User login
Low serum LDH shows potential as depression biomarker
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM GENERAL HOSPITAL PSYCHIATRY
Mood disorder? Assessment in primary care
The assessment and diagnosis of bipolar disorder in youth has a complicated and controversial history. I recall from my child and adolescent fellowship training that there was a thinly veiled faculty argument about the diagnosis itself with strong opinions on each side. To revisit this quandary, I reviewed the most up-to-date literature and outlined a case-based approach to the initial screening assessment. Certainly, the assessment by a child and adolescent psychiatrist would be the standard for diagnosis, but we do know that the pediatrician’s office may be the first setting for a child and parent to present with mood symptoms and concerns about bipolar disorder. What can you do to address this adolescent, Carrie, and her mother’s concerns?
Case
Carrie is a 17-year-old girl who has struggled through her childhood and adolescence with anxious and depressive symptoms which have ebbed and flowed with major life stressors, including her parent’s divorce. She has tried cognitive-behavioral therapy and selective serotonin reuptake inhibitors, but the SSRI seemed to cause feelings of anxiousness and agitation, so she stopped it within weeks.
Her mother presents to you concerned that Carrie has had a more persistently irritable mood toward her, often just wanting to be with her friends or otherwise isolate in her room when home to study.
Most concerning to her mother is that Carrie, as a straight A student, has also developed a pattern of staying up all night to study for tests and then “crashes” and sleeps through the weekend, avoiding her mother and only brightening with her friends.
To complicate matters, Carrie’s biological father had type 1 bipolar disorder and an addiction. Her mother comes to you with an initially nonparticipatory Carrie in tow and says: “My former husband began his manic episodes with a lack of sleep and Carrie is so irritable towards me. I feel like I am walking on eggshells all the time. Could this be bipolar disorder?”
Case discussion
First, it’s always useful to frame a visit stating that you will spend some time with the patient and some time with both the patient and parent. Emphasizing confidentiality about issues such as drug use, which can be comorbid with mood symptoms and go undetected in high-achieving students such as Carrie, is also important. Further emphasizing that information will not be reflexively shared with the parent unless the child presents a danger to herself or others is also paramount to receive an honest report of symptoms.
Second, there are many signs and symptoms of bipolar disorder that naturally overlap with other conditions such as distractibility with attention-deficit/hyperactivity disorder, or irritability in either a unipolar depression or disruptive mood dysregulation disorder.1 You are looking for an episodic (not chronic) course of symptoms with episodes that last over 5 days for hypomania and over the course of weeks for mania all while meeting all the classic criteria for bipolar disorder.
Note that the broadening of diagnostic criteria has been thought to contribute to an inflated sense of prevalence. The actual expert estimate of prevalence is around 0.8%-1.8% in pediatric populations, although there is a large published range depending on whether the criteria are modified or not.2 Use of the unmodified criteria from the DSM-5 is the recommended approach. Bipolar disorder is exceedingly rare in prepubertal children, and it would be more common for prodromal symptoms such as Carrie’s to emerge and escalate over the teenage years, culminating in a clearer diagnosis in the later teens or 20s.3
In my screening questions, I find the idea of an “infatiguable state” is the most pathognomonic one in considering mania in bipolar disorder.4 Carrie’s “crashing” after nights of studying shows that she clearly fatigues. Patients with bipolar disorder within episodes of hypomania or mania have a seismic shift in perceived energy and a matching lack of ability to sleep that can affect their thought processes, speech, and decision-making. At first blush, Carrie’s history does not indicate current symptoms of bipolar disorder.3
Case, continued
When you meet with Carrie alone she shares that she has been experimenting with prescribed stimulants from her older college-aged brother in order to study and ace her tests. She is also experimenting with alcohol and marijuana with her friends. You provide her the CRAFFT tool to deepen your screening of this issue.5
With her mother, you administer the Parent General Behavior Inventory6 and the and the Child Mania Rating Scale7. From these scales, you note that the irritability is more specific to Carrie’s family than pan-present in school and with friends. Her lack of sleep occurs at high-pressure and discreet times.
At this point, you reassure Carrie and her mother that Carrie does not present with symptoms of bipolar disorder but that certainly you will continue screening assessments over time, as they are a good means to track symptoms. You also recommend that Carrie consider mood tracking so she can develop insights into her mood and its relationship to sleep and other events as she prepares for college.8
Case discussion, continued
The strongest risk factor for bipolar disorder in youth is family history (specifically a parent) with bipolar disorder).9 If there is the chance to explore the parent’s illness with open-ended questions, you will want to hear about the parent’s age of symptom onset, course of treatment, any hospitalizations, and stabilizing medications because this has prognostic power for your patient. It is important to ensure that the parent indeed has a diagnosis of bipolar disorder and that it is not just being used colloquially to characterize an adult who has labile moods from hour to hour or day to day. This would give undue anticipatory anxiety to a youth about their risk, which is up to 8- to 10-fold greater with a parent with bipolar disorder.9
Even with a strong family history, we do not often see bipolar disorder emerge in prepubertal children.10,11 There may be still concerning prodromal symptoms in which a diagnosis of unipolar depression with more irritable features and mood lability seems more commonly complicated by substance use, as with Carrie.
Activation with an SSRI, as in Carrie’s case, even if not resulting in full mania or hypomania, can also be a soft sign of the serotonergic sensitivity present in bipolar disorder. However, if there are not additional symptoms of bipolar disorder and you are concerned based on family history alone, you do not want to withhold antidepressant treatment because fear of risk. You would want to consider a “dose low and go slow” titration process with more frequent monitoring.
A diagnostic interview with a child and adolescent psychiatrist and administration of scales such as the Young Mania Rating Scale and the Modified Child Depression Rating Scale are the standard means to assess for bipolar symptoms.12 Considering the dearth of child psychiatrists nationally, it would be useful to improve one’s screening in primary care so as to not inadvertently “refer out” all patients for whom mood dysregulation is a concern.
There is also a more expanded tool that includes several scales integrated with clinical information (parent’s age of mood disorder onset, child’s age) which can culminate in a risk score.13
Lastly, I provide my patients with a handout of the Young Mania Rating Scale to take home as a reference and to complete before our next visit.14
You can repeat scales to monitor for more striking bipolar disorder signs and symptoms that emerge over the course of one’s longitudinal treatment of a pediatric patient. This can be an ongoing, episodic assessment since the emergence of bipolar disorder has been shown to range from the teenage years and beyond into the 20s and sometimes 30s.
Case, continued
Carrie presents to you again while in her first semester of college at the age of 19. She is taking a leave of absence after she began experimenting with cocaine at college and had a manic episode characterized by a lack of sleep without fatigue, persistent unabating energy, rapid and pressured speech, and ultimately, concern from her college friends. She was admitted to a psychiatric unit and stabilized on a second-generation antipsychotic, risperidone, which has solid evidence for mania, but she and you are now concerned about longer-term metabolic effects.15,16
You discuss monitoring her lipid profile and hemoglobin A1c, in addition to weight gain and waist circumference. She has connected with a therapist and psychiatrist through the college counseling center and hopes to return next semester with a fresh start and commitment to sobriety and social rhythms therapy known to be helpful for patients with bipolar disorder.17
While it is challenging to manage a chronic illness at her age, she feels hopeful that she can make better choices for her overall health with your support and the support of her family and mental health team.
Dr. Pawlowski is a child and adolescent consulting psychiatrist. She is a division chief at the University of Vermont Medical Center, Burlington, where she focuses on primary care mental health integration within primary care pediatrics, internal medicine, and family medicine.
References
1. Bipolar Disord. 2016 Jan 9 doi: 10.1111/bdi.12358.
2. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
3. Am J Psychiatry. 2018 Dec 11. doi: 10.1176/appi.ajp.2018.18040461.
4. DSM-5 Changes: Implications for Child Serious Emotional Disturbance. Rockville, Md.: Substance Abuse and Mental Health Services Administration, 2016.
5. The CRAFFT tool.
6. General Behavior Inventory. Parent Version (P-GBI) Short Form – H/B (Revised Version, 2008).
7. Child Mania Rating Scale, Parent Version (CMRS-P).
8. https://www.moodtracker.com.
9. J Clin Psychiatry. 2000 Sep. doi: 10.4088/jcp.v61n0906.
10. Int J Bipolar Disord. 2020 Apr 20. doi: 10.1186/s40345-020-00185-2.
11. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
12. Bipolar Disord. 2017 Sep 25. doi: 10.1111/bdi.12556.
13. www.cabsresearch.pitt.edu/bpriskcalculator/.
14. Parent Version of the Young Mania Rating Scale (PYMRS).
15. Arch Gen Psychiatry. 2012 Jan 2. doi: 10.1001/archgenpsychiatry.2011.1508.
16. The Carlat Child Psychiatry Report. “Bipolar Disorder” Newburyport, Mass.: Carlat Publishing, 2012.
17. https://www.ipsrt.org/.
The assessment and diagnosis of bipolar disorder in youth has a complicated and controversial history. I recall from my child and adolescent fellowship training that there was a thinly veiled faculty argument about the diagnosis itself with strong opinions on each side. To revisit this quandary, I reviewed the most up-to-date literature and outlined a case-based approach to the initial screening assessment. Certainly, the assessment by a child and adolescent psychiatrist would be the standard for diagnosis, but we do know that the pediatrician’s office may be the first setting for a child and parent to present with mood symptoms and concerns about bipolar disorder. What can you do to address this adolescent, Carrie, and her mother’s concerns?
Case
Carrie is a 17-year-old girl who has struggled through her childhood and adolescence with anxious and depressive symptoms which have ebbed and flowed with major life stressors, including her parent’s divorce. She has tried cognitive-behavioral therapy and selective serotonin reuptake inhibitors, but the SSRI seemed to cause feelings of anxiousness and agitation, so she stopped it within weeks.
Her mother presents to you concerned that Carrie has had a more persistently irritable mood toward her, often just wanting to be with her friends or otherwise isolate in her room when home to study.
Most concerning to her mother is that Carrie, as a straight A student, has also developed a pattern of staying up all night to study for tests and then “crashes” and sleeps through the weekend, avoiding her mother and only brightening with her friends.
To complicate matters, Carrie’s biological father had type 1 bipolar disorder and an addiction. Her mother comes to you with an initially nonparticipatory Carrie in tow and says: “My former husband began his manic episodes with a lack of sleep and Carrie is so irritable towards me. I feel like I am walking on eggshells all the time. Could this be bipolar disorder?”
Case discussion
First, it’s always useful to frame a visit stating that you will spend some time with the patient and some time with both the patient and parent. Emphasizing confidentiality about issues such as drug use, which can be comorbid with mood symptoms and go undetected in high-achieving students such as Carrie, is also important. Further emphasizing that information will not be reflexively shared with the parent unless the child presents a danger to herself or others is also paramount to receive an honest report of symptoms.
Second, there are many signs and symptoms of bipolar disorder that naturally overlap with other conditions such as distractibility with attention-deficit/hyperactivity disorder, or irritability in either a unipolar depression or disruptive mood dysregulation disorder.1 You are looking for an episodic (not chronic) course of symptoms with episodes that last over 5 days for hypomania and over the course of weeks for mania all while meeting all the classic criteria for bipolar disorder.
