User login
Mindful kids, part 1: Origins and evidence
Open a magazine or turn on the radio and you are likely to hear someone extolling the benefits of mindfulness for any number of purposes, conditions, or age groups. Businesses, schools, and health care organizations are incorporating mindfulness techniques to boost employee, student, and patient well-being and engagement, as well as to help employers, teachers, and providers to thrive. In this two-part series, part 1 will attempt to distill some of the fundamentals with regard to the following questions: 1. What is mindfulness? 2. What is the evidence for mindfulness, particularly in youth? and 3. How would you apply mindfulness techniques in your office setting?
Mindfulness was largely brought into the mainstream health care world by Jon Kabat-Zinn, PhD, of the University of Massachusetts Medical Center, Worcester. Drawing on Buddhist traditions, he created a secularized version of meditative and movement techniques used for thousands of years to promote healthy living. A growing evidence base showed that these practices, combined in a formal curriculum dubbed mindfulness-based stress reduction (MBSR), could alleviate symptoms and distress in conditions as diverse as chronic pain, psoriasis, and anxiety. This has spawned numerous research programs and spin-offs, and remains a foundational approach to utilizing mindfulness in medical care. Dr. Kabat-Zinn’s definition of the term is thus worth noting – mindfulness is “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1 Put simply, mindfulness means having your mind and your body in the same place at the same time. If your mind is wandering to what happened yesterday or planning for what might happen later today, then your mind and body are not in the same time. If your mind is thinking about what is going on at home while you are at work, or what your friends are doing, your mind and body are not in the same place.
A study of a modified version of Dr. Kabat-Zinn’s MBSR in middle schoolers in an inner city environment compared 12 weeks of mindfulness training versus a typical health curriculum discussing adolescence, stress, and puberty. In this inner city environment, students randomized to mindfulness training reported less depression, less hostility, fewer ruminations, and fewer PTSD symptoms as well as fewer physical complaints.10 Regarding clinical populations, mindfulness training in adolescents has shown promise for ADHD, with improvement in both core symptoms and functionality.11 This especially seems pronounced when caregivers are supported in learning mindful parenting techniques alongside their teens’ mindfulness training.12
In a general psychiatry clinic, an 8-week adolescent MBSR program was added to supplement treatment as usual – psychotherapy and medication management. Those randomized to mindfulness showed improvements in sleep and self-esteem, as well as a decline in depressive and anxiety symptoms, perceived stress, and interpersonal problems.13 Perhaps most impressively, half of the MBSR group dropped at least one diagnosis after the 8-week program, whereas none of those in the wait list group, receiving psychiatric specialty care as usual, decreased their diagnosis count.
While the sum of such research in adults and children builds a strong case for the value of mindfulness at both the universal (well-child check) and problem-focused levels, there are limitations to our knowledge base. The number of studies and total number of children and adolescents enrolled in mindfulness research is far fewer than in studies with adults. A variety of mindfulness practices have been incorporated into study interventions such that results are not always comparable and distinguishing the mechanism of action is difficult. Additionally, double-blind and placebo-controlled studies are harder to accomplish with such active interventions, although headway is being made.14
Despite what remains to be discovered, bringing mindfulness into the lives of children and adolescents seems increasingly sensible, given the growing body of scientific support for the benefits of mindfulness practices at the behavioral and functional neuroanatomic levels. As is the case with recommending healthy diets, exercise, and other universal health-promoting behaviors, the knowledge that mindfulness practices are beneficial may not be enough to get patients and their families engaged in these methods. The second article in this series will address some nuts and bolts of prescribing mindfulness in a pediatric health care setting.
Dr. Rosenfeld is an assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center, Robert Larner College of Medicine, Burlington. He said he has no relevant disclosures.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness (New York: Bantam Books, Penguin Random House, 2013).
2. J Pers Soc Psychol. 2003 Apr;84(4):822-48.
3. Gen Hosp Psychiatry. 1982 Apr;4(1):33-47.
4. Am J Psychiatry. 1992 Jul;149(7):936-43.
5. Clin Psychol Rev. 2011 Aug;31(6):1041-56.
6. Neuroreport. 2005 Nov 28;16(17):1893-7.
7. Soc Cogn Affect Neurosci. 2010 Mar;5(1):11-7.
8. Neuroimage. 2009 Apr 15;45(3):672-8.
9. Psychiatry Res. 2011 Jan 30;191(1):36-43.
10. Pediatrics. 2016 Jan;137(1):e20152532.
11. J Atten Disord. 2008 May;11(6):737-46.
12. J Child Fam Stud. 2012 Oct;21(5):775-87.
13. J Consult Clin Psychol. 2009 Oct;77(5):855-66.
14. Biol Psychiatry. 2016 Jul 1;80(1):53-61.
Open a magazine or turn on the radio and you are likely to hear someone extolling the benefits of mindfulness for any number of purposes, conditions, or age groups. Businesses, schools, and health care organizations are incorporating mindfulness techniques to boost employee, student, and patient well-being and engagement, as well as to help employers, teachers, and providers to thrive. In this two-part series, part 1 will attempt to distill some of the fundamentals with regard to the following questions: 1. What is mindfulness? 2. What is the evidence for mindfulness, particularly in youth? and 3. How would you apply mindfulness techniques in your office setting?
Mindfulness was largely brought into the mainstream health care world by Jon Kabat-Zinn, PhD, of the University of Massachusetts Medical Center, Worcester. Drawing on Buddhist traditions, he created a secularized version of meditative and movement techniques used for thousands of years to promote healthy living. A growing evidence base showed that these practices, combined in a formal curriculum dubbed mindfulness-based stress reduction (MBSR), could alleviate symptoms and distress in conditions as diverse as chronic pain, psoriasis, and anxiety. This has spawned numerous research programs and spin-offs, and remains a foundational approach to utilizing mindfulness in medical care. Dr. Kabat-Zinn’s definition of the term is thus worth noting – mindfulness is “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1 Put simply, mindfulness means having your mind and your body in the same place at the same time. If your mind is wandering to what happened yesterday or planning for what might happen later today, then your mind and body are not in the same time. If your mind is thinking about what is going on at home while you are at work, or what your friends are doing, your mind and body are not in the same place.
A study of a modified version of Dr. Kabat-Zinn’s MBSR in middle schoolers in an inner city environment compared 12 weeks of mindfulness training versus a typical health curriculum discussing adolescence, stress, and puberty. In this inner city environment, students randomized to mindfulness training reported less depression, less hostility, fewer ruminations, and fewer PTSD symptoms as well as fewer physical complaints.10 Regarding clinical populations, mindfulness training in adolescents has shown promise for ADHD, with improvement in both core symptoms and functionality.11 This especially seems pronounced when caregivers are supported in learning mindful parenting techniques alongside their teens’ mindfulness training.12
In a general psychiatry clinic, an 8-week adolescent MBSR program was added to supplement treatment as usual – psychotherapy and medication management. Those randomized to mindfulness showed improvements in sleep and self-esteem, as well as a decline in depressive and anxiety symptoms, perceived stress, and interpersonal problems.13 Perhaps most impressively, half of the MBSR group dropped at least one diagnosis after the 8-week program, whereas none of those in the wait list group, receiving psychiatric specialty care as usual, decreased their diagnosis count.
While the sum of such research in adults and children builds a strong case for the value of mindfulness at both the universal (well-child check) and problem-focused levels, there are limitations to our knowledge base. The number of studies and total number of children and adolescents enrolled in mindfulness research is far fewer than in studies with adults. A variety of mindfulness practices have been incorporated into study interventions such that results are not always comparable and distinguishing the mechanism of action is difficult. Additionally, double-blind and placebo-controlled studies are harder to accomplish with such active interventions, although headway is being made.14
Despite what remains to be discovered, bringing mindfulness into the lives of children and adolescents seems increasingly sensible, given the growing body of scientific support for the benefits of mindfulness practices at the behavioral and functional neuroanatomic levels. As is the case with recommending healthy diets, exercise, and other universal health-promoting behaviors, the knowledge that mindfulness practices are beneficial may not be enough to get patients and their families engaged in these methods. The second article in this series will address some nuts and bolts of prescribing mindfulness in a pediatric health care setting.
Dr. Rosenfeld is an assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center, Robert Larner College of Medicine, Burlington. He said he has no relevant disclosures.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness (New York: Bantam Books, Penguin Random House, 2013).
2. J Pers Soc Psychol. 2003 Apr;84(4):822-48.
3. Gen Hosp Psychiatry. 1982 Apr;4(1):33-47.
4. Am J Psychiatry. 1992 Jul;149(7):936-43.
5. Clin Psychol Rev. 2011 Aug;31(6):1041-56.
6. Neuroreport. 2005 Nov 28;16(17):1893-7.
7. Soc Cogn Affect Neurosci. 2010 Mar;5(1):11-7.
8. Neuroimage. 2009 Apr 15;45(3):672-8.
9. Psychiatry Res. 2011 Jan 30;191(1):36-43.
10. Pediatrics. 2016 Jan;137(1):e20152532.
11. J Atten Disord. 2008 May;11(6):737-46.
12. J Child Fam Stud. 2012 Oct;21(5):775-87.
13. J Consult Clin Psychol. 2009 Oct;77(5):855-66.
14. Biol Psychiatry. 2016 Jul 1;80(1):53-61.
Open a magazine or turn on the radio and you are likely to hear someone extolling the benefits of mindfulness for any number of purposes, conditions, or age groups. Businesses, schools, and health care organizations are incorporating mindfulness techniques to boost employee, student, and patient well-being and engagement, as well as to help employers, teachers, and providers to thrive. In this two-part series, part 1 will attempt to distill some of the fundamentals with regard to the following questions: 1. What is mindfulness? 2. What is the evidence for mindfulness, particularly in youth? and 3. How would you apply mindfulness techniques in your office setting?
Mindfulness was largely brought into the mainstream health care world by Jon Kabat-Zinn, PhD, of the University of Massachusetts Medical Center, Worcester. Drawing on Buddhist traditions, he created a secularized version of meditative and movement techniques used for thousands of years to promote healthy living. A growing evidence base showed that these practices, combined in a formal curriculum dubbed mindfulness-based stress reduction (MBSR), could alleviate symptoms and distress in conditions as diverse as chronic pain, psoriasis, and anxiety. This has spawned numerous research programs and spin-offs, and remains a foundational approach to utilizing mindfulness in medical care. Dr. Kabat-Zinn’s definition of the term is thus worth noting – mindfulness is “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1 Put simply, mindfulness means having your mind and your body in the same place at the same time. If your mind is wandering to what happened yesterday or planning for what might happen later today, then your mind and body are not in the same time. If your mind is thinking about what is going on at home while you are at work, or what your friends are doing, your mind and body are not in the same place.
A study of a modified version of Dr. Kabat-Zinn’s MBSR in middle schoolers in an inner city environment compared 12 weeks of mindfulness training versus a typical health curriculum discussing adolescence, stress, and puberty. In this inner city environment, students randomized to mindfulness training reported less depression, less hostility, fewer ruminations, and fewer PTSD symptoms as well as fewer physical complaints.10 Regarding clinical populations, mindfulness training in adolescents has shown promise for ADHD, with improvement in both core symptoms and functionality.11 This especially seems pronounced when caregivers are supported in learning mindful parenting techniques alongside their teens’ mindfulness training.12
In a general psychiatry clinic, an 8-week adolescent MBSR program was added to supplement treatment as usual – psychotherapy and medication management. Those randomized to mindfulness showed improvements in sleep and self-esteem, as well as a decline in depressive and anxiety symptoms, perceived stress, and interpersonal problems.13 Perhaps most impressively, half of the MBSR group dropped at least one diagnosis after the 8-week program, whereas none of those in the wait list group, receiving psychiatric specialty care as usual, decreased their diagnosis count.
While the sum of such research in adults and children builds a strong case for the value of mindfulness at both the universal (well-child check) and problem-focused levels, there are limitations to our knowledge base. The number of studies and total number of children and adolescents enrolled in mindfulness research is far fewer than in studies with adults. A variety of mindfulness practices have been incorporated into study interventions such that results are not always comparable and distinguishing the mechanism of action is difficult. Additionally, double-blind and placebo-controlled studies are harder to accomplish with such active interventions, although headway is being made.14
Despite what remains to be discovered, bringing mindfulness into the lives of children and adolescents seems increasingly sensible, given the growing body of scientific support for the benefits of mindfulness practices at the behavioral and functional neuroanatomic levels. As is the case with recommending healthy diets, exercise, and other universal health-promoting behaviors, the knowledge that mindfulness practices are beneficial may not be enough to get patients and their families engaged in these methods. The second article in this series will address some nuts and bolts of prescribing mindfulness in a pediatric health care setting.
Dr. Rosenfeld is an assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center, Robert Larner College of Medicine, Burlington. He said he has no relevant disclosures.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness (New York: Bantam Books, Penguin Random House, 2013).
2. J Pers Soc Psychol. 2003 Apr;84(4):822-48.
3. Gen Hosp Psychiatry. 1982 Apr;4(1):33-47.
4. Am J Psychiatry. 1992 Jul;149(7):936-43.
5. Clin Psychol Rev. 2011 Aug;31(6):1041-56.
6. Neuroreport. 2005 Nov 28;16(17):1893-7.
7. Soc Cogn Affect Neurosci. 2010 Mar;5(1):11-7.
8. Neuroimage. 2009 Apr 15;45(3):672-8.
9. Psychiatry Res. 2011 Jan 30;191(1):36-43.
10. Pediatrics. 2016 Jan;137(1):e20152532.
11. J Atten Disord. 2008 May;11(6):737-46.
12. J Child Fam Stud. 2012 Oct;21(5):775-87.
13. J Consult Clin Psychol. 2009 Oct;77(5):855-66.
14. Biol Psychiatry. 2016 Jul 1;80(1):53-61.
Mindful kids, part 2: Integration into practice
In this follow-up to last month’s column on mindfulness, in which the evidence base makes a compelling argument for incorporating mindfulness into our list of healthy practices for youth brain development, the challenge of implementing mindfulness “prescriptions” in practice is considered in more depth. As a reminder, a working definition of mindfulness was offered as, “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1
An important piece of prescribing, either pharmaceuticals or health-promoting practices, is sharing the risks, benefits, and alternatives to the recommended treatment. Last month’s article considered the potential benefits of cultivating a mindfulness practice. Few risks have been well-documented, particularly in the pediatric population. While some case reports describe adults having profoundly disturbing emotional reactions,these are in the context of intensive meditation experiences (think 10-day silent retreat).2 While there is not evidence of harm in youth, the lesson to be learned from adult experiences may be to consult with an advanced teacher if a patient chooses to become intensely involved in any meditative practice.
Bringing mindfulness practices to your office practice could occur anywhere along the spectrum from integrating some mindfulness moments into your standard physical exam to collaborating with an experienced mindfulness or yoga instructor to offer individual and group support to patients and families. My focus here is on simple practices and tools to begin introducing mindfulness to families.
A key component is clinician and caregiver buy-in. Developing your own practice, even if it’s simply three mindful breaths before entering each patient exam room, goes miles in terms of your being able to speak genuinely about the benefits and challenges of mindfulness in a relatable way. Similarly, the more kids see their families practicing and supporting mindfulness, the more likely they are to develop their own routines.
Legitimizing mindfulness practices with a “prescription” also can add to success rates. Considering diaphragmatic breathing as a foundational technique, the following prescription can be printed on cards and reviewed briefly in a visit:
- Show me how you breathe. Now let’s practice belly (abdominal/diaphragmatic) breathing.
- Move both hands to your belly. Imagine you are breathing behind your belly button. Feel your belly rise like a balloon.
- As you breathe out, feel your belly drop as you let air out.
- Bonus: Now breathe through your nose only as you continue belly breathing. Next, notice your belly rising and falling without placing your hands on it.
In a physical exam, the following might work: When you place your stethoscope on the chest and back to auscultate the lungs, instruct the child to “place a hand on your belly and take a deep breath into your belly button so that your hand moves out. Keep taking slow, deep belly breaths while I listen.”
This breathing technique activates the parasympathetic nervous system, quelling the fight-or-flight response that may contribute to anxiety, aggressive reactivity, and interfere with sleep. Prescribing five of these belly breaths before bedtime is a good beginning, increasing frequency and duration over time as the practice becomes routine, then adding the “bonus” techniques. Introducing abdominal breathing also makes a good opportunity to ask the child about sources of stress in their lives.
For the distracted or stressed-out youth, focus is key. Those children who seem to be always multitasking or never sit still may benefit from cultivating a focus practice. It also may help still the mind before bedtime. A mindfulness prescription for focus is as follows:
- The rays of the sun are much more powerful when they are brought into focus. Just like building a muscle, focus can be built up to be stronger. Let’s practice focusing.
- As you breathe in, count slowly to 5, raising one finger for each count. As you breathe out, count down to 0, lowering each finger.
- Notice when you get distracted during the counting. Exercise your focus by coming back to counting your breath.
- Let your hands rest in your lap. Then, move to counting silently in your head.
Alternative options for focus objects include watching the secondhand on a clock, balancing a peacock feather on a fingertip, listening to a bell or chime until it can no longer be heard, watching a sand timer until every grain falls.
In a physical exam, the following might work: During the neurologic exam for cranial nerves (eye movements), direct the child to focus on your finger. Hold it still for 10 seconds, gently reminding them to keep their focus on your finger if needed. Then, as you move to each quadrant, move slowly and stay in each quadrant for 5 seconds. Encourage them to “keep your focus on my finger.”
After practicing a focus exercise, inquire about the patient’s focus during school, homework, and activities. Suggest making the focused breathing, or an alternative focus activity, part of the daily routine. Parents are encouraged to participate alongside their children.
Depending on the amount of time you have in the visit, your mindfulness intervention may simply be how you conduct the physical exam. With more time or a child or family who seems to have an indication for prescribing mindfulness (stress, anxiety, inattention, insomnia, etc.), a more didactic approach toward mindfulness techniques accompanied by a specific prescription may be in order. Developmentally, clinicians in our practice have found that hands-on activities and games can help involve younger children, while teens can get into one of the apps developed to facilitate mindful practices. (See Online resources.) Diagnostically, more hyperactive or distractible children may mesh better with movement-based practices. Depressed or anxious children may enjoy quieter activities or benefit from small incentives to increase motivation. Children with traumatic histories may benefit from a slow pace, keeping their eyes open and looking at the floor rather than eyes closed and avoiding physical contact initially.
Methods of meditation and mindfulness exist in most every philosophical and religious tradition, but the neuroscientific value of these practices is a more recent take on these wisdom traditions. As we follow the growing research literature on mindfulness, consider incorporating this “new” prescription into your toolbox of healthy practices for the developing brain.
Dr. Rosenfeld is assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center and the university’s Robert Larner College of Medicine, Burlington. He reported no relevant disclosures. Email him at pdnews@frontlinemedcom.com.
Online resources:
- A Sesame Street video on belly breathing (for younger children): “Belly Breathe” with Elmo at www.youtube.com/watch?v=_mZbzDOpylA.
- Card decks: Growing Mindful: Mindfulness Practices for All Ages; Be Mindful Card Deck for Teens; Yoga 4 Classrooms Activity Card Deck.
- Apps: Smiling Mind (differentiated by age); Calm; Breathe; Breathe2Relax; Insight Timer; Grow (mindfulness for teens).
- Props: peacock feathers; sand timers; clock with secondhand; Tibetan singing bell or other; Hoberman spheres (“breathing balls”) to visualize belly breathing.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness. (New York: Bantam Books, Penguin Random House, 2013).
2. Rocha, Tomas. “The Dark Knight of the Soul.” The Atlantic. June 25, 2014.
In this follow-up to last month’s column on mindfulness, in which the evidence base makes a compelling argument for incorporating mindfulness into our list of healthy practices for youth brain development, the challenge of implementing mindfulness “prescriptions” in practice is considered in more depth. As a reminder, a working definition of mindfulness was offered as, “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1
An important piece of prescribing, either pharmaceuticals or health-promoting practices, is sharing the risks, benefits, and alternatives to the recommended treatment. Last month’s article considered the potential benefits of cultivating a mindfulness practice. Few risks have been well-documented, particularly in the pediatric population. While some case reports describe adults having profoundly disturbing emotional reactions,these are in the context of intensive meditation experiences (think 10-day silent retreat).2 While there is not evidence of harm in youth, the lesson to be learned from adult experiences may be to consult with an advanced teacher if a patient chooses to become intensely involved in any meditative practice.
Bringing mindfulness practices to your office practice could occur anywhere along the spectrum from integrating some mindfulness moments into your standard physical exam to collaborating with an experienced mindfulness or yoga instructor to offer individual and group support to patients and families. My focus here is on simple practices and tools to begin introducing mindfulness to families.
A key component is clinician and caregiver buy-in. Developing your own practice, even if it’s simply three mindful breaths before entering each patient exam room, goes miles in terms of your being able to speak genuinely about the benefits and challenges of mindfulness in a relatable way. Similarly, the more kids see their families practicing and supporting mindfulness, the more likely they are to develop their own routines.
Legitimizing mindfulness practices with a “prescription” also can add to success rates. Considering diaphragmatic breathing as a foundational technique, the following prescription can be printed on cards and reviewed briefly in a visit:
- Show me how you breathe. Now let’s practice belly (abdominal/diaphragmatic) breathing.
- Move both hands to your belly. Imagine you are breathing behind your belly button. Feel your belly rise like a balloon.
- As you breathe out, feel your belly drop as you let air out.
- Bonus: Now breathe through your nose only as you continue belly breathing. Next, notice your belly rising and falling without placing your hands on it.
In a physical exam, the following might work: When you place your stethoscope on the chest and back to auscultate the lungs, instruct the child to “place a hand on your belly and take a deep breath into your belly button so that your hand moves out. Keep taking slow, deep belly breaths while I listen.”
This breathing technique activates the parasympathetic nervous system, quelling the fight-or-flight response that may contribute to anxiety, aggressive reactivity, and interfere with sleep. Prescribing five of these belly breaths before bedtime is a good beginning, increasing frequency and duration over time as the practice becomes routine, then adding the “bonus” techniques. Introducing abdominal breathing also makes a good opportunity to ask the child about sources of stress in their lives.
For the distracted or stressed-out youth, focus is key. Those children who seem to be always multitasking or never sit still may benefit from cultivating a focus practice. It also may help still the mind before bedtime. A mindfulness prescription for focus is as follows:
- The rays of the sun are much more powerful when they are brought into focus. Just like building a muscle, focus can be built up to be stronger. Let’s practice focusing.
- As you breathe in, count slowly to 5, raising one finger for each count. As you breathe out, count down to 0, lowering each finger.
- Notice when you get distracted during the counting. Exercise your focus by coming back to counting your breath.
- Let your hands rest in your lap. Then, move to counting silently in your head.
Alternative options for focus objects include watching the secondhand on a clock, balancing a peacock feather on a fingertip, listening to a bell or chime until it can no longer be heard, watching a sand timer until every grain falls.
In a physical exam, the following might work: During the neurologic exam for cranial nerves (eye movements), direct the child to focus on your finger. Hold it still for 10 seconds, gently reminding them to keep their focus on your finger if needed. Then, as you move to each quadrant, move slowly and stay in each quadrant for 5 seconds. Encourage them to “keep your focus on my finger.”
After practicing a focus exercise, inquire about the patient’s focus during school, homework, and activities. Suggest making the focused breathing, or an alternative focus activity, part of the daily routine. Parents are encouraged to participate alongside their children.
Depending on the amount of time you have in the visit, your mindfulness intervention may simply be how you conduct the physical exam. With more time or a child or family who seems to have an indication for prescribing mindfulness (stress, anxiety, inattention, insomnia, etc.), a more didactic approach toward mindfulness techniques accompanied by a specific prescription may be in order. Developmentally, clinicians in our practice have found that hands-on activities and games can help involve younger children, while teens can get into one of the apps developed to facilitate mindful practices. (See Online resources.) Diagnostically, more hyperactive or distractible children may mesh better with movement-based practices. Depressed or anxious children may enjoy quieter activities or benefit from small incentives to increase motivation. Children with traumatic histories may benefit from a slow pace, keeping their eyes open and looking at the floor rather than eyes closed and avoiding physical contact initially.
Methods of meditation and mindfulness exist in most every philosophical and religious tradition, but the neuroscientific value of these practices is a more recent take on these wisdom traditions. As we follow the growing research literature on mindfulness, consider incorporating this “new” prescription into your toolbox of healthy practices for the developing brain.
Dr. Rosenfeld is assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center and the university’s Robert Larner College of Medicine, Burlington. He reported no relevant disclosures. Email him at pdnews@frontlinemedcom.com.
Online resources:
- A Sesame Street video on belly breathing (for younger children): “Belly Breathe” with Elmo at www.youtube.com/watch?v=_mZbzDOpylA.
- Card decks: Growing Mindful: Mindfulness Practices for All Ages; Be Mindful Card Deck for Teens; Yoga 4 Classrooms Activity Card Deck.
- Apps: Smiling Mind (differentiated by age); Calm; Breathe; Breathe2Relax; Insight Timer; Grow (mindfulness for teens).
- Props: peacock feathers; sand timers; clock with secondhand; Tibetan singing bell or other; Hoberman spheres (“breathing balls”) to visualize belly breathing.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness. (New York: Bantam Books, Penguin Random House, 2013).
2. Rocha, Tomas. “The Dark Knight of the Soul.” The Atlantic. June 25, 2014.
In this follow-up to last month’s column on mindfulness, in which the evidence base makes a compelling argument for incorporating mindfulness into our list of healthy practices for youth brain development, the challenge of implementing mindfulness “prescriptions” in practice is considered in more depth. As a reminder, a working definition of mindfulness was offered as, “the awareness that arises by paying attention on purpose, in the present moment, and nonjudgmentally.”1
An important piece of prescribing, either pharmaceuticals or health-promoting practices, is sharing the risks, benefits, and alternatives to the recommended treatment. Last month’s article considered the potential benefits of cultivating a mindfulness practice. Few risks have been well-documented, particularly in the pediatric population. While some case reports describe adults having profoundly disturbing emotional reactions,these are in the context of intensive meditation experiences (think 10-day silent retreat).2 While there is not evidence of harm in youth, the lesson to be learned from adult experiences may be to consult with an advanced teacher if a patient chooses to become intensely involved in any meditative practice.
Bringing mindfulness practices to your office practice could occur anywhere along the spectrum from integrating some mindfulness moments into your standard physical exam to collaborating with an experienced mindfulness or yoga instructor to offer individual and group support to patients and families. My focus here is on simple practices and tools to begin introducing mindfulness to families.
A key component is clinician and caregiver buy-in. Developing your own practice, even if it’s simply three mindful breaths before entering each patient exam room, goes miles in terms of your being able to speak genuinely about the benefits and challenges of mindfulness in a relatable way. Similarly, the more kids see their families practicing and supporting mindfulness, the more likely they are to develop their own routines.
Legitimizing mindfulness practices with a “prescription” also can add to success rates. Considering diaphragmatic breathing as a foundational technique, the following prescription can be printed on cards and reviewed briefly in a visit:
- Show me how you breathe. Now let’s practice belly (abdominal/diaphragmatic) breathing.
- Move both hands to your belly. Imagine you are breathing behind your belly button. Feel your belly rise like a balloon.
- As you breathe out, feel your belly drop as you let air out.
- Bonus: Now breathe through your nose only as you continue belly breathing. Next, notice your belly rising and falling without placing your hands on it.
In a physical exam, the following might work: When you place your stethoscope on the chest and back to auscultate the lungs, instruct the child to “place a hand on your belly and take a deep breath into your belly button so that your hand moves out. Keep taking slow, deep belly breaths while I listen.”
This breathing technique activates the parasympathetic nervous system, quelling the fight-or-flight response that may contribute to anxiety, aggressive reactivity, and interfere with sleep. Prescribing five of these belly breaths before bedtime is a good beginning, increasing frequency and duration over time as the practice becomes routine, then adding the “bonus” techniques. Introducing abdominal breathing also makes a good opportunity to ask the child about sources of stress in their lives.
For the distracted or stressed-out youth, focus is key. Those children who seem to be always multitasking or never sit still may benefit from cultivating a focus practice. It also may help still the mind before bedtime. A mindfulness prescription for focus is as follows:
- The rays of the sun are much more powerful when they are brought into focus. Just like building a muscle, focus can be built up to be stronger. Let’s practice focusing.
- As you breathe in, count slowly to 5, raising one finger for each count. As you breathe out, count down to 0, lowering each finger.
- Notice when you get distracted during the counting. Exercise your focus by coming back to counting your breath.
- Let your hands rest in your lap. Then, move to counting silently in your head.
Alternative options for focus objects include watching the secondhand on a clock, balancing a peacock feather on a fingertip, listening to a bell or chime until it can no longer be heard, watching a sand timer until every grain falls.
In a physical exam, the following might work: During the neurologic exam for cranial nerves (eye movements), direct the child to focus on your finger. Hold it still for 10 seconds, gently reminding them to keep their focus on your finger if needed. Then, as you move to each quadrant, move slowly and stay in each quadrant for 5 seconds. Encourage them to “keep your focus on my finger.”
After practicing a focus exercise, inquire about the patient’s focus during school, homework, and activities. Suggest making the focused breathing, or an alternative focus activity, part of the daily routine. Parents are encouraged to participate alongside their children.
Depending on the amount of time you have in the visit, your mindfulness intervention may simply be how you conduct the physical exam. With more time or a child or family who seems to have an indication for prescribing mindfulness (stress, anxiety, inattention, insomnia, etc.), a more didactic approach toward mindfulness techniques accompanied by a specific prescription may be in order. Developmentally, clinicians in our practice have found that hands-on activities and games can help involve younger children, while teens can get into one of the apps developed to facilitate mindful practices. (See Online resources.) Diagnostically, more hyperactive or distractible children may mesh better with movement-based practices. Depressed or anxious children may enjoy quieter activities or benefit from small incentives to increase motivation. Children with traumatic histories may benefit from a slow pace, keeping their eyes open and looking at the floor rather than eyes closed and avoiding physical contact initially.
Methods of meditation and mindfulness exist in most every philosophical and religious tradition, but the neuroscientific value of these practices is a more recent take on these wisdom traditions. As we follow the growing research literature on mindfulness, consider incorporating this “new” prescription into your toolbox of healthy practices for the developing brain.
Dr. Rosenfeld is assistant professor in the departments of psychiatry and pediatrics at the University of Vermont Medical Center and the university’s Robert Larner College of Medicine, Burlington. He reported no relevant disclosures. Email him at pdnews@frontlinemedcom.com.
Online resources:
- A Sesame Street video on belly breathing (for younger children): “Belly Breathe” with Elmo at www.youtube.com/watch?v=_mZbzDOpylA.
- Card decks: Growing Mindful: Mindfulness Practices for All Ages; Be Mindful Card Deck for Teens; Yoga 4 Classrooms Activity Card Deck.
- Apps: Smiling Mind (differentiated by age); Calm; Breathe; Breathe2Relax; Insight Timer; Grow (mindfulness for teens).
- Props: peacock feathers; sand timers; clock with secondhand; Tibetan singing bell or other; Hoberman spheres (“breathing balls”) to visualize belly breathing.
References
1. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness. (New York: Bantam Books, Penguin Random House, 2013).
2. Rocha, Tomas. “The Dark Knight of the Soul.” The Atlantic. June 25, 2014.
A veteran who is suicidal while sleeping
CASE Suicidal while asleep
Mr. R, age 28, an Iraq and Afghanistan veteran with major depressive disorder and posttraumatic stress disorder (PTSD), is awoken by his wife to check on their daughter approximately 30 minutes after he takes his nightly regimen of zolpidem, 10 mg, melatonin, 6 mg, and hydroxyzine, 20 mg. When Mr. R returns to the bedroom, he appears to be confused. Mr. R grabs an unloaded gun from under the mattress, puts it in his mouth, and pulls the trigger. Then Mr. R holds the gun to his head and pulls the trigger while saying that his wife and children will be better off without him. His wife takes the gun away, but he grabs another gun from his gun box and loads it. His wife convinces him to remove the ammunition; however, Mr. R gets the other unloaded gun and pulls the trigger on himself again. After his wife takes this gun away, he tries cutting himself with a pocketknife, causing superficial cuts. Eventually, Mr. R goes back to bed. He does not remember these events in the morning.
