User login
Unusual Case of Pemphigus Vulgaris Mimicking Localized Pustular Psoriasis of the Hands and Feet
Improvement in Psoriasis During Rituximab Therapy for Mixed Cryoglobulinemia Type II
Joint Involvement Predicts Aggressive Systemic Sclerosis
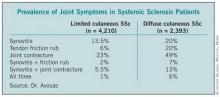
ROME — Clinical articular involvement is strikingly common in patients with systemic sclerosis and is associated with a more active and severe disease phenotype, according to an analysis of the world’s largest systemic sclerosis (SSc) registry.
Synovitis proved to have a particularly strong association with systemic inflammation in this study. However, synovitis often occurred in concert with joint contractures and/or tendon friction rubs, raising the possibility of a shared underlying mechanism in their development, Dr. Jérôme Avouac said at the annual European Congress of Rheumatology.
He presented an analysis of joint symptoms and their clinical implications in 7,286 patients with systemic sclerosis (SSc) enrolled in the EULAR Scleroderma Trials and Research (EUSTAR) registry, the world’s largest SSc database. Of registrants, 58% had the limited cutaneous subtype of SSc, 33% had the diffuse cutaneous subtype, and 9% were not classified.
Active disease, as defined by the 17-point dermal ultrasound, modified Rodnan skin scoring system, was present in 29% of the overall study population. Synovitis was associated with a highly significant 70% increased likelihood of having active disease, while tendon friction rubs were associated with a 51% increased rate. In contrast, joint contractures had no impact upon the risk of having active disease.
Synovitis was also associated with a 47% increased likelihood of having muscle weakness, a 49% increase in the risk of having elevated systolic pulmonary artery pressure, a 49% increase in the odds of having elevation of acute phase reactants, and a 29% increase in anti-topoisomerase-1 antibody positivity.
Patients with joint contractures were significantly more likely to have digital ulcerations (93% increased risk), elevated systolic pulmonary artery pressure (38% increased risk), muscle weakness (41% increase), and pulmonary fibrosis (23% increased risk), the rheumatologist continued.
Tendon friction rubs were associated with a 21% increased likelihood of experiencing digital ulceration, a 22% increase in pulmonary fibrosis, and a 38% greater likelihood of proteinuria.
These study findings provide support for making early detection of articular involvement in SSc a disease-management priority, according to Dr. Avouac.
He noted, however, that this was a cross-sectional analysis. Ongoing prospective follow-up of the EUSTAR registry participants will enable investigators to make a definitive determination of the merits of articular involvement as a predictor of disease severity.
This study was funded by the European League Against Rheumatism. Dr. Avouac declared having no financial conflicts.
ROME — Clinical articular involvement is strikingly common in patients with systemic sclerosis and is associated with a more active and severe disease phenotype, according to an analysis of the world’s largest systemic sclerosis (SSc) registry.
Synovitis proved to have a particularly strong association with systemic inflammation in this study. However, synovitis often occurred in concert with joint contractures and/or tendon friction rubs, raising the possibility of a shared underlying mechanism in their development, Dr. Jérôme Avouac said at the annual European Congress of Rheumatology.
He presented an analysis of joint symptoms and their clinical implications in 7,286 patients with systemic sclerosis (SSc) enrolled in the EULAR Scleroderma Trials and Research (EUSTAR) registry, the world’s largest SSc database. Of registrants, 58% had the limited cutaneous subtype of SSc, 33% had the diffuse cutaneous subtype, and 9% were not classified.
Active disease, as defined by the 17-point dermal ultrasound, modified Rodnan skin scoring system, was present in 29% of the overall study population. Synovitis was associated with a highly significant 70% increased likelihood of having active disease, while tendon friction rubs were associated with a 51% increased rate. In contrast, joint contractures had no impact upon the risk of having active disease.
Synovitis was also associated with a 47% increased likelihood of having muscle weakness, a 49% increase in the risk of having elevated systolic pulmonary artery pressure, a 49% increase in the odds of having elevation of acute phase reactants, and a 29% increase in anti-topoisomerase-1 antibody positivity.
Patients with joint contractures were significantly more likely to have digital ulcerations (93% increased risk), elevated systolic pulmonary artery pressure (38% increased risk), muscle weakness (41% increase), and pulmonary fibrosis (23% increased risk), the rheumatologist continued.
Tendon friction rubs were associated with a 21% increased likelihood of experiencing digital ulceration, a 22% increase in pulmonary fibrosis, and a 38% greater likelihood of proteinuria.
These study findings provide support for making early detection of articular involvement in SSc a disease-management priority, according to Dr. Avouac.
He noted, however, that this was a cross-sectional analysis. Ongoing prospective follow-up of the EUSTAR registry participants will enable investigators to make a definitive determination of the merits of articular involvement as a predictor of disease severity.
This study was funded by the European League Against Rheumatism. Dr. Avouac declared having no financial conflicts.
ROME — Clinical articular involvement is strikingly common in patients with systemic sclerosis and is associated with a more active and severe disease phenotype, according to an analysis of the world’s largest systemic sclerosis (SSc) registry.
Synovitis proved to have a particularly strong association with systemic inflammation in this study. However, synovitis often occurred in concert with joint contractures and/or tendon friction rubs, raising the possibility of a shared underlying mechanism in their development, Dr. Jérôme Avouac said at the annual European Congress of Rheumatology.
He presented an analysis of joint symptoms and their clinical implications in 7,286 patients with systemic sclerosis (SSc) enrolled in the EULAR Scleroderma Trials and Research (EUSTAR) registry, the world’s largest SSc database. Of registrants, 58% had the limited cutaneous subtype of SSc, 33% had the diffuse cutaneous subtype, and 9% were not classified.
Active disease, as defined by the 17-point dermal ultrasound, modified Rodnan skin scoring system, was present in 29% of the overall study population. Synovitis was associated with a highly significant 70% increased likelihood of having active disease, while tendon friction rubs were associated with a 51% increased rate. In contrast, joint contractures had no impact upon the risk of having active disease.
Synovitis was also associated with a 47% increased likelihood of having muscle weakness, a 49% increase in the risk of having elevated systolic pulmonary artery pressure, a 49% increase in the odds of having elevation of acute phase reactants, and a 29% increase in anti-topoisomerase-1 antibody positivity.
Patients with joint contractures were significantly more likely to have digital ulcerations (93% increased risk), elevated systolic pulmonary artery pressure (38% increased risk), muscle weakness (41% increase), and pulmonary fibrosis (23% increased risk), the rheumatologist continued.
Tendon friction rubs were associated with a 21% increased likelihood of experiencing digital ulceration, a 22% increase in pulmonary fibrosis, and a 38% greater likelihood of proteinuria.
These study findings provide support for making early detection of articular involvement in SSc a disease-management priority, according to Dr. Avouac.
He noted, however, that this was a cross-sectional analysis. Ongoing prospective follow-up of the EUSTAR registry participants will enable investigators to make a definitive determination of the merits of articular involvement as a predictor of disease severity.
This study was funded by the European League Against Rheumatism. Dr. Avouac declared having no financial conflicts.
New RA Guidelines Emphasize Early Treatment
The promised overhaul of treatment guidelines for rheumatoid arthritis has finally arrived, and with it, a "new paradigm" that focuses on early identification and treatment of the disabling disease.
The guidelines, which were developed by a joint committee from the American College of Rheumatology and the European League Against Rheumatism, are the latest update since the current guidelines were created in 1987.
Published jointly in the EULAR journal Annals of the Rheumatic Diseases (2010;69:1580-8) and the ACR's Arthritis and Rheumatism (2010;62:2569-81), the new guidelines were created in three phases over 2 years.
In the first phase, the goal was to "to identify the contributions of clinical and laboratory variables that in practice were the most predictive of the decision to initiate [disease-modifying antirheumatic drug] therapy in a population of patients with early undifferentiated synovitis," wrote the authors, led by Dr. Daniel Aletaha of the Medical University of Vienna.
To do this, a working group from both societies looked at data from 3,115 patients, and correlated whether or not the patients were ultimately prescribed methotrexate to an "agreed-upon list of standardized clinical and laboratory variables collected at baseline."
The odds of eventual methotrexate initiation were calculated for each variable. For example, swelling of the metacarpophalangeal joint had an odds ratio of 1.5, as did swelling of the proximal interphalangeal joint and the wrist. Tenderness of the hand (either the MCP, PIP, or wrist) was assigned an odds ratio of 2.0.
A moderate elevation of C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR) was assigned an OR of 1.0; a high elevation of either assay carried an OR of 2.0.
Finally, moderate levels of either rheumatoid factor or anti–citrullinated protein antibodies had an OR of 2.0 for the eventual prescription of a DMARD; high levels had an OR of 4.0.
In phase II, an "expert panel" of 12 rheumatologists related the above clinical and laboratory factors to the "probability of developing 'persistent inflammatory and/or erosive arthritis that is currently considered to be RA.'?"
The panel also looked at duration of symptoms (longer or shorter than 6 weeks) and the number and size of joints (large or small), in addition to the variables that were assessed in phase I. Using a computer program, the panel assigned each variable a point value of 1-100, with high scores indicating greater likelihood of RA.
Finally, phase III aimed to utilize the results of phases I and II "to develop a scoring system that would be applicable to newly presenting patients with undifferentiated inflammatory arthritis, to permit identification of those with a high probability of developing persistent and/or erosive RA."
This final scale assigns points in the following manner:
– One swollen "large joint" (defined as shoulders, elbows, hips, knees, and ankles) gets 0 points; involvement of 2-10 large joints gets 1 point.
– Involvement of 1-3 "small" joints (defined as metacarpophalangeal joints, proximal interphalangeal joints, second to fifth metatarsophalangeal joints, thumb interphalangeal joints, and wrists) gets 2 points, regardless of large-joint involvement; involvement of 4-10 small joints gets 3 points.
– Involvement of more than 10 joints, including at least one small joint, gets 5 points.
– Both a negative rheumatoid factor (RF) test and a negative anti–citrullinated protein antibody test (ACPA) gets 0 points, whereas having a "low-positive" RF or ACPA (defined as lower than three times the upper limit of normal) gets 2 points. A "high-positive" of either test gets 3 points.
– A normal CRP and normal ESR get 0 points, whereas at least one abnormal test gets 1 point.
– Symptom duration of fewer than 6 weeks gets 0 points; duration of 6 weeks or longer gets 1 point.
Scores of 6 or more out of 10 are classified as "definite RA."
Commenting on the new criteria in an interview, Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic in Rochester, Minn., and was not involved in the study, said, "A major useful feature is that the new guidelines do not require multiple joints to be inflamed before a diagnosis can be [made] of early inflammatory rheumatoid arthritis."
Indeed, a patient may score 6 points without multiple joint inflammation, according to the new guidelines.
The authors also pointed out that symmetry is not a criterion for diagnosis, as it did not show significance in either phase of the new guidelines’ development. Nevertheless, they wrote, "Inevitably ... the greater the number of involved joints the higher the likelihood of bilateral involvement."
When Dr. Matteson was asked what was missing from the new guidelines, he pointed to a lack of awareness of extra-articular components of RA, which also can occur early in the course of the disease.
"When they do [occur], they can be very useful in identifying the disease, and they are important markers and predictors of disease severity and need for therapy," he said.
The guidelines also lack biomarkers for treatment response, he added.
Several of the guideline authors disclosed financial and other relationships with multiple pharmaceutical companies. Dr. Matteson stated that he had no financial disclosures relative to his comments.