Note that the broadening of diagnostic criteria has been thought to contribute to an inflated sense of prevalence. The actual expert estimate of prevalence is around 0.8%-1.8% in pediatric populations, although there is a large published range depending on whether the criteria are modified or not.2 Use of the unmodified criteria from the DSM-5 is the recommended approach. Bipolar disorder is exceedingly rare in prepubertal children, and it would be more common for prodromal symptoms such as Carrie’s to emerge and escalate over the teenage years, culminating in a clearer diagnosis in the later teens or 20s.3
In my screening questions, I find the idea of an “infatiguable state” is the most pathognomonic one in considering mania in bipolar disorder.4 Carrie’s “crashing” after nights of studying shows that she clearly fatigues. Patients with bipolar disorder within episodes of hypomania or mania have a seismic shift in perceived energy and a matching lack of ability to sleep that can affect their thought processes, speech, and decision-making. At first blush, Carrie’s history does not indicate current symptoms of bipolar disorder.3
Case, continued
When you meet with Carrie alone she shares that she has been experimenting with prescribed stimulants from her older college-aged brother in order to study and ace her tests. She is also experimenting with alcohol and marijuana with her friends. You provide her the CRAFFT tool to deepen your screening of this issue.5
With her mother, you administer the Parent General Behavior Inventory6 and the and the Child Mania Rating Scale7. From these scales, you note that the irritability is more specific to Carrie’s family than pan-present in school and with friends. Her lack of sleep occurs at high-pressure and discreet times.
At this point, you reassure Carrie and her mother that Carrie does not present with symptoms of bipolar disorder but that certainly you will continue screening assessments over time, as they are a good means to track symptoms. You also recommend that Carrie consider mood tracking so she can develop insights into her mood and its relationship to sleep and other events as she prepares for college.8
Case discussion, continued
The strongest risk factor for bipolar disorder in youth is family history (specifically a parent) with bipolar disorder).9 If there is the chance to explore the parent’s illness with open-ended questions, you will want to hear about the parent’s age of symptom onset, course of treatment, any hospitalizations, and stabilizing medications because this has prognostic power for your patient. It is important to ensure that the parent indeed has a diagnosis of bipolar disorder and that it is not just being used colloquially to characterize an adult who has labile moods from hour to hour or day to day. This would give undue anticipatory anxiety to a youth about their risk, which is up to 8- to 10-fold greater with a parent with bipolar disorder.9
Even with a strong family history, we do not often see bipolar disorder emerge in prepubertal children.10,11 There may be still concerning prodromal symptoms in which a diagnosis of unipolar depression with more irritable features and mood lability seems more commonly complicated by substance use, as with Carrie.
Activation with an SSRI, as in Carrie’s case, even if not resulting in full mania or hypomania, can also be a soft sign of the serotonergic sensitivity present in bipolar disorder. However, if there are not additional symptoms of bipolar disorder and you are concerned based on family history alone, you do not want to withhold antidepressant treatment because fear of risk. You would want to consider a “dose low and go slow” titration process with more frequent monitoring.
A diagnostic interview with a child and adolescent psychiatrist and administration of scales such as the Young Mania Rating Scale and the Modified Child Depression Rating Scale are the standard means to assess for bipolar symptoms.12 Considering the dearth of child psychiatrists nationally, it would be useful to improve one’s screening in primary care so as to not inadvertently “refer out” all patients for whom mood dysregulation is a concern.
There is also a more expanded tool that includes several scales integrated with clinical information (parent’s age of mood disorder onset, child’s age) which can culminate in a risk score.13
Lastly, I provide my patients with a handout of the Young Mania Rating Scale to take home as a reference and to complete before our next visit.14
You can repeat scales to monitor for more striking bipolar disorder signs and symptoms that emerge over the course of one’s longitudinal treatment of a pediatric patient. This can be an ongoing, episodic assessment since the emergence of bipolar disorder has been shown to range from the teenage years and beyond into the 20s and sometimes 30s.
Case, continued
Carrie presents to you again while in her first semester of college at the age of 19. She is taking a leave of absence after she began experimenting with cocaine at college and had a manic episode characterized by a lack of sleep without fatigue, persistent unabating energy, rapid and pressured speech, and ultimately, concern from her college friends. She was admitted to a psychiatric unit and stabilized on a second-generation antipsychotic, risperidone, which has solid evidence for mania, but she and you are now concerned about longer-term metabolic effects.15,16
You discuss monitoring her lipid profile and hemoglobin A1c, in addition to weight gain and waist circumference. She has connected with a therapist and psychiatrist through the college counseling center and hopes to return next semester with a fresh start and commitment to sobriety and social rhythms therapy known to be helpful for patients with bipolar disorder.17
While it is challenging to manage a chronic illness at her age, she feels hopeful that she can make better choices for her overall health with your support and the support of her family and mental health team.
Dr. Pawlowski is a child and adolescent consulting psychiatrist. She is a division chief at the University of Vermont Medical Center, Burlington, where she focuses on primary care mental health integration within primary care pediatrics, internal medicine, and family medicine.
References
1. Bipolar Disord. 2016 Jan 9 doi: 10.1111/bdi.12358.
2. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
3. Am J Psychiatry. 2018 Dec 11. doi: 10.1176/appi.ajp.2018.18040461.
4. DSM-5 Changes: Implications for Child Serious Emotional Disturbance. Rockville, Md.: Substance Abuse and Mental Health Services Administration, 2016.
5. The CRAFFT tool.
6. General Behavior Inventory. Parent Version (P-GBI) Short Form – H/B (Revised Version, 2008).
7. Child Mania Rating Scale, Parent Version (CMRS-P).
8. https://www.moodtracker.com.
9. J Clin Psychiatry. 2000 Sep. doi: 10.4088/jcp.v61n0906.
10. Int J Bipolar Disord. 2020 Apr 20. doi: 10.1186/s40345-020-00185-2.
11. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
12. Bipolar Disord. 2017 Sep 25. doi: 10.1111/bdi.12556.
13. www.cabsresearch.pitt.edu/bpriskcalculator/.
14. Parent Version of the Young Mania Rating Scale (PYMRS).
15. Arch Gen Psychiatry. 2012 Jan 2. doi: 10.1001/archgenpsychiatry.2011.1508.
16. The Carlat Child Psychiatry Report. “Bipolar Disorder” Newburyport, Mass.: Carlat Publishing, 2012.
17. https://www.ipsrt.org/.
The assessment and diagnosis of bipolar disorder in youth has a complicated and controversial history. I recall from my child and adolescent fellowship training that there was a thinly veiled faculty argument about the diagnosis itself with strong opinions on each side. To revisit this quandary, I reviewed the most up-to-date literature and outlined a case-based approach to the initial screening assessment. Certainly, the assessment by a child and adolescent psychiatrist would be the standard for diagnosis, but we do know that the pediatrician’s office may be the first setting for a child and parent to present with mood symptoms and concerns about bipolar disorder. What can you do to address this adolescent, Carrie, and her mother’s concerns?
Case
Carrie is a 17-year-old girl who has struggled through her childhood and adolescence with anxious and depressive symptoms which have ebbed and flowed with major life stressors, including her parent’s divorce. She has tried cognitive-behavioral therapy and selective serotonin reuptake inhibitors, but the SSRI seemed to cause feelings of anxiousness and agitation, so she stopped it within weeks.
Her mother presents to you concerned that Carrie has had a more persistently irritable mood toward her, often just wanting to be with her friends or otherwise isolate in her room when home to study.
Most concerning to her mother is that Carrie, as a straight A student, has also developed a pattern of staying up all night to study for tests and then “crashes” and sleeps through the weekend, avoiding her mother and only brightening with her friends.
To complicate matters, Carrie’s biological father had type 1 bipolar disorder and an addiction. Her mother comes to you with an initially nonparticipatory Carrie in tow and says: “My former husband began his manic episodes with a lack of sleep and Carrie is so irritable towards me. I feel like I am walking on eggshells all the time. Could this be bipolar disorder?”
Case discussion
First, it’s always useful to frame a visit stating that you will spend some time with the patient and some time with both the patient and parent. Emphasizing confidentiality about issues such as drug use, which can be comorbid with mood symptoms and go undetected in high-achieving students such as Carrie, is also important. Further emphasizing that information will not be reflexively shared with the parent unless the child presents a danger to herself or others is also paramount to receive an honest report of symptoms.
Second, there are many signs and symptoms of bipolar disorder that naturally overlap with other conditions such as distractibility with attention-deficit/hyperactivity disorder, or irritability in either a unipolar depression or disruptive mood dysregulation disorder.1 You are looking for an episodic (not chronic) course of symptoms with episodes that last over 5 days for hypomania and over the course of weeks for mania all while meeting all the classic criteria for bipolar disorder.
Note that the broadening of diagnostic criteria has been thought to contribute to an inflated sense of prevalence. The actual expert estimate of prevalence is around 0.8%-1.8% in pediatric populations, although there is a large published range depending on whether the criteria are modified or not.2 Use of the unmodified criteria from the DSM-5 is the recommended approach. Bipolar disorder is exceedingly rare in prepubertal children, and it would be more common for prodromal symptoms such as Carrie’s to emerge and escalate over the teenage years, culminating in a clearer diagnosis in the later teens or 20s.3
In my screening questions, I find the idea of an “infatiguable state” is the most pathognomonic one in considering mania in bipolar disorder.4 Carrie’s “crashing” after nights of studying shows that she clearly fatigues. Patients with bipolar disorder within episodes of hypomania or mania have a seismic shift in perceived energy and a matching lack of ability to sleep that can affect their thought processes, speech, and decision-making. At first blush, Carrie’s history does not indicate current symptoms of bipolar disorder.3
Case, continued
When you meet with Carrie alone she shares that she has been experimenting with prescribed stimulants from her older college-aged brother in order to study and ace her tests. She is also experimenting with alcohol and marijuana with her friends. You provide her the CRAFFT tool to deepen your screening of this issue.5
With her mother, you administer the Parent General Behavior Inventory6 and the and the Child Mania Rating Scale7. From these scales, you note that the irritability is more specific to Carrie’s family than pan-present in school and with friends. Her lack of sleep occurs at high-pressure and discreet times.
At this point, you reassure Carrie and her mother that Carrie does not present with symptoms of bipolar disorder but that certainly you will continue screening assessments over time, as they are a good means to track symptoms. You also recommend that Carrie consider mood tracking so she can develop insights into her mood and its relationship to sleep and other events as she prepares for college.8
Case discussion, continued
The strongest risk factor for bipolar disorder in youth is family history (specifically a parent) with bipolar disorder).9 If there is the chance to explore the parent’s illness with open-ended questions, you will want to hear about the parent’s age of symptom onset, course of treatment, any hospitalizations, and stabilizing medications because this has prognostic power for your patient. It is important to ensure that the parent indeed has a diagnosis of bipolar disorder and that it is not just being used colloquially to characterize an adult who has labile moods from hour to hour or day to day. This would give undue anticipatory anxiety to a youth about their risk, which is up to 8- to 10-fold greater with a parent with bipolar disorder.9
Even with a strong family history, we do not often see bipolar disorder emerge in prepubertal children.10,11 There may be still concerning prodromal symptoms in which a diagnosis of unipolar depression with more irritable features and mood lability seems more commonly complicated by substance use, as with Carrie.
