User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
HPV4 vaccination in boys may prevent oropharyngeal cancer and cut costs
Vaccinating 12-year-old boys against human papillomavirus appears to be a cost-effective strategy for preventing HPV-related oropharyngeal cancer, according to an analysis performed using a theoretical cohort.
In a Markov model, a theoretical Canadian cohort of 192,940 boys aged 12 years in 2012 showed that use of the quadrivalent HPV (HPV4) vaccine produced 0.05 more quality-adjusted life-years and saved $145 Canadian dollars per person vs. no vaccine, assuming 99% vaccine efficacy and 70% uptake. The findings were reported by Dr. Donna M. Graham of the Princess Margaret Cancer Center, Toronto, and her colleagues online April 13 in the journal Cancer.
When 50% vaccine efficacy and 50% uptake were assumed, the HPV4 vaccine produced 0.023 more quality-adjusted life-years and saved $42 Canadian dollars, the investigators said. Based on these findings, use of HPV4 in boys could save between $8 million and $28 million Canadian dollars over the lifetime of the cohort (Cancer 2015 April 13 [doi:10.1002/cncr.29111]).
“The argument for funding male HPV vaccination in North America is becoming more compelling given the additional benefits of reduction in genital warts and anal cancer and the potential benefits for the female population because of increased herd immunity. Prospective data collection for male HPV vaccination and [oropharyngeal cancer] may validate these findings in the future,” they concluded.
Study coauthor Dr. Geoffrey Liu serves on the advisory boards of Pfizer and Novartis.
Vaccinating 12-year-old boys against human papillomavirus appears to be a cost-effective strategy for preventing HPV-related oropharyngeal cancer, according to an analysis performed using a theoretical cohort.
In a Markov model, a theoretical Canadian cohort of 192,940 boys aged 12 years in 2012 showed that use of the quadrivalent HPV (HPV4) vaccine produced 0.05 more quality-adjusted life-years and saved $145 Canadian dollars per person vs. no vaccine, assuming 99% vaccine efficacy and 70% uptake. The findings were reported by Dr. Donna M. Graham of the Princess Margaret Cancer Center, Toronto, and her colleagues online April 13 in the journal Cancer.
When 50% vaccine efficacy and 50% uptake were assumed, the HPV4 vaccine produced 0.023 more quality-adjusted life-years and saved $42 Canadian dollars, the investigators said. Based on these findings, use of HPV4 in boys could save between $8 million and $28 million Canadian dollars over the lifetime of the cohort (Cancer 2015 April 13 [doi:10.1002/cncr.29111]).
“The argument for funding male HPV vaccination in North America is becoming more compelling given the additional benefits of reduction in genital warts and anal cancer and the potential benefits for the female population because of increased herd immunity. Prospective data collection for male HPV vaccination and [oropharyngeal cancer] may validate these findings in the future,” they concluded.
Study coauthor Dr. Geoffrey Liu serves on the advisory boards of Pfizer and Novartis.
Vaccinating 12-year-old boys against human papillomavirus appears to be a cost-effective strategy for preventing HPV-related oropharyngeal cancer, according to an analysis performed using a theoretical cohort.
In a Markov model, a theoretical Canadian cohort of 192,940 boys aged 12 years in 2012 showed that use of the quadrivalent HPV (HPV4) vaccine produced 0.05 more quality-adjusted life-years and saved $145 Canadian dollars per person vs. no vaccine, assuming 99% vaccine efficacy and 70% uptake. The findings were reported by Dr. Donna M. Graham of the Princess Margaret Cancer Center, Toronto, and her colleagues online April 13 in the journal Cancer.
When 50% vaccine efficacy and 50% uptake were assumed, the HPV4 vaccine produced 0.023 more quality-adjusted life-years and saved $42 Canadian dollars, the investigators said. Based on these findings, use of HPV4 in boys could save between $8 million and $28 million Canadian dollars over the lifetime of the cohort (Cancer 2015 April 13 [doi:10.1002/cncr.29111]).
“The argument for funding male HPV vaccination in North America is becoming more compelling given the additional benefits of reduction in genital warts and anal cancer and the potential benefits for the female population because of increased herd immunity. Prospective data collection for male HPV vaccination and [oropharyngeal cancer] may validate these findings in the future,” they concluded.
Study coauthor Dr. Geoffrey Liu serves on the advisory boards of Pfizer and Novartis.
FROM CANCER
Key clinical point: HPV vaccination in boys appears to be feasible for preventing oropharyngeal cancer.
Major finding: Use of the HPV4 vaccine in boys could save between $8 million and $28 million Canadian dollars over the lifetime of the cohort.
Data source: A cost-effectiveness analysis in a theoretical Canadian cohort of 192,940 boys.
Disclosures: Coauthor Dr. Geoffrey Liu serves on the advisory boards of Pfizer and Novartis.
Plant-based supplement safely protects against UV radiation
MIAMI BEACH – An oral Polypodium leucotomos extract supplement was safe and effective for sun protection in a randomized, double-blind, placebo-controlled study of 40 participants.
The plant-based supplement, marketed as Heliocare by Ferndale Healthcare, was associated with a significant reduction in the risk of sunburn and the risk of erythema after ultraviolet B exposure, and with a significant increase in sun tolerability, according to study coauthor Dr. Brian Berman, who presented the findings at the South Beach Symposium.
After 2 months of treatment, 8 of 20 participants in the placebo group experienced at least one episode of sunburn, compared with 2 of 20 in the treatment group. At day 28 of treatment, 1 vs. 8 participants in the placebo and treatment groups, respectively, experienced an increased minimal erythema dose (MED), and 3 vs. 10 experienced decreased ultraviolet-induced erythema intensity (J. Clin. Aesthet. Dermatol. 2015;8:19-23).
Study participants were adults aged 18-65 years with Fitzpatrick skin types I-IV. They took 240-mg Heliocare capsules twice daily for 60 days and were assessed by physical examination. Vital signs were measured and clinical laboratory parameters, including hematology, comprehensive metabolic panel, and prothrombin time-partial thromboplastin time, were assessed at baseline and at the end of treatment. Twelve participants in each group also underwent MED testing. The treatment and placebo groups were similar with respect to the prestudy number of sunburns and the number of hours of sun exposure both before and during the study, said Dr. Berman of the University of Miami.
No safety issues associated with treatment were detected. Four participants in the treatment group reported transient mild fatigue, bloating, and headache, and one in the placebo group reported headache.
Polypodium leucotomos is a South American species of fern. Extracts from the fern have been used for at least 4 decades for photoprotection and for treatment of various skin disorders. The current findings suggest that the supplement is a safe and effective means for reducing the damaging effects of ultraviolet radiation, he said.
As with any dietary supplement, Polypodium leucotomos extract is not approved by the Food and Drug Administration to diagnose, treat, cure, or prevent any disease.
Dr. Berman and his colleagues concluded that, based on the excellent safety profile, additional studies assessing higher doses may be warranted.
Dr. Berman is a consultant/speaker for Ferndale Pharmaceuticals and has relationships with several other pharmaceutical companies.
MIAMI BEACH – An oral Polypodium leucotomos extract supplement was safe and effective for sun protection in a randomized, double-blind, placebo-controlled study of 40 participants.
The plant-based supplement, marketed as Heliocare by Ferndale Healthcare, was associated with a significant reduction in the risk of sunburn and the risk of erythema after ultraviolet B exposure, and with a significant increase in sun tolerability, according to study coauthor Dr. Brian Berman, who presented the findings at the South Beach Symposium.
After 2 months of treatment, 8 of 20 participants in the placebo group experienced at least one episode of sunburn, compared with 2 of 20 in the treatment group. At day 28 of treatment, 1 vs. 8 participants in the placebo and treatment groups, respectively, experienced an increased minimal erythema dose (MED), and 3 vs. 10 experienced decreased ultraviolet-induced erythema intensity (J. Clin. Aesthet. Dermatol. 2015;8:19-23).
Study participants were adults aged 18-65 years with Fitzpatrick skin types I-IV. They took 240-mg Heliocare capsules twice daily for 60 days and were assessed by physical examination. Vital signs were measured and clinical laboratory parameters, including hematology, comprehensive metabolic panel, and prothrombin time-partial thromboplastin time, were assessed at baseline and at the end of treatment. Twelve participants in each group also underwent MED testing. The treatment and placebo groups were similar with respect to the prestudy number of sunburns and the number of hours of sun exposure both before and during the study, said Dr. Berman of the University of Miami.
No safety issues associated with treatment were detected. Four participants in the treatment group reported transient mild fatigue, bloating, and headache, and one in the placebo group reported headache.
Polypodium leucotomos is a South American species of fern. Extracts from the fern have been used for at least 4 decades for photoprotection and for treatment of various skin disorders. The current findings suggest that the supplement is a safe and effective means for reducing the damaging effects of ultraviolet radiation, he said.
As with any dietary supplement, Polypodium leucotomos extract is not approved by the Food and Drug Administration to diagnose, treat, cure, or prevent any disease.
Dr. Berman and his colleagues concluded that, based on the excellent safety profile, additional studies assessing higher doses may be warranted.
Dr. Berman is a consultant/speaker for Ferndale Pharmaceuticals and has relationships with several other pharmaceutical companies.
MIAMI BEACH – An oral Polypodium leucotomos extract supplement was safe and effective for sun protection in a randomized, double-blind, placebo-controlled study of 40 participants.
The plant-based supplement, marketed as Heliocare by Ferndale Healthcare, was associated with a significant reduction in the risk of sunburn and the risk of erythema after ultraviolet B exposure, and with a significant increase in sun tolerability, according to study coauthor Dr. Brian Berman, who presented the findings at the South Beach Symposium.
After 2 months of treatment, 8 of 20 participants in the placebo group experienced at least one episode of sunburn, compared with 2 of 20 in the treatment group. At day 28 of treatment, 1 vs. 8 participants in the placebo and treatment groups, respectively, experienced an increased minimal erythema dose (MED), and 3 vs. 10 experienced decreased ultraviolet-induced erythema intensity (J. Clin. Aesthet. Dermatol. 2015;8:19-23).
Study participants were adults aged 18-65 years with Fitzpatrick skin types I-IV. They took 240-mg Heliocare capsules twice daily for 60 days and were assessed by physical examination. Vital signs were measured and clinical laboratory parameters, including hematology, comprehensive metabolic panel, and prothrombin time-partial thromboplastin time, were assessed at baseline and at the end of treatment. Twelve participants in each group also underwent MED testing. The treatment and placebo groups were similar with respect to the prestudy number of sunburns and the number of hours of sun exposure both before and during the study, said Dr. Berman of the University of Miami.
No safety issues associated with treatment were detected. Four participants in the treatment group reported transient mild fatigue, bloating, and headache, and one in the placebo group reported headache.
Polypodium leucotomos is a South American species of fern. Extracts from the fern have been used for at least 4 decades for photoprotection and for treatment of various skin disorders. The current findings suggest that the supplement is a safe and effective means for reducing the damaging effects of ultraviolet radiation, he said.
As with any dietary supplement, Polypodium leucotomos extract is not approved by the Food and Drug Administration to diagnose, treat, cure, or prevent any disease.
Dr. Berman and his colleagues concluded that, based on the excellent safety profile, additional studies assessing higher doses may be warranted.
Dr. Berman is a consultant/speaker for Ferndale Pharmaceuticals and has relationships with several other pharmaceutical companies.
AT THE SOUTH BEACH SYMPOSIUM
Key clinical point: An oral Polypodium leucotomos supplement provides safe and effective photoprotection.
Major finding:. At 2 months, eight placebo-group and two treatment-group participants experienced at least one episode of sunburn.
Data source: A randomized, double-blind, placebo-controlled study of 40 subjects.
Disclosures: Dr. Berman is a consultant/speaker for Ferndale Pharmaceuticals and has relationships with several other pharmaceutical companies.
Medicare beneficiaries often experience delays before melanoma surgery
About 22% of Medicare beneficiaries experience a delay of longer than 1.5 months between melanoma diagnosis and surgery, according to findings from a retrospective cohort study.
Of 32,501 melanoma cases from the Surveillance, Epidemiology, and End Results (SEER)–Medicare database, about 77.7% underwent excision within 1.5 months of biopsy, and of those who underwent excision after 1.5 months, 8.1% experienced a delay of more than 3 months. The risk-adjusted incidence of surgical delay longer than 1.5 months was significantly increased for those aged 85 years and older, compared with those aged 65 years and younger (odds ratio, 1.28), for those with a prior melanoma (OR, 1.20), and for those with three or more Elixhauser comorbidities (OR, 1.18), Dr. Jason P. Lott of Yale University, New Haven, Conn. and his colleagues reported online April 8 in JAMA Dermatology.
