User login
Focus on the Psychosocial Impacts of Psoriasis in Women
SAN FRANCISCO – It falls into a dermatologist's realm of responsibility to help women with psoriasis overcome treatment barriers and improve their psychosocial well-being, according to Dr. Jennifer C. Cather.
Women often are the main caregivers for their families and thus may put their family's health care needs before their own, Dr. Cather noted at a seminar on women's and pediatric dermatology sponsored by Skin Disease Education Foundation. Dermatologists should stress to these patients the importance of making their medical needs a priority.
"I see many women who have gone untreated or undertreated for many years despite being under the care of a dermatologist, or even several other dermatologists, before arriving in our office. It is important for women to make their own health a priority and then to seek out a dermatologist with whom they can frankly discuss the range of therapeutic options and their correlating implications," Dr. Cather said in an interview.
"The reality is that, however complicated it is to treat psoriasis, the complications of not treating the disease can often be much worse," said Dr. Cather, medical director at Modern Dermatology and Modern Research Associates, Dallas.
Although the prevalence of psoriasis is approximately the same by gender, the condition has a greater psychosocial impact on women than on men, multiple surveys have indicated. For example, 20% of women vs. 12% of men reported their psoriasis was a "very large problem in their everyday lives," the National Psoriasis Foundation discovered when they asked 4,725 people from 2004 to 2009 ("Report on the Psycho-Social Impacts of Psoriasis").
In addition, women reported a higher symptom burden than did men. Women were 17% more likely to report itching; 16% more likely to report physical irritation; and 24% more likely to report pain associated with their psoriasis, compared with men, according to the online report.
Other researchers discovered a statistically significant reduction in quality of life for women with psoriasis versus men, based on a survey of 266 psoriasis patients (J. Am. Acad. Dermatol. 2004:51:704-8).
"Wearing a chronic disease on the outside of your body is a real struggle in a society as focused on appearance as ours is," Dr. Cather said. The average age of psoriasis onset is between 15 and 35 years, a time when many women are making important choices about their future. The impact of low self-esteem associated with this condition can have far reaching implications.
Therefore, it is important for dermatologists to take the time to assess the patient's well-being. In addition to measurement of affected body surface area, inflammatory burden, joint involvement, and other medical issues using validated instruments, ask women some simple questions, Dr. Cather said. Examples include: "How is your psoriasis today?" and "Is the cost of your therapy worth it?"
"We need to understand how each patient is experiencing the disease and whether he or she is satisfied with the treatment option we have selected," Dr. Cather said. "Satisfied patients are compliant with the treatment regimen and have a greater chance of success over the long term."
The good news is that systemic medication can make a big difference in the lives of women affected by psoriasis. Tumor necrosis factor (TNF) antagonists are the treatment of choice for Dr. Cather when patients present with psoriasis and psoriatic arthritis. These agents can have a synergistic benefit when given with methotrexate, she said.
Interestingly, women with higher body mass index tend to respond better to monoclonal antibody treatments, Dr. Cather said. Also, keep in mind that psoriasis treatment needs may change for a particular woman throughout her life.
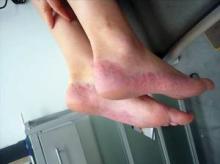
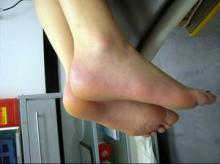
Dr. Cather presented several cases of women who responded well to systemic therapy. These included a young teenage girl whose plaques cleared following treatment with etanercept; and an older teenage girl – treated previously with adalimumab, efalizumab, and cyclosporine A – who responded to two injections of ustekinumab 45 mg. She also presented the case of a 55-year-old woman previously prescribed methotrexate and etanercept. The patient’s subsequent treatment with adalimumab has been "life changing"; her psoriasis has been clear since 2003.
Begin with a thorough physical examination, including a total body skin evaluation, and medical history, Dr. Cather said. Physical exam also can include a Pap smear, mammogram, and colonoscopy. Ask women about their history of malignancy, infections, and vaccinations. Inquire about their social history as well, she recommended.
A complete blood count, comprehensive metabolic panel, and high-sensitivity C-reaction protein assay are recommended laboratory tests. A screen for hepatitis B and C, HIV infection, and tuberculosis (repeated annually) can be helpful in the differential diagnosis.
In terms of future research, Dr. Cather said:"In our clinic, we are currently exploring the incorporation of more formal validated quality-of-life measures to dig deeper into how psoriasis is impacting life decisions and productivity."
Disclosures: Dr. Cather disclosed that she is a consultant for and on the speaker’s bureau of Abbott Laboratories and Centocor Inc. She also is a researcher for Amgen, Celgene Corp., and Pfizer.
SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – It falls into a dermatologist's realm of responsibility to help women with psoriasis overcome treatment barriers and improve their psychosocial well-being, according to Dr. Jennifer C. Cather.
Women often are the main caregivers for their families and thus may put their family's health care needs before their own, Dr. Cather noted at a seminar on women's and pediatric dermatology sponsored by Skin Disease Education Foundation. Dermatologists should stress to these patients the importance of making their medical needs a priority.
"I see many women who have gone untreated or undertreated for many years despite being under the care of a dermatologist, or even several other dermatologists, before arriving in our office. It is important for women to make their own health a priority and then to seek out a dermatologist with whom they can frankly discuss the range of therapeutic options and their correlating implications," Dr. Cather said in an interview.
"The reality is that, however complicated it is to treat psoriasis, the complications of not treating the disease can often be much worse," said Dr. Cather, medical director at Modern Dermatology and Modern Research Associates, Dallas.
Although the prevalence of psoriasis is approximately the same by gender, the condition has a greater psychosocial impact on women than on men, multiple surveys have indicated. For example, 20% of women vs. 12% of men reported their psoriasis was a "very large problem in their everyday lives," the National Psoriasis Foundation discovered when they asked 4,725 people from 2004 to 2009 ("Report on the Psycho-Social Impacts of Psoriasis").
In addition, women reported a higher symptom burden than did men. Women were 17% more likely to report itching; 16% more likely to report physical irritation; and 24% more likely to report pain associated with their psoriasis, compared with men, according to the online report.
Other researchers discovered a statistically significant reduction in quality of life for women with psoriasis versus men, based on a survey of 266 psoriasis patients (J. Am. Acad. Dermatol. 2004:51:704-8).
"Wearing a chronic disease on the outside of your body is a real struggle in a society as focused on appearance as ours is," Dr. Cather said. The average age of psoriasis onset is between 15 and 35 years, a time when many women are making important choices about their future. The impact of low self-esteem associated with this condition can have far reaching implications.
Therefore, it is important for dermatologists to take the time to assess the patient's well-being. In addition to measurement of affected body surface area, inflammatory burden, joint involvement, and other medical issues using validated instruments, ask women some simple questions, Dr. Cather said. Examples include: "How is your psoriasis today?" and "Is the cost of your therapy worth it?"
"We need to understand how each patient is experiencing the disease and whether he or she is satisfied with the treatment option we have selected," Dr. Cather said. "Satisfied patients are compliant with the treatment regimen and have a greater chance of success over the long term."
The good news is that systemic medication can make a big difference in the lives of women affected by psoriasis. Tumor necrosis factor (TNF) antagonists are the treatment of choice for Dr. Cather when patients present with psoriasis and psoriatic arthritis. These agents can have a synergistic benefit when given with methotrexate, she said.
Interestingly, women with higher body mass index tend to respond better to monoclonal antibody treatments, Dr. Cather said. Also, keep in mind that psoriasis treatment needs may change for a particular woman throughout her life.
Dr. Cather presented several cases of women who responded well to systemic therapy. These included a young teenage girl whose plaques cleared following treatment with etanercept; and an older teenage girl – treated previously with adalimumab, efalizumab, and cyclosporine A – who responded to two injections of ustekinumab 45 mg. She also presented the case of a 55-year-old woman previously prescribed methotrexate and etanercept. The patient’s subsequent treatment with adalimumab has been "life changing"; her psoriasis has been clear since 2003.
Begin with a thorough physical examination, including a total body skin evaluation, and medical history, Dr. Cather said. Physical exam also can include a Pap smear, mammogram, and colonoscopy. Ask women about their history of malignancy, infections, and vaccinations. Inquire about their social history as well, she recommended.
A complete blood count, comprehensive metabolic panel, and high-sensitivity C-reaction protein assay are recommended laboratory tests. A screen for hepatitis B and C, HIV infection, and tuberculosis (repeated annually) can be helpful in the differential diagnosis.
In terms of future research, Dr. Cather said:"In our clinic, we are currently exploring the incorporation of more formal validated quality-of-life measures to dig deeper into how psoriasis is impacting life decisions and productivity."
Disclosures: Dr. Cather disclosed that she is a consultant for and on the speaker’s bureau of Abbott Laboratories and Centocor Inc. She also is a researcher for Amgen, Celgene Corp., and Pfizer.
SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – It falls into a dermatologist's realm of responsibility to help women with psoriasis overcome treatment barriers and improve their psychosocial well-being, according to Dr. Jennifer C. Cather.
Women often are the main caregivers for their families and thus may put their family's health care needs before their own, Dr. Cather noted at a seminar on women's and pediatric dermatology sponsored by Skin Disease Education Foundation. Dermatologists should stress to these patients the importance of making their medical needs a priority.
"I see many women who have gone untreated or undertreated for many years despite being under the care of a dermatologist, or even several other dermatologists, before arriving in our office. It is important for women to make their own health a priority and then to seek out a dermatologist with whom they can frankly discuss the range of therapeutic options and their correlating implications," Dr. Cather said in an interview.
"The reality is that, however complicated it is to treat psoriasis, the complications of not treating the disease can often be much worse," said Dr. Cather, medical director at Modern Dermatology and Modern Research Associates, Dallas.
Although the prevalence of psoriasis is approximately the same by gender, the condition has a greater psychosocial impact on women than on men, multiple surveys have indicated. For example, 20% of women vs. 12% of men reported their psoriasis was a "very large problem in their everyday lives," the National Psoriasis Foundation discovered when they asked 4,725 people from 2004 to 2009 ("Report on the Psycho-Social Impacts of Psoriasis").
In addition, women reported a higher symptom burden than did men. Women were 17% more likely to report itching; 16% more likely to report physical irritation; and 24% more likely to report pain associated with their psoriasis, compared with men, according to the online report.
Other researchers discovered a statistically significant reduction in quality of life for women with psoriasis versus men, based on a survey of 266 psoriasis patients (J. Am. Acad. Dermatol. 2004:51:704-8).
"Wearing a chronic disease on the outside of your body is a real struggle in a society as focused on appearance as ours is," Dr. Cather said. The average age of psoriasis onset is between 15 and 35 years, a time when many women are making important choices about their future. The impact of low self-esteem associated with this condition can have far reaching implications.
Therefore, it is important for dermatologists to take the time to assess the patient's well-being. In addition to measurement of affected body surface area, inflammatory burden, joint involvement, and other medical issues using validated instruments, ask women some simple questions, Dr. Cather said. Examples include: "How is your psoriasis today?" and "Is the cost of your therapy worth it?"
"We need to understand how each patient is experiencing the disease and whether he or she is satisfied with the treatment option we have selected," Dr. Cather said. "Satisfied patients are compliant with the treatment regimen and have a greater chance of success over the long term."
The good news is that systemic medication can make a big difference in the lives of women affected by psoriasis. Tumor necrosis factor (TNF) antagonists are the treatment of choice for Dr. Cather when patients present with psoriasis and psoriatic arthritis. These agents can have a synergistic benefit when given with methotrexate, she said.
Interestingly, women with higher body mass index tend to respond better to monoclonal antibody treatments, Dr. Cather said. Also, keep in mind that psoriasis treatment needs may change for a particular woman throughout her life.
Dr. Cather presented several cases of women who responded well to systemic therapy. These included a young teenage girl whose plaques cleared following treatment with etanercept; and an older teenage girl – treated previously with adalimumab, efalizumab, and cyclosporine A – who responded to two injections of ustekinumab 45 mg. She also presented the case of a 55-year-old woman previously prescribed methotrexate and etanercept. The patient’s subsequent treatment with adalimumab has been "life changing"; her psoriasis has been clear since 2003.
Begin with a thorough physical examination, including a total body skin evaluation, and medical history, Dr. Cather said. Physical exam also can include a Pap smear, mammogram, and colonoscopy. Ask women about their history of malignancy, infections, and vaccinations. Inquire about their social history as well, she recommended.
A complete blood count, comprehensive metabolic panel, and high-sensitivity C-reaction protein assay are recommended laboratory tests. A screen for hepatitis B and C, HIV infection, and tuberculosis (repeated annually) can be helpful in the differential diagnosis.
In terms of future research, Dr. Cather said:"In our clinic, we are currently exploring the incorporation of more formal validated quality-of-life measures to dig deeper into how psoriasis is impacting life decisions and productivity."
