User login
Compounding rules challenge practice norms
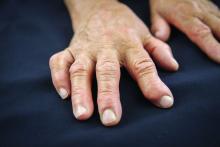
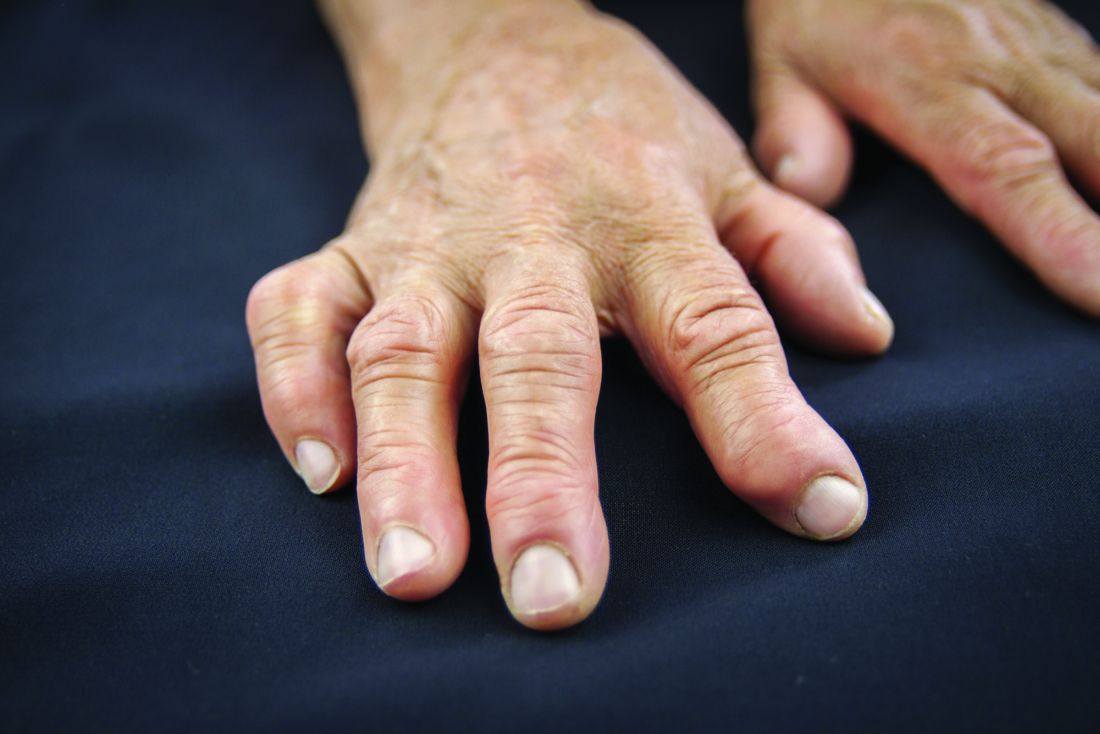
As new rules about drug compounding get shaped, rheumatologists seek to protect their ability to combine injectable drugs – most commonly a steroid and a local anesthetic – in their own offices.
In a position statement sent to government agencies and members of Congress in February, the American College of Rheumatology voiced concerns that the practice, which it called “critical,” could become a casualty of drug-compounding regulations under revision by the United States Pharmacopeial Convention (USP), a nonprofit group whose standards are enforceable by state and federal regulators.
In the same position statement on compounding, the ACR said it also seeks a change to a Food and Drug Administration rule limiting practitioners’ access to quinacrine, a drug only available through compounding pharmacies that is sometimes used to treat lupus patients. Quinacrine is not on the FDA’s current list of bulk substances approved for compounding, except by special permission. The ACR has asked the agency to add quinacrine to the list, but no one knows when this will happen.
Rheumatologists may also be more restricted than before in terms of which compounding pharmacies they can turn to, as new federal standards divide them into two types – those that can provide medicines in larger quantities and those that can’t.
Steroid fiasco sparked rule revisions
The ACR’s concerns follow a tighter focus by state and federal agencies on drug compounding after a fungal meningitis outbreak in 2012 was traced to contaminated steroids produced in bulk by a compounding pharmacy.
More than 800 infections, 64 of them fatal, occurred after the New England Compounding Center in Framingham, Mass., sold contaminated methylprednisolone acetate that was used in epidural and intra-articular joint injections.
The following year Congress passed the Drug Quality and Security Act, which aims, in part, to prevent compounding pharmacies from engaging in what amounts to unregulated manufacturing.
As part of the law, the FDA created a list of drugs appropriate for compounding and a process by which larger compounding pharmacies must register with the FDA, and agree to inspections. The USP standards, meanwhile, address detailed technical and safety aspects of compounding and are enforceable by the FDA and state agencies.
“USP and FDA have had the ability to regulate compounding for over a decade, but only recently have the rules become actively enforced,” said Donald Miller, PharmD, of North Dakota State University, Fargo, who helped shape the ACR’s position statement on compounding with the help of rheumatologists in private practice.
“When you make guidelines for safety, they make sense, but then you can’t anticipate the way it’s going to affect individuals’ practice. And that’s where rheumatology got caught up,” said Dr. Miller, who was a member of the FDA Arthritis Advisory Committee in 2014-2016.
In-office mixing a top concern
Other specialties, including dermatology and immunology, also stand to be affected by various changes to compounding law and practice – and their societies have been active in voicing concerns.
Though the latest revisions of USP chapter 797, which impacts in-office mixing, are still being sorted out, it’s the No. 1 compounding-related concern for rheumatologists, Dr. Miller said.
Rheumatologists routinely mix an analgesic and a steroid for injection. The analgesic makes the steroids less viscous, and offers patients hours of immediate relief. They also add analgesics to hyaluronic acid injected for viscosupplementation. The mixing is usually conducted bedside, and the injections are administered right away.
Technically, combining these products amounts to sterile compounding, Dr. Miller explained. “And theoretically, under these rules, a physician could still do this, but they’d have to do it under a sterile hood like you find in a pharmacy, and that’s just not practical. It also becomes a matter of interpretation.”
USP chapter 797 sanctions in-office mixing for “immediate use” with individual patients – which is nearly always the case for the steroid injections used in rheumatology. But it’s unclear whether “immediate use” means emergency use only, or allows for routine use, as rheumatologists hope.
“One reason this came to rheumatology’s attention is that some state boards of medicine were inspecting and saying ‘Hey, you can’t do that,’ ” Dr. Miller said.
“There’s that law of unintended consequences where you snare things in a net that you really don’t want to,” Dr. Huffstutter said.
Marcus Snow, MD, a rheumatologist at the University of Nebraska, Omaha, who also worked on the statement, said that most rheumatologists are likely unaware that their ability to mix drugs in-office has been called into question.
“I brought it up at our division meeting with a group of 10 rheumatologists, and no one was aware that this was coming down the pike,” Dr. Snow said in an interview.
Pediatric issues
Pediatric rheumatologists, and adult rheumatologists who see children occasionally, use compounding pharmacies to create palatable oral medicines and adjusted doses of adult treatments.
They also use injections combining steroids with analgesics, and consider the addition of the analgesic a key aid to compliance.
“The biggest barrier we have is patient and parent anxiety about doing the procedure and the associated pain. We always administer our steroids mixed with lidocaine to help with the postprocedural discomfort,” said Adam Reinhardt, MD, chief of pediatric rheumatology at the University of Nebraska and Children’s Hospital and Medical Center in Omaha.
Steroid injections can mean avoiding or delaying systemic treatment in children with oligoarticular arthritis, he said. “Most of us consider them a first-line therapy. The hope is that you can get by without having to use meds like methotrexate if you can get a prolonged response in the one or two joints that are active in that patient.”
But Dr. Reinhardt said that, while he mixed his own injections during his fellowship training, Children’s of Omaha now insists that they be prepared by in-house pharmacists, working under sterile hoods. The delay to receiving them in the clinic or procedure room is 40 minutes to an hour, he said, which the clinicians accommodate through careful scheduling.
The change from mixing in-clinic to relying on the central pharmacy came about in recent years, Dr. Reinhardt said, because of broader concerns related to medication storage in the clinics. While ordering from the central pharmacy works for his practice, he said, “I probably only inject maybe 50-70 joints a year, while adult rheumatologists are injecting far more than that. For a busy private practice, I can see that being a huge time constraint,” he said.
Relevance of rules
None of the rheumatologists interviewed questioned the need for tightened state and federal oversight of compounding practices overall – just the applicability of certain rules to their own practice.
Dr. Snow and Dr. Huffstutter noted that reports of infected joints – a potential result of a contaminated injection – are sporadic and rare. “There’s very little research in this, but [these types of injections] have been standard practice for decades,” Dr. Snow said.
Srikanth Mukkera, MD, a rheumatologist in Tupelo, Miss., agreed that “sporadic cases of joint infection do happen following injection, but it can be hard to show if an injection was the cause.”
Assuring that medicines are mixed only immediately prior to injection, and not stored, reduces the likelihood of contamination, Dr. Mukkera said. Moreover, he noted, epidural injections such as those that resulted in the 2012 meningitis outbreak carry different risks than those seen in intra-articular injections.
Dr. Miller, the lead author of the ACR statement, said that the rheumatologists on our committee “don’t know of anyone that’s had a knee or other joint infection from a contaminated injection. They feel that unless somebody finds some evidence of that, they should be allowed to continue” with their usual practice.
He said that he feels that the USP will ultimately heed the concerns of rheumatologists and hopefully provide a more relaxed interpretation of in-office compounding. “We’re hoping they’ll make some exceptions when they revise 797 standards or at least maybe leave room for organizations to create a best practice statement. We’ll see,” Dr. Miller said.
But this is in no way guaranteed. Dr. Huffstutter said he fears that, if the rules come to be interpreted more narrowly, even standard practices like reconstituting biologic drugs for infusion – something that’s also a routine part of in-office practice – could fall under the rubric of sterile compounding and come into question.
The quinacrine problem
A separate compounding-related issue in rheumatology is clinicians’ access to quinacrine, an antimalarial rheumatology drug that, while infrequently used, represents the only alternative to hydroxychloroquine for some lupus patients.
“There are no alternatives out there for hydroxychloroquine, so we need it as a backup,” Dr. Snow said. “If hydroxychloroquine isn’t an option, there’s nothing out there that we can use. There’s no easy replacement.”
Dr. Huffstutter said he currently had no patients on quinacrine. “It’s not very often that we use it, but in those patients that really need it, it can make a huge difference in how they do.”
Quinacrine is no longer manufactured commercially as a finished drug product but is available in a powder that compounding physicians put into 100-mg capsules. It is not on the FDA’s current list of drugs available for compounding except with special permission.
While the ACR has requested that the FDA add it the list of bulk drug substances that can be used in compounding, quinacrine remains off the list for now – and, providers say, hard to find.
Moreover, while rheumatologists may have previously been able to order and store quantities of quinacrine and other compounded nonsterile medications to dispense to their patients, they can no longer easily do so, as only the FDA-approved compounding “outsourcing facilities” are allowed to process larger orders; the rest can only respond to prescriptions for individual patients.
Dr. Miller said it’s likely that quinacrine will make it onto the FDA’s next list of bulk drugs available for compounding. “The FDA has kind of said, ‘Don’t worry about it,’ ” he said.
As new rules about drug compounding get shaped, rheumatologists seek to protect their ability to combine injectable drugs – most commonly a steroid and a local anesthetic – in their own offices.
In a position statement sent to government agencies and members of Congress in February, the American College of Rheumatology voiced concerns that the practice, which it called “critical,” could become a casualty of drug-compounding regulations under revision by the United States Pharmacopeial Convention (USP), a nonprofit group whose standards are enforceable by state and federal regulators.
In the same position statement on compounding, the ACR said it also seeks a change to a Food and Drug Administration rule limiting practitioners’ access to quinacrine, a drug only available through compounding pharmacies that is sometimes used to treat lupus patients. Quinacrine is not on the FDA’s current list of bulk substances approved for compounding, except by special permission. The ACR has asked the agency to add quinacrine to the list, but no one knows when this will happen.
Rheumatologists may also be more restricted than before in terms of which compounding pharmacies they can turn to, as new federal standards divide them into two types – those that can provide medicines in larger quantities and those that can’t.
Steroid fiasco sparked rule revisions
The ACR’s concerns follow a tighter focus by state and federal agencies on drug compounding after a fungal meningitis outbreak in 2012 was traced to contaminated steroids produced in bulk by a compounding pharmacy.
More than 800 infections, 64 of them fatal, occurred after the New England Compounding Center in Framingham, Mass., sold contaminated methylprednisolone acetate that was used in epidural and intra-articular joint injections.
The following year Congress passed the Drug Quality and Security Act, which aims, in part, to prevent compounding pharmacies from engaging in what amounts to unregulated manufacturing.
As part of the law, the FDA created a list of drugs appropriate for compounding and a process by which larger compounding pharmacies must register with the FDA, and agree to inspections. The USP standards, meanwhile, address detailed technical and safety aspects of compounding and are enforceable by the FDA and state agencies.
“USP and FDA have had the ability to regulate compounding for over a decade, but only recently have the rules become actively enforced,” said Donald Miller, PharmD, of North Dakota State University, Fargo, who helped shape the ACR’s position statement on compounding with the help of rheumatologists in private practice.
“When you make guidelines for safety, they make sense, but then you can’t anticipate the way it’s going to affect individuals’ practice. And that’s where rheumatology got caught up,” said Dr. Miller, who was a member of the FDA Arthritis Advisory Committee in 2014-2016.
In-office mixing a top concern
Other specialties, including dermatology and immunology, also stand to be affected by various changes to compounding law and practice – and their societies have been active in voicing concerns.
Though the latest revisions of USP chapter 797, which impacts in-office mixing, are still being sorted out, it’s the No. 1 compounding-related concern for rheumatologists, Dr. Miller said.
Rheumatologists routinely mix an analgesic and a steroid for injection. The analgesic makes the steroids less viscous, and offers patients hours of immediate relief. They also add analgesics to hyaluronic acid injected for viscosupplementation. The mixing is usually conducted bedside, and the injections are administered right away.
Technically, combining these products amounts to sterile compounding, Dr. Miller explained. “And theoretically, under these rules, a physician could still do this, but they’d have to do it under a sterile hood like you find in a pharmacy, and that’s just not practical. It also becomes a matter of interpretation.”
USP chapter 797 sanctions in-office mixing for “immediate use” with individual patients – which is nearly always the case for the steroid injections used in rheumatology. But it’s unclear whether “immediate use” means emergency use only, or allows for routine use, as rheumatologists hope.
“One reason this came to rheumatology’s attention is that some state boards of medicine were inspecting and saying ‘Hey, you can’t do that,’ ” Dr. Miller said.
“There’s that law of unintended consequences where you snare things in a net that you really don’t want to,” Dr. Huffstutter said.
Marcus Snow, MD, a rheumatologist at the University of Nebraska, Omaha, who also worked on the statement, said that most rheumatologists are likely unaware that their ability to mix drugs in-office has been called into question.
“I brought it up at our division meeting with a group of 10 rheumatologists, and no one was aware that this was coming down the pike,” Dr. Snow said in an interview.
Pediatric issues
Pediatric rheumatologists, and adult rheumatologists who see children occasionally, use compounding pharmacies to create palatable oral medicines and adjusted doses of adult treatments.
They also use injections combining steroids with analgesics, and consider the addition of the analgesic a key aid to compliance.
“The biggest barrier we have is patient and parent anxiety about doing the procedure and the associated pain. We always administer our steroids mixed with lidocaine to help with the postprocedural discomfort,” said Adam Reinhardt, MD, chief of pediatric rheumatology at the University of Nebraska and Children’s Hospital and Medical Center in Omaha.
Steroid injections can mean avoiding or delaying systemic treatment in children with oligoarticular arthritis, he said. “Most of us consider them a first-line therapy. The hope is that you can get by without having to use meds like methotrexate if you can get a prolonged response in the one or two joints that are active in that patient.”
But Dr. Reinhardt said that, while he mixed his own injections during his fellowship training, Children’s of Omaha now insists that they be prepared by in-house pharmacists, working under sterile hoods. The delay to receiving them in the clinic or procedure room is 40 minutes to an hour, he said, which the clinicians accommodate through careful scheduling.
The change from mixing in-clinic to relying on the central pharmacy came about in recent years, Dr. Reinhardt said, because of broader concerns related to medication storage in the clinics. While ordering from the central pharmacy works for his practice, he said, “I probably only inject maybe 50-70 joints a year, while adult rheumatologists are injecting far more than that. For a busy private practice, I can see that being a huge time constraint,” he said.
Relevance of rules
None of the rheumatologists interviewed questioned the need for tightened state and federal oversight of compounding practices overall – just the applicability of certain rules to their own practice.
Dr. Snow and Dr. Huffstutter noted that reports of infected joints – a potential result of a contaminated injection – are sporadic and rare. “There’s very little research in this, but [these types of injections] have been standard practice for decades,” Dr. Snow said.
Srikanth Mukkera, MD, a rheumatologist in Tupelo, Miss., agreed that “sporadic cases of joint infection do happen following injection, but it can be hard to show if an injection was the cause.”
Assuring that medicines are mixed only immediately prior to injection, and not stored, reduces the likelihood of contamination, Dr. Mukkera said. Moreover, he noted, epidural injections such as those that resulted in the 2012 meningitis outbreak carry different risks than those seen in intra-articular injections.
Dr. Miller, the lead author of the ACR statement, said that the rheumatologists on our committee “don’t know of anyone that’s had a knee or other joint infection from a contaminated injection. They feel that unless somebody finds some evidence of that, they should be allowed to continue” with their usual practice.
