User login
Price Display Systematic Review
Rising healthcare spending has garnered significant public attention, and is considered a threat to other national priorities. Up to one‐third of national health expenditures are wasteful, the largest fraction generated through unnecessary services that could be substituted for less‐costly alternatives or omitted altogether.[1] Physicians play a central role in health spending, as they purchase nearly all tests and therapies on behalf of patients.
One strategy to enhance cost‐conscious physician ordering is to increase transparency of cost data for providers.[2, 3, 4] Although physicians consider price an important factor in ordering decisions, they have difficulty estimating costs accurately or finding price information easily.[5, 6] Improving physicians' knowledge of order costs may prompt them to forego diagnostic tests or therapies of low utility, or shift ordering to lower‐cost alternatives. Real‐time price display during provider order entry is 1 approach for achieving this goal. Modern electronic health records (EHRs) with computerized physician order entry (CPOE) make price display not only practical but also scalable. Integrating price display into clinical workflow, however, can be challenging, and there remains lack of clarity about potential risks and benefits. The dissemination of real‐time CPOE price display, therefore, requires an understanding of its impact on clinical care.
Over the past 3 decades, several studies in the medical literature have evaluated the effect of price display on physician ordering behavior. To date, however, there has been only 1 narrative review of this literature, which did not include several recent studies on the topic or formally address study quality and physician acceptance of price display modules.[7] Therefore, to help inform healthcare leaders, technology innovators, and policy makers, we conducted a systematic review to address 4 key questions: (1) What are the characteristics of interventions that have displayed order prices to physicians in the context of actual practice? (2) To what degree does real‐time display of order prices impact order costs and order volume? (3) Does price display impact patient safety outcomes, and is it acceptable to providers? (4) What is the quality of the current literature on this topic?
METHODS
Data Sources
We searched 2 electronic databases, MEDLINE and Embase, using a combination of controlled vocabulary terms and keywords that covered both the targeted intervention (eg, fees and charges) and the outcome of interest (eg, physician's practice patterns), limited to English language articles with no restriction on country or year of publication (see Supporting Information, Appendix 1, in the online version of this article). The search was run through August 2014. Results from both database searches were combined and duplicates eliminated. We also ran a MEDLINE keyword search on titles and abstracts of articles from 2014 that were not yet indexed. A medical librarian was involved in all aspects of the search process.[8]
Study Selection
Studies were included if they evaluated the effect of displaying actual order prices to providers during the ordering process and reported the impact on provider ordering practices. Reports in any clinical context and with any study design were included. To assess most accurately the effect of price display on real‐life ordering and patient outcomes, studies were excluded if: (1) they were review articles, commentaries, or editorials; (2) they did not show order prices to providers; (3) the context was a simulation; (4) the prices displayed were relative (eg, $/$$/$$$) or were only cumulative; (5) prices were not presented real‐time during the ordering process; or (6) the primary outcome was neither order costs nor order volume. We decided a priori to exclude simulations because these may not accurately reflect provider behavior when treating real patients, and to exclude studies showing relative prices due to concerns that it is a less significant price transparency intervention and that providers may interpret relative prices differently from actual prices.
Two reviewers, both physicians and health service researchers (M.T.S. and T.R.B.), separately reviewed the full list of titles and abstracts. For studies that potentially met inclusion criteria, full articles were obtained and were independently read for inclusion in the final review. The references of all included studies were searched manually, and the Scopus database was used to search all studies that cited the included studies. We also searched the references of relevant literature reviews.[9, 10, 11] Articles of interest discovered through manual search were then subjected to the same process.
Data Extraction and Quality Assessment
Two reviewers (M.T.S. and T.R.B.) independently performed data extraction using a standardized spreadsheet. Discrepancies were resolved by reviewer consensus. Extracted study characteristics included study design and duration, clinical setting, study size, type of orders involved, characteristics of price display intervention and control, and type of outcome. Findings regarding patient safety and provider acceptability were also extracted when available.
Study quality was independently evaluated and scored by both reviewers using the Downs and Black checklist, designed to assess quality of both randomized and nonrandomized studies.[12] The checklist contains 5 items pertaining to allocation concealment, blinding, or follow‐up that are not applicable to an administrative intervention like price display, so these questions were excluded. Additionally, few studies calculated sample size or reported post hoc statistical power, so we also excluded this question, leaving a modified 21‐item checklist. We also assessed each study for sources of bias that were not already assessed by the Downs and Black checklist, including contamination between study groups, confounding of results, and incomplete intervention or data collection.
Data Synthesis
Data are reported in tabular form for all included studies. Due to heterogeneity of study designs and outcome measures, data from the studies were not pooled quantitatively. This review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis guidelines.
RESULTS
Database searches yielded a total of 1400 articles, of which 18 were selected on the basis of title and abstract for detailed assessment. Reference searching led us to retrieve 94 further studies of possible interest, of which 23 were selected on the basis of abstract for detailed assessment. Thus, 41 publications underwent full manuscript review, 19 of which met all inclusion criteria (see Supporting Information, Appendix 2, in the online version of this article).[13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31] These studies were published between 1983 and 2014, and were conducted primarily in the United States.
Study Characteristics
There was considerable heterogeneity among the 19 studies with regard to design, setting, and scope (Table 1). There were 5 randomized trials, for which the units of randomization were patient (1), provider team (2), and test (2). There were 13 pre‐post intervention studies, 5 of which used a concomitant control group, and 2 of which included a washout period. There was 1 interrupted time series study. Studies were conducted within inpatient hospital floors (8), outpatient clinics (4), emergency departments (ED) or urgent care facilities (4), and hospital operating rooms (3).
| Study | Design | Clinical Setting | Providers | Intervention and Duration | Order(s) Studied | Type of Price Displayed | Concurrent Interventions |
|---|---|---|---|---|---|---|---|
| |||||||
| Fang et al.[14] 2014 | Pre‐post study with control group | Academic hospital (USA) | All inpatient ordering providers | CPOE system with prices displayed for reference lab tests; 8 months | All send‐out lab tests | Charge from send‐out laboratory, displayed as range (eg, $100300) | Display also contained expected lab turnaround time |
| Nougon et al.[13] 2014 | Pre‐post study with washout | Academic adult emergency department (Belgium) | 9 ED house staff | CPOE system with prices displayed on common orders form, and price list displayed above all workstations and in patient rooms; 2 months | Common lab and imaging tests | Reference costs from Belgian National Institute for Health Insurance and Invalidity | None |
| Durand et al.[17] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 10 common imaging tests | Medicare allowable fee | None |
| Feldman et al.[16] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 61 lab tests | Medicare allowable fee | None |
| Horn et al.[15] 2014 | Interrupted time series study with control group | Private outpatient group practice alliance (USA) | 215 primary care physicians | CPOE system with prices displayed; 6 months | 27 lab tests | Medicare allowable fee, displayed as narrow range (eg, $5$10) | None |
| Ellemdin et al.[18] 2011 | Pre‐post study with control group | Academic hospital, internal medicine units (South Africa) | Internal medicine physicians (number NR) | Sheet with lab test costs given to intervention group physicians who were required to write out cost for each order; 4 months | Common lab tests | Not reported | None |
| Schilling,[19] 2010 | Pre‐post study with control group | Academic adult emergency department (Sweden) | All internal medicine physicians in ED | Standard provider workstations with price lists posted on each; 2 months | 91 common lab tests, 39 common imaging tests | Not reported | None |
| Guterman et al.[21] 2002 | Pre‐post study | Academic‐affiliated urgent care clinic (USA) | 51 attendings and housestaff | Preformatted paper prescription form with medication prices displayed; 2 weeks | 2 H2‐blocker medications | Acquisition cost of medication plus fill fee | None |
| Seguin et al.[20] 2002 | Pre‐post study | Academic surgical intensive care unit (France) | All intensive care unit physicians | Paper quick‐order checklist with prices displayed; 2 months | 6 common lab tests, 1 imaging test | Not reported | None |
| Hampers et al.[23] 1999 | Pre‐post study with washout | Academic pediatric emergency department (USA) | Pediatric ED attendings and housestaff (number NR) | Paper common‐order checklist with prices displayed; 3 months | 22 common lab and imaging tests | Chargemaster price* | Physicians required to calculate total charges for diagnostic workup |
| Ornstein et al.[22] 1999 | Pre‐post study | Academic family medicine outpatient clinic (USA) | 46 attendings and housestaff | Microcomputer CPOE system with medication prices displayed; 6 months | All medications | AWP for total supply (acute medications) or 30‐day supply (chronic medications) | Additional keystroke produced list of less costly alternative medications |
| Lin et al.[25] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | All anesthesia providers | Standard muscle relaxant drug vials with price stickers displayed; 12 months | All muscle relaxant medications | Not reported | None |
| McNitt et al.[24] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | 90 anesthesia attendings, housestaff and anesthetists | List of drug costs displayed in operating rooms, anesthesia lounge, and anesthesia satellite pharmacy; 10 months | 22 common anesthesia medications | Hospital acquisition cost | Regular anesthesia department reviews of drug usage and cost |
| Bates et al.[27] 1997 | RCT (randomized by patient) | Academic hospital, medical and surgical inpatients (USA) | All inpatient ordering providers | CPOE system with display of test price and running total of prices for the ordering session; 4 months (lab) and 7 months (imaging) | All lab tests, 35 common imaging tests | Chargemaster price | None |
| Vedsted et al.[26] 1997 | Pre‐post study with control group | Outpatient general practices (Denmark) | 231 general practitioners | In practices already using APEX CPOE system, introduction of medication price display (control practices used non‐APEX computer system or paper‐based prescribing); 12 months | All medications | Chargemaster price | Medication price comparison module (stars indicated availability of cheaper option) |
| Horrow et al.[28] 1994 | Pre‐post study | Private tertiary care hospital operating rooms (USA) | 56 anesthesia attendings, housestaff and anesthetists | Standard anesthesia drug vials and syringes with supermarket price stickers displayed; 3 months | 13 neuromuscular relaxant and sedative‐hypnotic medications | Hospital acquisition cost | None |
| Tierney et al.[29] 1993 | Cluster RCT (randomized by provider team) | Public hospital, internal medicine services (USA) | 68 teams of internal medicine attendings and housestaff | Microcomputer CPOE system with prices displayed (control group used written order sheets); 17 months | All orders | Chargemaster price | CPOE system listed cost‐effective tests for common problems and displayed reasonable test intervals |
| Tierney et al.[30] 1990 | Cluster RCT (randomized by clinic session) | Academic, outpatient, general medicine practice (USA) | 121 internal medicine attendings and housestaff | Microcomputer CPOE system with pop‐up window displaying price for current test and running total of cumulative test prices for current visit; 6 months | All lab and imaging tests | Chargemaster price | None |
| Everett et al.[31] 1983 | Pre‐post study with control group | Academic hospital, general internal medicine wards (USA) | Internal medicine attendings and housestaff (number NR) | Written order sheet with adjacent sheet of lab test prices; 3 months | Common lab tests | Chargemaster price | None |
Prices were displayed for laboratory tests (12 studies), imaging tests (8 studies), and medications (7 studies). Study scope ranged from examining a single medication class to evaluating all inpatient orders. The type of price used for the display varied, with the most common being the facility charges or chargemaster prices (6 studies), and Medicare prices (3 studies). In several cases, price display was only 1 component of the study, and 6 studies introduced additional interventions concurrent with price display, such as cost‐effective ordering menus,[29] medication comparison modules,[26] or display of test turnaround times.[14] Seven of the 19 studies were conducted in the past decade, of which 5 displayed prices within an EHR.[13, 14, 15, 16, 17]
Order Costs and Volume
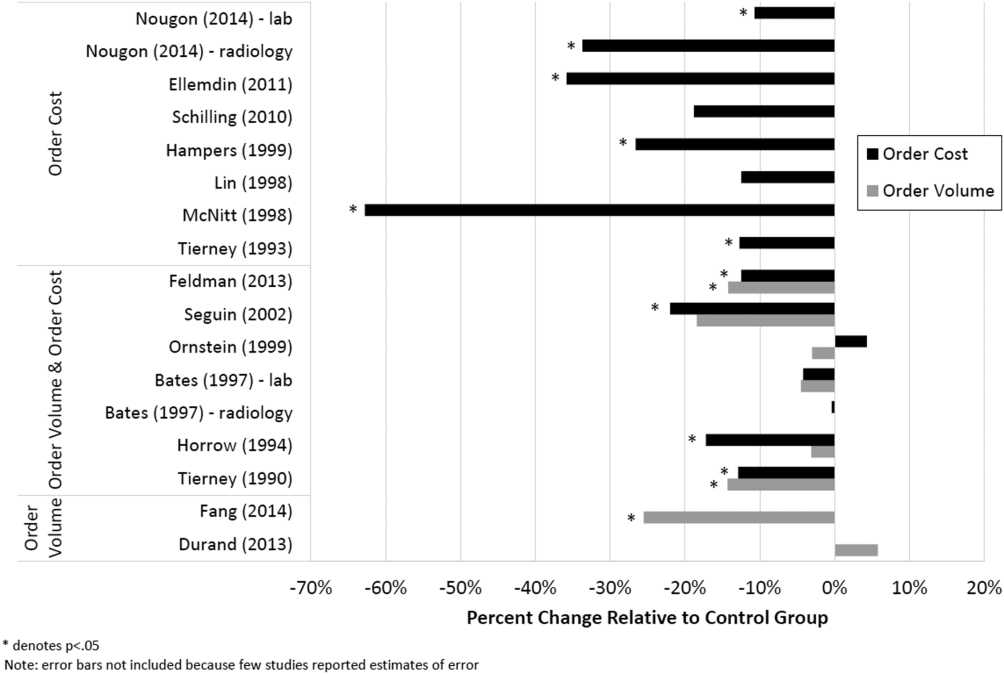
Thirteen studies reported the numeric impact of price display on aggregate order costs (Table 2). Nine of these demonstrated a statistically significant (P 0.05) decrease in order costs, with effect sizes ranging from 10.7% to 62.8%.[13, 16, 18, 20, 23, 24, 28, 29, 30] Decreases were found for lab costs, imaging costs, and medication costs, and were observed in both the inpatient and outpatient settings. Three of these 9 studies were randomized. For example, in 1 study randomizing 61 lab tests to price display or no price display, costs for the intervention labs dropped 9.6% compared to the year prior, whereas costs for control labs increased 2.9% (P 0.001).[16] Two studies randomized by provider group showed that providers seeing order prices accrued 12.7% fewer charges per inpatient admission (P = 0.02) and 12.9% fewer test charges per outpatient visit (P 0.05).[29, 30] Three studies found no significant association between price display and order costs, with effect sizes ranging from a decrease of 18.8% to an increase of 4.3%.[19, 22, 27] These studies also evaluated lab, imaging, and medication costs, and included 1 randomized trial. One additional large study noted a 12.5% decrease in medication costs after initiation of price display, but did not statistically evaluate this difference.[25]
| Study | No. of Encounters | Primary Outcome Measure(s) | Impact on Order Costs | Impact on Order Volume | ||||
|---|---|---|---|---|---|---|---|---|
| Control Group Outcome | Intervention Group Outcome | Relative Change | Control Group Outcome | Intervention Group Outcome | Relative Change | |||
| ||||||||
| Fang et al.[14] 2014 | 378,890 patient‐days | Reference lab orders per 1000 patient‐days | NR | NR | NA | 51 orders/1000 patient‐days | 38 orders/1000 patient‐days | 25.5% orders/1000 patient‐days (P 0.001) |
| Nougon et al.[13] 2015 | 2422 ED visits (excluding washout) | Lab and imaging test costs per ED visit | 7.1/visit (lab); 21.8/visit (imaging) | 6.4/visit (lab); 14.4/visit (imaging) | 10.7% lab costs/ visit (P = 0.02); 33.7% imaging costs/visit (P 0.001) | NR | NR | NA |
| Durand et al.[17] 2013 | NR | Imaging orders compared to baseline 1 year prior | NR | NR | NA | 3.0% total orders | +2.8% total orders | +5.8% total orders (P = 0.10) |
| Feldman et al.[16] 2013 | 245,758 patient‐days | Lab orders and fees per patient‐day compared to baseline 1 year prior | +2.9% fees/ patient‐day | 9.6% fees/ patient‐day | 12.5% fees/patient‐day (P 0.001) | +5.6% orders/patient‐day | 8.6% orders/ patient‐day | 14.2% orders/patient‐day (P 0.001) |
| Horn et al.[15] 2014 | NR | Lab test volume per patient visit, by individual lab test | NR | NR | NA | Aggregate data not reported | Aggregate data not reported | 5 of 27 tests had significant reduction in ordering (2.1% to 15.2%/patient visit) |
| Ellemdin et al.[18] 2011 | 897 admissions | Lab cost per hospital day | R442.90/day | R284.14/day | 35.8% lab costs/patient‐day (P = 0.001) | NR | NR | NA |
| Schilling[19] 2010 | 3222 ED visits | Combined lab and imaging test costs per ED visit | 108/visit | 88/visit | 18.8% test costs/visit (P = 0.07) | NR | NR | NA |
| Guterman et al.[21] 2002 | 168 urgent care visits | Percent of acid reducer prescriptions for ranitidine (the higher‐cost option) | NR | NR | NA | 49% ranitidine | 21% ranitidine | 57.1% ranitidine (P = 0.007) |
| Seguin et al.[20] 2002 | 287 SICU admissions | Tests ordered per admission; test costs per admission | 341/admission | 266/admission | 22.0% test costs/admission (P 0.05) | 13.6 tests/admission | 11.1 tests/ admission | 18.4% tests/admission (P = 0.12) |
| Hampers et al.[23] 1999 | 4881 ED visits (excluding washout) | Adjusted mean test charges per patient visit | $86.79/visit | $63.74/visit | 26.6% test charges/visit (P 0.01) | NR | NR | NA |
| Ornstein et al.[22] 1999 | 30,461 outpatient visits | Prescriptions per visit; prescription cost per visit; cost per prescription | $12.49/visit; $21.83/ prescription | $13.03/visit; $22.03/prescription |
+4.3% prescription costs/visit (P = 0.12); +0.9% cost/prescription (P = 0.61) |
0.66 prescriptions/visit | 0.64 prescriptions/ visit | 3.0% prescriptions/visit (P value not reported) |
| Lin et al.[25] 1998 | 40,747 surgical cases | Annual spending on muscle relaxants medication |
$378,234/year (20,389 cases) |
$330,923/year (20,358 cases) |
12.5% | NR | NR | NA |
| McNitt et al.[24] 1998 | 15,130 surgical cases | Anesthesia drug cost per case | $51.02/case | $18.99/case | 62.8% drug costs/case (P 0.05) | NR | NR | NA |
| Bates et al.[27] 1997 | 7090 admissions (lab); 17,381 admissions (imaging) | Tests ordered per admission; charges for tests ordered per admission |
$771/ admission (lab); $276/admission (imaging) |
$739/admission (lab); $275/admission (imaging) |
4.2% lab charges/admission (P = 0.97); 0.4% imaging charges/admission (P = 0.10) |
26.8 lab tests/admission; 1.76 imaging tests/admission |
25.6 lab tests/ admission; 1.76 imaging tests/ admission |
4.5% lab tests/admission (P = 0.74); 0% imaging tests/admission (P = 0.13) |
| Vedsted et al.[26] 1997 | NR | Prescribed daily doses per 1000 insured; total drug reimbursement per 1000 insured; reimbursement per daily dose | Reported graphically only | Reported graphically only | No difference | Reported graphically only | Reported graphically only | No difference |
| Horrow et al.[28] 1994 | NR | Anesthetic drugs used per week; anesthetic drug cost per week | $3837/week | $3179/week | 17.1% drug costs/week (P = 0.04) | 97 drugs/week | 94 drugs/week | 3.1% drugs/week (P = 0.56) |
| Tierney et al.[29] 1993 | 5219 admissions | Total charges per admission | $6964/admission | $6077/admission | 12.7% total charges/admission (P = 0.02) | NR | NR | NA |
| Tierney et al.[30] 1990 | 15,257 outpatient visits | Test orders per outpatient visit; test charges per outpatient visit | $51.81/visit | $45.13/visit | 12.9% test charges/visit (P 0.05) | 1.82 tests/visit | 1.56 tests/visit | 14.3% tests/visit (P 0.005) |
| Everett et al.[31] 1983 | NR | Lab tests per admission; charges per admission | NR | NR | NA | NR | NR | No statistically significant changes |
Eight studies reported the numeric impact of price display on aggregate order volume. Three of these demonstrated a statistically significant decrease in order volume, with effect sizes ranging from 14.2% to 25.5%.[14, 16, 30] Decreases were found for lab and imaging tests, and were observed in both inpatient and outpatient settings. For example, 1 pre‐post study displaying prices for inpatient send‐out lab tests demonstrated a 25.5% reduction in send‐out labs per 1000 patient‐days (P 0.001), whereas there was no change for the control group in‐house lab tests, for which prices were not shown.[14] The other 5 studies reported no significant association between price display and order volume, with effect sizes ranging from a decrease of 18.4% to an increase of 5.8%.[17, 20, 22, 27, 28] These studies evaluated lab, imaging, and medication volume. One trial randomizing by individual inpatient showed a nonsignificant decrease of 4.5% in lab orders per admission in the intervention group (P = 0.74), although the authors noted that their study had insufficient power to detect differences less than 10%.[27] Of note, 2 of the 5 studies reporting nonsignificant impacts on order volume (3.1%, P = 0.56; and 18.4%, P = 0.12) did demonstrate significant decreases in order costs (17.1%, P = 0.04; and 22.0%, P 0.05).[20, 28]
There were an additional 2 studies that reported the impact of price display on order volume for individual orders only. In 1 time‐series study showing lab test prices, there was a statistically significant decrease in order volume for 5 of 27 individual tests studied (using a Bonferroni‐adjusted threshold of significance), with no tests showing a significant increase.[15] In 1 pre‐post study showing prices for H2‐antagonist drugs, there was a statistically significant 57.1% decrease in order volume for the high‐cost medication, with a corresponding 58.7% increase in the low‐cost option.[21] These studies did not report impact on aggregate order costs. Two further studies in this review did not report outcomes numerically, but did state in their articles that significant impacts on order volume were not observed.[26, 31]
Therefore, of the 19 studies included in this review, 17 reported numeric results. Of these 17 studies, 12 showed that price display was associated with statistically significant decreases in either order costs or volume, either in aggregate (10 studies; Figure 1) or for individual orders (2 studies). Of the 7 studies conducted within the past decade, 5 noted significant decreases in order costs or volume. Prices were embedded into an EHR in 5 of these recent studies, and 4 of the 5 observed significant decreases in order costs or volume. Only 2 studies from the past decade1 from Belgium and 1 from the United Statesincorporated prices into an EHR and reported aggregate order costs. Both found statistically significant decreases in order costs with price display.[13, 16]
Patient Safety and Provider Acceptability
Five studies reported patient‐safety outcomes. One inpatient randomized trial showed similar rates of postdischarge utilization and charges between the intervention and control groups.[29] An outpatient randomized trial showed similar rates of hospital admissions, ED visits, and outpatient visits between the intervention and control groups.[30] Two pre‐post studies showing anesthesia prices in hospital operating rooms included a quality assurance review and showed no changes in adverse outcomes such as prolonged postoperative intubation, recovery room stay, or unplanned intensive care unit admissions.[24, 25] The only adverse safety finding was in a pre‐post study in a pediatric ED, which showed a higher rate of unscheduled follow‐up care during the intervention period compared to the control period (24.4% vs 17.8%, P 0.01) but similar rates of patients feeling better (83.4% vs 86.7%, P = 0.05). These findings, however, were based on self‐report during telephone follow‐up with a 47% response rate.[23]
Five studies reported on provider acceptability of price display. Two conducted questionnaires as part of the study plan, whereas the other 3 offered general provider feedback. One questionnaire revealed that 83% of practices were satisfied or very satisfied with the price display.[26] The other questionnaire found that 81% of physicians felt the price display improved my knowledge of the relative costs of tests I order and similarly 81% would like additional cost information displayed for other orders.[15] Three studies reported subjectively that showing prices initially caused questions from most physicians,[13] but that ultimately, physicians like seeing this information[27] and gave feedback that was generally positive.[21] One study evaluated the impact of price display on provider cost knowledge. Providers in the intervention group did not improve in their cost‐awareness, with average errors in cost estimates exceeding 40% even after 6 months of price display.[30]
Study Quality
Using a modified Downs and Black checklist of 21 items, studies in this review ranged in scores from 5 to 20, with a median score of 15. Studies most frequently lost points for being nonrandomized, failing to describe or adjust for potential confounders, being prone to historical confounding, or not evaluating potential adverse events.