In an editorial accompanying the guidelines in the Annals of the Rheumatic Diseases, ACR President Stanley Cohen and EULAR President Paul Emery said that the 1987 criteria, although useful for differentiating “established” RA from other rheumatologic diseases, are “widely regarded as unsatisfactory for the diagnosis of RA (for which they were not designed)” (Ann. Rheum. Dis. 2010;69:1575-6).
However, in 2010, “the need for the new classification criteria has been made more urgent by the understanding that, at presentation, RA may be an evolving disease, the final phenotype of which can be altered by interventions.”
And despite the fact that “change can be difficult for a generation of rheumatologists used to classifying RA with the old criteria,” the authors were hopeful that “these new classification criteria will be rapidly adopted in daily practice, and we look forward to their implementation in clinical trials.”
Indeed, they added, “How these criteria might impact patient selection for clinical trials will be of great interest.”
Dr. Cohen is affiliated with the clinical research center at the University of Texas Southwestern Medical School in Dallas. Dr. Emery is from the University of Leeds (England). Both stated that they had no competing interests to disclose in relation to this editorial.
In an editorial accompanying the guidelines in the Annals of the Rheumatic Diseases, ACR President Stanley Cohen and EULAR President Paul Emery said that the 1987 criteria, although useful for differentiating “established” RA from other rheumatologic diseases, are “widely regarded as unsatisfactory for the diagnosis of RA (for which they were not designed)” (Ann. Rheum. Dis. 2010;69:1575-6).
However, in 2010, “the need for the new classification criteria has been made more urgent by the understanding that, at presentation, RA may be an evolving disease, the final phenotype of which can be altered by interventions.”
And despite the fact that “change can be difficult for a generation of rheumatologists used to classifying RA with the old criteria,” the authors were hopeful that “these new classification criteria will be rapidly adopted in daily practice, and we look forward to their implementation in clinical trials.”
Indeed, they added, “How these criteria might impact patient selection for clinical trials will be of great interest.”
Dr. Cohen is affiliated with the clinical research center at the University of Texas Southwestern Medical School in Dallas. Dr. Emery is from the University of Leeds (England). Both stated that they had no competing interests to disclose in relation to this editorial.
In an editorial accompanying the guidelines in the Annals of the Rheumatic Diseases, ACR President Stanley Cohen and EULAR President Paul Emery said that the 1987 criteria, although useful for differentiating “established” RA from other rheumatologic diseases, are “widely regarded as unsatisfactory for the diagnosis of RA (for which they were not designed)” (Ann. Rheum. Dis. 2010;69:1575-6).
However, in 2010, “the need for the new classification criteria has been made more urgent by the understanding that, at presentation, RA may be an evolving disease, the final phenotype of which can be altered by interventions.”
And despite the fact that “change can be difficult for a generation of rheumatologists used to classifying RA with the old criteria,” the authors were hopeful that “these new classification criteria will be rapidly adopted in daily practice, and we look forward to their implementation in clinical trials.”
Indeed, they added, “How these criteria might impact patient selection for clinical trials will be of great interest.”
Dr. Cohen is affiliated with the clinical research center at the University of Texas Southwestern Medical School in Dallas. Dr. Emery is from the University of Leeds (England). Both stated that they had no competing interests to disclose in relation to this editorial.
The promised overhaul of treatment guidelines for rheumatoid arthritis has finally arrived, and with it, a "new paradigm" that focuses on early identification and treatment of the disabling disease.
The guidelines, which were developed by a joint committee from the American College of Rheumatology and the European League Against Rheumatism, are the latest update since the current guidelines were created in 1987.
Published jointly in the EULAR journal Annals of the Rheumatic Diseases (2010;69:1580-8) and the ACR's Arthritis and Rheumatism (2010;62:2569-81), the new guidelines were created in three phases over 2 years.
In the first phase, the goal was to "to identify the contributions of clinical and laboratory variables that in practice were the most predictive of the decision to initiate [disease-modifying antirheumatic drug] therapy in a population of patients with early undifferentiated synovitis," wrote the authors, led by Dr. Daniel Aletaha of the Medical University of Vienna.
To do this, a working group from both societies looked at data from 3,115 patients, and correlated whether or not the patients were ultimately prescribed methotrexate to an "agreed-upon list of standardized clinical and laboratory variables collected at baseline."
The odds of eventual methotrexate initiation were calculated for each variable. For example, swelling of the metacarpophalangeal joint had an odds ratio of 1.5, as did swelling of the proximal interphalangeal joint and the wrist. Tenderness of the hand (either the MCP, PIP, or wrist) was assigned an odds ratio of 2.0.
A moderate elevation of C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR) was assigned an OR of 1.0; a high elevation of either assay carried an OR of 2.0.
Finally, moderate levels of either rheumatoid factor or anti–citrullinated protein antibodies had an OR of 2.0 for the eventual prescription of a DMARD; high levels had an OR of 4.0.
In phase II, an "expert panel" of 12 rheumatologists related the above clinical and laboratory factors to the "probability of developing 'persistent inflammatory and/or erosive arthritis that is currently considered to be RA.'?"
The panel also looked at duration of symptoms (longer or shorter than 6 weeks) and the number and size of joints (large or small), in addition to the variables that were assessed in phase I. Using a computer program, the panel assigned each variable a point value of 1-100, with high scores indicating greater likelihood of RA.
Finally, phase III aimed to utilize the results of phases I and II "to develop a scoring system that would be applicable to newly presenting patients with undifferentiated inflammatory arthritis, to permit identification of those with a high probability of developing persistent and/or erosive RA."
This final scale assigns points in the following manner:
– One swollen "large joint" (defined as shoulders, elbows, hips, knees, and ankles) gets 0 points; involvement of 2-10 large joints gets 1 point.
– Involvement of 1-3 "small" joints (defined as metacarpophalangeal joints, proximal interphalangeal joints, second to fifth metatarsophalangeal joints, thumb interphalangeal joints, and wrists) gets 2 points, regardless of large-joint involvement; involvement of 4-10 small joints gets 3 points.
– Involvement of more than 10 joints, including at least one small joint, gets 5 points.
– Both a negative rheumatoid factor (RF) test and a negative anti–citrullinated protein antibody test (ACPA) gets 0 points, whereas having a "low-positive" RF or ACPA (defined as lower than three times the upper limit of normal) gets 2 points. A "high-positive" of either test gets 3 points.
– A normal CRP and normal ESR get 0 points, whereas at least one abnormal test gets 1 point.
– Symptom duration of fewer than 6 weeks gets 0 points; duration of 6 weeks or longer gets 1 point.
Scores of 6 or more out of 10 are classified as "definite RA."
Commenting on the new criteria in an interview, Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic in Rochester, Minn., and was not involved in the study, said, "A major useful feature is that the new guidelines do not require multiple joints to be inflamed before a diagnosis can be [made] of early inflammatory rheumatoid arthritis."
Indeed, a patient may score 6 points without multiple joint inflammation, according to the new guidelines.
The authors also pointed out that symmetry is not a criterion for diagnosis, as it did not show significance in either phase of the new guidelines’ development. Nevertheless, they wrote, "Inevitably ... the greater the number of involved joints the higher the likelihood of bilateral involvement."
When Dr. Matteson was asked what was missing from the new guidelines, he pointed to a lack of awareness of extra-articular components of RA, which also can occur early in the course of the disease.
"When they do [occur], they can be very useful in identifying the disease, and they are important markers and predictors of disease severity and need for therapy," he said.
The guidelines also lack biomarkers for treatment response, he added.
Several of the guideline authors disclosed financial and other relationships with multiple pharmaceutical companies. Dr. Matteson stated that he had no financial disclosures relative to his comments.
The promised overhaul of treatment guidelines for rheumatoid arthritis has finally arrived, and with it, a "new paradigm" that focuses on early identification and treatment of the disabling disease.
The guidelines, which were developed by a joint committee from the American College of Rheumatology and the European League Against Rheumatism, are the latest update since the current guidelines were created in 1987.
Published jointly in the EULAR journal Annals of the Rheumatic Diseases (2010;69:1580-8) and the ACR's Arthritis and Rheumatism (2010;62:2569-81), the new guidelines were created in three phases over 2 years.
In the first phase, the goal was to "to identify the contributions of clinical and laboratory variables that in practice were the most predictive of the decision to initiate [disease-modifying antirheumatic drug] therapy in a population of patients with early undifferentiated synovitis," wrote the authors, led by Dr. Daniel Aletaha of the Medical University of Vienna.
To do this, a working group from both societies looked at data from 3,115 patients, and correlated whether or not the patients were ultimately prescribed methotrexate to an "agreed-upon list of standardized clinical and laboratory variables collected at baseline."
The odds of eventual methotrexate initiation were calculated for each variable. For example, swelling of the metacarpophalangeal joint had an odds ratio of 1.5, as did swelling of the proximal interphalangeal joint and the wrist. Tenderness of the hand (either the MCP, PIP, or wrist) was assigned an odds ratio of 2.0.
A moderate elevation of C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR) was assigned an OR of 1.0; a high elevation of either assay carried an OR of 2.0.
Finally, moderate levels of either rheumatoid factor or anti–citrullinated protein antibodies had an OR of 2.0 for the eventual prescription of a DMARD; high levels had an OR of 4.0.
In phase II, an "expert panel" of 12 rheumatologists related the above clinical and laboratory factors to the "probability of developing 'persistent inflammatory and/or erosive arthritis that is currently considered to be RA.'?"
The panel also looked at duration of symptoms (longer or shorter than 6 weeks) and the number and size of joints (large or small), in addition to the variables that were assessed in phase I. Using a computer program, the panel assigned each variable a point value of 1-100, with high scores indicating greater likelihood of RA.
Finally, phase III aimed to utilize the results of phases I and II "to develop a scoring system that would be applicable to newly presenting patients with undifferentiated inflammatory arthritis, to permit identification of those with a high probability of developing persistent and/or erosive RA."
This final scale assigns points in the following manner:
– One swollen "large joint" (defined as shoulders, elbows, hips, knees, and ankles) gets 0 points; involvement of 2-10 large joints gets 1 point.
– Involvement of 1-3 "small" joints (defined as metacarpophalangeal joints, proximal interphalangeal joints, second to fifth metatarsophalangeal joints, thumb interphalangeal joints, and wrists) gets 2 points, regardless of large-joint involvement; involvement of 4-10 small joints gets 3 points.
– Involvement of more than 10 joints, including at least one small joint, gets 5 points.
– Both a negative rheumatoid factor (RF) test and a negative anti–citrullinated protein antibody test (ACPA) gets 0 points, whereas having a "low-positive" RF or ACPA (defined as lower than three times the upper limit of normal) gets 2 points. A "high-positive" of either test gets 3 points.
– A normal CRP and normal ESR get 0 points, whereas at least one abnormal test gets 1 point.
– Symptom duration of fewer than 6 weeks gets 0 points; duration of 6 weeks or longer gets 1 point.
Scores of 6 or more out of 10 are classified as "definite RA."
Commenting on the new criteria in an interview, Dr. Eric L. Matteson, who is a professor of medicine at the division of rheumatology at the Mayo Clinic in Rochester, Minn., and was not involved in the study, said, "A major useful feature is that the new guidelines do not require multiple joints to be inflamed before a diagnosis can be [made] of early inflammatory rheumatoid arthritis."
Indeed, a patient may score 6 points without multiple joint inflammation, according to the new guidelines.
The authors also pointed out that symmetry is not a criterion for diagnosis, as it did not show significance in either phase of the new guidelines’ development. Nevertheless, they wrote, "Inevitably ... the greater the number of involved joints the higher the likelihood of bilateral involvement."
When Dr. Matteson was asked what was missing from the new guidelines, he pointed to a lack of awareness of extra-articular components of RA, which also can occur early in the course of the disease.