He said that he feels that the USP will ultimately heed the concerns of rheumatologists and hopefully provide a more relaxed interpretation of in-office compounding. “We’re hoping they’ll make some exceptions when they revise 797 standards or at least maybe leave room for organizations to create a best practice statement. We’ll see,” Dr. Miller said.
But this is in no way guaranteed. Dr. Huffstutter said he fears that, if the rules come to be interpreted more narrowly, even standard practices like reconstituting biologic drugs for infusion – something that’s also a routine part of in-office practice – could fall under the rubric of sterile compounding and come into question.
The quinacrine problem
A separate compounding-related issue in rheumatology is clinicians’ access to quinacrine, an antimalarial rheumatology drug that, while infrequently used, represents the only alternative to hydroxychloroquine for some lupus patients.
“There are no alternatives out there for hydroxychloroquine, so we need it as a backup,” Dr. Snow said. “If hydroxychloroquine isn’t an option, there’s nothing out there that we can use. There’s no easy replacement.”
Dr. Huffstutter said he currently had no patients on quinacrine. “It’s not very often that we use it, but in those patients that really need it, it can make a huge difference in how they do.”
Quinacrine is no longer manufactured commercially as a finished drug product but is available in a powder that compounding physicians put into 100-mg capsules. It is not on the FDA’s current list of drugs available for compounding except with special permission.
While the ACR has requested that the FDA add it the list of bulk drug substances that can be used in compounding, quinacrine remains off the list for now – and, providers say, hard to find.
Moreover, while rheumatologists may have previously been able to order and store quantities of quinacrine and other compounded nonsterile medications to dispense to their patients, they can no longer easily do so, as only the FDA-approved compounding “outsourcing facilities” are allowed to process larger orders; the rest can only respond to prescriptions for individual patients.
Dr. Miller said it’s likely that quinacrine will make it onto the FDA’s next list of bulk drugs available for compounding. “The FDA has kind of said, ‘Don’t worry about it,’ ” he said.
As new rules about drug compounding get shaped, rheumatologists seek to protect their ability to combine injectable drugs – most commonly a steroid and a local anesthetic – in their own offices.
In a position statement sent to government agencies and members of Congress in February, the American College of Rheumatology voiced concerns that the practice, which it called “critical,” could become a casualty of drug-compounding regulations under revision by the United States Pharmacopeial Convention (USP), a nonprofit group whose standards are enforceable by state and federal regulators.
In the same position statement on compounding, the ACR said it also seeks a change to a Food and Drug Administration rule limiting practitioners’ access to quinacrine, a drug only available through compounding pharmacies that is sometimes used to treat lupus patients. Quinacrine is not on the FDA’s current list of bulk substances approved for compounding, except by special permission. The ACR has asked the agency to add quinacrine to the list, but no one knows when this will happen.
Rheumatologists may also be more restricted than before in terms of which compounding pharmacies they can turn to, as new federal standards divide them into two types – those that can provide medicines in larger quantities and those that can’t.
Steroid fiasco sparked rule revisions
The ACR’s concerns follow a tighter focus by state and federal agencies on drug compounding after a fungal meningitis outbreak in 2012 was traced to contaminated steroids produced in bulk by a compounding pharmacy.
More than 800 infections, 64 of them fatal, occurred after the New England Compounding Center in Framingham, Mass., sold contaminated methylprednisolone acetate that was used in epidural and intra-articular joint injections.
The following year Congress passed the Drug Quality and Security Act, which aims, in part, to prevent compounding pharmacies from engaging in what amounts to unregulated manufacturing.
As part of the law, the FDA created a list of drugs appropriate for compounding and a process by which larger compounding pharmacies must register with the FDA, and agree to inspections. The USP standards, meanwhile, address detailed technical and safety aspects of compounding and are enforceable by the FDA and state agencies.
“USP and FDA have had the ability to regulate compounding for over a decade, but only recently have the rules become actively enforced,” said Donald Miller, PharmD, of North Dakota State University, Fargo, who helped shape the ACR’s position statement on compounding with the help of rheumatologists in private practice.
“When you make guidelines for safety, they make sense, but then you can’t anticipate the way it’s going to affect individuals’ practice. And that’s where rheumatology got caught up,” said Dr. Miller, who was a member of the FDA Arthritis Advisory Committee in 2014-2016.
In-office mixing a top concern
Other specialties, including dermatology and immunology, also stand to be affected by various changes to compounding law and practice – and their societies have been active in voicing concerns.
Though the latest revisions of USP chapter 797, which impacts in-office mixing, are still being sorted out, it’s the No. 1 compounding-related concern for rheumatologists, Dr. Miller said.
Rheumatologists routinely mix an analgesic and a steroid for injection. The analgesic makes the steroids less viscous, and offers patients hours of immediate relief. They also add analgesics to hyaluronic acid injected for viscosupplementation. The mixing is usually conducted bedside, and the injections are administered right away.
Technically, combining these products amounts to sterile compounding, Dr. Miller explained. “And theoretically, under these rules, a physician could still do this, but they’d have to do it under a sterile hood like you find in a pharmacy, and that’s just not practical. It also becomes a matter of interpretation.”
USP chapter 797 sanctions in-office mixing for “immediate use” with individual patients – which is nearly always the case for the steroid injections used in rheumatology. But it’s unclear whether “immediate use” means emergency use only, or allows for routine use, as rheumatologists hope.
“One reason this came to rheumatology’s attention is that some state boards of medicine were inspecting and saying ‘Hey, you can’t do that,’ ” Dr. Miller said.
“There’s that law of unintended consequences where you snare things in a net that you really don’t want to,” Dr. Huffstutter said.
Marcus Snow, MD, a rheumatologist at the University of Nebraska, Omaha, who also worked on the statement, said that most rheumatologists are likely unaware that their ability to mix drugs in-office has been called into question.
“I brought it up at our division meeting with a group of 10 rheumatologists, and no one was aware that this was coming down the pike,” Dr. Snow said in an interview.
Pediatric issues
Pediatric rheumatologists, and adult rheumatologists who see children occasionally, use compounding pharmacies to create palatable oral medicines and adjusted doses of adult treatments.
They also use injections combining steroids with analgesics, and consider the addition of the analgesic a key aid to compliance.
“The biggest barrier we have is patient and parent anxiety about doing the procedure and the associated pain. We always administer our steroids mixed with lidocaine to help with the postprocedural discomfort,” said Adam Reinhardt, MD, chief of pediatric rheumatology at the University of Nebraska and Children’s Hospital and Medical Center in Omaha.
Steroid injections can mean avoiding or delaying systemic treatment in children with oligoarticular arthritis, he said. “Most of us consider them a first-line therapy. The hope is that you can get by without having to use meds like methotrexate if you can get a prolonged response in the one or two joints that are active in that patient.”
But Dr. Reinhardt said that, while he mixed his own injections during his fellowship training, Children’s of Omaha now insists that they be prepared by in-house pharmacists, working under sterile hoods. The delay to receiving them in the clinic or procedure room is 40 minutes to an hour, he said, which the clinicians accommodate through careful scheduling.
The change from mixing in-clinic to relying on the central pharmacy came about in recent years, Dr. Reinhardt said, because of broader concerns related to medication storage in the clinics. While ordering from the central pharmacy works for his practice, he said, “I probably only inject maybe 50-70 joints a year, while adult rheumatologists are injecting far more than that. For a busy private practice, I can see that being a huge time constraint,” he said.
Relevance of rules
None of the rheumatologists interviewed questioned the need for tightened state and federal oversight of compounding practices overall – just the applicability of certain rules to their own practice.
Dr. Snow and Dr. Huffstutter noted that reports of infected joints – a potential result of a contaminated injection – are sporadic and rare. “There’s very little research in this, but [these types of injections] have been standard practice for decades,” Dr. Snow said.
Srikanth Mukkera, MD, a rheumatologist in Tupelo, Miss., agreed that “sporadic cases of joint infection do happen following injection, but it can be hard to show if an injection was the cause.”
Assuring that medicines are mixed only immediately prior to injection, and not stored, reduces the likelihood of contamination, Dr. Mukkera said. Moreover, he noted, epidural injections such as those that resulted in the 2012 meningitis outbreak carry different risks than those seen in intra-articular injections.
Dr. Miller, the lead author of the ACR statement, said that the rheumatologists on our committee “don’t know of anyone that’s had a knee or other joint infection from a contaminated injection. They feel that unless somebody finds some evidence of that, they should be allowed to continue” with their usual practice.
He said that he feels that the USP will ultimately heed the concerns of rheumatologists and hopefully provide a more relaxed interpretation of in-office compounding. “We’re hoping they’ll make some exceptions when they revise 797 standards or at least maybe leave room for organizations to create a best practice statement. We’ll see,” Dr. Miller said.
But this is in no way guaranteed. Dr. Huffstutter said he fears that, if the rules come to be interpreted more narrowly, even standard practices like reconstituting biologic drugs for infusion – something that’s also a routine part of in-office practice – could fall under the rubric of sterile compounding and come into question.
The quinacrine problem
A separate compounding-related issue in rheumatology is clinicians’ access to quinacrine, an antimalarial rheumatology drug that, while infrequently used, represents the only alternative to hydroxychloroquine for some lupus patients.
“There are no alternatives out there for hydroxychloroquine, so we need it as a backup,” Dr. Snow said. “If hydroxychloroquine isn’t an option, there’s nothing out there that we can use. There’s no easy replacement.”
Dr. Huffstutter said he currently had no patients on quinacrine. “It’s not very often that we use it, but in those patients that really need it, it can make a huge difference in how they do.”
Quinacrine is no longer manufactured commercially as a finished drug product but is available in a powder that compounding physicians put into 100-mg capsules. It is not on the FDA’s current list of drugs available for compounding except with special permission.
While the ACR has requested that the FDA add it the list of bulk drug substances that can be used in compounding, quinacrine remains off the list for now – and, providers say, hard to find.
Moreover, while rheumatologists may have previously been able to order and store quantities of quinacrine and other compounded nonsterile medications to dispense to their patients, they can no longer easily do so, as only the FDA-approved compounding “outsourcing facilities” are allowed to process larger orders; the rest can only respond to prescriptions for individual patients.
Dr. Miller said it’s likely that quinacrine will make it onto the FDA’s next list of bulk drugs available for compounding. “The FDA has kind of said, ‘Don’t worry about it,’ ” he said.
Fatigue in rheumatoid arthritis declines with pedometer use
Rheumatoid arthritis patients given pedometers reported a decrease in fatigue over a 21-week period, coupled with a small but significant increase in steps walked, according to a study from investigators at the University of California, San Francisco.
“Our results provide evidence that pedometers are effective at increasing physical activity among people with RA and provide support for the hypothesis that increasing physical activity by walking has important effects on fatigue and other RA symptoms,” wrote the researchers, led by Patricia Katz, PhD.
Patients who were given only pedometers increased their walking from baseline to week 21 by an average of 1,441 steps, while those given pedometers plus a step goal walked a mean of 1,656 steps additionally. The control group had a mean decrease of 747 steps.
Scores on the Patient-Reported Outcome Measurement Information System fatigue short form at 21 weeks declined by –3.2 for those who received pedometers only (P = .02) and by –4.8 for those who received pedometers plus a step goal (P = .0002), compared with the control group, which reported a decrease of –1.6 (P = .26).
While both intervention groups saw increased activity when compared with controls, patients given a step goal saw constant improvement, averaging an increase of 22 steps per week, while those without a step goal reported a decrease of 37 steps per week, which the researchers suspected could be from a larger initial increase in activity that could have discouraged a continued increase over time.
Dr. Katz and her associates said that the positive change that a rise in physical activity had on all other aspects of patients’ self-reported assessment shows promising results for the efficacy of exercise in patients, which some physicians had questioned.
“In the past, there was concern that physical activity or exercise might exacerbate rheumatoid arthritis symptoms, [however], our findings are similar to previous reports that exercise did not lead to worsened rheumatoid arthritis,” they wrote.
This study was limited by the small number of participants in the study, as well as the self-reported method of data collection. Researchers were also unable to gather activity intensity, as well as the amount of time patients wore the pedometer, causing the measure of adherence to “only be considered an estimate.”
The researchers reported technical difficulties with the devices, creating missing data.
The study was supported by the Rheumatology Research Foundation. The researchers reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
On Twitter @EAZTweets
Rheumatoid arthritis patients given pedometers reported a decrease in fatigue over a 21-week period, coupled with a small but significant increase in steps walked, according to a study from investigators at the University of California, San Francisco.
“Our results provide evidence that pedometers are effective at increasing physical activity among people with RA and provide support for the hypothesis that increasing physical activity by walking has important effects on fatigue and other RA symptoms,” wrote the researchers, led by Patricia Katz, PhD.
Patients who were given only pedometers increased their walking from baseline to week 21 by an average of 1,441 steps, while those given pedometers plus a step goal walked a mean of 1,656 steps additionally. The control group had a mean decrease of 747 steps.
Scores on the Patient-Reported Outcome Measurement Information System fatigue short form at 21 weeks declined by –3.2 for those who received pedometers only (P = .02) and by –4.8 for those who received pedometers plus a step goal (P = .0002), compared with the control group, which reported a decrease of –1.6 (P = .26).
While both intervention groups saw increased activity when compared with controls, patients given a step goal saw constant improvement, averaging an increase of 22 steps per week, while those without a step goal reported a decrease of 37 steps per week, which the researchers suspected could be from a larger initial increase in activity that could have discouraged a continued increase over time.
Dr. Katz and her associates said that the positive change that a rise in physical activity had on all other aspects of patients’ self-reported assessment shows promising results for the efficacy of exercise in patients, which some physicians had questioned.
“In the past, there was concern that physical activity or exercise might exacerbate rheumatoid arthritis symptoms, [however], our findings are similar to previous reports that exercise did not lead to worsened rheumatoid arthritis,” they wrote.
This study was limited by the small number of participants in the study, as well as the self-reported method of data collection. Researchers were also unable to gather activity intensity, as well as the amount of time patients wore the pedometer, causing the measure of adherence to “only be considered an estimate.”
The researchers reported technical difficulties with the devices, creating missing data.
The study was supported by the Rheumatology Research Foundation. The researchers reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
On Twitter @EAZTweets
Rheumatoid arthritis patients given pedometers reported a decrease in fatigue over a 21-week period, coupled with a small but significant increase in steps walked, according to a study from investigators at the University of California, San Francisco.
“Our results provide evidence that pedometers are effective at increasing physical activity among people with RA and provide support for the hypothesis that increasing physical activity by walking has important effects on fatigue and other RA symptoms,” wrote the researchers, led by Patricia Katz, PhD.
Patients who were given only pedometers increased their walking from baseline to week 21 by an average of 1,441 steps, while those given pedometers plus a step goal walked a mean of 1,656 steps additionally. The control group had a mean decrease of 747 steps.
Scores on the Patient-Reported Outcome Measurement Information System fatigue short form at 21 weeks declined by –3.2 for those who received pedometers only (P = .02) and by –4.8 for those who received pedometers plus a step goal (P = .0002), compared with the control group, which reported a decrease of –1.6 (P = .26).
While both intervention groups saw increased activity when compared with controls, patients given a step goal saw constant improvement, averaging an increase of 22 steps per week, while those without a step goal reported a decrease of 37 steps per week, which the researchers suspected could be from a larger initial increase in activity that could have discouraged a continued increase over time.
Dr. Katz and her associates said that the positive change that a rise in physical activity had on all other aspects of patients’ self-reported assessment shows promising results for the efficacy of exercise in patients, which some physicians had questioned.
“In the past, there was concern that physical activity or exercise might exacerbate rheumatoid arthritis symptoms, [however], our findings are similar to previous reports that exercise did not lead to worsened rheumatoid arthritis,” they wrote.
This study was limited by the small number of participants in the study, as well as the self-reported method of data collection. Researchers were also unable to gather activity intensity, as well as the amount of time patients wore the pedometer, causing the measure of adherence to “only be considered an estimate.”
The researchers reported technical difficulties with the devices, creating missing data.
The study was supported by the Rheumatology Research Foundation. The researchers reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
On Twitter @EAZTweets
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: Mean fatigue scores dropped –3.2 (P = .02) and –4.8 (P = .0002) for patients given only pedometers and pedometers with step goals, respectively.
Data source: Controlled study of 96 rheumatoid arthritis patients observed over a 21-week period.
Disclosures: The study was supported by the Rheumatology Research Foundation. The researchers reported no relevant financial disclosures.
Stopping TNF inhibitors for pregnancy may invite flares
Women with rheumatoid arthritis or axial spondyloarthritis who stop treatment with tumor necrosis factor inhibitors when they become pregnant may be inviting disease flares during the pregnancy, according to a report published in Arthritis Research & Therapy.
To examine the frequency of rheumatoid arthritis (RA) and axial spondyloarthritis (axSpA) flares during pregnancy, researchers prospectively followed 136 women treated at the Center for Pregnancy in Rheumatic Diseases at Inselspital Bern (Switzerland) during a 5-year period. These patients – 75 with RA and 61 with axSpA – were assessed before conception, during each trimester, and 6-8 weeks postpartum for disease activity and medication use, said Stephanie van den Brandt, MD, of the department of rheumatology, immunology, and allergology at the University of Bern, and her associates.
The relative risk of a disease flare was 3.33 among RA patients and 3.08 among axSpA patients who discontinued TNF inhibitors at the time of a positive pregnancy test. In comparison, rheumatic disease remained stable throughout pregnancy in most women who were not taking TNF inhibitors before pregnancy, the investigators said (Arthritis Res Ther. 2017 Mar 20. doi: 10.1186/s13075-017-1269-1).
