User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
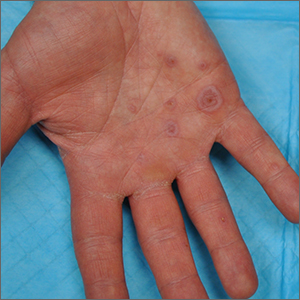
Palmar rash
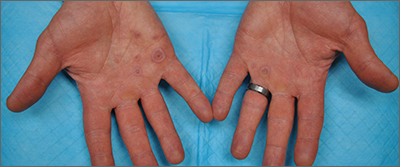
This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828

This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828
Elevated PCSK9 levels associated with psoriasis suggest new treatment target
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
FROM JAMA DERMATOLOGY
Angioedema risk jumps when switching HF meds
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Long COVID affecting more than one-third of college students, faculty
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
FROM EMERGING INFECTIOUS DISEASES
‘Sugar tax’ prevented thousands of girls becoming obese
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
Novel resuscitation for patients with nonshockable rhythms in cardiac arrest
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
Don’t cross the friends line with patients
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
More type 2 diabetes deaths from cancer than heart disease
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.
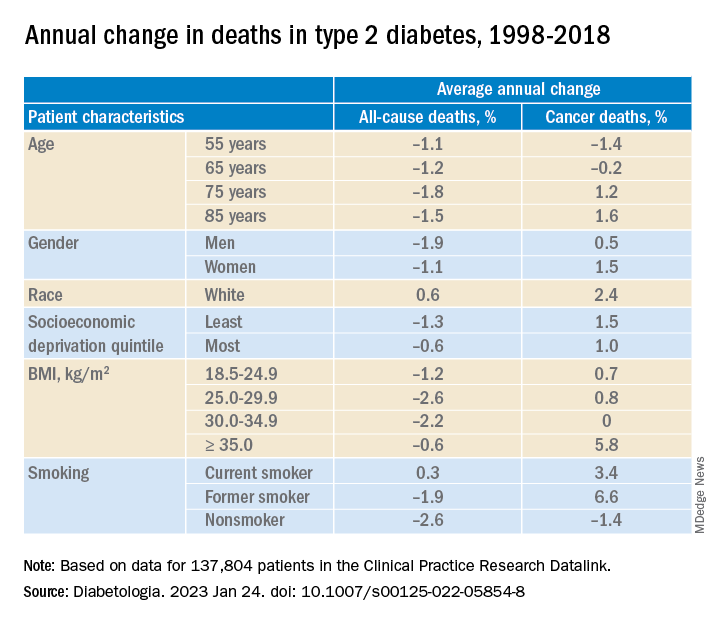
In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.

In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.

In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DIABETOLOGIA
FDA panel backs shift toward one-dose COVID shot
The FDA is looking to give clearer direction to vaccine makers about future development of COVID-19 vaccines. The plan is to narrow down the current complex landscape of options for vaccinations, and thus help increase use of these shots.
COVID remains a serious threat, causing about 4,000 deaths a week recently, according to the Centers for Disease Control and Prevention. The 21 members of the Vaccines and Related Biological Products Advisory Committee (VRBPAC) on Jan. 26 voted unanimously “yes” on a single question posed by the FDA:
“Does the committee recommend harmonizing the vaccine strain composition of primary series and booster doses in the U.S. to a single composition, e.g., the composition for all vaccines administered currently would be a bivalent vaccine (Original plus Omicron BA.4/BA.5)?”
In other words, would it be better to have one vaccine potentially combining multiple strains of the virus, instead of multiple vaccines – such as a two-shot primary series then a booster containing different combinations of viral strains.
The FDA will consider the panel’s advice as it outlines new strategies for keeping ahead of the evolving virus.
In explaining their support for the FDA plan, panel members said they hoped that a simpler regime would aid in persuading more people to get COVID vaccines.
Pamela McInnes, DDS, MSc, noted that it’s difficult to explain to many people that the vaccine works to protect them from more severe illness if they contract COVID after getting vaccinated.
“That is a real challenge,” said Dr. McInness, retired deputy director of the National Center for Advancing Translational Sciences at the National Institutes of Health.
“The message that you would have gotten more sick and landed in the hospital resonates with me, but I’m not sure if it resonates with” many people who become infected, she said.
The plan
In the briefing document for the meeting, the FDA outlined a plan for transitioning from the current complex landscape of COVID-19 vaccines to a single vaccine composition for the primary series and booster vaccination.
This would require harmonizing the strain composition of all COVID-19 vaccines; simplifying the immunization schedule for future vaccination campaigns to administer a two-dose series in certain young children and in older adults and persons with compromised immunity, and only one dose in all other individuals; and establishing a process for vaccine strain selection recommendations, similar in many ways to that used for seasonal influenza vaccines, based on prevailing and predicted variants that would take place by June to allow for vaccine production by September.
During the discussion, though, questions arose about the June target date. Given the production schedule for some vaccines, that date might need to shift, said Jerry Weir, PhD, director of the division of viral products at FDA’s Center for Biologics Evaluation and Research.
“We’re all just going to have to maintain flexibility,” Dr. Weir said, adding that there is not yet a “good pattern” established for updating these vaccines.
Increasing vaccination rates
There was broad consensus about the need to boost public support for COVID-19 vaccinations. While about 81% of the U.S. population has had at least one dose of this vaccine, only 15.3% have had an updated bivalent booster dose, according to the CDC.
“Anything that results in better public communication would be extremely valuable,” said committee member Henry H. Bernstein, DO, MHCM, of the Zucker School of Medicine at Hofstra/Northwell Health in Hempstead, N.Y.
But it’s unclear what expectations will be prioritized for the COVID vaccine program, he said.
“Realistically, I don’t think we can have it all – less infection, less transmission, less severe disease, and less long COVID,” Dr. Bernstein said. “And that seems to be a major challenge for public messaging.”
Panelists press for more data
Other committee members also pressed for clearer targets in evaluating the goals for COVID vaccines, and for more robust data.
