User login
VIDEO: What’s new on atopic dermatitis drugs and cancer concerns?
WAIKOLOA, HAWAII – Topical calcineurin inhibitors’ boxed warnings give many patients and physicians pause over cancer concerns – but a new database analysis may put some minds at ease about the drugs’ use for atopic dermatitis.
“Pimecrolimus and tacrolimus are given topically, not internally – very little absorption occurs. So, it was hoped that ... we wouldn’t see cancer increases in these patients,” explained Dr. Joseph F. Fowler Jr., clinical professor of dermatology at the University of Louisville (Ky.). “And in fact, that’s exactly what was shown in this large study.”
In an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation, Dr. Fowler discussed the data from new research examining cancer incidence and calcineurin inhibitor use.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WAIKOLOA, HAWAII – Topical calcineurin inhibitors’ boxed warnings give many patients and physicians pause over cancer concerns – but a new database analysis may put some minds at ease about the drugs’ use for atopic dermatitis.
“Pimecrolimus and tacrolimus are given topically, not internally – very little absorption occurs. So, it was hoped that ... we wouldn’t see cancer increases in these patients,” explained Dr. Joseph F. Fowler Jr., clinical professor of dermatology at the University of Louisville (Ky.). “And in fact, that’s exactly what was shown in this large study.”
In an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation, Dr. Fowler discussed the data from new research examining cancer incidence and calcineurin inhibitor use.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WAIKOLOA, HAWAII – Topical calcineurin inhibitors’ boxed warnings give many patients and physicians pause over cancer concerns – but a new database analysis may put some minds at ease about the drugs’ use for atopic dermatitis.
“Pimecrolimus and tacrolimus are given topically, not internally – very little absorption occurs. So, it was hoped that ... we wouldn’t see cancer increases in these patients,” explained Dr. Joseph F. Fowler Jr., clinical professor of dermatology at the University of Louisville (Ky.). “And in fact, that’s exactly what was shown in this large study.”
In an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation, Dr. Fowler discussed the data from new research examining cancer incidence and calcineurin inhibitor use.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SDEF HAWAII DERMATOLOGY SEMINAR
When the Doctor Is Not a Doctor

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.
New tanning dependence screening tool ‘promising’
The validity of a new screening tool used to identify people who are addicted to indoor tanning has been confirmed in a recent study.
In background information to their study, the researchers, led by Jerod L. Stapleton of the Rutgers Cancer Institute of New Jersey, said tools for assessing tanning addiction had been developed by adapting existing substance addiction screening tools. However, concerns had been raised about their validity, with some research suggesting assessment results did not correspond to actual tanning behaviors.
The Behavioral Addiction Indoor Tanning Screener (BAITS) screening tool was first described in the DSM-5. The tool was designed to capture the experience of diminished control and urges to use indoor tanning, the research team explained in a short communication published in Acta Dermato-Venereologica (2015 [doi: 10.2340/00015555-2290]).
The aim of the current study was to validate BAITS classification criteria by comparing it with the Structured Interview for Tanning Abuse and Dependence (SITAD) clinical assessment as well as indoor tanning use 6 months later.
Of the 164 participants, 81% did not agree with any BAITS items, 10% agreed with one, and 9% agreed with two or more. Those who agreed with two or more BAITS items were highly likely to be diagnosed as tanning dependent on the SITAD (73%), compared with those with one response (25%) or zero responses (1%) (P less than .001).
Participants who agreed with two or more BAITS items also reported an indoor tanning frequency at 6 months more than two and a half times higher than that of those agreeing with one item, and more than four and a half times higher than that of participants who endorsed no items.
“The BAITS represents a promising screening tool for symptoms of tanning addiction with preliminary evidence of validity,“ the research team wrote.
The BAITS could be used to identify patients in need of counseling to avoid indoor tanning, they suggested. “Although there is a lack of behavioral interventions targeted to individuals who are addicted to tanning, even brief counseling by clinicians regarding addictive behaviors, like smoking, can lead to measurable reductions in use.”
No conflicts of interest were declared. The study was funded by a grant from the National Cancer Institute.
The validity of a new screening tool used to identify people who are addicted to indoor tanning has been confirmed in a recent study.
In background information to their study, the researchers, led by Jerod L. Stapleton of the Rutgers Cancer Institute of New Jersey, said tools for assessing tanning addiction had been developed by adapting existing substance addiction screening tools. However, concerns had been raised about their validity, with some research suggesting assessment results did not correspond to actual tanning behaviors.
The Behavioral Addiction Indoor Tanning Screener (BAITS) screening tool was first described in the DSM-5. The tool was designed to capture the experience of diminished control and urges to use indoor tanning, the research team explained in a short communication published in Acta Dermato-Venereologica (2015 [doi: 10.2340/00015555-2290]).
The aim of the current study was to validate BAITS classification criteria by comparing it with the Structured Interview for Tanning Abuse and Dependence (SITAD) clinical assessment as well as indoor tanning use 6 months later.
Of the 164 participants, 81% did not agree with any BAITS items, 10% agreed with one, and 9% agreed with two or more. Those who agreed with two or more BAITS items were highly likely to be diagnosed as tanning dependent on the SITAD (73%), compared with those with one response (25%) or zero responses (1%) (P less than .001).
Participants who agreed with two or more BAITS items also reported an indoor tanning frequency at 6 months more than two and a half times higher than that of those agreeing with one item, and more than four and a half times higher than that of participants who endorsed no items.
“The BAITS represents a promising screening tool for symptoms of tanning addiction with preliminary evidence of validity,“ the research team wrote.
The BAITS could be used to identify patients in need of counseling to avoid indoor tanning, they suggested. “Although there is a lack of behavioral interventions targeted to individuals who are addicted to tanning, even brief counseling by clinicians regarding addictive behaviors, like smoking, can lead to measurable reductions in use.”
No conflicts of interest were declared. The study was funded by a grant from the National Cancer Institute.
The validity of a new screening tool used to identify people who are addicted to indoor tanning has been confirmed in a recent study.
In background information to their study, the researchers, led by Jerod L. Stapleton of the Rutgers Cancer Institute of New Jersey, said tools for assessing tanning addiction had been developed by adapting existing substance addiction screening tools. However, concerns had been raised about their validity, with some research suggesting assessment results did not correspond to actual tanning behaviors.
The Behavioral Addiction Indoor Tanning Screener (BAITS) screening tool was first described in the DSM-5. The tool was designed to capture the experience of diminished control and urges to use indoor tanning, the research team explained in a short communication published in Acta Dermato-Venereologica (2015 [doi: 10.2340/00015555-2290]).
The aim of the current study was to validate BAITS classification criteria by comparing it with the Structured Interview for Tanning Abuse and Dependence (SITAD) clinical assessment as well as indoor tanning use 6 months later.
Of the 164 participants, 81% did not agree with any BAITS items, 10% agreed with one, and 9% agreed with two or more. Those who agreed with two or more BAITS items were highly likely to be diagnosed as tanning dependent on the SITAD (73%), compared with those with one response (25%) or zero responses (1%) (P less than .001).
Participants who agreed with two or more BAITS items also reported an indoor tanning frequency at 6 months more than two and a half times higher than that of those agreeing with one item, and more than four and a half times higher than that of participants who endorsed no items.
“The BAITS represents a promising screening tool for symptoms of tanning addiction with preliminary evidence of validity,“ the research team wrote.
