User login
Climate change expected to impose major burden on mental health
NEW YORK – Of the broad range of direct and indirect threats to public health anticipated from climate change, those involving mental health will place psychiatrists on the front lines of efforts to mitigate the impact, a member of the Climate Psychiatry Alliance said at the annual meeting of the American Psychiatric Association.
“One thing climate changes mean for us in psychiatry is more work,” reported Janet L. Lewis, MD, an assistant clinical professor of psychiatry at the University of Rochester (N.Y.).
Mental health is sensitive to climate. “Psychiatric patients can be particularly vulnerable to the medical effects of climate change,” Dr. Lewis said. “People with schizophrenia exhibit impaired thermoregulatory functioning, and many of our medications can impair the body’s normal heat regulation.”
The evidence of increased death rates among schizophrenia patients during heat waves has been attributed to this phenomenon as well as to the failure of patients with mental disorders to seek or obtain relief from heat, according to Dr. Lewis, but she noted that These links are true for the individual, and they affect trends in communities.
As a cause of societal stresses, such as food and water insecurity, climate change also has the very real potential of producing traumatic disruptions commensurate with disasters such as hurricanes or earthquakes. Noting that the rate of PTSD after such natural disasters typically runs at around 30%, Dr. Lewis suggested that psychiatrists might face large challenges from major upheavals induced by climate change.
However, even in the absence of catastrophic consequences, significant psychiatric morbidity may be generated by climate change in the form of “ecoanxieties” or “solastalgia,” a term coined about 10 years ago to describe psychic anxiety induced by environmental change. While many individuals continue to function normally despite fear or anxiety about climate change, Dr. Lewis said that there are many reports in the literature now show that psychoterratic illness, another term for this phenomenon, is associated with degraded or threatened environments linked to climate change.
The Climate Psychiatry Alliance is one of several professional psychiatry groups that is engaged in evaluating how psychiatry as a profession should react to climate change. The Climate Psychiatry committee of the Group for the Advancement of Psychiatry is another. Dr. Lewis, addressing the potential criticism that climate is a political issue, said that “we bring some very particular things to this 21st-century disaster … hopefully, everything I have said about the mental effects of climate change convinces you that it is not just a political problem.”
It is, however, a problem that is complex. Differentiating complex problems from complicated problems – which can be solved eventually with sufficient information – Dr. Lewis explained that complex problems are dynamic with an interplay between components that make solutions uncertain without experimentation and continual reassessment. She believes both mitigation of the problem and adaption to the inevitability of rising temperatures will be necessary.
This is relevant for psychiatrists who also must adapt to the environmental changes and develop resilience that will help them deal objectively with the mental health consequences of climate change. She noted that environmentalists recognize two traps in approaching solutions to climate change. The first is proposing overly simplistic solutions that fail to address the profound implications of climate change. The second is being rendered inactive by the overwhelming complexity of this growing problem.
Ultimately, Dr. Lewis called for psychiatrists to be proactive in dealing the mental health consequences of climate change. She noted that the APA issued a position statement in 2017, which emphasized that individuals with mental health disorders are disproportionately affected by climate change.
“We as psychiatrists know what it is to deal with complex systems, and we understand through our own work with traumatized patients how to manage patients with trauma responses and how to get empowered and engaged in the rebuilding of realistic lives,” Dr. Lewis said. She believes those skills will be important as the impact of climate change on mental health unfolds.
NEW YORK – Of the broad range of direct and indirect threats to public health anticipated from climate change, those involving mental health will place psychiatrists on the front lines of efforts to mitigate the impact, a member of the Climate Psychiatry Alliance said at the annual meeting of the American Psychiatric Association.
“One thing climate changes mean for us in psychiatry is more work,” reported Janet L. Lewis, MD, an assistant clinical professor of psychiatry at the University of Rochester (N.Y.).
Mental health is sensitive to climate. “Psychiatric patients can be particularly vulnerable to the medical effects of climate change,” Dr. Lewis said. “People with schizophrenia exhibit impaired thermoregulatory functioning, and many of our medications can impair the body’s normal heat regulation.”
The evidence of increased death rates among schizophrenia patients during heat waves has been attributed to this phenomenon as well as to the failure of patients with mental disorders to seek or obtain relief from heat, according to Dr. Lewis, but she noted that These links are true for the individual, and they affect trends in communities.
As a cause of societal stresses, such as food and water insecurity, climate change also has the very real potential of producing traumatic disruptions commensurate with disasters such as hurricanes or earthquakes. Noting that the rate of PTSD after such natural disasters typically runs at around 30%, Dr. Lewis suggested that psychiatrists might face large challenges from major upheavals induced by climate change.
However, even in the absence of catastrophic consequences, significant psychiatric morbidity may be generated by climate change in the form of “ecoanxieties” or “solastalgia,” a term coined about 10 years ago to describe psychic anxiety induced by environmental change. While many individuals continue to function normally despite fear or anxiety about climate change, Dr. Lewis said that there are many reports in the literature now show that psychoterratic illness, another term for this phenomenon, is associated with degraded or threatened environments linked to climate change.
The Climate Psychiatry Alliance is one of several professional psychiatry groups that is engaged in evaluating how psychiatry as a profession should react to climate change. The Climate Psychiatry committee of the Group for the Advancement of Psychiatry is another. Dr. Lewis, addressing the potential criticism that climate is a political issue, said that “we bring some very particular things to this 21st-century disaster … hopefully, everything I have said about the mental effects of climate change convinces you that it is not just a political problem.”
It is, however, a problem that is complex. Differentiating complex problems from complicated problems – which can be solved eventually with sufficient information – Dr. Lewis explained that complex problems are dynamic with an interplay between components that make solutions uncertain without experimentation and continual reassessment. She believes both mitigation of the problem and adaption to the inevitability of rising temperatures will be necessary.
This is relevant for psychiatrists who also must adapt to the environmental changes and develop resilience that will help them deal objectively with the mental health consequences of climate change. She noted that environmentalists recognize two traps in approaching solutions to climate change. The first is proposing overly simplistic solutions that fail to address the profound implications of climate change. The second is being rendered inactive by the overwhelming complexity of this growing problem.
Ultimately, Dr. Lewis called for psychiatrists to be proactive in dealing the mental health consequences of climate change. She noted that the APA issued a position statement in 2017, which emphasized that individuals with mental health disorders are disproportionately affected by climate change.
“We as psychiatrists know what it is to deal with complex systems, and we understand through our own work with traumatized patients how to manage patients with trauma responses and how to get empowered and engaged in the rebuilding of realistic lives,” Dr. Lewis said. She believes those skills will be important as the impact of climate change on mental health unfolds.
NEW YORK – Of the broad range of direct and indirect threats to public health anticipated from climate change, those involving mental health will place psychiatrists on the front lines of efforts to mitigate the impact, a member of the Climate Psychiatry Alliance said at the annual meeting of the American Psychiatric Association.
“One thing climate changes mean for us in psychiatry is more work,” reported Janet L. Lewis, MD, an assistant clinical professor of psychiatry at the University of Rochester (N.Y.).
Mental health is sensitive to climate. “Psychiatric patients can be particularly vulnerable to the medical effects of climate change,” Dr. Lewis said. “People with schizophrenia exhibit impaired thermoregulatory functioning, and many of our medications can impair the body’s normal heat regulation.”
The evidence of increased death rates among schizophrenia patients during heat waves has been attributed to this phenomenon as well as to the failure of patients with mental disorders to seek or obtain relief from heat, according to Dr. Lewis, but she noted that These links are true for the individual, and they affect trends in communities.
As a cause of societal stresses, such as food and water insecurity, climate change also has the very real potential of producing traumatic disruptions commensurate with disasters such as hurricanes or earthquakes. Noting that the rate of PTSD after such natural disasters typically runs at around 30%, Dr. Lewis suggested that psychiatrists might face large challenges from major upheavals induced by climate change.
However, even in the absence of catastrophic consequences, significant psychiatric morbidity may be generated by climate change in the form of “ecoanxieties” or “solastalgia,” a term coined about 10 years ago to describe psychic anxiety induced by environmental change. While many individuals continue to function normally despite fear or anxiety about climate change, Dr. Lewis said that there are many reports in the literature now show that psychoterratic illness, another term for this phenomenon, is associated with degraded or threatened environments linked to climate change.
The Climate Psychiatry Alliance is one of several professional psychiatry groups that is engaged in evaluating how psychiatry as a profession should react to climate change. The Climate Psychiatry committee of the Group for the Advancement of Psychiatry is another. Dr. Lewis, addressing the potential criticism that climate is a political issue, said that “we bring some very particular things to this 21st-century disaster … hopefully, everything I have said about the mental effects of climate change convinces you that it is not just a political problem.”
It is, however, a problem that is complex. Differentiating complex problems from complicated problems – which can be solved eventually with sufficient information – Dr. Lewis explained that complex problems are dynamic with an interplay between components that make solutions uncertain without experimentation and continual reassessment. She believes both mitigation of the problem and adaption to the inevitability of rising temperatures will be necessary.
This is relevant for psychiatrists who also must adapt to the environmental changes and develop resilience that will help them deal objectively with the mental health consequences of climate change. She noted that environmentalists recognize two traps in approaching solutions to climate change. The first is proposing overly simplistic solutions that fail to address the profound implications of climate change. The second is being rendered inactive by the overwhelming complexity of this growing problem.
Ultimately, Dr. Lewis called for psychiatrists to be proactive in dealing the mental health consequences of climate change. She noted that the APA issued a position statement in 2017, which emphasized that individuals with mental health disorders are disproportionately affected by climate change.
“We as psychiatrists know what it is to deal with complex systems, and we understand through our own work with traumatized patients how to manage patients with trauma responses and how to get empowered and engaged in the rebuilding of realistic lives,” Dr. Lewis said. She believes those skills will be important as the impact of climate change on mental health unfolds.
Ten-step trauma intervention offers help for foster families
WASHINGTON – Trauma-Informed Parenting Skills for Resource Parents, a new intervention program, might be an answer to addressing trauma symptoms in foster homes, according to a presentation at the annual conference of the Anxiety and Depression Association of America.
Rates of trauma exposure range from 80% to 93% in child welfare populations. In light of those statistics, foster parents are left to deal with the effects of traumatic stress symptoms without proper preparation or tools. Trauma-Informed Parenting Skills for Resource Parents targets different aspects of the way in which trauma can affect both the foster child and other members of the family.
The program is structured over the course of 10 weekly, 60- to 90-minute sessions for parents with foster children or those who plan to begin fostering. It is designed for caregivers of children aged 0-17 years. In addition, the intervention uses four key components: trauma awareness, caregiver relationships as the context for healing, trauma-informed parenting strategies, and creating physical and psychological safety, according to the program’s website.
“Trauma awareness is a large part of this intervention [in order to] help resource parents understand what’s happening,” Dr. Eslinger said. “There is trauma 101, orientation to what happens in the body when a child is exposed to a traumatic event, and this is followed by learning how to use the caregiver relationship.”
The 10 sessions were structured carefully, starting by addressing end goals, moving to education on the effects of early childhood trauma, transitioning to relaxation and coping skills, followed by teaching how to deal with challenging behaviors, and finishing with a final session where participants have a chance to bring it all together.
Caregivers also are instructed on using the cognitive triangle to understand their children’s feelings and build the framework to develop healthy reactions to behavior caused by traumatic stress.
“We work to help parents learn how to instill safety messages that the child needs to hear, creating a sense of safety in the home, and operating in the relationship in such a way to create psychological safety for their child,” Dr. Sprang said. “For many of [the parents], they’ve never understood that their disappointment and their hopelessness were a danger to the child – that children pick up on this.”
Neither Dr. Eslinger nor Dr. Sprang reported financial disclosures.
WASHINGTON – Trauma-Informed Parenting Skills for Resource Parents, a new intervention program, might be an answer to addressing trauma symptoms in foster homes, according to a presentation at the annual conference of the Anxiety and Depression Association of America.
Rates of trauma exposure range from 80% to 93% in child welfare populations. In light of those statistics, foster parents are left to deal with the effects of traumatic stress symptoms without proper preparation or tools. Trauma-Informed Parenting Skills for Resource Parents targets different aspects of the way in which trauma can affect both the foster child and other members of the family.
The program is structured over the course of 10 weekly, 60- to 90-minute sessions for parents with foster children or those who plan to begin fostering. It is designed for caregivers of children aged 0-17 years. In addition, the intervention uses four key components: trauma awareness, caregiver relationships as the context for healing, trauma-informed parenting strategies, and creating physical and psychological safety, according to the program’s website.
“Trauma awareness is a large part of this intervention [in order to] help resource parents understand what’s happening,” Dr. Eslinger said. “There is trauma 101, orientation to what happens in the body when a child is exposed to a traumatic event, and this is followed by learning how to use the caregiver relationship.”
The 10 sessions were structured carefully, starting by addressing end goals, moving to education on the effects of early childhood trauma, transitioning to relaxation and coping skills, followed by teaching how to deal with challenging behaviors, and finishing with a final session where participants have a chance to bring it all together.
Caregivers also are instructed on using the cognitive triangle to understand their children’s feelings and build the framework to develop healthy reactions to behavior caused by traumatic stress.
“We work to help parents learn how to instill safety messages that the child needs to hear, creating a sense of safety in the home, and operating in the relationship in such a way to create psychological safety for their child,” Dr. Sprang said. “For many of [the parents], they’ve never understood that their disappointment and their hopelessness were a danger to the child – that children pick up on this.”
Neither Dr. Eslinger nor Dr. Sprang reported financial disclosures.
WASHINGTON – Trauma-Informed Parenting Skills for Resource Parents, a new intervention program, might be an answer to addressing trauma symptoms in foster homes, according to a presentation at the annual conference of the Anxiety and Depression Association of America.
Rates of trauma exposure range from 80% to 93% in child welfare populations. In light of those statistics, foster parents are left to deal with the effects of traumatic stress symptoms without proper preparation or tools. Trauma-Informed Parenting Skills for Resource Parents targets different aspects of the way in which trauma can affect both the foster child and other members of the family.
The program is structured over the course of 10 weekly, 60- to 90-minute sessions for parents with foster children or those who plan to begin fostering. It is designed for caregivers of children aged 0-17 years. In addition, the intervention uses four key components: trauma awareness, caregiver relationships as the context for healing, trauma-informed parenting strategies, and creating physical and psychological safety, according to the program’s website.
“Trauma awareness is a large part of this intervention [in order to] help resource parents understand what’s happening,” Dr. Eslinger said. “There is trauma 101, orientation to what happens in the body when a child is exposed to a traumatic event, and this is followed by learning how to use the caregiver relationship.”
The 10 sessions were structured carefully, starting by addressing end goals, moving to education on the effects of early childhood trauma, transitioning to relaxation and coping skills, followed by teaching how to deal with challenging behaviors, and finishing with a final session where participants have a chance to bring it all together.
Caregivers also are instructed on using the cognitive triangle to understand their children’s feelings and build the framework to develop healthy reactions to behavior caused by traumatic stress.
“We work to help parents learn how to instill safety messages that the child needs to hear, creating a sense of safety in the home, and operating in the relationship in such a way to create psychological safety for their child,” Dr. Sprang said. “For many of [the parents], they’ve never understood that their disappointment and their hopelessness were a danger to the child – that children pick up on this.”
Neither Dr. Eslinger nor Dr. Sprang reported financial disclosures.
REPORTING FROM THE ANXIETY AND DEPRESSION CONFERENCE 2018
Social support after Katrina may ease depressive, PTSD symptoms
The availability of social support after a traumatic event such as a natural disaster could help buffer the development of depressive and PTSD symptoms in individuals exposed to the event, according to research published April 5.
In the Journal of Traumatic Stress, researchers reported the results of a survey of 810 adults who were exposed to the third-deadliest hurricane in U.S. history – Hurricane Katrina – in August 2005. Of those adults, 259 were displaced by the hurricane and 546 were not displaced. All of the adults were residents of Mississippi before the hurricane. More than half of the participants were women (52.2%), most were white (73.5%), and their ages ranged from 18 to 91 years.
Interviewers who were supervised by doctoral level clinicians administered numerous self-report questionnaires 18-24 months after the hurricane. Among other measures, the interviews included the Composite International Interview for DSM-IV.
The researchers found a significant negative interaction between perceived social support received in the 2 months after the hurricane and depressive symptoms, both in displaced and nondisplaced individuals.
The study also showed that the number of Katrina-related traumatic events, and whether an individual had been displaced or not, were associated with depressive symptoms – even after accounting for potential confounders, such as the number of previous traumatic events, age, and minority status.
In addition, the study explored the interaction between the number of hurricane-related traumatic events, perceived social support received, and displacement status as predictors of each cluster of PTSD symptoms. Individuals who experienced greater numbers of hurricane-related traumatic events and were displaced by the event showed more reexperiencing, avoidance, and arousal symptoms. However, social support was associated with lower likelihood of all PTSD symptom clusters.
Nondisplaced individuals who experienced a greater number of hurricane-related traumatic events showed higher arousal and avoidance symptoms, but this was only significant in individuals who reported lower levels of social support.
“Unlike previous studies, by controlling for highly correlated variables: number of previous traumatic events experienced and disaster-related stressors,” wrote Adam P. McGuire, PhD, formerly of the University of Mississippi, Jackson, and now at the Veterans Integrated Service Network, and his coauthors.
The authors also commented on the “unexpected” finding that the significant buffering effect of social support was seen both in displaced and nondisplaced residents, “which suggests that perceived social support is linked to important cognitive and behavioral processes that reduce the likelihood of developing depressive symptoms [e.g., challenging negative beliefs about self], and those effects are not limited to nondisplaced disaster survivors.”
The study was supported by the National Institutes of Health and the Midwest Regional Postdoctoral Program in Eating Disorder Research. The authors had no conflicts of interest.
SOURCE: McGuire AP et al. J Trauma Stress. 2018 Apr 5. doi: 10.1002/jts.22270.
The availability of social support after a traumatic event such as a natural disaster could help buffer the development of depressive and PTSD symptoms in individuals exposed to the event, according to research published April 5.
In the Journal of Traumatic Stress, researchers reported the results of a survey of 810 adults who were exposed to the third-deadliest hurricane in U.S. history – Hurricane Katrina – in August 2005. Of those adults, 259 were displaced by the hurricane and 546 were not displaced. All of the adults were residents of Mississippi before the hurricane. More than half of the participants were women (52.2%), most were white (73.5%), and their ages ranged from 18 to 91 years.
Interviewers who were supervised by doctoral level clinicians administered numerous self-report questionnaires 18-24 months after the hurricane. Among other measures, the interviews included the Composite International Interview for DSM-IV.
The researchers found a significant negative interaction between perceived social support received in the 2 months after the hurricane and depressive symptoms, both in displaced and nondisplaced individuals.
The study also showed that the number of Katrina-related traumatic events, and whether an individual had been displaced or not, were associated with depressive symptoms – even after accounting for potential confounders, such as the number of previous traumatic events, age, and minority status.
In addition, the study explored the interaction between the number of hurricane-related traumatic events, perceived social support received, and displacement status as predictors of each cluster of PTSD symptoms. Individuals who experienced greater numbers of hurricane-related traumatic events and were displaced by the event showed more reexperiencing, avoidance, and arousal symptoms. However, social support was associated with lower likelihood of all PTSD symptom clusters.
Nondisplaced individuals who experienced a greater number of hurricane-related traumatic events showed higher arousal and avoidance symptoms, but this was only significant in individuals who reported lower levels of social support.
“Unlike previous studies, by controlling for highly correlated variables: number of previous traumatic events experienced and disaster-related stressors,” wrote Adam P. McGuire, PhD, formerly of the University of Mississippi, Jackson, and now at the Veterans Integrated Service Network, and his coauthors.
The authors also commented on the “unexpected” finding that the significant buffering effect of social support was seen both in displaced and nondisplaced residents, “which suggests that perceived social support is linked to important cognitive and behavioral processes that reduce the likelihood of developing depressive symptoms [e.g., challenging negative beliefs about self], and those effects are not limited to nondisplaced disaster survivors.”
The study was supported by the National Institutes of Health and the Midwest Regional Postdoctoral Program in Eating Disorder Research. The authors had no conflicts of interest.
SOURCE: McGuire AP et al. J Trauma Stress. 2018 Apr 5. doi: 10.1002/jts.22270.
The availability of social support after a traumatic event such as a natural disaster could help buffer the development of depressive and PTSD symptoms in individuals exposed to the event, according to research published April 5.
In the Journal of Traumatic Stress, researchers reported the results of a survey of 810 adults who were exposed to the third-deadliest hurricane in U.S. history – Hurricane Katrina – in August 2005. Of those adults, 259 were displaced by the hurricane and 546 were not displaced. All of the adults were residents of Mississippi before the hurricane. More than half of the participants were women (52.2%), most were white (73.5%), and their ages ranged from 18 to 91 years.
Interviewers who were supervised by doctoral level clinicians administered numerous self-report questionnaires 18-24 months after the hurricane. Among other measures, the interviews included the Composite International Interview for DSM-IV.
The researchers found a significant negative interaction between perceived social support received in the 2 months after the hurricane and depressive symptoms, both in displaced and nondisplaced individuals.
The study also showed that the number of Katrina-related traumatic events, and whether an individual had been displaced or not, were associated with depressive symptoms – even after accounting for potential confounders, such as the number of previous traumatic events, age, and minority status.
In addition, the study explored the interaction between the number of hurricane-related traumatic events, perceived social support received, and displacement status as predictors of each cluster of PTSD symptoms. Individuals who experienced greater numbers of hurricane-related traumatic events and were displaced by the event showed more reexperiencing, avoidance, and arousal symptoms. However, social support was associated with lower likelihood of all PTSD symptom clusters.
Nondisplaced individuals who experienced a greater number of hurricane-related traumatic events showed higher arousal and avoidance symptoms, but this was only significant in individuals who reported lower levels of social support.
“Unlike previous studies, by controlling for highly correlated variables: number of previous traumatic events experienced and disaster-related stressors,” wrote Adam P. McGuire, PhD, formerly of the University of Mississippi, Jackson, and now at the Veterans Integrated Service Network, and his coauthors.
The authors also commented on the “unexpected” finding that the significant buffering effect of social support was seen both in displaced and nondisplaced residents, “which suggests that perceived social support is linked to important cognitive and behavioral processes that reduce the likelihood of developing depressive symptoms [e.g., challenging negative beliefs about self], and those effects are not limited to nondisplaced disaster survivors.”
The study was supported by the National Institutes of Health and the Midwest Regional Postdoctoral Program in Eating Disorder Research. The authors had no conflicts of interest.
SOURCE: McGuire AP et al. J Trauma Stress. 2018 Apr 5. doi: 10.1002/jts.22270.
FROM THE JOURNAL OF TRAUMATIC STRESS
Key clinical point: Social support can reduce the impact of traumatic events such as hurricanes.
Major finding: Perceived social support was associated with reduced depressive symptoms.
Study details: A survey of 810 adults who were exposed to Hurricane Katrina while living in Mississippi.
Disclosures: The study was supported by the National Institutes of Health and the Midwest Regional Postdoctoral Program in Eating Disorder Research. The presenters had no conflicts of interest.
Source: McGuire AP et al. J Trauma Stress. 2018 Apr 5. doi: 10.1002/jts.22270.
Understanding, Assessing, and Conceptualizing Suicide Risk Among Veterans With PTSD
Increased risk of suicide among veterans with posttraumatic stress disorder (PTSD) is well established. Posttraumatic stress disorder and related consequences are associated with higher rates of suicidal ideation and suicidal self-directed violence (S-SDV).1 Based on a systematic review, several explanations for this relationship have been hypothesized.1 Particular emphasis has been placed on trauma type (eg, premilitary childhood abuse, combat exposure), frequency of trauma exposure (ie, a single traumatic episode vs multiple traumatic experiences), specific PTSD symptoms (eg, avoidance, sleep disturbance, alteration in mood and cognitions, risky behaviors), and other psychosocial consequences associated with PTSD (eg, low social support, psychiatric comorbidity, substance use). However, there is limited understanding regarding how to conceptualize and assess risk for suicide when treating veterans who have PTSD.
PTSD and the Interpersonal-Psychological Theory of Suicide
Although PTSD is associated with risk for S-SDV among veterans, a diagnosis-specific approach to conceptualizing risk of suicide (ie, an explanation specific to PTSD) might not be enough because most individuals with a psychiatric diagnosis do not engage in S-SDV.2 Rather, theories that are able to conceptualize suicide risk across many different psychiatric diagnoses are likely to improve mental health providers’ ability to understand risk of suicide. Although many theories attempt to understand suicide risk, the Interpersonal-Psychological Theory of Suicide (IPTS) has robust empirical support.3
The IPTS proposes that suicidal ideation is driven by perceptions of stable and unchanging thwarted belongingness (TB), defined as an unmet psychological need to socially belong, and perceived burdensomeness (PB), defined as the perception that one is a burden on others.4 However, PB and TB are not considered sufficient for S-SDV to occur unless an individual also has acquired the capability for suicide. Capability for suicide is thought to happen when an individual loses the fear of dying by suicide and develops tolerance to physical pain, which is proposed to occur through habituation or repeated exposure to painful stimuli.3
Several studies have examined the IPTS in a number of clinical populations, including veterans and active-duty service members; yet limited research has applied the IPTS to veterans with PTSD.3 However, a recent article proposed that a number of PTSD-related factors increase risk of suicide through the lens of the IPTS.5 In particular, repeated exposure to painful and provocative events—especially those characterized by violence and aggression—might increase acquired capability for suicide by causing habituation to physical pain and discomfort and reducing fear of injury and death. This concept is especially concerning because of the frequent occurrence of both military- (eg, combat, military sexual trauma) and nonmilitary-related (eg, childhood abuse, intimate partner violence) stressful and traumatic events among veterans, especially individuals with PTSD.
