User login
Endoscopic ultrasonography to evaluate pancreatitis
Endoscopic ultrasonography (EUS) is a minimally invasive test that provides high-resolution imaging of the pancreas.1,2 As such, it is proving useful.
Accurate diagnosis and timely intervention are essential in managing acute and chronic pancreatitis, which are often encountered in the clinic and the hospital. However, the cause of acute pancreatitis is not always easy to determine. Furthermore, recurrent bouts can progress to chronic pancreatitis if the cause is not identified and eliminated. EUS has been studied extensively in the evaluation of both acute and chronic pancreatitis, as it can identify obstructive and biliary causes of acute pancreatitis and early structural features of chronic pancreatitis.
This article will review the indications and evidence for EUS in the evaluation of acute and chronic pancreatitis.
SPECIALIZED TRAINING REQUIRED
EUS involves passage of a specialized endoscope through the esophagus and stomach and into the duodenum. The scope has a very small ultrasound probe at the tip, allowing detailed imaging of the upper gastrointestinal tract and surrounding organs.
There are two types of EUS endoscope: radial and linear. A radial scope provides a 360° range of view perpendicular to the long axis of the scope. A linear scope provides a 150° view parallel to the long axis of the scope. Many endosonographers favor linear EUS for imaging the pancreas because it permits fine-needle aspiration biopsy of masses, cysts, and lymph nodes.
Specialized training beyond the gastroenterology fellowship is usually required to become proficient in performing EUS, in recognizing the anatomy it reveals, and in performing fine-needle aspiration biopsy.
ENDOSCOPIC ULTRASONOGRAPHY IN ACUTE PANCREATITIS
Finding the cause of acute pancreatitis can be challenging in patients who do not have typical risk factors, eg, those who do not drink substantial amounts of alcohol and in whom transabdominal ultrasonography fails to reveal gallstones.
Several studies have evaluated the role of EUS in recurrent “idiopathic” pancreatitis.3–5 Causes of acute pancreatitis detectable with EUS included gallbladder and bile duct microlithiasis (stones smaller than 3 mm), cysts, intraductal papillary mucinous neoplasms, ampullary neoplasms, pancreas divisum, and pancreatic masses.
Stones, sludge. Transabdominal ultrasonography is often performed in the workup of acute pancreatitis to rule out gallbladder stones and biliary dilation. Unfortunately, it does a poor job of imaging the distal common bile duct, where culprit stones may reside.
EUS provides a high-quality view of the bile duct from the ampulla of Vater to the region of the hepatic hilum and is safer than endoscopic retrograde cholangiopancreatography (ERCP). The available evidence supports the use of EUS as a diagnostic test for bile duct stones.3–7 In fact, using ERCP as the reference standard, EUS has been found to be more sensitive than transabdominal ultrasonography for bile duct stones.4
The yield of EUS for finding biliary sludge and stones may be high in patients with unexplained pancreatitis. EUS detected sludge, microlithiasis, or both in 33 of 35 patients with idiopathic acute pancreatitis who underwent transabdominal ultrasonography with negative results.8 Furthermore, most were symptom-free at an average of 10 months after cholecystectomy, suggesting that microlithiasis was the cause of the “idiopathic” pancreatitis.
EUS can also decrease the number of unnecessary ERCP procedures in patients with suspected biliary pancreatitis. In these patients, EUS can be performed as an initial diagnostic test to exclude retained biliary stones. If a stone is present, the endoscopist can proceed to ERCP for sphincterotomy and stone removal during the same endoscopic session. If EUS is negative, the endoscopy can be concluded without cannulating the bile duct and putting the patient at risk of acute pancreatitis. In one report, this approach eliminated the need for ERCP in five of six patients with suspected biliary pancreatitis.6
Tumors and other causes of bile duct obstruction can also cause recurrent acute pancreatitis and may be difficult to detect with cross-sectional imaging. EUS, on the other hand, can detect small pancreatic masses (< 2 cm), which may be missed by conventional computed tomography. Also, a linear EUS scope, with its forward oblique view, can image the duodenum and ampulla, where obstructing inflammation, tumors, and polyps may be found. One should strongly suspect occult malignancy in elderly patients with unexplained acute pancreatitis. In those patients, repeat imaging with high-resolution dual-phase computed tomography or with EUS should be considered after a few weeks once the acute inflammation resolves.
Pancreas divisum is a relatively common congenital abnormality in which the dorsal and ventral pancreatic ducts do not properly fuse during embryonic development. To rule out pancreas divisum, the endosonographer must carefully trace the pancreatic duct from the dorsal pancreas into the ventral pancreas, where it connects with the bile duct at the duodenal wall.
In summary, EUS appears to be safe and accurate for diagnosing bile duct stones and other structural causes of idiopathic acute pancreatitis.
ENDOSCOPIC ULTRASONOGRAPHY IN CHRONIC PANCREATITIS
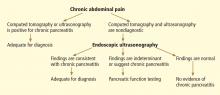
Chronic pancreatitis, a relatively common and sometimes debilitating cause of chronic upper abdominal pain, may be difficult to diagnose using noninvasive imaging tests. Minimal-change chronic pancreatitis is defined as a syndrome of pancreatic abdominal pain with no or slight structural changes detected on imaging but with histologic inflammation and fibrosis diagnostic of chronic pancreatitis.9
A clinical rationale for trying to detect chronic pancreatitis early in its course is that interventions can be started earlier. These include abstinence from alcohol, giving exogenous pancreatic enzymes, and advanced interventions such as celiac plexus blocks for pain control. Some patients may even benefit from resection of the pancreas if pain is severe and resistant to conservative measures.
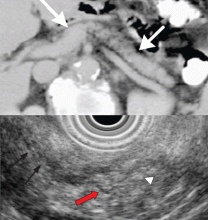
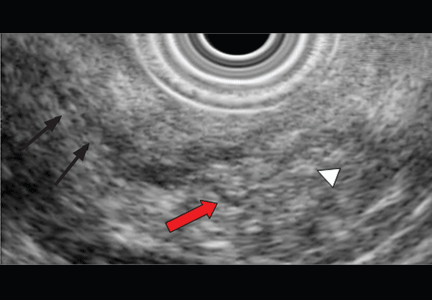
EUS can detect both parenchymal and ductal changes that correlate with histologic fibrosis.10 Parenchymal changes include hyperechoic foci, hyperechoic strands, lobularity, cysts, and shadowing calcifications. Ductal changes include dilation of the main pancreatic duct, irregularity, hyperechoic duct margins, and visible side branches.
Several studies have evaluated the ability of EUS to diagnose early chronic pancreatitis.9,11–15 Reference standards used to determine the accuracy of EUS have included histology,10,16–18 pancreatic function testing,19–22 and ERCP.11,15,23,24
The best diagnostic test may be pancreatic histology. However, biopsy of the pancreas is impractical and exposes patients to high risk. In addition, the patchy and focal distribution of histologic changes may decrease its reliability. Fortunately, the histologic findings of fibrosis have been shown to correlate with EUS criteria in patients undergoing EUS before surgical resection in three recent studies.16–18 A threshold of four or more criteria out of a possible nine was found to provide the optimal sensitivity and specificity for histologic pancreatic fibrosis.16,17 The criteria used were four parenchymal features (hyperechoic foci, strands, hypoechoic lobules, cysts) and five ductal features (irregularity of the main pancreatic duct, dilation, hyperechoic duct walls, visible side branches, and calcifications or stones).
EUS is sensitive for chronic pancreatitis, but ‘true’ accuracy is impossible to know
Unfortunately, greater sensitivity may come at the expense of worse specificity. Certain demographic variables may alter the EUS appearance of the pancreas. A multivariate analysis25 found several variables that predicted abnormalities on EUS even in the absence of clinically evident pancreatitis; the strongest were heavy ethanol use (odds ratio [OR] 5.1, 95% confidence interval [CI] 3.1–8.5), male sex (OR 1.8, 95% CI 1.3–2.55), clinical suspicion of pancreatic disease (OR 1.7, 95% CI 1.2–2.3), and heavy smoking (OR 1.7, 95% CI 1.2–2.4). More prospective studies are needed to further differentiate true disease from false-positive findings of chronic pancreatitis.
Also, traditional EUS scoring symptoms have counted features in an unweighted fashion and assigned an arbitrary cut point (eg, four or more features) for diagnosis. This approach fails to account for the greater importance of some features (eg, calcifications) compared with others.
Interobserver variability is another important limitation of EUS in diagnosing chronic pancreatitis.26,27 In one multicenter study of EUS interpretation, the overall kappa (agreement beyond chance) was only 0.45 for overall chronic pancreatitis diagnosis and worse for many individual criteria for chronic pancreatitis. The endosonographers disagreed most about hyperechoic strands and foci, main pancreatic duct irregularity, and visible side branches (kappa < 0.4).
The Rosemont classification
These limitations led a group of experts to meet in Chicago, IL, to develop a consensus-based and weighted EUS scoring system for the diagnosis of chronic pancreatitis, termed the Rosemont classification.
In this system, the previous parenchymal and ductal features are assigned stricter definitions and reclassified as major and minor criteria. Based on the presence of major and minor features, EUS results are stratified as “normal,” “indeterminate for chronic pancreatitis,” “suggestive of chronic pancreatitis,” or “most consistent with chronic pancreatitis.”15,28
Further validation of this scoring system is needed before it can be used widely.
ENDOSCOPIC ULTRASONOGRAPHY PLUS PANCREATIC FUNCTION TESTING
The best way to diagnose minimal-change chronic pancreatitis may be a combination of sensitive structural and functional testing. Although clinically apparent steatorrhea typically occurs late in the course of chronic pancreatitis, mild exocrine insufficiency may occur early and is detectable with hormone-stimulated pancreatic function testing. Therefore, pancreatic function tests are considered sensitive for diagnosing chronic pancreatitis.20,21,29
Endoscopic pancreatic function testing involves injecting secretin intravenously and then collecting duodenal aspirates through the endoscope. The duodenal fluid is analyzed for bicarbonate concentration as a measure of exocrine function.29
We have studied combined EUS and endoscopic pancreatic function testing in the diagnosis of chronic pancreatitis.16 The combination gives a simultaneous structural and functional assessment of the pancreas and may optimize sensitivity for detecting minimal-change chronic pancreatitis. In a small study, we found the combination had 100% sensitivity for noncalcific chronic pancreatitis compared with a histologic reference standard.16
- Sivak MV, Kaufman A. Endoscopic ultrasonography in the differential diagnosis of pancreatic disease. A preliminary report. Scand J Gastroenterol Suppl 1986; 123:130–134.
- Hisanaga K, Hisanaga A, Nagata K, Ichie Y. High speed rotating scanner for transgastric sonography. AJR Am J Roentgenol 1980; 135:627–629.
- Frossard JL, Sosa-Valencia L, Amouyal G, Marty O, Hadengue A, Amouyal P. Usefulness of endoscopic ultrasonography in patients with “idiopathic” acute pancreatitis. Am J Med 2000; 109:196–200.
- Sugiyama M, Wada N, Atomi Y, Kuroda A, Muto T. Diagnosis of acute pancreatitis: value of endoscopic sonography. AJR Am J Roentgenol 1995; 165:867–872.
- Tandon M, Topazian M. Endoscopic ultrasound in idiopathic acute pancreatitis. Am J Gastroenterol 2001; 96:705–709.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Liu CL, Lo CM, Chan JK, et al. Detection of choledocholithiasis by EUS in acute pancreatitis: a prospective evaluation in 100 consecutive patients. Gastrointest Endosc 2001; 54:325–330.
- Mirbagheri SA, Mohamadnejad M, Nasiri J, Vahid AA, Ghadimi R, Malekzadeh R. Prospective evaluation of endoscopic ultrasonography in the diagnosis of biliary microlithiasis in patients with normal transabdominal ultrasonography. J Gastrointest Surg 2005; 9:961–964.
- Walsh TN, Rode J, Theis BA, Russell RC. Minimal change chronic pancreatitis. Gut 1992; 33:1566–1571.
- Bhutani MJ, Arantes VN, Verma D, et al. Histopathologic correlation of endoscopic ultrasound findings of chronic pancreatitis in human autopsies. Pancreas 2009; 38:820–824.
- Wiersema MJ, Hawes RH, Lehman GA, Kochman ML, Sherman S, Kopecky KK. Prospective evaluation of endoscopic ultrasonography and endoscopic retrograde cholangiopancreatography in patients with chronic abdominal pain of suspected pancreatic origin. Endoscopy 1993; 25:555–564.
- Kahl S, Glasbrenner B, Leodolter A, Pross M, Schulz HU, Malfertheiner P. EUS in the diagnosis of early chronic pancreatitis: a prospective follow-up study. Gastrointest Endosc 2002; 55:507–511.
- Jones SN, Lees WR, Frost RA. Diagnosis and grading of chronic pancreatitis by morphological criteria derived by ultrasound and pancreatography. Clin Radiol 1988; 39:43–48.
- Lees WR. Endoscopic ultrasonography of chronic pancreatitis and pancreatic pseudocysts. Scand J Gastroenterol Suppl 1986; 123:123–129.
- Sahai AV, Zimmerman M, Aabakken L, et al. Prospective assessment of the ability of endoscopic ultrasound to diagnose, exclude, or establish the severity of chronic pancreatitis found by endoscopic retrograde cholangiopancreatography. Gastrointest Endosc 1998; 48:18–25.
- Albashir S, Bronner MP, Parsi MA, Walsh RM, Stevens T. Endoscopic ultrasound, secretin endoscopic pancreatic function test, and histology: correlation in chronic pancreatitis. Am J Gastroenterol 2010; 105:2498–2503.
- Varadarajulu S, Eltoum I, Tamhane A, Eloubeidi MA. Histopathologic correlates of noncalcific chronic pancreatitis by EUS: a prospective tissue characterization study. Gastrointest Endosc 2007; 66:501–509.
- Chong AK, Hawes RH, Hoffman BJ, Adams DB, Lewin DN, Romagnuolo J. Diagnostic performance of EUS for chronic pancreatitis: a comparison with histopathology. Gastrointest Endosc 2007; 65:808–814.
- Chowdhury R, Bhutani MS, Mishra G, Toskes PP, Forsmark CE. Comparative analysis of direct pancreatic function testing versus morphological assessment by endoscopic ultrasonography for the evaluation of chronic unexplained abdominal pain of presumed pancreatic origin. Pancreas 2005; 31:63–68.
- Conwell DL, Zuccaro G, Purich E, et al. Comparison of endoscopic ultrasound chronic pancreatitis criteria to the endoscopic secretinstimulated pancreatic function test. Dig Dis Sci 2007; 52:1206–1210.
- Stevens T, Conwell DL, Zuccaro G, Vargo JJ, Dumot JA, Lopez R. Comparison of endoscopic ultrasound and endoscopic retrograde pancreatography for the prediction of pancreatic exocrine insufficiency. Dig Dis Sci 2008; 53:1146–1151.
- Stevens T, Dumot JA, Parsi MA, Zuccaro G, Vargo JJ. Combined endoscopic ultrasound and secretin endoscopic pancreatic function test in patients evaluated for chronic pancreatitis. Dig Dis Sci 2010; 55:2681–2687.
- Catalano MF, Lahoti S, Geenen JE, Hogan WJ. Prospective evaluation of endoscopic ultrasonography, endoscopic retrograde pancreatography, and secretin test in the diagnosis of chronic pancreatitis. Gastrointest Endosc 1998; 48:11–17.
- Irisawa A, Katakura K, Ohira H, et al. Usefulness of endoscopic ultrasound to diagnose the severity of chronic pancreatitis. J Gastroenterol 2007; 42(suppl 17):90–94.
- Yusoff IF, Sahai AV. A prospective, quantitative assessment of the effect of ethanol and other variables on the endosonographic appearance of the pancreas. Clin Gastroenterol Hepatol 2004; 2:405–409.
- Stevens T, Lopez R, Adler DG, et al. Multicenter comparison of the interobserver agreement of standard EUS scoring and Rosemont classification scoring for diagnosis of chronic pancreatitis. Gastrointest Endosc 2010; 71:519–526.
- Wallace MB, Hawes RH, Durkalski V, et al. The reliability of EUS for the diagnosis of chronic pancreatitis: interobserver agreement among experienced endosonographers. Gastrointest Endosc 2001; 53:294–299.
- Catalano MF, Sahai A, Levy M, et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest Endosc 2009; 69:1251–1261.
- Stevens T, Conwell DL, Zuccaro G, et al. A prospective crossover study comparing secretin-stimulated endoscopic and Dreiling tube pancreatic function testing in patients evaluated for chronic pancreatitis. Gastrointest Endosc 2008; 67:458–466.
Endoscopic ultrasonography (EUS) is a minimally invasive test that provides high-resolution imaging of the pancreas.1,2 As such, it is proving useful.
Accurate diagnosis and timely intervention are essential in managing acute and chronic pancreatitis, which are often encountered in the clinic and the hospital. However, the cause of acute pancreatitis is not always easy to determine. Furthermore, recurrent bouts can progress to chronic pancreatitis if the cause is not identified and eliminated. EUS has been studied extensively in the evaluation of both acute and chronic pancreatitis, as it can identify obstructive and biliary causes of acute pancreatitis and early structural features of chronic pancreatitis.
This article will review the indications and evidence for EUS in the evaluation of acute and chronic pancreatitis.
SPECIALIZED TRAINING REQUIRED
EUS involves passage of a specialized endoscope through the esophagus and stomach and into the duodenum. The scope has a very small ultrasound probe at the tip, allowing detailed imaging of the upper gastrointestinal tract and surrounding organs.
There are two types of EUS endoscope: radial and linear. A radial scope provides a 360° range of view perpendicular to the long axis of the scope. A linear scope provides a 150° view parallel to the long axis of the scope. Many endosonographers favor linear EUS for imaging the pancreas because it permits fine-needle aspiration biopsy of masses, cysts, and lymph nodes.
Specialized training beyond the gastroenterology fellowship is usually required to become proficient in performing EUS, in recognizing the anatomy it reveals, and in performing fine-needle aspiration biopsy.
ENDOSCOPIC ULTRASONOGRAPHY IN ACUTE PANCREATITIS
Finding the cause of acute pancreatitis can be challenging in patients who do not have typical risk factors, eg, those who do not drink substantial amounts of alcohol and in whom transabdominal ultrasonography fails to reveal gallstones.
Several studies have evaluated the role of EUS in recurrent “idiopathic” pancreatitis.3–5 Causes of acute pancreatitis detectable with EUS included gallbladder and bile duct microlithiasis (stones smaller than 3 mm), cysts, intraductal papillary mucinous neoplasms, ampullary neoplasms, pancreas divisum, and pancreatic masses.
Stones, sludge. Transabdominal ultrasonography is often performed in the workup of acute pancreatitis to rule out gallbladder stones and biliary dilation. Unfortunately, it does a poor job of imaging the distal common bile duct, where culprit stones may reside.
EUS provides a high-quality view of the bile duct from the ampulla of Vater to the region of the hepatic hilum and is safer than endoscopic retrograde cholangiopancreatography (ERCP). The available evidence supports the use of EUS as a diagnostic test for bile duct stones.3–7 In fact, using ERCP as the reference standard, EUS has been found to be more sensitive than transabdominal ultrasonography for bile duct stones.4
The yield of EUS for finding biliary sludge and stones may be high in patients with unexplained pancreatitis. EUS detected sludge, microlithiasis, or both in 33 of 35 patients with idiopathic acute pancreatitis who underwent transabdominal ultrasonography with negative results.8 Furthermore, most were symptom-free at an average of 10 months after cholecystectomy, suggesting that microlithiasis was the cause of the “idiopathic” pancreatitis.
EUS can also decrease the number of unnecessary ERCP procedures in patients with suspected biliary pancreatitis. In these patients, EUS can be performed as an initial diagnostic test to exclude retained biliary stones. If a stone is present, the endoscopist can proceed to ERCP for sphincterotomy and stone removal during the same endoscopic session. If EUS is negative, the endoscopy can be concluded without cannulating the bile duct and putting the patient at risk of acute pancreatitis. In one report, this approach eliminated the need for ERCP in five of six patients with suspected biliary pancreatitis.6
Tumors and other causes of bile duct obstruction can also cause recurrent acute pancreatitis and may be difficult to detect with cross-sectional imaging. EUS, on the other hand, can detect small pancreatic masses (< 2 cm), which may be missed by conventional computed tomography. Also, a linear EUS scope, with its forward oblique view, can image the duodenum and ampulla, where obstructing inflammation, tumors, and polyps may be found. One should strongly suspect occult malignancy in elderly patients with unexplained acute pancreatitis. In those patients, repeat imaging with high-resolution dual-phase computed tomography or with EUS should be considered after a few weeks once the acute inflammation resolves.
Pancreas divisum is a relatively common congenital abnormality in which the dorsal and ventral pancreatic ducts do not properly fuse during embryonic development. To rule out pancreas divisum, the endosonographer must carefully trace the pancreatic duct from the dorsal pancreas into the ventral pancreas, where it connects with the bile duct at the duodenal wall.
In summary, EUS appears to be safe and accurate for diagnosing bile duct stones and other structural causes of idiopathic acute pancreatitis.
ENDOSCOPIC ULTRASONOGRAPHY IN CHRONIC PANCREATITIS
Chronic pancreatitis, a relatively common and sometimes debilitating cause of chronic upper abdominal pain, may be difficult to diagnose using noninvasive imaging tests. Minimal-change chronic pancreatitis is defined as a syndrome of pancreatic abdominal pain with no or slight structural changes detected on imaging but with histologic inflammation and fibrosis diagnostic of chronic pancreatitis.9
A clinical rationale for trying to detect chronic pancreatitis early in its course is that interventions can be started earlier. These include abstinence from alcohol, giving exogenous pancreatic enzymes, and advanced interventions such as celiac plexus blocks for pain control. Some patients may even benefit from resection of the pancreas if pain is severe and resistant to conservative measures.
EUS can detect both parenchymal and ductal changes that correlate with histologic fibrosis.10 Parenchymal changes include hyperechoic foci, hyperechoic strands, lobularity, cysts, and shadowing calcifications. Ductal changes include dilation of the main pancreatic duct, irregularity, hyperechoic duct margins, and visible side branches.
Several studies have evaluated the ability of EUS to diagnose early chronic pancreatitis.9,11–15 Reference standards used to determine the accuracy of EUS have included histology,10,16–18 pancreatic function testing,19–22 and ERCP.11,15,23,24
The best diagnostic test may be pancreatic histology. However, biopsy of the pancreas is impractical and exposes patients to high risk. In addition, the patchy and focal distribution of histologic changes may decrease its reliability. Fortunately, the histologic findings of fibrosis have been shown to correlate with EUS criteria in patients undergoing EUS before surgical resection in three recent studies.16–18 A threshold of four or more criteria out of a possible nine was found to provide the optimal sensitivity and specificity for histologic pancreatic fibrosis.16,17 The criteria used were four parenchymal features (hyperechoic foci, strands, hypoechoic lobules, cysts) and five ductal features (irregularity of the main pancreatic duct, dilation, hyperechoic duct walls, visible side branches, and calcifications or stones).
EUS is sensitive for chronic pancreatitis, but ‘true’ accuracy is impossible to know
Unfortunately, greater sensitivity may come at the expense of worse specificity. Certain demographic variables may alter the EUS appearance of the pancreas. A multivariate analysis25 found several variables that predicted abnormalities on EUS even in the absence of clinically evident pancreatitis; the strongest were heavy ethanol use (odds ratio [OR] 5.1, 95% confidence interval [CI] 3.1–8.5), male sex (OR 1.8, 95% CI 1.3–2.55), clinical suspicion of pancreatic disease (OR 1.7, 95% CI 1.2–2.3), and heavy smoking (OR 1.7, 95% CI 1.2–2.4). More prospective studies are needed to further differentiate true disease from false-positive findings of chronic pancreatitis.
Also, traditional EUS scoring symptoms have counted features in an unweighted fashion and assigned an arbitrary cut point (eg, four or more features) for diagnosis. This approach fails to account for the greater importance of some features (eg, calcifications) compared with others.
Interobserver variability is another important limitation of EUS in diagnosing chronic pancreatitis.26,27 In one multicenter study of EUS interpretation, the overall kappa (agreement beyond chance) was only 0.45 for overall chronic pancreatitis diagnosis and worse for many individual criteria for chronic pancreatitis. The endosonographers disagreed most about hyperechoic strands and foci, main pancreatic duct irregularity, and visible side branches (kappa < 0.4).
The Rosemont classification
These limitations led a group of experts to meet in Chicago, IL, to develop a consensus-based and weighted EUS scoring system for the diagnosis of chronic pancreatitis, termed the Rosemont classification.
In this system, the previous parenchymal and ductal features are assigned stricter definitions and reclassified as major and minor criteria. Based on the presence of major and minor features, EUS results are stratified as “normal,” “indeterminate for chronic pancreatitis,” “suggestive of chronic pancreatitis,” or “most consistent with chronic pancreatitis.”15,28
Further validation of this scoring system is needed before it can be used widely.
ENDOSCOPIC ULTRASONOGRAPHY PLUS PANCREATIC FUNCTION TESTING
The best way to diagnose minimal-change chronic pancreatitis may be a combination of sensitive structural and functional testing. Although clinically apparent steatorrhea typically occurs late in the course of chronic pancreatitis, mild exocrine insufficiency may occur early and is detectable with hormone-stimulated pancreatic function testing. Therefore, pancreatic function tests are considered sensitive for diagnosing chronic pancreatitis.20,21,29
Endoscopic pancreatic function testing involves injecting secretin intravenously and then collecting duodenal aspirates through the endoscope. The duodenal fluid is analyzed for bicarbonate concentration as a measure of exocrine function.29
We have studied combined EUS and endoscopic pancreatic function testing in the diagnosis of chronic pancreatitis.16 The combination gives a simultaneous structural and functional assessment of the pancreas and may optimize sensitivity for detecting minimal-change chronic pancreatitis. In a small study, we found the combination had 100% sensitivity for noncalcific chronic pancreatitis compared with a histologic reference standard.16
Endoscopic ultrasonography (EUS) is a minimally invasive test that provides high-resolution imaging of the pancreas.1,2 As such, it is proving useful.
Accurate diagnosis and timely intervention are essential in managing acute and chronic pancreatitis, which are often encountered in the clinic and the hospital. However, the cause of acute pancreatitis is not always easy to determine. Furthermore, recurrent bouts can progress to chronic pancreatitis if the cause is not identified and eliminated. EUS has been studied extensively in the evaluation of both acute and chronic pancreatitis, as it can identify obstructive and biliary causes of acute pancreatitis and early structural features of chronic pancreatitis.
This article will review the indications and evidence for EUS in the evaluation of acute and chronic pancreatitis.
SPECIALIZED TRAINING REQUIRED
EUS involves passage of a specialized endoscope through the esophagus and stomach and into the duodenum. The scope has a very small ultrasound probe at the tip, allowing detailed imaging of the upper gastrointestinal tract and surrounding organs.
There are two types of EUS endoscope: radial and linear. A radial scope provides a 360° range of view perpendicular to the long axis of the scope. A linear scope provides a 150° view parallel to the long axis of the scope. Many endosonographers favor linear EUS for imaging the pancreas because it permits fine-needle aspiration biopsy of masses, cysts, and lymph nodes.
Specialized training beyond the gastroenterology fellowship is usually required to become proficient in performing EUS, in recognizing the anatomy it reveals, and in performing fine-needle aspiration biopsy.
ENDOSCOPIC ULTRASONOGRAPHY IN ACUTE PANCREATITIS
Finding the cause of acute pancreatitis can be challenging in patients who do not have typical risk factors, eg, those who do not drink substantial amounts of alcohol and in whom transabdominal ultrasonography fails to reveal gallstones.
Several studies have evaluated the role of EUS in recurrent “idiopathic” pancreatitis.3–5 Causes of acute pancreatitis detectable with EUS included gallbladder and bile duct microlithiasis (stones smaller than 3 mm), cysts, intraductal papillary mucinous neoplasms, ampullary neoplasms, pancreas divisum, and pancreatic masses.
Stones, sludge. Transabdominal ultrasonography is often performed in the workup of acute pancreatitis to rule out gallbladder stones and biliary dilation. Unfortunately, it does a poor job of imaging the distal common bile duct, where culprit stones may reside.
EUS provides a high-quality view of the bile duct from the ampulla of Vater to the region of the hepatic hilum and is safer than endoscopic retrograde cholangiopancreatography (ERCP). The available evidence supports the use of EUS as a diagnostic test for bile duct stones.3–7 In fact, using ERCP as the reference standard, EUS has been found to be more sensitive than transabdominal ultrasonography for bile duct stones.4
The yield of EUS for finding biliary sludge and stones may be high in patients with unexplained pancreatitis. EUS detected sludge, microlithiasis, or both in 33 of 35 patients with idiopathic acute pancreatitis who underwent transabdominal ultrasonography with negative results.8 Furthermore, most were symptom-free at an average of 10 months after cholecystectomy, suggesting that microlithiasis was the cause of the “idiopathic” pancreatitis.
EUS can also decrease the number of unnecessary ERCP procedures in patients with suspected biliary pancreatitis. In these patients, EUS can be performed as an initial diagnostic test to exclude retained biliary stones. If a stone is present, the endoscopist can proceed to ERCP for sphincterotomy and stone removal during the same endoscopic session. If EUS is negative, the endoscopy can be concluded without cannulating the bile duct and putting the patient at risk of acute pancreatitis. In one report, this approach eliminated the need for ERCP in five of six patients with suspected biliary pancreatitis.6
Tumors and other causes of bile duct obstruction can also cause recurrent acute pancreatitis and may be difficult to detect with cross-sectional imaging. EUS, on the other hand, can detect small pancreatic masses (< 2 cm), which may be missed by conventional computed tomography. Also, a linear EUS scope, with its forward oblique view, can image the duodenum and ampulla, where obstructing inflammation, tumors, and polyps may be found. One should strongly suspect occult malignancy in elderly patients with unexplained acute pancreatitis. In those patients, repeat imaging with high-resolution dual-phase computed tomography or with EUS should be considered after a few weeks once the acute inflammation resolves.
Pancreas divisum is a relatively common congenital abnormality in which the dorsal and ventral pancreatic ducts do not properly fuse during embryonic development. To rule out pancreas divisum, the endosonographer must carefully trace the pancreatic duct from the dorsal pancreas into the ventral pancreas, where it connects with the bile duct at the duodenal wall.
In summary, EUS appears to be safe and accurate for diagnosing bile duct stones and other structural causes of idiopathic acute pancreatitis.
ENDOSCOPIC ULTRASONOGRAPHY IN CHRONIC PANCREATITIS
Chronic pancreatitis, a relatively common and sometimes debilitating cause of chronic upper abdominal pain, may be difficult to diagnose using noninvasive imaging tests. Minimal-change chronic pancreatitis is defined as a syndrome of pancreatic abdominal pain with no or slight structural changes detected on imaging but with histologic inflammation and fibrosis diagnostic of chronic pancreatitis.9
A clinical rationale for trying to detect chronic pancreatitis early in its course is that interventions can be started earlier. These include abstinence from alcohol, giving exogenous pancreatic enzymes, and advanced interventions such as celiac plexus blocks for pain control. Some patients may even benefit from resection of the pancreas if pain is severe and resistant to conservative measures.
EUS can detect both parenchymal and ductal changes that correlate with histologic fibrosis.10 Parenchymal changes include hyperechoic foci, hyperechoic strands, lobularity, cysts, and shadowing calcifications. Ductal changes include dilation of the main pancreatic duct, irregularity, hyperechoic duct margins, and visible side branches.
Several studies have evaluated the ability of EUS to diagnose early chronic pancreatitis.9,11–15 Reference standards used to determine the accuracy of EUS have included histology,10,16–18 pancreatic function testing,19–22 and ERCP.11,15,23,24
The best diagnostic test may be pancreatic histology. However, biopsy of the pancreas is impractical and exposes patients to high risk. In addition, the patchy and focal distribution of histologic changes may decrease its reliability. Fortunately, the histologic findings of fibrosis have been shown to correlate with EUS criteria in patients undergoing EUS before surgical resection in three recent studies.16–18 A threshold of four or more criteria out of a possible nine was found to provide the optimal sensitivity and specificity for histologic pancreatic fibrosis.16,17 The criteria used were four parenchymal features (hyperechoic foci, strands, hypoechoic lobules, cysts) and five ductal features (irregularity of the main pancreatic duct, dilation, hyperechoic duct walls, visible side branches, and calcifications or stones).
EUS is sensitive for chronic pancreatitis, but ‘true’ accuracy is impossible to know
Unfortunately, greater sensitivity may come at the expense of worse specificity. Certain demographic variables may alter the EUS appearance of the pancreas. A multivariate analysis25 found several variables that predicted abnormalities on EUS even in the absence of clinically evident pancreatitis; the strongest were heavy ethanol use (odds ratio [OR] 5.1, 95% confidence interval [CI] 3.1–8.5), male sex (OR 1.8, 95% CI 1.3–2.55), clinical suspicion of pancreatic disease (OR 1.7, 95% CI 1.2–2.3), and heavy smoking (OR 1.7, 95% CI 1.2–2.4). More prospective studies are needed to further differentiate true disease from false-positive findings of chronic pancreatitis.
Also, traditional EUS scoring symptoms have counted features in an unweighted fashion and assigned an arbitrary cut point (eg, four or more features) for diagnosis. This approach fails to account for the greater importance of some features (eg, calcifications) compared with others.
