User login
ACP Weighs in on Medical Test Overuse
Hospitalists should view a new American College of Physicians (ACP) list of three dozen commonly overused clinical tests that offer lower value as an opportunity to review their use of screening and diagnostic tools, according to one of the list's authors.
Jeff Wiese, MD, SFHM, professor of medicine and residency program director at Tulane University Health Sciences Center in New Orleans, coauthored research published in January in the Annals of Internal Medicine that he says represents clinical situations in which tests have historically been administered but, upon further review, do not reflect "high-value care."
"Nobody is trying to waste money," says Dr. Wiese, a former president of SHM who adds that physicians over time might learn that tests that once offered higher value may no longer do so. "Only by critically reviewing our habits are we able to make the necessary adjustments to ensure we are delivering high-value, cost-conscious care."
ACP convened an ad hoc workgroup of internal-medicine specialists to review lower-value tests; the list that the team came up with includes:
- Repeat screening ultrasonography for abdominal aortic aneurysm following a negative study;
- Screening for prostate cancer in men older than 75 or with a life expectancy of less than 10 years; and
- Performing serologic testing for suspected early Lyme disease.
Dr. Wiese emphasizes that decisions regarding "cost-conscious care" must be interpreted in the context of the specific patient in front of them.
"There is no decision rule that applies to all patients," he says. "The tests addressed in the article are examples of tests that do not routinely offer high value, but this is not to say that there are not specific circumstances when they might be useful."
Hospitalists should view a new American College of Physicians (ACP) list of three dozen commonly overused clinical tests that offer lower value as an opportunity to review their use of screening and diagnostic tools, according to one of the list's authors.
Jeff Wiese, MD, SFHM, professor of medicine and residency program director at Tulane University Health Sciences Center in New Orleans, coauthored research published in January in the Annals of Internal Medicine that he says represents clinical situations in which tests have historically been administered but, upon further review, do not reflect "high-value care."
"Nobody is trying to waste money," says Dr. Wiese, a former president of SHM who adds that physicians over time might learn that tests that once offered higher value may no longer do so. "Only by critically reviewing our habits are we able to make the necessary adjustments to ensure we are delivering high-value, cost-conscious care."
ACP convened an ad hoc workgroup of internal-medicine specialists to review lower-value tests; the list that the team came up with includes:
- Repeat screening ultrasonography for abdominal aortic aneurysm following a negative study;
- Screening for prostate cancer in men older than 75 or with a life expectancy of less than 10 years; and
- Performing serologic testing for suspected early Lyme disease.
Dr. Wiese emphasizes that decisions regarding "cost-conscious care" must be interpreted in the context of the specific patient in front of them.
"There is no decision rule that applies to all patients," he says. "The tests addressed in the article are examples of tests that do not routinely offer high value, but this is not to say that there are not specific circumstances when they might be useful."
Hospitalists should view a new American College of Physicians (ACP) list of three dozen commonly overused clinical tests that offer lower value as an opportunity to review their use of screening and diagnostic tools, according to one of the list's authors.
Jeff Wiese, MD, SFHM, professor of medicine and residency program director at Tulane University Health Sciences Center in New Orleans, coauthored research published in January in the Annals of Internal Medicine that he says represents clinical situations in which tests have historically been administered but, upon further review, do not reflect "high-value care."
"Nobody is trying to waste money," says Dr. Wiese, a former president of SHM who adds that physicians over time might learn that tests that once offered higher value may no longer do so. "Only by critically reviewing our habits are we able to make the necessary adjustments to ensure we are delivering high-value, cost-conscious care."
ACP convened an ad hoc workgroup of internal-medicine specialists to review lower-value tests; the list that the team came up with includes:
- Repeat screening ultrasonography for abdominal aortic aneurysm following a negative study;
- Screening for prostate cancer in men older than 75 or with a life expectancy of less than 10 years; and
- Performing serologic testing for suspected early Lyme disease.
Dr. Wiese emphasizes that decisions regarding "cost-conscious care" must be interpreted in the context of the specific patient in front of them.
"There is no decision rule that applies to all patients," he says. "The tests addressed in the article are examples of tests that do not routinely offer high value, but this is not to say that there are not specific circumstances when they might be useful."
In the Literature: Research You Need to Know
Clinical question: What is the current level of continuity of care, and what factors affect continuity of care in the hospital setting? Has this changed with increasing use of hospitalists and limits on residency duty hours?
Background: Outpatient continuity of care leads to lower costs, better quality of life, and less emergency room use. Recent changes in residency hours have increased hand-offs and decreased inpatient continuity, but to what extent is unknown.
Study design: Retrospective cohort of 5% of Medicare claims data (530,000 patients in all) from 1996 to 2006, including patients admitted for COPD, congestive heart failure, or pneumonia who were cared for by a general internist or family practitioner.
Setting: Nationwide in the U.S.
Synopsis: The authors defined patients as having a primary-care physician (PCP) if they had three billed visits with the PCP in the last year, hospitalists as those who derived at least 90% of their Medicare claims from inpatient billing, and other generalists as those who met criteria as a generalist but did not fit these categories. Inpatient continuity of care decreased to 59% of patients seeing a single physician in the hospital in 2006 from 71% in 1996, with an accompanying decrease in the length of stay of one full day. There were large variations by geographic region, population size, and hospital characteristics. Patients cared for by hospitalists had slightly better continuity of care than those cared for by nonhospitalist generalists, and those who were cared for by both hospitalists and nonhospitalist generalists had the worst continuity of care. Having a PCP was associated with increased discontinuity of care as an inpatient, perhaps because of individual members of a practice rounding on all of the practice’s inpatients.
Bottom line: Patients were 5% less likely per year between 1996 and 2006 to have a single physician be their primary caregiver in the hospital, but the rise of the hospitalist movement does not seem to be the cause.
Citation: Fletcher KE, Sharma G, Zhang D, Kuo YF, Goodwin JS. Trends in inpatient continuity of care for a cohort of Medicare patients 1996-2006. J Hosp Med. 2011;6:441-447.
For more physician reviews of HM-relevant literature, visit our website.
Clinical question: What is the current level of continuity of care, and what factors affect continuity of care in the hospital setting? Has this changed with increasing use of hospitalists and limits on residency duty hours?
Background: Outpatient continuity of care leads to lower costs, better quality of life, and less emergency room use. Recent changes in residency hours have increased hand-offs and decreased inpatient continuity, but to what extent is unknown.
Study design: Retrospective cohort of 5% of Medicare claims data (530,000 patients in all) from 1996 to 2006, including patients admitted for COPD, congestive heart failure, or pneumonia who were cared for by a general internist or family practitioner.
Setting: Nationwide in the U.S.
Synopsis: The authors defined patients as having a primary-care physician (PCP) if they had three billed visits with the PCP in the last year, hospitalists as those who derived at least 90% of their Medicare claims from inpatient billing, and other generalists as those who met criteria as a generalist but did not fit these categories. Inpatient continuity of care decreased to 59% of patients seeing a single physician in the hospital in 2006 from 71% in 1996, with an accompanying decrease in the length of stay of one full day. There were large variations by geographic region, population size, and hospital characteristics. Patients cared for by hospitalists had slightly better continuity of care than those cared for by nonhospitalist generalists, and those who were cared for by both hospitalists and nonhospitalist generalists had the worst continuity of care. Having a PCP was associated with increased discontinuity of care as an inpatient, perhaps because of individual members of a practice rounding on all of the practice’s inpatients.
Bottom line: Patients were 5% less likely per year between 1996 and 2006 to have a single physician be their primary caregiver in the hospital, but the rise of the hospitalist movement does not seem to be the cause.
Citation: Fletcher KE, Sharma G, Zhang D, Kuo YF, Goodwin JS. Trends in inpatient continuity of care for a cohort of Medicare patients 1996-2006. J Hosp Med. 2011;6:441-447.
For more physician reviews of HM-relevant literature, visit our website.
Clinical question: What is the current level of continuity of care, and what factors affect continuity of care in the hospital setting? Has this changed with increasing use of hospitalists and limits on residency duty hours?
Background: Outpatient continuity of care leads to lower costs, better quality of life, and less emergency room use. Recent changes in residency hours have increased hand-offs and decreased inpatient continuity, but to what extent is unknown.
Study design: Retrospective cohort of 5% of Medicare claims data (530,000 patients in all) from 1996 to 2006, including patients admitted for COPD, congestive heart failure, or pneumonia who were cared for by a general internist or family practitioner.
Setting: Nationwide in the U.S.
Synopsis: The authors defined patients as having a primary-care physician (PCP) if they had three billed visits with the PCP in the last year, hospitalists as those who derived at least 90% of their Medicare claims from inpatient billing, and other generalists as those who met criteria as a generalist but did not fit these categories. Inpatient continuity of care decreased to 59% of patients seeing a single physician in the hospital in 2006 from 71% in 1996, with an accompanying decrease in the length of stay of one full day. There were large variations by geographic region, population size, and hospital characteristics. Patients cared for by hospitalists had slightly better continuity of care than those cared for by nonhospitalist generalists, and those who were cared for by both hospitalists and nonhospitalist generalists had the worst continuity of care. Having a PCP was associated with increased discontinuity of care as an inpatient, perhaps because of individual members of a practice rounding on all of the practice’s inpatients.
Bottom line: Patients were 5% less likely per year between 1996 and 2006 to have a single physician be their primary caregiver in the hospital, but the rise of the hospitalist movement does not seem to be the cause.
Citation: Fletcher KE, Sharma G, Zhang D, Kuo YF, Goodwin JS. Trends in inpatient continuity of care for a cohort of Medicare patients 1996-2006. J Hosp Med. 2011;6:441-447.
For more physician reviews of HM-relevant literature, visit our website.
The Dwindling Relevance of Annual Meetings
The annual meetings of the American College of Cardiology and the American Heart Association have been the centerpiece for the international exchange of ideas in clinical and basic cardiology for the last half century. They have attracted research scientists from around the world as a result of their preeminence as a platform for the presentation of new concepts.
They provided the setting for Mason Sones to show the first direct angiographic imaging of the coronary artery, for Andreis Grunzig to demonstrate the first percutaneous dilatation of the coronary artery, and for Michel Mirowski to present the first demonstration of the automatic implanted defibrillator. Those events caused gasps in the audience as we all saw, for the first time, a major breakthrough in cardiovascular medicine.
The scramble to get onto that platform or to be in that audience when the newest discovery was presented drew large audiences. The decrease in attendance in the past few years can be viewed as an index of the decrease in the importance of that platform.
The decreased attendance in the AHA and ACC annual scientific sessions provides ample evidence of the reduced role of American leadership and the ascendancy of European leadership in the world of cardiology. As the attendance at American meetings has ebbed, the European Society of Cardiology has seen a progressive increase in attendance of its annual meeting.
The AHA reached its highest attendance in the later part of the last century when its professional attendance in 1999 topped just over 20,000. It gradually slipped to 19,169 in 2010, and was 15,553 in Orlando in 2011. The ACC professional attendance has also fallen, from 18,542 in 2008 to 12,980 in 2011. At the same time, the attendance at the European Society of Cardiology has increased from 18,413 in 2002 in Berlin to almost 27,080 professional attendees in 2011 in Paris.
This decrease has not been observed in all American medical specialties. The Radiological Society of North America has had approximately 27,000 professional attendees for the last 5 years, and it advertises nearly eight football fields of exhibits.
One factor limiting attendance at some meetings is the expense, including the increase in admission fees, which now have gotten well into the four-figure level even if you are a member.
One of the most striking changes at the recent AHA and ACC meetings was the stark decrease in exhibitors. Exhibits that seemed to go on for miles in previous years, requiring rest stops at coffee stands along the way, have now became accessible with a casual walk. Many of the high-tech exhibitors either shrank their exhibit space or were entirely absent. Attendance and the number of exhibitors at the recent AHA meeting were impacted by the annual meeting of Transcatheter Cardiovascular Therapeutics, which had more than 12,000 attendees and was being held almost simultaneously a continent away. In order to fill the exhibition space at the recent AHA meeting, booth space was given over to displays of costume jewelry and pashmina scarves.
It is estimated by representatives of the ACC that international attendance at American meetings has decreased by at least one-third. This decrease is open to different interpretations. It has become increasingly difficult, for example, for many Asian and Eastern European cardiologists to obtain U.S. visas. But the fact that the Europeans do have an excellent meeting on their own soil has made that meeting more accessible to them. It is also clear that more Asian and American cardiologists are attending the European meeting. Part of this attraction has been related to a friendlier environment for the performance and consequent presentation of clinical trials in Europe.
The decrease in attendance at the American meetings is, in large part, a result of the balkanization of the "big tent" of cardiology. The creation of specialty associations by electrophysiologists, interventionalists, and heart failure specialists, to name but a few, has impacted on the appeal of – and need to engage in – the large annual meeting in order to satisfy professional requirements. Specialty cardiologists now have not only their own meeting platforms, but also their own specialty journals, which are in direct competition with JACC and Circulation.
What is lost, however, is the integrated educational experience that the practicing cardiologist needs in order to bring the entirety of cardiovascular science to the individual patient. When the field of cardiology was smaller and its scientific and clinical horizons were nearer at hand, this could be accomplished at one meeting.
We have, unfortunately, outgrown our "tent," but the Internet is now there to help us. Access to the meeting’s scientific presentations is now readily available through a variety of electronic media sites, including Cardiology News.
The annual meetings of the American College of Cardiology and the American Heart Association have been the centerpiece for the international exchange of ideas in clinical and basic cardiology for the last half century. They have attracted research scientists from around the world as a result of their preeminence as a platform for the presentation of new concepts.
They provided the setting for Mason Sones to show the first direct angiographic imaging of the coronary artery, for Andreis Grunzig to demonstrate the first percutaneous dilatation of the coronary artery, and for Michel Mirowski to present the first demonstration of the automatic implanted defibrillator. Those events caused gasps in the audience as we all saw, for the first time, a major breakthrough in cardiovascular medicine.
The scramble to get onto that platform or to be in that audience when the newest discovery was presented drew large audiences. The decrease in attendance in the past few years can be viewed as an index of the decrease in the importance of that platform.
The decreased attendance in the AHA and ACC annual scientific sessions provides ample evidence of the reduced role of American leadership and the ascendancy of European leadership in the world of cardiology. As the attendance at American meetings has ebbed, the European Society of Cardiology has seen a progressive increase in attendance of its annual meeting.
The AHA reached its highest attendance in the later part of the last century when its professional attendance in 1999 topped just over 20,000. It gradually slipped to 19,169 in 2010, and was 15,553 in Orlando in 2011. The ACC professional attendance has also fallen, from 18,542 in 2008 to 12,980 in 2011. At the same time, the attendance at the European Society of Cardiology has increased from 18,413 in 2002 in Berlin to almost 27,080 professional attendees in 2011 in Paris.
This decrease has not been observed in all American medical specialties. The Radiological Society of North America has had approximately 27,000 professional attendees for the last 5 years, and it advertises nearly eight football fields of exhibits.
One factor limiting attendance at some meetings is the expense, including the increase in admission fees, which now have gotten well into the four-figure level even if you are a member.
One of the most striking changes at the recent AHA and ACC meetings was the stark decrease in exhibitors. Exhibits that seemed to go on for miles in previous years, requiring rest stops at coffee stands along the way, have now became accessible with a casual walk. Many of the high-tech exhibitors either shrank their exhibit space or were entirely absent. Attendance and the number of exhibitors at the recent AHA meeting were impacted by the annual meeting of Transcatheter Cardiovascular Therapeutics, which had more than 12,000 attendees and was being held almost simultaneously a continent away. In order to fill the exhibition space at the recent AHA meeting, booth space was given over to displays of costume jewelry and pashmina scarves.
It is estimated by representatives of the ACC that international attendance at American meetings has decreased by at least one-third. This decrease is open to different interpretations. It has become increasingly difficult, for example, for many Asian and Eastern European cardiologists to obtain U.S. visas. But the fact that the Europeans do have an excellent meeting on their own soil has made that meeting more accessible to them. It is also clear that more Asian and American cardiologists are attending the European meeting. Part of this attraction has been related to a friendlier environment for the performance and consequent presentation of clinical trials in Europe.
The decrease in attendance at the American meetings is, in large part, a result of the balkanization of the "big tent" of cardiology. The creation of specialty associations by electrophysiologists, interventionalists, and heart failure specialists, to name but a few, has impacted on the appeal of – and need to engage in – the large annual meeting in order to satisfy professional requirements. Specialty cardiologists now have not only their own meeting platforms, but also their own specialty journals, which are in direct competition with JACC and Circulation.
What is lost, however, is the integrated educational experience that the practicing cardiologist needs in order to bring the entirety of cardiovascular science to the individual patient. When the field of cardiology was smaller and its scientific and clinical horizons were nearer at hand, this could be accomplished at one meeting.
We have, unfortunately, outgrown our "tent," but the Internet is now there to help us. Access to the meeting’s scientific presentations is now readily available through a variety of electronic media sites, including Cardiology News.
The annual meetings of the American College of Cardiology and the American Heart Association have been the centerpiece for the international exchange of ideas in clinical and basic cardiology for the last half century. They have attracted research scientists from around the world as a result of their preeminence as a platform for the presentation of new concepts.
They provided the setting for Mason Sones to show the first direct angiographic imaging of the coronary artery, for Andreis Grunzig to demonstrate the first percutaneous dilatation of the coronary artery, and for Michel Mirowski to present the first demonstration of the automatic implanted defibrillator. Those events caused gasps in the audience as we all saw, for the first time, a major breakthrough in cardiovascular medicine.
The scramble to get onto that platform or to be in that audience when the newest discovery was presented drew large audiences. The decrease in attendance in the past few years can be viewed as an index of the decrease in the importance of that platform.
The decreased attendance in the AHA and ACC annual scientific sessions provides ample evidence of the reduced role of American leadership and the ascendancy of European leadership in the world of cardiology. As the attendance at American meetings has ebbed, the European Society of Cardiology has seen a progressive increase in attendance of its annual meeting.
The AHA reached its highest attendance in the later part of the last century when its professional attendance in 1999 topped just over 20,000. It gradually slipped to 19,169 in 2010, and was 15,553 in Orlando in 2011. The ACC professional attendance has also fallen, from 18,542 in 2008 to 12,980 in 2011. At the same time, the attendance at the European Society of Cardiology has increased from 18,413 in 2002 in Berlin to almost 27,080 professional attendees in 2011 in Paris.
This decrease has not been observed in all American medical specialties. The Radiological Society of North America has had approximately 27,000 professional attendees for the last 5 years, and it advertises nearly eight football fields of exhibits.
One factor limiting attendance at some meetings is the expense, including the increase in admission fees, which now have gotten well into the four-figure level even if you are a member.
One of the most striking changes at the recent AHA and ACC meetings was the stark decrease in exhibitors. Exhibits that seemed to go on for miles in previous years, requiring rest stops at coffee stands along the way, have now became accessible with a casual walk. Many of the high-tech exhibitors either shrank their exhibit space or were entirely absent. Attendance and the number of exhibitors at the recent AHA meeting were impacted by the annual meeting of Transcatheter Cardiovascular Therapeutics, which had more than 12,000 attendees and was being held almost simultaneously a continent away. In order to fill the exhibition space at the recent AHA meeting, booth space was given over to displays of costume jewelry and pashmina scarves.
It is estimated by representatives of the ACC that international attendance at American meetings has decreased by at least one-third. This decrease is open to different interpretations. It has become increasingly difficult, for example, for many Asian and Eastern European cardiologists to obtain U.S. visas. But the fact that the Europeans do have an excellent meeting on their own soil has made that meeting more accessible to them. It is also clear that more Asian and American cardiologists are attending the European meeting. Part of this attraction has been related to a friendlier environment for the performance and consequent presentation of clinical trials in Europe.
The decrease in attendance at the American meetings is, in large part, a result of the balkanization of the "big tent" of cardiology. The creation of specialty associations by electrophysiologists, interventionalists, and heart failure specialists, to name but a few, has impacted on the appeal of – and need to engage in – the large annual meeting in order to satisfy professional requirements. Specialty cardiologists now have not only their own meeting platforms, but also their own specialty journals, which are in direct competition with JACC and Circulation.
What is lost, however, is the integrated educational experience that the practicing cardiologist needs in order to bring the entirety of cardiovascular science to the individual patient. When the field of cardiology was smaller and its scientific and clinical horizons were nearer at hand, this could be accomplished at one meeting.
We have, unfortunately, outgrown our "tent," but the Internet is now there to help us. Access to the meeting’s scientific presentations is now readily available through a variety of electronic media sites, including Cardiology News.
Infants Left Out of Survival Gains in Childhood Leukemia
Overall 5-year survival in children with acute lymphoblastic leukemia improved from 83.7% to 90.4% between the early 1990s and the early 2000s, according to a report published online March 12 in the Journal of Clinical Oncology.
Five-year survival improved by 30%-50% across all subgroups of patients during this interval, with one discouraging exception: It remained steady and much lower in infants aged less than 1 year, said Dr. Stephen P. Hunger of the University of Colorado Cancer Center and his associates in the Children’s Oncology Group (COG).
The investigators assessed outcomes in 21,626 children and adolescents participating in 36 ALL clinical trials in 1990-2005, which they described as "the largest childhood ALL cohort ever reported." The COG includes more than 200 member institutions in the United States, Canada, and other countries, which together have enrolled 56% of all cases of pediatric ALL that occurred in the United States between 1990 and 2005 in clinical trials.
"Thus, our results are representative of survival following contemporary therapy in the U.S." and differ somewhat from those of other large groups such as the National Cancer Institute’s SEER program, they said.
Dr. Hunger and his colleagues divided this cohort into three similar-sized groups in three eras: 7,304 patients treated in 1990-1994; 7,169 treated in 1995-1999; and 7,153 treated in 2000-2005. The median follow-up was 9.13 years, 8.02 years, and 5.35 years, respectively.
Most (92%) of these study subjects were treated in the United States, with 6% treated in Canada and 2% treated elsewhere.
Overall 5-year survival increased from 83.7% in the first era to 87.7% in the second and to 90.4% in the third. "We believe that the major reason for improved survival was decreased risk of relapse," they said (J. Clin. Oncol. 2012 March 12 [doi:10.1200/JCO.20911.37.8018]).
This is because the rate of deaths from relapse decreased markedly during this interval, from 43% in 1990-1994 to 27% in 2000-2005.
Among infants, the 5-year risk of death changed little, at 52.1% in 1990-1994 and 50.3% in 2000-2005. During the study period, "the COG pursued several strategies to attempt to increase survival for infants with ALL. Chemotherapy treatment was intensified significantly" in two clinical trials, while stem-cell transplantation was explored in others.
Stem-cell therapy was not found to be beneficial in infants in these studies, and chemotherapy intensification raised the rate of treatment-related death to the same degree that it lowered the rate of death from disease progression, with no net improvement in survival.
"Infant ALL is a unique high-risk subset that requires new therapeutic strategies," the researchers said.
Five-year survival improved 30%-50% across all other subgroups of patients: in all age groups (except infants), in both sexes, in all races and ethnicities, in patients with B-cell or T-cell disease, in patients with average-risk or high-risk profiles, and across all features of ALL according to NCI criteria.
However, the risk of death still remained higher in patients aged 10 or older, compared with those aged 1-10, higher in boys than in girls, higher in blacks and Hispanics than in whites, and higher in T-cell than in B-cell disease. Risk of death was two- to fourfold higher in patients high-risk than in average-risk patients.
It was encouraging that the "racial gap" in outcomes between whites and nonwhites narrowed during the study period. For example, the absolute difference in 5-year survival between whites and blacks declined from 11.0% in 1990-1994 to only 3.3% in 2000-2005. Racial differences between whites and nonwhites in ALL biology are still evident, with black and Hispanic patients being more likely than whites to carry higher-risk forms of the disease or higher-risk genetic profiles, the investigators noted.
A total of 36% of the ALL deaths in these study subjects occurred in patients who were at average risk. "Thus, efforts to decrease ALL deaths must focus both on high-risk patient subsets and on the large subset of patients with favorable clinical characteristics," Dr. Hunger and his associates said.
Based on their data, the researchers also "anticipate significant improvements in 10-year survival."
Children’s Oncology Group studies are supported by the National Cancer Institute. Dr. Hunger reported no financial conflicts of interest. One associate reported ties to EUSA Pharma, Sanofi-Aventis, Teva Pharmaceutical Industries, Bristol-Myers Squibb, Enzon Pharmaceuticals, Sigma Tau Pharmaceuticals, and Genzyme.
Overall 5-year survival in children with acute lymphoblastic leukemia improved from 83.7% to 90.4% between the early 1990s and the early 2000s, according to a report published online March 12 in the Journal of Clinical Oncology.
Five-year survival improved by 30%-50% across all subgroups of patients during this interval, with one discouraging exception: It remained steady and much lower in infants aged less than 1 year, said Dr. Stephen P. Hunger of the University of Colorado Cancer Center and his associates in the Children’s Oncology Group (COG).
The investigators assessed outcomes in 21,626 children and adolescents participating in 36 ALL clinical trials in 1990-2005, which they described as "the largest childhood ALL cohort ever reported." The COG includes more than 200 member institutions in the United States, Canada, and other countries, which together have enrolled 56% of all cases of pediatric ALL that occurred in the United States between 1990 and 2005 in clinical trials.
"Thus, our results are representative of survival following contemporary therapy in the U.S." and differ somewhat from those of other large groups such as the National Cancer Institute’s SEER program, they said.
