User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
Statins cut limb events in PAD patients
AMSTERDAM – Treatment with a statin cut the relative rate of worsening peripheral artery disease by roughly 20% in a registry with nearly 6,000 peripheral artery disease patients.
But the registry data also showed that more than a third of patients with peripheral artery disease (PAD) failed to receive statin treatment, Dr. Dharam Kumbhani said at the annual Congress of the European Society of Cardiology.
"This is one of the first, and largest, studies to demonstrate an impact [of statin treatment] on adverse limb outcomes in patients with PAD, including worsening claudication, new critical limb ischemia, need for revascularization, and notably need for ischemic amputations. But despite having a class I recommendation for use in patients with PAD, data from this large, international registry suggest that statin use remains suboptimal," said Dr. Kumbhani, an interventional cardiologist at the University of Texas Southwestern Medical Center in Dallas.
"Future research should focus on improving patient and physician compliance with statin use across the entire spectrum of PAD patients," he said.
Concomitant coronary artery disease, as well as the specialty of the treating physician, seemed to link with statin use in these patients, Dr. Kumbhani added. "In PAD patients with CAD, statin use occurred in about 75%; in PAD patients without CAD, it was less than 50%."
Statins were most reliably prescribed to PAD patients by cardiologists, who administered the drugs to about 80% of all their PAD patients, regardless of concomitant CAD. In contrast, vascular surgeons prescribed statins to fewer than half their PAD patients, and in those who did not have concomitant CAD, statin use fell to less than a third. About 70% of all patients enrolled in the registry received their treatment from a primary care physician.
The data came from the Reduction of Atherothrombosis for Continued Health (REACH) registry, which followed more than 45,000 enrolled patients for at least 4 years at more than 3,600 centers in 29 countries; more than a quarter were U.S. patients (JAMA 2010;304:1350-7). Enrolled patients were at least 45 years old and had at least three atherosclerosis risk factors or established vascular disease in the coronary, cerebral, or peripheral domains. The enrolled patients included 5,861 with established, symptomatic PAD, of whom 3,643 (62%) were on statin treatment at the time they entered the registry.
During 4-year follow-up the rate of the primary systemic outcome – a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke – decreased by a statistically significant, relative 15% among patients on a statin compared with those not on a statin at baseline, in a propensity-score adjusted analysis.
Statin use linked with a statistically significant, 21% relative drop in worsening PAD events, compared with nonuse, in the propensity-score adjusted model. Adjusted analyses also showed statistically significant reductions for most of the individual outcomes that formed the composites, including a 27% relative reduction in nonfatal strokes and a 43% relative drop in limb amputations.
The systemic benefits seen in the registry from statin use in patients with PAD are consistent with results from randomized, controlled trials, but finding statin use also linked with decreased adverse limb outcomes is new, Dr. Kumbhani said.
The REACH registry is funded by Sanofi and Bristol-Myers Squibb. Dr. Kumbhani reported having no relevant financial disclosures.
On Twitter @mitchelzoler
AMSTERDAM – Treatment with a statin cut the relative rate of worsening peripheral artery disease by roughly 20% in a registry with nearly 6,000 peripheral artery disease patients.
But the registry data also showed that more than a third of patients with peripheral artery disease (PAD) failed to receive statin treatment, Dr. Dharam Kumbhani said at the annual Congress of the European Society of Cardiology.
"This is one of the first, and largest, studies to demonstrate an impact [of statin treatment] on adverse limb outcomes in patients with PAD, including worsening claudication, new critical limb ischemia, need for revascularization, and notably need for ischemic amputations. But despite having a class I recommendation for use in patients with PAD, data from this large, international registry suggest that statin use remains suboptimal," said Dr. Kumbhani, an interventional cardiologist at the University of Texas Southwestern Medical Center in Dallas.
"Future research should focus on improving patient and physician compliance with statin use across the entire spectrum of PAD patients," he said.
Concomitant coronary artery disease, as well as the specialty of the treating physician, seemed to link with statin use in these patients, Dr. Kumbhani added. "In PAD patients with CAD, statin use occurred in about 75%; in PAD patients without CAD, it was less than 50%."
Statins were most reliably prescribed to PAD patients by cardiologists, who administered the drugs to about 80% of all their PAD patients, regardless of concomitant CAD. In contrast, vascular surgeons prescribed statins to fewer than half their PAD patients, and in those who did not have concomitant CAD, statin use fell to less than a third. About 70% of all patients enrolled in the registry received their treatment from a primary care physician.
The data came from the Reduction of Atherothrombosis for Continued Health (REACH) registry, which followed more than 45,000 enrolled patients for at least 4 years at more than 3,600 centers in 29 countries; more than a quarter were U.S. patients (JAMA 2010;304:1350-7). Enrolled patients were at least 45 years old and had at least three atherosclerosis risk factors or established vascular disease in the coronary, cerebral, or peripheral domains. The enrolled patients included 5,861 with established, symptomatic PAD, of whom 3,643 (62%) were on statin treatment at the time they entered the registry.
During 4-year follow-up the rate of the primary systemic outcome – a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke – decreased by a statistically significant, relative 15% among patients on a statin compared with those not on a statin at baseline, in a propensity-score adjusted analysis.
Statin use linked with a statistically significant, 21% relative drop in worsening PAD events, compared with nonuse, in the propensity-score adjusted model. Adjusted analyses also showed statistically significant reductions for most of the individual outcomes that formed the composites, including a 27% relative reduction in nonfatal strokes and a 43% relative drop in limb amputations.
The systemic benefits seen in the registry from statin use in patients with PAD are consistent with results from randomized, controlled trials, but finding statin use also linked with decreased adverse limb outcomes is new, Dr. Kumbhani said.
The REACH registry is funded by Sanofi and Bristol-Myers Squibb. Dr. Kumbhani reported having no relevant financial disclosures.
On Twitter @mitchelzoler
AMSTERDAM – Treatment with a statin cut the relative rate of worsening peripheral artery disease by roughly 20% in a registry with nearly 6,000 peripheral artery disease patients.
But the registry data also showed that more than a third of patients with peripheral artery disease (PAD) failed to receive statin treatment, Dr. Dharam Kumbhani said at the annual Congress of the European Society of Cardiology.
"This is one of the first, and largest, studies to demonstrate an impact [of statin treatment] on adverse limb outcomes in patients with PAD, including worsening claudication, new critical limb ischemia, need for revascularization, and notably need for ischemic amputations. But despite having a class I recommendation for use in patients with PAD, data from this large, international registry suggest that statin use remains suboptimal," said Dr. Kumbhani, an interventional cardiologist at the University of Texas Southwestern Medical Center in Dallas.
"Future research should focus on improving patient and physician compliance with statin use across the entire spectrum of PAD patients," he said.
Concomitant coronary artery disease, as well as the specialty of the treating physician, seemed to link with statin use in these patients, Dr. Kumbhani added. "In PAD patients with CAD, statin use occurred in about 75%; in PAD patients without CAD, it was less than 50%."
Statins were most reliably prescribed to PAD patients by cardiologists, who administered the drugs to about 80% of all their PAD patients, regardless of concomitant CAD. In contrast, vascular surgeons prescribed statins to fewer than half their PAD patients, and in those who did not have concomitant CAD, statin use fell to less than a third. About 70% of all patients enrolled in the registry received their treatment from a primary care physician.
The data came from the Reduction of Atherothrombosis for Continued Health (REACH) registry, which followed more than 45,000 enrolled patients for at least 4 years at more than 3,600 centers in 29 countries; more than a quarter were U.S. patients (JAMA 2010;304:1350-7). Enrolled patients were at least 45 years old and had at least three atherosclerosis risk factors or established vascular disease in the coronary, cerebral, or peripheral domains. The enrolled patients included 5,861 with established, symptomatic PAD, of whom 3,643 (62%) were on statin treatment at the time they entered the registry.
During 4-year follow-up the rate of the primary systemic outcome – a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke – decreased by a statistically significant, relative 15% among patients on a statin compared with those not on a statin at baseline, in a propensity-score adjusted analysis.
Statin use linked with a statistically significant, 21% relative drop in worsening PAD events, compared with nonuse, in the propensity-score adjusted model. Adjusted analyses also showed statistically significant reductions for most of the individual outcomes that formed the composites, including a 27% relative reduction in nonfatal strokes and a 43% relative drop in limb amputations.
The systemic benefits seen in the registry from statin use in patients with PAD are consistent with results from randomized, controlled trials, but finding statin use also linked with decreased adverse limb outcomes is new, Dr. Kumbhani said.
The REACH registry is funded by Sanofi and Bristol-Myers Squibb. Dr. Kumbhani reported having no relevant financial disclosures.
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2013
Major finding: In patients with peripheral artery disease, statin use was linked with an adjusted, relative 21% cut in adverse limb outcomes.
Data source: Four-year follow-up in the REACH registry, which enrolled more than 45,000 patients with atherothrombotic disease in 29 countries.
Disclosures: The REACH registry is funded by Sanofi and Bristol-Myers Squibb. Dr. Kumbhani reported having no relevant financial conflicts.
Registry-based randomized clinical trials are here
The results from the TASTE trial reported last week at the annual congress of the European Society of Cardiology and in the New England Journal of Medicine – which showed no 30-day survival benefit for ST-segment elevation myocardial infarction patients treated with thrombus aspiration compared with conventional stenting – received more kudos for the study’s design than for its primary finding.
A big reason was that the study design was so unprecedented. Called a "registry-based randomized clinical trial" (RRCT), the Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia (TASTE) trial enrolled STEMI patients as they entered the long-standing, pan–Sweden and Iceland Swedish Web System for Enhancement and Development of Evidence-Based Care in Heart Disease Evaluated According to Recommended Therapies (SWEDEHEART) registry.
On the SWEDEHEART registry backbone, which automatically provided comprehensive data collection and follow-up, the researchers who collaborated on TASTE built in a web-based randomization that allocated more than 7,200 patients to either treatment by thrombus aspiration followed by percutaneous coronary intervention (PCI) or to PCI only.
The main result was clear, but several experts who heard it hedged on the immediate impact of the TASTE results on use of thrombus aspiration.
This treatment "is still logical, feasible, and relatively easy, and will continue to be attractive to many," especially until the results of a more conventionally designed, similarly-sized trial, TOTAL, are available, commented Dr. Raffaele De Caterina, the designated discussant for the report at the meeting.
Despite their skepticism on what the TASTE findings mean for current practice, Dr. De Caterina and others had unbridled enthusiasm for the new RRCT. The design "allowed completeness of follow-up and a much lower cost," and with no commercial involvement, he said.
"This is an exciting and innovative approach. Automatic collection of outcomes makes it incredibly cost effective, and a number of countries have large registry programs," said Dr. Keith A.A. Fox, a professor of cardiology at the University of Edinburgh.
A commentary that ran with the published report on TASTE called the RRCT concept "a disruptive technology" that "transforms existing standards, procedures, and cost structures." The commentary, cowritten by Dr. Michael S. Lauer, director of cardiovascular sciences at the National Heart, Lung, and Blood Institute, noted that the incremental cost to run TASTE was about $300,000, roughly the size of an average research grant from the National Institutes of Health to an academic laboratory.
The RRCT model inaugurated by TASTE produced enough interest to have the TASTE investigators give a second talk at the ESC meeting focused just on their study’s design and its implications. Dr. Stefan James, a TASTE researcher, highlighted several other clinical questions that he and his colleagues are addressing in ongoing RRCTS: the role of supplemental oxygen in acute MI patients, a comparison of bivalirudin (Angiomax) and heparin in acute MI patients on contemporary antiplatelet treatment, the value of drug-eluting technologies for treating peripheral artery disease, and the ability of bioabsorbable vascular scaffolds to prevent events.
The RRCT is "ideal for one clinically important hypothesis with reliable, hard endpoints," Dr. James said.
On Twitter @mitchelzoler
The results from the TASTE trial reported last week at the annual congress of the European Society of Cardiology and in the New England Journal of Medicine – which showed no 30-day survival benefit for ST-segment elevation myocardial infarction patients treated with thrombus aspiration compared with conventional stenting – received more kudos for the study’s design than for its primary finding.
A big reason was that the study design was so unprecedented. Called a "registry-based randomized clinical trial" (RRCT), the Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia (TASTE) trial enrolled STEMI patients as they entered the long-standing, pan–Sweden and Iceland Swedish Web System for Enhancement and Development of Evidence-Based Care in Heart Disease Evaluated According to Recommended Therapies (SWEDEHEART) registry.
On the SWEDEHEART registry backbone, which automatically provided comprehensive data collection and follow-up, the researchers who collaborated on TASTE built in a web-based randomization that allocated more than 7,200 patients to either treatment by thrombus aspiration followed by percutaneous coronary intervention (PCI) or to PCI only.
The main result was clear, but several experts who heard it hedged on the immediate impact of the TASTE results on use of thrombus aspiration.
This treatment "is still logical, feasible, and relatively easy, and will continue to be attractive to many," especially until the results of a more conventionally designed, similarly-sized trial, TOTAL, are available, commented Dr. Raffaele De Caterina, the designated discussant for the report at the meeting.
Despite their skepticism on what the TASTE findings mean for current practice, Dr. De Caterina and others had unbridled enthusiasm for the new RRCT. The design "allowed completeness of follow-up and a much lower cost," and with no commercial involvement, he said.
