effective alternative for women with menorrhagia.
All classes of hormonal contraception carry a similar risk of side effects, such as headache, breast tenderness, nausea, irregular bleeding, and mood changes. Some women have no side effects.
CASE 2: THROMBOPHILIA
A 39-year-old woman with a body mass index of 31 kg/m2 (obese) has a history of protein S deficiency with active lower-extremity deep vein thrombosis, for which she is taking warfarin (Coumadin). She experiences menorrhagia and dysmenorrhea due to intramural fibroids and possible adenomyosis seen on transvaginal ultrasonography and confirmed by magnetic resonance imaging. Hysteroscopy reveals no polyps or submucosal fibroids. An endometrial biopsy is negative for malignancy.
She desires contraception. How do you counsel her?
Estrogens are contraindicated—except, perhaps, in select cases
This patient has many reasons for heavy bleeding. She is on warfarin, which effectively inhibits synthesis of vitamin K-dependent coagulation factor. She also has fibroids and adenomyosis. The latter is a difficult condition to control, as the location of the intramuscular glands makes treatments such as ablation, dilation and curettage, and oral agents ineffective.
All estrogen-containing formulations (pills, ring, patch) are contraindicated in women with acute venous thromboembolism (VTE) and known thrombophilia. A newer agent approved for treating menorrhagia (not for contraception), tranexamic acid (Lysteda), also carries a contraindication for patients with thrombophilia or history of VTE; however, the evidence for the latter is controversial.7
The updated CDC guidelines for the use of hormonal contraceptives state that patients who receive anticoagulation for at least 3 months and who have no history of VTE or a low risk of recurrent VTE (no evidence of active cancer, no known thrombophilia) may use estrogen-containing contraceptives in select cases (category 3—theoretical risk outweighs benefits, but not an absolute contraindication).5 Although this is not common clinical practice, select patients may benefit from menstrual cycle control while receiving anticoagulation. However, other contraceptive alternatives are preferred if possible.
Progestin-only treatments such as the Mirena IUS (if the fibroids do not distort the uterine cavity) and the etonogestrel implant (Implanon) are nonsurgical options that may reduce menorrhagia and are safer alternatives for patients with thrombophilia.
The Paragard (copper) intrauterine device would provide nonhormonal contraception without diminishing menorrhagia. Obviously, barrier methods (which are less effective than hormonal contraception) can be suggested for contraception alone. A viable option for women finished with childbearing is hysterectomy, which provides contraceptive benefit and definitive treatment of menorrhagia due to adenomyosis.
Laboratory screening for VTE is not required before starting estrogen-containing contraceptives. However, one should take a detailed history and inquire about VTE events or a family history of recurrent VTE.
VTE rates among reproductive-age women are 4 to 5 per 10,000 women per year.8 The rate of VTE in oral contraceptive users is estimated as 9 to 10 per 10,000 women per year.9 However, rates of VTE associated with pregnancy and postpartum states are exponentially greater. Although recent studies have shown some discrepancy in rates of VTE across different classes of progestins,10,11 the absolute risk of VTE with hormonal contraceptives is very low.
In December 2011, an FDA panel voted 15 to 11 that the benefits of drospirenone-containing contraceptives (eg, Yaz, Yasmin, Beyaz, Safyral), such as preventing pregnancy, outweigh the potential risk. However, product labeling may change in the future to more accurately reflect the risk-benefit ratio. Stay tuned for better-designed trials to further assess VTE risk across progestins.
Health care providers should engage patients in an informed discussion about all risks and benefits of hormonal contraceptives and note this risk of VTE is higher in gravid women.
CASE 3: FUTURE FERTILITY
A 30-year-old surgical resident who has never been pregnant comes for her annual examination. She currently desires birth control but would like to be pregnant 1 to 2 years from now. She has no history of significant medical illness. Her body mass index is 23 kg/m2, and she takes no medications. How do you counsel her?
Many options; also consider folic acid
Effective counseling leads to patient-centered decision-making for all treatments and procedures. Contraceptive counseling should elicit the patient’s perspective about hormonal methods and educate her on efficacy, proper use, and common adverse effects.
Contraception should fit the patient’s lifestyle. Questions as simple as “Are you a good pill-taker?” or “Are you comfortable with injections?” will help you and the patient assess what will work effectively and will maintain good adherence.
