User login
Hydroxychloroquine dosage recommendations often ignored
Evidence continues to mount that some North American rheumatologists are not following practice recommendations for minimizing the retinal toxicity risk of patients on long-term hydroxychloroquine treatment.
An audit of 100 patients seen at any of nine Canadian rheumatology clinics during early 2016 showed that 30% of patients were not on appropriate weight-based hydroxychloroquine dosages, and 13% of patients on the drug had not received a baseline retinal assessment during their first year of treatment, Sahil Koppikar, MD, reported in a poster presented at the annual meeting of the Canadian Rheumatology Association in Ottawa in February.
In a second recently reported study, researchers from the Chicago area documented that roughly half of the 554 rheumatology patients on hydroxychloroquine (HCQ) in a regional health system and seen by an ophthalmologist during 2009-2016 received an excessive dosage of the drug (Ophthalmology. 2017 Jan 30. doi: 10.1016/j.ophtha.2016.12.021).
Although his study did not examine reasons for the compliance shortfall, Dr. Koppikar proposed some possible factors.
HCQ comes only as 200-mg tablets, and prescribing intermediate dosages can be a challenge (although veteran clinicians know that a safe and easy way to dial down a dosage is to have the patient periodically skip a dose). Also, “it is more convenient to prescribe 400 mg daily rather than calculate an exact dosage,” Dr. Koppikar said in an interview. In addition, rheumatologists may be unaware that the prevalence of retinopathy in patients on HCQ is fairly common (about 8% in one large recent study), assessment for risk factors that heighten sensitivity to the drug isn’t always done, and appointments for retinal screening can fall through the cracks.
“It behooves rheumatologists to adopt the [HCQ] recommendations of the American Academy of Ophthalmology [AAO] because there is more toxicity than we previously appreciated,” he added. A new version of the AAO’s recommendations came out in March 2016.
The Committee on Rheumatologic Care of the American College of Rheumatology (ACR) has regularly updated the ACR’s position statement on screening for HCQ retinopathy and appropriate dosages, with the most recent version out in August 2016. The August statement “is very similar” to the AAO’s 2016 recommendations, said Vinicius Domingues, MD, a member of the committee and an ACR spokesman for the revision. The ACR statement acknowledges and cites the AAO 2016 recommendations.
The ACR’s 2016 statement also does not fully endorse the AAO’s 2016 firm statement that “all patients using HCQ keep daily dosage less than 5.0 mg/kg real weight,” aside from “rare instances” when a higher dosage is needed to treat a “life-threatening disease.”
The ACR 2016 statement goes on to note that other authors have recommended a dosage of 6.5 mg/kg of actual body weight but capped at 400 mg/day and adjusted for renal insufficiency, and the ACR statement stops short of specifying which dosage strategy it recommends.
“The AAO recommendations are much more definitive and state more specifically what screening is recommended and what is a safe dosage,” commented Dr. Rosenbaum.
Dr. Domingues agreed that rheumatologist compliance with HCQ best practices has been spotty.
“In the past few years, more studies have used new ways to detect macular abnormalities and have identified a higher-than-expected incidence of maculopathy. Through lectures, CME, and articles, rheumatologists have received a tremendous amount of information with regard to screening and preventing retinal toxicity,” he said in an interview. “There are still gaps, and some rheumatologists still prescribe HCQ without taking into consideration the patient’s weight.”
That was a key finding in the poster presented at the Canadian Rheumatology Association by Dr. Koppikar and his collaborator on the study, Henry Averns, MD. The 100 patients assessed through the nine-clinic audit process averaged 58 years old, 81% were women, and patients had been taking HCQ for an average of just over 6 years, primarily for rheumatoid arthritis or systemic lupus erythematosus. Nearly two-thirds had a high risk for retinal toxicity. Based on the 2011 recommendations from the AAO and ACR, 17% of the patients were receiving an HCQ overdose that was more than 10% above the recommended dosage, and another 13% received a smaller overdose. If the 2016 dosage guidelines were applied, the extent of overdosing might be even greater, Dr. Koppikar said.
Dr. Koppikar and Dr. Averns said they believe that one way to address HCQ overdosing is by giving clinicians a dosing chart to easily find the right dosage for a patient’s weight. They have distributed these charts to the practices they audited and plan to do a follow-up audit to measure the effect of the intervention on HCQ prescribing.
Results from the initial clinical audit showed that “clinicians were not meeting standards, and we needed an intervention [a dosing chart] to implement a change,” Dr. Koppikar said. “Clinical audits are easy to implement, cost effective, and help improve patient care.”
Dr. Koppikar, Dr. Rosenbaum, Dr. Domingues, and Dr. Averns had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Evidence continues to mount that some North American rheumatologists are not following practice recommendations for minimizing the retinal toxicity risk of patients on long-term hydroxychloroquine treatment.
An audit of 100 patients seen at any of nine Canadian rheumatology clinics during early 2016 showed that 30% of patients were not on appropriate weight-based hydroxychloroquine dosages, and 13% of patients on the drug had not received a baseline retinal assessment during their first year of treatment, Sahil Koppikar, MD, reported in a poster presented at the annual meeting of the Canadian Rheumatology Association in Ottawa in February.
In a second recently reported study, researchers from the Chicago area documented that roughly half of the 554 rheumatology patients on hydroxychloroquine (HCQ) in a regional health system and seen by an ophthalmologist during 2009-2016 received an excessive dosage of the drug (Ophthalmology. 2017 Jan 30. doi: 10.1016/j.ophtha.2016.12.021).
Although his study did not examine reasons for the compliance shortfall, Dr. Koppikar proposed some possible factors.
HCQ comes only as 200-mg tablets, and prescribing intermediate dosages can be a challenge (although veteran clinicians know that a safe and easy way to dial down a dosage is to have the patient periodically skip a dose). Also, “it is more convenient to prescribe 400 mg daily rather than calculate an exact dosage,” Dr. Koppikar said in an interview. In addition, rheumatologists may be unaware that the prevalence of retinopathy in patients on HCQ is fairly common (about 8% in one large recent study), assessment for risk factors that heighten sensitivity to the drug isn’t always done, and appointments for retinal screening can fall through the cracks.
“It behooves rheumatologists to adopt the [HCQ] recommendations of the American Academy of Ophthalmology [AAO] because there is more toxicity than we previously appreciated,” he added. A new version of the AAO’s recommendations came out in March 2016.
The Committee on Rheumatologic Care of the American College of Rheumatology (ACR) has regularly updated the ACR’s position statement on screening for HCQ retinopathy and appropriate dosages, with the most recent version out in August 2016. The August statement “is very similar” to the AAO’s 2016 recommendations, said Vinicius Domingues, MD, a member of the committee and an ACR spokesman for the revision. The ACR statement acknowledges and cites the AAO 2016 recommendations.
The ACR’s 2016 statement also does not fully endorse the AAO’s 2016 firm statement that “all patients using HCQ keep daily dosage less than 5.0 mg/kg real weight,” aside from “rare instances” when a higher dosage is needed to treat a “life-threatening disease.”
The ACR 2016 statement goes on to note that other authors have recommended a dosage of 6.5 mg/kg of actual body weight but capped at 400 mg/day and adjusted for renal insufficiency, and the ACR statement stops short of specifying which dosage strategy it recommends.
“The AAO recommendations are much more definitive and state more specifically what screening is recommended and what is a safe dosage,” commented Dr. Rosenbaum.
Dr. Domingues agreed that rheumatologist compliance with HCQ best practices has been spotty.
“In the past few years, more studies have used new ways to detect macular abnormalities and have identified a higher-than-expected incidence of maculopathy. Through lectures, CME, and articles, rheumatologists have received a tremendous amount of information with regard to screening and preventing retinal toxicity,” he said in an interview. “There are still gaps, and some rheumatologists still prescribe HCQ without taking into consideration the patient’s weight.”
That was a key finding in the poster presented at the Canadian Rheumatology Association by Dr. Koppikar and his collaborator on the study, Henry Averns, MD. The 100 patients assessed through the nine-clinic audit process averaged 58 years old, 81% were women, and patients had been taking HCQ for an average of just over 6 years, primarily for rheumatoid arthritis or systemic lupus erythematosus. Nearly two-thirds had a high risk for retinal toxicity. Based on the 2011 recommendations from the AAO and ACR, 17% of the patients were receiving an HCQ overdose that was more than 10% above the recommended dosage, and another 13% received a smaller overdose. If the 2016 dosage guidelines were applied, the extent of overdosing might be even greater, Dr. Koppikar said.
Dr. Koppikar and Dr. Averns said they believe that one way to address HCQ overdosing is by giving clinicians a dosing chart to easily find the right dosage for a patient’s weight. They have distributed these charts to the practices they audited and plan to do a follow-up audit to measure the effect of the intervention on HCQ prescribing.
Results from the initial clinical audit showed that “clinicians were not meeting standards, and we needed an intervention [a dosing chart] to implement a change,” Dr. Koppikar said. “Clinical audits are easy to implement, cost effective, and help improve patient care.”
Dr. Koppikar, Dr. Rosenbaum, Dr. Domingues, and Dr. Averns had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Evidence continues to mount that some North American rheumatologists are not following practice recommendations for minimizing the retinal toxicity risk of patients on long-term hydroxychloroquine treatment.
An audit of 100 patients seen at any of nine Canadian rheumatology clinics during early 2016 showed that 30% of patients were not on appropriate weight-based hydroxychloroquine dosages, and 13% of patients on the drug had not received a baseline retinal assessment during their first year of treatment, Sahil Koppikar, MD, reported in a poster presented at the annual meeting of the Canadian Rheumatology Association in Ottawa in February.
In a second recently reported study, researchers from the Chicago area documented that roughly half of the 554 rheumatology patients on hydroxychloroquine (HCQ) in a regional health system and seen by an ophthalmologist during 2009-2016 received an excessive dosage of the drug (Ophthalmology. 2017 Jan 30. doi: 10.1016/j.ophtha.2016.12.021).