Dr. Hunger and his colleagues divided this cohort into three similar-sized groups in three eras: 7,304 patients treated in 1990-1994; 7,169 treated in 1995-1999; and 7,153 treated in 2000-2005. The median follow-up was 9.13 years, 8.02 years, and 5.35 years, respectively.
Most (92%) of these study subjects were treated in the United States, with 6% treated in Canada and 2% treated elsewhere.
Overall 5-year survival increased from 83.7% in the first era to 87.7% in the second and to 90.4% in the third. "We believe that the major reason for improved survival was decreased risk of relapse," they said (J. Clin. Oncol. 2012 March 12 [doi:10.1200/JCO.20911.37.8018]).
This is because the rate of deaths from relapse decreased markedly during this interval, from 43% in 1990-1994 to 27% in 2000-2005.
Among infants, the 5-year risk of death changed little, at 52.1% in 1990-1994 and 50.3% in 2000-2005. During the study period, "the COG pursued several strategies to attempt to increase survival for infants with ALL. Chemotherapy treatment was intensified significantly" in two clinical trials, while stem-cell transplantation was explored in others.
Stem-cell therapy was not found to be beneficial in infants in these studies, and chemotherapy intensification raised the rate of treatment-related death to the same degree that it lowered the rate of death from disease progression, with no net improvement in survival.
"Infant ALL is a unique high-risk subset that requires new therapeutic strategies," the researchers said.
Five-year survival improved 30%-50% across all other subgroups of patients: in all age groups (except infants), in both sexes, in all races and ethnicities, in patients with B-cell or T-cell disease, in patients with average-risk or high-risk profiles, and across all features of ALL according to NCI criteria.
However, the risk of death still remained higher in patients aged 10 or older, compared with those aged 1-10, higher in boys than in girls, higher in blacks and Hispanics than in whites, and higher in T-cell than in B-cell disease. Risk of death was two- to fourfold higher in patients high-risk than in average-risk patients.
It was encouraging that the "racial gap" in outcomes between whites and nonwhites narrowed during the study period. For example, the absolute difference in 5-year survival between whites and blacks declined from 11.0% in 1990-1994 to only 3.3% in 2000-2005. Racial differences between whites and nonwhites in ALL biology are still evident, with black and Hispanic patients being more likely than whites to carry higher-risk forms of the disease or higher-risk genetic profiles, the investigators noted.
A total of 36% of the ALL deaths in these study subjects occurred in patients who were at average risk. "Thus, efforts to decrease ALL deaths must focus both on high-risk patient subsets and on the large subset of patients with favorable clinical characteristics," Dr. Hunger and his associates said.
Based on their data, the researchers also "anticipate significant improvements in 10-year survival."
Children’s Oncology Group studies are supported by the National Cancer Institute. Dr. Hunger reported no financial conflicts of interest. One associate reported ties to EUSA Pharma, Sanofi-Aventis, Teva Pharmaceutical Industries, Bristol-Myers Squibb, Enzon Pharmaceuticals, Sigma Tau Pharmaceuticals, and Genzyme.
Overall 5-year survival in children with acute lymphoblastic leukemia improved from 83.7% to 90.4% between the early 1990s and the early 2000s, according to a report published online March 12 in the Journal of Clinical Oncology.
Five-year survival improved by 30%-50% across all subgroups of patients during this interval, with one discouraging exception: It remained steady and much lower in infants aged less than 1 year, said Dr. Stephen P. Hunger of the University of Colorado Cancer Center and his associates in the Children’s Oncology Group (COG).
The investigators assessed outcomes in 21,626 children and adolescents participating in 36 ALL clinical trials in 1990-2005, which they described as "the largest childhood ALL cohort ever reported." The COG includes more than 200 member institutions in the United States, Canada, and other countries, which together have enrolled 56% of all cases of pediatric ALL that occurred in the United States between 1990 and 2005 in clinical trials.
"Thus, our results are representative of survival following contemporary therapy in the U.S." and differ somewhat from those of other large groups such as the National Cancer Institute’s SEER program, they said.
Dr. Hunger and his colleagues divided this cohort into three similar-sized groups in three eras: 7,304 patients treated in 1990-1994; 7,169 treated in 1995-1999; and 7,153 treated in 2000-2005. The median follow-up was 9.13 years, 8.02 years, and 5.35 years, respectively.
Most (92%) of these study subjects were treated in the United States, with 6% treated in Canada and 2% treated elsewhere.
Overall 5-year survival increased from 83.7% in the first era to 87.7% in the second and to 90.4% in the third. "We believe that the major reason for improved survival was decreased risk of relapse," they said (J. Clin. Oncol. 2012 March 12 [doi:10.1200/JCO.20911.37.8018]).
This is because the rate of deaths from relapse decreased markedly during this interval, from 43% in 1990-1994 to 27% in 2000-2005.
Among infants, the 5-year risk of death changed little, at 52.1% in 1990-1994 and 50.3% in 2000-2005. During the study period, "the COG pursued several strategies to attempt to increase survival for infants with ALL. Chemotherapy treatment was intensified significantly" in two clinical trials, while stem-cell transplantation was explored in others.
Stem-cell therapy was not found to be beneficial in infants in these studies, and chemotherapy intensification raised the rate of treatment-related death to the same degree that it lowered the rate of death from disease progression, with no net improvement in survival.
"Infant ALL is a unique high-risk subset that requires new therapeutic strategies," the researchers said.
Five-year survival improved 30%-50% across all other subgroups of patients: in all age groups (except infants), in both sexes, in all races and ethnicities, in patients with B-cell or T-cell disease, in patients with average-risk or high-risk profiles, and across all features of ALL according to NCI criteria.
However, the risk of death still remained higher in patients aged 10 or older, compared with those aged 1-10, higher in boys than in girls, higher in blacks and Hispanics than in whites, and higher in T-cell than in B-cell disease. Risk of death was two- to fourfold higher in patients high-risk than in average-risk patients.
It was encouraging that the "racial gap" in outcomes between whites and nonwhites narrowed during the study period. For example, the absolute difference in 5-year survival between whites and blacks declined from 11.0% in 1990-1994 to only 3.3% in 2000-2005. Racial differences between whites and nonwhites in ALL biology are still evident, with black and Hispanic patients being more likely than whites to carry higher-risk forms of the disease or higher-risk genetic profiles, the investigators noted.
A total of 36% of the ALL deaths in these study subjects occurred in patients who were at average risk. "Thus, efforts to decrease ALL deaths must focus both on high-risk patient subsets and on the large subset of patients with favorable clinical characteristics," Dr. Hunger and his associates said.
Based on their data, the researchers also "anticipate significant improvements in 10-year survival."
Children’s Oncology Group studies are supported by the National Cancer Institute. Dr. Hunger reported no financial conflicts of interest. One associate reported ties to EUSA Pharma, Sanofi-Aventis, Teva Pharmaceutical Industries, Bristol-Myers Squibb, Enzon Pharmaceuticals, Sigma Tau Pharmaceuticals, and Genzyme.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Major Finding: Five-year survival in children with acute lymphoblastic leukemia steadily increased from 83.7% in 1990-1994 to 87.7% in 1994-1999 and to 90.4% in 2000-2005, largely from a decreased risk of relapse.
Data Source: An analysis of data from 36 clinical trials of pediatric ALL involving 21,626 study subjects treated in 1990-2005.
Disclosures: Children’s Oncology Group studies are supported by the National Cancer Institute. Dr. Hunger reported no financial conflicts of interest. One associate reported ties to EUSA Pharma, Sanofi-Aventis, Teva Pharmaceutical Industries, Bristol-Myers Squibb, Enzon Pharmaceuticals, Sigma Tau Pharmaceuticals, and Genzyme.
Slight Hyperglycemia Risk Shouldn't Deter Statin Use
The Food and Drug Administration's announcement that the labeling of statins will now note their potential for raising a patient’s blood sugar and glycosylated hemoglobin levels is a reminder that, despite their relative safety, statin treatment poses some level of risk and hence should not be prescribed indiscriminately, experts said.
On the other hand, the risk for blood sugar elevation is modest enough that for the vast majority of patients who have significant cardiovascular disease (CVD) risk, the potential benefit from statin treatment continues to far outweigh the risk patients might face from statin-induced hyperglycemia, according to several experts interviewed for this article. Patients with cardiovascular disease risk who could remain on statins include those who have already had a cardiovascular event, the secondary prevention population, and patients who already have diabetes, considered a coronary risk equivalent because of the sizeable risk that diabetes confers for a future cardiovascular event.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease."
Boosted hyperglycemia that pushes a person’s fasting plasma glucose level to 126 mg/dL or above, the range diagnosed as type 2 diabetes, "is probably the most frequent quantifiable harm from statins" but is still uncommon, noted Dr. Jennifer G. Robinson, professor of medicine and epidemiology at the University of Iowa in Iowa City. She estimated that of the 10%-15% of patients who will develop type 2 diabetes over a period of several years on statin treatment, roughly 1 new case of diabetes out of every 500 incident cases will be attributable to statin treatment, based on the risk information available today.
"It's not very much. It should not change any clinician’s day to day practice in any way," said Dr. Robinson, who is also a vice-chair of the Adult Treatment Panel IV, the group assembled by the National Heart, Lung, and Blood Institute to issue new U.S. cholesterol management guidelines, expected later this year. "It just means that you don’t give a statin to everyone, not someone with a 1% risk for a cardiovascular event over the next 10 years," she said.
Primary prevention poses the most complicated issues, when physicians prescribe statins to people who have not yet had any cardiovascular event. Prescribers face the difficult question of when the risk for incident hyperglycemia triggered by a statin starts to outweigh the benefit from cardiovascular risk reduction. Further muddying the question of whom to exclude from primary prevention with statin treatment are the unknowns that shroud the effect: How do statins cause this? Which patients are most susceptible? Do different statins pose varying levels of hyperglycemia risk?
"As increasingly large populations become candidates for statin treatment, with new guidelines and new methods for CVD risk-prediction modeling, it will be very important to look at the benefit to risk ratio of treatment, including the risk for developing diabetes," said Dr. JoAnn E. Manson, professor of medicine at Harvard Medical School and chief of preventive medicine at Brigham and Women’s Hospital in Boston.
Relatively low-risk groups of patients who are increasingly prescribed statins include adolescents, young adults, and middle-aged women, she noted. "The key is the absolute risk of CVD in these groups, more than their relative risk. In a population with a low absolute risk of CVD events, we need to look very carefully to see where the crossover occurs from net benefit to net risk of treatment."
A problem for the time being is that no good way exists for identifying what factors, beyond borderline high blood glucose at baseline, help identify patients at increased risk for developing diabetes while on statin treatment. Additional research and guidance about what level of fasting plasma glucose at baseline, before a statin regimen starts, should trigger concern, and how often plasma glucose should be monitored once a patient is on a statin, will be helpful, Dr. Manson said.
"Professional societies and expert groups should make clear recommendations about the need for routine vs. targeted glucose testing, as well as the frequency. Consensus guidelines don’t yet exist. The new statin label eliminates testing liver function. Will this be replaced by excessive testing of blood glucose, a practice that could be burdensome to patients and clinicians and drive up health care costs?" she said in an interview.
The dilemma physicians also face when deciding whether to prescribe a statin to patients toward the low end of the cardiovascular risk spectrum is that the same risk factors that might flag patients with a high risk for insulin resistance, hyperglycemia, and the development of type 2 diabetes – factors such as obesity, inadequate physical activity, elements of metabolic syndrome, and a "prediabetic" fasting plasma glucose level of 110-125 mg/dL – also function as cardiovascular disease risk factors.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease," Dr. Manson said. She recommended that patients on statin treatment at least be told to be on the alert for developing new symptoms of diabetes: frequent thirst, frequent urination, and blurred vision. "And lifestyle modifications should be intensified to reduce both diabetes and CVD risk."
"The excess risk for diabetes is concentrated in the people with fasting blood sugars in the 115- to 125-mg/dL range" said Dr. Roger S. Blumenthal, professor of medicine and director of preventive medicine at Johns Hopkins Medical Institutions in Baltimore. "A lot of those people are clearly insulin resistant. We almost always have a fasting plasma glucose [when patients are about to start on a statin] as part of a basic lipid profile. With physicians aware of the association, I think this will focus more attention on vulnerable patients in the 115- to 125-mg/dL range, who are headed for diabetes if they don’t make significant improvements in their diet and exercise habits. It’s reasonable to look at glucose and tell patients that a statin might potentially raise their blood sugar by 5-7 mg/dL, but exercising and dropping some excess weight will significantly improve their blood sugar."
Targeted use of plasma glucose testing in statin recipients who seem to have the greatest risk for developing diabetes also received endorsement from Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, in Fresno. He suggested possibly doing annual testing of patients with metabolic syndrome, those who are obese, those with a family history of diabetes, and patients who have previously shown impaired glucose tolerance on a tolerance test.
If a patient’s fasting plasma glucose began to creep up on a statin regimen, "I’d look for other reasons, such as did they gain weight?" he said. Seeing a possible hyperglycemic effect should also prompt a reassessment of whether the patient benefited from the statin, and whether they have made necessary lifestyle changes like improved diet and increased exercise. Rising blood sugar could be used to help motivate a patient to do better on lifestyle measures, and trigger a reevaluation of whether the patient is, on balance, benefiting from the statin, he said. Changing the statin used or the dosage is tricky, because no evidence exists now to support such steps.
But because the biggest signal for the prodiabetic effect of statins came in results from the Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) (N. Engl. J. Med. 2008;359:2195-207), the current perception among at least some physicians is that rosuvastatin (Crestor) poses the biggest hyperglycemic risk. "Some physicians might consider changing [the prescribed statin] from rosuvastatin to simvastatin or atorvastatin," Dr. Blumenthal said.
The FDA took the right step in adding the hyperglycemia information to statin labeling, said Dr. Deedwania. "They give the data, and leave it up to physicians to make their own conclusions."
As a consequence of the FDA’s actions "a lot more physicians will pay attention to glucose as they put patients on statins. The evidence is consistent, and most now agree that it’s real," said Dr. Stephen J. Nicholls, a cardiologist at the Cleveland Clinic. "But these are not completely healthy people with low glucose levels who suddenly, on a statin, become diabetic. What this reinforces is that while there will continue to be a lot of people who require statin treatment, the cornerstone of treatment is lifestyle change: diet, exercise, and weight loss. There is a continuum of risk: Patients at higher risk will benefit from a statin; for patients at very low risk use lifestyle. And if you put a patient on a statin, you need to keep an eye on them."
Dr. Robinson, Dr. Manson, and Dr. Blumenthal said that they had no relevant financial disclosures. Dr. Deedwania said that he has been a consultant to Pfizer, Amarin, and Amgen. Dr. Nicholls said he has received research support from AstraZeneca and has been a consultant to AstraZeneca, Merck, and Pfizer.
The Food and Drug Administration's announcement that the labeling of statins will now note their potential for raising a patient’s blood sugar and glycosylated hemoglobin levels is a reminder that, despite their relative safety, statin treatment poses some level of risk and hence should not be prescribed indiscriminately, experts said.
On the other hand, the risk for blood sugar elevation is modest enough that for the vast majority of patients who have significant cardiovascular disease (CVD) risk, the potential benefit from statin treatment continues to far outweigh the risk patients might face from statin-induced hyperglycemia, according to several experts interviewed for this article. Patients with cardiovascular disease risk who could remain on statins include those who have already had a cardiovascular event, the secondary prevention population, and patients who already have diabetes, considered a coronary risk equivalent because of the sizeable risk that diabetes confers for a future cardiovascular event.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease."
Boosted hyperglycemia that pushes a person’s fasting plasma glucose level to 126 mg/dL or above, the range diagnosed as type 2 diabetes, "is probably the most frequent quantifiable harm from statins" but is still uncommon, noted Dr. Jennifer G. Robinson, professor of medicine and epidemiology at the University of Iowa in Iowa City. She estimated that of the 10%-15% of patients who will develop type 2 diabetes over a period of several years on statin treatment, roughly 1 new case of diabetes out of every 500 incident cases will be attributable to statin treatment, based on the risk information available today.
"It's not very much. It should not change any clinician’s day to day practice in any way," said Dr. Robinson, who is also a vice-chair of the Adult Treatment Panel IV, the group assembled by the National Heart, Lung, and Blood Institute to issue new U.S. cholesterol management guidelines, expected later this year. "It just means that you don’t give a statin to everyone, not someone with a 1% risk for a cardiovascular event over the next 10 years," she said.
Primary prevention poses the most complicated issues, when physicians prescribe statins to people who have not yet had any cardiovascular event. Prescribers face the difficult question of when the risk for incident hyperglycemia triggered by a statin starts to outweigh the benefit from cardiovascular risk reduction. Further muddying the question of whom to exclude from primary prevention with statin treatment are the unknowns that shroud the effect: How do statins cause this? Which patients are most susceptible? Do different statins pose varying levels of hyperglycemia risk?
"As increasingly large populations become candidates for statin treatment, with new guidelines and new methods for CVD risk-prediction modeling, it will be very important to look at the benefit to risk ratio of treatment, including the risk for developing diabetes," said Dr. JoAnn E. Manson, professor of medicine at Harvard Medical School and chief of preventive medicine at Brigham and Women’s Hospital in Boston.
Relatively low-risk groups of patients who are increasingly prescribed statins include adolescents, young adults, and middle-aged women, she noted. "The key is the absolute risk of CVD in these groups, more than their relative risk. In a population with a low absolute risk of CVD events, we need to look very carefully to see where the crossover occurs from net benefit to net risk of treatment."
A problem for the time being is that no good way exists for identifying what factors, beyond borderline high blood glucose at baseline, help identify patients at increased risk for developing diabetes while on statin treatment. Additional research and guidance about what level of fasting plasma glucose at baseline, before a statin regimen starts, should trigger concern, and how often plasma glucose should be monitored once a patient is on a statin, will be helpful, Dr. Manson said.
"Professional societies and expert groups should make clear recommendations about the need for routine vs. targeted glucose testing, as well as the frequency. Consensus guidelines don’t yet exist. The new statin label eliminates testing liver function. Will this be replaced by excessive testing of blood glucose, a practice that could be burdensome to patients and clinicians and drive up health care costs?" she said in an interview.
The dilemma physicians also face when deciding whether to prescribe a statin to patients toward the low end of the cardiovascular risk spectrum is that the same risk factors that might flag patients with a high risk for insulin resistance, hyperglycemia, and the development of type 2 diabetes – factors such as obesity, inadequate physical activity, elements of metabolic syndrome, and a "prediabetic" fasting plasma glucose level of 110-125 mg/dL – also function as cardiovascular disease risk factors.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease," Dr. Manson said. She recommended that patients on statin treatment at least be told to be on the alert for developing new symptoms of diabetes: frequent thirst, frequent urination, and blurred vision. "And lifestyle modifications should be intensified to reduce both diabetes and CVD risk."
"The excess risk for diabetes is concentrated in the people with fasting blood sugars in the 115- to 125-mg/dL range" said Dr. Roger S. Blumenthal, professor of medicine and director of preventive medicine at Johns Hopkins Medical Institutions in Baltimore. "A lot of those people are clearly insulin resistant. We almost always have a fasting plasma glucose [when patients are about to start on a statin] as part of a basic lipid profile. With physicians aware of the association, I think this will focus more attention on vulnerable patients in the 115- to 125-mg/dL range, who are headed for diabetes if they don’t make significant improvements in their diet and exercise habits. It’s reasonable to look at glucose and tell patients that a statin might potentially raise their blood sugar by 5-7 mg/dL, but exercising and dropping some excess weight will significantly improve their blood sugar."
Targeted use of plasma glucose testing in statin recipients who seem to have the greatest risk for developing diabetes also received endorsement from Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, in Fresno. He suggested possibly doing annual testing of patients with metabolic syndrome, those who are obese, those with a family history of diabetes, and patients who have previously shown impaired glucose tolerance on a tolerance test.
If a patient’s fasting plasma glucose began to creep up on a statin regimen, "I’d look for other reasons, such as did they gain weight?" he said. Seeing a possible hyperglycemic effect should also prompt a reassessment of whether the patient benefited from the statin, and whether they have made necessary lifestyle changes like improved diet and increased exercise. Rising blood sugar could be used to help motivate a patient to do better on lifestyle measures, and trigger a reevaluation of whether the patient is, on balance, benefiting from the statin, he said. Changing the statin used or the dosage is tricky, because no evidence exists now to support such steps.
But because the biggest signal for the prodiabetic effect of statins came in results from the Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) (N. Engl. J. Med. 2008;359:2195-207), the current perception among at least some physicians is that rosuvastatin (Crestor) poses the biggest hyperglycemic risk. "Some physicians might consider changing [the prescribed statin] from rosuvastatin to simvastatin or atorvastatin," Dr. Blumenthal said.
The FDA took the right step in adding the hyperglycemia information to statin labeling, said Dr. Deedwania. "They give the data, and leave it up to physicians to make their own conclusions."
As a consequence of the FDA’s actions "a lot more physicians will pay attention to glucose as they put patients on statins. The evidence is consistent, and most now agree that it’s real," said Dr. Stephen J. Nicholls, a cardiologist at the Cleveland Clinic. "But these are not completely healthy people with low glucose levels who suddenly, on a statin, become diabetic. What this reinforces is that while there will continue to be a lot of people who require statin treatment, the cornerstone of treatment is lifestyle change: diet, exercise, and weight loss. There is a continuum of risk: Patients at higher risk will benefit from a statin; for patients at very low risk use lifestyle. And if you put a patient on a statin, you need to keep an eye on them."
Dr. Robinson, Dr. Manson, and Dr. Blumenthal said that they had no relevant financial disclosures. Dr. Deedwania said that he has been a consultant to Pfizer, Amarin, and Amgen. Dr. Nicholls said he has received research support from AstraZeneca and has been a consultant to AstraZeneca, Merck, and Pfizer.
The Food and Drug Administration's announcement that the labeling of statins will now note their potential for raising a patient’s blood sugar and glycosylated hemoglobin levels is a reminder that, despite their relative safety, statin treatment poses some level of risk and hence should not be prescribed indiscriminately, experts said.
On the other hand, the risk for blood sugar elevation is modest enough that for the vast majority of patients who have significant cardiovascular disease (CVD) risk, the potential benefit from statin treatment continues to far outweigh the risk patients might face from statin-induced hyperglycemia, according to several experts interviewed for this article. Patients with cardiovascular disease risk who could remain on statins include those who have already had a cardiovascular event, the secondary prevention population, and patients who already have diabetes, considered a coronary risk equivalent because of the sizeable risk that diabetes confers for a future cardiovascular event.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease."
Boosted hyperglycemia that pushes a person’s fasting plasma glucose level to 126 mg/dL or above, the range diagnosed as type 2 diabetes, "is probably the most frequent quantifiable harm from statins" but is still uncommon, noted Dr. Jennifer G. Robinson, professor of medicine and epidemiology at the University of Iowa in Iowa City. She estimated that of the 10%-15% of patients who will develop type 2 diabetes over a period of several years on statin treatment, roughly 1 new case of diabetes out of every 500 incident cases will be attributable to statin treatment, based on the risk information available today.
"It's not very much. It should not change any clinician’s day to day practice in any way," said Dr. Robinson, who is also a vice-chair of the Adult Treatment Panel IV, the group assembled by the National Heart, Lung, and Blood Institute to issue new U.S. cholesterol management guidelines, expected later this year. "It just means that you don’t give a statin to everyone, not someone with a 1% risk for a cardiovascular event over the next 10 years," she said.
Primary prevention poses the most complicated issues, when physicians prescribe statins to people who have not yet had any cardiovascular event. Prescribers face the difficult question of when the risk for incident hyperglycemia triggered by a statin starts to outweigh the benefit from cardiovascular risk reduction. Further muddying the question of whom to exclude from primary prevention with statin treatment are the unknowns that shroud the effect: How do statins cause this? Which patients are most susceptible? Do different statins pose varying levels of hyperglycemia risk?
"As increasingly large populations become candidates for statin treatment, with new guidelines and new methods for CVD risk-prediction modeling, it will be very important to look at the benefit to risk ratio of treatment, including the risk for developing diabetes," said Dr. JoAnn E. Manson, professor of medicine at Harvard Medical School and chief of preventive medicine at Brigham and Women’s Hospital in Boston.
Relatively low-risk groups of patients who are increasingly prescribed statins include adolescents, young adults, and middle-aged women, she noted. "The key is the absolute risk of CVD in these groups, more than their relative risk. In a population with a low absolute risk of CVD events, we need to look very carefully to see where the crossover occurs from net benefit to net risk of treatment."
A problem for the time being is that no good way exists for identifying what factors, beyond borderline high blood glucose at baseline, help identify patients at increased risk for developing diabetes while on statin treatment. Additional research and guidance about what level of fasting plasma glucose at baseline, before a statin regimen starts, should trigger concern, and how often plasma glucose should be monitored once a patient is on a statin, will be helpful, Dr. Manson said.
"Professional societies and expert groups should make clear recommendations about the need for routine vs. targeted glucose testing, as well as the frequency. Consensus guidelines don’t yet exist. The new statin label eliminates testing liver function. Will this be replaced by excessive testing of blood glucose, a practice that could be burdensome to patients and clinicians and drive up health care costs?" she said in an interview.
The dilemma physicians also face when deciding whether to prescribe a statin to patients toward the low end of the cardiovascular risk spectrum is that the same risk factors that might flag patients with a high risk for insulin resistance, hyperglycemia, and the development of type 2 diabetes – factors such as obesity, inadequate physical activity, elements of metabolic syndrome, and a "prediabetic" fasting plasma glucose level of 110-125 mg/dL – also function as cardiovascular disease risk factors.