Interobserver variability is another important limitation of EUS in diagnosing chronic pancreatitis.26,27 In one multicenter study of EUS interpretation, the overall kappa (agreement beyond chance) was only 0.45 for overall chronic pancreatitis diagnosis and worse for many individual criteria for chronic pancreatitis. The endosonographers disagreed most about hyperechoic strands and foci, main pancreatic duct irregularity, and visible side branches (kappa < 0.4).
The Rosemont classification
These limitations led a group of experts to meet in Chicago, IL, to develop a consensus-based and weighted EUS scoring system for the diagnosis of chronic pancreatitis, termed the Rosemont classification.
In this system, the previous parenchymal and ductal features are assigned stricter definitions and reclassified as major and minor criteria. Based on the presence of major and minor features, EUS results are stratified as “normal,” “indeterminate for chronic pancreatitis,” “suggestive of chronic pancreatitis,” or “most consistent with chronic pancreatitis.”15,28
Further validation of this scoring system is needed before it can be used widely.
ENDOSCOPIC ULTRASONOGRAPHY PLUS PANCREATIC FUNCTION TESTING
The best way to diagnose minimal-change chronic pancreatitis may be a combination of sensitive structural and functional testing. Although clinically apparent steatorrhea typically occurs late in the course of chronic pancreatitis, mild exocrine insufficiency may occur early and is detectable with hormone-stimulated pancreatic function testing. Therefore, pancreatic function tests are considered sensitive for diagnosing chronic pancreatitis.20,21,29
Endoscopic pancreatic function testing involves injecting secretin intravenously and then collecting duodenal aspirates through the endoscope. The duodenal fluid is analyzed for bicarbonate concentration as a measure of exocrine function.29
We have studied combined EUS and endoscopic pancreatic function testing in the diagnosis of chronic pancreatitis.16 The combination gives a simultaneous structural and functional assessment of the pancreas and may optimize sensitivity for detecting minimal-change chronic pancreatitis. In a small study, we found the combination had 100% sensitivity for noncalcific chronic pancreatitis compared with a histologic reference standard.16
- Sivak MV, Kaufman A. Endoscopic ultrasonography in the differential diagnosis of pancreatic disease. A preliminary report. Scand J Gastroenterol Suppl 1986; 123:130–134.
- Hisanaga K, Hisanaga A, Nagata K, Ichie Y. High speed rotating scanner for transgastric sonography. AJR Am J Roentgenol 1980; 135:627–629.
- Frossard JL, Sosa-Valencia L, Amouyal G, Marty O, Hadengue A, Amouyal P. Usefulness of endoscopic ultrasonography in patients with “idiopathic” acute pancreatitis. Am J Med 2000; 109:196–200.
- Sugiyama M, Wada N, Atomi Y, Kuroda A, Muto T. Diagnosis of acute pancreatitis: value of endoscopic sonography. AJR Am J Roentgenol 1995; 165:867–872.
- Tandon M, Topazian M. Endoscopic ultrasound in idiopathic acute pancreatitis. Am J Gastroenterol 2001; 96:705–709.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Liu CL, Lo CM, Chan JK, et al. Detection of choledocholithiasis by EUS in acute pancreatitis: a prospective evaluation in 100 consecutive patients. Gastrointest Endosc 2001; 54:325–330.
- Mirbagheri SA, Mohamadnejad M, Nasiri J, Vahid AA, Ghadimi R, Malekzadeh R. Prospective evaluation of endoscopic ultrasonography in the diagnosis of biliary microlithiasis in patients with normal transabdominal ultrasonography. J Gastrointest Surg 2005; 9:961–964.
- Walsh TN, Rode J, Theis BA, Russell RC. Minimal change chronic pancreatitis. Gut 1992; 33:1566–1571.
- Bhutani MJ, Arantes VN, Verma D, et al. Histopathologic correlation of endoscopic ultrasound findings of chronic pancreatitis in human autopsies. Pancreas 2009; 38:820–824.
- Wiersema MJ, Hawes RH, Lehman GA, Kochman ML, Sherman S, Kopecky KK. Prospective evaluation of endoscopic ultrasonography and endoscopic retrograde cholangiopancreatography in patients with chronic abdominal pain of suspected pancreatic origin. Endoscopy 1993; 25:555–564.
- Kahl S, Glasbrenner B, Leodolter A, Pross M, Schulz HU, Malfertheiner P. EUS in the diagnosis of early chronic pancreatitis: a prospective follow-up study. Gastrointest Endosc 2002; 55:507–511.
- Jones SN, Lees WR, Frost RA. Diagnosis and grading of chronic pancreatitis by morphological criteria derived by ultrasound and pancreatography. Clin Radiol 1988; 39:43–48.
- Lees WR. Endoscopic ultrasonography of chronic pancreatitis and pancreatic pseudocysts. Scand J Gastroenterol Suppl 1986; 123:123–129.
- Sahai AV, Zimmerman M, Aabakken L, et al. Prospective assessment of the ability of endoscopic ultrasound to diagnose, exclude, or establish the severity of chronic pancreatitis found by endoscopic retrograde cholangiopancreatography. Gastrointest Endosc 1998; 48:18–25.
- Albashir S, Bronner MP, Parsi MA, Walsh RM, Stevens T. Endoscopic ultrasound, secretin endoscopic pancreatic function test, and histology: correlation in chronic pancreatitis. Am J Gastroenterol 2010; 105:2498–2503.
- Varadarajulu S, Eltoum I, Tamhane A, Eloubeidi MA. Histopathologic correlates of noncalcific chronic pancreatitis by EUS: a prospective tissue characterization study. Gastrointest Endosc 2007; 66:501–509.
- Chong AK, Hawes RH, Hoffman BJ, Adams DB, Lewin DN, Romagnuolo J. Diagnostic performance of EUS for chronic pancreatitis: a comparison with histopathology. Gastrointest Endosc 2007; 65:808–814.
- Chowdhury R, Bhutani MS, Mishra G, Toskes PP, Forsmark CE. Comparative analysis of direct pancreatic function testing versus morphological assessment by endoscopic ultrasonography for the evaluation of chronic unexplained abdominal pain of presumed pancreatic origin. Pancreas 2005; 31:63–68.
- Conwell DL, Zuccaro G, Purich E, et al. Comparison of endoscopic ultrasound chronic pancreatitis criteria to the endoscopic secretinstimulated pancreatic function test. Dig Dis Sci 2007; 52:1206–1210.
- Stevens T, Conwell DL, Zuccaro G, Vargo JJ, Dumot JA, Lopez R. Comparison of endoscopic ultrasound and endoscopic retrograde pancreatography for the prediction of pancreatic exocrine insufficiency. Dig Dis Sci 2008; 53:1146–1151.
- Stevens T, Dumot JA, Parsi MA, Zuccaro G, Vargo JJ. Combined endoscopic ultrasound and secretin endoscopic pancreatic function test in patients evaluated for chronic pancreatitis. Dig Dis Sci 2010; 55:2681–2687.
- Catalano MF, Lahoti S, Geenen JE, Hogan WJ. Prospective evaluation of endoscopic ultrasonography, endoscopic retrograde pancreatography, and secretin test in the diagnosis of chronic pancreatitis. Gastrointest Endosc 1998; 48:11–17.
- Irisawa A, Katakura K, Ohira H, et al. Usefulness of endoscopic ultrasound to diagnose the severity of chronic pancreatitis. J Gastroenterol 2007; 42(suppl 17):90–94.
- Yusoff IF, Sahai AV. A prospective, quantitative assessment of the effect of ethanol and other variables on the endosonographic appearance of the pancreas. Clin Gastroenterol Hepatol 2004; 2:405–409.
- Stevens T, Lopez R, Adler DG, et al. Multicenter comparison of the interobserver agreement of standard EUS scoring and Rosemont classification scoring for diagnosis of chronic pancreatitis. Gastrointest Endosc 2010; 71:519–526.
- Wallace MB, Hawes RH, Durkalski V, et al. The reliability of EUS for the diagnosis of chronic pancreatitis: interobserver agreement among experienced endosonographers. Gastrointest Endosc 2001; 53:294–299.
- Catalano MF, Sahai A, Levy M, et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest Endosc 2009; 69:1251–1261.
- Stevens T, Conwell DL, Zuccaro G, et al. A prospective crossover study comparing secretin-stimulated endoscopic and Dreiling tube pancreatic function testing in patients evaluated for chronic pancreatitis. Gastrointest Endosc 2008; 67:458–466.
- Sivak MV, Kaufman A. Endoscopic ultrasonography in the differential diagnosis of pancreatic disease. A preliminary report. Scand J Gastroenterol Suppl 1986; 123:130–134.
- Hisanaga K, Hisanaga A, Nagata K, Ichie Y. High speed rotating scanner for transgastric sonography. AJR Am J Roentgenol 1980; 135:627–629.
- Frossard JL, Sosa-Valencia L, Amouyal G, Marty O, Hadengue A, Amouyal P. Usefulness of endoscopic ultrasonography in patients with “idiopathic” acute pancreatitis. Am J Med 2000; 109:196–200.
- Sugiyama M, Wada N, Atomi Y, Kuroda A, Muto T. Diagnosis of acute pancreatitis: value of endoscopic sonography. AJR Am J Roentgenol 1995; 165:867–872.
- Tandon M, Topazian M. Endoscopic ultrasound in idiopathic acute pancreatitis. Am J Gastroenterol 2001; 96:705–709.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Liu CL, Lo CM, Chan JK, et al. Detection of choledocholithiasis by EUS in acute pancreatitis: a prospective evaluation in 100 consecutive patients. Gastrointest Endosc 2001; 54:325–330.
- Mirbagheri SA, Mohamadnejad M, Nasiri J, Vahid AA, Ghadimi R, Malekzadeh R. Prospective evaluation of endoscopic ultrasonography in the diagnosis of biliary microlithiasis in patients with normal transabdominal ultrasonography. J Gastrointest Surg 2005; 9:961–964.
- Walsh TN, Rode J, Theis BA, Russell RC. Minimal change chronic pancreatitis. Gut 1992; 33:1566–1571.
- Bhutani MJ, Arantes VN, Verma D, et al. Histopathologic correlation of endoscopic ultrasound findings of chronic pancreatitis in human autopsies. Pancreas 2009; 38:820–824.
- Wiersema MJ, Hawes RH, Lehman GA, Kochman ML, Sherman S, Kopecky KK. Prospective evaluation of endoscopic ultrasonography and endoscopic retrograde cholangiopancreatography in patients with chronic abdominal pain of suspected pancreatic origin. Endoscopy 1993; 25:555–564.
- Kahl S, Glasbrenner B, Leodolter A, Pross M, Schulz HU, Malfertheiner P. EUS in the diagnosis of early chronic pancreatitis: a prospective follow-up study. Gastrointest Endosc 2002; 55:507–511.
- Jones SN, Lees WR, Frost RA. Diagnosis and grading of chronic pancreatitis by morphological criteria derived by ultrasound and pancreatography. Clin Radiol 1988; 39:43–48.
- Lees WR. Endoscopic ultrasonography of chronic pancreatitis and pancreatic pseudocysts. Scand J Gastroenterol Suppl 1986; 123:123–129.
- Sahai AV, Zimmerman M, Aabakken L, et al. Prospective assessment of the ability of endoscopic ultrasound to diagnose, exclude, or establish the severity of chronic pancreatitis found by endoscopic retrograde cholangiopancreatography. Gastrointest Endosc 1998; 48:18–25.
- Albashir S, Bronner MP, Parsi MA, Walsh RM, Stevens T. Endoscopic ultrasound, secretin endoscopic pancreatic function test, and histology: correlation in chronic pancreatitis. Am J Gastroenterol 2010; 105:2498–2503.
- Varadarajulu S, Eltoum I, Tamhane A, Eloubeidi MA. Histopathologic correlates of noncalcific chronic pancreatitis by EUS: a prospective tissue characterization study. Gastrointest Endosc 2007; 66:501–509.
- Chong AK, Hawes RH, Hoffman BJ, Adams DB, Lewin DN, Romagnuolo J. Diagnostic performance of EUS for chronic pancreatitis: a comparison with histopathology. Gastrointest Endosc 2007; 65:808–814.
- Chowdhury R, Bhutani MS, Mishra G, Toskes PP, Forsmark CE. Comparative analysis of direct pancreatic function testing versus morphological assessment by endoscopic ultrasonography for the evaluation of chronic unexplained abdominal pain of presumed pancreatic origin. Pancreas 2005; 31:63–68.
- Conwell DL, Zuccaro G, Purich E, et al. Comparison of endoscopic ultrasound chronic pancreatitis criteria to the endoscopic secretinstimulated pancreatic function test. Dig Dis Sci 2007; 52:1206–1210.
- Stevens T, Conwell DL, Zuccaro G, Vargo JJ, Dumot JA, Lopez R. Comparison of endoscopic ultrasound and endoscopic retrograde pancreatography for the prediction of pancreatic exocrine insufficiency. Dig Dis Sci 2008; 53:1146–1151.
- Stevens T, Dumot JA, Parsi MA, Zuccaro G, Vargo JJ. Combined endoscopic ultrasound and secretin endoscopic pancreatic function test in patients evaluated for chronic pancreatitis. Dig Dis Sci 2010; 55:2681–2687.
- Catalano MF, Lahoti S, Geenen JE, Hogan WJ. Prospective evaluation of endoscopic ultrasonography, endoscopic retrograde pancreatography, and secretin test in the diagnosis of chronic pancreatitis. Gastrointest Endosc 1998; 48:11–17.
- Irisawa A, Katakura K, Ohira H, et al. Usefulness of endoscopic ultrasound to diagnose the severity of chronic pancreatitis. J Gastroenterol 2007; 42(suppl 17):90–94.
- Yusoff IF, Sahai AV. A prospective, quantitative assessment of the effect of ethanol and other variables on the endosonographic appearance of the pancreas. Clin Gastroenterol Hepatol 2004; 2:405–409.
- Stevens T, Lopez R, Adler DG, et al. Multicenter comparison of the interobserver agreement of standard EUS scoring and Rosemont classification scoring for diagnosis of chronic pancreatitis. Gastrointest Endosc 2010; 71:519–526.
- Wallace MB, Hawes RH, Durkalski V, et al. The reliability of EUS for the diagnosis of chronic pancreatitis: interobserver agreement among experienced endosonographers. Gastrointest Endosc 2001; 53:294–299.
- Catalano MF, Sahai A, Levy M, et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest Endosc 2009; 69:1251–1261.
- Stevens T, Conwell DL, Zuccaro G, et al. A prospective crossover study comparing secretin-stimulated endoscopic and Dreiling tube pancreatic function testing in patients evaluated for chronic pancreatitis. Gastrointest Endosc 2008; 67:458–466.
KEY POINTS
- EUS can identify the cause of acute pancreatitis when other imaging tests (computed tomography, transabdominal ultrasonography) are unrevealing.
- EUS can safely and accurately detect bile duct stones and other causes of recurrent acute pancreatitis. It can also detect mild and severe structural features of chronic pancreatitis.
- An endoscopic pancreatic function test may be a useful adjunct to EUS to detect mild exocrine insufficiency in early chronic pancreatitis.
Asthma in African Americans: What can we do about the higher rates of disease?
The last several decades have seen a dramatic surge in the prevalence of asthma. In 2009, there were an estimated 17.5 million adults and almost 7.1 million children with asthma in the United States,1 up from 9.5 million adults and slightly more than 5 million children in 1995.2
Multiple factors contribute to these disparities, including genetics, socioeconomic factors, cultural factors, health maintenance behaviors, provider-patient communication, air quality, and obesity.
GENETICS: 70% OF DESTINY?
The trend towards personalized medicine has spurred extensive research into the genetics of asthma. Studies in twins and familial aggregation studies suggest genetics plays a significant role, with estimates of the heritability of asthma as high as 70%.4,5 More than 100 candidate genes have been shown to be associated with asthma and atopy, 30 of them in five or more independent studies.6
Researchers face many challenges when investigating the genetics involved in asthma for a particular race. Race is both a biologic and a social construct and, as such, is a poor substitute for genetics. Race constitutes not only genetic differences in individuals, but also the behaviors, beliefs, and experiences that vary among races.
The clinical disease—the phenotype—is the product of the interaction of genes and these differing behaviors and exposures. Genetics can affect how environmental factors found in association with socioeconomic factors relate to asthma morbidity and mortality.
For example, as we will discuss below, African Americans are more likely than whites to be sensitized to cockroach allergen, even after controlling for socioeconomic variables that may be associated with greater exposure.7 High-level exposure to cockroach allergen in sensitized children has been associated with poor asthma outcomes.8 This suggests that a genetic difference may exist between African Americans and whites with respect to the potential to develop cockroach sensitization, and this difference may be of particular importance for those African Americans living in areas with higher levels of cockroach exposure.
Two polymorphisms
Two polymorphisms have garnered attention for their influence on African Americans with asthma:
TheADRB2gene. This gene codes for the beta-2 adrenergic receptor and resides at chromosome 5q13.9 The receptor is found on several types of cells in the lung, including airway smooth muscle and epithelial cells, and is responsible for the salutary effects of inhaled beta-2 agonists such as albuterol (eg, Proventil).
Allelic polymorphisms of this gene are clinically relevant. The substitution of arginine (Arg) for glycine (Gly) at codon 16 of this gene is responsible for differences in response to short-acting beta-2 agonists. The allelic frequency of Arg16 is lower in white Americans (39.3%) than in African Americans (49.2%), and thus African Americans are more likely to be homozygous for Arg16 (ie, to have the Arg/Arg genotype).10
People who are homozygous for Arg16 who use albuterol on a regular basis are at higher risk of untoward asthma outcomes.11 This is important, for several reasons. In general, adherence to inhaled corticosteroids is poor (not only in African Americans),12 and patients who do not take their inhaled corticosteroids as they should may rely on short-acting beta-2 agonists more frequently. Furthermore, African Americans may have a poorer response to the repeated doses of albuterol that are typically given in the emergency department and in the hospital for severe asthma exacerbations.13 Additionally, data suggest that Arg/Arg individuals have more frequent exacerbations independent of beta-agonist use,14 although curiously, patients who are homozygous for Arg16 have a greater benefit from single doses of short-acting beta-2 agonists than those who are Gly16 homozygous.15
TheCD14gene. An interesting relationship between innate immunity and asthma has recently been described. Polymorphisms of CD14, which codes for a receptor for endotoxin, have been uncovered. The single-nucleotide polymorphism variant thymine (T) at position −260 has been found in greater frequency in whites than in African Americans, who are more likely to have the cytosine (C) allele.16 An association between the CC genotype and atopy has been reported,16 although this has not been consistent.17
A possible explanation for these inconsistencies may lie in complex gene-environment interactions. The amount of endotoxin exposure may play a role in phenotypic expression. Individuals with the CC genotype were at lower risk of developing atopy when exposed to high levels of endotoxin; however, when exposed to lower levels of endotoxin, the CC genotype was associated with a higher risk of atopy.18 Nonfarm homes in westernized countries tend to have lower levels of endotoxin than farm homes, even in low-income urban areas.19 This implies that individuals with the CC allele, who are more likely to be African American, would be at greater risk for atopy in the United States. Greater knowledge of these types of gene-environment interactions may lead to improved understanding of the observations that have generated controversy concerning the “hygiene hypothesis.”
The details of how microbial exposure can influence the human immune response to antigen exposure are still being elucidated.20
These examples highlight not only the importance of genetics in the development of asthma, but also the role genes play in variation of treatment response and subsequent risk of morbidity and death. An understanding of these genetic differences among patients is clearly important for moving towards personalized treatment strategies for asthma.
Ancestry-informative markers
A developing strategy to assess the differences in asthma prevalence, severity, and response to treatment between racial groups is the use of ancestry-informative markers (AIMs).
AIMs are single-nucleotide polymorphisms that occur in varying allelic frequencies between ancestral groups, eg, continental Africans or European whites.21 AIMs provide an estimate of an individual’s proportion of ancestry—ie, of how “African” an African American is genetically.
African ancestry, determined using AIMs, was found to be associated with asthma in people living on the Caribbean coast of Colombia.22 However, one study found that AIMs could not predict an individual’s response to inhaled corticosteroids.23
Further research is necessary to find a technique to determine how groups of individuals can be characterized more precisely and managed more appropriately.
SOCIOECONOMIC FACTORS
African Americans living in low-income urban areas have an even greater prevalence of asthma and a greater risk of asthma-related morbidity and death than African Americans overall.3,24,25 Urban areas typically have a high proportion of residents living at or below the poverty level, and minorities often constitute a substantial proportion of the population in these areas. Evidence suggests that both African American race and lower socioeconomic status are independent risk factors for asthma prevalence, morbidity, and death.3,25
To provide better care for African Americans living in low-income urban areas, it is important to understand the factors that may be contributing to the higher morbidity and mortality rates in low-income urban areas.
Inadequate follow-up
Proper and routine follow-up for evaluation of asthma symptoms is essential for appropriate management. The Expert Panel Report 3 (EPR-3) of the National Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma,26 published in 2007, recommends that patients be seen at least every 6 months if they have been experiencing good control. While gaining control, patients should be seen every 2 to 6 weeks.26
Despite these recommendations, numerous studies have suggested that African Americans do not receive adequate follow-up. Children who are poor, are nonwhite or Hispanic, or are underinsured are more likely to lack routine health care27 and, more specifically, routine asthma care.28 Low-income patients are also more likely to receive care in a large hospital-run clinic or neighborhood clinic,27,28 where continuity of care may be less than ideal.29 Even among patients enrolled in Medicaid or Medicare, African American children with a primary care provider have fewer asthma visits compared with white Medicaid-insured children.30
Insufficient follow-up care contributes to greater asthma morbidity, resulting in, for example, more emergency department visits for asthma in African Americans.27,31,32
Suboptimal care
Data also suggest that the quality of care that residents of low-income urban areas receive is often suboptimal. Many people living in low-income urban areas are not provided with the knowledge and tools to treat asthma exacerbations at home.33 African Americans are also less likely to be seen by an asthma specialist31,34 as recommended for those with moderate or severe asthma.26
The EPR-3 guidelines also stress the importance of inhaled corticosteroids as the preferred therapy for all patients with persistent asthma. Even after controlling for the number of primary care visits, insurance status, and disease severity, African Americans are less likely to receive a prescription for inhaled corticosteroids, or they receive the same dosage of inhaled corticosteroids in the face of more severe disease.31,33,35,36
The reasons for these differences in treatment are not fully understood but are likely multiple. Lack of access to an asthma specialist and financial or formulary constraints are some of the potential barriers to optimal asthma care outcomes.
Misdiagnosis in the acute setting may also be a source of less-than-ideal care, as patients seen in emergency departments may be misdiagnosed with viral infection or bronchitis.
African Americans may report different symptoms than whites
Intriguing studies suggest that African Americans report different symptoms while describing asthma exacerbations.
In one study, compared with whites, African Americans were less likely to report nocturnal symptoms, dyspnea, or chest pain during exacerbations.37 In another study, when given a methacholine challenge that induced a significant drop in forced expiratory volume in 1 second (FEV1), African Americans with asthma were more likely to complain of upper airway symptoms as opposed to lower airway symptoms, compared with white patients.38
The symptoms that African Americans describe, such as having a tight throat or voice, are not typically regarded as related to asthma; for this reason, such descriptions may be an obstacle to correct diagnosis, management, and follow-up.
Asthma care providers should be aware of these observations to ensure that their patients are managed appropriately.
Lack of social support
Living in a low-income urban area presents many challenges that can interfere with proper asthma control.
Asthma diagnosis, management, and morbidity are affected by family support.39 Patients with asthma who lack sufficient financial support for treatment, who lack adequate psychological support, and who have more major life stressors are at higher risk of untoward outcomes. Disruption and dysfunction of the family and the supports available have been associated with greater asthma morbidity.40–42 Unfortunately, these types of stressors are all too common in families living in low-income urban areas.43–45
Multiple stressors that can occur more often in low-income urban areas, including exposure to violent crime, have also been linked to greater asthma morbidity.45–47
POOR PHYSICIAN-PATIENT COMMUNICATION
A consistent theme in focus groups of African Americans living in inner-city areas is the perception that health care providers are not effectively communicating and taking the time to listen to their concerns.48,49 Respondents believed they had better insight into their illness than their providers, and for this reason were better able to manage their disease.
The importance of an optimal provider-patient relationship was highlighted by a prospective cohort study in which Medicaid children receiving care at physician’s offices with the highest cultural competency scores were more adherent with their asthma controller medications.50
MEDICATION ADHERENCE RATES ARE DISTURBINGLY LOW
Rates of medication adherence for chronic diseases is disturbingly low, and may be even worse for pulmonary diseases.51 Reported rates of adherence to asthma medications among all patients range from 50% to 60%.52,53 Several studies showed that African Americans have a lower rate of adherence than do whites,53–55 even after adjusting for multiple socioeconomic variables.56
Many explanations have been proposed for this discrepancy, and all likely play a role in particular environments. For example, the incidence of crime in the surrounding area was inversely related to medication adherence after adjusting for socioeconomic factors.57 African Americans may have more concern about side effects associated with inhaled corticosteroid use and may be less likely to understand how these drugs work.52,53 A poor provider-patient relationship has also been cited as a barrier to adherence.55,57 Finally, physicians are more likely to underestimate asthma severity in an African American patient than in a white patient.58
Taking the time to ensure that patients truly understand all aspects of their disease and establishing a health care environment that is culturally appropriate may have a significant impact in patients with asthma.
ENVIRONMENTAL EXPOSURES
Air quality contributes to the greater asthma morbidity observed in urban residents, including African Americans. While poor outdoor air quality has not been clearly linked to a higher incidence of asthma, it has been associated with greater asthma morbidity. Poor air quality may affect individuals of all races, but with respect to ambient pollutants such as particulate matter and diesel exhaust, outdoor air quality is worse in urban environments where greater proportions of people of low socioeconomic status reside.59,60
The most extensively studied components of air pollution are ozone, sulfur dioxide, and particulate matter. These pollutants have been associated with a higher rate of emergency department visits,61,62 worse asthma symptoms,63,64 and higher exhaled nitric oxide levels.65
Tobacco smoke
Despite the substantial success of smoking cessation efforts nationwide, exposure to tobacco smoke continues to be common and is a significant risk factor for poor asthma control. Recent data suggest that African Americans and whites have a similar prevalence of smoking,66 but a study found a very high prevalence in low-income African Americans.67
Active smoking has been associated with worse asthma control and a higher risk of death.68 People with asthma who smoke are less likely to improve in their lung function and symptom scores when treated with short courses of oral glucocorticoids compared with both nonsmokers and former smokers.69
Secondhand smoke hurts too. Many children living in low-income urban areas are exposed to secondhand smoke or environmental tobacco smoke.70,71 Passive exposure in children has been associated with worse asthma outcomes, and data suggest such exposure may be a cause of asthma.68,72–74
Environmental tobacco smoke has also been implicated in gene-environment interactions. Patients who are either homozygous or heterozygous for the Arg allele at codon 16 of the ADRB2 gene (discussed above) had significantly lower FEV1 and forced vital capacity (FVC) values when exposed to passive tobacco smoke. This difference was not seen in people who were not exposed.75
Cockroach allergen
The type and condition of a person’s housing also plays a role in asthma-related morbidity and death. Across several socioeconomic levels, it has been suggested that African Americans have poorer-quality housing compared with whites.76 Some of the conditions found in low-quality houses, such as interruptions in heat, plumbing leaks, and the presence of rodents, have been associated with a higher prevalence of asthma in the household.77
Cockroach allergen exposure and sensitization is a major contributor to asthma morbidity in African Americans, particularly those living in poorer urban areas where cockroach allergen may be the most common indoor allergen.8 Living in older housing in urban areas is associated with higher exposure to cockroach allergen, and with subsequent sensitization.78,79 Exposure to high levels of the major cockroach allergen, Bla g 1, in sensitized individuals has been linked to a greater risk of hospitalization and unscheduled medical visits for asthma. This was not found to be the case for other common indoor allergens, such as dust mite and cat dander.8
However, it is not only exposure to high cockroach allergen levels that puts African Americans at risk. African Americans living in low-income urban areas may also be more likely than whites living in low-income urban areas to become sensitized to cockroach allergen.7,80 This suggests a gene-environment interaction that may be unique to African Americans. Moreover, cockroach sensitization may occur early in life.81,82
While successful cockroach avoidance measures and environmental control may be challenging, such measures have been shown to decrease rates of asthma morbidity.83
OBESITY
Obesity has been linked to an ever-growing list of diseases, one of which is asthma. Obesity is not a unique challenge for African Americans, but recent data from the US Centers for Disease Control and Prevention show that African Americans have a 51% higher prevalence of obesity compared with whites.84
Obesity is a risk factor for greater asthma morbidity and is a significant challenge in the African American community. The rise in obesity rates has paralleled the rise in asthma in recent decades. The higher one’s body mass index, the higher one’s risk of asthma.85 This association appears to be stronger in people without concurrent atopic disease.86 Obesity has also been associated with a poorer response to inhaled corticosteroids and a higher risk of asthma exacerbations.87 Interestingly, significant weight loss has been associated with improvements in both asthma control and lung function.88,89
What is the mechanism?
The underlying pathogenic mechanisms have not been completely elucidated, and they are likely multiple.
Adipokines (cytokines secreted by adipocytes) have been implicated. Two of the most extensively studied adipokines are leptin and adiponectin. Leptin production is increased in obesity, and it has inflammatory effects on both the innate and adaptive immune systems.90 The opposite is true for adiponectin, which may have anti-inflammatory properties and which decreases as the body mass index increases.90 The precise role these molecules may have in lung disease is undergoing further investigation.
Mechanical alterations in lung function may also contribute. Obese people have a lower functional residual capacity and expiratory reserve volume. Breathing with a lower-volume functional residual capacity results in decreased airway diameter and contributes to increased airway resistance.90 The decreased airway diameter may alter the contractile properties of airway smooth muscle and lead to increased airway responsiveness.90 These differences are in addition to the lower mean values of common spirometry indices such as the FEV1 and FVC, found in nonasthmatic African Americans compared with whites.91
Data suggest these differences are primarily due to anthropometric factors, with nutritional and environmental factors playing a less significant role.92 On this basis, the American Thoracic Society recommends applying race-specific reference standards for use with spirometry in order to accurately gauge lung function in African Americans.
APPROPRIATE CARE AND EDUCATION
The cause of greater asthma prevalence and severity among African Americans is multifactorial. It is likely that a number of factors work together, rather than separately, in influencing the development of asthma and its course.
Some risk factors are avoidable, and it is important to identify and ameliorate them. Others are not preventable, but knowledge of them may provide more specific management strategies and may lead to new therapies in the future.
While more work is needed to further unravel the complex risk factors associated with asthma, ensuring that higher-risk patients are provided the appropriate care and the knowledge to help control their disease is a necessary step in improving the disparities in asthma care outcomes.
- Akinbami LJ, Moorman JE, Liu X. Asthma prevalence, health care use, and mortality: United States, 2005–2009. Natl Health Stat Report 2011; 32:1–14.
- National Institutes of Health. National Heart, Lung, and Blood Institute. Data Fact Sheet. Asthma statistics. January 1999. http://www.nhlbi.nih.gov/health/prof/lung/asthma/asthstat.pdf. Accessed February 1, 2012.
- Lang DM, Polansky M, Sherman MS. Hospitalizations for asthma in an urban population: 1995–1999. Ann Allergy Asthma Immunol 2009; 103:128–133.
- Duffy DL, Martin NG, Battistutta D, Hopper JL, Mathews JD. Genetics of asthma and hay fever in Australian twins. Am Rev Respir Dis 1990; 142:1351–1358.
- Koeppen-Schomerus G, Stevenson J, Plomin R. Genes and environment in asthma: a study of 4 year old twins. Arch Dis Child 2001; 85:398–400.
- Meng JF, Rosenwasser LJ. Unraveling the genetic basis of asthma and allergic diseases. Allergy Asthma Immunol Res 2010; 2:215–227.
- Stevenson LA, Gergen PJ, Hoover DR, Rosenstreich D, Mannino DM, Matte TD. Sociodemographic correlates of indoor allergen sensitivity among United States children. J Allergy Clin Immunol 2001; 108:747–752.
- Rosenstreich DL, Eggleston P, Kattan M, et al. The role of cockroach allergy and exposure to cockroach allergen in causing morbidity among inner-city children with asthma. N Engl J Med 1997; 336:1356–1363.
- Kobilka BK, Dixon RA, Frielle T, et al. cDNA for the human beta 2-adrenergic receptor: a protein with multiple membrane-spanning domains and encoded by a gene whose chromosomal location is shared with that of the receptor for platelet-derived growth factor. Proc Natl Acad Sci U S A 1987; 84:46–50.
- Maxwell TJ, Ameyaw MM, Pritchard S, et al. Beta-2 adrenergic receptor genotypes and haplotypes in different ethnic groups. Int J Mol Med 2005; 16:573–580.
- Israel E, Chinchilli VM, Ford JG, et al; National Heart, Lung, and Blood Institute’s Asthma Clinical Research Network. Use of regularly scheduled albuterol treatment in asthma: genotype-stratified, randomised, placebo-controlled cross-over trial. Lancet 2004; 364:1505–1512.
- Wells K, Pladevall M, Peterson EL, et al. Race-ethnic differences in factors associated with inhaled steroid adherence among adults with asthma. Am J Respir Crit Care Med 2008; 178:1194–1201.
- Carroll CL, Stoltz P, Schramm CM, Zucker AR. Beta2-adrenergic receptor polymorphisms affect response to treatment in children with severe asthma exacerbations. Chest 2009; 135:1186–1192.
- Bleecker ER, Nelson HS, Kraft M, et al. Beta2-receptor polymorphisms in patients receiving salmeterol with or without fluticasone propionate. Am J Respir Crit Care Med 2010; 181:676–687.
