User login
The Official Newspaper of the American Association for Thoracic Surgery
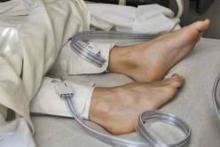
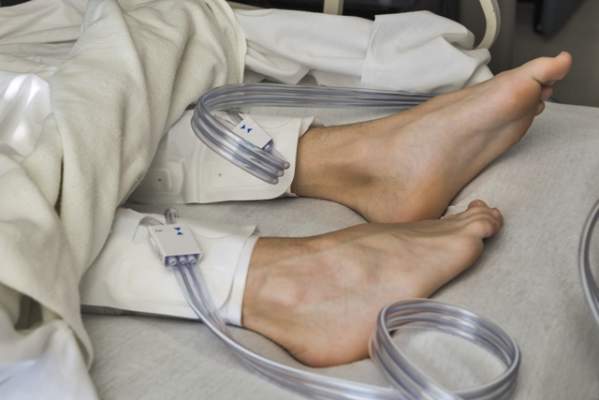
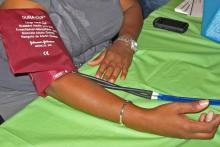
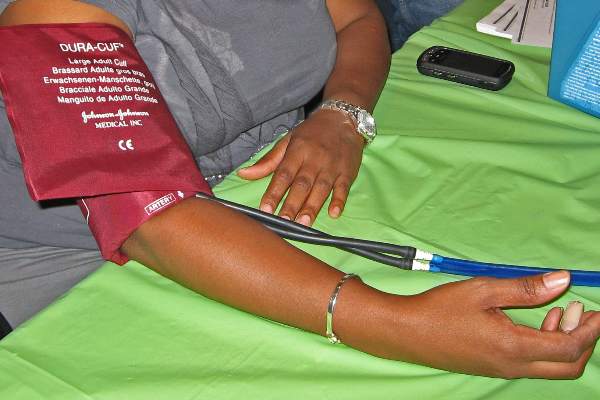
IPC maintains first-line status in preventing DVT in most surgical patients
Intermittent pneumatic compression remains the consensus choice as the sole prophylactic agent for deep vein thrombosis in low- or moderate-risk surgical patients, according to a literature analysis published online ahead of print in September in the Journal of Vascular Surgery: Venous and Lymphatic Disorders.
Dr. Nirvana Sadaghianloo of the University of Nice (France) Sophia Antipolis and Dr. Alan Dardik of Yale University in New Haven, Conn., used the MEDLINE and Cochrane libraries to find individual studies and meta-analyses published in English since 2011 assessing the efficacy of intermittent pneumatic compression (IPC) in preventing deep vein thrombosis (DVT), which included the American College of Chest Physicians ninth edition guidelines (2012). They stated that, although the overall quality of studies regarding the use of IPC was low, IPC showed efficacy in prevention of DVT for more than 30 years (J Vasc Surg Venous Lymphat Disord. 2015 doi: 10.1016/j.jvsv.2015.07.006).
“IPC represents a good alternative to pharmacologic agents when the risk of thrombosis is moderate or low or when the risk of bleeding is high or may have serious consequences for the patient,” they stated.
However, they also found that most recommendations suggested that, in high-risk patients, IPC plays a role primarily as an additional modality to provide additional benefit in preventing DVT when is used in combination with pharmacologic therapy. They highlighted how the choice of any thromboprophylactic agent required a systematic risk assessment as a critical prerequisite.
Overall, risk stratification was most frequently assessed by the Caprini or Rogers score for most general, abdominal-pelvic, bariatric, vascular, plastic, and gynecologic surgery patients and by the Padua Prediction Score for hospitalized medical patients. In addition, major orthopedic surgery patients and stroke patients with restricted mobility were usually considered high risk, Dr. Sadaghianloo and Dr. Dardik said.
From their assessment of the literature, they determined that “further studies are needed to assess practical clinical questions that remain unanswered, including optimal cuff length and location, sequence and duration of pressure, and whether use of IPC in an outpatient setting can be effective and achieve good compliance.”
The authors reported that they had no conflicts of interest.
Intermittent pneumatic compression remains the consensus choice as the sole prophylactic agent for deep vein thrombosis in low- or moderate-risk surgical patients, according to a literature analysis published online ahead of print in September in the Journal of Vascular Surgery: Venous and Lymphatic Disorders.
Dr. Nirvana Sadaghianloo of the University of Nice (France) Sophia Antipolis and Dr. Alan Dardik of Yale University in New Haven, Conn., used the MEDLINE and Cochrane libraries to find individual studies and meta-analyses published in English since 2011 assessing the efficacy of intermittent pneumatic compression (IPC) in preventing deep vein thrombosis (DVT), which included the American College of Chest Physicians ninth edition guidelines (2012). They stated that, although the overall quality of studies regarding the use of IPC was low, IPC showed efficacy in prevention of DVT for more than 30 years (J Vasc Surg Venous Lymphat Disord. 2015 doi: 10.1016/j.jvsv.2015.07.006).
“IPC represents a good alternative to pharmacologic agents when the risk of thrombosis is moderate or low or when the risk of bleeding is high or may have serious consequences for the patient,” they stated.
However, they also found that most recommendations suggested that, in high-risk patients, IPC plays a role primarily as an additional modality to provide additional benefit in preventing DVT when is used in combination with pharmacologic therapy. They highlighted how the choice of any thromboprophylactic agent required a systematic risk assessment as a critical prerequisite.
Overall, risk stratification was most frequently assessed by the Caprini or Rogers score for most general, abdominal-pelvic, bariatric, vascular, plastic, and gynecologic surgery patients and by the Padua Prediction Score for hospitalized medical patients. In addition, major orthopedic surgery patients and stroke patients with restricted mobility were usually considered high risk, Dr. Sadaghianloo and Dr. Dardik said.
From their assessment of the literature, they determined that “further studies are needed to assess practical clinical questions that remain unanswered, including optimal cuff length and location, sequence and duration of pressure, and whether use of IPC in an outpatient setting can be effective and achieve good compliance.”
The authors reported that they had no conflicts of interest.
Intermittent pneumatic compression remains the consensus choice as the sole prophylactic agent for deep vein thrombosis in low- or moderate-risk surgical patients, according to a literature analysis published online ahead of print in September in the Journal of Vascular Surgery: Venous and Lymphatic Disorders.
Dr. Nirvana Sadaghianloo of the University of Nice (France) Sophia Antipolis and Dr. Alan Dardik of Yale University in New Haven, Conn., used the MEDLINE and Cochrane libraries to find individual studies and meta-analyses published in English since 2011 assessing the efficacy of intermittent pneumatic compression (IPC) in preventing deep vein thrombosis (DVT), which included the American College of Chest Physicians ninth edition guidelines (2012). They stated that, although the overall quality of studies regarding the use of IPC was low, IPC showed efficacy in prevention of DVT for more than 30 years (J Vasc Surg Venous Lymphat Disord. 2015 doi: 10.1016/j.jvsv.2015.07.006).
“IPC represents a good alternative to pharmacologic agents when the risk of thrombosis is moderate or low or when the risk of bleeding is high or may have serious consequences for the patient,” they stated.
However, they also found that most recommendations suggested that, in high-risk patients, IPC plays a role primarily as an additional modality to provide additional benefit in preventing DVT when is used in combination with pharmacologic therapy. They highlighted how the choice of any thromboprophylactic agent required a systematic risk assessment as a critical prerequisite.
Overall, risk stratification was most frequently assessed by the Caprini or Rogers score for most general, abdominal-pelvic, bariatric, vascular, plastic, and gynecologic surgery patients and by the Padua Prediction Score for hospitalized medical patients. In addition, major orthopedic surgery patients and stroke patients with restricted mobility were usually considered high risk, Dr. Sadaghianloo and Dr. Dardik said.
From their assessment of the literature, they determined that “further studies are needed to assess practical clinical questions that remain unanswered, including optimal cuff length and location, sequence and duration of pressure, and whether use of IPC in an outpatient setting can be effective and achieve good compliance.”
The authors reported that they had no conflicts of interest.
FROM JOURNAL OF VASCULAR SURGERY: VENOUS AND LYMPHATIC DISORDERS
Key clinical point: IPC is efficacious as the sole prophylactic agent in low- or moderate-risk surgical patients and in patients with high risk of bleeding with drug therapy.
Major finding: In high-risk patients, IPC is an added modality for preventing DVT in combination with pharmacologic prophylaxis.
Data source: Researchers performed an assessment of the literature in English since 2011 in MEDLINE and the Cochrane libraries.
Disclosures: The authors reported that they had no conflicts of interest.
ICU care improves survival without raising costs
Compared with care on a general hospital ward, ICU care improved survival without raising costs significantly in a study of more than 1 million Medicare patients hospitalized with pneumonia, published online Sept. 22 in JAMA.
The retrospective cohort study involved older patients whose condition was considered “borderline” – not one that would clearly benefit from ICU admission but also not one for which ICU admission could clearly be ruled out. The decision of whether to admit these study participants to a general ward or an ICU was deemed discretionary. “Contrary to [our] prespecified hypothesis, [our] findings suggest that ICU admission for borderline patients … is associated with reduced mortality without a considerable increase in costs,” said Dr. Thomas S. Valley of the division of pulmonary and critical care medicine, University of Michigan, Ann Arbor, and his associates.
The investigators analyzed data from the American Hospital Association’s annual surveys and the Healthcare Cost Reporting Information Systems regarding 1,327,370 Medicare patients admitted to 2,988 hospitals across the country during a recent 2-year period. A total of 328,404 patients (29.5% of the study population) were admitted to ICUs and the remainder to general hospital wards.
After the data were adjusted to account for numerous patient, disease, and hospital variables, ICU admission was associated with significantly lower 30-day mortality (14.8%), compared with general ward admission (20.5%) – an absolute reduction of 5.7%. Yet the differences between the two groups were nonsignificant regarding payments by Medicare ($9,918 for ICU vs. $11,238 for general ward care) and hospital costs ($14,162 for ICU vs $11,320 for general ward care).
These findings were consistent across numerous sensitivity analyses, including some that compared urban against rural hospitals, white against nonwhite patients, small against large ICUs, and severely ill against less severely ill patients, Dr. Valley and his associates said (JAMA. 2015 Sep 22;314[12]:1272-79. doi: 10.1001.jama.2015.11068).
There are several reasons why ICU care might be beneficial for “borderline” patients with pneumonia. Greater attention from nurses and other clinicians could allow for more timely recognition of decompensation; more aggressive care is more likely to head off the development of sepsis; better adherence to guideline-based treatment is known to improve mortality; and a greater likelihood of being managed by a pulmonary or critical care specialist with greater expertise in pneumonia care should improve outcomes, the researchers noted.
Their study findings have important implications for health care reform. “In order to contain U.S. health care costs, it has been suggested that reducing critical care bed supply would result in more efficient admission decisions and cost savings with minimal mortality decrements.” This “presumes that ICU admission for discretionary patients provides minimal benefit but substantially increases costs.” The results of this study clearly refute that assumption, Dr. Valley and his associates said.
This study provides important empirical evidence that ICU admission can benefit “low-risk” patients. It demonstrates that the value of intensive care extends beyond mere life support for patients with an acutely failing organ and instead includes all the organizational and human resources that comprise an ICU.
It would be tempting to use these results to justify more liberal ICU admission, but that would be untenable in this era of constrained health care resources. Rather than increasing ICU use, we should make general wards function more like ICUs. The task at hand is to study why intensive care saves lives, then use that information to make hospital care safe and effective for all patients, regardless of where in the hospital they are cared for.
Dr. Ian J. Barbash is in the division of pulmonary, allergy, and critical care medicine at the University of Pittsburgh. Dr. Jeremy M. Kahn is in the department of health policy and management at the university’s Graduate School of Public Health. Both Dr. Barbash and Dr. Kahn are also at the university’s Clinical Research, Investigation, and Systems Modeling of Acute Illness Center. Both authors reported having no relevant financial disclosures. They made these remarks in an editorial accompanying Dr. Valley’s report (JAMA. 2015;314:1240-41. doi: 10.1001/jama.2015.11171).
This study provides important empirical evidence that ICU admission can benefit “low-risk” patients. It demonstrates that the value of intensive care extends beyond mere life support for patients with an acutely failing organ and instead includes all the organizational and human resources that comprise an ICU.
It would be tempting to use these results to justify more liberal ICU admission, but that would be untenable in this era of constrained health care resources. Rather than increasing ICU use, we should make general wards function more like ICUs. The task at hand is to study why intensive care saves lives, then use that information to make hospital care safe and effective for all patients, regardless of where in the hospital they are cared for.
Dr. Ian J. Barbash is in the division of pulmonary, allergy, and critical care medicine at the University of Pittsburgh. Dr. Jeremy M. Kahn is in the department of health policy and management at the university’s Graduate School of Public Health. Both Dr. Barbash and Dr. Kahn are also at the university’s Clinical Research, Investigation, and Systems Modeling of Acute Illness Center. Both authors reported having no relevant financial disclosures. They made these remarks in an editorial accompanying Dr. Valley’s report (JAMA. 2015;314:1240-41. doi: 10.1001/jama.2015.11171).
This study provides important empirical evidence that ICU admission can benefit “low-risk” patients. It demonstrates that the value of intensive care extends beyond mere life support for patients with an acutely failing organ and instead includes all the organizational and human resources that comprise an ICU.
It would be tempting to use these results to justify more liberal ICU admission, but that would be untenable in this era of constrained health care resources. Rather than increasing ICU use, we should make general wards function more like ICUs. The task at hand is to study why intensive care saves lives, then use that information to make hospital care safe and effective for all patients, regardless of where in the hospital they are cared for.
Dr. Ian J. Barbash is in the division of pulmonary, allergy, and critical care medicine at the University of Pittsburgh. Dr. Jeremy M. Kahn is in the department of health policy and management at the university’s Graduate School of Public Health. Both Dr. Barbash and Dr. Kahn are also at the university’s Clinical Research, Investigation, and Systems Modeling of Acute Illness Center. Both authors reported having no relevant financial disclosures. They made these remarks in an editorial accompanying Dr. Valley’s report (JAMA. 2015;314:1240-41. doi: 10.1001/jama.2015.11171).
