User login
Cannabis shows inconsistent benefits for pain, PTSD
Chronic pain and posttraumatic stress disorder are among the top reasons given by patients seeking medical marijuana in states where it is legal, but there is little scientific evidence to support its value for treating either condition, based on the results of a pair of systemic evidence reviews conducted by the U.S. Department of Veterans Affairs.
The findings were published online Aug. 14 in the Annals of Internal Medicine.
In the PTSD study, Maya E. O’Neil, PhD, of the VA Portland (Ore.) Health Care System, and colleagues found no significant evidence to support the effectiveness of cannabis for relieving symptoms (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0477). The researchers reviewed data from two systematic reviews and three primary studies.
One of the larger studies (included in one of the systematic reviews) included 47,000 veterans in VA intensive PTSD programs during 1992-2011. In fact, after controlling for demographic factors and other confounding variables, individuals who continued to use cannabis or started using cannabis showed worse PTSD symptoms than did nonusers after 4 months.
“Findings from [randomized, controlled trials] are needed to help determine whether and to what extent cannabis may improve PTSD symptoms, and further studies also are needed to clarify harms in patients with PTSD,” the researchers noted.
In the review of chronic pain literature, Shannon M. Nugent, PhD, also of the VA Portland (Ore.) Health Care System, and her colleagues examined 27 trials, 11 reviews, and 32 primary studies (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0155).
“Across nine studies, intervention patients were more likely to report at least 30% improvement in pain,” the investigators said. But this finding was specific to tetrahydrocannabinol (THC), the researchers said, and evidence of the ability of cannabis to relieve other types of pain, such as cancer pain and multiple sclerosis pain, was insufficient.
In addition, the researchers found a low strength of evidence association between cannabis use and the development of psychotic symptoms, and a moderate strength of evidence association between cannabis use and impaired cognitive function in the general population. “However, our confidence in the findings is limited by inconsistent findings among included studies, inadequate assessment of exposure, and inadequate adjustment for confounding among the studies” they said.
Although no significant differences were noted in rates of adverse events between cannabis users and nonusers, some data suggested users had an increased risk for short-term adverse events that ranged from dizziness to paranoia and suicide attempts.
Other potential harms associated with cannabis use included decreased lung function, increased risk of complications from infectious diseases, cannabis hyperemesis syndrome, and increased risk of violent behavior.
“Even though we did not find strong, consistent evidence of benefit, clinicians will still need to engage in evidence-based discussions with patients managing chronic pain who are using or requesting to use cannabis,” the researchers wrote.
The researchers had no financial conflicts to disclose. The study was commissioned by the Veterans Health Administration.
“The systematic reviews highlight an alarming lack of high-quality data from which to draw firm conclusions about the efficacy of cannabis for these conditions, for which cannabis is both sanctioned and commonly used,” wrote Sachin Patel, MD, PhD, in an accompanying editorial (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-1713).
“Even if future studies reveal a clear lack of substantial benefit of cannabis for pain or PTSD, legislation is unlikely to remove these conditions from the lists of indications for medical cannabis,” he cautioned. “It will be up to front-line practicing physicians to learn about the harms and benefits of cannabis, educate their patients on these topics, and make evidence-based recommendations about using cannabis and related products for various health conditions.
“In parallel, the research community must pursue high-quality studies and disseminate the results to clinicians and the public. In this context, these reviews are must-reads for all physicians, especially those practicing in states where medical cannabis is legal,” Dr. Patel added.
Dr. Patel is affiliated with Vanderbilt Psychiatric Hospital in Nashville, Tenn. He had no relevant financial conflicts to disclose, but reported receiving grants from Lundbeck.
“The systematic reviews highlight an alarming lack of high-quality data from which to draw firm conclusions about the efficacy of cannabis for these conditions, for which cannabis is both sanctioned and commonly used,” wrote Sachin Patel, MD, PhD, in an accompanying editorial (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-1713).
“Even if future studies reveal a clear lack of substantial benefit of cannabis for pain or PTSD, legislation is unlikely to remove these conditions from the lists of indications for medical cannabis,” he cautioned. “It will be up to front-line practicing physicians to learn about the harms and benefits of cannabis, educate their patients on these topics, and make evidence-based recommendations about using cannabis and related products for various health conditions.
“In parallel, the research community must pursue high-quality studies and disseminate the results to clinicians and the public. In this context, these reviews are must-reads for all physicians, especially those practicing in states where medical cannabis is legal,” Dr. Patel added.
Dr. Patel is affiliated with Vanderbilt Psychiatric Hospital in Nashville, Tenn. He had no relevant financial conflicts to disclose, but reported receiving grants from Lundbeck.
“The systematic reviews highlight an alarming lack of high-quality data from which to draw firm conclusions about the efficacy of cannabis for these conditions, for which cannabis is both sanctioned and commonly used,” wrote Sachin Patel, MD, PhD, in an accompanying editorial (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-1713).
“Even if future studies reveal a clear lack of substantial benefit of cannabis for pain or PTSD, legislation is unlikely to remove these conditions from the lists of indications for medical cannabis,” he cautioned. “It will be up to front-line practicing physicians to learn about the harms and benefits of cannabis, educate their patients on these topics, and make evidence-based recommendations about using cannabis and related products for various health conditions.
“In parallel, the research community must pursue high-quality studies and disseminate the results to clinicians and the public. In this context, these reviews are must-reads for all physicians, especially those practicing in states where medical cannabis is legal,” Dr. Patel added.
Dr. Patel is affiliated with Vanderbilt Psychiatric Hospital in Nashville, Tenn. He had no relevant financial conflicts to disclose, but reported receiving grants from Lundbeck.
Chronic pain and posttraumatic stress disorder are among the top reasons given by patients seeking medical marijuana in states where it is legal, but there is little scientific evidence to support its value for treating either condition, based on the results of a pair of systemic evidence reviews conducted by the U.S. Department of Veterans Affairs.
The findings were published online Aug. 14 in the Annals of Internal Medicine.
In the PTSD study, Maya E. O’Neil, PhD, of the VA Portland (Ore.) Health Care System, and colleagues found no significant evidence to support the effectiveness of cannabis for relieving symptoms (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0477). The researchers reviewed data from two systematic reviews and three primary studies.
One of the larger studies (included in one of the systematic reviews) included 47,000 veterans in VA intensive PTSD programs during 1992-2011. In fact, after controlling for demographic factors and other confounding variables, individuals who continued to use cannabis or started using cannabis showed worse PTSD symptoms than did nonusers after 4 months.
“Findings from [randomized, controlled trials] are needed to help determine whether and to what extent cannabis may improve PTSD symptoms, and further studies also are needed to clarify harms in patients with PTSD,” the researchers noted.
In the review of chronic pain literature, Shannon M. Nugent, PhD, also of the VA Portland (Ore.) Health Care System, and her colleagues examined 27 trials, 11 reviews, and 32 primary studies (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0155).
“Across nine studies, intervention patients were more likely to report at least 30% improvement in pain,” the investigators said. But this finding was specific to tetrahydrocannabinol (THC), the researchers said, and evidence of the ability of cannabis to relieve other types of pain, such as cancer pain and multiple sclerosis pain, was insufficient.
In addition, the researchers found a low strength of evidence association between cannabis use and the development of psychotic symptoms, and a moderate strength of evidence association between cannabis use and impaired cognitive function in the general population. “However, our confidence in the findings is limited by inconsistent findings among included studies, inadequate assessment of exposure, and inadequate adjustment for confounding among the studies” they said.
Although no significant differences were noted in rates of adverse events between cannabis users and nonusers, some data suggested users had an increased risk for short-term adverse events that ranged from dizziness to paranoia and suicide attempts.
Other potential harms associated with cannabis use included decreased lung function, increased risk of complications from infectious diseases, cannabis hyperemesis syndrome, and increased risk of violent behavior.
“Even though we did not find strong, consistent evidence of benefit, clinicians will still need to engage in evidence-based discussions with patients managing chronic pain who are using or requesting to use cannabis,” the researchers wrote.
The researchers had no financial conflicts to disclose. The study was commissioned by the Veterans Health Administration.
Chronic pain and posttraumatic stress disorder are among the top reasons given by patients seeking medical marijuana in states where it is legal, but there is little scientific evidence to support its value for treating either condition, based on the results of a pair of systemic evidence reviews conducted by the U.S. Department of Veterans Affairs.
The findings were published online Aug. 14 in the Annals of Internal Medicine.
In the PTSD study, Maya E. O’Neil, PhD, of the VA Portland (Ore.) Health Care System, and colleagues found no significant evidence to support the effectiveness of cannabis for relieving symptoms (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0477). The researchers reviewed data from two systematic reviews and three primary studies.
One of the larger studies (included in one of the systematic reviews) included 47,000 veterans in VA intensive PTSD programs during 1992-2011. In fact, after controlling for demographic factors and other confounding variables, individuals who continued to use cannabis or started using cannabis showed worse PTSD symptoms than did nonusers after 4 months.
“Findings from [randomized, controlled trials] are needed to help determine whether and to what extent cannabis may improve PTSD symptoms, and further studies also are needed to clarify harms in patients with PTSD,” the researchers noted.
In the review of chronic pain literature, Shannon M. Nugent, PhD, also of the VA Portland (Ore.) Health Care System, and her colleagues examined 27 trials, 11 reviews, and 32 primary studies (Ann Intern Med. 2017 Aug 14. doi: 10.7326/M17-0155).
“Across nine studies, intervention patients were more likely to report at least 30% improvement in pain,” the investigators said. But this finding was specific to tetrahydrocannabinol (THC), the researchers said, and evidence of the ability of cannabis to relieve other types of pain, such as cancer pain and multiple sclerosis pain, was insufficient.
In addition, the researchers found a low strength of evidence association between cannabis use and the development of psychotic symptoms, and a moderate strength of evidence association between cannabis use and impaired cognitive function in the general population. “However, our confidence in the findings is limited by inconsistent findings among included studies, inadequate assessment of exposure, and inadequate adjustment for confounding among the studies” they said.
Although no significant differences were noted in rates of adverse events between cannabis users and nonusers, some data suggested users had an increased risk for short-term adverse events that ranged from dizziness to paranoia and suicide attempts.
Other potential harms associated with cannabis use included decreased lung function, increased risk of complications from infectious diseases, cannabis hyperemesis syndrome, and increased risk of violent behavior.
“Even though we did not find strong, consistent evidence of benefit, clinicians will still need to engage in evidence-based discussions with patients managing chronic pain who are using or requesting to use cannabis,” the researchers wrote.
The researchers had no financial conflicts to disclose. The study was commissioned by the Veterans Health Administration.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point:
Major finding: One study of cannabis use for neuropathic pain showed an approximately 30% improvement in pain, but researchers found no significant impact on relieving other types of chronic pain or PTSD symptoms.
Data source: The data come from a chronic pain review that included 27 trials, 11 systematic reviews, and 32 primary studies; and a PTSD review that included 2 systematic reviews and 3 primary studies.
Disclosures: The researchers had no financial conflicts to disclose. The study was commissioned by the Veterans Health Administration.
RAND Study Provides Report Card for MHS PTSD and Depression Care
Deck: Study finds overall high quality of care but identifies important gaps in assessment and treatment.
Active-duty service members with posttraumatic stress disorder (PTSD) or depression are frequent users of both inpatient and outpatient Military Health System (MHS) care, but there were significant variations in the quality and type of care they received, according to a recent RAND Corporation study. According to the authors, the MHS “performed well in providing initial screening for suicide and substance abuse but needs to improve at providing adequate follow-up to service members with suicide risk.”
The Quality of Care for PTSD and Depression in the Military Health System study is the third in a series of reports on the quality of depression and PTSD care for active-duty service members. The study examined data for 2013-2014 and included 14,654 service members with PTSD and 30,496 with depression; 6,322 service members were in both groups. The goal of the report was to determine whether the service members with PTSD or depression receive evidence-based care and whether there were disparities in care quality by branch of service, geographic region, and other characteristics, such as gender, age, pay grade, race/ethnicity, or deployment history.
Related: New Center of Excellence to Lead Research of “Signature Wounds”
In both the PTSD and depression cohorts, the majority of active-component service members were enlisted soldiers and had 1 or more deployments. A large number of patients were in both the PTSD and depression cohorts. The study noted that more than half of patients in the PTSD cohort had a diagnosis of depression, and more than one-fourth of those in the depression cohort received a PTSD diagnosis during the 12-month observation period.
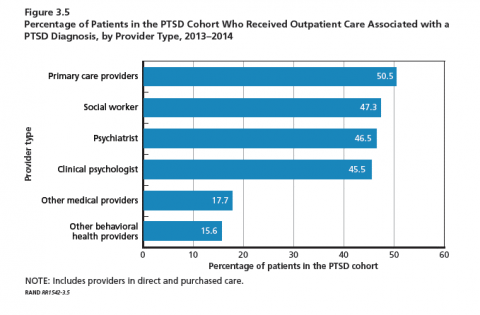
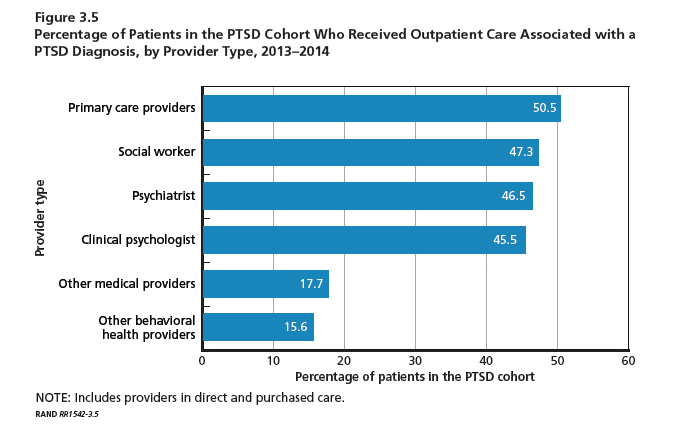
Service members with PTSD or depression were willing to engage the health system with medians of 40 and 31 visits for PTSD and depression, respectively, during the 1-year observation period. Most of these visits were for unrelated conditions. Importantly, more than half of patients received their care from primary care providers. Social workers, psychiatrists, and clinical psychologists provided care for slightly less than half of the PTSD cohort. These mental health provider groups saw between 33% and 40% of the depression cohort. The majority of patients in both the PTSD and depression cohorts received care for their cohort diagnosis solely at military treatment facilities
“The high utilization for both medical and psychological conditions combined with the high number of different providers raise questions about the extent of coordination vs fragmentation of care for all the care these service members received,” the study reported. “The high number of psychotherapy visits received by members of these cohorts suggests that the MHS may be more successful than the civilian sector in engaging patients with PTSD or depression in psychosocial interventions.”
The study also highlighted the following important gaps in treatment, assessment, and follow-up:
- Assessment of baseline symptom severity of PTSD for a new treatment episode with the PCL (PTSD Checklist) was not as frequent, though current efforts are under way within the MHS to increase the regular use of the PCL to monitor PTSD patient symptoms.
- Standardized tools were used in less than half of the assessments for depression, suicide risk, and recent substance use and almost never used for assessment of function.
- Appropriate minimal care for patients with suicidal ideation was less than optimal (54%), primarily due to a lack of documentation regarding addressing access to lethal means. A Safety Plan Worksheet has recently been added to the clinical guideline for assessment and management of suicide risk which may improve this performance in the future.