"It would be a mistake to say that anyone at high risk for diabetes should be denied a statin because these people are also at high risk for cardiovascular disease," Dr. Manson said. She recommended that patients on statin treatment at least be told to be on the alert for developing new symptoms of diabetes: frequent thirst, frequent urination, and blurred vision. "And lifestyle modifications should be intensified to reduce both diabetes and CVD risk."
"The excess risk for diabetes is concentrated in the people with fasting blood sugars in the 115- to 125-mg/dL range" said Dr. Roger S. Blumenthal, professor of medicine and director of preventive medicine at Johns Hopkins Medical Institutions in Baltimore. "A lot of those people are clearly insulin resistant. We almost always have a fasting plasma glucose [when patients are about to start on a statin] as part of a basic lipid profile. With physicians aware of the association, I think this will focus more attention on vulnerable patients in the 115- to 125-mg/dL range, who are headed for diabetes if they don’t make significant improvements in their diet and exercise habits. It’s reasonable to look at glucose and tell patients that a statin might potentially raise their blood sugar by 5-7 mg/dL, but exercising and dropping some excess weight will significantly improve their blood sugar."
Targeted use of plasma glucose testing in statin recipients who seem to have the greatest risk for developing diabetes also received endorsement from Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, in Fresno. He suggested possibly doing annual testing of patients with metabolic syndrome, those who are obese, those with a family history of diabetes, and patients who have previously shown impaired glucose tolerance on a tolerance test.
If a patient’s fasting plasma glucose began to creep up on a statin regimen, "I’d look for other reasons, such as did they gain weight?" he said. Seeing a possible hyperglycemic effect should also prompt a reassessment of whether the patient benefited from the statin, and whether they have made necessary lifestyle changes like improved diet and increased exercise. Rising blood sugar could be used to help motivate a patient to do better on lifestyle measures, and trigger a reevaluation of whether the patient is, on balance, benefiting from the statin, he said. Changing the statin used or the dosage is tricky, because no evidence exists now to support such steps.
But because the biggest signal for the prodiabetic effect of statins came in results from the Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) (N. Engl. J. Med. 2008;359:2195-207), the current perception among at least some physicians is that rosuvastatin (Crestor) poses the biggest hyperglycemic risk. "Some physicians might consider changing [the prescribed statin] from rosuvastatin to simvastatin or atorvastatin," Dr. Blumenthal said.
The FDA took the right step in adding the hyperglycemia information to statin labeling, said Dr. Deedwania. "They give the data, and leave it up to physicians to make their own conclusions."
As a consequence of the FDA’s actions "a lot more physicians will pay attention to glucose as they put patients on statins. The evidence is consistent, and most now agree that it’s real," said Dr. Stephen J. Nicholls, a cardiologist at the Cleveland Clinic. "But these are not completely healthy people with low glucose levels who suddenly, on a statin, become diabetic. What this reinforces is that while there will continue to be a lot of people who require statin treatment, the cornerstone of treatment is lifestyle change: diet, exercise, and weight loss. There is a continuum of risk: Patients at higher risk will benefit from a statin; for patients at very low risk use lifestyle. And if you put a patient on a statin, you need to keep an eye on them."
Dr. Robinson, Dr. Manson, and Dr. Blumenthal said that they had no relevant financial disclosures. Dr. Deedwania said that he has been a consultant to Pfizer, Amarin, and Amgen. Dr. Nicholls said he has received research support from AstraZeneca and has been a consultant to AstraZeneca, Merck, and Pfizer.
Care Transitions for the Underserved
Hospital readmissions are common and costly, and represent a significant burden to the healthcare system. The challenges of postdischarge medication uncertainty, lack of self‐management support, and lack of timely access to health professionals1 are compounded in uninsured and Medicaid individuals by limited access to medications and primary care, financial strain, insecure housing, and limited social support.2
Our hospital cares for a large number of uninsured and low‐income publicly insured patients. The Portland area safety‐net, which consists of a network of 14 federally qualified health centers and free clinics, has limited capacity for uncompensated care. Uninsured patientsand to a lesser degree, Medicaid patientshave difficulty establishing primary care. Prior to the implementation of our program, uninsured and Medicaid patients without a usual source of care were given a list of safety‐net clinics at discharge, but frequently could not access appointments or navigate the complex system. There were no well‐developed partnerships between hospital and outpatient clinics for uninsured or Medicaid patients. The hospital lacked a systematic approach to securing postdischarge follow‐up and peridischarge patient education, and uninsured patients were financially responsible for most medications upon discharge. The costs of uncompensated or undercompensated potentially preventable readmissions for these patients, along with the recognition of gaps in quality, ultimately provided the rationale for a medical center‐funded transitional care intervention for uninsured and low‐income publicly insured patients.
Several transitional care improvement programs have shown effectiveness in reducing hospital readmissions,1, 35 but most have been conducted in settings where patients have secure access to outpatient care, and none have focused specifically on uninsured or Medicaid patients. Moreover, the development of these programs requires time and capital. Transitional care programs that have published results, to date, have been funded through government or private foundation grants1, 35; however, broader implementation of transitional care innovations will require financial and intellectual engagement of healthcare institutions themselves.
This report describes development of the Care Transitions Innovation (C‐TraIn), a multicomponent transitional care intervention for uninsured and low‐income publicly insured adults at a large, urban academic medical center, Oregon Health & Science University (OHSU). Because institutional funding and engagement is critical to the sustainability and scalability of similar programs, we also describe our process for gaining institutional support. Our hypothesis is that C‐TraIn can reduce readmissions and emergency department (ED) use at 30 days after hospital discharge, compared with usual care.
METHODS
Engaging Institutional Leaders
Early and continued efforts to engage hospital administrators were integral to our ultimate success in gaining institutional funding and leadership support. Initially, we convened what we called a Health Systems Morbidity and Mortality conference, featuring an uninsured patient who told of his postdischarge experiences and costly, potentially preventable readmission. We invited a broad array of potential stakeholders, including representatives from hospital administration, hospital case managers and social workers, community safety‐net providers, inpatient and outpatient physicians, residents, and medical students. Our patient was previously admitted to OHSU and diagnosed with pneumonia, hypothyroidism, sleep apnea, and depression. At discharge, he was given a list of low‐cost clinics; however, he was unable to arrange follow‐up, could not afford prescriptions, and felt overwhelmed trying to navigate a complex system. Consequently, he received no outpatient healthcare and his illnesses progressed. Unable to stay awake as a long‐haul trucker, he lost his job and subsequently his housing, and was readmitted to the intensive care unit with severe hypercarbic respiratory failure, volume overload, and hypothyroidism. The $130,000 charge for his 19‐day rehospitalization was largely un‐recuperated by the hospital. The case was a stark example of the patient‐safety and financial costs of fragmented care, and the conference was a nidus for further institutional engagement and program development, the key steps of which are described in Table 1.
| Time | Key Step | How Step Was Achieved | Take Home Points |
|---|---|---|---|
| |||
| July 2008July 2009 | 1. Identified key stakeholders | Considered varied stakeholders impacted by transitional care gaps for uninsured and Medicaid patients | Casting a wide net early in the process promoted high level of engagement and allowed self‐identification of some stakeholders |
| 2. Framed problems and opportunities; exposed costs of existing system shortcomings | Educational conference (that we called a Health Systems M&M) fostered a blame‐free environment to explore varied perspectives | Individual patient story made policy issue more accessible to a wide range of stakeholders | |
| Discussion of exposed drivers and costs of misaligned incentives; highlighted inroads to developing a business case for change | |||
| Oct 2008June 2009 | 3. Identified administrative allies and leaders with high bridging capital | Follow‐up with administrator after Health System M&M allowed further identification of key administrative stakeholders | Administrator insight highlighted institutional priorities and strategic plans |
| Ongoing meetings over 9 moto advocate for change, explore support for program development | Key ally within administration facilitated conversation with executive leadership whose support was a critical for program success | ||
| July 2009June 2010 | 4. Framed processes locally with continued involvement from multiple stakeholders | Performed multicomponent needs assessment | Patient assessment included inpatients for ease of survey administration |
| Utilized efforts of student volunteers for low‐budget option | |||
| Existing administrative support aided patient tracking | |||
| Non‐integrated health system and lack of claims data for uninsured limited usefulness of administrative utilization data | |||
| 5. Performed cost analysis to further support the business and quality case | Used OHSU data from needs assessment patient sample to estimate potential costs and savings of saved readmissions and avoided ED visits | Business case highlighted existing costs to OHSU for uncompensated care; program presented a solution to realign incentives and better allocate existing hospital expenditures | |
| Qualitative patient interviews exposed opportunity for quality improvement | Highlighted pilot as an opportunity for institutional learning about transitional care improvements | ||
| 6. Use needs assessment to map intervention | Drew upon local and national health systems expertise through literature review and consultation with local and national program leaders | OHSU's Care Transitions Innovation (C‐TraIn) includes elements aimed at improving access, patient education, care coordination, and systems integration (Table 2) | |
| Matched patient needs to specific elements of program design | |||
Planning the Intervention
Findings from a patient needs assessment and community stakeholder meetingsdescribed belowdirectly informed a multicomponent intervention that includes linkages and payment for medical homes for uninsured patients who lack access to outpatient care, a transitional care nurse whose care bridges inpatient and outpatient settings, inpatient pharmacy consultation, and provision of 30 days of medications at hospital discharge for uninsured patients (Table 2).
| Program Element | Description | Resources per 200 Patients |
|---|---|---|
| ||
| Transitional care RN | Augments patient education and care coordination in the hospital until 30 days after discharge. Tasks include: | 1.0 FTE nurse salary* |
| developing a personal health record with inpatients | ||
| completing a home visit within 72 hr of discharge to focus on medication reconciliation and patient self‐management | ||
| low‐risk patients receive 3 calls and no home visit (see Supporting Information, Appendix 1, in the online version of this article) | ||
| 2 subsequent phone calls to provide additional coaching, identify unmet needs, and close the loop on incomplete financial paperwork | ||
| The nurse provides a warm handoff with clinic staff, assists in scheduling timely posthospital follow‐up, and assures timely transfer of DC summaries. She coordinates posthospital care management with Medicaid case‐workers when available. | ||
| Pharmacy | Consultation: Inpatient pharmacists reconcile and simplify medication regimens, educate patients, and assess adherence barriers. | 0.4 FTE inpatient pharmacist salary |
| Prescription support: For uninsured patients, pharmacists guide MD prescribing towards medications available on the C‐TraIn value‐based formulary, a low‐cost formulary that reflects medications available through $4 plans, a Medicaid formulary, and FQHC on‐site pharmacies. | Estimated $12/prescription; 6.5 prescriptions/patient | |
| Uninsured patients are given 30 days of bridging prescription medications at hospital discharge free of charge. | ||
| Outpatient medical home and specialty care linkages | OHSU has partnered with outpatient clinics on a per‐patient basis to support funding of primary care for uninsured patients who lack a usual source of care. Clinics also provide coordinated care for Medicaid patients without assigned primary care, and have committed to engaging in continuous quality improvement. Clinics include an academic general internal medicine practice, an FQHC specializing in addiction and care for the homeless, and an FQHC that serves a low‐income rural population. | Estimated 8 primary care visits/yr at $205/visit (FQHC reimbursement rate) equates to $1640/ patient/yr. |
| Timely posthospital specialty care related to index admission diagnoses is coordinated through OHSU's outpatient specialty clinics. | ||
| Monthly care coordination meetings | We convene a diverse team of community clinic champions, OHSU inpatient and outpatient pharmacy and nurse representatives, hospital administrative support, and a CareOregon representive. | |
| At each meeting, we review individual patient cases, seek feedback from diverse, and previously siloed, team members, and engage in ongoing quality improvement. | ||
Needs Assessment
We conducted a mixed‐methods needs assessment of consecutive nonelderly adult inpatients (65 years old) admitted to general medicine and cardiology, between July and October 2009, with no insurance, Medicaid, or MedicareMedicaid. Five volunteer medical and pre‐medical students surveyed 116 patients (see Supporting Information survey, Appendix 2, in the online version of this article). Forty patients reported prior admission within the last 6 months. With these participants, we conducted in‐depth semi‐structured interviews assessing self‐perceived transitional care barriers. Investigators drew preliminary themes from the interviews but delayed a scientifically rigorous qualitative analysis, given a compressed timeline in which to meet program development needs. Of the 116 patients surveyed, 22 had MedicareMedicaid. Given that many of these patients discharged to skilled nursing facilities, we focused program development using data from the 94 uninsured and Medicaid patients (Table 3).
| Uninsured (n = 43 patients) | Medicaid (n = 51 patients) | |
|---|---|---|
| ||
| Lack usual source of care (%) | 33.3 | 11.1* |
| Self‐reported 6 mo rehospitalization (%) | 60.0 | 48.6 |
| Average no. Rx prior to hospitalization | 4.4 | 13.8 |
| Barriers to taking meds as prescribed (%) | 42.9 | 21.6* |
| Cost of meds as leading barrier (%) | 30.0 | 2.9* |
| Marginal housing (%) | 40.5 | 32.4 |
| Low health literacy (%) | 41.5 | 41.7 |
| Transportation barrier (%) | 11.9 | 31.4* |
| Comorbid depression (%) | 54.8 | 45.9 |
| Income 30 K (%) | 79.5 | 96.8 |
Finding 1: Thirty‐three percent of uninsured and 11% of Medicaid patients lacked a usual source of care. This was highest among Portland‐area residents (45%). Program element: We forged relationships with 3 outpatient clinics and developed a contractual relationship whereby OHSU pays for medical homes for uninsured patients lacking usual care. Finding 2: Patients were unclear as to how to self‐manage care or who to contact with questions after hospitalization. Program element: Transitional care nurse provides intensive peridischarge education, performs home visits within 3 days of discharge, and serves as a point person for patients during the peridischarge period. Finding 3: Among uninsured patients, cost was the leading barrier to taking medications as prescribed and often led to self‐rationing of medications without provider input. Program element: We developed a low‐cost, value‐based formulary for uninsured patients that parallels partnering clinic formularies, $4 plans, and medication assistance programs. After 30 days of program‐funded medications, patients then get medications through these other sources. Inpatient pharmacists consult on all patients to reconcile medications, identify access and adherence gaps, provide patient education, and communicate across settings. Finding 4: Comorbid depression was common. Program element: We sought partnerships with clinics with integrated mental health services. Finding 5: Over half of patients live in 3 counties surrounding Portland. Program element: We restricted our intervention to patients residing in local counties and included postdischarge home visits in our model. Partnering clinics match patient geographic distribution. Finding 6: Self‐ reported 6‐month readmission (60%) rates exceeded rates estimated by hospital administrative data (18%), supporting qualitative findings that patients seek care at numerous hospitals. Program element: Given that utilization claims data are unavailable for the uninsured, we included phone follow‐up surveys to assess self‐reported utilization 30 days postdischarge. Finding 7: Using administrative data, we estimated that the hospital loses an average of $11,000 per readmission per patient in direct, unremunerated costs. Indirect costs (such as costs of hospital staff) and opportunity costs (of potential revenue from an insured patient occupying the bed) were excluded, thus presenting a conservative estimate of cost savings. Program element: We used local cost data to support the business case and emphasize potential value of an up‐front investment in transitional care.
Defining the Setting
We convened a series of 3 work group meetings with diverse internal and external stakeholders (Table 4) to further define an intervention in the context of local health system realities. Work groups shaped the program in several specific ways. First, community clinic leaders emphasized that limited specialty access is an important barrier when caring for recently hospitalized uninsured and Medicaid patients. They felt expanded postdischarge access to specialists would be important to increase their capacity for recently discharged patients. Thus, we streamlined patients' posthospital specialty access for conditions treated during hospitalization. Second, initially we considered linking with 1 clinic; however, health systems researchers and clinic providers cautioned us, suggesting that partnering with multiple clinics would make our work more broadly applicable. Finally, pharmacists and financial assistance staff revealed that financial assistance forms are often not completed during hospitalization because inpatients lack access to income documentation. This led us to incorporate help with financial paperwork into the postdischarge intervention.
| Clinical staff |
| Hospital medicine physician |
| General internal medicine physician |
| Hospital ward nurse staff |
| Pharmacy (inpatient, outpatient, medication assistance programs) |
| Care management/social work |
| Emergency medicine |
| Health system leadership |
| Hospital administrative leadership |
| Primary care clinic leadership |
| Safety‐net clinic leadership |
| Specialty clinic leadership |
| Hospital business development and strategic planning |
| CareOregon (Medicaid managed care) leadership |
| Other |
| Patients |
| Health systems researchers |
| Clinical informatics |
| Hospital financials (billing, financial screening, admitting) |
Pilot Testing
We conducted pilot testing over 4 weeks, incorporating a Plan‐Do‐Study‐Act approach. For example, our transitional care nurse initially used an intervention guide with a list of steps outlined; however, we quickly discovered that the multiple and varied needs of this patient populationincluding housing, transportation, and foodwere overwhelming and pulled the nurse in many directions. In consultation with our quality improvement experts, we reframed the intervention guide as a checklist to be completed for each patient.
Pilot testing also underscored the importance of monthly meetings to promote shared learning and create a forum for communication and problem solving across settings. During these meetings, patient case discussions inform continuous quality improvement and promote energy‐sustaining team‐building. Information is then disseminated to each clinic site and arm of the intervention through a designated champion from each group. We also planned to meet monthly with the hospital executive director to balance service and research needs, and engage in rapid‐cycle change throughout our 1‐year demonstration project.
Funding the Program
We talked to others with experience implementing nurse‐led transitional care interventions. Based on these discussions, we anticipated our nurse would be able to see 200 patients over the course of 1 year, and we developed our budget accordingly (Table 2). From our needs assessment, we knew 60% of patients reported at least 1 hospitalization in the 6 months prior. If we assumed that 60% (120) of the 200 patients randomized to our intervention would get readmitted, then a 20% reduction would lead to 24 avoided readmissions and translate into $264,000 in savings for the health system. Even though the hospital would not reap all of these savings, as patients get admitted to other area hospitals, hospital administration acknowledged the value of setting the stage for community‐wide solutions. Moreover, the benefit was felt to extend beyond financial savings to improved quality and institutional learning around transitional care.
PROGRAM EVALUATION
We are conducting a clustered, randomized controlled trial to evaluate C‐TraIn's impact on quality, access, and high‐cost utilization at 30 days after hospital discharge. Results are anticipated in mid‐2012. We chose to perform an analysis clustered by admitting team, because communication between the C‐TraIn nurse, physician team, and pharmacist consult services could introduce secular change effects that could impact the care received by other patients on a given team. There are 5 general medicine resident teams, 1 hospitalist service, and 1 cardiology service, and the physician personnel for each team changes from month to month. Because the cardiology and hospitalist services differ slightly from resident teams, we chose a randomized cross‐over design such that intervention and control teams are redesignated every 3 months. To enhance internal validity, study personnel who enroll patients and administer baseline and 30‐day surveys are blinded to intervention status. We are collecting data on prior utilization, usual source of care, outpatient access, insurance, patient activation,6 functional status,7, 8 self‐rated health,7 health literacy, care transitions education,9 alcohol and substance abuse, and social support.10 Our primary outcome will be self‐reported 30‐day hospital readmission and ED use. We will also evaluate administrative claims data to identify 30‐day OHSU readmission and ED utilization rates. We will assess whether improved access to medications, rates of outpatient follow‐up and time to follow‐up mediate any effect on primary outcomes. Secondary outcomes will include outpatient utilization, patient activation, self‐rated health, and functional status.
Given limited experience with transitional care programs in socioeconomically disadvantaged patients, we are measuring acceptability and feasibility by tracking rates of those declining the intervention, and through semi‐structured interviews at 30 days. We are monitoring fidelity to core elements of the program through chart and checklist reviews, and seeking provider feedback through in‐person meetings with key implementers. To ensure possibility of broader adoption beyond OHSU, we are developing a toolkit that defines core program elements and can be adapted for use in various settings.
DISCUSSION
Using a process of broad stakeholder engagement, exposure of financial incentives, and data‐driven understanding of institutional and population needs, we built consensus and gained institutional financial commitment for implementation of a multicomponent transitional care program for uninsured and Medicaid patients. Our experience is relevant to other hospital systems, and may have particular relevance to academic medical centers, whose tripartite mission of clinical care, research, and education make them a natural place for healthcare reform.11
Several key lessons from our experience may be widely applicable. First, key administrative allies helped us understand institutional priorities and identify key institutional change‐agents. Though initial attempts to gain support were met cautiously, persistent advocacy, development of a strong business case, and support from several administrative allies compelled further leadership support. Second, unlike traditional grant funding cycles, hospital budgets operate in real‐time rapid‐change cycles, necessitating rapid data collection, analysis, and program design. Such demands could potentially threaten the viability of the program itself, or result in premature diffusion of novel practices into disparate populations. Communication with administrative leadership about the value of sound research design within the context of faster‐paced institutional needs was important and allowed time for data‐driven program development and diffusion. Simultaneously, we recognized the need to move quickly, provide regular progress updates, and use existing institutional resources, such as volunteer students and business development office, when possible.
We found that cross‐site hospitalcommunity partnerships are an essential program element. Partnership occurs through a payment agreement and through active engagement in ongoing quality improvement, including clinic representation at monthly team meetings. Clinic partnerships have enabled multidisciplinary cross‐site communication and relationships that facilitate innovation across routinely siloed elements of the system, allowing the team to anticipate and respond to patient problems before they lead to readmissions or poor outcomes. Our experience matches findings from recent program evaluations that found that care coordination attempts are unsuccessful without strong cross‐site linkages.12 These linkages are especially challenging and needed for uninsured and Medicaid patients, given their traditional lack of access and the additional social and financial barriers that influence their care.13
Limitations of our study include: implementation at a single, academic medical center; secular changes (which we mitigate against using randomized trial design); and potential for low power, if readmission rates are lower than anticipated from needs assessment data. Additionally, the need for a willing and invested program champion to coordinate an often messy, complex intervention may limit generalizability.
While transitional care programs continue to proliferate in response to increasingly recognized gaps in a fragmented care system,14, 15 few interventions specifically address the needs of socioeconomically disadvantaged patients. The major study that did5 was conducted in Massachusetts, where many patients received care through a state Free Care program and robust local safety‐net. Others have largely been tested in integrated care settings,1 and target patients who are part of managed care programs.1, 4, 16
To our knowledge, there are no well‐described programs that include explicit purchasing of outpatient medical homes for uninsured patients who would not otherwise have access to care. Our experience shifts the paradigm of the role of hospitals in care for the uninsured and underinsured: instead of a reactive, uncoordinated role, we assert that the hospital's strategic up‐front allocation of resources has a sound business, quality, and ethical foundation. This is especially important, given a new era of payment reform and coordinated care organizations. There is an opportunity to both improve quality for the uninsured and Medicaid patients, control costs, and gain valuable experience that can inform transitional care improvements for broader patient populations. If our study is successful in reducing readmissions, there may be important implications as to how to redefine the hospital's role in outpatient access to care linkages, especially for uninsured and Medicaid patients.
Acknowledgements
The authors acknowledge Char Riley, Dawn Whitney, and Tara Harben of OHSU, as well as volunteer research assistants Amie Leaverton, Molly McClain, Emily Johnson, Travis Geraci, and Claudia Sells.
- ,,,.The care transitions intervention: results of a randomized controlled trial.Arch Intern Med.2006;166(17):1822–1828.
- ,,,,.Medicaid patients at high risk for frequent hospital admission: real‐time identification and remediable risks.J Urban Health.2009;86(2):230–241.
- ,,,,,.Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial.J Am Geriatr Soc.2004;52(5):675–684.
- ,,,,.The effect of Evercare on hospital use.J Am Geriatr Soc.2003;51(10):1427–1434.
- ,,, et al.A reengineered hospital discharge program to decrease rehospitalization: a randomized trial.Ann Intern Med.2009;150(3):178–187.
- ,,,.Development of the patient activation measure (PAM): conceptualizing and measuring activation in patients and consumers.Health Serv Res.2004;39(4 pt 1):1005–1026.
- The EuroQol Group.EuroQol—a new facility for the measurement of health‐related quality of life.Health Policy.1990;16(3):199–208.
- ,,,,,.Trajectories of life‐space mobility after hospitalization.Ann Intern Med.2009;150(6):372–378.
- ,,.Assessing the quality of preparation for posthospital care from the patient's perspective: the care transitions measure.Med Care.2005;43(3):246–255.
- ,,,.Assessing social support: the social support questionnaire.J Pers Soc Psychol.1983;44(1):127–139.
- .Payment reform and the mission of academic medical centers.N Engl J Med.2010;363(19):1784–1786.
- ,,,.Effects of care coordination on hospitalization, quality of care, and health care expenditures among Medicare beneficiaries: 15 randomized trials.JAMA.2009;301(6):603–618.
- ,,,.Post‐discharge intervention in vulnerable, chronically ill patients.J Hosp Med.2012;7(2):124–130.
- ,,, et al.Discharge planning from hospital to home.Cochrane Database Syst Rev.2010(1):000313.
- .Preventing the rebound: improving care transition in hospital discharge processes.Aust Health Rev.2010;34(4):445–451.
- ,,, et al.Comprehensive discharge planning and home follow‐up of hospitalized elders: a randomized clinical trial.JAMA.1999;281(7):613–620.
