User login
Pharyngeal reservoir drives gonorrhea epidemic in gay men
VIENNA – , Colm O’Mahony, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
“We think pharyngeal gonorrhea is now the most important factor in this continuing epidemic,” according to Dr. O’Mahony, professor of medicine and a venereologist at the University of Chester (England).
Recent studies have shown that men can carry Neisseria gonorrhoeae in their throats for weeks or months without symptoms and can readily spread the infection through unprotected oral sex.
Also, surveys of men who have sex with men (MSM) indicate that saliva is commonly used as a lubricant for anal sex. Australian investigators have estimated that about half of rectal gonorrhea in MSM would be eliminated if they stopped using their partner’s saliva for this purpose (Sex Trans Infect. 2016 Mar 3. pii:sextrans-2015-052502. doi: 10.1136/sextrans-2015-052502).
And then there is French kissing.
“Men don’t go out to a nightclub and have indiscriminate anal sex anymore. It’s not like that,” according to Dr. O’Mahony. “But they do kiss quite a lot of other men over the course of an evening, and it’s deep kissing. They may French kiss 15-20 other young men. And we think there’s actually a significant risk of transmission of gonorrhea from this simple deep kissing.”
Indeed, Australian investigators are now conducting a study examining whether having young gay men take a bottle of mouthwash with them when they go clubbing so they can take a good swish in between kissing will protect against N. gonorrhoeae infection.
“Apparently gonorrhea is quite sensitive to mouthwashes like Listerine. So we await those study results with interest,” he continued.
Dr. O’Mahony warned that the problem of multidrug-resistant gonorrhea is further along than most noninfectious disease experts realize. That’s a frightening prospect, given that an estimated 800,000 cases of gonorrhea occur annually in the United States alone. Because of well-documented treatment failures with cefixime and other oral cephalosporins, the Centers for Disease Control and Prevention now recommends only one regimen for the treatment of gonorrhea: dual treatment with a single intramuscular dose of 250 mg of ceftriaxone (Rocephin) plus 1 g of azithromycin in a single dose.
“There have already been some cases of ceftriaxone-resistant gonorrhea reported in Japan, Spain, and other parts of Europe. And we’re now seeing azithromycin-resistant gonorrhea throughout the U.K., which is a problem. So we are really worried that we will end up with untreatable gonorrhea within a couple of years,” Dr. O’Mahony said.
The evolving antimicrobial resistance scenario is reminiscent of the quinolone experience, he added.
“In 1992, I published the first reported case of quinolone-resistant gonorrhea in Liverpool. Five years later we had to stop using quinolones because more than 10% of gonorrhea was resistant,” the venereologist said.
In mid-2016 the CDC published the first-ever comprehensive data from its Gonococcal Isolate Surveillance Project. An analysis of more than 5,000 N. gonorrhoeae isolates obtained from men with gonococcal urethritis presenting at U.S. STD clinics showed that 25.3% of samples were resistant to tetracycline, 19.2% to ciprofloxacin, and 16.2% to penicillin. The prevalence of reduced azithromycin susceptibility jumped from 0.6% in 2013 to 2.5% in 2014. Reduced ceftriaxone susceptibility doubled from 0.4% in 2013 to 0.8% the following year. Antimicrobial susceptibility patterns varied by geographic region, with the highest rates of reduced susceptibility being seen in the Midwest and among MSM (MMWR Surveill Summ. 2016;65[No. SS-7]:1–19. doi: 10.15585/mmwr.ss6507a1).
Dr. O’Mahony reported having no relevant financial conflicts.
VIENNA – , Colm O’Mahony, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
“We think pharyngeal gonorrhea is now the most important factor in this continuing epidemic,” according to Dr. O’Mahony, professor of medicine and a venereologist at the University of Chester (England).
Recent studies have shown that men can carry Neisseria gonorrhoeae in their throats for weeks or months without symptoms and can readily spread the infection through unprotected oral sex.
Also, surveys of men who have sex with men (MSM) indicate that saliva is commonly used as a lubricant for anal sex. Australian investigators have estimated that about half of rectal gonorrhea in MSM would be eliminated if they stopped using their partner’s saliva for this purpose (Sex Trans Infect. 2016 Mar 3. pii:sextrans-2015-052502. doi: 10.1136/sextrans-2015-052502).
And then there is French kissing.
“Men don’t go out to a nightclub and have indiscriminate anal sex anymore. It’s not like that,” according to Dr. O’Mahony. “But they do kiss quite a lot of other men over the course of an evening, and it’s deep kissing. They may French kiss 15-20 other young men. And we think there’s actually a significant risk of transmission of gonorrhea from this simple deep kissing.”
Indeed, Australian investigators are now conducting a study examining whether having young gay men take a bottle of mouthwash with them when they go clubbing so they can take a good swish in between kissing will protect against N. gonorrhoeae infection.
“Apparently gonorrhea is quite sensitive to mouthwashes like Listerine. So we await those study results with interest,” he continued.
Dr. O’Mahony warned that the problem of multidrug-resistant gonorrhea is further along than most noninfectious disease experts realize. That’s a frightening prospect, given that an estimated 800,000 cases of gonorrhea occur annually in the United States alone. Because of well-documented treatment failures with cefixime and other oral cephalosporins, the Centers for Disease Control and Prevention now recommends only one regimen for the treatment of gonorrhea: dual treatment with a single intramuscular dose of 250 mg of ceftriaxone (Rocephin) plus 1 g of azithromycin in a single dose.
“There have already been some cases of ceftriaxone-resistant gonorrhea reported in Japan, Spain, and other parts of Europe. And we’re now seeing azithromycin-resistant gonorrhea throughout the U.K., which is a problem. So we are really worried that we will end up with untreatable gonorrhea within a couple of years,” Dr. O’Mahony said.
The evolving antimicrobial resistance scenario is reminiscent of the quinolone experience, he added.
“In 1992, I published the first reported case of quinolone-resistant gonorrhea in Liverpool. Five years later we had to stop using quinolones because more than 10% of gonorrhea was resistant,” the venereologist said.
In mid-2016 the CDC published the first-ever comprehensive data from its Gonococcal Isolate Surveillance Project. An analysis of more than 5,000 N. gonorrhoeae isolates obtained from men with gonococcal urethritis presenting at U.S. STD clinics showed that 25.3% of samples were resistant to tetracycline, 19.2% to ciprofloxacin, and 16.2% to penicillin. The prevalence of reduced azithromycin susceptibility jumped from 0.6% in 2013 to 2.5% in 2014. Reduced ceftriaxone susceptibility doubled from 0.4% in 2013 to 0.8% the following year. Antimicrobial susceptibility patterns varied by geographic region, with the highest rates of reduced susceptibility being seen in the Midwest and among MSM (MMWR Surveill Summ. 2016;65[No. SS-7]:1–19. doi: 10.15585/mmwr.ss6507a1).
Dr. O’Mahony reported having no relevant financial conflicts.
VIENNA – , Colm O’Mahony, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
“We think pharyngeal gonorrhea is now the most important factor in this continuing epidemic,” according to Dr. O’Mahony, professor of medicine and a venereologist at the University of Chester (England).
Recent studies have shown that men can carry Neisseria gonorrhoeae in their throats for weeks or months without symptoms and can readily spread the infection through unprotected oral sex.
Also, surveys of men who have sex with men (MSM) indicate that saliva is commonly used as a lubricant for anal sex. Australian investigators have estimated that about half of rectal gonorrhea in MSM would be eliminated if they stopped using their partner’s saliva for this purpose (Sex Trans Infect. 2016 Mar 3. pii:sextrans-2015-052502. doi: 10.1136/sextrans-2015-052502).
And then there is French kissing.
“Men don’t go out to a nightclub and have indiscriminate anal sex anymore. It’s not like that,” according to Dr. O’Mahony. “But they do kiss quite a lot of other men over the course of an evening, and it’s deep kissing. They may French kiss 15-20 other young men. And we think there’s actually a significant risk of transmission of gonorrhea from this simple deep kissing.”
Indeed, Australian investigators are now conducting a study examining whether having young gay men take a bottle of mouthwash with them when they go clubbing so they can take a good swish in between kissing will protect against N. gonorrhoeae infection.
“Apparently gonorrhea is quite sensitive to mouthwashes like Listerine. So we await those study results with interest,” he continued.
Dr. O’Mahony warned that the problem of multidrug-resistant gonorrhea is further along than most noninfectious disease experts realize. That’s a frightening prospect, given that an estimated 800,000 cases of gonorrhea occur annually in the United States alone. Because of well-documented treatment failures with cefixime and other oral cephalosporins, the Centers for Disease Control and Prevention now recommends only one regimen for the treatment of gonorrhea: dual treatment with a single intramuscular dose of 250 mg of ceftriaxone (Rocephin) plus 1 g of azithromycin in a single dose.
“There have already been some cases of ceftriaxone-resistant gonorrhea reported in Japan, Spain, and other parts of Europe. And we’re now seeing azithromycin-resistant gonorrhea throughout the U.K., which is a problem. So we are really worried that we will end up with untreatable gonorrhea within a couple of years,” Dr. O’Mahony said.
The evolving antimicrobial resistance scenario is reminiscent of the quinolone experience, he added.
“In 1992, I published the first reported case of quinolone-resistant gonorrhea in Liverpool. Five years later we had to stop using quinolones because more than 10% of gonorrhea was resistant,” the venereologist said.
In mid-2016 the CDC published the first-ever comprehensive data from its Gonococcal Isolate Surveillance Project. An analysis of more than 5,000 N. gonorrhoeae isolates obtained from men with gonococcal urethritis presenting at U.S. STD clinics showed that 25.3% of samples were resistant to tetracycline, 19.2% to ciprofloxacin, and 16.2% to penicillin. The prevalence of reduced azithromycin susceptibility jumped from 0.6% in 2013 to 2.5% in 2014. Reduced ceftriaxone susceptibility doubled from 0.4% in 2013 to 0.8% the following year. Antimicrobial susceptibility patterns varied by geographic region, with the highest rates of reduced susceptibility being seen in the Midwest and among MSM (MMWR Surveill Summ. 2016;65[No. SS-7]:1–19. doi: 10.15585/mmwr.ss6507a1).
Dr. O’Mahony reported having no relevant financial conflicts.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Centers of excellence program raises quality bar in lung cancer management
VIENNA – A grassroots, patient-centric program aimed at encouraging U.S. community hospitals in underserved areas to adopt a comprehensive centers-of-excellence model for treatment of lung cancer has demonstrated that community cancer centers can achieve quality of care comparable to that found in academic medical centers, Raymond Osarogiagbon, MD, declared at the World Congress on Lung Cancer.
One major reason why aggregate lung cancer survival in the United States has barely inched upward during the past 3 decades is there is often a huge gap between the quality of care patients get at academic research centers – including access to clinical trials – and what they can get in community hospitals. Eighty percent of lung cancer patients receive their care in these community cancer centers, where not all physicians and surgeons may be up to speed with guideline-recommended best practices, observed Dr. Osarogiagbon, a medical oncologist and director of the multidisciplinary thoracic oncology program at Baptist Cancer Center in Memphis.
The centers of excellence program, supported by the Bonnie J. Addario Lung Cancer Foundation, is an attempt to address this disparity. In 3 years, 13 hospitals in areas with large underserved patient populations in nine states have qualified. Dr. Osarogiagbon anticipates that the competitive advantage this designation provides will spur more and more community hospitals with cancer centers to get on board.
To qualify, community cancer centers have to commit to following specific best practices as standards of care in accord with guidelines issued by groups including the National Comprehensive Cancer Network and the American Society of Clinical Oncology. Requirements include the use of multidisciplinary teams for treatment decisions, routine use of molecular diagnostics and targeted therapies, patient access to clinical trials, longitudinal institutional data tracking, patient and caregiver education programs, and minimally invasive surgical and staging techniques. A lung cancer screening program is required. So is a systematic program for management of all incidentally detected lung nodules, many of which today fall between the cracks.
In an interview, Dr. Osarogiagbon shared several examples of how achieving the foundation’s center of excellence designation enables community cancer centers to achieve top-quality care on a par with academic medical centers. At Memorial Cancer Institute in Hollywood, Fla., which serves large Hispanic and black populations, implementation of a lung cancer screening program and other measures has resulted in 40% of patients with lung cancer being diagnosed with stage 1 or 2 disease amenable to curative surgery. The overall U.S. rate is significantly lower at 29%.
And in an area composed of western Tennessee, northern Mississippi, and eastern Arkansas, Dr. Osarogiagbon and coinvestigators at the Baptist Cancer Center conducted a study demonstrating that the use of two complementary surgical staging interventions resulted in improved rates of guideline-recommended surgical staging quality.
