User login
Grand Rounds: Man, 30, With Traumatic Finger Amputations
A 30-year-old man sustained traumatic amputations of three of his left fingers while at work. A heavy object fell when a supporting chain snapped; although he moved quickly, three of his left distal fingers were caught under the object. He was flown to a hospital for definitive hand care.
During the preadmission history and physical, it was noted that the patient had mild right knee pain in addition to his finger injuries. He had experienced no head injury and no loss of consciousness or other complaints. He did not remember injuring his leg, although he said it might have been struck by the falling object; all he could remember was the injury to his fingers.
On physical exam, the only abnormality other than the man’s traumatic finger amputations was mild right knee edema and a small bruised area medially. Initially, he complained of mild pain on palpation and moderate pain with passive range of motion, but range of motion was intact. His pain was worse at the proximal, medial tibial area, and he had mild lateral mid-calf tenderness though no bruising. Distally, his right lower extremity motor and sensory function were intact, and he had no open wounds or skin breakdown. He had 2+ dorsalis pedis pulse and 1+ posterior tibial pulse. The toes were pink and warm with brisk capillary refill. All compartments were soft and compressible.
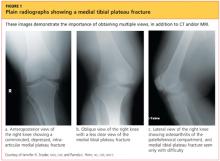
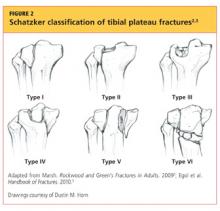
Upon review of his plain radiographs (three views of the right knee), the patient was noted to have a severely comminuted medial tibial plateau fracture that extended to the midline in the region of the tibial spine, with mild depression of the fracture fragments measuring about 6 mm (see Figures 1a, 1b, and 1c). This would translate into a Schatzker IV classification type1 fracture (see Figure 22,3).
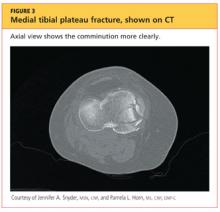
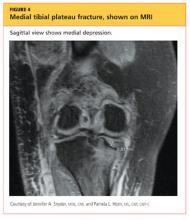
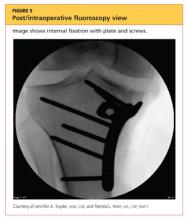
The man was admitted and underwent emergent surgery on his injured left fingers that night. Further diagnostic knee testing was performed, including CT and MRI (see Figures 3 and 4). Three days after admission, the patient underwent open reduction and internal fixation (plating) of the right medial, proximal tibia (see Figure 5). He has done very well since without issue.
DISCUSSION
Fractures of the tibial plateau occur along the articular, or joint, surface of the proximal tibia. The plateau consists of lateral and medial condylar surfaces. These concave structures function as an articulation point for the cartilaginous menisci and the femoral condyles.4 The medial plateau and condyle are stronger than those of the lateral side, and therefore are less commonly fractured. An elevated intercondylar eminence divides the lateral and medial plateaus, providing an attachment site for the cruciate ligaments.3
The Schatzker classification system1 is most commonly used to describe the types of tibial plateau fractures (as seen in Figure 22,3). Schatzker et al1 divided these injuries into six categories, according to the impact of increased energy exerted onto the bone; the rising classification numbers indicate an increase in complexity and severity and usually a worsening prognosis.
The type I fracture represents a split fracture of the lateral plateau. Typically, a fracture of this type has depression or displacement measuring less than 4 mm.
Type II tibial plateau fractures, the most common Schatzker injury, are lateral plateau fractures with depression noted at the split. Not always evident on plain radiographs, this depression can often be overlooked, and the injury mistaken for a type I fracture. The depression is measured vertically from the lower edge of the medial plateau to the lowest depression point of the lateral plateau.5
Type III fractures, the least common among the Schatzker injuries, are described as pure depression fractures of the lateral plateau. These fractures do not have an appreciable “split” along the plateau and are usually found in older patients with osteopenia.2
The Schatzker type IV injury is a medial fracture with displacement or depression to a portion of the plateau. The fracture may be split or comminuted and may originate in the intercondylar area.
Type V fractures, also known as “bicondylar fractures,” affect both the lateral and medial plateau. An inverted “Y” pattern is frequently seen, and there may be additional involvement of the intercondylar eminence. Type V fractures differ from type VI injuries in that there is no disturbance of the metaphyseal-diaphyseal connection. Thus, type VI fractures also include a transverse component that separates the condyles (metaphysis) of the bone from the shaft (diaphysis). Wide variation is seen among type VI fractures.5
Assessment and Diagnosis
Originally termed “fender fractures” due to their frequent association with automobile injuries, fractures of the tibial plateau account for 1% of all fractures and 8% of fractures in elderly patients.6 Tibial plateau fractures occur when varus or valgus force is combined with axial loading. The fracture itself occurs when the femoral condyle is driven into the lateral or medial plateau. Bicondylar injuries occur when rigorous axial force is sustained in a fully extended knee.
Injuries may also include those of the ligaments or menisci, resulting in joint instability. Patients may present with generalized knee pain or difficulty bearing weight after sustaining injuries, such as being struck in a motor vehicle accident, being tackled, or falling from some height.4
Evaluation of a patient with a suspected tibial plateau fracture begins with a detailed history and thorough physical examination. Details regarding the mechanism of injury help to predict the pattern of the fracture and may indicate whether a more focused neurovascular exam is warranted. Low-energy injuries (often seen with Schatzker types I to III) or twisting injuries yield low suspicion for neurovascular injury or compartment syndrome. However, high-energy injuries (seen often with Schatzker types IV through VI) have a greater likelihood of resulting in complicated injuries that must be urgently or emergently treated.5
The popliteal artery is bound posteriorly and distally to the tibial plateau, and the peroneal nerve is located laterally and positioned around the fibular head. It is essential to assess for the popliteal pulse, as well as lateral lower-extremity sensation and the patient’s ability to dorsiflex. Along with motor and neurovascular injuries, presentation with a painful, strikingly swollen knee and difficulty bearing weight may indicate a hemarthrosis. Soft tissue injuries over the knee resulting from direct trauma may require a saline arthrogram to rule out communication into the joint. Furthermore, a thorough ligamentous exam of the knee is helpful in determining the extent of the injuries.3
Compartment syndrome is a serious, emergent complication that can occur with tibial plateau fractures, especially those sustained during high-energy trauma.7 The health care provider must perform serial exams of the lower extremity to assess for classic signs of compartment syndrome. Are the compartments tense or noncompressible? Does the patient have pain with passive stretch or with range of motion of the lower extremity? Is there pallor or paresthesia to the affected limb? Is the pulse weak or absent? Presence of any of the aforementioned symptoms should prompt a high suspicion for compartment syndrome, and the patient must be sent to an emergency department for urgent evaluation.5
Treatment/Rehabilitation
For Schatzker types I through III, intervention focuses on the articular cartilage examination and repair. Type IV injuries often include corresponding damage to the popliteal artery and/or peroneal nerve, and types V and VI often have such overlying soft tissue damage that temporary placement of an external fixation device is required before definitive surgical intervention can be performed.8
However, it should be noted that conservative versus surgical treatment is often debated among surgeons for treatment of Schatzker fractures. The management of a tibial plateau fracture depends on the physical demands and health of the patient, the severity of the fracture, the stability of the joint, and the surgeon’s skill set and preferences.4 Operative intervention is generally indicated for fractures with depressions greater than 2 mm (although some surgeons allow up to 1 cm of depression), fractures with joint instability, or open fractures. Injuries with concern for vascular injury or compartment syndrome are also treated both operatively and emergently. Postoperatively, patients will remain non–weight-bearing for eight to 12 weeks after surgery, and in the interim, depending on the surgeon’s preference, may or may not engage in active or passive range of motion of the knee.
Advocates of open reduction and internal fixation (ORIF) argue that this method allows for the fracture reduction and anatomic alignment to be directly examined, but they also acknowledge that this approach compromises a great deal of soft tissue surrounding the proximal tibia.9,10
In order to reduce soft tissue damage, some surgeons favor external fixation. Initial use of this surgical technique results in minimal soft tissue swelling and allows early range of motion. While the external fixation device is in place, there is a risk for pin site infection, and proper site care must be provided.6,11
Generally, the treatment of tibial plateau fractures is considered successful when the fracture reduction is sustained, the patient’s functional capacity and axial loading are restored, and the articular surface is reconstructed. As a rule, nonoperative treatment is reserved for tibial plateau fractures that are minimally depressed or nondisplaced, or for patients with advanced osteoporosis. Under these circumstances, after a non–weight-bearing period of four to eight weeks, patients will begin to perform protected and partial weight bearing using a hinged knee brace.2 Early active range of motion, along with isometric exercises to strengthen the quadriceps, is recommended.
Whether surgical or conservative treatment is chosen, complications of tibial plateau fractures include knee stiffness, wound breakdown and infection, malunion or nonunion, vascular or neurologic injury, prominent or painful hardware, or avascular necrosis of fragmented bone pieces.4
CONCLUSION
The primary care practitioner must never overlook patients’ complaints of knee pain, especially after varus or valgus stress injuries or axial loading injuries to the knee. The patient may be able to ambulate; however, ordering a radiograph is an easy method for evaluation and for ruling out tibial plateau injuries. If there is any question regarding the presence of fracture with plain radiographs and/or the clinical exam warrants it, CT is an appropriate second diagnostic intervention.
Should a tibial plateau fracture present in a primary care or urgent care setting, thorough examination of neurovascular status and risk for compartment syndrome must be done urgently, followed by a referral to an orthopedic surgeon or emergency department.
REFERENCES
1. Schatzker J, McBroom R, Bruce D. The tibial plateau fracture: the Toronto experience, 1968–1975. Clin Orthop Relat Res. 1979;(138): 94-104.
2. Marsh JL. Tibial plateau fractures. In: Bucholz RW, Court-Brown CM, Heckman HD, Tornetta P. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:1780-1831.
3. Egol K, Koval KJ, Zuckerman JD. Tibial plateau. In: Egol K, Koval KJ, Zuckerman JD. Handbook of Fractures. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:455-463.
4. Fenton PP, Porter KK. Tibial plateau fractures: a review. Trauma. 2011;13(3):181-187.
5. Markhardt BK, Gross JM, Monu JU. Schatzker classification of tibial plateau fractures: use of CT and MR imaging improves assessment. Radiographics. 2009;29(2):585-597.
6. Lewis C. Does the mode of fixation of tibial plateau fractures, i.e. external fixation versus internal fixation, influence the time to union? A systematic review of the literature. Eur J Orthopaed Surg Traumatol. 2008;18(5):365-370.
7. Weinlein J, Schmidt A. Acute compartment syndrome in tibial plateau fractures—beware! J Knee Surg. 2010;31(1):9-16.
8. te Stroet MA, Holla M, Biert J, van Kampen A. The value of CT scan compared to plain radiographs for the classification and treatment plan in tibial plateau fractures. Emerg Radiol. 2011;18(4):279-283.
9. Musahl V, Tarkin I, Kobbe P, et al. New trends and techniques in open reduction and internal fixation of fractures of the tibial plateau. J Bone Joint Surg Br. 2009;91(4):426-433.
10. Toro-Arbelaez JB, Gardner MJ, Shindle MK, et al. Open reduction and internal fixation of intraarticular tibial plateau nonunions. Injury. 2007;38(3):378-383.
11. Marsh JL, Smith ST, Do TT. External fixation and limited internal fixation for complex fractures of the tibial plateau. J Bone Joint Surg Am. 1995;77(5):661-673.
A 30-year-old man sustained traumatic amputations of three of his left fingers while at work. A heavy object fell when a supporting chain snapped; although he moved quickly, three of his left distal fingers were caught under the object. He was flown to a hospital for definitive hand care.
During the preadmission history and physical, it was noted that the patient had mild right knee pain in addition to his finger injuries. He had experienced no head injury and no loss of consciousness or other complaints. He did not remember injuring his leg, although he said it might have been struck by the falling object; all he could remember was the injury to his fingers.
On physical exam, the only abnormality other than the man’s traumatic finger amputations was mild right knee edema and a small bruised area medially. Initially, he complained of mild pain on palpation and moderate pain with passive range of motion, but range of motion was intact. His pain was worse at the proximal, medial tibial area, and he had mild lateral mid-calf tenderness though no bruising. Distally, his right lower extremity motor and sensory function were intact, and he had no open wounds or skin breakdown. He had 2+ dorsalis pedis pulse and 1+ posterior tibial pulse. The toes were pink and warm with brisk capillary refill. All compartments were soft and compressible.
Upon review of his plain radiographs (three views of the right knee), the patient was noted to have a severely comminuted medial tibial plateau fracture that extended to the midline in the region of the tibial spine, with mild depression of the fracture fragments measuring about 6 mm (see Figures 1a, 1b, and 1c). This would translate into a Schatzker IV classification type1 fracture (see Figure 22,3).
The man was admitted and underwent emergent surgery on his injured left fingers that night. Further diagnostic knee testing was performed, including CT and MRI (see Figures 3 and 4). Three days after admission, the patient underwent open reduction and internal fixation (plating) of the right medial, proximal tibia (see Figure 5). He has done very well since without issue.
DISCUSSION
Fractures of the tibial plateau occur along the articular, or joint, surface of the proximal tibia. The plateau consists of lateral and medial condylar surfaces. These concave structures function as an articulation point for the cartilaginous menisci and the femoral condyles.4 The medial plateau and condyle are stronger than those of the lateral side, and therefore are less commonly fractured. An elevated intercondylar eminence divides the lateral and medial plateaus, providing an attachment site for the cruciate ligaments.3
The Schatzker classification system1 is most commonly used to describe the types of tibial plateau fractures (as seen in Figure 22,3). Schatzker et al1 divided these injuries into six categories, according to the impact of increased energy exerted onto the bone; the rising classification numbers indicate an increase in complexity and severity and usually a worsening prognosis.
The type I fracture represents a split fracture of the lateral plateau. Typically, a fracture of this type has depression or displacement measuring less than 4 mm.
Type II tibial plateau fractures, the most common Schatzker injury, are lateral plateau fractures with depression noted at the split. Not always evident on plain radiographs, this depression can often be overlooked, and the injury mistaken for a type I fracture. The depression is measured vertically from the lower edge of the medial plateau to the lowest depression point of the lateral plateau.5
Type III fractures, the least common among the Schatzker injuries, are described as pure depression fractures of the lateral plateau. These fractures do not have an appreciable “split” along the plateau and are usually found in older patients with osteopenia.2
The Schatzker type IV injury is a medial fracture with displacement or depression to a portion of the plateau. The fracture may be split or comminuted and may originate in the intercondylar area.
Type V fractures, also known as “bicondylar fractures,” affect both the lateral and medial plateau. An inverted “Y” pattern is frequently seen, and there may be additional involvement of the intercondylar eminence. Type V fractures differ from type VI injuries in that there is no disturbance of the metaphyseal-diaphyseal connection. Thus, type VI fractures also include a transverse component that separates the condyles (metaphysis) of the bone from the shaft (diaphysis). Wide variation is seen among type VI fractures.5
Assessment and Diagnosis
Originally termed “fender fractures” due to their frequent association with automobile injuries, fractures of the tibial plateau account for 1% of all fractures and 8% of fractures in elderly patients.6 Tibial plateau fractures occur when varus or valgus force is combined with axial loading. The fracture itself occurs when the femoral condyle is driven into the lateral or medial plateau. Bicondylar injuries occur when rigorous axial force is sustained in a fully extended knee.
Injuries may also include those of the ligaments or menisci, resulting in joint instability. Patients may present with generalized knee pain or difficulty bearing weight after sustaining injuries, such as being struck in a motor vehicle accident, being tackled, or falling from some height.4
Evaluation of a patient with a suspected tibial plateau fracture begins with a detailed history and thorough physical examination. Details regarding the mechanism of injury help to predict the pattern of the fracture and may indicate whether a more focused neurovascular exam is warranted. Low-energy injuries (often seen with Schatzker types I to III) or twisting injuries yield low suspicion for neurovascular injury or compartment syndrome. However, high-energy injuries (seen often with Schatzker types IV through VI) have a greater likelihood of resulting in complicated injuries that must be urgently or emergently treated.5
The popliteal artery is bound posteriorly and distally to the tibial plateau, and the peroneal nerve is located laterally and positioned around the fibular head. It is essential to assess for the popliteal pulse, as well as lateral lower-extremity sensation and the patient’s ability to dorsiflex. Along with motor and neurovascular injuries, presentation with a painful, strikingly swollen knee and difficulty bearing weight may indicate a hemarthrosis. Soft tissue injuries over the knee resulting from direct trauma may require a saline arthrogram to rule out communication into the joint. Furthermore, a thorough ligamentous exam of the knee is helpful in determining the extent of the injuries.3
Compartment syndrome is a serious, emergent complication that can occur with tibial plateau fractures, especially those sustained during high-energy trauma.7 The health care provider must perform serial exams of the lower extremity to assess for classic signs of compartment syndrome. Are the compartments tense or noncompressible? Does the patient have pain with passive stretch or with range of motion of the lower extremity? Is there pallor or paresthesia to the affected limb? Is the pulse weak or absent? Presence of any of the aforementioned symptoms should prompt a high suspicion for compartment syndrome, and the patient must be sent to an emergency department for urgent evaluation.5
Treatment/Rehabilitation
For Schatzker types I through III, intervention focuses on the articular cartilage examination and repair. Type IV injuries often include corresponding damage to the popliteal artery and/or peroneal nerve, and types V and VI often have such overlying soft tissue damage that temporary placement of an external fixation device is required before definitive surgical intervention can be performed.8
However, it should be noted that conservative versus surgical treatment is often debated among surgeons for treatment of Schatzker fractures. The management of a tibial plateau fracture depends on the physical demands and health of the patient, the severity of the fracture, the stability of the joint, and the surgeon’s skill set and preferences.4 Operative intervention is generally indicated for fractures with depressions greater than 2 mm (although some surgeons allow up to 1 cm of depression), fractures with joint instability, or open fractures. Injuries with concern for vascular injury or compartment syndrome are also treated both operatively and emergently. Postoperatively, patients will remain non–weight-bearing for eight to 12 weeks after surgery, and in the interim, depending on the surgeon’s preference, may or may not engage in active or passive range of motion of the knee.
Advocates of open reduction and internal fixation (ORIF) argue that this method allows for the fracture reduction and anatomic alignment to be directly examined, but they also acknowledge that this approach compromises a great deal of soft tissue surrounding the proximal tibia.9,10
In order to reduce soft tissue damage, some surgeons favor external fixation. Initial use of this surgical technique results in minimal soft tissue swelling and allows early range of motion. While the external fixation device is in place, there is a risk for pin site infection, and proper site care must be provided.6,11
Generally, the treatment of tibial plateau fractures is considered successful when the fracture reduction is sustained, the patient’s functional capacity and axial loading are restored, and the articular surface is reconstructed. As a rule, nonoperative treatment is reserved for tibial plateau fractures that are minimally depressed or nondisplaced, or for patients with advanced osteoporosis. Under these circumstances, after a non–weight-bearing period of four to eight weeks, patients will begin to perform protected and partial weight bearing using a hinged knee brace.2 Early active range of motion, along with isometric exercises to strengthen the quadriceps, is recommended.
Whether surgical or conservative treatment is chosen, complications of tibial plateau fractures include knee stiffness, wound breakdown and infection, malunion or nonunion, vascular or neurologic injury, prominent or painful hardware, or avascular necrosis of fragmented bone pieces.4
CONCLUSION
The primary care practitioner must never overlook patients’ complaints of knee pain, especially after varus or valgus stress injuries or axial loading injuries to the knee. The patient may be able to ambulate; however, ordering a radiograph is an easy method for evaluation and for ruling out tibial plateau injuries. If there is any question regarding the presence of fracture with plain radiographs and/or the clinical exam warrants it, CT is an appropriate second diagnostic intervention.
Should a tibial plateau fracture present in a primary care or urgent care setting, thorough examination of neurovascular status and risk for compartment syndrome must be done urgently, followed by a referral to an orthopedic surgeon or emergency department.
REFERENCES
1. Schatzker J, McBroom R, Bruce D. The tibial plateau fracture: the Toronto experience, 1968–1975. Clin Orthop Relat Res. 1979;(138): 94-104.
2. Marsh JL. Tibial plateau fractures. In: Bucholz RW, Court-Brown CM, Heckman HD, Tornetta P. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:1780-1831.
3. Egol K, Koval KJ, Zuckerman JD. Tibial plateau. In: Egol K, Koval KJ, Zuckerman JD. Handbook of Fractures. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:455-463.
4. Fenton PP, Porter KK. Tibial plateau fractures: a review. Trauma. 2011;13(3):181-187.
5. Markhardt BK, Gross JM, Monu JU. Schatzker classification of tibial plateau fractures: use of CT and MR imaging improves assessment. Radiographics. 2009;29(2):585-597.
6. Lewis C. Does the mode of fixation of tibial plateau fractures, i.e. external fixation versus internal fixation, influence the time to union? A systematic review of the literature. Eur J Orthopaed Surg Traumatol. 2008;18(5):365-370.
7. Weinlein J, Schmidt A. Acute compartment syndrome in tibial plateau fractures—beware! J Knee Surg. 2010;31(1):9-16.
8. te Stroet MA, Holla M, Biert J, van Kampen A. The value of CT scan compared to plain radiographs for the classification and treatment plan in tibial plateau fractures. Emerg Radiol. 2011;18(4):279-283.
9. Musahl V, Tarkin I, Kobbe P, et al. New trends and techniques in open reduction and internal fixation of fractures of the tibial plateau. J Bone Joint Surg Br. 2009;91(4):426-433.
10. Toro-Arbelaez JB, Gardner MJ, Shindle MK, et al. Open reduction and internal fixation of intraarticular tibial plateau nonunions. Injury. 2007;38(3):378-383.
11. Marsh JL, Smith ST, Do TT. External fixation and limited internal fixation for complex fractures of the tibial plateau. J Bone Joint Surg Am. 1995;77(5):661-673.
A 30-year-old man sustained traumatic amputations of three of his left fingers while at work. A heavy object fell when a supporting chain snapped; although he moved quickly, three of his left distal fingers were caught under the object. He was flown to a hospital for definitive hand care.
During the preadmission history and physical, it was noted that the patient had mild right knee pain in addition to his finger injuries. He had experienced no head injury and no loss of consciousness or other complaints. He did not remember injuring his leg, although he said it might have been struck by the falling object; all he could remember was the injury to his fingers.