Deciding on a contraceptive option that is cost-effective is crucial, particularly for many young women or adolescents. Many oral contraceptives are widely available as generic formulations for less than $10 per month. Although generic drugs are not required to be 100% bioequivalent to their brand-name counterparts, they can provide a more economical option. For a complete guide to different hormonal contraceptive formulations, we suggest Choosing a birth control method, available on the Web site of the Association of Reproductive Health Professionals at www.arhp.org/upload-Docs/choosingqrg.pdf.12
As discussed earlier, half of all pregnancies are unplanned, and so women of childbearing age should be ingesting 400 μg of folic acid daily. Debate exists as to whether Americans who eat a balanced diet need a multivitamin.13 However, there is no debate about folic acid, which is proven to prevent neural tube defects. Newer formulations of ethinyl estradiol-drospirenone (Beyaz, Safyral) now contain an active form of folic acid (levomefolate calcium 451 mg in each pill). For the above patient who needs contraception and is willing to take birth control, the addition of folic acid provides an essential element in preconception counseling.
Regardless of the current contraceptive choice, patients who actively desire pregnancy should take a prenatal vitamin that contains folic acid and iron.
In addition to combined oral contraceptives, other options for this patient include medroxyprogesterone acetate (intramuscular or subcutaneous), NuvaRing, or intrauterine devices. The Ortho Evra patch is also an option for this patient. However, since 2008 the patch has carried an FDA warning that the risk of VTE is twice as high with this product than with oral contraceptives that contain 30 μg of ethinyl estradiol plus levonorgestrel.14 Postmarketing data did not show any higher risk of VTE in patch users compared with oral contraceptive users less than 40 years of age, however.15
CASE 4: PSYCHIATRIC ILLNESS
A 21-year-old woman who has bipolar II disorder comes to your office for her annual gynecologic evaluation. She has one sexual partner and desires oral contraceptive pills. Lithium treatment has failed for her, but her condition is stable on carbamazepine (Tegretol). She asks if it is true that women can still get pregnant while on the birth control pill. How do you counsel her?
Possible interactions with psychiatric drugs
Like the woman in case 3, this patient has many options, including estrogen-containing pills, the vaginal ring, the patch, injectable contraceptives, and intrauterine devices.
Certain antiepileptic, antipsychotic, or headache medications such as carbamezapine, phenytoin (Dilantin), oxcarbazepine (Trileptal), and topiramate (Topamax) decrease levels of hormonal contraceptives by induction of the CYP450 enzymes. Conversely, it is suggested that lamotrigine (Lamictal) levels decrease by up to 49% while patients concomitantly take oral contraceptive pills, which can induce seizure activity.16 Also, antibiotics such as rifampin (Rifadin) and even herbs such as St. John’s Wort can decrease the effectiveness of hormonal contraceptives by increasing their metabolism.
On the positive side, depot medroxyprogesterone acetate raises the seizure threshold by a mechanism attributed to high levels of progestins and is a better option for epileptic patients. A bulletin of the American College of Gynecologists addresses the paucity of data on hormonal treatments in depressed patients. However, some evidence points to slight improvement of depressive symptoms after 1 year in patients who took Depo-Provera compared with those who discontinued the drug.17
The Pearl index, a measure of contraceptive efficacy
We refer to the Pearl index when answering our patients’ questions about contraceptive efficacy. The Pearl index is defined as the number of unintended pregnancies per 100 women per year. The typical (or actual) effectiveness for each contraceptive method is quoted rather than the theoretical (perfect-use) efficacy.
We suggest simplifying this discussion with patients. For example, for every 100 women using male condoms for contraception, 15 women have unintended pregnancies per year. With hormonal contraceptives (pill, patch, or ring), for every 100 women there are 8 per year with unintended pregnancy, 3 of 100 with Depo-Provera, and less than 1 in 100 using intrauterine devices or female or male sterilization.18
Efficacy decreases (and the failure rate increases) with frequency of intercourse, irregular menstrual cycles, missed pills, improper dosing, and drug-drug interactions as described above.
CASE 5: HYPERTENSION
A 33-year-old woman who has been pregnant twice experienced preeclampsia in her last pregnancy, and now her blood pressure is consistently approximately 140/90 mm Hg on multiple office visits and ambulatory monitoring. She desires contraception. How do you counsel her?