Although his study did not examine reasons for the compliance shortfall, Dr. Koppikar proposed some possible factors.
HCQ comes only as 200-mg tablets, and prescribing intermediate dosages can be a challenge (although veteran clinicians know that a safe and easy way to dial down a dosage is to have the patient periodically skip a dose). Also, “it is more convenient to prescribe 400 mg daily rather than calculate an exact dosage,” Dr. Koppikar said in an interview. In addition, rheumatologists may be unaware that the prevalence of retinopathy in patients on HCQ is fairly common (about 8% in one large recent study), assessment for risk factors that heighten sensitivity to the drug isn’t always done, and appointments for retinal screening can fall through the cracks.
“It behooves rheumatologists to adopt the [HCQ] recommendations of the American Academy of Ophthalmology [AAO] because there is more toxicity than we previously appreciated,” he added. A new version of the AAO’s recommendations came out in March 2016.
The Committee on Rheumatologic Care of the American College of Rheumatology (ACR) has regularly updated the ACR’s position statement on screening for HCQ retinopathy and appropriate dosages, with the most recent version out in August 2016. The August statement “is very similar” to the AAO’s 2016 recommendations, said Vinicius Domingues, MD, a member of the committee and an ACR spokesman for the revision. The ACR statement acknowledges and cites the AAO 2016 recommendations.
The ACR’s 2016 statement also does not fully endorse the AAO’s 2016 firm statement that “all patients using HCQ keep daily dosage less than 5.0 mg/kg real weight,” aside from “rare instances” when a higher dosage is needed to treat a “life-threatening disease.”
The ACR 2016 statement goes on to note that other authors have recommended a dosage of 6.5 mg/kg of actual body weight but capped at 400 mg/day and adjusted for renal insufficiency, and the ACR statement stops short of specifying which dosage strategy it recommends.
“The AAO recommendations are much more definitive and state more specifically what screening is recommended and what is a safe dosage,” commented Dr. Rosenbaum.
Dr. Domingues agreed that rheumatologist compliance with HCQ best practices has been spotty.
“In the past few years, more studies have used new ways to detect macular abnormalities and have identified a higher-than-expected incidence of maculopathy. Through lectures, CME, and articles, rheumatologists have received a tremendous amount of information with regard to screening and preventing retinal toxicity,” he said in an interview. “There are still gaps, and some rheumatologists still prescribe HCQ without taking into consideration the patient’s weight.”
That was a key finding in the poster presented at the Canadian Rheumatology Association by Dr. Koppikar and his collaborator on the study, Henry Averns, MD. The 100 patients assessed through the nine-clinic audit process averaged 58 years old, 81% were women, and patients had been taking HCQ for an average of just over 6 years, primarily for rheumatoid arthritis or systemic lupus erythematosus. Nearly two-thirds had a high risk for retinal toxicity. Based on the 2011 recommendations from the AAO and ACR, 17% of the patients were receiving an HCQ overdose that was more than 10% above the recommended dosage, and another 13% received a smaller overdose. If the 2016 dosage guidelines were applied, the extent of overdosing might be even greater, Dr. Koppikar said.
Dr. Koppikar and Dr. Averns said they believe that one way to address HCQ overdosing is by giving clinicians a dosing chart to easily find the right dosage for a patient’s weight. They have distributed these charts to the practices they audited and plan to do a follow-up audit to measure the effect of the intervention on HCQ prescribing.
Results from the initial clinical audit showed that “clinicians were not meeting standards, and we needed an intervention [a dosing chart] to implement a change,” Dr. Koppikar said. “Clinical audits are easy to implement, cost effective, and help improve patient care.”
Dr. Koppikar, Dr. Rosenbaum, Dr. Domingues, and Dr. Averns had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Key clinical point:
Major finding: A practice audit showed that 30% of rheumatology patients treated with hydroxychloroquine received an excessive dosage.
Data source: An audit of 100 rheumatology patients seen at any of nine rheumatology clinics in Eastern Ontario, Canada.
Disclosures: Dr. Koppikar, Dr. Rosenbaum, Dr. Domingues, and Dr. Averns had no relevant financial disclosures.
CDC: Greater activity limitations accompany rising arthritis prevalence
The number of adults with arthritis in the United States continues to rise, with the number projected to climb as high as 78 million by the year 2040. Some of the keys to stemming this rising tide are exercise, along with greater knowledge of how to manage symptoms, according to a new report from the Centers for Disease Control and Prevention.
“Arthritis is at an all-time high: more than 54 million people report a diagnosis of it, and alarmingly, more people with arthritis are suffering from it,” said Anne Schuchat, MD, acting director of the CDC, during a conference call regarding the agency’s latest Vital Signs report (MMWR Morb Mortal Wkly Rep. 2017 Mar 7. doi: org/10.15585/mmwr.mm6609e1). “Among adults with arthritis, the percentage whose lives are particularly limited has increased by about 20% since 2002, from about 36% in 2002 to 43% in 2015. We’re seeing this increase independent of aging of the population.”
“Physical activity can be the antidote for many people [and] can actually decrease pain and improve function by almost 40%,” Dr. Schuchat explained. “Right now, one in three adults with arthritis report being inactive [because of] pain or fear of pain or not knowing what exercise is safe for their joints.”
This inactivity can lead to arthritis patients developing other serious chronic conditions, such as heart disease, diabetes and obesity – conditions that all require physical activity in order to properly manage them. Arthritis alone puts an extraordinary financial burden on the domestic health care industry, as direct medical costs associated with the condition total roughly $81 billion per year, according to the CDC. Additionally, about half of all adults with heart disease or diabetes, and about one-third of obese adults, also have arthritis.
In addition to engaging in regular physical activity, the Vital Signs report also recommends that arthritis patients attend disease management education programs, which are available regionally but often go underutilized, largely due to lack of awareness about them or trepidation regarding how effective the programs really are. To combat this, the CDC is calling on health care providers to help them educate patients about these classes and spread the word about the steps that can be taken to manage arthritis. In 2017, the CDC is funding arthritis programs in 12 states (California, Kansas, Kentucky, Michigan, Missouri, Montana, New York, Oregon, Pennsylvania, Rhode Island, South Carolina, and Utah) to disseminate arthritis-appropriate evidence-based physical activity and self-management education interventions.
“Men or women with arthritis can reduce their symptoms by 10%-20% by participating in disease management education programs to acquire skills to better manage their symptoms. Right now, these programs are only reaching about 1 in 10 people with arthritis, but the classes are available in many community settings,” Dr. Schuchat said. “We know that adults with arthritis are significantly more likely to attend a disease management education program when a health care provider recommends it to them.”
When seeing patients with arthritis, Dr. Schuchat advised health care providers to recommend routine physical activity, such as walking, biking, swimming, and physical activity programs offered by local parks and recreation centers, as well as weight loss, in order to ease joint pain. The American College of Rheumatology and other professional organizations provide guidelines for discussing treatment options with patients. Providing treatment or additional services for depression or anxiety, which occur in about one-third of adult arthritis patients, may help individuals to better manage their arthritis symptoms.
The agency’s report derives from its analysis of 2013-2015 data from the National Health Interview Survey, which comprised a nationally representative sample of about 36,000 in-person interviews. The survey classifies individuals with physician-diagnosed arthritis as those who answered “yes” to the question “Have you ever been told by a doctor or other health professional that you have some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia?”
The number of adults with arthritis in the United States continues to rise, with the number projected to climb as high as 78 million by the year 2040. Some of the keys to stemming this rising tide are exercise, along with greater knowledge of how to manage symptoms, according to a new report from the Centers for Disease Control and Prevention.
“Arthritis is at an all-time high: more than 54 million people report a diagnosis of it, and alarmingly, more people with arthritis are suffering from it,” said Anne Schuchat, MD, acting director of the CDC, during a conference call regarding the agency’s latest Vital Signs report (MMWR Morb Mortal Wkly Rep. 2017 Mar 7. doi: org/10.15585/mmwr.mm6609e1). “Among adults with arthritis, the percentage whose lives are particularly limited has increased by about 20% since 2002, from about 36% in 2002 to 43% in 2015. We’re seeing this increase independent of aging of the population.”
“Physical activity can be the antidote for many people [and] can actually decrease pain and improve function by almost 40%,” Dr. Schuchat explained. “Right now, one in three adults with arthritis report being inactive [because of] pain or fear of pain or not knowing what exercise is safe for their joints.”
This inactivity can lead to arthritis patients developing other serious chronic conditions, such as heart disease, diabetes and obesity – conditions that all require physical activity in order to properly manage them. Arthritis alone puts an extraordinary financial burden on the domestic health care industry, as direct medical costs associated with the condition total roughly $81 billion per year, according to the CDC. Additionally, about half of all adults with heart disease or diabetes, and about one-third of obese adults, also have arthritis.
In addition to engaging in regular physical activity, the Vital Signs report also recommends that arthritis patients attend disease management education programs, which are available regionally but often go underutilized, largely due to lack of awareness about them or trepidation regarding how effective the programs really are. To combat this, the CDC is calling on health care providers to help them educate patients about these classes and spread the word about the steps that can be taken to manage arthritis. In 2017, the CDC is funding arthritis programs in 12 states (California, Kansas, Kentucky, Michigan, Missouri, Montana, New York, Oregon, Pennsylvania, Rhode Island, South Carolina, and Utah) to disseminate arthritis-appropriate evidence-based physical activity and self-management education interventions.
“Men or women with arthritis can reduce their symptoms by 10%-20% by participating in disease management education programs to acquire skills to better manage their symptoms. Right now, these programs are only reaching about 1 in 10 people with arthritis, but the classes are available in many community settings,” Dr. Schuchat said. “We know that adults with arthritis are significantly more likely to attend a disease management education program when a health care provider recommends it to them.”
When seeing patients with arthritis, Dr. Schuchat advised health care providers to recommend routine physical activity, such as walking, biking, swimming, and physical activity programs offered by local parks and recreation centers, as well as weight loss, in order to ease joint pain. The American College of Rheumatology and other professional organizations provide guidelines for discussing treatment options with patients. Providing treatment or additional services for depression or anxiety, which occur in about one-third of adult arthritis patients, may help individuals to better manage their arthritis symptoms.
