User login
Looking up patients online: Why it’s a bad idea
Searching for someone on the Internet and viewing his or her social media profile is an effective way to obtain information about people, including patients. Following our patients’ “digital footprint” may help us understand the context of their lives, reconcile discrepancies in what they have told us, or allow us to confront denial and address incomplete reporting.1 However, perusing our patients’ online profiles could negatively impact treatment and adherence. Consider these factors before looking up your patients’ online profiles1-3:
Inaccurate information. Information on the Internet, especially what you can find on user-generated forums, is largely unregulated; as a result, the veracity of that information cannot be guaranteed.1 Patients may choose to portray themselves inaccurately on their online profiles, and their identities often cannot be confirmed. Even if some information is accurate, you might discover things that you did not expect to learn about your patients, including important information that they did not share, or even something they lied about. This can create the conundrums of what to do with such information and how to discuss it at the next visit.
Impact on treatment. Despite patients’ online activities being displayed for the world to see, many patients do not expect their clinicians to access their online information. They might perceive such perusal as a breach of trust, which might lead some to view the doctor–patient relationship as adversarial. Accessing this information also could create a more intimate relationship than intended. Even if a clinician acquires consent to perform a search, patients may still feel coerced into allowing it because they might feel that declining to grant permission would make the clinician suspect that they have something to hide, or that the clinician would search without consent.2
In addition, if patients are aware that their psychiatrists are monitoring them, they might change their behavior. For example, they may delete certain data, add additional information that may not be accurate, or censor future social media posts. Knowing that their clinicians could be paying attention to them around the clock also might motivate certain patients to act out more or become withdrawn.
Possible medicolegal repercussions. If clinicians are able to access their patients’ electronic profiles, are they then legally obligated to monitor them? For example, if a patient who posts a picture with a noose around his neck later completes suicide, does the clinician who intermittently monitored this patient’s online profile face legal ramifications for not seeing the post? Do clinicians have to call 911 for vaguely suicidal tweets? What responsibilities does a clinician have at the first sign of an innocuous “sad” emoji? The sheer volume of online content that patients can create over different outlets is staggering. It can be overwhelming and ineffective to attempt to monitor patients’ online activities in addition to attending to one’s usual clinical duties, and the medicolegal repercussions of doing so are largely unknown.
Before searching the Internet to learn more about your patients, first consider the ramifications of doing so. While such searches could be helpful, they may lead to poor adherence, a lack of trust, or legal quagmires.
1. Fisher CE, Appelbaum PS. Beyond Googling: the ethics of using patients’ electronic footprints in psychiatric practice. Harv Rev Psychiatry. 2017;25(4):170-179.
2. Ashby GA, O’Brien A, Bowman
3. Cox-George C. The changing face(book) of psychiatry: can we justify ‘following’ patients’ social media activity? BJPsych Bulletin. 2015;39(6):284-285.
Searching for someone on the Internet and viewing his or her social media profile is an effective way to obtain information about people, including patients. Following our patients’ “digital footprint” may help us understand the context of their lives, reconcile discrepancies in what they have told us, or allow us to confront denial and address incomplete reporting.1 However, perusing our patients’ online profiles could negatively impact treatment and adherence. Consider these factors before looking up your patients’ online profiles1-3:
Inaccurate information. Information on the Internet, especially what you can find on user-generated forums, is largely unregulated; as a result, the veracity of that information cannot be guaranteed.1 Patients may choose to portray themselves inaccurately on their online profiles, and their identities often cannot be confirmed. Even if some information is accurate, you might discover things that you did not expect to learn about your patients, including important information that they did not share, or even something they lied about. This can create the conundrums of what to do with such information and how to discuss it at the next visit.
Impact on treatment. Despite patients’ online activities being displayed for the world to see, many patients do not expect their clinicians to access their online information. They might perceive such perusal as a breach of trust, which might lead some to view the doctor–patient relationship as adversarial. Accessing this information also could create a more intimate relationship than intended. Even if a clinician acquires consent to perform a search, patients may still feel coerced into allowing it because they might feel that declining to grant permission would make the clinician suspect that they have something to hide, or that the clinician would search without consent.2
In addition, if patients are aware that their psychiatrists are monitoring them, they might change their behavior. For example, they may delete certain data, add additional information that may not be accurate, or censor future social media posts. Knowing that their clinicians could be paying attention to them around the clock also might motivate certain patients to act out more or become withdrawn.
Possible medicolegal repercussions. If clinicians are able to access their patients’ electronic profiles, are they then legally obligated to monitor them? For example, if a patient who posts a picture with a noose around his neck later completes suicide, does the clinician who intermittently monitored this patient’s online profile face legal ramifications for not seeing the post? Do clinicians have to call 911 for vaguely suicidal tweets? What responsibilities does a clinician have at the first sign of an innocuous “sad” emoji? The sheer volume of online content that patients can create over different outlets is staggering. It can be overwhelming and ineffective to attempt to monitor patients’ online activities in addition to attending to one’s usual clinical duties, and the medicolegal repercussions of doing so are largely unknown.
Before searching the Internet to learn more about your patients, first consider the ramifications of doing so. While such searches could be helpful, they may lead to poor adherence, a lack of trust, or legal quagmires.
Searching for someone on the Internet and viewing his or her social media profile is an effective way to obtain information about people, including patients. Following our patients’ “digital footprint” may help us understand the context of their lives, reconcile discrepancies in what they have told us, or allow us to confront denial and address incomplete reporting.1 However, perusing our patients’ online profiles could negatively impact treatment and adherence. Consider these factors before looking up your patients’ online profiles1-3:
Inaccurate information. Information on the Internet, especially what you can find on user-generated forums, is largely unregulated; as a result, the veracity of that information cannot be guaranteed.1 Patients may choose to portray themselves inaccurately on their online profiles, and their identities often cannot be confirmed. Even if some information is accurate, you might discover things that you did not expect to learn about your patients, including important information that they did not share, or even something they lied about. This can create the conundrums of what to do with such information and how to discuss it at the next visit.
Impact on treatment. Despite patients’ online activities being displayed for the world to see, many patients do not expect their clinicians to access their online information. They might perceive such perusal as a breach of trust, which might lead some to view the doctor–patient relationship as adversarial. Accessing this information also could create a more intimate relationship than intended. Even if a clinician acquires consent to perform a search, patients may still feel coerced into allowing it because they might feel that declining to grant permission would make the clinician suspect that they have something to hide, or that the clinician would search without consent.2
In addition, if patients are aware that their psychiatrists are monitoring them, they might change their behavior. For example, they may delete certain data, add additional information that may not be accurate, or censor future social media posts. Knowing that their clinicians could be paying attention to them around the clock also might motivate certain patients to act out more or become withdrawn.
Possible medicolegal repercussions. If clinicians are able to access their patients’ electronic profiles, are they then legally obligated to monitor them? For example, if a patient who posts a picture with a noose around his neck later completes suicide, does the clinician who intermittently monitored this patient’s online profile face legal ramifications for not seeing the post? Do clinicians have to call 911 for vaguely suicidal tweets? What responsibilities does a clinician have at the first sign of an innocuous “sad” emoji? The sheer volume of online content that patients can create over different outlets is staggering. It can be overwhelming and ineffective to attempt to monitor patients’ online activities in addition to attending to one’s usual clinical duties, and the medicolegal repercussions of doing so are largely unknown.
Before searching the Internet to learn more about your patients, first consider the ramifications of doing so. While such searches could be helpful, they may lead to poor adherence, a lack of trust, or legal quagmires.
1. Fisher CE, Appelbaum PS. Beyond Googling: the ethics of using patients’ electronic footprints in psychiatric practice. Harv Rev Psychiatry. 2017;25(4):170-179.
2. Ashby GA, O’Brien A, Bowman
3. Cox-George C. The changing face(book) of psychiatry: can we justify ‘following’ patients’ social media activity? BJPsych Bulletin. 2015;39(6):284-285.
1. Fisher CE, Appelbaum PS. Beyond Googling: the ethics of using patients’ electronic footprints in psychiatric practice. Harv Rev Psychiatry. 2017;25(4):170-179.
2. Ashby GA, O’Brien A, Bowman
3. Cox-George C. The changing face(book) of psychiatry: can we justify ‘following’ patients’ social media activity? BJPsych Bulletin. 2015;39(6):284-285.
Novel risk factors for febrile neutropenia in NHL, other cancers
A retrospective study has revealed new potential risk factors for chemotherapy-induced febrile neutropenia (FN) in patients with solid tumors and non-Hodgkin lymphoma (NHL).
Researchers found the timing and duration of corticosteroid use were both associated with FN.
The team also observed “marginal” associations between FN and certain dermatologic and mucosal conditions as well as the use of intravenous (IV) antibiotics before chemotherapy.
On the other hand, there was no association between oral antibiotic use and FN or between radiation therapy (RT) and FN.
Chun Rebecca Chao, PhD, of Kaiser Permanente Southern California in Pasadena, and her colleagues reported these findings in JNCCN.
“Febrile neutropenia is life-threatening and often requires hospitalization,” Dr. Chao noted. “Furthermore, FN can lead to chemotherapy dose delay and dose reduction, which, in turn, negatively impacts antitumor efficacy. However, it can be prevented if high-risk individuals are identified and treated prophylactically.”
With this in mind, Dr. Chao and her colleagues set out to identify novel risk factors for FN by analyzing 15,971 patients who were treated with myelosuppressive chemotherapy at Kaiser Permanente Southern California between 2000 and 2009.
Patients had been diagnosed with NHL (n=1,617) or breast (n=6,323), lung (n=3,584), colorectal (n=3,062), ovarian (n=924), or gastric (n=461) cancers.
In all, 4.3% of patients developed FN during their first cycle of chemotherapy.
Corticosteroid use
The researchers found corticosteroid use was associated with an increased risk of FN in a propensity score-adjusted (PSA) model (adjusted for age, sex, socioeconomic factors, comorbidities, etc.). The hazard ratio (HR) was 1.53 (95% CI, 1.17-1.98; P<0.01) for patients who received corticosteroids.
A longer duration of corticosteroid use was associated with a greater risk of FN. The adjusted HR (compared to no corticosteroid use) was:
- 1.78 for corticosteroid treatment lasting less than 15 days (P<0.01)
- 1.84 for treatment lasting 15 to 29 days (P<0.01)
- 2.27 for treatment lasting 30 to 44 days (P<0.01)
- 2.86 for treatment lasting 45 to 90 days (P<0.01).
More recent corticosteroid use was associated with a greater risk of FN as well. The adjusted HR was:
- 1.88 for corticosteroid treatment less than 15 days before chemotherapy (P<0.01)
- 1.13 for treatment 15 to 29 days before chemotherapy (P=0.72)
- 1.22 for treatment 30 to 44 days before chemotherapy (P=0.66)
- 1.41 for treatment 45 to 90 days before chemotherapy (P=0.32).
“One way to reduce the incidence rate for FN could be to schedule prior corticosteroid use and subsequent chemotherapy with at least 2 weeks between them, given the magnitude of the risk increase and prevalence of this risk factor,” Dr. Chao said.
Other potential risk factors
The researchers found a “marginally” increased risk of FN in patients with certain dermatologic conditions (dermatitis, psoriasis, pruritus, etc.) and mucosal conditions (gastritis, stomatitis, mucositis, etc.).
In the PSA model, the HR was 1.40 (95% CI, 0.98-1.93; P=0.05) for patients with these conditions.
IV antibiotic use was also found to be marginally associated with an increased risk of FN in a restricted analysis covering patients treated in 2008 and 2009. In the PSA model, the HR was 1.35 (95% CI, 0.97-1.87; P=0.08).
On the other hand, there was no association between FN and oral antibiotic use in the restricted analysis. In the PSA model, the HR was 1.07 (95% CI, 0.77-1.48; P=0.70) for patients who received oral antibiotics.
Dr. Chao and her colleagues said these results suggest IV antibiotics may have a more profound impact than oral antibiotics on the balance of bacterial flora and other immune functions. Another possible explanation is that patients who received IV antibiotics were generally sicker and more prone to severe infection than patients who received oral antibiotics.
As with oral antibiotics, the researchers found no association between FN and the following factors (with the PSA model):
- Prior surgery (HR=0.89; 95% CI, 0.72-1.11; P=0.30)
- Prior RT (HR=0.91; 95% CI, 0.64-1.27; P=0.61)
- Concurrent RT (HR=1.32; 95% CI, 0.69-2.37; P=0.37).
The researchers noted that they did not account for radiation field or dose in this study, so additional evaluation of RT as a risk factor is needed.
In closing, Dr. Chao and her colleagues said these results suggest corticosteroid use, IV antibiotics, and certain dermatologic and mucosal conditions should be taken into consideration when monitoring patients receiving myelosuppressive chemotherapy and when evaluating the need for prophylactic granulocyte colony-stimulating factor or chemotherapy dose reduction.
Dr. Chao and her colleagues received funding from Amgen, Inc., to perform this study.
A retrospective study has revealed new potential risk factors for chemotherapy-induced febrile neutropenia (FN) in patients with solid tumors and non-Hodgkin lymphoma (NHL).
Researchers found the timing and duration of corticosteroid use were both associated with FN.
The team also observed “marginal” associations between FN and certain dermatologic and mucosal conditions as well as the use of intravenous (IV) antibiotics before chemotherapy.
On the other hand, there was no association between oral antibiotic use and FN or between radiation therapy (RT) and FN.
Chun Rebecca Chao, PhD, of Kaiser Permanente Southern California in Pasadena, and her colleagues reported these findings in JNCCN.
“Febrile neutropenia is life-threatening and often requires hospitalization,” Dr. Chao noted. “Furthermore, FN can lead to chemotherapy dose delay and dose reduction, which, in turn, negatively impacts antitumor efficacy. However, it can be prevented if high-risk individuals are identified and treated prophylactically.”
With this in mind, Dr. Chao and her colleagues set out to identify novel risk factors for FN by analyzing 15,971 patients who were treated with myelosuppressive chemotherapy at Kaiser Permanente Southern California between 2000 and 2009.
Patients had been diagnosed with NHL (n=1,617) or breast (n=6,323), lung (n=3,584), colorectal (n=3,062), ovarian (n=924), or gastric (n=461) cancers.
In all, 4.3% of patients developed FN during their first cycle of chemotherapy.
Corticosteroid use
The researchers found corticosteroid use was associated with an increased risk of FN in a propensity score-adjusted (PSA) model (adjusted for age, sex, socioeconomic factors, comorbidities, etc.). The hazard ratio (HR) was 1.53 (95% CI, 1.17-1.98; P<0.01) for patients who received corticosteroids.
A longer duration of corticosteroid use was associated with a greater risk of FN. The adjusted HR (compared to no corticosteroid use) was:
- 1.78 for corticosteroid treatment lasting less than 15 days (P<0.01)
- 1.84 for treatment lasting 15 to 29 days (P<0.01)
- 2.27 for treatment lasting 30 to 44 days (P<0.01)
- 2.86 for treatment lasting 45 to 90 days (P<0.01).
More recent corticosteroid use was associated with a greater risk of FN as well. The adjusted HR was:
- 1.88 for corticosteroid treatment less than 15 days before chemotherapy (P<0.01)
- 1.13 for treatment 15 to 29 days before chemotherapy (P=0.72)
- 1.22 for treatment 30 to 44 days before chemotherapy (P=0.66)
- 1.41 for treatment 45 to 90 days before chemotherapy (P=0.32).
“One way to reduce the incidence rate for FN could be to schedule prior corticosteroid use and subsequent chemotherapy with at least 2 weeks between them, given the magnitude of the risk increase and prevalence of this risk factor,” Dr. Chao said.
Other potential risk factors
The researchers found a “marginally” increased risk of FN in patients with certain dermatologic conditions (dermatitis, psoriasis, pruritus, etc.) and mucosal conditions (gastritis, stomatitis, mucositis, etc.).
In the PSA model, the HR was 1.40 (95% CI, 0.98-1.93; P=0.05) for patients with these conditions.
IV antibiotic use was also found to be marginally associated with an increased risk of FN in a restricted analysis covering patients treated in 2008 and 2009. In the PSA model, the HR was 1.35 (95% CI, 0.97-1.87; P=0.08).
On the other hand, there was no association between FN and oral antibiotic use in the restricted analysis. In the PSA model, the HR was 1.07 (95% CI, 0.77-1.48; P=0.70) for patients who received oral antibiotics.
Dr. Chao and her colleagues said these results suggest IV antibiotics may have a more profound impact than oral antibiotics on the balance of bacterial flora and other immune functions. Another possible explanation is that patients who received IV antibiotics were generally sicker and more prone to severe infection than patients who received oral antibiotics.
As with oral antibiotics, the researchers found no association between FN and the following factors (with the PSA model):
- Prior surgery (HR=0.89; 95% CI, 0.72-1.11; P=0.30)
- Prior RT (HR=0.91; 95% CI, 0.64-1.27; P=0.61)
- Concurrent RT (HR=1.32; 95% CI, 0.69-2.37; P=0.37).
The researchers noted that they did not account for radiation field or dose in this study, so additional evaluation of RT as a risk factor is needed.
In closing, Dr. Chao and her colleagues said these results suggest corticosteroid use, IV antibiotics, and certain dermatologic and mucosal conditions should be taken into consideration when monitoring patients receiving myelosuppressive chemotherapy and when evaluating the need for prophylactic granulocyte colony-stimulating factor or chemotherapy dose reduction.
Dr. Chao and her colleagues received funding from Amgen, Inc., to perform this study.
A retrospective study has revealed new potential risk factors for chemotherapy-induced febrile neutropenia (FN) in patients with solid tumors and non-Hodgkin lymphoma (NHL).
Researchers found the timing and duration of corticosteroid use were both associated with FN.
The team also observed “marginal” associations between FN and certain dermatologic and mucosal conditions as well as the use of intravenous (IV) antibiotics before chemotherapy.
On the other hand, there was no association between oral antibiotic use and FN or between radiation therapy (RT) and FN.
Chun Rebecca Chao, PhD, of Kaiser Permanente Southern California in Pasadena, and her colleagues reported these findings in JNCCN.
“Febrile neutropenia is life-threatening and often requires hospitalization,” Dr. Chao noted. “Furthermore, FN can lead to chemotherapy dose delay and dose reduction, which, in turn, negatively impacts antitumor efficacy. However, it can be prevented if high-risk individuals are identified and treated prophylactically.”
With this in mind, Dr. Chao and her colleagues set out to identify novel risk factors for FN by analyzing 15,971 patients who were treated with myelosuppressive chemotherapy at Kaiser Permanente Southern California between 2000 and 2009.
Patients had been diagnosed with NHL (n=1,617) or breast (n=6,323), lung (n=3,584), colorectal (n=3,062), ovarian (n=924), or gastric (n=461) cancers.
In all, 4.3% of patients developed FN during their first cycle of chemotherapy.
Corticosteroid use
The researchers found corticosteroid use was associated with an increased risk of FN in a propensity score-adjusted (PSA) model (adjusted for age, sex, socioeconomic factors, comorbidities, etc.). The hazard ratio (HR) was 1.53 (95% CI, 1.17-1.98; P<0.01) for patients who received corticosteroids.
A longer duration of corticosteroid use was associated with a greater risk of FN. The adjusted HR (compared to no corticosteroid use) was:
- 1.78 for corticosteroid treatment lasting less than 15 days (P<0.01)
- 1.84 for treatment lasting 15 to 29 days (P<0.01)
- 2.27 for treatment lasting 30 to 44 days (P<0.01)
- 2.86 for treatment lasting 45 to 90 days (P<0.01).
More recent corticosteroid use was associated with a greater risk of FN as well. The adjusted HR was:
- 1.88 for corticosteroid treatment less than 15 days before chemotherapy (P<0.01)
- 1.13 for treatment 15 to 29 days before chemotherapy (P=0.72)
- 1.22 for treatment 30 to 44 days before chemotherapy (P=0.66)
- 1.41 for treatment 45 to 90 days before chemotherapy (P=0.32).
“One way to reduce the incidence rate for FN could be to schedule prior corticosteroid use and subsequent chemotherapy with at least 2 weeks between them, given the magnitude of the risk increase and prevalence of this risk factor,” Dr. Chao said.
Other potential risk factors
The researchers found a “marginally” increased risk of FN in patients with certain dermatologic conditions (dermatitis, psoriasis, pruritus, etc.) and mucosal conditions (gastritis, stomatitis, mucositis, etc.).
In the PSA model, the HR was 1.40 (95% CI, 0.98-1.93; P=0.05) for patients with these conditions.
IV antibiotic use was also found to be marginally associated with an increased risk of FN in a restricted analysis covering patients treated in 2008 and 2009. In the PSA model, the HR was 1.35 (95% CI, 0.97-1.87; P=0.08).
On the other hand, there was no association between FN and oral antibiotic use in the restricted analysis. In the PSA model, the HR was 1.07 (95% CI, 0.77-1.48; P=0.70) for patients who received oral antibiotics.
Dr. Chao and her colleagues said these results suggest IV antibiotics may have a more profound impact than oral antibiotics on the balance of bacterial flora and other immune functions. Another possible explanation is that patients who received IV antibiotics were generally sicker and more prone to severe infection than patients who received oral antibiotics.
As with oral antibiotics, the researchers found no association between FN and the following factors (with the PSA model):
- Prior surgery (HR=0.89; 95% CI, 0.72-1.11; P=0.30)
- Prior RT (HR=0.91; 95% CI, 0.64-1.27; P=0.61)
- Concurrent RT (HR=1.32; 95% CI, 0.69-2.37; P=0.37).
The researchers noted that they did not account for radiation field or dose in this study, so additional evaluation of RT as a risk factor is needed.
In closing, Dr. Chao and her colleagues said these results suggest corticosteroid use, IV antibiotics, and certain dermatologic and mucosal conditions should be taken into consideration when monitoring patients receiving myelosuppressive chemotherapy and when evaluating the need for prophylactic granulocyte colony-stimulating factor or chemotherapy dose reduction.
Dr. Chao and her colleagues received funding from Amgen, Inc., to perform this study.
EVI1 overexpression promotes leukemogenesis, study suggests
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Palliative care guidelines relevant for hematologists, doc says
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
A closer look at an ezetimibe discussion
Although I look forward to receiving JFP each month, I was initially disappointed in Dr. Jonathon M. Firnhaber’s article, “Newer cholesterol-lowering agents: What you must know” (J Fam Pract. 2018;67:339-341,344,345), because of what appeared to be a superficial discussion of the medication ezetimibe. The potential role of PCSK9 inhibitors in extremely high-risk individuals was well discussed, but my first read left me with the impression that ezetimibe should be used more widely.
It seemed that in the section for ezetimibe, the author was suggesting using it for primary prevention. The line, “Consider adding ezetimibe to maximally tolerated statin therapy for patients not meeting LDL-C goals with a statin alone” left me a bit confused, as the most widely used guideline (that by the American College of Cardiology/American Heart Association Task Force on Practice Guidelines) states that there is no goal low-density lipoprotein cholesterol (LDL-C) level for primary prevention in patients without known cardiovascular disease (CVD) because studies have not been done to support this concept.1
But upon rereading the article, I realized the statement was placed at the end of a section that discussed secondary prevention based on the IMPROVE-IT study.2 This trial included only patients with previous acute coronary syndrome, one of the populations at highest risk.
I write just to reinforce the importance of considering what evidence we have for primary prevention. Although there is a value to rechecking LDL-C levels to assess compliance, there really is no convincing evidence that we should treat to a goal LDL-C level in someone who does not already have CVD. So the addition of ezetimibe to a statin in these patients is not recommended. Thus, the often-quoted strategy: “Start them on the right statin, and don’t look back.”
Bill Crump, MD
Madisonville, Ky
1. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(suppl 2):S1-S45.
2. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
Continue to: Authors' response
Author’s response:
Thank you, Dr. Crump, for your feedback. I suspect that most clinicians would welcome more robust outcomes data on ezetimibe, but to date none have been published.
The IMPROVE-IT trial1 offers the best supportive evidence for the use of ezetimibe, but still finds only a 2% absolute risk reduction (ARR) in a composite endpoint (cardiovascular death, nonfatal myocardial infarction, unstable angina requiring rehospitalization, coronary revascularization ≥30 days after randomization, or nonfatal stroke), equating to a number needed to treat (NNT) of 50.