- Most service members with PTSD or depression received at least some psychotherapy, but the MHS could increase delivery of evidence-based psychotherapy.
- Improvements are still needed in rates of outcome monitoring and performance on outcome measures.
- Multiple “statistically significant and clinically meaningful differences” were found in measure scores by service branch, TRICARE region, and service member.
Deck: Study finds overall high quality of care but identifies important gaps in assessment and treatment.
Active-duty service members with posttraumatic stress disorder (PTSD) or depression are frequent users of both inpatient and outpatient Military Health System (MHS) care, but there were significant variations in the quality and type of care they received, according to a recent RAND Corporation study. According to the authors, the MHS “performed well in providing initial screening for suicide and substance abuse but needs to improve at providing adequate follow-up to service members with suicide risk.”
The Quality of Care for PTSD and Depression in the Military Health System study is the third in a series of reports on the quality of depression and PTSD care for active-duty service members. The study examined data for 2013-2014 and included 14,654 service members with PTSD and 30,496 with depression; 6,322 service members were in both groups. The goal of the report was to determine whether the service members with PTSD or depression receive evidence-based care and whether there were disparities in care quality by branch of service, geographic region, and other characteristics, such as gender, age, pay grade, race/ethnicity, or deployment history.
Related: New Center of Excellence to Lead Research of “Signature Wounds”
In both the PTSD and depression cohorts, the majority of active-component service members were enlisted soldiers and had 1 or more deployments. A large number of patients were in both the PTSD and depression cohorts. The study noted that more than half of patients in the PTSD cohort had a diagnosis of depression, and more than one-fourth of those in the depression cohort received a PTSD diagnosis during the 12-month observation period.
Service members with PTSD or depression were willing to engage the health system with medians of 40 and 31 visits for PTSD and depression, respectively, during the 1-year observation period. Most of these visits were for unrelated conditions. Importantly, more than half of patients received their care from primary care providers. Social workers, psychiatrists, and clinical psychologists provided care for slightly less than half of the PTSD cohort. These mental health provider groups saw between 33% and 40% of the depression cohort. The majority of patients in both the PTSD and depression cohorts received care for their cohort diagnosis solely at military treatment facilities
“The high utilization for both medical and psychological conditions combined with the high number of different providers raise questions about the extent of coordination vs fragmentation of care for all the care these service members received,” the study reported. “The high number of psychotherapy visits received by members of these cohorts suggests that the MHS may be more successful than the civilian sector in engaging patients with PTSD or depression in psychosocial interventions.”
The study also highlighted the following important gaps in treatment, assessment, and follow-up:
- Assessment of baseline symptom severity of PTSD for a new treatment episode with the PCL (PTSD Checklist) was not as frequent, though current efforts are under way within the MHS to increase the regular use of the PCL to monitor PTSD patient symptoms.
- Standardized tools were used in less than half of the assessments for depression, suicide risk, and recent substance use and almost never used for assessment of function.
- Appropriate minimal care for patients with suicidal ideation was less than optimal (54%), primarily due to a lack of documentation regarding addressing access to lethal means. A Safety Plan Worksheet has recently been added to the clinical guideline for assessment and management of suicide risk which may improve this performance in the future.
- Most service members with PTSD or depression received at least some psychotherapy, but the MHS could increase delivery of evidence-based psychotherapy.
- Improvements are still needed in rates of outcome monitoring and performance on outcome measures.
- Multiple “statistically significant and clinically meaningful differences” were found in measure scores by service branch, TRICARE region, and service member.
Deck: Study finds overall high quality of care but identifies important gaps in assessment and treatment.
Active-duty service members with posttraumatic stress disorder (PTSD) or depression are frequent users of both inpatient and outpatient Military Health System (MHS) care, but there were significant variations in the quality and type of care they received, according to a recent RAND Corporation study. According to the authors, the MHS “performed well in providing initial screening for suicide and substance abuse but needs to improve at providing adequate follow-up to service members with suicide risk.”
The Quality of Care for PTSD and Depression in the Military Health System study is the third in a series of reports on the quality of depression and PTSD care for active-duty service members. The study examined data for 2013-2014 and included 14,654 service members with PTSD and 30,496 with depression; 6,322 service members were in both groups. The goal of the report was to determine whether the service members with PTSD or depression receive evidence-based care and whether there were disparities in care quality by branch of service, geographic region, and other characteristics, such as gender, age, pay grade, race/ethnicity, or deployment history.
Related: New Center of Excellence to Lead Research of “Signature Wounds”
In both the PTSD and depression cohorts, the majority of active-component service members were enlisted soldiers and had 1 or more deployments. A large number of patients were in both the PTSD and depression cohorts. The study noted that more than half of patients in the PTSD cohort had a diagnosis of depression, and more than one-fourth of those in the depression cohort received a PTSD diagnosis during the 12-month observation period.
Service members with PTSD or depression were willing to engage the health system with medians of 40 and 31 visits for PTSD and depression, respectively, during the 1-year observation period. Most of these visits were for unrelated conditions. Importantly, more than half of patients received their care from primary care providers. Social workers, psychiatrists, and clinical psychologists provided care for slightly less than half of the PTSD cohort. These mental health provider groups saw between 33% and 40% of the depression cohort. The majority of patients in both the PTSD and depression cohorts received care for their cohort diagnosis solely at military treatment facilities
“The high utilization for both medical and psychological conditions combined with the high number of different providers raise questions about the extent of coordination vs fragmentation of care for all the care these service members received,” the study reported. “The high number of psychotherapy visits received by members of these cohorts suggests that the MHS may be more successful than the civilian sector in engaging patients with PTSD or depression in psychosocial interventions.”
The study also highlighted the following important gaps in treatment, assessment, and follow-up:
- Assessment of baseline symptom severity of PTSD for a new treatment episode with the PCL (PTSD Checklist) was not as frequent, though current efforts are under way within the MHS to increase the regular use of the PCL to monitor PTSD patient symptoms.
- Standardized tools were used in less than half of the assessments for depression, suicide risk, and recent substance use and almost never used for assessment of function.
- Appropriate minimal care for patients with suicidal ideation was less than optimal (54%), primarily due to a lack of documentation regarding addressing access to lethal means. A Safety Plan Worksheet has recently been added to the clinical guideline for assessment and management of suicide risk which may improve this performance in the future.
- Most service members with PTSD or depression received at least some psychotherapy, but the MHS could increase delivery of evidence-based psychotherapy.
- Improvements are still needed in rates of outcome monitoring and performance on outcome measures.
- Multiple “statistically significant and clinically meaningful differences” were found in measure scores by service branch, TRICARE region, and service member.
Depression, PTSD double risk of dementia for older female veterans
LONDON – Women veterans with either depression or post-traumatic stress disorder face a doubling in their risk of dementia – and having both increases the risk even more, Dr. Kristine Yaffe reported at the Alzheimer’s Association International Conference.*
The risk ratios for incident dementia that Dr. Yaffe of the University of California, San Francisco, and her colleagues calculated from their analysis of a cohort of 149,000 older female veterans in the national Veterans Health Administration (VHA) database remained unchanged even when they adjusted for age, education, medical comorbidities, and other confounders.
Not only are older women veterans a growing group; they are frequently diagnosed with mental health disorders. In 2012, 45% of women veteran patients in the VHA had a mental health condition, Dr. Yaffe noted.
“Over 9% of all veterans in the U.S. are women, accounting for more than 2 million women veterans. And 30% of those are more than 55 years old. Additionally, the number of women utilizing the Veterans Healthcare Administration system has nearly doubled in the last decade.”
The study of the impact of depression and PTSD on incident dementia is the first of its kind, Dr. Yaffe noted. The cohort comprised women without dementia who had at least two VHA visits during 2005-2015. They were followed for a mean of 5 years. A diagnosis of depression or PTSD had to occur during a 2-year baseline period. Confounders considered in the analysis were demographics, medical comorbidities, and health habits, including alcohol and tobacco use. The primary outcome was time to incident dementia.
At baseline, the group was a mean of 67 years old. Most subjects (70%) were white. Hypertension was common (46%), as was diabetes (16%). About 6% had cardiovascular disease. Depression was present in 18% and PTSD in 4%.
When parsed by diagnosis, there were some significant between-group differences at baseline. Women with depression or PTSD were younger than those without (65 and 63 vs. 67 years). Women who had both disorders were the youngest group, at 62 years.
Hypertension was least common in women without depression or PTSD (41%), and most common among those with depression (65%). Diabetes was almost more common among women with depression than among those without (24% vs. 14%).
Dr. Yaffe created two regression analyses. Model 1 controlled for age, race, and education. Model 2 controlled for the factors in Model 1, plus diabetes, hypertension, and cardiovascular disease.
By the end of follow-up, 4% of the group had developed dementia. The presence of depression approximately doubled the risk of dementia (hazard ratio, 2.14), compared with women who had neither depression nor PTSD. This risk was virtually unchanged in both Model 1 and Model 2 (HRs, 2.12 and 2.00).
The risk associated with PTSD was quite similar, increasing the risk of dementia twofold (HR, 2.19). Again, this was similar after controlling for the confounders in both Model 1 (HR, 2.20) and Model 2 (HR, 2.16).
Women with both depression and PTSD had almost a tripling of risk for dementia (HR, 2.71). Adjustment for confounders did not significantly alter this risk, either in Model 1 (HR, 2.59) or Model 2 (HR, 2.42).
“A question that often comes up in these types of studies is, ‘Is this a reverse causation?’ ” Dr. Yaffe said. “In other words, are people with dementia somehow getting more depression? We conducted a lag-time analysis that allowed a 2-year lag time for dementia, and also adjusted for the number of clinic visits. The results were almost identical.”
“This consistent doubling of risk is quite high,” Dr. Yaffe said. “In our prior work with male veterans, we didn’t see this robust an association.”
The study was funded by the Department of Defense and the National Institutes of Health. Dr. Yaffe had no financial disclosures.
Correction, 8/7/17: An earlier version of this article misstated Dr. Kristine Yaffe's degree.
msullivan@frontlinemedcom.com
On Twitter @alz_gal
LONDON – Women veterans with either depression or post-traumatic stress disorder face a doubling in their risk of dementia – and having both increases the risk even more, Dr. Kristine Yaffe reported at the Alzheimer’s Association International Conference.*
The risk ratios for incident dementia that Dr. Yaffe of the University of California, San Francisco, and her colleagues calculated from their analysis of a cohort of 149,000 older female veterans in the national Veterans Health Administration (VHA) database remained unchanged even when they adjusted for age, education, medical comorbidities, and other confounders.
Not only are older women veterans a growing group; they are frequently diagnosed with mental health disorders. In 2012, 45% of women veteran patients in the VHA had a mental health condition, Dr. Yaffe noted.
“Over 9% of all veterans in the U.S. are women, accounting for more than 2 million women veterans. And 30% of those are more than 55 years old. Additionally, the number of women utilizing the Veterans Healthcare Administration system has nearly doubled in the last decade.”
The study of the impact of depression and PTSD on incident dementia is the first of its kind, Dr. Yaffe noted. The cohort comprised women without dementia who had at least two VHA visits during 2005-2015. They were followed for a mean of 5 years. A diagnosis of depression or PTSD had to occur during a 2-year baseline period. Confounders considered in the analysis were demographics, medical comorbidities, and health habits, including alcohol and tobacco use. The primary outcome was time to incident dementia.
At baseline, the group was a mean of 67 years old. Most subjects (70%) were white. Hypertension was common (46%), as was diabetes (16%). About 6% had cardiovascular disease. Depression was present in 18% and PTSD in 4%.
When parsed by diagnosis, there were some significant between-group differences at baseline. Women with depression or PTSD were younger than those without (65 and 63 vs. 67 years). Women who had both disorders were the youngest group, at 62 years.
Hypertension was least common in women without depression or PTSD (41%), and most common among those with depression (65%). Diabetes was almost more common among women with depression than among those without (24% vs. 14%).
Dr. Yaffe created two regression analyses. Model 1 controlled for age, race, and education. Model 2 controlled for the factors in Model 1, plus diabetes, hypertension, and cardiovascular disease.
By the end of follow-up, 4% of the group had developed dementia. The presence of depression approximately doubled the risk of dementia (hazard ratio, 2.14), compared with women who had neither depression nor PTSD. This risk was virtually unchanged in both Model 1 and Model 2 (HRs, 2.12 and 2.00).
The risk associated with PTSD was quite similar, increasing the risk of dementia twofold (HR, 2.19). Again, this was similar after controlling for the confounders in both Model 1 (HR, 2.20) and Model 2 (HR, 2.16).
Women with both depression and PTSD had almost a tripling of risk for dementia (HR, 2.71). Adjustment for confounders did not significantly alter this risk, either in Model 1 (HR, 2.59) or Model 2 (HR, 2.42).
“A question that often comes up in these types of studies is, ‘Is this a reverse causation?’ ” Dr. Yaffe said. “In other words, are people with dementia somehow getting more depression? We conducted a lag-time analysis that allowed a 2-year lag time for dementia, and also adjusted for the number of clinic visits. The results were almost identical.”
“This consistent doubling of risk is quite high,” Dr. Yaffe said. “In our prior work with male veterans, we didn’t see this robust an association.”
The study was funded by the Department of Defense and the National Institutes of Health. Dr. Yaffe had no financial disclosures.
Correction, 8/7/17: An earlier version of this article misstated Dr. Kristine Yaffe's degree.
msullivan@frontlinemedcom.com
On Twitter @alz_gal
LONDON – Women veterans with either depression or post-traumatic stress disorder face a doubling in their risk of dementia – and having both increases the risk even more, Dr. Kristine Yaffe reported at the Alzheimer’s Association International Conference.*
The risk ratios for incident dementia that Dr. Yaffe of the University of California, San Francisco, and her colleagues calculated from their analysis of a cohort of 149,000 older female veterans in the national Veterans Health Administration (VHA) database remained unchanged even when they adjusted for age, education, medical comorbidities, and other confounders.
Not only are older women veterans a growing group; they are frequently diagnosed with mental health disorders. In 2012, 45% of women veteran patients in the VHA had a mental health condition, Dr. Yaffe noted.
“Over 9% of all veterans in the U.S. are women, accounting for more than 2 million women veterans. And 30% of those are more than 55 years old. Additionally, the number of women utilizing the Veterans Healthcare Administration system has nearly doubled in the last decade.”
The study of the impact of depression and PTSD on incident dementia is the first of its kind, Dr. Yaffe noted. The cohort comprised women without dementia who had at least two VHA visits during 2005-2015. They were followed for a mean of 5 years. A diagnosis of depression or PTSD had to occur during a 2-year baseline period. Confounders considered in the analysis were demographics, medical comorbidities, and health habits, including alcohol and tobacco use. The primary outcome was time to incident dementia.
At baseline, the group was a mean of 67 years old. Most subjects (70%) were white. Hypertension was common (46%), as was diabetes (16%). About 6% had cardiovascular disease. Depression was present in 18% and PTSD in 4%.
When parsed by diagnosis, there were some significant between-group differences at baseline. Women with depression or PTSD were younger than those without (65 and 63 vs. 67 years). Women who had both disorders were the youngest group, at 62 years.
Hypertension was least common in women without depression or PTSD (41%), and most common among those with depression (65%). Diabetes was almost more common among women with depression than among those without (24% vs. 14%).
Dr. Yaffe created two regression analyses. Model 1 controlled for age, race, and education. Model 2 controlled for the factors in Model 1, plus diabetes, hypertension, and cardiovascular disease.
