User login
Simple points about sunscreen products can address confusion over these products
GRAND CAYMAN – Are your patients noncompliant about sun protection? Do they want to use photoprotection but aren’t sure what is best for them? Perhaps they are confused by the multitude of sunscreen manufacturer’s claims or even some claims by consumer and environmental advocates.
Patients are often confused by how much sun protection factor they actually need and marketing hype often makes things worse, according to Dr. Vincent DeLeoof the department of dermatology at the University of Southern California, Los Angeles. “The use of the words ‘babies, natural, hypoallergenic, or organic’ all mean absolutely nothing when it comes to an SPF’s actual efficacy,” he said at the meeting provided by Global Academy of Medical Education.
The degree of confusion surrounding these products was illustrated in a survey of 114 patients at Chicago dermatology clinic. Of those surveyed, 80% said they purchased a sunscreen product to prevent sunburn (75%) and to prevent skin cancer (66%); having the highest SPF, a sensitive skin formulation, or being water resistant were reasons the products were chosen. But 62% could not identify information explaining its role in cancer prevention, and 77% could not identify how well the product would prevent sunburn. And nearly all participants were unsure how products prevented photoaging (JAMA Dermatol. 2015 Sep;151[9]:1028-30).
Adding to the confusion, Dr. DeLeo said, are warnings issued by some consumer advocacy groups about sunscreens that are not based on actual clinical or animal studies, but from data simulation. For example, he said there are no data to substantiate claims from such groups that oxybenzone has estrogenic effects in humans.
Consumer advocacy groups who say that sunscreens interfere with vitamin D levels may have a point, however, Dr. DeLeo noted, adding that it is time public health officials look into this issue, particularly with regards to revising recommended levels of vitamin D upward for the elderly, dark-skinned persons, and breastfed babies.
Although the Food and Drug Administration does not directly test sunscreens, it issued a 2014 guidance to manufacturers regarding which ingredients can be used and at what concentrations. Based on the FDA’s 2011 final rule on Labeling and Effectiveness Testing of OTC sunscreen products, the highest SPF currently allowed is “50+” without specifying actual numbers above 50, although Dr. DeLeo said this could be revised if industry is able to demonstrate efficacy at higher SPF values.
In an interview, however, he said that this is not likely to happen anytime soon. Citing a recent study of 40 commercially available sunscreens, including top brand names, professional skincare lines, and eponymously labeled products sold at drug store chains, more than half failed to reach the SPF level they purported to have and nine did not provide adequate broad spectrum protection.
“The takeaway is that the FDA is probably correct. Sunscreens should not be labeled higher than 50,” he said.
With the Sunscreen Innovation Act passed in 2014, the FDA is now evaluating data on new sunscreen agents that have proven effective overseas. So far, these agents have not been approved, Dr. DeLeo said in the interview.
With regards to the currently available products, “what patients really need to know is pretty simple,” Dr. DeLeo said during his presentation. An important point is that sunscreens – which work by forming a film on the stratum corneum, preventing the penetration of radiation – are effective only if used properly, he said.
A concept that is helpful for patients to understand is that while an SPF 30 product allows a person to stay in the sun twice as long as an SPF 15 product, with the same amount of protection from burning, it does not block twice as many burn rays, he noted. “When you combine them, it’s not additive, it dilutes” he said. For example, if an SPF 10 product is mixed with an SPF 20 product, the SPF is 15.
According to Dr. DeLeo, other important messages about sunscreen for patients are as follows:
• Use SPF 30 or higher.
• Apply 20-30 minutes before exposure to give the product a chance to create an effective barrier on the skin; and reapply every 2 hours or after going in the water or sweating outside.
• Use lotions or sprays, based on patient preference.
• For pregnant women or parents who don’t want to use chemical sunscreens, inorganic physical screens can be considered, although they will likely be less effective.
• Babies younger than 6 months of age can tolerate very small amounts of sunscreens.
Still, Dr. DeLeo said that the most foolproof form of photoprotection is to stay out of the sun, particularly between 10 a.m. and 4 p.m.
He disclosed being a consultant to LaRoche-Posay and Estée Lauder. Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
GRAND CAYMAN – Are your patients noncompliant about sun protection? Do they want to use photoprotection but aren’t sure what is best for them? Perhaps they are confused by the multitude of sunscreen manufacturer’s claims or even some claims by consumer and environmental advocates.