- Finkelstein Y, Bournissen FG, Hutson JR, Shannon M. Polymorphism of the ADRB2 gene and response to inhaled beta-agonists in children with asthma: a meta-analysis. J Asthma 2009; 46:900–905.
- Baldini M, Lohman IC, Halonen M, Erickson RP, Holt PG, Martinez FD. A polymorphism in the 5’ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am J Respir Cell Mol Biol 1999; 20:976–983.
- Kedda MA, Lose F, Duffy D, Bell E, Thompson PJ, Upham J. The CD14 C-159T polymorphism is not associated with asthma or asthma severity in an Australian adult population. Thorax 2005; 60:211–214.
- Zambelli-Weiner A, Ehrlich E, Stockton ML, et al. Evaluation of the CD14/-260 polymorphism and house dust endotoxin exposure in the Barbados Asthma Genetics Study. J Allergy Clin Immunol 2005; 115:1203–1209.
- Perzanowski MS, Miller RL, Thorne PS, et al. Endotoxin in inner-city homes: associations with wheeze and eczema in early childhood. J Allergy Clin Immunol 2006; 117:1082–1089.
- Williams LK, Oliver J, Peterson EL, et al. Gene-environment interactions between CD14 C-260T and endotoxin exposure on Foxp3+ and Foxp3− CD4+ lymphocyte numbers and total serum IgE levels in early childhood. Ann Allergy Asthma Immunol 2008; 100:128–136.
- Barnes KC. Ancestry, ancestry-informative markers, asthma, and the quest for personalized medicine. J Allergy Clin Immunol 2010; 126:1139–1140.
- Vergara C, Caraballo L, Mercado D, et al. African ancestry is associated with risk of asthma and high total serum IgE in a population from the Caribbean Coast of Colombia. Hum Genet 2009; 125:565–579.
- Gould W, Peterson EL, Karungi G, et al. Factors predicting inhaled corticosteroid responsiveness in African American patients with asthma. J Allergy Clin Immunol 2010; 126:1131–1138.
- Lang DM, Polansky M. Patterns of asthma mortality in Philadelphia from 1969 to 1991. N Engl J Med 1994; 331:1542–1546.
- Schwartz J, Gold D, Dockery DW, Weiss ST, Speizer FE. Predictors of asthma and persistent wheeze in a national sample of children in the United States. Association with social class, perinatal events, and race. Am Rev Respir Dis 1990; 142:555–562.
- National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma. Summary Report 2007. J Allergy Clin Immunol 2007; 120( suppl 5):S94–S138.
- Newacheck PW, Hughes DC, Stoddard JJ. Children’s access to primary care: differences by race, income, and insurance status. Pediatrics 1996; 97:26–32.
- Halfon N, Newacheck PW. Childhood asthma and poverty: differential impacts and utilization of health services. Pediatrics 1993; 91:56–61.
- Burt CW, Knapp DE. Ambulatory care visits for asthma: United States, 1993–94. Adv Data 1996; 277:1.
- Lieu TA, Lozano P, Finkelstein JA, et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics 2002; 109:857–865.
- Zoratti EM, Havstad S, Rodriguez J, Robens-Paradise Y, Lafata JE, McCarthy B. Health service use by African Americans and Caucasians with asthma in a managed care setting. Am J Respir Crit Care Med 1998; 158:371–377.
- Wisnivesky JP, Leventhal H, Halm EA. Predictors of asthma-related health care utilization and quality of life among inner-city patients with asthma. J Allergy Clin Immunol 2005; 116:636–642.
- Halm EA, Wisnivesky JP, Leventhal H. Quality and access to care among a cohort of inner-city adults with asthma: who gets guideline concordant care? Chest 2005; 128:1943–1950.
- Krishnan JA, Diette GB, Skinner EA, Clark BD, Steinwachs D, Wu AW. Race and sex differences in consistency of care with national asthma guidelines in managed care organizations. Arch Intern Med 2001; 161:1660–1668.
- Nelson A. Unequal treatment: confronting racial and ethnic disparities in health care. J Natl Med Assoc 2002; 94:666–668.
- Boudreaux ED, Emond SD, Clark S, Camargo CA; Multicenter Airway Research Collaboration Investigators. Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics 2003; 111:e615–e621.
- Trochtenberg DS, BeLue R, Piphus S, Washington N. Differing reports of asthma symptoms in African Americans and Caucasians. J Asthma 2008; 45:165–170.
- Hardie GE, Janson S, Gold WM, Carrieri-Kohlman V, Boushey HA. Ethnic differences: word descriptors used by African-American and white asthma patients during induced bronchoconstriction. Chest 2000; 117:935–943.
- Clark NM, Levison MJ, Evans D, Wasilewski Y, Feldman CH, Mellins RB. Communication within low income families and the management of asthma. Patient Educ Couns 1990; 15:191–210.
- Rhee H, Belyea MJ, Brasch J. Family support and asthma outcomes in adolescents: barriers to adherence as a mediator. J Adolesc Health 2010; 47:472–478.
- Quinn K, Kaufman JS, Siddiqi A, Yeatts KB. Parent perceptions of neighborhood stressors are associated with general health and child respiratory health among low-income, urban families. J Asthma 2010; 47:281–289.
- Loerbroks A, Apfelbacher CJ, Bosch JA, Stürmer T. Depressive symptoms, social support, and risk of adult asthma in a population-based cohort study. Psychosom Med 2010; 72:309–315.
- Wood DL, Valdez RB, Hayashi T, Shen A. Health of homeless children and housed, poor children. Pediatrics 1990; 86:858–866.
- O’Donnell L, Stueve A, Myint-U A. Parenting and violence toward self, partners, and others among inner-city young adults. Am J Public Health 2009; 99:2255–2260.
- Leventhal T, Brooks-Gunn J. The neighborhoods they live in: the effects of neighborhood residence on child and adolescent outcomes. Psychol Bull 2000; 126:309–337.
- Shankardass K, McConnell R, Jerrett M, Milam J, Richardson J, Berhane K. Parental stress increases the effect of traffic-related air pollution on childhood asthma incidence. Proc Natl Acad Sci U S A 2009; 106:12406–12411.
- Wright RJ, Mitchell H, Visness CM, et al. Community violence and asthma morbidity: the Inner-City Asthma Study. Am J Public Health 2004; 94:625–632.
- George M, Freedman TG, Norfleet AL, Feldman HI, Apter AJ. Qualitative research-enhanced understanding of patients’ beliefs: results of focus groups with low-income, urban, African American adults with asthma. J Allergy Clin Immunol 2003; 111:967–973.
- Laster N, Holsey CN, Shendell DG, Mccarty FA, Celano M. Barriers to asthma management among urban families: caregiver and child perspectives. J Asthma 2009; 46:731–739.
- Lieu TA, Finkelstein JA, Lozano P, et al. Cultural competence policies and other predictors of asthma care quality for Medicaid-insured children. Pediatrics 2004; 114:e102–e110.
- DiMatteo MR. Variations in patients’ adherence to medical recommendations: a quantitative review of 50 years of research. Med Care 2004; 42:200–209.
- Bender B, Wamboldt FS, O’Connor SL, et al. Measurement of children’s asthma medication adherence by self report, mother report, canister weight, and Doser CT. Ann Allergy Asthma Immunol 2000; 85:416–421.
- Williams LK, Pladevall M, Xi H, et al. Relationship between adherence to inhaled corticosteroids and poor outcomes among adults with asthma. J Allergy Clin Immunol 2004; 114:1288–1293.
- Apter AJ, Boston RC, George M, et al. Modifiable barriers to adherence to inhaled steroids among adults with asthma: it’s not just black and white. J Allergy Clin Immunol 2003; 111:1219–1226.
- Apter AJ, Reisine ST, Affleck G, Barrows E, ZuWallack RL. Adherence with twice-daily dosing of inhaled steroids. Socioeconomic and health-belief differences. Am J Respir Crit Care Med 1998; 157:1810–1817.
- Williams LK, Joseph CL, Peterson EL, et al. Race-ethnicity, crime, and other factors associated with adherence to inhaled corticosteroids. J Allergy Clin Immunol 2007; 119:168–175.
- O’Malley AS, Sheppard VB, Schwartz M, Mandelblatt J. The role of trust in use of preventive services among low-income African-American women. Prev Med 2004; 38:777–785.
- Okelo SO, Wu AW, Merriman B, Krishnan JA, Diette GB. Are physician estimates of asthma severity less accurate in black than in white patients? J Gen Intern Med 2007; 22:976–981.
- Kinney PL, Aggarwal M, Northridge ME, Janssen NA, Shepard P. Airborne concentrations of PM(2.5) and diesel exhaust particles on Harlem sidewalks: a community-based pilot study. Environ Health Perspect 2000; 108:213–218.
- O’Neill MS, Jerrett M, Kawachi I, et al; Workshop on Air Pollution and Socioeconomic Conditions. Health, wealth, and air pollution: advancing theory and methods. Environ Health Perspect 2003; 111:1861–1870.
- Schwartz J, Slater D, Larson TV, Pierson WE, Koenig JQ. Particulate air pollution and hospital emergency room visits for asthma in Seattle. Am Rev Respir Dis 1993; 147:826–831.
- Norris G, YoungPong SN, Koenig JQ, Larson TV, Sheppard L, Stout JW. An association between fine particles and asthma emergency department visits for children in Seattle. Environ Health Perspect 1999; 107:489–493.
- Yu O, Sheppard L, Lumley T, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptoms of asthma in Seattle-area children enrolled in the CAMP study. Environ Health Perspect 2000; 108:1209–1214.
- Slaughter JC, Lumley T, Sheppard L, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptom severity and medication use in children with asthma. Ann Allergy Asthma Immunol 2003; 91:346–353.
- Koenig JQ, Jansen K, Mar TF, et al. Measurement of offline exhaled nitric oxide in a study of community exposure to air pollution. Environ Health Perspect 2003; 111:1625–1629.
- Centers for Disease Control and Prevention (CDC). Cigarette smoking among adults and trends in smoking cessation—United States, 2008. MMWR Morb Mortal Wkly Rep 2009; 58:1227–1232.
- Delva J, Tellez M, Finlayson TL, et al. Cigarette smoking among low-income African Americans: a serious public health problem. Am J Prev Med 2005; 29:218–220.
- McLeish AC, Zvolensky MJ. Asthma and cigarette smoking: a review of the empirical literature. J Asthma 2010; 47:345–361.
- Chaudhuri R, Livingston E, McMahon AD, Thomson L, Borland W, Thomson NC. Cigarette smoking impairs the therapeutic response to oral corticosteroids in chronic asthma. Am J Respir Crit Care Med 2003; 168:1308–1311.
- Wilson SE, Kahn RS, Khoury J, Lanphear BP. Racial differences in exposure to environmental tobacco smoke among children. Environ Health Perspect 2005; 113:362–367.
- Huss K, Rand CS, Butz AM, et al. Home environmental risk factors in urban minority asthmatic children. Ann Allergy 1994; 72:173–177.
- Samir S, Colin Y, Thomas S. Impact of environmental tobacco smoke on children admitted with status asthmaticus in the pediatric intensive care unit. Pediatr Pulmonol 2010. [Epub ahead of print]
- Lannerö E, Wickman M, Pershagen G, Nordvall L. Maternal smoking during pregnancy increases the risk of recurrent wheezing during the first years of life (BAMSE). Respir Res 2006; 7:3.
- Hedman L, Bjerg A, Sundberg S, Forsberg B, Rönmark E. Both environmental tobacco smoke and personal smoking is related to asthma and wheeze in teenagers. Thorax 2011; 66:20–25.
- Zhang G, Hayden CM, Khoo SK, et al. Beta2-adrenoceptor polymorphisms and asthma phenotypes: interactions with passive smoking. Eur Respir J 2007; 30:48–55.
- Rosenbaum E, Friedman S. The Housing Divide: How Generations of Immigrants Fare in New York’s Housing Market. New York, NY: New York University Press; 2007.
- Rosenbaum E. Racial/ethnic differences in asthma prevalence: the role of housing and neighborhood environments. J Health Soc Behav 2008; 49:131–145.
- Rauh VA, Chew GR, Garfinkel RS. Deteriorated housing contributes to high cockroach allergen levels in inner-city households. Environ Health Perspect 2002; 110( suppl 2):323–327.
- Eggleston PA, Rosenstreich D, Lynn H, et al. Relationship of indoor allergen exposure to skin test sensitivity in inner-city children with asthma. J Allergy Clin Immunol 1998; 102:563–570.
- Togias A, Horowitz E, Joyner D, Guydon L, Malveaux F. Evaluating the factors that relate to asthma severity in adolescents. Int Arch Allergy Immunol 1997; 113:87–95.
- Alp H, Yu BH, Grant EN, Rao V, Moy JN. Cockroach allergy appears early in life in inner-city children with recurrent wheezing. Ann Allergy Asthma Immunol 2001; 86:51–54.
- Miller RL, Chew GL, Bell CA, et al. Prenatal exposure, maternal sensitization, and sensitization in utero to indoor allergens in an inner-city cohort. Am J Respir Crit Care Med 2001; 164:995–1001.
- Morgan WJ, Crain EF, Gruchalla RS, et al; Inner-City Asthma Study Group. Results of a home-based environmental intervention among urban children with asthma. N Engl J Med 2004; 351:1068–1080.
- Centers for Disease Control and Prevention (CDC). Overweight and Obesity. US Obesity Trends. http://templatelab.com/us-obesity-trends/. Accessed February 1, 2012.
- Beuther DA, Sutherland ER. Overweight, obesity, and incident asthma: a meta-analysis of prospective epidemiologic studies. Am J Respir Crit Care Med 2007; 175:661–666.
- Visness CM, London SJ, Daniels JL, et al. Association of childhood obesity with atopic and nonatopic asthma: results from the National Health and Nutrition Examination Survey 1999–2006. J Asthma 2010; 47:822–829.
- Camargo CA, Sutherland ER, Bailey W, et al. Effect of increased body mass index on asthma risk, impairment and response to asthma controller therapy in African Americans. Curr Med Res Opin 2010; 26:1629–1635.
- Hakala K, Stenius-Aarniala B, Sovijärvi A. Effects of weight loss on peak flow variability, airways obstruction, and lung volumes in obese patients with asthma. Chest 2000; 118:1315–1321.
- Stenius-Aarniala B, Poussa T, Kvarnström J, Grönlund EL, Ylikahri M, Mustajoki P. Immediate and long term effects of weight reduction in obese people with asthma: randomised controlled study. BMJ 2000; 320:827–832.
- Dixon AE, Holguin F, Sood A, et al; American Thoracic Society Ad Hoc Subcommittee on Obesity and Lung Disease. An official American Thoracic Society Workshop report: obesity and asthma. Proc Am Thorac Soc 2010; 7:325–335.
- Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general US population. Am J Respir Crit Care Med 1999; 159:179–187.
- Harik-Khan RI, Muller DC, Wise RA. Racial difference in lung function in African-American and white children: effect of anthropometric, socioeconomic, nutritional, and environmental factors. Am J Epidemiol 2004; 160:893–900.
The last several decades have seen a dramatic surge in the prevalence of asthma. In 2009, there were an estimated 17.5 million adults and almost 7.1 million children with asthma in the United States,1 up from 9.5 million adults and slightly more than 5 million children in 1995.2
Multiple factors contribute to these disparities, including genetics, socioeconomic factors, cultural factors, health maintenance behaviors, provider-patient communication, air quality, and obesity.
GENETICS: 70% OF DESTINY?
The trend towards personalized medicine has spurred extensive research into the genetics of asthma. Studies in twins and familial aggregation studies suggest genetics plays a significant role, with estimates of the heritability of asthma as high as 70%.4,5 More than 100 candidate genes have been shown to be associated with asthma and atopy, 30 of them in five or more independent studies.6
Researchers face many challenges when investigating the genetics involved in asthma for a particular race. Race is both a biologic and a social construct and, as such, is a poor substitute for genetics. Race constitutes not only genetic differences in individuals, but also the behaviors, beliefs, and experiences that vary among races.
The clinical disease—the phenotype—is the product of the interaction of genes and these differing behaviors and exposures. Genetics can affect how environmental factors found in association with socioeconomic factors relate to asthma morbidity and mortality.
For example, as we will discuss below, African Americans are more likely than whites to be sensitized to cockroach allergen, even after controlling for socioeconomic variables that may be associated with greater exposure.7 High-level exposure to cockroach allergen in sensitized children has been associated with poor asthma outcomes.8 This suggests that a genetic difference may exist between African Americans and whites with respect to the potential to develop cockroach sensitization, and this difference may be of particular importance for those African Americans living in areas with higher levels of cockroach exposure.
Two polymorphisms
Two polymorphisms have garnered attention for their influence on African Americans with asthma:
TheADRB2gene. This gene codes for the beta-2 adrenergic receptor and resides at chromosome 5q13.9 The receptor is found on several types of cells in the lung, including airway smooth muscle and epithelial cells, and is responsible for the salutary effects of inhaled beta-2 agonists such as albuterol (eg, Proventil).
Allelic polymorphisms of this gene are clinically relevant. The substitution of arginine (Arg) for glycine (Gly) at codon 16 of this gene is responsible for differences in response to short-acting beta-2 agonists. The allelic frequency of Arg16 is lower in white Americans (39.3%) than in African Americans (49.2%), and thus African Americans are more likely to be homozygous for Arg16 (ie, to have the Arg/Arg genotype).10
People who are homozygous for Arg16 who use albuterol on a regular basis are at higher risk of untoward asthma outcomes.11 This is important, for several reasons. In general, adherence to inhaled corticosteroids is poor (not only in African Americans),12 and patients who do not take their inhaled corticosteroids as they should may rely on short-acting beta-2 agonists more frequently. Furthermore, African Americans may have a poorer response to the repeated doses of albuterol that are typically given in the emergency department and in the hospital for severe asthma exacerbations.13 Additionally, data suggest that Arg/Arg individuals have more frequent exacerbations independent of beta-agonist use,14 although curiously, patients who are homozygous for Arg16 have a greater benefit from single doses of short-acting beta-2 agonists than those who are Gly16 homozygous.15
TheCD14gene. An interesting relationship between innate immunity and asthma has recently been described. Polymorphisms of CD14, which codes for a receptor for endotoxin, have been uncovered. The single-nucleotide polymorphism variant thymine (T) at position −260 has been found in greater frequency in whites than in African Americans, who are more likely to have the cytosine (C) allele.16 An association between the CC genotype and atopy has been reported,16 although this has not been consistent.17
A possible explanation for these inconsistencies may lie in complex gene-environment interactions. The amount of endotoxin exposure may play a role in phenotypic expression. Individuals with the CC genotype were at lower risk of developing atopy when exposed to high levels of endotoxin; however, when exposed to lower levels of endotoxin, the CC genotype was associated with a higher risk of atopy.18 Nonfarm homes in westernized countries tend to have lower levels of endotoxin than farm homes, even in low-income urban areas.19 This implies that individuals with the CC allele, who are more likely to be African American, would be at greater risk for atopy in the United States. Greater knowledge of these types of gene-environment interactions may lead to improved understanding of the observations that have generated controversy concerning the “hygiene hypothesis.”
The details of how microbial exposure can influence the human immune response to antigen exposure are still being elucidated.20
These examples highlight not only the importance of genetics in the development of asthma, but also the role genes play in variation of treatment response and subsequent risk of morbidity and death. An understanding of these genetic differences among patients is clearly important for moving towards personalized treatment strategies for asthma.
Ancestry-informative markers
A developing strategy to assess the differences in asthma prevalence, severity, and response to treatment between racial groups is the use of ancestry-informative markers (AIMs).
AIMs are single-nucleotide polymorphisms that occur in varying allelic frequencies between ancestral groups, eg, continental Africans or European whites.21 AIMs provide an estimate of an individual’s proportion of ancestry—ie, of how “African” an African American is genetically.
African ancestry, determined using AIMs, was found to be associated with asthma in people living on the Caribbean coast of Colombia.22 However, one study found that AIMs could not predict an individual’s response to inhaled corticosteroids.23
Further research is necessary to find a technique to determine how groups of individuals can be characterized more precisely and managed more appropriately.
SOCIOECONOMIC FACTORS
African Americans living in low-income urban areas have an even greater prevalence of asthma and a greater risk of asthma-related morbidity and death than African Americans overall.3,24,25 Urban areas typically have a high proportion of residents living at or below the poverty level, and minorities often constitute a substantial proportion of the population in these areas. Evidence suggests that both African American race and lower socioeconomic status are independent risk factors for asthma prevalence, morbidity, and death.3,25
To provide better care for African Americans living in low-income urban areas, it is important to understand the factors that may be contributing to the higher morbidity and mortality rates in low-income urban areas.
Inadequate follow-up
Proper and routine follow-up for evaluation of asthma symptoms is essential for appropriate management. The Expert Panel Report 3 (EPR-3) of the National Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma,26 published in 2007, recommends that patients be seen at least every 6 months if they have been experiencing good control. While gaining control, patients should be seen every 2 to 6 weeks.26
Despite these recommendations, numerous studies have suggested that African Americans do not receive adequate follow-up. Children who are poor, are nonwhite or Hispanic, or are underinsured are more likely to lack routine health care27 and, more specifically, routine asthma care.28 Low-income patients are also more likely to receive care in a large hospital-run clinic or neighborhood clinic,27,28 where continuity of care may be less than ideal.29 Even among patients enrolled in Medicaid or Medicare, African American children with a primary care provider have fewer asthma visits compared with white Medicaid-insured children.30
Insufficient follow-up care contributes to greater asthma morbidity, resulting in, for example, more emergency department visits for asthma in African Americans.27,31,32
Suboptimal care
Data also suggest that the quality of care that residents of low-income urban areas receive is often suboptimal. Many people living in low-income urban areas are not provided with the knowledge and tools to treat asthma exacerbations at home.33 African Americans are also less likely to be seen by an asthma specialist31,34 as recommended for those with moderate or severe asthma.26
The EPR-3 guidelines also stress the importance of inhaled corticosteroids as the preferred therapy for all patients with persistent asthma. Even after controlling for the number of primary care visits, insurance status, and disease severity, African Americans are less likely to receive a prescription for inhaled corticosteroids, or they receive the same dosage of inhaled corticosteroids in the face of more severe disease.31,33,35,36
The reasons for these differences in treatment are not fully understood but are likely multiple. Lack of access to an asthma specialist and financial or formulary constraints are some of the potential barriers to optimal asthma care outcomes.
Misdiagnosis in the acute setting may also be a source of less-than-ideal care, as patients seen in emergency departments may be misdiagnosed with viral infection or bronchitis.
African Americans may report different symptoms than whites
Intriguing studies suggest that African Americans report different symptoms while describing asthma exacerbations.
In one study, compared with whites, African Americans were less likely to report nocturnal symptoms, dyspnea, or chest pain during exacerbations.37 In another study, when given a methacholine challenge that induced a significant drop in forced expiratory volume in 1 second (FEV1), African Americans with asthma were more likely to complain of upper airway symptoms as opposed to lower airway symptoms, compared with white patients.38
The symptoms that African Americans describe, such as having a tight throat or voice, are not typically regarded as related to asthma; for this reason, such descriptions may be an obstacle to correct diagnosis, management, and follow-up.
Asthma care providers should be aware of these observations to ensure that their patients are managed appropriately.
Lack of social support
Living in a low-income urban area presents many challenges that can interfere with proper asthma control.
Asthma diagnosis, management, and morbidity are affected by family support.39 Patients with asthma who lack sufficient financial support for treatment, who lack adequate psychological support, and who have more major life stressors are at higher risk of untoward outcomes. Disruption and dysfunction of the family and the supports available have been associated with greater asthma morbidity.40–42 Unfortunately, these types of stressors are all too common in families living in low-income urban areas.43–45
Multiple stressors that can occur more often in low-income urban areas, including exposure to violent crime, have also been linked to greater asthma morbidity.45–47
POOR PHYSICIAN-PATIENT COMMUNICATION
A consistent theme in focus groups of African Americans living in inner-city areas is the perception that health care providers are not effectively communicating and taking the time to listen to their concerns.48,49 Respondents believed they had better insight into their illness than their providers, and for this reason were better able to manage their disease.
The importance of an optimal provider-patient relationship was highlighted by a prospective cohort study in which Medicaid children receiving care at physician’s offices with the highest cultural competency scores were more adherent with their asthma controller medications.50
MEDICATION ADHERENCE RATES ARE DISTURBINGLY LOW
Rates of medication adherence for chronic diseases is disturbingly low, and may be even worse for pulmonary diseases.51 Reported rates of adherence to asthma medications among all patients range from 50% to 60%.52,53 Several studies showed that African Americans have a lower rate of adherence than do whites,53–55 even after adjusting for multiple socioeconomic variables.56
Many explanations have been proposed for this discrepancy, and all likely play a role in particular environments. For example, the incidence of crime in the surrounding area was inversely related to medication adherence after adjusting for socioeconomic factors.57 African Americans may have more concern about side effects associated with inhaled corticosteroid use and may be less likely to understand how these drugs work.52,53 A poor provider-patient relationship has also been cited as a barrier to adherence.55,57 Finally, physicians are more likely to underestimate asthma severity in an African American patient than in a white patient.58
Taking the time to ensure that patients truly understand all aspects of their disease and establishing a health care environment that is culturally appropriate may have a significant impact in patients with asthma.
ENVIRONMENTAL EXPOSURES
Air quality contributes to the greater asthma morbidity observed in urban residents, including African Americans. While poor outdoor air quality has not been clearly linked to a higher incidence of asthma, it has been associated with greater asthma morbidity. Poor air quality may affect individuals of all races, but with respect to ambient pollutants such as particulate matter and diesel exhaust, outdoor air quality is worse in urban environments where greater proportions of people of low socioeconomic status reside.59,60
The most extensively studied components of air pollution are ozone, sulfur dioxide, and particulate matter. These pollutants have been associated with a higher rate of emergency department visits,61,62 worse asthma symptoms,63,64 and higher exhaled nitric oxide levels.65
Tobacco smoke
Despite the substantial success of smoking cessation efforts nationwide, exposure to tobacco smoke continues to be common and is a significant risk factor for poor asthma control. Recent data suggest that African Americans and whites have a similar prevalence of smoking,66 but a study found a very high prevalence in low-income African Americans.67
Active smoking has been associated with worse asthma control and a higher risk of death.68 People with asthma who smoke are less likely to improve in their lung function and symptom scores when treated with short courses of oral glucocorticoids compared with both nonsmokers and former smokers.69
Secondhand smoke hurts too. Many children living in low-income urban areas are exposed to secondhand smoke or environmental tobacco smoke.70,71 Passive exposure in children has been associated with worse asthma outcomes, and data suggest such exposure may be a cause of asthma.68,72–74
Environmental tobacco smoke has also been implicated in gene-environment interactions. Patients who are either homozygous or heterozygous for the Arg allele at codon 16 of the ADRB2 gene (discussed above) had significantly lower FEV1 and forced vital capacity (FVC) values when exposed to passive tobacco smoke. This difference was not seen in people who were not exposed.75
Cockroach allergen
The type and condition of a person’s housing also plays a role in asthma-related morbidity and death. Across several socioeconomic levels, it has been suggested that African Americans have poorer-quality housing compared with whites.76 Some of the conditions found in low-quality houses, such as interruptions in heat, plumbing leaks, and the presence of rodents, have been associated with a higher prevalence of asthma in the household.77
Cockroach allergen exposure and sensitization is a major contributor to asthma morbidity in African Americans, particularly those living in poorer urban areas where cockroach allergen may be the most common indoor allergen.8 Living in older housing in urban areas is associated with higher exposure to cockroach allergen, and with subsequent sensitization.78,79 Exposure to high levels of the major cockroach allergen, Bla g 1, in sensitized individuals has been linked to a greater risk of hospitalization and unscheduled medical visits for asthma. This was not found to be the case for other common indoor allergens, such as dust mite and cat dander.8
However, it is not only exposure to high cockroach allergen levels that puts African Americans at risk. African Americans living in low-income urban areas may also be more likely than whites living in low-income urban areas to become sensitized to cockroach allergen.7,80 This suggests a gene-environment interaction that may be unique to African Americans. Moreover, cockroach sensitization may occur early in life.81,82
While successful cockroach avoidance measures and environmental control may be challenging, such measures have been shown to decrease rates of asthma morbidity.83
OBESITY
Obesity has been linked to an ever-growing list of diseases, one of which is asthma. Obesity is not a unique challenge for African Americans, but recent data from the US Centers for Disease Control and Prevention show that African Americans have a 51% higher prevalence of obesity compared with whites.84
Obesity is a risk factor for greater asthma morbidity and is a significant challenge in the African American community. The rise in obesity rates has paralleled the rise in asthma in recent decades. The higher one’s body mass index, the higher one’s risk of asthma.85 This association appears to be stronger in people without concurrent atopic disease.86 Obesity has also been associated with a poorer response to inhaled corticosteroids and a higher risk of asthma exacerbations.87 Interestingly, significant weight loss has been associated with improvements in both asthma control and lung function.88,89
What is the mechanism?
The underlying pathogenic mechanisms have not been completely elucidated, and they are likely multiple.
Adipokines (cytokines secreted by adipocytes) have been implicated. Two of the most extensively studied adipokines are leptin and adiponectin. Leptin production is increased in obesity, and it has inflammatory effects on both the innate and adaptive immune systems.90 The opposite is true for adiponectin, which may have anti-inflammatory properties and which decreases as the body mass index increases.90 The precise role these molecules may have in lung disease is undergoing further investigation.
Mechanical alterations in lung function may also contribute. Obese people have a lower functional residual capacity and expiratory reserve volume. Breathing with a lower-volume functional residual capacity results in decreased airway diameter and contributes to increased airway resistance.90 The decreased airway diameter may alter the contractile properties of airway smooth muscle and lead to increased airway responsiveness.90 These differences are in addition to the lower mean values of common spirometry indices such as the FEV1 and FVC, found in nonasthmatic African Americans compared with whites.91
Data suggest these differences are primarily due to anthropometric factors, with nutritional and environmental factors playing a less significant role.92 On this basis, the American Thoracic Society recommends applying race-specific reference standards for use with spirometry in order to accurately gauge lung function in African Americans.
APPROPRIATE CARE AND EDUCATION
The cause of greater asthma prevalence and severity among African Americans is multifactorial. It is likely that a number of factors work together, rather than separately, in influencing the development of asthma and its course.
Some risk factors are avoidable, and it is important to identify and ameliorate them. Others are not preventable, but knowledge of them may provide more specific management strategies and may lead to new therapies in the future.
While more work is needed to further unravel the complex risk factors associated with asthma, ensuring that higher-risk patients are provided the appropriate care and the knowledge to help control their disease is a necessary step in improving the disparities in asthma care outcomes.
The last several decades have seen a dramatic surge in the prevalence of asthma. In 2009, there were an estimated 17.5 million adults and almost 7.1 million children with asthma in the United States,1 up from 9.5 million adults and slightly more than 5 million children in 1995.2
Multiple factors contribute to these disparities, including genetics, socioeconomic factors, cultural factors, health maintenance behaviors, provider-patient communication, air quality, and obesity.
GENETICS: 70% OF DESTINY?
The trend towards personalized medicine has spurred extensive research into the genetics of asthma. Studies in twins and familial aggregation studies suggest genetics plays a significant role, with estimates of the heritability of asthma as high as 70%.4,5 More than 100 candidate genes have been shown to be associated with asthma and atopy, 30 of them in five or more independent studies.6
Researchers face many challenges when investigating the genetics involved in asthma for a particular race. Race is both a biologic and a social construct and, as such, is a poor substitute for genetics. Race constitutes not only genetic differences in individuals, but also the behaviors, beliefs, and experiences that vary among races.
The clinical disease—the phenotype—is the product of the interaction of genes and these differing behaviors and exposures. Genetics can affect how environmental factors found in association with socioeconomic factors relate to asthma morbidity and mortality.
For example, as we will discuss below, African Americans are more likely than whites to be sensitized to cockroach allergen, even after controlling for socioeconomic variables that may be associated with greater exposure.7 High-level exposure to cockroach allergen in sensitized children has been associated with poor asthma outcomes.8 This suggests that a genetic difference may exist between African Americans and whites with respect to the potential to develop cockroach sensitization, and this difference may be of particular importance for those African Americans living in areas with higher levels of cockroach exposure.
Two polymorphisms
Two polymorphisms have garnered attention for their influence on African Americans with asthma:
TheADRB2gene. This gene codes for the beta-2 adrenergic receptor and resides at chromosome 5q13.9 The receptor is found on several types of cells in the lung, including airway smooth muscle and epithelial cells, and is responsible for the salutary effects of inhaled beta-2 agonists such as albuterol (eg, Proventil).