The observational study, presented elsewhere at the world congress by Nicholas Faris, MD, of the Thoracic Oncology Research Group, Baptist Cancer Center, Memphis, entailed analysis of curative-intent resections in 2,094 patients with non–small-cell lung cancer during 2004-2016. A novel anatomically sound gross dissection protocol was provided to assist pathologists in retrieving the intrapulmonary lymph nodes required for staging in 161 patients undergoing curative resection. A special lymph node specimen collection kit was utilized in 152. Another 289 resections utilized both interventions. And 1,492 patients received neither intervention.
Use of the interventions was associated with higher rates of adherence to various professional organizations’ guidelines for high-quality surgical staging. For example, the American College of Surgeons Commission on Cancer recommends that at least 10 lymph nodes be examined in patients with stage 1a-2b NSCLC. This was achieved in 71% of patients who had both interventions, 56% of those where the lymph node specimen collection kit was utilized but not the pathology intervention, 48% of patients where the pathology intervention but not the kit was employed, and in only 25% of patients where neither was used.
Similarly, more than three mediastinal lymph node stations were sampled in accord with National Comprehensive Cancer Network guidelines in 96% of patients with both interventions, 90% with the lymph node collection kit, 54% with the pathology intervention, and only 44% with neither, Dr. Osarogiagbon said in the interview.
Dr. Osarogiagbon reported serving as a consultant to Eli Lilly, Genentech, and the Association of Community Cancer Centers.
VIENNA – A grassroots, patient-centric program aimed at encouraging U.S. community hospitals in underserved areas to adopt a comprehensive centers-of-excellence model for treatment of lung cancer has demonstrated that community cancer centers can achieve quality of care comparable to that found in academic medical centers, Raymond Osarogiagbon, MD, declared at the World Congress on Lung Cancer.
One major reason why aggregate lung cancer survival in the United States has barely inched upward during the past 3 decades is there is often a huge gap between the quality of care patients get at academic research centers – including access to clinical trials – and what they can get in community hospitals. Eighty percent of lung cancer patients receive their care in these community cancer centers, where not all physicians and surgeons may be up to speed with guideline-recommended best practices, observed Dr. Osarogiagbon, a medical oncologist and director of the multidisciplinary thoracic oncology program at Baptist Cancer Center in Memphis.
The centers of excellence program, supported by the Bonnie J. Addario Lung Cancer Foundation, is an attempt to address this disparity. In 3 years, 13 hospitals in areas with large underserved patient populations in nine states have qualified. Dr. Osarogiagbon anticipates that the competitive advantage this designation provides will spur more and more community hospitals with cancer centers to get on board.
To qualify, community cancer centers have to commit to following specific best practices as standards of care in accord with guidelines issued by groups including the National Comprehensive Cancer Network and the American Society of Clinical Oncology. Requirements include the use of multidisciplinary teams for treatment decisions, routine use of molecular diagnostics and targeted therapies, patient access to clinical trials, longitudinal institutional data tracking, patient and caregiver education programs, and minimally invasive surgical and staging techniques. A lung cancer screening program is required. So is a systematic program for management of all incidentally detected lung nodules, many of which today fall between the cracks.
In an interview, Dr. Osarogiagbon shared several examples of how achieving the foundation’s center of excellence designation enables community cancer centers to achieve top-quality care on a par with academic medical centers. At Memorial Cancer Institute in Hollywood, Fla., which serves large Hispanic and black populations, implementation of a lung cancer screening program and other measures has resulted in 40% of patients with lung cancer being diagnosed with stage 1 or 2 disease amenable to curative surgery. The overall U.S. rate is significantly lower at 29%.
And in an area composed of western Tennessee, northern Mississippi, and eastern Arkansas, Dr. Osarogiagbon and coinvestigators at the Baptist Cancer Center conducted a study demonstrating that the use of two complementary surgical staging interventions resulted in improved rates of guideline-recommended surgical staging quality.
The observational study, presented elsewhere at the world congress by Nicholas Faris, MD, of the Thoracic Oncology Research Group, Baptist Cancer Center, Memphis, entailed analysis of curative-intent resections in 2,094 patients with non–small-cell lung cancer during 2004-2016. A novel anatomically sound gross dissection protocol was provided to assist pathologists in retrieving the intrapulmonary lymph nodes required for staging in 161 patients undergoing curative resection. A special lymph node specimen collection kit was utilized in 152. Another 289 resections utilized both interventions. And 1,492 patients received neither intervention.
Use of the interventions was associated with higher rates of adherence to various professional organizations’ guidelines for high-quality surgical staging. For example, the American College of Surgeons Commission on Cancer recommends that at least 10 lymph nodes be examined in patients with stage 1a-2b NSCLC. This was achieved in 71% of patients who had both interventions, 56% of those where the lymph node specimen collection kit was utilized but not the pathology intervention, 48% of patients where the pathology intervention but not the kit was employed, and in only 25% of patients where neither was used.
Similarly, more than three mediastinal lymph node stations were sampled in accord with National Comprehensive Cancer Network guidelines in 96% of patients with both interventions, 90% with the lymph node collection kit, 54% with the pathology intervention, and only 44% with neither, Dr. Osarogiagbon said in the interview.
Dr. Osarogiagbon reported serving as a consultant to Eli Lilly, Genentech, and the Association of Community Cancer Centers.
VIENNA – A grassroots, patient-centric program aimed at encouraging U.S. community hospitals in underserved areas to adopt a comprehensive centers-of-excellence model for treatment of lung cancer has demonstrated that community cancer centers can achieve quality of care comparable to that found in academic medical centers, Raymond Osarogiagbon, MD, declared at the World Congress on Lung Cancer.
One major reason why aggregate lung cancer survival in the United States has barely inched upward during the past 3 decades is there is often a huge gap between the quality of care patients get at academic research centers – including access to clinical trials – and what they can get in community hospitals. Eighty percent of lung cancer patients receive their care in these community cancer centers, where not all physicians and surgeons may be up to speed with guideline-recommended best practices, observed Dr. Osarogiagbon, a medical oncologist and director of the multidisciplinary thoracic oncology program at Baptist Cancer Center in Memphis.
The centers of excellence program, supported by the Bonnie J. Addario Lung Cancer Foundation, is an attempt to address this disparity. In 3 years, 13 hospitals in areas with large underserved patient populations in nine states have qualified. Dr. Osarogiagbon anticipates that the competitive advantage this designation provides will spur more and more community hospitals with cancer centers to get on board.
To qualify, community cancer centers have to commit to following specific best practices as standards of care in accord with guidelines issued by groups including the National Comprehensive Cancer Network and the American Society of Clinical Oncology. Requirements include the use of multidisciplinary teams for treatment decisions, routine use of molecular diagnostics and targeted therapies, patient access to clinical trials, longitudinal institutional data tracking, patient and caregiver education programs, and minimally invasive surgical and staging techniques. A lung cancer screening program is required. So is a systematic program for management of all incidentally detected lung nodules, many of which today fall between the cracks.
In an interview, Dr. Osarogiagbon shared several examples of how achieving the foundation’s center of excellence designation enables community cancer centers to achieve top-quality care on a par with academic medical centers. At Memorial Cancer Institute in Hollywood, Fla., which serves large Hispanic and black populations, implementation of a lung cancer screening program and other measures has resulted in 40% of patients with lung cancer being diagnosed with stage 1 or 2 disease amenable to curative surgery. The overall U.S. rate is significantly lower at 29%.
And in an area composed of western Tennessee, northern Mississippi, and eastern Arkansas, Dr. Osarogiagbon and coinvestigators at the Baptist Cancer Center conducted a study demonstrating that the use of two complementary surgical staging interventions resulted in improved rates of guideline-recommended surgical staging quality.
The observational study, presented elsewhere at the world congress by Nicholas Faris, MD, of the Thoracic Oncology Research Group, Baptist Cancer Center, Memphis, entailed analysis of curative-intent resections in 2,094 patients with non–small-cell lung cancer during 2004-2016. A novel anatomically sound gross dissection protocol was provided to assist pathologists in retrieving the intrapulmonary lymph nodes required for staging in 161 patients undergoing curative resection. A special lymph node specimen collection kit was utilized in 152. Another 289 resections utilized both interventions. And 1,492 patients received neither intervention.
Use of the interventions was associated with higher rates of adherence to various professional organizations’ guidelines for high-quality surgical staging. For example, the American College of Surgeons Commission on Cancer recommends that at least 10 lymph nodes be examined in patients with stage 1a-2b NSCLC. This was achieved in 71% of patients who had both interventions, 56% of those where the lymph node specimen collection kit was utilized but not the pathology intervention, 48% of patients where the pathology intervention but not the kit was employed, and in only 25% of patients where neither was used.
Similarly, more than three mediastinal lymph node stations were sampled in accord with National Comprehensive Cancer Network guidelines in 96% of patients with both interventions, 90% with the lymph node collection kit, 54% with the pathology intervention, and only 44% with neither, Dr. Osarogiagbon said in the interview.
Dr. Osarogiagbon reported serving as a consultant to Eli Lilly, Genentech, and the Association of Community Cancer Centers.
EXPERT ANALYSIS FROM WCLC 2016
Screening tool spots teens headed for substance-dependent adulthood
VIENNA – The creation of a simple risk score that accurately predicts which adolescents in the general population will develop persistent substance dependence as adults has been one of the highlights of the year in addiction medicine, Wim van den Brink, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“These predictors are not very difficult to assess. Clinicians will be interested to know that the positive predictive value of the screen is threefold greater than the persistent prevalence rate,” noted Dr. van den Brink, professor of psychiatry and addiction at the University of Amsterdam and director of the Amsterdam Institute for Addiction Research.
The New Zealand researchers developed what they call “a universal screening tool” by working backward in an analysis of a representative group of 1,037 individuals born in Dunedin, New Zealand, in 1972-1973 and prospectively followed to age 38 years, with a 95% study retention rate. Along the way, participants were assessed for dependence on alcohol, tobacco, cannabis, or hard drugs at ages 21, 26, 32, and 38.
Persistent substance dependence in adulthood, defined as dependence at a minimum of three of the assessments, was present in 19% of subjects.
The investigators found that the presence in childhood or adolescence of any four of nine risk factors had an area under the curve of 80% for persistent substance dependence as an adult. The sensitivity was 43%, with a 93% specificity. The positive predictive value was 60%, and the negative predictive value was 87% (Psychol Med. 2016 Mar;46[4]:877-89).
The nine risk factors are low family socioeconomic status, a family history of substance dependence, childhood depression, childhood conduct disorder, early exposure to substances, adolescent frequent alcohol use, adolescent frequent cannabis use, male gender, and adolescent frequent tobacco use.
The single least potent predictor was low family socioeconomic status, with an associated 1.73-fold increased risk. The strongest predictors were adolescent frequent tobacco use, which conferred a 5.41-fold increased risk; adolescent frequent cannabis use, with a 4.25-fold risk; and childhood conduct disorder, with a 3.2-fold increased risk.
The investigators also analyzed the screening tool’s performance in predicting a modified outcome consisting of adult persistent dependence on any of the target substances except for tobacco. The predictive power of having any four of the risk factors was similar to that found in the main analysis; however, the two strongest predictors now became adolescent frequent cannabis use, with a 9.5-fold increased risk, and childhood conduct disorder, with a relative risk of 5.42.
Regarding childhood conduct disorder as a risk factor, Dr. van den Brink said, “If you are a child with conduct disorder, your chances of becoming substance dependent in coming years is more than fivefold greater than in a child without conduct disorder.”
This raises the question of whether effective treatment of childhood conduct disorder might prevent later development of persistent substance dependence in adulthood. The answer remains unknown. Although there is no approved drug therapy for conduct disorder, methylphenidate is widely prescribed, especially in young patients with comorbid attention-deficit/hyperactivity disorder.
Several years ago a meta-analysis of 15 longitudinal studies with more than 2,500 participants concluded that stimulant therapy of childhood ADHD neither increased nor reduced the risk of subsequent substance use disorders (JAMA Psychiatry. 2013 Jul;70[7]:740-9). Prescribing physicians were happy to hear they weren’t causing iatrogenic injury, but Dr. van den Brink said he was never comfortable with the investigators’ conclusion.
“There was a lot of heterogeneity in the data, so the overall conclusion might not be the best conclusion,” he said.
He said has become more convinced of that than ever as a result of a recent randomized, double-blind, placebo-controlled MRI study of cerebral blood flow in response to methylphenidate in stimulant-naive patients with childhood or adult AHDH. The investigators found that MRIs obtained 1 week after the conclusion of 16 weeks of methylphenidate therapy showed increased blood flow in the strial and thalamic areas in the pediatric ADHD patients but not in the adults with ADHD (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
This is evidence of an age-dependent sustained effect of methylphenidate therapy on dopamine striatal-thalamic circuitry in children that’s not related to the drug’s clinical effects, which were gone after a week off therapy. The question is, Does this effect represent neurotoxicity, or is it an expression of enhanced brain maturation? Dr. van den Brink said he suspects it’s the latter but cannot exclude the former possibility.
