User login
Dabigatran
To the Editor: In their response to a letter to the editor (December 2011), Drs. Wartak and Bartholomew suggested the use of recombinant activated factor VIIa (NovoSeven) for bleeding in patients on dabigatran. They based this recommendation on a review by Stangier and Clemens,1 which was based on phase II and III data on the efficacy and safety of dabigatran. There have been no controlled trials or prospective data on the use of this agent for this indication, nor are there data on its use in bleeding after intracranial hemorrhage, bleeding related to cardiac surgery, or trauma-related bleeding. In a systematic review, Yank et al2 found that there is no lower mortality rate and an increased risk of thromboembolism when activated factor VIIa is used off-label. This agent is approved for use only in patients with hemophilia, and in fact Novo Nordisk paid a $25 million settlement for off-label promotion of this drug for nonapproved indications.3 Recombinant factor VIIa costs up to $10,000 per vial, and if it is used off-label, that cost is not reimbursed to the hospital.
Just because we can do something does not mean that we should do it. The use of recombinant factor VIIa for dabigatran-related bleeding needs to be studied in a controlled trial before it is routinely used. As seen in the cited review, indication drift can lead to adverse patient outcomes and will certainly lead to financial peril in hospitals across the country.
- Stangier J, Clemens A. Pharmacology, pharmacokinetics, and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor. Clin Appl Thromb Hemost 2009; 15(suppl 1):9S–16S.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Silverman E. Novo Nordisk pays $25M for off-label marketing. Pharmalot. www.pharmalot.com/2011/06/novo-nordisk-pays-25m-for-off-label-marketing. Accessed February 9, 2012.
To the Editor: In their response to a letter to the editor (December 2011), Drs. Wartak and Bartholomew suggested the use of recombinant activated factor VIIa (NovoSeven) for bleeding in patients on dabigatran. They based this recommendation on a review by Stangier and Clemens,1 which was based on phase II and III data on the efficacy and safety of dabigatran. There have been no controlled trials or prospective data on the use of this agent for this indication, nor are there data on its use in bleeding after intracranial hemorrhage, bleeding related to cardiac surgery, or trauma-related bleeding. In a systematic review, Yank et al2 found that there is no lower mortality rate and an increased risk of thromboembolism when activated factor VIIa is used off-label. This agent is approved for use only in patients with hemophilia, and in fact Novo Nordisk paid a $25 million settlement for off-label promotion of this drug for nonapproved indications.3 Recombinant factor VIIa costs up to $10,000 per vial, and if it is used off-label, that cost is not reimbursed to the hospital.
Just because we can do something does not mean that we should do it. The use of recombinant factor VIIa for dabigatran-related bleeding needs to be studied in a controlled trial before it is routinely used. As seen in the cited review, indication drift can lead to adverse patient outcomes and will certainly lead to financial peril in hospitals across the country.
To the Editor: In their response to a letter to the editor (December 2011), Drs. Wartak and Bartholomew suggested the use of recombinant activated factor VIIa (NovoSeven) for bleeding in patients on dabigatran. They based this recommendation on a review by Stangier and Clemens,1 which was based on phase II and III data on the efficacy and safety of dabigatran. There have been no controlled trials or prospective data on the use of this agent for this indication, nor are there data on its use in bleeding after intracranial hemorrhage, bleeding related to cardiac surgery, or trauma-related bleeding. In a systematic review, Yank et al2 found that there is no lower mortality rate and an increased risk of thromboembolism when activated factor VIIa is used off-label. This agent is approved for use only in patients with hemophilia, and in fact Novo Nordisk paid a $25 million settlement for off-label promotion of this drug for nonapproved indications.3 Recombinant factor VIIa costs up to $10,000 per vial, and if it is used off-label, that cost is not reimbursed to the hospital.
Just because we can do something does not mean that we should do it. The use of recombinant factor VIIa for dabigatran-related bleeding needs to be studied in a controlled trial before it is routinely used. As seen in the cited review, indication drift can lead to adverse patient outcomes and will certainly lead to financial peril in hospitals across the country.
- Stangier J, Clemens A. Pharmacology, pharmacokinetics, and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor. Clin Appl Thromb Hemost 2009; 15(suppl 1):9S–16S.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Silverman E. Novo Nordisk pays $25M for off-label marketing. Pharmalot. www.pharmalot.com/2011/06/novo-nordisk-pays-25m-for-off-label-marketing. Accessed February 9, 2012.
- Stangier J, Clemens A. Pharmacology, pharmacokinetics, and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor. Clin Appl Thromb Hemost 2009; 15(suppl 1):9S–16S.
- Yank V, Tuohy CV, Logan AC, et al. Systematic review: benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann Intern Med 2011; 154:529–540.
- Silverman E. Novo Nordisk pays $25M for off-label marketing. Pharmalot. www.pharmalot.com/2011/06/novo-nordisk-pays-25m-for-off-label-marketing. Accessed February 9, 2012.
Prenatal Exposure to Valproate Is Associated With Increased Risk of Autism and Lower IQ
Researchers advise physicians to discuss the risks with women who have epilepsy and are of childbearing potential.
BALTIMORE—Fetal exposure to the antiepileptic drug (AED) valproate increases a child’s risk of autism and impairs his or her IQ until the age of 6, according to two studies presented at the 65th Annual Meeting of the American Epilepsy Society.
Children born to mothers on valproate monotherapy have a risk of childhood autism that is five times greater than that of children without prenatal exposure to the drug. In addition, children’s IQs are negatively associated with valproate dose, but not with carbamazepine, lamotrigine, or phenytoin.
Increased Risk of Autism
Previous research in animals and small studies involving humans have suggested that valproate treatment during pregnancy was associated with an increased risk of autism in the child.
To investigate this link, Jakob Christensen, PhD, a consultant neurologist at Aarhus University Hospital in Denmark, carried out a population-based cohort study. Using the Danish Civil Registration System, he identified 655,691 children born to 428,431 mothers between 1996 and 2006.
He looked to the Danish Prescription Register to identify women who had filled prescriptions for valproate from 30 days before the estimated date of conception to the day of birth.
Dr. Christensen also used the Danish Psychiatric Register to identify children diagnosed with an autism spectrum disorder and focused particularly on the subgroup that had been diagnosed with childhood autism. He and his colleagues then estimated the risk of autism in children born to mothers who used valproate during pregnancy and adjusted the risk estimates for parental psychiatric history, maternal age, and gender of the child.
Children born after prenatal exposure to valproate had more than twice the risk of an autism spectrum disorder than those without such exposure, according to the investigators. The risk of an autism spectrum disorder was 2.6 following valproate monotherapy and 2.5 following valproate polytherapy. The risk of childhood autism in children with prenatal exposure to valproate was 4.1 following valproate monotherapy and 6.8 following valproate polytherapy.
"Stopping any anticonvulsant medication poses a danger,” Dr. Christensen commented. “Women taking valproate who are contemplating pregnancy should consult with their doctors about the possibility of transitioning to another drug or reducing the dosage of their present medication when that isn’t possible.”
NeurodevelopmentalEffects of AEDs
Kimford Meador, MD, Director of the Emory Epilepsy Center and Professor of Neurology at Emory University in Atlanta, has directed the Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study. His preliminary analysis indicated that fetal exposure to valproate impaired children’s IQ at age 3. Dr. Meador and his colleagues conducted a new analysis to determine whether valproate’s effects on IQ continued until age 6.
The NEAD study enrolled pregnant women with epilepsy on AED monotherapy from 1999 to 2004. It aims to examine the long-term neurodevelopmental effects of four common AEDs: carbamazepine, lamotrigine, phenytoin, and valproate. The primary outcome for Dr. Meador’s new analysis was IQ at age 6, as measured by the Differential Ability Scale (DAS). The team also performed a secondary analysis of verbal and nonverbal cluster scores from the DAS. The sample size was 310 children.
Dr. Meador found that child IQ was lower with valproate exposure than it was with exposure to any of the other AEDs. The adjusted mean IQ for children born after valproate exposure was 97. By contrast, the adjusted mean IQ for carbamazepine was 105, the adjusted mean IQ for lamotrigine was 108, and the adjusted mean IQ for phenytoin was 108. The verbal cluster score was less than the nonverbal cluster score for lamotrigine and valproate.
In children born after fetal exposure to valproate, the risks of low IQ and decreased verbal and nonverbal abilities are dose-dependent. However, “the dose-dependent effect is not seen with other AEDs on any of these factors,” said Dr. Meador.
Women of childbearing potential need to understand the risks that valproate entails, said Dr. Meador. “In my mind, it’s a very poor first choice for women of childbearing potential.
“The quandary is that there’s a subgroup of women with primary generalized epilepsy who will only respond to valproate,” he added. “Because it is not possible to predict which women are in this category, neurologists should treat them with another AED before trying valproate,” said Dr. Meador.
To hear an audiocast related to this news article, please click here.
Suggested Reading
Meador KJ, Baker GA, Browning N, et al. Cognitive function at 3 years of age after fetal exposure to antiepileptic drugs. N Engl J Med. 2009;360(16):1597-1605.
Sun Y, Vestergaard M, Christensen J, et al. Intake of marine n-3 fatty acids during pregnancy and risk for epilepsy in the offspring: a population-based cohort study. Epilepsy Res. 2010;91(2-3):267-272.
Researchers advise physicians to discuss the risks with women who have epilepsy and are of childbearing potential.
BALTIMORE—Fetal exposure to the antiepileptic drug (AED) valproate increases a child’s risk of autism and impairs his or her IQ until the age of 6, according to two studies presented at the 65th Annual Meeting of the American Epilepsy Society.
Children born to mothers on valproate monotherapy have a risk of childhood autism that is five times greater than that of children without prenatal exposure to the drug. In addition, children’s IQs are negatively associated with valproate dose, but not with carbamazepine, lamotrigine, or phenytoin.
Increased Risk of Autism
Previous research in animals and small studies involving humans have suggested that valproate treatment during pregnancy was associated with an increased risk of autism in the child.
To investigate this link, Jakob Christensen, PhD, a consultant neurologist at Aarhus University Hospital in Denmark, carried out a population-based cohort study. Using the Danish Civil Registration System, he identified 655,691 children born to 428,431 mothers between 1996 and 2006.
He looked to the Danish Prescription Register to identify women who had filled prescriptions for valproate from 30 days before the estimated date of conception to the day of birth.
Dr. Christensen also used the Danish Psychiatric Register to identify children diagnosed with an autism spectrum disorder and focused particularly on the subgroup that had been diagnosed with childhood autism. He and his colleagues then estimated the risk of autism in children born to mothers who used valproate during pregnancy and adjusted the risk estimates for parental psychiatric history, maternal age, and gender of the child.
Children born after prenatal exposure to valproate had more than twice the risk of an autism spectrum disorder than those without such exposure, according to the investigators. The risk of an autism spectrum disorder was 2.6 following valproate monotherapy and 2.5 following valproate polytherapy. The risk of childhood autism in children with prenatal exposure to valproate was 4.1 following valproate monotherapy and 6.8 following valproate polytherapy.
"Stopping any anticonvulsant medication poses a danger,” Dr. Christensen commented. “Women taking valproate who are contemplating pregnancy should consult with their doctors about the possibility of transitioning to another drug or reducing the dosage of their present medication when that isn’t possible.”
NeurodevelopmentalEffects of AEDs
Kimford Meador, MD, Director of the Emory Epilepsy Center and Professor of Neurology at Emory University in Atlanta, has directed the Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study. His preliminary analysis indicated that fetal exposure to valproate impaired children’s IQ at age 3. Dr. Meador and his colleagues conducted a new analysis to determine whether valproate’s effects on IQ continued until age 6.
The NEAD study enrolled pregnant women with epilepsy on AED monotherapy from 1999 to 2004. It aims to examine the long-term neurodevelopmental effects of four common AEDs: carbamazepine, lamotrigine, phenytoin, and valproate. The primary outcome for Dr. Meador’s new analysis was IQ at age 6, as measured by the Differential Ability Scale (DAS). The team also performed a secondary analysis of verbal and nonverbal cluster scores from the DAS. The sample size was 310 children.