The lowest risk of delay was among those who underwent biopsy and excision by dermatologists (probability, 16.5%), and the highest was among those with a biopsy performed by a nondermatologist and excised by a primary care physician (probability, 30.7%), the investigators said (JAMA Dermatol. 2015 April 8 [doi:10.1001/jamadermatol.2015.119]).
The study, which provides the first population-based estimates of melanoma surgery delay among Medicare beneficiaries, and which shows that delays are relatively common, highlights “opportunities for quality improvement in dermatologic care and suggests that efforts to minimize the delay of surgery for melanoma might focus on increased access to dermatologic expertise and enhanced coordination of care among different specialists,” the investigators concluded.
This study was supported by the Robert Wood Johnson Foundation and the P30 Cancer Center Support Grant at the Yale Comprehensive Cancer Center.
Dr. Lott reported having no disclosures. Coauthor Dr. Cary P. Gross received research grant support from Johnson & Johnson, Merck, and 21st Century Oncology.
About 22% of Medicare beneficiaries experience a delay of longer than 1.5 months between melanoma diagnosis and surgery, according to findings from a retrospective cohort study.
Of 32,501 melanoma cases from the Surveillance, Epidemiology, and End Results (SEER)–Medicare database, about 77.7% underwent excision within 1.5 months of biopsy, and of those who underwent excision after 1.5 months, 8.1% experienced a delay of more than 3 months. The risk-adjusted incidence of surgical delay longer than 1.5 months was significantly increased for those aged 85 years and older, compared with those aged 65 years and younger (odds ratio, 1.28), for those with a prior melanoma (OR, 1.20), and for those with three or more Elixhauser comorbidities (OR, 1.18), Dr. Jason P. Lott of Yale University, New Haven, Conn. and his colleagues reported online April 8 in JAMA Dermatology.
The lowest risk of delay was among those who underwent biopsy and excision by dermatologists (probability, 16.5%), and the highest was among those with a biopsy performed by a nondermatologist and excised by a primary care physician (probability, 30.7%), the investigators said (JAMA Dermatol. 2015 April 8 [doi:10.1001/jamadermatol.2015.119]).
The study, which provides the first population-based estimates of melanoma surgery delay among Medicare beneficiaries, and which shows that delays are relatively common, highlights “opportunities for quality improvement in dermatologic care and suggests that efforts to minimize the delay of surgery for melanoma might focus on increased access to dermatologic expertise and enhanced coordination of care among different specialists,” the investigators concluded.
This study was supported by the Robert Wood Johnson Foundation and the P30 Cancer Center Support Grant at the Yale Comprehensive Cancer Center.
Dr. Lott reported having no disclosures. Coauthor Dr. Cary P. Gross received research grant support from Johnson & Johnson, Merck, and 21st Century Oncology.
About 22% of Medicare beneficiaries experience a delay of longer than 1.5 months between melanoma diagnosis and surgery, according to findings from a retrospective cohort study.
Of 32,501 melanoma cases from the Surveillance, Epidemiology, and End Results (SEER)–Medicare database, about 77.7% underwent excision within 1.5 months of biopsy, and of those who underwent excision after 1.5 months, 8.1% experienced a delay of more than 3 months. The risk-adjusted incidence of surgical delay longer than 1.5 months was significantly increased for those aged 85 years and older, compared with those aged 65 years and younger (odds ratio, 1.28), for those with a prior melanoma (OR, 1.20), and for those with three or more Elixhauser comorbidities (OR, 1.18), Dr. Jason P. Lott of Yale University, New Haven, Conn. and his colleagues reported online April 8 in JAMA Dermatology.
The lowest risk of delay was among those who underwent biopsy and excision by dermatologists (probability, 16.5%), and the highest was among those with a biopsy performed by a nondermatologist and excised by a primary care physician (probability, 30.7%), the investigators said (JAMA Dermatol. 2015 April 8 [doi:10.1001/jamadermatol.2015.119]).
The study, which provides the first population-based estimates of melanoma surgery delay among Medicare beneficiaries, and which shows that delays are relatively common, highlights “opportunities for quality improvement in dermatologic care and suggests that efforts to minimize the delay of surgery for melanoma might focus on increased access to dermatologic expertise and enhanced coordination of care among different specialists,” the investigators concluded.
This study was supported by the Robert Wood Johnson Foundation and the P30 Cancer Center Support Grant at the Yale Comprehensive Cancer Center.
Dr. Lott reported having no disclosures. Coauthor Dr. Cary P. Gross received research grant support from Johnson & Johnson, Merck, and 21st Century Oncology.
FROM JAMA DERMATOLOGY
Key clinical point: Opportunities exist for improving the timely delivery of melanoma surgery for Medicare beneficiaries.
Major finding: A total of 22% of Medicare beneficiaries had a delay of more than 1.5 months between melanoma diagnosis and surgery.
Data source: A retrospective cohort study involving 32,501 melanoma cases.
Disclosures:This study was supported by the Robert Wood Johnson Foundation and the P30 Cancer Center Support Grant at the Yale Comprehensive Cancer Center. Dr. Lott reported having no disclosures. Coauthor Dr. Cary P. Gross received research grant support from Johnson & Johnson, Merck, and 21st Century Oncology.
KOH solution, AK treatment both improve genital warts
MIAMI BEACH – Two novel therapies have potential for the treatment of genital warts, according to Dr. Theodore Rosen.
The first of these “way-off-label” treatments involves application of 5% potassium hydroxide (KOH) solution daily for 12 weeks, said Dr. Rosen, professor of dermatology at Baylor College of Medicine in Houston, Tex.
In a randomized, open-label study of 60 patients conducted in Turkey, researchers compared the KOH treatment with a 10% salicylic acid and 0.5% 5-fluorouracil compound available outside of the United States. The compound used in the study is similar to the WartPEEL (MedCara Pharmaceuticals) product available in the United States, Dr. Rosen said at the South Beach Symposium.
Both treatments were associated with a significant decrease in the number of lesions, and the outcomes were similar in both groups at 12 weeks (mean decrease from 17.03 to 3.73 lesions with KOH and from 16.13 to 3.10 with the 5-FU product). The investigators reported that excellent clearance was achieved by 70% and 76.7% of patients in the KOH and 5-FU groups, respectively, and marked improvement was seen in 13.3% and 20% of patients in the groups, respectively (Int. J. Dermatol. 2014; 53:1145-50).
No difference was seen between the groups in the rate of relapse at 16 weeks, and no serious adverse events were reported.
“This is a dirt-cheap way to treat genital warts,” Dr. Rosen said, adding that clinicians can simply give patients 5% KOH in a small bottle and instruct them to apply it once daily.
A second potential – but “not-so-dirt-cheap” – treatment worth noting is ingenol mebutate, based on findings from another small study, Dr. Rosen said.
Ingenol mebutate (Picato), which is approved for the treatment of actinic keratosis, was shown in 10 patients with human papillomovirus-6–positive genital warts to provide complete clearance with a single application.
The treatment was compared with vehicle in each patient – ingenol mebutate was applied to one affected area, and vehicle was applied to another affected area. The areas with active treatment were completely clear within 3-7 days, and the areas where vehicle was applied were not clear. No recurrence was noted at 3 months in the areas treated with ingenol mebutate.
Not surprisingly, the treatment was associated with mild to moderate burning, but it is a “very, very interesting, very short treatment,” Dr. Rosen said.
Dr. Rosen reported having no relevant disclosures.
MIAMI BEACH – Two novel therapies have potential for the treatment of genital warts, according to Dr. Theodore Rosen.
The first of these “way-off-label” treatments involves application of 5% potassium hydroxide (KOH) solution daily for 12 weeks, said Dr. Rosen, professor of dermatology at Baylor College of Medicine in Houston, Tex.
In a randomized, open-label study of 60 patients conducted in Turkey, researchers compared the KOH treatment with a 10% salicylic acid and 0.5% 5-fluorouracil compound available outside of the United States. The compound used in the study is similar to the WartPEEL (MedCara Pharmaceuticals) product available in the United States, Dr. Rosen said at the South Beach Symposium.
Both treatments were associated with a significant decrease in the number of lesions, and the outcomes were similar in both groups at 12 weeks (mean decrease from 17.03 to 3.73 lesions with KOH and from 16.13 to 3.10 with the 5-FU product). The investigators reported that excellent clearance was achieved by 70% and 76.7% of patients in the KOH and 5-FU groups, respectively, and marked improvement was seen in 13.3% and 20% of patients in the groups, respectively (Int. J. Dermatol. 2014; 53:1145-50).
No difference was seen between the groups in the rate of relapse at 16 weeks, and no serious adverse events were reported.
“This is a dirt-cheap way to treat genital warts,” Dr. Rosen said, adding that clinicians can simply give patients 5% KOH in a small bottle and instruct them to apply it once daily.
A second potential – but “not-so-dirt-cheap” – treatment worth noting is ingenol mebutate, based on findings from another small study, Dr. Rosen said.
Ingenol mebutate (Picato), which is approved for the treatment of actinic keratosis, was shown in 10 patients with human papillomovirus-6–positive genital warts to provide complete clearance with a single application.
The treatment was compared with vehicle in each patient – ingenol mebutate was applied to one affected area, and vehicle was applied to another affected area. The areas with active treatment were completely clear within 3-7 days, and the areas where vehicle was applied were not clear. No recurrence was noted at 3 months in the areas treated with ingenol mebutate.
Not surprisingly, the treatment was associated with mild to moderate burning, but it is a “very, very interesting, very short treatment,” Dr. Rosen said.
Dr. Rosen reported having no relevant disclosures.
MIAMI BEACH – Two novel therapies have potential for the treatment of genital warts, according to Dr. Theodore Rosen.
The first of these “way-off-label” treatments involves application of 5% potassium hydroxide (KOH) solution daily for 12 weeks, said Dr. Rosen, professor of dermatology at Baylor College of Medicine in Houston, Tex.
In a randomized, open-label study of 60 patients conducted in Turkey, researchers compared the KOH treatment with a 10% salicylic acid and 0.5% 5-fluorouracil compound available outside of the United States. The compound used in the study is similar to the WartPEEL (MedCara Pharmaceuticals) product available in the United States, Dr. Rosen said at the South Beach Symposium.
Both treatments were associated with a significant decrease in the number of lesions, and the outcomes were similar in both groups at 12 weeks (mean decrease from 17.03 to 3.73 lesions with KOH and from 16.13 to 3.10 with the 5-FU product). The investigators reported that excellent clearance was achieved by 70% and 76.7% of patients in the KOH and 5-FU groups, respectively, and marked improvement was seen in 13.3% and 20% of patients in the groups, respectively (Int. J. Dermatol. 2014; 53:1145-50).
No difference was seen between the groups in the rate of relapse at 16 weeks, and no serious adverse events were reported.
“This is a dirt-cheap way to treat genital warts,” Dr. Rosen said, adding that clinicians can simply give patients 5% KOH in a small bottle and instruct them to apply it once daily.
A second potential – but “not-so-dirt-cheap” – treatment worth noting is ingenol mebutate, based on findings from another small study, Dr. Rosen said.
Ingenol mebutate (Picato), which is approved for the treatment of actinic keratosis, was shown in 10 patients with human papillomovirus-6–positive genital warts to provide complete clearance with a single application.
The treatment was compared with vehicle in each patient – ingenol mebutate was applied to one affected area, and vehicle was applied to another affected area. The areas with active treatment were completely clear within 3-7 days, and the areas where vehicle was applied were not clear. No recurrence was noted at 3 months in the areas treated with ingenol mebutate.
Not surprisingly, the treatment was associated with mild to moderate burning, but it is a “very, very interesting, very short treatment,” Dr. Rosen said.
Dr. Rosen reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM
cfDNA testing benefits extend to routine aneuploidy screening population
Cell-free DNA (cfDNA) testing performed better than did standard screening plus measurement of nuchal translucency and biochemical analytes for detecting trisomy 21 in a large, prospective, multicenter study of women undergoing routine prenatal screening.