Disclosures: Dr. Cather disclosed that she is a consultant for and on the speaker’s bureau of Abbott Laboratories and Centocor Inc. She also is a researcher for Amgen, Celgene Corp., and Pfizer.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM A SEMINAR ON WOMEN'S AND PEDIATRIC DERMATOLOGY
TNF Antagonists in Rheumatic Patients Linked to Increased Hospitalization for Varicella Zoster
Rheumatic disease patients who are exposed to tumor necrosis factor antagonists have a 10-fold increased risk of hospitalization for varicella zoster virus infections, compared with the general population, according to a secondary analysis of two large databases.
Nonetheless, the absolute incidence of varicella-related hospitalizations remains low at about three cases per 10,000 person-years of exposure, and the risks of using vaccination for prevention likely outweigh the benefits, Dr. Ignacio Garcia-Doval of Complexo Hospitalario de Pontevedra (Spain) and colleagues have reported in the October issue of the Annals of the Rheumatic Diseases.
The estimated incidence rate of hospitalization for shingles in the rheumatic population was 32 cases per 100,000 patient years, compared with an expected rate of 3.4 in the general population, and the estimated incidence of hospitalization for chickenpox in the rheumatic patients was 26 per 100,000, compared with 1.9 in the general population. This finding is based on analysis of data from a national registry of rheumatic disease patients who were treated with TNF agents (BIOBADASER database) and from an administrative database of all hospital admissions in public centers in Spain (Conjunto Mínimo Básico de Datos al Alta Hospitalaria, or CMBD), which together represent more than 114 million patient-years.
The estimated age- and sex-standardized incidence rate per 100,000 person-years, and the estimated standardized incidence difference were 9 and 26, respectively, for shingles, and 19 and 33, respectively, for chickenpox, they said (Ann. Rheum. Dis. 2010;69:1751-5).
TNF antagonists are known to be associated with an increased risk of tuberculosis in particular and of opportunistic infections in general. There is a biological basis for an increased risk of viral infections, the investigators said, noting that although some studies have shown an increased rate of viral infection in TNF antagonist–treated patients, the clinical relevance of the increase is uncertain.
The current study does not allow differentiation of the causes for the increased risk, but it does show that the absolute rate is low.
The investigators said it is unlikely that the cohorts received systematic vaccination against varicella zoster virus, since the general health mandate in Spain was given in 2005 and only for children aged 11-14 years.
"Standard guidelines for chickenpox vaccination probably apply to the population included in our study," they wrote.
However, shingles vaccine, which is an attenuated vaccine with a higher dose of antigen, could potentially lead to more side effects in an immunosuppressed population, they said.
For example, in a randomized trial of adults older than age 60 years, shingles vaccine was associated with 7 cases of severe adverse events and 14 cases of vaccine-related adverse events per 10,000 vaccinations, they noted.
"These vaccination-associated risks are similar in rate and severity to the risks of hospitalized infections in our study. Hence, shingles vaccination before starting a TNF antagonist may not be warranted at present," they wrote, concluding that although vaccination in healthy children is warranted, it is not warranted in adults with "immunosuppression secondary to the baseline inflammatory disease and its complications."
Disclosures: Various study authors reported serving on the advisory board for, and/or receiving lecture fees or honoraria from, Wyeth, Abbott, Schering-Plough, Roche, and/or Bristol-Myers Squibb.
The rationale for zoster vaccination goes beyond the goal of simply preventing hospitalized herpes zoster. Vaccination in rheumatoid arthritis patients who are 60 years of age or older should be the standard of care before initiation of anti-TNF or other long-term immunosuppressive therapy.
Prospective data on the efficacy of herpes zoster vaccination, particularly in patients with rheumatoid arthritis, are lacking. But there is strong evidence for the protective effects and importance of vaccination in adults aged 60 years and older. Given that patients with rheumatoid arthritis are at increased risk for herpes zoster and that vaccination with live viruses is contraindicated while biological therapies are used, it would make sense to target this group for vaccination before anti-TNF therapy is initiated.
The purpose of vaccination is not only to lower the risk of rare, serious manifestations of herpes zoster, but also to lower the risk of uncomplicated herpes zoster, which causes considerable morbidity. Future studies should look at the potential benefits of vaccinating those younger than age 60 and those receiving other types of immunosuppressive therapy.
The opinions above are excerpted from an editorial accompanying the research report (Ann. Rheum. Dis. 2010;69:1735-7) by Dr. Kevin L. Winthrop, of the department of infectious diseases at the Oregon Health and Science University, Portland, and Dr. Daniel E. Furst, a Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles. Dr. Winthrop reported receiving funding from the Agency for Healthcare Research and Quality for work on the manuscript, and receiving a grant from UCB Pharmaceuticals, as well as consulting fees from Amgen, Wyeth, and Genentech. Dr. Furst reported receiving research support for studies of abatacept, adalimumab, certolizumab, etanercept, infliximab, rituximab, and tocilizumab, and consulting with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Genentech, and UCB.
The rationale for zoster vaccination goes beyond the goal of simply preventing hospitalized herpes zoster. Vaccination in rheumatoid arthritis patients who are 60 years of age or older should be the standard of care before initiation of anti-TNF or other long-term immunosuppressive therapy.
Prospective data on the efficacy of herpes zoster vaccination, particularly in patients with rheumatoid arthritis, are lacking. But there is strong evidence for the protective effects and importance of vaccination in adults aged 60 years and older. Given that patients with rheumatoid arthritis are at increased risk for herpes zoster and that vaccination with live viruses is contraindicated while biological therapies are used, it would make sense to target this group for vaccination before anti-TNF therapy is initiated.
The purpose of vaccination is not only to lower the risk of rare, serious manifestations of herpes zoster, but also to lower the risk of uncomplicated herpes zoster, which causes considerable morbidity. Future studies should look at the potential benefits of vaccinating those younger than age 60 and those receiving other types of immunosuppressive therapy.
The opinions above are excerpted from an editorial accompanying the research report (Ann. Rheum. Dis. 2010;69:1735-7) by Dr. Kevin L. Winthrop, of the department of infectious diseases at the Oregon Health and Science University, Portland, and Dr. Daniel E. Furst, a Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles. Dr. Winthrop reported receiving funding from the Agency for Healthcare Research and Quality for work on the manuscript, and receiving a grant from UCB Pharmaceuticals, as well as consulting fees from Amgen, Wyeth, and Genentech. Dr. Furst reported receiving research support for studies of abatacept, adalimumab, certolizumab, etanercept, infliximab, rituximab, and tocilizumab, and consulting with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Genentech, and UCB.
The rationale for zoster vaccination goes beyond the goal of simply preventing hospitalized herpes zoster. Vaccination in rheumatoid arthritis patients who are 60 years of age or older should be the standard of care before initiation of anti-TNF or other long-term immunosuppressive therapy.
Prospective data on the efficacy of herpes zoster vaccination, particularly in patients with rheumatoid arthritis, are lacking. But there is strong evidence for the protective effects and importance of vaccination in adults aged 60 years and older. Given that patients with rheumatoid arthritis are at increased risk for herpes zoster and that vaccination with live viruses is contraindicated while biological therapies are used, it would make sense to target this group for vaccination before anti-TNF therapy is initiated.
The purpose of vaccination is not only to lower the risk of rare, serious manifestations of herpes zoster, but also to lower the risk of uncomplicated herpes zoster, which causes considerable morbidity. Future studies should look at the potential benefits of vaccinating those younger than age 60 and those receiving other types of immunosuppressive therapy.
The opinions above are excerpted from an editorial accompanying the research report (Ann. Rheum. Dis. 2010;69:1735-7) by Dr. Kevin L. Winthrop, of the department of infectious diseases at the Oregon Health and Science University, Portland, and Dr. Daniel E. Furst, a Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles. Dr. Winthrop reported receiving funding from the Agency for Healthcare Research and Quality for work on the manuscript, and receiving a grant from UCB Pharmaceuticals, as well as consulting fees from Amgen, Wyeth, and Genentech. Dr. Furst reported receiving research support for studies of abatacept, adalimumab, certolizumab, etanercept, infliximab, rituximab, and tocilizumab, and consulting with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Genentech, and UCB.
Rheumatic disease patients who are exposed to tumor necrosis factor antagonists have a 10-fold increased risk of hospitalization for varicella zoster virus infections, compared with the general population, according to a secondary analysis of two large databases.
Nonetheless, the absolute incidence of varicella-related hospitalizations remains low at about three cases per 10,000 person-years of exposure, and the risks of using vaccination for prevention likely outweigh the benefits, Dr. Ignacio Garcia-Doval of Complexo Hospitalario de Pontevedra (Spain) and colleagues have reported in the October issue of the Annals of the Rheumatic Diseases.
The estimated incidence rate of hospitalization for shingles in the rheumatic population was 32 cases per 100,000 patient years, compared with an expected rate of 3.4 in the general population, and the estimated incidence of hospitalization for chickenpox in the rheumatic patients was 26 per 100,000, compared with 1.9 in the general population. This finding is based on analysis of data from a national registry of rheumatic disease patients who were treated with TNF agents (BIOBADASER database) and from an administrative database of all hospital admissions in public centers in Spain (Conjunto Mínimo Básico de Datos al Alta Hospitalaria, or CMBD), which together represent more than 114 million patient-years.
The estimated age- and sex-standardized incidence rate per 100,000 person-years, and the estimated standardized incidence difference were 9 and 26, respectively, for shingles, and 19 and 33, respectively, for chickenpox, they said (Ann. Rheum. Dis. 2010;69:1751-5).
TNF antagonists are known to be associated with an increased risk of tuberculosis in particular and of opportunistic infections in general. There is a biological basis for an increased risk of viral infections, the investigators said, noting that although some studies have shown an increased rate of viral infection in TNF antagonist–treated patients, the clinical relevance of the increase is uncertain.
The current study does not allow differentiation of the causes for the increased risk, but it does show that the absolute rate is low.
The investigators said it is unlikely that the cohorts received systematic vaccination against varicella zoster virus, since the general health mandate in Spain was given in 2005 and only for children aged 11-14 years.
"Standard guidelines for chickenpox vaccination probably apply to the population included in our study," they wrote.
However, shingles vaccine, which is an attenuated vaccine with a higher dose of antigen, could potentially lead to more side effects in an immunosuppressed population, they said.
For example, in a randomized trial of adults older than age 60 years, shingles vaccine was associated with 7 cases of severe adverse events and 14 cases of vaccine-related adverse events per 10,000 vaccinations, they noted.
"These vaccination-associated risks are similar in rate and severity to the risks of hospitalized infections in our study. Hence, shingles vaccination before starting a TNF antagonist may not be warranted at present," they wrote, concluding that although vaccination in healthy children is warranted, it is not warranted in adults with "immunosuppression secondary to the baseline inflammatory disease and its complications."
Disclosures: Various study authors reported serving on the advisory board for, and/or receiving lecture fees or honoraria from, Wyeth, Abbott, Schering-Plough, Roche, and/or Bristol-Myers Squibb.
Rheumatic disease patients who are exposed to tumor necrosis factor antagonists have a 10-fold increased risk of hospitalization for varicella zoster virus infections, compared with the general population, according to a secondary analysis of two large databases.
Nonetheless, the absolute incidence of varicella-related hospitalizations remains low at about three cases per 10,000 person-years of exposure, and the risks of using vaccination for prevention likely outweigh the benefits, Dr. Ignacio Garcia-Doval of Complexo Hospitalario de Pontevedra (Spain) and colleagues have reported in the October issue of the Annals of the Rheumatic Diseases.
The estimated incidence rate of hospitalization for shingles in the rheumatic population was 32 cases per 100,000 patient years, compared with an expected rate of 3.4 in the general population, and the estimated incidence of hospitalization for chickenpox in the rheumatic patients was 26 per 100,000, compared with 1.9 in the general population. This finding is based on analysis of data from a national registry of rheumatic disease patients who were treated with TNF agents (BIOBADASER database) and from an administrative database of all hospital admissions in public centers in Spain (Conjunto Mínimo Básico de Datos al Alta Hospitalaria, or CMBD), which together represent more than 114 million patient-years.
The estimated age- and sex-standardized incidence rate per 100,000 person-years, and the estimated standardized incidence difference were 9 and 26, respectively, for shingles, and 19 and 33, respectively, for chickenpox, they said (Ann. Rheum. Dis. 2010;69:1751-5).
TNF antagonists are known to be associated with an increased risk of tuberculosis in particular and of opportunistic infections in general. There is a biological basis for an increased risk of viral infections, the investigators said, noting that although some studies have shown an increased rate of viral infection in TNF antagonist–treated patients, the clinical relevance of the increase is uncertain.
The current study does not allow differentiation of the causes for the increased risk, but it does show that the absolute rate is low.
The investigators said it is unlikely that the cohorts received systematic vaccination against varicella zoster virus, since the general health mandate in Spain was given in 2005 and only for children aged 11-14 years.
"Standard guidelines for chickenpox vaccination probably apply to the population included in our study," they wrote.
However, shingles vaccine, which is an attenuated vaccine with a higher dose of antigen, could potentially lead to more side effects in an immunosuppressed population, they said.
For example, in a randomized trial of adults older than age 60 years, shingles vaccine was associated with 7 cases of severe adverse events and 14 cases of vaccine-related adverse events per 10,000 vaccinations, they noted.