Dr. Meador found that child IQ was lower with valproate exposure than it was with exposure to any of the other AEDs. The adjusted mean IQ for children born after valproate exposure was 97. By contrast, the adjusted mean IQ for carbamazepine was 105, the adjusted mean IQ for lamotrigine was 108, and the adjusted mean IQ for phenytoin was 108. The verbal cluster score was less than the nonverbal cluster score for lamotrigine and valproate.
In children born after fetal exposure to valproate, the risks of low IQ and decreased verbal and nonverbal abilities are dose-dependent. However, “the dose-dependent effect is not seen with other AEDs on any of these factors,” said Dr. Meador.
Women of childbearing potential need to understand the risks that valproate entails, said Dr. Meador. “In my mind, it’s a very poor first choice for women of childbearing potential.
“The quandary is that there’s a subgroup of women with primary generalized epilepsy who will only respond to valproate,” he added. “Because it is not possible to predict which women are in this category, neurologists should treat them with another AED before trying valproate,” said Dr. Meador.
To hear an audiocast related to this news article, please click here.
Researchers advise physicians to discuss the risks with women who have epilepsy and are of childbearing potential.
BALTIMORE—Fetal exposure to the antiepileptic drug (AED) valproate increases a child’s risk of autism and impairs his or her IQ until the age of 6, according to two studies presented at the 65th Annual Meeting of the American Epilepsy Society.
Children born to mothers on valproate monotherapy have a risk of childhood autism that is five times greater than that of children without prenatal exposure to the drug. In addition, children’s IQs are negatively associated with valproate dose, but not with carbamazepine, lamotrigine, or phenytoin.
Increased Risk of Autism
Previous research in animals and small studies involving humans have suggested that valproate treatment during pregnancy was associated with an increased risk of autism in the child.
To investigate this link, Jakob Christensen, PhD, a consultant neurologist at Aarhus University Hospital in Denmark, carried out a population-based cohort study. Using the Danish Civil Registration System, he identified 655,691 children born to 428,431 mothers between 1996 and 2006.
He looked to the Danish Prescription Register to identify women who had filled prescriptions for valproate from 30 days before the estimated date of conception to the day of birth.
Dr. Christensen also used the Danish Psychiatric Register to identify children diagnosed with an autism spectrum disorder and focused particularly on the subgroup that had been diagnosed with childhood autism. He and his colleagues then estimated the risk of autism in children born to mothers who used valproate during pregnancy and adjusted the risk estimates for parental psychiatric history, maternal age, and gender of the child.
Children born after prenatal exposure to valproate had more than twice the risk of an autism spectrum disorder than those without such exposure, according to the investigators. The risk of an autism spectrum disorder was 2.6 following valproate monotherapy and 2.5 following valproate polytherapy. The risk of childhood autism in children with prenatal exposure to valproate was 4.1 following valproate monotherapy and 6.8 following valproate polytherapy.
"Stopping any anticonvulsant medication poses a danger,” Dr. Christensen commented. “Women taking valproate who are contemplating pregnancy should consult with their doctors about the possibility of transitioning to another drug or reducing the dosage of their present medication when that isn’t possible.”
NeurodevelopmentalEffects of AEDs
Kimford Meador, MD, Director of the Emory Epilepsy Center and Professor of Neurology at Emory University in Atlanta, has directed the Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study. His preliminary analysis indicated that fetal exposure to valproate impaired children’s IQ at age 3. Dr. Meador and his colleagues conducted a new analysis to determine whether valproate’s effects on IQ continued until age 6.
The NEAD study enrolled pregnant women with epilepsy on AED monotherapy from 1999 to 2004. It aims to examine the long-term neurodevelopmental effects of four common AEDs: carbamazepine, lamotrigine, phenytoin, and valproate. The primary outcome for Dr. Meador’s new analysis was IQ at age 6, as measured by the Differential Ability Scale (DAS). The team also performed a secondary analysis of verbal and nonverbal cluster scores from the DAS. The sample size was 310 children.
Dr. Meador found that child IQ was lower with valproate exposure than it was with exposure to any of the other AEDs. The adjusted mean IQ for children born after valproate exposure was 97. By contrast, the adjusted mean IQ for carbamazepine was 105, the adjusted mean IQ for lamotrigine was 108, and the adjusted mean IQ for phenytoin was 108. The verbal cluster score was less than the nonverbal cluster score for lamotrigine and valproate.
In children born after fetal exposure to valproate, the risks of low IQ and decreased verbal and nonverbal abilities are dose-dependent. However, “the dose-dependent effect is not seen with other AEDs on any of these factors,” said Dr. Meador.
Women of childbearing potential need to understand the risks that valproate entails, said Dr. Meador. “In my mind, it’s a very poor first choice for women of childbearing potential.
“The quandary is that there’s a subgroup of women with primary generalized epilepsy who will only respond to valproate,” he added. “Because it is not possible to predict which women are in this category, neurologists should treat them with another AED before trying valproate,” said Dr. Meador.
To hear an audiocast related to this news article, please click here.
Suggested Reading
Meador KJ, Baker GA, Browning N, et al. Cognitive function at 3 years of age after fetal exposure to antiepileptic drugs. N Engl J Med. 2009;360(16):1597-1605.
Sun Y, Vestergaard M, Christensen J, et al. Intake of marine n-3 fatty acids during pregnancy and risk for epilepsy in the offspring: a population-based cohort study. Epilepsy Res. 2010;91(2-3):267-272.
Suggested Reading
Meador KJ, Baker GA, Browning N, et al. Cognitive function at 3 years of age after fetal exposure to antiepileptic drugs. N Engl J Med. 2009;360(16):1597-1605.
Sun Y, Vestergaard M, Christensen J, et al. Intake of marine n-3 fatty acids during pregnancy and risk for epilepsy in the offspring: a population-based cohort study. Epilepsy Res. 2010;91(2-3):267-272.
How Safe Is 3D TV for Children With Epilepsy?
When 150 children, 84 with epilepsy, were exposed to 15 minutes of three-dimensional TV, no seizures occurred—but some important EEG changes were observed.
BALTIMORE—Preliminary results from an ongoing study indicate that three-dimensional (3D) TV does not pose a significant safety hazard for children with epilepsy, researchers reported at the 65th Annual Meeting of the American Epilepsy Society.
The risk of suffering a seizure by watching 3D TV is very low, but not zero, and we think that it’s probably basically linked not to the 3D technique itself but to the content of the video,” said study coauthor Gerhard Kluger, MD, of Schön Klinik, Vogtareuth, Germany, and Paracelsus Medical University, Salzburg, Austria.
Although the electronics manufacturer Samsung has warned consumers that 3D TV may cause epileptic seizures, no previous research has investigated this possibility in children, especially in children with epilepsy, Dr. Kluger said. His study team, together with Herbert Plischke, MD, Managing Director of the Generation Research Program at the University of Munich, and other researchers, are doing so through a study that will eventually include at least 200 children with the condition.
The researchers are providing each study participant with two EEGs—a 20-minute, routine EEG that includes photo stimulation, and an EEG performed while the participant views 15 minutes of 3D video footage from the film Ice Age 3. Participants view this footage on a 50-inch 3D plasma TV while sitting about 2 meters away and wearing 3D shutter glasses.
The researchers have compiled data on 150 children and adolescents—84 with epilepsy and 66 with other neurologic conditions, such as headache, that suggested the risk of epilepsy. Participants had a mean age of 12, and 77 (51%) were male.
“We did not observe a single patient suffering a seizure during the 15 minutes” of 3D viewing, Dr. Kluger said. However, one participant with epilepsy, who typically experiences seizures four times per day, had a seizure two minutes following the viewing. “We cannot be sure it was linked to the 3D viewing, but we cannot exclude that a longer observation period might cause harm,” said Dr. Kluger.
In addition, three participants—all of whom were between the ages of 14 and 16 and had idiopathic generalized epilepsy—experienced a paroxysm increase of more than 100% during the 3D viewing. None of these adolescents made any complaints, showed any clinical signs, or experienced any subclinical seizures during the viewing.
“Interestingly, one boy with the diagnosis of ADHD (under treatment with methylphenidate) without a history of seizures showed a normal routine EEG with no epileptiform spikes but some spike during 3D viewing,” Dr. Kluger told Neurology Reviews.
It is likely that the observed paroxysm increases were triggered by non-3D content of the film, such as surprises and motion, according to Dr. Kluger. Laboratory time constraints necessitated the 15-minute period for 3D viewing and prevented researchers from comparing children’s reactions to 3D TV with their reactions to normal TV, he added. The study team is planning to perform research with a longer 3D viewing period and different video material in the future.
Most participants experienced a significant increase in lambda waves during the viewing period. The researchers attributed this increase to normal augmentation of saccadic eye movements.
Although 15 children with no history of seizures were photosensitive, as indicated by photoparoxysmal reactions during the pre-viewing EEG, no child showed any paroxysmal increase while viewing 3D. “3D seems to be no special problem for this population—at least with the 3D technology and the method we have used so far,” Dr. Kluger said.
In 10 patients with epilepsy, there was a reduction in the frequency of epileptiform activity during the 3D viewing. “This is not unusual, and it may be easily explained by an increase in alertness” during the viewing, Dr. Kluger noted. “Typically, if a child with epilepsy is alert, then he or she doesn’t seize so often.”
Seventeen percent of the children complained of dizziness, headache, or nausea. “That’s quite a lot,” Dr. Kluger said. “And my impression is that the older the children, the more likely they didn’t like the 3D.
“I think most of the children can go home and go to the video and watch it, but in some there’s doubt,” Dr. Kluger concluded. “Maybe when we study 200 children with epilepsy, we can answer these questions more precisely.”
Apart from the study, Dr. Kluger said, “I think we will continue offering this simulation for the parents, because they go home happy there was no reaction.” He also emphasized, “Children with epilepsy should do the same as children without epilepsy as much as possible.”
—Jack Baney
When 150 children, 84 with epilepsy, were exposed to 15 minutes of three-dimensional TV, no seizures occurred—but some important EEG changes were observed.
BALTIMORE—Preliminary results from an ongoing study indicate that three-dimensional (3D) TV does not pose a significant safety hazard for children with epilepsy, researchers reported at the 65th Annual Meeting of the American Epilepsy Society.
The risk of suffering a seizure by watching 3D TV is very low, but not zero, and we think that it’s probably basically linked not to the 3D technique itself but to the content of the video,” said study coauthor Gerhard Kluger, MD, of Schön Klinik, Vogtareuth, Germany, and Paracelsus Medical University, Salzburg, Austria.
Although the electronics manufacturer Samsung has warned consumers that 3D TV may cause epileptic seizures, no previous research has investigated this possibility in children, especially in children with epilepsy, Dr. Kluger said. His study team, together with Herbert Plischke, MD, Managing Director of the Generation Research Program at the University of Munich, and other researchers, are doing so through a study that will eventually include at least 200 children with the condition.
The researchers are providing each study participant with two EEGs—a 20-minute, routine EEG that includes photo stimulation, and an EEG performed while the participant views 15 minutes of 3D video footage from the film Ice Age 3. Participants view this footage on a 50-inch 3D plasma TV while sitting about 2 meters away and wearing 3D shutter glasses.
The researchers have compiled data on 150 children and adolescents—84 with epilepsy and 66 with other neurologic conditions, such as headache, that suggested the risk of epilepsy. Participants had a mean age of 12, and 77 (51%) were male.
“We did not observe a single patient suffering a seizure during the 15 minutes” of 3D viewing, Dr. Kluger said. However, one participant with epilepsy, who typically experiences seizures four times per day, had a seizure two minutes following the viewing. “We cannot be sure it was linked to the 3D viewing, but we cannot exclude that a longer observation period might cause harm,” said Dr. Kluger.