Compared with care on a general hospital ward, ICU care improved survival without raising costs significantly in a study of more than 1 million Medicare patients hospitalized with pneumonia, published online Sept. 22 in JAMA.
The retrospective cohort study involved older patients whose condition was considered “borderline” – not one that would clearly benefit from ICU admission but also not one for which ICU admission could clearly be ruled out. The decision of whether to admit these study participants to a general ward or an ICU was deemed discretionary. “Contrary to [our] prespecified hypothesis, [our] findings suggest that ICU admission for borderline patients … is associated with reduced mortality without a considerable increase in costs,” said Dr. Thomas S. Valley of the division of pulmonary and critical care medicine, University of Michigan, Ann Arbor, and his associates.
The investigators analyzed data from the American Hospital Association’s annual surveys and the Healthcare Cost Reporting Information Systems regarding 1,327,370 Medicare patients admitted to 2,988 hospitals across the country during a recent 2-year period. A total of 328,404 patients (29.5% of the study population) were admitted to ICUs and the remainder to general hospital wards.
After the data were adjusted to account for numerous patient, disease, and hospital variables, ICU admission was associated with significantly lower 30-day mortality (14.8%), compared with general ward admission (20.5%) – an absolute reduction of 5.7%. Yet the differences between the two groups were nonsignificant regarding payments by Medicare ($9,918 for ICU vs. $11,238 for general ward care) and hospital costs ($14,162 for ICU vs $11,320 for general ward care).
These findings were consistent across numerous sensitivity analyses, including some that compared urban against rural hospitals, white against nonwhite patients, small against large ICUs, and severely ill against less severely ill patients, Dr. Valley and his associates said (JAMA. 2015 Sep 22;314[12]:1272-79. doi: 10.1001.jama.2015.11068).
There are several reasons why ICU care might be beneficial for “borderline” patients with pneumonia. Greater attention from nurses and other clinicians could allow for more timely recognition of decompensation; more aggressive care is more likely to head off the development of sepsis; better adherence to guideline-based treatment is known to improve mortality; and a greater likelihood of being managed by a pulmonary or critical care specialist with greater expertise in pneumonia care should improve outcomes, the researchers noted.
Their study findings have important implications for health care reform. “In order to contain U.S. health care costs, it has been suggested that reducing critical care bed supply would result in more efficient admission decisions and cost savings with minimal mortality decrements.” This “presumes that ICU admission for discretionary patients provides minimal benefit but substantially increases costs.” The results of this study clearly refute that assumption, Dr. Valley and his associates said.
Compared with care on a general hospital ward, ICU care improved survival without raising costs significantly in a study of more than 1 million Medicare patients hospitalized with pneumonia, published online Sept. 22 in JAMA.
The retrospective cohort study involved older patients whose condition was considered “borderline” – not one that would clearly benefit from ICU admission but also not one for which ICU admission could clearly be ruled out. The decision of whether to admit these study participants to a general ward or an ICU was deemed discretionary. “Contrary to [our] prespecified hypothesis, [our] findings suggest that ICU admission for borderline patients … is associated with reduced mortality without a considerable increase in costs,” said Dr. Thomas S. Valley of the division of pulmonary and critical care medicine, University of Michigan, Ann Arbor, and his associates.
The investigators analyzed data from the American Hospital Association’s annual surveys and the Healthcare Cost Reporting Information Systems regarding 1,327,370 Medicare patients admitted to 2,988 hospitals across the country during a recent 2-year period. A total of 328,404 patients (29.5% of the study population) were admitted to ICUs and the remainder to general hospital wards.
After the data were adjusted to account for numerous patient, disease, and hospital variables, ICU admission was associated with significantly lower 30-day mortality (14.8%), compared with general ward admission (20.5%) – an absolute reduction of 5.7%. Yet the differences between the two groups were nonsignificant regarding payments by Medicare ($9,918 for ICU vs. $11,238 for general ward care) and hospital costs ($14,162 for ICU vs $11,320 for general ward care).
These findings were consistent across numerous sensitivity analyses, including some that compared urban against rural hospitals, white against nonwhite patients, small against large ICUs, and severely ill against less severely ill patients, Dr. Valley and his associates said (JAMA. 2015 Sep 22;314[12]:1272-79. doi: 10.1001.jama.2015.11068).
There are several reasons why ICU care might be beneficial for “borderline” patients with pneumonia. Greater attention from nurses and other clinicians could allow for more timely recognition of decompensation; more aggressive care is more likely to head off the development of sepsis; better adherence to guideline-based treatment is known to improve mortality; and a greater likelihood of being managed by a pulmonary or critical care specialist with greater expertise in pneumonia care should improve outcomes, the researchers noted.
Their study findings have important implications for health care reform. “In order to contain U.S. health care costs, it has been suggested that reducing critical care bed supply would result in more efficient admission decisions and cost savings with minimal mortality decrements.” This “presumes that ICU admission for discretionary patients provides minimal benefit but substantially increases costs.” The results of this study clearly refute that assumption, Dr. Valley and his associates said.
FROM JAMA
Key clinical point: ICU care improved survival without raising costs in older patients with pneumonia, compared with care on a general hospital ward.
Major finding: ICU admission was associated with significantly lower 30-day mortality (14.8%), compared with general ward admission (20.5%).
Data source: A retrospective cohort study involving 1,112,394 Medicare patients treated for pneumonia at 2,988 U.S. hospitals during a 2-year period.
Disclosures: This study was supported by the National Institutes of Health, the Department of Veterans Affairs Health Services Research and Development Service, and the Agency for Healthcare Research and Quality. Dr. Valley and his associates reported having no relevant financial disclosures.
Point/Counterpoint: Does surgery play a role in N2 disease treatment following induction therapy?
POINT: Surgery has its uses for some
BY DR. STEPHEN G. SWISHER
When talking about the role of surgery after induction therapy with persistent N2 disease, one must acknowledge that this is such a heterogeneous disease. You can have single-station N2; resectable, bulky multistation N2; and so on. Then there’s unresectable stage IIIA, but let’s focus mainly on resectable stage IIIA disease.
I can’t tell you how many audiences I’ve faced that absolutely believe the myth that surgery plays no role in Stage IIIA non–small cell lung cancer based on data from stage IIIA disease patients randomized to chemoradiation followed by surgery. The problem with these study results is the high mortality in the pneumonectomy subset. There’s no difference in the overall survival of the two groups, but that doesn’t mean that everyone in that group wouldn’t benefit from surgery.
The curve showed that pneumonectomy did not benefit after chemoradiation in a non–high-volume center. You can see a steep drop in the mortality early on, but it catches up again at the end. If you look at the overall 5-year survival rate, even in the pneumonectomy subset, you’re looking at 22% vs. 24%.
But in the lobectomy set, you see something completely different. You’ve got a doubling of survivors and no mortality early on, and a doubling of 5-year survival from 18% to 36%.
And yet, people continue saying that there’s no role for surgery. Well, I think there is a role for surgery, and there are subsets of N2 for which surgery can be particularly beneficial. We have to move more toward what the medical oncologists do, which is personalize therapy and look at subsets of N2 disease so that we know which patients we can benefit and which ones we can’t.
Moving on to the second myth: Surgery plays no role in N2 residual disease after induction therapy. This myth is based on the results of a couple of prospective studies in the 1980s and ’90s that showed residual N2 disease after chemo- and radiotherapy leads to survival of 16-35 months in most cases. I’d say that those results are true, but it’s not to say there aren’t subsets within these populations that benefited. With preoperative chemo and radiation, it’s basically the same thing – poor prognoses in patients with N2 or N3 disease, so the standard becomes never to operate on these individuals.
A European study prospectively took 30 patients and treated them with induction chemotherapy. They saw a 5-year survival rate of 62% if a patient downgrades to lymph node–negative disease and the positron-emission tomographic (PET) findings were good. But they also saw a subset with a small amount of disease within the lymph nodes at the N2 stage and a poor response on PET; Their 5-year survival rate was around 19%. So I’d argue that PET response and the number of lymph nodes involved are the key criteria, and you shouldn’t routinely deny surgery to these patients.
Our experience at MD Anderson Cancer Center over the last 10 years has been to treat N2 and N3 admissions, with surgery, followed by postoperative radiation of 50 Gy. We’re able to achieve very-low morbidity with this regimen, and no mortality after 30 and 90 days. Just to show the heterogeneity: Single-station, microscopic N2 disease should really be resected.
You just can’t lump together everyone with residual N2 after induction therapy, since PET-CTs and most other diagnostic procedures have high false-negative rates. And like I’ve said, it doesn’t matter because N2 disease is really a subset disease. Microscopic N2 disease behaves in a completely different way than does macroscopic, multiple-level N2 disease. And even more important is how the patient’s primary tumor responds to the chemotherapy or chemoradiation; that will tell you how well they’re going to do even if they have a small amount of residual disease in the lymph nodes.
Dr. Swisher is at the University of Texas MD Anderson Cancer Center in Houston. He disclosed that he is a consultant/advisory board member for GlaxoSmithKline.
COUNTERPOINT: Surgery seems to have little value, adds risk.
BY DR. SCOTT J. SWANSON
Dr. Swisher and I probably agree more than we disagree, but I’m going to start by saying that N2 disease is bad, and most of these populations are heterogeneous. But if you feel that a curve toward the bottom of a graph is good, then you should think twice. Anywhere from a 15%-30% survival rate is not great and shows that we have a long way to go. The overall impression among oncologists in several countries is that it’s not really clear whether surgery adds value. Even in very good centers like MD Anderson where there is minimal risk, surgery inherently still involves some risk.
So then, what do we do with persistent N2 disease? I’d say that most of the time, it should be treated with chemotherapy or chemotherapy and radiation. In some cases, N2 disease can be treated with a creative, mutation-driven immunotherapy approach. Most of the time though, surgery is just not a good idea.
Interestingly, about one-third of lung cancer patients present with stage IIIA disease, so it’s important that we as a medical community sort out these treatment options. I think we’re all in agreement that single-station, microscopic, PET-negative/CT-negative disease is not the same as extranodal or multistation PET-positive disease, so we’ve got to begin to substratify N2 disease.
In an intergroup trial of about 200 patients per arm published in the Lancet, patients went to surgery if they didn’t regress after evaluation. The progression-free survival rate did seem to favor surgery; and, if you look at the lobectomy subset, the results are certainly strong. But again, we’re dealing with curves that are pretty low. The pneumonectomy subset drops off and then starts to catch up, but clearly pneumonectomy was a problem in this multicenter trial. The most important graph to this debate shows subjects that persistently had nodal disease had very poor survival. It’s hard to argue for surgery when results show that only about 24% of them are going to be alive down the road. If oncologists across most of the United States say they believe that surgery isn’t a good idea, we’re not going to use this graph to change their minds; we need to change our way of thinking.
So the conclusion to take away from that presentation is that N0 status at surgery significantly predicts greater 5-year survival. So, conversely, surgery is not helpful for the patient with node-positive disease. Surgical resection should only be considered if lobectomy is the operation in question. Pneumonectomy carries risk, and surgery has no beneficial value, compared with chemotherapy unless the patient has been downstaged.
Our experience at Brigham and Women’s Hospital in Boston is similar to Dr. Swisher’s at MD Anderson. During the first 8 years of our thoracic division, we looked at 103 patients who had surgery after induction therapy for N2 disease. The induction plan in those patients was chemotherapy only for 75, radiation only for 18, and chemoradiation for 10. Almost 40% of patients had pneumonectomies, and the rest had lobectomies.
Mortality was 3.9%, major morbidity was 7%, and about 30% were downstaged. Those 5-year survival rates were about 36%. Persistent nodal disease, either N1 or N2, was seen in about 75%, and most of them were N2; the 5-year survival rate there was about 9% with a median of about 15.9 months. Beauty is in the eye of the beholder, so you may look at that and say that 15.9 months isn’t bad, but there’s still a huge subgroup that’s node positive, so here I’d say that pushing surgery is not the best strategy. We also found in this group that adenocarcinomas were much harder to clear.
We’re on very-safe ground to push surgery in the node-negative group, but you’ve got to be careful in the node-positive group.
Survival is relatively limited in N2 disease in general. Surgery may be of value if you downstage the patient, if you’re doing a lobectomy or if you see squamous cell carcinoma. Going forward, we really ought to focus our attention on identifying responders more reliably and improving downstaging – with different or individualized chemotherapy, or perhaps even immunologic therapy.
For the present, we can talk about radiation dosing. High-dose radiation is clearly a viable option for some patients. In addition, we can improve identification of N2 subgroups. Not all N2 disease is the same, so it should not be treated the same across the board.
Dr. Swanson is at Brigham and Women’s Hospital in Boston. He disclosed that he is a consultant/advisory board member for Covidien and Ethicon Endo-Surgery.
This article grew out of the debate by Dr. Swisher and Dr. Swanson at the annual meeting of the Society of Thoracic Surgeons.
POINT: Surgery has its uses for some
BY DR. STEPHEN G. SWISHER
When talking about the role of surgery after induction therapy with persistent N2 disease, one must acknowledge that this is such a heterogeneous disease. You can have single-station N2; resectable, bulky multistation N2; and so on. Then there’s unresectable stage IIIA, but let’s focus mainly on resectable stage IIIA disease.
I can’t tell you how many audiences I’ve faced that absolutely believe the myth that surgery plays no role in Stage IIIA non–small cell lung cancer based on data from stage IIIA disease patients randomized to chemoradiation followed by surgery. The problem with these study results is the high mortality in the pneumonectomy subset. There’s no difference in the overall survival of the two groups, but that doesn’t mean that everyone in that group wouldn’t benefit from surgery.
The curve showed that pneumonectomy did not benefit after chemoradiation in a non–high-volume center. You can see a steep drop in the mortality early on, but it catches up again at the end. If you look at the overall 5-year survival rate, even in the pneumonectomy subset, you’re looking at 22% vs. 24%.
