User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Indiana HIV outbreak prompts national advisory
As health officials continue to battle an intravenous drug use–related HIV outbreak in a rural county in Indiana, the Centers for Disease Control and Prevention has issued an official health advisory stressing the need for vigilance at the state and local levels with respect to detecting and controlling similar outbreaks in other communities across the United States.
The Indiana outbreak began in November, with 11 new HIV infections diagnosed by January in Scott County, where fewer than 5 infections per year had been identified previously; since January, an unprecedented 135 infections have been confirmed, and 6 others are under investigation, according to Dr. Jerome M. Adams, Indiana State Health Commissioner.
“To put this in further perspective, from 2009 through 2013, the county only reported three new cases of HIV,” Dr. Adams said in a joint Indiana State Department of Health/CDC teleconference.
New cases are being reported every day on an hourly basis, he added.
The affected community includes only 4,200 people, and 84% of those diagnosed with HIV also tested positive for hepatitis C virus (HCV) infection, Dr. Joan Duwve, chief medical consultant, Indiana State Department of Health, said during the teleconference.
According to the CDC advisory, 96% of 108 infected individuals who were interviewed reported injecting dissolved prescription-type oxymorphone, as well as sharing syringes. The advisory states that “urgent action is needed to prevent further HIV and HCV transmission in this area and to investigate and control any similar outbreaks in other communities.”
Although the national incidence of HIV infections has declined markedly – by about 90% – since peaking in the 1990s, about 50,000 new infections occur each year in the United States, and about 8% of those are associated with intravenous drug use.
“The United States is facing an epidemic of prescription opioid abuse that must be addressed. Opioid poisoning deaths have nearly quadrupled from 1999 through 2011. This epidemic of opioid abuse has already contributed to a severe and growing epidemic of viral hepatitis among people who inject drugs,” said Dr. Jonathan Mermin, director of the National Center for HIV/AIDS Viral Hepatitis, STD, and TB Prevention, who also participated in the teleconference.
New CDC data on HCV show a 150% increase in reports of acute HCV infections nationwide between 2010 to 2013, he said.
“The majority of these infections are believed to be attributable to injection drug use, so we must act now to reverse this trend and to prevent this from undoing progress in HIV prevention to date,” he said.
To identify and control outbreaks quickly, the CDC made several recommendations for both health departments and healthcare providers.
Health departments should review the most recent sources of data on HIV and HCV diagnoses, overdose deaths, admissions for drug treatment, and drug arrests, looking specifically for recent increases in the number of HIV infections attributed to intravenous drug use, and the number of HCV infections among those aged 35 years and younger.
These, as well as high rates of prescription-type opioid abuse and overdose, drug treatment admissions, or drug arrests, are attributes of communities at risk for unrecognized clusters of infections, according to the advisory.
Other recommendations for health departments include ensuring complete contact tracing for new diagnoses and testing of all contacts, ensuring that people who inject drugs and those at high risk of drug injection have access to integrated prevention services, and reminding venues such as emergency departments and community-based clinical practices of the importance of routine opt-out of HIV testing as well as HCV testing per current recommendations.
Additionally, local health department should notify state health departments and the CDC of any suspected clusters of HIV or HCV infection.
Likewise, health care professionals should report suspected clusters to their local or state health department, ensure that HCV-infected patients are also tested for HIV and vice versa, and ensure that those receiving treatment for either HIV or HCV are adhering to prescribed therapy and are engaged in ongoing care.
Syringe-sharing and sexual partners of those diagnosed with HIV or HCV should be encouraged to undergo testing, and providers should report all newly diagnosed infections to the health department, and should refer all persons with substance abuse problems for medication-assisted treatment and counseling. Effective treatments should be used as appropriately indicated.
The risks and benefits of all pain treatment options, and the fact that long-term opioid therapy is not associated with reduced chronic pain, should be discussed with any person for whom opioids are under consideration for pain management, according to the advisory.
In Indiana, where a public health emergency was declared on March 26 and has been extended until May 24, the only local health care provider in the county where all the cases occurred, along with local health officials, law enforcement, community partners, regional health care providers, and the CDC, provided a comprehensive response, including a public education campaign, establishment of an incident command center and community outreach center, short-term authorization of syringe exchange, and support for comprehensive medical care. The care includes HIV and HCV treatment, as well as substance abuse counseling and treatment, according to a “Notes From the Field Report” in the Morbidity and Mortality Weekly Report (MMWR 2015 April 24;64 [early release]:1-2).
“State and local health departments and academic partners, with the assistance of CDC, are working to implement and improve the community outreach programs supported by the executive order and to interrupt injection drug use-related HIV and hepatitis C virus transmission. Contact tracing by state and CDC disease intervention specialists continues to identify those potentially exposed,” the report states.
During the teleconference, Dr. Duwve described the response in detail, noting that the HIV treatment clinic is staffed by Indiana University infectious disease physicians and is supported by the Indiana State Department of Health and local partners.
Testing and treatment services are also provided at the county jail, she said.
“The outbreak highlights the vulnerability of many rural, resource poor populations to drug use, misuse, and addiction. This outbreak also demonstrates the importance of timely HIV surveillance activity and rapid response to interrupt disease transmission,” she said, noting that it also points to the need for expanded mental health and substance use treatment programs in medically under-served rural areas.
Dr. Mermin of the CDC added that “the situation in Indiana should serve as a warning that we cannot let down our guard against these deadly infections.”
As health officials continue to battle an intravenous drug use–related HIV outbreak in a rural county in Indiana, the Centers for Disease Control and Prevention has issued an official health advisory stressing the need for vigilance at the state and local levels with respect to detecting and controlling similar outbreaks in other communities across the United States.
The Indiana outbreak began in November, with 11 new HIV infections diagnosed by January in Scott County, where fewer than 5 infections per year had been identified previously; since January, an unprecedented 135 infections have been confirmed, and 6 others are under investigation, according to Dr. Jerome M. Adams, Indiana State Health Commissioner.
“To put this in further perspective, from 2009 through 2013, the county only reported three new cases of HIV,” Dr. Adams said in a joint Indiana State Department of Health/CDC teleconference.
New cases are being reported every day on an hourly basis, he added.
The affected community includes only 4,200 people, and 84% of those diagnosed with HIV also tested positive for hepatitis C virus (HCV) infection, Dr. Joan Duwve, chief medical consultant, Indiana State Department of Health, said during the teleconference.
According to the CDC advisory, 96% of 108 infected individuals who were interviewed reported injecting dissolved prescription-type oxymorphone, as well as sharing syringes. The advisory states that “urgent action is needed to prevent further HIV and HCV transmission in this area and to investigate and control any similar outbreaks in other communities.”
Although the national incidence of HIV infections has declined markedly – by about 90% – since peaking in the 1990s, about 50,000 new infections occur each year in the United States, and about 8% of those are associated with intravenous drug use.
“The United States is facing an epidemic of prescription opioid abuse that must be addressed. Opioid poisoning deaths have nearly quadrupled from 1999 through 2011. This epidemic of opioid abuse has already contributed to a severe and growing epidemic of viral hepatitis among people who inject drugs,” said Dr. Jonathan Mermin, director of the National Center for HIV/AIDS Viral Hepatitis, STD, and TB Prevention, who also participated in the teleconference.
New CDC data on HCV show a 150% increase in reports of acute HCV infections nationwide between 2010 to 2013, he said.
“The majority of these infections are believed to be attributable to injection drug use, so we must act now to reverse this trend and to prevent this from undoing progress in HIV prevention to date,” he said.
To identify and control outbreaks quickly, the CDC made several recommendations for both health departments and healthcare providers.
Health departments should review the most recent sources of data on HIV and HCV diagnoses, overdose deaths, admissions for drug treatment, and drug arrests, looking specifically for recent increases in the number of HIV infections attributed to intravenous drug use, and the number of HCV infections among those aged 35 years and younger.
These, as well as high rates of prescription-type opioid abuse and overdose, drug treatment admissions, or drug arrests, are attributes of communities at risk for unrecognized clusters of infections, according to the advisory.
Other recommendations for health departments include ensuring complete contact tracing for new diagnoses and testing of all contacts, ensuring that people who inject drugs and those at high risk of drug injection have access to integrated prevention services, and reminding venues such as emergency departments and community-based clinical practices of the importance of routine opt-out of HIV testing as well as HCV testing per current recommendations.
Additionally, local health department should notify state health departments and the CDC of any suspected clusters of HIV or HCV infection.
Likewise, health care professionals should report suspected clusters to their local or state health department, ensure that HCV-infected patients are also tested for HIV and vice versa, and ensure that those receiving treatment for either HIV or HCV are adhering to prescribed therapy and are engaged in ongoing care.
Syringe-sharing and sexual partners of those diagnosed with HIV or HCV should be encouraged to undergo testing, and providers should report all newly diagnosed infections to the health department, and should refer all persons with substance abuse problems for medication-assisted treatment and counseling. Effective treatments should be used as appropriately indicated.
The risks and benefits of all pain treatment options, and the fact that long-term opioid therapy is not associated with reduced chronic pain, should be discussed with any person for whom opioids are under consideration for pain management, according to the advisory.
In Indiana, where a public health emergency was declared on March 26 and has been extended until May 24, the only local health care provider in the county where all the cases occurred, along with local health officials, law enforcement, community partners, regional health care providers, and the CDC, provided a comprehensive response, including a public education campaign, establishment of an incident command center and community outreach center, short-term authorization of syringe exchange, and support for comprehensive medical care. The care includes HIV and HCV treatment, as well as substance abuse counseling and treatment, according to a “Notes From the Field Report” in the Morbidity and Mortality Weekly Report (MMWR 2015 April 24;64 [early release]:1-2).
“State and local health departments and academic partners, with the assistance of CDC, are working to implement and improve the community outreach programs supported by the executive order and to interrupt injection drug use-related HIV and hepatitis C virus transmission. Contact tracing by state and CDC disease intervention specialists continues to identify those potentially exposed,” the report states.
During the teleconference, Dr. Duwve described the response in detail, noting that the HIV treatment clinic is staffed by Indiana University infectious disease physicians and is supported by the Indiana State Department of Health and local partners.
Testing and treatment services are also provided at the county jail, she said.
“The outbreak highlights the vulnerability of many rural, resource poor populations to drug use, misuse, and addiction. This outbreak also demonstrates the importance of timely HIV surveillance activity and rapid response to interrupt disease transmission,” she said, noting that it also points to the need for expanded mental health and substance use treatment programs in medically under-served rural areas.
Dr. Mermin of the CDC added that “the situation in Indiana should serve as a warning that we cannot let down our guard against these deadly infections.”
As health officials continue to battle an intravenous drug use–related HIV outbreak in a rural county in Indiana, the Centers for Disease Control and Prevention has issued an official health advisory stressing the need for vigilance at the state and local levels with respect to detecting and controlling similar outbreaks in other communities across the United States.
The Indiana outbreak began in November, with 11 new HIV infections diagnosed by January in Scott County, where fewer than 5 infections per year had been identified previously; since January, an unprecedented 135 infections have been confirmed, and 6 others are under investigation, according to Dr. Jerome M. Adams, Indiana State Health Commissioner.
“To put this in further perspective, from 2009 through 2013, the county only reported three new cases of HIV,” Dr. Adams said in a joint Indiana State Department of Health/CDC teleconference.
New cases are being reported every day on an hourly basis, he added.
The affected community includes only 4,200 people, and 84% of those diagnosed with HIV also tested positive for hepatitis C virus (HCV) infection, Dr. Joan Duwve, chief medical consultant, Indiana State Department of Health, said during the teleconference.
According to the CDC advisory, 96% of 108 infected individuals who were interviewed reported injecting dissolved prescription-type oxymorphone, as well as sharing syringes. The advisory states that “urgent action is needed to prevent further HIV and HCV transmission in this area and to investigate and control any similar outbreaks in other communities.”
Although the national incidence of HIV infections has declined markedly – by about 90% – since peaking in the 1990s, about 50,000 new infections occur each year in the United States, and about 8% of those are associated with intravenous drug use.
“The United States is facing an epidemic of prescription opioid abuse that must be addressed. Opioid poisoning deaths have nearly quadrupled from 1999 through 2011. This epidemic of opioid abuse has already contributed to a severe and growing epidemic of viral hepatitis among people who inject drugs,” said Dr. Jonathan Mermin, director of the National Center for HIV/AIDS Viral Hepatitis, STD, and TB Prevention, who also participated in the teleconference.
New CDC data on HCV show a 150% increase in reports of acute HCV infections nationwide between 2010 to 2013, he said.
“The majority of these infections are believed to be attributable to injection drug use, so we must act now to reverse this trend and to prevent this from undoing progress in HIV prevention to date,” he said.
To identify and control outbreaks quickly, the CDC made several recommendations for both health departments and healthcare providers.
Health departments should review the most recent sources of data on HIV and HCV diagnoses, overdose deaths, admissions for drug treatment, and drug arrests, looking specifically for recent increases in the number of HIV infections attributed to intravenous drug use, and the number of HCV infections among those aged 35 years and younger.
These, as well as high rates of prescription-type opioid abuse and overdose, drug treatment admissions, or drug arrests, are attributes of communities at risk for unrecognized clusters of infections, according to the advisory.
Other recommendations for health departments include ensuring complete contact tracing for new diagnoses and testing of all contacts, ensuring that people who inject drugs and those at high risk of drug injection have access to integrated prevention services, and reminding venues such as emergency departments and community-based clinical practices of the importance of routine opt-out of HIV testing as well as HCV testing per current recommendations.
Additionally, local health department should notify state health departments and the CDC of any suspected clusters of HIV or HCV infection.
Likewise, health care professionals should report suspected clusters to their local or state health department, ensure that HCV-infected patients are also tested for HIV and vice versa, and ensure that those receiving treatment for either HIV or HCV are adhering to prescribed therapy and are engaged in ongoing care.
Syringe-sharing and sexual partners of those diagnosed with HIV or HCV should be encouraged to undergo testing, and providers should report all newly diagnosed infections to the health department, and should refer all persons with substance abuse problems for medication-assisted treatment and counseling. Effective treatments should be used as appropriately indicated.
The risks and benefits of all pain treatment options, and the fact that long-term opioid therapy is not associated with reduced chronic pain, should be discussed with any person for whom opioids are under consideration for pain management, according to the advisory.