What increased the likelihood of parasomnia in Mr. R?
a) high zolpidem dosage
b) concomitant use of other sedating agents
c) sleep deprivation
d) dehydration
[polldaddy:9712545]
The authors’ observations
Parasomnias are sleep-wake transition disorders classified by the sleep stage from which they arise, either NREM or rapid eye movement (REM). NREM parasomnias could result from incomplete awakening from NREM sleep, typically in Stage N3 (slow-wave) sleep.1 DSM-5 describes NREM parasomnias as arousal disorders in which the disturbance is not attributable to the physiological effects of substance; substance/medication-induced sleep disorder, parasomnia type, is when the disturbance can be attributed to a substance.2 The latter also can occur during REM sleep.
NREM parasomnias are characterized by abnormal behaviors during sleep with significant harm potential.3 Somnambulism or sleepwalking and sleep terrors are the 2 types of NREM parasomnias in DSM-5. Sleepwalking could involve complex behaviors, including:
- eating
- talking
- cooking
- shopping
- driving
- sexual activity.
Zolpidem, a benzodiazepine receptor agonist, is a preferred hypnotic agent for insomnia because of its low risk for abuse and daytime sedation.4 However, the drug has been associated with NREM parasomnias, namely somnambulism or sleepwalking, and its variants including sleep-driving, sleep-related eating disorder, and rarely sexsomnia (sleep-sex), with anterograde amnesia for the event.5 Suicidal behavior that occurs while the patient is asleep with next-day amnesia is another variant of somnambulism. There are several reports of suicidal behavior during sleep,6,7 but to our knowledge, there are only 2 previous cases implicating zolpidem as the cause:
- Gibson et al8 described a 49-year-old man who sustained a self-inflicted gunshot wound to his head while asleep. He just had started taking zolpidem, and in the weeks before the incident he had several episodes of sleepwalking and sleep-eating. He had consumed alcohol the night of the self-inflicted gunshot wound, but had no other psychiatric history.
- Chopra et al4 described a 37-year-old man, with no prior episodes of sleepwalking or associated complex behaviors, who was taking zolpidem, 10 mg/d, for chronic insomnia. He shot a gun in the basement of his home, and then held the loaded gun to his neck while asleep. The authors attributed the event to zolpidem in combination with other predisposing factors, including dehydration after intense exercise and alcohol use. The authors categorized this type of event as “para-suicidal amnestic behavior,” although “sleep-related pseudo-suicidal behavior” might be a better term for this type of parasomnia because of its occurrence during sleep and non-deliberate nature.
In another case report, a 27-year-old man took additional zolpidem after he did not experience desired sedative effects from an initial 20 mg.9 Because the patient remembered the suicidal thoughts, the authors believed that the patient attempted suicide while under the influence of zolpidem. The authors did not believe the incident to be sleep-related suicidal behavior, because it was uncertain if he attempted suicide while asleep.
Mr. R does not remember the events his wife witnessed while he was asleep. To our knowledge, Mr. R’s case is the first sleep-related pseudo-suicidal behavior case resulting from zolpidem, 10 mg/d, without concurrent alcohol use in an adult male veteran with PTSD and no suicidal ideation while awake.
HISTORY Further details revealed
Mr. R says that in the days leading to the incident he was not sleep-deprived and was getting at least 6 hours of restful sleep every night. He had been taking zolpidem every night. He has no childhood or family history of NREM parasomnias. He says he did not engage in intense exercise that evening or have a fever the night of the incident and has abstained from alcohol for 2 years.
His wife says that after he took zolpidem, when he was woken up, “He was not there; his eyes were glazed and glossy, and it’s like he was in another world,” and his speech and behavior were bizarre. She also reports that his eyes were open when he engaged in this behavior that appeared suicidal.
Three months before the incident, Mr. R had reported nightmares with dream enactment behaviors, hypervigilance on awakening and during the daytime, irritability, and anxious and depressed mood with neurovegetative symptoms, and was referred to our clinic for medication management. He also reported no prior or current manic or psychotic symptoms, denied suicidal thoughts, and had no history of suicide attempts. Mr. R’s medication regimen included tramadol, 400 mg/d, for chronic knee pain; fluoxetine, 60 mg/d, for depression and PTSD; and propranolol ER, 60 mg/d, and propranolol, 10 mg/d as needed, for anxiety. He was started on prazosin, 2 mg/d, titrated to 4 mg/d, for medication management of nightmares.
Mr. R also was referred to the sleep laboratory for a polysomnogram (PSG) because of reported loud snoring and witnessed apneas, especially because sleep apnea can cause nightmares and dream enactment behaviors. The PSG was negative for sleep apnea or excessive periodic limb movements of sleep, but showed increased electromyographic (EMG) activity during REM sleep, which was consistent with his report of dream enactment behaviors. Two months later, he reported improvement in nightmares and depression, but not in dream enactment behaviors. Because of prominent anxiety and irritability, he was started on gabapentin, 300 mg, 3 times a day.
What factor increases the risk of NREM parasomnias with zolpidem compared with benzodiazepines?
a) greater preservation of Stage N3 sleep
b) lesser degree of muscle relaxation
c) both a and b
d) none of the above
[polldaddy:9712556]
The authors’ observations
Factors that increase the likelihood of parasomnias include:
- zolpidem >10 mg at bedtime
- concomitant use of other CNS depressants, including sedative hypnotic agents and alcohol
- female sex
- not falling asleep immediately after taking zolpidem
- personal or family history of parasomnias
- living alone
- poor pill management
- presence of sleep disruptors such as sleep apnea and periodic limb movements of sleep.1,4,5,10
Higher dosages of zolpidem (>10 mg/d) have been identified as the predictive risk factor.5 In the Chopra et al4 case report on sleep-related suicidal behavior related to zolpidem, 10 mg at bedtime, concomitant dehydration and alcohol use were implicated as facilitating factors. Dehydration could increase serum levels of zolpidem resulting in greater CNS effects. Alcohol use was implicated in the Gibson et al8 case report as well, and the patient had multiple episodes of sleepwalking and sleep-related eating.However, Mr. R was not dehydrated or using alcohol.
An interesting feature of Mr. R’s case is that he was taking fluoxetine. Cytochrome P450 (CYP) 3A4 is involved in metabolizing zolpidem, and norfluoxetine, a metabolite of fluoxetine, inhibits CYP3A4. Although studies have not found pharmacokinetic interactions between fluoxetine and zolpidem, these studies did not investigate fluoxetine dosages >20 mg/d.11 The inhibition of CYP enzymes by fluoxetine likely is dose-dependent,12 and therefore concomitant administration of high-dosage fluoxetine (>20 mg/d) with zolpidem might result in higher serum levels of zolpidem.
Mr. R also was taking several sedating agents (gabapentin, hydroxyzine, melatonin, and tramadol). The concomitant use of these sedative-hypnotic agents could have increased his risk of parasomnia. A review of the literature did not reveal any reports of gabapentin, hydroxyzine, melatonin, or tramadol causing parasomnias. This observation, as well as the well-known role of zolpidem5 in etiopathogenesis of parasomnias, indicates that the pseudo-suicidal behavior Mr. R displayed while asleep likely was a direct result of zolpidem use in presence of other facilitating factors. Gabapentin, which is known to increase the depth of sleep, was added to his regimen 1 month before his parasomnia episode. Therefore, gabapentin could have triggered parasomnia with zolpidem therapy.1,13
Conditions that provoke repeated cortical arousals (eg, periodic limb movement disorder [PLMD] and sleep apnea) or increase depth or pressure of sleep (intense exercise in the evening, fever, sleep deprivation) are thought to be associated with NREM parasomnias.1-4 However, Mr. R underwent in-laboratory PSG and tested negative for major cortical arousal-inducing conditions, such as PLMD and sleep apnea.
Some other sleep disruptors likely were involved in Mr. R’s case. Auditory and tactile stimuli are known to cause cortical arousals, with additive effect seen when these 2 stimuli are combined.3,14 Additionally, these exogenous stimuli are known to trigger sleep-related violent parasomnias.15 Mr. R displayed this behavior after his wife woke him up. The auditory stimulus of his wife’s voice and/or tactile stimulus involved in the act of waking Mr. R likely played a role in the suicidal and violent nature of his NREM parasomnia.
[polldaddy:9712581]
The authors’ observations
In general, the mechanisms by which zolpidem causes NREM parasomnias are not completely understood. The sedation-related amnestic properties of zolpidem might explain some of these behaviors. Patients could perform these behaviors after waking and have subsequent amnesia.4 There is greater preservation of Stage N3 sleep with zolpidem compared with benzodiazepines. Benzodiazepines also cause muscle relaxation while the motor system remains relatively more active during sleep with zolpidem because of its selectivity for α-1 subunit of gamma-aminobutyric acid A receptor. These factors might increase the likelihood of NREM parasomnias with zolpidem compared with benzodiazepines.4
Types of parasomnias
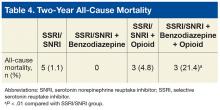
According to DSM-5, there are 2 categories of parasomnias based on the sleep stage from which a parasomnia emerges.2 REM sleep behavior disorder (RBD) refers to complex motor and/or vocalizations during REM sleep, accompanied by increased EMG activity during REM sleep (Table).2,3

The pseudo-suicidal behavior Mr. R displayed likely was NREM parasomnia because it occurred in the first third of the night with his eyes open and impaired recall after the event. Interestingly, Mr. R had RBD in addition to the NREM parasomnia likely caused by zolpidem. This is evident from Mr. R’s frequent dream enactment behaviors, such as kicking, thrashing, and punching during sleep, along with increased EMG activity during REM sleep as recorded on the PSG.10 The presence of RBD could be explained by selective serotonin reuptake inhibitor (fluoxetine) use, and comorbidity with PTSD.2,16
Management of parasomnias
Initial management of parasomnias involves decreasing the risk of parasomnia-related injury. Suggested safety measures include:
- sleeping away from windows
- sleeping in a sleeping bag
- sleeping on a lower floor
- locking windows and doors
- removing potentially dangerous objects from the bedroom
- putting gates across stairwells
- installing bells or alarms on door knobs.15
Removing access to firearms or other weapons such as knives is of utmost importance especially with patients who have easy access during wakefulness. If removing weapons is not feasible, consider disarming, securing, or locking them.15 These considerations are relevant to veterans with PTSD because of the high prevalence of symptoms, including depression, insomnia, and pain, which require sedating medications.17 A review of parasomnias among a large sample of psychiatric outpatients revealed that a variety of sedating medications, including antidepressants, can lead to NREM parasomnias.18 Therefore, exercise caution when prescribing sedating medications, especially in patients vulnerable to developing dangerous parasomnias, such as a veteran with PTSD and easy access to guns.19
TREATMENT Zolpidem stopped
Mr. R immediately stops taking zolpidem because he is aware of its association with abnormal behaviors during sleep, and his wife removes his access to firearms and knives at night. Because of his history of clinical benefit and no history of parasomnias with mirtazapine, Mr. R is started on mirtazapine for insomnia that previously was treated with zolpidem, and residual depression. Six months after discontinuing zolpidem, he does not experience NREM parasomnias, and there are no changes in his dream enactment behaviors.
Summing up
Zolpidem therapy could be associated with unusual variants of NREM parasomnia, sleepwalking type; sleep-related pseudo-suicidal behavior is one such variant. Several factors could play a role in increasing the likelihood of NREM parasomnia with zolpidem therapy. In Mr. R’s case, the pharmacokinetic drug interactions between fluoxetine and zolpidem, as well as concomitant use of several sedating agents could have played a role in increasing the likelihood of NREM parasomnia, with audio-tactile stimuli contributing to the violent and suicidal nature of the parasomnia. Exercise caution when using CYP enzyme inhibitors, such as fluoxetine and paroxetine, in combination with zolpidem. Knowledge of the potential interaction between zolpidem and fluoxetine is important because antidepressants and hypnotics are commonly co-prescribed because insomnia often is comorbid with other psychiatric disorders.
In veterans with PTSD who do not have suicidal ideations while awake, life-threatening non-intentional behavior is a risk because of easy access to guns or other weapons. Sedative-hypnotic medications commonly are prescribed to patients with PTSD. Exercise caution when using hypnotic agents such as zolpidem, and consider sleep aids with a lower risk of parasomnias (based on the author’s experience, trazodone, mirtazapine, melatonin, and gabapentin) when possible. Non-pharmacologic treatments of insomnia, such as sleep hygiene education and, more importantly, cognitive-behavioral therapy for insomnia, are preferred. If a patient is already taking zolpidem, nightly dosage should not be >10 mg. Polypharmacy with other sedating medications should be avoided when possible and both exogenous (noise, pets) and endogenous sleep disruptors (sleep apnea, PLMD) should be addressed. Advise the patient to avoid alcohol and remove firearms and other potential weapons. Discontinue zolpidem if the patient develops sleep-related abnormal behavior because of its potential to take on violent forms.
1. Howell MJ. Parasomnias: an updated review. Neurotherapeutics. 2012;9(4):753-775.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
3. Zadra A, Desautels A, Petit D, et al. Somnambulism: clinical aspects and pathophysiological hypotheses. Lancet Neurol. 2013;12(3):285-294.
4. Chopra A, Selim B, Silber MH, et al. Para-suicidal amnestic behavior associated with chronic zolpidem use: implications for patient safety. Psychosomatics. 2013;54(5):498-501.
5. Hwang TJ, Ni HC, Chen HC, et al. Risk predictors for hypnosedative-related complex sleep behaviors: a retrospective, cross-sectional pilot study. J Clin Psychiatry. 2010;71(10):1331-1335.
6. Shatkin JP, Feinfield K, Strober M. The misinterpretation of a non-REM sleep parasomnia as suicidal behavior in an adolescent. Sleep Breath. 2002;6(4):175-179.
7. Mahowald MW, Schenck CH, Goldner M, et al. Parasomnia pseudo-suicide. J Forensic Sci. 2003;48(5):1158-1162.
8. Gibson CE, Caplan JP. Zolpidem-associated parasomnia with serious self-injury: a shot in the dark. Psychosomatics. 2011;52(1):88-91.
9. Mortaz Hejri S, Faizi M, Babaeian M. Zolpidem-induced suicide attempt: a case report. Daru. 2013;20;21(1):77.
10. Poceta JS. Zolpidem ingestion, automatisms, and sleep driving: a clinical and legal case series. J Clin Sleep Med. 2011;7(6):632-638.
11. Hesse LM, von Moltke LL, Greenblatt DJ. Clinically important drug interactions with zopiclone, zolpidem and zaleplon. CNS Drugs. 2003;17(7):513-532.
12. Catterson ML, Preskorn SH. Pharmacokinetics of selective serotonin reuptake inhibitors: clinical relevance. Pharmacol Toxicol. 1996;78(4):203-208.
13. Rosenberg RP, Hull SG, Lankford DA, et al. A randomized, double-blind, single-dose, placebo-controlled, multicenter, polysomnographic study of gabapentin in transient insomnia induced by sleep phase advance. J Clin Sleep Med. 2014;10(10):1093-1100.
14. Kato T, Montplaisir JY, Lavigne GJ. Experimentally induced arousals during sleep: a cross-modality matching paradigm. J Sleep Res. 2004;13(3):229-238.
15. Siclari F, Khatami R, Urbaniok F, et al. Violence in sleep. Brain. 2010;133(pt 12):3494-3509.
16. Husain AM, Miller PP, Carwile ST. Rem sleep behavior disorder: potential relationship to post-traumatic stress disorder. J Clin Neurophysiol. 2001;18(2):148-157.
17. Bernardy NC, Lund BC, Alexander B, et al. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
18. Lam SP, Fong SY, Ho CK, et al. Parasomnia among psychiatric outpatients: a clinical, epidemiologic, cross-sectional study. J Clin Psychiatry. 2008;69(9):1374-1382.
19. Freeman TW, Roca V, Kimbrell T. A survey of gun collection and use among three groups of veteran patients admitted to veterans affairs hospital treatment programs. South Med J. 2003;96(3):240-243.
CASE Suicidal while asleep
Mr. R, age 28, an Iraq and Afghanistan veteran with major depressive disorder and posttraumatic stress disorder (PTSD), is awoken by his wife to check on their daughter approximately 30 minutes after he takes his nightly regimen of zolpidem, 10 mg, melatonin, 6 mg, and hydroxyzine, 20 mg. When Mr. R returns to the bedroom, he appears to be confused. Mr. R grabs an unloaded gun from under the mattress, puts it in his mouth, and pulls the trigger. Then Mr. R holds the gun to his head and pulls the trigger while saying that his wife and children will be better off without him. His wife takes the gun away, but he grabs another gun from his gun box and loads it. His wife convinces him to remove the ammunition; however, Mr. R gets the other unloaded gun and pulls the trigger on himself again. After his wife takes this gun away, he tries cutting himself with a pocketknife, causing superficial cuts. Eventually, Mr. R goes back to bed. He does not remember these events in the morning.
What increased the likelihood of parasomnia in Mr. R?
a) high zolpidem dosage
b) concomitant use of other sedating agents
c) sleep deprivation
d) dehydration
[polldaddy:9712545]
The authors’ observations
Parasomnias are sleep-wake transition disorders classified by the sleep stage from which they arise, either NREM or rapid eye movement (REM). NREM parasomnias could result from incomplete awakening from NREM sleep, typically in Stage N3 (slow-wave) sleep.1 DSM-5 describes NREM parasomnias as arousal disorders in which the disturbance is not attributable to the physiological effects of substance; substance/medication-induced sleep disorder, parasomnia type, is when the disturbance can be attributed to a substance.2 The latter also can occur during REM sleep.
NREM parasomnias are characterized by abnormal behaviors during sleep with significant harm potential.3 Somnambulism or sleepwalking and sleep terrors are the 2 types of NREM parasomnias in DSM-5. Sleepwalking could involve complex behaviors, including:
- eating
- talking
- cooking
- shopping
- driving
- sexual activity.
Zolpidem, a benzodiazepine receptor agonist, is a preferred hypnotic agent for insomnia because of its low risk for abuse and daytime sedation.4 However, the drug has been associated with NREM parasomnias, namely somnambulism or sleepwalking, and its variants including sleep-driving, sleep-related eating disorder, and rarely sexsomnia (sleep-sex), with anterograde amnesia for the event.5 Suicidal behavior that occurs while the patient is asleep with next-day amnesia is another variant of somnambulism. There are several reports of suicidal behavior during sleep,6,7 but to our knowledge, there are only 2 previous cases implicating zolpidem as the cause:
- Gibson et al8 described a 49-year-old man who sustained a self-inflicted gunshot wound to his head while asleep. He just had started taking zolpidem, and in the weeks before the incident he had several episodes of sleepwalking and sleep-eating. He had consumed alcohol the night of the self-inflicted gunshot wound, but had no other psychiatric history.
- Chopra et al4 described a 37-year-old man, with no prior episodes of sleepwalking or associated complex behaviors, who was taking zolpidem, 10 mg/d, for chronic insomnia. He shot a gun in the basement of his home, and then held the loaded gun to his neck while asleep. The authors attributed the event to zolpidem in combination with other predisposing factors, including dehydration after intense exercise and alcohol use. The authors categorized this type of event as “para-suicidal amnestic behavior,” although “sleep-related pseudo-suicidal behavior” might be a better term for this type of parasomnia because of its occurrence during sleep and non-deliberate nature.
In another case report, a 27-year-old man took additional zolpidem after he did not experience desired sedative effects from an initial 20 mg.9 Because the patient remembered the suicidal thoughts, the authors believed that the patient attempted suicide while under the influence of zolpidem. The authors did not believe the incident to be sleep-related suicidal behavior, because it was uncertain if he attempted suicide while asleep.
Mr. R does not remember the events his wife witnessed while he was asleep. To our knowledge, Mr. R’s case is the first sleep-related pseudo-suicidal behavior case resulting from zolpidem, 10 mg/d, without concurrent alcohol use in an adult male veteran with PTSD and no suicidal ideation while awake.
HISTORY Further details revealed
Mr. R says that in the days leading to the incident he was not sleep-deprived and was getting at least 6 hours of restful sleep every night. He had been taking zolpidem every night. He has no childhood or family history of NREM parasomnias. He says he did not engage in intense exercise that evening or have a fever the night of the incident and has abstained from alcohol for 2 years.
His wife says that after he took zolpidem, when he was woken up, “He was not there; his eyes were glazed and glossy, and it’s like he was in another world,” and his speech and behavior were bizarre. She also reports that his eyes were open when he engaged in this behavior that appeared suicidal.
Three months before the incident, Mr. R had reported nightmares with dream enactment behaviors, hypervigilance on awakening and during the daytime, irritability, and anxious and depressed mood with neurovegetative symptoms, and was referred to our clinic for medication management. He also reported no prior or current manic or psychotic symptoms, denied suicidal thoughts, and had no history of suicide attempts. Mr. R’s medication regimen included tramadol, 400 mg/d, for chronic knee pain; fluoxetine, 60 mg/d, for depression and PTSD; and propranolol ER, 60 mg/d, and propranolol, 10 mg/d as needed, for anxiety. He was started on prazosin, 2 mg/d, titrated to 4 mg/d, for medication management of nightmares.
Mr. R also was referred to the sleep laboratory for a polysomnogram (PSG) because of reported loud snoring and witnessed apneas, especially because sleep apnea can cause nightmares and dream enactment behaviors. The PSG was negative for sleep apnea or excessive periodic limb movements of sleep, but showed increased electromyographic (EMG) activity during REM sleep, which was consistent with his report of dream enactment behaviors. Two months later, he reported improvement in nightmares and depression, but not in dream enactment behaviors. Because of prominent anxiety and irritability, he was started on gabapentin, 300 mg, 3 times a day.
What factor increases the risk of NREM parasomnias with zolpidem compared with benzodiazepines?
a) greater preservation of Stage N3 sleep
b) lesser degree of muscle relaxation
c) both a and b
d) none of the above
[polldaddy:9712556]
The authors’ observations
Factors that increase the likelihood of parasomnias include:
- zolpidem >10 mg at bedtime
- concomitant use of other CNS depressants, including sedative hypnotic agents and alcohol
- female sex
- not falling asleep immediately after taking zolpidem
- personal or family history of parasomnias
- living alone
- poor pill management
- presence of sleep disruptors such as sleep apnea and periodic limb movements of sleep.1,4,5,10
Higher dosages of zolpidem (>10 mg/d) have been identified as the predictive risk factor.5 In the Chopra et al4 case report on sleep-related suicidal behavior related to zolpidem, 10 mg at bedtime, concomitant dehydration and alcohol use were implicated as facilitating factors. Dehydration could increase serum levels of zolpidem resulting in greater CNS effects. Alcohol use was implicated in the Gibson et al8 case report as well, and the patient had multiple episodes of sleepwalking and sleep-related eating.However, Mr. R was not dehydrated or using alcohol.
An interesting feature of Mr. R’s case is that he was taking fluoxetine. Cytochrome P450 (CYP) 3A4 is involved in metabolizing zolpidem, and norfluoxetine, a metabolite of fluoxetine, inhibits CYP3A4. Although studies have not found pharmacokinetic interactions between fluoxetine and zolpidem, these studies did not investigate fluoxetine dosages >20 mg/d.11 The inhibition of CYP enzymes by fluoxetine likely is dose-dependent,12 and therefore concomitant administration of high-dosage fluoxetine (>20 mg/d) with zolpidem might result in higher serum levels of zolpidem.
Mr. R also was taking several sedating agents (gabapentin, hydroxyzine, melatonin, and tramadol). The concomitant use of these sedative-hypnotic agents could have increased his risk of parasomnia. A review of the literature did not reveal any reports of gabapentin, hydroxyzine, melatonin, or tramadol causing parasomnias. This observation, as well as the well-known role of zolpidem5 in etiopathogenesis of parasomnias, indicates that the pseudo-suicidal behavior Mr. R displayed while asleep likely was a direct result of zolpidem use in presence of other facilitating factors. Gabapentin, which is known to increase the depth of sleep, was added to his regimen 1 month before his parasomnia episode. Therefore, gabapentin could have triggered parasomnia with zolpidem therapy.1,13
Conditions that provoke repeated cortical arousals (eg, periodic limb movement disorder [PLMD] and sleep apnea) or increase depth or pressure of sleep (intense exercise in the evening, fever, sleep deprivation) are thought to be associated with NREM parasomnias.1-4 However, Mr. R underwent in-laboratory PSG and tested negative for major cortical arousal-inducing conditions, such as PLMD and sleep apnea.
Some other sleep disruptors likely were involved in Mr. R’s case. Auditory and tactile stimuli are known to cause cortical arousals, with additive effect seen when these 2 stimuli are combined.3,14 Additionally, these exogenous stimuli are known to trigger sleep-related violent parasomnias.15 Mr. R displayed this behavior after his wife woke him up. The auditory stimulus of his wife’s voice and/or tactile stimulus involved in the act of waking Mr. R likely played a role in the suicidal and violent nature of his NREM parasomnia.
[polldaddy:9712581]
The authors’ observations
In general, the mechanisms by which zolpidem causes NREM parasomnias are not completely understood. The sedation-related amnestic properties of zolpidem might explain some of these behaviors. Patients could perform these behaviors after waking and have subsequent amnesia.4 There is greater preservation of Stage N3 sleep with zolpidem compared with benzodiazepines. Benzodiazepines also cause muscle relaxation while the motor system remains relatively more active during sleep with zolpidem because of its selectivity for α-1 subunit of gamma-aminobutyric acid A receptor. These factors might increase the likelihood of NREM parasomnias with zolpidem compared with benzodiazepines.4
Types of parasomnias
According to DSM-5, there are 2 categories of parasomnias based on the sleep stage from which a parasomnia emerges.2 REM sleep behavior disorder (RBD) refers to complex motor and/or vocalizations during REM sleep, accompanied by increased EMG activity during REM sleep (Table).2,3

The pseudo-suicidal behavior Mr. R displayed likely was NREM parasomnia because it occurred in the first third of the night with his eyes open and impaired recall after the event. Interestingly, Mr. R had RBD in addition to the NREM parasomnia likely caused by zolpidem. This is evident from Mr. R’s frequent dream enactment behaviors, such as kicking, thrashing, and punching during sleep, along with increased EMG activity during REM sleep as recorded on the PSG.10 The presence of RBD could be explained by selective serotonin reuptake inhibitor (fluoxetine) use, and comorbidity with PTSD.2,16
Management of parasomnias
Initial management of parasomnias involves decreasing the risk of parasomnia-related injury. Suggested safety measures include:
- sleeping away from windows
- sleeping in a sleeping bag
- sleeping on a lower floor
- locking windows and doors
- removing potentially dangerous objects from the bedroom
- putting gates across stairwells
- installing bells or alarms on door knobs.15
Removing access to firearms or other weapons such as knives is of utmost importance especially with patients who have easy access during wakefulness. If removing weapons is not feasible, consider disarming, securing, or locking them.15 These considerations are relevant to veterans with PTSD because of the high prevalence of symptoms, including depression, insomnia, and pain, which require sedating medications.17 A review of parasomnias among a large sample of psychiatric outpatients revealed that a variety of sedating medications, including antidepressants, can lead to NREM parasomnias.18 Therefore, exercise caution when prescribing sedating medications, especially in patients vulnerable to developing dangerous parasomnias, such as a veteran with PTSD and easy access to guns.19
TREATMENT Zolpidem stopped
Mr. R immediately stops taking zolpidem because he is aware of its association with abnormal behaviors during sleep, and his wife removes his access to firearms and knives at night. Because of his history of clinical benefit and no history of parasomnias with mirtazapine, Mr. R is started on mirtazapine for insomnia that previously was treated with zolpidem, and residual depression. Six months after discontinuing zolpidem, he does not experience NREM parasomnias, and there are no changes in his dream enactment behaviors.
Summing up
Zolpidem therapy could be associated with unusual variants of NREM parasomnia, sleepwalking type; sleep-related pseudo-suicidal behavior is one such variant. Several factors could play a role in increasing the likelihood of NREM parasomnia with zolpidem therapy. In Mr. R’s case, the pharmacokinetic drug interactions between fluoxetine and zolpidem, as well as concomitant use of several sedating agents could have played a role in increasing the likelihood of NREM parasomnia, with audio-tactile stimuli contributing to the violent and suicidal nature of the parasomnia. Exercise caution when using CYP enzyme inhibitors, such as fluoxetine and paroxetine, in combination with zolpidem. Knowledge of the potential interaction between zolpidem and fluoxetine is important because antidepressants and hypnotics are commonly co-prescribed because insomnia often is comorbid with other psychiatric disorders.
In veterans with PTSD who do not have suicidal ideations while awake, life-threatening non-intentional behavior is a risk because of easy access to guns or other weapons. Sedative-hypnotic medications commonly are prescribed to patients with PTSD. Exercise caution when using hypnotic agents such as zolpidem, and consider sleep aids with a lower risk of parasomnias (based on the author’s experience, trazodone, mirtazapine, melatonin, and gabapentin) when possible. Non-pharmacologic treatments of insomnia, such as sleep hygiene education and, more importantly, cognitive-behavioral therapy for insomnia, are preferred. If a patient is already taking zolpidem, nightly dosage should not be >10 mg. Polypharmacy with other sedating medications should be avoided when possible and both exogenous (noise, pets) and endogenous sleep disruptors (sleep apnea, PLMD) should be addressed. Advise the patient to avoid alcohol and remove firearms and other potential weapons. Discontinue zolpidem if the patient develops sleep-related abnormal behavior because of its potential to take on violent forms.
CASE Suicidal while asleep
Mr. R, age 28, an Iraq and Afghanistan veteran with major depressive disorder and posttraumatic stress disorder (PTSD), is awoken by his wife to check on their daughter approximately 30 minutes after he takes his nightly regimen of zolpidem, 10 mg, melatonin, 6 mg, and hydroxyzine, 20 mg. When Mr. R returns to the bedroom, he appears to be confused. Mr. R grabs an unloaded gun from under the mattress, puts it in his mouth, and pulls the trigger. Then Mr. R holds the gun to his head and pulls the trigger while saying that his wife and children will be better off without him. His wife takes the gun away, but he grabs another gun from his gun box and loads it. His wife convinces him to remove the ammunition; however, Mr. R gets the other unloaded gun and pulls the trigger on himself again. After his wife takes this gun away, he tries cutting himself with a pocketknife, causing superficial cuts. Eventually, Mr. R goes back to bed. He does not remember these events in the morning.
What increased the likelihood of parasomnia in Mr. R?
a) high zolpidem dosage
b) concomitant use of other sedating agents
c) sleep deprivation
d) dehydration
[polldaddy:9712545]
The authors’ observations
Parasomnias are sleep-wake transition disorders classified by the sleep stage from which they arise, either NREM or rapid eye movement (REM). NREM parasomnias could result from incomplete awakening from NREM sleep, typically in Stage N3 (slow-wave) sleep.1 DSM-5 describes NREM parasomnias as arousal disorders in which the disturbance is not attributable to the physiological effects of substance; substance/medication-induced sleep disorder, parasomnia type, is when the disturbance can be attributed to a substance.2 The latter also can occur during REM sleep.
NREM parasomnias are characterized by abnormal behaviors during sleep with significant harm potential.3 Somnambulism or sleepwalking and sleep terrors are the 2 types of NREM parasomnias in DSM-5. Sleepwalking could involve complex behaviors, including:
- eating
- talking
- cooking
- shopping
- driving
- sexual activity.
Zolpidem, a benzodiazepine receptor agonist, is a preferred hypnotic agent for insomnia because of its low risk for abuse and daytime sedation.4 However, the drug has been associated with NREM parasomnias, namely somnambulism or sleepwalking, and its variants including sleep-driving, sleep-related eating disorder, and rarely sexsomnia (sleep-sex), with anterograde amnesia for the event.5 Suicidal behavior that occurs while the patient is asleep with next-day amnesia is another variant of somnambulism. There are several reports of suicidal behavior during sleep,6,7 but to our knowledge, there are only 2 previous cases implicating zolpidem as the cause:
- Gibson et al8 described a 49-year-old man who sustained a self-inflicted gunshot wound to his head while asleep. He just had started taking zolpidem, and in the weeks before the incident he had several episodes of sleepwalking and sleep-eating. He had consumed alcohol the night of the self-inflicted gunshot wound, but had no other psychiatric history.
- Chopra et al4 described a 37-year-old man, with no prior episodes of sleepwalking or associated complex behaviors, who was taking zolpidem, 10 mg/d, for chronic insomnia. He shot a gun in the basement of his home, and then held the loaded gun to his neck while asleep. The authors attributed the event to zolpidem in combination with other predisposing factors, including dehydration after intense exercise and alcohol use. The authors categorized this type of event as “para-suicidal amnestic behavior,” although “sleep-related pseudo-suicidal behavior” might be a better term for this type of parasomnia because of its occurrence during sleep and non-deliberate nature.