Avoid estrogen-containing products
According to the WHO and CDC guidelines,5 women with controlled or uncontrolled hypertension should not be offered combined oral contraceptives, the patch, or the ring (category 3—theoretical or proven risks outweigh the benefits, and category 4 for systolic blood pressure greater than 160 mm Hg or diastolic blood pressure greater than 100 mm Hg).
The progesterone-only pill (“mini pill”), medroxyprogesterone acetate (intramuscular or subcutaneous), Mirena IUS, the copper intrauterine device, and the etonogestrel implant are all safer options.
A small subset of patients develop elevated blood pressure after starting hormonal contraceptives. Estrogen-containing hormones can increase the liver’s output of angiotensinogen, which is a renin substrate that activates the renin-angiotensin-aldosterone system. If this becomes clinically apparent, these patients should refrain from estrogen-containing products and use progestin-only formulations as a safer alternative.
Patients with isolated elevated hypertriglyceridemia should avoid oral contraceptives. However, the patch, the ring, and progestin-only methods may be acceptable.
Diabetic patients with microvascular complications of retinopathy or nephropathy and any patient with macrovascular disease (stroke, cardiovascular disease) should not be offered estrogen-containing contraception.
Contraceptive counseling is both an art and a science. The role of the health care provider is to determine the patient’s medical eligibility and match her preferences and lifestyle to an appropriate method for both contraceptive and potentially noncontraceptive benefits, while minimizing the risk of unintended pregnancy.
Women throughout the range of reproductive years need appropriate counseling and education on hormones, the menstrual cycle, and the efficacy of contraception as part of their routine gynecologic evaluation. Issues of access to birth control, cost, possible side effects, and actual effectiveness of methods are important to discuss.
In this paper we will discuss common clinical practice case scenarios to illustrate contraceptive counseling and management, including:
- Perimenopausal women
- Women with thrombophilia
- Women who contemplate becoming pregnant in the future
- Women with psychiatric illness
- Women with hypertension.
HALF OF ALL PREGNANCIES ARE UNPLANNED
Although many contraceptive options are available, 48% of all pregnancies in the United States are unintended.1 In 2009, the national teen birth rate was 39.1 births per 1,000 girls and women age 15 to 19 years, which was 37% lower than in 1991.2 Still, African American and Hispanic teenagers living in southern states have disproportionately higher rates.
The rate of unintended pregnancy is a little lower at the older end of the reproductive age range, but still high: 35% of all pregnancies in women over 40 years old are also unintended.2
To find out why these numbers are so high, in 2007 the US Centers for Disease Control and Prevention (CDC) conducted a survey3 that included 8,000 women reporting unintended pregnancy who had not used contraception. Of these, 39% were married. Surprisingly, more than one-third of women said they did not know they could get pregnant when they did.3
WHAT’S NEW IN CONTRACEPTION?
The “pill” was approved by the US Food and Drug Administration (FDA) more than 50 years ago, and it is still the most commonly used contraceptive method (followed by surgical sterilization). Enovid, the pill formulated by Dr. John Rock and Dr. Gregory Pincus in the 1950s, contained 150 μg of mestranol (equivalent to 90 μg of ethinyl estradiol) and 9.85 mg of norethynodrel, a very potent progestin. Our current oral contraceptive pills contain much lower hormone doses and have fewer androgenic side effects.4
In May 2010, the CDC and the World Health Organization (WHO) updated their safety guidelines for all hormonal contraceptives and the use of these agents in patients with various medical and family histories. They ranked contraceptive methods from those with no restriction to those with unacceptable risk to their use. This document can be accessed at www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm.5
New developments in oral contraceptives are notably in the 19-nortestosterone derivatives, the family that includes the second-generation progestogens already available such as norgestimate (contained in Ortho-Cyclen) and norethindrone (contained in Loestrin). A newer progestin, dienogest, is available in a preparation that also contains estradiol valerate (Natazia). Drospirenone, which is similar to spironolactone, is contained in Yaz, Yasmin, and newer products that also contain levomefolate calcium (Beyaz, Safyral).