Hospital readmissions are common and costly, and represent a significant burden to the healthcare system. The challenges of postdischarge medication uncertainty, lack of self‐management support, and lack of timely access to health professionals1 are compounded in uninsured and Medicaid individuals by limited access to medications and primary care, financial strain, insecure housing, and limited social support.2
Our hospital cares for a large number of uninsured and low‐income publicly insured patients. The Portland area safety‐net, which consists of a network of 14 federally qualified health centers and free clinics, has limited capacity for uncompensated care. Uninsured patientsand to a lesser degree, Medicaid patientshave difficulty establishing primary care. Prior to the implementation of our program, uninsured and Medicaid patients without a usual source of care were given a list of safety‐net clinics at discharge, but frequently could not access appointments or navigate the complex system. There were no well‐developed partnerships between hospital and outpatient clinics for uninsured or Medicaid patients. The hospital lacked a systematic approach to securing postdischarge follow‐up and peridischarge patient education, and uninsured patients were financially responsible for most medications upon discharge. The costs of uncompensated or undercompensated potentially preventable readmissions for these patients, along with the recognition of gaps in quality, ultimately provided the rationale for a medical center‐funded transitional care intervention for uninsured and low‐income publicly insured patients.
Several transitional care improvement programs have shown effectiveness in reducing hospital readmissions,1, 35 but most have been conducted in settings where patients have secure access to outpatient care, and none have focused specifically on uninsured or Medicaid patients. Moreover, the development of these programs requires time and capital. Transitional care programs that have published results, to date, have been funded through government or private foundation grants1, 35; however, broader implementation of transitional care innovations will require financial and intellectual engagement of healthcare institutions themselves.
This report describes development of the Care Transitions Innovation (C‐TraIn), a multicomponent transitional care intervention for uninsured and low‐income publicly insured adults at a large, urban academic medical center, Oregon Health & Science University (OHSU). Because institutional funding and engagement is critical to the sustainability and scalability of similar programs, we also describe our process for gaining institutional support. Our hypothesis is that C‐TraIn can reduce readmissions and emergency department (ED) use at 30 days after hospital discharge, compared with usual care.
METHODS
Engaging Institutional Leaders
Early and continued efforts to engage hospital administrators were integral to our ultimate success in gaining institutional funding and leadership support. Initially, we convened what we called a Health Systems Morbidity and Mortality conference, featuring an uninsured patient who told of his postdischarge experiences and costly, potentially preventable readmission. We invited a broad array of potential stakeholders, including representatives from hospital administration, hospital case managers and social workers, community safety‐net providers, inpatient and outpatient physicians, residents, and medical students. Our patient was previously admitted to OHSU and diagnosed with pneumonia, hypothyroidism, sleep apnea, and depression. At discharge, he was given a list of low‐cost clinics; however, he was unable to arrange follow‐up, could not afford prescriptions, and felt overwhelmed trying to navigate a complex system. Consequently, he received no outpatient healthcare and his illnesses progressed. Unable to stay awake as a long‐haul trucker, he lost his job and subsequently his housing, and was readmitted to the intensive care unit with severe hypercarbic respiratory failure, volume overload, and hypothyroidism. The $130,000 charge for his 19‐day rehospitalization was largely un‐recuperated by the hospital. The case was a stark example of the patient‐safety and financial costs of fragmented care, and the conference was a nidus for further institutional engagement and program development, the key steps of which are described in Table 1.
| Time | Key Step | How Step Was Achieved | Take Home Points |
|---|---|---|---|
| |||
| July 2008July 2009 | 1. Identified key stakeholders | Considered varied stakeholders impacted by transitional care gaps for uninsured and Medicaid patients | Casting a wide net early in the process promoted high level of engagement and allowed self‐identification of some stakeholders |
| 2. Framed problems and opportunities; exposed costs of existing system shortcomings | Educational conference (that we called a Health Systems M&M) fostered a blame‐free environment to explore varied perspectives | Individual patient story made policy issue more accessible to a wide range of stakeholders | |
| Discussion of exposed drivers and costs of misaligned incentives; highlighted inroads to developing a business case for change | |||
| Oct 2008June 2009 | 3. Identified administrative allies and leaders with high bridging capital | Follow‐up with administrator after Health System M&M allowed further identification of key administrative stakeholders | Administrator insight highlighted institutional priorities and strategic plans |
| Ongoing meetings over 9 moto advocate for change, explore support for program development | Key ally within administration facilitated conversation with executive leadership whose support was a critical for program success | ||
| July 2009June 2010 | 4. Framed processes locally with continued involvement from multiple stakeholders | Performed multicomponent needs assessment | Patient assessment included inpatients for ease of survey administration |
| Utilized efforts of student volunteers for low‐budget option | |||
| Existing administrative support aided patient tracking | |||
| Non‐integrated health system and lack of claims data for uninsured limited usefulness of administrative utilization data | |||
| 5. Performed cost analysis to further support the business and quality case | Used OHSU data from needs assessment patient sample to estimate potential costs and savings of saved readmissions and avoided ED visits | Business case highlighted existing costs to OHSU for uncompensated care; program presented a solution to realign incentives and better allocate existing hospital expenditures | |
| Qualitative patient interviews exposed opportunity for quality improvement | Highlighted pilot as an opportunity for institutional learning about transitional care improvements | ||
| 6. Use needs assessment to map intervention | Drew upon local and national health systems expertise through literature review and consultation with local and national program leaders | OHSU's Care Transitions Innovation (C‐TraIn) includes elements aimed at improving access, patient education, care coordination, and systems integration (Table 2) | |
| Matched patient needs to specific elements of program design | |||
Planning the Intervention
Findings from a patient needs assessment and community stakeholder meetingsdescribed belowdirectly informed a multicomponent intervention that includes linkages and payment for medical homes for uninsured patients who lack access to outpatient care, a transitional care nurse whose care bridges inpatient and outpatient settings, inpatient pharmacy consultation, and provision of 30 days of medications at hospital discharge for uninsured patients (Table 2).
| Program Element | Description | Resources per 200 Patients |
|---|---|---|
| ||
| Transitional care RN | Augments patient education and care coordination in the hospital until 30 days after discharge. Tasks include: | 1.0 FTE nurse salary* |
| developing a personal health record with inpatients | ||
| completing a home visit within 72 hr of discharge to focus on medication reconciliation and patient self‐management | ||
| low‐risk patients receive 3 calls and no home visit (see Supporting Information, Appendix 1, in the online version of this article) | ||
| 2 subsequent phone calls to provide additional coaching, identify unmet needs, and close the loop on incomplete financial paperwork | ||
| The nurse provides a warm handoff with clinic staff, assists in scheduling timely posthospital follow‐up, and assures timely transfer of DC summaries. She coordinates posthospital care management with Medicaid case‐workers when available. | ||
| Pharmacy | Consultation: Inpatient pharmacists reconcile and simplify medication regimens, educate patients, and assess adherence barriers. | 0.4 FTE inpatient pharmacist salary |
| Prescription support: For uninsured patients, pharmacists guide MD prescribing towards medications available on the C‐TraIn value‐based formulary, a low‐cost formulary that reflects medications available through $4 plans, a Medicaid formulary, and FQHC on‐site pharmacies. | Estimated $12/prescription; 6.5 prescriptions/patient | |
| Uninsured patients are given 30 days of bridging prescription medications at hospital discharge free of charge. | ||
| Outpatient medical home and specialty care linkages | OHSU has partnered with outpatient clinics on a per‐patient basis to support funding of primary care for uninsured patients who lack a usual source of care. Clinics also provide coordinated care for Medicaid patients without assigned primary care, and have committed to engaging in continuous quality improvement. Clinics include an academic general internal medicine practice, an FQHC specializing in addiction and care for the homeless, and an FQHC that serves a low‐income rural population. | Estimated 8 primary care visits/yr at $205/visit (FQHC reimbursement rate) equates to $1640/ patient/yr. |
| Timely posthospital specialty care related to index admission diagnoses is coordinated through OHSU's outpatient specialty clinics. | ||
| Monthly care coordination meetings | We convene a diverse team of community clinic champions, OHSU inpatient and outpatient pharmacy and nurse representatives, hospital administrative support, and a CareOregon representive. | |
| At each meeting, we review individual patient cases, seek feedback from diverse, and previously siloed, team members, and engage in ongoing quality improvement. | ||
Needs Assessment
We conducted a mixed‐methods needs assessment of consecutive nonelderly adult inpatients (65 years old) admitted to general medicine and cardiology, between July and October 2009, with no insurance, Medicaid, or MedicareMedicaid. Five volunteer medical and pre‐medical students surveyed 116 patients (see Supporting Information survey, Appendix 2, in the online version of this article). Forty patients reported prior admission within the last 6 months. With these participants, we conducted in‐depth semi‐structured interviews assessing self‐perceived transitional care barriers. Investigators drew preliminary themes from the interviews but delayed a scientifically rigorous qualitative analysis, given a compressed timeline in which to meet program development needs. Of the 116 patients surveyed, 22 had MedicareMedicaid. Given that many of these patients discharged to skilled nursing facilities, we focused program development using data from the 94 uninsured and Medicaid patients (Table 3).
| Uninsured (n = 43 patients) | Medicaid (n = 51 patients) | |
|---|---|---|
| ||
| Lack usual source of care (%) | 33.3 | 11.1* |
| Self‐reported 6 mo rehospitalization (%) | 60.0 | 48.6 |
| Average no. Rx prior to hospitalization | 4.4 | 13.8 |
| Barriers to taking meds as prescribed (%) | 42.9 | 21.6* |
| Cost of meds as leading barrier (%) | 30.0 | 2.9* |
| Marginal housing (%) | 40.5 | 32.4 |
| Low health literacy (%) | 41.5 | 41.7 |
| Transportation barrier (%) | 11.9 | 31.4* |
| Comorbid depression (%) | 54.8 | 45.9 |
| Income 30 K (%) | 79.5 | 96.8 |
Finding 1: Thirty‐three percent of uninsured and 11% of Medicaid patients lacked a usual source of care. This was highest among Portland‐area residents (45%). Program element: We forged relationships with 3 outpatient clinics and developed a contractual relationship whereby OHSU pays for medical homes for uninsured patients lacking usual care. Finding 2: Patients were unclear as to how to self‐manage care or who to contact with questions after hospitalization. Program element: Transitional care nurse provides intensive peridischarge education, performs home visits within 3 days of discharge, and serves as a point person for patients during the peridischarge period. Finding 3: Among uninsured patients, cost was the leading barrier to taking medications as prescribed and often led to self‐rationing of medications without provider input. Program element: We developed a low‐cost, value‐based formulary for uninsured patients that parallels partnering clinic formularies, $4 plans, and medication assistance programs. After 30 days of program‐funded medications, patients then get medications through these other sources. Inpatient pharmacists consult on all patients to reconcile medications, identify access and adherence gaps, provide patient education, and communicate across settings. Finding 4: Comorbid depression was common. Program element: We sought partnerships with clinics with integrated mental health services. Finding 5: Over half of patients live in 3 counties surrounding Portland. Program element: We restricted our intervention to patients residing in local counties and included postdischarge home visits in our model. Partnering clinics match patient geographic distribution. Finding 6: Self‐ reported 6‐month readmission (60%) rates exceeded rates estimated by hospital administrative data (18%), supporting qualitative findings that patients seek care at numerous hospitals. Program element: Given that utilization claims data are unavailable for the uninsured, we included phone follow‐up surveys to assess self‐reported utilization 30 days postdischarge. Finding 7: Using administrative data, we estimated that the hospital loses an average of $11,000 per readmission per patient in direct, unremunerated costs. Indirect costs (such as costs of hospital staff) and opportunity costs (of potential revenue from an insured patient occupying the bed) were excluded, thus presenting a conservative estimate of cost savings. Program element: We used local cost data to support the business case and emphasize potential value of an up‐front investment in transitional care.
Defining the Setting
We convened a series of 3 work group meetings with diverse internal and external stakeholders (Table 4) to further define an intervention in the context of local health system realities. Work groups shaped the program in several specific ways. First, community clinic leaders emphasized that limited specialty access is an important barrier when caring for recently hospitalized uninsured and Medicaid patients. They felt expanded postdischarge access to specialists would be important to increase their capacity for recently discharged patients. Thus, we streamlined patients' posthospital specialty access for conditions treated during hospitalization. Second, initially we considered linking with 1 clinic; however, health systems researchers and clinic providers cautioned us, suggesting that partnering with multiple clinics would make our work more broadly applicable. Finally, pharmacists and financial assistance staff revealed that financial assistance forms are often not completed during hospitalization because inpatients lack access to income documentation. This led us to incorporate help with financial paperwork into the postdischarge intervention.
| Clinical staff |
| Hospital medicine physician |
| General internal medicine physician |
| Hospital ward nurse staff |
| Pharmacy (inpatient, outpatient, medication assistance programs) |
| Care management/social work |
| Emergency medicine |
| Health system leadership |
| Hospital administrative leadership |
| Primary care clinic leadership |
| Safety‐net clinic leadership |
| Specialty clinic leadership |
| Hospital business development and strategic planning |
| CareOregon (Medicaid managed care) leadership |
| Other |
| Patients |
| Health systems researchers |
| Clinical informatics |
| Hospital financials (billing, financial screening, admitting) |
Pilot Testing
We conducted pilot testing over 4 weeks, incorporating a Plan‐Do‐Study‐Act approach. For example, our transitional care nurse initially used an intervention guide with a list of steps outlined; however, we quickly discovered that the multiple and varied needs of this patient populationincluding housing, transportation, and foodwere overwhelming and pulled the nurse in many directions. In consultation with our quality improvement experts, we reframed the intervention guide as a checklist to be completed for each patient.
Pilot testing also underscored the importance of monthly meetings to promote shared learning and create a forum for communication and problem solving across settings. During these meetings, patient case discussions inform continuous quality improvement and promote energy‐sustaining team‐building. Information is then disseminated to each clinic site and arm of the intervention through a designated champion from each group. We also planned to meet monthly with the hospital executive director to balance service and research needs, and engage in rapid‐cycle change throughout our 1‐year demonstration project.
Funding the Program
We talked to others with experience implementing nurse‐led transitional care interventions. Based on these discussions, we anticipated our nurse would be able to see 200 patients over the course of 1 year, and we developed our budget accordingly (Table 2). From our needs assessment, we knew 60% of patients reported at least 1 hospitalization in the 6 months prior. If we assumed that 60% (120) of the 200 patients randomized to our intervention would get readmitted, then a 20% reduction would lead to 24 avoided readmissions and translate into $264,000 in savings for the health system. Even though the hospital would not reap all of these savings, as patients get admitted to other area hospitals, hospital administration acknowledged the value of setting the stage for community‐wide solutions. Moreover, the benefit was felt to extend beyond financial savings to improved quality and institutional learning around transitional care.
PROGRAM EVALUATION
We are conducting a clustered, randomized controlled trial to evaluate C‐TraIn's impact on quality, access, and high‐cost utilization at 30 days after hospital discharge. Results are anticipated in mid‐2012. We chose to perform an analysis clustered by admitting team, because communication between the C‐TraIn nurse, physician team, and pharmacist consult services could introduce secular change effects that could impact the care received by other patients on a given team. There are 5 general medicine resident teams, 1 hospitalist service, and 1 cardiology service, and the physician personnel for each team changes from month to month. Because the cardiology and hospitalist services differ slightly from resident teams, we chose a randomized cross‐over design such that intervention and control teams are redesignated every 3 months. To enhance internal validity, study personnel who enroll patients and administer baseline and 30‐day surveys are blinded to intervention status. We are collecting data on prior utilization, usual source of care, outpatient access, insurance, patient activation,6 functional status,7, 8 self‐rated health,7 health literacy, care transitions education,9 alcohol and substance abuse, and social support.10 Our primary outcome will be self‐reported 30‐day hospital readmission and ED use. We will also evaluate administrative claims data to identify 30‐day OHSU readmission and ED utilization rates. We will assess whether improved access to medications, rates of outpatient follow‐up and time to follow‐up mediate any effect on primary outcomes. Secondary outcomes will include outpatient utilization, patient activation, self‐rated health, and functional status.
Given limited experience with transitional care programs in socioeconomically disadvantaged patients, we are measuring acceptability and feasibility by tracking rates of those declining the intervention, and through semi‐structured interviews at 30 days. We are monitoring fidelity to core elements of the program through chart and checklist reviews, and seeking provider feedback through in‐person meetings with key implementers. To ensure possibility of broader adoption beyond OHSU, we are developing a toolkit that defines core program elements and can be adapted for use in various settings.
DISCUSSION
Using a process of broad stakeholder engagement, exposure of financial incentives, and data‐driven understanding of institutional and population needs, we built consensus and gained institutional financial commitment for implementation of a multicomponent transitional care program for uninsured and Medicaid patients. Our experience is relevant to other hospital systems, and may have particular relevance to academic medical centers, whose tripartite mission of clinical care, research, and education make them a natural place for healthcare reform.11
Several key lessons from our experience may be widely applicable. First, key administrative allies helped us understand institutional priorities and identify key institutional change‐agents. Though initial attempts to gain support were met cautiously, persistent advocacy, development of a strong business case, and support from several administrative allies compelled further leadership support. Second, unlike traditional grant funding cycles, hospital budgets operate in real‐time rapid‐change cycles, necessitating rapid data collection, analysis, and program design. Such demands could potentially threaten the viability of the program itself, or result in premature diffusion of novel practices into disparate populations. Communication with administrative leadership about the value of sound research design within the context of faster‐paced institutional needs was important and allowed time for data‐driven program development and diffusion. Simultaneously, we recognized the need to move quickly, provide regular progress updates, and use existing institutional resources, such as volunteer students and business development office, when possible.
We found that cross‐site hospitalcommunity partnerships are an essential program element. Partnership occurs through a payment agreement and through active engagement in ongoing quality improvement, including clinic representation at monthly team meetings. Clinic partnerships have enabled multidisciplinary cross‐site communication and relationships that facilitate innovation across routinely siloed elements of the system, allowing the team to anticipate and respond to patient problems before they lead to readmissions or poor outcomes. Our experience matches findings from recent program evaluations that found that care coordination attempts are unsuccessful without strong cross‐site linkages.12 These linkages are especially challenging and needed for uninsured and Medicaid patients, given their traditional lack of access and the additional social and financial barriers that influence their care.13
Limitations of our study include: implementation at a single, academic medical center; secular changes (which we mitigate against using randomized trial design); and potential for low power, if readmission rates are lower than anticipated from needs assessment data. Additionally, the need for a willing and invested program champion to coordinate an often messy, complex intervention may limit generalizability.
While transitional care programs continue to proliferate in response to increasingly recognized gaps in a fragmented care system,14, 15 few interventions specifically address the needs of socioeconomically disadvantaged patients. The major study that did5 was conducted in Massachusetts, where many patients received care through a state Free Care program and robust local safety‐net. Others have largely been tested in integrated care settings,1 and target patients who are part of managed care programs.1, 4, 16
To our knowledge, there are no well‐described programs that include explicit purchasing of outpatient medical homes for uninsured patients who would not otherwise have access to care. Our experience shifts the paradigm of the role of hospitals in care for the uninsured and underinsured: instead of a reactive, uncoordinated role, we assert that the hospital's strategic up‐front allocation of resources has a sound business, quality, and ethical foundation. This is especially important, given a new era of payment reform and coordinated care organizations. There is an opportunity to both improve quality for the uninsured and Medicaid patients, control costs, and gain valuable experience that can inform transitional care improvements for broader patient populations. If our study is successful in reducing readmissions, there may be important implications as to how to redefine the hospital's role in outpatient access to care linkages, especially for uninsured and Medicaid patients.
Acknowledgements
The authors acknowledge Char Riley, Dawn Whitney, and Tara Harben of OHSU, as well as volunteer research assistants Amie Leaverton, Molly McClain, Emily Johnson, Travis Geraci, and Claudia Sells.
Hospital readmissions are common and costly, and represent a significant burden to the healthcare system. The challenges of postdischarge medication uncertainty, lack of self‐management support, and lack of timely access to health professionals1 are compounded in uninsured and Medicaid individuals by limited access to medications and primary care, financial strain, insecure housing, and limited social support.2
Our hospital cares for a large number of uninsured and low‐income publicly insured patients. The Portland area safety‐net, which consists of a network of 14 federally qualified health centers and free clinics, has limited capacity for uncompensated care. Uninsured patientsand to a lesser degree, Medicaid patientshave difficulty establishing primary care. Prior to the implementation of our program, uninsured and Medicaid patients without a usual source of care were given a list of safety‐net clinics at discharge, but frequently could not access appointments or navigate the complex system. There were no well‐developed partnerships between hospital and outpatient clinics for uninsured or Medicaid patients. The hospital lacked a systematic approach to securing postdischarge follow‐up and peridischarge patient education, and uninsured patients were financially responsible for most medications upon discharge. The costs of uncompensated or undercompensated potentially preventable readmissions for these patients, along with the recognition of gaps in quality, ultimately provided the rationale for a medical center‐funded transitional care intervention for uninsured and low‐income publicly insured patients.
Several transitional care improvement programs have shown effectiveness in reducing hospital readmissions,1, 35 but most have been conducted in settings where patients have secure access to outpatient care, and none have focused specifically on uninsured or Medicaid patients. Moreover, the development of these programs requires time and capital. Transitional care programs that have published results, to date, have been funded through government or private foundation grants1, 35; however, broader implementation of transitional care innovations will require financial and intellectual engagement of healthcare institutions themselves.
This report describes development of the Care Transitions Innovation (C‐TraIn), a multicomponent transitional care intervention for uninsured and low‐income publicly insured adults at a large, urban academic medical center, Oregon Health & Science University (OHSU). Because institutional funding and engagement is critical to the sustainability and scalability of similar programs, we also describe our process for gaining institutional support. Our hypothesis is that C‐TraIn can reduce readmissions and emergency department (ED) use at 30 days after hospital discharge, compared with usual care.
METHODS
Engaging Institutional Leaders
Early and continued efforts to engage hospital administrators were integral to our ultimate success in gaining institutional funding and leadership support. Initially, we convened what we called a Health Systems Morbidity and Mortality conference, featuring an uninsured patient who told of his postdischarge experiences and costly, potentially preventable readmission. We invited a broad array of potential stakeholders, including representatives from hospital administration, hospital case managers and social workers, community safety‐net providers, inpatient and outpatient physicians, residents, and medical students. Our patient was previously admitted to OHSU and diagnosed with pneumonia, hypothyroidism, sleep apnea, and depression. At discharge, he was given a list of low‐cost clinics; however, he was unable to arrange follow‐up, could not afford prescriptions, and felt overwhelmed trying to navigate a complex system. Consequently, he received no outpatient healthcare and his illnesses progressed. Unable to stay awake as a long‐haul trucker, he lost his job and subsequently his housing, and was readmitted to the intensive care unit with severe hypercarbic respiratory failure, volume overload, and hypothyroidism. The $130,000 charge for his 19‐day rehospitalization was largely un‐recuperated by the hospital. The case was a stark example of the patient‐safety and financial costs of fragmented care, and the conference was a nidus for further institutional engagement and program development, the key steps of which are described in Table 1.
| Time | Key Step | How Step Was Achieved | Take Home Points |
|---|---|---|---|
| |||
| July 2008July 2009 | 1. Identified key stakeholders | Considered varied stakeholders impacted by transitional care gaps for uninsured and Medicaid patients | Casting a wide net early in the process promoted high level of engagement and allowed self‐identification of some stakeholders |
| 2. Framed problems and opportunities; exposed costs of existing system shortcomings | Educational conference (that we called a Health Systems M&M) fostered a blame‐free environment to explore varied perspectives | Individual patient story made policy issue more accessible to a wide range of stakeholders | |
| Discussion of exposed drivers and costs of misaligned incentives; highlighted inroads to developing a business case for change | |||
| Oct 2008June 2009 | 3. Identified administrative allies and leaders with high bridging capital | Follow‐up with administrator after Health System M&M allowed further identification of key administrative stakeholders | Administrator insight highlighted institutional priorities and strategic plans |
| Ongoing meetings over 9 moto advocate for change, explore support for program development | Key ally within administration facilitated conversation with executive leadership whose support was a critical for program success | ||
| July 2009June 2010 | 4. Framed processes locally with continued involvement from multiple stakeholders | Performed multicomponent needs assessment | Patient assessment included inpatients for ease of survey administration |
| Utilized efforts of student volunteers for low‐budget option | |||
| Existing administrative support aided patient tracking | |||
| Non‐integrated health system and lack of claims data for uninsured limited usefulness of administrative utilization data | |||
| 5. Performed cost analysis to further support the business and quality case | Used OHSU data from needs assessment patient sample to estimate potential costs and savings of saved readmissions and avoided ED visits | Business case highlighted existing costs to OHSU for uncompensated care; program presented a solution to realign incentives and better allocate existing hospital expenditures | |
| Qualitative patient interviews exposed opportunity for quality improvement | Highlighted pilot as an opportunity for institutional learning about transitional care improvements | ||
| 6. Use needs assessment to map intervention | Drew upon local and national health systems expertise through literature review and consultation with local and national program leaders | OHSU's Care Transitions Innovation (C‐TraIn) includes elements aimed at improving access, patient education, care coordination, and systems integration (Table 2) | |
| Matched patient needs to specific elements of program design | |||
Planning the Intervention
Findings from a patient needs assessment and community stakeholder meetingsdescribed belowdirectly informed a multicomponent intervention that includes linkages and payment for medical homes for uninsured patients who lack access to outpatient care, a transitional care nurse whose care bridges inpatient and outpatient settings, inpatient pharmacy consultation, and provision of 30 days of medications at hospital discharge for uninsured patients (Table 2).