Moreover, the acquired capability for suicide correlates highly with anxious, intrusive, and hyperarousal symptoms of PTSD.5-7 Over time, these PTSD symptoms are thought to increase habituation to the physically painful and frightening aspects of S-SDV, resulting in increased pain tolerance and fearlessness about death.3
In addition, PTSD-related cognitive-affective states (ie, thoughts and emotions), such as guilt, shame, and self-deprecation, might drive beliefs of PB and TB.5,8 Repeated exposure to such trauma-related thoughts and emotions could further reinforce beliefs of self-hate or inadequacy (PB).2 Trauma-related beliefs that the world or others are unsafe also might reduce the likelihood of seeking social support, thereby increasing TB.2 The PTSD symptoms of avoidance and self-blame also are likely to reinforce beliefs of PB and TB.2
Assessing Suicide Risk in the Context of PTSD
The IPTS framework is one that can be used by mental health providers to conceptualize risk of suicide across populations and psychiatric diagnoses, including veterans with PTSD. However, integrating additional risk assessment and management techniques is essential to guide appropriate risk stratification and treatment.
One such method of suicide risk assessment and management is therapeutic risk management (TRM).9 Therapeutic risk management involves a stratification process by which temporal aspects (ie, acute and chronic) and severity (ie, low, moderate, and high) of suicide risk are assessed using a combination of clinical interview and psychometrically sound self-report measures, such as the Beck Scale for Suicide Ideation, Beck Hopelessness Scale, and Reasons for Living Inventory. Appropriate clinical interventions that correspond to acute and chronic suicide risk stratification are then implemented (eg, safety planning, lethal means counseling, increasing frequency of care, hospitalization if warranted).
Therapeutic risk management emphasizes the necessity of assessing current and past suicidal ideation, intent, plan, and access to means. Moreover, additional considerations might be indicated when assessing and conceptualizing suicide risk among veterans with PTSD. Assessing lifetime trauma history, including traumas that occurred before, during, and after military service, is important for understanding whether traumatic experiences influence acute and chronic risks of suicide. As previously described, careful attention to stressful and traumatic experiences with violent and aggressive characteristics is recommended because research suggests that these experiences are associated with increased capability for suicide.5 Awareness of the diversity of traumatic experiences and the importance of contextual factors surrounding such experiences also are essential. For example, the nature of violence and proximity to violence (eg, directly involved in a firefight vs hearing a mortar explosion in the distance) are key components of military-related combat trauma that might differentially influence risk of suicide.10
Similarly, although military sexual trauma can include repeated threatening sexual harassment or sexual assault, research suggests that military sexual assault is particularly important for understanding suicidal ideation, and experiences of military sexual harassment are less important.11 Therefore, a careful and nuanced understanding of how contextual aspects of a veteran’s trauma history might relate to his or her chronic and acute risk of suicide is critical.
Also important is considering the individual and institutional reactions to trauma. For example, veterans whose behaviors during traumatic experiences violated their values and moral code (ie, moral injury) might be at increased risk for S-SDV. Similarly, veterans who believe that the military institution did not adequately protect them from or support them in the aftermath of traumatic experience(s) (ie, institutional betrayal) might be at higher risk of suicide.
During a clinical interview, mental health providers should pay attention to beliefs and behaviors the veteran is reporting. For example, endorsement of perceptions of low social support (eg, “no one likes me”) or self-esteem (eg, “I’m just not as good as I used to be”) might be indicative of TB or PB, respectively. Additionally, providers should be aware of current or lifetime exposure to painful stimuli (eg, nonsuicidal self-injury, such as cutting or burning, previous suicide attempts) because these exposures might increase the veteran’s acquired capability of future S-SDV.
Although unstructured clinical interviews are a common suicide risk assessment approach, TRM proposes that using a thorough clinical interview along with valid self-report measures could further illuminate a patient’s risk of suicide.9 Implementing brief measures allows mental health providers to quickly assess several risk factors and decrease the likelihood of missing important aspects of suicide risk assessment. Providers can use a number of measures to inform their suicide risk assessment, including augmenting a clinical interview of suicide risk with a valid self-report measure of recent suicidal ideation (eg, Beck Scale for Suicide Ideation, which assesses the severity of suicidal ideation in the past week).
Additionally for veterans with PTSD, mental health providers can include measures of PTSD symptoms (eg, PTSD checklist in the Diagnostic and Statistical Manual of Mental Disorders–5) and common PTSD comorbidities (eg, Beck Depression Inventory-II for depressive symptoms) that might contribute to current risk of suicide. Based on previous research, providers also might consider adding measures of trauma-related beliefs (eg, Posttraumatic Cognitions Inventory) and emotions, such as guilt (eg, Trauma-Related Guilt Inventory).5
These measures could aid in identifying modifiable risk factors of suicide among veterans with PTSD, such as the extent to which certain beliefs or emotions relate to an individual’s risk of suicide. In addition to asking about characteristics of traumatic events during the clinical interview, measures of moral injury (eg, Moral Injury Events Scale) and institutional betrayal (eg, Institutional Betrayal Questionnaire) might further inform understanding of contextual aspects of trauma that could help explain an individual’s risk of suicide.
Finally, interpersonal measures also could be helpful. For example, because avoidance and social isolation are risk factors for suicidal ideation among veterans with PTSD, measures of perceived interpersonal functioning (eg, Interpersonal Needs Questionnaire) might add further data to assist in suicide risk conceptualization. Although the selection of specific measures likely varies based on the specific needs of an individual patient, these are examples of measures that can be used with veterans with PTSD to inform suicide risk assessment and conceptualization.
By combining data from various measures across multiple domains with a thorough clinical interview, mental health providers can use a TRM approach to understand and conceptualize suicide risk among veterans with PTSD. This approach can facilitate mental health providers’ ability to provide optimal care and guide intervention(s) for veterans with PTSD. One brief intervention that has been used with veterans is safety planning. During safety planning, the provider assists the veteran in identifying warning signs, internal and external coping strategies, and individuals the veteran can reach out to for help (eg, friends and family, providers, Veterans Crisis Line), in addition to collaboratively brainstorming ways the veteran can make his or her environment safer (eg, reducing access to lethal means, identifying reminders of their reasons for living).
Specific to veterans with PTSD, symptoms such as avoidance, hyperarousal, social isolation, and beliefs that others and the world are unsafe might affect safety planning. Such symptoms could hinder identification and use of coping strategies while deterring openness to reach out to others for help. A collaborative method can be used to identify alternate means of coping that take into account PTSD-related avoidance and hyperarousal (eg, rather than going to a crowded store or isolating at home, taking a walk in a quiet park with few people). Similarly, because substance use and risky behaviors are common among veterans with PTSD and might further increase risk of suicide, exploring healthy (eg, exercise) vs unhealthy (eg, substance use; unprotected sex) coping strategies could be helpful.
Further, based on their lived experience, veterans with PTSD could experience difficulty identifying a support system or be reluctant to reach out to others during acute crisis. This might be particularly daunting in the presence of PB and TB. In these situations, it is important to validate the veteran’s difficulty with reaching out while simultaneously encouraging the veteran to examine the accuracy of such beliefs and/or helping the veteran develop skills to overcome these obstacles.
The mental health provider also can work with the individual to ensure that the veteran understands that if he or she does engage emergency resources (eg, Veterans Crisis Line), information likely will be held confidential. Providers can tell their patients that breaks in confidentiality are rare and occur only in circumstances in which it is necessary to protect the veteran. In doing so, the provider facilitates the veteran’s understanding of the role of crisis resources and clarifies any misconceptions the veteran might have (eg, calling the crisis line will always result in hospitalization or police presence).
Conclusion
Several PTSD-related factors might increase PB, TB, and the acquired capability for suicide among veterans with PTSD. Because suicide risk assessment and management can be time sensitive and anxiety provoking, mental health providers can use a TRM approach to increase their confidence in instituting optimal care and mitigating risk by having a structured, therapeutic assessment process that gathers appropriate suicide- and PTSD-related data to assist in developing suicide risk-related treatment. However, more research is needed to determine the most useful self-report measures and effective interventions when working with veterans with PTSD at risk of suicide.
1. Pompili M, Sher L, Serafini G, et al. Posttraumatic stress disorder and suicide risk among veterans: a literature review
2. Goldsmith SK, Pellmar TC, Kleinman AM, Bunney WE, eds. Reducing Suicide: A National Imperative. Washington, DC: The National Academies Press; 2002.
3. Chu C, Buchman-Schmitt JM, Stanley IH, et al. The interpersonal psychological theory of suicide: a systematic review and meta-analysis of a decade of cross-national research. Psychol Bull. 2017;143(12):1313-1345.
4. Van Orden KA, Witte TK, Cukrowicz KC, et al. The interpersonal theory of suicide. Psychol Rev. 2010;117(2):575-600.
5. Bryan CJ, Grove JL, Kimbrel NA. Theory-driven models of self-directed violence among individuals with PTSD. Curr Opin Psychol. 2017;14:12-17.
6. Bryan CJ, Anestis M. Reexperiencing symptoms and the interpersonal-psychological theory of suicidal behavior among deployed service members evaluated for traumatic brain injury. J Clin Psychol. 2011;67(9):856-865.
7. Zuromski KL, Davis MT, Witte TK, Weathers F, Blevins C. PTSD symptom clusters are differentially associated with components of the acquired capability for suicide. Suicide Life Threat Behav. 2014;44(6):682-697.
8. Davis MT, Witte TK, Weathers FW, Blevins CA. The role of posttraumatic stress disorder symptom clusters in the prediction of passive suicidal ideation. Psychol Trauma. 2014;6(suppl 1):S82-S91.
9. Wortzel HS, Matarazzo B, Homaifar B. A model for therapeutic risk management of the suicidal patient. J Psychiatr Pract. 2013;19(4):323-326.
10. Bryan CJ, Cukrowicz KC. Associations between types of combat violence and the acquired capability for suicide. Suicide Life Threat Behav. 2011;41(2):126-136.
11. Monteith LL, Menefee DS, Forster JE, Bahraini NH. A closer examination of sexual trauma during deployment: not all sexual traumas are associated with suicidal ideation. Suicide Life Threat Behav. 2016;46(1):46-54.
Increased risk of suicide among veterans with posttraumatic stress disorder (PTSD) is well established. Posttraumatic stress disorder and related consequences are associated with higher rates of suicidal ideation and suicidal self-directed violence (S-SDV).1 Based on a systematic review, several explanations for this relationship have been hypothesized.1 Particular emphasis has been placed on trauma type (eg, premilitary childhood abuse, combat exposure), frequency of trauma exposure (ie, a single traumatic episode vs multiple traumatic experiences), specific PTSD symptoms (eg, avoidance, sleep disturbance, alteration in mood and cognitions, risky behaviors), and other psychosocial consequences associated with PTSD (eg, low social support, psychiatric comorbidity, substance use). However, there is limited understanding regarding how to conceptualize and assess risk for suicide when treating veterans who have PTSD.
PTSD and the Interpersonal-Psychological Theory of Suicide
Although PTSD is associated with risk for S-SDV among veterans, a diagnosis-specific approach to conceptualizing risk of suicide (ie, an explanation specific to PTSD) might not be enough because most individuals with a psychiatric diagnosis do not engage in S-SDV.2 Rather, theories that are able to conceptualize suicide risk across many different psychiatric diagnoses are likely to improve mental health providers’ ability to understand risk of suicide. Although many theories attempt to understand suicide risk, the Interpersonal-Psychological Theory of Suicide (IPTS) has robust empirical support.3
The IPTS proposes that suicidal ideation is driven by perceptions of stable and unchanging thwarted belongingness (TB), defined as an unmet psychological need to socially belong, and perceived burdensomeness (PB), defined as the perception that one is a burden on others.4 However, PB and TB are not considered sufficient for S-SDV to occur unless an individual also has acquired the capability for suicide. Capability for suicide is thought to happen when an individual loses the fear of dying by suicide and develops tolerance to physical pain, which is proposed to occur through habituation or repeated exposure to painful stimuli.3
Several studies have examined the IPTS in a number of clinical populations, including veterans and active-duty service members; yet limited research has applied the IPTS to veterans with PTSD.3 However, a recent article proposed that a number of PTSD-related factors increase risk of suicide through the lens of the IPTS.5 In particular, repeated exposure to painful and provocative events—especially those characterized by violence and aggression—might increase acquired capability for suicide by causing habituation to physical pain and discomfort and reducing fear of injury and death. This concept is especially concerning because of the frequent occurrence of both military- (eg, combat, military sexual trauma) and nonmilitary-related (eg, childhood abuse, intimate partner violence) stressful and traumatic events among veterans, especially individuals with PTSD.
Moreover, the acquired capability for suicide correlates highly with anxious, intrusive, and hyperarousal symptoms of PTSD.5-7 Over time, these PTSD symptoms are thought to increase habituation to the physically painful and frightening aspects of S-SDV, resulting in increased pain tolerance and fearlessness about death.3
In addition, PTSD-related cognitive-affective states (ie, thoughts and emotions), such as guilt, shame, and self-deprecation, might drive beliefs of PB and TB.5,8 Repeated exposure to such trauma-related thoughts and emotions could further reinforce beliefs of self-hate or inadequacy (PB).2 Trauma-related beliefs that the world or others are unsafe also might reduce the likelihood of seeking social support, thereby increasing TB.2 The PTSD symptoms of avoidance and self-blame also are likely to reinforce beliefs of PB and TB.2
Assessing Suicide Risk in the Context of PTSD
The IPTS framework is one that can be used by mental health providers to conceptualize risk of suicide across populations and psychiatric diagnoses, including veterans with PTSD. However, integrating additional risk assessment and management techniques is essential to guide appropriate risk stratification and treatment.
One such method of suicide risk assessment and management is therapeutic risk management (TRM).9 Therapeutic risk management involves a stratification process by which temporal aspects (ie, acute and chronic) and severity (ie, low, moderate, and high) of suicide risk are assessed using a combination of clinical interview and psychometrically sound self-report measures, such as the Beck Scale for Suicide Ideation, Beck Hopelessness Scale, and Reasons for Living Inventory. Appropriate clinical interventions that correspond to acute and chronic suicide risk stratification are then implemented (eg, safety planning, lethal means counseling, increasing frequency of care, hospitalization if warranted).
Therapeutic risk management emphasizes the necessity of assessing current and past suicidal ideation, intent, plan, and access to means. Moreover, additional considerations might be indicated when assessing and conceptualizing suicide risk among veterans with PTSD. Assessing lifetime trauma history, including traumas that occurred before, during, and after military service, is important for understanding whether traumatic experiences influence acute and chronic risks of suicide. As previously described, careful attention to stressful and traumatic experiences with violent and aggressive characteristics is recommended because research suggests that these experiences are associated with increased capability for suicide.5 Awareness of the diversity of traumatic experiences and the importance of contextual factors surrounding such experiences also are essential. For example, the nature of violence and proximity to violence (eg, directly involved in a firefight vs hearing a mortar explosion in the distance) are key components of military-related combat trauma that might differentially influence risk of suicide.10
Similarly, although military sexual trauma can include repeated threatening sexual harassment or sexual assault, research suggests that military sexual assault is particularly important for understanding suicidal ideation, and experiences of military sexual harassment are less important.11 Therefore, a careful and nuanced understanding of how contextual aspects of a veteran’s trauma history might relate to his or her chronic and acute risk of suicide is critical.
Also important is considering the individual and institutional reactions to trauma. For example, veterans whose behaviors during traumatic experiences violated their values and moral code (ie, moral injury) might be at increased risk for S-SDV. Similarly, veterans who believe that the military institution did not adequately protect them from or support them in the aftermath of traumatic experience(s) (ie, institutional betrayal) might be at higher risk of suicide.
During a clinical interview, mental health providers should pay attention to beliefs and behaviors the veteran is reporting. For example, endorsement of perceptions of low social support (eg, “no one likes me”) or self-esteem (eg, “I’m just not as good as I used to be”) might be indicative of TB or PB, respectively. Additionally, providers should be aware of current or lifetime exposure to painful stimuli (eg, nonsuicidal self-injury, such as cutting or burning, previous suicide attempts) because these exposures might increase the veteran’s acquired capability of future S-SDV.
Although unstructured clinical interviews are a common suicide risk assessment approach, TRM proposes that using a thorough clinical interview along with valid self-report measures could further illuminate a patient’s risk of suicide.9 Implementing brief measures allows mental health providers to quickly assess several risk factors and decrease the likelihood of missing important aspects of suicide risk assessment. Providers can use a number of measures to inform their suicide risk assessment, including augmenting a clinical interview of suicide risk with a valid self-report measure of recent suicidal ideation (eg, Beck Scale for Suicide Ideation, which assesses the severity of suicidal ideation in the past week).
Additionally for veterans with PTSD, mental health providers can include measures of PTSD symptoms (eg, PTSD checklist in the Diagnostic and Statistical Manual of Mental Disorders–5) and common PTSD comorbidities (eg, Beck Depression Inventory-II for depressive symptoms) that might contribute to current risk of suicide. Based on previous research, providers also might consider adding measures of trauma-related beliefs (eg, Posttraumatic Cognitions Inventory) and emotions, such as guilt (eg, Trauma-Related Guilt Inventory).5
These measures could aid in identifying modifiable risk factors of suicide among veterans with PTSD, such as the extent to which certain beliefs or emotions relate to an individual’s risk of suicide. In addition to asking about characteristics of traumatic events during the clinical interview, measures of moral injury (eg, Moral Injury Events Scale) and institutional betrayal (eg, Institutional Betrayal Questionnaire) might further inform understanding of contextual aspects of trauma that could help explain an individual’s risk of suicide.
Finally, interpersonal measures also could be helpful. For example, because avoidance and social isolation are risk factors for suicidal ideation among veterans with PTSD, measures of perceived interpersonal functioning (eg, Interpersonal Needs Questionnaire) might add further data to assist in suicide risk conceptualization. Although the selection of specific measures likely varies based on the specific needs of an individual patient, these are examples of measures that can be used with veterans with PTSD to inform suicide risk assessment and conceptualization.
By combining data from various measures across multiple domains with a thorough clinical interview, mental health providers can use a TRM approach to understand and conceptualize suicide risk among veterans with PTSD. This approach can facilitate mental health providers’ ability to provide optimal care and guide intervention(s) for veterans with PTSD. One brief intervention that has been used with veterans is safety planning. During safety planning, the provider assists the veteran in identifying warning signs, internal and external coping strategies, and individuals the veteran can reach out to for help (eg, friends and family, providers, Veterans Crisis Line), in addition to collaboratively brainstorming ways the veteran can make his or her environment safer (eg, reducing access to lethal means, identifying reminders of their reasons for living).
Specific to veterans with PTSD, symptoms such as avoidance, hyperarousal, social isolation, and beliefs that others and the world are unsafe might affect safety planning. Such symptoms could hinder identification and use of coping strategies while deterring openness to reach out to others for help. A collaborative method can be used to identify alternate means of coping that take into account PTSD-related avoidance and hyperarousal (eg, rather than going to a crowded store or isolating at home, taking a walk in a quiet park with few people). Similarly, because substance use and risky behaviors are common among veterans with PTSD and might further increase risk of suicide, exploring healthy (eg, exercise) vs unhealthy (eg, substance use; unprotected sex) coping strategies could be helpful.
Further, based on their lived experience, veterans with PTSD could experience difficulty identifying a support system or be reluctant to reach out to others during acute crisis. This might be particularly daunting in the presence of PB and TB. In these situations, it is important to validate the veteran’s difficulty with reaching out while simultaneously encouraging the veteran to examine the accuracy of such beliefs and/or helping the veteran develop skills to overcome these obstacles.
The mental health provider also can work with the individual to ensure that the veteran understands that if he or she does engage emergency resources (eg, Veterans Crisis Line), information likely will be held confidential. Providers can tell their patients that breaks in confidentiality are rare and occur only in circumstances in which it is necessary to protect the veteran. In doing so, the provider facilitates the veteran’s understanding of the role of crisis resources and clarifies any misconceptions the veteran might have (eg, calling the crisis line will always result in hospitalization or police presence).
Conclusion
Several PTSD-related factors might increase PB, TB, and the acquired capability for suicide among veterans with PTSD. Because suicide risk assessment and management can be time sensitive and anxiety provoking, mental health providers can use a TRM approach to increase their confidence in instituting optimal care and mitigating risk by having a structured, therapeutic assessment process that gathers appropriate suicide- and PTSD-related data to assist in developing suicide risk-related treatment. However, more research is needed to determine the most useful self-report measures and effective interventions when working with veterans with PTSD at risk of suicide.
Increased risk of suicide among veterans with posttraumatic stress disorder (PTSD) is well established. Posttraumatic stress disorder and related consequences are associated with higher rates of suicidal ideation and suicidal self-directed violence (S-SDV).1 Based on a systematic review, several explanations for this relationship have been hypothesized.1 Particular emphasis has been placed on trauma type (eg, premilitary childhood abuse, combat exposure), frequency of trauma exposure (ie, a single traumatic episode vs multiple traumatic experiences), specific PTSD symptoms (eg, avoidance, sleep disturbance, alteration in mood and cognitions, risky behaviors), and other psychosocial consequences associated with PTSD (eg, low social support, psychiatric comorbidity, substance use). However, there is limited understanding regarding how to conceptualize and assess risk for suicide when treating veterans who have PTSD.
PTSD and the Interpersonal-Psychological Theory of Suicide
Although PTSD is associated with risk for S-SDV among veterans, a diagnosis-specific approach to conceptualizing risk of suicide (ie, an explanation specific to PTSD) might not be enough because most individuals with a psychiatric diagnosis do not engage in S-SDV.2 Rather, theories that are able to conceptualize suicide risk across many different psychiatric diagnoses are likely to improve mental health providers’ ability to understand risk of suicide. Although many theories attempt to understand suicide risk, the Interpersonal-Psychological Theory of Suicide (IPTS) has robust empirical support.3
The IPTS proposes that suicidal ideation is driven by perceptions of stable and unchanging thwarted belongingness (TB), defined as an unmet psychological need to socially belong, and perceived burdensomeness (PB), defined as the perception that one is a burden on others.4 However, PB and TB are not considered sufficient for S-SDV to occur unless an individual also has acquired the capability for suicide. Capability for suicide is thought to happen when an individual loses the fear of dying by suicide and develops tolerance to physical pain, which is proposed to occur through habituation or repeated exposure to painful stimuli.3
Several studies have examined the IPTS in a number of clinical populations, including veterans and active-duty service members; yet limited research has applied the IPTS to veterans with PTSD.3 However, a recent article proposed that a number of PTSD-related factors increase risk of suicide through the lens of the IPTS.5 In particular, repeated exposure to painful and provocative events—especially those characterized by violence and aggression—might increase acquired capability for suicide by causing habituation to physical pain and discomfort and reducing fear of injury and death. This concept is especially concerning because of the frequent occurrence of both military- (eg, combat, military sexual trauma) and nonmilitary-related (eg, childhood abuse, intimate partner violence) stressful and traumatic events among veterans, especially individuals with PTSD.
Moreover, the acquired capability for suicide correlates highly with anxious, intrusive, and hyperarousal symptoms of PTSD.5-7 Over time, these PTSD symptoms are thought to increase habituation to the physically painful and frightening aspects of S-SDV, resulting in increased pain tolerance and fearlessness about death.3
In addition, PTSD-related cognitive-affective states (ie, thoughts and emotions), such as guilt, shame, and self-deprecation, might drive beliefs of PB and TB.5,8 Repeated exposure to such trauma-related thoughts and emotions could further reinforce beliefs of self-hate or inadequacy (PB).2 Trauma-related beliefs that the world or others are unsafe also might reduce the likelihood of seeking social support, thereby increasing TB.2 The PTSD symptoms of avoidance and self-blame also are likely to reinforce beliefs of PB and TB.2
Assessing Suicide Risk in the Context of PTSD
The IPTS framework is one that can be used by mental health providers to conceptualize risk of suicide across populations and psychiatric diagnoses, including veterans with PTSD. However, integrating additional risk assessment and management techniques is essential to guide appropriate risk stratification and treatment.
One such method of suicide risk assessment and management is therapeutic risk management (TRM).9 Therapeutic risk management involves a stratification process by which temporal aspects (ie, acute and chronic) and severity (ie, low, moderate, and high) of suicide risk are assessed using a combination of clinical interview and psychometrically sound self-report measures, such as the Beck Scale for Suicide Ideation, Beck Hopelessness Scale, and Reasons for Living Inventory. Appropriate clinical interventions that correspond to acute and chronic suicide risk stratification are then implemented (eg, safety planning, lethal means counseling, increasing frequency of care, hospitalization if warranted).
Therapeutic risk management emphasizes the necessity of assessing current and past suicidal ideation, intent, plan, and access to means. Moreover, additional considerations might be indicated when assessing and conceptualizing suicide risk among veterans with PTSD. Assessing lifetime trauma history, including traumas that occurred before, during, and after military service, is important for understanding whether traumatic experiences influence acute and chronic risks of suicide. As previously described, careful attention to stressful and traumatic experiences with violent and aggressive characteristics is recommended because research suggests that these experiences are associated with increased capability for suicide.5 Awareness of the diversity of traumatic experiences and the importance of contextual factors surrounding such experiences also are essential. For example, the nature of violence and proximity to violence (eg, directly involved in a firefight vs hearing a mortar explosion in the distance) are key components of military-related combat trauma that might differentially influence risk of suicide.10
Similarly, although military sexual trauma can include repeated threatening sexual harassment or sexual assault, research suggests that military sexual assault is particularly important for understanding suicidal ideation, and experiences of military sexual harassment are less important.11 Therefore, a careful and nuanced understanding of how contextual aspects of a veteran’s trauma history might relate to his or her chronic and acute risk of suicide is critical.