In addition, three participants—all of whom were between the ages of 14 and 16 and had idiopathic generalized epilepsy—experienced a paroxysm increase of more than 100% during the 3D viewing. None of these adolescents made any complaints, showed any clinical signs, or experienced any subclinical seizures during the viewing.
“Interestingly, one boy with the diagnosis of ADHD (under treatment with methylphenidate) without a history of seizures showed a normal routine EEG with no epileptiform spikes but some spike during 3D viewing,” Dr. Kluger told Neurology Reviews.
It is likely that the observed paroxysm increases were triggered by non-3D content of the film, such as surprises and motion, according to Dr. Kluger. Laboratory time constraints necessitated the 15-minute period for 3D viewing and prevented researchers from comparing children’s reactions to 3D TV with their reactions to normal TV, he added. The study team is planning to perform research with a longer 3D viewing period and different video material in the future.
Most participants experienced a significant increase in lambda waves during the viewing period. The researchers attributed this increase to normal augmentation of saccadic eye movements.
Although 15 children with no history of seizures were photosensitive, as indicated by photoparoxysmal reactions during the pre-viewing EEG, no child showed any paroxysmal increase while viewing 3D. “3D seems to be no special problem for this population—at least with the 3D technology and the method we have used so far,” Dr. Kluger said.
In 10 patients with epilepsy, there was a reduction in the frequency of epileptiform activity during the 3D viewing. “This is not unusual, and it may be easily explained by an increase in alertness” during the viewing, Dr. Kluger noted. “Typically, if a child with epilepsy is alert, then he or she doesn’t seize so often.”
Seventeen percent of the children complained of dizziness, headache, or nausea. “That’s quite a lot,” Dr. Kluger said. “And my impression is that the older the children, the more likely they didn’t like the 3D.
“I think most of the children can go home and go to the video and watch it, but in some there’s doubt,” Dr. Kluger concluded. “Maybe when we study 200 children with epilepsy, we can answer these questions more precisely.”
Apart from the study, Dr. Kluger said, “I think we will continue offering this simulation for the parents, because they go home happy there was no reaction.” He also emphasized, “Children with epilepsy should do the same as children without epilepsy as much as possible.”
—Jack Baney
When 150 children, 84 with epilepsy, were exposed to 15 minutes of three-dimensional TV, no seizures occurred—but some important EEG changes were observed.
BALTIMORE—Preliminary results from an ongoing study indicate that three-dimensional (3D) TV does not pose a significant safety hazard for children with epilepsy, researchers reported at the 65th Annual Meeting of the American Epilepsy Society.
The risk of suffering a seizure by watching 3D TV is very low, but not zero, and we think that it’s probably basically linked not to the 3D technique itself but to the content of the video,” said study coauthor Gerhard Kluger, MD, of Schön Klinik, Vogtareuth, Germany, and Paracelsus Medical University, Salzburg, Austria.
Although the electronics manufacturer Samsung has warned consumers that 3D TV may cause epileptic seizures, no previous research has investigated this possibility in children, especially in children with epilepsy, Dr. Kluger said. His study team, together with Herbert Plischke, MD, Managing Director of the Generation Research Program at the University of Munich, and other researchers, are doing so through a study that will eventually include at least 200 children with the condition.
The researchers are providing each study participant with two EEGs—a 20-minute, routine EEG that includes photo stimulation, and an EEG performed while the participant views 15 minutes of 3D video footage from the film Ice Age 3. Participants view this footage on a 50-inch 3D plasma TV while sitting about 2 meters away and wearing 3D shutter glasses.
The researchers have compiled data on 150 children and adolescents—84 with epilepsy and 66 with other neurologic conditions, such as headache, that suggested the risk of epilepsy. Participants had a mean age of 12, and 77 (51%) were male.
“We did not observe a single patient suffering a seizure during the 15 minutes” of 3D viewing, Dr. Kluger said. However, one participant with epilepsy, who typically experiences seizures four times per day, had a seizure two minutes following the viewing. “We cannot be sure it was linked to the 3D viewing, but we cannot exclude that a longer observation period might cause harm,” said Dr. Kluger.
In addition, three participants—all of whom were between the ages of 14 and 16 and had idiopathic generalized epilepsy—experienced a paroxysm increase of more than 100% during the 3D viewing. None of these adolescents made any complaints, showed any clinical signs, or experienced any subclinical seizures during the viewing.
“Interestingly, one boy with the diagnosis of ADHD (under treatment with methylphenidate) without a history of seizures showed a normal routine EEG with no epileptiform spikes but some spike during 3D viewing,” Dr. Kluger told Neurology Reviews.
It is likely that the observed paroxysm increases were triggered by non-3D content of the film, such as surprises and motion, according to Dr. Kluger. Laboratory time constraints necessitated the 15-minute period for 3D viewing and prevented researchers from comparing children’s reactions to 3D TV with their reactions to normal TV, he added. The study team is planning to perform research with a longer 3D viewing period and different video material in the future.
Most participants experienced a significant increase in lambda waves during the viewing period. The researchers attributed this increase to normal augmentation of saccadic eye movements.
Although 15 children with no history of seizures were photosensitive, as indicated by photoparoxysmal reactions during the pre-viewing EEG, no child showed any paroxysmal increase while viewing 3D. “3D seems to be no special problem for this population—at least with the 3D technology and the method we have used so far,” Dr. Kluger said.
In 10 patients with epilepsy, there was a reduction in the frequency of epileptiform activity during the 3D viewing. “This is not unusual, and it may be easily explained by an increase in alertness” during the viewing, Dr. Kluger noted. “Typically, if a child with epilepsy is alert, then he or she doesn’t seize so often.”
Seventeen percent of the children complained of dizziness, headache, or nausea. “That’s quite a lot,” Dr. Kluger said. “And my impression is that the older the children, the more likely they didn’t like the 3D.
“I think most of the children can go home and go to the video and watch it, but in some there’s doubt,” Dr. Kluger concluded. “Maybe when we study 200 children with epilepsy, we can answer these questions more precisely.”
Apart from the study, Dr. Kluger said, “I think we will continue offering this simulation for the parents, because they go home happy there was no reaction.” He also emphasized, “Children with epilepsy should do the same as children without epilepsy as much as possible.”
—Jack Baney
New and Noteworthy Information for March 2012
Men show higher rates of amnestic mild cognitive impairment (aMCI) and nonamnestic mild cognitive impairment (naMCI) than women, researchers reported in the January 31 Neurology. Starting in 2004, a cohort of Olmsted County, Minnesota, residents ages 70 to 89 underwent baseline and 15-month interval evaluations to assess their cognitive status. Of the 1,450 participants who were cognitively normal at baseline, 296 developed MCI, with an age- and sex-standardized incidence rate of 63.6 (per 1,000 person years) overall. Men (72.4) showed higher rates of MCI than women (57.3), and this trend continued for both aMCI and naMCI. Participants with fewer years of education had higher rates of MCI. “Differences in incidence rates by clinical subtype and by sex suggest that risk factors for MCI should be investigated separately for aMCI and naMCI, and in men and women,” the study authors stated.
Infants who are eventually diagnosed with autism spectrum disorders (ASD) show abnormal development of white matter pathways starting as early as 6 months of age, according to a study published in the February 17 online American Journal of Psychiatry. Researchers analyzed imaging results from 92 high-risk infants who had siblings with autism. The participants underwent diffusion tensor imaging at 6 months, 12 months, and 24 months. After characterizing the white matter fiber tracts of the 28 infants who met criteria for ASD at 24 months and the 64 infants who did not meet the criteria, the investigators found notable differences in the infants’ development. Compared with the 64 infants who did not develop ASD, infants who had ASD had different white matter development for 12 of the 15 brain pathways studied. “These results suggest that aberrant development of white matter pathways may precede the manifestation of autistic symptoms in the first year of life,” the researchers concluded.
Epilepsy surgery has long-term beneficial effects on patients’ seizure control and overall quality of life, according to research published in the February 7 online Epilepsia. Patients who underwent epilepsy surgery by Sidney Goldring, MD, from 1967 to 1990 were followed up for a mean duration of 26 years. Of the 361 patients who had epilepsy surgery, 117 completed follow-up interviews, and 80% reported a higher quality of life on the Quality of Life in Epilepsy (QOLIE-31) questionnaire after surgery. In addition, an association was observed between seizure freedom and better quality of life, and patients who underwent temporal lobe resection showed better seizure outcomes than those who had different procedures. Considering that the positive outcomes of epilepsy surgery from decades ago seem sustainable, the researchers said they are “optimistic that the outcomes from modern epilepsy surgery will be even better and that our present enthusiasm for this treatment modality is not misplaced.”
Elderly nursing home residents show an increased mortality risk with higher doses of antipsychotic drugs, according to a study in the February 23 online BMJ. All 75,445 study participants were age 65 or older, lived in nursing homes from 2001 to 2005, and were new users of antipsychotic drugs (haloperidol, aripiprazole, olanzapine, quetiapine, risperidone, and ziprasidone). After comparing 180-day risks of all-cause and cause-specific mortality by individual drug, the researchers found that users of haloperidol had an increased rate of mortality and users of quetiapine had a decreased risk of mortality, compared with users of risperidone. These effects were seen for all causes of mortality examined, remained after adjustment for dose, and were strongest immediately following the start of treatment. “There was no evidence that the treatment effect differed for patients with a diagnosis of dementia or behavioral disturbances,” noted the researchers. They added that although their findings do not prove causality, “….they provide more evidence of the risk of using these drugs in older patients, reinforcing the concept that they should not be used in the absence of clear need.”
—Lauren LeBano
Men show higher rates of amnestic mild cognitive impairment (aMCI) and nonamnestic mild cognitive impairment (naMCI) than women, researchers reported in the January 31 Neurology. Starting in 2004, a cohort of Olmsted County, Minnesota, residents ages 70 to 89 underwent baseline and 15-month interval evaluations to assess their cognitive status. Of the 1,450 participants who were cognitively normal at baseline, 296 developed MCI, with an age- and sex-standardized incidence rate of 63.6 (per 1,000 person years) overall. Men (72.4) showed higher rates of MCI than women (57.3), and this trend continued for both aMCI and naMCI. Participants with fewer years of education had higher rates of MCI. “Differences in incidence rates by clinical subtype and by sex suggest that risk factors for MCI should be investigated separately for aMCI and naMCI, and in men and women,” the study authors stated.
Infants who are eventually diagnosed with autism spectrum disorders (ASD) show abnormal development of white matter pathways starting as early as 6 months of age, according to a study published in the February 17 online American Journal of Psychiatry. Researchers analyzed imaging results from 92 high-risk infants who had siblings with autism. The participants underwent diffusion tensor imaging at 6 months, 12 months, and 24 months. After characterizing the white matter fiber tracts of the 28 infants who met criteria for ASD at 24 months and the 64 infants who did not meet the criteria, the investigators found notable differences in the infants’ development. Compared with the 64 infants who did not develop ASD, infants who had ASD had different white matter development for 12 of the 15 brain pathways studied. “These results suggest that aberrant development of white matter pathways may precede the manifestation of autistic symptoms in the first year of life,” the researchers concluded.
Epilepsy surgery has long-term beneficial effects on patients’ seizure control and overall quality of life, according to research published in the February 7 online Epilepsia. Patients who underwent epilepsy surgery by Sidney Goldring, MD, from 1967 to 1990 were followed up for a mean duration of 26 years. Of the 361 patients who had epilepsy surgery, 117 completed follow-up interviews, and 80% reported a higher quality of life on the Quality of Life in Epilepsy (QOLIE-31) questionnaire after surgery. In addition, an association was observed between seizure freedom and better quality of life, and patients who underwent temporal lobe resection showed better seizure outcomes than those who had different procedures. Considering that the positive outcomes of epilepsy surgery from decades ago seem sustainable, the researchers said they are “optimistic that the outcomes from modern epilepsy surgery will be even better and that our present enthusiasm for this treatment modality is not misplaced.”
