User login
Telehealth apps in ObGyn practice
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.
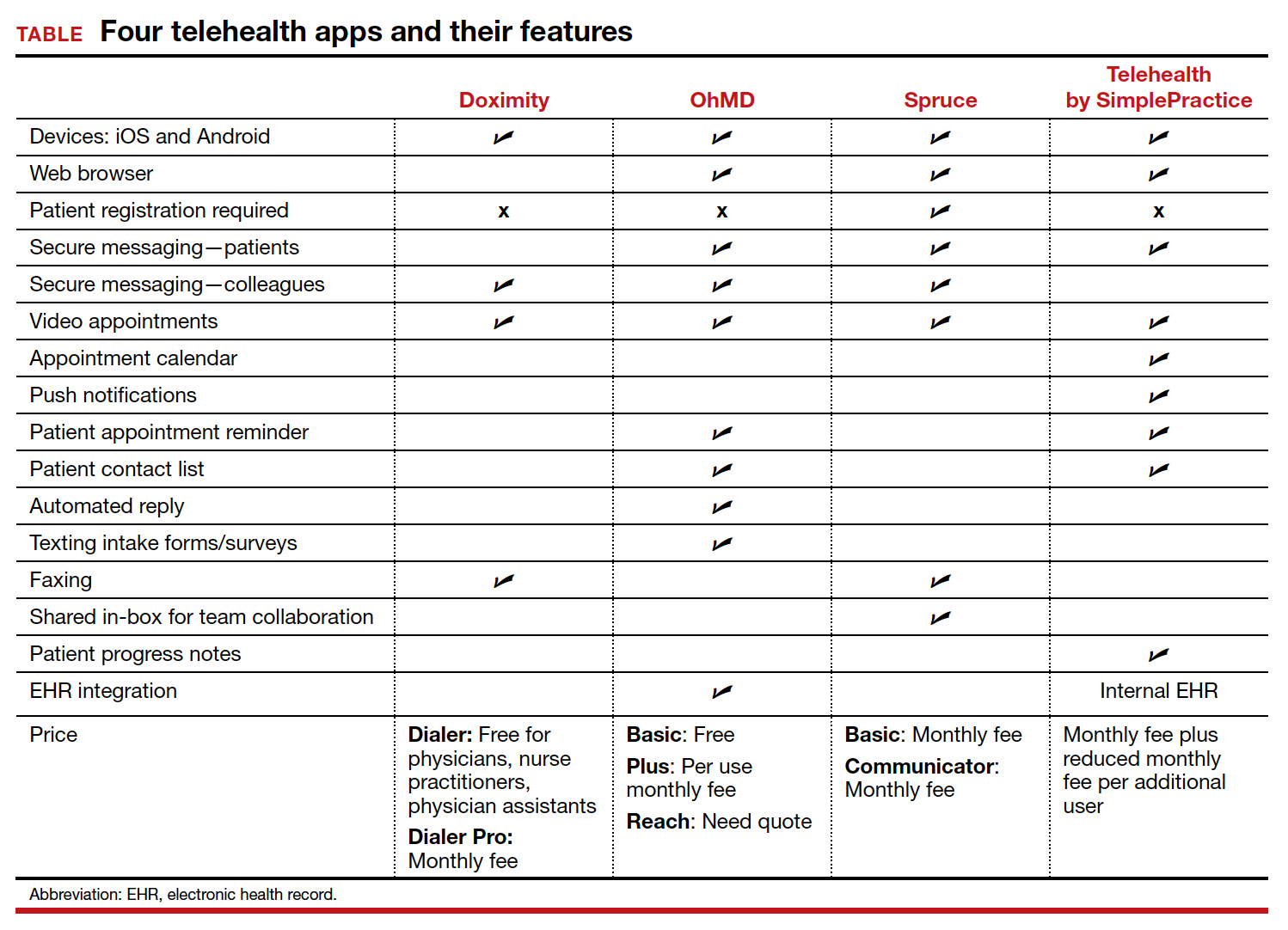
These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
Free now to speak, nine oncologists spill the beans over firing
Last year, nine oncologists filed a lawsuit against the Anne Arundel Medical Center (AAMC), in Annapolis, Md., alleging that the hospital had fired them and had refused to allow them privileges to see their patients.
The oncologists said that the hospital chose profit over the needs of cancer patients, as it slashed oncology care services to cut costs.
The hospital denied any wrongdoing and alleged that the oncologists were not fired but that they had quit because they had been offered a more profitable opportunity.
At that time, the oncologists were not free to respond because of the ongoing litigation. But now that the lawsuit is over and the dust has settled, they are free to speak, and they contacted this news organization to tell their side of the story.
AAMC is a private, not-for-profit corporation that operates a large acute care hospital in Annapolis. It is affiliated with Luminis Health, the parent company of the medical center. Until October 23, 2020, the nine oncologists were employed by the AA Physician Group.
The doctors are Jason Taksey, MD, Benjamin Bridges, MD, Ravin Garg, MD, Adam Goldrich, MD, Carol Tweed, MD, Peter Graze, MD, Stuart Selonick, MD, David Weng, MD, and Jeanine Werner, MD.
They are all “highly respected, board certified oncologists and hematologists, with regional and, for some, national reputations in their medical specialty. The oncologists have had privileges at AAMC for many years and their capability as physicians is unquestioned,” according to the court filing made on behalf of the oncologists.
“Most of us have been in this town for decades,” said Dr. Tweed, who served as the unofficial spokesperson for the group. “Some of us are faculty members at Johns Hopkins, and this hospital’s oncology service was historically defined by our group.”
AAMC has a good reputation for providing high-quality medicine, “which is what brought many of us there in the first place,” Dr. Tweed said in an interview.
Triggered by cost cutting
The situation began when the hospital began cutting services to curtail costs, which directly affected the delivery of oncology care, Dr. Tweed explained. “They were also creating a very toxic and difficult interpersonal work environment, and that made it difficult to do patient care,” she said. “We would go to them and let them know that we were having difficulty delivering optimal patient care because we didn’t have enough staff or the resources we needed for safety — and it got to the point where we were being ignored and our input was no longer welcome.”
Dr. Tweed explained that the administrators announced which patient-care services would be cut without asking for their input as to the safety of those decisions. “Perhaps the most notorious was when they shut down the oncology lab,” she said. “That lab to an oncologist/hematologist is like a scalpel to a surgeon. I need lab results immediately — I need to know if I can give chemotherapy right now, or do I need to hold a dose. The lab is intrinsic to oncology care anywhere.”
There was a continuing cascade of events, and the oncology group mulled over some ideas as to how to provide optimal patient care in this increasingly difficult environment. The decision they reached was to discuss running their own practice with the hospital administrators as a means of making up for the gaps that they were now having to contend with. “As physicians, we do a lot of non-billable work, such as patient education, nighttime rounds for our cancer patients, and so on, and we told them that we would continue doing that,” said Dr. Tweed. “They said that they would talk to us, but they didn’t.”
Within a week of sending their proposal for setting up their own practice, all nine physicians were fired. “Instead of arranging a discussion, we received termination letters,” she explained. “We were terminated without cause.”
As physicians, Dr. Tweed explained that they were by contract obligated to arbitrate. It dragged on for weeks and months, to the tune of hundreds of thousands of dollars in legal fees.
“The only thing we wanted was to be able to practice in this town,” said Dr. Tweed. “And what is important to know is that it was never for money, and that was never our motivation for wanting to form our own practice.”
Dr. Tweed was referring to the hospital’s allegations that the oncologists had left their employment for monetary gain. A statement given to this news organization by the Luminis Health Anne Arundel Medical Center at the time stated that “this dispute started after nine oncologists left their employment to join a for-profit organization. We tried repeatedly to remain aligned with them.”
The oncologists had resigned during the height of the coronavirus pandemic to “pursue lucrative contracts” with a “major pharmaceutical distribution,” according to Todd M. Reinecker, attorney for Luminis Health, as reported by the Capital Gazette (this news organization reached out to Mr. Reinecker at that time but did not receive a response).
This was not the case, Dr. Tweed emphasized. “We took a great financial risk in doing this for patient care. It was pretty disgusting that was in print from the hospital’s lawyer.”
“The doctors anticipated Luminis Health would be unable to recruit new physicians and be forced to continue to use their services,” Mr. Reinecker maintained.
In fact, the medical center hired seven new oncologists to replace them.
Noncompete covenant
In filing their lawsuit, the nine oncologists put before the arbitrator the issue of the enforceability of the noncompete provision in their employment agreement, which prohibited the oncologists from working in the geographic area that includes the hospital. Their position was that the agreement was overly broad and thus unenforceable.
“We sign noncompete restrictive covenant contracts and we’re told that they are nonenforceable, and that’s the general discourse,” said Dr. Tweed. “Some states don’t even allow them. Well, we found out that they are very enforceable.”
The arbitrator eventually determined that three of the oncologists, including Dr. Tweed, had enforceable noncompete contracts.
“During the year or so while this was all going on, I would say that 90% of my patients wanted to stay with me,” said Dr. Tweed. “Patients were looking all over the place for us because, in many cases, the hospital did not tell them where to find us. In fact, they told us that we couldn’t contact the patients — they said it was ‘solicitation of a patient.’ “
In addition, the hospital continued to put more restrictions on the doctors. Six of the nine oncologists were able to continue practicing in Annapolis, and the remaining three will be able to join them in October 2022 when their noncompete contracts expire.
Now that the hospital has seen that there was a new oncology practice in town, Dr. Tweed noted, they changed their bylaws, and they now forbid hospital privileges to every physician in that group.
“The new bylaws do not restrict all private oncologists, just specifically our group, which prevents us from being able to do rounds in the hospital,” said Dr. Tweed. “If I want to see any of my patients, I have to get a visitor badge.”
Dr. Tweed contends that this move was purely for financial and business reasons to keep the oncologists from their patients. This is the primary hospital where their patients would be admitted if they need hospital care. AAMC is the only hospital within a 15-mile radius, and it serves as the regional hospital for the greater Annapolis area and for many Eastern Shore communities, whose hospitals do not offer various specialty services, such as oncology care.
“This was done purely because they were finance focused and not patient care focused,” Dr. Tweed emphasized. “We basically had to bargain with the hospital to let us even transfuse our patients.”
Telemedicine added to the mix
Yet another restriction that surfaced during the arbitration involved telemedicine. Dr. Tweed explained that as soon as the hospital realized that the three oncologists planned to stay in town and that their patients wanted to continue receiving care with them, they put telemedicine on the chopping block.
As if the restrictions and removal of hospital privileges wasn’t enough, the hospital decided to go after telemedicine during arbitration, Dr. Tweed said. If patients lived in any of the restricted ZIP codes, they were forbidden to conduct virtual visits with them.
“This isn’t ethical, but they tried to do everything to keep us from seeing our patients,” she said. “This is patient choice, but they were telling patients that if you live in any of these ZIP codes, you cannot do telemedicine if you choose Carol Tweed as your doctor,” Dr. Tweed said.
Of course, a patient isn’t bound by the arbitration and can see any doctor, but Dr. Tweed explained that the hospital threatened to come after her with a lawsuit.
One of the other physicians, Stuart Selonick, MD, said in an interview that he wasn’t quite sure how the idea of prohibiting telemedicine even came up. “There is little precedence for telemedicine in the U.S.,” he said. “They’ve extended the restrictions to telemedicine, and this is a new legal boundary, and it was new to the judge. But they made it part of the definition of the restrictive covenant. But to fight it would mean another lawsuit,” he added.
A separate lawsuit had previously been filed in an effort to regain hospital privileges, but the decision was made not to continue, owing to the amount of litigation it would involve.
“We can’t spend a lifetime and millions on another legal battle,” said Dr. Tweed. “We don’t have the corporate legal pool that the hospital has, and they know it.”
Patients have written endless letters supporting the doctors, Dr. Tweed said, but to no avail, as the hospital did not change course.
Litigation is now completed, and in about 9 months, the remaining three physicians will be able to rejoin their colleagues and put this behind them as best they can.
“The hospital knows that they harmed patient care for financial gain -- that’s the tagline,” said Tweed.
Approached for a response, Justin McLeod, spokesperson for Luminis Health, said that they are “pleased with the outcome of the case and the resolution agreed to by both sides. This agreement ensures patient access and continuity of care for patients with cancer. These providers have access to their patients’ electronic medical records, can order outpatient services, and attend quarterly cancer committee meetings with other providers.
“Our focus is the future of cancer care for our community. Luminis Health Anne Arundel Medical Center is committed to providing patients with high quality, comprehensive cancer care that is accessible to all,” he added.
A version of this article first appeared on Medscape.com.
Last year, nine oncologists filed a lawsuit against the Anne Arundel Medical Center (AAMC), in Annapolis, Md., alleging that the hospital had fired them and had refused to allow them privileges to see their patients.
The oncologists said that the hospital chose profit over the needs of cancer patients, as it slashed oncology care services to cut costs.
The hospital denied any wrongdoing and alleged that the oncologists were not fired but that they had quit because they had been offered a more profitable opportunity.
At that time, the oncologists were not free to respond because of the ongoing litigation. But now that the lawsuit is over and the dust has settled, they are free to speak, and they contacted this news organization to tell their side of the story.