LoLoestrin Fe, which contains active pills containing 10 μg of ethinyl estradiol and 1 mg of norethindrone and placebo pills with 75 mg of ferrous fumarate, was recently approved by the FDA and offers an ultra-low dose of estrogen.
Depot medroxyprogesterone acetate now comes in a 104-mg suspension for subcutaneous injection every 3 months; it is called depo-subQ provera 104. Standard medroxyprogesterone acetate 150 mg for intramuscular injection every 3 months (Depo-Provera) is still available and has gone generic. The newer product offers the advantages of lower dose and less weight-gain. Also, it allows capable and willing patients to self-administer their contraceptives. However, it is more expensive—$ 104 per injection for a patient without insurance at Cleveland Clinic, compared with $46 for Depo-Provera and $10 for the generic intramuscular preparation for a patient with insurance.
A new option for emergency contraception, ulipristal (ella) is a progesterone antagonist-agonist available only by prescription. Taken in a single oral dose of 30 μg, it is effective for up to 120 hours after unprotected intercourse. It joins Plan B (levonorgestrel 1.5 mg in a single dose) and Next Choice (two doses of levonorgestrel 0.75 mg each), which are available over-the-counter for women age 17 years or older, and by prescription for those 16 years and younger, for use up to 72 hours after unprotected intercourse.
CASE 1: CONTRACEPTION IN PERIMENOPAUSE
A 48-year-old attorney who has had two children complains of irregular menstrual cycles and of occasional hot flashes at night that wake her from sleep. She keeps a menstrual calendar; it shows her last menstrual period was 3 months ago. She took oral contraceptives for 15 years before she had her first child. She is using condoms intermittently for contraception. Her body mass index is normal at 24 kg/m2, and she does not smoke. How do you counsel her?
A variety of hormonal options
This healthy perimenopausal woman has a variety of hormonal contraception options that would have the added benefit of regulating her menstrual cycle or suppressing it altogether. These include the levonorgestrel intrauterine system (Mirena IUS), various injectable products (such as Depo-Provera or the newer depo-subQ provera 104), contraceptive pills, the Ortho Evra contraceptive patch, and the vaginal contraceptive ring (NuvaRing). Of these, low-dose birth control pills may be the best option, as they would help with cycle control, offer contraception, and better regulate hormonal fluctuations to reduce her hot flashes.
Hormonal contraception can safely be used in women in their 30s and 40s, and often until menopause if the benefit outweighs the risk.
An estradiol valerate-dienogest oral contraceptive with a quadriphasic dosing schedule (Natazia) has been studied in women up to age 50. Although it was approved in 2010 in the United States, this pill has been used in Europe since the 1990s. The 26 active pills contain tapering doses of the active drugs, with the aim of mimicking the natural menstrual cycle, similar to triphasic pills. Estradiol valerate is a bioidentical estrogen, as it is rapidly metabolized to estradiol (E2), which is identical to 17-beta estradiol and estrone (E3) produced by the ovary. A dose of 2 mg of estradiol valerate is equivalent to 10 μg of ethinyl estradiol, which is the estrogen component in most other oral contraceptives. Low-dose pills by definition contain less than 50 μg of ethinyl estradiol. Dienogest, the progesterone component, has a 17-cyanomethyl group that accounts for its strongly progestogenic and weakly antiandrogenic properties.
All oral hormonal contraceptives can increase triglycerides by inducing the CYP450 system in the liver. However, in clinical trials, estradiol valerate-dienogest also caused other changes in lipid metabolism, such as a nonsignificant increase in high-density lipoprotein cholesterol and a slight reduction in low-density lipoprotein cholesterol and lipoprotein(a) compared with ethinyl estradiol-levonorgestrel preparations.6
It is important to advise patients that, compared with users of other oral contraceptives, estradiol valerate-dienogest users may experience fewer days of menstrual bleeding and more cycles without withdrawal bleeding. This product can therefore be an effective alternative for women with menorrhagia.
All classes of hormonal contraception carry a similar risk of side effects, such as headache, breast tenderness, nausea, irregular bleeding, and mood changes. Some women have no side effects.