On physical exam, the only abnormality other than the man’s traumatic finger amputations was mild right knee edema and a small bruised area medially. Initially, he complained of mild pain on palpation and moderate pain with passive range of motion, but range of motion was intact. His pain was worse at the proximal, medial tibial area, and he had mild lateral mid-calf tenderness though no bruising. Distally, his right lower extremity motor and sensory function were intact, and he had no open wounds or skin breakdown. He had 2+ dorsalis pedis pulse and 1+ posterior tibial pulse. The toes were pink and warm with brisk capillary refill. All compartments were soft and compressible.
Upon review of his plain radiographs (three views of the right knee), the patient was noted to have a severely comminuted medial tibial plateau fracture that extended to the midline in the region of the tibial spine, with mild depression of the fracture fragments measuring about 6 mm (see Figures 1a, 1b, and 1c). This would translate into a Schatzker IV classification type1 fracture (see Figure 22,3).
The man was admitted and underwent emergent surgery on his injured left fingers that night. Further diagnostic knee testing was performed, including CT and MRI (see Figures 3 and 4). Three days after admission, the patient underwent open reduction and internal fixation (plating) of the right medial, proximal tibia (see Figure 5). He has done very well since without issue.
DISCUSSION
Fractures of the tibial plateau occur along the articular, or joint, surface of the proximal tibia. The plateau consists of lateral and medial condylar surfaces. These concave structures function as an articulation point for the cartilaginous menisci and the femoral condyles.4 The medial plateau and condyle are stronger than those of the lateral side, and therefore are less commonly fractured. An elevated intercondylar eminence divides the lateral and medial plateaus, providing an attachment site for the cruciate ligaments.3
The Schatzker classification system1 is most commonly used to describe the types of tibial plateau fractures (as seen in Figure 22,3). Schatzker et al1 divided these injuries into six categories, according to the impact of increased energy exerted onto the bone; the rising classification numbers indicate an increase in complexity and severity and usually a worsening prognosis.
The type I fracture represents a split fracture of the lateral plateau. Typically, a fracture of this type has depression or displacement measuring less than 4 mm.
Type II tibial plateau fractures, the most common Schatzker injury, are lateral plateau fractures with depression noted at the split. Not always evident on plain radiographs, this depression can often be overlooked, and the injury mistaken for a type I fracture. The depression is measured vertically from the lower edge of the medial plateau to the lowest depression point of the lateral plateau.5
Type III fractures, the least common among the Schatzker injuries, are described as pure depression fractures of the lateral plateau. These fractures do not have an appreciable “split” along the plateau and are usually found in older patients with osteopenia.2
The Schatzker type IV injury is a medial fracture with displacement or depression to a portion of the plateau. The fracture may be split or comminuted and may originate in the intercondylar area.
Type V fractures, also known as “bicondylar fractures,” affect both the lateral and medial plateau. An inverted “Y” pattern is frequently seen, and there may be additional involvement of the intercondylar eminence. Type V fractures differ from type VI injuries in that there is no disturbance of the metaphyseal-diaphyseal connection. Thus, type VI fractures also include a transverse component that separates the condyles (metaphysis) of the bone from the shaft (diaphysis). Wide variation is seen among type VI fractures.5
Assessment and Diagnosis
Originally termed “fender fractures” due to their frequent association with automobile injuries, fractures of the tibial plateau account for 1% of all fractures and 8% of fractures in elderly patients.6 Tibial plateau fractures occur when varus or valgus force is combined with axial loading. The fracture itself occurs when the femoral condyle is driven into the lateral or medial plateau. Bicondylar injuries occur when rigorous axial force is sustained in a fully extended knee.
Injuries may also include those of the ligaments or menisci, resulting in joint instability. Patients may present with generalized knee pain or difficulty bearing weight after sustaining injuries, such as being struck in a motor vehicle accident, being tackled, or falling from some height.4
Evaluation of a patient with a suspected tibial plateau fracture begins with a detailed history and thorough physical examination. Details regarding the mechanism of injury help to predict the pattern of the fracture and may indicate whether a more focused neurovascular exam is warranted. Low-energy injuries (often seen with Schatzker types I to III) or twisting injuries yield low suspicion for neurovascular injury or compartment syndrome. However, high-energy injuries (seen often with Schatzker types IV through VI) have a greater likelihood of resulting in complicated injuries that must be urgently or emergently treated.5
The popliteal artery is bound posteriorly and distally to the tibial plateau, and the peroneal nerve is located laterally and positioned around the fibular head. It is essential to assess for the popliteal pulse, as well as lateral lower-extremity sensation and the patient’s ability to dorsiflex. Along with motor and neurovascular injuries, presentation with a painful, strikingly swollen knee and difficulty bearing weight may indicate a hemarthrosis. Soft tissue injuries over the knee resulting from direct trauma may require a saline arthrogram to rule out communication into the joint. Furthermore, a thorough ligamentous exam of the knee is helpful in determining the extent of the injuries.3
Compartment syndrome is a serious, emergent complication that can occur with tibial plateau fractures, especially those sustained during high-energy trauma.7 The health care provider must perform serial exams of the lower extremity to assess for classic signs of compartment syndrome. Are the compartments tense or noncompressible? Does the patient have pain with passive stretch or with range of motion of the lower extremity? Is there pallor or paresthesia to the affected limb? Is the pulse weak or absent? Presence of any of the aforementioned symptoms should prompt a high suspicion for compartment syndrome, and the patient must be sent to an emergency department for urgent evaluation.5
Treatment/Rehabilitation
For Schatzker types I through III, intervention focuses on the articular cartilage examination and repair. Type IV injuries often include corresponding damage to the popliteal artery and/or peroneal nerve, and types V and VI often have such overlying soft tissue damage that temporary placement of an external fixation device is required before definitive surgical intervention can be performed.8
However, it should be noted that conservative versus surgical treatment is often debated among surgeons for treatment of Schatzker fractures. The management of a tibial plateau fracture depends on the physical demands and health of the patient, the severity of the fracture, the stability of the joint, and the surgeon’s skill set and preferences.4 Operative intervention is generally indicated for fractures with depressions greater than 2 mm (although some surgeons allow up to 1 cm of depression), fractures with joint instability, or open fractures. Injuries with concern for vascular injury or compartment syndrome are also treated both operatively and emergently. Postoperatively, patients will remain non–weight-bearing for eight to 12 weeks after surgery, and in the interim, depending on the surgeon’s preference, may or may not engage in active or passive range of motion of the knee.
Advocates of open reduction and internal fixation (ORIF) argue that this method allows for the fracture reduction and anatomic alignment to be directly examined, but they also acknowledge that this approach compromises a great deal of soft tissue surrounding the proximal tibia.9,10
In order to reduce soft tissue damage, some surgeons favor external fixation. Initial use of this surgical technique results in minimal soft tissue swelling and allows early range of motion. While the external fixation device is in place, there is a risk for pin site infection, and proper site care must be provided.6,11
Generally, the treatment of tibial plateau fractures is considered successful when the fracture reduction is sustained, the patient’s functional capacity and axial loading are restored, and the articular surface is reconstructed. As a rule, nonoperative treatment is reserved for tibial plateau fractures that are minimally depressed or nondisplaced, or for patients with advanced osteoporosis. Under these circumstances, after a non–weight-bearing period of four to eight weeks, patients will begin to perform protected and partial weight bearing using a hinged knee brace.2 Early active range of motion, along with isometric exercises to strengthen the quadriceps, is recommended.
Whether surgical or conservative treatment is chosen, complications of tibial plateau fractures include knee stiffness, wound breakdown and infection, malunion or nonunion, vascular or neurologic injury, prominent or painful hardware, or avascular necrosis of fragmented bone pieces.4
CONCLUSION
The primary care practitioner must never overlook patients’ complaints of knee pain, especially after varus or valgus stress injuries or axial loading injuries to the knee. The patient may be able to ambulate; however, ordering a radiograph is an easy method for evaluation and for ruling out tibial plateau injuries. If there is any question regarding the presence of fracture with plain radiographs and/or the clinical exam warrants it, CT is an appropriate second diagnostic intervention.
Should a tibial plateau fracture present in a primary care or urgent care setting, thorough examination of neurovascular status and risk for compartment syndrome must be done urgently, followed by a referral to an orthopedic surgeon or emergency department.
REFERENCES
1. Schatzker J, McBroom R, Bruce D. The tibial plateau fracture: the Toronto experience, 1968–1975. Clin Orthop Relat Res. 1979;(138): 94-104.
2. Marsh JL. Tibial plateau fractures. In: Bucholz RW, Court-Brown CM, Heckman HD, Tornetta P. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:1780-1831.
3. Egol K, Koval KJ, Zuckerman JD. Tibial plateau. In: Egol K, Koval KJ, Zuckerman JD. Handbook of Fractures. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:455-463.
4. Fenton PP, Porter KK. Tibial plateau fractures: a review. Trauma. 2011;13(3):181-187.
5. Markhardt BK, Gross JM, Monu JU. Schatzker classification of tibial plateau fractures: use of CT and MR imaging improves assessment. Radiographics. 2009;29(2):585-597.
6. Lewis C. Does the mode of fixation of tibial plateau fractures, i.e. external fixation versus internal fixation, influence the time to union? A systematic review of the literature. Eur J Orthopaed Surg Traumatol. 2008;18(5):365-370.
7. Weinlein J, Schmidt A. Acute compartment syndrome in tibial plateau fractures—beware! J Knee Surg. 2010;31(1):9-16.
8. te Stroet MA, Holla M, Biert J, van Kampen A. The value of CT scan compared to plain radiographs for the classification and treatment plan in tibial plateau fractures. Emerg Radiol. 2011;18(4):279-283.
9. Musahl V, Tarkin I, Kobbe P, et al. New trends and techniques in open reduction and internal fixation of fractures of the tibial plateau. J Bone Joint Surg Br. 2009;91(4):426-433.
10. Toro-Arbelaez JB, Gardner MJ, Shindle MK, et al. Open reduction and internal fixation of intraarticular tibial plateau nonunions. Injury. 2007;38(3):378-383.
11. Marsh JL, Smith ST, Do TT. External fixation and limited internal fixation for complex fractures of the tibial plateau. J Bone Joint Surg Am. 1995;77(5):661-673.
Thyroid Peroxidase Antibodies
Q: I have a patient with premature ovarian failure (diagnosed when she was 32) who is now in her late 40s. She is fatigued, and a blood test revealed a thyroid peroxidase antibodies level of 587 IU/mL. Would you supplement with thyroid replacement hormone, even though she has a TSH of 1.004?
The short answer is: No. Thyroid peroxidase (TPO) antibodies are a marker for the presence of autoimmune thyroid disease. Blood test results for TPO antibodies are positive in 95% of patients with chronic lymphocytic thyroiditis, also known as Hashimoto’s disease, and in 50% to 80% of patients with Graves’ disease.
Patients with high levels of TPO antibodies are at risk for future thyroid dysfunction. Not all patients with Hashimoto’s develop hypothyroidism, and if present, it may not persist. Patients with Hashimoto’s, although rarely, can experience a change from a hypothyroid to a euthyroid or even a hyperthyroid state, because of the development of coexisting TSH-receptor antibodies (TRAb), which include thyroid-stimulating immunoglobulin (TSI) and thyrotropin-binding inhibitory immunoglobulin (TBII), as seen in Graves’ disease.
Thyroid nodules are common with Hashimoto’s and are associated with a small risk (5% to 7%) for thyroid cancer. Sudden enlargement of the thyroid gland in a patient with Hashimoto’s should raise concern about thyroid lymphoma. Some endocrinologists will give supplemental thyroid hormone to a patient with Hashimoto’s, even if the TSH is normal, in an attempt to shrink the size of the gland. However, the closer the TSH is to < 1, the less room there is to further suppress it without making the patient overtly hyperthyroid, and the less likely it is that you will achieve much shrinkage of the gland.
Therefore, in the absence of a symptomatic goiter, there is no clinical reason to initiate any therapy. Even with mildly elevated TSH levels (5 to 10 mIU/L; ie, subclinical hypothyroidism) in an asymptomatic patient, there is considerable controversy about thyroid hormone initiation when the free T4 and T3 levels are normal. Most authorities agree that treatment should be initiated in most patients when the TSH rises above 10 mIU/L, regardless of symptoms. However, there are clearer indications to start thyroid hormone in women who want to become, or who are, pregnant, to maintain a TSH of < 2.5 mIU/L. Also, individuals with depression or hyperlipidemia warrant extra consideration for the use of thyroid hormone.
Since this particular patient had premature ovarian failure, which is often autoimmune in nature, she must be considered at risk for future development of hypothyroidism. This patient should be followed annually to ensure that her TSH does not rise. Should she develop symptoms suggestive of hypothyroidism and her TSH rises above 3, some endocrinologists would initiate a brief empiric trial of thyroid replacement to see if her symptoms respond when the TSH lowers again. If they do not, the thyroid hormone might be stopped, and the patient should continue to be followed.
Note: The definition of a “normal” TSH is evolving. Levels above 3.0 (suggested normal therapeutic range: 0.5 to 3.0) are considered possibly suspicious in symptomatic young people, while levels slightly above the normal reference range (5 to 7 mIU/L) may be deemed normal for the asymptomatic geriatric population.
The other point to remember is that when a clinician initiates any thyroid therapy, some patients fixate on the thyroid as the only source of their symptoms, such as fatigue, weight gain, and obesity, to the exclusion of any other etiologies. For example, sleep deprivation is a far more common cause of fatigue in our “open 24 hours” society, and lifestyle remains the major cause of obesity. Thus, there can be unintended consequences of a diagnosis of thyroid “disease.”
SUGGESTED READING
American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. www.aace.com/publications/guidelines. Accessed March 5, 2012.
Bremner AP, Feddema P, Leedman PJ, et al. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012 Feb 16; [Epub ahead of print].
Hutfless S, Matos P, Talor MV, et al. Significance of prediagnostic thyroid antibodies in women with autoimmune thyroid disease. J Clin Endocrinol Metab. 2011;96(9):E1466-E1471.
Kauffman RP, Castracane VD. Premature ovarian failure associated with autoimmune polyglandular syndrome: pathophysiological mechanisms and future fertility. J Womens Health (Larchmt). 2003;12(5):513-520.
Q: I have a patient with premature ovarian failure (diagnosed when she was 32) who is now in her late 40s. She is fatigued, and a blood test revealed a thyroid peroxidase antibodies level of 587 IU/mL. Would you supplement with thyroid replacement hormone, even though she has a TSH of 1.004?
The short answer is: No. Thyroid peroxidase (TPO) antibodies are a marker for the presence of autoimmune thyroid disease. Blood test results for TPO antibodies are positive in 95% of patients with chronic lymphocytic thyroiditis, also known as Hashimoto’s disease, and in 50% to 80% of patients with Graves’ disease.
Patients with high levels of TPO antibodies are at risk for future thyroid dysfunction. Not all patients with Hashimoto’s develop hypothyroidism, and if present, it may not persist. Patients with Hashimoto’s, although rarely, can experience a change from a hypothyroid to a euthyroid or even a hyperthyroid state, because of the development of coexisting TSH-receptor antibodies (TRAb), which include thyroid-stimulating immunoglobulin (TSI) and thyrotropin-binding inhibitory immunoglobulin (TBII), as seen in Graves’ disease.
Thyroid nodules are common with Hashimoto’s and are associated with a small risk (5% to 7%) for thyroid cancer. Sudden enlargement of the thyroid gland in a patient with Hashimoto’s should raise concern about thyroid lymphoma. Some endocrinologists will give supplemental thyroid hormone to a patient with Hashimoto’s, even if the TSH is normal, in an attempt to shrink the size of the gland. However, the closer the TSH is to < 1, the less room there is to further suppress it without making the patient overtly hyperthyroid, and the less likely it is that you will achieve much shrinkage of the gland.
Therefore, in the absence of a symptomatic goiter, there is no clinical reason to initiate any therapy. Even with mildly elevated TSH levels (5 to 10 mIU/L; ie, subclinical hypothyroidism) in an asymptomatic patient, there is considerable controversy about thyroid hormone initiation when the free T4 and T3 levels are normal. Most authorities agree that treatment should be initiated in most patients when the TSH rises above 10 mIU/L, regardless of symptoms. However, there are clearer indications to start thyroid hormone in women who want to become, or who are, pregnant, to maintain a TSH of < 2.5 mIU/L. Also, individuals with depression or hyperlipidemia warrant extra consideration for the use of thyroid hormone.
Since this particular patient had premature ovarian failure, which is often autoimmune in nature, she must be considered at risk for future development of hypothyroidism. This patient should be followed annually to ensure that her TSH does not rise. Should she develop symptoms suggestive of hypothyroidism and her TSH rises above 3, some endocrinologists would initiate a brief empiric trial of thyroid replacement to see if her symptoms respond when the TSH lowers again. If they do not, the thyroid hormone might be stopped, and the patient should continue to be followed.
Note: The definition of a “normal” TSH is evolving. Levels above 3.0 (suggested normal therapeutic range: 0.5 to 3.0) are considered possibly suspicious in symptomatic young people, while levels slightly above the normal reference range (5 to 7 mIU/L) may be deemed normal for the asymptomatic geriatric population.
The other point to remember is that when a clinician initiates any thyroid therapy, some patients fixate on the thyroid as the only source of their symptoms, such as fatigue, weight gain, and obesity, to the exclusion of any other etiologies. For example, sleep deprivation is a far more common cause of fatigue in our “open 24 hours” society, and lifestyle remains the major cause of obesity. Thus, there can be unintended consequences of a diagnosis of thyroid “disease.”
SUGGESTED READING
American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. www.aace.com/publications/guidelines. Accessed March 5, 2012.
Bremner AP, Feddema P, Leedman PJ, et al. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012 Feb 16; [Epub ahead of print].
Hutfless S, Matos P, Talor MV, et al. Significance of prediagnostic thyroid antibodies in women with autoimmune thyroid disease. J Clin Endocrinol Metab. 2011;96(9):E1466-E1471.
Kauffman RP, Castracane VD. Premature ovarian failure associated with autoimmune polyglandular syndrome: pathophysiological mechanisms and future fertility. J Womens Health (Larchmt). 2003;12(5):513-520.
Q: I have a patient with premature ovarian failure (diagnosed when she was 32) who is now in her late 40s. She is fatigued, and a blood test revealed a thyroid peroxidase antibodies level of 587 IU/mL. Would you supplement with thyroid replacement hormone, even though she has a TSH of 1.004?
The short answer is: No. Thyroid peroxidase (TPO) antibodies are a marker for the presence of autoimmune thyroid disease. Blood test results for TPO antibodies are positive in 95% of patients with chronic lymphocytic thyroiditis, also known as Hashimoto’s disease, and in 50% to 80% of patients with Graves’ disease.
Patients with high levels of TPO antibodies are at risk for future thyroid dysfunction. Not all patients with Hashimoto’s develop hypothyroidism, and if present, it may not persist. Patients with Hashimoto’s, although rarely, can experience a change from a hypothyroid to a euthyroid or even a hyperthyroid state, because of the development of coexisting TSH-receptor antibodies (TRAb), which include thyroid-stimulating immunoglobulin (TSI) and thyrotropin-binding inhibitory immunoglobulin (TBII), as seen in Graves’ disease.
Thyroid nodules are common with Hashimoto’s and are associated with a small risk (5% to 7%) for thyroid cancer. Sudden enlargement of the thyroid gland in a patient with Hashimoto’s should raise concern about thyroid lymphoma. Some endocrinologists will give supplemental thyroid hormone to a patient with Hashimoto’s, even if the TSH is normal, in an attempt to shrink the size of the gland. However, the closer the TSH is to < 1, the less room there is to further suppress it without making the patient overtly hyperthyroid, and the less likely it is that you will achieve much shrinkage of the gland.
Therefore, in the absence of a symptomatic goiter, there is no clinical reason to initiate any therapy. Even with mildly elevated TSH levels (5 to 10 mIU/L; ie, subclinical hypothyroidism) in an asymptomatic patient, there is considerable controversy about thyroid hormone initiation when the free T4 and T3 levels are normal. Most authorities agree that treatment should be initiated in most patients when the TSH rises above 10 mIU/L, regardless of symptoms. However, there are clearer indications to start thyroid hormone in women who want to become, or who are, pregnant, to maintain a TSH of < 2.5 mIU/L. Also, individuals with depression or hyperlipidemia warrant extra consideration for the use of thyroid hormone.
Since this particular patient had premature ovarian failure, which is often autoimmune in nature, she must be considered at risk for future development of hypothyroidism. This patient should be followed annually to ensure that her TSH does not rise. Should she develop symptoms suggestive of hypothyroidism and her TSH rises above 3, some endocrinologists would initiate a brief empiric trial of thyroid replacement to see if her symptoms respond when the TSH lowers again. If they do not, the thyroid hormone might be stopped, and the patient should continue to be followed.
Note: The definition of a “normal” TSH is evolving. Levels above 3.0 (suggested normal therapeutic range: 0.5 to 3.0) are considered possibly suspicious in symptomatic young people, while levels slightly above the normal reference range (5 to 7 mIU/L) may be deemed normal for the asymptomatic geriatric population.
The other point to remember is that when a clinician initiates any thyroid therapy, some patients fixate on the thyroid as the only source of their symptoms, such as fatigue, weight gain, and obesity, to the exclusion of any other etiologies. For example, sleep deprivation is a far more common cause of fatigue in our “open 24 hours” society, and lifestyle remains the major cause of obesity. Thus, there can be unintended consequences of a diagnosis of thyroid “disease.”
SUGGESTED READING
American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. www.aace.com/publications/guidelines. Accessed March 5, 2012.
Bremner AP, Feddema P, Leedman PJ, et al. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012 Feb 16; [Epub ahead of print].
Hutfless S, Matos P, Talor MV, et al. Significance of prediagnostic thyroid antibodies in women with autoimmune thyroid disease. J Clin Endocrinol Metab. 2011;96(9):E1466-E1471.
Kauffman RP, Castracane VD. Premature ovarian failure associated with autoimmune polyglandular syndrome: pathophysiological mechanisms and future fertility. J Womens Health (Larchmt). 2003;12(5):513-520.