"This is an exciting and innovative approach. Automatic collection of outcomes makes it incredibly cost effective, and a number of countries have large registry programs," said Dr. Keith A.A. Fox, a professor of cardiology at the University of Edinburgh.
A commentary that ran with the published report on TASTE called the RRCT concept "a disruptive technology" that "transforms existing standards, procedures, and cost structures." The commentary, cowritten by Dr. Michael S. Lauer, director of cardiovascular sciences at the National Heart, Lung, and Blood Institute, noted that the incremental cost to run TASTE was about $300,000, roughly the size of an average research grant from the National Institutes of Health to an academic laboratory.
The RRCT model inaugurated by TASTE produced enough interest to have the TASTE investigators give a second talk at the ESC meeting focused just on their study’s design and its implications. Dr. Stefan James, a TASTE researcher, highlighted several other clinical questions that he and his colleagues are addressing in ongoing RRCTS: the role of supplemental oxygen in acute MI patients, a comparison of bivalirudin (Angiomax) and heparin in acute MI patients on contemporary antiplatelet treatment, the value of drug-eluting technologies for treating peripheral artery disease, and the ability of bioabsorbable vascular scaffolds to prevent events.
The RRCT is "ideal for one clinically important hypothesis with reliable, hard endpoints," Dr. James said.
On Twitter @mitchelzoler
The results from the TASTE trial reported last week at the annual congress of the European Society of Cardiology and in the New England Journal of Medicine – which showed no 30-day survival benefit for ST-segment elevation myocardial infarction patients treated with thrombus aspiration compared with conventional stenting – received more kudos for the study’s design than for its primary finding.
A big reason was that the study design was so unprecedented. Called a "registry-based randomized clinical trial" (RRCT), the Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia (TASTE) trial enrolled STEMI patients as they entered the long-standing, pan–Sweden and Iceland Swedish Web System for Enhancement and Development of Evidence-Based Care in Heart Disease Evaluated According to Recommended Therapies (SWEDEHEART) registry.
On the SWEDEHEART registry backbone, which automatically provided comprehensive data collection and follow-up, the researchers who collaborated on TASTE built in a web-based randomization that allocated more than 7,200 patients to either treatment by thrombus aspiration followed by percutaneous coronary intervention (PCI) or to PCI only.
The main result was clear, but several experts who heard it hedged on the immediate impact of the TASTE results on use of thrombus aspiration.
This treatment "is still logical, feasible, and relatively easy, and will continue to be attractive to many," especially until the results of a more conventionally designed, similarly-sized trial, TOTAL, are available, commented Dr. Raffaele De Caterina, the designated discussant for the report at the meeting.
Despite their skepticism on what the TASTE findings mean for current practice, Dr. De Caterina and others had unbridled enthusiasm for the new RRCT. The design "allowed completeness of follow-up and a much lower cost," and with no commercial involvement, he said.
"This is an exciting and innovative approach. Automatic collection of outcomes makes it incredibly cost effective, and a number of countries have large registry programs," said Dr. Keith A.A. Fox, a professor of cardiology at the University of Edinburgh.
A commentary that ran with the published report on TASTE called the RRCT concept "a disruptive technology" that "transforms existing standards, procedures, and cost structures." The commentary, cowritten by Dr. Michael S. Lauer, director of cardiovascular sciences at the National Heart, Lung, and Blood Institute, noted that the incremental cost to run TASTE was about $300,000, roughly the size of an average research grant from the National Institutes of Health to an academic laboratory.
The RRCT model inaugurated by TASTE produced enough interest to have the TASTE investigators give a second talk at the ESC meeting focused just on their study’s design and its implications. Dr. Stefan James, a TASTE researcher, highlighted several other clinical questions that he and his colleagues are addressing in ongoing RRCTS: the role of supplemental oxygen in acute MI patients, a comparison of bivalirudin (Angiomax) and heparin in acute MI patients on contemporary antiplatelet treatment, the value of drug-eluting technologies for treating peripheral artery disease, and the ability of bioabsorbable vascular scaffolds to prevent events.
The RRCT is "ideal for one clinically important hypothesis with reliable, hard endpoints," Dr. James said.
On Twitter @mitchelzoler
Correlates of excellent type 1 diabetes control found
PHILADELPHIA – U.S. patients with type 1 diabetes who maintain excellent glycemic control have the best diabetes-control practices, use a pump, and have higher incomes and educational attainment, compared with patients who maintain fair or poor glycemic control, based on data collected from more than 2,000 registry patients.
Patients with excellent control had, by definition, the lowest levels of hemoglobin A1c, and also had a quarter of the episodes of diabetic ketoacidosis as did the patients who maintained fair or poor glycemic control, Elaine Massaro, R.N., said at the annual meeting of the American Association of Diabetes Educators.
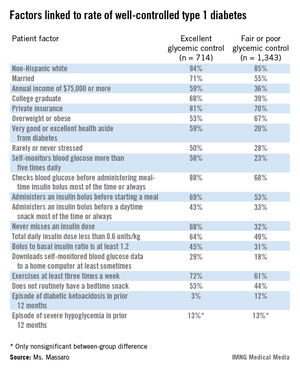
But better glycemic control did not result in an increased incidence of severe hypoglycemic episodes; the rate of severe hypoglycemia was 13% during the preceding 12 months among patients with excellent glycemic control and those with fair or poor control, said Ms. Massaro, an advanced practice nurse at Northwestern University in Chicago.
Between September 2010 and May 2013, more than 26,000 U.S. adult and pediatric patients with type 1 diabetes treated at any of 70 participating centers enrolled in the T1D Exchange Clinic Registry. From the total group of enrollees, Ms. Massaro and her associates identified 714 patients (3%) who, at their entry examination, were at least 26 years old, had been diagnosed with type 1 diabetes for at least 2 years, were not using a real-time continuous glucose monitor, and had excellent glycemic control, with a HbA1c of less than 6.5%. The researchers also identified a second subgroup of 1,343 (5%) from the registered patients who matched the first group by age, duration of diagnosis, and lack of continuous monitoring, but differed by having fair or poor glycemic control, with a HbA1c of 8.5% or higher.
Comparison of these two subgroups identified several factors that significantly correlated with excellent glycemic control. For example, 68% of patients with excellent control never missed an insulin dose, compared with 32% of those with fair or poor control. The prevalence of patients who said they self-monitored their blood glucose more than five times a day was 58% among those with excellent control and 23% among those with fair or poor control.
Four times as many patients with fair or poor control reported having an episode of diabetic ketoacidosis during the prior 12 months, compared with patients who maintained excellent glycemic control (12% vs. 3%).
The analysis failed to find any correlation between the level of glycemic control and the type of insulin that patients used, or whether they administered a long-acting insulin formulation once or twice a day. The results also showed that insulin pump use was significantly more prevalent among patients with excellent glycemic control than among those with fair or poor control, but Ms. Massaro did not report specific rates of pump use in the two subgroups.
Ms. Massaro had no disclosures.
On Twitter @mitchelzoler
PHILADELPHIA – U.S. patients with type 1 diabetes who maintain excellent glycemic control have the best diabetes-control practices, use a pump, and have higher incomes and educational attainment, compared with patients who maintain fair or poor glycemic control, based on data collected from more than 2,000 registry patients.
Patients with excellent control had, by definition, the lowest levels of hemoglobin A1c, and also had a quarter of the episodes of diabetic ketoacidosis as did the patients who maintained fair or poor glycemic control, Elaine Massaro, R.N., said at the annual meeting of the American Association of Diabetes Educators.

But better glycemic control did not result in an increased incidence of severe hypoglycemic episodes; the rate of severe hypoglycemia was 13% during the preceding 12 months among patients with excellent glycemic control and those with fair or poor control, said Ms. Massaro, an advanced practice nurse at Northwestern University in Chicago.
Between September 2010 and May 2013, more than 26,000 U.S. adult and pediatric patients with type 1 diabetes treated at any of 70 participating centers enrolled in the T1D Exchange Clinic Registry. From the total group of enrollees, Ms. Massaro and her associates identified 714 patients (3%) who, at their entry examination, were at least 26 years old, had been diagnosed with type 1 diabetes for at least 2 years, were not using a real-time continuous glucose monitor, and had excellent glycemic control, with a HbA1c of less than 6.5%. The researchers also identified a second subgroup of 1,343 (5%) from the registered patients who matched the first group by age, duration of diagnosis, and lack of continuous monitoring, but differed by having fair or poor glycemic control, with a HbA1c of 8.5% or higher.
Comparison of these two subgroups identified several factors that significantly correlated with excellent glycemic control. For example, 68% of patients with excellent control never missed an insulin dose, compared with 32% of those with fair or poor control. The prevalence of patients who said they self-monitored their blood glucose more than five times a day was 58% among those with excellent control and 23% among those with fair or poor control.
Four times as many patients with fair or poor control reported having an episode of diabetic ketoacidosis during the prior 12 months, compared with patients who maintained excellent glycemic control (12% vs. 3%).
The analysis failed to find any correlation between the level of glycemic control and the type of insulin that patients used, or whether they administered a long-acting insulin formulation once or twice a day. The results also showed that insulin pump use was significantly more prevalent among patients with excellent glycemic control than among those with fair or poor control, but Ms. Massaro did not report specific rates of pump use in the two subgroups.
Ms. Massaro had no disclosures.
On Twitter @mitchelzoler
PHILADELPHIA – U.S. patients with type 1 diabetes who maintain excellent glycemic control have the best diabetes-control practices, use a pump, and have higher incomes and educational attainment, compared with patients who maintain fair or poor glycemic control, based on data collected from more than 2,000 registry patients.
Patients with excellent control had, by definition, the lowest levels of hemoglobin A1c, and also had a quarter of the episodes of diabetic ketoacidosis as did the patients who maintained fair or poor glycemic control, Elaine Massaro, R.N., said at the annual meeting of the American Association of Diabetes Educators.

But better glycemic control did not result in an increased incidence of severe hypoglycemic episodes; the rate of severe hypoglycemia was 13% during the preceding 12 months among patients with excellent glycemic control and those with fair or poor control, said Ms. Massaro, an advanced practice nurse at Northwestern University in Chicago.
Between September 2010 and May 2013, more than 26,000 U.S. adult and pediatric patients with type 1 diabetes treated at any of 70 participating centers enrolled in the T1D Exchange Clinic Registry. From the total group of enrollees, Ms. Massaro and her associates identified 714 patients (3%) who, at their entry examination, were at least 26 years old, had been diagnosed with type 1 diabetes for at least 2 years, were not using a real-time continuous glucose monitor, and had excellent glycemic control, with a HbA1c of less than 6.5%. The researchers also identified a second subgroup of 1,343 (5%) from the registered patients who matched the first group by age, duration of diagnosis, and lack of continuous monitoring, but differed by having fair or poor glycemic control, with a HbA1c of 8.5% or higher.
Comparison of these two subgroups identified several factors that significantly correlated with excellent glycemic control. For example, 68% of patients with excellent control never missed an insulin dose, compared with 32% of those with fair or poor control. The prevalence of patients who said they self-monitored their blood glucose more than five times a day was 58% among those with excellent control and 23% among those with fair or poor control.
Four times as many patients with fair or poor control reported having an episode of diabetic ketoacidosis during the prior 12 months, compared with patients who maintained excellent glycemic control (12% vs. 3%).
The analysis failed to find any correlation between the level of glycemic control and the type of insulin that patients used, or whether they administered a long-acting insulin formulation once or twice a day. The results also showed that insulin pump use was significantly more prevalent among patients with excellent glycemic control than among those with fair or poor control, but Ms. Massaro did not report specific rates of pump use in the two subgroups.
Ms. Massaro had no disclosures.
On Twitter @mitchelzoler
AT AADE 13
Major finding: Of patients with excellent type 1 diabetes control, 68% never miss an insulin dose, compared with 32% of patients with fair or poor control.
Data source: Data came from 2,057 U.S. patients with type 1 diabetes and excellent or fair-to-poor glycemic control enrolled in a nationwide patient registry.
Disclosures: Ms. Massaro had no disclosures.
CRT appears deadly in short-QRS patients
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
The results from EchoCRT fit well with the results of a meta-analysis that my associates and I recently ran on data from 3,782 heart failure patients treated with CRT in five trials sponsored by Medtronic. The results showed that QRS duration was a powerful predictor of the effects of CRT on morbidity and mortality.
The meta-analysis results confirmed the benefit of CRT in patients with mild, moderate, or severe heart failure symptoms, in sinus rhythm, and with a QRS duration of at least 140 msec, and in these patients CRT is standard of care. The results also showed that the benefits from CRT diminish as the QRS duration goes below 140 msec. Patients with a QRS duration of 130-139 msec are in a gray zone. If they need a defibrillator, then making it a CRT device makes sense, but if no device implant is planned then continued attempts at medical treatment are probably better than going to CRT.

|
|
I would avoid CRT in patients with a QRS of less than 130 msec, and now the EchoCRT results show evidence of harm in these patients. A lot of patients with a QRS duration of 120-129 msec have been receiving CRT devices when the chances of benefit are small. This might be a treatment of last resort, but only when you have exhausted all the other treatment alternatives.
