User login
Resecting residual gastrointestinal stromal tumors improved survival
SAN FRANCISCO – Surgically removing residual gastrointestinal stromal tumors in patients who respond to imatinib therapy significantly increased time to tumor progression to 88 months, compared with 43 months using imatinib alone, based on findings from a retrospective study of 134 patients.
After controlling for the effects of other risk factors, the surgery decreased threefold the likelihood of disease progression and decreased fivefold the risk of death, Dr. Seong Joon Park reported in a press briefing sponsored by the American Society of Clinical Oncology (ASCO). The press conference was held in advance of at a meeting on gastrointestinal cancers sponsored by ASCO and three other cancer organizations.
The findings support the widely adopted practice of removing residual tumors in these patients, despite the retrospective and observational design of the study, Dr. Park said. A prospective European study of similar design to this one terminated early due to poor patient enrollment. "It’s really hard to conduct a prospective study of this design," said Dr. Park of Asan Medical Center, Seoul, South Korea.
He and his associates reviewed the records of patients who showed at least 6 months of disease stabilization or response to imatinib (Gleevec) treatment, 92 of whom got the drug treatment alone and 42 of whom underwent surgery to remove residual tumors after a median of 19 months of imatinib therapy. The imatinib therapy was restarted after surgery. Median follow-up for the cohort as a whole was 59 months.
"This treatment strategy is worth trying as a clinical practice if the medical center is large enough to have an experienced multidisciplinary team and to have low morbidity and mortality associated with surgery," he said.
Each year, approximately 5,000 new cases of gastrointestinal stromal tumors are diagnosed in the United States, most often in the stomach and small intestine, though they can occur anywhere in or near the GI tract. Imatinib typically is first-line therapy, and 80%-85% of patients will respond to the treatment, he said. A majority of patients who respond to imatinib will have residual tumors, however, which are believed to contribute to the development of drug resistance, leading to the hypothesis that removing the residual tumors would improve survival.
In general, one-third of patients are candidates for surgical removal of residual lesions, depending on the tumor size and other tumor and patient characteristics, Dr. Park said.
The two patient groups in the study were similar except that the surgery group was significantly younger (51 vs. 58 years) and was less likely to have metastases in the peritoneum (41% in the surgery group vs. 61% in the control group).
As it is an aggressive and difficult treatment, surgery is more likely to be considered and recommended to younger patients who have a good performance status and, thus, less likely to be recommended in patients with multiple peritoneal metastases.
Factors associated with longer progression-free and overall survival included surgery and having an initial tumor size less than 150 mm, multivariate analyses showed. Female sex and having the KIT exon 11 mutation also were associated with longer progression-free survival. The researchers used propensity scores and inverse-probability-weighting adjustments to account for the effects of factors other than surgery.
The gastrointestinal cancers meeting, where Dr. Park will present the results, is cosponsored by ASCO, the American Gastroenterological Association Institute, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Park reported having no financial disclosures.
On Twitter @sherryboschert
GI stromal tumors are an uncommon type of gastrointestinal tumor that can arise at many different places within the GI tract. This disease is notable because it’s really been a triumph of molecularly targeted therapy with imatinib (Gleevec), a drug that targets a particular molecular abnormality present in GI stromal tumors. Because of this, there is an extremely high response rate in patients with GI stromal tumors and drug therapy can control the disease for years.
Unfortunately, resistance ultimately develops to imatinib. This study provides provocative evidence that taking an aggressive approach surgically in addition to medical treatment with imatinib may result in longer survival of patients with GI stromal tumors.
Dr. Neal J. Meropol is chief of hematology and oncology at Case Western Reserve University, Cleveland. He gave these comments as moderator of the press briefing. He has been a consultant or advisor to Precision Therapeutics.
GI stromal tumors are an uncommon type of gastrointestinal tumor that can arise at many different places within the GI tract. This disease is notable because it’s really been a triumph of molecularly targeted therapy with imatinib (Gleevec), a drug that targets a particular molecular abnormality present in GI stromal tumors. Because of this, there is an extremely high response rate in patients with GI stromal tumors and drug therapy can control the disease for years.
Unfortunately, resistance ultimately develops to imatinib. This study provides provocative evidence that taking an aggressive approach surgically in addition to medical treatment with imatinib may result in longer survival of patients with GI stromal tumors.
Dr. Neal J. Meropol is chief of hematology and oncology at Case Western Reserve University, Cleveland. He gave these comments as moderator of the press briefing. He has been a consultant or advisor to Precision Therapeutics.
GI stromal tumors are an uncommon type of gastrointestinal tumor that can arise at many different places within the GI tract. This disease is notable because it’s really been a triumph of molecularly targeted therapy with imatinib (Gleevec), a drug that targets a particular molecular abnormality present in GI stromal tumors. Because of this, there is an extremely high response rate in patients with GI stromal tumors and drug therapy can control the disease for years.
Unfortunately, resistance ultimately develops to imatinib. This study provides provocative evidence that taking an aggressive approach surgically in addition to medical treatment with imatinib may result in longer survival of patients with GI stromal tumors.
Dr. Neal J. Meropol is chief of hematology and oncology at Case Western Reserve University, Cleveland. He gave these comments as moderator of the press briefing. He has been a consultant or advisor to Precision Therapeutics.
SAN FRANCISCO – Surgically removing residual gastrointestinal stromal tumors in patients who respond to imatinib therapy significantly increased time to tumor progression to 88 months, compared with 43 months using imatinib alone, based on findings from a retrospective study of 134 patients.
After controlling for the effects of other risk factors, the surgery decreased threefold the likelihood of disease progression and decreased fivefold the risk of death, Dr. Seong Joon Park reported in a press briefing sponsored by the American Society of Clinical Oncology (ASCO). The press conference was held in advance of at a meeting on gastrointestinal cancers sponsored by ASCO and three other cancer organizations.
The findings support the widely adopted practice of removing residual tumors in these patients, despite the retrospective and observational design of the study, Dr. Park said. A prospective European study of similar design to this one terminated early due to poor patient enrollment. "It’s really hard to conduct a prospective study of this design," said Dr. Park of Asan Medical Center, Seoul, South Korea.
He and his associates reviewed the records of patients who showed at least 6 months of disease stabilization or response to imatinib (Gleevec) treatment, 92 of whom got the drug treatment alone and 42 of whom underwent surgery to remove residual tumors after a median of 19 months of imatinib therapy. The imatinib therapy was restarted after surgery. Median follow-up for the cohort as a whole was 59 months.
"This treatment strategy is worth trying as a clinical practice if the medical center is large enough to have an experienced multidisciplinary team and to have low morbidity and mortality associated with surgery," he said.
Each year, approximately 5,000 new cases of gastrointestinal stromal tumors are diagnosed in the United States, most often in the stomach and small intestine, though they can occur anywhere in or near the GI tract. Imatinib typically is first-line therapy, and 80%-85% of patients will respond to the treatment, he said. A majority of patients who respond to imatinib will have residual tumors, however, which are believed to contribute to the development of drug resistance, leading to the hypothesis that removing the residual tumors would improve survival.
In general, one-third of patients are candidates for surgical removal of residual lesions, depending on the tumor size and other tumor and patient characteristics, Dr. Park said.
The two patient groups in the study were similar except that the surgery group was significantly younger (51 vs. 58 years) and was less likely to have metastases in the peritoneum (41% in the surgery group vs. 61% in the control group).
As it is an aggressive and difficult treatment, surgery is more likely to be considered and recommended to younger patients who have a good performance status and, thus, less likely to be recommended in patients with multiple peritoneal metastases.
Factors associated with longer progression-free and overall survival included surgery and having an initial tumor size less than 150 mm, multivariate analyses showed. Female sex and having the KIT exon 11 mutation also were associated with longer progression-free survival. The researchers used propensity scores and inverse-probability-weighting adjustments to account for the effects of factors other than surgery.
The gastrointestinal cancers meeting, where Dr. Park will present the results, is cosponsored by ASCO, the American Gastroenterological Association Institute, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Park reported having no financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – Surgically removing residual gastrointestinal stromal tumors in patients who respond to imatinib therapy significantly increased time to tumor progression to 88 months, compared with 43 months using imatinib alone, based on findings from a retrospective study of 134 patients.
After controlling for the effects of other risk factors, the surgery decreased threefold the likelihood of disease progression and decreased fivefold the risk of death, Dr. Seong Joon Park reported in a press briefing sponsored by the American Society of Clinical Oncology (ASCO). The press conference was held in advance of at a meeting on gastrointestinal cancers sponsored by ASCO and three other cancer organizations.
The findings support the widely adopted practice of removing residual tumors in these patients, despite the retrospective and observational design of the study, Dr. Park said. A prospective European study of similar design to this one terminated early due to poor patient enrollment. "It’s really hard to conduct a prospective study of this design," said Dr. Park of Asan Medical Center, Seoul, South Korea.
He and his associates reviewed the records of patients who showed at least 6 months of disease stabilization or response to imatinib (Gleevec) treatment, 92 of whom got the drug treatment alone and 42 of whom underwent surgery to remove residual tumors after a median of 19 months of imatinib therapy. The imatinib therapy was restarted after surgery. Median follow-up for the cohort as a whole was 59 months.
"This treatment strategy is worth trying as a clinical practice if the medical center is large enough to have an experienced multidisciplinary team and to have low morbidity and mortality associated with surgery," he said.
Each year, approximately 5,000 new cases of gastrointestinal stromal tumors are diagnosed in the United States, most often in the stomach and small intestine, though they can occur anywhere in or near the GI tract. Imatinib typically is first-line therapy, and 80%-85% of patients will respond to the treatment, he said. A majority of patients who respond to imatinib will have residual tumors, however, which are believed to contribute to the development of drug resistance, leading to the hypothesis that removing the residual tumors would improve survival.
In general, one-third of patients are candidates for surgical removal of residual lesions, depending on the tumor size and other tumor and patient characteristics, Dr. Park said.
The two patient groups in the study were similar except that the surgery group was significantly younger (51 vs. 58 years) and was less likely to have metastases in the peritoneum (41% in the surgery group vs. 61% in the control group).
As it is an aggressive and difficult treatment, surgery is more likely to be considered and recommended to younger patients who have a good performance status and, thus, less likely to be recommended in patients with multiple peritoneal metastases.
Factors associated with longer progression-free and overall survival included surgery and having an initial tumor size less than 150 mm, multivariate analyses showed. Female sex and having the KIT exon 11 mutation also were associated with longer progression-free survival. The researchers used propensity scores and inverse-probability-weighting adjustments to account for the effects of factors other than surgery.
The gastrointestinal cancers meeting, where Dr. Park will present the results, is cosponsored by ASCO, the American Gastroenterological Association Institute, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Park reported having no financial disclosures.