VIENNA – The creation of a simple risk score that accurately predicts which adolescents in the general population will develop persistent substance dependence as adults has been one of the highlights of the year in addiction medicine, Wim van den Brink, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“These predictors are not very difficult to assess. Clinicians will be interested to know that the positive predictive value of the screen is threefold greater than the persistent prevalence rate,” noted Dr. van den Brink, professor of psychiatry and addiction at the University of Amsterdam and director of the Amsterdam Institute for Addiction Research.
The New Zealand researchers developed what they call “a universal screening tool” by working backward in an analysis of a representative group of 1,037 individuals born in Dunedin, New Zealand, in 1972-1973 and prospectively followed to age 38 years, with a 95% study retention rate. Along the way, participants were assessed for dependence on alcohol, tobacco, cannabis, or hard drugs at ages 21, 26, 32, and 38.
Persistent substance dependence in adulthood, defined as dependence at a minimum of three of the assessments, was present in 19% of subjects.
The investigators found that the presence in childhood or adolescence of any four of nine risk factors had an area under the curve of 80% for persistent substance dependence as an adult. The sensitivity was 43%, with a 93% specificity. The positive predictive value was 60%, and the negative predictive value was 87% (Psychol Med. 2016 Mar;46[4]:877-89).
The nine risk factors are low family socioeconomic status, a family history of substance dependence, childhood depression, childhood conduct disorder, early exposure to substances, adolescent frequent alcohol use, adolescent frequent cannabis use, male gender, and adolescent frequent tobacco use.
The single least potent predictor was low family socioeconomic status, with an associated 1.73-fold increased risk. The strongest predictors were adolescent frequent tobacco use, which conferred a 5.41-fold increased risk; adolescent frequent cannabis use, with a 4.25-fold risk; and childhood conduct disorder, with a 3.2-fold increased risk.
The investigators also analyzed the screening tool’s performance in predicting a modified outcome consisting of adult persistent dependence on any of the target substances except for tobacco. The predictive power of having any four of the risk factors was similar to that found in the main analysis; however, the two strongest predictors now became adolescent frequent cannabis use, with a 9.5-fold increased risk, and childhood conduct disorder, with a relative risk of 5.42.
Regarding childhood conduct disorder as a risk factor, Dr. van den Brink said, “If you are a child with conduct disorder, your chances of becoming substance dependent in coming years is more than fivefold greater than in a child without conduct disorder.”
This raises the question of whether effective treatment of childhood conduct disorder might prevent later development of persistent substance dependence in adulthood. The answer remains unknown. Although there is no approved drug therapy for conduct disorder, methylphenidate is widely prescribed, especially in young patients with comorbid attention-deficit/hyperactivity disorder.
Several years ago a meta-analysis of 15 longitudinal studies with more than 2,500 participants concluded that stimulant therapy of childhood ADHD neither increased nor reduced the risk of subsequent substance use disorders (JAMA Psychiatry. 2013 Jul;70[7]:740-9). Prescribing physicians were happy to hear they weren’t causing iatrogenic injury, but Dr. van den Brink said he was never comfortable with the investigators’ conclusion.
“There was a lot of heterogeneity in the data, so the overall conclusion might not be the best conclusion,” he said.
He said has become more convinced of that than ever as a result of a recent randomized, double-blind, placebo-controlled MRI study of cerebral blood flow in response to methylphenidate in stimulant-naive patients with childhood or adult AHDH. The investigators found that MRIs obtained 1 week after the conclusion of 16 weeks of methylphenidate therapy showed increased blood flow in the strial and thalamic areas in the pediatric ADHD patients but not in the adults with ADHD (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
This is evidence of an age-dependent sustained effect of methylphenidate therapy on dopamine striatal-thalamic circuitry in children that’s not related to the drug’s clinical effects, which were gone after a week off therapy. The question is, Does this effect represent neurotoxicity, or is it an expression of enhanced brain maturation? Dr. van den Brink said he suspects it’s the latter but cannot exclude the former possibility.
VIENNA – The creation of a simple risk score that accurately predicts which adolescents in the general population will develop persistent substance dependence as adults has been one of the highlights of the year in addiction medicine, Wim van den Brink, MD, said at the annual congress of the European College of Neuropsychopharmacology.
“These predictors are not very difficult to assess. Clinicians will be interested to know that the positive predictive value of the screen is threefold greater than the persistent prevalence rate,” noted Dr. van den Brink, professor of psychiatry and addiction at the University of Amsterdam and director of the Amsterdam Institute for Addiction Research.
The New Zealand researchers developed what they call “a universal screening tool” by working backward in an analysis of a representative group of 1,037 individuals born in Dunedin, New Zealand, in 1972-1973 and prospectively followed to age 38 years, with a 95% study retention rate. Along the way, participants were assessed for dependence on alcohol, tobacco, cannabis, or hard drugs at ages 21, 26, 32, and 38.
Persistent substance dependence in adulthood, defined as dependence at a minimum of three of the assessments, was present in 19% of subjects.
The investigators found that the presence in childhood or adolescence of any four of nine risk factors had an area under the curve of 80% for persistent substance dependence as an adult. The sensitivity was 43%, with a 93% specificity. The positive predictive value was 60%, and the negative predictive value was 87% (Psychol Med. 2016 Mar;46[4]:877-89).
The nine risk factors are low family socioeconomic status, a family history of substance dependence, childhood depression, childhood conduct disorder, early exposure to substances, adolescent frequent alcohol use, adolescent frequent cannabis use, male gender, and adolescent frequent tobacco use.
The single least potent predictor was low family socioeconomic status, with an associated 1.73-fold increased risk. The strongest predictors were adolescent frequent tobacco use, which conferred a 5.41-fold increased risk; adolescent frequent cannabis use, with a 4.25-fold risk; and childhood conduct disorder, with a 3.2-fold increased risk.
The investigators also analyzed the screening tool’s performance in predicting a modified outcome consisting of adult persistent dependence on any of the target substances except for tobacco. The predictive power of having any four of the risk factors was similar to that found in the main analysis; however, the two strongest predictors now became adolescent frequent cannabis use, with a 9.5-fold increased risk, and childhood conduct disorder, with a relative risk of 5.42.
Regarding childhood conduct disorder as a risk factor, Dr. van den Brink said, “If you are a child with conduct disorder, your chances of becoming substance dependent in coming years is more than fivefold greater than in a child without conduct disorder.”
This raises the question of whether effective treatment of childhood conduct disorder might prevent later development of persistent substance dependence in adulthood. The answer remains unknown. Although there is no approved drug therapy for conduct disorder, methylphenidate is widely prescribed, especially in young patients with comorbid attention-deficit/hyperactivity disorder.
Several years ago a meta-analysis of 15 longitudinal studies with more than 2,500 participants concluded that stimulant therapy of childhood ADHD neither increased nor reduced the risk of subsequent substance use disorders (JAMA Psychiatry. 2013 Jul;70[7]:740-9). Prescribing physicians were happy to hear they weren’t causing iatrogenic injury, but Dr. van den Brink said he was never comfortable with the investigators’ conclusion.
“There was a lot of heterogeneity in the data, so the overall conclusion might not be the best conclusion,” he said.
He said has become more convinced of that than ever as a result of a recent randomized, double-blind, placebo-controlled MRI study of cerebral blood flow in response to methylphenidate in stimulant-naive patients with childhood or adult AHDH. The investigators found that MRIs obtained 1 week after the conclusion of 16 weeks of methylphenidate therapy showed increased blood flow in the strial and thalamic areas in the pediatric ADHD patients but not in the adults with ADHD (JAMA Psychiatry. 2016 Sep 1;73[9]:955-62).
This is evidence of an age-dependent sustained effect of methylphenidate therapy on dopamine striatal-thalamic circuitry in children that’s not related to the drug’s clinical effects, which were gone after a week off therapy. The question is, Does this effect represent neurotoxicity, or is it an expression of enhanced brain maturation? Dr. van den Brink said he suspects it’s the latter but cannot exclude the former possibility.
EXPERT ANALYSIS FROM THE ECNP CONGRESS
Novel oral antifungal headed to phase III for onychomycosis
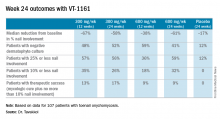
VIENNA – A once-weekly oral antifungal drug known as VT-1161 will move into phase III clinical testing in 2017 based on its impressive performance in an interim analysis of a phase IIb study, Amir Tavakkol, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We saw robust evidence of clinical and mycologic efficacy in patients with moderate to severe onychomycosis. This was true even in patients with nails considered very difficult to treat because of dermatophytomas and spikes, which are usually poor prognostic elements,” said Dr. Tavakkol, chief development officer at Viamet Pharmaceuticals in Durham, N.C., which is developing VT-1161.
He reported on 107 patients with toenail onychomycosis who enrolled in the phase IIb, double-blind, placebo-controlled RENOVATE (Restoring Nail: An Oral VT-1161 Tablet Evaluation) study. At enrollment they averaged 47% disease involvement of the big toenail, the target nail for the trial. Participants in the five study arms had an average of 4.2-5.0 affected toenails. Both the percentage of nail involvement and the number of diseased toenails were roughly twice as great as is typical in studies of topical antifungals, underscoring that participants in the VT-1161 trial had fairly severe onychomycosis.
Patients were assigned to one of five study arms: 300 mg of VT-1161 once weekly for 12 weeks, then 12 weeks of placebo; 600 mg of VT-1161 once weekly for 12 weeks, followed by 12 weeks of placebo; either 300 or 600 mg of the antifungal agent once weekly for the full 24 weeks; or 24 weeks of once-per-week placebo. Immediately prior to the formal start of the study, however, everyone received 14 days of a once-daily loading dose of VT-1161 or placebo at the dose they would subsequently take once weekly during the trial.
The primary outcome in the ongoing study is the percentage of patients with a complete cure, both mycologic and clinical, at 48 weeks. Those data aren’t in yet, but Dr. Tavakkol presented the results of the prespecified interim analysis at 24 weeks.
“Please keep in mind that this is only at most 24 weeks of therapy. Given the rate of nail growth at 1 mm per month when it is healthy, these are remarkable data. Ten percent or less nail involvement is basically 1-2 mm at the distal end. I believe a substantial percentage of these patients will reach clinical cure by 48 weeks,” he said.
VT-1161, a molecule with high selectivity for fungal CYP51, blocks the production of ergosterol, a key component of the fungal cell membrane, according to the company. It has no known drug interactions. At all doses studied in the trial, it was safe and well tolerated, with no abnormal liver function tests, no effect on bilirubin, and no change in QTc interval. Only 8 of the 107 patients reported adverse events deemed possibly related to the study drug by blinded investigators. No one dropped out of the study.
“VT-1161 is also being developed for recurrent vulvovaginal candidiasis. The results there are outstanding, too,” Dr. Tavakkol said.
The trial was funded by Viamet, where he is employed.
VIENNA – A once-weekly oral antifungal drug known as VT-1161 will move into phase III clinical testing in 2017 based on its impressive performance in an interim analysis of a phase IIb study, Amir Tavakkol, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We saw robust evidence of clinical and mycologic efficacy in patients with moderate to severe onychomycosis. This was true even in patients with nails considered very difficult to treat because of dermatophytomas and spikes, which are usually poor prognostic elements,” said Dr. Tavakkol, chief development officer at Viamet Pharmaceuticals in Durham, N.C., which is developing VT-1161.
He reported on 107 patients with toenail onychomycosis who enrolled in the phase IIb, double-blind, placebo-controlled RENOVATE (Restoring Nail: An Oral VT-1161 Tablet Evaluation) study. At enrollment they averaged 47% disease involvement of the big toenail, the target nail for the trial. Participants in the five study arms had an average of 4.2-5.0 affected toenails. Both the percentage of nail involvement and the number of diseased toenails were roughly twice as great as is typical in studies of topical antifungals, underscoring that participants in the VT-1161 trial had fairly severe onychomycosis.
Patients were assigned to one of five study arms: 300 mg of VT-1161 once weekly for 12 weeks, then 12 weeks of placebo; 600 mg of VT-1161 once weekly for 12 weeks, followed by 12 weeks of placebo; either 300 or 600 mg of the antifungal agent once weekly for the full 24 weeks; or 24 weeks of once-per-week placebo. Immediately prior to the formal start of the study, however, everyone received 14 days of a once-daily loading dose of VT-1161 or placebo at the dose they would subsequently take once weekly during the trial.
The primary outcome in the ongoing study is the percentage of patients with a complete cure, both mycologic and clinical, at 48 weeks. Those data aren’t in yet, but Dr. Tavakkol presented the results of the prespecified interim analysis at 24 weeks.