Allelic polymorphisms of this gene are clinically relevant. The substitution of arginine (Arg) for glycine (Gly) at codon 16 of this gene is responsible for differences in response to short-acting beta-2 agonists. The allelic frequency of Arg16 is lower in white Americans (39.3%) than in African Americans (49.2%), and thus African Americans are more likely to be homozygous for Arg16 (ie, to have the Arg/Arg genotype).10
People who are homozygous for Arg16 who use albuterol on a regular basis are at higher risk of untoward asthma outcomes.11 This is important, for several reasons. In general, adherence to inhaled corticosteroids is poor (not only in African Americans),12 and patients who do not take their inhaled corticosteroids as they should may rely on short-acting beta-2 agonists more frequently. Furthermore, African Americans may have a poorer response to the repeated doses of albuterol that are typically given in the emergency department and in the hospital for severe asthma exacerbations.13 Additionally, data suggest that Arg/Arg individuals have more frequent exacerbations independent of beta-agonist use,14 although curiously, patients who are homozygous for Arg16 have a greater benefit from single doses of short-acting beta-2 agonists than those who are Gly16 homozygous.15
TheCD14gene. An interesting relationship between innate immunity and asthma has recently been described. Polymorphisms of CD14, which codes for a receptor for endotoxin, have been uncovered. The single-nucleotide polymorphism variant thymine (T) at position −260 has been found in greater frequency in whites than in African Americans, who are more likely to have the cytosine (C) allele.16 An association between the CC genotype and atopy has been reported,16 although this has not been consistent.17
A possible explanation for these inconsistencies may lie in complex gene-environment interactions. The amount of endotoxin exposure may play a role in phenotypic expression. Individuals with the CC genotype were at lower risk of developing atopy when exposed to high levels of endotoxin; however, when exposed to lower levels of endotoxin, the CC genotype was associated with a higher risk of atopy.18 Nonfarm homes in westernized countries tend to have lower levels of endotoxin than farm homes, even in low-income urban areas.19 This implies that individuals with the CC allele, who are more likely to be African American, would be at greater risk for atopy in the United States. Greater knowledge of these types of gene-environment interactions may lead to improved understanding of the observations that have generated controversy concerning the “hygiene hypothesis.”
The details of how microbial exposure can influence the human immune response to antigen exposure are still being elucidated.20
These examples highlight not only the importance of genetics in the development of asthma, but also the role genes play in variation of treatment response and subsequent risk of morbidity and death. An understanding of these genetic differences among patients is clearly important for moving towards personalized treatment strategies for asthma.
Ancestry-informative markers
A developing strategy to assess the differences in asthma prevalence, severity, and response to treatment between racial groups is the use of ancestry-informative markers (AIMs).
AIMs are single-nucleotide polymorphisms that occur in varying allelic frequencies between ancestral groups, eg, continental Africans or European whites.21 AIMs provide an estimate of an individual’s proportion of ancestry—ie, of how “African” an African American is genetically.
African ancestry, determined using AIMs, was found to be associated with asthma in people living on the Caribbean coast of Colombia.22 However, one study found that AIMs could not predict an individual’s response to inhaled corticosteroids.23
Further research is necessary to find a technique to determine how groups of individuals can be characterized more precisely and managed more appropriately.
SOCIOECONOMIC FACTORS
African Americans living in low-income urban areas have an even greater prevalence of asthma and a greater risk of asthma-related morbidity and death than African Americans overall.3,24,25 Urban areas typically have a high proportion of residents living at or below the poverty level, and minorities often constitute a substantial proportion of the population in these areas. Evidence suggests that both African American race and lower socioeconomic status are independent risk factors for asthma prevalence, morbidity, and death.3,25
To provide better care for African Americans living in low-income urban areas, it is important to understand the factors that may be contributing to the higher morbidity and mortality rates in low-income urban areas.
Inadequate follow-up
Proper and routine follow-up for evaluation of asthma symptoms is essential for appropriate management. The Expert Panel Report 3 (EPR-3) of the National Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma,26 published in 2007, recommends that patients be seen at least every 6 months if they have been experiencing good control. While gaining control, patients should be seen every 2 to 6 weeks.26
Despite these recommendations, numerous studies have suggested that African Americans do not receive adequate follow-up. Children who are poor, are nonwhite or Hispanic, or are underinsured are more likely to lack routine health care27 and, more specifically, routine asthma care.28 Low-income patients are also more likely to receive care in a large hospital-run clinic or neighborhood clinic,27,28 where continuity of care may be less than ideal.29 Even among patients enrolled in Medicaid or Medicare, African American children with a primary care provider have fewer asthma visits compared with white Medicaid-insured children.30
Insufficient follow-up care contributes to greater asthma morbidity, resulting in, for example, more emergency department visits for asthma in African Americans.27,31,32
Suboptimal care
Data also suggest that the quality of care that residents of low-income urban areas receive is often suboptimal. Many people living in low-income urban areas are not provided with the knowledge and tools to treat asthma exacerbations at home.33 African Americans are also less likely to be seen by an asthma specialist31,34 as recommended for those with moderate or severe asthma.26
The EPR-3 guidelines also stress the importance of inhaled corticosteroids as the preferred therapy for all patients with persistent asthma. Even after controlling for the number of primary care visits, insurance status, and disease severity, African Americans are less likely to receive a prescription for inhaled corticosteroids, or they receive the same dosage of inhaled corticosteroids in the face of more severe disease.31,33,35,36
The reasons for these differences in treatment are not fully understood but are likely multiple. Lack of access to an asthma specialist and financial or formulary constraints are some of the potential barriers to optimal asthma care outcomes.
Misdiagnosis in the acute setting may also be a source of less-than-ideal care, as patients seen in emergency departments may be misdiagnosed with viral infection or bronchitis.
African Americans may report different symptoms than whites
Intriguing studies suggest that African Americans report different symptoms while describing asthma exacerbations.
In one study, compared with whites, African Americans were less likely to report nocturnal symptoms, dyspnea, or chest pain during exacerbations.37 In another study, when given a methacholine challenge that induced a significant drop in forced expiratory volume in 1 second (FEV1), African Americans with asthma were more likely to complain of upper airway symptoms as opposed to lower airway symptoms, compared with white patients.38
The symptoms that African Americans describe, such as having a tight throat or voice, are not typically regarded as related to asthma; for this reason, such descriptions may be an obstacle to correct diagnosis, management, and follow-up.
Asthma care providers should be aware of these observations to ensure that their patients are managed appropriately.
Lack of social support
Living in a low-income urban area presents many challenges that can interfere with proper asthma control.
Asthma diagnosis, management, and morbidity are affected by family support.39 Patients with asthma who lack sufficient financial support for treatment, who lack adequate psychological support, and who have more major life stressors are at higher risk of untoward outcomes. Disruption and dysfunction of the family and the supports available have been associated with greater asthma morbidity.40–42 Unfortunately, these types of stressors are all too common in families living in low-income urban areas.43–45
Multiple stressors that can occur more often in low-income urban areas, including exposure to violent crime, have also been linked to greater asthma morbidity.45–47
POOR PHYSICIAN-PATIENT COMMUNICATION
A consistent theme in focus groups of African Americans living in inner-city areas is the perception that health care providers are not effectively communicating and taking the time to listen to their concerns.48,49 Respondents believed they had better insight into their illness than their providers, and for this reason were better able to manage their disease.
The importance of an optimal provider-patient relationship was highlighted by a prospective cohort study in which Medicaid children receiving care at physician’s offices with the highest cultural competency scores were more adherent with their asthma controller medications.50
MEDICATION ADHERENCE RATES ARE DISTURBINGLY LOW
Rates of medication adherence for chronic diseases is disturbingly low, and may be even worse for pulmonary diseases.51 Reported rates of adherence to asthma medications among all patients range from 50% to 60%.52,53 Several studies showed that African Americans have a lower rate of adherence than do whites,53–55 even after adjusting for multiple socioeconomic variables.56
Many explanations have been proposed for this discrepancy, and all likely play a role in particular environments. For example, the incidence of crime in the surrounding area was inversely related to medication adherence after adjusting for socioeconomic factors.57 African Americans may have more concern about side effects associated with inhaled corticosteroid use and may be less likely to understand how these drugs work.52,53 A poor provider-patient relationship has also been cited as a barrier to adherence.55,57 Finally, physicians are more likely to underestimate asthma severity in an African American patient than in a white patient.58
Taking the time to ensure that patients truly understand all aspects of their disease and establishing a health care environment that is culturally appropriate may have a significant impact in patients with asthma.
ENVIRONMENTAL EXPOSURES
Air quality contributes to the greater asthma morbidity observed in urban residents, including African Americans. While poor outdoor air quality has not been clearly linked to a higher incidence of asthma, it has been associated with greater asthma morbidity. Poor air quality may affect individuals of all races, but with respect to ambient pollutants such as particulate matter and diesel exhaust, outdoor air quality is worse in urban environments where greater proportions of people of low socioeconomic status reside.59,60
The most extensively studied components of air pollution are ozone, sulfur dioxide, and particulate matter. These pollutants have been associated with a higher rate of emergency department visits,61,62 worse asthma symptoms,63,64 and higher exhaled nitric oxide levels.65
Tobacco smoke
Despite the substantial success of smoking cessation efforts nationwide, exposure to tobacco smoke continues to be common and is a significant risk factor for poor asthma control. Recent data suggest that African Americans and whites have a similar prevalence of smoking,66 but a study found a very high prevalence in low-income African Americans.67
Active smoking has been associated with worse asthma control and a higher risk of death.68 People with asthma who smoke are less likely to improve in their lung function and symptom scores when treated with short courses of oral glucocorticoids compared with both nonsmokers and former smokers.69
Secondhand smoke hurts too. Many children living in low-income urban areas are exposed to secondhand smoke or environmental tobacco smoke.70,71 Passive exposure in children has been associated with worse asthma outcomes, and data suggest such exposure may be a cause of asthma.68,72–74
Environmental tobacco smoke has also been implicated in gene-environment interactions. Patients who are either homozygous or heterozygous for the Arg allele at codon 16 of the ADRB2 gene (discussed above) had significantly lower FEV1 and forced vital capacity (FVC) values when exposed to passive tobacco smoke. This difference was not seen in people who were not exposed.75
Cockroach allergen
The type and condition of a person’s housing also plays a role in asthma-related morbidity and death. Across several socioeconomic levels, it has been suggested that African Americans have poorer-quality housing compared with whites.76 Some of the conditions found in low-quality houses, such as interruptions in heat, plumbing leaks, and the presence of rodents, have been associated with a higher prevalence of asthma in the household.77
Cockroach allergen exposure and sensitization is a major contributor to asthma morbidity in African Americans, particularly those living in poorer urban areas where cockroach allergen may be the most common indoor allergen.8 Living in older housing in urban areas is associated with higher exposure to cockroach allergen, and with subsequent sensitization.78,79 Exposure to high levels of the major cockroach allergen, Bla g 1, in sensitized individuals has been linked to a greater risk of hospitalization and unscheduled medical visits for asthma. This was not found to be the case for other common indoor allergens, such as dust mite and cat dander.8
However, it is not only exposure to high cockroach allergen levels that puts African Americans at risk. African Americans living in low-income urban areas may also be more likely than whites living in low-income urban areas to become sensitized to cockroach allergen.7,80 This suggests a gene-environment interaction that may be unique to African Americans. Moreover, cockroach sensitization may occur early in life.81,82
While successful cockroach avoidance measures and environmental control may be challenging, such measures have been shown to decrease rates of asthma morbidity.83
OBESITY
Obesity has been linked to an ever-growing list of diseases, one of which is asthma. Obesity is not a unique challenge for African Americans, but recent data from the US Centers for Disease Control and Prevention show that African Americans have a 51% higher prevalence of obesity compared with whites.84
Obesity is a risk factor for greater asthma morbidity and is a significant challenge in the African American community. The rise in obesity rates has paralleled the rise in asthma in recent decades. The higher one’s body mass index, the higher one’s risk of asthma.85 This association appears to be stronger in people without concurrent atopic disease.86 Obesity has also been associated with a poorer response to inhaled corticosteroids and a higher risk of asthma exacerbations.87 Interestingly, significant weight loss has been associated with improvements in both asthma control and lung function.88,89
What is the mechanism?
The underlying pathogenic mechanisms have not been completely elucidated, and they are likely multiple.
Adipokines (cytokines secreted by adipocytes) have been implicated. Two of the most extensively studied adipokines are leptin and adiponectin. Leptin production is increased in obesity, and it has inflammatory effects on both the innate and adaptive immune systems.90 The opposite is true for adiponectin, which may have anti-inflammatory properties and which decreases as the body mass index increases.90 The precise role these molecules may have in lung disease is undergoing further investigation.
Mechanical alterations in lung function may also contribute. Obese people have a lower functional residual capacity and expiratory reserve volume. Breathing with a lower-volume functional residual capacity results in decreased airway diameter and contributes to increased airway resistance.90 The decreased airway diameter may alter the contractile properties of airway smooth muscle and lead to increased airway responsiveness.90 These differences are in addition to the lower mean values of common spirometry indices such as the FEV1 and FVC, found in nonasthmatic African Americans compared with whites.91
Data suggest these differences are primarily due to anthropometric factors, with nutritional and environmental factors playing a less significant role.92 On this basis, the American Thoracic Society recommends applying race-specific reference standards for use with spirometry in order to accurately gauge lung function in African Americans.
APPROPRIATE CARE AND EDUCATION
The cause of greater asthma prevalence and severity among African Americans is multifactorial. It is likely that a number of factors work together, rather than separately, in influencing the development of asthma and its course.
Some risk factors are avoidable, and it is important to identify and ameliorate them. Others are not preventable, but knowledge of them may provide more specific management strategies and may lead to new therapies in the future.
While more work is needed to further unravel the complex risk factors associated with asthma, ensuring that higher-risk patients are provided the appropriate care and the knowledge to help control their disease is a necessary step in improving the disparities in asthma care outcomes.
- Akinbami LJ, Moorman JE, Liu X. Asthma prevalence, health care use, and mortality: United States, 2005–2009. Natl Health Stat Report 2011; 32:1–14.
- National Institutes of Health. National Heart, Lung, and Blood Institute. Data Fact Sheet. Asthma statistics. January 1999. http://www.nhlbi.nih.gov/health/prof/lung/asthma/asthstat.pdf. Accessed February 1, 2012.
- Lang DM, Polansky M, Sherman MS. Hospitalizations for asthma in an urban population: 1995–1999. Ann Allergy Asthma Immunol 2009; 103:128–133.
- Duffy DL, Martin NG, Battistutta D, Hopper JL, Mathews JD. Genetics of asthma and hay fever in Australian twins. Am Rev Respir Dis 1990; 142:1351–1358.
- Koeppen-Schomerus G, Stevenson J, Plomin R. Genes and environment in asthma: a study of 4 year old twins. Arch Dis Child 2001; 85:398–400.
- Meng JF, Rosenwasser LJ. Unraveling the genetic basis of asthma and allergic diseases. Allergy Asthma Immunol Res 2010; 2:215–227.
- Stevenson LA, Gergen PJ, Hoover DR, Rosenstreich D, Mannino DM, Matte TD. Sociodemographic correlates of indoor allergen sensitivity among United States children. J Allergy Clin Immunol 2001; 108:747–752.
- Rosenstreich DL, Eggleston P, Kattan M, et al. The role of cockroach allergy and exposure to cockroach allergen in causing morbidity among inner-city children with asthma. N Engl J Med 1997; 336:1356–1363.
- Kobilka BK, Dixon RA, Frielle T, et al. cDNA for the human beta 2-adrenergic receptor: a protein with multiple membrane-spanning domains and encoded by a gene whose chromosomal location is shared with that of the receptor for platelet-derived growth factor. Proc Natl Acad Sci U S A 1987; 84:46–50.
- Maxwell TJ, Ameyaw MM, Pritchard S, et al. Beta-2 adrenergic receptor genotypes and haplotypes in different ethnic groups. Int J Mol Med 2005; 16:573–580.
- Israel E, Chinchilli VM, Ford JG, et al; National Heart, Lung, and Blood Institute’s Asthma Clinical Research Network. Use of regularly scheduled albuterol treatment in asthma: genotype-stratified, randomised, placebo-controlled cross-over trial. Lancet 2004; 364:1505–1512.
- Wells K, Pladevall M, Peterson EL, et al. Race-ethnic differences in factors associated with inhaled steroid adherence among adults with asthma. Am J Respir Crit Care Med 2008; 178:1194–1201.
- Carroll CL, Stoltz P, Schramm CM, Zucker AR. Beta2-adrenergic receptor polymorphisms affect response to treatment in children with severe asthma exacerbations. Chest 2009; 135:1186–1192.
- Bleecker ER, Nelson HS, Kraft M, et al. Beta2-receptor polymorphisms in patients receiving salmeterol with or without fluticasone propionate. Am J Respir Crit Care Med 2010; 181:676–687.
- Finkelstein Y, Bournissen FG, Hutson JR, Shannon M. Polymorphism of the ADRB2 gene and response to inhaled beta-agonists in children with asthma: a meta-analysis. J Asthma 2009; 46:900–905.
- Baldini M, Lohman IC, Halonen M, Erickson RP, Holt PG, Martinez FD. A polymorphism in the 5’ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am J Respir Cell Mol Biol 1999; 20:976–983.
- Kedda MA, Lose F, Duffy D, Bell E, Thompson PJ, Upham J. The CD14 C-159T polymorphism is not associated with asthma or asthma severity in an Australian adult population. Thorax 2005; 60:211–214.
- Zambelli-Weiner A, Ehrlich E, Stockton ML, et al. Evaluation of the CD14/-260 polymorphism and house dust endotoxin exposure in the Barbados Asthma Genetics Study. J Allergy Clin Immunol 2005; 115:1203–1209.
- Perzanowski MS, Miller RL, Thorne PS, et al. Endotoxin in inner-city homes: associations with wheeze and eczema in early childhood. J Allergy Clin Immunol 2006; 117:1082–1089.
- Williams LK, Oliver J, Peterson EL, et al. Gene-environment interactions between CD14 C-260T and endotoxin exposure on Foxp3+ and Foxp3− CD4+ lymphocyte numbers and total serum IgE levels in early childhood. Ann Allergy Asthma Immunol 2008; 100:128–136.
- Barnes KC. Ancestry, ancestry-informative markers, asthma, and the quest for personalized medicine. J Allergy Clin Immunol 2010; 126:1139–1140.
- Vergara C, Caraballo L, Mercado D, et al. African ancestry is associated with risk of asthma and high total serum IgE in a population from the Caribbean Coast of Colombia. Hum Genet 2009; 125:565–579.
- Gould W, Peterson EL, Karungi G, et al. Factors predicting inhaled corticosteroid responsiveness in African American patients with asthma. J Allergy Clin Immunol 2010; 126:1131–1138.
- Lang DM, Polansky M. Patterns of asthma mortality in Philadelphia from 1969 to 1991. N Engl J Med 1994; 331:1542–1546.
- Schwartz J, Gold D, Dockery DW, Weiss ST, Speizer FE. Predictors of asthma and persistent wheeze in a national sample of children in the United States. Association with social class, perinatal events, and race. Am Rev Respir Dis 1990; 142:555–562.
- National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma. Summary Report 2007. J Allergy Clin Immunol 2007; 120( suppl 5):S94–S138.
- Newacheck PW, Hughes DC, Stoddard JJ. Children’s access to primary care: differences by race, income, and insurance status. Pediatrics 1996; 97:26–32.
- Halfon N, Newacheck PW. Childhood asthma and poverty: differential impacts and utilization of health services. Pediatrics 1993; 91:56–61.
- Burt CW, Knapp DE. Ambulatory care visits for asthma: United States, 1993–94. Adv Data 1996; 277:1.
- Lieu TA, Lozano P, Finkelstein JA, et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics 2002; 109:857–865.
- Zoratti EM, Havstad S, Rodriguez J, Robens-Paradise Y, Lafata JE, McCarthy B. Health service use by African Americans and Caucasians with asthma in a managed care setting. Am J Respir Crit Care Med 1998; 158:371–377.
- Wisnivesky JP, Leventhal H, Halm EA. Predictors of asthma-related health care utilization and quality of life among inner-city patients with asthma. J Allergy Clin Immunol 2005; 116:636–642.
- Halm EA, Wisnivesky JP, Leventhal H. Quality and access to care among a cohort of inner-city adults with asthma: who gets guideline concordant care? Chest 2005; 128:1943–1950.
- Krishnan JA, Diette GB, Skinner EA, Clark BD, Steinwachs D, Wu AW. Race and sex differences in consistency of care with national asthma guidelines in managed care organizations. Arch Intern Med 2001; 161:1660–1668.
- Nelson A. Unequal treatment: confronting racial and ethnic disparities in health care. J Natl Med Assoc 2002; 94:666–668.
- Boudreaux ED, Emond SD, Clark S, Camargo CA; Multicenter Airway Research Collaboration Investigators. Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics 2003; 111:e615–e621.
- Trochtenberg DS, BeLue R, Piphus S, Washington N. Differing reports of asthma symptoms in African Americans and Caucasians. J Asthma 2008; 45:165–170.
- Hardie GE, Janson S, Gold WM, Carrieri-Kohlman V, Boushey HA. Ethnic differences: word descriptors used by African-American and white asthma patients during induced bronchoconstriction. Chest 2000; 117:935–943.
- Clark NM, Levison MJ, Evans D, Wasilewski Y, Feldman CH, Mellins RB. Communication within low income families and the management of asthma. Patient Educ Couns 1990; 15:191–210.
- Rhee H, Belyea MJ, Brasch J. Family support and asthma outcomes in adolescents: barriers to adherence as a mediator. J Adolesc Health 2010; 47:472–478.
- Quinn K, Kaufman JS, Siddiqi A, Yeatts KB. Parent perceptions of neighborhood stressors are associated with general health and child respiratory health among low-income, urban families. J Asthma 2010; 47:281–289.
- Loerbroks A, Apfelbacher CJ, Bosch JA, Stürmer T. Depressive symptoms, social support, and risk of adult asthma in a population-based cohort study. Psychosom Med 2010; 72:309–315.
- Wood DL, Valdez RB, Hayashi T, Shen A. Health of homeless children and housed, poor children. Pediatrics 1990; 86:858–866.
- O’Donnell L, Stueve A, Myint-U A. Parenting and violence toward self, partners, and others among inner-city young adults. Am J Public Health 2009; 99:2255–2260.
- Leventhal T, Brooks-Gunn J. The neighborhoods they live in: the effects of neighborhood residence on child and adolescent outcomes. Psychol Bull 2000; 126:309–337.
- Shankardass K, McConnell R, Jerrett M, Milam J, Richardson J, Berhane K. Parental stress increases the effect of traffic-related air pollution on childhood asthma incidence. Proc Natl Acad Sci U S A 2009; 106:12406–12411.
- Wright RJ, Mitchell H, Visness CM, et al. Community violence and asthma morbidity: the Inner-City Asthma Study. Am J Public Health 2004; 94:625–632.
- George M, Freedman TG, Norfleet AL, Feldman HI, Apter AJ. Qualitative research-enhanced understanding of patients’ beliefs: results of focus groups with low-income, urban, African American adults with asthma. J Allergy Clin Immunol 2003; 111:967–973.
- Laster N, Holsey CN, Shendell DG, Mccarty FA, Celano M. Barriers to asthma management among urban families: caregiver and child perspectives. J Asthma 2009; 46:731–739.
- Lieu TA, Finkelstein JA, Lozano P, et al. Cultural competence policies and other predictors of asthma care quality for Medicaid-insured children. Pediatrics 2004; 114:e102–e110.
- DiMatteo MR. Variations in patients’ adherence to medical recommendations: a quantitative review of 50 years of research. Med Care 2004; 42:200–209.
- Bender B, Wamboldt FS, O’Connor SL, et al. Measurement of children’s asthma medication adherence by self report, mother report, canister weight, and Doser CT. Ann Allergy Asthma Immunol 2000; 85:416–421.
- Williams LK, Pladevall M, Xi H, et al. Relationship between adherence to inhaled corticosteroids and poor outcomes among adults with asthma. J Allergy Clin Immunol 2004; 114:1288–1293.
- Apter AJ, Boston RC, George M, et al. Modifiable barriers to adherence to inhaled steroids among adults with asthma: it’s not just black and white. J Allergy Clin Immunol 2003; 111:1219–1226.
- Apter AJ, Reisine ST, Affleck G, Barrows E, ZuWallack RL. Adherence with twice-daily dosing of inhaled steroids. Socioeconomic and health-belief differences. Am J Respir Crit Care Med 1998; 157:1810–1817.
- Williams LK, Joseph CL, Peterson EL, et al. Race-ethnicity, crime, and other factors associated with adherence to inhaled corticosteroids. J Allergy Clin Immunol 2007; 119:168–175.
- O’Malley AS, Sheppard VB, Schwartz M, Mandelblatt J. The role of trust in use of preventive services among low-income African-American women. Prev Med 2004; 38:777–785.
- Okelo SO, Wu AW, Merriman B, Krishnan JA, Diette GB. Are physician estimates of asthma severity less accurate in black than in white patients? J Gen Intern Med 2007; 22:976–981.
- Kinney PL, Aggarwal M, Northridge ME, Janssen NA, Shepard P. Airborne concentrations of PM(2.5) and diesel exhaust particles on Harlem sidewalks: a community-based pilot study. Environ Health Perspect 2000; 108:213–218.
- O’Neill MS, Jerrett M, Kawachi I, et al; Workshop on Air Pollution and Socioeconomic Conditions. Health, wealth, and air pollution: advancing theory and methods. Environ Health Perspect 2003; 111:1861–1870.
- Schwartz J, Slater D, Larson TV, Pierson WE, Koenig JQ. Particulate air pollution and hospital emergency room visits for asthma in Seattle. Am Rev Respir Dis 1993; 147:826–831.
- Norris G, YoungPong SN, Koenig JQ, Larson TV, Sheppard L, Stout JW. An association between fine particles and asthma emergency department visits for children in Seattle. Environ Health Perspect 1999; 107:489–493.
- Yu O, Sheppard L, Lumley T, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptoms of asthma in Seattle-area children enrolled in the CAMP study. Environ Health Perspect 2000; 108:1209–1214.
- Slaughter JC, Lumley T, Sheppard L, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptom severity and medication use in children with asthma. Ann Allergy Asthma Immunol 2003; 91:346–353.
- Koenig JQ, Jansen K, Mar TF, et al. Measurement of offline exhaled nitric oxide in a study of community exposure to air pollution. Environ Health Perspect 2003; 111:1625–1629.
- Centers for Disease Control and Prevention (CDC). Cigarette smoking among adults and trends in smoking cessation—United States, 2008. MMWR Morb Mortal Wkly Rep 2009; 58:1227–1232.
- Delva J, Tellez M, Finlayson TL, et al. Cigarette smoking among low-income African Americans: a serious public health problem. Am J Prev Med 2005; 29:218–220.
- McLeish AC, Zvolensky MJ. Asthma and cigarette smoking: a review of the empirical literature. J Asthma 2010; 47:345–361.
- Chaudhuri R, Livingston E, McMahon AD, Thomson L, Borland W, Thomson NC. Cigarette smoking impairs the therapeutic response to oral corticosteroids in chronic asthma. Am J Respir Crit Care Med 2003; 168:1308–1311.
- Wilson SE, Kahn RS, Khoury J, Lanphear BP. Racial differences in exposure to environmental tobacco smoke among children. Environ Health Perspect 2005; 113:362–367.
- Huss K, Rand CS, Butz AM, et al. Home environmental risk factors in urban minority asthmatic children. Ann Allergy 1994; 72:173–177.
- Samir S, Colin Y, Thomas S. Impact of environmental tobacco smoke on children admitted with status asthmaticus in the pediatric intensive care unit. Pediatr Pulmonol 2010. [Epub ahead of print]
- Lannerö E, Wickman M, Pershagen G, Nordvall L. Maternal smoking during pregnancy increases the risk of recurrent wheezing during the first years of life (BAMSE). Respir Res 2006; 7:3.
- Hedman L, Bjerg A, Sundberg S, Forsberg B, Rönmark E. Both environmental tobacco smoke and personal smoking is related to asthma and wheeze in teenagers. Thorax 2011; 66:20–25.
- Zhang G, Hayden CM, Khoo SK, et al. Beta2-adrenoceptor polymorphisms and asthma phenotypes: interactions with passive smoking. Eur Respir J 2007; 30:48–55.
- Rosenbaum E, Friedman S. The Housing Divide: How Generations of Immigrants Fare in New York’s Housing Market. New York, NY: New York University Press; 2007.
- Rosenbaum E. Racial/ethnic differences in asthma prevalence: the role of housing and neighborhood environments. J Health Soc Behav 2008; 49:131–145.
- Rauh VA, Chew GR, Garfinkel RS. Deteriorated housing contributes to high cockroach allergen levels in inner-city households. Environ Health Perspect 2002; 110( suppl 2):323–327.
- Eggleston PA, Rosenstreich D, Lynn H, et al. Relationship of indoor allergen exposure to skin test sensitivity in inner-city children with asthma. J Allergy Clin Immunol 1998; 102:563–570.
- Togias A, Horowitz E, Joyner D, Guydon L, Malveaux F. Evaluating the factors that relate to asthma severity in adolescents. Int Arch Allergy Immunol 1997; 113:87–95.
- Alp H, Yu BH, Grant EN, Rao V, Moy JN. Cockroach allergy appears early in life in inner-city children with recurrent wheezing. Ann Allergy Asthma Immunol 2001; 86:51–54.
- Miller RL, Chew GL, Bell CA, et al. Prenatal exposure, maternal sensitization, and sensitization in utero to indoor allergens in an inner-city cohort. Am J Respir Crit Care Med 2001; 164:995–1001.
- Morgan WJ, Crain EF, Gruchalla RS, et al; Inner-City Asthma Study Group. Results of a home-based environmental intervention among urban children with asthma. N Engl J Med 2004; 351:1068–1080.
- Centers for Disease Control and Prevention (CDC). Overweight and Obesity. US Obesity Trends. http://templatelab.com/us-obesity-trends/. Accessed February 1, 2012.
- Beuther DA, Sutherland ER. Overweight, obesity, and incident asthma: a meta-analysis of prospective epidemiologic studies. Am J Respir Crit Care Med 2007; 175:661–666.
- Visness CM, London SJ, Daniels JL, et al. Association of childhood obesity with atopic and nonatopic asthma: results from the National Health and Nutrition Examination Survey 1999–2006. J Asthma 2010; 47:822–829.
- Camargo CA, Sutherland ER, Bailey W, et al. Effect of increased body mass index on asthma risk, impairment and response to asthma controller therapy in African Americans. Curr Med Res Opin 2010; 26:1629–1635.
- Hakala K, Stenius-Aarniala B, Sovijärvi A. Effects of weight loss on peak flow variability, airways obstruction, and lung volumes in obese patients with asthma. Chest 2000; 118:1315–1321.
- Stenius-Aarniala B, Poussa T, Kvarnström J, Grönlund EL, Ylikahri M, Mustajoki P. Immediate and long term effects of weight reduction in obese people with asthma: randomised controlled study. BMJ 2000; 320:827–832.
- Dixon AE, Holguin F, Sood A, et al; American Thoracic Society Ad Hoc Subcommittee on Obesity and Lung Disease. An official American Thoracic Society Workshop report: obesity and asthma. Proc Am Thorac Soc 2010; 7:325–335.
- Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general US population. Am J Respir Crit Care Med 1999; 159:179–187.
- Harik-Khan RI, Muller DC, Wise RA. Racial difference in lung function in African-American and white children: effect of anthropometric, socioeconomic, nutritional, and environmental factors. Am J Epidemiol 2004; 160:893–900.
- Akinbami LJ, Moorman JE, Liu X. Asthma prevalence, health care use, and mortality: United States, 2005–2009. Natl Health Stat Report 2011; 32:1–14.
- National Institutes of Health. National Heart, Lung, and Blood Institute. Data Fact Sheet. Asthma statistics. January 1999. http://www.nhlbi.nih.gov/health/prof/lung/asthma/asthstat.pdf. Accessed February 1, 2012.
- Lang DM, Polansky M, Sherman MS. Hospitalizations for asthma in an urban population: 1995–1999. Ann Allergy Asthma Immunol 2009; 103:128–133.
- Duffy DL, Martin NG, Battistutta D, Hopper JL, Mathews JD. Genetics of asthma and hay fever in Australian twins. Am Rev Respir Dis 1990; 142:1351–1358.
- Koeppen-Schomerus G, Stevenson J, Plomin R. Genes and environment in asthma: a study of 4 year old twins. Arch Dis Child 2001; 85:398–400.
- Meng JF, Rosenwasser LJ. Unraveling the genetic basis of asthma and allergic diseases. Allergy Asthma Immunol Res 2010; 2:215–227.
- Stevenson LA, Gergen PJ, Hoover DR, Rosenstreich D, Mannino DM, Matte TD. Sociodemographic correlates of indoor allergen sensitivity among United States children. J Allergy Clin Immunol 2001; 108:747–752.
- Rosenstreich DL, Eggleston P, Kattan M, et al. The role of cockroach allergy and exposure to cockroach allergen in causing morbidity among inner-city children with asthma. N Engl J Med 1997; 336:1356–1363.
- Kobilka BK, Dixon RA, Frielle T, et al. cDNA for the human beta 2-adrenergic receptor: a protein with multiple membrane-spanning domains and encoded by a gene whose chromosomal location is shared with that of the receptor for platelet-derived growth factor. Proc Natl Acad Sci U S A 1987; 84:46–50.
- Maxwell TJ, Ameyaw MM, Pritchard S, et al. Beta-2 adrenergic receptor genotypes and haplotypes in different ethnic groups. Int J Mol Med 2005; 16:573–580.
- Israel E, Chinchilli VM, Ford JG, et al; National Heart, Lung, and Blood Institute’s Asthma Clinical Research Network. Use of regularly scheduled albuterol treatment in asthma: genotype-stratified, randomised, placebo-controlled cross-over trial. Lancet 2004; 364:1505–1512.
- Wells K, Pladevall M, Peterson EL, et al. Race-ethnic differences in factors associated with inhaled steroid adherence among adults with asthma. Am J Respir Crit Care Med 2008; 178:1194–1201.
- Carroll CL, Stoltz P, Schramm CM, Zucker AR. Beta2-adrenergic receptor polymorphisms affect response to treatment in children with severe asthma exacerbations. Chest 2009; 135:1186–1192.