The largest meta-analysis of ezetimibe trials—published prior to IMPROVE-IT—combined 31,048 patients to find an ARR for myocardial infarction of 1.1% (NNT=91) and an ARR for stroke of 0.6% (NNT=167), with no difference in cardiovascular death.2
Because of its limited outcomes data, ezetimibe is best reserved for patients unable to tolerate statin therapy, for those in whom statin therapy is contraindicated, or for those not meeting LDL-C reduction goals with a statin alone. This position is also supported by the United Kingdom’s National Institute for Health and Care Excellence (NICE).3
Finally, you are correct that the 2013 American College of Cardiology/American Heart Association Guideline on the Assessment of Cardiovascular Risk does not advocate a number-driven LDL-C goal, but rather recommends a risk-based moderate (30%-50%) or high-intensity (>50%) LDL-C reduction goal.4
Jonathon Firnhaber, MD
Greenville, NC
1. Cannon C, Blazing M, Giugliano R, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
2. Savarese G, Ferrari G, Rosano G, et al. Safety and efficacy of ezetimibe: a meta-analysis. Int J Cardiol. 2015;201:247-252.
3. National Institute for Health and Care Excellence. Ezetimibe for treating primary heterozygous-familial and non-familial hypercholesterolaemia. Technology appraisal guidance [TA385]. February 24, 2016. www.nice.org.uk/guidance/ta385. Accessed September 12, 2018.
4. Goff DC Jr, Lloyd-Jones DM, Bennett G, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2935-2959.
Although I look forward to receiving JFP each month, I was initially disappointed in Dr. Jonathon M. Firnhaber’s article, “Newer cholesterol-lowering agents: What you must know” (J Fam Pract. 2018;67:339-341,344,345), because of what appeared to be a superficial discussion of the medication ezetimibe. The potential role of PCSK9 inhibitors in extremely high-risk individuals was well discussed, but my first read left me with the impression that ezetimibe should be used more widely.
It seemed that in the section for ezetimibe, the author was suggesting using it for primary prevention. The line, “Consider adding ezetimibe to maximally tolerated statin therapy for patients not meeting LDL-C goals with a statin alone” left me a bit confused, as the most widely used guideline (that by the American College of Cardiology/American Heart Association Task Force on Practice Guidelines) states that there is no goal low-density lipoprotein cholesterol (LDL-C) level for primary prevention in patients without known cardiovascular disease (CVD) because studies have not been done to support this concept.1
But upon rereading the article, I realized the statement was placed at the end of a section that discussed secondary prevention based on the IMPROVE-IT study.2 This trial included only patients with previous acute coronary syndrome, one of the populations at highest risk.
I write just to reinforce the importance of considering what evidence we have for primary prevention. Although there is a value to rechecking LDL-C levels to assess compliance, there really is no convincing evidence that we should treat to a goal LDL-C level in someone who does not already have CVD. So the addition of ezetimibe to a statin in these patients is not recommended. Thus, the often-quoted strategy: “Start them on the right statin, and don’t look back.”
Bill Crump, MD
Madisonville, Ky
1. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(suppl 2):S1-S45.
2. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
Continue to: Authors' response
Author’s response:
Thank you, Dr. Crump, for your feedback. I suspect that most clinicians would welcome more robust outcomes data on ezetimibe, but to date none have been published.
The IMPROVE-IT trial1 offers the best supportive evidence for the use of ezetimibe, but still finds only a 2% absolute risk reduction (ARR) in a composite endpoint (cardiovascular death, nonfatal myocardial infarction, unstable angina requiring rehospitalization, coronary revascularization ≥30 days after randomization, or nonfatal stroke), equating to a number needed to treat (NNT) of 50.
The largest meta-analysis of ezetimibe trials—published prior to IMPROVE-IT—combined 31,048 patients to find an ARR for myocardial infarction of 1.1% (NNT=91) and an ARR for stroke of 0.6% (NNT=167), with no difference in cardiovascular death.2
Because of its limited outcomes data, ezetimibe is best reserved for patients unable to tolerate statin therapy, for those in whom statin therapy is contraindicated, or for those not meeting LDL-C reduction goals with a statin alone. This position is also supported by the United Kingdom’s National Institute for Health and Care Excellence (NICE).3
Finally, you are correct that the 2013 American College of Cardiology/American Heart Association Guideline on the Assessment of Cardiovascular Risk does not advocate a number-driven LDL-C goal, but rather recommends a risk-based moderate (30%-50%) or high-intensity (>50%) LDL-C reduction goal.4
Jonathon Firnhaber, MD
Greenville, NC
1. Cannon C, Blazing M, Giugliano R, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
2. Savarese G, Ferrari G, Rosano G, et al. Safety and efficacy of ezetimibe: a meta-analysis. Int J Cardiol. 2015;201:247-252.
3. National Institute for Health and Care Excellence. Ezetimibe for treating primary heterozygous-familial and non-familial hypercholesterolaemia. Technology appraisal guidance [TA385]. February 24, 2016. www.nice.org.uk/guidance/ta385. Accessed September 12, 2018.
4. Goff DC Jr, Lloyd-Jones DM, Bennett G, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2935-2959.
Although I look forward to receiving JFP each month, I was initially disappointed in Dr. Jonathon M. Firnhaber’s article, “Newer cholesterol-lowering agents: What you must know” (J Fam Pract. 2018;67:339-341,344,345), because of what appeared to be a superficial discussion of the medication ezetimibe. The potential role of PCSK9 inhibitors in extremely high-risk individuals was well discussed, but my first read left me with the impression that ezetimibe should be used more widely.
It seemed that in the section for ezetimibe, the author was suggesting using it for primary prevention. The line, “Consider adding ezetimibe to maximally tolerated statin therapy for patients not meeting LDL-C goals with a statin alone” left me a bit confused, as the most widely used guideline (that by the American College of Cardiology/American Heart Association Task Force on Practice Guidelines) states that there is no goal low-density lipoprotein cholesterol (LDL-C) level for primary prevention in patients without known cardiovascular disease (CVD) because studies have not been done to support this concept.1
But upon rereading the article, I realized the statement was placed at the end of a section that discussed secondary prevention based on the IMPROVE-IT study.2 This trial included only patients with previous acute coronary syndrome, one of the populations at highest risk.
I write just to reinforce the importance of considering what evidence we have for primary prevention. Although there is a value to rechecking LDL-C levels to assess compliance, there really is no convincing evidence that we should treat to a goal LDL-C level in someone who does not already have CVD. So the addition of ezetimibe to a statin in these patients is not recommended. Thus, the often-quoted strategy: “Start them on the right statin, and don’t look back.”
Bill Crump, MD
Madisonville, Ky
1. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(suppl 2):S1-S45.
2. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
Continue to: Authors' response
Author’s response:
Thank you, Dr. Crump, for your feedback. I suspect that most clinicians would welcome more robust outcomes data on ezetimibe, but to date none have been published.
The IMPROVE-IT trial1 offers the best supportive evidence for the use of ezetimibe, but still finds only a 2% absolute risk reduction (ARR) in a composite endpoint (cardiovascular death, nonfatal myocardial infarction, unstable angina requiring rehospitalization, coronary revascularization ≥30 days after randomization, or nonfatal stroke), equating to a number needed to treat (NNT) of 50.
The largest meta-analysis of ezetimibe trials—published prior to IMPROVE-IT—combined 31,048 patients to find an ARR for myocardial infarction of 1.1% (NNT=91) and an ARR for stroke of 0.6% (NNT=167), with no difference in cardiovascular death.2
Because of its limited outcomes data, ezetimibe is best reserved for patients unable to tolerate statin therapy, for those in whom statin therapy is contraindicated, or for those not meeting LDL-C reduction goals with a statin alone. This position is also supported by the United Kingdom’s National Institute for Health and Care Excellence (NICE).3
Finally, you are correct that the 2013 American College of Cardiology/American Heart Association Guideline on the Assessment of Cardiovascular Risk does not advocate a number-driven LDL-C goal, but rather recommends a risk-based moderate (30%-50%) or high-intensity (>50%) LDL-C reduction goal.4
Jonathon Firnhaber, MD
Greenville, NC
1. Cannon C, Blazing M, Giugliano R, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
2. Savarese G, Ferrari G, Rosano G, et al. Safety and efficacy of ezetimibe: a meta-analysis. Int J Cardiol. 2015;201:247-252.
3. National Institute for Health and Care Excellence. Ezetimibe for treating primary heterozygous-familial and non-familial hypercholesterolaemia. Technology appraisal guidance [TA385]. February 24, 2016. www.nice.org.uk/guidance/ta385. Accessed September 12, 2018.
4. Goff DC Jr, Lloyd-Jones DM, Bennett G, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2935-2959.
Disagreement over a Case Report Dx
Based on the magnetic resonance imaging (MRI) scans presented in the Case Report, “Bilateral wrist pain • limited range of motion • tenderness to palpation • Dx?” (J Fam Pract. 2018;67:160-162), I disagree with the diagnosis.
Contrary to the assertion by Drs. Shehata and Hizon that the patient had “fractures extending through the scaphoid waist,” this young girl actually had bilateral osseous contusions (ie, microtrabecular fractures) of the radial aspect of the scaphoid and did not have complete scaphoid waist fractures. Also, the MRI scans demonstrate intact ulnar cortices bilaterally, indicating that there is no complete scaphoid waist fracture.
These are typical “FOOSH” (fall on outstretched hand) injuries and would be expected to have an exceedingly good prognosis with immobilization. As to whether or not this affects medical management, such as how long the cast remains on the arm, I would have to defer to an orthopedic surgeon’s judgment.
David R. Pennes, MD
Grand Rapids, Mich
Continue to: Author's response
Author’s response:
Thank you for your comments. You are correct that the MRI scans shown do not demonstrate a complete fracture through the scaphoid, but rather a microtrabecular fracture. We did not intend to make the distinction between the 2 entities because management for both is similar. The teaching point of this case was to impress upon clinicians that these types of fractures may be subtle even on MRI, and that if they are not treated appropriately, they can progress to complete fracture or result in non-union and long-term pain and disability.
Jerry Hizon, MD
Riverside, Calif
Based on the magnetic resonance imaging (MRI) scans presented in the Case Report, “Bilateral wrist pain • limited range of motion • tenderness to palpation • Dx?” (J Fam Pract. 2018;67:160-162), I disagree with the diagnosis.
Contrary to the assertion by Drs. Shehata and Hizon that the patient had “fractures extending through the scaphoid waist,” this young girl actually had bilateral osseous contusions (ie, microtrabecular fractures) of the radial aspect of the scaphoid and did not have complete scaphoid waist fractures. Also, the MRI scans demonstrate intact ulnar cortices bilaterally, indicating that there is no complete scaphoid waist fracture.
These are typical “FOOSH” (fall on outstretched hand) injuries and would be expected to have an exceedingly good prognosis with immobilization. As to whether or not this affects medical management, such as how long the cast remains on the arm, I would have to defer to an orthopedic surgeon’s judgment.
David R. Pennes, MD
Grand Rapids, Mich
Continue to: Author's response
Author’s response:
Thank you for your comments. You are correct that the MRI scans shown do not demonstrate a complete fracture through the scaphoid, but rather a microtrabecular fracture. We did not intend to make the distinction between the 2 entities because management for both is similar. The teaching point of this case was to impress upon clinicians that these types of fractures may be subtle even on MRI, and that if they are not treated appropriately, they can progress to complete fracture or result in non-union and long-term pain and disability.
Jerry Hizon, MD
Riverside, Calif
Based on the magnetic resonance imaging (MRI) scans presented in the Case Report, “Bilateral wrist pain • limited range of motion • tenderness to palpation • Dx?” (J Fam Pract. 2018;67:160-162), I disagree with the diagnosis.
Contrary to the assertion by Drs. Shehata and Hizon that the patient had “fractures extending through the scaphoid waist,” this young girl actually had bilateral osseous contusions (ie, microtrabecular fractures) of the radial aspect of the scaphoid and did not have complete scaphoid waist fractures. Also, the MRI scans demonstrate intact ulnar cortices bilaterally, indicating that there is no complete scaphoid waist fracture.
These are typical “FOOSH” (fall on outstretched hand) injuries and would be expected to have an exceedingly good prognosis with immobilization. As to whether or not this affects medical management, such as how long the cast remains on the arm, I would have to defer to an orthopedic surgeon’s judgment.
David R. Pennes, MD
Grand Rapids, Mich
Continue to: Author's response
Author’s response:
Thank you for your comments. You are correct that the MRI scans shown do not demonstrate a complete fracture through the scaphoid, but rather a microtrabecular fracture. We did not intend to make the distinction between the 2 entities because management for both is similar. The teaching point of this case was to impress upon clinicians that these types of fractures may be subtle even on MRI, and that if they are not treated appropriately, they can progress to complete fracture or result in non-union and long-term pain and disability.
Jerry Hizon, MD
Riverside, Calif
New feature debuts, how to address reviewer criticism, and more!
Dear Colleagues,
The November issue of The New Gastroenterologist is packed with some great articles! First, this issue’s In Focus article addresses the increasingly important topic of endoscopic management of obesity. In the article, the authors, Pichamol Jirapinyo and Christopher Thompson (Brigham and Women’s Hospital), provide an outstanding overview of the approved and up-and-coming endoscopic therapies that can be used to help treat the obesity epidemic. This is an area that we will inevitably see more of in our practices.
A new feature in this issue of The New Gastroenterologist is a column focused on early career gastroenterologists who are going into private practice, which was curated in conjunction with the Digestive Health Physicians Association. This month’s article by Fred Rosenberg (North Shore Endoscopy Center) provides an overview of private practice gastroenterology models. I look forward to making this column a recurring feature of future issues.
Additionally, using their wealth of experience, former CGH editor in chief Hashem El-Serag and current CGH editor in chief Fasiha Kanwal (Baylor) provide an enlightening piece on how to address reviewer criticism, which will no doubt be very helpful for those of us looking to publish. There is also a helpful article about grant writing tips authored by two successfully funded early career basic scientists, Arthur Beyder (Mayo) and Christina Twyman-Saint Victor (University of Pennsylvania).
For those considering pursuing extra training in IBD either during or after GI fellowship, Siddharth Singh (UCSD) goes through the different advanced training options that are now available in IBD. And finally, as many are laying down roots in new places, buying a house will almost inevitably be on the horizon. To help guide you through the mortgage preapproval process, Rob Wishnick (Guaranteed Rate) provides some useful insights from his many years of experience in the home loan industry.
Please check out “In Case You Missed It” to see other articles from the last quarter in AGA publications that may be of interest to you. And, if you have any ideas or want to contribute to The New Gastroenterologist, please contact me at bryson.katona@uphs.upenn.edu or the managing editor, Ryan Farrell, at rfarrell@gastro.org.
Sincerely,
Bryson W. Katona, MD, PhD
Editor in Chief
Dr. Katona is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia.
Dear Colleagues,
The November issue of The New Gastroenterologist is packed with some great articles! First, this issue’s In Focus article addresses the increasingly important topic of endoscopic management of obesity. In the article, the authors, Pichamol Jirapinyo and Christopher Thompson (Brigham and Women’s Hospital), provide an outstanding overview of the approved and up-and-coming endoscopic therapies that can be used to help treat the obesity epidemic. This is an area that we will inevitably see more of in our practices.
A new feature in this issue of The New Gastroenterologist is a column focused on early career gastroenterologists who are going into private practice, which was curated in conjunction with the Digestive Health Physicians Association. This month’s article by Fred Rosenberg (North Shore Endoscopy Center) provides an overview of private practice gastroenterology models. I look forward to making this column a recurring feature of future issues.
Additionally, using their wealth of experience, former CGH editor in chief Hashem El-Serag and current CGH editor in chief Fasiha Kanwal (Baylor) provide an enlightening piece on how to address reviewer criticism, which will no doubt be very helpful for those of us looking to publish. There is also a helpful article about grant writing tips authored by two successfully funded early career basic scientists, Arthur Beyder (Mayo) and Christina Twyman-Saint Victor (University of Pennsylvania).
For those considering pursuing extra training in IBD either during or after GI fellowship, Siddharth Singh (UCSD) goes through the different advanced training options that are now available in IBD. And finally, as many are laying down roots in new places, buying a house will almost inevitably be on the horizon. To help guide you through the mortgage preapproval process, Rob Wishnick (Guaranteed Rate) provides some useful insights from his many years of experience in the home loan industry.
Please check out “In Case You Missed It” to see other articles from the last quarter in AGA publications that may be of interest to you. And, if you have any ideas or want to contribute to The New Gastroenterologist, please contact me at bryson.katona@uphs.upenn.edu or the managing editor, Ryan Farrell, at rfarrell@gastro.org.
Sincerely,
Bryson W. Katona, MD, PhD
Editor in Chief
Dr. Katona is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia.
Dear Colleagues,
The November issue of The New Gastroenterologist is packed with some great articles! First, this issue’s In Focus article addresses the increasingly important topic of endoscopic management of obesity. In the article, the authors, Pichamol Jirapinyo and Christopher Thompson (Brigham and Women’s Hospital), provide an outstanding overview of the approved and up-and-coming endoscopic therapies that can be used to help treat the obesity epidemic. This is an area that we will inevitably see more of in our practices.
A new feature in this issue of The New Gastroenterologist is a column focused on early career gastroenterologists who are going into private practice, which was curated in conjunction with the Digestive Health Physicians Association. This month’s article by Fred Rosenberg (North Shore Endoscopy Center) provides an overview of private practice gastroenterology models. I look forward to making this column a recurring feature of future issues.
Additionally, using their wealth of experience, former CGH editor in chief Hashem El-Serag and current CGH editor in chief Fasiha Kanwal (Baylor) provide an enlightening piece on how to address reviewer criticism, which will no doubt be very helpful for those of us looking to publish. There is also a helpful article about grant writing tips authored by two successfully funded early career basic scientists, Arthur Beyder (Mayo) and Christina Twyman-Saint Victor (University of Pennsylvania).
For those considering pursuing extra training in IBD either during or after GI fellowship, Siddharth Singh (UCSD) goes through the different advanced training options that are now available in IBD. And finally, as many are laying down roots in new places, buying a house will almost inevitably be on the horizon. To help guide you through the mortgage preapproval process, Rob Wishnick (Guaranteed Rate) provides some useful insights from his many years of experience in the home loan industry.
Please check out “In Case You Missed It” to see other articles from the last quarter in AGA publications that may be of interest to you. And, if you have any ideas or want to contribute to The New Gastroenterologist, please contact me at bryson.katona@uphs.upenn.edu or the managing editor, Ryan Farrell, at rfarrell@gastro.org.
Sincerely,
Bryson W. Katona, MD, PhD
Editor in Chief
Dr. Katona is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia.
Persistent erythematous papulonodular rash
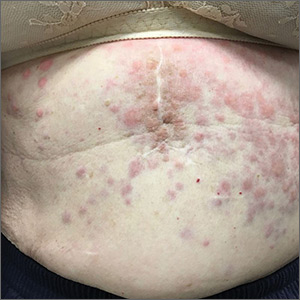
An 80-year-old white woman presented to our dermatology clinic with a rash across her abdomen that had been there for more than a year. While not itchy or painful, the rash was slowly expanding. The patient had tried treatments including topical antifungals and topical corticosteroids, but none had helped.
Her medical history was significant for dementia and stage III triple-negative breast cancer in the left breast (diagnosed 8 years prior), which was treated with a simple left mastectomy, chemotherapy, and radiation. She reported no history of skin cancer. She was not taking any medications and had no known drug allergies. A physical examination revealed an erythematous, papulonodular rash with diffuse induration in a band-like pattern across her entire upper abdomen and left flank (FIGURE).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cutaneous metastasis of primary breast cancer
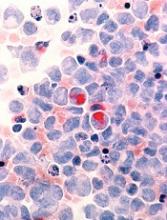
Based on our patient’s history, we gave a presumptive diagnosis of cutaneous breast cancer metastasis. A punch biopsy was performed. The pathology report showed nests of neoplastic cells within the dermis, which was consistent with this diagnosis. Immunohistochemical stains and fluorescence in-situ hybridization confirmed triple-negative breast markers for estrogen receptor, progesterone receptor, and human epidermal growth factor receptor-2.
An uncommon phenomenonseen mostly with breast cancer
Cutaneous metastatic carcinoma is relatively uncommon; one meta-analysis reported the overall incidence to be 5.3%.1 While it is unusual, any internal malignancy can metastasize to the skin. In women, the most common malignancy to do so is breast cancer. One study found breast cancer to be associated with 26.5% of cutaneous metastatic cases.2 These metastases often occur well after the patient has been treated for the primary malignancy.
Identifying features. Most cutaneous metastases occur near the site of the primary tumor, initially in the form of a firm, mobile, nonpainful nodule.3 This nodule is typically skin-colored or red, but in the case of cutaneous metastases of melanomas, it can appear blue or black. In the case of breast cancer, the lesions most often arise on the chest and abdomen.4 Occasionally, metastases can ulcerate through the skin.
Some forms of cutaneous metastasis, such as carcinoma erysipeloides, can appear in specific patterns. Carcinoma erysipeloides has a similar appearance to cellulitis; it manifests as a sharply demarcated, red, inflammatory patch in the skin adjacent to the primary tumor.
Consider the clinical picture
Cutaneous metastatic lesions have a wide range of differential diagnoses due to their varied appearances. It is important to view the overall clinical picture when distinguishing such lesions. Although cutaneous metastasis is uncommon, it should always be considered when asymptomatic skin lesions resist treatment—even in someone without a known history of malignancy.
Perform a biopsy. The diagnosis can be confirmed with a skin biopsy. A punch biopsy is preferable, as visualization of the dermis is crucial, and histology often reveals nests of pleomorphic cells. Further cellular cytology can elicit the primary malignancy of origin.
Making our diagnosis
We ruled out several possibilities before arriving at our diagnosis. An infectious etiology (eg, cutaneous candidiasis) was considered, as was a cutaneous change due to radiation therapy. We also considered shingles, the early stages of which would have been similar in appearance to our patient’s lesions, and urticaria, which can manifest as erythematous papules and wheals across various parts of the body. A lack of specific symptoms (eg, pruritis, pain, fever) made these alternative diagnoses less likely. The fact that our patient’s lesions persisted for more than a year without any response to treatment—and that they continued to grow—alerted us of a more sinister etiology.
Continue to: Treating the tumor is often not possible
Treating the tumor is often not possible
Treatment first involves treating the underlying tumor. For cases in which cutaneous lesions are the first manifestation of an internal malignancy, investigation as to the source should be performed. The lesions can then be treated with a combination of chemotherapy, radiation, and surgery.5,6
Unfortunately, in most cases of cutaneous metastases, the primary malignancy is already widespread and possibly untreatable. In such instances, palliative care is offered. Lesions are managed symptomatically, and prevention of skin irritation becomes the primary focus. Keeping the skin clean and dry helps to prevent ulceration and secondary infection.
In cases where the lesions ulcerate or crust, debridement can help. Excision of lesions, as well as pairing laser therapy with electrochemotherapy, may be helpful to improve the patient’s quality of life when lesions cause discomfort.
The prognosis for cutaneous metastasis due to breast cancer is often hard to predict because it is determined by other factors, such as the presence of internal metastases, which indicates a worse prognosis (on the scale of months). Some case reports have demonstrated that patients with metastases limited to the skin may have prolonged survival (on the scale of years).7
Our patient was offered an initial trial of radiation therapy, but she refused all treatment because the lesions did not cause discomfort, and she preferred to not go through further aggressive cancer treatment that could potentially cause complications and pain. We respected the patient’s wishes and counseled her on follow-up if the lesions became symptomatic or she decided she wanted to try treatment.
CORRESPONDENCE
Araya Zaesim, 1550 College St, Macon, GA, 31207; Zaesim.araya@gmail.com
1. Krathen RA, Orengo IF, Rosen T. Cutaneous metastasis: a meta-analysis of data. South Med J. 2003;96:164-167.
2. Brownstein MH, Helwig EB. Patterns of cutaneous metastasis. Arch Dermatol. 1972;105:862-868.
3. De Giorgi V, Grazzini M, Alfaioli B, et al. Cutaneous manifestations of breast carcinoma. Dermatol Ther. 2010;23:581-589.
4. Wong CYB, Helm MA, Kalb RE, et al. The presentation, pathology, and current management strategies of cutaneous metastasis. N Am J Med Sci. 2013;5:499-504.
5. Moore S. Cutaneous metastatic breast cancer. Clin J Oncol Nurs. 2002;6:255-260.