Premature Hair Graying
What's Eating You? The Sticktight Flea (Echidnophaga gallinacea)
Clonazepam dosing
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Your surgical toolbox should include topical hemostatic agents—here is why
Vessel-sealing devices and hemostatic adjuvants are expanding the surgical armamentarium. These products provide a spectrum of alternatives that can serve you and your surgical patient well when traditional techniques for obtaining hemostasis fail to provide a satisfactory result. (Keep in mind, however, that technology is no substitute for excellent technique!)
In this article, we highlight three common scenarios in which topical hemostatic agents may be useful during gynecologic surgery. In addition, in the sidebar, five surgeons describe the hemostatic products they rely on most often—and tell why.

Following hysterectomy, persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to the site of bladder mobilization, may be managed with the aid of a topical hemostatic agent—in this case, a fibrin sealant.
When the site of bleeding is difficult to reach
CASE 1: Oozing at the site of bladder mobilization
You perform total hysterectomy in a 44-year-old woman who has uterine fibroids. After the procedure, you notice persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to where the bladder was mobilized.
How do you manage the oozing?
Wide mobilization of the bladder is a vital step in the safe performance of hysterectomy. Adhesions may complicate the process if the patient has had previous abdominal surgery, infection, or inflammation. Following mobilization of the bladder and removal of the uterus, bleeding may be visible along the adventitia of the posterior bladder wall or along the anterior surface of the vagina, distal to the cuff, as it is in this case (see the illustration).
Judicious application of an energy source is an option, but thermal injury to the bladder is a concern. A good alternative is proper placement of a hemostatic suture, but it can sometimes be difficult to avoid incorporating the bladder or injuring or obstructing the nearby ureter.
In this case, the location of the bleeding deep in the operative field poses a challenge, because of limited exposure and the proximity of the bladder and ureters. Virtually any hemostatic agent would work well in this circumstance (TABLE). For example, a flowable agent or fibrin sealant could be thoroughly applied to the area during a minimally invasive or open procedure and would naturally conform to the irregularities in the tissue, particularly the junction between the vagina and bladder flap.
A pliable product such as Surgicel Nu-Knit or Fibrillar would also work well in these circumstances, although successful application during laparoscopy may depend on the size of the trocar. For example, Nu-Knit would require trimming to a size suitable for passage through a trocar, made easier by moistening with saline. The weave of Fibrillar makes it more challenging to pass, intact, through a trocar; rolling the material into a cylindrical shape may reduce its diameter and allow it to pass more easily.
CASE 1: Resolved
You apply a fibrin sealant to the site of bleeding, and the oozing abates. Once complete hemostasis is ensured, you conclude the surgery and transfer the patient to recovery, where she does well.
Profiles in hemostasis: Strengths and weaknesses of topical agent
| Agent (brands) | Composition | Forms available | Mechanism of action | Advantages | Caveats | Duration | Relative cost* |
|---|---|---|---|---|---|---|---|
| Physical agents | |||||||
| Gelatin matrix (Gelfoam, Gelfilm, Surgifoam) | Porcine- derived collagen | Sponge, film, powder | Provides physical matrix for clot formation | Non-antigenic; neutral pH; may be used with thrombin | Material expansion may cause compression; Not for use in closed spaces or near nerve structures | 4–6 weeks | $ |
| Oxidized regenerated cellulose (Surgicel Fibrillar, Surgicel Nu-Knit) | Wood pulp | Mesh or packed fibers | Provides physical matrix for clot formation; acidic pH causes hemolysis and local clot formation | Pliable, easy to place through laparoscope; acidic pH has antimicrobial effect | Works best in a dry field. Acidic pH inactivates biologic agents, such as thrombin, and may increase inflammation. Avoid using excess material. | 2–4 weeks | $ |
| Microfibrillar collagen (Avitene, Instat, Helitene Helistat) | Bovine-derived collagen | Powder, non-woven sheet, sponge | Absorbable acid salt. Provides physical scaffold for platelet activation and clot initiation. | Sheet form may be passed through laparoscope; minimal expansion | Rare allergic reactions reported; may contribute to granuloma formation | 8–12 weeks | $$ |
| Biologically active agents | |||||||
| Topical thrombin (Thrombin-JMI, Recothrom, Evithrom, rh Thrombin) | Bovine, human, or recombinant | Liquid | Promotes conversion of fibrinogen to fibrin | May be combined effectively with physical agents of neutral pH; recombinant human thrombin will be available in the near future | Risk of blood-borne infection with non-recombinant human thrombin; risk of anaphylaxis and antibody formation with bovine thrombin | N/A | $$ |
| Hemostatic matrix (Floseal, Surgiflo) | Thrombin plus gelatin | Foam | Gelatin granules provide expansion and compression while thrombin initiates clot formation | May be used in areas of small arterial bleeding | Requires contact with blood | 6–8 weeks | $$$ |
| Fibrin sealants (Evicel, Tisseel, Crosseal) | Human | Liquid | Combination of fibrinogen and thrombin causes cleavage of fibrinogen to fibrin and resultant clot initiation | Fast-acting; hemostatic and adhesive properties; works well for diffusely oozing surfaces | Contraindicated in patients who have a history of anaphylactic reaction to serum-derived products or IgA deficiency | 10–14 days | $$$ |
| * Median cost for use in one case Key: $=inexpensive; $$=moderately expensive; $$$=expensive | |||||||
Controlling bleeding without injuring underlying tissue
CASE 2: After adhesiolysis, bleeding at multiple sites
You perform adnexectomy on a 47-year-old woman who has a large (7 to 8 cm), benign ovarian mass. As you operate, you discover that the lesion is adherent to the sigmoid mesentery and the posterior aspect of the uterus; it is also adherent to the pelvic sidewall, directly along the course of the ureter. Although you are able to release the various adhesive attachments, persistent bleeding is noted at multiple pinpoint areas along the mesentery, uterine serosa, and pelvic sidewall, even after the application of direct pressure.
What do you do next?
Although cautery can be used liberally on the uterus, its application to mesentery carries a risk of injury to the mesenteric vessels and bowel wall. Caution is advised when you are attempting to control bleeding on the peritoneum overlying the ureter, whether you are using suture ligature or an energy source. Ideally, you should identify the ureter using a retroperitoneal approach and mobilize it laterally before employing any of these techniques.
There are several potential approaches to the bleeding described in Case 2, all of them involving hemostatic adjuvants. The first decision you need to make, however, is whether to address each region separately or all sites in unison. If you opt to address them together—either during an open procedure or laparoscopy—a fibrin sealant (e.g., Evicel, Tisseel) is one option. It can be applied using a dripping technique or aerosolization, either of which allows for broad application of a thin film of the agent. The limitation of this approach is the volume of agent required to resolve the bleeding, with a potential need for multiple doses to completely coat the area.
Because fibrin sealants function independently of the patient’s coagulation cascade, they are particularly useful in the presence of disseminated intravascular coagulation (DIC) and other coagulopathies that might limit the effectiveness of preparations that require the patient’s own serum.
An alternative approach to Case 2 is to apply an oxidized regenerated cellulose (ORC) derivative directly to the affected areas. Various forms are available (e.g., Surgicel Fibrillar, Surgicel Nu-Knit). These ORC products can be cut and customized to the area in need of hemostasis, allowing each site to be addressed individually. These agents typically remain adherent after they are applied due to the nature of the interaction between the product, blood, and tissue.
A liquid or foam hemostatic agent (e.g., Surgiflo, Floseal, topical thrombin) could also be employed in this case, but application can be a challenge on a large area with a heterogeneous topography because of the tendency of such agents to migrate under the force of gravity, pooling away from the source of bleeding.
Is combining agents a good idea?
Although they are not typically approved for use in combination, sequential application of hemostatic agents may be considered when bleeding persists.
All hemostatic agents work best in combination with the application of pressure. It usually is advisable to use moist gauze for this purpose because it can be lifted away without significant adherence to the underlying hemostatic complex, avoiding clot
disruption.
CASE 2: Resolved
You opt to use an ORC product, customizing it to fit each bleeding site, and apply direct pressure. When hemostasis has been achieved at all sites, you complete the operation. The patient has an uneventful postoperative course.
Protect structures along the pelvic sidewall
CASE 3: When the application of pressure isn’t enough
While performing a left salpingo-oophorectomy for a 12-cm ovarian lesion, you use a retroperitoneal approach to identify the structures along the pelvic sidewall. During identification of the ureter, you encounter bleeding from a small vessel in the adjacent fatty areolar tissue. After a period of observation, during which you apply pressure to the area of concern, bleeding persists.
What hemostatic agent do you employ to stop it?
The careful application of steady pressure is often enough to safely control bleeding in the area of the pelvic sidewall. In the event that pressure alone fails to resolve the bleeding, however, it is critical to choose a remedy that avoids injuring the ureter, iliac vessels, and infundibulopelvic ligament. Wide exposure of the space may allow for direct identification of the point of bleeding and precise application of cautery, a hemoclip, or a tie. When this approach is not feasible, other solutions must be sought.
When traditional hemostatic techniques fail in delicate anatomic sites, such as the periureteral area, hemostatic agents are an effective option that can minimize the risk of injury to surrounding vital structures. The contour of the space calls for a product that can intercalate, such as a foam, sealant, or Surgicel Fibrillar. Direct, precise application to the point of bleeding is critical, and the “bunching up” of a more rigid and bulky agent may limit its application to the area of concern. Use of a moist gauze to apply direct pressure after application of the agent will increase the likelihood of success.
CASE 3: Resolved
You decide to apply a foam hemostatic agent because of its ability to conform to the irregular space. You also continue to apply gentle pressure to the point of bleeding, using a moist gauze. Within minutes, hemostasis is achieved. You are then able to finish the operation.
Other variables to consider
As these three cases illustrate, the use of hemostatic agents to control surgical bleeding requires an individualized approach. The site and amount of bleeding, as well as the patient’s hemodynamic and coagulation status, are key variables to be considered when selecting an agent.
For instance, because of their components, fibrin sealants can function independently of the patient’s coagulation status. ORC products provide a matrix that facilitates platelet aggregration and may be less effective when anti-platelet agents have been used.
It is also appropriate for the surgeon to be familiar with the relative cost of the agents available at his or her institution. In particular, when several agents may be equally effective in a particular set of circumstances, cost may be the determining factor.
Availability of these agents varies from one institution to the next; as a result, it can be challenging to maintain familiarity with all of the products in the marketplace. Having access to a diverse, readily available set of “go to” agents is critical to ensure rapid application in a clinical setting.
The surgeon’s preference also is important, particularly in regard to the ease of preparation and handling. Some agents may not be as suitable for minimally invasive procedures (see TABLE). For others, special laparoscopic applicators are available.
When using a hemostatic agent, it pays to consider the duration of its effect in the surgical site. Both the quantity of the agent that is applied and characteristics of the local operative site influence how quickly the agent degrades. Keep this in mind when imaging studies are planned for the early postoperative period. An ORC preparation, for example, may appear with small pockets of air that resemble an abscess. Effective communication with the radiology team is critical to avoid the misinterpretation of findings.
Curious to discover the preferences and practices of surgeons likely to utilize topical hemostatic agents, OBG Management polled several experienced and expert surgeons, including members of the journal’s Board of Editors and Virtual Board of Editors. Their diverse responses offer a snapshot of gynecologic surgical practice in 2012—but all agree that hemostatic products are no substitute for sound surgical technique.
JANELLE YATES, SENIOR EDITOR





We want to hear from you! Tell us what you think.
Recommended reading
Achneck HE, Sileshi B, Jamiolkowski RM, et al. A comprehensive review of topical hemostatic agents: efficacy and recommendations for use. Ann Surg. 2010;251(2):217-228.
Chapman WC, Singla N, Genyk Y, et al. A phase 3, randomized, double-blind comparative study of the efficacy and safety of topical recombinant human thrombin and bovine thrombin in surgical hemostasis. J Am Coll Surg. 2007;205(2):256-265.
Holub Z, Jabor A. Laparoscopic management of bleeding after laparoscopic or vaginal hysterectomy. JSLS. 2004;8(3):235-238.
Sharma JB, Malhotra M. Laparoscopic oxidized cellulose (Surgicel) application for small uterine perforations. Int J Gynaecol Obstet. 2003;83(3):271-275.
Sharma JB, Malhotra M. Topical oxidized cellulose for tubal hemorrhage hemostasis during laparoscopic sterilization. Int J Gynaecol Obstet. 2003;82(2):221-222.
Vessel-sealing devices and hemostatic adjuvants are expanding the surgical armamentarium. These products provide a spectrum of alternatives that can serve you and your surgical patient well when traditional techniques for obtaining hemostasis fail to provide a satisfactory result. (Keep in mind, however, that technology is no substitute for excellent technique!)
In this article, we highlight three common scenarios in which topical hemostatic agents may be useful during gynecologic surgery. In addition, in the sidebar, five surgeons describe the hemostatic products they rely on most often—and tell why.

Following hysterectomy, persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to the site of bladder mobilization, may be managed with the aid of a topical hemostatic agent—in this case, a fibrin sealant.
When the site of bleeding is difficult to reach
CASE 1: Oozing at the site of bladder mobilization
You perform total hysterectomy in a 44-year-old woman who has uterine fibroids. After the procedure, you notice persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to where the bladder was mobilized.
How do you manage the oozing?
Wide mobilization of the bladder is a vital step in the safe performance of hysterectomy. Adhesions may complicate the process if the patient has had previous abdominal surgery, infection, or inflammation. Following mobilization of the bladder and removal of the uterus, bleeding may be visible along the adventitia of the posterior bladder wall or along the anterior surface of the vagina, distal to the cuff, as it is in this case (see the illustration).
Judicious application of an energy source is an option, but thermal injury to the bladder is a concern. A good alternative is proper placement of a hemostatic suture, but it can sometimes be difficult to avoid incorporating the bladder or injuring or obstructing the nearby ureter.
In this case, the location of the bleeding deep in the operative field poses a challenge, because of limited exposure and the proximity of the bladder and ureters. Virtually any hemostatic agent would work well in this circumstance (TABLE). For example, a flowable agent or fibrin sealant could be thoroughly applied to the area during a minimally invasive or open procedure and would naturally conform to the irregularities in the tissue, particularly the junction between the vagina and bladder flap.
A pliable product such as Surgicel Nu-Knit or Fibrillar would also work well in these circumstances, although successful application during laparoscopy may depend on the size of the trocar. For example, Nu-Knit would require trimming to a size suitable for passage through a trocar, made easier by moistening with saline. The weave of Fibrillar makes it more challenging to pass, intact, through a trocar; rolling the material into a cylindrical shape may reduce its diameter and allow it to pass more easily.
CASE 1: Resolved
You apply a fibrin sealant to the site of bleeding, and the oozing abates. Once complete hemostasis is ensured, you conclude the surgery and transfer the patient to recovery, where she does well.
Profiles in hemostasis: Strengths and weaknesses of topical agent
| Agent (brands) | Composition | Forms available | Mechanism of action | Advantages | Caveats | Duration | Relative cost* |
|---|---|---|---|---|---|---|---|
| Physical agents | |||||||
| Gelatin matrix (Gelfoam, Gelfilm, Surgifoam) | Porcine- derived collagen | Sponge, film, powder | Provides physical matrix for clot formation | Non-antigenic; neutral pH; may be used with thrombin | Material expansion may cause compression; Not for use in closed spaces or near nerve structures | 4–6 weeks | $ |
| Oxidized regenerated cellulose (Surgicel Fibrillar, Surgicel Nu-Knit) | Wood pulp | Mesh or packed fibers | Provides physical matrix for clot formation; acidic pH causes hemolysis and local clot formation | Pliable, easy to place through laparoscope; acidic pH has antimicrobial effect | Works best in a dry field. Acidic pH inactivates biologic agents, such as thrombin, and may increase inflammation. Avoid using excess material. | 2–4 weeks | $ |
| Microfibrillar collagen (Avitene, Instat, Helitene Helistat) | Bovine-derived collagen | Powder, non-woven sheet, sponge | Absorbable acid salt. Provides physical scaffold for platelet activation and clot initiation. | Sheet form may be passed through laparoscope; minimal expansion | Rare allergic reactions reported; may contribute to granuloma formation | 8–12 weeks | $$ |
| Biologically active agents | |||||||
| Topical thrombin (Thrombin-JMI, Recothrom, Evithrom, rh Thrombin) | Bovine, human, or recombinant | Liquid | Promotes conversion of fibrinogen to fibrin | May be combined effectively with physical agents of neutral pH; recombinant human thrombin will be available in the near future | Risk of blood-borne infection with non-recombinant human thrombin; risk of anaphylaxis and antibody formation with bovine thrombin | N/A | $$ |
| Hemostatic matrix (Floseal, Surgiflo) | Thrombin plus gelatin | Foam | Gelatin granules provide expansion and compression while thrombin initiates clot formation | May be used in areas of small arterial bleeding | Requires contact with blood | 6–8 weeks | $$$ |
| Fibrin sealants (Evicel, Tisseel, Crosseal) | Human | Liquid | Combination of fibrinogen and thrombin causes cleavage of fibrinogen to fibrin and resultant clot initiation | Fast-acting; hemostatic and adhesive properties; works well for diffusely oozing surfaces | Contraindicated in patients who have a history of anaphylactic reaction to serum-derived products or IgA deficiency | 10–14 days | $$$ |
| * Median cost for use in one case Key: $=inexpensive; $$=moderately expensive; $$$=expensive | |||||||
Controlling bleeding without injuring underlying tissue
CASE 2: After adhesiolysis, bleeding at multiple sites
You perform adnexectomy on a 47-year-old woman who has a large (7 to 8 cm), benign ovarian mass. As you operate, you discover that the lesion is adherent to the sigmoid mesentery and the posterior aspect of the uterus; it is also adherent to the pelvic sidewall, directly along the course of the ureter. Although you are able to release the various adhesive attachments, persistent bleeding is noted at multiple pinpoint areas along the mesentery, uterine serosa, and pelvic sidewall, even after the application of direct pressure.
What do you do next?
Although cautery can be used liberally on the uterus, its application to mesentery carries a risk of injury to the mesenteric vessels and bowel wall. Caution is advised when you are attempting to control bleeding on the peritoneum overlying the ureter, whether you are using suture ligature or an energy source. Ideally, you should identify the ureter using a retroperitoneal approach and mobilize it laterally before employing any of these techniques.
There are several potential approaches to the bleeding described in Case 2, all of them involving hemostatic adjuvants. The first decision you need to make, however, is whether to address each region separately or all sites in unison. If you opt to address them together—either during an open procedure or laparoscopy—a fibrin sealant (e.g., Evicel, Tisseel) is one option. It can be applied using a dripping technique or aerosolization, either of which allows for broad application of a thin film of the agent. The limitation of this approach is the volume of agent required to resolve the bleeding, with a potential need for multiple doses to completely coat the area.
Because fibrin sealants function independently of the patient’s coagulation cascade, they are particularly useful in the presence of disseminated intravascular coagulation (DIC) and other coagulopathies that might limit the effectiveness of preparations that require the patient’s own serum.
An alternative approach to Case 2 is to apply an oxidized regenerated cellulose (ORC) derivative directly to the affected areas. Various forms are available (e.g., Surgicel Fibrillar, Surgicel Nu-Knit). These ORC products can be cut and customized to the area in need of hemostasis, allowing each site to be addressed individually. These agents typically remain adherent after they are applied due to the nature of the interaction between the product, blood, and tissue.
A liquid or foam hemostatic agent (e.g., Surgiflo, Floseal, topical thrombin) could also be employed in this case, but application can be a challenge on a large area with a heterogeneous topography because of the tendency of such agents to migrate under the force of gravity, pooling away from the source of bleeding.
Is combining agents a good idea?
Although they are not typically approved for use in combination, sequential application of hemostatic agents may be considered when bleeding persists.
All hemostatic agents work best in combination with the application of pressure. It usually is advisable to use moist gauze for this purpose because it can be lifted away without significant adherence to the underlying hemostatic complex, avoiding clot
disruption.
CASE 2: Resolved
You opt to use an ORC product, customizing it to fit each bleeding site, and apply direct pressure. When hemostasis has been achieved at all sites, you complete the operation. The patient has an uneventful postoperative course.
Protect structures along the pelvic sidewall
CASE 3: When the application of pressure isn’t enough
While performing a left salpingo-oophorectomy for a 12-cm ovarian lesion, you use a retroperitoneal approach to identify the structures along the pelvic sidewall. During identification of the ureter, you encounter bleeding from a small vessel in the adjacent fatty areolar tissue. After a period of observation, during which you apply pressure to the area of concern, bleeding persists.
What hemostatic agent do you employ to stop it?
The careful application of steady pressure is often enough to safely control bleeding in the area of the pelvic sidewall. In the event that pressure alone fails to resolve the bleeding, however, it is critical to choose a remedy that avoids injuring the ureter, iliac vessels, and infundibulopelvic ligament. Wide exposure of the space may allow for direct identification of the point of bleeding and precise application of cautery, a hemoclip, or a tie. When this approach is not feasible, other solutions must be sought.
When traditional hemostatic techniques fail in delicate anatomic sites, such as the periureteral area, hemostatic agents are an effective option that can minimize the risk of injury to surrounding vital structures. The contour of the space calls for a product that can intercalate, such as a foam, sealant, or Surgicel Fibrillar. Direct, precise application to the point of bleeding is critical, and the “bunching up” of a more rigid and bulky agent may limit its application to the area of concern. Use of a moist gauze to apply direct pressure after application of the agent will increase the likelihood of success.
CASE 3: Resolved
You decide to apply a foam hemostatic agent because of its ability to conform to the irregular space. You also continue to apply gentle pressure to the point of bleeding, using a moist gauze. Within minutes, hemostasis is achieved. You are then able to finish the operation.
Other variables to consider
As these three cases illustrate, the use of hemostatic agents to control surgical bleeding requires an individualized approach. The site and amount of bleeding, as well as the patient’s hemodynamic and coagulation status, are key variables to be considered when selecting an agent.
For instance, because of their components, fibrin sealants can function independently of the patient’s coagulation status. ORC products provide a matrix that facilitates platelet aggregration and may be less effective when anti-platelet agents have been used.
It is also appropriate for the surgeon to be familiar with the relative cost of the agents available at his or her institution. In particular, when several agents may be equally effective in a particular set of circumstances, cost may be the determining factor.
Availability of these agents varies from one institution to the next; as a result, it can be challenging to maintain familiarity with all of the products in the marketplace. Having access to a diverse, readily available set of “go to” agents is critical to ensure rapid application in a clinical setting.