CASE 2: THROMBOPHILIA
A 39-year-old woman with a body mass index of 31 kg/m2 (obese) has a history of protein S deficiency with active lower-extremity deep vein thrombosis, for which she is taking warfarin (Coumadin). She experiences menorrhagia and dysmenorrhea due to intramural fibroids and possible adenomyosis seen on transvaginal ultrasonography and confirmed by magnetic resonance imaging. Hysteroscopy reveals no polyps or submucosal fibroids. An endometrial biopsy is negative for malignancy.
She desires contraception. How do you counsel her?
Estrogens are contraindicated—except, perhaps, in select cases
This patient has many reasons for heavy bleeding. She is on warfarin, which effectively inhibits synthesis of vitamin K-dependent coagulation factor. She also has fibroids and adenomyosis. The latter is a difficult condition to control, as the location of the intramuscular glands makes treatments such as ablation, dilation and curettage, and oral agents ineffective.
All estrogen-containing formulations (pills, ring, patch) are contraindicated in women with acute venous thromboembolism (VTE) and known thrombophilia. A newer agent approved for treating menorrhagia (not for contraception), tranexamic acid (Lysteda), also carries a contraindication for patients with thrombophilia or history of VTE; however, the evidence for the latter is controversial.7
The updated CDC guidelines for the use of hormonal contraceptives state that patients who receive anticoagulation for at least 3 months and who have no history of VTE or a low risk of recurrent VTE (no evidence of active cancer, no known thrombophilia) may use estrogen-containing contraceptives in select cases (category 3—theoretical risk outweighs benefits, but not an absolute contraindication).5 Although this is not common clinical practice, select patients may benefit from menstrual cycle control while receiving anticoagulation. However, other contraceptive alternatives are preferred if possible.
Progestin-only treatments such as the Mirena IUS (if the fibroids do not distort the uterine cavity) and the etonogestrel implant (Implanon) are nonsurgical options that may reduce menorrhagia and are safer alternatives for patients with thrombophilia.
The Paragard (copper) intrauterine device would provide nonhormonal contraception without diminishing menorrhagia. Obviously, barrier methods (which are less effective than hormonal contraception) can be suggested for contraception alone. A viable option for women finished with childbearing is hysterectomy, which provides contraceptive benefit and definitive treatment of menorrhagia due to adenomyosis.
Laboratory screening for VTE is not required before starting estrogen-containing contraceptives. However, one should take a detailed history and inquire about VTE events or a family history of recurrent VTE.
VTE rates among reproductive-age women are 4 to 5 per 10,000 women per year.8 The rate of VTE in oral contraceptive users is estimated as 9 to 10 per 10,000 women per year.9 However, rates of VTE associated with pregnancy and postpartum states are exponentially greater. Although recent studies have shown some discrepancy in rates of VTE across different classes of progestins,10,11 the absolute risk of VTE with hormonal contraceptives is very low.
In December 2011, an FDA panel voted 15 to 11 that the benefits of drospirenone-containing contraceptives (eg, Yaz, Yasmin, Beyaz, Safyral), such as preventing pregnancy, outweigh the potential risk. However, product labeling may change in the future to more accurately reflect the risk-benefit ratio. Stay tuned for better-designed trials to further assess VTE risk across progestins.
Health care providers should engage patients in an informed discussion about all risks and benefits of hormonal contraceptives and note this risk of VTE is higher in gravid women.
CASE 3: FUTURE FERTILITY
A 30-year-old surgical resident who has never been pregnant comes for her annual examination. She currently desires birth control but would like to be pregnant 1 to 2 years from now. She has no history of significant medical illness. Her body mass index is 23 kg/m2, and she takes no medications. How do you counsel her?
Many options; also consider folic acid
Effective counseling leads to patient-centered decision-making for all treatments and procedures. Contraceptive counseling should elicit the patient’s perspective about hormonal methods and educate her on efficacy, proper use, and common adverse effects.
Contraception should fit the patient’s lifestyle. Questions as simple as “Are you a good pill-taker?” or “Are you comfortable with injections?” will help you and the patient assess what will work effectively and will maintain good adherence.