Also important is considering the individual and institutional reactions to trauma. For example, veterans whose behaviors during traumatic experiences violated their values and moral code (ie, moral injury) might be at increased risk for S-SDV. Similarly, veterans who believe that the military institution did not adequately protect them from or support them in the aftermath of traumatic experience(s) (ie, institutional betrayal) might be at higher risk of suicide.
During a clinical interview, mental health providers should pay attention to beliefs and behaviors the veteran is reporting. For example, endorsement of perceptions of low social support (eg, “no one likes me”) or self-esteem (eg, “I’m just not as good as I used to be”) might be indicative of TB or PB, respectively. Additionally, providers should be aware of current or lifetime exposure to painful stimuli (eg, nonsuicidal self-injury, such as cutting or burning, previous suicide attempts) because these exposures might increase the veteran’s acquired capability of future S-SDV.
Although unstructured clinical interviews are a common suicide risk assessment approach, TRM proposes that using a thorough clinical interview along with valid self-report measures could further illuminate a patient’s risk of suicide.9 Implementing brief measures allows mental health providers to quickly assess several risk factors and decrease the likelihood of missing important aspects of suicide risk assessment. Providers can use a number of measures to inform their suicide risk assessment, including augmenting a clinical interview of suicide risk with a valid self-report measure of recent suicidal ideation (eg, Beck Scale for Suicide Ideation, which assesses the severity of suicidal ideation in the past week).
Additionally for veterans with PTSD, mental health providers can include measures of PTSD symptoms (eg, PTSD checklist in the Diagnostic and Statistical Manual of Mental Disorders–5) and common PTSD comorbidities (eg, Beck Depression Inventory-II for depressive symptoms) that might contribute to current risk of suicide. Based on previous research, providers also might consider adding measures of trauma-related beliefs (eg, Posttraumatic Cognitions Inventory) and emotions, such as guilt (eg, Trauma-Related Guilt Inventory).5
These measures could aid in identifying modifiable risk factors of suicide among veterans with PTSD, such as the extent to which certain beliefs or emotions relate to an individual’s risk of suicide. In addition to asking about characteristics of traumatic events during the clinical interview, measures of moral injury (eg, Moral Injury Events Scale) and institutional betrayal (eg, Institutional Betrayal Questionnaire) might further inform understanding of contextual aspects of trauma that could help explain an individual’s risk of suicide.
Finally, interpersonal measures also could be helpful. For example, because avoidance and social isolation are risk factors for suicidal ideation among veterans with PTSD, measures of perceived interpersonal functioning (eg, Interpersonal Needs Questionnaire) might add further data to assist in suicide risk conceptualization. Although the selection of specific measures likely varies based on the specific needs of an individual patient, these are examples of measures that can be used with veterans with PTSD to inform suicide risk assessment and conceptualization.
By combining data from various measures across multiple domains with a thorough clinical interview, mental health providers can use a TRM approach to understand and conceptualize suicide risk among veterans with PTSD. This approach can facilitate mental health providers’ ability to provide optimal care and guide intervention(s) for veterans with PTSD. One brief intervention that has been used with veterans is safety planning. During safety planning, the provider assists the veteran in identifying warning signs, internal and external coping strategies, and individuals the veteran can reach out to for help (eg, friends and family, providers, Veterans Crisis Line), in addition to collaboratively brainstorming ways the veteran can make his or her environment safer (eg, reducing access to lethal means, identifying reminders of their reasons for living).
Specific to veterans with PTSD, symptoms such as avoidance, hyperarousal, social isolation, and beliefs that others and the world are unsafe might affect safety planning. Such symptoms could hinder identification and use of coping strategies while deterring openness to reach out to others for help. A collaborative method can be used to identify alternate means of coping that take into account PTSD-related avoidance and hyperarousal (eg, rather than going to a crowded store or isolating at home, taking a walk in a quiet park with few people). Similarly, because substance use and risky behaviors are common among veterans with PTSD and might further increase risk of suicide, exploring healthy (eg, exercise) vs unhealthy (eg, substance use; unprotected sex) coping strategies could be helpful.
Further, based on their lived experience, veterans with PTSD could experience difficulty identifying a support system or be reluctant to reach out to others during acute crisis. This might be particularly daunting in the presence of PB and TB. In these situations, it is important to validate the veteran’s difficulty with reaching out while simultaneously encouraging the veteran to examine the accuracy of such beliefs and/or helping the veteran develop skills to overcome these obstacles.
The mental health provider also can work with the individual to ensure that the veteran understands that if he or she does engage emergency resources (eg, Veterans Crisis Line), information likely will be held confidential. Providers can tell their patients that breaks in confidentiality are rare and occur only in circumstances in which it is necessary to protect the veteran. In doing so, the provider facilitates the veteran’s understanding of the role of crisis resources and clarifies any misconceptions the veteran might have (eg, calling the crisis line will always result in hospitalization or police presence).
Conclusion
Several PTSD-related factors might increase PB, TB, and the acquired capability for suicide among veterans with PTSD. Because suicide risk assessment and management can be time sensitive and anxiety provoking, mental health providers can use a TRM approach to increase their confidence in instituting optimal care and mitigating risk by having a structured, therapeutic assessment process that gathers appropriate suicide- and PTSD-related data to assist in developing suicide risk-related treatment. However, more research is needed to determine the most useful self-report measures and effective interventions when working with veterans with PTSD at risk of suicide.
1. Pompili M, Sher L, Serafini G, et al. Posttraumatic stress disorder and suicide risk among veterans: a literature review
2. Goldsmith SK, Pellmar TC, Kleinman AM, Bunney WE, eds. Reducing Suicide: A National Imperative. Washington, DC: The National Academies Press; 2002.
3. Chu C, Buchman-Schmitt JM, Stanley IH, et al. The interpersonal psychological theory of suicide: a systematic review and meta-analysis of a decade of cross-national research. Psychol Bull. 2017;143(12):1313-1345.
4. Van Orden KA, Witte TK, Cukrowicz KC, et al. The interpersonal theory of suicide. Psychol Rev. 2010;117(2):575-600.
5. Bryan CJ, Grove JL, Kimbrel NA. Theory-driven models of self-directed violence among individuals with PTSD. Curr Opin Psychol. 2017;14:12-17.
6. Bryan CJ, Anestis M. Reexperiencing symptoms and the interpersonal-psychological theory of suicidal behavior among deployed service members evaluated for traumatic brain injury. J Clin Psychol. 2011;67(9):856-865.
7. Zuromski KL, Davis MT, Witte TK, Weathers F, Blevins C. PTSD symptom clusters are differentially associated with components of the acquired capability for suicide. Suicide Life Threat Behav. 2014;44(6):682-697.
8. Davis MT, Witte TK, Weathers FW, Blevins CA. The role of posttraumatic stress disorder symptom clusters in the prediction of passive suicidal ideation. Psychol Trauma. 2014;6(suppl 1):S82-S91.
9. Wortzel HS, Matarazzo B, Homaifar B. A model for therapeutic risk management of the suicidal patient. J Psychiatr Pract. 2013;19(4):323-326.
10. Bryan CJ, Cukrowicz KC. Associations between types of combat violence and the acquired capability for suicide. Suicide Life Threat Behav. 2011;41(2):126-136.
11. Monteith LL, Menefee DS, Forster JE, Bahraini NH. A closer examination of sexual trauma during deployment: not all sexual traumas are associated with suicidal ideation. Suicide Life Threat Behav. 2016;46(1):46-54.
1. Pompili M, Sher L, Serafini G, et al. Posttraumatic stress disorder and suicide risk among veterans: a literature review
2. Goldsmith SK, Pellmar TC, Kleinman AM, Bunney WE, eds. Reducing Suicide: A National Imperative. Washington, DC: The National Academies Press; 2002.
3. Chu C, Buchman-Schmitt JM, Stanley IH, et al. The interpersonal psychological theory of suicide: a systematic review and meta-analysis of a decade of cross-national research. Psychol Bull. 2017;143(12):1313-1345.
4. Van Orden KA, Witte TK, Cukrowicz KC, et al. The interpersonal theory of suicide. Psychol Rev. 2010;117(2):575-600.
5. Bryan CJ, Grove JL, Kimbrel NA. Theory-driven models of self-directed violence among individuals with PTSD. Curr Opin Psychol. 2017;14:12-17.
6. Bryan CJ, Anestis M. Reexperiencing symptoms and the interpersonal-psychological theory of suicidal behavior among deployed service members evaluated for traumatic brain injury. J Clin Psychol. 2011;67(9):856-865.
7. Zuromski KL, Davis MT, Witte TK, Weathers F, Blevins C. PTSD symptom clusters are differentially associated with components of the acquired capability for suicide. Suicide Life Threat Behav. 2014;44(6):682-697.
8. Davis MT, Witte TK, Weathers FW, Blevins CA. The role of posttraumatic stress disorder symptom clusters in the prediction of passive suicidal ideation. Psychol Trauma. 2014;6(suppl 1):S82-S91.
9. Wortzel HS, Matarazzo B, Homaifar B. A model for therapeutic risk management of the suicidal patient. J Psychiatr Pract. 2013;19(4):323-326.
10. Bryan CJ, Cukrowicz KC. Associations between types of combat violence and the acquired capability for suicide. Suicide Life Threat Behav. 2011;41(2):126-136.
11. Monteith LL, Menefee DS, Forster JE, Bahraini NH. A closer examination of sexual trauma during deployment: not all sexual traumas are associated with suicidal ideation. Suicide Life Threat Behav. 2016;46(1):46-54.
PTSD: A systematic approach to diagnosis and treatment
Posttraumatic stress disorder (PTSD) has increasingly become a part of American culture since its introduction in the American Psychiatric Association’s third edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III) in 1980.1 Since then, a proliferation of material about this disorder—both academic and popular—has been generated, yet much confusion persists surrounding the definition of the disorder, its prevalence, and its management. This review addresses the essential elements for diagnosis and treatment of PTSD.
Diagnosis: A closer look at the criteria
Criteria for the diagnosis of PTSD have evolved since 1980, with changes in the definition of trauma and the addition of symptoms and symptom groups.2 Table 13 summarizes the current DSM-5 criteria for PTSD.

Trauma exposure. An essential first step in the diagnosis of PTSD is to determine whether the individual has experienced exposure to trauma. This concept is defined in Criterion A (trauma exposure).3 PTSD is nonconformist among the psychiatric diagnoses in that it requires a specific external event as part of its definition. Misapplication of the trauma exposure criterion by many clinicians and researchers has led to misdiagnosis and erroneously high prevalence estimates of PTSD.4,5
A traumatic event is one that represents a threat to life or limb, specifically defined as “actual or threatened death, serious injury, or sexual violence.”3 DSM-5 does not allow for just any stressful event to be considered trauma. For example, no matter how distressing, failing an important test at school or being served with divorce proceedings do not represent a requisite trauma6 because these examples do not entail a threat to life or limb.
DSM-5 PTSD Criterion A also requires a qualifying exposure to the traumatic event. There are 4 types of qualifying exposures:
- direct experience of immediate serious physical danger
- eyewitness of trauma to others
- indirect exposure via violent or accidental trauma experienced by a close family member or close friend
- repeated or extreme exposure to aversive details of trauma, such as first responders collecting human remains or law enforcement officers being repeatedly exposed to horrific details of child abuse.3
Witnessed trauma must be in person; thus, viewing trauma in media reports would not constitute a qualifying exposure. Indirect trauma exposure can occur through learning of the experience of a qualifying trauma exposure by a close family member or personal friend.
It is critical to differentiate exposure to trauma (an objective construct) from the subjective distress that may be associated with it. If trauma has not occurred or a qualifying exposure is not established, no amount of distress associated with it can establish the experience as meeting Criterion A for PTSD. This does not mean that nonqualifying experiences of stressful events are not distressing; in fact, such experiences can result in substantial psychological angst. Conversely, exposure to trauma is not tantamount to a diagnosis of PTSD, as most trauma exposures do not result in PTSD.7,8
Continue to: Symptom groups
Symptom groups. DSM-5 symptom criteria for PTSD include 4 symptom groups, Criteria B to E, respectively:
- intrusion
- avoidance
- negative cognitions and mood (numbing)
- hyperarousal/reactivity.
A specific number of symptoms must be present in all 4 of the symptom groups to fulfill diagnostic criteria. Importantly, these symptoms must be linked temporally and conceptually to the traumatic exposure to qualify as PTSD symptoms. Specifically, the symptoms must be new or substantially worsened after the event. For example, continuing sleep disturbance in someone who has had lifetime difficulty sleeping would not count as a trauma-related symptom. Most symptom checklists do not properly assess diagnostic criteria for PTSD because they do not anchor the symptoms in an exposure to a traumatic event; diagnosis requires an interview to fully assess all the diagnostic criteria. Finally, the symptoms must have been present for >1 month for the diagnosis, and the symptoms must have resulted in clinically significant distress or functional impairment to qualify.
The Algorithm provides a practical way to systematically assess all DSM-5 criteria for PTSD to arrive at a diagnosis. The clinician begins by determining whether a traumatic event has occurred and whether the individual had a qualifying exposure to it. If not, PTSD cannot be diagnosed. Alternative diagnoses to consider for new disorders that arise in the context of trauma among patients who are not exposed to trauma include major depressive disorder, adjustment disorder, and bereavement, as well as acute stress disorder (which is not validated but has potential utility as a billable diagnosis).
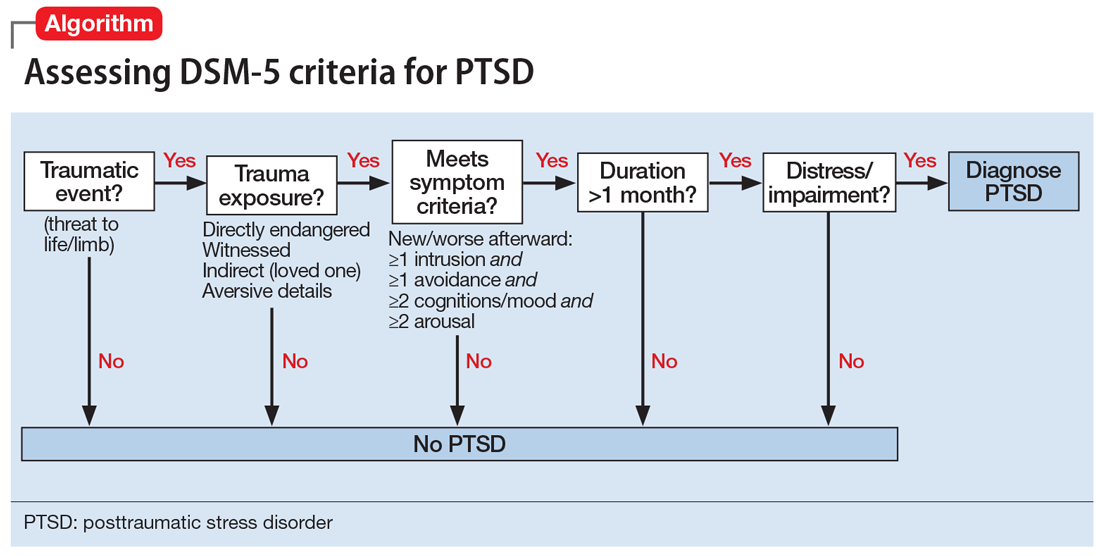
Avoidance and numbing symptoms (present in Criteria C and D) have been shown to represent markers of illness and can be useful in predicting PTSD.8-10 Unlike symptoms of intrusion and hyperarousal (Criteria B and E, respectively), which are very common and by themselves are nonpathological, avoidance/numbing symptoms occur much less commonly, are associated with functional impairment and other indicators of illness, and are strongly associated with PTSD.6 Prominent avoidance/numbing profiles have been demonstrated to predict PTSD in the first 1 to 2 weeks after trauma exposure, before PTSD can be formally diagnosed.11 Posttraumatic stress symptoms are nearly universal after trauma exposure, even in people who do not develop PTSD.5 Intrusion and hyperarousal symptoms constitute most of such symptoms,7 and these symptoms in the absence of prominent avoidance/numbing can be considered normative distress responses to trauma exposure.12
Some PTSD symptoms may seem quite similar to symptoms of depressive disorders and anxiety disorders. PTSD can be differentiated from these other disorders by linking the symptoms temporally and contextually to a qualifying exposure to a traumatic event. More often than not, PTSD presents with comorbid psychiatric disorders, especially depressive disorders, anxiety disorders, and/or substance use disorders.
Continue to: Treatment: Medication, psychotherapy, or both
Treatment: Medication, psychotherapy, or both
Both pharmacotherapy and psychotherapy—as monotherapy or in combination—are beneficial for treatment of PTSD. Research has not conclusively shown either treatment modality to be superior, because adequate head-to-head trials have not been conducted.4 Therefore, the choice of initial treatment is based on individual circumstances, such as patient preference for medication and/or psychotherapy, or the availability of therapists trained in evidence-based PTSD psychotherapy. Pharmacotherapeutic approaches are considered especially beneficial for depressive- and anxiety-like symptoms of PTSD, and trauma-focused psychotherapies are presumed to address the neuropathology of conditioned fear and anxiety responses involved in PTSD.14 Table 214-25 provides a list of published treatment guidelines and reviews to help clinicians seeking further detail beyond that provided in this article.
Antidepressants are the mainstay of pharmacotherapy for PTSD. These medications are effective for treating major depressive disorder, and have beneficial properties for PTSD independent of their antidepressant effects. The serotonin selective reuptake inhibitors (SSRIs) sertraline and paroxetine are FDA-approved for the treatment of PTSD.6 Other recommended medications include the serotonin-norepinephrine reuptake inhibitor (SNRI) venlafaxine, and nefazodone, an atypical serotoninergic agent.13 Other antidepressants with less published evidence of effectiveness are used as second-line pharmacotherapies for PTSD, including fluoxetine (SSRI), and mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA).4 Older medications, such as the tricyclic antidepressant amitriptyline and the monoamine oxidase inhibitor phenelzine, have also been used successfully as second-line treatments, but evidence of their benefit is less convincing than that supporting the first-line SSRIs/SNRIs. Additionally, their less favorable adverse effect and safety profiles make them less attractive treatment choices.13 Table 314-25 provides a list of first- and second-line medications for PTSD with recommended dosages and adverse effect profiles.
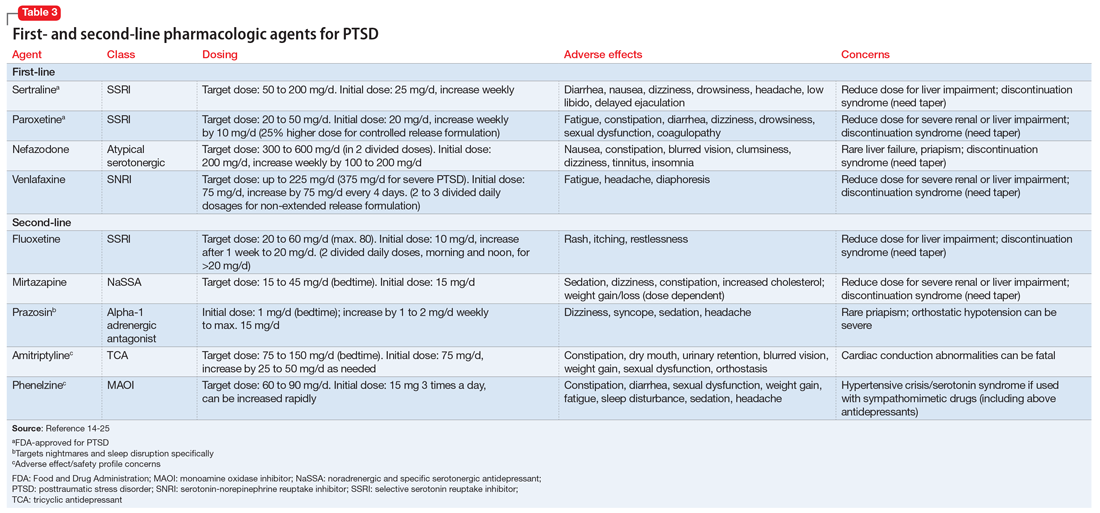
Other medications. Antiepileptics, antipsychotics, and benzodiazepines have not been demonstrated to have efficacy for primary treatment of PTSD, and none of the medications are considered first-line treatments, although sometimes they are used adjunctively in attempts to enhance the effectiveness of antidepressants. Benzodiazepines are sometimes used to target symptoms, such as sleep disturbance or hyperarousal, but only for very short periods. Several authoritative reviews strongly recommend against practices of polypharmacy that commonly involves use of these agents.4,14 Prazosin, an alpha-1 adrenergic antagonist, has been demonstrated to be an effective treatment for nightmares and sleep disturbances, and has grown increasingly popular for treating these symptoms in PTSD, especially in military veterans.13
A well-established barrier to effective pharmacotherapy of PTSD is medication nonadherence.13 Two common underlying sources of nonadherence are inconsistency with the patient’s treatment preference and intolerable adverse effects. Because SSRIs/SNRIs require 8 to 12 weeks of adequate dosing for symptom relief,13 medication adherence is vital. Explaining to patients that it takes many weeks of consistent dosing for clinical effects and reassuring them that the antidepressant agents used to treat PTSD are not habit-forming may help improve adherence.4
Psychotherapy. Prolonged exposure therapy and cognitive processing therapy—both trauma-focused therapies—have the best empirical evidence for efficacy for PTSD.4,14,26 Some patients are too anxious or avoidant to participate in trauma-focused psychotherapy and may benefit from a course of antidepressant treatment before initiating psychotherapy to reduce hyperarousal and avoidance symptoms enough to allow them to tolerate therapy that incorporates trauma memories.6 However, current PTSD treatment guidelines no longer recommend stabilization with medication or preparatory therapy as a routine prerequisite to trauma-focused psychotherapy.4
Continue to: Eye movement desensitization and reprocessing (EMDR) therapy...
Eye movement desensitization and reprocessing (EMDR) therapy has emerged as a popular trauma-focused therapy with documented effectiveness. During EMDR, the patient attends to emotionally disturbing material in brief sequential doses (which varies with individual patients) while simultaneously focusing on an external stimulus, typically therapist-directed lateral eye movements. Critics of EMDR point out that the theoretical concepts and therapeutic maneuvers (eg, finger movements to guide eye gaze) in EMDR are not consistent with current understanding of the neurobiological processes involved in PTSD. Further, studies testing separate components of the therapy have not established independent effectiveness of the therapeutic maneuvers beyond the therapeutic effects of the psychotherapy components of the procedure.4
Other psychotherapies might also be beneficial, but not enough research has been conducted to provide evidence for their effectiveness.4 Non-trauma–focused psychotherapies used for PTSD include supportive therapy, motivational interviewing, relaxation, and mindfulness. Because these therapies have less evidence of effectiveness, they are now widely considered second-line options. Psychological first aid is not a treatment for PTSD, but rather a nontreatment intervention for distress that is widely used by first responders and crisis counselors to provide compassion, support, and stabilization for people exposed to trauma, whether or not they have developed PTSD. Psychological first aid is supported by expert consensus, but it has not been studied enough to demonstrate how helpful it is as a treatment.6
Comorbidities require careful consideration
PTSD in the presence of other psychiatric disorders may require a unique and specialized approach to pharmacotherapy and psychotherapy. For instance, for a patient who has a comorbid substance use disorder, acute substance withdrawal can exacerbate PTSD symptoms. Sertraline is considered a medication of choice for these patients,13 and having a substance abuse specialist on the treatment team is desirable.4,13 A patient with comorbid traumatic brain injury (TBI) may have reduced tolerance to medications, and may require an individually-tailored and elongated titration strategy. Additionally, stimulants sometimes used to improve cognition for patients with comorbid TBI can exacerbate symptoms of hyperarousal, and these patients may need stabilization before beginning PTSD treatment. Antidepressant treatment for PTSD among patients with comorbid bipolar disorder has the potential to induce mania. Psychiatrists must consider these issues when formulating treatment plans for patients with PTSD and specific psychiatric comorbidities.4,6
PTSD symptoms can be chronic, sometimes lasting many years or even decades.27 In a longitudinal study of 716 survivors of 10 different disasters, 62% of those diagnosed with PTSD were still symptomatic 1 to 3 years after the disaster, demonstrating the enduring nature of PTSD symptoms.12 Similarly, a follow-up study of survivors of the Oklahoma City bombing found 58% of those with PTSD and 39% of those without PTSD were still reporting posttraumatic stress symptoms 7 years after the incident.28 Remarkably, these same individuals reported substantially improved functioning at work, with family and personal activities, and social interactions,28 and long-term employment disability specifically related to PTSD is highly unusual.29 Even individuals who continued to report active posttraumatic stress symptoms experienced a return of functioning equivalent to levels in individuals with no PTSD.28 These data suggest that treating psychiatrists and other mental health clinicians can be optimistic that functioning can improve remarkably over the long term, even if posttraumatic stress symptoms persist.