In Indiana, where a public health emergency was declared on March 26 and has been extended until May 24, the only local health care provider in the county where all the cases occurred, along with local health officials, law enforcement, community partners, regional health care providers, and the CDC, provided a comprehensive response, including a public education campaign, establishment of an incident command center and community outreach center, short-term authorization of syringe exchange, and support for comprehensive medical care. The care includes HIV and HCV treatment, as well as substance abuse counseling and treatment, according to a “Notes From the Field Report” in the Morbidity and Mortality Weekly Report (MMWR 2015 April 24;64 [early release]:1-2).
“State and local health departments and academic partners, with the assistance of CDC, are working to implement and improve the community outreach programs supported by the executive order and to interrupt injection drug use-related HIV and hepatitis C virus transmission. Contact tracing by state and CDC disease intervention specialists continues to identify those potentially exposed,” the report states.
During the teleconference, Dr. Duwve described the response in detail, noting that the HIV treatment clinic is staffed by Indiana University infectious disease physicians and is supported by the Indiana State Department of Health and local partners.
Testing and treatment services are also provided at the county jail, she said.
“The outbreak highlights the vulnerability of many rural, resource poor populations to drug use, misuse, and addiction. This outbreak also demonstrates the importance of timely HIV surveillance activity and rapid response to interrupt disease transmission,” she said, noting that it also points to the need for expanded mental health and substance use treatment programs in medically under-served rural areas.
Dr. Mermin of the CDC added that “the situation in Indiana should serve as a warning that we cannot let down our guard against these deadly infections.”
May 2015: Click for Credit
Here are 5 articles in the May issue of Clinician Reviews (accreditation valid until January 1, 2016):
1. Clindamycin, TMP-SMX Are Equally Effective for Skin Infections
To take the posttest, go to: http://bit.ly/1cVxR85
VITALS
Key clinical point: Clindamycin and TMP-SMX had similar efficacy and adverse-effect profiles for treating uncomplicated skin infections, including both abscesses and cellulitis.
Major finding: At 7-10 days after therapy completion, the rates of cure in the evaluable population were 89.5% with clindamycin and 88.2% with TMP-SMX.
Data source: A prospective, multicenter, randomized, double-blind clinical trial involving 524 adults and children followed for 1 month after treatment.
Disclosures: This trial was supported by the National Institutes of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences (NCT00730028). Dr Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
2. Hormone Therapy 10 Years Postmenopause Increases Risks
To take the posttest, go to: http://bit.ly/1OBYcUe
VITALS
Key clinical point: Hormone therapy in postmenopausal women increases stroke risk.
Major finding: Stroke increased by 24%, venous thromboembolism by 92%, and pulmonary embolism by 81% in postmenopausal women receiving hormone therapy.
Data source: A review and meta-analysis of 19 randomized controlled trials involving 40,140 postmenopausal women who received orally administered hormone therapy, placebo, or no treatment for prevention of cardiovascular disease.
Disclosures: One study was funded by Wyeth-Ayerst. Two studies received partial funding from Novo-Nordisk Pharmaceutical, and one study was funded by the National Institutes of Health with support from Wyeth-Ayerst, Hoffman-La Roche, Pharmacia, and Upjohn. Eight other studies used medication provided by various pharmaceutical companies.
3. "Perfect storm" of Depression, Stress Raises Risk for MI, Death
To take the posttest, go to: http://bit.ly/1yM2HtF
VITALS
Key clinical point: Concurrent depression and stress in coronary heart disease patients may increase early risk for MI and death.
Major finding: CHD patients with high depressive symptoms and high stress at baseline had an increased risk for MI and death early during follow-up (adjusted HR, 1.48).
Data source: A prospective cohort study of 4,487 adults.
Disclosures: The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr Alcántara reported having no disclosures; two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
4. Type 2 Diabetes Lower in Familial Hypercholesterolemia
To take the posttest, go to: http://bit.ly/1bplTDc
VITALS
Key clinical point: The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives.
Major finding: The prevalence of type 2 diabetes was 1.75% in 25,137 patients with familial hypercholesterolemia, compared with 2.93% in 38,183 of their unaffected relatives.
Data source: An observational cross-sectional analysis of data for 63,320 people in the Dutch national registry of familial hypercholesterolemia.
Disclosures: The study sponsor was not specified; the familial hypercholesterolemia registry is subsidized by the Dutch government. Dr Besseling reported having no financial disclosures; his associates reported ties to Aegerion, Amgen, AstraZeneca, Boehringer Ingelheim, Cerenis, Eli Lilly, Genzyme, JSiS, MSD, Novartis, Pfizer, Regeneron, Roche, and Sanofi.
5. Mongersen Induces 55%-65% Remission Rates in Crohn’s
To take the posttest, go to: http://bit.ly/1DctonL
VITALS
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized, placebo-controlled, double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sirtris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
Here are 5 articles in the May issue of Clinician Reviews (accreditation valid until January 1, 2016):
1. Clindamycin, TMP-SMX Are Equally Effective for Skin Infections
To take the posttest, go to: http://bit.ly/1cVxR85
VITALS
Key clinical point: Clindamycin and TMP-SMX had similar efficacy and adverse-effect profiles for treating uncomplicated skin infections, including both abscesses and cellulitis.
Major finding: At 7-10 days after therapy completion, the rates of cure in the evaluable population were 89.5% with clindamycin and 88.2% with TMP-SMX.
Data source: A prospective, multicenter, randomized, double-blind clinical trial involving 524 adults and children followed for 1 month after treatment.
Disclosures: This trial was supported by the National Institutes of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences (NCT00730028). Dr Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
2. Hormone Therapy 10 Years Postmenopause Increases Risks
To take the posttest, go to: http://bit.ly/1OBYcUe
VITALS
Key clinical point: Hormone therapy in postmenopausal women increases stroke risk.
Major finding: Stroke increased by 24%, venous thromboembolism by 92%, and pulmonary embolism by 81% in postmenopausal women receiving hormone therapy.
Data source: A review and meta-analysis of 19 randomized controlled trials involving 40,140 postmenopausal women who received orally administered hormone therapy, placebo, or no treatment for prevention of cardiovascular disease.
Disclosures: One study was funded by Wyeth-Ayerst. Two studies received partial funding from Novo-Nordisk Pharmaceutical, and one study was funded by the National Institutes of Health with support from Wyeth-Ayerst, Hoffman-La Roche, Pharmacia, and Upjohn. Eight other studies used medication provided by various pharmaceutical companies.
3. "Perfect storm" of Depression, Stress Raises Risk for MI, Death
To take the posttest, go to: http://bit.ly/1yM2HtF
VITALS
Key clinical point: Concurrent depression and stress in coronary heart disease patients may increase early risk for MI and death.
Major finding: CHD patients with high depressive symptoms and high stress at baseline had an increased risk for MI and death early during follow-up (adjusted HR, 1.48).
Data source: A prospective cohort study of 4,487 adults.
Disclosures: The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr Alcántara reported having no disclosures; two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
4. Type 2 Diabetes Lower in Familial Hypercholesterolemia
To take the posttest, go to: http://bit.ly/1bplTDc
VITALS
Key clinical point: The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives.
Major finding: The prevalence of type 2 diabetes was 1.75% in 25,137 patients with familial hypercholesterolemia, compared with 2.93% in 38,183 of their unaffected relatives.
Data source: An observational cross-sectional analysis of data for 63,320 people in the Dutch national registry of familial hypercholesterolemia.
Disclosures: The study sponsor was not specified; the familial hypercholesterolemia registry is subsidized by the Dutch government. Dr Besseling reported having no financial disclosures; his associates reported ties to Aegerion, Amgen, AstraZeneca, Boehringer Ingelheim, Cerenis, Eli Lilly, Genzyme, JSiS, MSD, Novartis, Pfizer, Regeneron, Roche, and Sanofi.
5. Mongersen Induces 55%-65% Remission Rates in Crohn’s
To take the posttest, go to: http://bit.ly/1DctonL
VITALS
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized, placebo-controlled, double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sirtris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
Here are 5 articles in the May issue of Clinician Reviews (accreditation valid until January 1, 2016):
1. Clindamycin, TMP-SMX Are Equally Effective for Skin Infections
To take the posttest, go to: http://bit.ly/1cVxR85
VITALS
Key clinical point: Clindamycin and TMP-SMX had similar efficacy and adverse-effect profiles for treating uncomplicated skin infections, including both abscesses and cellulitis.
Major finding: At 7-10 days after therapy completion, the rates of cure in the evaluable population were 89.5% with clindamycin and 88.2% with TMP-SMX.
Data source: A prospective, multicenter, randomized, double-blind clinical trial involving 524 adults and children followed for 1 month after treatment.
Disclosures: This trial was supported by the National Institutes of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences (NCT00730028). Dr Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
2. Hormone Therapy 10 Years Postmenopause Increases Risks
To take the posttest, go to: http://bit.ly/1OBYcUe
VITALS
Key clinical point: Hormone therapy in postmenopausal women increases stroke risk.
Major finding: Stroke increased by 24%, venous thromboembolism by 92%, and pulmonary embolism by 81% in postmenopausal women receiving hormone therapy.
Data source: A review and meta-analysis of 19 randomized controlled trials involving 40,140 postmenopausal women who received orally administered hormone therapy, placebo, or no treatment for prevention of cardiovascular disease.
Disclosures: One study was funded by Wyeth-Ayerst. Two studies received partial funding from Novo-Nordisk Pharmaceutical, and one study was funded by the National Institutes of Health with support from Wyeth-Ayerst, Hoffman-La Roche, Pharmacia, and Upjohn. Eight other studies used medication provided by various pharmaceutical companies.
3. "Perfect storm" of Depression, Stress Raises Risk for MI, Death
To take the posttest, go to: http://bit.ly/1yM2HtF
VITALS
Key clinical point: Concurrent depression and stress in coronary heart disease patients may increase early risk for MI and death.
Major finding: CHD patients with high depressive symptoms and high stress at baseline had an increased risk for MI and death early during follow-up (adjusted HR, 1.48).
Data source: A prospective cohort study of 4,487 adults.
Disclosures: The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr Alcántara reported having no disclosures; two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
4. Type 2 Diabetes Lower in Familial Hypercholesterolemia
To take the posttest, go to: http://bit.ly/1bplTDc
VITALS
Key clinical point: The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives.
Major finding: The prevalence of type 2 diabetes was 1.75% in 25,137 patients with familial hypercholesterolemia, compared with 2.93% in 38,183 of their unaffected relatives.
Data source: An observational cross-sectional analysis of data for 63,320 people in the Dutch national registry of familial hypercholesterolemia.
Disclosures: The study sponsor was not specified; the familial hypercholesterolemia registry is subsidized by the Dutch government. Dr Besseling reported having no financial disclosures; his associates reported ties to Aegerion, Amgen, AstraZeneca, Boehringer Ingelheim, Cerenis, Eli Lilly, Genzyme, JSiS, MSD, Novartis, Pfizer, Regeneron, Roche, and Sanofi.
5. Mongersen Induces 55%-65% Remission Rates in Crohn’s
To take the posttest, go to: http://bit.ly/1DctonL
VITALS
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized, placebo-controlled, double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sirtris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
OTC phototherapy devices can enhance outcomes
MIAMI BEACH – Over-the-counter phototherapy devices for anti-aging and acne treatment can improve office-based procedure results and enhance clinical practice, according to Dr. Z. Paul Lorenc.
Light-emitting diode (LED) devices now available for home use, such as the illuMask (La Lumiere) anti-aging and anti-acne masks, are safe and effective and can be used along with office-based treatments to speed healing and boost results, Dr. Lorenc said at the South Beach Symposium.
The illuMask anti-aging mask uses a combination of 630 nm wavelength red light and 855 nm wavelength infrared light, and the two work synergistically to improve signs of aging, such as wrinkles, photoaging, and hyperpigmentation.
Dr. Lorenc, chief medical and scientific officer for La Lumiere, worked on the pivotal trials for the devices, and said that in a study of 30 patients aged 14 -40 years, 15 minutes of daily mask use was associated with 82% and 94% improvement in 17 clinical skin attributes at weeks 4 and 8, respectively. Improvement in 20 sham-treated subjects was 24% at both 4 and 8 weeks.
Subject self-assessment showed significant improvement in 93% and 100% of 15 clinical attributes at weeks 4 and 8, respectively, in the treated subjects, compared with 33% and 40% in the sham-treated subjects, he said.
The illuMask anti-acne mask uses 630 nm red light and 440 nm blue light, which also work synergistically.
In addition, 15 minutes of daily use was associated with a 71% average decrease in the number of noninflammatory lesions and an 83% decrease in the number of inflammatory lesions at 8 weeks. The median decrease in the 5-point Acne Severity Assessment Scale score was 2 points – from 4 (moderate) to 2 (almost clear).
Dr. Lorenc, who practices plastic surgery in New York, said he does not treat acne very often, but he does encounter patients with adult acne who benefit from the acne mask.
He said he uses the anti-aging mask often.
“I’m using it on every single resurfacing patient,” he said, explaining that red light is anti-inflammatory in nature, and increases production of collagen and elastin.
Patients undergoing partial or total resurfacing, those undergoing other procedures such as microneedling, and even those undergoing a face-lift, can benefit from use of the anti-aging mask beginning the day after the procedure, he said, adding that it decreases inflammatory processes, and can provide some added anti-aging effects that may not be addressed by the initial procedures (dyschromia in face-lift patients, for example).
The masks are simple to use, don’t take much time, can be used with existing treatment regimens, are appropriate for all Fitzpatrick skin types, and are associated with a high compliance rate – even among teens, Dr. Lorenc said.
While some physicians have expressed concern that the OTC availability of phototherapy devices could hurt clinical practice, his experience has been quite the opposite.
The boost that the masks give both to the recovery time and efficacy of office-based procedures makes for happier patients who come back – and send their friends, he said.
“I have incorporated [the masks] into my practice, and what it has done is the opposite of what you would think intuitively; it has expanded my practice – both surgical and nonsurgical, and we think that this is a very good adjunct therapy to both acne and anti-aging treatment,” he said.
Dr. Lorenc had no additional disclosures.
MIAMI BEACH – Over-the-counter phototherapy devices for anti-aging and acne treatment can improve office-based procedure results and enhance clinical practice, according to Dr. Z. Paul Lorenc.