The meta-analysis also showed that QRS duration was the only independent predictor of CRT outcomes (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht290]). QRS morphology – whether or not there is a left bundle branch block – was not a significant factor relative to QRS duration. The EchoCRT results also showed that echocardiography used to diagnose left-ventricular dyssynchrony failed to identify a subgroup of patients with a narrow QRS duration who benefited from CRT.
Dr. John G.F. Cleland is professor of medicine at the University of Hull (England). He has been a consultant to Biotronik, St. Jude, and Medtronic. He made these comments in an interview.
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
AMSTERDAM – Patients with severe heart failure and a narrow QRS duration who received cardiac resynchronization therapy had more than twice the rate of cardiovascular deaths as patients who did not undergo active CRT treatment in a multinational randomized study with 809 patients.
The study’s enrollment criteria focused on patients with a QRS duration of less than 130 msec, and in this group active CRT produced no benefit, compared with the CRT function turned off in control patients for the study’s primary efficacy endpoint of death from any cause or first hospitalization for worsening heart failure.
In addition, during an average 19 months’ follow-up, patients in the CRT-on group had an all-cause death rate nearly five percentage points above the controls, an 80% relatively increased hazard, and a cardiovascular death rate also five percentage points above the controls, a greater than two-fold relative hazard, Dr. Johannes Holzmeister said at the annual congress of the European Society of Cardiology.
Both increased hazard rates were statistically significant and were a "surprising, unexpected finding," said Dr. Holzmeister, a cardiologist at the University of Zurich.
"This is the final nail in the coffin for CRT in patients with only slightly-prolonged QRS," commented Dr. Douglas P. Zipes, a professor and electrophysiologist at Indiana University in Indianapolis.
Existing recommendations from the American College of Cardiology and American Heart Association (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jacc2013.05.019]) restrict unequivocal endorsement of CRT to heart failure patients with a QRS duration of at least 150 msec and say that CRT can be useful for patients with New York Heart Association class II, III, or IV heart failure, left bundle branch block (LBBB), and a QRS duration of 120-149 msec. Recommendations from the European Society of Cardiology are more permissive, calling CRT recommended for patients in sinus rhythm with a QRS duration of at least 120 msec, LBBB morphology on their QRS wave, and an ejection fraction of at least 35% who are expected to remain in good functional status for more than 1 year (Eur. Heart J. 2012;33:1787-847).
The ESC recommendations also say that CRT should be considered in patients with a QRS duration of at least 150 msec, irrespective of their QRS morphology.
It seems like many patients with shorter QRS durations have been receiving CRT treatment. During his report at the meeting, Dr. Holzmeister cited data from a 2008-2009 European-wide survey of CRT patients by the European Heart Rhythm Association, which showed that, 4-5 years ago, 9% of patients in Europe who received a CRT device had a QRS duration of less than 120 msec and another 10% of the CRT recipients had durations of 120-129 msec (Eur. Heart J. 2009;30:2450-60).
Substantial numbers of patients continue to receive CRT treatment with a QRS duration of 120-129 msec, a treatment that the data indicate should be a "last resort," said Dr. John G.F. Cleland, professor of medicine and a heart failure specialist at the University of Hull (England).
Although existing society recommendations sanction CRT for selected heart failure patients with a QRS duration of 120-149 msec, many experts have become convinced that the field needs a new set of QRS criteria.
"I think the cutoff for CRT implantation should be 150 msec," said Dr. Gordon F. Tomaselli, professor of cardiology and an electrophysiologist at Johns Hopkins University in Baltimore. "Patients with a QRS duration of less than 150 msec, particularly without left bundle branch block, should probably not get CRTs unless some special circumstances are present," he said in an interview.
"This reinforces the conclusion that we should not use CRT devices in heart failure patients with a QRS duration of less than 130 msec," commented Dr. Stefan D. Anker, professor and cardiologist at Charitè Hospital in Berlin and designated discussant for Dr. Holzmeister’s talk at the meeting. Dr. Anker called CRT use in patients with QRS durations of 130-149 msec uncertain and in need of more evidence for efficacy and safety from trials. "What is certain is that in patients with a QRS of 150 msec or greater there is a strong mortality reduction with CRT," he said.
The Echocardiography Guided Cardiac Resynchronization Therapy (EchoCRT) study randomized 809 patients with a QRS duration of less than 130 msec and left ventricular ejection fraction of 35% or less, and also required that patients meet criteria for having left-ventricular dyssynchrony detected by echocardiography. All patients received a CRT device, which was turned on in 404 patients. The study began in August 2008, and enrollment stopped on March 13, 2013, because of futility and a potential for harm.
At that time, cardiovascular deaths had occurred in 9% of patients with their CRT turned on and in 4% of those with the device turned off. Concurrent with Dr. Holzmeister’s report at the meeting, an article with the results was published online (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1306687]).
The EchoCRT trial was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister said that he has been a consultant to Biotronik. Dr. Zipes said that he has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker said that he has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures.
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2013
Major finding: Cardiovascular death occurred in 9% of patients on active cardiac resynchronization therapy and in 4% of control patients.
Data source: Data came from the EchoCRT trial, a multicenter, randomized trial with 809 patients with NYHA class III or IV heart failure and a QRS duration of less than 130 msec.
Disclosures: EchoCRT was sponsored by Biotronik and GE Healthcare. Dr. Holzmeister has been a consultant to Biotronik. Dr. Zipes has been a consultant to Health System Networks and consults for and is a co-owner of Insight Telehealth. Dr. Anker has received fees and honoraria from St. Jude, Abbott Vascular, and several other drug and device companies. Dr. Tomaselli said that he had no disclosures. Dr. Cleland said that he has been a consultant to Biotronik, Medtronic, and St. Jude.
Depression in pediatric SLE matches average rates
Children and adolescents with systemic lupus erythematosus do not have significantly more depression than does the general population of the same age, according to an assessment of 54 pediatric lupus patients at one Canadian center.
"The overall percentage of children and adolescents reporting depressive symptoms was within the range reported in the general adolescent population," said Sara Ahola Kohut, Ph.D.
The finding suggests that "childhood-onset systemic lupus erythematosus [SLE] may not be an additional risk factor for depression," they reported (Lupus 2013;22:712-20).
But despite these "encouraging" findings, health care providers should be aware of, and continue to screen for, depressive symptoms in this at-risk population, children and adolescents with SLE, said Dr. Kohut, a pediatric pain researcher at the Hospital for Sick Children in Toronto.
Dr. Kohut and her associates at the hospital and at the University of Toronto assembled and reviewed two groups of SLE patients during April 2010-January 2012. All patients enrolled in the two groups had been diagnosed with SLE at the Hospital for Sick Children prior to reaching the age of 18. The 38 patients who supplied data in cohort 1 were aged 10-18 years and had been first diagnosed within the prior 18 months. Based on their demographics and disease characteristics, they were representative of a broader group of 41 other patients with childhood-onset SLE (cSLE) seen at the hospital during this period who did not participate in the study.
All 16 patients who provided data in the second cohort were aged 18-24 years and had been diagnosed at least 3 years previously at the hospital. Again, these patients did not significantly differ by demographics or disease duration from 81 other potentially eligible patients who elected not to participate.
The average age of the patients in cohort 1 was 14 years; 84% were girls, and they had been diagnosed with SLE for an average of 11 months. Sixteen of the 38 had been diagnosed with neuropsychiatric SLE (NPSLE).
Patients in cohort 1 underwent assessment with the Children’s Depression Inventory. Their mean total and subscale scores fell into the nondepressed range. Ten of the 38 (26%) had elevated depression scores, and three (8%) had clinically significant depression scores, but none of the patients endorsed suicidality.
The average age of patients in cohort 2 was 22 years; all were women, and their average time since diagnosis of SLE was nearly 8 years. Half had been diagnosed with NPSLE.
Cohort 2 patients were assessed with the Beck Depression Inventory II. Once again, the average scores of the 16 patients did not fall into the depressed range for both their total Beck score and their subscale scores, and none of the 16 endorsed suicidality. Seven of the 16 (44%) had elevated depression scores on the Beck Inventory, with two of them scoring in the moderate to severe range for depressive symptoms. The prevalence of elevated depression scores in this cohort included half of the 8 patients previously diagnosed with NPSLE and three of the eight without a neuropsychiatric diagnosis.
The prevalence of depression in cohorts 1 and 2 did not differ significantly.
The analysis also looked at the severity of the more prevalent symptoms of depression in each of the two cohorts. In cohort 1, the most severe symptoms were fatigue, school problems, indecisiveness, despair, and sleep disturbances. In cohort 2, the most common symptoms were physical, and the most severe symptoms were sleep disturbances, loss of energy, fatigue, and changes in appetite.
"SLE is a chronic disease with symptoms of active disease that may include fatigue, sleep disturbances, and anorexia; therefore it is not surprising that physical depressive symptoms were the most frequent," said Dr. Kohut and her associates. "It may be more useful to monitor affective mood disorders (e.g., irritability, negative mood, negative self-esteem, worthlessness) when assessing depression in cSLE patients as these symptoms are less likely to coincide with symptoms of active SLE leading to (falsely) elevated composite depression scores," they noted.
The percentage of cSLE patients in the study with elevated levels of negative mood measures, including affective symptoms, and cognitive depressive symptoms was consistent with what has been previously reported for the general adolescent population. In addition, the authors noted that the children and adolescents who had previously been diagnosed with NPSLE did not have significantly higher depression scores than those of the cSLE patients without the neuropsychiatric diagnosis.
"The CDI and BDI-II remain valid instruments to screen for mood disorders. However, our paper highlights the utility of identifying which items children and adolescents with cSLE endorse. The CDI includes subscales to facilitate the identification of mood-related indicators such as the Negative Mood subscale," Dr. Kohut said in an interview. "To screen specifically for affective mood indicators, health care professionals may ask about loss of interest, the last time the patients can remember enjoying themselves, or more generally about feelings of sadness or low mood."
The analysis also showed a significant link between higher corticosteroid dose and more symptoms of physical depression and lower self-esteem. The authors found this "not surprising" since the changes in physical appearance associated with higher dose corticosteroids could be expected to lead to issues with self-esteem and depressive symptoms.
The new finding that the prevalence of depression in children and young adults with cSLE matches the general population contrasts with what’s seen in adults with SLE, where the rate of depression exceeds the general adult prevalence. "There may be other factors affecting adults with SLE such as duration, severity or impact of illness, side effects from medications, loss of opportunities, available supports, etc.," Dr. Kohut said. "Further longitudinal research would be needed to clarify such differences in disease."
Dr. Kohut and her associates said that they had no disclosures.
On Twitter @mitchelzoler
Children and adolescents with systemic lupus erythematosus do not have significantly more depression than does the general population of the same age, according to an assessment of 54 pediatric lupus patients at one Canadian center.
"The overall percentage of children and adolescents reporting depressive symptoms was within the range reported in the general adolescent population," said Sara Ahola Kohut, Ph.D.
The finding suggests that "childhood-onset systemic lupus erythematosus [SLE] may not be an additional risk factor for depression," they reported (Lupus 2013;22:712-20).
But despite these "encouraging" findings, health care providers should be aware of, and continue to screen for, depressive symptoms in this at-risk population, children and adolescents with SLE, said Dr. Kohut, a pediatric pain researcher at the Hospital for Sick Children in Toronto.
Dr. Kohut and her associates at the hospital and at the University of Toronto assembled and reviewed two groups of SLE patients during April 2010-January 2012. All patients enrolled in the two groups had been diagnosed with SLE at the Hospital for Sick Children prior to reaching the age of 18. The 38 patients who supplied data in cohort 1 were aged 10-18 years and had been first diagnosed within the prior 18 months. Based on their demographics and disease characteristics, they were representative of a broader group of 41 other patients with childhood-onset SLE (cSLE) seen at the hospital during this period who did not participate in the study.
All 16 patients who provided data in the second cohort were aged 18-24 years and had been diagnosed at least 3 years previously at the hospital. Again, these patients did not significantly differ by demographics or disease duration from 81 other potentially eligible patients who elected not to participate.
The average age of the patients in cohort 1 was 14 years; 84% were girls, and they had been diagnosed with SLE for an average of 11 months. Sixteen of the 38 had been diagnosed with neuropsychiatric SLE (NPSLE).
Patients in cohort 1 underwent assessment with the Children’s Depression Inventory. Their mean total and subscale scores fell into the nondepressed range. Ten of the 38 (26%) had elevated depression scores, and three (8%) had clinically significant depression scores, but none of the patients endorsed suicidality.
The average age of patients in cohort 2 was 22 years; all were women, and their average time since diagnosis of SLE was nearly 8 years. Half had been diagnosed with NPSLE.
Cohort 2 patients were assessed with the Beck Depression Inventory II. Once again, the average scores of the 16 patients did not fall into the depressed range for both their total Beck score and their subscale scores, and none of the 16 endorsed suicidality. Seven of the 16 (44%) had elevated depression scores on the Beck Inventory, with two of them scoring in the moderate to severe range for depressive symptoms. The prevalence of elevated depression scores in this cohort included half of the 8 patients previously diagnosed with NPSLE and three of the eight without a neuropsychiatric diagnosis.
The prevalence of depression in cohorts 1 and 2 did not differ significantly.