Elderly nursing home residents show an increased mortality risk with higher doses of antipsychotic drugs, according to a study in the February 23 online BMJ. All 75,445 study participants were age 65 or older, lived in nursing homes from 2001 to 2005, and were new users of antipsychotic drugs (haloperidol, aripiprazole, olanzapine, quetiapine, risperidone, and ziprasidone). After comparing 180-day risks of all-cause and cause-specific mortality by individual drug, the researchers found that users of haloperidol had an increased rate of mortality and users of quetiapine had a decreased risk of mortality, compared with users of risperidone. These effects were seen for all causes of mortality examined, remained after adjustment for dose, and were strongest immediately following the start of treatment. “There was no evidence that the treatment effect differed for patients with a diagnosis of dementia or behavioral disturbances,” noted the researchers. They added that although their findings do not prove causality, “….they provide more evidence of the risk of using these drugs in older patients, reinforcing the concept that they should not be used in the absence of clear need.”
—Lauren LeBano
Men show higher rates of amnestic mild cognitive impairment (aMCI) and nonamnestic mild cognitive impairment (naMCI) than women, researchers reported in the January 31 Neurology. Starting in 2004, a cohort of Olmsted County, Minnesota, residents ages 70 to 89 underwent baseline and 15-month interval evaluations to assess their cognitive status. Of the 1,450 participants who were cognitively normal at baseline, 296 developed MCI, with an age- and sex-standardized incidence rate of 63.6 (per 1,000 person years) overall. Men (72.4) showed higher rates of MCI than women (57.3), and this trend continued for both aMCI and naMCI. Participants with fewer years of education had higher rates of MCI. “Differences in incidence rates by clinical subtype and by sex suggest that risk factors for MCI should be investigated separately for aMCI and naMCI, and in men and women,” the study authors stated.
Infants who are eventually diagnosed with autism spectrum disorders (ASD) show abnormal development of white matter pathways starting as early as 6 months of age, according to a study published in the February 17 online American Journal of Psychiatry. Researchers analyzed imaging results from 92 high-risk infants who had siblings with autism. The participants underwent diffusion tensor imaging at 6 months, 12 months, and 24 months. After characterizing the white matter fiber tracts of the 28 infants who met criteria for ASD at 24 months and the 64 infants who did not meet the criteria, the investigators found notable differences in the infants’ development. Compared with the 64 infants who did not develop ASD, infants who had ASD had different white matter development for 12 of the 15 brain pathways studied. “These results suggest that aberrant development of white matter pathways may precede the manifestation of autistic symptoms in the first year of life,” the researchers concluded.
Epilepsy surgery has long-term beneficial effects on patients’ seizure control and overall quality of life, according to research published in the February 7 online Epilepsia. Patients who underwent epilepsy surgery by Sidney Goldring, MD, from 1967 to 1990 were followed up for a mean duration of 26 years. Of the 361 patients who had epilepsy surgery, 117 completed follow-up interviews, and 80% reported a higher quality of life on the Quality of Life in Epilepsy (QOLIE-31) questionnaire after surgery. In addition, an association was observed between seizure freedom and better quality of life, and patients who underwent temporal lobe resection showed better seizure outcomes than those who had different procedures. Considering that the positive outcomes of epilepsy surgery from decades ago seem sustainable, the researchers said they are “optimistic that the outcomes from modern epilepsy surgery will be even better and that our present enthusiasm for this treatment modality is not misplaced.”
Elderly nursing home residents show an increased mortality risk with higher doses of antipsychotic drugs, according to a study in the February 23 online BMJ. All 75,445 study participants were age 65 or older, lived in nursing homes from 2001 to 2005, and were new users of antipsychotic drugs (haloperidol, aripiprazole, olanzapine, quetiapine, risperidone, and ziprasidone). After comparing 180-day risks of all-cause and cause-specific mortality by individual drug, the researchers found that users of haloperidol had an increased rate of mortality and users of quetiapine had a decreased risk of mortality, compared with users of risperidone. These effects were seen for all causes of mortality examined, remained after adjustment for dose, and were strongest immediately following the start of treatment. “There was no evidence that the treatment effect differed for patients with a diagnosis of dementia or behavioral disturbances,” noted the researchers. They added that although their findings do not prove causality, “….they provide more evidence of the risk of using these drugs in older patients, reinforcing the concept that they should not be used in the absence of clear need.”
—Lauren LeBano
Photoparoxysmal Response Is High Among Teenagers With Autism and Epilepsy
A recent finding may indicate brain irritability or hypersensitivity in children with autism spectrum disorders.
BALTIMORE—The incidence of a photoparoxysmal response among children older than 15 with autism spectrum disorders (ASD) is 25%, which is much higher than that for younger children with ASD, according to a study presented at the 65th Annual Meeting of the American Epilepsy Society. When children in the sample population without epilepsy are excluded, the incidence of a photoparoxysmal response among children older than 15 with ASD is 29.4%.
Epilepsy and frequent interictal discharges are common among children with ASD. Photoparoxysmal responses are interictal EEG discharges correlated with generalized, photosensitive epilepsy. A flashing strobe light, for example, can trigger a photoparoxysmal response in a predisposed individual. “The brain picks up that stimulus through the eye, and for some children, it can cause an abnormal brainwave pattern,” explained Jill Miller-Horn, MD, an epilepsy fellow at Children’s Hospital Boston.
“We already know that abnormal brainwave responses to flashing light can be associated with epilepsy. You might be familiar with what happened in Japan with the Pokemon cartoon, where there was bright, flashing colored light on the screen and hundreds of children then had seizures,” she added. Dr. Miller-Horn and her colleagues decided to study photoparoxysmal responses to photic stimulation in children with ASD, a topic that she said no researchers had previously investigated.
Identifying a Cohort of Children With ASD
The team conducted a retrospective pilot study of children with ASD to determine the rate of the photoparoxysmal response caused by intermittent photic stimulation during EEG studies at Children’s Hospital Boston. The investigators searched medical records that identified 333 children with ASD who were treated at the hospital between December 2010 and May 2011.
Of these children, 206 had had EEGs. In the group of 206 children, 118 had comorbid ASD and epilepsy, and 88 had ASD without seizures. Intermittent photic stimulation was part of 177 children’s EEG studies. The group of 177 included 138 boys and 39 girls, and the children’s average age was 9.
Photoparoxysmal Response Increases in Adolescence
A photoparoxysmal response was elicited in 13 of the 177 children who received photic stimulation during their EEGs. The 7.3% incidence of a photoparoxysmal response in children with ASD was within the range previously reported in the normal population, according to Dr. Miller-Horn. “Our study found that in the ASD population, there is an association between the photoparoxysmal response and epilepsy, as has been previously reported in children with epilepsy without ASD,” she said.
“When we subdivided these children with autism by age, we found that there’s an increase in the photoparoxysmal response as they entered adolescence,” commented Dr. Miller-Horn. The meaning of the result is unclear, “but it’s a difference from the normal population, and it’s a difference from other children who have epilepsy,” she added.
“This is a new finding that may be a clue to the pathophysiology for the high rate of ASD and epilepsy comorbidity,” Dr. Miller-Horn continued. “There may be irritability or hypersensitivity in the brain for children with autism that’s being revealed with the clue that they are more photosensitive.”
Large-scale and prospective studies are needed to confirm the trend, according to the investigators. Further studies could reveal the findings’ significance in the pathophysiology of epilepsy in children with ASD.
—Erik Greb
Suggested Reading
Lo C, Shorvon S, Davis M, et al. Genetic linkage analysis of a large family with photoparoxysmal response. Epilepsy Res. 2011 Nov 7; [Epub ahead of print]
Lopes da Silva FH, Harding GF. Transition to seizure in photosensitive epilepsy. Epilepsy Res. 2011;97(3):278-282.
A recent finding may indicate brain irritability or hypersensitivity in children with autism spectrum disorders.
BALTIMORE—The incidence of a photoparoxysmal response among children older than 15 with autism spectrum disorders (ASD) is 25%, which is much higher than that for younger children with ASD, according to a study presented at the 65th Annual Meeting of the American Epilepsy Society. When children in the sample population without epilepsy are excluded, the incidence of a photoparoxysmal response among children older than 15 with ASD is 29.4%.
Epilepsy and frequent interictal discharges are common among children with ASD. Photoparoxysmal responses are interictal EEG discharges correlated with generalized, photosensitive epilepsy. A flashing strobe light, for example, can trigger a photoparoxysmal response in a predisposed individual. “The brain picks up that stimulus through the eye, and for some children, it can cause an abnormal brainwave pattern,” explained Jill Miller-Horn, MD, an epilepsy fellow at Children’s Hospital Boston.
“We already know that abnormal brainwave responses to flashing light can be associated with epilepsy. You might be familiar with what happened in Japan with the Pokemon cartoon, where there was bright, flashing colored light on the screen and hundreds of children then had seizures,” she added. Dr. Miller-Horn and her colleagues decided to study photoparoxysmal responses to photic stimulation in children with ASD, a topic that she said no researchers had previously investigated.
Identifying a Cohort of Children With ASD
The team conducted a retrospective pilot study of children with ASD to determine the rate of the photoparoxysmal response caused by intermittent photic stimulation during EEG studies at Children’s Hospital Boston. The investigators searched medical records that identified 333 children with ASD who were treated at the hospital between December 2010 and May 2011.
Of these children, 206 had had EEGs. In the group of 206 children, 118 had comorbid ASD and epilepsy, and 88 had ASD without seizures. Intermittent photic stimulation was part of 177 children’s EEG studies. The group of 177 included 138 boys and 39 girls, and the children’s average age was 9.
Photoparoxysmal Response Increases in Adolescence
A photoparoxysmal response was elicited in 13 of the 177 children who received photic stimulation during their EEGs. The 7.3% incidence of a photoparoxysmal response in children with ASD was within the range previously reported in the normal population, according to Dr. Miller-Horn. “Our study found that in the ASD population, there is an association between the photoparoxysmal response and epilepsy, as has been previously reported in children with epilepsy without ASD,” she said.
“When we subdivided these children with autism by age, we found that there’s an increase in the photoparoxysmal response as they entered adolescence,” commented Dr. Miller-Horn. The meaning of the result is unclear, “but it’s a difference from the normal population, and it’s a difference from other children who have epilepsy,” she added.
“This is a new finding that may be a clue to the pathophysiology for the high rate of ASD and epilepsy comorbidity,” Dr. Miller-Horn continued. “There may be irritability or hypersensitivity in the brain for children with autism that’s being revealed with the clue that they are more photosensitive.”
Large-scale and prospective studies are needed to confirm the trend, according to the investigators. Further studies could reveal the findings’ significance in the pathophysiology of epilepsy in children with ASD.
—Erik Greb
A recent finding may indicate brain irritability or hypersensitivity in children with autism spectrum disorders.
BALTIMORE—The incidence of a photoparoxysmal response among children older than 15 with autism spectrum disorders (ASD) is 25%, which is much higher than that for younger children with ASD, according to a study presented at the 65th Annual Meeting of the American Epilepsy Society. When children in the sample population without epilepsy are excluded, the incidence of a photoparoxysmal response among children older than 15 with ASD is 29.4%.
Epilepsy and frequent interictal discharges are common among children with ASD. Photoparoxysmal responses are interictal EEG discharges correlated with generalized, photosensitive epilepsy. A flashing strobe light, for example, can trigger a photoparoxysmal response in a predisposed individual. “The brain picks up that stimulus through the eye, and for some children, it can cause an abnormal brainwave pattern,” explained Jill Miller-Horn, MD, an epilepsy fellow at Children’s Hospital Boston.
“We already know that abnormal brainwave responses to flashing light can be associated with epilepsy. You might be familiar with what happened in Japan with the Pokemon cartoon, where there was bright, flashing colored light on the screen and hundreds of children then had seizures,” she added. Dr. Miller-Horn and her colleagues decided to study photoparoxysmal responses to photic stimulation in children with ASD, a topic that she said no researchers had previously investigated.