The agency’s report derives from its analysis of 2013-2015 data from the National Health Interview Survey, which comprised a nationally representative sample of about 36,000 in-person interviews. The survey classifies individuals with physician-diagnosed arthritis as those who answered “yes” to the question “Have you ever been told by a doctor or other health professional that you have some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia?”
The number of adults with arthritis in the United States continues to rise, with the number projected to climb as high as 78 million by the year 2040. Some of the keys to stemming this rising tide are exercise, along with greater knowledge of how to manage symptoms, according to a new report from the Centers for Disease Control and Prevention.
“Arthritis is at an all-time high: more than 54 million people report a diagnosis of it, and alarmingly, more people with arthritis are suffering from it,” said Anne Schuchat, MD, acting director of the CDC, during a conference call regarding the agency’s latest Vital Signs report (MMWR Morb Mortal Wkly Rep. 2017 Mar 7. doi: org/10.15585/mmwr.mm6609e1). “Among adults with arthritis, the percentage whose lives are particularly limited has increased by about 20% since 2002, from about 36% in 2002 to 43% in 2015. We’re seeing this increase independent of aging of the population.”
“Physical activity can be the antidote for many people [and] can actually decrease pain and improve function by almost 40%,” Dr. Schuchat explained. “Right now, one in three adults with arthritis report being inactive [because of] pain or fear of pain or not knowing what exercise is safe for their joints.”
This inactivity can lead to arthritis patients developing other serious chronic conditions, such as heart disease, diabetes and obesity – conditions that all require physical activity in order to properly manage them. Arthritis alone puts an extraordinary financial burden on the domestic health care industry, as direct medical costs associated with the condition total roughly $81 billion per year, according to the CDC. Additionally, about half of all adults with heart disease or diabetes, and about one-third of obese adults, also have arthritis.
In addition to engaging in regular physical activity, the Vital Signs report also recommends that arthritis patients attend disease management education programs, which are available regionally but often go underutilized, largely due to lack of awareness about them or trepidation regarding how effective the programs really are. To combat this, the CDC is calling on health care providers to help them educate patients about these classes and spread the word about the steps that can be taken to manage arthritis. In 2017, the CDC is funding arthritis programs in 12 states (California, Kansas, Kentucky, Michigan, Missouri, Montana, New York, Oregon, Pennsylvania, Rhode Island, South Carolina, and Utah) to disseminate arthritis-appropriate evidence-based physical activity and self-management education interventions.
“Men or women with arthritis can reduce their symptoms by 10%-20% by participating in disease management education programs to acquire skills to better manage their symptoms. Right now, these programs are only reaching about 1 in 10 people with arthritis, but the classes are available in many community settings,” Dr. Schuchat said. “We know that adults with arthritis are significantly more likely to attend a disease management education program when a health care provider recommends it to them.”
When seeing patients with arthritis, Dr. Schuchat advised health care providers to recommend routine physical activity, such as walking, biking, swimming, and physical activity programs offered by local parks and recreation centers, as well as weight loss, in order to ease joint pain. The American College of Rheumatology and other professional organizations provide guidelines for discussing treatment options with patients. Providing treatment or additional services for depression or anxiety, which occur in about one-third of adult arthritis patients, may help individuals to better manage their arthritis symptoms.
The agency’s report derives from its analysis of 2013-2015 data from the National Health Interview Survey, which comprised a nationally representative sample of about 36,000 in-person interviews. The survey classifies individuals with physician-diagnosed arthritis as those who answered “yes” to the question “Have you ever been told by a doctor or other health professional that you have some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia?”
FROM MMWR
Key clinical point:
Major finding: About 24 million American adults report being significantly limited due to their arthritis.
Data source: 2013-2015 data from the National Health Interview Survey.
Disclosures: No disclosures were reported.
Biosimilars: No big dollar savings, but are clinically ‘dead on’
SNOWMASS, COLO. – If you thought biosimilars would bring sharply reduced pricing compared with their parent agents, with resultant greater patient access to highly effective therapies for rheumatic diseases ... think again.
“The promise to our patients of biosimilars – greater access to treatments – is something I think we’re just not going to see, at least not here in the U.S.,” Michael E. Weinblatt, MD, declared at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
In contrast, the safety and efficacy of the biosimilars, as well as their interchangeability with their reference products, appear to be as hoped for. At the 2016 annual meeting of the American College of Rheumatology, Dr. Weinblatt presented the week 24 results of a phase III, randomized trial involving rheumatoid arthritis patients on background methotrexate plus either adalimumab (Humira) or its biosimilar SB5.
“Essentially, they’re dead on in clinical response, they’re dead on in antibody levels, and they’re dead on in toxicity. And, you can put any of the biosimilars up there and the results are the same. If they get approved, this is what you’re going to see,” the rheumatologist said.
Also at the 2016 ACR annual meeting, he noted, Danish investigators presented reassuring 1-year follow-up data on 802 Danes with inflammatory rheumatic diseases who switched from infliximab (Remicade) to its biosimilar Remsima. Disease activity and flare rates in the year following the switch were similar to those in the year before. The 1-year rate of adherence to Remsima was 84%, similar to the historical 86% 1-year rate with infliximab.
“So, I’m pretty comfortable with the biosimilars,” Dr. Weinblatt continued.
He observed that, of all the systemic rheumatic diseases, the greatest progress has occurred in the treatment of rheumatoid arthritis.
“We have made great advances in the treatment of this disease, unlike many of our other diseases. Methotrexate and combination therapies with small molecules and biologics has dramatically changed the course of the disease,” he noted. “The greatest challenge we have now as rheumatologists is access barriers for our patients.”
Dr. Weinblatt reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
SNOWMASS, COLO. – If you thought biosimilars would bring sharply reduced pricing compared with their parent agents, with resultant greater patient access to highly effective therapies for rheumatic diseases ... think again.
“The promise to our patients of biosimilars – greater access to treatments – is something I think we’re just not going to see, at least not here in the U.S.,” Michael E. Weinblatt, MD, declared at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
In contrast, the safety and efficacy of the biosimilars, as well as their interchangeability with their reference products, appear to be as hoped for. At the 2016 annual meeting of the American College of Rheumatology, Dr. Weinblatt presented the week 24 results of a phase III, randomized trial involving rheumatoid arthritis patients on background methotrexate plus either adalimumab (Humira) or its biosimilar SB5.
“Essentially, they’re dead on in clinical response, they’re dead on in antibody levels, and they’re dead on in toxicity. And, you can put any of the biosimilars up there and the results are the same. If they get approved, this is what you’re going to see,” the rheumatologist said.
Also at the 2016 ACR annual meeting, he noted, Danish investigators presented reassuring 1-year follow-up data on 802 Danes with inflammatory rheumatic diseases who switched from infliximab (Remicade) to its biosimilar Remsima. Disease activity and flare rates in the year following the switch were similar to those in the year before. The 1-year rate of adherence to Remsima was 84%, similar to the historical 86% 1-year rate with infliximab.
“So, I’m pretty comfortable with the biosimilars,” Dr. Weinblatt continued.
He observed that, of all the systemic rheumatic diseases, the greatest progress has occurred in the treatment of rheumatoid arthritis.
“We have made great advances in the treatment of this disease, unlike many of our other diseases. Methotrexate and combination therapies with small molecules and biologics has dramatically changed the course of the disease,” he noted. “The greatest challenge we have now as rheumatologists is access barriers for our patients.”
Dr. Weinblatt reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
SNOWMASS, COLO. – If you thought biosimilars would bring sharply reduced pricing compared with their parent agents, with resultant greater patient access to highly effective therapies for rheumatic diseases ... think again.
“The promise to our patients of biosimilars – greater access to treatments – is something I think we’re just not going to see, at least not here in the U.S.,” Michael E. Weinblatt, MD, declared at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
In contrast, the safety and efficacy of the biosimilars, as well as their interchangeability with their reference products, appear to be as hoped for. At the 2016 annual meeting of the American College of Rheumatology, Dr. Weinblatt presented the week 24 results of a phase III, randomized trial involving rheumatoid arthritis patients on background methotrexate plus either adalimumab (Humira) or its biosimilar SB5.
“Essentially, they’re dead on in clinical response, they’re dead on in antibody levels, and they’re dead on in toxicity. And, you can put any of the biosimilars up there and the results are the same. If they get approved, this is what you’re going to see,” the rheumatologist said.
Also at the 2016 ACR annual meeting, he noted, Danish investigators presented reassuring 1-year follow-up data on 802 Danes with inflammatory rheumatic diseases who switched from infliximab (Remicade) to its biosimilar Remsima. Disease activity and flare rates in the year following the switch were similar to those in the year before. The 1-year rate of adherence to Remsima was 84%, similar to the historical 86% 1-year rate with infliximab.
“So, I’m pretty comfortable with the biosimilars,” Dr. Weinblatt continued.
He observed that, of all the systemic rheumatic diseases, the greatest progress has occurred in the treatment of rheumatoid arthritis.
“We have made great advances in the treatment of this disease, unlike many of our other diseases. Methotrexate and combination therapies with small molecules and biologics has dramatically changed the course of the disease,” he noted. “The greatest challenge we have now as rheumatologists is access barriers for our patients.”
Dr. Weinblatt reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
EXPERT ANALYSIS FROM THE WINTER RHEUMATOLOGY SYMPOSIUM
Chikungunya implicated in long-term joint disease
A majority of Chikungunya infections can cause arthritis and arthralgia months or years after the initial infection, based on data from a prospective study of 307 patients.
“The most common symptoms of Chikungunya virus infection are fever associated with rheumatic manifestations,” wrote rheumatologist Eric Bouquillard, MD, of Saint-Pierre, Reunion, France, and his colleagues.