6. Ahmed M. Cutaneous metastases from breast carcinoma. BMJ Case Rep. 2011;2011: bcr0620114398.
7. Cho J, Park Y, Lee JC, et al. Case series of different onset of skin metastasis according to the breast cancer subtypes. Cancer Res Treat. 2014;46:194-199.
An 80-year-old white woman presented to our dermatology clinic with a rash across her abdomen that had been there for more than a year. While not itchy or painful, the rash was slowly expanding. The patient had tried treatments including topical antifungals and topical corticosteroids, but none had helped.
Her medical history was significant for dementia and stage III triple-negative breast cancer in the left breast (diagnosed 8 years prior), which was treated with a simple left mastectomy, chemotherapy, and radiation. She reported no history of skin cancer. She was not taking any medications and had no known drug allergies. A physical examination revealed an erythematous, papulonodular rash with diffuse induration in a band-like pattern across her entire upper abdomen and left flank (FIGURE).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cutaneous metastasis of primary breast cancer
Based on our patient’s history, we gave a presumptive diagnosis of cutaneous breast cancer metastasis. A punch biopsy was performed. The pathology report showed nests of neoplastic cells within the dermis, which was consistent with this diagnosis. Immunohistochemical stains and fluorescence in-situ hybridization confirmed triple-negative breast markers for estrogen receptor, progesterone receptor, and human epidermal growth factor receptor-2.
An uncommon phenomenonseen mostly with breast cancer
Cutaneous metastatic carcinoma is relatively uncommon; one meta-analysis reported the overall incidence to be 5.3%.1 While it is unusual, any internal malignancy can metastasize to the skin. In women, the most common malignancy to do so is breast cancer. One study found breast cancer to be associated with 26.5% of cutaneous metastatic cases.2 These metastases often occur well after the patient has been treated for the primary malignancy.
Identifying features. Most cutaneous metastases occur near the site of the primary tumor, initially in the form of a firm, mobile, nonpainful nodule.3 This nodule is typically skin-colored or red, but in the case of cutaneous metastases of melanomas, it can appear blue or black. In the case of breast cancer, the lesions most often arise on the chest and abdomen.4 Occasionally, metastases can ulcerate through the skin.
Some forms of cutaneous metastasis, such as carcinoma erysipeloides, can appear in specific patterns. Carcinoma erysipeloides has a similar appearance to cellulitis; it manifests as a sharply demarcated, red, inflammatory patch in the skin adjacent to the primary tumor.
Consider the clinical picture
Cutaneous metastatic lesions have a wide range of differential diagnoses due to their varied appearances. It is important to view the overall clinical picture when distinguishing such lesions. Although cutaneous metastasis is uncommon, it should always be considered when asymptomatic skin lesions resist treatment—even in someone without a known history of malignancy.
Perform a biopsy. The diagnosis can be confirmed with a skin biopsy. A punch biopsy is preferable, as visualization of the dermis is crucial, and histology often reveals nests of pleomorphic cells. Further cellular cytology can elicit the primary malignancy of origin.
Making our diagnosis
We ruled out several possibilities before arriving at our diagnosis. An infectious etiology (eg, cutaneous candidiasis) was considered, as was a cutaneous change due to radiation therapy. We also considered shingles, the early stages of which would have been similar in appearance to our patient’s lesions, and urticaria, which can manifest as erythematous papules and wheals across various parts of the body. A lack of specific symptoms (eg, pruritis, pain, fever) made these alternative diagnoses less likely. The fact that our patient’s lesions persisted for more than a year without any response to treatment—and that they continued to grow—alerted us of a more sinister etiology.
Continue to: Treating the tumor is often not possible
Treating the tumor is often not possible
Treatment first involves treating the underlying tumor. For cases in which cutaneous lesions are the first manifestation of an internal malignancy, investigation as to the source should be performed. The lesions can then be treated with a combination of chemotherapy, radiation, and surgery.5,6
Unfortunately, in most cases of cutaneous metastases, the primary malignancy is already widespread and possibly untreatable. In such instances, palliative care is offered. Lesions are managed symptomatically, and prevention of skin irritation becomes the primary focus. Keeping the skin clean and dry helps to prevent ulceration and secondary infection.
In cases where the lesions ulcerate or crust, debridement can help. Excision of lesions, as well as pairing laser therapy with electrochemotherapy, may be helpful to improve the patient’s quality of life when lesions cause discomfort.
The prognosis for cutaneous metastasis due to breast cancer is often hard to predict because it is determined by other factors, such as the presence of internal metastases, which indicates a worse prognosis (on the scale of months). Some case reports have demonstrated that patients with metastases limited to the skin may have prolonged survival (on the scale of years).7
Our patient was offered an initial trial of radiation therapy, but she refused all treatment because the lesions did not cause discomfort, and she preferred to not go through further aggressive cancer treatment that could potentially cause complications and pain. We respected the patient’s wishes and counseled her on follow-up if the lesions became symptomatic or she decided she wanted to try treatment.
CORRESPONDENCE
Araya Zaesim, 1550 College St, Macon, GA, 31207; Zaesim.araya@gmail.com
An 80-year-old white woman presented to our dermatology clinic with a rash across her abdomen that had been there for more than a year. While not itchy or painful, the rash was slowly expanding. The patient had tried treatments including topical antifungals and topical corticosteroids, but none had helped.
Her medical history was significant for dementia and stage III triple-negative breast cancer in the left breast (diagnosed 8 years prior), which was treated with a simple left mastectomy, chemotherapy, and radiation. She reported no history of skin cancer. She was not taking any medications and had no known drug allergies. A physical examination revealed an erythematous, papulonodular rash with diffuse induration in a band-like pattern across her entire upper abdomen and left flank (FIGURE).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cutaneous metastasis of primary breast cancer
Based on our patient’s history, we gave a presumptive diagnosis of cutaneous breast cancer metastasis. A punch biopsy was performed. The pathology report showed nests of neoplastic cells within the dermis, which was consistent with this diagnosis. Immunohistochemical stains and fluorescence in-situ hybridization confirmed triple-negative breast markers for estrogen receptor, progesterone receptor, and human epidermal growth factor receptor-2.
An uncommon phenomenonseen mostly with breast cancer
Cutaneous metastatic carcinoma is relatively uncommon; one meta-analysis reported the overall incidence to be 5.3%.1 While it is unusual, any internal malignancy can metastasize to the skin. In women, the most common malignancy to do so is breast cancer. One study found breast cancer to be associated with 26.5% of cutaneous metastatic cases.2 These metastases often occur well after the patient has been treated for the primary malignancy.
Identifying features. Most cutaneous metastases occur near the site of the primary tumor, initially in the form of a firm, mobile, nonpainful nodule.3 This nodule is typically skin-colored or red, but in the case of cutaneous metastases of melanomas, it can appear blue or black. In the case of breast cancer, the lesions most often arise on the chest and abdomen.4 Occasionally, metastases can ulcerate through the skin.
Some forms of cutaneous metastasis, such as carcinoma erysipeloides, can appear in specific patterns. Carcinoma erysipeloides has a similar appearance to cellulitis; it manifests as a sharply demarcated, red, inflammatory patch in the skin adjacent to the primary tumor.
Consider the clinical picture
Cutaneous metastatic lesions have a wide range of differential diagnoses due to their varied appearances. It is important to view the overall clinical picture when distinguishing such lesions. Although cutaneous metastasis is uncommon, it should always be considered when asymptomatic skin lesions resist treatment—even in someone without a known history of malignancy.
Perform a biopsy. The diagnosis can be confirmed with a skin biopsy. A punch biopsy is preferable, as visualization of the dermis is crucial, and histology often reveals nests of pleomorphic cells. Further cellular cytology can elicit the primary malignancy of origin.
Making our diagnosis
We ruled out several possibilities before arriving at our diagnosis. An infectious etiology (eg, cutaneous candidiasis) was considered, as was a cutaneous change due to radiation therapy. We also considered shingles, the early stages of which would have been similar in appearance to our patient’s lesions, and urticaria, which can manifest as erythematous papules and wheals across various parts of the body. A lack of specific symptoms (eg, pruritis, pain, fever) made these alternative diagnoses less likely. The fact that our patient’s lesions persisted for more than a year without any response to treatment—and that they continued to grow—alerted us of a more sinister etiology.
Continue to: Treating the tumor is often not possible
Treating the tumor is often not possible
Treatment first involves treating the underlying tumor. For cases in which cutaneous lesions are the first manifestation of an internal malignancy, investigation as to the source should be performed. The lesions can then be treated with a combination of chemotherapy, radiation, and surgery.5,6
Unfortunately, in most cases of cutaneous metastases, the primary malignancy is already widespread and possibly untreatable. In such instances, palliative care is offered. Lesions are managed symptomatically, and prevention of skin irritation becomes the primary focus. Keeping the skin clean and dry helps to prevent ulceration and secondary infection.
In cases where the lesions ulcerate or crust, debridement can help. Excision of lesions, as well as pairing laser therapy with electrochemotherapy, may be helpful to improve the patient’s quality of life when lesions cause discomfort.
The prognosis for cutaneous metastasis due to breast cancer is often hard to predict because it is determined by other factors, such as the presence of internal metastases, which indicates a worse prognosis (on the scale of months). Some case reports have demonstrated that patients with metastases limited to the skin may have prolonged survival (on the scale of years).7
Our patient was offered an initial trial of radiation therapy, but she refused all treatment because the lesions did not cause discomfort, and she preferred to not go through further aggressive cancer treatment that could potentially cause complications and pain. We respected the patient’s wishes and counseled her on follow-up if the lesions became symptomatic or she decided she wanted to try treatment.
CORRESPONDENCE
Araya Zaesim, 1550 College St, Macon, GA, 31207; Zaesim.araya@gmail.com
1. Krathen RA, Orengo IF, Rosen T. Cutaneous metastasis: a meta-analysis of data. South Med J. 2003;96:164-167.
2. Brownstein MH, Helwig EB. Patterns of cutaneous metastasis. Arch Dermatol. 1972;105:862-868.
3. De Giorgi V, Grazzini M, Alfaioli B, et al. Cutaneous manifestations of breast carcinoma. Dermatol Ther. 2010;23:581-589.
4. Wong CYB, Helm MA, Kalb RE, et al. The presentation, pathology, and current management strategies of cutaneous metastasis. N Am J Med Sci. 2013;5:499-504.
5. Moore S. Cutaneous metastatic breast cancer. Clin J Oncol Nurs. 2002;6:255-260.
6. Ahmed M. Cutaneous metastases from breast carcinoma. BMJ Case Rep. 2011;2011: bcr0620114398.
7. Cho J, Park Y, Lee JC, et al. Case series of different onset of skin metastasis according to the breast cancer subtypes. Cancer Res Treat. 2014;46:194-199.
1. Krathen RA, Orengo IF, Rosen T. Cutaneous metastasis: a meta-analysis of data. South Med J. 2003;96:164-167.
2. Brownstein MH, Helwig EB. Patterns of cutaneous metastasis. Arch Dermatol. 1972;105:862-868.
3. De Giorgi V, Grazzini M, Alfaioli B, et al. Cutaneous manifestations of breast carcinoma. Dermatol Ther. 2010;23:581-589.
4. Wong CYB, Helm MA, Kalb RE, et al. The presentation, pathology, and current management strategies of cutaneous metastasis. N Am J Med Sci. 2013;5:499-504.
5. Moore S. Cutaneous metastatic breast cancer. Clin J Oncol Nurs. 2002;6:255-260.
6. Ahmed M. Cutaneous metastases from breast carcinoma. BMJ Case Rep. 2011;2011: bcr0620114398.
7. Cho J, Park Y, Lee JC, et al. Case series of different onset of skin metastasis according to the breast cancer subtypes. Cancer Res Treat. 2014;46:194-199.
Primary hyperparathyroidism: Labs to order, Tx to consider
Since the advent of multichannel serum chemistry screening in the 1970s, large numbers of asymptomatic cases of primary hyperparathyroidism (PHPT) have been discovered. The clinical spectrum of the disease has changed from the classic “moans, groans, bones, and stones” to an asymptomatic and subtle presentation of hypercalcemia.1,2 PHPT and malignancy are the most common causes for hypercalcemia, accounting for 90% of cases.3 In the United States, the estimated incidence of PHPT between 1998 and 2010 was about 50 per 100,000 person-years. Most patients with PHPT are older women (ages >50 years) who are asymptomatic at the time of diagnosis.1
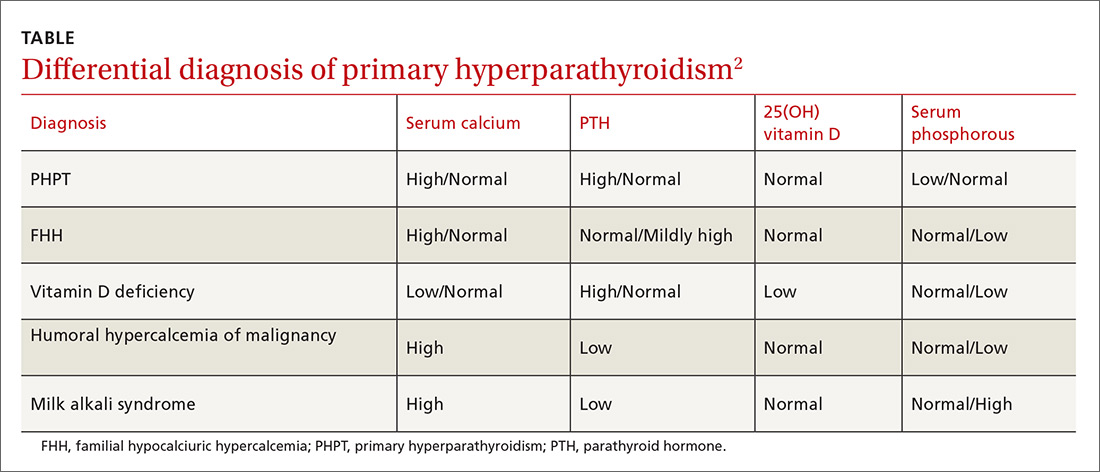
Vigilance needed in primary care. PHPT is slowly progressive, and the patient might accept symptoms as a process of aging. Therefore, it is essential that primary care physicians (PCPs) be aware of the diagnostic and management options. A systematic approach to the diagnosis of PHPT helps differentiate the causes of hypercalcemia (TABLE2; FIGURE2). But before we discuss PHPT diagnostic clues, it’s helpful to quickly review the workings of the parathyroid glands.
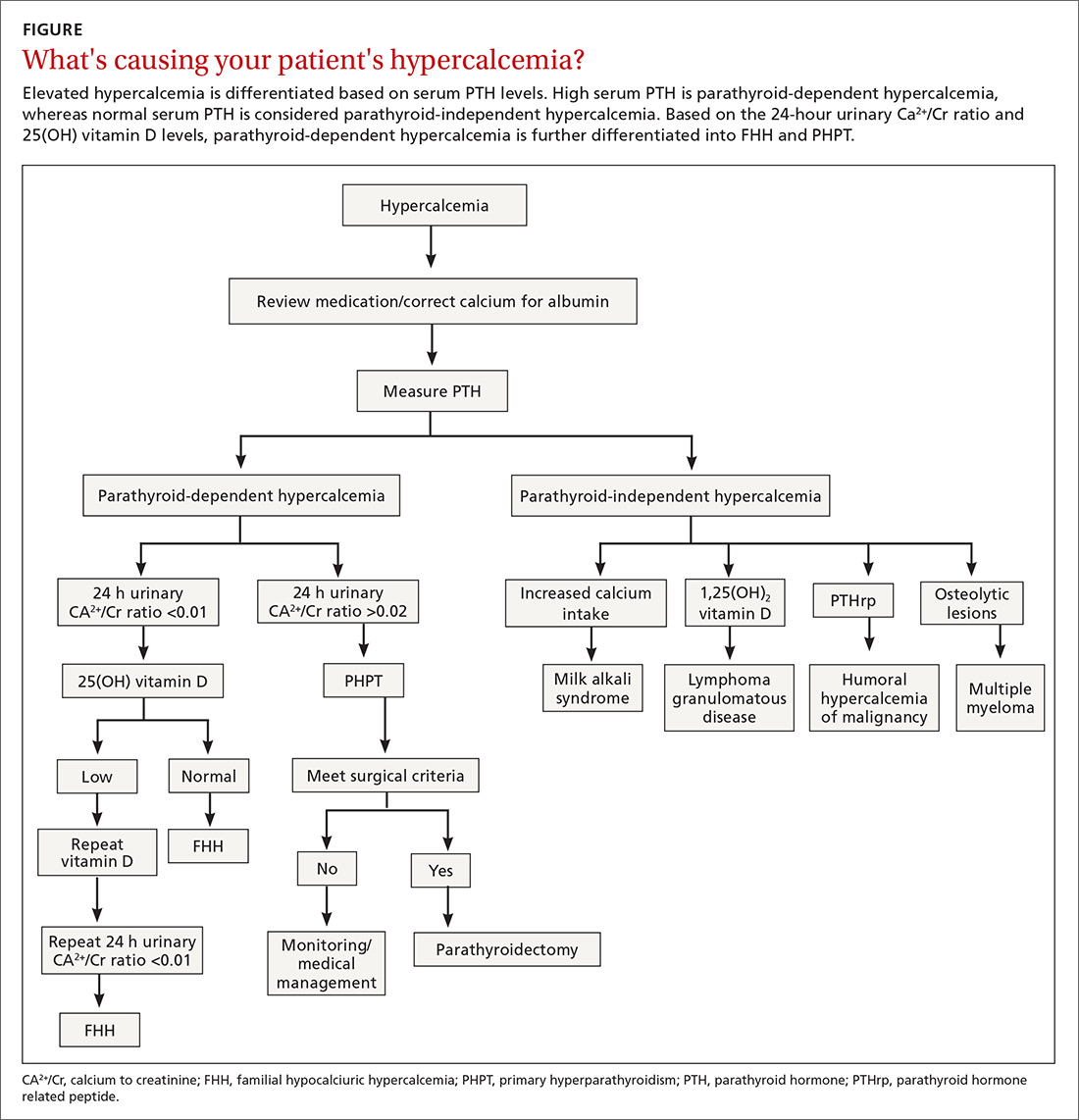
How the glands work, what can go wrong
Parathyroid hormone (PTH) is secreted by 4 pea-sized parathyroid glands located posterior to the thyroid. PTH regulates the levels of calcium (Ca2+) and phosphorous and controls the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D by activating the enzyme 1 alpha-hydroxylase.
PHPT is regarded as an abnormal secretion of PTH that does not correlate with the levels of Ca2+ in the blood.1 Eighty percent of PHPT is due to a solitary adenoma in one of the parathyroid glands, 2% to 4% is secondary to multiple parathyroid adenomas, 15% is due to parathyroid hyperplasia, and 0.5% due to parathyroid carcinoma.4
Nonspecific symptomsare subtle clues of PHPT
Patients with PHPT can present with nonspecific symptoms, such as weakness, fatigue, anorexia, polyuria, polydipsia, bone and joint pain, mild depression, and mild cognitive or neuromuscular dysfunction.5 A careful history is essential to elicit these symptoms, as the patient may attribute these to aging or other causes. PHPT should also be considered when patients present with kidney stones, unexplained osteoporosis, or fragility fractures. A physical examination is seldom helpful, as parathyroid adenomas are hardly ever palpable. A slit-lamp examination may reveal corneal diseases in rare cases of hypercalcemia.6
Which lab tests, imaging should you order?
Serum Ca2+
Repeat measurements of serum Ca2+ to confirm hypercalcemia. Volume depletion, underlying malignancy, medications such as hydrochlorothiazide, lithium, and excess intake of Ca2+ carbonate can cause hypercalcemia.7 Therefore, a review of the patients’ home medications and dietary preferences in the evaluation of hypercalcemia is essential. The 2 most common causes of hypercalcemia are hyperparathyroidism and hypercalcemia of malignancy.
For hypercalcemia, establish a differential diagnosis by measuring intact PTH. An increased serum Ca2+ level along with an elevated PTH concentration should suggest PTH-dependent hypercalcemia, whereas hypercalcemia with suppressed or low-normal PTH values should suggest PTH-independent hypercalcemia (granulomatous disorders, hypercalcemia of malignancy).
Continue to: Hypercalcemia of malignancy is due to...
Hypercalcemia of malignancy is due to increased production of parathyroid hormone-related peptide from various tumor cells that initiate bone resorption and increased renal Ca2+ absorption. It can also be due to osteolysis from bone metastasis.7 It is generally severe and is a common cause of hypercalcemia in the inpatient setting.
Meticulous evaluation is vital to diagnose PHPT. Measurement of serum ionized Ca2+ reflects the biologically active Ca2+. Studies by Ong and colleagues suggest that about 24% of patients with the histologically proven parathyroid disease had isolated ionized hypercalcemia.8 It is also an important adjunct to diagnose the presumed normocalcemic PHPT in which both the ionized Ca2+ levels and serum total Ca2+ levels should be normal.9
In patients with hypoalbuminemia, a corrected serum Ca2+ is calculated using the equation: corrected Ca2+ = [0.8 × (normal albumin-patient’s albumin)] + serum Ca2+ level.
Serum PTH
Second-generation PTH assays (intact PTH) and third-generation PTH assays (bioactive PTH) are equally reliable in diagnosing PHPT.10 The results obtained with intact and bioactive PTH assays are highly correlated. Several studies have found no improvement in diagnostic accuracy when using the bioactive PTH assay.11,12
Serum PTH can be low, normal, or elevated in hypercalcemia. Hypercalcemia with a high PTH level is parathyroid-dependent hypercalcemia, whereas hypercalcemia with a suppressed PTH level is considered parathyroid-independent.
Continue to: Serum 25(OH) vitamin D
Serum 25(OH) vitamin D
Vitamin D levels are normal in PHPT and normocalcemic PHPT. However, measuring 25(OH) vitamin D in all patients with suspected PHPT is recommended to evaluate for secondary hyperparathyroidism that is due to hypocalcemia or renal failure, which can occur concomitantly with PHPT.
Normocalcemic PHPT can be differentiated from secondary hyperparathyroidism of chronic kidney disease by measuring the 1,25(OH)2 vitamin D level; it will be low in secondary hyperparathyroidism.4
Serum 1,25(OH)2 vitamin D
1,25(OH)2 vitaminD levels are elevated in about one-third of patients with PHPT, as PTH stimulates the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D.13 Although this is not a routine test, it is useful in the evaluation of parathyroid-independent hypercalcemia caused by granulomatous disease, such as sarcoidosis where there is an autonomous production of 1,25(OH)2 vitamin D leading to hypercalcemia.14
Serum creatinine and estimated glomerular filtration rate
Serum creatinine (Cr) helps assess renal function. Reduction in serum Cr clearance to <60 mL/min with no other underlying cause is an indication for parathyroidectomy.10
Serum phosphorous
PTH increases the excretion of phosphorous by inhibiting reabsorption from the proximal tubule. Therefore, serum phosphorus tends to be in the lower range of normal in PHPT, but hypophosphatemia is present in less than a quarter of patients.4
Continue to: 24-hour urinary Ca2+
24-hour urinary Ca2+
A 24-hour urinary Ca2+ excretion is used to assess the risk of renal stones and to differentiate PHPT from familial hypocalciuric hypercalcemia (FHH). Patients with FHH have an abnormality in Ca2+ receptor gene expression in parathyroid cells and renal tubular cells that could lead to parathyroid-mediated hypercalcemia and hypocalciuria. FHH is differentiated from PHPT by calculating a 24-hour urinary Ca2+/Cr ratio. A value of <0.01 is diagnostic of FHH; whereas values >0.02 indicate PHPT. The test can be more accurate when the patient is on a normal Ca2+ and salt diet, when the estimated glomerular filtration rate is >60 mL/min/1.73 m2, and when the serum 25(OH) vitamin D level is >30 ng/dL.15 Adequate urine volume is necessary for the 24-hour Ca2+/Cr ratio to be valid.
Renal imaging
Kidney stones and high Ca2+ deposits in the kidneys are the common manifestations of PHPT. Renal X-ray, computed tomography (CT), or ultrasonography are recommended in the evaluation of patients with PHPT. An incidental finding of either kidney stones or high Ca2+ deposits in the kidneys is an indication for surgery.10
Bone density/DEXA (dual energy X-ray absorptiometry) scan with a vertebral fracture assessment (VFA)
Asymptomatic PHPT individuals with osteoporosis (T-score < 2.5) or vertebral compression fracture benefit from surgical management.10 It is essential to obtain densitometry at 3 sites: the lumbar spine, the hip, and the distal third of the radius. Due to differing amounts of cortical and cancellous bone at the 3 sites and the differential effects of PTH on the cortical and cancellous bone, measurement at all 3 sites allows a clear estimation of the severity of the hyperparathyroid process on the skeleton.16 Therefore, consider measuring serum PTH if the patient has severe osteoporosis or fragility fractures that cannot be explained or that are unresponsive to treatment.
Management
The primary modality of treatment in PHPT is parathyroidectomy. The benefits are many, including an increase in bone mineral density (BMD) and reduction in fractures and kidney stones.10 With modern imaging and intra-operative PTH measurement, the success of minimally invasive parathyroidectomy is high in experienced hands. Patients with PHPT should be referred to an endocrinologist before surgery.
Surgery
Consider surgery if the patient meets any one of the following criteria:
1) overt clinical manifestations (stones, fractures)
2) serum Ca2+ >1 mg/dL above the upper limit of normal
3) Cr clearance <60 mL/min
4) low BMD with a T score ≤2.5 at any site
5) age <50 years
6) uncertain prospect for follow-up.
Continue to: Perform imaging before surgery to identify...
Perform imaging before surgery to identify the overactive parathyroid glands. Ultrasound can detect enlargement of the parathyroid glands. A sestamibi scan, which measures the uptake of Tc99-sestamibi by the parathyroid glands, reflects the activity of the parathyroid glands. In cases of nonlocalization by these 2 modalities, other imaging techniques like 4D CT scan and contrast-enhanced ultrasound can be used. Of note: Imaging is used for localization, but not for diagnosis.
Intra-operative PTH measurement has added to the efficacy of minimally invasive parathyroidectomy. A drop in PTH of >50% after 10 to 15 minutes of excising the gland is considered to be positive.10
Medication management
Monitor patients who refuse surgery or those who do not meet the criteria after surgery. Serum Ca2+ and PTH are monitored annually. DEXA scan needs to be repeated every 1 to 2 years based on the clinical picture. Also assess patients for any fragility fractures and renal endpoints. Recommend taking vitamin D to keep the level above 20 ng/dL.10 Ca2+ intake should follow normally recommended guidelines.
Bisphosphonates are primarily used for the treatment of osteoporosis accompanying PHPT. They decrease bone resorption and, to a lesser extent, bone formation. Alendronate increases BMD at the lumbar spine, but does not have much effect on Ca2+ and PTH levels.
Calcimimetics act by mimicking the effects of Ca2+ on the Ca2+ receptors present on the surface of the parathyroid cells. Therefore, calcimimetics reduce the level of parathyroid hormone and Ca2+ levels. (Long-term benefits have not been established.) Bisphosphonates are prescribed for osteoporosis and calcimimetics for hypercalcemia.10
Continue to: Conclusion
Conclusion
Although largely asymptomatic, consider PHPT when patients present with unexplained kidney stones, osteoporosis, or any nonspecific symptoms described earlier. PHPT is diagnosed by detecting an inappropriately high or normal PTH in relation to the Ca2+ level. Medications need to be reviewed, and conditions such as FHH that produce similar symptoms need to be ruled out. Measurement of 25(OH) vitamin D levels is recommended in all patients with PHPT.
Parathyroidectomy is the definitive form of treatment and should be offered to patients who meet any one of the surgical criteria, as described earlier. It can also be offered to patients who do not meet the criteria if they prefer. It is known to decrease the risk of kidney stones and osteoporosis. Medical therapy is primarily for patients who do not meet the criteria as mentioned earlier and for those who cannot and/or are unwilling to undergo surgery.
CORRESPONDENCE
Padmaja Sanapureddy, MD, Department of Primary Care and Medicine, G.V. (Sonny) Montgomery VA Medical Center, 1500 E Woodrow Wilson Ave, Jackson, MS 39216; sanapureddypadmaja@gmail.com.
1. Griebeler ML, Kearns AE, Ryu E, et al. Secular trends in the incidence of primary hyperparathyroidism over five decades (1965-2010). Bone. 2015;73:1-7.
2. Melmed S, Polonsky, KS, Larsen PR, et al. Williams Textbook of Endocrinology: Hormones and Disorders of Mineral Metabolism, 12th ed. Philadelphia, PA: Elsevier Inc, 2011:1262-1263.
3. Assadi F. Hypercalcemia: an evidence-based approach to clinical cases. Iran J Kidney Dis. 2009;3(2):71-79.
4. Bilezikian JP, Cusano NE, Khan AA, et al. Primary hyperparathyroidism. Nat Rev Dis Primers. 2016;19;2:16033.
5. Roman S, Sosa JA. Psychiatric and cognitive aspects of primary hyperparathyroidism. Curr Opin Oncol. 2007;19:1-5.
6. Berkow JW, Fine BS, Zimmerman LE. Unusual ocular calcification in hyperparathyroidism. Am J Ophthalmol. 1968;66:812-824.
7. Carroll MF, Schade DS. A practical approach to hypercalcemia. Am Fam Physician. 2003;67:1959-1966.
8. Ong GSY, Walsh JP, Stuckey BGA, et al. The importance of measuring ionized calcium in characterizing calcium status and diagnosing primary hyperparathyroidism. J Clin Endocrinol Metab. 2012;97:3138-3145.
9. Silverberg SJ, Bilezikian JP. “Incipient” primary hyperparathyroidism: a “forme fruste” of an old disease. J Clin Endocrinol Metab. 2003;88:5348-5352.
10. Eastell R, Brandi ML, Costa AG, et al. Diagnosis of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014;99:3570-3579.
11. Boudou P, Ibrahim F, Cormier C, et al. Third- or second-generation parathyroid hormone assays: a remaining debate in the diagnosis of primary hyperparathyroidism. J Clin Endocrinol Metab. 2005;90:6370-6372.
12. Carnevale V, Dionisi S, Nofroni I, et al. Potential clinical utility of a new IRMA for parathyroid hormone in postmenopausal patients with primary hyperparathyroidism. Clin Chem. 2004;50:626-631.
13. Jameson JL, De Groot L. Endocrinology: Adult and Pediatric. 7thed. Philadelphia, PA: Elsevier Inc, 2016:1109.
14. Tebben PJ, Singh RJ, Kumar R. Vitamin d-mediated hypercalcemia: mechanisms, diagnosis and treatment. Endocr Rev. 2016;37:521-547.
15. Shinall MC Jr, Dahir KM, Broome JT. Differentiating familial hypocalciuric hypercalcemia from primary hyperparathyroidism. Endocr Pract. 2013;19:697-702.
16. Castellano E, Attanasio R, Gianotti L, et al. Forearm DXA increases the rate of patients with asymptomatic primary hyperparathyroidism meeting surgical criteria. J Clin Endocrinol Metab. 2016;101:2728-2732.
Since the advent of multichannel serum chemistry screening in the 1970s, large numbers of asymptomatic cases of primary hyperparathyroidism (PHPT) have been discovered. The clinical spectrum of the disease has changed from the classic “moans, groans, bones, and stones” to an asymptomatic and subtle presentation of hypercalcemia.1,2 PHPT and malignancy are the most common causes for hypercalcemia, accounting for 90% of cases.3 In the United States, the estimated incidence of PHPT between 1998 and 2010 was about 50 per 100,000 person-years. Most patients with PHPT are older women (ages >50 years) who are asymptomatic at the time of diagnosis.1