In 15,841 women included in the study, known as the Noninvasive Examination of Trisomy (NEXT) study, the area under the curve (AUC) for trisomy 21 was 0.999 for cell-free DNA testing, compared with 0.958 for standard screening – a statistically significant difference. Trisomy 21 was detected in 38 of 38 cases using cfDNA, compared with 30 of 38 cases using standard screening (sensitivity of 100% vs. 78.9%), Dr. Mary E. Norton of the University of California, San Francisco, and her colleagues reported online in the New England Journal of Medicine.
Further, the false positive rate with cfDNA was nearly 100 times lower than that with standard screening (0.06% vs. 5.4%), and the positive predictive value was 80.9% vs. 3.4%, respectively, the investigators said (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMoal407349]).
Although the study was powered to compare only the detection of trisomy 21, cfDNA also appeared to be better for detecting trisomies 18 and 13, and the lower false-positive rate and higher positive predictive value with cfDNA testing support its use in risk assessment for these trisomies, they said.
Participants in the blinded study, which was conducted at 35 centers in six countries in the United States, Canada, and Europe, were pregnant adult women with a mean age of 31 years and a mean gestational age of 12.5 weeks at the time of testing. The women presented for aneuploidy screening between March 2012 and April 2013 and underwent both cfDNA screening and standard screening, which included measurement of serum pregnancy-associated plasma protein A, total or free beta-subunit of human chorionic gonadotropin, and nuchal translucency.
Birth outcome was determined by diagnostic genetic testing in 557 women, and by newborn examination in the remaining subjects.
Cell-free DNA testing was introduced in 2011 and is reported to be highly effective for detecting trisomy 21.
“In practice, the use of this test could result in a significant reduction in diagnostic procedures,” Dr. Norton and her associates wrote.
They noted, however, that while several large proof-of-principle studies have confirmed the high sensitivity and specificity of cfDNA testing for detecting trisomy 21, few direct, well-powered studies have compared cfDNA testing and standard screening in a routine prenatal screening population. Rather, most studies have included only selected populations of high risk women who were sampled prior to invasive testing.
The current findings demonstrating the value of cfDNA in a general screening population are encouraging, but careful consideration of the screening method and costs is necessary before cfDNA testing can be widely implemented for general prenatal aneuploidy screening, Dr. Norton and her associates said.
Expectations regarding prenatal genetic testing also should be considered, they noted.
“For trisomy 21 and other common aneuploidies, cfDNA testing represents a highly accurate screening option … however, maternal serum and nuchal translucency screening can identify risk for a broad array of abnormalities that are not detectable on cfDNA testing,” they said, explaining that cases of trisomy 21 comprised just over 50% of aneuploidy in the study population and that women who desire a comprehensive assessment may prefer to undergo karyotyping or chromosomal microarray analysis.
This study is strengthened by the large sample size in a general prenatal screening population but is limited by the comparison between cfDNA testing and only standard first-trimester screening, the investigators said, explaining that methods such as integrated first- and second-trimester screening with nuchal translucency and biochemical analytes have higher sensitivity and specificity.
Further, the trisomy 21 detection rate of standard screening in this study was 79%, which is lower than the 82%-87% seen in a prior study.
“It is possible that standard screening has worse performance in clinical practice than under the stringent experimental conditions in which previously reported data were collected,” Dr. Norton and her associates noted.
This study was supported by Ariosa Diagnostics and the Perinatal Quality Foundation. Dr. Norton, in addition to most of her coauthors, reported receiving grant or other support from Ariosa. Multiple authors also reported a financial relationship with Natera outside the submitted work. Additionally, Howard Cuckle, Ph.D., is director of Genome Ltd., a provider of prenatal screening services including noninvasive prenatal testing, and Dr. Ronald J. Wapner reported relationships with Illumine and Sequenom, in addition to Ariosa and Natera. The remaining authors had no relevant financial disclosures.
The findings by Norton, et al. allay fears that cfDNA testing performance – shown to be strong in high risk populations – would be compromised in populations with a lower risk of trisomy 21, according to Dr. Lyn S. Chitty.
The positive predictive value of 80.9% with cfDNA testing compared very favorably with the 3.4% positive predictive value with standard screening in their large general aneuploidy screening population, Dr. Chitty wrote .
The findings also underscore the need for invasive testing to confirm a positive cfDNA result, as 9 of 47 results indicating high trisomy 21 risk were false positives.
The study “adds to the evidence showing good efficacy of such analysis in all women seeking screening for trisomy 21,” she said, adding that such well-conducted studies improve understanding of the performance of cfDNA testing.
“However, test uptake, economic aspects, and clinical utility will depend on local cultural and society factors, including attitudes with respect to disability, laws around termination of pregnancy, and the existing health care structure,” Dr. Chitty said.
The performance of cfDNA testing for trisomy 21 is clearly superior to traditional approaches in unselected pregnancies, but its use for screening for other chromosomal abnormalities requires further validation, she concluded.
Dr. Chitty is with the UCL Institute of Child Health and the Great Ormond Street Hospital for Children NHS Foundation Trust, both in London. She reported receiving grant support from Oxford Genome Technology, nonfinancial support from Verinata, and other support from Illumina and Ariosa outside the submitted work. Dr. Chitty wrote in an editorial responding to Dr. Norton and her associates’ study (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMe1502441]).
The findings by Norton, et al. allay fears that cfDNA testing performance – shown to be strong in high risk populations – would be compromised in populations with a lower risk of trisomy 21, according to Dr. Lyn S. Chitty.
The positive predictive value of 80.9% with cfDNA testing compared very favorably with the 3.4% positive predictive value with standard screening in their large general aneuploidy screening population, Dr. Chitty wrote .
The findings also underscore the need for invasive testing to confirm a positive cfDNA result, as 9 of 47 results indicating high trisomy 21 risk were false positives.
The study “adds to the evidence showing good efficacy of such analysis in all women seeking screening for trisomy 21,” she said, adding that such well-conducted studies improve understanding of the performance of cfDNA testing.
“However, test uptake, economic aspects, and clinical utility will depend on local cultural and society factors, including attitudes with respect to disability, laws around termination of pregnancy, and the existing health care structure,” Dr. Chitty said.
The performance of cfDNA testing for trisomy 21 is clearly superior to traditional approaches in unselected pregnancies, but its use for screening for other chromosomal abnormalities requires further validation, she concluded.
Dr. Chitty is with the UCL Institute of Child Health and the Great Ormond Street Hospital for Children NHS Foundation Trust, both in London. She reported receiving grant support from Oxford Genome Technology, nonfinancial support from Verinata, and other support from Illumina and Ariosa outside the submitted work. Dr. Chitty wrote in an editorial responding to Dr. Norton and her associates’ study (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMe1502441]).
The findings by Norton, et al. allay fears that cfDNA testing performance – shown to be strong in high risk populations – would be compromised in populations with a lower risk of trisomy 21, according to Dr. Lyn S. Chitty.
The positive predictive value of 80.9% with cfDNA testing compared very favorably with the 3.4% positive predictive value with standard screening in their large general aneuploidy screening population, Dr. Chitty wrote .
The findings also underscore the need for invasive testing to confirm a positive cfDNA result, as 9 of 47 results indicating high trisomy 21 risk were false positives.
The study “adds to the evidence showing good efficacy of such analysis in all women seeking screening for trisomy 21,” she said, adding that such well-conducted studies improve understanding of the performance of cfDNA testing.
“However, test uptake, economic aspects, and clinical utility will depend on local cultural and society factors, including attitudes with respect to disability, laws around termination of pregnancy, and the existing health care structure,” Dr. Chitty said.
The performance of cfDNA testing for trisomy 21 is clearly superior to traditional approaches in unselected pregnancies, but its use for screening for other chromosomal abnormalities requires further validation, she concluded.
Dr. Chitty is with the UCL Institute of Child Health and the Great Ormond Street Hospital for Children NHS Foundation Trust, both in London. She reported receiving grant support from Oxford Genome Technology, nonfinancial support from Verinata, and other support from Illumina and Ariosa outside the submitted work. Dr. Chitty wrote in an editorial responding to Dr. Norton and her associates’ study (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMe1502441]).
Cell-free DNA (cfDNA) testing performed better than did standard screening plus measurement of nuchal translucency and biochemical analytes for detecting trisomy 21 in a large, prospective, multicenter study of women undergoing routine prenatal screening.
In 15,841 women included in the study, known as the Noninvasive Examination of Trisomy (NEXT) study, the area under the curve (AUC) for trisomy 21 was 0.999 for cell-free DNA testing, compared with 0.958 for standard screening – a statistically significant difference. Trisomy 21 was detected in 38 of 38 cases using cfDNA, compared with 30 of 38 cases using standard screening (sensitivity of 100% vs. 78.9%), Dr. Mary E. Norton of the University of California, San Francisco, and her colleagues reported online in the New England Journal of Medicine.
Further, the false positive rate with cfDNA was nearly 100 times lower than that with standard screening (0.06% vs. 5.4%), and the positive predictive value was 80.9% vs. 3.4%, respectively, the investigators said (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMoal407349]).
Although the study was powered to compare only the detection of trisomy 21, cfDNA also appeared to be better for detecting trisomies 18 and 13, and the lower false-positive rate and higher positive predictive value with cfDNA testing support its use in risk assessment for these trisomies, they said.
Participants in the blinded study, which was conducted at 35 centers in six countries in the United States, Canada, and Europe, were pregnant adult women with a mean age of 31 years and a mean gestational age of 12.5 weeks at the time of testing. The women presented for aneuploidy screening between March 2012 and April 2013 and underwent both cfDNA screening and standard screening, which included measurement of serum pregnancy-associated plasma protein A, total or free beta-subunit of human chorionic gonadotropin, and nuchal translucency.
Birth outcome was determined by diagnostic genetic testing in 557 women, and by newborn examination in the remaining subjects.
Cell-free DNA testing was introduced in 2011 and is reported to be highly effective for detecting trisomy 21.
“In practice, the use of this test could result in a significant reduction in diagnostic procedures,” Dr. Norton and her associates wrote.
They noted, however, that while several large proof-of-principle studies have confirmed the high sensitivity and specificity of cfDNA testing for detecting trisomy 21, few direct, well-powered studies have compared cfDNA testing and standard screening in a routine prenatal screening population. Rather, most studies have included only selected populations of high risk women who were sampled prior to invasive testing.
The current findings demonstrating the value of cfDNA in a general screening population are encouraging, but careful consideration of the screening method and costs is necessary before cfDNA testing can be widely implemented for general prenatal aneuploidy screening, Dr. Norton and her associates said.
Expectations regarding prenatal genetic testing also should be considered, they noted.
“For trisomy 21 and other common aneuploidies, cfDNA testing represents a highly accurate screening option … however, maternal serum and nuchal translucency screening can identify risk for a broad array of abnormalities that are not detectable on cfDNA testing,” they said, explaining that cases of trisomy 21 comprised just over 50% of aneuploidy in the study population and that women who desire a comprehensive assessment may prefer to undergo karyotyping or chromosomal microarray analysis.
This study is strengthened by the large sample size in a general prenatal screening population but is limited by the comparison between cfDNA testing and only standard first-trimester screening, the investigators said, explaining that methods such as integrated first- and second-trimester screening with nuchal translucency and biochemical analytes have higher sensitivity and specificity.
Further, the trisomy 21 detection rate of standard screening in this study was 79%, which is lower than the 82%-87% seen in a prior study.
“It is possible that standard screening has worse performance in clinical practice than under the stringent experimental conditions in which previously reported data were collected,” Dr. Norton and her associates noted.
This study was supported by Ariosa Diagnostics and the Perinatal Quality Foundation. Dr. Norton, in addition to most of her coauthors, reported receiving grant or other support from Ariosa. Multiple authors also reported a financial relationship with Natera outside the submitted work. Additionally, Howard Cuckle, Ph.D., is director of Genome Ltd., a provider of prenatal screening services including noninvasive prenatal testing, and Dr. Ronald J. Wapner reported relationships with Illumine and Sequenom, in addition to Ariosa and Natera. The remaining authors had no relevant financial disclosures.
Cell-free DNA (cfDNA) testing performed better than did standard screening plus measurement of nuchal translucency and biochemical analytes for detecting trisomy 21 in a large, prospective, multicenter study of women undergoing routine prenatal screening.