The analysis also looked at the severity of the more prevalent symptoms of depression in each of the two cohorts. In cohort 1, the most severe symptoms were fatigue, school problems, indecisiveness, despair, and sleep disturbances. In cohort 2, the most common symptoms were physical, and the most severe symptoms were sleep disturbances, loss of energy, fatigue, and changes in appetite.
"SLE is a chronic disease with symptoms of active disease that may include fatigue, sleep disturbances, and anorexia; therefore it is not surprising that physical depressive symptoms were the most frequent," said Dr. Kohut and her associates. "It may be more useful to monitor affective mood disorders (e.g., irritability, negative mood, negative self-esteem, worthlessness) when assessing depression in cSLE patients as these symptoms are less likely to coincide with symptoms of active SLE leading to (falsely) elevated composite depression scores," they noted.
The percentage of cSLE patients in the study with elevated levels of negative mood measures, including affective symptoms, and cognitive depressive symptoms was consistent with what has been previously reported for the general adolescent population. In addition, the authors noted that the children and adolescents who had previously been diagnosed with NPSLE did not have significantly higher depression scores than those of the cSLE patients without the neuropsychiatric diagnosis.
"The CDI and BDI-II remain valid instruments to screen for mood disorders. However, our paper highlights the utility of identifying which items children and adolescents with cSLE endorse. The CDI includes subscales to facilitate the identification of mood-related indicators such as the Negative Mood subscale," Dr. Kohut said in an interview. "To screen specifically for affective mood indicators, health care professionals may ask about loss of interest, the last time the patients can remember enjoying themselves, or more generally about feelings of sadness or low mood."
The analysis also showed a significant link between higher corticosteroid dose and more symptoms of physical depression and lower self-esteem. The authors found this "not surprising" since the changes in physical appearance associated with higher dose corticosteroids could be expected to lead to issues with self-esteem and depressive symptoms.
The new finding that the prevalence of depression in children and young adults with cSLE matches the general population contrasts with what’s seen in adults with SLE, where the rate of depression exceeds the general adult prevalence. "There may be other factors affecting adults with SLE such as duration, severity or impact of illness, side effects from medications, loss of opportunities, available supports, etc.," Dr. Kohut said. "Further longitudinal research would be needed to clarify such differences in disease."
Dr. Kohut and her associates said that they had no disclosures.
On Twitter @mitchelzoler
Children and adolescents with systemic lupus erythematosus do not have significantly more depression than does the general population of the same age, according to an assessment of 54 pediatric lupus patients at one Canadian center.
"The overall percentage of children and adolescents reporting depressive symptoms was within the range reported in the general adolescent population," said Sara Ahola Kohut, Ph.D.
The finding suggests that "childhood-onset systemic lupus erythematosus [SLE] may not be an additional risk factor for depression," they reported (Lupus 2013;22:712-20).
But despite these "encouraging" findings, health care providers should be aware of, and continue to screen for, depressive symptoms in this at-risk population, children and adolescents with SLE, said Dr. Kohut, a pediatric pain researcher at the Hospital for Sick Children in Toronto.
Dr. Kohut and her associates at the hospital and at the University of Toronto assembled and reviewed two groups of SLE patients during April 2010-January 2012. All patients enrolled in the two groups had been diagnosed with SLE at the Hospital for Sick Children prior to reaching the age of 18. The 38 patients who supplied data in cohort 1 were aged 10-18 years and had been first diagnosed within the prior 18 months. Based on their demographics and disease characteristics, they were representative of a broader group of 41 other patients with childhood-onset SLE (cSLE) seen at the hospital during this period who did not participate in the study.
All 16 patients who provided data in the second cohort were aged 18-24 years and had been diagnosed at least 3 years previously at the hospital. Again, these patients did not significantly differ by demographics or disease duration from 81 other potentially eligible patients who elected not to participate.
The average age of the patients in cohort 1 was 14 years; 84% were girls, and they had been diagnosed with SLE for an average of 11 months. Sixteen of the 38 had been diagnosed with neuropsychiatric SLE (NPSLE).
Patients in cohort 1 underwent assessment with the Children’s Depression Inventory. Their mean total and subscale scores fell into the nondepressed range. Ten of the 38 (26%) had elevated depression scores, and three (8%) had clinically significant depression scores, but none of the patients endorsed suicidality.
The average age of patients in cohort 2 was 22 years; all were women, and their average time since diagnosis of SLE was nearly 8 years. Half had been diagnosed with NPSLE.
Cohort 2 patients were assessed with the Beck Depression Inventory II. Once again, the average scores of the 16 patients did not fall into the depressed range for both their total Beck score and their subscale scores, and none of the 16 endorsed suicidality. Seven of the 16 (44%) had elevated depression scores on the Beck Inventory, with two of them scoring in the moderate to severe range for depressive symptoms. The prevalence of elevated depression scores in this cohort included half of the 8 patients previously diagnosed with NPSLE and three of the eight without a neuropsychiatric diagnosis.
The prevalence of depression in cohorts 1 and 2 did not differ significantly.
The analysis also looked at the severity of the more prevalent symptoms of depression in each of the two cohorts. In cohort 1, the most severe symptoms were fatigue, school problems, indecisiveness, despair, and sleep disturbances. In cohort 2, the most common symptoms were physical, and the most severe symptoms were sleep disturbances, loss of energy, fatigue, and changes in appetite.
"SLE is a chronic disease with symptoms of active disease that may include fatigue, sleep disturbances, and anorexia; therefore it is not surprising that physical depressive symptoms were the most frequent," said Dr. Kohut and her associates. "It may be more useful to monitor affective mood disorders (e.g., irritability, negative mood, negative self-esteem, worthlessness) when assessing depression in cSLE patients as these symptoms are less likely to coincide with symptoms of active SLE leading to (falsely) elevated composite depression scores," they noted.
The percentage of cSLE patients in the study with elevated levels of negative mood measures, including affective symptoms, and cognitive depressive symptoms was consistent with what has been previously reported for the general adolescent population. In addition, the authors noted that the children and adolescents who had previously been diagnosed with NPSLE did not have significantly higher depression scores than those of the cSLE patients without the neuropsychiatric diagnosis.
"The CDI and BDI-II remain valid instruments to screen for mood disorders. However, our paper highlights the utility of identifying which items children and adolescents with cSLE endorse. The CDI includes subscales to facilitate the identification of mood-related indicators such as the Negative Mood subscale," Dr. Kohut said in an interview. "To screen specifically for affective mood indicators, health care professionals may ask about loss of interest, the last time the patients can remember enjoying themselves, or more generally about feelings of sadness or low mood."
The analysis also showed a significant link between higher corticosteroid dose and more symptoms of physical depression and lower self-esteem. The authors found this "not surprising" since the changes in physical appearance associated with higher dose corticosteroids could be expected to lead to issues with self-esteem and depressive symptoms.
The new finding that the prevalence of depression in children and young adults with cSLE matches the general population contrasts with what’s seen in adults with SLE, where the rate of depression exceeds the general adult prevalence. "There may be other factors affecting adults with SLE such as duration, severity or impact of illness, side effects from medications, loss of opportunities, available supports, etc.," Dr. Kohut said. "Further longitudinal research would be needed to clarify such differences in disease."
Dr. Kohut and her associates said that they had no disclosures.
On Twitter @mitchelzoler
FROM LUPUS
Major finding: The prevalence of depression in pediatric lupus was 26%-44%, not significantly different from reported levels in the general pediatric population.
Data source: An assessment of 54 children, adolescents, and young adults diagnosed with childhood-onset systemic lupus erythematosus at one Canadian center.
Disclosures: Dr. Kohut and her associates said that they had no disclosures.
Edoxaban pivotal trial shows VTE efficacy, safety (copy 1)
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler

|
| Dr. Steven Q. Simpson |
Dr. Steven Q. Simpson, FCCP, comments: This study is relevant, because investigators treated VTE patients in a standard fashion, but randomized to Factor Xa inhibition vs. warfarin.It is important that this strategy provides equivalent inhibition of recurrent clot, with a safety profile that is better. However, it is a very interesting finding that patients with evidence of right ventricular dysfunction actually had better outcomes on the new regimen than on warfarin.
We may now have entered the era where we can liberate our patients from the periodic blood draws, concerns about diet and concomitant medications, and much of the bleeding risk associated with warfarin therapy.Warfarin has saved many lives in its career, but it is time to consider retiring.

|
| Dr. Steven Q. Simpson |
Dr. Steven Q. Simpson, FCCP, comments: This study is relevant, because investigators treated VTE patients in a standard fashion, but randomized to Factor Xa inhibition vs. warfarin.It is important that this strategy provides equivalent inhibition of recurrent clot, with a safety profile that is better. However, it is a very interesting finding that patients with evidence of right ventricular dysfunction actually had better outcomes on the new regimen than on warfarin.
We may now have entered the era where we can liberate our patients from the periodic blood draws, concerns about diet and concomitant medications, and much of the bleeding risk associated with warfarin therapy.Warfarin has saved many lives in its career, but it is time to consider retiring.

|
| Dr. Steven Q. Simpson |
Dr. Steven Q. Simpson, FCCP, comments: This study is relevant, because investigators treated VTE patients in a standard fashion, but randomized to Factor Xa inhibition vs. warfarin.It is important that this strategy provides equivalent inhibition of recurrent clot, with a safety profile that is better. However, it is a very interesting finding that patients with evidence of right ventricular dysfunction actually had better outcomes on the new regimen than on warfarin.
We may now have entered the era where we can liberate our patients from the periodic blood draws, concerns about diet and concomitant medications, and much of the bleeding risk associated with warfarin therapy.Warfarin has saved many lives in its career, but it is time to consider retiring.
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler
AT ESC 2013
Major finding: Treatment of acute venous thromboembolism with edoxaban reduced clinically relevant bleeds by a relative 19%, compared with warfarin.
Data source: The data came from Hokusai-VTE, a multicenter, randomized, pivotal trial that enrolled 8,240 patients with acute venous thromboembolism.
Disclosures: Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing edoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Edoxaban pivotal trial shows VTE efficacy, safety
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler
It appears that the Hokusai-VTE results proved the hypothesis that edoxaban is noninferior to warfarin, and also important was that in higher-risk patients with significant pulmonary embolisms, edoxaban was better.
The emergence of the oral factor X inhibitors is giving physicians a new option for treatment. These drugs are much more convenient to use than warfarin; diet does not interfere with treatment, and blood draws aren’t necessary. It looks like the benefits from the new factor X inhibitors may be class effects; the results have been consistent from one drug to the next. But there is the limitation right now of their relatively high cost. I hope that eventually the cost differential won’t be as prohibitive as it probably is today for some patients.
Physicians and patients in the United States are slowly starting to use these drugs routinely in patients with VTE, and in other settings, such as to prevent clot formation and strokes in patients with atrial fibrillation. In mid-2013, about 10%-12% of patients with atrial fibrillation are being treated with one of the new oral anticoagulants, a group of drugs that also includes dabigatran (Pradaxa). I think there will gradually be increasing use of these drugs in place of warfarin as people grow more familiar and comfortable with them.
Dr. William A. Zoghbi is professor of medicine and director of the Cardiovascular Imaging Institute at the DeBakey Heart and Vascular Center of Methodist Hospital in Houston. He said that he had no relevant disclosures. Dr. Zoghbi made these comments in an interview.
It appears that the Hokusai-VTE results proved the hypothesis that edoxaban is noninferior to warfarin, and also important was that in higher-risk patients with significant pulmonary embolisms, edoxaban was better.
The emergence of the oral factor X inhibitors is giving physicians a new option for treatment. These drugs are much more convenient to use than warfarin; diet does not interfere with treatment, and blood draws aren’t necessary. It looks like the benefits from the new factor X inhibitors may be class effects; the results have been consistent from one drug to the next. But there is the limitation right now of their relatively high cost. I hope that eventually the cost differential won’t be as prohibitive as it probably is today for some patients.
Physicians and patients in the United States are slowly starting to use these drugs routinely in patients with VTE, and in other settings, such as to prevent clot formation and strokes in patients with atrial fibrillation. In mid-2013, about 10%-12% of patients with atrial fibrillation are being treated with one of the new oral anticoagulants, a group of drugs that also includes dabigatran (Pradaxa). I think there will gradually be increasing use of these drugs in place of warfarin as people grow more familiar and comfortable with them.
Dr. William A. Zoghbi is professor of medicine and director of the Cardiovascular Imaging Institute at the DeBakey Heart and Vascular Center of Methodist Hospital in Houston. He said that he had no relevant disclosures. Dr. Zoghbi made these comments in an interview.
It appears that the Hokusai-VTE results proved the hypothesis that edoxaban is noninferior to warfarin, and also important was that in higher-risk patients with significant pulmonary embolisms, edoxaban was better.
The emergence of the oral factor X inhibitors is giving physicians a new option for treatment. These drugs are much more convenient to use than warfarin; diet does not interfere with treatment, and blood draws aren’t necessary. It looks like the benefits from the new factor X inhibitors may be class effects; the results have been consistent from one drug to the next. But there is the limitation right now of their relatively high cost. I hope that eventually the cost differential won’t be as prohibitive as it probably is today for some patients.