The surgeon’s preference also is important, particularly in regard to the ease of preparation and handling. Some agents may not be as suitable for minimally invasive procedures (see TABLE). For others, special laparoscopic applicators are available.
When using a hemostatic agent, it pays to consider the duration of its effect in the surgical site. Both the quantity of the agent that is applied and characteristics of the local operative site influence how quickly the agent degrades. Keep this in mind when imaging studies are planned for the early postoperative period. An ORC preparation, for example, may appear with small pockets of air that resemble an abscess. Effective communication with the radiology team is critical to avoid the misinterpretation of findings.
Curious to discover the preferences and practices of surgeons likely to utilize topical hemostatic agents, OBG Management polled several experienced and expert surgeons, including members of the journal’s Board of Editors and Virtual Board of Editors. Their diverse responses offer a snapshot of gynecologic surgical practice in 2012—but all agree that hemostatic products are no substitute for sound surgical technique.
JANELLE YATES, SENIOR EDITOR





We want to hear from you! Tell us what you think.
Vessel-sealing devices and hemostatic adjuvants are expanding the surgical armamentarium. These products provide a spectrum of alternatives that can serve you and your surgical patient well when traditional techniques for obtaining hemostasis fail to provide a satisfactory result. (Keep in mind, however, that technology is no substitute for excellent technique!)
In this article, we highlight three common scenarios in which topical hemostatic agents may be useful during gynecologic surgery. In addition, in the sidebar, five surgeons describe the hemostatic products they rely on most often—and tell why.

Following hysterectomy, persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to the site of bladder mobilization, may be managed with the aid of a topical hemostatic agent—in this case, a fibrin sealant.
When the site of bleeding is difficult to reach
CASE 1: Oozing at the site of bladder mobilization
You perform total hysterectomy in a 44-year-old woman who has uterine fibroids. After the procedure, you notice persistent oozing along the anterior vaginal margin, distal to the cuff and adjacent to where the bladder was mobilized.
How do you manage the oozing?
Wide mobilization of the bladder is a vital step in the safe performance of hysterectomy. Adhesions may complicate the process if the patient has had previous abdominal surgery, infection, or inflammation. Following mobilization of the bladder and removal of the uterus, bleeding may be visible along the adventitia of the posterior bladder wall or along the anterior surface of the vagina, distal to the cuff, as it is in this case (see the illustration).
Judicious application of an energy source is an option, but thermal injury to the bladder is a concern. A good alternative is proper placement of a hemostatic suture, but it can sometimes be difficult to avoid incorporating the bladder or injuring or obstructing the nearby ureter.
In this case, the location of the bleeding deep in the operative field poses a challenge, because of limited exposure and the proximity of the bladder and ureters. Virtually any hemostatic agent would work well in this circumstance (TABLE). For example, a flowable agent or fibrin sealant could be thoroughly applied to the area during a minimally invasive or open procedure and would naturally conform to the irregularities in the tissue, particularly the junction between the vagina and bladder flap.
A pliable product such as Surgicel Nu-Knit or Fibrillar would also work well in these circumstances, although successful application during laparoscopy may depend on the size of the trocar. For example, Nu-Knit would require trimming to a size suitable for passage through a trocar, made easier by moistening with saline. The weave of Fibrillar makes it more challenging to pass, intact, through a trocar; rolling the material into a cylindrical shape may reduce its diameter and allow it to pass more easily.
CASE 1: Resolved
You apply a fibrin sealant to the site of bleeding, and the oozing abates. Once complete hemostasis is ensured, you conclude the surgery and transfer the patient to recovery, where she does well.
Profiles in hemostasis: Strengths and weaknesses of topical agent
| Agent (brands) | Composition | Forms available | Mechanism of action | Advantages | Caveats | Duration | Relative cost* |
|---|---|---|---|---|---|---|---|
| Physical agents | |||||||
| Gelatin matrix (Gelfoam, Gelfilm, Surgifoam) | Porcine- derived collagen | Sponge, film, powder | Provides physical matrix for clot formation | Non-antigenic; neutral pH; may be used with thrombin | Material expansion may cause compression; Not for use in closed spaces or near nerve structures | 4–6 weeks | $ |
| Oxidized regenerated cellulose (Surgicel Fibrillar, Surgicel Nu-Knit) | Wood pulp | Mesh or packed fibers | Provides physical matrix for clot formation; acidic pH causes hemolysis and local clot formation | Pliable, easy to place through laparoscope; acidic pH has antimicrobial effect | Works best in a dry field. Acidic pH inactivates biologic agents, such as thrombin, and may increase inflammation. Avoid using excess material. | 2–4 weeks | $ |
| Microfibrillar collagen (Avitene, Instat, Helitene Helistat) | Bovine-derived collagen | Powder, non-woven sheet, sponge | Absorbable acid salt. Provides physical scaffold for platelet activation and clot initiation. | Sheet form may be passed through laparoscope; minimal expansion | Rare allergic reactions reported; may contribute to granuloma formation | 8–12 weeks | $$ |
| Biologically active agents | |||||||
| Topical thrombin (Thrombin-JMI, Recothrom, Evithrom, rh Thrombin) | Bovine, human, or recombinant | Liquid | Promotes conversion of fibrinogen to fibrin | May be combined effectively with physical agents of neutral pH; recombinant human thrombin will be available in the near future | Risk of blood-borne infection with non-recombinant human thrombin; risk of anaphylaxis and antibody formation with bovine thrombin | N/A | $$ |
| Hemostatic matrix (Floseal, Surgiflo) | Thrombin plus gelatin | Foam | Gelatin granules provide expansion and compression while thrombin initiates clot formation | May be used in areas of small arterial bleeding | Requires contact with blood | 6–8 weeks | $$$ |
| Fibrin sealants (Evicel, Tisseel, Crosseal) | Human | Liquid | Combination of fibrinogen and thrombin causes cleavage of fibrinogen to fibrin and resultant clot initiation | Fast-acting; hemostatic and adhesive properties; works well for diffusely oozing surfaces | Contraindicated in patients who have a history of anaphylactic reaction to serum-derived products or IgA deficiency | 10–14 days | $$$ |
| * Median cost for use in one case Key: $=inexpensive; $$=moderately expensive; $$$=expensive | |||||||
Controlling bleeding without injuring underlying tissue
CASE 2: After adhesiolysis, bleeding at multiple sites
You perform adnexectomy on a 47-year-old woman who has a large (7 to 8 cm), benign ovarian mass. As you operate, you discover that the lesion is adherent to the sigmoid mesentery and the posterior aspect of the uterus; it is also adherent to the pelvic sidewall, directly along the course of the ureter. Although you are able to release the various adhesive attachments, persistent bleeding is noted at multiple pinpoint areas along the mesentery, uterine serosa, and pelvic sidewall, even after the application of direct pressure.
What do you do next?
Although cautery can be used liberally on the uterus, its application to mesentery carries a risk of injury to the mesenteric vessels and bowel wall. Caution is advised when you are attempting to control bleeding on the peritoneum overlying the ureter, whether you are using suture ligature or an energy source. Ideally, you should identify the ureter using a retroperitoneal approach and mobilize it laterally before employing any of these techniques.
There are several potential approaches to the bleeding described in Case 2, all of them involving hemostatic adjuvants. The first decision you need to make, however, is whether to address each region separately or all sites in unison. If you opt to address them together—either during an open procedure or laparoscopy—a fibrin sealant (e.g., Evicel, Tisseel) is one option. It can be applied using a dripping technique or aerosolization, either of which allows for broad application of a thin film of the agent. The limitation of this approach is the volume of agent required to resolve the bleeding, with a potential need for multiple doses to completely coat the area.
Because fibrin sealants function independently of the patient’s coagulation cascade, they are particularly useful in the presence of disseminated intravascular coagulation (DIC) and other coagulopathies that might limit the effectiveness of preparations that require the patient’s own serum.
An alternative approach to Case 2 is to apply an oxidized regenerated cellulose (ORC) derivative directly to the affected areas. Various forms are available (e.g., Surgicel Fibrillar, Surgicel Nu-Knit). These ORC products can be cut and customized to the area in need of hemostasis, allowing each site to be addressed individually. These agents typically remain adherent after they are applied due to the nature of the interaction between the product, blood, and tissue.
A liquid or foam hemostatic agent (e.g., Surgiflo, Floseal, topical thrombin) could also be employed in this case, but application can be a challenge on a large area with a heterogeneous topography because of the tendency of such agents to migrate under the force of gravity, pooling away from the source of bleeding.
Is combining agents a good idea?
Although they are not typically approved for use in combination, sequential application of hemostatic agents may be considered when bleeding persists.
All hemostatic agents work best in combination with the application of pressure. It usually is advisable to use moist gauze for this purpose because it can be lifted away without significant adherence to the underlying hemostatic complex, avoiding clot
disruption.
CASE 2: Resolved
You opt to use an ORC product, customizing it to fit each bleeding site, and apply direct pressure. When hemostasis has been achieved at all sites, you complete the operation. The patient has an uneventful postoperative course.
Protect structures along the pelvic sidewall
CASE 3: When the application of pressure isn’t enough
While performing a left salpingo-oophorectomy for a 12-cm ovarian lesion, you use a retroperitoneal approach to identify the structures along the pelvic sidewall. During identification of the ureter, you encounter bleeding from a small vessel in the adjacent fatty areolar tissue. After a period of observation, during which you apply pressure to the area of concern, bleeding persists.
What hemostatic agent do you employ to stop it?
The careful application of steady pressure is often enough to safely control bleeding in the area of the pelvic sidewall. In the event that pressure alone fails to resolve the bleeding, however, it is critical to choose a remedy that avoids injuring the ureter, iliac vessels, and infundibulopelvic ligament. Wide exposure of the space may allow for direct identification of the point of bleeding and precise application of cautery, a hemoclip, or a tie. When this approach is not feasible, other solutions must be sought.
When traditional hemostatic techniques fail in delicate anatomic sites, such as the periureteral area, hemostatic agents are an effective option that can minimize the risk of injury to surrounding vital structures. The contour of the space calls for a product that can intercalate, such as a foam, sealant, or Surgicel Fibrillar. Direct, precise application to the point of bleeding is critical, and the “bunching up” of a more rigid and bulky agent may limit its application to the area of concern. Use of a moist gauze to apply direct pressure after application of the agent will increase the likelihood of success.
CASE 3: Resolved
You decide to apply a foam hemostatic agent because of its ability to conform to the irregular space. You also continue to apply gentle pressure to the point of bleeding, using a moist gauze. Within minutes, hemostasis is achieved. You are then able to finish the operation.
Other variables to consider
As these three cases illustrate, the use of hemostatic agents to control surgical bleeding requires an individualized approach. The site and amount of bleeding, as well as the patient’s hemodynamic and coagulation status, are key variables to be considered when selecting an agent.
For instance, because of their components, fibrin sealants can function independently of the patient’s coagulation status. ORC products provide a matrix that facilitates platelet aggregration and may be less effective when anti-platelet agents have been used.
It is also appropriate for the surgeon to be familiar with the relative cost of the agents available at his or her institution. In particular, when several agents may be equally effective in a particular set of circumstances, cost may be the determining factor.
Availability of these agents varies from one institution to the next; as a result, it can be challenging to maintain familiarity with all of the products in the marketplace. Having access to a diverse, readily available set of “go to” agents is critical to ensure rapid application in a clinical setting.
The surgeon’s preference also is important, particularly in regard to the ease of preparation and handling. Some agents may not be as suitable for minimally invasive procedures (see TABLE). For others, special laparoscopic applicators are available.
When using a hemostatic agent, it pays to consider the duration of its effect in the surgical site. Both the quantity of the agent that is applied and characteristics of the local operative site influence how quickly the agent degrades. Keep this in mind when imaging studies are planned for the early postoperative period. An ORC preparation, for example, may appear with small pockets of air that resemble an abscess. Effective communication with the radiology team is critical to avoid the misinterpretation of findings.
Curious to discover the preferences and practices of surgeons likely to utilize topical hemostatic agents, OBG Management polled several experienced and expert surgeons, including members of the journal’s Board of Editors and Virtual Board of Editors. Their diverse responses offer a snapshot of gynecologic surgical practice in 2012—but all agree that hemostatic products are no substitute for sound surgical technique.
JANELLE YATES, SENIOR EDITOR





We want to hear from you! Tell us what you think.
Recommended reading
Achneck HE, Sileshi B, Jamiolkowski RM, et al. A comprehensive review of topical hemostatic agents: efficacy and recommendations for use. Ann Surg. 2010;251(2):217-228.
Chapman WC, Singla N, Genyk Y, et al. A phase 3, randomized, double-blind comparative study of the efficacy and safety of topical recombinant human thrombin and bovine thrombin in surgical hemostasis. J Am Coll Surg. 2007;205(2):256-265.
Holub Z, Jabor A. Laparoscopic management of bleeding after laparoscopic or vaginal hysterectomy. JSLS. 2004;8(3):235-238.
Sharma JB, Malhotra M. Laparoscopic oxidized cellulose (Surgicel) application for small uterine perforations. Int J Gynaecol Obstet. 2003;83(3):271-275.
Sharma JB, Malhotra M. Topical oxidized cellulose for tubal hemorrhage hemostasis during laparoscopic sterilization. Int J Gynaecol Obstet. 2003;82(2):221-222.
Recommended reading
Achneck HE, Sileshi B, Jamiolkowski RM, et al. A comprehensive review of topical hemostatic agents: efficacy and recommendations for use. Ann Surg. 2010;251(2):217-228.
Chapman WC, Singla N, Genyk Y, et al. A phase 3, randomized, double-blind comparative study of the efficacy and safety of topical recombinant human thrombin and bovine thrombin in surgical hemostasis. J Am Coll Surg. 2007;205(2):256-265.
Holub Z, Jabor A. Laparoscopic management of bleeding after laparoscopic or vaginal hysterectomy. JSLS. 2004;8(3):235-238.
Sharma JB, Malhotra M. Laparoscopic oxidized cellulose (Surgicel) application for small uterine perforations. Int J Gynaecol Obstet. 2003;83(3):271-275.
Sharma JB, Malhotra M. Topical oxidized cellulose for tubal hemorrhage hemostasis during laparoscopic sterilization. Int J Gynaecol Obstet. 2003;82(2):221-222.
Clonazepam dosing
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Dr. Scott Freeman’s useful discussion of targeting acute risk factors in suicidal patients (“Suicide assessment: Targeting acute risk factors,” Current Psychiatry, January 2012, p. 52-57) ends by resolving the clinical vignette with a summary of hospital treatment. Apart from failing to indicate any psychotherapeutic inroads, Dr. Freeman seems to support prescribing clonazepam, 0.5 mg twice daily and 1 mg at bedtime. Clonazepam apparently “worked” by alleviating the patient’s anxiety and insomnia, but defied any pharmacologic rationale insofar as clonazepam has a slow onset and long half-life, making 3 doses per day irrational. This treatment strategy also risks problems of cumulative excess in the long run after discharge.
Aggressive pharmacotherapy may be the hallmark of modern acute hospital treatment, but surely it should incorporate careful understanding of specific medications’ pharmacodynamics, especially when relying on benzodiazepines. Needless to say, beginning a psychological process in the hospital also appears to have been shortchanged.
Sara Hartley, MD
Lecturer, Clinical Skills Program
University of California,
Berkeley-University of California,
San Francisco Joint Medical Program
Berkeley, CA
Dr. Freeman responds
I appreciate Dr. Hartley’s interest in my article. Although I agree with her that psychotherapy is an integral part of any treatment plan, the clinical vignette was used only to emphasize the need to aggressively and quickly start antidepressant and, more importantly, anxiolytic pharmacologic treatment in acutely suicidal patients with severe anxiety and depression.
With regard to clonazepam’s pharmacokinetics, although it does have a long half-life, it is only weakly lipophilic compared with other long-acting benzodiazepines such as diazepam. In fact, clonazepam has been shown to be less lipophilic than lorazepam,1 meaning it has a much smaller volume of distribution and less accumulation in peripheral adipose tissue. Therefore, one would not be concerned about significant drug accumulation leading to unexpected toxicity with a less lipophilic agent such as clonazepam.
I do not agree that dosing clonazepam 3 times a day, especially in an acute crisis, is “irrational,” as Dr. Hartley suggests. According to the package insert, although clonazepam is recommended to be administered twice daily for panic disorder, it can be given 3 times a day for seizure disorders.2
Scott A. Freeman, MD
Medical Director
Schizophrenia and Bipolar Disorder Inpatient Unit
McLean Hospital
Belmont, MA
Using melatonin to reset the clock of hospitalized older patients
Helping hospitalized geriatric patients maintain an appropriate sleep-wake cycle can be a challenge. Older patients’ circadian rhythm may be affected by several factors—eg, obstructive sleep apnea and restless leg syndrome—that contribute to disrupted sleep and daytime fatigue. Some patients may have dementing illnesses that could dysregulate sleep. Many older patients experience delirium during hospitalization, of which sleep-wake cycle disturbances are a hallmark. Finally, geriatric patients’ natural sleep pattern often does not mimic a hospital’s typical schedule.
Sleep medication side effects
Medications used to promote sleep can cause side effects in geriatric patients. Benzodiazepine use by older adults is discouraged because these medications could cause falls or contribute to delirium. Non-benzodiazepine hypnotics such as zolpidem, zaleplon, and eszopiclone pose a similar risk. Medications containing diphenhydramine predispose patients to deliriogenic effects via their anticholinergic properties. Tricyclic antidepressants carry risks, such as delirium secondary to anticholinergic effects, orthostatic hypotension, falls from α-1 blockade, and cardiac arrythmias.
Atypical antipsychotics sometimes are used off-label to help initiate sleep, but they carry a “black-box” warning regarding sudden death from cardiovascular events in geriatric patients with dementia. Hydroxyzine and trazodone also are associated with side effects such as orthostatic hypotension and daytime sedation, and are not always effective.
Melatonin is a hormone secreted by the pineal gland in response to darkness, under the control of the suprachiasmatic nucleus (SCN), and is thought to promote sleep via synchronizing effects on the SCN.1 Melatonin is available as an over-the-counter dietary supplement and via prescription in dosages of 1 or 3 mg. The typical effective dose is 3 to 9 mg.1 Patients should take melatonin in the mid-evening, ideally between 7 pm and 8 pm, and effects become evident after a few days. Side effects are rare; the most common are headache and nausea. Daytime sedation and vivid dreams also have been reported. Melatonin can be used safely in conjunction with other sleep aids and its major drug-drug interactions involve enhancing the effects of other sedatives.2
We have found melatonin to be effective for treating sleep disturbances in older hospitalized patients. Its effectiveness may stem from the high incidence of dysregulated or calcified pineal glands in geriatric patients, which leads to a marked reduction in melatonin secretion.3 Recent evidence also suggests melatonin may reduce the incidence of delirium in older adults, and it has been proposed as a delirium treatment in post-operative and intensive care unit settings.4
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. de Jonghe A, Korevaar JC, van Munster BC, et al. Effectiveness of melatonin treatment on circadian rhythm disturbances in dementia. Are there implications for delirium? A systematic review. Int J Geriatr Psychiatry. 2010;25(12):1201-1208.
2. Werneke U, Turner T, Priebe S. Complementary medicines in psychiatry: review of effectiveness and safety. Br J Psychiatry. 2006;188:109-121.
3. Schmid HA. Decreased melatonin biosynthesis calcium flux, pineal gland calcification and aging: a hypothetical framework. Gerontology. 1993;39(4):189-199.
4. Al-Aama T, Brymer C, Gutmanis I, et al. Melatonin decreases delirium in elderly patients: a randomized, placebo-controlled trial. Int J Geriatr Psychiatry. 2011;26(7):687-694.
Helping hospitalized geriatric patients maintain an appropriate sleep-wake cycle can be a challenge. Older patients’ circadian rhythm may be affected by several factors—eg, obstructive sleep apnea and restless leg syndrome—that contribute to disrupted sleep and daytime fatigue. Some patients may have dementing illnesses that could dysregulate sleep. Many older patients experience delirium during hospitalization, of which sleep-wake cycle disturbances are a hallmark. Finally, geriatric patients’ natural sleep pattern often does not mimic a hospital’s typical schedule.
Sleep medication side effects
Medications used to promote sleep can cause side effects in geriatric patients. Benzodiazepine use by older adults is discouraged because these medications could cause falls or contribute to delirium. Non-benzodiazepine hypnotics such as zolpidem, zaleplon, and eszopiclone pose a similar risk. Medications containing diphenhydramine predispose patients to deliriogenic effects via their anticholinergic properties. Tricyclic antidepressants carry risks, such as delirium secondary to anticholinergic effects, orthostatic hypotension, falls from α-1 blockade, and cardiac arrythmias.
Atypical antipsychotics sometimes are used off-label to help initiate sleep, but they carry a “black-box” warning regarding sudden death from cardiovascular events in geriatric patients with dementia. Hydroxyzine and trazodone also are associated with side effects such as orthostatic hypotension and daytime sedation, and are not always effective.
Melatonin is a hormone secreted by the pineal gland in response to darkness, under the control of the suprachiasmatic nucleus (SCN), and is thought to promote sleep via synchronizing effects on the SCN.1 Melatonin is available as an over-the-counter dietary supplement and via prescription in dosages of 1 or 3 mg. The typical effective dose is 3 to 9 mg.1 Patients should take melatonin in the mid-evening, ideally between 7 pm and 8 pm, and effects become evident after a few days. Side effects are rare; the most common are headache and nausea. Daytime sedation and vivid dreams also have been reported. Melatonin can be used safely in conjunction with other sleep aids and its major drug-drug interactions involve enhancing the effects of other sedatives.2
We have found melatonin to be effective for treating sleep disturbances in older hospitalized patients. Its effectiveness may stem from the high incidence of dysregulated or calcified pineal glands in geriatric patients, which leads to a marked reduction in melatonin secretion.3 Recent evidence also suggests melatonin may reduce the incidence of delirium in older adults, and it has been proposed as a delirium treatment in post-operative and intensive care unit settings.4
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Helping hospitalized geriatric patients maintain an appropriate sleep-wake cycle can be a challenge. Older patients’ circadian rhythm may be affected by several factors—eg, obstructive sleep apnea and restless leg syndrome—that contribute to disrupted sleep and daytime fatigue. Some patients may have dementing illnesses that could dysregulate sleep. Many older patients experience delirium during hospitalization, of which sleep-wake cycle disturbances are a hallmark. Finally, geriatric patients’ natural sleep pattern often does not mimic a hospital’s typical schedule.