"These vaccination-associated risks are similar in rate and severity to the risks of hospitalized infections in our study. Hence, shingles vaccination before starting a TNF antagonist may not be warranted at present," they wrote, concluding that although vaccination in healthy children is warranted, it is not warranted in adults with "immunosuppression secondary to the baseline inflammatory disease and its complications."
Disclosures: Various study authors reported serving on the advisory board for, and/or receiving lecture fees or honoraria from, Wyeth, Abbott, Schering-Plough, Roche, and/or Bristol-Myers Squibb.
FROM THE ANNALS OF THE RHEUMATIC DISEASES
Major Finding: The estimated incidence rate of hospitalization for shingles in the rheumatic population was 32 cases per 100,000 patient-years, compared with an expected rate of 3.4 in the general population. The estimated incidence of hospitalization for chickenpox in the rheumatic patients was 26 per 100,000, compared with 1.9 in the general population. The absolute incidence of varicella-related hospitalizations remains low, at about 3 cases per 10,000 person-years of exposure.
Data Source: A secondary analysis of data from two large databases.
Disclosures: Various authors on the study reported serving on the advisory board for, and/or receiving lecture fees or honoraria from Wyeth, Abbott, Schering-Plough, Roche, and/or Bristol-Myers Squibb. Editorial author Dr. Winthrop reported receiving funding from the Agency for Healthcare Research and Quality for work on the manuscript, and receiving a grant from UCB Pharmaceuticals, and consulting fees from Amgen, Wyeth, and Genentech. Dr. Furst reported receiving research support for studies of abatacept, adalimumab, certolizumab, etanercept, infliximab, rituximab, and tocilizumab, and consulting with Abbott, Amgen, Bristol-Myers Squibb, Centocor, Genentech, and UCB.
Dressing Up Psoriasis
Suffering for fashion is a time-honored tradition, from bustles and corsets to sky-high heels and pointy toes. But psoriasis patients (approximately 7.5 million adults in the United States) suffer even more than most. And 80% have plaque-type psoriasis, which can make it especially tough to find clothes that don't irritate their skin, let alone clothes with style.
Fortunately, there are some fashion doctors in town: Tim Gunn, who mentors up-and-coming designers on "Project Runway," and Dr. Susan Taylor, founder of the Skin of Color Center at St. Luke’s and Roosevelt Hospitals in New York, Ny.
In an online collection of videos called "Psophisticated Style: A Guide to Everyday Style and Psoriasis," Dr. Taylor and Gunn provide personalized fashion advice to five psoriasis patients. The patients were selected in a contest that was part of an ongoing "Addressing Psoriasis" public awareness campaign sponsored by Amgen and Pfizer (yes, both of these companies make psoriasis drugs, as if you had to ask).
The videos are fun, funky, and fashion-forward, with some style tips that anyone – psoriasis patient or not – can use.
The Fabulous Fabrics video includes suggestions about choosing breathable fabrics, such as cotton, linen, and silk. But, as Dr. Taylor notes, it's important to experiment with different fabrics, because what feels good to one person may not be comfortable for another, breathable or not. She mentioned how one of her patients prefers the feel of polyester to cotton, and simply makes an effort to stay in air-conditioned comfort when wearing it.
A key tip from the Awesome Accessories video reminds us about using scarves to update a look – and cleverly conceal problem plaques on the neck.
In the Seasonal Styling video, we learn to try gray instead of black for suits or dresses, because gray is less likely to show the skin flakes that are inevitable for many psoriasis patients.
And here's Gunn's tip on proportion and the rule of thirds: Don't let your outfit cut you exactly in half; it’s not a good look for anyone. I feel more stylish already!
Heidi Splete (on twitter @hsplete)
Suffering for fashion is a time-honored tradition, from bustles and corsets to sky-high heels and pointy toes. But psoriasis patients (approximately 7.5 million adults in the United States) suffer even more than most. And 80% have plaque-type psoriasis, which can make it especially tough to find clothes that don't irritate their skin, let alone clothes with style.
Fortunately, there are some fashion doctors in town: Tim Gunn, who mentors up-and-coming designers on "Project Runway," and Dr. Susan Taylor, founder of the Skin of Color Center at St. Luke’s and Roosevelt Hospitals in New York, Ny.
In an online collection of videos called "Psophisticated Style: A Guide to Everyday Style and Psoriasis," Dr. Taylor and Gunn provide personalized fashion advice to five psoriasis patients. The patients were selected in a contest that was part of an ongoing "Addressing Psoriasis" public awareness campaign sponsored by Amgen and Pfizer (yes, both of these companies make psoriasis drugs, as if you had to ask).
The videos are fun, funky, and fashion-forward, with some style tips that anyone – psoriasis patient or not – can use.
The Fabulous Fabrics video includes suggestions about choosing breathable fabrics, such as cotton, linen, and silk. But, as Dr. Taylor notes, it's important to experiment with different fabrics, because what feels good to one person may not be comfortable for another, breathable or not. She mentioned how one of her patients prefers the feel of polyester to cotton, and simply makes an effort to stay in air-conditioned comfort when wearing it.
A key tip from the Awesome Accessories video reminds us about using scarves to update a look – and cleverly conceal problem plaques on the neck.
In the Seasonal Styling video, we learn to try gray instead of black for suits or dresses, because gray is less likely to show the skin flakes that are inevitable for many psoriasis patients.
And here's Gunn's tip on proportion and the rule of thirds: Don't let your outfit cut you exactly in half; it’s not a good look for anyone. I feel more stylish already!
Heidi Splete (on twitter @hsplete)
Suffering for fashion is a time-honored tradition, from bustles and corsets to sky-high heels and pointy toes. But psoriasis patients (approximately 7.5 million adults in the United States) suffer even more than most. And 80% have plaque-type psoriasis, which can make it especially tough to find clothes that don't irritate their skin, let alone clothes with style.
Fortunately, there are some fashion doctors in town: Tim Gunn, who mentors up-and-coming designers on "Project Runway," and Dr. Susan Taylor, founder of the Skin of Color Center at St. Luke’s and Roosevelt Hospitals in New York, Ny.
In an online collection of videos called "Psophisticated Style: A Guide to Everyday Style and Psoriasis," Dr. Taylor and Gunn provide personalized fashion advice to five psoriasis patients. The patients were selected in a contest that was part of an ongoing "Addressing Psoriasis" public awareness campaign sponsored by Amgen and Pfizer (yes, both of these companies make psoriasis drugs, as if you had to ask).
The videos are fun, funky, and fashion-forward, with some style tips that anyone – psoriasis patient or not – can use.
The Fabulous Fabrics video includes suggestions about choosing breathable fabrics, such as cotton, linen, and silk. But, as Dr. Taylor notes, it's important to experiment with different fabrics, because what feels good to one person may not be comfortable for another, breathable or not. She mentioned how one of her patients prefers the feel of polyester to cotton, and simply makes an effort to stay in air-conditioned comfort when wearing it.
A key tip from the Awesome Accessories video reminds us about using scarves to update a look – and cleverly conceal problem plaques on the neck.
In the Seasonal Styling video, we learn to try gray instead of black for suits or dresses, because gray is less likely to show the skin flakes that are inevitable for many psoriasis patients.
And here's Gunn's tip on proportion and the rule of thirds: Don't let your outfit cut you exactly in half; it’s not a good look for anyone. I feel more stylish already!
Heidi Splete (on twitter @hsplete)
Bosentan Reduced Occurrence of Digital Ulcers in Systemic Sclerosis
Treatment with bosentan reduced the occurrence of new ischemic digital ulcers in patients with systemic sclerosis, but did not hasten healing of existing ulcers, findings from a study have shown.
The dual endothelin receptor antagonist was associated with a 30% reduction in the number of new digital ulcers in a double-blind, placebo-controlled trial reported online in the Annals of the Rheumatic Diseases.
In the United States, the Food and Drug Administration has approved bosentan (Tracleer) for the treatment of pulmonary artery hypertension (PAH). Both PAH and digital ulcers are complications of systemic sclerosis (SSc). In Europe, bosentan is approved for pulmonary artery hypertension.
As a follow-up to a previous study of 122 SSc patients in whom bosentan treatment was associated with a 48% reduction, compared with placebo, in the mean number of new digital ulcers but no differences in healing after 16 weeks of treatment, Dr. Marco Matucci-Cerinic, professor of rheumatology and medicine at the University of Florence (Italy) and colleagues designed the current trial to evaluate the effects of the drug in a larger population of patients over a longer period of time.
Toward this end, the investigators enrolled 188 SSc patients with at least one active digital ulcer (the cardinal ulcer) to receive 62.5 mg of bosentan twice daily for 4 weeks and 125 mg twice daily for 20 weeks, or matching placebo.
Patients in both groups were at least 18 years old at the time of enrollment (between October 2003 and May 2005) and were well matched with respect to demographics, baseline disease characteristics, and concomitant treatment for digital ulcers at baseline.
The study's primary end points were the number of new digital ulcers and the time to healing of the cardinal ulcer; the secondary end points were pain, disability, and safety, the authors wrote (Ann. Rheum. Dis. 2010 Aug. 30 [doi:10.1136/ard.2010.130658]).
After 24 weeks, the mean total number of digital ulcers per patient was similar in the two groups, but the mean number of new digital ulcers was 1.9 in the treatment group and 2.7 in the placebo group, the authors reported. "Fewer new [digital ulcers] were observed with bosentan than placebo in all subgroups except among current smokers," they stated. "This included subgroups of limited and diffuse [SSc], with no difference between the two subgroups in the treatment effect."
In post hoc analysis, more pronounced reductions were seen in patients who had multiple digital ulcers, the authors observed. Specifically, they wrote, "the reduction of new [digital ulcers] appeared to be greater in patients with at least four [digital ulcers] at baseline."
The reduction of new digital ulcers in bosentan-treated patients "did not translate into a smaller ulcer burden, as was seen in the previous study," the authors stated, noting that this could be attributed to the fact that 38% of patients in the previous study did not have an active digital ulcer at baseline and had fewer digital ulcers to heal, possibly giving more weight to prevention in the reduction of overall ulcer burden.
In terms of healing, there was no difference in the time to healing of the cardinal ulcer between bosentan treatment and placebo. At 24 weeks, more than half of the patients in both groups had healing of the cardinal ulcer that was maintained for a minimum of 12 weeks, the authors wrote.
Additionally, no treatment effects were observed in patient-rated measures of overall hand pain and pain of the cardinal ulcer, as assessed by visual analog scales. Similarly, there were no significant differences in the changes from baseline in the Health Assessment Questionnaire–Disability Index and hand disability index at week 24, they reported.
The fact that the reduction in new ulcers was not associated with decreased pain or disability, relative to placebo, could be explained by the similar number of digital ulcers between treatment groups, or a lack of sensitivity to change and discriminative value in the current instruments that are used to assess hand function in systemic sclerosis, the authors hypothesized. It might also be indicative of the fact that bosentan treatment does not improve pain and disability in spite of the reduction of new digital ulcers, they wrote.
Safety and tolerability assessments showed that serious adverse events occurred in 9.4% and 16.7%, respectively, of patients in the bosentan and placebo groups, and that similar proportions of patients in both groups experienced at least one adverse event, the authors reported. More treatment vs. placebo patients experienced peripheral edema and events denoting elevated aminotransferases.
Laboratory analyses showed increases in aminotransferases in 10.5% of the bosentan patients, compared with 1.1% of the placebo patients, reinforcing "the need for continual monitoring of liver function with this treatment," the authors wrote.
The clinical utility of bosentan treatment for digital ulcer prevention in SSc "may be challenged," the authors wrote. "In a patient encountered with a single [digital ulcer], initiation of bosentan would not be expected to facilitate healing, and at least 66% of all bosentan-treated subjects would develop at least one additional [digital ulcer] over 6 months of follow-up." However, they noted, bosentan treatment offers greater potential benefit to patients who present with multiple digital ulcers. In this regard, the treatment "may be a useful adjunct in the management of patients with [SSc] and recurrent [digital ulcers]."
Disclosures: This study was funded by Actelion Pharmaceuticals. The authors disclosed financial relationships with Actelion, Pfizer, GlaxoSmithKline Beecham, Encysive, Genzyme, Aspreva, Biovitrum, DiGNA, Gilead, MediQuest, Centocor, FibroGen, Bristol-Myers Squibb, Lilly, and United Therapeutics.
Treatment with bosentan reduced the occurrence of new ischemic digital ulcers in patients with systemic sclerosis, but did not hasten healing of existing ulcers, findings from a study have shown.
The dual endothelin receptor antagonist was associated with a 30% reduction in the number of new digital ulcers in a double-blind, placebo-controlled trial reported online in the Annals of the Rheumatic Diseases.
In the United States, the Food and Drug Administration has approved bosentan (Tracleer) for the treatment of pulmonary artery hypertension (PAH). Both PAH and digital ulcers are complications of systemic sclerosis (SSc). In Europe, bosentan is approved for pulmonary artery hypertension.
As a follow-up to a previous study of 122 SSc patients in whom bosentan treatment was associated with a 48% reduction, compared with placebo, in the mean number of new digital ulcers but no differences in healing after 16 weeks of treatment, Dr. Marco Matucci-Cerinic, professor of rheumatology and medicine at the University of Florence (Italy) and colleagues designed the current trial to evaluate the effects of the drug in a larger population of patients over a longer period of time.
