User login
Ketamine infusions may be helpful in central sensitization pain syndromes
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
EXPERT ANALYSIS FROM PAINWEEK 2018
What is the Relation Between PTSD and Medical Conditions?
Posttraumatic stress disorder (PTSD) develops after exposure to a traumatic event, which can involve witnessing the traumatic event or directly experiencing the trauma.1 The prevalence of PTSD in the general population is approximately 7% to 8%.1 However, not everyone who experiences trauma develops PTSD since the majority of men and women experience at least 1 traumatic event in their lifetimes but do not develop PTSD.1
In order to be diagnosed with PTSD, a patient must meet several criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5).2 The patient is required to have exposure to trauma, begin having a certain number of prespecified symptoms, and these symptoms must persist for at least a month.2 Symptoms of PTSD include re-experiencing the traumatic event, avoidance of stimuli associated with the trauma, negative cognitions and mood, and hyperarousal.3,4 The hyperarousal that is associated with PTSD has been theorized to be either a result of the trauma experienced or exacerbation of a pre-existing tendency.5 This can manifest in various ways, such as hypervigilance, exaggerated startle response, trouble sleeping, problems concentrating, or irritability.3,5 These symptoms can cause individuals with PTSD to have elevated levels of stress and to experience difficulties with completing everyday tasks.6
PTSD and Inflimmation-Related Medical Conditions
Posttraumatic stress disorder has been linked to various physical health problems. Studies have found that PTSD is often comorbid with cardiovascular, autoimmune, musculoskeletal, digestive, chronic pain and respiratory disorders.3,7-11 Inflammation may be a contributing factor in the associations between PTSD and these conditions.12-16 Studies have found that increases in pro-inflammatory cytokines and interferons are associated with PTSD, as well as changes in immune-related blood cells.12-16
Considering that PTSD has been linked to many medical conditions that have inflammatory components, especially cardiovascular disease, inflammatory markers may be early indicators of PTSD.12-16 Additionally, inflammatory markers such as cytokines and interferons can be targeted through medications, and potentially influence symptoms.13 However, the relation between PTSD and inflammation remains unclear. Associations between PTSD and inflammation-related medical conditions may be due to confounding variables, such as sociodemographic characteristics and health behaviors. Moreover, the list of inflammation-related medical conditions is long and there is no universal agreement of what conditions are related to inflammation.
We recently conducted an epidemiological study using a representative sample of residents living in New York City and found significant associations between PTSD and some inflammation-related medical conditions.8 We found that participants who had PTSD were more than 4 times more likely to report having had a heart attack or emphysema than were those without PTSD. In addition, participants with PTSD were 2 times more likely to report having hypercholesterolemia, insulin resistance, and angina than were those without PTSD. However, we also found that participants who had PTSD were less likely to develop other inflammation-related conditions like hypertension, type 1 diabetes mellitus, asthma, coronary heart disease, stroke, osteoporosis, and failing kidney.
Together, these associations suggest there is a strong link between PTSD and certain medical conditions, but the link may not be solely based on inflammation.8 Moreover, positive associations between PTSD and hypertension, asthma, and coronary heart disease disappeared when depression was controlled for. This finding points to depression as a major factor, consistent with previous findings that depression is associated with the development of various medical conditions and may be a stronger factor than PTSD.8
Nonetheless, findings concerning the increased risk for heart problems among adults with PTSD are striking and important given that heart disease is one of the main causes of death in the United States.9 Specifically, well over half a million people in the United States die of heart disease annually as the leading cause of death.17 Heart disease has been one of the top 2 leading causes of death for Americans since 1975.18
In the veteran population, heart disease has also been found to be a leading cause of death, accounting for 20 percent of all deaths in veterans from 1993 to 2002.19 Posttraumatic stress disorder has been linked to a 55% increase in the chance of developing heart disease or dying from a heart-related medical problem.9 For example, data from the World Trade Center Registry showed that on average adults who developed PTSD from the 9/11 terrorist attack had a heightened risk for heart disease for 3 years after the event.9 Other studies of the U.S. veteran population have shown that veterans with PTSD are more likely to experience heart failure, myocardial infarction, and cardiac arrhythmia than other veterans.10,20
Veteran-Specific Issues
In the US veteran population, there is a higher prevalence of PTSD and physical health conditions when compared with the general population.21,21 The prevalence of combat-related PTSD in veterans ranges from 2% to 17%, compared with a 7% to 8% prevalence of PTSD in the general population.1,22 In a study of veterans who were seen in patient-aligned care teams (PACTs) > 1 year, 9.3% were diagnosed with PTSD and many of those with PTSD also had other medical conditions.21 It was found that 43% of veterans seen by PACTs with chronic pain had PTSD, 33% with hypertension had PTSD, and 32% with diabetes mellitus had PTSD.21 In another study of combat veterans it was found that those who were trauma-exposed had more physical health problems, regardless of the amount of time spent in combat.19 Consequentially, veterans with PTSD have been found to make more frequent visits to primary care and specialty medical care clinics. 21
Integrated healthcare has been a main service model for the Department of Veteran Affairs (VA) and several programs have been created to integrate mental health and primary care. For example, the VA primary care-mental health integration (PCMHI) program places mental health services within primary care services.21 Assessments of this program have demonstrated that it improves the screening of psychological disorders and preventive care of patients who have psychological disorders.21 Specifically, it has been found that contact with PCMHI diminishes risks for poor outcomes among psychiatric patients.21 Another program called SCAN-ECHO, provides specialized training for VA general practitioners on treating specific health conditions through a specialty care team and video conferencing.23 This VA program allows for patients in more remote locations to receive specialty care from generalists.23 While there has not yet been a focus in SCAN-ECHO on PTSD, this may be considered in the future as a way to better train primary care and mental health providers about PTSD and common comorbid medical conditions.
Through their professional experiences, VA practitioners have knowledge of the link between PTSD and various medical conditions. The VA has already implemented screening for PTSD in primary care clinics, but it is important for mental health providers and medical practitioners to continue educating themselves about medical comorbidities and the possible exacerbation of medical conditions due to PTSD.21 Some physical manifestations of PTSD symptoms, such as sleep disturbances, avoidance of crowds, or hypervigilance, can affect overall health. Hypervigilance can result in over-activation of stress pathways, which puts patients with PTSD at a heighted risk for medical conditions.11 Additionally, some of the cognitive symptoms of PTSD, such as sleep problems, may worsen current health problems. Therefore, further collaboration between primary care physicians and mental health providers is beneficial in treating clients that have PTSD.
Conclusion
Posttraumatic stress disorder is a prevalent condition among veterans that is often comorbid with other medical conditions, which may have important implications for VA healthcare teams.3 It can manifest both psychologically and physiologically, and can greatly affect a patient’s quality of life.3 Veterans with PTSD may be at increased risk for certain medical conditions, such as cardiovascular disease.9,10,20 However, preventive screenings for medical conditions linked to PTSD and regular health assessments may reduce these risks.21 The VA’s infrastructure of integrated medical and mental healthcare can help provide comprehensive care to the many veterans who have both PTSD and serious medical conditions.21 While the relation between PTSD and inflammation remains unclear, it is clear that many people with PTSD have medical conditions that may be affected by PTSD symptoms.
1. US Department of Veteran Affairs. How common is PTSD? https://www.ptsd.va.gov/public/PTSD-overview/basics/how-common-is-ptsd.asp. Updated October 3, 2016. Accessed September 14, 2018.
2. Pai A, Suris AM, North CS. Posttraumatic stress disorder in the DSM-5: controversy, change, and conceptual considerations. Behav Sci (Basel). 2017;7(1):pii E7.
3. Gupta MA. Review of somatic symptoms in post-traumatic stress disorder. Int Rev Psychiatry. 2013;25(1):86-99.
4. Tsai J, Harpaz-Rotem I, Armour C, Southwick SM, Krystal JH, Pietrzak RH. Dimensional structure of DSM-5 posttraumatic stress disorder symptoms: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2015;76(5):546-553.
5. Schalinski I, Elbert TR, Schauer M. Cardiac defense in response to imminent threat in women with multiple trauma and severe PTSD. Psychophysiology. 2013;50(7):691-700.
6. National Institute of Mental Health. Post-traumatic stress disorder. https://www.nimh.nih.gov/health/topics/post-traumatic-stress-disorder-ptsd/index.shtml. Updated February 2016. Accessed September 14, 2018.
7. Sledjeski EM, Speisman B, Dierker LC. Does number of lifetime traumas explain the relationship between PTSD and chronic medical conditions? Answers from the National Comorbidity Survey-Replication (NCS-R). J Behav Med. 2008;31(4):341-349.
8. Tsai J, Shen J. Exploring the link between posttraumatic stress disorder and inflammation-related medical conditions: an epidemiological examination. Psychiatr Q. 2017;88(4):909-916.
9. Tulloch H, Greenman PS, Tassé V. Post-traumatic stress disorder among cardiac patients: Prevalence, risk factors, and considerations for assessment and treatment. Behav Sci (Basel). 2014;5(1):27-40.
10. Britvić D, Antičević V, Kaliterna M, et al. Comorbidities with posttraumatic stress disorder (PTSD) among combat veterans: 15 years postwar analysis. Int J Clin Health Psychol. 2015;15(2):81-92.
11. Pacella ML, Hruska B, Delahanty DL. The physical health consequences of PTSD and PTSD symptoms: a meta-analytic review. J Anxiety Disord. 2013;27(1):33-46.
12. Brouwers C, Wolf J, von Känel R. Inflammatory markers in PTSD. In: Martin CR, Preedy VR, Patel VB, eds. Comprehensive Guide to Post-Traumatic Stress Disorder. Zürich, Switzerland: Springer; 2016:979-993.
13. Passos IC, Vasconcelos-Moreno MP, Costa LG, et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry. 2015;2(11):1002-1012.
14. von Känel R, Begré S, Abbas CC, Saner H, Gander ML, Schmid JP. Inflammatory biomarkers in patients with posttraumatic stress disorder caused by myocardial infarction and the role of depressive symptoms. Neuroimmunomodulation. 2010;17(1):39-46.
15. Spitzer C, Barnow S, Völzke H, et al. Association of posttraumatic stress disorder with low-grade elevation of C-reactive protein: evidence from the general population. J Psychiatr Res. 2010;44(1):15-21.
16. Gola H, Engler H, Sommershof A, et al. Posttraumatic stress disorder is associated with an enhanced spontaneous production of pro-inflammatory cytokines by peripheral blood mononuclear cells. BMC Psychiatry. 2013;13:40.
17. Sidney S, Sorel ME, Quesenberry CP, et al. Comparative trends in heart disease, stroke, and all-cause mortality in the United States and a large integrated healthcare delivery system. Am J Med. 2018;131(7):829-836.e1.
18. US Department of Health and Human Service, Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2016: with chartbook on long-term trends in health. https://www.cdc.gov/nchs/data/hus/hus16.pdf. Published May 2017. Accessed September 14, 2018.
19. Weiner J, Richmond TS, Conigliaro J, Wiebe DJ. Military veteran mortality following a survived suicide attempt. BMC Public Health. 2011;11:374.
20. Roy SS, Foraker RE, Girton RA, Mansfield AJ. Posttraumatic stress disorder and incident heart failure among a community-based sample of US veterans. Am J Public Health. 2015;105(4):757-763.
21. Trivedi RB, Post EP, Sun H, et al. Prevalence, comorbidity, and prognosis of mental health among US veterans. Am J Public Health. 2015;105(12):2564-2569.
22. Richardson LK, Frueh BC, Acierno R. Prevalence estimates of combat-related post-traumatic stress disorder: a critical review. Aust N Z J Psychiatry. 2010;44(1):4-19.
23. US Department of Veterans Affairs. In the spotlight: VA uses technology to provide rural veterans greater access to specialty care services. https://www.patientcare.va.gov/In_the_Spotlight.asp. Updated June 3, 2015. Accessed September 14, 2018.
Posttraumatic stress disorder (PTSD) develops after exposure to a traumatic event, which can involve witnessing the traumatic event or directly experiencing the trauma.1 The prevalence of PTSD in the general population is approximately 7% to 8%.1 However, not everyone who experiences trauma develops PTSD since the majority of men and women experience at least 1 traumatic event in their lifetimes but do not develop PTSD.1
In order to be diagnosed with PTSD, a patient must meet several criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5).2 The patient is required to have exposure to trauma, begin having a certain number of prespecified symptoms, and these symptoms must persist for at least a month.2 Symptoms of PTSD include re-experiencing the traumatic event, avoidance of stimuli associated with the trauma, negative cognitions and mood, and hyperarousal.3,4 The hyperarousal that is associated with PTSD has been theorized to be either a result of the trauma experienced or exacerbation of a pre-existing tendency.5 This can manifest in various ways, such as hypervigilance, exaggerated startle response, trouble sleeping, problems concentrating, or irritability.3,5 These symptoms can cause individuals with PTSD to have elevated levels of stress and to experience difficulties with completing everyday tasks.6
PTSD and Inflimmation-Related Medical Conditions
Posttraumatic stress disorder has been linked to various physical health problems. Studies have found that PTSD is often comorbid with cardiovascular, autoimmune, musculoskeletal, digestive, chronic pain and respiratory disorders.3,7-11 Inflammation may be a contributing factor in the associations between PTSD and these conditions.12-16 Studies have found that increases in pro-inflammatory cytokines and interferons are associated with PTSD, as well as changes in immune-related blood cells.12-16
Considering that PTSD has been linked to many medical conditions that have inflammatory components, especially cardiovascular disease, inflammatory markers may be early indicators of PTSD.12-16 Additionally, inflammatory markers such as cytokines and interferons can be targeted through medications, and potentially influence symptoms.13 However, the relation between PTSD and inflammation remains unclear. Associations between PTSD and inflammation-related medical conditions may be due to confounding variables, such as sociodemographic characteristics and health behaviors. Moreover, the list of inflammation-related medical conditions is long and there is no universal agreement of what conditions are related to inflammation.
We recently conducted an epidemiological study using a representative sample of residents living in New York City and found significant associations between PTSD and some inflammation-related medical conditions.8 We found that participants who had PTSD were more than 4 times more likely to report having had a heart attack or emphysema than were those without PTSD. In addition, participants with PTSD were 2 times more likely to report having hypercholesterolemia, insulin resistance, and angina than were those without PTSD. However, we also found that participants who had PTSD were less likely to develop other inflammation-related conditions like hypertension, type 1 diabetes mellitus, asthma, coronary heart disease, stroke, osteoporosis, and failing kidney.
Together, these associations suggest there is a strong link between PTSD and certain medical conditions, but the link may not be solely based on inflammation.8 Moreover, positive associations between PTSD and hypertension, asthma, and coronary heart disease disappeared when depression was controlled for. This finding points to depression as a major factor, consistent with previous findings that depression is associated with the development of various medical conditions and may be a stronger factor than PTSD.8
Nonetheless, findings concerning the increased risk for heart problems among adults with PTSD are striking and important given that heart disease is one of the main causes of death in the United States.9 Specifically, well over half a million people in the United States die of heart disease annually as the leading cause of death.17 Heart disease has been one of the top 2 leading causes of death for Americans since 1975.18
In the veteran population, heart disease has also been found to be a leading cause of death, accounting for 20 percent of all deaths in veterans from 1993 to 2002.19 Posttraumatic stress disorder has been linked to a 55% increase in the chance of developing heart disease or dying from a heart-related medical problem.9 For example, data from the World Trade Center Registry showed that on average adults who developed PTSD from the 9/11 terrorist attack had a heightened risk for heart disease for 3 years after the event.9 Other studies of the U.S. veteran population have shown that veterans with PTSD are more likely to experience heart failure, myocardial infarction, and cardiac arrhythmia than other veterans.10,20
Veteran-Specific Issues
In the US veteran population, there is a higher prevalence of PTSD and physical health conditions when compared with the general population.21,21 The prevalence of combat-related PTSD in veterans ranges from 2% to 17%, compared with a 7% to 8% prevalence of PTSD in the general population.1,22 In a study of veterans who were seen in patient-aligned care teams (PACTs) > 1 year, 9.3% were diagnosed with PTSD and many of those with PTSD also had other medical conditions.21 It was found that 43% of veterans seen by PACTs with chronic pain had PTSD, 33% with hypertension had PTSD, and 32% with diabetes mellitus had PTSD.21 In another study of combat veterans it was found that those who were trauma-exposed had more physical health problems, regardless of the amount of time spent in combat.19 Consequentially, veterans with PTSD have been found to make more frequent visits to primary care and specialty medical care clinics. 21
Integrated healthcare has been a main service model for the Department of Veteran Affairs (VA) and several programs have been created to integrate mental health and primary care. For example, the VA primary care-mental health integration (PCMHI) program places mental health services within primary care services.21 Assessments of this program have demonstrated that it improves the screening of psychological disorders and preventive care of patients who have psychological disorders.21 Specifically, it has been found that contact with PCMHI diminishes risks for poor outcomes among psychiatric patients.21 Another program called SCAN-ECHO, provides specialized training for VA general practitioners on treating specific health conditions through a specialty care team and video conferencing.23 This VA program allows for patients in more remote locations to receive specialty care from generalists.23 While there has not yet been a focus in SCAN-ECHO on PTSD, this may be considered in the future as a way to better train primary care and mental health providers about PTSD and common comorbid medical conditions.
Through their professional experiences, VA practitioners have knowledge of the link between PTSD and various medical conditions. The VA has already implemented screening for PTSD in primary care clinics, but it is important for mental health providers and medical practitioners to continue educating themselves about medical comorbidities and the possible exacerbation of medical conditions due to PTSD.21 Some physical manifestations of PTSD symptoms, such as sleep disturbances, avoidance of crowds, or hypervigilance, can affect overall health. Hypervigilance can result in over-activation of stress pathways, which puts patients with PTSD at a heighted risk for medical conditions.11 Additionally, some of the cognitive symptoms of PTSD, such as sleep problems, may worsen current health problems. Therefore, further collaboration between primary care physicians and mental health providers is beneficial in treating clients that have PTSD.
Conclusion
Posttraumatic stress disorder is a prevalent condition among veterans that is often comorbid with other medical conditions, which may have important implications for VA healthcare teams.3 It can manifest both psychologically and physiologically, and can greatly affect a patient’s quality of life.3 Veterans with PTSD may be at increased risk for certain medical conditions, such as cardiovascular disease.9,10,20 However, preventive screenings for medical conditions linked to PTSD and regular health assessments may reduce these risks.21 The VA’s infrastructure of integrated medical and mental healthcare can help provide comprehensive care to the many veterans who have both PTSD and serious medical conditions.21 While the relation between PTSD and inflammation remains unclear, it is clear that many people with PTSD have medical conditions that may be affected by PTSD symptoms.
Posttraumatic stress disorder (PTSD) develops after exposure to a traumatic event, which can involve witnessing the traumatic event or directly experiencing the trauma.1 The prevalence of PTSD in the general population is approximately 7% to 8%.1 However, not everyone who experiences trauma develops PTSD since the majority of men and women experience at least 1 traumatic event in their lifetimes but do not develop PTSD.1
In order to be diagnosed with PTSD, a patient must meet several criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5).2 The patient is required to have exposure to trauma, begin having a certain number of prespecified symptoms, and these symptoms must persist for at least a month.2 Symptoms of PTSD include re-experiencing the traumatic event, avoidance of stimuli associated with the trauma, negative cognitions and mood, and hyperarousal.3,4 The hyperarousal that is associated with PTSD has been theorized to be either a result of the trauma experienced or exacerbation of a pre-existing tendency.5 This can manifest in various ways, such as hypervigilance, exaggerated startle response, trouble sleeping, problems concentrating, or irritability.3,5 These symptoms can cause individuals with PTSD to have elevated levels of stress and to experience difficulties with completing everyday tasks.6
PTSD and Inflimmation-Related Medical Conditions
Posttraumatic stress disorder has been linked to various physical health problems. Studies have found that PTSD is often comorbid with cardiovascular, autoimmune, musculoskeletal, digestive, chronic pain and respiratory disorders.3,7-11 Inflammation may be a contributing factor in the associations between PTSD and these conditions.12-16 Studies have found that increases in pro-inflammatory cytokines and interferons are associated with PTSD, as well as changes in immune-related blood cells.12-16
Considering that PTSD has been linked to many medical conditions that have inflammatory components, especially cardiovascular disease, inflammatory markers may be early indicators of PTSD.12-16 Additionally, inflammatory markers such as cytokines and interferons can be targeted through medications, and potentially influence symptoms.13 However, the relation between PTSD and inflammation remains unclear. Associations between PTSD and inflammation-related medical conditions may be due to confounding variables, such as sociodemographic characteristics and health behaviors. Moreover, the list of inflammation-related medical conditions is long and there is no universal agreement of what conditions are related to inflammation.
We recently conducted an epidemiological study using a representative sample of residents living in New York City and found significant associations between PTSD and some inflammation-related medical conditions.8 We found that participants who had PTSD were more than 4 times more likely to report having had a heart attack or emphysema than were those without PTSD. In addition, participants with PTSD were 2 times more likely to report having hypercholesterolemia, insulin resistance, and angina than were those without PTSD. However, we also found that participants who had PTSD were less likely to develop other inflammation-related conditions like hypertension, type 1 diabetes mellitus, asthma, coronary heart disease, stroke, osteoporosis, and failing kidney.