"When they do [occur], they can be very useful in identifying the disease, and they are important markers and predictors of disease severity and need for therapy," he said.
The guidelines also lack biomarkers for treatment response, he added.
Several of the guideline authors disclosed financial and other relationships with multiple pharmaceutical companies. Dr. Matteson stated that he had no financial disclosures relative to his comments.
Major Finding: A new criteria set for rheumatoid arthritis identifies early disease based on a score of 6 or higher out of 10.
Data Source: An international committee of rheumatologists from both the European League Against Rheumatism and the American College of Rheumatology.
Disclosures: Several of the authors of the guidelines disclosed financial relationships with multiple drug makers.
TNF Blockers Raise Atypical Mycobacteria Infection Risk
PORTLAND, Ore. - Patients with suspicious, productive coughs should be worked up to rule out atypical mycobacterial infections before they are treated with tumor necrosis factor inhibitors, even if their chest x-rays and tuberculosis tests are negative, according to Dr. Kevin Winthrop.
"These biologics seem to promote mycobacterial growth; you have to be careful an infection isn’t hiding," he said at the annual meeting of the Society for Pediatric Dermatology.
Chest x-rays are not sensitive enough to pick up infection by Micobacterium avium and other nontuberculous mycobacteria (NTM), which are twice as likely to cause pulmonary infection in the United States as M. tuberculosis is, Dr. Winthrop of the division of infectious diseases at Oregon Health and Science University, Portland, said in a later interview.
Chest computed tomography and sputum culture are typically needed to make the diagnosis.
It's important information for dermatologists because tumor necrosis factor (TNF) inhibitors are being used “more and more in dermatology,” he said.
Adalimumab, etanercept, and infliximab are indicated for plaque psoriasis; those and other TNF inhibitors have been tried off label for many ailments with dermatologic manifestations, including Behçet’s disease, Crohn’s disease, dermatomyositis, and scleroderma (J. Cutan. Med. Surg. 2005;9:296-302).
Black boxes on product labels warn that TNF inhibition increases the risk of TB, but the risk of NTM infection is not similarly emphasized.
Dr. Winthrop and his colleagues identified 105 anti-TNF therapy–associated NTM infections in FDA’s MedWatch database between 1999 and 2006; 56% were pulmonary, 26% skin and soft tissue, 9% bone and joint, and 8% disseminated. There was one eye infection; overall, M. avium caused half of the infections (Emerg. Infect. Dis. 2009:15:1556-61).
Most of the patients were older women treated for rheumatoid arthritis; only a few of the cases were associated with psoriasis therapy, but TNF inhibitors were not indicated for psoriasis during most of the study period.
Seventy-three infections in the MedWatch database were associated with infliximab, 25 with etanercept, and 7 with adalimumab.
"Use of infliximab may pose a greater risk for NTM disease. If true, the risk could be caused by the drug itself or differences in the characteristics of patients given infliximab relative to users of the other anti-TNF-alpha compounds," Dr. Winthrop and his colleagues wrote.
"Infliximab users were more likely to be concomitantly using methotrexate at the time of diagnosis," they said.
During his presentation at the annual meeting, Dr. Winthrop said he suspects the number of anti-TNF–associated NTM infections is "much lower" in psoriasis than in rheumatoid arthritis, which can affect the lungs and increase susceptibility to opportunistic infection by nontuberculous mycobacteria, which are ubiquitous in soil and water.
He and his colleagues noted in their report, however, that MedWatch – a voluntary reporting system – likely underestimated the incidence of TNF blocker–associated NTM infection.
A large, epidemiologic safety study of TNF inhibitors and other biologics is underway, and should further define the risks of NTM infections in dermatology patients, said Dr. Winthrop, a coinvestigator in the project. Results could begin to be published in 2011.
He said M. avium is most likely to infect the lungs, while Micobacterium abscessus, Micobacterium chelonae, Micobacterium marinum, and Micobacterium fortuitum are more likely to infect skin and soft tissue.
Micobacterium kansasii is a likely pathogen of both skin and lung in the Southern United States, he said.
As with pulmonary infections, NTM skin infections are difficult to diagnose; diagnosis typically requires a punch biopsy along with a culture or polymerase chain reaction analysis.
NTM infections do not typically respond to TB antibiotics; other antibiotics must be used in combination, Dr. Winthrop said.
Pulmonary infections typically are treated for 18 months, longer than for TB, because NTM infections are generally less susceptible to antibiotics.
Disclosures: Dr. Winthrop said he has no conflicts of interests.
PORTLAND, Ore. - Patients with suspicious, productive coughs should be worked up to rule out atypical mycobacterial infections before they are treated with tumor necrosis factor inhibitors, even if their chest x-rays and tuberculosis tests are negative, according to Dr. Kevin Winthrop.
"These biologics seem to promote mycobacterial growth; you have to be careful an infection isn’t hiding," he said at the annual meeting of the Society for Pediatric Dermatology.
Chest x-rays are not sensitive enough to pick up infection by Micobacterium avium and other nontuberculous mycobacteria (NTM), which are twice as likely to cause pulmonary infection in the United States as M. tuberculosis is, Dr. Winthrop of the division of infectious diseases at Oregon Health and Science University, Portland, said in a later interview.
Chest computed tomography and sputum culture are typically needed to make the diagnosis.
It's important information for dermatologists because tumor necrosis factor (TNF) inhibitors are being used “more and more in dermatology,” he said.
Adalimumab, etanercept, and infliximab are indicated for plaque psoriasis; those and other TNF inhibitors have been tried off label for many ailments with dermatologic manifestations, including Behçet’s disease, Crohn’s disease, dermatomyositis, and scleroderma (J. Cutan. Med. Surg. 2005;9:296-302).
Black boxes on product labels warn that TNF inhibition increases the risk of TB, but the risk of NTM infection is not similarly emphasized.
Dr. Winthrop and his colleagues identified 105 anti-TNF therapy–associated NTM infections in FDA’s MedWatch database between 1999 and 2006; 56% were pulmonary, 26% skin and soft tissue, 9% bone and joint, and 8% disseminated. There was one eye infection; overall, M. avium caused half of the infections (Emerg. Infect. Dis. 2009:15:1556-61).
Most of the patients were older women treated for rheumatoid arthritis; only a few of the cases were associated with psoriasis therapy, but TNF inhibitors were not indicated for psoriasis during most of the study period.
Seventy-three infections in the MedWatch database were associated with infliximab, 25 with etanercept, and 7 with adalimumab.
"Use of infliximab may pose a greater risk for NTM disease. If true, the risk could be caused by the drug itself or differences in the characteristics of patients given infliximab relative to users of the other anti-TNF-alpha compounds," Dr. Winthrop and his colleagues wrote.
"Infliximab users were more likely to be concomitantly using methotrexate at the time of diagnosis," they said.
During his presentation at the annual meeting, Dr. Winthrop said he suspects the number of anti-TNF–associated NTM infections is "much lower" in psoriasis than in rheumatoid arthritis, which can affect the lungs and increase susceptibility to opportunistic infection by nontuberculous mycobacteria, which are ubiquitous in soil and water.
He and his colleagues noted in their report, however, that MedWatch – a voluntary reporting system – likely underestimated the incidence of TNF blocker–associated NTM infection.
A large, epidemiologic safety study of TNF inhibitors and other biologics is underway, and should further define the risks of NTM infections in dermatology patients, said Dr. Winthrop, a coinvestigator in the project. Results could begin to be published in 2011.
He said M. avium is most likely to infect the lungs, while Micobacterium abscessus, Micobacterium chelonae, Micobacterium marinum, and Micobacterium fortuitum are more likely to infect skin and soft tissue.
Micobacterium kansasii is a likely pathogen of both skin and lung in the Southern United States, he said.
As with pulmonary infections, NTM skin infections are difficult to diagnose; diagnosis typically requires a punch biopsy along with a culture or polymerase chain reaction analysis.
NTM infections do not typically respond to TB antibiotics; other antibiotics must be used in combination, Dr. Winthrop said.
Pulmonary infections typically are treated for 18 months, longer than for TB, because NTM infections are generally less susceptible to antibiotics.
Disclosures: Dr. Winthrop said he has no conflicts of interests.
PORTLAND, Ore. - Patients with suspicious, productive coughs should be worked up to rule out atypical mycobacterial infections before they are treated with tumor necrosis factor inhibitors, even if their chest x-rays and tuberculosis tests are negative, according to Dr. Kevin Winthrop.
"These biologics seem to promote mycobacterial growth; you have to be careful an infection isn’t hiding," he said at the annual meeting of the Society for Pediatric Dermatology.
Chest x-rays are not sensitive enough to pick up infection by Micobacterium avium and other nontuberculous mycobacteria (NTM), which are twice as likely to cause pulmonary infection in the United States as M. tuberculosis is, Dr. Winthrop of the division of infectious diseases at Oregon Health and Science University, Portland, said in a later interview.
Chest computed tomography and sputum culture are typically needed to make the diagnosis.
It's important information for dermatologists because tumor necrosis factor (TNF) inhibitors are being used “more and more in dermatology,” he said.
Adalimumab, etanercept, and infliximab are indicated for plaque psoriasis; those and other TNF inhibitors have been tried off label for many ailments with dermatologic manifestations, including Behçet’s disease, Crohn’s disease, dermatomyositis, and scleroderma (J. Cutan. Med. Surg. 2005;9:296-302).
Black boxes on product labels warn that TNF inhibition increases the risk of TB, but the risk of NTM infection is not similarly emphasized.
Dr. Winthrop and his colleagues identified 105 anti-TNF therapy–associated NTM infections in FDA’s MedWatch database between 1999 and 2006; 56% were pulmonary, 26% skin and soft tissue, 9% bone and joint, and 8% disseminated. There was one eye infection; overall, M. avium caused half of the infections (Emerg. Infect. Dis. 2009:15:1556-61).
Most of the patients were older women treated for rheumatoid arthritis; only a few of the cases were associated with psoriasis therapy, but TNF inhibitors were not indicated for psoriasis during most of the study period.
Seventy-three infections in the MedWatch database were associated with infliximab, 25 with etanercept, and 7 with adalimumab.
"Use of infliximab may pose a greater risk for NTM disease. If true, the risk could be caused by the drug itself or differences in the characteristics of patients given infliximab relative to users of the other anti-TNF-alpha compounds," Dr. Winthrop and his colleagues wrote.
"Infliximab users were more likely to be concomitantly using methotrexate at the time of diagnosis," they said.
During his presentation at the annual meeting, Dr. Winthrop said he suspects the number of anti-TNF–associated NTM infections is "much lower" in psoriasis than in rheumatoid arthritis, which can affect the lungs and increase susceptibility to opportunistic infection by nontuberculous mycobacteria, which are ubiquitous in soil and water.
He and his colleagues noted in their report, however, that MedWatch – a voluntary reporting system – likely underestimated the incidence of TNF blocker–associated NTM infection.
A large, epidemiologic safety study of TNF inhibitors and other biologics is underway, and should further define the risks of NTM infections in dermatology patients, said Dr. Winthrop, a coinvestigator in the project. Results could begin to be published in 2011.
He said M. avium is most likely to infect the lungs, while Micobacterium abscessus, Micobacterium chelonae, Micobacterium marinum, and Micobacterium fortuitum are more likely to infect skin and soft tissue.
Micobacterium kansasii is a likely pathogen of both skin and lung in the Southern United States, he said.
As with pulmonary infections, NTM skin infections are difficult to diagnose; diagnosis typically requires a punch biopsy along with a culture or polymerase chain reaction analysis.