AAMC is a private, not-for-profit corporation that operates a large acute care hospital in Annapolis. It is affiliated with Luminis Health, the parent company of the medical center. Until October 23, 2020, the nine oncologists were employed by the AA Physician Group.
The doctors are Jason Taksey, MD, Benjamin Bridges, MD, Ravin Garg, MD, Adam Goldrich, MD, Carol Tweed, MD, Peter Graze, MD, Stuart Selonick, MD, David Weng, MD, and Jeanine Werner, MD.
They are all “highly respected, board certified oncologists and hematologists, with regional and, for some, national reputations in their medical specialty. The oncologists have had privileges at AAMC for many years and their capability as physicians is unquestioned,” according to the court filing made on behalf of the oncologists.
“Most of us have been in this town for decades,” said Dr. Tweed, who served as the unofficial spokesperson for the group. “Some of us are faculty members at Johns Hopkins, and this hospital’s oncology service was historically defined by our group.”
AAMC has a good reputation for providing high-quality medicine, “which is what brought many of us there in the first place,” Dr. Tweed said in an interview.
Triggered by cost cutting
The situation began when the hospital began cutting services to curtail costs, which directly affected the delivery of oncology care, Dr. Tweed explained. “They were also creating a very toxic and difficult interpersonal work environment, and that made it difficult to do patient care,” she said. “We would go to them and let them know that we were having difficulty delivering optimal patient care because we didn’t have enough staff or the resources we needed for safety — and it got to the point where we were being ignored and our input was no longer welcome.”
Dr. Tweed explained that the administrators announced which patient-care services would be cut without asking for their input as to the safety of those decisions. “Perhaps the most notorious was when they shut down the oncology lab,” she said. “That lab to an oncologist/hematologist is like a scalpel to a surgeon. I need lab results immediately — I need to know if I can give chemotherapy right now, or do I need to hold a dose. The lab is intrinsic to oncology care anywhere.”
There was a continuing cascade of events, and the oncology group mulled over some ideas as to how to provide optimal patient care in this increasingly difficult environment. The decision they reached was to discuss running their own practice with the hospital administrators as a means of making up for the gaps that they were now having to contend with. “As physicians, we do a lot of non-billable work, such as patient education, nighttime rounds for our cancer patients, and so on, and we told them that we would continue doing that,” said Dr. Tweed. “They said that they would talk to us, but they didn’t.”
Within a week of sending their proposal for setting up their own practice, all nine physicians were fired. “Instead of arranging a discussion, we received termination letters,” she explained. “We were terminated without cause.”
As physicians, Dr. Tweed explained that they were by contract obligated to arbitrate. It dragged on for weeks and months, to the tune of hundreds of thousands of dollars in legal fees.
“The only thing we wanted was to be able to practice in this town,” said Dr. Tweed. “And what is important to know is that it was never for money, and that was never our motivation for wanting to form our own practice.”
Dr. Tweed was referring to the hospital’s allegations that the oncologists had left their employment for monetary gain. A statement given to this news organization by the Luminis Health Anne Arundel Medical Center at the time stated that “this dispute started after nine oncologists left their employment to join a for-profit organization. We tried repeatedly to remain aligned with them.”
The oncologists had resigned during the height of the coronavirus pandemic to “pursue lucrative contracts” with a “major pharmaceutical distribution,” according to Todd M. Reinecker, attorney for Luminis Health, as reported by the Capital Gazette (this news organization reached out to Mr. Reinecker at that time but did not receive a response).
This was not the case, Dr. Tweed emphasized. “We took a great financial risk in doing this for patient care. It was pretty disgusting that was in print from the hospital’s lawyer.”
“The doctors anticipated Luminis Health would be unable to recruit new physicians and be forced to continue to use their services,” Mr. Reinecker maintained.
In fact, the medical center hired seven new oncologists to replace them.
Noncompete covenant
In filing their lawsuit, the nine oncologists put before the arbitrator the issue of the enforceability of the noncompete provision in their employment agreement, which prohibited the oncologists from working in the geographic area that includes the hospital. Their position was that the agreement was overly broad and thus unenforceable.
“We sign noncompete restrictive covenant contracts and we’re told that they are nonenforceable, and that’s the general discourse,” said Dr. Tweed. “Some states don’t even allow them. Well, we found out that they are very enforceable.”
The arbitrator eventually determined that three of the oncologists, including Dr. Tweed, had enforceable noncompete contracts.
“During the year or so while this was all going on, I would say that 90% of my patients wanted to stay with me,” said Dr. Tweed. “Patients were looking all over the place for us because, in many cases, the hospital did not tell them where to find us. In fact, they told us that we couldn’t contact the patients — they said it was ‘solicitation of a patient.’ “
In addition, the hospital continued to put more restrictions on the doctors. Six of the nine oncologists were able to continue practicing in Annapolis, and the remaining three will be able to join them in October 2022 when their noncompete contracts expire.
Now that the hospital has seen that there was a new oncology practice in town, Dr. Tweed noted, they changed their bylaws, and they now forbid hospital privileges to every physician in that group.
“The new bylaws do not restrict all private oncologists, just specifically our group, which prevents us from being able to do rounds in the hospital,” said Dr. Tweed. “If I want to see any of my patients, I have to get a visitor badge.”
Dr. Tweed contends that this move was purely for financial and business reasons to keep the oncologists from their patients. This is the primary hospital where their patients would be admitted if they need hospital care. AAMC is the only hospital within a 15-mile radius, and it serves as the regional hospital for the greater Annapolis area and for many Eastern Shore communities, whose hospitals do not offer various specialty services, such as oncology care.
“This was done purely because they were finance focused and not patient care focused,” Dr. Tweed emphasized. “We basically had to bargain with the hospital to let us even transfuse our patients.”
Telemedicine added to the mix
Yet another restriction that surfaced during the arbitration involved telemedicine. Dr. Tweed explained that as soon as the hospital realized that the three oncologists planned to stay in town and that their patients wanted to continue receiving care with them, they put telemedicine on the chopping block.
As if the restrictions and removal of hospital privileges wasn’t enough, the hospital decided to go after telemedicine during arbitration, Dr. Tweed said. If patients lived in any of the restricted ZIP codes, they were forbidden to conduct virtual visits with them.
“This isn’t ethical, but they tried to do everything to keep us from seeing our patients,” she said. “This is patient choice, but they were telling patients that if you live in any of these ZIP codes, you cannot do telemedicine if you choose Carol Tweed as your doctor,” Dr. Tweed said.
Of course, a patient isn’t bound by the arbitration and can see any doctor, but Dr. Tweed explained that the hospital threatened to come after her with a lawsuit.
One of the other physicians, Stuart Selonick, MD, said in an interview that he wasn’t quite sure how the idea of prohibiting telemedicine even came up. “There is little precedence for telemedicine in the U.S.,” he said. “They’ve extended the restrictions to telemedicine, and this is a new legal boundary, and it was new to the judge. But they made it part of the definition of the restrictive covenant. But to fight it would mean another lawsuit,” he added.
A separate lawsuit had previously been filed in an effort to regain hospital privileges, but the decision was made not to continue, owing to the amount of litigation it would involve.
“We can’t spend a lifetime and millions on another legal battle,” said Dr. Tweed. “We don’t have the corporate legal pool that the hospital has, and they know it.”
Patients have written endless letters supporting the doctors, Dr. Tweed said, but to no avail, as the hospital did not change course.
Litigation is now completed, and in about 9 months, the remaining three physicians will be able to rejoin their colleagues and put this behind them as best they can.
“The hospital knows that they harmed patient care for financial gain -- that’s the tagline,” said Tweed.
Approached for a response, Justin McLeod, spokesperson for Luminis Health, said that they are “pleased with the outcome of the case and the resolution agreed to by both sides. This agreement ensures patient access and continuity of care for patients with cancer. These providers have access to their patients’ electronic medical records, can order outpatient services, and attend quarterly cancer committee meetings with other providers.
“Our focus is the future of cancer care for our community. Luminis Health Anne Arundel Medical Center is committed to providing patients with high quality, comprehensive cancer care that is accessible to all,” he added.
A version of this article first appeared on Medscape.com.
Last year, nine oncologists filed a lawsuit against the Anne Arundel Medical Center (AAMC), in Annapolis, Md., alleging that the hospital had fired them and had refused to allow them privileges to see their patients.
The oncologists said that the hospital chose profit over the needs of cancer patients, as it slashed oncology care services to cut costs.
The hospital denied any wrongdoing and alleged that the oncologists were not fired but that they had quit because they had been offered a more profitable opportunity.
At that time, the oncologists were not free to respond because of the ongoing litigation. But now that the lawsuit is over and the dust has settled, they are free to speak, and they contacted this news organization to tell their side of the story.
AAMC is a private, not-for-profit corporation that operates a large acute care hospital in Annapolis. It is affiliated with Luminis Health, the parent company of the medical center. Until October 23, 2020, the nine oncologists were employed by the AA Physician Group.
The doctors are Jason Taksey, MD, Benjamin Bridges, MD, Ravin Garg, MD, Adam Goldrich, MD, Carol Tweed, MD, Peter Graze, MD, Stuart Selonick, MD, David Weng, MD, and Jeanine Werner, MD.
They are all “highly respected, board certified oncologists and hematologists, with regional and, for some, national reputations in their medical specialty. The oncologists have had privileges at AAMC for many years and their capability as physicians is unquestioned,” according to the court filing made on behalf of the oncologists.
“Most of us have been in this town for decades,” said Dr. Tweed, who served as the unofficial spokesperson for the group. “Some of us are faculty members at Johns Hopkins, and this hospital’s oncology service was historically defined by our group.”
AAMC has a good reputation for providing high-quality medicine, “which is what brought many of us there in the first place,” Dr. Tweed said in an interview.
Triggered by cost cutting
The situation began when the hospital began cutting services to curtail costs, which directly affected the delivery of oncology care, Dr. Tweed explained. “They were also creating a very toxic and difficult interpersonal work environment, and that made it difficult to do patient care,” she said. “We would go to them and let them know that we were having difficulty delivering optimal patient care because we didn’t have enough staff or the resources we needed for safety — and it got to the point where we were being ignored and our input was no longer welcome.”
Dr. Tweed explained that the administrators announced which patient-care services would be cut without asking for their input as to the safety of those decisions. “Perhaps the most notorious was when they shut down the oncology lab,” she said. “That lab to an oncologist/hematologist is like a scalpel to a surgeon. I need lab results immediately — I need to know if I can give chemotherapy right now, or do I need to hold a dose. The lab is intrinsic to oncology care anywhere.”
There was a continuing cascade of events, and the oncology group mulled over some ideas as to how to provide optimal patient care in this increasingly difficult environment. The decision they reached was to discuss running their own practice with the hospital administrators as a means of making up for the gaps that they were now having to contend with. “As physicians, we do a lot of non-billable work, such as patient education, nighttime rounds for our cancer patients, and so on, and we told them that we would continue doing that,” said Dr. Tweed. “They said that they would talk to us, but they didn’t.”
Within a week of sending their proposal for setting up their own practice, all nine physicians were fired. “Instead of arranging a discussion, we received termination letters,” she explained. “We were terminated without cause.”
As physicians, Dr. Tweed explained that they were by contract obligated to arbitrate. It dragged on for weeks and months, to the tune of hundreds of thousands of dollars in legal fees.
“The only thing we wanted was to be able to practice in this town,” said Dr. Tweed. “And what is important to know is that it was never for money, and that was never our motivation for wanting to form our own practice.”
Dr. Tweed was referring to the hospital’s allegations that the oncologists had left their employment for monetary gain. A statement given to this news organization by the Luminis Health Anne Arundel Medical Center at the time stated that “this dispute started after nine oncologists left their employment to join a for-profit organization. We tried repeatedly to remain aligned with them.”
The oncologists had resigned during the height of the coronavirus pandemic to “pursue lucrative contracts” with a “major pharmaceutical distribution,” according to Todd M. Reinecker, attorney for Luminis Health, as reported by the Capital Gazette (this news organization reached out to Mr. Reinecker at that time but did not receive a response).
This was not the case, Dr. Tweed emphasized. “We took a great financial risk in doing this for patient care. It was pretty disgusting that was in print from the hospital’s lawyer.”
“The doctors anticipated Luminis Health would be unable to recruit new physicians and be forced to continue to use their services,” Mr. Reinecker maintained.
In fact, the medical center hired seven new oncologists to replace them.
Noncompete covenant
In filing their lawsuit, the nine oncologists put before the arbitrator the issue of the enforceability of the noncompete provision in their employment agreement, which prohibited the oncologists from working in the geographic area that includes the hospital. Their position was that the agreement was overly broad and thus unenforceable.
“We sign noncompete restrictive covenant contracts and we’re told that they are nonenforceable, and that’s the general discourse,” said Dr. Tweed. “Some states don’t even allow them. Well, we found out that they are very enforceable.”
The arbitrator eventually determined that three of the oncologists, including Dr. Tweed, had enforceable noncompete contracts.