Vigilance needed in primary care. PHPT is slowly progressive, and the patient might accept symptoms as a process of aging. Therefore, it is essential that primary care physicians (PCPs) be aware of the diagnostic and management options. A systematic approach to the diagnosis of PHPT helps differentiate the causes of hypercalcemia (TABLE2; FIGURE2). But before we discuss PHPT diagnostic clues, it’s helpful to quickly review the workings of the parathyroid glands.

How the glands work, what can go wrong
Parathyroid hormone (PTH) is secreted by 4 pea-sized parathyroid glands located posterior to the thyroid. PTH regulates the levels of calcium (Ca2+) and phosphorous and controls the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D by activating the enzyme 1 alpha-hydroxylase.
PHPT is regarded as an abnormal secretion of PTH that does not correlate with the levels of Ca2+ in the blood.1 Eighty percent of PHPT is due to a solitary adenoma in one of the parathyroid glands, 2% to 4% is secondary to multiple parathyroid adenomas, 15% is due to parathyroid hyperplasia, and 0.5% due to parathyroid carcinoma.4
Nonspecific symptomsare subtle clues of PHPT
Patients with PHPT can present with nonspecific symptoms, such as weakness, fatigue, anorexia, polyuria, polydipsia, bone and joint pain, mild depression, and mild cognitive or neuromuscular dysfunction.5 A careful history is essential to elicit these symptoms, as the patient may attribute these to aging or other causes. PHPT should also be considered when patients present with kidney stones, unexplained osteoporosis, or fragility fractures. A physical examination is seldom helpful, as parathyroid adenomas are hardly ever palpable. A slit-lamp examination may reveal corneal diseases in rare cases of hypercalcemia.6
Which lab tests, imaging should you order?
Serum Ca2+
Repeat measurements of serum Ca2+ to confirm hypercalcemia. Volume depletion, underlying malignancy, medications such as hydrochlorothiazide, lithium, and excess intake of Ca2+ carbonate can cause hypercalcemia.7 Therefore, a review of the patients’ home medications and dietary preferences in the evaluation of hypercalcemia is essential. The 2 most common causes of hypercalcemia are hyperparathyroidism and hypercalcemia of malignancy.
For hypercalcemia, establish a differential diagnosis by measuring intact PTH. An increased serum Ca2+ level along with an elevated PTH concentration should suggest PTH-dependent hypercalcemia, whereas hypercalcemia with suppressed or low-normal PTH values should suggest PTH-independent hypercalcemia (granulomatous disorders, hypercalcemia of malignancy).
Continue to: Hypercalcemia of malignancy is due to...
Hypercalcemia of malignancy is due to increased production of parathyroid hormone-related peptide from various tumor cells that initiate bone resorption and increased renal Ca2+ absorption. It can also be due to osteolysis from bone metastasis.7 It is generally severe and is a common cause of hypercalcemia in the inpatient setting.
Meticulous evaluation is vital to diagnose PHPT. Measurement of serum ionized Ca2+ reflects the biologically active Ca2+. Studies by Ong and colleagues suggest that about 24% of patients with the histologically proven parathyroid disease had isolated ionized hypercalcemia.8 It is also an important adjunct to diagnose the presumed normocalcemic PHPT in which both the ionized Ca2+ levels and serum total Ca2+ levels should be normal.9
In patients with hypoalbuminemia, a corrected serum Ca2+ is calculated using the equation: corrected Ca2+ = [0.8 × (normal albumin-patient’s albumin)] + serum Ca2+ level.
Serum PTH
Second-generation PTH assays (intact PTH) and third-generation PTH assays (bioactive PTH) are equally reliable in diagnosing PHPT.10 The results obtained with intact and bioactive PTH assays are highly correlated. Several studies have found no improvement in diagnostic accuracy when using the bioactive PTH assay.11,12
Serum PTH can be low, normal, or elevated in hypercalcemia. Hypercalcemia with a high PTH level is parathyroid-dependent hypercalcemia, whereas hypercalcemia with a suppressed PTH level is considered parathyroid-independent.
Continue to: Serum 25(OH) vitamin D
Serum 25(OH) vitamin D
Vitamin D levels are normal in PHPT and normocalcemic PHPT. However, measuring 25(OH) vitamin D in all patients with suspected PHPT is recommended to evaluate for secondary hyperparathyroidism that is due to hypocalcemia or renal failure, which can occur concomitantly with PHPT.
Normocalcemic PHPT can be differentiated from secondary hyperparathyroidism of chronic kidney disease by measuring the 1,25(OH)2 vitamin D level; it will be low in secondary hyperparathyroidism.4
Serum 1,25(OH)2 vitamin D
1,25(OH)2 vitaminD levels are elevated in about one-third of patients with PHPT, as PTH stimulates the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D.13 Although this is not a routine test, it is useful in the evaluation of parathyroid-independent hypercalcemia caused by granulomatous disease, such as sarcoidosis where there is an autonomous production of 1,25(OH)2 vitamin D leading to hypercalcemia.14
Serum creatinine and estimated glomerular filtration rate
Serum creatinine (Cr) helps assess renal function. Reduction in serum Cr clearance to <60 mL/min with no other underlying cause is an indication for parathyroidectomy.10
Serum phosphorous
PTH increases the excretion of phosphorous by inhibiting reabsorption from the proximal tubule. Therefore, serum phosphorus tends to be in the lower range of normal in PHPT, but hypophosphatemia is present in less than a quarter of patients.4
Continue to: 24-hour urinary Ca2+
24-hour urinary Ca2+
A 24-hour urinary Ca2+ excretion is used to assess the risk of renal stones and to differentiate PHPT from familial hypocalciuric hypercalcemia (FHH). Patients with FHH have an abnormality in Ca2+ receptor gene expression in parathyroid cells and renal tubular cells that could lead to parathyroid-mediated hypercalcemia and hypocalciuria. FHH is differentiated from PHPT by calculating a 24-hour urinary Ca2+/Cr ratio. A value of <0.01 is diagnostic of FHH; whereas values >0.02 indicate PHPT. The test can be more accurate when the patient is on a normal Ca2+ and salt diet, when the estimated glomerular filtration rate is >60 mL/min/1.73 m2, and when the serum 25(OH) vitamin D level is >30 ng/dL.15 Adequate urine volume is necessary for the 24-hour Ca2+/Cr ratio to be valid.
Renal imaging
Kidney stones and high Ca2+ deposits in the kidneys are the common manifestations of PHPT. Renal X-ray, computed tomography (CT), or ultrasonography are recommended in the evaluation of patients with PHPT. An incidental finding of either kidney stones or high Ca2+ deposits in the kidneys is an indication for surgery.10
Bone density/DEXA (dual energy X-ray absorptiometry) scan with a vertebral fracture assessment (VFA)
Asymptomatic PHPT individuals with osteoporosis (T-score < 2.5) or vertebral compression fracture benefit from surgical management.10 It is essential to obtain densitometry at 3 sites: the lumbar spine, the hip, and the distal third of the radius. Due to differing amounts of cortical and cancellous bone at the 3 sites and the differential effects of PTH on the cortical and cancellous bone, measurement at all 3 sites allows a clear estimation of the severity of the hyperparathyroid process on the skeleton.16 Therefore, consider measuring serum PTH if the patient has severe osteoporosis or fragility fractures that cannot be explained or that are unresponsive to treatment.
Management
The primary modality of treatment in PHPT is parathyroidectomy. The benefits are many, including an increase in bone mineral density (BMD) and reduction in fractures and kidney stones.10 With modern imaging and intra-operative PTH measurement, the success of minimally invasive parathyroidectomy is high in experienced hands. Patients with PHPT should be referred to an endocrinologist before surgery.
Surgery
Consider surgery if the patient meets any one of the following criteria:
1) overt clinical manifestations (stones, fractures)
2) serum Ca2+ >1 mg/dL above the upper limit of normal
3) Cr clearance <60 mL/min
4) low BMD with a T score ≤2.5 at any site
5) age <50 years
6) uncertain prospect for follow-up.
Continue to: Perform imaging before surgery to identify...
Perform imaging before surgery to identify the overactive parathyroid glands. Ultrasound can detect enlargement of the parathyroid glands. A sestamibi scan, which measures the uptake of Tc99-sestamibi by the parathyroid glands, reflects the activity of the parathyroid glands. In cases of nonlocalization by these 2 modalities, other imaging techniques like 4D CT scan and contrast-enhanced ultrasound can be used. Of note: Imaging is used for localization, but not for diagnosis.
Intra-operative PTH measurement has added to the efficacy of minimally invasive parathyroidectomy. A drop in PTH of >50% after 10 to 15 minutes of excising the gland is considered to be positive.10
Medication management
Monitor patients who refuse surgery or those who do not meet the criteria after surgery. Serum Ca2+ and PTH are monitored annually. DEXA scan needs to be repeated every 1 to 2 years based on the clinical picture. Also assess patients for any fragility fractures and renal endpoints. Recommend taking vitamin D to keep the level above 20 ng/dL.10 Ca2+ intake should follow normally recommended guidelines.
Bisphosphonates are primarily used for the treatment of osteoporosis accompanying PHPT. They decrease bone resorption and, to a lesser extent, bone formation. Alendronate increases BMD at the lumbar spine, but does not have much effect on Ca2+ and PTH levels.
Calcimimetics act by mimicking the effects of Ca2+ on the Ca2+ receptors present on the surface of the parathyroid cells. Therefore, calcimimetics reduce the level of parathyroid hormone and Ca2+ levels. (Long-term benefits have not been established.) Bisphosphonates are prescribed for osteoporosis and calcimimetics for hypercalcemia.10
Continue to: Conclusion
Conclusion
Although largely asymptomatic, consider PHPT when patients present with unexplained kidney stones, osteoporosis, or any nonspecific symptoms described earlier. PHPT is diagnosed by detecting an inappropriately high or normal PTH in relation to the Ca2+ level. Medications need to be reviewed, and conditions such as FHH that produce similar symptoms need to be ruled out. Measurement of 25(OH) vitamin D levels is recommended in all patients with PHPT.
Parathyroidectomy is the definitive form of treatment and should be offered to patients who meet any one of the surgical criteria, as described earlier. It can also be offered to patients who do not meet the criteria if they prefer. It is known to decrease the risk of kidney stones and osteoporosis. Medical therapy is primarily for patients who do not meet the criteria as mentioned earlier and for those who cannot and/or are unwilling to undergo surgery.
CORRESPONDENCE
Padmaja Sanapureddy, MD, Department of Primary Care and Medicine, G.V. (Sonny) Montgomery VA Medical Center, 1500 E Woodrow Wilson Ave, Jackson, MS 39216; sanapureddypadmaja@gmail.com.
Since the advent of multichannel serum chemistry screening in the 1970s, large numbers of asymptomatic cases of primary hyperparathyroidism (PHPT) have been discovered. The clinical spectrum of the disease has changed from the classic “moans, groans, bones, and stones” to an asymptomatic and subtle presentation of hypercalcemia.1,2 PHPT and malignancy are the most common causes for hypercalcemia, accounting for 90% of cases.3 In the United States, the estimated incidence of PHPT between 1998 and 2010 was about 50 per 100,000 person-years. Most patients with PHPT are older women (ages >50 years) who are asymptomatic at the time of diagnosis.1