Identifying a Cohort of Children With ASD
The team conducted a retrospective pilot study of children with ASD to determine the rate of the photoparoxysmal response caused by intermittent photic stimulation during EEG studies at Children’s Hospital Boston. The investigators searched medical records that identified 333 children with ASD who were treated at the hospital between December 2010 and May 2011.
Of these children, 206 had had EEGs. In the group of 206 children, 118 had comorbid ASD and epilepsy, and 88 had ASD without seizures. Intermittent photic stimulation was part of 177 children’s EEG studies. The group of 177 included 138 boys and 39 girls, and the children’s average age was 9.
Photoparoxysmal Response Increases in Adolescence
A photoparoxysmal response was elicited in 13 of the 177 children who received photic stimulation during their EEGs. The 7.3% incidence of a photoparoxysmal response in children with ASD was within the range previously reported in the normal population, according to Dr. Miller-Horn. “Our study found that in the ASD population, there is an association between the photoparoxysmal response and epilepsy, as has been previously reported in children with epilepsy without ASD,” she said.
“When we subdivided these children with autism by age, we found that there’s an increase in the photoparoxysmal response as they entered adolescence,” commented Dr. Miller-Horn. The meaning of the result is unclear, “but it’s a difference from the normal population, and it’s a difference from other children who have epilepsy,” she added.
“This is a new finding that may be a clue to the pathophysiology for the high rate of ASD and epilepsy comorbidity,” Dr. Miller-Horn continued. “There may be irritability or hypersensitivity in the brain for children with autism that’s being revealed with the clue that they are more photosensitive.”
Large-scale and prospective studies are needed to confirm the trend, according to the investigators. Further studies could reveal the findings’ significance in the pathophysiology of epilepsy in children with ASD.
—Erik Greb
Suggested Reading
Lo C, Shorvon S, Davis M, et al. Genetic linkage analysis of a large family with photoparoxysmal response. Epilepsy Res. 2011 Nov 7; [Epub ahead of print]
Lopes da Silva FH, Harding GF. Transition to seizure in photosensitive epilepsy. Epilepsy Res. 2011;97(3):278-282.
Suggested Reading
Lo C, Shorvon S, Davis M, et al. Genetic linkage analysis of a large family with photoparoxysmal response. Epilepsy Res. 2011 Nov 7; [Epub ahead of print]
Lopes da Silva FH, Harding GF. Transition to seizure in photosensitive epilepsy. Epilepsy Res. 2011;97(3):278-282.
Grand Rounds: Man, 61, With Painful Oral Ulcerations
A 61-year-old man, who had recently emigrated from the Ukraine, presented to his primary care provider with a chief complaint of painful oral lesions and weight loss. The patient described the gradual onset of a severe sore throat and mouth pain three months earlier. Originally, he attributed his symptoms to an upper respiratory infection but became concerned when his symptoms did not resolve.
He reported that the pain had worsened over time and that he was now barely able to swallow solid food or tolerate acidic beverages due to considerable discomfort. His son, who accompanied him to the appointment, had also noted weight loss.
The patient denied any concomitant symptoms, including fever, cough, night sweats, fatigue, lymphadenopathy, abdominal pain, diarrhea, melena, or concomitant rash. His medical history was remarkable only for stage 1 hypertension, which had been well controlled on hydrochlorothiazide 12.5 mg/d for the previous three years. However, the patient had received only minimal preventive health care while living in the Ukraine. His family history was unknown.
One week earlier, the patient had seen a dentist complaining of mouth pain, and was referred to an oral medicine specialist; this specialist, in turn, referred the patient to a primary care nurse practitioner for lab work to confirm the suspected diagnosis of pemphigus vulgaris.
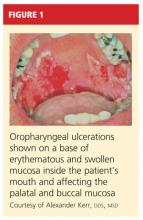
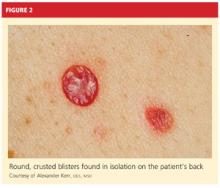
On physical examination, the patient appeared older than his stated age. He was a thin, mildly ill–appearing man, afebrile and normotensive, with heart rate and respirations within normal limits. However, intraoral examination revealed multiple oropharyngeal ulcerations of varying size on a base of erythematous and swollen mucosa on the inside of the man’s cheek and palatal and buccal mucosa (see Figure 1). On his upper back, two round, crusted blisters were noted in isolation (Figure 2). The remaining findings in the physical examination were unremarkable.
Based on the patient’s physical exam findings and clinical guideline recommendations regarding chronic oral ulcerations of unknown etiology,1,2 the patient was scheduled for a cytologic smear to be performed by oral medicine, followed by a gingival biopsy for a direct immunofluorescence test and routine histopathology.3 Unfortunately, due to extensive involvement and concern for possible mucosal shredding, an oral biopsy was not deemed possible.
However, the oral medicine specialist, because he strongly suspected pemphigus vulgaris, recommended testing for circulating autoantibodies against the antigens desmogleins 1 and/or 3 in the epidermis, which are responsible for cellular adhesion. (A positive test result supports, but does not confirm, a diagnosis of pemphigus vulgaris.4)
Additionally, baseline labs were performed for signs of systemic illness, including infection, anemia, and liver and kidney disease. Frequent monitoring was conducted for steroid-induced symptoms of elevated blood sugars; the primary care provider was responsible for monitoring the patient for weight gain and steroid-induced psychosis. The patient was referred to gastroenterology for a colonoscopy to ensure that his weight loss and anorexia were not the result of gastrointestinal malignancy. However, the patient declined this test.
DISCUSSION
Painful oral lesions can have numerous etiologies of varying severity and complexity, including herpes simplex virus infection, aphthae, lichen planus, erythema multiforme, squamous cell and other oral carcinomas, primary HIV infection, lupus, and pemphigus. Differentiating among these conditions requires a careful medical history and complete physical exam.5
Pemphigus vulgaris (PV) is the most common variant of pemphigus, a group of chronic autoimmune diseases that cause blistering and ulceration of the mucous membranes and the skin.6 From the Greek pemphix (bubble), PV is more common in people of Ashkenazi Jewish or Mediterranean descent,6,7 usually occurs in middle-aged and older persons, and occurs about 1.5 times more commonly in women than men.5,7 Until the introduction of systemic steroids, pemphigus was often a fatal disease. Significant mortality still exists, mainly as a result of infection or adverse reactions to medication therapy.5
In patients with PV, flaccid bullae are formed on the skin in a process called acantholysis, in which epidermal cells lose their ability to adhere to one another. This results in rapidly expanding, thin-walled blisters on the oral mucosa, scalp, face, axillae, and groin. The blisters burst easily, leaving irregularly shaped, painful ulcerations.4 Painful oral mucosal membrane erosions are the first presenting sign of PV and often the only sign for an average of five months before other skin lesions develop.3 These lesions are noninfectious.
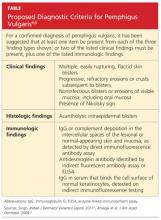
To make a definitive diagnosis of PV, clinical lesions must be present, with a confirmation of histologic findings, acantholysis on biopsy, and a confirmation of autoantibodies present in tissue and/or serum.4 (For proposed detailed diagnostic criteria, see table4,8.)
Initial misdiagnoses, which often lead to delayed or incorrect treatment, usually include aphthous stomatitis, gingivostomatitis, erythema multiforme, erosive lichen planus, herpes simplex virus, and/or oral candidiasis.3
Common Differentials
Herpes simplex virus. Affecting between 15% and 45% of the population, herpes simplex virus (HSV) infection, also known as cold sores, is the most common cause of recurrent oral ulcers.9 HSV is transmitted through direct contact with lesions or via viral shedding. Primary infection, which may occur with flu-like symptoms, causes the sudden onset of multiple clustered vesicles on an erythematous base that quickly ulcerate and crust. Recurrent infections tend to be less severe and are accompanied by minimal systemic symptoms.10
Diagnosis is usually made through history and physical exam. However, diagnostic tests, including Tzanck smears, biopsy, polymerase chain reaction (PCR) assay, and/or viral isolation in culture, are sometimes used to confirm a suspected case.10
Oral lichen planus (OLP). This is a common, chronic, mucocutaneous inflammatory disease of unknown etiology that affects skin and mucous membranes of the mouth, including the buccal mucosa, tongue, and/or gums. These lesions are noninfectious and are an immunologically mediated disease. Stress, anxiety, genetic predisposition, NSAID use, antihypertensive medications (eg, captopril, enalapril, propranolol; considered an oral lichenoid drug reaction), and altered cell-mediated immune response have been considered possible causative factors.11,12 Recent reports suggest an association between hepatitis C virus and OLP.13
Affecting about 4% of the general population, and more predominate in perimenopausal women, OLP lesions appear as white, lacey patches; red, swollen tissues; or open sores, most commonly on the inside of the mouth bilaterally. Patients will present with complaints of burning, roughness, or pain in the mouth, dry mouth, sensitivity to hot or spicy foods, and difficulty swallowing if the throat is involved. Diagnosis is based on history and physical examination and often a confirmatory biopsy. Topical high-potency corticosteroids are generally first-line therapy, with systemic medications such as oral prednisone used to treat severe cases.14,15
Oral candidiasis. Up to 80% of healthy individuals carry Candida albicans in their mouths16; this pathogen accounts for about half of all cases of oral candidiasis (oral thrush). Oral infections occur only with an underlying predisposing condition in the host. Oral thrush presents as creamy white lesions on the oral mucosa; a diagnostic feature is that the plaques can be removed to reveal an erythematous base.16,17
In the chronic form of candidiasis, the mucosal surface is bright red and smooth. When the tongue is involved, it may appear dry, fissured, or cracked. Patients may report a dry mouth, burning pain, and difficulty eating. Infection can be confirmed with periodic acid-Schiff staining of a smear to detect candidal hyphae.9
Use of antifungal creams and lozenges, as well as improved oral hygiene, will often lead to resolution of symptoms.9 Management of any associated underlying conditions, such as diabetes, asthma requiring long-term use of steroid inhalers, or infection with HIV/AIDS, is essential.18
Oral aphthae. Recurrent aphthous ulcers (commonly called canker sores; also referred to as recurrent aphthous stomatitis [RAS]) are a common oral condition. Etiology is unknown and most likely multifactorial, with a strong genetic tendency and multiple predisposing factors, including trauma, stress, food allergies, hormones, and smoking.19 Certain chronic illnesses, including celiac disease, inflammatory bowel disease (IBD), HIV, and neutropenia may also predispose patients to RAS or RAS-like syndromes.
Aphthous ulcers are classified as minor or major. Minor aphthae, which account for 90% of RAS cases, present as single or multiple, small, oval or round ulcers with an erythematous halo on the buccal or labial mucosa or tongue.19 The ulcers last 7 to 10 days and heal spontaneously without scarring.
Diagnosis, based on history and clinical presentation, may include evaluation for systemic causes of oral ulcers. Treatment for both minor and major apthae is palliative, with mainstays including topical corticosteroids, mouth rinses, and, in severe cases, thalidomide, although randomized controlled trials have not shown this agent to be of benefit.9
Treatment for Pemphigus Vulgaris
The outcome goal for management of pemphigus is to achieve and maintain remission. This includes the epithelialization of all skin and mucosal lesions, prevention of relapse, minimization of adverse treatment effects, and successful withdrawal of therapeutic medications.20
The response to treatment varies greatly among patients, as the optimal therapeutic regimen for pemphigus is unknown.20 Systemic glucocorticoids are considered the gold standard of treatment and management, but their use has been associated with several adverse effects, including weight gain and elevated blood sugar levels. Recently, the combination of IV immune globulin and biological therapies (eg, rituximab) that target specific molecules in the inflammatory process have been demonstrated as effective in cases of refractory pemphigus.21,22
PATIENT MANAGEMENT AND OUTCOME
Several referrals were made, including dermatology, for its familiarity with autoimmune diseases of the skin. There, the patient was fully examined and found to have a small truncal lesion compatible with PV. He was referred to an otolaryngologist for a nasal endoscopy to determine the extent of the lesions. They were found to extend far beyond his oral cavity into his esophagus.