Like his fellow VRBPAC members, Cody Meissner, MD, of Dartmouth’s Geisel School of Medicine, Hanover, N.H., supported a move toward harmonizing the strains used in different companies’ vaccines. But he added that it wasn’t clear yet how frequently they should be administered.
“We need to see what happens with disease burden,” Dr. Meissner said. “We may or may not need annual vaccination. It’s just awfully early, it seems to me, in this process to answer that question.”
Among those serving on VRBPAC was one of the FDA’s more vocal critics on these points, Paul A. Offit, MD, a vaccine expert from Children’s Hospital of Philadelphia. Dr. Offit, for example, joined former FDA officials in writing a November opinion article for the Washington Post, arguing that the evidence for boosters for healthy younger adults was not strong.
At the Jan. 26 meeting, he supported the drive toward simplification of COVID vaccine schedules, while arguing for more data about how well these products are working.
“This virus is going to be with us for years, if not decades, and there will always be vulnerable groups who are going to be hospitalized and killed by the virus,” Dr. Offit said.
The CDC needs to provide more information about the characteristics of people being hospitalized with COVID infections, including their ages and comorbidities as well as details about their vaccine history, he said. In addition, academic researchers should provide a clearer picture of what immunological predictors are at play in increasing people’s risk from COVID.
“Then and only then can we really best make the decision about who gets vaccinated with what and when,” Dr. Offit said.
VRBPAC member Ofer Levy, MD, PhD, also urged the FDA to press for a collection of more robust and detailed information about the immune response to COVID-19 vaccinations, such as a deeper look at what’s happening with antibodies.
“I hope FDA will continue to reflect on how to best take this information forward, and encourage – or require – sponsors to gather more information in a standardized way across these different arms of the human immune system,” Dr. Levy said. “So we keep learning and keep doing this better.”
In recapping the panel’s suggestions at the end of the meeting, Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research, addressed the requests made during the day’s meeting about better data on how the vaccines work.
“We heard loud and clear that we need to use a data-driven approach to get to the simplest possible scheme that we can for vaccination,” Dr. Marks said. “And it should be as simple as possible but not oversimplified, a little bit like they say about Mozart’s music.”
A version of this article first appeared on WebMD.com.
The FDA is looking to give clearer direction to vaccine makers about future development of COVID-19 vaccines. The plan is to narrow down the current complex landscape of options for vaccinations, and thus help increase use of these shots.
COVID remains a serious threat, causing about 4,000 deaths a week recently, according to the Centers for Disease Control and Prevention. The 21 members of the Vaccines and Related Biological Products Advisory Committee (VRBPAC) on Jan. 26 voted unanimously “yes” on a single question posed by the FDA:
“Does the committee recommend harmonizing the vaccine strain composition of primary series and booster doses in the U.S. to a single composition, e.g., the composition for all vaccines administered currently would be a bivalent vaccine (Original plus Omicron BA.4/BA.5)?”
In other words, would it be better to have one vaccine potentially combining multiple strains of the virus, instead of multiple vaccines – such as a two-shot primary series then a booster containing different combinations of viral strains.
The FDA will consider the panel’s advice as it outlines new strategies for keeping ahead of the evolving virus.
In explaining their support for the FDA plan, panel members said they hoped that a simpler regime would aid in persuading more people to get COVID vaccines.
Pamela McInnes, DDS, MSc, noted that it’s difficult to explain to many people that the vaccine works to protect them from more severe illness if they contract COVID after getting vaccinated.
“That is a real challenge,” said Dr. McInness, retired deputy director of the National Center for Advancing Translational Sciences at the National Institutes of Health.
“The message that you would have gotten more sick and landed in the hospital resonates with me, but I’m not sure if it resonates with” many people who become infected, she said.
The plan
In the briefing document for the meeting, the FDA outlined a plan for transitioning from the current complex landscape of COVID-19 vaccines to a single vaccine composition for the primary series and booster vaccination.
This would require harmonizing the strain composition of all COVID-19 vaccines; simplifying the immunization schedule for future vaccination campaigns to administer a two-dose series in certain young children and in older adults and persons with compromised immunity, and only one dose in all other individuals; and establishing a process for vaccine strain selection recommendations, similar in many ways to that used for seasonal influenza vaccines, based on prevailing and predicted variants that would take place by June to allow for vaccine production by September.
During the discussion, though, questions arose about the June target date. Given the production schedule for some vaccines, that date might need to shift, said Jerry Weir, PhD, director of the division of viral products at FDA’s Center for Biologics Evaluation and Research.
“We’re all just going to have to maintain flexibility,” Dr. Weir said, adding that there is not yet a “good pattern” established for updating these vaccines.
Increasing vaccination rates
There was broad consensus about the need to boost public support for COVID-19 vaccinations. While about 81% of the U.S. population has had at least one dose of this vaccine, only 15.3% have had an updated bivalent booster dose, according to the CDC.
“Anything that results in better public communication would be extremely valuable,” said committee member Henry H. Bernstein, DO, MHCM, of the Zucker School of Medicine at Hofstra/Northwell Health in Hempstead, N.Y.
But it’s unclear what expectations will be prioritized for the COVID vaccine program, he said.
“Realistically, I don’t think we can have it all – less infection, less transmission, less severe disease, and less long COVID,” Dr. Bernstein said. “And that seems to be a major challenge for public messaging.”
Panelists press for more data
Other committee members also pressed for clearer targets in evaluating the goals for COVID vaccines, and for more robust data.
Like his fellow VRBPAC members, Cody Meissner, MD, of Dartmouth’s Geisel School of Medicine, Hanover, N.H., supported a move toward harmonizing the strains used in different companies’ vaccines. But he added that it wasn’t clear yet how frequently they should be administered.
“We need to see what happens with disease burden,” Dr. Meissner said. “We may or may not need annual vaccination. It’s just awfully early, it seems to me, in this process to answer that question.”
Among those serving on VRBPAC was one of the FDA’s more vocal critics on these points, Paul A. Offit, MD, a vaccine expert from Children’s Hospital of Philadelphia. Dr. Offit, for example, joined former FDA officials in writing a November opinion article for the Washington Post, arguing that the evidence for boosters for healthy younger adults was not strong.