NTM infections do not typically respond to TB antibiotics; other antibiotics must be used in combination, Dr. Winthrop said.
Pulmonary infections typically are treated for 18 months, longer than for TB, because NTM infections are generally less susceptible to antibiotics.
Disclosures: Dr. Winthrop said he has no conflicts of interests.
BASDAI as Good as ASDAS, but Easier to Use
Two scores used to measure disease activity in axial psoriatic arthritis are similarly accurate, but the older measure is easier to employ and thus, probably more clinically practical, a study has shown.
A comparative study determined that the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), with its single patient-derived format of six questions, is just as accurate as the newer Ankylosing Spondylitis Disease Activity Score (ASDAS). That measure has four formulas including two that assess both C-reactive protein (CRP) level and erythrocyte sedimentation rate (ESR), and two that assess each of the factors separately.
ASDAS was introduced in 2007 because researchers felt the BASDAI had limited face and construct validity, reported Dr. Lihi Eder and colleagues (Ann. Rheum. Dis. 2010 July 13 [doi:10.1136/ard.2010.129726]).
Dr. Eder of Toronto Western Hospital and the coauthors compared the scores’ validity in 201 patients with axial psoriatic arthritis. Their mean age was 53 years; the mean duration of psoriatic arthritis was 18.5 years, with a mean of 9 years since the diagnosis of axial involvement. For each patient, the researchers calculated the BASDAI and ASDAS and correlated those with the patients’ and their physicians’ rating of disease.
The BASDAI includes patient rating of six aspects of disease: fatigue, total back pain, pain and swelling of peripheral joints, pain at entheseal sites, severity of morning stiffness, and duration of morning stiffness. The ASDAS involves only three of those factors: total back pain, pain and swelling of peripheral joints, and duration of morning stiffness. All patients gave a global assessment of their disease activity and were examined by a rheumatologist who gave a global assessment of disease activity as well.
Both the patient and physician global ratings are given on a scale of 0-10 with 10 being the most severe disease. Any patient with either a patient or physician global assessment score on either scale equal to or above 6 was considered to have highly active disease.
Both the BASDAI and ASDAS correlated well with disease activity, which is not surprising given that both consist largely of patient-derived information, the investigators said. Physician global assessment correlated less well with BASDAI and ASDAS.
Both scores were able to discriminate between high and low disease activity, an important function in that the scores are used to guide treatment decisions.
The addition of the CRP and ESR components to the ASDAS score showed poor correlation with both patient and physician ratings of disease activity. "While CRP showed only marginally significant and weakly positive correlation with patient or physician global scores, ESR did not show any significant correlation with either of those scores," the authors noted.
In a logistic regression analysis, both scores discriminated well between high and low disease activity. "As expected, the scores showed better discrimination when the definition of the disease activity was based on patient- rather than physician-derived scores," the authors wrote.
Because the scores are similarly accurate, the authors endorsed the BASDAI score on the basis of its relative simplicity. "The ASDAS score did not improve discriminative ability compared with BASDAI," they wrote. “Therefore, because BASDAI is easier to calculate, it may be more practical for clinical use in patients with axial psoriatic arthritis."
Disclosures: The study was funded by the University of Toronto Psoriatic Arthritis Program. The authors said they had no conflicts of interest.
Two scores used to measure disease activity in axial psoriatic arthritis are similarly accurate, but the older measure is easier to employ and thus, probably more clinically practical, a study has shown.
A comparative study determined that the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), with its single patient-derived format of six questions, is just as accurate as the newer Ankylosing Spondylitis Disease Activity Score (ASDAS). That measure has four formulas including two that assess both C-reactive protein (CRP) level and erythrocyte sedimentation rate (ESR), and two that assess each of the factors separately.
ASDAS was introduced in 2007 because researchers felt the BASDAI had limited face and construct validity, reported Dr. Lihi Eder and colleagues (Ann. Rheum. Dis. 2010 July 13 [doi:10.1136/ard.2010.129726]).
Dr. Eder of Toronto Western Hospital and the coauthors compared the scores’ validity in 201 patients with axial psoriatic arthritis. Their mean age was 53 years; the mean duration of psoriatic arthritis was 18.5 years, with a mean of 9 years since the diagnosis of axial involvement. For each patient, the researchers calculated the BASDAI and ASDAS and correlated those with the patients’ and their physicians’ rating of disease.
The BASDAI includes patient rating of six aspects of disease: fatigue, total back pain, pain and swelling of peripheral joints, pain at entheseal sites, severity of morning stiffness, and duration of morning stiffness. The ASDAS involves only three of those factors: total back pain, pain and swelling of peripheral joints, and duration of morning stiffness. All patients gave a global assessment of their disease activity and were examined by a rheumatologist who gave a global assessment of disease activity as well.
Both the patient and physician global ratings are given on a scale of 0-10 with 10 being the most severe disease. Any patient with either a patient or physician global assessment score on either scale equal to or above 6 was considered to have highly active disease.
Both the BASDAI and ASDAS correlated well with disease activity, which is not surprising given that both consist largely of patient-derived information, the investigators said. Physician global assessment correlated less well with BASDAI and ASDAS.
Both scores were able to discriminate between high and low disease activity, an important function in that the scores are used to guide treatment decisions.
The addition of the CRP and ESR components to the ASDAS score showed poor correlation with both patient and physician ratings of disease activity. "While CRP showed only marginally significant and weakly positive correlation with patient or physician global scores, ESR did not show any significant correlation with either of those scores," the authors noted.
In a logistic regression analysis, both scores discriminated well between high and low disease activity. "As expected, the scores showed better discrimination when the definition of the disease activity was based on patient- rather than physician-derived scores," the authors wrote.
Because the scores are similarly accurate, the authors endorsed the BASDAI score on the basis of its relative simplicity. "The ASDAS score did not improve discriminative ability compared with BASDAI," they wrote. “Therefore, because BASDAI is easier to calculate, it may be more practical for clinical use in patients with axial psoriatic arthritis."
Disclosures: The study was funded by the University of Toronto Psoriatic Arthritis Program. The authors said they had no conflicts of interest.
Two scores used to measure disease activity in axial psoriatic arthritis are similarly accurate, but the older measure is easier to employ and thus, probably more clinically practical, a study has shown.
A comparative study determined that the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), with its single patient-derived format of six questions, is just as accurate as the newer Ankylosing Spondylitis Disease Activity Score (ASDAS). That measure has four formulas including two that assess both C-reactive protein (CRP) level and erythrocyte sedimentation rate (ESR), and two that assess each of the factors separately.
ASDAS was introduced in 2007 because researchers felt the BASDAI had limited face and construct validity, reported Dr. Lihi Eder and colleagues (Ann. Rheum. Dis. 2010 July 13 [doi:10.1136/ard.2010.129726]).
Dr. Eder of Toronto Western Hospital and the coauthors compared the scores’ validity in 201 patients with axial psoriatic arthritis. Their mean age was 53 years; the mean duration of psoriatic arthritis was 18.5 years, with a mean of 9 years since the diagnosis of axial involvement. For each patient, the researchers calculated the BASDAI and ASDAS and correlated those with the patients’ and their physicians’ rating of disease.
The BASDAI includes patient rating of six aspects of disease: fatigue, total back pain, pain and swelling of peripheral joints, pain at entheseal sites, severity of morning stiffness, and duration of morning stiffness. The ASDAS involves only three of those factors: total back pain, pain and swelling of peripheral joints, and duration of morning stiffness. All patients gave a global assessment of their disease activity and were examined by a rheumatologist who gave a global assessment of disease activity as well.
Both the patient and physician global ratings are given on a scale of 0-10 with 10 being the most severe disease. Any patient with either a patient or physician global assessment score on either scale equal to or above 6 was considered to have highly active disease.
Both the BASDAI and ASDAS correlated well with disease activity, which is not surprising given that both consist largely of patient-derived information, the investigators said. Physician global assessment correlated less well with BASDAI and ASDAS.
Both scores were able to discriminate between high and low disease activity, an important function in that the scores are used to guide treatment decisions.
The addition of the CRP and ESR components to the ASDAS score showed poor correlation with both patient and physician ratings of disease activity. "While CRP showed only marginally significant and weakly positive correlation with patient or physician global scores, ESR did not show any significant correlation with either of those scores," the authors noted.
In a logistic regression analysis, both scores discriminated well between high and low disease activity. "As expected, the scores showed better discrimination when the definition of the disease activity was based on patient- rather than physician-derived scores," the authors wrote.
Because the scores are similarly accurate, the authors endorsed the BASDAI score on the basis of its relative simplicity. "The ASDAS score did not improve discriminative ability compared with BASDAI," they wrote. “Therefore, because BASDAI is easier to calculate, it may be more practical for clinical use in patients with axial psoriatic arthritis."
Disclosures: The study was funded by the University of Toronto Psoriatic Arthritis Program. The authors said they had no conflicts of interest.
Dark Beer Ups Psoriasis Risk in Women
Women who drink more than two drinks per week are significantly more likely to develop psoriasis than are women who abstain from alcohol.
Moreover, when stratified by type of alcohol consumed, it is full-calorie, non-light beer – not wine, liquor, or light beer – that appears to raise the risk for the skin condition, according to a study of over 82,000 women published online in the Archives of Dermatology.
Dr. Abrar A. Qureshi, director of the Translational Research Resource Center in the department of dermatology at Brigham and Women’s Hospital, Boston, looked at 116,430 female registered nurses from the Nurses’ Health Study II (an ongoing longitudinal study begun in 1989).
The nurses were asked about whether they had ever had a diagnosis of psoriasis between 1991 and 2005, with 1991 being the first year in the study in which alcohol intake data patterns were assessed. Weekly drinking was also assessed in 1995, 1999, and 2003 (doi:10.1001/archdermatol.2010.204).
Overall, among the 47,614 women who responded to the survey and reported consuming alcohol, the mean age was 36 years. It was the same for the 35,058 abstainers who responded.
The only differences between the cohorts were that the abstainers had a slightly higher body mass index (BMI) and were less physically active; drinkers were more likely to smoke or have ever smoked.
Dr. Qureshi and his colleagues found that with adjustment for age only, there was a 1.89 relative risk of developing psoriasis among patients who reported drinking more than 2.3 drinks per week (95% confidence interval, 1.29-2.77).
When adjusted for age, smoking, BMI, dietary folate, and physical exercise, the risk dropped, but only slightly, to 1.72 (95% CI, 1.15-2.57).
Furthermore, when stratified by type of alcohol, having five or more glasses of non-light beer per week was the only beverage to be significantly associated with incident psoriasis, after the multivariate adjustment (RR 1.76; 95% CI 1.15-2.69). Five or more glasses of light beer, white wine, or red wine were not significantly associated with psoriasis, nor were two or more glasses per week of liquor.
Finally, the authors analyzed alcohol intake and confirmed psoriasis according to the Psoriasis Screening Tool questionnaire, a one-page, self-administered, seven-question survey sent to all study participants who reported prior psoriasis diagnosis.
Among this subgroup of confirmed cases, after the same multivariate adjustment used in the earlier analysis, the relative risk associated with any alcohol intake above 2.3 drinks per week was even more pronounced – 2.54 (95% CI, 1.57-4.10), and for 5 or more non-light beer drinks per week, it was 2.29 (95% CI, 1.36-3.85).
The authors postulated that beer’s gluten content could be the culprit, since gluten has been tied to psoriasis in prior studies, and beer is one of the few nondistilled alcoholic beverages to use a starch source for fermentation – barley, most commonly. However, light beer also is made with grain and therefore contains gluten, albeit lower amounts.