“Please keep in mind that this is only at most 24 weeks of therapy. Given the rate of nail growth at 1 mm per month when it is healthy, these are remarkable data. Ten percent or less nail involvement is basically 1-2 mm at the distal end. I believe a substantial percentage of these patients will reach clinical cure by 48 weeks,” he said.
VT-1161, a molecule with high selectivity for fungal CYP51, blocks the production of ergosterol, a key component of the fungal cell membrane, according to the company. It has no known drug interactions. At all doses studied in the trial, it was safe and well tolerated, with no abnormal liver function tests, no effect on bilirubin, and no change in QTc interval. Only 8 of the 107 patients reported adverse events deemed possibly related to the study drug by blinded investigators. No one dropped out of the study.
“VT-1161 is also being developed for recurrent vulvovaginal candidiasis. The results there are outstanding, too,” Dr. Tavakkol said.
The trial was funded by Viamet, where he is employed.
VIENNA – A once-weekly oral antifungal drug known as VT-1161 will move into phase III clinical testing in 2017 based on its impressive performance in an interim analysis of a phase IIb study, Amir Tavakkol, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“We saw robust evidence of clinical and mycologic efficacy in patients with moderate to severe onychomycosis. This was true even in patients with nails considered very difficult to treat because of dermatophytomas and spikes, which are usually poor prognostic elements,” said Dr. Tavakkol, chief development officer at Viamet Pharmaceuticals in Durham, N.C., which is developing VT-1161.
He reported on 107 patients with toenail onychomycosis who enrolled in the phase IIb, double-blind, placebo-controlled RENOVATE (Restoring Nail: An Oral VT-1161 Tablet Evaluation) study. At enrollment they averaged 47% disease involvement of the big toenail, the target nail for the trial. Participants in the five study arms had an average of 4.2-5.0 affected toenails. Both the percentage of nail involvement and the number of diseased toenails were roughly twice as great as is typical in studies of topical antifungals, underscoring that participants in the VT-1161 trial had fairly severe onychomycosis.
Patients were assigned to one of five study arms: 300 mg of VT-1161 once weekly for 12 weeks, then 12 weeks of placebo; 600 mg of VT-1161 once weekly for 12 weeks, followed by 12 weeks of placebo; either 300 or 600 mg of the antifungal agent once weekly for the full 24 weeks; or 24 weeks of once-per-week placebo. Immediately prior to the formal start of the study, however, everyone received 14 days of a once-daily loading dose of VT-1161 or placebo at the dose they would subsequently take once weekly during the trial.
The primary outcome in the ongoing study is the percentage of patients with a complete cure, both mycologic and clinical, at 48 weeks. Those data aren’t in yet, but Dr. Tavakkol presented the results of the prespecified interim analysis at 24 weeks.
“Please keep in mind that this is only at most 24 weeks of therapy. Given the rate of nail growth at 1 mm per month when it is healthy, these are remarkable data. Ten percent or less nail involvement is basically 1-2 mm at the distal end. I believe a substantial percentage of these patients will reach clinical cure by 48 weeks,” he said.
VT-1161, a molecule with high selectivity for fungal CYP51, blocks the production of ergosterol, a key component of the fungal cell membrane, according to the company. It has no known drug interactions. At all doses studied in the trial, it was safe and well tolerated, with no abnormal liver function tests, no effect on bilirubin, and no change in QTc interval. Only 8 of the 107 patients reported adverse events deemed possibly related to the study drug by blinded investigators. No one dropped out of the study.
“VT-1161 is also being developed for recurrent vulvovaginal candidiasis. The results there are outstanding, too,” Dr. Tavakkol said.
The trial was funded by Viamet, where he is employed.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Among onychomycosis patients with an average 47% target toenail involvement at baseline, 35% had improved to no more than 10% nail involvement at 24 weeks after 12 weeks of once-weekly VT-1161 followed by 12 weeks of placebo.
Data source: A double-blind, randomized, phase IIb clinical trial involving 107 patients with toenail onychomycosis.
Disclosures: The trial was funded by Viamet Pharmaceuticals, where the study presenter is employed.
Adjunctive brexpiprazole improves cognitive function in major depression
VIENNA – Adjunctive brexpiprazole in patients with an inadequate response to antidepressant monotherapy for major depressive disorder resulted in clinically meaningful improvement in multiple aspects of functional impairment, including cognitive dysfunction, in three open-label exploratory studies, Ross A. Baker, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
Clinician-rated depressive symptoms also showed significant improvement once brexpiprazole (Rexulti) was on board, added Dr. Baker of Otsuka Pharmaceutical in Princeton, N.J.
But fatigue and cognitive impairment are another matter. They are now recognized as core aspects of major depression. Even after patients’ depressive symptoms have gone into remission, lingering cognitive dysfunction often impairs performance at work or school, social life, and family/home life. Antidepressants don’t improve this depression-related cognitive impairment. This was the impetus for the three open-label exploratory studies, which addressed these common problems of functional disability and cognitive impairment in different populations with major depressive disorder. The target dose of brexpiprazole in all three studies was 2 mg per day in addition to whatever antidepressant medication patients were already on.
One 12-week study included 47 young patients working at a job or in school. Their mean baseline score on the Sheehan Disability Scale (SDS) was 6.5; a score of 5 or more on the 0-10 scale indicates significant functional impairment in the three domains covered by the SDS: home responsibilities, social life, and work or school performance.
After 12 weeks of adjunctive brexpiprazole, the patients’ mean SDS score had improved by 3.7 points. The improvement was significant across all three domains in this study, as well as in the other two open-label studies.
Patients also demonstrated significant improvement on the Cognitive and Physical Functioning Questionnaire (CPFQ), a self-rated scale that addresses cognitive impairment and executive function. The CPFQ consists of seven items, each scored 1-6, with a higher score indicating worse function in that domain. After 12 weeks on adjunctive brexpiprazole, the young patients experienced a mean 8.1-point improvement from a baseline CPFQ score of 26.1.
Another study included 37 patients with prominent anxiety as a feature of their major depressive disorder. Over the course of 6 weeks of treatment, their mean SDS score improved by 3.6 points from a baseline of 6.5. Also, their CPFQ score fell by 9.9 points from a baseline of 29.3.
The third study involved 61 patients who had switched antidepressants after an inadequate response. After 6 weeks on adjunctive brexpiprazole, their mean SDS score improved from 6.3 to 3.2, and their CPFQ dropped by 9.9 points from a baseline of 29.3 points.
Increased energy and alertness were commonly reported by participants in all three studies.
MADRS scores improved by a mean of 18.1 points from 28.3 points at baseline in the youth study, by 19.8 points from a baseline of 30.1 in the high-anxiety group, and by 17.3 points from a baseline of 29.6 in the study of patients who’d switched antidepressants.
These exploratory studies were sponsored by Otsuka Pharmaceutical. Dr. Baker is a company employee.
Brexpiprazole also is FDA-approved for treatment of schizophrenia.
VIENNA – Adjunctive brexpiprazole in patients with an inadequate response to antidepressant monotherapy for major depressive disorder resulted in clinically meaningful improvement in multiple aspects of functional impairment, including cognitive dysfunction, in three open-label exploratory studies, Ross A. Baker, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
Clinician-rated depressive symptoms also showed significant improvement once brexpiprazole (Rexulti) was on board, added Dr. Baker of Otsuka Pharmaceutical in Princeton, N.J.
But fatigue and cognitive impairment are another matter. They are now recognized as core aspects of major depression. Even after patients’ depressive symptoms have gone into remission, lingering cognitive dysfunction often impairs performance at work or school, social life, and family/home life. Antidepressants don’t improve this depression-related cognitive impairment. This was the impetus for the three open-label exploratory studies, which addressed these common problems of functional disability and cognitive impairment in different populations with major depressive disorder. The target dose of brexpiprazole in all three studies was 2 mg per day in addition to whatever antidepressant medication patients were already on.
One 12-week study included 47 young patients working at a job or in school. Their mean baseline score on the Sheehan Disability Scale (SDS) was 6.5; a score of 5 or more on the 0-10 scale indicates significant functional impairment in the three domains covered by the SDS: home responsibilities, social life, and work or school performance.
After 12 weeks of adjunctive brexpiprazole, the patients’ mean SDS score had improved by 3.7 points. The improvement was significant across all three domains in this study, as well as in the other two open-label studies.
Patients also demonstrated significant improvement on the Cognitive and Physical Functioning Questionnaire (CPFQ), a self-rated scale that addresses cognitive impairment and executive function. The CPFQ consists of seven items, each scored 1-6, with a higher score indicating worse function in that domain. After 12 weeks on adjunctive brexpiprazole, the young patients experienced a mean 8.1-point improvement from a baseline CPFQ score of 26.1.
Another study included 37 patients with prominent anxiety as a feature of their major depressive disorder. Over the course of 6 weeks of treatment, their mean SDS score improved by 3.6 points from a baseline of 6.5. Also, their CPFQ score fell by 9.9 points from a baseline of 29.3.
The third study involved 61 patients who had switched antidepressants after an inadequate response. After 6 weeks on adjunctive brexpiprazole, their mean SDS score improved from 6.3 to 3.2, and their CPFQ dropped by 9.9 points from a baseline of 29.3 points.
Increased energy and alertness were commonly reported by participants in all three studies.
MADRS scores improved by a mean of 18.1 points from 28.3 points at baseline in the youth study, by 19.8 points from a baseline of 30.1 in the high-anxiety group, and by 17.3 points from a baseline of 29.6 in the study of patients who’d switched antidepressants.
These exploratory studies were sponsored by Otsuka Pharmaceutical. Dr. Baker is a company employee.
Brexpiprazole also is FDA-approved for treatment of schizophrenia.
VIENNA – Adjunctive brexpiprazole in patients with an inadequate response to antidepressant monotherapy for major depressive disorder resulted in clinically meaningful improvement in multiple aspects of functional impairment, including cognitive dysfunction, in three open-label exploratory studies, Ross A. Baker, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
Clinician-rated depressive symptoms also showed significant improvement once brexpiprazole (Rexulti) was on board, added Dr. Baker of Otsuka Pharmaceutical in Princeton, N.J.
But fatigue and cognitive impairment are another matter. They are now recognized as core aspects of major depression. Even after patients’ depressive symptoms have gone into remission, lingering cognitive dysfunction often impairs performance at work or school, social life, and family/home life. Antidepressants don’t improve this depression-related cognitive impairment. This was the impetus for the three open-label exploratory studies, which addressed these common problems of functional disability and cognitive impairment in different populations with major depressive disorder. The target dose of brexpiprazole in all three studies was 2 mg per day in addition to whatever antidepressant medication patients were already on.
One 12-week study included 47 young patients working at a job or in school. Their mean baseline score on the Sheehan Disability Scale (SDS) was 6.5; a score of 5 or more on the 0-10 scale indicates significant functional impairment in the three domains covered by the SDS: home responsibilities, social life, and work or school performance.
After 12 weeks of adjunctive brexpiprazole, the patients’ mean SDS score had improved by 3.7 points. The improvement was significant across all three domains in this study, as well as in the other two open-label studies.
Patients also demonstrated significant improvement on the Cognitive and Physical Functioning Questionnaire (CPFQ), a self-rated scale that addresses cognitive impairment and executive function. The CPFQ consists of seven items, each scored 1-6, with a higher score indicating worse function in that domain. After 12 weeks on adjunctive brexpiprazole, the young patients experienced a mean 8.1-point improvement from a baseline CPFQ score of 26.1.
Another study included 37 patients with prominent anxiety as a feature of their major depressive disorder. Over the course of 6 weeks of treatment, their mean SDS score improved by 3.6 points from a baseline of 6.5. Also, their CPFQ score fell by 9.9 points from a baseline of 29.3.
The third study involved 61 patients who had switched antidepressants after an inadequate response. After 6 weeks on adjunctive brexpiprazole, their mean SDS score improved from 6.3 to 3.2, and their CPFQ dropped by 9.9 points from a baseline of 29.3 points.
Increased energy and alertness were commonly reported by participants in all three studies.
MADRS scores improved by a mean of 18.1 points from 28.3 points at baseline in the youth study, by 19.8 points from a baseline of 30.1 in the high-anxiety group, and by 17.3 points from a baseline of 29.6 in the study of patients who’d switched antidepressants.
These exploratory studies were sponsored by Otsuka Pharmaceutical. Dr. Baker is a company employee.
Brexpiprazole also is FDA-approved for treatment of schizophrenia.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Mean scores on the Sheehan Disability Scale improved by 3.1-3.7 points from a baseline of 6.3-6.5 after 6 or 12 weeks of adjunctive brexpiprazole in three studies.
Data source: The three open-label exploratory studies included a total of 145 patients with major depressive disorder.