| Program Element | Description | Resources per 200 Patients |
|---|---|---|
| ||
| Transitional care RN | Augments patient education and care coordination in the hospital until 30 days after discharge. Tasks include: | 1.0 FTE nurse salary* |
| developing a personal health record with inpatients | ||
| completing a home visit within 72 hr of discharge to focus on medication reconciliation and patient self‐management | ||
| low‐risk patients receive 3 calls and no home visit (see Supporting Information, Appendix 1, in the online version of this article) | ||
| 2 subsequent phone calls to provide additional coaching, identify unmet needs, and close the loop on incomplete financial paperwork | ||
| The nurse provides a warm handoff with clinic staff, assists in scheduling timely posthospital follow‐up, and assures timely transfer of DC summaries. She coordinates posthospital care management with Medicaid case‐workers when available. | ||
| Pharmacy | Consultation: Inpatient pharmacists reconcile and simplify medication regimens, educate patients, and assess adherence barriers. | 0.4 FTE inpatient pharmacist salary |
| Prescription support: For uninsured patients, pharmacists guide MD prescribing towards medications available on the C‐TraIn value‐based formulary, a low‐cost formulary that reflects medications available through $4 plans, a Medicaid formulary, and FQHC on‐site pharmacies. | Estimated $12/prescription; 6.5 prescriptions/patient | |
| Uninsured patients are given 30 days of bridging prescription medications at hospital discharge free of charge. | ||
| Outpatient medical home and specialty care linkages | OHSU has partnered with outpatient clinics on a per‐patient basis to support funding of primary care for uninsured patients who lack a usual source of care. Clinics also provide coordinated care for Medicaid patients without assigned primary care, and have committed to engaging in continuous quality improvement. Clinics include an academic general internal medicine practice, an FQHC specializing in addiction and care for the homeless, and an FQHC that serves a low‐income rural population. | Estimated 8 primary care visits/yr at $205/visit (FQHC reimbursement rate) equates to $1640/ patient/yr. |
| Timely posthospital specialty care related to index admission diagnoses is coordinated through OHSU's outpatient specialty clinics. | ||
| Monthly care coordination meetings | We convene a diverse team of community clinic champions, OHSU inpatient and outpatient pharmacy and nurse representatives, hospital administrative support, and a CareOregon representive. | |
| At each meeting, we review individual patient cases, seek feedback from diverse, and previously siloed, team members, and engage in ongoing quality improvement. | ||
Needs Assessment
We conducted a mixed‐methods needs assessment of consecutive nonelderly adult inpatients (65 years old) admitted to general medicine and cardiology, between July and October 2009, with no insurance, Medicaid, or MedicareMedicaid. Five volunteer medical and pre‐medical students surveyed 116 patients (see Supporting Information survey, Appendix 2, in the online version of this article). Forty patients reported prior admission within the last 6 months. With these participants, we conducted in‐depth semi‐structured interviews assessing self‐perceived transitional care barriers. Investigators drew preliminary themes from the interviews but delayed a scientifically rigorous qualitative analysis, given a compressed timeline in which to meet program development needs. Of the 116 patients surveyed, 22 had MedicareMedicaid. Given that many of these patients discharged to skilled nursing facilities, we focused program development using data from the 94 uninsured and Medicaid patients (Table 3).
| Uninsured (n = 43 patients) | Medicaid (n = 51 patients) | |
|---|---|---|
| ||
| Lack usual source of care (%) | 33.3 | 11.1* |
| Self‐reported 6 mo rehospitalization (%) | 60.0 | 48.6 |
| Average no. Rx prior to hospitalization | 4.4 | 13.8 |
| Barriers to taking meds as prescribed (%) | 42.9 | 21.6* |
| Cost of meds as leading barrier (%) | 30.0 | 2.9* |
| Marginal housing (%) | 40.5 | 32.4 |
| Low health literacy (%) | 41.5 | 41.7 |
| Transportation barrier (%) | 11.9 | 31.4* |
| Comorbid depression (%) | 54.8 | 45.9 |
| Income 30 K (%) | 79.5 | 96.8 |
Finding 1: Thirty‐three percent of uninsured and 11% of Medicaid patients lacked a usual source of care. This was highest among Portland‐area residents (45%). Program element: We forged relationships with 3 outpatient clinics and developed a contractual relationship whereby OHSU pays for medical homes for uninsured patients lacking usual care. Finding 2: Patients were unclear as to how to self‐manage care or who to contact with questions after hospitalization. Program element: Transitional care nurse provides intensive peridischarge education, performs home visits within 3 days of discharge, and serves as a point person for patients during the peridischarge period. Finding 3: Among uninsured patients, cost was the leading barrier to taking medications as prescribed and often led to self‐rationing of medications without provider input. Program element: We developed a low‐cost, value‐based formulary for uninsured patients that parallels partnering clinic formularies, $4 plans, and medication assistance programs. After 30 days of program‐funded medications, patients then get medications through these other sources. Inpatient pharmacists consult on all patients to reconcile medications, identify access and adherence gaps, provide patient education, and communicate across settings. Finding 4: Comorbid depression was common. Program element: We sought partnerships with clinics with integrated mental health services. Finding 5: Over half of patients live in 3 counties surrounding Portland. Program element: We restricted our intervention to patients residing in local counties and included postdischarge home visits in our model. Partnering clinics match patient geographic distribution. Finding 6: Self‐ reported 6‐month readmission (60%) rates exceeded rates estimated by hospital administrative data (18%), supporting qualitative findings that patients seek care at numerous hospitals. Program element: Given that utilization claims data are unavailable for the uninsured, we included phone follow‐up surveys to assess self‐reported utilization 30 days postdischarge. Finding 7: Using administrative data, we estimated that the hospital loses an average of $11,000 per readmission per patient in direct, unremunerated costs. Indirect costs (such as costs of hospital staff) and opportunity costs (of potential revenue from an insured patient occupying the bed) were excluded, thus presenting a conservative estimate of cost savings. Program element: We used local cost data to support the business case and emphasize potential value of an up‐front investment in transitional care.
Defining the Setting
We convened a series of 3 work group meetings with diverse internal and external stakeholders (Table 4) to further define an intervention in the context of local health system realities. Work groups shaped the program in several specific ways. First, community clinic leaders emphasized that limited specialty access is an important barrier when caring for recently hospitalized uninsured and Medicaid patients. They felt expanded postdischarge access to specialists would be important to increase their capacity for recently discharged patients. Thus, we streamlined patients' posthospital specialty access for conditions treated during hospitalization. Second, initially we considered linking with 1 clinic; however, health systems researchers and clinic providers cautioned us, suggesting that partnering with multiple clinics would make our work more broadly applicable. Finally, pharmacists and financial assistance staff revealed that financial assistance forms are often not completed during hospitalization because inpatients lack access to income documentation. This led us to incorporate help with financial paperwork into the postdischarge intervention.
| Clinical staff |
| Hospital medicine physician |
| General internal medicine physician |
| Hospital ward nurse staff |
| Pharmacy (inpatient, outpatient, medication assistance programs) |
| Care management/social work |
| Emergency medicine |
| Health system leadership |
| Hospital administrative leadership |
| Primary care clinic leadership |
| Safety‐net clinic leadership |
| Specialty clinic leadership |
| Hospital business development and strategic planning |
| CareOregon (Medicaid managed care) leadership |
| Other |
| Patients |
| Health systems researchers |
| Clinical informatics |
| Hospital financials (billing, financial screening, admitting) |
Pilot Testing
We conducted pilot testing over 4 weeks, incorporating a Plan‐Do‐Study‐Act approach. For example, our transitional care nurse initially used an intervention guide with a list of steps outlined; however, we quickly discovered that the multiple and varied needs of this patient populationincluding housing, transportation, and foodwere overwhelming and pulled the nurse in many directions. In consultation with our quality improvement experts, we reframed the intervention guide as a checklist to be completed for each patient.
Pilot testing also underscored the importance of monthly meetings to promote shared learning and create a forum for communication and problem solving across settings. During these meetings, patient case discussions inform continuous quality improvement and promote energy‐sustaining team‐building. Information is then disseminated to each clinic site and arm of the intervention through a designated champion from each group. We also planned to meet monthly with the hospital executive director to balance service and research needs, and engage in rapid‐cycle change throughout our 1‐year demonstration project.
Funding the Program
We talked to others with experience implementing nurse‐led transitional care interventions. Based on these discussions, we anticipated our nurse would be able to see 200 patients over the course of 1 year, and we developed our budget accordingly (Table 2). From our needs assessment, we knew 60% of patients reported at least 1 hospitalization in the 6 months prior. If we assumed that 60% (120) of the 200 patients randomized to our intervention would get readmitted, then a 20% reduction would lead to 24 avoided readmissions and translate into $264,000 in savings for the health system. Even though the hospital would not reap all of these savings, as patients get admitted to other area hospitals, hospital administration acknowledged the value of setting the stage for community‐wide solutions. Moreover, the benefit was felt to extend beyond financial savings to improved quality and institutional learning around transitional care.
PROGRAM EVALUATION
We are conducting a clustered, randomized controlled trial to evaluate C‐TraIn's impact on quality, access, and high‐cost utilization at 30 days after hospital discharge. Results are anticipated in mid‐2012. We chose to perform an analysis clustered by admitting team, because communication between the C‐TraIn nurse, physician team, and pharmacist consult services could introduce secular change effects that could impact the care received by other patients on a given team. There are 5 general medicine resident teams, 1 hospitalist service, and 1 cardiology service, and the physician personnel for each team changes from month to month. Because the cardiology and hospitalist services differ slightly from resident teams, we chose a randomized cross‐over design such that intervention and control teams are redesignated every 3 months. To enhance internal validity, study personnel who enroll patients and administer baseline and 30‐day surveys are blinded to intervention status. We are collecting data on prior utilization, usual source of care, outpatient access, insurance, patient activation,6 functional status,7, 8 self‐rated health,7 health literacy, care transitions education,9 alcohol and substance abuse, and social support.10 Our primary outcome will be self‐reported 30‐day hospital readmission and ED use. We will also evaluate administrative claims data to identify 30‐day OHSU readmission and ED utilization rates. We will assess whether improved access to medications, rates of outpatient follow‐up and time to follow‐up mediate any effect on primary outcomes. Secondary outcomes will include outpatient utilization, patient activation, self‐rated health, and functional status.
Given limited experience with transitional care programs in socioeconomically disadvantaged patients, we are measuring acceptability and feasibility by tracking rates of those declining the intervention, and through semi‐structured interviews at 30 days. We are monitoring fidelity to core elements of the program through chart and checklist reviews, and seeking provider feedback through in‐person meetings with key implementers. To ensure possibility of broader adoption beyond OHSU, we are developing a toolkit that defines core program elements and can be adapted for use in various settings.
DISCUSSION
Using a process of broad stakeholder engagement, exposure of financial incentives, and data‐driven understanding of institutional and population needs, we built consensus and gained institutional financial commitment for implementation of a multicomponent transitional care program for uninsured and Medicaid patients. Our experience is relevant to other hospital systems, and may have particular relevance to academic medical centers, whose tripartite mission of clinical care, research, and education make them a natural place for healthcare reform.11
Several key lessons from our experience may be widely applicable. First, key administrative allies helped us understand institutional priorities and identify key institutional change‐agents. Though initial attempts to gain support were met cautiously, persistent advocacy, development of a strong business case, and support from several administrative allies compelled further leadership support. Second, unlike traditional grant funding cycles, hospital budgets operate in real‐time rapid‐change cycles, necessitating rapid data collection, analysis, and program design. Such demands could potentially threaten the viability of the program itself, or result in premature diffusion of novel practices into disparate populations. Communication with administrative leadership about the value of sound research design within the context of faster‐paced institutional needs was important and allowed time for data‐driven program development and diffusion. Simultaneously, we recognized the need to move quickly, provide regular progress updates, and use existing institutional resources, such as volunteer students and business development office, when possible.
We found that cross‐site hospitalcommunity partnerships are an essential program element. Partnership occurs through a payment agreement and through active engagement in ongoing quality improvement, including clinic representation at monthly team meetings. Clinic partnerships have enabled multidisciplinary cross‐site communication and relationships that facilitate innovation across routinely siloed elements of the system, allowing the team to anticipate and respond to patient problems before they lead to readmissions or poor outcomes. Our experience matches findings from recent program evaluations that found that care coordination attempts are unsuccessful without strong cross‐site linkages.12 These linkages are especially challenging and needed for uninsured and Medicaid patients, given their traditional lack of access and the additional social and financial barriers that influence their care.13
Limitations of our study include: implementation at a single, academic medical center; secular changes (which we mitigate against using randomized trial design); and potential for low power, if readmission rates are lower than anticipated from needs assessment data. Additionally, the need for a willing and invested program champion to coordinate an often messy, complex intervention may limit generalizability.
While transitional care programs continue to proliferate in response to increasingly recognized gaps in a fragmented care system,14, 15 few interventions specifically address the needs of socioeconomically disadvantaged patients. The major study that did5 was conducted in Massachusetts, where many patients received care through a state Free Care program and robust local safety‐net. Others have largely been tested in integrated care settings,1 and target patients who are part of managed care programs.1, 4, 16
To our knowledge, there are no well‐described programs that include explicit purchasing of outpatient medical homes for uninsured patients who would not otherwise have access to care. Our experience shifts the paradigm of the role of hospitals in care for the uninsured and underinsured: instead of a reactive, uncoordinated role, we assert that the hospital's strategic up‐front allocation of resources has a sound business, quality, and ethical foundation. This is especially important, given a new era of payment reform and coordinated care organizations. There is an opportunity to both improve quality for the uninsured and Medicaid patients, control costs, and gain valuable experience that can inform transitional care improvements for broader patient populations. If our study is successful in reducing readmissions, there may be important implications as to how to redefine the hospital's role in outpatient access to care linkages, especially for uninsured and Medicaid patients.
Acknowledgements
The authors acknowledge Char Riley, Dawn Whitney, and Tara Harben of OHSU, as well as volunteer research assistants Amie Leaverton, Molly McClain, Emily Johnson, Travis Geraci, and Claudia Sells.
- ,,,.The care transitions intervention: results of a randomized controlled trial.Arch Intern Med.2006;166(17):1822–1828.
- ,,,,.Medicaid patients at high risk for frequent hospital admission: real‐time identification and remediable risks.J Urban Health.2009;86(2):230–241.
- ,,,,,.Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial.J Am Geriatr Soc.2004;52(5):675–684.
- ,,,,.The effect of Evercare on hospital use.J Am Geriatr Soc.2003;51(10):1427–1434.
- ,,, et al.A reengineered hospital discharge program to decrease rehospitalization: a randomized trial.Ann Intern Med.2009;150(3):178–187.
- ,,,.Development of the patient activation measure (PAM): conceptualizing and measuring activation in patients and consumers.Health Serv Res.2004;39(4 pt 1):1005–1026.
- The EuroQol Group.EuroQol—a new facility for the measurement of health‐related quality of life.Health Policy.1990;16(3):199–208.
- ,,,,,.Trajectories of life‐space mobility after hospitalization.Ann Intern Med.2009;150(6):372–378.
- ,,.Assessing the quality of preparation for posthospital care from the patient's perspective: the care transitions measure.Med Care.2005;43(3):246–255.
- ,,,.Assessing social support: the social support questionnaire.J Pers Soc Psychol.1983;44(1):127–139.
- .Payment reform and the mission of academic medical centers.N Engl J Med.2010;363(19):1784–1786.
- ,,,.Effects of care coordination on hospitalization, quality of care, and health care expenditures among Medicare beneficiaries: 15 randomized trials.JAMA.2009;301(6):603–618.
- ,,,.Post‐discharge intervention in vulnerable, chronically ill patients.J Hosp Med.2012;7(2):124–130.
- ,,, et al.Discharge planning from hospital to home.Cochrane Database Syst Rev.2010(1):000313.
- .Preventing the rebound: improving care transition in hospital discharge processes.Aust Health Rev.2010;34(4):445–451.
- ,,, et al.Comprehensive discharge planning and home follow‐up of hospitalized elders: a randomized clinical trial.JAMA.1999;281(7):613–620.
- ,,,.The care transitions intervention: results of a randomized controlled trial.Arch Intern Med.2006;166(17):1822–1828.
- ,,,,.Medicaid patients at high risk for frequent hospital admission: real‐time identification and remediable risks.J Urban Health.2009;86(2):230–241.
- ,,,,,.Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial.J Am Geriatr Soc.2004;52(5):675–684.
- ,,,,.The effect of Evercare on hospital use.J Am Geriatr Soc.2003;51(10):1427–1434.
- ,,, et al.A reengineered hospital discharge program to decrease rehospitalization: a randomized trial.Ann Intern Med.2009;150(3):178–187.
- ,,,.Development of the patient activation measure (PAM): conceptualizing and measuring activation in patients and consumers.Health Serv Res.2004;39(4 pt 1):1005–1026.
- The EuroQol Group.EuroQol—a new facility for the measurement of health‐related quality of life.Health Policy.1990;16(3):199–208.
- ,,,,,.Trajectories of life‐space mobility after hospitalization.Ann Intern Med.2009;150(6):372–378.
- ,,.Assessing the quality of preparation for posthospital care from the patient's perspective: the care transitions measure.Med Care.2005;43(3):246–255.
- ,,,.Assessing social support: the social support questionnaire.J Pers Soc Psychol.1983;44(1):127–139.
- .Payment reform and the mission of academic medical centers.N Engl J Med.2010;363(19):1784–1786.
- ,,,.Effects of care coordination on hospitalization, quality of care, and health care expenditures among Medicare beneficiaries: 15 randomized trials.JAMA.2009;301(6):603–618.
- ,,,.Post‐discharge intervention in vulnerable, chronically ill patients.J Hosp Med.2012;7(2):124–130.
- ,,, et al.Discharge planning from hospital to home.Cochrane Database Syst Rev.2010(1):000313.
- .Preventing the rebound: improving care transition in hospital discharge processes.Aust Health Rev.2010;34(4):445–451.
- ,,, et al.Comprehensive discharge planning and home follow‐up of hospitalized elders: a randomized clinical trial.JAMA.1999;281(7):613–620.
FDA Announces Nationwide Recall of Three Cytarabine Lots
Three lots of the antineoplastic drug cytarabine have been recalled because they may not be sterile, according to a posting on the Food and Drug Administration’s MedWatch site.
The nationwide recall applies to lots of cytarabine for injection (1 gm/vial), manufactured by Bedford Laboratories. The risk for lack of sterility was determined from an investigation of the manufacturing area, after the product was released, according to the FDA.
The affected lot numbers are: 2066986, 2111675, and 2131148. Any hospital, physician, clinic, or other health care facility that has any of the affected products should not use it for patients and "should immediately quarantine any product for return," the statement said. Wholesalers, distributors, or retailers that have the recalled product should stop using it and contact the company at 800-562-4797. Click here to report the product to the FDA Medwatch program.
Bedford is one of several generic manufacturers of cytarabine.
Cytarabine is approved for use in combination with other approved cancer treatments for inducing remission of acute nonlymphocytic leukemia in children and adults, and has also been found to be useful in treating acute lymphocytic leukemia and the blast phase of chronic myelocytic leukemia. It is also indicated in the prophylaxis and treatment of meningeal leukemia, administered intrathecally for this indication.
To view the notice, visit the MedWatch site.
Three lots of the antineoplastic drug cytarabine have been recalled because they may not be sterile, according to a posting on the Food and Drug Administration’s MedWatch site.
The nationwide recall applies to lots of cytarabine for injection (1 gm/vial), manufactured by Bedford Laboratories. The risk for lack of sterility was determined from an investigation of the manufacturing area, after the product was released, according to the FDA.
The affected lot numbers are: 2066986, 2111675, and 2131148. Any hospital, physician, clinic, or other health care facility that has any of the affected products should not use it for patients and "should immediately quarantine any product for return," the statement said. Wholesalers, distributors, or retailers that have the recalled product should stop using it and contact the company at 800-562-4797. Click here to report the product to the FDA Medwatch program.
Bedford is one of several generic manufacturers of cytarabine.
Cytarabine is approved for use in combination with other approved cancer treatments for inducing remission of acute nonlymphocytic leukemia in children and adults, and has also been found to be useful in treating acute lymphocytic leukemia and the blast phase of chronic myelocytic leukemia. It is also indicated in the prophylaxis and treatment of meningeal leukemia, administered intrathecally for this indication.
To view the notice, visit the MedWatch site.
Three lots of the antineoplastic drug cytarabine have been recalled because they may not be sterile, according to a posting on the Food and Drug Administration’s MedWatch site.
The nationwide recall applies to lots of cytarabine for injection (1 gm/vial), manufactured by Bedford Laboratories. The risk for lack of sterility was determined from an investigation of the manufacturing area, after the product was released, according to the FDA.
The affected lot numbers are: 2066986, 2111675, and 2131148. Any hospital, physician, clinic, or other health care facility that has any of the affected products should not use it for patients and "should immediately quarantine any product for return," the statement said. Wholesalers, distributors, or retailers that have the recalled product should stop using it and contact the company at 800-562-4797. Click here to report the product to the FDA Medwatch program.
Bedford is one of several generic manufacturers of cytarabine.
Cytarabine is approved for use in combination with other approved cancer treatments for inducing remission of acute nonlymphocytic leukemia in children and adults, and has also been found to be useful in treating acute lymphocytic leukemia and the blast phase of chronic myelocytic leukemia. It is also indicated in the prophylaxis and treatment of meningeal leukemia, administered intrathecally for this indication.
To view the notice, visit the MedWatch site.
Duodenal Switch May Excel at Type 2 Diabetes Resolution
MADISON, WIS. – Total complication rates are high but comparable over the long term between duodenal switch surgery and Roux-en-Y gastric bypass, according to a propensity matched analysis of 309 superobese patients.
"Duodenal switch is a valid alternative to the Roux-en-Y gastric bypass, especially if significant comorbid illnesses are present, particularly diabetes," Dr. Robert B. Dorman said.
His conclusion is drawn from a study that focused on the long-term outcomes of 178 consecutive patients who underwent duodenal switch (DS) surgery and 139 propensity matched patients undergoing Roux-en-Y gastric bypass (RYGB). In addition to a chart review, the University of Minnesota Bariatric Surgery Outcomes Survey tool was used to prospectively track patients’ weight, comorbid illnesses, adverse outcomes, readmissions, and general health status. Mean follow-up was 3.7 years in the DS group and 6.2 years in the RYGB group.
There were five deaths in the DS group (postop day 38 and months 5, 7, 16, and 66) and three deaths in the RYGB group (postop months 3, 7, and 72), leaving 173 patients and 136 patients, respectively, in the analysis, Dr. Dorman said at the annual meeting of the Central Surgical Association.
Notably, weight loss in the two groups was comparable, decreasing from an average body mass index of 52 kg/m2 to 31 kg/m2 in the DS group and from 51 kg/m2 to 34 kg/m2 in the RYGB group, said Dr. Dorman, a general surgery resident at the University of Minnesota, Minneapolis.
Resolution of type 2 diabetes, hypertension, and hyperlipidemia was greatest among DS patients at 82%, 67%, and 81%, respectively, compared with 64%, 39%, and 55%, respectively, among RYGB patients.
DS patients, however, experienced significantly more loose stools, bloating, and heartburn than did RYGB patients, who had significantly more constipation. Nausea and emesis were comparable between the two groups.
With regard to complications, DS patients were significantly more likely to visit the emergency department (ED) than were RYGB patients (40% vs. 25%; P value less than .01) and to experience hair loss (67% vs. 41%; P less than .01), Dr. Dorman said.
There was also a nonsignificant trend for DS patients to be readmitted more often than RYGB patients (25.4% vs. 23.5%) and to have more gastrointestinal leaks (1.7% vs. 0%), abdominal reoperations (29% vs. 23%), total parenteral nutrition/tube feeds (7.6% vs. 3%), and infusion therapy (28.5% vs. 23.5%). The RYGB patients, however, underwent more endoscopy (22% vs. 14%).
Dr. Dorman said providers should explain to patients the adverse symptoms they can expect following duodenal switch, but noted that the investigators "still feel DS should be limited to surgeons and centers with experience."
Invited discussant Dr. James Wallace, a bariatric and general surgeon from the Medical College of Wisconsin, Milwaukee, described the 40% rate of ED visits in the DS group as "extreme," and questioned the use of nutritional, vitamin, and protein supplementation – particularly in light of the observed hair loss.
"I commend the authors for their excellent surgical outcomes with the duodenal switch – much better than others have reported in the literature – but I’m unconvinced that the incremental improvement in weight loss and resolution of metabolic derangements justifies the increased nutritional risk of the duodenal switch," he said.
Dr. Dorman responded that the ED visits may represent a "knee-jerk reflex" on the part of DS patients when they experience a complication. He added that the university has instituted more frequent checks and phone calls, particularly to high-risk DS patients, and has partnered with their transplant clinic’s infusion center to provide IV fluids. Nutritional support data in this study was insufficient to report on for all patients, but supplementary nutrition is implemented based on factors such as vitamin and albumin levels or difficulty with eating, according to Dr. Dorman.
Invited discussant Dr. Bradley Needleman, director of the bariatric surgery program at Ohio State University in Columbus, said he was most fascinated by the lack of a significant difference in weight loss between the two groups and asked how this finding would influence patient consultations.
Dr. Dorman said a recent case-matched study at their institution also found no significant difference in weight loss between the two procedures (Ann. Surg. 2012;255:287-93), although a recent prospective randomized European study reported that weight loss was significantly greater with duodenal switch surgery than with gastric bypass (Ann. Intern. Med. 2011;155:281-91).