By the end of follow-up, 4% of the group had developed dementia. The presence of depression approximately doubled the risk of dementia (hazard ratio, 2.14), compared with women who had neither depression nor PTSD. This risk was virtually unchanged in both Model 1 and Model 2 (HRs, 2.12 and 2.00).
The risk associated with PTSD was quite similar, increasing the risk of dementia twofold (HR, 2.19). Again, this was similar after controlling for the confounders in both Model 1 (HR, 2.20) and Model 2 (HR, 2.16).
Women with both depression and PTSD had almost a tripling of risk for dementia (HR, 2.71). Adjustment for confounders did not significantly alter this risk, either in Model 1 (HR, 2.59) or Model 2 (HR, 2.42).
“A question that often comes up in these types of studies is, ‘Is this a reverse causation?’ ” Dr. Yaffe said. “In other words, are people with dementia somehow getting more depression? We conducted a lag-time analysis that allowed a 2-year lag time for dementia, and also adjusted for the number of clinic visits. The results were almost identical.”
“This consistent doubling of risk is quite high,” Dr. Yaffe said. “In our prior work with male veterans, we didn’t see this robust an association.”
The study was funded by the Department of Defense and the National Institutes of Health. Dr. Yaffe had no financial disclosures.
Correction, 8/7/17: An earlier version of this article misstated Dr. Kristine Yaffe's degree.
msullivan@frontlinemedcom.com
On Twitter @alz_gal
AT AAIC 2017
Key clinical point:
Major finding: Depression or PTSD both doubled the risk of dementia; both conditions together increased the risk by almost 2.5 times.
Data source: The retrospective cohort study comprised 149,000 women in the national Veterans Health Administration database.
Disclosures: The Department of Defense and National Institutes of Health Funded the study. The presenter had no financial disclosures.
Treating comorbid posttraumatic stress disorder and cardiovascular disease
Mr. S, 64, has a history of posttraumatic stress disorder (PTSD), which has been well controlled for the past 15 years with cognitive-processing therapy and fluoxetine, 40 mg/d. However, over the past 6 weeks, Mr. S has experienced increased hypervigilance, nightmares, and flashbacks. He states that his primary care provider recommended an adjustment in pharmacotherapy to address this exacerbation of symptoms. Previous medication trials include sertraline, 200 mg/d, discontinued due to lack of perceived efficacy, and venlafaxine, 150 mg/d, discontinued due to increased blood pressure.
Mr. S’s medical history includes hypertension, dyslipidemia, and myocardial infarction (MI) 5 years ago. His family history includes sudden cardiac death (mother and father) and major depressive disorder (sister). His blood pressure is currently uncontrolled on lisinopril, 5 mg/d, and metoprolol succinate, 50 mg/d. Today, serial blood pressure readings measured approximately 180/90 mm Hg, with a pulse 50-60 beats per minute.
What is the next step in treating Mr. S’s hypertension and PTSD symptoms? Is there any evidence to support concomitant therapy?
PTSD is characterized by emotional and behavioral symptoms following exposure to a traumatic event. Its 12-month prevalence in the United States is estimated at 3.5%. Diagnostic criteria necessitate the presence of intrusive symptoms, persistent effortful avoidance of distressing trauma-related stimuli, negative cognitions or mood, and alterations in arousal and reactivity. PTSD negatively impacts social and occupational functioning.1
Studies have revealed a correlation between the presence of psychosocial factors, such as depression and anxiety, and the occurrence of cardiovascular events. The mechanism appears to consist of a behavioral component (eg, poor diet, tobacco use) and a direct pathophysiologic component (eg, excessive sympathetic nervous system activation) (Table 13).4 Management of concomitant PTSD and CVD presents a challenge to clinicians.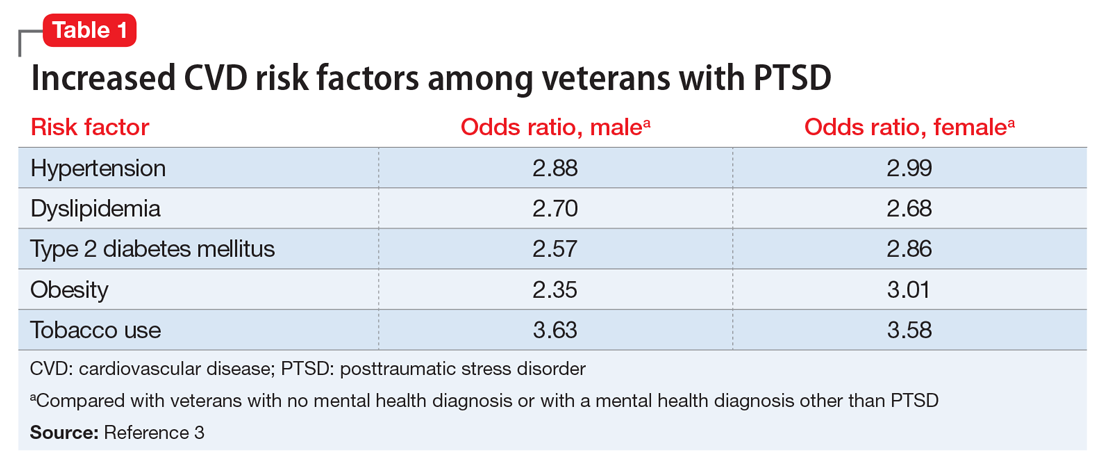
This article summarizes the evidence for the use of CVD medications in treating PTSD (Table 2) and how to apply these principles in patient care (Table 35-14).
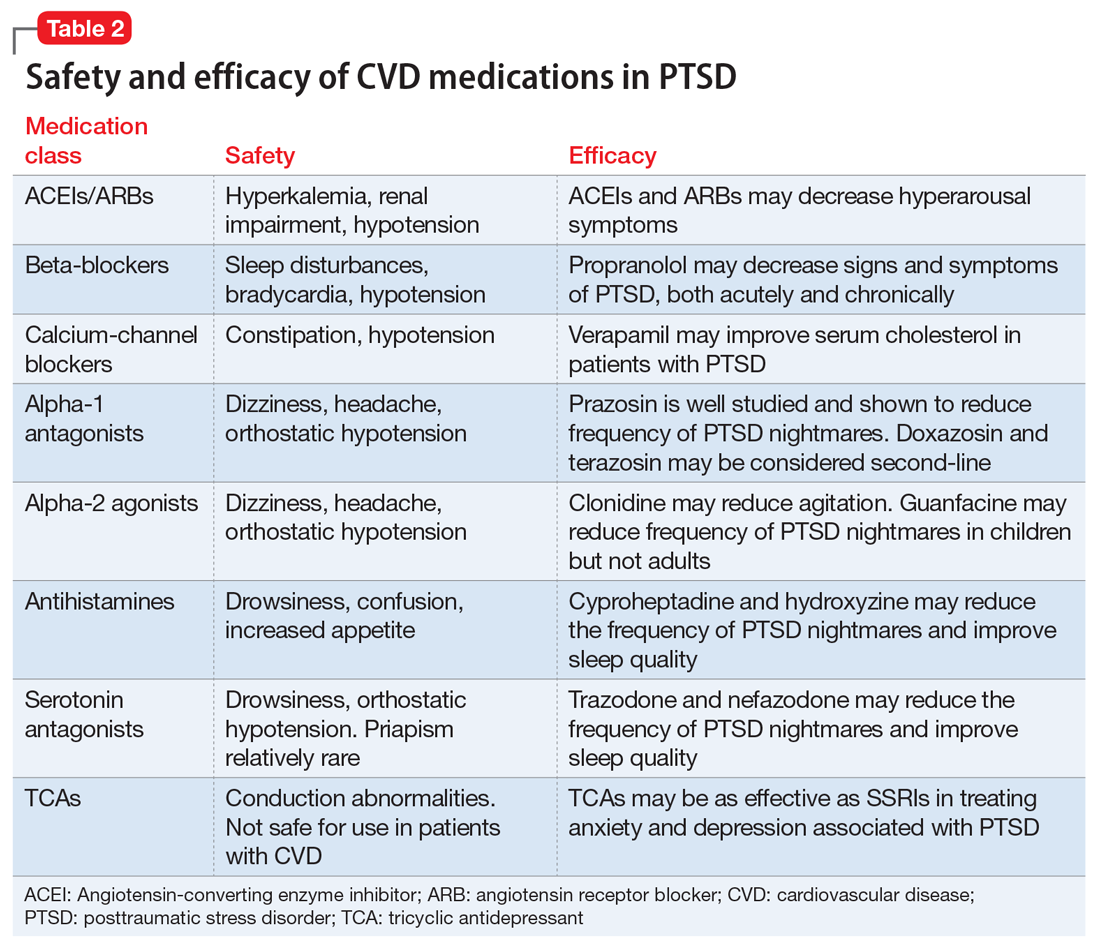
ACEIs, ARBs, beta blockers, and calcium channel blockers
Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) inhibit the renin-angiotensin system: ACEIs prevent formation of angiotensin II, a potent vasoconstrictor, and ARBs prevent interaction between angiotensin II and its receptor. In one study, patients were recruited from a large public hospital serving primarily a highly traumatized, low-income population. Patients taking an ACEI or ARB who had experienced at least 1 traumatic event exhibited significantly decreased hyperarousal symptoms and decreased intrusive thoughts on the PTSD Symptom Scale and Clinician Administered PTSD Scale.5 Other studies have reported that blockade of angiotensin II AT1 receptors may result in decreased stress, anxiety, and inflammation.15

Evidence supports the use of the centrally acting, beta-adrenergic antagonist propranolol for decreasing the physiologic reactivity to acute trauma. Emotional arousal enhances the consolidation of emotional experiences into long-term memories via the adrenal stress hormones epinephrine and corticosterone. The amygdala mediates these stress hormones and releases norepinephrine, which subsequently activates noradrenergic receptors essential for memory enhancement. Several studies have reported that patients who received propranolol within several hours of a traumatic event experienced fewer physiologic signs of PTSD at follow-up 1 month later.16 Moreover, researchers have hypothesized that chronic treatment with propranolol may be effective in decreasing hyperarousal symptoms in patients with chronic PTSD by reducing tonically elevated norepinephrine signaling.6
Chronic elevation of noradrenergic activity may induce lipoprotein lipase and suppress low-density lipoprotein (LDL) receptor activity, which in turn elevates serum cholesterol levels. The results of one study suggested that verapamil, a non-dihydropyridine calcium channel blocker, significantly improves serum cholesterol levels in patients with PTSD by increasing LDL receptor activity and decreasing norepinephrine release.7
Alpha-1 and alpha-2 antagonists
Alpha-1 antagonists relax vascular smooth muscle by blocking norepinephrine stimulation at postsynaptic α-1-adrenergic receptors. They frequently are prescribed for hypertension and benign prostatic hypertrophy. One α-1 antagonist in particular, prazosin, appears especially useful in treating sleep disturbances, which occur in up to 90% of patients with PTSD.17 Because of its relatively greater lipophilicity, prazosin crosses the blood–brain barrier and acts centrally to reduce the fight-or-flight and hyperarousal reactions related to nightmares caused by PTSD.18 Common adverse effects include dizziness and orthostatic hypotension. These usually can be mitigated with titration to effective dose. In a study of active-duty soldiers who returned from Iraq and Afghanistan, Raskind et al8 found that prazosin doses up to 25 mg/d in men and 12 mg/d in women were tolerated with weekly adjustments and blood pressure monitoring.
Other α-1 antagonists have shown efficacy in a limited number of trials and may be considered second-line treatment of PTSD hyperarousal symptoms. Doxazosin has a longer half-life compared with prazosin (22 hours vs 3 hours) and may be useful in treating daytime hyperarousal with once-daily dosing. However, its hydrophilicity prevents it from crossing the blood–brain barrier to the same degree as prazosin.19 Terazosin also has a longer half-life (12 hours) and reaches peak plasma concentration in 1 hour. It undergoes minimal first-pass metabolism, leaving almost the entire circulating dose in the parent form, but clinical data are limited to only a small case report.10
Alpha-2 agonists inhibit sympathetic outflow in the CNS, which ultimately relaxes vascular smooth muscle like α-1 antagonists. Clonidine exhibits sedative properties, which derive from its nonspecific binding to α-2a-, -2b-, and -2c-adrenergic receptors. Several case studies have described a reduction in agitation in PTSD patients with the use of clonidine, likely through the induction of sleep and relaxation. Guanfacine, on the other hand, selectively binds to the α-2a-adrenergic receptor and therefore lacks the sedative properties of clonidine. Several placebo-controlled trials showed no alleviation of PTSD symptoms in adults with the use of guanfacine.11 However, case reports and open-label trials have suggested that guanfacine may reduce trauma-induced nightmares in pediatric patients. Further investigation is needed to clarify the potential use of guanfacine in pediatric PTSD.19
Antihistamines and antidepressants
Several second-line pharmacologic agents may be useful in patients with PTSD who are already taking cardiovascular medication. A limited number of studies have demonstrated reduced frequency of PTSD nightmares with the histamine-1 antagonists cyproheptadine and hydroxyzine, both of which exhibit minor anti-serotonergic properties.12,13 Likewise, the serotonin antagonists nefazodone and trazodone have been shown to reduce the frequency of PTSD nightmares, as well as improve overall sleep quality.14 Nefazodone should be considered an option only after treatment failure of multiple other medications, because it is associated with a small, but significant, risk of life-threatening hepatotoxicity.20
Tricyclic antidepressants (TCAs) may reduce anxiety and depression associated with PTSD to the same degree as SSRIs.21 However, their effect on PTSD-associated sleep disturbances is much less pronounced than other available medications.14 TCAs should be avoided in patients with CVD because they may exacerbate cardiac conduction abnormalities. This is especially true for those recovering from acute MI.22
CASE CONTINUED
Mr. S is started on prazosin, 1 mg at bedtime, titrated weekly to 6 mg at bedtime with regular blood pressure monitoring because of the risk of orthostatic hypotension. Although the frequency of his nightmares decreases to 1 or 2 per month, he still experiences flashbacks at the same frequency and intensity as before. Prazosin, 1 mg every morning, is added, titrated weekly to 4 mg every morning. This combination of morning and bedtime dosing leads to resolution of both nightmares and flashbacks along with a significant reduction in hyperarousal. Lisinopril is increased from 5 to 10 mg/d to address Mr. S’s uncontrolled hypertension; this change also could have contributed to the reduction in hyperarousal. CPT and fluoxetine are continued.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Laslett LJ, Alagona P Jr, Clark BA 3rd, et al. The worldwide environment of cardiovascular disease: prevalence, diagnosis, therapy, and policy issues: a report from the American College of Cardiology. J Am Coll Cardiol. 2012;60(suppl 25):S1-S49.
3. Cohen BE, Marmar C, Ren L, et al. Association of cardiovascular risk factors with mental health diagnoses in Iraq and Afghanistan war veterans using VA health care. JAMA. 2009;302(5):489-492.
4. Rozanski A, Blumenthal JA, Kaplan J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation. 1999;99(16):2192-2217.
5. Khoury NM, Marvar PJ, Gillespie CF, et al. The renin-angiotensin pathway in posttraumatic stress disorder: angiotensin-converting enzyme inhibitors and angiotensin receptor blockers are associated with fewer traumatic stress symptoms. J Clin Psychiatry. 2012;73(6):849-855.
6. Giustino TF, Fitzgerald PJ, Maren S. Revisiting propranolol and PTSD: memory erasure or extinction enhancement? Neurobiol Learn Mem. 2016;130:26-33.