Together, these associations suggest there is a strong link between PTSD and certain medical conditions, but the link may not be solely based on inflammation.8 Moreover, positive associations between PTSD and hypertension, asthma, and coronary heart disease disappeared when depression was controlled for. This finding points to depression as a major factor, consistent with previous findings that depression is associated with the development of various medical conditions and may be a stronger factor than PTSD.8
Nonetheless, findings concerning the increased risk for heart problems among adults with PTSD are striking and important given that heart disease is one of the main causes of death in the United States.9 Specifically, well over half a million people in the United States die of heart disease annually as the leading cause of death.17 Heart disease has been one of the top 2 leading causes of death for Americans since 1975.18
In the veteran population, heart disease has also been found to be a leading cause of death, accounting for 20 percent of all deaths in veterans from 1993 to 2002.19 Posttraumatic stress disorder has been linked to a 55% increase in the chance of developing heart disease or dying from a heart-related medical problem.9 For example, data from the World Trade Center Registry showed that on average adults who developed PTSD from the 9/11 terrorist attack had a heightened risk for heart disease for 3 years after the event.9 Other studies of the U.S. veteran population have shown that veterans with PTSD are more likely to experience heart failure, myocardial infarction, and cardiac arrhythmia than other veterans.10,20
Veteran-Specific Issues
In the US veteran population, there is a higher prevalence of PTSD and physical health conditions when compared with the general population.21,21 The prevalence of combat-related PTSD in veterans ranges from 2% to 17%, compared with a 7% to 8% prevalence of PTSD in the general population.1,22 In a study of veterans who were seen in patient-aligned care teams (PACTs) > 1 year, 9.3% were diagnosed with PTSD and many of those with PTSD also had other medical conditions.21 It was found that 43% of veterans seen by PACTs with chronic pain had PTSD, 33% with hypertension had PTSD, and 32% with diabetes mellitus had PTSD.21 In another study of combat veterans it was found that those who were trauma-exposed had more physical health problems, regardless of the amount of time spent in combat.19 Consequentially, veterans with PTSD have been found to make more frequent visits to primary care and specialty medical care clinics. 21
Integrated healthcare has been a main service model for the Department of Veteran Affairs (VA) and several programs have been created to integrate mental health and primary care. For example, the VA primary care-mental health integration (PCMHI) program places mental health services within primary care services.21 Assessments of this program have demonstrated that it improves the screening of psychological disorders and preventive care of patients who have psychological disorders.21 Specifically, it has been found that contact with PCMHI diminishes risks for poor outcomes among psychiatric patients.21 Another program called SCAN-ECHO, provides specialized training for VA general practitioners on treating specific health conditions through a specialty care team and video conferencing.23 This VA program allows for patients in more remote locations to receive specialty care from generalists.23 While there has not yet been a focus in SCAN-ECHO on PTSD, this may be considered in the future as a way to better train primary care and mental health providers about PTSD and common comorbid medical conditions.
Through their professional experiences, VA practitioners have knowledge of the link between PTSD and various medical conditions. The VA has already implemented screening for PTSD in primary care clinics, but it is important for mental health providers and medical practitioners to continue educating themselves about medical comorbidities and the possible exacerbation of medical conditions due to PTSD.21 Some physical manifestations of PTSD symptoms, such as sleep disturbances, avoidance of crowds, or hypervigilance, can affect overall health. Hypervigilance can result in over-activation of stress pathways, which puts patients with PTSD at a heighted risk for medical conditions.11 Additionally, some of the cognitive symptoms of PTSD, such as sleep problems, may worsen current health problems. Therefore, further collaboration between primary care physicians and mental health providers is beneficial in treating clients that have PTSD.
Conclusion
Posttraumatic stress disorder is a prevalent condition among veterans that is often comorbid with other medical conditions, which may have important implications for VA healthcare teams.3 It can manifest both psychologically and physiologically, and can greatly affect a patient’s quality of life.3 Veterans with PTSD may be at increased risk for certain medical conditions, such as cardiovascular disease.9,10,20 However, preventive screenings for medical conditions linked to PTSD and regular health assessments may reduce these risks.21 The VA’s infrastructure of integrated medical and mental healthcare can help provide comprehensive care to the many veterans who have both PTSD and serious medical conditions.21 While the relation between PTSD and inflammation remains unclear, it is clear that many people with PTSD have medical conditions that may be affected by PTSD symptoms.
1. US Department of Veteran Affairs. How common is PTSD? https://www.ptsd.va.gov/public/PTSD-overview/basics/how-common-is-ptsd.asp. Updated October 3, 2016. Accessed September 14, 2018.
2. Pai A, Suris AM, North CS. Posttraumatic stress disorder in the DSM-5: controversy, change, and conceptual considerations. Behav Sci (Basel). 2017;7(1):pii E7.
3. Gupta MA. Review of somatic symptoms in post-traumatic stress disorder. Int Rev Psychiatry. 2013;25(1):86-99.
4. Tsai J, Harpaz-Rotem I, Armour C, Southwick SM, Krystal JH, Pietrzak RH. Dimensional structure of DSM-5 posttraumatic stress disorder symptoms: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2015;76(5):546-553.
5. Schalinski I, Elbert TR, Schauer M. Cardiac defense in response to imminent threat in women with multiple trauma and severe PTSD. Psychophysiology. 2013;50(7):691-700.
6. National Institute of Mental Health. Post-traumatic stress disorder. https://www.nimh.nih.gov/health/topics/post-traumatic-stress-disorder-ptsd/index.shtml. Updated February 2016. Accessed September 14, 2018.
7. Sledjeski EM, Speisman B, Dierker LC. Does number of lifetime traumas explain the relationship between PTSD and chronic medical conditions? Answers from the National Comorbidity Survey-Replication (NCS-R). J Behav Med. 2008;31(4):341-349.
8. Tsai J, Shen J. Exploring the link between posttraumatic stress disorder and inflammation-related medical conditions: an epidemiological examination. Psychiatr Q. 2017;88(4):909-916.
9. Tulloch H, Greenman PS, Tassé V. Post-traumatic stress disorder among cardiac patients: Prevalence, risk factors, and considerations for assessment and treatment. Behav Sci (Basel). 2014;5(1):27-40.
10. Britvić D, Antičević V, Kaliterna M, et al. Comorbidities with posttraumatic stress disorder (PTSD) among combat veterans: 15 years postwar analysis. Int J Clin Health Psychol. 2015;15(2):81-92.
11. Pacella ML, Hruska B, Delahanty DL. The physical health consequences of PTSD and PTSD symptoms: a meta-analytic review. J Anxiety Disord. 2013;27(1):33-46.
12. Brouwers C, Wolf J, von Känel R. Inflammatory markers in PTSD. In: Martin CR, Preedy VR, Patel VB, eds. Comprehensive Guide to Post-Traumatic Stress Disorder. Zürich, Switzerland: Springer; 2016:979-993.
13. Passos IC, Vasconcelos-Moreno MP, Costa LG, et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry. 2015;2(11):1002-1012.
14. von Känel R, Begré S, Abbas CC, Saner H, Gander ML, Schmid JP. Inflammatory biomarkers in patients with posttraumatic stress disorder caused by myocardial infarction and the role of depressive symptoms. Neuroimmunomodulation. 2010;17(1):39-46.
15. Spitzer C, Barnow S, Völzke H, et al. Association of posttraumatic stress disorder with low-grade elevation of C-reactive protein: evidence from the general population. J Psychiatr Res. 2010;44(1):15-21.
16. Gola H, Engler H, Sommershof A, et al. Posttraumatic stress disorder is associated with an enhanced spontaneous production of pro-inflammatory cytokines by peripheral blood mononuclear cells. BMC Psychiatry. 2013;13:40.
17. Sidney S, Sorel ME, Quesenberry CP, et al. Comparative trends in heart disease, stroke, and all-cause mortality in the United States and a large integrated healthcare delivery system. Am J Med. 2018;131(7):829-836.e1.
18. US Department of Health and Human Service, Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2016: with chartbook on long-term trends in health. https://www.cdc.gov/nchs/data/hus/hus16.pdf. Published May 2017. Accessed September 14, 2018.
19. Weiner J, Richmond TS, Conigliaro J, Wiebe DJ. Military veteran mortality following a survived suicide attempt. BMC Public Health. 2011;11:374.
20. Roy SS, Foraker RE, Girton RA, Mansfield AJ. Posttraumatic stress disorder and incident heart failure among a community-based sample of US veterans. Am J Public Health. 2015;105(4):757-763.
21. Trivedi RB, Post EP, Sun H, et al. Prevalence, comorbidity, and prognosis of mental health among US veterans. Am J Public Health. 2015;105(12):2564-2569.
22. Richardson LK, Frueh BC, Acierno R. Prevalence estimates of combat-related post-traumatic stress disorder: a critical review. Aust N Z J Psychiatry. 2010;44(1):4-19.
23. US Department of Veterans Affairs. In the spotlight: VA uses technology to provide rural veterans greater access to specialty care services. https://www.patientcare.va.gov/In_the_Spotlight.asp. Updated June 3, 2015. Accessed September 14, 2018.
1. US Department of Veteran Affairs. How common is PTSD? https://www.ptsd.va.gov/public/PTSD-overview/basics/how-common-is-ptsd.asp. Updated October 3, 2016. Accessed September 14, 2018.
2. Pai A, Suris AM, North CS. Posttraumatic stress disorder in the DSM-5: controversy, change, and conceptual considerations. Behav Sci (Basel). 2017;7(1):pii E7.
3. Gupta MA. Review of somatic symptoms in post-traumatic stress disorder. Int Rev Psychiatry. 2013;25(1):86-99.
4. Tsai J, Harpaz-Rotem I, Armour C, Southwick SM, Krystal JH, Pietrzak RH. Dimensional structure of DSM-5 posttraumatic stress disorder symptoms: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2015;76(5):546-553.
5. Schalinski I, Elbert TR, Schauer M. Cardiac defense in response to imminent threat in women with multiple trauma and severe PTSD. Psychophysiology. 2013;50(7):691-700.
6. National Institute of Mental Health. Post-traumatic stress disorder. https://www.nimh.nih.gov/health/topics/post-traumatic-stress-disorder-ptsd/index.shtml. Updated February 2016. Accessed September 14, 2018.
7. Sledjeski EM, Speisman B, Dierker LC. Does number of lifetime traumas explain the relationship between PTSD and chronic medical conditions? Answers from the National Comorbidity Survey-Replication (NCS-R). J Behav Med. 2008;31(4):341-349.
8. Tsai J, Shen J. Exploring the link between posttraumatic stress disorder and inflammation-related medical conditions: an epidemiological examination. Psychiatr Q. 2017;88(4):909-916.
9. Tulloch H, Greenman PS, Tassé V. Post-traumatic stress disorder among cardiac patients: Prevalence, risk factors, and considerations for assessment and treatment. Behav Sci (Basel). 2014;5(1):27-40.
10. Britvić D, Antičević V, Kaliterna M, et al. Comorbidities with posttraumatic stress disorder (PTSD) among combat veterans: 15 years postwar analysis. Int J Clin Health Psychol. 2015;15(2):81-92.
11. Pacella ML, Hruska B, Delahanty DL. The physical health consequences of PTSD and PTSD symptoms: a meta-analytic review. J Anxiety Disord. 2013;27(1):33-46.
12. Brouwers C, Wolf J, von Känel R. Inflammatory markers in PTSD. In: Martin CR, Preedy VR, Patel VB, eds. Comprehensive Guide to Post-Traumatic Stress Disorder. Zürich, Switzerland: Springer; 2016:979-993.
13. Passos IC, Vasconcelos-Moreno MP, Costa LG, et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry. 2015;2(11):1002-1012.
14. von Känel R, Begré S, Abbas CC, Saner H, Gander ML, Schmid JP. Inflammatory biomarkers in patients with posttraumatic stress disorder caused by myocardial infarction and the role of depressive symptoms. Neuroimmunomodulation. 2010;17(1):39-46.
15. Spitzer C, Barnow S, Völzke H, et al. Association of posttraumatic stress disorder with low-grade elevation of C-reactive protein: evidence from the general population. J Psychiatr Res. 2010;44(1):15-21.
16. Gola H, Engler H, Sommershof A, et al. Posttraumatic stress disorder is associated with an enhanced spontaneous production of pro-inflammatory cytokines by peripheral blood mononuclear cells. BMC Psychiatry. 2013;13:40.
17. Sidney S, Sorel ME, Quesenberry CP, et al. Comparative trends in heart disease, stroke, and all-cause mortality in the United States and a large integrated healthcare delivery system. Am J Med. 2018;131(7):829-836.e1.
18. US Department of Health and Human Service, Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2016: with chartbook on long-term trends in health. https://www.cdc.gov/nchs/data/hus/hus16.pdf. Published May 2017. Accessed September 14, 2018.
19. Weiner J, Richmond TS, Conigliaro J, Wiebe DJ. Military veteran mortality following a survived suicide attempt. BMC Public Health. 2011;11:374.
20. Roy SS, Foraker RE, Girton RA, Mansfield AJ. Posttraumatic stress disorder and incident heart failure among a community-based sample of US veterans. Am J Public Health. 2015;105(4):757-763.
21. Trivedi RB, Post EP, Sun H, et al. Prevalence, comorbidity, and prognosis of mental health among US veterans. Am J Public Health. 2015;105(12):2564-2569.
22. Richardson LK, Frueh BC, Acierno R. Prevalence estimates of combat-related post-traumatic stress disorder: a critical review. Aust N Z J Psychiatry. 2010;44(1):4-19.
23. US Department of Veterans Affairs. In the spotlight: VA uses technology to provide rural veterans greater access to specialty care services. https://www.patientcare.va.gov/In_the_Spotlight.asp. Updated June 3, 2015. Accessed September 14, 2018.
Reducing Benzodiazepine Prescribing in Older Veterans: A Direct-to-Consumer Educational Brochure
This quality improvement project used an educational brochure to help older veterans reduce their benzodiazepine use.
Benzodiazepines (BZDs) are among the most commonly prescribed medications. A recent study found that in 2008, more than 5% of Americans used a BZD, and the percentage was almost 9% among Americans aged ≥ 65 years.1,2 Among veterans, BZD use is even higher, in part because of the high prevalence of posttraumatic stress disorder (PTSD). One study found that more than 30% of veterans with PTSD received at least 1 BZD prescription.3 The risks associated with BZD treatment for PTSD are compounded by concurrent use of other sedatives and opioids prescribed for co-occurring chronic pain and insomnia.3
Older adults metabolize long-acting BZDs more slowly and generally have an increased sensitivity to the adverse effects (AEs) of all BZDs.4 In older adults, BZD use has been associated with cognitive decline, dementia, falls and consequent fractures, and adverse respiratory outcomes.5-12 The risk of most but not all of these AEs was increased with higher BZD dose or long-term BZD use, which this quality improvement project (QIP) defines as having at least a 60-day supply of BZD prescriptions dispensed within the past year.
Long-term BZD use increases with age. One study found that, among patients receiving a BZD, the rate of long-term BZD use was more than double in older adults (31.4%) than it was in adults aged between 18 and 35 years (14.7%).2 For these reasons, the 2012 Beers criteria of the American Geriatrics Society recommend avoiding all types of BZDs in the treatment of insomnia, agitation, or delirium in patients aged > 65 years.13 Despite this recommendation, the prevalence of BZD use in older adults remains high.14
Some innovative approaches have been developed to address the inappropriate use, including overuse and misuse, of BZDs in older adults.15 In one approach, direct-to-consumer (DTC) information is used to empower patients to collaborate with their physician to manage their health. Results from several studies suggest that providing older patients with information on BZD risks and benefits increases patient–physician interaction and thereby decreases inappropriate BZD use and improves health outcomes.4,16,17 One study found that perceptions of BZD risks increased 1 week after exposure to a DTC educational brochure (EB), with intention to discuss BZD discontinuation with their physician higher for patients who received the EB than it was for those who did not (83.1% vs 44.3%; P < .0001).16 The EMPOWER (Eliminating Medications Through Patient Ownership of End Results) cluster randomized controlled trial assessed the effectiveness of a DTC EB focused on BZD risks in older adults.17 In that seminal study, patients who received a DTC EB were more likely than were comparison patients to discontinue BZD within 6 months (27% vs 5%; risk difference, 23%; 95% CI, 14%-32%).
The Veterans Integrated Systems Network (VISN) 22 Academic Detailing Program is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20 With BZD use among older veterans remaining high, the VISN 22 program initiated a clinical QIP modeled on the EMPOWER trial. Veterans in VISN 22 received the DTC EB, which included information on BZD risks and encouraged them to discuss their BZD treatment with their health care provider. VISN 22 was the first VISN in the VHA to implement the EMPOWER protocol.
As this was a QIP, all eligible veterans in VISN 22 were mailed the DTC EB, thus making it difficult to estimate the impact of the EB on BZD discontinuation in this VISN. Therefore, DTC EB efficacy was estimated by comparing BZD discontinuation between VISN 22 and VISN 21, an adjacent VISN that did not mail the DTC EB.
Methods
Two QIPs were undertaken to determine the impact of DTC EB on BZD use in older veterans in the VHA.
Quality Improvement Project 1
Design. A retrospective cohort analysis was performed. The VISN 22 catchment area, which encompasses VA facilities and clinics in southern California and southern Nevada, serves about 500,000 veterans, a substantial proportion of whom are aged ≥ 65 years. Among these older veterans are active long-term BZD users, who were defined as having ≥ 60-day supply of BZD prescriptions dispensed within the past year. Each active long-term user with a BZD prescription released within 200 days before the index date (the date the user was to meet with the prescribing physician) was mailed an EB 2 to 8 weeks in advance of the visit. Excluded from analysis were veterans with a schizophrenia, spinal cord injury, or seizure disorder diagnosis recorded in both their inpatient and outpatient medical records; veterans seen by Palliative Care within the past year; and veterans who died before analysis was completed.
Education Brochure. The EB for VISN 22 (Figure 1, see
Patients. The sample consisted of all veterans identified as meeting the inclusion criteria and being enrolled in VISN 22. The EB was mailed once to veterans on a rolling basis from December 2014 to February 2016. Change in BZD use was analyzed only after 9 to 24 months had passed since the index appointment with the prescribing physician. This period included 12 weeks for BZD taper and then 6 months after taper.
Analysis. For each veteran, monthly mean lorazepam equivalent (LE) was calculated using as many as 12 fills before the index date. Average daily dose of LE was calculated by dividing the sum of LE from all included prescriptions by total number of days between the first fill and the index date. The BZD prescription fills were evaluated after the index date. Veterans who received at least 1 prescription after the index date but then had no BZD prescription activity in VA clinics for 3 consecutive months during the 9-month observation period were recorded as having tapered and then discontinued BZD. Veterans who had no BZD prescription activity in VA clinics after the index date and during the 9-month observation period were recorded as having discontinued BZD without tapering. For veterans who had BZD prescription activity in VA clinics after the index date and during the 9-month observation period, mean LE was calculated by dividing the total LE for BZD prescriptions after the index date by number of days from the first fill after the index date to the date of analysis.
Quality Improvement Project 2
Design. A retrospective cohort analysis using PSM was performed on a subgroup of the QIP-1 sample to evaluate the impact of EB on BZD prescribing in the VA during 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. Veterans in the analysis were active long-term BZD users, had at least 1 BZD prescription released within 200 days before the index date, were aged ≥ 65 years, and had an appointment scheduled with their BZD prescriber within 2 to 8 weeks (Figure 2).
Patients. VISN 22 implemented QIP-2, a real-world application of a modified EMPOWER program, by identifying eligible veterans on a rolling basis from December 2014 to August 2015. All veterans who were identified and sent an EB during this period were included in the case group. The index date was defined as the first of the month the EB was mailed. Veterans with a pending appointment were chosen because the lead time would allow them to receive the EB and prepare to discuss it with the physician during the visit.
A comparator group was drawn from the adjacent VISN 21 catchment area, which encompasses VA facilities and clinics in Hawaii, northern California, and northern Nevada. During the observation period, VISN 21 did not mail any EBs specifically addressing BZD risks. Veterans in the comparator group had an appointment scheduled with their BZD prescribing physician within 4 weeks, were aged ≥ 65 years on the index date (first of the month before the next appointment, coinciding with the date EBs were sent to VISN 22 veterans), were active long-term BZD users, and had at least 1 BZD prescription released within 200 days before the index date. All patients were followed for up to 12 months after the index date, with BZD discontinuation recorded 9 and 12 months after the index date.
Propensity Score Matching
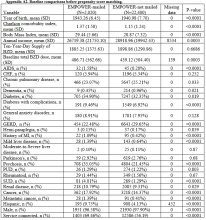
Propensity score (PS) was estimated with logistic regression analysis with treatment as the dependent variable and baseline characteristics as the independent variables.21,22 One-to-one matching on the PS was performed using the nearest neighbor approach without replacements. Independent variables related to outcome but unrelated to EB exposure were selected for PS development.22 These variables included year of birth; male sex; Hispanic ethnicity; annual income; service connection status; region; body mass index; Charlson Comorbidity Index category; total baseline BZD dose; and diagnosis of AIDS, nonmetastatic cancer, metastatic cancer, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), dementia, diabetes mellitus (DM), DM with complications, gastroesophageal reflux disease (GERD), general anxiety disorder (GAD), hemiparaplegia, liver disease (mild), liver disease (moderate to severe), myocardial infarction (MI), Parkinson disease, peptic ulcer disease (PUD), psychosis, renal disease, rheumatoid arthritis (RA), or substance use disorder (SUD).
The EMPOWER cluster randomized controlled trial (RCT) demonstrated the effectiveness of EB exposure in a Canadian population of elderly patients who were long-term BZD users.17 Randomized controlled trials are the gold standard for clinical trials because they can establish causal inference.23-25 Given ethical and practical concerns, however, RCTs cannot be applied to all clinical scenarios. Although EMPOWER is reported to be an effective tool in reducing BZD use in older adults, its application in a real-world, large, integrated health care system remains untested. Observational studies are often conducted as an alternative to RCTs but are subject to selection bias because of their lack of randomization.26 Therefore, robust research methods are needed to generate unbiased estimates of the impact of an intervention on an outcome. Propensity score matching simulates an RCT by balancing the covariates across treatment groups.21,22,27 Observed patient characteristics are used to estimate PS, the probability that treatment will be received. Logistic or probit regression is used to balance the potential confounding covariates between the treatment groups.Once PSs are known, mean treatment effect can be estimated without the mean model.28 In other words, PSM methods can be used to generate an unbiased estimate of the treatment.