On Twitter @sherryboschert
FROM A PRESS BRIEFING SPONSORED BY THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: Time to tumor progression was 88 months in 42 patients who had surgery for residual disease and 42 months in 92 patients given imatinib alone. Metastases in the peritoneum were present in 41% of the imatinib-and-surgery group and 61% of the imatinib-only group.
Data Source: Retrospective study of 134 patients with metastatic or recurrent gastrointestinal tumors who showed at least 6 months of disease stabilization or response to imatinib, 42 of whom had residual tumors resected.
Disclosures: Dr. Park reported having no financial disclosures.
Helping patients cope with traumatic loss
When tragedies like the Aurora, Colo., and Newtown, Conn., shootings occur, we wonder whether people can ever overcome their grief and find a way to move on with their lives. Froma Walsh, Ph.D., a leading authority on family resilience, has developed an approach that can be used to strengthen families and communities that have suffered such tragedies.
Dr. Walsh is the codirector and cofounder of the Chicago Center for Family Health, and the Mose and Sylvia Firestone Professor Emerita in the school of social service administration and department of psychiatry at the University of Chicago.
Resilience originally was described as individual resilience. Dr. Walsh, who has broadened that concept, provides us with important steps to understand how to help heal families and communities that suffer from unimaginable trauma and loss.
Individual resilience
Beginning in the 1960s, British psychiatrist Sir Michael Rutter studied youth from inner-city London and on the Isle of Wight. Even though they experienced many risk factors, 25% of the children had a good outcome. The children who showed individual resilience had the following characteristics: an easy temperament; female; a positive school climate; self-mastery; self-efficacy; planning skills; and a warm, close, personal relationship with an adult.
From 1955 to 1985, American researchers Emmy E. Werner, Ph.D., and Ruth S. Smith, Ph.D., studied a high-risk population of children in Hawaii and found that 36% showed individual resilience, despite their risk factors. Resilient personal characteristics found in this study were similar: being female, socially responsible, adaptable, tolerant, achievement oriented, a good communicator, and having good self-esteem as well as a caring environment inside and outside the family.
Acquiring resilient qualities
After traumatic events happen, how do people cope? How does the coping process result in growth? We are all familiar with the saying: "What doesn’t break you makes you stronger." This is resilience. How can we help families and communities become stronger?
Family resilience
Dr. Walsh developed the concept of family resilience and a practice framework, identifying key family processes that clinicians can target to strengthen family resilience. She is quick to point out that resilience does not lie in specific characteristics, but rather in family processes that promote resilience. These family processes include a family belief system that pulls family members together and forward, the ability to have hope, a spiritual or value orientation to draw meaning and new purpose out of a tragedy, being able to organize family life to meet challenges, reaching out to extended kin and community networks, collaborative problem solving, and clear communication between members.
Her article, "Traumatic loss and major disasters: strengthening family and community resilience," identifies ways that professionals can help families cope with traumatic loss (Fam. Process 2007;46:207-27). She identifies nine aspects of family life as being key factors in families being able to cope well with traumatic loss:.
• Making meaning of traumatic loss experience. Families ask "Why us?" Families need to make sense of their experiences in a way that allows them to live on. "New meaning" highlights the strengths of the survivors and shows a "new way" of relating to the world and to others. As psychiatrists, we can support the family in finding their own meaning, by helping them examine their beliefs about blame, responsibility, or negligence; and helping them come to terms with accountability and limits of control in the situation.
• Hope. Families need to regain hope in their future possibilities. How does this family want to continue and what does this family want to become? Although they cannot change what has happened, we can support their efforts to "master the possible" by promoting active agency in doing all they can as they move forward.
• Transcendence and spirituality. Families can use their personal values and faith communities for support and to help with forgiveness. Honoring those who died with memorials, anniversary remembrances, and celebrations of milestones in recovery will facilitate healing and growth. These rituals help affirm social values of goodness and compassion. "Recovery is a journey of the heart and spirit, bringing survivors back to the fullness of life," Dr. Walsh wrote in her article.
• Flexibility and stability. Flexibility allows adaptation in the family and allows it to stabilize. Families need to buffer the upheaval and restore security, reliability, and continuity in daily routines. They might need to construct a "new normal" in their lives.
• Connectedness. Each family member has her own adaptation to loss, but family members need to remain mutually supportive. If relationships have prior unresolved conflicts, distress can be worse. Family therapy can prove helpful.
• Extended kin and social resources. Reach out to friends, neighbors, health care providers, clergy and congregational support, school teachers and counselors, employers and coworkers, and neighborhood or community organizations. Multifamily community support groups can help families exchange information, share painful memories and feelings, provide mutual support, and encourage hope and efforts for recovery.
• Clear, consistent information. Families often need help to clarify facts and circumstances of traumatic events and to help their children understand, as age appropriate.
• Emotional sharing and support. Families and the community will experience a wide range of feelings. It is important to allow painful or unacceptable feelings to be expressed and supported, even when differences are viewed as threatening, to avoid the risk of future somatic and emotional disturbance, destructive behavior, or substance abuse. Journals and artwork are helpful, especially with children.
• Collaborative problem solving. Family and communities can coordinate collaborative efforts to promote recovery and resilience. Learning from their experience, they can take steps proactively to prevent future tragedies. One mother who lost a child in the Newtown massacre said she wanted her family and her community to be defined not by their tragedy and suffering, but by the way they are responding, by galvanizing action to stop gun violence.
Community resilience
Community resilience is promoted by Dr. Judith Landau and Jack Saul, Ph.D. in a book edited by Dr. Walsh and Monica McGoldrick, Ph.D., called "Living Beyond Loss: Death in the Family" (see list of further reading below). Community members can create a support system that connects individuals and families. Community support provides a highly connected and reality based understanding. Ongoing support groups can respond accurately to the community needs. Perhaps the community might create a neighborhood resource center or a public space to gather, a witness project, or a community website. Such interventions can have a long-lasting positive impact on the community and the families.
Professional resilience
Compassion fatigue can occur when witnessing trauma and in experiencing ongoing distress. Mental health professionals cannot heal all wounds, but we can create a safe haven for family and community members to share pain and their seeking a new path.
Kaethe Weingarten, Ph.D., has developed a compassionate witnessing project aimed at helping professionals understand the importance of "witnessing" suffering and struggle. She encourages professionals to become aware of their own responses to witnessing trauma and to develop tools to cope with the effects of witnessing trauma.
A family resilience approach is a very positive and constructive way that we can use to teach families how to make sense of what has happened and how to move forward, as difficult as that may currently seem to be.
Further reading:
Here is a list of additional reading you can do to help patients who are facing unimaginable loss:
• "Common Shock: Witnessing Violence Every Day," (New York: Dutton, 2003)
• "Family Resilience: A Framework for Clinical Practice," (Family Process 2003;42:1-18)
• "Living Beyond Loss: Death in the Family," (New York: Norton, 2004, 2nd ed.).
• "Resilience Concepts and Findings: Implications for Family Therapy," (J. Family Therapy 1999;21:119-44).
• "Strengthening Family Resilience," (New York: Guilford Press, 2006)
Dr. Heru is with the department of psychiatry at the University of Colorado at Denver, Aurora. E-mail Dr. Heru at cpnews@elsevier.com.
When tragedies like the Aurora, Colo., and Newtown, Conn., shootings occur, we wonder whether people can ever overcome their grief and find a way to move on with their lives. Froma Walsh, Ph.D., a leading authority on family resilience, has developed an approach that can be used to strengthen families and communities that have suffered such tragedies.
Dr. Walsh is the codirector and cofounder of the Chicago Center for Family Health, and the Mose and Sylvia Firestone Professor Emerita in the school of social service administration and department of psychiatry at the University of Chicago.
Resilience originally was described as individual resilience. Dr. Walsh, who has broadened that concept, provides us with important steps to understand how to help heal families and communities that suffer from unimaginable trauma and loss.
Individual resilience
Beginning in the 1960s, British psychiatrist Sir Michael Rutter studied youth from inner-city London and on the Isle of Wight. Even though they experienced many risk factors, 25% of the children had a good outcome. The children who showed individual resilience had the following characteristics: an easy temperament; female; a positive school climate; self-mastery; self-efficacy; planning skills; and a warm, close, personal relationship with an adult.
From 1955 to 1985, American researchers Emmy E. Werner, Ph.D., and Ruth S. Smith, Ph.D., studied a high-risk population of children in Hawaii and found that 36% showed individual resilience, despite their risk factors. Resilient personal characteristics found in this study were similar: being female, socially responsible, adaptable, tolerant, achievement oriented, a good communicator, and having good self-esteem as well as a caring environment inside and outside the family.
Acquiring resilient qualities
After traumatic events happen, how do people cope? How does the coping process result in growth? We are all familiar with the saying: "What doesn’t break you makes you stronger." This is resilience. How can we help families and communities become stronger?
Family resilience
Dr. Walsh developed the concept of family resilience and a practice framework, identifying key family processes that clinicians can target to strengthen family resilience. She is quick to point out that resilience does not lie in specific characteristics, but rather in family processes that promote resilience. These family processes include a family belief system that pulls family members together and forward, the ability to have hope, a spiritual or value orientation to draw meaning and new purpose out of a tragedy, being able to organize family life to meet challenges, reaching out to extended kin and community networks, collaborative problem solving, and clear communication between members.
Her article, "Traumatic loss and major disasters: strengthening family and community resilience," identifies ways that professionals can help families cope with traumatic loss (Fam. Process 2007;46:207-27). She identifies nine aspects of family life as being key factors in families being able to cope well with traumatic loss:.
• Making meaning of traumatic loss experience. Families ask "Why us?" Families need to make sense of their experiences in a way that allows them to live on. "New meaning" highlights the strengths of the survivors and shows a "new way" of relating to the world and to others. As psychiatrists, we can support the family in finding their own meaning, by helping them examine their beliefs about blame, responsibility, or negligence; and helping them come to terms with accountability and limits of control in the situation.
• Hope. Families need to regain hope in their future possibilities. How does this family want to continue and what does this family want to become? Although they cannot change what has happened, we can support their efforts to "master the possible" by promoting active agency in doing all they can as they move forward.
• Transcendence and spirituality. Families can use their personal values and faith communities for support and to help with forgiveness. Honoring those who died with memorials, anniversary remembrances, and celebrations of milestones in recovery will facilitate healing and growth. These rituals help affirm social values of goodness and compassion. "Recovery is a journey of the heart and spirit, bringing survivors back to the fullness of life," Dr. Walsh wrote in her article.
• Flexibility and stability. Flexibility allows adaptation in the family and allows it to stabilize. Families need to buffer the upheaval and restore security, reliability, and continuity in daily routines. They might need to construct a "new normal" in their lives.