Most disease flares occurred in the first trimester among women with RA and in the second half of pregnancy among women with axSpA. Most women with RA who resumed taking TNF inhibitors when their disease flared responded well to the treatment, with CRP levels dropping by 70% and remission being achieved rapidly. In contrast, most women with axSpA who resumed taking TNF inhibitors did not respond as well, with CRP levels dropping by only 35%. Their disease was ameliorated but not controlled by restarting the therapy.
No sponsor was cited for this study. Dr. van den Brandt and her associates reported having no relevant financial disclosures.
Women with rheumatoid arthritis or axial spondyloarthritis who stop treatment with tumor necrosis factor inhibitors when they become pregnant may be inviting disease flares during the pregnancy, according to a report published in Arthritis Research & Therapy.
To examine the frequency of rheumatoid arthritis (RA) and axial spondyloarthritis (axSpA) flares during pregnancy, researchers prospectively followed 136 women treated at the Center for Pregnancy in Rheumatic Diseases at Inselspital Bern (Switzerland) during a 5-year period. These patients – 75 with RA and 61 with axSpA – were assessed before conception, during each trimester, and 6-8 weeks postpartum for disease activity and medication use, said Stephanie van den Brandt, MD, of the department of rheumatology, immunology, and allergology at the University of Bern, and her associates.
The relative risk of a disease flare was 3.33 among RA patients and 3.08 among axSpA patients who discontinued TNF inhibitors at the time of a positive pregnancy test. In comparison, rheumatic disease remained stable throughout pregnancy in most women who were not taking TNF inhibitors before pregnancy, the investigators said (Arthritis Res Ther. 2017 Mar 20. doi: 10.1186/s13075-017-1269-1).
Most disease flares occurred in the first trimester among women with RA and in the second half of pregnancy among women with axSpA. Most women with RA who resumed taking TNF inhibitors when their disease flared responded well to the treatment, with CRP levels dropping by 70% and remission being achieved rapidly. In contrast, most women with axSpA who resumed taking TNF inhibitors did not respond as well, with CRP levels dropping by only 35%. Their disease was ameliorated but not controlled by restarting the therapy.
No sponsor was cited for this study. Dr. van den Brandt and her associates reported having no relevant financial disclosures.
Women with rheumatoid arthritis or axial spondyloarthritis who stop treatment with tumor necrosis factor inhibitors when they become pregnant may be inviting disease flares during the pregnancy, according to a report published in Arthritis Research & Therapy.
To examine the frequency of rheumatoid arthritis (RA) and axial spondyloarthritis (axSpA) flares during pregnancy, researchers prospectively followed 136 women treated at the Center for Pregnancy in Rheumatic Diseases at Inselspital Bern (Switzerland) during a 5-year period. These patients – 75 with RA and 61 with axSpA – were assessed before conception, during each trimester, and 6-8 weeks postpartum for disease activity and medication use, said Stephanie van den Brandt, MD, of the department of rheumatology, immunology, and allergology at the University of Bern, and her associates.
The relative risk of a disease flare was 3.33 among RA patients and 3.08 among axSpA patients who discontinued TNF inhibitors at the time of a positive pregnancy test. In comparison, rheumatic disease remained stable throughout pregnancy in most women who were not taking TNF inhibitors before pregnancy, the investigators said (Arthritis Res Ther. 2017 Mar 20. doi: 10.1186/s13075-017-1269-1).
Most disease flares occurred in the first trimester among women with RA and in the second half of pregnancy among women with axSpA. Most women with RA who resumed taking TNF inhibitors when their disease flared responded well to the treatment, with CRP levels dropping by 70% and remission being achieved rapidly. In contrast, most women with axSpA who resumed taking TNF inhibitors did not respond as well, with CRP levels dropping by only 35%. Their disease was ameliorated but not controlled by restarting the therapy.
No sponsor was cited for this study. Dr. van den Brandt and her associates reported having no relevant financial disclosures.
Key clinical point:
Major finding: The relative risk of a disease flare was 3.33 among RA patients and 3.08 among axSpA patients who discontinued TNF inhibitors at conception.
Data source: A prospective cohort study involving 75 pregnant women with RA and 61 with axial spondyloarthritis treated at one Swiss specialty center in 2000-2015.
Disclosures: No sponsor was cited for this study. Dr. van den Brandt and her associates reported having no relevant financial disclosures.
New antibody shows potential as prognostic marker in early rheumatoid arthritis
Individuals with rheumatoid arthritis who have antifibrillar collagen type II antibodies appear to have a unique disease phenotype characterized by acute but transient inflammation around the time of diagnosis, as well as a favorable prognosis, according to findings from a retrospective cohort study.
The antifibrillar collagen type II (anti-CII) phenotype also differed from the phenotype of individuals with anti–cyclic citrullinated peptide 2 (anti-CCP2) antibodies who typically exhibit inflammatory markers and higher disease activity later in the disease course, reported Vivek Anand Manivel of Uppsala (Sweden) University and his colleagues (Ann Rheum Dis. 2017 Mar 23. doi: 10.1136/annrheumdis-2016-210873).
Overall, the investigators detected anti-CII positivity in 6.6% and anti-CCP2 in 57.9%, including only anti-CII antibodies in 2.6%, only anti-CCP2 in 54%, double positivity in 3.9%, and double negativity in 39.4%. A control group of 960 patients from the Swedish general population who were matched for age, locality, and sex were positive for anti-CII in 1.6% and anti-CCP2 in 1.7%.
Significant differences were discovered between anti-CII positive and negative groups for C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), swollen joint count (SJC), and Disease Activity Score encompassing 28 joints (DAS28). Anti-CII seropositive patients had higher CRP at 0-6 months, higher ESR at 0-3 months, a higher SJC at 0-6 months, and a higher DAS28 at 0-3 months.
“Anti-CCP2 [seropositivity] on the other hand was associated with higher disease activity later during the 5-year follow-up, and with CRP and ESR during the full period,” the investigators wrote.
The same respective phenotypic patterns occurred when the comparisons were limited to patients with anti-CII positivity alone or anti-CCP2 positivity alone, the investigators noted. However, “patients in the anti-CII and anti-CCP2 double-positive group mainly followed the anti-CII pattern, with early but not late increased values for CRP, ESR, DAS28, and DAS28CRP, but with a mixed pattern for SJC.”
In comparisons where baseline clinical and laboratory measures were associated with anti-CII or anti-CCP2 positivity, anti-CII was more often associated with favorable late changes in CRP, ESR, SJC, TJC, DAS28, DAS28CRP, Health Assessment Questionnaire, and pain and global measurements on a visual analog scale when compared with antibody double-negative patients. There were fewer changes in clinical and laboratory measures associated with anti-CCP2 positivity. These significant changes associated with anti-CII positivity were always larger improvements than were seen for antibody double-negative patients, whereas the significant changes observed with anti-CCP2 patients were always smaller improvements than those for antibody double-negative patients.
Moderate or good European League Against Rheumatism responses were significantly associated with anti-CII positivity at 12, 24, 36, and 60 months, whereas they were negatively associated with anti-CCP2 positivity at 36 and 60 months.
Overall, the findings of the study show that since “anti-CII-positive patients with RA have an acute onset, but favorable prognosis” and anti-CCP2 is associated with poor prognosis, “the combined analysis of anti-CII and ACPA/anti-CCP2 may be a new two-dimensional tool for predicting the prognosis and choosing therapy in newly diagnosed patients with RA,” the authors suggested.
The study was funded by grants from the Swedish Research Council, Swedish Rheumatism Association, King Gustav Vth 80-Year Foundation, and Uppsala County Council. Dr. Manivel and his coauthors reported no relevant financial disclosures.
Individuals with rheumatoid arthritis who have antifibrillar collagen type II antibodies appear to have a unique disease phenotype characterized by acute but transient inflammation around the time of diagnosis, as well as a favorable prognosis, according to findings from a retrospective cohort study.
The antifibrillar collagen type II (anti-CII) phenotype also differed from the phenotype of individuals with anti–cyclic citrullinated peptide 2 (anti-CCP2) antibodies who typically exhibit inflammatory markers and higher disease activity later in the disease course, reported Vivek Anand Manivel of Uppsala (Sweden) University and his colleagues (Ann Rheum Dis. 2017 Mar 23. doi: 10.1136/annrheumdis-2016-210873).
Overall, the investigators detected anti-CII positivity in 6.6% and anti-CCP2 in 57.9%, including only anti-CII antibodies in 2.6%, only anti-CCP2 in 54%, double positivity in 3.9%, and double negativity in 39.4%. A control group of 960 patients from the Swedish general population who were matched for age, locality, and sex were positive for anti-CII in 1.6% and anti-CCP2 in 1.7%.
Significant differences were discovered between anti-CII positive and negative groups for C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), swollen joint count (SJC), and Disease Activity Score encompassing 28 joints (DAS28). Anti-CII seropositive patients had higher CRP at 0-6 months, higher ESR at 0-3 months, a higher SJC at 0-6 months, and a higher DAS28 at 0-3 months.
“Anti-CCP2 [seropositivity] on the other hand was associated with higher disease activity later during the 5-year follow-up, and with CRP and ESR during the full period,” the investigators wrote.
The same respective phenotypic patterns occurred when the comparisons were limited to patients with anti-CII positivity alone or anti-CCP2 positivity alone, the investigators noted. However, “patients in the anti-CII and anti-CCP2 double-positive group mainly followed the anti-CII pattern, with early but not late increased values for CRP, ESR, DAS28, and DAS28CRP, but with a mixed pattern for SJC.”
In comparisons where baseline clinical and laboratory measures were associated with anti-CII or anti-CCP2 positivity, anti-CII was more often associated with favorable late changes in CRP, ESR, SJC, TJC, DAS28, DAS28CRP, Health Assessment Questionnaire, and pain and global measurements on a visual analog scale when compared with antibody double-negative patients. There were fewer changes in clinical and laboratory measures associated with anti-CCP2 positivity. These significant changes associated with anti-CII positivity were always larger improvements than were seen for antibody double-negative patients, whereas the significant changes observed with anti-CCP2 patients were always smaller improvements than those for antibody double-negative patients.
Moderate or good European League Against Rheumatism responses were significantly associated with anti-CII positivity at 12, 24, 36, and 60 months, whereas they were negatively associated with anti-CCP2 positivity at 36 and 60 months.
Overall, the findings of the study show that since “anti-CII-positive patients with RA have an acute onset, but favorable prognosis” and anti-CCP2 is associated with poor prognosis, “the combined analysis of anti-CII and ACPA/anti-CCP2 may be a new two-dimensional tool for predicting the prognosis and choosing therapy in newly diagnosed patients with RA,” the authors suggested.
The study was funded by grants from the Swedish Research Council, Swedish Rheumatism Association, King Gustav Vth 80-Year Foundation, and Uppsala County Council. Dr. Manivel and his coauthors reported no relevant financial disclosures.
Individuals with rheumatoid arthritis who have antifibrillar collagen type II antibodies appear to have a unique disease phenotype characterized by acute but transient inflammation around the time of diagnosis, as well as a favorable prognosis, according to findings from a retrospective cohort study.
The antifibrillar collagen type II (anti-CII) phenotype also differed from the phenotype of individuals with anti–cyclic citrullinated peptide 2 (anti-CCP2) antibodies who typically exhibit inflammatory markers and higher disease activity later in the disease course, reported Vivek Anand Manivel of Uppsala (Sweden) University and his colleagues (Ann Rheum Dis. 2017 Mar 23. doi: 10.1136/annrheumdis-2016-210873).
Overall, the investigators detected anti-CII positivity in 6.6% and anti-CCP2 in 57.9%, including only anti-CII antibodies in 2.6%, only anti-CCP2 in 54%, double positivity in 3.9%, and double negativity in 39.4%. A control group of 960 patients from the Swedish general population who were matched for age, locality, and sex were positive for anti-CII in 1.6% and anti-CCP2 in 1.7%.
Significant differences were discovered between anti-CII positive and negative groups for C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), swollen joint count (SJC), and Disease Activity Score encompassing 28 joints (DAS28). Anti-CII seropositive patients had higher CRP at 0-6 months, higher ESR at 0-3 months, a higher SJC at 0-6 months, and a higher DAS28 at 0-3 months.
“Anti-CCP2 [seropositivity] on the other hand was associated with higher disease activity later during the 5-year follow-up, and with CRP and ESR during the full period,” the investigators wrote.
The same respective phenotypic patterns occurred when the comparisons were limited to patients with anti-CII positivity alone or anti-CCP2 positivity alone, the investigators noted. However, “patients in the anti-CII and anti-CCP2 double-positive group mainly followed the anti-CII pattern, with early but not late increased values for CRP, ESR, DAS28, and DAS28CRP, but with a mixed pattern for SJC.”
In comparisons where baseline clinical and laboratory measures were associated with anti-CII or anti-CCP2 positivity, anti-CII was more often associated with favorable late changes in CRP, ESR, SJC, TJC, DAS28, DAS28CRP, Health Assessment Questionnaire, and pain and global measurements on a visual analog scale when compared with antibody double-negative patients. There were fewer changes in clinical and laboratory measures associated with anti-CCP2 positivity. These significant changes associated with anti-CII positivity were always larger improvements than were seen for antibody double-negative patients, whereas the significant changes observed with anti-CCP2 patients were always smaller improvements than those for antibody double-negative patients.
Moderate or good European League Against Rheumatism responses were significantly associated with anti-CII positivity at 12, 24, 36, and 60 months, whereas they were negatively associated with anti-CCP2 positivity at 36 and 60 months.
Overall, the findings of the study show that since “anti-CII-positive patients with RA have an acute onset, but favorable prognosis” and anti-CCP2 is associated with poor prognosis, “the combined analysis of anti-CII and ACPA/anti-CCP2 may be a new two-dimensional tool for predicting the prognosis and choosing therapy in newly diagnosed patients with RA,” the authors suggested.
The study was funded by grants from the Swedish Research Council, Swedish Rheumatism Association, King Gustav Vth 80-Year Foundation, and Uppsala County Council. Dr. Manivel and his coauthors reported no relevant financial disclosures.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point:
Major finding: RA patients with anti-CII seropositivity had significantly higher C-reactive protein levels at 0-6 months, higher ESR at 0-3 months, a higher SJC at 0-6 months, and a higher DAS28 at 0-3 months than did anti-CII seronegative patients.
Data source: Retrospective cohort study of EIRA study patients and 960 controls.
Disclosures: Study funded by Swedish Research Council, Swedish Rheumatism Association, King Gustav Vth 80-Year Foundation, and Uppsala County Council. The authors reported no relevant financial disclosures.
Get ready for cancer immunotherapy-induced rheumatic diseases
SNOWMASS, COLO. – Physicians can expect to encounter more and more patients with inflammatory arthritis and other rheumatic adverse events induced by immune checkpoint inhibitors as a result of anticipated exponential growth in the use of these drugs to treat an expanding list of cancers, Clifton O. Bingham III, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
These cancer immunotherapy–induced rheumatic diseases may superficially look like the classic forms of familiar autoimmune diseases, but they have highly atypical features that will affect treatment decisions.
“What we’ve seen consistently is that the normal doses of prednisone we would use to treat an inflammatory arthritis are really ineffective in most of these patients. We’ve had to use super doses – up to 120 mg/day – for initial control, and then 7.5-40 mg daily for maintenance of response,” according to Dr. Bingham, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
To date, only limited data from case series are available on rheumatic IRAEs. There are no prospective patient registries logging accurate data on the incidence of these rheumatic adverse events among cancer patients treated with immune checkpoint inhibitors (ICIs). These IRAEs, which lie at the intersection of rheumatology and oncology, are of special interest to Dr. Bingham – he and his coinvestigators have published five articles on the topic over the course of a single year.
In a wide-ranging talk at the symposium, he touched on the phenotypic spectrum of rheumatologic IRAEs, his conviction that they are greatly underdiagnosed, why physicians can expect to encounter them much more frequently, rheumatologic IRAE treatment issues, and the risks of prescribing ICIs in patients with known preexisting rheumatologic disease.
Rheumatologic IRAE presentations
Inflammatory arthritis is the most common form of rheumatologic IRAE, followed by sicca syndrome. At the Johns Hopkins Arthritis Center, Dr. Bingham and his coworkers have 25 well-characterized patients with inflammatory arthritis resulting from an ICI, only 1 of whom is HLA-B27-positive.
“Also, just one is autoantibody-positive, even though they all look for all the world as though they have rheumatoid arthritis,” the rheumatologist observed.
This ICI-induced inflammatory arthritis initially presents most commonly in the midsize and large joints – knees, ankles, elbows – then expands to include small joints such as the wrists, proximal interphalangeal joints, and the metacarpophalangeal joints.
Notably, the Hopkins group also has three patients with classic reactive arthritis marked by conjunctivitis, urethritis, arthritis, and dactylitis.
“I don’t know about you, but, in our general rheumatology practice, we see maybe one case of reactive arthritis in several years, so this is something that has struck us as really quite interesting,” said Dr. Bingham, who is also director of research in the division of rheumatology at Johns Hopkins.
The arthritis center is also managing a group of patients with ICI-induced sicca syndrome, which is uniformly extremely severe and treatment resistant, as well as a couple of patients with myositis IRAE, one with polymyalgia rheumatica, and two with crystal disease that is highly inflammatory in nature, difficult to treat, and includes an inflammatory polyarthritis component not typical in patients with crystal arthritis.
Why physicians will see more rheumatologic IRAEs
ICIs have dramatically transformed the treatment of selected advanced-stage cancers. For example, whereas patients with metastatic melanoma historically had a 2-year survival rate of 5%, combination therapy with the ICIs ipilimumab (Yervoy) and nivolumab (Opdivo) resulted in a 60% rate of partial or complete remission in a landmark clinical trial.