Propensity Score Analysis
Baseline characteristics were compared using Student t test (continuous variables) and χ2 test (discrete variables). Results are presented as means and standard deviations (continuous variables) and frequency and percentage (discrete variables).
The main outcome was BZD discontinuation 9 and 12 months after the index date. A postindex lag of 6 months was used to capture any tapering (Figure 2). Discontinuation, defined as 3 consecutive months of no BZD prescription on hand, was measured for 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. An estimate was made of the difference in the proportions of BZD discontinuers who received the EB and BZD discontinuers who did not receive the EB, where mean treatment (risk difference) was presented as the absolute risk difference with a 95% CI. Standard errors and 95% CIs for the risk differences were generated with biased-corrected CIs from 1,000 bootstrap samples.
Sensitivity Analyses
Naïve multivariate logistic regression analysis was performed to evaluate the association between EB exposure and BZD discontinuation while controlling for potential confounders. Results are presented as odds ratios (ORs) and 95% CIs. Confounders identified were the same covariates used to generate the PSs.
Several analyses were performed to test the sensitivity of the methods applied using PSM by changing caliber size while maintaining the nearest neighbor approach without replacement. Linear regression analysis was performed with robust standard errors to estimate the risk difference of BZD discontinuation between EB-exposed and EB-unexposed veterans.
Statistical significance was set at P < .05. All statistical analyses were performed with Stata/SE Version 13 (College Station, TX).
Results
Quality Improvement Project 1
On a rolling basis from December 2014 to February 2016, the EB was mailed once to 3,896 VISN 22 veterans 2 to 8 weeks before a clinic appointment with their BZD prescribing physician.
Quality Improvement Project 2
Of all the VISN 22 and VISN 21 veterans, 24,420 met the inclusion and exclusion criteria. Of these 24,420 veterans, 2,020 (8.3%) were in VISN 22 and received the EB between December 2014 and August 2015 (QIP-1), and 22,400 (91.7%) were in VISN 21 and did not receive the EB.
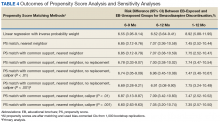
Naïve Results Before PS Matching. In the naïve analyses, a larger proportion of EB-exposed vs unexposed veterans discontinued BZD; in addition, reductions were 6.6%, 7.4%, and 9.5% larger for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (P < .0001 for all comparisons; Table 2).
After controlling for potential confounders, the naïve logistic regression analyses found EB exposure was significantly associated with 44%, 32%, and 42% increases in the odds of BZD discontinuation for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (Table 3).
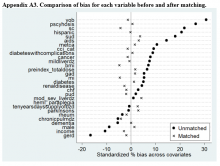
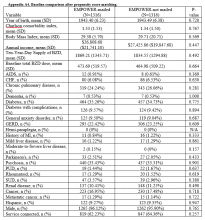
Propensity Score Matching. Before matching, there were significant differences in baseline characteristics of veterans who met the inclusion and exclusion criteria, with few exceptions (eAppendices 2 and 3, ).
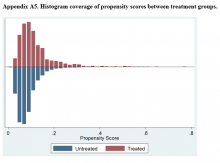
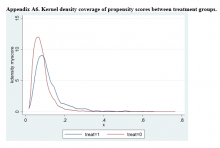
Propensity Score Matching Results. Inspection of PSs revealed good coverage across treatment groups on a histogram plot and a kernel density plot (eAppendices 5 and 6).
Discussion
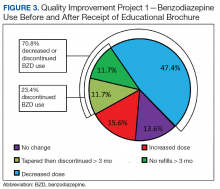
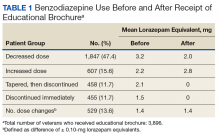
This QIP was the first to evaluate the impact of an EMPOWER-modeled DTC EB in a large, integrated health care system in the U.S. It was also the first to demonstrate potential benefits of a DTC EB designed for older veterans who are long-term BZD users. In this QIP, which mailed the EB to 3,896 veterans, 1,847 (47.4%) decreased their BZD dose, 458 (11.7%) tapered and then discontinued BZD, and 455 (11.7%) immediately discontinued BZD. The total percentage of veterans who discontinued BZD (23.4%; 913/3,896) was similar to the 27% reported in the EMPOWER trial.17 However, the risk difference between the 1,316 EB-exposed VISN 22 veterans (QIP-1) and the 1,316 EB-unexposed VISN 21 veterans in this QIP was significantly lower than the 23% risk difference in EMPOWER (though it still demonstrated a significantly larger reduction for EB-exposed veterans).17
Given this inclusion of all qualifying veterans from the catchment area studied in this QIP, and given the ethical and practical concerns, an RCT was not possible. Therefore, PSM methods were used to balance the covariates across treatment groups and thereby simulate an RCT.21,22,27 With use of the PSM approach, findings from the descriptive analysis were confirmed and potential selection bias reduced.
Study Limitations
The less robust risk difference found in this QIP has several possible explanations. The authors’ use of a DTC EB coincided with a national VA effort to reduce older veterans’ use of BZDs and other inappropriate medications. For instance, during the study period, academic detailing was being implemented to reduce use of BZDs, particularly in combination with opioids, across VHA facilities and clinics. (Academic detailing is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20) However, QIP-2 results and PS analysis of a subgroup of the original sample suggest that EB-exposed veterans were significantly more likely than were their unexposed counterparts were to discontinue BZD. To an extent, this analysis controlled for these other efforts to reduce BZD use in VHA clinics and can be considered a study strength.
Another limitation is the study design, which lacked a control group and did not consider the possibility that some facility or clinic physicians might influence others. Although the region variable was controlled for in PSM, the authors did not capture facility characteristics, including frequency of prescribing BZD and use of a protocol for enforcing the Beers criteria. Such confounders might have influenced outcomes. Unlike the EMPOWER trial,17 this QIP did not assess or exclude cognitively impaired veterans. It is reasonable to assume that these veterans might not understand some EB messages and consequently might fail to engage their physicians. Failure to initiate discussion with a physician would attenuate the impact of the EB.
Study Strengths
A strength of this QIP was its use of a DTC EB in a large, regional sample of older veterans in a real-world clinical setting. In addition, the study group (EB-exposed veterans) and the comparator group (EB-unexposed veterans) were from similar geographic areas (primarily California and Nevada).
Conclusion
Results of this study suggest that a DTC EB, designed to reduce BZD use among older veterans, was effective in helping patients lower their BZD dose and discontinue BZD. The likelihood of discontinuing BZD 9 and 12 months after the index date was significantly higher for veterans who received an EB modeled on the EMPOWER educational brochure than for a comparator group of veterans who did not receive the EB and were receiving care during the same observation period. In the future, it would be beneficial to use a design that controls for physician exposure to academic detailing focused on BZD reduction and that accounts for the cluster effects of facility practice. Despite these limitations, this QIP is the first real-world empirical example of using an EMPOWER-modeled DTC EB to decrease BZD use among older veterans. Furthermore, these results suggest that a DTC EB can be used to target other high-risk prescription drugs, such as opioids, particularly if alternative treatment options can be provided.
Acknowledgments
Dr. Hauser thanks Cathy, Anika, Katia, and Max Hauser, and Alba and Kevin Quinlan, for their support. In memory of Jirina Hauser, who died on Mother’s Day, May 14, 2017, at the age of 100.
1. Dell’osso B, Lader M. Do benzodiazepines still deserve a major role in the treatment of psychiatric disorders? A critical reappraisal. Eur Psychiatry. 2013;28(1):7-20.
2. Olfson M, King M, Schoenbaum M. Benzodiazepine use in the United States. JAMA Psychiatry. 2015;72(2):136-142.
3. Bernardy NC, Lund BC, Alexander B, Friedman MJ. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
4. Roberts KJ. Patient empowerment in the United States: a critical commentary. Health Expect. 1999;2(2):82-92.
5. Paterniti S, Dufouil C, Alpérovitch A. Long-term benzodiazepine use and cognitive decline in the elderly: the Epidemiology of Vascular Aging Study. J Clin Psychopharmacol. 2002;22(3):285-293.
6. van der Hooft CS, Schoofs MW, Ziere G, et al. Inappropriate benzodiazepine use in older adults and the risk of fracture. Br J Clin Pharmacol. 2008;66(2):276-282.
7. Zint K, Haefeli WE, Glynn RJ, Mogun H, Avorn J, Stürmer T. Impact of drug interactions, dosage, and duration of therapy on the risk of hip fracture associated with benzodiazepine use in older adults. Pharmacoepidemiol Drug Saf. 2010;19(12):1248-1255.
8. Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc. 2011;59(10):1883-1890.
9. de Gage SB, Bégaud B, Bazin F, et al. Benzodiazepine use and risk of dementia: prospective population based study. BMJ. 2012;345:e6231
10. Tannenbaum C, Paquette A, Hilmer S, Holroyd-Leduc J, Carnahan R. A systematic review of amnestic and non-amnestic mild cognitive impairment induced by anticholinergic, antihistamine, GABAergic and opioid drugs. Drugs Aging. 2012;29(8):639-658.
11. Vozoris NT, Fischer HD, Wang X, et al. Benzodiazepine drug use and adverse respiratory outcomes among older adults with chronic obstructive pulmonary disease. Eur Respir J. 2014;44(2):332-340.
12. Gomm W, von Holt K, Thomé F, et al. Regular benzodiazepine and z-substance use and risk of dementia: an analysis of German claims data. J Alzheimers Dis. 2016;54(2):801-808.
13. American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60(4):616-631.
14. National Institutes of Health. Despite risks, benzodiazepine use highest in older people. https://www.nih.gov/news-events/news-releases/despite-risks-benzodiaze pine-use-highest-older-people. Published December 17, 2014. Accessed July 31, 2018.
15. Airagnes G, Pelissolo A, Lavallée M, Flament M, Limosin F. Benzodiazepine misuse in the elderly: risk factors, consequences, and management. Curr Psychiatry Rep. 2016;18(10):89.
16. Martin P, Tamblyn R, Ahmed S, Tannenbaum C. A drug education tool developed for older adults changes knowledge, beliefs and risk perceptions about inappropriate benzodiazepine prescriptions in the elderly. Patient Educ Couns. 2013;92(1):81-87.
17. Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898.
18. Soumerai SB, Avorn J. Principles of educational outreach (‘academic detailing’) to improve clinical decision making. JAMA. 1990;263(4):549-556.
19. Fischer MA, Avorn J. Academic detailing can play a key role in assessing and implementing comparative effectiveness research findings. Health Aff (Millwood). 2012;31(10):2206-2212.
20. Wells DL, Popish S, Kay C, Torrise V, Christopher ML. VA Academic Detailing Service: implementation and lessons learned. Fed Pract. 2016;33(5):38-42.
21. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424.
22. Brookhart MA, Schneeweiss S, Rothman KJ, Glynn RJ, Avorn J, Stürmer T. Variable selection for propensity score models. Am J Epidemiol. 2006;163(12):1149-1156.
23. Rubin DB. Estimating causal effects of treatments in randomized and nonrandomized studies. J Ed Psych. 1974;66(5):688-701.
24. Greenland S. An introduction to instrumental variables for epidemiologists. Int J Epidemiol. 2000;29(4):722-729.
25. Cartwright N. What are randomized controlled trials good for? Philos Stud. 2010;147(1):59.
26. Kleinbaum DG, Morgenstern H, Kupper LL. Selection bias in epidemiologic studies. Am J Epidemiol. 1981;113(4):452-463.
27. Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41-55.
28. Pirracchio R, Carone M, Rigon MR, Caruana E, Mebazaa A, Chevret S. Propensity score estimators for the average treatment effect and the average treatment effect on the treated may yield very different estimates. Stat Methods Med Res. 2016;25(5):1938-1954.
This quality improvement project used an educational brochure to help older veterans reduce their benzodiazepine use.
This quality improvement project used an educational brochure to help older veterans reduce their benzodiazepine use.
Benzodiazepines (BZDs) are among the most commonly prescribed medications. A recent study found that in 2008, more than 5% of Americans used a BZD, and the percentage was almost 9% among Americans aged ≥ 65 years.1,2 Among veterans, BZD use is even higher, in part because of the high prevalence of posttraumatic stress disorder (PTSD). One study found that more than 30% of veterans with PTSD received at least 1 BZD prescription.3 The risks associated with BZD treatment for PTSD are compounded by concurrent use of other sedatives and opioids prescribed for co-occurring chronic pain and insomnia.3
Older adults metabolize long-acting BZDs more slowly and generally have an increased sensitivity to the adverse effects (AEs) of all BZDs.4 In older adults, BZD use has been associated with cognitive decline, dementia, falls and consequent fractures, and adverse respiratory outcomes.5-12 The risk of most but not all of these AEs was increased with higher BZD dose or long-term BZD use, which this quality improvement project (QIP) defines as having at least a 60-day supply of BZD prescriptions dispensed within the past year.
Long-term BZD use increases with age. One study found that, among patients receiving a BZD, the rate of long-term BZD use was more than double in older adults (31.4%) than it was in adults aged between 18 and 35 years (14.7%).2 For these reasons, the 2012 Beers criteria of the American Geriatrics Society recommend avoiding all types of BZDs in the treatment of insomnia, agitation, or delirium in patients aged > 65 years.13 Despite this recommendation, the prevalence of BZD use in older adults remains high.14
Some innovative approaches have been developed to address the inappropriate use, including overuse and misuse, of BZDs in older adults.15 In one approach, direct-to-consumer (DTC) information is used to empower patients to collaborate with their physician to manage their health. Results from several studies suggest that providing older patients with information on BZD risks and benefits increases patient–physician interaction and thereby decreases inappropriate BZD use and improves health outcomes.4,16,17 One study found that perceptions of BZD risks increased 1 week after exposure to a DTC educational brochure (EB), with intention to discuss BZD discontinuation with their physician higher for patients who received the EB than it was for those who did not (83.1% vs 44.3%; P < .0001).16 The EMPOWER (Eliminating Medications Through Patient Ownership of End Results) cluster randomized controlled trial assessed the effectiveness of a DTC EB focused on BZD risks in older adults.17 In that seminal study, patients who received a DTC EB were more likely than were comparison patients to discontinue BZD within 6 months (27% vs 5%; risk difference, 23%; 95% CI, 14%-32%).
The Veterans Integrated Systems Network (VISN) 22 Academic Detailing Program is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20 With BZD use among older veterans remaining high, the VISN 22 program initiated a clinical QIP modeled on the EMPOWER trial. Veterans in VISN 22 received the DTC EB, which included information on BZD risks and encouraged them to discuss their BZD treatment with their health care provider. VISN 22 was the first VISN in the VHA to implement the EMPOWER protocol.
As this was a QIP, all eligible veterans in VISN 22 were mailed the DTC EB, thus making it difficult to estimate the impact of the EB on BZD discontinuation in this VISN. Therefore, DTC EB efficacy was estimated by comparing BZD discontinuation between VISN 22 and VISN 21, an adjacent VISN that did not mail the DTC EB.
Methods
Two QIPs were undertaken to determine the impact of DTC EB on BZD use in older veterans in the VHA.
Quality Improvement Project 1
Design. A retrospective cohort analysis was performed. The VISN 22 catchment area, which encompasses VA facilities and clinics in southern California and southern Nevada, serves about 500,000 veterans, a substantial proportion of whom are aged ≥ 65 years. Among these older veterans are active long-term BZD users, who were defined as having ≥ 60-day supply of BZD prescriptions dispensed within the past year. Each active long-term user with a BZD prescription released within 200 days before the index date (the date the user was to meet with the prescribing physician) was mailed an EB 2 to 8 weeks in advance of the visit. Excluded from analysis were veterans with a schizophrenia, spinal cord injury, or seizure disorder diagnosis recorded in both their inpatient and outpatient medical records; veterans seen by Palliative Care within the past year; and veterans who died before analysis was completed.
Education Brochure. The EB for VISN 22 (Figure 1, see
Patients. The sample consisted of all veterans identified as meeting the inclusion criteria and being enrolled in VISN 22. The EB was mailed once to veterans on a rolling basis from December 2014 to February 2016. Change in BZD use was analyzed only after 9 to 24 months had passed since the index appointment with the prescribing physician. This period included 12 weeks for BZD taper and then 6 months after taper.
Analysis. For each veteran, monthly mean lorazepam equivalent (LE) was calculated using as many as 12 fills before the index date. Average daily dose of LE was calculated by dividing the sum of LE from all included prescriptions by total number of days between the first fill and the index date. The BZD prescription fills were evaluated after the index date. Veterans who received at least 1 prescription after the index date but then had no BZD prescription activity in VA clinics for 3 consecutive months during the 9-month observation period were recorded as having tapered and then discontinued BZD. Veterans who had no BZD prescription activity in VA clinics after the index date and during the 9-month observation period were recorded as having discontinued BZD without tapering. For veterans who had BZD prescription activity in VA clinics after the index date and during the 9-month observation period, mean LE was calculated by dividing the total LE for BZD prescriptions after the index date by number of days from the first fill after the index date to the date of analysis.
Quality Improvement Project 2
Design. A retrospective cohort analysis using PSM was performed on a subgroup of the QIP-1 sample to evaluate the impact of EB on BZD prescribing in the VA during 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. Veterans in the analysis were active long-term BZD users, had at least 1 BZD prescription released within 200 days before the index date, were aged ≥ 65 years, and had an appointment scheduled with their BZD prescriber within 2 to 8 weeks (Figure 2).
Patients. VISN 22 implemented QIP-2, a real-world application of a modified EMPOWER program, by identifying eligible veterans on a rolling basis from December 2014 to August 2015. All veterans who were identified and sent an EB during this period were included in the case group. The index date was defined as the first of the month the EB was mailed. Veterans with a pending appointment were chosen because the lead time would allow them to receive the EB and prepare to discuss it with the physician during the visit.
A comparator group was drawn from the adjacent VISN 21 catchment area, which encompasses VA facilities and clinics in Hawaii, northern California, and northern Nevada. During the observation period, VISN 21 did not mail any EBs specifically addressing BZD risks. Veterans in the comparator group had an appointment scheduled with their BZD prescribing physician within 4 weeks, were aged ≥ 65 years on the index date (first of the month before the next appointment, coinciding with the date EBs were sent to VISN 22 veterans), were active long-term BZD users, and had at least 1 BZD prescription released within 200 days before the index date. All patients were followed for up to 12 months after the index date, with BZD discontinuation recorded 9 and 12 months after the index date.
Propensity Score Matching
Propensity score (PS) was estimated with logistic regression analysis with treatment as the dependent variable and baseline characteristics as the independent variables.21,22 One-to-one matching on the PS was performed using the nearest neighbor approach without replacements. Independent variables related to outcome but unrelated to EB exposure were selected for PS development.22 These variables included year of birth; male sex; Hispanic ethnicity; annual income; service connection status; region; body mass index; Charlson Comorbidity Index category; total baseline BZD dose; and diagnosis of AIDS, nonmetastatic cancer, metastatic cancer, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), dementia, diabetes mellitus (DM), DM with complications, gastroesophageal reflux disease (GERD), general anxiety disorder (GAD), hemiparaplegia, liver disease (mild), liver disease (moderate to severe), myocardial infarction (MI), Parkinson disease, peptic ulcer disease (PUD), psychosis, renal disease, rheumatoid arthritis (RA), or substance use disorder (SUD).
The EMPOWER cluster randomized controlled trial (RCT) demonstrated the effectiveness of EB exposure in a Canadian population of elderly patients who were long-term BZD users.17 Randomized controlled trials are the gold standard for clinical trials because they can establish causal inference.23-25 Given ethical and practical concerns, however, RCTs cannot be applied to all clinical scenarios. Although EMPOWER is reported to be an effective tool in reducing BZD use in older adults, its application in a real-world, large, integrated health care system remains untested. Observational studies are often conducted as an alternative to RCTs but are subject to selection bias because of their lack of randomization.26 Therefore, robust research methods are needed to generate unbiased estimates of the impact of an intervention on an outcome. Propensity score matching simulates an RCT by balancing the covariates across treatment groups.21,22,27 Observed patient characteristics are used to estimate PS, the probability that treatment will be received. Logistic or probit regression is used to balance the potential confounding covariates between the treatment groups.Once PSs are known, mean treatment effect can be estimated without the mean model.28 In other words, PSM methods can be used to generate an unbiased estimate of the treatment.
Propensity Score Analysis
Baseline characteristics were compared using Student t test (continuous variables) and χ2 test (discrete variables). Results are presented as means and standard deviations (continuous variables) and frequency and percentage (discrete variables).
The main outcome was BZD discontinuation 9 and 12 months after the index date. A postindex lag of 6 months was used to capture any tapering (Figure 2). Discontinuation, defined as 3 consecutive months of no BZD prescription on hand, was measured for 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. An estimate was made of the difference in the proportions of BZD discontinuers who received the EB and BZD discontinuers who did not receive the EB, where mean treatment (risk difference) was presented as the absolute risk difference with a 95% CI. Standard errors and 95% CIs for the risk differences were generated with biased-corrected CIs from 1,000 bootstrap samples.
Sensitivity Analyses
Naïve multivariate logistic regression analysis was performed to evaluate the association between EB exposure and BZD discontinuation while controlling for potential confounders. Results are presented as odds ratios (ORs) and 95% CIs. Confounders identified were the same covariates used to generate the PSs.
Several analyses were performed to test the sensitivity of the methods applied using PSM by changing caliber size while maintaining the nearest neighbor approach without replacement. Linear regression analysis was performed with robust standard errors to estimate the risk difference of BZD discontinuation between EB-exposed and EB-unexposed veterans.
Statistical significance was set at P < .05. All statistical analyses were performed with Stata/SE Version 13 (College Station, TX).
Results
Quality Improvement Project 1
On a rolling basis from December 2014 to February 2016, the EB was mailed once to 3,896 VISN 22 veterans 2 to 8 weeks before a clinic appointment with their BZD prescribing physician.