Overall, 83% of the patients showed persistent joint pain after an average of 32 months. In addition, synovitis occurred in 64% of the patients who experienced chronic joint pain, mainly in the wrists, fingers, and ankles.
At baseline, the average number of painful joints was 6.5. At follow-up, the average number of painful joints was 3.3, and 43% of patients reported persistence of one or more swollen joints.
However, the patients reported little functional impairment; the average Health Assessment Questionnaire score was 0.44.
“RT-PCR [reverse transcription–polymerase chain reaction] was used in an attempt to detect the viral genome in synovial fluid samples from 10 patients, including 2 patients in the viremic phase, but the results were always negative,” the researchers noted.
Dr. Bouquillard and his colleagues enrolled the patients during April 2005-December 2006. Rheumatologic exams were conducted at baseline, and follow-up data were collected by phone surveys at 1 and 2 years after the onset of Chikungunya infection. Phone surveys were conducted by the Reunion Island Clinical Investigation Centre for Clinical Epidemiology, and interviewers also assessed patients for signs of anxiety, depression, and weakness.
The study was not designed to address treatment, but data from previous studies suggest that combination disease-modifying antirheumatic drug therapy may be more effective than hydroxychloroquine monotherapy for chronic joint pain post Chikungunya, the researchers noted.
The researchers had no financial conflicts to disclose. The study was supported in part by the Union Régionale des Médecins Libéraux de La Réunion.
A majority of Chikungunya infections can cause arthritis and arthralgia months or years after the initial infection, based on data from a prospective study of 307 patients.
“The most common symptoms of Chikungunya virus infection are fever associated with rheumatic manifestations,” wrote rheumatologist Eric Bouquillard, MD, of Saint-Pierre, Reunion, France, and his colleagues.
Overall, 83% of the patients showed persistent joint pain after an average of 32 months. In addition, synovitis occurred in 64% of the patients who experienced chronic joint pain, mainly in the wrists, fingers, and ankles.
At baseline, the average number of painful joints was 6.5. At follow-up, the average number of painful joints was 3.3, and 43% of patients reported persistence of one or more swollen joints.
However, the patients reported little functional impairment; the average Health Assessment Questionnaire score was 0.44.
“RT-PCR [reverse transcription–polymerase chain reaction] was used in an attempt to detect the viral genome in synovial fluid samples from 10 patients, including 2 patients in the viremic phase, but the results were always negative,” the researchers noted.
Dr. Bouquillard and his colleagues enrolled the patients during April 2005-December 2006. Rheumatologic exams were conducted at baseline, and follow-up data were collected by phone surveys at 1 and 2 years after the onset of Chikungunya infection. Phone surveys were conducted by the Reunion Island Clinical Investigation Centre for Clinical Epidemiology, and interviewers also assessed patients for signs of anxiety, depression, and weakness.
The study was not designed to address treatment, but data from previous studies suggest that combination disease-modifying antirheumatic drug therapy may be more effective than hydroxychloroquine monotherapy for chronic joint pain post Chikungunya, the researchers noted.
The researchers had no financial conflicts to disclose. The study was supported in part by the Union Régionale des Médecins Libéraux de La Réunion.
A majority of Chikungunya infections can cause arthritis and arthralgia months or years after the initial infection, based on data from a prospective study of 307 patients.
“The most common symptoms of Chikungunya virus infection are fever associated with rheumatic manifestations,” wrote rheumatologist Eric Bouquillard, MD, of Saint-Pierre, Reunion, France, and his colleagues.
Overall, 83% of the patients showed persistent joint pain after an average of 32 months. In addition, synovitis occurred in 64% of the patients who experienced chronic joint pain, mainly in the wrists, fingers, and ankles.
At baseline, the average number of painful joints was 6.5. At follow-up, the average number of painful joints was 3.3, and 43% of patients reported persistence of one or more swollen joints.
However, the patients reported little functional impairment; the average Health Assessment Questionnaire score was 0.44.
“RT-PCR [reverse transcription–polymerase chain reaction] was used in an attempt to detect the viral genome in synovial fluid samples from 10 patients, including 2 patients in the viremic phase, but the results were always negative,” the researchers noted.
Dr. Bouquillard and his colleagues enrolled the patients during April 2005-December 2006. Rheumatologic exams were conducted at baseline, and follow-up data were collected by phone surveys at 1 and 2 years after the onset of Chikungunya infection. Phone surveys were conducted by the Reunion Island Clinical Investigation Centre for Clinical Epidemiology, and interviewers also assessed patients for signs of anxiety, depression, and weakness.
The study was not designed to address treatment, but data from previous studies suggest that combination disease-modifying antirheumatic drug therapy may be more effective than hydroxychloroquine monotherapy for chronic joint pain post Chikungunya, the researchers noted.
The researchers had no financial conflicts to disclose. The study was supported in part by the Union Régionale des Médecins Libéraux de La Réunion.
FROM JOINT BONE SPINE
Key clinical point:
Major finding: Approximately 83% of adults with Chikungunya virus infections reported persistent joint pain after an average of 32 months.
Data source: A prospective, multicenter study of 307 adults with a history of Chikungunya virus infections.
Disclosures: The researchers had no financial conflicts to disclose. The study was supported in part by the Union Régionale des Médecins Libéraux de La Réunion.
Humira Pen topped per-person drug spending in 2016
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”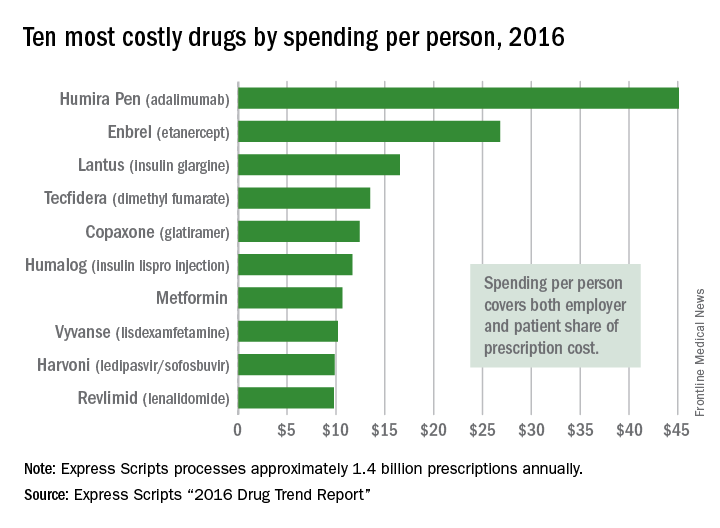
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
Sirukumab found effective, safe for highly refractory RA
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
FROM THE LANCET
Key clinical point: Sirukumab proved effective and safe for RA patients who failed to respond to or were intolerant of multiple previous therapies.
Key numerical finding: The primary efficacy endpoint – the proportion of patients achieving an ACR20 response at week 16 – was 40% for low-dose and 45% for high-dose sirukumab, compared with 24% for placebo.
Data source: A manufacturer-sponsored, international, randomized, double-blind, placebo-controlled, phase III trial involving 878 adults with refractory RA.
Disclosures: This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing of the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
Survey highlights interest in diet’s effects on RA
Nearly one-quarter of patients with long-standing rheumatoid arthritis who participated in a recent survey reported that their diets affect their RA symptoms.
Of 217 participants with a median disease duration of 17 years, 52 (24%) reported that certain foods either improve or worsen symptoms. Foods most commonly associated with improved symptoms were blueberries (11.1%), fish (10.9%), and spinach; foods most commonly associated with exacerbated symptoms were desserts (12.7%) and soda with sugar (12.4%, ), Sara K. Tedeschi, MD, of Brigham and Women’s Hospital, Boston, and her colleagues reported online in Arthritis Care & Research.
Participants came from a single-center RA registry (the Brigham RA Sequential Study, or BRASS) at a large academic center and were surveyed between May 2015 and December 2015. They were asked about the effects of 20 different foods that have been popularized as “inflammatory” or “anti-inflammatory” and about the effects of four lifestyle/environment factors. Most (83%) were women, and 58% were using a biologic disease-modifying antirheumatic drug.
The findings indicate that there is substantial patient interest in the effects of diet on RA symptoms and highlight the need for prospective studies on the topic, the investigators concluded. While strong conclusions cannot be drawn based on this survey, further study regarding a potential link between sugar consumption and inflammation is warranted.
Dr. Tedeschi’s work on this project was supported by the National Institutes of Health. The Brigham RA Sequential Study received funding from UCB, Crescendo Biosciences, Bristol-Myers Squibb, Amgen, and DxTerity.
Nearly one-quarter of patients with long-standing rheumatoid arthritis who participated in a recent survey reported that their diets affect their RA symptoms.
Of 217 participants with a median disease duration of 17 years, 52 (24%) reported that certain foods either improve or worsen symptoms. Foods most commonly associated with improved symptoms were blueberries (11.1%), fish (10.9%), and spinach; foods most commonly associated with exacerbated symptoms were desserts (12.7%) and soda with sugar (12.4%, ), Sara K. Tedeschi, MD, of Brigham and Women’s Hospital, Boston, and her colleagues reported online in Arthritis Care & Research.
Participants came from a single-center RA registry (the Brigham RA Sequential Study, or BRASS) at a large academic center and were surveyed between May 2015 and December 2015. They were asked about the effects of 20 different foods that have been popularized as “inflammatory” or “anti-inflammatory” and about the effects of four lifestyle/environment factors. Most (83%) were women, and 58% were using a biologic disease-modifying antirheumatic drug.
The findings indicate that there is substantial patient interest in the effects of diet on RA symptoms and highlight the need for prospective studies on the topic, the investigators concluded. While strong conclusions cannot be drawn based on this survey, further study regarding a potential link between sugar consumption and inflammation is warranted.
Dr. Tedeschi’s work on this project was supported by the National Institutes of Health. The Brigham RA Sequential Study received funding from UCB, Crescendo Biosciences, Bristol-Myers Squibb, Amgen, and DxTerity.
Nearly one-quarter of patients with long-standing rheumatoid arthritis who participated in a recent survey reported that their diets affect their RA symptoms.