The basis of cancer immunotherapy is the discovery that, in order for cancer cells to thrive, they emit blocking signals that downregulate the native ability of T cells to recognize and kill them. This is true for both solid tumors and hematologic malignancies. The ICIs inhibit these blocking signals, which include cytotoxic T-lymphocyte–associated protein 4 (CTLA4), programmed death-1 (PD-1), and programmed death ligand-1 (PDL-1), thereby freeing up the T cells for tumor fighting.
Because of the nonspecific mechanism of this T-cell activation, however, ICIs have, as their main toxicities, T-cell–mediated autoimmune inflammatory tissue damage, which gets lumped under the umbrella term IRAEs. It can affect almost every organ system. Skin rashes are the most common, colitis second. Other commonly encountered IRAEs include thyroiditis, hypophysitis, hepatitis, peripheral neuropathy, and pneumonitis.
In addition to the four currently approved ICIs – ipilimumab, nivolumab, pembrolizumab (Keytruda), and atezolizumab (Tecentriq) – investigational ICIs targeting CTLA4, PD-1, and/or PDL-1 are in development. Plus, new ICIs targeting other blocking signals, including lymphocyte activation gene-3, CD137, and T-cell immunoglobulin and mucin domain-3, are now in clinical trials.
Clinical trials aimed at expanding the indications of existing ICIs and using ICIs in earlier-stage cancers in an effort to improve rates of lasting remission are also underway.
All told, probably at least 400 clinical trials of ICIs are ongoing worldwide, the rheumatologist estimated.
“More people will be exposed to these drugs, and we’ll see more and more of these rheumatologic IRAEs,” Dr. Bingham predicted.
Rheumatologic IRAEs are seriously underdiagnosed
Back in the pre-ICI days, Dr. Bingham was coauthor of a major study which concluded that clinical trialists in oncology consistently downgrade the severity of rheumatologic adverse events, often by 1 or 2 grades (J Rheumatol. 2007 Jun;34[6]:1401-14).
Unpublished details of ICI clinical trials in melanoma that he obtained from Bristol-Myers Squibb suggest that the true rate of rheumatologic IRAEs is about 20%, or roughly double that reported in the studies. That’s because the adverse events–grading system used in oncology undercalls the severity of arthritis and autoimmune disorders.
Indeed, the National Cancer Institute’s Common Terminology Criteria for Adverse Events, used in oncology clinical trials, is confusing on the topic of musculoskeletal and connective tissue disorders as treatment-emergent adverse events, according to Dr. Bingham. He noted that an oncologist can code a swollen joint in three different ways – joint effusion, arthritis, or arthralgia – and it takes disabling interference with self-care in activities of daily living for that swollen joint to rise to the level of a Grade 3 adverse event. From a rheumatology trialist’s perspective, that would be a Grade 4 disability.
Plus, neither the product labeling nor the patient information guides for the approved immunotherapy drugs mention the importance of monitoring for rheumatologic IRAEs or their management.
“There is poor awareness of musculoskeletal and rheumatic IRAEs in the general oncology community,” Dr. Bingham asserted. “But, if you talk with any oncology nurses who work in a clinical trial, they will tell you they’re seeing these events with significant frequency and severity.”
Treatment and response
It’s critical to gain control of rheumatologic IRAEs quickly so that patients can get on with their cancer immunotherapy. Dr. Bingham uses intra-articular steroid injections for patients with oligoarthritis and high-dose oral prednisone for polyarticular disease. He starts methotrexate and/or leflunomide early because the conventional disease-modifying antirheumatic drugs have roughly a 2-month delay in onset of action. He has had several patients who are unable to taper steroids despite background methotrexate.
In the most severely affected patients, he has turned to biologic agents in consultation with their oncologists. Tumor necrosis factor (TNF) inhibitors are the ones he and other rheumatologists have used most often.
“Notably, we have not been able to taper down very well. We have patients who are out more than 2 years now who still require their TNF inhibitor to treat their inflammatory arthritis, and these are patients on conventional disease–modifying antirheumatic drugs as well. As soon as it’s tapered, the arthritis begins to come back,” according to Dr. Bingham.
In marked contrast, colitis as an IRAE typically clears in response to just one or two doses of a TNF inhibitor.
One audience member related that she’d encountered a roadblock in trying to get authorization for a TNF inhibitor for a patient with a rheumatologic IRAE secondary to ICI treatment for metastatic melanoma because the labeling states these agents are relatively contraindicated in melanoma patients. Dr. Bingham offered a tip: Collaborate with the patient’s oncologist.
“In most cases, oncologists can get infliximab for these patients and administer it in their infusion centers. They are able to get things authorized with very little trouble,” he said.
Besides, most of these patients with severe inflammatory arthritis meet conventional criteria for TNF inhibitor therapy, based on their number of infected joints and elevated acute phase reactants for longer than 6 weeks, Dr. Bingham noted.
“We’ve had some very interesting conversations with patients. It’s impressive to see the impact arthritis can have on people. A lot of patients have said, ‘I don’t care if I die. Get me functional right now.’ That’s pretty profound. Quality of life is still very important for people, even when dealing with life-threatening diseases,” he observed.
Oncologists are actually eager for their patients to get on steroid-sparing therapy because of concern that high doses of steroids may reduce the efficacy of cancer immunotherapy. That’s not an issue with the TNF inhibitors, the rheumatologist continued.
Turning to the utility of other classes of biologic agents, Dr. Bingham advised avoiding abatacept (Orencia) because its mechanism of action is likely to cause interference with the cancer immunotherapy. Rituximab (Rituxan) takes too long to act. Anakinra (Kineret), tofacitinib (Xeljanz), and tocilizumab (Actemra), on the other hand, are agents he is interested in using as alternatives to TNF inhibitors, although he hasn’t done so yet.
Use of ICIs in patients with preexisting autoimmune disease
The experience here is entirely anecdotal, since such patients have been excluded from ICI clinical trials, but the available evidence suggests physicians should be prepared for higher rheumatologic IRAE rates in this setting. Investigators at Vanderbilt University reported that 8 of 30 cancer patients with known preexisting autoimmune disease experienced flares of that disease when treated with ipilimumab, and 10 developed a new IRAE (Therap Adv Gastroenterol. 2016 Jul;9[4]:457-62).
The Hopkins group has three patients with preexisting rheumatoid arthritis and two with preexisting scleroderma who have received ICIs. All three rheumatoid arthritis patients flared. Rheumatologists are trying to manage these flares so the patients can continue on their ICI. One of the scleroderma patients experienced no change in that disease while on an ICI, while the other showed a definite improvement in scleroderma symptoms.
“I think the jury’s still out in terms of what you do about ICI therapy in patients with preexisting autoimmunity. The data would say that there’s maybe a 50-50 chance of the autoimmune disease becoming worse, but, if patients have an otherwise fatal cancer, I think it’s probably worth the chance,” Dr. Bingham said.
Anecdotal reports suggest that more severe IRAEs may be a favorable prognostic sign in terms of cancer eradication, but a lot more patient experience will be needed in order to be sure, the rheumatologist said.
Dr. Bingham reported serving as a consultant to Bristol-Myers Squibb.
SNOWMASS, COLO. – Physicians can expect to encounter more and more patients with inflammatory arthritis and other rheumatic adverse events induced by immune checkpoint inhibitors as a result of anticipated exponential growth in the use of these drugs to treat an expanding list of cancers, Clifton O. Bingham III, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
These cancer immunotherapy–induced rheumatic diseases may superficially look like the classic forms of familiar autoimmune diseases, but they have highly atypical features that will affect treatment decisions.
“What we’ve seen consistently is that the normal doses of prednisone we would use to treat an inflammatory arthritis are really ineffective in most of these patients. We’ve had to use super doses – up to 120 mg/day – for initial control, and then 7.5-40 mg daily for maintenance of response,” according to Dr. Bingham, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
To date, only limited data from case series are available on rheumatic IRAEs. There are no prospective patient registries logging accurate data on the incidence of these rheumatic adverse events among cancer patients treated with immune checkpoint inhibitors (ICIs). These IRAEs, which lie at the intersection of rheumatology and oncology, are of special interest to Dr. Bingham – he and his coinvestigators have published five articles on the topic over the course of a single year.
In a wide-ranging talk at the symposium, he touched on the phenotypic spectrum of rheumatologic IRAEs, his conviction that they are greatly underdiagnosed, why physicians can expect to encounter them much more frequently, rheumatologic IRAE treatment issues, and the risks of prescribing ICIs in patients with known preexisting rheumatologic disease.
Rheumatologic IRAE presentations
Inflammatory arthritis is the most common form of rheumatologic IRAE, followed by sicca syndrome. At the Johns Hopkins Arthritis Center, Dr. Bingham and his coworkers have 25 well-characterized patients with inflammatory arthritis resulting from an ICI, only 1 of whom is HLA-B27-positive.
“Also, just one is autoantibody-positive, even though they all look for all the world as though they have rheumatoid arthritis,” the rheumatologist observed.
This ICI-induced inflammatory arthritis initially presents most commonly in the midsize and large joints – knees, ankles, elbows – then expands to include small joints such as the wrists, proximal interphalangeal joints, and the metacarpophalangeal joints.
Notably, the Hopkins group also has three patients with classic reactive arthritis marked by conjunctivitis, urethritis, arthritis, and dactylitis.
“I don’t know about you, but, in our general rheumatology practice, we see maybe one case of reactive arthritis in several years, so this is something that has struck us as really quite interesting,” said Dr. Bingham, who is also director of research in the division of rheumatology at Johns Hopkins.
The arthritis center is also managing a group of patients with ICI-induced sicca syndrome, which is uniformly extremely severe and treatment resistant, as well as a couple of patients with myositis IRAE, one with polymyalgia rheumatica, and two with crystal disease that is highly inflammatory in nature, difficult to treat, and includes an inflammatory polyarthritis component not typical in patients with crystal arthritis.
Why physicians will see more rheumatologic IRAEs
ICIs have dramatically transformed the treatment of selected advanced-stage cancers. For example, whereas patients with metastatic melanoma historically had a 2-year survival rate of 5%, combination therapy with the ICIs ipilimumab (Yervoy) and nivolumab (Opdivo) resulted in a 60% rate of partial or complete remission in a landmark clinical trial.
The basis of cancer immunotherapy is the discovery that, in order for cancer cells to thrive, they emit blocking signals that downregulate the native ability of T cells to recognize and kill them. This is true for both solid tumors and hematologic malignancies. The ICIs inhibit these blocking signals, which include cytotoxic T-lymphocyte–associated protein 4 (CTLA4), programmed death-1 (PD-1), and programmed death ligand-1 (PDL-1), thereby freeing up the T cells for tumor fighting.
Because of the nonspecific mechanism of this T-cell activation, however, ICIs have, as their main toxicities, T-cell–mediated autoimmune inflammatory tissue damage, which gets lumped under the umbrella term IRAEs. It can affect almost every organ system. Skin rashes are the most common, colitis second. Other commonly encountered IRAEs include thyroiditis, hypophysitis, hepatitis, peripheral neuropathy, and pneumonitis.
In addition to the four currently approved ICIs – ipilimumab, nivolumab, pembrolizumab (Keytruda), and atezolizumab (Tecentriq) – investigational ICIs targeting CTLA4, PD-1, and/or PDL-1 are in development. Plus, new ICIs targeting other blocking signals, including lymphocyte activation gene-3, CD137, and T-cell immunoglobulin and mucin domain-3, are now in clinical trials.
Clinical trials aimed at expanding the indications of existing ICIs and using ICIs in earlier-stage cancers in an effort to improve rates of lasting remission are also underway.
All told, probably at least 400 clinical trials of ICIs are ongoing worldwide, the rheumatologist estimated.
“More people will be exposed to these drugs, and we’ll see more and more of these rheumatologic IRAEs,” Dr. Bingham predicted.
Rheumatologic IRAEs are seriously underdiagnosed
Back in the pre-ICI days, Dr. Bingham was coauthor of a major study which concluded that clinical trialists in oncology consistently downgrade the severity of rheumatologic adverse events, often by 1 or 2 grades (J Rheumatol. 2007 Jun;34[6]:1401-14).
Unpublished details of ICI clinical trials in melanoma that he obtained from Bristol-Myers Squibb suggest that the true rate of rheumatologic IRAEs is about 20%, or roughly double that reported in the studies. That’s because the adverse events–grading system used in oncology undercalls the severity of arthritis and autoimmune disorders.
Indeed, the National Cancer Institute’s Common Terminology Criteria for Adverse Events, used in oncology clinical trials, is confusing on the topic of musculoskeletal and connective tissue disorders as treatment-emergent adverse events, according to Dr. Bingham. He noted that an oncologist can code a swollen joint in three different ways – joint effusion, arthritis, or arthralgia – and it takes disabling interference with self-care in activities of daily living for that swollen joint to rise to the level of a Grade 3 adverse event. From a rheumatology trialist’s perspective, that would be a Grade 4 disability.
Plus, neither the product labeling nor the patient information guides for the approved immunotherapy drugs mention the importance of monitoring for rheumatologic IRAEs or their management.
“There is poor awareness of musculoskeletal and rheumatic IRAEs in the general oncology community,” Dr. Bingham asserted. “But, if you talk with any oncology nurses who work in a clinical trial, they will tell you they’re seeing these events with significant frequency and severity.”
Treatment and response
It’s critical to gain control of rheumatologic IRAEs quickly so that patients can get on with their cancer immunotherapy. Dr. Bingham uses intra-articular steroid injections for patients with oligoarthritis and high-dose oral prednisone for polyarticular disease. He starts methotrexate and/or leflunomide early because the conventional disease-modifying antirheumatic drugs have roughly a 2-month delay in onset of action. He has had several patients who are unable to taper steroids despite background methotrexate.
In the most severely affected patients, he has turned to biologic agents in consultation with their oncologists. Tumor necrosis factor (TNF) inhibitors are the ones he and other rheumatologists have used most often.
“Notably, we have not been able to taper down very well. We have patients who are out more than 2 years now who still require their TNF inhibitor to treat their inflammatory arthritis, and these are patients on conventional disease–modifying antirheumatic drugs as well. As soon as it’s tapered, the arthritis begins to come back,” according to Dr. Bingham.
In marked contrast, colitis as an IRAE typically clears in response to just one or two doses of a TNF inhibitor.
One audience member related that she’d encountered a roadblock in trying to get authorization for a TNF inhibitor for a patient with a rheumatologic IRAE secondary to ICI treatment for metastatic melanoma because the labeling states these agents are relatively contraindicated in melanoma patients. Dr. Bingham offered a tip: Collaborate with the patient’s oncologist.
“In most cases, oncologists can get infliximab for these patients and administer it in their infusion centers. They are able to get things authorized with very little trouble,” he said.
Besides, most of these patients with severe inflammatory arthritis meet conventional criteria for TNF inhibitor therapy, based on their number of infected joints and elevated acute phase reactants for longer than 6 weeks, Dr. Bingham noted.
“We’ve had some very interesting conversations with patients. It’s impressive to see the impact arthritis can have on people. A lot of patients have said, ‘I don’t care if I die. Get me functional right now.’ That’s pretty profound. Quality of life is still very important for people, even when dealing with life-threatening diseases,” he observed.
Oncologists are actually eager for their patients to get on steroid-sparing therapy because of concern that high doses of steroids may reduce the efficacy of cancer immunotherapy. That’s not an issue with the TNF inhibitors, the rheumatologist continued.
Turning to the utility of other classes of biologic agents, Dr. Bingham advised avoiding abatacept (Orencia) because its mechanism of action is likely to cause interference with the cancer immunotherapy. Rituximab (Rituxan) takes too long to act. Anakinra (Kineret), tofacitinib (Xeljanz), and tocilizumab (Actemra), on the other hand, are agents he is interested in using as alternatives to TNF inhibitors, although he hasn’t done so yet.
Use of ICIs in patients with preexisting autoimmune disease
The experience here is entirely anecdotal, since such patients have been excluded from ICI clinical trials, but the available evidence suggests physicians should be prepared for higher rheumatologic IRAE rates in this setting. Investigators at Vanderbilt University reported that 8 of 30 cancer patients with known preexisting autoimmune disease experienced flares of that disease when treated with ipilimumab, and 10 developed a new IRAE (Therap Adv Gastroenterol. 2016 Jul;9[4]:457-62).
The Hopkins group has three patients with preexisting rheumatoid arthritis and two with preexisting scleroderma who have received ICIs. All three rheumatoid arthritis patients flared. Rheumatologists are trying to manage these flares so the patients can continue on their ICI. One of the scleroderma patients experienced no change in that disease while on an ICI, while the other showed a definite improvement in scleroderma symptoms.
“I think the jury’s still out in terms of what you do about ICI therapy in patients with preexisting autoimmunity. The data would say that there’s maybe a 50-50 chance of the autoimmune disease becoming worse, but, if patients have an otherwise fatal cancer, I think it’s probably worth the chance,” Dr. Bingham said.
Anecdotal reports suggest that more severe IRAEs may be a favorable prognostic sign in terms of cancer eradication, but a lot more patient experience will be needed in order to be sure, the rheumatologist said.
Dr. Bingham reported serving as a consultant to Bristol-Myers Squibb.
SNOWMASS, COLO. – Physicians can expect to encounter more and more patients with inflammatory arthritis and other rheumatic adverse events induced by immune checkpoint inhibitors as a result of anticipated exponential growth in the use of these drugs to treat an expanding list of cancers, Clifton O. Bingham III, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
These cancer immunotherapy–induced rheumatic diseases may superficially look like the classic forms of familiar autoimmune diseases, but they have highly atypical features that will affect treatment decisions.