Sleep medication side effects
Medications used to promote sleep can cause side effects in geriatric patients. Benzodiazepine use by older adults is discouraged because these medications could cause falls or contribute to delirium. Non-benzodiazepine hypnotics such as zolpidem, zaleplon, and eszopiclone pose a similar risk. Medications containing diphenhydramine predispose patients to deliriogenic effects via their anticholinergic properties. Tricyclic antidepressants carry risks, such as delirium secondary to anticholinergic effects, orthostatic hypotension, falls from α-1 blockade, and cardiac arrythmias.
Atypical antipsychotics sometimes are used off-label to help initiate sleep, but they carry a “black-box” warning regarding sudden death from cardiovascular events in geriatric patients with dementia. Hydroxyzine and trazodone also are associated with side effects such as orthostatic hypotension and daytime sedation, and are not always effective.
Melatonin is a hormone secreted by the pineal gland in response to darkness, under the control of the suprachiasmatic nucleus (SCN), and is thought to promote sleep via synchronizing effects on the SCN.1 Melatonin is available as an over-the-counter dietary supplement and via prescription in dosages of 1 or 3 mg. The typical effective dose is 3 to 9 mg.1 Patients should take melatonin in the mid-evening, ideally between 7 pm and 8 pm, and effects become evident after a few days. Side effects are rare; the most common are headache and nausea. Daytime sedation and vivid dreams also have been reported. Melatonin can be used safely in conjunction with other sleep aids and its major drug-drug interactions involve enhancing the effects of other sedatives.2
We have found melatonin to be effective for treating sleep disturbances in older hospitalized patients. Its effectiveness may stem from the high incidence of dysregulated or calcified pineal glands in geriatric patients, which leads to a marked reduction in melatonin secretion.3 Recent evidence also suggests melatonin may reduce the incidence of delirium in older adults, and it has been proposed as a delirium treatment in post-operative and intensive care unit settings.4
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. de Jonghe A, Korevaar JC, van Munster BC, et al. Effectiveness of melatonin treatment on circadian rhythm disturbances in dementia. Are there implications for delirium? A systematic review. Int J Geriatr Psychiatry. 2010;25(12):1201-1208.
2. Werneke U, Turner T, Priebe S. Complementary medicines in psychiatry: review of effectiveness and safety. Br J Psychiatry. 2006;188:109-121.
3. Schmid HA. Decreased melatonin biosynthesis calcium flux, pineal gland calcification and aging: a hypothetical framework. Gerontology. 1993;39(4):189-199.
4. Al-Aama T, Brymer C, Gutmanis I, et al. Melatonin decreases delirium in elderly patients: a randomized, placebo-controlled trial. Int J Geriatr Psychiatry. 2011;26(7):687-694.
1. de Jonghe A, Korevaar JC, van Munster BC, et al. Effectiveness of melatonin treatment on circadian rhythm disturbances in dementia. Are there implications for delirium? A systematic review. Int J Geriatr Psychiatry. 2010;25(12):1201-1208.
2. Werneke U, Turner T, Priebe S. Complementary medicines in psychiatry: review of effectiveness and safety. Br J Psychiatry. 2006;188:109-121.
3. Schmid HA. Decreased melatonin biosynthesis calcium flux, pineal gland calcification and aging: a hypothetical framework. Gerontology. 1993;39(4):189-199.
4. Al-Aama T, Brymer C, Gutmanis I, et al. Melatonin decreases delirium in elderly patients: a randomized, placebo-controlled trial. Int J Geriatr Psychiatry. 2011;26(7):687-694.
Benzodiazepines: A versatile clinical tool
Since the discovery of chlordiazepoxide in the 1950s, benzodiazepines have revolutionized the treatment of anxiety and insomnia, largely because of their improved safety profile compared with barbiturates, formerly the preferred sedative-hypnotic.1 In addition to their anxiolytic and sedative-hypnotic effects, benzodiazepines exhibit anterograde amnesia, anticonvulsant, and muscle relaxant properties.1 Psychiatrists use benzodiazepines to treat anxiety and sleep disorders, acute agitation, alcohol withdrawal, catatonia, and psychotropic side effects such as akathisia. This article highlights the evidence for using benzodiazepines in anxiety and other disorders and why they generally should not be used for obsessive-compulsive disorder and posttraumatic stress disorder (Box 1).
Current evidence indicates little support for using benzodiazepines for obsessive-compulsive disorder (OCD). The American Psychiatric Association (APA) and the World Federation of Biological Psychiatry do not recommend benzodiazepines for treating OCD because of a lack of evidence for efficacy.a,b An earlier study suggested clonazepam monotherapy was effective for OCDc; however, a more recent study did not show a benefit on rate of response or degree of symptom improvement.d Augmentation strategies with benzodiazepines also do not appear to be beneficial for OCD management. A recent double-blind, placebo-controlled study failed to demonstrate faster symptom improvement by augmenting sertraline with clonazepam, although the study had a small sample size and high drop-out rate.e
Because benzodiazepines have negligible action on core posttraumatic stress disorder (PTSD) symptoms (re-experiencing, avoidance, and hyperarousal), selective serotonin reuptake inhibitors and other agents largely have supplanted them for PTSD treatment.f Use of benzodiazepines for PTSD is associated with withdrawal symptoms, more severe symptoms after discontinuation, and possible disinhibition, and may interfere with patients’ efforts to integrate trauma experiences. Although benzodiazepines may reduce distress associated with acute trauma, there is evidence—in clinical studies and animal models—that early benzodiazepine administration fails to prevent PTSD and may increase its incidence.g The International Consensus Group on Depression and Anxiety, the APA, and the British Association for Psychopharmacology all highlight the limited role, if any, for benzodiazepines in PTSD.h-j
References
- Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
- American Psychiatric Association. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
- Hewlett WA, Vinogradov S, Agras WS. Clomipramine, clonazepam, and clonidine treatment of obsessive compulsive disorder. J Clin Psychopharmacol. 1992;12(6):420-430.
- Hollander E, Kaplan A, Stahl SM. A double-blind, placebo-controlled trial of clonazepam in obsessive-compulsive disorder. World J Biol Psychiatry. 2003;4(1):30-34.
- Crockett BA, Churchill E, Davidson JR. A double-blind combination study of clonazepam and sertraline in OCD. Ann Clin Psychiatry. 2004;16(3):127-132.
- Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs. Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
- Matar MA, Zohar J, Kaplan Z, et al. Alprazolam treatment immediately after stress exposure interferes with the normal HPA-stress response and increases vulnerability to subsequent stress in an animal model of PTSD. Eur Neuropsychopharmacol. 2009;19(4):283-295.
- Ballenger JC, Davidson JR, Lecrubier Y, et al. Consensus statement update on posttraumatic stress disorder from the international consensus group on depression and anxiety. J Clin Psychiatry. 2004;65(suppl 1):55-62.
- Ursano RJ, Bell C, Eth S, et al. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Am J Psychiatry. 2004;161(11 suppl):3-31.
- Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based guidelines for the pharmacological treatment of anxiety disorders: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2005;19(6):567-596.
Pharmacokinetic properties
Most benzodiazepines are considered to have similar efficacy; therefore, selection is based on pharmacokinetic considerations. Table 1 compares the indication, onset, and half-life of 12 commonly used benzodiazepines.2-6 Although Table 1 lists approximate equivalent doses, studies report inconsistent data. These are approximations only and should not be used independently to make therapy decisions.
Table 1
Oral benzodiazepines: Indications, onset, half-life, and equivalent doses
| Drug | FDA-approved indication(s) | Onset of action | Approximate half-life (hours) in healthy adults | Approximate equivalent dose (mg)a | Comments |
|---|---|---|---|---|---|
| Alprazolam | Anxiety disorders, panic disorder | Intermediate | 6.3 to 26.9 (IR), 10.7 to 15.8 (XR) | 0.5 | Increased risk for abuse because of greater lipid solubility |
| Chlordiazepoxide | Anxiety disorders, acute alcohol withdrawal, preoperative apprehension and anxiety | Intermediate | 24 to 48 | 10 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Clonazepam | Seizure disorders, panic disorder | Intermediate | 18 to 50 | 0.25 to 0.5 | Use caution in patients with liver disease |
| Clorazepate | Anxiety, seizures, acute alcohol withdrawal | Fast | 40 to 50 | 7.5 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Diazepam | Anxiety disorders, acute alcohol withdrawal, muscle spasms, convulsive disorders | Very fast | 20 to 100 | 5 | Risk for accumulation because of long-acting metabolites (temazepam, desmethyldiazepam, oxazepam). Increased risk for abuse because of quick onset |
| Estazolam | Insomnia | Intermediate | 10 to 24 | 0.3 to 2 | None |
| Flurazepam | Insomnia | Intermediate | 47 to 100 | 30 | Avoid in geriatric patients or patients with liver impairment |
| Lorazepam | Anxiety | Intermediate | 10 to 20 | 1 | Preferred for patients with liver impairment and geriatric patients |
| Oxazepam | Anxiety, acute alcohol withdrawal | Slow to intermediate | 5 to 20 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Quazepam | Insomnia | Intermediate | 39 to 73 | 5 to 15 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Temazepam | Insomnia | Intermediate | 3.5 to 18.4 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Triazolam | Insomnia | Fast | 1.5 to 5.5 | 0.25 | Lacks active metabolites |
| IR: immediate release; XR: extended release aInterpret with caution, conflicting data exist Source: References 2-6 | |||||
A diverse range of indications
Alcohol withdrawal. Benzodiazepines are the treatment of choice for alcohol withdrawal syndrome, particularly to prevent seizures.7 Research supports symptom-triggered therapy using the revised Clinical Institute Withdrawal Assessment for Alcohol. Benzodiazepines reduce CNS sympathetic hyperactivity to mitigate withdrawal from alcohol by decreasing tachycardia, tremor, insomnia, agitation, and anxiety. Furthermore, these agents provide prophylaxis against serious sequelae such as seizures and delirium.
Insomnia. The American Academy of Sleep Medicine considers benzodiazepine receptor agonists (BzRAs, which include benzodiazepines and non-benzodiazepines) and ramelteon first-line pharmacotherapy for primary insomnia.8 However, pharmacologic treatment should be short-term. Agents with short to intermediate half-lives and rapid onset, such as triazolam, can aid sleep initiation. Those with longer half-lives, such as temazepam, could address sleep maintenance. If a patient does not respond to the initial agent, try another medication within the same class, because patients may respond differently. Use lower starting doses in geriatric patients.9 Closely monitor for adverse effects, rebound insomnia, and potential abuse or tolerance. Identify comorbid conditions and medications that may impair sleep, and address them accordingly.
Psychological and behavioral treatments given over 4 to 8 weeks can yield stable sleep improvements for up to 2 years. If available, these interventions may be considered first-line for treating insomnia because of their lasting effects compared with BzRAs.10
Generalized anxiety disorder (GAD). Benzodiazepines effectively treat GAD because they work quickly and are well tolerated. However, there are better first-line treatment options when considering efficacy studies and dependence and tolerance concerns. One effect-size comparison of 21 double-blind, placebo-controlled trials showed that the efficacy of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and pregabalin are comparable to benzodiazepines.11 Benzodiazepines can be used in the first 2 to 3 weeks after initiating antidepressants to alleviate and prevent worsening of anxiety that may occur at the start of antidepressant therapy. Recent treatment guidelines recommend benzodiazepines as a second-line treatment or for treatment-resistant GAD in patients who do not have a substance abuse history.12,13
Panic disorder. Efficacy of benzodiazepines for panic disorder is comparable to SSRIs, SNRIs, and tricyclic antidepressants (TCAs). SSRIs and SNRIs are considered first-line treatments for panic disorder because of their favorable side effect profile.14 In practice, benzodiazepines often are combined with SSRIs, SNRIs, or TCAs. A randomized controlled trial demonstrated that paroxetine and clonazepam (mean dose 1.6 mg/d at 5 weeks) resulted in a more rapid response compared with paroxetine alone, although this difference lasted only a few weeks.15 Furthermore, this study suggested that brief treatment with clonazepam followed by a taper is as effective as sustained treatment with paroxetine and clonazepam.15
There is a lack of high-quality data on combining cognitive-behavioral therapy (CBT) and benzodiazepines for panic disorder, although a Cochrane Review found that adding a benzodiazepine to CBT did not lead to a significant difference in response compared with psychotherapy alone.16 A recent randomized controlled trial demonstrated that tapering benzodiazepines combined with CBT was associated with successful discontinuation of the drug and prevented return of panic symptoms.17
Social anxiety. A meta-analysis found that for treating social anxiety, benzodiazepines have better efficacy than SSRIs, monoamine oxidase inhibitors, and anticonvulsants.18 Longer-acting benzodiazepines may be more effective than shorter-acting agents. One study of patients with social anxiety showed a 38% response rate for alprazolam vs 20% for placebo over 12 weeks, and a similar 10-week study demonstrated a 73% recovery rate with clonazepam vs 22% for placebo.19 In addition, studies have observed that patients can be maintained on clonazepam for up to 2 years without symptom relapse and will tolerate slow-taper discontinuation.18,20 Sedation and drowsiness can be lessened by limiting clonazepam doses to 2 to 3 mg/d.
Akathisia and tremor. Akathisia, a syndrome of motor restlessness and inner turmoil, is associated with antipsychotics but can occur with SSRIs. Reducing the dosage or switching to another, usually less potent agent often can relieve akathisia. When these remedies are not tenable, consider benzodiazepines along with other medications—including beta blockers and anticholinergic agents—with demonstrated efficacy in reducing akathisia symptoms. Lorazepam, diazepam, and clonazepam have demonstrated efficacy for relieving akathisia in comparison studies with placebo, propranolol, and diphenhydramine.21,22
Drug-induced postural tremor can occur with several psychotropics, including lithium, valproic acid, antidepressants, and antipsychotics. A tremor is considered mild if a patient can drink a glass of water with 1 hand without spilling and severe if holding a glass with 2 hands is difficult. Propranolol is most commonly prescribed for these tremors, but alprazolam and clonazepam have demonstrated efficacy, either as monotherapy or coadministered with a beta blocker.23
Acute agitation. Agitated patients often have acute psychosis and/or mania or dyscontrol secondary to axis II disorders.24 Patients may be paranoid, hostile, disruptive, and combative. Rapidly initiating medication can prevent the need for more restrictive measures, such as seclusion or restraint. Antipsychotics—especially high-potency agents such as haloperidol—and benzodiazepines, as monotherapy or in combination, are a mainstay treatment. Although treatment protocols favor atypical antipsychotics over typical antipsychotics, benzodiazepines are a viable option because of their anxiolytic and sedative effects. Advantages of benzodiazepine monotherapy include decreased extrapyramidal symptoms, greater patient acceptance/preference, and increased sedation compared with antipsychotics. Lorazepam, 1 to 2 mg intramuscularly (IM) or orally, is well tolerated because of its favorable drug-drug interaction profile and lack of significant cardiac side effects. Benzodiazepines can cause respiratory depression in patients with chronic lung disease and additive sedation secondary to opiates, other sedatives/hypnotics, or alcohol. Behavioral disinhibition is rare and is associated with preexisting CNS pathology or mental retardation.25 The IM olanzapine package insert warns against coadministering IM lorazepam because of additive cardiorespiratory depressive effects and excessive somnolence.26
Catatonia. The characteristic symptoms of catatonia are immobility, negativism, muteness, and failure to eat or drink. Benzodiazepines improve these symptoms in approximately 70% to 80% of catatonic patients with affective disorders. Response rates are lower in catatonia in patients with schizophrenia.27 If catatonia in a patient with psychosis is missed, giving antipsychotics before benzodiazepines may worsen catatonic symptoms or precipitate neuroleptic malignant syndrome in some cases. When you suspect a patient has catatonia, start with lorazepam, 1 to 2 mg IV or IM, and examine the patient for diminishing catatonic signs within 1 to 2 hours. If catatonia signs lessen, begin regularly scheduled lorazepam, with dosing varying by age—be more cautious in geriatric patients—and symptom severity. Titrate benzodiazepines for stuporous patients more slowly (eg, 1 mg 3 times a day as a starting dose) than for excited catatonic patients. Lorazepam can be increased gradually as tolerated; it is not unusual for patients to require up to 8 to 12 mg/d. Electroconvulsive therapy (ECT) is the treatment of choice when catatonic patients respond poorly or partially to high-dose benzodiazepines.28,29
Benzodiazepine reversal for ECT
Benzodiazepines have anticonvulsant properties that may interfere with the therapeutic efficacy of ECT.30 A multi-center study demonstrated that lorazepam (up to 4 mg/d as needed) in the 48 hours before the first ECT session was not associated with effects on seizure threshold or duration; however, larger lorazepam dosages were associated with briefer EEG seizure duration.31 Some patients may not tolerate withholding or tapering benzodiazepines in preparation for ECT. Studies investigating flumazenil for pre-ECT benzodiazepine reversal are lacking. One retrospective analysis showed that flumazenil administration immediately before and after ECT resulted in adequate seizures with no difference in clinical outcome compared with patients who were not receiving benzodiazepines or flumazenil.32
Tapering benzodiazepines
Slow discontinuation of benzodiazepines is recommended to avoid withdrawal symptoms, such as rebound anxiety, agitation, insomnia, or seizures, particularly when use exceeds 8 weeks. The onset of withdrawal symptoms varies, depending on the medication used. Withdrawal symptoms may appear in 1 to 2 days for agents with shorter half-lives, but may not appear until 3 to 7 days for agents with longer half-lives.33Table 2 lists recommended durations for tapering benzodiazepines.33,34 In general, decrease the total daily dose by 25% the first week, another 25% the second week, then 10% a week until discontinuation. When benzodiazepine use exceeds 1 year, a slower taper is recommended; for example, decrease 10% every 1 to 2 weeks. When 20% of the dosage remains, begin a 5% dose reduction every 2 to 4 weeks. Monitor patients for withdrawal symptoms or symptom exacerbation. If either occur, consider maintaining the current benzodiazepine dose or increasing the dose for 1 to 2 weeks or longer, if necessary, then continue to taper at a slower rate.34
Table 2
Recommendations for tapering benzodiazepines
| Duration of use | Recommended taper length | Comments |
| <6 to 8 weeks | Taper may not be required | Depending on clinical judgment and patient stability/preference, consider implementing a taper, particularly if using a high-dose benzodiazepine or an agent with a short or intermediate half-life, such as alprazolam or triazolam |
| 8 weeks to 6 months | Slowly over 2 to 3 weeks | Go slower during latter half of taper. Tapering will reduce, not eliminate, withdrawal symptoms. Patients should avoid alcohol and stimulants during benzodiazepine withdrawal |
| 6 months to 1 year | Slowly over 4 to 8 weeks | |
| >1 year | Slowly over 2 to 4 months | |
| Source: References 33,34 | ||
Risks of benzodiazepine use
For most indications, benzodiazepine therapy should be short-term.35 Use exceeding 2 to 4 weeks increases the risk for dependence and withdrawal. Tell patients to avoid alcohol while taking a benzodiazepine because this combination is potentially lethal. Benzodiazepines are commonly abused and abuse can lead to unintentional drug overdose. Benzodiazepines accounted for 37% of unintentional drug overdose deaths in West Virginia in 2006; in 46% of these cases, benzodiazepines were used for nonmedical purposes. Clinicians can help reduce the risk of diversion by limiting prescriptions to 30 days with no refills.36
Older patients taking benzodiazepines are at increased risk of falls and hip fractures.37 Lorazepam, oxazepam, and temazepam—agents with shorter half-lives that are not greatly affected by pharmacokinetic changes associated with aging—are preferred for these patients.34 Patients with dementia or other CNS-compromising conditions may become confused or delirious with regular benzodiazepine dosing. Educate patients to whom you prescribe benzodiazepines about the importance of gauging their level of sedation before driving or engaging in other tasks for which sedation could compromise their safety. Benzodiazepine use during pregnancy requires a careful discussion of risks and benefits (Box 2).38
Benzodiazepine use during pregnancy has been associated with cleft palate and urogenital and neurologic malformations in the fetus.38 Although data are conflicting—particularly among recent meta-analyses that fail to demonstrate an association—some experts advise against benzodiazepine use in the first trimester. Participate in shared decision making with your patients and educate them about the potential risks and benefits of benzodiazepine use during the first trimester and throughout pregnancy. After delivery, newborns may develop “floppy baby syndrome”—which is associated with lethargy, difficulty eating, and respiratory depression—or withdrawal.38 To minimize this risk, consider tapering the benzodiazepine as the patient approaches delivery.
Related Resources
- Substance Abuse and Mental Health Services Administration. www.samhsa.gov.
- National Institute on Drug Abuse resources for medical and health professionals. www.drugabuse.gov/medical-health-professionals.
- American Academy of Sleep Medicine. www.aasmnet.org.
Drug Brand Names
- Alprazolam • Xanax
- Chlordiazepoxide • Librium, Limbitrol
- Clonazepam • Klonopin
- Clorazepate • Tranxene
- Diazepam • Valium
- Diphenhydramine • Benadryl, others
- Estazolam • ProSom
- Flumazenil • Romazicon
- Flurazepam • Dalmane
- Haloperidol • Haldol
- Lithium • Lithobid
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Paroxetine • Paxil
- Pregabalin • Lyrica
- Propranolol • Inderal, InnoPran XL, others
- Quazepam • Doral
- Ramelteon • Rozerem
- Sertraline • Zoloft
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakene, Stavzor, others
Disclosures
Drs. Bostwick and Yasugi report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Casher is a speaker for AstraZeneca and Sunovion Pharmaceuticals.
1. Mihic SJ, Harris RA. Hypnotics and sedatives. In: Brunton LL Chabner BA, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. New York, NY: McGraw Hill and Company; 2011:457-480.
2. Facts and comparisons Web site. 2011 Wolters Kluwer Health Inc. http://online.factsandcomparisons.com. Accessed August 16, 2011.
3. DuPont RL, Greene W, Lydiard RB. Sedatives and hypnotics: pharmacology and epidemiology. In: Gold MS Hermann R, eds. UpToDate. http://www.uptodate.com/contents/sedatives-and-hypnotics-abuse-and-dependence-pharmacology-and-epidemiology. Accessed August 16, 2011.