Vigilance needed in primary care. PHPT is slowly progressive, and the patient might accept symptoms as a process of aging. Therefore, it is essential that primary care physicians (PCPs) be aware of the diagnostic and management options. A systematic approach to the diagnosis of PHPT helps differentiate the causes of hypercalcemia (TABLE2; FIGURE2). But before we discuss PHPT diagnostic clues, it’s helpful to quickly review the workings of the parathyroid glands.

How the glands work, what can go wrong
Parathyroid hormone (PTH) is secreted by 4 pea-sized parathyroid glands located posterior to the thyroid. PTH regulates the levels of calcium (Ca2+) and phosphorous and controls the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D by activating the enzyme 1 alpha-hydroxylase.
PHPT is regarded as an abnormal secretion of PTH that does not correlate with the levels of Ca2+ in the blood.1 Eighty percent of PHPT is due to a solitary adenoma in one of the parathyroid glands, 2% to 4% is secondary to multiple parathyroid adenomas, 15% is due to parathyroid hyperplasia, and 0.5% due to parathyroid carcinoma.4
Nonspecific symptomsare subtle clues of PHPT
Patients with PHPT can present with nonspecific symptoms, such as weakness, fatigue, anorexia, polyuria, polydipsia, bone and joint pain, mild depression, and mild cognitive or neuromuscular dysfunction.5 A careful history is essential to elicit these symptoms, as the patient may attribute these to aging or other causes. PHPT should also be considered when patients present with kidney stones, unexplained osteoporosis, or fragility fractures. A physical examination is seldom helpful, as parathyroid adenomas are hardly ever palpable. A slit-lamp examination may reveal corneal diseases in rare cases of hypercalcemia.6
Which lab tests, imaging should you order?
Serum Ca2+
Repeat measurements of serum Ca2+ to confirm hypercalcemia. Volume depletion, underlying malignancy, medications such as hydrochlorothiazide, lithium, and excess intake of Ca2+ carbonate can cause hypercalcemia.7 Therefore, a review of the patients’ home medications and dietary preferences in the evaluation of hypercalcemia is essential. The 2 most common causes of hypercalcemia are hyperparathyroidism and hypercalcemia of malignancy.
For hypercalcemia, establish a differential diagnosis by measuring intact PTH. An increased serum Ca2+ level along with an elevated PTH concentration should suggest PTH-dependent hypercalcemia, whereas hypercalcemia with suppressed or low-normal PTH values should suggest PTH-independent hypercalcemia (granulomatous disorders, hypercalcemia of malignancy).
Continue to: Hypercalcemia of malignancy is due to...
Hypercalcemia of malignancy is due to increased production of parathyroid hormone-related peptide from various tumor cells that initiate bone resorption and increased renal Ca2+ absorption. It can also be due to osteolysis from bone metastasis.7 It is generally severe and is a common cause of hypercalcemia in the inpatient setting.
Meticulous evaluation is vital to diagnose PHPT. Measurement of serum ionized Ca2+ reflects the biologically active Ca2+. Studies by Ong and colleagues suggest that about 24% of patients with the histologically proven parathyroid disease had isolated ionized hypercalcemia.8 It is also an important adjunct to diagnose the presumed normocalcemic PHPT in which both the ionized Ca2+ levels and serum total Ca2+ levels should be normal.9
In patients with hypoalbuminemia, a corrected serum Ca2+ is calculated using the equation: corrected Ca2+ = [0.8 × (normal albumin-patient’s albumin)] + serum Ca2+ level.
Serum PTH
Second-generation PTH assays (intact PTH) and third-generation PTH assays (bioactive PTH) are equally reliable in diagnosing PHPT.10 The results obtained with intact and bioactive PTH assays are highly correlated. Several studies have found no improvement in diagnostic accuracy when using the bioactive PTH assay.11,12
Serum PTH can be low, normal, or elevated in hypercalcemia. Hypercalcemia with a high PTH level is parathyroid-dependent hypercalcemia, whereas hypercalcemia with a suppressed PTH level is considered parathyroid-independent.
Continue to: Serum 25(OH) vitamin D
Serum 25(OH) vitamin D
Vitamin D levels are normal in PHPT and normocalcemic PHPT. However, measuring 25(OH) vitamin D in all patients with suspected PHPT is recommended to evaluate for secondary hyperparathyroidism that is due to hypocalcemia or renal failure, which can occur concomitantly with PHPT.
Normocalcemic PHPT can be differentiated from secondary hyperparathyroidism of chronic kidney disease by measuring the 1,25(OH)2 vitamin D level; it will be low in secondary hyperparathyroidism.4
Serum 1,25(OH)2 vitamin D
1,25(OH)2 vitaminD levels are elevated in about one-third of patients with PHPT, as PTH stimulates the conversion of 25(OH) vitamin D to 1,25(OH)2 vitamin D.13 Although this is not a routine test, it is useful in the evaluation of parathyroid-independent hypercalcemia caused by granulomatous disease, such as sarcoidosis where there is an autonomous production of 1,25(OH)2 vitamin D leading to hypercalcemia.14
Serum creatinine and estimated glomerular filtration rate
Serum creatinine (Cr) helps assess renal function. Reduction in serum Cr clearance to <60 mL/min with no other underlying cause is an indication for parathyroidectomy.10
Serum phosphorous
PTH increases the excretion of phosphorous by inhibiting reabsorption from the proximal tubule. Therefore, serum phosphorus tends to be in the lower range of normal in PHPT, but hypophosphatemia is present in less than a quarter of patients.4
Continue to: 24-hour urinary Ca2+
24-hour urinary Ca2+
A 24-hour urinary Ca2+ excretion is used to assess the risk of renal stones and to differentiate PHPT from familial hypocalciuric hypercalcemia (FHH). Patients with FHH have an abnormality in Ca2+ receptor gene expression in parathyroid cells and renal tubular cells that could lead to parathyroid-mediated hypercalcemia and hypocalciuria. FHH is differentiated from PHPT by calculating a 24-hour urinary Ca2+/Cr ratio. A value of <0.01 is diagnostic of FHH; whereas values >0.02 indicate PHPT. The test can be more accurate when the patient is on a normal Ca2+ and salt diet, when the estimated glomerular filtration rate is >60 mL/min/1.73 m2, and when the serum 25(OH) vitamin D level is >30 ng/dL.15 Adequate urine volume is necessary for the 24-hour Ca2+/Cr ratio to be valid.
Renal imaging
Kidney stones and high Ca2+ deposits in the kidneys are the common manifestations of PHPT. Renal X-ray, computed tomography (CT), or ultrasonography are recommended in the evaluation of patients with PHPT. An incidental finding of either kidney stones or high Ca2+ deposits in the kidneys is an indication for surgery.10
Bone density/DEXA (dual energy X-ray absorptiometry) scan with a vertebral fracture assessment (VFA)
Asymptomatic PHPT individuals with osteoporosis (T-score < 2.5) or vertebral compression fracture benefit from surgical management.10 It is essential to obtain densitometry at 3 sites: the lumbar spine, the hip, and the distal third of the radius. Due to differing amounts of cortical and cancellous bone at the 3 sites and the differential effects of PTH on the cortical and cancellous bone, measurement at all 3 sites allows a clear estimation of the severity of the hyperparathyroid process on the skeleton.16 Therefore, consider measuring serum PTH if the patient has severe osteoporosis or fragility fractures that cannot be explained or that are unresponsive to treatment.
Management
The primary modality of treatment in PHPT is parathyroidectomy. The benefits are many, including an increase in bone mineral density (BMD) and reduction in fractures and kidney stones.10 With modern imaging and intra-operative PTH measurement, the success of minimally invasive parathyroidectomy is high in experienced hands. Patients with PHPT should be referred to an endocrinologist before surgery.
Surgery
Consider surgery if the patient meets any one of the following criteria:
1) overt clinical manifestations (stones, fractures)
2) serum Ca2+ >1 mg/dL above the upper limit of normal
3) Cr clearance <60 mL/min
4) low BMD with a T score ≤2.5 at any site
5) age <50 years
6) uncertain prospect for follow-up.
Continue to: Perform imaging before surgery to identify...
Perform imaging before surgery to identify the overactive parathyroid glands. Ultrasound can detect enlargement of the parathyroid glands. A sestamibi scan, which measures the uptake of Tc99-sestamibi by the parathyroid glands, reflects the activity of the parathyroid glands. In cases of nonlocalization by these 2 modalities, other imaging techniques like 4D CT scan and contrast-enhanced ultrasound can be used. Of note: Imaging is used for localization, but not for diagnosis.
Intra-operative PTH measurement has added to the efficacy of minimally invasive parathyroidectomy. A drop in PTH of >50% after 10 to 15 minutes of excising the gland is considered to be positive.10
Medication management
Monitor patients who refuse surgery or those who do not meet the criteria after surgery. Serum Ca2+ and PTH are monitored annually. DEXA scan needs to be repeated every 1 to 2 years based on the clinical picture. Also assess patients for any fragility fractures and renal endpoints. Recommend taking vitamin D to keep the level above 20 ng/dL.10 Ca2+ intake should follow normally recommended guidelines.
Bisphosphonates are primarily used for the treatment of osteoporosis accompanying PHPT. They decrease bone resorption and, to a lesser extent, bone formation. Alendronate increases BMD at the lumbar spine, but does not have much effect on Ca2+ and PTH levels.
Calcimimetics act by mimicking the effects of Ca2+ on the Ca2+ receptors present on the surface of the parathyroid cells. Therefore, calcimimetics reduce the level of parathyroid hormone and Ca2+ levels. (Long-term benefits have not been established.) Bisphosphonates are prescribed for osteoporosis and calcimimetics for hypercalcemia.10
Continue to: Conclusion
Conclusion
Although largely asymptomatic, consider PHPT when patients present with unexplained kidney stones, osteoporosis, or any nonspecific symptoms described earlier. PHPT is diagnosed by detecting an inappropriately high or normal PTH in relation to the Ca2+ level. Medications need to be reviewed, and conditions such as FHH that produce similar symptoms need to be ruled out. Measurement of 25(OH) vitamin D levels is recommended in all patients with PHPT.
Parathyroidectomy is the definitive form of treatment and should be offered to patients who meet any one of the surgical criteria, as described earlier. It can also be offered to patients who do not meet the criteria if they prefer. It is known to decrease the risk of kidney stones and osteoporosis. Medical therapy is primarily for patients who do not meet the criteria as mentioned earlier and for those who cannot and/or are unwilling to undergo surgery.
CORRESPONDENCE
Padmaja Sanapureddy, MD, Department of Primary Care and Medicine, G.V. (Sonny) Montgomery VA Medical Center, 1500 E Woodrow Wilson Ave, Jackson, MS 39216; sanapureddypadmaja@gmail.com.
1. Griebeler ML, Kearns AE, Ryu E, et al. Secular trends in the incidence of primary hyperparathyroidism over five decades (1965-2010). Bone. 2015;73:1-7.
2. Melmed S, Polonsky, KS, Larsen PR, et al. Williams Textbook of Endocrinology: Hormones and Disorders of Mineral Metabolism, 12th ed. Philadelphia, PA: Elsevier Inc, 2011:1262-1263.
3. Assadi F. Hypercalcemia: an evidence-based approach to clinical cases. Iran J Kidney Dis. 2009;3(2):71-79.
4. Bilezikian JP, Cusano NE, Khan AA, et al. Primary hyperparathyroidism. Nat Rev Dis Primers. 2016;19;2:16033.
5. Roman S, Sosa JA. Psychiatric and cognitive aspects of primary hyperparathyroidism. Curr Opin Oncol. 2007;19:1-5.
6. Berkow JW, Fine BS, Zimmerman LE. Unusual ocular calcification in hyperparathyroidism. Am J Ophthalmol. 1968;66:812-824.
7. Carroll MF, Schade DS. A practical approach to hypercalcemia. Am Fam Physician. 2003;67:1959-1966.
8. Ong GSY, Walsh JP, Stuckey BGA, et al. The importance of measuring ionized calcium in characterizing calcium status and diagnosing primary hyperparathyroidism. J Clin Endocrinol Metab. 2012;97:3138-3145.
9. Silverberg SJ, Bilezikian JP. “Incipient” primary hyperparathyroidism: a “forme fruste” of an old disease. J Clin Endocrinol Metab. 2003;88:5348-5352.
10. Eastell R, Brandi ML, Costa AG, et al. Diagnosis of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014;99:3570-3579.
11. Boudou P, Ibrahim F, Cormier C, et al. Third- or second-generation parathyroid hormone assays: a remaining debate in the diagnosis of primary hyperparathyroidism. J Clin Endocrinol Metab. 2005;90:6370-6372.
12. Carnevale V, Dionisi S, Nofroni I, et al. Potential clinical utility of a new IRMA for parathyroid hormone in postmenopausal patients with primary hyperparathyroidism. Clin Chem. 2004;50:626-631.
13. Jameson JL, De Groot L. Endocrinology: Adult and Pediatric. 7thed. Philadelphia, PA: Elsevier Inc, 2016:1109.
14. Tebben PJ, Singh RJ, Kumar R. Vitamin d-mediated hypercalcemia: mechanisms, diagnosis and treatment. Endocr Rev. 2016;37:521-547.
15. Shinall MC Jr, Dahir KM, Broome JT. Differentiating familial hypocalciuric hypercalcemia from primary hyperparathyroidism. Endocr Pract. 2013;19:697-702.
16. Castellano E, Attanasio R, Gianotti L, et al. Forearm DXA increases the rate of patients with asymptomatic primary hyperparathyroidism meeting surgical criteria. J Clin Endocrinol Metab. 2016;101:2728-2732.
1. Griebeler ML, Kearns AE, Ryu E, et al. Secular trends in the incidence of primary hyperparathyroidism over five decades (1965-2010). Bone. 2015;73:1-7.
2. Melmed S, Polonsky, KS, Larsen PR, et al. Williams Textbook of Endocrinology: Hormones and Disorders of Mineral Metabolism, 12th ed. Philadelphia, PA: Elsevier Inc, 2011:1262-1263.
3. Assadi F. Hypercalcemia: an evidence-based approach to clinical cases. Iran J Kidney Dis. 2009;3(2):71-79.
4. Bilezikian JP, Cusano NE, Khan AA, et al. Primary hyperparathyroidism. Nat Rev Dis Primers. 2016;19;2:16033.
5. Roman S, Sosa JA. Psychiatric and cognitive aspects of primary hyperparathyroidism. Curr Opin Oncol. 2007;19:1-5.
6. Berkow JW, Fine BS, Zimmerman LE. Unusual ocular calcification in hyperparathyroidism. Am J Ophthalmol. 1968;66:812-824.
7. Carroll MF, Schade DS. A practical approach to hypercalcemia. Am Fam Physician. 2003;67:1959-1966.
8. Ong GSY, Walsh JP, Stuckey BGA, et al. The importance of measuring ionized calcium in characterizing calcium status and diagnosing primary hyperparathyroidism. J Clin Endocrinol Metab. 2012;97:3138-3145.
9. Silverberg SJ, Bilezikian JP. “Incipient” primary hyperparathyroidism: a “forme fruste” of an old disease. J Clin Endocrinol Metab. 2003;88:5348-5352.
10. Eastell R, Brandi ML, Costa AG, et al. Diagnosis of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014;99:3570-3579.
11. Boudou P, Ibrahim F, Cormier C, et al. Third- or second-generation parathyroid hormone assays: a remaining debate in the diagnosis of primary hyperparathyroidism. J Clin Endocrinol Metab. 2005;90:6370-6372.
12. Carnevale V, Dionisi S, Nofroni I, et al. Potential clinical utility of a new IRMA for parathyroid hormone in postmenopausal patients with primary hyperparathyroidism. Clin Chem. 2004;50:626-631.
13. Jameson JL, De Groot L. Endocrinology: Adult and Pediatric. 7thed. Philadelphia, PA: Elsevier Inc, 2016:1109.
14. Tebben PJ, Singh RJ, Kumar R. Vitamin d-mediated hypercalcemia: mechanisms, diagnosis and treatment. Endocr Rev. 2016;37:521-547.
15. Shinall MC Jr, Dahir KM, Broome JT. Differentiating familial hypocalciuric hypercalcemia from primary hyperparathyroidism. Endocr Pract. 2013;19:697-702.
16. Castellano E, Attanasio R, Gianotti L, et al. Forearm DXA increases the rate of patients with asymptomatic primary hyperparathyroidism meeting surgical criteria. J Clin Endocrinol Metab. 2016;101:2728-2732.
PRACTICE RECOMMENDATIONS
› Evaluate suspected cases of primary hyperparathyroidism (PHPT) with serum total calcium, parathyroid hormone (PTH), creatinine, and 25-hydroxy vitamin D levels. A
› Consider 24-hour urine measurement of calcium and creatinine in patients undergoing evaluation for possible PHPT. A
› Obtain bone densitometry at the spine, hip, and distal radius in patients with PHPT. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Adult foot fractures: A guide
The evaluation and management of acute musculoskeletal conditions are frequently handled by primary care providers.1 It’s estimated that up to 14% of orthopedic complaints encountered by family physicians involve fractures,2 and approximately 15% of these are foot fractures.2 Diagnosis requires radiographic evaluation, but ultrasound is proving useful, too. This article reviews the diagnosis and management of adult foot fractures, with an emphasis on when advanced imaging and referral are indicated (TABLE1,3-10).
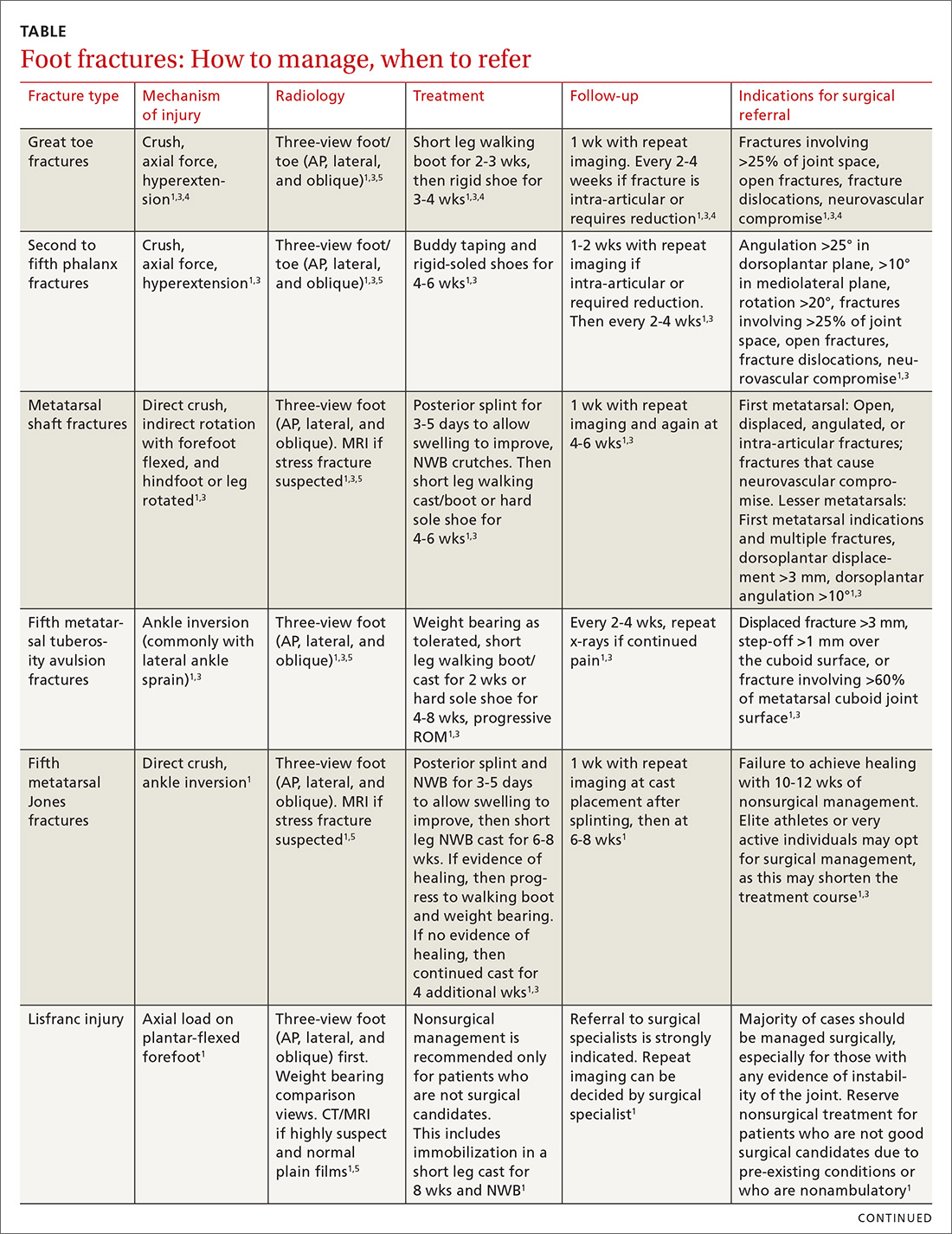
Phalanx fractures: The most common foot fractures
Phalanx fractures typically occur by crush injury, hyperextension, or direct axial force (eg, stubbing the toe).3 Patients with phalanx fractures typically present with pain at or near the site of injury, edema, ecchymosis, and erythema. Throbbing pain is characteristic, and dependent position may worsen the pain.1 Emergently evaluate any fracture causing tenting of the skin, protrusion from the skin, or neurovascular compromise, and attempt realignment to regain neurovascular function.