“What we’ve seen consistently is that the normal doses of prednisone we would use to treat an inflammatory arthritis are really ineffective in most of these patients. We’ve had to use super doses – up to 120 mg/day – for initial control, and then 7.5-40 mg daily for maintenance of response,” according to Dr. Bingham, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
To date, only limited data from case series are available on rheumatic IRAEs. There are no prospective patient registries logging accurate data on the incidence of these rheumatic adverse events among cancer patients treated with immune checkpoint inhibitors (ICIs). These IRAEs, which lie at the intersection of rheumatology and oncology, are of special interest to Dr. Bingham – he and his coinvestigators have published five articles on the topic over the course of a single year.
In a wide-ranging talk at the symposium, he touched on the phenotypic spectrum of rheumatologic IRAEs, his conviction that they are greatly underdiagnosed, why physicians can expect to encounter them much more frequently, rheumatologic IRAE treatment issues, and the risks of prescribing ICIs in patients with known preexisting rheumatologic disease.
Rheumatologic IRAE presentations
Inflammatory arthritis is the most common form of rheumatologic IRAE, followed by sicca syndrome. At the Johns Hopkins Arthritis Center, Dr. Bingham and his coworkers have 25 well-characterized patients with inflammatory arthritis resulting from an ICI, only 1 of whom is HLA-B27-positive.
“Also, just one is autoantibody-positive, even though they all look for all the world as though they have rheumatoid arthritis,” the rheumatologist observed.
This ICI-induced inflammatory arthritis initially presents most commonly in the midsize and large joints – knees, ankles, elbows – then expands to include small joints such as the wrists, proximal interphalangeal joints, and the metacarpophalangeal joints.
Notably, the Hopkins group also has three patients with classic reactive arthritis marked by conjunctivitis, urethritis, arthritis, and dactylitis.
“I don’t know about you, but, in our general rheumatology practice, we see maybe one case of reactive arthritis in several years, so this is something that has struck us as really quite interesting,” said Dr. Bingham, who is also director of research in the division of rheumatology at Johns Hopkins.
The arthritis center is also managing a group of patients with ICI-induced sicca syndrome, which is uniformly extremely severe and treatment resistant, as well as a couple of patients with myositis IRAE, one with polymyalgia rheumatica, and two with crystal disease that is highly inflammatory in nature, difficult to treat, and includes an inflammatory polyarthritis component not typical in patients with crystal arthritis.
Why physicians will see more rheumatologic IRAEs
ICIs have dramatically transformed the treatment of selected advanced-stage cancers. For example, whereas patients with metastatic melanoma historically had a 2-year survival rate of 5%, combination therapy with the ICIs ipilimumab (Yervoy) and nivolumab (Opdivo) resulted in a 60% rate of partial or complete remission in a landmark clinical trial.
The basis of cancer immunotherapy is the discovery that, in order for cancer cells to thrive, they emit blocking signals that downregulate the native ability of T cells to recognize and kill them. This is true for both solid tumors and hematologic malignancies. The ICIs inhibit these blocking signals, which include cytotoxic T-lymphocyte–associated protein 4 (CTLA4), programmed death-1 (PD-1), and programmed death ligand-1 (PDL-1), thereby freeing up the T cells for tumor fighting.
Because of the nonspecific mechanism of this T-cell activation, however, ICIs have, as their main toxicities, T-cell–mediated autoimmune inflammatory tissue damage, which gets lumped under the umbrella term IRAEs. It can affect almost every organ system. Skin rashes are the most common, colitis second. Other commonly encountered IRAEs include thyroiditis, hypophysitis, hepatitis, peripheral neuropathy, and pneumonitis.
In addition to the four currently approved ICIs – ipilimumab, nivolumab, pembrolizumab (Keytruda), and atezolizumab (Tecentriq) – investigational ICIs targeting CTLA4, PD-1, and/or PDL-1 are in development. Plus, new ICIs targeting other blocking signals, including lymphocyte activation gene-3, CD137, and T-cell immunoglobulin and mucin domain-3, are now in clinical trials.
Clinical trials aimed at expanding the indications of existing ICIs and using ICIs in earlier-stage cancers in an effort to improve rates of lasting remission are also underway.
All told, probably at least 400 clinical trials of ICIs are ongoing worldwide, the rheumatologist estimated.
“More people will be exposed to these drugs, and we’ll see more and more of these rheumatologic IRAEs,” Dr. Bingham predicted.
Rheumatologic IRAEs are seriously underdiagnosed
Back in the pre-ICI days, Dr. Bingham was coauthor of a major study which concluded that clinical trialists in oncology consistently downgrade the severity of rheumatologic adverse events, often by 1 or 2 grades (J Rheumatol. 2007 Jun;34[6]:1401-14).
Unpublished details of ICI clinical trials in melanoma that he obtained from Bristol-Myers Squibb suggest that the true rate of rheumatologic IRAEs is about 20%, or roughly double that reported in the studies. That’s because the adverse events–grading system used in oncology undercalls the severity of arthritis and autoimmune disorders.
Indeed, the National Cancer Institute’s Common Terminology Criteria for Adverse Events, used in oncology clinical trials, is confusing on the topic of musculoskeletal and connective tissue disorders as treatment-emergent adverse events, according to Dr. Bingham. He noted that an oncologist can code a swollen joint in three different ways – joint effusion, arthritis, or arthralgia – and it takes disabling interference with self-care in activities of daily living for that swollen joint to rise to the level of a Grade 3 adverse event. From a rheumatology trialist’s perspective, that would be a Grade 4 disability.
Plus, neither the product labeling nor the patient information guides for the approved immunotherapy drugs mention the importance of monitoring for rheumatologic IRAEs or their management.
“There is poor awareness of musculoskeletal and rheumatic IRAEs in the general oncology community,” Dr. Bingham asserted. “But, if you talk with any oncology nurses who work in a clinical trial, they will tell you they’re seeing these events with significant frequency and severity.”
Treatment and response
It’s critical to gain control of rheumatologic IRAEs quickly so that patients can get on with their cancer immunotherapy. Dr. Bingham uses intra-articular steroid injections for patients with oligoarthritis and high-dose oral prednisone for polyarticular disease. He starts methotrexate and/or leflunomide early because the conventional disease-modifying antirheumatic drugs have roughly a 2-month delay in onset of action. He has had several patients who are unable to taper steroids despite background methotrexate.
In the most severely affected patients, he has turned to biologic agents in consultation with their oncologists. Tumor necrosis factor (TNF) inhibitors are the ones he and other rheumatologists have used most often.
“Notably, we have not been able to taper down very well. We have patients who are out more than 2 years now who still require their TNF inhibitor to treat their inflammatory arthritis, and these are patients on conventional disease–modifying antirheumatic drugs as well. As soon as it’s tapered, the arthritis begins to come back,” according to Dr. Bingham.
In marked contrast, colitis as an IRAE typically clears in response to just one or two doses of a TNF inhibitor.
One audience member related that she’d encountered a roadblock in trying to get authorization for a TNF inhibitor for a patient with a rheumatologic IRAE secondary to ICI treatment for metastatic melanoma because the labeling states these agents are relatively contraindicated in melanoma patients. Dr. Bingham offered a tip: Collaborate with the patient’s oncologist.
“In most cases, oncologists can get infliximab for these patients and administer it in their infusion centers. They are able to get things authorized with very little trouble,” he said.
Besides, most of these patients with severe inflammatory arthritis meet conventional criteria for TNF inhibitor therapy, based on their number of infected joints and elevated acute phase reactants for longer than 6 weeks, Dr. Bingham noted.
“We’ve had some very interesting conversations with patients. It’s impressive to see the impact arthritis can have on people. A lot of patients have said, ‘I don’t care if I die. Get me functional right now.’ That’s pretty profound. Quality of life is still very important for people, even when dealing with life-threatening diseases,” he observed.
Oncologists are actually eager for their patients to get on steroid-sparing therapy because of concern that high doses of steroids may reduce the efficacy of cancer immunotherapy. That’s not an issue with the TNF inhibitors, the rheumatologist continued.
Turning to the utility of other classes of biologic agents, Dr. Bingham advised avoiding abatacept (Orencia) because its mechanism of action is likely to cause interference with the cancer immunotherapy. Rituximab (Rituxan) takes too long to act. Anakinra (Kineret), tofacitinib (Xeljanz), and tocilizumab (Actemra), on the other hand, are agents he is interested in using as alternatives to TNF inhibitors, although he hasn’t done so yet.
Use of ICIs in patients with preexisting autoimmune disease
The experience here is entirely anecdotal, since such patients have been excluded from ICI clinical trials, but the available evidence suggests physicians should be prepared for higher rheumatologic IRAE rates in this setting. Investigators at Vanderbilt University reported that 8 of 30 cancer patients with known preexisting autoimmune disease experienced flares of that disease when treated with ipilimumab, and 10 developed a new IRAE (Therap Adv Gastroenterol. 2016 Jul;9[4]:457-62).
The Hopkins group has three patients with preexisting rheumatoid arthritis and two with preexisting scleroderma who have received ICIs. All three rheumatoid arthritis patients flared. Rheumatologists are trying to manage these flares so the patients can continue on their ICI. One of the scleroderma patients experienced no change in that disease while on an ICI, while the other showed a definite improvement in scleroderma symptoms.
“I think the jury’s still out in terms of what you do about ICI therapy in patients with preexisting autoimmunity. The data would say that there’s maybe a 50-50 chance of the autoimmune disease becoming worse, but, if patients have an otherwise fatal cancer, I think it’s probably worth the chance,” Dr. Bingham said.
Anecdotal reports suggest that more severe IRAEs may be a favorable prognostic sign in terms of cancer eradication, but a lot more patient experience will be needed in order to be sure, the rheumatologist said.
Dr. Bingham reported serving as a consultant to Bristol-Myers Squibb.
EXPERT ANALYSIS FROM THE WINTER RHEUMATOLOGY SYMPOSIUM
Shingles vaccine deemed effective in people with autoimmune disease
The herpes zoster vaccine reduces the risk of shingles in older adults with autoimmune disease, even if they are taking immunosuppressants for their condition, but the protection begins to wane after about 5 years, a recent retrospective study found.
“There has been some concern that patients with autoimmune conditions might have a lower immunogenic response to herpes zoster vaccination, especially when treated with immunosuppressive medications such as glucocorticoids,” wrote Huifeng Yun, PhD, of the University of Alabama at Birmingham, and her colleagues.
The researchers used 2006-2013 Medicare data to calculate the risk of shingles among Medicare recipients who had an autoimmune disease and either did or did not receive the herpes zoster vaccine. All the patients had been enrolled in Medicare for at least 12 continuous months and had a diagnosis of ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis.
The researchers matched 59,627 patients who received the herpes zoster vaccine with 119,254 unvaccinated patients, based on age, sex, race, calendar year, autoimmune disease type, and use of autoimmune drugs (biologics, disease-modifying antirheumatic drugs, and glucocorticoids). During a follow-up of up to 7 years, the researchers additionally accounted for comorbid medical conditions and concurrent medications each year.
The cohort, with an average age of 73.5 years in both groups, included 53.1% of adults with rheumatoid arthritis, 31.6% with psoriasis, 20.9% with inflammatory bowel disease, 4.7% with psoriatic arthritis, and 1.4% with ankylosing spondylitis.
Those who received the vaccine had a rate of 0.75 herpes zoster cases per 100 people during the first year, which rose to 1.25 cases per 100 people per year at the seventh year after vaccination. The rate among unvaccinated individuals stayed steady at approximately 1.3-1.7 cases per 100 people per year throughout the study period. These rates, as expected, were approximately 50% higher than in the general population over age 70 without autoimmune disease.
Compared with unvaccinated individuals, vaccinated individuals had a reduced relative risk for shingles of 0.74-0.77 after adjustment for confounders, but the risk reduction only remained statistically significant for the first 5 years after vaccination.
The waning seen with the vaccine’s effectiveness “raises the possibility that patients might benefit from a booster vaccine at some point after initial vaccination, although no recommendation currently exists that would support such a practice,” the authors wrote.
Dr. Yun has received research funding from Amgen. Other authors disclosed ties to Amgen, AstraZeneca, Bristol-Myers Squibb, Crescendo Bioscience, Janssen, and Pfizer. One author has received research support and consulting fees from Corrona. The study did not note an external source of funding.
The herpes zoster vaccine reduces the risk of shingles in older adults with autoimmune disease, even if they are taking immunosuppressants for their condition, but the protection begins to wane after about 5 years, a recent retrospective study found.
“There has been some concern that patients with autoimmune conditions might have a lower immunogenic response to herpes zoster vaccination, especially when treated with immunosuppressive medications such as glucocorticoids,” wrote Huifeng Yun, PhD, of the University of Alabama at Birmingham, and her colleagues.
The researchers used 2006-2013 Medicare data to calculate the risk of shingles among Medicare recipients who had an autoimmune disease and either did or did not receive the herpes zoster vaccine. All the patients had been enrolled in Medicare for at least 12 continuous months and had a diagnosis of ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis.
The researchers matched 59,627 patients who received the herpes zoster vaccine with 119,254 unvaccinated patients, based on age, sex, race, calendar year, autoimmune disease type, and use of autoimmune drugs (biologics, disease-modifying antirheumatic drugs, and glucocorticoids). During a follow-up of up to 7 years, the researchers additionally accounted for comorbid medical conditions and concurrent medications each year.
The cohort, with an average age of 73.5 years in both groups, included 53.1% of adults with rheumatoid arthritis, 31.6% with psoriasis, 20.9% with inflammatory bowel disease, 4.7% with psoriatic arthritis, and 1.4% with ankylosing spondylitis.
Those who received the vaccine had a rate of 0.75 herpes zoster cases per 100 people during the first year, which rose to 1.25 cases per 100 people per year at the seventh year after vaccination. The rate among unvaccinated individuals stayed steady at approximately 1.3-1.7 cases per 100 people per year throughout the study period. These rates, as expected, were approximately 50% higher than in the general population over age 70 without autoimmune disease.
Compared with unvaccinated individuals, vaccinated individuals had a reduced relative risk for shingles of 0.74-0.77 after adjustment for confounders, but the risk reduction only remained statistically significant for the first 5 years after vaccination.
The waning seen with the vaccine’s effectiveness “raises the possibility that patients might benefit from a booster vaccine at some point after initial vaccination, although no recommendation currently exists that would support such a practice,” the authors wrote.
Dr. Yun has received research funding from Amgen. Other authors disclosed ties to Amgen, AstraZeneca, Bristol-Myers Squibb, Crescendo Bioscience, Janssen, and Pfizer. One author has received research support and consulting fees from Corrona. The study did not note an external source of funding.
The herpes zoster vaccine reduces the risk of shingles in older adults with autoimmune disease, even if they are taking immunosuppressants for their condition, but the protection begins to wane after about 5 years, a recent retrospective study found.
“There has been some concern that patients with autoimmune conditions might have a lower immunogenic response to herpes zoster vaccination, especially when treated with immunosuppressive medications such as glucocorticoids,” wrote Huifeng Yun, PhD, of the University of Alabama at Birmingham, and her colleagues.
The researchers used 2006-2013 Medicare data to calculate the risk of shingles among Medicare recipients who had an autoimmune disease and either did or did not receive the herpes zoster vaccine. All the patients had been enrolled in Medicare for at least 12 continuous months and had a diagnosis of ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis.
The researchers matched 59,627 patients who received the herpes zoster vaccine with 119,254 unvaccinated patients, based on age, sex, race, calendar year, autoimmune disease type, and use of autoimmune drugs (biologics, disease-modifying antirheumatic drugs, and glucocorticoids). During a follow-up of up to 7 years, the researchers additionally accounted for comorbid medical conditions and concurrent medications each year.
The cohort, with an average age of 73.5 years in both groups, included 53.1% of adults with rheumatoid arthritis, 31.6% with psoriasis, 20.9% with inflammatory bowel disease, 4.7% with psoriatic arthritis, and 1.4% with ankylosing spondylitis.
Those who received the vaccine had a rate of 0.75 herpes zoster cases per 100 people during the first year, which rose to 1.25 cases per 100 people per year at the seventh year after vaccination. The rate among unvaccinated individuals stayed steady at approximately 1.3-1.7 cases per 100 people per year throughout the study period. These rates, as expected, were approximately 50% higher than in the general population over age 70 without autoimmune disease.
Compared with unvaccinated individuals, vaccinated individuals had a reduced relative risk for shingles of 0.74-0.77 after adjustment for confounders, but the risk reduction only remained statistically significant for the first 5 years after vaccination.
The waning seen with the vaccine’s effectiveness “raises the possibility that patients might benefit from a booster vaccine at some point after initial vaccination, although no recommendation currently exists that would support such a practice,” the authors wrote.
Dr. Yun has received research funding from Amgen. Other authors disclosed ties to Amgen, AstraZeneca, Bristol-Myers Squibb, Crescendo Bioscience, Janssen, and Pfizer. One author has received research support and consulting fees from Corrona. The study did not note an external source of funding.
Key clinical point:
Major finding: Medicare patients with autoimmune disease had a 23%-26% reduced risk of shingles for 5 years after receiving the herpes zoster vaccine.
Data source: The findings are based on analysis of 2006-2013 Medicare data on 59,627 patients who received the herpes zoster vaccine and 119,254 patients who didn’t.
Disclosures: Dr. Yun has received research funding from Amgen. Other authors disclosed ties to Amgen, AstraZeneca, Bristol-Myers Squibb, Crescendo Bioscience, Janssen, and Pfizer. One author has received research support and consulting fees from Corrona. The study did not note an external source of funding.