- Bleecker ER, Nelson HS, Kraft M, et al. Beta2-receptor polymorphisms in patients receiving salmeterol with or without fluticasone propionate. Am J Respir Crit Care Med 2010; 181:676–687.
- Finkelstein Y, Bournissen FG, Hutson JR, Shannon M. Polymorphism of the ADRB2 gene and response to inhaled beta-agonists in children with asthma: a meta-analysis. J Asthma 2009; 46:900–905.
- Baldini M, Lohman IC, Halonen M, Erickson RP, Holt PG, Martinez FD. A polymorphism in the 5’ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am J Respir Cell Mol Biol 1999; 20:976–983.
- Kedda MA, Lose F, Duffy D, Bell E, Thompson PJ, Upham J. The CD14 C-159T polymorphism is not associated with asthma or asthma severity in an Australian adult population. Thorax 2005; 60:211–214.
- Zambelli-Weiner A, Ehrlich E, Stockton ML, et al. Evaluation of the CD14/-260 polymorphism and house dust endotoxin exposure in the Barbados Asthma Genetics Study. J Allergy Clin Immunol 2005; 115:1203–1209.
- Perzanowski MS, Miller RL, Thorne PS, et al. Endotoxin in inner-city homes: associations with wheeze and eczema in early childhood. J Allergy Clin Immunol 2006; 117:1082–1089.
- Williams LK, Oliver J, Peterson EL, et al. Gene-environment interactions between CD14 C-260T and endotoxin exposure on Foxp3+ and Foxp3− CD4+ lymphocyte numbers and total serum IgE levels in early childhood. Ann Allergy Asthma Immunol 2008; 100:128–136.
- Barnes KC. Ancestry, ancestry-informative markers, asthma, and the quest for personalized medicine. J Allergy Clin Immunol 2010; 126:1139–1140.
- Vergara C, Caraballo L, Mercado D, et al. African ancestry is associated with risk of asthma and high total serum IgE in a population from the Caribbean Coast of Colombia. Hum Genet 2009; 125:565–579.
- Gould W, Peterson EL, Karungi G, et al. Factors predicting inhaled corticosteroid responsiveness in African American patients with asthma. J Allergy Clin Immunol 2010; 126:1131–1138.
- Lang DM, Polansky M. Patterns of asthma mortality in Philadelphia from 1969 to 1991. N Engl J Med 1994; 331:1542–1546.
- Schwartz J, Gold D, Dockery DW, Weiss ST, Speizer FE. Predictors of asthma and persistent wheeze in a national sample of children in the United States. Association with social class, perinatal events, and race. Am Rev Respir Dis 1990; 142:555–562.
- National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma. Summary Report 2007. J Allergy Clin Immunol 2007; 120( suppl 5):S94–S138.
- Newacheck PW, Hughes DC, Stoddard JJ. Children’s access to primary care: differences by race, income, and insurance status. Pediatrics 1996; 97:26–32.
- Halfon N, Newacheck PW. Childhood asthma and poverty: differential impacts and utilization of health services. Pediatrics 1993; 91:56–61.
- Burt CW, Knapp DE. Ambulatory care visits for asthma: United States, 1993–94. Adv Data 1996; 277:1.
- Lieu TA, Lozano P, Finkelstein JA, et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics 2002; 109:857–865.
- Zoratti EM, Havstad S, Rodriguez J, Robens-Paradise Y, Lafata JE, McCarthy B. Health service use by African Americans and Caucasians with asthma in a managed care setting. Am J Respir Crit Care Med 1998; 158:371–377.
- Wisnivesky JP, Leventhal H, Halm EA. Predictors of asthma-related health care utilization and quality of life among inner-city patients with asthma. J Allergy Clin Immunol 2005; 116:636–642.
- Halm EA, Wisnivesky JP, Leventhal H. Quality and access to care among a cohort of inner-city adults with asthma: who gets guideline concordant care? Chest 2005; 128:1943–1950.
- Krishnan JA, Diette GB, Skinner EA, Clark BD, Steinwachs D, Wu AW. Race and sex differences in consistency of care with national asthma guidelines in managed care organizations. Arch Intern Med 2001; 161:1660–1668.
- Nelson A. Unequal treatment: confronting racial and ethnic disparities in health care. J Natl Med Assoc 2002; 94:666–668.
- Boudreaux ED, Emond SD, Clark S, Camargo CA; Multicenter Airway Research Collaboration Investigators. Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics 2003; 111:e615–e621.
- Trochtenberg DS, BeLue R, Piphus S, Washington N. Differing reports of asthma symptoms in African Americans and Caucasians. J Asthma 2008; 45:165–170.
- Hardie GE, Janson S, Gold WM, Carrieri-Kohlman V, Boushey HA. Ethnic differences: word descriptors used by African-American and white asthma patients during induced bronchoconstriction. Chest 2000; 117:935–943.
- Clark NM, Levison MJ, Evans D, Wasilewski Y, Feldman CH, Mellins RB. Communication within low income families and the management of asthma. Patient Educ Couns 1990; 15:191–210.
- Rhee H, Belyea MJ, Brasch J. Family support and asthma outcomes in adolescents: barriers to adherence as a mediator. J Adolesc Health 2010; 47:472–478.
- Quinn K, Kaufman JS, Siddiqi A, Yeatts KB. Parent perceptions of neighborhood stressors are associated with general health and child respiratory health among low-income, urban families. J Asthma 2010; 47:281–289.
- Loerbroks A, Apfelbacher CJ, Bosch JA, Stürmer T. Depressive symptoms, social support, and risk of adult asthma in a population-based cohort study. Psychosom Med 2010; 72:309–315.
- Wood DL, Valdez RB, Hayashi T, Shen A. Health of homeless children and housed, poor children. Pediatrics 1990; 86:858–866.
- O’Donnell L, Stueve A, Myint-U A. Parenting and violence toward self, partners, and others among inner-city young adults. Am J Public Health 2009; 99:2255–2260.
- Leventhal T, Brooks-Gunn J. The neighborhoods they live in: the effects of neighborhood residence on child and adolescent outcomes. Psychol Bull 2000; 126:309–337.
- Shankardass K, McConnell R, Jerrett M, Milam J, Richardson J, Berhane K. Parental stress increases the effect of traffic-related air pollution on childhood asthma incidence. Proc Natl Acad Sci U S A 2009; 106:12406–12411.
- Wright RJ, Mitchell H, Visness CM, et al. Community violence and asthma morbidity: the Inner-City Asthma Study. Am J Public Health 2004; 94:625–632.
- George M, Freedman TG, Norfleet AL, Feldman HI, Apter AJ. Qualitative research-enhanced understanding of patients’ beliefs: results of focus groups with low-income, urban, African American adults with asthma. J Allergy Clin Immunol 2003; 111:967–973.
- Laster N, Holsey CN, Shendell DG, Mccarty FA, Celano M. Barriers to asthma management among urban families: caregiver and child perspectives. J Asthma 2009; 46:731–739.
- Lieu TA, Finkelstein JA, Lozano P, et al. Cultural competence policies and other predictors of asthma care quality for Medicaid-insured children. Pediatrics 2004; 114:e102–e110.
- DiMatteo MR. Variations in patients’ adherence to medical recommendations: a quantitative review of 50 years of research. Med Care 2004; 42:200–209.
- Bender B, Wamboldt FS, O’Connor SL, et al. Measurement of children’s asthma medication adherence by self report, mother report, canister weight, and Doser CT. Ann Allergy Asthma Immunol 2000; 85:416–421.
- Williams LK, Pladevall M, Xi H, et al. Relationship between adherence to inhaled corticosteroids and poor outcomes among adults with asthma. J Allergy Clin Immunol 2004; 114:1288–1293.
- Apter AJ, Boston RC, George M, et al. Modifiable barriers to adherence to inhaled steroids among adults with asthma: it’s not just black and white. J Allergy Clin Immunol 2003; 111:1219–1226.
- Apter AJ, Reisine ST, Affleck G, Barrows E, ZuWallack RL. Adherence with twice-daily dosing of inhaled steroids. Socioeconomic and health-belief differences. Am J Respir Crit Care Med 1998; 157:1810–1817.
- Williams LK, Joseph CL, Peterson EL, et al. Race-ethnicity, crime, and other factors associated with adherence to inhaled corticosteroids. J Allergy Clin Immunol 2007; 119:168–175.
- O’Malley AS, Sheppard VB, Schwartz M, Mandelblatt J. The role of trust in use of preventive services among low-income African-American women. Prev Med 2004; 38:777–785.
- Okelo SO, Wu AW, Merriman B, Krishnan JA, Diette GB. Are physician estimates of asthma severity less accurate in black than in white patients? J Gen Intern Med 2007; 22:976–981.
- Kinney PL, Aggarwal M, Northridge ME, Janssen NA, Shepard P. Airborne concentrations of PM(2.5) and diesel exhaust particles on Harlem sidewalks: a community-based pilot study. Environ Health Perspect 2000; 108:213–218.
- O’Neill MS, Jerrett M, Kawachi I, et al; Workshop on Air Pollution and Socioeconomic Conditions. Health, wealth, and air pollution: advancing theory and methods. Environ Health Perspect 2003; 111:1861–1870.
- Schwartz J, Slater D, Larson TV, Pierson WE, Koenig JQ. Particulate air pollution and hospital emergency room visits for asthma in Seattle. Am Rev Respir Dis 1993; 147:826–831.
- Norris G, YoungPong SN, Koenig JQ, Larson TV, Sheppard L, Stout JW. An association between fine particles and asthma emergency department visits for children in Seattle. Environ Health Perspect 1999; 107:489–493.
- Yu O, Sheppard L, Lumley T, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptoms of asthma in Seattle-area children enrolled in the CAMP study. Environ Health Perspect 2000; 108:1209–1214.
- Slaughter JC, Lumley T, Sheppard L, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptom severity and medication use in children with asthma. Ann Allergy Asthma Immunol 2003; 91:346–353.
- Koenig JQ, Jansen K, Mar TF, et al. Measurement of offline exhaled nitric oxide in a study of community exposure to air pollution. Environ Health Perspect 2003; 111:1625–1629.
- Centers for Disease Control and Prevention (CDC). Cigarette smoking among adults and trends in smoking cessation—United States, 2008. MMWR Morb Mortal Wkly Rep 2009; 58:1227–1232.
- Delva J, Tellez M, Finlayson TL, et al. Cigarette smoking among low-income African Americans: a serious public health problem. Am J Prev Med 2005; 29:218–220.
- McLeish AC, Zvolensky MJ. Asthma and cigarette smoking: a review of the empirical literature. J Asthma 2010; 47:345–361.
- Chaudhuri R, Livingston E, McMahon AD, Thomson L, Borland W, Thomson NC. Cigarette smoking impairs the therapeutic response to oral corticosteroids in chronic asthma. Am J Respir Crit Care Med 2003; 168:1308–1311.
- Wilson SE, Kahn RS, Khoury J, Lanphear BP. Racial differences in exposure to environmental tobacco smoke among children. Environ Health Perspect 2005; 113:362–367.
- Huss K, Rand CS, Butz AM, et al. Home environmental risk factors in urban minority asthmatic children. Ann Allergy 1994; 72:173–177.
- Samir S, Colin Y, Thomas S. Impact of environmental tobacco smoke on children admitted with status asthmaticus in the pediatric intensive care unit. Pediatr Pulmonol 2010. [Epub ahead of print]
- Lannerö E, Wickman M, Pershagen G, Nordvall L. Maternal smoking during pregnancy increases the risk of recurrent wheezing during the first years of life (BAMSE). Respir Res 2006; 7:3.
- Hedman L, Bjerg A, Sundberg S, Forsberg B, Rönmark E. Both environmental tobacco smoke and personal smoking is related to asthma and wheeze in teenagers. Thorax 2011; 66:20–25.
- Zhang G, Hayden CM, Khoo SK, et al. Beta2-adrenoceptor polymorphisms and asthma phenotypes: interactions with passive smoking. Eur Respir J 2007; 30:48–55.
- Rosenbaum E, Friedman S. The Housing Divide: How Generations of Immigrants Fare in New York’s Housing Market. New York, NY: New York University Press; 2007.
- Rosenbaum E. Racial/ethnic differences in asthma prevalence: the role of housing and neighborhood environments. J Health Soc Behav 2008; 49:131–145.
- Rauh VA, Chew GR, Garfinkel RS. Deteriorated housing contributes to high cockroach allergen levels in inner-city households. Environ Health Perspect 2002; 110( suppl 2):323–327.
- Eggleston PA, Rosenstreich D, Lynn H, et al. Relationship of indoor allergen exposure to skin test sensitivity in inner-city children with asthma. J Allergy Clin Immunol 1998; 102:563–570.
- Togias A, Horowitz E, Joyner D, Guydon L, Malveaux F. Evaluating the factors that relate to asthma severity in adolescents. Int Arch Allergy Immunol 1997; 113:87–95.
- Alp H, Yu BH, Grant EN, Rao V, Moy JN. Cockroach allergy appears early in life in inner-city children with recurrent wheezing. Ann Allergy Asthma Immunol 2001; 86:51–54.
- Miller RL, Chew GL, Bell CA, et al. Prenatal exposure, maternal sensitization, and sensitization in utero to indoor allergens in an inner-city cohort. Am J Respir Crit Care Med 2001; 164:995–1001.
- Morgan WJ, Crain EF, Gruchalla RS, et al; Inner-City Asthma Study Group. Results of a home-based environmental intervention among urban children with asthma. N Engl J Med 2004; 351:1068–1080.
- Centers for Disease Control and Prevention (CDC). Overweight and Obesity. US Obesity Trends. http://templatelab.com/us-obesity-trends/. Accessed February 1, 2012.
- Beuther DA, Sutherland ER. Overweight, obesity, and incident asthma: a meta-analysis of prospective epidemiologic studies. Am J Respir Crit Care Med 2007; 175:661–666.
- Visness CM, London SJ, Daniels JL, et al. Association of childhood obesity with atopic and nonatopic asthma: results from the National Health and Nutrition Examination Survey 1999–2006. J Asthma 2010; 47:822–829.
- Camargo CA, Sutherland ER, Bailey W, et al. Effect of increased body mass index on asthma risk, impairment and response to asthma controller therapy in African Americans. Curr Med Res Opin 2010; 26:1629–1635.
- Hakala K, Stenius-Aarniala B, Sovijärvi A. Effects of weight loss on peak flow variability, airways obstruction, and lung volumes in obese patients with asthma. Chest 2000; 118:1315–1321.
- Stenius-Aarniala B, Poussa T, Kvarnström J, Grönlund EL, Ylikahri M, Mustajoki P. Immediate and long term effects of weight reduction in obese people with asthma: randomised controlled study. BMJ 2000; 320:827–832.
- Dixon AE, Holguin F, Sood A, et al; American Thoracic Society Ad Hoc Subcommittee on Obesity and Lung Disease. An official American Thoracic Society Workshop report: obesity and asthma. Proc Am Thorac Soc 2010; 7:325–335.
- Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general US population. Am J Respir Crit Care Med 1999; 159:179–187.
- Harik-Khan RI, Muller DC, Wise RA. Racial difference in lung function in African-American and white children: effect of anthropometric, socioeconomic, nutritional, and environmental factors. Am J Epidemiol 2004; 160:893–900.
KEY POINTS
- To better identify those at risk, researchers are looking at genetic markers such as polymorphisms in ADRB2 and CD14.
- Exposure to tobacco smoke and to cockroach allergen contribute to higher rates of asthma prevalence and morbidity.
- African Americans are more likely to receive suboptimal care, in particular to be misdiagnosed with other conditions, to not receive inhaled corticosteroids, and to not receive proper follow-up.
- Better physician-patient communication is one of the keys to improving this problem.
Peer-reviewers for 2011
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in the year ending December 31, 2011. Reviewing papers for scientific journals is an arduous task and involves considerable time and effort. We are grateful to these reviewers for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in the year ending December 31, 2011. Reviewing papers for scientific journals is an arduous task and involves considerable time and effort. We are grateful to these reviewers for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in the year ending December 31, 2011. Reviewing papers for scientific journals is an arduous task and involves considerable time and effort. We are grateful to these reviewers for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
A 26-year-old woman with a lump in her chest
A 26-year-old Filipino woman presented for evaluation of sternal pain associated with a palpable mass that she had noticed 8 months earlier. She had no history of significant medical illness. She had recently immigrated to El Paso, TX, from the Philippines.
Q: Which is the most likely diagnosis?
- Plasmacytoma
- Chondrosarcoma
- Extrapulmonary tuberculosis
- Lymphoma
- Metastatic breast cancer
EXTRAPULMONARY TUBERCULOSIS
Extrapulmonary tuberculosis accounts for about 20% of all cases of tuberculosis.1
Risk factors for tuberculosis include advanced age, immunosuppression (eg, as occurs in HIV infection), organ transplantation, and therapy with a tumor necrosis factor alpha inhibitor.1–4 Risk factors unique to extrapulmonary tuberculosis infection include female sex and non-Hispanic black ethnicity.2 Because of the high prevalence of tuberculosis in certain parts of the world, obtaining a travel or residence history is an essential part of the clinical evaluation.
Skeletal tuberculosis accounts for 11% to 27% of extrapulmonary cases and, by extrapolation, 2% to 5% of all cases of tuberculosis.1–3 Although the spine is the site most commonly involved, any bone may be affected. When the chest wall is involved, the most common locations are the margin of the sternum and along rib shafts.5
Most patients present with pain and swelling. The presence of constitutional symptoms is variable, occurring in about one-third of patients.6 Classically, the lesion of tuberculous osteomyelitis is described as a “cold abscess,” as it is characterized by swelling and erythema with little or no warmth. Spontaneous drainage and sinus tract formation may occur.5
The differential diagnosis of tuberculous osteomyelitis includes pyogenic bacterial infection, atypical bacterial infection (nocardia, meliodosis, brucellosis), fungal infection (coccidioidomycosis, histoplasmosis, blastomycosis), and metastatic and primary bone malignancies. Diagnosis requires a high index of suspicion, biopsy for histopathologic examination, acid-fast staining, and mycobacterial culture.7
Patients generally respond well to 6 months of a standard four-drug regimen for tuberculosis. Surgery is indicated for abscess drainage, debridement of infected tissue, spine stabilization, and relief of spinal cord compression.5
Our patient had complete resolution of her sternal mass with drug therapy alone.
- Peto HM, Pratt RH, Harrington TA, LoBue PA, Armstrong LR. Epidemiology of extrapulmonary tuberculosis in the United States, 1993–2006. Clin Infect Dis 2009; 49:1350–1357.
- Yang Z, Kong Y, Wilson F, et al. Identification of risk factors for extrapulmonary tuberculosis. Clin Infect Dis 2004; 38:199–205.
- Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001; 345:1098–1104.
- Alagarsamy S, Dhand S, Aung S, Wolff M, Bahrain M. Sternal tuberculosis: a rare case mimicking sarcoma and review of the literature. Infect Dis Clin Pract 2009; 17:138–143.
- Morris BS, Maheshwari M, Chalwa A. Chest wall tuberculosis: a review of CT appearances. Br J Radiol 2004; 77:449–457.
- Sandher DS, Al-Jibury M, Paton RW, Ormerod LP. Bone and joint tuberculosis: cases in Blackburn between 1988 and 2005. J Bone Joint Surg Br 2007; 89:1379–1381.
- Centers for Disease Control and Prevention (CDC). Case definitions for infectious conditions under public health surveillance. http://cdc.gov/mmwr/preview/mmwrhtml/00047449.htm. Accessed October 6, 2011.
A 26-year-old Filipino woman presented for evaluation of sternal pain associated with a palpable mass that she had noticed 8 months earlier. She had no history of significant medical illness. She had recently immigrated to El Paso, TX, from the Philippines.
Q: Which is the most likely diagnosis?
- Plasmacytoma
- Chondrosarcoma
- Extrapulmonary tuberculosis
- Lymphoma
- Metastatic breast cancer
EXTRAPULMONARY TUBERCULOSIS
Extrapulmonary tuberculosis accounts for about 20% of all cases of tuberculosis.1
Risk factors for tuberculosis include advanced age, immunosuppression (eg, as occurs in HIV infection), organ transplantation, and therapy with a tumor necrosis factor alpha inhibitor.1–4 Risk factors unique to extrapulmonary tuberculosis infection include female sex and non-Hispanic black ethnicity.2 Because of the high prevalence of tuberculosis in certain parts of the world, obtaining a travel or residence history is an essential part of the clinical evaluation.
Skeletal tuberculosis accounts for 11% to 27% of extrapulmonary cases and, by extrapolation, 2% to 5% of all cases of tuberculosis.1–3 Although the spine is the site most commonly involved, any bone may be affected. When the chest wall is involved, the most common locations are the margin of the sternum and along rib shafts.5
Most patients present with pain and swelling. The presence of constitutional symptoms is variable, occurring in about one-third of patients.6 Classically, the lesion of tuberculous osteomyelitis is described as a “cold abscess,” as it is characterized by swelling and erythema with little or no warmth. Spontaneous drainage and sinus tract formation may occur.5
The differential diagnosis of tuberculous osteomyelitis includes pyogenic bacterial infection, atypical bacterial infection (nocardia, meliodosis, brucellosis), fungal infection (coccidioidomycosis, histoplasmosis, blastomycosis), and metastatic and primary bone malignancies. Diagnosis requires a high index of suspicion, biopsy for histopathologic examination, acid-fast staining, and mycobacterial culture.7
Patients generally respond well to 6 months of a standard four-drug regimen for tuberculosis. Surgery is indicated for abscess drainage, debridement of infected tissue, spine stabilization, and relief of spinal cord compression.5
Our patient had complete resolution of her sternal mass with drug therapy alone.
A 26-year-old Filipino woman presented for evaluation of sternal pain associated with a palpable mass that she had noticed 8 months earlier. She had no history of significant medical illness. She had recently immigrated to El Paso, TX, from the Philippines.
Q: Which is the most likely diagnosis?
- Plasmacytoma
- Chondrosarcoma
- Extrapulmonary tuberculosis
- Lymphoma
- Metastatic breast cancer
EXTRAPULMONARY TUBERCULOSIS
Extrapulmonary tuberculosis accounts for about 20% of all cases of tuberculosis.1
Risk factors for tuberculosis include advanced age, immunosuppression (eg, as occurs in HIV infection), organ transplantation, and therapy with a tumor necrosis factor alpha inhibitor.1–4 Risk factors unique to extrapulmonary tuberculosis infection include female sex and non-Hispanic black ethnicity.2 Because of the high prevalence of tuberculosis in certain parts of the world, obtaining a travel or residence history is an essential part of the clinical evaluation.
Skeletal tuberculosis accounts for 11% to 27% of extrapulmonary cases and, by extrapolation, 2% to 5% of all cases of tuberculosis.1–3 Although the spine is the site most commonly involved, any bone may be affected. When the chest wall is involved, the most common locations are the margin of the sternum and along rib shafts.5
Most patients present with pain and swelling. The presence of constitutional symptoms is variable, occurring in about one-third of patients.6 Classically, the lesion of tuberculous osteomyelitis is described as a “cold abscess,” as it is characterized by swelling and erythema with little or no warmth. Spontaneous drainage and sinus tract formation may occur.5
The differential diagnosis of tuberculous osteomyelitis includes pyogenic bacterial infection, atypical bacterial infection (nocardia, meliodosis, brucellosis), fungal infection (coccidioidomycosis, histoplasmosis, blastomycosis), and metastatic and primary bone malignancies. Diagnosis requires a high index of suspicion, biopsy for histopathologic examination, acid-fast staining, and mycobacterial culture.7
Patients generally respond well to 6 months of a standard four-drug regimen for tuberculosis. Surgery is indicated for abscess drainage, debridement of infected tissue, spine stabilization, and relief of spinal cord compression.5
Our patient had complete resolution of her sternal mass with drug therapy alone.
- Peto HM, Pratt RH, Harrington TA, LoBue PA, Armstrong LR. Epidemiology of extrapulmonary tuberculosis in the United States, 1993–2006. Clin Infect Dis 2009; 49:1350–1357.
- Yang Z, Kong Y, Wilson F, et al. Identification of risk factors for extrapulmonary tuberculosis. Clin Infect Dis 2004; 38:199–205.
- Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001; 345:1098–1104.
- Alagarsamy S, Dhand S, Aung S, Wolff M, Bahrain M. Sternal tuberculosis: a rare case mimicking sarcoma and review of the literature. Infect Dis Clin Pract 2009; 17:138–143.
- Morris BS, Maheshwari M, Chalwa A. Chest wall tuberculosis: a review of CT appearances. Br J Radiol 2004; 77:449–457.
- Sandher DS, Al-Jibury M, Paton RW, Ormerod LP. Bone and joint tuberculosis: cases in Blackburn between 1988 and 2005. J Bone Joint Surg Br 2007; 89:1379–1381.
- Centers for Disease Control and Prevention (CDC). Case definitions for infectious conditions under public health surveillance. http://cdc.gov/mmwr/preview/mmwrhtml/00047449.htm. Accessed October 6, 2011.
- Peto HM, Pratt RH, Harrington TA, LoBue PA, Armstrong LR. Epidemiology of extrapulmonary tuberculosis in the United States, 1993–2006. Clin Infect Dis 2009; 49:1350–1357.
- Yang Z, Kong Y, Wilson F, et al. Identification of risk factors for extrapulmonary tuberculosis. Clin Infect Dis 2004; 38:199–205.
- Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001; 345:1098–1104.
- Alagarsamy S, Dhand S, Aung S, Wolff M, Bahrain M. Sternal tuberculosis: a rare case mimicking sarcoma and review of the literature. Infect Dis Clin Pract 2009; 17:138–143.
- Morris BS, Maheshwari M, Chalwa A. Chest wall tuberculosis: a review of CT appearances. Br J Radiol 2004; 77:449–457.
- Sandher DS, Al-Jibury M, Paton RW, Ormerod LP. Bone and joint tuberculosis: cases in Blackburn between 1988 and 2005. J Bone Joint Surg Br 2007; 89:1379–1381.
- Centers for Disease Control and Prevention (CDC). Case definitions for infectious conditions under public health surveillance. http://cdc.gov/mmwr/preview/mmwrhtml/00047449.htm. Accessed October 6, 2011.
High creatinine 6 months after renal transplant
A 42-year-old man presented with acute renal failure with a serum creatinine of 6.07 mg/dL (baseline 2.0 mg/dL) 6 months after receiving a kidney transplant from a deceased donor. He was asymptomatic, had no previous symptoms of transplant rejection, and was compliant with his immunosuppressive regimen. The physical examination and the rest of the laboratory workup were normal.
Q: Which is the most likely diagnosis?
- A lymphocele
- A hematoma
- A urinoma
- A perirenal abscess
- A simple renal cyst
A: A lymphocele is the most likely diagnosis. A lymphocele— a collection of lymph without an epithelial lining—develops in as many as 20% of kidney transplant recipients.1 Many causative factors have been proposed, including leakage of lymph from recipient lymphatic channels,2 use of diuretics,2 obesity,3 kidney biopsy,4 acute rejection,3 and the use of sirolimus5 (Rapamune) and high-dose corticosteroids.6 Some believe that lymphoceles may also arise from severed lymphatic vessels of the donor-kidney allograft.7
Ultrasonography can usually distinguish a lymphocele from other fluid collections on the basis of fluid appearance, shape, and position. In most cases, the lymphocele is adjacent to the lower pole and medial to the allograft, and appears anechoic on ultrasonography, with a thin, distinct wall. The typical features on analysis of aspirated fluid—ie, a creatinine level approximately the same as in the serum, a low protein value, and a high lymphocyte count compared with serum values—confirm the diagnosis of lymphocele.
A hematoma can occur in any location and have a heterogeneous appearance, as it contains both clotted (echogenic) and unclotted (anechoic) blood. They are usually seen within the first 1 to 2 weeks after surgery and may also develop after trauma or renal biopsy.
A urinoma is a collection of urine outside the bladder, resulting from a ureteral leak. They are predominantly anechoic, with an often indistinct wall. If there is a clinical suspicion, the diagnosis can be confirmed on aspiration by a high creatinine level in the fluid compared with the serum value.
A perirenal abscess commonly presents with pain, fever, and a complex fluid collection on ultrasonography, sometimes with an air fluid level. Aspiration of purulent fluid confirms the diagnosis.
A simple renal cyst appears within or protruding from the renal parenchyma as a spherical or eggshaped fluid-filled sac with an anechoic lumen and no measurable wall thickness.
SYMPTOMS AND MANAGEMENT
Lymphoceles are mostly inconsequential but can cause renal failure by compressing the ureter, renal vessels, or renal allograft. Other manifestations may include pain and swelling at the kidney allograft site, wound drainage, unilateral lower-extremity edema, deep vein thrombosis due to compression of iliac veins,8 urinary urgency or frequency due to extrinsic bladder compression, and urinary retention.9
If the lymphocele is clinically significant, percutaneous drainage guided by ultrasonography is recommended as the initial curative procedure.10 Sclerotherapy with different chemical agents is effective, but success depends on the size of the lymphocele cavity.11
If these conservative therapies fail, lymphocele unroofing into the peritoneal cavity is needed. This is accomplished by laparoscopy12,13 or open surgery. Although laparoscopic drainage is considered the procedure of choice, open surgery may be required for multiloculated lymphoceles and those adjacent to vital structures.14,15
Kidneys are the most commonly transplanted solid organs. Every year, about 16,000 kidney transplantations are performed in the United States. It is common for the primary care physician to initially see these patients in cases of associated complications. Internists must be aware of the common causes of acute renal failure in this population, eg, acute rejection, drug toxicity, and obstruction. Lymphoceles are an important cause of renal failure due to obstruction. Early recognition and appropriate treatment of this complication can improve the outcome of the allograft.
- O’neill WC, Baumgarten DA. Ultrasonography in renal transplantation. Am J Kidney Dis 2002; 39:663–678.
- Braun WE, Banowsky LH, Straffon RA, et al. Lymphocytes associated with renal transplantation. Report of 15 cases and review of the literature. Am J Med 1974; 57:714–729.
- Goel M, Flechner SM, Zhou L, et al. The influence of various maintenance immunosuppressive drugs on lymphocele formation and treatment after kidney transplantation. J Urol 2004; 171:1788–1792.
- Mundy AR, Podesta ML, Bewick M, Rudge CJ, Ellis FG. The urological complications of 1000 renal transplants. Br J Urol 1981; 53:397–402.
- Giessing M, Fischer TJ, Deger S, et al. Increased frequency of lymphoceles under treatment with sirolimus following renal transplantation: a single center experience. Transplant Proc 2002; 34:1815–1816.
- Amante AJ, Kahan BD. Technical complications of renal transplantation. Surg Clin North Am 1994; 74:1117–1131.
- Saidi RF, Wertheim JA, Ko DS, et al. Impact of donor kidney recovery method on lymphatic complications in kidney transplantation. Transplant Proc 2008; 40:1054–1055.
- Iwan-Zietek I, Zietek Z, Sulikowski T, et al. Minimally invasive methods for the treatment of lymphocele after kidney transplantation. Transplant Proc 2009; 41:3073–3076.
- Hwang EC, Kang TW, Koh YS, et al. Post-transplant lymphocele: an unusual cause of acute urinary retention mimicking urethral injury. Int J Urol 2006; 13:468–470.
- Zietek Z, Sulikowski T, Tejchman K, et al. Lymphocele after kidney transplantation. Transplant Proc 2007; 39:2744–2747.
- Mahrer A, Ramchandani P, Trerotola SO, Shlansky-Goldberg RD, Itkin M. Sclerotherapy in the management of postoperative lymphocele. J Vasc Interv Radiol 2010; 21:1050–1053.
- Risaliti A, Corno V, Donini A, et al. Laparoscopic treatment of symptomatic lymphoceles after kidney transplantation. Surg Endosc 2000; 14:293–295.
- Ostrowski M, Lubikowski J, Kowalczyk M, Power J. Laparoscopic lymphocele drainage after renal transplantation. Ann Transplant 2000; 5:25–27.
- Fuller TF, Kang SM, Hirose R, Feng S, Stock PG, Freise CE. Management of lymphoceles after renal transplantation: laparoscopic versus open drainage. J Urol 2003; 169:2022–2025.
- Hsu TH, Gill IS, Grune MT, et al. Laparoscopic lymphocelectomy: a multi-institutional analysis. J Urol 2000; 163:1096–1098.
A 42-year-old man presented with acute renal failure with a serum creatinine of 6.07 mg/dL (baseline 2.0 mg/dL) 6 months after receiving a kidney transplant from a deceased donor. He was asymptomatic, had no previous symptoms of transplant rejection, and was compliant with his immunosuppressive regimen. The physical examination and the rest of the laboratory workup were normal.
Q: Which is the most likely diagnosis?
- A lymphocele
- A hematoma
- A urinoma
- A perirenal abscess
- A simple renal cyst
A: A lymphocele is the most likely diagnosis. A lymphocele— a collection of lymph without an epithelial lining—develops in as many as 20% of kidney transplant recipients.1 Many causative factors have been proposed, including leakage of lymph from recipient lymphatic channels,2 use of diuretics,2 obesity,3 kidney biopsy,4 acute rejection,3 and the use of sirolimus5 (Rapamune) and high-dose corticosteroids.6 Some believe that lymphoceles may also arise from severed lymphatic vessels of the donor-kidney allograft.7
Ultrasonography can usually distinguish a lymphocele from other fluid collections on the basis of fluid appearance, shape, and position. In most cases, the lymphocele is adjacent to the lower pole and medial to the allograft, and appears anechoic on ultrasonography, with a thin, distinct wall. The typical features on analysis of aspirated fluid—ie, a creatinine level approximately the same as in the serum, a low protein value, and a high lymphocyte count compared with serum values—confirm the diagnosis of lymphocele.