• Connectedness. Each family member has her own adaptation to loss, but family members need to remain mutually supportive. If relationships have prior unresolved conflicts, distress can be worse. Family therapy can prove helpful.
• Extended kin and social resources. Reach out to friends, neighbors, health care providers, clergy and congregational support, school teachers and counselors, employers and coworkers, and neighborhood or community organizations. Multifamily community support groups can help families exchange information, share painful memories and feelings, provide mutual support, and encourage hope and efforts for recovery.
• Clear, consistent information. Families often need help to clarify facts and circumstances of traumatic events and to help their children understand, as age appropriate.
• Emotional sharing and support. Families and the community will experience a wide range of feelings. It is important to allow painful or unacceptable feelings to be expressed and supported, even when differences are viewed as threatening, to avoid the risk of future somatic and emotional disturbance, destructive behavior, or substance abuse. Journals and artwork are helpful, especially with children.
• Collaborative problem solving. Family and communities can coordinate collaborative efforts to promote recovery and resilience. Learning from their experience, they can take steps proactively to prevent future tragedies. One mother who lost a child in the Newtown massacre said she wanted her family and her community to be defined not by their tragedy and suffering, but by the way they are responding, by galvanizing action to stop gun violence.
Community resilience
Community resilience is promoted by Dr. Judith Landau and Jack Saul, Ph.D. in a book edited by Dr. Walsh and Monica McGoldrick, Ph.D., called "Living Beyond Loss: Death in the Family" (see list of further reading below). Community members can create a support system that connects individuals and families. Community support provides a highly connected and reality based understanding. Ongoing support groups can respond accurately to the community needs. Perhaps the community might create a neighborhood resource center or a public space to gather, a witness project, or a community website. Such interventions can have a long-lasting positive impact on the community and the families.
Professional resilience
Compassion fatigue can occur when witnessing trauma and in experiencing ongoing distress. Mental health professionals cannot heal all wounds, but we can create a safe haven for family and community members to share pain and their seeking a new path.
Kaethe Weingarten, Ph.D., has developed a compassionate witnessing project aimed at helping professionals understand the importance of "witnessing" suffering and struggle. She encourages professionals to become aware of their own responses to witnessing trauma and to develop tools to cope with the effects of witnessing trauma.
A family resilience approach is a very positive and constructive way that we can use to teach families how to make sense of what has happened and how to move forward, as difficult as that may currently seem to be.
Further reading:
Here is a list of additional reading you can do to help patients who are facing unimaginable loss:
• "Common Shock: Witnessing Violence Every Day," (New York: Dutton, 2003)
• "Family Resilience: A Framework for Clinical Practice," (Family Process 2003;42:1-18)
• "Living Beyond Loss: Death in the Family," (New York: Norton, 2004, 2nd ed.).
• "Resilience Concepts and Findings: Implications for Family Therapy," (J. Family Therapy 1999;21:119-44).
• "Strengthening Family Resilience," (New York: Guilford Press, 2006)
Dr. Heru is with the department of psychiatry at the University of Colorado at Denver, Aurora. E-mail Dr. Heru at cpnews@elsevier.com.
When tragedies like the Aurora, Colo., and Newtown, Conn., shootings occur, we wonder whether people can ever overcome their grief and find a way to move on with their lives. Froma Walsh, Ph.D., a leading authority on family resilience, has developed an approach that can be used to strengthen families and communities that have suffered such tragedies.
Dr. Walsh is the codirector and cofounder of the Chicago Center for Family Health, and the Mose and Sylvia Firestone Professor Emerita in the school of social service administration and department of psychiatry at the University of Chicago.
Resilience originally was described as individual resilience. Dr. Walsh, who has broadened that concept, provides us with important steps to understand how to help heal families and communities that suffer from unimaginable trauma and loss.
Individual resilience
Beginning in the 1960s, British psychiatrist Sir Michael Rutter studied youth from inner-city London and on the Isle of Wight. Even though they experienced many risk factors, 25% of the children had a good outcome. The children who showed individual resilience had the following characteristics: an easy temperament; female; a positive school climate; self-mastery; self-efficacy; planning skills; and a warm, close, personal relationship with an adult.
From 1955 to 1985, American researchers Emmy E. Werner, Ph.D., and Ruth S. Smith, Ph.D., studied a high-risk population of children in Hawaii and found that 36% showed individual resilience, despite their risk factors. Resilient personal characteristics found in this study were similar: being female, socially responsible, adaptable, tolerant, achievement oriented, a good communicator, and having good self-esteem as well as a caring environment inside and outside the family.
Acquiring resilient qualities
After traumatic events happen, how do people cope? How does the coping process result in growth? We are all familiar with the saying: "What doesn’t break you makes you stronger." This is resilience. How can we help families and communities become stronger?
Family resilience
Dr. Walsh developed the concept of family resilience and a practice framework, identifying key family processes that clinicians can target to strengthen family resilience. She is quick to point out that resilience does not lie in specific characteristics, but rather in family processes that promote resilience. These family processes include a family belief system that pulls family members together and forward, the ability to have hope, a spiritual or value orientation to draw meaning and new purpose out of a tragedy, being able to organize family life to meet challenges, reaching out to extended kin and community networks, collaborative problem solving, and clear communication between members.
Her article, "Traumatic loss and major disasters: strengthening family and community resilience," identifies ways that professionals can help families cope with traumatic loss (Fam. Process 2007;46:207-27). She identifies nine aspects of family life as being key factors in families being able to cope well with traumatic loss:.
• Making meaning of traumatic loss experience. Families ask "Why us?" Families need to make sense of their experiences in a way that allows them to live on. "New meaning" highlights the strengths of the survivors and shows a "new way" of relating to the world and to others. As psychiatrists, we can support the family in finding their own meaning, by helping them examine their beliefs about blame, responsibility, or negligence; and helping them come to terms with accountability and limits of control in the situation.
• Hope. Families need to regain hope in their future possibilities. How does this family want to continue and what does this family want to become? Although they cannot change what has happened, we can support their efforts to "master the possible" by promoting active agency in doing all they can as they move forward.
• Transcendence and spirituality. Families can use their personal values and faith communities for support and to help with forgiveness. Honoring those who died with memorials, anniversary remembrances, and celebrations of milestones in recovery will facilitate healing and growth. These rituals help affirm social values of goodness and compassion. "Recovery is a journey of the heart and spirit, bringing survivors back to the fullness of life," Dr. Walsh wrote in her article.
• Flexibility and stability. Flexibility allows adaptation in the family and allows it to stabilize. Families need to buffer the upheaval and restore security, reliability, and continuity in daily routines. They might need to construct a "new normal" in their lives.
• Connectedness. Each family member has her own adaptation to loss, but family members need to remain mutually supportive. If relationships have prior unresolved conflicts, distress can be worse. Family therapy can prove helpful.
• Extended kin and social resources. Reach out to friends, neighbors, health care providers, clergy and congregational support, school teachers and counselors, employers and coworkers, and neighborhood or community organizations. Multifamily community support groups can help families exchange information, share painful memories and feelings, provide mutual support, and encourage hope and efforts for recovery.
• Clear, consistent information. Families often need help to clarify facts and circumstances of traumatic events and to help their children understand, as age appropriate.
• Emotional sharing and support. Families and the community will experience a wide range of feelings. It is important to allow painful or unacceptable feelings to be expressed and supported, even when differences are viewed as threatening, to avoid the risk of future somatic and emotional disturbance, destructive behavior, or substance abuse. Journals and artwork are helpful, especially with children.
• Collaborative problem solving. Family and communities can coordinate collaborative efforts to promote recovery and resilience. Learning from their experience, they can take steps proactively to prevent future tragedies. One mother who lost a child in the Newtown massacre said she wanted her family and her community to be defined not by their tragedy and suffering, but by the way they are responding, by galvanizing action to stop gun violence.
Community resilience
Community resilience is promoted by Dr. Judith Landau and Jack Saul, Ph.D. in a book edited by Dr. Walsh and Monica McGoldrick, Ph.D., called "Living Beyond Loss: Death in the Family" (see list of further reading below). Community members can create a support system that connects individuals and families. Community support provides a highly connected and reality based understanding. Ongoing support groups can respond accurately to the community needs. Perhaps the community might create a neighborhood resource center or a public space to gather, a witness project, or a community website. Such interventions can have a long-lasting positive impact on the community and the families.
Professional resilience
Compassion fatigue can occur when witnessing trauma and in experiencing ongoing distress. Mental health professionals cannot heal all wounds, but we can create a safe haven for family and community members to share pain and their seeking a new path.
Kaethe Weingarten, Ph.D., has developed a compassionate witnessing project aimed at helping professionals understand the importance of "witnessing" suffering and struggle. She encourages professionals to become aware of their own responses to witnessing trauma and to develop tools to cope with the effects of witnessing trauma.
A family resilience approach is a very positive and constructive way that we can use to teach families how to make sense of what has happened and how to move forward, as difficult as that may currently seem to be.
Further reading:
Here is a list of additional reading you can do to help patients who are facing unimaginable loss:
• "Common Shock: Witnessing Violence Every Day," (New York: Dutton, 2003)
• "Family Resilience: A Framework for Clinical Practice," (Family Process 2003;42:1-18)
• "Living Beyond Loss: Death in the Family," (New York: Norton, 2004, 2nd ed.).
• "Resilience Concepts and Findings: Implications for Family Therapy," (J. Family Therapy 1999;21:119-44).
• "Strengthening Family Resilience," (New York: Guilford Press, 2006)
Dr. Heru is with the department of psychiatry at the University of Colorado at Denver, Aurora. E-mail Dr. Heru at cpnews@elsevier.com.
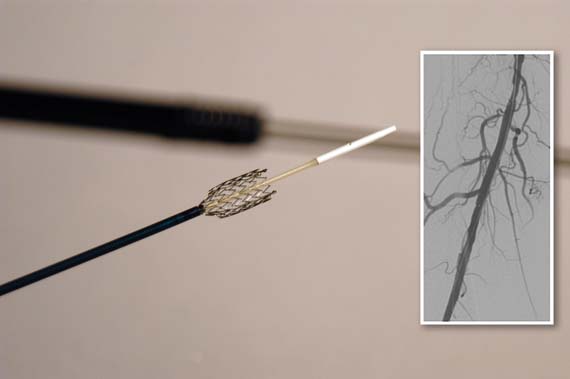
Paclitaxel-eluting stent approved for PAD
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.