Disclosures: The studies were sponsored by Otsuka Pharmaceuticals. The presenter is employed by the company.
Mixed findings on iron repletion in HF
NEW ORLEANS – Oral iron supplementation was declared ineffective for the treatment of iron deficiency in patients with heart failure with reduced ejection fraction in one major randomized trial while intravenous ferric carboxymaltose improved exercise capacity and quality of life in another study presented at the American Heart Association scientific sessions.
Iron deficiency is present in roughly half of patients with heart failure with reduced ejection fraction (HFrEF). In affected patients it’s associated with diminished functional capacity and poor quality of life, and is an independent predictor of mortality. Thus, it has emerged as a potential therapeutic target in HFrEF.
The IRONOUT HF (Oral Iron Repletion Effects on Oxygen UpTake in Heart Failure) trial was a multicenter, randomized, double-blind, placebo-controlled clinical trial conducted by the National Heart, Lung, and Blood Institute’s Clinical Heart Failure Network investigators. It included 225 patients with HFrEF and iron deficiency who were randomized to 16 weeks of oral iron polysaccharide at 150 mg b.i.d. or matching placebo.
The primary endpoint was change from baseline to 16 weeks in exercise capacity, as measured via peak oxygen uptake during cardiopulmonary exercise testing. The results were no better in the iron-supplemented group than in placebo-treated controls. Nor was there any benefit for oral iron therapy in terms of quality of life, as assessed by the Kansas City Cardiomyopathy Questionnaire or any of numerous other secondary endpoints, according to Dr. Lewis, head of the heart failure section and director of the cardiopulmonary exercise testing laboratory at Massachusetts General Hospital, Boston.
Participants’ mean baseline hemoglobin level was 12.6 g/dL, but whether patients were anemic at baseline or not made no difference in terms of outcomes. Rates of venous congestion and bleeding were low during the trial.
The problem, the investigators found, was that high-dose oral iron only minimally repleted iron stores. Transferrin saturation increased by a paltry absolute 3% after 16 weeks of twice-daily therapy. Serum ferritin levels increased only 1/20th as much as after intravenous iron ferric carboxymaltose therapy in the earlier positive FAIR-HF (Ferinject Assessment in Patients With Iron Deficiency and Chronic Heart Failure) trial (N Engl J Med. 2009 Dec 17;361:2436-48).
Levels of hepcidin were elevated in study participants. And the higher the level of hepcidin – which Dr. Lewis called “a massive regulator of iron bioavailability and absorption” – the more refractory patients were to oral iron repletion.
“Hepcidin was invented by nature to protect against iron from stimulating growth of bacteria. And when it’s elevated, taking iron orally just has no chance of success,” explained Dr. Anker, professor of cardiology at the University of Gottingen (Germany).
Dr. Anker, who chaired the positive FAIR-HF trial of intravenous iron, said IRONOUT HF was a very well-conducted and definitive clinical trial of oral iron supplementation in HFrEF.
“It’s very simple: Oral iron does not work in patients with chronic heart failure. That’s the take home message. It’s true for peak VO2 [oxygen consumption], for 6-minute walk distance, for symptoms, for quality of life, and even for surrogate markers like NT-proBNP [N-terminal pro b-type natriuretic peptide]. If iron doesn’t get into the body, it’s really difficult to [imagine] that the iron that doesn’t get into the body can exert an effect,” he said.
The primary endpoint in EFFECT-HF was assessor-blinded change in peak VO2 from baseline to week 24. The control group experienced a decrease in peak VO2 over time such that there was a significant difference of 1.0 mL/kg per minute between the two groups.
The ferric carboxymaltose recipients also did significantly better in terms of secondary endpoints including improvement in New York Heart Association functional class, self-reported Patient Global Assessment score, and quality-of-life measures, reported Dr. van Veldhuisen, professor and chairman of cardiology at University Medical Center Groningen (the Netherlands).
Session moderator Clyde Yancy, MD, commented that intravenous iron is not ready for prime time use in clinical practice for several reasons. The open-label EFFECT-HF trial, like the earlier positive double-blind FAIR-HF and CONFIRM-HF IV ferric carboxymaltose trials, was too modest in size to be convincing, especially since this is an expensive and intrusive therapy.
“The endpoint of peak VO2, although a very powerful endpoint, is still one for which there may be some subjectivity, and so we need a more definitive endpoint to be absolutely certain about the potential benefit of the administration of ferric carboxymaltose,” said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University in Chicago.
Discussant Adrian Hernandez, MD, of the Duke Clinical Research Institute in Durham, N.C., said he considers peak VO2 an important endpoint.
“When you talk to patients, exercise capacity is an outcome that matters to them,” he said. “They often comment that what matters to them is living longer with a better quality of life, free of worsening heart failure, and having improvement in everyday functional status. So the cardiopulmonary exercise test is not just a surrogate endpoint; it’s a measure of functional outcome that matters to patients,” he said.
Still, like Dr. Yancy, Dr. Hernandez said he thinks it’s time to have larger, longer, definitive trials with clinical endpoints in order to understand the role of intravenous iron. Both cardiologists applauded Dr. Anker’s announcement that such a trial, known as FAIR-HF2, is now getting started.
The IRONOUT HF trial was funded by the National Institutes of Health. Dr. Lewis reported receiving research support from a handful of pharmaceutical and medical device companies. Dr. van Velduisen reported serving as a scientific adviser to Vifor Pharma, which sponsored the EFFECT-HF trial. Dr. Anker, who was an EFFECT-HF investigator, serves as a consultant to Vifor and several other companies. Dr. Hernandez was an IRONOUT HF investigator and reported receiving research grants from a handful of pharmaceutical companies.
NEW ORLEANS – Oral iron supplementation was declared ineffective for the treatment of iron deficiency in patients with heart failure with reduced ejection fraction in one major randomized trial while intravenous ferric carboxymaltose improved exercise capacity and quality of life in another study presented at the American Heart Association scientific sessions.
Iron deficiency is present in roughly half of patients with heart failure with reduced ejection fraction (HFrEF). In affected patients it’s associated with diminished functional capacity and poor quality of life, and is an independent predictor of mortality. Thus, it has emerged as a potential therapeutic target in HFrEF.
The IRONOUT HF (Oral Iron Repletion Effects on Oxygen UpTake in Heart Failure) trial was a multicenter, randomized, double-blind, placebo-controlled clinical trial conducted by the National Heart, Lung, and Blood Institute’s Clinical Heart Failure Network investigators. It included 225 patients with HFrEF and iron deficiency who were randomized to 16 weeks of oral iron polysaccharide at 150 mg b.i.d. or matching placebo.
The primary endpoint was change from baseline to 16 weeks in exercise capacity, as measured via peak oxygen uptake during cardiopulmonary exercise testing. The results were no better in the iron-supplemented group than in placebo-treated controls. Nor was there any benefit for oral iron therapy in terms of quality of life, as assessed by the Kansas City Cardiomyopathy Questionnaire or any of numerous other secondary endpoints, according to Dr. Lewis, head of the heart failure section and director of the cardiopulmonary exercise testing laboratory at Massachusetts General Hospital, Boston.
Participants’ mean baseline hemoglobin level was 12.6 g/dL, but whether patients were anemic at baseline or not made no difference in terms of outcomes. Rates of venous congestion and bleeding were low during the trial.
The problem, the investigators found, was that high-dose oral iron only minimally repleted iron stores. Transferrin saturation increased by a paltry absolute 3% after 16 weeks of twice-daily therapy. Serum ferritin levels increased only 1/20th as much as after intravenous iron ferric carboxymaltose therapy in the earlier positive FAIR-HF (Ferinject Assessment in Patients With Iron Deficiency and Chronic Heart Failure) trial (N Engl J Med. 2009 Dec 17;361:2436-48).
Levels of hepcidin were elevated in study participants. And the higher the level of hepcidin – which Dr. Lewis called “a massive regulator of iron bioavailability and absorption” – the more refractory patients were to oral iron repletion.
“Hepcidin was invented by nature to protect against iron from stimulating growth of bacteria. And when it’s elevated, taking iron orally just has no chance of success,” explained Dr. Anker, professor of cardiology at the University of Gottingen (Germany).
Dr. Anker, who chaired the positive FAIR-HF trial of intravenous iron, said IRONOUT HF was a very well-conducted and definitive clinical trial of oral iron supplementation in HFrEF.
“It’s very simple: Oral iron does not work in patients with chronic heart failure. That’s the take home message. It’s true for peak VO2 [oxygen consumption], for 6-minute walk distance, for symptoms, for quality of life, and even for surrogate markers like NT-proBNP [N-terminal pro b-type natriuretic peptide]. If iron doesn’t get into the body, it’s really difficult to [imagine] that the iron that doesn’t get into the body can exert an effect,” he said.
The primary endpoint in EFFECT-HF was assessor-blinded change in peak VO2 from baseline to week 24. The control group experienced a decrease in peak VO2 over time such that there was a significant difference of 1.0 mL/kg per minute between the two groups.
The ferric carboxymaltose recipients also did significantly better in terms of secondary endpoints including improvement in New York Heart Association functional class, self-reported Patient Global Assessment score, and quality-of-life measures, reported Dr. van Veldhuisen, professor and chairman of cardiology at University Medical Center Groningen (the Netherlands).
Session moderator Clyde Yancy, MD, commented that intravenous iron is not ready for prime time use in clinical practice for several reasons. The open-label EFFECT-HF trial, like the earlier positive double-blind FAIR-HF and CONFIRM-HF IV ferric carboxymaltose trials, was too modest in size to be convincing, especially since this is an expensive and intrusive therapy.
“The endpoint of peak VO2, although a very powerful endpoint, is still one for which there may be some subjectivity, and so we need a more definitive endpoint to be absolutely certain about the potential benefit of the administration of ferric carboxymaltose,” said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University in Chicago.
Discussant Adrian Hernandez, MD, of the Duke Clinical Research Institute in Durham, N.C., said he considers peak VO2 an important endpoint.
“When you talk to patients, exercise capacity is an outcome that matters to them,” he said. “They often comment that what matters to them is living longer with a better quality of life, free of worsening heart failure, and having improvement in everyday functional status. So the cardiopulmonary exercise test is not just a surrogate endpoint; it’s a measure of functional outcome that matters to patients,” he said.
Still, like Dr. Yancy, Dr. Hernandez said he thinks it’s time to have larger, longer, definitive trials with clinical endpoints in order to understand the role of intravenous iron. Both cardiologists applauded Dr. Anker’s announcement that such a trial, known as FAIR-HF2, is now getting started.
The IRONOUT HF trial was funded by the National Institutes of Health. Dr. Lewis reported receiving research support from a handful of pharmaceutical and medical device companies. Dr. van Velduisen reported serving as a scientific adviser to Vifor Pharma, which sponsored the EFFECT-HF trial. Dr. Anker, who was an EFFECT-HF investigator, serves as a consultant to Vifor and several other companies. Dr. Hernandez was an IRONOUT HF investigator and reported receiving research grants from a handful of pharmaceutical companies.
NEW ORLEANS – Oral iron supplementation was declared ineffective for the treatment of iron deficiency in patients with heart failure with reduced ejection fraction in one major randomized trial while intravenous ferric carboxymaltose improved exercise capacity and quality of life in another study presented at the American Heart Association scientific sessions.
Iron deficiency is present in roughly half of patients with heart failure with reduced ejection fraction (HFrEF). In affected patients it’s associated with diminished functional capacity and poor quality of life, and is an independent predictor of mortality. Thus, it has emerged as a potential therapeutic target in HFrEF.
The IRONOUT HF (Oral Iron Repletion Effects on Oxygen UpTake in Heart Failure) trial was a multicenter, randomized, double-blind, placebo-controlled clinical trial conducted by the National Heart, Lung, and Blood Institute’s Clinical Heart Failure Network investigators. It included 225 patients with HFrEF and iron deficiency who were randomized to 16 weeks of oral iron polysaccharide at 150 mg b.i.d. or matching placebo.
The primary endpoint was change from baseline to 16 weeks in exercise capacity, as measured via peak oxygen uptake during cardiopulmonary exercise testing. The results were no better in the iron-supplemented group than in placebo-treated controls. Nor was there any benefit for oral iron therapy in terms of quality of life, as assessed by the Kansas City Cardiomyopathy Questionnaire or any of numerous other secondary endpoints, according to Dr. Lewis, head of the heart failure section and director of the cardiopulmonary exercise testing laboratory at Massachusetts General Hospital, Boston.
Participants’ mean baseline hemoglobin level was 12.6 g/dL, but whether patients were anemic at baseline or not made no difference in terms of outcomes. Rates of venous congestion and bleeding were low during the trial.