Most patients with phalanx fractures have point tenderness over the site of the fracture; however, this may also occur with contusions. Placing a gentle loading force along the long axis of the bone distal to the injury may help you differentiate between a contusion and a fracture.4 Pain observed with axial loading of the bone during examination points to a fracture rather than a contusion.
Differential diagnosis
Obtain imaging, including anterior-posterior (AP), lateral, and oblique views at a minimum, for all patients in whom you suspect fractures.5 Multiple fractures of the phalanges are common; therefore, always thoroughly examine the phalanges adjacent to the injured one.
Sesamoid bone fractures are uncommon but do occur and are usually due to direct injury from jumping or landing. The most common sesamoid to be injured is the medial sesamoid of the great toe, although the lateral sesamoid can also be injured. Bipartite sesamoids can occur and may confuse the examiner due to their similar appearance on x-rays to a sesamoid fracture.1 These normal variants often appear smooth and are commonly bilateral as opposed to the jagged or abrupt edges of a fracture. Stress fractures occur as well and are typically due to overuse-type injuries.
Other causes of pain similar to that experienced with phalanx fractures include soft tissue injuries to adjacent ligaments, tendons, and muscles. To help discern the cause of pain, evaluate nail beds for subungual hematomas, indicating injury to the nail bed causing bleeding and pressure under the nail. Obvious deformities of the toes or metatarsal-phalangeal joints signal the possibility of a fracture-dislocation. First metatarsophalangeal (MTP) sprain (“turf toe,” a condition common in athletes who hyperextend the toe, such as when pushing off from hard surfaces like turf) and gout can also present with acute pain in the first phalanx.
Treatment
Due to the role of the great toe in weight bearing and balance, great toe fractures are sometimes managed differently than fractures in Toes 2 through 5. Proper alignment and healing from a fracture in the first toe are critical to prevent future pain and other sequelae. Refer for orthopedic evaluation great toe fractures with displacement, angulation, rotational deformity, neurovascular compromise, >25% involvement of the joint space, or obvious dislocation.1 If referral is not indicated, treat great toe fractures with a short leg walking cast/boot for 2 to 3 weeks followed by buddy taping and use of a hard-soled shoe for 3 to 4 weeks.1
Continue to: With regard to the lesser toes...
With regard to the lesser toes, refer patients with fracture-dislocations, displaced intra-articular fractures, and fractures that do not reduce easily. Nondisplaced fractures of the lesser toes do not require surgical referral.1 These can be treated with splinting (buddy taping) and use of rigid-sole shoes for 4 to 6 weeks. Treatment duration depends largely on patient compliance; generally, continue treatment until point tenderness resolves.1
Treatment of sesamoid fractures consists of resting the affected foot with a walking boot, hard-soled shoe, or “donut” pad under the sesamoid bone to help distribute weight on the foot when standing. Length of treatment is approximately 6 to 8 weeks for most fractures.1 Consider surgical referral if nonoperative management is unsuccessful.
Metatarsal fractures: Look for malalignment
Metatarsal fractures account for 5% of all foot fractures encountered in primary care.2 These fractures typically occur as a result of falls, direct trauma, or rotational injuries (eg, ankle and foot sprains).1 In athletes, the most common cause of these fractures is high rotational force. Patients typically present with pain over the injury site, swelling, bruising, and pain with weight bearing.
As part of your exam, look for malalignment, rotational deformities, and evidence of open fracture. Palpation at the site of the fracture may increase the pain; however, as is true with phalanx fractures, contusions may also cause significant tenderness upon palpation. Also assess range of motion—with special attention to signs of malrotation—and evaluate the adjacent metatarsals, as multiple bones, ligaments, or both are often involved.1
Fifth metatarsal fractures are the most common in adults, likely because of decreased cortical thickness as compared with the other metatarsals.3,11 In addition, multiple soft tissue attachments connect at the proximal aspect of the fifth metatarsal. Classification of these types of metatarsal fractures is based on anatomic location.3 Jones fractures are one type of fracture at the proximal aspect of the fifth metatarsal that occur at the metaphyseal-diaphyseal junction specifically (FIGURE 1). Because this area receives its blood supply from small terminal vessels, fractures here have a high risk of non-union and, thus, should be top of mind in any patient with tenderness at the base of the fifth metatarsal.
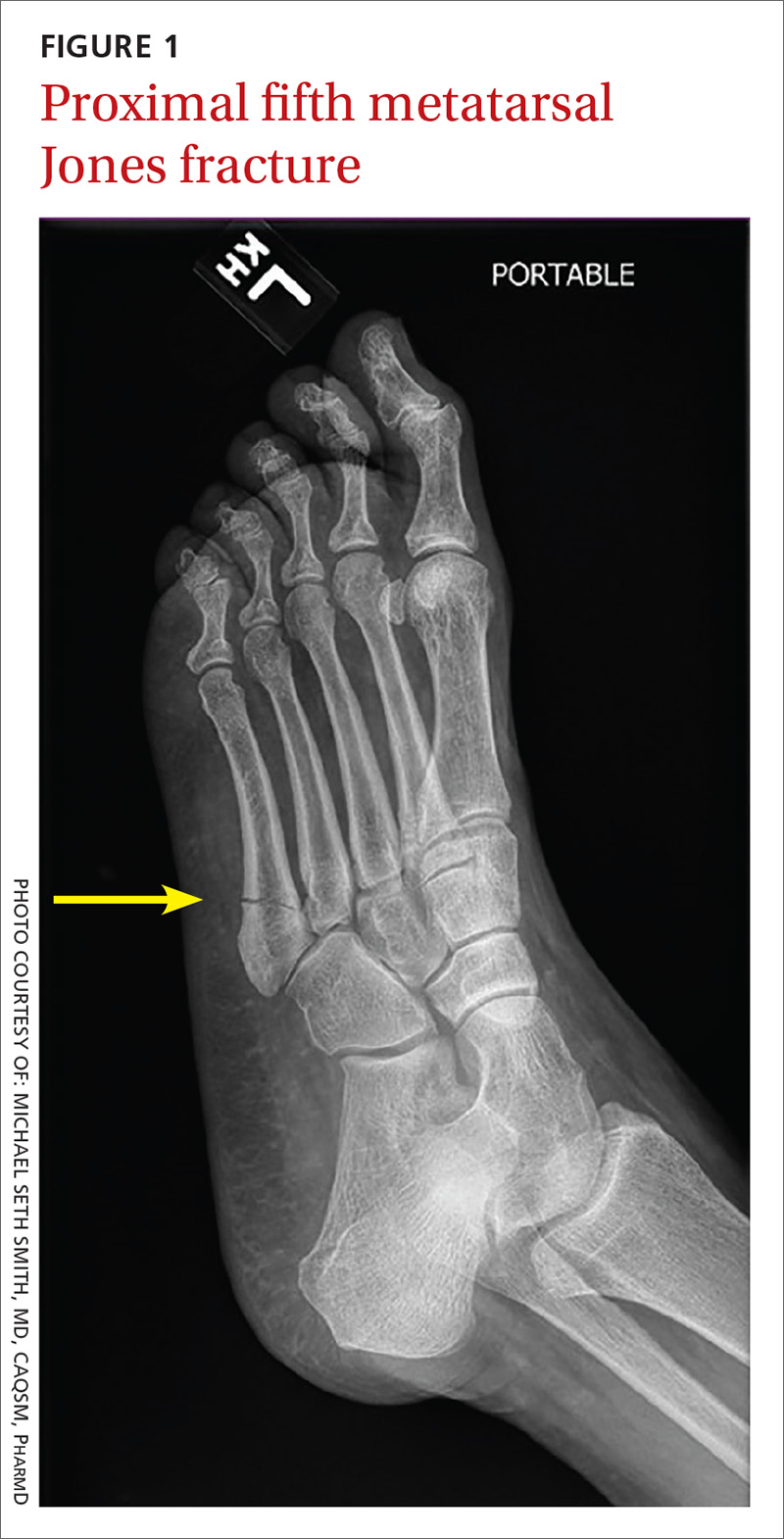
Continue to: MRI/ultrasound in addition to plain films?
MRI/ultrasound in addition to plain films?
Use the Ottawa Ankle and Foot Rules to determine the need for foot radiographs in the acute setting.12 If indicated, imaging should include AP, lateral, and oblique views of the foot.5 Consider magnetic resonance imaging (MRI) if you suspect a stress fracture, which typically presents as an overuse injury in athletes.
Ultrasound may be effective for identifying metatarsal fractures, as well.13 Ultrasound can be used to visualize fractures of the long metatarsal bones and identify displacement, angulation, and step-offs. Although its use in the United States has been limited for this purpose, studies in other countries are showing that it yields results comparable to plain films in the emergency department setting for diagnosis and initial management of these fractures.13
Differential diagnosis
Multiple other diagnoses present similarly to metatarsal fractures including: ligamentous/tendon/soft tissue injuries, interdigital neuroma, sesamoid fractures, and Lisfranc ligament injury (which we'll discuss in a bit). Stress fractures of the metatarsal, most commonly seen in the second metatarsal, are insidious in nature and are common with repetitive movement such as that made by gymnasts, dancers, and track athletes.14 Metatarsalgia, which causes pain at the ball of the foot, is a condition that can stem from a myriad of causes including a low or high arch, biomechanics, and previous injury.
Treatment
Nondisplaced, minimally displaced (<3 mm), and minimally angulated (<10° dorsal/plantar angulation) fractures of any metatarsal shaft may be managed conservatively with a hard-soled shoe or walking boot with weight bearing as tolerated for 4 to 6 weeks.1 Most stress fractures (excluding fifth metatarsal stress fractures) may be treated similarly. Surgical referral is indicated for patients with any open fracture, first metatarsal fractures with displacement, central metatarsal fractures with >10° deformity, >3 mm of translation, multiple fractures, or fifth metatarsal stress fracture.1
Base of fifth metatarsal tuberosity avulsion fractures can be managed nonoperatively with protected weight bearing in a boot or cast for 2 weeks or hard sole shoe for 4 to 8 weeks if the fracture is at the proximal tubercle (see Zone I, FIGURE 2). Jones fractures (Zone II, FIGURE 2) or fractures of the proximal diaphysis (Zone III, FIGURE 2) of the fifth metatarsal may also be managed nonoperatively in patients who are not competitive athletes. This can be done with a non-weight bearing (NWB) short leg cast for 6 to 8 weeks at a minimum.4 If the patient is an athlete who wishes to resume competition, surgical referral is indicated, as the risk of nonunion is high with these fractures.15
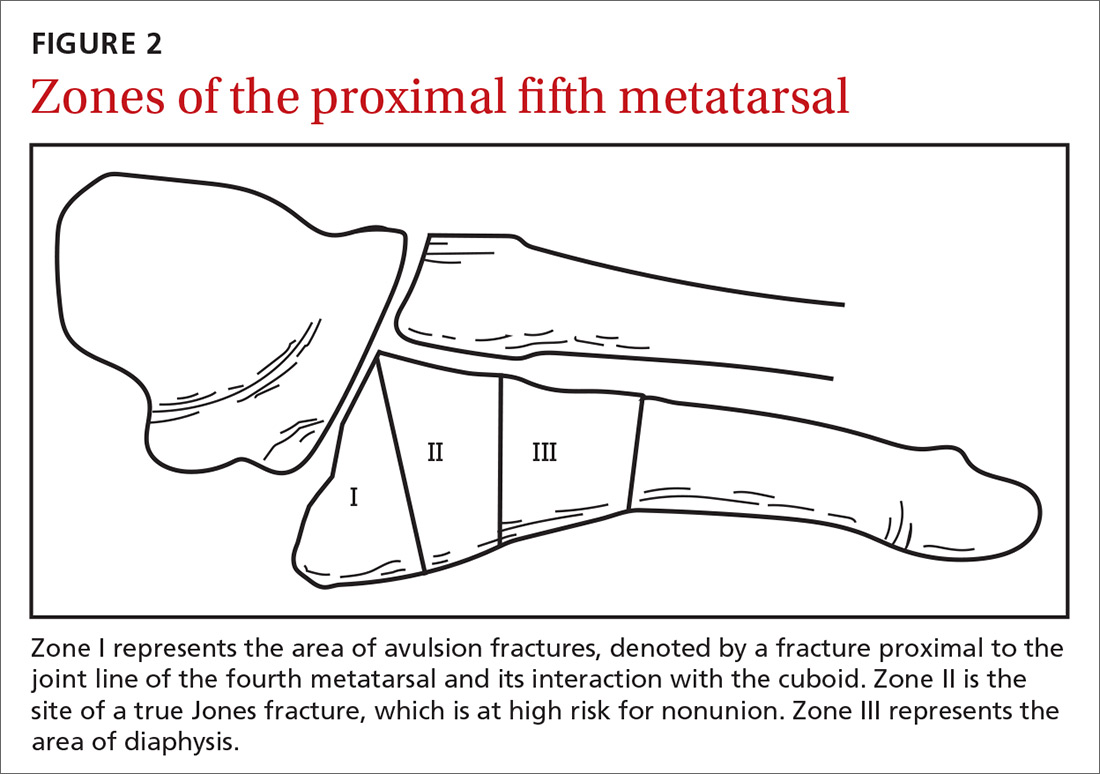
Continue to: Stress fractures and stress reactions...
Stress fractures and stress reactions (early evidence of bone edema seen on MRI, indicating progression to stress fracture) of the proximal fifth metatarsal should be managed with strict activity modifications and a NWB short leg cast for 6 to 8 weeks, given the high risk of nonunion.
Midfoot fractures: The cuboid, cuneiforms, and navicular bone
Although fractures are less common in the midfoot, the midfoot serves an important role in weight bearing and stabilization. In addition, along with the metatarsal bones, the midfoot is critical to proper alignment and articulation.
Cuboid and cuneiform fractures
Cuboid fractures may occur with high-velocity trauma, foot inversion with external rotation of the tibia, a direct blow, or axial load on a plantar-flexed heel. Pain with weight bearing or with walking on toes is usually present. Cuneiform fractures are less common and rarely occur in isolation.6 Mechanisms of injury for cuneiform fractures include direct impact, axial loading on a dorsiflexed or plantar-flexed foot with rotational force, and severe rotation of the midfoot section in a fixed foot. Pain is usually localized to the dorsal or dorsomedial aspect of the foot.
With these 2 midfoot injuries, the exam should include palpation of the cuboid, navicular, and cuneiform bones and careful inspection of the Lisfranc joint, as this can be injured in midfoot fractures.7 Obtain AP, lateral, and oblique views of the foot,5 and strongly consider bilateral weight-bearing films to evaluate for tarsometatarsal (TMT) joint complex injuries (typically Lisfranc joint complex injuries), given their association with midfoot fractures1 (FIGURE 3).
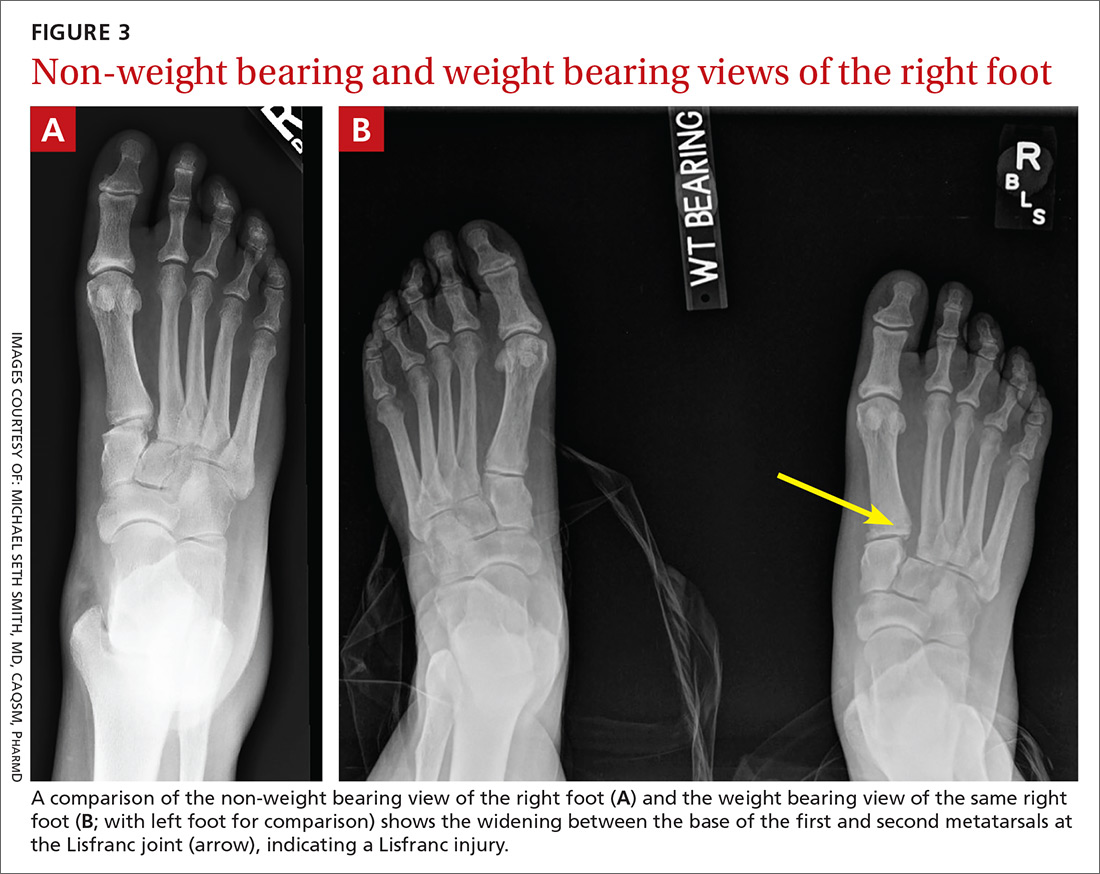
If the fracture is at or near the TMT joint complex, obtain a CT scan or MRI regardless of plain film findings to evaluate the Lisfranc joint complex. Because the Lisfranc joint is particularly important in midfoot stability, untreated injuries can lead to impaired gait or chronic foot pain and deformity. Early identification and surgical referral for these injuries is crucial.
Continue to: Navicular fractures
Navicular fractures
Navicular fractures are typically caused by a twisting mechanism with forced plantar flexion or forced dorsiflexion of the midfoot. They present with severe pain over the dorsal or dorsomedial foot, particularly while bearing weight. Tenderness to palpation over the navicular bone generally warrants imaging studies to rule out fracture, as undiagnosed fractures can lead to severe long-term disability. Use Ottawa Ankle and Foot Rules12 in the acute setting to determine the need for radiographs. If imaging is indicated, obtain AP, lateral, and oblique views.
Tuberosity fractures may be seen on the AP view, while dorsal avulsions, talonavicular joint disruptions, and naviculo-cuneiform joint injuries are better seen on lateral views.16 Patients, particularly cross-country and track athletes, presenting with insidious onset of pain over the navicular bone should be evaluated for stress fracture using MRI, even in the presence of normal radiographs.
Differential diagnosis. Suspect Lisfranc joint complex injuries in any patient with mid-foot pain or fracture. The transverse arch of the foot is reliant upon the articulation of the second metatarsal with all 5 neighboring bones. The Lisfranc ligament is the strongest of 3 supporting ligaments to anchor the TMT joint complex. Other causes of mid-foot pain include soft tissue injury, contusion, and tendinopathy. In addition, other conditions that may cause pain in this area include cuboid syndrome, peroneal tendinopathy, Jones fracture, stress fracture, anterior calcaneal fracture, and sinus tarsi syndrome.
Treatment. Nondisplaced fractures of the cuboid or cuneiform may be treated with a short leg walking cast/boot for 6 weeks followed by the use of a shoe with a thin, rigid, longitudinal arch support for an additional 6 weeks.1 Fractures requiring referral for surgical evaluation include fractures that are open and fractures with vascular or neurological compromise. Also refer comminuted fractures and those that present with >2 mm step-off. Lastly, midfoot fractures that involve the Lisfranc joint should be immobilized and referred for orthopedic evaluation with instructions to the patient to avoid weight bearing until orthopedic evaluation.8
Avulsion fractures of the navicular bone may be managed nonoperatively with a short leg walking cast/boot if there is <20% involvement of the talonavicular surface. Simple nondisplaced body fractures may also be managed conservatively with immobilization and protected weight bearing for 6 to 8 weeks.15 Refer for surgical evaluation avulsion fractures that are intra-articular or dorsal involving 20% or more of the talonavicular surface and tuberosity fractures, given their risk of nonunion.9 All navicular body fractures that are not longitudinal in nature should also be referred for surgical evaluation. Navicular stress fractures that do not extend into the plantar cortex may be managed conservatively with a minimum of 6 weeks of a short leg cast and strict NWB with close follow-up.17
Continue to: Calcaneal fractures
Calcaneal fractures
Calcaneal fractures typically occur from severe axial load or fall from a height. Weight bearing is usually limited and secondary to significant pain. Tenderness to palpation over the calcaneus or with squeezing of the heel will produce pain on exam. Initial x-rays should include a lateral and axial view of the calcaneus. Additional imaging, including a CT scan, may be indicated for further evaluation to determine the extent of the fracture or to determine if a fracture is present despite normal x-rays.10
Acute compartment syndrome occurs in 10% of calcaneal fractures and must be considered in patients with calcaneal fractures from severe trauma.18 Tendon injuries of the ankle, hindfoot, and midfoot may present similarly but can be ruled out with clinical exam and appropriate imaging.
Treatment
Avulsion fractures that do not involve more than 25% of the calcaneocuboid joint and nondisplaced calcaneal fractures may be managed conservatively by instructing patients to wear a NWB short leg cast/boot for 4 to 6 weeks.1 Refer for surgical evaluation patients with calcaneal fracture fragments >1 cm, displacement >3 mm, open fractures, joint involvement >25%, and those whose symptoms fail to resolve with conservative management. Stress fractures can be managed conservatively with cessation of aggravating activities and immobilization in a walking boot until symptoms resolve, which typically takes 4 to 6 weeks.1
CORRESPONDENCE
Michael Seth Smith, MD, CAQSM, PharmD, 3450 Hull Road, Gainesville, FL 32611; smithms@ortho.ufl.edu
1. Eiff MP, Hatch RL, et al. Fracture Management for Primary Care. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012.
2. Hatch RL, Rosenbaum Cl. Fracture care by family physicians. A review of 295 cases. J Fam Pract. 1994;38:238-244.
3. Bica D, Sprouse RA, Armen J. Diagnosis and management of common foot fractures. Am Fam Physician. 2016;93:183-191.
4. Hatch RL, Hacking S. Evaluation and management of toe fractures. Am Fam Physician. 2003;68:2413-2418.
5. Snider RK. Essentials of Musculoskeletal Care. 2nd ed. Rosemont, IL: American Orthopedic Surgeons; 2001.
6. Guler F, Baz AB, Turan A, et al. Isolated medial cuneiform fractures: report of two cases and review of the literature. Foot Ankle Spec. 2011;4:306-309.
7. Borrelli J, De S, Van Pelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20:472.
8. Pinney SJ, Sangeorzan BJ. Fractures of the tarsal bones. Orthop Clin North Am. 2001:32:21-33.
9. Rosenbaum AJ, Uhl R, DiPreta JA. Acute fractures of the tarsal navicular. Orthopedics. 2014;37:541-546.
10. Sanders RW, Clare MP. Calcaneous fractures. In: Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:2064.
11. Singer G, Cichocki M, Schalamon J, et al. A study of metatarsal fractures in children. J Bone Joint Surg Am. 2008;90:772-776.
12. Stiell IG, Greenberg GH, McKnight RD, et al. A study to develop clinical decision rules for the use of radiography in acute ankle injuries. Ann Emerg Med. 1992;21:384-390.
13. Ekinci S, Polat O, Günalp M, et al. The accuracy of ultrasound evaluation in foot and ankle trauma. Am J Emerg Med. 2013;31:1551-1555.
14. Welck MJ, Hayes T, Pastides P, et al. Stress fractures of the foot and ankle. Injury. 2017;48:1722-1726.
15. Hatch RL, Alsobrook JA, Clugston JR. Diagnosis and management of metatarsal fractures. Am Fam Physician. 2007;76:817-826.
16. Schildhauer TA, Coulibaly MO, Hoffman MF. Fractures and dislocations of the midfoot and forefoot. In: Rockwood and Green’s Fractures in Adults. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2015:2690.
17. Mayer SW, Joyner PW, Almekinders LC, et al. Stress fractures of the foot and ankle in athletes. Sports Health. 2014;6:481-491.
18. Kalsi R, Dempsey A, Bunney EB. Compartment syndrome of the foot after calcaneal fracture. J Emerg Med. 2012;43:e101-106.
The evaluation and management of acute musculoskeletal conditions are frequently handled by primary care providers.1 It’s estimated that up to 14% of orthopedic complaints encountered by family physicians involve fractures,2 and approximately 15% of these are foot fractures.2 Diagnosis requires radiographic evaluation, but ultrasound is proving useful, too. This article reviews the diagnosis and management of adult foot fractures, with an emphasis on when advanced imaging and referral are indicated (TABLE1,3-10).