We supplemented this modified Downs and Black checklist by reviewing 3 categories of study limitations not well‐reflected in the checklist scoring (Table 3). The first was potential for contamination between study groups, which was a concern in 4 studies. For example, 1 pre‐post study assessing medication ordering included clinical pharmacists in patient encounters both before and after the price display intervention.[22] This may have enhanced cost‐awareness even before prices were shown. The second set of limitations, present in 12 studies, included confounders that were not addressed by study design or analysis. For example, the intervention in 1 study displayed not just test cost but also test turnaround time, which may have separately influenced providers against ordering a particular test.[14] The third set of limitations included unanticipated gaps in the display of prices or in the collection of ordering data, which occurred in 5 studies. If studies did not report on gaps in the intervention or data collection, we assumed there were none.
| Study | Modified Downs & Black Score (Max Score 21) | Other Price Display Quality Criteria (Not Included in Downs & Black Score) | ||
|---|---|---|---|---|
| Potential for Contamination Between Study Groups | Potential Confounders of Results Not Addressed by Study Design or Analysis | Incomplete Price Display Intervention or Data Collection | ||
| ||||
| Fang et al.[14] 2014 | 14 | None | Concurrent display of test turnaround time may have independently contributed to decreased test ordering | 21% of reference lab orders were excluded from analysis because no price or turnaround‐time data were available |
| Nougon et al.[13] 2015 | 16 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Durand et al.[17] 2013 | 17 | Providers seeing test prices for intervention tests (including lab tests in concurrent Feldman study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, chest x‐ray) was not independent of control test ordering (eg, CT chest) | None |
| Feldman et al.[16] 2013 | 18 | Providers seeing test prices for intervention tests (including imaging tests in concurrent Durand study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, CMP) was not independent of control test ordering (eg, BMP) | None |
| Horn et al.[15] 2014 | 15 | None | None | None |
| Ellemdin et al.[18] 2011 | 15 | None | None | None |
| Schilling[19] 2010 | 12 | None | None | None |
| Guterman et al.[21] 2002 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Seguin et al.[20] 2002 | 17 | None | Because primary outcome was not adjusted for length of stay, the 30% shorter average length of stay during intervention period may have contributed to decreased costs per admission; historical confounding may have existed due to pre‐post study design without control group | None |
| Hampers et al.[23] 1999 | 17 | None | Requirement that physicians calculate total charges for each visit may have independently contributed to decreased test ordering; historical confounding may have existed due to pre‐post study design without control group | 10% of eligible patient visits were excluded from analysis because prices were not displayed or ordering data were not collected |
| Ornstein et al.[22] 1999 | 15 | Clinical pharmacists and pharmacy students involved in half of all patient contacts may have enhanced cost‐awareness during control period | Emergence of new drugs during intervention period and an ongoing quality improvement activity to increase prescribing of lipid‐lowering medications may have contributed to increased medication costs; historical confounding may have existed due to pre‐post study design without control group | 25% of prescription orders had no price displayed, and average prices were imputed for purposes of analysis |
| Lin et al.[25] 1998 | 12 | None | Emergence of new drug during intervention period and changes in several drug prices may have contributed to decreased order costs; historical confounding may have existed due to pre‐post study design without control group | None |
| McNitt et al.[24] 1998 | 15 | None | Intensive drug‐utilization review and cost‐reduction efforts may have independently contributed to decreased drug costs; historical confounding may have existed due to pre‐post study design without control group | None |
| Bates et al.[27] 1997 | 18 | Providers seeing test prices on intervention patients may have remembered prices or remained cost‐conscious when placing orders for control patients | None | 47% of lab tests and 26% of imaging tests were ordered manually outside of the trial's CPOE display system* |
| Vedsted et al.[26] 1997 | 5 | None | Medication price comparison module may have independently influenced physician ordering | None |
| Horrow et al.[28] 1994 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | Ordering data for 2 medications during 2 of 24 weeks were excluded from analysis due to internal inconsistency in the data |
| Tierney et al.[29] 1993 | 20 | None | Introduction of computerized order entry and menus for cost‐effective ordering may have independently contributed to decreased test ordering | None |
| Tierney et al.[30] 1990 | 20 | None | None | None |
| Everett et al.[31] 1983 | 7 | None | None | None |
Even among the 5 randomized trials there were substantial limitations. For example, 2 trials used individual tests as the unit of randomization, although ordering patterns for these tests are not independent of each other (eg, ordering rates for comprehensive metabolic panels are not independent of ordering rates for basic metabolic panels).[16, 17] This creates interference between units that was not accounted for in the analysis.[32] A third trial was randomized at the level of the patient, so was subject to contamination as providers seeing the price display for intervention group patients may have remained cost‐conscious while placing orders for control group patients.[27] In a fourth trial, the measured impact of the price display may have been confounded by other aspects of the overall cost intervention, which included cost‐effective test menus and suggestions for reasonable testing intervals.[29]
The highest‐quality study was a cluster‐randomized trial published in 1990 specifically measuring the effect of price display on a wide range of orders.[30] Providers and patients were separated by clinic session so as to avoid contamination between groups, and the trial included more than 15,000 outpatient visits. The intervention group providers ordered 14.3% fewer tests than control group providers, which resulted in 12.9% lower charges.
DISCUSSION
We identified 19 published reports of interventions that displayed real‐time order prices to providers and evaluated the impact on provider ordering. There was substantial heterogeneity in study setting, design, and quality. Although there is insufficient evidence on which to base strong conclusions, these studies collectively suggest that provider price display likely reduces order costs to a modest degree. Data on patient safety were largely lacking, although in the few studies that examined patient outcomes, there was little evidence that patient safety was adversely affected by the intervention. Providers widely viewed display of prices positively.
Our findings align with those of a recent systematic review that concluded that real‐time price information changed provider ordering in the majority of studies.[7] Whereas that review evaluated 17 studies from both clinical settings and simulations, our review focused exclusively on studies conducted in actual ordering environments. Additionally, our literature search yielded 8 studies not previously reviewed. We believe that the alignment of our findings with the prior review, despite the differences in studies included, adds validity to the conclusion that price display likely has a modest impact on reducing order costs. Our review contains several additions important for those considering price display interventions. We provide detailed information on study settings and intervention characteristics. We present a formal assessment of study quality to evaluate the strength of individual study findings and to guide future research in this area. Finally, because both patient safety and provider acceptability may be a concern when prices are shown, we describe all safety outcomes and provider feedback that these studies reported.
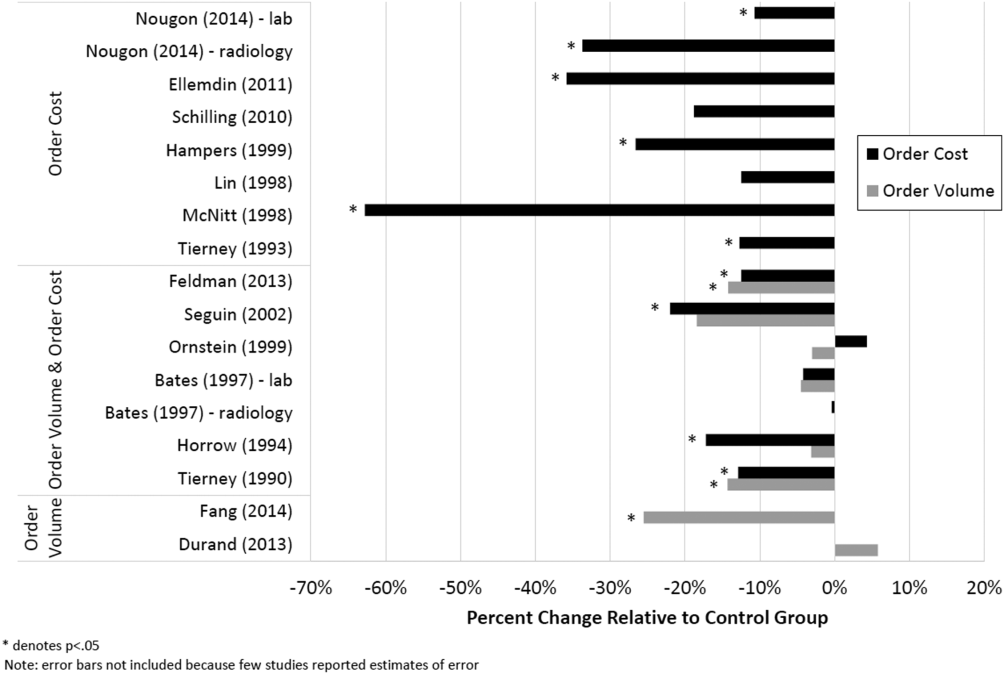
The largest effect sizes were noted in 5 studies reporting decreases in order volume or costs greater than 25%.[13, 14, 18, 23, 24] These were all pre‐post intervention studies, so the effect sizes may have been exaggerated by historical confounding. However, the 2 studies with concurrent control groups found no decreases in order volume or cost in the control group.[14, 18] Among the 5 studies that did not find a significant association between price display and provider ordering, 3 were subject to contamination between study groups,[17, 22, 27] 1 was underpowered,[19] and 1 noted a substantial effect size but did not perform a statistical analysis.[25] We also found that order costs were more frequently reduced than order volume, likely because shifts in ordering to less expensive alternatives may cause costs to decrease while volume remains unchanged.[20, 28]
If price display reduces order costs, as the majority of studies in this review indicate, this finding carries broad implications. Policy makers could promote cost‐conscious care by creating incentives for widespread adoption of price display. Hospital and health system leaders could improve transparency and reduce expenses by prioritizing price display. The specific beneficiaries of any reduced spending would depend on payment structures. With shifts toward financial risk‐bearing arrangements like accountable care organizations, healthcare institutions may have a financial interest in adopting price display. Because price display is an administrative intervention that can be developed within EHRs, it is potentially 1 of the most rapidly scalable strategies for reducing healthcare spending. Even modest reductions in spending on laboratory tests, imaging studies, and medications would result in substantial savings on a system‐wide basis.
Implementing price display does not come without challenges. Prices need to be calculated or obtained, loaded into an EHR system, and updated periodically. Technology innovators could enhance EHR software by making these processes easier. Healthcare institutions may find displaying relative prices (eg, $/$$/$$$) logistically simpler in some contexts than showing actual prices (eg, purchase cost), such as when contracts require prices to be confidential. Although we decided to exclude studies displaying relative prices, our search identified no studies that met other inclusion criteria but displayed relative prices, suggesting a lack of evidence regarding the impact of relative price display as an alternative to actual price display.
There are 4 key limitations to our review. First, the heterogeneity of the study designs and reported outcomes precluded pooling of data. The variety of clinical settings and mechanisms through which prices were displayed enhances the generalizability of our findings, but makes it difficult to identify particular contexts (eg, type of price or type of order) in which the intervention may be most effective. Second, although the presence of negative studies on this subject reduces the concern for reporting bias, it remains possible that sites willing to implement and study price displays may be inherently more sensitive to prices, such that published results might be more pronounced than if the intervention were widely implemented across multiple sites. Third, the mixed study quality limits the strength of conclusions that can be drawn. Several studies with both positive and negative findings had issues of bias, contamination, or confounding that make it difficult to be confident of the direction or magnitude of the main findings. Studies evaluating price display are challenging to conduct without these limitations, and that was apparent in our review. Finally, because over half of the studies were conducted over 15 years ago, it may limit their generalizability to modern ordering environments.
We believe there remains a need for high‐quality evidence on this subject within a contemporary context to confirm these findings. The optimal methodology for evaluating this intervention is a cluster randomized trial by facility or provider group, similar to that reported by Tierney et al. in 1990, with a primary outcome of aggregate order costs.[30] Given the substantial investment this would require, a large time series study could also be informative. As most prior price display interventions have been under 6 months in duration, it would be useful to know if the impact on order costs is sustained over a longer time period. The concurrent introduction of any EHR alerts that could impact ordering (eg, duplicate test warnings) should be simultaneously measured and reported. Studies also need to determine the impact of price display alone compared to price comparison displays (displaying prices for the selected order along with reasonable alternatives). Although price comparison was a component of the intervention in some of the studies in this review, it was not evaluated relative to price display alone. Furthermore, it would be helpful to know if the type of price displayed affects its impact. For instance, if providers are most sensitive to the absolute magnitude of prices, then displaying chargemaster prices may impact ordering more than showing hospital costs. If, however, relative prices are all that providers need, then showing lower numbers, such as Medicare prices or hospital costs, may be sufficient. Finally, it would be reassuring to have additional evidence that price display does not adversely impact patient outcomes.
Although some details need elucidation, the studies synthesized in this review provide valuable data in the current climate of increased emphasis on price transparency. Although substantial attention has been devoted by the academic community, technology start‐ups, private insurers, and even state legislatures to improving price transparency to patients, less focus has been given to physicians, for whom healthcare prices are often just as opaque.[4] The findings from this review suggest that provider price display may be an effective, safe, and acceptable approach to empower physicians to control healthcare spending.
Disclosures: Dr. Silvestri, Dr. Bongiovanni, and Ms. Glover have nothing to disclose. Dr. Gross reports grants from Johnson & Johnson, Medtronic Inc., and 21st Century Oncology during the conduct of this study. In addition, he received payment from Fair Health Inc. and ASTRO outside the submitted work.
- Institute of Medicine. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America: Washington, DC: National Academies Press; 2012.
- . Do physicians need a “shopping cart” for health care services? JAMA. 2012;307(8):791–792.
- . The disruptive innovation of price transparency in health care. JAMA. 2013;310(18):1927–1928.
- , . Providing price displays for physicians: which price is right? JAMA. 2014;312(16):1631–1632.
- , . Physician awareness of diagnostic and nondrug therapeutic costs: a systematic review. Int J Tech Assess Health Care. 2008;24(2):158–165.
- , , . Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4(9):e283.
- , , , . The effect of charge display on cost of care and physician practice behaviors: a systematic review. J Gen Intern Med. 2015;30:835–842.
- , , . Engaging medical librarians to improve the quality of review articles. JAMA. 2014;312(10):999–1000.
- , , . Influencing behavior of physicians ordering laboratory tests: a literature study. Med Care. 1993;31(9):784–794.
- , . Trials of providing costing information to general practitioners: a systematic review. Med J Aust. 1997;167(2):89–92.
- . A review of physician cost‐containment strategies for laboratory testing. Med Care. 1983;21(8):783–802.
- , . The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non‐randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377–384.
- , , , et al. Does offering pricing information to resident physicians in the emergency department potentially reduce laboratory and radiology costs? Eur J Emerg Med. 2015;22:247–252.
- , , , et al. Cost and turn‐around time display decreases inpatient ordering of reference laboratory tests: a time series. BMJ Qual Saf. 2014;23:994–1000.
- , , , , . The impact of cost displays on primary care physician laboratory test ordering. J Gen Intern Med. 2014;29:708–714.
- , , , et al. Impact of providing fee data on laboratory test ordering: a controlled clinical trial. JAMA Intern Med. 2013;173(10):903–908.
- , , , . Provider cost transparency alone has no impact on inpatient imaging utilization. J Am Coll Radiol. 2013;10(2):108–113.
- , , . Providing clinicians with information on laboratory test costs leads to reduction in hospital expenditure. S Afr Med J. 2011;101(10):746–748.
- . Cutting costs: the impact of price lists on the cost development at the emergency department. Eur J Emerg Med. 2010;17(6):337–339.
- , , , , . Effects of price information on test ordering in an intensive care unit. Intens Care Med. 2002;28(3):332–335.
- , , , , , . Modifying provider behavior: a low‐tech approach to pharmaceutical ordering. J Gen Intern Med. 2002;17(10):792–796.
- , , , , . Medication cost information in a computer‐based patient record system. Impact on prescribing in a family medicine clinical practice. Arch Fam Med. 1999;8(2):118–121.
- , , , , . The effect of price information on test‐ordering behavior and patient outcomes in a pediatric emergency department. Pediatrics. 1999;103(4 pt 2):877–882.
- , , . Long‐term pharmaceutical cost reduction using a data management system. Anesth Analg. 1998;87(4):837–842.
- , . The impact of price labeling of muscle relaxants on cost consciousness among anesthesiologists. J Clin Anesth. 1998;10(5):401–403.
- , , . Does a computerized price comparison module reduce prescribing costs in general practice? Fam Pract. 1997;14(3):199–203.
- , , , et al. Does the computerized display of charges affect inpatient ancillary test utilization? Arch Intern Med. 1997;157(21):2501–2508.
- , . Price stickers do not alter drug usage. Can J Anaesth. 1994;41(11):1047–1052.
- , , , . Physician inpatient order writing on microcomputer workstations. Effects on resource utilization. JAMA. 1993;269(3):379–383.
- , , . The effect on test ordering of informing physicians of the charges for outpatient diagnostic tests. N Engl J Med. 1990;322(21):1499–1504.
- , , , . Effect of cost education, cost audits, and faculty chart review on the use of laboratory services. Arch Intern Med. 1983;143(5):942–944.
- . Interference between units in randomized experiments. J Am Stat Assoc. 2007;102(477):191–200.
Rising healthcare spending has garnered significant public attention, and is considered a threat to other national priorities. Up to one‐third of national health expenditures are wasteful, the largest fraction generated through unnecessary services that could be substituted for less‐costly alternatives or omitted altogether.[1] Physicians play a central role in health spending, as they purchase nearly all tests and therapies on behalf of patients.
One strategy to enhance cost‐conscious physician ordering is to increase transparency of cost data for providers.[2, 3, 4] Although physicians consider price an important factor in ordering decisions, they have difficulty estimating costs accurately or finding price information easily.[5, 6] Improving physicians' knowledge of order costs may prompt them to forego diagnostic tests or therapies of low utility, or shift ordering to lower‐cost alternatives. Real‐time price display during provider order entry is 1 approach for achieving this goal. Modern electronic health records (EHRs) with computerized physician order entry (CPOE) make price display not only practical but also scalable. Integrating price display into clinical workflow, however, can be challenging, and there remains lack of clarity about potential risks and benefits. The dissemination of real‐time CPOE price display, therefore, requires an understanding of its impact on clinical care.
Over the past 3 decades, several studies in the medical literature have evaluated the effect of price display on physician ordering behavior. To date, however, there has been only 1 narrative review of this literature, which did not include several recent studies on the topic or formally address study quality and physician acceptance of price display modules.[7] Therefore, to help inform healthcare leaders, technology innovators, and policy makers, we conducted a systematic review to address 4 key questions: (1) What are the characteristics of interventions that have displayed order prices to physicians in the context of actual practice? (2) To what degree does real‐time display of order prices impact order costs and order volume? (3) Does price display impact patient safety outcomes, and is it acceptable to providers? (4) What is the quality of the current literature on this topic?
METHODS
Data Sources
We searched 2 electronic databases, MEDLINE and Embase, using a combination of controlled vocabulary terms and keywords that covered both the targeted intervention (eg, fees and charges) and the outcome of interest (eg, physician's practice patterns), limited to English language articles with no restriction on country or year of publication (see Supporting Information, Appendix 1, in the online version of this article). The search was run through August 2014. Results from both database searches were combined and duplicates eliminated. We also ran a MEDLINE keyword search on titles and abstracts of articles from 2014 that were not yet indexed. A medical librarian was involved in all aspects of the search process.[8]
Study Selection
Studies were included if they evaluated the effect of displaying actual order prices to providers during the ordering process and reported the impact on provider ordering practices. Reports in any clinical context and with any study design were included. To assess most accurately the effect of price display on real‐life ordering and patient outcomes, studies were excluded if: (1) they were review articles, commentaries, or editorials; (2) they did not show order prices to providers; (3) the context was a simulation; (4) the prices displayed were relative (eg, $/$$/$$$) or were only cumulative; (5) prices were not presented real‐time during the ordering process; or (6) the primary outcome was neither order costs nor order volume. We decided a priori to exclude simulations because these may not accurately reflect provider behavior when treating real patients, and to exclude studies showing relative prices due to concerns that it is a less significant price transparency intervention and that providers may interpret relative prices differently from actual prices.
Two reviewers, both physicians and health service researchers (M.T.S. and T.R.B.), separately reviewed the full list of titles and abstracts. For studies that potentially met inclusion criteria, full articles were obtained and were independently read for inclusion in the final review. The references of all included studies were searched manually, and the Scopus database was used to search all studies that cited the included studies. We also searched the references of relevant literature reviews.[9, 10, 11] Articles of interest discovered through manual search were then subjected to the same process.
Data Extraction and Quality Assessment
Two reviewers (M.T.S. and T.R.B.) independently performed data extraction using a standardized spreadsheet. Discrepancies were resolved by reviewer consensus. Extracted study characteristics included study design and duration, clinical setting, study size, type of orders involved, characteristics of price display intervention and control, and type of outcome. Findings regarding patient safety and provider acceptability were also extracted when available.
Study quality was independently evaluated and scored by both reviewers using the Downs and Black checklist, designed to assess quality of both randomized and nonrandomized studies.[12] The checklist contains 5 items pertaining to allocation concealment, blinding, or follow‐up that are not applicable to an administrative intervention like price display, so these questions were excluded. Additionally, few studies calculated sample size or reported post hoc statistical power, so we also excluded this question, leaving a modified 21‐item checklist. We also assessed each study for sources of bias that were not already assessed by the Downs and Black checklist, including contamination between study groups, confounding of results, and incomplete intervention or data collection.
Data Synthesis
Data are reported in tabular form for all included studies. Due to heterogeneity of study designs and outcome measures, data from the studies were not pooled quantitatively. This review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis guidelines.
RESULTS
Database searches yielded a total of 1400 articles, of which 18 were selected on the basis of title and abstract for detailed assessment. Reference searching led us to retrieve 94 further studies of possible interest, of which 23 were selected on the basis of abstract for detailed assessment. Thus, 41 publications underwent full manuscript review, 19 of which met all inclusion criteria (see Supporting Information, Appendix 2, in the online version of this article).[13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31] These studies were published between 1983 and 2014, and were conducted primarily in the United States.