Deciding on a contraceptive option that is cost-effective is crucial, particularly for many young women or adolescents. Many oral contraceptives are widely available as generic formulations for less than $10 per month. Although generic drugs are not required to be 100% bioequivalent to their brand-name counterparts, they can provide a more economical option. For a complete guide to different hormonal contraceptive formulations, we suggest Choosing a birth control method, available on the Web site of the Association of Reproductive Health Professionals at www.arhp.org/upload-Docs/choosingqrg.pdf.12
As discussed earlier, half of all pregnancies are unplanned, and so women of childbearing age should be ingesting 400 μg of folic acid daily. Debate exists as to whether Americans who eat a balanced diet need a multivitamin.13 However, there is no debate about folic acid, which is proven to prevent neural tube defects. Newer formulations of ethinyl estradiol-drospirenone (Beyaz, Safyral) now contain an active form of folic acid (levomefolate calcium 451 mg in each pill). For the above patient who needs contraception and is willing to take birth control, the addition of folic acid provides an essential element in preconception counseling.
Regardless of the current contraceptive choice, patients who actively desire pregnancy should take a prenatal vitamin that contains folic acid and iron.
In addition to combined oral contraceptives, other options for this patient include medroxyprogesterone acetate (intramuscular or subcutaneous), NuvaRing, or intrauterine devices. The Ortho Evra patch is also an option for this patient. However, since 2008 the patch has carried an FDA warning that the risk of VTE is twice as high with this product than with oral contraceptives that contain 30 μg of ethinyl estradiol plus levonorgestrel.14 Postmarketing data did not show any higher risk of VTE in patch users compared with oral contraceptive users less than 40 years of age, however.15
CASE 4: PSYCHIATRIC ILLNESS
A 21-year-old woman who has bipolar II disorder comes to your office for her annual gynecologic evaluation. She has one sexual partner and desires oral contraceptive pills. Lithium treatment has failed for her, but her condition is stable on carbamazepine (Tegretol). She asks if it is true that women can still get pregnant while on the birth control pill. How do you counsel her?
Possible interactions with psychiatric drugs
Like the woman in case 3, this patient has many options, including estrogen-containing pills, the vaginal ring, the patch, injectable contraceptives, and intrauterine devices.
Certain antiepileptic, antipsychotic, or headache medications such as carbamezapine, phenytoin (Dilantin), oxcarbazepine (Trileptal), and topiramate (Topamax) decrease levels of hormonal contraceptives by induction of the CYP450 enzymes. Conversely, it is suggested that lamotrigine (Lamictal) levels decrease by up to 49% while patients concomitantly take oral contraceptive pills, which can induce seizure activity.16 Also, antibiotics such as rifampin (Rifadin) and even herbs such as St. John’s Wort can decrease the effectiveness of hormonal contraceptives by increasing their metabolism.
On the positive side, depot medroxyprogesterone acetate raises the seizure threshold by a mechanism attributed to high levels of progestins and is a better option for epileptic patients. A bulletin of the American College of Gynecologists addresses the paucity of data on hormonal treatments in depressed patients. However, some evidence points to slight improvement of depressive symptoms after 1 year in patients who took Depo-Provera compared with those who discontinued the drug.17
The Pearl index, a measure of contraceptive efficacy
We refer to the Pearl index when answering our patients’ questions about contraceptive efficacy. The Pearl index is defined as the number of unintended pregnancies per 100 women per year. The typical (or actual) effectiveness for each contraceptive method is quoted rather than the theoretical (perfect-use) efficacy.
We suggest simplifying this discussion with patients. For example, for every 100 women using male condoms for contraception, 15 women have unintended pregnancies per year. With hormonal contraceptives (pill, patch, or ring), for every 100 women there are 8 per year with unintended pregnancy, 3 of 100 with Depo-Provera, and less than 1 in 100 using intrauterine devices or female or male sterilization.18
Efficacy decreases (and the failure rate increases) with frequency of intercourse, irregular menstrual cycles, missed pills, improper dosing, and drug-drug interactions as described above.
CASE 5: HYPERTENSION
A 33-year-old woman who has been pregnant twice experienced preeclampsia in her last pregnancy, and now her blood pressure is consistently approximately 140/90 mm Hg on multiple office visits and ambulatory monitoring. She desires contraception. How do you counsel her?
Avoid estrogen-containing products
According to the WHO and CDC guidelines,5 women with controlled or uncontrolled hypertension should not be offered combined oral contraceptives, the patch, or the ring (category 3—theoretical or proven risks outweigh the benefits, and category 4 for systolic blood pressure greater than 160 mm Hg or diastolic blood pressure greater than 100 mm Hg).