Phalanx fractures: The most common foot fractures
Phalanx fractures typically occur by crush injury, hyperextension, or direct axial force (eg, stubbing the toe).3 Patients with phalanx fractures typically present with pain at or near the site of injury, edema, ecchymosis, and erythema. Throbbing pain is characteristic, and dependent position may worsen the pain.1 Emergently evaluate any fracture causing tenting of the skin, protrusion from the skin, or neurovascular compromise, and attempt realignment to regain neurovascular function.

Most patients with phalanx fractures have point tenderness over the site of the fracture; however, this may also occur with contusions. Placing a gentle loading force along the long axis of the bone distal to the injury may help you differentiate between a contusion and a fracture.4 Pain observed with axial loading of the bone during examination points to a fracture rather than a contusion.
Differential diagnosis
Obtain imaging, including anterior-posterior (AP), lateral, and oblique views at a minimum, for all patients in whom you suspect fractures.5 Multiple fractures of the phalanges are common; therefore, always thoroughly examine the phalanges adjacent to the injured one.
Sesamoid bone fractures are uncommon but do occur and are usually due to direct injury from jumping or landing. The most common sesamoid to be injured is the medial sesamoid of the great toe, although the lateral sesamoid can also be injured. Bipartite sesamoids can occur and may confuse the examiner due to their similar appearance on x-rays to a sesamoid fracture.1 These normal variants often appear smooth and are commonly bilateral as opposed to the jagged or abrupt edges of a fracture. Stress fractures occur as well and are typically due to overuse-type injuries.
Other causes of pain similar to that experienced with phalanx fractures include soft tissue injuries to adjacent ligaments, tendons, and muscles. To help discern the cause of pain, evaluate nail beds for subungual hematomas, indicating injury to the nail bed causing bleeding and pressure under the nail. Obvious deformities of the toes or metatarsal-phalangeal joints signal the possibility of a fracture-dislocation. First metatarsophalangeal (MTP) sprain (“turf toe,” a condition common in athletes who hyperextend the toe, such as when pushing off from hard surfaces like turf) and gout can also present with acute pain in the first phalanx.
Treatment
Due to the role of the great toe in weight bearing and balance, great toe fractures are sometimes managed differently than fractures in Toes 2 through 5. Proper alignment and healing from a fracture in the first toe are critical to prevent future pain and other sequelae. Refer for orthopedic evaluation great toe fractures with displacement, angulation, rotational deformity, neurovascular compromise, >25% involvement of the joint space, or obvious dislocation.1 If referral is not indicated, treat great toe fractures with a short leg walking cast/boot for 2 to 3 weeks followed by buddy taping and use of a hard-soled shoe for 3 to 4 weeks.1
Continue to: With regard to the lesser toes...
With regard to the lesser toes, refer patients with fracture-dislocations, displaced intra-articular fractures, and fractures that do not reduce easily. Nondisplaced fractures of the lesser toes do not require surgical referral.1 These can be treated with splinting (buddy taping) and use of rigid-sole shoes for 4 to 6 weeks. Treatment duration depends largely on patient compliance; generally, continue treatment until point tenderness resolves.1
Treatment of sesamoid fractures consists of resting the affected foot with a walking boot, hard-soled shoe, or “donut” pad under the sesamoid bone to help distribute weight on the foot when standing. Length of treatment is approximately 6 to 8 weeks for most fractures.1 Consider surgical referral if nonoperative management is unsuccessful.
Metatarsal fractures: Look for malalignment
Metatarsal fractures account for 5% of all foot fractures encountered in primary care.2 These fractures typically occur as a result of falls, direct trauma, or rotational injuries (eg, ankle and foot sprains).1 In athletes, the most common cause of these fractures is high rotational force. Patients typically present with pain over the injury site, swelling, bruising, and pain with weight bearing.
As part of your exam, look for malalignment, rotational deformities, and evidence of open fracture. Palpation at the site of the fracture may increase the pain; however, as is true with phalanx fractures, contusions may also cause significant tenderness upon palpation. Also assess range of motion—with special attention to signs of malrotation—and evaluate the adjacent metatarsals, as multiple bones, ligaments, or both are often involved.1
Fifth metatarsal fractures are the most common in adults, likely because of decreased cortical thickness as compared with the other metatarsals.3,11 In addition, multiple soft tissue attachments connect at the proximal aspect of the fifth metatarsal. Classification of these types of metatarsal fractures is based on anatomic location.3 Jones fractures are one type of fracture at the proximal aspect of the fifth metatarsal that occur at the metaphyseal-diaphyseal junction specifically (FIGURE 1). Because this area receives its blood supply from small terminal vessels, fractures here have a high risk of non-union and, thus, should be top of mind in any patient with tenderness at the base of the fifth metatarsal.

Continue to: MRI/ultrasound in addition to plain films?
MRI/ultrasound in addition to plain films?
Use the Ottawa Ankle and Foot Rules to determine the need for foot radiographs in the acute setting.12 If indicated, imaging should include AP, lateral, and oblique views of the foot.5 Consider magnetic resonance imaging (MRI) if you suspect a stress fracture, which typically presents as an overuse injury in athletes.
Ultrasound may be effective for identifying metatarsal fractures, as well.13 Ultrasound can be used to visualize fractures of the long metatarsal bones and identify displacement, angulation, and step-offs. Although its use in the United States has been limited for this purpose, studies in other countries are showing that it yields results comparable to plain films in the emergency department setting for diagnosis and initial management of these fractures.13
Differential diagnosis
Multiple other diagnoses present similarly to metatarsal fractures including: ligamentous/tendon/soft tissue injuries, interdigital neuroma, sesamoid fractures, and Lisfranc ligament injury (which we'll discuss in a bit). Stress fractures of the metatarsal, most commonly seen in the second metatarsal, are insidious in nature and are common with repetitive movement such as that made by gymnasts, dancers, and track athletes.14 Metatarsalgia, which causes pain at the ball of the foot, is a condition that can stem from a myriad of causes including a low or high arch, biomechanics, and previous injury.
Treatment
Nondisplaced, minimally displaced (<3 mm), and minimally angulated (<10° dorsal/plantar angulation) fractures of any metatarsal shaft may be managed conservatively with a hard-soled shoe or walking boot with weight bearing as tolerated for 4 to 6 weeks.1 Most stress fractures (excluding fifth metatarsal stress fractures) may be treated similarly. Surgical referral is indicated for patients with any open fracture, first metatarsal fractures with displacement, central metatarsal fractures with >10° deformity, >3 mm of translation, multiple fractures, or fifth metatarsal stress fracture.1
Base of fifth metatarsal tuberosity avulsion fractures can be managed nonoperatively with protected weight bearing in a boot or cast for 2 weeks or hard sole shoe for 4 to 8 weeks if the fracture is at the proximal tubercle (see Zone I, FIGURE 2). Jones fractures (Zone II, FIGURE 2) or fractures of the proximal diaphysis (Zone III, FIGURE 2) of the fifth metatarsal may also be managed nonoperatively in patients who are not competitive athletes. This can be done with a non-weight bearing (NWB) short leg cast for 6 to 8 weeks at a minimum.4 If the patient is an athlete who wishes to resume competition, surgical referral is indicated, as the risk of nonunion is high with these fractures.15

Continue to: Stress fractures and stress reactions...
Stress fractures and stress reactions (early evidence of bone edema seen on MRI, indicating progression to stress fracture) of the proximal fifth metatarsal should be managed with strict activity modifications and a NWB short leg cast for 6 to 8 weeks, given the high risk of nonunion.
Midfoot fractures: The cuboid, cuneiforms, and navicular bone
Although fractures are less common in the midfoot, the midfoot serves an important role in weight bearing and stabilization. In addition, along with the metatarsal bones, the midfoot is critical to proper alignment and articulation.
Cuboid and cuneiform fractures
Cuboid fractures may occur with high-velocity trauma, foot inversion with external rotation of the tibia, a direct blow, or axial load on a plantar-flexed heel. Pain with weight bearing or with walking on toes is usually present. Cuneiform fractures are less common and rarely occur in isolation.6 Mechanisms of injury for cuneiform fractures include direct impact, axial loading on a dorsiflexed or plantar-flexed foot with rotational force, and severe rotation of the midfoot section in a fixed foot. Pain is usually localized to the dorsal or dorsomedial aspect of the foot.
With these 2 midfoot injuries, the exam should include palpation of the cuboid, navicular, and cuneiform bones and careful inspection of the Lisfranc joint, as this can be injured in midfoot fractures.7 Obtain AP, lateral, and oblique views of the foot,5 and strongly consider bilateral weight-bearing films to evaluate for tarsometatarsal (TMT) joint complex injuries (typically Lisfranc joint complex injuries), given their association with midfoot fractures1 (FIGURE 3).