Toward this end, the investigators enrolled 188 SSc patients with at least one active digital ulcer (the cardinal ulcer) to receive 62.5 mg of bosentan twice daily for 4 weeks and 125 mg twice daily for 20 weeks, or matching placebo.
Patients in both groups were at least 18 years old at the time of enrollment (between October 2003 and May 2005) and were well matched with respect to demographics, baseline disease characteristics, and concomitant treatment for digital ulcers at baseline.
The study's primary end points were the number of new digital ulcers and the time to healing of the cardinal ulcer; the secondary end points were pain, disability, and safety, the authors wrote (Ann. Rheum. Dis. 2010 Aug. 30 [doi:10.1136/ard.2010.130658]).
After 24 weeks, the mean total number of digital ulcers per patient was similar in the two groups, but the mean number of new digital ulcers was 1.9 in the treatment group and 2.7 in the placebo group, the authors reported. "Fewer new [digital ulcers] were observed with bosentan than placebo in all subgroups except among current smokers," they stated. "This included subgroups of limited and diffuse [SSc], with no difference between the two subgroups in the treatment effect."
In post hoc analysis, more pronounced reductions were seen in patients who had multiple digital ulcers, the authors observed. Specifically, they wrote, "the reduction of new [digital ulcers] appeared to be greater in patients with at least four [digital ulcers] at baseline."
The reduction of new digital ulcers in bosentan-treated patients "did not translate into a smaller ulcer burden, as was seen in the previous study," the authors stated, noting that this could be attributed to the fact that 38% of patients in the previous study did not have an active digital ulcer at baseline and had fewer digital ulcers to heal, possibly giving more weight to prevention in the reduction of overall ulcer burden.
In terms of healing, there was no difference in the time to healing of the cardinal ulcer between bosentan treatment and placebo. At 24 weeks, more than half of the patients in both groups had healing of the cardinal ulcer that was maintained for a minimum of 12 weeks, the authors wrote.
Additionally, no treatment effects were observed in patient-rated measures of overall hand pain and pain of the cardinal ulcer, as assessed by visual analog scales. Similarly, there were no significant differences in the changes from baseline in the Health Assessment Questionnaire–Disability Index and hand disability index at week 24, they reported.
The fact that the reduction in new ulcers was not associated with decreased pain or disability, relative to placebo, could be explained by the similar number of digital ulcers between treatment groups, or a lack of sensitivity to change and discriminative value in the current instruments that are used to assess hand function in systemic sclerosis, the authors hypothesized. It might also be indicative of the fact that bosentan treatment does not improve pain and disability in spite of the reduction of new digital ulcers, they wrote.
Safety and tolerability assessments showed that serious adverse events occurred in 9.4% and 16.7%, respectively, of patients in the bosentan and placebo groups, and that similar proportions of patients in both groups experienced at least one adverse event, the authors reported. More treatment vs. placebo patients experienced peripheral edema and events denoting elevated aminotransferases.
Laboratory analyses showed increases in aminotransferases in 10.5% of the bosentan patients, compared with 1.1% of the placebo patients, reinforcing "the need for continual monitoring of liver function with this treatment," the authors wrote.
The clinical utility of bosentan treatment for digital ulcer prevention in SSc "may be challenged," the authors wrote. "In a patient encountered with a single [digital ulcer], initiation of bosentan would not be expected to facilitate healing, and at least 66% of all bosentan-treated subjects would develop at least one additional [digital ulcer] over 6 months of follow-up." However, they noted, bosentan treatment offers greater potential benefit to patients who present with multiple digital ulcers. In this regard, the treatment "may be a useful adjunct in the management of patients with [SSc] and recurrent [digital ulcers]."
Disclosures: This study was funded by Actelion Pharmaceuticals. The authors disclosed financial relationships with Actelion, Pfizer, GlaxoSmithKline Beecham, Encysive, Genzyme, Aspreva, Biovitrum, DiGNA, Gilead, MediQuest, Centocor, FibroGen, Bristol-Myers Squibb, Lilly, and United Therapeutics.
Treatment with bosentan reduced the occurrence of new ischemic digital ulcers in patients with systemic sclerosis, but did not hasten healing of existing ulcers, findings from a study have shown.
The dual endothelin receptor antagonist was associated with a 30% reduction in the number of new digital ulcers in a double-blind, placebo-controlled trial reported online in the Annals of the Rheumatic Diseases.
In the United States, the Food and Drug Administration has approved bosentan (Tracleer) for the treatment of pulmonary artery hypertension (PAH). Both PAH and digital ulcers are complications of systemic sclerosis (SSc). In Europe, bosentan is approved for pulmonary artery hypertension.
As a follow-up to a previous study of 122 SSc patients in whom bosentan treatment was associated with a 48% reduction, compared with placebo, in the mean number of new digital ulcers but no differences in healing after 16 weeks of treatment, Dr. Marco Matucci-Cerinic, professor of rheumatology and medicine at the University of Florence (Italy) and colleagues designed the current trial to evaluate the effects of the drug in a larger population of patients over a longer period of time.
Toward this end, the investigators enrolled 188 SSc patients with at least one active digital ulcer (the cardinal ulcer) to receive 62.5 mg of bosentan twice daily for 4 weeks and 125 mg twice daily for 20 weeks, or matching placebo.
Patients in both groups were at least 18 years old at the time of enrollment (between October 2003 and May 2005) and were well matched with respect to demographics, baseline disease characteristics, and concomitant treatment for digital ulcers at baseline.
The study's primary end points were the number of new digital ulcers and the time to healing of the cardinal ulcer; the secondary end points were pain, disability, and safety, the authors wrote (Ann. Rheum. Dis. 2010 Aug. 30 [doi:10.1136/ard.2010.130658]).
After 24 weeks, the mean total number of digital ulcers per patient was similar in the two groups, but the mean number of new digital ulcers was 1.9 in the treatment group and 2.7 in the placebo group, the authors reported. "Fewer new [digital ulcers] were observed with bosentan than placebo in all subgroups except among current smokers," they stated. "This included subgroups of limited and diffuse [SSc], with no difference between the two subgroups in the treatment effect."
In post hoc analysis, more pronounced reductions were seen in patients who had multiple digital ulcers, the authors observed. Specifically, they wrote, "the reduction of new [digital ulcers] appeared to be greater in patients with at least four [digital ulcers] at baseline."
The reduction of new digital ulcers in bosentan-treated patients "did not translate into a smaller ulcer burden, as was seen in the previous study," the authors stated, noting that this could be attributed to the fact that 38% of patients in the previous study did not have an active digital ulcer at baseline and had fewer digital ulcers to heal, possibly giving more weight to prevention in the reduction of overall ulcer burden.
In terms of healing, there was no difference in the time to healing of the cardinal ulcer between bosentan treatment and placebo. At 24 weeks, more than half of the patients in both groups had healing of the cardinal ulcer that was maintained for a minimum of 12 weeks, the authors wrote.
Additionally, no treatment effects were observed in patient-rated measures of overall hand pain and pain of the cardinal ulcer, as assessed by visual analog scales. Similarly, there were no significant differences in the changes from baseline in the Health Assessment Questionnaire–Disability Index and hand disability index at week 24, they reported.
The fact that the reduction in new ulcers was not associated with decreased pain or disability, relative to placebo, could be explained by the similar number of digital ulcers between treatment groups, or a lack of sensitivity to change and discriminative value in the current instruments that are used to assess hand function in systemic sclerosis, the authors hypothesized. It might also be indicative of the fact that bosentan treatment does not improve pain and disability in spite of the reduction of new digital ulcers, they wrote.
Safety and tolerability assessments showed that serious adverse events occurred in 9.4% and 16.7%, respectively, of patients in the bosentan and placebo groups, and that similar proportions of patients in both groups experienced at least one adverse event, the authors reported. More treatment vs. placebo patients experienced peripheral edema and events denoting elevated aminotransferases.
Laboratory analyses showed increases in aminotransferases in 10.5% of the bosentan patients, compared with 1.1% of the placebo patients, reinforcing "the need for continual monitoring of liver function with this treatment," the authors wrote.
The clinical utility of bosentan treatment for digital ulcer prevention in SSc "may be challenged," the authors wrote. "In a patient encountered with a single [digital ulcer], initiation of bosentan would not be expected to facilitate healing, and at least 66% of all bosentan-treated subjects would develop at least one additional [digital ulcer] over 6 months of follow-up." However, they noted, bosentan treatment offers greater potential benefit to patients who present with multiple digital ulcers. In this regard, the treatment "may be a useful adjunct in the management of patients with [SSc] and recurrent [digital ulcers]."
Disclosures: This study was funded by Actelion Pharmaceuticals. The authors disclosed financial relationships with Actelion, Pfizer, GlaxoSmithKline Beecham, Encysive, Genzyme, Aspreva, Biovitrum, DiGNA, Gilead, MediQuest, Centocor, FibroGen, Bristol-Myers Squibb, Lilly, and United Therapeutics.
Major Finding: Treatment with bosentan reduced the occurrence of new digital ulcers in SSc patients who had multiple ulcers at treatment initiation by 30%.
Data Source: A randomized, double-blind, placebo-controlled study comprising 188 systemic sclerosis patients.
Disclosures: This study was funded by Actelion Pharmaceuticals. The authors disclosed financial relationships with Actelion, Pfizer, GlaxoSmithKline Beecham, Encysive, Genzyme, Aspreva, Biovitrum, DiGNA, Gilead, MediQuest, Centocor, FibroGen, Bristol-Myers Squibb, Lilly, and United Therapeutics.
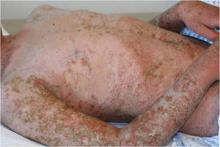
Cyclosporine Often Best Option for Pediatric Pustular Psoriasis Flare
PORTLAND, Ore. - Cyclosporine is often the best rescue therapy for acute generalized pustular psoriasis in children, according to Dr. Kelly M. Cordoro.
“When speed is important, my first choice is cyclosporine,” said Dr. Cordoro of the department of dermatology at the University of California, San Francisco. When adequately dosed, it can end pustulation within days.
Methotrexate and acitretin are also options, but can take several weeks to have maximum benefit, which is too slow for acute generalized pustular psoriasis (GPP) patients. GPP can cause fatal secondary infections, calcium imbalances, and cardiorespiratory collapse, although these outcomes are rare in children. She also noted methotrexate’s hepatotoxic potential, a concern in the treatment of GPP because the disease itself can cause acute liver dysfunction.
Acute GPP patients, apart from the pathognomonic skin findings, can present with fever, anorexia, and chills. Patients “are very sick,” she said.
Severe cases warrant hospital admission for treatment and supportive care, including rehydration, electrolyte imbalance correction, and bed rest. Compresses followed by bland emollients, such as petrolatum jelly, are essential to help prevent secondary infections and sepsis.
Rescue therapy is only the first of three stages by which Dr. Cordoro conceptualizes GPP management.
The second, transition therapy, usually includes tapering the cyclosporine and initiating other treatments for long-term control, such as acitretin, phototherapy, or both.
The maintenance stage comes after the patient has been stable or clear for months. Phototherapy and topical vitamin D analogues, such as Dovonex, are “great choices for maintenance,” Dr. Cordoro said. Low-dose acitretin, topical corticosteroids, and topical calcineurin inhibitors are other options.
Dr. Cordoro also tests the pharynx and perianal area for group A streptococcus colonization, and treats to eliminate it once her GPP patients are well enough for antibiotics. “It’s not known if it helps in the acute setting, but strep antigen may trigger psoriasis again at some point,” she said.
There are no consensus treatment guidelines for GPP in children; clinical decisions are based on patient characteristics, clinical experience and setting, and hunches about the best way to proceed. “There’s no cookbook approach, no one-size-fits-all treatment,” Dr. Cordoro said.
One of her patients, an 8-year-old boy with severe, relapsing/remitting GPP, illustrates the point.
He had been doing fairly well on low-dose acitretin, but then flared. His mother took him to an urgent care center, and the doctor there, mistaking GPP for an infection, started the boy on a cephalosporin.
His condition worsened and he was toxic by the time Dr. Cordoro arrived.
She admitted the patient to the hospital and initiated rescue doses of cyclosporine 5 mg/kg per day, along with supportive care. His condition deteriorated over the next few days, and his liver enzymes became elevated. Since he was failing on cyclosporine, she wanted to use infliximab for rescue therapy.
The elevated enzymes, though, caused a dilemma: Was it safe to start a potentially hepatotoxic tumor necrosis factor inhibitor in a patient with elevated liver enzymes? And were the enzymes elevated because of the GPP flare itself, or one of the concomitant medications?
Because cephalosporins can cause transient liver enzyme elevations, Dr. Cordoro discontinued the cephalosporin; the boy’s enzymes dropped within a day, which cleared the way for infliximab.
He turned the corner in 2 days; pustulation ceased and his skin began to heal. Given the severity of his flare, he is on adalimumab for maintenance therapy, Dr. Cordoro said.
“It gets complicated. You have to take everything into account,” she said, but “children with GPP tend to have an excellent prognosis as long as secondary complications are prevented or managed.”