Of 217 participants with a median disease duration of 17 years, 52 (24%) reported that certain foods either improve or worsen symptoms. Foods most commonly associated with improved symptoms were blueberries (11.1%), fish (10.9%), and spinach; foods most commonly associated with exacerbated symptoms were desserts (12.7%) and soda with sugar (12.4%, ), Sara K. Tedeschi, MD, of Brigham and Women’s Hospital, Boston, and her colleagues reported online in Arthritis Care & Research.
Participants came from a single-center RA registry (the Brigham RA Sequential Study, or BRASS) at a large academic center and were surveyed between May 2015 and December 2015. They were asked about the effects of 20 different foods that have been popularized as “inflammatory” or “anti-inflammatory” and about the effects of four lifestyle/environment factors. Most (83%) were women, and 58% were using a biologic disease-modifying antirheumatic drug.
The findings indicate that there is substantial patient interest in the effects of diet on RA symptoms and highlight the need for prospective studies on the topic, the investigators concluded. While strong conclusions cannot be drawn based on this survey, further study regarding a potential link between sugar consumption and inflammation is warranted.
Dr. Tedeschi’s work on this project was supported by the National Institutes of Health. The Brigham RA Sequential Study received funding from UCB, Crescendo Biosciences, Bristol-Myers Squibb, Amgen, and DxTerity.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: 24% of respondents reported that diet affects RA symptoms.
Data source: A survey of 217 participants in the Brigham RA Sequential Study.
Disclosures: Dr. Tedeschi’s work on this project was supported by the National Institutes of Health. The Brigham RA Sequential Study received funding from UCB, Crescendo Biosciences, Bristol-Myers Squibb, Amgen, and DxTerity.
Pilot trial of first in kind biologic shows RA treatment potential
The novel biologic mavrilimumab that targets the GM-CSF pathway has shown therapeutic potential in the treatment of rheumatoid arthritis in a proof-of-concept trial, particularly in patients who have failed to respond to biologics that target other pathways.
Writing in the Annals of the Rheumatic Diseases, researchers led by Gerd R. Burmester, MD, director and professor of medicine in the department of rheumatology and clinical immunology at Charité University Hospital and the Free University and Humboldt University of Berlin, noted that despite the success of the currently available biologics to treat rheumatoid arthritis (RA), a considerable number of patients do not achieve long-term responses (Ann Rheum Dis. 2017 Feb 17. doi: 10.1136/annrheumdis-2016-210624).
Mavrilimumab is a fully human monoclonal antibody that blocks GM-CSFR and is the first biologic of its kind to target the GM-CSF pathway, they noted.
The multicenter, phase IIb, randomized trial EARTH EXPLORER 1 involved 305 patients with moderate to severe RA who were randomized in a ratio of 1:1:1:1 to subcutaneous mavrilimumab 150 mg, 100 mg, 30 mg, or placebo every other week plus methotrexate for 24 weeks.
The results showed that the GM-CSFR blocker met one of its primary endpoints by significantly reducing the 28-joint Disease Activity Score based on C-reactive protein (DAS28–CRP) from baseline to week 12 when compared with placebo. The amount of reduction increased with the dose, from –1.37 with 30 mg, to –1.64 with 100 mg, to –1.90 with 150 mg, compared with –0.68 with placebo (P less than .001 for all vs. placebo).
The biologic also met its other primary endpoint of achieving ACR20 level of response by 24 weeks in significantly more patients than with placebo: 73.4% for 150 mg, 61.2% for 100 mg, and 50.6% for 30 mg vs. 24.7% for placebo (P less than .001).
Adverse events were reported in all treatment dose groups (42%-54% of patients in each group), and no treatment-related safety signals were observed. Only one case each of pneumonia and angioedema were considered to be related to treatment by the investigators.
The research team suggested that the blocking of GM-CSF signaling could be applicable to patients who have failed treatment with biologics that target other pathways or for people with other inflammatory or autoimmune diseases.
“This proof-of-concept study confirms that inhibition of GM-CSF activity is a promising and novel therapeutic approach for patients with RA, including those who do not adequately respond to currently available therapies,” they concluded.
The study was funded by AstraZeneca/MedImmune. Several of the authors are employees of MedImmune and several reported financial ties to other pharmaceutical companies.
The novel biologic mavrilimumab that targets the GM-CSF pathway has shown therapeutic potential in the treatment of rheumatoid arthritis in a proof-of-concept trial, particularly in patients who have failed to respond to biologics that target other pathways.
Writing in the Annals of the Rheumatic Diseases, researchers led by Gerd R. Burmester, MD, director and professor of medicine in the department of rheumatology and clinical immunology at Charité University Hospital and the Free University and Humboldt University of Berlin, noted that despite the success of the currently available biologics to treat rheumatoid arthritis (RA), a considerable number of patients do not achieve long-term responses (Ann Rheum Dis. 2017 Feb 17. doi: 10.1136/annrheumdis-2016-210624).
Mavrilimumab is a fully human monoclonal antibody that blocks GM-CSFR and is the first biologic of its kind to target the GM-CSF pathway, they noted.
The multicenter, phase IIb, randomized trial EARTH EXPLORER 1 involved 305 patients with moderate to severe RA who were randomized in a ratio of 1:1:1:1 to subcutaneous mavrilimumab 150 mg, 100 mg, 30 mg, or placebo every other week plus methotrexate for 24 weeks.
The results showed that the GM-CSFR blocker met one of its primary endpoints by significantly reducing the 28-joint Disease Activity Score based on C-reactive protein (DAS28–CRP) from baseline to week 12 when compared with placebo. The amount of reduction increased with the dose, from –1.37 with 30 mg, to –1.64 with 100 mg, to –1.90 with 150 mg, compared with –0.68 with placebo (P less than .001 for all vs. placebo).
The biologic also met its other primary endpoint of achieving ACR20 level of response by 24 weeks in significantly more patients than with placebo: 73.4% for 150 mg, 61.2% for 100 mg, and 50.6% for 30 mg vs. 24.7% for placebo (P less than .001).
Adverse events were reported in all treatment dose groups (42%-54% of patients in each group), and no treatment-related safety signals were observed. Only one case each of pneumonia and angioedema were considered to be related to treatment by the investigators.
The research team suggested that the blocking of GM-CSF signaling could be applicable to patients who have failed treatment with biologics that target other pathways or for people with other inflammatory or autoimmune diseases.
“This proof-of-concept study confirms that inhibition of GM-CSF activity is a promising and novel therapeutic approach for patients with RA, including those who do not adequately respond to currently available therapies,” they concluded.
The study was funded by AstraZeneca/MedImmune. Several of the authors are employees of MedImmune and several reported financial ties to other pharmaceutical companies.
The novel biologic mavrilimumab that targets the GM-CSF pathway has shown therapeutic potential in the treatment of rheumatoid arthritis in a proof-of-concept trial, particularly in patients who have failed to respond to biologics that target other pathways.
Writing in the Annals of the Rheumatic Diseases, researchers led by Gerd R. Burmester, MD, director and professor of medicine in the department of rheumatology and clinical immunology at Charité University Hospital and the Free University and Humboldt University of Berlin, noted that despite the success of the currently available biologics to treat rheumatoid arthritis (RA), a considerable number of patients do not achieve long-term responses (Ann Rheum Dis. 2017 Feb 17. doi: 10.1136/annrheumdis-2016-210624).
Mavrilimumab is a fully human monoclonal antibody that blocks GM-CSFR and is the first biologic of its kind to target the GM-CSF pathway, they noted.
The multicenter, phase IIb, randomized trial EARTH EXPLORER 1 involved 305 patients with moderate to severe RA who were randomized in a ratio of 1:1:1:1 to subcutaneous mavrilimumab 150 mg, 100 mg, 30 mg, or placebo every other week plus methotrexate for 24 weeks.
The results showed that the GM-CSFR blocker met one of its primary endpoints by significantly reducing the 28-joint Disease Activity Score based on C-reactive protein (DAS28–CRP) from baseline to week 12 when compared with placebo. The amount of reduction increased with the dose, from –1.37 with 30 mg, to –1.64 with 100 mg, to –1.90 with 150 mg, compared with –0.68 with placebo (P less than .001 for all vs. placebo).
The biologic also met its other primary endpoint of achieving ACR20 level of response by 24 weeks in significantly more patients than with placebo: 73.4% for 150 mg, 61.2% for 100 mg, and 50.6% for 30 mg vs. 24.7% for placebo (P less than .001).
Adverse events were reported in all treatment dose groups (42%-54% of patients in each group), and no treatment-related safety signals were observed. Only one case each of pneumonia and angioedema were considered to be related to treatment by the investigators.
The research team suggested that the blocking of GM-CSF signaling could be applicable to patients who have failed treatment with biologics that target other pathways or for people with other inflammatory or autoimmune diseases.
“This proof-of-concept study confirms that inhibition of GM-CSF activity is a promising and novel therapeutic approach for patients with RA, including those who do not adequately respond to currently available therapies,” they concluded.
The study was funded by AstraZeneca/MedImmune. Several of the authors are employees of MedImmune and several reported financial ties to other pharmaceutical companies.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point:
Major finding: Mavrilimumab significantly reduced DAS28–CRP scores from baseline to week 12 when compared with placebo, and the mount of reduction increased with the dose, from –1.37 with 30 mg, to –1.64 with 100 mg, to –1.90 with 150 mg, compared with –0.68 with placebo (P less than .001 for all vs. placebo).
Data source: A multicenter, randomized, double-blind, placebo-controlled, phase IIb trial of 305 RA patients with moderate to severe RA.
Disclosures: The study was funded by AstraZeneca/MedImmune. Several of the authors are employees of MedImmune and several reported financial ties to other pharmaceutical companies.