Study Characteristics
There was considerable heterogeneity among the 19 studies with regard to design, setting, and scope (Table 1). There were 5 randomized trials, for which the units of randomization were patient (1), provider team (2), and test (2). There were 13 pre‐post intervention studies, 5 of which used a concomitant control group, and 2 of which included a washout period. There was 1 interrupted time series study. Studies were conducted within inpatient hospital floors (8), outpatient clinics (4), emergency departments (ED) or urgent care facilities (4), and hospital operating rooms (3).
| Study | Design | Clinical Setting | Providers | Intervention and Duration | Order(s) Studied | Type of Price Displayed | Concurrent Interventions |
|---|---|---|---|---|---|---|---|
| |||||||
| Fang et al.[14] 2014 | Pre‐post study with control group | Academic hospital (USA) | All inpatient ordering providers | CPOE system with prices displayed for reference lab tests; 8 months | All send‐out lab tests | Charge from send‐out laboratory, displayed as range (eg, $100300) | Display also contained expected lab turnaround time |
| Nougon et al.[13] 2014 | Pre‐post study with washout | Academic adult emergency department (Belgium) | 9 ED house staff | CPOE system with prices displayed on common orders form, and price list displayed above all workstations and in patient rooms; 2 months | Common lab and imaging tests | Reference costs from Belgian National Institute for Health Insurance and Invalidity | None |
| Durand et al.[17] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 10 common imaging tests | Medicare allowable fee | None |
| Feldman et al.[16] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 61 lab tests | Medicare allowable fee | None |
| Horn et al.[15] 2014 | Interrupted time series study with control group | Private outpatient group practice alliance (USA) | 215 primary care physicians | CPOE system with prices displayed; 6 months | 27 lab tests | Medicare allowable fee, displayed as narrow range (eg, $5$10) | None |
| Ellemdin et al.[18] 2011 | Pre‐post study with control group | Academic hospital, internal medicine units (South Africa) | Internal medicine physicians (number NR) | Sheet with lab test costs given to intervention group physicians who were required to write out cost for each order; 4 months | Common lab tests | Not reported | None |
| Schilling,[19] 2010 | Pre‐post study with control group | Academic adult emergency department (Sweden) | All internal medicine physicians in ED | Standard provider workstations with price lists posted on each; 2 months | 91 common lab tests, 39 common imaging tests | Not reported | None |
| Guterman et al.[21] 2002 | Pre‐post study | Academic‐affiliated urgent care clinic (USA) | 51 attendings and housestaff | Preformatted paper prescription form with medication prices displayed; 2 weeks | 2 H2‐blocker medications | Acquisition cost of medication plus fill fee | None |
| Seguin et al.[20] 2002 | Pre‐post study | Academic surgical intensive care unit (France) | All intensive care unit physicians | Paper quick‐order checklist with prices displayed; 2 months | 6 common lab tests, 1 imaging test | Not reported | None |
| Hampers et al.[23] 1999 | Pre‐post study with washout | Academic pediatric emergency department (USA) | Pediatric ED attendings and housestaff (number NR) | Paper common‐order checklist with prices displayed; 3 months | 22 common lab and imaging tests | Chargemaster price* | Physicians required to calculate total charges for diagnostic workup |
| Ornstein et al.[22] 1999 | Pre‐post study | Academic family medicine outpatient clinic (USA) | 46 attendings and housestaff | Microcomputer CPOE system with medication prices displayed; 6 months | All medications | AWP for total supply (acute medications) or 30‐day supply (chronic medications) | Additional keystroke produced list of less costly alternative medications |
| Lin et al.[25] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | All anesthesia providers | Standard muscle relaxant drug vials with price stickers displayed; 12 months | All muscle relaxant medications | Not reported | None |
| McNitt et al.[24] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | 90 anesthesia attendings, housestaff and anesthetists | List of drug costs displayed in operating rooms, anesthesia lounge, and anesthesia satellite pharmacy; 10 months | 22 common anesthesia medications | Hospital acquisition cost | Regular anesthesia department reviews of drug usage and cost |
| Bates et al.[27] 1997 | RCT (randomized by patient) | Academic hospital, medical and surgical inpatients (USA) | All inpatient ordering providers | CPOE system with display of test price and running total of prices for the ordering session; 4 months (lab) and 7 months (imaging) | All lab tests, 35 common imaging tests | Chargemaster price | None |
| Vedsted et al.[26] 1997 | Pre‐post study with control group | Outpatient general practices (Denmark) | 231 general practitioners | In practices already using APEX CPOE system, introduction of medication price display (control practices used non‐APEX computer system or paper‐based prescribing); 12 months | All medications | Chargemaster price | Medication price comparison module (stars indicated availability of cheaper option) |
| Horrow et al.[28] 1994 | Pre‐post study | Private tertiary care hospital operating rooms (USA) | 56 anesthesia attendings, housestaff and anesthetists | Standard anesthesia drug vials and syringes with supermarket price stickers displayed; 3 months | 13 neuromuscular relaxant and sedative‐hypnotic medications | Hospital acquisition cost | None |
| Tierney et al.[29] 1993 | Cluster RCT (randomized by provider team) | Public hospital, internal medicine services (USA) | 68 teams of internal medicine attendings and housestaff | Microcomputer CPOE system with prices displayed (control group used written order sheets); 17 months | All orders | Chargemaster price | CPOE system listed cost‐effective tests for common problems and displayed reasonable test intervals |
| Tierney et al.[30] 1990 | Cluster RCT (randomized by clinic session) | Academic, outpatient, general medicine practice (USA) | 121 internal medicine attendings and housestaff | Microcomputer CPOE system with pop‐up window displaying price for current test and running total of cumulative test prices for current visit; 6 months | All lab and imaging tests | Chargemaster price | None |
| Everett et al.[31] 1983 | Pre‐post study with control group | Academic hospital, general internal medicine wards (USA) | Internal medicine attendings and housestaff (number NR) | Written order sheet with adjacent sheet of lab test prices; 3 months | Common lab tests | Chargemaster price | None |
Prices were displayed for laboratory tests (12 studies), imaging tests (8 studies), and medications (7 studies). Study scope ranged from examining a single medication class to evaluating all inpatient orders. The type of price used for the display varied, with the most common being the facility charges or chargemaster prices (6 studies), and Medicare prices (3 studies). In several cases, price display was only 1 component of the study, and 6 studies introduced additional interventions concurrent with price display, such as cost‐effective ordering menus,[29] medication comparison modules,[26] or display of test turnaround times.[14] Seven of the 19 studies were conducted in the past decade, of which 5 displayed prices within an EHR.[13, 14, 15, 16, 17]
Order Costs and Volume
Thirteen studies reported the numeric impact of price display on aggregate order costs (Table 2). Nine of these demonstrated a statistically significant (P 0.05) decrease in order costs, with effect sizes ranging from 10.7% to 62.8%.[13, 16, 18, 20, 23, 24, 28, 29, 30] Decreases were found for lab costs, imaging costs, and medication costs, and were observed in both the inpatient and outpatient settings. Three of these 9 studies were randomized. For example, in 1 study randomizing 61 lab tests to price display or no price display, costs for the intervention labs dropped 9.6% compared to the year prior, whereas costs for control labs increased 2.9% (P 0.001).[16] Two studies randomized by provider group showed that providers seeing order prices accrued 12.7% fewer charges per inpatient admission (P = 0.02) and 12.9% fewer test charges per outpatient visit (P 0.05).[29, 30] Three studies found no significant association between price display and order costs, with effect sizes ranging from a decrease of 18.8% to an increase of 4.3%.[19, 22, 27] These studies also evaluated lab, imaging, and medication costs, and included 1 randomized trial. One additional large study noted a 12.5% decrease in medication costs after initiation of price display, but did not statistically evaluate this difference.[25]
| Study | No. of Encounters | Primary Outcome Measure(s) | Impact on Order Costs | Impact on Order Volume | ||||
|---|---|---|---|---|---|---|---|---|
| Control Group Outcome | Intervention Group Outcome | Relative Change | Control Group Outcome | Intervention Group Outcome | Relative Change | |||
| ||||||||
| Fang et al.[14] 2014 | 378,890 patient‐days | Reference lab orders per 1000 patient‐days | NR | NR | NA | 51 orders/1000 patient‐days | 38 orders/1000 patient‐days | 25.5% orders/1000 patient‐days (P 0.001) |
| Nougon et al.[13] 2015 | 2422 ED visits (excluding washout) | Lab and imaging test costs per ED visit | 7.1/visit (lab); 21.8/visit (imaging) | 6.4/visit (lab); 14.4/visit (imaging) | 10.7% lab costs/ visit (P = 0.02); 33.7% imaging costs/visit (P 0.001) | NR | NR | NA |
| Durand et al.[17] 2013 | NR | Imaging orders compared to baseline 1 year prior | NR | NR | NA | 3.0% total orders | +2.8% total orders | +5.8% total orders (P = 0.10) |
| Feldman et al.[16] 2013 | 245,758 patient‐days | Lab orders and fees per patient‐day compared to baseline 1 year prior | +2.9% fees/ patient‐day | 9.6% fees/ patient‐day | 12.5% fees/patient‐day (P 0.001) | +5.6% orders/patient‐day | 8.6% orders/ patient‐day | 14.2% orders/patient‐day (P 0.001) |
| Horn et al.[15] 2014 | NR | Lab test volume per patient visit, by individual lab test | NR | NR | NA | Aggregate data not reported | Aggregate data not reported | 5 of 27 tests had significant reduction in ordering (2.1% to 15.2%/patient visit) |
| Ellemdin et al.[18] 2011 | 897 admissions | Lab cost per hospital day | R442.90/day | R284.14/day | 35.8% lab costs/patient‐day (P = 0.001) | NR | NR | NA |
| Schilling[19] 2010 | 3222 ED visits | Combined lab and imaging test costs per ED visit | 108/visit | 88/visit | 18.8% test costs/visit (P = 0.07) | NR | NR | NA |
| Guterman et al.[21] 2002 | 168 urgent care visits | Percent of acid reducer prescriptions for ranitidine (the higher‐cost option) | NR | NR | NA | 49% ranitidine | 21% ranitidine | 57.1% ranitidine (P = 0.007) |
| Seguin et al.[20] 2002 | 287 SICU admissions | Tests ordered per admission; test costs per admission | 341/admission | 266/admission | 22.0% test costs/admission (P 0.05) | 13.6 tests/admission | 11.1 tests/ admission | 18.4% tests/admission (P = 0.12) |
| Hampers et al.[23] 1999 | 4881 ED visits (excluding washout) | Adjusted mean test charges per patient visit | $86.79/visit | $63.74/visit | 26.6% test charges/visit (P 0.01) | NR | NR | NA |
| Ornstein et al.[22] 1999 | 30,461 outpatient visits | Prescriptions per visit; prescription cost per visit; cost per prescription | $12.49/visit; $21.83/ prescription | $13.03/visit; $22.03/prescription |
+4.3% prescription costs/visit (P = 0.12); +0.9% cost/prescription (P = 0.61) |
0.66 prescriptions/visit | 0.64 prescriptions/ visit | 3.0% prescriptions/visit (P value not reported) |
| Lin et al.[25] 1998 | 40,747 surgical cases | Annual spending on muscle relaxants medication |
$378,234/year (20,389 cases) |
$330,923/year (20,358 cases) |
12.5% | NR | NR | NA |
| McNitt et al.[24] 1998 | 15,130 surgical cases | Anesthesia drug cost per case | $51.02/case | $18.99/case | 62.8% drug costs/case (P 0.05) | NR | NR | NA |
| Bates et al.[27] 1997 | 7090 admissions (lab); 17,381 admissions (imaging) | Tests ordered per admission; charges for tests ordered per admission |
$771/ admission (lab); $276/admission (imaging) |
$739/admission (lab); $275/admission (imaging) |
4.2% lab charges/admission (P = 0.97); 0.4% imaging charges/admission (P = 0.10) |
26.8 lab tests/admission; 1.76 imaging tests/admission |
25.6 lab tests/ admission; 1.76 imaging tests/ admission |
4.5% lab tests/admission (P = 0.74); 0% imaging tests/admission (P = 0.13) |
| Vedsted et al.[26] 1997 | NR | Prescribed daily doses per 1000 insured; total drug reimbursement per 1000 insured; reimbursement per daily dose | Reported graphically only | Reported graphically only | No difference | Reported graphically only | Reported graphically only | No difference |
| Horrow et al.[28] 1994 | NR | Anesthetic drugs used per week; anesthetic drug cost per week | $3837/week | $3179/week | 17.1% drug costs/week (P = 0.04) | 97 drugs/week | 94 drugs/week | 3.1% drugs/week (P = 0.56) |
| Tierney et al.[29] 1993 | 5219 admissions | Total charges per admission | $6964/admission | $6077/admission | 12.7% total charges/admission (P = 0.02) | NR | NR | NA |
| Tierney et al.[30] 1990 | 15,257 outpatient visits | Test orders per outpatient visit; test charges per outpatient visit | $51.81/visit | $45.13/visit | 12.9% test charges/visit (P 0.05) | 1.82 tests/visit | 1.56 tests/visit | 14.3% tests/visit (P 0.005) |
| Everett et al.[31] 1983 | NR | Lab tests per admission; charges per admission | NR | NR | NA | NR | NR | No statistically significant changes |
Eight studies reported the numeric impact of price display on aggregate order volume. Three of these demonstrated a statistically significant decrease in order volume, with effect sizes ranging from 14.2% to 25.5%.[14, 16, 30] Decreases were found for lab and imaging tests, and were observed in both inpatient and outpatient settings. For example, 1 pre‐post study displaying prices for inpatient send‐out lab tests demonstrated a 25.5% reduction in send‐out labs per 1000 patient‐days (P 0.001), whereas there was no change for the control group in‐house lab tests, for which prices were not shown.[14] The other 5 studies reported no significant association between price display and order volume, with effect sizes ranging from a decrease of 18.4% to an increase of 5.8%.[17, 20, 22, 27, 28] These studies evaluated lab, imaging, and medication volume. One trial randomizing by individual inpatient showed a nonsignificant decrease of 4.5% in lab orders per admission in the intervention group (P = 0.74), although the authors noted that their study had insufficient power to detect differences less than 10%.[27] Of note, 2 of the 5 studies reporting nonsignificant impacts on order volume (3.1%, P = 0.56; and 18.4%, P = 0.12) did demonstrate significant decreases in order costs (17.1%, P = 0.04; and 22.0%, P 0.05).[20, 28]
There were an additional 2 studies that reported the impact of price display on order volume for individual orders only. In 1 time‐series study showing lab test prices, there was a statistically significant decrease in order volume for 5 of 27 individual tests studied (using a Bonferroni‐adjusted threshold of significance), with no tests showing a significant increase.[15] In 1 pre‐post study showing prices for H2‐antagonist drugs, there was a statistically significant 57.1% decrease in order volume for the high‐cost medication, with a corresponding 58.7% increase in the low‐cost option.[21] These studies did not report impact on aggregate order costs. Two further studies in this review did not report outcomes numerically, but did state in their articles that significant impacts on order volume were not observed.[26, 31]
Therefore, of the 19 studies included in this review, 17 reported numeric results. Of these 17 studies, 12 showed that price display was associated with statistically significant decreases in either order costs or volume, either in aggregate (10 studies; Figure 1) or for individual orders (2 studies). Of the 7 studies conducted within the past decade, 5 noted significant decreases in order costs or volume. Prices were embedded into an EHR in 5 of these recent studies, and 4 of the 5 observed significant decreases in order costs or volume. Only 2 studies from the past decade1 from Belgium and 1 from the United Statesincorporated prices into an EHR and reported aggregate order costs. Both found statistically significant decreases in order costs with price display.[13, 16]

Patient Safety and Provider Acceptability
Five studies reported patient‐safety outcomes. One inpatient randomized trial showed similar rates of postdischarge utilization and charges between the intervention and control groups.[29] An outpatient randomized trial showed similar rates of hospital admissions, ED visits, and outpatient visits between the intervention and control groups.[30] Two pre‐post studies showing anesthesia prices in hospital operating rooms included a quality assurance review and showed no changes in adverse outcomes such as prolonged postoperative intubation, recovery room stay, or unplanned intensive care unit admissions.[24, 25] The only adverse safety finding was in a pre‐post study in a pediatric ED, which showed a higher rate of unscheduled follow‐up care during the intervention period compared to the control period (24.4% vs 17.8%, P 0.01) but similar rates of patients feeling better (83.4% vs 86.7%, P = 0.05). These findings, however, were based on self‐report during telephone follow‐up with a 47% response rate.[23]
Five studies reported on provider acceptability of price display. Two conducted questionnaires as part of the study plan, whereas the other 3 offered general provider feedback. One questionnaire revealed that 83% of practices were satisfied or very satisfied with the price display.[26] The other questionnaire found that 81% of physicians felt the price display improved my knowledge of the relative costs of tests I order and similarly 81% would like additional cost information displayed for other orders.[15] Three studies reported subjectively that showing prices initially caused questions from most physicians,[13] but that ultimately, physicians like seeing this information[27] and gave feedback that was generally positive.[21] One study evaluated the impact of price display on provider cost knowledge. Providers in the intervention group did not improve in their cost‐awareness, with average errors in cost estimates exceeding 40% even after 6 months of price display.[30]
Study Quality
Using a modified Downs and Black checklist of 21 items, studies in this review ranged in scores from 5 to 20, with a median score of 15. Studies most frequently lost points for being nonrandomized, failing to describe or adjust for potential confounders, being prone to historical confounding, or not evaluating potential adverse events.
We supplemented this modified Downs and Black checklist by reviewing 3 categories of study limitations not well‐reflected in the checklist scoring (Table 3). The first was potential for contamination between study groups, which was a concern in 4 studies. For example, 1 pre‐post study assessing medication ordering included clinical pharmacists in patient encounters both before and after the price display intervention.[22] This may have enhanced cost‐awareness even before prices were shown. The second set of limitations, present in 12 studies, included confounders that were not addressed by study design or analysis. For example, the intervention in 1 study displayed not just test cost but also test turnaround time, which may have separately influenced providers against ordering a particular test.[14] The third set of limitations included unanticipated gaps in the display of prices or in the collection of ordering data, which occurred in 5 studies. If studies did not report on gaps in the intervention or data collection, we assumed there were none.
| Study | Modified Downs & Black Score (Max Score 21) | Other Price Display Quality Criteria (Not Included in Downs & Black Score) | ||
|---|---|---|---|---|
| Potential for Contamination Between Study Groups | Potential Confounders of Results Not Addressed by Study Design or Analysis | Incomplete Price Display Intervention or Data Collection | ||
| ||||
| Fang et al.[14] 2014 | 14 | None | Concurrent display of test turnaround time may have independently contributed to decreased test ordering | 21% of reference lab orders were excluded from analysis because no price or turnaround‐time data were available |
| Nougon et al.[13] 2015 | 16 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Durand et al.[17] 2013 | 17 | Providers seeing test prices for intervention tests (including lab tests in concurrent Feldman study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, chest x‐ray) was not independent of control test ordering (eg, CT chest) | None |
| Feldman et al.[16] 2013 | 18 | Providers seeing test prices for intervention tests (including imaging tests in concurrent Durand study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, CMP) was not independent of control test ordering (eg, BMP) | None |
| Horn et al.[15] 2014 | 15 | None | None | None |
| Ellemdin et al.[18] 2011 | 15 | None | None | None |
| Schilling[19] 2010 | 12 | None | None | None |
| Guterman et al.[21] 2002 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Seguin et al.[20] 2002 | 17 | None | Because primary outcome was not adjusted for length of stay, the 30% shorter average length of stay during intervention period may have contributed to decreased costs per admission; historical confounding may have existed due to pre‐post study design without control group | None |
| Hampers et al.[23] 1999 | 17 | None | Requirement that physicians calculate total charges for each visit may have independently contributed to decreased test ordering; historical confounding may have existed due to pre‐post study design without control group | 10% of eligible patient visits were excluded from analysis because prices were not displayed or ordering data were not collected |
| Ornstein et al.[22] 1999 | 15 | Clinical pharmacists and pharmacy students involved in half of all patient contacts may have enhanced cost‐awareness during control period | Emergence of new drugs during intervention period and an ongoing quality improvement activity to increase prescribing of lipid‐lowering medications may have contributed to increased medication costs; historical confounding may have existed due to pre‐post study design without control group | 25% of prescription orders had no price displayed, and average prices were imputed for purposes of analysis |
| Lin et al.[25] 1998 | 12 | None | Emergence of new drug during intervention period and changes in several drug prices may have contributed to decreased order costs; historical confounding may have existed due to pre‐post study design without control group | None |
| McNitt et al.[24] 1998 | 15 | None | Intensive drug‐utilization review and cost‐reduction efforts may have independently contributed to decreased drug costs; historical confounding may have existed due to pre‐post study design without control group | None |
| Bates et al.[27] 1997 | 18 | Providers seeing test prices on intervention patients may have remembered prices or remained cost‐conscious when placing orders for control patients | None | 47% of lab tests and 26% of imaging tests were ordered manually outside of the trial's CPOE display system* |
| Vedsted et al.[26] 1997 | 5 | None | Medication price comparison module may have independently influenced physician ordering | None |
| Horrow et al.[28] 1994 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | Ordering data for 2 medications during 2 of 24 weeks were excluded from analysis due to internal inconsistency in the data |
| Tierney et al.[29] 1993 | 20 | None | Introduction of computerized order entry and menus for cost‐effective ordering may have independently contributed to decreased test ordering | None |
| Tierney et al.[30] 1990 | 20 | None | None | None |
| Everett et al.[31] 1983 | 7 | None | None | None |
Even among the 5 randomized trials there were substantial limitations. For example, 2 trials used individual tests as the unit of randomization, although ordering patterns for these tests are not independent of each other (eg, ordering rates for comprehensive metabolic panels are not independent of ordering rates for basic metabolic panels).[16, 17] This creates interference between units that was not accounted for in the analysis.[32] A third trial was randomized at the level of the patient, so was subject to contamination as providers seeing the price display for intervention group patients may have remained cost‐conscious while placing orders for control group patients.[27] In a fourth trial, the measured impact of the price display may have been confounded by other aspects of the overall cost intervention, which included cost‐effective test menus and suggestions for reasonable testing intervals.[29]
The highest‐quality study was a cluster‐randomized trial published in 1990 specifically measuring the effect of price display on a wide range of orders.[30] Providers and patients were separated by clinic session so as to avoid contamination between groups, and the trial included more than 15,000 outpatient visits. The intervention group providers ordered 14.3% fewer tests than control group providers, which resulted in 12.9% lower charges.
DISCUSSION
We identified 19 published reports of interventions that displayed real‐time order prices to providers and evaluated the impact on provider ordering. There was substantial heterogeneity in study setting, design, and quality. Although there is insufficient evidence on which to base strong conclusions, these studies collectively suggest that provider price display likely reduces order costs to a modest degree. Data on patient safety were largely lacking, although in the few studies that examined patient outcomes, there was little evidence that patient safety was adversely affected by the intervention. Providers widely viewed display of prices positively.