Dr. Cordoro noted an educational Web site for children where they can learn about psoriasis and interact with other children with the condition.
Disclosures: Dr. Cordoro said she had no conflicts of interest.
PORTLAND, Ore. - Cyclosporine is often the best rescue therapy for acute generalized pustular psoriasis in children, according to Dr. Kelly M. Cordoro.
“When speed is important, my first choice is cyclosporine,” said Dr. Cordoro of the department of dermatology at the University of California, San Francisco. When adequately dosed, it can end pustulation within days.
Methotrexate and acitretin are also options, but can take several weeks to have maximum benefit, which is too slow for acute generalized pustular psoriasis (GPP) patients. GPP can cause fatal secondary infections, calcium imbalances, and cardiorespiratory collapse, although these outcomes are rare in children. She also noted methotrexate’s hepatotoxic potential, a concern in the treatment of GPP because the disease itself can cause acute liver dysfunction.
Acute GPP patients, apart from the pathognomonic skin findings, can present with fever, anorexia, and chills. Patients “are very sick,” she said.
Severe cases warrant hospital admission for treatment and supportive care, including rehydration, electrolyte imbalance correction, and bed rest. Compresses followed by bland emollients, such as petrolatum jelly, are essential to help prevent secondary infections and sepsis.
Rescue therapy is only the first of three stages by which Dr. Cordoro conceptualizes GPP management.
The second, transition therapy, usually includes tapering the cyclosporine and initiating other treatments for long-term control, such as acitretin, phototherapy, or both.
The maintenance stage comes after the patient has been stable or clear for months. Phototherapy and topical vitamin D analogues, such as Dovonex, are “great choices for maintenance,” Dr. Cordoro said. Low-dose acitretin, topical corticosteroids, and topical calcineurin inhibitors are other options.
Dr. Cordoro also tests the pharynx and perianal area for group A streptococcus colonization, and treats to eliminate it once her GPP patients are well enough for antibiotics. “It’s not known if it helps in the acute setting, but strep antigen may trigger psoriasis again at some point,” she said.
There are no consensus treatment guidelines for GPP in children; clinical decisions are based on patient characteristics, clinical experience and setting, and hunches about the best way to proceed. “There’s no cookbook approach, no one-size-fits-all treatment,” Dr. Cordoro said.
One of her patients, an 8-year-old boy with severe, relapsing/remitting GPP, illustrates the point.
He had been doing fairly well on low-dose acitretin, but then flared. His mother took him to an urgent care center, and the doctor there, mistaking GPP for an infection, started the boy on a cephalosporin.
His condition worsened and he was toxic by the time Dr. Cordoro arrived.
She admitted the patient to the hospital and initiated rescue doses of cyclosporine 5 mg/kg per day, along with supportive care. His condition deteriorated over the next few days, and his liver enzymes became elevated. Since he was failing on cyclosporine, she wanted to use infliximab for rescue therapy.
The elevated enzymes, though, caused a dilemma: Was it safe to start a potentially hepatotoxic tumor necrosis factor inhibitor in a patient with elevated liver enzymes? And were the enzymes elevated because of the GPP flare itself, or one of the concomitant medications?
Because cephalosporins can cause transient liver enzyme elevations, Dr. Cordoro discontinued the cephalosporin; the boy’s enzymes dropped within a day, which cleared the way for infliximab.
He turned the corner in 2 days; pustulation ceased and his skin began to heal. Given the severity of his flare, he is on adalimumab for maintenance therapy, Dr. Cordoro said.
“It gets complicated. You have to take everything into account,” she said, but “children with GPP tend to have an excellent prognosis as long as secondary complications are prevented or managed.”
Dr. Cordoro noted an educational Web site for children where they can learn about psoriasis and interact with other children with the condition.
Disclosures: Dr. Cordoro said she had no conflicts of interest.
PORTLAND, Ore. - Cyclosporine is often the best rescue therapy for acute generalized pustular psoriasis in children, according to Dr. Kelly M. Cordoro.
“When speed is important, my first choice is cyclosporine,” said Dr. Cordoro of the department of dermatology at the University of California, San Francisco. When adequately dosed, it can end pustulation within days.
Methotrexate and acitretin are also options, but can take several weeks to have maximum benefit, which is too slow for acute generalized pustular psoriasis (GPP) patients. GPP can cause fatal secondary infections, calcium imbalances, and cardiorespiratory collapse, although these outcomes are rare in children. She also noted methotrexate’s hepatotoxic potential, a concern in the treatment of GPP because the disease itself can cause acute liver dysfunction.
Acute GPP patients, apart from the pathognomonic skin findings, can present with fever, anorexia, and chills. Patients “are very sick,” she said.
Severe cases warrant hospital admission for treatment and supportive care, including rehydration, electrolyte imbalance correction, and bed rest. Compresses followed by bland emollients, such as petrolatum jelly, are essential to help prevent secondary infections and sepsis.
Rescue therapy is only the first of three stages by which Dr. Cordoro conceptualizes GPP management.
The second, transition therapy, usually includes tapering the cyclosporine and initiating other treatments for long-term control, such as acitretin, phototherapy, or both.
The maintenance stage comes after the patient has been stable or clear for months. Phototherapy and topical vitamin D analogues, such as Dovonex, are “great choices for maintenance,” Dr. Cordoro said. Low-dose acitretin, topical corticosteroids, and topical calcineurin inhibitors are other options.
Dr. Cordoro also tests the pharynx and perianal area for group A streptococcus colonization, and treats to eliminate it once her GPP patients are well enough for antibiotics. “It’s not known if it helps in the acute setting, but strep antigen may trigger psoriasis again at some point,” she said.
There are no consensus treatment guidelines for GPP in children; clinical decisions are based on patient characteristics, clinical experience and setting, and hunches about the best way to proceed. “There’s no cookbook approach, no one-size-fits-all treatment,” Dr. Cordoro said.
One of her patients, an 8-year-old boy with severe, relapsing/remitting GPP, illustrates the point.
He had been doing fairly well on low-dose acitretin, but then flared. His mother took him to an urgent care center, and the doctor there, mistaking GPP for an infection, started the boy on a cephalosporin.
His condition worsened and he was toxic by the time Dr. Cordoro arrived.
She admitted the patient to the hospital and initiated rescue doses of cyclosporine 5 mg/kg per day, along with supportive care. His condition deteriorated over the next few days, and his liver enzymes became elevated. Since he was failing on cyclosporine, she wanted to use infliximab for rescue therapy.
The elevated enzymes, though, caused a dilemma: Was it safe to start a potentially hepatotoxic tumor necrosis factor inhibitor in a patient with elevated liver enzymes? And were the enzymes elevated because of the GPP flare itself, or one of the concomitant medications?
Because cephalosporins can cause transient liver enzyme elevations, Dr. Cordoro discontinued the cephalosporin; the boy’s enzymes dropped within a day, which cleared the way for infliximab.
He turned the corner in 2 days; pustulation ceased and his skin began to heal. Given the severity of his flare, he is on adalimumab for maintenance therapy, Dr. Cordoro said.
“It gets complicated. You have to take everything into account,” she said, but “children with GPP tend to have an excellent prognosis as long as secondary complications are prevented or managed.”
Dr. Cordoro noted an educational Web site for children where they can learn about psoriasis and interact with other children with the condition.
Disclosures: Dr. Cordoro said she had no conflicts of interest.
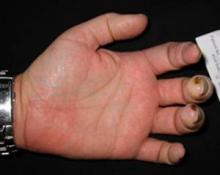
Adalimumab Found Safe for Psoriatic Diseases
CHICAGO - Adalimumab was well tolerated, with a low incidence of adverse events and serious infections, in two studies totaling almost 1,800 patients who took the drug for up to 5 years.
The first study was a meta-analysis of 11 global clinical trials, totaling almost 1,721 patients. All of the phase II or III studies included safety data analyzed in 2007 and 2008, according to a report by Dr. Richard Langley and his associates presented at the summer meeting of the American Academy of Dermatology.
The drug was administered subcutaneously in all treatment groups. Dosing regimens included 80 mg/week initially, followed by 40 mg every other week (1,039 patients were on this regimen); 40 mg every other week (422 were on this regimen); 80 mg for the first 2 weeks, followed by 40 mg every other week (198 patients were on this regimen); and 80 mg every other week (62 patients).
All patients were about 44 years old, with disease duration of 18 years. The percentage of body involved was 27%; psoriatic arthritis was present in 28%, according to Dr. Langley of Dalhousie University, Halifax.
The length of adalimumab exposure ranged from 48 weeks to 5.5 years; the mean exposure time was 2 years. At both the 2007 and 2008 safety analyses, the overall rate of adverse events was 3 events per person-year.
The most commonly reported were nasopharyngitis, upper respiratory infection, and sinusitis, which accounted for 47% of the 3,489 infectious adverse events (AEs) reported.
In the 2008 analysis, 60 serious AEs were reported in 50 patients. There were two occurrences of sepsis; the incidence rates of pneumonia were 0.002 per person-year; limb abscess occurred at a rate of 0.001 abscess per person-year; and other serious infections, less than 0.001 per person-year.
Tuberculin skin tests were positive in 138 patients in the 6% of patients who received a test at baseline. There were no cases of latent TB reactivation in those who complied with prophylactic treatment. Six patients developed tuberculosis; four of them were Asian males.
Twelve opportunistic infections occurred in 11 patients; none was considered serious. The infections included 11 cases of candidiasis and 1 case of coccidioidomycosis, which occurred in the southwestern United States where the disease is endemic.
The second study examined efficacy and adverse event rates in patients who took adalimumab for moderate-severe plaque psoriasis of the hands and feet. These 72 patients were included in a single 28-week randomized, placebo-controlled crossover trial.
The patients' mean age was 50 years. The duration of psoriasis was about 12 years; the duration of hand/feet psoriasis, about 8 years.
The crossover trial randomized 49 patients to 40 mg adalimumab every other week and 23 to placebo for 16 weeks; after that, an open-label extension trial included all patients taking 30 mg every other week.
At week 16, significantly more of the adalimumab-treated patients reported being clear or almost clear of their plaque lesions, compared with placebo (31% vs. 4%). At weeks 16 and 28 of the trial, 51% of the active group and 47% of the crossover group reported scores of clear or almost clear.
During the first 16 weeks, adverse events occurred in 63% of the active group and 70% of the placebo group. Serious adverse events occurred in none of the active group and 4% of the placebo group. Infectious AEs occurred in 35% of the active group and 44% of the placebo group. One malignancy occurred in the placebo group.
There was one opportunistic infection in the active group. Hepatic events occurred in one patient in the adalimumab group.
During the crossover phase, AEs occurred in 43% of the adalimumab group and 39% of the crossover group; the most commonly reported were nasopharyngitis, headache, diarrhea, and infection site reaction.
AEs were considered serious in two adalimumab patients. Infectious adverse events occurred in 24% of the adalimumab group and 17% of the crossover group. There was one case of heart failure in the adalimumab group.
Three adalimumab patients and two crossover patients discontinued treatment because of the AEs. There were no deaths, serious infections, cases of tuberculosis, demyelinating diseases, lupus-like syndromes, lymphomas, or non-melanoma skins cancers reported.
Disclosures: All of the studies were supported by Abbott Laboratories. Dr. Langley disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Isotechnika, LEO Pharma, NanoBio, Novartis, Pfizer, Schering, and Serono.
CHICAGO - Adalimumab was well tolerated, with a low incidence of adverse events and serious infections, in two studies totaling almost 1,800 patients who took the drug for up to 5 years.
The first study was a meta-analysis of 11 global clinical trials, totaling almost 1,721 patients. All of the phase II or III studies included safety data analyzed in 2007 and 2008, according to a report by Dr. Richard Langley and his associates presented at the summer meeting of the American Academy of Dermatology.
The drug was administered subcutaneously in all treatment groups. Dosing regimens included 80 mg/week initially, followed by 40 mg every other week (1,039 patients were on this regimen); 40 mg every other week (422 were on this regimen); 80 mg for the first 2 weeks, followed by 40 mg every other week (198 patients were on this regimen); and 80 mg every other week (62 patients).
All patients were about 44 years old, with disease duration of 18 years. The percentage of body involved was 27%; psoriatic arthritis was present in 28%, according to Dr. Langley of Dalhousie University, Halifax.
The length of adalimumab exposure ranged from 48 weeks to 5.5 years; the mean exposure time was 2 years. At both the 2007 and 2008 safety analyses, the overall rate of adverse events was 3 events per person-year.
The most commonly reported were nasopharyngitis, upper respiratory infection, and sinusitis, which accounted for 47% of the 3,489 infectious adverse events (AEs) reported.
In the 2008 analysis, 60 serious AEs were reported in 50 patients. There were two occurrences of sepsis; the incidence rates of pneumonia were 0.002 per person-year; limb abscess occurred at a rate of 0.001 abscess per person-year; and other serious infections, less than 0.001 per person-year.
Tuberculin skin tests were positive in 138 patients in the 6% of patients who received a test at baseline. There were no cases of latent TB reactivation in those who complied with prophylactic treatment. Six patients developed tuberculosis; four of them were Asian males.