At the Jan. 26 meeting, he supported the drive toward simplification of COVID vaccine schedules, while arguing for more data about how well these products are working.
“This virus is going to be with us for years, if not decades, and there will always be vulnerable groups who are going to be hospitalized and killed by the virus,” Dr. Offit said.
The CDC needs to provide more information about the characteristics of people being hospitalized with COVID infections, including their ages and comorbidities as well as details about their vaccine history, he said. In addition, academic researchers should provide a clearer picture of what immunological predictors are at play in increasing people’s risk from COVID.
“Then and only then can we really best make the decision about who gets vaccinated with what and when,” Dr. Offit said.
VRBPAC member Ofer Levy, MD, PhD, also urged the FDA to press for a collection of more robust and detailed information about the immune response to COVID-19 vaccinations, such as a deeper look at what’s happening with antibodies.
“I hope FDA will continue to reflect on how to best take this information forward, and encourage – or require – sponsors to gather more information in a standardized way across these different arms of the human immune system,” Dr. Levy said. “So we keep learning and keep doing this better.”
In recapping the panel’s suggestions at the end of the meeting, Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research, addressed the requests made during the day’s meeting about better data on how the vaccines work.
“We heard loud and clear that we need to use a data-driven approach to get to the simplest possible scheme that we can for vaccination,” Dr. Marks said. “And it should be as simple as possible but not oversimplified, a little bit like they say about Mozart’s music.”
A version of this article first appeared on WebMD.com.
The FDA is looking to give clearer direction to vaccine makers about future development of COVID-19 vaccines. The plan is to narrow down the current complex landscape of options for vaccinations, and thus help increase use of these shots.
COVID remains a serious threat, causing about 4,000 deaths a week recently, according to the Centers for Disease Control and Prevention. The 21 members of the Vaccines and Related Biological Products Advisory Committee (VRBPAC) on Jan. 26 voted unanimously “yes” on a single question posed by the FDA:
“Does the committee recommend harmonizing the vaccine strain composition of primary series and booster doses in the U.S. to a single composition, e.g., the composition for all vaccines administered currently would be a bivalent vaccine (Original plus Omicron BA.4/BA.5)?”
In other words, would it be better to have one vaccine potentially combining multiple strains of the virus, instead of multiple vaccines – such as a two-shot primary series then a booster containing different combinations of viral strains.
The FDA will consider the panel’s advice as it outlines new strategies for keeping ahead of the evolving virus.
In explaining their support for the FDA plan, panel members said they hoped that a simpler regime would aid in persuading more people to get COVID vaccines.
Pamela McInnes, DDS, MSc, noted that it’s difficult to explain to many people that the vaccine works to protect them from more severe illness if they contract COVID after getting vaccinated.
“That is a real challenge,” said Dr. McInness, retired deputy director of the National Center for Advancing Translational Sciences at the National Institutes of Health.
“The message that you would have gotten more sick and landed in the hospital resonates with me, but I’m not sure if it resonates with” many people who become infected, she said.
The plan
In the briefing document for the meeting, the FDA outlined a plan for transitioning from the current complex landscape of COVID-19 vaccines to a single vaccine composition for the primary series and booster vaccination.
This would require harmonizing the strain composition of all COVID-19 vaccines; simplifying the immunization schedule for future vaccination campaigns to administer a two-dose series in certain young children and in older adults and persons with compromised immunity, and only one dose in all other individuals; and establishing a process for vaccine strain selection recommendations, similar in many ways to that used for seasonal influenza vaccines, based on prevailing and predicted variants that would take place by June to allow for vaccine production by September.
During the discussion, though, questions arose about the June target date. Given the production schedule for some vaccines, that date might need to shift, said Jerry Weir, PhD, director of the division of viral products at FDA’s Center for Biologics Evaluation and Research.
“We’re all just going to have to maintain flexibility,” Dr. Weir said, adding that there is not yet a “good pattern” established for updating these vaccines.
Increasing vaccination rates
There was broad consensus about the need to boost public support for COVID-19 vaccinations. While about 81% of the U.S. population has had at least one dose of this vaccine, only 15.3% have had an updated bivalent booster dose, according to the CDC.
“Anything that results in better public communication would be extremely valuable,” said committee member Henry H. Bernstein, DO, MHCM, of the Zucker School of Medicine at Hofstra/Northwell Health in Hempstead, N.Y.
But it’s unclear what expectations will be prioritized for the COVID vaccine program, he said.
“Realistically, I don’t think we can have it all – less infection, less transmission, less severe disease, and less long COVID,” Dr. Bernstein said. “And that seems to be a major challenge for public messaging.”
Panelists press for more data
Other committee members also pressed for clearer targets in evaluating the goals for COVID vaccines, and for more robust data.
Like his fellow VRBPAC members, Cody Meissner, MD, of Dartmouth’s Geisel School of Medicine, Hanover, N.H., supported a move toward harmonizing the strains used in different companies’ vaccines. But he added that it wasn’t clear yet how frequently they should be administered.
“We need to see what happens with disease burden,” Dr. Meissner said. “We may or may not need annual vaccination. It’s just awfully early, it seems to me, in this process to answer that question.”
Among those serving on VRBPAC was one of the FDA’s more vocal critics on these points, Paul A. Offit, MD, a vaccine expert from Children’s Hospital of Philadelphia. Dr. Offit, for example, joined former FDA officials in writing a November opinion article for the Washington Post, arguing that the evidence for boosters for healthy younger adults was not strong.
At the Jan. 26 meeting, he supported the drive toward simplification of COVID vaccine schedules, while arguing for more data about how well these products are working.
“This virus is going to be with us for years, if not decades, and there will always be vulnerable groups who are going to be hospitalized and killed by the virus,” Dr. Offit said.
The CDC needs to provide more information about the characteristics of people being hospitalized with COVID infections, including their ages and comorbidities as well as details about their vaccine history, he said. In addition, academic researchers should provide a clearer picture of what immunological predictors are at play in increasing people’s risk from COVID.