7. Ansari MA, Ahmed S. Calcium channel blocker verapamil: a new intervention for high cholesterol levels in patients with PTSD. Turk Jem. 2007;11:93-97.
8. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. De Jong J, Wauben P, Huijbrechts I, et al. Doxazosin treatment for posttraumatic stress disorder. J Clin Psychopharmacol. 2010;30(1):84-85.
10. Nirmalani-Gandhy A, Sanchez D, Catalano G. Terazosin for the treatment of trauma-related nightmares: a report of four cases. Clin Neuropharmacol. 2015;38(3):109-111.
11. Belkin MR, Schwartz TL. Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context. 2015;4:212286. doi: 10.7573/dic.212286.
12. Gupta S, Popli A, Bathurst E, et al. Efficacy of cyproheptadine for nightmares associated with posttraumatic stress disorder. Compr Psychiatry. 1998;39(3):160-164.
13. Ahmadpanah M, Sabzeiee P, Hosseini SM, et al. Comparing the effect of prazosin and hydroxyzine on sleep quality in patients suffering from posttraumatic stress disorder. Neuropsychobiology. 2014;69(4):235-242.
14. Maher MJ, Rego SA, Asnis GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
15. Saavedra JM, Sánchez-Lemus E, Benicky J. Blockade of brain angiotensin II AT1 receptors ameliorates stress, anxiety, brain inflammation, and ischemia: therapeutic implications. Psychoneuroendocrinology. 2011;36(1):1-18.
16. McGaugh JL. Making lasting memories: remembering the significant. Proc Natl Acad Sci U S A. 2013;110(suppl 2):10402-10407.
17. Writer BW, Meyer EG, Schillerstrom JE. Prazosin for military combat-related PTSD nightmares: a critical review. J Neuropsychiatry Clin Neurosci. 2014;26(1):24-33.
18. Kung S, Espinel Z, Lapid MI. Treatment of nightmares with prazosin: a systematic review. Mayo Clin Proc. 2012;87(9):890-900.
19. Arnsten AF, Raskind MA, Taylor FB, et al. The effects of stress exposure on prefrontal cortex: translating basic research into successful treatments for post-traumatic stress disorder. Neurobiol Stress. 2015;1:89-99.
20. Serzone [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2003.
21. Puetz TW, Youngstedt SD, Herring MP. Effects of pharmacotherapy on combat-related PTSD, anxiety, and depression: a systematic review and meta-regression analysis. PLoS One. 2015;10(5):e0126529. doi: 10.1371/journal. pone.0126529.
22. Glassman AH. Cardiovascular effects of tricyclic antidepressants. Annu Rev Med. 1984;35:503-511.
Mr. S, 64, has a history of posttraumatic stress disorder (PTSD), which has been well controlled for the past 15 years with cognitive-processing therapy and fluoxetine, 40 mg/d. However, over the past 6 weeks, Mr. S has experienced increased hypervigilance, nightmares, and flashbacks. He states that his primary care provider recommended an adjustment in pharmacotherapy to address this exacerbation of symptoms. Previous medication trials include sertraline, 200 mg/d, discontinued due to lack of perceived efficacy, and venlafaxine, 150 mg/d, discontinued due to increased blood pressure.
Mr. S’s medical history includes hypertension, dyslipidemia, and myocardial infarction (MI) 5 years ago. His family history includes sudden cardiac death (mother and father) and major depressive disorder (sister). His blood pressure is currently uncontrolled on lisinopril, 5 mg/d, and metoprolol succinate, 50 mg/d. Today, serial blood pressure readings measured approximately 180/90 mm Hg, with a pulse 50-60 beats per minute.
What is the next step in treating Mr. S’s hypertension and PTSD symptoms? Is there any evidence to support concomitant therapy?
PTSD is characterized by emotional and behavioral symptoms following exposure to a traumatic event. Its 12-month prevalence in the United States is estimated at 3.5%. Diagnostic criteria necessitate the presence of intrusive symptoms, persistent effortful avoidance of distressing trauma-related stimuli, negative cognitions or mood, and alterations in arousal and reactivity. PTSD negatively impacts social and occupational functioning.1
Studies have revealed a correlation between the presence of psychosocial factors, such as depression and anxiety, and the occurrence of cardiovascular events. The mechanism appears to consist of a behavioral component (eg, poor diet, tobacco use) and a direct pathophysiologic component (eg, excessive sympathetic nervous system activation) (Table 13).4 Management of concomitant PTSD and CVD presents a challenge to clinicians.
This article summarizes the evidence for the use of CVD medications in treating PTSD (Table 2) and how to apply these principles in patient care (Table 35-14).

ACEIs, ARBs, beta blockers, and calcium channel blockers
Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) inhibit the renin-angiotensin system: ACEIs prevent formation of angiotensin II, a potent vasoconstrictor, and ARBs prevent interaction between angiotensin II and its receptor. In one study, patients were recruited from a large public hospital serving primarily a highly traumatized, low-income population. Patients taking an ACEI or ARB who had experienced at least 1 traumatic event exhibited significantly decreased hyperarousal symptoms and decreased intrusive thoughts on the PTSD Symptom Scale and Clinician Administered PTSD Scale.5 Other studies have reported that blockade of angiotensin II AT1 receptors may result in decreased stress, anxiety, and inflammation.15

Evidence supports the use of the centrally acting, beta-adrenergic antagonist propranolol for decreasing the physiologic reactivity to acute trauma. Emotional arousal enhances the consolidation of emotional experiences into long-term memories via the adrenal stress hormones epinephrine and corticosterone. The amygdala mediates these stress hormones and releases norepinephrine, which subsequently activates noradrenergic receptors essential for memory enhancement. Several studies have reported that patients who received propranolol within several hours of a traumatic event experienced fewer physiologic signs of PTSD at follow-up 1 month later.16 Moreover, researchers have hypothesized that chronic treatment with propranolol may be effective in decreasing hyperarousal symptoms in patients with chronic PTSD by reducing tonically elevated norepinephrine signaling.6
Chronic elevation of noradrenergic activity may induce lipoprotein lipase and suppress low-density lipoprotein (LDL) receptor activity, which in turn elevates serum cholesterol levels. The results of one study suggested that verapamil, a non-dihydropyridine calcium channel blocker, significantly improves serum cholesterol levels in patients with PTSD by increasing LDL receptor activity and decreasing norepinephrine release.7
Alpha-1 and alpha-2 antagonists
Alpha-1 antagonists relax vascular smooth muscle by blocking norepinephrine stimulation at postsynaptic α-1-adrenergic receptors. They frequently are prescribed for hypertension and benign prostatic hypertrophy. One α-1 antagonist in particular, prazosin, appears especially useful in treating sleep disturbances, which occur in up to 90% of patients with PTSD.17 Because of its relatively greater lipophilicity, prazosin crosses the blood–brain barrier and acts centrally to reduce the fight-or-flight and hyperarousal reactions related to nightmares caused by PTSD.18 Common adverse effects include dizziness and orthostatic hypotension. These usually can be mitigated with titration to effective dose. In a study of active-duty soldiers who returned from Iraq and Afghanistan, Raskind et al8 found that prazosin doses up to 25 mg/d in men and 12 mg/d in women were tolerated with weekly adjustments and blood pressure monitoring.
Other α-1 antagonists have shown efficacy in a limited number of trials and may be considered second-line treatment of PTSD hyperarousal symptoms. Doxazosin has a longer half-life compared with prazosin (22 hours vs 3 hours) and may be useful in treating daytime hyperarousal with once-daily dosing. However, its hydrophilicity prevents it from crossing the blood–brain barrier to the same degree as prazosin.19 Terazosin also has a longer half-life (12 hours) and reaches peak plasma concentration in 1 hour. It undergoes minimal first-pass metabolism, leaving almost the entire circulating dose in the parent form, but clinical data are limited to only a small case report.10
Alpha-2 agonists inhibit sympathetic outflow in the CNS, which ultimately relaxes vascular smooth muscle like α-1 antagonists. Clonidine exhibits sedative properties, which derive from its nonspecific binding to α-2a-, -2b-, and -2c-adrenergic receptors. Several case studies have described a reduction in agitation in PTSD patients with the use of clonidine, likely through the induction of sleep and relaxation. Guanfacine, on the other hand, selectively binds to the α-2a-adrenergic receptor and therefore lacks the sedative properties of clonidine. Several placebo-controlled trials showed no alleviation of PTSD symptoms in adults with the use of guanfacine.11 However, case reports and open-label trials have suggested that guanfacine may reduce trauma-induced nightmares in pediatric patients. Further investigation is needed to clarify the potential use of guanfacine in pediatric PTSD.19
Antihistamines and antidepressants
Several second-line pharmacologic agents may be useful in patients with PTSD who are already taking cardiovascular medication. A limited number of studies have demonstrated reduced frequency of PTSD nightmares with the histamine-1 antagonists cyproheptadine and hydroxyzine, both of which exhibit minor anti-serotonergic properties.12,13 Likewise, the serotonin antagonists nefazodone and trazodone have been shown to reduce the frequency of PTSD nightmares, as well as improve overall sleep quality.14 Nefazodone should be considered an option only after treatment failure of multiple other medications, because it is associated with a small, but significant, risk of life-threatening hepatotoxicity.20
Tricyclic antidepressants (TCAs) may reduce anxiety and depression associated with PTSD to the same degree as SSRIs.21 However, their effect on PTSD-associated sleep disturbances is much less pronounced than other available medications.14 TCAs should be avoided in patients with CVD because they may exacerbate cardiac conduction abnormalities. This is especially true for those recovering from acute MI.22
CASE CONTINUED
Mr. S is started on prazosin, 1 mg at bedtime, titrated weekly to 6 mg at bedtime with regular blood pressure monitoring because of the risk of orthostatic hypotension. Although the frequency of his nightmares decreases to 1 or 2 per month, he still experiences flashbacks at the same frequency and intensity as before. Prazosin, 1 mg every morning, is added, titrated weekly to 4 mg every morning. This combination of morning and bedtime dosing leads to resolution of both nightmares and flashbacks along with a significant reduction in hyperarousal. Lisinopril is increased from 5 to 10 mg/d to address Mr. S’s uncontrolled hypertension; this change also could have contributed to the reduction in hyperarousal. CPT and fluoxetine are continued.
Mr. S, 64, has a history of posttraumatic stress disorder (PTSD), which has been well controlled for the past 15 years with cognitive-processing therapy and fluoxetine, 40 mg/d. However, over the past 6 weeks, Mr. S has experienced increased hypervigilance, nightmares, and flashbacks. He states that his primary care provider recommended an adjustment in pharmacotherapy to address this exacerbation of symptoms. Previous medication trials include sertraline, 200 mg/d, discontinued due to lack of perceived efficacy, and venlafaxine, 150 mg/d, discontinued due to increased blood pressure.
Mr. S’s medical history includes hypertension, dyslipidemia, and myocardial infarction (MI) 5 years ago. His family history includes sudden cardiac death (mother and father) and major depressive disorder (sister). His blood pressure is currently uncontrolled on lisinopril, 5 mg/d, and metoprolol succinate, 50 mg/d. Today, serial blood pressure readings measured approximately 180/90 mm Hg, with a pulse 50-60 beats per minute.
What is the next step in treating Mr. S’s hypertension and PTSD symptoms? Is there any evidence to support concomitant therapy?
PTSD is characterized by emotional and behavioral symptoms following exposure to a traumatic event. Its 12-month prevalence in the United States is estimated at 3.5%. Diagnostic criteria necessitate the presence of intrusive symptoms, persistent effortful avoidance of distressing trauma-related stimuli, negative cognitions or mood, and alterations in arousal and reactivity. PTSD negatively impacts social and occupational functioning.1
Studies have revealed a correlation between the presence of psychosocial factors, such as depression and anxiety, and the occurrence of cardiovascular events. The mechanism appears to consist of a behavioral component (eg, poor diet, tobacco use) and a direct pathophysiologic component (eg, excessive sympathetic nervous system activation) (Table 13).4 Management of concomitant PTSD and CVD presents a challenge to clinicians.
This article summarizes the evidence for the use of CVD medications in treating PTSD (Table 2) and how to apply these principles in patient care (Table 35-14).

ACEIs, ARBs, beta blockers, and calcium channel blockers
Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) inhibit the renin-angiotensin system: ACEIs prevent formation of angiotensin II, a potent vasoconstrictor, and ARBs prevent interaction between angiotensin II and its receptor. In one study, patients were recruited from a large public hospital serving primarily a highly traumatized, low-income population. Patients taking an ACEI or ARB who had experienced at least 1 traumatic event exhibited significantly decreased hyperarousal symptoms and decreased intrusive thoughts on the PTSD Symptom Scale and Clinician Administered PTSD Scale.5 Other studies have reported that blockade of angiotensin II AT1 receptors may result in decreased stress, anxiety, and inflammation.15

Evidence supports the use of the centrally acting, beta-adrenergic antagonist propranolol for decreasing the physiologic reactivity to acute trauma. Emotional arousal enhances the consolidation of emotional experiences into long-term memories via the adrenal stress hormones epinephrine and corticosterone. The amygdala mediates these stress hormones and releases norepinephrine, which subsequently activates noradrenergic receptors essential for memory enhancement. Several studies have reported that patients who received propranolol within several hours of a traumatic event experienced fewer physiologic signs of PTSD at follow-up 1 month later.16 Moreover, researchers have hypothesized that chronic treatment with propranolol may be effective in decreasing hyperarousal symptoms in patients with chronic PTSD by reducing tonically elevated norepinephrine signaling.6
Chronic elevation of noradrenergic activity may induce lipoprotein lipase and suppress low-density lipoprotein (LDL) receptor activity, which in turn elevates serum cholesterol levels. The results of one study suggested that verapamil, a non-dihydropyridine calcium channel blocker, significantly improves serum cholesterol levels in patients with PTSD by increasing LDL receptor activity and decreasing norepinephrine release.7
Alpha-1 and alpha-2 antagonists
Alpha-1 antagonists relax vascular smooth muscle by blocking norepinephrine stimulation at postsynaptic α-1-adrenergic receptors. They frequently are prescribed for hypertension and benign prostatic hypertrophy. One α-1 antagonist in particular, prazosin, appears especially useful in treating sleep disturbances, which occur in up to 90% of patients with PTSD.17 Because of its relatively greater lipophilicity, prazosin crosses the blood–brain barrier and acts centrally to reduce the fight-or-flight and hyperarousal reactions related to nightmares caused by PTSD.18 Common adverse effects include dizziness and orthostatic hypotension. These usually can be mitigated with titration to effective dose. In a study of active-duty soldiers who returned from Iraq and Afghanistan, Raskind et al8 found that prazosin doses up to 25 mg/d in men and 12 mg/d in women were tolerated with weekly adjustments and blood pressure monitoring.