4. U.S. Food and Drug Administration. Orange book: approved drug products with therapeutic equivalence evaluations. http://www.accessdata.fda.gov/scripts/cder/ob/default.cfm. Accessed August 16, 2011.
5. Chouinard G. Issues in the clinical use of benzodiazepines: potency withdrawal, and rebound. J Clin Psychiatry. 2004;65(suppl 5):7-12.
6. Shader RI, Greenblatt DJ. Can you provide a table of equivalencies for benzodiazepines and other marketed benzodiazepine receptor agonists? J Clin Psychopharmacol. 1997;17(4):331.-
7. Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacologic interventions for the treatment of the alcohol withdrawal syndrome. Cochrane Database Syst Rev. 2011;15(6):CD008537.-
8. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4(5):487-504.
9. Foral P, Dewan N, Malesker M. Insomnia: a therapeutic review for pharmacists. Consult Pharm. 2011;26(5):332-341.
10. Riemann D, Perlis ML. The treatments of chronic insomnia: a review of benzodiazepine receptor agonists and psychological and behavioral therapies. Sleep Med Rev. 2009;13(3):205-214.
11. Hidalgo RB, Tupler LA, Davidson JR. An effect-size analysis of pharmacologic treatments of generalized anxiety disorder. J Psychopharmacol. 2007;21(8):864-872.
12. Davidson JR, Zhang W, Connor KM, et al. A psychopharmacological treatment algorithm for generalised anxiety disorder (GAD). J Psychopharmacol. 2010;24(1):3-26.
13. Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
14. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder. 2nd ed. Arlington VA: American Psychiatric Publishing, Inc.; 2009.
15. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
16. Watanabe N, Churchill R, Furukawa TA. Combined psychotherapy plus benzodiazepines for panic disorder. Cochrane Database Syst Rev. 2009;(1):CD005335.-
17. Otto MW, McHugh RK, Simon NM, et al. Efficacy of CBT for benzodiazepine discontinuation in patients with panic disorder: further evaluation. Behav Res Ther. 2010;48(8):720-727.
18. Davidson JR. Use of benzodiazepines in social anxiety disorder generalized anxiety disorder, and posttraumatic stress disorder. J Clin Psychiatry. 2004;65(suppl 5):29-33.
19. Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
20. Connor KM, Davidson JR, Potts NL, et al. Discontinuation of clonazepam in the treatment of social phobia. J Clin Psychopharmacol. 1998;18(5):373-378.
21. Miller CH, Fleischhacker WW. Managing antipsychotic-induced acute and chronic akathisia. Drug Saf. 2000;22(1):73-81.
22. Rodnitzky RL. Drug-induced movement disorders. Clin Neuropharmacol. 2002;25(3):142-151.
23. Arbaizar B, Gómez-Acebo I, Llorca J. Postural induced tremor in psychiatry. Psychiatry Clin Neurosci. 2008;62(6):638-645.
24. Casher MI, Bess JD. Manual of inpatient psychiatry. New York NY: Cambridge University Press; 2010.
25. Battaglia J. Pharmacological management of acute agitation. Drugs. 2005;65(9):1207-1222.
26. Physicians’ desk reference. Montvale NJ: PDR Network, LLC; 2010.
27. Rosebush PI, Mazurek MF. Catatonia and its treatment. Schizophr Bull. 2010;36(2):239-242.
28. Ungvari GS, Kau LS, Wai-Kwong T, et al. The pharmacological treatment of catatonia: an overview. Eur Arch Psychiatry Clin Neurosci. 2001;251(suppl 1):I31-I34.
29. Fink M, Taylor MA. Catatonia: a clinician’s guide to diagnosis and treatment. New York NY: Cambridge University Press; 2003.
30. Naguib N, Koorn R. Interactions between psychotropics anaesthetics and electroconvulsive therapy: implications for drug choice and patient management. CNS Drugs. 2002;16(4):229-247.
31. Boylan LS, Haskett RF, Mulsant BH, et al. Determinants of seizure threshold in ECT: benzodiazepine use, anesthetic dosage, and other factors. J ECT. 2000;16(1):3-18.
32. Krystal AD, Watts BV, Weiner RD, et al. The use of flumazenil in the anxious and benzodiazepine-dependent ECT patient. J ECT. 1998;14(1):5-14.
33. Melton ST, Kirkwood CK. Anxiety disorders I: generalized anxiety panic, and social anxiety disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: a pathophysiologic approach. New York, NY: McGraw-Hill Companies; 2011:1209-1228.
34. Benzodiazepine toolkit. The Pharmacist’s Letter/Prescriber’s Letter. 2011;27(4):270406.-
35. Lader M. Benzodiazepines revisited – will we ever learn? Addiction. 2011;106(12):2086-2109.
36. Toblin RL, Paulozzi LJ, Logan JE, et al. Mental illness and psychotropic drug use among prescription drug overdose deaths: a medical examiner chart review. J Clin Psychiatry. 2010;71(4):491-496.
37. Ashton H. The diagnosis and management of benzodiazepine dependence. Curr Opin Psychiatry. 2005;18(3):249-255.
38. Menon SJ. Psychotropic medication during pregnancy and lactation. Arch Gynecol Obstet. 2008;277(1):1-13.
Since the discovery of chlordiazepoxide in the 1950s, benzodiazepines have revolutionized the treatment of anxiety and insomnia, largely because of their improved safety profile compared with barbiturates, formerly the preferred sedative-hypnotic.1 In addition to their anxiolytic and sedative-hypnotic effects, benzodiazepines exhibit anterograde amnesia, anticonvulsant, and muscle relaxant properties.1 Psychiatrists use benzodiazepines to treat anxiety and sleep disorders, acute agitation, alcohol withdrawal, catatonia, and psychotropic side effects such as akathisia. This article highlights the evidence for using benzodiazepines in anxiety and other disorders and why they generally should not be used for obsessive-compulsive disorder and posttraumatic stress disorder (Box 1).
Current evidence indicates little support for using benzodiazepines for obsessive-compulsive disorder (OCD). The American Psychiatric Association (APA) and the World Federation of Biological Psychiatry do not recommend benzodiazepines for treating OCD because of a lack of evidence for efficacy.a,b An earlier study suggested clonazepam monotherapy was effective for OCDc; however, a more recent study did not show a benefit on rate of response or degree of symptom improvement.d Augmentation strategies with benzodiazepines also do not appear to be beneficial for OCD management. A recent double-blind, placebo-controlled study failed to demonstrate faster symptom improvement by augmenting sertraline with clonazepam, although the study had a small sample size and high drop-out rate.e
Because benzodiazepines have negligible action on core posttraumatic stress disorder (PTSD) symptoms (re-experiencing, avoidance, and hyperarousal), selective serotonin reuptake inhibitors and other agents largely have supplanted them for PTSD treatment.f Use of benzodiazepines for PTSD is associated with withdrawal symptoms, more severe symptoms after discontinuation, and possible disinhibition, and may interfere with patients’ efforts to integrate trauma experiences. Although benzodiazepines may reduce distress associated with acute trauma, there is evidence—in clinical studies and animal models—that early benzodiazepine administration fails to prevent PTSD and may increase its incidence.g The International Consensus Group on Depression and Anxiety, the APA, and the British Association for Psychopharmacology all highlight the limited role, if any, for benzodiazepines in PTSD.h-j
References
- Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
- American Psychiatric Association. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
- Hewlett WA, Vinogradov S, Agras WS. Clomipramine, clonazepam, and clonidine treatment of obsessive compulsive disorder. J Clin Psychopharmacol. 1992;12(6):420-430.
- Hollander E, Kaplan A, Stahl SM. A double-blind, placebo-controlled trial of clonazepam in obsessive-compulsive disorder. World J Biol Psychiatry. 2003;4(1):30-34.
- Crockett BA, Churchill E, Davidson JR. A double-blind combination study of clonazepam and sertraline in OCD. Ann Clin Psychiatry. 2004;16(3):127-132.
- Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs. Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
- Matar MA, Zohar J, Kaplan Z, et al. Alprazolam treatment immediately after stress exposure interferes with the normal HPA-stress response and increases vulnerability to subsequent stress in an animal model of PTSD. Eur Neuropsychopharmacol. 2009;19(4):283-295.
- Ballenger JC, Davidson JR, Lecrubier Y, et al. Consensus statement update on posttraumatic stress disorder from the international consensus group on depression and anxiety. J Clin Psychiatry. 2004;65(suppl 1):55-62.
- Ursano RJ, Bell C, Eth S, et al. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Am J Psychiatry. 2004;161(11 suppl):3-31.
- Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based guidelines for the pharmacological treatment of anxiety disorders: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2005;19(6):567-596.
Pharmacokinetic properties
Most benzodiazepines are considered to have similar efficacy; therefore, selection is based on pharmacokinetic considerations. Table 1 compares the indication, onset, and half-life of 12 commonly used benzodiazepines.2-6 Although Table 1 lists approximate equivalent doses, studies report inconsistent data. These are approximations only and should not be used independently to make therapy decisions.
Table 1
Oral benzodiazepines: Indications, onset, half-life, and equivalent doses
| Drug | FDA-approved indication(s) | Onset of action | Approximate half-life (hours) in healthy adults | Approximate equivalent dose (mg)a | Comments |
|---|---|---|---|---|---|
| Alprazolam | Anxiety disorders, panic disorder | Intermediate | 6.3 to 26.9 (IR), 10.7 to 15.8 (XR) | 0.5 | Increased risk for abuse because of greater lipid solubility |
| Chlordiazepoxide | Anxiety disorders, acute alcohol withdrawal, preoperative apprehension and anxiety | Intermediate | 24 to 48 | 10 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Clonazepam | Seizure disorders, panic disorder | Intermediate | 18 to 50 | 0.25 to 0.5 | Use caution in patients with liver disease |
| Clorazepate | Anxiety, seizures, acute alcohol withdrawal | Fast | 40 to 50 | 7.5 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Diazepam | Anxiety disorders, acute alcohol withdrawal, muscle spasms, convulsive disorders | Very fast | 20 to 100 | 5 | Risk for accumulation because of long-acting metabolites (temazepam, desmethyldiazepam, oxazepam). Increased risk for abuse because of quick onset |
| Estazolam | Insomnia | Intermediate | 10 to 24 | 0.3 to 2 | None |
| Flurazepam | Insomnia | Intermediate | 47 to 100 | 30 | Avoid in geriatric patients or patients with liver impairment |
| Lorazepam | Anxiety | Intermediate | 10 to 20 | 1 | Preferred for patients with liver impairment and geriatric patients |
| Oxazepam | Anxiety, acute alcohol withdrawal | Slow to intermediate | 5 to 20 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Quazepam | Insomnia | Intermediate | 39 to 73 | 5 to 15 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Temazepam | Insomnia | Intermediate | 3.5 to 18.4 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Triazolam | Insomnia | Fast | 1.5 to 5.5 | 0.25 | Lacks active metabolites |
| IR: immediate release; XR: extended release aInterpret with caution, conflicting data exist Source: References 2-6 | |||||
A diverse range of indications
Alcohol withdrawal. Benzodiazepines are the treatment of choice for alcohol withdrawal syndrome, particularly to prevent seizures.7 Research supports symptom-triggered therapy using the revised Clinical Institute Withdrawal Assessment for Alcohol. Benzodiazepines reduce CNS sympathetic hyperactivity to mitigate withdrawal from alcohol by decreasing tachycardia, tremor, insomnia, agitation, and anxiety. Furthermore, these agents provide prophylaxis against serious sequelae such as seizures and delirium.
Insomnia. The American Academy of Sleep Medicine considers benzodiazepine receptor agonists (BzRAs, which include benzodiazepines and non-benzodiazepines) and ramelteon first-line pharmacotherapy for primary insomnia.8 However, pharmacologic treatment should be short-term. Agents with short to intermediate half-lives and rapid onset, such as triazolam, can aid sleep initiation. Those with longer half-lives, such as temazepam, could address sleep maintenance. If a patient does not respond to the initial agent, try another medication within the same class, because patients may respond differently. Use lower starting doses in geriatric patients.9 Closely monitor for adverse effects, rebound insomnia, and potential abuse or tolerance. Identify comorbid conditions and medications that may impair sleep, and address them accordingly.
Psychological and behavioral treatments given over 4 to 8 weeks can yield stable sleep improvements for up to 2 years. If available, these interventions may be considered first-line for treating insomnia because of their lasting effects compared with BzRAs.10
Generalized anxiety disorder (GAD). Benzodiazepines effectively treat GAD because they work quickly and are well tolerated. However, there are better first-line treatment options when considering efficacy studies and dependence and tolerance concerns. One effect-size comparison of 21 double-blind, placebo-controlled trials showed that the efficacy of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and pregabalin are comparable to benzodiazepines.11 Benzodiazepines can be used in the first 2 to 3 weeks after initiating antidepressants to alleviate and prevent worsening of anxiety that may occur at the start of antidepressant therapy. Recent treatment guidelines recommend benzodiazepines as a second-line treatment or for treatment-resistant GAD in patients who do not have a substance abuse history.12,13
Panic disorder. Efficacy of benzodiazepines for panic disorder is comparable to SSRIs, SNRIs, and tricyclic antidepressants (TCAs). SSRIs and SNRIs are considered first-line treatments for panic disorder because of their favorable side effect profile.14 In practice, benzodiazepines often are combined with SSRIs, SNRIs, or TCAs. A randomized controlled trial demonstrated that paroxetine and clonazepam (mean dose 1.6 mg/d at 5 weeks) resulted in a more rapid response compared with paroxetine alone, although this difference lasted only a few weeks.15 Furthermore, this study suggested that brief treatment with clonazepam followed by a taper is as effective as sustained treatment with paroxetine and clonazepam.15
There is a lack of high-quality data on combining cognitive-behavioral therapy (CBT) and benzodiazepines for panic disorder, although a Cochrane Review found that adding a benzodiazepine to CBT did not lead to a significant difference in response compared with psychotherapy alone.16 A recent randomized controlled trial demonstrated that tapering benzodiazepines combined with CBT was associated with successful discontinuation of the drug and prevented return of panic symptoms.17
Social anxiety. A meta-analysis found that for treating social anxiety, benzodiazepines have better efficacy than SSRIs, monoamine oxidase inhibitors, and anticonvulsants.18 Longer-acting benzodiazepines may be more effective than shorter-acting agents. One study of patients with social anxiety showed a 38% response rate for alprazolam vs 20% for placebo over 12 weeks, and a similar 10-week study demonstrated a 73% recovery rate with clonazepam vs 22% for placebo.19 In addition, studies have observed that patients can be maintained on clonazepam for up to 2 years without symptom relapse and will tolerate slow-taper discontinuation.18,20 Sedation and drowsiness can be lessened by limiting clonazepam doses to 2 to 3 mg/d.
Akathisia and tremor. Akathisia, a syndrome of motor restlessness and inner turmoil, is associated with antipsychotics but can occur with SSRIs. Reducing the dosage or switching to another, usually less potent agent often can relieve akathisia. When these remedies are not tenable, consider benzodiazepines along with other medications—including beta blockers and anticholinergic agents—with demonstrated efficacy in reducing akathisia symptoms. Lorazepam, diazepam, and clonazepam have demonstrated efficacy for relieving akathisia in comparison studies with placebo, propranolol, and diphenhydramine.21,22
Drug-induced postural tremor can occur with several psychotropics, including lithium, valproic acid, antidepressants, and antipsychotics. A tremor is considered mild if a patient can drink a glass of water with 1 hand without spilling and severe if holding a glass with 2 hands is difficult. Propranolol is most commonly prescribed for these tremors, but alprazolam and clonazepam have demonstrated efficacy, either as monotherapy or coadministered with a beta blocker.23
Acute agitation. Agitated patients often have acute psychosis and/or mania or dyscontrol secondary to axis II disorders.24 Patients may be paranoid, hostile, disruptive, and combative. Rapidly initiating medication can prevent the need for more restrictive measures, such as seclusion or restraint. Antipsychotics—especially high-potency agents such as haloperidol—and benzodiazepines, as monotherapy or in combination, are a mainstay treatment. Although treatment protocols favor atypical antipsychotics over typical antipsychotics, benzodiazepines are a viable option because of their anxiolytic and sedative effects. Advantages of benzodiazepine monotherapy include decreased extrapyramidal symptoms, greater patient acceptance/preference, and increased sedation compared with antipsychotics. Lorazepam, 1 to 2 mg intramuscularly (IM) or orally, is well tolerated because of its favorable drug-drug interaction profile and lack of significant cardiac side effects. Benzodiazepines can cause respiratory depression in patients with chronic lung disease and additive sedation secondary to opiates, other sedatives/hypnotics, or alcohol. Behavioral disinhibition is rare and is associated with preexisting CNS pathology or mental retardation.25 The IM olanzapine package insert warns against coadministering IM lorazepam because of additive cardiorespiratory depressive effects and excessive somnolence.26
Catatonia. The characteristic symptoms of catatonia are immobility, negativism, muteness, and failure to eat or drink. Benzodiazepines improve these symptoms in approximately 70% to 80% of catatonic patients with affective disorders. Response rates are lower in catatonia in patients with schizophrenia.27 If catatonia in a patient with psychosis is missed, giving antipsychotics before benzodiazepines may worsen catatonic symptoms or precipitate neuroleptic malignant syndrome in some cases. When you suspect a patient has catatonia, start with lorazepam, 1 to 2 mg IV or IM, and examine the patient for diminishing catatonic signs within 1 to 2 hours. If catatonia signs lessen, begin regularly scheduled lorazepam, with dosing varying by age—be more cautious in geriatric patients—and symptom severity. Titrate benzodiazepines for stuporous patients more slowly (eg, 1 mg 3 times a day as a starting dose) than for excited catatonic patients. Lorazepam can be increased gradually as tolerated; it is not unusual for patients to require up to 8 to 12 mg/d. Electroconvulsive therapy (ECT) is the treatment of choice when catatonic patients respond poorly or partially to high-dose benzodiazepines.28,29
Benzodiazepine reversal for ECT
Benzodiazepines have anticonvulsant properties that may interfere with the therapeutic efficacy of ECT.30 A multi-center study demonstrated that lorazepam (up to 4 mg/d as needed) in the 48 hours before the first ECT session was not associated with effects on seizure threshold or duration; however, larger lorazepam dosages were associated with briefer EEG seizure duration.31 Some patients may not tolerate withholding or tapering benzodiazepines in preparation for ECT. Studies investigating flumazenil for pre-ECT benzodiazepine reversal are lacking. One retrospective analysis showed that flumazenil administration immediately before and after ECT resulted in adequate seizures with no difference in clinical outcome compared with patients who were not receiving benzodiazepines or flumazenil.32
Tapering benzodiazepines
Slow discontinuation of benzodiazepines is recommended to avoid withdrawal symptoms, such as rebound anxiety, agitation, insomnia, or seizures, particularly when use exceeds 8 weeks. The onset of withdrawal symptoms varies, depending on the medication used. Withdrawal symptoms may appear in 1 to 2 days for agents with shorter half-lives, but may not appear until 3 to 7 days for agents with longer half-lives.33Table 2 lists recommended durations for tapering benzodiazepines.33,34 In general, decrease the total daily dose by 25% the first week, another 25% the second week, then 10% a week until discontinuation. When benzodiazepine use exceeds 1 year, a slower taper is recommended; for example, decrease 10% every 1 to 2 weeks. When 20% of the dosage remains, begin a 5% dose reduction every 2 to 4 weeks. Monitor patients for withdrawal symptoms or symptom exacerbation. If either occur, consider maintaining the current benzodiazepine dose or increasing the dose for 1 to 2 weeks or longer, if necessary, then continue to taper at a slower rate.34
Table 2
Recommendations for tapering benzodiazepines
| Duration of use | Recommended taper length | Comments |
| <6 to 8 weeks | Taper may not be required | Depending on clinical judgment and patient stability/preference, consider implementing a taper, particularly if using a high-dose benzodiazepine or an agent with a short or intermediate half-life, such as alprazolam or triazolam |
| 8 weeks to 6 months | Slowly over 2 to 3 weeks | Go slower during latter half of taper. Tapering will reduce, not eliminate, withdrawal symptoms. Patients should avoid alcohol and stimulants during benzodiazepine withdrawal |
| 6 months to 1 year | Slowly over 4 to 8 weeks | |
| >1 year | Slowly over 2 to 4 months | |
| Source: References 33,34 | ||
Risks of benzodiazepine use
For most indications, benzodiazepine therapy should be short-term.35 Use exceeding 2 to 4 weeks increases the risk for dependence and withdrawal. Tell patients to avoid alcohol while taking a benzodiazepine because this combination is potentially lethal. Benzodiazepines are commonly abused and abuse can lead to unintentional drug overdose. Benzodiazepines accounted for 37% of unintentional drug overdose deaths in West Virginia in 2006; in 46% of these cases, benzodiazepines were used for nonmedical purposes. Clinicians can help reduce the risk of diversion by limiting prescriptions to 30 days with no refills.36
Older patients taking benzodiazepines are at increased risk of falls and hip fractures.37 Lorazepam, oxazepam, and temazepam—agents with shorter half-lives that are not greatly affected by pharmacokinetic changes associated with aging—are preferred for these patients.34 Patients with dementia or other CNS-compromising conditions may become confused or delirious with regular benzodiazepine dosing. Educate patients to whom you prescribe benzodiazepines about the importance of gauging their level of sedation before driving or engaging in other tasks for which sedation could compromise their safety. Benzodiazepine use during pregnancy requires a careful discussion of risks and benefits (Box 2).38
Benzodiazepine use during pregnancy has been associated with cleft palate and urogenital and neurologic malformations in the fetus.38 Although data are conflicting—particularly among recent meta-analyses that fail to demonstrate an association—some experts advise against benzodiazepine use in the first trimester. Participate in shared decision making with your patients and educate them about the potential risks and benefits of benzodiazepine use during the first trimester and throughout pregnancy. After delivery, newborns may develop “floppy baby syndrome”—which is associated with lethargy, difficulty eating, and respiratory depression—or withdrawal.38 To minimize this risk, consider tapering the benzodiazepine as the patient approaches delivery.
Related Resources
- Substance Abuse and Mental Health Services Administration. www.samhsa.gov.
- National Institute on Drug Abuse resources for medical and health professionals. www.drugabuse.gov/medical-health-professionals.
- American Academy of Sleep Medicine. www.aasmnet.org.