In 15,841 women included in the study, known as the Noninvasive Examination of Trisomy (NEXT) study, the area under the curve (AUC) for trisomy 21 was 0.999 for cell-free DNA testing, compared with 0.958 for standard screening – a statistically significant difference. Trisomy 21 was detected in 38 of 38 cases using cfDNA, compared with 30 of 38 cases using standard screening (sensitivity of 100% vs. 78.9%), Dr. Mary E. Norton of the University of California, San Francisco, and her colleagues reported online in the New England Journal of Medicine.
Further, the false positive rate with cfDNA was nearly 100 times lower than that with standard screening (0.06% vs. 5.4%), and the positive predictive value was 80.9% vs. 3.4%, respectively, the investigators said (N. Engl. J. Med. 2015 April 1 [doi:10.1056/NEJMoal407349]).
Although the study was powered to compare only the detection of trisomy 21, cfDNA also appeared to be better for detecting trisomies 18 and 13, and the lower false-positive rate and higher positive predictive value with cfDNA testing support its use in risk assessment for these trisomies, they said.
Participants in the blinded study, which was conducted at 35 centers in six countries in the United States, Canada, and Europe, were pregnant adult women with a mean age of 31 years and a mean gestational age of 12.5 weeks at the time of testing. The women presented for aneuploidy screening between March 2012 and April 2013 and underwent both cfDNA screening and standard screening, which included measurement of serum pregnancy-associated plasma protein A, total or free beta-subunit of human chorionic gonadotropin, and nuchal translucency.
Birth outcome was determined by diagnostic genetic testing in 557 women, and by newborn examination in the remaining subjects.
Cell-free DNA testing was introduced in 2011 and is reported to be highly effective for detecting trisomy 21.
“In practice, the use of this test could result in a significant reduction in diagnostic procedures,” Dr. Norton and her associates wrote.
They noted, however, that while several large proof-of-principle studies have confirmed the high sensitivity and specificity of cfDNA testing for detecting trisomy 21, few direct, well-powered studies have compared cfDNA testing and standard screening in a routine prenatal screening population. Rather, most studies have included only selected populations of high risk women who were sampled prior to invasive testing.
The current findings demonstrating the value of cfDNA in a general screening population are encouraging, but careful consideration of the screening method and costs is necessary before cfDNA testing can be widely implemented for general prenatal aneuploidy screening, Dr. Norton and her associates said.
Expectations regarding prenatal genetic testing also should be considered, they noted.
“For trisomy 21 and other common aneuploidies, cfDNA testing represents a highly accurate screening option … however, maternal serum and nuchal translucency screening can identify risk for a broad array of abnormalities that are not detectable on cfDNA testing,” they said, explaining that cases of trisomy 21 comprised just over 50% of aneuploidy in the study population and that women who desire a comprehensive assessment may prefer to undergo karyotyping or chromosomal microarray analysis.
This study is strengthened by the large sample size in a general prenatal screening population but is limited by the comparison between cfDNA testing and only standard first-trimester screening, the investigators said, explaining that methods such as integrated first- and second-trimester screening with nuchal translucency and biochemical analytes have higher sensitivity and specificity.
Further, the trisomy 21 detection rate of standard screening in this study was 79%, which is lower than the 82%-87% seen in a prior study.
“It is possible that standard screening has worse performance in clinical practice than under the stringent experimental conditions in which previously reported data were collected,” Dr. Norton and her associates noted.
This study was supported by Ariosa Diagnostics and the Perinatal Quality Foundation. Dr. Norton, in addition to most of her coauthors, reported receiving grant or other support from Ariosa. Multiple authors also reported a financial relationship with Natera outside the submitted work. Additionally, Howard Cuckle, Ph.D., is director of Genome Ltd., a provider of prenatal screening services including noninvasive prenatal testing, and Dr. Ronald J. Wapner reported relationships with Illumine and Sequenom, in addition to Ariosa and Natera. The remaining authors had no relevant financial disclosures.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: cfDNA testing has high sensitivity and specificity for detecting trisomy 21 in the general prenatal screening population.
Major finding: The AUC for trisomy 21 was 0.999 for cfDNA testing, compared with 0.958 for standard screening.
Data source: A prospective, multicenter, blinded study of 15,841 women.
Disclosures: This study was supported by Ariosa Diagnostics and the Perinatal Quality Foundation. Dr. Norton, in addition to most of her coauthors, reported receiving grant or other support from Ariosa. Multiple authors also reported a financial relationship with Natera outside the submitted work. Additionally, Howard Cuckle, Ph.D., is director of Genome Ltd., a provider of prenatal screening services including noninvasive prenatal testing, and Dr. Ronald J. Wapner reported relationships with Illumine and Sequenom. The remaining authors had no relevant financial disclosures.
Physician-patient communication, terminology play important role in CPM decisions
BIRMINGHAM, ALA. – The perception that one’s physician had recommended contralateral prophylactic mastectomy was a particularly important factor in the decision to undergo the procedure among BRCA1/2 mutation noncarriers with newly diagnosed breast cancer in a prospective study.
Of 90 BRCA noncarriers with newly diagnosed breast cancer, a “sizable minority” (24.4%) chose to undergo contralateral prophylactic mastectomy (CPM) after learning their mutation status, Jada G. Hamilton, Ph.D., reported at the annual meeting of the American Society of Preventive Oncology.
By comparison, 88% of eight BRCA1/2 carriers who participated in the study chose to undergo CPM, and neither of two women with a BRCA1/2 variant of undetermined significance chose to undergo CPM.
On multivariate analysis, the perception that one’s physician had recommended CPM was most strongly associated with the decision to undergo the procedure (odds ratio, 11.1), said Dr. Hamilton of Memorial Sloan-Kettering Cancer Center, New York.
Other factors associated with the decision were a perception of greater risk for contralateral breast cancer (OR, 6.46) and a perception of greater pros of CPM (OR, 1.37), she said, noting that those who indicated they would feel good about having CPM and those who indicated they might feel regret if they didn’t have CPM were most likely to elect CPM.
Age, Ashkenazi Jewish ethnicity, breast cancer–related distress, perceived cons of CPM (such as disfigurement and concerns regarding a negative impact on one’s sex life), and decisional conflict regarding CPM were not significantly associated with the decision.
Presurgical genetic testing provides valuable information to women with newly diagnosed breast cancer as they begin to make decisions about treatment, Dr. Hamilton said. Although BRCA1/2 mutation noncarriers have a low (3%-10%) risk, compared with carriers (27%-37%), studies suggest that about 18% nevertheless choose to undergo CPM.
The psychosocial factors that may affect the decision are not well understood, Dr. Hamilton said.
For the current analysis, participants who were part of a larger prospective study on presurgical BRCA1/2 testing completed a questionnaire, and the frequency and psychosocial correlates of the decision to undergo a CPM were assessed. The participants were adult women with a median age of 43 years (range of 29-59 years).
The findings raise interesting questions for future work, Dr. Hamilton said.
“I think it’s really critical for future studies to dig in to what’s happening in terms of physician-patient communication around CPM,” she said, noting that it will be important to explore how such communication interacts with a woman’s past experiences, emotions, and beliefs to shape her cancer prevention decisions.
Further, the women who undergo CPM should be followed to assess their long-term outcomes with respect to factors such as quality of life, satisfaction, and decisional regret, she concluded.
The decision to undergo CPM and the effects of physician-patient communication on that decision were also addressed in another study presented at the meeting.
In that study – a mixed methods pilot study looking mainly at factors affecting informed decision making in women who had ductal carcinoma in situ (DCIS) or who were considered to be at increased risk of invasive breast cancer risk because of a diagnosis of lobular carcinoma in situ (LCIS), BRCA positivity, or 20% or greater calculated lifetime risk – anxiety about cancer recurrence was the top reason for pursuing CPM.
Despite a lack of survival benefit, an increasing number of women with DCIS are undergoing CPM, but little is known about the decision-making process, said Jessica Valente of Emory University, Atlanta.
She and her colleagues sought to identify factors impacting risk comprehension and decision making. Of 68 women with DCIS or who were at high risk for development of invasive breast cancer, 33 considered CPM and 11 underwent the procedure.
The choice to undergo CPM was significantly associated with plastic surgery consultation, increased 10-year breast cancer risk, genetic counseling, genetic testing, and higher income, she said.
The study also highlighted a lack of health literacy and understanding of related terminology.
Most participants (nearly 84%) stated that DCIS qualified as breast cancer, but only about 40% correctly defined DCIS, Ms. Valente said.
When asked what they would recommend as a treatment strategy for a friend with DCIS, 35% thought surgery would be best. A similar percentage would recommend surgery for LCIS.
“When we looked at ductal hyperplasia, fewer people thought that that qualified as cancer, and they were more likely to recommend observation,” she said.
Further, when asked to interpret the phrase “indolent lesion of epithelial origin,” which is a phrase that has been promoted as a replacement for “DCIS” in light of concerns that women are increasingly electing CPM for DCIS because of fear of the word “carcinoma” despite a 99% survival rate, only 28% of patients believed it referred to cancer.
Observation was one of the highly recommended interventions for “indolent lesion of epithelial origin,” followed by biopsy, she said, noting that only 13% recommended surgery when this phrase was used.
“Interestingly, 7.4% said an oral or topical medication [should be used for “indolent lesion of epithelial origin]” – a finding likely explained by the fact that some women interpreted the word “lesion” to mean a wound or sore on the skin, she said.
Additionally, few women were able to define contralateral prophylactic mastectomy.
Overall, despite the study population being very well educated and from a higher socioeconomic status, they had low scores for understanding terminology (8.21 out of 20), Ms. Valente noted.
The findings demonstrate that decision making in the context of DCIS remains complex and underscore the importance of recognizing patients’ knowledge of risk communication and terminology for supporting shared and informed surgical decision making, she said.
The findings also demonstrate that while fewer women felt that surgery was appropriate when the term “indolent lesion of epithelial origin” was used instead of “DCIS,” the proposed new terminology doesn’t necessarily provide the desired clarity, she noted.
“They still came up with such a broad range of interpretations that we really might be introducing a new set of conflicts and confusion when we think about changing to that terminology when we talk to patients,” she concluded.
Dr. Hamilton and Ms. Valente each reported having no disclosures.
BIRMINGHAM, ALA. – The perception that one’s physician had recommended contralateral prophylactic mastectomy was a particularly important factor in the decision to undergo the procedure among BRCA1/2 mutation noncarriers with newly diagnosed breast cancer in a prospective study.
Of 90 BRCA noncarriers with newly diagnosed breast cancer, a “sizable minority” (24.4%) chose to undergo contralateral prophylactic mastectomy (CPM) after learning their mutation status, Jada G. Hamilton, Ph.D., reported at the annual meeting of the American Society of Preventive Oncology.
By comparison, 88% of eight BRCA1/2 carriers who participated in the study chose to undergo CPM, and neither of two women with a BRCA1/2 variant of undetermined significance chose to undergo CPM.
On multivariate analysis, the perception that one’s physician had recommended CPM was most strongly associated with the decision to undergo the procedure (odds ratio, 11.1), said Dr. Hamilton of Memorial Sloan-Kettering Cancer Center, New York.
Other factors associated with the decision were a perception of greater risk for contralateral breast cancer (OR, 6.46) and a perception of greater pros of CPM (OR, 1.37), she said, noting that those who indicated they would feel good about having CPM and those who indicated they might feel regret if they didn’t have CPM were most likely to elect CPM.
Age, Ashkenazi Jewish ethnicity, breast cancer–related distress, perceived cons of CPM (such as disfigurement and concerns regarding a negative impact on one’s sex life), and decisional conflict regarding CPM were not significantly associated with the decision.
Presurgical genetic testing provides valuable information to women with newly diagnosed breast cancer as they begin to make decisions about treatment, Dr. Hamilton said. Although BRCA1/2 mutation noncarriers have a low (3%-10%) risk, compared with carriers (27%-37%), studies suggest that about 18% nevertheless choose to undergo CPM.
The psychosocial factors that may affect the decision are not well understood, Dr. Hamilton said.
For the current analysis, participants who were part of a larger prospective study on presurgical BRCA1/2 testing completed a questionnaire, and the frequency and psychosocial correlates of the decision to undergo a CPM were assessed. The participants were adult women with a median age of 43 years (range of 29-59 years).
The findings raise interesting questions for future work, Dr. Hamilton said.
“I think it’s really critical for future studies to dig in to what’s happening in terms of physician-patient communication around CPM,” she said, noting that it will be important to explore how such communication interacts with a woman’s past experiences, emotions, and beliefs to shape her cancer prevention decisions.