The problem, the investigators found, was that high-dose oral iron only minimally repleted iron stores. Transferrin saturation increased by a paltry absolute 3% after 16 weeks of twice-daily therapy. Serum ferritin levels increased only 1/20th as much as after intravenous iron ferric carboxymaltose therapy in the earlier positive FAIR-HF (Ferinject Assessment in Patients With Iron Deficiency and Chronic Heart Failure) trial (N Engl J Med. 2009 Dec 17;361:2436-48).
Levels of hepcidin were elevated in study participants. And the higher the level of hepcidin – which Dr. Lewis called “a massive regulator of iron bioavailability and absorption” – the more refractory patients were to oral iron repletion.
“Hepcidin was invented by nature to protect against iron from stimulating growth of bacteria. And when it’s elevated, taking iron orally just has no chance of success,” explained Dr. Anker, professor of cardiology at the University of Gottingen (Germany).
Dr. Anker, who chaired the positive FAIR-HF trial of intravenous iron, said IRONOUT HF was a very well-conducted and definitive clinical trial of oral iron supplementation in HFrEF.
“It’s very simple: Oral iron does not work in patients with chronic heart failure. That’s the take home message. It’s true for peak VO2 [oxygen consumption], for 6-minute walk distance, for symptoms, for quality of life, and even for surrogate markers like NT-proBNP [N-terminal pro b-type natriuretic peptide]. If iron doesn’t get into the body, it’s really difficult to [imagine] that the iron that doesn’t get into the body can exert an effect,” he said.
The primary endpoint in EFFECT-HF was assessor-blinded change in peak VO2 from baseline to week 24. The control group experienced a decrease in peak VO2 over time such that there was a significant difference of 1.0 mL/kg per minute between the two groups.
The ferric carboxymaltose recipients also did significantly better in terms of secondary endpoints including improvement in New York Heart Association functional class, self-reported Patient Global Assessment score, and quality-of-life measures, reported Dr. van Veldhuisen, professor and chairman of cardiology at University Medical Center Groningen (the Netherlands).
Session moderator Clyde Yancy, MD, commented that intravenous iron is not ready for prime time use in clinical practice for several reasons. The open-label EFFECT-HF trial, like the earlier positive double-blind FAIR-HF and CONFIRM-HF IV ferric carboxymaltose trials, was too modest in size to be convincing, especially since this is an expensive and intrusive therapy.
“The endpoint of peak VO2, although a very powerful endpoint, is still one for which there may be some subjectivity, and so we need a more definitive endpoint to be absolutely certain about the potential benefit of the administration of ferric carboxymaltose,” said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University in Chicago.
Discussant Adrian Hernandez, MD, of the Duke Clinical Research Institute in Durham, N.C., said he considers peak VO2 an important endpoint.
“When you talk to patients, exercise capacity is an outcome that matters to them,” he said. “They often comment that what matters to them is living longer with a better quality of life, free of worsening heart failure, and having improvement in everyday functional status. So the cardiopulmonary exercise test is not just a surrogate endpoint; it’s a measure of functional outcome that matters to patients,” he said.
Still, like Dr. Yancy, Dr. Hernandez said he thinks it’s time to have larger, longer, definitive trials with clinical endpoints in order to understand the role of intravenous iron. Both cardiologists applauded Dr. Anker’s announcement that such a trial, known as FAIR-HF2, is now getting started.
The IRONOUT HF trial was funded by the National Institutes of Health. Dr. Lewis reported receiving research support from a handful of pharmaceutical and medical device companies. Dr. van Velduisen reported serving as a scientific adviser to Vifor Pharma, which sponsored the EFFECT-HF trial. Dr. Anker, who was an EFFECT-HF investigator, serves as a consultant to Vifor and several other companies. Dr. Hernandez was an IRONOUT HF investigator and reported receiving research grants from a handful of pharmaceutical companies.
EXPERT ANALYSIS FROM THE AHA SCIENTIFIC SESSIONS
PPIs may boost ischemic stroke risk
NEW ORLEANS – The use of proton pump inhibitors (PPIs) was associated with significantly increased risk of having a first ischemic stroke in a large nationwide Danish cohort study, Thomas S. Sehested, MD, reported at the American Heart Association scientific sessions.
The relationship was dose dependent. At the lowest available dose of each of the four PPIs studied there was no significantly increased risk. At the intermediate doses of three of the four PPIs studied, the increased risk of ischemic stroke became statistically significant. And the highest dose of each drug was associated with the greatest ischemic stroke risk.
In Denmark, for instance, where most PPIs are prescription only and use is easily trackable, it’s estimated that, at any given time, 7% of the adult population is taking a PPI, often not as directed in the labeling.
The impetus for this study, Dr. Sehested explained, was the mounting evidence that PPIs may constitute an independent risk factor for acute MI and other cardiovascular events. For example, a recent meta-analysis of 17 randomized controlled trials totaling 7,540 participants published through mid-2015 concluded that the use of PPIs was associated with a 70% increase in cardiovascular risk (Neurogastroenterol Motil. 2016 Aug 30. doi: 10.1111/nmo.12926).
He reported on 245,676 Danes above age 30 who were free of prior MI or stroke when they underwent elective GI endoscopy during 1997-2012. After a 30-day postendoscopy grace period during which 1,476 patients had a first MI, stroke, or died of any cause, the final study population was 244,200, of whom 43.7% were PPI users during the grace period and beyond.
During a median 5.8 years of follow-up, 9,489 subjects (3.9%) had a first ischemic stroke. Because of the comprehensive nature of Denmark’s interlocking birth to death registries, there was virtually no loss to follow-up in this study.
The unadjusted incidence of ischemic stroke in PPI nonusers was 55.7 per 10,000 person-years, compared with 88.9 per 10,000 in PPI users.
The PPI users were slightly older than nonusers by roughly 3 years. They were also an absolute 5% more likely to be hypertensive and an absolute 1.7% more likely to be regular users of NSAIDs. All of these differences, while modest, were statistically significant because of the large patient numbers involved.
In a multivariate analysis adjusted for age, sex, calendar year, comorbid diabetes, hypertension, alcohol use disorder, heart failure, peptic ulcer, peripheral artery disease, kidney disease, aspirin, oral anticoagulants and other medications, and socioeconomic status, current users of PPIs were 19% more likely to have a first ischemic stroke than nonusers. That difference is statistically significant and clinically meaningful, Dr. Sehested said.
In contrast, when the same sort of nationwide analysis was repeated, comparing current users of histamine-2 receptor antagonists to nonusers of those drugs or PPIs, there was no difference in ischemic stroke risk between the two groups.
The message, according to Dr. Sehested, is that physicians should encourage more cautious use of PPIs. And especially in the United States, where most PPIs are available over the counter, it’s prudent during office visits to ask what nonprescription drugs a patient is taking.
Dr. Sehested presented his study findings in a session devoted to original research in cardiovascular epidemiology. Many top American epidemiologists were present in the audience, and several rose to congratulate him on his presentation of the latest elegant epidemiologic study to come out of Denmark, the only place in the world where this sort of nationwide comprehensive research is possible.
“Wow! I just love the work you do in Denmark. It’s really inspiring,” commented David Siscovick, MD, senior vice president for research at the New York Academy of Medicine and professor emeritus of medicine and epidemiology at the University of Washington in Seattle.
He had a question: “Did you deal with PPI starters and stoppers and compliance in any way?”
Dr. Sehested replied that he and his coinvestigators were able to see who was on a PPI at any given point in the study, and they accounted for that. One issue the researchers plan to examine but haven’t yet had a chance to, however, is the relationship between duration of PPI therapy and ischemic stroke risk. It’s likely that some patients had already been on a PPI for a lengthy time at elective endoscopy, which is when the study in its current form began.
“I think that would strengthen the study,” he said.
Comoderator Jorge Kizer, MD, of Albert Einstein College of Medicine in New York, commented, “Confounding by indication is clearly the elephant in the room. The guidelines actually recommend adding a PPI if a patient is on dual-antiplatelet therapy and has an NSAID added. Did you adjust for that? It would boost confidence that the results are actually due to the PPI.”
Dr. Sehested answered that the great majority of individuals with cardiovascular disease at baseline were excluded from the analysis.
“I don’t think we had that many on dual-antiplatelet therapy,” he added.
Preclinical studies suggest a possible mechanism by which PPIs may harm cardiovascular health. The drugs reduce nitric oxide synthase levels, with resultant endothelial dysfunction, he said.
Dr. Sehested is employed at the Danish Heart Foundation, which funded the study.
A growing number of retrospective studies have associated proton pump inhibitors with a host of serious adverse effects. These include chronic kidney disease, dementia, osteoporosis, cardiovascular events, pneumonia, enteric infections, and others. The authors of this large, retrospective Danish study have now added ischemic stroke to the list.
Nonetheless, this study should serve as wake-up call to closely examine the risks and benefits of ongoing PPI use for each individual patient. For example, guidelines clearly advocate the use of PPIs in patients at high risk for peptic ulcer disease (for example, use of aspirin and warfarin together), and these patients should continue PPIs unless more convincing evidence of serious side effects emerge. On the other hand, several studies have shown that many patients with uncomplicated gastroesophageal reflux disease symptoms can achieve symptom control with substitution of histamine2 blockers, p.r.n. dosing of PPIs, or without acid-reducing medications entirely. Still, many patients with confirmed pathologic acid reflux are likely to require ongoing PPIs. For patients who continue PPIs, they should use the lowest effective dose.
Surely, physicians will be discussing PPI adverse effects with increasing numbers of patients. Until higher-quality evidence in the form of a randomized controlled trial emerges, physicians should get used to explaining the principles of epidemiology.
Jacob Kurlander, MD, is a clinical lecturer in the division of gastroenterology, University of Michigan, Ann Arbor. He has received research funding from Ironwood Pharmaceuticals.
A growing number of retrospective studies have associated proton pump inhibitors with a host of serious adverse effects. These include chronic kidney disease, dementia, osteoporosis, cardiovascular events, pneumonia, enteric infections, and others. The authors of this large, retrospective Danish study have now added ischemic stroke to the list.
Nonetheless, this study should serve as wake-up call to closely examine the risks and benefits of ongoing PPI use for each individual patient. For example, guidelines clearly advocate the use of PPIs in patients at high risk for peptic ulcer disease (for example, use of aspirin and warfarin together), and these patients should continue PPIs unless more convincing evidence of serious side effects emerge. On the other hand, several studies have shown that many patients with uncomplicated gastroesophageal reflux disease symptoms can achieve symptom control with substitution of histamine2 blockers, p.r.n. dosing of PPIs, or without acid-reducing medications entirely. Still, many patients with confirmed pathologic acid reflux are likely to require ongoing PPIs. For patients who continue PPIs, they should use the lowest effective dose.
Surely, physicians will be discussing PPI adverse effects with increasing numbers of patients. Until higher-quality evidence in the form of a randomized controlled trial emerges, physicians should get used to explaining the principles of epidemiology.
Jacob Kurlander, MD, is a clinical lecturer in the division of gastroenterology, University of Michigan, Ann Arbor. He has received research funding from Ironwood Pharmaceuticals.
A growing number of retrospective studies have associated proton pump inhibitors with a host of serious adverse effects. These include chronic kidney disease, dementia, osteoporosis, cardiovascular events, pneumonia, enteric infections, and others. The authors of this large, retrospective Danish study have now added ischemic stroke to the list.
Nonetheless, this study should serve as wake-up call to closely examine the risks and benefits of ongoing PPI use for each individual patient. For example, guidelines clearly advocate the use of PPIs in patients at high risk for peptic ulcer disease (for example, use of aspirin and warfarin together), and these patients should continue PPIs unless more convincing evidence of serious side effects emerge. On the other hand, several studies have shown that many patients with uncomplicated gastroesophageal reflux disease symptoms can achieve symptom control with substitution of histamine2 blockers, p.r.n. dosing of PPIs, or without acid-reducing medications entirely. Still, many patients with confirmed pathologic acid reflux are likely to require ongoing PPIs. For patients who continue PPIs, they should use the lowest effective dose.
Surely, physicians will be discussing PPI adverse effects with increasing numbers of patients. Until higher-quality evidence in the form of a randomized controlled trial emerges, physicians should get used to explaining the principles of epidemiology.
Jacob Kurlander, MD, is a clinical lecturer in the division of gastroenterology, University of Michigan, Ann Arbor. He has received research funding from Ironwood Pharmaceuticals.
NEW ORLEANS – The use of proton pump inhibitors (PPIs) was associated with significantly increased risk of having a first ischemic stroke in a large nationwide Danish cohort study, Thomas S. Sehested, MD, reported at the American Heart Association scientific sessions.
The relationship was dose dependent. At the lowest available dose of each of the four PPIs studied there was no significantly increased risk. At the intermediate doses of three of the four PPIs studied, the increased risk of ischemic stroke became statistically significant. And the highest dose of each drug was associated with the greatest ischemic stroke risk.