Drug Brand Names
- Alprazolam • Xanax
- Chlordiazepoxide • Librium, Limbitrol
- Clonazepam • Klonopin
- Clorazepate • Tranxene
- Diazepam • Valium
- Diphenhydramine • Benadryl, others
- Estazolam • ProSom
- Flumazenil • Romazicon
- Flurazepam • Dalmane
- Haloperidol • Haldol
- Lithium • Lithobid
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Paroxetine • Paxil
- Pregabalin • Lyrica
- Propranolol • Inderal, InnoPran XL, others
- Quazepam • Doral
- Ramelteon • Rozerem
- Sertraline • Zoloft
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakene, Stavzor, others
Disclosures
Drs. Bostwick and Yasugi report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Casher is a speaker for AstraZeneca and Sunovion Pharmaceuticals.
Since the discovery of chlordiazepoxide in the 1950s, benzodiazepines have revolutionized the treatment of anxiety and insomnia, largely because of their improved safety profile compared with barbiturates, formerly the preferred sedative-hypnotic.1 In addition to their anxiolytic and sedative-hypnotic effects, benzodiazepines exhibit anterograde amnesia, anticonvulsant, and muscle relaxant properties.1 Psychiatrists use benzodiazepines to treat anxiety and sleep disorders, acute agitation, alcohol withdrawal, catatonia, and psychotropic side effects such as akathisia. This article highlights the evidence for using benzodiazepines in anxiety and other disorders and why they generally should not be used for obsessive-compulsive disorder and posttraumatic stress disorder (Box 1).
Current evidence indicates little support for using benzodiazepines for obsessive-compulsive disorder (OCD). The American Psychiatric Association (APA) and the World Federation of Biological Psychiatry do not recommend benzodiazepines for treating OCD because of a lack of evidence for efficacy.a,b An earlier study suggested clonazepam monotherapy was effective for OCDc; however, a more recent study did not show a benefit on rate of response or degree of symptom improvement.d Augmentation strategies with benzodiazepines also do not appear to be beneficial for OCD management. A recent double-blind, placebo-controlled study failed to demonstrate faster symptom improvement by augmenting sertraline with clonazepam, although the study had a small sample size and high drop-out rate.e
Because benzodiazepines have negligible action on core posttraumatic stress disorder (PTSD) symptoms (re-experiencing, avoidance, and hyperarousal), selective serotonin reuptake inhibitors and other agents largely have supplanted them for PTSD treatment.f Use of benzodiazepines for PTSD is associated with withdrawal symptoms, more severe symptoms after discontinuation, and possible disinhibition, and may interfere with patients’ efforts to integrate trauma experiences. Although benzodiazepines may reduce distress associated with acute trauma, there is evidence—in clinical studies and animal models—that early benzodiazepine administration fails to prevent PTSD and may increase its incidence.g The International Consensus Group on Depression and Anxiety, the APA, and the British Association for Psychopharmacology all highlight the limited role, if any, for benzodiazepines in PTSD.h-j
References
- Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
- American Psychiatric Association. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
- Hewlett WA, Vinogradov S, Agras WS. Clomipramine, clonazepam, and clonidine treatment of obsessive compulsive disorder. J Clin Psychopharmacol. 1992;12(6):420-430.
- Hollander E, Kaplan A, Stahl SM. A double-blind, placebo-controlled trial of clonazepam in obsessive-compulsive disorder. World J Biol Psychiatry. 2003;4(1):30-34.
- Crockett BA, Churchill E, Davidson JR. A double-blind combination study of clonazepam and sertraline in OCD. Ann Clin Psychiatry. 2004;16(3):127-132.
- Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs. Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
- Matar MA, Zohar J, Kaplan Z, et al. Alprazolam treatment immediately after stress exposure interferes with the normal HPA-stress response and increases vulnerability to subsequent stress in an animal model of PTSD. Eur Neuropsychopharmacol. 2009;19(4):283-295.
- Ballenger JC, Davidson JR, Lecrubier Y, et al. Consensus statement update on posttraumatic stress disorder from the international consensus group on depression and anxiety. J Clin Psychiatry. 2004;65(suppl 1):55-62.
- Ursano RJ, Bell C, Eth S, et al. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Am J Psychiatry. 2004;161(11 suppl):3-31.
- Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based guidelines for the pharmacological treatment of anxiety disorders: recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2005;19(6):567-596.
Pharmacokinetic properties
Most benzodiazepines are considered to have similar efficacy; therefore, selection is based on pharmacokinetic considerations. Table 1 compares the indication, onset, and half-life of 12 commonly used benzodiazepines.2-6 Although Table 1 lists approximate equivalent doses, studies report inconsistent data. These are approximations only and should not be used independently to make therapy decisions.
Table 1
Oral benzodiazepines: Indications, onset, half-life, and equivalent doses
| Drug | FDA-approved indication(s) | Onset of action | Approximate half-life (hours) in healthy adults | Approximate equivalent dose (mg)a | Comments |
|---|---|---|---|---|---|
| Alprazolam | Anxiety disorders, panic disorder | Intermediate | 6.3 to 26.9 (IR), 10.7 to 15.8 (XR) | 0.5 | Increased risk for abuse because of greater lipid solubility |
| Chlordiazepoxide | Anxiety disorders, acute alcohol withdrawal, preoperative apprehension and anxiety | Intermediate | 24 to 48 | 10 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Clonazepam | Seizure disorders, panic disorder | Intermediate | 18 to 50 | 0.25 to 0.5 | Use caution in patients with liver disease |
| Clorazepate | Anxiety, seizures, acute alcohol withdrawal | Fast | 40 to 50 | 7.5 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Diazepam | Anxiety disorders, acute alcohol withdrawal, muscle spasms, convulsive disorders | Very fast | 20 to 100 | 5 | Risk for accumulation because of long-acting metabolites (temazepam, desmethyldiazepam, oxazepam). Increased risk for abuse because of quick onset |
| Estazolam | Insomnia | Intermediate | 10 to 24 | 0.3 to 2 | None |
| Flurazepam | Insomnia | Intermediate | 47 to 100 | 30 | Avoid in geriatric patients or patients with liver impairment |
| Lorazepam | Anxiety | Intermediate | 10 to 20 | 1 | Preferred for patients with liver impairment and geriatric patients |
| Oxazepam | Anxiety, acute alcohol withdrawal | Slow to intermediate | 5 to 20 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Quazepam | Insomnia | Intermediate | 39 to 73 | 5 to 15 | Risk for accumulation because of long-acting metabolites (desmethyldiazepam, oxazepam) |
| Temazepam | Insomnia | Intermediate | 3.5 to 18.4 | 30 | Preferred for patients with liver impairment and geriatric patients |
| Triazolam | Insomnia | Fast | 1.5 to 5.5 | 0.25 | Lacks active metabolites |
| IR: immediate release; XR: extended release aInterpret with caution, conflicting data exist Source: References 2-6 | |||||
A diverse range of indications
Alcohol withdrawal. Benzodiazepines are the treatment of choice for alcohol withdrawal syndrome, particularly to prevent seizures.7 Research supports symptom-triggered therapy using the revised Clinical Institute Withdrawal Assessment for Alcohol. Benzodiazepines reduce CNS sympathetic hyperactivity to mitigate withdrawal from alcohol by decreasing tachycardia, tremor, insomnia, agitation, and anxiety. Furthermore, these agents provide prophylaxis against serious sequelae such as seizures and delirium.
Insomnia. The American Academy of Sleep Medicine considers benzodiazepine receptor agonists (BzRAs, which include benzodiazepines and non-benzodiazepines) and ramelteon first-line pharmacotherapy for primary insomnia.8 However, pharmacologic treatment should be short-term. Agents with short to intermediate half-lives and rapid onset, such as triazolam, can aid sleep initiation. Those with longer half-lives, such as temazepam, could address sleep maintenance. If a patient does not respond to the initial agent, try another medication within the same class, because patients may respond differently. Use lower starting doses in geriatric patients.9 Closely monitor for adverse effects, rebound insomnia, and potential abuse or tolerance. Identify comorbid conditions and medications that may impair sleep, and address them accordingly.
Psychological and behavioral treatments given over 4 to 8 weeks can yield stable sleep improvements for up to 2 years. If available, these interventions may be considered first-line for treating insomnia because of their lasting effects compared with BzRAs.10
Generalized anxiety disorder (GAD). Benzodiazepines effectively treat GAD because they work quickly and are well tolerated. However, there are better first-line treatment options when considering efficacy studies and dependence and tolerance concerns. One effect-size comparison of 21 double-blind, placebo-controlled trials showed that the efficacy of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and pregabalin are comparable to benzodiazepines.11 Benzodiazepines can be used in the first 2 to 3 weeks after initiating antidepressants to alleviate and prevent worsening of anxiety that may occur at the start of antidepressant therapy. Recent treatment guidelines recommend benzodiazepines as a second-line treatment or for treatment-resistant GAD in patients who do not have a substance abuse history.12,13
Panic disorder. Efficacy of benzodiazepines for panic disorder is comparable to SSRIs, SNRIs, and tricyclic antidepressants (TCAs). SSRIs and SNRIs are considered first-line treatments for panic disorder because of their favorable side effect profile.14 In practice, benzodiazepines often are combined with SSRIs, SNRIs, or TCAs. A randomized controlled trial demonstrated that paroxetine and clonazepam (mean dose 1.6 mg/d at 5 weeks) resulted in a more rapid response compared with paroxetine alone, although this difference lasted only a few weeks.15 Furthermore, this study suggested that brief treatment with clonazepam followed by a taper is as effective as sustained treatment with paroxetine and clonazepam.15
There is a lack of high-quality data on combining cognitive-behavioral therapy (CBT) and benzodiazepines for panic disorder, although a Cochrane Review found that adding a benzodiazepine to CBT did not lead to a significant difference in response compared with psychotherapy alone.16 A recent randomized controlled trial demonstrated that tapering benzodiazepines combined with CBT was associated with successful discontinuation of the drug and prevented return of panic symptoms.17
Social anxiety. A meta-analysis found that for treating social anxiety, benzodiazepines have better efficacy than SSRIs, monoamine oxidase inhibitors, and anticonvulsants.18 Longer-acting benzodiazepines may be more effective than shorter-acting agents. One study of patients with social anxiety showed a 38% response rate for alprazolam vs 20% for placebo over 12 weeks, and a similar 10-week study demonstrated a 73% recovery rate with clonazepam vs 22% for placebo.19 In addition, studies have observed that patients can be maintained on clonazepam for up to 2 years without symptom relapse and will tolerate slow-taper discontinuation.18,20 Sedation and drowsiness can be lessened by limiting clonazepam doses to 2 to 3 mg/d.
Akathisia and tremor. Akathisia, a syndrome of motor restlessness and inner turmoil, is associated with antipsychotics but can occur with SSRIs. Reducing the dosage or switching to another, usually less potent agent often can relieve akathisia. When these remedies are not tenable, consider benzodiazepines along with other medications—including beta blockers and anticholinergic agents—with demonstrated efficacy in reducing akathisia symptoms. Lorazepam, diazepam, and clonazepam have demonstrated efficacy for relieving akathisia in comparison studies with placebo, propranolol, and diphenhydramine.21,22
Drug-induced postural tremor can occur with several psychotropics, including lithium, valproic acid, antidepressants, and antipsychotics. A tremor is considered mild if a patient can drink a glass of water with 1 hand without spilling and severe if holding a glass with 2 hands is difficult. Propranolol is most commonly prescribed for these tremors, but alprazolam and clonazepam have demonstrated efficacy, either as monotherapy or coadministered with a beta blocker.23
Acute agitation. Agitated patients often have acute psychosis and/or mania or dyscontrol secondary to axis II disorders.24 Patients may be paranoid, hostile, disruptive, and combative. Rapidly initiating medication can prevent the need for more restrictive measures, such as seclusion or restraint. Antipsychotics—especially high-potency agents such as haloperidol—and benzodiazepines, as monotherapy or in combination, are a mainstay treatment. Although treatment protocols favor atypical antipsychotics over typical antipsychotics, benzodiazepines are a viable option because of their anxiolytic and sedative effects. Advantages of benzodiazepine monotherapy include decreased extrapyramidal symptoms, greater patient acceptance/preference, and increased sedation compared with antipsychotics. Lorazepam, 1 to 2 mg intramuscularly (IM) or orally, is well tolerated because of its favorable drug-drug interaction profile and lack of significant cardiac side effects. Benzodiazepines can cause respiratory depression in patients with chronic lung disease and additive sedation secondary to opiates, other sedatives/hypnotics, or alcohol. Behavioral disinhibition is rare and is associated with preexisting CNS pathology or mental retardation.25 The IM olanzapine package insert warns against coadministering IM lorazepam because of additive cardiorespiratory depressive effects and excessive somnolence.26
Catatonia. The characteristic symptoms of catatonia are immobility, negativism, muteness, and failure to eat or drink. Benzodiazepines improve these symptoms in approximately 70% to 80% of catatonic patients with affective disorders. Response rates are lower in catatonia in patients with schizophrenia.27 If catatonia in a patient with psychosis is missed, giving antipsychotics before benzodiazepines may worsen catatonic symptoms or precipitate neuroleptic malignant syndrome in some cases. When you suspect a patient has catatonia, start with lorazepam, 1 to 2 mg IV or IM, and examine the patient for diminishing catatonic signs within 1 to 2 hours. If catatonia signs lessen, begin regularly scheduled lorazepam, with dosing varying by age—be more cautious in geriatric patients—and symptom severity. Titrate benzodiazepines for stuporous patients more slowly (eg, 1 mg 3 times a day as a starting dose) than for excited catatonic patients. Lorazepam can be increased gradually as tolerated; it is not unusual for patients to require up to 8 to 12 mg/d. Electroconvulsive therapy (ECT) is the treatment of choice when catatonic patients respond poorly or partially to high-dose benzodiazepines.28,29
Benzodiazepine reversal for ECT
Benzodiazepines have anticonvulsant properties that may interfere with the therapeutic efficacy of ECT.30 A multi-center study demonstrated that lorazepam (up to 4 mg/d as needed) in the 48 hours before the first ECT session was not associated with effects on seizure threshold or duration; however, larger lorazepam dosages were associated with briefer EEG seizure duration.31 Some patients may not tolerate withholding or tapering benzodiazepines in preparation for ECT. Studies investigating flumazenil for pre-ECT benzodiazepine reversal are lacking. One retrospective analysis showed that flumazenil administration immediately before and after ECT resulted in adequate seizures with no difference in clinical outcome compared with patients who were not receiving benzodiazepines or flumazenil.32
Tapering benzodiazepines
Slow discontinuation of benzodiazepines is recommended to avoid withdrawal symptoms, such as rebound anxiety, agitation, insomnia, or seizures, particularly when use exceeds 8 weeks. The onset of withdrawal symptoms varies, depending on the medication used. Withdrawal symptoms may appear in 1 to 2 days for agents with shorter half-lives, but may not appear until 3 to 7 days for agents with longer half-lives.33Table 2 lists recommended durations for tapering benzodiazepines.33,34 In general, decrease the total daily dose by 25% the first week, another 25% the second week, then 10% a week until discontinuation. When benzodiazepine use exceeds 1 year, a slower taper is recommended; for example, decrease 10% every 1 to 2 weeks. When 20% of the dosage remains, begin a 5% dose reduction every 2 to 4 weeks. Monitor patients for withdrawal symptoms or symptom exacerbation. If either occur, consider maintaining the current benzodiazepine dose or increasing the dose for 1 to 2 weeks or longer, if necessary, then continue to taper at a slower rate.34
Table 2
Recommendations for tapering benzodiazepines
| Duration of use | Recommended taper length | Comments |
| <6 to 8 weeks | Taper may not be required | Depending on clinical judgment and patient stability/preference, consider implementing a taper, particularly if using a high-dose benzodiazepine or an agent with a short or intermediate half-life, such as alprazolam or triazolam |
| 8 weeks to 6 months | Slowly over 2 to 3 weeks | Go slower during latter half of taper. Tapering will reduce, not eliminate, withdrawal symptoms. Patients should avoid alcohol and stimulants during benzodiazepine withdrawal |
| 6 months to 1 year | Slowly over 4 to 8 weeks | |
| >1 year | Slowly over 2 to 4 months | |
| Source: References 33,34 | ||
Risks of benzodiazepine use
For most indications, benzodiazepine therapy should be short-term.35 Use exceeding 2 to 4 weeks increases the risk for dependence and withdrawal. Tell patients to avoid alcohol while taking a benzodiazepine because this combination is potentially lethal. Benzodiazepines are commonly abused and abuse can lead to unintentional drug overdose. Benzodiazepines accounted for 37% of unintentional drug overdose deaths in West Virginia in 2006; in 46% of these cases, benzodiazepines were used for nonmedical purposes. Clinicians can help reduce the risk of diversion by limiting prescriptions to 30 days with no refills.36
Older patients taking benzodiazepines are at increased risk of falls and hip fractures.37 Lorazepam, oxazepam, and temazepam—agents with shorter half-lives that are not greatly affected by pharmacokinetic changes associated with aging—are preferred for these patients.34 Patients with dementia or other CNS-compromising conditions may become confused or delirious with regular benzodiazepine dosing. Educate patients to whom you prescribe benzodiazepines about the importance of gauging their level of sedation before driving or engaging in other tasks for which sedation could compromise their safety. Benzodiazepine use during pregnancy requires a careful discussion of risks and benefits (Box 2).38
Benzodiazepine use during pregnancy has been associated with cleft palate and urogenital and neurologic malformations in the fetus.38 Although data are conflicting—particularly among recent meta-analyses that fail to demonstrate an association—some experts advise against benzodiazepine use in the first trimester. Participate in shared decision making with your patients and educate them about the potential risks and benefits of benzodiazepine use during the first trimester and throughout pregnancy. After delivery, newborns may develop “floppy baby syndrome”—which is associated with lethargy, difficulty eating, and respiratory depression—or withdrawal.38 To minimize this risk, consider tapering the benzodiazepine as the patient approaches delivery.
Related Resources
- Substance Abuse and Mental Health Services Administration. www.samhsa.gov.
- National Institute on Drug Abuse resources for medical and health professionals. www.drugabuse.gov/medical-health-professionals.
- American Academy of Sleep Medicine. www.aasmnet.org.
Drug Brand Names
- Alprazolam • Xanax
- Chlordiazepoxide • Librium, Limbitrol
- Clonazepam • Klonopin
- Clorazepate • Tranxene
- Diazepam • Valium
- Diphenhydramine • Benadryl, others
- Estazolam • ProSom
- Flumazenil • Romazicon
- Flurazepam • Dalmane
- Haloperidol • Haldol
- Lithium • Lithobid
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Oxazepam • Serax
- Paroxetine • Paxil
- Pregabalin • Lyrica
- Propranolol • Inderal, InnoPran XL, others
- Quazepam • Doral
- Ramelteon • Rozerem
- Sertraline • Zoloft
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakene, Stavzor, others
Disclosures
Drs. Bostwick and Yasugi report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Casher is a speaker for AstraZeneca and Sunovion Pharmaceuticals.
1. Mihic SJ, Harris RA. Hypnotics and sedatives. In: Brunton LL Chabner BA, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. New York, NY: McGraw Hill and Company; 2011:457-480.
2. Facts and comparisons Web site. 2011 Wolters Kluwer Health Inc. http://online.factsandcomparisons.com. Accessed August 16, 2011.
3. DuPont RL, Greene W, Lydiard RB. Sedatives and hypnotics: pharmacology and epidemiology. In: Gold MS Hermann R, eds. UpToDate. http://www.uptodate.com/contents/sedatives-and-hypnotics-abuse-and-dependence-pharmacology-and-epidemiology. Accessed August 16, 2011.
4. U.S. Food and Drug Administration. Orange book: approved drug products with therapeutic equivalence evaluations. http://www.accessdata.fda.gov/scripts/cder/ob/default.cfm. Accessed August 16, 2011.
5. Chouinard G. Issues in the clinical use of benzodiazepines: potency withdrawal, and rebound. J Clin Psychiatry. 2004;65(suppl 5):7-12.
6. Shader RI, Greenblatt DJ. Can you provide a table of equivalencies for benzodiazepines and other marketed benzodiazepine receptor agonists? J Clin Psychopharmacol. 1997;17(4):331.-
7. Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacologic interventions for the treatment of the alcohol withdrawal syndrome. Cochrane Database Syst Rev. 2011;15(6):CD008537.-
8. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4(5):487-504.
9. Foral P, Dewan N, Malesker M. Insomnia: a therapeutic review for pharmacists. Consult Pharm. 2011;26(5):332-341.
10. Riemann D, Perlis ML. The treatments of chronic insomnia: a review of benzodiazepine receptor agonists and psychological and behavioral therapies. Sleep Med Rev. 2009;13(3):205-214.
11. Hidalgo RB, Tupler LA, Davidson JR. An effect-size analysis of pharmacologic treatments of generalized anxiety disorder. J Psychopharmacol. 2007;21(8):864-872.
12. Davidson JR, Zhang W, Connor KM, et al. A psychopharmacological treatment algorithm for generalised anxiety disorder (GAD). J Psychopharmacol. 2010;24(1):3-26.
13. Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
14. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder. 2nd ed. Arlington VA: American Psychiatric Publishing, Inc.; 2009.
15. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
16. Watanabe N, Churchill R, Furukawa TA. Combined psychotherapy plus benzodiazepines for panic disorder. Cochrane Database Syst Rev. 2009;(1):CD005335.-
17. Otto MW, McHugh RK, Simon NM, et al. Efficacy of CBT for benzodiazepine discontinuation in patients with panic disorder: further evaluation. Behav Res Ther. 2010;48(8):720-727.
18. Davidson JR. Use of benzodiazepines in social anxiety disorder generalized anxiety disorder, and posttraumatic stress disorder. J Clin Psychiatry. 2004;65(suppl 5):29-33.
19. Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
20. Connor KM, Davidson JR, Potts NL, et al. Discontinuation of clonazepam in the treatment of social phobia. J Clin Psychopharmacol. 1998;18(5):373-378.
21. Miller CH, Fleischhacker WW. Managing antipsychotic-induced acute and chronic akathisia. Drug Saf. 2000;22(1):73-81.
22. Rodnitzky RL. Drug-induced movement disorders. Clin Neuropharmacol. 2002;25(3):142-151.
23. Arbaizar B, Gómez-Acebo I, Llorca J. Postural induced tremor in psychiatry. Psychiatry Clin Neurosci. 2008;62(6):638-645.