Activation with an SSRI, as in Carrie’s case, even if not resulting in full mania or hypomania, can also be a soft sign of the serotonergic sensitivity present in bipolar disorder. However, if there are not additional symptoms of bipolar disorder and you are concerned based on family history alone, you do not want to withhold antidepressant treatment because fear of risk. You would want to consider a “dose low and go slow” titration process with more frequent monitoring.
A diagnostic interview with a child and adolescent psychiatrist and administration of scales such as the Young Mania Rating Scale and the Modified Child Depression Rating Scale are the standard means to assess for bipolar symptoms.12 Considering the dearth of child psychiatrists nationally, it would be useful to improve one’s screening in primary care so as to not inadvertently “refer out” all patients for whom mood dysregulation is a concern.
There is also a more expanded tool that includes several scales integrated with clinical information (parent’s age of mood disorder onset, child’s age) which can culminate in a risk score.13
Lastly, I provide my patients with a handout of the Young Mania Rating Scale to take home as a reference and to complete before our next visit.14
You can repeat scales to monitor for more striking bipolar disorder signs and symptoms that emerge over the course of one’s longitudinal treatment of a pediatric patient. This can be an ongoing, episodic assessment since the emergence of bipolar disorder has been shown to range from the teenage years and beyond into the 20s and sometimes 30s.
Case, continued
Carrie presents to you again while in her first semester of college at the age of 19. She is taking a leave of absence after she began experimenting with cocaine at college and had a manic episode characterized by a lack of sleep without fatigue, persistent unabating energy, rapid and pressured speech, and ultimately, concern from her college friends. She was admitted to a psychiatric unit and stabilized on a second-generation antipsychotic, risperidone, which has solid evidence for mania, but she and you are now concerned about longer-term metabolic effects.15,16
You discuss monitoring her lipid profile and hemoglobin A1c, in addition to weight gain and waist circumference. She has connected with a therapist and psychiatrist through the college counseling center and hopes to return next semester with a fresh start and commitment to sobriety and social rhythms therapy known to be helpful for patients with bipolar disorder.17
While it is challenging to manage a chronic illness at her age, she feels hopeful that she can make better choices for her overall health with your support and the support of her family and mental health team.
Dr. Pawlowski is a child and adolescent consulting psychiatrist. She is a division chief at the University of Vermont Medical Center, Burlington, where she focuses on primary care mental health integration within primary care pediatrics, internal medicine, and family medicine.
References
1. Bipolar Disord. 2016 Jan 9 doi: 10.1111/bdi.12358.
2. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
3. Am J Psychiatry. 2018 Dec 11. doi: 10.1176/appi.ajp.2018.18040461.
4. DSM-5 Changes: Implications for Child Serious Emotional Disturbance. Rockville, Md.: Substance Abuse and Mental Health Services Administration, 2016.
5. The CRAFFT tool.
6. General Behavior Inventory. Parent Version (P-GBI) Short Form – H/B (Revised Version, 2008).
7. Child Mania Rating Scale, Parent Version (CMRS-P).
8. https://www.moodtracker.com.
9. J Clin Psychiatry. 2000 Sep. doi: 10.4088/jcp.v61n0906.
10. Int J Bipolar Disord. 2020 Apr 20. doi: 10.1186/s40345-020-00185-2.
11. Int J Bipolar Disord. 2021 Jun 25. doi: 10.1186/s40345-021-00225-5.
12. Bipolar Disord. 2017 Sep 25. doi: 10.1111/bdi.12556.
13. www.cabsresearch.pitt.edu/bpriskcalculator/.
14. Parent Version of the Young Mania Rating Scale (PYMRS).
15. Arch Gen Psychiatry. 2012 Jan 2. doi: 10.1001/archgenpsychiatry.2011.1508.
16. The Carlat Child Psychiatry Report. “Bipolar Disorder” Newburyport, Mass.: Carlat Publishing, 2012.
17. https://www.ipsrt.org/.
The pediatrician’s office may be the first setting for a child to present with mood symptoms.
Self-management app may boost quality of life
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Postconcussion symptoms tied to high risk of depression
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Sleep complaints in major depression flag risk for other psychiatric disorders
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.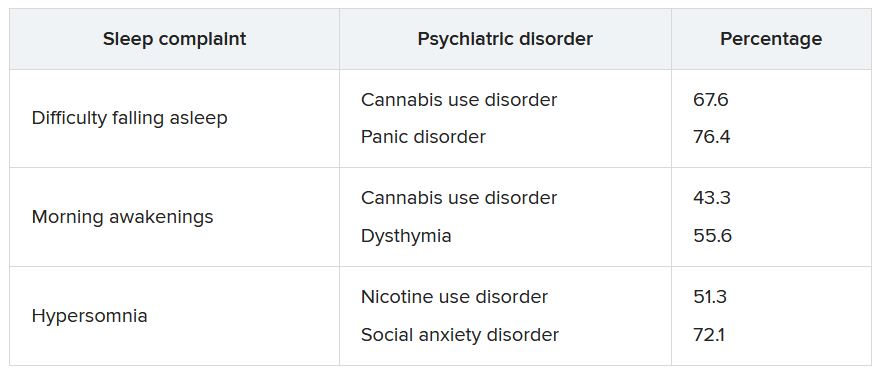
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Hearing loss strongly tied to increased dementia risk
Investigators also found that even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and that hearing aid use was tied to a 32% decrease in dementia prevalence.
“Every 10-decibel increase in hearing loss was associated with 16% greater prevalence of dementia, such that prevalence of dementia in older adults with moderate or greater hearing loss was 61% higher than prevalence in those with normal hearing,” lead investigator Alison Huang, PhD, senior research associate in epidemiology at Johns Hopkins Bloomberg School of Public Health and core faculty in the Cochlear Center for Hearing and Public Health, Baltimore, Md., told this news organization.
The findings were published online in JAMA.
Dose-dependent effect
For the study, researchers analyzed data on 2,413 community-dwelling participants in the National Health and Aging Trends Study, a nationally representative, continuous panel study of U.S. Medicare beneficiaries aged 65 and older.
Data from the study were collected during in-home interviews, setting it apart from previous work that relied on data collected in a clinical setting, Dr. Huang said.
“This study was able to capture more vulnerable populations, such as the oldest old and older adults with disabilities, typically excluded from prior epidemiologic studies of the hearing loss–dementia association that use clinic-based data collection, which only captures people who have the ability and means to get to clinics,” Dr. Huang said.
Weighted hearing loss prevalence was 36.7% for mild and 29.8% for moderate to severe hearing loss, and weighted prevalence of dementia was 10.3%.
Those with moderate to severe hearing loss were 61% more likely to have dementia than those with normal hearing (prevalence ratio, 1.61; 95% confidence interval, 1.09-2.38).
Dementia prevalence increased with increasing severity of hearing loss: normal hearing: 6.19% (95% CI, 4.31-8.80); mild hearing loss: 8.93% (95% CI, 6.99-11.34); moderate/severe hearing loss: 16.52% (95% CI, 13.81-19.64). But only moderate to severe hearing loss showed a statistically significant association with dementia (P = .02).
Dementia prevalence increased 16% per 10-decibel increase in hearing loss (prevalence ratio 1.16; P < .001).
Among the 853 individuals in the study with moderate to severe hearing loss, those who used hearing aids (n = 414) had a 32% lower risk of dementia compared with those who didn’t use assistive devices (prevalence ratio, 0.68; 95% CI, 0.47-1.00). This news organization last month reported on similar data published in JAMA Neurology suggesting that hearing aids reduce dementia risk.
“With this study, we were able to refine our understanding of the strength of the hearing loss–dementia association in a study more representative of older adults in the United States,” said Dr. Huang.
Robust association
Commenting on the findings, Justin S. Golub, MD, associate professor in the department of otolaryngology–head and neck surgery at Columbia University, New York, said the study supports earlier research and suggests a “robust” association between hearing loss and dementia.
“The particular advantage of this study was that it was high quality and nationally representative,” Dr. Golub said. “It is also among a smaller set of studies that have shown hearing aid use to be associated with lower risk of dementia.”
Although not statistically significant, researchers did find increasing prevalence of dementia among people with only mild hearing loss, and clinicians should take note, said Dr. Golub, who was not involved with this study.
“We would expect the relationship between mild hearing loss and dementia to be weaker than severe hearing loss and dementia and, as a result, it might take more participants to show an association among the mild group,” Dr. Golub said.
“Even though this particular study did not specifically find a relationship between mild hearing loss and dementia, I would still recommend people to start treating their hearing loss when it is early,” Dr. Golub added.
The study was funded by the National Institute on Aging. Dr. Golub reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators also found that even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and that hearing aid use was tied to a 32% decrease in dementia prevalence.
“Every 10-decibel increase in hearing loss was associated with 16% greater prevalence of dementia, such that prevalence of dementia in older adults with moderate or greater hearing loss was 61% higher than prevalence in those with normal hearing,” lead investigator Alison Huang, PhD, senior research associate in epidemiology at Johns Hopkins Bloomberg School of Public Health and core faculty in the Cochlear Center for Hearing and Public Health, Baltimore, Md., told this news organization.
The findings were published online in JAMA.
Dose-dependent effect
For the study, researchers analyzed data on 2,413 community-dwelling participants in the National Health and Aging Trends Study, a nationally representative, continuous panel study of U.S. Medicare beneficiaries aged 65 and older.
Data from the study were collected during in-home interviews, setting it apart from previous work that relied on data collected in a clinical setting, Dr. Huang said.
“This study was able to capture more vulnerable populations, such as the oldest old and older adults with disabilities, typically excluded from prior epidemiologic studies of the hearing loss–dementia association that use clinic-based data collection, which only captures people who have the ability and means to get to clinics,” Dr. Huang said.
Weighted hearing loss prevalence was 36.7% for mild and 29.8% for moderate to severe hearing loss, and weighted prevalence of dementia was 10.3%.
Those with moderate to severe hearing loss were 61% more likely to have dementia than those with normal hearing (prevalence ratio, 1.61; 95% confidence interval, 1.09-2.38).
Dementia prevalence increased with increasing severity of hearing loss: normal hearing: 6.19% (95% CI, 4.31-8.80); mild hearing loss: 8.93% (95% CI, 6.99-11.34); moderate/severe hearing loss: 16.52% (95% CI, 13.81-19.64). But only moderate to severe hearing loss showed a statistically significant association with dementia (P = .02).
Dementia prevalence increased 16% per 10-decibel increase in hearing loss (prevalence ratio 1.16; P < .001).
Among the 853 individuals in the study with moderate to severe hearing loss, those who used hearing aids (n = 414) had a 32% lower risk of dementia compared with those who didn’t use assistive devices (prevalence ratio, 0.68; 95% CI, 0.47-1.00). This news organization last month reported on similar data published in JAMA Neurology suggesting that hearing aids reduce dementia risk.
“With this study, we were able to refine our understanding of the strength of the hearing loss–dementia association in a study more representative of older adults in the United States,” said Dr. Huang.