Quality Improvement Project 2
Of all the VISN 22 and VISN 21 veterans, 24,420 met the inclusion and exclusion criteria. Of these 24,420 veterans, 2,020 (8.3%) were in VISN 22 and received the EB between December 2014 and August 2015 (QIP-1), and 22,400 (91.7%) were in VISN 21 and did not receive the EB.
Naïve Results Before PS Matching. In the naïve analyses, a larger proportion of EB-exposed vs unexposed veterans discontinued BZD; in addition, reductions were 6.6%, 7.4%, and 9.5% larger for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (P < .0001 for all comparisons; Table 2).
After controlling for potential confounders, the naïve logistic regression analyses found EB exposure was significantly associated with 44%, 32%, and 42% increases in the odds of BZD discontinuation for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (Table 3).
Propensity Score Matching. Before matching, there were significant differences in baseline characteristics of veterans who met the inclusion and exclusion criteria, with few exceptions (eAppendices 2 and 3, ).
Propensity Score Matching Results. Inspection of PSs revealed good coverage across treatment groups on a histogram plot and a kernel density plot (eAppendices 5 and 6).
Discussion
This QIP was the first to evaluate the impact of an EMPOWER-modeled DTC EB in a large, integrated health care system in the U.S. It was also the first to demonstrate potential benefits of a DTC EB designed for older veterans who are long-term BZD users. In this QIP, which mailed the EB to 3,896 veterans, 1,847 (47.4%) decreased their BZD dose, 458 (11.7%) tapered and then discontinued BZD, and 455 (11.7%) immediately discontinued BZD. The total percentage of veterans who discontinued BZD (23.4%; 913/3,896) was similar to the 27% reported in the EMPOWER trial.17 However, the risk difference between the 1,316 EB-exposed VISN 22 veterans (QIP-1) and the 1,316 EB-unexposed VISN 21 veterans in this QIP was significantly lower than the 23% risk difference in EMPOWER (though it still demonstrated a significantly larger reduction for EB-exposed veterans).17
Given this inclusion of all qualifying veterans from the catchment area studied in this QIP, and given the ethical and practical concerns, an RCT was not possible. Therefore, PSM methods were used to balance the covariates across treatment groups and thereby simulate an RCT.21,22,27 With use of the PSM approach, findings from the descriptive analysis were confirmed and potential selection bias reduced.
Study Limitations
The less robust risk difference found in this QIP has several possible explanations. The authors’ use of a DTC EB coincided with a national VA effort to reduce older veterans’ use of BZDs and other inappropriate medications. For instance, during the study period, academic detailing was being implemented to reduce use of BZDs, particularly in combination with opioids, across VHA facilities and clinics. (Academic detailing is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20) However, QIP-2 results and PS analysis of a subgroup of the original sample suggest that EB-exposed veterans were significantly more likely than were their unexposed counterparts were to discontinue BZD. To an extent, this analysis controlled for these other efforts to reduce BZD use in VHA clinics and can be considered a study strength.
Another limitation is the study design, which lacked a control group and did not consider the possibility that some facility or clinic physicians might influence others. Although the region variable was controlled for in PSM, the authors did not capture facility characteristics, including frequency of prescribing BZD and use of a protocol for enforcing the Beers criteria. Such confounders might have influenced outcomes. Unlike the EMPOWER trial,17 this QIP did not assess or exclude cognitively impaired veterans. It is reasonable to assume that these veterans might not understand some EB messages and consequently might fail to engage their physicians. Failure to initiate discussion with a physician would attenuate the impact of the EB.
Study Strengths
A strength of this QIP was its use of a DTC EB in a large, regional sample of older veterans in a real-world clinical setting. In addition, the study group (EB-exposed veterans) and the comparator group (EB-unexposed veterans) were from similar geographic areas (primarily California and Nevada).
Conclusion
Results of this study suggest that a DTC EB, designed to reduce BZD use among older veterans, was effective in helping patients lower their BZD dose and discontinue BZD. The likelihood of discontinuing BZD 9 and 12 months after the index date was significantly higher for veterans who received an EB modeled on the EMPOWER educational brochure than for a comparator group of veterans who did not receive the EB and were receiving care during the same observation period. In the future, it would be beneficial to use a design that controls for physician exposure to academic detailing focused on BZD reduction and that accounts for the cluster effects of facility practice. Despite these limitations, this QIP is the first real-world empirical example of using an EMPOWER-modeled DTC EB to decrease BZD use among older veterans. Furthermore, these results suggest that a DTC EB can be used to target other high-risk prescription drugs, such as opioids, particularly if alternative treatment options can be provided.
Acknowledgments
Dr. Hauser thanks Cathy, Anika, Katia, and Max Hauser, and Alba and Kevin Quinlan, for their support. In memory of Jirina Hauser, who died on Mother’s Day, May 14, 2017, at the age of 100.
Benzodiazepines (BZDs) are among the most commonly prescribed medications. A recent study found that in 2008, more than 5% of Americans used a BZD, and the percentage was almost 9% among Americans aged ≥ 65 years.1,2 Among veterans, BZD use is even higher, in part because of the high prevalence of posttraumatic stress disorder (PTSD). One study found that more than 30% of veterans with PTSD received at least 1 BZD prescription.3 The risks associated with BZD treatment for PTSD are compounded by concurrent use of other sedatives and opioids prescribed for co-occurring chronic pain and insomnia.3
Older adults metabolize long-acting BZDs more slowly and generally have an increased sensitivity to the adverse effects (AEs) of all BZDs.4 In older adults, BZD use has been associated with cognitive decline, dementia, falls and consequent fractures, and adverse respiratory outcomes.5-12 The risk of most but not all of these AEs was increased with higher BZD dose or long-term BZD use, which this quality improvement project (QIP) defines as having at least a 60-day supply of BZD prescriptions dispensed within the past year.
Long-term BZD use increases with age. One study found that, among patients receiving a BZD, the rate of long-term BZD use was more than double in older adults (31.4%) than it was in adults aged between 18 and 35 years (14.7%).2 For these reasons, the 2012 Beers criteria of the American Geriatrics Society recommend avoiding all types of BZDs in the treatment of insomnia, agitation, or delirium in patients aged > 65 years.13 Despite this recommendation, the prevalence of BZD use in older adults remains high.14
Some innovative approaches have been developed to address the inappropriate use, including overuse and misuse, of BZDs in older adults.15 In one approach, direct-to-consumer (DTC) information is used to empower patients to collaborate with their physician to manage their health. Results from several studies suggest that providing older patients with information on BZD risks and benefits increases patient–physician interaction and thereby decreases inappropriate BZD use and improves health outcomes.4,16,17 One study found that perceptions of BZD risks increased 1 week after exposure to a DTC educational brochure (EB), with intention to discuss BZD discontinuation with their physician higher for patients who received the EB than it was for those who did not (83.1% vs 44.3%; P < .0001).16 The EMPOWER (Eliminating Medications Through Patient Ownership of End Results) cluster randomized controlled trial assessed the effectiveness of a DTC EB focused on BZD risks in older adults.17 In that seminal study, patients who received a DTC EB were more likely than were comparison patients to discontinue BZD within 6 months (27% vs 5%; risk difference, 23%; 95% CI, 14%-32%).
The Veterans Integrated Systems Network (VISN) 22 Academic Detailing Program is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20 With BZD use among older veterans remaining high, the VISN 22 program initiated a clinical QIP modeled on the EMPOWER trial. Veterans in VISN 22 received the DTC EB, which included information on BZD risks and encouraged them to discuss their BZD treatment with their health care provider. VISN 22 was the first VISN in the VHA to implement the EMPOWER protocol.
As this was a QIP, all eligible veterans in VISN 22 were mailed the DTC EB, thus making it difficult to estimate the impact of the EB on BZD discontinuation in this VISN. Therefore, DTC EB efficacy was estimated by comparing BZD discontinuation between VISN 22 and VISN 21, an adjacent VISN that did not mail the DTC EB.
Methods
Two QIPs were undertaken to determine the impact of DTC EB on BZD use in older veterans in the VHA.
Quality Improvement Project 1
Design. A retrospective cohort analysis was performed. The VISN 22 catchment area, which encompasses VA facilities and clinics in southern California and southern Nevada, serves about 500,000 veterans, a substantial proportion of whom are aged ≥ 65 years. Among these older veterans are active long-term BZD users, who were defined as having ≥ 60-day supply of BZD prescriptions dispensed within the past year. Each active long-term user with a BZD prescription released within 200 days before the index date (the date the user was to meet with the prescribing physician) was mailed an EB 2 to 8 weeks in advance of the visit. Excluded from analysis were veterans with a schizophrenia, spinal cord injury, or seizure disorder diagnosis recorded in both their inpatient and outpatient medical records; veterans seen by Palliative Care within the past year; and veterans who died before analysis was completed.
Education Brochure. The EB for VISN 22 (Figure 1, see
Patients. The sample consisted of all veterans identified as meeting the inclusion criteria and being enrolled in VISN 22. The EB was mailed once to veterans on a rolling basis from December 2014 to February 2016. Change in BZD use was analyzed only after 9 to 24 months had passed since the index appointment with the prescribing physician. This period included 12 weeks for BZD taper and then 6 months after taper.
Analysis. For each veteran, monthly mean lorazepam equivalent (LE) was calculated using as many as 12 fills before the index date. Average daily dose of LE was calculated by dividing the sum of LE from all included prescriptions by total number of days between the first fill and the index date. The BZD prescription fills were evaluated after the index date. Veterans who received at least 1 prescription after the index date but then had no BZD prescription activity in VA clinics for 3 consecutive months during the 9-month observation period were recorded as having tapered and then discontinued BZD. Veterans who had no BZD prescription activity in VA clinics after the index date and during the 9-month observation period were recorded as having discontinued BZD without tapering. For veterans who had BZD prescription activity in VA clinics after the index date and during the 9-month observation period, mean LE was calculated by dividing the total LE for BZD prescriptions after the index date by number of days from the first fill after the index date to the date of analysis.
Quality Improvement Project 2
Design. A retrospective cohort analysis using PSM was performed on a subgroup of the QIP-1 sample to evaluate the impact of EB on BZD prescribing in the VA during 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. Veterans in the analysis were active long-term BZD users, had at least 1 BZD prescription released within 200 days before the index date, were aged ≥ 65 years, and had an appointment scheduled with their BZD prescriber within 2 to 8 weeks (Figure 2).
Patients. VISN 22 implemented QIP-2, a real-world application of a modified EMPOWER program, by identifying eligible veterans on a rolling basis from December 2014 to August 2015. All veterans who were identified and sent an EB during this period were included in the case group. The index date was defined as the first of the month the EB was mailed. Veterans with a pending appointment were chosen because the lead time would allow them to receive the EB and prepare to discuss it with the physician during the visit.
A comparator group was drawn from the adjacent VISN 21 catchment area, which encompasses VA facilities and clinics in Hawaii, northern California, and northern Nevada. During the observation period, VISN 21 did not mail any EBs specifically addressing BZD risks. Veterans in the comparator group had an appointment scheduled with their BZD prescribing physician within 4 weeks, were aged ≥ 65 years on the index date (first of the month before the next appointment, coinciding with the date EBs were sent to VISN 22 veterans), were active long-term BZD users, and had at least 1 BZD prescription released within 200 days before the index date. All patients were followed for up to 12 months after the index date, with BZD discontinuation recorded 9 and 12 months after the index date.
Propensity Score Matching
Propensity score (PS) was estimated with logistic regression analysis with treatment as the dependent variable and baseline characteristics as the independent variables.21,22 One-to-one matching on the PS was performed using the nearest neighbor approach without replacements. Independent variables related to outcome but unrelated to EB exposure were selected for PS development.22 These variables included year of birth; male sex; Hispanic ethnicity; annual income; service connection status; region; body mass index; Charlson Comorbidity Index category; total baseline BZD dose; and diagnosis of AIDS, nonmetastatic cancer, metastatic cancer, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), dementia, diabetes mellitus (DM), DM with complications, gastroesophageal reflux disease (GERD), general anxiety disorder (GAD), hemiparaplegia, liver disease (mild), liver disease (moderate to severe), myocardial infarction (MI), Parkinson disease, peptic ulcer disease (PUD), psychosis, renal disease, rheumatoid arthritis (RA), or substance use disorder (SUD).
The EMPOWER cluster randomized controlled trial (RCT) demonstrated the effectiveness of EB exposure in a Canadian population of elderly patients who were long-term BZD users.17 Randomized controlled trials are the gold standard for clinical trials because they can establish causal inference.23-25 Given ethical and practical concerns, however, RCTs cannot be applied to all clinical scenarios. Although EMPOWER is reported to be an effective tool in reducing BZD use in older adults, its application in a real-world, large, integrated health care system remains untested. Observational studies are often conducted as an alternative to RCTs but are subject to selection bias because of their lack of randomization.26 Therefore, robust research methods are needed to generate unbiased estimates of the impact of an intervention on an outcome. Propensity score matching simulates an RCT by balancing the covariates across treatment groups.21,22,27 Observed patient characteristics are used to estimate PS, the probability that treatment will be received. Logistic or probit regression is used to balance the potential confounding covariates between the treatment groups.Once PSs are known, mean treatment effect can be estimated without the mean model.28 In other words, PSM methods can be used to generate an unbiased estimate of the treatment.
Propensity Score Analysis
Baseline characteristics were compared using Student t test (continuous variables) and χ2 test (discrete variables). Results are presented as means and standard deviations (continuous variables) and frequency and percentage (discrete variables).
The main outcome was BZD discontinuation 9 and 12 months after the index date. A postindex lag of 6 months was used to capture any tapering (Figure 2). Discontinuation, defined as 3 consecutive months of no BZD prescription on hand, was measured for 2 periods: 6 to 9 months and 6 to 12 months after the index date. A secondary outcome was discontinuation 1 to 12 months after the index date. An estimate was made of the difference in the proportions of BZD discontinuers who received the EB and BZD discontinuers who did not receive the EB, where mean treatment (risk difference) was presented as the absolute risk difference with a 95% CI. Standard errors and 95% CIs for the risk differences were generated with biased-corrected CIs from 1,000 bootstrap samples.
Sensitivity Analyses
Naïve multivariate logistic regression analysis was performed to evaluate the association between EB exposure and BZD discontinuation while controlling for potential confounders. Results are presented as odds ratios (ORs) and 95% CIs. Confounders identified were the same covariates used to generate the PSs.
Several analyses were performed to test the sensitivity of the methods applied using PSM by changing caliber size while maintaining the nearest neighbor approach without replacement. Linear regression analysis was performed with robust standard errors to estimate the risk difference of BZD discontinuation between EB-exposed and EB-unexposed veterans.
Statistical significance was set at P < .05. All statistical analyses were performed with Stata/SE Version 13 (College Station, TX).
Results
Quality Improvement Project 1
On a rolling basis from December 2014 to February 2016, the EB was mailed once to 3,896 VISN 22 veterans 2 to 8 weeks before a clinic appointment with their BZD prescribing physician.
Quality Improvement Project 2
Of all the VISN 22 and VISN 21 veterans, 24,420 met the inclusion and exclusion criteria. Of these 24,420 veterans, 2,020 (8.3%) were in VISN 22 and received the EB between December 2014 and August 2015 (QIP-1), and 22,400 (91.7%) were in VISN 21 and did not receive the EB.
Naïve Results Before PS Matching. In the naïve analyses, a larger proportion of EB-exposed vs unexposed veterans discontinued BZD; in addition, reductions were 6.6%, 7.4%, and 9.5% larger for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (P < .0001 for all comparisons; Table 2).
After controlling for potential confounders, the naïve logistic regression analyses found EB exposure was significantly associated with 44%, 32%, and 42% increases in the odds of BZD discontinuation for 6 to 9 months, 6 to 12 months, and 1 to 12 months after the index date, respectively (Table 3).
Propensity Score Matching. Before matching, there were significant differences in baseline characteristics of veterans who met the inclusion and exclusion criteria, with few exceptions (eAppendices 2 and 3, ).
Propensity Score Matching Results. Inspection of PSs revealed good coverage across treatment groups on a histogram plot and a kernel density plot (eAppendices 5 and 6).
Discussion
This QIP was the first to evaluate the impact of an EMPOWER-modeled DTC EB in a large, integrated health care system in the U.S. It was also the first to demonstrate potential benefits of a DTC EB designed for older veterans who are long-term BZD users. In this QIP, which mailed the EB to 3,896 veterans, 1,847 (47.4%) decreased their BZD dose, 458 (11.7%) tapered and then discontinued BZD, and 455 (11.7%) immediately discontinued BZD. The total percentage of veterans who discontinued BZD (23.4%; 913/3,896) was similar to the 27% reported in the EMPOWER trial.17 However, the risk difference between the 1,316 EB-exposed VISN 22 veterans (QIP-1) and the 1,316 EB-unexposed VISN 21 veterans in this QIP was significantly lower than the 23% risk difference in EMPOWER (though it still demonstrated a significantly larger reduction for EB-exposed veterans).17
Given this inclusion of all qualifying veterans from the catchment area studied in this QIP, and given the ethical and practical concerns, an RCT was not possible. Therefore, PSM methods were used to balance the covariates across treatment groups and thereby simulate an RCT.21,22,27 With use of the PSM approach, findings from the descriptive analysis were confirmed and potential selection bias reduced.
Study Limitations
The less robust risk difference found in this QIP has several possible explanations. The authors’ use of a DTC EB coincided with a national VA effort to reduce older veterans’ use of BZDs and other inappropriate medications. For instance, during the study period, academic detailing was being implemented to reduce use of BZDs, particularly in combination with opioids, across VHA facilities and clinics. (Academic detailing is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote physicians’ safety initiatives and align prescribing behavior with best practices.18-20) However, QIP-2 results and PS analysis of a subgroup of the original sample suggest that EB-exposed veterans were significantly more likely than were their unexposed counterparts were to discontinue BZD. To an extent, this analysis controlled for these other efforts to reduce BZD use in VHA clinics and can be considered a study strength.
Another limitation is the study design, which lacked a control group and did not consider the possibility that some facility or clinic physicians might influence others. Although the region variable was controlled for in PSM, the authors did not capture facility characteristics, including frequency of prescribing BZD and use of a protocol for enforcing the Beers criteria. Such confounders might have influenced outcomes. Unlike the EMPOWER trial,17 this QIP did not assess or exclude cognitively impaired veterans. It is reasonable to assume that these veterans might not understand some EB messages and consequently might fail to engage their physicians. Failure to initiate discussion with a physician would attenuate the impact of the EB.
Study Strengths
A strength of this QIP was its use of a DTC EB in a large, regional sample of older veterans in a real-world clinical setting. In addition, the study group (EB-exposed veterans) and the comparator group (EB-unexposed veterans) were from similar geographic areas (primarily California and Nevada).
Conclusion
Results of this study suggest that a DTC EB, designed to reduce BZD use among older veterans, was effective in helping patients lower their BZD dose and discontinue BZD. The likelihood of discontinuing BZD 9 and 12 months after the index date was significantly higher for veterans who received an EB modeled on the EMPOWER educational brochure than for a comparator group of veterans who did not receive the EB and were receiving care during the same observation period. In the future, it would be beneficial to use a design that controls for physician exposure to academic detailing focused on BZD reduction and that accounts for the cluster effects of facility practice. Despite these limitations, this QIP is the first real-world empirical example of using an EMPOWER-modeled DTC EB to decrease BZD use among older veterans. Furthermore, these results suggest that a DTC EB can be used to target other high-risk prescription drugs, such as opioids, particularly if alternative treatment options can be provided.
Acknowledgments
Dr. Hauser thanks Cathy, Anika, Katia, and Max Hauser, and Alba and Kevin Quinlan, for their support. In memory of Jirina Hauser, who died on Mother’s Day, May 14, 2017, at the age of 100.
1. Dell’osso B, Lader M. Do benzodiazepines still deserve a major role in the treatment of psychiatric disorders? A critical reappraisal. Eur Psychiatry. 2013;28(1):7-20.
2. Olfson M, King M, Schoenbaum M. Benzodiazepine use in the United States. JAMA Psychiatry. 2015;72(2):136-142.
3. Bernardy NC, Lund BC, Alexander B, Friedman MJ. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
4. Roberts KJ. Patient empowerment in the United States: a critical commentary. Health Expect. 1999;2(2):82-92.
5. Paterniti S, Dufouil C, Alpérovitch A. Long-term benzodiazepine use and cognitive decline in the elderly: the Epidemiology of Vascular Aging Study. J Clin Psychopharmacol. 2002;22(3):285-293.
6. van der Hooft CS, Schoofs MW, Ziere G, et al. Inappropriate benzodiazepine use in older adults and the risk of fracture. Br J Clin Pharmacol. 2008;66(2):276-282.
7. Zint K, Haefeli WE, Glynn RJ, Mogun H, Avorn J, Stürmer T. Impact of drug interactions, dosage, and duration of therapy on the risk of hip fracture associated with benzodiazepine use in older adults. Pharmacoepidemiol Drug Saf. 2010;19(12):1248-1255.
8. Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc. 2011;59(10):1883-1890.
9. de Gage SB, Bégaud B, Bazin F, et al. Benzodiazepine use and risk of dementia: prospective population based study. BMJ. 2012;345:e6231
10. Tannenbaum C, Paquette A, Hilmer S, Holroyd-Leduc J, Carnahan R. A systematic review of amnestic and non-amnestic mild cognitive impairment induced by anticholinergic, antihistamine, GABAergic and opioid drugs. Drugs Aging. 2012;29(8):639-658.
11. Vozoris NT, Fischer HD, Wang X, et al. Benzodiazepine drug use and adverse respiratory outcomes among older adults with chronic obstructive pulmonary disease. Eur Respir J. 2014;44(2):332-340.
12. Gomm W, von Holt K, Thomé F, et al. Regular benzodiazepine and z-substance use and risk of dementia: an analysis of German claims data. J Alzheimers Dis. 2016;54(2):801-808.