Thirty-day readmissions and the posthospital syndrome
Most of us struggle to be proficient in that fine art of balancing high-quality, cost-effective medical care, patient and family satisfaction, and length of stay. One, or even two, of these without the other, just won’t cut muster these days, and with the CMS Hospital Readmissions Reduction Program, the pressure is on to cut readmissions within 30 days of discharge, or else! (Section 3025 of the Affordable Care Act requires the CMS to reduce payments to acute care hospitals with excessive readmission rates as of Oct. 1, 2012.)
Realistically, we all know that even if we provide the best medical care possible, there will always be patients with chronic, end-stage disease whom we will never "fix." The best we can do is optimize the quality of their lives until they die or are readmitted and the cycle begins again. But there may be much more we as hospitalists can do to optimize the overall care of our patients to improve their outcomes, and save our hospitals a great deal of money at the same time.
"Post-Hospital Syndrome – An Acquired, Transient Condition of Generalized Risk" an article published Jan. 10 in the New England Journal of Medicine, gives excellent insight into underappreciated factors that result in readmission to the hospital (N. Engl. J. Med. 2013;368:100-2). Approximately one-fifth of Medicare patients admitted to a hospital require readmission within 30 days, surprisingly often for conditions completely unrelated to the original reason for admission. For instance, among patients initially admitted for heart failure, only 37% of readmissions within 30 days were for worsening heart failure. Likewise, after a hospitalization for a COPD exacerbation, only 36% of readmissions within 30 days were for another exacerbation.
Regardless of the original reason for admission, common causes of readmission include infection, gastrointestinal conditions, metabolic derangements, mental illness, trauma, heart failure, COPD, and pneumonia. And, despite what would seem intuitive, the severity of the initial illness did not help predict which patients would require readmission.
Sleep deprivation is a major contributor to postdischarge morbidity. Polysomnographic studies have demonstrated a reduction in REM sleep and an increase in non-REM sleep in hospitalized patients. Sleep deprivation is known to adversely impact immune function, cardiac risk, and even the coagulation cascade, in addition to the more obvious things such as mental function, ability to ambulate safely, and emotional well-being.
Inadequate nutrition is another culprit. Between being NPO for procedures, which are commonly rescheduled, feeling too sick (or too groggy) to eat, and the increased catabolic demands of many acute illnesses, the pendulum often swings far away from a homeostasis into a potentially dangerous zone, despite how the patient may look at first glance. Some potential consequences of poor nutrition include impaired wound healing, increased infection risk, and decreased cardiac and respiratory function.
Other factors, such as uncontrolled pain, a host of new medications – with myriad side effects – and deconditioning from lying in bed for prolonged periods also play key roles in setting patients up for a potentially debilitating vulnerability that often results in yet another acute illness requiring hospitalization not long after their initial discharge.
We ought to start thinking about discharge planning when we first admit patients. Based on this article, we need to include innovative ways to decrease the posthospital syndrome. There are some simple things we can do: Optimize pain control, get them out of bed as soon as possible, and create an environment to facilitate peaceful sleep. For example, we can easily minimize interruptions during early morning hours for vital sign checks and blood draws in stable patients. (Do we really need routine follow-up lab to be drawn at 6 a.m. in every patient?)
We should all think about our workflow and what we can do differently to minimize the physiologic vulnerability of our patients at discharge so they can be safely discharged (and stay discharged).
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center, Glen Burnie, Md., who has a passion for empowering patients to partner in their health care.
Most of us struggle to be proficient in that fine art of balancing high-quality, cost-effective medical care, patient and family satisfaction, and length of stay. One, or even two, of these without the other, just won’t cut muster these days, and with the CMS Hospital Readmissions Reduction Program, the pressure is on to cut readmissions within 30 days of discharge, or else! (Section 3025 of the Affordable Care Act requires the CMS to reduce payments to acute care hospitals with excessive readmission rates as of Oct. 1, 2012.)
Realistically, we all know that even if we provide the best medical care possible, there will always be patients with chronic, end-stage disease whom we will never "fix." The best we can do is optimize the quality of their lives until they die or are readmitted and the cycle begins again. But there may be much more we as hospitalists can do to optimize the overall care of our patients to improve their outcomes, and save our hospitals a great deal of money at the same time.
"Post-Hospital Syndrome – An Acquired, Transient Condition of Generalized Risk" an article published Jan. 10 in the New England Journal of Medicine, gives excellent insight into underappreciated factors that result in readmission to the hospital (N. Engl. J. Med. 2013;368:100-2). Approximately one-fifth of Medicare patients admitted to a hospital require readmission within 30 days, surprisingly often for conditions completely unrelated to the original reason for admission. For instance, among patients initially admitted for heart failure, only 37% of readmissions within 30 days were for worsening heart failure. Likewise, after a hospitalization for a COPD exacerbation, only 36% of readmissions within 30 days were for another exacerbation.
Regardless of the original reason for admission, common causes of readmission include infection, gastrointestinal conditions, metabolic derangements, mental illness, trauma, heart failure, COPD, and pneumonia. And, despite what would seem intuitive, the severity of the initial illness did not help predict which patients would require readmission.
Sleep deprivation is a major contributor to postdischarge morbidity. Polysomnographic studies have demonstrated a reduction in REM sleep and an increase in non-REM sleep in hospitalized patients. Sleep deprivation is known to adversely impact immune function, cardiac risk, and even the coagulation cascade, in addition to the more obvious things such as mental function, ability to ambulate safely, and emotional well-being.
Inadequate nutrition is another culprit. Between being NPO for procedures, which are commonly rescheduled, feeling too sick (or too groggy) to eat, and the increased catabolic demands of many acute illnesses, the pendulum often swings far away from a homeostasis into a potentially dangerous zone, despite how the patient may look at first glance. Some potential consequences of poor nutrition include impaired wound healing, increased infection risk, and decreased cardiac and respiratory function.
Other factors, such as uncontrolled pain, a host of new medications – with myriad side effects – and deconditioning from lying in bed for prolonged periods also play key roles in setting patients up for a potentially debilitating vulnerability that often results in yet another acute illness requiring hospitalization not long after their initial discharge.
We ought to start thinking about discharge planning when we first admit patients. Based on this article, we need to include innovative ways to decrease the posthospital syndrome. There are some simple things we can do: Optimize pain control, get them out of bed as soon as possible, and create an environment to facilitate peaceful sleep. For example, we can easily minimize interruptions during early morning hours for vital sign checks and blood draws in stable patients. (Do we really need routine follow-up lab to be drawn at 6 a.m. in every patient?)
We should all think about our workflow and what we can do differently to minimize the physiologic vulnerability of our patients at discharge so they can be safely discharged (and stay discharged).
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center, Glen Burnie, Md., who has a passion for empowering patients to partner in their health care.
Most of us struggle to be proficient in that fine art of balancing high-quality, cost-effective medical care, patient and family satisfaction, and length of stay. One, or even two, of these without the other, just won’t cut muster these days, and with the CMS Hospital Readmissions Reduction Program, the pressure is on to cut readmissions within 30 days of discharge, or else! (Section 3025 of the Affordable Care Act requires the CMS to reduce payments to acute care hospitals with excessive readmission rates as of Oct. 1, 2012.)
Realistically, we all know that even if we provide the best medical care possible, there will always be patients with chronic, end-stage disease whom we will never "fix." The best we can do is optimize the quality of their lives until they die or are readmitted and the cycle begins again. But there may be much more we as hospitalists can do to optimize the overall care of our patients to improve their outcomes, and save our hospitals a great deal of money at the same time.
"Post-Hospital Syndrome – An Acquired, Transient Condition of Generalized Risk" an article published Jan. 10 in the New England Journal of Medicine, gives excellent insight into underappreciated factors that result in readmission to the hospital (N. Engl. J. Med. 2013;368:100-2). Approximately one-fifth of Medicare patients admitted to a hospital require readmission within 30 days, surprisingly often for conditions completely unrelated to the original reason for admission. For instance, among patients initially admitted for heart failure, only 37% of readmissions within 30 days were for worsening heart failure. Likewise, after a hospitalization for a COPD exacerbation, only 36% of readmissions within 30 days were for another exacerbation.
Regardless of the original reason for admission, common causes of readmission include infection, gastrointestinal conditions, metabolic derangements, mental illness, trauma, heart failure, COPD, and pneumonia. And, despite what would seem intuitive, the severity of the initial illness did not help predict which patients would require readmission.
Sleep deprivation is a major contributor to postdischarge morbidity. Polysomnographic studies have demonstrated a reduction in REM sleep and an increase in non-REM sleep in hospitalized patients. Sleep deprivation is known to adversely impact immune function, cardiac risk, and even the coagulation cascade, in addition to the more obvious things such as mental function, ability to ambulate safely, and emotional well-being.
Inadequate nutrition is another culprit. Between being NPO for procedures, which are commonly rescheduled, feeling too sick (or too groggy) to eat, and the increased catabolic demands of many acute illnesses, the pendulum often swings far away from a homeostasis into a potentially dangerous zone, despite how the patient may look at first glance. Some potential consequences of poor nutrition include impaired wound healing, increased infection risk, and decreased cardiac and respiratory function.
Other factors, such as uncontrolled pain, a host of new medications – with myriad side effects – and deconditioning from lying in bed for prolonged periods also play key roles in setting patients up for a potentially debilitating vulnerability that often results in yet another acute illness requiring hospitalization not long after their initial discharge.
We ought to start thinking about discharge planning when we first admit patients. Based on this article, we need to include innovative ways to decrease the posthospital syndrome. There are some simple things we can do: Optimize pain control, get them out of bed as soon as possible, and create an environment to facilitate peaceful sleep. For example, we can easily minimize interruptions during early morning hours for vital sign checks and blood draws in stable patients. (Do we really need routine follow-up lab to be drawn at 6 a.m. in every patient?)
We should all think about our workflow and what we can do differently to minimize the physiologic vulnerability of our patients at discharge so they can be safely discharged (and stay discharged).
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center, Glen Burnie, Md., who has a passion for empowering patients to partner in their health care.
The power of culture
We psychiatrists should take a biopsychosocial approach to assessing our patients. However, we are enamored with biology and individual psychodynamics. Thus, we often overlook the influence of culture, or the lack thereof, on human behavior.
The assertion of Dr. Douglas K. Novins that using foundational cultural beliefs and practices strengthens interventions with people of color is particularly powerful. Furthermore, Dr. Novins’s findings inform us about the importance of culture in the protective factors and risky behaviors of our patients.
Culture Protects
While doing HIV prevention work in Durban, South Africa, I found it striking that 40% of the black African Zulu people were HIV positive, 6% of the white South Africans were HIV positive, but only 1% of the East Indian South Africans were HIV positive.
As it turns out, the East Indian South African culture (with its intact religious rituals, proscribed clothing customs, age-old mating practices, and so on) protected them. Meanwhile, the black African Zulu culture and its protective cultural influence had been stripped from them, making them vulnerable to activities such as risky sexual behavior, substance abuse, and violence.