But in the lobectomy set, you see something completely different. You’ve got a doubling of survivors and no mortality early on, and a doubling of 5-year survival from 18% to 36%.
And yet, people continue saying that there’s no role for surgery. Well, I think there is a role for surgery, and there are subsets of N2 for which surgery can be particularly beneficial. We have to move more toward what the medical oncologists do, which is personalize therapy and look at subsets of N2 disease so that we know which patients we can benefit and which ones we can’t.
Moving on to the second myth: Surgery plays no role in N2 residual disease after induction therapy. This myth is based on the results of a couple of prospective studies in the 1980s and ’90s that showed residual N2 disease after chemo- and radiotherapy leads to survival of 16-35 months in most cases. I’d say that those results are true, but it’s not to say there aren’t subsets within these populations that benefited. With preoperative chemo and radiation, it’s basically the same thing – poor prognoses in patients with N2 or N3 disease, so the standard becomes never to operate on these individuals.
A European study prospectively took 30 patients and treated them with induction chemotherapy. They saw a 5-year survival rate of 62% if a patient downgrades to lymph node–negative disease and the positron-emission tomographic (PET) findings were good. But they also saw a subset with a small amount of disease within the lymph nodes at the N2 stage and a poor response on PET; Their 5-year survival rate was around 19%. So I’d argue that PET response and the number of lymph nodes involved are the key criteria, and you shouldn’t routinely deny surgery to these patients.
Our experience at MD Anderson Cancer Center over the last 10 years has been to treat N2 and N3 admissions, with surgery, followed by postoperative radiation of 50 Gy. We’re able to achieve very-low morbidity with this regimen, and no mortality after 30 and 90 days. Just to show the heterogeneity: Single-station, microscopic N2 disease should really be resected.
You just can’t lump together everyone with residual N2 after induction therapy, since PET-CTs and most other diagnostic procedures have high false-negative rates. And like I’ve said, it doesn’t matter because N2 disease is really a subset disease. Microscopic N2 disease behaves in a completely different way than does macroscopic, multiple-level N2 disease. And even more important is how the patient’s primary tumor responds to the chemotherapy or chemoradiation; that will tell you how well they’re going to do even if they have a small amount of residual disease in the lymph nodes.
Dr. Swisher is at the University of Texas MD Anderson Cancer Center in Houston. He disclosed that he is a consultant/advisory board member for GlaxoSmithKline.
COUNTERPOINT: Surgery seems to have little value, adds risk.
BY DR. SCOTT J. SWANSON
Dr. Swisher and I probably agree more than we disagree, but I’m going to start by saying that N2 disease is bad, and most of these populations are heterogeneous. But if you feel that a curve toward the bottom of a graph is good, then you should think twice. Anywhere from a 15%-30% survival rate is not great and shows that we have a long way to go. The overall impression among oncologists in several countries is that it’s not really clear whether surgery adds value. Even in very good centers like MD Anderson where there is minimal risk, surgery inherently still involves some risk.
So then, what do we do with persistent N2 disease? I’d say that most of the time, it should be treated with chemotherapy or chemotherapy and radiation. In some cases, N2 disease can be treated with a creative, mutation-driven immunotherapy approach. Most of the time though, surgery is just not a good idea.
Interestingly, about one-third of lung cancer patients present with stage IIIA disease, so it’s important that we as a medical community sort out these treatment options. I think we’re all in agreement that single-station, microscopic, PET-negative/CT-negative disease is not the same as extranodal or multistation PET-positive disease, so we’ve got to begin to substratify N2 disease.
In an intergroup trial of about 200 patients per arm published in the Lancet, patients went to surgery if they didn’t regress after evaluation. The progression-free survival rate did seem to favor surgery; and, if you look at the lobectomy subset, the results are certainly strong. But again, we’re dealing with curves that are pretty low. The pneumonectomy subset drops off and then starts to catch up, but clearly pneumonectomy was a problem in this multicenter trial. The most important graph to this debate shows subjects that persistently had nodal disease had very poor survival. It’s hard to argue for surgery when results show that only about 24% of them are going to be alive down the road. If oncologists across most of the United States say they believe that surgery isn’t a good idea, we’re not going to use this graph to change their minds; we need to change our way of thinking.
So the conclusion to take away from that presentation is that N0 status at surgery significantly predicts greater 5-year survival. So, conversely, surgery is not helpful for the patient with node-positive disease. Surgical resection should only be considered if lobectomy is the operation in question. Pneumonectomy carries risk, and surgery has no beneficial value, compared with chemotherapy unless the patient has been downstaged.
Our experience at Brigham and Women’s Hospital in Boston is similar to Dr. Swisher’s at MD Anderson. During the first 8 years of our thoracic division, we looked at 103 patients who had surgery after induction therapy for N2 disease. The induction plan in those patients was chemotherapy only for 75, radiation only for 18, and chemoradiation for 10. Almost 40% of patients had pneumonectomies, and the rest had lobectomies.
Mortality was 3.9%, major morbidity was 7%, and about 30% were downstaged. Those 5-year survival rates were about 36%. Persistent nodal disease, either N1 or N2, was seen in about 75%, and most of them were N2; the 5-year survival rate there was about 9% with a median of about 15.9 months. Beauty is in the eye of the beholder, so you may look at that and say that 15.9 months isn’t bad, but there’s still a huge subgroup that’s node positive, so here I’d say that pushing surgery is not the best strategy. We also found in this group that adenocarcinomas were much harder to clear.
We’re on very-safe ground to push surgery in the node-negative group, but you’ve got to be careful in the node-positive group.
Survival is relatively limited in N2 disease in general. Surgery may be of value if you downstage the patient, if you’re doing a lobectomy or if you see squamous cell carcinoma. Going forward, we really ought to focus our attention on identifying responders more reliably and improving downstaging – with different or individualized chemotherapy, or perhaps even immunologic therapy.
For the present, we can talk about radiation dosing. High-dose radiation is clearly a viable option for some patients. In addition, we can improve identification of N2 subgroups. Not all N2 disease is the same, so it should not be treated the same across the board.
Dr. Swanson is at Brigham and Women’s Hospital in Boston. He disclosed that he is a consultant/advisory board member for Covidien and Ethicon Endo-Surgery.
This article grew out of the debate by Dr. Swisher and Dr. Swanson at the annual meeting of the Society of Thoracic Surgeons.
POINT: Surgery has its uses for some
BY DR. STEPHEN G. SWISHER
When talking about the role of surgery after induction therapy with persistent N2 disease, one must acknowledge that this is such a heterogeneous disease. You can have single-station N2; resectable, bulky multistation N2; and so on. Then there’s unresectable stage IIIA, but let’s focus mainly on resectable stage IIIA disease.
I can’t tell you how many audiences I’ve faced that absolutely believe the myth that surgery plays no role in Stage IIIA non–small cell lung cancer based on data from stage IIIA disease patients randomized to chemoradiation followed by surgery. The problem with these study results is the high mortality in the pneumonectomy subset. There’s no difference in the overall survival of the two groups, but that doesn’t mean that everyone in that group wouldn’t benefit from surgery.
The curve showed that pneumonectomy did not benefit after chemoradiation in a non–high-volume center. You can see a steep drop in the mortality early on, but it catches up again at the end. If you look at the overall 5-year survival rate, even in the pneumonectomy subset, you’re looking at 22% vs. 24%.
But in the lobectomy set, you see something completely different. You’ve got a doubling of survivors and no mortality early on, and a doubling of 5-year survival from 18% to 36%.
And yet, people continue saying that there’s no role for surgery. Well, I think there is a role for surgery, and there are subsets of N2 for which surgery can be particularly beneficial. We have to move more toward what the medical oncologists do, which is personalize therapy and look at subsets of N2 disease so that we know which patients we can benefit and which ones we can’t.
Moving on to the second myth: Surgery plays no role in N2 residual disease after induction therapy. This myth is based on the results of a couple of prospective studies in the 1980s and ’90s that showed residual N2 disease after chemo- and radiotherapy leads to survival of 16-35 months in most cases. I’d say that those results are true, but it’s not to say there aren’t subsets within these populations that benefited. With preoperative chemo and radiation, it’s basically the same thing – poor prognoses in patients with N2 or N3 disease, so the standard becomes never to operate on these individuals.
A European study prospectively took 30 patients and treated them with induction chemotherapy. They saw a 5-year survival rate of 62% if a patient downgrades to lymph node–negative disease and the positron-emission tomographic (PET) findings were good. But they also saw a subset with a small amount of disease within the lymph nodes at the N2 stage and a poor response on PET; Their 5-year survival rate was around 19%. So I’d argue that PET response and the number of lymph nodes involved are the key criteria, and you shouldn’t routinely deny surgery to these patients.
Our experience at MD Anderson Cancer Center over the last 10 years has been to treat N2 and N3 admissions, with surgery, followed by postoperative radiation of 50 Gy. We’re able to achieve very-low morbidity with this regimen, and no mortality after 30 and 90 days. Just to show the heterogeneity: Single-station, microscopic N2 disease should really be resected.
You just can’t lump together everyone with residual N2 after induction therapy, since PET-CTs and most other diagnostic procedures have high false-negative rates. And like I’ve said, it doesn’t matter because N2 disease is really a subset disease. Microscopic N2 disease behaves in a completely different way than does macroscopic, multiple-level N2 disease. And even more important is how the patient’s primary tumor responds to the chemotherapy or chemoradiation; that will tell you how well they’re going to do even if they have a small amount of residual disease in the lymph nodes.
Dr. Swisher is at the University of Texas MD Anderson Cancer Center in Houston. He disclosed that he is a consultant/advisory board member for GlaxoSmithKline.
COUNTERPOINT: Surgery seems to have little value, adds risk.
BY DR. SCOTT J. SWANSON
Dr. Swisher and I probably agree more than we disagree, but I’m going to start by saying that N2 disease is bad, and most of these populations are heterogeneous. But if you feel that a curve toward the bottom of a graph is good, then you should think twice. Anywhere from a 15%-30% survival rate is not great and shows that we have a long way to go. The overall impression among oncologists in several countries is that it’s not really clear whether surgery adds value. Even in very good centers like MD Anderson where there is minimal risk, surgery inherently still involves some risk.
So then, what do we do with persistent N2 disease? I’d say that most of the time, it should be treated with chemotherapy or chemotherapy and radiation. In some cases, N2 disease can be treated with a creative, mutation-driven immunotherapy approach. Most of the time though, surgery is just not a good idea.
Interestingly, about one-third of lung cancer patients present with stage IIIA disease, so it’s important that we as a medical community sort out these treatment options. I think we’re all in agreement that single-station, microscopic, PET-negative/CT-negative disease is not the same as extranodal or multistation PET-positive disease, so we’ve got to begin to substratify N2 disease.
In an intergroup trial of about 200 patients per arm published in the Lancet, patients went to surgery if they didn’t regress after evaluation. The progression-free survival rate did seem to favor surgery; and, if you look at the lobectomy subset, the results are certainly strong. But again, we’re dealing with curves that are pretty low. The pneumonectomy subset drops off and then starts to catch up, but clearly pneumonectomy was a problem in this multicenter trial. The most important graph to this debate shows subjects that persistently had nodal disease had very poor survival. It’s hard to argue for surgery when results show that only about 24% of them are going to be alive down the road. If oncologists across most of the United States say they believe that surgery isn’t a good idea, we’re not going to use this graph to change their minds; we need to change our way of thinking.
So the conclusion to take away from that presentation is that N0 status at surgery significantly predicts greater 5-year survival. So, conversely, surgery is not helpful for the patient with node-positive disease. Surgical resection should only be considered if lobectomy is the operation in question. Pneumonectomy carries risk, and surgery has no beneficial value, compared with chemotherapy unless the patient has been downstaged.
Our experience at Brigham and Women’s Hospital in Boston is similar to Dr. Swisher’s at MD Anderson. During the first 8 years of our thoracic division, we looked at 103 patients who had surgery after induction therapy for N2 disease. The induction plan in those patients was chemotherapy only for 75, radiation only for 18, and chemoradiation for 10. Almost 40% of patients had pneumonectomies, and the rest had lobectomies.
Mortality was 3.9%, major morbidity was 7%, and about 30% were downstaged. Those 5-year survival rates were about 36%. Persistent nodal disease, either N1 or N2, was seen in about 75%, and most of them were N2; the 5-year survival rate there was about 9% with a median of about 15.9 months. Beauty is in the eye of the beholder, so you may look at that and say that 15.9 months isn’t bad, but there’s still a huge subgroup that’s node positive, so here I’d say that pushing surgery is not the best strategy. We also found in this group that adenocarcinomas were much harder to clear.
We’re on very-safe ground to push surgery in the node-negative group, but you’ve got to be careful in the node-positive group.
Survival is relatively limited in N2 disease in general. Surgery may be of value if you downstage the patient, if you’re doing a lobectomy or if you see squamous cell carcinoma. Going forward, we really ought to focus our attention on identifying responders more reliably and improving downstaging – with different or individualized chemotherapy, or perhaps even immunologic therapy.
For the present, we can talk about radiation dosing. High-dose radiation is clearly a viable option for some patients. In addition, we can improve identification of N2 subgroups. Not all N2 disease is the same, so it should not be treated the same across the board.
Dr. Swanson is at Brigham and Women’s Hospital in Boston. He disclosed that he is a consultant/advisory board member for Covidien and Ethicon Endo-Surgery.
This article grew out of the debate by Dr. Swisher and Dr. Swanson at the annual meeting of the Society of Thoracic Surgeons.
Asthma exacerbations seen in 32% of patients within 2 years of drug step-down
Nearly a third of patients have asthma exacerbation in the 2 years after medication step-down, according to a new study published in the September issue of Chest.
With the goal of using the least amount of medication to control asthma, guidelines recommend considering medication step-down after 3 months of stabilized asthma. However, there is limited evidence backing these recommendations, especially when it comes to understanding the long-term outcomes after asthma medication step-down.