In another case report, a 27-year-old man took additional zolpidem after he did not experience desired sedative effects from an initial 20 mg.9 Because the patient remembered the suicidal thoughts, the authors believed that the patient attempted suicide while under the influence of zolpidem. The authors did not believe the incident to be sleep-related suicidal behavior, because it was uncertain if he attempted suicide while asleep.
Mr. R does not remember the events his wife witnessed while he was asleep. To our knowledge, Mr. R’s case is the first sleep-related pseudo-suicidal behavior case resulting from zolpidem, 10 mg/d, without concurrent alcohol use in an adult male veteran with PTSD and no suicidal ideation while awake.
HISTORY Further details revealed
Mr. R says that in the days leading to the incident he was not sleep-deprived and was getting at least 6 hours of restful sleep every night. He had been taking zolpidem every night. He has no childhood or family history of NREM parasomnias. He says he did not engage in intense exercise that evening or have a fever the night of the incident and has abstained from alcohol for 2 years.
His wife says that after he took zolpidem, when he was woken up, “He was not there; his eyes were glazed and glossy, and it’s like he was in another world,” and his speech and behavior were bizarre. She also reports that his eyes were open when he engaged in this behavior that appeared suicidal.
Three months before the incident, Mr. R had reported nightmares with dream enactment behaviors, hypervigilance on awakening and during the daytime, irritability, and anxious and depressed mood with neurovegetative symptoms, and was referred to our clinic for medication management. He also reported no prior or current manic or psychotic symptoms, denied suicidal thoughts, and had no history of suicide attempts. Mr. R’s medication regimen included tramadol, 400 mg/d, for chronic knee pain; fluoxetine, 60 mg/d, for depression and PTSD; and propranolol ER, 60 mg/d, and propranolol, 10 mg/d as needed, for anxiety. He was started on prazosin, 2 mg/d, titrated to 4 mg/d, for medication management of nightmares.
Mr. R also was referred to the sleep laboratory for a polysomnogram (PSG) because of reported loud snoring and witnessed apneas, especially because sleep apnea can cause nightmares and dream enactment behaviors. The PSG was negative for sleep apnea or excessive periodic limb movements of sleep, but showed increased electromyographic (EMG) activity during REM sleep, which was consistent with his report of dream enactment behaviors. Two months later, he reported improvement in nightmares and depression, but not in dream enactment behaviors. Because of prominent anxiety and irritability, he was started on gabapentin, 300 mg, 3 times a day.
What factor increases the risk of NREM parasomnias with zolpidem compared with benzodiazepines?
a) greater preservation of Stage N3 sleep
b) lesser degree of muscle relaxation
c) both a and b
d) none of the above
[polldaddy:9712556]
The authors’ observations
Factors that increase the likelihood of parasomnias include:
- zolpidem >10 mg at bedtime
- concomitant use of other CNS depressants, including sedative hypnotic agents and alcohol
- female sex
- not falling asleep immediately after taking zolpidem
- personal or family history of parasomnias
- living alone
- poor pill management
- presence of sleep disruptors such as sleep apnea and periodic limb movements of sleep.1,4,5,10
Higher dosages of zolpidem (>10 mg/d) have been identified as the predictive risk factor.5 In the Chopra et al4 case report on sleep-related suicidal behavior related to zolpidem, 10 mg at bedtime, concomitant dehydration and alcohol use were implicated as facilitating factors. Dehydration could increase serum levels of zolpidem resulting in greater CNS effects. Alcohol use was implicated in the Gibson et al8 case report as well, and the patient had multiple episodes of sleepwalking and sleep-related eating.However, Mr. R was not dehydrated or using alcohol.
An interesting feature of Mr. R’s case is that he was taking fluoxetine. Cytochrome P450 (CYP) 3A4 is involved in metabolizing zolpidem, and norfluoxetine, a metabolite of fluoxetine, inhibits CYP3A4. Although studies have not found pharmacokinetic interactions between fluoxetine and zolpidem, these studies did not investigate fluoxetine dosages >20 mg/d.11 The inhibition of CYP enzymes by fluoxetine likely is dose-dependent,12 and therefore concomitant administration of high-dosage fluoxetine (>20 mg/d) with zolpidem might result in higher serum levels of zolpidem.
Mr. R also was taking several sedating agents (gabapentin, hydroxyzine, melatonin, and tramadol). The concomitant use of these sedative-hypnotic agents could have increased his risk of parasomnia. A review of the literature did not reveal any reports of gabapentin, hydroxyzine, melatonin, or tramadol causing parasomnias. This observation, as well as the well-known role of zolpidem5 in etiopathogenesis of parasomnias, indicates that the pseudo-suicidal behavior Mr. R displayed while asleep likely was a direct result of zolpidem use in presence of other facilitating factors. Gabapentin, which is known to increase the depth of sleep, was added to his regimen 1 month before his parasomnia episode. Therefore, gabapentin could have triggered parasomnia with zolpidem therapy.1,13
Conditions that provoke repeated cortical arousals (eg, periodic limb movement disorder [PLMD] and sleep apnea) or increase depth or pressure of sleep (intense exercise in the evening, fever, sleep deprivation) are thought to be associated with NREM parasomnias.1-4 However, Mr. R underwent in-laboratory PSG and tested negative for major cortical arousal-inducing conditions, such as PLMD and sleep apnea.
Some other sleep disruptors likely were involved in Mr. R’s case. Auditory and tactile stimuli are known to cause cortical arousals, with additive effect seen when these 2 stimuli are combined.3,14 Additionally, these exogenous stimuli are known to trigger sleep-related violent parasomnias.15 Mr. R displayed this behavior after his wife woke him up. The auditory stimulus of his wife’s voice and/or tactile stimulus involved in the act of waking Mr. R likely played a role in the suicidal and violent nature of his NREM parasomnia.
[polldaddy:9712581]
The authors’ observations
In general, the mechanisms by which zolpidem causes NREM parasomnias are not completely understood. The sedation-related amnestic properties of zolpidem might explain some of these behaviors. Patients could perform these behaviors after waking and have subsequent amnesia.4 There is greater preservation of Stage N3 sleep with zolpidem compared with benzodiazepines. Benzodiazepines also cause muscle relaxation while the motor system remains relatively more active during sleep with zolpidem because of its selectivity for α-1 subunit of gamma-aminobutyric acid A receptor. These factors might increase the likelihood of NREM parasomnias with zolpidem compared with benzodiazepines.4
Types of parasomnias
According to DSM-5, there are 2 categories of parasomnias based on the sleep stage from which a parasomnia emerges.2 REM sleep behavior disorder (RBD) refers to complex motor and/or vocalizations during REM sleep, accompanied by increased EMG activity during REM sleep (Table).2,3

The pseudo-suicidal behavior Mr. R displayed likely was NREM parasomnia because it occurred in the first third of the night with his eyes open and impaired recall after the event. Interestingly, Mr. R had RBD in addition to the NREM parasomnia likely caused by zolpidem. This is evident from Mr. R’s frequent dream enactment behaviors, such as kicking, thrashing, and punching during sleep, along with increased EMG activity during REM sleep as recorded on the PSG.10 The presence of RBD could be explained by selective serotonin reuptake inhibitor (fluoxetine) use, and comorbidity with PTSD.2,16
Management of parasomnias
Initial management of parasomnias involves decreasing the risk of parasomnia-related injury. Suggested safety measures include:
- sleeping away from windows
- sleeping in a sleeping bag
- sleeping on a lower floor
- locking windows and doors
- removing potentially dangerous objects from the bedroom
- putting gates across stairwells
- installing bells or alarms on door knobs.15
Removing access to firearms or other weapons such as knives is of utmost importance especially with patients who have easy access during wakefulness. If removing weapons is not feasible, consider disarming, securing, or locking them.15 These considerations are relevant to veterans with PTSD because of the high prevalence of symptoms, including depression, insomnia, and pain, which require sedating medications.17 A review of parasomnias among a large sample of psychiatric outpatients revealed that a variety of sedating medications, including antidepressants, can lead to NREM parasomnias.18 Therefore, exercise caution when prescribing sedating medications, especially in patients vulnerable to developing dangerous parasomnias, such as a veteran with PTSD and easy access to guns.19
TREATMENT Zolpidem stopped
Mr. R immediately stops taking zolpidem because he is aware of its association with abnormal behaviors during sleep, and his wife removes his access to firearms and knives at night. Because of his history of clinical benefit and no history of parasomnias with mirtazapine, Mr. R is started on mirtazapine for insomnia that previously was treated with zolpidem, and residual depression. Six months after discontinuing zolpidem, he does not experience NREM parasomnias, and there are no changes in his dream enactment behaviors.
Summing up
Zolpidem therapy could be associated with unusual variants of NREM parasomnia, sleepwalking type; sleep-related pseudo-suicidal behavior is one such variant. Several factors could play a role in increasing the likelihood of NREM parasomnia with zolpidem therapy. In Mr. R’s case, the pharmacokinetic drug interactions between fluoxetine and zolpidem, as well as concomitant use of several sedating agents could have played a role in increasing the likelihood of NREM parasomnia, with audio-tactile stimuli contributing to the violent and suicidal nature of the parasomnia. Exercise caution when using CYP enzyme inhibitors, such as fluoxetine and paroxetine, in combination with zolpidem. Knowledge of the potential interaction between zolpidem and fluoxetine is important because antidepressants and hypnotics are commonly co-prescribed because insomnia often is comorbid with other psychiatric disorders.
In veterans with PTSD who do not have suicidal ideations while awake, life-threatening non-intentional behavior is a risk because of easy access to guns or other weapons. Sedative-hypnotic medications commonly are prescribed to patients with PTSD. Exercise caution when using hypnotic agents such as zolpidem, and consider sleep aids with a lower risk of parasomnias (based on the author’s experience, trazodone, mirtazapine, melatonin, and gabapentin) when possible. Non-pharmacologic treatments of insomnia, such as sleep hygiene education and, more importantly, cognitive-behavioral therapy for insomnia, are preferred. If a patient is already taking zolpidem, nightly dosage should not be >10 mg. Polypharmacy with other sedating medications should be avoided when possible and both exogenous (noise, pets) and endogenous sleep disruptors (sleep apnea, PLMD) should be addressed. Advise the patient to avoid alcohol and remove firearms and other potential weapons. Discontinue zolpidem if the patient develops sleep-related abnormal behavior because of its potential to take on violent forms.
1. Howell MJ. Parasomnias: an updated review. Neurotherapeutics. 2012;9(4):753-775.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
3. Zadra A, Desautels A, Petit D, et al. Somnambulism: clinical aspects and pathophysiological hypotheses. Lancet Neurol. 2013;12(3):285-294.
4. Chopra A, Selim B, Silber MH, et al. Para-suicidal amnestic behavior associated with chronic zolpidem use: implications for patient safety. Psychosomatics. 2013;54(5):498-501.
5. Hwang TJ, Ni HC, Chen HC, et al. Risk predictors for hypnosedative-related complex sleep behaviors: a retrospective, cross-sectional pilot study. J Clin Psychiatry. 2010;71(10):1331-1335.
6. Shatkin JP, Feinfield K, Strober M. The misinterpretation of a non-REM sleep parasomnia as suicidal behavior in an adolescent. Sleep Breath. 2002;6(4):175-179.
7. Mahowald MW, Schenck CH, Goldner M, et al. Parasomnia pseudo-suicide. J Forensic Sci. 2003;48(5):1158-1162.
8. Gibson CE, Caplan JP. Zolpidem-associated parasomnia with serious self-injury: a shot in the dark. Psychosomatics. 2011;52(1):88-91.
9. Mortaz Hejri S, Faizi M, Babaeian M. Zolpidem-induced suicide attempt: a case report. Daru. 2013;20;21(1):77.
10. Poceta JS. Zolpidem ingestion, automatisms, and sleep driving: a clinical and legal case series. J Clin Sleep Med. 2011;7(6):632-638.
11. Hesse LM, von Moltke LL, Greenblatt DJ. Clinically important drug interactions with zopiclone, zolpidem and zaleplon. CNS Drugs. 2003;17(7):513-532.
12. Catterson ML, Preskorn SH. Pharmacokinetics of selective serotonin reuptake inhibitors: clinical relevance. Pharmacol Toxicol. 1996;78(4):203-208.
13. Rosenberg RP, Hull SG, Lankford DA, et al. A randomized, double-blind, single-dose, placebo-controlled, multicenter, polysomnographic study of gabapentin in transient insomnia induced by sleep phase advance. J Clin Sleep Med. 2014;10(10):1093-1100.
14. Kato T, Montplaisir JY, Lavigne GJ. Experimentally induced arousals during sleep: a cross-modality matching paradigm. J Sleep Res. 2004;13(3):229-238.
15. Siclari F, Khatami R, Urbaniok F, et al. Violence in sleep. Brain. 2010;133(pt 12):3494-3509.
16. Husain AM, Miller PP, Carwile ST. Rem sleep behavior disorder: potential relationship to post-traumatic stress disorder. J Clin Neurophysiol. 2001;18(2):148-157.
17. Bernardy NC, Lund BC, Alexander B, et al. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
18. Lam SP, Fong SY, Ho CK, et al. Parasomnia among psychiatric outpatients: a clinical, epidemiologic, cross-sectional study. J Clin Psychiatry. 2008;69(9):1374-1382.
19. Freeman TW, Roca V, Kimbrell T. A survey of gun collection and use among three groups of veteran patients admitted to veterans affairs hospital treatment programs. South Med J. 2003;96(3):240-243.
1. Howell MJ. Parasomnias: an updated review. Neurotherapeutics. 2012;9(4):753-775.
2. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
3. Zadra A, Desautels A, Petit D, et al. Somnambulism: clinical aspects and pathophysiological hypotheses. Lancet Neurol. 2013;12(3):285-294.
4. Chopra A, Selim B, Silber MH, et al. Para-suicidal amnestic behavior associated with chronic zolpidem use: implications for patient safety. Psychosomatics. 2013;54(5):498-501.
5. Hwang TJ, Ni HC, Chen HC, et al. Risk predictors for hypnosedative-related complex sleep behaviors: a retrospective, cross-sectional pilot study. J Clin Psychiatry. 2010;71(10):1331-1335.
6. Shatkin JP, Feinfield K, Strober M. The misinterpretation of a non-REM sleep parasomnia as suicidal behavior in an adolescent. Sleep Breath. 2002;6(4):175-179.
7. Mahowald MW, Schenck CH, Goldner M, et al. Parasomnia pseudo-suicide. J Forensic Sci. 2003;48(5):1158-1162.
8. Gibson CE, Caplan JP. Zolpidem-associated parasomnia with serious self-injury: a shot in the dark. Psychosomatics. 2011;52(1):88-91.
9. Mortaz Hejri S, Faizi M, Babaeian M. Zolpidem-induced suicide attempt: a case report. Daru. 2013;20;21(1):77.
10. Poceta JS. Zolpidem ingestion, automatisms, and sleep driving: a clinical and legal case series. J Clin Sleep Med. 2011;7(6):632-638.
11. Hesse LM, von Moltke LL, Greenblatt DJ. Clinically important drug interactions with zopiclone, zolpidem and zaleplon. CNS Drugs. 2003;17(7):513-532.
12. Catterson ML, Preskorn SH. Pharmacokinetics of selective serotonin reuptake inhibitors: clinical relevance. Pharmacol Toxicol. 1996;78(4):203-208.
13. Rosenberg RP, Hull SG, Lankford DA, et al. A randomized, double-blind, single-dose, placebo-controlled, multicenter, polysomnographic study of gabapentin in transient insomnia induced by sleep phase advance. J Clin Sleep Med. 2014;10(10):1093-1100.
14. Kato T, Montplaisir JY, Lavigne GJ. Experimentally induced arousals during sleep: a cross-modality matching paradigm. J Sleep Res. 2004;13(3):229-238.
15. Siclari F, Khatami R, Urbaniok F, et al. Violence in sleep. Brain. 2010;133(pt 12):3494-3509.
16. Husain AM, Miller PP, Carwile ST. Rem sleep behavior disorder: potential relationship to post-traumatic stress disorder. J Clin Neurophysiol. 2001;18(2):148-157.
17. Bernardy NC, Lund BC, Alexander B, et al. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
18. Lam SP, Fong SY, Ho CK, et al. Parasomnia among psychiatric outpatients: a clinical, epidemiologic, cross-sectional study. J Clin Psychiatry. 2008;69(9):1374-1382.
19. Freeman TW, Roca V, Kimbrell T. A survey of gun collection and use among three groups of veteran patients admitted to veterans affairs hospital treatment programs. South Med J. 2003;96(3):240-243.
Following the Trajectory of PTSD
Does the course of posttraumatic stress disorder (PTSD) differ depending on whether the person is in the military or has left? Researchers from Naval Health Research Center and the VA wondered whether separation from the military could create a “significant disruption of routine, order, and structure,” which might exacerbate PTSD symptoms, and would the symptoms subside as the veteran adjusted to civilian life?
Using data from the Millennium Cohort Study, researchers examined trajectories of PTSD among 22,080 military personnel across 4 time points, about 3 years apart, from 2001 to 2013. They compared trajectories between people who separated before the second time point or remained in the military across the entire study period. The researchers assessed PTSD screening and symptoms using the PTSD Checklist-Civilian, for which higher scores represent more severe symptoms.
The researchers say 4 distinct classes described symptom trajectories: resilient, delayed onset, improving, and elevated-recovering. Overall, the trajectories were similar for veterans and active-duty personnel. Veterans had a higher likelihood of screening positive for PTSD at baseline before separation and were more likely to newly screen positive for PTSD at waves 2, 3, and 4. Of participants who screened positive for PTSD, veterans had more severe symptoms compared with active-duty personnel at baseline but not at any subsequent assessments.
However, differences between the “elevated-recovering” classes grew over time, showing that veterans did not recover as soon or as “dramatically,” the researchers say. This might be due to symptoms being exacerbated by the change in routine.
The good news is that most veterans and active-duty personnel fell into the resilient class (82% and 87%, respectively). The researchers cite other studies that have found resilience is the most common response to PTSD.
The researchers noted several risk factors for slower recovery, such as lower physical well-being and a history of multiple life stressors. The “delayed onset” group may be a good target for interventions, they suggest. This group reported high use of VA care, but still 26% reported no VA care, indicating that they could benefit from continued efforts to identify and treat them.
Does the course of posttraumatic stress disorder (PTSD) differ depending on whether the person is in the military or has left? Researchers from Naval Health Research Center and the VA wondered whether separation from the military could create a “significant disruption of routine, order, and structure,” which might exacerbate PTSD symptoms, and would the symptoms subside as the veteran adjusted to civilian life?
Using data from the Millennium Cohort Study, researchers examined trajectories of PTSD among 22,080 military personnel across 4 time points, about 3 years apart, from 2001 to 2013. They compared trajectories between people who separated before the second time point or remained in the military across the entire study period. The researchers assessed PTSD screening and symptoms using the PTSD Checklist-Civilian, for which higher scores represent more severe symptoms.
The researchers say 4 distinct classes described symptom trajectories: resilient, delayed onset, improving, and elevated-recovering. Overall, the trajectories were similar for veterans and active-duty personnel. Veterans had a higher likelihood of screening positive for PTSD at baseline before separation and were more likely to newly screen positive for PTSD at waves 2, 3, and 4. Of participants who screened positive for PTSD, veterans had more severe symptoms compared with active-duty personnel at baseline but not at any subsequent assessments.
However, differences between the “elevated-recovering” classes grew over time, showing that veterans did not recover as soon or as “dramatically,” the researchers say. This might be due to symptoms being exacerbated by the change in routine.
The good news is that most veterans and active-duty personnel fell into the resilient class (82% and 87%, respectively). The researchers cite other studies that have found resilience is the most common response to PTSD.
The researchers noted several risk factors for slower recovery, such as lower physical well-being and a history of multiple life stressors. The “delayed onset” group may be a good target for interventions, they suggest. This group reported high use of VA care, but still 26% reported no VA care, indicating that they could benefit from continued efforts to identify and treat them.
Does the course of posttraumatic stress disorder (PTSD) differ depending on whether the person is in the military or has left? Researchers from Naval Health Research Center and the VA wondered whether separation from the military could create a “significant disruption of routine, order, and structure,” which might exacerbate PTSD symptoms, and would the symptoms subside as the veteran adjusted to civilian life?
Using data from the Millennium Cohort Study, researchers examined trajectories of PTSD among 22,080 military personnel across 4 time points, about 3 years apart, from 2001 to 2013. They compared trajectories between people who separated before the second time point or remained in the military across the entire study period. The researchers assessed PTSD screening and symptoms using the PTSD Checklist-Civilian, for which higher scores represent more severe symptoms.
The researchers say 4 distinct classes described symptom trajectories: resilient, delayed onset, improving, and elevated-recovering. Overall, the trajectories were similar for veterans and active-duty personnel. Veterans had a higher likelihood of screening positive for PTSD at baseline before separation and were more likely to newly screen positive for PTSD at waves 2, 3, and 4. Of participants who screened positive for PTSD, veterans had more severe symptoms compared with active-duty personnel at baseline but not at any subsequent assessments.
However, differences between the “elevated-recovering” classes grew over time, showing that veterans did not recover as soon or as “dramatically,” the researchers say. This might be due to symptoms being exacerbated by the change in routine.
The good news is that most veterans and active-duty personnel fell into the resilient class (82% and 87%, respectively). The researchers cite other studies that have found resilience is the most common response to PTSD.
The researchers noted several risk factors for slower recovery, such as lower physical well-being and a history of multiple life stressors. The “delayed onset” group may be a good target for interventions, they suggest. This group reported high use of VA care, but still 26% reported no VA care, indicating that they could benefit from continued efforts to identify and treat them.
Posttraumatic Stress Disorder: Developments in Assessment and Treatment
The U.S. veteran population of about 22 million is estimated to represent > 9% of U.S. adults.1 Of veterans using health services, a growing proportion have been deployed in Iraq or Afghanistan. The VA and DoD have made it a priority to address the health care needs of service members and veterans affected by these wars, with a particular focus on posttraumatic stress disorder (PTSD) and mild traumatic brain injury (mTBI). This review highlights key developments in assessment and treatment of PTSD.
Epidemiology
Although studies have produced widely varying estimates of the prevalence of PTSD following deployment to Iraq or Afghanistan, more consistent estimates have been obtained by appropriately grouping studies that used a similar methodology.2 Current PTSD prevalence after Iraq and Afghanistan deployments has averaged 5% to 6% in general military population samples that included support personnel from all services (constituting about two-thirds of deployed forces) and 13% in studies that focused on operational infantry units exposed to direct combat. 2
Among Iraq and Afghanistan war veterans who have sought care at VA treatment facilities, > 25% have received a diagnosis of PTSD (Karen H. Seal, MD, written communication, December 2014). Although combat frequency and intensity is the most prevalent predictor of PTSD in this population, other types of trauma, such as sexual assault, can confer a risk as high as direct combat.3 Another strong correlate with postdeployment PTSD has been deployment-related mTBIs (concussions), especially following blast exposure. The most likely mechanism for this seems to be the extreme life-threatening context in which these concussions occur.4
Posttraumatic stress disorder has been linked with a host of comorbid conditions, including depression, anxiety disorders, substance use disorders, physical symptoms, anger, aggression, complicated grief, and risky behaviors.5-7 Deployments also have been shown to have cumulative effects on the psychological health of military spouses and children.8,9
Diagnosis
One of the most important developments in PTSD is the new definition, which was released in May 2013 in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5), replacing the definition that has proved highly useful for clinical, neurobiologic, research, and public health purposes for > 25 years.10 The PTSD definition underwent much more extensive changes in the DSM-5 than did any other common mental disorder affecting adults. Changes included moving this diagnosis out of the anxiety disorders chapter into a separate “Trauma- and Stressor-Related Disorders” chapter, which now includes adjustment disorder.
The A trauma criteria were revised, including removal of criterion A2 (response of “fear, helplessness, or horror”), in part because individuals who train for traumatic events as part of military and first responder occupations often do not report this response. The 3 symptom-criteria clusters were divided into 4 clusters: B (intrusion), C (avoidance), D (negative alternations in cognitions and mood), and E (alternations in arousal and reactivity). Three symptoms were added, bringing the total from 17 to 20, and many symptoms were substantially reworded.
The implications of these revisions are an active area of investigation. However, initial evidence suggests that these changes have not necessarily improved the clinical utility of the definition.11,12 Although research suggests that the 2 criteria sets result in a similar prevalence of PTSD, they do not identify the same individuals, and there is no evidence that clinical accuracy is any greater when using the revised definition.
One head-to-head comparison of symptom criteria between the DSM-5 version of the PTSD Checklist (PCL-5) and the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) version (PCL-S) among 1,822 infantry soldiers showed high discordance.11 Of soldiers who met DSM-IV symptom criteria, 30% did not meet DSM-5 criteria, and an equivalent number only met criteria under DSM-5. This was one of the only studies that maintained independence of the 2 clinical scales (rather than adding DSM-5 symptoms to an existing DSM-IV scale). This study also controlled for the order in which the 2 scales were presented on the survey, which was found to be critically important.11 Furthermore, there was no difference in the overlap with comorbid conditions (eg, major depression, generalized anxiety, alcohol misuse) and no difference in level of functional impairment, suggesting that the new definition has no greater specificity or clinical utility than does the original.
The diagnostic discordance between DSM-IV and DSM-5 was mostly accounted for by the redefinition of C criterion (avoidance) in DSM-5. In an editorial accompanying the comparison study, Alexander C. McFarlane, MD, AO, raised concerns about this decision and the decision to effectively remove the important symptom of numbing of emotions.12 These changes could disenfranchise service members and veterans who learn to override avoidant behaviors through training and who often cope with their reactivity through emotional numbing.12 These various concerns, as well as years of experience
with the original definition and importance of aligning trauma-focused treatment with the most appropriate diagnosis, suggest that clinicians have reasonable justification for continuing to assign the PTSD diagnosis for veterans who meet full criteria for PTSD under the previous DSM-IV definition.
Another clinical concern of the revision is the potential problem with selecting a diagnosis for subthreshold PTSD. Subthreshold PTSD can be broadly defined as PTSD symptoms that do not meet full criteria for diagnosis but impair functioning sufficiently for traumafocused treatment to be indicated. DSM-5 recommends use of adjustment disorder in this circumstance.10 However, in military populations, this definition carries a pejorative connotation (weakness or failure to adapt) and can also lead to administrative separation without medical benefits if the condition has lasted < 6 months.13 Chronic
adjustment disorder is medically compensable under VA and DoD regulations but suffers from poorly defined clinical criteria, in addition to the pejorative connotation. (Chronic adjustment disorder was inadvertently left out of the print version of DSM-5 but corrected online.)
As a result of these concerns, the VA National Center for PTSD has recommended that clinicians use ICD-9 code 309.89 (“other specified trauma- and stressor-related disorder” in DSM-5) instead of adjustment disorder for subthreshold PTSD.14 However, current DoD electronic medical record systems map this code to even more pejorative labels (eg, adjustment disorder with aggression, antisocial behavior, or destructiveness). Thus, until the
VA and DoD adopt ICD-10 coding, which is expected to occur within the next year, clinicians will probably have to continue to use ICD-9 code 300.00 (“unspecified anxiety disorder” or “anxiety disorder not otherwise specified”) for clinically significant subthreshold PTSD.
Clinical Treatment
Although the VA and DoD have codeveloped a PTSD clinical practice guideline (CPG) and are working closely to ensure seamless transition of care, their policy approaches to PTSD treatment have been quite different.15 The VA has placed particular attention on training PTSD specialists in prolonged exposure (PE) and cognitive processing therapy (CPT) and ensuring that all veterans have access to one of these two “formulary,” trauma-focused psychotherapies.16 U.S. Army and DoD policies have emphasized that PTSD treatment should be a core competency for all mental health providers and have interpreted the literature and CPG recommendations broadly, emphasizing the core components found in many different trauma-focused psychotherapies and encouraging use of a wide range of options.16
One of the reasons for DoD’s approach has been the recognition that the most important threat to treatment efficacy is not the fidelity in which manualized treatments are delivered but the willingness of patients to engage in care and their rate of dropping out.16,17 Several studies from VA health care settings have shown that only one-third of Operation Enduring Freedom/Operation Iraqi Freedom veterans diagnosed with PTSD end up receiving a minimally acceptable number of treatment sessions to have reasonable expectation of recovery.18-20 One study suggested that treatment adequacy is somewhat higher in DoD among active-duty soldiers (41%-52%), though still far from optimal.17
For treatment interventions, the VA/DoD CPG remains an authoritative guide, with A-level recommendations (strong evidence benefits outweigh harms)
assigned to a wide range of trauma-focused psychotherapies as well as medications in the serotonin reuptake inhibitor and serotonin-norepinephrine reuptake inhibitor classes.15 However, despite a growing list of available pharmacologic choices, new developments in PTSD pharmacotherapy remain elusive. Prazosin, an alpha-1 receptor antagonist, has become widely used in service members and veterans, based on initial clinical trials, but release of findings from a recently completed VA multicenter cooperative clinical trial are still pending.21,22
Pharmacologic enhancement of exposure treatment is an active area of research but has not yet produced sufficient evidence to change treatment recommendations.23 Expensive clinical trials of hyperbaric oxygen for chronic postconcussive symptoms and PTSD in combat veterans have informed clinicians more about the nature of placebo responses than of anything else.24 Benzodiazepines have received D-level recommendations (harm outweighs benefits) in the VA/DoD CPG yet continue to be prescribed relatively frequently to service members and veterans with PTSD.15 Temporary relief of anxiety symptoms is offset by the propensity of benzodiazepines to worsen or impede PTSD recovery through tolerance and dependence, rebound sleep disturbance or anger, and they seem to enhance rather than alleviate fear conditioning.15,16 Risperidone has received a D-level recommendation, based on the results of a large VA cooperative trial, and other atypical antipsychotics carry similar concerns, including metabolic and cardiovascular adverse effects.25
Trauma-Focused Psychotherapy
For mental health professionals working with service members and veterans, the most important clinical strategy supported by strong evidence is to have a firm understanding of the core components of effective trauma-focused psychotherapy and deliver these in a patient-centered manner that fosters continued treatment engagement, which is the most important variable in predicting treatment effectiveness.15-17 The core components are:
- Narration of the traumatic event. This is the single most essential component, besides the underlying therapeutic alliance, and there are many effective ways for therapists to facilitate the narrative process. These include imaginal exposure (as in PE), a linear life narrative approach (as in narrative exposure therapy [NET]), visualization of the traumatic event (as in eye movement desensitization and reprocessing [EMDR]), written narration (shown to be equivalent to the full manualized CPT package in a dismantling study26), facilitation through virtual reality, and inherent narrative processes built into all forms of trauma-focused psychotherapies.15,16
- Cognitive restructuring. This may be delivered in a systematized manner, such as in CPT or EMDR, or more organically, as occurs in PE or NET. Cognitive restructuring, which fundamentally involves gaining a different perspective on the traumatic event(s) and one’s responses (eg, coming to terms with guilt, self-blame, or pervasive mistrust), often emerges naturally during narrative processes.
- In-vivo exposure. This involves graded exposure to activities that trigger symptoms, such as being able to shop at a grocery store during peak hours. Invivo exposure is an inherent component of many trauma-focused treatments, including PE and Stress Inoculation Training. In-vitro techniques that rely on visualization (eg, EMDR) provide another viable delivery method.
- Relaxation exercises. Aimed at addressing physiologic reactivity, these can include diaphragmatic breathing, progressive muscle relaxation, mindfulness, eye movements, and various other approaches, including simply bringing greater awareness to the level of distress through routine
use of a subjective distress measure. - Psychoeducation. This provides an understanding of why PTSD occurs and the underlying physiologic mechanisms and correlates, including the effects of trauma on neuroendocrine and autonomic nervous system regulation, and the strong association of PTSD with physical health effects. Tailoring psychoeducation to military and veteran populations is critical, because of the occupational context in which PTSD can occur and the fact that PTSD symptoms are rooted in skills and responses that are adaptive and beneficial in military occupational and war-zone environments.16
Complementary and alternative medicine approaches also have a role in augmenting evidence-based PTSD treatment, for example, through facilitating relaxation responses, helping with chronic pain or sleep, or facilitating narrative processes through expressive arts.27
New Developments in Psychotherapy
In terms of new developments, 2 trauma-focused psychotherapies deserve particular consideration. The first is NET, which has been shown to be highly effective in severely traumatized populations and has even been delivered by lay counselors with only a few weeks of training.28 Narrative exposure therapy has a solid evidence base, is within the A-level exposure therapy category in the VA/DoD CPG, is simple and straightforward in its approach, easy to learn, and specifically designed for multiple traumas, which can be very relevant to military and veteran populations. However, NET is almost never used by clinicians in DoD and VA facilities. In addition to offering it as a treatment option in DoD and VA mental health clinics, research is needed to explore the potential use of NET to reach veterans who do not engage in traditional care but may be willing to receive services in primary care or even nonmedical settings through peerto-peer models.