The BAITS could be used to identify patients in need of counseling to avoid indoor tanning, they suggested. “Although there is a lack of behavioral interventions targeted to individuals who are addicted to tanning, even brief counseling by clinicians regarding addictive behaviors, like smoking, can lead to measurable reductions in use.”
No conflicts of interest were declared. The study was funded by a grant from the National Cancer Institute.
FROM ACTA DERMATO-VENEREOLOGICA
Key clinical point: A new screening tool for symptoms of tanning addiction has shown preliminary evidence of validity.
Major finding: Participants who agreed with two or more BAITS items were highly likely to be diagnosed as tanning dependent on the SITAD (73%), compared with those with one response (25%) or zero responses (1%).
Data source: An online survey that included the BAITS screening tool sent to a random sample of 700 students, and a follow-up survey at 6 months. Overall, 164 participants were included in the study.
Disclosures: No conflicts of interest were declared. The study was funded by a grant from the National Cancer Institute.
Average person with atopic dermatitis has no increased risk of actinic keratosis or nonmelanoma skin cancer
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer.
Major finding: In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more actinic keratoses than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted OR, 0.71) and squamous cell cancer (adjusted OR, 1.54).
Data source: A prospective, Dutch population-based cohort study of 4,375 participants who had undergone full body skin examinations.
Disclosures: The investigators said they had no relevant financial disclosures.
Young women who tan indoors are at two- to sixfold increased risk of melanoma
Women under age 50 years who regularly tanned indoors were at a twofold to sixfold increased risk of melanoma, compared with age-matched women who had not engaged in indoor tanning, according to a population-based control study.
The 681 individuals with melanoma, drawn from the Skin Health Study, a population-based case-control study evaluating the indoor tanning and the risk of melanoma, conducted in Minnesota, were diagnosed during 2004-2007. Of these, 68% were women; of the 654 controls, 68% were women. For the women younger than 30 years who had ever tanned indoors, compared with controls, the crude odds ratio of developing melanoma was 6.0; for women aged 30-39 years, the adjusted odds ratio was 3.5; and for women aged 40-49 years, the adjusted odds ratio was 2.3.
Indoor tanning was most common in 95% of women in the case group who were younger than 40 years, compared with 81% of controls of the same age (P less than .001), although “The median hours of routine and recreational sun exposure were fairly similar among females in both groups.” Women younger than age 40 years at diagnosis or reference age started tanning indoors at a median age of 16 years, compared with 25 years among women aged 40 years and older (P less than .001). The results were reported on line on Jan. 27 (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermaool.2015.2938).
“Given the substantial proportion of young women today who began indoor tanning as adolescents, this result is particularly concerning because their risk of developing melanoma in the future may be very high,” said DeAnn Lazovich, Ph.D., of the University of Minnesota, Minneapolis, and her associates.
Nearly all women who developed melanoma who were younger than 30 at diagnosis reported tanning indoors more than 10 times. In the older age groups (30-50 years), women were nearly three to four times more likely to develop melanoma if they reported tanning indoors more than 10 times. The strongest association between use of indoor tanning and melanoma was observed among women who developed melanoma on their trunk, with an odds ratio of 3.7.
In men, the strength of the association between indoor tanning and melanoma was variable. This was likely because of the small sample size for some age groups, Dr. Lazovich and her associates said. There was no clear pattern between the age at initiation of indoor tanning or a dose response in relation to development of melanoma in men depending on their age at diagnosis.

Melanoma rates generally are higher in women than in men until about age 50 years, but rates have been increasing in both younger men and women. In 1995, the rising rates seemed to diverge by sex, but in 2006, the incidence of melanoma started to increase more steeply in younger women than men, according to some sources.
This current analysis “provides evidence that indoor tanning is likely driver of diverging trends in men and women younger than 50 years in the United States,” the authors wrote.
The study was supported by National Institutes of Health grants. No conflicts of interest were reported.
In an accompanying editorial, the authors pointed out that the study found that “nearly all women in the study (96.8%) diagnosed with melanoma [while] younger than 30 years had engaged in indoor tanning, all initiating indoor tanning before age 25, and nearly all (90.5%) engaging in frequent indoor tanning,” and that the results build “on the previous literature and demonstrates the importance of public health efforts in reducing indoor tanning.”
By June 2015, “42 states had laws addressing youth access: 13 states restricted indoor tanning among minors (less than 18 years), 12 states prohibited minors at a younger age (e.g., 14-17 years), and 17 states required parents to give permission or to accompany minors while indoor tanning,” which have had some effect, they wrote.
“Indoor tanning among female high school students decreased from 25.4% in 2009 to 20.2% in 2013. Declines have also been noted among adults, with national rates decreasing from 8.6% to 6.5% among women and from 2.2% to 1.7% among men between 2010 and 2013. Despite these reductions, an estimated 11.3 million Americans continue to engage in indoor tanning each year, [more than] 85% (9.7 million) of whom are adults.”
Areas of concerns, however, are indoor tanning facilities available at college and university campuses, direct sale of indoor tanning devices to the public “and their use in unsupervised settings,” and deceptive advertising practices by the indoor tanning industry, they added.
These comments were excerpted from an editorial by Gery P. Guy Jr., Ph.D., of the division of cancer prevention and control, Centers for Disease Control and Prevention, and his associates (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermatol.2015.3007). The authors said they had no conflicts of interest.
In an accompanying editorial, the authors pointed out that the study found that “nearly all women in the study (96.8%) diagnosed with melanoma [while] younger than 30 years had engaged in indoor tanning, all initiating indoor tanning before age 25, and nearly all (90.5%) engaging in frequent indoor tanning,” and that the results build “on the previous literature and demonstrates the importance of public health efforts in reducing indoor tanning.”
By June 2015, “42 states had laws addressing youth access: 13 states restricted indoor tanning among minors (less than 18 years), 12 states prohibited minors at a younger age (e.g., 14-17 years), and 17 states required parents to give permission or to accompany minors while indoor tanning,” which have had some effect, they wrote.
“Indoor tanning among female high school students decreased from 25.4% in 2009 to 20.2% in 2013. Declines have also been noted among adults, with national rates decreasing from 8.6% to 6.5% among women and from 2.2% to 1.7% among men between 2010 and 2013. Despite these reductions, an estimated 11.3 million Americans continue to engage in indoor tanning each year, [more than] 85% (9.7 million) of whom are adults.”
Areas of concerns, however, are indoor tanning facilities available at college and university campuses, direct sale of indoor tanning devices to the public “and their use in unsupervised settings,” and deceptive advertising practices by the indoor tanning industry, they added.
These comments were excerpted from an editorial by Gery P. Guy Jr., Ph.D., of the division of cancer prevention and control, Centers for Disease Control and Prevention, and his associates (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermatol.2015.3007). The authors said they had no conflicts of interest.
In an accompanying editorial, the authors pointed out that the study found that “nearly all women in the study (96.8%) diagnosed with melanoma [while] younger than 30 years had engaged in indoor tanning, all initiating indoor tanning before age 25, and nearly all (90.5%) engaging in frequent indoor tanning,” and that the results build “on the previous literature and demonstrates the importance of public health efforts in reducing indoor tanning.”
By June 2015, “42 states had laws addressing youth access: 13 states restricted indoor tanning among minors (less than 18 years), 12 states prohibited minors at a younger age (e.g., 14-17 years), and 17 states required parents to give permission or to accompany minors while indoor tanning,” which have had some effect, they wrote.