In addition, it appears that the white South African culture is eroding, which is resulting in higher levels of HIV-positive individuals.
Culture Destroys
The latest Youth Risk Behavior Surveillance data offer a glimpse into just how paradoxical our world has become. The investigators looked at six categories of health-risk behaviors among young people and young adults.
Among their findings: The prevalence of having carried a weapon in general was higher among white males (27.2%) than among their black counterparts (21%). The prevalence of having carried a weapon onto school property was higher among white males (7.8%) than black males (6.7%). The prevalence of having ever used cocaine was higher among white males (7.6%) than black males (4.2%). Yet, people of color make up a higher proportion of children and young adults who are incarcerated. In fact, in 2010, the imprisonment rate for black non-Hispanic males (3,074/100,000 U.S. black male residents) was almost seven times higher than it was for white non-Hispanic males (459/100,000), according to the U.S. Bureau of Justice Statistics.
Some of these disparities can be deconstructed by looking at housing patterns. Structurally, we understand that most mid- and large-size cities have more absolute numbers of low-income whites than low-income blacks. But few low-income white neighborhoods exist because low-income whites have scattered-site housing, while low-income blacks are concentrated in inner cities. Police have a more difficult time finding and incarcerating illegal drug users when they live in scattered-site housing. Therefore, blacks who use illegal drugs are incarcerated more often than whites who use illegal drugs.
We also must acknowledge that some of these disparities are tied to the human construct of race. Buy-in to this construct explains why law enforcement officers traditionally "hunted" runaway slaves and returned them to their owners. It potentially explains the motivations of former Chicago police officer Jon Burge, who was convicted 2 years ago of lying about the torture of innocent black men in order to get confessions over many decades. Finally, this reality explains the thinking behind this saying in Chicago’s black community: "The police hunt black males!"
International psychiatrist Suman Fernando makes the point in his book "Mental Health, Race and Culture: Third Edition" (New York: Palgrave Macmillan, 2010) that much of Western culture is inherently racist. Derald Wing Sue, Ph.D., the preeminent multicultural scholar, reminds us of "ethnocentric monoculturalism," the notion that the only culture in the Western world that has any value is Western culture, and all other cultural values and practices are "primitive." Dr. Sue points out that ethnocentric monoculturalism and whiteness define a reality that puts those who are white European American males at an advantage (American Psychologist 2004;59:761-9).
In Canada, these destructive, entrenched views led to the removal of children from First Nations communities. This cultural dislocation, in turn, led to the loss of cultural protective factors, which ultimately contributed to the engagement in risky behaviors tied to suicide, intragroup homicide, and substance abuse. As I’ve discussed previously, many of the disruptive behaviors that result in incarceration can be traced back to alcohol consumption (Preventing fetal alcohol syndrome, April 12, 2012). It is well known that this syndrome is a leading cause of speech and language disorders, attention-deficit/hyperactivity disorder, and other developmental/cognitive disorders. These are often responsible for affect dysregulation, which leads to disruptive behaviors – which, in turn, can lead to incarceration.
It is heartening to see research like that produced by Dr. Novins and his colleagues. Their work reaffirms that culture protects. It also is a reminder that psychiatrists need to understand the sociological forces that exacerbate the emotional pain suffered by our patients – particularly those who are marginalized. We must redouble our efforts to incorporate respectful cultural components into our interventions. Doing so will produce better outcomes.
Dr. Bell is president and chief executive officer of Community Mental Health Council Inc. in Chicago. He also serves as director of the Institute for Juvenile Research at the University of Illinois at Chicago, and is director of public health and community psychiatry at the university.
We psychiatrists should take a biopsychosocial approach to assessing our patients. However, we are enamored with biology and individual psychodynamics. Thus, we often overlook the influence of culture, or the lack thereof, on human behavior.
The assertion of Dr. Douglas K. Novins that using foundational cultural beliefs and practices strengthens interventions with people of color is particularly powerful. Furthermore, Dr. Novins’s findings inform us about the importance of culture in the protective factors and risky behaviors of our patients.
Culture Protects
While doing HIV prevention work in Durban, South Africa, I found it striking that 40% of the black African Zulu people were HIV positive, 6% of the white South Africans were HIV positive, but only 1% of the East Indian South Africans were HIV positive.
As it turns out, the East Indian South African culture (with its intact religious rituals, proscribed clothing customs, age-old mating practices, and so on) protected them. Meanwhile, the black African Zulu culture and its protective cultural influence had been stripped from them, making them vulnerable to activities such as risky sexual behavior, substance abuse, and violence.
In addition, it appears that the white South African culture is eroding, which is resulting in higher levels of HIV-positive individuals.
Culture Destroys
The latest Youth Risk Behavior Surveillance data offer a glimpse into just how paradoxical our world has become. The investigators looked at six categories of health-risk behaviors among young people and young adults.
Among their findings: The prevalence of having carried a weapon in general was higher among white males (27.2%) than among their black counterparts (21%). The prevalence of having carried a weapon onto school property was higher among white males (7.8%) than black males (6.7%). The prevalence of having ever used cocaine was higher among white males (7.6%) than black males (4.2%). Yet, people of color make up a higher proportion of children and young adults who are incarcerated. In fact, in 2010, the imprisonment rate for black non-Hispanic males (3,074/100,000 U.S. black male residents) was almost seven times higher than it was for white non-Hispanic males (459/100,000), according to the U.S. Bureau of Justice Statistics.
Some of these disparities can be deconstructed by looking at housing patterns. Structurally, we understand that most mid- and large-size cities have more absolute numbers of low-income whites than low-income blacks. But few low-income white neighborhoods exist because low-income whites have scattered-site housing, while low-income blacks are concentrated in inner cities. Police have a more difficult time finding and incarcerating illegal drug users when they live in scattered-site housing. Therefore, blacks who use illegal drugs are incarcerated more often than whites who use illegal drugs.
We also must acknowledge that some of these disparities are tied to the human construct of race. Buy-in to this construct explains why law enforcement officers traditionally "hunted" runaway slaves and returned them to their owners. It potentially explains the motivations of former Chicago police officer Jon Burge, who was convicted 2 years ago of lying about the torture of innocent black men in order to get confessions over many decades. Finally, this reality explains the thinking behind this saying in Chicago’s black community: "The police hunt black males!"
International psychiatrist Suman Fernando makes the point in his book "Mental Health, Race and Culture: Third Edition" (New York: Palgrave Macmillan, 2010) that much of Western culture is inherently racist. Derald Wing Sue, Ph.D., the preeminent multicultural scholar, reminds us of "ethnocentric monoculturalism," the notion that the only culture in the Western world that has any value is Western culture, and all other cultural values and practices are "primitive." Dr. Sue points out that ethnocentric monoculturalism and whiteness define a reality that puts those who are white European American males at an advantage (American Psychologist 2004;59:761-9).
In Canada, these destructive, entrenched views led to the removal of children from First Nations communities. This cultural dislocation, in turn, led to the loss of cultural protective factors, which ultimately contributed to the engagement in risky behaviors tied to suicide, intragroup homicide, and substance abuse. As I’ve discussed previously, many of the disruptive behaviors that result in incarceration can be traced back to alcohol consumption (Preventing fetal alcohol syndrome, April 12, 2012). It is well known that this syndrome is a leading cause of speech and language disorders, attention-deficit/hyperactivity disorder, and other developmental/cognitive disorders. These are often responsible for affect dysregulation, which leads to disruptive behaviors – which, in turn, can lead to incarceration.
It is heartening to see research like that produced by Dr. Novins and his colleagues. Their work reaffirms that culture protects. It also is a reminder that psychiatrists need to understand the sociological forces that exacerbate the emotional pain suffered by our patients – particularly those who are marginalized. We must redouble our efforts to incorporate respectful cultural components into our interventions. Doing so will produce better outcomes.
Dr. Bell is president and chief executive officer of Community Mental Health Council Inc. in Chicago. He also serves as director of the Institute for Juvenile Research at the University of Illinois at Chicago, and is director of public health and community psychiatry at the university.
We psychiatrists should take a biopsychosocial approach to assessing our patients. However, we are enamored with biology and individual psychodynamics. Thus, we often overlook the influence of culture, or the lack thereof, on human behavior.
The assertion of Dr. Douglas K. Novins that using foundational cultural beliefs and practices strengthens interventions with people of color is particularly powerful. Furthermore, Dr. Novins’s findings inform us about the importance of culture in the protective factors and risky behaviors of our patients.
Culture Protects
While doing HIV prevention work in Durban, South Africa, I found it striking that 40% of the black African Zulu people were HIV positive, 6% of the white South Africans were HIV positive, but only 1% of the East Indian South Africans were HIV positive.
As it turns out, the East Indian South African culture (with its intact religious rituals, proscribed clothing customs, age-old mating practices, and so on) protected them. Meanwhile, the black African Zulu culture and its protective cultural influence had been stripped from them, making them vulnerable to activities such as risky sexual behavior, substance abuse, and violence.
In addition, it appears that the white South African culture is eroding, which is resulting in higher levels of HIV-positive individuals.
Culture Destroys
The latest Youth Risk Behavior Surveillance data offer a glimpse into just how paradoxical our world has become. The investigators looked at six categories of health-risk behaviors among young people and young adults.
Among their findings: The prevalence of having carried a weapon in general was higher among white males (27.2%) than among their black counterparts (21%). The prevalence of having carried a weapon onto school property was higher among white males (7.8%) than black males (6.7%). The prevalence of having ever used cocaine was higher among white males (7.6%) than black males (4.2%). Yet, people of color make up a higher proportion of children and young adults who are incarcerated. In fact, in 2010, the imprisonment rate for black non-Hispanic males (3,074/100,000 U.S. black male residents) was almost seven times higher than it was for white non-Hispanic males (459/100,000), according to the U.S. Bureau of Justice Statistics.
Some of these disparities can be deconstructed by looking at housing patterns. Structurally, we understand that most mid- and large-size cities have more absolute numbers of low-income whites than low-income blacks. But few low-income white neighborhoods exist because low-income whites have scattered-site housing, while low-income blacks are concentrated in inner cities. Police have a more difficult time finding and incarcerating illegal drug users when they live in scattered-site housing. Therefore, blacks who use illegal drugs are incarcerated more often than whites who use illegal drugs.
We also must acknowledge that some of these disparities are tied to the human construct of race. Buy-in to this construct explains why law enforcement officers traditionally "hunted" runaway slaves and returned them to their owners. It potentially explains the motivations of former Chicago police officer Jon Burge, who was convicted 2 years ago of lying about the torture of innocent black men in order to get confessions over many decades. Finally, this reality explains the thinking behind this saying in Chicago’s black community: "The police hunt black males!"