“During the year or so while this was all going on, I would say that 90% of my patients wanted to stay with me,” said Dr. Tweed. “Patients were looking all over the place for us because, in many cases, the hospital did not tell them where to find us. In fact, they told us that we couldn’t contact the patients — they said it was ‘solicitation of a patient.’ “
In addition, the hospital continued to put more restrictions on the doctors. Six of the nine oncologists were able to continue practicing in Annapolis, and the remaining three will be able to join them in October 2022 when their noncompete contracts expire.
Now that the hospital has seen that there was a new oncology practice in town, Dr. Tweed noted, they changed their bylaws, and they now forbid hospital privileges to every physician in that group.
“The new bylaws do not restrict all private oncologists, just specifically our group, which prevents us from being able to do rounds in the hospital,” said Dr. Tweed. “If I want to see any of my patients, I have to get a visitor badge.”
Dr. Tweed contends that this move was purely for financial and business reasons to keep the oncologists from their patients. This is the primary hospital where their patients would be admitted if they need hospital care. AAMC is the only hospital within a 15-mile radius, and it serves as the regional hospital for the greater Annapolis area and for many Eastern Shore communities, whose hospitals do not offer various specialty services, such as oncology care.
“This was done purely because they were finance focused and not patient care focused,” Dr. Tweed emphasized. “We basically had to bargain with the hospital to let us even transfuse our patients.”
Telemedicine added to the mix
Yet another restriction that surfaced during the arbitration involved telemedicine. Dr. Tweed explained that as soon as the hospital realized that the three oncologists planned to stay in town and that their patients wanted to continue receiving care with them, they put telemedicine on the chopping block.
As if the restrictions and removal of hospital privileges wasn’t enough, the hospital decided to go after telemedicine during arbitration, Dr. Tweed said. If patients lived in any of the restricted ZIP codes, they were forbidden to conduct virtual visits with them.
“This isn’t ethical, but they tried to do everything to keep us from seeing our patients,” she said. “This is patient choice, but they were telling patients that if you live in any of these ZIP codes, you cannot do telemedicine if you choose Carol Tweed as your doctor,” Dr. Tweed said.
Of course, a patient isn’t bound by the arbitration and can see any doctor, but Dr. Tweed explained that the hospital threatened to come after her with a lawsuit.
One of the other physicians, Stuart Selonick, MD, said in an interview that he wasn’t quite sure how the idea of prohibiting telemedicine even came up. “There is little precedence for telemedicine in the U.S.,” he said. “They’ve extended the restrictions to telemedicine, and this is a new legal boundary, and it was new to the judge. But they made it part of the definition of the restrictive covenant. But to fight it would mean another lawsuit,” he added.
A separate lawsuit had previously been filed in an effort to regain hospital privileges, but the decision was made not to continue, owing to the amount of litigation it would involve.
“We can’t spend a lifetime and millions on another legal battle,” said Dr. Tweed. “We don’t have the corporate legal pool that the hospital has, and they know it.”
Patients have written endless letters supporting the doctors, Dr. Tweed said, but to no avail, as the hospital did not change course.
Litigation is now completed, and in about 9 months, the remaining three physicians will be able to rejoin their colleagues and put this behind them as best they can.
“The hospital knows that they harmed patient care for financial gain -- that’s the tagline,” said Tweed.
Approached for a response, Justin McLeod, spokesperson for Luminis Health, said that they are “pleased with the outcome of the case and the resolution agreed to by both sides. This agreement ensures patient access and continuity of care for patients with cancer. These providers have access to their patients’ electronic medical records, can order outpatient services, and attend quarterly cancer committee meetings with other providers.
“Our focus is the future of cancer care for our community. Luminis Health Anne Arundel Medical Center is committed to providing patients with high quality, comprehensive cancer care that is accessible to all,” he added.
A version of this article first appeared on Medscape.com.
Which companies aren’t exiting Russia? Big pharma
Even as the war in Ukraine has prompted an exodus of international companies — from fast-food chains and oil producers to luxury retailers — from Russia,
Airlines, automakers, banks, and technology giants — at least 320 companies by one count — are among the businesses curtailing operations or making high-profile exits from Russia as its invasion of Ukraine intensifies. McDonald’s, Starbucks, and Coca-Cola announced a pause in sales recently.
But drugmakers, medical device manufacturers, and health care companies, which are exempted from U.S. and European sanctions, said Russians need access to medicines and medical equipment and contend that international humanitarian law requires they keep supply chains open.
“As a health care company, we have an important purpose, which is why at this time we continue to serve people in all countries in which we operate who depend on us for essential products, some life-sustaining,” said Scott Stoffel, divisional vice president for Illinois-based Abbott Laboratories, which manufactures and sells medicines in Russia for oncology, women’s health, pancreatic insufficiency, and liver health.
Johnson & Johnson — which has corporate offices in Moscow, Novosibirsk, St. Petersburg, and Yekaterinburg — said in a statement, “We remain committed to providing essential health products to those in need in Ukraine, Russia, and the region, in compliance with current sanctions and while adapting to the rapidly changing situation on the ground.”
The reluctance of drugmakers to pause operations in Russia is being met with a growing chorus of criticism.
Pharmaceutical companies that say they must continue to manufacture drugs in Russia for humanitarian reasons are “being misguided at best, cynical in the medium case, and outright deplorably misleading and deceptive,” said Jeffrey Sonnenfeld, DBA, a professor at the Yale School of Management who is tracking which companies have curtailed operations in Russia. He noted that banks and technology companies also provide essential services.
“Russians are put in a tragic position of unearned suffering. If we continue to make life palatable for them, then we are continuing to support the regime,” Dr. Sonnenfeld said. “These drug companies will be seen as complicit with the most vicious operation on the planet. Instead of protecting life, they are going to be seen as destroying life. The goal here is to show that Putin is not in control of all sectors of the economy.”
U.S. pharmaceutical and medical companies have operated in Russia for decades, and many ramped up operations after Russia invaded and annexed Crimea in 2014, navigating the fraught relationship between the United States and Russia amid sanctions. In 2010, Vladimir Putin, then Russian prime minister, announced an ambitious national plan for the Russian pharmaceutical industry that would be a pillar in his efforts to reestablish his country as an influential superpower and wean the country off Western pharmaceutical imports. Under the plan, called “Pharma-2020” and “Pharma-2030,” the government required Western pharmaceutical companies eager to sell to Russia’s growing middle class to locate production inside the country.
Pfizer, Johnson & Johnson, Novartis, and Abbott are among the drugmakers that manufacture pharmaceutical drugs at facilities in St. Petersburg and elsewhere in the country and typically sell those drugs as branded generics or under Russian brands.
Pfizer’s CEO, Albert Bourla, said on CBS that the giant drugmaker is not going to make further investments in Russia, but that it will not cut ties with Russia, as multinational companies in other industries are doing.
Pharmaceutical manufacturing plants in Kaluga, a major manufacturing center for Volkswagen and Volvo southwest of Moscow, have been funded through a partnership between Rusnano, a state-owned venture that promotes the development of high-tech enterprises, and U.S. venture capital firms.
Russia also has sought to position itself as an attractive research market, offering an inexpensive and lax regulatory environment for clinical drug trials. Last year, Pfizer conducted in Russia clinical trials of Paxlovid, its experimental antiviral pill to treat covid-19. Before the invasion began in late February, 3,072 trials were underway in Russia and 503 were underway in Ukraine, according to BioWorld, a reporting hub focused on drug development that features data from Cortellis.
AstraZeneca is the top sponsor of clinical trials in Russia, with 49 trials, followed by a subsidiary of Merck, with 48 trials.
So far, drugmakers’ response to the Ukraine invasion has largely centered on public pledges to donate essential medicines and vaccines to Ukrainian patients and refugees. They’ve also made general comments about the need to keep open the supply of medicines flowing within Russia.
Abbott has pledged $2 million to support humanitarian efforts in Ukraine, and Pfizer, based in New York, said it has supplied $1 million in humanitarian grants. Swiss drug maker Novartis said it was expanding humanitarian efforts in Ukraine and working to “ensure the continued supply of our medicines in Ukraine.”
But no major pharmaceutical or medical device maker has announced plans to shutter manufacturing plants or halt sales inside Russia.
In an open letter, hundreds of leaders of mainly smaller biotechnology companies have called on industry members to cease business activities in Russia, including “investment in Russian companies and new investment within the borders of Russia,” and to halt trade and collaboration with Russian companies, except for supplying food and medicines. How many of the signatories have business operations in Russia was unclear.
Ulrich Neumann, director for market access at Janssen, a Johnson & Johnson company, was among those who signed the letter, but whether he was speaking for the company was unclear. In its own statement posted on social media, the company said it’s “committed to providing access to our essential medical products in the countries where we operate, in compliance with current international sanctions.”
GlaxoSmithKline, headquartered in the United Kingdom, said in a statement that it’s stopping all advertising in Russia and will not enter into contracts that “directly support the Russian administration or military.” But the company said that as a “supplier of needed medicines, vaccines and everyday health products, we have a responsibility to do all we can to make them available. For this reason, we will continue to supply our products to the people of Russia, while we can.”
Nell Minow, vice chair of ValueEdge Advisors, an investment consulting firm, noted that drug companies have been treated differently than other industries during previous global conflicts. For example, some corporate ethicists advised against pharmaceutical companies’ total divestment from South Africa’s apartheid regime to ensure essential medicines flowed to the country.
“There is a difference between a hamburger and a pill,” Mr. Minow said. Companies should strongly condemn Russia’s actions, she said, but unless the United States enters directly into a war with Russia, companies that make essential medicines and health care products should continue to operate. Before U.S. involvement in World War II, she added, there were “some American companies that did business with Germany until the last minute.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation. KHN senior correspondent Arthur Allen contributed to this article.
Even as the war in Ukraine has prompted an exodus of international companies — from fast-food chains and oil producers to luxury retailers — from Russia,
Airlines, automakers, banks, and technology giants — at least 320 companies by one count — are among the businesses curtailing operations or making high-profile exits from Russia as its invasion of Ukraine intensifies. McDonald’s, Starbucks, and Coca-Cola announced a pause in sales recently.
But drugmakers, medical device manufacturers, and health care companies, which are exempted from U.S. and European sanctions, said Russians need access to medicines and medical equipment and contend that international humanitarian law requires they keep supply chains open.
“As a health care company, we have an important purpose, which is why at this time we continue to serve people in all countries in which we operate who depend on us for essential products, some life-sustaining,” said Scott Stoffel, divisional vice president for Illinois-based Abbott Laboratories, which manufactures and sells medicines in Russia for oncology, women’s health, pancreatic insufficiency, and liver health.
Johnson & Johnson — which has corporate offices in Moscow, Novosibirsk, St. Petersburg, and Yekaterinburg — said in a statement, “We remain committed to providing essential health products to those in need in Ukraine, Russia, and the region, in compliance with current sanctions and while adapting to the rapidly changing situation on the ground.”
The reluctance of drugmakers to pause operations in Russia is being met with a growing chorus of criticism.
Pharmaceutical companies that say they must continue to manufacture drugs in Russia for humanitarian reasons are “being misguided at best, cynical in the medium case, and outright deplorably misleading and deceptive,” said Jeffrey Sonnenfeld, DBA, a professor at the Yale School of Management who is tracking which companies have curtailed operations in Russia. He noted that banks and technology companies also provide essential services.
“Russians are put in a tragic position of unearned suffering. If we continue to make life palatable for them, then we are continuing to support the regime,” Dr. Sonnenfeld said. “These drug companies will be seen as complicit with the most vicious operation on the planet. Instead of protecting life, they are going to be seen as destroying life. The goal here is to show that Putin is not in control of all sectors of the economy.”
U.S. pharmaceutical and medical companies have operated in Russia for decades, and many ramped up operations after Russia invaded and annexed Crimea in 2014, navigating the fraught relationship between the United States and Russia amid sanctions. In 2010, Vladimir Putin, then Russian prime minister, announced an ambitious national plan for the Russian pharmaceutical industry that would be a pillar in his efforts to reestablish his country as an influential superpower and wean the country off Western pharmaceutical imports. Under the plan, called “Pharma-2020” and “Pharma-2030,” the government required Western pharmaceutical companies eager to sell to Russia’s growing middle class to locate production inside the country.
Pfizer, Johnson & Johnson, Novartis, and Abbott are among the drugmakers that manufacture pharmaceutical drugs at facilities in St. Petersburg and elsewhere in the country and typically sell those drugs as branded generics or under Russian brands.
Pfizer’s CEO, Albert Bourla, said on CBS that the giant drugmaker is not going to make further investments in Russia, but that it will not cut ties with Russia, as multinational companies in other industries are doing.
Pharmaceutical manufacturing plants in Kaluga, a major manufacturing center for Volkswagen and Volvo southwest of Moscow, have been funded through a partnership between Rusnano, a state-owned venture that promotes the development of high-tech enterprises, and U.S. venture capital firms.
Russia also has sought to position itself as an attractive research market, offering an inexpensive and lax regulatory environment for clinical drug trials. Last year, Pfizer conducted in Russia clinical trials of Paxlovid, its experimental antiviral pill to treat covid-19. Before the invasion began in late February, 3,072 trials were underway in Russia and 503 were underway in Ukraine, according to BioWorld, a reporting hub focused on drug development that features data from Cortellis.