Patients are often confused by how much sun protection factor they actually need and marketing hype often makes things worse, according to Dr. Vincent DeLeoof the department of dermatology at the University of Southern California, Los Angeles. “The use of the words ‘babies, natural, hypoallergenic, or organic’ all mean absolutely nothing when it comes to an SPF’s actual efficacy,” he said at the meeting provided by Global Academy of Medical Education.
The degree of confusion surrounding these products was illustrated in a survey of 114 patients at Chicago dermatology clinic. Of those surveyed, 80% said they purchased a sunscreen product to prevent sunburn (75%) and to prevent skin cancer (66%); having the highest SPF, a sensitive skin formulation, or being water resistant were reasons the products were chosen. But 62% could not identify information explaining its role in cancer prevention, and 77% could not identify how well the product would prevent sunburn. And nearly all participants were unsure how products prevented photoaging (JAMA Dermatol. 2015 Sep;151[9]:1028-30).
Adding to the confusion, Dr. DeLeo said, are warnings issued by some consumer advocacy groups about sunscreens that are not based on actual clinical or animal studies, but from data simulation. For example, he said there are no data to substantiate claims from such groups that oxybenzone has estrogenic effects in humans.
Consumer advocacy groups who say that sunscreens interfere with vitamin D levels may have a point, however, Dr. DeLeo noted, adding that it is time public health officials look into this issue, particularly with regards to revising recommended levels of vitamin D upward for the elderly, dark-skinned persons, and breastfed babies.
Although the Food and Drug Administration does not directly test sunscreens, it issued a 2014 guidance to manufacturers regarding which ingredients can be used and at what concentrations. Based on the FDA’s 2011 final rule on Labeling and Effectiveness Testing of OTC sunscreen products, the highest SPF currently allowed is “50+” without specifying actual numbers above 50, although Dr. DeLeo said this could be revised if industry is able to demonstrate efficacy at higher SPF values.
In an interview, however, he said that this is not likely to happen anytime soon. Citing a recent study of 40 commercially available sunscreens, including top brand names, professional skincare lines, and eponymously labeled products sold at drug store chains, more than half failed to reach the SPF level they purported to have and nine did not provide adequate broad spectrum protection.
“The takeaway is that the FDA is probably correct. Sunscreens should not be labeled higher than 50,” he said.
With the Sunscreen Innovation Act passed in 2014, the FDA is now evaluating data on new sunscreen agents that have proven effective overseas. So far, these agents have not been approved, Dr. DeLeo said in the interview.
With regards to the currently available products, “what patients really need to know is pretty simple,” Dr. DeLeo said during his presentation. An important point is that sunscreens – which work by forming a film on the stratum corneum, preventing the penetration of radiation – are effective only if used properly, he said.
A concept that is helpful for patients to understand is that while an SPF 30 product allows a person to stay in the sun twice as long as an SPF 15 product, with the same amount of protection from burning, it does not block twice as many burn rays, he noted. “When you combine them, it’s not additive, it dilutes” he said. For example, if an SPF 10 product is mixed with an SPF 20 product, the SPF is 15.
According to Dr. DeLeo, other important messages about sunscreen for patients are as follows:
• Use SPF 30 or higher.
• Apply 20-30 minutes before exposure to give the product a chance to create an effective barrier on the skin; and reapply every 2 hours or after going in the water or sweating outside.
• Use lotions or sprays, based on patient preference.
• For pregnant women or parents who don’t want to use chemical sunscreens, inorganic physical screens can be considered, although they will likely be less effective.
• Babies younger than 6 months of age can tolerate very small amounts of sunscreens.
Still, Dr. DeLeo said that the most foolproof form of photoprotection is to stay out of the sun, particularly between 10 a.m. and 4 p.m.
He disclosed being a consultant to LaRoche-Posay and Estée Lauder. Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
GRAND CAYMAN – Are your patients noncompliant about sun protection? Do they want to use photoprotection but aren’t sure what is best for them? Perhaps they are confused by the multitude of sunscreen manufacturer’s claims or even some claims by consumer and environmental advocates.