A hematoma can occur in any location and have a heterogeneous appearance, as it contains both clotted (echogenic) and unclotted (anechoic) blood. They are usually seen within the first 1 to 2 weeks after surgery and may also develop after trauma or renal biopsy.
A urinoma is a collection of urine outside the bladder, resulting from a ureteral leak. They are predominantly anechoic, with an often indistinct wall. If there is a clinical suspicion, the diagnosis can be confirmed on aspiration by a high creatinine level in the fluid compared with the serum value.
A perirenal abscess commonly presents with pain, fever, and a complex fluid collection on ultrasonography, sometimes with an air fluid level. Aspiration of purulent fluid confirms the diagnosis.
A simple renal cyst appears within or protruding from the renal parenchyma as a spherical or eggshaped fluid-filled sac with an anechoic lumen and no measurable wall thickness.
SYMPTOMS AND MANAGEMENT
Lymphoceles are mostly inconsequential but can cause renal failure by compressing the ureter, renal vessels, or renal allograft. Other manifestations may include pain and swelling at the kidney allograft site, wound drainage, unilateral lower-extremity edema, deep vein thrombosis due to compression of iliac veins,8 urinary urgency or frequency due to extrinsic bladder compression, and urinary retention.9
If the lymphocele is clinically significant, percutaneous drainage guided by ultrasonography is recommended as the initial curative procedure.10 Sclerotherapy with different chemical agents is effective, but success depends on the size of the lymphocele cavity.11
If these conservative therapies fail, lymphocele unroofing into the peritoneal cavity is needed. This is accomplished by laparoscopy12,13 or open surgery. Although laparoscopic drainage is considered the procedure of choice, open surgery may be required for multiloculated lymphoceles and those adjacent to vital structures.14,15
Kidneys are the most commonly transplanted solid organs. Every year, about 16,000 kidney transplantations are performed in the United States. It is common for the primary care physician to initially see these patients in cases of associated complications. Internists must be aware of the common causes of acute renal failure in this population, eg, acute rejection, drug toxicity, and obstruction. Lymphoceles are an important cause of renal failure due to obstruction. Early recognition and appropriate treatment of this complication can improve the outcome of the allograft.
A 42-year-old man presented with acute renal failure with a serum creatinine of 6.07 mg/dL (baseline 2.0 mg/dL) 6 months after receiving a kidney transplant from a deceased donor. He was asymptomatic, had no previous symptoms of transplant rejection, and was compliant with his immunosuppressive regimen. The physical examination and the rest of the laboratory workup were normal.
Q: Which is the most likely diagnosis?
- A lymphocele
- A hematoma
- A urinoma
- A perirenal abscess
- A simple renal cyst
A: A lymphocele is the most likely diagnosis. A lymphocele— a collection of lymph without an epithelial lining—develops in as many as 20% of kidney transplant recipients.1 Many causative factors have been proposed, including leakage of lymph from recipient lymphatic channels,2 use of diuretics,2 obesity,3 kidney biopsy,4 acute rejection,3 and the use of sirolimus5 (Rapamune) and high-dose corticosteroids.6 Some believe that lymphoceles may also arise from severed lymphatic vessels of the donor-kidney allograft.7
Ultrasonography can usually distinguish a lymphocele from other fluid collections on the basis of fluid appearance, shape, and position. In most cases, the lymphocele is adjacent to the lower pole and medial to the allograft, and appears anechoic on ultrasonography, with a thin, distinct wall. The typical features on analysis of aspirated fluid—ie, a creatinine level approximately the same as in the serum, a low protein value, and a high lymphocyte count compared with serum values—confirm the diagnosis of lymphocele.
A hematoma can occur in any location and have a heterogeneous appearance, as it contains both clotted (echogenic) and unclotted (anechoic) blood. They are usually seen within the first 1 to 2 weeks after surgery and may also develop after trauma or renal biopsy.
A urinoma is a collection of urine outside the bladder, resulting from a ureteral leak. They are predominantly anechoic, with an often indistinct wall. If there is a clinical suspicion, the diagnosis can be confirmed on aspiration by a high creatinine level in the fluid compared with the serum value.
A perirenal abscess commonly presents with pain, fever, and a complex fluid collection on ultrasonography, sometimes with an air fluid level. Aspiration of purulent fluid confirms the diagnosis.
A simple renal cyst appears within or protruding from the renal parenchyma as a spherical or eggshaped fluid-filled sac with an anechoic lumen and no measurable wall thickness.
SYMPTOMS AND MANAGEMENT
Lymphoceles are mostly inconsequential but can cause renal failure by compressing the ureter, renal vessels, or renal allograft. Other manifestations may include pain and swelling at the kidney allograft site, wound drainage, unilateral lower-extremity edema, deep vein thrombosis due to compression of iliac veins,8 urinary urgency or frequency due to extrinsic bladder compression, and urinary retention.9
If the lymphocele is clinically significant, percutaneous drainage guided by ultrasonography is recommended as the initial curative procedure.10 Sclerotherapy with different chemical agents is effective, but success depends on the size of the lymphocele cavity.11
If these conservative therapies fail, lymphocele unroofing into the peritoneal cavity is needed. This is accomplished by laparoscopy12,13 or open surgery. Although laparoscopic drainage is considered the procedure of choice, open surgery may be required for multiloculated lymphoceles and those adjacent to vital structures.14,15
Kidneys are the most commonly transplanted solid organs. Every year, about 16,000 kidney transplantations are performed in the United States. It is common for the primary care physician to initially see these patients in cases of associated complications. Internists must be aware of the common causes of acute renal failure in this population, eg, acute rejection, drug toxicity, and obstruction. Lymphoceles are an important cause of renal failure due to obstruction. Early recognition and appropriate treatment of this complication can improve the outcome of the allograft.
- O’neill WC, Baumgarten DA. Ultrasonography in renal transplantation. Am J Kidney Dis 2002; 39:663–678.
- Braun WE, Banowsky LH, Straffon RA, et al. Lymphocytes associated with renal transplantation. Report of 15 cases and review of the literature. Am J Med 1974; 57:714–729.
- Goel M, Flechner SM, Zhou L, et al. The influence of various maintenance immunosuppressive drugs on lymphocele formation and treatment after kidney transplantation. J Urol 2004; 171:1788–1792.
- Mundy AR, Podesta ML, Bewick M, Rudge CJ, Ellis FG. The urological complications of 1000 renal transplants. Br J Urol 1981; 53:397–402.
- Giessing M, Fischer TJ, Deger S, et al. Increased frequency of lymphoceles under treatment with sirolimus following renal transplantation: a single center experience. Transplant Proc 2002; 34:1815–1816.
- Amante AJ, Kahan BD. Technical complications of renal transplantation. Surg Clin North Am 1994; 74:1117–1131.
- Saidi RF, Wertheim JA, Ko DS, et al. Impact of donor kidney recovery method on lymphatic complications in kidney transplantation. Transplant Proc 2008; 40:1054–1055.
- Iwan-Zietek I, Zietek Z, Sulikowski T, et al. Minimally invasive methods for the treatment of lymphocele after kidney transplantation. Transplant Proc 2009; 41:3073–3076.
- Hwang EC, Kang TW, Koh YS, et al. Post-transplant lymphocele: an unusual cause of acute urinary retention mimicking urethral injury. Int J Urol 2006; 13:468–470.
- Zietek Z, Sulikowski T, Tejchman K, et al. Lymphocele after kidney transplantation. Transplant Proc 2007; 39:2744–2747.
- Mahrer A, Ramchandani P, Trerotola SO, Shlansky-Goldberg RD, Itkin M. Sclerotherapy in the management of postoperative lymphocele. J Vasc Interv Radiol 2010; 21:1050–1053.
- Risaliti A, Corno V, Donini A, et al. Laparoscopic treatment of symptomatic lymphoceles after kidney transplantation. Surg Endosc 2000; 14:293–295.
- Ostrowski M, Lubikowski J, Kowalczyk M, Power J. Laparoscopic lymphocele drainage after renal transplantation. Ann Transplant 2000; 5:25–27.
- Fuller TF, Kang SM, Hirose R, Feng S, Stock PG, Freise CE. Management of lymphoceles after renal transplantation: laparoscopic versus open drainage. J Urol 2003; 169:2022–2025.
- Hsu TH, Gill IS, Grune MT, et al. Laparoscopic lymphocelectomy: a multi-institutional analysis. J Urol 2000; 163:1096–1098.
- O’neill WC, Baumgarten DA. Ultrasonography in renal transplantation. Am J Kidney Dis 2002; 39:663–678.
- Braun WE, Banowsky LH, Straffon RA, et al. Lymphocytes associated with renal transplantation. Report of 15 cases and review of the literature. Am J Med 1974; 57:714–729.
- Goel M, Flechner SM, Zhou L, et al. The influence of various maintenance immunosuppressive drugs on lymphocele formation and treatment after kidney transplantation. J Urol 2004; 171:1788–1792.
- Mundy AR, Podesta ML, Bewick M, Rudge CJ, Ellis FG. The urological complications of 1000 renal transplants. Br J Urol 1981; 53:397–402.
- Giessing M, Fischer TJ, Deger S, et al. Increased frequency of lymphoceles under treatment with sirolimus following renal transplantation: a single center experience. Transplant Proc 2002; 34:1815–1816.
- Amante AJ, Kahan BD. Technical complications of renal transplantation. Surg Clin North Am 1994; 74:1117–1131.
- Saidi RF, Wertheim JA, Ko DS, et al. Impact of donor kidney recovery method on lymphatic complications in kidney transplantation. Transplant Proc 2008; 40:1054–1055.
- Iwan-Zietek I, Zietek Z, Sulikowski T, et al. Minimally invasive methods for the treatment of lymphocele after kidney transplantation. Transplant Proc 2009; 41:3073–3076.
- Hwang EC, Kang TW, Koh YS, et al. Post-transplant lymphocele: an unusual cause of acute urinary retention mimicking urethral injury. Int J Urol 2006; 13:468–470.
- Zietek Z, Sulikowski T, Tejchman K, et al. Lymphocele after kidney transplantation. Transplant Proc 2007; 39:2744–2747.
- Mahrer A, Ramchandani P, Trerotola SO, Shlansky-Goldberg RD, Itkin M. Sclerotherapy in the management of postoperative lymphocele. J Vasc Interv Radiol 2010; 21:1050–1053.
- Risaliti A, Corno V, Donini A, et al. Laparoscopic treatment of symptomatic lymphoceles after kidney transplantation. Surg Endosc 2000; 14:293–295.
- Ostrowski M, Lubikowski J, Kowalczyk M, Power J. Laparoscopic lymphocele drainage after renal transplantation. Ann Transplant 2000; 5:25–27.
- Fuller TF, Kang SM, Hirose R, Feng S, Stock PG, Freise CE. Management of lymphoceles after renal transplantation: laparoscopic versus open drainage. J Urol 2003; 169:2022–2025.
- Hsu TH, Gill IS, Grune MT, et al. Laparoscopic lymphocelectomy: a multi-institutional analysis. J Urol 2000; 163:1096–1098.
Cervical cancer screening
To the Editor: In their excellent review of cervical cancer screening,1 Jin and colleagues discussed the current screening guidelines advocated by various medical organizations. The authors wisely advised clinicians to modify these guidelines when the lifestyle of an individual patient differs from the expected behavior of the patient’s peer group. For example, they said “it is probably reasonable to continue screening in women age 70 and older who are sexually active with multiple partners and who have a history of abnormal Pap test results.”
To this I would add that it seems reasonable to continue screening a woman over 70 who is sexually active with multiple partners, even if she still has no history of abnormal Pap test results. Similar reasoning might be applied to the statement, “women age 30 and older who had negative results on both Pap and HPV testing should be screened no more often than every 3 years.” This makes sense on a population-wide basis, since women over 30 are more likely to be married and have fewer sexual partners. But why should women who continue to have multiple sex partners into their 30s be screened any less frequently than women in their 20s?
The high negative predictive value of HPV-plus-Pap testing is based on the risk characteristics of the population being screened, as well as on the technical characteristics of the tests. Rigid adherence to screening guidelines may be a disservice to individuals whose lifestyles place them at higher risk than the norm for their age cohort.
- Jin XW, Sikon A, Yen-Lieberman B. Cervical cancer screening: less testing, smarter testing. Cleve Clin J Med 2011; 78:737–747.
To the Editor: In their excellent review of cervical cancer screening,1 Jin and colleagues discussed the current screening guidelines advocated by various medical organizations. The authors wisely advised clinicians to modify these guidelines when the lifestyle of an individual patient differs from the expected behavior of the patient’s peer group. For example, they said “it is probably reasonable to continue screening in women age 70 and older who are sexually active with multiple partners and who have a history of abnormal Pap test results.”
To this I would add that it seems reasonable to continue screening a woman over 70 who is sexually active with multiple partners, even if she still has no history of abnormal Pap test results. Similar reasoning might be applied to the statement, “women age 30 and older who had negative results on both Pap and HPV testing should be screened no more often than every 3 years.” This makes sense on a population-wide basis, since women over 30 are more likely to be married and have fewer sexual partners. But why should women who continue to have multiple sex partners into their 30s be screened any less frequently than women in their 20s?
The high negative predictive value of HPV-plus-Pap testing is based on the risk characteristics of the population being screened, as well as on the technical characteristics of the tests. Rigid adherence to screening guidelines may be a disservice to individuals whose lifestyles place them at higher risk than the norm for their age cohort.
To the Editor: In their excellent review of cervical cancer screening,1 Jin and colleagues discussed the current screening guidelines advocated by various medical organizations. The authors wisely advised clinicians to modify these guidelines when the lifestyle of an individual patient differs from the expected behavior of the patient’s peer group. For example, they said “it is probably reasonable to continue screening in women age 70 and older who are sexually active with multiple partners and who have a history of abnormal Pap test results.”
To this I would add that it seems reasonable to continue screening a woman over 70 who is sexually active with multiple partners, even if she still has no history of abnormal Pap test results. Similar reasoning might be applied to the statement, “women age 30 and older who had negative results on both Pap and HPV testing should be screened no more often than every 3 years.” This makes sense on a population-wide basis, since women over 30 are more likely to be married and have fewer sexual partners. But why should women who continue to have multiple sex partners into their 30s be screened any less frequently than women in their 20s?
The high negative predictive value of HPV-plus-Pap testing is based on the risk characteristics of the population being screened, as well as on the technical characteristics of the tests. Rigid adherence to screening guidelines may be a disservice to individuals whose lifestyles place them at higher risk than the norm for their age cohort.
- Jin XW, Sikon A, Yen-Lieberman B. Cervical cancer screening: less testing, smarter testing. Cleve Clin J Med 2011; 78:737–747.
- Jin XW, Sikon A, Yen-Lieberman B. Cervical cancer screening: less testing, smarter testing. Cleve Clin J Med 2011; 78:737–747.
Exploring the human genome, and relearning genetics by necessity
The ability to scan the entire human genome and to recognize variations in specific nucleotides within recognized genes is more than a technologic feat. It is now possible to assess the risk of some genetic diseases before they are phenotypically expressed. We are increasingly able to predict whether specific drugs will be effective or pose higher risks of adverse effects in individual patients, a field called pharmacogenomics. How much pharmacogenomics can and should be incorporated into our practice as part of personalized medicine remains to be determined,
Genome-wide association studies can answer certain research questions, but also raise additional ones. In some ways, these studies are like molecular epidemiology—they can demonstrate a statistical association between a risk factor and a clinical event such as a heart attack, but just as in traditional epidemiologic studies, association does not always equate with causation.
As discussed by Drs. Manace and Babyatsky in this issue of the Journal, additional techniques can be used to try to sort out the issue of association vs causation—in this case, whether C-reactive protein (CRP) is merely associated with cardiovascular events or is a cause of them. Using the tools of traditional clinical research, it would be ideal to demonstrate that the use of a highly specific inhibitor of the risk factor (CRP) prevents the disease. CRP levels can be lowered with statins, but these drugs also reduce levels of low-density lipoprotein cholesterol, which will lower the risk of cardiac events. Thus, statins do not have the specificity to prove that CRP causes myocardial infarction.
This paper is one of the first in the Journal to discuss advances in genomics that may affect our practice. Beginning in May, the Journal will begin a new series on personalized medicine to highlight the role that genetics and molecular medicine can play in our clinical practice and in our understanding of pathophysiology.
The ability to scan the entire human genome and to recognize variations in specific nucleotides within recognized genes is more than a technologic feat. It is now possible to assess the risk of some genetic diseases before they are phenotypically expressed. We are increasingly able to predict whether specific drugs will be effective or pose higher risks of adverse effects in individual patients, a field called pharmacogenomics. How much pharmacogenomics can and should be incorporated into our practice as part of personalized medicine remains to be determined,
Genome-wide association studies can answer certain research questions, but also raise additional ones. In some ways, these studies are like molecular epidemiology—they can demonstrate a statistical association between a risk factor and a clinical event such as a heart attack, but just as in traditional epidemiologic studies, association does not always equate with causation.
As discussed by Drs. Manace and Babyatsky in this issue of the Journal, additional techniques can be used to try to sort out the issue of association vs causation—in this case, whether C-reactive protein (CRP) is merely associated with cardiovascular events or is a cause of them. Using the tools of traditional clinical research, it would be ideal to demonstrate that the use of a highly specific inhibitor of the risk factor (CRP) prevents the disease. CRP levels can be lowered with statins, but these drugs also reduce levels of low-density lipoprotein cholesterol, which will lower the risk of cardiac events. Thus, statins do not have the specificity to prove that CRP causes myocardial infarction.
This paper is one of the first in the Journal to discuss advances in genomics that may affect our practice. Beginning in May, the Journal will begin a new series on personalized medicine to highlight the role that genetics and molecular medicine can play in our clinical practice and in our understanding of pathophysiology.
The ability to scan the entire human genome and to recognize variations in specific nucleotides within recognized genes is more than a technologic feat. It is now possible to assess the risk of some genetic diseases before they are phenotypically expressed. We are increasingly able to predict whether specific drugs will be effective or pose higher risks of adverse effects in individual patients, a field called pharmacogenomics. How much pharmacogenomics can and should be incorporated into our practice as part of personalized medicine remains to be determined,
Genome-wide association studies can answer certain research questions, but also raise additional ones. In some ways, these studies are like molecular epidemiology—they can demonstrate a statistical association between a risk factor and a clinical event such as a heart attack, but just as in traditional epidemiologic studies, association does not always equate with causation.
As discussed by Drs. Manace and Babyatsky in this issue of the Journal, additional techniques can be used to try to sort out the issue of association vs causation—in this case, whether C-reactive protein (CRP) is merely associated with cardiovascular events or is a cause of them. Using the tools of traditional clinical research, it would be ideal to demonstrate that the use of a highly specific inhibitor of the risk factor (CRP) prevents the disease. CRP levels can be lowered with statins, but these drugs also reduce levels of low-density lipoprotein cholesterol, which will lower the risk of cardiac events. Thus, statins do not have the specificity to prove that CRP causes myocardial infarction.
This paper is one of the first in the Journal to discuss advances in genomics that may affect our practice. Beginning in May, the Journal will begin a new series on personalized medicine to highlight the role that genetics and molecular medicine can play in our clinical practice and in our understanding of pathophysiology.
Putting genome analysis to good use: Lessons from C-reactive protein and cardiovascular disease
Genomics research is paying off, not only by identifying people at risk of rare inherited diseases but also by clarifying the pathogenic mechanisms of important, common ones.
Thanks to advances in technology, we can now, at a reasonable cost, simultaneously screen for millions of genetic variants in thousands of people to find variants that are more common in people with a given disease than without the disease, a fruitful method called a genome-wide association study. Moreover, an epidemiologic method called mendelian randomization takes advantage of the natural reshuffling of the genetic deck that occurs with each generation to give an estimate of whether certain gene products are mediators—or merely markers—of disease.
In a landmark study published in 2009, Elliott et al1 used mendelian randomization to evaluate the role of C-reactive protein (CRP) in coronary artery disease.
Here, we review the use of genetic tools in a clinical context, highlighting CRP to illustrate some of the potential uses and limitations of applied genomics in clinical investigation.
NATURE VS NURTURE: AN AGE-OLD DEBATE
The relative contributions of genetic and environmental factors to human health and disease— nature vs nurture—is an age-old debate in which interest has been renewed in this era of intensive research in molecular genetics.
In the 19th century, Charles Darwin proposed that evolution proceeds through natural selection of variations in inherited traits. His contemporary, Gregor Mendel, showed that traits are inherited in discrete units, later named genes. Just what genes were and how they worked had to await the discovery of the structure of DNA in 1953, by Watson and Crick.2
Since then, progress has accelerated. Advances in recombinant DNA and DNA-sequencing technologies enabled sequencing of the entire human genome only 50 years later. More recently, we have seen automated rapid sequencing, the HapMap project (more on this below), and the advent of genome-wide association studies that uncover genetic variants correlated with or predisposing to common, complex human diseases.
Until recent years, medical genetics was mostly confined to the study of rare syndromes, such as Huntington disease, that are due either to a change in a single gene or to abnormal quantities of large swaths of chromosomes containing many genes. It had little application to most of the common disorders seen by primary care physicians. However, the genes and pathways implicated in rare monogenic disorders have provided key insights into common diseases. For example, defining the genes and mutations underlying familial hypercholesterolemia highlighted the role of low-density lipoprotein cholesterol (LDL-C) in the pathogenesis of atherosclerotic disease.
3.4 BILLION BASE PAIRS, 23,000 GENES
The DNA molecule consists of two strings of the nucleotides guanine (G), cytosine (C), thymine (T), and adenine (A). The human genome contains about 3.4 billion of these nucleotides, also called base pairs, as they bind G to C and A to T across the length of the double helix of the DNA molecule.
Only about 2% of these 3.4 billion base pairs make up genes, ie, sequences that are transcribed into RNA and then translated into protein. Humans have only about 23,000 genes, which is less than in some plant species.
What about the rest of the human genome, ie, most of it? Previously dismissed as “junk,” these regions likely possess more elusive regulatory functions, controlling gene expression (ultimately, the production of protein), which varies considerably from tissue to tissue and over a person’s lifetime.
It is the orchestration of gene expression over time and cell type that gives the human body its intricate complexity. The study of how all our genes and gene products interact is called genomics and is part of the larger topic of the network of protein interactions (proteomics) and of the integration of various protein pathways (metabolomics).
We are all 99% identical—or 12 million nucleotides different
Human genome sequences are 99% identical across populations. But the remaining 1% is still a big number: there are more than 12 million variants between any two individuals’ genomes. These variants include:
- Single-nucleotide polymorphisms (SNPs), ie, a single-nucleotide change that is present in at least 1% of the population
- Copy number variants (CNVs), ie, a stretch of DNA that is either missing or duplicated
- Repeating patterns of DNA that vary in the number of repeated sequences.
THE EVOLUTION OF GENOMICS RESEARCH
Much of the initial focus of research in the genomics era consisted of identifying these variants and discovering associations between them and particular human diseases or clinical outcomes. In this way, we uncovered a multitude of potential new biomarkers and therapeutic targets, requiring further investigation into the connection between the DNA variant and the clinical state.
At the close of the 20th century, genetic factors were correlated with human disease by linkage analysis (a method of mapping patterns of markers that congregate in relatively narrow regions of DNA in families with specific diseases), and candidate gene approaches, whereby genes were investigated on the basis of their postulated biology and of previous studies. These techniques were relatively low-yield and cumbersome; years of work uncovered only a handful of genes proven to be associated with diseases.
Newer tools can look at scores of genes linked to common diseases. Researchers now rely on sophisticated DNA sequencing tools and interpretation software to sift masses of data to find meaningful markers (DNA variants or mutations).
Genomics research in the past few years has been mostly hypothesis-independent. Investigators are no longer limited to the small cache of genes whose corresponding proteins are well characterized, but can instead probe the entire genome for connections between our DNA and our physiology.
The rise of genome-wide association studies
Over the past decade, much clinically useful information has been gathered in genome-wide association studies.
The rise of this type of study rested on our emerging understanding of the architecture of our genome. When the genomes of multiple humans were fully sequenced, we discovered that specific variants do not occur randomly in relation to each other. Rather, they tend to be inherited in particular blocks called haplotypes, and some SNPs or combinations of SNPS are very rare or essentially never seen.
In its first phase, the HapMap project organized these useful blocks of variants, genotyping 1 million SNPs for each of 270 individuals from mother-father-offspring trios from distinct geographic regions of the world.3 The second phase of the HapMap project extended the analysis to more than 3 million SNPs and to other populations.4
While the HapMap should be generally applicable to other populations not yet studied, limitations of the first two HapMap phases include rare SNPs or CNVs, or variants outside of haplotype regions.
The 1,000 Genomes Project, now under way, will develop an even more comprehensive catalog of human genetic variants in much broader populations.
The success of genome-wide association studies is also partly attributable to progress in DNA-sequencing technology. Using microarray chips, we can now look at millions of SNPs per patient or the entire coding sequence of the genome (termed the exome) in a single experiment that is both time-effecient and cost-effective.
What is a genome-wide association study?
A genome-wide association study generally compares genetic variants between patients with a particular clinical condition (cases) and people without the condition (controls), looking for statistically significant differences. As a tool for genetic discovery, these studies have revealed many avenues for further investigation in the pathogenesis of disease, as well as potential targets of therapy.
Using these studies, research groups around the world have found reproducible correlations between genetic variants and susceptibility to common adult-onset diseases.
Although many of the variants identified in these studies are associated with only a slightly higher risk of disease, the method is free of many of the inherent biases associated with clinical research. These studies permit a comprehensive, hypothesis-independent and unbiased scan of the genome to identify novel susceptibility factors, whereas earlier genetic epidemiology studies could take on only a handful of variables to evaluate at a time. Additionally, they are powered to detect very small increases (or decreases) in disease risk, previously outside the reach of linkage analysis. Polymorphisms (or, presumably, non-disease-causing DNA changes) discovered using these studies often correlate with clinical phenotypes or with levels of biomarkers, even if the genetic variants are not necessarily pathologic in themselves.
Thus, genome-wide association studies have led to important insights into the pathogenesis of multiple common diseases, such as inflammatory bowel disease and diabetes mellitus, and they are facilitating new treatment approaches. For instance, multiple studies have reproduced an association between Crohn disease and variation in the gene NOD2, whose protein product is implicated in bacterial product recognition, autophagy, and apoptosis.5 This discovery led to the investigation of new potential therapies for Crohn disease, ie, the tyrosine kinase inhibitors gefitinib (Iressa) and erlotinib (Tarceva), known to inhibit NOD2 activity, and to the prognostic use of the NOD2 genotype in Crohn disease (a field of study known as genotype-phenotype correlation).
Future advances will likely come from looking at combinations of variants, which may carry a higher risk of disease than single variants.
CORONARY HEART DISEASE: FRESH INSIGHT INTO AN OLD PROBLEM
Cardiovascular disease accounts for 30% of deaths worldwide.6 Of all the cardiovascular disorders, coronary heart disease is rising most rapidly in incidence, as the rest of the world adopts Western practices such as a high-calorie, high-fat, high-glycemic diet.
Hundreds of risk factors for coronary heart disease have been described.7 Three of them are clearly modifiable participants in the pathogenesis of atherosclerosis: hypertension, smoking, and elevated LDL-C. These and others form the basis for risk-assessment tools such as the Framingham risk score and the Prospective Cardiovascular Münster (PROCAM) study score. Other possible markers require further evaluation as to whether they are clinically useful and are direct mediators of coronary heart disease.
Because up to 40% of coronary deaths occur in people who lack conventional risk factors for it (eg, they do not smoke and they have normal levels of LDL-C and blood pressure), researchers are searching hard for new, potentially treatable risk factors.8 Of particular interest are components of inflammatory pathways linked with atherosclerosis and coronary heart disease. The identity of the key inflammatory factors that cause arterial plaque formation and rupture continues to be studied.
CRP, an acute-phase reactant produced by the liver in response to inflammation, has received much attention, as serum CRP levels correlate strongly with coronary events. Researchers have used modifiers of CRP to try to alter the course of coronary heart disease, but traditional research has so far failed to establish a causal relationship between CRP and coronary heart disease.9
How we know that LDL-C is a mediator, not just a marker
As a risk factor, LDL-C resembles CRP in that its levels correlate with a number of other, confounding risk factors. Therefore, much basic research and clinical observation had to be done before we could say that LDL-C plays a role in the pathogenesis of coronary heart disease.
Initially an association between LDL-C and heart disease was noted.10 Then, studies of familial hypercholesterolemia uncovered genetic abnormalities that increase LDL-C levels and, thereby, the risk of coronary heart disease—eg, mutations in the LDL receptor gene,11–14 the apolipoprotein B (APOB) gene at its LDL receptor-binding domain,15LDL-RAP1 (a gene encoding an accessory adaptor protein that interacts with the LDL receptor),16 and PCSK9 (a gene that codes for proprotein convertase subtilisin-kexin type 9 protease).17
Conversely, specific loss-of-function truncating mutations of PCSK9 that reduce LDL-C levels are associated with strong protection against coronary heart disease.18 Other gene mutations that reduce LDL-C also lower the risk.19,20
Further, a genome-wide association study21 identified multiple genetic variations associated with different forms of dyslipidemia, uncovering additional links between LDL-C and coronary heart disease.
Finally, randomized controlled trials of niacin, fibrates, and statins showed that these potent LDL-C-lowering agents reduce the rate of development or progression of coronary heart disease.22,23
C-reactive protein: Marker or mediator?
Unlike LDL-C, no familial syndromes of coronary heart disease have been recognized in patients who have isolated high serum levels of CRP.
Since many substances in addition to CRP increase in concentration in both acute and chronic inflammatory states, agents that lower CRP in a targeted manner would be needed for large prospective, randomized trials to show whether CRP plays a direct role in coronary heart disease. A specific CRP inhibitor, 1,6-bis(phosphocholine)-hexane, may aid in these efforts, although it is not orally bioavailable and has a very short serum half-life.24
The JUPITER trial. Statins lower levels of both LDL-C and CRP. The Justification for the Use of Statins in Primary Prevention: an Intervention Evaluating Rosuvastatin (JUPITER) trial was designed to find out whether statins alter coronary risk in patients with “normal” LDL-C levels (< 130 mg/dL) and elevated CRP levels (> 2 g/L).25
In this prospective, randomized trial, statin treatment resulted in a dramatic risk reduction of 40% to 50% in multiple coronary end points, as well as a reduction in CRP levels of 37% compared with placebo. However, LDL-C levels fell by 50%, confounding the effect on CRP, as the lower coronary event rate could alternatively be explained by the effect of lower-than-normal LDL-C levels. Thus, a causative link between CRP and coronary heart disease could not be proved.26
Though ongoing trials may further illuminate the role of inflammation in the development of coronary heart disease, and specific CRP inhibitors are in development, we have few tools to answer the fundamental question of whether CRP itself is an active participant in cardiovascular disease progression or if it is a bystander marker, helping to define risk for patients who develop coronary heart disease without other known risk factors.
Of note, adding CRP to the Framingham risk score does not improve its predictive power very much in any age group.27,28 Nevertheless, for certain end points, such as the long-term rate of death after percutaneous coronary intervention29 or of cardiovascular death immediately after coronary artery bypass grafting,30 CRP levels predict coronary events reliably.
BIOMARKERS AND MENDELIAN RANDOMIZATION
Further insight into the CRP-coronary association may lie in the genes. Intriguingly, while mutations have been found that alter the serum concentration of CRP, these isolated changes in CRP levels have not yet been shown to affect heart disease risk.9,31,32
If one were to design a prospective, interventional study to evaluate the role of CRP in coronary heart disease, it would be very difficult to tease apart the specific impact of CRP from that of other variables that are often present in people with high CRP, such as obesity and hyperlipidemia. The technique of mendelian randomization offers a way to evaluate the correlation between coronary heart disease development and CRP levels independent of other risk factors.
How many heart attacks in people with or without polymorphisms?
Mendelian randomization takes advantage of a basic genetic principle, ie, the independent assortment of traits. According to Mendel’s second law, alleles for different traits are inherited independently of one another. Therefore, the gene that encodes CRP and other genes that influence its circulating level are presumably inherited independently from other genes that influence coronary risk.
In typical studies of CRP, participants are grouped according to whether they have high or low CRP levels. In these studies, confounding variables congregate in these two groups. For example, people with high CRP may be more likely to smoke and to have a higher body mass index and higher lipid levels—all of which influence cardiovascular outcomes. It is therefore difficult to tease out the effect of CRP levels from other background risk factors.
In contrast, in studies using mendelian randomization, patients are grouped according to whether they have a variant that affects the substance being studied (eg, CRP), and outcomes are compared between the two genetic groups.
Strengths and limitations of this method
By randomizing research subjects by gene variants affecting CRP levels, it is theoretically possible to achieve more equal stratification and minimize confounding between subgroups.33
Mendelian randomization should also address the possibility of “reverse causality,” when the intermediate trait with a potential role in disease development (eg, CRP) is actually regulated by the disease state itself (ie, “inflammation of atherosclerotic cardiovascular disease”).34
A limitation of mendelian randomization is that different genes influencing the biomarker under investigation must be proven to be truly randomly assorted among populations. It cannot be assumed that levels of a biomarker are equally distributed across cases and controls when there may in fact be non-random genetic associations.