Based on a positive enzyme-linked immunosorbent assay (ELISA) for PV antibodies, a cytologic smear with acantholytic cells, and a classic clinical presentation of PV, the patient was started on prednisone 80 mg/d with azathioprine 50 mg/d for the first 14 days.23,24 He responded quickly to these oral medications and underwent a confirmatory oral biopsy within a few weeks.
After several months, the patient was slowly titrated down to lower maintenance doses of prednisone and azathioprine. Now in remission, he continues to receive collaborative management from oral medicine, dermatology, and a nurse practitioner–managed primary care practice. Health care maintenance has included appropriate vaccination and discussion regarding prostate cancer screening, per 2010 guidelines from the US Preventive Services Task Force.25
CONCLUSION
Since the differential diagnosis for pemphigus vulgaris is extensive and the diagnostic criteria are exacting, many affected patients are undiagnosed or misdiagnosed, with a resulting delay in effective treatment. It is important for the primary care clinician to undertake a frequent review of common oral infections, particularly those with similar presentations.
The authors extend their thanks to Alexander Kerr, DDS, MSD, Clinical Associate Professor, Department of Oral and Maxillary Pathology, Radiology and Medicine, New York University College of Dentistry, for the images included in this article and for Dr. Kerr’s clinical expertise and partnership.
REFERENCES
1. Sciubba JJ. Oral mucosal diseases in the office setting. Part II. Oral lichen planus, pemphigus vulgaris, and mucosal pemphigoid. Gen Dent. 2007;55(5):464-476.
2. Muñoz-Corcuera M, Esparza-Gómez G, González-Moles MA, Bascones-Martínez A. Oral ulcers: clinical aspects. A tool for dermatologists. Part II. Chronic ulcers. Clin Exp Dermatol. 2009; 34(4):456-461.
3. Dagistan S, Goregen M, Miloglu O, Cakur B. Oral pemphigus vulgaris: a case report with review of the literature. J Oral Sci. 2008;50(3):359-362.
4. Singh S. Evidence-based treatments for pemphigus vulgaris, pemphigus foliaceus and bullous pemphigoid: a systematic review. Indian J Dermatol Venereol Leprol. 2011;77(4):456-469.
5. Ohta M, Osawa S, Endo H, et al. Pemphigus vulgaris confined to the gingiva: a case report. Int J Dent. 2011;2011:207153. Epub 2011 May 11.
6. Mignona MD, Fortuna G, Leuci S. Oral pemphigus. Minerva Stomatol. 2009;58(10):501-518.
7. Mimouni D, Bar H, Gdalevich M, et al. Pemphigus: analysis of epidemiological factors in 155 patients. J Eur Acad Dermatol Venereol. 2008; 22(10):1232-1235.
8. Amagai M, Ikeda S, Shimizu H, et al. A randomized double-blind trial of intravenous immunoglobulin for pemphigus. J Am Acad Dermatol. 2009;60(4):595-603.
9. Gonsalves WC, Chi AC, Neville BW. Common oral lesions: Part I. Superficial mucosal lesions. Am Fam Physician. 2007;75(4):501-507.
10. Fatahzadeh M, Schwartz R. Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management. J Am Acad Dermatol. 2007;57(5):737-763.
11. Sugerman PB, Savage NW. Oral lichen planus: causes, diagnosis and management. Aust Dent J. 2002;47(4):290-297.
12. Kaomongkolgit R. Oral lichenoid drug reaction associated with antihypertensive and hypoglycemic drugs. J Drugs Dermatol. 2010;9(1):73-75.
13. Petti S, Rabiei M, De Luca M, Scully C. The magnitude of the association between hepatitis C virus infection and oral lichen planus: meta-analysis and case control study. Odontology. 2011;99(2):168-178.
14. Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84(1): 53-60.
15. Thongprasom K, Carrozzo M, Furness S, Lodi G. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011 Jul 6; (7):CD001168.
16. Giannini PJ, Shetty KV. Diagnosis and management of oral candidiasis. Otolaryngol Clin North Am. 2011;44(1):231-240, vii.
17. Lynch DP. Oral candidiasis. History, classification, and clinical presentation. Oral Surg Oral Med Oral Pathol. 1994;78(2):189-193.
18. Williams D, Lewis M. Pathogenesis and treatment of oral candidosis. J Oral Microbiol. 2011 Jan 28;3. doi: 10.3402/jom.v3i0.5771.
19. Scully C, Challacombe SJ. Pemphigus vulgaris: update on etiopathogenesis, oral manifestations, and management. Crit Rev Oral Biol Med. 2002;13(5):397-408.
20. Martin LK, Werth V, Villanueva E, Murrell DF. A systematic review of randomized controlled trials for pemphigus vulgaris and pemphigus foliaceus. J Am Acad Dermatol. 2011;64(5):903-908.
21. Joly P, Mouquet H, Roujeau JC, et al. A single cycle of rituximab for the treatment of severe pemphigus. N Engl J Med. 2007;357(6):545-552.
22. Diaz LA. Rituximab and pemphigus: a therapeutic advance. N Engl J Med. 2007;357(6):605-607.
23. Anstey AV, Wakelin S, Reynolds NJ. Guidelines for prescribing azathioprine in dermatology. Br J Dermatol. 2004;151(6):1123-1132.
24. Chams-Davatchi C, Daneshpazhooh M. Prednisolone dosage in pemphigus vulgaris. J Am Acad Dermatol. 2005;53(3):547.
25. Agency for Healthcare Research and Quality. Guide to Clinical Preventive Services, 2010-2011: recommendations of the US Preventive Services Task Force. AHRQ Publication No. 10-05145, September 2010. www.ahrq.gov/clinic/pocketgd1011/pocketgd1011.pdf. Accessed January 23, 2012.
A 61-year-old man, who had recently emigrated from the Ukraine, presented to his primary care provider with a chief complaint of painful oral lesions and weight loss. The patient described the gradual onset of a severe sore throat and mouth pain three months earlier. Originally, he attributed his symptoms to an upper respiratory infection but became concerned when his symptoms did not resolve.
He reported that the pain had worsened over time and that he was now barely able to swallow solid food or tolerate acidic beverages due to considerable discomfort. His son, who accompanied him to the appointment, had also noted weight loss.
The patient denied any concomitant symptoms, including fever, cough, night sweats, fatigue, lymphadenopathy, abdominal pain, diarrhea, melena, or concomitant rash. His medical history was remarkable only for stage 1 hypertension, which had been well controlled on hydrochlorothiazide 12.5 mg/d for the previous three years. However, the patient had received only minimal preventive health care while living in the Ukraine. His family history was unknown.
One week earlier, the patient had seen a dentist complaining of mouth pain, and was referred to an oral medicine specialist; this specialist, in turn, referred the patient to a primary care nurse practitioner for lab work to confirm the suspected diagnosis of pemphigus vulgaris.
On physical examination, the patient appeared older than his stated age. He was a thin, mildly ill–appearing man, afebrile and normotensive, with heart rate and respirations within normal limits. However, intraoral examination revealed multiple oropharyngeal ulcerations of varying size on a base of erythematous and swollen mucosa on the inside of the man’s cheek and palatal and buccal mucosa (see Figure 1). On his upper back, two round, crusted blisters were noted in isolation (Figure 2). The remaining findings in the physical examination were unremarkable.
Based on the patient’s physical exam findings and clinical guideline recommendations regarding chronic oral ulcerations of unknown etiology,1,2 the patient was scheduled for a cytologic smear to be performed by oral medicine, followed by a gingival biopsy for a direct immunofluorescence test and routine histopathology.3 Unfortunately, due to extensive involvement and concern for possible mucosal shredding, an oral biopsy was not deemed possible.
However, the oral medicine specialist, because he strongly suspected pemphigus vulgaris, recommended testing for circulating autoantibodies against the antigens desmogleins 1 and/or 3 in the epidermis, which are responsible for cellular adhesion. (A positive test result supports, but does not confirm, a diagnosis of pemphigus vulgaris.4)
Additionally, baseline labs were performed for signs of systemic illness, including infection, anemia, and liver and kidney disease. Frequent monitoring was conducted for steroid-induced symptoms of elevated blood sugars; the primary care provider was responsible for monitoring the patient for weight gain and steroid-induced psychosis. The patient was referred to gastroenterology for a colonoscopy to ensure that his weight loss and anorexia were not the result of gastrointestinal malignancy. However, the patient declined this test.
DISCUSSION
Painful oral lesions can have numerous etiologies of varying severity and complexity, including herpes simplex virus infection, aphthae, lichen planus, erythema multiforme, squamous cell and other oral carcinomas, primary HIV infection, lupus, and pemphigus. Differentiating among these conditions requires a careful medical history and complete physical exam.5
Pemphigus vulgaris (PV) is the most common variant of pemphigus, a group of chronic autoimmune diseases that cause blistering and ulceration of the mucous membranes and the skin.6 From the Greek pemphix (bubble), PV is more common in people of Ashkenazi Jewish or Mediterranean descent,6,7 usually occurs in middle-aged and older persons, and occurs about 1.5 times more commonly in women than men.5,7 Until the introduction of systemic steroids, pemphigus was often a fatal disease. Significant mortality still exists, mainly as a result of infection or adverse reactions to medication therapy.5
In patients with PV, flaccid bullae are formed on the skin in a process called acantholysis, in which epidermal cells lose their ability to adhere to one another. This results in rapidly expanding, thin-walled blisters on the oral mucosa, scalp, face, axillae, and groin. The blisters burst easily, leaving irregularly shaped, painful ulcerations.4 Painful oral mucosal membrane erosions are the first presenting sign of PV and often the only sign for an average of five months before other skin lesions develop.3 These lesions are noninfectious.
To make a definitive diagnosis of PV, clinical lesions must be present, with a confirmation of histologic findings, acantholysis on biopsy, and a confirmation of autoantibodies present in tissue and/or serum.4 (For proposed detailed diagnostic criteria, see table4,8.)