Other α-1 antagonists have shown efficacy in a limited number of trials and may be considered second-line treatment of PTSD hyperarousal symptoms. Doxazosin has a longer half-life compared with prazosin (22 hours vs 3 hours) and may be useful in treating daytime hyperarousal with once-daily dosing. However, its hydrophilicity prevents it from crossing the blood–brain barrier to the same degree as prazosin.19 Terazosin also has a longer half-life (12 hours) and reaches peak plasma concentration in 1 hour. It undergoes minimal first-pass metabolism, leaving almost the entire circulating dose in the parent form, but clinical data are limited to only a small case report.10
Alpha-2 agonists inhibit sympathetic outflow in the CNS, which ultimately relaxes vascular smooth muscle like α-1 antagonists. Clonidine exhibits sedative properties, which derive from its nonspecific binding to α-2a-, -2b-, and -2c-adrenergic receptors. Several case studies have described a reduction in agitation in PTSD patients with the use of clonidine, likely through the induction of sleep and relaxation. Guanfacine, on the other hand, selectively binds to the α-2a-adrenergic receptor and therefore lacks the sedative properties of clonidine. Several placebo-controlled trials showed no alleviation of PTSD symptoms in adults with the use of guanfacine.11 However, case reports and open-label trials have suggested that guanfacine may reduce trauma-induced nightmares in pediatric patients. Further investigation is needed to clarify the potential use of guanfacine in pediatric PTSD.19
Antihistamines and antidepressants
Several second-line pharmacologic agents may be useful in patients with PTSD who are already taking cardiovascular medication. A limited number of studies have demonstrated reduced frequency of PTSD nightmares with the histamine-1 antagonists cyproheptadine and hydroxyzine, both of which exhibit minor anti-serotonergic properties.12,13 Likewise, the serotonin antagonists nefazodone and trazodone have been shown to reduce the frequency of PTSD nightmares, as well as improve overall sleep quality.14 Nefazodone should be considered an option only after treatment failure of multiple other medications, because it is associated with a small, but significant, risk of life-threatening hepatotoxicity.20
Tricyclic antidepressants (TCAs) may reduce anxiety and depression associated with PTSD to the same degree as SSRIs.21 However, their effect on PTSD-associated sleep disturbances is much less pronounced than other available medications.14 TCAs should be avoided in patients with CVD because they may exacerbate cardiac conduction abnormalities. This is especially true for those recovering from acute MI.22
CASE CONTINUED
Mr. S is started on prazosin, 1 mg at bedtime, titrated weekly to 6 mg at bedtime with regular blood pressure monitoring because of the risk of orthostatic hypotension. Although the frequency of his nightmares decreases to 1 or 2 per month, he still experiences flashbacks at the same frequency and intensity as before. Prazosin, 1 mg every morning, is added, titrated weekly to 4 mg every morning. This combination of morning and bedtime dosing leads to resolution of both nightmares and flashbacks along with a significant reduction in hyperarousal. Lisinopril is increased from 5 to 10 mg/d to address Mr. S’s uncontrolled hypertension; this change also could have contributed to the reduction in hyperarousal. CPT and fluoxetine are continued.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Laslett LJ, Alagona P Jr, Clark BA 3rd, et al. The worldwide environment of cardiovascular disease: prevalence, diagnosis, therapy, and policy issues: a report from the American College of Cardiology. J Am Coll Cardiol. 2012;60(suppl 25):S1-S49.
3. Cohen BE, Marmar C, Ren L, et al. Association of cardiovascular risk factors with mental health diagnoses in Iraq and Afghanistan war veterans using VA health care. JAMA. 2009;302(5):489-492.
4. Rozanski A, Blumenthal JA, Kaplan J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation. 1999;99(16):2192-2217.
5. Khoury NM, Marvar PJ, Gillespie CF, et al. The renin-angiotensin pathway in posttraumatic stress disorder: angiotensin-converting enzyme inhibitors and angiotensin receptor blockers are associated with fewer traumatic stress symptoms. J Clin Psychiatry. 2012;73(6):849-855.
6. Giustino TF, Fitzgerald PJ, Maren S. Revisiting propranolol and PTSD: memory erasure or extinction enhancement? Neurobiol Learn Mem. 2016;130:26-33.
7. Ansari MA, Ahmed S. Calcium channel blocker verapamil: a new intervention for high cholesterol levels in patients with PTSD. Turk Jem. 2007;11:93-97.
8. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. De Jong J, Wauben P, Huijbrechts I, et al. Doxazosin treatment for posttraumatic stress disorder. J Clin Psychopharmacol. 2010;30(1):84-85.
10. Nirmalani-Gandhy A, Sanchez D, Catalano G. Terazosin for the treatment of trauma-related nightmares: a report of four cases. Clin Neuropharmacol. 2015;38(3):109-111.
11. Belkin MR, Schwartz TL. Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context. 2015;4:212286. doi: 10.7573/dic.212286.
12. Gupta S, Popli A, Bathurst E, et al. Efficacy of cyproheptadine for nightmares associated with posttraumatic stress disorder. Compr Psychiatry. 1998;39(3):160-164.
13. Ahmadpanah M, Sabzeiee P, Hosseini SM, et al. Comparing the effect of prazosin and hydroxyzine on sleep quality in patients suffering from posttraumatic stress disorder. Neuropsychobiology. 2014;69(4):235-242.
14. Maher MJ, Rego SA, Asnis GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
15. Saavedra JM, Sánchez-Lemus E, Benicky J. Blockade of brain angiotensin II AT1 receptors ameliorates stress, anxiety, brain inflammation, and ischemia: therapeutic implications. Psychoneuroendocrinology. 2011;36(1):1-18.
16. McGaugh JL. Making lasting memories: remembering the significant. Proc Natl Acad Sci U S A. 2013;110(suppl 2):10402-10407.
17. Writer BW, Meyer EG, Schillerstrom JE. Prazosin for military combat-related PTSD nightmares: a critical review. J Neuropsychiatry Clin Neurosci. 2014;26(1):24-33.
18. Kung S, Espinel Z, Lapid MI. Treatment of nightmares with prazosin: a systematic review. Mayo Clin Proc. 2012;87(9):890-900.
19. Arnsten AF, Raskind MA, Taylor FB, et al. The effects of stress exposure on prefrontal cortex: translating basic research into successful treatments for post-traumatic stress disorder. Neurobiol Stress. 2015;1:89-99.
20. Serzone [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2003.
21. Puetz TW, Youngstedt SD, Herring MP. Effects of pharmacotherapy on combat-related PTSD, anxiety, and depression: a systematic review and meta-regression analysis. PLoS One. 2015;10(5):e0126529. doi: 10.1371/journal. pone.0126529.
22. Glassman AH. Cardiovascular effects of tricyclic antidepressants. Annu Rev Med. 1984;35:503-511.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Laslett LJ, Alagona P Jr, Clark BA 3rd, et al. The worldwide environment of cardiovascular disease: prevalence, diagnosis, therapy, and policy issues: a report from the American College of Cardiology. J Am Coll Cardiol. 2012;60(suppl 25):S1-S49.
3. Cohen BE, Marmar C, Ren L, et al. Association of cardiovascular risk factors with mental health diagnoses in Iraq and Afghanistan war veterans using VA health care. JAMA. 2009;302(5):489-492.
4. Rozanski A, Blumenthal JA, Kaplan J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation. 1999;99(16):2192-2217.
5. Khoury NM, Marvar PJ, Gillespie CF, et al. The renin-angiotensin pathway in posttraumatic stress disorder: angiotensin-converting enzyme inhibitors and angiotensin receptor blockers are associated with fewer traumatic stress symptoms. J Clin Psychiatry. 2012;73(6):849-855.
6. Giustino TF, Fitzgerald PJ, Maren S. Revisiting propranolol and PTSD: memory erasure or extinction enhancement? Neurobiol Learn Mem. 2016;130:26-33.
7. Ansari MA, Ahmed S. Calcium channel blocker verapamil: a new intervention for high cholesterol levels in patients with PTSD. Turk Jem. 2007;11:93-97.
8. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. De Jong J, Wauben P, Huijbrechts I, et al. Doxazosin treatment for posttraumatic stress disorder. J Clin Psychopharmacol. 2010;30(1):84-85.
10. Nirmalani-Gandhy A, Sanchez D, Catalano G. Terazosin for the treatment of trauma-related nightmares: a report of four cases. Clin Neuropharmacol. 2015;38(3):109-111.
11. Belkin MR, Schwartz TL. Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context. 2015;4:212286. doi: 10.7573/dic.212286.
12. Gupta S, Popli A, Bathurst E, et al. Efficacy of cyproheptadine for nightmares associated with posttraumatic stress disorder. Compr Psychiatry. 1998;39(3):160-164.
13. Ahmadpanah M, Sabzeiee P, Hosseini SM, et al. Comparing the effect of prazosin and hydroxyzine on sleep quality in patients suffering from posttraumatic stress disorder. Neuropsychobiology. 2014;69(4):235-242.
14. Maher MJ, Rego SA, Asnis GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
15. Saavedra JM, Sánchez-Lemus E, Benicky J. Blockade of brain angiotensin II AT1 receptors ameliorates stress, anxiety, brain inflammation, and ischemia: therapeutic implications. Psychoneuroendocrinology. 2011;36(1):1-18.
16. McGaugh JL. Making lasting memories: remembering the significant. Proc Natl Acad Sci U S A. 2013;110(suppl 2):10402-10407.
17. Writer BW, Meyer EG, Schillerstrom JE. Prazosin for military combat-related PTSD nightmares: a critical review. J Neuropsychiatry Clin Neurosci. 2014;26(1):24-33.
18. Kung S, Espinel Z, Lapid MI. Treatment of nightmares with prazosin: a systematic review. Mayo Clin Proc. 2012;87(9):890-900.
19. Arnsten AF, Raskind MA, Taylor FB, et al. The effects of stress exposure on prefrontal cortex: translating basic research into successful treatments for post-traumatic stress disorder. Neurobiol Stress. 2015;1:89-99.
20. Serzone [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2003.
21. Puetz TW, Youngstedt SD, Herring MP. Effects of pharmacotherapy on combat-related PTSD, anxiety, and depression: a systematic review and meta-regression analysis. PLoS One. 2015;10(5):e0126529. doi: 10.1371/journal. pone.0126529.
22. Glassman AH. Cardiovascular effects of tricyclic antidepressants. Annu Rev Med. 1984;35:503-511.
FIGHT to remember PTSD
Certain clinical features of posttraumatic stress disorder (PTSD) appear in other psychiatric diagnoses and therefore can confound accurate diagnosis and treatment. PTSD is frequently comorbid with other classes of psychiatric disorders, including mood, personality, substance use, and psychotic disorders, which can further complicate diagnostic clarity. Comorbidity in PTSD is important to recognize because it has been associated with worse treatment outcomes.1
In DSM-5, the updated criteria for PTSD included Criterion D: “Negative alterations in cognitions and mood associated with the traumatic event(s) ….”2 In addition to inability to remember an important aspect of the trau
We created the mnemonic FIGHT to help remember the updated DSM-5 criteria for PTSD when considering the differential diagnosis.
Flight. Avoidant symptoms, including efforts to avoid distressing memories, thoughts, or feelings about the traumatic event, as well as avoidance of external reminders.
Intrusive symptoms, such as distressing dreams, intrusive memories, and physiological distress when exposed to cues.
Gloomy cognitions. Negative cognitions and mood associated with the traumatic event.
Hypervigilance. Alterations in arousal, such as irritability, angry outbursts, reckless behavior, and exaggerated startle response.
Trauma. Exposure to actual or threatened death, serious injury, or sexual violence.
A diagnosis of PTSD requires ≥1 month of symptoms that cause significant distress or impairment and are not attributable to the physiological effects of a substance or medical condition. Specifiers in DSM-5 include with depersonalization or derealization, as well as delayed expression.2
Vigilance in the assessment and treatment of PTSD will aid the clinician and patient in producing better care outcomes.
1. Angstman KB, Marcelin A, Gonzalez CA, et al. The impact of posttraumatic stress disorder on the 6-month outcomes in collaborative care management for depression. J Prim Care Community Health. 2016;7(3):159-164.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Publishing; 2013.
Certain clinical features of posttraumatic stress disorder (PTSD) appear in other psychiatric diagnoses and therefore can confound accurate diagnosis and treatment. PTSD is frequently comorbid with other classes of psychiatric disorders, including mood, personality, substance use, and psychotic disorders, which can further complicate diagnostic clarity. Comorbidity in PTSD is important to recognize because it has been associated with worse treatment outcomes.1
In DSM-5, the updated criteria for PTSD included Criterion D: “Negative alterations in cognitions and mood associated with the traumatic event(s) ….”2 In addition to inability to remember an important aspect of the trau
We created the mnemonic FIGHT to help remember the updated DSM-5 criteria for PTSD when considering the differential diagnosis.
Flight. Avoidant symptoms, including efforts to avoid distressing memories, thoughts, or feelings about the traumatic event, as well as avoidance of external reminders.
Intrusive symptoms, such as distressing dreams, intrusive memories, and physiological distress when exposed to cues.
Gloomy cognitions. Negative cognitions and mood associated with the traumatic event.
Hypervigilance. Alterations in arousal, such as irritability, angry outbursts, reckless behavior, and exaggerated startle response.
Trauma. Exposure to actual or threatened death, serious injury, or sexual violence.
A diagnosis of PTSD requires ≥1 month of symptoms that cause significant distress or impairment and are not attributable to the physiological effects of a substance or medical condition. Specifiers in DSM-5 include with depersonalization or derealization, as well as delayed expression.2
Vigilance in the assessment and treatment of PTSD will aid the clinician and patient in producing better care outcomes.
Certain clinical features of posttraumatic stress disorder (PTSD) appear in other psychiatric diagnoses and therefore can confound accurate diagnosis and treatment. PTSD is frequently comorbid with other classes of psychiatric disorders, including mood, personality, substance use, and psychotic disorders, which can further complicate diagnostic clarity. Comorbidity in PTSD is important to recognize because it has been associated with worse treatment outcomes.1
In DSM-5, the updated criteria for PTSD included Criterion D: “Negative alterations in cognitions and mood associated with the traumatic event(s) ….”2 In addition to inability to remember an important aspect of the trau
We created the mnemonic FIGHT to help remember the updated DSM-5 criteria for PTSD when considering the differential diagnosis.
Flight. Avoidant symptoms, including efforts to avoid distressing memories, thoughts, or feelings about the traumatic event, as well as avoidance of external reminders.
Intrusive symptoms, such as distressing dreams, intrusive memories, and physiological distress when exposed to cues.
Gloomy cognitions. Negative cognitions and mood associated with the traumatic event.
Hypervigilance. Alterations in arousal, such as irritability, angry outbursts, reckless behavior, and exaggerated startle response.
Trauma. Exposure to actual or threatened death, serious injury, or sexual violence.
A diagnosis of PTSD requires ≥1 month of symptoms that cause significant distress or impairment and are not attributable to the physiological effects of a substance or medical condition. Specifiers in DSM-5 include with depersonalization or derealization, as well as delayed expression.2
Vigilance in the assessment and treatment of PTSD will aid the clinician and patient in producing better care outcomes.
1. Angstman KB, Marcelin A, Gonzalez CA, et al. The impact of posttraumatic stress disorder on the 6-month outcomes in collaborative care management for depression. J Prim Care Community Health. 2016;7(3):159-164.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Publishing; 2013.
1. Angstman KB, Marcelin A, Gonzalez CA, et al. The impact of posttraumatic stress disorder on the 6-month outcomes in collaborative care management for depression. J Prim Care Community Health. 2016;7(3):159-164.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Publishing; 2013.
VA/DOD elevate psychotherapy over medication in updated PTSD guidelines
MIAMI – New federal clinical practice guidelines for treating posttraumatic stress disorder move trauma-focused psychotherapy ahead of pharmacotherapy as first-line treatment.