Robust association
Commenting on the findings, Justin S. Golub, MD, associate professor in the department of otolaryngology–head and neck surgery at Columbia University, New York, said the study supports earlier research and suggests a “robust” association between hearing loss and dementia.
“The particular advantage of this study was that it was high quality and nationally representative,” Dr. Golub said. “It is also among a smaller set of studies that have shown hearing aid use to be associated with lower risk of dementia.”
Although not statistically significant, researchers did find increasing prevalence of dementia among people with only mild hearing loss, and clinicians should take note, said Dr. Golub, who was not involved with this study.
“We would expect the relationship between mild hearing loss and dementia to be weaker than severe hearing loss and dementia and, as a result, it might take more participants to show an association among the mild group,” Dr. Golub said.
“Even though this particular study did not specifically find a relationship between mild hearing loss and dementia, I would still recommend people to start treating their hearing loss when it is early,” Dr. Golub added.
The study was funded by the National Institute on Aging. Dr. Golub reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators also found that even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and that hearing aid use was tied to a 32% decrease in dementia prevalence.
“Every 10-decibel increase in hearing loss was associated with 16% greater prevalence of dementia, such that prevalence of dementia in older adults with moderate or greater hearing loss was 61% higher than prevalence in those with normal hearing,” lead investigator Alison Huang, PhD, senior research associate in epidemiology at Johns Hopkins Bloomberg School of Public Health and core faculty in the Cochlear Center for Hearing and Public Health, Baltimore, Md., told this news organization.
The findings were published online in JAMA.
Dose-dependent effect
For the study, researchers analyzed data on 2,413 community-dwelling participants in the National Health and Aging Trends Study, a nationally representative, continuous panel study of U.S. Medicare beneficiaries aged 65 and older.
Data from the study were collected during in-home interviews, setting it apart from previous work that relied on data collected in a clinical setting, Dr. Huang said.
“This study was able to capture more vulnerable populations, such as the oldest old and older adults with disabilities, typically excluded from prior epidemiologic studies of the hearing loss–dementia association that use clinic-based data collection, which only captures people who have the ability and means to get to clinics,” Dr. Huang said.
Weighted hearing loss prevalence was 36.7% for mild and 29.8% for moderate to severe hearing loss, and weighted prevalence of dementia was 10.3%.
Those with moderate to severe hearing loss were 61% more likely to have dementia than those with normal hearing (prevalence ratio, 1.61; 95% confidence interval, 1.09-2.38).
Dementia prevalence increased with increasing severity of hearing loss: normal hearing: 6.19% (95% CI, 4.31-8.80); mild hearing loss: 8.93% (95% CI, 6.99-11.34); moderate/severe hearing loss: 16.52% (95% CI, 13.81-19.64). But only moderate to severe hearing loss showed a statistically significant association with dementia (P = .02).
Dementia prevalence increased 16% per 10-decibel increase in hearing loss (prevalence ratio 1.16; P < .001).
Among the 853 individuals in the study with moderate to severe hearing loss, those who used hearing aids (n = 414) had a 32% lower risk of dementia compared with those who didn’t use assistive devices (prevalence ratio, 0.68; 95% CI, 0.47-1.00). This news organization last month reported on similar data published in JAMA Neurology suggesting that hearing aids reduce dementia risk.
“With this study, we were able to refine our understanding of the strength of the hearing loss–dementia association in a study more representative of older adults in the United States,” said Dr. Huang.
Robust association
Commenting on the findings, Justin S. Golub, MD, associate professor in the department of otolaryngology–head and neck surgery at Columbia University, New York, said the study supports earlier research and suggests a “robust” association between hearing loss and dementia.
“The particular advantage of this study was that it was high quality and nationally representative,” Dr. Golub said. “It is also among a smaller set of studies that have shown hearing aid use to be associated with lower risk of dementia.”
Although not statistically significant, researchers did find increasing prevalence of dementia among people with only mild hearing loss, and clinicians should take note, said Dr. Golub, who was not involved with this study.
“We would expect the relationship between mild hearing loss and dementia to be weaker than severe hearing loss and dementia and, as a result, it might take more participants to show an association among the mild group,” Dr. Golub said.
“Even though this particular study did not specifically find a relationship between mild hearing loss and dementia, I would still recommend people to start treating their hearing loss when it is early,” Dr. Golub added.
The study was funded by the National Institute on Aging. Dr. Golub reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA
Telehealth parent-child interaction therapy improved behavior in children with developmental delay
The children received the therapy with their parents or caregivers, who were more likely to demonstrate positive parenting behaviors than parents in the control group, authors of the new research published in JAMA Pediatrics found.
Approximately 13% of children have some form of developmental delay (DD) and more than half of these children also have at least one mental health disorder, which makes behavior problems a common and ongoing challenge, Daniel M. Bagner, PhD, a psychologist at Florida International University, Miami, and colleagues wrote.
Clinic-based interventions such as parent-child interaction therapy (PCIT) have been effective for improving behavior in children with DD, the researchers said. PCIT involves in-session caregiver coaching using a 1-way mirror and a wireless earpiece worn by the caregiver.
Barriers to the use of PCIT, especially in marginalized and low-income communities, include transportation, clinician shortages, and stigma-related concerns about a clinic visit, the researchers wrote. Technology now allows for Internet-delivered PCIT to reach more children and families, but its effectiveness for children with DD has not been well studied.
In the new study, the researchers randomized 150 children with DD and externalizing behavior problems to up to 20 weeks of Internet-delivered parent-child interaction therapy (iPCIT) or to referral as usual (RAU, the control group). The children were randomized after completion of early intervention services within 3 months of their third birthday, and participated in the sessions with a parent or caregiver. Most of the participants were from economically disadvantaged households and underrepresented ethnic backgrounds.
The iPCIT intervention was conducted weekly with a remote therapist and lasted for 1-1.5 hours; approximately half of the families received the intervention in Spanish.
The primary outcome was rating on the Child Behavior Checklist (CBCL) and assessment of children and caregivers using the Dyadic Parent-Child Interaction Coding System, fourth edition (DPICS). Assessments occurred at baseline and at week 20 (post treatment), with follow ups at 6 and 12 months.
Scores on the CBCL in the iPCIT group decreased from a mean of 61.18 at baseline to 53.83 post intervention. Scores for the control group started at 64.05 and decreased to 59.49 post intervention. At 6-12 months, the scores for both groups remained stable.
Children who received iPCIT with their parent or caregiver also showed significantly lower levels of externalizing behavior problems, compared with the RAU controls post treatment, and at 6-month and 12-month follow-ups based on the Cohen d measure of standardized effect size for differences between groups.
Significantly more children in the iPCIT group showed clinically significant improvements in externalizing problems at post treatment, compared with the RAU group (74% vs. 42%; P < .001) and at 6 months’ follow-up (73% vs. 45%; P = .002). However, the differences from baseline were not significantly different between the two groups after 12 months, which suggests that the effects may wane over time, the researchers noted.
In addition, the rate of child compliance with parent commands, as measured by a cleanup task, approximately doubled by the 12-month follow-up among children in the iPCIT group versus an increase of approximately one-third in the RAU group.
For secondary outcome measures related to caregiver behaviors, the proportion of observed positive parenting behaviors increased in the iPCIT group during the course of the intervention (postintervention odds ratio, 1.10), and the proportion of controlling and critical behaviors decreased (postintervention OR, 1.40). Harsh and inconsistent discipline decreased in both groups based on self-reports, but the decrease was steeper in iPCIT families.
iPCIT did not have a greater impact than RAU in reducing caregiver stress. The researchers wrote that they were not surprised by the lack of stress reduction “given mixed findings on the impact of parenting interventions on stress in caregivers of children with DD.”
Data support iPCIT potential
Overall, the results support findings from previous studies of clinic-based PCIT for children with DD and previous studies of telehealth interventions for typically developing children, the researchers said.
“Moreover, iPCIT-treated children not only showed reductions in behavior problems, such as aggression, but demonstrated higher rates of following directions, which is especially important for children entering kindergarten,” they wrote.
The findings were limited by several factors including the narrow focus on the primary and secondary outcomes, the use of data from a single site in a single metropolitan area – which may limit generalizability – and the lack of comparison between iPCIT and a clinic-based PCIT control group, the researchers noted. The equipment in the current study was provided to families; therefore, differences in treatment response could not be attributed to differences in technology.
The study represents the first known randomized controlled trial to evaluate a telehealth parenting intervention for children with, according to the researchers. The results suggest that technology can be leveraged to help these patients, including those from ethnic minority families who may be underserved by clinic-based care in overcoming barriers to treatment such as transportation and availability of clinicians. Use of iPCIT could be a critical resource as young children with DD complete Part C services and enter the school system.
Practical pediatric takeaways
“This was a great study, well-designed and very important and helpful for pediatric providers,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Young children with developmental delay and/or mental and behavioral health disorders require early identification and intervention,” said Dr. Haut. However, obstacles to intervention include stigma or parental denial of the disorder, as well as more practical challenges related to transportation, time to access a clinic or office, potential long length of treatment, and cost.
“Despite availability of state programs for young children, follow up and continued services can be challenging to complete. Once the child outgrows the state program finding alternative therapy can be difficult with the current shortage of pediatric mental health providers,” Dr. Haut noted.
“I was surprised to see that this study treatment phase was completed prior to the COVID-19 pandemic, when telehealth was not as popular a mode for health care and was not utilized to the extent that it is now, especially for pediatric care,” said Dr. Haut. “I was not surprised at the results, as the traditional mode of PCIT includes therapy and training in a space that may not be as familiar to the child as their home environment, and would include live presence of the therapist/s, which may add to anxiety for both the parent and child.”
That almost half of the parents participating in the study had graduated from college and/or completed graduate degrees “may have contributed to some of the success of this study,” Dr. Haut noted.
Benefits and barriers
“The COVID-19 pandemic brought significant change to the frequency of use and overall success of telehealth services,” Dr. Haut said. “Additional provider education in aspects such as provider technique and the use of medical devices with improved specific health care technology assisted in advancing the experience and opportunity for successful telehealth visits. Telehealth therapy offers a cost-effective option for any pediatric patients and for providers, as the time and space commitment for the patient visit can be considerably less than live office visits.
“Unfortunately, there are still overall barriers that I have personally experienced with telehealth, including interruptions in connectivity, background noise, and lack of an available computer or tablet; and with the use of cell phones not always allowing full inclusion of the caregiver and child,” said Dr. Haut. Children with DD, behavioral problems, or other mental health disorders may pose challenges for parents to manage at home while simultaneously trying to fully focus on the therapy in an online setting.
Although the current study is encouraging, “larger studies focused on specific or individual pediatric mental health and/or behavioral disorders may offer more information for providers, and better document the success of telehealth delivery of services,” Dr. Haut said.
The study was supported by the National Institute of Child Health and Human Development. Dr. Bagner disclosed funding from the National Institutes of Health. He also disclosed personal fees from PCIT International to train clinicians in PCIT supported by a grant from the Florida Department of Children and Families outside the current study. Dr. Haut had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
The children received the therapy with their parents or caregivers, who were more likely to demonstrate positive parenting behaviors than parents in the control group, authors of the new research published in JAMA Pediatrics found.