If the fracture is at or near the TMT joint complex, obtain a CT scan or MRI regardless of plain film findings to evaluate the Lisfranc joint complex. Because the Lisfranc joint is particularly important in midfoot stability, untreated injuries can lead to impaired gait or chronic foot pain and deformity. Early identification and surgical referral for these injuries is crucial.
Continue to: Navicular fractures
Navicular fractures
Navicular fractures are typically caused by a twisting mechanism with forced plantar flexion or forced dorsiflexion of the midfoot. They present with severe pain over the dorsal or dorsomedial foot, particularly while bearing weight. Tenderness to palpation over the navicular bone generally warrants imaging studies to rule out fracture, as undiagnosed fractures can lead to severe long-term disability. Use Ottawa Ankle and Foot Rules12 in the acute setting to determine the need for radiographs. If imaging is indicated, obtain AP, lateral, and oblique views.
Tuberosity fractures may be seen on the AP view, while dorsal avulsions, talonavicular joint disruptions, and naviculo-cuneiform joint injuries are better seen on lateral views.16 Patients, particularly cross-country and track athletes, presenting with insidious onset of pain over the navicular bone should be evaluated for stress fracture using MRI, even in the presence of normal radiographs.
Differential diagnosis. Suspect Lisfranc joint complex injuries in any patient with mid-foot pain or fracture. The transverse arch of the foot is reliant upon the articulation of the second metatarsal with all 5 neighboring bones. The Lisfranc ligament is the strongest of 3 supporting ligaments to anchor the TMT joint complex. Other causes of mid-foot pain include soft tissue injury, contusion, and tendinopathy. In addition, other conditions that may cause pain in this area include cuboid syndrome, peroneal tendinopathy, Jones fracture, stress fracture, anterior calcaneal fracture, and sinus tarsi syndrome.
Treatment. Nondisplaced fractures of the cuboid or cuneiform may be treated with a short leg walking cast/boot for 6 weeks followed by the use of a shoe with a thin, rigid, longitudinal arch support for an additional 6 weeks.1 Fractures requiring referral for surgical evaluation include fractures that are open and fractures with vascular or neurological compromise. Also refer comminuted fractures and those that present with >2 mm step-off. Lastly, midfoot fractures that involve the Lisfranc joint should be immobilized and referred for orthopedic evaluation with instructions to the patient to avoid weight bearing until orthopedic evaluation.8
Avulsion fractures of the navicular bone may be managed nonoperatively with a short leg walking cast/boot if there is <20% involvement of the talonavicular surface. Simple nondisplaced body fractures may also be managed conservatively with immobilization and protected weight bearing for 6 to 8 weeks.15 Refer for surgical evaluation avulsion fractures that are intra-articular or dorsal involving 20% or more of the talonavicular surface and tuberosity fractures, given their risk of nonunion.9 All navicular body fractures that are not longitudinal in nature should also be referred for surgical evaluation. Navicular stress fractures that do not extend into the plantar cortex may be managed conservatively with a minimum of 6 weeks of a short leg cast and strict NWB with close follow-up.17
Continue to: Calcaneal fractures
Calcaneal fractures
Calcaneal fractures typically occur from severe axial load or fall from a height. Weight bearing is usually limited and secondary to significant pain. Tenderness to palpation over the calcaneus or with squeezing of the heel will produce pain on exam. Initial x-rays should include a lateral and axial view of the calcaneus. Additional imaging, including a CT scan, may be indicated for further evaluation to determine the extent of the fracture or to determine if a fracture is present despite normal x-rays.10
Acute compartment syndrome occurs in 10% of calcaneal fractures and must be considered in patients with calcaneal fractures from severe trauma.18 Tendon injuries of the ankle, hindfoot, and midfoot may present similarly but can be ruled out with clinical exam and appropriate imaging.
Treatment
Avulsion fractures that do not involve more than 25% of the calcaneocuboid joint and nondisplaced calcaneal fractures may be managed conservatively by instructing patients to wear a NWB short leg cast/boot for 4 to 6 weeks.1 Refer for surgical evaluation patients with calcaneal fracture fragments >1 cm, displacement >3 mm, open fractures, joint involvement >25%, and those whose symptoms fail to resolve with conservative management. Stress fractures can be managed conservatively with cessation of aggravating activities and immobilization in a walking boot until symptoms resolve, which typically takes 4 to 6 weeks.1
CORRESPONDENCE
Michael Seth Smith, MD, CAQSM, PharmD, 3450 Hull Road, Gainesville, FL 32611; smithms@ortho.ufl.edu
The evaluation and management of acute musculoskeletal conditions are frequently handled by primary care providers.1 It’s estimated that up to 14% of orthopedic complaints encountered by family physicians involve fractures,2 and approximately 15% of these are foot fractures.2 Diagnosis requires radiographic evaluation, but ultrasound is proving useful, too. This article reviews the diagnosis and management of adult foot fractures, with an emphasis on when advanced imaging and referral are indicated (TABLE1,3-10).

Phalanx fractures: The most common foot fractures
Phalanx fractures typically occur by crush injury, hyperextension, or direct axial force (eg, stubbing the toe).3 Patients with phalanx fractures typically present with pain at or near the site of injury, edema, ecchymosis, and erythema. Throbbing pain is characteristic, and dependent position may worsen the pain.1 Emergently evaluate any fracture causing tenting of the skin, protrusion from the skin, or neurovascular compromise, and attempt realignment to regain neurovascular function.

Most patients with phalanx fractures have point tenderness over the site of the fracture; however, this may also occur with contusions. Placing a gentle loading force along the long axis of the bone distal to the injury may help you differentiate between a contusion and a fracture.4 Pain observed with axial loading of the bone during examination points to a fracture rather than a contusion.
Differential diagnosis
Obtain imaging, including anterior-posterior (AP), lateral, and oblique views at a minimum, for all patients in whom you suspect fractures.5 Multiple fractures of the phalanges are common; therefore, always thoroughly examine the phalanges adjacent to the injured one.
Sesamoid bone fractures are uncommon but do occur and are usually due to direct injury from jumping or landing. The most common sesamoid to be injured is the medial sesamoid of the great toe, although the lateral sesamoid can also be injured. Bipartite sesamoids can occur and may confuse the examiner due to their similar appearance on x-rays to a sesamoid fracture.1 These normal variants often appear smooth and are commonly bilateral as opposed to the jagged or abrupt edges of a fracture. Stress fractures occur as well and are typically due to overuse-type injuries.
Other causes of pain similar to that experienced with phalanx fractures include soft tissue injuries to adjacent ligaments, tendons, and muscles. To help discern the cause of pain, evaluate nail beds for subungual hematomas, indicating injury to the nail bed causing bleeding and pressure under the nail. Obvious deformities of the toes or metatarsal-phalangeal joints signal the possibility of a fracture-dislocation. First metatarsophalangeal (MTP) sprain (“turf toe,” a condition common in athletes who hyperextend the toe, such as when pushing off from hard surfaces like turf) and gout can also present with acute pain in the first phalanx.
Treatment
Due to the role of the great toe in weight bearing and balance, great toe fractures are sometimes managed differently than fractures in Toes 2 through 5. Proper alignment and healing from a fracture in the first toe are critical to prevent future pain and other sequelae. Refer for orthopedic evaluation great toe fractures with displacement, angulation, rotational deformity, neurovascular compromise, >25% involvement of the joint space, or obvious dislocation.1 If referral is not indicated, treat great toe fractures with a short leg walking cast/boot for 2 to 3 weeks followed by buddy taping and use of a hard-soled shoe for 3 to 4 weeks.1
Continue to: With regard to the lesser toes...
With regard to the lesser toes, refer patients with fracture-dislocations, displaced intra-articular fractures, and fractures that do not reduce easily. Nondisplaced fractures of the lesser toes do not require surgical referral.1 These can be treated with splinting (buddy taping) and use of rigid-sole shoes for 4 to 6 weeks. Treatment duration depends largely on patient compliance; generally, continue treatment until point tenderness resolves.1
Treatment of sesamoid fractures consists of resting the affected foot with a walking boot, hard-soled shoe, or “donut” pad under the sesamoid bone to help distribute weight on the foot when standing. Length of treatment is approximately 6 to 8 weeks for most fractures.1 Consider surgical referral if nonoperative management is unsuccessful.
Metatarsal fractures: Look for malalignment
Metatarsal fractures account for 5% of all foot fractures encountered in primary care.2 These fractures typically occur as a result of falls, direct trauma, or rotational injuries (eg, ankle and foot sprains).1 In athletes, the most common cause of these fractures is high rotational force. Patients typically present with pain over the injury site, swelling, bruising, and pain with weight bearing.
As part of your exam, look for malalignment, rotational deformities, and evidence of open fracture. Palpation at the site of the fracture may increase the pain; however, as is true with phalanx fractures, contusions may also cause significant tenderness upon palpation. Also assess range of motion—with special attention to signs of malrotation—and evaluate the adjacent metatarsals, as multiple bones, ligaments, or both are often involved.1
Fifth metatarsal fractures are the most common in adults, likely because of decreased cortical thickness as compared with the other metatarsals.3,11 In addition, multiple soft tissue attachments connect at the proximal aspect of the fifth metatarsal. Classification of these types of metatarsal fractures is based on anatomic location.3 Jones fractures are one type of fracture at the proximal aspect of the fifth metatarsal that occur at the metaphyseal-diaphyseal junction specifically (FIGURE 1). Because this area receives its blood supply from small terminal vessels, fractures here have a high risk of non-union and, thus, should be top of mind in any patient with tenderness at the base of the fifth metatarsal.

Continue to: MRI/ultrasound in addition to plain films?
MRI/ultrasound in addition to plain films?
Use the Ottawa Ankle and Foot Rules to determine the need for foot radiographs in the acute setting.12 If indicated, imaging should include AP, lateral, and oblique views of the foot.5 Consider magnetic resonance imaging (MRI) if you suspect a stress fracture, which typically presents as an overuse injury in athletes.
Ultrasound may be effective for identifying metatarsal fractures, as well.13 Ultrasound can be used to visualize fractures of the long metatarsal bones and identify displacement, angulation, and step-offs. Although its use in the United States has been limited for this purpose, studies in other countries are showing that it yields results comparable to plain films in the emergency department setting for diagnosis and initial management of these fractures.13
Differential diagnosis
Multiple other diagnoses present similarly to metatarsal fractures including: ligamentous/tendon/soft tissue injuries, interdigital neuroma, sesamoid fractures, and Lisfranc ligament injury (which we'll discuss in a bit). Stress fractures of the metatarsal, most commonly seen in the second metatarsal, are insidious in nature and are common with repetitive movement such as that made by gymnasts, dancers, and track athletes.14 Metatarsalgia, which causes pain at the ball of the foot, is a condition that can stem from a myriad of causes including a low or high arch, biomechanics, and previous injury.
Treatment
Nondisplaced, minimally displaced (<3 mm), and minimally angulated (<10° dorsal/plantar angulation) fractures of any metatarsal shaft may be managed conservatively with a hard-soled shoe or walking boot with weight bearing as tolerated for 4 to 6 weeks.1 Most stress fractures (excluding fifth metatarsal stress fractures) may be treated similarly. Surgical referral is indicated for patients with any open fracture, first metatarsal fractures with displacement, central metatarsal fractures with >10° deformity, >3 mm of translation, multiple fractures, or fifth metatarsal stress fracture.1
Base of fifth metatarsal tuberosity avulsion fractures can be managed nonoperatively with protected weight bearing in a boot or cast for 2 weeks or hard sole shoe for 4 to 8 weeks if the fracture is at the proximal tubercle (see Zone I, FIGURE 2). Jones fractures (Zone II, FIGURE 2) or fractures of the proximal diaphysis (Zone III, FIGURE 2) of the fifth metatarsal may also be managed nonoperatively in patients who are not competitive athletes. This can be done with a non-weight bearing (NWB) short leg cast for 6 to 8 weeks at a minimum.4 If the patient is an athlete who wishes to resume competition, surgical referral is indicated, as the risk of nonunion is high with these fractures.15

Continue to: Stress fractures and stress reactions...
Stress fractures and stress reactions (early evidence of bone edema seen on MRI, indicating progression to stress fracture) of the proximal fifth metatarsal should be managed with strict activity modifications and a NWB short leg cast for 6 to 8 weeks, given the high risk of nonunion.
Midfoot fractures: The cuboid, cuneiforms, and navicular bone
Although fractures are less common in the midfoot, the midfoot serves an important role in weight bearing and stabilization. In addition, along with the metatarsal bones, the midfoot is critical to proper alignment and articulation.
Cuboid and cuneiform fractures
Cuboid fractures may occur with high-velocity trauma, foot inversion with external rotation of the tibia, a direct blow, or axial load on a plantar-flexed heel. Pain with weight bearing or with walking on toes is usually present. Cuneiform fractures are less common and rarely occur in isolation.6 Mechanisms of injury for cuneiform fractures include direct impact, axial loading on a dorsiflexed or plantar-flexed foot with rotational force, and severe rotation of the midfoot section in a fixed foot. Pain is usually localized to the dorsal or dorsomedial aspect of the foot.
With these 2 midfoot injuries, the exam should include palpation of the cuboid, navicular, and cuneiform bones and careful inspection of the Lisfranc joint, as this can be injured in midfoot fractures.7 Obtain AP, lateral, and oblique views of the foot,5 and strongly consider bilateral weight-bearing films to evaluate for tarsometatarsal (TMT) joint complex injuries (typically Lisfranc joint complex injuries), given their association with midfoot fractures1 (FIGURE 3).

If the fracture is at or near the TMT joint complex, obtain a CT scan or MRI regardless of plain film findings to evaluate the Lisfranc joint complex. Because the Lisfranc joint is particularly important in midfoot stability, untreated injuries can lead to impaired gait or chronic foot pain and deformity. Early identification and surgical referral for these injuries is crucial.
Continue to: Navicular fractures
Navicular fractures
Navicular fractures are typically caused by a twisting mechanism with forced plantar flexion or forced dorsiflexion of the midfoot. They present with severe pain over the dorsal or dorsomedial foot, particularly while bearing weight. Tenderness to palpation over the navicular bone generally warrants imaging studies to rule out fracture, as undiagnosed fractures can lead to severe long-term disability. Use Ottawa Ankle and Foot Rules12 in the acute setting to determine the need for radiographs. If imaging is indicated, obtain AP, lateral, and oblique views.
Tuberosity fractures may be seen on the AP view, while dorsal avulsions, talonavicular joint disruptions, and naviculo-cuneiform joint injuries are better seen on lateral views.16 Patients, particularly cross-country and track athletes, presenting with insidious onset of pain over the navicular bone should be evaluated for stress fracture using MRI, even in the presence of normal radiographs.
Differential diagnosis. Suspect Lisfranc joint complex injuries in any patient with mid-foot pain or fracture. The transverse arch of the foot is reliant upon the articulation of the second metatarsal with all 5 neighboring bones. The Lisfranc ligament is the strongest of 3 supporting ligaments to anchor the TMT joint complex. Other causes of mid-foot pain include soft tissue injury, contusion, and tendinopathy. In addition, other conditions that may cause pain in this area include cuboid syndrome, peroneal tendinopathy, Jones fracture, stress fracture, anterior calcaneal fracture, and sinus tarsi syndrome.
Treatment. Nondisplaced fractures of the cuboid or cuneiform may be treated with a short leg walking cast/boot for 6 weeks followed by the use of a shoe with a thin, rigid, longitudinal arch support for an additional 6 weeks.1 Fractures requiring referral for surgical evaluation include fractures that are open and fractures with vascular or neurological compromise. Also refer comminuted fractures and those that present with >2 mm step-off. Lastly, midfoot fractures that involve the Lisfranc joint should be immobilized and referred for orthopedic evaluation with instructions to the patient to avoid weight bearing until orthopedic evaluation.8
Avulsion fractures of the navicular bone may be managed nonoperatively with a short leg walking cast/boot if there is <20% involvement of the talonavicular surface. Simple nondisplaced body fractures may also be managed conservatively with immobilization and protected weight bearing for 6 to 8 weeks.15 Refer for surgical evaluation avulsion fractures that are intra-articular or dorsal involving 20% or more of the talonavicular surface and tuberosity fractures, given their risk of nonunion.9 All navicular body fractures that are not longitudinal in nature should also be referred for surgical evaluation. Navicular stress fractures that do not extend into the plantar cortex may be managed conservatively with a minimum of 6 weeks of a short leg cast and strict NWB with close follow-up.17
Continue to: Calcaneal fractures
Calcaneal fractures
Calcaneal fractures typically occur from severe axial load or fall from a height. Weight bearing is usually limited and secondary to significant pain. Tenderness to palpation over the calcaneus or with squeezing of the heel will produce pain on exam. Initial x-rays should include a lateral and axial view of the calcaneus. Additional imaging, including a CT scan, may be indicated for further evaluation to determine the extent of the fracture or to determine if a fracture is present despite normal x-rays.10
Acute compartment syndrome occurs in 10% of calcaneal fractures and must be considered in patients with calcaneal fractures from severe trauma.18 Tendon injuries of the ankle, hindfoot, and midfoot may present similarly but can be ruled out with clinical exam and appropriate imaging.
Treatment
Avulsion fractures that do not involve more than 25% of the calcaneocuboid joint and nondisplaced calcaneal fractures may be managed conservatively by instructing patients to wear a NWB short leg cast/boot for 4 to 6 weeks.1 Refer for surgical evaluation patients with calcaneal fracture fragments >1 cm, displacement >3 mm, open fractures, joint involvement >25%, and those whose symptoms fail to resolve with conservative management. Stress fractures can be managed conservatively with cessation of aggravating activities and immobilization in a walking boot until symptoms resolve, which typically takes 4 to 6 weeks.1
CORRESPONDENCE
Michael Seth Smith, MD, CAQSM, PharmD, 3450 Hull Road, Gainesville, FL 32611; smithms@ortho.ufl.edu
1. Eiff MP, Hatch RL, et al. Fracture Management for Primary Care. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012.
2. Hatch RL, Rosenbaum Cl. Fracture care by family physicians. A review of 295 cases. J Fam Pract. 1994;38:238-244.
3. Bica D, Sprouse RA, Armen J. Diagnosis and management of common foot fractures. Am Fam Physician. 2016;93:183-191.
4. Hatch RL, Hacking S. Evaluation and management of toe fractures. Am Fam Physician. 2003;68:2413-2418.
5. Snider RK. Essentials of Musculoskeletal Care. 2nd ed. Rosemont, IL: American Orthopedic Surgeons; 2001.
6. Guler F, Baz AB, Turan A, et al. Isolated medial cuneiform fractures: report of two cases and review of the literature. Foot Ankle Spec. 2011;4:306-309.
7. Borrelli J, De S, Van Pelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20:472.
8. Pinney SJ, Sangeorzan BJ. Fractures of the tarsal bones. Orthop Clin North Am. 2001:32:21-33.
9. Rosenbaum AJ, Uhl R, DiPreta JA. Acute fractures of the tarsal navicular. Orthopedics. 2014;37:541-546.
10. Sanders RW, Clare MP. Calcaneous fractures. In: Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:2064.
11. Singer G, Cichocki M, Schalamon J, et al. A study of metatarsal fractures in children. J Bone Joint Surg Am. 2008;90:772-776.
12. Stiell IG, Greenberg GH, McKnight RD, et al. A study to develop clinical decision rules for the use of radiography in acute ankle injuries. Ann Emerg Med. 1992;21:384-390.
13. Ekinci S, Polat O, Günalp M, et al. The accuracy of ultrasound evaluation in foot and ankle trauma. Am J Emerg Med. 2013;31:1551-1555.
14. Welck MJ, Hayes T, Pastides P, et al. Stress fractures of the foot and ankle. Injury. 2017;48:1722-1726.
15. Hatch RL, Alsobrook JA, Clugston JR. Diagnosis and management of metatarsal fractures. Am Fam Physician. 2007;76:817-826.
16. Schildhauer TA, Coulibaly MO, Hoffman MF. Fractures and dislocations of the midfoot and forefoot. In: Rockwood and Green’s Fractures in Adults. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2015:2690.
17. Mayer SW, Joyner PW, Almekinders LC, et al. Stress fractures of the foot and ankle in athletes. Sports Health. 2014;6:481-491.
18. Kalsi R, Dempsey A, Bunney EB. Compartment syndrome of the foot after calcaneal fracture. J Emerg Med. 2012;43:e101-106.
1. Eiff MP, Hatch RL, et al. Fracture Management for Primary Care. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012.
2. Hatch RL, Rosenbaum Cl. Fracture care by family physicians. A review of 295 cases. J Fam Pract. 1994;38:238-244.
3. Bica D, Sprouse RA, Armen J. Diagnosis and management of common foot fractures. Am Fam Physician. 2016;93:183-191.
4. Hatch RL, Hacking S. Evaluation and management of toe fractures. Am Fam Physician. 2003;68:2413-2418.
5. Snider RK. Essentials of Musculoskeletal Care. 2nd ed. Rosemont, IL: American Orthopedic Surgeons; 2001.
6. Guler F, Baz AB, Turan A, et al. Isolated medial cuneiform fractures: report of two cases and review of the literature. Foot Ankle Spec. 2011;4:306-309.
7. Borrelli J, De S, Van Pelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20:472.
8. Pinney SJ, Sangeorzan BJ. Fractures of the tarsal bones. Orthop Clin North Am. 2001:32:21-33.
9. Rosenbaum AJ, Uhl R, DiPreta JA. Acute fractures of the tarsal navicular. Orthopedics. 2014;37:541-546.
10. Sanders RW, Clare MP. Calcaneous fractures. In: Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:2064.
11. Singer G, Cichocki M, Schalamon J, et al. A study of metatarsal fractures in children. J Bone Joint Surg Am. 2008;90:772-776.
12. Stiell IG, Greenberg GH, McKnight RD, et al. A study to develop clinical decision rules for the use of radiography in acute ankle injuries. Ann Emerg Med. 1992;21:384-390.
13. Ekinci S, Polat O, Günalp M, et al. The accuracy of ultrasound evaluation in foot and ankle trauma. Am J Emerg Med. 2013;31:1551-1555.
14. Welck MJ, Hayes T, Pastides P, et al. Stress fractures of the foot and ankle. Injury. 2017;48:1722-1726.
15. Hatch RL, Alsobrook JA, Clugston JR. Diagnosis and management of metatarsal fractures. Am Fam Physician. 2007;76:817-826.
16. Schildhauer TA, Coulibaly MO, Hoffman MF. Fractures and dislocations of the midfoot and forefoot. In: Rockwood and Green’s Fractures in Adults. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2015:2690.
17. Mayer SW, Joyner PW, Almekinders LC, et al. Stress fractures of the foot and ankle in athletes. Sports Health. 2014;6:481-491.
18. Kalsi R, Dempsey A, Bunney EB. Compartment syndrome of the foot after calcaneal fracture. J Emerg Med. 2012;43:e101-106.
PRACTICE RECOMMENDATIONS
› Manage most fractures of the proximal fifth metatarsal metaphyseal-diaphyseal junction (Jones fracture) conservatively with appropriate treatment and close follow-up; refer for early surgical evaluation only those patients who are highly active and who are interested in a faster return to activity. B
› Use the Ottawa Ankle and Foot Rules to determine whether x-rays are needed in a patient with foot pain and a suspected fracture. A
› Start with weight-bearing x-rays and then consider computed tomography or magnetic resonance imaging for complete evaluation of suspected injury to the Lisfranc joint. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series