“Then and only then can we really best make the decision about who gets vaccinated with what and when,” Dr. Offit said.
VRBPAC member Ofer Levy, MD, PhD, also urged the FDA to press for a collection of more robust and detailed information about the immune response to COVID-19 vaccinations, such as a deeper look at what’s happening with antibodies.
“I hope FDA will continue to reflect on how to best take this information forward, and encourage – or require – sponsors to gather more information in a standardized way across these different arms of the human immune system,” Dr. Levy said. “So we keep learning and keep doing this better.”
In recapping the panel’s suggestions at the end of the meeting, Peter Marks, MD, PhD, the director of the FDA’s Center for Biologics Evaluation and Research, addressed the requests made during the day’s meeting about better data on how the vaccines work.
“We heard loud and clear that we need to use a data-driven approach to get to the simplest possible scheme that we can for vaccination,” Dr. Marks said. “And it should be as simple as possible but not oversimplified, a little bit like they say about Mozart’s music.”
A version of this article first appeared on WebMD.com.
Q&A with long COVID patient-researcher: Treatments lagging as cases rise
For Julia Moore Vogel, PhD, a cup of peppermint tea marked the moment her life would change forever.
One morning in early July 2020, she took a sip of her favorite strongly flavored pick-me-up and couldn’t taste it. She knew loss of taste and smell were symptoms of COVID-19, and she suspected she had contracted the virus. A doctor’s visit confirmed her fears.
“I remember trying the tea and just being so shocked and thinking: How can this be happening to me?” said Dr. Moore Vogel, a COVID-19 researcher with the Scripps Research Translational Institute in San Diego. “I’d been so incredibly careful.”
Her physician assured her that as a healthy woman in her mid-30s, she’d be “back to normal” in 2 weeks’ time and that her loss of taste and smell “very likely will be your only symptom,” she recalled.
. Now, 2½ years later, Dr. Moore Vogel is among the tens of millions of Americans with long COVID.
As a COVID-19 patient-researcher who still struggles with fatigue and migraines, she has learned to cope with her condition. She directs the Participant Center for the All of Us Research Program, a National Institutes of Health collaboration to build the largest, most diverse health database in history. She relies on a practice called pacing, which helps conserve physical, mental, and emotional energy, to avoid making her symptoms worse.
And she is a coauthor of a landmark 200-study review of long COVID published Jan. 13 in the journal Nature, with Scripps Executive Vice President and Medscape Editor-in-Chief Eric Topol, MD. Two other institute long COVID researchers and patient advocates who have the condition coauthored the review – Lisa McCorkell and Hannah E. Davis, cofounders of the Patient-Led Research Collaborative , a group of long COVID patients who study the virus.
Dr. Moore Vogel discussed the key findings of the new review and her personal experiences with this news organization.
Q: When you contracted COVID, no treatments or vaccines existed. Can you talk about what the experience was like for you?
A: “It was July 2020. The loss of taste and smell was the first symptom, and what was interesting was that was my only symptom for a little bit. Being the goal-oriented, work-oriented person that I am, I just worked the rest of the week and hoped that it wasn’t real.
“But that was a Wednesday, and by Friday, I was just getting really tired, and it was really hard to finish my workday. I ended up taking 3 weeks off to recover from the acute phase. At the time, I had read early discussions about long COVID, and it was always on my [mind] – how long was it going to take to recover?”
Q: You went to see a doctor that first week?
A: “I called them when I had the loss of taste and smell, and they said, ‘It’s very likely this will be your only symptom.’ And when I first talked to a physician, they were saying, ‘Oh, you’re young and healthy, in 2 weeks you’ll be back to normal.’ But of course that turned out not to be true.
“It’s hard to remember what it was like at that time. There were so few treatments, it was all about rationing ventilators, and it was absolutely terrifying at the time to just not know what was going to happen.”
Q: How are you managing your condition today?
A: “I have myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), one of the really common diagnoses that come out of long COVID. So, that’s the biggest thing for me to manage now, and the main management is pacing.
“I also have medications for migraine management. I’ve always had some degree of migraines, like 2 a month, but now it’s like 15 a month, so it’s a totally different scale of management.”
Q: It must be frustrating, personally and professionally, that long COVID treatments remain elusive.
A: “I’m disappointed at the pace of testing things in clinical trials. There was so much progress made, so much innovation in the early stages of the pandemic to treat the acute phase, and it led to amazing things. We have all these monoclonal antibodies, the steroids are really effective, not to mention the antivirals and the vaccines, of course, on the prevention side. It’s been amazing.
“But for some reason, long COVID treatment is really lagging. What I hypothesize as part of the reason for that is that it doesn’t feel as dramatic. When you see someone on a ventilator or hear about death, it feels very dramatic, and people really worry about that.”
Q: So, let’s talk about the research. How did your personal experiences – and those of the two other coauthors with long COVID – help inform this review?
A: “I work with Eric Topol on a regular basis, and it was amazing that he invited patients to work with him on this review ... I have to say of my other long COVID patient coauthors, Hannah Davis and Lisa McCorkell, it was amazing to work with them.
“It was my first time working with people who have long COVID on a big project. The understanding that we had of each other [where] one of us might say, Oh, I’m crashing today, I can’t work on this. Can you help get us across the finish line for this deadline? That was really amazing to me in terms of how a workplace can be with real disability accommodations.
“It’s really changed my personal outlook on how important it is to have patients involved in the process.”
Q: What was the most surprising or significant finding of the review, in your view?
A: “I would say the most impactful thing to me in the process of writing this review is how much research has been done in such a short time. We started with over 250 studies that we wanted to reference in the review, and we actually had to cut out 50 in the editorial process, which was really hard!
“There’s just been so much progress that’s been made in the past couple of years. And then thinking about the progress on long COVID in general, the other things that’s important to acknowledge is all the work that’s been done on other postviral illnesses that present very similarly to long COVID in many patients, ME/CFS, and postural orthostatic tachycardia syndrome (POTS).”
Q: One thing that stood out is the review’s finding that long COVID is potentially lifelong COVID and, in some ways, is closer to HIV-AIDS than, say, influenza. Is that right?