Approximately 13% of children have some form of developmental delay (DD) and more than half of these children also have at least one mental health disorder, which makes behavior problems a common and ongoing challenge, Daniel M. Bagner, PhD, a psychologist at Florida International University, Miami, and colleagues wrote.
Clinic-based interventions such as parent-child interaction therapy (PCIT) have been effective for improving behavior in children with DD, the researchers said. PCIT involves in-session caregiver coaching using a 1-way mirror and a wireless earpiece worn by the caregiver.
Barriers to the use of PCIT, especially in marginalized and low-income communities, include transportation, clinician shortages, and stigma-related concerns about a clinic visit, the researchers wrote. Technology now allows for Internet-delivered PCIT to reach more children and families, but its effectiveness for children with DD has not been well studied.
In the new study, the researchers randomized 150 children with DD and externalizing behavior problems to up to 20 weeks of Internet-delivered parent-child interaction therapy (iPCIT) or to referral as usual (RAU, the control group). The children were randomized after completion of early intervention services within 3 months of their third birthday, and participated in the sessions with a parent or caregiver. Most of the participants were from economically disadvantaged households and underrepresented ethnic backgrounds.
The iPCIT intervention was conducted weekly with a remote therapist and lasted for 1-1.5 hours; approximately half of the families received the intervention in Spanish.
The primary outcome was rating on the Child Behavior Checklist (CBCL) and assessment of children and caregivers using the Dyadic Parent-Child Interaction Coding System, fourth edition (DPICS). Assessments occurred at baseline and at week 20 (post treatment), with follow ups at 6 and 12 months.
Scores on the CBCL in the iPCIT group decreased from a mean of 61.18 at baseline to 53.83 post intervention. Scores for the control group started at 64.05 and decreased to 59.49 post intervention. At 6-12 months, the scores for both groups remained stable.
Children who received iPCIT with their parent or caregiver also showed significantly lower levels of externalizing behavior problems, compared with the RAU controls post treatment, and at 6-month and 12-month follow-ups based on the Cohen d measure of standardized effect size for differences between groups.
Significantly more children in the iPCIT group showed clinically significant improvements in externalizing problems at post treatment, compared with the RAU group (74% vs. 42%; P < .001) and at 6 months’ follow-up (73% vs. 45%; P = .002). However, the differences from baseline were not significantly different between the two groups after 12 months, which suggests that the effects may wane over time, the researchers noted.
In addition, the rate of child compliance with parent commands, as measured by a cleanup task, approximately doubled by the 12-month follow-up among children in the iPCIT group versus an increase of approximately one-third in the RAU group.
For secondary outcome measures related to caregiver behaviors, the proportion of observed positive parenting behaviors increased in the iPCIT group during the course of the intervention (postintervention odds ratio, 1.10), and the proportion of controlling and critical behaviors decreased (postintervention OR, 1.40). Harsh and inconsistent discipline decreased in both groups based on self-reports, but the decrease was steeper in iPCIT families.
iPCIT did not have a greater impact than RAU in reducing caregiver stress. The researchers wrote that they were not surprised by the lack of stress reduction “given mixed findings on the impact of parenting interventions on stress in caregivers of children with DD.”
Data support iPCIT potential
Overall, the results support findings from previous studies of clinic-based PCIT for children with DD and previous studies of telehealth interventions for typically developing children, the researchers said.
“Moreover, iPCIT-treated children not only showed reductions in behavior problems, such as aggression, but demonstrated higher rates of following directions, which is especially important for children entering kindergarten,” they wrote.
The findings were limited by several factors including the narrow focus on the primary and secondary outcomes, the use of data from a single site in a single metropolitan area – which may limit generalizability – and the lack of comparison between iPCIT and a clinic-based PCIT control group, the researchers noted. The equipment in the current study was provided to families; therefore, differences in treatment response could not be attributed to differences in technology.
The study represents the first known randomized controlled trial to evaluate a telehealth parenting intervention for children with, according to the researchers. The results suggest that technology can be leveraged to help these patients, including those from ethnic minority families who may be underserved by clinic-based care in overcoming barriers to treatment such as transportation and availability of clinicians. Use of iPCIT could be a critical resource as young children with DD complete Part C services and enter the school system.
Practical pediatric takeaways
“This was a great study, well-designed and very important and helpful for pediatric providers,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Young children with developmental delay and/or mental and behavioral health disorders require early identification and intervention,” said Dr. Haut. However, obstacles to intervention include stigma or parental denial of the disorder, as well as more practical challenges related to transportation, time to access a clinic or office, potential long length of treatment, and cost.
“Despite availability of state programs for young children, follow up and continued services can be challenging to complete. Once the child outgrows the state program finding alternative therapy can be difficult with the current shortage of pediatric mental health providers,” Dr. Haut noted.
“I was surprised to see that this study treatment phase was completed prior to the COVID-19 pandemic, when telehealth was not as popular a mode for health care and was not utilized to the extent that it is now, especially for pediatric care,” said Dr. Haut. “I was not surprised at the results, as the traditional mode of PCIT includes therapy and training in a space that may not be as familiar to the child as their home environment, and would include live presence of the therapist/s, which may add to anxiety for both the parent and child.”
That almost half of the parents participating in the study had graduated from college and/or completed graduate degrees “may have contributed to some of the success of this study,” Dr. Haut noted.
Benefits and barriers
“The COVID-19 pandemic brought significant change to the frequency of use and overall success of telehealth services,” Dr. Haut said. “Additional provider education in aspects such as provider technique and the use of medical devices with improved specific health care technology assisted in advancing the experience and opportunity for successful telehealth visits. Telehealth therapy offers a cost-effective option for any pediatric patients and for providers, as the time and space commitment for the patient visit can be considerably less than live office visits.
“Unfortunately, there are still overall barriers that I have personally experienced with telehealth, including interruptions in connectivity, background noise, and lack of an available computer or tablet; and with the use of cell phones not always allowing full inclusion of the caregiver and child,” said Dr. Haut. Children with DD, behavioral problems, or other mental health disorders may pose challenges for parents to manage at home while simultaneously trying to fully focus on the therapy in an online setting.
Although the current study is encouraging, “larger studies focused on specific or individual pediatric mental health and/or behavioral disorders may offer more information for providers, and better document the success of telehealth delivery of services,” Dr. Haut said.
The study was supported by the National Institute of Child Health and Human Development. Dr. Bagner disclosed funding from the National Institutes of Health. He also disclosed personal fees from PCIT International to train clinicians in PCIT supported by a grant from the Florida Department of Children and Families outside the current study. Dr. Haut had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
The children received the therapy with their parents or caregivers, who were more likely to demonstrate positive parenting behaviors than parents in the control group, authors of the new research published in JAMA Pediatrics found.
Approximately 13% of children have some form of developmental delay (DD) and more than half of these children also have at least one mental health disorder, which makes behavior problems a common and ongoing challenge, Daniel M. Bagner, PhD, a psychologist at Florida International University, Miami, and colleagues wrote.
Clinic-based interventions such as parent-child interaction therapy (PCIT) have been effective for improving behavior in children with DD, the researchers said. PCIT involves in-session caregiver coaching using a 1-way mirror and a wireless earpiece worn by the caregiver.
Barriers to the use of PCIT, especially in marginalized and low-income communities, include transportation, clinician shortages, and stigma-related concerns about a clinic visit, the researchers wrote. Technology now allows for Internet-delivered PCIT to reach more children and families, but its effectiveness for children with DD has not been well studied.
In the new study, the researchers randomized 150 children with DD and externalizing behavior problems to up to 20 weeks of Internet-delivered parent-child interaction therapy (iPCIT) or to referral as usual (RAU, the control group). The children were randomized after completion of early intervention services within 3 months of their third birthday, and participated in the sessions with a parent or caregiver. Most of the participants were from economically disadvantaged households and underrepresented ethnic backgrounds.
The iPCIT intervention was conducted weekly with a remote therapist and lasted for 1-1.5 hours; approximately half of the families received the intervention in Spanish.
The primary outcome was rating on the Child Behavior Checklist (CBCL) and assessment of children and caregivers using the Dyadic Parent-Child Interaction Coding System, fourth edition (DPICS). Assessments occurred at baseline and at week 20 (post treatment), with follow ups at 6 and 12 months.
Scores on the CBCL in the iPCIT group decreased from a mean of 61.18 at baseline to 53.83 post intervention. Scores for the control group started at 64.05 and decreased to 59.49 post intervention. At 6-12 months, the scores for both groups remained stable.
Children who received iPCIT with their parent or caregiver also showed significantly lower levels of externalizing behavior problems, compared with the RAU controls post treatment, and at 6-month and 12-month follow-ups based on the Cohen d measure of standardized effect size for differences between groups.
Significantly more children in the iPCIT group showed clinically significant improvements in externalizing problems at post treatment, compared with the RAU group (74% vs. 42%; P < .001) and at 6 months’ follow-up (73% vs. 45%; P = .002). However, the differences from baseline were not significantly different between the two groups after 12 months, which suggests that the effects may wane over time, the researchers noted.
In addition, the rate of child compliance with parent commands, as measured by a cleanup task, approximately doubled by the 12-month follow-up among children in the iPCIT group versus an increase of approximately one-third in the RAU group.
For secondary outcome measures related to caregiver behaviors, the proportion of observed positive parenting behaviors increased in the iPCIT group during the course of the intervention (postintervention odds ratio, 1.10), and the proportion of controlling and critical behaviors decreased (postintervention OR, 1.40). Harsh and inconsistent discipline decreased in both groups based on self-reports, but the decrease was steeper in iPCIT families.
iPCIT did not have a greater impact than RAU in reducing caregiver stress. The researchers wrote that they were not surprised by the lack of stress reduction “given mixed findings on the impact of parenting interventions on stress in caregivers of children with DD.”
Data support iPCIT potential
Overall, the results support findings from previous studies of clinic-based PCIT for children with DD and previous studies of telehealth interventions for typically developing children, the researchers said.
“Moreover, iPCIT-treated children not only showed reductions in behavior problems, such as aggression, but demonstrated higher rates of following directions, which is especially important for children entering kindergarten,” they wrote.
The findings were limited by several factors including the narrow focus on the primary and secondary outcomes, the use of data from a single site in a single metropolitan area – which may limit generalizability – and the lack of comparison between iPCIT and a clinic-based PCIT control group, the researchers noted. The equipment in the current study was provided to families; therefore, differences in treatment response could not be attributed to differences in technology.
The study represents the first known randomized controlled trial to evaluate a telehealth parenting intervention for children with, according to the researchers. The results suggest that technology can be leveraged to help these patients, including those from ethnic minority families who may be underserved by clinic-based care in overcoming barriers to treatment such as transportation and availability of clinicians. Use of iPCIT could be a critical resource as young children with DD complete Part C services and enter the school system.
Practical pediatric takeaways
“This was a great study, well-designed and very important and helpful for pediatric providers,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Young children with developmental delay and/or mental and behavioral health disorders require early identification and intervention,” said Dr. Haut. However, obstacles to intervention include stigma or parental denial of the disorder, as well as more practical challenges related to transportation, time to access a clinic or office, potential long length of treatment, and cost.