Physicians and patients in the United States are slowly starting to use these drugs routinely in patients with VTE, and in other settings, such as to prevent clot formation and strokes in patients with atrial fibrillation. In mid-2013, about 10%-12% of patients with atrial fibrillation are being treated with one of the new oral anticoagulants, a group of drugs that also includes dabigatran (Pradaxa). I think there will gradually be increasing use of these drugs in place of warfarin as people grow more familiar and comfortable with them.
Dr. William A. Zoghbi is professor of medicine and director of the Cardiovascular Imaging Institute at the DeBakey Heart and Vascular Center of Methodist Hospital in Houston. He said that he had no relevant disclosures. Dr. Zoghbi made these comments in an interview.
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler
AMSTERDAM – The growing role for the new factor Xa inhibitors in treating acute venous thromboembolism received another boost with the performance of a new drug from the class, edoxaban, in a pivotal, randomized, international trial with more than 8,000 patients.
Edoxaban showed noninferior efficacy and superior safety compared with the standard treatment, warfarin, during 12 months of follow-up, performance that closely matched prior results for the first two direct factor Xa inhibitors that were tested for treating venous thromboembolism (VTE), rivaroxaban (Xarelto) and apixaban (Eliquis). The edoxaban trial also showed an important new feature for drugs in this class, a statistically significant benefit compared with warfarin in the roughly one-third of enrolled patients with a severe pulmonary embolism causing right ventricular dysfunction.
"The most interesting observation is that the third of patients [with pulmonary embolism and] with right ventricular dysfunction were better off with edoxaban," Dr. Harry R. Büller said at the European Society of Cardiology Congress 2013.
"A criticism raised with [the pivotal rivaroxaban and apixaban trials treating acute VTE] was that the investigators were reluctant not to use low molecular weight heparin in the patients with large clots. In that group we’ve made a novel observation" in this prespecified subgroup analysis, said Dr. Büller, professor and chairman of vascular medicine at the Academic Medical Center in Amsterdam.
Investigators in the edoxaban trial may have been more willing to enroll their severely ill VTE patients because the treatment regimens compared began all patients with at least 5 days of treatment with either enoxaparin or unfractionated heparin. Intravenous treatment stopped after about a week and then continued with either oral warfarin or edoxaban. This treatment strategy contrasted with the prior rivaroxaban (EINSTEIN, N. Engl. J. Med. 2012;366:1287-97 and apixaban (AMPLIFY, N. Engl. J. Med. 2013;369:799-808) pivotal studies that began acute VTE patients on the oral drugs from the start of treatment. In the edoxaban study, participating physicians enrolled "the full spectrum of VTE patients," Dr. Büller said.
Physicians "feel comfortable intervening with heparin in these patients because you have quick, intravenous treatment with an anticoagulant," commented Dr. William A. Zoghbi, professor of medicine and director of the Cardiovascular Imaging Institute at the Debakey Heart and Vascular Center at Methodist Hospital in Houston. The results showing superior efficacy of edoxaban over warfarin in the most severely affected pulmonary embolism patients "makes you more confident that in the higher-risk patients edoxaban was not only not inferior but may even have an advantage," Dr. Zoghbi said in an interview.
The Hokusai-VTE trial randomized 4,921 patients with deep-vein thrombosis and 3,319 patients with pulmonary embolism at 439 centers in 37 countries during January 2010–October 2012. Patients averaged 56 years of age, a bit more than half were men, and they received heparin for a median of 7 days.
After 1 year, the primary efficacy endpoint of recurrent VTE in all patients occurred in 3.2% of the 4,118 patients randomized to received edoxaban and in 3.5% of the 4,122 randomized to receive warfarin, results that meet the study’s criterion for noninferiority.
Among the prespecified subgroup of pulmonary embolism patients with a larger clot and inferred right-ventricular dysfunction – based on both the anatomic size of their clot and on their blood level of N-terminal pro-brain natriuretic peptide – the rate of recurrent VTE during follow-up was 3.3% in the edoxaban group and 6.2% in the warfarin group, a statistically significant difference.
The study’s primary safety outcome was the rate of clinically relevant major or nonmajor bleeds, which occurred in 8.5% of the edoxaban patients and in 10.3% of the warfarin patients, for a relative hazard reduction of 19% by edoxaban that was statistically significant for superiority, reported Dr. Büller. Concurrent with his talk at the meeting a report of the trial results was published online (N. Engl. J. Med. 2013;doi:10.1056NEJMoa1306638).
The safety data also showed that the edoxaban-treated patients had a total of 5 intracranial or retroperitoneal bleeds, compared with 22 in the patients treated with warfarin, a difference that Dr. Büller said he believed was real and due to the effect of warfarin on clotting factor VII, and is something that has also been seen in the trials testing the other new factor Xa inhibitors.
The Hokusai-VTE study is unique among the trials of oral factor Xa inhibitors by being the only study that allowed for a flexible duration of anticoagulation treatment, followed all patients for 12 months, and used imaging and biomarkers to risk-stratify patients to prove efficacy in patients with severe pulmonary embolism, commented Dr. Stavros V. Konstantinides, professor and deputy scientific director of the Center for Thrombosis and Haemostasis of Johannes Gutenberg University in Mainz, Germany.
The data collected on all three oral factor X inhibitors so far suggest that they all are "noninferior to standard treatment and are also not inferior and most likely superior for safety, with apixaban showing the best safety profile so far," said Dr. Konstantinides, who was the designated discussant for Dr. Büller’s report. But Dr. Konstantinides also warned that these new drugs will need to "justify their high cost" by showing improvements in patient satisfaction with their treatment and quality of life, and by lowering overall health-care costs by lowering the number of rehospitalizations in VTE patients.
Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing endoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
Twitter:@mitchelzoler
AT ESC 2013
Major finding: Treatment of acute venous thromboembolism with edoxaban reduced clinically relevant bleeds by a relative 19%, compared with warfarin.
Data source: The data came from Hokusai-VTE, a multicenter, randomized, pivotal trial that enrolled 8,240 patients with acute venous thromboembolism.
Disclosures: Hokusai-VTE was sponsored by Daiichi-Sankyo, the company developing edoxaban. Dr. Büller said that he has received honoraria from Daiichi Sankyo, as well as from Bayer, Boehringer Ingelheim, Pfizer/Bristol-Myers Squibb, Isis, and Thrombogenics. Dr. Büller also served as a principal investigator for the VTE pivotal trials of rivaroxaban and apixaban. Dr. Zoghbi said that he had no relevant disclosures. Dr. Konstantinides has been a speaker for and an advisor to Boehringer Ingelheim, Bayer, and Pfizer/Bristol-Myers Squibb.
PCI in noninfarct coronaries helps STEMI patients
AMSTERDAM – In patients with acute ST-segment elevation myocardial infarction, stenting of significant coronary stenoses not responsible for the infarction as well as the infarct-producing lesion led to substantially better outcomes than an intervention that only targeted the infarct-related stenosis in a randomized, multicenter trial with 465 patients.
The results appeared to refute the prevailing recommendation from cardiology societies to limit the percutaneous coronary intervention (PCI) done during acute treatment of ST-segment elevation myocardial infarction (STEMI) to the infarct-related lesion, especially when the secondary lesions are not clearly causing ongoing hemodynamic instability.
The new findings "make it clear that preventive PCI is a better strategy than restricting a further intervention to those patients with refractory angina or a subsequent myocardial infarction," Dr. David S. Wald reported on Sept. 1 at the annual congress of the European Society of Cardiology. Simultaneous with Dr. Wald’s report, the results appeared in an article published online (N. Engl. J. Med. 2013 [doi:10.156/NEJMoa1305520]).
Current guidelines on management of STEMI recommend PCI for the infarct-related artery only (Eur. Heart J. 2012;33:2569-619; Circulation 2013;127:529-55), primarily because, until now, scant evidence existed that a different strategy helped patients. "This uncertainty has led to variations in practice, with some cardiologists performing immediate preventive PCI in spite of the guidelines, some delaying preventive PCI until recovery from the acute episode, and others limiting the procedure to patients with recurrent symptoms or evidence of ischemia," noted Dr. Wald, an interventional cardiologist at the Wolfson Institute of Preventive Medicine of the Queen Mary University of London.
Dr. Wald acknowledged, however, that his study did not address whether patients would be better served by a staged approach that delayed preventive PCI of significant, noninfarct related stenoses in a second PCI procedure, whether the benefits extend to treating noninfarct stenoses that occlude less than half of a coronary artery, and whether a similar strategy would help patients with non-ST–elevation MI (NSTEMI).
The Preventive Angioplasty in Acute Myocardial Infarction (PRAMI) trial enrolled consecutive patients of any age at five U.K. centers during April 2008 to January 2013. The study focused on STEMI patients because patients with a NSTEMI often do not have a clearly identifiable infarct-related artery. In addition, to qualify for entry, the patient’s infarct-related artery had to have been successfully treated by PCI and they had to have at least one other coronary artery with at least 50% stenosis judged treatable by PCI. Patients with coronary anatomy more appropriately treated with bypass surgery were excluded. The enrolled patients averaged 62 years of age, about three-quarters were men, and about 18% had diabetes. Roughly two-thirds had a second coronary artery with a greater than 50% stenosis, and the remaining patients had two coronary arteries with a significant lesion.
After successful treatment of the infarct-producing artery, patients with additional, significant lesions judged appropriate for PCI were randomized to either have their other significant lesions treated during the same procedure or to undergo no further intervention. This resulted in an average of 1.36 additional arteries undergoing PCI in the 234 patients randomized to the more aggressive protocol.
Following the PCI procedures, all patients received standard medical treatment: daily treatment with aspirin and a second antiplatelet drug, clopidogrel (Plavix), prasugrel (Effient), or ticagrelor (Brilinta). About 95% of patients in both study arms also received treatment with a statin, and about 90% received a beta-blocker and an angiotensin-converting enzyme inhibitor.
During an average follow-up of 23 months, the combined rate of death from cardiac causes, nonfatal MI, or refractory angina – the study’s primary endpoint – occurred in 9% of the patients randomized to immediate PCI of their noninfarct-related stenoses and in 23% of the 231 patients in the control arm, a statistically significant difference. The 14-percentage-point absolute reduction in endpoints translated into a 65% relative risk reduction. The rate of cardiac death or nonfatal MI was 5% in the patients who received added PCI while in the catheterization laboratory and 12% among those only treated in their infarct-related artery, also a statistically significant difference.
Additional analyses also showed statistically significantly lower rates of both nonfatal MI and refractory angina in the patients treated in multiple arteries when these endpoints were tallied individually and a strong trend toward fewer cardiac deaths in these patients as well. Kaplan-Meier analysis showed that the risk reduction in the preventive-PCI group was evident after 6 months of follow-up and then increased with continued follow-up.
These results were not materially affected by patient age, sex, diabetes status, infarct location, or the number of coronary arteries with significant stenoses. In addition, the complication rates, including procedure-related strokes, bleeding requiring transfusions or surgery, and contrast-induced nephropathy requiring dialysis, were similar in the two study arms.
The results were consistent with reports from two prior randomized trials that also assessed the value of preventive PCI in patients with acute STEMI, but in fewer patients. One of these prior studies involved a total of 69 patients (Int. J. Cardiovasc. Intervent. 2004;6:128-33), and the second enrolled a total of 214 patients (Heart 2010;96:662-7); both were limited by a lack of statistical power, and both relied on repeat revascularization as an endpoint, which may be subject to bias, Dr. Wald said.
Dr. Wald said that he a director for and shareholder in Polypill Ltd.
On Twitter @mitchelzoler
Based on this report from the PRAMI trial, we can no longer assume that secondary lesions in acute myocardial infarction are innocent until proven guilty.
It has been a core belief in interventional cardiology that percutaneous coronary intervention in noninfarct lesions does not prevent death and MI. Cardiologists have refrained from treating anything but the infarct lesion acutely, and they usually withhold further treatment unless a patient is symptomatic.
But patients with acute ST-segment elevation MI have a substantial risk for early, recurrent events, in contrast to patients with stable angina. Why? Coronary artery disease is accompanied by systemic abnormalities in coagulation, inflammation, and endothelial function, with multiple inflamed lesions. Patients with acute coronary syndrome have prominent, systemic derangements in these processes. The aggressive, acute treatment used in PRAMI may have stabilized these not-so-innocent lesions.
The PRAMI findings suggest that there are no healthy coronary arteries in a patient with acute STEMI. Does this mean that these patients need extensive revascularization? It is plausible that the risk from noninfarct lesions is independent of their hemodynamic severity.
The strategy employed in this study differs markedly from current practice. Guidelines have cautioned against treating multiple vessels during acute STEMI, particularly when the secondary sites are not clearly causing ongoing hemodynamic instability. The PRAMI results suggest that widening the scope of interventional therapy in acute STEMI patients is a promising new approach to management.
Dr. Laura Mauri is an interventional cardiologist at Brigham and Women’s Hospital, professor of medicine at Harvard University, and chief scientific officer of the Harvard Clinical Research Institute in Boston. She has been a consultant to Biotronik, has been on an advisory board of St. Jude, and she has received research grants from seven other drug or device-manufacturing companies. She made these comments in an editorial that accompanied the published version of the PRAMI report (N. Engl. J. Med. 2013 [doi:10.1056/NEJMe1309383]).
Based on this report from the PRAMI trial, we can no longer assume that secondary lesions in acute myocardial infarction are innocent until proven guilty.