International psychiatrist Suman Fernando makes the point in his book "Mental Health, Race and Culture: Third Edition" (New York: Palgrave Macmillan, 2010) that much of Western culture is inherently racist. Derald Wing Sue, Ph.D., the preeminent multicultural scholar, reminds us of "ethnocentric monoculturalism," the notion that the only culture in the Western world that has any value is Western culture, and all other cultural values and practices are "primitive." Dr. Sue points out that ethnocentric monoculturalism and whiteness define a reality that puts those who are white European American males at an advantage (American Psychologist 2004;59:761-9).
In Canada, these destructive, entrenched views led to the removal of children from First Nations communities. This cultural dislocation, in turn, led to the loss of cultural protective factors, which ultimately contributed to the engagement in risky behaviors tied to suicide, intragroup homicide, and substance abuse. As I’ve discussed previously, many of the disruptive behaviors that result in incarceration can be traced back to alcohol consumption (Preventing fetal alcohol syndrome, April 12, 2012). It is well known that this syndrome is a leading cause of speech and language disorders, attention-deficit/hyperactivity disorder, and other developmental/cognitive disorders. These are often responsible for affect dysregulation, which leads to disruptive behaviors – which, in turn, can lead to incarceration.
It is heartening to see research like that produced by Dr. Novins and his colleagues. Their work reaffirms that culture protects. It also is a reminder that psychiatrists need to understand the sociological forces that exacerbate the emotional pain suffered by our patients – particularly those who are marginalized. We must redouble our efforts to incorporate respectful cultural components into our interventions. Doing so will produce better outcomes.
Dr. Bell is president and chief executive officer of Community Mental Health Council Inc. in Chicago. He also serves as director of the Institute for Juvenile Research at the University of Illinois at Chicago, and is director of public health and community psychiatry at the university.
Lab test for multiple gastroenteritis pathogens cleared for use
A test that can detect multiple causes of infectious gastroenteritis in one stool sample has been cleared for marketing by the Food and Drug Administration.
The xTAG Gastrointestinal Pathogen Panel (GPP), a multiplexed nucleic acid test, is the "first test that can simultaneously detect 11 common viral, bacterial, and parasitic causes of infectious gastroenteritis from a single patient sample," the agency said in a Jan. 14 statement announcing the approval.
The xTAG GPP tests for the bacteria Campylobacter, Clostridium difficile toxin A/B, Escherichia coli O157, enterotoxigenic E. coli (ETEC) LT/ST, Salmonella, Shigella, and Shiga-like toxin-producing E. coli (STEC) stx1/stx2; the viruses norovirus and rotavirus A; and the parasites Cryptosporidium and Giardia lamblia.
This test can help clinicians identify and treat the cause of gastroenteritis in patients more quickly, and "could also allow clinicians and public health professionals to more quickly identify and investigate the source of potential gastroenteritis outbreaks," Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health at the FDA’s Center for Devices and Radiological Health, said in the statement.
In studies conducted by the manufacturer, Luminex, results of the xTAG GPP were "comparable" with the results of individual tests for the 11 pathogens in stool samples from 1,407 patients with suspected infectious gastroenteritis, 313 samples from pediatric patients with suspected infectious gastroenteritis, and 203 samples from patients with confirmed cases of infectious gastroenteritis, according to the FDA. Because of the risk of false positives, "all positive results from the xTAG GPP need to be confirmed by additional testing," the agency’s announcement said.
The test, which is now available in the United States, can provide multiple results within 5 hours. It is the first and most comprehensive multiplexed product of its kind in the United States, according to a statement from Luminex.
The FDA cites data from the Centers for Disease Control and Prevention reporting that the number of deaths associated with gastroenteritis increased from nearly 7,000 to more than 17,000 per year between 1999 and 2007 in the United States. Two-thirds of the deaths were attributed to norovirus and C. difficile.
A test that can detect multiple causes of infectious gastroenteritis in one stool sample has been cleared for marketing by the Food and Drug Administration.
The xTAG Gastrointestinal Pathogen Panel (GPP), a multiplexed nucleic acid test, is the "first test that can simultaneously detect 11 common viral, bacterial, and parasitic causes of infectious gastroenteritis from a single patient sample," the agency said in a Jan. 14 statement announcing the approval.
The xTAG GPP tests for the bacteria Campylobacter, Clostridium difficile toxin A/B, Escherichia coli O157, enterotoxigenic E. coli (ETEC) LT/ST, Salmonella, Shigella, and Shiga-like toxin-producing E. coli (STEC) stx1/stx2; the viruses norovirus and rotavirus A; and the parasites Cryptosporidium and Giardia lamblia.
This test can help clinicians identify and treat the cause of gastroenteritis in patients more quickly, and "could also allow clinicians and public health professionals to more quickly identify and investigate the source of potential gastroenteritis outbreaks," Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health at the FDA’s Center for Devices and Radiological Health, said in the statement.
In studies conducted by the manufacturer, Luminex, results of the xTAG GPP were "comparable" with the results of individual tests for the 11 pathogens in stool samples from 1,407 patients with suspected infectious gastroenteritis, 313 samples from pediatric patients with suspected infectious gastroenteritis, and 203 samples from patients with confirmed cases of infectious gastroenteritis, according to the FDA. Because of the risk of false positives, "all positive results from the xTAG GPP need to be confirmed by additional testing," the agency’s announcement said.
The test, which is now available in the United States, can provide multiple results within 5 hours. It is the first and most comprehensive multiplexed product of its kind in the United States, according to a statement from Luminex.
The FDA cites data from the Centers for Disease Control and Prevention reporting that the number of deaths associated with gastroenteritis increased from nearly 7,000 to more than 17,000 per year between 1999 and 2007 in the United States. Two-thirds of the deaths were attributed to norovirus and C. difficile.
A test that can detect multiple causes of infectious gastroenteritis in one stool sample has been cleared for marketing by the Food and Drug Administration.
The xTAG Gastrointestinal Pathogen Panel (GPP), a multiplexed nucleic acid test, is the "first test that can simultaneously detect 11 common viral, bacterial, and parasitic causes of infectious gastroenteritis from a single patient sample," the agency said in a Jan. 14 statement announcing the approval.
The xTAG GPP tests for the bacteria Campylobacter, Clostridium difficile toxin A/B, Escherichia coli O157, enterotoxigenic E. coli (ETEC) LT/ST, Salmonella, Shigella, and Shiga-like toxin-producing E. coli (STEC) stx1/stx2; the viruses norovirus and rotavirus A; and the parasites Cryptosporidium and Giardia lamblia.
This test can help clinicians identify and treat the cause of gastroenteritis in patients more quickly, and "could also allow clinicians and public health professionals to more quickly identify and investigate the source of potential gastroenteritis outbreaks," Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health at the FDA’s Center for Devices and Radiological Health, said in the statement.
In studies conducted by the manufacturer, Luminex, results of the xTAG GPP were "comparable" with the results of individual tests for the 11 pathogens in stool samples from 1,407 patients with suspected infectious gastroenteritis, 313 samples from pediatric patients with suspected infectious gastroenteritis, and 203 samples from patients with confirmed cases of infectious gastroenteritis, according to the FDA. Because of the risk of false positives, "all positive results from the xTAG GPP need to be confirmed by additional testing," the agency’s announcement said.
The test, which is now available in the United States, can provide multiple results within 5 hours. It is the first and most comprehensive multiplexed product of its kind in the United States, according to a statement from Luminex.
The FDA cites data from the Centers for Disease Control and Prevention reporting that the number of deaths associated with gastroenteritis increased from nearly 7,000 to more than 17,000 per year between 1999 and 2007 in the United States. Two-thirds of the deaths were attributed to norovirus and C. difficile.
TORS: Postop bleeding risk rises with antithrombotic use
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
AT THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF OTOLARYNGOLOGY; HEAD AND NECK SURGERY FOUNDATION
Major Finding: Approximately three-quarters of patients (8 of 11) who underwent TORS and experienced some postop bleeding were on an antithrombotic medication.
Data Source: A review of 147 consecutive patients at a single surgery center.
Disclosures: Dr. Asher said he had no relevant financial conflicts.
Statins cut risk of hepatocellular carcinoma
Statin therapy taken to prevent cardiovascular events also appears to protect against hepatocellular carcinoma, reducing the overall risk for the cancer by 37%, according to the results of a systematic review and meta-analysis.
In a meta-analysis of all the studies in the literature that have examined statins’ effect on HCC risk, use of the drugs was associated with a pronounced and consistent risk reduction (48%) in Asian populations, as well as a still-significant reduction (33%) in Western populations, reported Dr. Siddharth Singh and his associates at the Mayo Clinic, Rochester, Minn.
Video source: American Gastroenterological Association's YouTube page
At present, "it does not seem prudent to prescribe statins for chemoprevention" of HCC in the general population, mainly because of the high number of people who would need to be treated to prevent a single case of HCC. "However, in patients with multiple risk factors, such as East Asian men who have chronic HBV [hepatitis B virus] infection, statins may have a clinically relevant chemoprotective effect against HCC, the investigators said (Gastroenterology 2012 Oct. 15 [doi: 10.1053/j.gastro.2012.10.005]).
Prospective cohort studies or randomized clinical trials of the issue are warranted in populations at high risk for HCC, they noted.
The results of preclinical studies have suggested that statins may decrease the risk of cancers, perhaps because their antiproliferative, proapoptotic, antiangiogenic, immunomodulatory, and anti-infective effects may prevent cancer growth. But clinical studies have produced conflicting results.
Dr. Singh and his colleagues performed a systematic review of the literature for studies that clearly defined statin exposure, reported HCC risk, and either reported relative risks or odds ratios for the development of HCC or provided the data so those risks could be calculated. They then performed a meta-analysis of 10 studies: 7 observational studies and 3 that reported pooled data from 26 randomized clinical trials.
Most of the studies were considered to be of high quality. Most of them accounted for various potential confounders such as patient age; sex; medication use; and the presence of viral hepatitis, cirrhosis, diabetes, or alcoholic liver disease. The likelihood of selection bias and of publication bias in the included studies was judged to be very low.
Altogether the 10 studies included 1,459,417 subjects and 4,298 cases of HCC.
In an initial analysis of the data, the use of statins was associated with a significant 41% reduction in the rate of HCC. After the data were adjusted to account for several potential confounders, the risk reduction was altered slightly, but a robust 37% reduction in HCC rate remained.
The investigators also performed an analysis of the data based on the location of the studies, because the epidemiology of HCC is so different between Western and Asian populations. They found that statin use correlated with a 48% reduction in the rate of HCC in Asian populations, where viral hepatitis is the primary risk factor for the disease, and a 33% reduction in the rate of HCC in Western populations, where the metabolic syndrome, nonalcoholic fatty liver disease, and alcohol-related cirrhosis are the primary risk factors.