Patients are often confused by how much sun protection factor they actually need and marketing hype often makes things worse, according to Dr. Vincent DeLeoof the department of dermatology at the University of Southern California, Los Angeles. “The use of the words ‘babies, natural, hypoallergenic, or organic’ all mean absolutely nothing when it comes to an SPF’s actual efficacy,” he said at the meeting provided by Global Academy of Medical Education.
The degree of confusion surrounding these products was illustrated in a survey of 114 patients at Chicago dermatology clinic. Of those surveyed, 80% said they purchased a sunscreen product to prevent sunburn (75%) and to prevent skin cancer (66%); having the highest SPF, a sensitive skin formulation, or being water resistant were reasons the products were chosen. But 62% could not identify information explaining its role in cancer prevention, and 77% could not identify how well the product would prevent sunburn. And nearly all participants were unsure how products prevented photoaging (JAMA Dermatol. 2015 Sep;151[9]:1028-30).
Adding to the confusion, Dr. DeLeo said, are warnings issued by some consumer advocacy groups about sunscreens that are not based on actual clinical or animal studies, but from data simulation. For example, he said there are no data to substantiate claims from such groups that oxybenzone has estrogenic effects in humans.
Consumer advocacy groups who say that sunscreens interfere with vitamin D levels may have a point, however, Dr. DeLeo noted, adding that it is time public health officials look into this issue, particularly with regards to revising recommended levels of vitamin D upward for the elderly, dark-skinned persons, and breastfed babies.
Although the Food and Drug Administration does not directly test sunscreens, it issued a 2014 guidance to manufacturers regarding which ingredients can be used and at what concentrations. Based on the FDA’s 2011 final rule on Labeling and Effectiveness Testing of OTC sunscreen products, the highest SPF currently allowed is “50+” without specifying actual numbers above 50, although Dr. DeLeo said this could be revised if industry is able to demonstrate efficacy at higher SPF values.
In an interview, however, he said that this is not likely to happen anytime soon. Citing a recent study of 40 commercially available sunscreens, including top brand names, professional skincare lines, and eponymously labeled products sold at drug store chains, more than half failed to reach the SPF level they purported to have and nine did not provide adequate broad spectrum protection.
“The takeaway is that the FDA is probably correct. Sunscreens should not be labeled higher than 50,” he said.
With the Sunscreen Innovation Act passed in 2014, the FDA is now evaluating data on new sunscreen agents that have proven effective overseas. So far, these agents have not been approved, Dr. DeLeo said in the interview.
With regards to the currently available products, “what patients really need to know is pretty simple,” Dr. DeLeo said during his presentation. An important point is that sunscreens – which work by forming a film on the stratum corneum, preventing the penetration of radiation – are effective only if used properly, he said.
A concept that is helpful for patients to understand is that while an SPF 30 product allows a person to stay in the sun twice as long as an SPF 15 product, with the same amount of protection from burning, it does not block twice as many burn rays, he noted. “When you combine them, it’s not additive, it dilutes” he said. For example, if an SPF 10 product is mixed with an SPF 20 product, the SPF is 15.
According to Dr. DeLeo, other important messages about sunscreen for patients are as follows:
• Use SPF 30 or higher.
• Apply 20-30 minutes before exposure to give the product a chance to create an effective barrier on the skin; and reapply every 2 hours or after going in the water or sweating outside.
• Use lotions or sprays, based on patient preference.
• For pregnant women or parents who don’t want to use chemical sunscreens, inorganic physical screens can be considered, although they will likely be less effective.
• Babies younger than 6 months of age can tolerate very small amounts of sunscreens.
Still, Dr. DeLeo said that the most foolproof form of photoprotection is to stay out of the sun, particularly between 10 a.m. and 4 p.m.
He disclosed being a consultant to LaRoche-Posay and Estée Lauder. Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
AT THE CARIBBEAN DERMATOLOGY SYMPOSIUM
Time will tell if medical apps help or compete with dermatologists
GRAND CAYMAN ISLAND – Smartphones and other digital technologies could provide clinicians – and patients – the tools to greatly improve melanoma and other dermatology outcomes, but how might they change actual practice?
“Ten years from now, what is going to distinguish the app world from the dermatology community?” That was the question Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, posed at the meeting, sponsored by Global Academy for Medical Education.