Bottom Line
A thorough understanding of the criteria for posttraumatic stress disorder (PTSD) is necessary for accurate diagnosis and treatment. Evidence-based treatment options for adults with PTSD include certain antidepressants and trauma-focused psychotherapies.
Related Resources
- Bernadino M, Nelson KJ. FIGHT to remember PTSD. Current Psychiatry. 2017;16(8):17.
- Koola MM. Prazosin and doxazosin for PTSD are underutilized and underdosed. Current Psychiatry. 2017;16(3):19-20,47,e1.
Drug Brand Names
Amitriptyline • Elavil, Endep
Fluoxetine • Prozac, Sarafem
Mirtazapine • Remeron
Nefazodone • Serzone
Paroxetine • Paxil
Phenelzine • Nardil
Prazosin • Minipress
Sertraline • Zoloft
Venlafaxine • Effexor
1. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Washington, DC: American Psychiatric Association; 1980.
2. North CS, Surís AM, Smith RP, et al. The evolution of PTSD criteria across editions of the DSM. Ann Clin Psychiatry. 2016;28(3):197-208.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013
4. Downs DL, North CS. Trauma-related disorders. Overview of posttraumatic stress disorder. https://www.deckerip.com/products/scientific-american-psychiatry/table-of-contents/. Published July 2017. Accessed February 27, 2018.
5. North CS. Disaster mental health epidemiology: methodological review and interpretation of research findings. Psychiatry. 2016; 79(2):130-146.
6. North CS, Yutzy SH. Goodwin and Guze’s Psychiatric Diagnosis, 6th ed. New York, NY: Oxford University Press; 2010.
7. North CS, Nixon SJ, Shariat S, et al. Psychiatric disorders among survivors of the Oklahoma City bombing. JAMA. 1999;282(8):755-762.
8. North CS, Pfefferbaum B. Mental Health Response to Community Disasters: A Systematic Review. JAMA. 2013;310(5):507-518.
9. North CS, Pollio DE, Smith, RP, et al. Trauma exposure and posttraumatic stress disorder among employees of New York City companies affected by the September 11, 2001 attacks on the World Trade Center. Disaster Med Public Health Prep. 2011;5(suppl 2):S205-S213.
10. North CS, Oliver J, Pandya A. Examining a comprehensive model of disaster-related posttraumatic stress disorder in systematically studied survivors of 10 disasters. Am J Public Health. 2012;102(10):e40-e48.
11. Whitman JB, North CS, Downs DL, et al. A prospective study of the onset of PTSD symptoms in the first month after trauma exposure. Ann Clin Psychiatry. 2013;25(3):163-172.
12. North CS, Oliver J. Analysis of the longitudinal course of PTSD in 716 survivors of 10 disasters. Soc Psychiatry Psychiatr Epidemiol. 2013;48(8):1189-1197.
13. Jeffreys M, Capehart B, Friedman MJ. Pharmacotherapy for posttraumatic stress disorder: review with clinical applications. J Rehabil Res Dev. 2012;49(5):703-715.
14. Lee DJ, Schnitzlein CW, Wolf JP, et al. Psychotherapy versus pharmacotherapy for posttraumatic stress disorder: systemic review and meta-analyses to determine first-line treatments. Depress Anxiety. 2016;33(9):792-806.
15. Foa EB, Keane T, Friedman MJ. Effective treatments for PTSD: practice guidelines from the International Society for traumatic stress studies. New York, NY: The Guilford Press; 2000.
16. Ursano RJ, Bell C, Eth S, et al; Work Group on ASD and PTSD. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Arlington, VA: American Psychiatric Association Publishing; 2004.
17. National Collaborating Centre for Mental Health. Post-traumatic stress disorder: the management of PTSD in adults and children in primary and secondary care. London, UK: Gaskell and the British Psychological Society; 2005.
18. Foa EB, Keane TM, Friedman MJ, eds; The Board of Directors of the International Society for Traumatic Stress Studies. Effective treatments for PTSD. 2nd ed. Oakbrook Terrace, IL: The Guilford Press; 2005.
19. Department of Veterans Affairs and Department of Defense. VA/DoD clinical practice guidelines. Management of Posttraumatic Stress Disorder and Acute Stress Reaction 2017. https://www.healthquality.va.gov/guidelines/MH/ptsd/. Published June 2017. Accessed February 26, 2018.
20. Phoenix Australia -Centre for Posttraumatic Mental Health. Australian guidelines for the treatment of acute stress disorder and posttraumatic stress disorder. Melbourne, Australia: Phoenix Australia Centre for Posttraumatic Mental Health; 2013.
21. World Health Organization. Guidelines for the management of conditions specifically related to stress. Geneva, Switzerland: World Health Organization Press; 2013.
22. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):201-213.
23. Stein DJ, Ipser J, McAnda N. Pharmacotherapy of posttraumatic stress disorder: a review of meta-analyses and treatment guidelines. CNS Spectr. 2009;14(suppl 1):25-31.
24. Forbes D, Creamer M, Bisson JI, et al. A guide to guidelines for the treatment of PTSD and related conditions. J Trauma Stress. 2010;23(5):537-552.
25. Nash WP, Watson PJ. Review of VA/DOD clinical practice guideline on management of acute stress and interventions to prevent posttraumatic stress disorder. J Rehabil Res Dev. 2012;49(5):637-648.
26. Birur B, Moore NC, Davis LL. An evidence-based review of early intervention and prevention of posttraumatic stress disorder. Community Ment Health J. 2017;53(2):183-201.
27. Breslau N, Davis GC. Posttraumatic stress disorder in an urban population of young adults: Risk factors for chronicity. Am J Psychiatry. 1992;149(5):671-675.
28. North CS, Pfefferbaum B, Kawasaki A, et al. Psychosocial adjustment of directly exposed survivors seven years after the Oklahoma City bombing. Compr Psychiatry. 2011;52(1):1-8
29. Rasco SS, North CS. An empirical study of employment and disability over three years among survivors of major disasters. J Am Acad Psychiatry Law. 2010;38(1):80-86.
Posttraumatic stress disorder (PTSD) has increasingly become a part of American culture since its introduction in the American Psychiatric Association’s third edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III) in 1980.1 Since then, a proliferation of material about this disorder—both academic and popular—has been generated, yet much confusion persists surrounding the definition of the disorder, its prevalence, and its management. This review addresses the essential elements for diagnosis and treatment of PTSD.
Diagnosis: A closer look at the criteria
Criteria for the diagnosis of PTSD have evolved since 1980, with changes in the definition of trauma and the addition of symptoms and symptom groups.2 Table 13 summarizes the current DSM-5 criteria for PTSD.

Trauma exposure. An essential first step in the diagnosis of PTSD is to determine whether the individual has experienced exposure to trauma. This concept is defined in Criterion A (trauma exposure).3 PTSD is nonconformist among the psychiatric diagnoses in that it requires a specific external event as part of its definition. Misapplication of the trauma exposure criterion by many clinicians and researchers has led to misdiagnosis and erroneously high prevalence estimates of PTSD.4,5
A traumatic event is one that represents a threat to life or limb, specifically defined as “actual or threatened death, serious injury, or sexual violence.”3 DSM-5 does not allow for just any stressful event to be considered trauma. For example, no matter how distressing, failing an important test at school or being served with divorce proceedings do not represent a requisite trauma6 because these examples do not entail a threat to life or limb.
DSM-5 PTSD Criterion A also requires a qualifying exposure to the traumatic event. There are 4 types of qualifying exposures:
- direct experience of immediate serious physical danger
- eyewitness of trauma to others
- indirect exposure via violent or accidental trauma experienced by a close family member or close friend
- repeated or extreme exposure to aversive details of trauma, such as first responders collecting human remains or law enforcement officers being repeatedly exposed to horrific details of child abuse.3
Witnessed trauma must be in person; thus, viewing trauma in media reports would not constitute a qualifying exposure. Indirect trauma exposure can occur through learning of the experience of a qualifying trauma exposure by a close family member or personal friend.
It is critical to differentiate exposure to trauma (an objective construct) from the subjective distress that may be associated with it. If trauma has not occurred or a qualifying exposure is not established, no amount of distress associated with it can establish the experience as meeting Criterion A for PTSD. This does not mean that nonqualifying experiences of stressful events are not distressing; in fact, such experiences can result in substantial psychological angst. Conversely, exposure to trauma is not tantamount to a diagnosis of PTSD, as most trauma exposures do not result in PTSD.7,8
Continue to: Symptom groups
Symptom groups. DSM-5 symptom criteria for PTSD include 4 symptom groups, Criteria B to E, respectively:
- intrusion
- avoidance
- negative cognitions and mood (numbing)
- hyperarousal/reactivity.
A specific number of symptoms must be present in all 4 of the symptom groups to fulfill diagnostic criteria. Importantly, these symptoms must be linked temporally and conceptually to the traumatic exposure to qualify as PTSD symptoms. Specifically, the symptoms must be new or substantially worsened after the event. For example, continuing sleep disturbance in someone who has had lifetime difficulty sleeping would not count as a trauma-related symptom. Most symptom checklists do not properly assess diagnostic criteria for PTSD because they do not anchor the symptoms in an exposure to a traumatic event; diagnosis requires an interview to fully assess all the diagnostic criteria. Finally, the symptoms must have been present for >1 month for the diagnosis, and the symptoms must have resulted in clinically significant distress or functional impairment to qualify.
The Algorithm provides a practical way to systematically assess all DSM-5 criteria for PTSD to arrive at a diagnosis. The clinician begins by determining whether a traumatic event has occurred and whether the individual had a qualifying exposure to it. If not, PTSD cannot be diagnosed. Alternative diagnoses to consider for new disorders that arise in the context of trauma among patients who are not exposed to trauma include major depressive disorder, adjustment disorder, and bereavement, as well as acute stress disorder (which is not validated but has potential utility as a billable diagnosis).

Avoidance and numbing symptoms (present in Criteria C and D) have been shown to represent markers of illness and can be useful in predicting PTSD.8-10 Unlike symptoms of intrusion and hyperarousal (Criteria B and E, respectively), which are very common and by themselves are nonpathological, avoidance/numbing symptoms occur much less commonly, are associated with functional impairment and other indicators of illness, and are strongly associated with PTSD.6 Prominent avoidance/numbing profiles have been demonstrated to predict PTSD in the first 1 to 2 weeks after trauma exposure, before PTSD can be formally diagnosed.11 Posttraumatic stress symptoms are nearly universal after trauma exposure, even in people who do not develop PTSD.5 Intrusion and hyperarousal symptoms constitute most of such symptoms,7 and these symptoms in the absence of prominent avoidance/numbing can be considered normative distress responses to trauma exposure.12
Some PTSD symptoms may seem quite similar to symptoms of depressive disorders and anxiety disorders. PTSD can be differentiated from these other disorders by linking the symptoms temporally and contextually to a qualifying exposure to a traumatic event. More often than not, PTSD presents with comorbid psychiatric disorders, especially depressive disorders, anxiety disorders, and/or substance use disorders.
Continue to: Treatment: Medication, psychotherapy, or both
Treatment: Medication, psychotherapy, or both
Both pharmacotherapy and psychotherapy—as monotherapy or in combination—are beneficial for treatment of PTSD. Research has not conclusively shown either treatment modality to be superior, because adequate head-to-head trials have not been conducted.4 Therefore, the choice of initial treatment is based on individual circumstances, such as patient preference for medication and/or psychotherapy, or the availability of therapists trained in evidence-based PTSD psychotherapy. Pharmacotherapeutic approaches are considered especially beneficial for depressive- and anxiety-like symptoms of PTSD, and trauma-focused psychotherapies are presumed to address the neuropathology of conditioned fear and anxiety responses involved in PTSD.14 Table 214-25 provides a list of published treatment guidelines and reviews to help clinicians seeking further detail beyond that provided in this article.

Antidepressants are the mainstay of pharmacotherapy for PTSD. These medications are effective for treating major depressive disorder, and have beneficial properties for PTSD independent of their antidepressant effects. The serotonin selective reuptake inhibitors (SSRIs) sertraline and paroxetine are FDA-approved for the treatment of PTSD.6 Other recommended medications include the serotonin-norepinephrine reuptake inhibitor (SNRI) venlafaxine, and nefazodone, an atypical serotoninergic agent.13 Other antidepressants with less published evidence of effectiveness are used as second-line pharmacotherapies for PTSD, including fluoxetine (SSRI), and mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA).4 Older medications, such as the tricyclic antidepressant amitriptyline and the monoamine oxidase inhibitor phenelzine, have also been used successfully as second-line treatments, but evidence of their benefit is less convincing than that supporting the first-line SSRIs/SNRIs. Additionally, their less favorable adverse effect and safety profiles make them less attractive treatment choices.13 Table 314-25 provides a list of first- and second-line medications for PTSD with recommended dosages and adverse effect profiles.

Other medications. Antiepileptics, antipsychotics, and benzodiazepines have not been demonstrated to have efficacy for primary treatment of PTSD, and none of the medications are considered first-line treatments, although sometimes they are used adjunctively in attempts to enhance the effectiveness of antidepressants. Benzodiazepines are sometimes used to target symptoms, such as sleep disturbance or hyperarousal, but only for very short periods. Several authoritative reviews strongly recommend against practices of polypharmacy that commonly involves use of these agents.4,14 Prazosin, an alpha-1 adrenergic antagonist, has been demonstrated to be an effective treatment for nightmares and sleep disturbances, and has grown increasingly popular for treating these symptoms in PTSD, especially in military veterans.13
A well-established barrier to effective pharmacotherapy of PTSD is medication nonadherence.13 Two common underlying sources of nonadherence are inconsistency with the patient’s treatment preference and intolerable adverse effects. Because SSRIs/SNRIs require 8 to 12 weeks of adequate dosing for symptom relief,13 medication adherence is vital. Explaining to patients that it takes many weeks of consistent dosing for clinical effects and reassuring them that the antidepressant agents used to treat PTSD are not habit-forming may help improve adherence.4
Psychotherapy. Prolonged exposure therapy and cognitive processing therapy—both trauma-focused therapies—have the best empirical evidence for efficacy for PTSD.4,14,26 Some patients are too anxious or avoidant to participate in trauma-focused psychotherapy and may benefit from a course of antidepressant treatment before initiating psychotherapy to reduce hyperarousal and avoidance symptoms enough to allow them to tolerate therapy that incorporates trauma memories.6 However, current PTSD treatment guidelines no longer recommend stabilization with medication or preparatory therapy as a routine prerequisite to trauma-focused psychotherapy.4
Continue to: Eye movement desensitization and reprocessing (EMDR) therapy...
Eye movement desensitization and reprocessing (EMDR) therapy has emerged as a popular trauma-focused therapy with documented effectiveness. During EMDR, the patient attends to emotionally disturbing material in brief sequential doses (which varies with individual patients) while simultaneously focusing on an external stimulus, typically therapist-directed lateral eye movements. Critics of EMDR point out that the theoretical concepts and therapeutic maneuvers (eg, finger movements to guide eye gaze) in EMDR are not consistent with current understanding of the neurobiological processes involved in PTSD. Further, studies testing separate components of the therapy have not established independent effectiveness of the therapeutic maneuvers beyond the therapeutic effects of the psychotherapy components of the procedure.4
Other psychotherapies might also be beneficial, but not enough research has been conducted to provide evidence for their effectiveness.4 Non-trauma–focused psychotherapies used for PTSD include supportive therapy, motivational interviewing, relaxation, and mindfulness. Because these therapies have less evidence of effectiveness, they are now widely considered second-line options. Psychological first aid is not a treatment for PTSD, but rather a nontreatment intervention for distress that is widely used by first responders and crisis counselors to provide compassion, support, and stabilization for people exposed to trauma, whether or not they have developed PTSD. Psychological first aid is supported by expert consensus, but it has not been studied enough to demonstrate how helpful it is as a treatment.6
Comorbidities require careful consideration
PTSD in the presence of other psychiatric disorders may require a unique and specialized approach to pharmacotherapy and psychotherapy. For instance, for a patient who has a comorbid substance use disorder, acute substance withdrawal can exacerbate PTSD symptoms. Sertraline is considered a medication of choice for these patients,13 and having a substance abuse specialist on the treatment team is desirable.4,13 A patient with comorbid traumatic brain injury (TBI) may have reduced tolerance to medications, and may require an individually-tailored and elongated titration strategy. Additionally, stimulants sometimes used to improve cognition for patients with comorbid TBI can exacerbate symptoms of hyperarousal, and these patients may need stabilization before beginning PTSD treatment. Antidepressant treatment for PTSD among patients with comorbid bipolar disorder has the potential to induce mania. Psychiatrists must consider these issues when formulating treatment plans for patients with PTSD and specific psychiatric comorbidities.4,6
PTSD symptoms can be chronic, sometimes lasting many years or even decades.27 In a longitudinal study of 716 survivors of 10 different disasters, 62% of those diagnosed with PTSD were still symptomatic 1 to 3 years after the disaster, demonstrating the enduring nature of PTSD symptoms.12 Similarly, a follow-up study of survivors of the Oklahoma City bombing found 58% of those with PTSD and 39% of those without PTSD were still reporting posttraumatic stress symptoms 7 years after the incident.28 Remarkably, these same individuals reported substantially improved functioning at work, with family and personal activities, and social interactions,28 and long-term employment disability specifically related to PTSD is highly unusual.29 Even individuals who continued to report active posttraumatic stress symptoms experienced a return of functioning equivalent to levels in individuals with no PTSD.28 These data suggest that treating psychiatrists and other mental health clinicians can be optimistic that functioning can improve remarkably over the long term, even if posttraumatic stress symptoms persist.
Bottom Line
A thorough understanding of the criteria for posttraumatic stress disorder (PTSD) is necessary for accurate diagnosis and treatment. Evidence-based treatment options for adults with PTSD include certain antidepressants and trauma-focused psychotherapies.
Related Resources
- Bernadino M, Nelson KJ. FIGHT to remember PTSD. Current Psychiatry. 2017;16(8):17.
- Koola MM. Prazosin and doxazosin for PTSD are underutilized and underdosed. Current Psychiatry. 2017;16(3):19-20,47,e1.
Drug Brand Names
Amitriptyline • Elavil, Endep
Fluoxetine • Prozac, Sarafem
Mirtazapine • Remeron
Nefazodone • Serzone
Paroxetine • Paxil
Phenelzine • Nardil
Prazosin • Minipress
Sertraline • Zoloft
Venlafaxine • Effexor
Posttraumatic stress disorder (PTSD) has increasingly become a part of American culture since its introduction in the American Psychiatric Association’s third edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III) in 1980.1 Since then, a proliferation of material about this disorder—both academic and popular—has been generated, yet much confusion persists surrounding the definition of the disorder, its prevalence, and its management. This review addresses the essential elements for diagnosis and treatment of PTSD.
Diagnosis: A closer look at the criteria
Criteria for the diagnosis of PTSD have evolved since 1980, with changes in the definition of trauma and the addition of symptoms and symptom groups.2 Table 13 summarizes the current DSM-5 criteria for PTSD.

Trauma exposure. An essential first step in the diagnosis of PTSD is to determine whether the individual has experienced exposure to trauma. This concept is defined in Criterion A (trauma exposure).3 PTSD is nonconformist among the psychiatric diagnoses in that it requires a specific external event as part of its definition. Misapplication of the trauma exposure criterion by many clinicians and researchers has led to misdiagnosis and erroneously high prevalence estimates of PTSD.4,5
A traumatic event is one that represents a threat to life or limb, specifically defined as “actual or threatened death, serious injury, or sexual violence.”3 DSM-5 does not allow for just any stressful event to be considered trauma. For example, no matter how distressing, failing an important test at school or being served with divorce proceedings do not represent a requisite trauma6 because these examples do not entail a threat to life or limb.
DSM-5 PTSD Criterion A also requires a qualifying exposure to the traumatic event. There are 4 types of qualifying exposures:
- direct experience of immediate serious physical danger
- eyewitness of trauma to others
- indirect exposure via violent or accidental trauma experienced by a close family member or close friend
- repeated or extreme exposure to aversive details of trauma, such as first responders collecting human remains or law enforcement officers being repeatedly exposed to horrific details of child abuse.3
Witnessed trauma must be in person; thus, viewing trauma in media reports would not constitute a qualifying exposure. Indirect trauma exposure can occur through learning of the experience of a qualifying trauma exposure by a close family member or personal friend.
It is critical to differentiate exposure to trauma (an objective construct) from the subjective distress that may be associated with it. If trauma has not occurred or a qualifying exposure is not established, no amount of distress associated with it can establish the experience as meeting Criterion A for PTSD. This does not mean that nonqualifying experiences of stressful events are not distressing; in fact, such experiences can result in substantial psychological angst. Conversely, exposure to trauma is not tantamount to a diagnosis of PTSD, as most trauma exposures do not result in PTSD.7,8
Continue to: Symptom groups
Symptom groups. DSM-5 symptom criteria for PTSD include 4 symptom groups, Criteria B to E, respectively:
- intrusion
- avoidance
- negative cognitions and mood (numbing)
- hyperarousal/reactivity.
A specific number of symptoms must be present in all 4 of the symptom groups to fulfill diagnostic criteria. Importantly, these symptoms must be linked temporally and conceptually to the traumatic exposure to qualify as PTSD symptoms. Specifically, the symptoms must be new or substantially worsened after the event. For example, continuing sleep disturbance in someone who has had lifetime difficulty sleeping would not count as a trauma-related symptom. Most symptom checklists do not properly assess diagnostic criteria for PTSD because they do not anchor the symptoms in an exposure to a traumatic event; diagnosis requires an interview to fully assess all the diagnostic criteria. Finally, the symptoms must have been present for >1 month for the diagnosis, and the symptoms must have resulted in clinically significant distress or functional impairment to qualify.
The Algorithm provides a practical way to systematically assess all DSM-5 criteria for PTSD to arrive at a diagnosis. The clinician begins by determining whether a traumatic event has occurred and whether the individual had a qualifying exposure to it. If not, PTSD cannot be diagnosed. Alternative diagnoses to consider for new disorders that arise in the context of trauma among patients who are not exposed to trauma include major depressive disorder, adjustment disorder, and bereavement, as well as acute stress disorder (which is not validated but has potential utility as a billable diagnosis).

Avoidance and numbing symptoms (present in Criteria C and D) have been shown to represent markers of illness and can be useful in predicting PTSD.8-10 Unlike symptoms of intrusion and hyperarousal (Criteria B and E, respectively), which are very common and by themselves are nonpathological, avoidance/numbing symptoms occur much less commonly, are associated with functional impairment and other indicators of illness, and are strongly associated with PTSD.6 Prominent avoidance/numbing profiles have been demonstrated to predict PTSD in the first 1 to 2 weeks after trauma exposure, before PTSD can be formally diagnosed.11 Posttraumatic stress symptoms are nearly universal after trauma exposure, even in people who do not develop PTSD.5 Intrusion and hyperarousal symptoms constitute most of such symptoms,7 and these symptoms in the absence of prominent avoidance/numbing can be considered normative distress responses to trauma exposure.12
Some PTSD symptoms may seem quite similar to symptoms of depressive disorders and anxiety disorders. PTSD can be differentiated from these other disorders by linking the symptoms temporally and contextually to a qualifying exposure to a traumatic event. More often than not, PTSD presents with comorbid psychiatric disorders, especially depressive disorders, anxiety disorders, and/or substance use disorders.
Continue to: Treatment: Medication, psychotherapy, or both
Treatment: Medication, psychotherapy, or both
Both pharmacotherapy and psychotherapy—as monotherapy or in combination—are beneficial for treatment of PTSD. Research has not conclusively shown either treatment modality to be superior, because adequate head-to-head trials have not been conducted.4 Therefore, the choice of initial treatment is based on individual circumstances, such as patient preference for medication and/or psychotherapy, or the availability of therapists trained in evidence-based PTSD psychotherapy. Pharmacotherapeutic approaches are considered especially beneficial for depressive- and anxiety-like symptoms of PTSD, and trauma-focused psychotherapies are presumed to address the neuropathology of conditioned fear and anxiety responses involved in PTSD.14 Table 214-25 provides a list of published treatment guidelines and reviews to help clinicians seeking further detail beyond that provided in this article.

Antidepressants are the mainstay of pharmacotherapy for PTSD. These medications are effective for treating major depressive disorder, and have beneficial properties for PTSD independent of their antidepressant effects. The serotonin selective reuptake inhibitors (SSRIs) sertraline and paroxetine are FDA-approved for the treatment of PTSD.6 Other recommended medications include the serotonin-norepinephrine reuptake inhibitor (SNRI) venlafaxine, and nefazodone, an atypical serotoninergic agent.13 Other antidepressants with less published evidence of effectiveness are used as second-line pharmacotherapies for PTSD, including fluoxetine (SSRI), and mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA).4 Older medications, such as the tricyclic antidepressant amitriptyline and the monoamine oxidase inhibitor phenelzine, have also been used successfully as second-line treatments, but evidence of their benefit is less convincing than that supporting the first-line SSRIs/SNRIs. Additionally, their less favorable adverse effect and safety profiles make them less attractive treatment choices.13 Table 314-25 provides a list of first- and second-line medications for PTSD with recommended dosages and adverse effect profiles.