"It seems to be a finding that exists only within our institution and that should be taken into account when we discuss with our own patients which operations they should undergo," Dr. Dorman said. "I think that duodenal switch in a patient with diabetes and BMI over 45-50 [kg/m2] is certainly a valid operation at an experienced center, as long as we understand the symptoms they may experience afterward."
Duodenal switch should remain a valid alternative because RYGB has its own inherent downfalls – notably, high marginal ulcer and stricture rates, as indicated in the current study by the trend toward significantly greater use of endoscopic procedures in the RYGB patients, said Dr. Sayeed Ikramuddin. Also, the group has now twice shown in matched patient populations the superiority by which the duodenal switch provides resolution of type 2 diabetes when compared with RYGB, added Dr. Ikramuddin, senior author and director of gastrointestinal surgery at the University of Minnesota.
"Lastly, the Roux-en-Y gastric bypass has a high long-term failure rate resulting in patients regaining their weight," Dr. Ikramuddin said in an interview. "This is a phenomenon not as common among duodenal switch patients, likely due to the more malabsorptive nature of the operation."
When asked what contraindications exist for duodenal switch surgery, Dr. Dorman replied that the only absolute contraindications are women of reproductive age because of concerns of nutritional malabsorption and patients unwilling to commit to follow-up. A patient with significant gastroesophageal reflux disease would most likely be offered RYGB, he said, noting that DS surgery had been performed on 5%-10% of their last 100 GI patients.
Dr. Dorman reported no relevant conflicts of interest.
MADISON, WIS. – Total complication rates are high but comparable over the long term between duodenal switch surgery and Roux-en-Y gastric bypass, according to a propensity matched analysis of 309 superobese patients.
"Duodenal switch is a valid alternative to the Roux-en-Y gastric bypass, especially if significant comorbid illnesses are present, particularly diabetes," Dr. Robert B. Dorman said.
His conclusion is drawn from a study that focused on the long-term outcomes of 178 consecutive patients who underwent duodenal switch (DS) surgery and 139 propensity matched patients undergoing Roux-en-Y gastric bypass (RYGB). In addition to a chart review, the University of Minnesota Bariatric Surgery Outcomes Survey tool was used to prospectively track patients’ weight, comorbid illnesses, adverse outcomes, readmissions, and general health status. Mean follow-up was 3.7 years in the DS group and 6.2 years in the RYGB group.
There were five deaths in the DS group (postop day 38 and months 5, 7, 16, and 66) and three deaths in the RYGB group (postop months 3, 7, and 72), leaving 173 patients and 136 patients, respectively, in the analysis, Dr. Dorman said at the annual meeting of the Central Surgical Association.
Notably, weight loss in the two groups was comparable, decreasing from an average body mass index of 52 kg/m2 to 31 kg/m2 in the DS group and from 51 kg/m2 to 34 kg/m2 in the RYGB group, said Dr. Dorman, a general surgery resident at the University of Minnesota, Minneapolis.
Resolution of type 2 diabetes, hypertension, and hyperlipidemia was greatest among DS patients at 82%, 67%, and 81%, respectively, compared with 64%, 39%, and 55%, respectively, among RYGB patients.
DS patients, however, experienced significantly more loose stools, bloating, and heartburn than did RYGB patients, who had significantly more constipation. Nausea and emesis were comparable between the two groups.
With regard to complications, DS patients were significantly more likely to visit the emergency department (ED) than were RYGB patients (40% vs. 25%; P value less than .01) and to experience hair loss (67% vs. 41%; P less than .01), Dr. Dorman said.
There was also a nonsignificant trend for DS patients to be readmitted more often than RYGB patients (25.4% vs. 23.5%) and to have more gastrointestinal leaks (1.7% vs. 0%), abdominal reoperations (29% vs. 23%), total parenteral nutrition/tube feeds (7.6% vs. 3%), and infusion therapy (28.5% vs. 23.5%). The RYGB patients, however, underwent more endoscopy (22% vs. 14%).
Dr. Dorman said providers should explain to patients the adverse symptoms they can expect following duodenal switch, but noted that the investigators "still feel DS should be limited to surgeons and centers with experience."
Invited discussant Dr. James Wallace, a bariatric and general surgeon from the Medical College of Wisconsin, Milwaukee, described the 40% rate of ED visits in the DS group as "extreme," and questioned the use of nutritional, vitamin, and protein supplementation – particularly in light of the observed hair loss.
"I commend the authors for their excellent surgical outcomes with the duodenal switch – much better than others have reported in the literature – but I’m unconvinced that the incremental improvement in weight loss and resolution of metabolic derangements justifies the increased nutritional risk of the duodenal switch," he said.
Dr. Dorman responded that the ED visits may represent a "knee-jerk reflex" on the part of DS patients when they experience a complication. He added that the university has instituted more frequent checks and phone calls, particularly to high-risk DS patients, and has partnered with their transplant clinic’s infusion center to provide IV fluids. Nutritional support data in this study was insufficient to report on for all patients, but supplementary nutrition is implemented based on factors such as vitamin and albumin levels or difficulty with eating, according to Dr. Dorman.
Invited discussant Dr. Bradley Needleman, director of the bariatric surgery program at Ohio State University in Columbus, said he was most fascinated by the lack of a significant difference in weight loss between the two groups and asked how this finding would influence patient consultations.
Dr. Dorman said a recent case-matched study at their institution also found no significant difference in weight loss between the two procedures (Ann. Surg. 2012;255:287-93), although a recent prospective randomized European study reported that weight loss was significantly greater with duodenal switch surgery than with gastric bypass (Ann. Intern. Med. 2011;155:281-91).
"It seems to be a finding that exists only within our institution and that should be taken into account when we discuss with our own patients which operations they should undergo," Dr. Dorman said. "I think that duodenal switch in a patient with diabetes and BMI over 45-50 [kg/m2] is certainly a valid operation at an experienced center, as long as we understand the symptoms they may experience afterward."
Duodenal switch should remain a valid alternative because RYGB has its own inherent downfalls – notably, high marginal ulcer and stricture rates, as indicated in the current study by the trend toward significantly greater use of endoscopic procedures in the RYGB patients, said Dr. Sayeed Ikramuddin. Also, the group has now twice shown in matched patient populations the superiority by which the duodenal switch provides resolution of type 2 diabetes when compared with RYGB, added Dr. Ikramuddin, senior author and director of gastrointestinal surgery at the University of Minnesota.
"Lastly, the Roux-en-Y gastric bypass has a high long-term failure rate resulting in patients regaining their weight," Dr. Ikramuddin said in an interview. "This is a phenomenon not as common among duodenal switch patients, likely due to the more malabsorptive nature of the operation."
When asked what contraindications exist for duodenal switch surgery, Dr. Dorman replied that the only absolute contraindications are women of reproductive age because of concerns of nutritional malabsorption and patients unwilling to commit to follow-up. A patient with significant gastroesophageal reflux disease would most likely be offered RYGB, he said, noting that DS surgery had been performed on 5%-10% of their last 100 GI patients.
Dr. Dorman reported no relevant conflicts of interest.
MADISON, WIS. – Total complication rates are high but comparable over the long term between duodenal switch surgery and Roux-en-Y gastric bypass, according to a propensity matched analysis of 309 superobese patients.
"Duodenal switch is a valid alternative to the Roux-en-Y gastric bypass, especially if significant comorbid illnesses are present, particularly diabetes," Dr. Robert B. Dorman said.
His conclusion is drawn from a study that focused on the long-term outcomes of 178 consecutive patients who underwent duodenal switch (DS) surgery and 139 propensity matched patients undergoing Roux-en-Y gastric bypass (RYGB). In addition to a chart review, the University of Minnesota Bariatric Surgery Outcomes Survey tool was used to prospectively track patients’ weight, comorbid illnesses, adverse outcomes, readmissions, and general health status. Mean follow-up was 3.7 years in the DS group and 6.2 years in the RYGB group.
There were five deaths in the DS group (postop day 38 and months 5, 7, 16, and 66) and three deaths in the RYGB group (postop months 3, 7, and 72), leaving 173 patients and 136 patients, respectively, in the analysis, Dr. Dorman said at the annual meeting of the Central Surgical Association.
Notably, weight loss in the two groups was comparable, decreasing from an average body mass index of 52 kg/m2 to 31 kg/m2 in the DS group and from 51 kg/m2 to 34 kg/m2 in the RYGB group, said Dr. Dorman, a general surgery resident at the University of Minnesota, Minneapolis.
Resolution of type 2 diabetes, hypertension, and hyperlipidemia was greatest among DS patients at 82%, 67%, and 81%, respectively, compared with 64%, 39%, and 55%, respectively, among RYGB patients.
DS patients, however, experienced significantly more loose stools, bloating, and heartburn than did RYGB patients, who had significantly more constipation. Nausea and emesis were comparable between the two groups.
With regard to complications, DS patients were significantly more likely to visit the emergency department (ED) than were RYGB patients (40% vs. 25%; P value less than .01) and to experience hair loss (67% vs. 41%; P less than .01), Dr. Dorman said.
There was also a nonsignificant trend for DS patients to be readmitted more often than RYGB patients (25.4% vs. 23.5%) and to have more gastrointestinal leaks (1.7% vs. 0%), abdominal reoperations (29% vs. 23%), total parenteral nutrition/tube feeds (7.6% vs. 3%), and infusion therapy (28.5% vs. 23.5%). The RYGB patients, however, underwent more endoscopy (22% vs. 14%).
Dr. Dorman said providers should explain to patients the adverse symptoms they can expect following duodenal switch, but noted that the investigators "still feel DS should be limited to surgeons and centers with experience."
Invited discussant Dr. James Wallace, a bariatric and general surgeon from the Medical College of Wisconsin, Milwaukee, described the 40% rate of ED visits in the DS group as "extreme," and questioned the use of nutritional, vitamin, and protein supplementation – particularly in light of the observed hair loss.
"I commend the authors for their excellent surgical outcomes with the duodenal switch – much better than others have reported in the literature – but I’m unconvinced that the incremental improvement in weight loss and resolution of metabolic derangements justifies the increased nutritional risk of the duodenal switch," he said.
Dr. Dorman responded that the ED visits may represent a "knee-jerk reflex" on the part of DS patients when they experience a complication. He added that the university has instituted more frequent checks and phone calls, particularly to high-risk DS patients, and has partnered with their transplant clinic’s infusion center to provide IV fluids. Nutritional support data in this study was insufficient to report on for all patients, but supplementary nutrition is implemented based on factors such as vitamin and albumin levels or difficulty with eating, according to Dr. Dorman.
Invited discussant Dr. Bradley Needleman, director of the bariatric surgery program at Ohio State University in Columbus, said he was most fascinated by the lack of a significant difference in weight loss between the two groups and asked how this finding would influence patient consultations.
Dr. Dorman said a recent case-matched study at their institution also found no significant difference in weight loss between the two procedures (Ann. Surg. 2012;255:287-93), although a recent prospective randomized European study reported that weight loss was significantly greater with duodenal switch surgery than with gastric bypass (Ann. Intern. Med. 2011;155:281-91).
"It seems to be a finding that exists only within our institution and that should be taken into account when we discuss with our own patients which operations they should undergo," Dr. Dorman said. "I think that duodenal switch in a patient with diabetes and BMI over 45-50 [kg/m2] is certainly a valid operation at an experienced center, as long as we understand the symptoms they may experience afterward."
Duodenal switch should remain a valid alternative because RYGB has its own inherent downfalls – notably, high marginal ulcer and stricture rates, as indicated in the current study by the trend toward significantly greater use of endoscopic procedures in the RYGB patients, said Dr. Sayeed Ikramuddin. Also, the group has now twice shown in matched patient populations the superiority by which the duodenal switch provides resolution of type 2 diabetes when compared with RYGB, added Dr. Ikramuddin, senior author and director of gastrointestinal surgery at the University of Minnesota.
"Lastly, the Roux-en-Y gastric bypass has a high long-term failure rate resulting in patients regaining their weight," Dr. Ikramuddin said in an interview. "This is a phenomenon not as common among duodenal switch patients, likely due to the more malabsorptive nature of the operation."
When asked what contraindications exist for duodenal switch surgery, Dr. Dorman replied that the only absolute contraindications are women of reproductive age because of concerns of nutritional malabsorption and patients unwilling to commit to follow-up. A patient with significant gastroesophageal reflux disease would most likely be offered RYGB, he said, noting that DS surgery had been performed on 5%-10% of their last 100 GI patients.
Dr. Dorman reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE CENTRAL SURGICAL ASSOCIATION
Major Finding: Resolution of type 2 diabetes, hypertension, and hyperlipidemia was greatest among duodenal switch patients, at 82%, 67%, and 81%, vs. 64%, 39%, and 55% among Roux-en-Y gastric bypass patients.
Data Source: Data were taken from a chart review and prospective survey of 309 superobese patients.
Disclosures: Dr. Dorman reported no relevant conflicts of interest.
ONLINE EXCLUSIVE: How the School of Medicine at Stanford University Is Addressing Women Physicians and Leadership
Whenever Hannah Valantine, MD, needs reassurance that women leadership interventions at Stanford University’s School of Medicine are working, she looks at the numbers.
In the span of five to six years, the medical school increased the percentage of women at each faculty rank so that it now surpasses national averages as calculated by the Association of American Medical Colleges. Indeed, the percentage of women at the full professor rank jumped from 14.5 percent to 22 percent.
“We really are making progress,” says Dr. Valantine, full professor of medicine and the medical school’s senior associate dean for diversity and leadership.
With structural elements such as tenure clock extension, extended maternity and family leave, onsite childcare, early stage research funding support, and mentoring in place, Dr. Valantine is turning her attention to the next round of interventions, which focus more on psychological and social factors impairing women’s advancement.
She will use a National Institutes of Health grant to develop interventions for the phenomenon of stereotype threat, which is the fear that one's behavior will confirm an existing stereotype about one’s social group. This fear may lead to an impairment of performance.
Over the next six months, Dr. Valantine and her team will also conduct several pilot programs involving map career customization, a model that encourages people to chart their career over the next 5 to 10 to 20 years, taking into consideration their life outside of work. The intent is to help individuals identify their priorities and goals and how they change over time, and also help supervisors better match the ebbs and flows of a person’s life to the workplace and identify and develop aspiring leaders.
Stanford’s medical school is organized around teams of doctors that care for groups of patients. Each team must achieve excellence in four academic missions: clinical care, education, research, and administration. The map career customization pilot programs are aimed at helping doctors within the team plan their career path around these four missions and then putting the individual plans together in a team context in order to meet the team’s goals, says Dr. Valantine.
“This way the work and the four missions are entirely covered,” she says. “We create a vibrant academic environment where we create new things and have time to think and integrate our life and work… It’s a little countercultural, but I think people are crying out for that and I think it stands a great chance of making the culture change.”
Stanford’s burgeoning efforts in map career customization have intrigued SHM board member Janet Nagamine, RN, MD, FHM, a hospitalist at Kaiser Permanente Medical Center in Santa Clara, Calif., and Stanford alum.
She hopes to collaborate with Dr. Valantine and incorporate in hospital medicine the interventions that Stanford is doing while conducting studies and developing workforce planning initiatives specific to hospitalists. The goal is to create a hospital medicine model that replicates Stanford’s success in cultivating women physician leaders.
“We make this false assumption that your career is going to look the same throughout your life and that’s just not realistic,” Dr. Nagamine says.
Whenever Hannah Valantine, MD, needs reassurance that women leadership interventions at Stanford University’s School of Medicine are working, she looks at the numbers.
In the span of five to six years, the medical school increased the percentage of women at each faculty rank so that it now surpasses national averages as calculated by the Association of American Medical Colleges. Indeed, the percentage of women at the full professor rank jumped from 14.5 percent to 22 percent.
“We really are making progress,” says Dr. Valantine, full professor of medicine and the medical school’s senior associate dean for diversity and leadership.
With structural elements such as tenure clock extension, extended maternity and family leave, onsite childcare, early stage research funding support, and mentoring in place, Dr. Valantine is turning her attention to the next round of interventions, which focus more on psychological and social factors impairing women’s advancement.
She will use a National Institutes of Health grant to develop interventions for the phenomenon of stereotype threat, which is the fear that one's behavior will confirm an existing stereotype about one’s social group. This fear may lead to an impairment of performance.
Over the next six months, Dr. Valantine and her team will also conduct several pilot programs involving map career customization, a model that encourages people to chart their career over the next 5 to 10 to 20 years, taking into consideration their life outside of work. The intent is to help individuals identify their priorities and goals and how they change over time, and also help supervisors better match the ebbs and flows of a person’s life to the workplace and identify and develop aspiring leaders.
Stanford’s medical school is organized around teams of doctors that care for groups of patients. Each team must achieve excellence in four academic missions: clinical care, education, research, and administration. The map career customization pilot programs are aimed at helping doctors within the team plan their career path around these four missions and then putting the individual plans together in a team context in order to meet the team’s goals, says Dr. Valantine.
“This way the work and the four missions are entirely covered,” she says. “We create a vibrant academic environment where we create new things and have time to think and integrate our life and work… It’s a little countercultural, but I think people are crying out for that and I think it stands a great chance of making the culture change.”
Stanford’s burgeoning efforts in map career customization have intrigued SHM board member Janet Nagamine, RN, MD, FHM, a hospitalist at Kaiser Permanente Medical Center in Santa Clara, Calif., and Stanford alum.
She hopes to collaborate with Dr. Valantine and incorporate in hospital medicine the interventions that Stanford is doing while conducting studies and developing workforce planning initiatives specific to hospitalists. The goal is to create a hospital medicine model that replicates Stanford’s success in cultivating women physician leaders.
“We make this false assumption that your career is going to look the same throughout your life and that’s just not realistic,” Dr. Nagamine says.
Whenever Hannah Valantine, MD, needs reassurance that women leadership interventions at Stanford University’s School of Medicine are working, she looks at the numbers.
In the span of five to six years, the medical school increased the percentage of women at each faculty rank so that it now surpasses national averages as calculated by the Association of American Medical Colleges. Indeed, the percentage of women at the full professor rank jumped from 14.5 percent to 22 percent.
“We really are making progress,” says Dr. Valantine, full professor of medicine and the medical school’s senior associate dean for diversity and leadership.
With structural elements such as tenure clock extension, extended maternity and family leave, onsite childcare, early stage research funding support, and mentoring in place, Dr. Valantine is turning her attention to the next round of interventions, which focus more on psychological and social factors impairing women’s advancement.
She will use a National Institutes of Health grant to develop interventions for the phenomenon of stereotype threat, which is the fear that one's behavior will confirm an existing stereotype about one’s social group. This fear may lead to an impairment of performance.
Over the next six months, Dr. Valantine and her team will also conduct several pilot programs involving map career customization, a model that encourages people to chart their career over the next 5 to 10 to 20 years, taking into consideration their life outside of work. The intent is to help individuals identify their priorities and goals and how they change over time, and also help supervisors better match the ebbs and flows of a person’s life to the workplace and identify and develop aspiring leaders.
Stanford’s medical school is organized around teams of doctors that care for groups of patients. Each team must achieve excellence in four academic missions: clinical care, education, research, and administration. The map career customization pilot programs are aimed at helping doctors within the team plan their career path around these four missions and then putting the individual plans together in a team context in order to meet the team’s goals, says Dr. Valantine.
“This way the work and the four missions are entirely covered,” she says. “We create a vibrant academic environment where we create new things and have time to think and integrate our life and work… It’s a little countercultural, but I think people are crying out for that and I think it stands a great chance of making the culture change.”
Stanford’s burgeoning efforts in map career customization have intrigued SHM board member Janet Nagamine, RN, MD, FHM, a hospitalist at Kaiser Permanente Medical Center in Santa Clara, Calif., and Stanford alum.
She hopes to collaborate with Dr. Valantine and incorporate in hospital medicine the interventions that Stanford is doing while conducting studies and developing workforce planning initiatives specific to hospitalists. The goal is to create a hospital medicine model that replicates Stanford’s success in cultivating women physician leaders.
“We make this false assumption that your career is going to look the same throughout your life and that’s just not realistic,” Dr. Nagamine says.
CDI: The Scope of the Problem
Clostridium difficile is a gram‐positive, spore‐forming, toxin‐producing, anaerobic bacillus that was established as the causative pathogen of most cases of antibiotic‐associated colitis in 1978. 1, 2 The spectrum of possible clinical presentations of C. difficile range from asymptomatic colonization, uncomplicated diarrhea, severe pseudomembranous colitis, paralytic ileus, to sepsis and death, with a mortality rate upwards of 80% in fulminant cases requiring colectomy. 3
Vegetative C. difficile cells die rapidly on dry surfaces, but they have been found to remain viable for up to 6 hours on moist surfaces in room air. 4 Spores shed from the gastrointestinal (GI) tract, however, are highly resistant to common hospital disinfectants, and can survive in the environment for many months. 2 C. difficile spores are primarily transmitted from patient to patient on the hands or equipment of healthcare workers. 2 Once spores are ingested and reach the GI tract, they germinate in the vegetative form. 2, 5 In the GI tract, C. difficile causes disease by the production of toxins, primarily toxins A and B, both of which cause severe inflammation. 5 Toxin A attracts neutrophils and monocytes, and toxin B breaks down colonic epithelial cells. 5 Both of these mechanisms lead to colitis, formation of pseudomembranes, and watery diarrhea. 5
After alteration of the healthy colonic bacterial flora, the immune response to C. difficile toxins appears to play a major role in determining host susceptibility to C. difficile infection (CDI). 5, 6 Those with antitoxin immunity are more likely to become symptomless carriers than patients without preexisting immunity. 3 More than 60% of healthy adults have protective immunity against a primary CDI, demonstrated by detectable serum IgG and IgA to both toxins A and B, as a consequence of childhood immunity or frequent exposure to C. difficile in the environment. 3 After a primary episode of CDI, many patients acquire protective immunity against C. difficile toxins, seen as significantly higher serum concentrations of IgM against C. difficile toxin by the third day from onset of diarrhea, and significantly higher serum concentrations of IgG against toxin A by the 12th day. 7 Patients who experience recurrent CDI lack development of this protective immunity to C. difficile. 6, 7
CDI INCIDENCE IS ON THE RISE
During the past decade, rates of CDI have increased steadily to levels not previously seen. A report published by the Agency of Healthcare Research and Quality demonstrated that the number of CDI diagnoses on hospital discharge more than doubled in the United States from 139,000 to 301,200 between 2000 and 2005 (Figure 1). 8 Examination of a more recent Nationwide Inpatient Sample (NIS) indicates continuation of this trend, with nearly 350,000 CDI diagnoses recorded upon discharge from acute care hospitals in 2008. 9 Of note, in 2006 the state of Ohio mandated CDI reporting from both hospitals and nursing homes. It was estimated there were more than 18,000 cases of CDI during this year, of which more than 60% were diagnosed in nursing homes. 10 Based on the 2008 NIS data and the data from Ohio, it is conceivable there were as many as 1 million cases of CDI in the US in 2008.
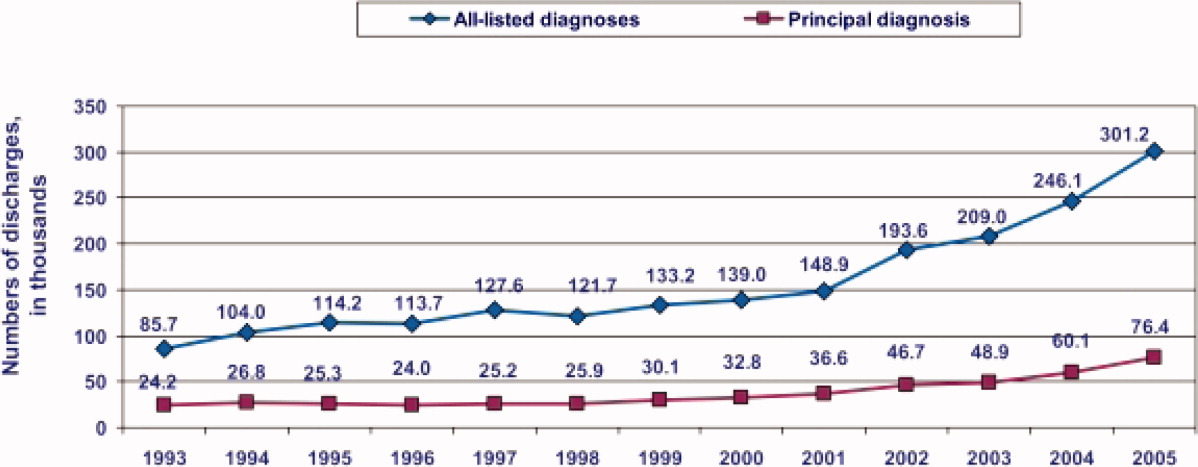
8
This increased incidence of CDI contrasts with several other healthcare‐associated infections, which have declined in incidence over the last decade. 1113 C. difficile is the most common causative agent of healthcare‐associated infections in some areas. A cohort study of common infections among inpatients at 30 community hospitals in the Duke Infection Control Outreach Network conducted between January 1, 2008 and June 30, 2009 found the incidence of CDI cases was 0.26 cases per 1000 patient‐days, which was higher than the incidence of methicillin‐resistant Staphylococcus aureus (MRSA) at 0.22 cases per 1000 patient‐days. 14 Another study utilizing the NIS data found that, while vancomycin‐resistant enterococcus and pseudomonas infections remained stable, CDI increased in many areas of the country and was more common than MRSA in some areas. 15
HYPERVIRULENT STRAIN OF C. DIFFICILE
In the early 2000s, an epidemic and hypervirulent strain of C. difficile emerged in North America and Europe that altered the epidemiology of CDI. 16 Due to multiple different methods for molecular typing of C. difficile, this strain has several names depending on the method of typing performed. The most common names for this strain are BI (REA typing), NAP1 (pulsed field gel electrophoresis), and 027 (PCR‐ribotyping). This strain has become the predominant strain of C. difficile in some areas, accounting for more than 80% of CDI cases in some areas. 3
The characteristics of this particular strain epidemic in North America typically include:
-
A deletion in the tcd gene that downregulates toxin production, which renders the gene nonfunctional in the epidemic strain. Some in vitro data have demonstrated that this epidemic strain produces 16‐fold higher concentrations of toxin A and 23‐fold higher concentrations of toxin B than nonepidemic strains of C. difficile. 17
-
Production of a third toxin, called binary toxin CDT. The role of this toxin in the pathogenesis of CDI is not clear, but the presence of this toxin has been associated with more severe CDI‐related diarrhea. 2, 16
-
High‐level resistance to fluoroquinolones, including moxifloxacin and gatifloxacin. 5, 16 It has been theorized that increasing use of fluoroquinolones during the past decade may have provided a selective advantage for the BI/NAP1/027 strain to predominate. 2
-
Production of more spores than other strains of C. difficile. 17, 18 This may increase its ability to contaminate the environment and be transmitted in a healthcare facility.