“Indoor tanning among female high school students decreased from 25.4% in 2009 to 20.2% in 2013. Declines have also been noted among adults, with national rates decreasing from 8.6% to 6.5% among women and from 2.2% to 1.7% among men between 2010 and 2013. Despite these reductions, an estimated 11.3 million Americans continue to engage in indoor tanning each year, [more than] 85% (9.7 million) of whom are adults.”
Areas of concerns, however, are indoor tanning facilities available at college and university campuses, direct sale of indoor tanning devices to the public “and their use in unsupervised settings,” and deceptive advertising practices by the indoor tanning industry, they added.
These comments were excerpted from an editorial by Gery P. Guy Jr., Ph.D., of the division of cancer prevention and control, Centers for Disease Control and Prevention, and his associates (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermatol.2015.3007). The authors said they had no conflicts of interest.
Women under age 50 years who regularly tanned indoors were at a twofold to sixfold increased risk of melanoma, compared with age-matched women who had not engaged in indoor tanning, according to a population-based control study.
The 681 individuals with melanoma, drawn from the Skin Health Study, a population-based case-control study evaluating the indoor tanning and the risk of melanoma, conducted in Minnesota, were diagnosed during 2004-2007. Of these, 68% were women; of the 654 controls, 68% were women. For the women younger than 30 years who had ever tanned indoors, compared with controls, the crude odds ratio of developing melanoma was 6.0; for women aged 30-39 years, the adjusted odds ratio was 3.5; and for women aged 40-49 years, the adjusted odds ratio was 2.3.
Indoor tanning was most common in 95% of women in the case group who were younger than 40 years, compared with 81% of controls of the same age (P less than .001), although “The median hours of routine and recreational sun exposure were fairly similar among females in both groups.” Women younger than age 40 years at diagnosis or reference age started tanning indoors at a median age of 16 years, compared with 25 years among women aged 40 years and older (P less than .001). The results were reported on line on Jan. 27 (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermaool.2015.2938).
“Given the substantial proportion of young women today who began indoor tanning as adolescents, this result is particularly concerning because their risk of developing melanoma in the future may be very high,” said DeAnn Lazovich, Ph.D., of the University of Minnesota, Minneapolis, and her associates.
Nearly all women who developed melanoma who were younger than 30 at diagnosis reported tanning indoors more than 10 times. In the older age groups (30-50 years), women were nearly three to four times more likely to develop melanoma if they reported tanning indoors more than 10 times. The strongest association between use of indoor tanning and melanoma was observed among women who developed melanoma on their trunk, with an odds ratio of 3.7.
In men, the strength of the association between indoor tanning and melanoma was variable. This was likely because of the small sample size for some age groups, Dr. Lazovich and her associates said. There was no clear pattern between the age at initiation of indoor tanning or a dose response in relation to development of melanoma in men depending on their age at diagnosis.

Melanoma rates generally are higher in women than in men until about age 50 years, but rates have been increasing in both younger men and women. In 1995, the rising rates seemed to diverge by sex, but in 2006, the incidence of melanoma started to increase more steeply in younger women than men, according to some sources.
This current analysis “provides evidence that indoor tanning is likely driver of diverging trends in men and women younger than 50 years in the United States,” the authors wrote.
The study was supported by National Institutes of Health grants. No conflicts of interest were reported.
Women under age 50 years who regularly tanned indoors were at a twofold to sixfold increased risk of melanoma, compared with age-matched women who had not engaged in indoor tanning, according to a population-based control study.
The 681 individuals with melanoma, drawn from the Skin Health Study, a population-based case-control study evaluating the indoor tanning and the risk of melanoma, conducted in Minnesota, were diagnosed during 2004-2007. Of these, 68% were women; of the 654 controls, 68% were women. For the women younger than 30 years who had ever tanned indoors, compared with controls, the crude odds ratio of developing melanoma was 6.0; for women aged 30-39 years, the adjusted odds ratio was 3.5; and for women aged 40-49 years, the adjusted odds ratio was 2.3.
Indoor tanning was most common in 95% of women in the case group who were younger than 40 years, compared with 81% of controls of the same age (P less than .001), although “The median hours of routine and recreational sun exposure were fairly similar among females in both groups.” Women younger than age 40 years at diagnosis or reference age started tanning indoors at a median age of 16 years, compared with 25 years among women aged 40 years and older (P less than .001). The results were reported on line on Jan. 27 (JAMA Dermatol. 2016 Jan 27. doi: 10.1001/jamadermaool.2015.2938).
“Given the substantial proportion of young women today who began indoor tanning as adolescents, this result is particularly concerning because their risk of developing melanoma in the future may be very high,” said DeAnn Lazovich, Ph.D., of the University of Minnesota, Minneapolis, and her associates.
Nearly all women who developed melanoma who were younger than 30 at diagnosis reported tanning indoors more than 10 times. In the older age groups (30-50 years), women were nearly three to four times more likely to develop melanoma if they reported tanning indoors more than 10 times. The strongest association between use of indoor tanning and melanoma was observed among women who developed melanoma on their trunk, with an odds ratio of 3.7.
In men, the strength of the association between indoor tanning and melanoma was variable. This was likely because of the small sample size for some age groups, Dr. Lazovich and her associates said. There was no clear pattern between the age at initiation of indoor tanning or a dose response in relation to development of melanoma in men depending on their age at diagnosis.

Melanoma rates generally are higher in women than in men until about age 50 years, but rates have been increasing in both younger men and women. In 1995, the rising rates seemed to diverge by sex, but in 2006, the incidence of melanoma started to increase more steeply in younger women than men, according to some sources.
This current analysis “provides evidence that indoor tanning is likely driver of diverging trends in men and women younger than 50 years in the United States,” the authors wrote.
The study was supported by National Institutes of Health grants. No conflicts of interest were reported.
FROM JAMA DERMATOLOGY
Key clinical point: Women under 50 years who regularly tan indoors are at a twofold to sixfold increased risk of melanoma, compared with women the same age who don’t tan indoors.
Major finding: For the women younger than 30 years who had ever tanned indoors, compared with controls, the adjusted odds ratio of developing melanoma was 6.0; for women aged 30-39 years, it was 3.5; and for women 40-49, it was 2.3.
Data source: Drawn from the Skin Health Study, 681 melanoma patients were age- and gender-matched with 654 controls.
Disclosures: The study was supported by National Institutes of Health grants. No conflicts of interest were reported.
High-dose interferon offered no survival benefit in patients with melanoma and a single tumor–positive sentinel lymph node
Patients with melanoma and a single tumor–positive sentinel lymph node (SLN) had no improvement in overall (OS) or disease-free survival (DFS) with adjuvant high-dose interferon alfa-2b (HDI), and patients with histologically negative, RT-PCR–positive SLNs had no improvement with completion lymph node dissection (CLND) or CLND plus interferon, according to researchers.
For patients with a single positive SLN who received HDI, compared with the observation-only group, 5-year OS was 71.4% and 74.8% and 5-year DFS was 70.9% and 67.1%, respectively. For patients with reverse transcription polymerase chain reaction (RT-PCR)–positive but histologically negative SLNs who received CLND plus interferon, or CLND only, compared with the observation-only group, 5-year OS was 86.9%, 85.9%, and 85.5%, and 5-year DFS was 83.9%, 84.0%, and 79.4%, respectively.
The finding that HDI offers no survival benefit contradicts an earlier study (ECOG E1694) that compared HDI with ganglioside vaccine treatment.