The researchers also performed sensitivity analyses according to the studies’ design (cohort vs. case control) and quality (high vs. low). Both cohort and case-control studies confirmed a protective effect of statins against the development of HCC, as did both high-quality and low-quality studies.
In a final sensitivity analysis, each study was serially excluded from the meta-analysis to determine whether any one study was having a dominant effect on the odds ratios. None of the studies was found to markedly affect the outcomes of the analyses.
The study design didn’t permit separate analyses of the protective effects of statins by drug type or by dose or duration of therapy.
The studies included in this meta-analysis were too heterogeneous to allow the investigators to calculate an overall number needed to treat. But the studies restricted to Asian patients were homogeneous and did allow this calculation for men of Asian ethnicity.
Dr. Singh and his associates determined that 5,209 East Asian men would need to be treated with statins to prevent 1 case of HCC per year. For very-high-risk Asian men with chronic HBV-associated cirrhosis, the number needed to treat with statins to prevent 1 case of HCC per year would be 57.
No financial conflicts of interest were reported.
Statin therapy taken to prevent cardiovascular events also appears to protect against hepatocellular carcinoma, reducing the overall risk for the cancer by 37%, according to the results of a systematic review and meta-analysis.
In a meta-analysis of all the studies in the literature that have examined statins’ effect on HCC risk, use of the drugs was associated with a pronounced and consistent risk reduction (48%) in Asian populations, as well as a still-significant reduction (33%) in Western populations, reported Dr. Siddharth Singh and his associates at the Mayo Clinic, Rochester, Minn.
Video source: American Gastroenterological Association's YouTube page
At present, "it does not seem prudent to prescribe statins for chemoprevention" of HCC in the general population, mainly because of the high number of people who would need to be treated to prevent a single case of HCC. "However, in patients with multiple risk factors, such as East Asian men who have chronic HBV [hepatitis B virus] infection, statins may have a clinically relevant chemoprotective effect against HCC, the investigators said (Gastroenterology 2012 Oct. 15 [doi: 10.1053/j.gastro.2012.10.005]).
Prospective cohort studies or randomized clinical trials of the issue are warranted in populations at high risk for HCC, they noted.
The results of preclinical studies have suggested that statins may decrease the risk of cancers, perhaps because their antiproliferative, proapoptotic, antiangiogenic, immunomodulatory, and anti-infective effects may prevent cancer growth. But clinical studies have produced conflicting results.
Dr. Singh and his colleagues performed a systematic review of the literature for studies that clearly defined statin exposure, reported HCC risk, and either reported relative risks or odds ratios for the development of HCC or provided the data so those risks could be calculated. They then performed a meta-analysis of 10 studies: 7 observational studies and 3 that reported pooled data from 26 randomized clinical trials.
Most of the studies were considered to be of high quality. Most of them accounted for various potential confounders such as patient age; sex; medication use; and the presence of viral hepatitis, cirrhosis, diabetes, or alcoholic liver disease. The likelihood of selection bias and of publication bias in the included studies was judged to be very low.
Altogether the 10 studies included 1,459,417 subjects and 4,298 cases of HCC.
In an initial analysis of the data, the use of statins was associated with a significant 41% reduction in the rate of HCC. After the data were adjusted to account for several potential confounders, the risk reduction was altered slightly, but a robust 37% reduction in HCC rate remained.
The investigators also performed an analysis of the data based on the location of the studies, because the epidemiology of HCC is so different between Western and Asian populations. They found that statin use correlated with a 48% reduction in the rate of HCC in Asian populations, where viral hepatitis is the primary risk factor for the disease, and a 33% reduction in the rate of HCC in Western populations, where the metabolic syndrome, nonalcoholic fatty liver disease, and alcohol-related cirrhosis are the primary risk factors.
The researchers also performed sensitivity analyses according to the studies’ design (cohort vs. case control) and quality (high vs. low). Both cohort and case-control studies confirmed a protective effect of statins against the development of HCC, as did both high-quality and low-quality studies.
In a final sensitivity analysis, each study was serially excluded from the meta-analysis to determine whether any one study was having a dominant effect on the odds ratios. None of the studies was found to markedly affect the outcomes of the analyses.
The study design didn’t permit separate analyses of the protective effects of statins by drug type or by dose or duration of therapy.
The studies included in this meta-analysis were too heterogeneous to allow the investigators to calculate an overall number needed to treat. But the studies restricted to Asian patients were homogeneous and did allow this calculation for men of Asian ethnicity.
Dr. Singh and his associates determined that 5,209 East Asian men would need to be treated with statins to prevent 1 case of HCC per year. For very-high-risk Asian men with chronic HBV-associated cirrhosis, the number needed to treat with statins to prevent 1 case of HCC per year would be 57.
No financial conflicts of interest were reported.
Statin therapy taken to prevent cardiovascular events also appears to protect against hepatocellular carcinoma, reducing the overall risk for the cancer by 37%, according to the results of a systematic review and meta-analysis.
In a meta-analysis of all the studies in the literature that have examined statins’ effect on HCC risk, use of the drugs was associated with a pronounced and consistent risk reduction (48%) in Asian populations, as well as a still-significant reduction (33%) in Western populations, reported Dr. Siddharth Singh and his associates at the Mayo Clinic, Rochester, Minn.
Video source: American Gastroenterological Association's YouTube page
At present, "it does not seem prudent to prescribe statins for chemoprevention" of HCC in the general population, mainly because of the high number of people who would need to be treated to prevent a single case of HCC. "However, in patients with multiple risk factors, such as East Asian men who have chronic HBV [hepatitis B virus] infection, statins may have a clinically relevant chemoprotective effect against HCC, the investigators said (Gastroenterology 2012 Oct. 15 [doi: 10.1053/j.gastro.2012.10.005]).
Prospective cohort studies or randomized clinical trials of the issue are warranted in populations at high risk for HCC, they noted.
The results of preclinical studies have suggested that statins may decrease the risk of cancers, perhaps because their antiproliferative, proapoptotic, antiangiogenic, immunomodulatory, and anti-infective effects may prevent cancer growth. But clinical studies have produced conflicting results.
Dr. Singh and his colleagues performed a systematic review of the literature for studies that clearly defined statin exposure, reported HCC risk, and either reported relative risks or odds ratios for the development of HCC or provided the data so those risks could be calculated. They then performed a meta-analysis of 10 studies: 7 observational studies and 3 that reported pooled data from 26 randomized clinical trials.
Most of the studies were considered to be of high quality. Most of them accounted for various potential confounders such as patient age; sex; medication use; and the presence of viral hepatitis, cirrhosis, diabetes, or alcoholic liver disease. The likelihood of selection bias and of publication bias in the included studies was judged to be very low.
Altogether the 10 studies included 1,459,417 subjects and 4,298 cases of HCC.
In an initial analysis of the data, the use of statins was associated with a significant 41% reduction in the rate of HCC. After the data were adjusted to account for several potential confounders, the risk reduction was altered slightly, but a robust 37% reduction in HCC rate remained.
The investigators also performed an analysis of the data based on the location of the studies, because the epidemiology of HCC is so different between Western and Asian populations. They found that statin use correlated with a 48% reduction in the rate of HCC in Asian populations, where viral hepatitis is the primary risk factor for the disease, and a 33% reduction in the rate of HCC in Western populations, where the metabolic syndrome, nonalcoholic fatty liver disease, and alcohol-related cirrhosis are the primary risk factors.
The researchers also performed sensitivity analyses according to the studies’ design (cohort vs. case control) and quality (high vs. low). Both cohort and case-control studies confirmed a protective effect of statins against the development of HCC, as did both high-quality and low-quality studies.
In a final sensitivity analysis, each study was serially excluded from the meta-analysis to determine whether any one study was having a dominant effect on the odds ratios. None of the studies was found to markedly affect the outcomes of the analyses.
The study design didn’t permit separate analyses of the protective effects of statins by drug type or by dose or duration of therapy.
The studies included in this meta-analysis were too heterogeneous to allow the investigators to calculate an overall number needed to treat. But the studies restricted to Asian patients were homogeneous and did allow this calculation for men of Asian ethnicity.
Dr. Singh and his associates determined that 5,209 East Asian men would need to be treated with statins to prevent 1 case of HCC per year. For very-high-risk Asian men with chronic HBV-associated cirrhosis, the number needed to treat with statins to prevent 1 case of HCC per year would be 57.
No financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Major Finding: Statin therapy reduced the risk of developing hepatocellular carcinoma by 37% overall; the risk reduction was stronger (48%) in Asian populations but still significant (33%) in Western populations.
Data Source: A systematic review and meta-analysis of 10 observational studies or randomized clinical trials involving 1,459,417 subjects, of whom 4,298 developed HCC during follow-up.
Disclosures: No financial conflicts of interest were reported.
Case of the Month
A 56-year-old Hispanic female with a past medical history significant for basal cell carcinoma presented with a history of itchy, erythematous papules on her right cheek. Four days prior, she presented for suture removal after reconstruction with an island pedicle flap following Mohs micrographic surgery. She experienced a similar rash on her forearm following another surgery in the past.
a) Cellulitis
b) Contact dermatitis
c) Herpes simplex virus
Diagnosis: Contact dermatitis secondary to Mastisol and Steri-Strips
Contact dermatitis is a localized, pruritic, erythematous rash that occurs after contact with a certain allergen or irritant. The disorder is typically classified as either allergic contact dermatitis or irritant contact dermatitis.
Allergic contact dermatitis is a T cell–mediated, type-IV, delayed-type hypersensitivity reaction that requires prior sensitization with the causative agent before the patient becomes allergic to it. The typical rash of pruritus, erythema, edema, and vesicle formation occurs with further exposures.
The mechanism of immune response to a particular allergen requires that the antigen be of low molecular weight (less than 500 d) in order to penetrate the stratum corneum and gain access to the immunologic system. CD4, CD8, T regulatory cells, and natural killer T cells have all been implicated.
The process is composed of an afferent (sensitization) phase and an efferent (elicitation) phase.
Common haptens or immunogenic agents include nickel, urushiol from poison ivy resin, ultraviolet light, dyes, and fragrances.
This patient revealed a history of a similar reaction to the one presented in this case following wound dressing on her forearm with two products: Steri-Strips and Mastisol liquid adhesive. Unfortunately, she did not reveal this history until she had experienced the reaction a second time. The patient was instructed to apply hydrocortisone 1% cream twice daily to the red areas only and to follow up with a clinic visit in 4-5 days. Documentation of her allergy was included in her medical record.