One major limitation of the study, according to Dr. Qureshi, is the retrospective recall of psoriasis onset, which might have led to misclassification of some psoriasis incidents. Additionally, the researchers wrote, “This well-educated female cohort provides high-quality data with little loss to follow-up but does not represent a random sample of U.S. women,” nor of alcohol consumption rates.
Nevertheless, they added, “the biological effects of alcohol intake on psoriasis should be similar.”
Dr. Qureshi disclosed serving as a consultant to pharmaceutical makers Amgen and Genentech; the study was funded by grants from the National Institutes of Health/National Cancer Institute.
Women who drink more than two drinks per week are significantly more likely to develop psoriasis than are women who abstain from alcohol.
Moreover, when stratified by type of alcohol consumed, it is full-calorie, non-light beer – not wine, liquor, or light beer – that appears to raise the risk for the skin condition, according to a study of over 82,000 women published online in the Archives of Dermatology.
Dr. Abrar A. Qureshi, director of the Translational Research Resource Center in the department of dermatology at Brigham and Women’s Hospital, Boston, looked at 116,430 female registered nurses from the Nurses’ Health Study II (an ongoing longitudinal study begun in 1989).
The nurses were asked about whether they had ever had a diagnosis of psoriasis between 1991 and 2005, with 1991 being the first year in the study in which alcohol intake data patterns were assessed. Weekly drinking was also assessed in 1995, 1999, and 2003 (doi:10.1001/archdermatol.2010.204).
Overall, among the 47,614 women who responded to the survey and reported consuming alcohol, the mean age was 36 years. It was the same for the 35,058 abstainers who responded.
The only differences between the cohorts were that the abstainers had a slightly higher body mass index (BMI) and were less physically active; drinkers were more likely to smoke or have ever smoked.
Dr. Qureshi and his colleagues found that with adjustment for age only, there was a 1.89 relative risk of developing psoriasis among patients who reported drinking more than 2.3 drinks per week (95% confidence interval, 1.29-2.77).
When adjusted for age, smoking, BMI, dietary folate, and physical exercise, the risk dropped, but only slightly, to 1.72 (95% CI, 1.15-2.57).
Furthermore, when stratified by type of alcohol, having five or more glasses of non-light beer per week was the only beverage to be significantly associated with incident psoriasis, after the multivariate adjustment (RR 1.76; 95% CI 1.15-2.69). Five or more glasses of light beer, white wine, or red wine were not significantly associated with psoriasis, nor were two or more glasses per week of liquor.
Finally, the authors analyzed alcohol intake and confirmed psoriasis according to the Psoriasis Screening Tool questionnaire, a one-page, self-administered, seven-question survey sent to all study participants who reported prior psoriasis diagnosis.
Among this subgroup of confirmed cases, after the same multivariate adjustment used in the earlier analysis, the relative risk associated with any alcohol intake above 2.3 drinks per week was even more pronounced – 2.54 (95% CI, 1.57-4.10), and for 5 or more non-light beer drinks per week, it was 2.29 (95% CI, 1.36-3.85).
The authors postulated that beer’s gluten content could be the culprit, since gluten has been tied to psoriasis in prior studies, and beer is one of the few nondistilled alcoholic beverages to use a starch source for fermentation – barley, most commonly. However, light beer also is made with grain and therefore contains gluten, albeit lower amounts.
One major limitation of the study, according to Dr. Qureshi, is the retrospective recall of psoriasis onset, which might have led to misclassification of some psoriasis incidents. Additionally, the researchers wrote, “This well-educated female cohort provides high-quality data with little loss to follow-up but does not represent a random sample of U.S. women,” nor of alcohol consumption rates.
Nevertheless, they added, “the biological effects of alcohol intake on psoriasis should be similar.”
Dr. Qureshi disclosed serving as a consultant to pharmaceutical makers Amgen and Genentech; the study was funded by grants from the National Institutes of Health/National Cancer Institute.
Women who drink more than two drinks per week are significantly more likely to develop psoriasis than are women who abstain from alcohol.
Moreover, when stratified by type of alcohol consumed, it is full-calorie, non-light beer – not wine, liquor, or light beer – that appears to raise the risk for the skin condition, according to a study of over 82,000 women published online in the Archives of Dermatology.
Dr. Abrar A. Qureshi, director of the Translational Research Resource Center in the department of dermatology at Brigham and Women’s Hospital, Boston, looked at 116,430 female registered nurses from the Nurses’ Health Study II (an ongoing longitudinal study begun in 1989).
The nurses were asked about whether they had ever had a diagnosis of psoriasis between 1991 and 2005, with 1991 being the first year in the study in which alcohol intake data patterns were assessed. Weekly drinking was also assessed in 1995, 1999, and 2003 (doi:10.1001/archdermatol.2010.204).
Overall, among the 47,614 women who responded to the survey and reported consuming alcohol, the mean age was 36 years. It was the same for the 35,058 abstainers who responded.
The only differences between the cohorts were that the abstainers had a slightly higher body mass index (BMI) and were less physically active; drinkers were more likely to smoke or have ever smoked.
Dr. Qureshi and his colleagues found that with adjustment for age only, there was a 1.89 relative risk of developing psoriasis among patients who reported drinking more than 2.3 drinks per week (95% confidence interval, 1.29-2.77).
When adjusted for age, smoking, BMI, dietary folate, and physical exercise, the risk dropped, but only slightly, to 1.72 (95% CI, 1.15-2.57).
Furthermore, when stratified by type of alcohol, having five or more glasses of non-light beer per week was the only beverage to be significantly associated with incident psoriasis, after the multivariate adjustment (RR 1.76; 95% CI 1.15-2.69). Five or more glasses of light beer, white wine, or red wine were not significantly associated with psoriasis, nor were two or more glasses per week of liquor.
Finally, the authors analyzed alcohol intake and confirmed psoriasis according to the Psoriasis Screening Tool questionnaire, a one-page, self-administered, seven-question survey sent to all study participants who reported prior psoriasis diagnosis.
Among this subgroup of confirmed cases, after the same multivariate adjustment used in the earlier analysis, the relative risk associated with any alcohol intake above 2.3 drinks per week was even more pronounced – 2.54 (95% CI, 1.57-4.10), and for 5 or more non-light beer drinks per week, it was 2.29 (95% CI, 1.36-3.85).
The authors postulated that beer’s gluten content could be the culprit, since gluten has been tied to psoriasis in prior studies, and beer is one of the few nondistilled alcoholic beverages to use a starch source for fermentation – barley, most commonly. However, light beer also is made with grain and therefore contains gluten, albeit lower amounts.
One major limitation of the study, according to Dr. Qureshi, is the retrospective recall of psoriasis onset, which might have led to misclassification of some psoriasis incidents. Additionally, the researchers wrote, “This well-educated female cohort provides high-quality data with little loss to follow-up but does not represent a random sample of U.S. women,” nor of alcohol consumption rates.
Nevertheless, they added, “the biological effects of alcohol intake on psoriasis should be similar.”
Dr. Qureshi disclosed serving as a consultant to pharmaceutical makers Amgen and Genentech; the study was funded by grants from the National Institutes of Health/National Cancer Institute.
Immunize Traveling Psoriatics Before Starting Biologic Therapy
CHICAGO — The patient on biologic therapy who must travel and requires immunization with a live attenuated virus or bacteria should be immunized before beginning biologic therapy, according to a case report presented to a symposium that was sponsored by the American College of Rheumatology.
“Think hard about the patient’s life before you put them on biologic therapy, and immunize before you give the medications,” advised Dr. Clifton O. Bingham, who is director of the rheumatology clinics at Johns Hopkins University, Baltimore. However, he acknowledged that this would not always be possible.
The case was a 35-year-old woman with psoriatic arthritis whose work required her to travel to Burkina Faso, a small, landlocked country in West Africa, where yellow fever is endemic. The patient’s skin and articular disease was well controlled with etanercept and methotrexate.
As a practical matter, Dr. Bingham said that the immunomodulators etanercept and methotrexate could be withheld for at least 1 month, and then the patient could be given yellow fever vaccine. He advised waiting 2-3 weeks before restarting the medication. “The disease may flare during that period of time, so that’s the risk you take,” he said.
If the patient was required to travel to the infected area with only 2 weeks’ notice, Dr. Bingham advised providing a letter of medical contraindication to yellow fever vaccination. “That should allow her to enter the country, but you tell her to be really careful.” When a patient is on biological therapy, he said it is important to understand which live vaccinations could potentially reactivate in the setting of immunosuppression.
Vaccines that contain live attenuated viruses or bacteria include: varicella; intranasal influenza/H1N1; measles, mumps, and rubella; yellow fever; oral polio; oral typhoid; vaccinia (smallpox); BCG; and rotavirus.
Disseminated disease has been reported in immunocompromised patients who were immunized with live-virus vaccines. Live viral dissemination has occurred in patients during chemotherapy, and there have been cases of infection in patients receiving immunosuppression for organ transplantation. Live virus dissemination has also been reported in patients with HIV infection. (J. Infect. Dis. 2008;197 [Suppl 2]; Clin. Infect. Dis. 2009;49:1550-6; AIDS Rev.2007;9:173-87).
“So, the recommendation is, with people who are on biologics and people who are immunosuppressed, they should not receive these live vaccinations,” Dr. Bingham said.
Up-to-date vaccine information is available online from the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/pubs/vis/default.htm).
An audience member asked, “Do you think that in some cases it may be a better choice to subject somebody to the risk of an attenuated organism, as opposed to the risk of wild-type disease in an area where the disease may be highly endemic?”
Dr. Bingham said, “The risk of developing yellow fever is about 50 per 100,000 in western Africa, and about 10 per 100,000 in Central and South America. So it is a low risk. But if you delve a little deeper into that information, you find out that many of those patients die. It’s potentially a fatal disease. So you do have to balance the risk.”
Disclosures: Dr. Bingham disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Cypress Bioscience, Genentech, Merck, Novartis, Osiris Therapeutics, Procter & Gamble, Roche, Sonosite, Targeted Genetics, UCB, and Wyeth.
CHICAGO — The patient on biologic therapy who must travel and requires immunization with a live attenuated virus or bacteria should be immunized before beginning biologic therapy, according to a case report presented to a symposium that was sponsored by the American College of Rheumatology.
“Think hard about the patient’s life before you put them on biologic therapy, and immunize before you give the medications,” advised Dr. Clifton O. Bingham, who is director of the rheumatology clinics at Johns Hopkins University, Baltimore. However, he acknowledged that this would not always be possible.
The case was a 35-year-old woman with psoriatic arthritis whose work required her to travel to Burkina Faso, a small, landlocked country in West Africa, where yellow fever is endemic. The patient’s skin and articular disease was well controlled with etanercept and methotrexate.
As a practical matter, Dr. Bingham said that the immunomodulators etanercept and methotrexate could be withheld for at least 1 month, and then the patient could be given yellow fever vaccine. He advised waiting 2-3 weeks before restarting the medication. “The disease may flare during that period of time, so that’s the risk you take,” he said.
If the patient was required to travel to the infected area with only 2 weeks’ notice, Dr. Bingham advised providing a letter of medical contraindication to yellow fever vaccination. “That should allow her to enter the country, but you tell her to be really careful.” When a patient is on biological therapy, he said it is important to understand which live vaccinations could potentially reactivate in the setting of immunosuppression.
Vaccines that contain live attenuated viruses or bacteria include: varicella; intranasal influenza/H1N1; measles, mumps, and rubella; yellow fever; oral polio; oral typhoid; vaccinia (smallpox); BCG; and rotavirus.