Further, the women who undergo CPM should be followed to assess their long-term outcomes with respect to factors such as quality of life, satisfaction, and decisional regret, she concluded.
The decision to undergo CPM and the effects of physician-patient communication on that decision were also addressed in another study presented at the meeting.
In that study – a mixed methods pilot study looking mainly at factors affecting informed decision making in women who had ductal carcinoma in situ (DCIS) or who were considered to be at increased risk of invasive breast cancer risk because of a diagnosis of lobular carcinoma in situ (LCIS), BRCA positivity, or 20% or greater calculated lifetime risk – anxiety about cancer recurrence was the top reason for pursuing CPM.
Despite a lack of survival benefit, an increasing number of women with DCIS are undergoing CPM, but little is known about the decision-making process, said Jessica Valente of Emory University, Atlanta.
She and her colleagues sought to identify factors impacting risk comprehension and decision making. Of 68 women with DCIS or who were at high risk for development of invasive breast cancer, 33 considered CPM and 11 underwent the procedure.
The choice to undergo CPM was significantly associated with plastic surgery consultation, increased 10-year breast cancer risk, genetic counseling, genetic testing, and higher income, she said.
The study also highlighted a lack of health literacy and understanding of related terminology.
Most participants (nearly 84%) stated that DCIS qualified as breast cancer, but only about 40% correctly defined DCIS, Ms. Valente said.
When asked what they would recommend as a treatment strategy for a friend with DCIS, 35% thought surgery would be best. A similar percentage would recommend surgery for LCIS.
“When we looked at ductal hyperplasia, fewer people thought that that qualified as cancer, and they were more likely to recommend observation,” she said.
Further, when asked to interpret the phrase “indolent lesion of epithelial origin,” which is a phrase that has been promoted as a replacement for “DCIS” in light of concerns that women are increasingly electing CPM for DCIS because of fear of the word “carcinoma” despite a 99% survival rate, only 28% of patients believed it referred to cancer.
Observation was one of the highly recommended interventions for “indolent lesion of epithelial origin,” followed by biopsy, she said, noting that only 13% recommended surgery when this phrase was used.
“Interestingly, 7.4% said an oral or topical medication [should be used for “indolent lesion of epithelial origin]” – a finding likely explained by the fact that some women interpreted the word “lesion” to mean a wound or sore on the skin, she said.
Additionally, few women were able to define contralateral prophylactic mastectomy.
Overall, despite the study population being very well educated and from a higher socioeconomic status, they had low scores for understanding terminology (8.21 out of 20), Ms. Valente noted.
The findings demonstrate that decision making in the context of DCIS remains complex and underscore the importance of recognizing patients’ knowledge of risk communication and terminology for supporting shared and informed surgical decision making, she said.
The findings also demonstrate that while fewer women felt that surgery was appropriate when the term “indolent lesion of epithelial origin” was used instead of “DCIS,” the proposed new terminology doesn’t necessarily provide the desired clarity, she noted.
“They still came up with such a broad range of interpretations that we really might be introducing a new set of conflicts and confusion when we think about changing to that terminology when we talk to patients,” she concluded.
Dr. Hamilton and Ms. Valente each reported having no disclosures.
BIRMINGHAM, ALA. – The perception that one’s physician had recommended contralateral prophylactic mastectomy was a particularly important factor in the decision to undergo the procedure among BRCA1/2 mutation noncarriers with newly diagnosed breast cancer in a prospective study.
Of 90 BRCA noncarriers with newly diagnosed breast cancer, a “sizable minority” (24.4%) chose to undergo contralateral prophylactic mastectomy (CPM) after learning their mutation status, Jada G. Hamilton, Ph.D., reported at the annual meeting of the American Society of Preventive Oncology.
By comparison, 88% of eight BRCA1/2 carriers who participated in the study chose to undergo CPM, and neither of two women with a BRCA1/2 variant of undetermined significance chose to undergo CPM.
On multivariate analysis, the perception that one’s physician had recommended CPM was most strongly associated with the decision to undergo the procedure (odds ratio, 11.1), said Dr. Hamilton of Memorial Sloan-Kettering Cancer Center, New York.
Other factors associated with the decision were a perception of greater risk for contralateral breast cancer (OR, 6.46) and a perception of greater pros of CPM (OR, 1.37), she said, noting that those who indicated they would feel good about having CPM and those who indicated they might feel regret if they didn’t have CPM were most likely to elect CPM.
Age, Ashkenazi Jewish ethnicity, breast cancer–related distress, perceived cons of CPM (such as disfigurement and concerns regarding a negative impact on one’s sex life), and decisional conflict regarding CPM were not significantly associated with the decision.
Presurgical genetic testing provides valuable information to women with newly diagnosed breast cancer as they begin to make decisions about treatment, Dr. Hamilton said. Although BRCA1/2 mutation noncarriers have a low (3%-10%) risk, compared with carriers (27%-37%), studies suggest that about 18% nevertheless choose to undergo CPM.
The psychosocial factors that may affect the decision are not well understood, Dr. Hamilton said.
For the current analysis, participants who were part of a larger prospective study on presurgical BRCA1/2 testing completed a questionnaire, and the frequency and psychosocial correlates of the decision to undergo a CPM were assessed. The participants were adult women with a median age of 43 years (range of 29-59 years).
The findings raise interesting questions for future work, Dr. Hamilton said.
“I think it’s really critical for future studies to dig in to what’s happening in terms of physician-patient communication around CPM,” she said, noting that it will be important to explore how such communication interacts with a woman’s past experiences, emotions, and beliefs to shape her cancer prevention decisions.
Further, the women who undergo CPM should be followed to assess their long-term outcomes with respect to factors such as quality of life, satisfaction, and decisional regret, she concluded.
The decision to undergo CPM and the effects of physician-patient communication on that decision were also addressed in another study presented at the meeting.
In that study – a mixed methods pilot study looking mainly at factors affecting informed decision making in women who had ductal carcinoma in situ (DCIS) or who were considered to be at increased risk of invasive breast cancer risk because of a diagnosis of lobular carcinoma in situ (LCIS), BRCA positivity, or 20% or greater calculated lifetime risk – anxiety about cancer recurrence was the top reason for pursuing CPM.
Despite a lack of survival benefit, an increasing number of women with DCIS are undergoing CPM, but little is known about the decision-making process, said Jessica Valente of Emory University, Atlanta.
She and her colleagues sought to identify factors impacting risk comprehension and decision making. Of 68 women with DCIS or who were at high risk for development of invasive breast cancer, 33 considered CPM and 11 underwent the procedure.
The choice to undergo CPM was significantly associated with plastic surgery consultation, increased 10-year breast cancer risk, genetic counseling, genetic testing, and higher income, she said.
The study also highlighted a lack of health literacy and understanding of related terminology.
Most participants (nearly 84%) stated that DCIS qualified as breast cancer, but only about 40% correctly defined DCIS, Ms. Valente said.
When asked what they would recommend as a treatment strategy for a friend with DCIS, 35% thought surgery would be best. A similar percentage would recommend surgery for LCIS.
“When we looked at ductal hyperplasia, fewer people thought that that qualified as cancer, and they were more likely to recommend observation,” she said.
Further, when asked to interpret the phrase “indolent lesion of epithelial origin,” which is a phrase that has been promoted as a replacement for “DCIS” in light of concerns that women are increasingly electing CPM for DCIS because of fear of the word “carcinoma” despite a 99% survival rate, only 28% of patients believed it referred to cancer.
Observation was one of the highly recommended interventions for “indolent lesion of epithelial origin,” followed by biopsy, she said, noting that only 13% recommended surgery when this phrase was used.
“Interestingly, 7.4% said an oral or topical medication [should be used for “indolent lesion of epithelial origin]” – a finding likely explained by the fact that some women interpreted the word “lesion” to mean a wound or sore on the skin, she said.
Additionally, few women were able to define contralateral prophylactic mastectomy.
Overall, despite the study population being very well educated and from a higher socioeconomic status, they had low scores for understanding terminology (8.21 out of 20), Ms. Valente noted.
The findings demonstrate that decision making in the context of DCIS remains complex and underscore the importance of recognizing patients’ knowledge of risk communication and terminology for supporting shared and informed surgical decision making, she said.
The findings also demonstrate that while fewer women felt that surgery was appropriate when the term “indolent lesion of epithelial origin” was used instead of “DCIS,” the proposed new terminology doesn’t necessarily provide the desired clarity, she noted.
“They still came up with such a broad range of interpretations that we really might be introducing a new set of conflicts and confusion when we think about changing to that terminology when we talk to patients,” she concluded.
Dr. Hamilton and Ms. Valente each reported having no disclosures.
AT THE ASPO ANNUAL MEETING
Key clinical point: Physician-patient communication plays an important role in a woman’s decision to undergo CPM.
Major finding: Of 90 women with invasive breast cancer and without BRCA mutations, 24% chose to undergo CPM; of 68 women with DCIS, 33 considered CPM and 11 underwent the procedure.The choice to undergo CPM was significantly associated with the perception that one’s physician had recommended CPM in the first study, and with a plastic surgery consultation, increased 10-year breast cancer risk, genetic counseling, genetic testing, and higher income in the DCIS study.
Data source: A prospective study of 90 patients with invasive breast cancer and a mixed methods pilot study of 68 patients with DCIS.
Disclosures: Dr. Hamilton and Ms. Valente each reported having no disclosures.
Nurses’ Health Study: No link between depression and breast cancer
BIRMINGHAM, ALA. – Neither depression nor antidepressant use are associated with an increased risk of breast cancer among participants in the Nurses’ Health Study, according to an analysis of data from 67,120 women enrolled in the ongoing prospective cohort study.
Of the women included in the analysis, 2,904 had confirmed breast cancer as of the end of December 2012, including 2,333 with invasive disease. After adjusting for age, body mass index, and menopausal status, no statistically significant associations were seen between invasive or in situ breast cancer and depression or antidepressant use, Katherine W. Reeves, Ph.D. reported at the annual meeting of the American Society of Preventive Oncology.
The point estimates for the odds ratios were all below 1 for in situ disease, indicating a potential protective effect of depression, Dr. Reeves said.
“These were not statistically significant, so I would caution against overinterpreting the results, but it is kind of curious,” she said, noting that the finding may indicate that depressed women are less likely than nondepressed women are to have a mammogram – and thus are less likely to have the opportunity to be diagnosed with in situ disease.
When depression and antidepressant use were included together in the same model, they remained unassociated with breast cancer risk (odds ratio, 0.87), said Dr. Reeves of the University of Massachusetts Amherst.
Study subjects were an average age of 66 years, 8.7% were clinically depressed, and 9.7% used antidepressants. Data on depression and antidepressant use among Nurses’ Health Study participants were collected simultaneously beginning in 2000.
Depression and antidepressant use were self-reported, and depressive symptoms were confirmed using the five-item Mental Health Inventory.
The findings are encouraging; depression and antidepressant use are common and both have been hypothesized to increase breast cancer risk. Some prior studies have found a link between either depression or antidepressant use and breast cancer, and others have not – but most have had important limitations, including retrospective design and inclusion of major depression only, among others, she said.
“To me, though, the most important limitation is that previous studies have not evaluated depression and antidepressant use together,” Dr. Reeves said.
In the current study, which did consider both, no evidence was seen to suggest that depression or antidepressant use affects breast cancer risk.
“It’s typically very unexciting to have to report null results, but in this case, I think this is excellent news and really the best we could have hoped for,” she said, adding that although they are preliminary, the findings should be “very reassuring for the millions of women with depression and/or those using antidepressants.”
“Depression is a very serious medical condition. It deserves to be treated, and it’s nice that these women can take the antidepressants, which so effectively treat this condition without worry that they’re doing something that would adversely affect their breast cancer risk in the future,” she concluded, noting that more sophisticated analyses of the data are planned to consider additional variables, including treatment duration. The analyses will also be repeated in the Nurses’ Health Study II cohort, which is a younger cohort with a higher incidence of premenopausal breast cancer and a greater prevalence of both depression and antidepressant use.
BIRMINGHAM, ALA. – Neither depression nor antidepressant use are associated with an increased risk of breast cancer among participants in the Nurses’ Health Study, according to an analysis of data from 67,120 women enrolled in the ongoing prospective cohort study.
Of the women included in the analysis, 2,904 had confirmed breast cancer as of the end of December 2012, including 2,333 with invasive disease. After adjusting for age, body mass index, and menopausal status, no statistically significant associations were seen between invasive or in situ breast cancer and depression or antidepressant use, Katherine W. Reeves, Ph.D. reported at the annual meeting of the American Society of Preventive Oncology.