For instance, if SNPs in various genes that affect creatine kinase levels were being compared to cardiovascular outcome, it would be important to take into account that baseline creatine kinase levels are higher in African Americans as well as in men in interpreting the study data.35
THE ELLIOTT STUDY (2009)
In a study published in 2009, Elliott et al1 mined genome-wide data collected over the last decade to bring more clarity to the issue of causality between elevated CRP and heart disease.
To accomplish mendelian randomization, the authors assessed SNPs that affect circulating CRP levels in combined sets of 28,000 cases and 100,000 controls—robust population sizes. The SNP variants included were associated with approximately 20% lower CRP levels. This degree of CRP reduction should correspond to a 6% reduction in coronary risk as predicted by meta-analysis of observational studies.
No association between low-CRP variants and heart disease
The authors found significant associations between these SNPs and CRP levels and between CRP levels and coronary heart disease, but not between the SNPs and coronary disease when results for three SNPs were combined and standardized to a 20% lower CRP level (odds ratio 1.00, 95% confidence interval 0.97–1.02).1
In view of the lack of association between coronary heart disease and SNPs that affect CRP levels, the authors suggested that the observational data linking CRP levels and coronary disease may have been confounded by other risk factors, or that the trend is due to reverse causation (the inflammatory response associated with atherosclerosis elevates CRP) rather than CRP’s directly causing heart disease.
These findings have important implications for management of cardiovascular disease, as therapeutic strategies to reduce plasma CRP levels are less likely to be beneficial.
The authors also described other genetic variants that may affect coronary heart disease. Carriers of minor alleles of SNPs in the gene for the leptin receptor LEPR and the APOE-CI-CII cluster showed a significantly higher risk of coronary heart disease.1 However, both variants were associated with lower levels of CRP (and, for the SNP in LEPR, lower body weight and body mass index), suggesting that the links with coronary heart disease are not mediated by CRP. These findings illustrate the ability of genome-wide association studies to identify novel susceptibility loci for complex disease without limiting investigation to genes previously thought to take part in coronary heart disease.
In view of the evidence from this study, it seems that the benefits accruing to patients with high CRP from lipid-lowering therapy as demonstrated in the JUPITER trial are likely not the result of CRP-lowering per se, but rather are the result of action on the underlying pathology that leads to elevation of inflammatory markers, including CRP. As an editorial accompanying the study by Elliot et al pointed out, the work not only provides important information in the effort to identify genetic markers associated with complex disease, but it also helps discern the role of the genes and their products in the progress and treatment of common diseases.36
Subsequent studies of CRP and the “directionality” of its role in coronary disease,37 as well as in other conditions such as obesity and cancer,38,39 have carried on the strategy of Elliott et al, providing further evidence for the function of CRP as a bystander in the inflammatory response and complex disease progression.
IMPLICATIONS OF THESE FINDINGS
Tools now exist to leapfrog the randomized controlled trials that have been the primary way of examining the role of potential mediators of common diseases. Mendelian randomization aids in determining whether biomarkers are involved in disease pathogenesis, are simply bystanders, or are secondary markers caused by the disease itself. While randomized controlled trials will still be important, this new approach offers the power of evaluating much larger sample sizes and more equally stratifying confounding factors between study groups by relying on independent assortment of genetic traits.
In medical care today, the prevention of coronary heart disease entails aggressive treatment of hypertension and hyperlipidemia, along with lifestyle modifications such as balanced diet, routine exercise, and smoking cessation. Given the large numbers of patients at risk, even with low risk scores using currently identified risk factors, more specific and sensitive markers (or panels of such markers) of cardiovascular risk are needed.
In the personalized medicine of the future, we will rely on markers that not only identify people at higher risk, but also tell us who would benefit from certain therapies. From the JUPITER trial, we understand that patients with elevated CRP levels may be appropriate candidates for statin therapy even if they have normal levels of LDL-C.36 The study by Elliott et al steers us away from using CRP-affecting SNPs in predicting the course of disease and also from the belief that targeting CRP alone would be a worthwhile therapeutic strategy.
The inflammatory hypothesis of coronary heart disease remains a very important area of investigation, and CRP may turn out to be one of the best biomarkers we have to predict the progression of coronary diseases. But the study by Elliott et al demonstrates that CRP-lowering drugs are unlikely to be magic bullets.
Most importantly, geneticists will partner with clinical researchers to answer important questions about biomarkers and genes, capitalizing on large sets of population data.
- Elliott P, Chambers JC, Zhang W, et al. Genetic loci associated with C-reactive protein levels and risk of coronary heart disease. JAMA 2009; 302:37–48.
- Watson JD, Crick FH. Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid. Nature 1953; 171:737–738.
- International HapMap Consortium. A haplotype map of the human genome. Nature 2005; 437:1299–1320.
- International HapMap Consortium; Frazer KA, Ballinger DG, Cox DR, et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 2007; 449:851–861.
- Hugot JP, Chamaillard M, Zouali H, et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001; 411:599–603.
- Anderson GF, Chu E. Expanding priorities—confronting chronic disease in countries with low income. N Engl J Med 2007; 356:209–211.
- Hingorani AD, Shah T, Casas JP, Humphries SE, Talmud PJ. C-reactive protein and coronary heart disease: predictive test or therapeutic target? Clin Chem 2009; 55:239–255.
- Smith SC. Current and future directions of cardiovascular risk prediction. Am J Cardiol 2006; 97:28A–32A.
- Zacho J, Tybjaerg-Hansen A, Jensen JS, Grande P, Sillesen H, Nordestgaard BG. Genetically elevated C-reactive protein and ischemic vascular disease. N Engl J Med 2008; 359:1897–1908.
- Stamler J, Wentworth D, Neaton JD. Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded? Findings in 356,222 primary screenees of the Multiple Risk Factor Intervention Trial (MRFIT). JAMA 1986; 256:2823–2838.
- Lehrman MA, Schneider WJ, Südhof TC, Brown MS, Goldstein JL, Russell DW. Mutation in LDL receptor: Alu-Alu recombination deletes exons encoding transmembrane and cytoplasmic domains. Science 1985; 227:140–146.
- Hobbs HH, Russell DW, Brown MS, Goldstein JL. The LDL receptor locus in familial hypercholesterolemia: mutational analysis of a membrane protein. Annu Rev Genet 1990; 24:133–170.
- Südhof TC, Goldstein JL, Brown MS, Russell DW. The LDL receptor gene: a mosaic of exons shared with different proteins. Science 1985; 228:815–822.
- Villéger L, Abifadel M, Allard D, et al. The UMD-LDLR database: additions to the software and 490 new entries to the database. Hum Mutat 2002; 20:81–87.
- Soria LF, Ludwig EH, Clarke HR, Vega GL, Grundy SM, McCarthy BJ. Association between a specific apolipoprotein B mutation and familial defective apolipoprotein B-100. Proc Natl Acad Sci U S A 1989; 86:587–591.
- Garcia CK, Wilund K, Arca M, et al. Autosomal recessive hypercholesterolemia caused by mutations in a putative LDL receptor adaptor protein. Science 2001; 292:1394–1398.
- Sun XM, Eden ER, Tosi I, et al. Evidence for effect of mutant PCSK9 on apolipoprotein B secretion as the cause of unusually severe dominant hypercholesterolaemia. Hum Mol Genet 2005; 14:1161–1169.
- Cohen JC, Boerwinkle E, Mosley TH, Hobbs HH. Sequence variations in PCSK9, low LDL, and protection against coronary heart disease. N Engl J Med 2006; 354:1264–1272.
- Linsel-Nitschke P, Götz A, Erdmann J, et al; Wellcome Trust Case Control Consortium (WTCCC). Lifelong reduction of LDL-cholesterol related to a common variant in the LDL-receptor gene decreases the risk of coronary artery disease—a Mendelian Randomisation study. PLoS One 2008; 3:e2986.
- Linsel-Nitschke P, Heeren J, Aherrahrou Z, et al. Genetic variation at chromosome 1p13.3 affects sortilin mRNA expression, cellular LDL-uptake and serum LDL levels which translates to the risk of coronary artery disease. Atherosclerosis 2010; 208:183–189.
- Kathiresan S, Willer CJ, Peloso GM, et al. Common variants at 30 loci contribute to polygenic dyslipidemia. Nat Genet 2009; 41:56–65.
- Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med 1996; 335:1001–1009.
- Cannon CP, Braunwald E, McCabe CH, et al; Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction 22 Investigators. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Pepys MB, Hirschfield GM, Tennent GA, et al. Targeting C-reactive protein for the treatment of cardiovascular disease. Nature 2006; 440:1217–1221.
- Ridker PM, Danielson E, Fonseca FA, et al; JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med 2008; 359:2195–2207.
- Shishehbor MH, Hazen SL. Jupiter to earth: a statin helps people with normal LDL-C and high hs-CRP, but what does it mean? Cleve Clin J Med 2009; 76:37–44.
- Shah T, Casas JP, Cooper JA, et al. Critical appraisal of CRP measurement for the prediction of coronary heart disease events: new data and systematic review of 31 prospective cohorts. Int J Epidemiol 2009; 38:217–231.
- Hamer M, Chida Y, Stamatakis E. Utility of C-reactive protein for cardiovascular risk stratification across three age groups in subjects without existing cardiovascular diseases. Am J Cardiol 2009; 104:538–542.
- Razzouk L, Muntner P, Bansilal S, et al. C-reactive protein predicts long-term mortality independently of low-density lipoprotein cholesterol in patients undergoing percutaneous coronary intervention. Am Heart J 2009; 158:277–283.
- Balciunas M, Bagdonaite L, Samalavicius R, Griskevicius L, Vuylsteke A. Pre-operative high sensitive C-reactive protein predicts cardiovascular events after coronary artery bypass grafting surgery: a prospective observational study. Ann Card Anaesth 2009; 12:127–132.
- Hunter DJ, Altshuler D, Rader DJ. From Darwin’s finches to canaries in the coal mine—mining the genome for new biology. N Engl J Med 2008; 358:2760–2763.
- Lawlor DA, Harbord RM, Timpson NJ, et al. The association of C-reactive protein and CRP genotype with coronary heart disease: findings from five studies with 4,610 cases amongst 18,637 participants. PLoS One 2008; 3:e3011.
- Lange LA, Carlson CS, Hindorff LA, et al. Association of polymorphisms in the CRP gene with circulating C-reactive protein levels and cardiovascular events. JAMA 2006; 296:2703–2711.
- Sheehan NA, Didelez V, Burton PR, Tobin MD. Mendelian randomisation and causal inference in observational epidemiology. PLoS Med 2008; 5:e177.
- Neal RC, Ferdinand KC, Ycas J, Miller E. Relationship of ethnic origin, gender, and age to blood creatine kinase levels. Am J Med 2009; 122:73–78.
- Shah SH, de Lemos JA. Biomarkers and cardiovascular disease: determining causality and quantifying contribution to risk assessment. JAMA 2009; 302:92–93.
- Nordestgaard BG, Zacho J. Lipids, atherosclerosis and CVD risk: is CRP an innocent bystander? Nutr Metab Cardiovasc Dis 2009; 19:521–524.
- Welsh P, Polisecki E, Robertson M, et al. Unraveling the directional link between adiposity and inflammation: a bidirectional Mendelian randomization approach. J Clin Endocrinol Metab 2010; 95:93–99.
- Allin KH, Nordestgaard BG, Zacho J, Tybjaerg-Hansen A, Bojesen SE. C-reactive protein and the risk of cancer: a mendelian randomization study. J Natl Cancer Inst 2010; 102:202–206.
Genomics research is paying off, not only by identifying people at risk of rare inherited diseases but also by clarifying the pathogenic mechanisms of important, common ones.
Thanks to advances in technology, we can now, at a reasonable cost, simultaneously screen for millions of genetic variants in thousands of people to find variants that are more common in people with a given disease than without the disease, a fruitful method called a genome-wide association study. Moreover, an epidemiologic method called mendelian randomization takes advantage of the natural reshuffling of the genetic deck that occurs with each generation to give an estimate of whether certain gene products are mediators—or merely markers—of disease.
In a landmark study published in 2009, Elliott et al1 used mendelian randomization to evaluate the role of C-reactive protein (CRP) in coronary artery disease.
Here, we review the use of genetic tools in a clinical context, highlighting CRP to illustrate some of the potential uses and limitations of applied genomics in clinical investigation.
NATURE VS NURTURE: AN AGE-OLD DEBATE
The relative contributions of genetic and environmental factors to human health and disease— nature vs nurture—is an age-old debate in which interest has been renewed in this era of intensive research in molecular genetics.
In the 19th century, Charles Darwin proposed that evolution proceeds through natural selection of variations in inherited traits. His contemporary, Gregor Mendel, showed that traits are inherited in discrete units, later named genes. Just what genes were and how they worked had to await the discovery of the structure of DNA in 1953, by Watson and Crick.2
Since then, progress has accelerated. Advances in recombinant DNA and DNA-sequencing technologies enabled sequencing of the entire human genome only 50 years later. More recently, we have seen automated rapid sequencing, the HapMap project (more on this below), and the advent of genome-wide association studies that uncover genetic variants correlated with or predisposing to common, complex human diseases.
Until recent years, medical genetics was mostly confined to the study of rare syndromes, such as Huntington disease, that are due either to a change in a single gene or to abnormal quantities of large swaths of chromosomes containing many genes. It had little application to most of the common disorders seen by primary care physicians. However, the genes and pathways implicated in rare monogenic disorders have provided key insights into common diseases. For example, defining the genes and mutations underlying familial hypercholesterolemia highlighted the role of low-density lipoprotein cholesterol (LDL-C) in the pathogenesis of atherosclerotic disease.
3.4 BILLION BASE PAIRS, 23,000 GENES
The DNA molecule consists of two strings of the nucleotides guanine (G), cytosine (C), thymine (T), and adenine (A). The human genome contains about 3.4 billion of these nucleotides, also called base pairs, as they bind G to C and A to T across the length of the double helix of the DNA molecule.
Only about 2% of these 3.4 billion base pairs make up genes, ie, sequences that are transcribed into RNA and then translated into protein. Humans have only about 23,000 genes, which is less than in some plant species.
What about the rest of the human genome, ie, most of it? Previously dismissed as “junk,” these regions likely possess more elusive regulatory functions, controlling gene expression (ultimately, the production of protein), which varies considerably from tissue to tissue and over a person’s lifetime.
It is the orchestration of gene expression over time and cell type that gives the human body its intricate complexity. The study of how all our genes and gene products interact is called genomics and is part of the larger topic of the network of protein interactions (proteomics) and of the integration of various protein pathways (metabolomics).
We are all 99% identical—or 12 million nucleotides different
Human genome sequences are 99% identical across populations. But the remaining 1% is still a big number: there are more than 12 million variants between any two individuals’ genomes. These variants include:
- Single-nucleotide polymorphisms (SNPs), ie, a single-nucleotide change that is present in at least 1% of the population
- Copy number variants (CNVs), ie, a stretch of DNA that is either missing or duplicated
- Repeating patterns of DNA that vary in the number of repeated sequences.
THE EVOLUTION OF GENOMICS RESEARCH
Much of the initial focus of research in the genomics era consisted of identifying these variants and discovering associations between them and particular human diseases or clinical outcomes. In this way, we uncovered a multitude of potential new biomarkers and therapeutic targets, requiring further investigation into the connection between the DNA variant and the clinical state.
At the close of the 20th century, genetic factors were correlated with human disease by linkage analysis (a method of mapping patterns of markers that congregate in relatively narrow regions of DNA in families with specific diseases), and candidate gene approaches, whereby genes were investigated on the basis of their postulated biology and of previous studies. These techniques were relatively low-yield and cumbersome; years of work uncovered only a handful of genes proven to be associated with diseases.
Newer tools can look at scores of genes linked to common diseases. Researchers now rely on sophisticated DNA sequencing tools and interpretation software to sift masses of data to find meaningful markers (DNA variants or mutations).
Genomics research in the past few years has been mostly hypothesis-independent. Investigators are no longer limited to the small cache of genes whose corresponding proteins are well characterized, but can instead probe the entire genome for connections between our DNA and our physiology.
The rise of genome-wide association studies
Over the past decade, much clinically useful information has been gathered in genome-wide association studies.
The rise of this type of study rested on our emerging understanding of the architecture of our genome. When the genomes of multiple humans were fully sequenced, we discovered that specific variants do not occur randomly in relation to each other. Rather, they tend to be inherited in particular blocks called haplotypes, and some SNPs or combinations of SNPS are very rare or essentially never seen.
In its first phase, the HapMap project organized these useful blocks of variants, genotyping 1 million SNPs for each of 270 individuals from mother-father-offspring trios from distinct geographic regions of the world.3 The second phase of the HapMap project extended the analysis to more than 3 million SNPs and to other populations.4
While the HapMap should be generally applicable to other populations not yet studied, limitations of the first two HapMap phases include rare SNPs or CNVs, or variants outside of haplotype regions.
The 1,000 Genomes Project, now under way, will develop an even more comprehensive catalog of human genetic variants in much broader populations.
The success of genome-wide association studies is also partly attributable to progress in DNA-sequencing technology. Using microarray chips, we can now look at millions of SNPs per patient or the entire coding sequence of the genome (termed the exome) in a single experiment that is both time-effecient and cost-effective.
What is a genome-wide association study?
A genome-wide association study generally compares genetic variants between patients with a particular clinical condition (cases) and people without the condition (controls), looking for statistically significant differences. As a tool for genetic discovery, these studies have revealed many avenues for further investigation in the pathogenesis of disease, as well as potential targets of therapy.
Using these studies, research groups around the world have found reproducible correlations between genetic variants and susceptibility to common adult-onset diseases.
Although many of the variants identified in these studies are associated with only a slightly higher risk of disease, the method is free of many of the inherent biases associated with clinical research. These studies permit a comprehensive, hypothesis-independent and unbiased scan of the genome to identify novel susceptibility factors, whereas earlier genetic epidemiology studies could take on only a handful of variables to evaluate at a time. Additionally, they are powered to detect very small increases (or decreases) in disease risk, previously outside the reach of linkage analysis. Polymorphisms (or, presumably, non-disease-causing DNA changes) discovered using these studies often correlate with clinical phenotypes or with levels of biomarkers, even if the genetic variants are not necessarily pathologic in themselves.
Thus, genome-wide association studies have led to important insights into the pathogenesis of multiple common diseases, such as inflammatory bowel disease and diabetes mellitus, and they are facilitating new treatment approaches. For instance, multiple studies have reproduced an association between Crohn disease and variation in the gene NOD2, whose protein product is implicated in bacterial product recognition, autophagy, and apoptosis.5 This discovery led to the investigation of new potential therapies for Crohn disease, ie, the tyrosine kinase inhibitors gefitinib (Iressa) and erlotinib (Tarceva), known to inhibit NOD2 activity, and to the prognostic use of the NOD2 genotype in Crohn disease (a field of study known as genotype-phenotype correlation).
Future advances will likely come from looking at combinations of variants, which may carry a higher risk of disease than single variants.
CORONARY HEART DISEASE: FRESH INSIGHT INTO AN OLD PROBLEM
Cardiovascular disease accounts for 30% of deaths worldwide.6 Of all the cardiovascular disorders, coronary heart disease is rising most rapidly in incidence, as the rest of the world adopts Western practices such as a high-calorie, high-fat, high-glycemic diet.
Hundreds of risk factors for coronary heart disease have been described.7 Three of them are clearly modifiable participants in the pathogenesis of atherosclerosis: hypertension, smoking, and elevated LDL-C. These and others form the basis for risk-assessment tools such as the Framingham risk score and the Prospective Cardiovascular Münster (PROCAM) study score. Other possible markers require further evaluation as to whether they are clinically useful and are direct mediators of coronary heart disease.
Because up to 40% of coronary deaths occur in people who lack conventional risk factors for it (eg, they do not smoke and they have normal levels of LDL-C and blood pressure), researchers are searching hard for new, potentially treatable risk factors.8 Of particular interest are components of inflammatory pathways linked with atherosclerosis and coronary heart disease. The identity of the key inflammatory factors that cause arterial plaque formation and rupture continues to be studied.
CRP, an acute-phase reactant produced by the liver in response to inflammation, has received much attention, as serum CRP levels correlate strongly with coronary events. Researchers have used modifiers of CRP to try to alter the course of coronary heart disease, but traditional research has so far failed to establish a causal relationship between CRP and coronary heart disease.9
How we know that LDL-C is a mediator, not just a marker
As a risk factor, LDL-C resembles CRP in that its levels correlate with a number of other, confounding risk factors. Therefore, much basic research and clinical observation had to be done before we could say that LDL-C plays a role in the pathogenesis of coronary heart disease.
Initially an association between LDL-C and heart disease was noted.10 Then, studies of familial hypercholesterolemia uncovered genetic abnormalities that increase LDL-C levels and, thereby, the risk of coronary heart disease—eg, mutations in the LDL receptor gene,11–14 the apolipoprotein B (APOB) gene at its LDL receptor-binding domain,15LDL-RAP1 (a gene encoding an accessory adaptor protein that interacts with the LDL receptor),16 and PCSK9 (a gene that codes for proprotein convertase subtilisin-kexin type 9 protease).17
Conversely, specific loss-of-function truncating mutations of PCSK9 that reduce LDL-C levels are associated with strong protection against coronary heart disease.18 Other gene mutations that reduce LDL-C also lower the risk.19,20
Further, a genome-wide association study21 identified multiple genetic variations associated with different forms of dyslipidemia, uncovering additional links between LDL-C and coronary heart disease.
Finally, randomized controlled trials of niacin, fibrates, and statins showed that these potent LDL-C-lowering agents reduce the rate of development or progression of coronary heart disease.22,23
C-reactive protein: Marker or mediator?
Unlike LDL-C, no familial syndromes of coronary heart disease have been recognized in patients who have isolated high serum levels of CRP.
Since many substances in addition to CRP increase in concentration in both acute and chronic inflammatory states, agents that lower CRP in a targeted manner would be needed for large prospective, randomized trials to show whether CRP plays a direct role in coronary heart disease. A specific CRP inhibitor, 1,6-bis(phosphocholine)-hexane, may aid in these efforts, although it is not orally bioavailable and has a very short serum half-life.24
The JUPITER trial. Statins lower levels of both LDL-C and CRP. The Justification for the Use of Statins in Primary Prevention: an Intervention Evaluating Rosuvastatin (JUPITER) trial was designed to find out whether statins alter coronary risk in patients with “normal” LDL-C levels (< 130 mg/dL) and elevated CRP levels (> 2 g/L).25
In this prospective, randomized trial, statin treatment resulted in a dramatic risk reduction of 40% to 50% in multiple coronary end points, as well as a reduction in CRP levels of 37% compared with placebo. However, LDL-C levels fell by 50%, confounding the effect on CRP, as the lower coronary event rate could alternatively be explained by the effect of lower-than-normal LDL-C levels. Thus, a causative link between CRP and coronary heart disease could not be proved.26
Though ongoing trials may further illuminate the role of inflammation in the development of coronary heart disease, and specific CRP inhibitors are in development, we have few tools to answer the fundamental question of whether CRP itself is an active participant in cardiovascular disease progression or if it is a bystander marker, helping to define risk for patients who develop coronary heart disease without other known risk factors.
Of note, adding CRP to the Framingham risk score does not improve its predictive power very much in any age group.27,28 Nevertheless, for certain end points, such as the long-term rate of death after percutaneous coronary intervention29 or of cardiovascular death immediately after coronary artery bypass grafting,30 CRP levels predict coronary events reliably.
BIOMARKERS AND MENDELIAN RANDOMIZATION
Further insight into the CRP-coronary association may lie in the genes. Intriguingly, while mutations have been found that alter the serum concentration of CRP, these isolated changes in CRP levels have not yet been shown to affect heart disease risk.9,31,32
If one were to design a prospective, interventional study to evaluate the role of CRP in coronary heart disease, it would be very difficult to tease apart the specific impact of CRP from that of other variables that are often present in people with high CRP, such as obesity and hyperlipidemia. The technique of mendelian randomization offers a way to evaluate the correlation between coronary heart disease development and CRP levels independent of other risk factors.
How many heart attacks in people with or without polymorphisms?
Mendelian randomization takes advantage of a basic genetic principle, ie, the independent assortment of traits. According to Mendel’s second law, alleles for different traits are inherited independently of one another. Therefore, the gene that encodes CRP and other genes that influence its circulating level are presumably inherited independently from other genes that influence coronary risk.
In typical studies of CRP, participants are grouped according to whether they have high or low CRP levels. In these studies, confounding variables congregate in these two groups. For example, people with high CRP may be more likely to smoke and to have a higher body mass index and higher lipid levels—all of which influence cardiovascular outcomes. It is therefore difficult to tease out the effect of CRP levels from other background risk factors.
In contrast, in studies using mendelian randomization, patients are grouped according to whether they have a variant that affects the substance being studied (eg, CRP), and outcomes are compared between the two genetic groups.
Strengths and limitations of this method
By randomizing research subjects by gene variants affecting CRP levels, it is theoretically possible to achieve more equal stratification and minimize confounding between subgroups.33
Mendelian randomization should also address the possibility of “reverse causality,” when the intermediate trait with a potential role in disease development (eg, CRP) is actually regulated by the disease state itself (ie, “inflammation of atherosclerotic cardiovascular disease”).34
A limitation of mendelian randomization is that different genes influencing the biomarker under investigation must be proven to be truly randomly assorted among populations. It cannot be assumed that levels of a biomarker are equally distributed across cases and controls when there may in fact be non-random genetic associations.
For instance, if SNPs in various genes that affect creatine kinase levels were being compared to cardiovascular outcome, it would be important to take into account that baseline creatine kinase levels are higher in African Americans as well as in men in interpreting the study data.35
THE ELLIOTT STUDY (2009)
In a study published in 2009, Elliott et al1 mined genome-wide data collected over the last decade to bring more clarity to the issue of causality between elevated CRP and heart disease.
To accomplish mendelian randomization, the authors assessed SNPs that affect circulating CRP levels in combined sets of 28,000 cases and 100,000 controls—robust population sizes. The SNP variants included were associated with approximately 20% lower CRP levels. This degree of CRP reduction should correspond to a 6% reduction in coronary risk as predicted by meta-analysis of observational studies.
No association between low-CRP variants and heart disease
The authors found significant associations between these SNPs and CRP levels and between CRP levels and coronary heart disease, but not between the SNPs and coronary disease when results for three SNPs were combined and standardized to a 20% lower CRP level (odds ratio 1.00, 95% confidence interval 0.97–1.02).1
In view of the lack of association between coronary heart disease and SNPs that affect CRP levels, the authors suggested that the observational data linking CRP levels and coronary disease may have been confounded by other risk factors, or that the trend is due to reverse causation (the inflammatory response associated with atherosclerosis elevates CRP) rather than CRP’s directly causing heart disease.
These findings have important implications for management of cardiovascular disease, as therapeutic strategies to reduce plasma CRP levels are less likely to be beneficial.
The authors also described other genetic variants that may affect coronary heart disease. Carriers of minor alleles of SNPs in the gene for the leptin receptor LEPR and the APOE-CI-CII cluster showed a significantly higher risk of coronary heart disease.1 However, both variants were associated with lower levels of CRP (and, for the SNP in LEPR, lower body weight and body mass index), suggesting that the links with coronary heart disease are not mediated by CRP. These findings illustrate the ability of genome-wide association studies to identify novel susceptibility loci for complex disease without limiting investigation to genes previously thought to take part in coronary heart disease.
In view of the evidence from this study, it seems that the benefits accruing to patients with high CRP from lipid-lowering therapy as demonstrated in the JUPITER trial are likely not the result of CRP-lowering per se, but rather are the result of action on the underlying pathology that leads to elevation of inflammatory markers, including CRP. As an editorial accompanying the study by Elliot et al pointed out, the work not only provides important information in the effort to identify genetic markers associated with complex disease, but it also helps discern the role of the genes and their products in the progress and treatment of common diseases.36
Subsequent studies of CRP and the “directionality” of its role in coronary disease,37 as well as in other conditions such as obesity and cancer,38,39 have carried on the strategy of Elliott et al, providing further evidence for the function of CRP as a bystander in the inflammatory response and complex disease progression.
IMPLICATIONS OF THESE FINDINGS
Tools now exist to leapfrog the randomized controlled trials that have been the primary way of examining the role of potential mediators of common diseases. Mendelian randomization aids in determining whether biomarkers are involved in disease pathogenesis, are simply bystanders, or are secondary markers caused by the disease itself. While randomized controlled trials will still be important, this new approach offers the power of evaluating much larger sample sizes and more equally stratifying confounding factors between study groups by relying on independent assortment of genetic traits.
In medical care today, the prevention of coronary heart disease entails aggressive treatment of hypertension and hyperlipidemia, along with lifestyle modifications such as balanced diet, routine exercise, and smoking cessation. Given the large numbers of patients at risk, even with low risk scores using currently identified risk factors, more specific and sensitive markers (or panels of such markers) of cardiovascular risk are needed.
In the personalized medicine of the future, we will rely on markers that not only identify people at higher risk, but also tell us who would benefit from certain therapies. From the JUPITER trial, we understand that patients with elevated CRP levels may be appropriate candidates for statin therapy even if they have normal levels of LDL-C.36 The study by Elliott et al steers us away from using CRP-affecting SNPs in predicting the course of disease and also from the belief that targeting CRP alone would be a worthwhile therapeutic strategy.
The inflammatory hypothesis of coronary heart disease remains a very important area of investigation, and CRP may turn out to be one of the best biomarkers we have to predict the progression of coronary diseases. But the study by Elliott et al demonstrates that CRP-lowering drugs are unlikely to be magic bullets.
Most importantly, geneticists will partner with clinical researchers to answer important questions about biomarkers and genes, capitalizing on large sets of population data.
Genomics research is paying off, not only by identifying people at risk of rare inherited diseases but also by clarifying the pathogenic mechanisms of important, common ones.
Thanks to advances in technology, we can now, at a reasonable cost, simultaneously screen for millions of genetic variants in thousands of people to find variants that are more common in people with a given disease than without the disease, a fruitful method called a genome-wide association study. Moreover, an epidemiologic method called mendelian randomization takes advantage of the natural reshuffling of the genetic deck that occurs with each generation to give an estimate of whether certain gene products are mediators—or merely markers—of disease.
In a landmark study published in 2009, Elliott et al1 used mendelian randomization to evaluate the role of C-reactive protein (CRP) in coronary artery disease.
Here, we review the use of genetic tools in a clinical context, highlighting CRP to illustrate some of the potential uses and limitations of applied genomics in clinical investigation.
NATURE VS NURTURE: AN AGE-OLD DEBATE
The relative contributions of genetic and environmental factors to human health and disease— nature vs nurture—is an age-old debate in which interest has been renewed in this era of intensive research in molecular genetics.
In the 19th century, Charles Darwin proposed that evolution proceeds through natural selection of variations in inherited traits. His contemporary, Gregor Mendel, showed that traits are inherited in discrete units, later named genes. Just what genes were and how they worked had to await the discovery of the structure of DNA in 1953, by Watson and Crick.2
Since then, progress has accelerated. Advances in recombinant DNA and DNA-sequencing technologies enabled sequencing of the entire human genome only 50 years later. More recently, we have seen automated rapid sequencing, the HapMap project (more on this below), and the advent of genome-wide association studies that uncover genetic variants correlated with or predisposing to common, complex human diseases.
Until recent years, medical genetics was mostly confined to the study of rare syndromes, such as Huntington disease, that are due either to a change in a single gene or to abnormal quantities of large swaths of chromosomes containing many genes. It had little application to most of the common disorders seen by primary care physicians. However, the genes and pathways implicated in rare monogenic disorders have provided key insights into common diseases. For example, defining the genes and mutations underlying familial hypercholesterolemia highlighted the role of low-density lipoprotein cholesterol (LDL-C) in the pathogenesis of atherosclerotic disease.
3.4 BILLION BASE PAIRS, 23,000 GENES
The DNA molecule consists of two strings of the nucleotides guanine (G), cytosine (C), thymine (T), and adenine (A). The human genome contains about 3.4 billion of these nucleotides, also called base pairs, as they bind G to C and A to T across the length of the double helix of the DNA molecule.
Only about 2% of these 3.4 billion base pairs make up genes, ie, sequences that are transcribed into RNA and then translated into protein. Humans have only about 23,000 genes, which is less than in some plant species.
What about the rest of the human genome, ie, most of it? Previously dismissed as “junk,” these regions likely possess more elusive regulatory functions, controlling gene expression (ultimately, the production of protein), which varies considerably from tissue to tissue and over a person’s lifetime.
It is the orchestration of gene expression over time and cell type that gives the human body its intricate complexity. The study of how all our genes and gene products interact is called genomics and is part of the larger topic of the network of protein interactions (proteomics) and of the integration of various protein pathways (metabolomics).
We are all 99% identical—or 12 million nucleotides different
Human genome sequences are 99% identical across populations. But the remaining 1% is still a big number: there are more than 12 million variants between any two individuals’ genomes. These variants include:
- Single-nucleotide polymorphisms (SNPs), ie, a single-nucleotide change that is present in at least 1% of the population
- Copy number variants (CNVs), ie, a stretch of DNA that is either missing or duplicated
- Repeating patterns of DNA that vary in the number of repeated sequences.
THE EVOLUTION OF GENOMICS RESEARCH
Much of the initial focus of research in the genomics era consisted of identifying these variants and discovering associations between them and particular human diseases or clinical outcomes. In this way, we uncovered a multitude of potential new biomarkers and therapeutic targets, requiring further investigation into the connection between the DNA variant and the clinical state.
At the close of the 20th century, genetic factors were correlated with human disease by linkage analysis (a method of mapping patterns of markers that congregate in relatively narrow regions of DNA in families with specific diseases), and candidate gene approaches, whereby genes were investigated on the basis of their postulated biology and of previous studies. These techniques were relatively low-yield and cumbersome; years of work uncovered only a handful of genes proven to be associated with diseases.