It has been a core belief in interventional cardiology that percutaneous coronary intervention in noninfarct lesions does not prevent death and MI. Cardiologists have refrained from treating anything but the infarct lesion acutely, and they usually withhold further treatment unless a patient is symptomatic.
But patients with acute ST-segment elevation MI have a substantial risk for early, recurrent events, in contrast to patients with stable angina. Why? Coronary artery disease is accompanied by systemic abnormalities in coagulation, inflammation, and endothelial function, with multiple inflamed lesions. Patients with acute coronary syndrome have prominent, systemic derangements in these processes. The aggressive, acute treatment used in PRAMI may have stabilized these not-so-innocent lesions.
The PRAMI findings suggest that there are no healthy coronary arteries in a patient with acute STEMI. Does this mean that these patients need extensive revascularization? It is plausible that the risk from noninfarct lesions is independent of their hemodynamic severity.
The strategy employed in this study differs markedly from current practice. Guidelines have cautioned against treating multiple vessels during acute STEMI, particularly when the secondary sites are not clearly causing ongoing hemodynamic instability. The PRAMI results suggest that widening the scope of interventional therapy in acute STEMI patients is a promising new approach to management.
Dr. Laura Mauri is an interventional cardiologist at Brigham and Women’s Hospital, professor of medicine at Harvard University, and chief scientific officer of the Harvard Clinical Research Institute in Boston. She has been a consultant to Biotronik, has been on an advisory board of St. Jude, and she has received research grants from seven other drug or device-manufacturing companies. She made these comments in an editorial that accompanied the published version of the PRAMI report (N. Engl. J. Med. 2013 [doi:10.1056/NEJMe1309383]).
Based on this report from the PRAMI trial, we can no longer assume that secondary lesions in acute myocardial infarction are innocent until proven guilty.
It has been a core belief in interventional cardiology that percutaneous coronary intervention in noninfarct lesions does not prevent death and MI. Cardiologists have refrained from treating anything but the infarct lesion acutely, and they usually withhold further treatment unless a patient is symptomatic.
But patients with acute ST-segment elevation MI have a substantial risk for early, recurrent events, in contrast to patients with stable angina. Why? Coronary artery disease is accompanied by systemic abnormalities in coagulation, inflammation, and endothelial function, with multiple inflamed lesions. Patients with acute coronary syndrome have prominent, systemic derangements in these processes. The aggressive, acute treatment used in PRAMI may have stabilized these not-so-innocent lesions.
The PRAMI findings suggest that there are no healthy coronary arteries in a patient with acute STEMI. Does this mean that these patients need extensive revascularization? It is plausible that the risk from noninfarct lesions is independent of their hemodynamic severity.
The strategy employed in this study differs markedly from current practice. Guidelines have cautioned against treating multiple vessels during acute STEMI, particularly when the secondary sites are not clearly causing ongoing hemodynamic instability. The PRAMI results suggest that widening the scope of interventional therapy in acute STEMI patients is a promising new approach to management.
Dr. Laura Mauri is an interventional cardiologist at Brigham and Women’s Hospital, professor of medicine at Harvard University, and chief scientific officer of the Harvard Clinical Research Institute in Boston. She has been a consultant to Biotronik, has been on an advisory board of St. Jude, and she has received research grants from seven other drug or device-manufacturing companies. She made these comments in an editorial that accompanied the published version of the PRAMI report (N. Engl. J. Med. 2013 [doi:10.1056/NEJMe1309383]).
AMSTERDAM – In patients with acute ST-segment elevation myocardial infarction, stenting of significant coronary stenoses not responsible for the infarction as well as the infarct-producing lesion led to substantially better outcomes than an intervention that only targeted the infarct-related stenosis in a randomized, multicenter trial with 465 patients.
The results appeared to refute the prevailing recommendation from cardiology societies to limit the percutaneous coronary intervention (PCI) done during acute treatment of ST-segment elevation myocardial infarction (STEMI) to the infarct-related lesion, especially when the secondary lesions are not clearly causing ongoing hemodynamic instability.
The new findings "make it clear that preventive PCI is a better strategy than restricting a further intervention to those patients with refractory angina or a subsequent myocardial infarction," Dr. David S. Wald reported on Sept. 1 at the annual congress of the European Society of Cardiology. Simultaneous with Dr. Wald’s report, the results appeared in an article published online (N. Engl. J. Med. 2013 [doi:10.156/NEJMoa1305520]).
Current guidelines on management of STEMI recommend PCI for the infarct-related artery only (Eur. Heart J. 2012;33:2569-619; Circulation 2013;127:529-55), primarily because, until now, scant evidence existed that a different strategy helped patients. "This uncertainty has led to variations in practice, with some cardiologists performing immediate preventive PCI in spite of the guidelines, some delaying preventive PCI until recovery from the acute episode, and others limiting the procedure to patients with recurrent symptoms or evidence of ischemia," noted Dr. Wald, an interventional cardiologist at the Wolfson Institute of Preventive Medicine of the Queen Mary University of London.
Dr. Wald acknowledged, however, that his study did not address whether patients would be better served by a staged approach that delayed preventive PCI of significant, noninfarct related stenoses in a second PCI procedure, whether the benefits extend to treating noninfarct stenoses that occlude less than half of a coronary artery, and whether a similar strategy would help patients with non-ST–elevation MI (NSTEMI).
The Preventive Angioplasty in Acute Myocardial Infarction (PRAMI) trial enrolled consecutive patients of any age at five U.K. centers during April 2008 to January 2013. The study focused on STEMI patients because patients with a NSTEMI often do not have a clearly identifiable infarct-related artery. In addition, to qualify for entry, the patient’s infarct-related artery had to have been successfully treated by PCI and they had to have at least one other coronary artery with at least 50% stenosis judged treatable by PCI. Patients with coronary anatomy more appropriately treated with bypass surgery were excluded. The enrolled patients averaged 62 years of age, about three-quarters were men, and about 18% had diabetes. Roughly two-thirds had a second coronary artery with a greater than 50% stenosis, and the remaining patients had two coronary arteries with a significant lesion.
After successful treatment of the infarct-producing artery, patients with additional, significant lesions judged appropriate for PCI were randomized to either have their other significant lesions treated during the same procedure or to undergo no further intervention. This resulted in an average of 1.36 additional arteries undergoing PCI in the 234 patients randomized to the more aggressive protocol.
Following the PCI procedures, all patients received standard medical treatment: daily treatment with aspirin and a second antiplatelet drug, clopidogrel (Plavix), prasugrel (Effient), or ticagrelor (Brilinta). About 95% of patients in both study arms also received treatment with a statin, and about 90% received a beta-blocker and an angiotensin-converting enzyme inhibitor.
During an average follow-up of 23 months, the combined rate of death from cardiac causes, nonfatal MI, or refractory angina – the study’s primary endpoint – occurred in 9% of the patients randomized to immediate PCI of their noninfarct-related stenoses and in 23% of the 231 patients in the control arm, a statistically significant difference. The 14-percentage-point absolute reduction in endpoints translated into a 65% relative risk reduction. The rate of cardiac death or nonfatal MI was 5% in the patients who received added PCI while in the catheterization laboratory and 12% among those only treated in their infarct-related artery, also a statistically significant difference.
Additional analyses also showed statistically significantly lower rates of both nonfatal MI and refractory angina in the patients treated in multiple arteries when these endpoints were tallied individually and a strong trend toward fewer cardiac deaths in these patients as well. Kaplan-Meier analysis showed that the risk reduction in the preventive-PCI group was evident after 6 months of follow-up and then increased with continued follow-up.
These results were not materially affected by patient age, sex, diabetes status, infarct location, or the number of coronary arteries with significant stenoses. In addition, the complication rates, including procedure-related strokes, bleeding requiring transfusions or surgery, and contrast-induced nephropathy requiring dialysis, were similar in the two study arms.
The results were consistent with reports from two prior randomized trials that also assessed the value of preventive PCI in patients with acute STEMI, but in fewer patients. One of these prior studies involved a total of 69 patients (Int. J. Cardiovasc. Intervent. 2004;6:128-33), and the second enrolled a total of 214 patients (Heart 2010;96:662-7); both were limited by a lack of statistical power, and both relied on repeat revascularization as an endpoint, which may be subject to bias, Dr. Wald said.
Dr. Wald said that he a director for and shareholder in Polypill Ltd.
On Twitter @mitchelzoler
AMSTERDAM – In patients with acute ST-segment elevation myocardial infarction, stenting of significant coronary stenoses not responsible for the infarction as well as the infarct-producing lesion led to substantially better outcomes than an intervention that only targeted the infarct-related stenosis in a randomized, multicenter trial with 465 patients.
The results appeared to refute the prevailing recommendation from cardiology societies to limit the percutaneous coronary intervention (PCI) done during acute treatment of ST-segment elevation myocardial infarction (STEMI) to the infarct-related lesion, especially when the secondary lesions are not clearly causing ongoing hemodynamic instability.
The new findings "make it clear that preventive PCI is a better strategy than restricting a further intervention to those patients with refractory angina or a subsequent myocardial infarction," Dr. David S. Wald reported on Sept. 1 at the annual congress of the European Society of Cardiology. Simultaneous with Dr. Wald’s report, the results appeared in an article published online (N. Engl. J. Med. 2013 [doi:10.156/NEJMoa1305520]).
Current guidelines on management of STEMI recommend PCI for the infarct-related artery only (Eur. Heart J. 2012;33:2569-619; Circulation 2013;127:529-55), primarily because, until now, scant evidence existed that a different strategy helped patients. "This uncertainty has led to variations in practice, with some cardiologists performing immediate preventive PCI in spite of the guidelines, some delaying preventive PCI until recovery from the acute episode, and others limiting the procedure to patients with recurrent symptoms or evidence of ischemia," noted Dr. Wald, an interventional cardiologist at the Wolfson Institute of Preventive Medicine of the Queen Mary University of London.
Dr. Wald acknowledged, however, that his study did not address whether patients would be better served by a staged approach that delayed preventive PCI of significant, noninfarct related stenoses in a second PCI procedure, whether the benefits extend to treating noninfarct stenoses that occlude less than half of a coronary artery, and whether a similar strategy would help patients with non-ST–elevation MI (NSTEMI).
The Preventive Angioplasty in Acute Myocardial Infarction (PRAMI) trial enrolled consecutive patients of any age at five U.K. centers during April 2008 to January 2013. The study focused on STEMI patients because patients with a NSTEMI often do not have a clearly identifiable infarct-related artery. In addition, to qualify for entry, the patient’s infarct-related artery had to have been successfully treated by PCI and they had to have at least one other coronary artery with at least 50% stenosis judged treatable by PCI. Patients with coronary anatomy more appropriately treated with bypass surgery were excluded. The enrolled patients averaged 62 years of age, about three-quarters were men, and about 18% had diabetes. Roughly two-thirds had a second coronary artery with a greater than 50% stenosis, and the remaining patients had two coronary arteries with a significant lesion.
After successful treatment of the infarct-producing artery, patients with additional, significant lesions judged appropriate for PCI were randomized to either have their other significant lesions treated during the same procedure or to undergo no further intervention. This resulted in an average of 1.36 additional arteries undergoing PCI in the 234 patients randomized to the more aggressive protocol.
Following the PCI procedures, all patients received standard medical treatment: daily treatment with aspirin and a second antiplatelet drug, clopidogrel (Plavix), prasugrel (Effient), or ticagrelor (Brilinta). About 95% of patients in both study arms also received treatment with a statin, and about 90% received a beta-blocker and an angiotensin-converting enzyme inhibitor.
During an average follow-up of 23 months, the combined rate of death from cardiac causes, nonfatal MI, or refractory angina – the study’s primary endpoint – occurred in 9% of the patients randomized to immediate PCI of their noninfarct-related stenoses and in 23% of the 231 patients in the control arm, a statistically significant difference. The 14-percentage-point absolute reduction in endpoints translated into a 65% relative risk reduction. The rate of cardiac death or nonfatal MI was 5% in the patients who received added PCI while in the catheterization laboratory and 12% among those only treated in their infarct-related artery, also a statistically significant difference.
Additional analyses also showed statistically significantly lower rates of both nonfatal MI and refractory angina in the patients treated in multiple arteries when these endpoints were tallied individually and a strong trend toward fewer cardiac deaths in these patients as well. Kaplan-Meier analysis showed that the risk reduction in the preventive-PCI group was evident after 6 months of follow-up and then increased with continued follow-up.
These results were not materially affected by patient age, sex, diabetes status, infarct location, or the number of coronary arteries with significant stenoses. In addition, the complication rates, including procedure-related strokes, bleeding requiring transfusions or surgery, and contrast-induced nephropathy requiring dialysis, were similar in the two study arms.
The results were consistent with reports from two prior randomized trials that also assessed the value of preventive PCI in patients with acute STEMI, but in fewer patients. One of these prior studies involved a total of 69 patients (Int. J. Cardiovasc. Intervent. 2004;6:128-33), and the second enrolled a total of 214 patients (Heart 2010;96:662-7); both were limited by a lack of statistical power, and both relied on repeat revascularization as an endpoint, which may be subject to bias, Dr. Wald said.
Dr. Wald said that he a director for and shareholder in Polypill Ltd.