Methotrexate for RA: A 'fascinating drug’
SNOWMASS, COLO. – “When I started working with methotrexate in 1982, I never would have predicted that methotrexate would become the standard of care in treating rheumatoid arthritis. There’s just no way,” Michael E. Weinblatt, MD, recalled at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“Even now, 35 years later, we continue to learn more about this fascinating drug,” added Dr. Weinblatt, professor of medicine at Harvard Medical School, Boston.
He highlighted recent developments in this ongoing story and presented some tricks of the trade gained in 35 years of up-close experience with the drug.
Enhancing effectiveness of biologics
One of the hottest topics in methotrexate research is the drug’s ability to enhance the effectiveness of many, but not all, biologic agents. All of the anti–tumor necrosis factor (anti-TNF) biologics as well as rituximab (Rituxan) are demonstrably more effective when used in combination with methotrexate. Dr. Weinblatt considers the widespread underutilization of this combination strategy scandalous.
“This is an incredibly important point: All of you use biologics and all of you use methotrexate, but I’ve been depressed by the fact that up to 30%-40% of patients – no matter which data set you look at – are on monotherapy biological therapy,” he said.
He cited data from the ongoing BRASS Registry (Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Registry) to underscore his point that good things happen when biologics and methotrexate are used together. Of 1,395 BRASS Registry participants prospectively followed since 2003, the proportion on biologic therapy has climbed steadily from 41% at the outset to 68% in 2016. Remarkably, 82% of patients on biologic therapy remain on their first biologic agent. Fewer than 4% have switched biologics more than twice. That’s very unlike the experiences reported elsewhere.
“I think one of the reasons we have such positive data is that we have a high percentage of patients staying on their background methotrexate,” said Dr. Weinblatt, codirector of clinical rheumatology and associate director of the Center for Arthritis and Joint Diseases at Brigham and Women’s Hospital, Boston.
He noted that Dutch investigators reported at the 2016 annual meeting of the American College of Rheumatology that among 1,230 consecutive rheumatoid arthritis patients started on etanercept (Enbrel) or adalimumab (Humira), 28% in the etanercept group were on concomitant methotrexate, as were 64% of those who started on adalimumab. The patients spent a median of 1.3-1.6 years and a maximum of 9.2-9.3 years on their biologic agent. Patients on adalimumab monotherapy were 2.61-fold more likely to drop out than were those on dual therapy with methotrexate. Patients on etanercept monotherapy were 1.2-fold more likely to drop out, a difference that didn’t achieve statistical significance.
Although the investigators did not study the mechanism of prolonged on-treatment survival, they speculated that it probably involved methotrexate’s documented ability to prevent formation of anti-adalimumab antibodies. In contrast, patients on etanercept don’t develop blocking antibodies, Dr. Weinblatt observed.
The randomized, double-blind CONCERTO trial conducted in 395 methotrexate- and biologic-naive RA patients demonstrated that methotrexate reduces the immunogenicity of adalimumab in dose-dependent fashion. Participants were randomized to open-label adalimumab at 40 mg every 2 weeks plus weekly double-blind methotrexate at 2.5, 5, 10, or 20 mg. Clinical outcomes at 26 weeks as reflected in 28-joint count disease activity score and the Clinical Disease Activity Index were significantly better in patients on 10 or 20 mg/week of methotrexate than in those on 2.5 or 5 mg/week. Serum adalimumab levels were higher in patients on the two higher doses of methotrexate as well (Ann Rheum Dis. 2015 Jun;74[6]:1037-44).
“It ends up that if you don’t use methotrexate or you use a very low dose you increase the risk of developing antibodies against adalimumab and decrease the efficacy of the drug. So in clinical practice, if you’re going to be working with dose titration of methotrexate and your patient is on adalimumab, there’s a threshold below which you probably shouldn’t go. In this study, doses of 10 mg/week or more induced a greater clinical response,” he said.
With infliximab (Remicade), based upon 20-year-old studies, the threshold is 7.5 mg of methotrexate per week.
“With etanercept, we don’t know what the threshold is. You don’t develop blocking anti-drug antibodies with etanercept, but we do know that methotrexate enhances the efficacy of etanercept, and it doesn’t do it by changing the biologic’s pharmacokinetics and there’s no increase in methotrexate blood levels,” the rheumatologist continued.
Unlike the anti-TNF biologics and rituximab, the efficacy of the Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib is not enhanced when the drugs are used in combination with methotrexate, studies indicate.
The efficacy of certolizumab pegol (Cimzia) wasn’t affected by methotrexate dose category in a prespecified pooled subgroup analysis of the phase III RAPID 1 and RAPID 2 clinical trials. In the 1,273 certolizumab-treated patients, the week-24 treatment response was similar regardless of whether patients were on methotrexate at 10 mg/week or less, 10-15 mg/week, or more than 15 mg/week. The investigators concluded that to minimize treatment-emergent adverse events, physicians can tailor background methotrexate dosing based upon individual patient tolerance without affecting certolizumab’s efficacy (Arthritis Care Res [Hoboken]. 2016 Mar;68[3]:299-307).
An important aspect of this analysis was that among the 325 subjects randomized to placebo rather than certolizumab, the treatment response at week 24 was significantly better in those on more than 15 mg/week of methotrexate than with lower doses of the drug.
“Most patients on methotrexate need more than 15 mg/week. So it astonishes me that such a high percentage of patients enrolled in clinical trials around the world are on, like, 14 mg/week. I mean, most patients need somewhere between 15 and 25 mg/week for a response, although over time you might be able to decrease that dose,” Dr. Weinblatt said.
Side effects of methotrexate
“The biggest issue with methotrexate is the tolerability problem, since serious adverse events are incredibly rare with this molecule,” he said.
Hepatotoxicity is a concern, but Dr. Weinblatt emphasized that elevated liver function tests do not equal cirrhosis.
“Historically, during the first 6 months on methotrexate 20%-25% of patients increase their transaminases in every clinical trial where that’s been looked at. Over time, the liver compensates for the drug. But 5%-6% of patients experience repeated moderate elevations more than 1.5 times the upper limit of normal,” he said.
Key risk factors for methotrexate-related hepatotoxicity were identified in a national observational cohort study of 659 military veterans over age 65 when they started methotrexate for rheumatic diseases. The investigators found a 6% incidence of moderately elevated liver enzymes during a mean follow-up period of 7 months. Obesity was associated with a 1.9-fold increased risk, a serum total cholesterol greater than 240 mg/dL conferred a 5.8-fold elevated risk, and abnormal liver function tests at baseline were associated with a 3.2-fold increased risk (Arthritis Care Res [Hoboken]. 2014 Aug;66[8]:1159-66).
“No surprise: It’s patients who weigh more who are at increased risk for methotrexate-related transaminase increases. I actually think the biggest factor with regard to methotrexate liver disease is the patient’s [body mass index]. Patients in North America aren’t getting any slimmer, so you need to look at this with your patients. If you have a morbidly obese patient on methotrexate whose transaminases suddenly start going up, that’s the patient who’s at greatest risk for methotrexate hepatotoxicity,” he cautioned.
The 3.2-fold increased risk of repeated elevated transaminases associated with abnormal baseline liver function tests in the Veterans Affairs study should be a red flag for rheumatologists.
“I personally think patients shouldn’t start on methotrexate if they have elevated transaminases. They ought to be normal at the start. There are too many other good options now to treat our patients,” Dr. Weinblatt said.
He reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
SNOWMASS, COLO. – “When I started working with methotrexate in 1982, I never would have predicted that methotrexate would become the standard of care in treating rheumatoid arthritis. There’s just no way,” Michael E. Weinblatt, MD, recalled at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“Even now, 35 years later, we continue to learn more about this fascinating drug,” added Dr. Weinblatt, professor of medicine at Harvard Medical School, Boston.
He highlighted recent developments in this ongoing story and presented some tricks of the trade gained in 35 years of up-close experience with the drug.
Enhancing effectiveness of biologics
One of the hottest topics in methotrexate research is the drug’s ability to enhance the effectiveness of many, but not all, biologic agents. All of the anti–tumor necrosis factor (anti-TNF) biologics as well as rituximab (Rituxan) are demonstrably more effective when used in combination with methotrexate. Dr. Weinblatt considers the widespread underutilization of this combination strategy scandalous.
“This is an incredibly important point: All of you use biologics and all of you use methotrexate, but I’ve been depressed by the fact that up to 30%-40% of patients – no matter which data set you look at – are on monotherapy biological therapy,” he said.
He cited data from the ongoing BRASS Registry (Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Registry) to underscore his point that good things happen when biologics and methotrexate are used together. Of 1,395 BRASS Registry participants prospectively followed since 2003, the proportion on biologic therapy has climbed steadily from 41% at the outset to 68% in 2016. Remarkably, 82% of patients on biologic therapy remain on their first biologic agent. Fewer than 4% have switched biologics more than twice. That’s very unlike the experiences reported elsewhere.
“I think one of the reasons we have such positive data is that we have a high percentage of patients staying on their background methotrexate,” said Dr. Weinblatt, codirector of clinical rheumatology and associate director of the Center for Arthritis and Joint Diseases at Brigham and Women’s Hospital, Boston.
He noted that Dutch investigators reported at the 2016 annual meeting of the American College of Rheumatology that among 1,230 consecutive rheumatoid arthritis patients started on etanercept (Enbrel) or adalimumab (Humira), 28% in the etanercept group were on concomitant methotrexate, as were 64% of those who started on adalimumab. The patients spent a median of 1.3-1.6 years and a maximum of 9.2-9.3 years on their biologic agent. Patients on adalimumab monotherapy were 2.61-fold more likely to drop out than were those on dual therapy with methotrexate. Patients on etanercept monotherapy were 1.2-fold more likely to drop out, a difference that didn’t achieve statistical significance.
Although the investigators did not study the mechanism of prolonged on-treatment survival, they speculated that it probably involved methotrexate’s documented ability to prevent formation of anti-adalimumab antibodies. In contrast, patients on etanercept don’t develop blocking antibodies, Dr. Weinblatt observed.