The point estimates for the odds ratios were all below 1 for in situ disease, indicating a potential protective effect of depression, Dr. Reeves said.
“These were not statistically significant, so I would caution against overinterpreting the results, but it is kind of curious,” she said, noting that the finding may indicate that depressed women are less likely than nondepressed women are to have a mammogram – and thus are less likely to have the opportunity to be diagnosed with in situ disease.
When depression and antidepressant use were included together in the same model, they remained unassociated with breast cancer risk (odds ratio, 0.87), said Dr. Reeves of the University of Massachusetts Amherst.
Study subjects were an average age of 66 years, 8.7% were clinically depressed, and 9.7% used antidepressants. Data on depression and antidepressant use among Nurses’ Health Study participants were collected simultaneously beginning in 2000.
Depression and antidepressant use were self-reported, and depressive symptoms were confirmed using the five-item Mental Health Inventory.
The findings are encouraging; depression and antidepressant use are common and both have been hypothesized to increase breast cancer risk. Some prior studies have found a link between either depression or antidepressant use and breast cancer, and others have not – but most have had important limitations, including retrospective design and inclusion of major depression only, among others, she said.
“To me, though, the most important limitation is that previous studies have not evaluated depression and antidepressant use together,” Dr. Reeves said.
In the current study, which did consider both, no evidence was seen to suggest that depression or antidepressant use affects breast cancer risk.
“It’s typically very unexciting to have to report null results, but in this case, I think this is excellent news and really the best we could have hoped for,” she said, adding that although they are preliminary, the findings should be “very reassuring for the millions of women with depression and/or those using antidepressants.”
“Depression is a very serious medical condition. It deserves to be treated, and it’s nice that these women can take the antidepressants, which so effectively treat this condition without worry that they’re doing something that would adversely affect their breast cancer risk in the future,” she concluded, noting that more sophisticated analyses of the data are planned to consider additional variables, including treatment duration. The analyses will also be repeated in the Nurses’ Health Study II cohort, which is a younger cohort with a higher incidence of premenopausal breast cancer and a greater prevalence of both depression and antidepressant use.
BIRMINGHAM, ALA. – Neither depression nor antidepressant use are associated with an increased risk of breast cancer among participants in the Nurses’ Health Study, according to an analysis of data from 67,120 women enrolled in the ongoing prospective cohort study.
Of the women included in the analysis, 2,904 had confirmed breast cancer as of the end of December 2012, including 2,333 with invasive disease. After adjusting for age, body mass index, and menopausal status, no statistically significant associations were seen between invasive or in situ breast cancer and depression or antidepressant use, Katherine W. Reeves, Ph.D. reported at the annual meeting of the American Society of Preventive Oncology.
The point estimates for the odds ratios were all below 1 for in situ disease, indicating a potential protective effect of depression, Dr. Reeves said.
“These were not statistically significant, so I would caution against overinterpreting the results, but it is kind of curious,” she said, noting that the finding may indicate that depressed women are less likely than nondepressed women are to have a mammogram – and thus are less likely to have the opportunity to be diagnosed with in situ disease.
When depression and antidepressant use were included together in the same model, they remained unassociated with breast cancer risk (odds ratio, 0.87), said Dr. Reeves of the University of Massachusetts Amherst.
Study subjects were an average age of 66 years, 8.7% were clinically depressed, and 9.7% used antidepressants. Data on depression and antidepressant use among Nurses’ Health Study participants were collected simultaneously beginning in 2000.
Depression and antidepressant use were self-reported, and depressive symptoms were confirmed using the five-item Mental Health Inventory.
The findings are encouraging; depression and antidepressant use are common and both have been hypothesized to increase breast cancer risk. Some prior studies have found a link between either depression or antidepressant use and breast cancer, and others have not – but most have had important limitations, including retrospective design and inclusion of major depression only, among others, she said.
“To me, though, the most important limitation is that previous studies have not evaluated depression and antidepressant use together,” Dr. Reeves said.
In the current study, which did consider both, no evidence was seen to suggest that depression or antidepressant use affects breast cancer risk.
“It’s typically very unexciting to have to report null results, but in this case, I think this is excellent news and really the best we could have hoped for,” she said, adding that although they are preliminary, the findings should be “very reassuring for the millions of women with depression and/or those using antidepressants.”
“Depression is a very serious medical condition. It deserves to be treated, and it’s nice that these women can take the antidepressants, which so effectively treat this condition without worry that they’re doing something that would adversely affect their breast cancer risk in the future,” she concluded, noting that more sophisticated analyses of the data are planned to consider additional variables, including treatment duration. The analyses will also be repeated in the Nurses’ Health Study II cohort, which is a younger cohort with a higher incidence of premenopausal breast cancer and a greater prevalence of both depression and antidepressant use.
AT THE ASPO ANNUAL MEETING
Key clinical point: Depression and antidepressant use do not appear to increase breast cancer risk.
Major finding: No statistically significant associations were seen between either invasive or in situ breast cancer and depression or antidepressant use.
Data source: 67,120 women fromthe Nurses’ Health Study.
Disclosures: The investigator reported no conflicts.
Colonoscopy follow-up varied widely after positive fecal blood test
BIRMINGHAM, ALA.– The time to colonoscopy after a positive fecal occult blood test varied widely between health systems and also varied based on age and comorbidity score, in a study of more than 62,000 patients from four health systems
The median time to colonoscopy after a positive fecal occult blood test (FOBT) in 62,384 patients in the Population-based Research Optimizing Screening through Personalized Regimens (PROSPR) Network ranged from 41 to 174 days. Most of those who received a follow-up colonoscopy did so within 3-6 months of their positive FOBT.
The percentage of patients followed up with colonoscopy within 12 months ranged from 58.1% to 83.8%, with lowest percentages occurring in the two systems with the longest median time to follow-up, Jessica Chubak, Ph.D., of Group Health Research Institute and her colleagues reported in a poster at the American Society of Preventive Oncology annual meeting.
The rate of colonoscopy follow-up within 12 months also decreased with advancing age and increasing Charlson comorbidity score. For those aged 55-59 years, 60-64 years, 65-69 years, or 70-75 years, the hazard ratios for follow-up were 1.02, 0.98, 0.98, and 0.90, respectively, compared with the youngest age group (50-54 years). But for those patients aged 76-84 years or 85-89 years, the hazard ratios were 0.65 and 0.34, respectively.
For those with a Charlson comorbidity score of 1, 2, or 3 or more, the adjusted hazard ratios for follow-up of were 0.93, 0.87, and 0.70, respectively, compared with those with a score of 0, the investigators said.
No significant differences in follow-up were seen based on gender, body mass index, or race/ethnicity.
The investigators used administrative and clinical data to estimate the time to follow-up and probability of follow-up for all persons with a positive FOBT in 2011 and 2012.
The findings have implications for future research on improving follow-up in older patients and those with comorbidities, the study authors said.
The differences between health care systems may be due to varying practices. The two organizations with the best time to colonoscopy follow-up in the network are health maintenance organizations with targets for time to colonoscopy and monitored appointment supply.
In contrast, the organization with the second longest time to follow-up was an HMO that contracted with external providers for about 60% of colonoscopies. The organization with the longest time to follow-up was a safety-net system with limited colonoscopy capacity that served socioeconomically disadvantaged patients who may have faced more barriers to colonoscopy completion.
Colorectal cancer is the fourth most common cancer in the United States, and FOBT is an important screening strategy that relies on follow-up in the event of a positive finding.
“Our findings that both individual-level factors as well as health care system were associated with follow-up strengthens the rationale for investigating multilevel interventions to improve follow-up after abnormal screening tests,” the researchers concluded.
The National Cancer Institute funded the study.
BIRMINGHAM, ALA.– The time to colonoscopy after a positive fecal occult blood test varied widely between health systems and also varied based on age and comorbidity score, in a study of more than 62,000 patients from four health systems
The median time to colonoscopy after a positive fecal occult blood test (FOBT) in 62,384 patients in the Population-based Research Optimizing Screening through Personalized Regimens (PROSPR) Network ranged from 41 to 174 days. Most of those who received a follow-up colonoscopy did so within 3-6 months of their positive FOBT.
The percentage of patients followed up with colonoscopy within 12 months ranged from 58.1% to 83.8%, with lowest percentages occurring in the two systems with the longest median time to follow-up, Jessica Chubak, Ph.D., of Group Health Research Institute and her colleagues reported in a poster at the American Society of Preventive Oncology annual meeting.
The rate of colonoscopy follow-up within 12 months also decreased with advancing age and increasing Charlson comorbidity score. For those aged 55-59 years, 60-64 years, 65-69 years, or 70-75 years, the hazard ratios for follow-up were 1.02, 0.98, 0.98, and 0.90, respectively, compared with the youngest age group (50-54 years). But for those patients aged 76-84 years or 85-89 years, the hazard ratios were 0.65 and 0.34, respectively.
For those with a Charlson comorbidity score of 1, 2, or 3 or more, the adjusted hazard ratios for follow-up of were 0.93, 0.87, and 0.70, respectively, compared with those with a score of 0, the investigators said.
No significant differences in follow-up were seen based on gender, body mass index, or race/ethnicity.
The investigators used administrative and clinical data to estimate the time to follow-up and probability of follow-up for all persons with a positive FOBT in 2011 and 2012.
The findings have implications for future research on improving follow-up in older patients and those with comorbidities, the study authors said.
The differences between health care systems may be due to varying practices. The two organizations with the best time to colonoscopy follow-up in the network are health maintenance organizations with targets for time to colonoscopy and monitored appointment supply.
In contrast, the organization with the second longest time to follow-up was an HMO that contracted with external providers for about 60% of colonoscopies. The organization with the longest time to follow-up was a safety-net system with limited colonoscopy capacity that served socioeconomically disadvantaged patients who may have faced more barriers to colonoscopy completion.
Colorectal cancer is the fourth most common cancer in the United States, and FOBT is an important screening strategy that relies on follow-up in the event of a positive finding.
“Our findings that both individual-level factors as well as health care system were associated with follow-up strengthens the rationale for investigating multilevel interventions to improve follow-up after abnormal screening tests,” the researchers concluded.
The National Cancer Institute funded the study.
BIRMINGHAM, ALA.– The time to colonoscopy after a positive fecal occult blood test varied widely between health systems and also varied based on age and comorbidity score, in a study of more than 62,000 patients from four health systems
The median time to colonoscopy after a positive fecal occult blood test (FOBT) in 62,384 patients in the Population-based Research Optimizing Screening through Personalized Regimens (PROSPR) Network ranged from 41 to 174 days. Most of those who received a follow-up colonoscopy did so within 3-6 months of their positive FOBT.
The percentage of patients followed up with colonoscopy within 12 months ranged from 58.1% to 83.8%, with lowest percentages occurring in the two systems with the longest median time to follow-up, Jessica Chubak, Ph.D., of Group Health Research Institute and her colleagues reported in a poster at the American Society of Preventive Oncology annual meeting.
The rate of colonoscopy follow-up within 12 months also decreased with advancing age and increasing Charlson comorbidity score. For those aged 55-59 years, 60-64 years, 65-69 years, or 70-75 years, the hazard ratios for follow-up were 1.02, 0.98, 0.98, and 0.90, respectively, compared with the youngest age group (50-54 years). But for those patients aged 76-84 years or 85-89 years, the hazard ratios were 0.65 and 0.34, respectively.
For those with a Charlson comorbidity score of 1, 2, or 3 or more, the adjusted hazard ratios for follow-up of were 0.93, 0.87, and 0.70, respectively, compared with those with a score of 0, the investigators said.
No significant differences in follow-up were seen based on gender, body mass index, or race/ethnicity.
The investigators used administrative and clinical data to estimate the time to follow-up and probability of follow-up for all persons with a positive FOBT in 2011 and 2012.
The findings have implications for future research on improving follow-up in older patients and those with comorbidities, the study authors said.
The differences between health care systems may be due to varying practices. The two organizations with the best time to colonoscopy follow-up in the network are health maintenance organizations with targets for time to colonoscopy and monitored appointment supply.
In contrast, the organization with the second longest time to follow-up was an HMO that contracted with external providers for about 60% of colonoscopies. The organization with the longest time to follow-up was a safety-net system with limited colonoscopy capacity that served socioeconomically disadvantaged patients who may have faced more barriers to colonoscopy completion.