“The results of this study refute the conclusion that improved DFS and OS in ECOG E1694 was due to a beneficial effect of HDI because HDI treatment in ECOG E1694 was not compared with observation or placebo, but to a vaccine that is now known to be associated with a greater risk of recurrence and mortality,” wrote Dr. Kelly M. McMasters, surgical oncologist at the University of Louisville (Ky.) and his colleagues (J Clin Oncol. 2016 Feb 8. doi: 10.1200/JCO.205.63.3776).
The prospective, randomized Sunbelt Melanoma Trial included patients with melanoma of thickness 1 mm or greater without evidence of metastasis. Protocol A, with 218 patients with histologically positive SLNs, did not meet its accrual goal of 150 patients each for arms 1 and 2. Protocol B had 556 patients with RT-PCR–positive but histologically negative SLNs. The median follow-up was 71 months.
The trial found that HDI therapy after CLND did not improve DFS or OS for patients with minimal nodal tumor burden. For patients randomly assigned to HDI versus observation after CLND, hazard ratios for DFS and OS were 0.82 (95% confidence interval, 0.50-1.36; P = .45) and 1.10 (0.69-1.76; P = .68).
In patients with stage I or II melanoma who have tumor-negative SLNs by hematoxylin and eosin histopathology and immunohistochemistry, but have molecular evidence of melanoma by RT-PCR analysis, no significant differences were observed among patients randomly assigned to CLND or CLND plus interferon treatment. Compared with observation, CLND alone showed a slight DFS improvement (hazard ratio, 0.58; 95% CI, 0.35-0.94; P = .0277), but no OS improvement (HR, 1.00; 95% CI, 0.634-1.59; P = .99). Patients who received CLND plus interferon had no significant improvement in DFS or OS, compared with observation.
Subgroup analysis showed that in patients with a single positive SLN, HDI was associated with improved DFS only in patients with ulceration (HR, 0.43; 95% CI, 0.21-0.87; P = .0183; n = 75) and with Breslow thickness more than 4 mm (HR, 0.35; 95% CI, 0.14-0.88; P = .0259; n = 42). No OS improvement was observed.
“Taken together, these data support the conclusion that the benefit of HDI is small, perhaps because only a fraction of the patient population responds to therapy,” the investigators wrote.
Patients with melanoma and a single tumor–positive sentinel lymph node (SLN) had no improvement in overall (OS) or disease-free survival (DFS) with adjuvant high-dose interferon alfa-2b (HDI), and patients with histologically negative, RT-PCR–positive SLNs had no improvement with completion lymph node dissection (CLND) or CLND plus interferon, according to researchers.
For patients with a single positive SLN who received HDI, compared with the observation-only group, 5-year OS was 71.4% and 74.8% and 5-year DFS was 70.9% and 67.1%, respectively. For patients with reverse transcription polymerase chain reaction (RT-PCR)–positive but histologically negative SLNs who received CLND plus interferon, or CLND only, compared with the observation-only group, 5-year OS was 86.9%, 85.9%, and 85.5%, and 5-year DFS was 83.9%, 84.0%, and 79.4%, respectively.
The finding that HDI offers no survival benefit contradicts an earlier study (ECOG E1694) that compared HDI with ganglioside vaccine treatment.
“The results of this study refute the conclusion that improved DFS and OS in ECOG E1694 was due to a beneficial effect of HDI because HDI treatment in ECOG E1694 was not compared with observation or placebo, but to a vaccine that is now known to be associated with a greater risk of recurrence and mortality,” wrote Dr. Kelly M. McMasters, surgical oncologist at the University of Louisville (Ky.) and his colleagues (J Clin Oncol. 2016 Feb 8. doi: 10.1200/JCO.205.63.3776).
The prospective, randomized Sunbelt Melanoma Trial included patients with melanoma of thickness 1 mm or greater without evidence of metastasis. Protocol A, with 218 patients with histologically positive SLNs, did not meet its accrual goal of 150 patients each for arms 1 and 2. Protocol B had 556 patients with RT-PCR–positive but histologically negative SLNs. The median follow-up was 71 months.
The trial found that HDI therapy after CLND did not improve DFS or OS for patients with minimal nodal tumor burden. For patients randomly assigned to HDI versus observation after CLND, hazard ratios for DFS and OS were 0.82 (95% confidence interval, 0.50-1.36; P = .45) and 1.10 (0.69-1.76; P = .68).
In patients with stage I or II melanoma who have tumor-negative SLNs by hematoxylin and eosin histopathology and immunohistochemistry, but have molecular evidence of melanoma by RT-PCR analysis, no significant differences were observed among patients randomly assigned to CLND or CLND plus interferon treatment. Compared with observation, CLND alone showed a slight DFS improvement (hazard ratio, 0.58; 95% CI, 0.35-0.94; P = .0277), but no OS improvement (HR, 1.00; 95% CI, 0.634-1.59; P = .99). Patients who received CLND plus interferon had no significant improvement in DFS or OS, compared with observation.
Subgroup analysis showed that in patients with a single positive SLN, HDI was associated with improved DFS only in patients with ulceration (HR, 0.43; 95% CI, 0.21-0.87; P = .0183; n = 75) and with Breslow thickness more than 4 mm (HR, 0.35; 95% CI, 0.14-0.88; P = .0259; n = 42). No OS improvement was observed.
“Taken together, these data support the conclusion that the benefit of HDI is small, perhaps because only a fraction of the patient population responds to therapy,” the investigators wrote.
Patients with melanoma and a single tumor–positive sentinel lymph node (SLN) had no improvement in overall (OS) or disease-free survival (DFS) with adjuvant high-dose interferon alfa-2b (HDI), and patients with histologically negative, RT-PCR–positive SLNs had no improvement with completion lymph node dissection (CLND) or CLND plus interferon, according to researchers.
For patients with a single positive SLN who received HDI, compared with the observation-only group, 5-year OS was 71.4% and 74.8% and 5-year DFS was 70.9% and 67.1%, respectively. For patients with reverse transcription polymerase chain reaction (RT-PCR)–positive but histologically negative SLNs who received CLND plus interferon, or CLND only, compared with the observation-only group, 5-year OS was 86.9%, 85.9%, and 85.5%, and 5-year DFS was 83.9%, 84.0%, and 79.4%, respectively.
The finding that HDI offers no survival benefit contradicts an earlier study (ECOG E1694) that compared HDI with ganglioside vaccine treatment.
“The results of this study refute the conclusion that improved DFS and OS in ECOG E1694 was due to a beneficial effect of HDI because HDI treatment in ECOG E1694 was not compared with observation or placebo, but to a vaccine that is now known to be associated with a greater risk of recurrence and mortality,” wrote Dr. Kelly M. McMasters, surgical oncologist at the University of Louisville (Ky.) and his colleagues (J Clin Oncol. 2016 Feb 8. doi: 10.1200/JCO.205.63.3776).
The prospective, randomized Sunbelt Melanoma Trial included patients with melanoma of thickness 1 mm or greater without evidence of metastasis. Protocol A, with 218 patients with histologically positive SLNs, did not meet its accrual goal of 150 patients each for arms 1 and 2. Protocol B had 556 patients with RT-PCR–positive but histologically negative SLNs. The median follow-up was 71 months.
The trial found that HDI therapy after CLND did not improve DFS or OS for patients with minimal nodal tumor burden. For patients randomly assigned to HDI versus observation after CLND, hazard ratios for DFS and OS were 0.82 (95% confidence interval, 0.50-1.36; P = .45) and 1.10 (0.69-1.76; P = .68).