Twelve opportunistic infections occurred in 11 patients; none was considered serious. The infections included 11 cases of candidiasis and 1 case of coccidioidomycosis, which occurred in the southwestern United States where the disease is endemic.
The second study examined efficacy and adverse event rates in patients who took adalimumab for moderate-severe plaque psoriasis of the hands and feet. These 72 patients were included in a single 28-week randomized, placebo-controlled crossover trial.
The patients' mean age was 50 years. The duration of psoriasis was about 12 years; the duration of hand/feet psoriasis, about 8 years.
The crossover trial randomized 49 patients to 40 mg adalimumab every other week and 23 to placebo for 16 weeks; after that, an open-label extension trial included all patients taking 30 mg every other week.
At week 16, significantly more of the adalimumab-treated patients reported being clear or almost clear of their plaque lesions, compared with placebo (31% vs. 4%). At weeks 16 and 28 of the trial, 51% of the active group and 47% of the crossover group reported scores of clear or almost clear.
During the first 16 weeks, adverse events occurred in 63% of the active group and 70% of the placebo group. Serious adverse events occurred in none of the active group and 4% of the placebo group. Infectious AEs occurred in 35% of the active group and 44% of the placebo group. One malignancy occurred in the placebo group.
There was one opportunistic infection in the active group. Hepatic events occurred in one patient in the adalimumab group.
During the crossover phase, AEs occurred in 43% of the adalimumab group and 39% of the crossover group; the most commonly reported were nasopharyngitis, headache, diarrhea, and infection site reaction.
AEs were considered serious in two adalimumab patients. Infectious adverse events occurred in 24% of the adalimumab group and 17% of the crossover group. There was one case of heart failure in the adalimumab group.
Three adalimumab patients and two crossover patients discontinued treatment because of the AEs. There were no deaths, serious infections, cases of tuberculosis, demyelinating diseases, lupus-like syndromes, lymphomas, or non-melanoma skins cancers reported.
Disclosures: All of the studies were supported by Abbott Laboratories. Dr. Langley disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Isotechnika, LEO Pharma, NanoBio, Novartis, Pfizer, Schering, and Serono.
CHICAGO - Adalimumab was well tolerated, with a low incidence of adverse events and serious infections, in two studies totaling almost 1,800 patients who took the drug for up to 5 years.
The first study was a meta-analysis of 11 global clinical trials, totaling almost 1,721 patients. All of the phase II or III studies included safety data analyzed in 2007 and 2008, according to a report by Dr. Richard Langley and his associates presented at the summer meeting of the American Academy of Dermatology.
The drug was administered subcutaneously in all treatment groups. Dosing regimens included 80 mg/week initially, followed by 40 mg every other week (1,039 patients were on this regimen); 40 mg every other week (422 were on this regimen); 80 mg for the first 2 weeks, followed by 40 mg every other week (198 patients were on this regimen); and 80 mg every other week (62 patients).
All patients were about 44 years old, with disease duration of 18 years. The percentage of body involved was 27%; psoriatic arthritis was present in 28%, according to Dr. Langley of Dalhousie University, Halifax.
The length of adalimumab exposure ranged from 48 weeks to 5.5 years; the mean exposure time was 2 years. At both the 2007 and 2008 safety analyses, the overall rate of adverse events was 3 events per person-year.
The most commonly reported were nasopharyngitis, upper respiratory infection, and sinusitis, which accounted for 47% of the 3,489 infectious adverse events (AEs) reported.
In the 2008 analysis, 60 serious AEs were reported in 50 patients. There were two occurrences of sepsis; the incidence rates of pneumonia were 0.002 per person-year; limb abscess occurred at a rate of 0.001 abscess per person-year; and other serious infections, less than 0.001 per person-year.
Tuberculin skin tests were positive in 138 patients in the 6% of patients who received a test at baseline. There were no cases of latent TB reactivation in those who complied with prophylactic treatment. Six patients developed tuberculosis; four of them were Asian males.
Twelve opportunistic infections occurred in 11 patients; none was considered serious. The infections included 11 cases of candidiasis and 1 case of coccidioidomycosis, which occurred in the southwestern United States where the disease is endemic.
The second study examined efficacy and adverse event rates in patients who took adalimumab for moderate-severe plaque psoriasis of the hands and feet. These 72 patients were included in a single 28-week randomized, placebo-controlled crossover trial.
The patients' mean age was 50 years. The duration of psoriasis was about 12 years; the duration of hand/feet psoriasis, about 8 years.
The crossover trial randomized 49 patients to 40 mg adalimumab every other week and 23 to placebo for 16 weeks; after that, an open-label extension trial included all patients taking 30 mg every other week.
At week 16, significantly more of the adalimumab-treated patients reported being clear or almost clear of their plaque lesions, compared with placebo (31% vs. 4%). At weeks 16 and 28 of the trial, 51% of the active group and 47% of the crossover group reported scores of clear or almost clear.
During the first 16 weeks, adverse events occurred in 63% of the active group and 70% of the placebo group. Serious adverse events occurred in none of the active group and 4% of the placebo group. Infectious AEs occurred in 35% of the active group and 44% of the placebo group. One malignancy occurred in the placebo group.
There was one opportunistic infection in the active group. Hepatic events occurred in one patient in the adalimumab group.
During the crossover phase, AEs occurred in 43% of the adalimumab group and 39% of the crossover group; the most commonly reported were nasopharyngitis, headache, diarrhea, and infection site reaction.
AEs were considered serious in two adalimumab patients. Infectious adverse events occurred in 24% of the adalimumab group and 17% of the crossover group. There was one case of heart failure in the adalimumab group.
Three adalimumab patients and two crossover patients discontinued treatment because of the AEs. There were no deaths, serious infections, cases of tuberculosis, demyelinating diseases, lupus-like syndromes, lymphomas, or non-melanoma skins cancers reported.
Disclosures: All of the studies were supported by Abbott Laboratories. Dr. Langley disclosed financial relationships with Abbott, Amgen, Bristol-Myers Squibb, Celgene, Centocor, Isotechnika, LEO Pharma, NanoBio, Novartis, Pfizer, Schering, and Serono.
Vitamin D - Myths or Truths?
CHICAGO – Dr. Richard L. Gallo put on his Myth Busters hat at a recent dermatology meeting to debunk – and in some cases uphold – some of the most popular ideas about vitamin D.
"This subject is nothing new," noted Dr. Gallo, recounting a bit of vitamin D yore. "In 1936, Schlitz beer urged customers to drink the beverage because it contained 100 units of vitamin D, and could ward off colds and flu. So even back then, they were on to something."
But, Dr. Gallo questioned, is the idea that vitamin D can strengthen the immune system a reality – or a myth? And how about other claims touted in the public press, that sunlight is the best source of vitamin D, that the vitamin strengthen bones and protects against cancer.
"Unfortunately, vitamin D information has become something of a shell game, with positions that overstate the strength of the evidence. As dermatologists, for example, we know the carcinogenic potential of sunlight, but there are now opposing groups that advocate health by increasing vitamin D through sun exposure."
Myth No. 1: Fifteen minutes per day of sunlight provides adequate amounts of vitamin D.
"In a test tube, ultraviolet B is the optimal spectrum for converting 25-hydroxy D into vitamin D in the human body," Dr. Gallo said. "But randomized studies on this vary in results."
One frequently cited study examined the issue in Denmark. "Northern latitudes are very useful for studies like this because of the high intensity of the sun during the summer, and the low intensity in winter," said Dr. Gallo, chief of dermatology and professor of medicine and pediatrics at the University of California San Diego. "In this study, the 25-hydroxy D in the population varied dramatically with change in sunlight exposure, and tended to lag about 1 month behind the sunlight levels."
But the study also found that 53% of the subjects who sought sun exposure were still suboptimal in their vitamin D levels. "So sun-seeking behavior in one of the most intense sun-exposed areas of the world is not sufficient to cover optimal vitamin D in a population." (Photochem. Photobiol. 2009;85:1,480-4).
A 2009 study looked at sunlight exposure and vitamin D in twins (PLoS One 2010 5(7):e11555 [doi: 10.1371/journal.pone.0011555]). More than 200 twins were evaluated for the seasonal impact of genetic factors on serum 25-hydroxy vitamin D concentrations. "This showed very wide distributions in levels during the different seasons, and concluded that more than 50% of the variation in summer levels was not due to sun or diet, but to genetic influences independent of skin pigment," Dr. Gallo said.
"So, Myth No. 1 – busted," he concluded.
Myth No. 2: Vitamin D improves bone health.
Prospective cohort studies such as the National Health and Nutrition Examination Survey show that hip fracture is reduced by more than one-third in patients with adequate vitamin D levels (more than 60 nmol/L). "However, we still have a lot to learn. Data from a recent 3-year study of 2,000 perimenopasual women concluded that if vitamin D were given as a single annual dose of 500,000 IU, the women had a 15% increased risk of falls and a 26% increase in the risk of fractures." (JAMA 2010;303:1815-22).
"As far as Myth No. 2 goes, I'd say it's true, but we don't understand everything yet."
Myth No. 3: Vitamin D protects against cancer.
"This has been quite a popular theme in the press for years now, but there are no great mechanistic explanations as to why it may be true," Dr Gallo said. "There are a number of randomized controlled trials, but the data are inconsistent."
A 2009 Agency for Healthcare Research and Quality review examined more than 170 studies and reviews for several health outcomes and vitamin D. "Only one study really showed a level of significance [for cancer reduction]. The others showed inconsistent data on cancer and some showed a slight trend toward an increased risk for colon cancer." (Evid. Rep. Technol. Assess. (Full Rep.) 2009;183:1-420).
"Myth No. 3 is still a plausible possibility, but no benefit has been clearly demonstrated."
Myth No. 4: Vitamin D improves immune function.
"We have excellent mechanistic data to support this claim, including a number of observational studies and a few randomized controlled trials," Dr. Gallo said.
He coauthored a 2009 study concluding that vitamin D activates an enzyme on the surface of monocytes and keratinocytes, increasing the cells' pattern recognition and boosting their antimicrobial effect. "This enhances the immune barrier in injured skin," Dr. Gallo said (J. Clin. Invest. 2007;117:803-11).
Animal models also "show quite clearly that the extent of infection can be limited in an animal supplemented with vitamin D compared to a deficient one," he added. "There also seems to be a relative association between viral infections and upper respiratory infections, with the highest incidence occurring at the lowest levels of vitamin D on a seasonal basis. So maybe Schlitz did have an idea there. Therefore I’d say Myth No. 4 is plausible, but not yet clearly defined."
Dr. Gallo did not have any relevant financial disclosures. However, he is a member of the Institute of Medicine’s committee on Dietary Reference Intakes for Vitamin D and Calcium. The committee will release new recommendations for national daily requirements of vitamin D and calcium later this year.
CHICAGO – Dr. Richard L. Gallo put on his Myth Busters hat at a recent dermatology meeting to debunk – and in some cases uphold – some of the most popular ideas about vitamin D.
"This subject is nothing new," noted Dr. Gallo, recounting a bit of vitamin D yore. "In 1936, Schlitz beer urged customers to drink the beverage because it contained 100 units of vitamin D, and could ward off colds and flu. So even back then, they were on to something."
But, Dr. Gallo questioned, is the idea that vitamin D can strengthen the immune system a reality – or a myth? And how about other claims touted in the public press, that sunlight is the best source of vitamin D, that the vitamin strengthen bones and protects against cancer.
"Unfortunately, vitamin D information has become something of a shell game, with positions that overstate the strength of the evidence. As dermatologists, for example, we know the carcinogenic potential of sunlight, but there are now opposing groups that advocate health by increasing vitamin D through sun exposure."
Myth No. 1: Fifteen minutes per day of sunlight provides adequate amounts of vitamin D.
"In a test tube, ultraviolet B is the optimal spectrum for converting 25-hydroxy D into vitamin D in the human body," Dr. Gallo said. "But randomized studies on this vary in results."
One frequently cited study examined the issue in Denmark. "Northern latitudes are very useful for studies like this because of the high intensity of the sun during the summer, and the low intensity in winter," said Dr. Gallo, chief of dermatology and professor of medicine and pediatrics at the University of California San Diego. "In this study, the 25-hydroxy D in the population varied dramatically with change in sunlight exposure, and tended to lag about 1 month behind the sunlight levels."
But the study also found that 53% of the subjects who sought sun exposure were still suboptimal in their vitamin D levels. "So sun-seeking behavior in one of the most intense sun-exposed areas of the world is not sufficient to cover optimal vitamin D in a population." (Photochem. Photobiol. 2009;85:1,480-4).
A 2009 study looked at sunlight exposure and vitamin D in twins (PLoS One 2010 5(7):e11555 [doi: 10.1371/journal.pone.0011555]). More than 200 twins were evaluated for the seasonal impact of genetic factors on serum 25-hydroxy vitamin D concentrations. "This showed very wide distributions in levels during the different seasons, and concluded that more than 50% of the variation in summer levels was not due to sun or diet, but to genetic influences independent of skin pigment," Dr. Gallo said.
"So, Myth No. 1 – busted," he concluded.
Myth No. 2: Vitamin D improves bone health.