Dr. Matthew A. Rank of the division of allergy, asthma, and clinical immunology at the Mayo Clinic in Scottsdale, Ariz., and colleagues analyzed the long-term outcomes of patients after asthma medication step-down.
The investigators conducted a retrospective claims-based analysis using data obtained from the Optum Labs Data Warehouse which contains information from more than 100 million deidentified patients with Medicare Advantage or commercial insurance plans (Chest. 2015;148[3]:630-39). .
Data was extracted on patients who had an asthma diagnosis code between 2000 and 2012 with continuous medical and pharmacy coverage for 3 or more years during the study period and with a history of medication step-down.
Investigators defined a medication step-down as greater or equal to 50% decrease in asthma controller medication between evaluations. Stability was defined as not having an asthma exacerbation requiring care in the hospital or ED, or systemic corticosteroids and claiming fewer than two rescue inhalers prescriptions in the 4 month study period.
The study cohort was divided into four asthma stability groups: 0-3 months, 4-7 months, 8-11 months, and greater to or equal to 12 months of stability.
Of the 26,292 individuals included in the study, 32% developed an asthma exacerbation during the 2 years after medication step-down. There was a strong association between the risk of developing an asthma exacerbation during the 2-year study period and the length of asthma stability prior to medication step-down. For instance, 44% in participants with less than 4 months of stability, 34% with 4-7 months of stability, 30% with 8-11 months of stability, and 21% with more than 12 months of stability (P less than .001).
In addition, study participants who were women, were black, were younger than 19 years old, had a Charlson comorbidity index great than or equal to 1, and at least two outpatient visits for asthma were significantly associated with a shorter interval to asthma exacerbation (P less than.001 for all variables).
Finally, most study participants had a hospital or ED visit, systemic corticosteroids, two rescue inhalers in a 4-month period, or needed to return to baseline asthma controller treatment. The authors suggest that this is evidence that most of the cohort continued to have underlying asthma during the 2-year study period. Furthermore, 33% of participants with less than 4 months of stability required return to baseline treatment versus 8%, 13%, and 15% for more than 12 months of stability, 8-11 months, and 4-7 months, respectively.
Among the limitations noted by the authors: Data were from insured patients, data did not indicate if step-down involved consultation with a health care provider, and the cohort did not include patients who did not step-down as a comparison.
“Individuals and their providers can cautiously apply the data from this study to decisions about stepping down asthma medications. The novel insights from this analysis that contribute to this decision making process are consideration to the length of stability prior to step-down and the rate of asthma exacerbations in the 24 months following step-down,” the authors wrote.
The study was funded by the Mayo Foundation for Medical Education and Research. The authors report no disclosures.
Nearly a third of patients have asthma exacerbation in the 2 years after medication step-down, according to a new study published in the September issue of Chest.
With the goal of using the least amount of medication to control asthma, guidelines recommend considering medication step-down after 3 months of stabilized asthma. However, there is limited evidence backing these recommendations, especially when it comes to understanding the long-term outcomes after asthma medication step-down.
Dr. Matthew A. Rank of the division of allergy, asthma, and clinical immunology at the Mayo Clinic in Scottsdale, Ariz., and colleagues analyzed the long-term outcomes of patients after asthma medication step-down.
The investigators conducted a retrospective claims-based analysis using data obtained from the Optum Labs Data Warehouse which contains information from more than 100 million deidentified patients with Medicare Advantage or commercial insurance plans (Chest. 2015;148[3]:630-39). .
Data was extracted on patients who had an asthma diagnosis code between 2000 and 2012 with continuous medical and pharmacy coverage for 3 or more years during the study period and with a history of medication step-down.
Investigators defined a medication step-down as greater or equal to 50% decrease in asthma controller medication between evaluations. Stability was defined as not having an asthma exacerbation requiring care in the hospital or ED, or systemic corticosteroids and claiming fewer than two rescue inhalers prescriptions in the 4 month study period.
The study cohort was divided into four asthma stability groups: 0-3 months, 4-7 months, 8-11 months, and greater to or equal to 12 months of stability.
Of the 26,292 individuals included in the study, 32% developed an asthma exacerbation during the 2 years after medication step-down. There was a strong association between the risk of developing an asthma exacerbation during the 2-year study period and the length of asthma stability prior to medication step-down. For instance, 44% in participants with less than 4 months of stability, 34% with 4-7 months of stability, 30% with 8-11 months of stability, and 21% with more than 12 months of stability (P less than .001).
In addition, study participants who were women, were black, were younger than 19 years old, had a Charlson comorbidity index great than or equal to 1, and at least two outpatient visits for asthma were significantly associated with a shorter interval to asthma exacerbation (P less than.001 for all variables).
Finally, most study participants had a hospital or ED visit, systemic corticosteroids, two rescue inhalers in a 4-month period, or needed to return to baseline asthma controller treatment. The authors suggest that this is evidence that most of the cohort continued to have underlying asthma during the 2-year study period. Furthermore, 33% of participants with less than 4 months of stability required return to baseline treatment versus 8%, 13%, and 15% for more than 12 months of stability, 8-11 months, and 4-7 months, respectively.
Among the limitations noted by the authors: Data were from insured patients, data did not indicate if step-down involved consultation with a health care provider, and the cohort did not include patients who did not step-down as a comparison.
“Individuals and their providers can cautiously apply the data from this study to decisions about stepping down asthma medications. The novel insights from this analysis that contribute to this decision making process are consideration to the length of stability prior to step-down and the rate of asthma exacerbations in the 24 months following step-down,” the authors wrote.
The study was funded by the Mayo Foundation for Medical Education and Research. The authors report no disclosures.
Nearly a third of patients have asthma exacerbation in the 2 years after medication step-down, according to a new study published in the September issue of Chest.
With the goal of using the least amount of medication to control asthma, guidelines recommend considering medication step-down after 3 months of stabilized asthma. However, there is limited evidence backing these recommendations, especially when it comes to understanding the long-term outcomes after asthma medication step-down.
Dr. Matthew A. Rank of the division of allergy, asthma, and clinical immunology at the Mayo Clinic in Scottsdale, Ariz., and colleagues analyzed the long-term outcomes of patients after asthma medication step-down.
The investigators conducted a retrospective claims-based analysis using data obtained from the Optum Labs Data Warehouse which contains information from more than 100 million deidentified patients with Medicare Advantage or commercial insurance plans (Chest. 2015;148[3]:630-39). .
Data was extracted on patients who had an asthma diagnosis code between 2000 and 2012 with continuous medical and pharmacy coverage for 3 or more years during the study period and with a history of medication step-down.
Investigators defined a medication step-down as greater or equal to 50% decrease in asthma controller medication between evaluations. Stability was defined as not having an asthma exacerbation requiring care in the hospital or ED, or systemic corticosteroids and claiming fewer than two rescue inhalers prescriptions in the 4 month study period.
The study cohort was divided into four asthma stability groups: 0-3 months, 4-7 months, 8-11 months, and greater to or equal to 12 months of stability.
Of the 26,292 individuals included in the study, 32% developed an asthma exacerbation during the 2 years after medication step-down. There was a strong association between the risk of developing an asthma exacerbation during the 2-year study period and the length of asthma stability prior to medication step-down. For instance, 44% in participants with less than 4 months of stability, 34% with 4-7 months of stability, 30% with 8-11 months of stability, and 21% with more than 12 months of stability (P less than .001).
In addition, study participants who were women, were black, were younger than 19 years old, had a Charlson comorbidity index great than or equal to 1, and at least two outpatient visits for asthma were significantly associated with a shorter interval to asthma exacerbation (P less than.001 for all variables).
Finally, most study participants had a hospital or ED visit, systemic corticosteroids, two rescue inhalers in a 4-month period, or needed to return to baseline asthma controller treatment. The authors suggest that this is evidence that most of the cohort continued to have underlying asthma during the 2-year study period. Furthermore, 33% of participants with less than 4 months of stability required return to baseline treatment versus 8%, 13%, and 15% for more than 12 months of stability, 8-11 months, and 4-7 months, respectively.
Among the limitations noted by the authors: Data were from insured patients, data did not indicate if step-down involved consultation with a health care provider, and the cohort did not include patients who did not step-down as a comparison.
“Individuals and their providers can cautiously apply the data from this study to decisions about stepping down asthma medications. The novel insights from this analysis that contribute to this decision making process are consideration to the length of stability prior to step-down and the rate of asthma exacerbations in the 24 months following step-down,” the authors wrote.
The study was funded by the Mayo Foundation for Medical Education and Research. The authors report no disclosures.
FROM CHEST
Key clinical point: Asthma exacerbation occurred in 32% of patients in the 2 years after medication step-down.
Major finding: Of the 26,292 individuals, 32% developed an asthma exacerbation during the 2 years after medication step-down. There was a strong association between the risk of developing an asthma exacerbation and the length of asthma stability prior to medication step-down.
Data source: A retrospective claims-based analysis using data obtained from the Optum Labs Data Warehouse.
Disclosures: The study was funded by the Mayo Foundation for Medical Education and Research. The authors report no disclosures.
View Webcast of Mitral Conclave 2015
If you were unable to attend Mitral Conclave 2015, catch up on all the state-of-the art information you missed.
The full event webcast is now available for purchase.
Experience it all!
Purchase the full webcast* today
View Interviews with AATS & Thought Leaders
*CME credits are only available to attendees at Mitral Conclave in New York City.
If you were unable to attend Mitral Conclave 2015, catch up on all the state-of-the art information you missed.
The full event webcast is now available for purchase.
Experience it all!
Purchase the full webcast* today
View Interviews with AATS & Thought Leaders
*CME credits are only available to attendees at Mitral Conclave in New York City.
If you were unable to attend Mitral Conclave 2015, catch up on all the state-of-the art information you missed.
The full event webcast is now available for purchase.
Experience it all!
Purchase the full webcast* today
View Interviews with AATS & Thought Leaders
*CME credits are only available to attendees at Mitral Conclave in New York City.
Register for Focus on Thoracic Surgery: Technical Challenges and Complications
October 23-24, 2015
Seaport Hotel & World Trade Center
Boston, MA
Overview
Complications remain one of the major challenges in treating patients. The 2015 Focus on Thoracic Surgery meeting will concentrate on the intraoperative challenges and management of complications facing thoracic surgeons in treating lung and esophageal cancers. This year’s course is particularly aimed at operating surgeons who are active and seek to learn innovative solutions to clinical problems during surgery.
This course will display new advanced diagnosis and prompt management approaches, allowing attendees to understand and readily translate them into daily practice. This two-day conference will have a faculty of experts in the field who are leaders in the application of new technologies in surgical management. It will combine informational sessions, panel discussions and complex case presentations.
Focus on Thoracic Surgery is a must for surgeons who specialize in the treatment of lung and esophageal cancers and want to enhance their skills so they can overcome challenges and complications in their surgical practices.
Session Highlights
- Intraoperative Challenges during Thoracoscopic Lobectomy
- Intraoperative Challenges during Extended Pulmonary Resections
- Managing Complications after Pneumonectomy
- Intraoperative Challenges during Complex Esophageal Surgery
- Intraoperative Challenges during Esophagectomy
- Managing Complications after Esophageal Resection
- Intraoperative Challenges in Airway SurgeryComplex Case Presentations
Hotel rooms are limited!
Register/Reserve Accommodations
Program Directors
G. Alec Patterson
David J. Sugarbaker
Program Committee Members
Thomas A. D’Amico
Shaf Keshavjee
James D. Luketich
Bryan F. Meyers
Scott J. Swanson
October 23-24, 2015
Seaport Hotel & World Trade Center
Boston, MA
Overview
Complications remain one of the major challenges in treating patients. The 2015 Focus on Thoracic Surgery meeting will concentrate on the intraoperative challenges and management of complications facing thoracic surgeons in treating lung and esophageal cancers. This year’s course is particularly aimed at operating surgeons who are active and seek to learn innovative solutions to clinical problems during surgery.
This course will display new advanced diagnosis and prompt management approaches, allowing attendees to understand and readily translate them into daily practice. This two-day conference will have a faculty of experts in the field who are leaders in the application of new technologies in surgical management. It will combine informational sessions, panel discussions and complex case presentations.
Focus on Thoracic Surgery is a must for surgeons who specialize in the treatment of lung and esophageal cancers and want to enhance their skills so they can overcome challenges and complications in their surgical practices.
Session Highlights
- Intraoperative Challenges during Thoracoscopic Lobectomy
- Intraoperative Challenges during Extended Pulmonary Resections
- Managing Complications after Pneumonectomy
- Intraoperative Challenges during Complex Esophageal Surgery
- Intraoperative Challenges during Esophagectomy
- Managing Complications after Esophageal Resection
- Intraoperative Challenges in Airway SurgeryComplex Case Presentations
Hotel rooms are limited!
Register/Reserve Accommodations
Program Directors
G. Alec Patterson
David J. Sugarbaker
Program Committee Members
Thomas A. D’Amico
Shaf Keshavjee
James D. Luketich
Bryan F. Meyers
Scott J. Swanson
October 23-24, 2015
Seaport Hotel & World Trade Center
Boston, MA
Overview
Complications remain one of the major challenges in treating patients. The 2015 Focus on Thoracic Surgery meeting will concentrate on the intraoperative challenges and management of complications facing thoracic surgeons in treating lung and esophageal cancers. This year’s course is particularly aimed at operating surgeons who are active and seek to learn innovative solutions to clinical problems during surgery.
This course will display new advanced diagnosis and prompt management approaches, allowing attendees to understand and readily translate them into daily practice. This two-day conference will have a faculty of experts in the field who are leaders in the application of new technologies in surgical management. It will combine informational sessions, panel discussions and complex case presentations.
Focus on Thoracic Surgery is a must for surgeons who specialize in the treatment of lung and esophageal cancers and want to enhance their skills so they can overcome challenges and complications in their surgical practices.