Other medications. Antiepileptics, antipsychotics, and benzodiazepines have not been demonstrated to have efficacy for primary treatment of PTSD, and none of the medications are considered first-line treatments, although sometimes they are used adjunctively in attempts to enhance the effectiveness of antidepressants. Benzodiazepines are sometimes used to target symptoms, such as sleep disturbance or hyperarousal, but only for very short periods. Several authoritative reviews strongly recommend against practices of polypharmacy that commonly involves use of these agents.4,14 Prazosin, an alpha-1 adrenergic antagonist, has been demonstrated to be an effective treatment for nightmares and sleep disturbances, and has grown increasingly popular for treating these symptoms in PTSD, especially in military veterans.13
A well-established barrier to effective pharmacotherapy of PTSD is medication nonadherence.13 Two common underlying sources of nonadherence are inconsistency with the patient’s treatment preference and intolerable adverse effects. Because SSRIs/SNRIs require 8 to 12 weeks of adequate dosing for symptom relief,13 medication adherence is vital. Explaining to patients that it takes many weeks of consistent dosing for clinical effects and reassuring them that the antidepressant agents used to treat PTSD are not habit-forming may help improve adherence.4
Psychotherapy. Prolonged exposure therapy and cognitive processing therapy—both trauma-focused therapies—have the best empirical evidence for efficacy for PTSD.4,14,26 Some patients are too anxious or avoidant to participate in trauma-focused psychotherapy and may benefit from a course of antidepressant treatment before initiating psychotherapy to reduce hyperarousal and avoidance symptoms enough to allow them to tolerate therapy that incorporates trauma memories.6 However, current PTSD treatment guidelines no longer recommend stabilization with medication or preparatory therapy as a routine prerequisite to trauma-focused psychotherapy.4
Continue to: Eye movement desensitization and reprocessing (EMDR) therapy...
Eye movement desensitization and reprocessing (EMDR) therapy has emerged as a popular trauma-focused therapy with documented effectiveness. During EMDR, the patient attends to emotionally disturbing material in brief sequential doses (which varies with individual patients) while simultaneously focusing on an external stimulus, typically therapist-directed lateral eye movements. Critics of EMDR point out that the theoretical concepts and therapeutic maneuvers (eg, finger movements to guide eye gaze) in EMDR are not consistent with current understanding of the neurobiological processes involved in PTSD. Further, studies testing separate components of the therapy have not established independent effectiveness of the therapeutic maneuvers beyond the therapeutic effects of the psychotherapy components of the procedure.4
Other psychotherapies might also be beneficial, but not enough research has been conducted to provide evidence for their effectiveness.4 Non-trauma–focused psychotherapies used for PTSD include supportive therapy, motivational interviewing, relaxation, and mindfulness. Because these therapies have less evidence of effectiveness, they are now widely considered second-line options. Psychological first aid is not a treatment for PTSD, but rather a nontreatment intervention for distress that is widely used by first responders and crisis counselors to provide compassion, support, and stabilization for people exposed to trauma, whether or not they have developed PTSD. Psychological first aid is supported by expert consensus, but it has not been studied enough to demonstrate how helpful it is as a treatment.6
Comorbidities require careful consideration
PTSD in the presence of other psychiatric disorders may require a unique and specialized approach to pharmacotherapy and psychotherapy. For instance, for a patient who has a comorbid substance use disorder, acute substance withdrawal can exacerbate PTSD symptoms. Sertraline is considered a medication of choice for these patients,13 and having a substance abuse specialist on the treatment team is desirable.4,13 A patient with comorbid traumatic brain injury (TBI) may have reduced tolerance to medications, and may require an individually-tailored and elongated titration strategy. Additionally, stimulants sometimes used to improve cognition for patients with comorbid TBI can exacerbate symptoms of hyperarousal, and these patients may need stabilization before beginning PTSD treatment. Antidepressant treatment for PTSD among patients with comorbid bipolar disorder has the potential to induce mania. Psychiatrists must consider these issues when formulating treatment plans for patients with PTSD and specific psychiatric comorbidities.4,6
PTSD symptoms can be chronic, sometimes lasting many years or even decades.27 In a longitudinal study of 716 survivors of 10 different disasters, 62% of those diagnosed with PTSD were still symptomatic 1 to 3 years after the disaster, demonstrating the enduring nature of PTSD symptoms.12 Similarly, a follow-up study of survivors of the Oklahoma City bombing found 58% of those with PTSD and 39% of those without PTSD were still reporting posttraumatic stress symptoms 7 years after the incident.28 Remarkably, these same individuals reported substantially improved functioning at work, with family and personal activities, and social interactions,28 and long-term employment disability specifically related to PTSD is highly unusual.29 Even individuals who continued to report active posttraumatic stress symptoms experienced a return of functioning equivalent to levels in individuals with no PTSD.28 These data suggest that treating psychiatrists and other mental health clinicians can be optimistic that functioning can improve remarkably over the long term, even if posttraumatic stress symptoms persist.
Bottom Line
A thorough understanding of the criteria for posttraumatic stress disorder (PTSD) is necessary for accurate diagnosis and treatment. Evidence-based treatment options for adults with PTSD include certain antidepressants and trauma-focused psychotherapies.
Related Resources
- Bernadino M, Nelson KJ. FIGHT to remember PTSD. Current Psychiatry. 2017;16(8):17.
- Koola MM. Prazosin and doxazosin for PTSD are underutilized and underdosed. Current Psychiatry. 2017;16(3):19-20,47,e1.
Drug Brand Names
Amitriptyline • Elavil, Endep
Fluoxetine • Prozac, Sarafem
Mirtazapine • Remeron
Nefazodone • Serzone
Paroxetine • Paxil
Phenelzine • Nardil
Prazosin • Minipress
Sertraline • Zoloft
Venlafaxine • Effexor
1. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Washington, DC: American Psychiatric Association; 1980.
2. North CS, Surís AM, Smith RP, et al. The evolution of PTSD criteria across editions of the DSM. Ann Clin Psychiatry. 2016;28(3):197-208.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013
4. Downs DL, North CS. Trauma-related disorders. Overview of posttraumatic stress disorder. https://www.deckerip.com/products/scientific-american-psychiatry/table-of-contents/. Published July 2017. Accessed February 27, 2018.
5. North CS. Disaster mental health epidemiology: methodological review and interpretation of research findings. Psychiatry. 2016; 79(2):130-146.
6. North CS, Yutzy SH. Goodwin and Guze’s Psychiatric Diagnosis, 6th ed. New York, NY: Oxford University Press; 2010.
7. North CS, Nixon SJ, Shariat S, et al. Psychiatric disorders among survivors of the Oklahoma City bombing. JAMA. 1999;282(8):755-762.
8. North CS, Pfefferbaum B. Mental Health Response to Community Disasters: A Systematic Review. JAMA. 2013;310(5):507-518.
9. North CS, Pollio DE, Smith, RP, et al. Trauma exposure and posttraumatic stress disorder among employees of New York City companies affected by the September 11, 2001 attacks on the World Trade Center. Disaster Med Public Health Prep. 2011;5(suppl 2):S205-S213.
10. North CS, Oliver J, Pandya A. Examining a comprehensive model of disaster-related posttraumatic stress disorder in systematically studied survivors of 10 disasters. Am J Public Health. 2012;102(10):e40-e48.
11. Whitman JB, North CS, Downs DL, et al. A prospective study of the onset of PTSD symptoms in the first month after trauma exposure. Ann Clin Psychiatry. 2013;25(3):163-172.
12. North CS, Oliver J. Analysis of the longitudinal course of PTSD in 716 survivors of 10 disasters. Soc Psychiatry Psychiatr Epidemiol. 2013;48(8):1189-1197.
13. Jeffreys M, Capehart B, Friedman MJ. Pharmacotherapy for posttraumatic stress disorder: review with clinical applications. J Rehabil Res Dev. 2012;49(5):703-715.
14. Lee DJ, Schnitzlein CW, Wolf JP, et al. Psychotherapy versus pharmacotherapy for posttraumatic stress disorder: systemic review and meta-analyses to determine first-line treatments. Depress Anxiety. 2016;33(9):792-806.
15. Foa EB, Keane T, Friedman MJ. Effective treatments for PTSD: practice guidelines from the International Society for traumatic stress studies. New York, NY: The Guilford Press; 2000.
16. Ursano RJ, Bell C, Eth S, et al; Work Group on ASD and PTSD. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Arlington, VA: American Psychiatric Association Publishing; 2004.
17. National Collaborating Centre for Mental Health. Post-traumatic stress disorder: the management of PTSD in adults and children in primary and secondary care. London, UK: Gaskell and the British Psychological Society; 2005.
18. Foa EB, Keane TM, Friedman MJ, eds; The Board of Directors of the International Society for Traumatic Stress Studies. Effective treatments for PTSD. 2nd ed. Oakbrook Terrace, IL: The Guilford Press; 2005.
19. Department of Veterans Affairs and Department of Defense. VA/DoD clinical practice guidelines. Management of Posttraumatic Stress Disorder and Acute Stress Reaction 2017. https://www.healthquality.va.gov/guidelines/MH/ptsd/. Published June 2017. Accessed February 26, 2018.
20. Phoenix Australia -Centre for Posttraumatic Mental Health. Australian guidelines for the treatment of acute stress disorder and posttraumatic stress disorder. Melbourne, Australia: Phoenix Australia Centre for Posttraumatic Mental Health; 2013.
21. World Health Organization. Guidelines for the management of conditions specifically related to stress. Geneva, Switzerland: World Health Organization Press; 2013.
22. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):201-213.
23. Stein DJ, Ipser J, McAnda N. Pharmacotherapy of posttraumatic stress disorder: a review of meta-analyses and treatment guidelines. CNS Spectr. 2009;14(suppl 1):25-31.
24. Forbes D, Creamer M, Bisson JI, et al. A guide to guidelines for the treatment of PTSD and related conditions. J Trauma Stress. 2010;23(5):537-552.
25. Nash WP, Watson PJ. Review of VA/DOD clinical practice guideline on management of acute stress and interventions to prevent posttraumatic stress disorder. J Rehabil Res Dev. 2012;49(5):637-648.
26. Birur B, Moore NC, Davis LL. An evidence-based review of early intervention and prevention of posttraumatic stress disorder. Community Ment Health J. 2017;53(2):183-201.
27. Breslau N, Davis GC. Posttraumatic stress disorder in an urban population of young adults: Risk factors for chronicity. Am J Psychiatry. 1992;149(5):671-675.
28. North CS, Pfefferbaum B, Kawasaki A, et al. Psychosocial adjustment of directly exposed survivors seven years after the Oklahoma City bombing. Compr Psychiatry. 2011;52(1):1-8
29. Rasco SS, North CS. An empirical study of employment and disability over three years among survivors of major disasters. J Am Acad Psychiatry Law. 2010;38(1):80-86.
1. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Washington, DC: American Psychiatric Association; 1980.
2. North CS, Surís AM, Smith RP, et al. The evolution of PTSD criteria across editions of the DSM. Ann Clin Psychiatry. 2016;28(3):197-208.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013
4. Downs DL, North CS. Trauma-related disorders. Overview of posttraumatic stress disorder. https://www.deckerip.com/products/scientific-american-psychiatry/table-of-contents/. Published July 2017. Accessed February 27, 2018.
5. North CS. Disaster mental health epidemiology: methodological review and interpretation of research findings. Psychiatry. 2016; 79(2):130-146.
6. North CS, Yutzy SH. Goodwin and Guze’s Psychiatric Diagnosis, 6th ed. New York, NY: Oxford University Press; 2010.
7. North CS, Nixon SJ, Shariat S, et al. Psychiatric disorders among survivors of the Oklahoma City bombing. JAMA. 1999;282(8):755-762.
8. North CS, Pfefferbaum B. Mental Health Response to Community Disasters: A Systematic Review. JAMA. 2013;310(5):507-518.
9. North CS, Pollio DE, Smith, RP, et al. Trauma exposure and posttraumatic stress disorder among employees of New York City companies affected by the September 11, 2001 attacks on the World Trade Center. Disaster Med Public Health Prep. 2011;5(suppl 2):S205-S213.
10. North CS, Oliver J, Pandya A. Examining a comprehensive model of disaster-related posttraumatic stress disorder in systematically studied survivors of 10 disasters. Am J Public Health. 2012;102(10):e40-e48.
11. Whitman JB, North CS, Downs DL, et al. A prospective study of the onset of PTSD symptoms in the first month after trauma exposure. Ann Clin Psychiatry. 2013;25(3):163-172.
12. North CS, Oliver J. Analysis of the longitudinal course of PTSD in 716 survivors of 10 disasters. Soc Psychiatry Psychiatr Epidemiol. 2013;48(8):1189-1197.
13. Jeffreys M, Capehart B, Friedman MJ. Pharmacotherapy for posttraumatic stress disorder: review with clinical applications. J Rehabil Res Dev. 2012;49(5):703-715.
14. Lee DJ, Schnitzlein CW, Wolf JP, et al. Psychotherapy versus pharmacotherapy for posttraumatic stress disorder: systemic review and meta-analyses to determine first-line treatments. Depress Anxiety. 2016;33(9):792-806.
15. Foa EB, Keane T, Friedman MJ. Effective treatments for PTSD: practice guidelines from the International Society for traumatic stress studies. New York, NY: The Guilford Press; 2000.
16. Ursano RJ, Bell C, Eth S, et al; Work Group on ASD and PTSD. Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Arlington, VA: American Psychiatric Association Publishing; 2004.
17. National Collaborating Centre for Mental Health. Post-traumatic stress disorder: the management of PTSD in adults and children in primary and secondary care. London, UK: Gaskell and the British Psychological Society; 2005.
18. Foa EB, Keane TM, Friedman MJ, eds; The Board of Directors of the International Society for Traumatic Stress Studies. Effective treatments for PTSD. 2nd ed. Oakbrook Terrace, IL: The Guilford Press; 2005.
19. Department of Veterans Affairs and Department of Defense. VA/DoD clinical practice guidelines. Management of Posttraumatic Stress Disorder and Acute Stress Reaction 2017. https://www.healthquality.va.gov/guidelines/MH/ptsd/. Published June 2017. Accessed February 26, 2018.
20. Phoenix Australia -Centre for Posttraumatic Mental Health. Australian guidelines for the treatment of acute stress disorder and posttraumatic stress disorder. Melbourne, Australia: Phoenix Australia Centre for Posttraumatic Mental Health; 2013.
21. World Health Organization. Guidelines for the management of conditions specifically related to stress. Geneva, Switzerland: World Health Organization Press; 2013.
22. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):201-213.
23. Stein DJ, Ipser J, McAnda N. Pharmacotherapy of posttraumatic stress disorder: a review of meta-analyses and treatment guidelines. CNS Spectr. 2009;14(suppl 1):25-31.
24. Forbes D, Creamer M, Bisson JI, et al. A guide to guidelines for the treatment of PTSD and related conditions. J Trauma Stress. 2010;23(5):537-552.
25. Nash WP, Watson PJ. Review of VA/DOD clinical practice guideline on management of acute stress and interventions to prevent posttraumatic stress disorder. J Rehabil Res Dev. 2012;49(5):637-648.
26. Birur B, Moore NC, Davis LL. An evidence-based review of early intervention and prevention of posttraumatic stress disorder. Community Ment Health J. 2017;53(2):183-201.
27. Breslau N, Davis GC. Posttraumatic stress disorder in an urban population of young adults: Risk factors for chronicity. Am J Psychiatry. 1992;149(5):671-675.
28. North CS, Pfefferbaum B, Kawasaki A, et al. Psychosocial adjustment of directly exposed survivors seven years after the Oklahoma City bombing. Compr Psychiatry. 2011;52(1):1-8
29. Rasco SS, North CS. An empirical study of employment and disability over three years among survivors of major disasters. J Am Acad Psychiatry Law. 2010;38(1):80-86.
Filling a Gender Gap in Research
Women, the VA wants your brains. It sounds a little disconcerting at first, but the National Center for PTSD and the nonprofit PINK Concussions are encouraging women to donate their brains for research.
In the past, says Dr. Carolyn Clancy, executive in charge of Veterans Health Administration, “the focus on TBI and PTSD brain research has primarily been based on male brains, without any active recruitment for women.” There has been almost no postmortem brain tissue available for study of injury in women. The VA also notes a lack of research on chronic traumatic encephalopathy in women. Only 2 peer-reviewed journal articles, both published in the early 1990s, have focused on women.
Women who are interested can take the “PINK Brain Pledge,” a nonbinding promise to leave their brains to science. They do not have to have a history of TBI or PTSD; brains also are needed for controls.
Women, the VA wants your brains. It sounds a little disconcerting at first, but the National Center for PTSD and the nonprofit PINK Concussions are encouraging women to donate their brains for research.
In the past, says Dr. Carolyn Clancy, executive in charge of Veterans Health Administration, “the focus on TBI and PTSD brain research has primarily been based on male brains, without any active recruitment for women.” There has been almost no postmortem brain tissue available for study of injury in women. The VA also notes a lack of research on chronic traumatic encephalopathy in women. Only 2 peer-reviewed journal articles, both published in the early 1990s, have focused on women.
Women who are interested can take the “PINK Brain Pledge,” a nonbinding promise to leave their brains to science. They do not have to have a history of TBI or PTSD; brains also are needed for controls.
Women, the VA wants your brains. It sounds a little disconcerting at first, but the National Center for PTSD and the nonprofit PINK Concussions are encouraging women to donate their brains for research.
In the past, says Dr. Carolyn Clancy, executive in charge of Veterans Health Administration, “the focus on TBI and PTSD brain research has primarily been based on male brains, without any active recruitment for women.” There has been almost no postmortem brain tissue available for study of injury in women. The VA also notes a lack of research on chronic traumatic encephalopathy in women. Only 2 peer-reviewed journal articles, both published in the early 1990s, have focused on women.
Women who are interested can take the “PINK Brain Pledge,” a nonbinding promise to leave their brains to science. They do not have to have a history of TBI or PTSD; brains also are needed for controls.
Medical Marijuana Redux
There were so many developments that occurred in the first months of 2018 that could potentially affect federal health care—the government shutdown, the proposed change in rights of conscience protections for federal health care professionals (HCPs), and more debate about medical marijuana in the VA—that it was hard to pick just one topic to discuss this month. In the end I felt it was time to examine how and in what ways the new VA policy on medical marijuana may have changed.
In 2014, before I became editor-in-chief of Federal Practitioner, I wrote an article analyzing the legal and ethical conflicts that arise for VA clinicians who practice under the federal regulations that prohibit them from prescribing medical marijuana or from completing forms or providing referrals for their patients who live in states where medical marijuana is legal.2 The article summarized the events and issues that led to the VA issuing a policy on medical marijuana in 2011. When that article was written, medical marijuana had been legalized in 20 states.
Now in March 2018, 29 states have passed legislation to permit marijuana use for medical purposes.3 Prior to issuing the revised version of its medical marijuana policy, the VA rumor mill went into high gear. Anticipatory stories predicted dramatic changes from the extreme of the VA penalizing veterans who used medical marijuana to allowing doctors to prescribe it. Such massive shifts are not typical of any bureaucracy, and indeed some VA officials denied that the revision represented any substantive movement in either direction.4
VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs was issued December 8, 2017.5 In accordance with federal regulation, its issuance superceded VHA Directive 2011-04 of the same title.6 According to the directive, its emphasis on discussion with veterans was a significant policy shift. “Major changes include adding policy to support the Veteran-provider relationship when discussing the use of medical marijuana and its impact on health including Veteran-specific treatment plans.” It should be noted that the prior directive did not prohibit or even discourage such conversations, and accompanying less official guidance actually promoted them.7
Interestingly, the new directive does not instruct HCPs to ask about medical marijuana in the way questions about alcohol, tobacco, and drug use as well as many other lifestyle factors are mandated. Asking a veteran about marijuana use would be a step toward medical mainstreaming. The burden is still on the veteran to bring up the subject—not an easy thing to do in light of the fear among some veterans that the VA will curtail benefits for a veteran caught using medical marijuana.
The new directive is a minor move toward appropriate medicalization. Practitioners are advised to discuss medical marijuana use with any veteran for whom it “may have clinical relevance” or who asks about medical marijuana. This underscores the need for VA practitioners to have access to up-to-date information in order to keep up with their Internet savvy patients and combat ever proliferating myths about the panacea-like properties of medical marijuana.
But when it comes down to the devilish details, the primary rules provide no deliverance from the impasse between state and federal law. Marijuana remains a Schedule I drug under the Controlled Substances Act. For purposes of federal health care, it still is, “a substance with a high potential for abuse without a currently acceptable medical use in treatment in the United States, and lacking accepted safety for use under medical supervision.”8 Although many vocal veterans as well as some federal practitioners, HCPs in the wider medical community, and more recently a number of politicians would challenge this regulation, federal lawprohibits prescribing medical marijuana. The new VA directive is more explicit in stating that VA practitioners cannot complete forms enrolling veterans or permitting their registration in state-approved medical marijuana programs. This restriction was implicit in the prior directive but has been a continuing source of confusion for HCPs. The new directive at least clarifies these restrictions.
Another point of clinical misunderstanding had been about whether HCPs in the VA could refer patients to state-approved medical marijuana programs and what exactly referral entails. There is a direct prohibition in the new directive on making referrals, yet the term remains undefined. Nothing in the directive contradicts the right of a veteran to access their medical records for purposes of registering for state-approved programs. But the directive does forcefully restate that if a veteran appears in an HCP’s office or at the pharmacy with an authorization or registration for medical marijuana from a state-approved program, the VA will neither provide the product nor pay for its purchase elsewhere. The more rules-based form of this directive also strongly states that possession of marijuana on VA grounds even for medical purposes and with state approval is a violation of federal regulation that may be prosecuted under the Controlled Substance Act.
The new directive does clarify a question that had arisen about VA employees’ participation in state-approved medical marijuana programs. VA employees, even those who do not receive their care at the VA, are prohibited from using medical marijuana. Individuals who use marijuana for medical indications often do so daily. Considering that a person may test positive for marijuana months after regular use, a segment of VA staff may be at risk for violating federal drug-free workplace regulations.9,10
The administrative aspects of the directive are tightened, which will help clinicians know what they are supposed to do when a veteran reports medical marijuana use; it is hoped that this will bring more consistency and fairness to the process. Practitioners continue to be required to enter a veteran’s reported use of medical marijuana in the electronic medical record under the section Non-VA/Herbal Medication/Over the Counter. When HCPs discuss the use of medical marijuana with patients, the requirement to document those discussions is instructive.
Those looking for a relaxation in the VA’s clinical approach will find little to cheer about. But there are a few rays of hope for those HCPs and patients trying to do the best they can in this catch-22 situation. First, the VA has stood firm that veterans cannot be excluded from other types of VA medical care due to their use of medical marijuana. “Veterans must not be denied VHA services solely because they are participating in State-approved marijuana programs.”5 The directive specifically acknowledges the clinical areas in which veteran medical marijuana use has been the most contentious: PTSD, substance use, and pain management. It also encourages HCPs to review potential drug interactions and how marijuana use may affect other types of medical or psychiatric care. These 3 areas also are the object of intensified congressional pressure and veteran service organization lobbying for the VA to not only incorporate these modalities into VA care, but also to expand research.11
Second, the phrase “modifying treatment plans,” which understandably makes patients and their advocates apprehensive, is qualified. To those clinicians who would prefer, either because of concerns of professional liability or personal belief, to have a black-and-white stance on the use of medical marijuana, the directive mandates that they must deal with the gray. “Providers need to make decisions to modify treatment plans based on marijuana use on a case-by-case basis.”5
Third, those modifications cannot be unilateral pronouncements, but must be the result of shared decisions making and mutual discussion. The only ground on which a practitioner can exercise any degree of soft paternalism is when the use of medical marijuana and treatment for another condition represents an evidence-based threat to the health and safety of the veteran. “Providers need to
Overall the policy has no big surprises, leaving those who hoped the revision would bring a softening of the VA’s institutional position and federal law frustrated. Those who sought a strengthening of VA policy based on those same regulations regarding the use of medical marijuana will be equally thwarted. And those clinicians who are just trying to do the right thing as HCPs who work for the federal government and for their patients who are interested only in relief from their most troubling ailments, will stay right where they were, suspended over the ethical chasm that medical marijuana generates between state and federal law.
1. Curie M. Pierre Curie With Autobiographical Notes. Kellogg C, Kellogg V, trans. New York: Macmillan; 1923.
2. Geppert CMA. Legal and clinical evolution of Veterans Health Administration policy on medical marijuana. Fed Pract. 2014;31(3):6-12.
3. National Conference of State Legislators. State Medical Marijuana Laws. http://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx Updated February 15, 2018. Accessed March 2, 2018.
4. Shane L. VA refutes rumors of new policy on medical marijuana. https://www.militarytimes.com/veterans/2017/12/19/va-refutes-rumors-of-a-new-policy-on-medical-marijuana. Published December 19, 2017. Accessed March 2, 2018.
5. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs. December 8, 2017.
6. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2011-004, Access to Clinical Programs for Veterans Participating in State-Approved Marijuana Programs, dated January 31, 2011 (rescinded).
7. U.S. Department of Veterans Affairs, Veterans Health Administration. Clinical considerations regarding veteran patients who participate in state-approved medical marijuana programs. Washington, DC; 2010. [Nonpublic document.]
8. 21 U.S.C. 801 et al, the Controlled Substances Act.
9. Welch SA. The pharmacology of cannabinoids. In: Principles of Addiction Medicine: The Essentials. Cavacuiti CA, ed. Philadelphia, PA: Lippincott-Williams & Wilkins; 2011:62.
10. U.S. Department of Veterans Affairs. VA Handbook 5383.2, VA drug-free workplace plan. https://www.va.gov/vapubs/search_action.cfm?dType=2. Published April 11, 1997. Accessed March 2, 2018.