Light-emitting diode (LED) devices now available for home use, such as the illuMask (La Lumiere) anti-aging and anti-acne masks, are safe and effective and can be used along with office-based treatments to speed healing and boost results, Dr. Lorenc said at the South Beach Symposium.
The illuMask anti-aging mask uses a combination of 630 nm wavelength red light and 855 nm wavelength infrared light, and the two work synergistically to improve signs of aging, such as wrinkles, photoaging, and hyperpigmentation.
Dr. Lorenc, chief medical and scientific officer for La Lumiere, worked on the pivotal trials for the devices, and said that in a study of 30 patients aged 14 -40 years, 15 minutes of daily mask use was associated with 82% and 94% improvement in 17 clinical skin attributes at weeks 4 and 8, respectively. Improvement in 20 sham-treated subjects was 24% at both 4 and 8 weeks.
Subject self-assessment showed significant improvement in 93% and 100% of 15 clinical attributes at weeks 4 and 8, respectively, in the treated subjects, compared with 33% and 40% in the sham-treated subjects, he said.
The illuMask anti-acne mask uses 630 nm red light and 440 nm blue light, which also work synergistically.
In addition, 15 minutes of daily use was associated with a 71% average decrease in the number of noninflammatory lesions and an 83% decrease in the number of inflammatory lesions at 8 weeks. The median decrease in the 5-point Acne Severity Assessment Scale score was 2 points – from 4 (moderate) to 2 (almost clear).
Dr. Lorenc, who practices plastic surgery in New York, said he does not treat acne very often, but he does encounter patients with adult acne who benefit from the acne mask.
He said he uses the anti-aging mask often.
“I’m using it on every single resurfacing patient,” he said, explaining that red light is anti-inflammatory in nature, and increases production of collagen and elastin.
Patients undergoing partial or total resurfacing, those undergoing other procedures such as microneedling, and even those undergoing a face-lift, can benefit from use of the anti-aging mask beginning the day after the procedure, he said, adding that it decreases inflammatory processes, and can provide some added anti-aging effects that may not be addressed by the initial procedures (dyschromia in face-lift patients, for example).
The masks are simple to use, don’t take much time, can be used with existing treatment regimens, are appropriate for all Fitzpatrick skin types, and are associated with a high compliance rate – even among teens, Dr. Lorenc said.
While some physicians have expressed concern that the OTC availability of phototherapy devices could hurt clinical practice, his experience has been quite the opposite.
The boost that the masks give both to the recovery time and efficacy of office-based procedures makes for happier patients who come back – and send their friends, he said.
“I have incorporated [the masks] into my practice, and what it has done is the opposite of what you would think intuitively; it has expanded my practice – both surgical and nonsurgical, and we think that this is a very good adjunct therapy to both acne and anti-aging treatment,” he said.
Dr. Lorenc had no additional disclosures.
MIAMI BEACH – Over-the-counter phototherapy devices for anti-aging and acne treatment can improve office-based procedure results and enhance clinical practice, according to Dr. Z. Paul Lorenc.
Light-emitting diode (LED) devices now available for home use, such as the illuMask (La Lumiere) anti-aging and anti-acne masks, are safe and effective and can be used along with office-based treatments to speed healing and boost results, Dr. Lorenc said at the South Beach Symposium.
The illuMask anti-aging mask uses a combination of 630 nm wavelength red light and 855 nm wavelength infrared light, and the two work synergistically to improve signs of aging, such as wrinkles, photoaging, and hyperpigmentation.
Dr. Lorenc, chief medical and scientific officer for La Lumiere, worked on the pivotal trials for the devices, and said that in a study of 30 patients aged 14 -40 years, 15 minutes of daily mask use was associated with 82% and 94% improvement in 17 clinical skin attributes at weeks 4 and 8, respectively. Improvement in 20 sham-treated subjects was 24% at both 4 and 8 weeks.
Subject self-assessment showed significant improvement in 93% and 100% of 15 clinical attributes at weeks 4 and 8, respectively, in the treated subjects, compared with 33% and 40% in the sham-treated subjects, he said.
The illuMask anti-acne mask uses 630 nm red light and 440 nm blue light, which also work synergistically.
In addition, 15 minutes of daily use was associated with a 71% average decrease in the number of noninflammatory lesions and an 83% decrease in the number of inflammatory lesions at 8 weeks. The median decrease in the 5-point Acne Severity Assessment Scale score was 2 points – from 4 (moderate) to 2 (almost clear).
Dr. Lorenc, who practices plastic surgery in New York, said he does not treat acne very often, but he does encounter patients with adult acne who benefit from the acne mask.
He said he uses the anti-aging mask often.
“I’m using it on every single resurfacing patient,” he said, explaining that red light is anti-inflammatory in nature, and increases production of collagen and elastin.
Patients undergoing partial or total resurfacing, those undergoing other procedures such as microneedling, and even those undergoing a face-lift, can benefit from use of the anti-aging mask beginning the day after the procedure, he said, adding that it decreases inflammatory processes, and can provide some added anti-aging effects that may not be addressed by the initial procedures (dyschromia in face-lift patients, for example).
The masks are simple to use, don’t take much time, can be used with existing treatment regimens, are appropriate for all Fitzpatrick skin types, and are associated with a high compliance rate – even among teens, Dr. Lorenc said.
While some physicians have expressed concern that the OTC availability of phototherapy devices could hurt clinical practice, his experience has been quite the opposite.
The boost that the masks give both to the recovery time and efficacy of office-based procedures makes for happier patients who come back – and send their friends, he said.
“I have incorporated [the masks] into my practice, and what it has done is the opposite of what you would think intuitively; it has expanded my practice – both surgical and nonsurgical, and we think that this is a very good adjunct therapy to both acne and anti-aging treatment,” he said.
Dr. Lorenc had no additional disclosures.
Push for family planning SAM gains momentum
About 25% of family physicians continue to do obstetric deliveries, while an estimated 75%-90% prescribe birth control.
Yet, while a self-assessment module (SAM) exists to test knowledge in maternity care, the same is not true for family planning. Thus, the creation of a family planning SAM is overdue, according to Dr. Linda Prine.
At press time, 958 family physicians agreed, as evidenced by their signatures on a letter urging the American Board of Family Medicine (ABFM) to develop a family planning SAM.
The letter was initiated by the Reproductive Health Care Member Interest Group of the American Academy of Family Physicians, which is chaired by Dr. Prine. The group has written a proposal for the SAM, assembled a team of volunteer physicians to work on the SAM, and launched the petition to demonstrate interest, which is a required first step in the process. As a result, the group is well on its way to reaching its goal of obtaining 1,000 signatures.
“Family planning is an integral part of family medicine care. Most women of reproductive age decide to have an average of two children during their childbearing years. During the rest of those years, many will need to use contraception,” Dr. Prine of Mount Sinai Hospital, New York, said in an interview.
Recent studies have found that there is a shortage of family physicians who are knowledgeable and trained in all aspects of family planning, especially long-acting contraceptives such as intrauterine devices and implants. A family planning SAM may motivate family doctors to become more up to date in newer contraceptive methods and at least recommend them, if not provide them to patients, she said.
Providing family planning services – as just one part of the overall cradle-to-grave care of patients and their children, partners, and parents – is what family medicine is all about, she added.
Indeed, according to the AAFP, family physicians play a critical role in family planning.
“On any given day, family physicians all over the country are inserting intrauterine devices and progesterone implants and performing vasectomies,” said Dr. Jennifer Frost, medical director of the Health of the Public and Science division at the AAFP.
The AAFP also has a policy promoting the use of long-acting reversible contraceptive (LARC) methods.
“Family physicians are well trained in this area and provide these services to patients every day. In fact, the insertion of LARCs is an integral part of our residency training,” Dr. Frost said, adding that the array of contraceptive methods available and the increasing amount of data on the safety and efficacy of various options requires that family physicians be knowledgeable about the options available for those who want to prevent pregnancy, and about providing preconception care to those who want to become pregnant.
A recent Centers for Disease Control and Prevention report stated that nearly all women use contraception at some point in their lifetimes. The report, published in December, cited data from the 2011-2013 National Survey of Family Growth showing that nearly 62% of the 60.9 million women aged 15-44 years in 2011-2013 were currently using contraception, and that most used oral contraceptive pills (16%), female sterilization (15.5%), male condoms (9.4%), and LARCs (7.2%).
These data underscore the importance of competency with respect to all aspects of family planning, Dr. Prine said.
The availability of a family planning SAM for those who desire to improve their knowledge of contraceptive technologies would not only provide family physicians with an important tool for keeping up to date on contraceptive methods and issues regarding family planning but would show that the specialty recognizes the importance of the topic and follows evidence-based practices, she said.
Dr. Prine continues to collect signatures. Once more than 1,000 supporters have signed, the petition will be delivered to the ABFM for consideration.
The petition, which is addressed to Dr. Michael D. Hagen, ABFM senior vice president, states in part that the undersigned “see women and couples to discuss family planning needs with great frequency,” and that family planning “is an area of care that is core to our practices, and one that has an increasingly large area of content as new products and devices are often emerging.”
The demonstration of interest and enthusiasm for creation of a SAM via the petition is just the first step in an involved and costly process, Dr. Hagen said in an interview.
Maintenance of certification for family physicians has four main components: professionalism, life-long learning, demonstration of cognitive expertise or knowledge via an examination taken every 7-10 years, and quality improvement.
SAMs fit into the second component – demonstration of cognitive expertise – and consist of both a knowledge assessment based on a 60-item multiple choice questionnaire on the specific content area and a clinical simulation, Dr. Hagen explained, noting that family physicians are required to complete SAMs of their choice periodically, in addition to other maintenance-of-certification requirements and accumulation of 150 continuing medical education credits every 3 years.
“So two processes have to occur for the creation of a SAM: the development of content for the knowledge assessment and development of a simulation model,” said Dr. Hagen, who is also with the department of family and community medicine at the University of Kentucky, Lexington.
The knowledge assessment would require not only applicable questions, but readily accessible references and critiques that provide a rationale for why an answer is correct or incorrect, and the simulation would require “quite a bit of development of information that we put into a knowledge base to generate a clinical scenario,” he said.
Typically, three to five family physicians, a content expert or specialist in the area being addressed, two ABFM editorial staff members, and one or two informaticists are involved in the process.
“The team is charged with creating a template of what the content should look like for a particular SAM, then the content areas are divided among the team members, who then write their respective items,” he said, noting that this process can take up to 9 months
The simulation aspect of the SAM is created at the same time, and in all – with necessary meetings and revisions – SAM finalization may take 12-15 months.
The process costs about $75,000 to $100,000, excluding periodic updates and other maintenance, so the decision to create a SAM is not made lightly, he said.
Dr. Prine said she hopes that the volunteer labor of the team of highly qualified family physicians she has brought together will keep the costs down for the ABFM.
Once the petition is delivered to the ABFM, the Maintenance of Certification Committee will review the matter, look at all of the related competencies, consider whether family planning is an area of importance with respect to content areas, and determine whether family physicians require educational materials in the area, Dr. Hagen said, noting that the topic is scheduled for review at the May ABFM meeting.
Should a family planning SAM be approved, it will join “medical genomics” as the first new SAMs created in nearly 4 years. SAMs have been used since about 2004, and at that time a decision was made to create SAMs for the top 20 areas of priority identified by the Institute of Medicine, including lung disease, diabetes, heart failure, hypertension, and depression, just to name a few. SAMs for those areas were developed over a decade, and the ABFM recently began entertaining possible new topics for development.
“Family planning obviously is a component of family medicine … How much interest there is among the 80,000-plus diplomates, I don’t know, but that’s one of the things the interest group is trying to assess and inform us about. We will look carefully at where this fits in the ongoing anticipated need for development,” he said.
Both Dr. Hagen and Dr. Prine reported having no relevant disclosures aside from their respective roles within the ABFM and the AAFP member interest group.
About 25% of family physicians continue to do obstetric deliveries, while an estimated 75%-90% prescribe birth control.
Yet, while a self-assessment module (SAM) exists to test knowledge in maternity care, the same is not true for family planning. Thus, the creation of a family planning SAM is overdue, according to Dr. Linda Prine.
At press time, 958 family physicians agreed, as evidenced by their signatures on a letter urging the American Board of Family Medicine (ABFM) to develop a family planning SAM.
The letter was initiated by the Reproductive Health Care Member Interest Group of the American Academy of Family Physicians, which is chaired by Dr. Prine. The group has written a proposal for the SAM, assembled a team of volunteer physicians to work on the SAM, and launched the petition to demonstrate interest, which is a required first step in the process. As a result, the group is well on its way to reaching its goal of obtaining 1,000 signatures.
“Family planning is an integral part of family medicine care. Most women of reproductive age decide to have an average of two children during their childbearing years. During the rest of those years, many will need to use contraception,” Dr. Prine of Mount Sinai Hospital, New York, said in an interview.
Recent studies have found that there is a shortage of family physicians who are knowledgeable and trained in all aspects of family planning, especially long-acting contraceptives such as intrauterine devices and implants. A family planning SAM may motivate family doctors to become more up to date in newer contraceptive methods and at least recommend them, if not provide them to patients, she said.
Providing family planning services – as just one part of the overall cradle-to-grave care of patients and their children, partners, and parents – is what family medicine is all about, she added.
Indeed, according to the AAFP, family physicians play a critical role in family planning.
“On any given day, family physicians all over the country are inserting intrauterine devices and progesterone implants and performing vasectomies,” said Dr. Jennifer Frost, medical director of the Health of the Public and Science division at the AAFP.
The AAFP also has a policy promoting the use of long-acting reversible contraceptive (LARC) methods.
“Family physicians are well trained in this area and provide these services to patients every day. In fact, the insertion of LARCs is an integral part of our residency training,” Dr. Frost said, adding that the array of contraceptive methods available and the increasing amount of data on the safety and efficacy of various options requires that family physicians be knowledgeable about the options available for those who want to prevent pregnancy, and about providing preconception care to those who want to become pregnant.
A recent Centers for Disease Control and Prevention report stated that nearly all women use contraception at some point in their lifetimes. The report, published in December, cited data from the 2011-2013 National Survey of Family Growth showing that nearly 62% of the 60.9 million women aged 15-44 years in 2011-2013 were currently using contraception, and that most used oral contraceptive pills (16%), female sterilization (15.5%), male condoms (9.4%), and LARCs (7.2%).
These data underscore the importance of competency with respect to all aspects of family planning, Dr. Prine said.