Many clinicians already use dermatoscopes, which Dr. Halpern said “significantly improves diagnostic accuracy for melanoma by making available to us visual cues that would not be available through routine naked-eye examination.” However, he said, a rapid digital evolution in dermoscopy is already underway, allowing clinicians to use mobile devices that store images and perform short term monitoring of any areas of concern.
“For suspicious lesions, rather than do a biopsy, one of the options is that you can take a dermoscopic image of it, then do so again in another couple of months. If it hasn’t changed, then the data are very convincing that you’re dealing with something other than melanoma,” Dr. Halpern said.
What gives him pause is that there are an increasing number of consumer-based smartphone medical products, including more than 250 for dermatology alone.
“For about 100 dollars or less, patients can purchase their own dermatoscope and attach it to their smartphone” and take images of their own lesions, Dr. Halpern said.
As a result, he believes that the field has reached the “tipping point” for successful teledermatology, where companies can accept and interpret these data.
Because of the exponential growth of mobile medical applications, the Food and Drug Administration issued guidance for their use in 2015, he noted.
Although he said that the FDA is overwhelmed by the task – and that the medical app marketplace is akin to the Wild West – medical apps are here to stay and will continue to develop quickly.
“For one thing, big players are now entering the field,” he said, alluding to a recent Apple initiativeto collect data uploaded by consumers to the Cloud, which can then be parsed by medical researchers. In addition to melanoma, Apple is currently focusing on autism and epilepsy.
“Apple is trying to make the iPhone a conduit for medical research,” Dr. Halpern said.
Over the next 5-10 years, there will be a race to see who can perfect this kind of data analysis. There is even an ongoing competition that pits melanoma researchers around the world against one another to see who can most accurately interpret images uploaded to the archives. The hope, he said, is that it will help dermatologists to develop and agree upon international standards for melanoma diagnosis.
With this kind of standardization, medical apps can help both consumers and practitioners triage care.
“I don’t know when it will happen, but there is no doubt we’re going to reach a point at which these apps are going to be as good as the average dermatologist is today when it comes to looking at a single spot,” Dr. Halpern said.
Aside from some cancer centers’ ability to create three-dimensional images of lesions, another distinguishing technology for dermatologists could be reflectance confocal microscopy.
“As dermatologists, we are trained well enough in pathology that we can use this technology, we will be able to do even better than with dermoscopy alone,” he said. This kind of technology captures images at the cellular level similar to histological examinations, but without the actual biopsy.
In an interview, Dr. Halpern said that while dermatologists have nothing to fear from apps in daily practice, there are some issues to be aware of. “The first is the one we’re most concerned about – automated diagnosis, before we know for certain that the apps work.”
Another issue is that these apps raise awareness among consumers about their individual risk for melanoma, which will “invariably help drive more people in the door,” but there may not be enough clinicians to manage these patients.
The group Dr. Halpern expects will be the most frequent users of personal dermatology technologies initially are “ultra–tech savvy young people motivated to avoid melanoma,” although he expects that over time, this could change as technologies become easier to use.
He disclosed financial relationships with Quintiles, Canfield Scientific, DermTech, SciBase, and Caliber ID.
Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
GRAND CAYMAN ISLAND – Smartphones and other digital technologies could provide clinicians – and patients – the tools to greatly improve melanoma and other dermatology outcomes, but how might they change actual practice?
“Ten years from now, what is going to distinguish the app world from the dermatology community?” That was the question Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, posed at the meeting, sponsored by Global Academy for Medical Education.
Many clinicians already use dermatoscopes, which Dr. Halpern said “significantly improves diagnostic accuracy for melanoma by making available to us visual cues that would not be available through routine naked-eye examination.” However, he said, a rapid digital evolution in dermoscopy is already underway, allowing clinicians to use mobile devices that store images and perform short term monitoring of any areas of concern.
“For suspicious lesions, rather than do a biopsy, one of the options is that you can take a dermoscopic image of it, then do so again in another couple of months. If it hasn’t changed, then the data are very convincing that you’re dealing with something other than melanoma,” Dr. Halpern said.
What gives him pause is that there are an increasing number of consumer-based smartphone medical products, including more than 250 for dermatology alone.
“For about 100 dollars or less, patients can purchase their own dermatoscope and attach it to their smartphone” and take images of their own lesions, Dr. Halpern said.