A: “Yes. I’m really glad you took that point away from the review because that was one of the things that I felt the most strongly about incorporating. For many people, based on the treatments that we have today, this is likely to lead to lifelong disability. And that’s something, from my personal experience, for sure. I’m seeing no improvement on the horizon.
“That’s part of why I’m so passionate about there being clinical trials because I know there are millions and millions of us. So for me, that wasn’t so surprising, because I’m living it, but I can see how for the general public that was a really surprising finding.”
Q: The review breaks down long COVID’s effects on various organs/systems, and it includes the most comprehensive look to date at the effects on pregnant women. Anything you’d care to stress about that?
A: “It really underlies the importance of vaccination, given that it can affect both the pregnant person and child. There is early evidence of development delays if there’s infection while the child is still gestating. So, I think it underscores the need for vaccination to reduce that risk.
“You know, pregnancy is a stressful and terrifying time anyway. So, if there’s anything you can do to reduce the risk to yourself and your unborn child, I think it’s really worthwhile.”
Q: Why do you think this exhaustive review was needed?
A: “Because of the massive amount of literature that’s out there, it’s so hard for anybody to sift through. Eric Topol and Hannah Davis, two of the coauthors, are two people who have done it, and they keep up with all the literature, and they are always tweeting about it.
“But most people don’t have the time to be able to sift through it, so what we did was take all of that literature, organize it into sections, and summarize the key findings. Then the other thing that I think is really important for the field right now is the recommendations piece.”
Q: What impact do you think the new long COVID review in Nature will have?
A: “The response to our review is way beyond what I expected, and I think that’s in part a sign that there is growing awareness of the issue of long COVID.
“I hope that helps spiral toward more treatment trials. Because there are a lot of great candidates out there. We have a whole table in the review about the different potential treatments that should be tested.”
Q: What’s the take-home message for physicians?
A: “One of the key recommendations is about physician education. We know that it is so hard for physicians to keep up with this massive amount of literature, and we really need more physician education that’s meant for busy physicians who really don’t have time to read all of the primary literature themselves.
“So many folks are not getting the care that they need. Because these types of conditions haven’t been seen as much by primary care providers, physical therapists, etc., there’s so much more education that’s needed.
“I think the basic tenets probably could be taught in a weekend course, [including] listening to the patients, believing patients. There are so many times patient symptoms are [dismissed and not] really being taken seriously by their physicians.
“I think part of the challenge behind that is the conflating of mental health issues with these other physiological symptoms. There’s a tendency to say, ‘Oh, all this is this caused by mental health issues’ and that mental health is the root cause, when actually it’s the illness that’s the root cause.”
Q: What’s the big picture: How significant is the public health crisis that long COVID represents?
A: “I believe it’s a massive crisis, a massive emergency. A lot of people in the long COVID community are calling it a mass-disabling event. There is concern that if we let the pandemic run unmitigated for long enough – given that we expect about 10% of folks that get COVID will end up with long COVID – we could end up eventually with a majority-disabled society.
“That would be devasting – to individuals, to the economy, the medical system. So, it’s absolutely a public health emergency in my view, and that’s part of why I’ve been so surprised by the lack of trials, the lack of awareness in the public. There hasn’t been as much public education about long COVID as there has about acute COVID. I think we can do more from a public health perspective.”
Q: What are the main challenges in combating long COVID?
A: “I think the lack of treatments is the most devastating part because it’s such a hard disease to contract, and there’s no end in sight, and so that time horizon can be really difficult. That’s part of why I’m pushing the treatments so much, because I want to offer hope to the community, you know, I want there to be hope around the corner.
“My hope is that within 5 years we’ll have treatments that can really improve quality of life for the community. And I know that that may seem like a long time for those who are suffering, and I hope that there will be some clinical trials of treatments that improve symptom management within 1-2 years. But I think for really more novel things, it’s really going to take at least 5.”
Q: Any advice you’d give to someone with long COVID today?
A: “Connecting with others that are going through the experience is extremely valuable and can really help with that mental component which can be really draining.
“The other thing, in terms of what’s important for the lives of people who are living with long COVID, I would say to everyone who doesn’t have long COVID but knows someone who does, being able to offer support is crucial and can make such a difference in quality of life.
“It is really crucial, for those who don’t have long COVID, to take it into account when you’re making your risk calculations. When you’re thinking: Am I going to wear a mask here? or Am I going to go to that bar?
“Really consider the possibility that if you get COVID, you have a 10% chance of getting long COVID. And if you get long COVID, you have a 25% chance of not being able to work anymore or being so ill that you can’t work anymore and you may lose your health insurance.
“The compounding effects are absolutely devastating, and I think that’s under-factored-in to the general risk calculations of the public.”
A version of this article first appeared on Medscape.com.
For Julia Moore Vogel, PhD, a cup of peppermint tea marked the moment her life would change forever.
One morning in early July 2020, she took a sip of her favorite strongly flavored pick-me-up and couldn’t taste it. She knew loss of taste and smell were symptoms of COVID-19, and she suspected she had contracted the virus. A doctor’s visit confirmed her fears.
“I remember trying the tea and just being so shocked and thinking: How can this be happening to me?” said Dr. Moore Vogel, a COVID-19 researcher with the Scripps Research Translational Institute in San Diego. “I’d been so incredibly careful.”
Her physician assured her that as a healthy woman in her mid-30s, she’d be “back to normal” in 2 weeks’ time and that her loss of taste and smell “very likely will be your only symptom,” she recalled.
. Now, 2½ years later, Dr. Moore Vogel is among the tens of millions of Americans with long COVID.
As a COVID-19 patient-researcher who still struggles with fatigue and migraines, she has learned to cope with her condition. She directs the Participant Center for the All of Us Research Program, a National Institutes of Health collaboration to build the largest, most diverse health database in history. She relies on a practice called pacing, which helps conserve physical, mental, and emotional energy, to avoid making her symptoms worse.