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2013
Major finding: Preventive PCI of significant, noninfarct-related coronary stenoses in acute STEMI cut the combined rate of death from cardiac causes, nonfatal MI, or refractory angina by 14 percentage points, compared with controls – a 65% relative risk reduction.
Data source: A multicenter, randomized trial with 465 patients.
Disclosures: Dr. Wald said that he a director for and shareholder in Polypill Ltd.
Acute pulmonary embolism doesn’t always require hospitalization
AMSTERDAM – Two-thirds of patients who presented to the emergency department of a U.S. tertiary care hospital with an acute pulmonary embolism had no acute deterioration and required no short-term hospital-based interventions, in an analysis of 298 patients seen over a 2-year period.
The finding "supports the assertion that outpatient treatment of patients with pulmonary embolism [PE] is safe," said Dr. Christopher Kabrhel at the 24th Congress of the International Society on Thrombosis and Haemostasis.
"We want to identify patients for whom nothing bad will happen. We showed that two-thirds of patients did well and didn’t need anything from the hospital and didn’t benefit from being in the hospital. We need to identify some of these patients," soon after they present in the emergency department, said Dr. Kabrhel, a surgeon and emergency-medicine physician at Massachusetts General Hospital and Harvard Medical School in Boston. If reliable risk markers can be found with further research, "perhaps we can identify half of the two-thirds—a third of all patients who come to the emergency department with a PE—who we know will be safe with outpatient treatment so we can send those patients home from the emergency department and not admit them."
Most symptomatic U.S. patients who come to an emergency department, and are diagnosed with a PE are immediately admitted to the hospital. In the current study, the hospitalization rate was 92% with a median length of stay of 3 days. "We need a better rule to decide whether a patient needs hospitalization. We need to find which patients benefit from hospitalization," Dr. Kabrhel said in an interview.
He and his associates reviewed 298 adults 18 years or older who presented to the Massachusetts General Hospital emergency department during October 2009 through December 2011 with a radiographically proven PE diagnosed within 24 hours of arrival. They averaged 59 years old, half were women, and the most common comorbidity was malignancy in 107 patients (36%).
The study’s primary outcome was any clinical deterioration or need for hospital-based intervention during the 5 days following presentation at the emergency department, including the need for advanced cardiac life support, the development of a new cardiac dysrhythmia, the development of hypoxia or hypotension, the need for thrombolysis or thrombectomy, recurrent PE, or death. These events occurred in 99 patients (33%); of these, 28 patients (9% of the total group) had "severe" deterioration or required a "major" intervention. Twelve patients (4%) died within 30 days of their initial emergency presentation. The most common acute complication was the need for respiratory support, in about 55 patients, followed by hypotension, in about 34.
A multivariate analysis identified five baseline factors that significantly correlated with the primary outcome. Patients who had normal vital signs at baseline had a 79% reduced rate of significant deterioration or need for hospital-based intervention. The other four factors were linked with increased rates of deterioration and need for intervention: Right heart strain caused by the PE and identified by an echocardiogram boosted the risk of a bad outcome more than fourfold, coronary disease and cerebrovascular disease each were tied to a more than threefold increased rate, and residual deep vein thrombosis was linked with a more than doubled rate of bad outcomes.
The subset of patients with the most severe outcomes had only one direct correlation with bad outcomes, right heart strain on echo. This subset of patients also showed a protective link against bad outcomes when their systolic blood pressure never fell below 90 mm Hg.
In contrast to these factors linked to 5-day outcomes, two different types of patient factors were significantly linked with 30-day mortality: having a malignancy and having chronic lung disease.
"Previously validated clinical prediction rules that looked at outcomes after PE were primarily validated based on 30-day mortality or recurrent PE, and included factors like having cancer, heart failure, or chronic lung disease. But these scores are only able to predict the outcomes we examined with 70% sensitivity," Dr. Kabrhel said. He found this out by running the numbers he collected through three validated scores for predicting PE outcome: the Geneva Prediction Score (Ann. Intern. Med. 2006;144:165-71), the Severity Index (Am. J. Respir. Crit. Care Med. 2005; 172:1041-6), and the Simplified Pulmonary Embolism Severity Index (Arch. Intern. Med. 2010;170:1383-9). "Predictors of all-cause 30-day mortality are different than predictors of short-term outcomes" in PE patients, he said.
"We found that echo is a very good predictor of short-term outcomes, and also abnormal vital signs. The key point is we need to look at outcomes that are relevant to the decisions made" in the emergency department, Dr. Kabrhel said. "Looking at 30-day mortality in patients who are only hospitalized for 3 days doesn’t really inform the decision on who should be in the hospital. I would suggest caution on using [prediction] tools validated against 30-day mortality and recurrent PE to determine what to do acutely. We need better rules to decide which PE patients need hospitalization."
Dr. Kabrhel said he has been a consultant to Diagnostica Stago, and is an officer for LitPulse.
On Twitter @mitchelzoler
AMSTERDAM – Two-thirds of patients who presented to the emergency department of a U.S. tertiary care hospital with an acute pulmonary embolism had no acute deterioration and required no short-term hospital-based interventions, in an analysis of 298 patients seen over a 2-year period.
The finding "supports the assertion that outpatient treatment of patients with pulmonary embolism [PE] is safe," said Dr. Christopher Kabrhel at the 24th Congress of the International Society on Thrombosis and Haemostasis.
"We want to identify patients for whom nothing bad will happen. We showed that two-thirds of patients did well and didn’t need anything from the hospital and didn’t benefit from being in the hospital. We need to identify some of these patients," soon after they present in the emergency department, said Dr. Kabrhel, a surgeon and emergency-medicine physician at Massachusetts General Hospital and Harvard Medical School in Boston. If reliable risk markers can be found with further research, "perhaps we can identify half of the two-thirds—a third of all patients who come to the emergency department with a PE—who we know will be safe with outpatient treatment so we can send those patients home from the emergency department and not admit them."
Most symptomatic U.S. patients who come to an emergency department, and are diagnosed with a PE are immediately admitted to the hospital. In the current study, the hospitalization rate was 92% with a median length of stay of 3 days. "We need a better rule to decide whether a patient needs hospitalization. We need to find which patients benefit from hospitalization," Dr. Kabrhel said in an interview.
He and his associates reviewed 298 adults 18 years or older who presented to the Massachusetts General Hospital emergency department during October 2009 through December 2011 with a radiographically proven PE diagnosed within 24 hours of arrival. They averaged 59 years old, half were women, and the most common comorbidity was malignancy in 107 patients (36%).
The study’s primary outcome was any clinical deterioration or need for hospital-based intervention during the 5 days following presentation at the emergency department, including the need for advanced cardiac life support, the development of a new cardiac dysrhythmia, the development of hypoxia or hypotension, the need for thrombolysis or thrombectomy, recurrent PE, or death. These events occurred in 99 patients (33%); of these, 28 patients (9% of the total group) had "severe" deterioration or required a "major" intervention. Twelve patients (4%) died within 30 days of their initial emergency presentation. The most common acute complication was the need for respiratory support, in about 55 patients, followed by hypotension, in about 34.
A multivariate analysis identified five baseline factors that significantly correlated with the primary outcome. Patients who had normal vital signs at baseline had a 79% reduced rate of significant deterioration or need for hospital-based intervention. The other four factors were linked with increased rates of deterioration and need for intervention: Right heart strain caused by the PE and identified by an echocardiogram boosted the risk of a bad outcome more than fourfold, coronary disease and cerebrovascular disease each were tied to a more than threefold increased rate, and residual deep vein thrombosis was linked with a more than doubled rate of bad outcomes.
The subset of patients with the most severe outcomes had only one direct correlation with bad outcomes, right heart strain on echo. This subset of patients also showed a protective link against bad outcomes when their systolic blood pressure never fell below 90 mm Hg.
In contrast to these factors linked to 5-day outcomes, two different types of patient factors were significantly linked with 30-day mortality: having a malignancy and having chronic lung disease.
"Previously validated clinical prediction rules that looked at outcomes after PE were primarily validated based on 30-day mortality or recurrent PE, and included factors like having cancer, heart failure, or chronic lung disease. But these scores are only able to predict the outcomes we examined with 70% sensitivity," Dr. Kabrhel said. He found this out by running the numbers he collected through three validated scores for predicting PE outcome: the Geneva Prediction Score (Ann. Intern. Med. 2006;144:165-71), the Severity Index (Am. J. Respir. Crit. Care Med. 2005; 172:1041-6), and the Simplified Pulmonary Embolism Severity Index (Arch. Intern. Med. 2010;170:1383-9). "Predictors of all-cause 30-day mortality are different than predictors of short-term outcomes" in PE patients, he said.
"We found that echo is a very good predictor of short-term outcomes, and also abnormal vital signs. The key point is we need to look at outcomes that are relevant to the decisions made" in the emergency department, Dr. Kabrhel said. "Looking at 30-day mortality in patients who are only hospitalized for 3 days doesn’t really inform the decision on who should be in the hospital. I would suggest caution on using [prediction] tools validated against 30-day mortality and recurrent PE to determine what to do acutely. We need better rules to decide which PE patients need hospitalization."
Dr. Kabrhel said he has been a consultant to Diagnostica Stago, and is an officer for LitPulse.
On Twitter @mitchelzoler
AMSTERDAM – Two-thirds of patients who presented to the emergency department of a U.S. tertiary care hospital with an acute pulmonary embolism had no acute deterioration and required no short-term hospital-based interventions, in an analysis of 298 patients seen over a 2-year period.
The finding "supports the assertion that outpatient treatment of patients with pulmonary embolism [PE] is safe," said Dr. Christopher Kabrhel at the 24th Congress of the International Society on Thrombosis and Haemostasis.
"We want to identify patients for whom nothing bad will happen. We showed that two-thirds of patients did well and didn’t need anything from the hospital and didn’t benefit from being in the hospital. We need to identify some of these patients," soon after they present in the emergency department, said Dr. Kabrhel, a surgeon and emergency-medicine physician at Massachusetts General Hospital and Harvard Medical School in Boston. If reliable risk markers can be found with further research, "perhaps we can identify half of the two-thirds—a third of all patients who come to the emergency department with a PE—who we know will be safe with outpatient treatment so we can send those patients home from the emergency department and not admit them."
Most symptomatic U.S. patients who come to an emergency department, and are diagnosed with a PE are immediately admitted to the hospital. In the current study, the hospitalization rate was 92% with a median length of stay of 3 days. "We need a better rule to decide whether a patient needs hospitalization. We need to find which patients benefit from hospitalization," Dr. Kabrhel said in an interview.
He and his associates reviewed 298 adults 18 years or older who presented to the Massachusetts General Hospital emergency department during October 2009 through December 2011 with a radiographically proven PE diagnosed within 24 hours of arrival. They averaged 59 years old, half were women, and the most common comorbidity was malignancy in 107 patients (36%).
The study’s primary outcome was any clinical deterioration or need for hospital-based intervention during the 5 days following presentation at the emergency department, including the need for advanced cardiac life support, the development of a new cardiac dysrhythmia, the development of hypoxia or hypotension, the need for thrombolysis or thrombectomy, recurrent PE, or death. These events occurred in 99 patients (33%); of these, 28 patients (9% of the total group) had "severe" deterioration or required a "major" intervention. Twelve patients (4%) died within 30 days of their initial emergency presentation. The most common acute complication was the need for respiratory support, in about 55 patients, followed by hypotension, in about 34.
A multivariate analysis identified five baseline factors that significantly correlated with the primary outcome. Patients who had normal vital signs at baseline had a 79% reduced rate of significant deterioration or need for hospital-based intervention. The other four factors were linked with increased rates of deterioration and need for intervention: Right heart strain caused by the PE and identified by an echocardiogram boosted the risk of a bad outcome more than fourfold, coronary disease and cerebrovascular disease each were tied to a more than threefold increased rate, and residual deep vein thrombosis was linked with a more than doubled rate of bad outcomes.
The subset of patients with the most severe outcomes had only one direct correlation with bad outcomes, right heart strain on echo. This subset of patients also showed a protective link against bad outcomes when their systolic blood pressure never fell below 90 mm Hg.
In contrast to these factors linked to 5-day outcomes, two different types of patient factors were significantly linked with 30-day mortality: having a malignancy and having chronic lung disease.
"Previously validated clinical prediction rules that looked at outcomes after PE were primarily validated based on 30-day mortality or recurrent PE, and included factors like having cancer, heart failure, or chronic lung disease. But these scores are only able to predict the outcomes we examined with 70% sensitivity," Dr. Kabrhel said. He found this out by running the numbers he collected through three validated scores for predicting PE outcome: the Geneva Prediction Score (Ann. Intern. Med. 2006;144:165-71), the Severity Index (Am. J. Respir. Crit. Care Med. 2005; 172:1041-6), and the Simplified Pulmonary Embolism Severity Index (Arch. Intern. Med. 2010;170:1383-9). "Predictors of all-cause 30-day mortality are different than predictors of short-term outcomes" in PE patients, he said.
"We found that echo is a very good predictor of short-term outcomes, and also abnormal vital signs. The key point is we need to look at outcomes that are relevant to the decisions made" in the emergency department, Dr. Kabrhel said. "Looking at 30-day mortality in patients who are only hospitalized for 3 days doesn’t really inform the decision on who should be in the hospital. I would suggest caution on using [prediction] tools validated against 30-day mortality and recurrent PE to determine what to do acutely. We need better rules to decide which PE patients need hospitalization."