13. American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60(4):616-631.
14. National Institutes of Health. Despite risks, benzodiazepine use highest in older people. https://www.nih.gov/news-events/news-releases/despite-risks-benzodiaze pine-use-highest-older-people. Published December 17, 2014. Accessed July 31, 2018.
15. Airagnes G, Pelissolo A, Lavallée M, Flament M, Limosin F. Benzodiazepine misuse in the elderly: risk factors, consequences, and management. Curr Psychiatry Rep. 2016;18(10):89.
16. Martin P, Tamblyn R, Ahmed S, Tannenbaum C. A drug education tool developed for older adults changes knowledge, beliefs and risk perceptions about inappropriate benzodiazepine prescriptions in the elderly. Patient Educ Couns. 2013;92(1):81-87.
17. Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898.
18. Soumerai SB, Avorn J. Principles of educational outreach (‘academic detailing’) to improve clinical decision making. JAMA. 1990;263(4):549-556.
19. Fischer MA, Avorn J. Academic detailing can play a key role in assessing and implementing comparative effectiveness research findings. Health Aff (Millwood). 2012;31(10):2206-2212.
20. Wells DL, Popish S, Kay C, Torrise V, Christopher ML. VA Academic Detailing Service: implementation and lessons learned. Fed Pract. 2016;33(5):38-42.
21. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424.
22. Brookhart MA, Schneeweiss S, Rothman KJ, Glynn RJ, Avorn J, Stürmer T. Variable selection for propensity score models. Am J Epidemiol. 2006;163(12):1149-1156.
23. Rubin DB. Estimating causal effects of treatments in randomized and nonrandomized studies. J Ed Psych. 1974;66(5):688-701.
24. Greenland S. An introduction to instrumental variables for epidemiologists. Int J Epidemiol. 2000;29(4):722-729.
25. Cartwright N. What are randomized controlled trials good for? Philos Stud. 2010;147(1):59.
26. Kleinbaum DG, Morgenstern H, Kupper LL. Selection bias in epidemiologic studies. Am J Epidemiol. 1981;113(4):452-463.
27. Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41-55.
28. Pirracchio R, Carone M, Rigon MR, Caruana E, Mebazaa A, Chevret S. Propensity score estimators for the average treatment effect and the average treatment effect on the treated may yield very different estimates. Stat Methods Med Res. 2016;25(5):1938-1954.
1. Dell’osso B, Lader M. Do benzodiazepines still deserve a major role in the treatment of psychiatric disorders? A critical reappraisal. Eur Psychiatry. 2013;28(1):7-20.
2. Olfson M, King M, Schoenbaum M. Benzodiazepine use in the United States. JAMA Psychiatry. 2015;72(2):136-142.
3. Bernardy NC, Lund BC, Alexander B, Friedman MJ. Increased polysedative use in veterans with posttraumatic stress disorder. Pain Med. 2014;15(7):1083-1090.
4. Roberts KJ. Patient empowerment in the United States: a critical commentary. Health Expect. 1999;2(2):82-92.
5. Paterniti S, Dufouil C, Alpérovitch A. Long-term benzodiazepine use and cognitive decline in the elderly: the Epidemiology of Vascular Aging Study. J Clin Psychopharmacol. 2002;22(3):285-293.
6. van der Hooft CS, Schoofs MW, Ziere G, et al. Inappropriate benzodiazepine use in older adults and the risk of fracture. Br J Clin Pharmacol. 2008;66(2):276-282.
7. Zint K, Haefeli WE, Glynn RJ, Mogun H, Avorn J, Stürmer T. Impact of drug interactions, dosage, and duration of therapy on the risk of hip fracture associated with benzodiazepine use in older adults. Pharmacoepidemiol Drug Saf. 2010;19(12):1248-1255.
8. Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc. 2011;59(10):1883-1890.
9. de Gage SB, Bégaud B, Bazin F, et al. Benzodiazepine use and risk of dementia: prospective population based study. BMJ. 2012;345:e6231
10. Tannenbaum C, Paquette A, Hilmer S, Holroyd-Leduc J, Carnahan R. A systematic review of amnestic and non-amnestic mild cognitive impairment induced by anticholinergic, antihistamine, GABAergic and opioid drugs. Drugs Aging. 2012;29(8):639-658.
11. Vozoris NT, Fischer HD, Wang X, et al. Benzodiazepine drug use and adverse respiratory outcomes among older adults with chronic obstructive pulmonary disease. Eur Respir J. 2014;44(2):332-340.
12. Gomm W, von Holt K, Thomé F, et al. Regular benzodiazepine and z-substance use and risk of dementia: an analysis of German claims data. J Alzheimers Dis. 2016;54(2):801-808.
13. American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60(4):616-631.
14. National Institutes of Health. Despite risks, benzodiazepine use highest in older people. https://www.nih.gov/news-events/news-releases/despite-risks-benzodiaze pine-use-highest-older-people. Published December 17, 2014. Accessed July 31, 2018.
15. Airagnes G, Pelissolo A, Lavallée M, Flament M, Limosin F. Benzodiazepine misuse in the elderly: risk factors, consequences, and management. Curr Psychiatry Rep. 2016;18(10):89.
16. Martin P, Tamblyn R, Ahmed S, Tannenbaum C. A drug education tool developed for older adults changes knowledge, beliefs and risk perceptions about inappropriate benzodiazepine prescriptions in the elderly. Patient Educ Couns. 2013;92(1):81-87.
17. Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898.
18. Soumerai SB, Avorn J. Principles of educational outreach (‘academic detailing’) to improve clinical decision making. JAMA. 1990;263(4):549-556.
19. Fischer MA, Avorn J. Academic detailing can play a key role in assessing and implementing comparative effectiveness research findings. Health Aff (Millwood). 2012;31(10):2206-2212.
20. Wells DL, Popish S, Kay C, Torrise V, Christopher ML. VA Academic Detailing Service: implementation and lessons learned. Fed Pract. 2016;33(5):38-42.
21. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424.
22. Brookhart MA, Schneeweiss S, Rothman KJ, Glynn RJ, Avorn J, Stürmer T. Variable selection for propensity score models. Am J Epidemiol. 2006;163(12):1149-1156.
23. Rubin DB. Estimating causal effects of treatments in randomized and nonrandomized studies. J Ed Psych. 1974;66(5):688-701.
24. Greenland S. An introduction to instrumental variables for epidemiologists. Int J Epidemiol. 2000;29(4):722-729.
25. Cartwright N. What are randomized controlled trials good for? Philos Stud. 2010;147(1):59.
26. Kleinbaum DG, Morgenstern H, Kupper LL. Selection bias in epidemiologic studies. Am J Epidemiol. 1981;113(4):452-463.
27. Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41-55.
28. Pirracchio R, Carone M, Rigon MR, Caruana E, Mebazaa A, Chevret S. Propensity score estimators for the average treatment effect and the average treatment effect on the treated may yield very different estimates. Stat Methods Med Res. 2016;25(5):1938-1954.
Focus on symptoms over stories is detrimental to separated families
The horrors faced by migrant families forced to separate under the new U.S. “zero tolerance” policy continue to unfold. Tragic emblems of this policy include tapes of crying children and the reported suicide of a father who had been separated from his children.
A federal judge had issued an injunction requiring the reunification of thousands of families by July 26. Despite that deadline, hundreds of adults are no longer in the United States, and hundreds of children are scattered in shelters across the country.
In response to those events, mental health and medical organizations have released powerful statements. The American Psychological Association stated: “The administration’s policy ... is not only needless and cruel, it threatens the mental and physical health of both the children and their caregivers.” The American Medical Association issued a call asserting that separating children from their parents “will do great harm” and “create negative health impacts that will last an individual’s entire lifespan.” Meanwhile, the American Psychiatric Association’s president, Altha J. Stewart, MD, released a statement affirming that “any forced separation is highly stressful for children and can cause lifelong trauma, as well as an increased risk of other mental illnesses, such as depression, anxiety, and posttraumatic stress disorder.”
As forensic experts who testify about the mental well-being of immigration detainees, we applaud those powerful and unambiguous messages from the leaders in our fields. Yet, their statements also underscore the limitations of our diagnostic models: Our field is caught in the difficult position of either applying ill-fitting diagnostic labels or overpathologizing a normal reaction to horrific circumstances. While not applying diagnoses potentially minimizes the enormous psychological burden of separation, diagnosing depression or PTSD as catchalls for suffering incorrectly defines the experience of many survivors of ongoing trauma.
Currently, most providers, in trying to communicate the effects of ongoing trauma, rely on the diagnoses of depression or PTSD. Both of these diagnoses, however, are problematic. The diagnosis of major depressive disorder, for example, is useful in communicating a loss of hope, and the inability to enjoy pleasurable things. However, depression is an episodic illness, often part of a larger chronic disorder.1 Depression often has a genetic-hereditary component. On the other hand, children suffering from childhood traumas often present lifelong and wide-ranging problems, which may be triggered by reminders but are not episodic. For example, children experiencing parental separation have difficulty forming attachments, which, in turn, leads to subsequent difficulty forming meaningful interpersonal relationships.
The diagnosis of PTSD is useful in communicating a myriad of possible symptoms, which may accompany the trauma. However, PTSD implies a traumatic event as described in criteria A of the DSM-5: “exposure to actual or threatened death, serious injury, or sexual violence.” As such, PTSD poorly encompasses the wide array of smaller yet repetitive traumas experienced by victims of ongoing trauma, such as those youth separated from their parents at the U.S. border. Furthermore, PTSD is a disorder with specific symptoms that, based on a vast body of research,2,3 inadequately describes the multitude of interpersonal, psychological, and physical consequences associated with the type of trauma caused by family separations.
Our understanding of the long-term sequelae of childhood trauma has been greatly influenced by the adverse childhood experiences (ACE) study. The ACE study, one of the largest investigations ever conducted to assess associations between childhood maltreatment and later-life health and well-being, collected the life histories of more than 17,000 patients in a collaborative effort between the Centers for Disease Control and Prevention and Kaiser Permanente’s Health Appraisal Clinic.
The ACE study identified 10 forms of childhood trauma, including: abuse, neglect, abandonment, household dysfunction, and exposure to violence, that were strongly associated with negative psychological outcomes such as depression, suicide attempts, and engagement in high-risk behaviors, as well as significant medical consequences, including higher incidence of heart disease, diabetes, and stroke. Ultimately, having four or more ACEs was associated with early death.
In response to the emerging body of research on childhood trauma, various terms, including complex trauma, type-II trauma, and complex PTSD, have entered our professional lexicon as a means of communicating the wide-ranging consequences of developmental trauma. On the one hand, the less defined and rigid nature of these terms permits mental health providers to develop a rich narrative of a patient’s background, encompassing the patient’s behavior, character, and symptoms. However, the absence of formal terminology also has its drawbacks: Courts and juries have grown accustomed to diagnoses, labels, and syndromes. Most forensic mental health providers who testify about developmental trauma in court can predict the question: “So doctor, you are saying that the individual’s presentation is not severe enough to be considered PTSD, am I correct?” Disorders justify treatment, can explain disability, and warrant empathy; concomitantly, “complex trauma” runs the risk of being considered an academic explanation for trauma victims’ lifelong problems, rather than a societal failure that merits care.
Recognizing the limitations of our current diagnoses, the forthcoming update to the International Classification of Diseases (ICD-11) will add a new category: complex PTSD. The ICD-11 will attempt to widen the concept of trauma to include “conditions of prolonged adversity, in the form of sustained, repeated, or multiple forms of traumatic exposure.” Trauma exposure examples include genocide campaigns, childhood sexual abuse, child soldiering, severe domestic violence, torture, or slavery. The ICD-11 also expands our understanding of the consequences of trauma to include “affective dysregulation,” “negative self-concept,” and “disturbances in relationships” as part of a concept called “disturbances in self-organization.” Those are important steps in acknowledging the consequences of different forms of trauma as well as noticing a richer array of damages from those incidents.4
While the World Health Organization’s latest iteration of the ICD takes an important step in widening the scope of our diagnostic tools, we are cognizant that our field’s obsessional search for diagnoses, labels, and nomenclature reinforces a detrimental focus on symptoms over stories. However, as forensic mental health providers, we also are keenly aware that a failure to adopt common definitions impedes forensic evaluations, patient advocacy, public policy, and most importantly, patient care.
In the end, we have trained society to understand pathology through narrow lenses, and therefore, . So, despite the limitations of labels, let’s be encouraged by the World Health Organization’s efforts and continue in that direction.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Lehman is a licensed clinical and forensic psychologist in San Diego. Her practice consists of conducting forensic psychological evaluations for the courts with children, adolescents, and adults. Dr. Lehman has been qualified as an expert witness in California as well as in the federal courts. She previously was a supervisor at Sharper Future, a forensic rehabilitation program, and previously served as an adjunct faculty member at Alliant International University, San Diego. Dr. Lehman can be reached at fran@lehmanforensicpsych.com.
References
1. Am J Psychiatry. 2000 Sep;157(9):1501-4.
2. Psychiatr Ann. 2005;35(5):390-8.
3. Psychiatr Ann. 2005;35(5):401-8.
4. Eur J Psychotraumatol. 2018 Jan 15. doi: 10.1080/20008198.2017.1418103.
The horrors faced by migrant families forced to separate under the new U.S. “zero tolerance” policy continue to unfold. Tragic emblems of this policy include tapes of crying children and the reported suicide of a father who had been separated from his children.
A federal judge had issued an injunction requiring the reunification of thousands of families by July 26. Despite that deadline, hundreds of adults are no longer in the United States, and hundreds of children are scattered in shelters across the country.
In response to those events, mental health and medical organizations have released powerful statements. The American Psychological Association stated: “The administration’s policy ... is not only needless and cruel, it threatens the mental and physical health of both the children and their caregivers.” The American Medical Association issued a call asserting that separating children from their parents “will do great harm” and “create negative health impacts that will last an individual’s entire lifespan.” Meanwhile, the American Psychiatric Association’s president, Altha J. Stewart, MD, released a statement affirming that “any forced separation is highly stressful for children and can cause lifelong trauma, as well as an increased risk of other mental illnesses, such as depression, anxiety, and posttraumatic stress disorder.”
As forensic experts who testify about the mental well-being of immigration detainees, we applaud those powerful and unambiguous messages from the leaders in our fields. Yet, their statements also underscore the limitations of our diagnostic models: Our field is caught in the difficult position of either applying ill-fitting diagnostic labels or overpathologizing a normal reaction to horrific circumstances. While not applying diagnoses potentially minimizes the enormous psychological burden of separation, diagnosing depression or PTSD as catchalls for suffering incorrectly defines the experience of many survivors of ongoing trauma.
Currently, most providers, in trying to communicate the effects of ongoing trauma, rely on the diagnoses of depression or PTSD. Both of these diagnoses, however, are problematic. The diagnosis of major depressive disorder, for example, is useful in communicating a loss of hope, and the inability to enjoy pleasurable things. However, depression is an episodic illness, often part of a larger chronic disorder.1 Depression often has a genetic-hereditary component. On the other hand, children suffering from childhood traumas often present lifelong and wide-ranging problems, which may be triggered by reminders but are not episodic. For example, children experiencing parental separation have difficulty forming attachments, which, in turn, leads to subsequent difficulty forming meaningful interpersonal relationships.
The diagnosis of PTSD is useful in communicating a myriad of possible symptoms, which may accompany the trauma. However, PTSD implies a traumatic event as described in criteria A of the DSM-5: “exposure to actual or threatened death, serious injury, or sexual violence.” As such, PTSD poorly encompasses the wide array of smaller yet repetitive traumas experienced by victims of ongoing trauma, such as those youth separated from their parents at the U.S. border. Furthermore, PTSD is a disorder with specific symptoms that, based on a vast body of research,2,3 inadequately describes the multitude of interpersonal, psychological, and physical consequences associated with the type of trauma caused by family separations.
Our understanding of the long-term sequelae of childhood trauma has been greatly influenced by the adverse childhood experiences (ACE) study. The ACE study, one of the largest investigations ever conducted to assess associations between childhood maltreatment and later-life health and well-being, collected the life histories of more than 17,000 patients in a collaborative effort between the Centers for Disease Control and Prevention and Kaiser Permanente’s Health Appraisal Clinic.
The ACE study identified 10 forms of childhood trauma, including: abuse, neglect, abandonment, household dysfunction, and exposure to violence, that were strongly associated with negative psychological outcomes such as depression, suicide attempts, and engagement in high-risk behaviors, as well as significant medical consequences, including higher incidence of heart disease, diabetes, and stroke. Ultimately, having four or more ACEs was associated with early death.
In response to the emerging body of research on childhood trauma, various terms, including complex trauma, type-II trauma, and complex PTSD, have entered our professional lexicon as a means of communicating the wide-ranging consequences of developmental trauma. On the one hand, the less defined and rigid nature of these terms permits mental health providers to develop a rich narrative of a patient’s background, encompassing the patient’s behavior, character, and symptoms. However, the absence of formal terminology also has its drawbacks: Courts and juries have grown accustomed to diagnoses, labels, and syndromes. Most forensic mental health providers who testify about developmental trauma in court can predict the question: “So doctor, you are saying that the individual’s presentation is not severe enough to be considered PTSD, am I correct?” Disorders justify treatment, can explain disability, and warrant empathy; concomitantly, “complex trauma” runs the risk of being considered an academic explanation for trauma victims’ lifelong problems, rather than a societal failure that merits care.
Recognizing the limitations of our current diagnoses, the forthcoming update to the International Classification of Diseases (ICD-11) will add a new category: complex PTSD. The ICD-11 will attempt to widen the concept of trauma to include “conditions of prolonged adversity, in the form of sustained, repeated, or multiple forms of traumatic exposure.” Trauma exposure examples include genocide campaigns, childhood sexual abuse, child soldiering, severe domestic violence, torture, or slavery. The ICD-11 also expands our understanding of the consequences of trauma to include “affective dysregulation,” “negative self-concept,” and “disturbances in relationships” as part of a concept called “disturbances in self-organization.” Those are important steps in acknowledging the consequences of different forms of trauma as well as noticing a richer array of damages from those incidents.4
While the World Health Organization’s latest iteration of the ICD takes an important step in widening the scope of our diagnostic tools, we are cognizant that our field’s obsessional search for diagnoses, labels, and nomenclature reinforces a detrimental focus on symptoms over stories. However, as forensic mental health providers, we also are keenly aware that a failure to adopt common definitions impedes forensic evaluations, patient advocacy, public policy, and most importantly, patient care.
In the end, we have trained society to understand pathology through narrow lenses, and therefore, . So, despite the limitations of labels, let’s be encouraged by the World Health Organization’s efforts and continue in that direction.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Lehman is a licensed clinical and forensic psychologist in San Diego. Her practice consists of conducting forensic psychological evaluations for the courts with children, adolescents, and adults. Dr. Lehman has been qualified as an expert witness in California as well as in the federal courts. She previously was a supervisor at Sharper Future, a forensic rehabilitation program, and previously served as an adjunct faculty member at Alliant International University, San Diego. Dr. Lehman can be reached at fran@lehmanforensicpsych.com.
References
1. Am J Psychiatry. 2000 Sep;157(9):1501-4.
2. Psychiatr Ann. 2005;35(5):390-8.
3. Psychiatr Ann. 2005;35(5):401-8.
4. Eur J Psychotraumatol. 2018 Jan 15. doi: 10.1080/20008198.2017.1418103.
The horrors faced by migrant families forced to separate under the new U.S. “zero tolerance” policy continue to unfold. Tragic emblems of this policy include tapes of crying children and the reported suicide of a father who had been separated from his children.
A federal judge had issued an injunction requiring the reunification of thousands of families by July 26. Despite that deadline, hundreds of adults are no longer in the United States, and hundreds of children are scattered in shelters across the country.
In response to those events, mental health and medical organizations have released powerful statements. The American Psychological Association stated: “The administration’s policy ... is not only needless and cruel, it threatens the mental and physical health of both the children and their caregivers.” The American Medical Association issued a call asserting that separating children from their parents “will do great harm” and “create negative health impacts that will last an individual’s entire lifespan.” Meanwhile, the American Psychiatric Association’s president, Altha J. Stewart, MD, released a statement affirming that “any forced separation is highly stressful for children and can cause lifelong trauma, as well as an increased risk of other mental illnesses, such as depression, anxiety, and posttraumatic stress disorder.”
As forensic experts who testify about the mental well-being of immigration detainees, we applaud those powerful and unambiguous messages from the leaders in our fields. Yet, their statements also underscore the limitations of our diagnostic models: Our field is caught in the difficult position of either applying ill-fitting diagnostic labels or overpathologizing a normal reaction to horrific circumstances. While not applying diagnoses potentially minimizes the enormous psychological burden of separation, diagnosing depression or PTSD as catchalls for suffering incorrectly defines the experience of many survivors of ongoing trauma.
Currently, most providers, in trying to communicate the effects of ongoing trauma, rely on the diagnoses of depression or PTSD. Both of these diagnoses, however, are problematic. The diagnosis of major depressive disorder, for example, is useful in communicating a loss of hope, and the inability to enjoy pleasurable things. However, depression is an episodic illness, often part of a larger chronic disorder.1 Depression often has a genetic-hereditary component. On the other hand, children suffering from childhood traumas often present lifelong and wide-ranging problems, which may be triggered by reminders but are not episodic. For example, children experiencing parental separation have difficulty forming attachments, which, in turn, leads to subsequent difficulty forming meaningful interpersonal relationships.
The diagnosis of PTSD is useful in communicating a myriad of possible symptoms, which may accompany the trauma. However, PTSD implies a traumatic event as described in criteria A of the DSM-5: “exposure to actual or threatened death, serious injury, or sexual violence.” As such, PTSD poorly encompasses the wide array of smaller yet repetitive traumas experienced by victims of ongoing trauma, such as those youth separated from their parents at the U.S. border. Furthermore, PTSD is a disorder with specific symptoms that, based on a vast body of research,2,3 inadequately describes the multitude of interpersonal, psychological, and physical consequences associated with the type of trauma caused by family separations.