AstraZeneca is the top sponsor of clinical trials in Russia, with 49 trials, followed by a subsidiary of Merck, with 48 trials.
So far, drugmakers’ response to the Ukraine invasion has largely centered on public pledges to donate essential medicines and vaccines to Ukrainian patients and refugees. They’ve also made general comments about the need to keep open the supply of medicines flowing within Russia.
Abbott has pledged $2 million to support humanitarian efforts in Ukraine, and Pfizer, based in New York, said it has supplied $1 million in humanitarian grants. Swiss drug maker Novartis said it was expanding humanitarian efforts in Ukraine and working to “ensure the continued supply of our medicines in Ukraine.”
But no major pharmaceutical or medical device maker has announced plans to shutter manufacturing plants or halt sales inside Russia.
In an open letter, hundreds of leaders of mainly smaller biotechnology companies have called on industry members to cease business activities in Russia, including “investment in Russian companies and new investment within the borders of Russia,” and to halt trade and collaboration with Russian companies, except for supplying food and medicines. How many of the signatories have business operations in Russia was unclear.
Ulrich Neumann, director for market access at Janssen, a Johnson & Johnson company, was among those who signed the letter, but whether he was speaking for the company was unclear. In its own statement posted on social media, the company said it’s “committed to providing access to our essential medical products in the countries where we operate, in compliance with current international sanctions.”
GlaxoSmithKline, headquartered in the United Kingdom, said in a statement that it’s stopping all advertising in Russia and will not enter into contracts that “directly support the Russian administration or military.” But the company said that as a “supplier of needed medicines, vaccines and everyday health products, we have a responsibility to do all we can to make them available. For this reason, we will continue to supply our products to the people of Russia, while we can.”
Nell Minow, vice chair of ValueEdge Advisors, an investment consulting firm, noted that drug companies have been treated differently than other industries during previous global conflicts. For example, some corporate ethicists advised against pharmaceutical companies’ total divestment from South Africa’s apartheid regime to ensure essential medicines flowed to the country.
“There is a difference between a hamburger and a pill,” Mr. Minow said. Companies should strongly condemn Russia’s actions, she said, but unless the United States enters directly into a war with Russia, companies that make essential medicines and health care products should continue to operate. Before U.S. involvement in World War II, she added, there were “some American companies that did business with Germany until the last minute.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation. KHN senior correspondent Arthur Allen contributed to this article.
Even as the war in Ukraine has prompted an exodus of international companies — from fast-food chains and oil producers to luxury retailers — from Russia,
Airlines, automakers, banks, and technology giants — at least 320 companies by one count — are among the businesses curtailing operations or making high-profile exits from Russia as its invasion of Ukraine intensifies. McDonald’s, Starbucks, and Coca-Cola announced a pause in sales recently.
But drugmakers, medical device manufacturers, and health care companies, which are exempted from U.S. and European sanctions, said Russians need access to medicines and medical equipment and contend that international humanitarian law requires they keep supply chains open.
“As a health care company, we have an important purpose, which is why at this time we continue to serve people in all countries in which we operate who depend on us for essential products, some life-sustaining,” said Scott Stoffel, divisional vice president for Illinois-based Abbott Laboratories, which manufactures and sells medicines in Russia for oncology, women’s health, pancreatic insufficiency, and liver health.
Johnson & Johnson — which has corporate offices in Moscow, Novosibirsk, St. Petersburg, and Yekaterinburg — said in a statement, “We remain committed to providing essential health products to those in need in Ukraine, Russia, and the region, in compliance with current sanctions and while adapting to the rapidly changing situation on the ground.”
The reluctance of drugmakers to pause operations in Russia is being met with a growing chorus of criticism.
Pharmaceutical companies that say they must continue to manufacture drugs in Russia for humanitarian reasons are “being misguided at best, cynical in the medium case, and outright deplorably misleading and deceptive,” said Jeffrey Sonnenfeld, DBA, a professor at the Yale School of Management who is tracking which companies have curtailed operations in Russia. He noted that banks and technology companies also provide essential services.
“Russians are put in a tragic position of unearned suffering. If we continue to make life palatable for them, then we are continuing to support the regime,” Dr. Sonnenfeld said. “These drug companies will be seen as complicit with the most vicious operation on the planet. Instead of protecting life, they are going to be seen as destroying life. The goal here is to show that Putin is not in control of all sectors of the economy.”
U.S. pharmaceutical and medical companies have operated in Russia for decades, and many ramped up operations after Russia invaded and annexed Crimea in 2014, navigating the fraught relationship between the United States and Russia amid sanctions. In 2010, Vladimir Putin, then Russian prime minister, announced an ambitious national plan for the Russian pharmaceutical industry that would be a pillar in his efforts to reestablish his country as an influential superpower and wean the country off Western pharmaceutical imports. Under the plan, called “Pharma-2020” and “Pharma-2030,” the government required Western pharmaceutical companies eager to sell to Russia’s growing middle class to locate production inside the country.
Pfizer, Johnson & Johnson, Novartis, and Abbott are among the drugmakers that manufacture pharmaceutical drugs at facilities in St. Petersburg and elsewhere in the country and typically sell those drugs as branded generics or under Russian brands.
Pfizer’s CEO, Albert Bourla, said on CBS that the giant drugmaker is not going to make further investments in Russia, but that it will not cut ties with Russia, as multinational companies in other industries are doing.
Pharmaceutical manufacturing plants in Kaluga, a major manufacturing center for Volkswagen and Volvo southwest of Moscow, have been funded through a partnership between Rusnano, a state-owned venture that promotes the development of high-tech enterprises, and U.S. venture capital firms.
Russia also has sought to position itself as an attractive research market, offering an inexpensive and lax regulatory environment for clinical drug trials. Last year, Pfizer conducted in Russia clinical trials of Paxlovid, its experimental antiviral pill to treat covid-19. Before the invasion began in late February, 3,072 trials were underway in Russia and 503 were underway in Ukraine, according to BioWorld, a reporting hub focused on drug development that features data from Cortellis.
AstraZeneca is the top sponsor of clinical trials in Russia, with 49 trials, followed by a subsidiary of Merck, with 48 trials.
So far, drugmakers’ response to the Ukraine invasion has largely centered on public pledges to donate essential medicines and vaccines to Ukrainian patients and refugees. They’ve also made general comments about the need to keep open the supply of medicines flowing within Russia.
Abbott has pledged $2 million to support humanitarian efforts in Ukraine, and Pfizer, based in New York, said it has supplied $1 million in humanitarian grants. Swiss drug maker Novartis said it was expanding humanitarian efforts in Ukraine and working to “ensure the continued supply of our medicines in Ukraine.”
But no major pharmaceutical or medical device maker has announced plans to shutter manufacturing plants or halt sales inside Russia.
In an open letter, hundreds of leaders of mainly smaller biotechnology companies have called on industry members to cease business activities in Russia, including “investment in Russian companies and new investment within the borders of Russia,” and to halt trade and collaboration with Russian companies, except for supplying food and medicines. How many of the signatories have business operations in Russia was unclear.
Ulrich Neumann, director for market access at Janssen, a Johnson & Johnson company, was among those who signed the letter, but whether he was speaking for the company was unclear. In its own statement posted on social media, the company said it’s “committed to providing access to our essential medical products in the countries where we operate, in compliance with current international sanctions.”
GlaxoSmithKline, headquartered in the United Kingdom, said in a statement that it’s stopping all advertising in Russia and will not enter into contracts that “directly support the Russian administration or military.” But the company said that as a “supplier of needed medicines, vaccines and everyday health products, we have a responsibility to do all we can to make them available. For this reason, we will continue to supply our products to the people of Russia, while we can.”
Nell Minow, vice chair of ValueEdge Advisors, an investment consulting firm, noted that drug companies have been treated differently than other industries during previous global conflicts. For example, some corporate ethicists advised against pharmaceutical companies’ total divestment from South Africa’s apartheid regime to ensure essential medicines flowed to the country.
“There is a difference between a hamburger and a pill,” Mr. Minow said. Companies should strongly condemn Russia’s actions, she said, but unless the United States enters directly into a war with Russia, companies that make essential medicines and health care products should continue to operate. Before U.S. involvement in World War II, she added, there were “some American companies that did business with Germany until the last minute.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation. KHN senior correspondent Arthur Allen contributed to this article.
Wake Forest Cancer Center director fired, advisory board resigns
and withdrew their endorsement for renewal of the center’s National Cancer Institute comprehensive cancer center support grant.
The move was prompted by the abrupt firing of center director Boris Pasche, MD, PhD, on February 10, one day after NCI renewed a multimillion dollar grant.
The Cancer Letter broke the story and published the resignation letter from the EAB. It was signed by board chair Gerold Bepler, MD, PhD, CEO and director of the Karmanos Cancer Institute, Detroit, on behalf of the board.
The mass resignation of an EAB, a panel of outside experts that help shepherd cancer centers through the NCI grant process, is “highly unusual,” according to The Cancer Letter. It also raises concerns about the “immediate future” of Wake Forest’s cancer center, the publication added.
Numerous people involved with the situation did not respond or declined to comment when this news organization requested additional information and updates, including questions about the reason for Dr. Pasche’s termination; whether or not withdrawal of the endorsement puts Wake’s NCI designation in jeopardy; and if the EAB is being reconstituted.
A written statement from Wake Forest simply said that “the situation involving Dr. Pasche is an administrative decision. Various administrative changes occur regularly in organizations across the country. Dr. Pasche remains employed by Atrium Health Wake Forest Baptist. We are very grateful to Dr. Pasche for his years of service and many contributions to the mission and vision of our NCI-designated Comprehensive Cancer Center in Winston-Salem.”
Wake’s cancer center is in the process of combining with the Atrium Health Levine Cancer Center, which is not NCI-designated, following Atrium Health system’s recent acquisition of the Wake Forest Baptist Medical Center.
The NCI renewal notice, dated February 9, states that Dr. Pasche “and his leadership team have built a robust, transdisciplinary center that includes 140 scientists.”
Dr. Pasche was fired a day later.
The EAB resignation letter states that during Wake Forest’s recent NCI review process, “leadership gave their glowing endorsement of Dr. Pasche...This endorsement included unequivocal statements of support for Dr. Pasche’s oversight of the combined Atrium-Wake Forest cancer program.”
“What followed was his rapid dismissal after the...notice of award was issued, following a period during which the approach to integration was apparently being revisited,” Dr. Bepler said on behalf of the board.
“It is with sadness and dismay that we witnessed the change in approach by the institutional leadership towards” the merger, he wrote.
The Cancer Letter quotes an unnamed board member as saying, “EABs for cancer centers can only provide value to the center when there is openness and transparency in the process. In the absence of such, I believe the members felt that there was no further utility in providing guidance to the organization.”
The resignation letter was sent to the interim director of Wake’s cancer center, radiation oncologist William Blackstock, Jr, MD, and also copied to Atrium-Wake and NCI leadership.
The resignation letter endorsed Dr. Blackstock’s qualifications to run the center, and noted that as the board is reconstituted, “some of us would be honored to discuss participation...if there is unequivocal evidence from the health system’s senior management for support of a single, academically driven, comprehensive, and integrated cancer center.”
A version of this article first appeared on Medscape.com.
and withdrew their endorsement for renewal of the center’s National Cancer Institute comprehensive cancer center support grant.
The move was prompted by the abrupt firing of center director Boris Pasche, MD, PhD, on February 10, one day after NCI renewed a multimillion dollar grant.
The Cancer Letter broke the story and published the resignation letter from the EAB. It was signed by board chair Gerold Bepler, MD, PhD, CEO and director of the Karmanos Cancer Institute, Detroit, on behalf of the board.
The mass resignation of an EAB, a panel of outside experts that help shepherd cancer centers through the NCI grant process, is “highly unusual,” according to The Cancer Letter. It also raises concerns about the “immediate future” of Wake Forest’s cancer center, the publication added.
Numerous people involved with the situation did not respond or declined to comment when this news organization requested additional information and updates, including questions about the reason for Dr. Pasche’s termination; whether or not withdrawal of the endorsement puts Wake’s NCI designation in jeopardy; and if the EAB is being reconstituted.
A written statement from Wake Forest simply said that “the situation involving Dr. Pasche is an administrative decision. Various administrative changes occur regularly in organizations across the country. Dr. Pasche remains employed by Atrium Health Wake Forest Baptist. We are very grateful to Dr. Pasche for his years of service and many contributions to the mission and vision of our NCI-designated Comprehensive Cancer Center in Winston-Salem.”
Wake’s cancer center is in the process of combining with the Atrium Health Levine Cancer Center, which is not NCI-designated, following Atrium Health system’s recent acquisition of the Wake Forest Baptist Medical Center.
The NCI renewal notice, dated February 9, states that Dr. Pasche “and his leadership team have built a robust, transdisciplinary center that includes 140 scientists.”
Dr. Pasche was fired a day later.
The EAB resignation letter states that during Wake Forest’s recent NCI review process, “leadership gave their glowing endorsement of Dr. Pasche...This endorsement included unequivocal statements of support for Dr. Pasche’s oversight of the combined Atrium-Wake Forest cancer program.”