Session Highlights
- Intraoperative Challenges during Thoracoscopic Lobectomy
- Intraoperative Challenges during Extended Pulmonary Resections
- Managing Complications after Pneumonectomy
- Intraoperative Challenges during Complex Esophageal Surgery
- Intraoperative Challenges during Esophagectomy
- Managing Complications after Esophageal Resection
- Intraoperative Challenges in Airway SurgeryComplex Case Presentations
Hotel rooms are limited!
Register/Reserve Accommodations
Program Directors
G. Alec Patterson
David J. Sugarbaker
Program Committee Members
Thomas A. D’Amico
Shaf Keshavjee
James D. Luketich
Bryan F. Meyers
Scott J. Swanson
Clinical advances drive lung cancer staging, classification changes
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
AT THE IASLC WORLD CONFERENCE
Key clinical point: Improved understanding of lung cancer over the last decade has prompted updates to international tumor staging and classification systems.
Major finding: The WHO 2015 Classification of Lung Tumors is expected to aid clinical practice.
Data source: Conference presentation of key changes to the WHO Classification and TNM staging system.
Disclosures: Dr. Travis and Dr. Rami-Porta reported having no disclosures relevant to their presentations.
Lives saved with lower systolic BP: SPRINT trial
Deaths were reduced by nearly one-quarter when systolic blood pressure was treated to a target of 120 rather than 140 mm Hg, according to a large NIH-sponsored study comparing standard blood pressure treatment with more-intensive lowering of systolic blood pressure. The lower blood pressure group also saw a 30% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death.
The magnitude of the effect of the lower blood pressure target prompted the study’s data safety monitoring board to end the study early, said officials from several National Institutes of Health agencies at a telebriefing. The study was unblinded in August 2015, and a full report of the primary outcome measures will come in a paper due out by the end of the year, they said.
The Systolic Blood Pressure Intervention Trial, or SPRINT, is a 100-site trial that enrolled more than 9,300 people in the United States and Puerto Rico aged at least 50 years with high blood pressure and at risk for cardiovascular disease; those with diabetes were excluded. Patients were randomized to a standard treatment target of 140 mm Hg or less, or to a more intensive 120 mm Hg.
SPRINT participants received evidence-based treatment with a variety of antihypertensives, with the intervention arm requiring an average of almost three medications, compared with just under two for the less-intensive treatment arm.
Against a backdrop of uncertainty in the literature about what the target systolic blood pressure should be for those with hypertension and at risk for cardiovascular events or kidney disease, the study provides compelling evidence that more-aggressive blood pressure lowering is important. “More-intensive management of blood pressure can save lives,” said Dr. Gary Gibbons, director of the National Heart, Lung, and Blood Institute. This is good news, he said, since about one in three Americans has high blood pressure, and only about half of those 70 million currently have their blood pressure under control.
Dr. Jackson T. Wright Jr., SPRINT study lead and director of the clinical hypertension program at Case Western Reserve University in Cleveland, also emphasized that intensive blood pressure management can prevent the cardiovascular complications of hypertension. Though subgroup analysis is ongoing, the effect seems robust and consistent across age groups, sex, and ethnicity, he said. SPRINT, he said, also “offers an excellent opportunity to examine the tolerability and safety of the lower target.” The first look at the safety data shows that the more-intensive treatment is well tolerated, though data analysis is ongoing, he said.
Dr. Suzanne Oparil, director of the vascular biology and hypertension program at the University of Alabama-Birmingham, said, “This is a time of enlightenment.” The previous absence of compelling data played a part in the debate surrounding blood pressure levels that should be used in guidance documents, and Dr. Gibbons and Dr. Wright both emphasized that they would expect the forthcoming primary outcomes paper to have an impact on guideline-writing bodies. Dr. Wright said, however, “We are not providing guidance for providers or patients right now. The study was just unblinded a little less than 3 weeks ago.”
In 2014, the group of experts who had constituted the JNC 8 panel, a team assembled in 2008 by NHLBI to update official U.S. hypertension management guidelines, set the target blood pressure for the general population aged 60 years or older to less than 150/90 mm Hg, a major break from long-standing practice to treat such patients to a target systolic pressure of less than 140 mm Hg (JAMA. 2014;311[5]:507-20). These guidelines, released after SPRINT began, remain controversial.
The SPRINT MIND trial, tracking the relationship between systolic blood pressure and cognitive impairment or dementia, is ongoing. The study is also still collecting data about kidney function in study participants.
The study was funded by the National Institutes of Health. Two drug companies, Takeda and Arbor, provided some medication for the trial.
On Twitter @karioakes
Deaths were reduced by nearly one-quarter when systolic blood pressure was treated to a target of 120 rather than 140 mm Hg, according to a large NIH-sponsored study comparing standard blood pressure treatment with more-intensive lowering of systolic blood pressure. The lower blood pressure group also saw a 30% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death.
The magnitude of the effect of the lower blood pressure target prompted the study’s data safety monitoring board to end the study early, said officials from several National Institutes of Health agencies at a telebriefing. The study was unblinded in August 2015, and a full report of the primary outcome measures will come in a paper due out by the end of the year, they said.
The Systolic Blood Pressure Intervention Trial, or SPRINT, is a 100-site trial that enrolled more than 9,300 people in the United States and Puerto Rico aged at least 50 years with high blood pressure and at risk for cardiovascular disease; those with diabetes were excluded. Patients were randomized to a standard treatment target of 140 mm Hg or less, or to a more intensive 120 mm Hg.
SPRINT participants received evidence-based treatment with a variety of antihypertensives, with the intervention arm requiring an average of almost three medications, compared with just under two for the less-intensive treatment arm.
Against a backdrop of uncertainty in the literature about what the target systolic blood pressure should be for those with hypertension and at risk for cardiovascular events or kidney disease, the study provides compelling evidence that more-aggressive blood pressure lowering is important. “More-intensive management of blood pressure can save lives,” said Dr. Gary Gibbons, director of the National Heart, Lung, and Blood Institute. This is good news, he said, since about one in three Americans has high blood pressure, and only about half of those 70 million currently have their blood pressure under control.
Dr. Jackson T. Wright Jr., SPRINT study lead and director of the clinical hypertension program at Case Western Reserve University in Cleveland, also emphasized that intensive blood pressure management can prevent the cardiovascular complications of hypertension. Though subgroup analysis is ongoing, the effect seems robust and consistent across age groups, sex, and ethnicity, he said. SPRINT, he said, also “offers an excellent opportunity to examine the tolerability and safety of the lower target.” The first look at the safety data shows that the more-intensive treatment is well tolerated, though data analysis is ongoing, he said.
Dr. Suzanne Oparil, director of the vascular biology and hypertension program at the University of Alabama-Birmingham, said, “This is a time of enlightenment.” The previous absence of compelling data played a part in the debate surrounding blood pressure levels that should be used in guidance documents, and Dr. Gibbons and Dr. Wright both emphasized that they would expect the forthcoming primary outcomes paper to have an impact on guideline-writing bodies. Dr. Wright said, however, “We are not providing guidance for providers or patients right now. The study was just unblinded a little less than 3 weeks ago.”
In 2014, the group of experts who had constituted the JNC 8 panel, a team assembled in 2008 by NHLBI to update official U.S. hypertension management guidelines, set the target blood pressure for the general population aged 60 years or older to less than 150/90 mm Hg, a major break from long-standing practice to treat such patients to a target systolic pressure of less than 140 mm Hg (JAMA. 2014;311[5]:507-20). These guidelines, released after SPRINT began, remain controversial.
The SPRINT MIND trial, tracking the relationship between systolic blood pressure and cognitive impairment or dementia, is ongoing. The study is also still collecting data about kidney function in study participants.
The study was funded by the National Institutes of Health. Two drug companies, Takeda and Arbor, provided some medication for the trial.
On Twitter @karioakes
Deaths were reduced by nearly one-quarter when systolic blood pressure was treated to a target of 120 rather than 140 mm Hg, according to a large NIH-sponsored study comparing standard blood pressure treatment with more-intensive lowering of systolic blood pressure. The lower blood pressure group also saw a 30% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death.
The magnitude of the effect of the lower blood pressure target prompted the study’s data safety monitoring board to end the study early, said officials from several National Institutes of Health agencies at a telebriefing. The study was unblinded in August 2015, and a full report of the primary outcome measures will come in a paper due out by the end of the year, they said.
The Systolic Blood Pressure Intervention Trial, or SPRINT, is a 100-site trial that enrolled more than 9,300 people in the United States and Puerto Rico aged at least 50 years with high blood pressure and at risk for cardiovascular disease; those with diabetes were excluded. Patients were randomized to a standard treatment target of 140 mm Hg or less, or to a more intensive 120 mm Hg.
SPRINT participants received evidence-based treatment with a variety of antihypertensives, with the intervention arm requiring an average of almost three medications, compared with just under two for the less-intensive treatment arm.
Against a backdrop of uncertainty in the literature about what the target systolic blood pressure should be for those with hypertension and at risk for cardiovascular events or kidney disease, the study provides compelling evidence that more-aggressive blood pressure lowering is important. “More-intensive management of blood pressure can save lives,” said Dr. Gary Gibbons, director of the National Heart, Lung, and Blood Institute. This is good news, he said, since about one in three Americans has high blood pressure, and only about half of those 70 million currently have their blood pressure under control.
Dr. Jackson T. Wright Jr., SPRINT study lead and director of the clinical hypertension program at Case Western Reserve University in Cleveland, also emphasized that intensive blood pressure management can prevent the cardiovascular complications of hypertension. Though subgroup analysis is ongoing, the effect seems robust and consistent across age groups, sex, and ethnicity, he said. SPRINT, he said, also “offers an excellent opportunity to examine the tolerability and safety of the lower target.” The first look at the safety data shows that the more-intensive treatment is well tolerated, though data analysis is ongoing, he said.
Dr. Suzanne Oparil, director of the vascular biology and hypertension program at the University of Alabama-Birmingham, said, “This is a time of enlightenment.” The previous absence of compelling data played a part in the debate surrounding blood pressure levels that should be used in guidance documents, and Dr. Gibbons and Dr. Wright both emphasized that they would expect the forthcoming primary outcomes paper to have an impact on guideline-writing bodies. Dr. Wright said, however, “We are not providing guidance for providers or patients right now. The study was just unblinded a little less than 3 weeks ago.”
In 2014, the group of experts who had constituted the JNC 8 panel, a team assembled in 2008 by NHLBI to update official U.S. hypertension management guidelines, set the target blood pressure for the general population aged 60 years or older to less than 150/90 mm Hg, a major break from long-standing practice to treat such patients to a target systolic pressure of less than 140 mm Hg (JAMA. 2014;311[5]:507-20). These guidelines, released after SPRINT began, remain controversial.
The SPRINT MIND trial, tracking the relationship between systolic blood pressure and cognitive impairment or dementia, is ongoing. The study is also still collecting data about kidney function in study participants.
The study was funded by the National Institutes of Health. Two drug companies, Takeda and Arbor, provided some medication for the trial.
On Twitter @karioakes
FROM AN NHLBI TELEBRIEFING
The pros and cons of novel anticoagulants
Novel anticoagulants will likely replace need for vitamin K antagonists
BY MADHUKAR S. PATEL, M.D., AND ELLIOT L. CHAIKOF, M.D.
The discovery of oral anticoagulants began in 1924, when Schofield linked the death of grazing cattle from internal hemorrhage to the consumption of spoiled sweet clover hay.1 It was not until 1941, however, while trying to understand this observation, that Campbell & Link were able to identify the dicoumarol anticoagulant, which formed as a result of the spoiling process.2 Ultimately, after noting that vitamin K led to reversal of the dicoumarol effect, synthesis of the first class of oral anticoagulants, known as vitamin K antagonists (VKAs), began.
Despite the numerous challenges associated with managing patients using this class of anticoagulants, VKAs have become the mainstay of oral anticoagulation therapy for the past 70 years. Over the past 5 years, however, new oral anticoagulants (NOACs) have emerged and are changing clinical practice.
Mechanistically, these medications are targeted therapies and work as either direct thrombin inhibitors (dabigatran etexilate) or direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). Given their favorable pharmacologic design, NOACs have the potential to replace VKAs as they not only have an encouraging safety profile, but also are therapeutically equivalent or even superior to VKAs when used in certain patient populations.
Pharmacologic design
The targeted drug design of NOACs provides many pharmacologic advantages. Compared to VKAs, NOACs have a notably more predictable pharmacologic profile and relatively wide therapeutic window, which allows for fixed dosing, a rapid onset and offset, and fewer drug interactions.3 These characteristics eliminate the need for the routine dose monitoring and serial dose adjustments frequently associated with VKAs.
NOACs less commonly require bridging therapy with parenteral unfractionated heparin or low-molecular-weight heparins (LMWH) while awaiting therapeutic drug levels, as these levels are reached sooner and more predictably than with VKAs.4 As with any medication, however, appropriate consideration should to be given to specific patient populations such as those who are older or have significant comorbidities that may influence drug effect and clearance. Lastly, it should be mentioned that the pharmacologic benefits of NOACs apply not only from a patient perspective, but also from a health care systems standpoint, as their use may provide an opportunity to deliver more cost-effective care.
Specifically, economic models using available clinical trial data for stroke prevention in nonvalvular atrial fibrillation have shown that NOACs (apixaban, dabigatran, and rivaroxaban) are cost-effective alternatives when compared to warfarin.5 Although the results from such economic analyses are limited by the modeling assumptions they rely upon, these findings suggest that at least initially, cost should not be used as a prohibitive reason for adopting these new therapeutics.
Patient selection
The decision to institute oral anticoagulation therapy depends on each patient’s individualized bleeding risk to benefit of ischemia prevention ratio. A major determinant of this ratio is the clinical indication for which anticoagulation is begun. Numerous phase III clinical trials have been conducted comparing the use of NOACs to VKAs or placebos for the management of nonvalvular atrial fibrillation and venous thromboembolism, and as adjunctive therapy for patients with acute coronary syndrome.6
Meta-analyses of randomized trials have shown the most significant benefit to be in patients with nonvalvular atrial fibrillation, where NOACs yield significant reductions in stroke, intracranial hemorrhage, and all-cause mortality compared to warfarin, while displaying variable effects with regard to gastrointestinal bleeding.6,7 In patients with VTE, NOACs have been found to have efficacy similar to that of VKAs with regard to the prevention of VTE or VTE-related death, and have been noted to have a better safety profile.6
Lastly, when studied as an adjunctive agent to dual antiplatelet therapy in patients with acute coronary syndrome, NOACs have been associated with an increased bleeding risk without a significant decrease in thrombosis risk.6 Taken together, these data suggest that the primary indication for instituting NOAC therapy should be considered strongly when deciding upon which class of anticoagulant to use.