Another trauma-focused therapy worth highlighting is Accelerated Resolution Therapy (ART), a form of eye movement therapy with similarities to EMDR, which offers promise in delivering clinically meaningful results within < 6 treatment sessions.29,30 Although ART has been tested in only 1 randomized clinical trial (RCT) to date, it incorporates the core elements of other traumafocused therapies, which have an extensive evidence base.29 Accelerated Resolution Therapy is highly procedural, relatively simple for clinicians to learn, and focused on addressing physiologic and emotional reactivity linked to intrusive traumatic images and memories. It involves imaginal and in-vitro exposure through visualization, relaxation techniques, combined with rescripting traumatic imagery, using techniques similar to those used in cognitive therapies for insomnia and nightmares (eg, Imagery Rehearsal Therapy). All of the procedures are grounded in lateral eye movements.
The 2-arm, DoD-funded RCT involved evaluation of ART for treatment of combat-related PTSD (average 3.7 sessions) against an attention control condition among 57 veterans. Significant differences at 3 months’ follow-up were documented in PTSD, depression, anxiety, trauma-related guilt, sleep quality, and aggression.29 Limitations of this study included reliance on the PCL rather than on a gold-standard diagnostic instrument, lack of independent blinded outcomes, and a control condition similar to a wait list. More rigorous RCTs with active control conditions, as well as dismantling studies, are needed. Nevertheless, the impressive reduction in PCL scores (averaging 17-20 points) after only 3 to 4 sessions and firm grounding in trauma-focused psychotherapy components was sufficient for clinicians at several military treatment facilities (including Walter Reed National Military Medical Center and Fort Belvoir Community Hospital) to become trained in this technique and begin offering it as an option to service members with PTSD. The popularity of this technique is likely to grow if more clinicians test it and confirm that rapid improvements in PCL scores and functioning are possible within a few sessions.
Other Important Clinical Considerations
Ongoing clinical trials are actively looking at compressed delivery of CPT and PE psychotherapy (several sessions per week for 2-3 weeks), which will likely have considerable benefits in reducing patient dropout rates. Another active area of research involves interventions based in primary care, and there is good evidence to support expansion of step- and collaborative-care models in primary care to address postwar health concerns more holistically, particularly with regard to symptoms attributed to mTBI.4,7,24 Finally, considerable literature is emerging on conceptual problems with the overlap between mTBI and PTSD, problems of misattribution of generalized war-related health concerns to mTBI, and potential reinforcement of chronic postwar physical and cognitive symptoms through the current structure of mTBI specialty care.4,7,24 All of this research strongly supports prioritizing interventions based in primary care.
Summary and Conclusion
The most clinically important development in PTSD is the new DSM-5 definition. Clinicians need to be thoroughly aware of the concerns with its clinical utility and have appropriate strategies for dealing with the clinical implications of discordant results (a prominent expert has even called for putting the new definition on hold).11,12 Treatment strategies likely to have the greatest impact on improving effectiveness of treatment are those that emphasize engagement and retention in care and most importantly, delivery of the core components of trauma-focused psychotherapy in a patient-centered manner.16,17 Promising developments in trauma-focused therapy include NET and ART.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government or any of its agencies, or Foundation House, Victoria, Australia. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. United States Census Bureau. Veteran status: 2010 American Community Survey 1-year estimates. http://factfinder2.census.gov/faces/tableservices/jsf/pages/productview.xhtml?pid=ACS_10_1YR_S2101&prodType=table. Accessed February 27, 2015.
2. Kok BC, Herrell RK, Thomas JL, Hoge CW. Posttraumatic stress disorder associated with combat service in Iraq or Afghanistan: Reconciling prevalence differences between studies. J Nerv Ment Dis. 2012;200(5):4.
3. Norris FH, Slone LB. Epidemiology of trauma and PTSD. In: Friedman MJ, Keane TM, Resick PA, eds. Handbook of PTSD: Science and Practice. 2nd ed. New York, NY: The Guilford Press; 2004:100-120.
4. Hoge CW, Castro CA. Treatment of generalized war-related health concerns: Placing TBI and PTSD in context. JAMA. 2014;312(16):1685-1686.
5. Hoge CW, Terhakopian A, Castro CA, Messer SC, Engel CC. Association of posttraumatic stress disorder with somatic symptoms, health care visits, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164(1):150-153.
6. Thomas JL, Wilk JE, Riviere LA, McGurk D, Castro CA, Hoge CW. Prevalence of mental health problems and functional impairment among active component and National Guard soldiers 3 and 12 months following combat in Iraq. Arch Gen Psychiatry. 2010;67(6):614-623.
7. Toblin RL, Riviere LA, Thomas JL, Adler AB, Kok BC, Hoge CW. Grief and physical health outcomes in U.S. soldiers returning from combat. J Affect Disord. 2012;136(3):469-475.
8. Lester P, Peterson K, Reeves J, et al. The long war and parental combat deployment: Effects on military children and at-home spouses. J Am Acad Child Adolesc Psychiatry. 2010;49(4):310-320.
9. Mansfield AJ, Kaufman JS, Marshall SW, Gaynes BN, Morrissey JP, Engel CC. Deployment and the use of mental health services among U.S. Army wives. N Engl J Med. 2010;362(2):101-109.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
11. Hoge CW, Riviere LA, Wilk JE, Herrell RK, Weathers FW. The prevalence of posttraumatic stress disorder (PTSD) in US combat soldiers: A head-to-head comparison of DSM-5 versus DSM-IV-TR symptom criteria with the PTSD checklist. Lancet Psychiatry. 2014;1(4):269-277.
12. McFarlane AC. PTSD and DSM-5: Unintended consequences of change. Lancet Psychiatry. 2014;1(4):246-247.
13. Headquarters, U.S. Department of the Army. Standards of medical fitness. Army Regulation 40–501. Washington, DC: U.S. Department of the Army, 2011.
14. Friedman MJ, Hoge CW. Understanding changes to the posttraumatic stress disorder and acute stress disorder diagnoses in DSM-5. Presented at: Defense Centers of Excellence webinar; May 22, 2014. http://www.dcoe.mil/Libraries/Documents/DCoE-May-2014-PH-Webinar Presentation.pdf. Accessed February 27, 2015.
15. Veterans Health Administration, Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Post-Traumatic Stress, Version 2.0. Washington, DC: Veterans Health Administration and Department of Defense; 2010.
16. Hoge CW. Interventions for war-related posttraumatic stress disorder: Meeting veterans where they are. JAMA. 2011;306(5):549-551.
17. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: Low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
18. Lu MW, Duckart JP, O’Malley JP, Dobscha SK. Correlates of utilization of PTSD specialty treatment among recently diagnosed veterans at the VA. Psychiatr Serv. 2011;62(8):943-949.
19. Spoont MR, Murdoch M, Hodges J, Nugent S. Treatment receipt by veterans after a PTSD diagnosis in PTSD, mental health, or general medical clinics. Psychiatr Serv. 2010;61(1):58-63.
20. Harpaz-Rotem I, Rosenheck RA. Serving those who served: Retention of newly returning veterans from Iraq and Afghanistan in mental health treatment. Psychiatr Serv. 2011;62(1):22-27.
21. Raskind MA, Peterson K, Williams T, et. al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returning from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
22. Department of Veterans Affairs. CSP #563: Prazosin and combat trauma PTSD (PACT). https://clinicaltrials.gov/ct2/show/results/NCT00532493?sect=X70156. Updated July 28, 2014. Accessed February 27, 2015.
23. de Kleine RA, Rothbaum BO, van Minnen A. Pharmacological enhancement of exposure-based treatment in PTSD: A qualitative review. Eur J Psychotraumatol. 2013;4:21626.
24. Hoge CW, Jonas WB. The ritual of hyperbaric oxygen and lessons for the treatment of persistent postconcussion symptoms in military personnel. JAMA Intern Med. 2015;175(1):53-54.
25. Krystal JH, Rosenheck RA, Cramer JA, et al; Veterans Affairs Cooperative Study No. 504 Group. Adjunctive risperidone treatment for antidepressant-resistant symptoms of chronic military service-related PTSD: A randomized trial. JAMA. 2011;306(5):493-502.
26. Resick PA, Galovski TE, O’Brien Uhlmansiek M, Scher CD, Clum GA, Young-Xu Y. A randomized clinical trial to dismantle components of cognitive processing therapy for posttraumatic stress disorder in female victims of interpersonal violence. J Consult Clin Psychol. 2008;76(2):243-258.
27. Hoge CW. Integrating evidence-based treatments for PTSD with CAM practices. Psychiatr Ann. 2013;43(7):338-339.
28. Ertl V, Pfeiffer A, Schauer E, Elbert T, Neuner F. Community-implemented trauma therapy for former child soldiers in Northern Uganda: A randomized controlled trial. JAMA. 2011;306(5):503-512.
29. Kip KE, Rosenzweig L, Hernandez DF, et al. Randomized controlled trial of accelerated resolution therapy (ART) for symptoms of combat-related post-traumatic stress disorder (PTSD). Mil Med. 2013;178(12):1298-1309.
30. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART®). Behav Sci. 2012;2(2):115-134.
The U.S. veteran population of about 22 million is estimated to represent > 9% of U.S. adults.1 Of veterans using health services, a growing proportion have been deployed in Iraq or Afghanistan. The VA and DoD have made it a priority to address the health care needs of service members and veterans affected by these wars, with a particular focus on posttraumatic stress disorder (PTSD) and mild traumatic brain injury (mTBI). This review highlights key developments in assessment and treatment of PTSD.
Epidemiology
Although studies have produced widely varying estimates of the prevalence of PTSD following deployment to Iraq or Afghanistan, more consistent estimates have been obtained by appropriately grouping studies that used a similar methodology.2 Current PTSD prevalence after Iraq and Afghanistan deployments has averaged 5% to 6% in general military population samples that included support personnel from all services (constituting about two-thirds of deployed forces) and 13% in studies that focused on operational infantry units exposed to direct combat. 2
Among Iraq and Afghanistan war veterans who have sought care at VA treatment facilities, > 25% have received a diagnosis of PTSD (Karen H. Seal, MD, written communication, December 2014). Although combat frequency and intensity is the most prevalent predictor of PTSD in this population, other types of trauma, such as sexual assault, can confer a risk as high as direct combat.3 Another strong correlate with postdeployment PTSD has been deployment-related mTBIs (concussions), especially following blast exposure. The most likely mechanism for this seems to be the extreme life-threatening context in which these concussions occur.4
Posttraumatic stress disorder has been linked with a host of comorbid conditions, including depression, anxiety disorders, substance use disorders, physical symptoms, anger, aggression, complicated grief, and risky behaviors.5-7 Deployments also have been shown to have cumulative effects on the psychological health of military spouses and children.8,9
Diagnosis
One of the most important developments in PTSD is the new definition, which was released in May 2013 in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5), replacing the definition that has proved highly useful for clinical, neurobiologic, research, and public health purposes for > 25 years.10 The PTSD definition underwent much more extensive changes in the DSM-5 than did any other common mental disorder affecting adults. Changes included moving this diagnosis out of the anxiety disorders chapter into a separate “Trauma- and Stressor-Related Disorders” chapter, which now includes adjustment disorder.
The A trauma criteria were revised, including removal of criterion A2 (response of “fear, helplessness, or horror”), in part because individuals who train for traumatic events as part of military and first responder occupations often do not report this response. The 3 symptom-criteria clusters were divided into 4 clusters: B (intrusion), C (avoidance), D (negative alternations in cognitions and mood), and E (alternations in arousal and reactivity). Three symptoms were added, bringing the total from 17 to 20, and many symptoms were substantially reworded.
The implications of these revisions are an active area of investigation. However, initial evidence suggests that these changes have not necessarily improved the clinical utility of the definition.11,12 Although research suggests that the 2 criteria sets result in a similar prevalence of PTSD, they do not identify the same individuals, and there is no evidence that clinical accuracy is any greater when using the revised definition.
One head-to-head comparison of symptom criteria between the DSM-5 version of the PTSD Checklist (PCL-5) and the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) version (PCL-S) among 1,822 infantry soldiers showed high discordance.11 Of soldiers who met DSM-IV symptom criteria, 30% did not meet DSM-5 criteria, and an equivalent number only met criteria under DSM-5. This was one of the only studies that maintained independence of the 2 clinical scales (rather than adding DSM-5 symptoms to an existing DSM-IV scale). This study also controlled for the order in which the 2 scales were presented on the survey, which was found to be critically important.11 Furthermore, there was no difference in the overlap with comorbid conditions (eg, major depression, generalized anxiety, alcohol misuse) and no difference in level of functional impairment, suggesting that the new definition has no greater specificity or clinical utility than does the original.
The diagnostic discordance between DSM-IV and DSM-5 was mostly accounted for by the redefinition of C criterion (avoidance) in DSM-5. In an editorial accompanying the comparison study, Alexander C. McFarlane, MD, AO, raised concerns about this decision and the decision to effectively remove the important symptom of numbing of emotions.12 These changes could disenfranchise service members and veterans who learn to override avoidant behaviors through training and who often cope with their reactivity through emotional numbing.12 These various concerns, as well as years of experience
with the original definition and importance of aligning trauma-focused treatment with the most appropriate diagnosis, suggest that clinicians have reasonable justification for continuing to assign the PTSD diagnosis for veterans who meet full criteria for PTSD under the previous DSM-IV definition.
Another clinical concern of the revision is the potential problem with selecting a diagnosis for subthreshold PTSD. Subthreshold PTSD can be broadly defined as PTSD symptoms that do not meet full criteria for diagnosis but impair functioning sufficiently for traumafocused treatment to be indicated. DSM-5 recommends use of adjustment disorder in this circumstance.10 However, in military populations, this definition carries a pejorative connotation (weakness or failure to adapt) and can also lead to administrative separation without medical benefits if the condition has lasted < 6 months.13 Chronic
adjustment disorder is medically compensable under VA and DoD regulations but suffers from poorly defined clinical criteria, in addition to the pejorative connotation. (Chronic adjustment disorder was inadvertently left out of the print version of DSM-5 but corrected online.)
As a result of these concerns, the VA National Center for PTSD has recommended that clinicians use ICD-9 code 309.89 (“other specified trauma- and stressor-related disorder” in DSM-5) instead of adjustment disorder for subthreshold PTSD.14 However, current DoD electronic medical record systems map this code to even more pejorative labels (eg, adjustment disorder with aggression, antisocial behavior, or destructiveness). Thus, until the
VA and DoD adopt ICD-10 coding, which is expected to occur within the next year, clinicians will probably have to continue to use ICD-9 code 300.00 (“unspecified anxiety disorder” or “anxiety disorder not otherwise specified”) for clinically significant subthreshold PTSD.
Clinical Treatment
Although the VA and DoD have codeveloped a PTSD clinical practice guideline (CPG) and are working closely to ensure seamless transition of care, their policy approaches to PTSD treatment have been quite different.15 The VA has placed particular attention on training PTSD specialists in prolonged exposure (PE) and cognitive processing therapy (CPT) and ensuring that all veterans have access to one of these two “formulary,” trauma-focused psychotherapies.16 U.S. Army and DoD policies have emphasized that PTSD treatment should be a core competency for all mental health providers and have interpreted the literature and CPG recommendations broadly, emphasizing the core components found in many different trauma-focused psychotherapies and encouraging use of a wide range of options.16
One of the reasons for DoD’s approach has been the recognition that the most important threat to treatment efficacy is not the fidelity in which manualized treatments are delivered but the willingness of patients to engage in care and their rate of dropping out.16,17 Several studies from VA health care settings have shown that only one-third of Operation Enduring Freedom/Operation Iraqi Freedom veterans diagnosed with PTSD end up receiving a minimally acceptable number of treatment sessions to have reasonable expectation of recovery.18-20 One study suggested that treatment adequacy is somewhat higher in DoD among active-duty soldiers (41%-52%), though still far from optimal.17
For treatment interventions, the VA/DoD CPG remains an authoritative guide, with A-level recommendations (strong evidence benefits outweigh harms)
assigned to a wide range of trauma-focused psychotherapies as well as medications in the serotonin reuptake inhibitor and serotonin-norepinephrine reuptake inhibitor classes.15 However, despite a growing list of available pharmacologic choices, new developments in PTSD pharmacotherapy remain elusive. Prazosin, an alpha-1 receptor antagonist, has become widely used in service members and veterans, based on initial clinical trials, but release of findings from a recently completed VA multicenter cooperative clinical trial are still pending.21,22
Pharmacologic enhancement of exposure treatment is an active area of research but has not yet produced sufficient evidence to change treatment recommendations.23 Expensive clinical trials of hyperbaric oxygen for chronic postconcussive symptoms and PTSD in combat veterans have informed clinicians more about the nature of placebo responses than of anything else.24 Benzodiazepines have received D-level recommendations (harm outweighs benefits) in the VA/DoD CPG yet continue to be prescribed relatively frequently to service members and veterans with PTSD.15 Temporary relief of anxiety symptoms is offset by the propensity of benzodiazepines to worsen or impede PTSD recovery through tolerance and dependence, rebound sleep disturbance or anger, and they seem to enhance rather than alleviate fear conditioning.15,16 Risperidone has received a D-level recommendation, based on the results of a large VA cooperative trial, and other atypical antipsychotics carry similar concerns, including metabolic and cardiovascular adverse effects.25
Trauma-Focused Psychotherapy
For mental health professionals working with service members and veterans, the most important clinical strategy supported by strong evidence is to have a firm understanding of the core components of effective trauma-focused psychotherapy and deliver these in a patient-centered manner that fosters continued treatment engagement, which is the most important variable in predicting treatment effectiveness.15-17 The core components are:
- Narration of the traumatic event. This is the single most essential component, besides the underlying therapeutic alliance, and there are many effective ways for therapists to facilitate the narrative process. These include imaginal exposure (as in PE), a linear life narrative approach (as in narrative exposure therapy [NET]), visualization of the traumatic event (as in eye movement desensitization and reprocessing [EMDR]), written narration (shown to be equivalent to the full manualized CPT package in a dismantling study26), facilitation through virtual reality, and inherent narrative processes built into all forms of trauma-focused psychotherapies.15,16
- Cognitive restructuring. This may be delivered in a systematized manner, such as in CPT or EMDR, or more organically, as occurs in PE or NET. Cognitive restructuring, which fundamentally involves gaining a different perspective on the traumatic event(s) and one’s responses (eg, coming to terms with guilt, self-blame, or pervasive mistrust), often emerges naturally during narrative processes.
- In-vivo exposure. This involves graded exposure to activities that trigger symptoms, such as being able to shop at a grocery store during peak hours. Invivo exposure is an inherent component of many trauma-focused treatments, including PE and Stress Inoculation Training. In-vitro techniques that rely on visualization (eg, EMDR) provide another viable delivery method.
- Relaxation exercises. Aimed at addressing physiologic reactivity, these can include diaphragmatic breathing, progressive muscle relaxation, mindfulness, eye movements, and various other approaches, including simply bringing greater awareness to the level of distress through routine
use of a subjective distress measure. - Psychoeducation. This provides an understanding of why PTSD occurs and the underlying physiologic mechanisms and correlates, including the effects of trauma on neuroendocrine and autonomic nervous system regulation, and the strong association of PTSD with physical health effects. Tailoring psychoeducation to military and veteran populations is critical, because of the occupational context in which PTSD can occur and the fact that PTSD symptoms are rooted in skills and responses that are adaptive and beneficial in military occupational and war-zone environments.16
Complementary and alternative medicine approaches also have a role in augmenting evidence-based PTSD treatment, for example, through facilitating relaxation responses, helping with chronic pain or sleep, or facilitating narrative processes through expressive arts.27
New Developments in Psychotherapy
In terms of new developments, 2 trauma-focused psychotherapies deserve particular consideration. The first is NET, which has been shown to be highly effective in severely traumatized populations and has even been delivered by lay counselors with only a few weeks of training.28 Narrative exposure therapy has a solid evidence base, is within the A-level exposure therapy category in the VA/DoD CPG, is simple and straightforward in its approach, easy to learn, and specifically designed for multiple traumas, which can be very relevant to military and veteran populations. However, NET is almost never used by clinicians in DoD and VA facilities. In addition to offering it as a treatment option in DoD and VA mental health clinics, research is needed to explore the potential use of NET to reach veterans who do not engage in traditional care but may be willing to receive services in primary care or even nonmedical settings through peerto-peer models.
Another trauma-focused therapy worth highlighting is Accelerated Resolution Therapy (ART), a form of eye movement therapy with similarities to EMDR, which offers promise in delivering clinically meaningful results within < 6 treatment sessions.29,30 Although ART has been tested in only 1 randomized clinical trial (RCT) to date, it incorporates the core elements of other traumafocused therapies, which have an extensive evidence base.29 Accelerated Resolution Therapy is highly procedural, relatively simple for clinicians to learn, and focused on addressing physiologic and emotional reactivity linked to intrusive traumatic images and memories. It involves imaginal and in-vitro exposure through visualization, relaxation techniques, combined with rescripting traumatic imagery, using techniques similar to those used in cognitive therapies for insomnia and nightmares (eg, Imagery Rehearsal Therapy). All of the procedures are grounded in lateral eye movements.
The 2-arm, DoD-funded RCT involved evaluation of ART for treatment of combat-related PTSD (average 3.7 sessions) against an attention control condition among 57 veterans. Significant differences at 3 months’ follow-up were documented in PTSD, depression, anxiety, trauma-related guilt, sleep quality, and aggression.29 Limitations of this study included reliance on the PCL rather than on a gold-standard diagnostic instrument, lack of independent blinded outcomes, and a control condition similar to a wait list. More rigorous RCTs with active control conditions, as well as dismantling studies, are needed. Nevertheless, the impressive reduction in PCL scores (averaging 17-20 points) after only 3 to 4 sessions and firm grounding in trauma-focused psychotherapy components was sufficient for clinicians at several military treatment facilities (including Walter Reed National Military Medical Center and Fort Belvoir Community Hospital) to become trained in this technique and begin offering it as an option to service members with PTSD. The popularity of this technique is likely to grow if more clinicians test it and confirm that rapid improvements in PCL scores and functioning are possible within a few sessions.
Other Important Clinical Considerations
Ongoing clinical trials are actively looking at compressed delivery of CPT and PE psychotherapy (several sessions per week for 2-3 weeks), which will likely have considerable benefits in reducing patient dropout rates. Another active area of research involves interventions based in primary care, and there is good evidence to support expansion of step- and collaborative-care models in primary care to address postwar health concerns more holistically, particularly with regard to symptoms attributed to mTBI.4,7,24 Finally, considerable literature is emerging on conceptual problems with the overlap between mTBI and PTSD, problems of misattribution of generalized war-related health concerns to mTBI, and potential reinforcement of chronic postwar physical and cognitive symptoms through the current structure of mTBI specialty care.4,7,24 All of this research strongly supports prioritizing interventions based in primary care.
Summary and Conclusion
The most clinically important development in PTSD is the new DSM-5 definition. Clinicians need to be thoroughly aware of the concerns with its clinical utility and have appropriate strategies for dealing with the clinical implications of discordant results (a prominent expert has even called for putting the new definition on hold).11,12 Treatment strategies likely to have the greatest impact on improving effectiveness of treatment are those that emphasize engagement and retention in care and most importantly, delivery of the core components of trauma-focused psychotherapy in a patient-centered manner.16,17 Promising developments in trauma-focused therapy include NET and ART.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government or any of its agencies, or Foundation House, Victoria, Australia. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
The U.S. veteran population of about 22 million is estimated to represent > 9% of U.S. adults.1 Of veterans using health services, a growing proportion have been deployed in Iraq or Afghanistan. The VA and DoD have made it a priority to address the health care needs of service members and veterans affected by these wars, with a particular focus on posttraumatic stress disorder (PTSD) and mild traumatic brain injury (mTBI). This review highlights key developments in assessment and treatment of PTSD.
Epidemiology
Although studies have produced widely varying estimates of the prevalence of PTSD following deployment to Iraq or Afghanistan, more consistent estimates have been obtained by appropriately grouping studies that used a similar methodology.2 Current PTSD prevalence after Iraq and Afghanistan deployments has averaged 5% to 6% in general military population samples that included support personnel from all services (constituting about two-thirds of deployed forces) and 13% in studies that focused on operational infantry units exposed to direct combat. 2
Among Iraq and Afghanistan war veterans who have sought care at VA treatment facilities, > 25% have received a diagnosis of PTSD (Karen H. Seal, MD, written communication, December 2014). Although combat frequency and intensity is the most prevalent predictor of PTSD in this population, other types of trauma, such as sexual assault, can confer a risk as high as direct combat.3 Another strong correlate with postdeployment PTSD has been deployment-related mTBIs (concussions), especially following blast exposure. The most likely mechanism for this seems to be the extreme life-threatening context in which these concussions occur.4
Posttraumatic stress disorder has been linked with a host of comorbid conditions, including depression, anxiety disorders, substance use disorders, physical symptoms, anger, aggression, complicated grief, and risky behaviors.5-7 Deployments also have been shown to have cumulative effects on the psychological health of military spouses and children.8,9
Diagnosis
One of the most important developments in PTSD is the new definition, which was released in May 2013 in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5), replacing the definition that has proved highly useful for clinical, neurobiologic, research, and public health purposes for > 25 years.10 The PTSD definition underwent much more extensive changes in the DSM-5 than did any other common mental disorder affecting adults. Changes included moving this diagnosis out of the anxiety disorders chapter into a separate “Trauma- and Stressor-Related Disorders” chapter, which now includes adjustment disorder.
The A trauma criteria were revised, including removal of criterion A2 (response of “fear, helplessness, or horror”), in part because individuals who train for traumatic events as part of military and first responder occupations often do not report this response. The 3 symptom-criteria clusters were divided into 4 clusters: B (intrusion), C (avoidance), D (negative alternations in cognitions and mood), and E (alternations in arousal and reactivity). Three symptoms were added, bringing the total from 17 to 20, and many symptoms were substantially reworded.
The implications of these revisions are an active area of investigation. However, initial evidence suggests that these changes have not necessarily improved the clinical utility of the definition.11,12 Although research suggests that the 2 criteria sets result in a similar prevalence of PTSD, they do not identify the same individuals, and there is no evidence that clinical accuracy is any greater when using the revised definition.
One head-to-head comparison of symptom criteria between the DSM-5 version of the PTSD Checklist (PCL-5) and the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) version (PCL-S) among 1,822 infantry soldiers showed high discordance.11 Of soldiers who met DSM-IV symptom criteria, 30% did not meet DSM-5 criteria, and an equivalent number only met criteria under DSM-5. This was one of the only studies that maintained independence of the 2 clinical scales (rather than adding DSM-5 symptoms to an existing DSM-IV scale). This study also controlled for the order in which the 2 scales were presented on the survey, which was found to be critically important.11 Furthermore, there was no difference in the overlap with comorbid conditions (eg, major depression, generalized anxiety, alcohol misuse) and no difference in level of functional impairment, suggesting that the new definition has no greater specificity or clinical utility than does the original.
The diagnostic discordance between DSM-IV and DSM-5 was mostly accounted for by the redefinition of C criterion (avoidance) in DSM-5. In an editorial accompanying the comparison study, Alexander C. McFarlane, MD, AO, raised concerns about this decision and the decision to effectively remove the important symptom of numbing of emotions.12 These changes could disenfranchise service members and veterans who learn to override avoidant behaviors through training and who often cope with their reactivity through emotional numbing.12 These various concerns, as well as years of experience
with the original definition and importance of aligning trauma-focused treatment with the most appropriate diagnosis, suggest that clinicians have reasonable justification for continuing to assign the PTSD diagnosis for veterans who meet full criteria for PTSD under the previous DSM-IV definition.
Another clinical concern of the revision is the potential problem with selecting a diagnosis for subthreshold PTSD. Subthreshold PTSD can be broadly defined as PTSD symptoms that do not meet full criteria for diagnosis but impair functioning sufficiently for traumafocused treatment to be indicated. DSM-5 recommends use of adjustment disorder in this circumstance.10 However, in military populations, this definition carries a pejorative connotation (weakness or failure to adapt) and can also lead to administrative separation without medical benefits if the condition has lasted < 6 months.13 Chronic
adjustment disorder is medically compensable under VA and DoD regulations but suffers from poorly defined clinical criteria, in addition to the pejorative connotation. (Chronic adjustment disorder was inadvertently left out of the print version of DSM-5 but corrected online.)
As a result of these concerns, the VA National Center for PTSD has recommended that clinicians use ICD-9 code 309.89 (“other specified trauma- and stressor-related disorder” in DSM-5) instead of adjustment disorder for subthreshold PTSD.14 However, current DoD electronic medical record systems map this code to even more pejorative labels (eg, adjustment disorder with aggression, antisocial behavior, or destructiveness). Thus, until the
VA and DoD adopt ICD-10 coding, which is expected to occur within the next year, clinicians will probably have to continue to use ICD-9 code 300.00 (“unspecified anxiety disorder” or “anxiety disorder not otherwise specified”) for clinically significant subthreshold PTSD.
Clinical Treatment
Although the VA and DoD have codeveloped a PTSD clinical practice guideline (CPG) and are working closely to ensure seamless transition of care, their policy approaches to PTSD treatment have been quite different.15 The VA has placed particular attention on training PTSD specialists in prolonged exposure (PE) and cognitive processing therapy (CPT) and ensuring that all veterans have access to one of these two “formulary,” trauma-focused psychotherapies.16 U.S. Army and DoD policies have emphasized that PTSD treatment should be a core competency for all mental health providers and have interpreted the literature and CPG recommendations broadly, emphasizing the core components found in many different trauma-focused psychotherapies and encouraging use of a wide range of options.16
One of the reasons for DoD’s approach has been the recognition that the most important threat to treatment efficacy is not the fidelity in which manualized treatments are delivered but the willingness of patients to engage in care and their rate of dropping out.16,17 Several studies from VA health care settings have shown that only one-third of Operation Enduring Freedom/Operation Iraqi Freedom veterans diagnosed with PTSD end up receiving a minimally acceptable number of treatment sessions to have reasonable expectation of recovery.18-20 One study suggested that treatment adequacy is somewhat higher in DoD among active-duty soldiers (41%-52%), though still far from optimal.17
For treatment interventions, the VA/DoD CPG remains an authoritative guide, with A-level recommendations (strong evidence benefits outweigh harms)
assigned to a wide range of trauma-focused psychotherapies as well as medications in the serotonin reuptake inhibitor and serotonin-norepinephrine reuptake inhibitor classes.15 However, despite a growing list of available pharmacologic choices, new developments in PTSD pharmacotherapy remain elusive. Prazosin, an alpha-1 receptor antagonist, has become widely used in service members and veterans, based on initial clinical trials, but release of findings from a recently completed VA multicenter cooperative clinical trial are still pending.21,22
Pharmacologic enhancement of exposure treatment is an active area of research but has not yet produced sufficient evidence to change treatment recommendations.23 Expensive clinical trials of hyperbaric oxygen for chronic postconcussive symptoms and PTSD in combat veterans have informed clinicians more about the nature of placebo responses than of anything else.24 Benzodiazepines have received D-level recommendations (harm outweighs benefits) in the VA/DoD CPG yet continue to be prescribed relatively frequently to service members and veterans with PTSD.15 Temporary relief of anxiety symptoms is offset by the propensity of benzodiazepines to worsen or impede PTSD recovery through tolerance and dependence, rebound sleep disturbance or anger, and they seem to enhance rather than alleviate fear conditioning.15,16 Risperidone has received a D-level recommendation, based on the results of a large VA cooperative trial, and other atypical antipsychotics carry similar concerns, including metabolic and cardiovascular adverse effects.25
Trauma-Focused Psychotherapy
For mental health professionals working with service members and veterans, the most important clinical strategy supported by strong evidence is to have a firm understanding of the core components of effective trauma-focused psychotherapy and deliver these in a patient-centered manner that fosters continued treatment engagement, which is the most important variable in predicting treatment effectiveness.15-17 The core components are:
- Narration of the traumatic event. This is the single most essential component, besides the underlying therapeutic alliance, and there are many effective ways for therapists to facilitate the narrative process. These include imaginal exposure (as in PE), a linear life narrative approach (as in narrative exposure therapy [NET]), visualization of the traumatic event (as in eye movement desensitization and reprocessing [EMDR]), written narration (shown to be equivalent to the full manualized CPT package in a dismantling study26), facilitation through virtual reality, and inherent narrative processes built into all forms of trauma-focused psychotherapies.15,16
- Cognitive restructuring. This may be delivered in a systematized manner, such as in CPT or EMDR, or more organically, as occurs in PE or NET. Cognitive restructuring, which fundamentally involves gaining a different perspective on the traumatic event(s) and one’s responses (eg, coming to terms with guilt, self-blame, or pervasive mistrust), often emerges naturally during narrative processes.