24. Casher MI, Bess JD. Manual of inpatient psychiatry. New York NY: Cambridge University Press; 2010.
25. Battaglia J. Pharmacological management of acute agitation. Drugs. 2005;65(9):1207-1222.
26. Physicians’ desk reference. Montvale NJ: PDR Network, LLC; 2010.
27. Rosebush PI, Mazurek MF. Catatonia and its treatment. Schizophr Bull. 2010;36(2):239-242.
28. Ungvari GS, Kau LS, Wai-Kwong T, et al. The pharmacological treatment of catatonia: an overview. Eur Arch Psychiatry Clin Neurosci. 2001;251(suppl 1):I31-I34.
29. Fink M, Taylor MA. Catatonia: a clinician’s guide to diagnosis and treatment. New York NY: Cambridge University Press; 2003.
30. Naguib N, Koorn R. Interactions between psychotropics anaesthetics and electroconvulsive therapy: implications for drug choice and patient management. CNS Drugs. 2002;16(4):229-247.
31. Boylan LS, Haskett RF, Mulsant BH, et al. Determinants of seizure threshold in ECT: benzodiazepine use, anesthetic dosage, and other factors. J ECT. 2000;16(1):3-18.
32. Krystal AD, Watts BV, Weiner RD, et al. The use of flumazenil in the anxious and benzodiazepine-dependent ECT patient. J ECT. 1998;14(1):5-14.
33. Melton ST, Kirkwood CK. Anxiety disorders I: generalized anxiety panic, and social anxiety disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: a pathophysiologic approach. New York, NY: McGraw-Hill Companies; 2011:1209-1228.
34. Benzodiazepine toolkit. The Pharmacist’s Letter/Prescriber’s Letter. 2011;27(4):270406.-
35. Lader M. Benzodiazepines revisited – will we ever learn? Addiction. 2011;106(12):2086-2109.
36. Toblin RL, Paulozzi LJ, Logan JE, et al. Mental illness and psychotropic drug use among prescription drug overdose deaths: a medical examiner chart review. J Clin Psychiatry. 2010;71(4):491-496.
37. Ashton H. The diagnosis and management of benzodiazepine dependence. Curr Opin Psychiatry. 2005;18(3):249-255.
38. Menon SJ. Psychotropic medication during pregnancy and lactation. Arch Gynecol Obstet. 2008;277(1):1-13.
1. Mihic SJ, Harris RA. Hypnotics and sedatives. In: Brunton LL Chabner BA, Knollmann BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. New York, NY: McGraw Hill and Company; 2011:457-480.
2. Facts and comparisons Web site. 2011 Wolters Kluwer Health Inc. http://online.factsandcomparisons.com. Accessed August 16, 2011.
3. DuPont RL, Greene W, Lydiard RB. Sedatives and hypnotics: pharmacology and epidemiology. In: Gold MS Hermann R, eds. UpToDate. http://www.uptodate.com/contents/sedatives-and-hypnotics-abuse-and-dependence-pharmacology-and-epidemiology. Accessed August 16, 2011.
4. U.S. Food and Drug Administration. Orange book: approved drug products with therapeutic equivalence evaluations. http://www.accessdata.fda.gov/scripts/cder/ob/default.cfm. Accessed August 16, 2011.
5. Chouinard G. Issues in the clinical use of benzodiazepines: potency withdrawal, and rebound. J Clin Psychiatry. 2004;65(suppl 5):7-12.
6. Shader RI, Greenblatt DJ. Can you provide a table of equivalencies for benzodiazepines and other marketed benzodiazepine receptor agonists? J Clin Psychopharmacol. 1997;17(4):331.-
7. Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacologic interventions for the treatment of the alcohol withdrawal syndrome. Cochrane Database Syst Rev. 2011;15(6):CD008537.-
8. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4(5):487-504.
9. Foral P, Dewan N, Malesker M. Insomnia: a therapeutic review for pharmacists. Consult Pharm. 2011;26(5):332-341.
10. Riemann D, Perlis ML. The treatments of chronic insomnia: a review of benzodiazepine receptor agonists and psychological and behavioral therapies. Sleep Med Rev. 2009;13(3):205-214.
11. Hidalgo RB, Tupler LA, Davidson JR. An effect-size analysis of pharmacologic treatments of generalized anxiety disorder. J Psychopharmacol. 2007;21(8):864-872.
12. Davidson JR, Zhang W, Connor KM, et al. A psychopharmacological treatment algorithm for generalised anxiety disorder (GAD). J Psychopharmacol. 2010;24(1):3-26.
13. Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision. World J Biol Psychiatry. 2008;9(4):248-312.
14. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder. 2nd ed. Arlington VA: American Psychiatric Publishing, Inc.; 2009.
15. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
16. Watanabe N, Churchill R, Furukawa TA. Combined psychotherapy plus benzodiazepines for panic disorder. Cochrane Database Syst Rev. 2009;(1):CD005335.-
17. Otto MW, McHugh RK, Simon NM, et al. Efficacy of CBT for benzodiazepine discontinuation in patients with panic disorder: further evaluation. Behav Res Ther. 2010;48(8):720-727.
18. Davidson JR. Use of benzodiazepines in social anxiety disorder generalized anxiety disorder, and posttraumatic stress disorder. J Clin Psychiatry. 2004;65(suppl 5):29-33.
19. Argyropoulos SV, Sandford JJ, Nutt DJ. The psychobiology of anxiolytic drugs Part 2: pharmacological treatments of anxiety. Pharmacol Ther. 2000;88(3):213-227.
20. Connor KM, Davidson JR, Potts NL, et al. Discontinuation of clonazepam in the treatment of social phobia. J Clin Psychopharmacol. 1998;18(5):373-378.
21. Miller CH, Fleischhacker WW. Managing antipsychotic-induced acute and chronic akathisia. Drug Saf. 2000;22(1):73-81.
22. Rodnitzky RL. Drug-induced movement disorders. Clin Neuropharmacol. 2002;25(3):142-151.
23. Arbaizar B, Gómez-Acebo I, Llorca J. Postural induced tremor in psychiatry. Psychiatry Clin Neurosci. 2008;62(6):638-645.
24. Casher MI, Bess JD. Manual of inpatient psychiatry. New York NY: Cambridge University Press; 2010.
25. Battaglia J. Pharmacological management of acute agitation. Drugs. 2005;65(9):1207-1222.
26. Physicians’ desk reference. Montvale NJ: PDR Network, LLC; 2010.
27. Rosebush PI, Mazurek MF. Catatonia and its treatment. Schizophr Bull. 2010;36(2):239-242.
28. Ungvari GS, Kau LS, Wai-Kwong T, et al. The pharmacological treatment of catatonia: an overview. Eur Arch Psychiatry Clin Neurosci. 2001;251(suppl 1):I31-I34.
29. Fink M, Taylor MA. Catatonia: a clinician’s guide to diagnosis and treatment. New York NY: Cambridge University Press; 2003.
30. Naguib N, Koorn R. Interactions between psychotropics anaesthetics and electroconvulsive therapy: implications for drug choice and patient management. CNS Drugs. 2002;16(4):229-247.
31. Boylan LS, Haskett RF, Mulsant BH, et al. Determinants of seizure threshold in ECT: benzodiazepine use, anesthetic dosage, and other factors. J ECT. 2000;16(1):3-18.
32. Krystal AD, Watts BV, Weiner RD, et al. The use of flumazenil in the anxious and benzodiazepine-dependent ECT patient. J ECT. 1998;14(1):5-14.
33. Melton ST, Kirkwood CK. Anxiety disorders I: generalized anxiety panic, and social anxiety disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: a pathophysiologic approach. New York, NY: McGraw-Hill Companies; 2011:1209-1228.
34. Benzodiazepine toolkit. The Pharmacist’s Letter/Prescriber’s Letter. 2011;27(4):270406.-
35. Lader M. Benzodiazepines revisited – will we ever learn? Addiction. 2011;106(12):2086-2109.
36. Toblin RL, Paulozzi LJ, Logan JE, et al. Mental illness and psychotropic drug use among prescription drug overdose deaths: a medical examiner chart review. J Clin Psychiatry. 2010;71(4):491-496.
37. Ashton H. The diagnosis and management of benzodiazepine dependence. Curr Opin Psychiatry. 2005;18(3):249-255.
38. Menon SJ. Psychotropic medication during pregnancy and lactation. Arch Gynecol Obstet. 2008;277(1):1-13.
Bedside visit comes too late . . . Unrecognized spinal infection leads to paralysis . . .
Bedside visit comes too late
A 22-YEAR-OLD MAN underwent a liver biopsy after being admitted to the hospital a week earlier with fever, chills, diarrhea, and general malaise. A number of specialists had seen him in the hospital because of abnormal laboratory studies, increasing fever, and a maculopapular rash over his trunk and face.
After the biopsy, the patient was dizzy and diaphoretic. His attending physician ordered hemoglobin and hematocrit levels, which were lower than earlier that day. Repeat testing showed a further decrease, prompting the physician to order 2 units of red blood cells.
Typing and cross-matching delayed the transfusion for several hours. Before it could be started, the patient was found unresponsive. When the attending physician came to the bedside, the patient had no palpable pulse. A code was called, but resuscitation efforts failed.
An autopsy found a small hole in the liver and 3500 mL of blood in the peritoneal cavity, as well as hepatitis with zonal and submassive necrosis, hemoperitoneum, and hypertrophy of the heart. An HIV test performed before the biopsy eventually came back positive.
PLAINTIFF’S CLAIM The attending physician and nurses were negligent in failing to respond to signs and symptoms of internal bleeding, including falling hematocrit and hemoglobin levels. The attending physician, who was at the hospital when the patient’s condition deteriorated, should have gone to the bedside and taken steps to prevent his death.
THE DEFENSE The patient had been stable overnight; a bedside exam was unnecessary.
VERDICT $1,815,658 Texas verdict.
COMMENT Considering the many demands on clinicians’ time, it’s easy to postpone a face-to-face evaluation of a patient after a procedure. In this case, such a delay cost more than $1.8 million. A laboratory test or nurses’ notes are sometimes inadequate substitutes for a physician’s evaluation.
Failure to investigate suspicious symptoms ends badly
A MAN WITH SIGNS AND SYMPTOMS SUGGESTIVE OF AORTIC ANEURYSM/DISSECTION—including chest pain, pericardial effusion, aortic regurgitation, and aortic dilatation—saw his physician, but the doctor didn’t order any tests, such as computed tomography (CT) with contrast, magnetic resonance imaging (MRI), or transesophageal echocardiogram (TEE).
Two weeks later, the 43-year-old patient returned to the physician, who noted left ventricular hypertrophy with pericardial effusion and mild aortic loop dilatation. Once again, the doctor didn’t order tests to rule out aneurysm/dissection.
Three weeks after the second office visit, the patient collapsed and was taken by ambulance to a hospital, where he was pronounced dead. An autopsy indicated that the cause of death was cardiac tamponade resulting from an undiagnosed aortic dissection.
PLAINTIFF’S CLAIM The physician should have ordered a CT scan with contrast, an MRI, or a TEE, any of which would have confirmed an aortic aneurysm/dissection, mandating immediate admission to a hospital for surgery.
THE DEFENSE No information about the defense is available.
VERDICT $1 million Maryland settlement.
COMMENT Although many common conditions will resolve spontaneously, it’s hard to imagine temporizing in a patient with chest pain and presumed aortic dissection.
Unrecognized spinal infection leads to paralysis
A 355-LB MAN WITH DIABETES AND SPINAL DISC DISEASE experienced a sharp pain between his shoulder blades after playing golf, followed by constant back pain radiating to his chest. He went to the emergency department (ED) the next day and was admitted to the hospital to rule out a heart attack.
During a week in the hospital, the patient was seen by several doctors and diagnosed with pneumonia and excessive myoglobin levels. A computed tomography (CT) scan of the thorax and abdomen showing fluid buildup in the lining around the lungs led to the pneumonia diagnosis. No definitive spinal view was available, however, because of a mixup between a secretary and a radiology technician.
When the patient saw the hospital attending physician (at the family practice group where she was a partner) after discharge from the hospital, he complained of shooting pain down his spine. The doctor prescribed muscle relaxants. Soon afterward, the patient developed difficulty walking and reported no bowel movements for 13 days.
Almost 2 weeks after discharge from the hospital, the patient broke his ankle. He told the paramedics who responded that he felt numb from his nipples to his feet. He was taken to a community hospital, where a doctor ordered another CT scan. The radiologist who read the scan failed to identify the serious spinal infection it indicated.
The patient was transferred back to the original hospital. No doctor saw him for 8 hours after transfer, by which time he was paralyzed from the chest down.
PLAINTIFF’S CLAIM The fluid buildup on the first CT scan was caused not by pneumonia but by an infection in the spinal discs that had spread to the vertebrae and surrounding tissue.
THE DEFENSE The attending physician denied at trial that the patient had told her about the shooting pains down his spine during the posthospitalization visit.
VERDICT $4.75 million Illinois verdict, preceded by more than $2.7 million in settlements with some of the doctors involved and the community hospital.
COMMENT Careful follow-up of ED visits and coordinated care are essential to avoid large verdicts such as this one.
Bedside visit comes too late
A 22-YEAR-OLD MAN underwent a liver biopsy after being admitted to the hospital a week earlier with fever, chills, diarrhea, and general malaise. A number of specialists had seen him in the hospital because of abnormal laboratory studies, increasing fever, and a maculopapular rash over his trunk and face.
After the biopsy, the patient was dizzy and diaphoretic. His attending physician ordered hemoglobin and hematocrit levels, which were lower than earlier that day. Repeat testing showed a further decrease, prompting the physician to order 2 units of red blood cells.
Typing and cross-matching delayed the transfusion for several hours. Before it could be started, the patient was found unresponsive. When the attending physician came to the bedside, the patient had no palpable pulse. A code was called, but resuscitation efforts failed.
An autopsy found a small hole in the liver and 3500 mL of blood in the peritoneal cavity, as well as hepatitis with zonal and submassive necrosis, hemoperitoneum, and hypertrophy of the heart. An HIV test performed before the biopsy eventually came back positive.
PLAINTIFF’S CLAIM The attending physician and nurses were negligent in failing to respond to signs and symptoms of internal bleeding, including falling hematocrit and hemoglobin levels. The attending physician, who was at the hospital when the patient’s condition deteriorated, should have gone to the bedside and taken steps to prevent his death.
THE DEFENSE The patient had been stable overnight; a bedside exam was unnecessary.
VERDICT $1,815,658 Texas verdict.
COMMENT Considering the many demands on clinicians’ time, it’s easy to postpone a face-to-face evaluation of a patient after a procedure. In this case, such a delay cost more than $1.8 million. A laboratory test or nurses’ notes are sometimes inadequate substitutes for a physician’s evaluation.
Failure to investigate suspicious symptoms ends badly
A MAN WITH SIGNS AND SYMPTOMS SUGGESTIVE OF AORTIC ANEURYSM/DISSECTION—including chest pain, pericardial effusion, aortic regurgitation, and aortic dilatation—saw his physician, but the doctor didn’t order any tests, such as computed tomography (CT) with contrast, magnetic resonance imaging (MRI), or transesophageal echocardiogram (TEE).
Two weeks later, the 43-year-old patient returned to the physician, who noted left ventricular hypertrophy with pericardial effusion and mild aortic loop dilatation. Once again, the doctor didn’t order tests to rule out aneurysm/dissection.
Three weeks after the second office visit, the patient collapsed and was taken by ambulance to a hospital, where he was pronounced dead. An autopsy indicated that the cause of death was cardiac tamponade resulting from an undiagnosed aortic dissection.
PLAINTIFF’S CLAIM The physician should have ordered a CT scan with contrast, an MRI, or a TEE, any of which would have confirmed an aortic aneurysm/dissection, mandating immediate admission to a hospital for surgery.
THE DEFENSE No information about the defense is available.
VERDICT $1 million Maryland settlement.
COMMENT Although many common conditions will resolve spontaneously, it’s hard to imagine temporizing in a patient with chest pain and presumed aortic dissection.
Unrecognized spinal infection leads to paralysis
A 355-LB MAN WITH DIABETES AND SPINAL DISC DISEASE experienced a sharp pain between his shoulder blades after playing golf, followed by constant back pain radiating to his chest. He went to the emergency department (ED) the next day and was admitted to the hospital to rule out a heart attack.
During a week in the hospital, the patient was seen by several doctors and diagnosed with pneumonia and excessive myoglobin levels. A computed tomography (CT) scan of the thorax and abdomen showing fluid buildup in the lining around the lungs led to the pneumonia diagnosis. No definitive spinal view was available, however, because of a mixup between a secretary and a radiology technician.
When the patient saw the hospital attending physician (at the family practice group where she was a partner) after discharge from the hospital, he complained of shooting pain down his spine. The doctor prescribed muscle relaxants. Soon afterward, the patient developed difficulty walking and reported no bowel movements for 13 days.
Almost 2 weeks after discharge from the hospital, the patient broke his ankle. He told the paramedics who responded that he felt numb from his nipples to his feet. He was taken to a community hospital, where a doctor ordered another CT scan. The radiologist who read the scan failed to identify the serious spinal infection it indicated.
The patient was transferred back to the original hospital. No doctor saw him for 8 hours after transfer, by which time he was paralyzed from the chest down.
PLAINTIFF’S CLAIM The fluid buildup on the first CT scan was caused not by pneumonia but by an infection in the spinal discs that had spread to the vertebrae and surrounding tissue.
THE DEFENSE The attending physician denied at trial that the patient had told her about the shooting pains down his spine during the posthospitalization visit.
VERDICT $4.75 million Illinois verdict, preceded by more than $2.7 million in settlements with some of the doctors involved and the community hospital.
COMMENT Careful follow-up of ED visits and coordinated care are essential to avoid large verdicts such as this one.
Bedside visit comes too late
A 22-YEAR-OLD MAN underwent a liver biopsy after being admitted to the hospital a week earlier with fever, chills, diarrhea, and general malaise. A number of specialists had seen him in the hospital because of abnormal laboratory studies, increasing fever, and a maculopapular rash over his trunk and face.
After the biopsy, the patient was dizzy and diaphoretic. His attending physician ordered hemoglobin and hematocrit levels, which were lower than earlier that day. Repeat testing showed a further decrease, prompting the physician to order 2 units of red blood cells.
Typing and cross-matching delayed the transfusion for several hours. Before it could be started, the patient was found unresponsive. When the attending physician came to the bedside, the patient had no palpable pulse. A code was called, but resuscitation efforts failed.
An autopsy found a small hole in the liver and 3500 mL of blood in the peritoneal cavity, as well as hepatitis with zonal and submassive necrosis, hemoperitoneum, and hypertrophy of the heart. An HIV test performed before the biopsy eventually came back positive.
PLAINTIFF’S CLAIM The attending physician and nurses were negligent in failing to respond to signs and symptoms of internal bleeding, including falling hematocrit and hemoglobin levels. The attending physician, who was at the hospital when the patient’s condition deteriorated, should have gone to the bedside and taken steps to prevent his death.
THE DEFENSE The patient had been stable overnight; a bedside exam was unnecessary.
VERDICT $1,815,658 Texas verdict.
COMMENT Considering the many demands on clinicians’ time, it’s easy to postpone a face-to-face evaluation of a patient after a procedure. In this case, such a delay cost more than $1.8 million. A laboratory test or nurses’ notes are sometimes inadequate substitutes for a physician’s evaluation.
Failure to investigate suspicious symptoms ends badly
A MAN WITH SIGNS AND SYMPTOMS SUGGESTIVE OF AORTIC ANEURYSM/DISSECTION—including chest pain, pericardial effusion, aortic regurgitation, and aortic dilatation—saw his physician, but the doctor didn’t order any tests, such as computed tomography (CT) with contrast, magnetic resonance imaging (MRI), or transesophageal echocardiogram (TEE).
Two weeks later, the 43-year-old patient returned to the physician, who noted left ventricular hypertrophy with pericardial effusion and mild aortic loop dilatation. Once again, the doctor didn’t order tests to rule out aneurysm/dissection.
Three weeks after the second office visit, the patient collapsed and was taken by ambulance to a hospital, where he was pronounced dead. An autopsy indicated that the cause of death was cardiac tamponade resulting from an undiagnosed aortic dissection.
PLAINTIFF’S CLAIM The physician should have ordered a CT scan with contrast, an MRI, or a TEE, any of which would have confirmed an aortic aneurysm/dissection, mandating immediate admission to a hospital for surgery.
THE DEFENSE No information about the defense is available.
VERDICT $1 million Maryland settlement.
COMMENT Although many common conditions will resolve spontaneously, it’s hard to imagine temporizing in a patient with chest pain and presumed aortic dissection.
Unrecognized spinal infection leads to paralysis
A 355-LB MAN WITH DIABETES AND SPINAL DISC DISEASE experienced a sharp pain between his shoulder blades after playing golf, followed by constant back pain radiating to his chest. He went to the emergency department (ED) the next day and was admitted to the hospital to rule out a heart attack.
During a week in the hospital, the patient was seen by several doctors and diagnosed with pneumonia and excessive myoglobin levels. A computed tomography (CT) scan of the thorax and abdomen showing fluid buildup in the lining around the lungs led to the pneumonia diagnosis. No definitive spinal view was available, however, because of a mixup between a secretary and a radiology technician.
When the patient saw the hospital attending physician (at the family practice group where she was a partner) after discharge from the hospital, he complained of shooting pain down his spine. The doctor prescribed muscle relaxants. Soon afterward, the patient developed difficulty walking and reported no bowel movements for 13 days.
Almost 2 weeks after discharge from the hospital, the patient broke his ankle. He told the paramedics who responded that he felt numb from his nipples to his feet. He was taken to a community hospital, where a doctor ordered another CT scan. The radiologist who read the scan failed to identify the serious spinal infection it indicated.
The patient was transferred back to the original hospital. No doctor saw him for 8 hours after transfer, by which time he was paralyzed from the chest down.
PLAINTIFF’S CLAIM The fluid buildup on the first CT scan was caused not by pneumonia but by an infection in the spinal discs that had spread to the vertebrae and surrounding tissue.
THE DEFENSE The attending physician denied at trial that the patient had told her about the shooting pains down his spine during the posthospitalization visit.
VERDICT $4.75 million Illinois verdict, preceded by more than $2.7 million in settlements with some of the doctors involved and the community hospital.
COMMENT Careful follow-up of ED visits and coordinated care are essential to avoid large verdicts such as this one.