11. Zezima K. VA says it won’t study medical marijuana’s effect on veterans. The Washington Post. https://www.washingtonpost.com/news/post-nation/wp/2018/01/16/va-says-it-wont-study-medical-marijuanas-effect-on-veterans/?utm_term=.9d554109d135. Published January 16, 2018. Accessed March 2, 2018.
There were so many developments that occurred in the first months of 2018 that could potentially affect federal health care—the government shutdown, the proposed change in rights of conscience protections for federal health care professionals (HCPs), and more debate about medical marijuana in the VA—that it was hard to pick just one topic to discuss this month. In the end I felt it was time to examine how and in what ways the new VA policy on medical marijuana may have changed.
In 2014, before I became editor-in-chief of Federal Practitioner, I wrote an article analyzing the legal and ethical conflicts that arise for VA clinicians who practice under the federal regulations that prohibit them from prescribing medical marijuana or from completing forms or providing referrals for their patients who live in states where medical marijuana is legal.2 The article summarized the events and issues that led to the VA issuing a policy on medical marijuana in 2011. When that article was written, medical marijuana had been legalized in 20 states.
Now in March 2018, 29 states have passed legislation to permit marijuana use for medical purposes.3 Prior to issuing the revised version of its medical marijuana policy, the VA rumor mill went into high gear. Anticipatory stories predicted dramatic changes from the extreme of the VA penalizing veterans who used medical marijuana to allowing doctors to prescribe it. Such massive shifts are not typical of any bureaucracy, and indeed some VA officials denied that the revision represented any substantive movement in either direction.4
VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs was issued December 8, 2017.5 In accordance with federal regulation, its issuance superceded VHA Directive 2011-04 of the same title.6 According to the directive, its emphasis on discussion with veterans was a significant policy shift. “Major changes include adding policy to support the Veteran-provider relationship when discussing the use of medical marijuana and its impact on health including Veteran-specific treatment plans.” It should be noted that the prior directive did not prohibit or even discourage such conversations, and accompanying less official guidance actually promoted them.7
Interestingly, the new directive does not instruct HCPs to ask about medical marijuana in the way questions about alcohol, tobacco, and drug use as well as many other lifestyle factors are mandated. Asking a veteran about marijuana use would be a step toward medical mainstreaming. The burden is still on the veteran to bring up the subject—not an easy thing to do in light of the fear among some veterans that the VA will curtail benefits for a veteran caught using medical marijuana.
The new directive is a minor move toward appropriate medicalization. Practitioners are advised to discuss medical marijuana use with any veteran for whom it “may have clinical relevance” or who asks about medical marijuana. This underscores the need for VA practitioners to have access to up-to-date information in order to keep up with their Internet savvy patients and combat ever proliferating myths about the panacea-like properties of medical marijuana.
But when it comes down to the devilish details, the primary rules provide no deliverance from the impasse between state and federal law. Marijuana remains a Schedule I drug under the Controlled Substances Act. For purposes of federal health care, it still is, “a substance with a high potential for abuse without a currently acceptable medical use in treatment in the United States, and lacking accepted safety for use under medical supervision.”8 Although many vocal veterans as well as some federal practitioners, HCPs in the wider medical community, and more recently a number of politicians would challenge this regulation, federal lawprohibits prescribing medical marijuana. The new VA directive is more explicit in stating that VA practitioners cannot complete forms enrolling veterans or permitting their registration in state-approved medical marijuana programs. This restriction was implicit in the prior directive but has been a continuing source of confusion for HCPs. The new directive at least clarifies these restrictions.
Another point of clinical misunderstanding had been about whether HCPs in the VA could refer patients to state-approved medical marijuana programs and what exactly referral entails. There is a direct prohibition in the new directive on making referrals, yet the term remains undefined. Nothing in the directive contradicts the right of a veteran to access their medical records for purposes of registering for state-approved programs. But the directive does forcefully restate that if a veteran appears in an HCP’s office or at the pharmacy with an authorization or registration for medical marijuana from a state-approved program, the VA will neither provide the product nor pay for its purchase elsewhere. The more rules-based form of this directive also strongly states that possession of marijuana on VA grounds even for medical purposes and with state approval is a violation of federal regulation that may be prosecuted under the Controlled Substance Act.
The new directive does clarify a question that had arisen about VA employees’ participation in state-approved medical marijuana programs. VA employees, even those who do not receive their care at the VA, are prohibited from using medical marijuana. Individuals who use marijuana for medical indications often do so daily. Considering that a person may test positive for marijuana months after regular use, a segment of VA staff may be at risk for violating federal drug-free workplace regulations.9,10
The administrative aspects of the directive are tightened, which will help clinicians know what they are supposed to do when a veteran reports medical marijuana use; it is hoped that this will bring more consistency and fairness to the process. Practitioners continue to be required to enter a veteran’s reported use of medical marijuana in the electronic medical record under the section Non-VA/Herbal Medication/Over the Counter. When HCPs discuss the use of medical marijuana with patients, the requirement to document those discussions is instructive.
Those looking for a relaxation in the VA’s clinical approach will find little to cheer about. But there are a few rays of hope for those HCPs and patients trying to do the best they can in this catch-22 situation. First, the VA has stood firm that veterans cannot be excluded from other types of VA medical care due to their use of medical marijuana. “Veterans must not be denied VHA services solely because they are participating in State-approved marijuana programs.”5 The directive specifically acknowledges the clinical areas in which veteran medical marijuana use has been the most contentious: PTSD, substance use, and pain management. It also encourages HCPs to review potential drug interactions and how marijuana use may affect other types of medical or psychiatric care. These 3 areas also are the object of intensified congressional pressure and veteran service organization lobbying for the VA to not only incorporate these modalities into VA care, but also to expand research.11
Second, the phrase “modifying treatment plans,” which understandably makes patients and their advocates apprehensive, is qualified. To those clinicians who would prefer, either because of concerns of professional liability or personal belief, to have a black-and-white stance on the use of medical marijuana, the directive mandates that they must deal with the gray. “Providers need to make decisions to modify treatment plans based on marijuana use on a case-by-case basis.”5
Third, those modifications cannot be unilateral pronouncements, but must be the result of shared decisions making and mutual discussion. The only ground on which a practitioner can exercise any degree of soft paternalism is when the use of medical marijuana and treatment for another condition represents an evidence-based threat to the health and safety of the veteran. “Providers need to
Overall the policy has no big surprises, leaving those who hoped the revision would bring a softening of the VA’s institutional position and federal law frustrated. Those who sought a strengthening of VA policy based on those same regulations regarding the use of medical marijuana will be equally thwarted. And those clinicians who are just trying to do the right thing as HCPs who work for the federal government and for their patients who are interested only in relief from their most troubling ailments, will stay right where they were, suspended over the ethical chasm that medical marijuana generates between state and federal law.
There were so many developments that occurred in the first months of 2018 that could potentially affect federal health care—the government shutdown, the proposed change in rights of conscience protections for federal health care professionals (HCPs), and more debate about medical marijuana in the VA—that it was hard to pick just one topic to discuss this month. In the end I felt it was time to examine how and in what ways the new VA policy on medical marijuana may have changed.
In 2014, before I became editor-in-chief of Federal Practitioner, I wrote an article analyzing the legal and ethical conflicts that arise for VA clinicians who practice under the federal regulations that prohibit them from prescribing medical marijuana or from completing forms or providing referrals for their patients who live in states where medical marijuana is legal.2 The article summarized the events and issues that led to the VA issuing a policy on medical marijuana in 2011. When that article was written, medical marijuana had been legalized in 20 states.
Now in March 2018, 29 states have passed legislation to permit marijuana use for medical purposes.3 Prior to issuing the revised version of its medical marijuana policy, the VA rumor mill went into high gear. Anticipatory stories predicted dramatic changes from the extreme of the VA penalizing veterans who used medical marijuana to allowing doctors to prescribe it. Such massive shifts are not typical of any bureaucracy, and indeed some VA officials denied that the revision represented any substantive movement in either direction.4
VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs was issued December 8, 2017.5 In accordance with federal regulation, its issuance superceded VHA Directive 2011-04 of the same title.6 According to the directive, its emphasis on discussion with veterans was a significant policy shift. “Major changes include adding policy to support the Veteran-provider relationship when discussing the use of medical marijuana and its impact on health including Veteran-specific treatment plans.” It should be noted that the prior directive did not prohibit or even discourage such conversations, and accompanying less official guidance actually promoted them.7
Interestingly, the new directive does not instruct HCPs to ask about medical marijuana in the way questions about alcohol, tobacco, and drug use as well as many other lifestyle factors are mandated. Asking a veteran about marijuana use would be a step toward medical mainstreaming. The burden is still on the veteran to bring up the subject—not an easy thing to do in light of the fear among some veterans that the VA will curtail benefits for a veteran caught using medical marijuana.
The new directive is a minor move toward appropriate medicalization. Practitioners are advised to discuss medical marijuana use with any veteran for whom it “may have clinical relevance” or who asks about medical marijuana. This underscores the need for VA practitioners to have access to up-to-date information in order to keep up with their Internet savvy patients and combat ever proliferating myths about the panacea-like properties of medical marijuana.
But when it comes down to the devilish details, the primary rules provide no deliverance from the impasse between state and federal law. Marijuana remains a Schedule I drug under the Controlled Substances Act. For purposes of federal health care, it still is, “a substance with a high potential for abuse without a currently acceptable medical use in treatment in the United States, and lacking accepted safety for use under medical supervision.”8 Although many vocal veterans as well as some federal practitioners, HCPs in the wider medical community, and more recently a number of politicians would challenge this regulation, federal lawprohibits prescribing medical marijuana. The new VA directive is more explicit in stating that VA practitioners cannot complete forms enrolling veterans or permitting their registration in state-approved medical marijuana programs. This restriction was implicit in the prior directive but has been a continuing source of confusion for HCPs. The new directive at least clarifies these restrictions.
Another point of clinical misunderstanding had been about whether HCPs in the VA could refer patients to state-approved medical marijuana programs and what exactly referral entails. There is a direct prohibition in the new directive on making referrals, yet the term remains undefined. Nothing in the directive contradicts the right of a veteran to access their medical records for purposes of registering for state-approved programs. But the directive does forcefully restate that if a veteran appears in an HCP’s office or at the pharmacy with an authorization or registration for medical marijuana from a state-approved program, the VA will neither provide the product nor pay for its purchase elsewhere. The more rules-based form of this directive also strongly states that possession of marijuana on VA grounds even for medical purposes and with state approval is a violation of federal regulation that may be prosecuted under the Controlled Substance Act.
The new directive does clarify a question that had arisen about VA employees’ participation in state-approved medical marijuana programs. VA employees, even those who do not receive their care at the VA, are prohibited from using medical marijuana. Individuals who use marijuana for medical indications often do so daily. Considering that a person may test positive for marijuana months after regular use, a segment of VA staff may be at risk for violating federal drug-free workplace regulations.9,10
The administrative aspects of the directive are tightened, which will help clinicians know what they are supposed to do when a veteran reports medical marijuana use; it is hoped that this will bring more consistency and fairness to the process. Practitioners continue to be required to enter a veteran’s reported use of medical marijuana in the electronic medical record under the section Non-VA/Herbal Medication/Over the Counter. When HCPs discuss the use of medical marijuana with patients, the requirement to document those discussions is instructive.
Those looking for a relaxation in the VA’s clinical approach will find little to cheer about. But there are a few rays of hope for those HCPs and patients trying to do the best they can in this catch-22 situation. First, the VA has stood firm that veterans cannot be excluded from other types of VA medical care due to their use of medical marijuana. “Veterans must not be denied VHA services solely because they are participating in State-approved marijuana programs.”5 The directive specifically acknowledges the clinical areas in which veteran medical marijuana use has been the most contentious: PTSD, substance use, and pain management. It also encourages HCPs to review potential drug interactions and how marijuana use may affect other types of medical or psychiatric care. These 3 areas also are the object of intensified congressional pressure and veteran service organization lobbying for the VA to not only incorporate these modalities into VA care, but also to expand research.11
Second, the phrase “modifying treatment plans,” which understandably makes patients and their advocates apprehensive, is qualified. To those clinicians who would prefer, either because of concerns of professional liability or personal belief, to have a black-and-white stance on the use of medical marijuana, the directive mandates that they must deal with the gray. “Providers need to make decisions to modify treatment plans based on marijuana use on a case-by-case basis.”5
Third, those modifications cannot be unilateral pronouncements, but must be the result of shared decisions making and mutual discussion. The only ground on which a practitioner can exercise any degree of soft paternalism is when the use of medical marijuana and treatment for another condition represents an evidence-based threat to the health and safety of the veteran. “Providers need to
Overall the policy has no big surprises, leaving those who hoped the revision would bring a softening of the VA’s institutional position and federal law frustrated. Those who sought a strengthening of VA policy based on those same regulations regarding the use of medical marijuana will be equally thwarted. And those clinicians who are just trying to do the right thing as HCPs who work for the federal government and for their patients who are interested only in relief from their most troubling ailments, will stay right where they were, suspended over the ethical chasm that medical marijuana generates between state and federal law.
1. Curie M. Pierre Curie With Autobiographical Notes. Kellogg C, Kellogg V, trans. New York: Macmillan; 1923.
2. Geppert CMA. Legal and clinical evolution of Veterans Health Administration policy on medical marijuana. Fed Pract. 2014;31(3):6-12.
3. National Conference of State Legislators. State Medical Marijuana Laws. http://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx Updated February 15, 2018. Accessed March 2, 2018.
4. Shane L. VA refutes rumors of new policy on medical marijuana. https://www.militarytimes.com/veterans/2017/12/19/va-refutes-rumors-of-a-new-policy-on-medical-marijuana. Published December 19, 2017. Accessed March 2, 2018.
5. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs. December 8, 2017.
6. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2011-004, Access to Clinical Programs for Veterans Participating in State-Approved Marijuana Programs, dated January 31, 2011 (rescinded).
7. U.S. Department of Veterans Affairs, Veterans Health Administration. Clinical considerations regarding veteran patients who participate in state-approved medical marijuana programs. Washington, DC; 2010. [Nonpublic document.]
8. 21 U.S.C. 801 et al, the Controlled Substances Act.
9. Welch SA. The pharmacology of cannabinoids. In: Principles of Addiction Medicine: The Essentials. Cavacuiti CA, ed. Philadelphia, PA: Lippincott-Williams & Wilkins; 2011:62.
10. U.S. Department of Veterans Affairs. VA Handbook 5383.2, VA drug-free workplace plan. https://www.va.gov/vapubs/search_action.cfm?dType=2. Published April 11, 1997. Accessed March 2, 2018.
11. Zezima K. VA says it won’t study medical marijuana’s effect on veterans. The Washington Post. https://www.washingtonpost.com/news/post-nation/wp/2018/01/16/va-says-it-wont-study-medical-marijuanas-effect-on-veterans/?utm_term=.9d554109d135. Published January 16, 2018. Accessed March 2, 2018.
1. Curie M. Pierre Curie With Autobiographical Notes. Kellogg C, Kellogg V, trans. New York: Macmillan; 1923.
2. Geppert CMA. Legal and clinical evolution of Veterans Health Administration policy on medical marijuana. Fed Pract. 2014;31(3):6-12.
3. National Conference of State Legislators. State Medical Marijuana Laws. http://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx Updated February 15, 2018. Accessed March 2, 2018.
4. Shane L. VA refutes rumors of new policy on medical marijuana. https://www.militarytimes.com/veterans/2017/12/19/va-refutes-rumors-of-a-new-policy-on-medical-marijuana. Published December 19, 2017. Accessed March 2, 2018.
5. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1315, Access to Clinical Programs for Veterans Participating in State Medical Marijuana Programs. December 8, 2017.
6. U.S. Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2011-004, Access to Clinical Programs for Veterans Participating in State-Approved Marijuana Programs, dated January 31, 2011 (rescinded).
7. U.S. Department of Veterans Affairs, Veterans Health Administration. Clinical considerations regarding veteran patients who participate in state-approved medical marijuana programs. Washington, DC; 2010. [Nonpublic document.]
8. 21 U.S.C. 801 et al, the Controlled Substances Act.
9. Welch SA. The pharmacology of cannabinoids. In: Principles of Addiction Medicine: The Essentials. Cavacuiti CA, ed. Philadelphia, PA: Lippincott-Williams & Wilkins; 2011:62.
10. U.S. Department of Veterans Affairs. VA Handbook 5383.2, VA drug-free workplace plan. https://www.va.gov/vapubs/search_action.cfm?dType=2. Published April 11, 1997. Accessed March 2, 2018.
11. Zezima K. VA says it won’t study medical marijuana’s effect on veterans. The Washington Post. https://www.washingtonpost.com/news/post-nation/wp/2018/01/16/va-says-it-wont-study-medical-marijuanas-effect-on-veterans/?utm_term=.9d554109d135. Published January 16, 2018. Accessed March 2, 2018.
Neuromodulation for Treatment-Refractory PTSD (FULL)
Failure of fear extinction is a core feature of posttraumatic stress disorder (PTSD).1 Recently, it was confirmed that the amygdala and the orbitofrontal cortex are crucial for both fear acquisition and fear extinction.2 The amygdala was found to have neurons active only during fear acquisition, and other neurons active only during fear extinction.3 In essence, the balance of activity between these 2 neuronal populations determines whether if an incoming stimulus is feared or not feared. This balance is under the influence of several cognitive domains, including memory, reward, and executive function.
In PTSD, the equilibrium is shifted heavily toward fear acquisition. The majority of patients spontaneously regain the capacity for fear extinction over time4 or with the help of treatment.5,6 Nonetheless, some patients with severe PTSD seem unable to recover the ability of fear extinction and remain refractory to both standard and novel psychotherapeutic or psychopharmacologic treatments.7 For these patients, direct modulation of the neural activity in the amygdala may permit fear extinction. This article describes the rationale for using deep brain stimulation (DBS) and initial results from the first-ever clinical trial.
Deep Brain Stimulation
Deep brain stimulation involves inserting electrodes in precise cerebral targets and then connecting the leads to a pulse generator (similar to a pacemaker) inserted in a subclavicular pocket. The generator controls the electrical signal (amplitude, pulse width, pulse frequency) delivered to the brain target and can be adjusted with use of a noninvasive programmer. In 1997, the FDA approved DBS for patients with Parkinson disease or essential tremor. Since then, its efficacy in these movement disorders has been confirmed in several studies.8,9
The mechanism by which the small electrical pulses of DBS influence activity is not clear. Clinically, DBS functionally inhibits the activity of local neurons.10 One theory describes “frequency jamming,” a concept similar to cardiac overdrive pacing in which the resultant high-frequency neuronal signal is meaningless and discounted by the rest of the brain.11
Over the years, DBS has demonstrated a strong safety profile.12 The risks of electrode insertion are mitigated with targeting based on high-quality magnetic resonance imaging (MRI) and computed tomography (Figure). Unlike a destructive lesion, DBS is reversible, and the implanted system can be removed in its entirety. Histologic analyses have shown only a small amount of scarring around the electrode tip.13 In movement disorder treatment, clinical experience has shown that stimulation-related adverse effects (AEs) are reversible with readjustment of stimulation parameters by external programmer.14
Novel Applications of DBS
The advantageous safety profile of DBS has permitted its evaluation in the treatment of other conditions thought to have malfunctioning networks at their core—such as intractable epilepsy (in resective surgery noncandidates).15,16 Although several trials have shown promising results of using DBS for treatment-resistant depression,17 the results of pivotal sham-controlled trials have been mixed.18,19 Obsessive-compulsive disorder, on the other hand, received the FDA humanitarian device exemption designation on the basis of positive long-term results.20 In treatment-resistant depression and obsessive-compulsive disorder, functional neuroimaging has identified DBS targets.21,22 Functional MRI or positron emission tomography (PET) images can be compared at resting state, at symptomatic state, and after treatment response. Nodes hyperactive during a symptomatic state and less active after successful treatment can be targeted with high-frequency DBS to directly reduce the hyperactivity and indirectly modulate or normalize the overall function of the circuit.23
Given the functional MRI and O15 (oxygen-15) PET evidence of amygdala hyperactivity in patients with PTSD having core symptoms,24-26 the authors hypothesized that high-frequency DBS targeting of the amygdala would improve PTSD-associated hyperarousal and reexperiencing symptoms in treatment-refractory patients. Indirect data supporting this hypothesis include a correlation between amygdala hyperactivity of increased intensity and symptom severity measured with the Clinician-Administered PTSD Scale (CAPS),27 and a correlation between reduced pretreatment amygdala hyperactivity and successful cognitive-behavioral treatment.28,29
Preclinical Work
Using a rodent model in which a novel object serves as a cue reminder of foot shocks (traumatic event), the authors tested the hypothesis that amygdala DBS would reduce PTSD-like symptoms.30 When untreated rats were presented with the object in their cage a week after the initial exposure, they immediately buried the object under bedding to avoid being reminded of the shocks. In contrast, rats treated with DBS did not bury the object. In most cases, in fact, they played with it.
The authors also replicated their results but with the addition of rats treated with paroxetine.31 Using the same rodent model, they found DBS superior to paroxetine in treating PTSD-like symptoms. This study had a crossover design: DBS and sham DBS. Briefly, 20 rats received an electrode in the amygdala and were exposed to inescapable shocks in the presence of the cue object. The rats were then randomly assigned to a DBS group (10 rats) or a sham-DBS group (10 rats). After 1 week, behavioral testing showed fear extinction in the DBS group and no improvement in the sham-DBS group. Then the groups were switched: The rats originally treated with DBS received no treatment, and the rats that were originally sham-treated underwent DBS. One week later, behavioral testing showed acquisition of fear extinction in all the rats. These results suggested DBS can be effective even when delayed after establishment of fear persistence and PTSD symptoms. These results also showed that DBS effects persist even after therapy discontinuation.
Similarly, other investigators have reported that the role of the amygdala is not limited to fear acquisition; it extends to fear expression. A lesion in the amygdala can prevent fear expression even if the disruption is performed subsequent to fear-conditioning training.32 This finding is important for humans, as DBS would be initiated during the chronic phase of the disorder, after failure of less invasive treatment options, such as pharmacotherapy and psychotherapy.
Early Clinical Experience
The authors have initiated the first ever clinical trial (NCT02091843) evaluating use of DBS for PTSD and are now recruiting patients. Enrollment is limited to 6 combat veterans with disabling PTSD that has not responded to pharmacotherapy and psychotherapy. This VA-funded single-site study, being conducted at the VA Greater Los Angeles Healthcare System (VAGLAHS), was approved by the VAGLAHS Institutional Review Board and the FDA. The authors have published the 2-year trial’s protocol, which includes an active-versus-sham stimulation phase; continuous electroencephalogram monitoring; baseline and posttreatment 18FDG (fluorodeoxyglucose) PET performed during a resting state vs during investigator-guided exposure to trauma reminders; and extensive psychological and neuropsychological assessments.33 The literature includes only 1 case report on amygdala DBS.34 The authors of that report used DBS of the basolateral nucleus of the amygdala to treat a teenaged boy with severe autism and found that the therapy was safe.
As of this writing, the authors have recruited and implanted 1 patient and reported on his clinical results (including baseline PET) over the first 8 months of stimulation35 and on the electrophysiologic findings over the first year.36 After experiencing extremely severe combat PTSD refractory to pharmacotherapy and psychotherapy treatments for more than 20 years, the patient treated with DBS is now experiencing substantial symptom relief, and his CAPS score (primary outcome measure) has improved by about 40%. He has tolerated continuous stimulation without any serious DBS-related AEs for up to 16 months. Notably, he has not had a single severe combat nightmare in a year—in stark contrast to nightly combat nightmares during the 20-year period leading to the trial. Furthermore, he has not been having any episodes of severe dissociation, which had been a common disabling problem before the trial. He has taken a second trip out of the country, improved his relationships with family, and made strides (albeit limited) in pursuing additional social interactions.
Avoidance remains a major problem. He recently left his job after 7 years, because he prefers a more nature-oriented rather than people-oriented environment. In addition, his interest in intensive psychotherapy has increased, and he has been considering options for spending more time working on his therapy.
Over 15 months of treatment, the patient’s CAPS total and subscale scores have decreased—his symptoms have improved (Table).21 He has had rapid and substantial reductions in recurrence and hyperarousal symptoms but slower improvement in avoidance. Improvements in emotional reactivity would be expected to occur before any change in behavior (eg, avoidance). Patients likely must first recognize changes in emotional reactivity to events before they can engage in a cognitive process to modify learned behavioral responses to those events.
After about 9 months of treatment, all of the study patient’s symptoms were somewhat stabilized, and the authors began making gradual stimulation adjustments to the latest parameters—3.5 V, 60 µs, and 160 Hz for the right electrode and 1.5 V, 60 µs, and 160 Hz for the left electrode—using the contacts in the basolateral nucleus of the amygdala, per postoperative neuroimaging.3
After 15 to 18 months, when improvement peaked at 48% symptom reduction from baseline, the patient experienced psychiatric decompensation (depression, suicide gesture) not attributable to changes in stimulation settings and not associated with exacerbation of PTSD symptoms. Treatment team members and independent psychiatric consultants attributed the decompensation to the patient’s difficulty in changing a long-standing avoidant behavior routine, owing to severe recurrence and hyperarousal symptoms in the past. His persistent inability to overcome avoidance and isolation, despite core PTSD symptom improvement, had left him feeling worthless.
The patient remains in the study but also is participating in other medication and psychotherapy trials and is making a career change. Periodic decompensations may be part of the treatment course as patients reach a more complex and volatile phase of improvement that requires more intensive cognitive reprocessing. If this proves to be the case with other patients enrolling in the study, intensive psychotherapy that addresses cognitive and emotional PTSD symptoms may be needed once there is improvement in intrusive and hyperarousal symptoms.