Prospective cohort studies such as the National Health and Nutrition Examination Survey show that hip fracture is reduced by more than one-third in patients with adequate vitamin D levels (more than 60 nmol/L). "However, we still have a lot to learn. Data from a recent 3-year study of 2,000 perimenopasual women concluded that if vitamin D were given as a single annual dose of 500,000 IU, the women had a 15% increased risk of falls and a 26% increase in the risk of fractures." (JAMA 2010;303:1815-22).
"As far as Myth No. 2 goes, I'd say it's true, but we don't understand everything yet."
Myth No. 3: Vitamin D protects against cancer.
"This has been quite a popular theme in the press for years now, but there are no great mechanistic explanations as to why it may be true," Dr Gallo said. "There are a number of randomized controlled trials, but the data are inconsistent."
A 2009 Agency for Healthcare Research and Quality review examined more than 170 studies and reviews for several health outcomes and vitamin D. "Only one study really showed a level of significance [for cancer reduction]. The others showed inconsistent data on cancer and some showed a slight trend toward an increased risk for colon cancer." (Evid. Rep. Technol. Assess. (Full Rep.) 2009;183:1-420).
"Myth No. 3 is still a plausible possibility, but no benefit has been clearly demonstrated."
Myth No. 4: Vitamin D improves immune function.
"We have excellent mechanistic data to support this claim, including a number of observational studies and a few randomized controlled trials," Dr. Gallo said.
He coauthored a 2009 study concluding that vitamin D activates an enzyme on the surface of monocytes and keratinocytes, increasing the cells' pattern recognition and boosting their antimicrobial effect. "This enhances the immune barrier in injured skin," Dr. Gallo said (J. Clin. Invest. 2007;117:803-11).
Animal models also "show quite clearly that the extent of infection can be limited in an animal supplemented with vitamin D compared to a deficient one," he added. "There also seems to be a relative association between viral infections and upper respiratory infections, with the highest incidence occurring at the lowest levels of vitamin D on a seasonal basis. So maybe Schlitz did have an idea there. Therefore I’d say Myth No. 4 is plausible, but not yet clearly defined."
Dr. Gallo did not have any relevant financial disclosures. However, he is a member of the Institute of Medicine’s committee on Dietary Reference Intakes for Vitamin D and Calcium. The committee will release new recommendations for national daily requirements of vitamin D and calcium later this year.
CHICAGO – Dr. Richard L. Gallo put on his Myth Busters hat at a recent dermatology meeting to debunk – and in some cases uphold – some of the most popular ideas about vitamin D.
"This subject is nothing new," noted Dr. Gallo, recounting a bit of vitamin D yore. "In 1936, Schlitz beer urged customers to drink the beverage because it contained 100 units of vitamin D, and could ward off colds and flu. So even back then, they were on to something."
But, Dr. Gallo questioned, is the idea that vitamin D can strengthen the immune system a reality – or a myth? And how about other claims touted in the public press, that sunlight is the best source of vitamin D, that the vitamin strengthen bones and protects against cancer.
"Unfortunately, vitamin D information has become something of a shell game, with positions that overstate the strength of the evidence. As dermatologists, for example, we know the carcinogenic potential of sunlight, but there are now opposing groups that advocate health by increasing vitamin D through sun exposure."
Myth No. 1: Fifteen minutes per day of sunlight provides adequate amounts of vitamin D.
"In a test tube, ultraviolet B is the optimal spectrum for converting 25-hydroxy D into vitamin D in the human body," Dr. Gallo said. "But randomized studies on this vary in results."
One frequently cited study examined the issue in Denmark. "Northern latitudes are very useful for studies like this because of the high intensity of the sun during the summer, and the low intensity in winter," said Dr. Gallo, chief of dermatology and professor of medicine and pediatrics at the University of California San Diego. "In this study, the 25-hydroxy D in the population varied dramatically with change in sunlight exposure, and tended to lag about 1 month behind the sunlight levels."
But the study also found that 53% of the subjects who sought sun exposure were still suboptimal in their vitamin D levels. "So sun-seeking behavior in one of the most intense sun-exposed areas of the world is not sufficient to cover optimal vitamin D in a population." (Photochem. Photobiol. 2009;85:1,480-4).
A 2009 study looked at sunlight exposure and vitamin D in twins (PLoS One 2010 5(7):e11555 [doi: 10.1371/journal.pone.0011555]). More than 200 twins were evaluated for the seasonal impact of genetic factors on serum 25-hydroxy vitamin D concentrations. "This showed very wide distributions in levels during the different seasons, and concluded that more than 50% of the variation in summer levels was not due to sun or diet, but to genetic influences independent of skin pigment," Dr. Gallo said.
"So, Myth No. 1 – busted," he concluded.
Myth No. 2: Vitamin D improves bone health.
Prospective cohort studies such as the National Health and Nutrition Examination Survey show that hip fracture is reduced by more than one-third in patients with adequate vitamin D levels (more than 60 nmol/L). "However, we still have a lot to learn. Data from a recent 3-year study of 2,000 perimenopasual women concluded that if vitamin D were given as a single annual dose of 500,000 IU, the women had a 15% increased risk of falls and a 26% increase in the risk of fractures." (JAMA 2010;303:1815-22).
"As far as Myth No. 2 goes, I'd say it's true, but we don't understand everything yet."
Myth No. 3: Vitamin D protects against cancer.
"This has been quite a popular theme in the press for years now, but there are no great mechanistic explanations as to why it may be true," Dr Gallo said. "There are a number of randomized controlled trials, but the data are inconsistent."
A 2009 Agency for Healthcare Research and Quality review examined more than 170 studies and reviews for several health outcomes and vitamin D. "Only one study really showed a level of significance [for cancer reduction]. The others showed inconsistent data on cancer and some showed a slight trend toward an increased risk for colon cancer." (Evid. Rep. Technol. Assess. (Full Rep.) 2009;183:1-420).
"Myth No. 3 is still a plausible possibility, but no benefit has been clearly demonstrated."
Myth No. 4: Vitamin D improves immune function.
"We have excellent mechanistic data to support this claim, including a number of observational studies and a few randomized controlled trials," Dr. Gallo said.
He coauthored a 2009 study concluding that vitamin D activates an enzyme on the surface of monocytes and keratinocytes, increasing the cells' pattern recognition and boosting their antimicrobial effect. "This enhances the immune barrier in injured skin," Dr. Gallo said (J. Clin. Invest. 2007;117:803-11).
Animal models also "show quite clearly that the extent of infection can be limited in an animal supplemented with vitamin D compared to a deficient one," he added. "There also seems to be a relative association between viral infections and upper respiratory infections, with the highest incidence occurring at the lowest levels of vitamin D on a seasonal basis. So maybe Schlitz did have an idea there. Therefore I’d say Myth No. 4 is plausible, but not yet clearly defined."
Dr. Gallo did not have any relevant financial disclosures. However, he is a member of the Institute of Medicine’s committee on Dietary Reference Intakes for Vitamin D and Calcium. The committee will release new recommendations for national daily requirements of vitamin D and calcium later this year.
Generic Biologics' Safety Questioned as Approval Process Stalls
The Biologics Price Competition and Innovation Act, quietly tucked into the health reform bill passed earlier this year, mandated the creation of an abbreviated approval process for follow-on biologic agents similar to the existing pathway for small-molecule drugs.
But 5 months later, the Food and Drug Administration has yet to work out just what that process will entail – and physicians have yet to be reassured that whatever emerges won’t cut corners on safety. And if the safety can be assured, the fruits of such a pathway could be life changing for some patients.
“Tremendously overpriced as they are, the available biologic drugs prevent so much human suffering and disability,” added Dr. Karen Kolba, who is a rheumatologist in solo private practice in Santa Maria, Calif.
“All of my patients would benefit. Those who cannot afford the copays because they are up to 30% of the drug cost would benefit. Those who have private insurance with low copays would benefit because their insurance companies will ultimately be paying out less to drug companies. And those with Medicare or Medicaid will benefit, as will all taxpayers, with lower drug costs.”
The Back Story
Unlike small-molecule generics, which gain approval through an abbreviated new drug application that does not require clinical trials, biologic drugs are vastly more complex – so complex, in fact, that their exact composition is not even known in some cases, according to the Merck Manual.
That is why, in the past, all new biologic drugs for which pharmaceutical companies seek approval in the United States have had to go through a complete new drug application process – including evaluation in animal studies and a full round of human clinical trials – before gaining approval.
The new Biologics Price Competition and Innovation Act (BPCIA) “aligns with the FDA’s long-standing policy of permitting appropriate reliance on what is already known about a drug, thereby saving time and resources and avoiding unnecessary duplication of human or animal testing,” according to a statement by the FDA.
And the new law provides guidance on the intellectual property side of the issue. It grants 12 years of patent exclusivity for an original biologic drug, plus a hold on applications for generics until 4 years after the original’s approval. And once approved, the law states, the first generic will itself enjoy a 1-year period of exclusivity.
The Safety Debate
However, the BPCIA does not offer very much guidance on what the abbreviated approval process will entail, said Merrill Matthews, Ph.D., a medical ethicist and a resident scholar with the Institute for Policy Innovation, Lewisville, Tex., a conservative advocacy group that seeks “lower taxes, fewer regulations, and a smaller, less-intrusive government.” He also is the director of the Council for Affordable Health Insurance, a D.C.-based organization promoting free-market health insurance reforms.
The BPCIA “gives the FDA an awful lot of latitude and flexibility in the process of approving biosimilars, and though it requires biosimilars to demonstrate ‘similarity’ with their reference products, approval can be done without clinical trials,” he said at a July 19 press conference on Capitol Hill.
Indeed, the law reads: “An application submitted under this subsection shall include information demonstrating that ... the biological product is biosimilar to a reference product based upon data derived from ... analytical studies that demonstrate that the biological product is highly similar to the reference product, notwithstanding minor differences in clinically inactive components.”
Gordon Johnston, vice president of regulatory science at the Generic Pharmaceutical Association, said he does not find the law to be clear. “The legislation could have been clearer, in our view, in ... defining terms such as ‘highly similar’ and ‘no clinically meaningful difference,’ ” he said in an interview. “For this reason, the process now under way of writing the legislation into regulations is critically important,” he added.
Dr. Kolba also pointed out that the new law would have done well to include at least some requirement that new generic products be entered in a registry to collect safety data. “This would be a much less expensive alternative to years of clinical trials, but ultimately assures us all that they are as safe as the branded products,” she said.?
Dr. Craig Kessler, professor of medicine and pathology at Georgetown University, Washington, agreed. “As a physician, I’m very much in favor of having biosimilars in the marketplace,” he said. “I think we owe it to society to try to reduce the cost of care.”
Nevertheless, “I don’t want to sacrifice patient safety for the decrease in the cost of care, because in the long run, the cost of care will actually be increased if we make the wrong decision.”
Added Rep. Michael C. Burgess (R-Tex.), a physician and also the chairman of the congressional Health Care Caucus, at the July press conference: “Can you really go through an abbreviated process with a drug that is just simply similar to the complex biologic that has already been introduced?
“My concern as a physician, first, always comes back to safety,” he said.
The Next Steps
To resolve these terms, and to figure out a pathway for biogeneric approval, the FDA has formed the Biosimilar Implementation Committee and placed at its head Dr. Janet Woodcock, who is also director of the Center for Drug Evaluation and Research. The new center will hold public meetings to solicit comment from stakeholders, experts, innovators, patients, and the public on some of the concerns surrounding biosimilar approval, according to Dr. Woodcock in a recent press release.
Mr. Johnston said that he expects a meeting to be held sometime by the end of 2010. However, the details remain vague. FDA spokesperson Karen Mahoney could not confirm that any meeting had been scheduled, and would not give any information on when physicians and patients could expect to see the first approvals of generic biologics.
“There are so many factors that will impact when biosimilar products will enter the market,” added Ms. Mahoney. “Therefore,?it is not reasonable to speculate.”
Added Rep. Burgess: “This is a difficult concept, and it does involve a lot of moving parts, and a lot of different contingencies. “For your average member of Congress – or even for your member of Congress with some background in health – it does become difficult to think about these things,” he added.
Dr. Kolba, meanwhile, offered some ideas about how to move forward – without generics.
“If the companies currently manufacturing biologic drugs and selling them at huge profits (having made back their research and development costs years ago) are truly concerned about the potential harm of generic biologics [to their market share,] they can simply decrease their prices to costs plus 10%. This would decrease the overall cost of the drugs by about 65%,” she said.
“At the new prices, generic drug makers will no longer be willing to enter the market, there will be no expensive lawsuits or advertising to pay for, and the 1% of the population with rheumatoid arthritis and other TNF [tumor necrosis factor]-driven diseases will be assured of safe and effective drugs.”
Dr. Kolba disclosed being a past consultant or speaker for the pharmaceutical companies Abbott, Amgen, Bristol-Myers Squibb, Genentech, and UCB. She is currently a principal investigator in clinical trials sponsored by Abbott, Amgen, Lilly, Pfizer, Sanofi-Aventis, and UCB. Dr. Craig Kessler has also disclosed consulting for and receiving research support from pharmaceutical companies including Amgen, Eisai, GlaxoSmithKline, and Sanofi-Aventis.