“Despite availability of state programs for young children, follow up and continued services can be challenging to complete. Once the child outgrows the state program finding alternative therapy can be difficult with the current shortage of pediatric mental health providers,” Dr. Haut noted.
“I was surprised to see that this study treatment phase was completed prior to the COVID-19 pandemic, when telehealth was not as popular a mode for health care and was not utilized to the extent that it is now, especially for pediatric care,” said Dr. Haut. “I was not surprised at the results, as the traditional mode of PCIT includes therapy and training in a space that may not be as familiar to the child as their home environment, and would include live presence of the therapist/s, which may add to anxiety for both the parent and child.”
That almost half of the parents participating in the study had graduated from college and/or completed graduate degrees “may have contributed to some of the success of this study,” Dr. Haut noted.
Benefits and barriers
“The COVID-19 pandemic brought significant change to the frequency of use and overall success of telehealth services,” Dr. Haut said. “Additional provider education in aspects such as provider technique and the use of medical devices with improved specific health care technology assisted in advancing the experience and opportunity for successful telehealth visits. Telehealth therapy offers a cost-effective option for any pediatric patients and for providers, as the time and space commitment for the patient visit can be considerably less than live office visits.
“Unfortunately, there are still overall barriers that I have personally experienced with telehealth, including interruptions in connectivity, background noise, and lack of an available computer or tablet; and with the use of cell phones not always allowing full inclusion of the caregiver and child,” said Dr. Haut. Children with DD, behavioral problems, or other mental health disorders may pose challenges for parents to manage at home while simultaneously trying to fully focus on the therapy in an online setting.
Although the current study is encouraging, “larger studies focused on specific or individual pediatric mental health and/or behavioral disorders may offer more information for providers, and better document the success of telehealth delivery of services,” Dr. Haut said.
The study was supported by the National Institute of Child Health and Human Development. Dr. Bagner disclosed funding from the National Institutes of Health. He also disclosed personal fees from PCIT International to train clinicians in PCIT supported by a grant from the Florida Department of Children and Families outside the current study. Dr. Haut had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
FROM JAMA PEDIATRICS
Age competency exams for physicians – yes or no?
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
What to know about newly approved Alzheimer’s drug
, offering hope where there has been little for patients and their families affected by the devastating disease.
More than 6 million people in the United States live with Alzheimer’s.
It’s not a cure, but the drug, given intravenously every 2 weeks, has shown moderate positive effects in clinical trials in slowing early-stage disease.
But many are wary. As explained in an editorial in the journal The Lancet, “The Alzheimer’s disease community has become accustomed to false hope, disappointment, and controversy.”
Some worry about lecanemab’s safety as some people in clinical trials experienced serious side effects of bleeding and swelling in the brain. Scientists recently attributed a third death to lecanemab, brand name Leqembi, though the drugmaker disputed the medication was the cause.
So what should patients and their families make of this news? Here we answer some of the top questions surrounding the drug.
What does the FDA action mean?
The FDA granted accelerated approval to Leqembi after it showed positive trial results in slowing the progression of early-stage disease.
The FDA can grant accelerated approval for drugs that treat serious conditions and fill an unmet medical need while drugs continue to be studied in larger trials.
With the FDA approval in hand, doctors can now prescribe the medication.
Rebecca Edelmayer, PhD, the Alzheimer’s Association senior director of scientific engagement, says that with the FDA’s move, ramping up manufacturing – and eventually nationwide distribution and implementation – will take some time.
“Ask your doctor about availability,” she says. “The main issue is that, without insurance and Medicare coverage of this class of treatments, access for those who could benefit from the newly approved treatment will only be available to those who can pay out-of-pocket. Without coverage, people simply won’t be able to get the treatment.”
The Washington Post reports that with accelerated approval, drugmaker Eisai is expected to immediately apply for full FDA approval, which wouldn’t be likely to come before later this year. Full approval could help clear the path for Medicare coverage of the drug.
Potential benefit?
Those who got Leqembi in a clinical trial for 18 months experienced 27% less decline in memory and thinking relative to the group who got a placebo. It also reduced amyloid in the brain, the sticky protein that builds up in the brains of people with Alzheimer’s and is considered a hallmark of the disease.
Howard Fillit, MD, cofounder and chief science officer of the Alzheimer’s Drug Discovery Foundation, says, “It’s the first phase 3 study in our field of a disease-modifying drug where the clinical efficacy was very clear.”
Concerns about side effects
The drug has raised safety concerns as it has been linked with certain serious adverse events, including brain swelling and bleeding. In the trial, 14% of patients who received the drug experienced side effects that included brain swelling and bleeding, compared with about 11% in the placebo group.
Scientists have reportedly linked three deaths during the clinical trial to lecanemab, though it is unclear whether it caused the deaths.
Dr. Fillit notes that the first two people who died were on blood thinners when they received lecanemab.
“There are things about the use of the drug in the real world that we need to work out, especially in the context of people with comorbidities,” he says.
The third death is a little different, Dr. Fillit says. The patient, who had a stroke, showed signs of vasculitis, or inflammation of the blood vessels.
“We don’t know exactly what happened, but we do know it was very, very rare” among the people involved in the trials, he says.
Dr. Edelmayer says that the most common reported side effects during the trials were infusion-related reactions, headache, and amyloid-related imaging abnormalities (ARIA). According to the FDA, these abnormalities “are known to occur with antibodies of this class. ARIA usually does not have symptoms, although serious and life-threatening events rarely may occur.”
The FDA has added these as warnings to the drug’s label, describing the possible infusion-related reactions as flu-like symptoms, nausea, vomiting, and changes in blood pressure.
How much will it cost?
Eisai says that lecanemab will cost $26,500 a year.
In a draft report released in December, the Institute for Clinical and Economic Review said a price ranging from $8,500 to $20,600 a year would make the drug cost-effective. While the group has no authority to set prices, many large health insurers consider its reports when they negotiate prices and some drugmakers take into account ICER’s recommendations when setting prices.
An editorial in The Lancet last month warns that the cost will likely be “prohibitive” for low- and middle-income countries and many health systems don’t have the infrastructure for a widespread rollout.
Will Medicare cover it?
The Centers for Medicare & Medicaid Services, which runs Medicare, which covers most people with Alzheimer’s, has indicated it won’t broadly cover amyloid-lowering drugs until the drug gets full U.S. approval based on clinical benefits, as opposed to accelerated approval.
That means people would have to pay thousands out of pocket at first to get it.
The CMS decision effectively denies Medicare coverage of fast-tracked FDA-approved medications for Alzheimer’s disease unless the person is enrolled in an approved clinical trial.
On Dec. 19, the Alzheimer’s Association filed a formal request asking CMS to remove the trial-only requirement and provide full and unrestricted coverage for FDA-approved Alzheimer’s treatments.
CMS says in a statement issued after the announcement: “Because Eisai’s product, lecanemab, was granted accelerated approval by the FDA, it falls under CMS’s existing national coverage determination. CMS is examining available information and may reconsider its current coverage based on this review.”
“If lecanemab subsequently receives traditional FDA approval, CMS would provide broader coverage,” the statement says.
Who benefits most from this drug?
Lecanemab is a treatment for people with early-stage Alzheimer’s disease who have amyloid in their brain. This means people with other types of dementia, or those in the later stages of Alzheimer’s disease, are not likely to improve with this drug.
Who makes lecanemab?
Japan-based Eisai is developing the drug, a monoclonal antibody, in collaboration with the U.S. company Biogen.
What’s the Alzheimer’s Association’s view?
The association urged accelerated FDA approval. In a statement, it says it “welcomes and is further encouraged” by the clinical trial results.
It says data published in the New England Journal of Medicine confirms lecanemab “can meaningfully change the course of the disease for people in the earliest stages of Alzheimer’s disease.”
“We are energized at the progress we are seeing in the research pipeline. The science is telling us that although antiamyloid treatments are not a cure – they are not going to be the end of treating Alzheimer’s – they are certainly the beginning,” Dr. Edelmayer says.
Are there alternatives?
The FDA gave accelerated approval to Biogen to produce another drug for Alzheimer’s, Aduhelm (aducanemab), in 2021, but the move was controversial as the drug’s effectiveness was widely questioned. It has since largely been pulled from the market.
Aduhelm had been the first approved early-stage Alzheimer’s treatment since 2003.
A version of this article first appeared on WebMD.com.
, offering hope where there has been little for patients and their families affected by the devastating disease.
More than 6 million people in the United States live with Alzheimer’s.
It’s not a cure, but the drug, given intravenously every 2 weeks, has shown moderate positive effects in clinical trials in slowing early-stage disease.
But many are wary. As explained in an editorial in the journal The Lancet, “The Alzheimer’s disease community has become accustomed to false hope, disappointment, and controversy.”
Some worry about lecanemab’s safety as some people in clinical trials experienced serious side effects of bleeding and swelling in the brain. Scientists recently attributed a third death to lecanemab, brand name Leqembi, though the drugmaker disputed the medication was the cause.
So what should patients and their families make of this news? Here we answer some of the top questions surrounding the drug.
What does the FDA action mean?
The FDA granted accelerated approval to Leqembi after it showed positive trial results in slowing the progression of early-stage disease.
The FDA can grant accelerated approval for drugs that treat serious conditions and fill an unmet medical need while drugs continue to be studied in larger trials.
With the FDA approval in hand, doctors can now prescribe the medication.
Rebecca Edelmayer, PhD, the Alzheimer’s Association senior director of scientific engagement, says that with the FDA’s move, ramping up manufacturing – and eventually nationwide distribution and implementation – will take some time.
“Ask your doctor about availability,” she says. “The main issue is that, without insurance and Medicare coverage of this class of treatments, access for those who could benefit from the newly approved treatment will only be available to those who can pay out-of-pocket. Without coverage, people simply won’t be able to get the treatment.”
The Washington Post reports that with accelerated approval, drugmaker Eisai is expected to immediately apply for full FDA approval, which wouldn’t be likely to come before later this year. Full approval could help clear the path for Medicare coverage of the drug.
Potential benefit?
Those who got Leqembi in a clinical trial for 18 months experienced 27% less decline in memory and thinking relative to the group who got a placebo. It also reduced amyloid in the brain, the sticky protein that builds up in the brains of people with Alzheimer’s and is considered a hallmark of the disease.
Howard Fillit, MD, cofounder and chief science officer of the Alzheimer’s Drug Discovery Foundation, says, “It’s the first phase 3 study in our field of a disease-modifying drug where the clinical efficacy was very clear.”
Concerns about side effects
The drug has raised safety concerns as it has been linked with certain serious adverse events, including brain swelling and bleeding. In the trial, 14% of patients who received the drug experienced side effects that included brain swelling and bleeding, compared with about 11% in the placebo group.
Scientists have reportedly linked three deaths during the clinical trial to lecanemab, though it is unclear whether it caused the deaths.
Dr. Fillit notes that the first two people who died were on blood thinners when they received lecanemab.
“There are things about the use of the drug in the real world that we need to work out, especially in the context of people with comorbidities,” he says.
The third death is a little different, Dr. Fillit says. The patient, who had a stroke, showed signs of vasculitis, or inflammation of the blood vessels.