Colorectal cancer is the fourth most common cancer in the United States, and FOBT is an important screening strategy that relies on follow-up in the event of a positive finding.
“Our findings that both individual-level factors as well as health care system were associated with follow-up strengthens the rationale for investigating multilevel interventions to improve follow-up after abnormal screening tests,” the researchers concluded.
The National Cancer Institute funded the study.
AT THE ASPO ANNUAL MEETING
Key clinical point: Increasing age and comorbidities reduce the likelihood of colonoscopy follow-up for a positive fecal occult blood test.
Major finding: The percentage of patients followed up with colonoscopy within 12 months ranged from 58.1% to 83.8%.
Data source: An analysis of administrative and clinical data from four health systems with more than 62,000 patients in the PROSPR Network.
Disclosures: The National Cancer Institute funded the study.
‘Tailored navigation’ ups colorectal cancer screening adherence
BIRMINGHAM, ALA. – Determining whether patients prefer colonoscopy or a stool sample blood test for colorectal cancer screening, and then providing support for completing the preferred method, could increase screening rates and reduce racial disparities in screening, according to findings from two randomized, controlled trials.
Such “tailored navigation” was superior to a standard intervention in the first of the two trials, which compared these approaches with usual care in general-population primary care patients from 10 practices. The patients were considered nonadherent, because they had not undergone colorectal cancer screening.
Screening adherence at 6 months was highest at 38% in 312 patients randomized to the tailored navigation intervention, compared with 33% in 316 patients randomized to receive the standard intervention. Adherence was only 12% among 317 patients in the usual care control group, Ronald E. Myers, Ph.D., of Thomas Jefferson University, Philadelphia, said at the annual meeting of the American Society of Preventive Oncology.
That study, by Dr. Myers and his colleagues, was published in 2013 (Cancer Epidemiol. Biomarkers Prev. 2013;22:109-17).
Patients in the tailored navigation group were sent either colonoscopy instructions or stool blood tests, depending on their reported test preference at baseline. If they had no preference, they received both. All patients in that group also received a mailed reminder and a navigation call to help encourage test use.
The standard intervention group was sent both colonoscopy instructions and stool blood tests, as well as a mailed reminder.
A total of 42% of patients had no preference regarding type of screening test, 39% preferred colonoscopy, and 19% preferred stool testing. “That may be a surprise, but their primary care physicians are not pushing stool blood testing,” Dr. Myers said. “They are pushing and promoting colonoscopy.”
The difference in screening adherence was significant for both the tailored intervention and standard intervention groups, compared with the control group, but was not significant between the two intervention groups.
A secondary analysis showed that mailing the colonoscopy instructions and blood test had a substantial effect on overall screening, and that the navigation calls also had an effect on overall screening and on colonoscopy.
Patients who were mailed only the colonoscopy instructions were much less likely to do stool blood testing – largely because it wasn’t being offered in their primary care practice. However, those who received both colonoscopy instructions and a stool blood test were much more likely to do stool blood testing.
In a similar study involving only noncompliant African American primary care patients, Dr. Myers and his colleagues found that adherence at 6 months was significantly greater in 384 patients randomized to tailored navigation than in 380 patients who were randomized to the standard intervention (odds ratio, 2.1), and positive change in overall screening preference was also significantly greater in the tailored navigation group (odds ratio, 1.5).
As in the first study, the tailored intervention in the second study (J. Natl. Cancer Inst. 2014;106:pii:dju344) involved a mailed stool blood test kit or colonoscopy instructions based on patient preference. But those with no preference received only colonoscopy instructions rather than receiving both instructions and a stool blood test.
All those in the tailored navigation groups received a telephone call to encourage screening, as well as a mailed reminder. The standard intervention group received a mailed stool blood test kit, colonoscopy instructions, and a mailed reminder.
The test preferences in the second study were similar to those in the first study, with 57% having no preference, 25% of patients preferring colonoscopy, and 18% preferring stool blood testing. A survey at 6 months and a medical records review at 12 months were conducted to assess adherence, changes in screening preference, and perceptions about screening.
Despite those with no preference receiving only the colonoscopy instructions, the tailored navigation group had a significantly higher screening rate than did the standard intervention group (38% vs. 24%), Dr. Myer noted.
The higher adherence was attributed to a high screening rate (50%) among those who preferred and received the stool blood test and navigation calls. The adherence rate was 35% in those who received the colonoscopy instructions and the navigation call.
“We believe it’s the case that mailing both stool blood test and colonoscopy materials in this case increased overall adherence, but that was mostly due to stool blood testing,” Dr. Myer said. Tailored navigation increased overall adherence primarily by boosting both stool blood testing and colonoscopy.
“Preference actually influenced, not necessarily overall screening, but what type of test was performed,” he added, noting that among African Americans, tailored navigation had a greater effect on overall adherence than did the mailed stool blood test and colonoscopy materials.
“We believe that tailored navigation with mailed stool blood testing and colonoscopy materials for everyone may increase screening adherence and also reduce screening disparity,” Dr. Myer noted, adding that it appears best to offer both tests, identify the preferred test, and provide support for the performance of that preferred test.
Additional research is needed to determine how to maximize the effects of tailored navigation and to determine its impact on racial disparities in colorectal cancer outcomes, Dr. Myer said. A study similar in design to those conducted in general and African American populations is underway in a Hispanic population, he added. Dr. Myers reported having no disclosures.
BIRMINGHAM, ALA. – Determining whether patients prefer colonoscopy or a stool sample blood test for colorectal cancer screening, and then providing support for completing the preferred method, could increase screening rates and reduce racial disparities in screening, according to findings from two randomized, controlled trials.
Such “tailored navigation” was superior to a standard intervention in the first of the two trials, which compared these approaches with usual care in general-population primary care patients from 10 practices. The patients were considered nonadherent, because they had not undergone colorectal cancer screening.
Screening adherence at 6 months was highest at 38% in 312 patients randomized to the tailored navigation intervention, compared with 33% in 316 patients randomized to receive the standard intervention. Adherence was only 12% among 317 patients in the usual care control group, Ronald E. Myers, Ph.D., of Thomas Jefferson University, Philadelphia, said at the annual meeting of the American Society of Preventive Oncology.
That study, by Dr. Myers and his colleagues, was published in 2013 (Cancer Epidemiol. Biomarkers Prev. 2013;22:109-17).
Patients in the tailored navigation group were sent either colonoscopy instructions or stool blood tests, depending on their reported test preference at baseline. If they had no preference, they received both. All patients in that group also received a mailed reminder and a navigation call to help encourage test use.
The standard intervention group was sent both colonoscopy instructions and stool blood tests, as well as a mailed reminder.
A total of 42% of patients had no preference regarding type of screening test, 39% preferred colonoscopy, and 19% preferred stool testing. “That may be a surprise, but their primary care physicians are not pushing stool blood testing,” Dr. Myers said. “They are pushing and promoting colonoscopy.”
The difference in screening adherence was significant for both the tailored intervention and standard intervention groups, compared with the control group, but was not significant between the two intervention groups.
A secondary analysis showed that mailing the colonoscopy instructions and blood test had a substantial effect on overall screening, and that the navigation calls also had an effect on overall screening and on colonoscopy.
Patients who were mailed only the colonoscopy instructions were much less likely to do stool blood testing – largely because it wasn’t being offered in their primary care practice. However, those who received both colonoscopy instructions and a stool blood test were much more likely to do stool blood testing.
In a similar study involving only noncompliant African American primary care patients, Dr. Myers and his colleagues found that adherence at 6 months was significantly greater in 384 patients randomized to tailored navigation than in 380 patients who were randomized to the standard intervention (odds ratio, 2.1), and positive change in overall screening preference was also significantly greater in the tailored navigation group (odds ratio, 1.5).
As in the first study, the tailored intervention in the second study (J. Natl. Cancer Inst. 2014;106:pii:dju344) involved a mailed stool blood test kit or colonoscopy instructions based on patient preference. But those with no preference received only colonoscopy instructions rather than receiving both instructions and a stool blood test.
All those in the tailored navigation groups received a telephone call to encourage screening, as well as a mailed reminder. The standard intervention group received a mailed stool blood test kit, colonoscopy instructions, and a mailed reminder.
The test preferences in the second study were similar to those in the first study, with 57% having no preference, 25% of patients preferring colonoscopy, and 18% preferring stool blood testing. A survey at 6 months and a medical records review at 12 months were conducted to assess adherence, changes in screening preference, and perceptions about screening.
Despite those with no preference receiving only the colonoscopy instructions, the tailored navigation group had a significantly higher screening rate than did the standard intervention group (38% vs. 24%), Dr. Myer noted.
The higher adherence was attributed to a high screening rate (50%) among those who preferred and received the stool blood test and navigation calls. The adherence rate was 35% in those who received the colonoscopy instructions and the navigation call.
“We believe it’s the case that mailing both stool blood test and colonoscopy materials in this case increased overall adherence, but that was mostly due to stool blood testing,” Dr. Myer said. Tailored navigation increased overall adherence primarily by boosting both stool blood testing and colonoscopy.
“Preference actually influenced, not necessarily overall screening, but what type of test was performed,” he added, noting that among African Americans, tailored navigation had a greater effect on overall adherence than did the mailed stool blood test and colonoscopy materials.
“We believe that tailored navigation with mailed stool blood testing and colonoscopy materials for everyone may increase screening adherence and also reduce screening disparity,” Dr. Myer noted, adding that it appears best to offer both tests, identify the preferred test, and provide support for the performance of that preferred test.
Additional research is needed to determine how to maximize the effects of tailored navigation and to determine its impact on racial disparities in colorectal cancer outcomes, Dr. Myer said. A study similar in design to those conducted in general and African American populations is underway in a Hispanic population, he added. Dr. Myers reported having no disclosures.
BIRMINGHAM, ALA. – Determining whether patients prefer colonoscopy or a stool sample blood test for colorectal cancer screening, and then providing support for completing the preferred method, could increase screening rates and reduce racial disparities in screening, according to findings from two randomized, controlled trials.
Such “tailored navigation” was superior to a standard intervention in the first of the two trials, which compared these approaches with usual care in general-population primary care patients from 10 practices. The patients were considered nonadherent, because they had not undergone colorectal cancer screening.
Screening adherence at 6 months was highest at 38% in 312 patients randomized to the tailored navigation intervention, compared with 33% in 316 patients randomized to receive the standard intervention. Adherence was only 12% among 317 patients in the usual care control group, Ronald E. Myers, Ph.D., of Thomas Jefferson University, Philadelphia, said at the annual meeting of the American Society of Preventive Oncology.
That study, by Dr. Myers and his colleagues, was published in 2013 (Cancer Epidemiol. Biomarkers Prev. 2013;22:109-17).
Patients in the tailored navigation group were sent either colonoscopy instructions or stool blood tests, depending on their reported test preference at baseline. If they had no preference, they received both. All patients in that group also received a mailed reminder and a navigation call to help encourage test use.
The standard intervention group was sent both colonoscopy instructions and stool blood tests, as well as a mailed reminder.
A total of 42% of patients had no preference regarding type of screening test, 39% preferred colonoscopy, and 19% preferred stool testing. “That may be a surprise, but their primary care physicians are not pushing stool blood testing,” Dr. Myers said. “They are pushing and promoting colonoscopy.”
The difference in screening adherence was significant for both the tailored intervention and standard intervention groups, compared with the control group, but was not significant between the two intervention groups.
A secondary analysis showed that mailing the colonoscopy instructions and blood test had a substantial effect on overall screening, and that the navigation calls also had an effect on overall screening and on colonoscopy.
Patients who were mailed only the colonoscopy instructions were much less likely to do stool blood testing – largely because it wasn’t being offered in their primary care practice. However, those who received both colonoscopy instructions and a stool blood test were much more likely to do stool blood testing.
In a similar study involving only noncompliant African American primary care patients, Dr. Myers and his colleagues found that adherence at 6 months was significantly greater in 384 patients randomized to tailored navigation than in 380 patients who were randomized to the standard intervention (odds ratio, 2.1), and positive change in overall screening preference was also significantly greater in the tailored navigation group (odds ratio, 1.5).
As in the first study, the tailored intervention in the second study (J. Natl. Cancer Inst. 2014;106:pii:dju344) involved a mailed stool blood test kit or colonoscopy instructions based on patient preference. But those with no preference received only colonoscopy instructions rather than receiving both instructions and a stool blood test.