- In-vivo exposure. This involves graded exposure to activities that trigger symptoms, such as being able to shop at a grocery store during peak hours. Invivo exposure is an inherent component of many trauma-focused treatments, including PE and Stress Inoculation Training. In-vitro techniques that rely on visualization (eg, EMDR) provide another viable delivery method.
- Relaxation exercises. Aimed at addressing physiologic reactivity, these can include diaphragmatic breathing, progressive muscle relaxation, mindfulness, eye movements, and various other approaches, including simply bringing greater awareness to the level of distress through routine
use of a subjective distress measure. - Psychoeducation. This provides an understanding of why PTSD occurs and the underlying physiologic mechanisms and correlates, including the effects of trauma on neuroendocrine and autonomic nervous system regulation, and the strong association of PTSD with physical health effects. Tailoring psychoeducation to military and veteran populations is critical, because of the occupational context in which PTSD can occur and the fact that PTSD symptoms are rooted in skills and responses that are adaptive and beneficial in military occupational and war-zone environments.16
Complementary and alternative medicine approaches also have a role in augmenting evidence-based PTSD treatment, for example, through facilitating relaxation responses, helping with chronic pain or sleep, or facilitating narrative processes through expressive arts.27
New Developments in Psychotherapy
In terms of new developments, 2 trauma-focused psychotherapies deserve particular consideration. The first is NET, which has been shown to be highly effective in severely traumatized populations and has even been delivered by lay counselors with only a few weeks of training.28 Narrative exposure therapy has a solid evidence base, is within the A-level exposure therapy category in the VA/DoD CPG, is simple and straightforward in its approach, easy to learn, and specifically designed for multiple traumas, which can be very relevant to military and veteran populations. However, NET is almost never used by clinicians in DoD and VA facilities. In addition to offering it as a treatment option in DoD and VA mental health clinics, research is needed to explore the potential use of NET to reach veterans who do not engage in traditional care but may be willing to receive services in primary care or even nonmedical settings through peerto-peer models.
Another trauma-focused therapy worth highlighting is Accelerated Resolution Therapy (ART), a form of eye movement therapy with similarities to EMDR, which offers promise in delivering clinically meaningful results within < 6 treatment sessions.29,30 Although ART has been tested in only 1 randomized clinical trial (RCT) to date, it incorporates the core elements of other traumafocused therapies, which have an extensive evidence base.29 Accelerated Resolution Therapy is highly procedural, relatively simple for clinicians to learn, and focused on addressing physiologic and emotional reactivity linked to intrusive traumatic images and memories. It involves imaginal and in-vitro exposure through visualization, relaxation techniques, combined with rescripting traumatic imagery, using techniques similar to those used in cognitive therapies for insomnia and nightmares (eg, Imagery Rehearsal Therapy). All of the procedures are grounded in lateral eye movements.
The 2-arm, DoD-funded RCT involved evaluation of ART for treatment of combat-related PTSD (average 3.7 sessions) against an attention control condition among 57 veterans. Significant differences at 3 months’ follow-up were documented in PTSD, depression, anxiety, trauma-related guilt, sleep quality, and aggression.29 Limitations of this study included reliance on the PCL rather than on a gold-standard diagnostic instrument, lack of independent blinded outcomes, and a control condition similar to a wait list. More rigorous RCTs with active control conditions, as well as dismantling studies, are needed. Nevertheless, the impressive reduction in PCL scores (averaging 17-20 points) after only 3 to 4 sessions and firm grounding in trauma-focused psychotherapy components was sufficient for clinicians at several military treatment facilities (including Walter Reed National Military Medical Center and Fort Belvoir Community Hospital) to become trained in this technique and begin offering it as an option to service members with PTSD. The popularity of this technique is likely to grow if more clinicians test it and confirm that rapid improvements in PCL scores and functioning are possible within a few sessions.
Other Important Clinical Considerations
Ongoing clinical trials are actively looking at compressed delivery of CPT and PE psychotherapy (several sessions per week for 2-3 weeks), which will likely have considerable benefits in reducing patient dropout rates. Another active area of research involves interventions based in primary care, and there is good evidence to support expansion of step- and collaborative-care models in primary care to address postwar health concerns more holistically, particularly with regard to symptoms attributed to mTBI.4,7,24 Finally, considerable literature is emerging on conceptual problems with the overlap between mTBI and PTSD, problems of misattribution of generalized war-related health concerns to mTBI, and potential reinforcement of chronic postwar physical and cognitive symptoms through the current structure of mTBI specialty care.4,7,24 All of this research strongly supports prioritizing interventions based in primary care.
Summary and Conclusion
The most clinically important development in PTSD is the new DSM-5 definition. Clinicians need to be thoroughly aware of the concerns with its clinical utility and have appropriate strategies for dealing with the clinical implications of discordant results (a prominent expert has even called for putting the new definition on hold).11,12 Treatment strategies likely to have the greatest impact on improving effectiveness of treatment are those that emphasize engagement and retention in care and most importantly, delivery of the core components of trauma-focused psychotherapy in a patient-centered manner.16,17 Promising developments in trauma-focused therapy include NET and ART.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government or any of its agencies, or Foundation House, Victoria, Australia. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. United States Census Bureau. Veteran status: 2010 American Community Survey 1-year estimates. http://factfinder2.census.gov/faces/tableservices/jsf/pages/productview.xhtml?pid=ACS_10_1YR_S2101&prodType=table. Accessed February 27, 2015.
2. Kok BC, Herrell RK, Thomas JL, Hoge CW. Posttraumatic stress disorder associated with combat service in Iraq or Afghanistan: Reconciling prevalence differences between studies. J Nerv Ment Dis. 2012;200(5):4.
3. Norris FH, Slone LB. Epidemiology of trauma and PTSD. In: Friedman MJ, Keane TM, Resick PA, eds. Handbook of PTSD: Science and Practice. 2nd ed. New York, NY: The Guilford Press; 2004:100-120.
4. Hoge CW, Castro CA. Treatment of generalized war-related health concerns: Placing TBI and PTSD in context. JAMA. 2014;312(16):1685-1686.
5. Hoge CW, Terhakopian A, Castro CA, Messer SC, Engel CC. Association of posttraumatic stress disorder with somatic symptoms, health care visits, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164(1):150-153.
6. Thomas JL, Wilk JE, Riviere LA, McGurk D, Castro CA, Hoge CW. Prevalence of mental health problems and functional impairment among active component and National Guard soldiers 3 and 12 months following combat in Iraq. Arch Gen Psychiatry. 2010;67(6):614-623.
7. Toblin RL, Riviere LA, Thomas JL, Adler AB, Kok BC, Hoge CW. Grief and physical health outcomes in U.S. soldiers returning from combat. J Affect Disord. 2012;136(3):469-475.
8. Lester P, Peterson K, Reeves J, et al. The long war and parental combat deployment: Effects on military children and at-home spouses. J Am Acad Child Adolesc Psychiatry. 2010;49(4):310-320.
9. Mansfield AJ, Kaufman JS, Marshall SW, Gaynes BN, Morrissey JP, Engel CC. Deployment and the use of mental health services among U.S. Army wives. N Engl J Med. 2010;362(2):101-109.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
11. Hoge CW, Riviere LA, Wilk JE, Herrell RK, Weathers FW. The prevalence of posttraumatic stress disorder (PTSD) in US combat soldiers: A head-to-head comparison of DSM-5 versus DSM-IV-TR symptom criteria with the PTSD checklist. Lancet Psychiatry. 2014;1(4):269-277.
12. McFarlane AC. PTSD and DSM-5: Unintended consequences of change. Lancet Psychiatry. 2014;1(4):246-247.
13. Headquarters, U.S. Department of the Army. Standards of medical fitness. Army Regulation 40–501. Washington, DC: U.S. Department of the Army, 2011.
14. Friedman MJ, Hoge CW. Understanding changes to the posttraumatic stress disorder and acute stress disorder diagnoses in DSM-5. Presented at: Defense Centers of Excellence webinar; May 22, 2014. http://www.dcoe.mil/Libraries/Documents/DCoE-May-2014-PH-Webinar Presentation.pdf. Accessed February 27, 2015.
15. Veterans Health Administration, Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Post-Traumatic Stress, Version 2.0. Washington, DC: Veterans Health Administration and Department of Defense; 2010.
16. Hoge CW. Interventions for war-related posttraumatic stress disorder: Meeting veterans where they are. JAMA. 2011;306(5):549-551.
17. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: Low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
18. Lu MW, Duckart JP, O’Malley JP, Dobscha SK. Correlates of utilization of PTSD specialty treatment among recently diagnosed veterans at the VA. Psychiatr Serv. 2011;62(8):943-949.
19. Spoont MR, Murdoch M, Hodges J, Nugent S. Treatment receipt by veterans after a PTSD diagnosis in PTSD, mental health, or general medical clinics. Psychiatr Serv. 2010;61(1):58-63.
20. Harpaz-Rotem I, Rosenheck RA. Serving those who served: Retention of newly returning veterans from Iraq and Afghanistan in mental health treatment. Psychiatr Serv. 2011;62(1):22-27.
21. Raskind MA, Peterson K, Williams T, et. al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returning from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
22. Department of Veterans Affairs. CSP #563: Prazosin and combat trauma PTSD (PACT). https://clinicaltrials.gov/ct2/show/results/NCT00532493?sect=X70156. Updated July 28, 2014. Accessed February 27, 2015.
23. de Kleine RA, Rothbaum BO, van Minnen A. Pharmacological enhancement of exposure-based treatment in PTSD: A qualitative review. Eur J Psychotraumatol. 2013;4:21626.
24. Hoge CW, Jonas WB. The ritual of hyperbaric oxygen and lessons for the treatment of persistent postconcussion symptoms in military personnel. JAMA Intern Med. 2015;175(1):53-54.
25. Krystal JH, Rosenheck RA, Cramer JA, et al; Veterans Affairs Cooperative Study No. 504 Group. Adjunctive risperidone treatment for antidepressant-resistant symptoms of chronic military service-related PTSD: A randomized trial. JAMA. 2011;306(5):493-502.
26. Resick PA, Galovski TE, O’Brien Uhlmansiek M, Scher CD, Clum GA, Young-Xu Y. A randomized clinical trial to dismantle components of cognitive processing therapy for posttraumatic stress disorder in female victims of interpersonal violence. J Consult Clin Psychol. 2008;76(2):243-258.
27. Hoge CW. Integrating evidence-based treatments for PTSD with CAM practices. Psychiatr Ann. 2013;43(7):338-339.
28. Ertl V, Pfeiffer A, Schauer E, Elbert T, Neuner F. Community-implemented trauma therapy for former child soldiers in Northern Uganda: A randomized controlled trial. JAMA. 2011;306(5):503-512.
29. Kip KE, Rosenzweig L, Hernandez DF, et al. Randomized controlled trial of accelerated resolution therapy (ART) for symptoms of combat-related post-traumatic stress disorder (PTSD). Mil Med. 2013;178(12):1298-1309.
30. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART®). Behav Sci. 2012;2(2):115-134.
1. United States Census Bureau. Veteran status: 2010 American Community Survey 1-year estimates. http://factfinder2.census.gov/faces/tableservices/jsf/pages/productview.xhtml?pid=ACS_10_1YR_S2101&prodType=table. Accessed February 27, 2015.
2. Kok BC, Herrell RK, Thomas JL, Hoge CW. Posttraumatic stress disorder associated with combat service in Iraq or Afghanistan: Reconciling prevalence differences between studies. J Nerv Ment Dis. 2012;200(5):4.
3. Norris FH, Slone LB. Epidemiology of trauma and PTSD. In: Friedman MJ, Keane TM, Resick PA, eds. Handbook of PTSD: Science and Practice. 2nd ed. New York, NY: The Guilford Press; 2004:100-120.
4. Hoge CW, Castro CA. Treatment of generalized war-related health concerns: Placing TBI and PTSD in context. JAMA. 2014;312(16):1685-1686.
5. Hoge CW, Terhakopian A, Castro CA, Messer SC, Engel CC. Association of posttraumatic stress disorder with somatic symptoms, health care visits, and absenteeism among Iraq war veterans. Am J Psychiatry. 2007;164(1):150-153.
6. Thomas JL, Wilk JE, Riviere LA, McGurk D, Castro CA, Hoge CW. Prevalence of mental health problems and functional impairment among active component and National Guard soldiers 3 and 12 months following combat in Iraq. Arch Gen Psychiatry. 2010;67(6):614-623.
7. Toblin RL, Riviere LA, Thomas JL, Adler AB, Kok BC, Hoge CW. Grief and physical health outcomes in U.S. soldiers returning from combat. J Affect Disord. 2012;136(3):469-475.
8. Lester P, Peterson K, Reeves J, et al. The long war and parental combat deployment: Effects on military children and at-home spouses. J Am Acad Child Adolesc Psychiatry. 2010;49(4):310-320.
9. Mansfield AJ, Kaufman JS, Marshall SW, Gaynes BN, Morrissey JP, Engel CC. Deployment and the use of mental health services among U.S. Army wives. N Engl J Med. 2010;362(2):101-109.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
11. Hoge CW, Riviere LA, Wilk JE, Herrell RK, Weathers FW. The prevalence of posttraumatic stress disorder (PTSD) in US combat soldiers: A head-to-head comparison of DSM-5 versus DSM-IV-TR symptom criteria with the PTSD checklist. Lancet Psychiatry. 2014;1(4):269-277.
12. McFarlane AC. PTSD and DSM-5: Unintended consequences of change. Lancet Psychiatry. 2014;1(4):246-247.
13. Headquarters, U.S. Department of the Army. Standards of medical fitness. Army Regulation 40–501. Washington, DC: U.S. Department of the Army, 2011.
14. Friedman MJ, Hoge CW. Understanding changes to the posttraumatic stress disorder and acute stress disorder diagnoses in DSM-5. Presented at: Defense Centers of Excellence webinar; May 22, 2014. http://www.dcoe.mil/Libraries/Documents/DCoE-May-2014-PH-Webinar Presentation.pdf. Accessed February 27, 2015.
15. Veterans Health Administration, Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Post-Traumatic Stress, Version 2.0. Washington, DC: Veterans Health Administration and Department of Defense; 2010.
16. Hoge CW. Interventions for war-related posttraumatic stress disorder: Meeting veterans where they are. JAMA. 2011;306(5):549-551.
17. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: Low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
18. Lu MW, Duckart JP, O’Malley JP, Dobscha SK. Correlates of utilization of PTSD specialty treatment among recently diagnosed veterans at the VA. Psychiatr Serv. 2011;62(8):943-949.
19. Spoont MR, Murdoch M, Hodges J, Nugent S. Treatment receipt by veterans after a PTSD diagnosis in PTSD, mental health, or general medical clinics. Psychiatr Serv. 2010;61(1):58-63.
20. Harpaz-Rotem I, Rosenheck RA. Serving those who served: Retention of newly returning veterans from Iraq and Afghanistan in mental health treatment. Psychiatr Serv. 2011;62(1):22-27.
21. Raskind MA, Peterson K, Williams T, et. al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returning from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
22. Department of Veterans Affairs. CSP #563: Prazosin and combat trauma PTSD (PACT). https://clinicaltrials.gov/ct2/show/results/NCT00532493?sect=X70156. Updated July 28, 2014. Accessed February 27, 2015.
23. de Kleine RA, Rothbaum BO, van Minnen A. Pharmacological enhancement of exposure-based treatment in PTSD: A qualitative review. Eur J Psychotraumatol. 2013;4:21626.
24. Hoge CW, Jonas WB. The ritual of hyperbaric oxygen and lessons for the treatment of persistent postconcussion symptoms in military personnel. JAMA Intern Med. 2015;175(1):53-54.
25. Krystal JH, Rosenheck RA, Cramer JA, et al; Veterans Affairs Cooperative Study No. 504 Group. Adjunctive risperidone treatment for antidepressant-resistant symptoms of chronic military service-related PTSD: A randomized trial. JAMA. 2011;306(5):493-502.
26. Resick PA, Galovski TE, O’Brien Uhlmansiek M, Scher CD, Clum GA, Young-Xu Y. A randomized clinical trial to dismantle components of cognitive processing therapy for posttraumatic stress disorder in female victims of interpersonal violence. J Consult Clin Psychol. 2008;76(2):243-258.
27. Hoge CW. Integrating evidence-based treatments for PTSD with CAM practices. Psychiatr Ann. 2013;43(7):338-339.
28. Ertl V, Pfeiffer A, Schauer E, Elbert T, Neuner F. Community-implemented trauma therapy for former child soldiers in Northern Uganda: A randomized controlled trial. JAMA. 2011;306(5):503-512.
29. Kip KE, Rosenzweig L, Hernandez DF, et al. Randomized controlled trial of accelerated resolution therapy (ART) for symptoms of combat-related post-traumatic stress disorder (PTSD). Mil Med. 2013;178(12):1298-1309.
30. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART®). Behav Sci. 2012;2(2):115-134.
Hospitalization Risk With Benzodiazepine and Opioid Use in Veterans With Posttraumatic Stress Disorder (FULL)
Posttraumatic stress disorder (PTSD) is a mental health condition that may develop in response to a traumatic event, such as that experienced by a soldier during active combat duty. In 2009, more than 495,000 veterans within the VA health care system were treated for PTSD—nearly triple the number a decade earlier.1 Core symptoms of PTSD include alterations in arousal and reactivity, avoidant behaviors, negative alterations in mood and cognition, and intrusive thoughts and nightmares. All of the symptoms can be debilitating. First-line pharmacotherapy options that target these core symptoms include selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs).2
The anxiolytic and sedative effects of benzodiazepines may provide quick relief from many of the secondary symptoms of PTSD, including sleep disturbances, irritability, and panic attacks. However, benzodiazepines potentially interfere with the extinction of conditioned fear—a goal integral to certain types of psychotherapy, such as exposure therapy.3,4 In addition, the systematic review and meta-analysis by Guina and colleagues revealed that benzodiazepines are ineffective in the treatment of PTSD.5 The majority of the evaluated studies that used PTSD-specific measures (eg, Clinician-Administered PTSD Scale [CAPS]) found increased PTSD severity and worse prognosis with use of these medications.5 In 2010, the VA and the DoD released a joint guideline for PTSD management.2 According to the guideline, benzodiazepines cause harm when used in PTSD and are relatively contraindicated in combat veterans because of the higher incidence of comorbid substance use disorders (SUDs) in these veterans relative to the general population.2,6
Opioid use also has been linked to poor functional and clinical outcomes in veterans with PTSD. Among patients being treated for opioid use disorder, those with PTSD were less likely to endorse employment as a main source of income and had a higher incidence of recent attempted suicide.7 In a large retrospective cohort study, Operation Iraqi Freedom and Operation Enduring Freedom veterans with PTSD who were prescribed opioids were more likely to present to the emergency department (ED) and to be hospitalized for overdoses and injuries.8
Despite the risks of benzodiazepine and opioid use in this patient population, these medications are still often prescribed to veterans with PTSD for symptomatic relief. In fiscal year 2009, across the VHA system 37% of veterans diagnosed with PTSD were prescribed a benzodiazepine, 69% of the time by a mental health provider.9 Among Iraq and Afghanistan veterans, those with PTSD were significantly more likely to be prescribed an opioid for diagnosed pain—relative to those with a mental health disorder other than PTSD and those without a mental health disorder.8 Thus, there seems to be a disconnect between guideline recommendations and current practice.
The authors conducted a study to assess the potential risk of hospitalization for veterans with PTSD prescribed first-line pharmacotherapy and those also prescribed concurrent benzodiazepine and/or opioid therapy since the release of the PTSD guideline in 2010.2
Methods
In this retrospective cohort study, conducted at the Southern Arizona VA Health Care System (SAVAHCS), the authors analyzed electronic medical record data from November 1, 2009 to August 1, 2015. Study inclusion criteria were veteran, aged 18 to 89 years, diagnosis of PTSD (International Classification of Diseases, Ninth Revision, Clinical Modification code 309.81), and SSRI or SNRI newly prescribed between November 1, 2010 and August 1, 2013.
Any veteran prescribed at least one 30-day or longer supply of any benzodiazepine or opioid within 1 year before the SSRI/SNRI initial prescription date was excluded from the study. Also excluded was any patient treated for PTSD at a facility outside SAVAHCS or whose 2-year evaluation period extended past August 1, 2015.
Study Groups
An outpatient prescription was determined to be the initial SSRI/SNRI prescription for a patient who received less than a 30-day cumulative supply of any SSRI or SNRI within 1 year before that prescription date. Citalopram, desvenlafaxine, duloxetine, escitalopram, fluoxetine, fluvoxamine, levomilnacipran, milnacipran, paroxetine, sertraline, venlafaxine, vilazodone, and vortioxetine were the prespecified SSRI/SNRIs included in the study.
Patients who received at least 1 outpatient prescription for any benzodiazepine (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and benzodiazepine therapy. Alprazolam, chlordiazepoxide, clonazepam, clorazepate, diazepam, estazolam, flurazepam, lorazepam, oxazepam, temazepam, and triazolam were the prespecified benzodiazepines included in the study.
Patients who received at least 1 outpatient prescription for any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and opioid therapy. Codeine, fentanyl, hydrocodone, hydromorphone, levorphanol, meperidine, methadone, morphine, oxymorphone, pentazocine, propoxyphene, and tramadol were the prespecified opioids included in this study.
Patients who received at least 1 outpatient prescription for any benzodiazepine and any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI, benzodiazepine, and opioid therapy.
The index date was defined as the first date of prescription overlap. If there was no benzodiazepine or opioid prescription within 1 year after the initial SSRI/SNRI prescription date, the patient was categorized as being on SSRI/SNRI monotherapy, and the index date was the date of the initial SSRI/SNRI prescription. For each patient, hospitalization data from the 2-year period after the index date were evaluated.
Outcomes and Data Collection
For evaluation of the primary outcome (2-year overall hospitalization risk), the number of unique mental health and medical/surgical hospitalizations was identified by the number of discharge summaries documented in the patient chart during the evaluation period. Time to first hospitalization was recorded for the survival data analysis. Secondary outcomes were mental health hospitalization risk, medical/surgical hospitalization risk, and all-cause mortality within 2 years.
Demographic data that were collected included age, sex, comorbid mental health disorders, comorbid SUDs, and concomitant use of psychotropic medications at index date (baseline). Select comorbid mental health disorders (anxiety, schizophrenia, depression, bipolar disorder) and substance use disorders (alcohol, opioid, illicit drug) also were identified. Data on insomnia and pain comorbidities (headaches or migraines; neuropathy; head, neck, back, arthritis, or joint pain) were collected, as these comorbidities could be indications for prescribing benzodiazepines and opioids. Concomitant baseline use of classes of psychotropic medications (antipsychotics, non-SSRI/SNRI antidepressants, mood stabilizers, anxiolytics, nonbenzodiazepine sedatives/hypnotics) also were documented. Last, hospitalizations within 6 months before the initial SSRI/SNRI prescription date were noted.
Statistical Analysis
Descriptive statistics were used to analyze all baseline demographic data. Continuous measures were evaluated with 1-way analyses of variance and post hoc Bonferroni-corrected pairwise comparisons, and categorical measures with contingency tables and χ2 tests or Fisher exact tests. When the overall χ2 test was significant across all 4 study groups, post hoc comparisons were performed between the SSRI/SNRI monotherapy group and each other group with Bonferroni adjusted for 3 comparisons.
Unadjusted and adjusted Weibull proportional hazard regression models were used to estimate hospitalization risk within 2 years after the index date for the different study groups with the SSRI/SNRI monotherapy group as the referent. Robust standard errors were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs). The Weibull model (and not the Cox model) was used because it does not assume hazard remains constant over time, which is appropriate in this instance, as the risk of an adverse event (AE) may be higher when first starting a medication or combination of medications relative to when doses are stabilized. Models were adjusted for age, sex, baseline mental health disorders, and baseline psychotropic medications. As earlier hospitalizations showed evidence of effect modification when this covariate was tested, hazard analyses were limited to patients not previously hospitalized.
The effect size of differences in hospitalization risk meeting statistical significance was assessed by estimating the number needed to harm (NNH) and 95% CIs (not shown) to observe 1 additional hospitalization in each medication group relative to the SSRI/SNRI monotherapy group over a 90-day period. A 95% CI for NNH that did not include 0 indicated the NNH was significant at the .05 level.10 All-cause mortality was evaluated with the Fisher exact test with post hoc Bonferroni-corrected comparisons as appropriate.
Results
Of 1,703 patients screened, 613 met all study inclusion criteria (Figure 1).
Baseline characteristics revealed no significant differences between groups in age or comorbid depression, schizophrenia, or SUDs (Table 1).
With the SSRI/SNRI monotherapy group as the referent, all concurrent therapy groups were at significantly increased risk for overall hospitalization within 2 years after the index date (Tables 2 & 3, Figure 2).
Risk for mental health hospitalization was significantly increased in all concurrent therapy groups relative to the referent group.
Although the risk for medical/surgical hospitalization was not significantly increased in the SSRI/SNRI and benzodiazepine therapy group (AHR, 1.9; 95% CI, 0.67-5.6), a significant difference was found in the SSRI/SNRI and opioid therapy group (AHR, 4.4; 95% CI, 1.6-12.0; NNH, 42).
Discussion
In 2013, Hawkins and colleagues evaluated hospitalization risk in veterans treated for PTSD within the Northwest VISN 20 between 2004 and 2010.11 Compared with patients treated with only an SSRI or SNRI, those treated with 1 of those medications and a benzodiazepine were at significantly higher risk for overall hospitalization (AHR, 1.79; 95% CI, 1.38-2.32; P < .001) and mental health hospitalization (AHR, 1.87; 95% CI, 1.37-2.53; P < .001). Furthermore, those prescribed a benzodiazepine and an opioid along with an SSRI or SNRI were at higher risk for overall hospitalization (AHR, 2.98; 95% CI, 2.22-4.00; P < .001), mental health hospitalization (AHR, 2.00; 95% CI, 1.35-2.98; P < .01), medical/surgical hospitalization (AHR, 4.86; 95% CI, 3.30-7.14; P < .001), and ED visits (AHR, 2.01; 95% CI, 1.53-2.65; P < .001).
Findings from the present study, which covered a period after the newest PTSD guideline was released,support findings reported by Hawkins and colleagues in their retrospective cohort study covering an earlier period.2,11 In the present study, compared with the monotherapy group, the SSRI/SNRI and benzodiazepine therapy group and the SSRI/SNRI, benzodiazepine, and opioid therapy group were at higher risk for both overall hospitalization and mental health hospitalization within 2 years. However, in a subset of PTSD patients prescribed opioids along with first-line pharmacotherapy, this study found that overall, mental health, and medical/surgical hospitalizations were significantly increased as well. Furthermore, this study found 2-year mortality was significantly higher for the SSRI/SNRI, benzodiazepine, and opioid therapy group than for the SSRI/SNRI monotherapy group.
Adjusted hazard ratios were higher in the present study than those in the study by Hawkins and colleagues,but CIs were wider as well.11 These differences may be attributable to the relatively smaller sample size of the present study and may explain why the HR was higher for the SSRI/SNRI and opioid therapy group than for the SSRI/SNRI, benzodiazepine, and opioid therapy group.
Nevertheless, these results support the growing body of evidence establishing the many risks for AEs when benzodiazepines and opioids are prescribed in the setting of PTSD. Unfortunately, it seems that, against clear guideline recommendations and literature findings, these medications still are being prescribed to this vulnerable, high-risk population.
In the last few months of 2013, the VA health care system launched 2 important medication safety initiatives. The Psychotropic Drug Safety Initiative (PDSI) was established as a quality improvement initiative for evidence-based provision of psychotropic medications. One PDSI metric in particular focused on reducing the proportion of veterans with PTSD being treated with benzodiazepines. The Opioid Safety Initiative (OSI) came as a response to a dramatic increase in the number of fatal overdoses related to prescription opioids—an increase linked to an unprecedented jump in opioid use for nonmalignant pain. As the present study’s inclusion cutoff date of August 1, 2013, preceded the debut of both PDSI and OSI, the benzodiazepine and opioid prescription rates reported here might be higher than those currently being found under the 2 initiatives.
Limitations
This study had several limitations that might affect the interpretation or generalizability of findings. Requiring at least a 30-day supply for prescription eligibility was an attempt to focus on chronic use of medications rather than on, for example, onetime supplies of opioids for dental procedures. However, prescription fill history was not assessed. Therefore, patients could have been included in certain study groups even if their SSRI, SNRI, benzodiazepine, or opioid prescription was not refilled. Furthermore, only VA medical records were used; non-VA prescriptions were not captured.
In addition, this study was limited to patients who at bare minimum were prescribed an SSRI or an SNRI. Some patients may have been prescribed a benzodiazepine and/or an opioid but were not on appropriate first-line pharmacotherapy for PTSD. These patients were excluded from the study, and their relative hospitalization risk went unexplored. Therefore, the magnitude of the issue at hand might have been underestimated.
Although psychotherapy is a first-line treatment option for PTSD, the study did not assess the potential impact of psychotherapy on outcomes or the groups’ relative proportions of patients undergoing psychotherapy. It is unknown whether the groups were equivalent at baseline in regards to psychotherapy participation rates.
This study did not characterize the specific reasons for hospitalization beyond whether it was for a mental health or a medical/surgical issue; thus, no distinction was made between hospitalizations for an elective procedure and hospitalizations for a drug overdose or an injury. Investigators could characterize admission diagnoses to better assess whether hospitalizations are truly associated with study medications or whether patients are being hospitalized for unrelated reasons. In addition, they could elucidate the true nature of hospitalization risk associated with SSRI/SNRI, benzodiazepine, and opioid use by comparing admission diagnoses made before and after initiation of these pharmacologic therapies.
This study also could not assess outcomes for patients who presented to the ED but were not admitted. If the hospital’s floor and ED beds were at full capacity, some patients might have been transferred to an outside facility. However, this scenario is not common at SAVAHCS, where the study was conducted.
Although some comorbid conditions were noted, the study did not evaluate whether its patients had a compelling indication for benzodiazepines in particular. Opioid use is very limited to the treatment of pain, and the majority of the patients on opioid therapy in this study had a diagnosed pain syndrome.
Because of the study’s sample size and power limitations, patients were eligible to be included in a concurrent therapy group if a benzodiazepine, an opioid, or both were added no later than 1 year after SSRI/SNRI initiation. This gap of up to 1 year might have introduced some variability in exposure to risk from earlier prescribed medications. However, sensitivity analyses were performed with multiple constructed Weibull models of time to hospitalization based on subsets with varying overlapping medication gaps. Analyses revealed relatively stable HRs, suggesting that potential bias did not occur.
Future Directions
Investigators could explore the higher all-cause mortality rates in the SSRI/SNRI, benzodiazepine, and opioid therapy group, as this study did not assess cause of death in these patients. Whether any patients died of reasons directly attributable to benzodiazepines or opioids is unknown.
That SSRIs and SNRIs are the only established first-line pharmacologic treatment options for PTSD symptoms partly accounts for the widespread use of benzodiazepines in this population. For that reason, beyond characterizing the many risks associated with using benzodiazepines to manage these symptoms, there is a huge need to research the viability of other pharmacologic agents in treating PTSD. This is especially important given the slower onset to efficacy of the SSRIs and SNRIs; per estimates, only up to 60% of patients respond to SSRIs, and 20% to 30% achieve full remission of PTSD.12 Furthermore, these rates likely are even lower for combat veterans than those for the general population. Several trials discussed in a 2009 guideline review of the treatment of patients with acute stress disorder and PTSD have called into question the efficacy of SSRIs for combat-related PTSD.13 In these randomized, controlled trials, change in PTSD symptom severity as measured with CAPS was not significantly reduced with SSRIs compared with placebo.
A systematic review revealed that, of the nonantidepressants used as adjuncts in treating patients who do not achieve remission with SSRIs, the atypical antipsychotic risperidone may have the strongest supporting evidence.12 However, the present study found high rates of antipsychotic use in the SSRI/SNRI, benzodiazepine, and opioid therapy group, which also had the highest all-cause mortality rate. The safety of risperidone as an alternative treatment needs further evaluation.