In Denmark, for instance, where most PPIs are prescription only and use is easily trackable, it’s estimated that, at any given time, 7% of the adult population is taking a PPI, often not as directed in the labeling.
The impetus for this study, Dr. Sehested explained, was the mounting evidence that PPIs may constitute an independent risk factor for acute MI and other cardiovascular events. For example, a recent meta-analysis of 17 randomized controlled trials totaling 7,540 participants published through mid-2015 concluded that the use of PPIs was associated with a 70% increase in cardiovascular risk (Neurogastroenterol Motil. 2016 Aug 30. doi: 10.1111/nmo.12926).
He reported on 245,676 Danes above age 30 who were free of prior MI or stroke when they underwent elective GI endoscopy during 1997-2012. After a 30-day postendoscopy grace period during which 1,476 patients had a first MI, stroke, or died of any cause, the final study population was 244,200, of whom 43.7% were PPI users during the grace period and beyond.
During a median 5.8 years of follow-up, 9,489 subjects (3.9%) had a first ischemic stroke. Because of the comprehensive nature of Denmark’s interlocking birth to death registries, there was virtually no loss to follow-up in this study.
The unadjusted incidence of ischemic stroke in PPI nonusers was 55.7 per 10,000 person-years, compared with 88.9 per 10,000 in PPI users.
The PPI users were slightly older than nonusers by roughly 3 years. They were also an absolute 5% more likely to be hypertensive and an absolute 1.7% more likely to be regular users of NSAIDs. All of these differences, while modest, were statistically significant because of the large patient numbers involved.
In a multivariate analysis adjusted for age, sex, calendar year, comorbid diabetes, hypertension, alcohol use disorder, heart failure, peptic ulcer, peripheral artery disease, kidney disease, aspirin, oral anticoagulants and other medications, and socioeconomic status, current users of PPIs were 19% more likely to have a first ischemic stroke than nonusers. That difference is statistically significant and clinically meaningful, Dr. Sehested said.
In contrast, when the same sort of nationwide analysis was repeated, comparing current users of histamine-2 receptor antagonists to nonusers of those drugs or PPIs, there was no difference in ischemic stroke risk between the two groups.
The message, according to Dr. Sehested, is that physicians should encourage more cautious use of PPIs. And especially in the United States, where most PPIs are available over the counter, it’s prudent during office visits to ask what nonprescription drugs a patient is taking.
Dr. Sehested presented his study findings in a session devoted to original research in cardiovascular epidemiology. Many top American epidemiologists were present in the audience, and several rose to congratulate him on his presentation of the latest elegant epidemiologic study to come out of Denmark, the only place in the world where this sort of nationwide comprehensive research is possible.
“Wow! I just love the work you do in Denmark. It’s really inspiring,” commented David Siscovick, MD, senior vice president for research at the New York Academy of Medicine and professor emeritus of medicine and epidemiology at the University of Washington in Seattle.
He had a question: “Did you deal with PPI starters and stoppers and compliance in any way?”
Dr. Sehested replied that he and his coinvestigators were able to see who was on a PPI at any given point in the study, and they accounted for that. One issue the researchers plan to examine but haven’t yet had a chance to, however, is the relationship between duration of PPI therapy and ischemic stroke risk. It’s likely that some patients had already been on a PPI for a lengthy time at elective endoscopy, which is when the study in its current form began.
“I think that would strengthen the study,” he said.
Comoderator Jorge Kizer, MD, of Albert Einstein College of Medicine in New York, commented, “Confounding by indication is clearly the elephant in the room. The guidelines actually recommend adding a PPI if a patient is on dual-antiplatelet therapy and has an NSAID added. Did you adjust for that? It would boost confidence that the results are actually due to the PPI.”
Dr. Sehested answered that the great majority of individuals with cardiovascular disease at baseline were excluded from the analysis.
“I don’t think we had that many on dual-antiplatelet therapy,” he added.
Preclinical studies suggest a possible mechanism by which PPIs may harm cardiovascular health. The drugs reduce nitric oxide synthase levels, with resultant endothelial dysfunction, he said.
Dr. Sehested is employed at the Danish Heart Foundation, which funded the study.
NEW ORLEANS – The use of proton pump inhibitors (PPIs) was associated with significantly increased risk of having a first ischemic stroke in a large nationwide Danish cohort study, Thomas S. Sehested, MD, reported at the American Heart Association scientific sessions.
The relationship was dose dependent. At the lowest available dose of each of the four PPIs studied there was no significantly increased risk. At the intermediate doses of three of the four PPIs studied, the increased risk of ischemic stroke became statistically significant. And the highest dose of each drug was associated with the greatest ischemic stroke risk.
In Denmark, for instance, where most PPIs are prescription only and use is easily trackable, it’s estimated that, at any given time, 7% of the adult population is taking a PPI, often not as directed in the labeling.
The impetus for this study, Dr. Sehested explained, was the mounting evidence that PPIs may constitute an independent risk factor for acute MI and other cardiovascular events. For example, a recent meta-analysis of 17 randomized controlled trials totaling 7,540 participants published through mid-2015 concluded that the use of PPIs was associated with a 70% increase in cardiovascular risk (Neurogastroenterol Motil. 2016 Aug 30. doi: 10.1111/nmo.12926).
He reported on 245,676 Danes above age 30 who were free of prior MI or stroke when they underwent elective GI endoscopy during 1997-2012. After a 30-day postendoscopy grace period during which 1,476 patients had a first MI, stroke, or died of any cause, the final study population was 244,200, of whom 43.7% were PPI users during the grace period and beyond.
During a median 5.8 years of follow-up, 9,489 subjects (3.9%) had a first ischemic stroke. Because of the comprehensive nature of Denmark’s interlocking birth to death registries, there was virtually no loss to follow-up in this study.
The unadjusted incidence of ischemic stroke in PPI nonusers was 55.7 per 10,000 person-years, compared with 88.9 per 10,000 in PPI users.
The PPI users were slightly older than nonusers by roughly 3 years. They were also an absolute 5% more likely to be hypertensive and an absolute 1.7% more likely to be regular users of NSAIDs. All of these differences, while modest, were statistically significant because of the large patient numbers involved.
In a multivariate analysis adjusted for age, sex, calendar year, comorbid diabetes, hypertension, alcohol use disorder, heart failure, peptic ulcer, peripheral artery disease, kidney disease, aspirin, oral anticoagulants and other medications, and socioeconomic status, current users of PPIs were 19% more likely to have a first ischemic stroke than nonusers. That difference is statistically significant and clinically meaningful, Dr. Sehested said.
In contrast, when the same sort of nationwide analysis was repeated, comparing current users of histamine-2 receptor antagonists to nonusers of those drugs or PPIs, there was no difference in ischemic stroke risk between the two groups.
The message, according to Dr. Sehested, is that physicians should encourage more cautious use of PPIs. And especially in the United States, where most PPIs are available over the counter, it’s prudent during office visits to ask what nonprescription drugs a patient is taking.
Dr. Sehested presented his study findings in a session devoted to original research in cardiovascular epidemiology. Many top American epidemiologists were present in the audience, and several rose to congratulate him on his presentation of the latest elegant epidemiologic study to come out of Denmark, the only place in the world where this sort of nationwide comprehensive research is possible.
“Wow! I just love the work you do in Denmark. It’s really inspiring,” commented David Siscovick, MD, senior vice president for research at the New York Academy of Medicine and professor emeritus of medicine and epidemiology at the University of Washington in Seattle.
He had a question: “Did you deal with PPI starters and stoppers and compliance in any way?”
Dr. Sehested replied that he and his coinvestigators were able to see who was on a PPI at any given point in the study, and they accounted for that. One issue the researchers plan to examine but haven’t yet had a chance to, however, is the relationship between duration of PPI therapy and ischemic stroke risk. It’s likely that some patients had already been on a PPI for a lengthy time at elective endoscopy, which is when the study in its current form began.
“I think that would strengthen the study,” he said.
Comoderator Jorge Kizer, MD, of Albert Einstein College of Medicine in New York, commented, “Confounding by indication is clearly the elephant in the room. The guidelines actually recommend adding a PPI if a patient is on dual-antiplatelet therapy and has an NSAID added. Did you adjust for that? It would boost confidence that the results are actually due to the PPI.”
Dr. Sehested answered that the great majority of individuals with cardiovascular disease at baseline were excluded from the analysis.
“I don’t think we had that many on dual-antiplatelet therapy,” he added.
Preclinical studies suggest a possible mechanism by which PPIs may harm cardiovascular health. The drugs reduce nitric oxide synthase levels, with resultant endothelial dysfunction, he said.
Dr. Sehested is employed at the Danish Heart Foundation, which funded the study.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: Current use of a proton pump inhibitor was independently associated with a 19% increased risk of a first ischemic stroke, and the risk was greater at the top approved doses.
Data source: This retrospective nationwide Danish study involved 244,200 adults age 30 or older followed for a median of nearly 6 years following elective GI endoscopy.
Disclosures: The presenter is employed at the Danish Heart Foundation, which funded the study.
A look at HIV-related cancers: incidence, screening, and stem transplantation
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
Pediatric psoriasis linked to multiple psychiatric comorbidities
VIENNA – Psoriasis in children and adolescents is associated with significantly increased risk of a variety of psychiatric comorbidities, according to a large Danish national study.
This finding has important public health implications. Psoriasis is a common skin disease, and 30% of cases have their onset in childhood or adolescence, Tanja Todberg, MD, observed at the annual congress of the European Academy of Dermatology and Venereology.
Diagnosis of psoriasis was based upon medical records and documentation that at least a second prescription for a topical vitamin D derivative had been filled. Those agents are the overwhelming choice as first-line therapy in the pediatric population, explained Dr. Todberg of the University of Copenhagen.
The pediatric psoriasis patients proved to be at significantly increased risk of being diagnosed with depression, eating disorders, drug abuse, and alcohol abuse. They were also more likely than controls to be prescribed antidepressants, antipsychotic agents, and benzodiazepines. That was every prespecified psychiatric outcome that Dr. Todberg and her coinvestigators included in the study except for one: anxiety disorders occurred at a similar rate in the pediatric psoriasis patients and controls.
Dr. Todberg reported having no financial conflicts of interest regarding this study, which was supported by Danish medical research funding.
bjancin@frontlinemedcom.com
VIENNA – Psoriasis in children and adolescents is associated with significantly increased risk of a variety of psychiatric comorbidities, according to a large Danish national study.
This finding has important public health implications. Psoriasis is a common skin disease, and 30% of cases have their onset in childhood or adolescence, Tanja Todberg, MD, observed at the annual congress of the European Academy of Dermatology and Venereology.
Diagnosis of psoriasis was based upon medical records and documentation that at least a second prescription for a topical vitamin D derivative had been filled. Those agents are the overwhelming choice as first-line therapy in the pediatric population, explained Dr. Todberg of the University of Copenhagen.
The pediatric psoriasis patients proved to be at significantly increased risk of being diagnosed with depression, eating disorders, drug abuse, and alcohol abuse. They were also more likely than controls to be prescribed antidepressants, antipsychotic agents, and benzodiazepines. That was every prespecified psychiatric outcome that Dr. Todberg and her coinvestigators included in the study except for one: anxiety disorders occurred at a similar rate in the pediatric psoriasis patients and controls.
Dr. Todberg reported having no financial conflicts of interest regarding this study, which was supported by Danish medical research funding.
bjancin@frontlinemedcom.com
VIENNA – Psoriasis in children and adolescents is associated with significantly increased risk of a variety of psychiatric comorbidities, according to a large Danish national study.
This finding has important public health implications. Psoriasis is a common skin disease, and 30% of cases have their onset in childhood or adolescence, Tanja Todberg, MD, observed at the annual congress of the European Academy of Dermatology and Venereology.
Diagnosis of psoriasis was based upon medical records and documentation that at least a second prescription for a topical vitamin D derivative had been filled. Those agents are the overwhelming choice as first-line therapy in the pediatric population, explained Dr. Todberg of the University of Copenhagen.
The pediatric psoriasis patients proved to be at significantly increased risk of being diagnosed with depression, eating disorders, drug abuse, and alcohol abuse. They were also more likely than controls to be prescribed antidepressants, antipsychotic agents, and benzodiazepines. That was every prespecified psychiatric outcome that Dr. Todberg and her coinvestigators included in the study except for one: anxiety disorders occurred at a similar rate in the pediatric psoriasis patients and controls.
Dr. Todberg reported having no financial conflicts of interest regarding this study, which was supported by Danish medical research funding.
bjancin@frontlinemedcom.com
AT THE EADV CONGRESS
Key clinical point:
Major finding: Danish pediatric patients with psoriasis were significantly more at risk of developing a range of psychiatric disorders than were control subjects.