“What followed was his rapid dismissal after the...notice of award was issued, following a period during which the approach to integration was apparently being revisited,” Dr. Bepler said on behalf of the board.
“It is with sadness and dismay that we witnessed the change in approach by the institutional leadership towards” the merger, he wrote.
The Cancer Letter quotes an unnamed board member as saying, “EABs for cancer centers can only provide value to the center when there is openness and transparency in the process. In the absence of such, I believe the members felt that there was no further utility in providing guidance to the organization.”
The resignation letter was sent to the interim director of Wake’s cancer center, radiation oncologist William Blackstock, Jr, MD, and also copied to Atrium-Wake and NCI leadership.
The resignation letter endorsed Dr. Blackstock’s qualifications to run the center, and noted that as the board is reconstituted, “some of us would be honored to discuss participation...if there is unequivocal evidence from the health system’s senior management for support of a single, academically driven, comprehensive, and integrated cancer center.”
A version of this article first appeared on Medscape.com.
and withdrew their endorsement for renewal of the center’s National Cancer Institute comprehensive cancer center support grant.
The move was prompted by the abrupt firing of center director Boris Pasche, MD, PhD, on February 10, one day after NCI renewed a multimillion dollar grant.
The Cancer Letter broke the story and published the resignation letter from the EAB. It was signed by board chair Gerold Bepler, MD, PhD, CEO and director of the Karmanos Cancer Institute, Detroit, on behalf of the board.
The mass resignation of an EAB, a panel of outside experts that help shepherd cancer centers through the NCI grant process, is “highly unusual,” according to The Cancer Letter. It also raises concerns about the “immediate future” of Wake Forest’s cancer center, the publication added.
Numerous people involved with the situation did not respond or declined to comment when this news organization requested additional information and updates, including questions about the reason for Dr. Pasche’s termination; whether or not withdrawal of the endorsement puts Wake’s NCI designation in jeopardy; and if the EAB is being reconstituted.
A written statement from Wake Forest simply said that “the situation involving Dr. Pasche is an administrative decision. Various administrative changes occur regularly in organizations across the country. Dr. Pasche remains employed by Atrium Health Wake Forest Baptist. We are very grateful to Dr. Pasche for his years of service and many contributions to the mission and vision of our NCI-designated Comprehensive Cancer Center in Winston-Salem.”
Wake’s cancer center is in the process of combining with the Atrium Health Levine Cancer Center, which is not NCI-designated, following Atrium Health system’s recent acquisition of the Wake Forest Baptist Medical Center.
The NCI renewal notice, dated February 9, states that Dr. Pasche “and his leadership team have built a robust, transdisciplinary center that includes 140 scientists.”
Dr. Pasche was fired a day later.
The EAB resignation letter states that during Wake Forest’s recent NCI review process, “leadership gave their glowing endorsement of Dr. Pasche...This endorsement included unequivocal statements of support for Dr. Pasche’s oversight of the combined Atrium-Wake Forest cancer program.”
“What followed was his rapid dismissal after the...notice of award was issued, following a period during which the approach to integration was apparently being revisited,” Dr. Bepler said on behalf of the board.
“It is with sadness and dismay that we witnessed the change in approach by the institutional leadership towards” the merger, he wrote.
The Cancer Letter quotes an unnamed board member as saying, “EABs for cancer centers can only provide value to the center when there is openness and transparency in the process. In the absence of such, I believe the members felt that there was no further utility in providing guidance to the organization.”
The resignation letter was sent to the interim director of Wake’s cancer center, radiation oncologist William Blackstock, Jr, MD, and also copied to Atrium-Wake and NCI leadership.
The resignation letter endorsed Dr. Blackstock’s qualifications to run the center, and noted that as the board is reconstituted, “some of us would be honored to discuss participation...if there is unequivocal evidence from the health system’s senior management for support of a single, academically driven, comprehensive, and integrated cancer center.”
A version of this article first appeared on Medscape.com.
Physicians beware: Feds start tracking information-blocking claims
The federal government’s efforts to thwart information blocking are underway. As such,
Recently, the Office of the National Coordinator revealed that the Department of Health & Humans Services has received 299 reports of information blocking since inviting anyone who suspected that health care providers, IT developers, or health information networks/exchanges might have interfered with access, exchange, or use of EHI through the Report Information Blocking Portal on April 5, 2021.
The vast majority of these claims – 211 – were filed against providers, while 46 alleged incidents of information blocking were by health IT developers, and two claims point to health information networks/ exchanges. The other 25 claims did not appear to present a claim of information blocking.
Of the 274 possible claims of information blocking recently released by ONC, 176 were made by patients.
The ONC has sent all possible claims to the HHS’s Office of the Inspector General. The claims have not yet been investigated and substantiated.
Do the stats tell the story?
The numbers in the recent ONC report do not shed much light on how much impact the regulations are having on information sharing. Health care providers, including physicians, might not yet be complying with the rules because monetary penalties are not in place.
Indeed, HHS has yet to spell out exactly what the disincentives on providers will be, though the 21st Century Cures Act stipulates that regulators could fine up to $1 million per information-blocking incident.
“Some providers might be saying, ‘I’m not going to be penalized at this point … so I can take a little bit longer to think about how I come into compliance.’ That could be just one factor of a host of many that are affecting compliance. We also are still in the middle of a public health emergency. So it’s hard to say at this point” exactly how the regulations will affect information blocking, Lauren Riplinger, vice president of policy and public affairs at the American Health Information Management Association, Chicago, said in an interview.
A long time coming
The government first zeroed in on ensuring that patients have access to their information in 2016 when President Obama signed the Cures Act into law. The legislation directed ONC to implement a standardized process for the public to report claims of possible information blocking.
The initiative appears to be picking up steam. The ONC is expected to release monthly reports on the cumulative number of information-blocking claims. The announcement of associated penalties is expected sometime in the future.
Industry leaders are advising health care providers to brush up on compliance. Physicians can look to professional groups such as the American Medical Association, the Medical Group Management Association, and other specialty associations for guidance. In addition, the ONC is educating providers on the rule.
“The ONC has provided a lot of great content for the past couple months, not only in terms of putting out FAQs to help clarify some of the gray areas in the rule, but they also have produced a series of provider-specific webinars where they walk through a potential scenario and address the extent to the rules apply,” Ms. Riplinger said.
With education, more is better
These efforts, however, could be expanded, according to MGMA.
“There is a general awareness of the rules, but we encourage ONC to continue educating the provider community: More FAQs and educational webinars would be helpful,” Claire Ernst, director of government affairs for MGMA, said in an interview. “A June 2021 MGMA poll found that 51% of medical groups said they needed more government guidance on complying with the new information-blocking rules.”
Although ONC already has provided some “scenario-based” education, more of this type of guidance could prove valuable.
“This rule is that it is very circumstance based. … and so it’s those more nuanced cases that I think are more challenging for providers to know whether or not they are engaging in information blocking,” Ms. Riplinger noted.
For example, a physician might choose to not upload lab test results to a patient portal and prefer to wait to discuss the results directly with the patient, which could potentially be construed as information blocking under the regulations.
The MGMA is requesting that ONC take a second look at these situations – and possibly adjust the regulations.
“MGMA has heard concerns about the impact of providing immediate results to patients before medical groups have the time to thoroughly review test results and discuss them compassionately with their patients,” Ms. Ernst said. “To address this, ONC could expand the current definition of harm to account for other unintended consequences, such as emotional distress, or provide more flexibility in terms of the time frame.”
A version of this article first appeared on Medscape.com.
The federal government’s efforts to thwart information blocking are underway. As such,
Recently, the Office of the National Coordinator revealed that the Department of Health & Humans Services has received 299 reports of information blocking since inviting anyone who suspected that health care providers, IT developers, or health information networks/exchanges might have interfered with access, exchange, or use of EHI through the Report Information Blocking Portal on April 5, 2021.
The vast majority of these claims – 211 – were filed against providers, while 46 alleged incidents of information blocking were by health IT developers, and two claims point to health information networks/ exchanges. The other 25 claims did not appear to present a claim of information blocking.
Of the 274 possible claims of information blocking recently released by ONC, 176 were made by patients.
The ONC has sent all possible claims to the HHS’s Office of the Inspector General. The claims have not yet been investigated and substantiated.
Do the stats tell the story?
The numbers in the recent ONC report do not shed much light on how much impact the regulations are having on information sharing. Health care providers, including physicians, might not yet be complying with the rules because monetary penalties are not in place.
Indeed, HHS has yet to spell out exactly what the disincentives on providers will be, though the 21st Century Cures Act stipulates that regulators could fine up to $1 million per information-blocking incident.
“Some providers might be saying, ‘I’m not going to be penalized at this point … so I can take a little bit longer to think about how I come into compliance.’ That could be just one factor of a host of many that are affecting compliance. We also are still in the middle of a public health emergency. So it’s hard to say at this point” exactly how the regulations will affect information blocking, Lauren Riplinger, vice president of policy and public affairs at the American Health Information Management Association, Chicago, said in an interview.
A long time coming
The government first zeroed in on ensuring that patients have access to their information in 2016 when President Obama signed the Cures Act into law. The legislation directed ONC to implement a standardized process for the public to report claims of possible information blocking.
The initiative appears to be picking up steam. The ONC is expected to release monthly reports on the cumulative number of information-blocking claims. The announcement of associated penalties is expected sometime in the future.
Industry leaders are advising health care providers to brush up on compliance. Physicians can look to professional groups such as the American Medical Association, the Medical Group Management Association, and other specialty associations for guidance. In addition, the ONC is educating providers on the rule.
“The ONC has provided a lot of great content for the past couple months, not only in terms of putting out FAQs to help clarify some of the gray areas in the rule, but they also have produced a series of provider-specific webinars where they walk through a potential scenario and address the extent to the rules apply,” Ms. Riplinger said.
With education, more is better
These efforts, however, could be expanded, according to MGMA.
“There is a general awareness of the rules, but we encourage ONC to continue educating the provider community: More FAQs and educational webinars would be helpful,” Claire Ernst, director of government affairs for MGMA, said in an interview. “A June 2021 MGMA poll found that 51% of medical groups said they needed more government guidance on complying with the new information-blocking rules.”
Although ONC already has provided some “scenario-based” education, more of this type of guidance could prove valuable.
“This rule is that it is very circumstance based. … and so it’s those more nuanced cases that I think are more challenging for providers to know whether or not they are engaging in information blocking,” Ms. Riplinger noted.
For example, a physician might choose to not upload lab test results to a patient portal and prefer to wait to discuss the results directly with the patient, which could potentially be construed as information blocking under the regulations.
The MGMA is requesting that ONC take a second look at these situations – and possibly adjust the regulations.
“MGMA has heard concerns about the impact of providing immediate results to patients before medical groups have the time to thoroughly review test results and discuss them compassionately with their patients,” Ms. Ernst said. “To address this, ONC could expand the current definition of harm to account for other unintended consequences, such as emotional distress, or provide more flexibility in terms of the time frame.”
A version of this article first appeared on Medscape.com.
The federal government’s efforts to thwart information blocking are underway. As such,
Recently, the Office of the National Coordinator revealed that the Department of Health & Humans Services has received 299 reports of information blocking since inviting anyone who suspected that health care providers, IT developers, or health information networks/exchanges might have interfered with access, exchange, or use of EHI through the Report Information Blocking Portal on April 5, 2021.
The vast majority of these claims – 211 – were filed against providers, while 46 alleged incidents of information blocking were by health IT developers, and two claims point to health information networks/ exchanges. The other 25 claims did not appear to present a claim of information blocking.
Of the 274 possible claims of information blocking recently released by ONC, 176 were made by patients.
The ONC has sent all possible claims to the HHS’s Office of the Inspector General. The claims have not yet been investigated and substantiated.
Do the stats tell the story?
The numbers in the recent ONC report do not shed much light on how much impact the regulations are having on information sharing. Health care providers, including physicians, might not yet be complying with the rules because monetary penalties are not in place.
Indeed, HHS has yet to spell out exactly what the disincentives on providers will be, though the 21st Century Cures Act stipulates that regulators could fine up to $1 million per information-blocking incident.
“Some providers might be saying, ‘I’m not going to be penalized at this point … so I can take a little bit longer to think about how I come into compliance.’ That could be just one factor of a host of many that are affecting compliance. We also are still in the middle of a public health emergency. So it’s hard to say at this point” exactly how the regulations will affect information blocking, Lauren Riplinger, vice president of policy and public affairs at the American Health Information Management Association, Chicago, said in an interview.
A long time coming
The government first zeroed in on ensuring that patients have access to their information in 2016 when President Obama signed the Cures Act into law. The legislation directed ONC to implement a standardized process for the public to report claims of possible information blocking.
The initiative appears to be picking up steam. The ONC is expected to release monthly reports on the cumulative number of information-blocking claims. The announcement of associated penalties is expected sometime in the future.
Industry leaders are advising health care providers to brush up on compliance. Physicians can look to professional groups such as the American Medical Association, the Medical Group Management Association, and other specialty associations for guidance. In addition, the ONC is educating providers on the rule.