“We don’t know exactly what happened, but we do know it was very, very rare” among the people involved in the trials, he says.
Dr. Edelmayer says that the most common reported side effects during the trials were infusion-related reactions, headache, and amyloid-related imaging abnormalities (ARIA). According to the FDA, these abnormalities “are known to occur with antibodies of this class. ARIA usually does not have symptoms, although serious and life-threatening events rarely may occur.”
The FDA has added these as warnings to the drug’s label, describing the possible infusion-related reactions as flu-like symptoms, nausea, vomiting, and changes in blood pressure.
How much will it cost?
Eisai says that lecanemab will cost $26,500 a year.
In a draft report released in December, the Institute for Clinical and Economic Review said a price ranging from $8,500 to $20,600 a year would make the drug cost-effective. While the group has no authority to set prices, many large health insurers consider its reports when they negotiate prices and some drugmakers take into account ICER’s recommendations when setting prices.
An editorial in The Lancet last month warns that the cost will likely be “prohibitive” for low- and middle-income countries and many health systems don’t have the infrastructure for a widespread rollout.
Will Medicare cover it?
The Centers for Medicare & Medicaid Services, which runs Medicare, which covers most people with Alzheimer’s, has indicated it won’t broadly cover amyloid-lowering drugs until the drug gets full U.S. approval based on clinical benefits, as opposed to accelerated approval.
That means people would have to pay thousands out of pocket at first to get it.
The CMS decision effectively denies Medicare coverage of fast-tracked FDA-approved medications for Alzheimer’s disease unless the person is enrolled in an approved clinical trial.
On Dec. 19, the Alzheimer’s Association filed a formal request asking CMS to remove the trial-only requirement and provide full and unrestricted coverage for FDA-approved Alzheimer’s treatments.
CMS says in a statement issued after the announcement: “Because Eisai’s product, lecanemab, was granted accelerated approval by the FDA, it falls under CMS’s existing national coverage determination. CMS is examining available information and may reconsider its current coverage based on this review.”
“If lecanemab subsequently receives traditional FDA approval, CMS would provide broader coverage,” the statement says.
Who benefits most from this drug?
Lecanemab is a treatment for people with early-stage Alzheimer’s disease who have amyloid in their brain. This means people with other types of dementia, or those in the later stages of Alzheimer’s disease, are not likely to improve with this drug.
Who makes lecanemab?
Japan-based Eisai is developing the drug, a monoclonal antibody, in collaboration with the U.S. company Biogen.
What’s the Alzheimer’s Association’s view?
The association urged accelerated FDA approval. In a statement, it says it “welcomes and is further encouraged” by the clinical trial results.
It says data published in the New England Journal of Medicine confirms lecanemab “can meaningfully change the course of the disease for people in the earliest stages of Alzheimer’s disease.”
“We are energized at the progress we are seeing in the research pipeline. The science is telling us that although antiamyloid treatments are not a cure – they are not going to be the end of treating Alzheimer’s – they are certainly the beginning,” Dr. Edelmayer says.
Are there alternatives?
The FDA gave accelerated approval to Biogen to produce another drug for Alzheimer’s, Aduhelm (aducanemab), in 2021, but the move was controversial as the drug’s effectiveness was widely questioned. It has since largely been pulled from the market.
Aduhelm had been the first approved early-stage Alzheimer’s treatment since 2003.
A version of this article first appeared on WebMD.com.
, offering hope where there has been little for patients and their families affected by the devastating disease.
More than 6 million people in the United States live with Alzheimer’s.
It’s not a cure, but the drug, given intravenously every 2 weeks, has shown moderate positive effects in clinical trials in slowing early-stage disease.
But many are wary. As explained in an editorial in the journal The Lancet, “The Alzheimer’s disease community has become accustomed to false hope, disappointment, and controversy.”
Some worry about lecanemab’s safety as some people in clinical trials experienced serious side effects of bleeding and swelling in the brain. Scientists recently attributed a third death to lecanemab, brand name Leqembi, though the drugmaker disputed the medication was the cause.
So what should patients and their families make of this news? Here we answer some of the top questions surrounding the drug.
What does the FDA action mean?
The FDA granted accelerated approval to Leqembi after it showed positive trial results in slowing the progression of early-stage disease.
The FDA can grant accelerated approval for drugs that treat serious conditions and fill an unmet medical need while drugs continue to be studied in larger trials.
With the FDA approval in hand, doctors can now prescribe the medication.
Rebecca Edelmayer, PhD, the Alzheimer’s Association senior director of scientific engagement, says that with the FDA’s move, ramping up manufacturing – and eventually nationwide distribution and implementation – will take some time.
“Ask your doctor about availability,” she says. “The main issue is that, without insurance and Medicare coverage of this class of treatments, access for those who could benefit from the newly approved treatment will only be available to those who can pay out-of-pocket. Without coverage, people simply won’t be able to get the treatment.”
The Washington Post reports that with accelerated approval, drugmaker Eisai is expected to immediately apply for full FDA approval, which wouldn’t be likely to come before later this year. Full approval could help clear the path for Medicare coverage of the drug.
Potential benefit?
Those who got Leqembi in a clinical trial for 18 months experienced 27% less decline in memory and thinking relative to the group who got a placebo. It also reduced amyloid in the brain, the sticky protein that builds up in the brains of people with Alzheimer’s and is considered a hallmark of the disease.
Howard Fillit, MD, cofounder and chief science officer of the Alzheimer’s Drug Discovery Foundation, says, “It’s the first phase 3 study in our field of a disease-modifying drug where the clinical efficacy was very clear.”
Concerns about side effects
The drug has raised safety concerns as it has been linked with certain serious adverse events, including brain swelling and bleeding. In the trial, 14% of patients who received the drug experienced side effects that included brain swelling and bleeding, compared with about 11% in the placebo group.
Scientists have reportedly linked three deaths during the clinical trial to lecanemab, though it is unclear whether it caused the deaths.
Dr. Fillit notes that the first two people who died were on blood thinners when they received lecanemab.
“There are things about the use of the drug in the real world that we need to work out, especially in the context of people with comorbidities,” he says.
The third death is a little different, Dr. Fillit says. The patient, who had a stroke, showed signs of vasculitis, or inflammation of the blood vessels.
“We don’t know exactly what happened, but we do know it was very, very rare” among the people involved in the trials, he says.
Dr. Edelmayer says that the most common reported side effects during the trials were infusion-related reactions, headache, and amyloid-related imaging abnormalities (ARIA). According to the FDA, these abnormalities “are known to occur with antibodies of this class. ARIA usually does not have symptoms, although serious and life-threatening events rarely may occur.”
The FDA has added these as warnings to the drug’s label, describing the possible infusion-related reactions as flu-like symptoms, nausea, vomiting, and changes in blood pressure.
How much will it cost?
Eisai says that lecanemab will cost $26,500 a year.
In a draft report released in December, the Institute for Clinical and Economic Review said a price ranging from $8,500 to $20,600 a year would make the drug cost-effective. While the group has no authority to set prices, many large health insurers consider its reports when they negotiate prices and some drugmakers take into account ICER’s recommendations when setting prices.
An editorial in The Lancet last month warns that the cost will likely be “prohibitive” for low- and middle-income countries and many health systems don’t have the infrastructure for a widespread rollout.
Will Medicare cover it?
The Centers for Medicare & Medicaid Services, which runs Medicare, which covers most people with Alzheimer’s, has indicated it won’t broadly cover amyloid-lowering drugs until the drug gets full U.S. approval based on clinical benefits, as opposed to accelerated approval.
That means people would have to pay thousands out of pocket at first to get it.
The CMS decision effectively denies Medicare coverage of fast-tracked FDA-approved medications for Alzheimer’s disease unless the person is enrolled in an approved clinical trial.
On Dec. 19, the Alzheimer’s Association filed a formal request asking CMS to remove the trial-only requirement and provide full and unrestricted coverage for FDA-approved Alzheimer’s treatments.
CMS says in a statement issued after the announcement: “Because Eisai’s product, lecanemab, was granted accelerated approval by the FDA, it falls under CMS’s existing national coverage determination. CMS is examining available information and may reconsider its current coverage based on this review.”
“If lecanemab subsequently receives traditional FDA approval, CMS would provide broader coverage,” the statement says.
Who benefits most from this drug?
Lecanemab is a treatment for people with early-stage Alzheimer’s disease who have amyloid in their brain. This means people with other types of dementia, or those in the later stages of Alzheimer’s disease, are not likely to improve with this drug.
Who makes lecanemab?
Japan-based Eisai is developing the drug, a monoclonal antibody, in collaboration with the U.S. company Biogen.
What’s the Alzheimer’s Association’s view?
The association urged accelerated FDA approval. In a statement, it says it “welcomes and is further encouraged” by the clinical trial results.
It says data published in the New England Journal of Medicine confirms lecanemab “can meaningfully change the course of the disease for people in the earliest stages of Alzheimer’s disease.”
“We are energized at the progress we are seeing in the research pipeline. The science is telling us that although antiamyloid treatments are not a cure – they are not going to be the end of treating Alzheimer’s – they are certainly the beginning,” Dr. Edelmayer says.
Are there alternatives?
The FDA gave accelerated approval to Biogen to produce another drug for Alzheimer’s, Aduhelm (aducanemab), in 2021, but the move was controversial as the drug’s effectiveness was widely questioned. It has since largely been pulled from the market.
Aduhelm had been the first approved early-stage Alzheimer’s treatment since 2003.
A version of this article first appeared on WebMD.com.
Screen all patients for cannabis use before surgery: Guideline
All patients who undergo procedures that require regional or general anesthesia should be asked if, how often, and in what forms they use the drug, according to recommendations from the American Society of Regional Anesthesia and Pain Medicine.
One reason: Patients who regularly use cannabis may experience worse pain and nausea after surgery and may require more opioid analgesia, the group said.
The society’s recommendations – published in Regional Anesthesia and Pain Medicine – are the first guidelines in the United States to cover cannabis use as it relates to surgery, the group said.
Possible interactions
Use of cannabis has increased in recent years, and researchers have been concerned that the drug may interact with anesthesia and complicate pain management. Few studies have evaluated interactions between cannabis and anesthetic agents, however, according to the authors of the new guidelines.
“With the rising prevalence of both medical and recreational cannabis use in the general population, anesthesiologists, surgeons, and perioperative physicians must have an understanding of the effects of cannabis on physiology in order to provide safe perioperative care,” the guideline said.
“Before surgery, anesthesiologists should ask patients if they use cannabis – whether medicinally or recreationally – and be prepared to possibly change the anesthesia plan or delay the procedure in certain situations,” Samer Narouze, MD, PhD, ASRA president and senior author of the guidelines, said in a news release about the recommendations.
Although some patients may use cannabis to relieve pain, research shows that “regular users may have more pain and nausea after surgery, not less, and may need more medications, including opioids, to manage the discomfort,” said Dr. Narouze, chairman of the Center for Pain Medicine at Western Reserve Hospital in Cuyahoga Falls, Ohio.
Risks for vomiting, heart attack
The new recommendations were created by a committee of 13 experts, including anesthesiologists, chronic pain physicians, and a patient advocate. Shalini Shah, MD, vice chair of anesthesiology at the University of California, Irvine, was lead author of the document.