And she is a coauthor of a landmark 200-study review of long COVID published Jan. 13 in the journal Nature, with Scripps Executive Vice President and Medscape Editor-in-Chief Eric Topol, MD. Two other institute long COVID researchers and patient advocates who have the condition coauthored the review – Lisa McCorkell and Hannah E. Davis, cofounders of the Patient-Led Research Collaborative , a group of long COVID patients who study the virus.
Dr. Moore Vogel discussed the key findings of the new review and her personal experiences with this news organization.
Q: When you contracted COVID, no treatments or vaccines existed. Can you talk about what the experience was like for you?
A: “It was July 2020. The loss of taste and smell was the first symptom, and what was interesting was that was my only symptom for a little bit. Being the goal-oriented, work-oriented person that I am, I just worked the rest of the week and hoped that it wasn’t real.
“But that was a Wednesday, and by Friday, I was just getting really tired, and it was really hard to finish my workday. I ended up taking 3 weeks off to recover from the acute phase. At the time, I had read early discussions about long COVID, and it was always on my [mind] – how long was it going to take to recover?”
Q: You went to see a doctor that first week?
A: “I called them when I had the loss of taste and smell, and they said, ‘It’s very likely this will be your only symptom.’ And when I first talked to a physician, they were saying, ‘Oh, you’re young and healthy, in 2 weeks you’ll be back to normal.’ But of course that turned out not to be true.
“It’s hard to remember what it was like at that time. There were so few treatments, it was all about rationing ventilators, and it was absolutely terrifying at the time to just not know what was going to happen.”
Q: How are you managing your condition today?
A: “I have myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), one of the really common diagnoses that come out of long COVID. So, that’s the biggest thing for me to manage now, and the main management is pacing.
“I also have medications for migraine management. I’ve always had some degree of migraines, like 2 a month, but now it’s like 15 a month, so it’s a totally different scale of management.”
Q: It must be frustrating, personally and professionally, that long COVID treatments remain elusive.
A: “I’m disappointed at the pace of testing things in clinical trials. There was so much progress made, so much innovation in the early stages of the pandemic to treat the acute phase, and it led to amazing things. We have all these monoclonal antibodies, the steroids are really effective, not to mention the antivirals and the vaccines, of course, on the prevention side. It’s been amazing.
“But for some reason, long COVID treatment is really lagging. What I hypothesize as part of the reason for that is that it doesn’t feel as dramatic. When you see someone on a ventilator or hear about death, it feels very dramatic, and people really worry about that.”
Q: So, let’s talk about the research. How did your personal experiences – and those of the two other coauthors with long COVID – help inform this review?
A: “I work with Eric Topol on a regular basis, and it was amazing that he invited patients to work with him on this review ... I have to say of my other long COVID patient coauthors, Hannah Davis and Lisa McCorkell, it was amazing to work with them.
“It was my first time working with people who have long COVID on a big project. The understanding that we had of each other [where] one of us might say, Oh, I’m crashing today, I can’t work on this. Can you help get us across the finish line for this deadline? That was really amazing to me in terms of how a workplace can be with real disability accommodations.
“It’s really changed my personal outlook on how important it is to have patients involved in the process.”
Q: What was the most surprising or significant finding of the review, in your view?
A: “I would say the most impactful thing to me in the process of writing this review is how much research has been done in such a short time. We started with over 250 studies that we wanted to reference in the review, and we actually had to cut out 50 in the editorial process, which was really hard!
“There’s just been so much progress that’s been made in the past couple of years. And then thinking about the progress on long COVID in general, the other things that’s important to acknowledge is all the work that’s been done on other postviral illnesses that present very similarly to long COVID in many patients, ME/CFS, and postural orthostatic tachycardia syndrome (POTS).”
Q: One thing that stood out is the review’s finding that long COVID is potentially lifelong COVID and, in some ways, is closer to HIV-AIDS than, say, influenza. Is that right?
A: “Yes. I’m really glad you took that point away from the review because that was one of the things that I felt the most strongly about incorporating. For many people, based on the treatments that we have today, this is likely to lead to lifelong disability. And that’s something, from my personal experience, for sure. I’m seeing no improvement on the horizon.
“That’s part of why I’m so passionate about there being clinical trials because I know there are millions and millions of us. So for me, that wasn’t so surprising, because I’m living it, but I can see how for the general public that was a really surprising finding.”
Q: The review breaks down long COVID’s effects on various organs/systems, and it includes the most comprehensive look to date at the effects on pregnant women. Anything you’d care to stress about that?
A: “It really underlies the importance of vaccination, given that it can affect both the pregnant person and child. There is early evidence of development delays if there’s infection while the child is still gestating. So, I think it underscores the need for vaccination to reduce that risk.
“You know, pregnancy is a stressful and terrifying time anyway. So, if there’s anything you can do to reduce the risk to yourself and your unborn child, I think it’s really worthwhile.”
Q: Why do you think this exhaustive review was needed?
A: “Because of the massive amount of literature that’s out there, it’s so hard for anybody to sift through. Eric Topol and Hannah Davis, two of the coauthors, are two people who have done it, and they keep up with all the literature, and they are always tweeting about it.
“But most people don’t have the time to be able to sift through it, so what we did was take all of that literature, organize it into sections, and summarize the key findings. Then the other thing that I think is really important for the field right now is the recommendations piece.”
Q: What impact do you think the new long COVID review in Nature will have?
A: “The response to our review is way beyond what I expected, and I think that’s in part a sign that there is growing awareness of the issue of long COVID.
“I hope that helps spiral toward more treatment trials. Because there are a lot of great candidates out there. We have a whole table in the review about the different potential treatments that should be tested.”
Q: What’s the take-home message for physicians?
A: “One of the key recommendations is about physician education. We know that it is so hard for physicians to keep up with this massive amount of literature, and we really need more physician education that’s meant for busy physicians who really don’t have time to read all of the primary literature themselves.
“So many folks are not getting the care that they need. Because these types of conditions haven’t been seen as much by primary care providers, physical therapists, etc., there’s so much more education that’s needed.
“I think the basic tenets probably could be taught in a weekend course, [including] listening to the patients, believing patients. There are so many times patient symptoms are [dismissed and not] really being taken seriously by their physicians.