The randomized, double-blind CONCERTO trial conducted in 395 methotrexate- and biologic-naive RA patients demonstrated that methotrexate reduces the immunogenicity of adalimumab in dose-dependent fashion. Participants were randomized to open-label adalimumab at 40 mg every 2 weeks plus weekly double-blind methotrexate at 2.5, 5, 10, or 20 mg. Clinical outcomes at 26 weeks as reflected in 28-joint count disease activity score and the Clinical Disease Activity Index were significantly better in patients on 10 or 20 mg/week of methotrexate than in those on 2.5 or 5 mg/week. Serum adalimumab levels were higher in patients on the two higher doses of methotrexate as well (Ann Rheum Dis. 2015 Jun;74[6]:1037-44).
“It ends up that if you don’t use methotrexate or you use a very low dose you increase the risk of developing antibodies against adalimumab and decrease the efficacy of the drug. So in clinical practice, if you’re going to be working with dose titration of methotrexate and your patient is on adalimumab, there’s a threshold below which you probably shouldn’t go. In this study, doses of 10 mg/week or more induced a greater clinical response,” he said.
With infliximab (Remicade), based upon 20-year-old studies, the threshold is 7.5 mg of methotrexate per week.
“With etanercept, we don’t know what the threshold is. You don’t develop blocking anti-drug antibodies with etanercept, but we do know that methotrexate enhances the efficacy of etanercept, and it doesn’t do it by changing the biologic’s pharmacokinetics and there’s no increase in methotrexate blood levels,” the rheumatologist continued.
Unlike the anti-TNF biologics and rituximab, the efficacy of the Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib is not enhanced when the drugs are used in combination with methotrexate, studies indicate.
The efficacy of certolizumab pegol (Cimzia) wasn’t affected by methotrexate dose category in a prespecified pooled subgroup analysis of the phase III RAPID 1 and RAPID 2 clinical trials. In the 1,273 certolizumab-treated patients, the week-24 treatment response was similar regardless of whether patients were on methotrexate at 10 mg/week or less, 10-15 mg/week, or more than 15 mg/week. The investigators concluded that to minimize treatment-emergent adverse events, physicians can tailor background methotrexate dosing based upon individual patient tolerance without affecting certolizumab’s efficacy (Arthritis Care Res [Hoboken]. 2016 Mar;68[3]:299-307).
An important aspect of this analysis was that among the 325 subjects randomized to placebo rather than certolizumab, the treatment response at week 24 was significantly better in those on more than 15 mg/week of methotrexate than with lower doses of the drug.
“Most patients on methotrexate need more than 15 mg/week. So it astonishes me that such a high percentage of patients enrolled in clinical trials around the world are on, like, 14 mg/week. I mean, most patients need somewhere between 15 and 25 mg/week for a response, although over time you might be able to decrease that dose,” Dr. Weinblatt said.
Side effects of methotrexate
“The biggest issue with methotrexate is the tolerability problem, since serious adverse events are incredibly rare with this molecule,” he said.
Hepatotoxicity is a concern, but Dr. Weinblatt emphasized that elevated liver function tests do not equal cirrhosis.
“Historically, during the first 6 months on methotrexate 20%-25% of patients increase their transaminases in every clinical trial where that’s been looked at. Over time, the liver compensates for the drug. But 5%-6% of patients experience repeated moderate elevations more than 1.5 times the upper limit of normal,” he said.
Key risk factors for methotrexate-related hepatotoxicity were identified in a national observational cohort study of 659 military veterans over age 65 when they started methotrexate for rheumatic diseases. The investigators found a 6% incidence of moderately elevated liver enzymes during a mean follow-up period of 7 months. Obesity was associated with a 1.9-fold increased risk, a serum total cholesterol greater than 240 mg/dL conferred a 5.8-fold elevated risk, and abnormal liver function tests at baseline were associated with a 3.2-fold increased risk (Arthritis Care Res [Hoboken]. 2014 Aug;66[8]:1159-66).
“No surprise: It’s patients who weigh more who are at increased risk for methotrexate-related transaminase increases. I actually think the biggest factor with regard to methotrexate liver disease is the patient’s [body mass index]. Patients in North America aren’t getting any slimmer, so you need to look at this with your patients. If you have a morbidly obese patient on methotrexate whose transaminases suddenly start going up, that’s the patient who’s at greatest risk for methotrexate hepatotoxicity,” he cautioned.
The 3.2-fold increased risk of repeated elevated transaminases associated with abnormal baseline liver function tests in the Veterans Affairs study should be a red flag for rheumatologists.
“I personally think patients shouldn’t start on methotrexate if they have elevated transaminases. They ought to be normal at the start. There are too many other good options now to treat our patients,” Dr. Weinblatt said.
He reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
SNOWMASS, COLO. – “When I started working with methotrexate in 1982, I never would have predicted that methotrexate would become the standard of care in treating rheumatoid arthritis. There’s just no way,” Michael E. Weinblatt, MD, recalled at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“Even now, 35 years later, we continue to learn more about this fascinating drug,” added Dr. Weinblatt, professor of medicine at Harvard Medical School, Boston.
He highlighted recent developments in this ongoing story and presented some tricks of the trade gained in 35 years of up-close experience with the drug.
Enhancing effectiveness of biologics
One of the hottest topics in methotrexate research is the drug’s ability to enhance the effectiveness of many, but not all, biologic agents. All of the anti–tumor necrosis factor (anti-TNF) biologics as well as rituximab (Rituxan) are demonstrably more effective when used in combination with methotrexate. Dr. Weinblatt considers the widespread underutilization of this combination strategy scandalous.
“This is an incredibly important point: All of you use biologics and all of you use methotrexate, but I’ve been depressed by the fact that up to 30%-40% of patients – no matter which data set you look at – are on monotherapy biological therapy,” he said.
He cited data from the ongoing BRASS Registry (Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Registry) to underscore his point that good things happen when biologics and methotrexate are used together. Of 1,395 BRASS Registry participants prospectively followed since 2003, the proportion on biologic therapy has climbed steadily from 41% at the outset to 68% in 2016. Remarkably, 82% of patients on biologic therapy remain on their first biologic agent. Fewer than 4% have switched biologics more than twice. That’s very unlike the experiences reported elsewhere.
“I think one of the reasons we have such positive data is that we have a high percentage of patients staying on their background methotrexate,” said Dr. Weinblatt, codirector of clinical rheumatology and associate director of the Center for Arthritis and Joint Diseases at Brigham and Women’s Hospital, Boston.
He noted that Dutch investigators reported at the 2016 annual meeting of the American College of Rheumatology that among 1,230 consecutive rheumatoid arthritis patients started on etanercept (Enbrel) or adalimumab (Humira), 28% in the etanercept group were on concomitant methotrexate, as were 64% of those who started on adalimumab. The patients spent a median of 1.3-1.6 years and a maximum of 9.2-9.3 years on their biologic agent. Patients on adalimumab monotherapy were 2.61-fold more likely to drop out than were those on dual therapy with methotrexate. Patients on etanercept monotherapy were 1.2-fold more likely to drop out, a difference that didn’t achieve statistical significance.
Although the investigators did not study the mechanism of prolonged on-treatment survival, they speculated that it probably involved methotrexate’s documented ability to prevent formation of anti-adalimumab antibodies. In contrast, patients on etanercept don’t develop blocking antibodies, Dr. Weinblatt observed.
The randomized, double-blind CONCERTO trial conducted in 395 methotrexate- and biologic-naive RA patients demonstrated that methotrexate reduces the immunogenicity of adalimumab in dose-dependent fashion. Participants were randomized to open-label adalimumab at 40 mg every 2 weeks plus weekly double-blind methotrexate at 2.5, 5, 10, or 20 mg. Clinical outcomes at 26 weeks as reflected in 28-joint count disease activity score and the Clinical Disease Activity Index were significantly better in patients on 10 or 20 mg/week of methotrexate than in those on 2.5 or 5 mg/week. Serum adalimumab levels were higher in patients on the two higher doses of methotrexate as well (Ann Rheum Dis. 2015 Jun;74[6]:1037-44).
“It ends up that if you don’t use methotrexate or you use a very low dose you increase the risk of developing antibodies against adalimumab and decrease the efficacy of the drug. So in clinical practice, if you’re going to be working with dose titration of methotrexate and your patient is on adalimumab, there’s a threshold below which you probably shouldn’t go. In this study, doses of 10 mg/week or more induced a greater clinical response,” he said.
With infliximab (Remicade), based upon 20-year-old studies, the threshold is 7.5 mg of methotrexate per week.
“With etanercept, we don’t know what the threshold is. You don’t develop blocking anti-drug antibodies with etanercept, but we do know that methotrexate enhances the efficacy of etanercept, and it doesn’t do it by changing the biologic’s pharmacokinetics and there’s no increase in methotrexate blood levels,” the rheumatologist continued.
Unlike the anti-TNF biologics and rituximab, the efficacy of the Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib is not enhanced when the drugs are used in combination with methotrexate, studies indicate.
The efficacy of certolizumab pegol (Cimzia) wasn’t affected by methotrexate dose category in a prespecified pooled subgroup analysis of the phase III RAPID 1 and RAPID 2 clinical trials. In the 1,273 certolizumab-treated patients, the week-24 treatment response was similar regardless of whether patients were on methotrexate at 10 mg/week or less, 10-15 mg/week, or more than 15 mg/week. The investigators concluded that to minimize treatment-emergent adverse events, physicians can tailor background methotrexate dosing based upon individual patient tolerance without affecting certolizumab’s efficacy (Arthritis Care Res [Hoboken]. 2016 Mar;68[3]:299-307).
An important aspect of this analysis was that among the 325 subjects randomized to placebo rather than certolizumab, the treatment response at week 24 was significantly better in those on more than 15 mg/week of methotrexate than with lower doses of the drug.
“Most patients on methotrexate need more than 15 mg/week. So it astonishes me that such a high percentage of patients enrolled in clinical trials around the world are on, like, 14 mg/week. I mean, most patients need somewhere between 15 and 25 mg/week for a response, although over time you might be able to decrease that dose,” Dr. Weinblatt said.