The availability of a family planning SAM for those who desire to improve their knowledge of contraceptive technologies would not only provide family physicians with an important tool for keeping up to date on contraceptive methods and issues regarding family planning but would show that the specialty recognizes the importance of the topic and follows evidence-based practices, she said.
Dr. Prine continues to collect signatures. Once more than 1,000 supporters have signed, the petition will be delivered to the ABFM for consideration.
The petition, which is addressed to Dr. Michael D. Hagen, ABFM senior vice president, states in part that the undersigned “see women and couples to discuss family planning needs with great frequency,” and that family planning “is an area of care that is core to our practices, and one that has an increasingly large area of content as new products and devices are often emerging.”
The demonstration of interest and enthusiasm for creation of a SAM via the petition is just the first step in an involved and costly process, Dr. Hagen said in an interview.
Maintenance of certification for family physicians has four main components: professionalism, life-long learning, demonstration of cognitive expertise or knowledge via an examination taken every 7-10 years, and quality improvement.
SAMs fit into the second component – demonstration of cognitive expertise – and consist of both a knowledge assessment based on a 60-item multiple choice questionnaire on the specific content area and a clinical simulation, Dr. Hagen explained, noting that family physicians are required to complete SAMs of their choice periodically, in addition to other maintenance-of-certification requirements and accumulation of 150 continuing medical education credits every 3 years.
“So two processes have to occur for the creation of a SAM: the development of content for the knowledge assessment and development of a simulation model,” said Dr. Hagen, who is also with the department of family and community medicine at the University of Kentucky, Lexington.
The knowledge assessment would require not only applicable questions, but readily accessible references and critiques that provide a rationale for why an answer is correct or incorrect, and the simulation would require “quite a bit of development of information that we put into a knowledge base to generate a clinical scenario,” he said.
Typically, three to five family physicians, a content expert or specialist in the area being addressed, two ABFM editorial staff members, and one or two informaticists are involved in the process.
“The team is charged with creating a template of what the content should look like for a particular SAM, then the content areas are divided among the team members, who then write their respective items,” he said, noting that this process can take up to 9 months
The simulation aspect of the SAM is created at the same time, and in all – with necessary meetings and revisions – SAM finalization may take 12-15 months.
The process costs about $75,000 to $100,000, excluding periodic updates and other maintenance, so the decision to create a SAM is not made lightly, he said.
Dr. Prine said she hopes that the volunteer labor of the team of highly qualified family physicians she has brought together will keep the costs down for the ABFM.
Once the petition is delivered to the ABFM, the Maintenance of Certification Committee will review the matter, look at all of the related competencies, consider whether family planning is an area of importance with respect to content areas, and determine whether family physicians require educational materials in the area, Dr. Hagen said, noting that the topic is scheduled for review at the May ABFM meeting.
Should a family planning SAM be approved, it will join “medical genomics” as the first new SAMs created in nearly 4 years. SAMs have been used since about 2004, and at that time a decision was made to create SAMs for the top 20 areas of priority identified by the Institute of Medicine, including lung disease, diabetes, heart failure, hypertension, and depression, just to name a few. SAMs for those areas were developed over a decade, and the ABFM recently began entertaining possible new topics for development.
“Family planning obviously is a component of family medicine … How much interest there is among the 80,000-plus diplomates, I don’t know, but that’s one of the things the interest group is trying to assess and inform us about. We will look carefully at where this fits in the ongoing anticipated need for development,” he said.
Both Dr. Hagen and Dr. Prine reported having no relevant disclosures aside from their respective roles within the ABFM and the AAFP member interest group.
About 25% of family physicians continue to do obstetric deliveries, while an estimated 75%-90% prescribe birth control.
Yet, while a self-assessment module (SAM) exists to test knowledge in maternity care, the same is not true for family planning. Thus, the creation of a family planning SAM is overdue, according to Dr. Linda Prine.
At press time, 958 family physicians agreed, as evidenced by their signatures on a letter urging the American Board of Family Medicine (ABFM) to develop a family planning SAM.
The letter was initiated by the Reproductive Health Care Member Interest Group of the American Academy of Family Physicians, which is chaired by Dr. Prine. The group has written a proposal for the SAM, assembled a team of volunteer physicians to work on the SAM, and launched the petition to demonstrate interest, which is a required first step in the process. As a result, the group is well on its way to reaching its goal of obtaining 1,000 signatures.
“Family planning is an integral part of family medicine care. Most women of reproductive age decide to have an average of two children during their childbearing years. During the rest of those years, many will need to use contraception,” Dr. Prine of Mount Sinai Hospital, New York, said in an interview.
Recent studies have found that there is a shortage of family physicians who are knowledgeable and trained in all aspects of family planning, especially long-acting contraceptives such as intrauterine devices and implants. A family planning SAM may motivate family doctors to become more up to date in newer contraceptive methods and at least recommend them, if not provide them to patients, she said.
Providing family planning services – as just one part of the overall cradle-to-grave care of patients and their children, partners, and parents – is what family medicine is all about, she added.
Indeed, according to the AAFP, family physicians play a critical role in family planning.
“On any given day, family physicians all over the country are inserting intrauterine devices and progesterone implants and performing vasectomies,” said Dr. Jennifer Frost, medical director of the Health of the Public and Science division at the AAFP.
The AAFP also has a policy promoting the use of long-acting reversible contraceptive (LARC) methods.
“Family physicians are well trained in this area and provide these services to patients every day. In fact, the insertion of LARCs is an integral part of our residency training,” Dr. Frost said, adding that the array of contraceptive methods available and the increasing amount of data on the safety and efficacy of various options requires that family physicians be knowledgeable about the options available for those who want to prevent pregnancy, and about providing preconception care to those who want to become pregnant.
A recent Centers for Disease Control and Prevention report stated that nearly all women use contraception at some point in their lifetimes. The report, published in December, cited data from the 2011-2013 National Survey of Family Growth showing that nearly 62% of the 60.9 million women aged 15-44 years in 2011-2013 were currently using contraception, and that most used oral contraceptive pills (16%), female sterilization (15.5%), male condoms (9.4%), and LARCs (7.2%).
These data underscore the importance of competency with respect to all aspects of family planning, Dr. Prine said.
The availability of a family planning SAM for those who desire to improve their knowledge of contraceptive technologies would not only provide family physicians with an important tool for keeping up to date on contraceptive methods and issues regarding family planning but would show that the specialty recognizes the importance of the topic and follows evidence-based practices, she said.
Dr. Prine continues to collect signatures. Once more than 1,000 supporters have signed, the petition will be delivered to the ABFM for consideration.
The petition, which is addressed to Dr. Michael D. Hagen, ABFM senior vice president, states in part that the undersigned “see women and couples to discuss family planning needs with great frequency,” and that family planning “is an area of care that is core to our practices, and one that has an increasingly large area of content as new products and devices are often emerging.”
The demonstration of interest and enthusiasm for creation of a SAM via the petition is just the first step in an involved and costly process, Dr. Hagen said in an interview.
Maintenance of certification for family physicians has four main components: professionalism, life-long learning, demonstration of cognitive expertise or knowledge via an examination taken every 7-10 years, and quality improvement.
SAMs fit into the second component – demonstration of cognitive expertise – and consist of both a knowledge assessment based on a 60-item multiple choice questionnaire on the specific content area and a clinical simulation, Dr. Hagen explained, noting that family physicians are required to complete SAMs of their choice periodically, in addition to other maintenance-of-certification requirements and accumulation of 150 continuing medical education credits every 3 years.
“So two processes have to occur for the creation of a SAM: the development of content for the knowledge assessment and development of a simulation model,” said Dr. Hagen, who is also with the department of family and community medicine at the University of Kentucky, Lexington.
The knowledge assessment would require not only applicable questions, but readily accessible references and critiques that provide a rationale for why an answer is correct or incorrect, and the simulation would require “quite a bit of development of information that we put into a knowledge base to generate a clinical scenario,” he said.
Typically, three to five family physicians, a content expert or specialist in the area being addressed, two ABFM editorial staff members, and one or two informaticists are involved in the process.
“The team is charged with creating a template of what the content should look like for a particular SAM, then the content areas are divided among the team members, who then write their respective items,” he said, noting that this process can take up to 9 months
The simulation aspect of the SAM is created at the same time, and in all – with necessary meetings and revisions – SAM finalization may take 12-15 months.
The process costs about $75,000 to $100,000, excluding periodic updates and other maintenance, so the decision to create a SAM is not made lightly, he said.
Dr. Prine said she hopes that the volunteer labor of the team of highly qualified family physicians she has brought together will keep the costs down for the ABFM.
Once the petition is delivered to the ABFM, the Maintenance of Certification Committee will review the matter, look at all of the related competencies, consider whether family planning is an area of importance with respect to content areas, and determine whether family physicians require educational materials in the area, Dr. Hagen said, noting that the topic is scheduled for review at the May ABFM meeting.
Should a family planning SAM be approved, it will join “medical genomics” as the first new SAMs created in nearly 4 years. SAMs have been used since about 2004, and at that time a decision was made to create SAMs for the top 20 areas of priority identified by the Institute of Medicine, including lung disease, diabetes, heart failure, hypertension, and depression, just to name a few. SAMs for those areas were developed over a decade, and the ABFM recently began entertaining possible new topics for development.
“Family planning obviously is a component of family medicine … How much interest there is among the 80,000-plus diplomates, I don’t know, but that’s one of the things the interest group is trying to assess and inform us about. We will look carefully at where this fits in the ongoing anticipated need for development,” he said.
Both Dr. Hagen and Dr. Prine reported having no relevant disclosures aside from their respective roles within the ABFM and the AAFP member interest group.
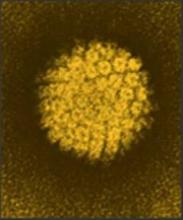
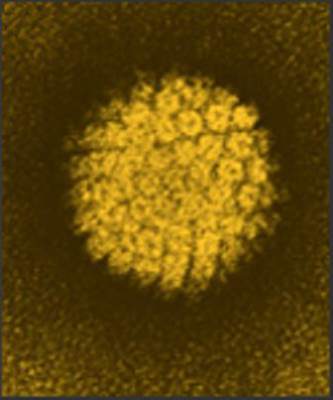
Serum antibodies don’t protect men against new oral HPV16
BIRMINGHAM, ALA. – Serum antibodies induced by a natural human papillomavirus type 16 infection do not appear to protect against acquisition of a subsequent oral HPV 16 infection in men, according to results of the HPV Infection in Men (HIM) study.
That finding is concerning, because about 70% of oropharyngeal cancers in the United States are caused by oral HPV infection. Most are caused by HPV type 16, Christine M. Pierce Campbell, Ph.D. of Moffitt Cancer Center & Research Institute, Tampa and her colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“HPV antibody status may reflect a man’s history of sexual behavior and partnerships, which may not be sufficiently measured in the risk factor questionnaire. In our study, the proportions of men who have sex with men and men who have sex with women and men who had high levels of antibodies at baseline were substantially greater than the proportion of heterosexual men with high antibody levels,” the researchers wrote. They noted that men with an antibody response may have been exposed to more HPV during their lifetimes, and thus were at greater risk of acquiring new oral HPV.
While naturally induced antibodies may not protect against oral HPV 16 infection, prophylactic HPV vaccination may provide such protection, the investigators said.
Of 1,618 men from the oral HPV 16 subcohort of the prospective natural history study of HPV who were followed for a median of 12.7 months after collection of an initial oral specimen, 17 developed a new incident oral HPV16 infection. Of these 17 men, 11 had low anti-HPV16 serum antibody levels, and 6 had high levels,
Anti-HPV 16 serum antibody levels were not associated with cumulative risk of oral HPV16 infection. During the first year of follow-up, 0.4% of those with low levels vs. 0.8% of those with high levels acquired a new infection. Within 24 months of follow-up, 3.0% of those with low levels vs. 3.5% of those with high levels acquired a new infection.
Those with higher levels were 60% more likely than those with low levels to acquire a new infection (hazard ratio, 1.6), but this finding was not statistically significant, the investigators reported.
Study subjects were men from the United States, Brazil, or Mexico who were aged 18 to 73 years and HIV negative at baseline.
Oral rinse-and-gargle specimens were collected from the men at 6-month intervals for up to 4 years. Low antibody levels were defined as the lowest 3 quartiles of antibody levels in the cohort combined, including those with no detectable antibody, and high levels were defined as the highest quartile.
Because of the rarity of oral HPV, additional study is needed to replicate the findings, they said.
Dr. Campbell was supported in part by a postdoctoral fellowship from the American Cancer Society. The HIM study was supported by a grant from the National Cancer Institute (NCI), by grants and/or other funding to individual authors from Merck, Sharp & Dohme, and from the NCI Intramural Program.
BIRMINGHAM, ALA. – Serum antibodies induced by a natural human papillomavirus type 16 infection do not appear to protect against acquisition of a subsequent oral HPV 16 infection in men, according to results of the HPV Infection in Men (HIM) study.
That finding is concerning, because about 70% of oropharyngeal cancers in the United States are caused by oral HPV infection. Most are caused by HPV type 16, Christine M. Pierce Campbell, Ph.D. of Moffitt Cancer Center & Research Institute, Tampa and her colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“HPV antibody status may reflect a man’s history of sexual behavior and partnerships, which may not be sufficiently measured in the risk factor questionnaire. In our study, the proportions of men who have sex with men and men who have sex with women and men who had high levels of antibodies at baseline were substantially greater than the proportion of heterosexual men with high antibody levels,” the researchers wrote. They noted that men with an antibody response may have been exposed to more HPV during their lifetimes, and thus were at greater risk of acquiring new oral HPV.
While naturally induced antibodies may not protect against oral HPV 16 infection, prophylactic HPV vaccination may provide such protection, the investigators said.
Of 1,618 men from the oral HPV 16 subcohort of the prospective natural history study of HPV who were followed for a median of 12.7 months after collection of an initial oral specimen, 17 developed a new incident oral HPV16 infection. Of these 17 men, 11 had low anti-HPV16 serum antibody levels, and 6 had high levels,
Anti-HPV 16 serum antibody levels were not associated with cumulative risk of oral HPV16 infection. During the first year of follow-up, 0.4% of those with low levels vs. 0.8% of those with high levels acquired a new infection. Within 24 months of follow-up, 3.0% of those with low levels vs. 3.5% of those with high levels acquired a new infection.