“I think part of the challenge behind that is the conflating of mental health issues with these other physiological symptoms. There’s a tendency to say, ‘Oh, all this is this caused by mental health issues’ and that mental health is the root cause, when actually it’s the illness that’s the root cause.”
Q: What’s the big picture: How significant is the public health crisis that long COVID represents?
A: “I believe it’s a massive crisis, a massive emergency. A lot of people in the long COVID community are calling it a mass-disabling event. There is concern that if we let the pandemic run unmitigated for long enough – given that we expect about 10% of folks that get COVID will end up with long COVID – we could end up eventually with a majority-disabled society.
“That would be devasting – to individuals, to the economy, the medical system. So, it’s absolutely a public health emergency in my view, and that’s part of why I’ve been so surprised by the lack of trials, the lack of awareness in the public. There hasn’t been as much public education about long COVID as there has about acute COVID. I think we can do more from a public health perspective.”
Q: What are the main challenges in combating long COVID?
A: “I think the lack of treatments is the most devastating part because it’s such a hard disease to contract, and there’s no end in sight, and so that time horizon can be really difficult. That’s part of why I’m pushing the treatments so much, because I want to offer hope to the community, you know, I want there to be hope around the corner.
“My hope is that within 5 years we’ll have treatments that can really improve quality of life for the community. And I know that that may seem like a long time for those who are suffering, and I hope that there will be some clinical trials of treatments that improve symptom management within 1-2 years. But I think for really more novel things, it’s really going to take at least 5.”
Q: Any advice you’d give to someone with long COVID today?
A: “Connecting with others that are going through the experience is extremely valuable and can really help with that mental component which can be really draining.
“The other thing, in terms of what’s important for the lives of people who are living with long COVID, I would say to everyone who doesn’t have long COVID but knows someone who does, being able to offer support is crucial and can make such a difference in quality of life.
“It is really crucial, for those who don’t have long COVID, to take it into account when you’re making your risk calculations. When you’re thinking: Am I going to wear a mask here? or Am I going to go to that bar?
“Really consider the possibility that if you get COVID, you have a 10% chance of getting long COVID. And if you get long COVID, you have a 25% chance of not being able to work anymore or being so ill that you can’t work anymore and you may lose your health insurance.
“The compounding effects are absolutely devastating, and I think that’s under-factored-in to the general risk calculations of the public.”
A version of this article first appeared on Medscape.com.
For Julia Moore Vogel, PhD, a cup of peppermint tea marked the moment her life would change forever.
One morning in early July 2020, she took a sip of her favorite strongly flavored pick-me-up and couldn’t taste it. She knew loss of taste and smell were symptoms of COVID-19, and she suspected she had contracted the virus. A doctor’s visit confirmed her fears.
“I remember trying the tea and just being so shocked and thinking: How can this be happening to me?” said Dr. Moore Vogel, a COVID-19 researcher with the Scripps Research Translational Institute in San Diego. “I’d been so incredibly careful.”
Her physician assured her that as a healthy woman in her mid-30s, she’d be “back to normal” in 2 weeks’ time and that her loss of taste and smell “very likely will be your only symptom,” she recalled.
. Now, 2½ years later, Dr. Moore Vogel is among the tens of millions of Americans with long COVID.
As a COVID-19 patient-researcher who still struggles with fatigue and migraines, she has learned to cope with her condition. She directs the Participant Center for the All of Us Research Program, a National Institutes of Health collaboration to build the largest, most diverse health database in history. She relies on a practice called pacing, which helps conserve physical, mental, and emotional energy, to avoid making her symptoms worse.
And she is a coauthor of a landmark 200-study review of long COVID published Jan. 13 in the journal Nature, with Scripps Executive Vice President and Medscape Editor-in-Chief Eric Topol, MD. Two other institute long COVID researchers and patient advocates who have the condition coauthored the review – Lisa McCorkell and Hannah E. Davis, cofounders of the Patient-Led Research Collaborative , a group of long COVID patients who study the virus.
Dr. Moore Vogel discussed the key findings of the new review and her personal experiences with this news organization.
Q: When you contracted COVID, no treatments or vaccines existed. Can you talk about what the experience was like for you?
A: “It was July 2020. The loss of taste and smell was the first symptom, and what was interesting was that was my only symptom for a little bit. Being the goal-oriented, work-oriented person that I am, I just worked the rest of the week and hoped that it wasn’t real.
“But that was a Wednesday, and by Friday, I was just getting really tired, and it was really hard to finish my workday. I ended up taking 3 weeks off to recover from the acute phase. At the time, I had read early discussions about long COVID, and it was always on my [mind] – how long was it going to take to recover?”
Q: You went to see a doctor that first week?
A: “I called them when I had the loss of taste and smell, and they said, ‘It’s very likely this will be your only symptom.’ And when I first talked to a physician, they were saying, ‘Oh, you’re young and healthy, in 2 weeks you’ll be back to normal.’ But of course that turned out not to be true.
“It’s hard to remember what it was like at that time. There were so few treatments, it was all about rationing ventilators, and it was absolutely terrifying at the time to just not know what was going to happen.”
Q: How are you managing your condition today?
A: “I have myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), one of the really common diagnoses that come out of long COVID. So, that’s the biggest thing for me to manage now, and the main management is pacing.
“I also have medications for migraine management. I’ve always had some degree of migraines, like 2 a month, but now it’s like 15 a month, so it’s a totally different scale of management.”
Q: It must be frustrating, personally and professionally, that long COVID treatments remain elusive.
A: “I’m disappointed at the pace of testing things in clinical trials. There was so much progress made, so much innovation in the early stages of the pandemic to treat the acute phase, and it led to amazing things. We have all these monoclonal antibodies, the steroids are really effective, not to mention the antivirals and the vaccines, of course, on the prevention side. It’s been amazing.
“But for some reason, long COVID treatment is really lagging. What I hypothesize as part of the reason for that is that it doesn’t feel as dramatic. When you see someone on a ventilator or hear about death, it feels very dramatic, and people really worry about that.”