“That is quite a statement that is being made, and it’s a big shift in our treatment guidelines,” Lori L. Davis, MD, said at a meeting of the American Society of Clinical Psychopharmacology, formerly the New Clinical Drug Evaluation Unit meeting. Dr. Davis, who coauthored the update for the Departments of Veterans Affairs and of Defense, discussed the guidelines at the meeting in advance of their release this summer.
The guidelines, dated June 2017 and based on evidence reviewed through March 2016, specify that evidence is strong for first-line psychotherapy that includes some form of exposure and/or cognitive restructuring. Such therapies include prolonged exposure, cognitive processing, eye movement desensitization and reprocessing, narrative exposure therapy, brief eclectic psychotherapy, and PTSD-specific cognitive-behavioral therapy.
The Management of Posttraumatic Stress Work Group found that evidence was weakest for non–trauma-focused therapies, such as stress inoculation training and present-centered and interpersonal psychotherapy. Manualized group therapy also was rated as weak, but insufficient evidence was found to rate one type of group therapy over another.
Insufficient evidence also was cited for psychotherapies not specified in other recommendations, such as dialectical behavior therapy, skills training in affect and interpersonal regulation, acceptance and commitment therapy (ACT), seeking safety therapy, or supportive counseling.
“Some of these treatments have been found to be effective for the treatment of other disorders (e.g., ACT for [major depressive disorder]),” the work group members wrote, “but do not have evidence of efficacy in patients with PTSD.”
The recommendations are based on a review of literature from the past 20 years and are congruent with other treatment guidelines internationally, according to Sheila A.M. Rausch, PhD, who directs mental health research and program evaluation for the VA Ann Arbor Healthcare System in Michigan, is a work group member, and served as a panelist at the meeting.
“What is striking is that, based on people reviewing all the evidence out there – and there are different methodologies for doing so – regardless, [the different groups] tend to come to the same conclusions,” Dr. Rausch said.
The updated guidelines cite insufficient evidence for making any recommendations for augmented or combined treatments of psychotherapy and pharmacotherapy.
Medications considered to have a strong evidence base for monotherapy were sertraline, paroxetine, fluoxetine, and venlafaxine. In cases in which a person declines psychotherapy, those medications are recommended as first-line treatment.
Prazosin, typically used to managing nightmares tied to PTSD, received a “suggest against” recommendation, in part because of the discovery of a publication bias, according to Dr. Davis, who is associate chief of staff for research and development at the Tuscaloosa (Ala.) VA Medical Center and a clinical professor of psychiatry and behavioral neurobiology at the University of Alabama in Birmingham and Tuscaloosa. She noted that the work group remained unsure of “how to grapple with this” news that the study drug did not break from placebo in the treatment of nightmares in more than 300 combat veterans with PTSD.
“We are not trying to tell you not to use antidepressants for major depressive disorder or panic disorders that are often comorbid with PTSD, so you have to keep that in mind as you read these guidelines,” Dr. Davis said. Many patients already take antidepressants because they are easily accessed in primary care practices, she added.
The work group found that evidence generally was weak against atypical antipsychotics – particularly in light of the known adverse side effects of those drugs. However, the group singled out risperidone. “We suggest against treatment of PTSD with quetiapine, olanzapine, and other atypical antipsychotics (except for risperidone, which is a ‘strong against,’ see recommendation 20),” the guidelines suggest.
They also “suggested against” two antidepressants – citalopram and amitriptyline, a tricyclic. The quality of evidence for those drugs was deemed low. Two drugs – lamotrigine and topiramate – also received “suggested against” rankings, and the evidence for the use of those drugs was deemed very low.
Meanwhile, the work group wrote that the quality of evidence was low and “recommended against” the use of three other drugs: divalproex, tiagabine, and guanfacine. They also found the quality of evidence to be very low for the drug class of benzodiazepines and four drugs – risperidone, D-cycloserine, ketamine, and hydrocortisone. They also found insufficient evidence to recommend complementary and integrative health (CIH) practices such as meditation and yoga for treating PTSD. The work group did offer a caveat, however. “It is important to clarify that we are not recommending against the treatments but rather are saying that, at this time, the research does not support the use of any CIH practice for the primary treatment of PTSD.”
Cannabis and its derivatives also were recommended against by the work group because of a “lack of evidence for their efficacy, known adverse effects, and associated risks.”
Dr. Davis said the work group’s findings underscored a crisis in pharmacotherapeutic research in PTSD. Since 2006, there have been only four industry-sponsored drugs for PTSD. “I find it kind of sad,” she said.
Currently, Dr. Rausch’s own study for the VA focuses on comparing the effects of combination therapies for PTSD in the context of patient preferences, in order to tailor the treatments accordingly. In 223 returning veterans with PTSD, she and her colleagues are conducting a head-to-head comparison of prolonged exposure therapy plus placebo, prolonged exposure plus sertraline, and sertraline plus emotion memory management therapy to determine how starting a patient on both kinds of treatments simultaneously affect response.
“It’s clinically important to find out what is really going on in the brain and body,” she said. “Although we know we have therapies that work, we need to find answers for partial responders ... and how to increase the speed and magnitude of response.”
Dr. Davis’s research is funded in part by the VA, Forest, Merck, Tonix, and Otsuka, for whom she also acts as a consultant. Dr. Rausch did not have any disclosures.
MIAMI – New federal clinical practice guidelines for treating posttraumatic stress disorder move trauma-focused psychotherapy ahead of pharmacotherapy as first-line treatment.
“That is quite a statement that is being made, and it’s a big shift in our treatment guidelines,” Lori L. Davis, MD, said at a meeting of the American Society of Clinical Psychopharmacology, formerly the New Clinical Drug Evaluation Unit meeting. Dr. Davis, who coauthored the update for the Departments of Veterans Affairs and of Defense, discussed the guidelines at the meeting in advance of their release this summer.
The guidelines, dated June 2017 and based on evidence reviewed through March 2016, specify that evidence is strong for first-line psychotherapy that includes some form of exposure and/or cognitive restructuring. Such therapies include prolonged exposure, cognitive processing, eye movement desensitization and reprocessing, narrative exposure therapy, brief eclectic psychotherapy, and PTSD-specific cognitive-behavioral therapy.
The Management of Posttraumatic Stress Work Group found that evidence was weakest for non–trauma-focused therapies, such as stress inoculation training and present-centered and interpersonal psychotherapy. Manualized group therapy also was rated as weak, but insufficient evidence was found to rate one type of group therapy over another.
Insufficient evidence also was cited for psychotherapies not specified in other recommendations, such as dialectical behavior therapy, skills training in affect and interpersonal regulation, acceptance and commitment therapy (ACT), seeking safety therapy, or supportive counseling.
“Some of these treatments have been found to be effective for the treatment of other disorders (e.g., ACT for [major depressive disorder]),” the work group members wrote, “but do not have evidence of efficacy in patients with PTSD.”
The recommendations are based on a review of literature from the past 20 years and are congruent with other treatment guidelines internationally, according to Sheila A.M. Rausch, PhD, who directs mental health research and program evaluation for the VA Ann Arbor Healthcare System in Michigan, is a work group member, and served as a panelist at the meeting.
“What is striking is that, based on people reviewing all the evidence out there – and there are different methodologies for doing so – regardless, [the different groups] tend to come to the same conclusions,” Dr. Rausch said.
The updated guidelines cite insufficient evidence for making any recommendations for augmented or combined treatments of psychotherapy and pharmacotherapy.
Medications considered to have a strong evidence base for monotherapy were sertraline, paroxetine, fluoxetine, and venlafaxine. In cases in which a person declines psychotherapy, those medications are recommended as first-line treatment.
Prazosin, typically used to managing nightmares tied to PTSD, received a “suggest against” recommendation, in part because of the discovery of a publication bias, according to Dr. Davis, who is associate chief of staff for research and development at the Tuscaloosa (Ala.) VA Medical Center and a clinical professor of psychiatry and behavioral neurobiology at the University of Alabama in Birmingham and Tuscaloosa. She noted that the work group remained unsure of “how to grapple with this” news that the study drug did not break from placebo in the treatment of nightmares in more than 300 combat veterans with PTSD.
“We are not trying to tell you not to use antidepressants for major depressive disorder or panic disorders that are often comorbid with PTSD, so you have to keep that in mind as you read these guidelines,” Dr. Davis said. Many patients already take antidepressants because they are easily accessed in primary care practices, she added.
The work group found that evidence generally was weak against atypical antipsychotics – particularly in light of the known adverse side effects of those drugs. However, the group singled out risperidone. “We suggest against treatment of PTSD with quetiapine, olanzapine, and other atypical antipsychotics (except for risperidone, which is a ‘strong against,’ see recommendation 20),” the guidelines suggest.
They also “suggested against” two antidepressants – citalopram and amitriptyline, a tricyclic. The quality of evidence for those drugs was deemed low. Two drugs – lamotrigine and topiramate – also received “suggested against” rankings, and the evidence for the use of those drugs was deemed very low.
Meanwhile, the work group wrote that the quality of evidence was low and “recommended against” the use of three other drugs: divalproex, tiagabine, and guanfacine. They also found the quality of evidence to be very low for the drug class of benzodiazepines and four drugs – risperidone, D-cycloserine, ketamine, and hydrocortisone. They also found insufficient evidence to recommend complementary and integrative health (CIH) practices such as meditation and yoga for treating PTSD. The work group did offer a caveat, however. “It is important to clarify that we are not recommending against the treatments but rather are saying that, at this time, the research does not support the use of any CIH practice for the primary treatment of PTSD.”
Cannabis and its derivatives also were recommended against by the work group because of a “lack of evidence for their efficacy, known adverse effects, and associated risks.”
Dr. Davis said the work group’s findings underscored a crisis in pharmacotherapeutic research in PTSD. Since 2006, there have been only four industry-sponsored drugs for PTSD. “I find it kind of sad,” she said.
Currently, Dr. Rausch’s own study for the VA focuses on comparing the effects of combination therapies for PTSD in the context of patient preferences, in order to tailor the treatments accordingly. In 223 returning veterans with PTSD, she and her colleagues are conducting a head-to-head comparison of prolonged exposure therapy plus placebo, prolonged exposure plus sertraline, and sertraline plus emotion memory management therapy to determine how starting a patient on both kinds of treatments simultaneously affect response.
“It’s clinically important to find out what is really going on in the brain and body,” she said. “Although we know we have therapies that work, we need to find answers for partial responders ... and how to increase the speed and magnitude of response.”
Dr. Davis’s research is funded in part by the VA, Forest, Merck, Tonix, and Otsuka, for whom she also acts as a consultant. Dr. Rausch did not have any disclosures.
MIAMI – New federal clinical practice guidelines for treating posttraumatic stress disorder move trauma-focused psychotherapy ahead of pharmacotherapy as first-line treatment.
“That is quite a statement that is being made, and it’s a big shift in our treatment guidelines,” Lori L. Davis, MD, said at a meeting of the American Society of Clinical Psychopharmacology, formerly the New Clinical Drug Evaluation Unit meeting. Dr. Davis, who coauthored the update for the Departments of Veterans Affairs and of Defense, discussed the guidelines at the meeting in advance of their release this summer.
The guidelines, dated June 2017 and based on evidence reviewed through March 2016, specify that evidence is strong for first-line psychotherapy that includes some form of exposure and/or cognitive restructuring. Such therapies include prolonged exposure, cognitive processing, eye movement desensitization and reprocessing, narrative exposure therapy, brief eclectic psychotherapy, and PTSD-specific cognitive-behavioral therapy.
The Management of Posttraumatic Stress Work Group found that evidence was weakest for non–trauma-focused therapies, such as stress inoculation training and present-centered and interpersonal psychotherapy. Manualized group therapy also was rated as weak, but insufficient evidence was found to rate one type of group therapy over another.
Insufficient evidence also was cited for psychotherapies not specified in other recommendations, such as dialectical behavior therapy, skills training in affect and interpersonal regulation, acceptance and commitment therapy (ACT), seeking safety therapy, or supportive counseling.
“Some of these treatments have been found to be effective for the treatment of other disorders (e.g., ACT for [major depressive disorder]),” the work group members wrote, “but do not have evidence of efficacy in patients with PTSD.”
The recommendations are based on a review of literature from the past 20 years and are congruent with other treatment guidelines internationally, according to Sheila A.M. Rausch, PhD, who directs mental health research and program evaluation for the VA Ann Arbor Healthcare System in Michigan, is a work group member, and served as a panelist at the meeting.
“What is striking is that, based on people reviewing all the evidence out there – and there are different methodologies for doing so – regardless, [the different groups] tend to come to the same conclusions,” Dr. Rausch said.
The updated guidelines cite insufficient evidence for making any recommendations for augmented or combined treatments of psychotherapy and pharmacotherapy.
Medications considered to have a strong evidence base for monotherapy were sertraline, paroxetine, fluoxetine, and venlafaxine. In cases in which a person declines psychotherapy, those medications are recommended as first-line treatment.
Prazosin, typically used to managing nightmares tied to PTSD, received a “suggest against” recommendation, in part because of the discovery of a publication bias, according to Dr. Davis, who is associate chief of staff for research and development at the Tuscaloosa (Ala.) VA Medical Center and a clinical professor of psychiatry and behavioral neurobiology at the University of Alabama in Birmingham and Tuscaloosa. She noted that the work group remained unsure of “how to grapple with this” news that the study drug did not break from placebo in the treatment of nightmares in more than 300 combat veterans with PTSD.
“We are not trying to tell you not to use antidepressants for major depressive disorder or panic disorders that are often comorbid with PTSD, so you have to keep that in mind as you read these guidelines,” Dr. Davis said. Many patients already take antidepressants because they are easily accessed in primary care practices, she added.
The work group found that evidence generally was weak against atypical antipsychotics – particularly in light of the known adverse side effects of those drugs. However, the group singled out risperidone. “We suggest against treatment of PTSD with quetiapine, olanzapine, and other atypical antipsychotics (except for risperidone, which is a ‘strong against,’ see recommendation 20),” the guidelines suggest.
They also “suggested against” two antidepressants – citalopram and amitriptyline, a tricyclic. The quality of evidence for those drugs was deemed low. Two drugs – lamotrigine and topiramate – also received “suggested against” rankings, and the evidence for the use of those drugs was deemed very low.
Meanwhile, the work group wrote that the quality of evidence was low and “recommended against” the use of three other drugs: divalproex, tiagabine, and guanfacine. They also found the quality of evidence to be very low for the drug class of benzodiazepines and four drugs – risperidone, D-cycloserine, ketamine, and hydrocortisone. They also found insufficient evidence to recommend complementary and integrative health (CIH) practices such as meditation and yoga for treating PTSD. The work group did offer a caveat, however. “It is important to clarify that we are not recommending against the treatments but rather are saying that, at this time, the research does not support the use of any CIH practice for the primary treatment of PTSD.”
Cannabis and its derivatives also were recommended against by the work group because of a “lack of evidence for their efficacy, known adverse effects, and associated risks.”
Dr. Davis said the work group’s findings underscored a crisis in pharmacotherapeutic research in PTSD. Since 2006, there have been only four industry-sponsored drugs for PTSD. “I find it kind of sad,” she said.
Currently, Dr. Rausch’s own study for the VA focuses on comparing the effects of combination therapies for PTSD in the context of patient preferences, in order to tailor the treatments accordingly. In 223 returning veterans with PTSD, she and her colleagues are conducting a head-to-head comparison of prolonged exposure therapy plus placebo, prolonged exposure plus sertraline, and sertraline plus emotion memory management therapy to determine how starting a patient on both kinds of treatments simultaneously affect response.