Those with higher levels were 60% more likely than those with low levels to acquire a new infection (hazard ratio, 1.6), but this finding was not statistically significant, the investigators reported.
Study subjects were men from the United States, Brazil, or Mexico who were aged 18 to 73 years and HIV negative at baseline.
Oral rinse-and-gargle specimens were collected from the men at 6-month intervals for up to 4 years. Low antibody levels were defined as the lowest 3 quartiles of antibody levels in the cohort combined, including those with no detectable antibody, and high levels were defined as the highest quartile.
Because of the rarity of oral HPV, additional study is needed to replicate the findings, they said.
Dr. Campbell was supported in part by a postdoctoral fellowship from the American Cancer Society. The HIM study was supported by a grant from the National Cancer Institute (NCI), by grants and/or other funding to individual authors from Merck, Sharp & Dohme, and from the NCI Intramural Program.
BIRMINGHAM, ALA. – Serum antibodies induced by a natural human papillomavirus type 16 infection do not appear to protect against acquisition of a subsequent oral HPV 16 infection in men, according to results of the HPV Infection in Men (HIM) study.
That finding is concerning, because about 70% of oropharyngeal cancers in the United States are caused by oral HPV infection. Most are caused by HPV type 16, Christine M. Pierce Campbell, Ph.D. of Moffitt Cancer Center & Research Institute, Tampa and her colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“HPV antibody status may reflect a man’s history of sexual behavior and partnerships, which may not be sufficiently measured in the risk factor questionnaire. In our study, the proportions of men who have sex with men and men who have sex with women and men who had high levels of antibodies at baseline were substantially greater than the proportion of heterosexual men with high antibody levels,” the researchers wrote. They noted that men with an antibody response may have been exposed to more HPV during their lifetimes, and thus were at greater risk of acquiring new oral HPV.
While naturally induced antibodies may not protect against oral HPV 16 infection, prophylactic HPV vaccination may provide such protection, the investigators said.
Of 1,618 men from the oral HPV 16 subcohort of the prospective natural history study of HPV who were followed for a median of 12.7 months after collection of an initial oral specimen, 17 developed a new incident oral HPV16 infection. Of these 17 men, 11 had low anti-HPV16 serum antibody levels, and 6 had high levels,
Anti-HPV 16 serum antibody levels were not associated with cumulative risk of oral HPV16 infection. During the first year of follow-up, 0.4% of those with low levels vs. 0.8% of those with high levels acquired a new infection. Within 24 months of follow-up, 3.0% of those with low levels vs. 3.5% of those with high levels acquired a new infection.
Those with higher levels were 60% more likely than those with low levels to acquire a new infection (hazard ratio, 1.6), but this finding was not statistically significant, the investigators reported.
Study subjects were men from the United States, Brazil, or Mexico who were aged 18 to 73 years and HIV negative at baseline.
Oral rinse-and-gargle specimens were collected from the men at 6-month intervals for up to 4 years. Low antibody levels were defined as the lowest 3 quartiles of antibody levels in the cohort combined, including those with no detectable antibody, and high levels were defined as the highest quartile.
Because of the rarity of oral HPV, additional study is needed to replicate the findings, they said.
Dr. Campbell was supported in part by a postdoctoral fellowship from the American Cancer Society. The HIM study was supported by a grant from the National Cancer Institute (NCI), by grants and/or other funding to individual authors from Merck, Sharp & Dohme, and from the NCI Intramural Program.
AT THE ASPO ANNUAL MEETING
Key clinical point: Healthy adult men with antibodies induced by a natural HPV16 infection may not be protected against a new oral HPV infection.
Major finding: During the first year of follow-up, 0.4% of men with low anti-HPV16 antibody levels vs. 0.8% of those with high levels after a natural infection acquired a new infection.
Data source: A prospective analysis of data from a subcohort of 1,618 men from the HIM study.
Disclosures: Dr. Campbell was supported in part by a postdoctoral fellowship from the American Cancer Society. The HIM study was supported by a grant from the National Cancer Institute (NCI), by grants and/or other funding to individual authors from Merck, Sharp & Dohme and from the NCI Intramural Program.
Racial disparities in HPV vaccine recommendations persist
BIRMINGHAM, ALA. – Minority adolescent girls are less likely than non-Hispanic white girls to receive a physician recommendation for human papillomavirus vaccination, according to a cross-sectional analysis of 2008-2012 national data from the National Immunization Survey-Teen.
Based on the survey of parents or guardians of 77,320 13-17-year old girls, the weighted proportion of non-Hispanic white girls who received a physician recommendation for HPV vaccination in 2008 was 54%, compared with 43%, 40%, and 47% for non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls, respectively.
The study was reported by Dr. Mahbubur Rahman and colleagues from The University of Texas Medical Branch at Galveston in a poster at the annual meeting of the American Society of Preventive Oncology.
A similar pattern was seen in the remaining years of the study. The weighted proportions for white vs. non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls were 60% vs. 50%, 51%, and 56% in 2009, 59% vs. 46%, 48%, and 56% in 2010, 63% vs. 52%, 51%, and 57% in 2011, and 70% vs. 63%, 56%, and 60% in 2012, the investigators said.
After adjustment for demographic characteristics, a separate weighted analysis for each year of data showed the differences between the non-Hispanic white girls and the other groups were statistically significant for all 5 years. The differences between the non-Hispanic black and Hispanic girls were not statistically significant.
The weighted adjusted odds ratio of receiving a physician recommendation for HPV vaccination among minority girls aged 13-17 varied from 0.81 to 0.87, between 2008 and 2012.
The findings confirm those from a 2009 study showing that white women were more likely than minority women to receive a recommendation for HPV vaccination in their adolescent daughters, and confirms that the findings is persistent, rather than a transient phenomenon.
This is notable, because according to 2013 data, HPV vaccine uptake among girls aged 13 to 17 years is lower than the Healthy People 2020 target of 80% for the 3-dose series among those aged 13 to 15 years. It is also below the level needed for herd immunity (75% to 94%), the investigators said, noting that studies show a strong association between provider recommendation and vaccine uptake.
In fact, provider recommendation is the best predictor of vaccine uptake; adolescent girls who receive a provider recommendation are five times more likely to initiate the HPV vaccine series than are those without a recommendation, they said.
“Reasons for racial disparity in receiving a physician recommendation need to be identified and interventions developed to achieve the targeted HPV vaccine uptake among U.S. adolescent girls irrespective of race/ethnicity,” they concluded.
The authors reported having no disclosures.
BIRMINGHAM, ALA. – Minority adolescent girls are less likely than non-Hispanic white girls to receive a physician recommendation for human papillomavirus vaccination, according to a cross-sectional analysis of 2008-2012 national data from the National Immunization Survey-Teen.
Based on the survey of parents or guardians of 77,320 13-17-year old girls, the weighted proportion of non-Hispanic white girls who received a physician recommendation for HPV vaccination in 2008 was 54%, compared with 43%, 40%, and 47% for non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls, respectively.
The study was reported by Dr. Mahbubur Rahman and colleagues from The University of Texas Medical Branch at Galveston in a poster at the annual meeting of the American Society of Preventive Oncology.
A similar pattern was seen in the remaining years of the study. The weighted proportions for white vs. non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls were 60% vs. 50%, 51%, and 56% in 2009, 59% vs. 46%, 48%, and 56% in 2010, 63% vs. 52%, 51%, and 57% in 2011, and 70% vs. 63%, 56%, and 60% in 2012, the investigators said.
After adjustment for demographic characteristics, a separate weighted analysis for each year of data showed the differences between the non-Hispanic white girls and the other groups were statistically significant for all 5 years. The differences between the non-Hispanic black and Hispanic girls were not statistically significant.
The weighted adjusted odds ratio of receiving a physician recommendation for HPV vaccination among minority girls aged 13-17 varied from 0.81 to 0.87, between 2008 and 2012.
The findings confirm those from a 2009 study showing that white women were more likely than minority women to receive a recommendation for HPV vaccination in their adolescent daughters, and confirms that the findings is persistent, rather than a transient phenomenon.
This is notable, because according to 2013 data, HPV vaccine uptake among girls aged 13 to 17 years is lower than the Healthy People 2020 target of 80% for the 3-dose series among those aged 13 to 15 years. It is also below the level needed for herd immunity (75% to 94%), the investigators said, noting that studies show a strong association between provider recommendation and vaccine uptake.
In fact, provider recommendation is the best predictor of vaccine uptake; adolescent girls who receive a provider recommendation are five times more likely to initiate the HPV vaccine series than are those without a recommendation, they said.
“Reasons for racial disparity in receiving a physician recommendation need to be identified and interventions developed to achieve the targeted HPV vaccine uptake among U.S. adolescent girls irrespective of race/ethnicity,” they concluded.
The authors reported having no disclosures.
BIRMINGHAM, ALA. – Minority adolescent girls are less likely than non-Hispanic white girls to receive a physician recommendation for human papillomavirus vaccination, according to a cross-sectional analysis of 2008-2012 national data from the National Immunization Survey-Teen.
Based on the survey of parents or guardians of 77,320 13-17-year old girls, the weighted proportion of non-Hispanic white girls who received a physician recommendation for HPV vaccination in 2008 was 54%, compared with 43%, 40%, and 47% for non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls, respectively.
The study was reported by Dr. Mahbubur Rahman and colleagues from The University of Texas Medical Branch at Galveston in a poster at the annual meeting of the American Society of Preventive Oncology.
A similar pattern was seen in the remaining years of the study. The weighted proportions for white vs. non-Hispanic black, Hispanic, and non-Hispanic other or multiple race girls were 60% vs. 50%, 51%, and 56% in 2009, 59% vs. 46%, 48%, and 56% in 2010, 63% vs. 52%, 51%, and 57% in 2011, and 70% vs. 63%, 56%, and 60% in 2012, the investigators said.
After adjustment for demographic characteristics, a separate weighted analysis for each year of data showed the differences between the non-Hispanic white girls and the other groups were statistically significant for all 5 years. The differences between the non-Hispanic black and Hispanic girls were not statistically significant.
The weighted adjusted odds ratio of receiving a physician recommendation for HPV vaccination among minority girls aged 13-17 varied from 0.81 to 0.87, between 2008 and 2012.
The findings confirm those from a 2009 study showing that white women were more likely than minority women to receive a recommendation for HPV vaccination in their adolescent daughters, and confirms that the findings is persistent, rather than a transient phenomenon.
This is notable, because according to 2013 data, HPV vaccine uptake among girls aged 13 to 17 years is lower than the Healthy People 2020 target of 80% for the 3-dose series among those aged 13 to 15 years. It is also below the level needed for herd immunity (75% to 94%), the investigators said, noting that studies show a strong association between provider recommendation and vaccine uptake.
In fact, provider recommendation is the best predictor of vaccine uptake; adolescent girls who receive a provider recommendation are five times more likely to initiate the HPV vaccine series than are those without a recommendation, they said.
“Reasons for racial disparity in receiving a physician recommendation need to be identified and interventions developed to achieve the targeted HPV vaccine uptake among U.S. adolescent girls irrespective of race/ethnicity,” they concluded.
The authors reported having no disclosures.
Key clinical point: Racial disparities in HPV vaccine recommendations are contributing to low rates of vaccine uptake.
Major finding: The weighted adjusted odds ratio of receiving a physician recommendation for HPV vaccination among minority (vs. non-Hispanic white) girls varied from 0.81 to 0.87 between 2008 and 2012.
Data source: A cross-sectional study of 77,320 adolescent girls.
Disclosures: The authors reported having no disclosures.
Most accidental genital trauma cases manageable in ED
ORLANDO – Most cases of accidental genital trauma in girls are caused by straddle injury and are isolated to the labia, and most can be managed expectantly or treated in the emergency department, according to findings from a retrospective cohort study.
Penetrating injuries, however, should be considered an indication for management in the operating room, Dr. Tazim Dowlut-McElroy reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
A review of 359 cases showed that the vast majority – 82% – were minor and managed expectantly.
“Only 18% required surgical management. Of those, 37% required general anesthesia in the OR, but 63% were adequately evaluated and treated in the ED,”said Dr. Dowlut-McElroy of Children’s Mercy Hospitals and Clinics, Kansas City.
About 2/3 (64%) of the patients presented during the day, and 36% presented at night. The most common presenting complaint was bleeding and pain (89% of cases), followed by voiding issues in 8% of cases. No presenting complaint was recorded in the remaining patient charts.
The most common mechanism of injury was straddle injury (73% of cases), followed by non-straddle blunt trauma in 15% of cases, and penetrating injury in the remaining cases, Dr. Dowlut-McElroy said.
Injuries included lacerations in 86% of cases, abrasions or contusions in 7%, and hematomas in 3%.
Factors associated with a need for treatment in the OR included greater mean age (mean age was 8 vs. 6 years for OR vs. ED cases), transfer from another institution (77% of OR cases vs. 31% of ED cases), penetrating injury (54% of OR cases vs. 8% of ED cases), injury to the urethra or anus (46% of OR cases vs. 7% of ED cases) and lesion size greater than 3 cm (63% of OR cases vs. 7% of ED cases), she noted.
Logistic regression analysis showed that factors significantly associated with treatment in the OR under general anesthesia were lesion size greater than 3 cm (odds ratio 5.5), and transfer from another hospital (odds ratio 4.1). Presentation at night approached significance (odds ratio 3.2).
Pediatric genital injuries comprise 0.2% to 8% of reported childhood trauma, and despite public health efforts to reduce injuries, the number of such pediatric injuries continues to rise, Dr. Dowlut-McElroy said.
As was the case in the current study, straddle injuries are the most common cause of accidental genital trauma (AGT), and most injuries are external and isolated to the labia.
However, genital bleeding, which is the most common presenting symptom after blunt genital trauma, usually requires a thorough evaluation of the location and severity of injury and may require surgical repair, she said.
Injuries may not consistently result in external bleeding, and without careful examination, injuries to the urogenital tract can be missed and result in delays in repair that can lead to urethral and vaginal stenosis and chronic fissures and fistulas, she said.
“Therefore, a key question is whether all patients should undergo examination under anesthesia in the operating room rather than undergoing evaluation and treatment in the emergency department,” she said, citing reports of discordant examination for the severity and extent of injury after AGT between an initial assessment in the ED and treatment in the OR, which have led some authors to recommend that all patients with AGT be evaluated under general anesthesia.