“The ONC has provided a lot of great content for the past couple months, not only in terms of putting out FAQs to help clarify some of the gray areas in the rule, but they also have produced a series of provider-specific webinars where they walk through a potential scenario and address the extent to the rules apply,” Ms. Riplinger said.
With education, more is better
These efforts, however, could be expanded, according to MGMA.
“There is a general awareness of the rules, but we encourage ONC to continue educating the provider community: More FAQs and educational webinars would be helpful,” Claire Ernst, director of government affairs for MGMA, said in an interview. “A June 2021 MGMA poll found that 51% of medical groups said they needed more government guidance on complying with the new information-blocking rules.”
Although ONC already has provided some “scenario-based” education, more of this type of guidance could prove valuable.
“This rule is that it is very circumstance based. … and so it’s those more nuanced cases that I think are more challenging for providers to know whether or not they are engaging in information blocking,” Ms. Riplinger noted.
For example, a physician might choose to not upload lab test results to a patient portal and prefer to wait to discuss the results directly with the patient, which could potentially be construed as information blocking under the regulations.
The MGMA is requesting that ONC take a second look at these situations – and possibly adjust the regulations.
“MGMA has heard concerns about the impact of providing immediate results to patients before medical groups have the time to thoroughly review test results and discuss them compassionately with their patients,” Ms. Ernst said. “To address this, ONC could expand the current definition of harm to account for other unintended consequences, such as emotional distress, or provide more flexibility in terms of the time frame.”
A version of this article first appeared on Medscape.com.
Former physician sentenced to 20 years in pill mill case
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
Depression, suicidal ideation continue to plague physicians: Survey
Now, as they bear the weight of a multiyear pandemic alongside the perpetual struggle to maintain some semblance of work-life balance, their resiliency has been stretched to the brink.
In 2022, the Medscape Physician Suicide Report surveyed more than 13,000 physicians in 29 specialties who were candid about their experiences with suicidal thoughts, how they support their besieged colleagues, and their go-to coping strategies.
Overall, 21% of physicians reported having feelings of depression. Of those, 24% had clinical depression and 64% had colloquial depression. Physicians who felt sad or blue decreased slightly, compared with the 2021 report, but the number of physicians experiencing severe depression rose 4%.
One in 10 physicians said they have thought about or attempted suicide. However, the number of physicians with suicidal thoughts dropped to 9%, down substantially from the 22% who reported similar feelings in 2020.
Still, there was a slight uptick in women physicians contemplating suicide, likely linked to their larger share of childcare and family responsibilities.
“They have needed to pull double duty even more than usual, and that may have increased their sense of burnout and vulnerability to suicidal thoughts,” said Andrea Giedinghagen, MD, assistant professor in the department of psychiatry at Washington University in St. Louis, and coauthor of “Physician Suicide: A Call to Action
Fighting the stigma of seeking mental health help
Although the number of physicians attempting, but not completing suicide, has remained steady at 1% for several years, the recent passage of the Dr. Lorna Breen Health Care Provider Protection Act by Congress aims to drive that figure even lower. Dr. Breen, an ED physician at New York–Presbyterian Hospital, died by suicide in April 2020. Overwhelmed by the onslaught of COVID patients, Dr. Breen was reluctant to seek mental health services for fear of being ostracized.
“Many physicians don’t seek mental health care due to fear of negative consequences in the workplace, including retribution, exclusion, loss of license, or even their job,” Gary Price, MD, president of The Physicians Foundation, told this news organization. “This was the experience of Dr. Lorna Breen. She was convinced that if she talked to a professional, she would lose her medical license. Perhaps if Dr. Breen was equipped with the accurate information – there is no mental health reporting requirement in her state’s medical license application – it might have saved her life.”
This same stigma was reflected in the survey, with one physician saying: “I’m afraid that if I spoke to a therapist, I’d have to report receiving psychiatric treatment to credentialing or licensing boards.” Roughly 40% of survey respondents, regardless of age, chose not to disclose their suicidal thoughts to anyone, not even a family member or suicide hotline. And just a tiny portion of physicians (10% of men and 13% of women) said that a colleague had discussed their suicidal thoughts with them.
“There is a longstanding culture of silence around physician mental health in the medical community,” said Dr. Price. “The strategies within the Act are critical to fixing this culture and making it acceptable and normalized for physicians to seek mental health care,” and for it to “become a fundamental and ongoing element of being a practicing physician.”
As part of the legislation, the Department of Health & Human Services must award grants to hospitals, medical associations, and other entities to facilitate mental health programs for providers. They must also establish policy recommendations and conduct campaigns to improve providers’ mental and behavioral health, encourage providers to seek mental health support and assistance, remove barriers to such treatment, and identify best practices to prevent suicide and promote resiliency
Addressing barriers to mental health
The new bill is a step in the right direction, but Dr. Price said health organizations must do more to address the six key structural barriers that are “discouraging physicians from seeking [mental health] help,” such as the inclusion of “intrusive mental health questions on medical board, hospital credentialing, and malpractice insurance applications.”
In addition, employers should allow physicians to seek out-of-network mental health services, if necessary, and not cause further humiliation by requiring them to be treated by colleagues within their hospital system. A similar proposal has recently been introduced and is gaining traction in Utah, following the suicide of ED physician Scott Jolley, MD, in 2021 after he was admitted for psychiatric care where he worked.
Diminishing the stigma surrounding physicians’ mental health encourages a more open dialogue, so if a colleague reaches out – listen. “Start by thanking the colleague for sharing the information: ‘I’m sure that wasn’t easy but I appreciate that you respect me enough to share this. Let’s talk more,’ ” said Michael F. Myers, MD, professor of clinical psychiatry at State University of New York, Brooklyn . “Then ask what you can do to help, which cuts down on the sense of isolation that colleague may feel.”
According to the survey, many physicians have developed strategies to support their happiness and mental health. Although fewer than 10% said reducing work hours or transitioning to a part-time schedule was most effective, the majority of physicians relied on spending time with family and friends (68%) – a choice that has considerable benefits.
“Close and intimate relationships are the single most protective factor for our mental health,” said Peter Yellowlees, MBBS, MD, chief wellness officer for UC Davis Health and professor of psychiatry at the University of California, Davis. “Isolation and loneliness are very important stressors, and we know that about 25% of the population reports being lonely.”
A version of this article first appeared on Medscape.com.
Now, as they bear the weight of a multiyear pandemic alongside the perpetual struggle to maintain some semblance of work-life balance, their resiliency has been stretched to the brink.
In 2022, the Medscape Physician Suicide Report surveyed more than 13,000 physicians in 29 specialties who were candid about their experiences with suicidal thoughts, how they support their besieged colleagues, and their go-to coping strategies.
Overall, 21% of physicians reported having feelings of depression. Of those, 24% had clinical depression and 64% had colloquial depression. Physicians who felt sad or blue decreased slightly, compared with the 2021 report, but the number of physicians experiencing severe depression rose 4%.
One in 10 physicians said they have thought about or attempted suicide. However, the number of physicians with suicidal thoughts dropped to 9%, down substantially from the 22% who reported similar feelings in 2020.
Still, there was a slight uptick in women physicians contemplating suicide, likely linked to their larger share of childcare and family responsibilities.
“They have needed to pull double duty even more than usual, and that may have increased their sense of burnout and vulnerability to suicidal thoughts,” said Andrea Giedinghagen, MD, assistant professor in the department of psychiatry at Washington University in St. Louis, and coauthor of “Physician Suicide: A Call to Action
Fighting the stigma of seeking mental health help
Although the number of physicians attempting, but not completing suicide, has remained steady at 1% for several years, the recent passage of the Dr. Lorna Breen Health Care Provider Protection Act by Congress aims to drive that figure even lower. Dr. Breen, an ED physician at New York–Presbyterian Hospital, died by suicide in April 2020. Overwhelmed by the onslaught of COVID patients, Dr. Breen was reluctant to seek mental health services for fear of being ostracized.
“Many physicians don’t seek mental health care due to fear of negative consequences in the workplace, including retribution, exclusion, loss of license, or even their job,” Gary Price, MD, president of The Physicians Foundation, told this news organization. “This was the experience of Dr. Lorna Breen. She was convinced that if she talked to a professional, she would lose her medical license. Perhaps if Dr. Breen was equipped with the accurate information – there is no mental health reporting requirement in her state’s medical license application – it might have saved her life.”
This same stigma was reflected in the survey, with one physician saying: “I’m afraid that if I spoke to a therapist, I’d have to report receiving psychiatric treatment to credentialing or licensing boards.” Roughly 40% of survey respondents, regardless of age, chose not to disclose their suicidal thoughts to anyone, not even a family member or suicide hotline. And just a tiny portion of physicians (10% of men and 13% of women) said that a colleague had discussed their suicidal thoughts with them.
“There is a longstanding culture of silence around physician mental health in the medical community,” said Dr. Price. “The strategies within the Act are critical to fixing this culture and making it acceptable and normalized for physicians to seek mental health care,” and for it to “become a fundamental and ongoing element of being a practicing physician.”
As part of the legislation, the Department of Health & Human Services must award grants to hospitals, medical associations, and other entities to facilitate mental health programs for providers. They must also establish policy recommendations and conduct campaigns to improve providers’ mental and behavioral health, encourage providers to seek mental health support and assistance, remove barriers to such treatment, and identify best practices to prevent suicide and promote resiliency
Addressing barriers to mental health
The new bill is a step in the right direction, but Dr. Price said health organizations must do more to address the six key structural barriers that are “discouraging physicians from seeking [mental health] help,” such as the inclusion of “intrusive mental health questions on medical board, hospital credentialing, and malpractice insurance applications.”
In addition, employers should allow physicians to seek out-of-network mental health services, if necessary, and not cause further humiliation by requiring them to be treated by colleagues within their hospital system. A similar proposal has recently been introduced and is gaining traction in Utah, following the suicide of ED physician Scott Jolley, MD, in 2021 after he was admitted for psychiatric care where he worked.
Diminishing the stigma surrounding physicians’ mental health encourages a more open dialogue, so if a colleague reaches out – listen. “Start by thanking the colleague for sharing the information: ‘I’m sure that wasn’t easy but I appreciate that you respect me enough to share this. Let’s talk more,’ ” said Michael F. Myers, MD, professor of clinical psychiatry at State University of New York, Brooklyn . “Then ask what you can do to help, which cuts down on the sense of isolation that colleague may feel.”
According to the survey, many physicians have developed strategies to support their happiness and mental health. Although fewer than 10% said reducing work hours or transitioning to a part-time schedule was most effective, the majority of physicians relied on spending time with family and friends (68%) – a choice that has considerable benefits.
“Close and intimate relationships are the single most protective factor for our mental health,” said Peter Yellowlees, MBBS, MD, chief wellness officer for UC Davis Health and professor of psychiatry at the University of California, Davis. “Isolation and loneliness are very important stressors, and we know that about 25% of the population reports being lonely.”
A version of this article first appeared on Medscape.com.
Now, as they bear the weight of a multiyear pandemic alongside the perpetual struggle to maintain some semblance of work-life balance, their resiliency has been stretched to the brink.
In 2022, the Medscape Physician Suicide Report surveyed more than 13,000 physicians in 29 specialties who were candid about their experiences with suicidal thoughts, how they support their besieged colleagues, and their go-to coping strategies.
Overall, 21% of physicians reported having feelings of depression. Of those, 24% had clinical depression and 64% had colloquial depression. Physicians who felt sad or blue decreased slightly, compared with the 2021 report, but the number of physicians experiencing severe depression rose 4%.
One in 10 physicians said they have thought about or attempted suicide. However, the number of physicians with suicidal thoughts dropped to 9%, down substantially from the 22% who reported similar feelings in 2020.
Still, there was a slight uptick in women physicians contemplating suicide, likely linked to their larger share of childcare and family responsibilities.
“They have needed to pull double duty even more than usual, and that may have increased their sense of burnout and vulnerability to suicidal thoughts,” said Andrea Giedinghagen, MD, assistant professor in the department of psychiatry at Washington University in St. Louis, and coauthor of “Physician Suicide: A Call to Action
Fighting the stigma of seeking mental health help
Although the number of physicians attempting, but not completing suicide, has remained steady at 1% for several years, the recent passage of the Dr. Lorna Breen Health Care Provider Protection Act by Congress aims to drive that figure even lower. Dr. Breen, an ED physician at New York–Presbyterian Hospital, died by suicide in April 2020. Overwhelmed by the onslaught of COVID patients, Dr. Breen was reluctant to seek mental health services for fear of being ostracized.
“Many physicians don’t seek mental health care due to fear of negative consequences in the workplace, including retribution, exclusion, loss of license, or even their job,” Gary Price, MD, president of The Physicians Foundation, told this news organization. “This was the experience of Dr. Lorna Breen. She was convinced that if she talked to a professional, she would lose her medical license. Perhaps if Dr. Breen was equipped with the accurate information – there is no mental health reporting requirement in her state’s medical license application – it might have saved her life.”