“It’s clinically important to find out what is really going on in the brain and body,” she said. “Although we know we have therapies that work, we need to find answers for partial responders ... and how to increase the speed and magnitude of response.”
Dr. Davis’s research is funded in part by the VA, Forest, Merck, Tonix, and Otsuka, for whom she also acts as a consultant. Dr. Rausch did not have any disclosures.
EXPERT ANALYSIS FROM THE ASCP ANNUAL MEETING
Add-on aripiprazole shows modest improvement over bupropion in depression
In men with major depressive disorder (MMD) who were not responding well to an antidepressant treatment, adding aripiprazole resulted in a modest increase in the likelihood of remission, compared with switching to bupropion monotherapy, according to results from a new randomized study.
The findings, published online July 11 in JAMA, come from a randomized, single-blinded trial enrolling more than 1,500 Veterans Health Administration patients (85% men; mean age, 54) with persistent MDD symptoms despite a trial of at least 1 antidepressant drug (JAMA. 2017;318[2]:132-45.).
The VA Augmentation and Switching Treatments for Improving Depression Outcomes (VAST-D) study, led by Somaia Mohamed, MD, PhD, of the VA Connecticut Healthcare System in West Haven, Conn., and carried out at 35 Veterans Health Administration treatment centers, was designed to help answer some of the lingering questions about augmentation strategies that the landmark, federally funded Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, from 2006, could not answer.
While the STAR*D investigators found bupropion to be an effective switching and augmentation agent in people who had failed citalopram monotherapy, the study was not powered to compare results for switching and augmentation. Nor did STAR*D include augmentation with atypical antipsychotics such as aripiprazole, a treatment strategy that was not yet in wide use.
The VAST-D study also differed from most depression trials in that most of the patients were men. The main outcome was clinical remission within 12 weeks of initiating one of the three treatment strategies. Subjects were randomized to augmentation of current treatment with aripiprazole 2-15 mg (n = 505) or bupropion 150-400 mg (n = 506) or were switched from current treatment to bupropion monotherapy (n = 511).
Remission rates during the first 12 weeks were 22% for patients switched from their current drug to bupropion, 27% for those who augmented treatment with bupropion, and 29% for those who augmented treatment with aripiprazole, though the difference in remission reached statistical significance only for the adjunctive aripiprazole group vs. the bupropion monotherapy group (relative risk, 1.30; 95% confidence interval, 1.05-1.60; P = .02).
Clinical response, as measured using two validated scoring systems, was higher (74%) in the aripiprazole group at 12 weeks, compared with the bupropion only (62%) or bupropion augmentation (66%) groups, a difference that reached statistical significance. Anxiety was more commonly reported among patients in the bupropion groups, while somnolence, akathisia, and weight gain were more frequently reported among patients treated with aripiprazole.
Though aripiprazole augmentation was seen associated with a higher rate of remission and greater response, Dr. Mohamed and her colleagues cautioned that, “given the small effect size and adverse effects” associated with the atypical antipsychotic drug, “further analysis, including cost-effectiveness, is needed to understand the net utility of this approach.”
The Department of Veterans Affairs sponsored the study. Bristol-Myers Squibb provided aripiprazole to investigators. Of the study’s 16 listed coauthors, 5 reported financial relationships with Bristol-Myers Squibb or other manufacturers.
“The modest advantage of aripiprazole augmentation over the two other treatment options in the VAST-D study suggests that this approach should be considered earlier by clinicians for patients with MDD who have an inadequate response to antidepressant therapies,” Maurizio Fava, MD, wrote in an editorial accompanying the study published by JAMA.
He also pointed out that the demographic makeup of the patients study was largely male, which is significantly different “from the usual study population in large trials of MDD, in which women typically far exceed the proportion of male study participants, as was the case with STAR*D.”
Dr. Fava cited the differences in the proportion of patients with posttraumatic stress disorder in the VAST-D and STAR*D study. In VAST-D, 44%-48% had PTSD, compared with a rate of 17% in the STAR*D cohort. “Did such enrichment potentially favor one of the treatment options of VAST-D? Although a placebo-controlled study of bupropion in chronic PTSD showed no benefit of this therapy, a meta-analysis of eight randomized, double-blind, placebo-controlled clinical trials of atypical antipsychotics for the treatment of PTSD found that these agents may be superior to placebo,” Dr. Fava wrote.
Finally, he cited the differences in mean age of onset of MDD in the two studies. In the VAST-D study, the mean age of onset ranged from 36 to 38 years and compared with a mean age of onset of 25 in STAR*D. “The older age of onset of MDD in the VAST-D study suggests that, in many cases, the MDD may have been secondary to other psychiatric conditions, such as PTSD. Therefore, there might be some significant neurobiological differences between the two populations,” Dr. Fava wrote (JAMA. 2017;318[2]:126-8).
Dr. Fava is affiliated with the division of clinical research at Massachusetts General Hospital, Boston. He reported financial relationships with numerous pharmaceutical companies.
“The modest advantage of aripiprazole augmentation over the two other treatment options in the VAST-D study suggests that this approach should be considered earlier by clinicians for patients with MDD who have an inadequate response to antidepressant therapies,” Maurizio Fava, MD, wrote in an editorial accompanying the study published by JAMA.
He also pointed out that the demographic makeup of the patients study was largely male, which is significantly different “from the usual study population in large trials of MDD, in which women typically far exceed the proportion of male study participants, as was the case with STAR*D.”
Dr. Fava cited the differences in the proportion of patients with posttraumatic stress disorder in the VAST-D and STAR*D study. In VAST-D, 44%-48% had PTSD, compared with a rate of 17% in the STAR*D cohort. “Did such enrichment potentially favor one of the treatment options of VAST-D? Although a placebo-controlled study of bupropion in chronic PTSD showed no benefit of this therapy, a meta-analysis of eight randomized, double-blind, placebo-controlled clinical trials of atypical antipsychotics for the treatment of PTSD found that these agents may be superior to placebo,” Dr. Fava wrote.
Finally, he cited the differences in mean age of onset of MDD in the two studies. In the VAST-D study, the mean age of onset ranged from 36 to 38 years and compared with a mean age of onset of 25 in STAR*D. “The older age of onset of MDD in the VAST-D study suggests that, in many cases, the MDD may have been secondary to other psychiatric conditions, such as PTSD. Therefore, there might be some significant neurobiological differences between the two populations,” Dr. Fava wrote (JAMA. 2017;318[2]:126-8).
Dr. Fava is affiliated with the division of clinical research at Massachusetts General Hospital, Boston. He reported financial relationships with numerous pharmaceutical companies.
“The modest advantage of aripiprazole augmentation over the two other treatment options in the VAST-D study suggests that this approach should be considered earlier by clinicians for patients with MDD who have an inadequate response to antidepressant therapies,” Maurizio Fava, MD, wrote in an editorial accompanying the study published by JAMA.
He also pointed out that the demographic makeup of the patients study was largely male, which is significantly different “from the usual study population in large trials of MDD, in which women typically far exceed the proportion of male study participants, as was the case with STAR*D.”
Dr. Fava cited the differences in the proportion of patients with posttraumatic stress disorder in the VAST-D and STAR*D study. In VAST-D, 44%-48% had PTSD, compared with a rate of 17% in the STAR*D cohort. “Did such enrichment potentially favor one of the treatment options of VAST-D? Although a placebo-controlled study of bupropion in chronic PTSD showed no benefit of this therapy, a meta-analysis of eight randomized, double-blind, placebo-controlled clinical trials of atypical antipsychotics for the treatment of PTSD found that these agents may be superior to placebo,” Dr. Fava wrote.
Finally, he cited the differences in mean age of onset of MDD in the two studies. In the VAST-D study, the mean age of onset ranged from 36 to 38 years and compared with a mean age of onset of 25 in STAR*D. “The older age of onset of MDD in the VAST-D study suggests that, in many cases, the MDD may have been secondary to other psychiatric conditions, such as PTSD. Therefore, there might be some significant neurobiological differences between the two populations,” Dr. Fava wrote (JAMA. 2017;318[2]:126-8).
Dr. Fava is affiliated with the division of clinical research at Massachusetts General Hospital, Boston. He reported financial relationships with numerous pharmaceutical companies.
In men with major depressive disorder (MMD) who were not responding well to an antidepressant treatment, adding aripiprazole resulted in a modest increase in the likelihood of remission, compared with switching to bupropion monotherapy, according to results from a new randomized study.
The findings, published online July 11 in JAMA, come from a randomized, single-blinded trial enrolling more than 1,500 Veterans Health Administration patients (85% men; mean age, 54) with persistent MDD symptoms despite a trial of at least 1 antidepressant drug (JAMA. 2017;318[2]:132-45.).
The VA Augmentation and Switching Treatments for Improving Depression Outcomes (VAST-D) study, led by Somaia Mohamed, MD, PhD, of the VA Connecticut Healthcare System in West Haven, Conn., and carried out at 35 Veterans Health Administration treatment centers, was designed to help answer some of the lingering questions about augmentation strategies that the landmark, federally funded Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, from 2006, could not answer.
While the STAR*D investigators found bupropion to be an effective switching and augmentation agent in people who had failed citalopram monotherapy, the study was not powered to compare results for switching and augmentation. Nor did STAR*D include augmentation with atypical antipsychotics such as aripiprazole, a treatment strategy that was not yet in wide use.
The VAST-D study also differed from most depression trials in that most of the patients were men. The main outcome was clinical remission within 12 weeks of initiating one of the three treatment strategies. Subjects were randomized to augmentation of current treatment with aripiprazole 2-15 mg (n = 505) or bupropion 150-400 mg (n = 506) or were switched from current treatment to bupropion monotherapy (n = 511).
Remission rates during the first 12 weeks were 22% for patients switched from their current drug to bupropion, 27% for those who augmented treatment with bupropion, and 29% for those who augmented treatment with aripiprazole, though the difference in remission reached statistical significance only for the adjunctive aripiprazole group vs. the bupropion monotherapy group (relative risk, 1.30; 95% confidence interval, 1.05-1.60; P = .02).
Clinical response, as measured using two validated scoring systems, was higher (74%) in the aripiprazole group at 12 weeks, compared with the bupropion only (62%) or bupropion augmentation (66%) groups, a difference that reached statistical significance. Anxiety was more commonly reported among patients in the bupropion groups, while somnolence, akathisia, and weight gain were more frequently reported among patients treated with aripiprazole.
Though aripiprazole augmentation was seen associated with a higher rate of remission and greater response, Dr. Mohamed and her colleagues cautioned that, “given the small effect size and adverse effects” associated with the atypical antipsychotic drug, “further analysis, including cost-effectiveness, is needed to understand the net utility of this approach.”
The Department of Veterans Affairs sponsored the study. Bristol-Myers Squibb provided aripiprazole to investigators. Of the study’s 16 listed coauthors, 5 reported financial relationships with Bristol-Myers Squibb or other manufacturers.
In men with major depressive disorder (MMD) who were not responding well to an antidepressant treatment, adding aripiprazole resulted in a modest increase in the likelihood of remission, compared with switching to bupropion monotherapy, according to results from a new randomized study.
The findings, published online July 11 in JAMA, come from a randomized, single-blinded trial enrolling more than 1,500 Veterans Health Administration patients (85% men; mean age, 54) with persistent MDD symptoms despite a trial of at least 1 antidepressant drug (JAMA. 2017;318[2]:132-45.).
The VA Augmentation and Switching Treatments for Improving Depression Outcomes (VAST-D) study, led by Somaia Mohamed, MD, PhD, of the VA Connecticut Healthcare System in West Haven, Conn., and carried out at 35 Veterans Health Administration treatment centers, was designed to help answer some of the lingering questions about augmentation strategies that the landmark, federally funded Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, from 2006, could not answer.
While the STAR*D investigators found bupropion to be an effective switching and augmentation agent in people who had failed citalopram monotherapy, the study was not powered to compare results for switching and augmentation. Nor did STAR*D include augmentation with atypical antipsychotics such as aripiprazole, a treatment strategy that was not yet in wide use.
The VAST-D study also differed from most depression trials in that most of the patients were men. The main outcome was clinical remission within 12 weeks of initiating one of the three treatment strategies. Subjects were randomized to augmentation of current treatment with aripiprazole 2-15 mg (n = 505) or bupropion 150-400 mg (n = 506) or were switched from current treatment to bupropion monotherapy (n = 511).
Remission rates during the first 12 weeks were 22% for patients switched from their current drug to bupropion, 27% for those who augmented treatment with bupropion, and 29% for those who augmented treatment with aripiprazole, though the difference in remission reached statistical significance only for the adjunctive aripiprazole group vs. the bupropion monotherapy group (relative risk, 1.30; 95% confidence interval, 1.05-1.60; P = .02).
Clinical response, as measured using two validated scoring systems, was higher (74%) in the aripiprazole group at 12 weeks, compared with the bupropion only (62%) or bupropion augmentation (66%) groups, a difference that reached statistical significance. Anxiety was more commonly reported among patients in the bupropion groups, while somnolence, akathisia, and weight gain were more frequently reported among patients treated with aripiprazole.
Though aripiprazole augmentation was seen associated with a higher rate of remission and greater response, Dr. Mohamed and her colleagues cautioned that, “given the small effect size and adverse effects” associated with the atypical antipsychotic drug, “further analysis, including cost-effectiveness, is needed to understand the net utility of this approach.”
The Department of Veterans Affairs sponsored the study. Bristol-Myers Squibb provided aripiprazole to investigators. Of the study’s 16 listed coauthors, 5 reported financial relationships with Bristol-Myers Squibb or other manufacturers.
FROM JAMA
Key clinical point: Aripiprazole added to a current antidepressant was associated with a modestly higher remission rate among men with MDD than was switching to bupropion monotherapy.
Major finding: Remission by 12 weeks was 28.9% among patients receiving add-on aripiprazole, vs. 22.3% for bupropion alone (RR, 1.30; 95% CI, 1.05-1.60; P = 0.02).
Data source: A randomized, single-blinded multicenter trial enrolling more than 1,500 patients (85% men) with persistent MDD despite treatment.
Disclosures: The Veterans Health Administration sponsored the study. One of the study drugs was donated by a manufacturer, and 5 of 16 coauthors disclosed financial conflicts of interest.
New Center of Excellence to Lead Research of “Signature Wounds”
Take a brand-new research facility, then add a neighboring U.S. Army base with one of the largest veteran populations of any health care network and a world-class team of researchers—that’s a “recipe for success,” says Dr. Michael Russell, director of the VA Center of Excellence for Research on Returning War Veterans in Waco, Texas.
The 53,000-square-foot center is designed to conduct state-of-the-art research on mental health problems associated with PTSD and TBI, “signature wounds” of conflicts in Afghanistan and the Middle East. The flagship study is named Project MAVEREX. Researchers will examine whether the inability of the regions in injured brains to communicate with one another worsens behavior outcomes. Using “cutting-edge data analysis techniques,” they hope to characterize the effects of TBI on brain structure and function “with very high precision,” says Dr. Evan Gordon, a cognitive neuroscientist working on MAVEREX.
The Center of Excellence is on the campus of the historic Doris Miller VAMC. The facility has space for 75 staff members and faculty as well as 25 trainees. It features multiple examination rooms, observation rooms, electrocardiography, electroencephalography, a 3 Tesla MRI, a transcranial magnetic stimulation suite, and a custom-built laboratory wing.