In one retrospective review of 22 patients with AGT, the most common mechanism of injury was straddle injury, and every patient was initially evaluated in the ED by an emergency physician and then by a pediatric surgeon. All were taken to the OR, and the findings on examination under anesthesia in the OR demonstrated significant disagreement with the findings in the ED, Dr. Dowlut-McElroy said.
In fact, 76% of patients had more significant injury than determined by the ED evaluation; 27% had complete disruption of the perianal sphincter, and 95% required surgical repair (Ped. Emerg. Care. 1995; 11:372-5).
In contrast to those findings, a 2008 review showed that only 19% of 105 cases - also due mainly to straddle injury - required examination under anesthesia in the OR, while 7% underwent conscious sedation in the ED. Only 21% of subjects required surgical repair (Ped. Emerg. Care 2008;24:831-5).
Conflicting reports such as these have results in considerable variation in recommendations for managing AGT, Dr. Dowlut-McElroy said.
“We sought to determine the characteristics of those managed in ED vs. those requiring treatment in the OR,” she added.
Patients included in the current review were identified by a medical database query from January 2000 to July 2014. They were aged 0-18 years and had been treated in the ED for genital trauma; those with obstetrical injuries were excluded.
Though limited by the retrospective and single center design, as well as by errors associated with misclassification of variables and missing data, the finding suggest that with adequate sedation, most girls with AGT, with the exception of those who experience penetrating injury, can undergo a thorough examination and repair in the ED, she concluded.
Dr. Dowlut-McElroy reported having no disclosures. One of her co-authors, Dr. Julie Strickland, is a Nexplanon trainer for Merck.
ORLANDO – Most cases of accidental genital trauma in girls are caused by straddle injury and are isolated to the labia, and most can be managed expectantly or treated in the emergency department, according to findings from a retrospective cohort study.
Penetrating injuries, however, should be considered an indication for management in the operating room, Dr. Tazim Dowlut-McElroy reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
A review of 359 cases showed that the vast majority – 82% – were minor and managed expectantly.
“Only 18% required surgical management. Of those, 37% required general anesthesia in the OR, but 63% were adequately evaluated and treated in the ED,”said Dr. Dowlut-McElroy of Children’s Mercy Hospitals and Clinics, Kansas City.
About 2/3 (64%) of the patients presented during the day, and 36% presented at night. The most common presenting complaint was bleeding and pain (89% of cases), followed by voiding issues in 8% of cases. No presenting complaint was recorded in the remaining patient charts.
The most common mechanism of injury was straddle injury (73% of cases), followed by non-straddle blunt trauma in 15% of cases, and penetrating injury in the remaining cases, Dr. Dowlut-McElroy said.
Injuries included lacerations in 86% of cases, abrasions or contusions in 7%, and hematomas in 3%.
Factors associated with a need for treatment in the OR included greater mean age (mean age was 8 vs. 6 years for OR vs. ED cases), transfer from another institution (77% of OR cases vs. 31% of ED cases), penetrating injury (54% of OR cases vs. 8% of ED cases), injury to the urethra or anus (46% of OR cases vs. 7% of ED cases) and lesion size greater than 3 cm (63% of OR cases vs. 7% of ED cases), she noted.
Logistic regression analysis showed that factors significantly associated with treatment in the OR under general anesthesia were lesion size greater than 3 cm (odds ratio 5.5), and transfer from another hospital (odds ratio 4.1). Presentation at night approached significance (odds ratio 3.2).
Pediatric genital injuries comprise 0.2% to 8% of reported childhood trauma, and despite public health efforts to reduce injuries, the number of such pediatric injuries continues to rise, Dr. Dowlut-McElroy said.
As was the case in the current study, straddle injuries are the most common cause of accidental genital trauma (AGT), and most injuries are external and isolated to the labia.
However, genital bleeding, which is the most common presenting symptom after blunt genital trauma, usually requires a thorough evaluation of the location and severity of injury and may require surgical repair, she said.
Injuries may not consistently result in external bleeding, and without careful examination, injuries to the urogenital tract can be missed and result in delays in repair that can lead to urethral and vaginal stenosis and chronic fissures and fistulas, she said.
“Therefore, a key question is whether all patients should undergo examination under anesthesia in the operating room rather than undergoing evaluation and treatment in the emergency department,” she said, citing reports of discordant examination for the severity and extent of injury after AGT between an initial assessment in the ED and treatment in the OR, which have led some authors to recommend that all patients with AGT be evaluated under general anesthesia.
In one retrospective review of 22 patients with AGT, the most common mechanism of injury was straddle injury, and every patient was initially evaluated in the ED by an emergency physician and then by a pediatric surgeon. All were taken to the OR, and the findings on examination under anesthesia in the OR demonstrated significant disagreement with the findings in the ED, Dr. Dowlut-McElroy said.
In fact, 76% of patients had more significant injury than determined by the ED evaluation; 27% had complete disruption of the perianal sphincter, and 95% required surgical repair (Ped. Emerg. Care. 1995; 11:372-5).
In contrast to those findings, a 2008 review showed that only 19% of 105 cases - also due mainly to straddle injury - required examination under anesthesia in the OR, while 7% underwent conscious sedation in the ED. Only 21% of subjects required surgical repair (Ped. Emerg. Care 2008;24:831-5).
Conflicting reports such as these have results in considerable variation in recommendations for managing AGT, Dr. Dowlut-McElroy said.
“We sought to determine the characteristics of those managed in ED vs. those requiring treatment in the OR,” she added.
Patients included in the current review were identified by a medical database query from January 2000 to July 2014. They were aged 0-18 years and had been treated in the ED for genital trauma; those with obstetrical injuries were excluded.
Though limited by the retrospective and single center design, as well as by errors associated with misclassification of variables and missing data, the finding suggest that with adequate sedation, most girls with AGT, with the exception of those who experience penetrating injury, can undergo a thorough examination and repair in the ED, she concluded.
Dr. Dowlut-McElroy reported having no disclosures. One of her co-authors, Dr. Julie Strickland, is a Nexplanon trainer for Merck.
ORLANDO – Most cases of accidental genital trauma in girls are caused by straddle injury and are isolated to the labia, and most can be managed expectantly or treated in the emergency department, according to findings from a retrospective cohort study.
Penetrating injuries, however, should be considered an indication for management in the operating room, Dr. Tazim Dowlut-McElroy reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
A review of 359 cases showed that the vast majority – 82% – were minor and managed expectantly.
“Only 18% required surgical management. Of those, 37% required general anesthesia in the OR, but 63% were adequately evaluated and treated in the ED,”said Dr. Dowlut-McElroy of Children’s Mercy Hospitals and Clinics, Kansas City.
About 2/3 (64%) of the patients presented during the day, and 36% presented at night. The most common presenting complaint was bleeding and pain (89% of cases), followed by voiding issues in 8% of cases. No presenting complaint was recorded in the remaining patient charts.
The most common mechanism of injury was straddle injury (73% of cases), followed by non-straddle blunt trauma in 15% of cases, and penetrating injury in the remaining cases, Dr. Dowlut-McElroy said.
Injuries included lacerations in 86% of cases, abrasions or contusions in 7%, and hematomas in 3%.
Factors associated with a need for treatment in the OR included greater mean age (mean age was 8 vs. 6 years for OR vs. ED cases), transfer from another institution (77% of OR cases vs. 31% of ED cases), penetrating injury (54% of OR cases vs. 8% of ED cases), injury to the urethra or anus (46% of OR cases vs. 7% of ED cases) and lesion size greater than 3 cm (63% of OR cases vs. 7% of ED cases), she noted.
Logistic regression analysis showed that factors significantly associated with treatment in the OR under general anesthesia were lesion size greater than 3 cm (odds ratio 5.5), and transfer from another hospital (odds ratio 4.1). Presentation at night approached significance (odds ratio 3.2).
Pediatric genital injuries comprise 0.2% to 8% of reported childhood trauma, and despite public health efforts to reduce injuries, the number of such pediatric injuries continues to rise, Dr. Dowlut-McElroy said.
As was the case in the current study, straddle injuries are the most common cause of accidental genital trauma (AGT), and most injuries are external and isolated to the labia.
However, genital bleeding, which is the most common presenting symptom after blunt genital trauma, usually requires a thorough evaluation of the location and severity of injury and may require surgical repair, she said.
Injuries may not consistently result in external bleeding, and without careful examination, injuries to the urogenital tract can be missed and result in delays in repair that can lead to urethral and vaginal stenosis and chronic fissures and fistulas, she said.
“Therefore, a key question is whether all patients should undergo examination under anesthesia in the operating room rather than undergoing evaluation and treatment in the emergency department,” she said, citing reports of discordant examination for the severity and extent of injury after AGT between an initial assessment in the ED and treatment in the OR, which have led some authors to recommend that all patients with AGT be evaluated under general anesthesia.
In one retrospective review of 22 patients with AGT, the most common mechanism of injury was straddle injury, and every patient was initially evaluated in the ED by an emergency physician and then by a pediatric surgeon. All were taken to the OR, and the findings on examination under anesthesia in the OR demonstrated significant disagreement with the findings in the ED, Dr. Dowlut-McElroy said.
In fact, 76% of patients had more significant injury than determined by the ED evaluation; 27% had complete disruption of the perianal sphincter, and 95% required surgical repair (Ped. Emerg. Care. 1995; 11:372-5).
In contrast to those findings, a 2008 review showed that only 19% of 105 cases - also due mainly to straddle injury - required examination under anesthesia in the OR, while 7% underwent conscious sedation in the ED. Only 21% of subjects required surgical repair (Ped. Emerg. Care 2008;24:831-5).
Conflicting reports such as these have results in considerable variation in recommendations for managing AGT, Dr. Dowlut-McElroy said.
“We sought to determine the characteristics of those managed in ED vs. those requiring treatment in the OR,” she added.
Patients included in the current review were identified by a medical database query from January 2000 to July 2014. They were aged 0-18 years and had been treated in the ED for genital trauma; those with obstetrical injuries were excluded.
Though limited by the retrospective and single center design, as well as by errors associated with misclassification of variables and missing data, the finding suggest that with adequate sedation, most girls with AGT, with the exception of those who experience penetrating injury, can undergo a thorough examination and repair in the ED, she concluded.
Dr. Dowlut-McElroy reported having no disclosures. One of her co-authors, Dr. Julie Strickland, is a Nexplanon trainer for Merck.
AT THE NASPAG ANNUAL MEETING
Key clinical point: With adequate sedation, most girls with AGT requiring treatment can undergo evaluation and repair in the ED.
Major finding: 82% of cases reviewed were minor and managed expectantly.
Data source: A retrospective medical records review of 359 cases of AGT.
Disclosures: Dr. Dowlut-McElroy reported having no disclosures. One of her co-authors, Dr. Julie Strickland, is a Nexplanon trainer for Merck.
Preliminary analysis confirms birth defect-cancer association
BIRMINGHAM, ALA. – Children with birth defects have been shown to have an increased risk of developing cancer, and preliminary findings from a large record-linkage study not only confirm and better define the risk, but also demonstrate the value of such studies for identifying new risk factors for childhood cancer, according to Philip J. Lupo, Ph.D.
Because birth defects and childhood cancers are relatively rare, large-scale linkages of state- and federally-funded disease registries may be helpful to identify new childhood cancer predisposition syndromes, Dr. Lupo of Baylor College of Medicine, Houston, and his colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“Epidemiological investigations have indicated that cancer risk is greater among those with major malformations such as spina bifida, as well as among those with relatively minor malformations, such as rib anomalies,” he said, noting that well-established birth defect-childhood cancer associations include Down syndrome and leukemia and Beckwith-Wiedemann syndrome and Wilms tumor.
Recent record-linkage studies have pointed to a number of previously unidentified associations, but these patterns require confirmation in larger studies; much more information is needed with respect to less common birth defects and childhood cancers. Additionally, understanding why some of these conditions overlap is critical for prevention efforts, he said.
Thus, he and his colleagues are planning to assemble a large population-based birth cohort to identify novel cancer predisposition syndromes, and enroll families where children have both a birth defect and cancer to better understand the molecular underpinnings of these conditions.
The project, titled the Genetic overlap Between Anomalies and Cancer in Kids (GOBACK) Study, involves registry linkages in several states, which together represent more than 10 million births.
In a preliminary assessment using data from the Michigan Department of Community Health for more than 2.5 million births, the investigators found increased risks of cancer among children with several specific birth defects. For instance, children with cardiac defects were twice as likely to develop cancer as were those without cardiac defects. Additionally, for those born with any birth defect, the risk of specific cancers was also elevated. For example, those with any birth defect were twice as likely as those without birth defects to develop rhabdomyosarcoma.
The findings are consistent with previous studies, and thus reinforce the utility of record linkages between population-based registries for epidemiologic assessments of these conditions, Dr. Lupo said, noting that identifying specific associations will allow for molecular studies to determine whether common developmental pathways underlie the etiology of birth defects and childhood cancer.
Dr. Lupo reported having no disclosures.
BIRMINGHAM, ALA. – Children with birth defects have been shown to have an increased risk of developing cancer, and preliminary findings from a large record-linkage study not only confirm and better define the risk, but also demonstrate the value of such studies for identifying new risk factors for childhood cancer, according to Philip J. Lupo, Ph.D.
Because birth defects and childhood cancers are relatively rare, large-scale linkages of state- and federally-funded disease registries may be helpful to identify new childhood cancer predisposition syndromes, Dr. Lupo of Baylor College of Medicine, Houston, and his colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“Epidemiological investigations have indicated that cancer risk is greater among those with major malformations such as spina bifida, as well as among those with relatively minor malformations, such as rib anomalies,” he said, noting that well-established birth defect-childhood cancer associations include Down syndrome and leukemia and Beckwith-Wiedemann syndrome and Wilms tumor.
Recent record-linkage studies have pointed to a number of previously unidentified associations, but these patterns require confirmation in larger studies; much more information is needed with respect to less common birth defects and childhood cancers. Additionally, understanding why some of these conditions overlap is critical for prevention efforts, he said.
Thus, he and his colleagues are planning to assemble a large population-based birth cohort to identify novel cancer predisposition syndromes, and enroll families where children have both a birth defect and cancer to better understand the molecular underpinnings of these conditions.
The project, titled the Genetic overlap Between Anomalies and Cancer in Kids (GOBACK) Study, involves registry linkages in several states, which together represent more than 10 million births.