Our findings align with those of a recent systematic review that concluded that real‐time price information changed provider ordering in the majority of studies.[7] Whereas that review evaluated 17 studies from both clinical settings and simulations, our review focused exclusively on studies conducted in actual ordering environments. Additionally, our literature search yielded 8 studies not previously reviewed. We believe that the alignment of our findings with the prior review, despite the differences in studies included, adds validity to the conclusion that price display likely has a modest impact on reducing order costs. Our review contains several additions important for those considering price display interventions. We provide detailed information on study settings and intervention characteristics. We present a formal assessment of study quality to evaluate the strength of individual study findings and to guide future research in this area. Finally, because both patient safety and provider acceptability may be a concern when prices are shown, we describe all safety outcomes and provider feedback that these studies reported.
The largest effect sizes were noted in 5 studies reporting decreases in order volume or costs greater than 25%.[13, 14, 18, 23, 24] These were all pre‐post intervention studies, so the effect sizes may have been exaggerated by historical confounding. However, the 2 studies with concurrent control groups found no decreases in order volume or cost in the control group.[14, 18] Among the 5 studies that did not find a significant association between price display and provider ordering, 3 were subject to contamination between study groups,[17, 22, 27] 1 was underpowered,[19] and 1 noted a substantial effect size but did not perform a statistical analysis.[25] We also found that order costs were more frequently reduced than order volume, likely because shifts in ordering to less expensive alternatives may cause costs to decrease while volume remains unchanged.[20, 28]
If price display reduces order costs, as the majority of studies in this review indicate, this finding carries broad implications. Policy makers could promote cost‐conscious care by creating incentives for widespread adoption of price display. Hospital and health system leaders could improve transparency and reduce expenses by prioritizing price display. The specific beneficiaries of any reduced spending would depend on payment structures. With shifts toward financial risk‐bearing arrangements like accountable care organizations, healthcare institutions may have a financial interest in adopting price display. Because price display is an administrative intervention that can be developed within EHRs, it is potentially 1 of the most rapidly scalable strategies for reducing healthcare spending. Even modest reductions in spending on laboratory tests, imaging studies, and medications would result in substantial savings on a system‐wide basis.
Implementing price display does not come without challenges. Prices need to be calculated or obtained, loaded into an EHR system, and updated periodically. Technology innovators could enhance EHR software by making these processes easier. Healthcare institutions may find displaying relative prices (eg, $/$$/$$$) logistically simpler in some contexts than showing actual prices (eg, purchase cost), such as when contracts require prices to be confidential. Although we decided to exclude studies displaying relative prices, our search identified no studies that met other inclusion criteria but displayed relative prices, suggesting a lack of evidence regarding the impact of relative price display as an alternative to actual price display.
There are 4 key limitations to our review. First, the heterogeneity of the study designs and reported outcomes precluded pooling of data. The variety of clinical settings and mechanisms through which prices were displayed enhances the generalizability of our findings, but makes it difficult to identify particular contexts (eg, type of price or type of order) in which the intervention may be most effective. Second, although the presence of negative studies on this subject reduces the concern for reporting bias, it remains possible that sites willing to implement and study price displays may be inherently more sensitive to prices, such that published results might be more pronounced than if the intervention were widely implemented across multiple sites. Third, the mixed study quality limits the strength of conclusions that can be drawn. Several studies with both positive and negative findings had issues of bias, contamination, or confounding that make it difficult to be confident of the direction or magnitude of the main findings. Studies evaluating price display are challenging to conduct without these limitations, and that was apparent in our review. Finally, because over half of the studies were conducted over 15 years ago, it may limit their generalizability to modern ordering environments.
We believe there remains a need for high‐quality evidence on this subject within a contemporary context to confirm these findings. The optimal methodology for evaluating this intervention is a cluster randomized trial by facility or provider group, similar to that reported by Tierney et al. in 1990, with a primary outcome of aggregate order costs.[30] Given the substantial investment this would require, a large time series study could also be informative. As most prior price display interventions have been under 6 months in duration, it would be useful to know if the impact on order costs is sustained over a longer time period. The concurrent introduction of any EHR alerts that could impact ordering (eg, duplicate test warnings) should be simultaneously measured and reported. Studies also need to determine the impact of price display alone compared to price comparison displays (displaying prices for the selected order along with reasonable alternatives). Although price comparison was a component of the intervention in some of the studies in this review, it was not evaluated relative to price display alone. Furthermore, it would be helpful to know if the type of price displayed affects its impact. For instance, if providers are most sensitive to the absolute magnitude of prices, then displaying chargemaster prices may impact ordering more than showing hospital costs. If, however, relative prices are all that providers need, then showing lower numbers, such as Medicare prices or hospital costs, may be sufficient. Finally, it would be reassuring to have additional evidence that price display does not adversely impact patient outcomes.
Although some details need elucidation, the studies synthesized in this review provide valuable data in the current climate of increased emphasis on price transparency. Although substantial attention has been devoted by the academic community, technology start‐ups, private insurers, and even state legislatures to improving price transparency to patients, less focus has been given to physicians, for whom healthcare prices are often just as opaque.[4] The findings from this review suggest that provider price display may be an effective, safe, and acceptable approach to empower physicians to control healthcare spending.
Disclosures: Dr. Silvestri, Dr. Bongiovanni, and Ms. Glover have nothing to disclose. Dr. Gross reports grants from Johnson & Johnson, Medtronic Inc., and 21st Century Oncology during the conduct of this study. In addition, he received payment from Fair Health Inc. and ASTRO outside the submitted work.
Rising healthcare spending has garnered significant public attention, and is considered a threat to other national priorities. Up to one‐third of national health expenditures are wasteful, the largest fraction generated through unnecessary services that could be substituted for less‐costly alternatives or omitted altogether.[1] Physicians play a central role in health spending, as they purchase nearly all tests and therapies on behalf of patients.
One strategy to enhance cost‐conscious physician ordering is to increase transparency of cost data for providers.[2, 3, 4] Although physicians consider price an important factor in ordering decisions, they have difficulty estimating costs accurately or finding price information easily.[5, 6] Improving physicians' knowledge of order costs may prompt them to forego diagnostic tests or therapies of low utility, or shift ordering to lower‐cost alternatives. Real‐time price display during provider order entry is 1 approach for achieving this goal. Modern electronic health records (EHRs) with computerized physician order entry (CPOE) make price display not only practical but also scalable. Integrating price display into clinical workflow, however, can be challenging, and there remains lack of clarity about potential risks and benefits. The dissemination of real‐time CPOE price display, therefore, requires an understanding of its impact on clinical care.
Over the past 3 decades, several studies in the medical literature have evaluated the effect of price display on physician ordering behavior. To date, however, there has been only 1 narrative review of this literature, which did not include several recent studies on the topic or formally address study quality and physician acceptance of price display modules.[7] Therefore, to help inform healthcare leaders, technology innovators, and policy makers, we conducted a systematic review to address 4 key questions: (1) What are the characteristics of interventions that have displayed order prices to physicians in the context of actual practice? (2) To what degree does real‐time display of order prices impact order costs and order volume? (3) Does price display impact patient safety outcomes, and is it acceptable to providers? (4) What is the quality of the current literature on this topic?
METHODS
Data Sources
We searched 2 electronic databases, MEDLINE and Embase, using a combination of controlled vocabulary terms and keywords that covered both the targeted intervention (eg, fees and charges) and the outcome of interest (eg, physician's practice patterns), limited to English language articles with no restriction on country or year of publication (see Supporting Information, Appendix 1, in the online version of this article). The search was run through August 2014. Results from both database searches were combined and duplicates eliminated. We also ran a MEDLINE keyword search on titles and abstracts of articles from 2014 that were not yet indexed. A medical librarian was involved in all aspects of the search process.[8]
Study Selection
Studies were included if they evaluated the effect of displaying actual order prices to providers during the ordering process and reported the impact on provider ordering practices. Reports in any clinical context and with any study design were included. To assess most accurately the effect of price display on real‐life ordering and patient outcomes, studies were excluded if: (1) they were review articles, commentaries, or editorials; (2) they did not show order prices to providers; (3) the context was a simulation; (4) the prices displayed were relative (eg, $/$$/$$$) or were only cumulative; (5) prices were not presented real‐time during the ordering process; or (6) the primary outcome was neither order costs nor order volume. We decided a priori to exclude simulations because these may not accurately reflect provider behavior when treating real patients, and to exclude studies showing relative prices due to concerns that it is a less significant price transparency intervention and that providers may interpret relative prices differently from actual prices.
Two reviewers, both physicians and health service researchers (M.T.S. and T.R.B.), separately reviewed the full list of titles and abstracts. For studies that potentially met inclusion criteria, full articles were obtained and were independently read for inclusion in the final review. The references of all included studies were searched manually, and the Scopus database was used to search all studies that cited the included studies. We also searched the references of relevant literature reviews.[9, 10, 11] Articles of interest discovered through manual search were then subjected to the same process.
Data Extraction and Quality Assessment
Two reviewers (M.T.S. and T.R.B.) independently performed data extraction using a standardized spreadsheet. Discrepancies were resolved by reviewer consensus. Extracted study characteristics included study design and duration, clinical setting, study size, type of orders involved, characteristics of price display intervention and control, and type of outcome. Findings regarding patient safety and provider acceptability were also extracted when available.
Study quality was independently evaluated and scored by both reviewers using the Downs and Black checklist, designed to assess quality of both randomized and nonrandomized studies.[12] The checklist contains 5 items pertaining to allocation concealment, blinding, or follow‐up that are not applicable to an administrative intervention like price display, so these questions were excluded. Additionally, few studies calculated sample size or reported post hoc statistical power, so we also excluded this question, leaving a modified 21‐item checklist. We also assessed each study for sources of bias that were not already assessed by the Downs and Black checklist, including contamination between study groups, confounding of results, and incomplete intervention or data collection.
Data Synthesis
Data are reported in tabular form for all included studies. Due to heterogeneity of study designs and outcome measures, data from the studies were not pooled quantitatively. This review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis guidelines.
RESULTS
Database searches yielded a total of 1400 articles, of which 18 were selected on the basis of title and abstract for detailed assessment. Reference searching led us to retrieve 94 further studies of possible interest, of which 23 were selected on the basis of abstract for detailed assessment. Thus, 41 publications underwent full manuscript review, 19 of which met all inclusion criteria (see Supporting Information, Appendix 2, in the online version of this article).[13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31] These studies were published between 1983 and 2014, and were conducted primarily in the United States.
Study Characteristics
There was considerable heterogeneity among the 19 studies with regard to design, setting, and scope (Table 1). There were 5 randomized trials, for which the units of randomization were patient (1), provider team (2), and test (2). There were 13 pre‐post intervention studies, 5 of which used a concomitant control group, and 2 of which included a washout period. There was 1 interrupted time series study. Studies were conducted within inpatient hospital floors (8), outpatient clinics (4), emergency departments (ED) or urgent care facilities (4), and hospital operating rooms (3).
| Study | Design | Clinical Setting | Providers | Intervention and Duration | Order(s) Studied | Type of Price Displayed | Concurrent Interventions |
|---|---|---|---|---|---|---|---|
| |||||||
| Fang et al.[14] 2014 | Pre‐post study with control group | Academic hospital (USA) | All inpatient ordering providers | CPOE system with prices displayed for reference lab tests; 8 months | All send‐out lab tests | Charge from send‐out laboratory, displayed as range (eg, $100300) | Display also contained expected lab turnaround time |
| Nougon et al.[13] 2014 | Pre‐post study with washout | Academic adult emergency department (Belgium) | 9 ED house staff | CPOE system with prices displayed on common orders form, and price list displayed above all workstations and in patient rooms; 2 months | Common lab and imaging tests | Reference costs from Belgian National Institute for Health Insurance and Invalidity | None |
| Durand et al.[17] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 10 common imaging tests | Medicare allowable fee | None |
| Feldman et al.[16] 2013 | RCT (randomized by test) | Academic hospital, all inpatients (USA) | All inpatient ordering providers | CPOE system with prices displayed; 6 months | 61 lab tests | Medicare allowable fee | None |
| Horn et al.[15] 2014 | Interrupted time series study with control group | Private outpatient group practice alliance (USA) | 215 primary care physicians | CPOE system with prices displayed; 6 months | 27 lab tests | Medicare allowable fee, displayed as narrow range (eg, $5$10) | None |
| Ellemdin et al.[18] 2011 | Pre‐post study with control group | Academic hospital, internal medicine units (South Africa) | Internal medicine physicians (number NR) | Sheet with lab test costs given to intervention group physicians who were required to write out cost for each order; 4 months | Common lab tests | Not reported | None |
| Schilling,[19] 2010 | Pre‐post study with control group | Academic adult emergency department (Sweden) | All internal medicine physicians in ED | Standard provider workstations with price lists posted on each; 2 months | 91 common lab tests, 39 common imaging tests | Not reported | None |
| Guterman et al.[21] 2002 | Pre‐post study | Academic‐affiliated urgent care clinic (USA) | 51 attendings and housestaff | Preformatted paper prescription form with medication prices displayed; 2 weeks | 2 H2‐blocker medications | Acquisition cost of medication plus fill fee | None |
| Seguin et al.[20] 2002 | Pre‐post study | Academic surgical intensive care unit (France) | All intensive care unit physicians | Paper quick‐order checklist with prices displayed; 2 months | 6 common lab tests, 1 imaging test | Not reported | None |
| Hampers et al.[23] 1999 | Pre‐post study with washout | Academic pediatric emergency department (USA) | Pediatric ED attendings and housestaff (number NR) | Paper common‐order checklist with prices displayed; 3 months | 22 common lab and imaging tests | Chargemaster price* | Physicians required to calculate total charges for diagnostic workup |
| Ornstein et al.[22] 1999 | Pre‐post study | Academic family medicine outpatient clinic (USA) | 46 attendings and housestaff | Microcomputer CPOE system with medication prices displayed; 6 months | All medications | AWP for total supply (acute medications) or 30‐day supply (chronic medications) | Additional keystroke produced list of less costly alternative medications |
| Lin et al.[25] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | All anesthesia providers | Standard muscle relaxant drug vials with price stickers displayed; 12 months | All muscle relaxant medications | Not reported | None |
| McNitt et al.[24] 1998 | Pre‐post study | Academic hospital operating rooms (USA) | 90 anesthesia attendings, housestaff and anesthetists | List of drug costs displayed in operating rooms, anesthesia lounge, and anesthesia satellite pharmacy; 10 months | 22 common anesthesia medications | Hospital acquisition cost | Regular anesthesia department reviews of drug usage and cost |
| Bates et al.[27] 1997 | RCT (randomized by patient) | Academic hospital, medical and surgical inpatients (USA) | All inpatient ordering providers | CPOE system with display of test price and running total of prices for the ordering session; 4 months (lab) and 7 months (imaging) | All lab tests, 35 common imaging tests | Chargemaster price | None |
| Vedsted et al.[26] 1997 | Pre‐post study with control group | Outpatient general practices (Denmark) | 231 general practitioners | In practices already using APEX CPOE system, introduction of medication price display (control practices used non‐APEX computer system or paper‐based prescribing); 12 months | All medications | Chargemaster price | Medication price comparison module (stars indicated availability of cheaper option) |
| Horrow et al.[28] 1994 | Pre‐post study | Private tertiary care hospital operating rooms (USA) | 56 anesthesia attendings, housestaff and anesthetists | Standard anesthesia drug vials and syringes with supermarket price stickers displayed; 3 months | 13 neuromuscular relaxant and sedative‐hypnotic medications | Hospital acquisition cost | None |
| Tierney et al.[29] 1993 | Cluster RCT (randomized by provider team) | Public hospital, internal medicine services (USA) | 68 teams of internal medicine attendings and housestaff | Microcomputer CPOE system with prices displayed (control group used written order sheets); 17 months | All orders | Chargemaster price | CPOE system listed cost‐effective tests for common problems and displayed reasonable test intervals |
| Tierney et al.[30] 1990 | Cluster RCT (randomized by clinic session) | Academic, outpatient, general medicine practice (USA) | 121 internal medicine attendings and housestaff | Microcomputer CPOE system with pop‐up window displaying price for current test and running total of cumulative test prices for current visit; 6 months | All lab and imaging tests | Chargemaster price | None |
| Everett et al.[31] 1983 | Pre‐post study with control group | Academic hospital, general internal medicine wards (USA) | Internal medicine attendings and housestaff (number NR) | Written order sheet with adjacent sheet of lab test prices; 3 months | Common lab tests | Chargemaster price | None |
Prices were displayed for laboratory tests (12 studies), imaging tests (8 studies), and medications (7 studies). Study scope ranged from examining a single medication class to evaluating all inpatient orders. The type of price used for the display varied, with the most common being the facility charges or chargemaster prices (6 studies), and Medicare prices (3 studies). In several cases, price display was only 1 component of the study, and 6 studies introduced additional interventions concurrent with price display, such as cost‐effective ordering menus,[29] medication comparison modules,[26] or display of test turnaround times.[14] Seven of the 19 studies were conducted in the past decade, of which 5 displayed prices within an EHR.[13, 14, 15, 16, 17]
Order Costs and Volume
Thirteen studies reported the numeric impact of price display on aggregate order costs (Table 2). Nine of these demonstrated a statistically significant (P 0.05) decrease in order costs, with effect sizes ranging from 10.7% to 62.8%.[13, 16, 18, 20, 23, 24, 28, 29, 30] Decreases were found for lab costs, imaging costs, and medication costs, and were observed in both the inpatient and outpatient settings. Three of these 9 studies were randomized. For example, in 1 study randomizing 61 lab tests to price display or no price display, costs for the intervention labs dropped 9.6% compared to the year prior, whereas costs for control labs increased 2.9% (P 0.001).[16] Two studies randomized by provider group showed that providers seeing order prices accrued 12.7% fewer charges per inpatient admission (P = 0.02) and 12.9% fewer test charges per outpatient visit (P 0.05).[29, 30] Three studies found no significant association between price display and order costs, with effect sizes ranging from a decrease of 18.8% to an increase of 4.3%.[19, 22, 27] These studies also evaluated lab, imaging, and medication costs, and included 1 randomized trial. One additional large study noted a 12.5% decrease in medication costs after initiation of price display, but did not statistically evaluate this difference.[25]
| Study | No. of Encounters | Primary Outcome Measure(s) | Impact on Order Costs | Impact on Order Volume | ||||
|---|---|---|---|---|---|---|---|---|
| Control Group Outcome | Intervention Group Outcome | Relative Change | Control Group Outcome | Intervention Group Outcome | Relative Change | |||
| ||||||||
| Fang et al.[14] 2014 | 378,890 patient‐days | Reference lab orders per 1000 patient‐days | NR | NR | NA | 51 orders/1000 patient‐days | 38 orders/1000 patient‐days | 25.5% orders/1000 patient‐days (P 0.001) |
| Nougon et al.[13] 2015 | 2422 ED visits (excluding washout) | Lab and imaging test costs per ED visit | 7.1/visit (lab); 21.8/visit (imaging) | 6.4/visit (lab); 14.4/visit (imaging) | 10.7% lab costs/ visit (P = 0.02); 33.7% imaging costs/visit (P 0.001) | NR | NR | NA |
| Durand et al.[17] 2013 | NR | Imaging orders compared to baseline 1 year prior | NR | NR | NA | 3.0% total orders | +2.8% total orders | +5.8% total orders (P = 0.10) |
| Feldman et al.[16] 2013 | 245,758 patient‐days | Lab orders and fees per patient‐day compared to baseline 1 year prior | +2.9% fees/ patient‐day | 9.6% fees/ patient‐day | 12.5% fees/patient‐day (P 0.001) | +5.6% orders/patient‐day | 8.6% orders/ patient‐day | 14.2% orders/patient‐day (P 0.001) |
| Horn et al.[15] 2014 | NR | Lab test volume per patient visit, by individual lab test | NR | NR | NA | Aggregate data not reported | Aggregate data not reported | 5 of 27 tests had significant reduction in ordering (2.1% to 15.2%/patient visit) |
| Ellemdin et al.[18] 2011 | 897 admissions | Lab cost per hospital day | R442.90/day | R284.14/day | 35.8% lab costs/patient‐day (P = 0.001) | NR | NR | NA |
| Schilling[19] 2010 | 3222 ED visits | Combined lab and imaging test costs per ED visit | 108/visit | 88/visit | 18.8% test costs/visit (P = 0.07) | NR | NR | NA |
| Guterman et al.[21] 2002 | 168 urgent care visits | Percent of acid reducer prescriptions for ranitidine (the higher‐cost option) | NR | NR | NA | 49% ranitidine | 21% ranitidine | 57.1% ranitidine (P = 0.007) |
| Seguin et al.[20] 2002 | 287 SICU admissions | Tests ordered per admission; test costs per admission | 341/admission | 266/admission | 22.0% test costs/admission (P 0.05) | 13.6 tests/admission | 11.1 tests/ admission | 18.4% tests/admission (P = 0.12) |
| Hampers et al.[23] 1999 | 4881 ED visits (excluding washout) | Adjusted mean test charges per patient visit | $86.79/visit | $63.74/visit | 26.6% test charges/visit (P 0.01) | NR | NR | NA |
| Ornstein et al.[22] 1999 | 30,461 outpatient visits | Prescriptions per visit; prescription cost per visit; cost per prescription | $12.49/visit; $21.83/ prescription | $13.03/visit; $22.03/prescription |
+4.3% prescription costs/visit (P = 0.12); +0.9% cost/prescription (P = 0.61) |
0.66 prescriptions/visit | 0.64 prescriptions/ visit | 3.0% prescriptions/visit (P value not reported) |
| Lin et al.[25] 1998 | 40,747 surgical cases | Annual spending on muscle relaxants medication |
$378,234/year (20,389 cases) |
$330,923/year (20,358 cases) |
12.5% | NR | NR | NA |
| McNitt et al.[24] 1998 | 15,130 surgical cases | Anesthesia drug cost per case | $51.02/case | $18.99/case | 62.8% drug costs/case (P 0.05) | NR | NR | NA |
| Bates et al.[27] 1997 | 7090 admissions (lab); 17,381 admissions (imaging) | Tests ordered per admission; charges for tests ordered per admission |
$771/ admission (lab); $276/admission (imaging) |
$739/admission (lab); $275/admission (imaging) |
4.2% lab charges/admission (P = 0.97); 0.4% imaging charges/admission (P = 0.10) |
26.8 lab tests/admission; 1.76 imaging tests/admission |
25.6 lab tests/ admission; 1.76 imaging tests/ admission |
4.5% lab tests/admission (P = 0.74); 0% imaging tests/admission (P = 0.13) |
| Vedsted et al.[26] 1997 | NR | Prescribed daily doses per 1000 insured; total drug reimbursement per 1000 insured; reimbursement per daily dose | Reported graphically only | Reported graphically only | No difference | Reported graphically only | Reported graphically only | No difference |
| Horrow et al.[28] 1994 | NR | Anesthetic drugs used per week; anesthetic drug cost per week | $3837/week | $3179/week | 17.1% drug costs/week (P = 0.04) | 97 drugs/week | 94 drugs/week | 3.1% drugs/week (P = 0.56) |
| Tierney et al.[29] 1993 | 5219 admissions | Total charges per admission | $6964/admission | $6077/admission | 12.7% total charges/admission (P = 0.02) | NR | NR | NA |
| Tierney et al.[30] 1990 | 15,257 outpatient visits | Test orders per outpatient visit; test charges per outpatient visit | $51.81/visit | $45.13/visit | 12.9% test charges/visit (P 0.05) | 1.82 tests/visit | 1.56 tests/visit | 14.3% tests/visit (P 0.005) |
| Everett et al.[31] 1983 | NR | Lab tests per admission; charges per admission | NR | NR | NA | NR | NR | No statistically significant changes |
Eight studies reported the numeric impact of price display on aggregate order volume. Three of these demonstrated a statistically significant decrease in order volume, with effect sizes ranging from 14.2% to 25.5%.[14, 16, 30] Decreases were found for lab and imaging tests, and were observed in both inpatient and outpatient settings. For example, 1 pre‐post study displaying prices for inpatient send‐out lab tests demonstrated a 25.5% reduction in send‐out labs per 1000 patient‐days (P 0.001), whereas there was no change for the control group in‐house lab tests, for which prices were not shown.[14] The other 5 studies reported no significant association between price display and order volume, with effect sizes ranging from a decrease of 18.4% to an increase of 5.8%.[17, 20, 22, 27, 28] These studies evaluated lab, imaging, and medication volume. One trial randomizing by individual inpatient showed a nonsignificant decrease of 4.5% in lab orders per admission in the intervention group (P = 0.74), although the authors noted that their study had insufficient power to detect differences less than 10%.[27] Of note, 2 of the 5 studies reporting nonsignificant impacts on order volume (3.1%, P = 0.56; and 18.4%, P = 0.12) did demonstrate significant decreases in order costs (17.1%, P = 0.04; and 22.0%, P 0.05).[20, 28]
There were an additional 2 studies that reported the impact of price display on order volume for individual orders only. In 1 time‐series study showing lab test prices, there was a statistically significant decrease in order volume for 5 of 27 individual tests studied (using a Bonferroni‐adjusted threshold of significance), with no tests showing a significant increase.[15] In 1 pre‐post study showing prices for H2‐antagonist drugs, there was a statistically significant 57.1% decrease in order volume for the high‐cost medication, with a corresponding 58.7% increase in the low‐cost option.[21] These studies did not report impact on aggregate order costs. Two further studies in this review did not report outcomes numerically, but did state in their articles that significant impacts on order volume were not observed.[26, 31]
Therefore, of the 19 studies included in this review, 17 reported numeric results. Of these 17 studies, 12 showed that price display was associated with statistically significant decreases in either order costs or volume, either in aggregate (10 studies; Figure 1) or for individual orders (2 studies). Of the 7 studies conducted within the past decade, 5 noted significant decreases in order costs or volume. Prices were embedded into an EHR in 5 of these recent studies, and 4 of the 5 observed significant decreases in order costs or volume. Only 2 studies from the past decade1 from Belgium and 1 from the United Statesincorporated prices into an EHR and reported aggregate order costs. Both found statistically significant decreases in order costs with price display.[13, 16]

Patient Safety and Provider Acceptability
Five studies reported patient‐safety outcomes. One inpatient randomized trial showed similar rates of postdischarge utilization and charges between the intervention and control groups.[29] An outpatient randomized trial showed similar rates of hospital admissions, ED visits, and outpatient visits between the intervention and control groups.[30] Two pre‐post studies showing anesthesia prices in hospital operating rooms included a quality assurance review and showed no changes in adverse outcomes such as prolonged postoperative intubation, recovery room stay, or unplanned intensive care unit admissions.[24, 25] The only adverse safety finding was in a pre‐post study in a pediatric ED, which showed a higher rate of unscheduled follow‐up care during the intervention period compared to the control period (24.4% vs 17.8%, P 0.01) but similar rates of patients feeling better (83.4% vs 86.7%, P = 0.05). These findings, however, were based on self‐report during telephone follow‐up with a 47% response rate.[23]
Five studies reported on provider acceptability of price display. Two conducted questionnaires as part of the study plan, whereas the other 3 offered general provider feedback. One questionnaire revealed that 83% of practices were satisfied or very satisfied with the price display.[26] The other questionnaire found that 81% of physicians felt the price display improved my knowledge of the relative costs of tests I order and similarly 81% would like additional cost information displayed for other orders.[15] Three studies reported subjectively that showing prices initially caused questions from most physicians,[13] but that ultimately, physicians like seeing this information[27] and gave feedback that was generally positive.[21] One study evaluated the impact of price display on provider cost knowledge. Providers in the intervention group did not improve in their cost‐awareness, with average errors in cost estimates exceeding 40% even after 6 months of price display.[30]
Study Quality
Using a modified Downs and Black checklist of 21 items, studies in this review ranged in scores from 5 to 20, with a median score of 15. Studies most frequently lost points for being nonrandomized, failing to describe or adjust for potential confounders, being prone to historical confounding, or not evaluating potential adverse events.