The progesterone-only pill (“mini pill”), medroxyprogesterone acetate (intramuscular or subcutaneous), Mirena IUS, the copper intrauterine device, and the etonogestrel implant are all safer options.
A small subset of patients develop elevated blood pressure after starting hormonal contraceptives. Estrogen-containing hormones can increase the liver’s output of angiotensinogen, which is a renin substrate that activates the renin-angiotensin-aldosterone system. If this becomes clinically apparent, these patients should refrain from estrogen-containing products and use progestin-only formulations as a safer alternative.
Patients with isolated elevated hypertriglyceridemia should avoid oral contraceptives. However, the patch, the ring, and progestin-only methods may be acceptable.
Diabetic patients with microvascular complications of retinopathy or nephropathy and any patient with macrovascular disease (stroke, cardiovascular disease) should not be offered estrogen-containing contraception.
- Finer LB, Henshaw SK. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspect Sex Reprod Health 2006; 38:90–96.
- Centers for Disease Control and Prevention. Vital signs: teenage pregnancy—United States 1991–2009. MMWR 2011; 60:414–420. www.cdc.gov/mmwr/preview/mmwrhtml/mm6013a5.htm. Accessed January 10, 2012.
- Nettleman MD, Chung H, Brewer J, Ayoola A, Reed PL. Reasons for unprotected intercourse: analysis of the PRAMS survey. Contraception 2007; 75:361–366.
- Nelson A. New low-dose, extended-cycle pills with levonorgestrel and ethinyl estradiol: an evolutionary step on birth control. Int J Womens Health 2010; 2:99–106.
- US Centers for Disease Control and Prevention. Appendix L. Summary of classifications for hormonal contraceptive methods and intrauterine devices. MMWR 2010; 59( RR04):76–81. www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm. Accessed January 10, 2012.
- Wiegratz L, Lee JH, Kutchera E, et al. Effect of dienogest-containing oral contraceptives in lipid metabolism. Contraception 2002; 65:223–229.
- Sundström A, Seaman H, Kieler H, Alfredsson L. The risk of venous thromboembolism associated with the use of tranexamic acid and other drugs used to treat menorrhagia: a case-control study using the General Practice Research Database. Br J Obstet Gynecol 2009; 116:91–97.
- Heinemann L, Dinger JC. Range of published estimates of venous thromboembolism incidence in young women. Contraception 2007; 75:328–336.
- Dinger JC, Heinemann LA, Kühl-Habich D. The safety of a drospirenone-containing oral contraceptive: final results from the European Active Surveillance Study on oral contraceptives based on 142,475 women-years of observation. Contraception 2007; 75:344–354.
- Parkin L, Sharples K, Hernandez RK, Jick SS. Risk of venous thromboembolism in users of oral contraceptives containing drospirenone or levonorgestrel: nested case-control study based on UK General Practice Research Database. BMJ 2011; 342:d2139.
- Jick SS, Hernandez RK. Risk of non-fatal venous thromboembolism in women using oral contraceptives containing drospirenone compared with women using oral contraceptives containing levonorgestrel: case-control study using United States claims data. BMJ 2011; 342:d2151.
- Association of Reproductive Health Professionals (ARHP). Choosing a birth control method. www.arhp.org/uploadDocs/choosingqrg.pdf. Accessed January 10, 2012.
- Caballero B. Should healthy people take a multivitamin? Cleve Clin J Med 2010; 77:656–657.
- US Food and Drug Administration. Ortho Evra questions and answers (1/18/2008). www.fda.gov/Drugs/DrugSafety/Postmarket-DrugSafetyInformationforPatientsandProviders/ucm110403.htm. Accessed January 10, 2012.
- Jick SS, Hagberg KW, Hernandez RK, Kaye JA. Postmarketing study of ORTHO EVRA and levonorgestrel oral contraceptives containing hormonal contraceptives with 30 mcg of ethinyl estradiol in relation to nonfatal venous thromboembolism. Contraception 2010; 81:16–21.
- Sabers A, Ohman I, Christensen J, Tomson T. Oral contraceptives reduce lamotrigine plasma levels. Neurology 2003; 61:570–571.
- Westhoff C, Truman C, Kalmuss D, et al; Depressive symptoms and Depo-Provera. Contraception 1998; 57:237–240.