Our understanding of the long-term sequelae of childhood trauma has been greatly influenced by the adverse childhood experiences (ACE) study. The ACE study, one of the largest investigations ever conducted to assess associations between childhood maltreatment and later-life health and well-being, collected the life histories of more than 17,000 patients in a collaborative effort between the Centers for Disease Control and Prevention and Kaiser Permanente’s Health Appraisal Clinic.
The ACE study identified 10 forms of childhood trauma, including: abuse, neglect, abandonment, household dysfunction, and exposure to violence, that were strongly associated with negative psychological outcomes such as depression, suicide attempts, and engagement in high-risk behaviors, as well as significant medical consequences, including higher incidence of heart disease, diabetes, and stroke. Ultimately, having four or more ACEs was associated with early death.
In response to the emerging body of research on childhood trauma, various terms, including complex trauma, type-II trauma, and complex PTSD, have entered our professional lexicon as a means of communicating the wide-ranging consequences of developmental trauma. On the one hand, the less defined and rigid nature of these terms permits mental health providers to develop a rich narrative of a patient’s background, encompassing the patient’s behavior, character, and symptoms. However, the absence of formal terminology also has its drawbacks: Courts and juries have grown accustomed to diagnoses, labels, and syndromes. Most forensic mental health providers who testify about developmental trauma in court can predict the question: “So doctor, you are saying that the individual’s presentation is not severe enough to be considered PTSD, am I correct?” Disorders justify treatment, can explain disability, and warrant empathy; concomitantly, “complex trauma” runs the risk of being considered an academic explanation for trauma victims’ lifelong problems, rather than a societal failure that merits care.
Recognizing the limitations of our current diagnoses, the forthcoming update to the International Classification of Diseases (ICD-11) will add a new category: complex PTSD. The ICD-11 will attempt to widen the concept of trauma to include “conditions of prolonged adversity, in the form of sustained, repeated, or multiple forms of traumatic exposure.” Trauma exposure examples include genocide campaigns, childhood sexual abuse, child soldiering, severe domestic violence, torture, or slavery. The ICD-11 also expands our understanding of the consequences of trauma to include “affective dysregulation,” “negative self-concept,” and “disturbances in relationships” as part of a concept called “disturbances in self-organization.” Those are important steps in acknowledging the consequences of different forms of trauma as well as noticing a richer array of damages from those incidents.4
While the World Health Organization’s latest iteration of the ICD takes an important step in widening the scope of our diagnostic tools, we are cognizant that our field’s obsessional search for diagnoses, labels, and nomenclature reinforces a detrimental focus on symptoms over stories. However, as forensic mental health providers, we also are keenly aware that a failure to adopt common definitions impedes forensic evaluations, patient advocacy, public policy, and most importantly, patient care.
In the end, we have trained society to understand pathology through narrow lenses, and therefore, . So, despite the limitations of labels, let’s be encouraged by the World Health Organization’s efforts and continue in that direction.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Lehman is a licensed clinical and forensic psychologist in San Diego. Her practice consists of conducting forensic psychological evaluations for the courts with children, adolescents, and adults. Dr. Lehman has been qualified as an expert witness in California as well as in the federal courts. She previously was a supervisor at Sharper Future, a forensic rehabilitation program, and previously served as an adjunct faculty member at Alliant International University, San Diego. Dr. Lehman can be reached at fran@lehmanforensicpsych.com.
References
1. Am J Psychiatry. 2000 Sep;157(9):1501-4.
2. Psychiatr Ann. 2005;35(5):390-8.
3. Psychiatr Ann. 2005;35(5):401-8.
4. Eur J Psychotraumatol. 2018 Jan 15. doi: 10.1080/20008198.2017.1418103.
VA Funds Intimate Partner Violence Programs
In the US, 36% of women and 29% of men have experienced rape, physical violence, or stalking by an intimate partner. Research suggests that veterans may be at greater risk for intimate partner violence than civilian counterparts, given the unique stressors posed by military life, such as military deployments that result in family separation, reintegration issues, and combat-related health issues, including PTSD and TBI. According to the VA’s Domestic Violence Task Force, the overall 12-month prevalence of inmate partner violence (IPV) perpetration among active duty service members was 22%, and victimization was 30%.
To help address this problem, the VA launched the IPV Assistance Program in 2014 and has since established coordinators at more than 115 facilities. The program coordinators use resources from mental health, primary care, women’s health, veterans’ justice outreach, and employee occupational health and assistance programs. The program also offers intervention through VA and community partnerships that address housing, education, and employment needs.
The program takes a holistic approach, focusing on developing a culture of safety, the VA says, with the goal of understanding, recognizing and responding to the effects of all types of trauma, including physical, sexual, and psychological. “We are giving careful attention to this program,” says Acting VA Secretary Peter O’Rourke, “ensuring it is integrated into clinical care and workplace safety.”
In the US, 36% of women and 29% of men have experienced rape, physical violence, or stalking by an intimate partner. Research suggests that veterans may be at greater risk for intimate partner violence than civilian counterparts, given the unique stressors posed by military life, such as military deployments that result in family separation, reintegration issues, and combat-related health issues, including PTSD and TBI. According to the VA’s Domestic Violence Task Force, the overall 12-month prevalence of inmate partner violence (IPV) perpetration among active duty service members was 22%, and victimization was 30%.
To help address this problem, the VA launched the IPV Assistance Program in 2014 and has since established coordinators at more than 115 facilities. The program coordinators use resources from mental health, primary care, women’s health, veterans’ justice outreach, and employee occupational health and assistance programs. The program also offers intervention through VA and community partnerships that address housing, education, and employment needs.
The program takes a holistic approach, focusing on developing a culture of safety, the VA says, with the goal of understanding, recognizing and responding to the effects of all types of trauma, including physical, sexual, and psychological. “We are giving careful attention to this program,” says Acting VA Secretary Peter O’Rourke, “ensuring it is integrated into clinical care and workplace safety.”
In the US, 36% of women and 29% of men have experienced rape, physical violence, or stalking by an intimate partner. Research suggests that veterans may be at greater risk for intimate partner violence than civilian counterparts, given the unique stressors posed by military life, such as military deployments that result in family separation, reintegration issues, and combat-related health issues, including PTSD and TBI. According to the VA’s Domestic Violence Task Force, the overall 12-month prevalence of inmate partner violence (IPV) perpetration among active duty service members was 22%, and victimization was 30%.
To help address this problem, the VA launched the IPV Assistance Program in 2014 and has since established coordinators at more than 115 facilities. The program coordinators use resources from mental health, primary care, women’s health, veterans’ justice outreach, and employee occupational health and assistance programs. The program also offers intervention through VA and community partnerships that address housing, education, and employment needs.
The program takes a holistic approach, focusing on developing a culture of safety, the VA says, with the goal of understanding, recognizing and responding to the effects of all types of trauma, including physical, sexual, and psychological. “We are giving careful attention to this program,” says Acting VA Secretary Peter O’Rourke, “ensuring it is integrated into clinical care and workplace safety.”
Puerto Rico after Maria: Trauma team returns
Ten months after Hurricane Maria pummeled into the island of Puerto Rico, things have begun to get better.
Despite some signs of recovery, mental – and physical – health problems are ongoing. The official death toll was recorded at 64, but a recent study by the Harvard School of Public Health estimates that it is closer to 5,000 (N Engl J Med. 2018 May 28. doi: 10.1056/NEJMsa1803972). Some reports show that the suicide rate on the island has soared by nearly 30%. Other reports show that unemployment has increased as has crime, and some estimates show that up to 200,000 people have left the island. As of this writing, thousands of people still are without power. And the hurricane season has begun yet again.
Week-long training gets underway
A few weeks ago, I joined a team of mental health professionals affiliated with the International Center for Psychosocial Trauma at the University of Missouri–Columbia (UMICPT) that went to Puerto Rico for a week. Under the leadership of UMICPT founder Syed Arshad Husain, MD, our goals were train our colleagues and teachers how to help children suffering from posttraumatic stress disorder after Maria. Several months earlier, our team had traveled to the island to train doctors, psychologists, social workers, and other mental health workers in San Juan and Ponce, and we were eager to return to continue our work.
Carlos Sellas, PsyD, a faculty member and supervisor of child and adolescent mental health clinics at Ponce Health Sciences University, attended the training. Dr. Sellas reported that somatic symptoms among the children had escalated after the hurricane. One child, whose grandfather suffered a myocardial infarction after Maria, repeatedly complains of chest pain. Pseudoseizures also have been observed.
Dr. Sellas said he also is seeing increased suicidal ideation and behavior in children and adolescents. In addition, some children are reporting auditory and visual hallucinations, and phobic reaction to rainstorms and lightening – in addition to fears of the dark.
Regressive behaviors cited
Laura Deliz, PsyD, director of the Autism Center at Ponce Health Sciences University, also attended the training. She reported that some of the autistic children under her care are manifesting regressive behaviors and are losing learned skills. They are more insecure, cling to transitional objects, and complain of pains, sleep problems, and show signs of having eating disorders. “Little things bother them more,” Dr. Deliz said. They cry more frequently, display more problems with concentration and attention, and are having more tantrums.
Comorbid with PTSD, symptoms of depression, anxiety disorders, conduct disorders, attention deficit disorders, and substance use disorders also are being encountered. Substance abuse more often is a comorbid condition in adolescents, but clinicians also are seeing this in children. Impulsive behaviors, self-destructive behaviors, and feelings of guilt also are being observed.
Compassion fatigue
Many trainees also are reporting symptoms of secondary traumatization and compassion fatigue. One trainee who lives in a mountain area had no electricity until 3 weeks before the training. Access to clean water has been sporadic, because power is required to pump the water.
Efforts to obtain gasoline has entailed waiting in line for 5 hours, sometimes only to have the supply run out upon reaching the pump. Puerto Rico continues to experience rolling blackouts. The island’s power company has lacked the proper materials to fix the problems. The elderly seem to be the main victims of this failing. Many of the elderly in the mountain areas, for example, still have no clean drinking water or electricity. Many of them live alone, and the churches are trying to help them.
Another trainee from the north coast, where the primary source of work is the dairy industry, reported that, when the power went off, the electric fences failed – and the cows wandered. Many became ill and died. An entire herd perished when an electric wire fell into nearby water.
Meanwhile, another trainee reported seeing a lot of anxiety and fear in the faces of the people waiting in long lines in the supermarkets trying to buy water, food that did not require refrigeration or cooking, and among people waiting in long lines at gas stations. Some people were sociable and supportive to one another; others were encouraging and telling stories. But there also were reports of fights breaking out. People were feeling frustrated because they could not get their basic needs met.
Among the adults, according to one observer, a sense of hopelessness and sadness prevailed. In the first weeks after the hurricane, just trying to communicate with other family members was a struggle because of the absence of cell phone service. In some ways, the children seemed more resilient, because they still managed to find ways to engage in play.
Compassion fatigue also is being experienced by many of the teachers on the island, our team learned. Many of them do not know whether they will have jobs at the beginning of the new school year. The public education system, already hit hard by a decade-long recession that preceded Maria, remains challenged. Of the 1,113 public schools, only 828 will remain operational, according to the Orlando Sentinel. Meanwhile, the psychosocial environment in many of the schools is not healthy, “not when you have students who are hungry and emotionally hurting,” according to one of our students.
UMICPT curriculum
When our team travels to a traumatized area, we use the model of “training the trainers.” We teach local mental health professionals and teachers how to recognize some of the negative sequelae of trauma in children, including PTSD, complex traumatic grief, depression, and phobias. It is our aim to train them, so they can train others to recognize these conditions, and provide evidence-based interventions, which in turn can help to alleviate symptoms and promote healing.
Our students already have some training in mental health. We seek to use their training and their experiences in our exercises. They learn from us, and we also learn much from them. When they share their experiences with us, we learn about their cultural values, which in turn enables us to provide culturally sensitive training. Skills for recovering from trauma include psychoeducation, relaxation and visualization training, dialectical behavioral therapy strategies for stress reduction, art therapy, narrative therapy, mindfulness training, and group therapy.
Future plans
UMICPT plans to make two more trips to Puerto Rico. A group of trainees will be further trained to serve as trainers to others in some of the techniques they have been taught. There is a plan to conduct a needs assessment in the schools and train teachers during the visit. Trained teachers would then have the option of introducing a weekly mental hygiene hour into the schools, with the aim of providing some relief to the children suffering from PTSD and other psychiatric problems.
Judith R. Milner, MD, MEd, SpecEd, is a general, child, and adolescent psychiatrist in private practice in Everett, Wash. She has traveled with various groups over the years in an effort to alleviate some of the suffering caused by war and natural disaster. Her predominant association has been with the International Center for Psychosocial Trauma. She also has worked with Step Up Rwanda Women and Pygmy Survival Alliance, as well as on the Committee for Women at the American Psychiatric Association and the Consumer Issues Committee, the Committee on Diversity and Culture, and the Membership Committee for the American Academy of Child and Adolescent Psychiatry.
Ten months after Hurricane Maria pummeled into the island of Puerto Rico, things have begun to get better.
Despite some signs of recovery, mental – and physical – health problems are ongoing. The official death toll was recorded at 64, but a recent study by the Harvard School of Public Health estimates that it is closer to 5,000 (N Engl J Med. 2018 May 28. doi: 10.1056/NEJMsa1803972). Some reports show that the suicide rate on the island has soared by nearly 30%. Other reports show that unemployment has increased as has crime, and some estimates show that up to 200,000 people have left the island. As of this writing, thousands of people still are without power. And the hurricane season has begun yet again.
Week-long training gets underway
A few weeks ago, I joined a team of mental health professionals affiliated with the International Center for Psychosocial Trauma at the University of Missouri–Columbia (UMICPT) that went to Puerto Rico for a week. Under the leadership of UMICPT founder Syed Arshad Husain, MD, our goals were train our colleagues and teachers how to help children suffering from posttraumatic stress disorder after Maria. Several months earlier, our team had traveled to the island to train doctors, psychologists, social workers, and other mental health workers in San Juan and Ponce, and we were eager to return to continue our work.
Carlos Sellas, PsyD, a faculty member and supervisor of child and adolescent mental health clinics at Ponce Health Sciences University, attended the training. Dr. Sellas reported that somatic symptoms among the children had escalated after the hurricane. One child, whose grandfather suffered a myocardial infarction after Maria, repeatedly complains of chest pain. Pseudoseizures also have been observed.
Dr. Sellas said he also is seeing increased suicidal ideation and behavior in children and adolescents. In addition, some children are reporting auditory and visual hallucinations, and phobic reaction to rainstorms and lightening – in addition to fears of the dark.
Regressive behaviors cited
Laura Deliz, PsyD, director of the Autism Center at Ponce Health Sciences University, also attended the training. She reported that some of the autistic children under her care are manifesting regressive behaviors and are losing learned skills. They are more insecure, cling to transitional objects, and complain of pains, sleep problems, and show signs of having eating disorders. “Little things bother them more,” Dr. Deliz said. They cry more frequently, display more problems with concentration and attention, and are having more tantrums.
Comorbid with PTSD, symptoms of depression, anxiety disorders, conduct disorders, attention deficit disorders, and substance use disorders also are being encountered. Substance abuse more often is a comorbid condition in adolescents, but clinicians also are seeing this in children. Impulsive behaviors, self-destructive behaviors, and feelings of guilt also are being observed.
Compassion fatigue
Many trainees also are reporting symptoms of secondary traumatization and compassion fatigue. One trainee who lives in a mountain area had no electricity until 3 weeks before the training. Access to clean water has been sporadic, because power is required to pump the water.
Efforts to obtain gasoline has entailed waiting in line for 5 hours, sometimes only to have the supply run out upon reaching the pump. Puerto Rico continues to experience rolling blackouts. The island’s power company has lacked the proper materials to fix the problems. The elderly seem to be the main victims of this failing. Many of the elderly in the mountain areas, for example, still have no clean drinking water or electricity. Many of them live alone, and the churches are trying to help them.
Another trainee from the north coast, where the primary source of work is the dairy industry, reported that, when the power went off, the electric fences failed – and the cows wandered. Many became ill and died. An entire herd perished when an electric wire fell into nearby water.
Meanwhile, another trainee reported seeing a lot of anxiety and fear in the faces of the people waiting in long lines in the supermarkets trying to buy water, food that did not require refrigeration or cooking, and among people waiting in long lines at gas stations. Some people were sociable and supportive to one another; others were encouraging and telling stories. But there also were reports of fights breaking out. People were feeling frustrated because they could not get their basic needs met.
Among the adults, according to one observer, a sense of hopelessness and sadness prevailed. In the first weeks after the hurricane, just trying to communicate with other family members was a struggle because of the absence of cell phone service. In some ways, the children seemed more resilient, because they still managed to find ways to engage in play.
Compassion fatigue also is being experienced by many of the teachers on the island, our team learned. Many of them do not know whether they will have jobs at the beginning of the new school year. The public education system, already hit hard by a decade-long recession that preceded Maria, remains challenged. Of the 1,113 public schools, only 828 will remain operational, according to the Orlando Sentinel. Meanwhile, the psychosocial environment in many of the schools is not healthy, “not when you have students who are hungry and emotionally hurting,” according to one of our students.
UMICPT curriculum
When our team travels to a traumatized area, we use the model of “training the trainers.” We teach local mental health professionals and teachers how to recognize some of the negative sequelae of trauma in children, including PTSD, complex traumatic grief, depression, and phobias. It is our aim to train them, so they can train others to recognize these conditions, and provide evidence-based interventions, which in turn can help to alleviate symptoms and promote healing.
Our students already have some training in mental health. We seek to use their training and their experiences in our exercises. They learn from us, and we also learn much from them. When they share their experiences with us, we learn about their cultural values, which in turn enables us to provide culturally sensitive training. Skills for recovering from trauma include psychoeducation, relaxation and visualization training, dialectical behavioral therapy strategies for stress reduction, art therapy, narrative therapy, mindfulness training, and group therapy.
Future plans
UMICPT plans to make two more trips to Puerto Rico. A group of trainees will be further trained to serve as trainers to others in some of the techniques they have been taught. There is a plan to conduct a needs assessment in the schools and train teachers during the visit. Trained teachers would then have the option of introducing a weekly mental hygiene hour into the schools, with the aim of providing some relief to the children suffering from PTSD and other psychiatric problems.
Judith R. Milner, MD, MEd, SpecEd, is a general, child, and adolescent psychiatrist in private practice in Everett, Wash. She has traveled with various groups over the years in an effort to alleviate some of the suffering caused by war and natural disaster. Her predominant association has been with the International Center for Psychosocial Trauma. She also has worked with Step Up Rwanda Women and Pygmy Survival Alliance, as well as on the Committee for Women at the American Psychiatric Association and the Consumer Issues Committee, the Committee on Diversity and Culture, and the Membership Committee for the American Academy of Child and Adolescent Psychiatry.
Ten months after Hurricane Maria pummeled into the island of Puerto Rico, things have begun to get better.
Despite some signs of recovery, mental – and physical – health problems are ongoing. The official death toll was recorded at 64, but a recent study by the Harvard School of Public Health estimates that it is closer to 5,000 (N Engl J Med. 2018 May 28. doi: 10.1056/NEJMsa1803972). Some reports show that the suicide rate on the island has soared by nearly 30%. Other reports show that unemployment has increased as has crime, and some estimates show that up to 200,000 people have left the island. As of this writing, thousands of people still are without power. And the hurricane season has begun yet again.
Week-long training gets underway
A few weeks ago, I joined a team of mental health professionals affiliated with the International Center for Psychosocial Trauma at the University of Missouri–Columbia (UMICPT) that went to Puerto Rico for a week. Under the leadership of UMICPT founder Syed Arshad Husain, MD, our goals were train our colleagues and teachers how to help children suffering from posttraumatic stress disorder after Maria. Several months earlier, our team had traveled to the island to train doctors, psychologists, social workers, and other mental health workers in San Juan and Ponce, and we were eager to return to continue our work.
Carlos Sellas, PsyD, a faculty member and supervisor of child and adolescent mental health clinics at Ponce Health Sciences University, attended the training. Dr. Sellas reported that somatic symptoms among the children had escalated after the hurricane. One child, whose grandfather suffered a myocardial infarction after Maria, repeatedly complains of chest pain. Pseudoseizures also have been observed.
Dr. Sellas said he also is seeing increased suicidal ideation and behavior in children and adolescents. In addition, some children are reporting auditory and visual hallucinations, and phobic reaction to rainstorms and lightening – in addition to fears of the dark.
Regressive behaviors cited
Laura Deliz, PsyD, director of the Autism Center at Ponce Health Sciences University, also attended the training. She reported that some of the autistic children under her care are manifesting regressive behaviors and are losing learned skills. They are more insecure, cling to transitional objects, and complain of pains, sleep problems, and show signs of having eating disorders. “Little things bother them more,” Dr. Deliz said. They cry more frequently, display more problems with concentration and attention, and are having more tantrums.
Comorbid with PTSD, symptoms of depression, anxiety disorders, conduct disorders, attention deficit disorders, and substance use disorders also are being encountered. Substance abuse more often is a comorbid condition in adolescents, but clinicians also are seeing this in children. Impulsive behaviors, self-destructive behaviors, and feelings of guilt also are being observed.
Compassion fatigue
Many trainees also are reporting symptoms of secondary traumatization and compassion fatigue. One trainee who lives in a mountain area had no electricity until 3 weeks before the training. Access to clean water has been sporadic, because power is required to pump the water.
Efforts to obtain gasoline has entailed waiting in line for 5 hours, sometimes only to have the supply run out upon reaching the pump. Puerto Rico continues to experience rolling blackouts. The island’s power company has lacked the proper materials to fix the problems. The elderly seem to be the main victims of this failing. Many of the elderly in the mountain areas, for example, still have no clean drinking water or electricity. Many of them live alone, and the churches are trying to help them.
Another trainee from the north coast, where the primary source of work is the dairy industry, reported that, when the power went off, the electric fences failed – and the cows wandered. Many became ill and died. An entire herd perished when an electric wire fell into nearby water.