Disseminated disease has been reported in immunocompromised patients who were immunized with live-virus vaccines. Live viral dissemination has occurred in patients during chemotherapy, and there have been cases of infection in patients receiving immunosuppression for organ transplantation. Live virus dissemination has also been reported in patients with HIV infection. (J. Infect. Dis. 2008;197 [Suppl 2]; Clin. Infect. Dis. 2009;49:1550-6; AIDS Rev.2007;9:173-87).
“So, the recommendation is, with people who are on biologics and people who are immunosuppressed, they should not receive these live vaccinations,” Dr. Bingham said.
Up-to-date vaccine information is available online from the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/pubs/vis/default.htm).
An audience member asked, “Do you think that in some cases it may be a better choice to subject somebody to the risk of an attenuated organism, as opposed to the risk of wild-type disease in an area where the disease may be highly endemic?”
Dr. Bingham said, “The risk of developing yellow fever is about 50 per 100,000 in western Africa, and about 10 per 100,000 in Central and South America. So it is a low risk. But if you delve a little deeper into that information, you find out that many of those patients die. It’s potentially a fatal disease. So you do have to balance the risk.”
Disclosures: Dr. Bingham disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Cypress Bioscience, Genentech, Merck, Novartis, Osiris Therapeutics, Procter & Gamble, Roche, Sonosite, Targeted Genetics, UCB, and Wyeth.
CHICAGO — The patient on biologic therapy who must travel and requires immunization with a live attenuated virus or bacteria should be immunized before beginning biologic therapy, according to a case report presented to a symposium that was sponsored by the American College of Rheumatology.
“Think hard about the patient’s life before you put them on biologic therapy, and immunize before you give the medications,” advised Dr. Clifton O. Bingham, who is director of the rheumatology clinics at Johns Hopkins University, Baltimore. However, he acknowledged that this would not always be possible.
The case was a 35-year-old woman with psoriatic arthritis whose work required her to travel to Burkina Faso, a small, landlocked country in West Africa, where yellow fever is endemic. The patient’s skin and articular disease was well controlled with etanercept and methotrexate.
As a practical matter, Dr. Bingham said that the immunomodulators etanercept and methotrexate could be withheld for at least 1 month, and then the patient could be given yellow fever vaccine. He advised waiting 2-3 weeks before restarting the medication. “The disease may flare during that period of time, so that’s the risk you take,” he said.
If the patient was required to travel to the infected area with only 2 weeks’ notice, Dr. Bingham advised providing a letter of medical contraindication to yellow fever vaccination. “That should allow her to enter the country, but you tell her to be really careful.” When a patient is on biological therapy, he said it is important to understand which live vaccinations could potentially reactivate in the setting of immunosuppression.
Vaccines that contain live attenuated viruses or bacteria include: varicella; intranasal influenza/H1N1; measles, mumps, and rubella; yellow fever; oral polio; oral typhoid; vaccinia (smallpox); BCG; and rotavirus.
Disseminated disease has been reported in immunocompromised patients who were immunized with live-virus vaccines. Live viral dissemination has occurred in patients during chemotherapy, and there have been cases of infection in patients receiving immunosuppression for organ transplantation. Live virus dissemination has also been reported in patients with HIV infection. (J. Infect. Dis. 2008;197 [Suppl 2]; Clin. Infect. Dis. 2009;49:1550-6; AIDS Rev.2007;9:173-87).
“So, the recommendation is, with people who are on biologics and people who are immunosuppressed, they should not receive these live vaccinations,” Dr. Bingham said.
Up-to-date vaccine information is available online from the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/pubs/vis/default.htm).
An audience member asked, “Do you think that in some cases it may be a better choice to subject somebody to the risk of an attenuated organism, as opposed to the risk of wild-type disease in an area where the disease may be highly endemic?”
Dr. Bingham said, “The risk of developing yellow fever is about 50 per 100,000 in western Africa, and about 10 per 100,000 in Central and South America. So it is a low risk. But if you delve a little deeper into that information, you find out that many of those patients die. It’s potentially a fatal disease. So you do have to balance the risk.”
Disclosures: Dr. Bingham disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Cypress Bioscience, Genentech, Merck, Novartis, Osiris Therapeutics, Procter & Gamble, Roche, Sonosite, Targeted Genetics, UCB, and Wyeth.
Dermatologists Excluded From PsA Recommendation Task Force
ROME - "No dermatologists wanted" was the apparent message sent by a task force that prepared a new set of recommendations for managing patients with psoriatic arthritis on behalf of the European League Against Rheumatism (EULAR).
By design, the panel that wrote the recommendations consisted entirely of rheumatologists, a fact proudly announced by French rheumatologist Laure Gossec as she gave the first public presentation of the recommendations during a session of the congress.
"Our goal was to develop easy-to-apply management recommendations for pharmacological, nontopical treatment from the rheumatologists point of view," according to Dr. Gossec, who is a rheumatologist at Cochin Hospital in Paris.
The parochial genesis of the new EULAR recommendations contrasts with another authoritative set of psoriatic arthritis (PsA) management recommendations published last year by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) (Ann. Rheum. Dis. 2009;68:1387-94), a document that involved participation by dermatologists.
The timing also raised questions on why the world needed a second guide to managing PsA so soon.
"We felt that the GRAPPA recommendations were rather complex to apply for rheumatologists. They require distinguishing mild, moderate, and severe disease and features of the disease," said Dr. Gossec.
In addition, creation of the GRAPPA recommendations, published in 2009, involved a high representation of dermatologists and were applicable to patients with high skin involvement, she said.
Drafting of the new EULAR recommendations began last January and finished in June, just days before Dr. Gossec presented them.
Some of this rationale is reasonable, said Dr. Christopher T. Ritchlin, a rheumatologist at the University of Rochester, N.Y., who chaired the GRAPPA panel that produced the 2009 treatment recommendations and was also a member of the EULAR task force that created the new guidelines.
"They're trying to be simple, which is fine. This is a great effort. I completely applaud trying to simplify, but that can be challenging because psoriatic arthritis is not only heterogeneous but can be complex within a patient. The GRAPPA recommendations are very complex; they took 2 years to develop. We worked with dermatologists because we felt there needs to be co-management for a large number of patients to effectively take care of their psoriatic disease. Interaction of the rheumatologist and dermatologist and even a psychologist or psychiatrist is critical for the successful management of a large number of cases. Not for every patient; some patients have no skin involvement. But there are a lot who have skin involvement."
Most dermatologists would likely agree.
"I think it is important for dermatologists to have a seat at the table," said Dr. Craig L. Leonardi, a dermatologist and psoriasis specialist at St. Louis University. "Results from published studies show that roughly 70% of PsA patients have skin symptoms before joint symptoms and, on average, they had skin symptoms for 10 years. If patients are pursuing treatment for their psoriasis, it may be a dermatologist who detects early PsA first. As a consequence, recommendations for early diagnosis and treatment need to consider the dermatologist."
Dr. Leonardi acknowledged that "rheumatologists are the ones formally trained in managing inflammatory joint disease. But there are some things about managing psoriasis that rheumatologists may not be aware of. For example, psoriatic skin has a wide range of responses to the various tumor necrosis factor antagonists" which should play a role in drug selection, he said.
A more skeptical view of the need for a new set of recommendations so soon on the heels of the GRAPPA paper came from Dr. Dafna D. Gladman, another member of the GRAPPA panel who had no involvement with the EULAR task force. "The EULAR group didn't come up with any more papers than GRAPPA. I'm not sure we have more information here than in the GRAPPA recommendations. I feel the GRAPPA recommendations are more helpful to clinicians because they tell them exactly what to do" for PsA patients with very specific disease presentations, he said.
"I'm not impressed that these recommendations are anything new," said Dr. Gladman, professor of medicine at the University of Toronto and director of the psoriatic arthritis program at Toronto Western Hospital.
During the brief discussion of the new EULAR recommendations following Dr. Gossec's presentation, most attention focused on the role of methotrexate and tumor necrosis factor (TNF) inhibitors for treating PsA.
The recommendations "emphasize how much we need controlled trial data on methotrexate," said Dr. Ritchlin.
One of the 10 recommendations from the EULAR panel gave the green light to starting treatment with a TNF inhibitor "in patients with active arthritis and an inadequate response to at least one synthetic disease-modifying antirheumatic drug, such as methotrexate."
"We thought that there was not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
"We thought that there were not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
The 10 recommendations are as follows:
1. NSAIDs may be used to relieve musculoskeletal signs and symptoms.
2. Treatment with a DMARD such as methotrexate, sulfasalazine, or leflunomide should be considered at an early stage of active disease, especially when it involves many swollen joints, structural damage in the presence of inflammation, high erythrocyte sedimentation rate or C-reactive protein, or clinically relevant extra-articular manifestations.
3. Consider use of methotrexate or another DMARD that also improves psoriasis in patients with active PsA that involves clinically relevant psoriasis.
4. Local injections of corticosteroids should be considered as adjunctive therapy. Systemic steroids at the lowest effective dose may be used with caution.
5. Therapy with a TNF inhibitor should be initiated in patients with active arthritis that responds inadequately to at least one synthetic DMARD, such as methotrexate.
6. Patients with active enthesitis, dactylitis, or both and insufficient response to NSAIDs or local steroid injections should be considered for anti-TNF therapy.
7. Consider anti-TNF therapy in cases of active, predominantly axial disease that has not responded sufficiently to NSAIDs.
8. In exceptional cases, a patient with very active disease who had not yet been treated with a synthetic DMARD may be considered for treatment with an anti TNF agent, especially when the patient has many swollen joints; structural damage in the presence of inflammation; or clinically relevant extra-articular manifestations, especially extensive skin involvement.
9. Consider switching to a different anti-TNF agent if the patient fails to respond adequately to the first drug from this class.
10. When adjusting treatment, take into account factors beyond disease activity, including comorbidities and safety issues.
Dr. Gossec, Dr. Ritchlin, and Dr. Gladman said that they had no relevant disclosures. Dr. Leonardi has been a consultant to, speaker for, and has received research support from Abbott Laboratories, Amgen Inc., and Genentech Inc. He has been a consultant to and received research support from Genentech. He has been a speaker for Warner Chilcott. He has received research support from Allergan Inc., Altana AG, Alza Corp., Astellis Pharma US Inc., Bristol-Myers Squibb, Celgene Corp., CombinatoRx, Eli Lilly & Co., Galderma Laboratories, Genzyme Corp., Incyte Corp., Israel Pharmaceutical, Novartis AG, Perrigo Co., Pfizer Inc., RTL Group, Schering-Plough Corp., 3M, Vitae Pharmaceuticals Inc., and Wyeth.
ROME - "No dermatologists wanted" was the apparent message sent by a task force that prepared a new set of recommendations for managing patients with psoriatic arthritis on behalf of the European League Against Rheumatism (EULAR).
By design, the panel that wrote the recommendations consisted entirely of rheumatologists, a fact proudly announced by French rheumatologist Laure Gossec as she gave the first public presentation of the recommendations during a session of the congress.
"Our goal was to develop easy-to-apply management recommendations for pharmacological, nontopical treatment from the rheumatologists point of view," according to Dr. Gossec, who is a rheumatologist at Cochin Hospital in Paris.
The parochial genesis of the new EULAR recommendations contrasts with another authoritative set of psoriatic arthritis (PsA) management recommendations published last year by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) (Ann. Rheum. Dis. 2009;68:1387-94), a document that involved participation by dermatologists.
The timing also raised questions on why the world needed a second guide to managing PsA so soon.
"We felt that the GRAPPA recommendations were rather complex to apply for rheumatologists. They require distinguishing mild, moderate, and severe disease and features of the disease," said Dr. Gossec.
In addition, creation of the GRAPPA recommendations, published in 2009, involved a high representation of dermatologists and were applicable to patients with high skin involvement, she said.