Some prospective studies have suggested that the α1 blockers doxazosin and prazosin, the latter of which is commonly used for PTSD nightmares, also may improve PTSD symptoms as assessed by CAPS.14,15 Although these results are promising, the trials to date have been conducted with relatively small sample sizes.
With more veterans being treated for PTSD within the VA health care system, the central treatment goal remains: Adequately address the symptoms of PTSD while minimizing the harm caused by medications. Prescribers should limit benzodiazepine and opioid use in this population and consider safer nonpharmacologic and pharmacologic treatment options when possible.
Conclusion
Combat veterans with PTSD who are prescribed benzodiazepines and/or opioids in addition to first-line pharmacotherapy are at significantly increased risk for overall and mental health hospitalization.
Click here to read the digital edition.
1. Bernardy NC, Lund BC, Alexander B, Jenkyn AB, Schnurr PP, Friedman MJ. Gender differences in prescribing among veterans diagnosed with posttraumatic stress disorder. J Gen Intern Med. 2013;28(suppl 2):S542-S548.
2. Management of Post-Traumatic Stress Working Group, Department of Veterans Affairs, Department of Defense. VA/DoD Clinical Practice Guideline for Management of Post-Traumatic Stress. http://www.healthquality.va.gov/PTSD-full-2010c .pdf. Published October 2010. Accessed July 12, 2015.
3. Marks IM, Swinson RP, Baso˘glu M, et al. Alprazolam and exposure alone and combined in panic disorder with agoraphobia. A controlled study in London and Toronto. Br J Psychiatry. 1993;162:776-787.
4. Wilhelm FH, Roth WT. Acute and delayed effects of alprazolam on flight phobics during exposure. Behav Res Ther. 1997;35(9):831-841.
5. Guina J, Rossetter SR, DeRhodes BJ, Nahhas RW, Welton RS. Benzodiazepines for PTSD: a systematic review and meta-analysis. J Psychiatr Pract. 2015;21(4):281-303.
6. Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Prevalence and Axis I comorbidity of full and partial posttraumatic stress disorder in the United States: results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. J Anxiety Disord. 2011;25(3):456-465.
7. Mills KL, Teesson M, Ross J, Darke S, Shanahan M. The costs and outcomes of treatment for opioid dependence associated with posttraumatic stress disorder. Psychiatr Serv. 2005;56(8):940-945.
8. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
9. Abrams TE, Lund BC, Bernardy NC, Friedman MJ. Aligning clinical practice to PTSD treatment guidelines: medication prescribing by provider type. Psychiatr Serv. 2013;64(2):142-148.
10. Altman DG, Andersen PK. Calculating the number needed to treat for trials where the outcome is time to an event. BMJ. 1999;319(7223):1492-1495.
11. Hawkins EJ, Malte CA, Grossbard J, Saxon AJ, Imel ZE, Kivlahan DR. Comparative safety of benzodiazepines and opioids among Veterans Affairs patients with posttraumatic stress disorder. J Addict Med. 2013;7(5):354-362.
12. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
13. Benedek DM, Friedman MJ, Zatzick D, Ursano RJ. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
14. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
15. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
Posttraumatic stress disorder (PTSD) is a mental health condition that may develop in response to a traumatic event, such as that experienced by a soldier during active combat duty. In 2009, more than 495,000 veterans within the VA health care system were treated for PTSD—nearly triple the number a decade earlier.1 Core symptoms of PTSD include alterations in arousal and reactivity, avoidant behaviors, negative alterations in mood and cognition, and intrusive thoughts and nightmares. All of the symptoms can be debilitating. First-line pharmacotherapy options that target these core symptoms include selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs).2
The anxiolytic and sedative effects of benzodiazepines may provide quick relief from many of the secondary symptoms of PTSD, including sleep disturbances, irritability, and panic attacks. However, benzodiazepines potentially interfere with the extinction of conditioned fear—a goal integral to certain types of psychotherapy, such as exposure therapy.3,4 In addition, the systematic review and meta-analysis by Guina and colleagues revealed that benzodiazepines are ineffective in the treatment of PTSD.5 The majority of the evaluated studies that used PTSD-specific measures (eg, Clinician-Administered PTSD Scale [CAPS]) found increased PTSD severity and worse prognosis with use of these medications.5 In 2010, the VA and the DoD released a joint guideline for PTSD management.2 According to the guideline, benzodiazepines cause harm when used in PTSD and are relatively contraindicated in combat veterans because of the higher incidence of comorbid substance use disorders (SUDs) in these veterans relative to the general population.2,6
Opioid use also has been linked to poor functional and clinical outcomes in veterans with PTSD. Among patients being treated for opioid use disorder, those with PTSD were less likely to endorse employment as a main source of income and had a higher incidence of recent attempted suicide.7 In a large retrospective cohort study, Operation Iraqi Freedom and Operation Enduring Freedom veterans with PTSD who were prescribed opioids were more likely to present to the emergency department (ED) and to be hospitalized for overdoses and injuries.8
Despite the risks of benzodiazepine and opioid use in this patient population, these medications are still often prescribed to veterans with PTSD for symptomatic relief. In fiscal year 2009, across the VHA system 37% of veterans diagnosed with PTSD were prescribed a benzodiazepine, 69% of the time by a mental health provider.9 Among Iraq and Afghanistan veterans, those with PTSD were significantly more likely to be prescribed an opioid for diagnosed pain—relative to those with a mental health disorder other than PTSD and those without a mental health disorder.8 Thus, there seems to be a disconnect between guideline recommendations and current practice.
The authors conducted a study to assess the potential risk of hospitalization for veterans with PTSD prescribed first-line pharmacotherapy and those also prescribed concurrent benzodiazepine and/or opioid therapy since the release of the PTSD guideline in 2010.2
Methods
In this retrospective cohort study, conducted at the Southern Arizona VA Health Care System (SAVAHCS), the authors analyzed electronic medical record data from November 1, 2009 to August 1, 2015. Study inclusion criteria were veteran, aged 18 to 89 years, diagnosis of PTSD (International Classification of Diseases, Ninth Revision, Clinical Modification code 309.81), and SSRI or SNRI newly prescribed between November 1, 2010 and August 1, 2013.
Any veteran prescribed at least one 30-day or longer supply of any benzodiazepine or opioid within 1 year before the SSRI/SNRI initial prescription date was excluded from the study. Also excluded was any patient treated for PTSD at a facility outside SAVAHCS or whose 2-year evaluation period extended past August 1, 2015.
Study Groups
An outpatient prescription was determined to be the initial SSRI/SNRI prescription for a patient who received less than a 30-day cumulative supply of any SSRI or SNRI within 1 year before that prescription date. Citalopram, desvenlafaxine, duloxetine, escitalopram, fluoxetine, fluvoxamine, levomilnacipran, milnacipran, paroxetine, sertraline, venlafaxine, vilazodone, and vortioxetine were the prespecified SSRI/SNRIs included in the study.
Patients who received at least 1 outpatient prescription for any benzodiazepine (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and benzodiazepine therapy. Alprazolam, chlordiazepoxide, clonazepam, clorazepate, diazepam, estazolam, flurazepam, lorazepam, oxazepam, temazepam, and triazolam were the prespecified benzodiazepines included in the study.
Patients who received at least 1 outpatient prescription for any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and opioid therapy. Codeine, fentanyl, hydrocodone, hydromorphone, levorphanol, meperidine, methadone, morphine, oxymorphone, pentazocine, propoxyphene, and tramadol were the prespecified opioids included in this study.
Patients who received at least 1 outpatient prescription for any benzodiazepine and any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI, benzodiazepine, and opioid therapy.
The index date was defined as the first date of prescription overlap. If there was no benzodiazepine or opioid prescription within 1 year after the initial SSRI/SNRI prescription date, the patient was categorized as being on SSRI/SNRI monotherapy, and the index date was the date of the initial SSRI/SNRI prescription. For each patient, hospitalization data from the 2-year period after the index date were evaluated.
Outcomes and Data Collection
For evaluation of the primary outcome (2-year overall hospitalization risk), the number of unique mental health and medical/surgical hospitalizations was identified by the number of discharge summaries documented in the patient chart during the evaluation period. Time to first hospitalization was recorded for the survival data analysis. Secondary outcomes were mental health hospitalization risk, medical/surgical hospitalization risk, and all-cause mortality within 2 years.
Demographic data that were collected included age, sex, comorbid mental health disorders, comorbid SUDs, and concomitant use of psychotropic medications at index date (baseline). Select comorbid mental health disorders (anxiety, schizophrenia, depression, bipolar disorder) and substance use disorders (alcohol, opioid, illicit drug) also were identified. Data on insomnia and pain comorbidities (headaches or migraines; neuropathy; head, neck, back, arthritis, or joint pain) were collected, as these comorbidities could be indications for prescribing benzodiazepines and opioids. Concomitant baseline use of classes of psychotropic medications (antipsychotics, non-SSRI/SNRI antidepressants, mood stabilizers, anxiolytics, nonbenzodiazepine sedatives/hypnotics) also were documented. Last, hospitalizations within 6 months before the initial SSRI/SNRI prescription date were noted.
Statistical Analysis
Descriptive statistics were used to analyze all baseline demographic data. Continuous measures were evaluated with 1-way analyses of variance and post hoc Bonferroni-corrected pairwise comparisons, and categorical measures with contingency tables and χ2 tests or Fisher exact tests. When the overall χ2 test was significant across all 4 study groups, post hoc comparisons were performed between the SSRI/SNRI monotherapy group and each other group with Bonferroni adjusted for 3 comparisons.
Unadjusted and adjusted Weibull proportional hazard regression models were used to estimate hospitalization risk within 2 years after the index date for the different study groups with the SSRI/SNRI monotherapy group as the referent. Robust standard errors were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs). The Weibull model (and not the Cox model) was used because it does not assume hazard remains constant over time, which is appropriate in this instance, as the risk of an adverse event (AE) may be higher when first starting a medication or combination of medications relative to when doses are stabilized. Models were adjusted for age, sex, baseline mental health disorders, and baseline psychotropic medications. As earlier hospitalizations showed evidence of effect modification when this covariate was tested, hazard analyses were limited to patients not previously hospitalized.
The effect size of differences in hospitalization risk meeting statistical significance was assessed by estimating the number needed to harm (NNH) and 95% CIs (not shown) to observe 1 additional hospitalization in each medication group relative to the SSRI/SNRI monotherapy group over a 90-day period. A 95% CI for NNH that did not include 0 indicated the NNH was significant at the .05 level.10 All-cause mortality was evaluated with the Fisher exact test with post hoc Bonferroni-corrected comparisons as appropriate.
Results
Of 1,703 patients screened, 613 met all study inclusion criteria (Figure 1).
Baseline characteristics revealed no significant differences between groups in age or comorbid depression, schizophrenia, or SUDs (Table 1).
With the SSRI/SNRI monotherapy group as the referent, all concurrent therapy groups were at significantly increased risk for overall hospitalization within 2 years after the index date (Tables 2 & 3, Figure 2).
Risk for mental health hospitalization was significantly increased in all concurrent therapy groups relative to the referent group.
Although the risk for medical/surgical hospitalization was not significantly increased in the SSRI/SNRI and benzodiazepine therapy group (AHR, 1.9; 95% CI, 0.67-5.6), a significant difference was found in the SSRI/SNRI and opioid therapy group (AHR, 4.4; 95% CI, 1.6-12.0; NNH, 42).
Discussion
In 2013, Hawkins and colleagues evaluated hospitalization risk in veterans treated for PTSD within the Northwest VISN 20 between 2004 and 2010.11 Compared with patients treated with only an SSRI or SNRI, those treated with 1 of those medications and a benzodiazepine were at significantly higher risk for overall hospitalization (AHR, 1.79; 95% CI, 1.38-2.32; P < .001) and mental health hospitalization (AHR, 1.87; 95% CI, 1.37-2.53; P < .001). Furthermore, those prescribed a benzodiazepine and an opioid along with an SSRI or SNRI were at higher risk for overall hospitalization (AHR, 2.98; 95% CI, 2.22-4.00; P < .001), mental health hospitalization (AHR, 2.00; 95% CI, 1.35-2.98; P < .01), medical/surgical hospitalization (AHR, 4.86; 95% CI, 3.30-7.14; P < .001), and ED visits (AHR, 2.01; 95% CI, 1.53-2.65; P < .001).
Findings from the present study, which covered a period after the newest PTSD guideline was released,support findings reported by Hawkins and colleagues in their retrospective cohort study covering an earlier period.2,11 In the present study, compared with the monotherapy group, the SSRI/SNRI and benzodiazepine therapy group and the SSRI/SNRI, benzodiazepine, and opioid therapy group were at higher risk for both overall hospitalization and mental health hospitalization within 2 years. However, in a subset of PTSD patients prescribed opioids along with first-line pharmacotherapy, this study found that overall, mental health, and medical/surgical hospitalizations were significantly increased as well. Furthermore, this study found 2-year mortality was significantly higher for the SSRI/SNRI, benzodiazepine, and opioid therapy group than for the SSRI/SNRI monotherapy group.
Adjusted hazard ratios were higher in the present study than those in the study by Hawkins and colleagues,but CIs were wider as well.11 These differences may be attributable to the relatively smaller sample size of the present study and may explain why the HR was higher for the SSRI/SNRI and opioid therapy group than for the SSRI/SNRI, benzodiazepine, and opioid therapy group.
Nevertheless, these results support the growing body of evidence establishing the many risks for AEs when benzodiazepines and opioids are prescribed in the setting of PTSD. Unfortunately, it seems that, against clear guideline recommendations and literature findings, these medications still are being prescribed to this vulnerable, high-risk population.
In the last few months of 2013, the VA health care system launched 2 important medication safety initiatives. The Psychotropic Drug Safety Initiative (PDSI) was established as a quality improvement initiative for evidence-based provision of psychotropic medications. One PDSI metric in particular focused on reducing the proportion of veterans with PTSD being treated with benzodiazepines. The Opioid Safety Initiative (OSI) came as a response to a dramatic increase in the number of fatal overdoses related to prescription opioids—an increase linked to an unprecedented jump in opioid use for nonmalignant pain. As the present study’s inclusion cutoff date of August 1, 2013, preceded the debut of both PDSI and OSI, the benzodiazepine and opioid prescription rates reported here might be higher than those currently being found under the 2 initiatives.
Limitations
This study had several limitations that might affect the interpretation or generalizability of findings. Requiring at least a 30-day supply for prescription eligibility was an attempt to focus on chronic use of medications rather than on, for example, onetime supplies of opioids for dental procedures. However, prescription fill history was not assessed. Therefore, patients could have been included in certain study groups even if their SSRI, SNRI, benzodiazepine, or opioid prescription was not refilled. Furthermore, only VA medical records were used; non-VA prescriptions were not captured.
In addition, this study was limited to patients who at bare minimum were prescribed an SSRI or an SNRI. Some patients may have been prescribed a benzodiazepine and/or an opioid but were not on appropriate first-line pharmacotherapy for PTSD. These patients were excluded from the study, and their relative hospitalization risk went unexplored. Therefore, the magnitude of the issue at hand might have been underestimated.
Although psychotherapy is a first-line treatment option for PTSD, the study did not assess the potential impact of psychotherapy on outcomes or the groups’ relative proportions of patients undergoing psychotherapy. It is unknown whether the groups were equivalent at baseline in regards to psychotherapy participation rates.
This study did not characterize the specific reasons for hospitalization beyond whether it was for a mental health or a medical/surgical issue; thus, no distinction was made between hospitalizations for an elective procedure and hospitalizations for a drug overdose or an injury. Investigators could characterize admission diagnoses to better assess whether hospitalizations are truly associated with study medications or whether patients are being hospitalized for unrelated reasons. In addition, they could elucidate the true nature of hospitalization risk associated with SSRI/SNRI, benzodiazepine, and opioid use by comparing admission diagnoses made before and after initiation of these pharmacologic therapies.
This study also could not assess outcomes for patients who presented to the ED but were not admitted. If the hospital’s floor and ED beds were at full capacity, some patients might have been transferred to an outside facility. However, this scenario is not common at SAVAHCS, where the study was conducted.
Although some comorbid conditions were noted, the study did not evaluate whether its patients had a compelling indication for benzodiazepines in particular. Opioid use is very limited to the treatment of pain, and the majority of the patients on opioid therapy in this study had a diagnosed pain syndrome.
Because of the study’s sample size and power limitations, patients were eligible to be included in a concurrent therapy group if a benzodiazepine, an opioid, or both were added no later than 1 year after SSRI/SNRI initiation. This gap of up to 1 year might have introduced some variability in exposure to risk from earlier prescribed medications. However, sensitivity analyses were performed with multiple constructed Weibull models of time to hospitalization based on subsets with varying overlapping medication gaps. Analyses revealed relatively stable HRs, suggesting that potential bias did not occur.
Future Directions
Investigators could explore the higher all-cause mortality rates in the SSRI/SNRI, benzodiazepine, and opioid therapy group, as this study did not assess cause of death in these patients. Whether any patients died of reasons directly attributable to benzodiazepines or opioids is unknown.
That SSRIs and SNRIs are the only established first-line pharmacologic treatment options for PTSD symptoms partly accounts for the widespread use of benzodiazepines in this population. For that reason, beyond characterizing the many risks associated with using benzodiazepines to manage these symptoms, there is a huge need to research the viability of other pharmacologic agents in treating PTSD. This is especially important given the slower onset to efficacy of the SSRIs and SNRIs; per estimates, only up to 60% of patients respond to SSRIs, and 20% to 30% achieve full remission of PTSD.12 Furthermore, these rates likely are even lower for combat veterans than those for the general population. Several trials discussed in a 2009 guideline review of the treatment of patients with acute stress disorder and PTSD have called into question the efficacy of SSRIs for combat-related PTSD.13 In these randomized, controlled trials, change in PTSD symptom severity as measured with CAPS was not significantly reduced with SSRIs compared with placebo.
A systematic review revealed that, of the nonantidepressants used as adjuncts in treating patients who do not achieve remission with SSRIs, the atypical antipsychotic risperidone may have the strongest supporting evidence.12 However, the present study found high rates of antipsychotic use in the SSRI/SNRI, benzodiazepine, and opioid therapy group, which also had the highest all-cause mortality rate. The safety of risperidone as an alternative treatment needs further evaluation.
Some prospective studies have suggested that the α1 blockers doxazosin and prazosin, the latter of which is commonly used for PTSD nightmares, also may improve PTSD symptoms as assessed by CAPS.14,15 Although these results are promising, the trials to date have been conducted with relatively small sample sizes.
With more veterans being treated for PTSD within the VA health care system, the central treatment goal remains: Adequately address the symptoms of PTSD while minimizing the harm caused by medications. Prescribers should limit benzodiazepine and opioid use in this population and consider safer nonpharmacologic and pharmacologic treatment options when possible.
Conclusion
Combat veterans with PTSD who are prescribed benzodiazepines and/or opioids in addition to first-line pharmacotherapy are at significantly increased risk for overall and mental health hospitalization.
Click here to read the digital edition.
Posttraumatic stress disorder (PTSD) is a mental health condition that may develop in response to a traumatic event, such as that experienced by a soldier during active combat duty. In 2009, more than 495,000 veterans within the VA health care system were treated for PTSD—nearly triple the number a decade earlier.1 Core symptoms of PTSD include alterations in arousal and reactivity, avoidant behaviors, negative alterations in mood and cognition, and intrusive thoughts and nightmares. All of the symptoms can be debilitating. First-line pharmacotherapy options that target these core symptoms include selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs).2
The anxiolytic and sedative effects of benzodiazepines may provide quick relief from many of the secondary symptoms of PTSD, including sleep disturbances, irritability, and panic attacks. However, benzodiazepines potentially interfere with the extinction of conditioned fear—a goal integral to certain types of psychotherapy, such as exposure therapy.3,4 In addition, the systematic review and meta-analysis by Guina and colleagues revealed that benzodiazepines are ineffective in the treatment of PTSD.5 The majority of the evaluated studies that used PTSD-specific measures (eg, Clinician-Administered PTSD Scale [CAPS]) found increased PTSD severity and worse prognosis with use of these medications.5 In 2010, the VA and the DoD released a joint guideline for PTSD management.2 According to the guideline, benzodiazepines cause harm when used in PTSD and are relatively contraindicated in combat veterans because of the higher incidence of comorbid substance use disorders (SUDs) in these veterans relative to the general population.2,6
Opioid use also has been linked to poor functional and clinical outcomes in veterans with PTSD. Among patients being treated for opioid use disorder, those with PTSD were less likely to endorse employment as a main source of income and had a higher incidence of recent attempted suicide.7 In a large retrospective cohort study, Operation Iraqi Freedom and Operation Enduring Freedom veterans with PTSD who were prescribed opioids were more likely to present to the emergency department (ED) and to be hospitalized for overdoses and injuries.8
Despite the risks of benzodiazepine and opioid use in this patient population, these medications are still often prescribed to veterans with PTSD for symptomatic relief. In fiscal year 2009, across the VHA system 37% of veterans diagnosed with PTSD were prescribed a benzodiazepine, 69% of the time by a mental health provider.9 Among Iraq and Afghanistan veterans, those with PTSD were significantly more likely to be prescribed an opioid for diagnosed pain—relative to those with a mental health disorder other than PTSD and those without a mental health disorder.8 Thus, there seems to be a disconnect between guideline recommendations and current practice.
The authors conducted a study to assess the potential risk of hospitalization for veterans with PTSD prescribed first-line pharmacotherapy and those also prescribed concurrent benzodiazepine and/or opioid therapy since the release of the PTSD guideline in 2010.2
Methods
In this retrospective cohort study, conducted at the Southern Arizona VA Health Care System (SAVAHCS), the authors analyzed electronic medical record data from November 1, 2009 to August 1, 2015. Study inclusion criteria were veteran, aged 18 to 89 years, diagnosis of PTSD (International Classification of Diseases, Ninth Revision, Clinical Modification code 309.81), and SSRI or SNRI newly prescribed between November 1, 2010 and August 1, 2013.
Any veteran prescribed at least one 30-day or longer supply of any benzodiazepine or opioid within 1 year before the SSRI/SNRI initial prescription date was excluded from the study. Also excluded was any patient treated for PTSD at a facility outside SAVAHCS or whose 2-year evaluation period extended past August 1, 2015.
Study Groups
An outpatient prescription was determined to be the initial SSRI/SNRI prescription for a patient who received less than a 30-day cumulative supply of any SSRI or SNRI within 1 year before that prescription date. Citalopram, desvenlafaxine, duloxetine, escitalopram, fluoxetine, fluvoxamine, levomilnacipran, milnacipran, paroxetine, sertraline, venlafaxine, vilazodone, and vortioxetine were the prespecified SSRI/SNRIs included in the study.
Patients who received at least 1 outpatient prescription for any benzodiazepine (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and benzodiazepine therapy. Alprazolam, chlordiazepoxide, clonazepam, clorazepate, diazepam, estazolam, flurazepam, lorazepam, oxazepam, temazepam, and triazolam were the prespecified benzodiazepines included in the study.
Patients who received at least 1 outpatient prescription for any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI and opioid therapy. Codeine, fentanyl, hydrocodone, hydromorphone, levorphanol, meperidine, methadone, morphine, oxymorphone, pentazocine, propoxyphene, and tramadol were the prespecified opioids included in this study.
Patients who received at least 1 outpatient prescription for any benzodiazepine and any opioid (minimum 30-day supply) within 1 year after the initial SSRI/SNRI prescription date were determined to be on concurrent SSRI/SNRI, benzodiazepine, and opioid therapy.
The index date was defined as the first date of prescription overlap. If there was no benzodiazepine or opioid prescription within 1 year after the initial SSRI/SNRI prescription date, the patient was categorized as being on SSRI/SNRI monotherapy, and the index date was the date of the initial SSRI/SNRI prescription. For each patient, hospitalization data from the 2-year period after the index date were evaluated.
Outcomes and Data Collection
For evaluation of the primary outcome (2-year overall hospitalization risk), the number of unique mental health and medical/surgical hospitalizations was identified by the number of discharge summaries documented in the patient chart during the evaluation period. Time to first hospitalization was recorded for the survival data analysis. Secondary outcomes were mental health hospitalization risk, medical/surgical hospitalization risk, and all-cause mortality within 2 years.
Demographic data that were collected included age, sex, comorbid mental health disorders, comorbid SUDs, and concomitant use of psychotropic medications at index date (baseline). Select comorbid mental health disorders (anxiety, schizophrenia, depression, bipolar disorder) and substance use disorders (alcohol, opioid, illicit drug) also were identified. Data on insomnia and pain comorbidities (headaches or migraines; neuropathy; head, neck, back, arthritis, or joint pain) were collected, as these comorbidities could be indications for prescribing benzodiazepines and opioids. Concomitant baseline use of classes of psychotropic medications (antipsychotics, non-SSRI/SNRI antidepressants, mood stabilizers, anxiolytics, nonbenzodiazepine sedatives/hypnotics) also were documented. Last, hospitalizations within 6 months before the initial SSRI/SNRI prescription date were noted.
Statistical Analysis
Descriptive statistics were used to analyze all baseline demographic data. Continuous measures were evaluated with 1-way analyses of variance and post hoc Bonferroni-corrected pairwise comparisons, and categorical measures with contingency tables and χ2 tests or Fisher exact tests. When the overall χ2 test was significant across all 4 study groups, post hoc comparisons were performed between the SSRI/SNRI monotherapy group and each other group with Bonferroni adjusted for 3 comparisons.
Unadjusted and adjusted Weibull proportional hazard regression models were used to estimate hospitalization risk within 2 years after the index date for the different study groups with the SSRI/SNRI monotherapy group as the referent. Robust standard errors were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs). The Weibull model (and not the Cox model) was used because it does not assume hazard remains constant over time, which is appropriate in this instance, as the risk of an adverse event (AE) may be higher when first starting a medication or combination of medications relative to when doses are stabilized. Models were adjusted for age, sex, baseline mental health disorders, and baseline psychotropic medications. As earlier hospitalizations showed evidence of effect modification when this covariate was tested, hazard analyses were limited to patients not previously hospitalized.
The effect size of differences in hospitalization risk meeting statistical significance was assessed by estimating the number needed to harm (NNH) and 95% CIs (not shown) to observe 1 additional hospitalization in each medication group relative to the SSRI/SNRI monotherapy group over a 90-day period. A 95% CI for NNH that did not include 0 indicated the NNH was significant at the .05 level.10 All-cause mortality was evaluated with the Fisher exact test with post hoc Bonferroni-corrected comparisons as appropriate.
Results
Of 1,703 patients screened, 613 met all study inclusion criteria (Figure 1).
Baseline characteristics revealed no significant differences between groups in age or comorbid depression, schizophrenia, or SUDs (Table 1).
With the SSRI/SNRI monotherapy group as the referent, all concurrent therapy groups were at significantly increased risk for overall hospitalization within 2 years after the index date (Tables 2 & 3, Figure 2).
Risk for mental health hospitalization was significantly increased in all concurrent therapy groups relative to the referent group.
Although the risk for medical/surgical hospitalization was not significantly increased in the SSRI/SNRI and benzodiazepine therapy group (AHR, 1.9; 95% CI, 0.67-5.6), a significant difference was found in the SSRI/SNRI and opioid therapy group (AHR, 4.4; 95% CI, 1.6-12.0; NNH, 42).
Discussion
In 2013, Hawkins and colleagues evaluated hospitalization risk in veterans treated for PTSD within the Northwest VISN 20 between 2004 and 2010.11 Compared with patients treated with only an SSRI or SNRI, those treated with 1 of those medications and a benzodiazepine were at significantly higher risk for overall hospitalization (AHR, 1.79; 95% CI, 1.38-2.32; P < .001) and mental health hospitalization (AHR, 1.87; 95% CI, 1.37-2.53; P < .001). Furthermore, those prescribed a benzodiazepine and an opioid along with an SSRI or SNRI were at higher risk for overall hospitalization (AHR, 2.98; 95% CI, 2.22-4.00; P < .001), mental health hospitalization (AHR, 2.00; 95% CI, 1.35-2.98; P < .01), medical/surgical hospitalization (AHR, 4.86; 95% CI, 3.30-7.14; P < .001), and ED visits (AHR, 2.01; 95% CI, 1.53-2.65; P < .001).
Findings from the present study, which covered a period after the newest PTSD guideline was released,support findings reported by Hawkins and colleagues in their retrospective cohort study covering an earlier period.2,11 In the present study, compared with the monotherapy group, the SSRI/SNRI and benzodiazepine therapy group and the SSRI/SNRI, benzodiazepine, and opioid therapy group were at higher risk for both overall hospitalization and mental health hospitalization within 2 years. However, in a subset of PTSD patients prescribed opioids along with first-line pharmacotherapy, this study found that overall, mental health, and medical/surgical hospitalizations were significantly increased as well. Furthermore, this study found 2-year mortality was significantly higher for the SSRI/SNRI, benzodiazepine, and opioid therapy group than for the SSRI/SNRI monotherapy group.
Adjusted hazard ratios were higher in the present study than those in the study by Hawkins and colleagues,but CIs were wider as well.11 These differences may be attributable to the relatively smaller sample size of the present study and may explain why the HR was higher for the SSRI/SNRI and opioid therapy group than for the SSRI/SNRI, benzodiazepine, and opioid therapy group.
Nevertheless, these results support the growing body of evidence establishing the many risks for AEs when benzodiazepines and opioids are prescribed in the setting of PTSD. Unfortunately, it seems that, against clear guideline recommendations and literature findings, these medications still are being prescribed to this vulnerable, high-risk population.
In the last few months of 2013, the VA health care system launched 2 important medication safety initiatives. The Psychotropic Drug Safety Initiative (PDSI) was established as a quality improvement initiative for evidence-based provision of psychotropic medications. One PDSI metric in particular focused on reducing the proportion of veterans with PTSD being treated with benzodiazepines. The Opioid Safety Initiative (OSI) came as a response to a dramatic increase in the number of fatal overdoses related to prescription opioids—an increase linked to an unprecedented jump in opioid use for nonmalignant pain. As the present study’s inclusion cutoff date of August 1, 2013, preceded the debut of both PDSI and OSI, the benzodiazepine and opioid prescription rates reported here might be higher than those currently being found under the 2 initiatives.
Limitations
This study had several limitations that might affect the interpretation or generalizability of findings. Requiring at least a 30-day supply for prescription eligibility was an attempt to focus on chronic use of medications rather than on, for example, onetime supplies of opioids for dental procedures. However, prescription fill history was not assessed. Therefore, patients could have been included in certain study groups even if their SSRI, SNRI, benzodiazepine, or opioid prescription was not refilled. Furthermore, only VA medical records were used; non-VA prescriptions were not captured.
In addition, this study was limited to patients who at bare minimum were prescribed an SSRI or an SNRI. Some patients may have been prescribed a benzodiazepine and/or an opioid but were not on appropriate first-line pharmacotherapy for PTSD. These patients were excluded from the study, and their relative hospitalization risk went unexplored. Therefore, the magnitude of the issue at hand might have been underestimated.
Although psychotherapy is a first-line treatment option for PTSD, the study did not assess the potential impact of psychotherapy on outcomes or the groups’ relative proportions of patients undergoing psychotherapy. It is unknown whether the groups were equivalent at baseline in regards to psychotherapy participation rates.
This study did not characterize the specific reasons for hospitalization beyond whether it was for a mental health or a medical/surgical issue; thus, no distinction was made between hospitalizations for an elective procedure and hospitalizations for a drug overdose or an injury. Investigators could characterize admission diagnoses to better assess whether hospitalizations are truly associated with study medications or whether patients are being hospitalized for unrelated reasons. In addition, they could elucidate the true nature of hospitalization risk associated with SSRI/SNRI, benzodiazepine, and opioid use by comparing admission diagnoses made before and after initiation of these pharmacologic therapies.
This study also could not assess outcomes for patients who presented to the ED but were not admitted. If the hospital’s floor and ED beds were at full capacity, some patients might have been transferred to an outside facility. However, this scenario is not common at SAVAHCS, where the study was conducted.
Although some comorbid conditions were noted, the study did not evaluate whether its patients had a compelling indication for benzodiazepines in particular. Opioid use is very limited to the treatment of pain, and the majority of the patients on opioid therapy in this study had a diagnosed pain syndrome.