- Trusell Wynn LL. Reducing unintended pregnancy in the United States. Contraception 2008; 77:1–5.
- Finer LB, Henshaw SK. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspect Sex Reprod Health 2006; 38:90–96.
- Centers for Disease Control and Prevention. Vital signs: teenage pregnancy—United States 1991–2009. MMWR 2011; 60:414–420. www.cdc.gov/mmwr/preview/mmwrhtml/mm6013a5.htm. Accessed January 10, 2012.
- Nettleman MD, Chung H, Brewer J, Ayoola A, Reed PL. Reasons for unprotected intercourse: analysis of the PRAMS survey. Contraception 2007; 75:361–366.
- Nelson A. New low-dose, extended-cycle pills with levonorgestrel and ethinyl estradiol: an evolutionary step on birth control. Int J Womens Health 2010; 2:99–106.
- US Centers for Disease Control and Prevention. Appendix L. Summary of classifications for hormonal contraceptive methods and intrauterine devices. MMWR 2010; 59( RR04):76–81. www.cdc.gov/mmwr/preview/mmwrhtml/rr5904a13.htm. Accessed January 10, 2012.
- Wiegratz L, Lee JH, Kutchera E, et al. Effect of dienogest-containing oral contraceptives in lipid metabolism. Contraception 2002; 65:223–229.
- Sundström A, Seaman H, Kieler H, Alfredsson L. The risk of venous thromboembolism associated with the use of tranexamic acid and other drugs used to treat menorrhagia: a case-control study using the General Practice Research Database. Br J Obstet Gynecol 2009; 116:91–97.
- Heinemann L, Dinger JC. Range of published estimates of venous thromboembolism incidence in young women. Contraception 2007; 75:328–336.
- Dinger JC, Heinemann LA, Kühl-Habich D. The safety of a drospirenone-containing oral contraceptive: final results from the European Active Surveillance Study on oral contraceptives based on 142,475 women-years of observation. Contraception 2007; 75:344–354.
- Parkin L, Sharples K, Hernandez RK, Jick SS. Risk of venous thromboembolism in users of oral contraceptives containing drospirenone or levonorgestrel: nested case-control study based on UK General Practice Research Database. BMJ 2011; 342:d2139.
- Jick SS, Hernandez RK. Risk of non-fatal venous thromboembolism in women using oral contraceptives containing drospirenone compared with women using oral contraceptives containing levonorgestrel: case-control study using United States claims data. BMJ 2011; 342:d2151.
- Association of Reproductive Health Professionals (ARHP). Choosing a birth control method. www.arhp.org/uploadDocs/choosingqrg.pdf. Accessed January 10, 2012.
- Caballero B. Should healthy people take a multivitamin? Cleve Clin J Med 2010; 77:656–657.
- US Food and Drug Administration. Ortho Evra questions and answers (1/18/2008). www.fda.gov/Drugs/DrugSafety/Postmarket-DrugSafetyInformationforPatientsandProviders/ucm110403.htm. Accessed January 10, 2012.
- Jick SS, Hagberg KW, Hernandez RK, Kaye JA. Postmarketing study of ORTHO EVRA and levonorgestrel oral contraceptives containing hormonal contraceptives with 30 mcg of ethinyl estradiol in relation to nonfatal venous thromboembolism. Contraception 2010; 81:16–21.
- Sabers A, Ohman I, Christensen J, Tomson T. Oral contraceptives reduce lamotrigine plasma levels. Neurology 2003; 61:570–571.
- Westhoff C, Truman C, Kalmuss D, et al; Depressive symptoms and Depo-Provera. Contraception 1998; 57:237–240.
- Trusell Wynn LL. Reducing unintended pregnancy in the United States. Contraception 2008; 77:1–5.
KEY POINTS
- Hormonal contraceptives have a number of noncontraceptive benefits, such as regulating the menstrual cycle.
- The Pearl index is the number of unintended pregnancies per 100 women per year. Rates are 15% using male condoms, 8% with oral contraceptives, 3% with depot medroxyprogesterone acetate (Depo-Provera) injections, and less than 1% with intrauterine devices or female or male sterilization.
- Estrogen-containing products should be avoided in patients with hypertension or who are at risk of venous thromboembolism.