This same stigma was reflected in the survey, with one physician saying: “I’m afraid that if I spoke to a therapist, I’d have to report receiving psychiatric treatment to credentialing or licensing boards.” Roughly 40% of survey respondents, regardless of age, chose not to disclose their suicidal thoughts to anyone, not even a family member or suicide hotline. And just a tiny portion of physicians (10% of men and 13% of women) said that a colleague had discussed their suicidal thoughts with them.
“There is a longstanding culture of silence around physician mental health in the medical community,” said Dr. Price. “The strategies within the Act are critical to fixing this culture and making it acceptable and normalized for physicians to seek mental health care,” and for it to “become a fundamental and ongoing element of being a practicing physician.”
As part of the legislation, the Department of Health & Human Services must award grants to hospitals, medical associations, and other entities to facilitate mental health programs for providers. They must also establish policy recommendations and conduct campaigns to improve providers’ mental and behavioral health, encourage providers to seek mental health support and assistance, remove barriers to such treatment, and identify best practices to prevent suicide and promote resiliency
Addressing barriers to mental health
The new bill is a step in the right direction, but Dr. Price said health organizations must do more to address the six key structural barriers that are “discouraging physicians from seeking [mental health] help,” such as the inclusion of “intrusive mental health questions on medical board, hospital credentialing, and malpractice insurance applications.”
In addition, employers should allow physicians to seek out-of-network mental health services, if necessary, and not cause further humiliation by requiring them to be treated by colleagues within their hospital system. A similar proposal has recently been introduced and is gaining traction in Utah, following the suicide of ED physician Scott Jolley, MD, in 2021 after he was admitted for psychiatric care where he worked.
Diminishing the stigma surrounding physicians’ mental health encourages a more open dialogue, so if a colleague reaches out – listen. “Start by thanking the colleague for sharing the information: ‘I’m sure that wasn’t easy but I appreciate that you respect me enough to share this. Let’s talk more,’ ” said Michael F. Myers, MD, professor of clinical psychiatry at State University of New York, Brooklyn . “Then ask what you can do to help, which cuts down on the sense of isolation that colleague may feel.”
According to the survey, many physicians have developed strategies to support their happiness and mental health. Although fewer than 10% said reducing work hours or transitioning to a part-time schedule was most effective, the majority of physicians relied on spending time with family and friends (68%) – a choice that has considerable benefits.
“Close and intimate relationships are the single most protective factor for our mental health,” said Peter Yellowlees, MBBS, MD, chief wellness officer for UC Davis Health and professor of psychiatry at the University of California, Davis. “Isolation and loneliness are very important stressors, and we know that about 25% of the population reports being lonely.”
A version of this article first appeared on Medscape.com.
Is a progression-free survival benefit alone really worth $10,000 a month?
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.
Proper steps for physicians to follow if they find themselves under investigation
Physician clients will find themselves in difficult legal situations from time to time. Sometimes it’s an investigation for Medicare fraud or other illegal conduct. Other times it’s a review related to Drug Enforcement Administration or licensure compliance. More commonly, physicians are involved in employer inquiries into workplace misconduct.
but how they choose to deal with the issue can have significant consequences.
In my opinion, physicians should have a relationship with a health care lawyer or firm in place before any investigation occurs. Whether they are being investigated for a license or medical staff issue, Medicare fraud, or contract issue, it’s important to know where to go for help quickly. Even if the physician does not retain a lawyer in advance, having the name of a qualified person who can be called for a variety of health care issues is already a step in the right direction.
More important than having a knowledgeable lawyer is actually contacting that lawyer. Some physicians will sit and chat with the Federal Bureau of Investigation or other investigators for hours, only to call me after the visitors leave. I have other clients who handle important medical staff hearings, discipline meetings, and license investigations on their own without consulting counsel first. In all of these situations, it can be too late to help a physician once their case has progressed too far down the road.
Employment issues arising in the workplace setting are the most common and troubling. Physicians will – without a second thought – attend a human resources–called or other meeting without thinking through the reason for the meeting, whether they are prepared or not, and without considering whether counsel could be helpful. Sometimes in the moment, there may be no choice, but most meetings are scheduled in advance with ample time for consultation and planning.
Many issues that arise in the workplace setting are troubling because they can be easily avoided. The No. 1 piece of advice which I offer to young physician clients as they enter the workplace is: Remember that nobody in the workplace is your friend. Every word that is said, text that is sent, gesture that is made, can put you at risk. You must assume that all conversations and messages will be shared with others. Joking around in the operating room about sexual escapades, sending texts with flirtatious comments, making comments that can be construed as racist or homophobic, or raising your voice in a moment of frustration are all real examples of situations where physicians ended up disciplined and terminated. Are these innocent comments or ones the doctor thought they could get away with among “friends?” From a human resources perspective, there is little tolerance for such conduct, regardless of the doctor’s intent.
There are also situations in the workplace that are more troubling. Many times a physician is accused of noncompliance with a contract or a policy, when in fact the accuser is retaliating or engaging in efforts to discredit a doctor. I have seen this happen where minority physicians complain about how they are treated and are suddenly investigated for a performance issue. I have had female physicians criticize a business decision at a committee meeting, only to receive a formal notice that their “negative attitude” violated a policy.
In these situations, talking with counsel before a meeting with the employer representative is recommended and can impact the trajectory of a physician’s career. Physicians cannot and should not handle such events on their own.
If a physician is forced or chooses to attend a meeting with an investigator or other party without counsel, there are some steps to consider (subject to the type of meeting and the specific circumstances).
- Listen more than you talk. Make sure you know the name of everyone who is present and their role within the organization.
- If you have previously provided any written or oral statements, or have written correspondence related to the issues at hand, review all materials in advance. If there is anything you think needs to be corrected or added, let the interviewer know that at the outset.
- Be familiar with your own employment agreement/policies and the terms that may be relevant to the discussion or meeting.
- Be calm, honest, and forthcoming in response to the questions, and don’t embellish or exaggerate.
- Avoid personal attacks on anyone. This generally serves to weaken an argument and credibility.
- Be prepared to explain your allegations or defense, and when you do so, keep in mind that the interviewer may not know the history, background, or details of any of the issues.
- If the reason for the situation relates to race or national origin, age, gender, sexual orientation, disability, or other protected category, don’t hesitate to say so.
- Answer the question you’re asked, but if you feel that the interviewer needs more information or is not understanding what you’ve said, feel free to explain. Be forthcoming, but don’t dominate the conversation.
- If they ask whether you have counsel, be honest, but decline to provide them any information about what you discussed with counsel, as those conversations are privileged.
- If the interviewer asks to record the conversation, you can agree, but ask to be provided a copy of the recording.
- Know your rights in advance. If the subject of the meeting is governed by bylaws or policies, for example, you may have the right to bring an attorney or adviser to the meeting, receive advance notice of who will be attending the meeting and the subject matter, and avail yourself of specific procedures or appeal rights of any discipline or decisions decided during the meeting.
There are many circumstances that can lead to a physician being under investigation or interrogation. In every single circumstance, it is ideal to seek legal counsel immediately. Whether the physician has actually engaged in wrongful conduct or not, without proper handling a physician’s career can be permanently, and sometimes irrevocably, affected.
Ms. Adler is a shareholder and health law practice group manager for Chicago-based law firm Roetzel, a member of the Illinois Association of Healthcare Attorneys, and a current advisory board member at DePaul College of Law Health Law Institute. She disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physician clients will find themselves in difficult legal situations from time to time. Sometimes it’s an investigation for Medicare fraud or other illegal conduct. Other times it’s a review related to Drug Enforcement Administration or licensure compliance. More commonly, physicians are involved in employer inquiries into workplace misconduct.
but how they choose to deal with the issue can have significant consequences.
In my opinion, physicians should have a relationship with a health care lawyer or firm in place before any investigation occurs. Whether they are being investigated for a license or medical staff issue, Medicare fraud, or contract issue, it’s important to know where to go for help quickly. Even if the physician does not retain a lawyer in advance, having the name of a qualified person who can be called for a variety of health care issues is already a step in the right direction.
More important than having a knowledgeable lawyer is actually contacting that lawyer. Some physicians will sit and chat with the Federal Bureau of Investigation or other investigators for hours, only to call me after the visitors leave. I have other clients who handle important medical staff hearings, discipline meetings, and license investigations on their own without consulting counsel first. In all of these situations, it can be too late to help a physician once their case has progressed too far down the road.
Employment issues arising in the workplace setting are the most common and troubling. Physicians will – without a second thought – attend a human resources–called or other meeting without thinking through the reason for the meeting, whether they are prepared or not, and without considering whether counsel could be helpful. Sometimes in the moment, there may be no choice, but most meetings are scheduled in advance with ample time for consultation and planning.
Many issues that arise in the workplace setting are troubling because they can be easily avoided. The No. 1 piece of advice which I offer to young physician clients as they enter the workplace is: Remember that nobody in the workplace is your friend. Every word that is said, text that is sent, gesture that is made, can put you at risk. You must assume that all conversations and messages will be shared with others. Joking around in the operating room about sexual escapades, sending texts with flirtatious comments, making comments that can be construed as racist or homophobic, or raising your voice in a moment of frustration are all real examples of situations where physicians ended up disciplined and terminated. Are these innocent comments or ones the doctor thought they could get away with among “friends?” From a human resources perspective, there is little tolerance for such conduct, regardless of the doctor’s intent.
There are also situations in the workplace that are more troubling. Many times a physician is accused of noncompliance with a contract or a policy, when in fact the accuser is retaliating or engaging in efforts to discredit a doctor. I have seen this happen where minority physicians complain about how they are treated and are suddenly investigated for a performance issue. I have had female physicians criticize a business decision at a committee meeting, only to receive a formal notice that their “negative attitude” violated a policy.
In these situations, talking with counsel before a meeting with the employer representative is recommended and can impact the trajectory of a physician’s career. Physicians cannot and should not handle such events on their own.
If a physician is forced or chooses to attend a meeting with an investigator or other party without counsel, there are some steps to consider (subject to the type of meeting and the specific circumstances).
- Listen more than you talk. Make sure you know the name of everyone who is present and their role within the organization.
- If you have previously provided any written or oral statements, or have written correspondence related to the issues at hand, review all materials in advance. If there is anything you think needs to be corrected or added, let the interviewer know that at the outset.
- Be familiar with your own employment agreement/policies and the terms that may be relevant to the discussion or meeting.
- Be calm, honest, and forthcoming in response to the questions, and don’t embellish or exaggerate.
- Avoid personal attacks on anyone. This generally serves to weaken an argument and credibility.
- Be prepared to explain your allegations or defense, and when you do so, keep in mind that the interviewer may not know the history, background, or details of any of the issues.
- If the reason for the situation relates to race or national origin, age, gender, sexual orientation, disability, or other protected category, don’t hesitate to say so.
- Answer the question you’re asked, but if you feel that the interviewer needs more information or is not understanding what you’ve said, feel free to explain. Be forthcoming, but don’t dominate the conversation.
- If they ask whether you have counsel, be honest, but decline to provide them any information about what you discussed with counsel, as those conversations are privileged.
- If the interviewer asks to record the conversation, you can agree, but ask to be provided a copy of the recording.
- Know your rights in advance. If the subject of the meeting is governed by bylaws or policies, for example, you may have the right to bring an attorney or adviser to the meeting, receive advance notice of who will be attending the meeting and the subject matter, and avail yourself of specific procedures or appeal rights of any discipline or decisions decided during the meeting.
There are many circumstances that can lead to a physician being under investigation or interrogation. In every single circumstance, it is ideal to seek legal counsel immediately. Whether the physician has actually engaged in wrongful conduct or not, without proper handling a physician’s career can be permanently, and sometimes irrevocably, affected.
Ms. Adler is a shareholder and health law practice group manager for Chicago-based law firm Roetzel, a member of the Illinois Association of Healthcare Attorneys, and a current advisory board member at DePaul College of Law Health Law Institute. She disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physician clients will find themselves in difficult legal situations from time to time. Sometimes it’s an investigation for Medicare fraud or other illegal conduct. Other times it’s a review related to Drug Enforcement Administration or licensure compliance. More commonly, physicians are involved in employer inquiries into workplace misconduct.
but how they choose to deal with the issue can have significant consequences.
In my opinion, physicians should have a relationship with a health care lawyer or firm in place before any investigation occurs. Whether they are being investigated for a license or medical staff issue, Medicare fraud, or contract issue, it’s important to know where to go for help quickly. Even if the physician does not retain a lawyer in advance, having the name of a qualified person who can be called for a variety of health care issues is already a step in the right direction.
More important than having a knowledgeable lawyer is actually contacting that lawyer. Some physicians will sit and chat with the Federal Bureau of Investigation or other investigators for hours, only to call me after the visitors leave. I have other clients who handle important medical staff hearings, discipline meetings, and license investigations on their own without consulting counsel first. In all of these situations, it can be too late to help a physician once their case has progressed too far down the road.
Employment issues arising in the workplace setting are the most common and troubling. Physicians will – without a second thought – attend a human resources–called or other meeting without thinking through the reason for the meeting, whether they are prepared or not, and without considering whether counsel could be helpful. Sometimes in the moment, there may be no choice, but most meetings are scheduled in advance with ample time for consultation and planning.