In a preliminary assessment using data from the Michigan Department of Community Health for more than 2.5 million births, the investigators found increased risks of cancer among children with several specific birth defects. For instance, children with cardiac defects were twice as likely to develop cancer as were those without cardiac defects. Additionally, for those born with any birth defect, the risk of specific cancers was also elevated. For example, those with any birth defect were twice as likely as those without birth defects to develop rhabdomyosarcoma.
The findings are consistent with previous studies, and thus reinforce the utility of record linkages between population-based registries for epidemiologic assessments of these conditions, Dr. Lupo said, noting that identifying specific associations will allow for molecular studies to determine whether common developmental pathways underlie the etiology of birth defects and childhood cancer.
Dr. Lupo reported having no disclosures.
BIRMINGHAM, ALA. – Children with birth defects have been shown to have an increased risk of developing cancer, and preliminary findings from a large record-linkage study not only confirm and better define the risk, but also demonstrate the value of such studies for identifying new risk factors for childhood cancer, according to Philip J. Lupo, Ph.D.
Because birth defects and childhood cancers are relatively rare, large-scale linkages of state- and federally-funded disease registries may be helpful to identify new childhood cancer predisposition syndromes, Dr. Lupo of Baylor College of Medicine, Houston, and his colleagues reported in a poster at the annual meeting of the American Society of Preventive Oncology.
“Epidemiological investigations have indicated that cancer risk is greater among those with major malformations such as spina bifida, as well as among those with relatively minor malformations, such as rib anomalies,” he said, noting that well-established birth defect-childhood cancer associations include Down syndrome and leukemia and Beckwith-Wiedemann syndrome and Wilms tumor.
Recent record-linkage studies have pointed to a number of previously unidentified associations, but these patterns require confirmation in larger studies; much more information is needed with respect to less common birth defects and childhood cancers. Additionally, understanding why some of these conditions overlap is critical for prevention efforts, he said.
Thus, he and his colleagues are planning to assemble a large population-based birth cohort to identify novel cancer predisposition syndromes, and enroll families where children have both a birth defect and cancer to better understand the molecular underpinnings of these conditions.
The project, titled the Genetic overlap Between Anomalies and Cancer in Kids (GOBACK) Study, involves registry linkages in several states, which together represent more than 10 million births.
In a preliminary assessment using data from the Michigan Department of Community Health for more than 2.5 million births, the investigators found increased risks of cancer among children with several specific birth defects. For instance, children with cardiac defects were twice as likely to develop cancer as were those without cardiac defects. Additionally, for those born with any birth defect, the risk of specific cancers was also elevated. For example, those with any birth defect were twice as likely as those without birth defects to develop rhabdomyosarcoma.
The findings are consistent with previous studies, and thus reinforce the utility of record linkages between population-based registries for epidemiologic assessments of these conditions, Dr. Lupo said, noting that identifying specific associations will allow for molecular studies to determine whether common developmental pathways underlie the etiology of birth defects and childhood cancer.
Dr. Lupo reported having no disclosures.
EXPERT ANALYSIS FROM THE ASPO ANNUAL MEETING
Lower DMPA dose provides comparable efficacy, tolerability in adolescents
ORLANDO – Depot medroxyprogesterone acetate (DMPA) given at a dose of 75 mg provided effective contraception with rates of weight gain and unscheduled bleeding in adolescent girls that were comparable to higher doses in a small randomized controlled trial.
Of 27 girls who completed the study, 8 were treated with a 150 mg dose, 9 were treated with 104 mg, and 10 were treated with 75 mg. Weight gain was greatest with the 75 mg dose, with 28.6% of girls in that group experiencing greater than 5% weight gain, compared with 12.5% and 22.2% of those in the 150 mg and 104 mg groups, respectively. The differences between groups did not reach statistical significance, however, Dr. Andrea E. Bonny reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
The combined number of days of bleeding or spotting during days 0 to 24 was lowest for those on the 75 mg dose (about 44 days vs. about 45 and 70 days for the 150 mg and 104 mg groups, respectively). These differences also failed to reach statistical significance, although the difference between the 104 mg and 75 mg groups trended toward significance, said Dr. Bonny of Nationwide Children’s Hospital, Columbus, Ohio.
Study subjects were healthy postmenarcheal females aged 12 to 21 years (mean of 18.5 years) who were initiating DMPA. Assigned doses were administered by intramuscular injection every 12 weeks. Subjects were assessed for weight/adiposity at baseline and at 12 and 24 weeks, and for serum MPA, estradiol, and progesterone weekly from 0-12 weeks, and also at 16, 20, and 24 weeks. Doses were escalated if MPA concentration was below 0.3 ng/ml, which is triple the previously demonstrated efficacy threshold of 0.1 ng/ml, Dr. Bonny said.
Doses were also escalated if progesterone concentration was 2 ng/ml or greater (which is suggestive of ovulation) at the time of an assessment. Escalations were required in 3 of the 10 girls in the 75 mg dose group, indicating 70% efficacy, but it is important to note that 98% of the measures of MPA concentration were above the standard 0.01 threshold for efficacy, she said.
All subjects kept a diary to track the number of days of spotting and bleeding.
An analysis of pharmacokinetic measures, including maximum MPA concentration, time needed to reach maximum MPA concentration, MPA elimination rate, and area under the concentration time curve from 0-12 weeks, showed a trend for an inverse relationship between MPA exposure and weight gain. The analysis also showed that time to maximum MPA concentration was a better predictor of unscheduled bleeding than was MPA dose; the time to maximum concentration was highly inversely correlated with the number of days of bleeding, Dr. Bonny said.
“The longer the time to get up to max concentration, the less bleeding they had,” she said.
Single intramuscular doses of 25, 50, and 100 mg have been shown to inhibit ovulation for at least 3 months in most individuals, but the relationship between exposure and side effects has been unclear, she said.
“We know that weight gain and unscheduled bleeding are highly variable among adolescents on DMPA, and despite a wide variety of studies examining predictors of DMPA-associated side effects, understanding of who is at risk is still limited,” she said, adding that pharmacokinetics also vary widely among patients.
“We found the 75 mg DMPA dose provided effective contraception in 70% of subjects, but that was using our very conservative cutoff of 0.03 ng/ml, and if we actually used the historical cutoff of 0.1, it would have reached 98%,” she said
The finding of a trend toward increased weight gain with the lowest dose seems counterintuitive, but has also been seen in at least one other cohort, she noted.
Aside from demonstrating the efficacy of the 75 mg dose of DMPA, this study demonstrated that adolescents “really can be enrolled in rigorous contraceptive trials,” she said, explaining that such studies rarely include individuals under age 18 years, and almost never include those under age 16 years.
“We had subjects who were 12 years old ... who were very compliant,” she said.
She also noted that the findings would be much more clinically useful if a simple clinical marker of pharmacokinetics were available.
“To that end, we have collected whole blood, and we will be analyzing it for genetic variants of the primary enzyme responsible for metabolizing DMPA to see if we can find a marker that can predict [time to maximum concentration] or some other pharmacokinetic measure,” she said.
This study was funded by a Society of Family Planning research grant and a Clinical and Translational Science Awards grant. Dr. Bonny reported having no disclosures.
ORLANDO – Depot medroxyprogesterone acetate (DMPA) given at a dose of 75 mg provided effective contraception with rates of weight gain and unscheduled bleeding in adolescent girls that were comparable to higher doses in a small randomized controlled trial.
Of 27 girls who completed the study, 8 were treated with a 150 mg dose, 9 were treated with 104 mg, and 10 were treated with 75 mg. Weight gain was greatest with the 75 mg dose, with 28.6% of girls in that group experiencing greater than 5% weight gain, compared with 12.5% and 22.2% of those in the 150 mg and 104 mg groups, respectively. The differences between groups did not reach statistical significance, however, Dr. Andrea E. Bonny reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
The combined number of days of bleeding or spotting during days 0 to 24 was lowest for those on the 75 mg dose (about 44 days vs. about 45 and 70 days for the 150 mg and 104 mg groups, respectively). These differences also failed to reach statistical significance, although the difference between the 104 mg and 75 mg groups trended toward significance, said Dr. Bonny of Nationwide Children’s Hospital, Columbus, Ohio.
Study subjects were healthy postmenarcheal females aged 12 to 21 years (mean of 18.5 years) who were initiating DMPA. Assigned doses were administered by intramuscular injection every 12 weeks. Subjects were assessed for weight/adiposity at baseline and at 12 and 24 weeks, and for serum MPA, estradiol, and progesterone weekly from 0-12 weeks, and also at 16, 20, and 24 weeks. Doses were escalated if MPA concentration was below 0.3 ng/ml, which is triple the previously demonstrated efficacy threshold of 0.1 ng/ml, Dr. Bonny said.
Doses were also escalated if progesterone concentration was 2 ng/ml or greater (which is suggestive of ovulation) at the time of an assessment. Escalations were required in 3 of the 10 girls in the 75 mg dose group, indicating 70% efficacy, but it is important to note that 98% of the measures of MPA concentration were above the standard 0.01 threshold for efficacy, she said.
All subjects kept a diary to track the number of days of spotting and bleeding.
An analysis of pharmacokinetic measures, including maximum MPA concentration, time needed to reach maximum MPA concentration, MPA elimination rate, and area under the concentration time curve from 0-12 weeks, showed a trend for an inverse relationship between MPA exposure and weight gain. The analysis also showed that time to maximum MPA concentration was a better predictor of unscheduled bleeding than was MPA dose; the time to maximum concentration was highly inversely correlated with the number of days of bleeding, Dr. Bonny said.
“The longer the time to get up to max concentration, the less bleeding they had,” she said.
Single intramuscular doses of 25, 50, and 100 mg have been shown to inhibit ovulation for at least 3 months in most individuals, but the relationship between exposure and side effects has been unclear, she said.
“We know that weight gain and unscheduled bleeding are highly variable among adolescents on DMPA, and despite a wide variety of studies examining predictors of DMPA-associated side effects, understanding of who is at risk is still limited,” she said, adding that pharmacokinetics also vary widely among patients.
“We found the 75 mg DMPA dose provided effective contraception in 70% of subjects, but that was using our very conservative cutoff of 0.03 ng/ml, and if we actually used the historical cutoff of 0.1, it would have reached 98%,” she said
The finding of a trend toward increased weight gain with the lowest dose seems counterintuitive, but has also been seen in at least one other cohort, she noted.
Aside from demonstrating the efficacy of the 75 mg dose of DMPA, this study demonstrated that adolescents “really can be enrolled in rigorous contraceptive trials,” she said, explaining that such studies rarely include individuals under age 18 years, and almost never include those under age 16 years.
“We had subjects who were 12 years old ... who were very compliant,” she said.
She also noted that the findings would be much more clinically useful if a simple clinical marker of pharmacokinetics were available.
“To that end, we have collected whole blood, and we will be analyzing it for genetic variants of the primary enzyme responsible for metabolizing DMPA to see if we can find a marker that can predict [time to maximum concentration] or some other pharmacokinetic measure,” she said.
This study was funded by a Society of Family Planning research grant and a Clinical and Translational Science Awards grant. Dr. Bonny reported having no disclosures.
ORLANDO – Depot medroxyprogesterone acetate (DMPA) given at a dose of 75 mg provided effective contraception with rates of weight gain and unscheduled bleeding in adolescent girls that were comparable to higher doses in a small randomized controlled trial.
Of 27 girls who completed the study, 8 were treated with a 150 mg dose, 9 were treated with 104 mg, and 10 were treated with 75 mg. Weight gain was greatest with the 75 mg dose, with 28.6% of girls in that group experiencing greater than 5% weight gain, compared with 12.5% and 22.2% of those in the 150 mg and 104 mg groups, respectively. The differences between groups did not reach statistical significance, however, Dr. Andrea E. Bonny reported at the annual meeting of the North American Society for Pediatric and Adolescent Gynecology.
The combined number of days of bleeding or spotting during days 0 to 24 was lowest for those on the 75 mg dose (about 44 days vs. about 45 and 70 days for the 150 mg and 104 mg groups, respectively). These differences also failed to reach statistical significance, although the difference between the 104 mg and 75 mg groups trended toward significance, said Dr. Bonny of Nationwide Children’s Hospital, Columbus, Ohio.
Study subjects were healthy postmenarcheal females aged 12 to 21 years (mean of 18.5 years) who were initiating DMPA. Assigned doses were administered by intramuscular injection every 12 weeks. Subjects were assessed for weight/adiposity at baseline and at 12 and 24 weeks, and for serum MPA, estradiol, and progesterone weekly from 0-12 weeks, and also at 16, 20, and 24 weeks. Doses were escalated if MPA concentration was below 0.3 ng/ml, which is triple the previously demonstrated efficacy threshold of 0.1 ng/ml, Dr. Bonny said.
Doses were also escalated if progesterone concentration was 2 ng/ml or greater (which is suggestive of ovulation) at the time of an assessment. Escalations were required in 3 of the 10 girls in the 75 mg dose group, indicating 70% efficacy, but it is important to note that 98% of the measures of MPA concentration were above the standard 0.01 threshold for efficacy, she said.
All subjects kept a diary to track the number of days of spotting and bleeding.
An analysis of pharmacokinetic measures, including maximum MPA concentration, time needed to reach maximum MPA concentration, MPA elimination rate, and area under the concentration time curve from 0-12 weeks, showed a trend for an inverse relationship between MPA exposure and weight gain. The analysis also showed that time to maximum MPA concentration was a better predictor of unscheduled bleeding than was MPA dose; the time to maximum concentration was highly inversely correlated with the number of days of bleeding, Dr. Bonny said.
“The longer the time to get up to max concentration, the less bleeding they had,” she said.
Single intramuscular doses of 25, 50, and 100 mg have been shown to inhibit ovulation for at least 3 months in most individuals, but the relationship between exposure and side effects has been unclear, she said.
“We know that weight gain and unscheduled bleeding are highly variable among adolescents on DMPA, and despite a wide variety of studies examining predictors of DMPA-associated side effects, understanding of who is at risk is still limited,” she said, adding that pharmacokinetics also vary widely among patients.
“We found the 75 mg DMPA dose provided effective contraception in 70% of subjects, but that was using our very conservative cutoff of 0.03 ng/ml, and if we actually used the historical cutoff of 0.1, it would have reached 98%,” she said
The finding of a trend toward increased weight gain with the lowest dose seems counterintuitive, but has also been seen in at least one other cohort, she noted.