In patients with stage I or II melanoma who have tumor-negative SLNs by hematoxylin and eosin histopathology and immunohistochemistry, but have molecular evidence of melanoma by RT-PCR analysis, no significant differences were observed among patients randomly assigned to CLND or CLND plus interferon treatment. Compared with observation, CLND alone showed a slight DFS improvement (hazard ratio, 0.58; 95% CI, 0.35-0.94; P = .0277), but no OS improvement (HR, 1.00; 95% CI, 0.634-1.59; P = .99). Patients who received CLND plus interferon had no significant improvement in DFS or OS, compared with observation.
Subgroup analysis showed that in patients with a single positive SLN, HDI was associated with improved DFS only in patients with ulceration (HR, 0.43; 95% CI, 0.21-0.87; P = .0183; n = 75) and with Breslow thickness more than 4 mm (HR, 0.35; 95% CI, 0.14-0.88; P = .0259; n = 42). No OS improvement was observed.
“Taken together, these data support the conclusion that the benefit of HDI is small, perhaps because only a fraction of the patient population responds to therapy,” the investigators wrote.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Adjuvant high-dose interferon (HDI) did not improve survival in patients with a single tumor–positive sentinel lymph node (SLN), and in patients with RT-PCR–positive but histologically negative SLNs, no survival benefit was observed with completion lymph node dissection (CLND) or adjuvant HDI.
Major finding: For patients with a single positive SLN who received HDI or observation, 5-year OS was 71.4% and 74.8% and 5-year DFS was 70.9% and 67.1%, respectively; for patients with RT-PCR–positive but histologically negative SLNs who received CLND plus interferon, CLND, or observation, 5-year OS was 86.9%, 85.9%, and 85.5%, and 5-year DFS was 83.9%, 84.0%, and 79.4%, respectively.
Data source: The prospective, randomized Sunbelt Melanoma Trial included patients with melanoma 1 mm or greater in thickness without evidence of metastasis. Protocol A had 218 patients with histologically positive SLNs and protocol B had 556 patients with RT-PCR–positive but histologically negative SLNs.
Disclosures: Dr. McMasters holds a leadership role with Provectus Biopharmaceuticals. Several of his coauthors reported ties to industry.
Books, text messages increase sun protection behaviors
An intervention consisting of text-message reminders, read-along books, and swim shirts achieved significant improvements in sun protection behaviors in children, compared with information about sun protection alone, according to the results of a randomized controlled trial.
The study, published online Feb. 8 in JAMA Pediatrics, enrolled 300 caregiver-child pairs (children aged 2-6 years), randomizing 153 to receive a read-along book emphasizing sun protection behaviors, a swim shirt, and weekly text messages asking about sun protection measures undertaken and 147 to the usual information about sun protection given at a well-child visit.
After 4 weeks, the intervention group showed significantly higher scores for sunscreen use both on sunny and cloudy days, significantly higher scores relating to wearing a shirt on sunny days, and significantly lower increases in skin melanin indices on the sun-protected upper arm, compared with the control group (JAMA Pediatr. 2016 Feb 8. doi: 10.1001/jamapediatrics.2015.4373).
“Pediatricians’ seasonal age-specific sun protection recommendations will be more effective if supported by an effective, easily accessible, multicomponent program that can be reinforced at home,” said Byron K. Ho of Northwestern University, Chicago, and his coauthors.
The study was funded by the Pediatric Sun Protection Foundation. No conflicts of interest were declared.
The choice by the investigators to educate both the caregiver and the child through the use of an active read-aloud book was a pragmatic one because this process engages both caregivers and their children to recruit each other in reinforcing recommended behaviors.
Supplying rather than simply recommending sun-protective clothing as part of the study encourages adherence by eliminating the obstacle of having families purchase the sun-protective clothing themselves, thus removing associated economic barriers.
D. Albert C. Yan and Dr. Leslie Castelo-Soccio are with the section of dermatology at the Children’s Hospital of Philadelphia and from the departments of pediatrics and dermatology at the University of Pennsylvania. These comments are excerpted from an accompanying editorial (JAMA Pediatr. Feb 8. doi: 10.1001/jamapediatrics.2015.4524). Dr. Yan declared consultancies for Galderma, Johnson & Johnson, Pierre Fabre, and Procter & Gamble. No other conflicts of interest were declared.
The choice by the investigators to educate both the caregiver and the child through the use of an active read-aloud book was a pragmatic one because this process engages both caregivers and their children to recruit each other in reinforcing recommended behaviors.
Supplying rather than simply recommending sun-protective clothing as part of the study encourages adherence by eliminating the obstacle of having families purchase the sun-protective clothing themselves, thus removing associated economic barriers.
D. Albert C. Yan and Dr. Leslie Castelo-Soccio are with the section of dermatology at the Children’s Hospital of Philadelphia and from the departments of pediatrics and dermatology at the University of Pennsylvania. These comments are excerpted from an accompanying editorial (JAMA Pediatr. Feb 8. doi: 10.1001/jamapediatrics.2015.4524). Dr. Yan declared consultancies for Galderma, Johnson & Johnson, Pierre Fabre, and Procter & Gamble. No other conflicts of interest were declared.
The choice by the investigators to educate both the caregiver and the child through the use of an active read-aloud book was a pragmatic one because this process engages both caregivers and their children to recruit each other in reinforcing recommended behaviors.
Supplying rather than simply recommending sun-protective clothing as part of the study encourages adherence by eliminating the obstacle of having families purchase the sun-protective clothing themselves, thus removing associated economic barriers.
D. Albert C. Yan and Dr. Leslie Castelo-Soccio are with the section of dermatology at the Children’s Hospital of Philadelphia and from the departments of pediatrics and dermatology at the University of Pennsylvania. These comments are excerpted from an accompanying editorial (JAMA Pediatr. Feb 8. doi: 10.1001/jamapediatrics.2015.4524). Dr. Yan declared consultancies for Galderma, Johnson & Johnson, Pierre Fabre, and Procter & Gamble. No other conflicts of interest were declared.
An intervention consisting of text-message reminders, read-along books, and swim shirts achieved significant improvements in sun protection behaviors in children, compared with information about sun protection alone, according to the results of a randomized controlled trial.
The study, published online Feb. 8 in JAMA Pediatrics, enrolled 300 caregiver-child pairs (children aged 2-6 years), randomizing 153 to receive a read-along book emphasizing sun protection behaviors, a swim shirt, and weekly text messages asking about sun protection measures undertaken and 147 to the usual information about sun protection given at a well-child visit.
After 4 weeks, the intervention group showed significantly higher scores for sunscreen use both on sunny and cloudy days, significantly higher scores relating to wearing a shirt on sunny days, and significantly lower increases in skin melanin indices on the sun-protected upper arm, compared with the control group (JAMA Pediatr. 2016 Feb 8. doi: 10.1001/jamapediatrics.2015.4373).
“Pediatricians’ seasonal age-specific sun protection recommendations will be more effective if supported by an effective, easily accessible, multicomponent program that can be reinforced at home,” said Byron K. Ho of Northwestern University, Chicago, and his coauthors.
The study was funded by the Pediatric Sun Protection Foundation. No conflicts of interest were declared.
An intervention consisting of text-message reminders, read-along books, and swim shirts achieved significant improvements in sun protection behaviors in children, compared with information about sun protection alone, according to the results of a randomized controlled trial.