As a result, he believes that the field has reached the “tipping point” for successful teledermatology, where companies can accept and interpret these data.
Because of the exponential growth of mobile medical applications, the Food and Drug Administration issued guidance for their use in 2015, he noted.
Although he said that the FDA is overwhelmed by the task – and that the medical app marketplace is akin to the Wild West – medical apps are here to stay and will continue to develop quickly.
“For one thing, big players are now entering the field,” he said, alluding to a recent Apple initiativeto collect data uploaded by consumers to the Cloud, which can then be parsed by medical researchers. In addition to melanoma, Apple is currently focusing on autism and epilepsy.
“Apple is trying to make the iPhone a conduit for medical research,” Dr. Halpern said.
Over the next 5-10 years, there will be a race to see who can perfect this kind of data analysis. There is even an ongoing competition that pits melanoma researchers around the world against one another to see who can most accurately interpret images uploaded to the archives. The hope, he said, is that it will help dermatologists to develop and agree upon international standards for melanoma diagnosis.
With this kind of standardization, medical apps can help both consumers and practitioners triage care.
“I don’t know when it will happen, but there is no doubt we’re going to reach a point at which these apps are going to be as good as the average dermatologist is today when it comes to looking at a single spot,” Dr. Halpern said.
Aside from some cancer centers’ ability to create three-dimensional images of lesions, another distinguishing technology for dermatologists could be reflectance confocal microscopy.
“As dermatologists, we are trained well enough in pathology that we can use this technology, we will be able to do even better than with dermoscopy alone,” he said. This kind of technology captures images at the cellular level similar to histological examinations, but without the actual biopsy.
In an interview, Dr. Halpern said that while dermatologists have nothing to fear from apps in daily practice, there are some issues to be aware of. “The first is the one we’re most concerned about – automated diagnosis, before we know for certain that the apps work.”
Another issue is that these apps raise awareness among consumers about their individual risk for melanoma, which will “invariably help drive more people in the door,” but there may not be enough clinicians to manage these patients.
The group Dr. Halpern expects will be the most frequent users of personal dermatology technologies initially are “ultra–tech savvy young people motivated to avoid melanoma,” although he expects that over time, this could change as technologies become easier to use.
He disclosed financial relationships with Quintiles, Canfield Scientific, DermTech, SciBase, and Caliber ID.
Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
GRAND CAYMAN ISLAND – Smartphones and other digital technologies could provide clinicians – and patients – the tools to greatly improve melanoma and other dermatology outcomes, but how might they change actual practice?
“Ten years from now, what is going to distinguish the app world from the dermatology community?” That was the question Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, posed at the meeting, sponsored by Global Academy for Medical Education.
Many clinicians already use dermatoscopes, which Dr. Halpern said “significantly improves diagnostic accuracy for melanoma by making available to us visual cues that would not be available through routine naked-eye examination.” However, he said, a rapid digital evolution in dermoscopy is already underway, allowing clinicians to use mobile devices that store images and perform short term monitoring of any areas of concern.
“For suspicious lesions, rather than do a biopsy, one of the options is that you can take a dermoscopic image of it, then do so again in another couple of months. If it hasn’t changed, then the data are very convincing that you’re dealing with something other than melanoma,” Dr. Halpern said.
What gives him pause is that there are an increasing number of consumer-based smartphone medical products, including more than 250 for dermatology alone.
“For about 100 dollars or less, patients can purchase their own dermatoscope and attach it to their smartphone” and take images of their own lesions, Dr. Halpern said.
As a result, he believes that the field has reached the “tipping point” for successful teledermatology, where companies can accept and interpret these data.
Because of the exponential growth of mobile medical applications, the Food and Drug Administration issued guidance for their use in 2015, he noted.
Although he said that the FDA is overwhelmed by the task – and that the medical app marketplace is akin to the Wild West – medical apps are here to stay and will continue to develop quickly.
“For one thing, big players are now entering the field,” he said, alluding to a recent Apple initiativeto collect data uploaded by consumers to the Cloud, which can then be parsed by medical researchers. In addition to melanoma, Apple is currently focusing on autism and epilepsy.
“Apple is trying to make the iPhone a conduit for medical research,” Dr. Halpern said.