Meanwhile, another trainee reported seeing a lot of anxiety and fear in the faces of the people waiting in long lines in the supermarkets trying to buy water, food that did not require refrigeration or cooking, and among people waiting in long lines at gas stations. Some people were sociable and supportive to one another; others were encouraging and telling stories. But there also were reports of fights breaking out. People were feeling frustrated because they could not get their basic needs met.
Among the adults, according to one observer, a sense of hopelessness and sadness prevailed. In the first weeks after the hurricane, just trying to communicate with other family members was a struggle because of the absence of cell phone service. In some ways, the children seemed more resilient, because they still managed to find ways to engage in play.
Compassion fatigue also is being experienced by many of the teachers on the island, our team learned. Many of them do not know whether they will have jobs at the beginning of the new school year. The public education system, already hit hard by a decade-long recession that preceded Maria, remains challenged. Of the 1,113 public schools, only 828 will remain operational, according to the Orlando Sentinel. Meanwhile, the psychosocial environment in many of the schools is not healthy, “not when you have students who are hungry and emotionally hurting,” according to one of our students.
UMICPT curriculum
When our team travels to a traumatized area, we use the model of “training the trainers.” We teach local mental health professionals and teachers how to recognize some of the negative sequelae of trauma in children, including PTSD, complex traumatic grief, depression, and phobias. It is our aim to train them, so they can train others to recognize these conditions, and provide evidence-based interventions, which in turn can help to alleviate symptoms and promote healing.
Our students already have some training in mental health. We seek to use their training and their experiences in our exercises. They learn from us, and we also learn much from them. When they share their experiences with us, we learn about their cultural values, which in turn enables us to provide culturally sensitive training. Skills for recovering from trauma include psychoeducation, relaxation and visualization training, dialectical behavioral therapy strategies for stress reduction, art therapy, narrative therapy, mindfulness training, and group therapy.
Future plans
UMICPT plans to make two more trips to Puerto Rico. A group of trainees will be further trained to serve as trainers to others in some of the techniques they have been taught. There is a plan to conduct a needs assessment in the schools and train teachers during the visit. Trained teachers would then have the option of introducing a weekly mental hygiene hour into the schools, with the aim of providing some relief to the children suffering from PTSD and other psychiatric problems.
Judith R. Milner, MD, MEd, SpecEd, is a general, child, and adolescent psychiatrist in private practice in Everett, Wash. She has traveled with various groups over the years in an effort to alleviate some of the suffering caused by war and natural disaster. Her predominant association has been with the International Center for Psychosocial Trauma. She also has worked with Step Up Rwanda Women and Pygmy Survival Alliance, as well as on the Committee for Women at the American Psychiatric Association and the Consumer Issues Committee, the Committee on Diversity and Culture, and the Membership Committee for the American Academy of Child and Adolescent Psychiatry.
Are PTSD Responses Inherited or Acquired?
Neuroimaging studies have consistently reported reduced activation of the medial prefrontal cortex (mPFC) in patients with posttraumatic stress disorder (PTSD) while they recall and imagine stressful personal events. During script-driven imagery (SDI) sessions, patients with PTSD exhibit increased psychophysiologic (eg, heart rate, skin conductance, and facial electromyographic) responses to trauma-related memories. However, the origin of the responses remained unclear. Are they familial, acquired, or resulting from trauma exposure?
Researchers from Harvard University, University of California Los Angeles, and University of New England conducted a study of 26 male identical twin pairs to help find the answer. The participants were divided into 4 groups: combat-exposed with PTSD (ExP+), their combat-unexposed twins without PTSD, combat-exposed participants without PTSD, and their combat-unexposed twins without PTSD. They engaged in SDI during functional magnetic resonance (fMRI) imaging and concurrent skin conductance measurement.
The results of the fMRI tests showed diminished activation in the medial prefrontal cortex of the patients with PTSD compared with the other groups. The SC response scores did not correlate significantly with PTSD symptom severity.
Contrary to the researchers’ predictions, mPFC activation was not inversely correlated with PTSD symptom severity. However, they say their finding of reduced mPFC activation in the ExP+ group provides evidence that the abnormality is an acquired characteristic. If those findings are replicated, such objectively measured biologic characteristics could potentially aid in diagnosing PTSD or assessing treatment response.
Source:
Dahlgren MK, Laifer LM, VanElzakker MB, et al. Psychol Med. 2018;48(7):1128-1138.
doi: 10.1017/S003329171700263X.
Neuroimaging studies have consistently reported reduced activation of the medial prefrontal cortex (mPFC) in patients with posttraumatic stress disorder (PTSD) while they recall and imagine stressful personal events. During script-driven imagery (SDI) sessions, patients with PTSD exhibit increased psychophysiologic (eg, heart rate, skin conductance, and facial electromyographic) responses to trauma-related memories. However, the origin of the responses remained unclear. Are they familial, acquired, or resulting from trauma exposure?
Researchers from Harvard University, University of California Los Angeles, and University of New England conducted a study of 26 male identical twin pairs to help find the answer. The participants were divided into 4 groups: combat-exposed with PTSD (ExP+), their combat-unexposed twins without PTSD, combat-exposed participants without PTSD, and their combat-unexposed twins without PTSD. They engaged in SDI during functional magnetic resonance (fMRI) imaging and concurrent skin conductance measurement.
The results of the fMRI tests showed diminished activation in the medial prefrontal cortex of the patients with PTSD compared with the other groups. The SC response scores did not correlate significantly with PTSD symptom severity.
Contrary to the researchers’ predictions, mPFC activation was not inversely correlated with PTSD symptom severity. However, they say their finding of reduced mPFC activation in the ExP+ group provides evidence that the abnormality is an acquired characteristic. If those findings are replicated, such objectively measured biologic characteristics could potentially aid in diagnosing PTSD or assessing treatment response.
Source:
Dahlgren MK, Laifer LM, VanElzakker MB, et al. Psychol Med. 2018;48(7):1128-1138.
doi: 10.1017/S003329171700263X.
Neuroimaging studies have consistently reported reduced activation of the medial prefrontal cortex (mPFC) in patients with posttraumatic stress disorder (PTSD) while they recall and imagine stressful personal events. During script-driven imagery (SDI) sessions, patients with PTSD exhibit increased psychophysiologic (eg, heart rate, skin conductance, and facial electromyographic) responses to trauma-related memories. However, the origin of the responses remained unclear. Are they familial, acquired, or resulting from trauma exposure?
Researchers from Harvard University, University of California Los Angeles, and University of New England conducted a study of 26 male identical twin pairs to help find the answer. The participants were divided into 4 groups: combat-exposed with PTSD (ExP+), their combat-unexposed twins without PTSD, combat-exposed participants without PTSD, and their combat-unexposed twins without PTSD. They engaged in SDI during functional magnetic resonance (fMRI) imaging and concurrent skin conductance measurement.
The results of the fMRI tests showed diminished activation in the medial prefrontal cortex of the patients with PTSD compared with the other groups. The SC response scores did not correlate significantly with PTSD symptom severity.
Contrary to the researchers’ predictions, mPFC activation was not inversely correlated with PTSD symptom severity. However, they say their finding of reduced mPFC activation in the ExP+ group provides evidence that the abnormality is an acquired characteristic. If those findings are replicated, such objectively measured biologic characteristics could potentially aid in diagnosing PTSD or assessing treatment response.
Source:
Dahlgren MK, Laifer LM, VanElzakker MB, et al. Psychol Med. 2018;48(7):1128-1138.
doi: 10.1017/S003329171700263X.
Extreme heat and mental health: Protecting patients
Now that the summer is in full swing, it is incumbent upon the psychiatric and mental health community to learn about the specific effects on behavior, psychiatric risks, and outcomes – and to plan for ways to protect our patients and communities.
Extreme heat has significant effects on mental health and behavior. Research shows1 that the number of people exposed to extreme heat is expected to rise in many American cities, particularly across the southern United States. Records were set in May 2018 across the United States and around the world. In the United States, those May temperatures were the warmest ever recorded, representing the hottest spring ever.2 Around the world, the warmer-than-average conditions that engulfed much of the land and sea surfaces made May the fourth-warmest since records started being kept in 1880.2
In short, these trends are not remitting. Extreme heat and climate disruption are the new normal.
Extreme heat makes many people cranky, agitated, or listless. However, heat waves are not benign, uncomfortable periods; they have profound health risks tied to increasing rates of anxiety, depression, posttraumatic stress disorder, and even death. In fact, extreme heat is now considered to be the single largest weather-related cause of death, exceeding hurricanes, lightning, tornadoes, floods, and earthquakes combined. The Centers for Disease Control and Prevention reports 7,800 deaths attributable to extreme heat between 1999 and 2009 – and predicts more frequent and extreme heat.3
In addition, extreme heat has been linked to increases in aggression and violence. One standard deviation of temperature increase and rainfall is associated with a 4% increase in interpersonal violence and 14% increase in intergroup violence.4 Anecdotal stories underscore the well known lore among prison staff of increased inmate violence during heat waves.5
Complex cognitive tasks such as working memory (spatial span test, pattern recognition) have been observed to be significantly impaired through heat stress.6 Increased heat also contributes to insomnia and worsens with increased humidity.7 A study in England and Wales showed a link between a possible association between hot weather and an increased risk of suicide.8 People with mental illness and those who abuse substances are considered an especially vulnerable population to the impacts of extreme heat and other climate change–related events. Co-occurring variables such as poverty, substandard housing, and lack of access to cool environments all contribute to this increased vulnerability. Homeless mentally ill have little control over their environments and have very limited ability to protect themselves from heat exposures and therefore are at extreme risk.9 . Regretfully, these kinds of cooling systems are out of reach for many people who live on the margins of society.
Furthermore, patients with severe psychotic or mood disorders, substance abuse disorders, and cognitive impairments who are able to compensate with marginal executive functioning during periods of normal weather are challenged during intense heat, and can lose their fragile ability to make plans, have good judgment, and care effectively for themselves. These patients are more likely to experience heat stroke and other heat-related morbidity.
Here is evidence that supports the greater impact of extreme heat on psychiatric patients:
- Increased emergency department and hospitalization for patients with preexisting psychiatric illness during heat waves.10,11
- Preexisting mental illness alone increases the risk of mortality during extreme heat events by 2 to 3 times.12,13
- Patients with schizophrenia might have underlying impairments in thermoregulation that are intrinsic to the disease. Such impairments would explain the perplexing sight of psychotic patients bundled up in layers on hot days.14
- Psychiatric medications (antipsychotics, anticholinergics, and antidepressants) have the potential to impair the body’s heat regulatory functioning; lithium affects fluid homeostasis.15
The negative effects of climate change are not equally distributed, and people with mental illness are among the most vulnerable. Given the predictable future of extreme heat waves (potentially increasing the population exposure by four- to sixfold by midcentury),1 we must do everything we can to educate our patients so that they take preventive measures to protect themselves from the adverse effects of extreme heat.
References
1. Nature Climate Change. 2015 May 18;5:652-5.
2. National Oceanic and Atmospheric Administration Global Climate Report. May 2018.
3. “Climate Change and Extreme Heat Events.” Centers for Disease Control and Prevention.
4. Science. 2013 Sep 13;341(6151).
5. Personal communication.
6. Int J Hyperthermia. 2003 May-Jun;19(3):355-72.
7. J Physiol Anthropol. 2012 May 31;31(14).
8. Br J Psychiatry. 2007 Aug;191:106-12.
9. U.S. Global Change Research Program, 2016. “The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment.” Chapters 8 and 9.
10. J Affect Disord. 2014 Feb;155:154-61.
11. Environ Health Perspect. 2008 Oct;116(10):1369-75.
12. Psychiatr Serv. 1998 Aug;49(8):1088-90.
13. Arch Intern Med. 2007 Nov. 12;167(20):2170-6.
14. Schizophr Res. 2004 Aug 1;69(2-3):149-57.
15. Eur Psychiatry. 2007 Sep;22(6):335-8.
Strategies for patients, communities
Part of the job of mental health professionals is psychoeducation, or teaching patients and families about the health risks tied to the psychological and physical impacts of heat exposure. Also, we should provide advice about effective management of psychiatric medications – such as monitoring lithium levels and considering medication dose adjustments – to reduce risks. Another key step is engaging caregivers, case managers, visiting nurses, and family members so that they closely monitor vulnerable populations. Providing information about the availability of respite care and cooling centers is another concrete step clinicians can take to help minimize the impact of extreme heat on patients.
Information that can be shared with patients about the threat include:
- A brochure from the Climate Psychiatry Alliance that offers specific ways to keep cool in extreme heat.
- A guidebook produced by the Environmental Protection Agency and the Centers for Disease Control and Prevention that explains what people can do to prepare for extreme heat.
- More extensive interventions can be found at https://www.climatepsychiatry.org/what-to-do/.
Dr. Cooper is in private practice and is affiliated with the department of psychiatry at the University of California, San Francisco. She is a Distinguished Life Fellow of the American Psychiatric Association.
Now that the summer is in full swing, it is incumbent upon the psychiatric and mental health community to learn about the specific effects on behavior, psychiatric risks, and outcomes – and to plan for ways to protect our patients and communities.
Extreme heat has significant effects on mental health and behavior. Research shows1 that the number of people exposed to extreme heat is expected to rise in many American cities, particularly across the southern United States. Records were set in May 2018 across the United States and around the world. In the United States, those May temperatures were the warmest ever recorded, representing the hottest spring ever.2 Around the world, the warmer-than-average conditions that engulfed much of the land and sea surfaces made May the fourth-warmest since records started being kept in 1880.2
In short, these trends are not remitting. Extreme heat and climate disruption are the new normal.
Extreme heat makes many people cranky, agitated, or listless. However, heat waves are not benign, uncomfortable periods; they have profound health risks tied to increasing rates of anxiety, depression, posttraumatic stress disorder, and even death. In fact, extreme heat is now considered to be the single largest weather-related cause of death, exceeding hurricanes, lightning, tornadoes, floods, and earthquakes combined. The Centers for Disease Control and Prevention reports 7,800 deaths attributable to extreme heat between 1999 and 2009 – and predicts more frequent and extreme heat.3
In addition, extreme heat has been linked to increases in aggression and violence. One standard deviation of temperature increase and rainfall is associated with a 4% increase in interpersonal violence and 14% increase in intergroup violence.4 Anecdotal stories underscore the well known lore among prison staff of increased inmate violence during heat waves.5
Complex cognitive tasks such as working memory (spatial span test, pattern recognition) have been observed to be significantly impaired through heat stress.6 Increased heat also contributes to insomnia and worsens with increased humidity.7 A study in England and Wales showed a link between a possible association between hot weather and an increased risk of suicide.8 People with mental illness and those who abuse substances are considered an especially vulnerable population to the impacts of extreme heat and other climate change–related events. Co-occurring variables such as poverty, substandard housing, and lack of access to cool environments all contribute to this increased vulnerability. Homeless mentally ill have little control over their environments and have very limited ability to protect themselves from heat exposures and therefore are at extreme risk.9 . Regretfully, these kinds of cooling systems are out of reach for many people who live on the margins of society.
Furthermore, patients with severe psychotic or mood disorders, substance abuse disorders, and cognitive impairments who are able to compensate with marginal executive functioning during periods of normal weather are challenged during intense heat, and can lose their fragile ability to make plans, have good judgment, and care effectively for themselves. These patients are more likely to experience heat stroke and other heat-related morbidity.
Here is evidence that supports the greater impact of extreme heat on psychiatric patients:
- Increased emergency department and hospitalization for patients with preexisting psychiatric illness during heat waves.10,11
- Preexisting mental illness alone increases the risk of mortality during extreme heat events by 2 to 3 times.12,13
- Patients with schizophrenia might have underlying impairments in thermoregulation that are intrinsic to the disease. Such impairments would explain the perplexing sight of psychotic patients bundled up in layers on hot days.14
- Psychiatric medications (antipsychotics, anticholinergics, and antidepressants) have the potential to impair the body’s heat regulatory functioning; lithium affects fluid homeostasis.15
The negative effects of climate change are not equally distributed, and people with mental illness are among the most vulnerable. Given the predictable future of extreme heat waves (potentially increasing the population exposure by four- to sixfold by midcentury),1 we must do everything we can to educate our patients so that they take preventive measures to protect themselves from the adverse effects of extreme heat.
References
1. Nature Climate Change. 2015 May 18;5:652-5.
2. National Oceanic and Atmospheric Administration Global Climate Report. May 2018.
3. “Climate Change and Extreme Heat Events.” Centers for Disease Control and Prevention.
4. Science. 2013 Sep 13;341(6151).
5. Personal communication.
6. Int J Hyperthermia. 2003 May-Jun;19(3):355-72.
7. J Physiol Anthropol. 2012 May 31;31(14).
8. Br J Psychiatry. 2007 Aug;191:106-12.
9. U.S. Global Change Research Program, 2016. “The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment.” Chapters 8 and 9.
10. J Affect Disord. 2014 Feb;155:154-61.
11. Environ Health Perspect. 2008 Oct;116(10):1369-75.
12. Psychiatr Serv. 1998 Aug;49(8):1088-90.
13. Arch Intern Med. 2007 Nov. 12;167(20):2170-6.
14. Schizophr Res. 2004 Aug 1;69(2-3):149-57.
15. Eur Psychiatry. 2007 Sep;22(6):335-8.
Strategies for patients, communities
Part of the job of mental health professionals is psychoeducation, or teaching patients and families about the health risks tied to the psychological and physical impacts of heat exposure. Also, we should provide advice about effective management of psychiatric medications – such as monitoring lithium levels and considering medication dose adjustments – to reduce risks. Another key step is engaging caregivers, case managers, visiting nurses, and family members so that they closely monitor vulnerable populations. Providing information about the availability of respite care and cooling centers is another concrete step clinicians can take to help minimize the impact of extreme heat on patients.
Information that can be shared with patients about the threat include:
- A brochure from the Climate Psychiatry Alliance that offers specific ways to keep cool in extreme heat.
- A guidebook produced by the Environmental Protection Agency and the Centers for Disease Control and Prevention that explains what people can do to prepare for extreme heat.
- More extensive interventions can be found at https://www.climatepsychiatry.org/what-to-do/.
Dr. Cooper is in private practice and is affiliated with the department of psychiatry at the University of California, San Francisco. She is a Distinguished Life Fellow of the American Psychiatric Association.
Now that the summer is in full swing, it is incumbent upon the psychiatric and mental health community to learn about the specific effects on behavior, psychiatric risks, and outcomes – and to plan for ways to protect our patients and communities.
Extreme heat has significant effects on mental health and behavior. Research shows1 that the number of people exposed to extreme heat is expected to rise in many American cities, particularly across the southern United States. Records were set in May 2018 across the United States and around the world. In the United States, those May temperatures were the warmest ever recorded, representing the hottest spring ever.2 Around the world, the warmer-than-average conditions that engulfed much of the land and sea surfaces made May the fourth-warmest since records started being kept in 1880.2
In short, these trends are not remitting. Extreme heat and climate disruption are the new normal.
Extreme heat makes many people cranky, agitated, or listless. However, heat waves are not benign, uncomfortable periods; they have profound health risks tied to increasing rates of anxiety, depression, posttraumatic stress disorder, and even death. In fact, extreme heat is now considered to be the single largest weather-related cause of death, exceeding hurricanes, lightning, tornadoes, floods, and earthquakes combined. The Centers for Disease Control and Prevention reports 7,800 deaths attributable to extreme heat between 1999 and 2009 – and predicts more frequent and extreme heat.3
In addition, extreme heat has been linked to increases in aggression and violence. One standard deviation of temperature increase and rainfall is associated with a 4% increase in interpersonal violence and 14% increase in intergroup violence.4 Anecdotal stories underscore the well known lore among prison staff of increased inmate violence during heat waves.5
Complex cognitive tasks such as working memory (spatial span test, pattern recognition) have been observed to be significantly impaired through heat stress.6 Increased heat also contributes to insomnia and worsens with increased humidity.7 A study in England and Wales showed a link between a possible association between hot weather and an increased risk of suicide.8 People with mental illness and those who abuse substances are considered an especially vulnerable population to the impacts of extreme heat and other climate change–related events. Co-occurring variables such as poverty, substandard housing, and lack of access to cool environments all contribute to this increased vulnerability. Homeless mentally ill have little control over their environments and have very limited ability to protect themselves from heat exposures and therefore are at extreme risk.9 . Regretfully, these kinds of cooling systems are out of reach for many people who live on the margins of society.
Furthermore, patients with severe psychotic or mood disorders, substance abuse disorders, and cognitive impairments who are able to compensate with marginal executive functioning during periods of normal weather are challenged during intense heat, and can lose their fragile ability to make plans, have good judgment, and care effectively for themselves. These patients are more likely to experience heat stroke and other heat-related morbidity.
Here is evidence that supports the greater impact of extreme heat on psychiatric patients:
- Increased emergency department and hospitalization for patients with preexisting psychiatric illness during heat waves.10,11
- Preexisting mental illness alone increases the risk of mortality during extreme heat events by 2 to 3 times.12,13
- Patients with schizophrenia might have underlying impairments in thermoregulation that are intrinsic to the disease. Such impairments would explain the perplexing sight of psychotic patients bundled up in layers on hot days.14
- Psychiatric medications (antipsychotics, anticholinergics, and antidepressants) have the potential to impair the body’s heat regulatory functioning; lithium affects fluid homeostasis.15
The negative effects of climate change are not equally distributed, and people with mental illness are among the most vulnerable. Given the predictable future of extreme heat waves (potentially increasing the population exposure by four- to sixfold by midcentury),1 we must do everything we can to educate our patients so that they take preventive measures to protect themselves from the adverse effects of extreme heat.
References
1. Nature Climate Change. 2015 May 18;5:652-5.
2. National Oceanic and Atmospheric Administration Global Climate Report. May 2018.
3. “Climate Change and Extreme Heat Events.” Centers for Disease Control and Prevention.