Overcoming challenges
Since the introduction of NOACs, there has been concern over the lack of specific antidotes to therapy, especially when administered in patients with impaired clearance, a high likelihood of need for an urgent or emergent procedure, or those presenting with life threatening bleeding complications.
Most recently, however, interim analysis from clinical trial data has shown complete reversal of the direct thrombin inhibitor dabigatran with the humanized monoclonal antibody idarucizumab within minutes of administration in greater than 88% of patients studied.8 Similarly, agents such as a PER977 are currently under phase II clinical trials as they have been shown to form noncovalent hydrogen bonds and charge-charge interactions with oral factor Xa inhibitors as well as oral thrombin inhibitors leading to their reversal.9
Given these promising findings, it likely will not be long until reversal agents for NOACs become clinically available. Until that time, it is encouraging that the bleeding profile of these drugs has been found to be favorable compared to VKAs and their short half-life allows for a relatively expeditious natural reversal of their anticoagulant effect as the drug is eliminated.
Conclusion
Unlike the serendipitous path leading to the discovery of the first class of oral anticoagulants (VKAs), NOACs have been specifically designed to provide targeted anticoagulation and to address the shortcomings of VKAs. To this end, NOACs are becoming increasingly important in the management of patients with specific clinical conditions such as nonvalvular atrial fibrillation and venous thromboembolism, where they have been shown to provide a larger net clinical benefit relative to the available alternatives. Furthermore, with economic analyses providing evidence that NOACs are cost-effective for the healthcare system and clinical trial results suggesting progress in the development of antidotes for reversal, it is likely that with growing experience, these agents will replace VKAs as the mainstay for prophylactic and therapeutic oral anticoagulation in targeted patient populations.
Dr. Patel is a research fellow and Dr. Chaikof is surgeon-in-chief, both at the department of surgery, Beth Israel Deaconess Medical Center, Boston. They reported no conflicts of interest.
References
1. J Am Vet Med Assoc. 1924;64:553-75 (See Br J Haematol 2008 Mar 18;141[6]:757-63).
2. J Biol Chem. 1941;138:21-33 (See Nutr Rev. 1974 Aug;32[8]:244-6).
3. Am Soc Hematol Educ Program. 2013;2013:464-70.
4. Eur Heart J. 2013 Jul;34(27):2094-2106.
5. Stroke. 2013 Jun;44(6):1676-81.
6. Nat Rev Cardiol. 2014 Dec;11(12):693-703.
7. Lancet. 2014 Mar 15;383(9921):955-62.
8. N Engl J Med. 2015;373(6):511-20.
9. N Engl J Med. 2014;371(22):2141-2.
What the doctor didn’t order: unintended consequences and pitfalls of NOACs
BY THOMAS WAKEFIELD, M.D., ANDREA OBI, M.D., AND DAWN COLEMAN, M.D.
Recently, several new oral anticoagulants have gained FDA approval to replace warfarin, capturing the attention of popular media. These include dabigatran, rivaroxaban, apixaban, and edoxaban. Dabigatran targets activated factor II (factor IIa), while rivaroxaban, apixaban, and edoxaban target activated factor X (factor Xa). Easy to take with a once- or twice-daily pill, with no cumbersome monitoring, they represent a seemingly ideal treatment for the chronically anticoagulated patient. All agents are currently FDA approved in the United States for treatment of acute venous thromboembolism (VTE) and atrial fibrillation (AF).
Dabigatran and edoxaban
As with warfarin, dabigatran and edoxaban require the use of a low-molecular-weight heparin (LMWH) or unfractionated heparin “bridge” when therapy is beginning, while rivaroxaban and apixaban are instituted as monotherapy without such a bridge. Dabigatran etexilate (PradaxaR, Boehringer Ingelheim) has the longest half-life of all of the NOACs at 12-17 hours, and this half-life is prolonged with increasing age and decreasing renal function.1 It is the only new agent that can be at least partially reversed with dialysis.2 Edoxaban (SavaysaR, Daiichi Sankyo) carries a boxed warning stating that this agent is less effective in AF patients with a creatinine clearance greater than 95 mL/min, and that kidney function should be assessed prior to starting treatment: Such patients have a greater risk of stroke compared with similar patients treated with warfarin. Edoxaban is the only agent specifically tested at a lower dose in patients at significantly increased risk of bleeding complications (low body weight and/or decreased creatinine clearance).3
Rivaroxaban and apixaban
Rivaroxaban (XareltoR, Bayer and Janssen), and apixaban (EliquisR, Bristol Myers-Squibb), unique among the NOACs, have been tested for extended therapy of acute DVT after treatment of 6-12 months. They were found to result in a significant decrease in recurrent VTE without an increase in major bleeding compared to placebo.4,5 Rivaroxaban has once-daily dosing and apixaban has twice-daily dosing; both are immediate monotherapy, making them quite convenient for patients. Apixaban is the only agent among the NOACs to have a slight decrease in gastrointestinal bleeding compared to warfarin.6
Consequences and pitfalls with NOACs
Problems with these new drugs, which may diminish our current level of enthusiasm for these agents to totally replace warfarin, include the inability to reliably follow their levels and to reverse their anticoagulant effects, the lack of data available on bridging when other procedures need to be performed, their short half-lives, and the lack of data on their anti-inflammatory effects.
With regard to monitoring of anticoagulation, the International Society of Thrombosis and Hemostasis (ISTH) has published a recommendation7 that lists these scenarios:
• When a patient is bleeding.
• Before surgery or an invasive procedure when the patient has taken the drug in the previous 24 hours, or longer if creatinine clearance (CrCl) is less than 50 mL/min.
• Identification of subtherapeutic or supratherapeutic levels in patients taking other drugs that are known to affect pharmacokinetics.
• Identification of subtherapeutic or supratherapeutic levels in patients at body weight extremes.
• Patients with deteriorating renal function.
• During perioperative management.
• During reversal of anticoagulation.
• When there is suspicion of overdose.
• Assessment of compliance in patients suffering thrombotic events while on treatment.
Currently, there exists no commercially available reversal agent for any of the NOACs and existing reversal agents for traditional anticoagulants are of limited, if any, use. Drugs under development include agents for the factor Xa inhibitors and for the thrombin inhibitor. Until the time that specific reversal agents exist, supportive care is the mainstay of therapy. In cases of trauma or severe or life-threatening bleeding, administration of concentrated clotting factors (prothrombin complex concentrate) or dialysis (dabigatran only) may be utilized. However, data from large clinical trials is lacking. A recent study of 90 patients receiving an antibody directed against dabigatran has revealed that the anticoagulant effects of dabigatran were reversed safely within minutes of administration; however, drug levels were not consistently suppressed at 24 hours in 20% of the cohort.8
There are no national guidelines nor large scale studies to guide bridging NOACs for procedures. The relatively short half-life for these agents makes it likely that traditional bridging as is practiced for warfarin is not necessary.9 However, this represents a double edged sword; withholding anticoagulation for two doses (such as if a patient becomes ill or a clinician is overly cautious around the time of a procedure) may leave the patient unprotected.
The final question with the new agents is their anti-inflammatory effects. We know that heparin and LMWH have significant pleiotropic effects that are not necessarily related to their anticoagulant effects. These effects are important to decrease the inflammatory nature of the thrombus and its effect on the vein wall. We do not know if the new oral agents have similar effects, as this has never fully been tested. In view of the fact that two of the agents are being used as monotherapy agents without any heparin/LMWH bridge, the anti-inflammatory properties of these new agents should be defined to make sure that such a bridge is not necessary.
Conclusion
So, in summary, although these agents have much to offer, there are many questions that remain to be addressed and answered before they totally replace traditional approaches to anticoagulation, in the realm of VTE. It must not be overlooked that for all the benefits, they each carry a risk of bleeding as they all target portions of the coagulation mechanism. We believe, that as with any “gift horse,” physicians should perhaps examine the data more closely and proceed with caution.
Dr. Wakefield is director of the Samuel and Jean Frankel Cardiovascular Center, Dr. Obi is a vascular surgery fellow, and Dr. Coleman is program director, section of vascular surgery, at the University of Michigan, Ann Arbor. They reported no conflicts of interest.
References
1. N Engl J Med. 2009;361:2342-52.
2. J Vasc Surg: Venous Lymphat Disord. 2013;1:418-26.
3. N Engl J Med. 2013;369:1406-15.
4. N Engl J Med. 2010;363:2499-2510.
5. N Engl J Med. 2013;368:699-708.
6. Arterioscler Thromb Vasc Biol. 2015;35:1056-65.
7. J Thromb Haemost. 2013;11:756-60.
Novel anticoagulants will likely replace need for vitamin K antagonists
BY MADHUKAR S. PATEL, M.D., AND ELLIOT L. CHAIKOF, M.D.
The discovery of oral anticoagulants began in 1924, when Schofield linked the death of grazing cattle from internal hemorrhage to the consumption of spoiled sweet clover hay.1 It was not until 1941, however, while trying to understand this observation, that Campbell & Link were able to identify the dicoumarol anticoagulant, which formed as a result of the spoiling process.2 Ultimately, after noting that vitamin K led to reversal of the dicoumarol effect, synthesis of the first class of oral anticoagulants, known as vitamin K antagonists (VKAs), began.
Despite the numerous challenges associated with managing patients using this class of anticoagulants, VKAs have become the mainstay of oral anticoagulation therapy for the past 70 years. Over the past 5 years, however, new oral anticoagulants (NOACs) have emerged and are changing clinical practice.
Mechanistically, these medications are targeted therapies and work as either direct thrombin inhibitors (dabigatran etexilate) or direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). Given their favorable pharmacologic design, NOACs have the potential to replace VKAs as they not only have an encouraging safety profile, but also are therapeutically equivalent or even superior to VKAs when used in certain patient populations.
Pharmacologic design
The targeted drug design of NOACs provides many pharmacologic advantages. Compared to VKAs, NOACs have a notably more predictable pharmacologic profile and relatively wide therapeutic window, which allows for fixed dosing, a rapid onset and offset, and fewer drug interactions.3 These characteristics eliminate the need for the routine dose monitoring and serial dose adjustments frequently associated with VKAs.
NOACs less commonly require bridging therapy with parenteral unfractionated heparin or low-molecular-weight heparins (LMWH) while awaiting therapeutic drug levels, as these levels are reached sooner and more predictably than with VKAs.4 As with any medication, however, appropriate consideration should to be given to specific patient populations such as those who are older or have significant comorbidities that may influence drug effect and clearance. Lastly, it should be mentioned that the pharmacologic benefits of NOACs apply not only from a patient perspective, but also from a health care systems standpoint, as their use may provide an opportunity to deliver more cost-effective care.
Specifically, economic models using available clinical trial data for stroke prevention in nonvalvular atrial fibrillation have shown that NOACs (apixaban, dabigatran, and rivaroxaban) are cost-effective alternatives when compared to warfarin.5 Although the results from such economic analyses are limited by the modeling assumptions they rely upon, these findings suggest that at least initially, cost should not be used as a prohibitive reason for adopting these new therapeutics.
Patient selection
The decision to institute oral anticoagulation therapy depends on each patient’s individualized bleeding risk to benefit of ischemia prevention ratio. A major determinant of this ratio is the clinical indication for which anticoagulation is begun. Numerous phase III clinical trials have been conducted comparing the use of NOACs to VKAs or placebos for the management of nonvalvular atrial fibrillation and venous thromboembolism, and as adjunctive therapy for patients with acute coronary syndrome.6
Meta-analyses of randomized trials have shown the most significant benefit to be in patients with nonvalvular atrial fibrillation, where NOACs yield significant reductions in stroke, intracranial hemorrhage, and all-cause mortality compared to warfarin, while displaying variable effects with regard to gastrointestinal bleeding.6,7 In patients with VTE, NOACs have been found to have efficacy similar to that of VKAs with regard to the prevention of VTE or VTE-related death, and have been noted to have a better safety profile.6
Lastly, when studied as an adjunctive agent to dual antiplatelet therapy in patients with acute coronary syndrome, NOACs have been associated with an increased bleeding risk without a significant decrease in thrombosis risk.6 Taken together, these data suggest that the primary indication for instituting NOAC therapy should be considered strongly when deciding upon which class of anticoagulant to use.
Overcoming challenges
Since the introduction of NOACs, there has been concern over the lack of specific antidotes to therapy, especially when administered in patients with impaired clearance, a high likelihood of need for an urgent or emergent procedure, or those presenting with life threatening bleeding complications.
Most recently, however, interim analysis from clinical trial data has shown complete reversal of the direct thrombin inhibitor dabigatran with the humanized monoclonal antibody idarucizumab within minutes of administration in greater than 88% of patients studied.8 Similarly, agents such as a PER977 are currently under phase II clinical trials as they have been shown to form noncovalent hydrogen bonds and charge-charge interactions with oral factor Xa inhibitors as well as oral thrombin inhibitors leading to their reversal.9
Given these promising findings, it likely will not be long until reversal agents for NOACs become clinically available. Until that time, it is encouraging that the bleeding profile of these drugs has been found to be favorable compared to VKAs and their short half-life allows for a relatively expeditious natural reversal of their anticoagulant effect as the drug is eliminated.