All those in the tailored navigation groups received a telephone call to encourage screening, as well as a mailed reminder. The standard intervention group received a mailed stool blood test kit, colonoscopy instructions, and a mailed reminder.
The test preferences in the second study were similar to those in the first study, with 57% having no preference, 25% of patients preferring colonoscopy, and 18% preferring stool blood testing. A survey at 6 months and a medical records review at 12 months were conducted to assess adherence, changes in screening preference, and perceptions about screening.
Despite those with no preference receiving only the colonoscopy instructions, the tailored navigation group had a significantly higher screening rate than did the standard intervention group (38% vs. 24%), Dr. Myer noted.
The higher adherence was attributed to a high screening rate (50%) among those who preferred and received the stool blood test and navigation calls. The adherence rate was 35% in those who received the colonoscopy instructions and the navigation call.
“We believe it’s the case that mailing both stool blood test and colonoscopy materials in this case increased overall adherence, but that was mostly due to stool blood testing,” Dr. Myer said. Tailored navigation increased overall adherence primarily by boosting both stool blood testing and colonoscopy.
“Preference actually influenced, not necessarily overall screening, but what type of test was performed,” he added, noting that among African Americans, tailored navigation had a greater effect on overall adherence than did the mailed stool blood test and colonoscopy materials.
“We believe that tailored navigation with mailed stool blood testing and colonoscopy materials for everyone may increase screening adherence and also reduce screening disparity,” Dr. Myer noted, adding that it appears best to offer both tests, identify the preferred test, and provide support for the performance of that preferred test.
Additional research is needed to determine how to maximize the effects of tailored navigation and to determine its impact on racial disparities in colorectal cancer outcomes, Dr. Myer said. A study similar in design to those conducted in general and African American populations is underway in a Hispanic population, he added. Dr. Myers reported having no disclosures.
AT THE ASPO ANNUAL MEETING
Key clinical point: Providing patients with colorectal cancer screening options and support could improve adherence and reduce racial disparities.
Major finding: Screening adherence was best with tailored navigation (38% at 6 months) in both the general and African American populations studied.
Data source: Two randomized, controlled trials involving 945 and 764 patients, respectively.
Disclosures: Dr. Myers reported having no disclosures.
Several factors predict postbariatric surgery readmission
Bariatric surgery is generally safe and readmissions are rare, but prolonged operative time, operation complexity, and major postoperative complications are among several risk factors for readmission identified in a large retrospective cohort.
Of 18,186 patients from the 2012 American College of Surgeons National Surgical Quality Improvement program (ACS NSQIP) database who had bariatric surgery as a primary procedure, 5% were readmitted. Of 815 patients with any major complication, 31% were readmitted. Factors found on multivariate analysis to significantly predict readmission within 30 days were age, sex, body mass index, American Society of Anesthesiology (ASA) risk class, diabetes status, hypertension, and steroid use, Dr. Christa R. Abraham of Albany (N.Y.) Medical College and her colleagues reported online in the Journal of the American College of Surgeons.
Further, all major postoperative complications were significant predictors of readmission, including bleeding requiring transfusion, urinary tract infections, and superficial surgical site infection (SSI). Other significant predictors were deep SSI, organ space SSI, wound disruption, pneumonia, unplanned intubation, mechanical ventilation for more than 48 hours, pulmonary embolism, deep vein thrombosis, and sepsis, the investigators said (J. Am. Coll. Surg. 2015 [doi:10.1016/j.jamcollsurg.2015.02.018]).
Of the patients included in the study, 1,819 had a laparoscopic gastric band, 9,613 had laparoscopic Roux-en-Y gastric bypass, 6,439 had gastroplasties, and 315 had open Roux-en-Y gastric bypass. All had a BMI of at least 30 kg/m2, and had a postsurgery length of stay of 14 days or fewer. Most were ASA risk class 3 or lower, and most were functionally independent.
Complications were more common with laparoscopic and open Roux-en-y gastric bypass (5.5% and 11.8%, respectively) rather than with gastroplasty and sleeve (3.4%) and laparoscopic banding (1.4%).
The findings are of value, because while bariatric surgery is a low-risk procedure, and it is extremely common; in 2013 there were 179,000 such surgeries performed in the United States.
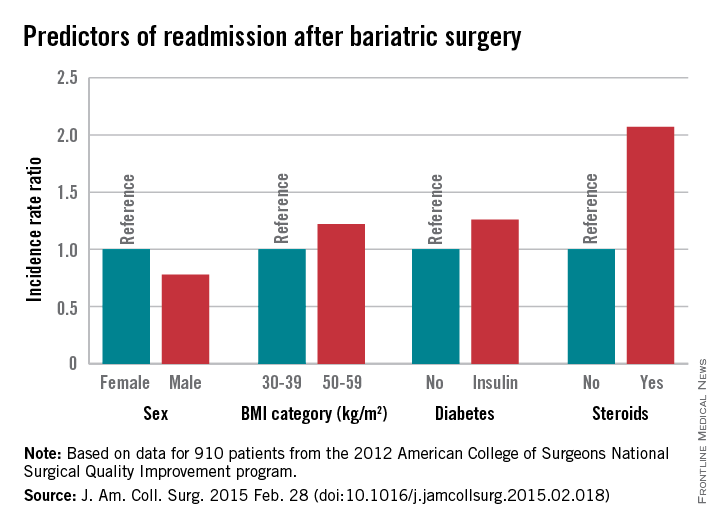
“Bariatric surgery is one of the fastest-growing surgical interest areas, making analysis of patient outcomes and reasons for readmission important,” the investigators explained.
The ability to identify high-risk patients could allow for targeted interventions to prevent readmission, they said.
For example, steroid use, which was identified as a risk factor in the current study, is modifiable.
“In our practice, steroids are discontinued for 6 weeks prior to bariatric surgery and patients who are steroid dependent are unlikely to undergo bariatric surgery,” they said.
Additionally, they “try to minimize readmission for patients with infections by treating with antibiotics following operation and continuing antibiotics at discharge.”
The investigators noted that the ACS NSQIP MORBPROB (estimated probability of morbidity) tool is a good tool for predicting readmission among prospective bariatric patients, although it may not fully capture the effect of preexisting conditions.
“These data led us to change our own practice by risk-stratifying patients with higher ASA and BMI to consider surgical options, and to begin early surveillance soon after discharge,” they said.
The authors reported having no disclosures.
Bariatric surgery is generally safe and readmissions are rare, but prolonged operative time, operation complexity, and major postoperative complications are among several risk factors for readmission identified in a large retrospective cohort.
Of 18,186 patients from the 2012 American College of Surgeons National Surgical Quality Improvement program (ACS NSQIP) database who had bariatric surgery as a primary procedure, 5% were readmitted. Of 815 patients with any major complication, 31% were readmitted. Factors found on multivariate analysis to significantly predict readmission within 30 days were age, sex, body mass index, American Society of Anesthesiology (ASA) risk class, diabetes status, hypertension, and steroid use, Dr. Christa R. Abraham of Albany (N.Y.) Medical College and her colleagues reported online in the Journal of the American College of Surgeons.
Further, all major postoperative complications were significant predictors of readmission, including bleeding requiring transfusion, urinary tract infections, and superficial surgical site infection (SSI). Other significant predictors were deep SSI, organ space SSI, wound disruption, pneumonia, unplanned intubation, mechanical ventilation for more than 48 hours, pulmonary embolism, deep vein thrombosis, and sepsis, the investigators said (J. Am. Coll. Surg. 2015 [doi:10.1016/j.jamcollsurg.2015.02.018]).
Of the patients included in the study, 1,819 had a laparoscopic gastric band, 9,613 had laparoscopic Roux-en-Y gastric bypass, 6,439 had gastroplasties, and 315 had open Roux-en-Y gastric bypass. All had a BMI of at least 30 kg/m2, and had a postsurgery length of stay of 14 days or fewer. Most were ASA risk class 3 or lower, and most were functionally independent.
Complications were more common with laparoscopic and open Roux-en-y gastric bypass (5.5% and 11.8%, respectively) rather than with gastroplasty and sleeve (3.4%) and laparoscopic banding (1.4%).
The findings are of value, because while bariatric surgery is a low-risk procedure, and it is extremely common; in 2013 there were 179,000 such surgeries performed in the United States.

“Bariatric surgery is one of the fastest-growing surgical interest areas, making analysis of patient outcomes and reasons for readmission important,” the investigators explained.
The ability to identify high-risk patients could allow for targeted interventions to prevent readmission, they said.
For example, steroid use, which was identified as a risk factor in the current study, is modifiable.
“In our practice, steroids are discontinued for 6 weeks prior to bariatric surgery and patients who are steroid dependent are unlikely to undergo bariatric surgery,” they said.
Additionally, they “try to minimize readmission for patients with infections by treating with antibiotics following operation and continuing antibiotics at discharge.”
The investigators noted that the ACS NSQIP MORBPROB (estimated probability of morbidity) tool is a good tool for predicting readmission among prospective bariatric patients, although it may not fully capture the effect of preexisting conditions.
“These data led us to change our own practice by risk-stratifying patients with higher ASA and BMI to consider surgical options, and to begin early surveillance soon after discharge,” they said.
The authors reported having no disclosures.
Bariatric surgery is generally safe and readmissions are rare, but prolonged operative time, operation complexity, and major postoperative complications are among several risk factors for readmission identified in a large retrospective cohort.
Of 18,186 patients from the 2012 American College of Surgeons National Surgical Quality Improvement program (ACS NSQIP) database who had bariatric surgery as a primary procedure, 5% were readmitted. Of 815 patients with any major complication, 31% were readmitted. Factors found on multivariate analysis to significantly predict readmission within 30 days were age, sex, body mass index, American Society of Anesthesiology (ASA) risk class, diabetes status, hypertension, and steroid use, Dr. Christa R. Abraham of Albany (N.Y.) Medical College and her colleagues reported online in the Journal of the American College of Surgeons.
Further, all major postoperative complications were significant predictors of readmission, including bleeding requiring transfusion, urinary tract infections, and superficial surgical site infection (SSI). Other significant predictors were deep SSI, organ space SSI, wound disruption, pneumonia, unplanned intubation, mechanical ventilation for more than 48 hours, pulmonary embolism, deep vein thrombosis, and sepsis, the investigators said (J. Am. Coll. Surg. 2015 [doi:10.1016/j.jamcollsurg.2015.02.018]).
Of the patients included in the study, 1,819 had a laparoscopic gastric band, 9,613 had laparoscopic Roux-en-Y gastric bypass, 6,439 had gastroplasties, and 315 had open Roux-en-Y gastric bypass. All had a BMI of at least 30 kg/m2, and had a postsurgery length of stay of 14 days or fewer. Most were ASA risk class 3 or lower, and most were functionally independent.
Complications were more common with laparoscopic and open Roux-en-y gastric bypass (5.5% and 11.8%, respectively) rather than with gastroplasty and sleeve (3.4%) and laparoscopic banding (1.4%).
The findings are of value, because while bariatric surgery is a low-risk procedure, and it is extremely common; in 2013 there were 179,000 such surgeries performed in the United States.

“Bariatric surgery is one of the fastest-growing surgical interest areas, making analysis of patient outcomes and reasons for readmission important,” the investigators explained.
The ability to identify high-risk patients could allow for targeted interventions to prevent readmission, they said.
For example, steroid use, which was identified as a risk factor in the current study, is modifiable.
“In our practice, steroids are discontinued for 6 weeks prior to bariatric surgery and patients who are steroid dependent are unlikely to undergo bariatric surgery,” they said.
Additionally, they “try to minimize readmission for patients with infections by treating with antibiotics following operation and continuing antibiotics at discharge.”
The investigators noted that the ACS NSQIP MORBPROB (estimated probability of morbidity) tool is a good tool for predicting readmission among prospective bariatric patients, although it may not fully capture the effect of preexisting conditions.
“These data led us to change our own practice by risk-stratifying patients with higher ASA and BMI to consider surgical options, and to begin early surveillance soon after discharge,” they said.
The authors reported having no disclosures.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF SURGEONS
Key clinical point: Knowing risk factors for readmission after bariatric surgery can allow for targeted interventions.
Major finding: Steroid use is among several risk factors for readmission following bariatric surgery (incidence rate ratio, 2.07)
Data source: A retrospective cohort study involving 18,186 patients.
Disclosures: The authors reported having no disclosures.