Many issues that arise in the workplace setting are troubling because they can be easily avoided. The No. 1 piece of advice which I offer to young physician clients as they enter the workplace is: Remember that nobody in the workplace is your friend. Every word that is said, text that is sent, gesture that is made, can put you at risk. You must assume that all conversations and messages will be shared with others. Joking around in the operating room about sexual escapades, sending texts with flirtatious comments, making comments that can be construed as racist or homophobic, or raising your voice in a moment of frustration are all real examples of situations where physicians ended up disciplined and terminated. Are these innocent comments or ones the doctor thought they could get away with among “friends?” From a human resources perspective, there is little tolerance for such conduct, regardless of the doctor’s intent.
There are also situations in the workplace that are more troubling. Many times a physician is accused of noncompliance with a contract or a policy, when in fact the accuser is retaliating or engaging in efforts to discredit a doctor. I have seen this happen where minority physicians complain about how they are treated and are suddenly investigated for a performance issue. I have had female physicians criticize a business decision at a committee meeting, only to receive a formal notice that their “negative attitude” violated a policy.
In these situations, talking with counsel before a meeting with the employer representative is recommended and can impact the trajectory of a physician’s career. Physicians cannot and should not handle such events on their own.
If a physician is forced or chooses to attend a meeting with an investigator or other party without counsel, there are some steps to consider (subject to the type of meeting and the specific circumstances).
- Listen more than you talk. Make sure you know the name of everyone who is present and their role within the organization.
- If you have previously provided any written or oral statements, or have written correspondence related to the issues at hand, review all materials in advance. If there is anything you think needs to be corrected or added, let the interviewer know that at the outset.
- Be familiar with your own employment agreement/policies and the terms that may be relevant to the discussion or meeting.
- Be calm, honest, and forthcoming in response to the questions, and don’t embellish or exaggerate.
- Avoid personal attacks on anyone. This generally serves to weaken an argument and credibility.
- Be prepared to explain your allegations or defense, and when you do so, keep in mind that the interviewer may not know the history, background, or details of any of the issues.
- If the reason for the situation relates to race or national origin, age, gender, sexual orientation, disability, or other protected category, don’t hesitate to say so.
- Answer the question you’re asked, but if you feel that the interviewer needs more information or is not understanding what you’ve said, feel free to explain. Be forthcoming, but don’t dominate the conversation.
- If they ask whether you have counsel, be honest, but decline to provide them any information about what you discussed with counsel, as those conversations are privileged.
- If the interviewer asks to record the conversation, you can agree, but ask to be provided a copy of the recording.
- Know your rights in advance. If the subject of the meeting is governed by bylaws or policies, for example, you may have the right to bring an attorney or adviser to the meeting, receive advance notice of who will be attending the meeting and the subject matter, and avail yourself of specific procedures or appeal rights of any discipline or decisions decided during the meeting.
There are many circumstances that can lead to a physician being under investigation or interrogation. In every single circumstance, it is ideal to seek legal counsel immediately. Whether the physician has actually engaged in wrongful conduct or not, without proper handling a physician’s career can be permanently, and sometimes irrevocably, affected.
Ms. Adler is a shareholder and health law practice group manager for Chicago-based law firm Roetzel, a member of the Illinois Association of Healthcare Attorneys, and a current advisory board member at DePaul College of Law Health Law Institute. She disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Self-care tips for clinicians as COVID-19 lingers
LAS VEGAS – according to Jon A. Levenson, MD.
“There are those who will need mental health treatment, so creating an easy way to reach out for help and facilitate linkage with care is critically important,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “The vast majority of our workforce will thrive with proper support. But what can each of us do to take care of ourselves?”
Step one is to recognize common stress reactions as well as signs of distress. He offered the oxygen mask metaphor, the idea that before we can take care of and support anyone else, we must first take care of ourselves. “When people are stressed, they don’t always think about the oxygen mask metaphor,” Dr. Levenson said. Step two is to practice and model self-care by adopting principles often discussed in acceptance and commitment therapy: to focus on what you can control, not on what you can’t control.
“We can’t control the amount of toilet paper at the grocery store, how long the pandemic will last, or how others have reacted,” Dr. Levenson said. “We also can’t control other people’s motives, predict what will happen, or the actions of others, including whether they will follow social distancing guidelines or not.”
How about what we can control? One is a positive attitude, “which can sustain people during times of intense stress,” he said. “Other things that we can do include turn off the news and find fun and enriching activities to do at home, whether it be playing a game with family or reaching out to friends through an iPad or a smartphone. You can also follow [Centers for Disease Control and Prevention] recommendations, control your own social distancing, and limit social media activity, which can be stressful. We can also control our kindness and grace.” He added that resilience does not mean “snapping back” to how you were before the pandemic, but rather “learning to integrate the adverse experiences into who you are and growing with them, which is sometimes known as posttraumatic growth.”
Dr. Levenson encouraged health care workers to use their coping resources, connect to others, and cultivate their values and purpose in life as they navigate these challenging times. “You also want to promote realistic optimism; find a way to stay positive,” he said. “We emphasize to our staff that while you won’t forget this time, focus on what you can control – your positive relationships – and remind yourself of your values and sources of gratitude. Figure out, and reflect on, what you care about, and then care about it. Remind yourself in a deliberate, purposeful way what anchors you to your job, which in the health care setting tends to be a desire to care for others, to assist those in need, and to work in teams. We also encourage staff to refrain from judgment. Guilt is a normal and near-universal response to this stressor, but there are many ways to contribute without a judgmental or guilty tone.”
Other tips for self-support are to remind yourself that it is not selfish to take breaks. “The needs of your patients are not more important than your own needs,” Dr. Levenson said. “Working nonstop can put you at higher risk for stress, exhaustion, and illness. You may need to give yourself more time to step back and recover from workplace challenges or extended coverage for peers; this is important. We remind our staff that your work may feel more emotionally draining than usual because everything is more intense overall during the COVID-19 pandemic. This reminder helps staff normalize what they already may be experiencing, and in turn, to further support each other.”
Soothing activities to relieve stress include meditation, prayer, deep and slow breathing, relaxation exercises, yoga, mindfulness, stretching, staying hydrated, eating healthfully, exercise, and getting sufficient sleep. Other stress management tips include avoiding excessive alcohol intake, reaching out to others, asking for assistance, and delegating when possible. “We want to promote psychological flexibility: the ability to stay in contact with the present moment,” he said. “We encourage our peers to be aware of unpleasant thoughts and feelings, and to try to redirect negative thought patterns to a proactive problem-solving approach; this includes choosing one’s behaviors based on the situation and personal values.”
Dr. Levenson reported having no disclosures related to his presentation.
LAS VEGAS – according to Jon A. Levenson, MD.
“There are those who will need mental health treatment, so creating an easy way to reach out for help and facilitate linkage with care is critically important,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “The vast majority of our workforce will thrive with proper support. But what can each of us do to take care of ourselves?”
Step one is to recognize common stress reactions as well as signs of distress. He offered the oxygen mask metaphor, the idea that before we can take care of and support anyone else, we must first take care of ourselves. “When people are stressed, they don’t always think about the oxygen mask metaphor,” Dr. Levenson said. Step two is to practice and model self-care by adopting principles often discussed in acceptance and commitment therapy: to focus on what you can control, not on what you can’t control.
“We can’t control the amount of toilet paper at the grocery store, how long the pandemic will last, or how others have reacted,” Dr. Levenson said. “We also can’t control other people’s motives, predict what will happen, or the actions of others, including whether they will follow social distancing guidelines or not.”
How about what we can control? One is a positive attitude, “which can sustain people during times of intense stress,” he said. “Other things that we can do include turn off the news and find fun and enriching activities to do at home, whether it be playing a game with family or reaching out to friends through an iPad or a smartphone. You can also follow [Centers for Disease Control and Prevention] recommendations, control your own social distancing, and limit social media activity, which can be stressful. We can also control our kindness and grace.” He added that resilience does not mean “snapping back” to how you were before the pandemic, but rather “learning to integrate the adverse experiences into who you are and growing with them, which is sometimes known as posttraumatic growth.”
Dr. Levenson encouraged health care workers to use their coping resources, connect to others, and cultivate their values and purpose in life as they navigate these challenging times. “You also want to promote realistic optimism; find a way to stay positive,” he said. “We emphasize to our staff that while you won’t forget this time, focus on what you can control – your positive relationships – and remind yourself of your values and sources of gratitude. Figure out, and reflect on, what you care about, and then care about it. Remind yourself in a deliberate, purposeful way what anchors you to your job, which in the health care setting tends to be a desire to care for others, to assist those in need, and to work in teams. We also encourage staff to refrain from judgment. Guilt is a normal and near-universal response to this stressor, but there are many ways to contribute without a judgmental or guilty tone.”
Other tips for self-support are to remind yourself that it is not selfish to take breaks. “The needs of your patients are not more important than your own needs,” Dr. Levenson said. “Working nonstop can put you at higher risk for stress, exhaustion, and illness. You may need to give yourself more time to step back and recover from workplace challenges or extended coverage for peers; this is important. We remind our staff that your work may feel more emotionally draining than usual because everything is more intense overall during the COVID-19 pandemic. This reminder helps staff normalize what they already may be experiencing, and in turn, to further support each other.”
Soothing activities to relieve stress include meditation, prayer, deep and slow breathing, relaxation exercises, yoga, mindfulness, stretching, staying hydrated, eating healthfully, exercise, and getting sufficient sleep. Other stress management tips include avoiding excessive alcohol intake, reaching out to others, asking for assistance, and delegating when possible. “We want to promote psychological flexibility: the ability to stay in contact with the present moment,” he said. “We encourage our peers to be aware of unpleasant thoughts and feelings, and to try to redirect negative thought patterns to a proactive problem-solving approach; this includes choosing one’s behaviors based on the situation and personal values.”
Dr. Levenson reported having no disclosures related to his presentation.
LAS VEGAS – according to Jon A. Levenson, MD.
“There are those who will need mental health treatment, so creating an easy way to reach out for help and facilitate linkage with care is critically important,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “The vast majority of our workforce will thrive with proper support. But what can each of us do to take care of ourselves?”
Step one is to recognize common stress reactions as well as signs of distress. He offered the oxygen mask metaphor, the idea that before we can take care of and support anyone else, we must first take care of ourselves. “When people are stressed, they don’t always think about the oxygen mask metaphor,” Dr. Levenson said. Step two is to practice and model self-care by adopting principles often discussed in acceptance and commitment therapy: to focus on what you can control, not on what you can’t control.
“We can’t control the amount of toilet paper at the grocery store, how long the pandemic will last, or how others have reacted,” Dr. Levenson said. “We also can’t control other people’s motives, predict what will happen, or the actions of others, including whether they will follow social distancing guidelines or not.”
How about what we can control? One is a positive attitude, “which can sustain people during times of intense stress,” he said. “Other things that we can do include turn off the news and find fun and enriching activities to do at home, whether it be playing a game with family or reaching out to friends through an iPad or a smartphone. You can also follow [Centers for Disease Control and Prevention] recommendations, control your own social distancing, and limit social media activity, which can be stressful. We can also control our kindness and grace.” He added that resilience does not mean “snapping back” to how you were before the pandemic, but rather “learning to integrate the adverse experiences into who you are and growing with them, which is sometimes known as posttraumatic growth.”
Dr. Levenson encouraged health care workers to use their coping resources, connect to others, and cultivate their values and purpose in life as they navigate these challenging times. “You also want to promote realistic optimism; find a way to stay positive,” he said. “We emphasize to our staff that while you won’t forget this time, focus on what you can control – your positive relationships – and remind yourself of your values and sources of gratitude. Figure out, and reflect on, what you care about, and then care about it. Remind yourself in a deliberate, purposeful way what anchors you to your job, which in the health care setting tends to be a desire to care for others, to assist those in need, and to work in teams. We also encourage staff to refrain from judgment. Guilt is a normal and near-universal response to this stressor, but there are many ways to contribute without a judgmental or guilty tone.”
Other tips for self-support are to remind yourself that it is not selfish to take breaks. “The needs of your patients are not more important than your own needs,” Dr. Levenson said. “Working nonstop can put you at higher risk for stress, exhaustion, and illness. You may need to give yourself more time to step back and recover from workplace challenges or extended coverage for peers; this is important. We remind our staff that your work may feel more emotionally draining than usual because everything is more intense overall during the COVID-19 pandemic. This reminder helps staff normalize what they already may be experiencing, and in turn, to further support each other.”
Soothing activities to relieve stress include meditation, prayer, deep and slow breathing, relaxation exercises, yoga, mindfulness, stretching, staying hydrated, eating healthfully, exercise, and getting sufficient sleep. Other stress management tips include avoiding excessive alcohol intake, reaching out to others, asking for assistance, and delegating when possible. “We want to promote psychological flexibility: the ability to stay in contact with the present moment,” he said. “We encourage our peers to be aware of unpleasant thoughts and feelings, and to try to redirect negative thought patterns to a proactive problem-solving approach; this includes choosing one’s behaviors based on the situation and personal values.”
Dr. Levenson reported having no disclosures related to his presentation.
AT NPA 2022