U.S. chikungunya epidemic would likely put rheumatologists on front line
SNOWMASS, COLO. – The continental United States is vulnerable to an epidemic of chikungunya virus disease, an event which would have profound consequences for rheumatologists, Robert T. Schoen, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“We certainly have all the factors in place where we could have a major epidemic of chikungunya, particularly in Florida, Texas, and neighboring states. As rheumatologists, I think we can own this infection if we want to because it causes an arthritis that is a true arthritis in a significant percentage of patients,” said Dr. Schoen, a rheumatologist at Yale University in New Haven, Conn.
“That’s a major impact when you think that these epidemics affect tens of thousands of individuals. If 25% of them develop long-term arthritis disability, that’s a whole new world for us,” the rheumatologist observed.
Chikungunya is a single-stranded RNA virus in the togaviridae family, which also contains rubella. The infection is transmitted by Aedes aegypti and A. albopictus, also known as the yellow fever and Asian tiger mosquitoes, respectively. Both mosquitoes are established inhabitants of much of the United States.
“This infection is a one-bite deal. These are very aggressive mosquitoes,” Dr. Schoen observed. “If you haven’t seen a case of chikungunya yet, you will soon,” he added.
In 2014, at the height of a massive Caribbean epidemic which included half a million cases in Puerto Rico, roughly 2,800 cases of chikungunya were imported into 46 U.S. states. In 2015, however, as the Caribbean epidemic waned and herd immunity developed, that figure fell to 653 imported cases. But the epidemic in India, which began in 2008, remains ongoing with no end in sight. Several of Dr. Schoen’s patients with chikungunya had recently returned from India when they first became ill.
“India provides an unlimited reservoir of immunologically naive patients to perpetuate the infection,” he observed.
Course of illness
The rate of asymptomatic infection has been variously estimated at 3%-25%. Symptomatic chikungunya is a biphasic illness. After a 2- to 6-day incubation period, patients develop rapid-onset fever with severe joint pain and muscle aches. Indeed, chikungunya means “bent over” in Makonde, an African Bantu language.
Roughly 60% of patients develop a rash during the acute febrile phase of the illness, which lasts about a week. The dermatitis is most often a maculopapular rash on the trunk which is “absolutely indistinguishable” from the rash caused by Zika virus infection, transmitted by the same vectors, Dr. Schoen noted.
During this acute phase, almost all patients develop a severe polyarthritis which can last for weeks or, less commonly, for months or years. This polyarthritis mimics seronegative rheumatoid arthritis. It is usually symmetric and often affects the hands, wrists, and feet.
The acute febrile phase is characterized by high levels of viremia, with up to 1 billion viral particles per milliliter of blood. During this period, definitive diagnosis of chikungunya can be made through reverse transcriptase–polymerase chain reaction testing or viral culture.
Typically, though, patients make their way to a rheumatologist only well after the viremic period is over. In that situation, the diagnostic mainstay is serologic testing. Antichikungunya virus Immunoglobulin M is detectable starting on about day 5 after symptom onset, and it persists for the next 1-3 months. Immunoglobulin G (IgG) antibodies become detectable in the same time frame and remain elevated for years.
Dr. Schoen highlighted a small but intriguing study from Singapore that suggests that the early appearance of chikungunya-specific neutralizing IgG3–antibodies may constitute a marker for favorable long-term prognosis. The observational study involved 30 patients hospitalized for severe acute chikungunya infection. The investigators classified them into two groups: 14 patients had low levels of acute viremia, a less severe acute illness, and late development of IgG3 antibodies; the other 16 had a high initial viral load, a more severe acute phase, and a rapid IgG3 and interleukin-6 response to infection.
The patients with a robust early IgG3 response cleared the virus faster and none of them developed chronic arthritis. In contrast, those without early, chikungunya-specific IgG3 had a high rate of persistent arthralgia (J Infect Dis. 2012 Apr 1;205[7]:1147-54).
Treatment
No evidence-based treatment for chikungunya exists. Treatment in the acute phase is primarily supportive care. Rheumatologists on the Caribbean island of Martinique have reported a favorable experience using methotrexate at doses up to 25 mg/week or with standard doses of anti–tumor necrosis factor agents in chikungunya patients with severe bilateral symmetric chronic inflammatory joint disease arising during the 2013-2015 epidemic. The treatment response and tolerability were comparable with the results typically obtained in rheumatoid arthritis patients, according to the rheumatologists (Arthritis Rheumatol. 2016 Nov;68[11]:2817-24).
Dr. Schoen found the report from Martinique to be reassuring. He too has resorted to familiar rheumatologic medications in his severely affected patients. Anecdotally, his greatest success involved a patient with a 5-year history of disabling chikungunya polyarthritis who showed marked improvement after several months on hydroxychloroquine.
The Martinique investigators also reported on 22 patients who developed chikungunya while on biologic agents for rheumatoid arthritis, Crohn’s disease, psoriatic arthritis, or systemic lupus erythematosus. The biologics weren’t protective against the viral disease in these patients. All 22 of them developed severe acute chikungunya polyarthritis with a mean swollen joint count of 9.6 (Joint Bone Spine. 2016 Mar;83[2]:245-6).
Infection during pregnancy
There is a legitimate concern about chikungunya infection occurring during pregnancy, but, based on the experiences documented on Reunion Island, the risk to the baby is confined to intrapartum exposure in a viremic mother.
Reunion Island is a French region in the Indian Ocean off the eastern coast of Africa. It has first-world medical care. Much of the best documented early work on chikungunya outbreaks grew out of the 2005-2006 epidemic there, which affected more than one-third of the island’s 800,000-plus residents.
In a prospective study of 7,504 deliveries on the island during a 22-month period, mother-to-child transmission of chikungunya occurred only in the context of intrapartum viremia, with 19 cases of vertical transmission occurring among 39 affected mothers who delivered at a median 38 weeks of gestation. Cesarean section had no protective effect. All 19 infected neonates were asymptomatic at birth, with median onset of pain, fever, and thrombocytopenia on day 4. Nine of the 19 infected neonates developed encephalopathy with pathologic MRI findings, including cerebral hemorrhage in two cases (PLoS Med. 2008 Mar 18;5[3]:e60).
The case fatality rate for chikungunya is low at 0.1%. Most deaths occur in young children or elderly individuals with major comorbid conditions. Despite this low mortality, interest in chikungunya vaccine development is being driven by the infection’s impact on tourism, its spread to temperate climates, the economic impact of the considerable time lost from work, and military need. A promising attenuated, virus-like, particle-based vaccine is currently in phase II testing in the Caribbean.
Dr. Schoen reported having no financial conflicts.
SNOWMASS, COLO. – The continental United States is vulnerable to an epidemic of chikungunya virus disease, an event which would have profound consequences for rheumatologists, Robert T. Schoen, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“We certainly have all the factors in place where we could have a major epidemic of chikungunya, particularly in Florida, Texas, and neighboring states. As rheumatologists, I think we can own this infection if we want to because it causes an arthritis that is a true arthritis in a significant percentage of patients,” said Dr. Schoen, a rheumatologist at Yale University in New Haven, Conn.
“That’s a major impact when you think that these epidemics affect tens of thousands of individuals. If 25% of them develop long-term arthritis disability, that’s a whole new world for us,” the rheumatologist observed.
Chikungunya is a single-stranded RNA virus in the togaviridae family, which also contains rubella. The infection is transmitted by Aedes aegypti and A. albopictus, also known as the yellow fever and Asian tiger mosquitoes, respectively. Both mosquitoes are established inhabitants of much of the United States.
“This infection is a one-bite deal. These are very aggressive mosquitoes,” Dr. Schoen observed. “If you haven’t seen a case of chikungunya yet, you will soon,” he added.
In 2014, at the height of a massive Caribbean epidemic which included half a million cases in Puerto Rico, roughly 2,800 cases of chikungunya were imported into 46 U.S. states. In 2015, however, as the Caribbean epidemic waned and herd immunity developed, that figure fell to 653 imported cases. But the epidemic in India, which began in 2008, remains ongoing with no end in sight. Several of Dr. Schoen’s patients with chikungunya had recently returned from India when they first became ill.
“India provides an unlimited reservoir of immunologically naive patients to perpetuate the infection,” he observed.
Course of illness
The rate of asymptomatic infection has been variously estimated at 3%-25%. Symptomatic chikungunya is a biphasic illness. After a 2- to 6-day incubation period, patients develop rapid-onset fever with severe joint pain and muscle aches. Indeed, chikungunya means “bent over” in Makonde, an African Bantu language.
Roughly 60% of patients develop a rash during the acute febrile phase of the illness, which lasts about a week. The dermatitis is most often a maculopapular rash on the trunk which is “absolutely indistinguishable” from the rash caused by Zika virus infection, transmitted by the same vectors, Dr. Schoen noted.
During this acute phase, almost all patients develop a severe polyarthritis which can last for weeks or, less commonly, for months or years. This polyarthritis mimics seronegative rheumatoid arthritis. It is usually symmetric and often affects the hands, wrists, and feet.
The acute febrile phase is characterized by high levels of viremia, with up to 1 billion viral particles per milliliter of blood. During this period, definitive diagnosis of chikungunya can be made through reverse transcriptase–polymerase chain reaction testing or viral culture.
Typically, though, patients make their way to a rheumatologist only well after the viremic period is over. In that situation, the diagnostic mainstay is serologic testing. Antichikungunya virus Immunoglobulin M is detectable starting on about day 5 after symptom onset, and it persists for the next 1-3 months. Immunoglobulin G (IgG) antibodies become detectable in the same time frame and remain elevated for years.
Dr. Schoen highlighted a small but intriguing study from Singapore that suggests that the early appearance of chikungunya-specific neutralizing IgG3–antibodies may constitute a marker for favorable long-term prognosis. The observational study involved 30 patients hospitalized for severe acute chikungunya infection. The investigators classified them into two groups: 14 patients had low levels of acute viremia, a less severe acute illness, and late development of IgG3 antibodies; the other 16 had a high initial viral load, a more severe acute phase, and a rapid IgG3 and interleukin-6 response to infection.
The patients with a robust early IgG3 response cleared the virus faster and none of them developed chronic arthritis. In contrast, those without early, chikungunya-specific IgG3 had a high rate of persistent arthralgia (J Infect Dis. 2012 Apr 1;205[7]:1147-54).
Treatment
No evidence-based treatment for chikungunya exists. Treatment in the acute phase is primarily supportive care. Rheumatologists on the Caribbean island of Martinique have reported a favorable experience using methotrexate at doses up to 25 mg/week or with standard doses of anti–tumor necrosis factor agents in chikungunya patients with severe bilateral symmetric chronic inflammatory joint disease arising during the 2013-2015 epidemic. The treatment response and tolerability were comparable with the results typically obtained in rheumatoid arthritis patients, according to the rheumatologists (Arthritis Rheumatol. 2016 Nov;68[11]:2817-24).
Dr. Schoen found the report from Martinique to be reassuring. He too has resorted to familiar rheumatologic medications in his severely affected patients. Anecdotally, his greatest success involved a patient with a 5-year history of disabling chikungunya polyarthritis who showed marked improvement after several months on hydroxychloroquine.
The Martinique investigators also reported on 22 patients who developed chikungunya while on biologic agents for rheumatoid arthritis, Crohn’s disease, psoriatic arthritis, or systemic lupus erythematosus. The biologics weren’t protective against the viral disease in these patients. All 22 of them developed severe acute chikungunya polyarthritis with a mean swollen joint count of 9.6 (Joint Bone Spine. 2016 Mar;83[2]:245-6).
Infection during pregnancy
There is a legitimate concern about chikungunya infection occurring during pregnancy, but, based on the experiences documented on Reunion Island, the risk to the baby is confined to intrapartum exposure in a viremic mother.
Reunion Island is a French region in the Indian Ocean off the eastern coast of Africa. It has first-world medical care. Much of the best documented early work on chikungunya outbreaks grew out of the 2005-2006 epidemic there, which affected more than one-third of the island’s 800,000-plus residents.
In a prospective study of 7,504 deliveries on the island during a 22-month period, mother-to-child transmission of chikungunya occurred only in the context of intrapartum viremia, with 19 cases of vertical transmission occurring among 39 affected mothers who delivered at a median 38 weeks of gestation. Cesarean section had no protective effect. All 19 infected neonates were asymptomatic at birth, with median onset of pain, fever, and thrombocytopenia on day 4. Nine of the 19 infected neonates developed encephalopathy with pathologic MRI findings, including cerebral hemorrhage in two cases (PLoS Med. 2008 Mar 18;5[3]:e60).
The case fatality rate for chikungunya is low at 0.1%. Most deaths occur in young children or elderly individuals with major comorbid conditions. Despite this low mortality, interest in chikungunya vaccine development is being driven by the infection’s impact on tourism, its spread to temperate climates, the economic impact of the considerable time lost from work, and military need. A promising attenuated, virus-like, particle-based vaccine is currently in phase II testing in the Caribbean.
Dr. Schoen reported having no financial conflicts.
SNOWMASS, COLO. – The continental United States is vulnerable to an epidemic of chikungunya virus disease, an event which would have profound consequences for rheumatologists, Robert T. Schoen, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
“We certainly have all the factors in place where we could have a major epidemic of chikungunya, particularly in Florida, Texas, and neighboring states. As rheumatologists, I think we can own this infection if we want to because it causes an arthritis that is a true arthritis in a significant percentage of patients,” said Dr. Schoen, a rheumatologist at Yale University in New Haven, Conn.
“That’s a major impact when you think that these epidemics affect tens of thousands of individuals. If 25% of them develop long-term arthritis disability, that’s a whole new world for us,” the rheumatologist observed.
Chikungunya is a single-stranded RNA virus in the togaviridae family, which also contains rubella. The infection is transmitted by Aedes aegypti and A. albopictus, also known as the yellow fever and Asian tiger mosquitoes, respectively. Both mosquitoes are established inhabitants of much of the United States.
“This infection is a one-bite deal. These are very aggressive mosquitoes,” Dr. Schoen observed. “If you haven’t seen a case of chikungunya yet, you will soon,” he added.
In 2014, at the height of a massive Caribbean epidemic which included half a million cases in Puerto Rico, roughly 2,800 cases of chikungunya were imported into 46 U.S. states. In 2015, however, as the Caribbean epidemic waned and herd immunity developed, that figure fell to 653 imported cases. But the epidemic in India, which began in 2008, remains ongoing with no end in sight. Several of Dr. Schoen’s patients with chikungunya had recently returned from India when they first became ill.
“India provides an unlimited reservoir of immunologically naive patients to perpetuate the infection,” he observed.
Course of illness
The rate of asymptomatic infection has been variously estimated at 3%-25%. Symptomatic chikungunya is a biphasic illness. After a 2- to 6-day incubation period, patients develop rapid-onset fever with severe joint pain and muscle aches. Indeed, chikungunya means “bent over” in Makonde, an African Bantu language.
Roughly 60% of patients develop a rash during the acute febrile phase of the illness, which lasts about a week. The dermatitis is most often a maculopapular rash on the trunk which is “absolutely indistinguishable” from the rash caused by Zika virus infection, transmitted by the same vectors, Dr. Schoen noted.
During this acute phase, almost all patients develop a severe polyarthritis which can last for weeks or, less commonly, for months or years. This polyarthritis mimics seronegative rheumatoid arthritis. It is usually symmetric and often affects the hands, wrists, and feet.
The acute febrile phase is characterized by high levels of viremia, with up to 1 billion viral particles per milliliter of blood. During this period, definitive diagnosis of chikungunya can be made through reverse transcriptase–polymerase chain reaction testing or viral culture.
Typically, though, patients make their way to a rheumatologist only well after the viremic period is over. In that situation, the diagnostic mainstay is serologic testing. Antichikungunya virus Immunoglobulin M is detectable starting on about day 5 after symptom onset, and it persists for the next 1-3 months. Immunoglobulin G (IgG) antibodies become detectable in the same time frame and remain elevated for years.
Dr. Schoen highlighted a small but intriguing study from Singapore that suggests that the early appearance of chikungunya-specific neutralizing IgG3–antibodies may constitute a marker for favorable long-term prognosis. The observational study involved 30 patients hospitalized for severe acute chikungunya infection. The investigators classified them into two groups: 14 patients had low levels of acute viremia, a less severe acute illness, and late development of IgG3 antibodies; the other 16 had a high initial viral load, a more severe acute phase, and a rapid IgG3 and interleukin-6 response to infection.
The patients with a robust early IgG3 response cleared the virus faster and none of them developed chronic arthritis. In contrast, those without early, chikungunya-specific IgG3 had a high rate of persistent arthralgia (J Infect Dis. 2012 Apr 1;205[7]:1147-54).
Treatment
No evidence-based treatment for chikungunya exists. Treatment in the acute phase is primarily supportive care. Rheumatologists on the Caribbean island of Martinique have reported a favorable experience using methotrexate at doses up to 25 mg/week or with standard doses of anti–tumor necrosis factor agents in chikungunya patients with severe bilateral symmetric chronic inflammatory joint disease arising during the 2013-2015 epidemic. The treatment response and tolerability were comparable with the results typically obtained in rheumatoid arthritis patients, according to the rheumatologists (Arthritis Rheumatol. 2016 Nov;68[11]:2817-24).
Dr. Schoen found the report from Martinique to be reassuring. He too has resorted to familiar rheumatologic medications in his severely affected patients. Anecdotally, his greatest success involved a patient with a 5-year history of disabling chikungunya polyarthritis who showed marked improvement after several months on hydroxychloroquine.
The Martinique investigators also reported on 22 patients who developed chikungunya while on biologic agents for rheumatoid arthritis, Crohn’s disease, psoriatic arthritis, or systemic lupus erythematosus. The biologics weren’t protective against the viral disease in these patients. All 22 of them developed severe acute chikungunya polyarthritis with a mean swollen joint count of 9.6 (Joint Bone Spine. 2016 Mar;83[2]:245-6).