Side effects of methotrexate
“The biggest issue with methotrexate is the tolerability problem, since serious adverse events are incredibly rare with this molecule,” he said.
Hepatotoxicity is a concern, but Dr. Weinblatt emphasized that elevated liver function tests do not equal cirrhosis.
“Historically, during the first 6 months on methotrexate 20%-25% of patients increase their transaminases in every clinical trial where that’s been looked at. Over time, the liver compensates for the drug. But 5%-6% of patients experience repeated moderate elevations more than 1.5 times the upper limit of normal,” he said.
Key risk factors for methotrexate-related hepatotoxicity were identified in a national observational cohort study of 659 military veterans over age 65 when they started methotrexate for rheumatic diseases. The investigators found a 6% incidence of moderately elevated liver enzymes during a mean follow-up period of 7 months. Obesity was associated with a 1.9-fold increased risk, a serum total cholesterol greater than 240 mg/dL conferred a 5.8-fold elevated risk, and abnormal liver function tests at baseline were associated with a 3.2-fold increased risk (Arthritis Care Res [Hoboken]. 2014 Aug;66[8]:1159-66).
“No surprise: It’s patients who weigh more who are at increased risk for methotrexate-related transaminase increases. I actually think the biggest factor with regard to methotrexate liver disease is the patient’s [body mass index]. Patients in North America aren’t getting any slimmer, so you need to look at this with your patients. If you have a morbidly obese patient on methotrexate whose transaminases suddenly start going up, that’s the patient who’s at greatest risk for methotrexate hepatotoxicity,” he cautioned.
The 3.2-fold increased risk of repeated elevated transaminases associated with abnormal baseline liver function tests in the Veterans Affairs study should be a red flag for rheumatologists.
“I personally think patients shouldn’t start on methotrexate if they have elevated transaminases. They ought to be normal at the start. There are too many other good options now to treat our patients,” Dr. Weinblatt said.
He reported receiving research grants from half a dozen companies and serving as a consultant to more than two dozen.
EXPERT ANALYSIS FROM THE WINTER RHEUMATOLOGY SYMPOSIUM
RA-BEAM trial shows that baricitinib improves RA symptoms, slows joint damage
Oral baricitinib, when added to standard rheumatoid arthritis treatment, improved symptoms and slowed the progression of joint damage, compared with both placebo and adalimumab, according to a report published online Feb. 15 in the New England Journal of Medicine.
The JAK inhibitor’s beneficial effects were noted as early as the first week of treatment and persisted throughout 1 year of follow-up in an international manufacturer-sponsored phase III trial (RA-BEAM) involving 1,305 adults with moderate to severe active rheumatoid arthritis (RA). The study participants had not responded to methotrexate and had never been treated with biologics; the majority had previously received at least two conventional synthetic disease-modifying antirheumatic drugs (DMARDs), said Peter C. Taylor, MD, PhD, of the Nuffield Department of Orthopedics, Rheumatology, and Musculoskeletal Sciences and the Kennedy Institute of Rheumatology at the University of Oxford (England), and his associates.
The primary efficacy endpoint – the proportion of patients at week 12 who showed a response of 20% or more according to American College of Rheumatology criteria (ACR20) – was 70% for baricitinib, compared with 40% for placebo. Baricitinib also was superior to placebo regarding all the major secondary efficacy endpoints of the study, including scores on the Health Assessment Questionnaire–Disability Index; results on the Disease Activity Score for 28 joints using high-sensitivity C-reactive protein (DAS28-CRP); remission according to the Simplified Disease Activity Index criteria; and the patients’ daily diary reports on duration and severity of morning joint stiffness, worst level of tiredness, and worst level of pain.
In addition, baricitinib was “considered to be significantly superior to adalimumab” according to the ACR20 (61% for adalimumab) and the DAS28-CRP responses at week 12. Both baricitinib and adalimumab were associated with significant reductions in the radiographic progression of structural joint damage, compared with placebo, at week 24 and week 52, the investigators reported (N Engl J Med. 2017;376:652-62).
Regarding safety outcomes, adverse events including infections were more frequent with baricitinib and adalimumab than with placebo, as were decreases in neutrophil counts, increases in aminotransferase and creatinine levels, and increases in low-density lipoprotein cholesterol levels. At 1 year, three patients in the baricitinib group and two in the adalimumab group discontinued treatment because of liver abnormalities.
Baricitinib is currently under consideration for approval by the U.S. Food and Drug Administration and was approved for marketing in the European Union on Feb. 13.
The RA-BEAM trial was supported by Eli Lilly and Incyte. Dr. Taylor reported receiving personal fees from Eli Lilly, and he and his associates reported ties to numerous other industry sources.
Oral baricitinib, when added to standard rheumatoid arthritis treatment, improved symptoms and slowed the progression of joint damage, compared with both placebo and adalimumab, according to a report published online Feb. 15 in the New England Journal of Medicine.
The JAK inhibitor’s beneficial effects were noted as early as the first week of treatment and persisted throughout 1 year of follow-up in an international manufacturer-sponsored phase III trial (RA-BEAM) involving 1,305 adults with moderate to severe active rheumatoid arthritis (RA). The study participants had not responded to methotrexate and had never been treated with biologics; the majority had previously received at least two conventional synthetic disease-modifying antirheumatic drugs (DMARDs), said Peter C. Taylor, MD, PhD, of the Nuffield Department of Orthopedics, Rheumatology, and Musculoskeletal Sciences and the Kennedy Institute of Rheumatology at the University of Oxford (England), and his associates.
The primary efficacy endpoint – the proportion of patients at week 12 who showed a response of 20% or more according to American College of Rheumatology criteria (ACR20) – was 70% for baricitinib, compared with 40% for placebo. Baricitinib also was superior to placebo regarding all the major secondary efficacy endpoints of the study, including scores on the Health Assessment Questionnaire–Disability Index; results on the Disease Activity Score for 28 joints using high-sensitivity C-reactive protein (DAS28-CRP); remission according to the Simplified Disease Activity Index criteria; and the patients’ daily diary reports on duration and severity of morning joint stiffness, worst level of tiredness, and worst level of pain.
In addition, baricitinib was “considered to be significantly superior to adalimumab” according to the ACR20 (61% for adalimumab) and the DAS28-CRP responses at week 12. Both baricitinib and adalimumab were associated with significant reductions in the radiographic progression of structural joint damage, compared with placebo, at week 24 and week 52, the investigators reported (N Engl J Med. 2017;376:652-62).
Regarding safety outcomes, adverse events including infections were more frequent with baricitinib and adalimumab than with placebo, as were decreases in neutrophil counts, increases in aminotransferase and creatinine levels, and increases in low-density lipoprotein cholesterol levels. At 1 year, three patients in the baricitinib group and two in the adalimumab group discontinued treatment because of liver abnormalities.
Baricitinib is currently under consideration for approval by the U.S. Food and Drug Administration and was approved for marketing in the European Union on Feb. 13.
The RA-BEAM trial was supported by Eli Lilly and Incyte. Dr. Taylor reported receiving personal fees from Eli Lilly, and he and his associates reported ties to numerous other industry sources.
Oral baricitinib, when added to standard rheumatoid arthritis treatment, improved symptoms and slowed the progression of joint damage, compared with both placebo and adalimumab, according to a report published online Feb. 15 in the New England Journal of Medicine.
The JAK inhibitor’s beneficial effects were noted as early as the first week of treatment and persisted throughout 1 year of follow-up in an international manufacturer-sponsored phase III trial (RA-BEAM) involving 1,305 adults with moderate to severe active rheumatoid arthritis (RA). The study participants had not responded to methotrexate and had never been treated with biologics; the majority had previously received at least two conventional synthetic disease-modifying antirheumatic drugs (DMARDs), said Peter C. Taylor, MD, PhD, of the Nuffield Department of Orthopedics, Rheumatology, and Musculoskeletal Sciences and the Kennedy Institute of Rheumatology at the University of Oxford (England), and his associates.
The primary efficacy endpoint – the proportion of patients at week 12 who showed a response of 20% or more according to American College of Rheumatology criteria (ACR20) – was 70% for baricitinib, compared with 40% for placebo. Baricitinib also was superior to placebo regarding all the major secondary efficacy endpoints of the study, including scores on the Health Assessment Questionnaire–Disability Index; results on the Disease Activity Score for 28 joints using high-sensitivity C-reactive protein (DAS28-CRP); remission according to the Simplified Disease Activity Index criteria; and the patients’ daily diary reports on duration and severity of morning joint stiffness, worst level of tiredness, and worst level of pain.
In addition, baricitinib was “considered to be significantly superior to adalimumab” according to the ACR20 (61% for adalimumab) and the DAS28-CRP responses at week 12. Both baricitinib and adalimumab were associated with significant reductions in the radiographic progression of structural joint damage, compared with placebo, at week 24 and week 52, the investigators reported (N Engl J Med. 2017;376:652-62).
Regarding safety outcomes, adverse events including infections were more frequent with baricitinib and adalimumab than with placebo, as were decreases in neutrophil counts, increases in aminotransferase and creatinine levels, and increases in low-density lipoprotein cholesterol levels. At 1 year, three patients in the baricitinib group and two in the adalimumab group discontinued treatment because of liver abnormalities.
Baricitinib is currently under consideration for approval by the U.S. Food and Drug Administration and was approved for marketing in the European Union on Feb. 13.
The RA-BEAM trial was supported by Eli Lilly and Incyte. Dr. Taylor reported receiving personal fees from Eli Lilly, and he and his associates reported ties to numerous other industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: The primary efficacy end point – the proportion of patients at week 12 who showed an ACR20 response – was 70% for baricitinib, compared with 40% for placebo.
Data source: A manufacturer-sponsored, international, randomized, double-blind phase III clinical trial involving 1,305 adults with moderate to severe active RA.
Disclosures: This study was supported by Eli Lilly and Incyte. Dr. Taylor reported receiving personal fees from Eli Lilly, and he and his associates reported ties to numerous other industry sources.