The study, published online Feb. 8 in JAMA Pediatrics, enrolled 300 caregiver-child pairs (children aged 2-6 years), randomizing 153 to receive a read-along book emphasizing sun protection behaviors, a swim shirt, and weekly text messages asking about sun protection measures undertaken and 147 to the usual information about sun protection given at a well-child visit.
After 4 weeks, the intervention group showed significantly higher scores for sunscreen use both on sunny and cloudy days, significantly higher scores relating to wearing a shirt on sunny days, and significantly lower increases in skin melanin indices on the sun-protected upper arm, compared with the control group (JAMA Pediatr. 2016 Feb 8. doi: 10.1001/jamapediatrics.2015.4373).
“Pediatricians’ seasonal age-specific sun protection recommendations will be more effective if supported by an effective, easily accessible, multicomponent program that can be reinforced at home,” said Byron K. Ho of Northwestern University, Chicago, and his coauthors.
The study was funded by the Pediatric Sun Protection Foundation. No conflicts of interest were declared.
FROM JAMA PEDIATRICS
Key clinical point: An intervention of books, text-message reminders, and swim shirts can improve sun protection behaviors in children.
Major finding: A multimodal 4-week intervention aimed at increasing sun protection behavior achieved significant increases in sunscreen use and shirt wearing.
Data source: Randomized controlled study involving 300 caregiver-child pairs.
Disclosures: The study was funded by the Pediatric Sun Protection Foundation. No conflicts of interest were declared.
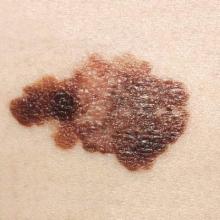
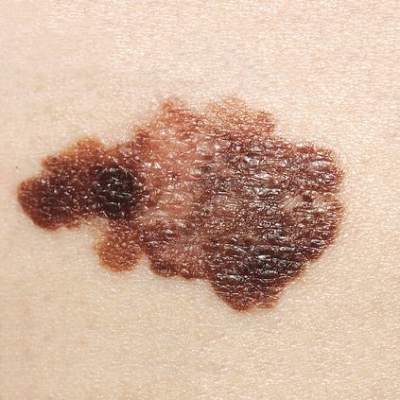
Who among us has not asked a patient to keep track of a mole?
“Keep an eye on that one, and call me if it changes,” is as much a stock phrase for dermatologists as “Wear your sunscreen.” Yet, how do patients know if a mole changes? I’m quite sure many of my patients wouldn’t notice if I shaved my head and grew a beard, let alone notice if 1 of 30 moles on their back changed color.
Mole Mapper is an iPhone app developed by the department of dermatology at Oregon Health and Science University (OHSU) to solve this problem. The app provides a framework for patients to photo, measure, and track their moles. With clear instructions, an anatomical map, and sophisticated markers, it is a significant aid for motivated patients who want medical-grade photos suitable for tracking.
To standardize the photos, the app prompts you to include a nickel, dime, or quarter in photos with nevi of interest. The user then calibrates the app by pinching onscreen circles overlying the photo such that they correspond exactly to the circumference of the coin and to the mole. Using a coin as a standard, the app then calculates the precise size of the mole regardless of the size of the photo. For example, photos taken 2 feet and 4 feet away both give the same diameter because both photos are calibrated by the dime in each.
The app was developed by a cancer biologist, Dan Webster, Ph.D., to help his wife monitor her moles between dermatology appointments. Interestingly, it was largely developed by a single person, a sign that creating apps is nearly entering into a DIY era. This increases the possibility for useful health care tools to be developed while also increasing the already crushing crowd of apps, few of which are truly useful.
The app’s functionality would not have been possible without the inclusion of Apple’s ResearchKit and Sage Bionetworks’ Bridge Server. ResearchKit provided open-source tools to facilitate informed consent over the phone and the ability to conduct participant surveys, among other activities. Bridge Server enabled the app to encrypt and securely transfer participant data from the phone to firewalled storage. The combination of these two software frameworks is paving the way for an exciting future of integrated technology and biomedical research.
According to Dan Webster, “ResearchKit is a game-changer because it provides an open-source platform for elegant informed consent, measurement tools, and participant data protection. The ability for participants to have so few barriers to contribute to a research study is the truly transformative aspect of ResearchKit, and we have seen unprecedented numbers of research study enrollees as a result.”
But that’s not all. The app is more than just a consumer tool for tracking – ResearchKit allows OHSU researchers to gather data on nevi, track them over time, and learn characteristics associated with melanoma from user-generated outcomes. This could significantly increase our understanding of melanoma and perhaps spawn an artificially intelligent app that learns to diagnose melanoma without human assistance.
Because of the institutional review board’s requirements for their research, users must be 18 years old to participate in the study. The app gracefully walks users through the consent process and even has a knowledge check at the end to ensure that they understood the risks and benefits of participating. The consent process is so streamlined that it ought to be a model for us to consent any patient for any reason.
To be clear, the app does not make diagnoses. It only provides a framework for patients to photograph their moles and track them. It also politely prompts users to rephotograph moles every 30 days so changes can be recorded.
There are apps with similar names, so be sure you have Mole Mapper from Sage Bionetworks. I tried it out to offer my experience here. Taking photos was as simple as any photo on an iPhone. Like any selfie, however, there are azimuth limits to the human arm – you can’t get shots in remote bodily corners easily. Also, placing a coin on yourself is easier said than done, unless you want to use your bubble gum to hold it in place while you take the shot. (I asked for assistance from my wife instead.)
The photos I took were accurate when compared with the measured diameter in real life, but there are still user-dependent adjustments that could lead to large artifacts. Making the measurement circles even slightly smaller or larger around the coin or the mole can lead to more than a millimeter of margins of error. If detecting melanoma requires less than 1-mm error in mole changes, then this could limit its usefulness.
Whether or not it leads to an app that automatically diagnoses melanoma from patient mole selfies, Mole Mapper has value. Any tool that empowers patients to be actively involved in their care and to meticulously monitor their moles will surely help us in keeping them safe.
Dr. Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego, and a volunteer clinical assistant professor at the University of California, San Diego. Dr. Benabio is @dermdoc on Twitter. He has no conflicts relating to the topic of this column.
“Keep an eye on that one, and call me if it changes,” is as much a stock phrase for dermatologists as “Wear your sunscreen.” Yet, how do patients know if a mole changes? I’m quite sure many of my patients wouldn’t notice if I shaved my head and grew a beard, let alone notice if 1 of 30 moles on their back changed color.
Mole Mapper is an iPhone app developed by the department of dermatology at Oregon Health and Science University (OHSU) to solve this problem. The app provides a framework for patients to photo, measure, and track their moles. With clear instructions, an anatomical map, and sophisticated markers, it is a significant aid for motivated patients who want medical-grade photos suitable for tracking.
To standardize the photos, the app prompts you to include a nickel, dime, or quarter in photos with nevi of interest. The user then calibrates the app by pinching onscreen circles overlying the photo such that they correspond exactly to the circumference of the coin and to the mole. Using a coin as a standard, the app then calculates the precise size of the mole regardless of the size of the photo. For example, photos taken 2 feet and 4 feet away both give the same diameter because both photos are calibrated by the dime in each.
The app was developed by a cancer biologist, Dan Webster, Ph.D., to help his wife monitor her moles between dermatology appointments. Interestingly, it was largely developed by a single person, a sign that creating apps is nearly entering into a DIY era. This increases the possibility for useful health care tools to be developed while also increasing the already crushing crowd of apps, few of which are truly useful.