We supplemented this modified Downs and Black checklist by reviewing 3 categories of study limitations not well‐reflected in the checklist scoring (Table 3). The first was potential for contamination between study groups, which was a concern in 4 studies. For example, 1 pre‐post study assessing medication ordering included clinical pharmacists in patient encounters both before and after the price display intervention.[22] This may have enhanced cost‐awareness even before prices were shown. The second set of limitations, present in 12 studies, included confounders that were not addressed by study design or analysis. For example, the intervention in 1 study displayed not just test cost but also test turnaround time, which may have separately influenced providers against ordering a particular test.[14] The third set of limitations included unanticipated gaps in the display of prices or in the collection of ordering data, which occurred in 5 studies. If studies did not report on gaps in the intervention or data collection, we assumed there were none.
| Study | Modified Downs & Black Score (Max Score 21) | Other Price Display Quality Criteria (Not Included in Downs & Black Score) | ||
|---|---|---|---|---|
| Potential for Contamination Between Study Groups | Potential Confounders of Results Not Addressed by Study Design or Analysis | Incomplete Price Display Intervention or Data Collection | ||
| ||||
| Fang et al.[14] 2014 | 14 | None | Concurrent display of test turnaround time may have independently contributed to decreased test ordering | 21% of reference lab orders were excluded from analysis because no price or turnaround‐time data were available |
| Nougon et al.[13] 2015 | 16 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Durand et al.[17] 2013 | 17 | Providers seeing test prices for intervention tests (including lab tests in concurrent Feldman study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, chest x‐ray) was not independent of control test ordering (eg, CT chest) | None |
| Feldman et al.[16] 2013 | 18 | Providers seeing test prices for intervention tests (including imaging tests in concurrent Durand study) may have remained cost‐conscious when placing orders for control tests | Interference between units likely occurred because intervention test ordering (eg, CMP) was not independent of control test ordering (eg, BMP) | None |
| Horn et al.[15] 2014 | 15 | None | None | None |
| Ellemdin et al.[18] 2011 | 15 | None | None | None |
| Schilling[19] 2010 | 12 | None | None | None |
| Guterman et al.[21] 2002 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | None |
| Seguin et al.[20] 2002 | 17 | None | Because primary outcome was not adjusted for length of stay, the 30% shorter average length of stay during intervention period may have contributed to decreased costs per admission; historical confounding may have existed due to pre‐post study design without control group | None |
| Hampers et al.[23] 1999 | 17 | None | Requirement that physicians calculate total charges for each visit may have independently contributed to decreased test ordering; historical confounding may have existed due to pre‐post study design without control group | 10% of eligible patient visits were excluded from analysis because prices were not displayed or ordering data were not collected |
| Ornstein et al.[22] 1999 | 15 | Clinical pharmacists and pharmacy students involved in half of all patient contacts may have enhanced cost‐awareness during control period | Emergence of new drugs during intervention period and an ongoing quality improvement activity to increase prescribing of lipid‐lowering medications may have contributed to increased medication costs; historical confounding may have existed due to pre‐post study design without control group | 25% of prescription orders had no price displayed, and average prices were imputed for purposes of analysis |
| Lin et al.[25] 1998 | 12 | None | Emergence of new drug during intervention period and changes in several drug prices may have contributed to decreased order costs; historical confounding may have existed due to pre‐post study design without control group | None |
| McNitt et al.[24] 1998 | 15 | None | Intensive drug‐utilization review and cost‐reduction efforts may have independently contributed to decreased drug costs; historical confounding may have existed due to pre‐post study design without control group | None |
| Bates et al.[27] 1997 | 18 | Providers seeing test prices on intervention patients may have remembered prices or remained cost‐conscious when placing orders for control patients | None | 47% of lab tests and 26% of imaging tests were ordered manually outside of the trial's CPOE display system* |
| Vedsted et al.[26] 1997 | 5 | None | Medication price comparison module may have independently influenced physician ordering | None |
| Horrow et al.[28] 1994 | 14 | None | Historical confounding may have existed due to pre‐post study design without control group | Ordering data for 2 medications during 2 of 24 weeks were excluded from analysis due to internal inconsistency in the data |
| Tierney et al.[29] 1993 | 20 | None | Introduction of computerized order entry and menus for cost‐effective ordering may have independently contributed to decreased test ordering | None |
| Tierney et al.[30] 1990 | 20 | None | None | None |
| Everett et al.[31] 1983 | 7 | None | None | None |
Even among the 5 randomized trials there were substantial limitations. For example, 2 trials used individual tests as the unit of randomization, although ordering patterns for these tests are not independent of each other (eg, ordering rates for comprehensive metabolic panels are not independent of ordering rates for basic metabolic panels).[16, 17] This creates interference between units that was not accounted for in the analysis.[32] A third trial was randomized at the level of the patient, so was subject to contamination as providers seeing the price display for intervention group patients may have remained cost‐conscious while placing orders for control group patients.[27] In a fourth trial, the measured impact of the price display may have been confounded by other aspects of the overall cost intervention, which included cost‐effective test menus and suggestions for reasonable testing intervals.[29]
The highest‐quality study was a cluster‐randomized trial published in 1990 specifically measuring the effect of price display on a wide range of orders.[30] Providers and patients were separated by clinic session so as to avoid contamination between groups, and the trial included more than 15,000 outpatient visits. The intervention group providers ordered 14.3% fewer tests than control group providers, which resulted in 12.9% lower charges.
DISCUSSION
We identified 19 published reports of interventions that displayed real‐time order prices to providers and evaluated the impact on provider ordering. There was substantial heterogeneity in study setting, design, and quality. Although there is insufficient evidence on which to base strong conclusions, these studies collectively suggest that provider price display likely reduces order costs to a modest degree. Data on patient safety were largely lacking, although in the few studies that examined patient outcomes, there was little evidence that patient safety was adversely affected by the intervention. Providers widely viewed display of prices positively.
Our findings align with those of a recent systematic review that concluded that real‐time price information changed provider ordering in the majority of studies.[7] Whereas that review evaluated 17 studies from both clinical settings and simulations, our review focused exclusively on studies conducted in actual ordering environments. Additionally, our literature search yielded 8 studies not previously reviewed. We believe that the alignment of our findings with the prior review, despite the differences in studies included, adds validity to the conclusion that price display likely has a modest impact on reducing order costs. Our review contains several additions important for those considering price display interventions. We provide detailed information on study settings and intervention characteristics. We present a formal assessment of study quality to evaluate the strength of individual study findings and to guide future research in this area. Finally, because both patient safety and provider acceptability may be a concern when prices are shown, we describe all safety outcomes and provider feedback that these studies reported.
The largest effect sizes were noted in 5 studies reporting decreases in order volume or costs greater than 25%.[13, 14, 18, 23, 24] These were all pre‐post intervention studies, so the effect sizes may have been exaggerated by historical confounding. However, the 2 studies with concurrent control groups found no decreases in order volume or cost in the control group.[14, 18] Among the 5 studies that did not find a significant association between price display and provider ordering, 3 were subject to contamination between study groups,[17, 22, 27] 1 was underpowered,[19] and 1 noted a substantial effect size but did not perform a statistical analysis.[25] We also found that order costs were more frequently reduced than order volume, likely because shifts in ordering to less expensive alternatives may cause costs to decrease while volume remains unchanged.[20, 28]
If price display reduces order costs, as the majority of studies in this review indicate, this finding carries broad implications. Policy makers could promote cost‐conscious care by creating incentives for widespread adoption of price display. Hospital and health system leaders could improve transparency and reduce expenses by prioritizing price display. The specific beneficiaries of any reduced spending would depend on payment structures. With shifts toward financial risk‐bearing arrangements like accountable care organizations, healthcare institutions may have a financial interest in adopting price display. Because price display is an administrative intervention that can be developed within EHRs, it is potentially 1 of the most rapidly scalable strategies for reducing healthcare spending. Even modest reductions in spending on laboratory tests, imaging studies, and medications would result in substantial savings on a system‐wide basis.
Implementing price display does not come without challenges. Prices need to be calculated or obtained, loaded into an EHR system, and updated periodically. Technology innovators could enhance EHR software by making these processes easier. Healthcare institutions may find displaying relative prices (eg, $/$$/$$$) logistically simpler in some contexts than showing actual prices (eg, purchase cost), such as when contracts require prices to be confidential. Although we decided to exclude studies displaying relative prices, our search identified no studies that met other inclusion criteria but displayed relative prices, suggesting a lack of evidence regarding the impact of relative price display as an alternative to actual price display.
There are 4 key limitations to our review. First, the heterogeneity of the study designs and reported outcomes precluded pooling of data. The variety of clinical settings and mechanisms through which prices were displayed enhances the generalizability of our findings, but makes it difficult to identify particular contexts (eg, type of price or type of order) in which the intervention may be most effective. Second, although the presence of negative studies on this subject reduces the concern for reporting bias, it remains possible that sites willing to implement and study price displays may be inherently more sensitive to prices, such that published results might be more pronounced than if the intervention were widely implemented across multiple sites. Third, the mixed study quality limits the strength of conclusions that can be drawn. Several studies with both positive and negative findings had issues of bias, contamination, or confounding that make it difficult to be confident of the direction or magnitude of the main findings. Studies evaluating price display are challenging to conduct without these limitations, and that was apparent in our review. Finally, because over half of the studies were conducted over 15 years ago, it may limit their generalizability to modern ordering environments.
We believe there remains a need for high‐quality evidence on this subject within a contemporary context to confirm these findings. The optimal methodology for evaluating this intervention is a cluster randomized trial by facility or provider group, similar to that reported by Tierney et al. in 1990, with a primary outcome of aggregate order costs.[30] Given the substantial investment this would require, a large time series study could also be informative. As most prior price display interventions have been under 6 months in duration, it would be useful to know if the impact on order costs is sustained over a longer time period. The concurrent introduction of any EHR alerts that could impact ordering (eg, duplicate test warnings) should be simultaneously measured and reported. Studies also need to determine the impact of price display alone compared to price comparison displays (displaying prices for the selected order along with reasonable alternatives). Although price comparison was a component of the intervention in some of the studies in this review, it was not evaluated relative to price display alone. Furthermore, it would be helpful to know if the type of price displayed affects its impact. For instance, if providers are most sensitive to the absolute magnitude of prices, then displaying chargemaster prices may impact ordering more than showing hospital costs. If, however, relative prices are all that providers need, then showing lower numbers, such as Medicare prices or hospital costs, may be sufficient. Finally, it would be reassuring to have additional evidence that price display does not adversely impact patient outcomes.
Although some details need elucidation, the studies synthesized in this review provide valuable data in the current climate of increased emphasis on price transparency. Although substantial attention has been devoted by the academic community, technology start‐ups, private insurers, and even state legislatures to improving price transparency to patients, less focus has been given to physicians, for whom healthcare prices are often just as opaque.[4] The findings from this review suggest that provider price display may be an effective, safe, and acceptable approach to empower physicians to control healthcare spending.
Disclosures: Dr. Silvestri, Dr. Bongiovanni, and Ms. Glover have nothing to disclose. Dr. Gross reports grants from Johnson & Johnson, Medtronic Inc., and 21st Century Oncology during the conduct of this study. In addition, he received payment from Fair Health Inc. and ASTRO outside the submitted work.
- Institute of Medicine. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America: Washington, DC: National Academies Press; 2012.
- . Do physicians need a “shopping cart” for health care services? JAMA. 2012;307(8):791–792.
- . The disruptive innovation of price transparency in health care. JAMA. 2013;310(18):1927–1928.
- , . Providing price displays for physicians: which price is right? JAMA. 2014;312(16):1631–1632.
- , . Physician awareness of diagnostic and nondrug therapeutic costs: a systematic review. Int J Tech Assess Health Care. 2008;24(2):158–165.
- , , . Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4(9):e283.
- , , , . The effect of charge display on cost of care and physician practice behaviors: a systematic review. J Gen Intern Med. 2015;30:835–842.
- , , . Engaging medical librarians to improve the quality of review articles. JAMA. 2014;312(10):999–1000.
- , , . Influencing behavior of physicians ordering laboratory tests: a literature study. Med Care. 1993;31(9):784–794.
- , . Trials of providing costing information to general practitioners: a systematic review. Med J Aust. 1997;167(2):89–92.
- . A review of physician cost‐containment strategies for laboratory testing. Med Care. 1983;21(8):783–802.
- , . The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non‐randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377–384.
- , , , et al. Does offering pricing information to resident physicians in the emergency department potentially reduce laboratory and radiology costs? Eur J Emerg Med. 2015;22:247–252.
- , , , et al. Cost and turn‐around time display decreases inpatient ordering of reference laboratory tests: a time series. BMJ Qual Saf. 2014;23:994–1000.
- , , , , . The impact of cost displays on primary care physician laboratory test ordering. J Gen Intern Med. 2014;29:708–714.
- , , , et al. Impact of providing fee data on laboratory test ordering: a controlled clinical trial. JAMA Intern Med. 2013;173(10):903–908.
- , , , . Provider cost transparency alone has no impact on inpatient imaging utilization. J Am Coll Radiol. 2013;10(2):108–113.
- , , . Providing clinicians with information on laboratory test costs leads to reduction in hospital expenditure. S Afr Med J. 2011;101(10):746–748.
- . Cutting costs: the impact of price lists on the cost development at the emergency department. Eur J Emerg Med. 2010;17(6):337–339.
- , , , , . Effects of price information on test ordering in an intensive care unit. Intens Care Med. 2002;28(3):332–335.
- , , , , , . Modifying provider behavior: a low‐tech approach to pharmaceutical ordering. J Gen Intern Med. 2002;17(10):792–796.
- , , , , . Medication cost information in a computer‐based patient record system. Impact on prescribing in a family medicine clinical practice. Arch Fam Med. 1999;8(2):118–121.
- , , , , . The effect of price information on test‐ordering behavior and patient outcomes in a pediatric emergency department. Pediatrics. 1999;103(4 pt 2):877–882.
- , , . Long‐term pharmaceutical cost reduction using a data management system. Anesth Analg. 1998;87(4):837–842.
- , . The impact of price labeling of muscle relaxants on cost consciousness among anesthesiologists. J Clin Anesth. 1998;10(5):401–403.
- , , . Does a computerized price comparison module reduce prescribing costs in general practice? Fam Pract. 1997;14(3):199–203.
- , , , et al. Does the computerized display of charges affect inpatient ancillary test utilization? Arch Intern Med. 1997;157(21):2501–2508.
- , . Price stickers do not alter drug usage. Can J Anaesth. 1994;41(11):1047–1052.
- , , , . Physician inpatient order writing on microcomputer workstations. Effects on resource utilization. JAMA. 1993;269(3):379–383.
- , , . The effect on test ordering of informing physicians of the charges for outpatient diagnostic tests. N Engl J Med. 1990;322(21):1499–1504.
- , , , . Effect of cost education, cost audits, and faculty chart review on the use of laboratory services. Arch Intern Med. 1983;143(5):942–944.
- . Interference between units in randomized experiments. J Am Stat Assoc. 2007;102(477):191–200.
- Institute of Medicine. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America: Washington, DC: National Academies Press; 2012.
- . Do physicians need a “shopping cart” for health care services? JAMA. 2012;307(8):791–792.
- . The disruptive innovation of price transparency in health care. JAMA. 2013;310(18):1927–1928.
- , . Providing price displays for physicians: which price is right? JAMA. 2014;312(16):1631–1632.
- , . Physician awareness of diagnostic and nondrug therapeutic costs: a systematic review. Int J Tech Assess Health Care. 2008;24(2):158–165.
- , , . Physician awareness of drug cost: a systematic review. PLoS Med. 2007;4(9):e283.
- , , , . The effect of charge display on cost of care and physician practice behaviors: a systematic review. J Gen Intern Med. 2015;30:835–842.
- , , . Engaging medical librarians to improve the quality of review articles. JAMA. 2014;312(10):999–1000.
- , , . Influencing behavior of physicians ordering laboratory tests: a literature study. Med Care. 1993;31(9):784–794.
- , . Trials of providing costing information to general practitioners: a systematic review. Med J Aust. 1997;167(2):89–92.
- . A review of physician cost‐containment strategies for laboratory testing. Med Care. 1983;21(8):783–802.
- , . The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non‐randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377–384.
- , , , et al. Does offering pricing information to resident physicians in the emergency department potentially reduce laboratory and radiology costs? Eur J Emerg Med. 2015;22:247–252.
- , , , et al. Cost and turn‐around time display decreases inpatient ordering of reference laboratory tests: a time series. BMJ Qual Saf. 2014;23:994–1000.
- , , , , . The impact of cost displays on primary care physician laboratory test ordering. J Gen Intern Med. 2014;29:708–714.
- , , , et al. Impact of providing fee data on laboratory test ordering: a controlled clinical trial. JAMA Intern Med. 2013;173(10):903–908.
- , , , . Provider cost transparency alone has no impact on inpatient imaging utilization. J Am Coll Radiol. 2013;10(2):108–113.
- , , . Providing clinicians with information on laboratory test costs leads to reduction in hospital expenditure. S Afr Med J. 2011;101(10):746–748.
- . Cutting costs: the impact of price lists on the cost development at the emergency department. Eur J Emerg Med. 2010;17(6):337–339.
- , , , , . Effects of price information on test ordering in an intensive care unit. Intens Care Med. 2002;28(3):332–335.
- , , , , , . Modifying provider behavior: a low‐tech approach to pharmaceutical ordering. J Gen Intern Med. 2002;17(10):792–796.
- , , , , . Medication cost information in a computer‐based patient record system. Impact on prescribing in a family medicine clinical practice. Arch Fam Med. 1999;8(2):118–121.
- , , , , . The effect of price information on test‐ordering behavior and patient outcomes in a pediatric emergency department. Pediatrics. 1999;103(4 pt 2):877–882.
- , , . Long‐term pharmaceutical cost reduction using a data management system. Anesth Analg. 1998;87(4):837–842.
- , . The impact of price labeling of muscle relaxants on cost consciousness among anesthesiologists. J Clin Anesth. 1998;10(5):401–403.
- , , . Does a computerized price comparison module reduce prescribing costs in general practice? Fam Pract. 1997;14(3):199–203.
- , , , et al. Does the computerized display of charges affect inpatient ancillary test utilization? Arch Intern Med. 1997;157(21):2501–2508.
- , . Price stickers do not alter drug usage. Can J Anaesth. 1994;41(11):1047–1052.
- , , , . Physician inpatient order writing on microcomputer workstations. Effects on resource utilization. JAMA. 1993;269(3):379–383.
- , , . The effect on test ordering of informing physicians of the charges for outpatient diagnostic tests. N Engl J Med. 1990;322(21):1499–1504.
- , , , . Effect of cost education, cost audits, and faculty chart review on the use of laboratory services. Arch Intern Med. 1983;143(5):942–944.