Drafting of the new EULAR recommendations began last January and finished in June, just days before Dr. Gossec presented them.
Some of this rationale is reasonable, said Dr. Christopher T. Ritchlin, a rheumatologist at the University of Rochester, N.Y., who chaired the GRAPPA panel that produced the 2009 treatment recommendations and was also a member of the EULAR task force that created the new guidelines.
"They're trying to be simple, which is fine. This is a great effort. I completely applaud trying to simplify, but that can be challenging because psoriatic arthritis is not only heterogeneous but can be complex within a patient. The GRAPPA recommendations are very complex; they took 2 years to develop. We worked with dermatologists because we felt there needs to be co-management for a large number of patients to effectively take care of their psoriatic disease. Interaction of the rheumatologist and dermatologist and even a psychologist or psychiatrist is critical for the successful management of a large number of cases. Not for every patient; some patients have no skin involvement. But there are a lot who have skin involvement."
Most dermatologists would likely agree.
"I think it is important for dermatologists to have a seat at the table," said Dr. Craig L. Leonardi, a dermatologist and psoriasis specialist at St. Louis University. "Results from published studies show that roughly 70% of PsA patients have skin symptoms before joint symptoms and, on average, they had skin symptoms for 10 years. If patients are pursuing treatment for their psoriasis, it may be a dermatologist who detects early PsA first. As a consequence, recommendations for early diagnosis and treatment need to consider the dermatologist."
Dr. Leonardi acknowledged that "rheumatologists are the ones formally trained in managing inflammatory joint disease. But there are some things about managing psoriasis that rheumatologists may not be aware of. For example, psoriatic skin has a wide range of responses to the various tumor necrosis factor antagonists" which should play a role in drug selection, he said.
A more skeptical view of the need for a new set of recommendations so soon on the heels of the GRAPPA paper came from Dr. Dafna D. Gladman, another member of the GRAPPA panel who had no involvement with the EULAR task force. "The EULAR group didn't come up with any more papers than GRAPPA. I'm not sure we have more information here than in the GRAPPA recommendations. I feel the GRAPPA recommendations are more helpful to clinicians because they tell them exactly what to do" for PsA patients with very specific disease presentations, he said.
"I'm not impressed that these recommendations are anything new," said Dr. Gladman, professor of medicine at the University of Toronto and director of the psoriatic arthritis program at Toronto Western Hospital.
During the brief discussion of the new EULAR recommendations following Dr. Gossec's presentation, most attention focused on the role of methotrexate and tumor necrosis factor (TNF) inhibitors for treating PsA.
The recommendations "emphasize how much we need controlled trial data on methotrexate," said Dr. Ritchlin.
One of the 10 recommendations from the EULAR panel gave the green light to starting treatment with a TNF inhibitor "in patients with active arthritis and an inadequate response to at least one synthetic disease-modifying antirheumatic drug, such as methotrexate."
"We thought that there was not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
"We thought that there were not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
The 10 recommendations are as follows:
1. NSAIDs may be used to relieve musculoskeletal signs and symptoms.
2. Treatment with a DMARD such as methotrexate, sulfasalazine, or leflunomide should be considered at an early stage of active disease, especially when it involves many swollen joints, structural damage in the presence of inflammation, high erythrocyte sedimentation rate or C-reactive protein, or clinically relevant extra-articular manifestations.
3. Consider use of methotrexate or another DMARD that also improves psoriasis in patients with active PsA that involves clinically relevant psoriasis.
4. Local injections of corticosteroids should be considered as adjunctive therapy. Systemic steroids at the lowest effective dose may be used with caution.
5. Therapy with a TNF inhibitor should be initiated in patients with active arthritis that responds inadequately to at least one synthetic DMARD, such as methotrexate.
6. Patients with active enthesitis, dactylitis, or both and insufficient response to NSAIDs or local steroid injections should be considered for anti-TNF therapy.
7. Consider anti-TNF therapy in cases of active, predominantly axial disease that has not responded sufficiently to NSAIDs.
8. In exceptional cases, a patient with very active disease who had not yet been treated with a synthetic DMARD may be considered for treatment with an anti TNF agent, especially when the patient has many swollen joints; structural damage in the presence of inflammation; or clinically relevant extra-articular manifestations, especially extensive skin involvement.
9. Consider switching to a different anti-TNF agent if the patient fails to respond adequately to the first drug from this class.
10. When adjusting treatment, take into account factors beyond disease activity, including comorbidities and safety issues.
Dr. Gossec, Dr. Ritchlin, and Dr. Gladman said that they had no relevant disclosures. Dr. Leonardi has been a consultant to, speaker for, and has received research support from Abbott Laboratories, Amgen Inc., and Genentech Inc. He has been a consultant to and received research support from Genentech. He has been a speaker for Warner Chilcott. He has received research support from Allergan Inc., Altana AG, Alza Corp., Astellis Pharma US Inc., Bristol-Myers Squibb, Celgene Corp., CombinatoRx, Eli Lilly & Co., Galderma Laboratories, Genzyme Corp., Incyte Corp., Israel Pharmaceutical, Novartis AG, Perrigo Co., Pfizer Inc., RTL Group, Schering-Plough Corp., 3M, Vitae Pharmaceuticals Inc., and Wyeth.
ROME - "No dermatologists wanted" was the apparent message sent by a task force that prepared a new set of recommendations for managing patients with psoriatic arthritis on behalf of the European League Against Rheumatism (EULAR).
By design, the panel that wrote the recommendations consisted entirely of rheumatologists, a fact proudly announced by French rheumatologist Laure Gossec as she gave the first public presentation of the recommendations during a session of the congress.
"Our goal was to develop easy-to-apply management recommendations for pharmacological, nontopical treatment from the rheumatologists point of view," according to Dr. Gossec, who is a rheumatologist at Cochin Hospital in Paris.
The parochial genesis of the new EULAR recommendations contrasts with another authoritative set of psoriatic arthritis (PsA) management recommendations published last year by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) (Ann. Rheum. Dis. 2009;68:1387-94), a document that involved participation by dermatologists.
The timing also raised questions on why the world needed a second guide to managing PsA so soon.
"We felt that the GRAPPA recommendations were rather complex to apply for rheumatologists. They require distinguishing mild, moderate, and severe disease and features of the disease," said Dr. Gossec.
In addition, creation of the GRAPPA recommendations, published in 2009, involved a high representation of dermatologists and were applicable to patients with high skin involvement, she said.
Drafting of the new EULAR recommendations began last January and finished in June, just days before Dr. Gossec presented them.
Some of this rationale is reasonable, said Dr. Christopher T. Ritchlin, a rheumatologist at the University of Rochester, N.Y., who chaired the GRAPPA panel that produced the 2009 treatment recommendations and was also a member of the EULAR task force that created the new guidelines.
"They're trying to be simple, which is fine. This is a great effort. I completely applaud trying to simplify, but that can be challenging because psoriatic arthritis is not only heterogeneous but can be complex within a patient. The GRAPPA recommendations are very complex; they took 2 years to develop. We worked with dermatologists because we felt there needs to be co-management for a large number of patients to effectively take care of their psoriatic disease. Interaction of the rheumatologist and dermatologist and even a psychologist or psychiatrist is critical for the successful management of a large number of cases. Not for every patient; some patients have no skin involvement. But there are a lot who have skin involvement."
Most dermatologists would likely agree.
"I think it is important for dermatologists to have a seat at the table," said Dr. Craig L. Leonardi, a dermatologist and psoriasis specialist at St. Louis University. "Results from published studies show that roughly 70% of PsA patients have skin symptoms before joint symptoms and, on average, they had skin symptoms for 10 years. If patients are pursuing treatment for their psoriasis, it may be a dermatologist who detects early PsA first. As a consequence, recommendations for early diagnosis and treatment need to consider the dermatologist."
Dr. Leonardi acknowledged that "rheumatologists are the ones formally trained in managing inflammatory joint disease. But there are some things about managing psoriasis that rheumatologists may not be aware of. For example, psoriatic skin has a wide range of responses to the various tumor necrosis factor antagonists" which should play a role in drug selection, he said.
A more skeptical view of the need for a new set of recommendations so soon on the heels of the GRAPPA paper came from Dr. Dafna D. Gladman, another member of the GRAPPA panel who had no involvement with the EULAR task force. "The EULAR group didn't come up with any more papers than GRAPPA. I'm not sure we have more information here than in the GRAPPA recommendations. I feel the GRAPPA recommendations are more helpful to clinicians because they tell them exactly what to do" for PsA patients with very specific disease presentations, he said.
"I'm not impressed that these recommendations are anything new," said Dr. Gladman, professor of medicine at the University of Toronto and director of the psoriatic arthritis program at Toronto Western Hospital.
During the brief discussion of the new EULAR recommendations following Dr. Gossec's presentation, most attention focused on the role of methotrexate and tumor necrosis factor (TNF) inhibitors for treating PsA.
The recommendations "emphasize how much we need controlled trial data on methotrexate," said Dr. Ritchlin.
One of the 10 recommendations from the EULAR panel gave the green light to starting treatment with a TNF inhibitor "in patients with active arthritis and an inadequate response to at least one synthetic disease-modifying antirheumatic drug, such as methotrexate."
"We thought that there was not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
"We thought that there were not enough data to limit [TNF inhibitor treatment] to patients with a specific number of affected joints," Dr. Gossec said.
The 10 recommendations are as follows:
1. NSAIDs may be used to relieve musculoskeletal signs and symptoms.
2. Treatment with a DMARD such as methotrexate, sulfasalazine, or leflunomide should be considered at an early stage of active disease, especially when it involves many swollen joints, structural damage in the presence of inflammation, high erythrocyte sedimentation rate or C-reactive protein, or clinically relevant extra-articular manifestations.
3. Consider use of methotrexate or another DMARD that also improves psoriasis in patients with active PsA that involves clinically relevant psoriasis.
4. Local injections of corticosteroids should be considered as adjunctive therapy. Systemic steroids at the lowest effective dose may be used with caution.
5. Therapy with a TNF inhibitor should be initiated in patients with active arthritis that responds inadequately to at least one synthetic DMARD, such as methotrexate.
6. Patients with active enthesitis, dactylitis, or both and insufficient response to NSAIDs or local steroid injections should be considered for anti-TNF therapy.
7. Consider anti-TNF therapy in cases of active, predominantly axial disease that has not responded sufficiently to NSAIDs.
8. In exceptional cases, a patient with very active disease who had not yet been treated with a synthetic DMARD may be considered for treatment with an anti TNF agent, especially when the patient has many swollen joints; structural damage in the presence of inflammation; or clinically relevant extra-articular manifestations, especially extensive skin involvement.
9. Consider switching to a different anti-TNF agent if the patient fails to respond adequately to the first drug from this class.
10. When adjusting treatment, take into account factors beyond disease activity, including comorbidities and safety issues.
Dr. Gossec, Dr. Ritchlin, and Dr. Gladman said that they had no relevant disclosures. Dr. Leonardi has been a consultant to, speaker for, and has received research support from Abbott Laboratories, Amgen Inc., and Genentech Inc. He has been a consultant to and received research support from Genentech. He has been a speaker for Warner Chilcott. He has received research support from Allergan Inc., Altana AG, Alza Corp., Astellis Pharma US Inc., Bristol-Myers Squibb, Celgene Corp., CombinatoRx, Eli Lilly & Co., Galderma Laboratories, Genzyme Corp., Incyte Corp., Israel Pharmaceutical, Novartis AG, Perrigo Co., Pfizer Inc., RTL Group, Schering-Plough Corp., 3M, Vitae Pharmaceuticals Inc., and Wyeth.