Because of the study’s sample size and power limitations, patients were eligible to be included in a concurrent therapy group if a benzodiazepine, an opioid, or both were added no later than 1 year after SSRI/SNRI initiation. This gap of up to 1 year might have introduced some variability in exposure to risk from earlier prescribed medications. However, sensitivity analyses were performed with multiple constructed Weibull models of time to hospitalization based on subsets with varying overlapping medication gaps. Analyses revealed relatively stable HRs, suggesting that potential bias did not occur.
Future Directions
Investigators could explore the higher all-cause mortality rates in the SSRI/SNRI, benzodiazepine, and opioid therapy group, as this study did not assess cause of death in these patients. Whether any patients died of reasons directly attributable to benzodiazepines or opioids is unknown.
That SSRIs and SNRIs are the only established first-line pharmacologic treatment options for PTSD symptoms partly accounts for the widespread use of benzodiazepines in this population. For that reason, beyond characterizing the many risks associated with using benzodiazepines to manage these symptoms, there is a huge need to research the viability of other pharmacologic agents in treating PTSD. This is especially important given the slower onset to efficacy of the SSRIs and SNRIs; per estimates, only up to 60% of patients respond to SSRIs, and 20% to 30% achieve full remission of PTSD.12 Furthermore, these rates likely are even lower for combat veterans than those for the general population. Several trials discussed in a 2009 guideline review of the treatment of patients with acute stress disorder and PTSD have called into question the efficacy of SSRIs for combat-related PTSD.13 In these randomized, controlled trials, change in PTSD symptom severity as measured with CAPS was not significantly reduced with SSRIs compared with placebo.
A systematic review revealed that, of the nonantidepressants used as adjuncts in treating patients who do not achieve remission with SSRIs, the atypical antipsychotic risperidone may have the strongest supporting evidence.12 However, the present study found high rates of antipsychotic use in the SSRI/SNRI, benzodiazepine, and opioid therapy group, which also had the highest all-cause mortality rate. The safety of risperidone as an alternative treatment needs further evaluation.
Some prospective studies have suggested that the α1 blockers doxazosin and prazosin, the latter of which is commonly used for PTSD nightmares, also may improve PTSD symptoms as assessed by CAPS.14,15 Although these results are promising, the trials to date have been conducted with relatively small sample sizes.
With more veterans being treated for PTSD within the VA health care system, the central treatment goal remains: Adequately address the symptoms of PTSD while minimizing the harm caused by medications. Prescribers should limit benzodiazepine and opioid use in this population and consider safer nonpharmacologic and pharmacologic treatment options when possible.
Conclusion
Combat veterans with PTSD who are prescribed benzodiazepines and/or opioids in addition to first-line pharmacotherapy are at significantly increased risk for overall and mental health hospitalization.
Click here to read the digital edition.
1. Bernardy NC, Lund BC, Alexander B, Jenkyn AB, Schnurr PP, Friedman MJ. Gender differences in prescribing among veterans diagnosed with posttraumatic stress disorder. J Gen Intern Med. 2013;28(suppl 2):S542-S548.
2. Management of Post-Traumatic Stress Working Group, Department of Veterans Affairs, Department of Defense. VA/DoD Clinical Practice Guideline for Management of Post-Traumatic Stress. http://www.healthquality.va.gov/PTSD-full-2010c .pdf. Published October 2010. Accessed July 12, 2015.
3. Marks IM, Swinson RP, Baso˘glu M, et al. Alprazolam and exposure alone and combined in panic disorder with agoraphobia. A controlled study in London and Toronto. Br J Psychiatry. 1993;162:776-787.
4. Wilhelm FH, Roth WT. Acute and delayed effects of alprazolam on flight phobics during exposure. Behav Res Ther. 1997;35(9):831-841.
5. Guina J, Rossetter SR, DeRhodes BJ, Nahhas RW, Welton RS. Benzodiazepines for PTSD: a systematic review and meta-analysis. J Psychiatr Pract. 2015;21(4):281-303.
6. Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Prevalence and Axis I comorbidity of full and partial posttraumatic stress disorder in the United States: results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. J Anxiety Disord. 2011;25(3):456-465.
7. Mills KL, Teesson M, Ross J, Darke S, Shanahan M. The costs and outcomes of treatment for opioid dependence associated with posttraumatic stress disorder. Psychiatr Serv. 2005;56(8):940-945.
8. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
9. Abrams TE, Lund BC, Bernardy NC, Friedman MJ. Aligning clinical practice to PTSD treatment guidelines: medication prescribing by provider type. Psychiatr Serv. 2013;64(2):142-148.
10. Altman DG, Andersen PK. Calculating the number needed to treat for trials where the outcome is time to an event. BMJ. 1999;319(7223):1492-1495.
11. Hawkins EJ, Malte CA, Grossbard J, Saxon AJ, Imel ZE, Kivlahan DR. Comparative safety of benzodiazepines and opioids among Veterans Affairs patients with posttraumatic stress disorder. J Addict Med. 2013;7(5):354-362.
12. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
13. Benedek DM, Friedman MJ, Zatzick D, Ursano RJ. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
14. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
15. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
1. Bernardy NC, Lund BC, Alexander B, Jenkyn AB, Schnurr PP, Friedman MJ. Gender differences in prescribing among veterans diagnosed with posttraumatic stress disorder. J Gen Intern Med. 2013;28(suppl 2):S542-S548.
2. Management of Post-Traumatic Stress Working Group, Department of Veterans Affairs, Department of Defense. VA/DoD Clinical Practice Guideline for Management of Post-Traumatic Stress. http://www.healthquality.va.gov/PTSD-full-2010c .pdf. Published October 2010. Accessed July 12, 2015.
3. Marks IM, Swinson RP, Baso˘glu M, et al. Alprazolam and exposure alone and combined in panic disorder with agoraphobia. A controlled study in London and Toronto. Br J Psychiatry. 1993;162:776-787.
4. Wilhelm FH, Roth WT. Acute and delayed effects of alprazolam on flight phobics during exposure. Behav Res Ther. 1997;35(9):831-841.
5. Guina J, Rossetter SR, DeRhodes BJ, Nahhas RW, Welton RS. Benzodiazepines for PTSD: a systematic review and meta-analysis. J Psychiatr Pract. 2015;21(4):281-303.
6. Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Prevalence and Axis I comorbidity of full and partial posttraumatic stress disorder in the United States: results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. J Anxiety Disord. 2011;25(3):456-465.
7. Mills KL, Teesson M, Ross J, Darke S, Shanahan M. The costs and outcomes of treatment for opioid dependence associated with posttraumatic stress disorder. Psychiatr Serv. 2005;56(8):940-945.
8. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
9. Abrams TE, Lund BC, Bernardy NC, Friedman MJ. Aligning clinical practice to PTSD treatment guidelines: medication prescribing by provider type. Psychiatr Serv. 2013;64(2):142-148.
10. Altman DG, Andersen PK. Calculating the number needed to treat for trials where the outcome is time to an event. BMJ. 1999;319(7223):1492-1495.
11. Hawkins EJ, Malte CA, Grossbard J, Saxon AJ, Imel ZE, Kivlahan DR. Comparative safety of benzodiazepines and opioids among Veterans Affairs patients with posttraumatic stress disorder. J Addict Med. 2013;7(5):354-362.
12. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
13. Benedek DM, Friedman MJ, Zatzick D, Ursano RJ. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
14. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
15. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
Data Trends on PTSD
Given the nature of active duty, service members are exposed to higher levels of stress and greater risk of posttraumatic stress disorder (PTSD). The comorbid conditions that often accompany a PTSD diagnosis, such as substance abuse or depression, can complicate treatment and threaten a veteran’s overall health. These factors place PTSD at the forefront of treatments the VA offers its patients. All veterans now are screened for symptoms of both PTSD and depression at their first visit to the VA facility. Mental health services overall remain a priority within the VA. The VA has requested that its 2017 budget include a 4.6% increase in funding for mental health services—boosting its funding to $7.8 billion. This increase will allow the VA to provide a high level of medical service and promote future research, such as a brain tissue biorepository launched in 2015, which supports research on the causes, progression, and treatment of veterans with PTSD.
“Understanding the cellular and circuit contributions to abnormal brain activity in PTSD is critical in the search for potential biomarkers of susceptibility, illness and treatment response and for developing new treatments targeting the conditions at the cellular level,” said Matthew J. Friedman, MD, PhD, senior advisor to the National Center for PTSD.
Click here to read the digital edition.
Given the nature of active duty, service members are exposed to higher levels of stress and greater risk of posttraumatic stress disorder (PTSD). The comorbid conditions that often accompany a PTSD diagnosis, such as substance abuse or depression, can complicate treatment and threaten a veteran’s overall health. These factors place PTSD at the forefront of treatments the VA offers its patients. All veterans now are screened for symptoms of both PTSD and depression at their first visit to the VA facility. Mental health services overall remain a priority within the VA. The VA has requested that its 2017 budget include a 4.6% increase in funding for mental health services—boosting its funding to $7.8 billion. This increase will allow the VA to provide a high level of medical service and promote future research, such as a brain tissue biorepository launched in 2015, which supports research on the causes, progression, and treatment of veterans with PTSD.
“Understanding the cellular and circuit contributions to abnormal brain activity in PTSD is critical in the search for potential biomarkers of susceptibility, illness and treatment response and for developing new treatments targeting the conditions at the cellular level,” said Matthew J. Friedman, MD, PhD, senior advisor to the National Center for PTSD.
Click here to read the digital edition.
Given the nature of active duty, service members are exposed to higher levels of stress and greater risk of posttraumatic stress disorder (PTSD). The comorbid conditions that often accompany a PTSD diagnosis, such as substance abuse or depression, can complicate treatment and threaten a veteran’s overall health. These factors place PTSD at the forefront of treatments the VA offers its patients. All veterans now are screened for symptoms of both PTSD and depression at their first visit to the VA facility. Mental health services overall remain a priority within the VA. The VA has requested that its 2017 budget include a 4.6% increase in funding for mental health services—boosting its funding to $7.8 billion. This increase will allow the VA to provide a high level of medical service and promote future research, such as a brain tissue biorepository launched in 2015, which supports research on the causes, progression, and treatment of veterans with PTSD.
“Understanding the cellular and circuit contributions to abnormal brain activity in PTSD is critical in the search for potential biomarkers of susceptibility, illness and treatment response and for developing new treatments targeting the conditions at the cellular level,” said Matthew J. Friedman, MD, PhD, senior advisor to the National Center for PTSD.
Click here to read the digital edition.
Prazosin and doxazosin for PTSD are underutilized and underdosed
The primary symptoms of PTSD are recurrent and include intrusive memories and dreams of the traumatic events, flashbacks, hypervigilance, irritability, sleep disturbances, and persistent avoidance of stimuli associated with the traumatic event. According to the National Comorbidity Survey, the estimated lifetime prevalence of PTSD among adults is 6.8% and is more common in women (9.7%) than men (3.6%).2 Among veterans, the prevalence of PTSD has been reported as:
- 31% among male Vietnam veterans (lifetime)
- 10% among Gulf War veterans
- 14% among Iraq and Afghanistan veterans.3
Why is PTSD overlooked in substance use?
Among individuals with SUD, 10% to 63% have comorbid PTSD.4 A recent report underscores the complexity and challenges of SUD–PTSD comorbidity.5 Most PTSD patients with comorbid SUD receive treatment only for SUD and the PTSD symptoms often are unaddressed.5 Those suffering from PTSD often abuse alcohol because they might consider it to be a coping strategy. Alcohol reduces hyperactivation of the dorsal anterior cingulate cortex caused by re-experiencing PTSD symptoms. Other substances of abuse, such as Cannabis, could suppress PTSD symptoms through alternate mechanisms (eg, endocannabinoid receptors). All of these could mask PTSD symptoms, which can delay diagnosis and treatment.
SUD is the tip of the “SUD-PTSD iceberg.” Some clinicians tend to focus on detoxification while completely ignoring the underlying psychopathology of SUD, which may be PTSD. Even during detoxification, PTSD should be aggressively treated.6 Lastly, practice guidelines for managing SUD–PTSD comorbidity are lacking.
Targeting mechanisms of action
Noradrenergic mechanisms have been strongly implicated in the pathophysiology of PTSD. However, selective serotonin reuptake inhibitors, such as sertraline and paroxetine, are the only FDA-approved pharmacotherapy options for PTSD, although their efficacy is limited, perhaps because they are serotonergic.
Prazosin, an alpha-1 (α-1) adrenergic antagonist that is FDA-approved for hypertension and benign prostatic hypertrophy, has been studied for treating nightmares in PTSD.7 Prazosin has shown efficacy for nightmares in PTSD and other daytime symptoms, such as flashbacks, hypervigilance, and irritability.8 Several studies support the efficacy of prazosin in persons suffering from PTSD.9-11 Use of lower dosages in clinical trials might explain why prazosin did not separate from placebo in some studies. (See Table summarizing studies of prazosin dosing for PTSD.)
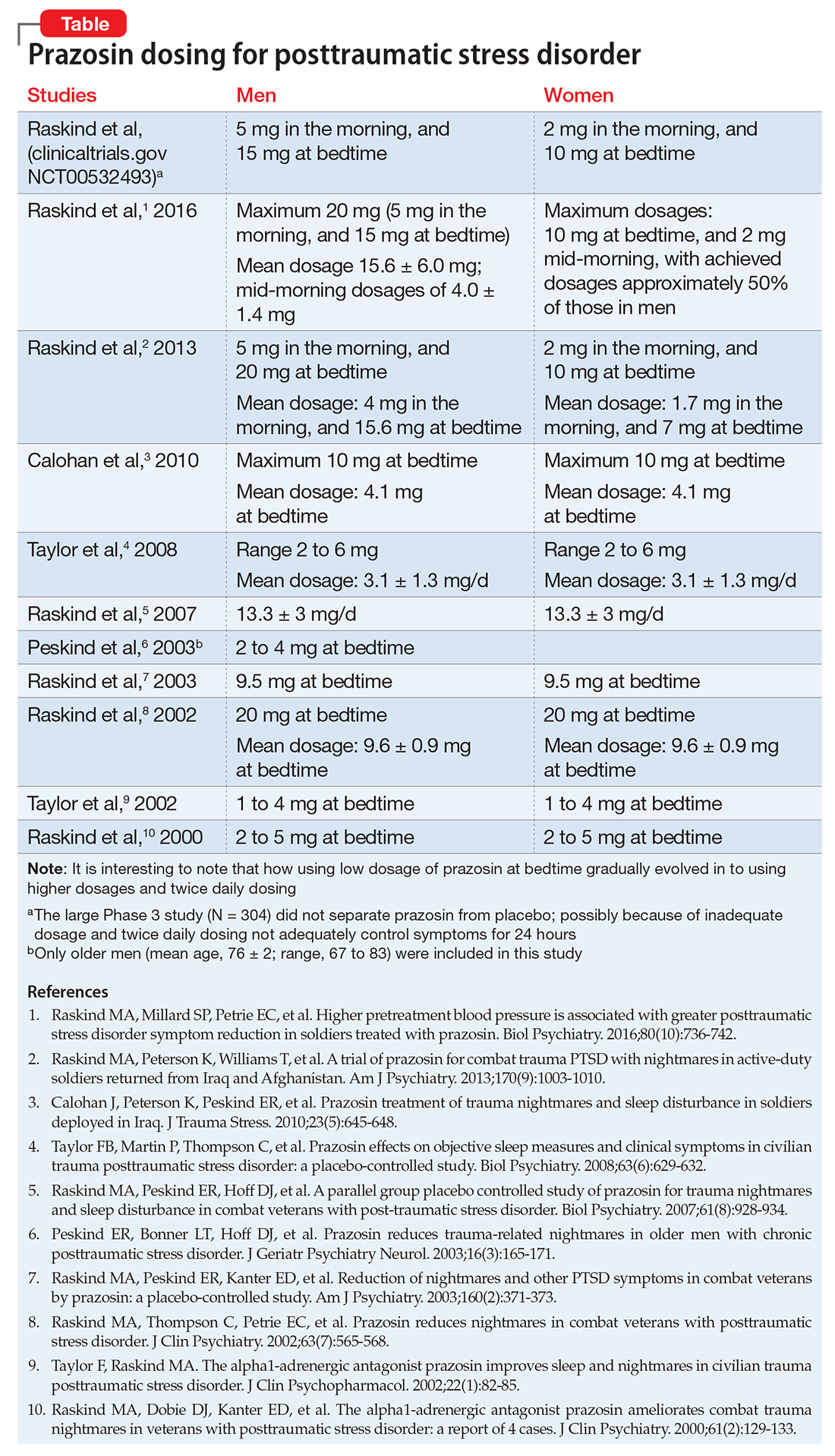
In a study of 12,844 veterans, the mean maximum prazosin dosage reached in the first year of treatment was 3.6 mg/d, and only 14% of patients reached the minimum Veterans Affairs recommended dosage of 6 mg/d.17 The most recent (March 2009) American Psychiatric Association practice guidelines recommend prazosin, 3 to 15 mg at bedtime.18
Prazosin has a short half-life of 2 to 3 hours and duration of action of 6 to 10 hours. Therefore, its use is limited to 2 or 3 times daily dosing. Higher (30 to 50 mg) and more frequent (2 to 3 times per day) dosages8,12,13 might be needed because of the drug’s short half-life.
Doxazosin. Another α-1 adrenergic drug, doxazosin, 8 to 16 mg/d, has shown benefit for PTSD as well.14,15 Doxazosin, which has a longer half-life (16 to 30 hours), requires only once-daily dosing.16 The most common side effects of prazosin and doxazosin are dizziness, headache, and drowsiness; syncope has been reported but is rare.
Prazosin and doxazosin also are used to treat substance abuse, such as alcohol use disorder19-21 and cocaine use disorder.22,23 This “two birds with one stone” approach could become more common in clinical practice.
Until a major breakthrough in PTSD treatment emerges, prazosin and doxazosin, although off-label, are reasonable treatment approaches.
1. Zimmerman M, Mattia JI. Is posttraumatic stress disorder underdiagnosed in routine clinical settings? J Nerv Ment Dis. 1999;187(7):420-428.
2. National Comorbidity Survey. 12-month prevalence of DSM-IV/WMH-CIDI disorders by sex and cohort (n=9282). http://www.hcp.med.harvard.edu/ncs/ftpdir/NCS-R_12-month_Prevalence_Estimates.pdf. Published 2005. Accessed February 10, 2017.
3. Gradus JL. Epidemiology of PTSD. http://www.ptsd.va.gov/professional/PTSD-overview/epidemiological-facts-ptsd.asp. Updated February 23, 2016. Accessed February 13, 2017.
4. Debell F, Fear NT, Head M, et al. A systematic review of the comorbidity between PTSD and alcohol misuse. Soc Psychiatry Psychiatr Epidemiol. 2014;49(9):1401-1425.
5. Vujanovic AA, Bonn-Miller MO, Petry NM. Co-occurring posttraumatic stress and substance use: emerging research on correlates, mechanisms, and treatments-introduction to the special issue. Psychol Addict Behav. 2016;30(7):713-719.
6. Jacobsen LK, Southwick SM, Kosten TR. Substance use disorders in patients with posttraumatic stress disorder: a review of the literature. Am J Psychiatry. 2001;158(8):1184-1190.
7. Raskind MA, Dobie DJ, Kanter ED, et al. The alpha1-adrenergic antagonist prazosin ameliorates combat trauma nightmares in veterans with posttraumatic stress disorder: a report of 4 cases. J Clin Psychiatry. 2000;61(2):129-133.
8. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
11. Raskind MA, Millard SP, Petrie EC, et al. Higher pretreatment blood pressure is associated with greater posttraumatic stress disorder symptom reduction in soldiers treated with prazosin. Biol Psychiatry. 2016;80(10):736-742.
12. Koola MM, Varghese SP, Fawcett JA. High-dose prazosin for the treatment of post-traumatic stress disorder. Ther Adv Psychopharmacol. 2014;4(1):43-47.
13. Vaishnav M, Patel V, Varghese SP, et al. Fludrocortisone in posttraumatic stress disorder: effective for symptoms and prazosin-induced hypotension. Prim Care Companion CNS Disord. 2014;16(6). doi: 10.4088/PCC.14l01676.
14. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
15. Roepke S, Danker-Hopfe H, Repantis D, et al. Doxazosin, an α-1-adrenergic-receptor antagonist, for nightmares in patients with posttraumatic stress disorder and/or borderline personality disorder: a chart review. Pharmacopsychiatry. 2017;50(1):26-31.
16. Smith C, Koola MM. Evidence for using doxazosin in the treatment of posttraumatic stress disorder. Psychiatr Ann. 2016;46(9):553-555.
17. Alexander B, Lund BC, Bernardy NC, et al. Early discontinuation and suboptimal dosing of prazosin: a potential missed opportunity for veterans with posttraumatic stress disorder. J Clin Psychiatry. 2015;76(5):e639-e644.
18. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf. Accessed February 10, 2017.
19. Qazi H, Wijegunaratne H, Savajiyani R, et al. Naltrexone and prazosin combination for posttraumatic stress disorder and alcohol use disorder. Prim Care Companion CNS Disord. 2014;16(4). doi: 10.4088/PCC.14l01638.
20. Simpson TL, Malte CA, Dietel B, et al. A pilot trial of prazosin, an alpha-1 adrenergic antagonist, for comorbid alcohol dependence and posttraumatic stress disorder. Alcohol Clin Exp Res. 2015;39(5):808-817.
21. Kenna GA, Haass-Koffler CL, Zywiak WH, et al. Role of the α1 blocker doxazosin in alcoholism: a proof-of-concept randomized controlled trial. Addict Biol. 2016;21(4):904-914.
22. Shorter D, Lindsay JA, Kosten TR. The alpha-1 adrenergic antagonist doxazosin for treatment of cocaine dependence: a pilot study. Drug Alcohol Depend. 2013;131(1-2):66-70.
23. Newton TF, De La Garza R II, Brown G, et al. Noradrenergic α1 receptor antagonist treatment attenuates positive subjective effects of cocaine in humans: a randomized trial. PLoS One. 2012;7(2):e30854.
The primary symptoms of PTSD are recurrent and include intrusive memories and dreams of the traumatic events, flashbacks, hypervigilance, irritability, sleep disturbances, and persistent avoidance of stimuli associated with the traumatic event. According to the National Comorbidity Survey, the estimated lifetime prevalence of PTSD among adults is 6.8% and is more common in women (9.7%) than men (3.6%).2 Among veterans, the prevalence of PTSD has been reported as:
- 31% among male Vietnam veterans (lifetime)
- 10% among Gulf War veterans
- 14% among Iraq and Afghanistan veterans.3
Why is PTSD overlooked in substance use?
Among individuals with SUD, 10% to 63% have comorbid PTSD.4 A recent report underscores the complexity and challenges of SUD–PTSD comorbidity.5 Most PTSD patients with comorbid SUD receive treatment only for SUD and the PTSD symptoms often are unaddressed.5 Those suffering from PTSD often abuse alcohol because they might consider it to be a coping strategy. Alcohol reduces hyperactivation of the dorsal anterior cingulate cortex caused by re-experiencing PTSD symptoms. Other substances of abuse, such as Cannabis, could suppress PTSD symptoms through alternate mechanisms (eg, endocannabinoid receptors). All of these could mask PTSD symptoms, which can delay diagnosis and treatment.
SUD is the tip of the “SUD-PTSD iceberg.” Some clinicians tend to focus on detoxification while completely ignoring the underlying psychopathology of SUD, which may be PTSD. Even during detoxification, PTSD should be aggressively treated.6 Lastly, practice guidelines for managing SUD–PTSD comorbidity are lacking.
Targeting mechanisms of action
Noradrenergic mechanisms have been strongly implicated in the pathophysiology of PTSD. However, selective serotonin reuptake inhibitors, such as sertraline and paroxetine, are the only FDA-approved pharmacotherapy options for PTSD, although their efficacy is limited, perhaps because they are serotonergic.
Prazosin, an alpha-1 (α-1) adrenergic antagonist that is FDA-approved for hypertension and benign prostatic hypertrophy, has been studied for treating nightmares in PTSD.7 Prazosin has shown efficacy for nightmares in PTSD and other daytime symptoms, such as flashbacks, hypervigilance, and irritability.8 Several studies support the efficacy of prazosin in persons suffering from PTSD.9-11 Use of lower dosages in clinical trials might explain why prazosin did not separate from placebo in some studies. (See Table summarizing studies of prazosin dosing for PTSD.)

In a study of 12,844 veterans, the mean maximum prazosin dosage reached in the first year of treatment was 3.6 mg/d, and only 14% of patients reached the minimum Veterans Affairs recommended dosage of 6 mg/d.17 The most recent (March 2009) American Psychiatric Association practice guidelines recommend prazosin, 3 to 15 mg at bedtime.18
Prazosin has a short half-life of 2 to 3 hours and duration of action of 6 to 10 hours. Therefore, its use is limited to 2 or 3 times daily dosing. Higher (30 to 50 mg) and more frequent (2 to 3 times per day) dosages8,12,13 might be needed because of the drug’s short half-life.
Doxazosin. Another α-1 adrenergic drug, doxazosin, 8 to 16 mg/d, has shown benefit for PTSD as well.14,15 Doxazosin, which has a longer half-life (16 to 30 hours), requires only once-daily dosing.16 The most common side effects of prazosin and doxazosin are dizziness, headache, and drowsiness; syncope has been reported but is rare.
Prazosin and doxazosin also are used to treat substance abuse, such as alcohol use disorder19-21 and cocaine use disorder.22,23 This “two birds with one stone” approach could become more common in clinical practice.
Until a major breakthrough in PTSD treatment emerges, prazosin and doxazosin, although off-label, are reasonable treatment approaches.
The primary symptoms of PTSD are recurrent and include intrusive memories and dreams of the traumatic events, flashbacks, hypervigilance, irritability, sleep disturbances, and persistent avoidance of stimuli associated with the traumatic event. According to the National Comorbidity Survey, the estimated lifetime prevalence of PTSD among adults is 6.8% and is more common in women (9.7%) than men (3.6%).2 Among veterans, the prevalence of PTSD has been reported as:
- 31% among male Vietnam veterans (lifetime)
- 10% among Gulf War veterans
- 14% among Iraq and Afghanistan veterans.3
Why is PTSD overlooked in substance use?
Among individuals with SUD, 10% to 63% have comorbid PTSD.4 A recent report underscores the complexity and challenges of SUD–PTSD comorbidity.5 Most PTSD patients with comorbid SUD receive treatment only for SUD and the PTSD symptoms often are unaddressed.5 Those suffering from PTSD often abuse alcohol because they might consider it to be a coping strategy. Alcohol reduces hyperactivation of the dorsal anterior cingulate cortex caused by re-experiencing PTSD symptoms. Other substances of abuse, such as Cannabis, could suppress PTSD symptoms through alternate mechanisms (eg, endocannabinoid receptors). All of these could mask PTSD symptoms, which can delay diagnosis and treatment.
SUD is the tip of the “SUD-PTSD iceberg.” Some clinicians tend to focus on detoxification while completely ignoring the underlying psychopathology of SUD, which may be PTSD. Even during detoxification, PTSD should be aggressively treated.6 Lastly, practice guidelines for managing SUD–PTSD comorbidity are lacking.
Targeting mechanisms of action
Noradrenergic mechanisms have been strongly implicated in the pathophysiology of PTSD. However, selective serotonin reuptake inhibitors, such as sertraline and paroxetine, are the only FDA-approved pharmacotherapy options for PTSD, although their efficacy is limited, perhaps because they are serotonergic.
Prazosin, an alpha-1 (α-1) adrenergic antagonist that is FDA-approved for hypertension and benign prostatic hypertrophy, has been studied for treating nightmares in PTSD.7 Prazosin has shown efficacy for nightmares in PTSD and other daytime symptoms, such as flashbacks, hypervigilance, and irritability.8 Several studies support the efficacy of prazosin in persons suffering from PTSD.9-11 Use of lower dosages in clinical trials might explain why prazosin did not separate from placebo in some studies. (See Table summarizing studies of prazosin dosing for PTSD.)

In a study of 12,844 veterans, the mean maximum prazosin dosage reached in the first year of treatment was 3.6 mg/d, and only 14% of patients reached the minimum Veterans Affairs recommended dosage of 6 mg/d.17 The most recent (March 2009) American Psychiatric Association practice guidelines recommend prazosin, 3 to 15 mg at bedtime.18
Prazosin has a short half-life of 2 to 3 hours and duration of action of 6 to 10 hours. Therefore, its use is limited to 2 or 3 times daily dosing. Higher (30 to 50 mg) and more frequent (2 to 3 times per day) dosages8,12,13 might be needed because of the drug’s short half-life.
Doxazosin. Another α-1 adrenergic drug, doxazosin, 8 to 16 mg/d, has shown benefit for PTSD as well.14,15 Doxazosin, which has a longer half-life (16 to 30 hours), requires only once-daily dosing.16 The most common side effects of prazosin and doxazosin are dizziness, headache, and drowsiness; syncope has been reported but is rare.
Prazosin and doxazosin also are used to treat substance abuse, such as alcohol use disorder19-21 and cocaine use disorder.22,23 This “two birds with one stone” approach could become more common in clinical practice.
Until a major breakthrough in PTSD treatment emerges, prazosin and doxazosin, although off-label, are reasonable treatment approaches.
1. Zimmerman M, Mattia JI. Is posttraumatic stress disorder underdiagnosed in routine clinical settings? J Nerv Ment Dis. 1999;187(7):420-428.
2. National Comorbidity Survey. 12-month prevalence of DSM-IV/WMH-CIDI disorders by sex and cohort (n=9282). http://www.hcp.med.harvard.edu/ncs/ftpdir/NCS-R_12-month_Prevalence_Estimates.pdf. Published 2005. Accessed February 10, 2017.
3. Gradus JL. Epidemiology of PTSD. http://www.ptsd.va.gov/professional/PTSD-overview/epidemiological-facts-ptsd.asp. Updated February 23, 2016. Accessed February 13, 2017.
4. Debell F, Fear NT, Head M, et al. A systematic review of the comorbidity between PTSD and alcohol misuse. Soc Psychiatry Psychiatr Epidemiol. 2014;49(9):1401-1425.
5. Vujanovic AA, Bonn-Miller MO, Petry NM. Co-occurring posttraumatic stress and substance use: emerging research on correlates, mechanisms, and treatments-introduction to the special issue. Psychol Addict Behav. 2016;30(7):713-719.
6. Jacobsen LK, Southwick SM, Kosten TR. Substance use disorders in patients with posttraumatic stress disorder: a review of the literature. Am J Psychiatry. 2001;158(8):1184-1190.
7. Raskind MA, Dobie DJ, Kanter ED, et al. The alpha1-adrenergic antagonist prazosin ameliorates combat trauma nightmares in veterans with posttraumatic stress disorder: a report of 4 cases. J Clin Psychiatry. 2000;61(2):129-133.
8. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
11. Raskind MA, Millard SP, Petrie EC, et al. Higher pretreatment blood pressure is associated with greater posttraumatic stress disorder symptom reduction in soldiers treated with prazosin. Biol Psychiatry. 2016;80(10):736-742.
12. Koola MM, Varghese SP, Fawcett JA. High-dose prazosin for the treatment of post-traumatic stress disorder. Ther Adv Psychopharmacol. 2014;4(1):43-47.
13. Vaishnav M, Patel V, Varghese SP, et al. Fludrocortisone in posttraumatic stress disorder: effective for symptoms and prazosin-induced hypotension. Prim Care Companion CNS Disord. 2014;16(6). doi: 10.4088/PCC.14l01676.
14. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
15. Roepke S, Danker-Hopfe H, Repantis D, et al. Doxazosin, an α-1-adrenergic-receptor antagonist, for nightmares in patients with posttraumatic stress disorder and/or borderline personality disorder: a chart review. Pharmacopsychiatry. 2017;50(1):26-31.
16. Smith C, Koola MM. Evidence for using doxazosin in the treatment of posttraumatic stress disorder. Psychiatr Ann. 2016;46(9):553-555.
17. Alexander B, Lund BC, Bernardy NC, et al. Early discontinuation and suboptimal dosing of prazosin: a potential missed opportunity for veterans with posttraumatic stress disorder. J Clin Psychiatry. 2015;76(5):e639-e644.
18. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf. Accessed February 10, 2017.
19. Qazi H, Wijegunaratne H, Savajiyani R, et al. Naltrexone and prazosin combination for posttraumatic stress disorder and alcohol use disorder. Prim Care Companion CNS Disord. 2014;16(4). doi: 10.4088/PCC.14l01638.