- . Interference between units in randomized experiments. J Am Stat Assoc. 2007;102(477):191–200.
Infections from endemic fungi, mycobacteria rare in patients on TNFIs
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: The incidence of select mycobacterial and fungal infections in patients taking TNFIs is low.
Major finding: Only 0.51% of patients taking TNFIs developed the mycobacterial and fungal infections targeted in this study.
Data source: A case-control study of 30,772 patients taking TNFIs.
Disclosures: The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
Thrombosis Management Demands Delicate, Balanced Approach
The delicate balance involved in providing hospitalized patients with needed anticoagulant, anti-platelet, and thrombolytic therapies for stroke and possible cardiac complications while minimizing bleed risks was explored by several speakers at the University of California San Francisco’s annual Management of the Hospitalized Patient Conference.
“These are dynamic issues and they’re moving all the time,” said Tracy Minichiello, MD, a former hospitalist who now runs the Anticoagulation and Thrombosis Service at the San Francisco VA Medical Center. Dosing and monitoring choices for physicians have grown more complicated with the new oral anticoagulants (apixaban, dabigatran, and rivaroxaban), and she said another balancing act is emerging in hospitals trying to avoid unnecessary and wasteful treatments.
“There is interest on both sides of that question,” Dr. Minichiello said, adding the stakes are high. “We don’t want to miss the diagnosis of pulmonary embolisms, which can be difficult to catch. But now there’s more discussion of the other side of the issue—over-diagnosis and over-treatment—where we’re also trying to avoid, for example, overuse of CT scans.”
Another major thrust of Dr. Minichiello’s presentations involved bridging therapies, the application of a parenteral, short-acting anticoagulant therapy during the temporary interruption of warfarin anticoagulation for an invasive procedure. Bridging decreases stroke and embolism risk, but with an increased risk for bleeding.
“Full intensity bridging therapy for anticoagulation potentially can do more harm than good,” she said, noting a dearth of data to support mortality benefits of bridging therapy.
Literature increasingly recommends hospitalists be more selective about the use of bridging therapies that might have been employed reflexively in the past, she noted.
“[Hospitalists] must be mindful of the risks and benefits,” she said.
Physicians should also think twice about concomitant antiplatelet therapy like aspirin with anticoagulants. “We need to work collaboratively with our cardiology colleagues when a patient is on two or three of these therapies,” she said. “Recommendations in this area are in evolution.”
Elise Bouchard, MD, an internist at Centre Maria-Chapdelaine in Dolbeau-Mistassini, Quebec, attended Dr. Minichiello’s breakout session on challenging cases.
“I learned that we shouldn’t use aspirin with Coumadin or other anticoagulants, except for cases like acute coronary syndrome,” Dr. Bouchard said. She also explained a number of her patients with cancer, for example, need anticoagulation treatment and hate getting another injection, so she tries when possible to offer the oral anticoagulants.
Dr. Minichiello works with hospitalists at the San Francisco VA who seek consults around procedures, anticoagulant choices, and when to restart treatments.
“Most hospitalists don’t have access to a service like ours, although they might be able to call on a hematology consult service [or pharmacist],” she said. She suggested hospitalists trying to develop their own evidenced-based protocols use websites like the University of Washington’s anticoagulation service website, or the American Society of Health System Pharmacists’ anticoagulation resource center. TH
The delicate balance involved in providing hospitalized patients with needed anticoagulant, anti-platelet, and thrombolytic therapies for stroke and possible cardiac complications while minimizing bleed risks was explored by several speakers at the University of California San Francisco’s annual Management of the Hospitalized Patient Conference.
“These are dynamic issues and they’re moving all the time,” said Tracy Minichiello, MD, a former hospitalist who now runs the Anticoagulation and Thrombosis Service at the San Francisco VA Medical Center. Dosing and monitoring choices for physicians have grown more complicated with the new oral anticoagulants (apixaban, dabigatran, and rivaroxaban), and she said another balancing act is emerging in hospitals trying to avoid unnecessary and wasteful treatments.
“There is interest on both sides of that question,” Dr. Minichiello said, adding the stakes are high. “We don’t want to miss the diagnosis of pulmonary embolisms, which can be difficult to catch. But now there’s more discussion of the other side of the issue—over-diagnosis and over-treatment—where we’re also trying to avoid, for example, overuse of CT scans.”
Another major thrust of Dr. Minichiello’s presentations involved bridging therapies, the application of a parenteral, short-acting anticoagulant therapy during the temporary interruption of warfarin anticoagulation for an invasive procedure. Bridging decreases stroke and embolism risk, but with an increased risk for bleeding.
“Full intensity bridging therapy for anticoagulation potentially can do more harm than good,” she said, noting a dearth of data to support mortality benefits of bridging therapy.
Literature increasingly recommends hospitalists be more selective about the use of bridging therapies that might have been employed reflexively in the past, she noted.
“[Hospitalists] must be mindful of the risks and benefits,” she said.
Physicians should also think twice about concomitant antiplatelet therapy like aspirin with anticoagulants. “We need to work collaboratively with our cardiology colleagues when a patient is on two or three of these therapies,” she said. “Recommendations in this area are in evolution.”
Elise Bouchard, MD, an internist at Centre Maria-Chapdelaine in Dolbeau-Mistassini, Quebec, attended Dr. Minichiello’s breakout session on challenging cases.
“I learned that we shouldn’t use aspirin with Coumadin or other anticoagulants, except for cases like acute coronary syndrome,” Dr. Bouchard said. She also explained a number of her patients with cancer, for example, need anticoagulation treatment and hate getting another injection, so she tries when possible to offer the oral anticoagulants.
Dr. Minichiello works with hospitalists at the San Francisco VA who seek consults around procedures, anticoagulant choices, and when to restart treatments.
“Most hospitalists don’t have access to a service like ours, although they might be able to call on a hematology consult service [or pharmacist],” she said. She suggested hospitalists trying to develop their own evidenced-based protocols use websites like the University of Washington’s anticoagulation service website, or the American Society of Health System Pharmacists’ anticoagulation resource center. TH
The delicate balance involved in providing hospitalized patients with needed anticoagulant, anti-platelet, and thrombolytic therapies for stroke and possible cardiac complications while minimizing bleed risks was explored by several speakers at the University of California San Francisco’s annual Management of the Hospitalized Patient Conference.
“These are dynamic issues and they’re moving all the time,” said Tracy Minichiello, MD, a former hospitalist who now runs the Anticoagulation and Thrombosis Service at the San Francisco VA Medical Center. Dosing and monitoring choices for physicians have grown more complicated with the new oral anticoagulants (apixaban, dabigatran, and rivaroxaban), and she said another balancing act is emerging in hospitals trying to avoid unnecessary and wasteful treatments.
“There is interest on both sides of that question,” Dr. Minichiello said, adding the stakes are high. “We don’t want to miss the diagnosis of pulmonary embolisms, which can be difficult to catch. But now there’s more discussion of the other side of the issue—over-diagnosis and over-treatment—where we’re also trying to avoid, for example, overuse of CT scans.”
Another major thrust of Dr. Minichiello’s presentations involved bridging therapies, the application of a parenteral, short-acting anticoagulant therapy during the temporary interruption of warfarin anticoagulation for an invasive procedure. Bridging decreases stroke and embolism risk, but with an increased risk for bleeding.
“Full intensity bridging therapy for anticoagulation potentially can do more harm than good,” she said, noting a dearth of data to support mortality benefits of bridging therapy.
Literature increasingly recommends hospitalists be more selective about the use of bridging therapies that might have been employed reflexively in the past, she noted.
“[Hospitalists] must be mindful of the risks and benefits,” she said.
Physicians should also think twice about concomitant antiplatelet therapy like aspirin with anticoagulants. “We need to work collaboratively with our cardiology colleagues when a patient is on two or three of these therapies,” she said. “Recommendations in this area are in evolution.”
Elise Bouchard, MD, an internist at Centre Maria-Chapdelaine in Dolbeau-Mistassini, Quebec, attended Dr. Minichiello’s breakout session on challenging cases.
“I learned that we shouldn’t use aspirin with Coumadin or other anticoagulants, except for cases like acute coronary syndrome,” Dr. Bouchard said. She also explained a number of her patients with cancer, for example, need anticoagulation treatment and hate getting another injection, so she tries when possible to offer the oral anticoagulants.
Dr. Minichiello works with hospitalists at the San Francisco VA who seek consults around procedures, anticoagulant choices, and when to restart treatments.
“Most hospitalists don’t have access to a service like ours, although they might be able to call on a hematology consult service [or pharmacist],” she said. She suggested hospitalists trying to develop their own evidenced-based protocols use websites like the University of Washington’s anticoagulation service website, or the American Society of Health System Pharmacists’ anticoagulation resource center. TH
Discussions about sexual orientation
The biological transition to puberty has always marked a critical point in a primary care pediatrician’s relationship to a patient. Adolescents’ capacity for abstract reasoning, their movement to autonomy, their nuanced sense of identity, their need for privacy, and their emerging sexuality together give the pediatrician an opportunity and a responsibility to create a safe place to talk. Your office can be an oasis from parents, peers, and a society that seems saturated with sexuality. You can be trusted more than the Internet and offer discussions that are leavened by your long-standing relationship with the patient.
The growing public awareness, acceptance, and legal standing given to gay, lesbian, bisexual, and transgender individuals represents welcome societal progress, and we sense that amidst this richer public conversation, a growing number of children and adolescents are presenting with questions or worries about their own emerging sexual orientation or gender identity. We would like to start with our key takeaway: Discussions about sexual orientation and gender identity do not require that you give answers or predict the future. Focus instead on being a curious, compassionate, and nonjudgmental listener, and you will be effective at helping your patient to better manage new, uncertain, and possibly stressful feelings.
Our focus today is how to create a safe setting and specifically how to ask about and discuss sexual orientation. Most teenagers will wonder at some point about their orientation. Studies suggest that among adults, 5%-10% are attracted to the same sex and 3% describe themselves as gay or bisexual. Such surveys are very challenging, though, and in our experience, these percentages are higher. Sexual orientation is believed to exist on a continuum rather than in a simple binary state – some people identify as purely homosexual or heterosexual, and the rest exist somewhere in the middle. Sharing this fact alone can offer a very helpful perspective to young people who are feeling pressure to “figure out” if they are gay or straight.
While sexual orientation describes whom a person is attracted to, gender identity is a person’s internal sense of his or her own gender. It emerges in childhood and becomes more rich and nuanced in adolescence and adulthood, and, like sexual orientation, it is also believed to exist on a continuum rather than in a simple binary state. Less than 0.1% of youth will experience gender dysphoria, or the pressing feeling that their gender identity is not the same as their phenotypic sex. While questions about gender identity should be approached with the same curious, compassionate, and nonjudgmental style, we will not discuss the management of patients with gender dysphoria here. It is a very complex (and controversial) topic. And, as a practical matter, sexual orientation will likely be a more common issue with your patients, whereas questions about gender identity will come up much less frequently.
It is worth knowing that there is a range of mental health issues that are associated with the stress of feeling comfortable with one’s sexual identity. There is some evidence that young people who identify as gay or bisexual have elevated risk for mood disorders (depression), anxiety disorders, conduct disorder, and substance use disorders, but this finding has not been consistent (Am J Public Health. 2010:100[12]; 2426-32). However, there is unequivocal evidence that there is an elevated risk for suicide attempts in lesbian, gay, or bisexual (LGB) youth above their heterosexual peers. One survey found that 9th-12th grade students who identified as LGB were up to seven times as likely to have a suicide attempt as were their peers who identify as heterosexual. This risk is especially pronounced in male adolescents and continues into adulthood, when there is an elevated risk for suicide completion among adult males who identify as homosexual, although not in adult females (J Homosex. 2011 Jan;58[1]:10-51). Importantly, the risk for suicide attempt in LGB adolescents remains elevated even in those adolescents without any diagnosable mental illness, likely attributable to the stresses of isolation, family conflict, stigmatization, or bullying that LGB adolescents are likely to experience.
Asking your early-adolescent patients in a calm and comfortable manner about sexual feelings builds an environment in which thoughts, feelings, and questions about sex and sexuality are more easily shared. It is important to find language that feels like yours, which you can use with ease. Perhaps starting with, “At about your age, I ask every patient of mine whether they are beginning to have sexual feelings. This is when you really want to be around someone, in a way that’s more powerful and different from even your favorite friend. Some people call it getting butterflies in your stomach.” If your patient recognizes what you are talking about, you might continue, “Do you feel attracted to boys or girls or both? Do you have those feelings about kids in your class or people you know, like a teacher? Perhaps about a celebrity in a TV show or a band?” You should absolutely reassure them, “You don’t have to talk about anything you do not want to, but you should know that this is a normal part of becoming a teenager. I talk about this a lot with patients who are younger and older than you are. I keep what we talk about very private, and sometimes this is the only place a teenager feels safe to ask questions.” If you start this process early enough – by the start of middle school – the patient will probably be a bit embarrassed or giggle and not talk much. But, by the next annual physical or the one after that, the issue will be more familiar and less charged. A meaningful discussion may start.
With patients who do describe feeling attracted to people of the same sex, more specific questions may be appropriate. You should expect these feelings to exist on a continuum: You may encounter a school-age child with great clarity about exactly whom she is attracted to and what that means, or an older teenager who is far less certain, responding to a less intense interest or having been told by a peer that he is probably gay. It can be powerfully reassuring to remind your patient that adolescence is when we start to figure out to whom we are attracted. They don’t have to decide, but just be aware of these feelings as they emerge, essentially getting to know themselves without any feelings of urgency or pressure. You might ask, “Have you wondered if you were gay or bisexual? Have you spoken to any friends about your feelings or have you experimented with a boy or girl to try and figure this out?” It’s very helpful if you ask if they are worried or stressed by these feelings. Some young people will suffer from internalized homophobia, which may be helped by your accepting stance or may require a referral for more ongoing support. It can be valuable to find out if they are dating people of the same sex, or if their “relationships” have all been online. While this “virtual” dating may seem safer, it may not help them better understand themselves and may expose them to exploitation or predatory behavior. If your patient is sexually active, you should be comfortable talking with them about the risks of unprotected sex and same-sex safe-sex practices.
It is particularly important to ask your adolescent LGB patient about whom they have told, and what responses have they gotten. The presence of good friends and loving family members is critical to all adolescents’ emotional well-being. If they have talked about their sexual orientation with their peers, have their friends been supportive, or has it left them more isolated at school? You should find out if they have been teased or bullied, and ask specifically about online teasing or harassment. If they are being bullied, how have they handled that? Find out also if they feel free to ask or talk about this subject with their parents. If not, try to understand if they are simply embarrassed and unsure how to bring it up, or if there is a strong sense that they will be shamed or even rejected by their parents. If the parents are truly shaming or rejecting, it will be critical to consider what kind of support may be necessary. Teens who are facing isolation or bullying at school may benefit from resources such as a gay-straight student alliance or a community organization dedicated to issues facing LGB youth. For patients who are facing hostile or rejecting parents, it can be protective to connect them with a therapist as well, as you are mindful of their marked isolation and subsequently heightened risk for mood problems and even suicide attempts.
Along a similar vein, it is very important that you are aware of your own comfort level with these issues. While discussing sexual orientation may feel awkward if it is new for you, it is important to be realistic if you cannot be supportive of your patients who are gay. If for religious or other reasons you are not comfortable talking about sexual orientation in an accepting, nonjudgmental manner, you should seek guidance on how to thoughtfully care for your LGB patients or appropriately refer them to someone who can provide a more-supportive treatment setting.
When you create an office that makes sexuality a safe topic for discussion, you should expect that you will hear questions or concerns about which you yourself may not know the answers. Do not panic, just maintain your posture of being a curious, compassionate, and nonjudgmental listener, and then look for the answers. We are delighted this news organization has devoted a column to the optimal care of LGBT youth (LGBT Youth Consult) and encourage the primary care pediatrician to “never to worry alone,” and instead get some advice and expert teammates when dealing with these complex and important issues.
Dr. Swick is an attending psychiatrist in the division of child psychiatry at Massachusetts General Hospital, Boston, and director of the Parenting at a Challenging Time (PACT) Program at the Vernon Cancer Center at Newton Wellesley Hospital, in Newton, Mass. Dr. Jellinek is professor of psychiatry and of pediatrics at Harvard Medical School, Boston.
The biological transition to puberty has always marked a critical point in a primary care pediatrician’s relationship to a patient. Adolescents’ capacity for abstract reasoning, their movement to autonomy, their nuanced sense of identity, their need for privacy, and their emerging sexuality together give the pediatrician an opportunity and a responsibility to create a safe place to talk. Your office can be an oasis from parents, peers, and a society that seems saturated with sexuality. You can be trusted more than the Internet and offer discussions that are leavened by your long-standing relationship with the patient.
The growing public awareness, acceptance, and legal standing given to gay, lesbian, bisexual, and transgender individuals represents welcome societal progress, and we sense that amidst this richer public conversation, a growing number of children and adolescents are presenting with questions or worries about their own emerging sexual orientation or gender identity. We would like to start with our key takeaway: Discussions about sexual orientation and gender identity do not require that you give answers or predict the future. Focus instead on being a curious, compassionate, and nonjudgmental listener, and you will be effective at helping your patient to better manage new, uncertain, and possibly stressful feelings.
Our focus today is how to create a safe setting and specifically how to ask about and discuss sexual orientation. Most teenagers will wonder at some point about their orientation. Studies suggest that among adults, 5%-10% are attracted to the same sex and 3% describe themselves as gay or bisexual. Such surveys are very challenging, though, and in our experience, these percentages are higher. Sexual orientation is believed to exist on a continuum rather than in a simple binary state – some people identify as purely homosexual or heterosexual, and the rest exist somewhere in the middle. Sharing this fact alone can offer a very helpful perspective to young people who are feeling pressure to “figure out” if they are gay or straight.
While sexual orientation describes whom a person is attracted to, gender identity is a person’s internal sense of his or her own gender. It emerges in childhood and becomes more rich and nuanced in adolescence and adulthood, and, like sexual orientation, it is also believed to exist on a continuum rather than in a simple binary state. Less than 0.1% of youth will experience gender dysphoria, or the pressing feeling that their gender identity is not the same as their phenotypic sex. While questions about gender identity should be approached with the same curious, compassionate, and nonjudgmental style, we will not discuss the management of patients with gender dysphoria here. It is a very complex (and controversial) topic. And, as a practical matter, sexual orientation will likely be a more common issue with your patients, whereas questions about gender identity will come up much less frequently.
It is worth knowing that there is a range of mental health issues that are associated with the stress of feeling comfortable with one’s sexual identity. There is some evidence that young people who identify as gay or bisexual have elevated risk for mood disorders (depression), anxiety disorders, conduct disorder, and substance use disorders, but this finding has not been consistent (Am J Public Health. 2010:100[12]; 2426-32). However, there is unequivocal evidence that there is an elevated risk for suicide attempts in lesbian, gay, or bisexual (LGB) youth above their heterosexual peers. One survey found that 9th-12th grade students who identified as LGB were up to seven times as likely to have a suicide attempt as were their peers who identify as heterosexual. This risk is especially pronounced in male adolescents and continues into adulthood, when there is an elevated risk for suicide completion among adult males who identify as homosexual, although not in adult females (J Homosex. 2011 Jan;58[1]:10-51). Importantly, the risk for suicide attempt in LGB adolescents remains elevated even in those adolescents without any diagnosable mental illness, likely attributable to the stresses of isolation, family conflict, stigmatization, or bullying that LGB adolescents are likely to experience.
Asking your early-adolescent patients in a calm and comfortable manner about sexual feelings builds an environment in which thoughts, feelings, and questions about sex and sexuality are more easily shared. It is important to find language that feels like yours, which you can use with ease. Perhaps starting with, “At about your age, I ask every patient of mine whether they are beginning to have sexual feelings. This is when you really want to be around someone, in a way that’s more powerful and different from even your favorite friend. Some people call it getting butterflies in your stomach.” If your patient recognizes what you are talking about, you might continue, “Do you feel attracted to boys or girls or both? Do you have those feelings about kids in your class or people you know, like a teacher? Perhaps about a celebrity in a TV show or a band?” You should absolutely reassure them, “You don’t have to talk about anything you do not want to, but you should know that this is a normal part of becoming a teenager. I talk about this a lot with patients who are younger and older than you are. I keep what we talk about very private, and sometimes this is the only place a teenager feels safe to ask questions.” If you start this process early enough – by the start of middle school – the patient will probably be a bit embarrassed or giggle and not talk much. But, by the next annual physical or the one after that, the issue will be more familiar and less charged. A meaningful discussion may start.
With patients who do describe feeling attracted to people of the same sex, more specific questions may be appropriate. You should expect these feelings to exist on a continuum: You may encounter a school-age child with great clarity about exactly whom she is attracted to and what that means, or an older teenager who is far less certain, responding to a less intense interest or having been told by a peer that he is probably gay. It can be powerfully reassuring to remind your patient that adolescence is when we start to figure out to whom we are attracted. They don’t have to decide, but just be aware of these feelings as they emerge, essentially getting to know themselves without any feelings of urgency or pressure. You might ask, “Have you wondered if you were gay or bisexual? Have you spoken to any friends about your feelings or have you experimented with a boy or girl to try and figure this out?” It’s very helpful if you ask if they are worried or stressed by these feelings. Some young people will suffer from internalized homophobia, which may be helped by your accepting stance or may require a referral for more ongoing support. It can be valuable to find out if they are dating people of the same sex, or if their “relationships” have all been online. While this “virtual” dating may seem safer, it may not help them better understand themselves and may expose them to exploitation or predatory behavior. If your patient is sexually active, you should be comfortable talking with them about the risks of unprotected sex and same-sex safe-sex practices.
It is particularly important to ask your adolescent LGB patient about whom they have told, and what responses have they gotten. The presence of good friends and loving family members is critical to all adolescents’ emotional well-being. If they have talked about their sexual orientation with their peers, have their friends been supportive, or has it left them more isolated at school? You should find out if they have been teased or bullied, and ask specifically about online teasing or harassment. If they are being bullied, how have they handled that? Find out also if they feel free to ask or talk about this subject with their parents. If not, try to understand if they are simply embarrassed and unsure how to bring it up, or if there is a strong sense that they will be shamed or even rejected by their parents. If the parents are truly shaming or rejecting, it will be critical to consider what kind of support may be necessary. Teens who are facing isolation or bullying at school may benefit from resources such as a gay-straight student alliance or a community organization dedicated to issues facing LGB youth. For patients who are facing hostile or rejecting parents, it can be protective to connect them with a therapist as well, as you are mindful of their marked isolation and subsequently heightened risk for mood problems and even suicide attempts.
Along a similar vein, it is very important that you are aware of your own comfort level with these issues. While discussing sexual orientation may feel awkward if it is new for you, it is important to be realistic if you cannot be supportive of your patients who are gay. If for religious or other reasons you are not comfortable talking about sexual orientation in an accepting, nonjudgmental manner, you should seek guidance on how to thoughtfully care for your LGB patients or appropriately refer them to someone who can provide a more-supportive treatment setting.
When you create an office that makes sexuality a safe topic for discussion, you should expect that you will hear questions or concerns about which you yourself may not know the answers. Do not panic, just maintain your posture of being a curious, compassionate, and nonjudgmental listener, and then look for the answers. We are delighted this news organization has devoted a column to the optimal care of LGBT youth (LGBT Youth Consult) and encourage the primary care pediatrician to “never to worry alone,” and instead get some advice and expert teammates when dealing with these complex and important issues.