Black SLE Patients Respond Well to Daily 2,000 IU Doses of Vitamin D
VANCOUVER, B.C. - A daily dose of at least 2,000 IU of vitamin D is required to elevate serum 25-hydroxyvitamin D levels above 30 ng/mL, the minimum threshold for optimal immune health, according to Dr. Diane Kamen, a rheumatologist at the Medical University of South Carolina in Charleston.
The conclusion is based on an open-label, phase I study of vitamin D repletion in 18 black patients with lupus that was presented by Dr. Kamen at the International Congress on Systemic Lupus Erythematosus.
Starting from a baseline mean serum 25-hydroxyvitamin D (25[OH]D) level of 13.3 ng/mL, six patients received 800 IU vitamin D once daily; six received 2,000 IU once daily; and six received 4,000 IU once daily.
After 12 weeks, 67% (four patients) in the 800 IU group, 83% (five) in the 2,000 IU group, and 67% (four) in the 4,000 IU group repleted to 30 ng/mL or greater.
In the 4,000 IU group, levels in 33% (two patients) rose above 40 ng/mL. That level was not reached at the lower doses.
The results are important, Dr. Kamen said in an interview after the conference, because although there is growing awareness that such high doses of vitamin D are needed to restore 25(OH)D levels in patients with autoimmune disease, the rheumatology literature still contains recommendations for doses of 600-800 IU/day.
“That’s just not going to cut it; 2,000 IU a day is the minimum effective dose for repletion,” especially if patients avoid the sun to prevent lupus flares, Dr. Kamen said.
Rheumatologists “need to know to recommend those higher doses, and to monitor levels” of 25(OH)D to make sure they are maintained, she said.
The 18 patients were enrolled from a population of blacks living on the Sea Islands of South Carolina and Georgia, a population known as the Gullah in whom there is a high incidence of lupus.
An earlier Gullah study found that 43% of 187 subjects had 25(OH)D levels below 10 ng/mL; in some, levels were undetectable. Lower levels correlated with higher SLEDAI (Systemic Lupus Erythematosus Disease Activity Index) scores and higher anti–double stranded DNA antibody levels, Dr. Kamen said.
The mean age in the phase I study was 44 years; mean prednisone dose 4.3 mg/day; and mean SLEDAI score 2.4. In all, 17 of 18 of the subjects were women, 50% (9) took hydroxychloroquine, and 50% (9) were anti-dsDNA antibody positive.
Compliance with the treatment regimen was 99%, by pill count. The doses were very well tolerated and safe, Dr. Kamen said.
Although 2,000 IU per day elevated 26(OH)D levels in most patients to at least 30 ng/mL, there’s debate in the rheumatology community about whether target blood levels should be higher in lupus patients.
“We know that 30 ng/mL is the minimum accepted as normal,” Dr. Kamen said, noting that secondary hyperparathyroidism can begin below that level.
“We also know [healthy] sun-exposed people tend to live closer to 60 ng/mL. The debate is over if the target should be 30, 40, 50, or 60,” she said.
“I tell my patients at high risk for conditions influenced by vitamin D, such as osteoporosis and inflammatory conditions, that we want them to stay between 40 and 60 ng/mL,” she said, but “it’s a gray zone” that awaits further research.
Levels of 25(OH)D are known to be low in lupus patients, but no one can say for sure whether that is a cause or a consequence of the disease, or if it results from the medications used to treat it, such as prednisone and hydroxychloroquine.
Vitamin D appears to help maintain protective immunologic responses and to enhance immunologic self-tolerance, so it’s possible a deficiency plays a role in the immune dysregulation that is seen in lupus, Dr. Kamen said.
A phase II trial is underway to assess the ability of daily vitamin D supplementation at 2,000 or 4,000 IU to reduce interferon-alpha expression in lupus patients.
The study has enrolled 44 of the 57 patients needed, and Dr. Kamen expects it to be completed and the results to be published in late 2011.
Dr. Kamen said she had no disclosures. The study was funded by the National Institutes of Health.
VANCOUVER, B.C. - A daily dose of at least 2,000 IU of vitamin D is required to elevate serum 25-hydroxyvitamin D levels above 30 ng/mL, the minimum threshold for optimal immune health, according to Dr. Diane Kamen, a rheumatologist at the Medical University of South Carolina in Charleston.
The conclusion is based on an open-label, phase I study of vitamin D repletion in 18 black patients with lupus that was presented by Dr. Kamen at the International Congress on Systemic Lupus Erythematosus.
Starting from a baseline mean serum 25-hydroxyvitamin D (25[OH]D) level of 13.3 ng/mL, six patients received 800 IU vitamin D once daily; six received 2,000 IU once daily; and six received 4,000 IU once daily.
After 12 weeks, 67% (four patients) in the 800 IU group, 83% (five) in the 2,000 IU group, and 67% (four) in the 4,000 IU group repleted to 30 ng/mL or greater.
In the 4,000 IU group, levels in 33% (two patients) rose above 40 ng/mL. That level was not reached at the lower doses.
The results are important, Dr. Kamen said in an interview after the conference, because although there is growing awareness that such high doses of vitamin D are needed to restore 25(OH)D levels in patients with autoimmune disease, the rheumatology literature still contains recommendations for doses of 600-800 IU/day.
“That’s just not going to cut it; 2,000 IU a day is the minimum effective dose for repletion,” especially if patients avoid the sun to prevent lupus flares, Dr. Kamen said.
Rheumatologists “need to know to recommend those higher doses, and to monitor levels” of 25(OH)D to make sure they are maintained, she said.
The 18 patients were enrolled from a population of blacks living on the Sea Islands of South Carolina and Georgia, a population known as the Gullah in whom there is a high incidence of lupus.
An earlier Gullah study found that 43% of 187 subjects had 25(OH)D levels below 10 ng/mL; in some, levels were undetectable. Lower levels correlated with higher SLEDAI (Systemic Lupus Erythematosus Disease Activity Index) scores and higher anti–double stranded DNA antibody levels, Dr. Kamen said.
The mean age in the phase I study was 44 years; mean prednisone dose 4.3 mg/day; and mean SLEDAI score 2.4. In all, 17 of 18 of the subjects were women, 50% (9) took hydroxychloroquine, and 50% (9) were anti-dsDNA antibody positive.
Compliance with the treatment regimen was 99%, by pill count. The doses were very well tolerated and safe, Dr. Kamen said.
Although 2,000 IU per day elevated 26(OH)D levels in most patients to at least 30 ng/mL, there’s debate in the rheumatology community about whether target blood levels should be higher in lupus patients.
“We know that 30 ng/mL is the minimum accepted as normal,” Dr. Kamen said, noting that secondary hyperparathyroidism can begin below that level.
“We also know [healthy] sun-exposed people tend to live closer to 60 ng/mL. The debate is over if the target should be 30, 40, 50, or 60,” she said.
“I tell my patients at high risk for conditions influenced by vitamin D, such as osteoporosis and inflammatory conditions, that we want them to stay between 40 and 60 ng/mL,” she said, but “it’s a gray zone” that awaits further research.
Levels of 25(OH)D are known to be low in lupus patients, but no one can say for sure whether that is a cause or a consequence of the disease, or if it results from the medications used to treat it, such as prednisone and hydroxychloroquine.
Vitamin D appears to help maintain protective immunologic responses and to enhance immunologic self-tolerance, so it’s possible a deficiency plays a role in the immune dysregulation that is seen in lupus, Dr. Kamen said.
A phase II trial is underway to assess the ability of daily vitamin D supplementation at 2,000 or 4,000 IU to reduce interferon-alpha expression in lupus patients.
The study has enrolled 44 of the 57 patients needed, and Dr. Kamen expects it to be completed and the results to be published in late 2011.
Dr. Kamen said she had no disclosures. The study was funded by the National Institutes of Health.
VANCOUVER, B.C. - A daily dose of at least 2,000 IU of vitamin D is required to elevate serum 25-hydroxyvitamin D levels above 30 ng/mL, the minimum threshold for optimal immune health, according to Dr. Diane Kamen, a rheumatologist at the Medical University of South Carolina in Charleston.
The conclusion is based on an open-label, phase I study of vitamin D repletion in 18 black patients with lupus that was presented by Dr. Kamen at the International Congress on Systemic Lupus Erythematosus.
Starting from a baseline mean serum 25-hydroxyvitamin D (25[OH]D) level of 13.3 ng/mL, six patients received 800 IU vitamin D once daily; six received 2,000 IU once daily; and six received 4,000 IU once daily.
After 12 weeks, 67% (four patients) in the 800 IU group, 83% (five) in the 2,000 IU group, and 67% (four) in the 4,000 IU group repleted to 30 ng/mL or greater.
In the 4,000 IU group, levels in 33% (two patients) rose above 40 ng/mL. That level was not reached at the lower doses.
The results are important, Dr. Kamen said in an interview after the conference, because although there is growing awareness that such high doses of vitamin D are needed to restore 25(OH)D levels in patients with autoimmune disease, the rheumatology literature still contains recommendations for doses of 600-800 IU/day.
“That’s just not going to cut it; 2,000 IU a day is the minimum effective dose for repletion,” especially if patients avoid the sun to prevent lupus flares, Dr. Kamen said.
Rheumatologists “need to know to recommend those higher doses, and to monitor levels” of 25(OH)D to make sure they are maintained, she said.
The 18 patients were enrolled from a population of blacks living on the Sea Islands of South Carolina and Georgia, a population known as the Gullah in whom there is a high incidence of lupus.
An earlier Gullah study found that 43% of 187 subjects had 25(OH)D levels below 10 ng/mL; in some, levels were undetectable. Lower levels correlated with higher SLEDAI (Systemic Lupus Erythematosus Disease Activity Index) scores and higher anti–double stranded DNA antibody levels, Dr. Kamen said.
The mean age in the phase I study was 44 years; mean prednisone dose 4.3 mg/day; and mean SLEDAI score 2.4. In all, 17 of 18 of the subjects were women, 50% (9) took hydroxychloroquine, and 50% (9) were anti-dsDNA antibody positive.
Compliance with the treatment regimen was 99%, by pill count. The doses were very well tolerated and safe, Dr. Kamen said.
Although 2,000 IU per day elevated 26(OH)D levels in most patients to at least 30 ng/mL, there’s debate in the rheumatology community about whether target blood levels should be higher in lupus patients.
“We know that 30 ng/mL is the minimum accepted as normal,” Dr. Kamen said, noting that secondary hyperparathyroidism can begin below that level.
“We also know [healthy] sun-exposed people tend to live closer to 60 ng/mL. The debate is over if the target should be 30, 40, 50, or 60,” she said.
“I tell my patients at high risk for conditions influenced by vitamin D, such as osteoporosis and inflammatory conditions, that we want them to stay between 40 and 60 ng/mL,” she said, but “it’s a gray zone” that awaits further research.
Levels of 25(OH)D are known to be low in lupus patients, but no one can say for sure whether that is a cause or a consequence of the disease, or if it results from the medications used to treat it, such as prednisone and hydroxychloroquine.
Vitamin D appears to help maintain protective immunologic responses and to enhance immunologic self-tolerance, so it’s possible a deficiency plays a role in the immune dysregulation that is seen in lupus, Dr. Kamen said.
A phase II trial is underway to assess the ability of daily vitamin D supplementation at 2,000 or 4,000 IU to reduce interferon-alpha expression in lupus patients.
The study has enrolled 44 of the 57 patients needed, and Dr. Kamen expects it to be completed and the results to be published in late 2011.
Dr. Kamen said she had no disclosures. The study was funded by the National Institutes of Health.