Data source: This was a retrospective study of prospectively collected registry data on all 4,410 Danish children and adolescents diagnosed with psoriasis during 1997-2012 and more than 41,000 Danish controls matched for age, sex, and calendar year.
Disclosures: The presenter reported having no financial conflicts of interest regarding this study, which was supported by Danish medical research funds.
GLAGOV finds evolocumab plus statin drives unprecedented plaque regression
NEW ORLEANS – Adding the PCSK9 inhibitor evolocumab to maximum tolerated statin therapy in subjects with symptomatic coronary disease induced atheromatous plaque regression of a previously unheard-of scale in the phase III GLAGOV trial, Steven E. Nissen, MD, reported at the American Heart Association scientific sessions.
Over 18 months of follow-up, percent atheroma volume was unchanged in patients treated with maximum tolerated statin therapy. But in patients on maximum tolerated statin therapy plus evolocumab, mean atheroma volume decreased by 0.95%. A reduction in atheroma volume as small as 0.5% has been associated with a reduced rate of cardiovascular events in previous studies, according to GLAGOV principal investigator Stephen J. Nicholls, MBBS, PhD, of the University of Adelaide in Australia.
Total atheroma volume was reduced by 5.8 mm3 with dual therapy, compared with a nonsignificant 0.9-mm3 decrease in patients on statin monotherapy.
Among 423 patients on statin monotherapy, 47% had regression and 53% had progression of atheroma volume. In contrast, 64% of 423 patients on a statin plus evolocumab had atheroma regressions and 36% had atheroma progression.
What’s more, plaque regression was directly related to declines in LDL levels.
“We saw a linear relationship between lower LDL and greater regression with no tailing off of benefit at very low LDL levels. Down to 20 mg/dL, it’s continuous and linear,” the cardiologist said. “I thought there might be diminishing return at low LDL levels. We don’t see that.”
GLAGOV (Global Assessment of Plaque Regression with a PCSK9 antibody as Measured by Intravascular Ultrasound) was a double-blind, placebo-controlled, randomized trial including 846 evaluable patients with symptomatic CAD at 197 centers. All were on a stable maximum tolerated dose of a statin at baseline, at which point they underwent intravascular ultrasound (IVUS) and were assigned to subcutaneous evolocumab (Repatha) at 420 mg once monthly or placebo injections while continuing on their statin. At 18 months, participants had a follow-up IVUS of the originally imaged target vessel.
The mean LDL-cholesterol level was 93 mg/dL at the start of the study. With the addition of evolocumab, the mean LDL level dropped to 37 mg/dL; that’s a 60% further reduction below the level on statin alone.
The investigators conducted a post hoc exploratory subgroup analysis confined to 144 patients whose baseline LDL on statin monotherapy was less than 70 mg/dL. In this group, the addition of evolocumab caused the mean LDL level to plunge to 24 mg/dL. Mean atheroma volume in dual-treatment patients with a baseline LDL below 70 mg/dL decreased by 1.97% – double the reduction seen in the overall statin/evolocumab study arm. Further, 81% of patients on dual therapy who started out with an LDL below 70 mg/dL showed IVUS evidence of plaque regression, compared with 48% of those on statin monotherapy. For those whose baseline LDL level was below 70 mg/dL and who remained on statin monotherapy, there was no change in atheroma volume over time.
No signals of any safety concerns arose in the group on a statin plus evolocumab. Of particular interest, there was no increase in new-onset diabetes, neurocognitive dysfunction, or myalgia in patients given dual therapy, compared with patients on a statin alone.
Dr. Santos, of the University of Sao Paolo in Brazil, noted that GLAGOV wasn’t powered to provide evidence of long-term safety or of hard clinical endpoints such as acute MI. For those outcomes, physicians must await the soon-to-come results of the nearly 28,000-patient FOURIER clinical outcomes trial.
The GLAGOV trial was sponsored by Amgen, the maker of evolocumab (Repatha). Dr. Santos reported serving as a consultant to and paid researcher for the company. Dr. Nissen reported serving as a consultant to Amgen and several other companies; any resultant consultant fees are directly paid to charities. Dr. Nicholls reported receiving research support from and serving as a consultant to Amgen and several other drug companies.
Simultaneously with Dr. Nissen’s presentation in New Orleans, the GLAGOV results were published online (JAMA. 2016 Nov 15. doi: 10.1001/jama.2016.16951).
NEW ORLEANS – Adding the PCSK9 inhibitor evolocumab to maximum tolerated statin therapy in subjects with symptomatic coronary disease induced atheromatous plaque regression of a previously unheard-of scale in the phase III GLAGOV trial, Steven E. Nissen, MD, reported at the American Heart Association scientific sessions.
Over 18 months of follow-up, percent atheroma volume was unchanged in patients treated with maximum tolerated statin therapy. But in patients on maximum tolerated statin therapy plus evolocumab, mean atheroma volume decreased by 0.95%. A reduction in atheroma volume as small as 0.5% has been associated with a reduced rate of cardiovascular events in previous studies, according to GLAGOV principal investigator Stephen J. Nicholls, MBBS, PhD, of the University of Adelaide in Australia.
Total atheroma volume was reduced by 5.8 mm3 with dual therapy, compared with a nonsignificant 0.9-mm3 decrease in patients on statin monotherapy.
Among 423 patients on statin monotherapy, 47% had regression and 53% had progression of atheroma volume. In contrast, 64% of 423 patients on a statin plus evolocumab had atheroma regressions and 36% had atheroma progression.
What’s more, plaque regression was directly related to declines in LDL levels.
“We saw a linear relationship between lower LDL and greater regression with no tailing off of benefit at very low LDL levels. Down to 20 mg/dL, it’s continuous and linear,” the cardiologist said. “I thought there might be diminishing return at low LDL levels. We don’t see that.”
GLAGOV (Global Assessment of Plaque Regression with a PCSK9 antibody as Measured by Intravascular Ultrasound) was a double-blind, placebo-controlled, randomized trial including 846 evaluable patients with symptomatic CAD at 197 centers. All were on a stable maximum tolerated dose of a statin at baseline, at which point they underwent intravascular ultrasound (IVUS) and were assigned to subcutaneous evolocumab (Repatha) at 420 mg once monthly or placebo injections while continuing on their statin. At 18 months, participants had a follow-up IVUS of the originally imaged target vessel.
The mean LDL-cholesterol level was 93 mg/dL at the start of the study. With the addition of evolocumab, the mean LDL level dropped to 37 mg/dL; that’s a 60% further reduction below the level on statin alone.
The investigators conducted a post hoc exploratory subgroup analysis confined to 144 patients whose baseline LDL on statin monotherapy was less than 70 mg/dL. In this group, the addition of evolocumab caused the mean LDL level to plunge to 24 mg/dL. Mean atheroma volume in dual-treatment patients with a baseline LDL below 70 mg/dL decreased by 1.97% – double the reduction seen in the overall statin/evolocumab study arm. Further, 81% of patients on dual therapy who started out with an LDL below 70 mg/dL showed IVUS evidence of plaque regression, compared with 48% of those on statin monotherapy. For those whose baseline LDL level was below 70 mg/dL and who remained on statin monotherapy, there was no change in atheroma volume over time.
No signals of any safety concerns arose in the group on a statin plus evolocumab. Of particular interest, there was no increase in new-onset diabetes, neurocognitive dysfunction, or myalgia in patients given dual therapy, compared with patients on a statin alone.
Dr. Santos, of the University of Sao Paolo in Brazil, noted that GLAGOV wasn’t powered to provide evidence of long-term safety or of hard clinical endpoints such as acute MI. For those outcomes, physicians must await the soon-to-come results of the nearly 28,000-patient FOURIER clinical outcomes trial.
The GLAGOV trial was sponsored by Amgen, the maker of evolocumab (Repatha). Dr. Santos reported serving as a consultant to and paid researcher for the company. Dr. Nissen reported serving as a consultant to Amgen and several other companies; any resultant consultant fees are directly paid to charities. Dr. Nicholls reported receiving research support from and serving as a consultant to Amgen and several other drug companies.
Simultaneously with Dr. Nissen’s presentation in New Orleans, the GLAGOV results were published online (JAMA. 2016 Nov 15. doi: 10.1001/jama.2016.16951).
NEW ORLEANS – Adding the PCSK9 inhibitor evolocumab to maximum tolerated statin therapy in subjects with symptomatic coronary disease induced atheromatous plaque regression of a previously unheard-of scale in the phase III GLAGOV trial, Steven E. Nissen, MD, reported at the American Heart Association scientific sessions.
Over 18 months of follow-up, percent atheroma volume was unchanged in patients treated with maximum tolerated statin therapy. But in patients on maximum tolerated statin therapy plus evolocumab, mean atheroma volume decreased by 0.95%. A reduction in atheroma volume as small as 0.5% has been associated with a reduced rate of cardiovascular events in previous studies, according to GLAGOV principal investigator Stephen J. Nicholls, MBBS, PhD, of the University of Adelaide in Australia.
Total atheroma volume was reduced by 5.8 mm3 with dual therapy, compared with a nonsignificant 0.9-mm3 decrease in patients on statin monotherapy.
Among 423 patients on statin monotherapy, 47% had regression and 53% had progression of atheroma volume. In contrast, 64% of 423 patients on a statin plus evolocumab had atheroma regressions and 36% had atheroma progression.
What’s more, plaque regression was directly related to declines in LDL levels.
“We saw a linear relationship between lower LDL and greater regression with no tailing off of benefit at very low LDL levels. Down to 20 mg/dL, it’s continuous and linear,” the cardiologist said. “I thought there might be diminishing return at low LDL levels. We don’t see that.”
GLAGOV (Global Assessment of Plaque Regression with a PCSK9 antibody as Measured by Intravascular Ultrasound) was a double-blind, placebo-controlled, randomized trial including 846 evaluable patients with symptomatic CAD at 197 centers. All were on a stable maximum tolerated dose of a statin at baseline, at which point they underwent intravascular ultrasound (IVUS) and were assigned to subcutaneous evolocumab (Repatha) at 420 mg once monthly or placebo injections while continuing on their statin. At 18 months, participants had a follow-up IVUS of the originally imaged target vessel.
The mean LDL-cholesterol level was 93 mg/dL at the start of the study. With the addition of evolocumab, the mean LDL level dropped to 37 mg/dL; that’s a 60% further reduction below the level on statin alone.
The investigators conducted a post hoc exploratory subgroup analysis confined to 144 patients whose baseline LDL on statin monotherapy was less than 70 mg/dL. In this group, the addition of evolocumab caused the mean LDL level to plunge to 24 mg/dL. Mean atheroma volume in dual-treatment patients with a baseline LDL below 70 mg/dL decreased by 1.97% – double the reduction seen in the overall statin/evolocumab study arm. Further, 81% of patients on dual therapy who started out with an LDL below 70 mg/dL showed IVUS evidence of plaque regression, compared with 48% of those on statin monotherapy. For those whose baseline LDL level was below 70 mg/dL and who remained on statin monotherapy, there was no change in atheroma volume over time.
No signals of any safety concerns arose in the group on a statin plus evolocumab. Of particular interest, there was no increase in new-onset diabetes, neurocognitive dysfunction, or myalgia in patients given dual therapy, compared with patients on a statin alone.
Dr. Santos, of the University of Sao Paolo in Brazil, noted that GLAGOV wasn’t powered to provide evidence of long-term safety or of hard clinical endpoints such as acute MI. For those outcomes, physicians must await the soon-to-come results of the nearly 28,000-patient FOURIER clinical outcomes trial.
The GLAGOV trial was sponsored by Amgen, the maker of evolocumab (Repatha). Dr. Santos reported serving as a consultant to and paid researcher for the company. Dr. Nissen reported serving as a consultant to Amgen and several other companies; any resultant consultant fees are directly paid to charities. Dr. Nicholls reported receiving research support from and serving as a consultant to Amgen and several other drug companies.
Simultaneously with Dr. Nissen’s presentation in New Orleans, the GLAGOV results were published online (JAMA. 2016 Nov 15. doi: 10.1001/jama.2016.16951).
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: Total atheroma volume was reduced by 5.8 mm3 with dual therapy, compared with a nonsignificant 0.9-mm3 decrease in patients on statin monotherapy.
Data source: GLAGOV, a phase III, randomized, double-blind, 18-month intravascular ultrasound study of 846 evaluable patients with symptomatic CAD.
Disclosures: The GLAGOV trial was sponsored by Amgen, maker of evolocumab (Repatha). Dr. Nissen reported serving as a consultant to Amgen and several other companies; any resultant consultant fees are directly paid to charities. Dr. Nicholls reported receiving research support from and serving as a consultant to Amgen and several other drug companies. Dr. Santos reported serving as a consultant to and paid researcher for the company.