Conclusion
Deep brain stimulation has been successful in treating Parkinson disease and essential tremor. Physiologically, DBS seems to inhibit specific brain regions’ dysfunctional activity stemming from a disease process. Deep brain stimulation-induced inhibition of a dysfunctional node improves clinical outcomes in movement disorders.
Given the reversibility and positive safety profile of DBS, new applications are being studied. The authors propose that DBS may benefit patients with severe treatment-refractory PTSD. Their first patient’s core PTSD symptoms have improved significantly, as expected, but as in other psychiatric DBS cases, the seriousness and chronicity of his illness may be complicating the course of recovery. The authors plan to recruit 6 patients for this early-phase safety trial.
Click here to read the digital edition.
1. Milad MR, Pitman RK, Ellis CB, et al. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry. 2009;66(12):1075-1082.
2. Marin MF, Song H, VanElzakker MB, et al. Association of resting metabolism in the fear neural network with extinction recall activations and clinical measures in trauma-exposed individuals. Am J Psychiatry. 2016;173(9):930-938.
3. Herry C, Ciocchi S, Senn V, Demmou L, Müller C, Lüthi A. Switching on and off fear by distinct neuronal circuits. Nature. 2008;454(7204):600-606.
4. Morina N, Wicherts JM, Lobbrecht J, Priebe S. Remission from post-traumatic stress disorder in adults: a systematic review and meta-analysis of long term outcome studies. Clin Psychol Rev. 2014;34(3):249-255.
5. Steenkamp MM, Litz BT, Hoge CW, Marmar CR. Psychotherapy for military-related PTSD: a review of randomized clinical trials. JAMA. 2015;314(5):489-500.
6. Hoskins M, Pearce J, Bethell A, et al. Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis. Br J Psychiatry. 2015;206(2):93-100.
7. Koek RJ, Schwartz HN, Scully S, et al. Treatment-refractory posttraumatic stress disorder (TRPTSD): a review and framework for the future. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:170-218.
8. Wagle Shukla A, Okun MS. State of the art for deep brain stimulation therapy in movement disorders: a clinical and technological perspective. IEEE Rev Biomed Eng. 2016;9:219-233.
9. Weaver FM, Follett K, Stern M, et al; CSP 468 Study Group. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. JAMA. 2009;301(1):63-73.
10. Benabid AL, Benazzouz A, Hoffmann D, Limousin P, Krack P, Pollack P. Long-term electrical inhibition of deep brain targets in movement disorders. Mov Disord. 1998;13(suppl 3):119-125.
11. Benabid AL, Wallace B, Mitrofanis J, et al. A putative generalized model of the effects and mechanism of action of high frequency electrical stimulation of the central nervous system. Acta Neurol Belg. 2005;105(3):149-157.
12. Fenoy AJ, Simpson RK Jr. Risks of common complications in deep brain stimulation surgery: management and avoidance. J Neurosurg. 2014;120(1):132-139.
13. DiLorenzo DJ, Jankovic J, Simpson RK, Takei H, Powell SZ. Neurohistopathological findings at the electrode–tissue interface in long-term deep brain stimulation: systematic literature review, case report, and assessment of stimulation threshold safety. Neuromodulation. 2014;17(5):405-418.
14. Revell MA. Deep brain stimulation for movement disorders. Nurs Clin North Am. 2015;50(4):691-701.
15. Fisher R, Salanova V, Witt T, et al; SANTE Study Group. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy. Epilepsia. 2010;51(5):899-908.
16. Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
17. Berlim MT, McGirr A, Van den Eynde F, Fleck MP, Giacobbe P. Effectiveness and acceptability of deep brain stimulation (DBS) of the subgenual cingulate cortex for treatment-resistant depression: a systematic review and exploratory meta-analysis. J Affect Disord. 2014;159:31-38.
18. Dougherty DD, Rezai AR, Carpenter LL, et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol Psychiatry. 2015;78(4):240-248.
19. Bergfeld IO, Mantione M, Hoogendoorn ML, et al. Deep brain stimulation of the ventral anterior limb of the internal capsule for treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2016;73(5):456-464.
20. Greenberg BD, Malone DA, Friehs GM, et al. Three-year outcomes in deep brain stimulation for highly resistant obsessive-compulsive disorder. Neuropsychopharmacology. 2006;31(11):2384-2393.
21. Mayber HS, Liotti M, Brannan SK, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156(5):675-682.
22. Rauch SL, Jenike MA, Alpert NM, et al. Regional cerebral blood flow measured during symptom provocation in obsessive-compulsive disorder using oxygen 15-labeled carbon dioxide and positron emission tomography. Arch Gen Psychiatry. 1994;51(1):62-70.
23. Williams NR, Taylor JJ, Lamb K, Hanlon CA, Short EB, George MS. Role of functional imaging in the development and refinement of invasive neuromodulation for psychiatric disorders. World J Radiol. 2014;6(10):756-778.
24. Francati V, Vermetten E, Bremner JD. Functional neuroimaging studies in posttraumatic stress disorder: review of current methods and findings. Depress Anxiety. 2007;24(3):202-218.
25. Shin LM, Orr SP, Carson MA, et al. Regional cerebral blood flow in the amygdala and medial prefrontal cortex during traumatic imagery in male and female Vietnam veterans with PTSD. Arch Gen Psychiatry. 2004;61(2):168-176.
26. Armony JL, Corbo V, Clément MH, Brunet A. Amygdala response in patients with acute PTSD to masked and unmasked emotional facial expressions. Am J Psychiatry. 2005;162(10):1961-1963.
27. Blake DD, Weathers FW, Nagy LM, et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress. 1995;8(1):75-90.
28. Felmingham K, Kemp A, Williams L, et al. Changes in anterior cingulate and amygdala after cognitive behavior therapy of posttraumatic stress disorder. Psychol Sci. 2007;18(2):127-129.
29. Peres JF, Newberg AB, Mercante JP, et al. Cerebral blood flow changes during retrieval of traumatic memories before and after psychotherapy: a SPECT study. Psychol Med. 2007;37(10):1481-1491.
30. Langevin JP, De Salles AA, Kosoyan HP, Krahl SE. Deep brain stimulation of the amygdala alleviates post-traumatic stress disorder symptoms in a rat model. J Psychiatr Res. 2010;44(16):1241-1245.
31. Stidd DA, Vogelsang K, Krahl SE, Langevin JP, Fellous JM. Amygdala deep brain stimulation is superior to paroxetine treatment in a rat model of posttraumatic stress disorder. Brain Stimul. 2013;6(6):837-844.
32. Anglada-Figueroa D, Quirk GJ. Lesions of the basal amygdala block expression of conditioned fear but not extinction. J Neurosci. 2005;25(42):9680-9685.
33. Koek RJ, Langevin JP, Krahl SE, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory combat post-traumatic stress disorder (PTSD): study protocol for a pilot randomized controlled trial with blinded, staggered onset of stimulation. Trials. 2014;15:356.
34. Sturm V, Fricke O, Bührle CP, et al. DBS in the basolateral amygdala improves symptoms of autism and related self-injurious behavior: a case report and hypothesis on the pathogenesis of the disorder. Front Hum Neurosci. 2013;6:341.
35. Langevin JP, Koek RJ, Schwartz HN, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory posttraumatic stress disorder. Biol Psychiatry. 2016;79(10):e82-e84.
36. Langevin JP, Chen JW, Koek RJ, et al. Deep brain stimulation of the basolateral amygdala: targeting technique and electrodiagnostic findings. Brain Sci. 2016;6(3):E28.
Failure of fear extinction is a core feature of posttraumatic stress disorder (PTSD).1 Recently, it was confirmed that the amygdala and the orbitofrontal cortex are crucial for both fear acquisition and fear extinction.2 The amygdala was found to have neurons active only during fear acquisition, and other neurons active only during fear extinction.3 In essence, the balance of activity between these 2 neuronal populations determines whether if an incoming stimulus is feared or not feared. This balance is under the influence of several cognitive domains, including memory, reward, and executive function.
In PTSD, the equilibrium is shifted heavily toward fear acquisition. The majority of patients spontaneously regain the capacity for fear extinction over time4 or with the help of treatment.5,6 Nonetheless, some patients with severe PTSD seem unable to recover the ability of fear extinction and remain refractory to both standard and novel psychotherapeutic or psychopharmacologic treatments.7 For these patients, direct modulation of the neural activity in the amygdala may permit fear extinction. This article describes the rationale for using deep brain stimulation (DBS) and initial results from the first-ever clinical trial.
Deep Brain Stimulation
Deep brain stimulation involves inserting electrodes in precise cerebral targets and then connecting the leads to a pulse generator (similar to a pacemaker) inserted in a subclavicular pocket. The generator controls the electrical signal (amplitude, pulse width, pulse frequency) delivered to the brain target and can be adjusted with use of a noninvasive programmer. In 1997, the FDA approved DBS for patients with Parkinson disease or essential tremor. Since then, its efficacy in these movement disorders has been confirmed in several studies.8,9
The mechanism by which the small electrical pulses of DBS influence activity is not clear. Clinically, DBS functionally inhibits the activity of local neurons.10 One theory describes “frequency jamming,” a concept similar to cardiac overdrive pacing in which the resultant high-frequency neuronal signal is meaningless and discounted by the rest of the brain.11
Over the years, DBS has demonstrated a strong safety profile.12 The risks of electrode insertion are mitigated with targeting based on high-quality magnetic resonance imaging (MRI) and computed tomography (Figure). Unlike a destructive lesion, DBS is reversible, and the implanted system can be removed in its entirety. Histologic analyses have shown only a small amount of scarring around the electrode tip.13 In movement disorder treatment, clinical experience has shown that stimulation-related adverse effects (AEs) are reversible with readjustment of stimulation parameters by external programmer.14
Novel Applications of DBS
The advantageous safety profile of DBS has permitted its evaluation in the treatment of other conditions thought to have malfunctioning networks at their core—such as intractable epilepsy (in resective surgery noncandidates).15,16 Although several trials have shown promising results of using DBS for treatment-resistant depression,17 the results of pivotal sham-controlled trials have been mixed.18,19 Obsessive-compulsive disorder, on the other hand, received the FDA humanitarian device exemption designation on the basis of positive long-term results.20 In treatment-resistant depression and obsessive-compulsive disorder, functional neuroimaging has identified DBS targets.21,22 Functional MRI or positron emission tomography (PET) images can be compared at resting state, at symptomatic state, and after treatment response. Nodes hyperactive during a symptomatic state and less active after successful treatment can be targeted with high-frequency DBS to directly reduce the hyperactivity and indirectly modulate or normalize the overall function of the circuit.23
Given the functional MRI and O15 (oxygen-15) PET evidence of amygdala hyperactivity in patients with PTSD having core symptoms,24-26 the authors hypothesized that high-frequency DBS targeting of the amygdala would improve PTSD-associated hyperarousal and reexperiencing symptoms in treatment-refractory patients. Indirect data supporting this hypothesis include a correlation between amygdala hyperactivity of increased intensity and symptom severity measured with the Clinician-Administered PTSD Scale (CAPS),27 and a correlation between reduced pretreatment amygdala hyperactivity and successful cognitive-behavioral treatment.28,29
Preclinical Work
Using a rodent model in which a novel object serves as a cue reminder of foot shocks (traumatic event), the authors tested the hypothesis that amygdala DBS would reduce PTSD-like symptoms.30 When untreated rats were presented with the object in their cage a week after the initial exposure, they immediately buried the object under bedding to avoid being reminded of the shocks. In contrast, rats treated with DBS did not bury the object. In most cases, in fact, they played with it.
The authors also replicated their results but with the addition of rats treated with paroxetine.31 Using the same rodent model, they found DBS superior to paroxetine in treating PTSD-like symptoms. This study had a crossover design: DBS and sham DBS. Briefly, 20 rats received an electrode in the amygdala and were exposed to inescapable shocks in the presence of the cue object. The rats were then randomly assigned to a DBS group (10 rats) or a sham-DBS group (10 rats). After 1 week, behavioral testing showed fear extinction in the DBS group and no improvement in the sham-DBS group. Then the groups were switched: The rats originally treated with DBS received no treatment, and the rats that were originally sham-treated underwent DBS. One week later, behavioral testing showed acquisition of fear extinction in all the rats. These results suggested DBS can be effective even when delayed after establishment of fear persistence and PTSD symptoms. These results also showed that DBS effects persist even after therapy discontinuation.
Similarly, other investigators have reported that the role of the amygdala is not limited to fear acquisition; it extends to fear expression. A lesion in the amygdala can prevent fear expression even if the disruption is performed subsequent to fear-conditioning training.32 This finding is important for humans, as DBS would be initiated during the chronic phase of the disorder, after failure of less invasive treatment options, such as pharmacotherapy and psychotherapy.
Early Clinical Experience
The authors have initiated the first ever clinical trial (NCT02091843) evaluating use of DBS for PTSD and are now recruiting patients. Enrollment is limited to 6 combat veterans with disabling PTSD that has not responded to pharmacotherapy and psychotherapy. This VA-funded single-site study, being conducted at the VA Greater Los Angeles Healthcare System (VAGLAHS), was approved by the VAGLAHS Institutional Review Board and the FDA. The authors have published the 2-year trial’s protocol, which includes an active-versus-sham stimulation phase; continuous electroencephalogram monitoring; baseline and posttreatment 18FDG (fluorodeoxyglucose) PET performed during a resting state vs during investigator-guided exposure to trauma reminders; and extensive psychological and neuropsychological assessments.33 The literature includes only 1 case report on amygdala DBS.34 The authors of that report used DBS of the basolateral nucleus of the amygdala to treat a teenaged boy with severe autism and found that the therapy was safe.
As of this writing, the authors have recruited and implanted 1 patient and reported on his clinical results (including baseline PET) over the first 8 months of stimulation35 and on the electrophysiologic findings over the first year.36 After experiencing extremely severe combat PTSD refractory to pharmacotherapy and psychotherapy treatments for more than 20 years, the patient treated with DBS is now experiencing substantial symptom relief, and his CAPS score (primary outcome measure) has improved by about 40%. He has tolerated continuous stimulation without any serious DBS-related AEs for up to 16 months. Notably, he has not had a single severe combat nightmare in a year—in stark contrast to nightly combat nightmares during the 20-year period leading to the trial. Furthermore, he has not been having any episodes of severe dissociation, which had been a common disabling problem before the trial. He has taken a second trip out of the country, improved his relationships with family, and made strides (albeit limited) in pursuing additional social interactions.
Avoidance remains a major problem. He recently left his job after 7 years, because he prefers a more nature-oriented rather than people-oriented environment. In addition, his interest in intensive psychotherapy has increased, and he has been considering options for spending more time working on his therapy.
Over 15 months of treatment, the patient’s CAPS total and subscale scores have decreased—his symptoms have improved (Table).21 He has had rapid and substantial reductions in recurrence and hyperarousal symptoms but slower improvement in avoidance. Improvements in emotional reactivity would be expected to occur before any change in behavior (eg, avoidance). Patients likely must first recognize changes in emotional reactivity to events before they can engage in a cognitive process to modify learned behavioral responses to those events.
After about 9 months of treatment, all of the study patient’s symptoms were somewhat stabilized, and the authors began making gradual stimulation adjustments to the latest parameters—3.5 V, 60 µs, and 160 Hz for the right electrode and 1.5 V, 60 µs, and 160 Hz for the left electrode—using the contacts in the basolateral nucleus of the amygdala, per postoperative neuroimaging.3
After 15 to 18 months, when improvement peaked at 48% symptom reduction from baseline, the patient experienced psychiatric decompensation (depression, suicide gesture) not attributable to changes in stimulation settings and not associated with exacerbation of PTSD symptoms. Treatment team members and independent psychiatric consultants attributed the decompensation to the patient’s difficulty in changing a long-standing avoidant behavior routine, owing to severe recurrence and hyperarousal symptoms in the past. His persistent inability to overcome avoidance and isolation, despite core PTSD symptom improvement, had left him feeling worthless.
The patient remains in the study but also is participating in other medication and psychotherapy trials and is making a career change. Periodic decompensations may be part of the treatment course as patients reach a more complex and volatile phase of improvement that requires more intensive cognitive reprocessing. If this proves to be the case with other patients enrolling in the study, intensive psychotherapy that addresses cognitive and emotional PTSD symptoms may be needed once there is improvement in intrusive and hyperarousal symptoms.
Conclusion
Deep brain stimulation has been successful in treating Parkinson disease and essential tremor. Physiologically, DBS seems to inhibit specific brain regions’ dysfunctional activity stemming from a disease process. Deep brain stimulation-induced inhibition of a dysfunctional node improves clinical outcomes in movement disorders.
Given the reversibility and positive safety profile of DBS, new applications are being studied. The authors propose that DBS may benefit patients with severe treatment-refractory PTSD. Their first patient’s core PTSD symptoms have improved significantly, as expected, but as in other psychiatric DBS cases, the seriousness and chronicity of his illness may be complicating the course of recovery. The authors plan to recruit 6 patients for this early-phase safety trial.
Click here to read the digital edition.
Failure of fear extinction is a core feature of posttraumatic stress disorder (PTSD).1 Recently, it was confirmed that the amygdala and the orbitofrontal cortex are crucial for both fear acquisition and fear extinction.2 The amygdala was found to have neurons active only during fear acquisition, and other neurons active only during fear extinction.3 In essence, the balance of activity between these 2 neuronal populations determines whether if an incoming stimulus is feared or not feared. This balance is under the influence of several cognitive domains, including memory, reward, and executive function.
In PTSD, the equilibrium is shifted heavily toward fear acquisition. The majority of patients spontaneously regain the capacity for fear extinction over time4 or with the help of treatment.5,6 Nonetheless, some patients with severe PTSD seem unable to recover the ability of fear extinction and remain refractory to both standard and novel psychotherapeutic or psychopharmacologic treatments.7 For these patients, direct modulation of the neural activity in the amygdala may permit fear extinction. This article describes the rationale for using deep brain stimulation (DBS) and initial results from the first-ever clinical trial.
Deep Brain Stimulation
Deep brain stimulation involves inserting electrodes in precise cerebral targets and then connecting the leads to a pulse generator (similar to a pacemaker) inserted in a subclavicular pocket. The generator controls the electrical signal (amplitude, pulse width, pulse frequency) delivered to the brain target and can be adjusted with use of a noninvasive programmer. In 1997, the FDA approved DBS for patients with Parkinson disease or essential tremor. Since then, its efficacy in these movement disorders has been confirmed in several studies.8,9
The mechanism by which the small electrical pulses of DBS influence activity is not clear. Clinically, DBS functionally inhibits the activity of local neurons.10 One theory describes “frequency jamming,” a concept similar to cardiac overdrive pacing in which the resultant high-frequency neuronal signal is meaningless and discounted by the rest of the brain.11
Over the years, DBS has demonstrated a strong safety profile.12 The risks of electrode insertion are mitigated with targeting based on high-quality magnetic resonance imaging (MRI) and computed tomography (Figure). Unlike a destructive lesion, DBS is reversible, and the implanted system can be removed in its entirety. Histologic analyses have shown only a small amount of scarring around the electrode tip.13 In movement disorder treatment, clinical experience has shown that stimulation-related adverse effects (AEs) are reversible with readjustment of stimulation parameters by external programmer.14
Novel Applications of DBS
The advantageous safety profile of DBS has permitted its evaluation in the treatment of other conditions thought to have malfunctioning networks at their core—such as intractable epilepsy (in resective surgery noncandidates).15,16 Although several trials have shown promising results of using DBS for treatment-resistant depression,17 the results of pivotal sham-controlled trials have been mixed.18,19 Obsessive-compulsive disorder, on the other hand, received the FDA humanitarian device exemption designation on the basis of positive long-term results.20 In treatment-resistant depression and obsessive-compulsive disorder, functional neuroimaging has identified DBS targets.21,22 Functional MRI or positron emission tomography (PET) images can be compared at resting state, at symptomatic state, and after treatment response. Nodes hyperactive during a symptomatic state and less active after successful treatment can be targeted with high-frequency DBS to directly reduce the hyperactivity and indirectly modulate or normalize the overall function of the circuit.23
Given the functional MRI and O15 (oxygen-15) PET evidence of amygdala hyperactivity in patients with PTSD having core symptoms,24-26 the authors hypothesized that high-frequency DBS targeting of the amygdala would improve PTSD-associated hyperarousal and reexperiencing symptoms in treatment-refractory patients. Indirect data supporting this hypothesis include a correlation between amygdala hyperactivity of increased intensity and symptom severity measured with the Clinician-Administered PTSD Scale (CAPS),27 and a correlation between reduced pretreatment amygdala hyperactivity and successful cognitive-behavioral treatment.28,29
Preclinical Work
Using a rodent model in which a novel object serves as a cue reminder of foot shocks (traumatic event), the authors tested the hypothesis that amygdala DBS would reduce PTSD-like symptoms.30 When untreated rats were presented with the object in their cage a week after the initial exposure, they immediately buried the object under bedding to avoid being reminded of the shocks. In contrast, rats treated with DBS did not bury the object. In most cases, in fact, they played with it.
The authors also replicated their results but with the addition of rats treated with paroxetine.31 Using the same rodent model, they found DBS superior to paroxetine in treating PTSD-like symptoms. This study had a crossover design: DBS and sham DBS. Briefly, 20 rats received an electrode in the amygdala and were exposed to inescapable shocks in the presence of the cue object. The rats were then randomly assigned to a DBS group (10 rats) or a sham-DBS group (10 rats). After 1 week, behavioral testing showed fear extinction in the DBS group and no improvement in the sham-DBS group. Then the groups were switched: The rats originally treated with DBS received no treatment, and the rats that were originally sham-treated underwent DBS. One week later, behavioral testing showed acquisition of fear extinction in all the rats. These results suggested DBS can be effective even when delayed after establishment of fear persistence and PTSD symptoms. These results also showed that DBS effects persist even after therapy discontinuation.
Similarly, other investigators have reported that the role of the amygdala is not limited to fear acquisition; it extends to fear expression. A lesion in the amygdala can prevent fear expression even if the disruption is performed subsequent to fear-conditioning training.32 This finding is important for humans, as DBS would be initiated during the chronic phase of the disorder, after failure of less invasive treatment options, such as pharmacotherapy and psychotherapy.
Early Clinical Experience
The authors have initiated the first ever clinical trial (NCT02091843) evaluating use of DBS for PTSD and are now recruiting patients. Enrollment is limited to 6 combat veterans with disabling PTSD that has not responded to pharmacotherapy and psychotherapy. This VA-funded single-site study, being conducted at the VA Greater Los Angeles Healthcare System (VAGLAHS), was approved by the VAGLAHS Institutional Review Board and the FDA. The authors have published the 2-year trial’s protocol, which includes an active-versus-sham stimulation phase; continuous electroencephalogram monitoring; baseline and posttreatment 18FDG (fluorodeoxyglucose) PET performed during a resting state vs during investigator-guided exposure to trauma reminders; and extensive psychological and neuropsychological assessments.33 The literature includes only 1 case report on amygdala DBS.34 The authors of that report used DBS of the basolateral nucleus of the amygdala to treat a teenaged boy with severe autism and found that the therapy was safe.
As of this writing, the authors have recruited and implanted 1 patient and reported on his clinical results (including baseline PET) over the first 8 months of stimulation35 and on the electrophysiologic findings over the first year.36 After experiencing extremely severe combat PTSD refractory to pharmacotherapy and psychotherapy treatments for more than 20 years, the patient treated with DBS is now experiencing substantial symptom relief, and his CAPS score (primary outcome measure) has improved by about 40%. He has tolerated continuous stimulation without any serious DBS-related AEs for up to 16 months. Notably, he has not had a single severe combat nightmare in a year—in stark contrast to nightly combat nightmares during the 20-year period leading to the trial. Furthermore, he has not been having any episodes of severe dissociation, which had been a common disabling problem before the trial. He has taken a second trip out of the country, improved his relationships with family, and made strides (albeit limited) in pursuing additional social interactions.
Avoidance remains a major problem. He recently left his job after 7 years, because he prefers a more nature-oriented rather than people-oriented environment. In addition, his interest in intensive psychotherapy has increased, and he has been considering options for spending more time working on his therapy.
Over 15 months of treatment, the patient’s CAPS total and subscale scores have decreased—his symptoms have improved (Table).21 He has had rapid and substantial reductions in recurrence and hyperarousal symptoms but slower improvement in avoidance. Improvements in emotional reactivity would be expected to occur before any change in behavior (eg, avoidance). Patients likely must first recognize changes in emotional reactivity to events before they can engage in a cognitive process to modify learned behavioral responses to those events.
After about 9 months of treatment, all of the study patient’s symptoms were somewhat stabilized, and the authors began making gradual stimulation adjustments to the latest parameters—3.5 V, 60 µs, and 160 Hz for the right electrode and 1.5 V, 60 µs, and 160 Hz for the left electrode—using the contacts in the basolateral nucleus of the amygdala, per postoperative neuroimaging.3
After 15 to 18 months, when improvement peaked at 48% symptom reduction from baseline, the patient experienced psychiatric decompensation (depression, suicide gesture) not attributable to changes in stimulation settings and not associated with exacerbation of PTSD symptoms. Treatment team members and independent psychiatric consultants attributed the decompensation to the patient’s difficulty in changing a long-standing avoidant behavior routine, owing to severe recurrence and hyperarousal symptoms in the past. His persistent inability to overcome avoidance and isolation, despite core PTSD symptom improvement, had left him feeling worthless.