CDI SEVERITY IS INCREASING
Paralleling the increased prevalence of CDI, C. difficile infections are generally becoming more severe. In Sherbrooke, Quebec, Canada, which experienced a dramatic outbreak of CDI associated with increased CDI severity, the cumulative 1‐year attributable mortality was nearly 37% (60 of 161 CDI cases) in a hospital case review of nonsurgical admissions between January 2003 and June 2004. 19 In St Louis, Missouri in 2003, a 5.7% 180‐day mortality rate was reported in an endemic setting. 20 Among the 24% of patients readmitted within 180 days of discharge (4207 of 17,492) in this retrospective case review, patients with CDI were more than twice as likely as non‐CDI patients to be readmitted to the hospital (52% vs 23%, N = 4207). 20 Furthermore, patients with CDI were significantly more likely to require discharge to a long‐term care facility (32%) than non‐CDI controls (23%). 19
Based on NIS data for CDI‐related hospitalizations between 2000 and 2005, the crude, age‐adjusted case‐fatality rate rose from 1.2% in 2000 to 2.2% in 2004. 21 This increase was mirrored by a doubling of CDI cases admitted for hospitalization during the same 6‐year period. 21 According to the investigators, these findings indirectly confirm that the doubling in CDI deaths is attributable to an increase in C. difficile virulence. 21 A 6‐month prospective surveillance of CDI patient outcomes in 29 Canadian hospitals was conducted by the Canadian Nosocomial Infection Surveillance Program (CNISP) beginning in November 2004. 22 At 30 days after onset of CDI, the percentage of deaths directly or indirectly attributable to CDI was 5.7%, which represented an almost 4‐fold increase over CDI‐attributable deaths recorded in the 1997 CNISP survey. 22 Overall 30‐day mortality was retrospectively analyzed among patients with CDI in a St Louis, Missouri 1200‐bed teaching hospital intensive care unit (ICU) over a 2‐year period (20042005). 23 The 30‐day crude mortality among 278 patients admitted to the ICU with CDI was 37% (n = 102), and mortality directly attributable to CDI in these critically ill patients was 6%. 23 The number of deaths in the United States due to CDI increased sharply from 793 patients in 1999 to 6225 patients in 2006. 24 In 2006, it ranked among the top 20 causes of death for those aged 65 years and older. 24
INCREASE IN TREATMENT FAILURES
In addition to being more severe, there have been several reports of increases in CDI treatment failures and/or increases in recurrent CDI. 6 Recent studies indicate there may be more metronidazole treatment failures regardless of whether the infecting strain is the BI/NAP1/027 strain, despite a lack of laboratory evidence indicating resistance to metronidazole. 2529 Regardless of the initial therapy chosen, patients must be carefully monitored to ensure they are responding appropriately to treatment and their condition is not deteriorating. 29 Some of the original trials of CDI treatments found relapse rates as low as 5% to 15%. 30 More recent data indicate relapse occurs after 30% of initial CDI episodes, and as frequent as 65% if the patient has had multiple prior CDI episodes. 3, 6, 31
COMMUNITY‐ASSOCIATED CDI
The epidemiology of community‐associated CDI may also be changing. Virulent strains, which cause more severe disease in high‐risk patients, may also cause more frequent, severe disease in populations previously thought to be at low risk. Some studies have found an increase in community‐associated CDI in otherwise healthy individuals with little or no exposure to a healthcare facility. Although antimicrobial exposure remains the most important risk factor for community‐associated CDI, antimicrobial exposure is less common in community‐associated CDI than healthcare‐associated CDI. 3235
In a Canadian study, the rate of diagnosed community‐acquired CDI cases was stable at about 22 cases per 100,000 patient‐years per calendar year between 1998 and 2002, but rose steadily for the next 2 years to 53 cases per 100,000 patient‐years in 2004. 33 Similar results were seen in the United Kingdom, with an exponential increase from fewer than 1 case per 100,000 person‐years in 1994 to 22 cases per 100,000 person‐years in 2004. 32 There are currently no comprehensive longitudinal studies in the United States investigating the incidence of purely community‐acquired CDI where a patient had no prior hospital exposure. However, regional surveys have reported an incidence of community‐acquired CDI of 12 cases per 100,000 person‐years during 1992 to 1994, 36 7.6 cases per 100,000 person‐years in 2005, 37 and 6.9 cases per 100,000 person‐years in 2006. 34, 37
One patient population generally thought to be at low risk for CDI that may be at increased risk for severe CDI is pregnant women. In one study 419 infectious disease consultants who responded to a survey conducted by the Emerging Infections Network had seen or were aware of 55 cases of CDI in peripartum women. 38 There were 21 cases with complications, including 10 relapses and 5 cases of toxic megacolon. 38 In a prior report of severe CDI among 10 peripartum women, 3 women died and 3 infants (2 were twins) were stillborn. 38 This data emphasizes why clinicians must have a high index of suspicion for CDI, and should be aware of the potential for severe outcomes, even in patients traditionally considered to be at low risk. 38
ECONOMIC IMPACT OF CDI
The economic burden of CDI in the United States is staggering, with estimates ranging from $1.1 to $3.2 billion annually (Table 1). 3941 These estimates are based on the cost of caring for patients with CDI in acute care facilities and are primarily driven by increased length of stay in the hospital due to CDI. These data also predate the emergence of the BI/NAP1/027 strain. Therefore, the costs of CDI are likely higher than these estimates due to the increases in CDI severity seen since these studies were performed. It is important to note that these studies did not include patients diagnosed and treated in nursing homes or the community, nor the increase in costs due to discharge to a long‐term care facility. 39
| Study | Patient Population | Estimated Attributable Cost per Episode* | Increase in LOS, days | Estimated Annual Attributable Cost, US |
|---|---|---|---|---|
| ||||
| Kyne et al 40 | Two medical wards (n = 40) | $3669 | 3.6 | $1.1 billion |
| Dubberke et al 39 | Nonsurgical patients∥ (n = 439) | $2454$3240 | 3.0 | $897 million$1.3 billion# |
| O'Brien et al 41 | Massachusetts discharge database (n = 3692)** | Primary diagnosis: $10,212; secondary diagnosis: $13,675 | Primary diagnosis: 6.4; secondary diagnosis: 2.9 | $3.2 billion |
SUMMARY
C. difficile infections are becoming more prevalent and more severe. The issue is sufficiently serious that healthcare‐onset CDI has recently been called a major public health threat. 42 For this reason, efforts to combat virulent C. difficile should include good antimicrobial stewardship, effective infection control, and control of environmental factors that promote transmission. 35 Healthcare professionals who oversee the care of inpatients should act as catalysts for improvement by taking a leadership role in the multidisciplinary approach needed to reduce the morbidity, mortality, and cost burden for patients and the healthcare system.
- . Narrative review: the new epidemic of Clostridium difficile‐associated enteric disease. Ann Intern Med. 2006;145(10): 758–764.
- Association for Professionals in Infection Control and Epidemiology, Inc (APIC). Guide to the elimination of Clostridium difficile in healthcare settings. Available at: http://www.apic.org/Content/NavigationMenu/PracticeGuidance/APICEliminationGuides/C.diff_Elimination_guide_logo.pdf. 2008. Accessed October 8, 2011.
- . Clostridium difficile and the disease it causes. Methods Mol Biol. 2010;646:9–35.
- , , . Vegetative Clostridium difficile survives in room air on moist surfaces and in gastric contents with reduced acidity: a potential mechanism to explain the association between proton pump inhibitors and C. difficile‐associated diarrhea?Antimicrob Agents Chemother. 2007;51(8): 2883–2887.
- , . Clostridium difficile‐associated disease: new challenges from an established pathogen. Cleve Clin J Med. 2006;73(2): 187–197.
- . A 76‐year‐old man with recurrent Clostridium difficile‐associated diarrhea: review of C difficile infection. JAMA. 2009;301(9): 954–962.
- , , , . Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet. 2001;357(9251): 189–193.
- , .Clostridium difficile‐associated disease in US hospitals, 1993–2005. Healthcare Cost and Utilization Project. Statistical Brief #50. April 2008. Available at: http://www.ncbi.nlm.nih.gov/books/NBK56038/pdf/sb50.pdf. Accessed December 12, 2011.
- Agency of Healthcare Research and Quality. Healthcare Cost and Utilization Project Database. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed April 2011.
- , , , et al. Clostridium difficile infection in Ohio hospitals and nursing homes during 2006. Infect Control Hosp Epidemiol. 2009;30(6): 526–533.
- , , , et al. Trends in catheter‐associated urinary tract infections in adult intensive care units—United States, 1990–2007. Infect Control Hosp Epidemiol. 2011;32(8): 748–756.
- , , , et al. Methicillin‐resistant Staphylococcus aureus central line‐associated bloodstream infections in US intensive care units, 1997–2007. JAMA. 2009;301(7): 727–736.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26): 2725–2732.
- , , , .The impact of hospital‐onset healthcare facility associated (HO‐HCFA) Clostridium difficile infection (CDI) in community hospitals: surpassing methicillin‐resistant Staphylococcus aureus (MRSA) as the new superbug [abstract 386]. Presented at: The Fifth Decennial International Conference on Healthcare‐Associated Infections (ICHAI). March 20, 2010; Atlanta, GA.
- , , . Growth and geographic variation in hospitalizations with resistant infections, United States, 2000–2005. Emerg Infect Dis. 2008;14(11): 1756–1758.
- , , . An epidemic, toxin gene‐variant strain of Clostridium difficile. New Engl J Med. 2005;353(23): 2433–2441.
- , , , et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366(9491): 1079–1084.
- , , , et al. Increased sporulation rate of epidemic Clostridium difficile type 027/NAP1. J Clin Microbiol. 2008;46(4): 1530–1533.
- , , . Mortality attributable to nosocomial Clostridium difficile‐ associated disease during an epidemic caused by a hypervirulent strain in Quebec. Can Med Assoc J. 2005;173(9): 1037–1042.
- , , , et al. Attributable outcomes of endemic Clostridium difficile‐associated disease in nonsurgical patients. Emerg Infect Dis. 2008;14:1031–1038.
- , , . Increase in adult Clostridium difficile‐related hospitalizations and case‐fatality rate, United States, 2000–2005. Emerg Infect Dis. 2008;14(6): 929–931.
- , , , et al. Health care‐associated Clostridium difficile infection in adults admitted to acute care hospitals in Canada: a Canadian Nosocomial Infection Surveillance Program study. Clin Infect Dis. 2009:48(5);568–576.
- , , , et al. Analysis of 30‐day mortality for Clostridium difficile‐associated disease in the ICU setting. Cheat. 2007;132(2): 418–424.
- , , , et al. Deaths: final data for 2006. Natl Vital Stat Rep. 2009;57(14): 1–134.
- , , . Factors associated with failure of metronidazole in Clostridium difficile‐associated disease. J Clin Gastroenterol. 2004;38(5): 414–418.
- , , , et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11): 1586–1590.
- , , , et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40(11): 1591–1597.
- , , , et al. Outcome of metronidazole therapy for Clostridium difficile disease and correlation with a scoring system. J Infect. 2007;55(6): 495–501.
- Centers for Disease Control and Prevention. Information about the current strain of Clostridium difficile. Available at: http://www.cdc.gov/HAI/organisms/cdiff/Cdiff‐current‐strain.html. Last updated: January 25, 2011. Accessed October 9, 2011.
- , , , et al. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile‐associated diarrhea. Clin Infect Dis. 1996;22(5): 813–818.
- , , . Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7): 1769–1775.
- , , , . Use of gastric acid‐suppressive agents and the risk of community‐acquired Clostridium difficile‐associated disease. JAMA. 2005;294(23): 2989–2995.
- , , , , . Patterns of antibiotic use and risk of hospital admission because of Clostridium difficile infection. Can Med Assoc J. 2008;179(8): 767–772.
- Centers for Disease Control and Prevention. Severe Clostridium difficile‐associated disease in populations previously at low risk—four states, 2005. MMWR. 2005;54(47): 1201–1205.
- . Clostridium difficile‐associated disease: an emerging threat to patient safety: insights from the Society of Infectious Diseases Pharmacists. Pharmacotherapy. 2006;26(3): 299–311.
- , , , et al. Antibiotics and Clostridium difficile in the ambulatory care setting. Clin Ther. 2000;22(1): 91–102.
- Centers for Disease Control and Prevention. Surveillance for community‐associated C. difficile—Connecticut, 2006. MMWR. 2008;57:340–343.
- , O', , et al. Clostridium difficile‐associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198(6):635.e1–635.e6.
- , , , et al. Short‐ and long‐term attributable costs of Clostridium difficile‐associated disease in nonsurgical inpatients. Clin Infect Dis. 2008;46(4): 497–504.
- , , , . Health care costs and mortality associated with nosocomial diarrhea due to Clostridium difficile. Clin Infect Dis. 2002;34(3): 346–353.
- , , , . The emerging infectious challenge of Clostridium difficile‐associated disease in Massachusetts hospitals: clinical and economic consequences. Infect Control Hosp Epidemiol. 2007;28(11): 1219–1227.
- , , , , . National Clostridium difficile infection (CDI) related hospitalizations approaches MRSA related hospitalizations. The need for antibiotic stewardship program [poster 94]. Presented at: The SHEA 2011 Annual Scientific Meeting. April 2, 2011; Dallas, TX.
Clostridium difficile is a gram‐positive, spore‐forming, toxin‐producing, anaerobic bacillus that was established as the causative pathogen of most cases of antibiotic‐associated colitis in 1978. 1, 2 The spectrum of possible clinical presentations of C. difficile range from asymptomatic colonization, uncomplicated diarrhea, severe pseudomembranous colitis, paralytic ileus, to sepsis and death, with a mortality rate upwards of 80% in fulminant cases requiring colectomy. 3
Vegetative C. difficile cells die rapidly on dry surfaces, but they have been found to remain viable for up to 6 hours on moist surfaces in room air. 4 Spores shed from the gastrointestinal (GI) tract, however, are highly resistant to common hospital disinfectants, and can survive in the environment for many months. 2 C. difficile spores are primarily transmitted from patient to patient on the hands or equipment of healthcare workers. 2 Once spores are ingested and reach the GI tract, they germinate in the vegetative form. 2, 5 In the GI tract, C. difficile causes disease by the production of toxins, primarily toxins A and B, both of which cause severe inflammation. 5 Toxin A attracts neutrophils and monocytes, and toxin B breaks down colonic epithelial cells. 5 Both of these mechanisms lead to colitis, formation of pseudomembranes, and watery diarrhea. 5
After alteration of the healthy colonic bacterial flora, the immune response to C. difficile toxins appears to play a major role in determining host susceptibility to C. difficile infection (CDI). 5, 6 Those with antitoxin immunity are more likely to become symptomless carriers than patients without preexisting immunity. 3 More than 60% of healthy adults have protective immunity against a primary CDI, demonstrated by detectable serum IgG and IgA to both toxins A and B, as a consequence of childhood immunity or frequent exposure to C. difficile in the environment. 3 After a primary episode of CDI, many patients acquire protective immunity against C. difficile toxins, seen as significantly higher serum concentrations of IgM against C. difficile toxin by the third day from onset of diarrhea, and significantly higher serum concentrations of IgG against toxin A by the 12th day. 7 Patients who experience recurrent CDI lack development of this protective immunity to C. difficile. 6, 7
CDI INCIDENCE IS ON THE RISE
During the past decade, rates of CDI have increased steadily to levels not previously seen. A report published by the Agency of Healthcare Research and Quality demonstrated that the number of CDI diagnoses on hospital discharge more than doubled in the United States from 139,000 to 301,200 between 2000 and 2005 (Figure 1). 8 Examination of a more recent Nationwide Inpatient Sample (NIS) indicates continuation of this trend, with nearly 350,000 CDI diagnoses recorded upon discharge from acute care hospitals in 2008. 9 Of note, in 2006 the state of Ohio mandated CDI reporting from both hospitals and nursing homes. It was estimated there were more than 18,000 cases of CDI during this year, of which more than 60% were diagnosed in nursing homes. 10 Based on the 2008 NIS data and the data from Ohio, it is conceivable there were as many as 1 million cases of CDI in the US in 2008.

8
This increased incidence of CDI contrasts with several other healthcare‐associated infections, which have declined in incidence over the last decade. 1113 C. difficile is the most common causative agent of healthcare‐associated infections in some areas. A cohort study of common infections among inpatients at 30 community hospitals in the Duke Infection Control Outreach Network conducted between January 1, 2008 and June 30, 2009 found the incidence of CDI cases was 0.26 cases per 1000 patient‐days, which was higher than the incidence of methicillin‐resistant Staphylococcus aureus (MRSA) at 0.22 cases per 1000 patient‐days. 14 Another study utilizing the NIS data found that, while vancomycin‐resistant enterococcus and pseudomonas infections remained stable, CDI increased in many areas of the country and was more common than MRSA in some areas. 15
HYPERVIRULENT STRAIN OF C. DIFFICILE
In the early 2000s, an epidemic and hypervirulent strain of C. difficile emerged in North America and Europe that altered the epidemiology of CDI. 16 Due to multiple different methods for molecular typing of C. difficile, this strain has several names depending on the method of typing performed. The most common names for this strain are BI (REA typing), NAP1 (pulsed field gel electrophoresis), and 027 (PCR‐ribotyping). This strain has become the predominant strain of C. difficile in some areas, accounting for more than 80% of CDI cases in some areas. 3
The characteristics of this particular strain epidemic in North America typically include:
-
A deletion in the tcd gene that downregulates toxin production, which renders the gene nonfunctional in the epidemic strain. Some in vitro data have demonstrated that this epidemic strain produces 16‐fold higher concentrations of toxin A and 23‐fold higher concentrations of toxin B than nonepidemic strains of C. difficile. 17
-
Production of a third toxin, called binary toxin CDT. The role of this toxin in the pathogenesis of CDI is not clear, but the presence of this toxin has been associated with more severe CDI‐related diarrhea. 2, 16
-
High‐level resistance to fluoroquinolones, including moxifloxacin and gatifloxacin. 5, 16 It has been theorized that increasing use of fluoroquinolones during the past decade may have provided a selective advantage for the BI/NAP1/027 strain to predominate. 2
-
Production of more spores than other strains of C. difficile. 17, 18 This may increase its ability to contaminate the environment and be transmitted in a healthcare facility.
CDI SEVERITY IS INCREASING
Paralleling the increased prevalence of CDI, C. difficile infections are generally becoming more severe. In Sherbrooke, Quebec, Canada, which experienced a dramatic outbreak of CDI associated with increased CDI severity, the cumulative 1‐year attributable mortality was nearly 37% (60 of 161 CDI cases) in a hospital case review of nonsurgical admissions between January 2003 and June 2004. 19 In St Louis, Missouri in 2003, a 5.7% 180‐day mortality rate was reported in an endemic setting. 20 Among the 24% of patients readmitted within 180 days of discharge (4207 of 17,492) in this retrospective case review, patients with CDI were more than twice as likely as non‐CDI patients to be readmitted to the hospital (52% vs 23%, N = 4207). 20 Furthermore, patients with CDI were significantly more likely to require discharge to a long‐term care facility (32%) than non‐CDI controls (23%). 19
Based on NIS data for CDI‐related hospitalizations between 2000 and 2005, the crude, age‐adjusted case‐fatality rate rose from 1.2% in 2000 to 2.2% in 2004. 21 This increase was mirrored by a doubling of CDI cases admitted for hospitalization during the same 6‐year period. 21 According to the investigators, these findings indirectly confirm that the doubling in CDI deaths is attributable to an increase in C. difficile virulence. 21 A 6‐month prospective surveillance of CDI patient outcomes in 29 Canadian hospitals was conducted by the Canadian Nosocomial Infection Surveillance Program (CNISP) beginning in November 2004. 22 At 30 days after onset of CDI, the percentage of deaths directly or indirectly attributable to CDI was 5.7%, which represented an almost 4‐fold increase over CDI‐attributable deaths recorded in the 1997 CNISP survey. 22 Overall 30‐day mortality was retrospectively analyzed among patients with CDI in a St Louis, Missouri 1200‐bed teaching hospital intensive care unit (ICU) over a 2‐year period (20042005). 23 The 30‐day crude mortality among 278 patients admitted to the ICU with CDI was 37% (n = 102), and mortality directly attributable to CDI in these critically ill patients was 6%. 23 The number of deaths in the United States due to CDI increased sharply from 793 patients in 1999 to 6225 patients in 2006. 24 In 2006, it ranked among the top 20 causes of death for those aged 65 years and older. 24
INCREASE IN TREATMENT FAILURES
In addition to being more severe, there have been several reports of increases in CDI treatment failures and/or increases in recurrent CDI. 6 Recent studies indicate there may be more metronidazole treatment failures regardless of whether the infecting strain is the BI/NAP1/027 strain, despite a lack of laboratory evidence indicating resistance to metronidazole. 2529 Regardless of the initial therapy chosen, patients must be carefully monitored to ensure they are responding appropriately to treatment and their condition is not deteriorating. 29 Some of the original trials of CDI treatments found relapse rates as low as 5% to 15%. 30 More recent data indicate relapse occurs after 30% of initial CDI episodes, and as frequent as 65% if the patient has had multiple prior CDI episodes. 3, 6, 31
COMMUNITY‐ASSOCIATED CDI
The epidemiology of community‐associated CDI may also be changing. Virulent strains, which cause more severe disease in high‐risk patients, may also cause more frequent, severe disease in populations previously thought to be at low risk. Some studies have found an increase in community‐associated CDI in otherwise healthy individuals with little or no exposure to a healthcare facility. Although antimicrobial exposure remains the most important risk factor for community‐associated CDI, antimicrobial exposure is less common in community‐associated CDI than healthcare‐associated CDI. 3235
In a Canadian study, the rate of diagnosed community‐acquired CDI cases was stable at about 22 cases per 100,000 patient‐years per calendar year between 1998 and 2002, but rose steadily for the next 2 years to 53 cases per 100,000 patient‐years in 2004. 33 Similar results were seen in the United Kingdom, with an exponential increase from fewer than 1 case per 100,000 person‐years in 1994 to 22 cases per 100,000 person‐years in 2004. 32 There are currently no comprehensive longitudinal studies in the United States investigating the incidence of purely community‐acquired CDI where a patient had no prior hospital exposure. However, regional surveys have reported an incidence of community‐acquired CDI of 12 cases per 100,000 person‐years during 1992 to 1994, 36 7.6 cases per 100,000 person‐years in 2005, 37 and 6.9 cases per 100,000 person‐years in 2006. 34, 37
One patient population generally thought to be at low risk for CDI that may be at increased risk for severe CDI is pregnant women. In one study 419 infectious disease consultants who responded to a survey conducted by the Emerging Infections Network had seen or were aware of 55 cases of CDI in peripartum women. 38 There were 21 cases with complications, including 10 relapses and 5 cases of toxic megacolon. 38 In a prior report of severe CDI among 10 peripartum women, 3 women died and 3 infants (2 were twins) were stillborn. 38 This data emphasizes why clinicians must have a high index of suspicion for CDI, and should be aware of the potential for severe outcomes, even in patients traditionally considered to be at low risk. 38
ECONOMIC IMPACT OF CDI
The economic burden of CDI in the United States is staggering, with estimates ranging from $1.1 to $3.2 billion annually (Table 1). 3941 These estimates are based on the cost of caring for patients with CDI in acute care facilities and are primarily driven by increased length of stay in the hospital due to CDI. These data also predate the emergence of the BI/NAP1/027 strain. Therefore, the costs of CDI are likely higher than these estimates due to the increases in CDI severity seen since these studies were performed. It is important to note that these studies did not include patients diagnosed and treated in nursing homes or the community, nor the increase in costs due to discharge to a long‐term care facility. 39
| Study | Patient Population | Estimated Attributable Cost per Episode* | Increase in LOS, days | Estimated Annual Attributable Cost, US |
|---|---|---|---|---|
| ||||
| Kyne et al 40 | Two medical wards (n = 40) | $3669 | 3.6 | $1.1 billion |
| Dubberke et al 39 | Nonsurgical patients∥ (n = 439) | $2454$3240 | 3.0 | $897 million$1.3 billion# |
| O'Brien et al 41 | Massachusetts discharge database (n = 3692)** | Primary diagnosis: $10,212; secondary diagnosis: $13,675 | Primary diagnosis: 6.4; secondary diagnosis: 2.9 | $3.2 billion |
SUMMARY
C. difficile infections are becoming more prevalent and more severe. The issue is sufficiently serious that healthcare‐onset CDI has recently been called a major public health threat. 42 For this reason, efforts to combat virulent C. difficile should include good antimicrobial stewardship, effective infection control, and control of environmental factors that promote transmission. 35 Healthcare professionals who oversee the care of inpatients should act as catalysts for improvement by taking a leadership role in the multidisciplinary approach needed to reduce the morbidity, mortality, and cost burden for patients and the healthcare system.