Infection during pregnancy
There is a legitimate concern about chikungunya infection occurring during pregnancy, but, based on the experiences documented on Reunion Island, the risk to the baby is confined to intrapartum exposure in a viremic mother.
Reunion Island is a French region in the Indian Ocean off the eastern coast of Africa. It has first-world medical care. Much of the best documented early work on chikungunya outbreaks grew out of the 2005-2006 epidemic there, which affected more than one-third of the island’s 800,000-plus residents.
In a prospective study of 7,504 deliveries on the island during a 22-month period, mother-to-child transmission of chikungunya occurred only in the context of intrapartum viremia, with 19 cases of vertical transmission occurring among 39 affected mothers who delivered at a median 38 weeks of gestation. Cesarean section had no protective effect. All 19 infected neonates were asymptomatic at birth, with median onset of pain, fever, and thrombocytopenia on day 4. Nine of the 19 infected neonates developed encephalopathy with pathologic MRI findings, including cerebral hemorrhage in two cases (PLoS Med. 2008 Mar 18;5[3]:e60).
The case fatality rate for chikungunya is low at 0.1%. Most deaths occur in young children or elderly individuals with major comorbid conditions. Despite this low mortality, interest in chikungunya vaccine development is being driven by the infection’s impact on tourism, its spread to temperate climates, the economic impact of the considerable time lost from work, and military need. A promising attenuated, virus-like, particle-based vaccine is currently in phase II testing in the Caribbean.
Dr. Schoen reported having no financial conflicts.
No rise in CV events seen with tocilizumab
Patients with refractory rheumatoid arthritis who switched to tocilizumab showed no increased cardiovascular risk when compared with those who switched to a tumor necrosis factor inhibitor in a large cohort study.
Rheumatoid arthritis is known to approximately double the risk of cardiovascular morbidity and mortality partly because of its associated chronic systemic inflammation. Several small trials and observational studies have reported that tocilizumab, an interleukin-6 receptor antagonist typically used as a second-line treatment for RA, elevates serum lipids, including LDL cholesterol. These serum lipid elevations caused by tocilizumab have brought concerns that the drug may further heighten CV risk in people with RA, said Seoyoung C. Kim, MD, ScD, of the division of pharmacoepidemiology and pharmacoeconomics and the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston, and her associates.
To minimize the effect of confounding by the severity of RA and the baseline CV risk, the researchers adjusted the data to account for more than 90 variables related to CV events and to RA severity.
The primary outcome — a composite of MI and stroke — occurred in 125 patients, 36 taking tocilizumab and 89 taking TNF inhibitors. The rate of this composite outcome was 0.52 per 100 person-years with tocilizumab and 0.59 per 100 person-years for TNF inhibitors, a nonsignificant difference, Dr. Kim and her associates reported (Arthritis Rheumatol. 2017 Feb 28. doi: 10.1002/art.40084).
There also were no significant differences between the two study groups in secondary endpoints, including rates of coronary revascularization, acute coronary syndrome, heart failure, and all-cause mortality. In addition, all subgroup analyses confirmed that tocilizumab did not raise CV risk, regardless of patient age (younger than or older than 60 years), the presence of cardiovascular disease at baseline, the presence of diabetes, the use of methotrexate, the use of oral steroids, or the use of statins.
These “reassuring” findings show that even though tocilizumab appears to raise LDL levels, “such increases do not appear to be associated with an increased risk of clinical CV events,” the investigators said.
The results confirm those reported at the 2016 American College of Rheumatology annual meeting for the 5-year, randomized, postmarketing ENTRACTE trial in which the lipid changes induced by tocilizumab did not translate into an increased risk of heart attack or stroke in RA patients.
This cohort study was sponsored by Genentech, which markets tocilizumab (Actemra). Dr. Kim reported ties to Genentech, Lilly, Pfizer, Bristol-Myers Squibb, and AstraZeneca. Her associates reported ties to Genentech, Lilly, Pfizer, AstraZeneca, Amgen, Corrona, Whiscon, Aetion, and Boehringer Ingelheim. Three of the seven authors were employees of Genentech.
Patients with refractory rheumatoid arthritis who switched to tocilizumab showed no increased cardiovascular risk when compared with those who switched to a tumor necrosis factor inhibitor in a large cohort study.
Rheumatoid arthritis is known to approximately double the risk of cardiovascular morbidity and mortality partly because of its associated chronic systemic inflammation. Several small trials and observational studies have reported that tocilizumab, an interleukin-6 receptor antagonist typically used as a second-line treatment for RA, elevates serum lipids, including LDL cholesterol. These serum lipid elevations caused by tocilizumab have brought concerns that the drug may further heighten CV risk in people with RA, said Seoyoung C. Kim, MD, ScD, of the division of pharmacoepidemiology and pharmacoeconomics and the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston, and her associates.
To minimize the effect of confounding by the severity of RA and the baseline CV risk, the researchers adjusted the data to account for more than 90 variables related to CV events and to RA severity.
The primary outcome — a composite of MI and stroke — occurred in 125 patients, 36 taking tocilizumab and 89 taking TNF inhibitors. The rate of this composite outcome was 0.52 per 100 person-years with tocilizumab and 0.59 per 100 person-years for TNF inhibitors, a nonsignificant difference, Dr. Kim and her associates reported (Arthritis Rheumatol. 2017 Feb 28. doi: 10.1002/art.40084).
There also were no significant differences between the two study groups in secondary endpoints, including rates of coronary revascularization, acute coronary syndrome, heart failure, and all-cause mortality. In addition, all subgroup analyses confirmed that tocilizumab did not raise CV risk, regardless of patient age (younger than or older than 60 years), the presence of cardiovascular disease at baseline, the presence of diabetes, the use of methotrexate, the use of oral steroids, or the use of statins.
These “reassuring” findings show that even though tocilizumab appears to raise LDL levels, “such increases do not appear to be associated with an increased risk of clinical CV events,” the investigators said.
The results confirm those reported at the 2016 American College of Rheumatology annual meeting for the 5-year, randomized, postmarketing ENTRACTE trial in which the lipid changes induced by tocilizumab did not translate into an increased risk of heart attack or stroke in RA patients.
This cohort study was sponsored by Genentech, which markets tocilizumab (Actemra). Dr. Kim reported ties to Genentech, Lilly, Pfizer, Bristol-Myers Squibb, and AstraZeneca. Her associates reported ties to Genentech, Lilly, Pfizer, AstraZeneca, Amgen, Corrona, Whiscon, Aetion, and Boehringer Ingelheim. Three of the seven authors were employees of Genentech.
Patients with refractory rheumatoid arthritis who switched to tocilizumab showed no increased cardiovascular risk when compared with those who switched to a tumor necrosis factor inhibitor in a large cohort study.
Rheumatoid arthritis is known to approximately double the risk of cardiovascular morbidity and mortality partly because of its associated chronic systemic inflammation. Several small trials and observational studies have reported that tocilizumab, an interleukin-6 receptor antagonist typically used as a second-line treatment for RA, elevates serum lipids, including LDL cholesterol. These serum lipid elevations caused by tocilizumab have brought concerns that the drug may further heighten CV risk in people with RA, said Seoyoung C. Kim, MD, ScD, of the division of pharmacoepidemiology and pharmacoeconomics and the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston, and her associates.
To minimize the effect of confounding by the severity of RA and the baseline CV risk, the researchers adjusted the data to account for more than 90 variables related to CV events and to RA severity.
The primary outcome — a composite of MI and stroke — occurred in 125 patients, 36 taking tocilizumab and 89 taking TNF inhibitors. The rate of this composite outcome was 0.52 per 100 person-years with tocilizumab and 0.59 per 100 person-years for TNF inhibitors, a nonsignificant difference, Dr. Kim and her associates reported (Arthritis Rheumatol. 2017 Feb 28. doi: 10.1002/art.40084).
There also were no significant differences between the two study groups in secondary endpoints, including rates of coronary revascularization, acute coronary syndrome, heart failure, and all-cause mortality. In addition, all subgroup analyses confirmed that tocilizumab did not raise CV risk, regardless of patient age (younger than or older than 60 years), the presence of cardiovascular disease at baseline, the presence of diabetes, the use of methotrexate, the use of oral steroids, or the use of statins.
These “reassuring” findings show that even though tocilizumab appears to raise LDL levels, “such increases do not appear to be associated with an increased risk of clinical CV events,” the investigators said.
The results confirm those reported at the 2016 American College of Rheumatology annual meeting for the 5-year, randomized, postmarketing ENTRACTE trial in which the lipid changes induced by tocilizumab did not translate into an increased risk of heart attack or stroke in RA patients.
This cohort study was sponsored by Genentech, which markets tocilizumab (Actemra). Dr. Kim reported ties to Genentech, Lilly, Pfizer, Bristol-Myers Squibb, and AstraZeneca. Her associates reported ties to Genentech, Lilly, Pfizer, AstraZeneca, Amgen, Corrona, Whiscon, Aetion, and Boehringer Ingelheim. Three of the seven authors were employees of Genentech.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Patients with refractory RA who switch to tocilizumab show no increased cardiovascular risk, compared with those who switch to a TNF inhibitor.
Major finding: The primary outcome – the rate of a composite of MI and stroke – was 0.52 per 100 person-years with tocilizumab and 0.59 per 100 person-years for TNF inhibitors, a nonsignificant difference.
Data source: A cohort study involving 28,028 adults with RA enrolled in three large health care claims databases from all 50 states who were followed for a median of 1 year.
Disclosures: This study was sponsored by Genentech, which markets tocilizumab (Actemra). Dr. Kim reported ties to Genentech, Lilly, Pfizer, Bristol-Myers Squibb, and AstraZeneca. Her associates reported ties to Genentech, Lilly, Pfizer, AstraZeneca, Amgen, Corrona, Whiscon, Aetion, and Boehringer Ingelheim. Three of the seven authors were employees of Genentech.
Biomarker score not predictive of successful TNF inhibitor tapering in RA
A multi-biomarker disease activity score in patients with longstanding rheumatoid arthritis and low disease activity prior to tapering adalimumab or etanercept did not predict flare-related outcomes in an 18-month, open-label, randomized clinical trial.
These findings seemed “robust and valid, at least in this specific context,” said investigators led by Chantal A. M. Bouman, MD, of Sint Maartenskliniek Nijmegen (the Netherlands), because multiple other outcomes measured in the trial, including discontinuation of biologic and radiographic progression, were not predicted by the multi-biomarker disease activity (MBDA) score, which is derived from an algorithm using a biomarker panel of 12 serum proteins and is marketed under the name Vectra DA.
The researchers sought to determine if measurement of disease activity using biomarkers with the MBDA score has a potential for smaller measurement error, compared with clinical decision making, in predicting the effects of dose tapering of biologic disease-modifying antirheumatic drugs by examining blood samples taken from 171 people with longstanding RA with low disease activity who were participating in the Dutch Dose Reduction Strategies of Subcutaneous TNF inhibitors (DRESS) trial (Rheumatology [Oxford]. 2017 Feb 22. doi: 10.1093/rheumatology/kex003).
The use of biomarkers could potentially overcome some of the drawbacks of the most widely used and extensively validated measure of RA disease activity, the Disease Activity Score in 28 joints (DAS28), which relies on “clinical assessments [that] are subject to interobserver variability, resulting in measurement error and suboptimal precision,” the investigators wrote. “Also, DAS28 can be influenced by factors other than RA disease activity (e.g., OA, [fibromyalgia], or other causes of inflammation, such an infection), resulting in clinical misclassification of disease activity state.”
The randomized DRESS trial investigated the noninferiority of a dose reduction strategy of adalimumab or etanercept (n = 115), compared with usual care (n = 56), on the rate of flare, defined as a DAS28 (using C-reactive protein) increase of more than 1.2 or 0.6 if the current DAS was more than or equal to 3.2. A major flare was one lasting more than 3 months despite treatment intervention.
The baseline MBDA score did not predict successful tapering based on an area under the receiver operating characteristic (AUROC) of 0.53 (95% confidence interval, 0.41-0.66), nor did it predict the discontinuation of either biologic (AUROC = 0.51; 95% CI, 0.36-0.66) or the occurrence of flare (AUROC = 0.50; 95% CI, 0.41-0.59 for both groups combined) or major flare (AUROC = 0.46; 95% CI, 0.32-0.65 for both groups combined).
Although the authors found a borderline positive predictive value of baseline MBDA score for major flare in the usual care group (AUROC = 0.72; 95% CI, 0.56-0.88), they said the finding should be “interpreted cautiously” as multiple testing may have resulted in false-positive findings.
The researchers also discovered that, in contrast to findings from five studies in four cohorts of patients with established or early RA, the MBDA did not predict radiographic progression (AUROC = 0.53 for predicting radiographic progression of more than 0.5 Sharp–van der Heijde points; 95% CI, 0.43-0.63 for both groups combined).
The inability of the MBDA score to predict radiographic outcomes “might be attributable to the low frequency and severity of radiographic progression in our study, with only a small difference in favor of the [usual care] group. It might reflect the strict tight control that was applied to patients who were already in low disease activity or remission,” the investigators suggested.
They also said that, in spite of the findings, the MBDA score may have predictive value in other groups of patients, such as those with early rheumatoid arthritis, higher disease activity, or suboptimal disease control, and suggested that the study’s findings be validated in further studies.
The study received no specific funding. However, one author is an employee of Crescendo Bioscience and reported receiving stock grants from its parent company, Myriad Genetics. Several authors reported relationships with industry.
A multi-biomarker disease activity score in patients with longstanding rheumatoid arthritis and low disease activity prior to tapering adalimumab or etanercept did not predict flare-related outcomes in an 18-month, open-label, randomized clinical trial.
These findings seemed “robust and valid, at least in this specific context,” said investigators led by Chantal A. M. Bouman, MD, of Sint Maartenskliniek Nijmegen (the Netherlands), because multiple other outcomes measured in the trial, including discontinuation of biologic and radiographic progression, were not predicted by the multi-biomarker disease activity (MBDA) score, which is derived from an algorithm using a biomarker panel of 12 serum proteins and is marketed under the name Vectra DA.
The researchers sought to determine if measurement of disease activity using biomarkers with the MBDA score has a potential for smaller measurement error, compared with clinical decision making, in predicting the effects of dose tapering of biologic disease-modifying antirheumatic drugs by examining blood samples taken from 171 people with longstanding RA with low disease activity who were participating in the Dutch Dose Reduction Strategies of Subcutaneous TNF inhibitors (DRESS) trial (Rheumatology [Oxford]. 2017 Feb 22. doi: 10.1093/rheumatology/kex003).
The use of biomarkers could potentially overcome some of the drawbacks of the most widely used and extensively validated measure of RA disease activity, the Disease Activity Score in 28 joints (DAS28), which relies on “clinical assessments [that] are subject to interobserver variability, resulting in measurement error and suboptimal precision,” the investigators wrote. “Also, DAS28 can be influenced by factors other than RA disease activity (e.g., OA, [fibromyalgia], or other causes of inflammation, such an infection), resulting in clinical misclassification of disease activity state.”
The randomized DRESS trial investigated the noninferiority of a dose reduction strategy of adalimumab or etanercept (n = 115), compared with usual care (n = 56), on the rate of flare, defined as a DAS28 (using C-reactive protein) increase of more than 1.2 or 0.6 if the current DAS was more than or equal to 3.2. A major flare was one lasting more than 3 months despite treatment intervention.
The baseline MBDA score did not predict successful tapering based on an area under the receiver operating characteristic (AUROC) of 0.53 (95% confidence interval, 0.41-0.66), nor did it predict the discontinuation of either biologic (AUROC = 0.51; 95% CI, 0.36-0.66) or the occurrence of flare (AUROC = 0.50; 95% CI, 0.41-0.59 for both groups combined) or major flare (AUROC = 0.46; 95% CI, 0.32-0.65 for both groups combined).
Although the authors found a borderline positive predictive value of baseline MBDA score for major flare in the usual care group (AUROC = 0.72; 95% CI, 0.56-0.88), they said the finding should be “interpreted cautiously” as multiple testing may have resulted in false-positive findings.
The researchers also discovered that, in contrast to findings from five studies in four cohorts of patients with established or early RA, the MBDA did not predict radiographic progression (AUROC = 0.53 for predicting radiographic progression of more than 0.5 Sharp–van der Heijde points; 95% CI, 0.43-0.63 for both groups combined).
The inability of the MBDA score to predict radiographic outcomes “might be attributable to the low frequency and severity of radiographic progression in our study, with only a small difference in favor of the [usual care] group. It might reflect the strict tight control that was applied to patients who were already in low disease activity or remission,” the investigators suggested.
They also said that, in spite of the findings, the MBDA score may have predictive value in other groups of patients, such as those with early rheumatoid arthritis, higher disease activity, or suboptimal disease control, and suggested that the study’s findings be validated in further studies.
The study received no specific funding. However, one author is an employee of Crescendo Bioscience and reported receiving stock grants from its parent company, Myriad Genetics. Several authors reported relationships with industry.
A multi-biomarker disease activity score in patients with longstanding rheumatoid arthritis and low disease activity prior to tapering adalimumab or etanercept did not predict flare-related outcomes in an 18-month, open-label, randomized clinical trial.
These findings seemed “robust and valid, at least in this specific context,” said investigators led by Chantal A. M. Bouman, MD, of Sint Maartenskliniek Nijmegen (the Netherlands), because multiple other outcomes measured in the trial, including discontinuation of biologic and radiographic progression, were not predicted by the multi-biomarker disease activity (MBDA) score, which is derived from an algorithm using a biomarker panel of 12 serum proteins and is marketed under the name Vectra DA.