4. Science. 2013 Sep 13;341(6151).
5. Personal communication.
6. Int J Hyperthermia. 2003 May-Jun;19(3):355-72.
7. J Physiol Anthropol. 2012 May 31;31(14).
8. Br J Psychiatry. 2007 Aug;191:106-12.
9. U.S. Global Change Research Program, 2016. “The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment.” Chapters 8 and 9.
10. J Affect Disord. 2014 Feb;155:154-61.
11. Environ Health Perspect. 2008 Oct;116(10):1369-75.
12. Psychiatr Serv. 1998 Aug;49(8):1088-90.
13. Arch Intern Med. 2007 Nov. 12;167(20):2170-6.
14. Schizophr Res. 2004 Aug 1;69(2-3):149-57.
15. Eur Psychiatry. 2007 Sep;22(6):335-8.
Strategies for patients, communities
Part of the job of mental health professionals is psychoeducation, or teaching patients and families about the health risks tied to the psychological and physical impacts of heat exposure. Also, we should provide advice about effective management of psychiatric medications – such as monitoring lithium levels and considering medication dose adjustments – to reduce risks. Another key step is engaging caregivers, case managers, visiting nurses, and family members so that they closely monitor vulnerable populations. Providing information about the availability of respite care and cooling centers is another concrete step clinicians can take to help minimize the impact of extreme heat on patients.
Information that can be shared with patients about the threat include:
- A brochure from the Climate Psychiatry Alliance that offers specific ways to keep cool in extreme heat.
- A guidebook produced by the Environmental Protection Agency and the Centers for Disease Control and Prevention that explains what people can do to prepare for extreme heat.
- More extensive interventions can be found at https://www.climatepsychiatry.org/what-to-do/.
Dr. Cooper is in private practice and is affiliated with the department of psychiatry at the University of California, San Francisco. She is a Distinguished Life Fellow of the American Psychiatric Association.
Family separations could lead to irreversible health outcomes
The recent crises of the separation of stressed children from their equally stressed parents at this country’s southern border raises the specter of emotional and cognitive reactions within these children – the negative ramifications of which will manifest themselves for years to come.
Stress is ubiquitous. Children experience stress in the normal everyday frustrations that come their way: going to day care for a few hours and leaving mommy, the tripping and falling as they learn to walk, a toy breaking. These stresses help a child develop the capacity for emotional regulation and are termed “tolerable stress.” There is, however, a big difference between tolerable stress and toxic stress.
When the stress reaction in response to perceived or actual danger is beyond tolerable levels by virtue of its quality, intensity, and longevity, it saps the body’s ability to rally or handle the trauma that is being faced because of depleted neurotransmitters that normally assist the body to fight or flee from danger. Unfortunately, that’s not the end of the story: As the stressor persists, it has the capacity to produce long-term alterations in the resilience of the brain and body of the young child. This change often is irreversible.
Scientists and the lay public have begun to actively discuss the impact of adverse childhood experiences and their causal link to irreversible negative adult health outcomes. We now know without a doubt the impact of toxic versus tolerable stress on the hypothalamic-pituitary-adrenal axis of the young child. We are aware that the brain of a young child is particularly vulnerable to stress during critical and sensitive periods of development and that downstream effects of early trauma show up as disorganized behavior and cognitive underperformance.
Development plays a central role in children’s behavioral response to separation from parents. Infants develop a sense of stranger anxiety and the primacy of one central figure between ages 8 and 10 months. The baby chooses the parent over strangers for comfort and care. Roughly between ages 3 and 4, a child develops an internal representation of the parent as the primary figure in their lives so that they can tolerate short periods of being away from the primary caregiver for, let’s say, half a day. They depend on the parent for all their physical and emotional needs, which includes the need for a stable, nurturing, and predictable presence.
Familiarity of the environment, family rituals, consistency of daily routines provided by the parent help neural pathways responsible for the biologic unfolding of developmental milestones. Only recently, the science of early brain development and the role of early childhood trauma on brain biology has caught up with the longitudinal observational studies of bereaved children who lost their parents under circumstances of acute stress (the blitzkrieg, the Yom Kippur War, the Hungarian orphans) followed by the chronic stress phase of no primary caregiver for months to years. These observational studies coupled by the emerging neuroscience of early brain development and trauma are powerful informants of what is tolerable stress for children and what is not.
As a psychiatrist and expert in early child development, I am concerned about these long-term effects on migrant children. ; frantically seeking the parent/comforting familiar caregiver. Gradually as that possibility fades, with their limited ability to verbalize needs, episodic weeping can give way to disorganized behavior, despondence, and finally apathy and regression of milestones and cognitive abilities already achieved. The separation is merely the proxy face of other disasters that are probably co-occurring for the child/family and multiplies the dose of the stress: loss of siblings, loss of familiar physical elements of the landscape, loss of adequate physical sustenance, loss of routine, loss of consistency, increased vulnerability for physical illness, and the list goes on.
By age 2 and above, children are more vocal in their desire for reunification and will weep inconsolably upon separation. At this time their bodies and the psychological lens through which they view the world is changing irrevocably. Stress hormones, noxious at high levels such as cortisol/adrenocorticotropic hormone and glutamate rise to levels that no longer respond to the traditional negative feedback loop of the body and reach levels that lead to cell death in areas of the brain responsible for modulating emotions that can permanently change brain architecture.
Psychologically, the child seeks out the parent by any means (crying, flailing, tantruming). When no positive outcome occurs, this is replaced by an unwillingness to engage with the world, or the child engages but by behavior that is aggressive, disorganized, and regressed. In their own way, young children’s mistrust of the world leads to a variety of behaviors that impede normal development: rejection of others, disengagement from social connections by quietly sitting in a corner, being unwilling to eat or sleeping fitfully.
As separation from the primary caregiver continues, there is a deepening sense of hopelessness. Generally, no amount of physical props (toys, playgrounds) without the human familiar faces can rectify the feeling of abandonment and hopelessness that children in these conditions face. Assuming they are no strangers to trauma experienced through the lives of their asylum-seeking parents, this separation refreshes all the wounds of days gone by. At least in those moments of initial trauma, they had the parent as a buffer. But with this new separation, they are left to emotionally fend for themselves with no tools to manage their emotions.
We have to deal with the aftereffects of so many natural disasters that cannot be avoided: earthquakes, tornadoes, tsunamis that displace children and families with horrific outcomes. Did we really need a man-made disaster that will irrevocably change the brains and behaviors of a generation of children who just happen to be on our southern borders because of an accident of birth? If parents abandoned their children this way it would be called child abuse. Deliberate policy decisions that have such dire effects on children also take on the label of child abuse.
Although images of my years as a young parent of three boys are turning a sepia color in faded memory, in my new role as a grandmother observing my son and his wife so in love with their 2-year-old reinforces how much mutual dependency of a child and parent on each other strengthens the very fabric of life. Children are our future and are the building blocks of a just and humane society.
As of this writing, the administration has vowed to end these separations. But how will children who have been separated be reunited with their families?
The education of decision makers about the far-reaching emotional and physical consequences on normative childhood development must be a top priority for all scientists and professionals interested in kids and families: I think that means all of us.
Dr. Sood is professor of psychiatry and pediatrics, and senior professor of child mental health policy at the Virginia Treatment Center at Virginia Commonwealth University, Richmond. She also is affiliated with the department of psychiatry at VCU and Children’s Hospital of Richmond.
The recent crises of the separation of stressed children from their equally stressed parents at this country’s southern border raises the specter of emotional and cognitive reactions within these children – the negative ramifications of which will manifest themselves for years to come.
Stress is ubiquitous. Children experience stress in the normal everyday frustrations that come their way: going to day care for a few hours and leaving mommy, the tripping and falling as they learn to walk, a toy breaking. These stresses help a child develop the capacity for emotional regulation and are termed “tolerable stress.” There is, however, a big difference between tolerable stress and toxic stress.
When the stress reaction in response to perceived or actual danger is beyond tolerable levels by virtue of its quality, intensity, and longevity, it saps the body’s ability to rally or handle the trauma that is being faced because of depleted neurotransmitters that normally assist the body to fight or flee from danger. Unfortunately, that’s not the end of the story: As the stressor persists, it has the capacity to produce long-term alterations in the resilience of the brain and body of the young child. This change often is irreversible.
Scientists and the lay public have begun to actively discuss the impact of adverse childhood experiences and their causal link to irreversible negative adult health outcomes. We now know without a doubt the impact of toxic versus tolerable stress on the hypothalamic-pituitary-adrenal axis of the young child. We are aware that the brain of a young child is particularly vulnerable to stress during critical and sensitive periods of development and that downstream effects of early trauma show up as disorganized behavior and cognitive underperformance.
Development plays a central role in children’s behavioral response to separation from parents. Infants develop a sense of stranger anxiety and the primacy of one central figure between ages 8 and 10 months. The baby chooses the parent over strangers for comfort and care. Roughly between ages 3 and 4, a child develops an internal representation of the parent as the primary figure in their lives so that they can tolerate short periods of being away from the primary caregiver for, let’s say, half a day. They depend on the parent for all their physical and emotional needs, which includes the need for a stable, nurturing, and predictable presence.
Familiarity of the environment, family rituals, consistency of daily routines provided by the parent help neural pathways responsible for the biologic unfolding of developmental milestones. Only recently, the science of early brain development and the role of early childhood trauma on brain biology has caught up with the longitudinal observational studies of bereaved children who lost their parents under circumstances of acute stress (the blitzkrieg, the Yom Kippur War, the Hungarian orphans) followed by the chronic stress phase of no primary caregiver for months to years. These observational studies coupled by the emerging neuroscience of early brain development and trauma are powerful informants of what is tolerable stress for children and what is not.
As a psychiatrist and expert in early child development, I am concerned about these long-term effects on migrant children. ; frantically seeking the parent/comforting familiar caregiver. Gradually as that possibility fades, with their limited ability to verbalize needs, episodic weeping can give way to disorganized behavior, despondence, and finally apathy and regression of milestones and cognitive abilities already achieved. The separation is merely the proxy face of other disasters that are probably co-occurring for the child/family and multiplies the dose of the stress: loss of siblings, loss of familiar physical elements of the landscape, loss of adequate physical sustenance, loss of routine, loss of consistency, increased vulnerability for physical illness, and the list goes on.
By age 2 and above, children are more vocal in their desire for reunification and will weep inconsolably upon separation. At this time their bodies and the psychological lens through which they view the world is changing irrevocably. Stress hormones, noxious at high levels such as cortisol/adrenocorticotropic hormone and glutamate rise to levels that no longer respond to the traditional negative feedback loop of the body and reach levels that lead to cell death in areas of the brain responsible for modulating emotions that can permanently change brain architecture.
Psychologically, the child seeks out the parent by any means (crying, flailing, tantruming). When no positive outcome occurs, this is replaced by an unwillingness to engage with the world, or the child engages but by behavior that is aggressive, disorganized, and regressed. In their own way, young children’s mistrust of the world leads to a variety of behaviors that impede normal development: rejection of others, disengagement from social connections by quietly sitting in a corner, being unwilling to eat or sleeping fitfully.
As separation from the primary caregiver continues, there is a deepening sense of hopelessness. Generally, no amount of physical props (toys, playgrounds) without the human familiar faces can rectify the feeling of abandonment and hopelessness that children in these conditions face. Assuming they are no strangers to trauma experienced through the lives of their asylum-seeking parents, this separation refreshes all the wounds of days gone by. At least in those moments of initial trauma, they had the parent as a buffer. But with this new separation, they are left to emotionally fend for themselves with no tools to manage their emotions.
We have to deal with the aftereffects of so many natural disasters that cannot be avoided: earthquakes, tornadoes, tsunamis that displace children and families with horrific outcomes. Did we really need a man-made disaster that will irrevocably change the brains and behaviors of a generation of children who just happen to be on our southern borders because of an accident of birth? If parents abandoned their children this way it would be called child abuse. Deliberate policy decisions that have such dire effects on children also take on the label of child abuse.
Although images of my years as a young parent of three boys are turning a sepia color in faded memory, in my new role as a grandmother observing my son and his wife so in love with their 2-year-old reinforces how much mutual dependency of a child and parent on each other strengthens the very fabric of life. Children are our future and are the building blocks of a just and humane society.
As of this writing, the administration has vowed to end these separations. But how will children who have been separated be reunited with their families?
The education of decision makers about the far-reaching emotional and physical consequences on normative childhood development must be a top priority for all scientists and professionals interested in kids and families: I think that means all of us.
Dr. Sood is professor of psychiatry and pediatrics, and senior professor of child mental health policy at the Virginia Treatment Center at Virginia Commonwealth University, Richmond. She also is affiliated with the department of psychiatry at VCU and Children’s Hospital of Richmond.
The recent crises of the separation of stressed children from their equally stressed parents at this country’s southern border raises the specter of emotional and cognitive reactions within these children – the negative ramifications of which will manifest themselves for years to come.
Stress is ubiquitous. Children experience stress in the normal everyday frustrations that come their way: going to day care for a few hours and leaving mommy, the tripping and falling as they learn to walk, a toy breaking. These stresses help a child develop the capacity for emotional regulation and are termed “tolerable stress.” There is, however, a big difference between tolerable stress and toxic stress.
When the stress reaction in response to perceived or actual danger is beyond tolerable levels by virtue of its quality, intensity, and longevity, it saps the body’s ability to rally or handle the trauma that is being faced because of depleted neurotransmitters that normally assist the body to fight or flee from danger. Unfortunately, that’s not the end of the story: As the stressor persists, it has the capacity to produce long-term alterations in the resilience of the brain and body of the young child. This change often is irreversible.
Scientists and the lay public have begun to actively discuss the impact of adverse childhood experiences and their causal link to irreversible negative adult health outcomes. We now know without a doubt the impact of toxic versus tolerable stress on the hypothalamic-pituitary-adrenal axis of the young child. We are aware that the brain of a young child is particularly vulnerable to stress during critical and sensitive periods of development and that downstream effects of early trauma show up as disorganized behavior and cognitive underperformance.
Development plays a central role in children’s behavioral response to separation from parents. Infants develop a sense of stranger anxiety and the primacy of one central figure between ages 8 and 10 months. The baby chooses the parent over strangers for comfort and care. Roughly between ages 3 and 4, a child develops an internal representation of the parent as the primary figure in their lives so that they can tolerate short periods of being away from the primary caregiver for, let’s say, half a day. They depend on the parent for all their physical and emotional needs, which includes the need for a stable, nurturing, and predictable presence.
Familiarity of the environment, family rituals, consistency of daily routines provided by the parent help neural pathways responsible for the biologic unfolding of developmental milestones. Only recently, the science of early brain development and the role of early childhood trauma on brain biology has caught up with the longitudinal observational studies of bereaved children who lost their parents under circumstances of acute stress (the blitzkrieg, the Yom Kippur War, the Hungarian orphans) followed by the chronic stress phase of no primary caregiver for months to years. These observational studies coupled by the emerging neuroscience of early brain development and trauma are powerful informants of what is tolerable stress for children and what is not.
As a psychiatrist and expert in early child development, I am concerned about these long-term effects on migrant children. ; frantically seeking the parent/comforting familiar caregiver. Gradually as that possibility fades, with their limited ability to verbalize needs, episodic weeping can give way to disorganized behavior, despondence, and finally apathy and regression of milestones and cognitive abilities already achieved. The separation is merely the proxy face of other disasters that are probably co-occurring for the child/family and multiplies the dose of the stress: loss of siblings, loss of familiar physical elements of the landscape, loss of adequate physical sustenance, loss of routine, loss of consistency, increased vulnerability for physical illness, and the list goes on.
By age 2 and above, children are more vocal in their desire for reunification and will weep inconsolably upon separation. At this time their bodies and the psychological lens through which they view the world is changing irrevocably. Stress hormones, noxious at high levels such as cortisol/adrenocorticotropic hormone and glutamate rise to levels that no longer respond to the traditional negative feedback loop of the body and reach levels that lead to cell death in areas of the brain responsible for modulating emotions that can permanently change brain architecture.
Psychologically, the child seeks out the parent by any means (crying, flailing, tantruming). When no positive outcome occurs, this is replaced by an unwillingness to engage with the world, or the child engages but by behavior that is aggressive, disorganized, and regressed. In their own way, young children’s mistrust of the world leads to a variety of behaviors that impede normal development: rejection of others, disengagement from social connections by quietly sitting in a corner, being unwilling to eat or sleeping fitfully.
As separation from the primary caregiver continues, there is a deepening sense of hopelessness. Generally, no amount of physical props (toys, playgrounds) without the human familiar faces can rectify the feeling of abandonment and hopelessness that children in these conditions face. Assuming they are no strangers to trauma experienced through the lives of their asylum-seeking parents, this separation refreshes all the wounds of days gone by. At least in those moments of initial trauma, they had the parent as a buffer. But with this new separation, they are left to emotionally fend for themselves with no tools to manage their emotions.
We have to deal with the aftereffects of so many natural disasters that cannot be avoided: earthquakes, tornadoes, tsunamis that displace children and families with horrific outcomes. Did we really need a man-made disaster that will irrevocably change the brains and behaviors of a generation of children who just happen to be on our southern borders because of an accident of birth? If parents abandoned their children this way it would be called child abuse. Deliberate policy decisions that have such dire effects on children also take on the label of child abuse.
Although images of my years as a young parent of three boys are turning a sepia color in faded memory, in my new role as a grandmother observing my son and his wife so in love with their 2-year-old reinforces how much mutual dependency of a child and parent on each other strengthens the very fabric of life. Children are our future and are the building blocks of a just and humane society.
As of this writing, the administration has vowed to end these separations. But how will children who have been separated be reunited with their families?
The education of decision makers about the far-reaching emotional and physical consequences on normative childhood development must be a top priority for all scientists and professionals interested in kids and families: I think that means all of us.
Dr. Sood is professor of psychiatry and pediatrics, and senior professor of child mental health policy at the Virginia Treatment Center at Virginia Commonwealth University, Richmond. She also is affiliated with the department of psychiatry at VCU and Children’s Hospital of Richmond.
Consider heterogeneous experiences among veteran cohorts when treating PTSD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW YORK – Veterans are not a homogeneous group, and when treating them for posttraumatic stress, it helps to consider their specific cohort, according to Elspeth Cameron Ritchie, MD.
Veterans from the first Gulf War, for example, have lingering concerns regarding medical illness (Gulf War syndrome); those from Vietnam are aging and might have medical problems or find that while they did well while working, now they are experiencing PTSD symptoms for the first time; and those returning from the conflicts in Iraq and Afghanistan might have physical injuries from blasts – the “signature weapon” of those wars. Such blasts can cause amputations, genital injuries, head trauma, and PTSD, said Dr. Ritchie, of the Uniformed Services University of the Health Sciences, Bethesda, Md.
In this video interview, Dr. Ritchie discusses these and other issues related to the treatment of PTSD among veterans as presented during a workshop entitled “Psychiatry and U.S. Veterans,” which she chaired at the annual meeting of the American Psychiatric Association.
The workshop covered the spectrum of treatments that might be helpful for veterans.
“ They don’t want it to just be the doctor giving them a pill,” she said. “Veterans are resilient; they’re tough; they don’t like to be thought of as victims ... and when you’re working with them, it’s very important to link into that dynamic resilient piece and capitalize on their strengths.”
Dr. Ritchie reported having no disclosures.
SOURCE: Ritchie EC et al. APA Workshop.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW YORK – Veterans are not a homogeneous group, and when treating them for posttraumatic stress, it helps to consider their specific cohort, according to Elspeth Cameron Ritchie, MD.
Veterans from the first Gulf War, for example, have lingering concerns regarding medical illness (Gulf War syndrome); those from Vietnam are aging and might have medical problems or find that while they did well while working, now they are experiencing PTSD symptoms for the first time; and those returning from the conflicts in Iraq and Afghanistan might have physical injuries from blasts – the “signature weapon” of those wars. Such blasts can cause amputations, genital injuries, head trauma, and PTSD, said Dr. Ritchie, of the Uniformed Services University of the Health Sciences, Bethesda, Md.
In this video interview, Dr. Ritchie discusses these and other issues related to the treatment of PTSD among veterans as presented during a workshop entitled “Psychiatry and U.S. Veterans,” which she chaired at the annual meeting of the American Psychiatric Association.
The workshop covered the spectrum of treatments that might be helpful for veterans.
“ They don’t want it to just be the doctor giving them a pill,” she said. “Veterans are resilient; they’re tough; they don’t like to be thought of as victims ... and when you’re working with them, it’s very important to link into that dynamic resilient piece and capitalize on their strengths.”
Dr. Ritchie reported having no disclosures.
SOURCE: Ritchie EC et al. APA Workshop.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW YORK – Veterans are not a homogeneous group, and when treating them for posttraumatic stress, it helps to consider their specific cohort, according to Elspeth Cameron Ritchie, MD.
Veterans from the first Gulf War, for example, have lingering concerns regarding medical illness (Gulf War syndrome); those from Vietnam are aging and might have medical problems or find that while they did well while working, now they are experiencing PTSD symptoms for the first time; and those returning from the conflicts in Iraq and Afghanistan might have physical injuries from blasts – the “signature weapon” of those wars. Such blasts can cause amputations, genital injuries, head trauma, and PTSD, said Dr. Ritchie, of the Uniformed Services University of the Health Sciences, Bethesda, Md.
In this video interview, Dr. Ritchie discusses these and other issues related to the treatment of PTSD among veterans as presented during a workshop entitled “Psychiatry and U.S. Veterans,” which she chaired at the annual meeting of the American Psychiatric Association.
The workshop covered the spectrum of treatments that might be helpful for veterans.
“ They don’t want it to just be the doctor giving them a pill,” she said. “Veterans are resilient; they’re tough; they don’t like to be thought of as victims ... and when you’re working with them, it’s very important to link into that dynamic resilient piece and capitalize on their strengths.”
Dr. Ritchie reported having no disclosures.
SOURCE: Ritchie EC et al. APA Workshop.
REPORTING FROM APA