Clostridium difficile is a gram‐positive, spore‐forming, toxin‐producing, anaerobic bacillus that was established as the causative pathogen of most cases of antibiotic‐associated colitis in 1978. 1, 2 The spectrum of possible clinical presentations of C. difficile range from asymptomatic colonization, uncomplicated diarrhea, severe pseudomembranous colitis, paralytic ileus, to sepsis and death, with a mortality rate upwards of 80% in fulminant cases requiring colectomy. 3
Vegetative C. difficile cells die rapidly on dry surfaces, but they have been found to remain viable for up to 6 hours on moist surfaces in room air. 4 Spores shed from the gastrointestinal (GI) tract, however, are highly resistant to common hospital disinfectants, and can survive in the environment for many months. 2 C. difficile spores are primarily transmitted from patient to patient on the hands or equipment of healthcare workers. 2 Once spores are ingested and reach the GI tract, they germinate in the vegetative form. 2, 5 In the GI tract, C. difficile causes disease by the production of toxins, primarily toxins A and B, both of which cause severe inflammation. 5 Toxin A attracts neutrophils and monocytes, and toxin B breaks down colonic epithelial cells. 5 Both of these mechanisms lead to colitis, formation of pseudomembranes, and watery diarrhea. 5
After alteration of the healthy colonic bacterial flora, the immune response to C. difficile toxins appears to play a major role in determining host susceptibility to C. difficile infection (CDI). 5, 6 Those with antitoxin immunity are more likely to become symptomless carriers than patients without preexisting immunity. 3 More than 60% of healthy adults have protective immunity against a primary CDI, demonstrated by detectable serum IgG and IgA to both toxins A and B, as a consequence of childhood immunity or frequent exposure to C. difficile in the environment. 3 After a primary episode of CDI, many patients acquire protective immunity against C. difficile toxins, seen as significantly higher serum concentrations of IgM against C. difficile toxin by the third day from onset of diarrhea, and significantly higher serum concentrations of IgG against toxin A by the 12th day. 7 Patients who experience recurrent CDI lack development of this protective immunity to C. difficile. 6, 7
CDI INCIDENCE IS ON THE RISE
During the past decade, rates of CDI have increased steadily to levels not previously seen. A report published by the Agency of Healthcare Research and Quality demonstrated that the number of CDI diagnoses on hospital discharge more than doubled in the United States from 139,000 to 301,200 between 2000 and 2005 (Figure 1). 8 Examination of a more recent Nationwide Inpatient Sample (NIS) indicates continuation of this trend, with nearly 350,000 CDI diagnoses recorded upon discharge from acute care hospitals in 2008. 9 Of note, in 2006 the state of Ohio mandated CDI reporting from both hospitals and nursing homes. It was estimated there were more than 18,000 cases of CDI during this year, of which more than 60% were diagnosed in nursing homes. 10 Based on the 2008 NIS data and the data from Ohio, it is conceivable there were as many as 1 million cases of CDI in the US in 2008.

8
This increased incidence of CDI contrasts with several other healthcare‐associated infections, which have declined in incidence over the last decade. 1113 C. difficile is the most common causative agent of healthcare‐associated infections in some areas. A cohort study of common infections among inpatients at 30 community hospitals in the Duke Infection Control Outreach Network conducted between January 1, 2008 and June 30, 2009 found the incidence of CDI cases was 0.26 cases per 1000 patient‐days, which was higher than the incidence of methicillin‐resistant Staphylococcus aureus (MRSA) at 0.22 cases per 1000 patient‐days. 14 Another study utilizing the NIS data found that, while vancomycin‐resistant enterococcus and pseudomonas infections remained stable, CDI increased in many areas of the country and was more common than MRSA in some areas. 15
HYPERVIRULENT STRAIN OF C. DIFFICILE
In the early 2000s, an epidemic and hypervirulent strain of C. difficile emerged in North America and Europe that altered the epidemiology of CDI. 16 Due to multiple different methods for molecular typing of C. difficile, this strain has several names depending on the method of typing performed. The most common names for this strain are BI (REA typing), NAP1 (pulsed field gel electrophoresis), and 027 (PCR‐ribotyping). This strain has become the predominant strain of C. difficile in some areas, accounting for more than 80% of CDI cases in some areas. 3
The characteristics of this particular strain epidemic in North America typically include:
-
A deletion in the tcd gene that downregulates toxin production, which renders the gene nonfunctional in the epidemic strain. Some in vitro data have demonstrated that this epidemic strain produces 16‐fold higher concentrations of toxin A and 23‐fold higher concentrations of toxin B than nonepidemic strains of C. difficile. 17
-
Production of a third toxin, called binary toxin CDT. The role of this toxin in the pathogenesis of CDI is not clear, but the presence of this toxin has been associated with more severe CDI‐related diarrhea. 2, 16
-
High‐level resistance to fluoroquinolones, including moxifloxacin and gatifloxacin. 5, 16 It has been theorized that increasing use of fluoroquinolones during the past decade may have provided a selective advantage for the BI/NAP1/027 strain to predominate. 2
-
Production of more spores than other strains of C. difficile. 17, 18 This may increase its ability to contaminate the environment and be transmitted in a healthcare facility.
CDI SEVERITY IS INCREASING
Paralleling the increased prevalence of CDI, C. difficile infections are generally becoming more severe. In Sherbrooke, Quebec, Canada, which experienced a dramatic outbreak of CDI associated with increased CDI severity, the cumulative 1‐year attributable mortality was nearly 37% (60 of 161 CDI cases) in a hospital case review of nonsurgical admissions between January 2003 and June 2004. 19 In St Louis, Missouri in 2003, a 5.7% 180‐day mortality rate was reported in an endemic setting. 20 Among the 24% of patients readmitted within 180 days of discharge (4207 of 17,492) in this retrospective case review, patients with CDI were more than twice as likely as non‐CDI patients to be readmitted to the hospital (52% vs 23%, N = 4207). 20 Furthermore, patients with CDI were significantly more likely to require discharge to a long‐term care facility (32%) than non‐CDI controls (23%). 19
Based on NIS data for CDI‐related hospitalizations between 2000 and 2005, the crude, age‐adjusted case‐fatality rate rose from 1.2% in 2000 to 2.2% in 2004. 21 This increase was mirrored by a doubling of CDI cases admitted for hospitalization during the same 6‐year period. 21 According to the investigators, these findings indirectly confirm that the doubling in CDI deaths is attributable to an increase in C. difficile virulence. 21 A 6‐month prospective surveillance of CDI patient outcomes in 29 Canadian hospitals was conducted by the Canadian Nosocomial Infection Surveillance Program (CNISP) beginning in November 2004. 22 At 30 days after onset of CDI, the percentage of deaths directly or indirectly attributable to CDI was 5.7%, which represented an almost 4‐fold increase over CDI‐attributable deaths recorded in the 1997 CNISP survey. 22 Overall 30‐day mortality was retrospectively analyzed among patients with CDI in a St Louis, Missouri 1200‐bed teaching hospital intensive care unit (ICU) over a 2‐year period (20042005). 23 The 30‐day crude mortality among 278 patients admitted to the ICU with CDI was 37% (n = 102), and mortality directly attributable to CDI in these critically ill patients was 6%. 23 The number of deaths in the United States due to CDI increased sharply from 793 patients in 1999 to 6225 patients in 2006. 24 In 2006, it ranked among the top 20 causes of death for those aged 65 years and older. 24
INCREASE IN TREATMENT FAILURES
In addition to being more severe, there have been several reports of increases in CDI treatment failures and/or increases in recurrent CDI. 6 Recent studies indicate there may be more metronidazole treatment failures regardless of whether the infecting strain is the BI/NAP1/027 strain, despite a lack of laboratory evidence indicating resistance to metronidazole. 2529 Regardless of the initial therapy chosen, patients must be carefully monitored to ensure they are responding appropriately to treatment and their condition is not deteriorating. 29 Some of the original trials of CDI treatments found relapse rates as low as 5% to 15%. 30 More recent data indicate relapse occurs after 30% of initial CDI episodes, and as frequent as 65% if the patient has had multiple prior CDI episodes. 3, 6, 31
COMMUNITY‐ASSOCIATED CDI
The epidemiology of community‐associated CDI may also be changing. Virulent strains, which cause more severe disease in high‐risk patients, may also cause more frequent, severe disease in populations previously thought to be at low risk. Some studies have found an increase in community‐associated CDI in otherwise healthy individuals with little or no exposure to a healthcare facility. Although antimicrobial exposure remains the most important risk factor for community‐associated CDI, antimicrobial exposure is less common in community‐associated CDI than healthcare‐associated CDI. 3235
In a Canadian study, the rate of diagnosed community‐acquired CDI cases was stable at about 22 cases per 100,000 patient‐years per calendar year between 1998 and 2002, but rose steadily for the next 2 years to 53 cases per 100,000 patient‐years in 2004. 33 Similar results were seen in the United Kingdom, with an exponential increase from fewer than 1 case per 100,000 person‐years in 1994 to 22 cases per 100,000 person‐years in 2004. 32 There are currently no comprehensive longitudinal studies in the United States investigating the incidence of purely community‐acquired CDI where a patient had no prior hospital exposure. However, regional surveys have reported an incidence of community‐acquired CDI of 12 cases per 100,000 person‐years during 1992 to 1994, 36 7.6 cases per 100,000 person‐years in 2005, 37 and 6.9 cases per 100,000 person‐years in 2006. 34, 37
One patient population generally thought to be at low risk for CDI that may be at increased risk for severe CDI is pregnant women. In one study 419 infectious disease consultants who responded to a survey conducted by the Emerging Infections Network had seen or were aware of 55 cases of CDI in peripartum women. 38 There were 21 cases with complications, including 10 relapses and 5 cases of toxic megacolon. 38 In a prior report of severe CDI among 10 peripartum women, 3 women died and 3 infants (2 were twins) were stillborn. 38 This data emphasizes why clinicians must have a high index of suspicion for CDI, and should be aware of the potential for severe outcomes, even in patients traditionally considered to be at low risk. 38
ECONOMIC IMPACT OF CDI
The economic burden of CDI in the United States is staggering, with estimates ranging from $1.1 to $3.2 billion annually (Table 1). 3941 These estimates are based on the cost of caring for patients with CDI in acute care facilities and are primarily driven by increased length of stay in the hospital due to CDI. These data also predate the emergence of the BI/NAP1/027 strain. Therefore, the costs of CDI are likely higher than these estimates due to the increases in CDI severity seen since these studies were performed. It is important to note that these studies did not include patients diagnosed and treated in nursing homes or the community, nor the increase in costs due to discharge to a long‐term care facility. 39
| Study | Patient Population | Estimated Attributable Cost per Episode* | Increase in LOS, days | Estimated Annual Attributable Cost, US |
|---|---|---|---|---|
| ||||
| Kyne et al 40 | Two medical wards (n = 40) | $3669 | 3.6 | $1.1 billion |
| Dubberke et al 39 | Nonsurgical patients∥ (n = 439) | $2454$3240 | 3.0 | $897 million$1.3 billion# |
| O'Brien et al 41 | Massachusetts discharge database (n = 3692)** | Primary diagnosis: $10,212; secondary diagnosis: $13,675 | Primary diagnosis: 6.4; secondary diagnosis: 2.9 | $3.2 billion |
SUMMARY
C. difficile infections are becoming more prevalent and more severe. The issue is sufficiently serious that healthcare‐onset CDI has recently been called a major public health threat. 42 For this reason, efforts to combat virulent C. difficile should include good antimicrobial stewardship, effective infection control, and control of environmental factors that promote transmission. 35 Healthcare professionals who oversee the care of inpatients should act as catalysts for improvement by taking a leadership role in the multidisciplinary approach needed to reduce the morbidity, mortality, and cost burden for patients and the healthcare system.
- . Narrative review: the new epidemic of Clostridium difficile‐associated enteric disease. Ann Intern Med. 2006;145(10): 758–764.
- Association for Professionals in Infection Control and Epidemiology, Inc (APIC). Guide to the elimination of Clostridium difficile in healthcare settings. Available at: http://www.apic.org/Content/NavigationMenu/PracticeGuidance/APICEliminationGuides/C.diff_Elimination_guide_logo.pdf. 2008. Accessed October 8, 2011.
- . Clostridium difficile and the disease it causes. Methods Mol Biol. 2010;646:9–35.
- , , . Vegetative Clostridium difficile survives in room air on moist surfaces and in gastric contents with reduced acidity: a potential mechanism to explain the association between proton pump inhibitors and C. difficile‐associated diarrhea?Antimicrob Agents Chemother. 2007;51(8): 2883–2887.
- , . Clostridium difficile‐associated disease: new challenges from an established pathogen. Cleve Clin J Med. 2006;73(2): 187–197.
- . A 76‐year‐old man with recurrent Clostridium difficile‐associated diarrhea: review of C difficile infection. JAMA. 2009;301(9): 954–962.
- , , , . Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet. 2001;357(9251): 189–193.
- , .Clostridium difficile‐associated disease in US hospitals, 1993–2005. Healthcare Cost and Utilization Project. Statistical Brief #50. April 2008. Available at: http://www.ncbi.nlm.nih.gov/books/NBK56038/pdf/sb50.pdf. Accessed December 12, 2011.
- Agency of Healthcare Research and Quality. Healthcare Cost and Utilization Project Database. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed April 2011.
- , , , et al. Clostridium difficile infection in Ohio hospitals and nursing homes during 2006. Infect Control Hosp Epidemiol. 2009;30(6): 526–533.
- , , , et al. Trends in catheter‐associated urinary tract infections in adult intensive care units—United States, 1990–2007. Infect Control Hosp Epidemiol. 2011;32(8): 748–756.
- , , , et al. Methicillin‐resistant Staphylococcus aureus central line‐associated bloodstream infections in US intensive care units, 1997–2007. JAMA. 2009;301(7): 727–736.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26): 2725–2732.
- , , , .The impact of hospital‐onset healthcare facility associated (HO‐HCFA) Clostridium difficile infection (CDI) in community hospitals: surpassing methicillin‐resistant Staphylococcus aureus (MRSA) as the new superbug [abstract 386]. Presented at: The Fifth Decennial International Conference on Healthcare‐Associated Infections (ICHAI). March 20, 2010; Atlanta, GA.
- , , . Growth and geographic variation in hospitalizations with resistant infections, United States, 2000–2005. Emerg Infect Dis. 2008;14(11): 1756–1758.
- , , . An epidemic, toxin gene‐variant strain of Clostridium difficile. New Engl J Med. 2005;353(23): 2433–2441.
- , , , et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366(9491): 1079–1084.
- , , , et al. Increased sporulation rate of epidemic Clostridium difficile type 027/NAP1. J Clin Microbiol. 2008;46(4): 1530–1533.
- , , . Mortality attributable to nosocomial Clostridium difficile‐ associated disease during an epidemic caused by a hypervirulent strain in Quebec. Can Med Assoc J. 2005;173(9): 1037–1042.
- , , , et al. Attributable outcomes of endemic Clostridium difficile‐associated disease in nonsurgical patients. Emerg Infect Dis. 2008;14:1031–1038.
- , , . Increase in adult Clostridium difficile‐related hospitalizations and case‐fatality rate, United States, 2000–2005. Emerg Infect Dis. 2008;14(6): 929–931.
- , , , et al. Health care‐associated Clostridium difficile infection in adults admitted to acute care hospitals in Canada: a Canadian Nosocomial Infection Surveillance Program study. Clin Infect Dis. 2009:48(5);568–576.
- , , , et al. Analysis of 30‐day mortality for Clostridium difficile‐associated disease in the ICU setting. Cheat. 2007;132(2): 418–424.
- , , , et al. Deaths: final data for 2006. Natl Vital Stat Rep. 2009;57(14): 1–134.
- , , . Factors associated with failure of metronidazole in Clostridium difficile‐associated disease. J Clin Gastroenterol. 2004;38(5): 414–418.
- , , , et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11): 1586–1590.
- , , , et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40(11): 1591–1597.
- , , , et al. Outcome of metronidazole therapy for Clostridium difficile disease and correlation with a scoring system. J Infect. 2007;55(6): 495–501.
- Centers for Disease Control and Prevention. Information about the current strain of Clostridium difficile. Available at: http://www.cdc.gov/HAI/organisms/cdiff/Cdiff‐current‐strain.html. Last updated: January 25, 2011. Accessed October 9, 2011.
- , , , et al. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile‐associated diarrhea. Clin Infect Dis. 1996;22(5): 813–818.
- , , . Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7): 1769–1775.
- , , , . Use of gastric acid‐suppressive agents and the risk of community‐acquired Clostridium difficile‐associated disease. JAMA. 2005;294(23): 2989–2995.
- , , , , . Patterns of antibiotic use and risk of hospital admission because of Clostridium difficile infection. Can Med Assoc J. 2008;179(8): 767–772.
- Centers for Disease Control and Prevention. Severe Clostridium difficile‐associated disease in populations previously at low risk—four states, 2005. MMWR. 2005;54(47): 1201–1205.
- . Clostridium difficile‐associated disease: an emerging threat to patient safety: insights from the Society of Infectious Diseases Pharmacists. Pharmacotherapy. 2006;26(3): 299–311.
- , , , et al. Antibiotics and Clostridium difficile in the ambulatory care setting. Clin Ther. 2000;22(1): 91–102.
- Centers for Disease Control and Prevention. Surveillance for community‐associated C. difficile—Connecticut, 2006. MMWR. 2008;57:340–343.
- , O', , et al. Clostridium difficile‐associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198(6):635.e1–635.e6.
- , , , et al. Short‐ and long‐term attributable costs of Clostridium difficile‐associated disease in nonsurgical inpatients. Clin Infect Dis. 2008;46(4): 497–504.
- , , , . Health care costs and mortality associated with nosocomial diarrhea due to Clostridium difficile. Clin Infect Dis. 2002;34(3): 346–353.
- , , , . The emerging infectious challenge of Clostridium difficile‐associated disease in Massachusetts hospitals: clinical and economic consequences. Infect Control Hosp Epidemiol. 2007;28(11): 1219–1227.
- , , , , . National Clostridium difficile infection (CDI) related hospitalizations approaches MRSA related hospitalizations. The need for antibiotic stewardship program [poster 94]. Presented at: The SHEA 2011 Annual Scientific Meeting. April 2, 2011; Dallas, TX.
- . Narrative review: the new epidemic of Clostridium difficile‐associated enteric disease. Ann Intern Med. 2006;145(10): 758–764.
- Association for Professionals in Infection Control and Epidemiology, Inc (APIC). Guide to the elimination of Clostridium difficile in healthcare settings. Available at: http://www.apic.org/Content/NavigationMenu/PracticeGuidance/APICEliminationGuides/C.diff_Elimination_guide_logo.pdf. 2008. Accessed October 8, 2011.
- . Clostridium difficile and the disease it causes. Methods Mol Biol. 2010;646:9–35.
- , , . Vegetative Clostridium difficile survives in room air on moist surfaces and in gastric contents with reduced acidity: a potential mechanism to explain the association between proton pump inhibitors and C. difficile‐associated diarrhea?Antimicrob Agents Chemother. 2007;51(8): 2883–2887.
- , . Clostridium difficile‐associated disease: new challenges from an established pathogen. Cleve Clin J Med. 2006;73(2): 187–197.
- . A 76‐year‐old man with recurrent Clostridium difficile‐associated diarrhea: review of C difficile infection. JAMA. 2009;301(9): 954–962.
- , , , . Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet. 2001;357(9251): 189–193.
- , .Clostridium difficile‐associated disease in US hospitals, 1993–2005. Healthcare Cost and Utilization Project. Statistical Brief #50. April 2008. Available at: http://www.ncbi.nlm.nih.gov/books/NBK56038/pdf/sb50.pdf. Accessed December 12, 2011.
- Agency of Healthcare Research and Quality. Healthcare Cost and Utilization Project Database. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed April 2011.
- , , , et al. Clostridium difficile infection in Ohio hospitals and nursing homes during 2006. Infect Control Hosp Epidemiol. 2009;30(6): 526–533.
- , , , et al. Trends in catheter‐associated urinary tract infections in adult intensive care units—United States, 1990–2007. Infect Control Hosp Epidemiol. 2011;32(8): 748–756.
- , , , et al. Methicillin‐resistant Staphylococcus aureus central line‐associated bloodstream infections in US intensive care units, 1997–2007. JAMA. 2009;301(7): 727–736.
- , , , et al. An intervention to decrease catheter‐related bloodstream infections in the ICU. N Engl J Med. 2006;355(26): 2725–2732.
- , , , .The impact of hospital‐onset healthcare facility associated (HO‐HCFA) Clostridium difficile infection (CDI) in community hospitals: surpassing methicillin‐resistant Staphylococcus aureus (MRSA) as the new superbug [abstract 386]. Presented at: The Fifth Decennial International Conference on Healthcare‐Associated Infections (ICHAI). March 20, 2010; Atlanta, GA.
- , , . Growth and geographic variation in hospitalizations with resistant infections, United States, 2000–2005. Emerg Infect Dis. 2008;14(11): 1756–1758.
- , , . An epidemic, toxin gene‐variant strain of Clostridium difficile. New Engl J Med. 2005;353(23): 2433–2441.
- , , , et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366(9491): 1079–1084.
- , , , et al. Increased sporulation rate of epidemic Clostridium difficile type 027/NAP1. J Clin Microbiol. 2008;46(4): 1530–1533.
- , , . Mortality attributable to nosocomial Clostridium difficile‐ associated disease during an epidemic caused by a hypervirulent strain in Quebec. Can Med Assoc J. 2005;173(9): 1037–1042.
- , , , et al. Attributable outcomes of endemic Clostridium difficile‐associated disease in nonsurgical patients. Emerg Infect Dis. 2008;14:1031–1038.
- , , . Increase in adult Clostridium difficile‐related hospitalizations and case‐fatality rate, United States, 2000–2005. Emerg Infect Dis. 2008;14(6): 929–931.
- , , , et al. Health care‐associated Clostridium difficile infection in adults admitted to acute care hospitals in Canada: a Canadian Nosocomial Infection Surveillance Program study. Clin Infect Dis. 2009:48(5);568–576.
- , , , et al. Analysis of 30‐day mortality for Clostridium difficile‐associated disease in the ICU setting. Cheat. 2007;132(2): 418–424.
- , , , et al. Deaths: final data for 2006. Natl Vital Stat Rep. 2009;57(14): 1–134.
- , , . Factors associated with failure of metronidazole in Clostridium difficile‐associated disease. J Clin Gastroenterol. 2004;38(5): 414–418.
- , , , et al. Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole. Clin Infect Dis. 2005;40(11): 1586–1590.
- , , , et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40(11): 1591–1597.
- , , , et al. Outcome of metronidazole therapy for Clostridium difficile disease and correlation with a scoring system. J Infect. 2007;55(6): 495–501.
- Centers for Disease Control and Prevention. Information about the current strain of Clostridium difficile. Available at: http://www.cdc.gov/HAI/organisms/cdiff/Cdiff‐current‐strain.html. Last updated: January 25, 2011. Accessed October 9, 2011.
- , , , et al. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile‐associated diarrhea. Clin Infect Dis. 1996;22(5): 813–818.
- , , . Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97(7): 1769–1775.
- , , , . Use of gastric acid‐suppressive agents and the risk of community‐acquired Clostridium difficile‐associated disease. JAMA. 2005;294(23): 2989–2995.
- , , , , . Patterns of antibiotic use and risk of hospital admission because of Clostridium difficile infection. Can Med Assoc J. 2008;179(8): 767–772.
- Centers for Disease Control and Prevention. Severe Clostridium difficile‐associated disease in populations previously at low risk—four states, 2005. MMWR. 2005;54(47): 1201–1205.
- . Clostridium difficile‐associated disease: an emerging threat to patient safety: insights from the Society of Infectious Diseases Pharmacists. Pharmacotherapy. 2006;26(3): 299–311.
- , , , et al. Antibiotics and Clostridium difficile in the ambulatory care setting. Clin Ther. 2000;22(1): 91–102.
- Centers for Disease Control and Prevention. Surveillance for community‐associated C. difficile—Connecticut, 2006. MMWR. 2008;57:340–343.
- , O', , et al. Clostridium difficile‐associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198(6):635.e1–635.e6.
- , , , et al. Short‐ and long‐term attributable costs of Clostridium difficile‐associated disease in nonsurgical inpatients. Clin Infect Dis. 2008;46(4): 497–504.
- , , , . Health care costs and mortality associated with nosocomial diarrhea due to Clostridium difficile. Clin Infect Dis. 2002;34(3): 346–353.
- , , , . The emerging infectious challenge of Clostridium difficile‐associated disease in Massachusetts hospitals: clinical and economic consequences. Infect Control Hosp Epidemiol. 2007;28(11): 1219–1227.
- , , , , . National Clostridium difficile infection (CDI) related hospitalizations approaches MRSA related hospitalizations. The need for antibiotic stewardship program [poster 94]. Presented at: The SHEA 2011 Annual Scientific Meeting. April 2, 2011; Dallas, TX.