Dr. Swick is an attending psychiatrist in the division of child psychiatry at Massachusetts General Hospital, Boston, and director of the Parenting at a Challenging Time (PACT) Program at the Vernon Cancer Center at Newton Wellesley Hospital, in Newton, Mass. Dr. Jellinek is professor of psychiatry and of pediatrics at Harvard Medical School, Boston.
The biological transition to puberty has always marked a critical point in a primary care pediatrician’s relationship to a patient. Adolescents’ capacity for abstract reasoning, their movement to autonomy, their nuanced sense of identity, their need for privacy, and their emerging sexuality together give the pediatrician an opportunity and a responsibility to create a safe place to talk. Your office can be an oasis from parents, peers, and a society that seems saturated with sexuality. You can be trusted more than the Internet and offer discussions that are leavened by your long-standing relationship with the patient.
The growing public awareness, acceptance, and legal standing given to gay, lesbian, bisexual, and transgender individuals represents welcome societal progress, and we sense that amidst this richer public conversation, a growing number of children and adolescents are presenting with questions or worries about their own emerging sexual orientation or gender identity. We would like to start with our key takeaway: Discussions about sexual orientation and gender identity do not require that you give answers or predict the future. Focus instead on being a curious, compassionate, and nonjudgmental listener, and you will be effective at helping your patient to better manage new, uncertain, and possibly stressful feelings.
Our focus today is how to create a safe setting and specifically how to ask about and discuss sexual orientation. Most teenagers will wonder at some point about their orientation. Studies suggest that among adults, 5%-10% are attracted to the same sex and 3% describe themselves as gay or bisexual. Such surveys are very challenging, though, and in our experience, these percentages are higher. Sexual orientation is believed to exist on a continuum rather than in a simple binary state – some people identify as purely homosexual or heterosexual, and the rest exist somewhere in the middle. Sharing this fact alone can offer a very helpful perspective to young people who are feeling pressure to “figure out” if they are gay or straight.
While sexual orientation describes whom a person is attracted to, gender identity is a person’s internal sense of his or her own gender. It emerges in childhood and becomes more rich and nuanced in adolescence and adulthood, and, like sexual orientation, it is also believed to exist on a continuum rather than in a simple binary state. Less than 0.1% of youth will experience gender dysphoria, or the pressing feeling that their gender identity is not the same as their phenotypic sex. While questions about gender identity should be approached with the same curious, compassionate, and nonjudgmental style, we will not discuss the management of patients with gender dysphoria here. It is a very complex (and controversial) topic. And, as a practical matter, sexual orientation will likely be a more common issue with your patients, whereas questions about gender identity will come up much less frequently.
It is worth knowing that there is a range of mental health issues that are associated with the stress of feeling comfortable with one’s sexual identity. There is some evidence that young people who identify as gay or bisexual have elevated risk for mood disorders (depression), anxiety disorders, conduct disorder, and substance use disorders, but this finding has not been consistent (Am J Public Health. 2010:100[12]; 2426-32). However, there is unequivocal evidence that there is an elevated risk for suicide attempts in lesbian, gay, or bisexual (LGB) youth above their heterosexual peers. One survey found that 9th-12th grade students who identified as LGB were up to seven times as likely to have a suicide attempt as were their peers who identify as heterosexual. This risk is especially pronounced in male adolescents and continues into adulthood, when there is an elevated risk for suicide completion among adult males who identify as homosexual, although not in adult females (J Homosex. 2011 Jan;58[1]:10-51). Importantly, the risk for suicide attempt in LGB adolescents remains elevated even in those adolescents without any diagnosable mental illness, likely attributable to the stresses of isolation, family conflict, stigmatization, or bullying that LGB adolescents are likely to experience.
Asking your early-adolescent patients in a calm and comfortable manner about sexual feelings builds an environment in which thoughts, feelings, and questions about sex and sexuality are more easily shared. It is important to find language that feels like yours, which you can use with ease. Perhaps starting with, “At about your age, I ask every patient of mine whether they are beginning to have sexual feelings. This is when you really want to be around someone, in a way that’s more powerful and different from even your favorite friend. Some people call it getting butterflies in your stomach.” If your patient recognizes what you are talking about, you might continue, “Do you feel attracted to boys or girls or both? Do you have those feelings about kids in your class or people you know, like a teacher? Perhaps about a celebrity in a TV show or a band?” You should absolutely reassure them, “You don’t have to talk about anything you do not want to, but you should know that this is a normal part of becoming a teenager. I talk about this a lot with patients who are younger and older than you are. I keep what we talk about very private, and sometimes this is the only place a teenager feels safe to ask questions.” If you start this process early enough – by the start of middle school – the patient will probably be a bit embarrassed or giggle and not talk much. But, by the next annual physical or the one after that, the issue will be more familiar and less charged. A meaningful discussion may start.
With patients who do describe feeling attracted to people of the same sex, more specific questions may be appropriate. You should expect these feelings to exist on a continuum: You may encounter a school-age child with great clarity about exactly whom she is attracted to and what that means, or an older teenager who is far less certain, responding to a less intense interest or having been told by a peer that he is probably gay. It can be powerfully reassuring to remind your patient that adolescence is when we start to figure out to whom we are attracted. They don’t have to decide, but just be aware of these feelings as they emerge, essentially getting to know themselves without any feelings of urgency or pressure. You might ask, “Have you wondered if you were gay or bisexual? Have you spoken to any friends about your feelings or have you experimented with a boy or girl to try and figure this out?” It’s very helpful if you ask if they are worried or stressed by these feelings. Some young people will suffer from internalized homophobia, which may be helped by your accepting stance or may require a referral for more ongoing support. It can be valuable to find out if they are dating people of the same sex, or if their “relationships” have all been online. While this “virtual” dating may seem safer, it may not help them better understand themselves and may expose them to exploitation or predatory behavior. If your patient is sexually active, you should be comfortable talking with them about the risks of unprotected sex and same-sex safe-sex practices.
It is particularly important to ask your adolescent LGB patient about whom they have told, and what responses have they gotten. The presence of good friends and loving family members is critical to all adolescents’ emotional well-being. If they have talked about their sexual orientation with their peers, have their friends been supportive, or has it left them more isolated at school? You should find out if they have been teased or bullied, and ask specifically about online teasing or harassment. If they are being bullied, how have they handled that? Find out also if they feel free to ask or talk about this subject with their parents. If not, try to understand if they are simply embarrassed and unsure how to bring it up, or if there is a strong sense that they will be shamed or even rejected by their parents. If the parents are truly shaming or rejecting, it will be critical to consider what kind of support may be necessary. Teens who are facing isolation or bullying at school may benefit from resources such as a gay-straight student alliance or a community organization dedicated to issues facing LGB youth. For patients who are facing hostile or rejecting parents, it can be protective to connect them with a therapist as well, as you are mindful of their marked isolation and subsequently heightened risk for mood problems and even suicide attempts.
Along a similar vein, it is very important that you are aware of your own comfort level with these issues. While discussing sexual orientation may feel awkward if it is new for you, it is important to be realistic if you cannot be supportive of your patients who are gay. If for religious or other reasons you are not comfortable talking about sexual orientation in an accepting, nonjudgmental manner, you should seek guidance on how to thoughtfully care for your LGB patients or appropriately refer them to someone who can provide a more-supportive treatment setting.
When you create an office that makes sexuality a safe topic for discussion, you should expect that you will hear questions or concerns about which you yourself may not know the answers. Do not panic, just maintain your posture of being a curious, compassionate, and nonjudgmental listener, and then look for the answers. We are delighted this news organization has devoted a column to the optimal care of LGBT youth (LGBT Youth Consult) and encourage the primary care pediatrician to “never to worry alone,” and instead get some advice and expert teammates when dealing with these complex and important issues.
Dr. Swick is an attending psychiatrist in the division of child psychiatry at Massachusetts General Hospital, Boston, and director of the Parenting at a Challenging Time (PACT) Program at the Vernon Cancer Center at Newton Wellesley Hospital, in Newton, Mass. Dr. Jellinek is professor of psychiatry and of pediatrics at Harvard Medical School, Boston.
EADV: Comorbid spondyloarthropathy common in hidradenitis suppurativa
COPENHAGEN – Back pain is surprisingly common in patients with hidradenitis suppurativa, and more than half of affected patients showed MRI evidence of axial spondyloarthropathy, Dr. Sylke Schneider-Burrus reported at the Annual Congress of the European Academy of Dermatology and Venereology.
“Our study demonstrates that back pain and spondyloarthropathy are very common among hidradenitis suppurativa patients and that neither history nor clinical parameters provide any hints for the presence of spondyloarthropathy. Therefore, we strongly suggest that hidradenitis suppurativa patients should be evaluated for spondyloarthropathy and affected patients should be treated systemically with TNF-alpha blockers in order to avoid chronic joint alterations,” said Dr. Schneider-Burrus, a dermatologist at Charite University Hospital in Berlin.
Hidradenitis suppurativa (HS) is a chronic, recurrent, scarring, inflammatory skin disease of the hair follicles. It causes painful, purulent, foul-smelling fistulating sinuses in the axillae, groin, and perianal region.
Because several other chronic inflammatory diseases affecting epithelial tissue have been associated with increased rates of axial spondyloarthropathy – notably, Crohn’s disease, ulcerative colitis, and psoriasis – Dr. Schneider-Burrus and coinvestigators wondered whether that might true of HS as well.
She presented a survey of 100 HS patients. To her surprise, fully 71% indicated they suffer from back pain, with lower back complaints predominating.
Forty-eight HS patients with back pain consented to undergo a pelvic MRI exam. Fifteen of the 48 (32%) showed clear MRI evidence of spondyloarthropathy, including sacroiliac erosions and subchondral sclerosis, while another 12 showed active sacroiliac synovitis and other acute inflammatory changes.
No significant differences were found between HS patients with and without axial spondyloarthropathy in terms of age at onset of HS, disease duration, HS severity as reflected in Sartorius score, age at MRI, body mass index, or smoking status.
Dr. Schneider-Burrus reported serving as a paid investigator for and consultant to Novartis and AbbVie.
COPENHAGEN – Back pain is surprisingly common in patients with hidradenitis suppurativa, and more than half of affected patients showed MRI evidence of axial spondyloarthropathy, Dr. Sylke Schneider-Burrus reported at the Annual Congress of the European Academy of Dermatology and Venereology.
“Our study demonstrates that back pain and spondyloarthropathy are very common among hidradenitis suppurativa patients and that neither history nor clinical parameters provide any hints for the presence of spondyloarthropathy. Therefore, we strongly suggest that hidradenitis suppurativa patients should be evaluated for spondyloarthropathy and affected patients should be treated systemically with TNF-alpha blockers in order to avoid chronic joint alterations,” said Dr. Schneider-Burrus, a dermatologist at Charite University Hospital in Berlin.
Hidradenitis suppurativa (HS) is a chronic, recurrent, scarring, inflammatory skin disease of the hair follicles. It causes painful, purulent, foul-smelling fistulating sinuses in the axillae, groin, and perianal region.
Because several other chronic inflammatory diseases affecting epithelial tissue have been associated with increased rates of axial spondyloarthropathy – notably, Crohn’s disease, ulcerative colitis, and psoriasis – Dr. Schneider-Burrus and coinvestigators wondered whether that might true of HS as well.
She presented a survey of 100 HS patients. To her surprise, fully 71% indicated they suffer from back pain, with lower back complaints predominating.
Forty-eight HS patients with back pain consented to undergo a pelvic MRI exam. Fifteen of the 48 (32%) showed clear MRI evidence of spondyloarthropathy, including sacroiliac erosions and subchondral sclerosis, while another 12 showed active sacroiliac synovitis and other acute inflammatory changes.
No significant differences were found between HS patients with and without axial spondyloarthropathy in terms of age at onset of HS, disease duration, HS severity as reflected in Sartorius score, age at MRI, body mass index, or smoking status.
Dr. Schneider-Burrus reported serving as a paid investigator for and consultant to Novartis and AbbVie.
COPENHAGEN – Back pain is surprisingly common in patients with hidradenitis suppurativa, and more than half of affected patients showed MRI evidence of axial spondyloarthropathy, Dr. Sylke Schneider-Burrus reported at the Annual Congress of the European Academy of Dermatology and Venereology.
“Our study demonstrates that back pain and spondyloarthropathy are very common among hidradenitis suppurativa patients and that neither history nor clinical parameters provide any hints for the presence of spondyloarthropathy. Therefore, we strongly suggest that hidradenitis suppurativa patients should be evaluated for spondyloarthropathy and affected patients should be treated systemically with TNF-alpha blockers in order to avoid chronic joint alterations,” said Dr. Schneider-Burrus, a dermatologist at Charite University Hospital in Berlin.
Hidradenitis suppurativa (HS) is a chronic, recurrent, scarring, inflammatory skin disease of the hair follicles. It causes painful, purulent, foul-smelling fistulating sinuses in the axillae, groin, and perianal region.
Because several other chronic inflammatory diseases affecting epithelial tissue have been associated with increased rates of axial spondyloarthropathy – notably, Crohn’s disease, ulcerative colitis, and psoriasis – Dr. Schneider-Burrus and coinvestigators wondered whether that might true of HS as well.
She presented a survey of 100 HS patients. To her surprise, fully 71% indicated they suffer from back pain, with lower back complaints predominating.
Forty-eight HS patients with back pain consented to undergo a pelvic MRI exam. Fifteen of the 48 (32%) showed clear MRI evidence of spondyloarthropathy, including sacroiliac erosions and subchondral sclerosis, while another 12 showed active sacroiliac synovitis and other acute inflammatory changes.
No significant differences were found between HS patients with and without axial spondyloarthropathy in terms of age at onset of HS, disease duration, HS severity as reflected in Sartorius score, age at MRI, body mass index, or smoking status.
Dr. Schneider-Burrus reported serving as a paid investigator for and consultant to Novartis and AbbVie.
AT THE EADV CONGRESS
Key clinical point: Axial spondyloarthropathy is extremely common in patients with hidradenitis suppurativa.
Major finding: Seventy-one percent of surveyed hidradenitis suppurativa patients reported suffering from back pain, and 56% of affected patients showed MRI evidence of axial spondyloarthropathy.
Data source: A back pain survey of 100 patients with hidradenitis suppurativa along with pelvic MRI exams in the 48 who reported back pain.
Disclosures: The presenter reported serving as a paid investigator for and consultant to Novartis and AbbVie.
Managing menopausal symptoms after risk-reducing salpingo-oophorectomy
Compared to the general population, women with mutations in the BRCA1 or BRCA2 genes have a significantly higher lifetime risk of ovarian and breast cancers (Science. 2003 Oct 24;302[5645]:643-6). Since the occurrence of ovarian and breast cancer in BRCA carriers is often prior to menopause, and because we have no screening test to detect early stage ovarian cancer, risk-reducing salpingo-oophorectomy has been recommended around age 40.
It has been shown that risk-reducing salpingo-oophorectomy significantly reduces ovarian cancer risk by 85%-95% in BRCA-affected women. Also, this surgery can reduce breast cancer risk by 53%-68% (N Engl J Med. 2002 May 23;346[21]:1609-15). The 2008 Practice Bulletin from the American College of Obstetricians and Gynecologists recommends that risk-reducing salpingo-oophorectomy should be performed in women with BRCA1 or BRCA2 mutations after the completion of childbearing or age 40 (Obstet Gynecol. 2008 Jan;111[1]:231-41).
Health implications
Nearly 60% of women who have a BRCA1 or BRCA2 mutation will elect to undergo risk-reducing salpingo-oophorectomy between the ages of 35 and 40 years (Open Med. 2007 Aug 13;1[2]:e92-8). As such, surgical menopause can result in hot flashes, vaginal dryness, sexual dysfunction, sleep disturbances, and cognitive changes, which may significantly impact a woman’s quality of life. In addition, increased risk of cardiovascular disease and osteoporosis following bilateral salpingo-oophorectomy may have a significant impact on a woman’s health.
Since these women undergo surgical menopause as opposed to natural menopause, they have an abrupt loss in hormones, and due to their younger age at the time of surgery, they may also have a longer exposure period to the detrimental effects of hypoestrogenism.
Symptom management
Various treatment options exist for relief of menopausal symptoms, including nonhormonal therapies and hormone replacement therapies (HT).
Nonhormonal therapies include serotonin receptor inhibitors (venlafaxine and paroxetine) and alpha-2 adrenergic agonists (clonidine), which are most appropriate for the treatment of vasomotor symptoms. Unfortunately, these options have proved to be as effective as HT. Also, women should be adequately counseled regarding the various side effects of these nonhormonal medications. Alternative approaches such as phytoestrogens are unproven and are still undergoing investigation. As such, HT remains the standard for treatment of menopausal symptoms, and many trials have confirmed that HT can effectively treat menopausal symptoms following risk-reducing salpingo-oophorectomy.
This then raises the question of safety regarding use of HT in this patient population; especially the possibility of increased risk of breast cancer. Interestingly, only 10%-25% of BRCA1 carriers will have estrogen receptor–positive breast cancer, while 65%-79% of BRCA2-associated breast cancers will be positive for the receptor (Clin Cancer Res. 2004 Mar 15;10[6]:2029-34).
Unfortunately, we do not have adequate trials or studies with sufficient long-term follow-up to validate whether HT increases the risk of breast cancer or recurrence. However, the PROSE Study Group did report on a prospective cohort of 462 women with BRCA1 or BRCA2 mutations. In this study, HT did not alter the reduction in breast cancer risk from risk-reducing salpingo-oophorectomy (J Clin Oncol. 2005 Nov 1;23[31]:7804-10). In addition to a paucity of data regarding systemic HT, there is little data in the BRCA-positive population to confirm the safety of local vaginal estrogen for treatment of vaginal atrophy (J Clin Oncol. 2004 Mar 15;22[6]:1045-54).
Understanding the options
Use of HT in women with BRCA1 and BRCA2 mutations requires further investigation. There should be shared decision making between the patient and provider when counseling on the management of menopausal symptoms following risk-reducing salpingo-oophorectomy. Most importantly, women should understand the options of nonhormonal therapies and their specific side effects. They should also understand the lack of significant data regarding use of systemic HT, and that if use is elected, there may be an increased risk of breast cancer.
Women who do elect to use systemic HT following risk-reducing salpingo-oophorectomy have options that can help reduce the risk of HT-associated breast cancer, including a shorter duration of systemic HT or concurrent hysterectomy to allow for estrogen-only HT, which has a decreased risk of breast cancer compared with combined therapies that include progestins. These women may also be considering prophylactic mastectomy, which would change the concerns regarding HT and an increased risk of breast cancer.
Increased awareness of these options among physicians and patients alike can help to decrease unsatisfactory symptoms and improve quality of life in women undergoing risk-reducing salpingo-oophorectomy.
Dr. Staley is a resident physician in the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at the university. They reported having no relevant financial disclosures. Email them at obnews@frontlinemedcom.com.
Compared to the general population, women with mutations in the BRCA1 or BRCA2 genes have a significantly higher lifetime risk of ovarian and breast cancers (Science. 2003 Oct 24;302[5645]:643-6). Since the occurrence of ovarian and breast cancer in BRCA carriers is often prior to menopause, and because we have no screening test to detect early stage ovarian cancer, risk-reducing salpingo-oophorectomy has been recommended around age 40.
It has been shown that risk-reducing salpingo-oophorectomy significantly reduces ovarian cancer risk by 85%-95% in BRCA-affected women. Also, this surgery can reduce breast cancer risk by 53%-68% (N Engl J Med. 2002 May 23;346[21]:1609-15). The 2008 Practice Bulletin from the American College of Obstetricians and Gynecologists recommends that risk-reducing salpingo-oophorectomy should be performed in women with BRCA1 or BRCA2 mutations after the completion of childbearing or age 40 (Obstet Gynecol. 2008 Jan;111[1]:231-41).
Health implications
Nearly 60% of women who have a BRCA1 or BRCA2 mutation will elect to undergo risk-reducing salpingo-oophorectomy between the ages of 35 and 40 years (Open Med. 2007 Aug 13;1[2]:e92-8). As such, surgical menopause can result in hot flashes, vaginal dryness, sexual dysfunction, sleep disturbances, and cognitive changes, which may significantly impact a woman’s quality of life. In addition, increased risk of cardiovascular disease and osteoporosis following bilateral salpingo-oophorectomy may have a significant impact on a woman’s health.
Since these women undergo surgical menopause as opposed to natural menopause, they have an abrupt loss in hormones, and due to their younger age at the time of surgery, they may also have a longer exposure period to the detrimental effects of hypoestrogenism.
Symptom management
Various treatment options exist for relief of menopausal symptoms, including nonhormonal therapies and hormone replacement therapies (HT).
Nonhormonal therapies include serotonin receptor inhibitors (venlafaxine and paroxetine) and alpha-2 adrenergic agonists (clonidine), which are most appropriate for the treatment of vasomotor symptoms. Unfortunately, these options have proved to be as effective as HT. Also, women should be adequately counseled regarding the various side effects of these nonhormonal medications. Alternative approaches such as phytoestrogens are unproven and are still undergoing investigation. As such, HT remains the standard for treatment of menopausal symptoms, and many trials have confirmed that HT can effectively treat menopausal symptoms following risk-reducing salpingo-oophorectomy.
This then raises the question of safety regarding use of HT in this patient population; especially the possibility of increased risk of breast cancer. Interestingly, only 10%-25% of BRCA1 carriers will have estrogen receptor–positive breast cancer, while 65%-79% of BRCA2-associated breast cancers will be positive for the receptor (Clin Cancer Res. 2004 Mar 15;10[6]:2029-34).
Unfortunately, we do not have adequate trials or studies with sufficient long-term follow-up to validate whether HT increases the risk of breast cancer or recurrence. However, the PROSE Study Group did report on a prospective cohort of 462 women with BRCA1 or BRCA2 mutations. In this study, HT did not alter the reduction in breast cancer risk from risk-reducing salpingo-oophorectomy (J Clin Oncol. 2005 Nov 1;23[31]:7804-10). In addition to a paucity of data regarding systemic HT, there is little data in the BRCA-positive population to confirm the safety of local vaginal estrogen for treatment of vaginal atrophy (J Clin Oncol. 2004 Mar 15;22[6]:1045-54).
Understanding the options
Use of HT in women with BRCA1 and BRCA2 mutations requires further investigation. There should be shared decision making between the patient and provider when counseling on the management of menopausal symptoms following risk-reducing salpingo-oophorectomy. Most importantly, women should understand the options of nonhormonal therapies and their specific side effects. They should also understand the lack of significant data regarding use of systemic HT, and that if use is elected, there may be an increased risk of breast cancer.
Women who do elect to use systemic HT following risk-reducing salpingo-oophorectomy have options that can help reduce the risk of HT-associated breast cancer, including a shorter duration of systemic HT or concurrent hysterectomy to allow for estrogen-only HT, which has a decreased risk of breast cancer compared with combined therapies that include progestins. These women may also be considering prophylactic mastectomy, which would change the concerns regarding HT and an increased risk of breast cancer.
Increased awareness of these options among physicians and patients alike can help to decrease unsatisfactory symptoms and improve quality of life in women undergoing risk-reducing salpingo-oophorectomy.