Four of 21 recommendations were classified as grade A, meaning that following them would be expected to provide substantial benefits. Those recommendations are to screen all patients before surgery; postpone elective surgery for patients who have altered mental status or impaired decision-making capacity at the time of surgery; counsel frequent, heavy users about the potential for cannabis use to impair postoperative pain control; and counsel pregnant patients about the risks of cannabis use to unborn children.
The authors cited studies to support their recommendations, including one showing that long-term cannabis use was associated with a 20% increase in the incidence of postoperative nausea and vomiting, a leading complaint of surgery patients. Other research has shown that cannabis use is linked to more pain and use of opioids after surgery.
Other recommendations include delaying elective surgery for at least 2 hours after a patient has smoked cannabis, owing to an increased risk for heart attack, and considering adjustment of ventilation settings during surgery for regular smokers of cannabis. Research has shown that smoking cannabis may be a rare trigger for myocardial infarction and is associated with airway inflammation and self-reported respiratory symptoms.
Nevertheless, doctors should not conduct universal toxicology screening, given a lack of evidence supporting this practice, the guideline stated.
The authors did not have enough information to make recommendations about reducing cannabis use before surgery or adjusting opioid prescriptions after surgery for patients who use cannabis, they said.
Kenneth Finn, MD, president of the American Board of Pain Medicine, welcomed the publication of the new guidelines. Dr. Finn, who practices at Springs Rehabilitation in Colorado Springs, has edited a textbook about cannabis in medicine and founded the International Academy on the Science and Impact of Cannabis.
“The vast majority of medical providers really have no idea about cannabis and what its impacts are on the human body,” Dr. Finn said.
For one, it can interact with numerous other drugs, including warfarin.
Guideline coauthor Eugene R. Viscusi, MD, professor of anesthesiology at the Sidney Kimmel Medical College, Philadelphia, emphasized that, while cannabis may be perceived as “natural,” it should not be considered differently from manufactured drugs.
Cannabis and cannabinoids represent “a class of very potent and pharmacologically active compounds,” Dr. Viscusi said in an interview. While researchers continue to assess possible medically beneficial effects of cannabis compounds, clinicians also need to be aware of the risks.
“The literature continues to emerge, and while we are always hopeful for good news, as physicians, we need to be very well versed on potential risks, especially in a high-risk situation like surgery,” he said.
Dr. Shah has consulted for companies that develop medical devices and drugs. Dr. Finn is the editor of the textbook, “Cannabis in Medicine: An Evidence-Based Approach” (Springer: New York, 2020), for which he receives royalties.
A version of this article first appeared on Medscape.com.
All patients who undergo procedures that require regional or general anesthesia should be asked if, how often, and in what forms they use the drug, according to recommendations from the American Society of Regional Anesthesia and Pain Medicine.
One reason: Patients who regularly use cannabis may experience worse pain and nausea after surgery and may require more opioid analgesia, the group said.
The society’s recommendations – published in Regional Anesthesia and Pain Medicine – are the first guidelines in the United States to cover cannabis use as it relates to surgery, the group said.
Possible interactions
Use of cannabis has increased in recent years, and researchers have been concerned that the drug may interact with anesthesia and complicate pain management. Few studies have evaluated interactions between cannabis and anesthetic agents, however, according to the authors of the new guidelines.
“With the rising prevalence of both medical and recreational cannabis use in the general population, anesthesiologists, surgeons, and perioperative physicians must have an understanding of the effects of cannabis on physiology in order to provide safe perioperative care,” the guideline said.
“Before surgery, anesthesiologists should ask patients if they use cannabis – whether medicinally or recreationally – and be prepared to possibly change the anesthesia plan or delay the procedure in certain situations,” Samer Narouze, MD, PhD, ASRA president and senior author of the guidelines, said in a news release about the recommendations.
Although some patients may use cannabis to relieve pain, research shows that “regular users may have more pain and nausea after surgery, not less, and may need more medications, including opioids, to manage the discomfort,” said Dr. Narouze, chairman of the Center for Pain Medicine at Western Reserve Hospital in Cuyahoga Falls, Ohio.
Risks for vomiting, heart attack
The new recommendations were created by a committee of 13 experts, including anesthesiologists, chronic pain physicians, and a patient advocate. Shalini Shah, MD, vice chair of anesthesiology at the University of California, Irvine, was lead author of the document.
Four of 21 recommendations were classified as grade A, meaning that following them would be expected to provide substantial benefits. Those recommendations are to screen all patients before surgery; postpone elective surgery for patients who have altered mental status or impaired decision-making capacity at the time of surgery; counsel frequent, heavy users about the potential for cannabis use to impair postoperative pain control; and counsel pregnant patients about the risks of cannabis use to unborn children.
The authors cited studies to support their recommendations, including one showing that long-term cannabis use was associated with a 20% increase in the incidence of postoperative nausea and vomiting, a leading complaint of surgery patients. Other research has shown that cannabis use is linked to more pain and use of opioids after surgery.
Other recommendations include delaying elective surgery for at least 2 hours after a patient has smoked cannabis, owing to an increased risk for heart attack, and considering adjustment of ventilation settings during surgery for regular smokers of cannabis. Research has shown that smoking cannabis may be a rare trigger for myocardial infarction and is associated with airway inflammation and self-reported respiratory symptoms.
Nevertheless, doctors should not conduct universal toxicology screening, given a lack of evidence supporting this practice, the guideline stated.
The authors did not have enough information to make recommendations about reducing cannabis use before surgery or adjusting opioid prescriptions after surgery for patients who use cannabis, they said.
Kenneth Finn, MD, president of the American Board of Pain Medicine, welcomed the publication of the new guidelines. Dr. Finn, who practices at Springs Rehabilitation in Colorado Springs, has edited a textbook about cannabis in medicine and founded the International Academy on the Science and Impact of Cannabis.
“The vast majority of medical providers really have no idea about cannabis and what its impacts are on the human body,” Dr. Finn said.
For one, it can interact with numerous other drugs, including warfarin.
Guideline coauthor Eugene R. Viscusi, MD, professor of anesthesiology at the Sidney Kimmel Medical College, Philadelphia, emphasized that, while cannabis may be perceived as “natural,” it should not be considered differently from manufactured drugs.
Cannabis and cannabinoids represent “a class of very potent and pharmacologically active compounds,” Dr. Viscusi said in an interview. While researchers continue to assess possible medically beneficial effects of cannabis compounds, clinicians also need to be aware of the risks.
“The literature continues to emerge, and while we are always hopeful for good news, as physicians, we need to be very well versed on potential risks, especially in a high-risk situation like surgery,” he said.
Dr. Shah has consulted for companies that develop medical devices and drugs. Dr. Finn is the editor of the textbook, “Cannabis in Medicine: An Evidence-Based Approach” (Springer: New York, 2020), for which he receives royalties.
A version of this article first appeared on Medscape.com.
All patients who undergo procedures that require regional or general anesthesia should be asked if, how often, and in what forms they use the drug, according to recommendations from the American Society of Regional Anesthesia and Pain Medicine.
One reason: Patients who regularly use cannabis may experience worse pain and nausea after surgery and may require more opioid analgesia, the group said.
The society’s recommendations – published in Regional Anesthesia and Pain Medicine – are the first guidelines in the United States to cover cannabis use as it relates to surgery, the group said.
Possible interactions
Use of cannabis has increased in recent years, and researchers have been concerned that the drug may interact with anesthesia and complicate pain management. Few studies have evaluated interactions between cannabis and anesthetic agents, however, according to the authors of the new guidelines.
“With the rising prevalence of both medical and recreational cannabis use in the general population, anesthesiologists, surgeons, and perioperative physicians must have an understanding of the effects of cannabis on physiology in order to provide safe perioperative care,” the guideline said.
“Before surgery, anesthesiologists should ask patients if they use cannabis – whether medicinally or recreationally – and be prepared to possibly change the anesthesia plan or delay the procedure in certain situations,” Samer Narouze, MD, PhD, ASRA president and senior author of the guidelines, said in a news release about the recommendations.
Although some patients may use cannabis to relieve pain, research shows that “regular users may have more pain and nausea after surgery, not less, and may need more medications, including opioids, to manage the discomfort,” said Dr. Narouze, chairman of the Center for Pain Medicine at Western Reserve Hospital in Cuyahoga Falls, Ohio.
Risks for vomiting, heart attack
The new recommendations were created by a committee of 13 experts, including anesthesiologists, chronic pain physicians, and a patient advocate. Shalini Shah, MD, vice chair of anesthesiology at the University of California, Irvine, was lead author of the document.
Four of 21 recommendations were classified as grade A, meaning that following them would be expected to provide substantial benefits. Those recommendations are to screen all patients before surgery; postpone elective surgery for patients who have altered mental status or impaired decision-making capacity at the time of surgery; counsel frequent, heavy users about the potential for cannabis use to impair postoperative pain control; and counsel pregnant patients about the risks of cannabis use to unborn children.
The authors cited studies to support their recommendations, including one showing that long-term cannabis use was associated with a 20% increase in the incidence of postoperative nausea and vomiting, a leading complaint of surgery patients. Other research has shown that cannabis use is linked to more pain and use of opioids after surgery.
Other recommendations include delaying elective surgery for at least 2 hours after a patient has smoked cannabis, owing to an increased risk for heart attack, and considering adjustment of ventilation settings during surgery for regular smokers of cannabis. Research has shown that smoking cannabis may be a rare trigger for myocardial infarction and is associated with airway inflammation and self-reported respiratory symptoms.
Nevertheless, doctors should not conduct universal toxicology screening, given a lack of evidence supporting this practice, the guideline stated.
The authors did not have enough information to make recommendations about reducing cannabis use before surgery or adjusting opioid prescriptions after surgery for patients who use cannabis, they said.
Kenneth Finn, MD, president of the American Board of Pain Medicine, welcomed the publication of the new guidelines. Dr. Finn, who practices at Springs Rehabilitation in Colorado Springs, has edited a textbook about cannabis in medicine and founded the International Academy on the Science and Impact of Cannabis.
“The vast majority of medical providers really have no idea about cannabis and what its impacts are on the human body,” Dr. Finn said.
For one, it can interact with numerous other drugs, including warfarin.
Guideline coauthor Eugene R. Viscusi, MD, professor of anesthesiology at the Sidney Kimmel Medical College, Philadelphia, emphasized that, while cannabis may be perceived as “natural,” it should not be considered differently from manufactured drugs.
Cannabis and cannabinoids represent “a class of very potent and pharmacologically active compounds,” Dr. Viscusi said in an interview. While researchers continue to assess possible medically beneficial effects of cannabis compounds, clinicians also need to be aware of the risks.
“The literature continues to emerge, and while we are always hopeful for good news, as physicians, we need to be very well versed on potential risks, especially in a high-risk situation like surgery,” he said.
Dr. Shah has consulted for companies that develop medical devices and drugs. Dr. Finn is the editor of the textbook, “Cannabis in Medicine: An Evidence-Based Approach” (Springer: New York, 2020), for which he receives royalties.
A version of this article first appeared on Medscape.com.
FROM REGIONAL ANETHESIA AND MEDICINE