20. Simpson TL, Malte CA, Dietel B, et al. A pilot trial of prazosin, an alpha-1 adrenergic antagonist, for comorbid alcohol dependence and posttraumatic stress disorder. Alcohol Clin Exp Res. 2015;39(5):808-817.
21. Kenna GA, Haass-Koffler CL, Zywiak WH, et al. Role of the α1 blocker doxazosin in alcoholism: a proof-of-concept randomized controlled trial. Addict Biol. 2016;21(4):904-914.
22. Shorter D, Lindsay JA, Kosten TR. The alpha-1 adrenergic antagonist doxazosin for treatment of cocaine dependence: a pilot study. Drug Alcohol Depend. 2013;131(1-2):66-70.
23. Newton TF, De La Garza R II, Brown G, et al. Noradrenergic α1 receptor antagonist treatment attenuates positive subjective effects of cocaine in humans: a randomized trial. PLoS One. 2012;7(2):e30854.
1. Zimmerman M, Mattia JI. Is posttraumatic stress disorder underdiagnosed in routine clinical settings? J Nerv Ment Dis. 1999;187(7):420-428.
2. National Comorbidity Survey. 12-month prevalence of DSM-IV/WMH-CIDI disorders by sex and cohort (n=9282). http://www.hcp.med.harvard.edu/ncs/ftpdir/NCS-R_12-month_Prevalence_Estimates.pdf. Published 2005. Accessed February 10, 2017.
3. Gradus JL. Epidemiology of PTSD. http://www.ptsd.va.gov/professional/PTSD-overview/epidemiological-facts-ptsd.asp. Updated February 23, 2016. Accessed February 13, 2017.
4. Debell F, Fear NT, Head M, et al. A systematic review of the comorbidity between PTSD and alcohol misuse. Soc Psychiatry Psychiatr Epidemiol. 2014;49(9):1401-1425.
5. Vujanovic AA, Bonn-Miller MO, Petry NM. Co-occurring posttraumatic stress and substance use: emerging research on correlates, mechanisms, and treatments-introduction to the special issue. Psychol Addict Behav. 2016;30(7):713-719.
6. Jacobsen LK, Southwick SM, Kosten TR. Substance use disorders in patients with posttraumatic stress disorder: a review of the literature. Am J Psychiatry. 2001;158(8):1184-1190.
7. Raskind MA, Dobie DJ, Kanter ED, et al. The alpha1-adrenergic antagonist prazosin ameliorates combat trauma nightmares in veterans with posttraumatic stress disorder: a report of 4 cases. J Clin Psychiatry. 2000;61(2):129-133.
8. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
11. Raskind MA, Millard SP, Petrie EC, et al. Higher pretreatment blood pressure is associated with greater posttraumatic stress disorder symptom reduction in soldiers treated with prazosin. Biol Psychiatry. 2016;80(10):736-742.
12. Koola MM, Varghese SP, Fawcett JA. High-dose prazosin for the treatment of post-traumatic stress disorder. Ther Adv Psychopharmacol. 2014;4(1):43-47.
13. Vaishnav M, Patel V, Varghese SP, et al. Fludrocortisone in posttraumatic stress disorder: effective for symptoms and prazosin-induced hypotension. Prim Care Companion CNS Disord. 2014;16(6). doi: 10.4088/PCC.14l01676.
14. Rodgman C, Verrico CD, Holst M, et al. Doxazosin XL reduces symptoms of posttraumatic stress disorder in veterans with PTSD: a pilot clinical trial. J Clin Psychiatry. 2016;77(5):e561-e565.
15. Roepke S, Danker-Hopfe H, Repantis D, et al. Doxazosin, an α-1-adrenergic-receptor antagonist, for nightmares in patients with posttraumatic stress disorder and/or borderline personality disorder: a chart review. Pharmacopsychiatry. 2017;50(1):26-31.
16. Smith C, Koola MM. Evidence for using doxazosin in the treatment of posttraumatic stress disorder. Psychiatr Ann. 2016;46(9):553-555.
17. Alexander B, Lund BC, Bernardy NC, et al. Early discontinuation and suboptimal dosing of prazosin: a potential missed opportunity for veterans with posttraumatic stress disorder. J Clin Psychiatry. 2015;76(5):e639-e644.
18. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf. Accessed February 10, 2017.
19. Qazi H, Wijegunaratne H, Savajiyani R, et al. Naltrexone and prazosin combination for posttraumatic stress disorder and alcohol use disorder. Prim Care Companion CNS Disord. 2014;16(4). doi: 10.4088/PCC.14l01638.
20. Simpson TL, Malte CA, Dietel B, et al. A pilot trial of prazosin, an alpha-1 adrenergic antagonist, for comorbid alcohol dependence and posttraumatic stress disorder. Alcohol Clin Exp Res. 2015;39(5):808-817.
21. Kenna GA, Haass-Koffler CL, Zywiak WH, et al. Role of the α1 blocker doxazosin in alcoholism: a proof-of-concept randomized controlled trial. Addict Biol. 2016;21(4):904-914.
22. Shorter D, Lindsay JA, Kosten TR. The alpha-1 adrenergic antagonist doxazosin for treatment of cocaine dependence: a pilot study. Drug Alcohol Depend. 2013;131(1-2):66-70.
23. Newton TF, De La Garza R II, Brown G, et al. Noradrenergic α1 receptor antagonist treatment attenuates positive subjective effects of cocaine in humans: a randomized trial. PLoS One. 2012;7(2):e30854.
When to consider cranial electrotherapy stimulation for patients with PTSD
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
MDMA – the love drug – makes a therapeutic comeback
VIENNA – What a difference a decade can make in the world of psychiatry.
Take, for example, the case of 3,4-methylenedioxymethamphetamine, better known as MDMA or, when used recreationally, as ecstasy, the love drug.
“Ten years ago at pretty much every scientific meeting where MDMA was being discussed, people were looking to find problems with it. People were dredging around trying to vilify this drug, because there was a hope that it might cause brain damage, which would justify having made its use illicit. Ten years later, we’ve changed direction completely, from fear and hating MDMA to loving it. Now we’re talking about the possibility that MDMA might actually heal the brain, and restoring MDMA to the therapeutic armamentarium,” David Nutt, MD, observed at the annual congress of the European College of Neuropsychopharmacology.
Indeed, the drug’s potential as an adjunct to psychotherapy in patients with posttraumatic stress disorder was the topic of a packed-to-the-gills session in the largest hall at the ECNP Congress, where Dr. Nutt highlighted recent insights into the psychopharmacology of MDMA and other speakers described evidence of the drug’s salutary effects on autobiographical memory and social cognition.
“The biggest problem with MDMA is its name,” quipped Dr. Nutt, professor of neuropsychopharmacology at Imperial College London.
“It used to be called ‘empathy,’ but when it started being used recreationally at raves and in the clubs, the dealers decided to change its name to ‘ecstasy.’ And that created havoc, because there’s nothing that aged editors of newspapers hate more than young people having ecstasy. They hated the term, and so the drug had to go,” according to the psychiatrist.
MDMA’s comeback as a potentially valuable medication in psychiatry can be traced to the first report of the drug’s impressive success when used as an adjunct to psychotherapy in a randomized, placebo-controlled pilot study. Michael C. Mithoefer, MD, a psychiatrist in private practice in South Carolina, and his coinvestigators stunned the psychiatric world by reporting that 10 of 12 patients with chronic PTSD refractory to both medications and psychotherapy showed significant clinical improvement in response to just two sessions of MDMA-assisted psychotherapy supplementing a more conventional course of psychotherapy (J Psychopharmacology. 2011 Apr;25[4]:439-52).
Moreover, the benefits proved durable: In a subsequent paper, the investigators reported the clinical benefit of this two-dose treatment program persisted at a mean 3.8 years of follow-up and no safety concerns had been seen (J Psychopharmacol. 2013 Jan;27[1]:28-39).
This study, which eventually drew the attention of military veterans’ groups with political clout, proved hugely influential, especially since PTSD is so common and often is highly treatment resistant.
“We’re now living in a very strange world where trauma has in some ways become the No. 1 problem facing many societies,” Dr. Nutt observed.
He predicted that with the Food and Drug Administration’s recent approval of clinical trials of MDMA in patients with PTSD, the drug will be licensed for that indication “within the next couple years.”
How MDMA works
The pharmacology of MDMA is complex, he continued. The drug is chiefly a serotonin-releasing agent and 5HT reuptake blocker, but it also acts as an agonist on alpha-adrenergic receptors, has muscarinic and histamine-blocking effects, and promotes release of oxytocin.
Animal studies have demonstrated that MDMA facilitates extinction of fear memories through a mechanism involving changes in levels of brain-derived neurotrophic factor. Experience in humans has shown that the drug has diverse pro-social effects: It is activating, enhances mood, promotes more flexible thinking, boosts tactile experiences, and increases empathy, which in turn aids patients in bonding with their therapists.
Dr. Nutt and his coinvestigators performed the first whole-brain study of the effects of MDMA using functional MRI. This double-blind, placebo-controlled, crossover study in healthy volunteers used measurements obtained through arterial spin labeling and analysis of blood oxygen level–dependent resting state functional connectivity. The investigators documented that the marked increase in positive mood and decreased magnitude of negative personal memories produced by MDMA was accompanied by profound reduction of cerebral blood flow in the right amygdala and hippocampus. Cerebral blood flow also was reduced in the right medial temporal lobe, thalamus, and inferior visual cortex. MDMA also resulted in decreased amygdala-cortical connectivity (Biol Psychiatry. 2015 Oct 15;78[8]:554-62; Int J Neuropsychopharmacol. 2014 Apr;17[4]:527-40).
The changes in those particular brain systems are consistent with and most likely underlie the drug’s therapeutic effects, he said. Taken together, they could serve to assist a patient in re-engaging with traumatic memories with less interference from emotional centers, thereby helping to gain executive control of the memory of the trauma.
H. Valerie Curran, PhD, a coinvestigator in the brain imaging study, cautioned the rapt audience that while there are abundant favorable anecdotal reports from psychotherapists going back as far as the 1970s, the actual evidence base for MDMA as a therapeutic adjunct to psychotherapy for PTSD is still pretty thin. She noted that in their groundbreaking study, Dr. Mithoefer and his colleagues used an unconventional form of psychotherapy modeled on the LSD therapy developed by Stanislav Grof, MD, PhD. The two MDMA-assisted sessions were each 8 hours long and included shamanistic techniques and specialized breathing to promote diminished oxygen to the brain. Also, the patient sat on a futon listening to music with a male therapist on one side and a female therapist on the other. As a clinical psychologist herself, she assured the audience that this is not standard practice in her field.
Only one other randomized, double-blind, placebo-controlled study of MDMA-assisted psychotherapy has been published to date (J Psychopharmacol. 2013 Jan;27[1]:40-52). With just 12 participants, it was too small to be conclusive. So there is a definite need for additional controlled studies on the interaction between MDMA and evidence-based forms of psychotherapy. Fortunately, additional clinical trials are ongoing, noted Dr. Curran, professor of psychopharmacology at University College London.
She presented highlights of a study she and her coinvestigators carried out to determine how MDMA affects the encoding and recall of emotional autobiographical memories, since the core of most psychotherapy for PTSD entails controlled revisiting of traumatic memories. The nonblinded study included a group of recreational MDMA users who – on two separate occasions, one under the influence of street-quality MDMA of uncertain dose and purity, the other on placebo – were tasked with responding to self-threatening scenarios, exposure to compassionate imagery, and a large series of positive and negative adjectives addressed at themselves or another person (J Psychopharmacol. 2015 Sep;29[9]:961-70).
The investigators found that MDMA enhanced the emotional intensity, vividness, and positivity of the subjects’ best autobiographical memories while modestly reducing the negativity of their worst memories. Structured ratings of compassion markedly increased while on MDMA. Overall, the drug’s effects were similar to those obtained through rigorous cognitive training methods developed in venerable Eastern contemplative practices in pursuit of a compassionate mindset, according to Dr. Curran.
The study results suggest a mechanism by which MDMA might enhance psychotherapy not only by improving the therapeutic alliance but also by reducing self-referential emotional processing without diminishing declarative memory, she added.
Findings of Swiss studies
Matthias E. Liechti, MD, head of the psychopharmacology research unit at the University of Basel, explained that at present Switzerland is the only country in the world where it’s legal to prescribe MDMA. Ditto LSD. Psychiatrists can do so on a case-by-case basis outside of a clinical trial setting in patients with treatment-resistant PTSD or anxiety disorders.
Dr. Liechti and his coinvestigators are interested in examining how MDMA affects social cognition as assessed by outcome measures, including a structured face emotion recognition test, the multifaceted empathy test, and a sexual arousal task.
In a series of studies in which they exposed subjects to MDMA, alcohol, methamphetamine, or LSD, they have established that both MDMA and LSD produce empathogenic effects that are possibly serotonin mediated. On a visual analog scale, subjects on those drugs gave high marks for feeling happy, open, trusting, and extroverted, and having a sense of well-being. MDMA impaired recognition of fearful, angry, and sad faces.
In contrast, methamphetamine, a pure stimulant that activates the norepinephrine/dopamine system, produced no empathogenic effects, but it enhanced recognition of sad or fearful faces. Alcohol slightly increased self-ratings for trust, happiness, and openness.
Methamphetamine increased ratings of sexual arousal in response to explicit sexual stimuli, while MDMA had no effect on sexual arousal.
MDMA and LSD increased oxytocin, prolactin, and cortisol levels consistent with their serotonergic effects. Methylphenidate did not, Dr. Liechti said.
A neuroscientist in the audience raised a possible safety concern regarding MDMA: If the drug has an agonist effect on serotonin receptors, couldn’t it have cardiac side effects similar to those of fenfluramine, a drug now banned because it stimulated the abundant 5HT-2b receptors present in the heart, resulting in increased risk of pulmonary hypertension and other adverse cardiovascular effects?
Dr. Nutt replied that there are multitudes of serotonin receptor subtypes, and it’s not yet known whether MDMA acts upon the 5HT-2b receptor. In any case, it shouldn’t be an issue for the drug’s medicinal use.
“Luckily, the effects of MDMA wear off quickly, and when it’s used with psychotherapy we may be giving only one or two doses in a lifetime, so it shouldn’t be a concern,” he said.
Dr. Nutt reported that the functional MRI brain imaging study was funded by a British television station and a private foundation.
“The reason for that is we’ve found it impossible to get any money from any traditional government funders to study drugs like MDMA unless you write grants to show they’re harmful,” he asserted.
Dr. Curran reported having no financial conflicts of interest regarding her studies. Dr. Liechti’s work is supported by the Swiss National Science Foundation.
VIENNA – What a difference a decade can make in the world of psychiatry.
Take, for example, the case of 3,4-methylenedioxymethamphetamine, better known as MDMA or, when used recreationally, as ecstasy, the love drug.
“Ten years ago at pretty much every scientific meeting where MDMA was being discussed, people were looking to find problems with it. People were dredging around trying to vilify this drug, because there was a hope that it might cause brain damage, which would justify having made its use illicit. Ten years later, we’ve changed direction completely, from fear and hating MDMA to loving it. Now we’re talking about the possibility that MDMA might actually heal the brain, and restoring MDMA to the therapeutic armamentarium,” David Nutt, MD, observed at the annual congress of the European College of Neuropsychopharmacology.
Indeed, the drug’s potential as an adjunct to psychotherapy in patients with posttraumatic stress disorder was the topic of a packed-to-the-gills session in the largest hall at the ECNP Congress, where Dr. Nutt highlighted recent insights into the psychopharmacology of MDMA and other speakers described evidence of the drug’s salutary effects on autobiographical memory and social cognition.
“The biggest problem with MDMA is its name,” quipped Dr. Nutt, professor of neuropsychopharmacology at Imperial College London.
“It used to be called ‘empathy,’ but when it started being used recreationally at raves and in the clubs, the dealers decided to change its name to ‘ecstasy.’ And that created havoc, because there’s nothing that aged editors of newspapers hate more than young people having ecstasy. They hated the term, and so the drug had to go,” according to the psychiatrist.
MDMA’s comeback as a potentially valuable medication in psychiatry can be traced to the first report of the drug’s impressive success when used as an adjunct to psychotherapy in a randomized, placebo-controlled pilot study. Michael C. Mithoefer, MD, a psychiatrist in private practice in South Carolina, and his coinvestigators stunned the psychiatric world by reporting that 10 of 12 patients with chronic PTSD refractory to both medications and psychotherapy showed significant clinical improvement in response to just two sessions of MDMA-assisted psychotherapy supplementing a more conventional course of psychotherapy (J Psychopharmacology. 2011 Apr;25[4]:439-52).
Moreover, the benefits proved durable: In a subsequent paper, the investigators reported the clinical benefit of this two-dose treatment program persisted at a mean 3.8 years of follow-up and no safety concerns had been seen (J Psychopharmacol. 2013 Jan;27[1]:28-39).
This study, which eventually drew the attention of military veterans’ groups with political clout, proved hugely influential, especially since PTSD is so common and often is highly treatment resistant.
“We’re now living in a very strange world where trauma has in some ways become the No. 1 problem facing many societies,” Dr. Nutt observed.
He predicted that with the Food and Drug Administration’s recent approval of clinical trials of MDMA in patients with PTSD, the drug will be licensed for that indication “within the next couple years.”
How MDMA works
The pharmacology of MDMA is complex, he continued. The drug is chiefly a serotonin-releasing agent and 5HT reuptake blocker, but it also acts as an agonist on alpha-adrenergic receptors, has muscarinic and histamine-blocking effects, and promotes release of oxytocin.
Animal studies have demonstrated that MDMA facilitates extinction of fear memories through a mechanism involving changes in levels of brain-derived neurotrophic factor. Experience in humans has shown that the drug has diverse pro-social effects: It is activating, enhances mood, promotes more flexible thinking, boosts tactile experiences, and increases empathy, which in turn aids patients in bonding with their therapists.
Dr. Nutt and his coinvestigators performed the first whole-brain study of the effects of MDMA using functional MRI. This double-blind, placebo-controlled, crossover study in healthy volunteers used measurements obtained through arterial spin labeling and analysis of blood oxygen level–dependent resting state functional connectivity. The investigators documented that the marked increase in positive mood and decreased magnitude of negative personal memories produced by MDMA was accompanied by profound reduction of cerebral blood flow in the right amygdala and hippocampus. Cerebral blood flow also was reduced in the right medial temporal lobe, thalamus, and inferior visual cortex. MDMA also resulted in decreased amygdala-cortical connectivity (Biol Psychiatry. 2015 Oct 15;78[8]:554-62; Int J Neuropsychopharmacol. 2014 Apr;17[4]:527-40).
The changes in those particular brain systems are consistent with and most likely underlie the drug’s therapeutic effects, he said. Taken together, they could serve to assist a patient in re-engaging with traumatic memories with less interference from emotional centers, thereby helping to gain executive control of the memory of the trauma.
H. Valerie Curran, PhD, a coinvestigator in the brain imaging study, cautioned the rapt audience that while there are abundant favorable anecdotal reports from psychotherapists going back as far as the 1970s, the actual evidence base for MDMA as a therapeutic adjunct to psychotherapy for PTSD is still pretty thin. She noted that in their groundbreaking study, Dr. Mithoefer and his colleagues used an unconventional form of psychotherapy modeled on the LSD therapy developed by Stanislav Grof, MD, PhD. The two MDMA-assisted sessions were each 8 hours long and included shamanistic techniques and specialized breathing to promote diminished oxygen to the brain. Also, the patient sat on a futon listening to music with a male therapist on one side and a female therapist on the other. As a clinical psychologist herself, she assured the audience that this is not standard practice in her field.
Only one other randomized, double-blind, placebo-controlled study of MDMA-assisted psychotherapy has been published to date (J Psychopharmacol. 2013 Jan;27[1]:40-52). With just 12 participants, it was too small to be conclusive. So there is a definite need for additional controlled studies on the interaction between MDMA and evidence-based forms of psychotherapy. Fortunately, additional clinical trials are ongoing, noted Dr. Curran, professor of psychopharmacology at University College London.
She presented highlights of a study she and her coinvestigators carried out to determine how MDMA affects the encoding and recall of emotional autobiographical memories, since the core of most psychotherapy for PTSD entails controlled revisiting of traumatic memories. The nonblinded study included a group of recreational MDMA users who – on two separate occasions, one under the influence of street-quality MDMA of uncertain dose and purity, the other on placebo – were tasked with responding to self-threatening scenarios, exposure to compassionate imagery, and a large series of positive and negative adjectives addressed at themselves or another person (J Psychopharmacol. 2015 Sep;29[9]:961-70).
The investigators found that MDMA enhanced the emotional intensity, vividness, and positivity of the subjects’ best autobiographical memories while modestly reducing the negativity of their worst memories. Structured ratings of compassion markedly increased while on MDMA. Overall, the drug’s effects were similar to those obtained through rigorous cognitive training methods developed in venerable Eastern contemplative practices in pursuit of a compassionate mindset, according to Dr. Curran.
The study results suggest a mechanism by which MDMA might enhance psychotherapy not only by improving the therapeutic alliance but also by reducing self-referential emotional processing without diminishing declarative memory, she added.
Findings of Swiss studies
Matthias E. Liechti, MD, head of the psychopharmacology research unit at the University of Basel, explained that at present Switzerland is the only country in the world where it’s legal to prescribe MDMA. Ditto LSD. Psychiatrists can do so on a case-by-case basis outside of a clinical trial setting in patients with treatment-resistant PTSD or anxiety disorders.
Dr. Liechti and his coinvestigators are interested in examining how MDMA affects social cognition as assessed by outcome measures, including a structured face emotion recognition test, the multifaceted empathy test, and a sexual arousal task.
In a series of studies in which they exposed subjects to MDMA, alcohol, methamphetamine, or LSD, they have established that both MDMA and LSD produce empathogenic effects that are possibly serotonin mediated. On a visual analog scale, subjects on those drugs gave high marks for feeling happy, open, trusting, and extroverted, and having a sense of well-being. MDMA impaired recognition of fearful, angry, and sad faces.
In contrast, methamphetamine, a pure stimulant that activates the norepinephrine/dopamine system, produced no empathogenic effects, but it enhanced recognition of sad or fearful faces. Alcohol slightly increased self-ratings for trust, happiness, and openness.
Methamphetamine increased ratings of sexual arousal in response to explicit sexual stimuli, while MDMA had no effect on sexual arousal.
MDMA and LSD increased oxytocin, prolactin, and cortisol levels consistent with their serotonergic effects. Methylphenidate did not, Dr. Liechti said.
A neuroscientist in the audience raised a possible safety concern regarding MDMA: If the drug has an agonist effect on serotonin receptors, couldn’t it have cardiac side effects similar to those of fenfluramine, a drug now banned because it stimulated the abundant 5HT-2b receptors present in the heart, resulting in increased risk of pulmonary hypertension and other adverse cardiovascular effects?
Dr. Nutt replied that there are multitudes of serotonin receptor subtypes, and it’s not yet known whether MDMA acts upon the 5HT-2b receptor. In any case, it shouldn’t be an issue for the drug’s medicinal use.
“Luckily, the effects of MDMA wear off quickly, and when it’s used with psychotherapy we may be giving only one or two doses in a lifetime, so it shouldn’t be a concern,” he said.
Dr. Nutt reported that the functional MRI brain imaging study was funded by a British television station and a private foundation.
“The reason for that is we’ve found it impossible to get any money from any traditional government funders to study drugs like MDMA unless you write grants to show they’re harmful,” he asserted.
Dr. Curran reported having no financial conflicts of interest regarding her studies. Dr. Liechti’s work is supported by the Swiss National Science Foundation.
VIENNA – What a difference a decade can make in the world of psychiatry.
Take, for example, the case of 3,4-methylenedioxymethamphetamine, better known as MDMA or, when used recreationally, as ecstasy, the love drug.
“Ten years ago at pretty much every scientific meeting where MDMA was being discussed, people were looking to find problems with it. People were dredging around trying to vilify this drug, because there was a hope that it might cause brain damage, which would justify having made its use illicit. Ten years later, we’ve changed direction completely, from fear and hating MDMA to loving it. Now we’re talking about the possibility that MDMA might actually heal the brain, and restoring MDMA to the therapeutic armamentarium,” David Nutt, MD, observed at the annual congress of the European College of Neuropsychopharmacology.
Indeed, the drug’s potential as an adjunct to psychotherapy in patients with posttraumatic stress disorder was the topic of a packed-to-the-gills session in the largest hall at the ECNP Congress, where Dr. Nutt highlighted recent insights into the psychopharmacology of MDMA and other speakers described evidence of the drug’s salutary effects on autobiographical memory and social cognition.
“The biggest problem with MDMA is its name,” quipped Dr. Nutt, professor of neuropsychopharmacology at Imperial College London.
“It used to be called ‘empathy,’ but when it started being used recreationally at raves and in the clubs, the dealers decided to change its name to ‘ecstasy.’ And that created havoc, because there’s nothing that aged editors of newspapers hate more than young people having ecstasy. They hated the term, and so the drug had to go,” according to the psychiatrist.
MDMA’s comeback as a potentially valuable medication in psychiatry can be traced to the first report of the drug’s impressive success when used as an adjunct to psychotherapy in a randomized, placebo-controlled pilot study. Michael C. Mithoefer, MD, a psychiatrist in private practice in South Carolina, and his coinvestigators stunned the psychiatric world by reporting that 10 of 12 patients with chronic PTSD refractory to both medications and psychotherapy showed significant clinical improvement in response to just two sessions of MDMA-assisted psychotherapy supplementing a more conventional course of psychotherapy (J Psychopharmacology. 2011 Apr;25[4]:439-52).
Moreover, the benefits proved durable: In a subsequent paper, the investigators reported the clinical benefit of this two-dose treatment program persisted at a mean 3.8 years of follow-up and no safety concerns had been seen (J Psychopharmacol. 2013 Jan;27[1]:28-39).
This study, which eventually drew the attention of military veterans’ groups with political clout, proved hugely influential, especially since PTSD is so common and often is highly treatment resistant.
“We’re now living in a very strange world where trauma has in some ways become the No. 1 problem facing many societies,” Dr. Nutt observed.
He predicted that with the Food and Drug Administration’s recent approval of clinical trials of MDMA in patients with PTSD, the drug will be licensed for that indication “within the next couple years.”
How MDMA works
The pharmacology of MDMA is complex, he continued. The drug is chiefly a serotonin-releasing agent and 5HT reuptake blocker, but it also acts as an agonist on alpha-adrenergic receptors, has muscarinic and histamine-blocking effects, and promotes release of oxytocin.
Animal studies have demonstrated that MDMA facilitates extinction of fear memories through a mechanism involving changes in levels of brain-derived neurotrophic factor. Experience in humans has shown that the drug has diverse pro-social effects: It is activating, enhances mood, promotes more flexible thinking, boosts tactile experiences, and increases empathy, which in turn aids patients in bonding with their therapists.
Dr. Nutt and his coinvestigators performed the first whole-brain study of the effects of MDMA using functional MRI. This double-blind, placebo-controlled, crossover study in healthy volunteers used measurements obtained through arterial spin labeling and analysis of blood oxygen level–dependent resting state functional connectivity. The investigators documented that the marked increase in positive mood and decreased magnitude of negative personal memories produced by MDMA was accompanied by profound reduction of cerebral blood flow in the right amygdala and hippocampus. Cerebral blood flow also was reduced in the right medial temporal lobe, thalamus, and inferior visual cortex. MDMA also resulted in decreased amygdala-cortical connectivity (Biol Psychiatry. 2015 Oct 15;78[8]:554-62; Int J Neuropsychopharmacol. 2014 Apr;17[4]:527-40).
The changes in those particular brain systems are consistent with and most likely underlie the drug’s therapeutic effects, he said. Taken together, they could serve to assist a patient in re-engaging with traumatic memories with less interference from emotional centers, thereby helping to gain executive control of the memory of the trauma.
H. Valerie Curran, PhD, a coinvestigator in the brain imaging study, cautioned the rapt audience that while there are abundant favorable anecdotal reports from psychotherapists going back as far as the 1970s, the actual evidence base for MDMA as a therapeutic adjunct to psychotherapy for PTSD is still pretty thin. She noted that in their groundbreaking study, Dr. Mithoefer and his colleagues used an unconventional form of psychotherapy modeled on the LSD therapy developed by Stanislav Grof, MD, PhD. The two MDMA-assisted sessions were each 8 hours long and included shamanistic techniques and specialized breathing to promote diminished oxygen to the brain. Also, the patient sat on a futon listening to music with a male therapist on one side and a female therapist on the other. As a clinical psychologist herself, she assured the audience that this is not standard practice in her field.
Only one other randomized, double-blind, placebo-controlled study of MDMA-assisted psychotherapy has been published to date (J Psychopharmacol. 2013 Jan;27[1]:40-52). With just 12 participants, it was too small to be conclusive. So there is a definite need for additional controlled studies on the interaction between MDMA and evidence-based forms of psychotherapy. Fortunately, additional clinical trials are ongoing, noted Dr. Curran, professor of psychopharmacology at University College London.
She presented highlights of a study she and her coinvestigators carried out to determine how MDMA affects the encoding and recall of emotional autobiographical memories, since the core of most psychotherapy for PTSD entails controlled revisiting of traumatic memories. The nonblinded study included a group of recreational MDMA users who – on two separate occasions, one under the influence of street-quality MDMA of uncertain dose and purity, the other on placebo – were tasked with responding to self-threatening scenarios, exposure to compassionate imagery, and a large series of positive and negative adjectives addressed at themselves or another person (J Psychopharmacol. 2015 Sep;29[9]:961-70).
The investigators found that MDMA enhanced the emotional intensity, vividness, and positivity of the subjects’ best autobiographical memories while modestly reducing the negativity of their worst memories. Structured ratings of compassion markedly increased while on MDMA. Overall, the drug’s effects were similar to those obtained through rigorous cognitive training methods developed in venerable Eastern contemplative practices in pursuit of a compassionate mindset, according to Dr. Curran.
The study results suggest a mechanism by which MDMA might enhance psychotherapy not only by improving the therapeutic alliance but also by reducing self-referential emotional processing without diminishing declarative memory, she added.
Findings of Swiss studies
Matthias E. Liechti, MD, head of the psychopharmacology research unit at the University of Basel, explained that at present Switzerland is the only country in the world where it’s legal to prescribe MDMA. Ditto LSD. Psychiatrists can do so on a case-by-case basis outside of a clinical trial setting in patients with treatment-resistant PTSD or anxiety disorders.
Dr. Liechti and his coinvestigators are interested in examining how MDMA affects social cognition as assessed by outcome measures, including a structured face emotion recognition test, the multifaceted empathy test, and a sexual arousal task.
In a series of studies in which they exposed subjects to MDMA, alcohol, methamphetamine, or LSD, they have established that both MDMA and LSD produce empathogenic effects that are possibly serotonin mediated. On a visual analog scale, subjects on those drugs gave high marks for feeling happy, open, trusting, and extroverted, and having a sense of well-being. MDMA impaired recognition of fearful, angry, and sad faces.
In contrast, methamphetamine, a pure stimulant that activates the norepinephrine/dopamine system, produced no empathogenic effects, but it enhanced recognition of sad or fearful faces. Alcohol slightly increased self-ratings for trust, happiness, and openness.
Methamphetamine increased ratings of sexual arousal in response to explicit sexual stimuli, while MDMA had no effect on sexual arousal.
MDMA and LSD increased oxytocin, prolactin, and cortisol levels consistent with their serotonergic effects. Methylphenidate did not, Dr. Liechti said.
A neuroscientist in the audience raised a possible safety concern regarding MDMA: If the drug has an agonist effect on serotonin receptors, couldn’t it have cardiac side effects similar to those of fenfluramine, a drug now banned because it stimulated the abundant 5HT-2b receptors present in the heart, resulting in increased risk of pulmonary hypertension and other adverse cardiovascular effects?
Dr. Nutt replied that there are multitudes of serotonin receptor subtypes, and it’s not yet known whether MDMA acts upon the 5HT-2b receptor. In any case, it shouldn’t be an issue for the drug’s medicinal use.
“Luckily, the effects of MDMA wear off quickly, and when it’s used with psychotherapy we may be giving only one or two doses in a lifetime, so it shouldn’t be a concern,” he said.
Dr. Nutt reported that the functional MRI brain imaging study was funded by a British television station and a private foundation.
“The reason for that is we’ve found it impossible to get any money from any traditional government funders to study drugs like MDMA unless you write grants to show they’re harmful,” he asserted.
Dr. Curran reported having no financial conflicts of interest regarding her studies. Dr. Liechti’s work is supported by the Swiss National Science Foundation.