Conclusion
Unlike the serendipitous path leading to the discovery of the first class of oral anticoagulants (VKAs), NOACs have been specifically designed to provide targeted anticoagulation and to address the shortcomings of VKAs. To this end, NOACs are becoming increasingly important in the management of patients with specific clinical conditions such as nonvalvular atrial fibrillation and venous thromboembolism, where they have been shown to provide a larger net clinical benefit relative to the available alternatives. Furthermore, with economic analyses providing evidence that NOACs are cost-effective for the healthcare system and clinical trial results suggesting progress in the development of antidotes for reversal, it is likely that with growing experience, these agents will replace VKAs as the mainstay for prophylactic and therapeutic oral anticoagulation in targeted patient populations.
Dr. Patel is a research fellow and Dr. Chaikof is surgeon-in-chief, both at the department of surgery, Beth Israel Deaconess Medical Center, Boston. They reported no conflicts of interest.
References
1. J Am Vet Med Assoc. 1924;64:553-75 (See Br J Haematol 2008 Mar 18;141[6]:757-63).
2. J Biol Chem. 1941;138:21-33 (See Nutr Rev. 1974 Aug;32[8]:244-6).
3. Am Soc Hematol Educ Program. 2013;2013:464-70.
4. Eur Heart J. 2013 Jul;34(27):2094-2106.
5. Stroke. 2013 Jun;44(6):1676-81.
6. Nat Rev Cardiol. 2014 Dec;11(12):693-703.
7. Lancet. 2014 Mar 15;383(9921):955-62.
8. N Engl J Med. 2015;373(6):511-20.
9. N Engl J Med. 2014;371(22):2141-2.
What the doctor didn’t order: unintended consequences and pitfalls of NOACs
BY THOMAS WAKEFIELD, M.D., ANDREA OBI, M.D., AND DAWN COLEMAN, M.D.
Recently, several new oral anticoagulants have gained FDA approval to replace warfarin, capturing the attention of popular media. These include dabigatran, rivaroxaban, apixaban, and edoxaban. Dabigatran targets activated factor II (factor IIa), while rivaroxaban, apixaban, and edoxaban target activated factor X (factor Xa). Easy to take with a once- or twice-daily pill, with no cumbersome monitoring, they represent a seemingly ideal treatment for the chronically anticoagulated patient. All agents are currently FDA approved in the United States for treatment of acute venous thromboembolism (VTE) and atrial fibrillation (AF).
Dabigatran and edoxaban
As with warfarin, dabigatran and edoxaban require the use of a low-molecular-weight heparin (LMWH) or unfractionated heparin “bridge” when therapy is beginning, while rivaroxaban and apixaban are instituted as monotherapy without such a bridge. Dabigatran etexilate (PradaxaR, Boehringer Ingelheim) has the longest half-life of all of the NOACs at 12-17 hours, and this half-life is prolonged with increasing age and decreasing renal function.1 It is the only new agent that can be at least partially reversed with dialysis.2 Edoxaban (SavaysaR, Daiichi Sankyo) carries a boxed warning stating that this agent is less effective in AF patients with a creatinine clearance greater than 95 mL/min, and that kidney function should be assessed prior to starting treatment: Such patients have a greater risk of stroke compared with similar patients treated with warfarin. Edoxaban is the only agent specifically tested at a lower dose in patients at significantly increased risk of bleeding complications (low body weight and/or decreased creatinine clearance).3
Rivaroxaban and apixaban
Rivaroxaban (XareltoR, Bayer and Janssen), and apixaban (EliquisR, Bristol Myers-Squibb), unique among the NOACs, have been tested for extended therapy of acute DVT after treatment of 6-12 months. They were found to result in a significant decrease in recurrent VTE without an increase in major bleeding compared to placebo.4,5 Rivaroxaban has once-daily dosing and apixaban has twice-daily dosing; both are immediate monotherapy, making them quite convenient for patients. Apixaban is the only agent among the NOACs to have a slight decrease in gastrointestinal bleeding compared to warfarin.6
Consequences and pitfalls with NOACs
Problems with these new drugs, which may diminish our current level of enthusiasm for these agents to totally replace warfarin, include the inability to reliably follow their levels and to reverse their anticoagulant effects, the lack of data available on bridging when other procedures need to be performed, their short half-lives, and the lack of data on their anti-inflammatory effects.
With regard to monitoring of anticoagulation, the International Society of Thrombosis and Hemostasis (ISTH) has published a recommendation7 that lists these scenarios:
• When a patient is bleeding.
• Before surgery or an invasive procedure when the patient has taken the drug in the previous 24 hours, or longer if creatinine clearance (CrCl) is less than 50 mL/min.
• Identification of subtherapeutic or supratherapeutic levels in patients taking other drugs that are known to affect pharmacokinetics.
• Identification of subtherapeutic or supratherapeutic levels in patients at body weight extremes.
• Patients with deteriorating renal function.
• During perioperative management.
• During reversal of anticoagulation.
• When there is suspicion of overdose.
• Assessment of compliance in patients suffering thrombotic events while on treatment.
Currently, there exists no commercially available reversal agent for any of the NOACs and existing reversal agents for traditional anticoagulants are of limited, if any, use. Drugs under development include agents for the factor Xa inhibitors and for the thrombin inhibitor. Until the time that specific reversal agents exist, supportive care is the mainstay of therapy. In cases of trauma or severe or life-threatening bleeding, administration of concentrated clotting factors (prothrombin complex concentrate) or dialysis (dabigatran only) may be utilized. However, data from large clinical trials is lacking. A recent study of 90 patients receiving an antibody directed against dabigatran has revealed that the anticoagulant effects of dabigatran were reversed safely within minutes of administration; however, drug levels were not consistently suppressed at 24 hours in 20% of the cohort.8
There are no national guidelines nor large scale studies to guide bridging NOACs for procedures. The relatively short half-life for these agents makes it likely that traditional bridging as is practiced for warfarin is not necessary.9 However, this represents a double edged sword; withholding anticoagulation for two doses (such as if a patient becomes ill or a clinician is overly cautious around the time of a procedure) may leave the patient unprotected.
The final question with the new agents is their anti-inflammatory effects. We know that heparin and LMWH have significant pleiotropic effects that are not necessarily related to their anticoagulant effects. These effects are important to decrease the inflammatory nature of the thrombus and its effect on the vein wall. We do not know if the new oral agents have similar effects, as this has never fully been tested. In view of the fact that two of the agents are being used as monotherapy agents without any heparin/LMWH bridge, the anti-inflammatory properties of these new agents should be defined to make sure that such a bridge is not necessary.
Conclusion
So, in summary, although these agents have much to offer, there are many questions that remain to be addressed and answered before they totally replace traditional approaches to anticoagulation, in the realm of VTE. It must not be overlooked that for all the benefits, they each carry a risk of bleeding as they all target portions of the coagulation mechanism. We believe, that as with any “gift horse,” physicians should perhaps examine the data more closely and proceed with caution.
Dr. Wakefield is director of the Samuel and Jean Frankel Cardiovascular Center, Dr. Obi is a vascular surgery fellow, and Dr. Coleman is program director, section of vascular surgery, at the University of Michigan, Ann Arbor. They reported no conflicts of interest.
References
1. N Engl J Med. 2009;361:2342-52.
2. J Vasc Surg: Venous Lymphat Disord. 2013;1:418-26.
3. N Engl J Med. 2013;369:1406-15.
4. N Engl J Med. 2010;363:2499-2510.
5. N Engl J Med. 2013;368:699-708.
6. Arterioscler Thromb Vasc Biol. 2015;35:1056-65.
7. J Thromb Haemost. 2013;11:756-60.
Novel anticoagulants will likely replace need for vitamin K antagonists
BY MADHUKAR S. PATEL, M.D., AND ELLIOT L. CHAIKOF, M.D.
The discovery of oral anticoagulants began in 1924, when Schofield linked the death of grazing cattle from internal hemorrhage to the consumption of spoiled sweet clover hay.1 It was not until 1941, however, while trying to understand this observation, that Campbell & Link were able to identify the dicoumarol anticoagulant, which formed as a result of the spoiling process.2 Ultimately, after noting that vitamin K led to reversal of the dicoumarol effect, synthesis of the first class of oral anticoagulants, known as vitamin K antagonists (VKAs), began.
Despite the numerous challenges associated with managing patients using this class of anticoagulants, VKAs have become the mainstay of oral anticoagulation therapy for the past 70 years. Over the past 5 years, however, new oral anticoagulants (NOACs) have emerged and are changing clinical practice.
Mechanistically, these medications are targeted therapies and work as either direct thrombin inhibitors (dabigatran etexilate) or direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). Given their favorable pharmacologic design, NOACs have the potential to replace VKAs as they not only have an encouraging safety profile, but also are therapeutically equivalent or even superior to VKAs when used in certain patient populations.
Pharmacologic design
The targeted drug design of NOACs provides many pharmacologic advantages. Compared to VKAs, NOACs have a notably more predictable pharmacologic profile and relatively wide therapeutic window, which allows for fixed dosing, a rapid onset and offset, and fewer drug interactions.3 These characteristics eliminate the need for the routine dose monitoring and serial dose adjustments frequently associated with VKAs.
NOACs less commonly require bridging therapy with parenteral unfractionated heparin or low-molecular-weight heparins (LMWH) while awaiting therapeutic drug levels, as these levels are reached sooner and more predictably than with VKAs.4 As with any medication, however, appropriate consideration should to be given to specific patient populations such as those who are older or have significant comorbidities that may influence drug effect and clearance. Lastly, it should be mentioned that the pharmacologic benefits of NOACs apply not only from a patient perspective, but also from a health care systems standpoint, as their use may provide an opportunity to deliver more cost-effective care.
Specifically, economic models using available clinical trial data for stroke prevention in nonvalvular atrial fibrillation have shown that NOACs (apixaban, dabigatran, and rivaroxaban) are cost-effective alternatives when compared to warfarin.5 Although the results from such economic analyses are limited by the modeling assumptions they rely upon, these findings suggest that at least initially, cost should not be used as a prohibitive reason for adopting these new therapeutics.
Patient selection
The decision to institute oral anticoagulation therapy depends on each patient’s individualized bleeding risk to benefit of ischemia prevention ratio. A major determinant of this ratio is the clinical indication for which anticoagulation is begun. Numerous phase III clinical trials have been conducted comparing the use of NOACs to VKAs or placebos for the management of nonvalvular atrial fibrillation and venous thromboembolism, and as adjunctive therapy for patients with acute coronary syndrome.6
Meta-analyses of randomized trials have shown the most significant benefit to be in patients with nonvalvular atrial fibrillation, where NOACs yield significant reductions in stroke, intracranial hemorrhage, and all-cause mortality compared to warfarin, while displaying variable effects with regard to gastrointestinal bleeding.6,7 In patients with VTE, NOACs have been found to have efficacy similar to that of VKAs with regard to the prevention of VTE or VTE-related death, and have been noted to have a better safety profile.6
Lastly, when studied as an adjunctive agent to dual antiplatelet therapy in patients with acute coronary syndrome, NOACs have been associated with an increased bleeding risk without a significant decrease in thrombosis risk.6 Taken together, these data suggest that the primary indication for instituting NOAC therapy should be considered strongly when deciding upon which class of anticoagulant to use.
Overcoming challenges
Since the introduction of NOACs, there has been concern over the lack of specific antidotes to therapy, especially when administered in patients with impaired clearance, a high likelihood of need for an urgent or emergent procedure, or those presenting with life threatening bleeding complications.
Most recently, however, interim analysis from clinical trial data has shown complete reversal of the direct thrombin inhibitor dabigatran with the humanized monoclonal antibody idarucizumab within minutes of administration in greater than 88% of patients studied.8 Similarly, agents such as a PER977 are currently under phase II clinical trials as they have been shown to form noncovalent hydrogen bonds and charge-charge interactions with oral factor Xa inhibitors as well as oral thrombin inhibitors leading to their reversal.9
Given these promising findings, it likely will not be long until reversal agents for NOACs become clinically available. Until that time, it is encouraging that the bleeding profile of these drugs has been found to be favorable compared to VKAs and their short half-life allows for a relatively expeditious natural reversal of their anticoagulant effect as the drug is eliminated.
Conclusion
Unlike the serendipitous path leading to the discovery of the first class of oral anticoagulants (VKAs), NOACs have been specifically designed to provide targeted anticoagulation and to address the shortcomings of VKAs. To this end, NOACs are becoming increasingly important in the management of patients with specific clinical conditions such as nonvalvular atrial fibrillation and venous thromboembolism, where they have been shown to provide a larger net clinical benefit relative to the available alternatives. Furthermore, with economic analyses providing evidence that NOACs are cost-effective for the healthcare system and clinical trial results suggesting progress in the development of antidotes for reversal, it is likely that with growing experience, these agents will replace VKAs as the mainstay for prophylactic and therapeutic oral anticoagulation in targeted patient populations.
Dr. Patel is a research fellow and Dr. Chaikof is surgeon-in-chief, both at the department of surgery, Beth Israel Deaconess Medical Center, Boston. They reported no conflicts of interest.
References
1. J Am Vet Med Assoc. 1924;64:553-75 (See Br J Haematol 2008 Mar 18;141[6]:757-63).
2. J Biol Chem. 1941;138:21-33 (See Nutr Rev. 1974 Aug;32[8]:244-6).
3. Am Soc Hematol Educ Program. 2013;2013:464-70.
4. Eur Heart J. 2013 Jul;34(27):2094-2106.
5. Stroke. 2013 Jun;44(6):1676-81.
6. Nat Rev Cardiol. 2014 Dec;11(12):693-703.
7. Lancet. 2014 Mar 15;383(9921):955-62.
8. N Engl J Med. 2015;373(6):511-20.
9. N Engl J Med. 2014;371(22):2141-2.
What the doctor didn’t order: unintended consequences and pitfalls of NOACs
BY THOMAS WAKEFIELD, M.D., ANDREA OBI, M.D., AND DAWN COLEMAN, M.D.
Recently, several new oral anticoagulants have gained FDA approval to replace warfarin, capturing the attention of popular media. These include dabigatran, rivaroxaban, apixaban, and edoxaban. Dabigatran targets activated factor II (factor IIa), while rivaroxaban, apixaban, and edoxaban target activated factor X (factor Xa). Easy to take with a once- or twice-daily pill, with no cumbersome monitoring, they represent a seemingly ideal treatment for the chronically anticoagulated patient. All agents are currently FDA approved in the United States for treatment of acute venous thromboembolism (VTE) and atrial fibrillation (AF).