The researchers sought to determine if measurement of disease activity using biomarkers with the MBDA score has a potential for smaller measurement error, compared with clinical decision making, in predicting the effects of dose tapering of biologic disease-modifying antirheumatic drugs by examining blood samples taken from 171 people with longstanding RA with low disease activity who were participating in the Dutch Dose Reduction Strategies of Subcutaneous TNF inhibitors (DRESS) trial (Rheumatology [Oxford]. 2017 Feb 22. doi: 10.1093/rheumatology/kex003).
The use of biomarkers could potentially overcome some of the drawbacks of the most widely used and extensively validated measure of RA disease activity, the Disease Activity Score in 28 joints (DAS28), which relies on “clinical assessments [that] are subject to interobserver variability, resulting in measurement error and suboptimal precision,” the investigators wrote. “Also, DAS28 can be influenced by factors other than RA disease activity (e.g., OA, [fibromyalgia], or other causes of inflammation, such an infection), resulting in clinical misclassification of disease activity state.”
The randomized DRESS trial investigated the noninferiority of a dose reduction strategy of adalimumab or etanercept (n = 115), compared with usual care (n = 56), on the rate of flare, defined as a DAS28 (using C-reactive protein) increase of more than 1.2 or 0.6 if the current DAS was more than or equal to 3.2. A major flare was one lasting more than 3 months despite treatment intervention.
The baseline MBDA score did not predict successful tapering based on an area under the receiver operating characteristic (AUROC) of 0.53 (95% confidence interval, 0.41-0.66), nor did it predict the discontinuation of either biologic (AUROC = 0.51; 95% CI, 0.36-0.66) or the occurrence of flare (AUROC = 0.50; 95% CI, 0.41-0.59 for both groups combined) or major flare (AUROC = 0.46; 95% CI, 0.32-0.65 for both groups combined).
Although the authors found a borderline positive predictive value of baseline MBDA score for major flare in the usual care group (AUROC = 0.72; 95% CI, 0.56-0.88), they said the finding should be “interpreted cautiously” as multiple testing may have resulted in false-positive findings.
The researchers also discovered that, in contrast to findings from five studies in four cohorts of patients with established or early RA, the MBDA did not predict radiographic progression (AUROC = 0.53 for predicting radiographic progression of more than 0.5 Sharp–van der Heijde points; 95% CI, 0.43-0.63 for both groups combined).
The inability of the MBDA score to predict radiographic outcomes “might be attributable to the low frequency and severity of radiographic progression in our study, with only a small difference in favor of the [usual care] group. It might reflect the strict tight control that was applied to patients who were already in low disease activity or remission,” the investigators suggested.
They also said that, in spite of the findings, the MBDA score may have predictive value in other groups of patients, such as those with early rheumatoid arthritis, higher disease activity, or suboptimal disease control, and suggested that the study’s findings be validated in further studies.
The study received no specific funding. However, one author is an employee of Crescendo Bioscience and reported receiving stock grants from its parent company, Myriad Genetics. Several authors reported relationships with industry.
FROM RHEUMATOLOGY
Key clinical point:
Main finding: The baseline MBDA score did not predict successful tapering based on an area under the receiver operating characteristic of 0.53 (95% CI, 0.41-0.66).
Data source: Analysis of serum samples from 171 RA patients taking part in the noninferiority, randomized, open-label, controlled Dose Reduction Strategies of Subcutaneous TNF inhibitors (DRESS) trial.
Disclosures: The study received no specific funding. However, one author is an employee of Crescendo Bioscience and reported receiving stock grants from its parent company, Myriad Genetics. Several authors reported relationships with industry.
Methotrexate use justified for early RA, disease prevention
Efforts to delay or prevent rheumatoid arthritis (RA) in individuals at high risk for the disease received a boost from the findings of a subgroup analysis of an older Dutch prevention trial involving methotrexate that were published recently in Arthritis & Rheumatology.
However, while the 22-patient subgroup analysis of the 2007 trial, called PROMPT (Probable RA: Methotrexate Versus Placebo Treatment), showed that 1 year of methotrexate prevented the development of rheumatoid arthritis in significantly more individuals at high risk for the disease than did placebo, hindsight has shown that most of these high-risk individuals actually had RA at baseline as defined by the 2010 American College or Rheumatology/European League Against Rheumatism classification criteria.
Even though the “landscape of RA has changed since this study was conceived, enrolled, and performed,” Dr. Sparks noted that the study provides evidence that today’s “standard of care really works” by making use of data that are unobtainable today.
“Nowadays, we wouldn’t be able to do a placebo-controlled trial in early RA because those patients need to be treated, but at the time, it was perfectly fine, based on the treatment landscape, to put these patients in a [placebo-controlled] trial,” he said in an interview.
“It also demonstrates that you really need to find high-risk individuals, because, if you recruit people who are never going to develop RA, you’re just diluting your effect, and you’re not going to find the difference in the groups based on that diluted effect when there could be a true effect in that special subgroup,” he added.
Senior study author Annette van der Helm-van Mil, MD, PhD, and her colleagues at Leiden (the Netherlands) University Medical Center performed the reanalysis after the initial finding that methotrexate had no preventative effect may have been falsely negative because the study included patients with a low risk of progressing to RA (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40062).
In the reanalysis, the investigators defined 22 out of the trial’s 110 participants as high risk based on their score of 8 or higher on the Leiden prediction rule at baseline. Of those 22, 18 also fulfilled the 2010 classification criteria for RA.
This “definition of high risk [in the trial] very much coincided with the new classification criteria and reinforces the standard of care that patients with new diagnosed RA should be treated with DMARDs [disease-modifying antirheumatic drugs] front line,” Dr. Sparks said.
The Leiden prediction rule score is based on ACPA statuses, as well as age, sex, distribution of involved joints, number of swollen and tender joints, severity of morning stiffness, C-reactive protein level, and rheumatoid factor. In previous studies of patients with undifferentiated arthritis, a score of 8 or higher with the prediction rule had a positive predictive value of 84% in patients with undifferentiated arthritis, according to the investigators.
Based on a primary outcome of fulfilling the 1987 classification criteria for RA after 5 years of follow-up, the investigators found that 6 of 11 (55%) in the methotrexate arm developed RA, compared with all 11 patients in the placebo arm (P = .011). “A 1-year course of methotrexate was associated with an absolute risk reduction of 45% on RA development, resulting in a number needed to treat of 2.2 (95% [confidence interval], 1.3-6.2),” the authors wrote.
Furthermore, the time to the development of RA based on the 1987 criteria was longer with methotrexate than with placebo (median of 22.5 months vs. 3 months; P less than .001).
Drug-free remission was also achieved by 4 of 11 (36%) patients taking methotrexate, compared with none of the patients taking placebo (P = .027).
The beneficial effects of methotrexate were seen in ACPA-positive and ACPA-negative high-risk patients but not in patients without a high risk of RA.
For the patients who did not fulfill the 2010 RA criteria at baseline, only one of four of those in the placebo arm did go on to develop RA based on 1987 criteria, whereas only one of three who received methotrexate did.
Overall, the reanalysis of PROMPT trial data illustrates how “noninformative inclusions can blur highly relevant study outcomes, such as prevention of RA,” the study authors concluded.
“As trials are now being conducted to evaluate the efficacy of treatment initiated in the phase of arthralgia, the development of adequate risk prediction models are of importance in order to prevent false-negative study results in the future,” they added.
The PROMPT trial was supported by the Dutch Arthritis Foundation and the Netherlands Organisation for Scientific Research.
Small numbers not withstanding, the findings of this reanalysis of the PROMPT trial are exciting, and they address the importance of identifying the right individuals to include in any type of prevention study in RA.
Given the temporal limits of clinical trials, inclusion criteria will need to incorporate both the likelihood of an important outcome and the timing of that outcome – right individuals, right time, right drug/intervention.
It is hoped that investigators will be able to demonstrate conclusively that preventive interventions work for rheumatic disease once these issues are addressed.
Kevin D. Deane, MD, PhD, and his coauthors are with the division of rheumatology at the University of Colorado, Denver, and made these remarks in an editorial accompanying the PROMPT trial reanalysis (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40061).
Small numbers not withstanding, the findings of this reanalysis of the PROMPT trial are exciting, and they address the importance of identifying the right individuals to include in any type of prevention study in RA.
Given the temporal limits of clinical trials, inclusion criteria will need to incorporate both the likelihood of an important outcome and the timing of that outcome – right individuals, right time, right drug/intervention.
It is hoped that investigators will be able to demonstrate conclusively that preventive interventions work for rheumatic disease once these issues are addressed.
Kevin D. Deane, MD, PhD, and his coauthors are with the division of rheumatology at the University of Colorado, Denver, and made these remarks in an editorial accompanying the PROMPT trial reanalysis (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40061).
Small numbers not withstanding, the findings of this reanalysis of the PROMPT trial are exciting, and they address the importance of identifying the right individuals to include in any type of prevention study in RA.
Given the temporal limits of clinical trials, inclusion criteria will need to incorporate both the likelihood of an important outcome and the timing of that outcome – right individuals, right time, right drug/intervention.
It is hoped that investigators will be able to demonstrate conclusively that preventive interventions work for rheumatic disease once these issues are addressed.
Kevin D. Deane, MD, PhD, and his coauthors are with the division of rheumatology at the University of Colorado, Denver, and made these remarks in an editorial accompanying the PROMPT trial reanalysis (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40061).
Efforts to delay or prevent rheumatoid arthritis (RA) in individuals at high risk for the disease received a boost from the findings of a subgroup analysis of an older Dutch prevention trial involving methotrexate that were published recently in Arthritis & Rheumatology.
However, while the 22-patient subgroup analysis of the 2007 trial, called PROMPT (Probable RA: Methotrexate Versus Placebo Treatment), showed that 1 year of methotrexate prevented the development of rheumatoid arthritis in significantly more individuals at high risk for the disease than did placebo, hindsight has shown that most of these high-risk individuals actually had RA at baseline as defined by the 2010 American College or Rheumatology/European League Against Rheumatism classification criteria.
Even though the “landscape of RA has changed since this study was conceived, enrolled, and performed,” Dr. Sparks noted that the study provides evidence that today’s “standard of care really works” by making use of data that are unobtainable today.
“Nowadays, we wouldn’t be able to do a placebo-controlled trial in early RA because those patients need to be treated, but at the time, it was perfectly fine, based on the treatment landscape, to put these patients in a [placebo-controlled] trial,” he said in an interview.
“It also demonstrates that you really need to find high-risk individuals, because, if you recruit people who are never going to develop RA, you’re just diluting your effect, and you’re not going to find the difference in the groups based on that diluted effect when there could be a true effect in that special subgroup,” he added.
Senior study author Annette van der Helm-van Mil, MD, PhD, and her colleagues at Leiden (the Netherlands) University Medical Center performed the reanalysis after the initial finding that methotrexate had no preventative effect may have been falsely negative because the study included patients with a low risk of progressing to RA (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40062).
In the reanalysis, the investigators defined 22 out of the trial’s 110 participants as high risk based on their score of 8 or higher on the Leiden prediction rule at baseline. Of those 22, 18 also fulfilled the 2010 classification criteria for RA.
This “definition of high risk [in the trial] very much coincided with the new classification criteria and reinforces the standard of care that patients with new diagnosed RA should be treated with DMARDs [disease-modifying antirheumatic drugs] front line,” Dr. Sparks said.
The Leiden prediction rule score is based on ACPA statuses, as well as age, sex, distribution of involved joints, number of swollen and tender joints, severity of morning stiffness, C-reactive protein level, and rheumatoid factor. In previous studies of patients with undifferentiated arthritis, a score of 8 or higher with the prediction rule had a positive predictive value of 84% in patients with undifferentiated arthritis, according to the investigators.
Based on a primary outcome of fulfilling the 1987 classification criteria for RA after 5 years of follow-up, the investigators found that 6 of 11 (55%) in the methotrexate arm developed RA, compared with all 11 patients in the placebo arm (P = .011). “A 1-year course of methotrexate was associated with an absolute risk reduction of 45% on RA development, resulting in a number needed to treat of 2.2 (95% [confidence interval], 1.3-6.2),” the authors wrote.
Furthermore, the time to the development of RA based on the 1987 criteria was longer with methotrexate than with placebo (median of 22.5 months vs. 3 months; P less than .001).
Drug-free remission was also achieved by 4 of 11 (36%) patients taking methotrexate, compared with none of the patients taking placebo (P = .027).
The beneficial effects of methotrexate were seen in ACPA-positive and ACPA-negative high-risk patients but not in patients without a high risk of RA.
For the patients who did not fulfill the 2010 RA criteria at baseline, only one of four of those in the placebo arm did go on to develop RA based on 1987 criteria, whereas only one of three who received methotrexate did.
Overall, the reanalysis of PROMPT trial data illustrates how “noninformative inclusions can blur highly relevant study outcomes, such as prevention of RA,” the study authors concluded.
“As trials are now being conducted to evaluate the efficacy of treatment initiated in the phase of arthralgia, the development of adequate risk prediction models are of importance in order to prevent false-negative study results in the future,” they added.
The PROMPT trial was supported by the Dutch Arthritis Foundation and the Netherlands Organisation for Scientific Research.
Efforts to delay or prevent rheumatoid arthritis (RA) in individuals at high risk for the disease received a boost from the findings of a subgroup analysis of an older Dutch prevention trial involving methotrexate that were published recently in Arthritis & Rheumatology.
However, while the 22-patient subgroup analysis of the 2007 trial, called PROMPT (Probable RA: Methotrexate Versus Placebo Treatment), showed that 1 year of methotrexate prevented the development of rheumatoid arthritis in significantly more individuals at high risk for the disease than did placebo, hindsight has shown that most of these high-risk individuals actually had RA at baseline as defined by the 2010 American College or Rheumatology/European League Against Rheumatism classification criteria.
Even though the “landscape of RA has changed since this study was conceived, enrolled, and performed,” Dr. Sparks noted that the study provides evidence that today’s “standard of care really works” by making use of data that are unobtainable today.
“Nowadays, we wouldn’t be able to do a placebo-controlled trial in early RA because those patients need to be treated, but at the time, it was perfectly fine, based on the treatment landscape, to put these patients in a [placebo-controlled] trial,” he said in an interview.
“It also demonstrates that you really need to find high-risk individuals, because, if you recruit people who are never going to develop RA, you’re just diluting your effect, and you’re not going to find the difference in the groups based on that diluted effect when there could be a true effect in that special subgroup,” he added.
Senior study author Annette van der Helm-van Mil, MD, PhD, and her colleagues at Leiden (the Netherlands) University Medical Center performed the reanalysis after the initial finding that methotrexate had no preventative effect may have been falsely negative because the study included patients with a low risk of progressing to RA (Arthritis Rheumatol. 2017 Feb 19. doi: 10.1002/art.40062).
In the reanalysis, the investigators defined 22 out of the trial’s 110 participants as high risk based on their score of 8 or higher on the Leiden prediction rule at baseline. Of those 22, 18 also fulfilled the 2010 classification criteria for RA.
This “definition of high risk [in the trial] very much coincided with the new classification criteria and reinforces the standard of care that patients with new diagnosed RA should be treated with DMARDs [disease-modifying antirheumatic drugs] front line,” Dr. Sparks said.
The Leiden prediction rule score is based on ACPA statuses, as well as age, sex, distribution of involved joints, number of swollen and tender joints, severity of morning stiffness, C-reactive protein level, and rheumatoid factor. In previous studies of patients with undifferentiated arthritis, a score of 8 or higher with the prediction rule had a positive predictive value of 84% in patients with undifferentiated arthritis, according to the investigators.
Based on a primary outcome of fulfilling the 1987 classification criteria for RA after 5 years of follow-up, the investigators found that 6 of 11 (55%) in the methotrexate arm developed RA, compared with all 11 patients in the placebo arm (P = .011). “A 1-year course of methotrexate was associated with an absolute risk reduction of 45% on RA development, resulting in a number needed to treat of 2.2 (95% [confidence interval], 1.3-6.2),” the authors wrote.
Furthermore, the time to the development of RA based on the 1987 criteria was longer with methotrexate than with placebo (median of 22.5 months vs. 3 months; P less than .001).
Drug-free remission was also achieved by 4 of 11 (36%) patients taking methotrexate, compared with none of the patients taking placebo (P = .027).
The beneficial effects of methotrexate were seen in ACPA-positive and ACPA-negative high-risk patients but not in patients without a high risk of RA.
For the patients who did not fulfill the 2010 RA criteria at baseline, only one of four of those in the placebo arm did go on to develop RA based on 1987 criteria, whereas only one of three who received methotrexate did.
Overall, the reanalysis of PROMPT trial data illustrates how “noninformative inclusions can blur highly relevant study outcomes, such as prevention of RA,” the study authors concluded.
“As trials are now being conducted to evaluate the efficacy of treatment initiated in the phase of arthralgia, the development of adequate risk prediction models are of importance in order to prevent false-negative study results in the future,” they added.
The PROMPT trial was supported by the Dutch Arthritis Foundation and the Netherlands Organisation for Scientific Research.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:
Main finding: In people deemed to be at high risk for RA, a 1-year course of methotrexate was associated with an absolute risk reduction of 45% of RA development, resulting in a number needed to treat of 2.2 (95% CI, 1.3-6.2).
Data source: A reanalysis of the PROMPT trial involving 110 patients with undifferentiated arthritis randomized to a 1-year course of methotrexate or placebo.
Disclosures: The PROMPT trial was supported by the Dutch Arthritis Foundation and the Netherlands Organisation for Scientific Research.