To view an online version of the July 19 press conference featuring Dr. Kessler, Dr. Matthews, and Congressman Burgess, visit http://health.burgess.house.gov/Blog/?postid=198224.
To view the entire text of the Biologics Price Competition and Innovation Act of 2009, visit www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/UCM216146.pdf.
The Biologics Price Competition and Innovation Act, quietly tucked into the health reform bill passed earlier this year, mandated the creation of an abbreviated approval process for follow-on biologic agents similar to the existing pathway for small-molecule drugs.
But 5 months later, the Food and Drug Administration has yet to work out just what that process will entail – and physicians have yet to be reassured that whatever emerges won’t cut corners on safety. And if the safety can be assured, the fruits of such a pathway could be life changing for some patients.
“Tremendously overpriced as they are, the available biologic drugs prevent so much human suffering and disability,” added Dr. Karen Kolba, who is a rheumatologist in solo private practice in Santa Maria, Calif.
“All of my patients would benefit. Those who cannot afford the copays because they are up to 30% of the drug cost would benefit. Those who have private insurance with low copays would benefit because their insurance companies will ultimately be paying out less to drug companies. And those with Medicare or Medicaid will benefit, as will all taxpayers, with lower drug costs.”
The Back Story
Unlike small-molecule generics, which gain approval through an abbreviated new drug application that does not require clinical trials, biologic drugs are vastly more complex – so complex, in fact, that their exact composition is not even known in some cases, according to the Merck Manual.
That is why, in the past, all new biologic drugs for which pharmaceutical companies seek approval in the United States have had to go through a complete new drug application process – including evaluation in animal studies and a full round of human clinical trials – before gaining approval.
The new Biologics Price Competition and Innovation Act (BPCIA) “aligns with the FDA’s long-standing policy of permitting appropriate reliance on what is already known about a drug, thereby saving time and resources and avoiding unnecessary duplication of human or animal testing,” according to a statement by the FDA.
And the new law provides guidance on the intellectual property side of the issue. It grants 12 years of patent exclusivity for an original biologic drug, plus a hold on applications for generics until 4 years after the original’s approval. And once approved, the law states, the first generic will itself enjoy a 1-year period of exclusivity.
The Safety Debate
However, the BPCIA does not offer very much guidance on what the abbreviated approval process will entail, said Merrill Matthews, Ph.D., a medical ethicist and a resident scholar with the Institute for Policy Innovation, Lewisville, Tex., a conservative advocacy group that seeks “lower taxes, fewer regulations, and a smaller, less-intrusive government.” He also is the director of the Council for Affordable Health Insurance, a D.C.-based organization promoting free-market health insurance reforms.
The BPCIA “gives the FDA an awful lot of latitude and flexibility in the process of approving biosimilars, and though it requires biosimilars to demonstrate ‘similarity’ with their reference products, approval can be done without clinical trials,” he said at a July 19 press conference on Capitol Hill.
Indeed, the law reads: “An application submitted under this subsection shall include information demonstrating that ... the biological product is biosimilar to a reference product based upon data derived from ... analytical studies that demonstrate that the biological product is highly similar to the reference product, notwithstanding minor differences in clinically inactive components.”
Gordon Johnston, vice president of regulatory science at the Generic Pharmaceutical Association, said he does not find the law to be clear. “The legislation could have been clearer, in our view, in ... defining terms such as ‘highly similar’ and ‘no clinically meaningful difference,’ ” he said in an interview. “For this reason, the process now under way of writing the legislation into regulations is critically important,” he added.
Dr. Kolba also pointed out that the new law would have done well to include at least some requirement that new generic products be entered in a registry to collect safety data. “This would be a much less expensive alternative to years of clinical trials, but ultimately assures us all that they are as safe as the branded products,” she said.?
Dr. Craig Kessler, professor of medicine and pathology at Georgetown University, Washington, agreed. “As a physician, I’m very much in favor of having biosimilars in the marketplace,” he said. “I think we owe it to society to try to reduce the cost of care.”
Nevertheless, “I don’t want to sacrifice patient safety for the decrease in the cost of care, because in the long run, the cost of care will actually be increased if we make the wrong decision.”
Added Rep. Michael C. Burgess (R-Tex.), a physician and also the chairman of the congressional Health Care Caucus, at the July press conference: “Can you really go through an abbreviated process with a drug that is just simply similar to the complex biologic that has already been introduced?
“My concern as a physician, first, always comes back to safety,” he said.
The Next Steps
To resolve these terms, and to figure out a pathway for biogeneric approval, the FDA has formed the Biosimilar Implementation Committee and placed at its head Dr. Janet Woodcock, who is also director of the Center for Drug Evaluation and Research. The new center will hold public meetings to solicit comment from stakeholders, experts, innovators, patients, and the public on some of the concerns surrounding biosimilar approval, according to Dr. Woodcock in a recent press release.
Mr. Johnston said that he expects a meeting to be held sometime by the end of 2010. However, the details remain vague. FDA spokesperson Karen Mahoney could not confirm that any meeting had been scheduled, and would not give any information on when physicians and patients could expect to see the first approvals of generic biologics.
“There are so many factors that will impact when biosimilar products will enter the market,” added Ms. Mahoney. “Therefore,?it is not reasonable to speculate.”
Added Rep. Burgess: “This is a difficult concept, and it does involve a lot of moving parts, and a lot of different contingencies. “For your average member of Congress – or even for your member of Congress with some background in health – it does become difficult to think about these things,” he added.
Dr. Kolba, meanwhile, offered some ideas about how to move forward – without generics.
“If the companies currently manufacturing biologic drugs and selling them at huge profits (having made back their research and development costs years ago) are truly concerned about the potential harm of generic biologics [to their market share,] they can simply decrease their prices to costs plus 10%. This would decrease the overall cost of the drugs by about 65%,” she said.
“At the new prices, generic drug makers will no longer be willing to enter the market, there will be no expensive lawsuits or advertising to pay for, and the 1% of the population with rheumatoid arthritis and other TNF [tumor necrosis factor]-driven diseases will be assured of safe and effective drugs.”
Dr. Kolba disclosed being a past consultant or speaker for the pharmaceutical companies Abbott, Amgen, Bristol-Myers Squibb, Genentech, and UCB. She is currently a principal investigator in clinical trials sponsored by Abbott, Amgen, Lilly, Pfizer, Sanofi-Aventis, and UCB. Dr. Craig Kessler has also disclosed consulting for and receiving research support from pharmaceutical companies including Amgen, Eisai, GlaxoSmithKline, and Sanofi-Aventis.
To view an online version of the July 19 press conference featuring Dr. Kessler, Dr. Matthews, and Congressman Burgess, visit http://health.burgess.house.gov/Blog/?postid=198224.
To view the entire text of the Biologics Price Competition and Innovation Act of 2009, visit www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/UCM216146.pdf.
The Biologics Price Competition and Innovation Act, quietly tucked into the health reform bill passed earlier this year, mandated the creation of an abbreviated approval process for follow-on biologic agents similar to the existing pathway for small-molecule drugs.
But 5 months later, the Food and Drug Administration has yet to work out just what that process will entail – and physicians have yet to be reassured that whatever emerges won’t cut corners on safety. And if the safety can be assured, the fruits of such a pathway could be life changing for some patients.
“Tremendously overpriced as they are, the available biologic drugs prevent so much human suffering and disability,” added Dr. Karen Kolba, who is a rheumatologist in solo private practice in Santa Maria, Calif.
“All of my patients would benefit. Those who cannot afford the copays because they are up to 30% of the drug cost would benefit. Those who have private insurance with low copays would benefit because their insurance companies will ultimately be paying out less to drug companies. And those with Medicare or Medicaid will benefit, as will all taxpayers, with lower drug costs.”
The Back Story
Unlike small-molecule generics, which gain approval through an abbreviated new drug application that does not require clinical trials, biologic drugs are vastly more complex – so complex, in fact, that their exact composition is not even known in some cases, according to the Merck Manual.
That is why, in the past, all new biologic drugs for which pharmaceutical companies seek approval in the United States have had to go through a complete new drug application process – including evaluation in animal studies and a full round of human clinical trials – before gaining approval.
The new Biologics Price Competition and Innovation Act (BPCIA) “aligns with the FDA’s long-standing policy of permitting appropriate reliance on what is already known about a drug, thereby saving time and resources and avoiding unnecessary duplication of human or animal testing,” according to a statement by the FDA.
And the new law provides guidance on the intellectual property side of the issue. It grants 12 years of patent exclusivity for an original biologic drug, plus a hold on applications for generics until 4 years after the original’s approval. And once approved, the law states, the first generic will itself enjoy a 1-year period of exclusivity.
The Safety Debate
However, the BPCIA does not offer very much guidance on what the abbreviated approval process will entail, said Merrill Matthews, Ph.D., a medical ethicist and a resident scholar with the Institute for Policy Innovation, Lewisville, Tex., a conservative advocacy group that seeks “lower taxes, fewer regulations, and a smaller, less-intrusive government.” He also is the director of the Council for Affordable Health Insurance, a D.C.-based organization promoting free-market health insurance reforms.
The BPCIA “gives the FDA an awful lot of latitude and flexibility in the process of approving biosimilars, and though it requires biosimilars to demonstrate ‘similarity’ with their reference products, approval can be done without clinical trials,” he said at a July 19 press conference on Capitol Hill.
Indeed, the law reads: “An application submitted under this subsection shall include information demonstrating that ... the biological product is biosimilar to a reference product based upon data derived from ... analytical studies that demonstrate that the biological product is highly similar to the reference product, notwithstanding minor differences in clinically inactive components.”
Gordon Johnston, vice president of regulatory science at the Generic Pharmaceutical Association, said he does not find the law to be clear. “The legislation could have been clearer, in our view, in ... defining terms such as ‘highly similar’ and ‘no clinically meaningful difference,’ ” he said in an interview. “For this reason, the process now under way of writing the legislation into regulations is critically important,” he added.
Dr. Kolba also pointed out that the new law would have done well to include at least some requirement that new generic products be entered in a registry to collect safety data. “This would be a much less expensive alternative to years of clinical trials, but ultimately assures us all that they are as safe as the branded products,” she said.?
Dr. Craig Kessler, professor of medicine and pathology at Georgetown University, Washington, agreed. “As a physician, I’m very much in favor of having biosimilars in the marketplace,” he said. “I think we owe it to society to try to reduce the cost of care.”
Nevertheless, “I don’t want to sacrifice patient safety for the decrease in the cost of care, because in the long run, the cost of care will actually be increased if we make the wrong decision.”
Added Rep. Michael C. Burgess (R-Tex.), a physician and also the chairman of the congressional Health Care Caucus, at the July press conference: “Can you really go through an abbreviated process with a drug that is just simply similar to the complex biologic that has already been introduced?
“My concern as a physician, first, always comes back to safety,” he said.
The Next Steps
To resolve these terms, and to figure out a pathway for biogeneric approval, the FDA has formed the Biosimilar Implementation Committee and placed at its head Dr. Janet Woodcock, who is also director of the Center for Drug Evaluation and Research. The new center will hold public meetings to solicit comment from stakeholders, experts, innovators, patients, and the public on some of the concerns surrounding biosimilar approval, according to Dr. Woodcock in a recent press release.
Mr. Johnston said that he expects a meeting to be held sometime by the end of 2010. However, the details remain vague. FDA spokesperson Karen Mahoney could not confirm that any meeting had been scheduled, and would not give any information on when physicians and patients could expect to see the first approvals of generic biologics.
“There are so many factors that will impact when biosimilar products will enter the market,” added Ms. Mahoney. “Therefore,?it is not reasonable to speculate.”
Added Rep. Burgess: “This is a difficult concept, and it does involve a lot of moving parts, and a lot of different contingencies. “For your average member of Congress – or even for your member of Congress with some background in health – it does become difficult to think about these things,” he added.
Dr. Kolba, meanwhile, offered some ideas about how to move forward – without generics.
“If the companies currently manufacturing biologic drugs and selling them at huge profits (having made back their research and development costs years ago) are truly concerned about the potential harm of generic biologics [to their market share,] they can simply decrease their prices to costs plus 10%. This would decrease the overall cost of the drugs by about 65%,” she said.
“At the new prices, generic drug makers will no longer be willing to enter the market, there will be no expensive lawsuits or advertising to pay for, and the 1% of the population with rheumatoid arthritis and other TNF [tumor necrosis factor]-driven diseases will be assured of safe and effective drugs.”
Dr. Kolba disclosed being a past consultant or speaker for the pharmaceutical companies Abbott, Amgen, Bristol-Myers Squibb, Genentech, and UCB. She is currently a principal investigator in clinical trials sponsored by Abbott, Amgen, Lilly, Pfizer, Sanofi-Aventis, and UCB. Dr. Craig Kessler has also disclosed consulting for and receiving research support from pharmaceutical companies including Amgen, Eisai, GlaxoSmithKline, and Sanofi-Aventis.
To view an online version of the July 19 press conference featuring Dr. Kessler, Dr. Matthews, and Congressman Burgess, visit http://health.burgess.house.gov/Blog/?postid=198224.
To view the entire text of the Biologics Price Competition and Innovation Act of 2009, visit www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/UCM216146.pdf.