Newer tools can look at scores of genes linked to common diseases. Researchers now rely on sophisticated DNA sequencing tools and interpretation software to sift masses of data to find meaningful markers (DNA variants or mutations).
Genomics research in the past few years has been mostly hypothesis-independent. Investigators are no longer limited to the small cache of genes whose corresponding proteins are well characterized, but can instead probe the entire genome for connections between our DNA and our physiology.
The rise of genome-wide association studies
Over the past decade, much clinically useful information has been gathered in genome-wide association studies.
The rise of this type of study rested on our emerging understanding of the architecture of our genome. When the genomes of multiple humans were fully sequenced, we discovered that specific variants do not occur randomly in relation to each other. Rather, they tend to be inherited in particular blocks called haplotypes, and some SNPs or combinations of SNPS are very rare or essentially never seen.
In its first phase, the HapMap project organized these useful blocks of variants, genotyping 1 million SNPs for each of 270 individuals from mother-father-offspring trios from distinct geographic regions of the world.3 The second phase of the HapMap project extended the analysis to more than 3 million SNPs and to other populations.4
While the HapMap should be generally applicable to other populations not yet studied, limitations of the first two HapMap phases include rare SNPs or CNVs, or variants outside of haplotype regions.
The 1,000 Genomes Project, now under way, will develop an even more comprehensive catalog of human genetic variants in much broader populations.
The success of genome-wide association studies is also partly attributable to progress in DNA-sequencing technology. Using microarray chips, we can now look at millions of SNPs per patient or the entire coding sequence of the genome (termed the exome) in a single experiment that is both time-effecient and cost-effective.
What is a genome-wide association study?
A genome-wide association study generally compares genetic variants between patients with a particular clinical condition (cases) and people without the condition (controls), looking for statistically significant differences. As a tool for genetic discovery, these studies have revealed many avenues for further investigation in the pathogenesis of disease, as well as potential targets of therapy.
Using these studies, research groups around the world have found reproducible correlations between genetic variants and susceptibility to common adult-onset diseases.
Although many of the variants identified in these studies are associated with only a slightly higher risk of disease, the method is free of many of the inherent biases associated with clinical research. These studies permit a comprehensive, hypothesis-independent and unbiased scan of the genome to identify novel susceptibility factors, whereas earlier genetic epidemiology studies could take on only a handful of variables to evaluate at a time. Additionally, they are powered to detect very small increases (or decreases) in disease risk, previously outside the reach of linkage analysis. Polymorphisms (or, presumably, non-disease-causing DNA changes) discovered using these studies often correlate with clinical phenotypes or with levels of biomarkers, even if the genetic variants are not necessarily pathologic in themselves.
Thus, genome-wide association studies have led to important insights into the pathogenesis of multiple common diseases, such as inflammatory bowel disease and diabetes mellitus, and they are facilitating new treatment approaches. For instance, multiple studies have reproduced an association between Crohn disease and variation in the gene NOD2, whose protein product is implicated in bacterial product recognition, autophagy, and apoptosis.5 This discovery led to the investigation of new potential therapies for Crohn disease, ie, the tyrosine kinase inhibitors gefitinib (Iressa) and erlotinib (Tarceva), known to inhibit NOD2 activity, and to the prognostic use of the NOD2 genotype in Crohn disease (a field of study known as genotype-phenotype correlation).
Future advances will likely come from looking at combinations of variants, which may carry a higher risk of disease than single variants.
CORONARY HEART DISEASE: FRESH INSIGHT INTO AN OLD PROBLEM
Cardiovascular disease accounts for 30% of deaths worldwide.6 Of all the cardiovascular disorders, coronary heart disease is rising most rapidly in incidence, as the rest of the world adopts Western practices such as a high-calorie, high-fat, high-glycemic diet.
Hundreds of risk factors for coronary heart disease have been described.7 Three of them are clearly modifiable participants in the pathogenesis of atherosclerosis: hypertension, smoking, and elevated LDL-C. These and others form the basis for risk-assessment tools such as the Framingham risk score and the Prospective Cardiovascular Münster (PROCAM) study score. Other possible markers require further evaluation as to whether they are clinically useful and are direct mediators of coronary heart disease.
Because up to 40% of coronary deaths occur in people who lack conventional risk factors for it (eg, they do not smoke and they have normal levels of LDL-C and blood pressure), researchers are searching hard for new, potentially treatable risk factors.8 Of particular interest are components of inflammatory pathways linked with atherosclerosis and coronary heart disease. The identity of the key inflammatory factors that cause arterial plaque formation and rupture continues to be studied.
CRP, an acute-phase reactant produced by the liver in response to inflammation, has received much attention, as serum CRP levels correlate strongly with coronary events. Researchers have used modifiers of CRP to try to alter the course of coronary heart disease, but traditional research has so far failed to establish a causal relationship between CRP and coronary heart disease.9
How we know that LDL-C is a mediator, not just a marker
As a risk factor, LDL-C resembles CRP in that its levels correlate with a number of other, confounding risk factors. Therefore, much basic research and clinical observation had to be done before we could say that LDL-C plays a role in the pathogenesis of coronary heart disease.
Initially an association between LDL-C and heart disease was noted.10 Then, studies of familial hypercholesterolemia uncovered genetic abnormalities that increase LDL-C levels and, thereby, the risk of coronary heart disease—eg, mutations in the LDL receptor gene,11–14 the apolipoprotein B (APOB) gene at its LDL receptor-binding domain,15LDL-RAP1 (a gene encoding an accessory adaptor protein that interacts with the LDL receptor),16 and PCSK9 (a gene that codes for proprotein convertase subtilisin-kexin type 9 protease).17
Conversely, specific loss-of-function truncating mutations of PCSK9 that reduce LDL-C levels are associated with strong protection against coronary heart disease.18 Other gene mutations that reduce LDL-C also lower the risk.19,20
Further, a genome-wide association study21 identified multiple genetic variations associated with different forms of dyslipidemia, uncovering additional links between LDL-C and coronary heart disease.
Finally, randomized controlled trials of niacin, fibrates, and statins showed that these potent LDL-C-lowering agents reduce the rate of development or progression of coronary heart disease.22,23
C-reactive protein: Marker or mediator?
Unlike LDL-C, no familial syndromes of coronary heart disease have been recognized in patients who have isolated high serum levels of CRP.
Since many substances in addition to CRP increase in concentration in both acute and chronic inflammatory states, agents that lower CRP in a targeted manner would be needed for large prospective, randomized trials to show whether CRP plays a direct role in coronary heart disease. A specific CRP inhibitor, 1,6-bis(phosphocholine)-hexane, may aid in these efforts, although it is not orally bioavailable and has a very short serum half-life.24
The JUPITER trial. Statins lower levels of both LDL-C and CRP. The Justification for the Use of Statins in Primary Prevention: an Intervention Evaluating Rosuvastatin (JUPITER) trial was designed to find out whether statins alter coronary risk in patients with “normal” LDL-C levels (< 130 mg/dL) and elevated CRP levels (> 2 g/L).25
In this prospective, randomized trial, statin treatment resulted in a dramatic risk reduction of 40% to 50% in multiple coronary end points, as well as a reduction in CRP levels of 37% compared with placebo. However, LDL-C levels fell by 50%, confounding the effect on CRP, as the lower coronary event rate could alternatively be explained by the effect of lower-than-normal LDL-C levels. Thus, a causative link between CRP and coronary heart disease could not be proved.26
Though ongoing trials may further illuminate the role of inflammation in the development of coronary heart disease, and specific CRP inhibitors are in development, we have few tools to answer the fundamental question of whether CRP itself is an active participant in cardiovascular disease progression or if it is a bystander marker, helping to define risk for patients who develop coronary heart disease without other known risk factors.
Of note, adding CRP to the Framingham risk score does not improve its predictive power very much in any age group.27,28 Nevertheless, for certain end points, such as the long-term rate of death after percutaneous coronary intervention29 or of cardiovascular death immediately after coronary artery bypass grafting,30 CRP levels predict coronary events reliably.
BIOMARKERS AND MENDELIAN RANDOMIZATION
Further insight into the CRP-coronary association may lie in the genes. Intriguingly, while mutations have been found that alter the serum concentration of CRP, these isolated changes in CRP levels have not yet been shown to affect heart disease risk.9,31,32
If one were to design a prospective, interventional study to evaluate the role of CRP in coronary heart disease, it would be very difficult to tease apart the specific impact of CRP from that of other variables that are often present in people with high CRP, such as obesity and hyperlipidemia. The technique of mendelian randomization offers a way to evaluate the correlation between coronary heart disease development and CRP levels independent of other risk factors.
How many heart attacks in people with or without polymorphisms?
Mendelian randomization takes advantage of a basic genetic principle, ie, the independent assortment of traits. According to Mendel’s second law, alleles for different traits are inherited independently of one another. Therefore, the gene that encodes CRP and other genes that influence its circulating level are presumably inherited independently from other genes that influence coronary risk.
In typical studies of CRP, participants are grouped according to whether they have high or low CRP levels. In these studies, confounding variables congregate in these two groups. For example, people with high CRP may be more likely to smoke and to have a higher body mass index and higher lipid levels—all of which influence cardiovascular outcomes. It is therefore difficult to tease out the effect of CRP levels from other background risk factors.
In contrast, in studies using mendelian randomization, patients are grouped according to whether they have a variant that affects the substance being studied (eg, CRP), and outcomes are compared between the two genetic groups.
Strengths and limitations of this method
By randomizing research subjects by gene variants affecting CRP levels, it is theoretically possible to achieve more equal stratification and minimize confounding between subgroups.33
Mendelian randomization should also address the possibility of “reverse causality,” when the intermediate trait with a potential role in disease development (eg, CRP) is actually regulated by the disease state itself (ie, “inflammation of atherosclerotic cardiovascular disease”).34
A limitation of mendelian randomization is that different genes influencing the biomarker under investigation must be proven to be truly randomly assorted among populations. It cannot be assumed that levels of a biomarker are equally distributed across cases and controls when there may in fact be non-random genetic associations.
For instance, if SNPs in various genes that affect creatine kinase levels were being compared to cardiovascular outcome, it would be important to take into account that baseline creatine kinase levels are higher in African Americans as well as in men in interpreting the study data.35
THE ELLIOTT STUDY (2009)
In a study published in 2009, Elliott et al1 mined genome-wide data collected over the last decade to bring more clarity to the issue of causality between elevated CRP and heart disease.
To accomplish mendelian randomization, the authors assessed SNPs that affect circulating CRP levels in combined sets of 28,000 cases and 100,000 controls—robust population sizes. The SNP variants included were associated with approximately 20% lower CRP levels. This degree of CRP reduction should correspond to a 6% reduction in coronary risk as predicted by meta-analysis of observational studies.
No association between low-CRP variants and heart disease
The authors found significant associations between these SNPs and CRP levels and between CRP levels and coronary heart disease, but not between the SNPs and coronary disease when results for three SNPs were combined and standardized to a 20% lower CRP level (odds ratio 1.00, 95% confidence interval 0.97–1.02).1
In view of the lack of association between coronary heart disease and SNPs that affect CRP levels, the authors suggested that the observational data linking CRP levels and coronary disease may have been confounded by other risk factors, or that the trend is due to reverse causation (the inflammatory response associated with atherosclerosis elevates CRP) rather than CRP’s directly causing heart disease.
These findings have important implications for management of cardiovascular disease, as therapeutic strategies to reduce plasma CRP levels are less likely to be beneficial.
The authors also described other genetic variants that may affect coronary heart disease. Carriers of minor alleles of SNPs in the gene for the leptin receptor LEPR and the APOE-CI-CII cluster showed a significantly higher risk of coronary heart disease.1 However, both variants were associated with lower levels of CRP (and, for the SNP in LEPR, lower body weight and body mass index), suggesting that the links with coronary heart disease are not mediated by CRP. These findings illustrate the ability of genome-wide association studies to identify novel susceptibility loci for complex disease without limiting investigation to genes previously thought to take part in coronary heart disease.
In view of the evidence from this study, it seems that the benefits accruing to patients with high CRP from lipid-lowering therapy as demonstrated in the JUPITER trial are likely not the result of CRP-lowering per se, but rather are the result of action on the underlying pathology that leads to elevation of inflammatory markers, including CRP. As an editorial accompanying the study by Elliot et al pointed out, the work not only provides important information in the effort to identify genetic markers associated with complex disease, but it also helps discern the role of the genes and their products in the progress and treatment of common diseases.36
Subsequent studies of CRP and the “directionality” of its role in coronary disease,37 as well as in other conditions such as obesity and cancer,38,39 have carried on the strategy of Elliott et al, providing further evidence for the function of CRP as a bystander in the inflammatory response and complex disease progression.
IMPLICATIONS OF THESE FINDINGS
Tools now exist to leapfrog the randomized controlled trials that have been the primary way of examining the role of potential mediators of common diseases. Mendelian randomization aids in determining whether biomarkers are involved in disease pathogenesis, are simply bystanders, or are secondary markers caused by the disease itself. While randomized controlled trials will still be important, this new approach offers the power of evaluating much larger sample sizes and more equally stratifying confounding factors between study groups by relying on independent assortment of genetic traits.
In medical care today, the prevention of coronary heart disease entails aggressive treatment of hypertension and hyperlipidemia, along with lifestyle modifications such as balanced diet, routine exercise, and smoking cessation. Given the large numbers of patients at risk, even with low risk scores using currently identified risk factors, more specific and sensitive markers (or panels of such markers) of cardiovascular risk are needed.
In the personalized medicine of the future, we will rely on markers that not only identify people at higher risk, but also tell us who would benefit from certain therapies. From the JUPITER trial, we understand that patients with elevated CRP levels may be appropriate candidates for statin therapy even if they have normal levels of LDL-C.36 The study by Elliott et al steers us away from using CRP-affecting SNPs in predicting the course of disease and also from the belief that targeting CRP alone would be a worthwhile therapeutic strategy.
The inflammatory hypothesis of coronary heart disease remains a very important area of investigation, and CRP may turn out to be one of the best biomarkers we have to predict the progression of coronary diseases. But the study by Elliott et al demonstrates that CRP-lowering drugs are unlikely to be magic bullets.
Most importantly, geneticists will partner with clinical researchers to answer important questions about biomarkers and genes, capitalizing on large sets of population data.
- Elliott P, Chambers JC, Zhang W, et al. Genetic loci associated with C-reactive protein levels and risk of coronary heart disease. JAMA 2009; 302:37–48.
- Watson JD, Crick FH. Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid. Nature 1953; 171:737–738.
- International HapMap Consortium. A haplotype map of the human genome. Nature 2005; 437:1299–1320.
- International HapMap Consortium; Frazer KA, Ballinger DG, Cox DR, et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 2007; 449:851–861.
- Hugot JP, Chamaillard M, Zouali H, et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001; 411:599–603.
- Anderson GF, Chu E. Expanding priorities—confronting chronic disease in countries with low income. N Engl J Med 2007; 356:209–211.
- Hingorani AD, Shah T, Casas JP, Humphries SE, Talmud PJ. C-reactive protein and coronary heart disease: predictive test or therapeutic target? Clin Chem 2009; 55:239–255.
- Smith SC. Current and future directions of cardiovascular risk prediction. Am J Cardiol 2006; 97:28A–32A.
- Zacho J, Tybjaerg-Hansen A, Jensen JS, Grande P, Sillesen H, Nordestgaard BG. Genetically elevated C-reactive protein and ischemic vascular disease. N Engl J Med 2008; 359:1897–1908.
- Stamler J, Wentworth D, Neaton JD. Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded? Findings in 356,222 primary screenees of the Multiple Risk Factor Intervention Trial (MRFIT). JAMA 1986; 256:2823–2838.
- Lehrman MA, Schneider WJ, Südhof TC, Brown MS, Goldstein JL, Russell DW. Mutation in LDL receptor: Alu-Alu recombination deletes exons encoding transmembrane and cytoplasmic domains. Science 1985; 227:140–146.
- Hobbs HH, Russell DW, Brown MS, Goldstein JL. The LDL receptor locus in familial hypercholesterolemia: mutational analysis of a membrane protein. Annu Rev Genet 1990; 24:133–170.
- Südhof TC, Goldstein JL, Brown MS, Russell DW. The LDL receptor gene: a mosaic of exons shared with different proteins. Science 1985; 228:815–822.
- Villéger L, Abifadel M, Allard D, et al. The UMD-LDLR database: additions to the software and 490 new entries to the database. Hum Mutat 2002; 20:81–87.
- Soria LF, Ludwig EH, Clarke HR, Vega GL, Grundy SM, McCarthy BJ. Association between a specific apolipoprotein B mutation and familial defective apolipoprotein B-100. Proc Natl Acad Sci U S A 1989; 86:587–591.
- Garcia CK, Wilund K, Arca M, et al. Autosomal recessive hypercholesterolemia caused by mutations in a putative LDL receptor adaptor protein. Science 2001; 292:1394–1398.
- Sun XM, Eden ER, Tosi I, et al. Evidence for effect of mutant PCSK9 on apolipoprotein B secretion as the cause of unusually severe dominant hypercholesterolaemia. Hum Mol Genet 2005; 14:1161–1169.
- Cohen JC, Boerwinkle E, Mosley TH, Hobbs HH. Sequence variations in PCSK9, low LDL, and protection against coronary heart disease. N Engl J Med 2006; 354:1264–1272.
- Linsel-Nitschke P, Götz A, Erdmann J, et al; Wellcome Trust Case Control Consortium (WTCCC). Lifelong reduction of LDL-cholesterol related to a common variant in the LDL-receptor gene decreases the risk of coronary artery disease—a Mendelian Randomisation study. PLoS One 2008; 3:e2986.
- Linsel-Nitschke P, Heeren J, Aherrahrou Z, et al. Genetic variation at chromosome 1p13.3 affects sortilin mRNA expression, cellular LDL-uptake and serum LDL levels which translates to the risk of coronary artery disease. Atherosclerosis 2010; 208:183–189.
- Kathiresan S, Willer CJ, Peloso GM, et al. Common variants at 30 loci contribute to polygenic dyslipidemia. Nat Genet 2009; 41:56–65.
- Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med 1996; 335:1001–1009.
- Cannon CP, Braunwald E, McCabe CH, et al; Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction 22 Investigators. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Pepys MB, Hirschfield GM, Tennent GA, et al. Targeting C-reactive protein for the treatment of cardiovascular disease. Nature 2006; 440:1217–1221.
- Ridker PM, Danielson E, Fonseca FA, et al; JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med 2008; 359:2195–2207.
- Shishehbor MH, Hazen SL. Jupiter to earth: a statin helps people with normal LDL-C and high hs-CRP, but what does it mean? Cleve Clin J Med 2009; 76:37–44.
- Shah T, Casas JP, Cooper JA, et al. Critical appraisal of CRP measurement for the prediction of coronary heart disease events: new data and systematic review of 31 prospective cohorts. Int J Epidemiol 2009; 38:217–231.
- Hamer M, Chida Y, Stamatakis E. Utility of C-reactive protein for cardiovascular risk stratification across three age groups in subjects without existing cardiovascular diseases. Am J Cardiol 2009; 104:538–542.
- Razzouk L, Muntner P, Bansilal S, et al. C-reactive protein predicts long-term mortality independently of low-density lipoprotein cholesterol in patients undergoing percutaneous coronary intervention. Am Heart J 2009; 158:277–283.
- Balciunas M, Bagdonaite L, Samalavicius R, Griskevicius L, Vuylsteke A. Pre-operative high sensitive C-reactive protein predicts cardiovascular events after coronary artery bypass grafting surgery: a prospective observational study. Ann Card Anaesth 2009; 12:127–132.
- Hunter DJ, Altshuler D, Rader DJ. From Darwin’s finches to canaries in the coal mine—mining the genome for new biology. N Engl J Med 2008; 358:2760–2763.
- Lawlor DA, Harbord RM, Timpson NJ, et al. The association of C-reactive protein and CRP genotype with coronary heart disease: findings from five studies with 4,610 cases amongst 18,637 participants. PLoS One 2008; 3:e3011.
- Lange LA, Carlson CS, Hindorff LA, et al. Association of polymorphisms in the CRP gene with circulating C-reactive protein levels and cardiovascular events. JAMA 2006; 296:2703–2711.
- Sheehan NA, Didelez V, Burton PR, Tobin MD. Mendelian randomisation and causal inference in observational epidemiology. PLoS Med 2008; 5:e177.
- Neal RC, Ferdinand KC, Ycas J, Miller E. Relationship of ethnic origin, gender, and age to blood creatine kinase levels. Am J Med 2009; 122:73–78.
- Shah SH, de Lemos JA. Biomarkers and cardiovascular disease: determining causality and quantifying contribution to risk assessment. JAMA 2009; 302:92–93.
- Nordestgaard BG, Zacho J. Lipids, atherosclerosis and CVD risk: is CRP an innocent bystander? Nutr Metab Cardiovasc Dis 2009; 19:521–524.
- Welsh P, Polisecki E, Robertson M, et al. Unraveling the directional link between adiposity and inflammation: a bidirectional Mendelian randomization approach. J Clin Endocrinol Metab 2010; 95:93–99.
- Allin KH, Nordestgaard BG, Zacho J, Tybjaerg-Hansen A, Bojesen SE. C-reactive protein and the risk of cancer: a mendelian randomization study. J Natl Cancer Inst 2010; 102:202–206.
- Elliott P, Chambers JC, Zhang W, et al. Genetic loci associated with C-reactive protein levels and risk of coronary heart disease. JAMA 2009; 302:37–48.
- Watson JD, Crick FH. Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid. Nature 1953; 171:737–738.
- International HapMap Consortium. A haplotype map of the human genome. Nature 2005; 437:1299–1320.
- International HapMap Consortium; Frazer KA, Ballinger DG, Cox DR, et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 2007; 449:851–861.
- Hugot JP, Chamaillard M, Zouali H, et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001; 411:599–603.
- Anderson GF, Chu E. Expanding priorities—confronting chronic disease in countries with low income. N Engl J Med 2007; 356:209–211.
- Hingorani AD, Shah T, Casas JP, Humphries SE, Talmud PJ. C-reactive protein and coronary heart disease: predictive test or therapeutic target? Clin Chem 2009; 55:239–255.
- Smith SC. Current and future directions of cardiovascular risk prediction. Am J Cardiol 2006; 97:28A–32A.
- Zacho J, Tybjaerg-Hansen A, Jensen JS, Grande P, Sillesen H, Nordestgaard BG. Genetically elevated C-reactive protein and ischemic vascular disease. N Engl J Med 2008; 359:1897–1908.
- Stamler J, Wentworth D, Neaton JD. Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded? Findings in 356,222 primary screenees of the Multiple Risk Factor Intervention Trial (MRFIT). JAMA 1986; 256:2823–2838.
- Lehrman MA, Schneider WJ, Südhof TC, Brown MS, Goldstein JL, Russell DW. Mutation in LDL receptor: Alu-Alu recombination deletes exons encoding transmembrane and cytoplasmic domains. Science 1985; 227:140–146.
- Hobbs HH, Russell DW, Brown MS, Goldstein JL. The LDL receptor locus in familial hypercholesterolemia: mutational analysis of a membrane protein. Annu Rev Genet 1990; 24:133–170.
- Südhof TC, Goldstein JL, Brown MS, Russell DW. The LDL receptor gene: a mosaic of exons shared with different proteins. Science 1985; 228:815–822.
- Villéger L, Abifadel M, Allard D, et al. The UMD-LDLR database: additions to the software and 490 new entries to the database. Hum Mutat 2002; 20:81–87.
- Soria LF, Ludwig EH, Clarke HR, Vega GL, Grundy SM, McCarthy BJ. Association between a specific apolipoprotein B mutation and familial defective apolipoprotein B-100. Proc Natl Acad Sci U S A 1989; 86:587–591.
- Garcia CK, Wilund K, Arca M, et al. Autosomal recessive hypercholesterolemia caused by mutations in a putative LDL receptor adaptor protein. Science 2001; 292:1394–1398.
- Sun XM, Eden ER, Tosi I, et al. Evidence for effect of mutant PCSK9 on apolipoprotein B secretion as the cause of unusually severe dominant hypercholesterolaemia. Hum Mol Genet 2005; 14:1161–1169.
- Cohen JC, Boerwinkle E, Mosley TH, Hobbs HH. Sequence variations in PCSK9, low LDL, and protection against coronary heart disease. N Engl J Med 2006; 354:1264–1272.
- Linsel-Nitschke P, Götz A, Erdmann J, et al; Wellcome Trust Case Control Consortium (WTCCC). Lifelong reduction of LDL-cholesterol related to a common variant in the LDL-receptor gene decreases the risk of coronary artery disease—a Mendelian Randomisation study. PLoS One 2008; 3:e2986.
- Linsel-Nitschke P, Heeren J, Aherrahrou Z, et al. Genetic variation at chromosome 1p13.3 affects sortilin mRNA expression, cellular LDL-uptake and serum LDL levels which translates to the risk of coronary artery disease. Atherosclerosis 2010; 208:183–189.
- Kathiresan S, Willer CJ, Peloso GM, et al. Common variants at 30 loci contribute to polygenic dyslipidemia. Nat Genet 2009; 41:56–65.
- Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med 1996; 335:1001–1009.
- Cannon CP, Braunwald E, McCabe CH, et al; Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction 22 Investigators. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Pepys MB, Hirschfield GM, Tennent GA, et al. Targeting C-reactive protein for the treatment of cardiovascular disease. Nature 2006; 440:1217–1221.
- Ridker PM, Danielson E, Fonseca FA, et al; JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med 2008; 359:2195–2207.
- Shishehbor MH, Hazen SL. Jupiter to earth: a statin helps people with normal LDL-C and high hs-CRP, but what does it mean? Cleve Clin J Med 2009; 76:37–44.
- Shah T, Casas JP, Cooper JA, et al. Critical appraisal of CRP measurement for the prediction of coronary heart disease events: new data and systematic review of 31 prospective cohorts. Int J Epidemiol 2009; 38:217–231.
- Hamer M, Chida Y, Stamatakis E. Utility of C-reactive protein for cardiovascular risk stratification across three age groups in subjects without existing cardiovascular diseases. Am J Cardiol 2009; 104:538–542.
- Razzouk L, Muntner P, Bansilal S, et al. C-reactive protein predicts long-term mortality independently of low-density lipoprotein cholesterol in patients undergoing percutaneous coronary intervention. Am Heart J 2009; 158:277–283.
- Balciunas M, Bagdonaite L, Samalavicius R, Griskevicius L, Vuylsteke A. Pre-operative high sensitive C-reactive protein predicts cardiovascular events after coronary artery bypass grafting surgery: a prospective observational study. Ann Card Anaesth 2009; 12:127–132.
- Hunter DJ, Altshuler D, Rader DJ. From Darwin’s finches to canaries in the coal mine—mining the genome for new biology. N Engl J Med 2008; 358:2760–2763.
- Lawlor DA, Harbord RM, Timpson NJ, et al. The association of C-reactive protein and CRP genotype with coronary heart disease: findings from five studies with 4,610 cases amongst 18,637 participants. PLoS One 2008; 3:e3011.
- Lange LA, Carlson CS, Hindorff LA, et al. Association of polymorphisms in the CRP gene with circulating C-reactive protein levels and cardiovascular events. JAMA 2006; 296:2703–2711.
- Sheehan NA, Didelez V, Burton PR, Tobin MD. Mendelian randomisation and causal inference in observational epidemiology. PLoS Med 2008; 5:e177.
- Neal RC, Ferdinand KC, Ycas J, Miller E. Relationship of ethnic origin, gender, and age to blood creatine kinase levels. Am J Med 2009; 122:73–78.
- Shah SH, de Lemos JA. Biomarkers and cardiovascular disease: determining causality and quantifying contribution to risk assessment. JAMA 2009; 302:92–93.
- Nordestgaard BG, Zacho J. Lipids, atherosclerosis and CVD risk: is CRP an innocent bystander? Nutr Metab Cardiovasc Dis 2009; 19:521–524.
- Welsh P, Polisecki E, Robertson M, et al. Unraveling the directional link between adiposity and inflammation: a bidirectional Mendelian randomization approach. J Clin Endocrinol Metab 2010; 95:93–99.
- Allin KH, Nordestgaard BG, Zacho J, Tybjaerg-Hansen A, Bojesen SE. C-reactive protein and the risk of cancer: a mendelian randomization study. J Natl Cancer Inst 2010; 102:202–206.
KEY POINTS
- Genome-wide association studies can uncover associations between genetic markers and medical conditions, but they fall short of establishing causality or even clear biologic interactions between a genetic variant and a disease state.
- Mendelian randomization is a method for addressing the relationship between genetic variants and disease, ie, whether a biomarker affected by the variant is a cause of the disease or merely a bystander.
- CRP, an acute-phase reactant produced by the liver in response to inflammation, is one of many inflammatory markers whose levels correlate with coronary disease and which has been suggested to play a role in its pathogenesis.
- The findings of Elliott et al suggest that therapies that specifically lower CRP levels are not likely to affect coronary artery disease.
Correction: Presumed premature ventricular contractions
In the article “Presumed premature ventricular contractions” by Drs. Moises Auron and Donald Underwood (Cleve Clin J Med 2011; 78:812–813), Figure 1 was incorrectly labelled. The corrected figure and legend appear below. The authors wish to thank Philippe Akhrass, MD, from the State University of New York, Brooklyn, and Shahrokh Rafii, MD, from Brookdale University Hospital and Medical Center, Brooklyn, NY, for pointing out this error.
In the article “Presumed premature ventricular contractions” by Drs. Moises Auron and Donald Underwood (Cleve Clin J Med 2011; 78:812–813), Figure 1 was incorrectly labelled. The corrected figure and legend appear below. The authors wish to thank Philippe Akhrass, MD, from the State University of New York, Brooklyn, and Shahrokh Rafii, MD, from Brookdale University Hospital and Medical Center, Brooklyn, NY, for pointing out this error.

In the article “Presumed premature ventricular contractions” by Drs. Moises Auron and Donald Underwood (Cleve Clin J Med 2011; 78:812–813), Figure 1 was incorrectly labelled. The corrected figure and legend appear below. The authors wish to thank Philippe Akhrass, MD, from the State University of New York, Brooklyn, and Shahrokh Rafii, MD, from Brookdale University Hospital and Medical Center, Brooklyn, NY, for pointing out this error.

In reply: Cervical cancer screening
In Reply: We thank Dr. Keller for his excellent comment. The rationale for discontinuing screening in a woman over 70 who has multiple sexual partners without a history of an abnormal Pap test is that she is at lower risk of new-onset cervical intraepithelial neoplasia (CIN) than a younger woman because of her decreased rate of metaplasia and less accessible transformation zone. In addition, postmenopausal mucosal atrophy may predispose to false-positive cytology. False-positive results are likely to be followed by additional invasive procedures, anxiety, and cost to the patient. However, she is still at risk for acquiring human papillomavirus (HPV) and CIN. Given that cervical cancer develops slowly and risk factors decrease with age, it is reasonable to stop screening at this point. Also, the recommendation of the 3-year screening interval in women over 30 with multiple sexual partners who had negative Pap and HPV tests is based on the fact that they can acquire HPV the day after screening and subsequently develop CIN, but we can detect HPV and CIN in the next round of screening (3 years later) and so will not miss the opportunity to treat cervical dysplasia.
However, practice guidelines are never meant to replace a physician’s sound clinical decision made on an individual basis.
In Reply: We thank Dr. Keller for his excellent comment. The rationale for discontinuing screening in a woman over 70 who has multiple sexual partners without a history of an abnormal Pap test is that she is at lower risk of new-onset cervical intraepithelial neoplasia (CIN) than a younger woman because of her decreased rate of metaplasia and less accessible transformation zone. In addition, postmenopausal mucosal atrophy may predispose to false-positive cytology. False-positive results are likely to be followed by additional invasive procedures, anxiety, and cost to the patient. However, she is still at risk for acquiring human papillomavirus (HPV) and CIN. Given that cervical cancer develops slowly and risk factors decrease with age, it is reasonable to stop screening at this point. Also, the recommendation of the 3-year screening interval in women over 30 with multiple sexual partners who had negative Pap and HPV tests is based on the fact that they can acquire HPV the day after screening and subsequently develop CIN, but we can detect HPV and CIN in the next round of screening (3 years later) and so will not miss the opportunity to treat cervical dysplasia.
However, practice guidelines are never meant to replace a physician’s sound clinical decision made on an individual basis.
In Reply: We thank Dr. Keller for his excellent comment. The rationale for discontinuing screening in a woman over 70 who has multiple sexual partners without a history of an abnormal Pap test is that she is at lower risk of new-onset cervical intraepithelial neoplasia (CIN) than a younger woman because of her decreased rate of metaplasia and less accessible transformation zone. In addition, postmenopausal mucosal atrophy may predispose to false-positive cytology. False-positive results are likely to be followed by additional invasive procedures, anxiety, and cost to the patient. However, she is still at risk for acquiring human papillomavirus (HPV) and CIN. Given that cervical cancer develops slowly and risk factors decrease with age, it is reasonable to stop screening at this point. Also, the recommendation of the 3-year screening interval in women over 30 with multiple sexual partners who had negative Pap and HPV tests is based on the fact that they can acquire HPV the day after screening and subsequently develop CIN, but we can detect HPV and CIN in the next round of screening (3 years later) and so will not miss the opportunity to treat cervical dysplasia.
However, practice guidelines are never meant to replace a physician’s sound clinical decision made on an individual basis.