Initial misdiagnoses, which often lead to delayed or incorrect treatment, usually include aphthous stomatitis, gingivostomatitis, erythema multiforme, erosive lichen planus, herpes simplex virus, and/or oral candidiasis.3
Common Differentials
Herpes simplex virus. Affecting between 15% and 45% of the population, herpes simplex virus (HSV) infection, also known as cold sores, is the most common cause of recurrent oral ulcers.9 HSV is transmitted through direct contact with lesions or via viral shedding. Primary infection, which may occur with flu-like symptoms, causes the sudden onset of multiple clustered vesicles on an erythematous base that quickly ulcerate and crust. Recurrent infections tend to be less severe and are accompanied by minimal systemic symptoms.10
Diagnosis is usually made through history and physical exam. However, diagnostic tests, including Tzanck smears, biopsy, polymerase chain reaction (PCR) assay, and/or viral isolation in culture, are sometimes used to confirm a suspected case.10
Oral lichen planus (OLP). This is a common, chronic, mucocutaneous inflammatory disease of unknown etiology that affects skin and mucous membranes of the mouth, including the buccal mucosa, tongue, and/or gums. These lesions are noninfectious and are an immunologically mediated disease. Stress, anxiety, genetic predisposition, NSAID use, antihypertensive medications (eg, captopril, enalapril, propranolol; considered an oral lichenoid drug reaction), and altered cell-mediated immune response have been considered possible causative factors.11,12 Recent reports suggest an association between hepatitis C virus and OLP.13
Affecting about 4% of the general population, and more predominate in perimenopausal women, OLP lesions appear as white, lacey patches; red, swollen tissues; or open sores, most commonly on the inside of the mouth bilaterally. Patients will present with complaints of burning, roughness, or pain in the mouth, dry mouth, sensitivity to hot or spicy foods, and difficulty swallowing if the throat is involved. Diagnosis is based on history and physical examination and often a confirmatory biopsy. Topical high-potency corticosteroids are generally first-line therapy, with systemic medications such as oral prednisone used to treat severe cases.14,15
Oral candidiasis. Up to 80% of healthy individuals carry Candida albicans in their mouths16; this pathogen accounts for about half of all cases of oral candidiasis (oral thrush). Oral infections occur only with an underlying predisposing condition in the host. Oral thrush presents as creamy white lesions on the oral mucosa; a diagnostic feature is that the plaques can be removed to reveal an erythematous base.16,17
In the chronic form of candidiasis, the mucosal surface is bright red and smooth. When the tongue is involved, it may appear dry, fissured, or cracked. Patients may report a dry mouth, burning pain, and difficulty eating. Infection can be confirmed with periodic acid-Schiff staining of a smear to detect candidal hyphae.9
Use of antifungal creams and lozenges, as well as improved oral hygiene, will often lead to resolution of symptoms.9 Management of any associated underlying conditions, such as diabetes, asthma requiring long-term use of steroid inhalers, or infection with HIV/AIDS, is essential.18
Oral aphthae. Recurrent aphthous ulcers (commonly called canker sores; also referred to as recurrent aphthous stomatitis [RAS]) are a common oral condition. Etiology is unknown and most likely multifactorial, with a strong genetic tendency and multiple predisposing factors, including trauma, stress, food allergies, hormones, and smoking.19 Certain chronic illnesses, including celiac disease, inflammatory bowel disease (IBD), HIV, and neutropenia may also predispose patients to RAS or RAS-like syndromes.
Aphthous ulcers are classified as minor or major. Minor aphthae, which account for 90% of RAS cases, present as single or multiple, small, oval or round ulcers with an erythematous halo on the buccal or labial mucosa or tongue.19 The ulcers last 7 to 10 days and heal spontaneously without scarring.
Diagnosis, based on history and clinical presentation, may include evaluation for systemic causes of oral ulcers. Treatment for both minor and major apthae is palliative, with mainstays including topical corticosteroids, mouth rinses, and, in severe cases, thalidomide, although randomized controlled trials have not shown this agent to be of benefit.9
Treatment for Pemphigus Vulgaris
The outcome goal for management of pemphigus is to achieve and maintain remission. This includes the epithelialization of all skin and mucosal lesions, prevention of relapse, minimization of adverse treatment effects, and successful withdrawal of therapeutic medications.20
The response to treatment varies greatly among patients, as the optimal therapeutic regimen for pemphigus is unknown.20 Systemic glucocorticoids are considered the gold standard of treatment and management, but their use has been associated with several adverse effects, including weight gain and elevated blood sugar levels. Recently, the combination of IV immune globulin and biological therapies (eg, rituximab) that target specific molecules in the inflammatory process have been demonstrated as effective in cases of refractory pemphigus.21,22
PATIENT MANAGEMENT AND OUTCOME
Several referrals were made, including dermatology, for its familiarity with autoimmune diseases of the skin. There, the patient was fully examined and found to have a small truncal lesion compatible with PV. He was referred to an otolaryngologist for a nasal endoscopy to determine the extent of the lesions. They were found to extend far beyond his oral cavity into his esophagus.
Based on a positive enzyme-linked immunosorbent assay (ELISA) for PV antibodies, a cytologic smear with acantholytic cells, and a classic clinical presentation of PV, the patient was started on prednisone 80 mg/d with azathioprine 50 mg/d for the first 14 days.23,24 He responded quickly to these oral medications and underwent a confirmatory oral biopsy within a few weeks.
After several months, the patient was slowly titrated down to lower maintenance doses of prednisone and azathioprine. Now in remission, he continues to receive collaborative management from oral medicine, dermatology, and a nurse practitioner–managed primary care practice. Health care maintenance has included appropriate vaccination and discussion regarding prostate cancer screening, per 2010 guidelines from the US Preventive Services Task Force.25
CONCLUSION
Since the differential diagnosis for pemphigus vulgaris is extensive and the diagnostic criteria are exacting, many affected patients are undiagnosed or misdiagnosed, with a resulting delay in effective treatment. It is important for the primary care clinician to undertake a frequent review of common oral infections, particularly those with similar presentations.
The authors extend their thanks to Alexander Kerr, DDS, MSD, Clinical Associate Professor, Department of Oral and Maxillary Pathology, Radiology and Medicine, New York University College of Dentistry, for the images included in this article and for Dr. Kerr’s clinical expertise and partnership.
REFERENCES
1. Sciubba JJ. Oral mucosal diseases in the office setting. Part II. Oral lichen planus, pemphigus vulgaris, and mucosal pemphigoid. Gen Dent. 2007;55(5):464-476.
2. Muñoz-Corcuera M, Esparza-Gómez G, González-Moles MA, Bascones-Martínez A. Oral ulcers: clinical aspects. A tool for dermatologists. Part II. Chronic ulcers. Clin Exp Dermatol. 2009; 34(4):456-461.
3. Dagistan S, Goregen M, Miloglu O, Cakur B. Oral pemphigus vulgaris: a case report with review of the literature. J Oral Sci. 2008;50(3):359-362.
4. Singh S. Evidence-based treatments for pemphigus vulgaris, pemphigus foliaceus and bullous pemphigoid: a systematic review. Indian J Dermatol Venereol Leprol. 2011;77(4):456-469.
5. Ohta M, Osawa S, Endo H, et al. Pemphigus vulgaris confined to the gingiva: a case report. Int J Dent. 2011;2011:207153. Epub 2011 May 11.
6. Mignona MD, Fortuna G, Leuci S. Oral pemphigus. Minerva Stomatol. 2009;58(10):501-518.
7. Mimouni D, Bar H, Gdalevich M, et al. Pemphigus: analysis of epidemiological factors in 155 patients. J Eur Acad Dermatol Venereol. 2008; 22(10):1232-1235.
8. Amagai M, Ikeda S, Shimizu H, et al. A randomized double-blind trial of intravenous immunoglobulin for pemphigus. J Am Acad Dermatol. 2009;60(4):595-603.
9. Gonsalves WC, Chi AC, Neville BW. Common oral lesions: Part I. Superficial mucosal lesions. Am Fam Physician. 2007;75(4):501-507.
10. Fatahzadeh M, Schwartz R. Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management. J Am Acad Dermatol. 2007;57(5):737-763.
11. Sugerman PB, Savage NW. Oral lichen planus: causes, diagnosis and management. Aust Dent J. 2002;47(4):290-297.
12. Kaomongkolgit R. Oral lichenoid drug reaction associated with antihypertensive and hypoglycemic drugs. J Drugs Dermatol. 2010;9(1):73-75.
13. Petti S, Rabiei M, De Luca M, Scully C. The magnitude of the association between hepatitis C virus infection and oral lichen planus: meta-analysis and case control study. Odontology. 2011;99(2):168-178.
14. Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84(1): 53-60.
15. Thongprasom K, Carrozzo M, Furness S, Lodi G. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011 Jul 6; (7):CD001168.
16. Giannini PJ, Shetty KV. Diagnosis and management of oral candidiasis. Otolaryngol Clin North Am. 2011;44(1):231-240, vii.
17. Lynch DP. Oral candidiasis. History, classification, and clinical presentation. Oral Surg Oral Med Oral Pathol. 1994;78(2):189-193.
18. Williams D, Lewis M. Pathogenesis and treatment of oral candidosis. J Oral Microbiol. 2011 Jan 28;3. doi: 10.3402/jom.v3i0.5771.
19. Scully C, Challacombe SJ. Pemphigus vulgaris: update on etiopathogenesis, oral manifestations, and management. Crit Rev Oral Biol Med. 2002;13(5):397-408.
20. Martin LK, Werth V, Villanueva E, Murrell DF. A systematic review of randomized controlled trials for pemphigus vulgaris and pemphigus foliaceus. J Am Acad Dermatol. 2011;64(5):903-908.
21. Joly P, Mouquet H, Roujeau JC, et al. A single cycle of rituximab for the treatment of severe pemphigus. N Engl J Med. 2007;357(6):545-552.
22. Diaz LA. Rituximab and pemphigus: a therapeutic advance. N Engl J Med. 2007;357(6):605-607.
23. Anstey AV, Wakelin S, Reynolds NJ. Guidelines for prescribing azathioprine in dermatology. Br J Dermatol. 2004;151(6):1123-1132.
24. Chams-Davatchi C, Daneshpazhooh M. Prednisolone dosage in pemphigus vulgaris. J Am Acad Dermatol. 2005;53(3):547.
25. Agency for Healthcare Research and Quality. Guide to Clinical Preventive Services, 2010-2011: recommendations of the US Preventive Services Task Force. AHRQ Publication No. 10-05145, September 2010. www.ahrq.gov/clinic/pocketgd1011/pocketgd1011.pdf. Accessed January 23, 2012.
A 61-year-old man, who had recently emigrated from the Ukraine, presented to his primary care provider with a chief complaint of painful oral lesions and weight loss. The patient described the gradual onset of a severe sore throat and mouth pain three months earlier. Originally, he attributed his symptoms to an upper respiratory infection but became concerned when his symptoms did not resolve.
He reported that the pain had worsened over time and that he was now barely able to swallow solid food or tolerate acidic beverages due to considerable discomfort. His son, who accompanied him to the appointment, had also noted weight loss.
The patient denied any concomitant symptoms, including fever, cough, night sweats, fatigue, lymphadenopathy, abdominal pain, diarrhea, melena, or concomitant rash. His medical history was remarkable only for stage 1 hypertension, which had been well controlled on hydrochlorothiazide 12.5 mg/d for the previous three years. However, the patient had received only minimal preventive health care while living in the Ukraine. His family history was unknown.
One week earlier, the patient had seen a dentist complaining of mouth pain, and was referred to an oral medicine specialist; this specialist, in turn, referred the patient to a primary care nurse practitioner for lab work to confirm the suspected diagnosis of pemphigus vulgaris.
On physical examination, the patient appeared older than his stated age. He was a thin, mildly ill–appearing man, afebrile and normotensive, with heart rate and respirations within normal limits. However, intraoral examination revealed multiple oropharyngeal ulcerations of varying size on a base of erythematous and swollen mucosa on the inside of the man’s cheek and palatal and buccal mucosa (see Figure 1). On his upper back, two round, crusted blisters were noted in isolation (Figure 2). The remaining findings in the physical examination were unremarkable.
Based on the patient’s physical exam findings and clinical guideline recommendations regarding chronic oral ulcerations of unknown etiology,1,2 the patient was scheduled for a cytologic smear to be performed by oral medicine, followed by a gingival biopsy for a direct immunofluorescence test and routine histopathology.3 Unfortunately, due to extensive involvement and concern for possible mucosal shredding, an oral biopsy was not deemed possible.
However, the oral medicine specialist, because he strongly suspected pemphigus vulgaris, recommended testing for circulating autoantibodies against the antigens desmogleins 1 and/or 3 in the epidermis, which are responsible for cellular adhesion. (A positive test result supports, but does not confirm, a diagnosis of pemphigus vulgaris.4)
Additionally, baseline labs were performed for signs of systemic illness, including infection, anemia, and liver and kidney disease. Frequent monitoring was conducted for steroid-induced symptoms of elevated blood sugars; the primary care provider was responsible for monitoring the patient for weight gain and steroid-induced psychosis. The patient was referred to gastroenterology for a colonoscopy to ensure that his weight loss and anorexia were not the result of gastrointestinal malignancy. However, the patient declined this test.