Dr. Kabrhel said he has been a consultant to Diagnostica Stago, and is an officer for LitPulse.
On Twitter @mitchelzoler
AT ISTH 2013
Major finding: Among patients newly diagnosed with acute pulmonary embolism, 67% had no acute complication and didn’t require a hospital-based intervention.
Data source: A review of 298 patients diagnosed in the emergency department with acute pulmonary embolism at one U.S. center during 2009-2011.
Disclosures: Dr. Kabrhel said that he has been a consultant to Diagnostica Stago, and is an officer for LitPulse.
Personalization declared route to improved AF care
Personalized management of atrial fibrillation is on its way, a new, more individualized approach to diagnosing and treating atrial fibrillation that will vary from patient to patient depending on the pathophysiology, clinical risk, and predisposition of each patient.
But while the trajectory toward personalization seems clear based on recent advances in understanding the many patient factors that produce variations in AF presentation and treatment response, the time for fully personalized management is not yet here, concluded a panel of experts who recently gathered to discuss the issue.
"Emerging markers may allow a more personalized management" in terms of selecting optimal strategies for anticoagulation or rhythm control in different patients based on factors including atrial morphology and damage, brain imaging, genetic predispositions, inflammatory state, and markers of coronary strain. But "each of these promising avenues requires validation in the context of existing risk factors in patients," wrote the 72-member task force in their consensus statement published online Aug. 27 (Europace 2013 [doi: 10.1093/europace/eut232]).
In short, a rough outline of the parameters that will be the basis for personalized assessment and management of patients with AF now exists, but specifics on which factors are proven useful and how to integrate this information into improved patient care remain to be determined from results of ongoing studies, concluded the panel, which represented the Fourth Atrial Fibrillation Competence Network/European Heart Rhythm Association (AFNET/EHRA) consensus conference.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease."
"The care of each patient with AF will depend upon how the patient presents, the medical history and treatment, and the presence or absence of an identifiable precipitant. Further refinements of the management plan will be based on ECG, cardiac and brain imaging, and laboratory findings," the panel wrote.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease," said Dr. Paulus Kirchhof, lead author of the consensus task force, in a statement. Personalized AF management "is the most promising way to further improve morbidity and mortality in AF patients," said Dr. Kirchhof, professor and chair of cardiovascular medicine at the University of Birmingham (England).
The panel noted that AF assessment and management already involve some degree of personalization, including assessing each patient’s presentation and need for restoration of sinus rhythm, the need for anticoagulant treatment and the type of treatment, choice of a rate-control agent, and the need for rhythm control by drug treatment or catheter ablation. Further refinement could come from applying other assessment tools. An ECG can help identify concurrent heart disease, silent AF, and the best candidates for catheter ablation. Long-term rhythm monitoring by ECG has the potential to better guide the type of antiarrhythmic therapy each patient receives.
The role of brain imaging by MRI remains poorly defined right now. Brain MRI scans hold promise to "help tailor individual treatment decisions on anticoagulation," but for now the evidence is inadequate. "Large prospective studies using serial MRI measurements in AF patients are needed to support" a role for this approach. "Present knowledge does not support the unselected or serial use of MRI to guide stroke prevention in AF patients," the task force said.
Heart imaging can provide information on valvular disease, left ventricular hypertrophy and dysfunction, and left atrial size and function, factors that can affect stroke risk and rhythm control. Biomarkers in a patient’s blood or urine, together with genetic markers, provide important information on bleeding and thrombosis risks as well as kidney function. Other important blood-based biomarkers relevant to assessing AF risk include natriuretic peptides, inflammatory markers, and unsaturated fatty acids. Genetic markers for familial AF may also provide potentially important risk information.
The task force also called for a new taxonomy for AF based on pathophysiology. The current system for classifying AF is "imperfect because there are overlaps between categories, and the majority of AF patients fall into the ‘unclassified AF’ group," Dr. Kirchhof said. "We need a better understanding of why AF develops in an individual patient before we can classify them based on biomarkers, imaging, or ECG, in addition to clinical parameters, and develop better therapies."
The consensus statement laid out a preliminary classification framework that drew distinctions among different types of AF etiologies that "could provide a step toward a taxonomy." The panel divided AF patients into seven types: monogenic for patients with an inherited cardiomyopathy, focally induced for patients with frequent paroxysmal episodes, postoperative, valvular, elderly for patients who first develop AF at age 80 years or older, polygenic for patients with genetic variants linked to early-onset AF, and unclassified.
This statement will be presented on Sept. 3 in a session on personalized medicine at the annual congress of the European Society of Cardiology in Amsterdam.
Dr. Kirchhof said that he has received consulting fees or honoraria from 16 different drug companies and device manufacturers. His complete disclosure statement as well as disclosures from the other task force members are listed in the statement document.
On Twitter @mitchelzoler
Personalized management of atrial fibrillation is on its way, a new, more individualized approach to diagnosing and treating atrial fibrillation that will vary from patient to patient depending on the pathophysiology, clinical risk, and predisposition of each patient.
But while the trajectory toward personalization seems clear based on recent advances in understanding the many patient factors that produce variations in AF presentation and treatment response, the time for fully personalized management is not yet here, concluded a panel of experts who recently gathered to discuss the issue.
"Emerging markers may allow a more personalized management" in terms of selecting optimal strategies for anticoagulation or rhythm control in different patients based on factors including atrial morphology and damage, brain imaging, genetic predispositions, inflammatory state, and markers of coronary strain. But "each of these promising avenues requires validation in the context of existing risk factors in patients," wrote the 72-member task force in their consensus statement published online Aug. 27 (Europace 2013 [doi: 10.1093/europace/eut232]).
In short, a rough outline of the parameters that will be the basis for personalized assessment and management of patients with AF now exists, but specifics on which factors are proven useful and how to integrate this information into improved patient care remain to be determined from results of ongoing studies, concluded the panel, which represented the Fourth Atrial Fibrillation Competence Network/European Heart Rhythm Association (AFNET/EHRA) consensus conference.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease."
"The care of each patient with AF will depend upon how the patient presents, the medical history and treatment, and the presence or absence of an identifiable precipitant. Further refinements of the management plan will be based on ECG, cardiac and brain imaging, and laboratory findings," the panel wrote.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease," said Dr. Paulus Kirchhof, lead author of the consensus task force, in a statement. Personalized AF management "is the most promising way to further improve morbidity and mortality in AF patients," said Dr. Kirchhof, professor and chair of cardiovascular medicine at the University of Birmingham (England).
The panel noted that AF assessment and management already involve some degree of personalization, including assessing each patient’s presentation and need for restoration of sinus rhythm, the need for anticoagulant treatment and the type of treatment, choice of a rate-control agent, and the need for rhythm control by drug treatment or catheter ablation. Further refinement could come from applying other assessment tools. An ECG can help identify concurrent heart disease, silent AF, and the best candidates for catheter ablation. Long-term rhythm monitoring by ECG has the potential to better guide the type of antiarrhythmic therapy each patient receives.
The role of brain imaging by MRI remains poorly defined right now. Brain MRI scans hold promise to "help tailor individual treatment decisions on anticoagulation," but for now the evidence is inadequate. "Large prospective studies using serial MRI measurements in AF patients are needed to support" a role for this approach. "Present knowledge does not support the unselected or serial use of MRI to guide stroke prevention in AF patients," the task force said.
Heart imaging can provide information on valvular disease, left ventricular hypertrophy and dysfunction, and left atrial size and function, factors that can affect stroke risk and rhythm control. Biomarkers in a patient’s blood or urine, together with genetic markers, provide important information on bleeding and thrombosis risks as well as kidney function. Other important blood-based biomarkers relevant to assessing AF risk include natriuretic peptides, inflammatory markers, and unsaturated fatty acids. Genetic markers for familial AF may also provide potentially important risk information.
The task force also called for a new taxonomy for AF based on pathophysiology. The current system for classifying AF is "imperfect because there are overlaps between categories, and the majority of AF patients fall into the ‘unclassified AF’ group," Dr. Kirchhof said. "We need a better understanding of why AF develops in an individual patient before we can classify them based on biomarkers, imaging, or ECG, in addition to clinical parameters, and develop better therapies."
The consensus statement laid out a preliminary classification framework that drew distinctions among different types of AF etiologies that "could provide a step toward a taxonomy." The panel divided AF patients into seven types: monogenic for patients with an inherited cardiomyopathy, focally induced for patients with frequent paroxysmal episodes, postoperative, valvular, elderly for patients who first develop AF at age 80 years or older, polygenic for patients with genetic variants linked to early-onset AF, and unclassified.
This statement will be presented on Sept. 3 in a session on personalized medicine at the annual congress of the European Society of Cardiology in Amsterdam.
Dr. Kirchhof said that he has received consulting fees or honoraria from 16 different drug companies and device manufacturers. His complete disclosure statement as well as disclosures from the other task force members are listed in the statement document.
On Twitter @mitchelzoler
Personalized management of atrial fibrillation is on its way, a new, more individualized approach to diagnosing and treating atrial fibrillation that will vary from patient to patient depending on the pathophysiology, clinical risk, and predisposition of each patient.
But while the trajectory toward personalization seems clear based on recent advances in understanding the many patient factors that produce variations in AF presentation and treatment response, the time for fully personalized management is not yet here, concluded a panel of experts who recently gathered to discuss the issue.
"Emerging markers may allow a more personalized management" in terms of selecting optimal strategies for anticoagulation or rhythm control in different patients based on factors including atrial morphology and damage, brain imaging, genetic predispositions, inflammatory state, and markers of coronary strain. But "each of these promising avenues requires validation in the context of existing risk factors in patients," wrote the 72-member task force in their consensus statement published online Aug. 27 (Europace 2013 [doi: 10.1093/europace/eut232]).
In short, a rough outline of the parameters that will be the basis for personalized assessment and management of patients with AF now exists, but specifics on which factors are proven useful and how to integrate this information into improved patient care remain to be determined from results of ongoing studies, concluded the panel, which represented the Fourth Atrial Fibrillation Competence Network/European Heart Rhythm Association (AFNET/EHRA) consensus conference.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease."
"The care of each patient with AF will depend upon how the patient presents, the medical history and treatment, and the presence or absence of an identifiable precipitant. Further refinements of the management plan will be based on ECG, cardiac and brain imaging, and laboratory findings," the panel wrote.
"The hope is that within the next few years, we will be able to propose personalized management of AF and thereby reduce the excess mortality associated with the disease," said Dr. Paulus Kirchhof, lead author of the consensus task force, in a statement. Personalized AF management "is the most promising way to further improve morbidity and mortality in AF patients," said Dr. Kirchhof, professor and chair of cardiovascular medicine at the University of Birmingham (England).
The panel noted that AF assessment and management already involve some degree of personalization, including assessing each patient’s presentation and need for restoration of sinus rhythm, the need for anticoagulant treatment and the type of treatment, choice of a rate-control agent, and the need for rhythm control by drug treatment or catheter ablation. Further refinement could come from applying other assessment tools. An ECG can help identify concurrent heart disease, silent AF, and the best candidates for catheter ablation. Long-term rhythm monitoring by ECG has the potential to better guide the type of antiarrhythmic therapy each patient receives.
The role of brain imaging by MRI remains poorly defined right now. Brain MRI scans hold promise to "help tailor individual treatment decisions on anticoagulation," but for now the evidence is inadequate. "Large prospective studies using serial MRI measurements in AF patients are needed to support" a role for this approach. "Present knowledge does not support the unselected or serial use of MRI to guide stroke prevention in AF patients," the task force said.
Heart imaging can provide information on valvular disease, left ventricular hypertrophy and dysfunction, and left atrial size and function, factors that can affect stroke risk and rhythm control. Biomarkers in a patient’s blood or urine, together with genetic markers, provide important information on bleeding and thrombosis risks as well as kidney function. Other important blood-based biomarkers relevant to assessing AF risk include natriuretic peptides, inflammatory markers, and unsaturated fatty acids. Genetic markers for familial AF may also provide potentially important risk information.
The task force also called for a new taxonomy for AF based on pathophysiology. The current system for classifying AF is "imperfect because there are overlaps between categories, and the majority of AF patients fall into the ‘unclassified AF’ group," Dr. Kirchhof said. "We need a better understanding of why AF develops in an individual patient before we can classify them based on biomarkers, imaging, or ECG, in addition to clinical parameters, and develop better therapies."
The consensus statement laid out a preliminary classification framework that drew distinctions among different types of AF etiologies that "could provide a step toward a taxonomy." The panel divided AF patients into seven types: monogenic for patients with an inherited cardiomyopathy, focally induced for patients with frequent paroxysmal episodes, postoperative, valvular, elderly for patients who first develop AF at age 80 years or older, polygenic for patients with genetic variants linked to early-onset AF, and unclassified.
This statement will be presented on Sept. 3 in a session on personalized medicine at the annual congress of the European Society of Cardiology in Amsterdam.
Dr. Kirchhof said that he has received consulting fees or honoraria from 16 different drug companies and device manufacturers. His complete disclosure statement as well as disclosures from the other task force members are listed in the statement document.
On Twitter @mitchelzoler
FROM EUROPACE