Q: So, let’s talk about the research. How did your personal experiences – and those of the two other coauthors with long COVID – help inform this review?
A: “I work with Eric Topol on a regular basis, and it was amazing that he invited patients to work with him on this review ... I have to say of my other long COVID patient coauthors, Hannah Davis and Lisa McCorkell, it was amazing to work with them.
“It was my first time working with people who have long COVID on a big project. The understanding that we had of each other [where] one of us might say, Oh, I’m crashing today, I can’t work on this. Can you help get us across the finish line for this deadline? That was really amazing to me in terms of how a workplace can be with real disability accommodations.
“It’s really changed my personal outlook on how important it is to have patients involved in the process.”
Q: What was the most surprising or significant finding of the review, in your view?
A: “I would say the most impactful thing to me in the process of writing this review is how much research has been done in such a short time. We started with over 250 studies that we wanted to reference in the review, and we actually had to cut out 50 in the editorial process, which was really hard!
“There’s just been so much progress that’s been made in the past couple of years. And then thinking about the progress on long COVID in general, the other things that’s important to acknowledge is all the work that’s been done on other postviral illnesses that present very similarly to long COVID in many patients, ME/CFS, and postural orthostatic tachycardia syndrome (POTS).”
Q: One thing that stood out is the review’s finding that long COVID is potentially lifelong COVID and, in some ways, is closer to HIV-AIDS than, say, influenza. Is that right?
A: “Yes. I’m really glad you took that point away from the review because that was one of the things that I felt the most strongly about incorporating. For many people, based on the treatments that we have today, this is likely to lead to lifelong disability. And that’s something, from my personal experience, for sure. I’m seeing no improvement on the horizon.
“That’s part of why I’m so passionate about there being clinical trials because I know there are millions and millions of us. So for me, that wasn’t so surprising, because I’m living it, but I can see how for the general public that was a really surprising finding.”
Q: The review breaks down long COVID’s effects on various organs/systems, and it includes the most comprehensive look to date at the effects on pregnant women. Anything you’d care to stress about that?
A: “It really underlies the importance of vaccination, given that it can affect both the pregnant person and child. There is early evidence of development delays if there’s infection while the child is still gestating. So, I think it underscores the need for vaccination to reduce that risk.
“You know, pregnancy is a stressful and terrifying time anyway. So, if there’s anything you can do to reduce the risk to yourself and your unborn child, I think it’s really worthwhile.”
Q: Why do you think this exhaustive review was needed?
A: “Because of the massive amount of literature that’s out there, it’s so hard for anybody to sift through. Eric Topol and Hannah Davis, two of the coauthors, are two people who have done it, and they keep up with all the literature, and they are always tweeting about it.
“But most people don’t have the time to be able to sift through it, so what we did was take all of that literature, organize it into sections, and summarize the key findings. Then the other thing that I think is really important for the field right now is the recommendations piece.”
Q: What impact do you think the new long COVID review in Nature will have?
A: “The response to our review is way beyond what I expected, and I think that’s in part a sign that there is growing awareness of the issue of long COVID.
“I hope that helps spiral toward more treatment trials. Because there are a lot of great candidates out there. We have a whole table in the review about the different potential treatments that should be tested.”
Q: What’s the take-home message for physicians?
A: “One of the key recommendations is about physician education. We know that it is so hard for physicians to keep up with this massive amount of literature, and we really need more physician education that’s meant for busy physicians who really don’t have time to read all of the primary literature themselves.
“So many folks are not getting the care that they need. Because these types of conditions haven’t been seen as much by primary care providers, physical therapists, etc., there’s so much more education that’s needed.
“I think the basic tenets probably could be taught in a weekend course, [including] listening to the patients, believing patients. There are so many times patient symptoms are [dismissed and not] really being taken seriously by their physicians.
“I think part of the challenge behind that is the conflating of mental health issues with these other physiological symptoms. There’s a tendency to say, ‘Oh, all this is this caused by mental health issues’ and that mental health is the root cause, when actually it’s the illness that’s the root cause.”
Q: What’s the big picture: How significant is the public health crisis that long COVID represents?
A: “I believe it’s a massive crisis, a massive emergency. A lot of people in the long COVID community are calling it a mass-disabling event. There is concern that if we let the pandemic run unmitigated for long enough – given that we expect about 10% of folks that get COVID will end up with long COVID – we could end up eventually with a majority-disabled society.
“That would be devasting – to individuals, to the economy, the medical system. So, it’s absolutely a public health emergency in my view, and that’s part of why I’ve been so surprised by the lack of trials, the lack of awareness in the public. There hasn’t been as much public education about long COVID as there has about acute COVID. I think we can do more from a public health perspective.”
Q: What are the main challenges in combating long COVID?
A: “I think the lack of treatments is the most devastating part because it’s such a hard disease to contract, and there’s no end in sight, and so that time horizon can be really difficult. That’s part of why I’m pushing the treatments so much, because I want to offer hope to the community, you know, I want there to be hope around the corner.
“My hope is that within 5 years we’ll have treatments that can really improve quality of life for the community. And I know that that may seem like a long time for those who are suffering, and I hope that there will be some clinical trials of treatments that improve symptom management within 1-2 years. But I think for really more novel things, it’s really going to take at least 5.”
Q: Any advice you’d give to someone with long COVID today?
A: “Connecting with others that are going through the experience is extremely valuable and can really help with that mental component which can be really draining.
“The other thing, in terms of what’s important for the lives of people who are living with long COVID, I would say to everyone who doesn’t have long COVID but knows someone who does, being able to offer support is crucial and can make such a difference in quality of life.
“It is really crucial, for those who don’t have long COVID, to take it into account when you’re making your risk calculations. When you’re thinking: Am I going to wear a mask here? or Am I going to go to that bar?
“Really consider the possibility that if you get COVID, you have a 10% chance of getting long COVID. And if you get long COVID, you have a 25% chance of not being able to work anymore or being so ill that you can’t work anymore and you may lose your health insurance.
“The compounding effects are absolutely devastating, and I think that’s under-factored-in to the general risk calculations of the public.”
A version of this article first appeared on Medscape.com.