This case was submitted by Dr. Keyvan Nouri; Dr. Katlein Franca; Jennifer Ledon; and Jessica Savas of the University of Miami.
–Donna Bilu Martin, M.D.
A 56-year-old Hispanic female with a past medical history significant for basal cell carcinoma presented with a history of itchy, erythematous papules on her right cheek. Four days prior, she presented for suture removal after reconstruction with an island pedicle flap following Mohs micrographic surgery. She experienced a similar rash on her forearm following another surgery in the past.
a) Cellulitis
b) Contact dermatitis
c) Herpes simplex virus
Diagnosis: Contact dermatitis secondary to Mastisol and Steri-Strips
Contact dermatitis is a localized, pruritic, erythematous rash that occurs after contact with a certain allergen or irritant. The disorder is typically classified as either allergic contact dermatitis or irritant contact dermatitis.
Allergic contact dermatitis is a T cell–mediated, type-IV, delayed-type hypersensitivity reaction that requires prior sensitization with the causative agent before the patient becomes allergic to it. The typical rash of pruritus, erythema, edema, and vesicle formation occurs with further exposures.
The mechanism of immune response to a particular allergen requires that the antigen be of low molecular weight (less than 500 d) in order to penetrate the stratum corneum and gain access to the immunologic system. CD4, CD8, T regulatory cells, and natural killer T cells have all been implicated.
The process is composed of an afferent (sensitization) phase and an efferent (elicitation) phase.
Common haptens or immunogenic agents include nickel, urushiol from poison ivy resin, ultraviolet light, dyes, and fragrances.
This patient revealed a history of a similar reaction to the one presented in this case following wound dressing on her forearm with two products: Steri-Strips and Mastisol liquid adhesive. Unfortunately, she did not reveal this history until she had experienced the reaction a second time. The patient was instructed to apply hydrocortisone 1% cream twice daily to the red areas only and to follow up with a clinic visit in 4-5 days. Documentation of her allergy was included in her medical record.
This case was submitted by Dr. Keyvan Nouri; Dr. Katlein Franca; Jennifer Ledon; and Jessica Savas of the University of Miami.
–Donna Bilu Martin, M.D.
A 56-year-old Hispanic female with a past medical history significant for basal cell carcinoma presented with a history of itchy, erythematous papules on her right cheek. Four days prior, she presented for suture removal after reconstruction with an island pedicle flap following Mohs micrographic surgery. She experienced a similar rash on her forearm following another surgery in the past.
a) Cellulitis
b) Contact dermatitis
c) Herpes simplex virus
Diagnosis: Contact dermatitis secondary to Mastisol and Steri-Strips
Contact dermatitis is a localized, pruritic, erythematous rash that occurs after contact with a certain allergen or irritant. The disorder is typically classified as either allergic contact dermatitis or irritant contact dermatitis.
Allergic contact dermatitis is a T cell–mediated, type-IV, delayed-type hypersensitivity reaction that requires prior sensitization with the causative agent before the patient becomes allergic to it. The typical rash of pruritus, erythema, edema, and vesicle formation occurs with further exposures.
The mechanism of immune response to a particular allergen requires that the antigen be of low molecular weight (less than 500 d) in order to penetrate the stratum corneum and gain access to the immunologic system. CD4, CD8, T regulatory cells, and natural killer T cells have all been implicated.
The process is composed of an afferent (sensitization) phase and an efferent (elicitation) phase.
Common haptens or immunogenic agents include nickel, urushiol from poison ivy resin, ultraviolet light, dyes, and fragrances.
This patient revealed a history of a similar reaction to the one presented in this case following wound dressing on her forearm with two products: Steri-Strips and Mastisol liquid adhesive. Unfortunately, she did not reveal this history until she had experienced the reaction a second time. The patient was instructed to apply hydrocortisone 1% cream twice daily to the red areas only and to follow up with a clinic visit in 4-5 days. Documentation of her allergy was included in her medical record.
This case was submitted by Dr. Keyvan Nouri; Dr. Katlein Franca; Jennifer Ledon; and Jessica Savas of the University of Miami.
–Donna Bilu Martin, M.D.
FDA approves plasma product
The FDA has approved a pooled plasma blood product (Octaplas) that can be used to replace coagulation factors in patients with certain medical conditions.
The product is a sterile, frozen solution of human plasma from several donors that has been treated with a solvent detergent process to minimize the risk of serious virus transmission.
The plasma used to manufacture Octaplas is collected from US donors who have been screened and tested for diseases transmitted by blood.
“For patients suffering with clotting disorders, this product provides a viable alternative to single-donor fresh-frozen plasma [FFP] and provides a reduced risk of certain viral transmissions,” said Karen Midthun, MD, director of the FDA’s Center for Biologics Evaluation and Research.
Indications and administration
Octaplas is indicated for the replacement of multiple coagulation factors in patients with acquired deficiencies due to liver disease or undergoing cardiac surgery or liver transplant. Octaplas can also be
used for plasma exchange in patients with thrombotic thrombocytopenic purpura (TTP).
Like FFP, Octaplas should be matched to the recipient’s blood group to help avoid transfusion reactions. Each lot of Octaplas is tested for composition of key clotting factors and is only released if the levels are within acceptable ranges.
The product is administered by intravenous infusion after thawing, using an infusion set with a filter. An aseptic technique must be used throughout the infusion.
The dosage depends upon the clinical situation and the underlying disorder. But 12-15 mL/kg of body weight is a generally accepted starting dose, and it should increase the patient’s plasma coagulation factor levels by about 25%.
It is important to monitor patient response, both clinically and with measurement of prothrombin time, partial thromboplastin time, and/or specific coagulation factor assays.
Prior experience with Octaplas
The FDA’s approval of Octaplas was primarily based on studies conducted in patients with liver disease, liver transplant, heart surgery, and TTP. The most common adverse reactions observed in these trials were shortness of breath, dizziness, chest discomfort, pruritis/rash, headache, and paresthesia.
Additional data supporting the safe use of Octaplas for the US market came from prior use of the product in Europe and other approved markets, where it has been used extensively.
A previous generation of Octaplas was first marketed in 1992, and the current version has been marketed since 2006. In total, more than 2 million patients outside the US have been treated with more than 7 million doses of Octaplas.
The product is manufactured by Octapharma, located in Vienna, Austria. For more information on Octaplas, visit the Octapharma website. ![]()

The FDA has approved a pooled plasma blood product (Octaplas) that can be used to replace coagulation factors in patients with certain medical conditions.
The product is a sterile, frozen solution of human plasma from several donors that has been treated with a solvent detergent process to minimize the risk of serious virus transmission.
The plasma used to manufacture Octaplas is collected from US donors who have been screened and tested for diseases transmitted by blood.
“For patients suffering with clotting disorders, this product provides a viable alternative to single-donor fresh-frozen plasma [FFP] and provides a reduced risk of certain viral transmissions,” said Karen Midthun, MD, director of the FDA’s Center for Biologics Evaluation and Research.
Indications and administration
Octaplas is indicated for the replacement of multiple coagulation factors in patients with acquired deficiencies due to liver disease or undergoing cardiac surgery or liver transplant. Octaplas can also be
used for plasma exchange in patients with thrombotic thrombocytopenic purpura (TTP).
Like FFP, Octaplas should be matched to the recipient’s blood group to help avoid transfusion reactions. Each lot of Octaplas is tested for composition of key clotting factors and is only released if the levels are within acceptable ranges.
The product is administered by intravenous infusion after thawing, using an infusion set with a filter. An aseptic technique must be used throughout the infusion.
The dosage depends upon the clinical situation and the underlying disorder. But 12-15 mL/kg of body weight is a generally accepted starting dose, and it should increase the patient’s plasma coagulation factor levels by about 25%.
It is important to monitor patient response, both clinically and with measurement of prothrombin time, partial thromboplastin time, and/or specific coagulation factor assays.
Prior experience with Octaplas
The FDA’s approval of Octaplas was primarily based on studies conducted in patients with liver disease, liver transplant, heart surgery, and TTP. The most common adverse reactions observed in these trials were shortness of breath, dizziness, chest discomfort, pruritis/rash, headache, and paresthesia.
Additional data supporting the safe use of Octaplas for the US market came from prior use of the product in Europe and other approved markets, where it has been used extensively.
A previous generation of Octaplas was first marketed in 1992, and the current version has been marketed since 2006. In total, more than 2 million patients outside the US have been treated with more than 7 million doses of Octaplas.
The product is manufactured by Octapharma, located in Vienna, Austria. For more information on Octaplas, visit the Octapharma website. ![]()

The FDA has approved a pooled plasma blood product (Octaplas) that can be used to replace coagulation factors in patients with certain medical conditions.
The product is a sterile, frozen solution of human plasma from several donors that has been treated with a solvent detergent process to minimize the risk of serious virus transmission.
The plasma used to manufacture Octaplas is collected from US donors who have been screened and tested for diseases transmitted by blood.
“For patients suffering with clotting disorders, this product provides a viable alternative to single-donor fresh-frozen plasma [FFP] and provides a reduced risk of certain viral transmissions,” said Karen Midthun, MD, director of the FDA’s Center for Biologics Evaluation and Research.
Indications and administration
Octaplas is indicated for the replacement of multiple coagulation factors in patients with acquired deficiencies due to liver disease or undergoing cardiac surgery or liver transplant. Octaplas can also be
used for plasma exchange in patients with thrombotic thrombocytopenic purpura (TTP).
Like FFP, Octaplas should be matched to the recipient’s blood group to help avoid transfusion reactions. Each lot of Octaplas is tested for composition of key clotting factors and is only released if the levels are within acceptable ranges.
The product is administered by intravenous infusion after thawing, using an infusion set with a filter. An aseptic technique must be used throughout the infusion.
The dosage depends upon the clinical situation and the underlying disorder. But 12-15 mL/kg of body weight is a generally accepted starting dose, and it should increase the patient’s plasma coagulation factor levels by about 25%.
It is important to monitor patient response, both clinically and with measurement of prothrombin time, partial thromboplastin time, and/or specific coagulation factor assays.
Prior experience with Octaplas
The FDA’s approval of Octaplas was primarily based on studies conducted in patients with liver disease, liver transplant, heart surgery, and TTP. The most common adverse reactions observed in these trials were shortness of breath, dizziness, chest discomfort, pruritis/rash, headache, and paresthesia.
Additional data supporting the safe use of Octaplas for the US market came from prior use of the product in Europe and other approved markets, where it has been used extensively.
A previous generation of Octaplas was first marketed in 1992, and the current version has been marketed since 2006. In total, more than 2 million patients outside the US have been treated with more than 7 million doses of Octaplas.
The product is manufactured by Octapharma, located in Vienna, Austria. For more information on Octaplas, visit the Octapharma website. ![]()