Aside from demonstrating the efficacy of the 75 mg dose of DMPA, this study demonstrated that adolescents “really can be enrolled in rigorous contraceptive trials,” she said, explaining that such studies rarely include individuals under age 18 years, and almost never include those under age 16 years.
“We had subjects who were 12 years old ... who were very compliant,” she said.
She also noted that the findings would be much more clinically useful if a simple clinical marker of pharmacokinetics were available.
“To that end, we have collected whole blood, and we will be analyzing it for genetic variants of the primary enzyme responsible for metabolizing DMPA to see if we can find a marker that can predict [time to maximum concentration] or some other pharmacokinetic measure,” she said.
This study was funded by a Society of Family Planning research grant and a Clinical and Translational Science Awards grant. Dr. Bonny reported having no disclosures.
AT THE NASPAG ANNUAL MEETING
Key clinical point: A 75 mg DMPA dose provides effective contraception with comparable bleeding and weight gain.
Major finding: Using a very conservative cutoff, a 75 mg DMPA dose provided effective contraception in 70% of subjects.
Data source: A randomized controlled trial involving 27 girls.
Disclosures: This study was funded by a Society of Family Planning research grant and a Clinical and Translational Science Awards grant. Dr. Bonny reported having no disclosures.
Combine topicals, orals for onychomycosis
MIAMI BEACH – Two new topical solutions approved in 2014 for the treatment of distal subungual onychomycosis don’t eliminate the need for oral treatment, but they do represent improvement in the options available to patients, according to Dr. Boni E. Elewski.
Oral treatments, including terbinafine, itraconazole, and fluconazole are still of value – either alone or in combination with the new solutions or other agents – for this type of onychomycosis, which is “essentially a nail bed dermatophytosis,” she said at the South Beach Symposium.
Terbinafine has been used for 2 decades, and is probably the most commonly used treatment for onychomycosis. It is approved as a once-daily pill given for 90 days, and reportedly has a cure rate just under 40%, she said.
Itraconazole is approved as a 200-mg daily dose for 12 weeks, although most physicians use pulse dosing, prescribing 400 mg daily for a week, then for 1 week each month.
“I like this drug for my nondermatophyte patients … if you have something that you don’t think is a dermatophyte, or if they’ve failed terbinafine, this is an excellent option,” she said.
Fluconazole, her “personal favorite,” is used off label for onychomycosis, but has been shown to have good cure rates with once-weekly treatment, said Dr. Elewski of the University of Alabama at Birmingham.
“I’ll confess, I like this drug because I feel comfortable with it,” she said adding that she tells her patients to think about “Fungal Fridays” or “Toesdays” as a way to remember to take their weekly treatments.
The cure rates are quite good, she said, noting that because the nails grow so slowly, the pace of the treatment matches patient expectations. They don’t finish treatment and still have a “cruddy-looking” nail, she explained.
If any of these oral drugs are used, laboratory monitoring and periodic assessments are necessary, so treatment is a bit complicated. Adverse events remain a concern – particularly drug-drug interactions, drug eruption, cardiac issues (with itraconazole), and loss of taste (with terbinafine).
“So we do have to worry about some of these conditions, which is why having other treatments is so nice,” Dr. Elewski said.
Another reason it’s good to have more options is that no matter which drug you choose, it won’t cure everyone, she noted. Sometimes that’s because the condition is severe; patients with a dermatophyte abscess, those with very thick nails, and those with complete nail involvement associated with a nondermatophyte mold, for example, will have a poorer prognosis, regardless of which oral treatment given. Also, the condition is often complicated by concomitant disorders such as psoriasis. About 5% of patients with psoriasis have nail-only disease, and about a third of them also have onychomycosis. Others are misdiagnosed as having onychomycosis.
Alternatives that can be used alone or in combination with the oral therapies, include the two new topical solutions: efinaconazole and tavaborole, Dr. Elewski said.
Solutions are good, because you can apply them on, under, and around the nail. Both of the drugs have demonstrated effective penetration of the nail plate, allowing penetration to the nail bed where the infection exists, she noted.
The mycological cure rate with efinaconazole yields outcomes comparable to those with oral drugs.
“I think [the mycological cure] is actually the most important endpoint. Because when you want to get rid of the fungus, what do you do? You want to kill the fungus,” said Dr. Elewski, adding that mycological cure is the first sign a patient will go on to experience complete cure.
The complete cure rate is lower, but that appears to be a time-related factor. The nail takes a long time to grow, so the complete cure rate will lag behind the mycological cure rate, Dr. Elewski explained.
The other topical solution – tavaborole – is a new molecule that contains boron. Mycological cure rates in studies of the drug were in the mid-30% range, and it appears able to be used with nail polish without issues to the polish or the outcomes, she said.
In Dr. Elewski’s experience, these topicals work better than expected in clinical practice, based on the clinical trials. One patient who wasn’t eligible for the clinical trials because of a dermatophytosis, for example, was nearly clear within 5 months, and is now totally cured, she said.
“So these treatments are effective as monotherapy, but could be used as an adjunct with systemic therapy, and perhaps also in nondermatophyte cases of onychomycosis,” Dr. Elewski noted. “I think it would be a better option to pick one of these topical drugs than putting someone on a prolonged course of itraconazole if at all possible.” The safety issue would be more favorable with the topical antifungal solution, she said.
A treatment that should never be used is ketoconazole, she noted, explaining that although the drug was never approved specifically for onychomycosis, it was often prescribed for the treatment of tinea versicolor. Because of safety concerns, the FDA removed its indication for all cutaneous fungal infections in 2013.
Dr. Elewski is a consultant for Valeant Pharmaceuticals and a contracted researcher for Anacor Pharmaceuticals.
MIAMI BEACH – Two new topical solutions approved in 2014 for the treatment of distal subungual onychomycosis don’t eliminate the need for oral treatment, but they do represent improvement in the options available to patients, according to Dr. Boni E. Elewski.
Oral treatments, including terbinafine, itraconazole, and fluconazole are still of value – either alone or in combination with the new solutions or other agents – for this type of onychomycosis, which is “essentially a nail bed dermatophytosis,” she said at the South Beach Symposium.
Terbinafine has been used for 2 decades, and is probably the most commonly used treatment for onychomycosis. It is approved as a once-daily pill given for 90 days, and reportedly has a cure rate just under 40%, she said.
Itraconazole is approved as a 200-mg daily dose for 12 weeks, although most physicians use pulse dosing, prescribing 400 mg daily for a week, then for 1 week each month.
“I like this drug for my nondermatophyte patients … if you have something that you don’t think is a dermatophyte, or if they’ve failed terbinafine, this is an excellent option,” she said.
Fluconazole, her “personal favorite,” is used off label for onychomycosis, but has been shown to have good cure rates with once-weekly treatment, said Dr. Elewski of the University of Alabama at Birmingham.
“I’ll confess, I like this drug because I feel comfortable with it,” she said adding that she tells her patients to think about “Fungal Fridays” or “Toesdays” as a way to remember to take their weekly treatments.
The cure rates are quite good, she said, noting that because the nails grow so slowly, the pace of the treatment matches patient expectations. They don’t finish treatment and still have a “cruddy-looking” nail, she explained.
If any of these oral drugs are used, laboratory monitoring and periodic assessments are necessary, so treatment is a bit complicated. Adverse events remain a concern – particularly drug-drug interactions, drug eruption, cardiac issues (with itraconazole), and loss of taste (with terbinafine).
“So we do have to worry about some of these conditions, which is why having other treatments is so nice,” Dr. Elewski said.
Another reason it’s good to have more options is that no matter which drug you choose, it won’t cure everyone, she noted. Sometimes that’s because the condition is severe; patients with a dermatophyte abscess, those with very thick nails, and those with complete nail involvement associated with a nondermatophyte mold, for example, will have a poorer prognosis, regardless of which oral treatment given. Also, the condition is often complicated by concomitant disorders such as psoriasis. About 5% of patients with psoriasis have nail-only disease, and about a third of them also have onychomycosis. Others are misdiagnosed as having onychomycosis.
Alternatives that can be used alone or in combination with the oral therapies, include the two new topical solutions: efinaconazole and tavaborole, Dr. Elewski said.
Solutions are good, because you can apply them on, under, and around the nail. Both of the drugs have demonstrated effective penetration of the nail plate, allowing penetration to the nail bed where the infection exists, she noted.
The mycological cure rate with efinaconazole yields outcomes comparable to those with oral drugs.
“I think [the mycological cure] is actually the most important endpoint. Because when you want to get rid of the fungus, what do you do? You want to kill the fungus,” said Dr. Elewski, adding that mycological cure is the first sign a patient will go on to experience complete cure.
The complete cure rate is lower, but that appears to be a time-related factor. The nail takes a long time to grow, so the complete cure rate will lag behind the mycological cure rate, Dr. Elewski explained.
The other topical solution – tavaborole – is a new molecule that contains boron. Mycological cure rates in studies of the drug were in the mid-30% range, and it appears able to be used with nail polish without issues to the polish or the outcomes, she said.
In Dr. Elewski’s experience, these topicals work better than expected in clinical practice, based on the clinical trials. One patient who wasn’t eligible for the clinical trials because of a dermatophytosis, for example, was nearly clear within 5 months, and is now totally cured, she said.
“So these treatments are effective as monotherapy, but could be used as an adjunct with systemic therapy, and perhaps also in nondermatophyte cases of onychomycosis,” Dr. Elewski noted. “I think it would be a better option to pick one of these topical drugs than putting someone on a prolonged course of itraconazole if at all possible.” The safety issue would be more favorable with the topical antifungal solution, she said.
A treatment that should never be used is ketoconazole, she noted, explaining that although the drug was never approved specifically for onychomycosis, it was often prescribed for the treatment of tinea versicolor. Because of safety concerns, the FDA removed its indication for all cutaneous fungal infections in 2013.
Dr. Elewski is a consultant for Valeant Pharmaceuticals and a contracted researcher for Anacor Pharmaceuticals.
MIAMI BEACH – Two new topical solutions approved in 2014 for the treatment of distal subungual onychomycosis don’t eliminate the need for oral treatment, but they do represent improvement in the options available to patients, according to Dr. Boni E. Elewski.
Oral treatments, including terbinafine, itraconazole, and fluconazole are still of value – either alone or in combination with the new solutions or other agents – for this type of onychomycosis, which is “essentially a nail bed dermatophytosis,” she said at the South Beach Symposium.
Terbinafine has been used for 2 decades, and is probably the most commonly used treatment for onychomycosis. It is approved as a once-daily pill given for 90 days, and reportedly has a cure rate just under 40%, she said.
Itraconazole is approved as a 200-mg daily dose for 12 weeks, although most physicians use pulse dosing, prescribing 400 mg daily for a week, then for 1 week each month.
“I like this drug for my nondermatophyte patients … if you have something that you don’t think is a dermatophyte, or if they’ve failed terbinafine, this is an excellent option,” she said.
Fluconazole, her “personal favorite,” is used off label for onychomycosis, but has been shown to have good cure rates with once-weekly treatment, said Dr. Elewski of the University of Alabama at Birmingham.
“I’ll confess, I like this drug because I feel comfortable with it,” she said adding that she tells her patients to think about “Fungal Fridays” or “Toesdays” as a way to remember to take their weekly treatments.
The cure rates are quite good, she said, noting that because the nails grow so slowly, the pace of the treatment matches patient expectations. They don’t finish treatment and still have a “cruddy-looking” nail, she explained.
If any of these oral drugs are used, laboratory monitoring and periodic assessments are necessary, so treatment is a bit complicated. Adverse events remain a concern – particularly drug-drug interactions, drug eruption, cardiac issues (with itraconazole), and loss of taste (with terbinafine).
“So we do have to worry about some of these conditions, which is why having other treatments is so nice,” Dr. Elewski said.
Another reason it’s good to have more options is that no matter which drug you choose, it won’t cure everyone, she noted. Sometimes that’s because the condition is severe; patients with a dermatophyte abscess, those with very thick nails, and those with complete nail involvement associated with a nondermatophyte mold, for example, will have a poorer prognosis, regardless of which oral treatment given. Also, the condition is often complicated by concomitant disorders such as psoriasis. About 5% of patients with psoriasis have nail-only disease, and about a third of them also have onychomycosis. Others are misdiagnosed as having onychomycosis.
Alternatives that can be used alone or in combination with the oral therapies, include the two new topical solutions: efinaconazole and tavaborole, Dr. Elewski said.
Solutions are good, because you can apply them on, under, and around the nail. Both of the drugs have demonstrated effective penetration of the nail plate, allowing penetration to the nail bed where the infection exists, she noted.
The mycological cure rate with efinaconazole yields outcomes comparable to those with oral drugs.
“I think [the mycological cure] is actually the most important endpoint. Because when you want to get rid of the fungus, what do you do? You want to kill the fungus,” said Dr. Elewski, adding that mycological cure is the first sign a patient will go on to experience complete cure.
The complete cure rate is lower, but that appears to be a time-related factor. The nail takes a long time to grow, so the complete cure rate will lag behind the mycological cure rate, Dr. Elewski explained.
The other topical solution – tavaborole – is a new molecule that contains boron. Mycological cure rates in studies of the drug were in the mid-30% range, and it appears able to be used with nail polish without issues to the polish or the outcomes, she said.
In Dr. Elewski’s experience, these topicals work better than expected in clinical practice, based on the clinical trials. One patient who wasn’t eligible for the clinical trials because of a dermatophytosis, for example, was nearly clear within 5 months, and is now totally cured, she said.
“So these treatments are effective as monotherapy, but could be used as an adjunct with systemic therapy, and perhaps also in nondermatophyte cases of onychomycosis,” Dr. Elewski noted. “I think it would be a better option to pick one of these topical drugs than putting someone on a prolonged course of itraconazole if at all possible.” The safety issue would be more favorable with the topical antifungal solution, she said.
A treatment that should never be used is ketoconazole, she noted, explaining that although the drug was never approved specifically for onychomycosis, it was often prescribed for the treatment of tinea versicolor. Because of safety concerns, the FDA removed its indication for all cutaneous fungal infections in 2013.
Dr. Elewski is a consultant for Valeant Pharmaceuticals and a contracted researcher for Anacor Pharmaceuticals.
AT THE SOUTH BEACH SYMPOSIUM