The patient remains in the study but also is participating in other medication and psychotherapy trials and is making a career change. Periodic decompensations may be part of the treatment course as patients reach a more complex and volatile phase of improvement that requires more intensive cognitive reprocessing. If this proves to be the case with other patients enrolling in the study, intensive psychotherapy that addresses cognitive and emotional PTSD symptoms may be needed once there is improvement in intrusive and hyperarousal symptoms.
Conclusion
Deep brain stimulation has been successful in treating Parkinson disease and essential tremor. Physiologically, DBS seems to inhibit specific brain regions’ dysfunctional activity stemming from a disease process. Deep brain stimulation-induced inhibition of a dysfunctional node improves clinical outcomes in movement disorders.
Given the reversibility and positive safety profile of DBS, new applications are being studied. The authors propose that DBS may benefit patients with severe treatment-refractory PTSD. Their first patient’s core PTSD symptoms have improved significantly, as expected, but as in other psychiatric DBS cases, the seriousness and chronicity of his illness may be complicating the course of recovery. The authors plan to recruit 6 patients for this early-phase safety trial.
Click here to read the digital edition.
1. Milad MR, Pitman RK, Ellis CB, et al. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry. 2009;66(12):1075-1082.
2. Marin MF, Song H, VanElzakker MB, et al. Association of resting metabolism in the fear neural network with extinction recall activations and clinical measures in trauma-exposed individuals. Am J Psychiatry. 2016;173(9):930-938.
3. Herry C, Ciocchi S, Senn V, Demmou L, Müller C, Lüthi A. Switching on and off fear by distinct neuronal circuits. Nature. 2008;454(7204):600-606.
4. Morina N, Wicherts JM, Lobbrecht J, Priebe S. Remission from post-traumatic stress disorder in adults: a systematic review and meta-analysis of long term outcome studies. Clin Psychol Rev. 2014;34(3):249-255.
5. Steenkamp MM, Litz BT, Hoge CW, Marmar CR. Psychotherapy for military-related PTSD: a review of randomized clinical trials. JAMA. 2015;314(5):489-500.
6. Hoskins M, Pearce J, Bethell A, et al. Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis. Br J Psychiatry. 2015;206(2):93-100.
7. Koek RJ, Schwartz HN, Scully S, et al. Treatment-refractory posttraumatic stress disorder (TRPTSD): a review and framework for the future. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:170-218.
8. Wagle Shukla A, Okun MS. State of the art for deep brain stimulation therapy in movement disorders: a clinical and technological perspective. IEEE Rev Biomed Eng. 2016;9:219-233.
9. Weaver FM, Follett K, Stern M, et al; CSP 468 Study Group. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. JAMA. 2009;301(1):63-73.
10. Benabid AL, Benazzouz A, Hoffmann D, Limousin P, Krack P, Pollack P. Long-term electrical inhibition of deep brain targets in movement disorders. Mov Disord. 1998;13(suppl 3):119-125.
11. Benabid AL, Wallace B, Mitrofanis J, et al. A putative generalized model of the effects and mechanism of action of high frequency electrical stimulation of the central nervous system. Acta Neurol Belg. 2005;105(3):149-157.
12. Fenoy AJ, Simpson RK Jr. Risks of common complications in deep brain stimulation surgery: management and avoidance. J Neurosurg. 2014;120(1):132-139.
13. DiLorenzo DJ, Jankovic J, Simpson RK, Takei H, Powell SZ. Neurohistopathological findings at the electrode–tissue interface in long-term deep brain stimulation: systematic literature review, case report, and assessment of stimulation threshold safety. Neuromodulation. 2014;17(5):405-418.
14. Revell MA. Deep brain stimulation for movement disorders. Nurs Clin North Am. 2015;50(4):691-701.
15. Fisher R, Salanova V, Witt T, et al; SANTE Study Group. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy. Epilepsia. 2010;51(5):899-908.
16. Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
17. Berlim MT, McGirr A, Van den Eynde F, Fleck MP, Giacobbe P. Effectiveness and acceptability of deep brain stimulation (DBS) of the subgenual cingulate cortex for treatment-resistant depression: a systematic review and exploratory meta-analysis. J Affect Disord. 2014;159:31-38.
18. Dougherty DD, Rezai AR, Carpenter LL, et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol Psychiatry. 2015;78(4):240-248.
19. Bergfeld IO, Mantione M, Hoogendoorn ML, et al. Deep brain stimulation of the ventral anterior limb of the internal capsule for treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2016;73(5):456-464.
20. Greenberg BD, Malone DA, Friehs GM, et al. Three-year outcomes in deep brain stimulation for highly resistant obsessive-compulsive disorder. Neuropsychopharmacology. 2006;31(11):2384-2393.
21. Mayber HS, Liotti M, Brannan SK, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156(5):675-682.
22. Rauch SL, Jenike MA, Alpert NM, et al. Regional cerebral blood flow measured during symptom provocation in obsessive-compulsive disorder using oxygen 15-labeled carbon dioxide and positron emission tomography. Arch Gen Psychiatry. 1994;51(1):62-70.
23. Williams NR, Taylor JJ, Lamb K, Hanlon CA, Short EB, George MS. Role of functional imaging in the development and refinement of invasive neuromodulation for psychiatric disorders. World J Radiol. 2014;6(10):756-778.
24. Francati V, Vermetten E, Bremner JD. Functional neuroimaging studies in posttraumatic stress disorder: review of current methods and findings. Depress Anxiety. 2007;24(3):202-218.
25. Shin LM, Orr SP, Carson MA, et al. Regional cerebral blood flow in the amygdala and medial prefrontal cortex during traumatic imagery in male and female Vietnam veterans with PTSD. Arch Gen Psychiatry. 2004;61(2):168-176.
26. Armony JL, Corbo V, Clément MH, Brunet A. Amygdala response in patients with acute PTSD to masked and unmasked emotional facial expressions. Am J Psychiatry. 2005;162(10):1961-1963.
27. Blake DD, Weathers FW, Nagy LM, et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress. 1995;8(1):75-90.
28. Felmingham K, Kemp A, Williams L, et al. Changes in anterior cingulate and amygdala after cognitive behavior therapy of posttraumatic stress disorder. Psychol Sci. 2007;18(2):127-129.
29. Peres JF, Newberg AB, Mercante JP, et al. Cerebral blood flow changes during retrieval of traumatic memories before and after psychotherapy: a SPECT study. Psychol Med. 2007;37(10):1481-1491.
30. Langevin JP, De Salles AA, Kosoyan HP, Krahl SE. Deep brain stimulation of the amygdala alleviates post-traumatic stress disorder symptoms in a rat model. J Psychiatr Res. 2010;44(16):1241-1245.
31. Stidd DA, Vogelsang K, Krahl SE, Langevin JP, Fellous JM. Amygdala deep brain stimulation is superior to paroxetine treatment in a rat model of posttraumatic stress disorder. Brain Stimul. 2013;6(6):837-844.
32. Anglada-Figueroa D, Quirk GJ. Lesions of the basal amygdala block expression of conditioned fear but not extinction. J Neurosci. 2005;25(42):9680-9685.
33. Koek RJ, Langevin JP, Krahl SE, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory combat post-traumatic stress disorder (PTSD): study protocol for a pilot randomized controlled trial with blinded, staggered onset of stimulation. Trials. 2014;15:356.
34. Sturm V, Fricke O, Bührle CP, et al. DBS in the basolateral amygdala improves symptoms of autism and related self-injurious behavior: a case report and hypothesis on the pathogenesis of the disorder. Front Hum Neurosci. 2013;6:341.
35. Langevin JP, Koek RJ, Schwartz HN, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory posttraumatic stress disorder. Biol Psychiatry. 2016;79(10):e82-e84.
36. Langevin JP, Chen JW, Koek RJ, et al. Deep brain stimulation of the basolateral amygdala: targeting technique and electrodiagnostic findings. Brain Sci. 2016;6(3):E28.
1. Milad MR, Pitman RK, Ellis CB, et al. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry. 2009;66(12):1075-1082.
2. Marin MF, Song H, VanElzakker MB, et al. Association of resting metabolism in the fear neural network with extinction recall activations and clinical measures in trauma-exposed individuals. Am J Psychiatry. 2016;173(9):930-938.
3. Herry C, Ciocchi S, Senn V, Demmou L, Müller C, Lüthi A. Switching on and off fear by distinct neuronal circuits. Nature. 2008;454(7204):600-606.
4. Morina N, Wicherts JM, Lobbrecht J, Priebe S. Remission from post-traumatic stress disorder in adults: a systematic review and meta-analysis of long term outcome studies. Clin Psychol Rev. 2014;34(3):249-255.
5. Steenkamp MM, Litz BT, Hoge CW, Marmar CR. Psychotherapy for military-related PTSD: a review of randomized clinical trials. JAMA. 2015;314(5):489-500.
6. Hoskins M, Pearce J, Bethell A, et al. Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis. Br J Psychiatry. 2015;206(2):93-100.
7. Koek RJ, Schwartz HN, Scully S, et al. Treatment-refractory posttraumatic stress disorder (TRPTSD): a review and framework for the future. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:170-218.
8. Wagle Shukla A, Okun MS. State of the art for deep brain stimulation therapy in movement disorders: a clinical and technological perspective. IEEE Rev Biomed Eng. 2016;9:219-233.
9. Weaver FM, Follett K, Stern M, et al; CSP 468 Study Group. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. JAMA. 2009;301(1):63-73.
10. Benabid AL, Benazzouz A, Hoffmann D, Limousin P, Krack P, Pollack P. Long-term electrical inhibition of deep brain targets in movement disorders. Mov Disord. 1998;13(suppl 3):119-125.
11. Benabid AL, Wallace B, Mitrofanis J, et al. A putative generalized model of the effects and mechanism of action of high frequency electrical stimulation of the central nervous system. Acta Neurol Belg. 2005;105(3):149-157.
12. Fenoy AJ, Simpson RK Jr. Risks of common complications in deep brain stimulation surgery: management and avoidance. J Neurosurg. 2014;120(1):132-139.
13. DiLorenzo DJ, Jankovic J, Simpson RK, Takei H, Powell SZ. Neurohistopathological findings at the electrode–tissue interface in long-term deep brain stimulation: systematic literature review, case report, and assessment of stimulation threshold safety. Neuromodulation. 2014;17(5):405-418.
14. Revell MA. Deep brain stimulation for movement disorders. Nurs Clin North Am. 2015;50(4):691-701.
15. Fisher R, Salanova V, Witt T, et al; SANTE Study Group. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy. Epilepsia. 2010;51(5):899-908.
16. Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
17. Berlim MT, McGirr A, Van den Eynde F, Fleck MP, Giacobbe P. Effectiveness and acceptability of deep brain stimulation (DBS) of the subgenual cingulate cortex for treatment-resistant depression: a systematic review and exploratory meta-analysis. J Affect Disord. 2014;159:31-38.
18. Dougherty DD, Rezai AR, Carpenter LL, et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol Psychiatry. 2015;78(4):240-248.
19. Bergfeld IO, Mantione M, Hoogendoorn ML, et al. Deep brain stimulation of the ventral anterior limb of the internal capsule for treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2016;73(5):456-464.
20. Greenberg BD, Malone DA, Friehs GM, et al. Three-year outcomes in deep brain stimulation for highly resistant obsessive-compulsive disorder. Neuropsychopharmacology. 2006;31(11):2384-2393.
21. Mayber HS, Liotti M, Brannan SK, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156(5):675-682.
22. Rauch SL, Jenike MA, Alpert NM, et al. Regional cerebral blood flow measured during symptom provocation in obsessive-compulsive disorder using oxygen 15-labeled carbon dioxide and positron emission tomography. Arch Gen Psychiatry. 1994;51(1):62-70.
23. Williams NR, Taylor JJ, Lamb K, Hanlon CA, Short EB, George MS. Role of functional imaging in the development and refinement of invasive neuromodulation for psychiatric disorders. World J Radiol. 2014;6(10):756-778.
24. Francati V, Vermetten E, Bremner JD. Functional neuroimaging studies in posttraumatic stress disorder: review of current methods and findings. Depress Anxiety. 2007;24(3):202-218.
25. Shin LM, Orr SP, Carson MA, et al. Regional cerebral blood flow in the amygdala and medial prefrontal cortex during traumatic imagery in male and female Vietnam veterans with PTSD. Arch Gen Psychiatry. 2004;61(2):168-176.
26. Armony JL, Corbo V, Clément MH, Brunet A. Amygdala response in patients with acute PTSD to masked and unmasked emotional facial expressions. Am J Psychiatry. 2005;162(10):1961-1963.
27. Blake DD, Weathers FW, Nagy LM, et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress. 1995;8(1):75-90.
28. Felmingham K, Kemp A, Williams L, et al. Changes in anterior cingulate and amygdala after cognitive behavior therapy of posttraumatic stress disorder. Psychol Sci. 2007;18(2):127-129.
29. Peres JF, Newberg AB, Mercante JP, et al. Cerebral blood flow changes during retrieval of traumatic memories before and after psychotherapy: a SPECT study. Psychol Med. 2007;37(10):1481-1491.
30. Langevin JP, De Salles AA, Kosoyan HP, Krahl SE. Deep brain stimulation of the amygdala alleviates post-traumatic stress disorder symptoms in a rat model. J Psychiatr Res. 2010;44(16):1241-1245.
31. Stidd DA, Vogelsang K, Krahl SE, Langevin JP, Fellous JM. Amygdala deep brain stimulation is superior to paroxetine treatment in a rat model of posttraumatic stress disorder. Brain Stimul. 2013;6(6):837-844.
32. Anglada-Figueroa D, Quirk GJ. Lesions of the basal amygdala block expression of conditioned fear but not extinction. J Neurosci. 2005;25(42):9680-9685.
33. Koek RJ, Langevin JP, Krahl SE, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory combat post-traumatic stress disorder (PTSD): study protocol for a pilot randomized controlled trial with blinded, staggered onset of stimulation. Trials. 2014;15:356.
34. Sturm V, Fricke O, Bührle CP, et al. DBS in the basolateral amygdala improves symptoms of autism and related self-injurious behavior: a case report and hypothesis on the pathogenesis of the disorder. Front Hum Neurosci. 2013;6:341.
35. Langevin JP, Koek RJ, Schwartz HN, et al. Deep brain stimulation of the basolateral amygdala for treatment-refractory posttraumatic stress disorder. Biol Psychiatry. 2016;79(10):e82-e84.
36. Langevin JP, Chen JW, Koek RJ, et al. Deep brain stimulation of the basolateral amygdala: targeting technique and electrodiagnostic findings. Brain Sci. 2016;6(3):E28.
VA is Trying New Ways to Treat TBI and PTSD
The VA is examining two alternative treatments for TBI and PTSD: a light-emitting diode (LED) treatment for mild to moderate TBI and stellate ganglion block (SGB) for PTSD.
In the LED treatment, which takes about 30 minutes, a lightweight LED-lined helmet is placed on the patient’s head, and more diodes are placed inside the nose to deliver photons to the deeper parts of the brain. The light is painless and generates no heat.
Although it is considered investigational, LED therapy is available at the VAHS Boston, as well as for veterans to use at home. Dr. Margaret Naeser, a professor of neurology at Boston University School of Medicine and lead investigator of the Boston study team, interviewed in VA Research Currents, says the technology has been around a while, but it was previously used on the body for wound healing and pain. Using it on the brain is new. The LED light has been shown to boost the output of nitric oxide, improving blood flow. Studies have shown that LED improves brain function, including attention and memory, emotions, and sleep.
Naeser says most of the TBI and PTSD cases helped so far with LEDs on the head included cognitive rehabilitation therapy. The patients showed additional progress after the LED treatments. A combination of both treatments would likely produce the best results, she says.
Providers at the Long Beach VAMC have been using SGB, commonly used in pain management with ropivacaine or bupivacaine, to reduce the symptoms of PTSD. According to the VA Evidence-based Synthesis Program (ESP), SGB may ease anxiety and the alert response by inhibiting connections between the peripheral sympathetic nerve system and regions of the cerebral cortex, such as the amygdala, thought to be abnormally activated in PTSD. Stellate ganglion block also has been associated with biologic markers of sedation.
The ESP experts say there is insufficient information to determine which veterans are most likely to benefit from SGB for PTSD, but an uncontrolled, unblinded case series of 30 active duty service members with combat-related PTSD suggests that those with predominantly hyperarousal and avoidance symptoms might be the best candidates. Patients who have undergone SGB have found it highly acceptable, although the invasive nature of SGB may be a barrier for some. Findings from the first randomized controlled trial of SGB for PTSD were inconclusive, the panel said; further research is warranted.
The VA is examining two alternative treatments for TBI and PTSD: a light-emitting diode (LED) treatment for mild to moderate TBI and stellate ganglion block (SGB) for PTSD.
In the LED treatment, which takes about 30 minutes, a lightweight LED-lined helmet is placed on the patient’s head, and more diodes are placed inside the nose to deliver photons to the deeper parts of the brain. The light is painless and generates no heat.
Although it is considered investigational, LED therapy is available at the VAHS Boston, as well as for veterans to use at home. Dr. Margaret Naeser, a professor of neurology at Boston University School of Medicine and lead investigator of the Boston study team, interviewed in VA Research Currents, says the technology has been around a while, but it was previously used on the body for wound healing and pain. Using it on the brain is new. The LED light has been shown to boost the output of nitric oxide, improving blood flow. Studies have shown that LED improves brain function, including attention and memory, emotions, and sleep.
Naeser says most of the TBI and PTSD cases helped so far with LEDs on the head included cognitive rehabilitation therapy. The patients showed additional progress after the LED treatments. A combination of both treatments would likely produce the best results, she says.
Providers at the Long Beach VAMC have been using SGB, commonly used in pain management with ropivacaine or bupivacaine, to reduce the symptoms of PTSD. According to the VA Evidence-based Synthesis Program (ESP), SGB may ease anxiety and the alert response by inhibiting connections between the peripheral sympathetic nerve system and regions of the cerebral cortex, such as the amygdala, thought to be abnormally activated in PTSD. Stellate ganglion block also has been associated with biologic markers of sedation.
The ESP experts say there is insufficient information to determine which veterans are most likely to benefit from SGB for PTSD, but an uncontrolled, unblinded case series of 30 active duty service members with combat-related PTSD suggests that those with predominantly hyperarousal and avoidance symptoms might be the best candidates. Patients who have undergone SGB have found it highly acceptable, although the invasive nature of SGB may be a barrier for some. Findings from the first randomized controlled trial of SGB for PTSD were inconclusive, the panel said; further research is warranted.
The VA is examining two alternative treatments for TBI and PTSD: a light-emitting diode (LED) treatment for mild to moderate TBI and stellate ganglion block (SGB) for PTSD.
In the LED treatment, which takes about 30 minutes, a lightweight LED-lined helmet is placed on the patient’s head, and more diodes are placed inside the nose to deliver photons to the deeper parts of the brain. The light is painless and generates no heat.
Although it is considered investigational, LED therapy is available at the VAHS Boston, as well as for veterans to use at home. Dr. Margaret Naeser, a professor of neurology at Boston University School of Medicine and lead investigator of the Boston study team, interviewed in VA Research Currents, says the technology has been around a while, but it was previously used on the body for wound healing and pain. Using it on the brain is new. The LED light has been shown to boost the output of nitric oxide, improving blood flow. Studies have shown that LED improves brain function, including attention and memory, emotions, and sleep.
Naeser says most of the TBI and PTSD cases helped so far with LEDs on the head included cognitive rehabilitation therapy. The patients showed additional progress after the LED treatments. A combination of both treatments would likely produce the best results, she says.
Providers at the Long Beach VAMC have been using SGB, commonly used in pain management with ropivacaine or bupivacaine, to reduce the symptoms of PTSD. According to the VA Evidence-based Synthesis Program (ESP), SGB may ease anxiety and the alert response by inhibiting connections between the peripheral sympathetic nerve system and regions of the cerebral cortex, such as the amygdala, thought to be abnormally activated in PTSD. Stellate ganglion block also has been associated with biologic markers of sedation.
The ESP experts say there is insufficient information to determine which veterans are most likely to benefit from SGB for PTSD, but an uncontrolled, unblinded case series of 30 active duty service members with combat-related PTSD suggests that those with predominantly hyperarousal and avoidance symptoms might be the best candidates. Patients who have undergone SGB have found it highly acceptable, although the invasive nature of SGB may be a barrier for some. Findings from the first randomized controlled trial of SGB for PTSD were inconclusive, the panel said; further research is warranted.
Therapeutic horseback riding may lower veterans’ PTSD symptoms
Veterans with posttraumatic stress disorder might benefit from therapeutic horseback riding, a small study suggests.
“Our findings provide empirical evidence that [therapeutic horseback riding] is effective at improving coping skills and in lessening one’s difficulty with emotional regulation, especially with longer riding interventions,” wrote Rebecca A. Johnson, PhD, of the University of Missouri, Columbia, and her associates.
Overall, 57 veterans were recruited and 28 enrolled in the randomized trial at baseline. Those individuals were randomized into two groups: a wait-list control group and a treatment group. Eventually, all of the veterans participated in the therapeutic riding program. Meanwhile, the riding center staff were not aware of which veterans had been assigned to either group. The Professional Association of Therapeutic Horsemanship, a nonprofit group that promotes equine-related activities for people with special needs, selected the horses that were used in the study. During the data collection periods, PTSD symptoms were measured via the PTSD Checklist–Military Version, or PCL-M. This self-report measure asks patients about problems in response to “stressful military experiences,” the researchers wrote. The Coping Self-Efficacy Scale and the Difficulties in Emotion Regulation Scale were among the other instruments used.
While riding, the results showed, participants had a statistically significant decrease in PTSD symptoms over the course of the 6-week program. “,” Dr. Johnson and her associates wrote. “Further detailed examination showed that participants had a 66.7% likelihood of having lower PTSD scores at 3 weeks, and an 87.5% likelihood at 6 weeks.”
Anecdotally, some of the veterans wanted to continue therapeutic riding after the end of the program, and they were able to do so.
“We conclude that [therapeutic horseback riding] shows promise as a beneficial intervention for veterans with PTSD, but did not measure functional ability,” they wrote.
Veterans with posttraumatic stress disorder might benefit from therapeutic horseback riding, a small study suggests.
“Our findings provide empirical evidence that [therapeutic horseback riding] is effective at improving coping skills and in lessening one’s difficulty with emotional regulation, especially with longer riding interventions,” wrote Rebecca A. Johnson, PhD, of the University of Missouri, Columbia, and her associates.
Overall, 57 veterans were recruited and 28 enrolled in the randomized trial at baseline. Those individuals were randomized into two groups: a wait-list control group and a treatment group. Eventually, all of the veterans participated in the therapeutic riding program. Meanwhile, the riding center staff were not aware of which veterans had been assigned to either group. The Professional Association of Therapeutic Horsemanship, a nonprofit group that promotes equine-related activities for people with special needs, selected the horses that were used in the study. During the data collection periods, PTSD symptoms were measured via the PTSD Checklist–Military Version, or PCL-M. This self-report measure asks patients about problems in response to “stressful military experiences,” the researchers wrote. The Coping Self-Efficacy Scale and the Difficulties in Emotion Regulation Scale were among the other instruments used.
While riding, the results showed, participants had a statistically significant decrease in PTSD symptoms over the course of the 6-week program. “,” Dr. Johnson and her associates wrote. “Further detailed examination showed that participants had a 66.7% likelihood of having lower PTSD scores at 3 weeks, and an 87.5% likelihood at 6 weeks.”
Anecdotally, some of the veterans wanted to continue therapeutic riding after the end of the program, and they were able to do so.
“We conclude that [therapeutic horseback riding] shows promise as a beneficial intervention for veterans with PTSD, but did not measure functional ability,” they wrote.
Veterans with posttraumatic stress disorder might benefit from therapeutic horseback riding, a small study suggests.
“Our findings provide empirical evidence that [therapeutic horseback riding] is effective at improving coping skills and in lessening one’s difficulty with emotional regulation, especially with longer riding interventions,” wrote Rebecca A. Johnson, PhD, of the University of Missouri, Columbia, and her associates.
Overall, 57 veterans were recruited and 28 enrolled in the randomized trial at baseline. Those individuals were randomized into two groups: a wait-list control group and a treatment group. Eventually, all of the veterans participated in the therapeutic riding program. Meanwhile, the riding center staff were not aware of which veterans had been assigned to either group. The Professional Association of Therapeutic Horsemanship, a nonprofit group that promotes equine-related activities for people with special needs, selected the horses that were used in the study. During the data collection periods, PTSD symptoms were measured via the PTSD Checklist–Military Version, or PCL-M. This self-report measure asks patients about problems in response to “stressful military experiences,” the researchers wrote. The Coping Self-Efficacy Scale and the Difficulties in Emotion Regulation Scale were among the other instruments used.
While riding, the results showed, participants had a statistically significant decrease in PTSD symptoms over the course of the 6-week program. “,” Dr. Johnson and her associates wrote. “Further detailed examination showed that participants had a 66.7% likelihood of having lower PTSD scores at 3 weeks, and an 87.5% likelihood at 6 weeks.”
Anecdotally, some of the veterans wanted to continue therapeutic riding after the end of the program, and they were able to do so.
“We conclude that [therapeutic horseback riding] shows promise as a beneficial intervention for veterans with PTSD, but did not measure functional ability,” they wrote.
FROM MILITARY MEDICAL RESEARCH