The app’s functionality would not have been possible without the inclusion of Apple’s ResearchKit and Sage Bionetworks’ Bridge Server. ResearchKit provided open-source tools to facilitate informed consent over the phone and the ability to conduct participant surveys, among other activities. Bridge Server enabled the app to encrypt and securely transfer participant data from the phone to firewalled storage. The combination of these two software frameworks is paving the way for an exciting future of integrated technology and biomedical research.
According to Dan Webster, “ResearchKit is a game-changer because it provides an open-source platform for elegant informed consent, measurement tools, and participant data protection. The ability for participants to have so few barriers to contribute to a research study is the truly transformative aspect of ResearchKit, and we have seen unprecedented numbers of research study enrollees as a result.”
But that’s not all. The app is more than just a consumer tool for tracking – ResearchKit allows OHSU researchers to gather data on nevi, track them over time, and learn characteristics associated with melanoma from user-generated outcomes. This could significantly increase our understanding of melanoma and perhaps spawn an artificially intelligent app that learns to diagnose melanoma without human assistance.
Because of the institutional review board’s requirements for their research, users must be 18 years old to participate in the study. The app gracefully walks users through the consent process and even has a knowledge check at the end to ensure that they understood the risks and benefits of participating. The consent process is so streamlined that it ought to be a model for us to consent any patient for any reason.
To be clear, the app does not make diagnoses. It only provides a framework for patients to photograph their moles and track them. It also politely prompts users to rephotograph moles every 30 days so changes can be recorded.
There are apps with similar names, so be sure you have Mole Mapper from Sage Bionetworks. I tried it out to offer my experience here. Taking photos was as simple as any photo on an iPhone. Like any selfie, however, there are azimuth limits to the human arm – you can’t get shots in remote bodily corners easily. Also, placing a coin on yourself is easier said than done, unless you want to use your bubble gum to hold it in place while you take the shot. (I asked for assistance from my wife instead.)
The photos I took were accurate when compared with the measured diameter in real life, but there are still user-dependent adjustments that could lead to large artifacts. Making the measurement circles even slightly smaller or larger around the coin or the mole can lead to more than a millimeter of margins of error. If detecting melanoma requires less than 1-mm error in mole changes, then this could limit its usefulness.
Whether or not it leads to an app that automatically diagnoses melanoma from patient mole selfies, Mole Mapper has value. Any tool that empowers patients to be actively involved in their care and to meticulously monitor their moles will surely help us in keeping them safe.
Dr. Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego, and a volunteer clinical assistant professor at the University of California, San Diego. Dr. Benabio is @dermdoc on Twitter. He has no conflicts relating to the topic of this column.
“Keep an eye on that one, and call me if it changes,” is as much a stock phrase for dermatologists as “Wear your sunscreen.” Yet, how do patients know if a mole changes? I’m quite sure many of my patients wouldn’t notice if I shaved my head and grew a beard, let alone notice if 1 of 30 moles on their back changed color.
Mole Mapper is an iPhone app developed by the department of dermatology at Oregon Health and Science University (OHSU) to solve this problem. The app provides a framework for patients to photo, measure, and track their moles. With clear instructions, an anatomical map, and sophisticated markers, it is a significant aid for motivated patients who want medical-grade photos suitable for tracking.
To standardize the photos, the app prompts you to include a nickel, dime, or quarter in photos with nevi of interest. The user then calibrates the app by pinching onscreen circles overlying the photo such that they correspond exactly to the circumference of the coin and to the mole. Using a coin as a standard, the app then calculates the precise size of the mole regardless of the size of the photo. For example, photos taken 2 feet and 4 feet away both give the same diameter because both photos are calibrated by the dime in each.
The app was developed by a cancer biologist, Dan Webster, Ph.D., to help his wife monitor her moles between dermatology appointments. Interestingly, it was largely developed by a single person, a sign that creating apps is nearly entering into a DIY era. This increases the possibility for useful health care tools to be developed while also increasing the already crushing crowd of apps, few of which are truly useful.
The app’s functionality would not have been possible without the inclusion of Apple’s ResearchKit and Sage Bionetworks’ Bridge Server. ResearchKit provided open-source tools to facilitate informed consent over the phone and the ability to conduct participant surveys, among other activities. Bridge Server enabled the app to encrypt and securely transfer participant data from the phone to firewalled storage. The combination of these two software frameworks is paving the way for an exciting future of integrated technology and biomedical research.
According to Dan Webster, “ResearchKit is a game-changer because it provides an open-source platform for elegant informed consent, measurement tools, and participant data protection. The ability for participants to have so few barriers to contribute to a research study is the truly transformative aspect of ResearchKit, and we have seen unprecedented numbers of research study enrollees as a result.”
But that’s not all. The app is more than just a consumer tool for tracking – ResearchKit allows OHSU researchers to gather data on nevi, track them over time, and learn characteristics associated with melanoma from user-generated outcomes. This could significantly increase our understanding of melanoma and perhaps spawn an artificially intelligent app that learns to diagnose melanoma without human assistance.
Because of the institutional review board’s requirements for their research, users must be 18 years old to participate in the study. The app gracefully walks users through the consent process and even has a knowledge check at the end to ensure that they understood the risks and benefits of participating. The consent process is so streamlined that it ought to be a model for us to consent any patient for any reason.
To be clear, the app does not make diagnoses. It only provides a framework for patients to photograph their moles and track them. It also politely prompts users to rephotograph moles every 30 days so changes can be recorded.
There are apps with similar names, so be sure you have Mole Mapper from Sage Bionetworks. I tried it out to offer my experience here. Taking photos was as simple as any photo on an iPhone. Like any selfie, however, there are azimuth limits to the human arm – you can’t get shots in remote bodily corners easily. Also, placing a coin on yourself is easier said than done, unless you want to use your bubble gum to hold it in place while you take the shot. (I asked for assistance from my wife instead.)
The photos I took were accurate when compared with the measured diameter in real life, but there are still user-dependent adjustments that could lead to large artifacts. Making the measurement circles even slightly smaller or larger around the coin or the mole can lead to more than a millimeter of margins of error. If detecting melanoma requires less than 1-mm error in mole changes, then this could limit its usefulness.
Whether or not it leads to an app that automatically diagnoses melanoma from patient mole selfies, Mole Mapper has value. Any tool that empowers patients to be actively involved in their care and to meticulously monitor their moles will surely help us in keeping them safe.
Dr. Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego, and a volunteer clinical assistant professor at the University of California, San Diego. Dr. Benabio is @dermdoc on Twitter. He has no conflicts relating to the topic of this column.
Dabrafenib may be useful in patients who discontinue vemurafenib
Treatment with vemurafenib may result in severe skin toxicity, and dabrafenib appears to be useful in patients who discontinue vemurafenib due to such toxicity, according to a retrospective analysis of the cohort.
About a quarter (26%) of 131 melanoma patients treated in real life conditions with the BRAF inhibitor vemurafenib developed grade 3-4 skin toxicity, and 44% (34) of those patients permanently discontinued treatment, corresponding to 11% of the overall cohort, Dr. Lucie Peuvrel of Nantes (France) University Hospital and colleagues reported in Journal of the European Academy of Dermatology and Venereology.
Discontinuations were mainly due to rash and classic adverse skin reactions, including Stevens-Johnson syndrome, drug reaction with eosinophilia, and systemic symptoms, whereas reactions involving only photosensitivity or cutaneous carcinomas rarely resulted in treatment adjustment; 14 of the remaining 19 patients with grade 3-4 toxicity who continued treatment had no dose adjustment, and 5 had a dose reduction.