DISCUSSION
Painful oral lesions can have numerous etiologies of varying severity and complexity, including herpes simplex virus infection, aphthae, lichen planus, erythema multiforme, squamous cell and other oral carcinomas, primary HIV infection, lupus, and pemphigus. Differentiating among these conditions requires a careful medical history and complete physical exam.5
Pemphigus vulgaris (PV) is the most common variant of pemphigus, a group of chronic autoimmune diseases that cause blistering and ulceration of the mucous membranes and the skin.6 From the Greek pemphix (bubble), PV is more common in people of Ashkenazi Jewish or Mediterranean descent,6,7 usually occurs in middle-aged and older persons, and occurs about 1.5 times more commonly in women than men.5,7 Until the introduction of systemic steroids, pemphigus was often a fatal disease. Significant mortality still exists, mainly as a result of infection or adverse reactions to medication therapy.5
In patients with PV, flaccid bullae are formed on the skin in a process called acantholysis, in which epidermal cells lose their ability to adhere to one another. This results in rapidly expanding, thin-walled blisters on the oral mucosa, scalp, face, axillae, and groin. The blisters burst easily, leaving irregularly shaped, painful ulcerations.4 Painful oral mucosal membrane erosions are the first presenting sign of PV and often the only sign for an average of five months before other skin lesions develop.3 These lesions are noninfectious.
To make a definitive diagnosis of PV, clinical lesions must be present, with a confirmation of histologic findings, acantholysis on biopsy, and a confirmation of autoantibodies present in tissue and/or serum.4 (For proposed detailed diagnostic criteria, see table4,8.)
Initial misdiagnoses, which often lead to delayed or incorrect treatment, usually include aphthous stomatitis, gingivostomatitis, erythema multiforme, erosive lichen planus, herpes simplex virus, and/or oral candidiasis.3
Common Differentials
Herpes simplex virus. Affecting between 15% and 45% of the population, herpes simplex virus (HSV) infection, also known as cold sores, is the most common cause of recurrent oral ulcers.9 HSV is transmitted through direct contact with lesions or via viral shedding. Primary infection, which may occur with flu-like symptoms, causes the sudden onset of multiple clustered vesicles on an erythematous base that quickly ulcerate and crust. Recurrent infections tend to be less severe and are accompanied by minimal systemic symptoms.10
Diagnosis is usually made through history and physical exam. However, diagnostic tests, including Tzanck smears, biopsy, polymerase chain reaction (PCR) assay, and/or viral isolation in culture, are sometimes used to confirm a suspected case.10
Oral lichen planus (OLP). This is a common, chronic, mucocutaneous inflammatory disease of unknown etiology that affects skin and mucous membranes of the mouth, including the buccal mucosa, tongue, and/or gums. These lesions are noninfectious and are an immunologically mediated disease. Stress, anxiety, genetic predisposition, NSAID use, antihypertensive medications (eg, captopril, enalapril, propranolol; considered an oral lichenoid drug reaction), and altered cell-mediated immune response have been considered possible causative factors.11,12 Recent reports suggest an association between hepatitis C virus and OLP.13
Affecting about 4% of the general population, and more predominate in perimenopausal women, OLP lesions appear as white, lacey patches; red, swollen tissues; or open sores, most commonly on the inside of the mouth bilaterally. Patients will present with complaints of burning, roughness, or pain in the mouth, dry mouth, sensitivity to hot or spicy foods, and difficulty swallowing if the throat is involved. Diagnosis is based on history and physical examination and often a confirmatory biopsy. Topical high-potency corticosteroids are generally first-line therapy, with systemic medications such as oral prednisone used to treat severe cases.14,15
Oral candidiasis. Up to 80% of healthy individuals carry Candida albicans in their mouths16; this pathogen accounts for about half of all cases of oral candidiasis (oral thrush). Oral infections occur only with an underlying predisposing condition in the host. Oral thrush presents as creamy white lesions on the oral mucosa; a diagnostic feature is that the plaques can be removed to reveal an erythematous base.16,17
In the chronic form of candidiasis, the mucosal surface is bright red and smooth. When the tongue is involved, it may appear dry, fissured, or cracked. Patients may report a dry mouth, burning pain, and difficulty eating. Infection can be confirmed with periodic acid-Schiff staining of a smear to detect candidal hyphae.9
Use of antifungal creams and lozenges, as well as improved oral hygiene, will often lead to resolution of symptoms.9 Management of any associated underlying conditions, such as diabetes, asthma requiring long-term use of steroid inhalers, or infection with HIV/AIDS, is essential.18
Oral aphthae. Recurrent aphthous ulcers (commonly called canker sores; also referred to as recurrent aphthous stomatitis [RAS]) are a common oral condition. Etiology is unknown and most likely multifactorial, with a strong genetic tendency and multiple predisposing factors, including trauma, stress, food allergies, hormones, and smoking.19 Certain chronic illnesses, including celiac disease, inflammatory bowel disease (IBD), HIV, and neutropenia may also predispose patients to RAS or RAS-like syndromes.
Aphthous ulcers are classified as minor or major. Minor aphthae, which account for 90% of RAS cases, present as single or multiple, small, oval or round ulcers with an erythematous halo on the buccal or labial mucosa or tongue.19 The ulcers last 7 to 10 days and heal spontaneously without scarring.
Diagnosis, based on history and clinical presentation, may include evaluation for systemic causes of oral ulcers. Treatment for both minor and major apthae is palliative, with mainstays including topical corticosteroids, mouth rinses, and, in severe cases, thalidomide, although randomized controlled trials have not shown this agent to be of benefit.9
Treatment for Pemphigus Vulgaris
The outcome goal for management of pemphigus is to achieve and maintain remission. This includes the epithelialization of all skin and mucosal lesions, prevention of relapse, minimization of adverse treatment effects, and successful withdrawal of therapeutic medications.20
The response to treatment varies greatly among patients, as the optimal therapeutic regimen for pemphigus is unknown.20 Systemic glucocorticoids are considered the gold standard of treatment and management, but their use has been associated with several adverse effects, including weight gain and elevated blood sugar levels. Recently, the combination of IV immune globulin and biological therapies (eg, rituximab) that target specific molecules in the inflammatory process have been demonstrated as effective in cases of refractory pemphigus.21,22
PATIENT MANAGEMENT AND OUTCOME
Several referrals were made, including dermatology, for its familiarity with autoimmune diseases of the skin. There, the patient was fully examined and found to have a small truncal lesion compatible with PV. He was referred to an otolaryngologist for a nasal endoscopy to determine the extent of the lesions. They were found to extend far beyond his oral cavity into his esophagus.
Based on a positive enzyme-linked immunosorbent assay (ELISA) for PV antibodies, a cytologic smear with acantholytic cells, and a classic clinical presentation of PV, the patient was started on prednisone 80 mg/d with azathioprine 50 mg/d for the first 14 days.23,24 He responded quickly to these oral medications and underwent a confirmatory oral biopsy within a few weeks.
After several months, the patient was slowly titrated down to lower maintenance doses of prednisone and azathioprine. Now in remission, he continues to receive collaborative management from oral medicine, dermatology, and a nurse practitioner–managed primary care practice. Health care maintenance has included appropriate vaccination and discussion regarding prostate cancer screening, per 2010 guidelines from the US Preventive Services Task Force.25
CONCLUSION
Since the differential diagnosis for pemphigus vulgaris is extensive and the diagnostic criteria are exacting, many affected patients are undiagnosed or misdiagnosed, with a resulting delay in effective treatment. It is important for the primary care clinician to undertake a frequent review of common oral infections, particularly those with similar presentations.
The authors extend their thanks to Alexander Kerr, DDS, MSD, Clinical Associate Professor, Department of Oral and Maxillary Pathology, Radiology and Medicine, New York University College of Dentistry, for the images included in this article and for Dr. Kerr’s clinical expertise and partnership.
REFERENCES
1. Sciubba JJ. Oral mucosal diseases in the office setting. Part II. Oral lichen planus, pemphigus vulgaris, and mucosal pemphigoid. Gen Dent. 2007;55(5):464-476.
2. Muñoz-Corcuera M, Esparza-Gómez G, González-Moles MA, Bascones-Martínez A. Oral ulcers: clinical aspects. A tool for dermatologists. Part II. Chronic ulcers. Clin Exp Dermatol. 2009; 34(4):456-461.
3. Dagistan S, Goregen M, Miloglu O, Cakur B. Oral pemphigus vulgaris: a case report with review of the literature. J Oral Sci. 2008;50(3):359-362.
4. Singh S. Evidence-based treatments for pemphigus vulgaris, pemphigus foliaceus and bullous pemphigoid: a systematic review. Indian J Dermatol Venereol Leprol. 2011;77(4):456-469.
5. Ohta M, Osawa S, Endo H, et al. Pemphigus vulgaris confined to the gingiva: a case report. Int J Dent. 2011;2011:207153. Epub 2011 May 11.
6. Mignona MD, Fortuna G, Leuci S. Oral pemphigus. Minerva Stomatol. 2009;58(10):501-518.
7. Mimouni D, Bar H, Gdalevich M, et al. Pemphigus: analysis of epidemiological factors in 155 patients. J Eur Acad Dermatol Venereol. 2008; 22(10):1232-1235.
8. Amagai M, Ikeda S, Shimizu H, et al. A randomized double-blind trial of intravenous immunoglobulin for pemphigus. J Am Acad Dermatol. 2009;60(4):595-603.
9. Gonsalves WC, Chi AC, Neville BW. Common oral lesions: Part I. Superficial mucosal lesions. Am Fam Physician. 2007;75(4):501-507.
10. Fatahzadeh M, Schwartz R. Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management. J Am Acad Dermatol. 2007;57(5):737-763.
11. Sugerman PB, Savage NW. Oral lichen planus: causes, diagnosis and management. Aust Dent J. 2002;47(4):290-297.
12. Kaomongkolgit R. Oral lichenoid drug reaction associated with antihypertensive and hypoglycemic drugs. J Drugs Dermatol. 2010;9(1):73-75.
13. Petti S, Rabiei M, De Luca M, Scully C. The magnitude of the association between hepatitis C virus infection and oral lichen planus: meta-analysis and case control study. Odontology. 2011;99(2):168-178.
14. Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84(1): 53-60.
15. Thongprasom K, Carrozzo M, Furness S, Lodi G. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011 Jul 6; (7):CD001168.
16. Giannini PJ, Shetty KV. Diagnosis and management of oral candidiasis. Otolaryngol Clin North Am. 2011;44(1):231-240, vii.
17. Lynch DP. Oral candidiasis. History, classification, and clinical presentation. Oral Surg Oral Med Oral Pathol. 1994;78(2):189-193.
18. Williams D, Lewis M. Pathogenesis and treatment of oral candidosis. J Oral Microbiol. 2011 Jan 28;3. doi: 10.3402/jom.v3i0.5771.
19. Scully C, Challacombe SJ. Pemphigus vulgaris: update on etiopathogenesis, oral manifestations, and management. Crit Rev Oral Biol Med. 2002;13(5):397-408.
20. Martin LK, Werth V, Villanueva E, Murrell DF. A systematic review of randomized controlled trials for pemphigus vulgaris and pemphigus foliaceus. J Am Acad Dermatol. 2011;64(5):903-908.
21. Joly P, Mouquet H, Roujeau JC, et al. A single cycle of rituximab for the treatment of severe pemphigus. N Engl J Med. 2007;357(6):545-552.
22. Diaz LA. Rituximab and pemphigus: a therapeutic advance. N Engl J Med. 2007;357(6):605-607.
23. Anstey AV, Wakelin S, Reynolds NJ. Guidelines for prescribing azathioprine in dermatology. Br J Dermatol. 2004;151(6):1123-1132.
24. Chams-Davatchi C, Daneshpazhooh M. Prednisolone dosage in pemphigus vulgaris. J Am Acad Dermatol. 2005;53(3):547.
25. Agency for Healthcare Research and Quality. Guide to Clinical Preventive Services, 2010-2011: recommendations of the US Preventive Services Task Force. AHRQ Publication No. 10-05145, September 2010. www.ahrq.gov/clinic/pocketgd1011/pocketgd1011.pdf. Accessed January 23, 2012.