Dabigatran and edoxaban
As with warfarin, dabigatran and edoxaban require the use of a low-molecular-weight heparin (LMWH) or unfractionated heparin “bridge” when therapy is beginning, while rivaroxaban and apixaban are instituted as monotherapy without such a bridge. Dabigatran etexilate (PradaxaR, Boehringer Ingelheim) has the longest half-life of all of the NOACs at 12-17 hours, and this half-life is prolonged with increasing age and decreasing renal function.1 It is the only new agent that can be at least partially reversed with dialysis.2 Edoxaban (SavaysaR, Daiichi Sankyo) carries a boxed warning stating that this agent is less effective in AF patients with a creatinine clearance greater than 95 mL/min, and that kidney function should be assessed prior to starting treatment: Such patients have a greater risk of stroke compared with similar patients treated with warfarin. Edoxaban is the only agent specifically tested at a lower dose in patients at significantly increased risk of bleeding complications (low body weight and/or decreased creatinine clearance).3
Rivaroxaban and apixaban
Rivaroxaban (XareltoR, Bayer and Janssen), and apixaban (EliquisR, Bristol Myers-Squibb), unique among the NOACs, have been tested for extended therapy of acute DVT after treatment of 6-12 months. They were found to result in a significant decrease in recurrent VTE without an increase in major bleeding compared to placebo.4,5 Rivaroxaban has once-daily dosing and apixaban has twice-daily dosing; both are immediate monotherapy, making them quite convenient for patients. Apixaban is the only agent among the NOACs to have a slight decrease in gastrointestinal bleeding compared to warfarin.6
Consequences and pitfalls with NOACs
Problems with these new drugs, which may diminish our current level of enthusiasm for these agents to totally replace warfarin, include the inability to reliably follow their levels and to reverse their anticoagulant effects, the lack of data available on bridging when other procedures need to be performed, their short half-lives, and the lack of data on their anti-inflammatory effects.
With regard to monitoring of anticoagulation, the International Society of Thrombosis and Hemostasis (ISTH) has published a recommendation7 that lists these scenarios:
• When a patient is bleeding.
• Before surgery or an invasive procedure when the patient has taken the drug in the previous 24 hours, or longer if creatinine clearance (CrCl) is less than 50 mL/min.
• Identification of subtherapeutic or supratherapeutic levels in patients taking other drugs that are known to affect pharmacokinetics.
• Identification of subtherapeutic or supratherapeutic levels in patients at body weight extremes.
• Patients with deteriorating renal function.
• During perioperative management.
• During reversal of anticoagulation.
• When there is suspicion of overdose.
• Assessment of compliance in patients suffering thrombotic events while on treatment.
Currently, there exists no commercially available reversal agent for any of the NOACs and existing reversal agents for traditional anticoagulants are of limited, if any, use. Drugs under development include agents for the factor Xa inhibitors and for the thrombin inhibitor. Until the time that specific reversal agents exist, supportive care is the mainstay of therapy. In cases of trauma or severe or life-threatening bleeding, administration of concentrated clotting factors (prothrombin complex concentrate) or dialysis (dabigatran only) may be utilized. However, data from large clinical trials is lacking. A recent study of 90 patients receiving an antibody directed against dabigatran has revealed that the anticoagulant effects of dabigatran were reversed safely within minutes of administration; however, drug levels were not consistently suppressed at 24 hours in 20% of the cohort.8
There are no national guidelines nor large scale studies to guide bridging NOACs for procedures. The relatively short half-life for these agents makes it likely that traditional bridging as is practiced for warfarin is not necessary.9 However, this represents a double edged sword; withholding anticoagulation for two doses (such as if a patient becomes ill or a clinician is overly cautious around the time of a procedure) may leave the patient unprotected.
The final question with the new agents is their anti-inflammatory effects. We know that heparin and LMWH have significant pleiotropic effects that are not necessarily related to their anticoagulant effects. These effects are important to decrease the inflammatory nature of the thrombus and its effect on the vein wall. We do not know if the new oral agents have similar effects, as this has never fully been tested. In view of the fact that two of the agents are being used as monotherapy agents without any heparin/LMWH bridge, the anti-inflammatory properties of these new agents should be defined to make sure that such a bridge is not necessary.
Conclusion
So, in summary, although these agents have much to offer, there are many questions that remain to be addressed and answered before they totally replace traditional approaches to anticoagulation, in the realm of VTE. It must not be overlooked that for all the benefits, they each carry a risk of bleeding as they all target portions of the coagulation mechanism. We believe, that as with any “gift horse,” physicians should perhaps examine the data more closely and proceed with caution.
Dr. Wakefield is director of the Samuel and Jean Frankel Cardiovascular Center, Dr. Obi is a vascular surgery fellow, and Dr. Coleman is program director, section of vascular surgery, at the University of Michigan, Ann Arbor. They reported no conflicts of interest.
References
1. N Engl J Med. 2009;361:2342-52.
2. J Vasc Surg: Venous Lymphat Disord. 2013;1:418-26.
3. N Engl J Med. 2013;369:1406-15.
4. N Engl J Med. 2010;363:2499-2510.
5. N Engl J Med. 2013;368:699-708.
6. Arterioscler Thromb Vasc Biol. 2015;35:1056-65.
7. J Thromb Haemost. 2013;11:756-60.
HHS: Expand antidiscrimination protections to transgender patients
Transgender patients who receive health care via government programs or funding must receive equal access to treatments and insurance coverage, according to a proposed rule issued Sept. 3 by the Health and Human Services department. The rule would extend antidiscrimination policies under the Affordable Care Act to include gender identity.
The rule would apply to health providers who accept patients covered by Medicare and Medicaid as well as insurance purchased via the health insurance marketplaces.
“The proposed rule clarifies and harmonizes existing well-established federal civil rights laws and clarifies the standards that HHS and in particular, the Office of Civil Rights, will apply in implementing [ACA] Section 1557,” Jocelyn Samuels, OCR director, said in a press conference. “Prior laws enforced by the Office of Civil Rights barred discrimination based only on race, color, national origin, age, or disability. All of the protections against sex discrimination that will be incorporated into the rule are new in this space.”
Section 1557 of the ACA extends civil rights protections to ban sex discrimination in federal health care programs and activities. The new proposed rule establishes that the prohibition on sex discrimination includes discrimination based on gender identity. The rule also includes requirements for effective communication for patients with disabilities and enhanced language assistance for patients with limited English proficiency.
Specifics of the proposed rule include:
• Patients must be treated equally and consistent with their gender identity by health providers. Insurers must provide fair access to coverage regardless of gender identity. For example, some insurers have historically excluded coverage of all care related to gender transition. Such categorical exclusions are prohibited under the proposed rule.
• Women must be treated equally with men in the health care they receive, not only in the health coverage they obtain but in the services they seek from providers.
• For patients with disabilities, the rule contains requirements for the provision of auxiliary aids and services, including alternative formats and sign language interpreters and the accessibility of programs offered through electronic and information technology.
• The rule bolsters language assistance for people with limited English proficiency so that patients are able to more effectively communicate with their providers to describe their symptoms and understand treatment.
During the press conference, Ms. Samuels clarified that the rule does not mean that health insurers must cover any specific treatments or procedures, rather they must apply nondiscriminatory criteria when assessing coverage requests.
The proposed extension of protections is, in part, driven by ongoing cases of sex and identity discrimination by some health providers. In one case, a hospital denied a transgender patient a room assignment consistent with her gender identity. In another, a male domestic violence victim was denied services at a hospital because he did not fit the traditional profile of a domestic violence victim, Ms. Samuels said. In another case, a health provider required that a husband be the guarantor for his wife’s medical bills but did not require the same for male patients and their spouses.
“There continue to be serious problems of discrimination in the health care arena,” she said. “This proposed rule provides very valuable tools for us to be able to appropriately address them.”
HHS is requesting comments on whether Section 1557 should include exemptions for religious organizations and, if so, to what extent. The administration notes that nothing in the proposed rule would affect the application of existing protections for religious beliefs and practices, such as provider conscience laws and regulations under the ACA involving preventive health services.
Comments on the rule will be accepted at www.regulations.gov until Nov. 6.
On Twitter @legal_med
Transgender patients who receive health care via government programs or funding must receive equal access to treatments and insurance coverage, according to a proposed rule issued Sept. 3 by the Health and Human Services department. The rule would extend antidiscrimination policies under the Affordable Care Act to include gender identity.
The rule would apply to health providers who accept patients covered by Medicare and Medicaid as well as insurance purchased via the health insurance marketplaces.
“The proposed rule clarifies and harmonizes existing well-established federal civil rights laws and clarifies the standards that HHS and in particular, the Office of Civil Rights, will apply in implementing [ACA] Section 1557,” Jocelyn Samuels, OCR director, said in a press conference. “Prior laws enforced by the Office of Civil Rights barred discrimination based only on race, color, national origin, age, or disability. All of the protections against sex discrimination that will be incorporated into the rule are new in this space.”
Section 1557 of the ACA extends civil rights protections to ban sex discrimination in federal health care programs and activities. The new proposed rule establishes that the prohibition on sex discrimination includes discrimination based on gender identity. The rule also includes requirements for effective communication for patients with disabilities and enhanced language assistance for patients with limited English proficiency.
Specifics of the proposed rule include:
• Patients must be treated equally and consistent with their gender identity by health providers. Insurers must provide fair access to coverage regardless of gender identity. For example, some insurers have historically excluded coverage of all care related to gender transition. Such categorical exclusions are prohibited under the proposed rule.
• Women must be treated equally with men in the health care they receive, not only in the health coverage they obtain but in the services they seek from providers.
• For patients with disabilities, the rule contains requirements for the provision of auxiliary aids and services, including alternative formats and sign language interpreters and the accessibility of programs offered through electronic and information technology.
• The rule bolsters language assistance for people with limited English proficiency so that patients are able to more effectively communicate with their providers to describe their symptoms and understand treatment.
During the press conference, Ms. Samuels clarified that the rule does not mean that health insurers must cover any specific treatments or procedures, rather they must apply nondiscriminatory criteria when assessing coverage requests.
The proposed extension of protections is, in part, driven by ongoing cases of sex and identity discrimination by some health providers. In one case, a hospital denied a transgender patient a room assignment consistent with her gender identity. In another, a male domestic violence victim was denied services at a hospital because he did not fit the traditional profile of a domestic violence victim, Ms. Samuels said. In another case, a health provider required that a husband be the guarantor for his wife’s medical bills but did not require the same for male patients and their spouses.
“There continue to be serious problems of discrimination in the health care arena,” she said. “This proposed rule provides very valuable tools for us to be able to appropriately address them.”
HHS is requesting comments on whether Section 1557 should include exemptions for religious organizations and, if so, to what extent. The administration notes that nothing in the proposed rule would affect the application of existing protections for religious beliefs and practices, such as provider conscience laws and regulations under the ACA involving preventive health services.
Comments on the rule will be accepted at www.regulations.gov until Nov. 6.
On Twitter @legal_med
Transgender patients who receive health care via government programs or funding must receive equal access to treatments and insurance coverage, according to a proposed rule issued Sept. 3 by the Health and Human Services department. The rule would extend antidiscrimination policies under the Affordable Care Act to include gender identity.
The rule would apply to health providers who accept patients covered by Medicare and Medicaid as well as insurance purchased via the health insurance marketplaces.
“The proposed rule clarifies and harmonizes existing well-established federal civil rights laws and clarifies the standards that HHS and in particular, the Office of Civil Rights, will apply in implementing [ACA] Section 1557,” Jocelyn Samuels, OCR director, said in a press conference. “Prior laws enforced by the Office of Civil Rights barred discrimination based only on race, color, national origin, age, or disability. All of the protections against sex discrimination that will be incorporated into the rule are new in this space.”
Section 1557 of the ACA extends civil rights protections to ban sex discrimination in federal health care programs and activities. The new proposed rule establishes that the prohibition on sex discrimination includes discrimination based on gender identity. The rule also includes requirements for effective communication for patients with disabilities and enhanced language assistance for patients with limited English proficiency.
Specifics of the proposed rule include:
• Patients must be treated equally and consistent with their gender identity by health providers. Insurers must provide fair access to coverage regardless of gender identity. For example, some insurers have historically excluded coverage of all care related to gender transition. Such categorical exclusions are prohibited under the proposed rule.
• Women must be treated equally with men in the health care they receive, not only in the health coverage they obtain but in the services they seek from providers.
• For patients with disabilities, the rule contains requirements for the provision of auxiliary aids and services, including alternative formats and sign language interpreters and the accessibility of programs offered through electronic and information technology.
• The rule bolsters language assistance for people with limited English proficiency so that patients are able to more effectively communicate with their providers to describe their symptoms and understand treatment.
During the press conference, Ms. Samuels clarified that the rule does not mean that health insurers must cover any specific treatments or procedures, rather they must apply nondiscriminatory criteria when assessing coverage requests.
The proposed extension of protections is, in part, driven by ongoing cases of sex and identity discrimination by some health providers. In one case, a hospital denied a transgender patient a room assignment consistent with her gender identity. In another, a male domestic violence victim was denied services at a hospital because he did not fit the traditional profile of a domestic violence victim, Ms. Samuels said. In another case, a health provider required that a husband be the guarantor for his wife’s medical bills but did not require the same for male patients and their spouses.
“There continue to be serious problems of discrimination in the health care arena,” she said. “This proposed rule provides very valuable tools for us to be able to appropriately address them.”
HHS is requesting comments on whether Section 1557 should include exemptions for religious organizations and, if so, to what extent. The administration notes that nothing in the proposed rule would affect the application of existing protections for religious beliefs and practices, such as provider conscience laws and regulations under the ACA involving preventive health services.
Comments on the rule will be accepted at www.regulations.gov until Nov. 6.
On Twitter @legal_med