Dr. Staley is a resident physician in the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at the university. They reported having no relevant financial disclosures. Email them at obnews@frontlinemedcom.com.
Compared to the general population, women with mutations in the BRCA1 or BRCA2 genes have a significantly higher lifetime risk of ovarian and breast cancers (Science. 2003 Oct 24;302[5645]:643-6). Since the occurrence of ovarian and breast cancer in BRCA carriers is often prior to menopause, and because we have no screening test to detect early stage ovarian cancer, risk-reducing salpingo-oophorectomy has been recommended around age 40.
It has been shown that risk-reducing salpingo-oophorectomy significantly reduces ovarian cancer risk by 85%-95% in BRCA-affected women. Also, this surgery can reduce breast cancer risk by 53%-68% (N Engl J Med. 2002 May 23;346[21]:1609-15). The 2008 Practice Bulletin from the American College of Obstetricians and Gynecologists recommends that risk-reducing salpingo-oophorectomy should be performed in women with BRCA1 or BRCA2 mutations after the completion of childbearing or age 40 (Obstet Gynecol. 2008 Jan;111[1]:231-41).
Health implications
Nearly 60% of women who have a BRCA1 or BRCA2 mutation will elect to undergo risk-reducing salpingo-oophorectomy between the ages of 35 and 40 years (Open Med. 2007 Aug 13;1[2]:e92-8). As such, surgical menopause can result in hot flashes, vaginal dryness, sexual dysfunction, sleep disturbances, and cognitive changes, which may significantly impact a woman’s quality of life. In addition, increased risk of cardiovascular disease and osteoporosis following bilateral salpingo-oophorectomy may have a significant impact on a woman’s health.
Since these women undergo surgical menopause as opposed to natural menopause, they have an abrupt loss in hormones, and due to their younger age at the time of surgery, they may also have a longer exposure period to the detrimental effects of hypoestrogenism.
Symptom management
Various treatment options exist for relief of menopausal symptoms, including nonhormonal therapies and hormone replacement therapies (HT).
Nonhormonal therapies include serotonin receptor inhibitors (venlafaxine and paroxetine) and alpha-2 adrenergic agonists (clonidine), which are most appropriate for the treatment of vasomotor symptoms. Unfortunately, these options have proved to be as effective as HT. Also, women should be adequately counseled regarding the various side effects of these nonhormonal medications. Alternative approaches such as phytoestrogens are unproven and are still undergoing investigation. As such, HT remains the standard for treatment of menopausal symptoms, and many trials have confirmed that HT can effectively treat menopausal symptoms following risk-reducing salpingo-oophorectomy.
This then raises the question of safety regarding use of HT in this patient population; especially the possibility of increased risk of breast cancer. Interestingly, only 10%-25% of BRCA1 carriers will have estrogen receptor–positive breast cancer, while 65%-79% of BRCA2-associated breast cancers will be positive for the receptor (Clin Cancer Res. 2004 Mar 15;10[6]:2029-34).
Unfortunately, we do not have adequate trials or studies with sufficient long-term follow-up to validate whether HT increases the risk of breast cancer or recurrence. However, the PROSE Study Group did report on a prospective cohort of 462 women with BRCA1 or BRCA2 mutations. In this study, HT did not alter the reduction in breast cancer risk from risk-reducing salpingo-oophorectomy (J Clin Oncol. 2005 Nov 1;23[31]:7804-10). In addition to a paucity of data regarding systemic HT, there is little data in the BRCA-positive population to confirm the safety of local vaginal estrogen for treatment of vaginal atrophy (J Clin Oncol. 2004 Mar 15;22[6]:1045-54).
Understanding the options
Use of HT in women with BRCA1 and BRCA2 mutations requires further investigation. There should be shared decision making between the patient and provider when counseling on the management of menopausal symptoms following risk-reducing salpingo-oophorectomy. Most importantly, women should understand the options of nonhormonal therapies and their specific side effects. They should also understand the lack of significant data regarding use of systemic HT, and that if use is elected, there may be an increased risk of breast cancer.
Women who do elect to use systemic HT following risk-reducing salpingo-oophorectomy have options that can help reduce the risk of HT-associated breast cancer, including a shorter duration of systemic HT or concurrent hysterectomy to allow for estrogen-only HT, which has a decreased risk of breast cancer compared with combined therapies that include progestins. These women may also be considering prophylactic mastectomy, which would change the concerns regarding HT and an increased risk of breast cancer.
Increased awareness of these options among physicians and patients alike can help to decrease unsatisfactory symptoms and improve quality of life in women undergoing risk-reducing salpingo-oophorectomy.
Dr. Staley is a resident physician in the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at the university. They reported having no relevant financial disclosures. Email them at obnews@frontlinemedcom.com.
Low incidence of DVT reported after percutaneous EVAR
Completely percutaneous endovascular aortic aneurysm repair (PEVAR) has become more common, using the suture-mediated “preclose” technique. The rate of periprocedural, iatrogenic, acute deep vein thrombosis (DVT), hitherto unknown, was found to be low for this approach, according to a study reported by Dr. Courtney E. Morgan and her colleagues at the Northwestern University, Chicago.
The researchers assessed 52 consecutive patients (44 men) with a mean age of 73 years, who underwent PEVAR at their center. Only 6% had a prior history of DVT (J Vasc Surg. 2015 Aug; 62:351-4).
Acute DVT was seen in four patients on postoperative day 1. These four DVTs comprised one femoropopliteal, and three calf DVTs. Three of these patients had associated risk factors: history of DVT (two patients); active smokers (one patient); and obesity (body mass index greater than 30 kg/m2 in all three patients).
At 2 weeks postoperatively, 75% of the DVTs had resolved.
“We found an overall rate of proximal DVT of 4% after PEVAR, which increased to 13% when calf-vein DVTs were included. Most patients with postoperative DVT had preexisting risk factors, which suggests that routine duplex ultrasound screening after PEVAR is not necessary unless there exist preclinical risk factors or postprocedural clinical indications suggestive of DVT,” the authors concluded.
Two of the researchers have received funding and/or served as speakers/consultants for device companies involved in EVAR.
Read the full study online in the Journal of Vascular Surgery.
Completely percutaneous endovascular aortic aneurysm repair (PEVAR) has become more common, using the suture-mediated “preclose” technique. The rate of periprocedural, iatrogenic, acute deep vein thrombosis (DVT), hitherto unknown, was found to be low for this approach, according to a study reported by Dr. Courtney E. Morgan and her colleagues at the Northwestern University, Chicago.
The researchers assessed 52 consecutive patients (44 men) with a mean age of 73 years, who underwent PEVAR at their center. Only 6% had a prior history of DVT (J Vasc Surg. 2015 Aug; 62:351-4).
Acute DVT was seen in four patients on postoperative day 1. These four DVTs comprised one femoropopliteal, and three calf DVTs. Three of these patients had associated risk factors: history of DVT (two patients); active smokers (one patient); and obesity (body mass index greater than 30 kg/m2 in all three patients).
At 2 weeks postoperatively, 75% of the DVTs had resolved.
“We found an overall rate of proximal DVT of 4% after PEVAR, which increased to 13% when calf-vein DVTs were included. Most patients with postoperative DVT had preexisting risk factors, which suggests that routine duplex ultrasound screening after PEVAR is not necessary unless there exist preclinical risk factors or postprocedural clinical indications suggestive of DVT,” the authors concluded.
Two of the researchers have received funding and/or served as speakers/consultants for device companies involved in EVAR.
Read the full study online in the Journal of Vascular Surgery.
Completely percutaneous endovascular aortic aneurysm repair (PEVAR) has become more common, using the suture-mediated “preclose” technique. The rate of periprocedural, iatrogenic, acute deep vein thrombosis (DVT), hitherto unknown, was found to be low for this approach, according to a study reported by Dr. Courtney E. Morgan and her colleagues at the Northwestern University, Chicago.
The researchers assessed 52 consecutive patients (44 men) with a mean age of 73 years, who underwent PEVAR at their center. Only 6% had a prior history of DVT (J Vasc Surg. 2015 Aug; 62:351-4).
Acute DVT was seen in four patients on postoperative day 1. These four DVTs comprised one femoropopliteal, and three calf DVTs. Three of these patients had associated risk factors: history of DVT (two patients); active smokers (one patient); and obesity (body mass index greater than 30 kg/m2 in all three patients).
At 2 weeks postoperatively, 75% of the DVTs had resolved.
“We found an overall rate of proximal DVT of 4% after PEVAR, which increased to 13% when calf-vein DVTs were included. Most patients with postoperative DVT had preexisting risk factors, which suggests that routine duplex ultrasound screening after PEVAR is not necessary unless there exist preclinical risk factors or postprocedural clinical indications suggestive of DVT,” the authors concluded.
Two of the researchers have received funding and/or served as speakers/consultants for device companies involved in EVAR.
Read the full study online in the Journal of Vascular Surgery.
FROM THE JOURNAL OF VASCULAR SURGERY
Will New American Cancer Society Mammography Policy Change VHA Practice?
The new American Cancer Society (ACS) policy for mammography screening now recommends annual mammograms from age 45 until age 54, suggesting a less aggressive approach to screening than in the 2003 guidelines. The recommendation is for asymptomatic women at average risk of developing breast cancer.
Related: How Much Is Too Much Cancer Screening?
“These recommendations are made with the intent of maximizing reductions in breast cancer mortality and years of life saved while being attentive to the need to minimize harms associated with screening,” said Dr. Kevin C. Oeffinger, chairman of the breast cancer guideline panel.
Related: Advances in Targeted Therapy for Breast Cancer
The guideline, developed by a panel of outside experts, was published October 20, 2015, in The Journal of the American Medical Association.
Other important recommendation changes include:
- ŸWomen aged 45 to 54 years should receive annual screening mammography and at age 55 women should transition to biennial screening.
- ŸWomen should continue screening mammography as long as their overall health is good and they have a life expectancy of 10 years or longer.
- ŸClinical breast examination is no longer recommended at any age.
- ŸThe panel noted that the recommendations are an effort to avoid false positive findings, which can take an emotional toll.
The current VHA recommendations state:
- Mammography is not recommended for women aged 13 to 39 years.
- Women aged 40 to 49 years should talk with their health care providers about screening.
- ŸMammograms are recommended every 2 years for women aged 50 to 74 years.
- ŸWomen aged > 75 years should talk with their health care providers.
According to Anita Aggarwal, DO, PhD, president of the Association of VA Hematology and Oncology (AVAHO), the VHA recommendations leave a lot of wiggle room; every year starting at age 40 is still acceptable practice at the VA.
Related: Breast Cancer Research Group Aims to Improve Veteran Survival Rates
“Keep in mind that recommendations are just recommendations,” Dr. Aggarwal told Federal Practitioner. “One size may not fit all. I believe we should use our sound judgment based on valid research to recommend what is best for the patient. Based on my literature search, annual screening starting at 40 saves the most lives via early detection.”
The new American Cancer Society (ACS) policy for mammography screening now recommends annual mammograms from age 45 until age 54, suggesting a less aggressive approach to screening than in the 2003 guidelines. The recommendation is for asymptomatic women at average risk of developing breast cancer.
Related: How Much Is Too Much Cancer Screening?
“These recommendations are made with the intent of maximizing reductions in breast cancer mortality and years of life saved while being attentive to the need to minimize harms associated with screening,” said Dr. Kevin C. Oeffinger, chairman of the breast cancer guideline panel.
Related: Advances in Targeted Therapy for Breast Cancer
The guideline, developed by a panel of outside experts, was published October 20, 2015, in The Journal of the American Medical Association.
Other important recommendation changes include:
- ŸWomen aged 45 to 54 years should receive annual screening mammography and at age 55 women should transition to biennial screening.
- ŸWomen should continue screening mammography as long as their overall health is good and they have a life expectancy of 10 years or longer.
- ŸClinical breast examination is no longer recommended at any age.
- ŸThe panel noted that the recommendations are an effort to avoid false positive findings, which can take an emotional toll.
The current VHA recommendations state:
- Mammography is not recommended for women aged 13 to 39 years.
- Women aged 40 to 49 years should talk with their health care providers about screening.
- ŸMammograms are recommended every 2 years for women aged 50 to 74 years.
- ŸWomen aged > 75 years should talk with their health care providers.
According to Anita Aggarwal, DO, PhD, president of the Association of VA Hematology and Oncology (AVAHO), the VHA recommendations leave a lot of wiggle room; every year starting at age 40 is still acceptable practice at the VA.
Related: Breast Cancer Research Group Aims to Improve Veteran Survival Rates
“Keep in mind that recommendations are just recommendations,” Dr. Aggarwal told Federal Practitioner. “One size may not fit all. I believe we should use our sound judgment based on valid research to recommend what is best for the patient. Based on my literature search, annual screening starting at 40 saves the most lives via early detection.”
The new American Cancer Society (ACS) policy for mammography screening now recommends annual mammograms from age 45 until age 54, suggesting a less aggressive approach to screening than in the 2003 guidelines. The recommendation is for asymptomatic women at average risk of developing breast cancer.
Related: How Much Is Too Much Cancer Screening?
“These recommendations are made with the intent of maximizing reductions in breast cancer mortality and years of life saved while being attentive to the need to minimize harms associated with screening,” said Dr. Kevin C. Oeffinger, chairman of the breast cancer guideline panel.
Related: Advances in Targeted Therapy for Breast Cancer
The guideline, developed by a panel of outside experts, was published October 20, 2015, in The Journal of the American Medical Association.
Other important recommendation changes include:
- ŸWomen aged 45 to 54 years should receive annual screening mammography and at age 55 women should transition to biennial screening.
- ŸWomen should continue screening mammography as long as their overall health is good and they have a life expectancy of 10 years or longer.
- ŸClinical breast examination is no longer recommended at any age.
- ŸThe panel noted that the recommendations are an effort to avoid false positive findings, which can take an emotional toll.
The current VHA recommendations state:
- Mammography is not recommended for women aged 13 to 39 years.
- Women aged 40 to 49 years should talk with their health care providers about screening.
- ŸMammograms are recommended every 2 years for women aged 50 to 74 years.
- ŸWomen aged > 75 years should talk with their health care providers.
According to Anita Aggarwal, DO, PhD, president of the Association of VA Hematology and Oncology (AVAHO), the VHA recommendations leave a lot of wiggle room; every year starting at age 40 is still acceptable practice at the VA.
Related: Breast Cancer Research Group Aims to Improve Veteran Survival Rates
“Keep in mind that recommendations are just recommendations,” Dr. Aggarwal told Federal Practitioner. “One size may not fit all. I believe we should use our sound judgment based on valid research to recommend what is best for the patient. Based on my literature search, annual screening starting at 40 saves the most lives via early detection.”
No flu vaccine for patients with egg allergy?
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
Percutaneous ethanol effective for small papillary thyroid cancers
LAKE BUENA VISTA, FLA. – An outpatient procedure may represent an efficacious and safe alternative to surgery for those patients with small papillary thyroid cancers who prefer definitive treatment over the “wait and watch” approach. Further, at one institution, the cost-effective alternative to surgery saved almost $40,000 per patient.
Ultrasound-guided percutaneous ethanol injection (UPEA) of small (cT1N0) intrathyroidal papillary thyroid cancer (SIPC) successfully reduced tumor volume by a median of 92%, eliminated tumor blood flow, and was very well tolerated by a series of 13 patients who received UPEA at the Mayo Clinic, Rochester, Minn.
Dr. Ian D. Hay, a consultant in Mayo’s division of endocrinology, diabetes, metabolism, and nutrition, presented the findings during a poster session at the International Thyroid Congress.
Dr. Hay and his colleagues treated 13 patients with a total of 15 tumors with injections of percutaneous ethanol. The first patient received just one injection; the remaining patients received one injection to each tumor site on each of 2 consecutive days. Five of the tumor foci had less than a 50% reduction in tumor volume at the first follow-up visit, so those tumors were injected a third time.
Patients in the series ranged from 38 to 86 years old (median 45), and five patients had significant comorbidities: one had congestive heart failure and the other four had concomitant unrelated cancers. Tumors were a median 8 mm in size, with volumes ranging from 25 to 676 mm3 (median 140 mm3).
All of the injections were performed under ultrasound guidance, and a median of 0.9 cc of ethanol was injected into each tumor. Ultrasound examination was performed at each follow-up visit to evaluate tumor volume and blood flow. Dr. Hay reported that the procedure was well tolerated: Local neck tenderness resolved within a day or two, and there were no reports of hoarseness or laryngeal nerve palsy.
Patients were followed for a mean 2.0 years (range, 0.4-5.7 years), with a median tumor reduction of 92% (range 46%-100%). For the nine tumors that were still identifiable on ultrasound at the time of reporting, the mean volume had decreased by 73%. Six tumor foci had completely disappeared, and no tumor had detectable blood flow on Doppler exam. Tumor thyroglobulin levels remained stable in all patients, and no nodal metastases were identified, Dr. Hay reported at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Internationally, the approach to managing SIPC varies from lobectomy to near-total thyroidectomy to active surveillance. For patients who prefer definitive management of their tumors but are reluctant to have surgery or who may have significant comorbidities, UPEA may represent a safe alternative, and at significant cost savings compared to surgery: Dr. Hay and his colleagues reported that they estimated the average cost savings at their institution to be over $38,000 per patient. “If prospective trials of observation vs. surgery for SIPC are to occur in the USA, perhaps it could be included as a ‘third arm’ in such trials,” Dr. Hay and his colleagues said.
On Twitter @karioakes
LAKE BUENA VISTA, FLA. – An outpatient procedure may represent an efficacious and safe alternative to surgery for those patients with small papillary thyroid cancers who prefer definitive treatment over the “wait and watch” approach. Further, at one institution, the cost-effective alternative to surgery saved almost $40,000 per patient.
Ultrasound-guided percutaneous ethanol injection (UPEA) of small (cT1N0) intrathyroidal papillary thyroid cancer (SIPC) successfully reduced tumor volume by a median of 92%, eliminated tumor blood flow, and was very well tolerated by a series of 13 patients who received UPEA at the Mayo Clinic, Rochester, Minn.
Dr. Ian D. Hay, a consultant in Mayo’s division of endocrinology, diabetes, metabolism, and nutrition, presented the findings during a poster session at the International Thyroid Congress.
Dr. Hay and his colleagues treated 13 patients with a total of 15 tumors with injections of percutaneous ethanol. The first patient received just one injection; the remaining patients received one injection to each tumor site on each of 2 consecutive days. Five of the tumor foci had less than a 50% reduction in tumor volume at the first follow-up visit, so those tumors were injected a third time.
Patients in the series ranged from 38 to 86 years old (median 45), and five patients had significant comorbidities: one had congestive heart failure and the other four had concomitant unrelated cancers. Tumors were a median 8 mm in size, with volumes ranging from 25 to 676 mm3 (median 140 mm3).
All of the injections were performed under ultrasound guidance, and a median of 0.9 cc of ethanol was injected into each tumor. Ultrasound examination was performed at each follow-up visit to evaluate tumor volume and blood flow. Dr. Hay reported that the procedure was well tolerated: Local neck tenderness resolved within a day or two, and there were no reports of hoarseness or laryngeal nerve palsy.
Patients were followed for a mean 2.0 years (range, 0.4-5.7 years), with a median tumor reduction of 92% (range 46%-100%). For the nine tumors that were still identifiable on ultrasound at the time of reporting, the mean volume had decreased by 73%. Six tumor foci had completely disappeared, and no tumor had detectable blood flow on Doppler exam. Tumor thyroglobulin levels remained stable in all patients, and no nodal metastases were identified, Dr. Hay reported at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Internationally, the approach to managing SIPC varies from lobectomy to near-total thyroidectomy to active surveillance. For patients who prefer definitive management of their tumors but are reluctant to have surgery or who may have significant comorbidities, UPEA may represent a safe alternative, and at significant cost savings compared to surgery: Dr. Hay and his colleagues reported that they estimated the average cost savings at their institution to be over $38,000 per patient. “If prospective trials of observation vs. surgery for SIPC are to occur in the USA, perhaps it could be included as a ‘third arm’ in such trials,” Dr. Hay and his colleagues said.
On Twitter @karioakes
LAKE BUENA VISTA, FLA. – An outpatient procedure may represent an efficacious and safe alternative to surgery for those patients with small papillary thyroid cancers who prefer definitive treatment over the “wait and watch” approach. Further, at one institution, the cost-effective alternative to surgery saved almost $40,000 per patient.
Ultrasound-guided percutaneous ethanol injection (UPEA) of small (cT1N0) intrathyroidal papillary thyroid cancer (SIPC) successfully reduced tumor volume by a median of 92%, eliminated tumor blood flow, and was very well tolerated by a series of 13 patients who received UPEA at the Mayo Clinic, Rochester, Minn.
Dr. Ian D. Hay, a consultant in Mayo’s division of endocrinology, diabetes, metabolism, and nutrition, presented the findings during a poster session at the International Thyroid Congress.
Dr. Hay and his colleagues treated 13 patients with a total of 15 tumors with injections of percutaneous ethanol. The first patient received just one injection; the remaining patients received one injection to each tumor site on each of 2 consecutive days. Five of the tumor foci had less than a 50% reduction in tumor volume at the first follow-up visit, so those tumors were injected a third time.
Patients in the series ranged from 38 to 86 years old (median 45), and five patients had significant comorbidities: one had congestive heart failure and the other four had concomitant unrelated cancers. Tumors were a median 8 mm in size, with volumes ranging from 25 to 676 mm3 (median 140 mm3).
All of the injections were performed under ultrasound guidance, and a median of 0.9 cc of ethanol was injected into each tumor. Ultrasound examination was performed at each follow-up visit to evaluate tumor volume and blood flow. Dr. Hay reported that the procedure was well tolerated: Local neck tenderness resolved within a day or two, and there were no reports of hoarseness or laryngeal nerve palsy.
Patients were followed for a mean 2.0 years (range, 0.4-5.7 years), with a median tumor reduction of 92% (range 46%-100%). For the nine tumors that were still identifiable on ultrasound at the time of reporting, the mean volume had decreased by 73%. Six tumor foci had completely disappeared, and no tumor had detectable blood flow on Doppler exam. Tumor thyroglobulin levels remained stable in all patients, and no nodal metastases were identified, Dr. Hay reported at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Internationally, the approach to managing SIPC varies from lobectomy to near-total thyroidectomy to active surveillance. For patients who prefer definitive management of their tumors but are reluctant to have surgery or who may have significant comorbidities, UPEA may represent a safe alternative, and at significant cost savings compared to surgery: Dr. Hay and his colleagues reported that they estimated the average cost savings at their institution to be over $38,000 per patient. “If prospective trials of observation vs. surgery for SIPC are to occur in the USA, perhaps it could be included as a ‘third arm’ in such trials,” Dr. Hay and his colleagues said.
On Twitter @karioakes
AT ITC 2015
Key clinical point: Ultrasound-guided percutaneous ethanol ablation (UPEA) is an efficacious, cost-effective, and noninvasive definitive treatment for small papillary thyroid cancers.
Major finding: Fifteen tumors in 13 patients were successfully treated with UPEA with a mean 92% reduction in tumor volume and no complications or metastasis at a mean 2-year follow-up.
Data source: Series of 13 patients with 15 tumors treated at the Mayo Clinic for small intrathyroidal papillary cancers.
Disclosures: No disclosures were identified.