Take a brand-new research facility, then add a neighboring U.S. Army base with one of the largest veteran populations of any health care network and a world-class team of researchers—that’s a “recipe for success,” says Dr. Michael Russell, director of the VA Center of Excellence for Research on Returning War Veterans in Waco, Texas.
The 53,000-square-foot center is designed to conduct state-of-the-art research on mental health problems associated with PTSD and TBI, “signature wounds” of conflicts in Afghanistan and the Middle East. The flagship study is named Project MAVEREX. Researchers will examine whether the inability of the regions in injured brains to communicate with one another worsens behavior outcomes. Using “cutting-edge data analysis techniques,” they hope to characterize the effects of TBI on brain structure and function “with very high precision,” says Dr. Evan Gordon, a cognitive neuroscientist working on MAVEREX.
The Center of Excellence is on the campus of the historic Doris Miller VAMC. The facility has space for 75 staff members and faculty as well as 25 trainees. It features multiple examination rooms, observation rooms, electrocardiography, electroencephalography, a 3 Tesla MRI, a transcranial magnetic stimulation suite, and a custom-built laboratory wing.
Take a brand-new research facility, then add a neighboring U.S. Army base with one of the largest veteran populations of any health care network and a world-class team of researchers—that’s a “recipe for success,” says Dr. Michael Russell, director of the VA Center of Excellence for Research on Returning War Veterans in Waco, Texas.
The 53,000-square-foot center is designed to conduct state-of-the-art research on mental health problems associated with PTSD and TBI, “signature wounds” of conflicts in Afghanistan and the Middle East. The flagship study is named Project MAVEREX. Researchers will examine whether the inability of the regions in injured brains to communicate with one another worsens behavior outcomes. Using “cutting-edge data analysis techniques,” they hope to characterize the effects of TBI on brain structure and function “with very high precision,” says Dr. Evan Gordon, a cognitive neuroscientist working on MAVEREX.
The Center of Excellence is on the campus of the historic Doris Miller VAMC. The facility has space for 75 staff members and faculty as well as 25 trainees. It features multiple examination rooms, observation rooms, electrocardiography, electroencephalography, a 3 Tesla MRI, a transcranial magnetic stimulation suite, and a custom-built laboratory wing.
Relieving PTSD Symptoms May Cut Risk of Myocardial Infarction and Stroke
Women with severe posttraumatic stress disorder (PTSD) symptoms have a nearly 70% increase in the incidence of cardiovascular disease (CVD), according to a study by researchers from Harvard and Brigham and Women’s Hospital in Boston, Massachusetts, Columbia University in New York, and University of California in San Francisco.
The researchers analyzed data from 49,859 women in the Nurses’ Health Study II. Over 20 years, there were 552 confirmed cases of myocardial infarction or stroke.
Women with 6 to 7 symptoms of trauma and PTSD had the highest risk. Women with trauma but no PTSD symptoms had a 30% higher risk. When women who said illness was their worst trauma were excluded, the risk of CVD doubled among those with trauma and severe PTSD symptoms and increased by 88% in women with trauma and moderate PTSD symptoms.
Strikingly, the researchers also found that when the PTSD symptoms declined so did the CVD risk. The researchers note that CVD risk due to other well-known risk factors, such as smoking, increases with exposure duration declines once the risk factor is eliminated. In this study, for every 5 additional years PTSD symptoms lasted, the odds of CVD were 9% higher.
A “more nuanced understanding” of the role of health behaviors could add insight into how PTSD influences the risk of CVD, the researchers say. They point to studies that have found a link between PTSD and cardiotoxic behaviors such as smoking, drinking, and diet. Physiologic alterations that occur with PTSD symptoms also may play an important role, they suggest, such as changes in neuropeptide Y in response to stress, which might contribute to metabolic syndrome.
Citing “particularly intriguing” findings from a study that found symptoms eventually remitted in 44% of individuals with PTSD, the researchers say providing treatment shortly after PTSD symptoms begin could limit the risk of CVD and, potentially, other disease-related risk.
Source:
Gilsanz P, Winning A, Koenen KC, et al. Psychol Med. 2017;47(8):1370-1378.
doi: 10.1017/S0033291716003378.
Women with severe posttraumatic stress disorder (PTSD) symptoms have a nearly 70% increase in the incidence of cardiovascular disease (CVD), according to a study by researchers from Harvard and Brigham and Women’s Hospital in Boston, Massachusetts, Columbia University in New York, and University of California in San Francisco.
The researchers analyzed data from 49,859 women in the Nurses’ Health Study II. Over 20 years, there were 552 confirmed cases of myocardial infarction or stroke.
Women with 6 to 7 symptoms of trauma and PTSD had the highest risk. Women with trauma but no PTSD symptoms had a 30% higher risk. When women who said illness was their worst trauma were excluded, the risk of CVD doubled among those with trauma and severe PTSD symptoms and increased by 88% in women with trauma and moderate PTSD symptoms.
Strikingly, the researchers also found that when the PTSD symptoms declined so did the CVD risk. The researchers note that CVD risk due to other well-known risk factors, such as smoking, increases with exposure duration declines once the risk factor is eliminated. In this study, for every 5 additional years PTSD symptoms lasted, the odds of CVD were 9% higher.
A “more nuanced understanding” of the role of health behaviors could add insight into how PTSD influences the risk of CVD, the researchers say. They point to studies that have found a link between PTSD and cardiotoxic behaviors such as smoking, drinking, and diet. Physiologic alterations that occur with PTSD symptoms also may play an important role, they suggest, such as changes in neuropeptide Y in response to stress, which might contribute to metabolic syndrome.
Citing “particularly intriguing” findings from a study that found symptoms eventually remitted in 44% of individuals with PTSD, the researchers say providing treatment shortly after PTSD symptoms begin could limit the risk of CVD and, potentially, other disease-related risk.
Source:
Gilsanz P, Winning A, Koenen KC, et al. Psychol Med. 2017;47(8):1370-1378.
doi: 10.1017/S0033291716003378.
Women with severe posttraumatic stress disorder (PTSD) symptoms have a nearly 70% increase in the incidence of cardiovascular disease (CVD), according to a study by researchers from Harvard and Brigham and Women’s Hospital in Boston, Massachusetts, Columbia University in New York, and University of California in San Francisco.
The researchers analyzed data from 49,859 women in the Nurses’ Health Study II. Over 20 years, there were 552 confirmed cases of myocardial infarction or stroke.
Women with 6 to 7 symptoms of trauma and PTSD had the highest risk. Women with trauma but no PTSD symptoms had a 30% higher risk. When women who said illness was their worst trauma were excluded, the risk of CVD doubled among those with trauma and severe PTSD symptoms and increased by 88% in women with trauma and moderate PTSD symptoms.
Strikingly, the researchers also found that when the PTSD symptoms declined so did the CVD risk. The researchers note that CVD risk due to other well-known risk factors, such as smoking, increases with exposure duration declines once the risk factor is eliminated. In this study, for every 5 additional years PTSD symptoms lasted, the odds of CVD were 9% higher.
A “more nuanced understanding” of the role of health behaviors could add insight into how PTSD influences the risk of CVD, the researchers say. They point to studies that have found a link between PTSD and cardiotoxic behaviors such as smoking, drinking, and diet. Physiologic alterations that occur with PTSD symptoms also may play an important role, they suggest, such as changes in neuropeptide Y in response to stress, which might contribute to metabolic syndrome.
Citing “particularly intriguing” findings from a study that found symptoms eventually remitted in 44% of individuals with PTSD, the researchers say providing treatment shortly after PTSD symptoms begin could limit the risk of CVD and, potentially, other disease-related risk.
Source:
Gilsanz P, Winning A, Koenen KC, et al. Psychol Med. 2017;47(8):1370-1378.
doi: 10.1017/S0033291716003378.
Almost one-third of Syrian refugees in U.S. met PTSD criteria
SAN FRANCISCO – Nearly one-third of adult Syrian civil war refugees who have resettled in the Detroit area meet diagnostic criteria for posttraumatic stress disorder, according to preliminary results of an ongoing study presented by Arash Javanbakht, MD, at the annual conference of the Anxiety and Depression Association of America.
That’s comparable to PTSD rates documented in Vietnam War combat veterans.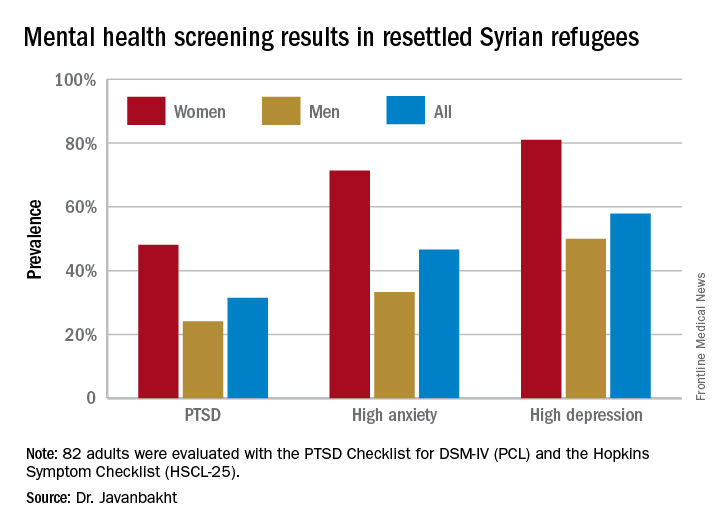
“Based on these data, mental health care for Syrian refugees resettling in the U.S. is highly needed,” he observed.
Michigan has a large population of Syrian war refugees. All who settle in southeastern Michigan undergo an initial health examination at Arab American and Chaldean Council primary care clinics staffed by bilingual teams. More than 90% of eligible adult refugees who have been invited to join Dr. Javanbakht’s mental health screening study have opted to participate. This extraordinarily high recruitment rate suggests that the findings are generalizable to the broader Syrian refugee community throughout the United States in his view.
The screening tools employed in the study are the PTSD Checklist, DSM-IV version (PCL), and the Hopkins Symptom Checklist (HSCL-25), which contains 10 anxiety questions and 15 depression questions.
Of the first 82 adult Syrian refugees evaluated, 26 (31.5%) were diagnosed as having PTSD on the basis of a PCL total score of 40 or more plus fulfillment of the DSM-IV diagnostic criteria for the disorder. In fact, this subgroup had a mean PCL score of 62.3. The prevalence of PTSD was twice as great in women, compared with men (see graphic).
Clinically impactful anxiety symptoms as defined by an HSCL-25 anxiety score greater than 1.79 was present in 38 refugees (47%), and clinically meaningful depressive symptoms were present in 47 (58%).
Anxiety, depression, and PTSD all were tightly correlated, complicating the clinical challenges, the psychiatrist noted.
Enrollment in the study is ongoing, but a first pass examination of a participant population that’s now twice the size of that in his presentation at the conference continues to show similar results, according to Dr. Javanbakht.
Dr. Javanbakht reported having no financial conflicts of interest regarding the study, which was funded by the state of Michigan and the Detroit Wayne Mental Health Authority.
SAN FRANCISCO – Nearly one-third of adult Syrian civil war refugees who have resettled in the Detroit area meet diagnostic criteria for posttraumatic stress disorder, according to preliminary results of an ongoing study presented by Arash Javanbakht, MD, at the annual conference of the Anxiety and Depression Association of America.
That’s comparable to PTSD rates documented in Vietnam War combat veterans.
“Based on these data, mental health care for Syrian refugees resettling in the U.S. is highly needed,” he observed.
Michigan has a large population of Syrian war refugees. All who settle in southeastern Michigan undergo an initial health examination at Arab American and Chaldean Council primary care clinics staffed by bilingual teams. More than 90% of eligible adult refugees who have been invited to join Dr. Javanbakht’s mental health screening study have opted to participate. This extraordinarily high recruitment rate suggests that the findings are generalizable to the broader Syrian refugee community throughout the United States in his view.
The screening tools employed in the study are the PTSD Checklist, DSM-IV version (PCL), and the Hopkins Symptom Checklist (HSCL-25), which contains 10 anxiety questions and 15 depression questions.
Of the first 82 adult Syrian refugees evaluated, 26 (31.5%) were diagnosed as having PTSD on the basis of a PCL total score of 40 or more plus fulfillment of the DSM-IV diagnostic criteria for the disorder. In fact, this subgroup had a mean PCL score of 62.3. The prevalence of PTSD was twice as great in women, compared with men (see graphic).
Clinically impactful anxiety symptoms as defined by an HSCL-25 anxiety score greater than 1.79 was present in 38 refugees (47%), and clinically meaningful depressive symptoms were present in 47 (58%).
Anxiety, depression, and PTSD all were tightly correlated, complicating the clinical challenges, the psychiatrist noted.
Enrollment in the study is ongoing, but a first pass examination of a participant population that’s now twice the size of that in his presentation at the conference continues to show similar results, according to Dr. Javanbakht.
Dr. Javanbakht reported having no financial conflicts of interest regarding the study, which was funded by the state of Michigan and the Detroit Wayne Mental Health Authority.
SAN FRANCISCO – Nearly one-third of adult Syrian civil war refugees who have resettled in the Detroit area meet diagnostic criteria for posttraumatic stress disorder, according to preliminary results of an ongoing study presented by Arash Javanbakht, MD, at the annual conference of the Anxiety and Depression Association of America.
That’s comparable to PTSD rates documented in Vietnam War combat veterans.
“Based on these data, mental health care for Syrian refugees resettling in the U.S. is highly needed,” he observed.
Michigan has a large population of Syrian war refugees. All who settle in southeastern Michigan undergo an initial health examination at Arab American and Chaldean Council primary care clinics staffed by bilingual teams. More than 90% of eligible adult refugees who have been invited to join Dr. Javanbakht’s mental health screening study have opted to participate. This extraordinarily high recruitment rate suggests that the findings are generalizable to the broader Syrian refugee community throughout the United States in his view.
The screening tools employed in the study are the PTSD Checklist, DSM-IV version (PCL), and the Hopkins Symptom Checklist (HSCL-25), which contains 10 anxiety questions and 15 depression questions.
Of the first 82 adult Syrian refugees evaluated, 26 (31.5%) were diagnosed as having PTSD on the basis of a PCL total score of 40 or more plus fulfillment of the DSM-IV diagnostic criteria for the disorder. In fact, this subgroup had a mean PCL score of 62.3. The prevalence of PTSD was twice as great in women, compared with men (see graphic).
Clinically impactful anxiety symptoms as defined by an HSCL-25 anxiety score greater than 1.79 was present in 38 refugees (47%), and clinically meaningful depressive symptoms were present in 47 (58%).
Anxiety, depression, and PTSD all were tightly correlated, complicating the clinical challenges, the psychiatrist noted.
Enrollment in the study is ongoing, but a first pass examination of a participant population that’s now twice the size of that in his presentation at the conference continues to show similar results, according to Dr. Javanbakht.
Dr. Javanbakht reported having no financial conflicts of interest regarding the study, which was funded by the state of Michigan and the Detroit Wayne Mental Health Authority.
AT THE ANXIETY AND DEPRESSION CONFERENCE 2017
Key clinical point:
Major finding: One-third of adult Syrian war refugees who have resettled in Michigan have posttraumatic stress disorder.
Data source: This ongoing cross-sectional study initially screened 82 adult Syrian civil war refugees for PTSD, anxiety, and depression.
Disclosures: This ongoing study is funded by the state of Michigan and the Detroit Wayne Mental Health Authority. The presenter reported having no financial conflicts of interest.