Clinical or biological abnormalities occurred in 94% of those with grade 3-4 rashes, the investigators noted (J Eur Acad Dermatol Venereol. 2016 Feb;30[2]:250-7).
Study subjects were patients with a mean age of 60 years who were treated with vemurafenib for melanoma between November 2010 and December 2014. Grade 3-4 skin toxicity that emerged within the first 4-8 weeks of treatment was significantly associated with prolonged overall survival; severe skin rashes also were associated with prolonged median overall survival. Of seven patients who switched to dabrafenib due to their skin reaction, only one experienced recurrence of the reaction.
Vemurafenib, which is approved for the treatment of unresectable stage III-IV BRAF mutant melanoma, is commonly associated with skin toxicity, with severe cases occurring only rarely; the impact of severe forms of toxicity on treatment and patient outcomes was unknown, the investigators said.
The current findings reaffirm that vemurafenib is commonly associated with skin toxicity, but rarely with severe cases. Severe skin adverse reactions permanently preclude use of vemurafenib, but dabrafenib – the only other BRAF inhibitor with market authorization for unresectable stage III-IV BRAF mutant melanoma – “seems to be beneficial in case of vemurafenib-induced skin intolerance,” they noted, adding that for other skin toxicities, including photosensitivity and cutaneous carcinoma, treatment adjustment is generally not required, and that treatment resumption at a reduced dose should be considered after skin improvement in patients with rashes.
The finding of increased survival in cases involving severe skin toxicity that emerges within 4-8 weeks of treatment initiation should be confirmed in other studies, Dr. Peuvrel and associates said.
The authors reported having no conflicts of interest.
Treatment with vemurafenib may result in severe skin toxicity, and dabrafenib appears to be useful in patients who discontinue vemurafenib due to such toxicity, according to a retrospective analysis of the cohort.
About a quarter (26%) of 131 melanoma patients treated in real life conditions with the BRAF inhibitor vemurafenib developed grade 3-4 skin toxicity, and 44% (34) of those patients permanently discontinued treatment, corresponding to 11% of the overall cohort, Dr. Lucie Peuvrel of Nantes (France) University Hospital and colleagues reported in Journal of the European Academy of Dermatology and Venereology.
Discontinuations were mainly due to rash and classic adverse skin reactions, including Stevens-Johnson syndrome, drug reaction with eosinophilia, and systemic symptoms, whereas reactions involving only photosensitivity or cutaneous carcinomas rarely resulted in treatment adjustment; 14 of the remaining 19 patients with grade 3-4 toxicity who continued treatment had no dose adjustment, and 5 had a dose reduction.
Clinical or biological abnormalities occurred in 94% of those with grade 3-4 rashes, the investigators noted (J Eur Acad Dermatol Venereol. 2016 Feb;30[2]:250-7).
Study subjects were patients with a mean age of 60 years who were treated with vemurafenib for melanoma between November 2010 and December 2014. Grade 3-4 skin toxicity that emerged within the first 4-8 weeks of treatment was significantly associated with prolonged overall survival; severe skin rashes also were associated with prolonged median overall survival. Of seven patients who switched to dabrafenib due to their skin reaction, only one experienced recurrence of the reaction.
Vemurafenib, which is approved for the treatment of unresectable stage III-IV BRAF mutant melanoma, is commonly associated with skin toxicity, with severe cases occurring only rarely; the impact of severe forms of toxicity on treatment and patient outcomes was unknown, the investigators said.
The current findings reaffirm that vemurafenib is commonly associated with skin toxicity, but rarely with severe cases. Severe skin adverse reactions permanently preclude use of vemurafenib, but dabrafenib – the only other BRAF inhibitor with market authorization for unresectable stage III-IV BRAF mutant melanoma – “seems to be beneficial in case of vemurafenib-induced skin intolerance,” they noted, adding that for other skin toxicities, including photosensitivity and cutaneous carcinoma, treatment adjustment is generally not required, and that treatment resumption at a reduced dose should be considered after skin improvement in patients with rashes.
The finding of increased survival in cases involving severe skin toxicity that emerges within 4-8 weeks of treatment initiation should be confirmed in other studies, Dr. Peuvrel and associates said.
The authors reported having no conflicts of interest.
Treatment with vemurafenib may result in severe skin toxicity, and dabrafenib appears to be useful in patients who discontinue vemurafenib due to such toxicity, according to a retrospective analysis of the cohort.
About a quarter (26%) of 131 melanoma patients treated in real life conditions with the BRAF inhibitor vemurafenib developed grade 3-4 skin toxicity, and 44% (34) of those patients permanently discontinued treatment, corresponding to 11% of the overall cohort, Dr. Lucie Peuvrel of Nantes (France) University Hospital and colleagues reported in Journal of the European Academy of Dermatology and Venereology.
Discontinuations were mainly due to rash and classic adverse skin reactions, including Stevens-Johnson syndrome, drug reaction with eosinophilia, and systemic symptoms, whereas reactions involving only photosensitivity or cutaneous carcinomas rarely resulted in treatment adjustment; 14 of the remaining 19 patients with grade 3-4 toxicity who continued treatment had no dose adjustment, and 5 had a dose reduction.
Clinical or biological abnormalities occurred in 94% of those with grade 3-4 rashes, the investigators noted (J Eur Acad Dermatol Venereol. 2016 Feb;30[2]:250-7).
Study subjects were patients with a mean age of 60 years who were treated with vemurafenib for melanoma between November 2010 and December 2014. Grade 3-4 skin toxicity that emerged within the first 4-8 weeks of treatment was significantly associated with prolonged overall survival; severe skin rashes also were associated with prolonged median overall survival. Of seven patients who switched to dabrafenib due to their skin reaction, only one experienced recurrence of the reaction.
Vemurafenib, which is approved for the treatment of unresectable stage III-IV BRAF mutant melanoma, is commonly associated with skin toxicity, with severe cases occurring only rarely; the impact of severe forms of toxicity on treatment and patient outcomes was unknown, the investigators said.
The current findings reaffirm that vemurafenib is commonly associated with skin toxicity, but rarely with severe cases. Severe skin adverse reactions permanently preclude use of vemurafenib, but dabrafenib – the only other BRAF inhibitor with market authorization for unresectable stage III-IV BRAF mutant melanoma – “seems to be beneficial in case of vemurafenib-induced skin intolerance,” they noted, adding that for other skin toxicities, including photosensitivity and cutaneous carcinoma, treatment adjustment is generally not required, and that treatment resumption at a reduced dose should be considered after skin improvement in patients with rashes.
The finding of increased survival in cases involving severe skin toxicity that emerges within 4-8 weeks of treatment initiation should be confirmed in other studies, Dr. Peuvrel and associates said.
The authors reported having no conflicts of interest.
FROM JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Treatment with vemurafenib may result in severe skin toxicity, and dabrafenib appears to be useful in patients who discontinue vemurafenib due to such toxicity.
Major finding: 26% of patients developed grade 3-4 skin toxicity, and 44% of those permanently discontinued treatment with vemurafenib.
Data source: A retrospective cohort study of 131 patients.
Disclosures: The authors reported having no conflicts of interest.
Organ transplant recipients face increased risk of BCC
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
FROM BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: The risk of a basal cell carcinoma increases after solid organ transplant.
Major finding: Transplant patients have a sixfold increased risk of BCC.
Data source: A retrospective database study of 4,000 transplant patients and almost 200,000 controls.
Disclosures: The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.