Over the next 5-10 years, there will be a race to see who can perfect this kind of data analysis. There is even an ongoing competition that pits melanoma researchers around the world against one another to see who can most accurately interpret images uploaded to the archives. The hope, he said, is that it will help dermatologists to develop and agree upon international standards for melanoma diagnosis.
With this kind of standardization, medical apps can help both consumers and practitioners triage care.
“I don’t know when it will happen, but there is no doubt we’re going to reach a point at which these apps are going to be as good as the average dermatologist is today when it comes to looking at a single spot,” Dr. Halpern said.
Aside from some cancer centers’ ability to create three-dimensional images of lesions, another distinguishing technology for dermatologists could be reflectance confocal microscopy.
“As dermatologists, we are trained well enough in pathology that we can use this technology, we will be able to do even better than with dermoscopy alone,” he said. This kind of technology captures images at the cellular level similar to histological examinations, but without the actual biopsy.
In an interview, Dr. Halpern said that while dermatologists have nothing to fear from apps in daily practice, there are some issues to be aware of. “The first is the one we’re most concerned about – automated diagnosis, before we know for certain that the apps work.”
Another issue is that these apps raise awareness among consumers about their individual risk for melanoma, which will “invariably help drive more people in the door,” but there may not be enough clinicians to manage these patients.
The group Dr. Halpern expects will be the most frequent users of personal dermatology technologies initially are “ultra–tech savvy young people motivated to avoid melanoma,” although he expects that over time, this could change as technologies become easier to use.
He disclosed financial relationships with Quintiles, Canfield Scientific, DermTech, SciBase, and Caliber ID.
Global Academy and this news organization are owned by the same parent company.
On Twitter @whitneymcknight
AT THE CARIBBEAN DERMATOLOGY SYMPOSIUM
MAPK inhibitor combo offers prolonged survival from BRAF+ advanced melanoma
Combination therapy to inhibit the MAP kinase pathway is associated with a median overall survival of more than 2 years for patients with metastatic melanoma positive for the BRAF V600 mutation who have not previously received a BRAF inhibitor.
An analysis of data from a phase I and II trial of the BRAF inhibitor dabrafenib (Tafinlar), 150 mg twice daily, and the MEK inhibitor trametinib (Mekinist), 2 mg once daily, in 78 BRAF inhibitor–naïve patients with metastatic melanoma showed that in a non-randomized cohort (part B) the median overall survival (OS) was 27.4 months. In a cohort randomized to the same doses (part C), the median OS was 25 months, reported Dr. Georgina V. Long and her colleagues from the Melanoma Institute Australia and the University of Sydney in the Journal of Clinical Oncology.
Among 24 patients treated in part B, progression-free survival (PFS) was 18% at 3 years.
“The combination has an acceptable long-term safety profile and is a standard of care for patients with BRAF mutation–positive metastatic melanoma, particularly given the recent publications demonstrating a significant improvement in the PFS and OS in phase III trials of combination versus single-agent BRAF inhibitors,” the investigators wrote (J Clin Oncol. 2016 Jan 25. doi: 10.1200/JCO.2015.62.9345).
The investigators conducted the analysis to determine which patients were the most likely to benefit from combination therapy of the MAP (mitogen-active protein) kinase pathway.
They observed that in the part B, non-randomized cohort, OS was 72% at 1 year, 60% at 2 tears, and 47% at 3 years. In the part C, randomized cohort, OS was 80%, 51%, and 38%, respectively.
Factors associated with better OS in Cox proportional hazards regression models included metastases to fewer than three organs sites, and lower levels of lactate dehydrogenase (LDH) at baseline. Not surprisingly, a complete response to therapy was also associated with better survival. Three-year OS rates were 62% for patients with normal baseline LDH levels, and 63% for patients who had a complete response.
Combination therapy to inhibit the MAP kinase pathway is associated with a median overall survival of more than 2 years for patients with metastatic melanoma positive for the BRAF V600 mutation who have not previously received a BRAF inhibitor.
An analysis of data from a phase I and II trial of the BRAF inhibitor dabrafenib (Tafinlar), 150 mg twice daily, and the MEK inhibitor trametinib (Mekinist), 2 mg once daily, in 78 BRAF inhibitor–naïve patients with metastatic melanoma showed that in a non-randomized cohort (part B) the median overall survival (OS) was 27.4 months. In a cohort randomized to the same doses (part C), the median OS was 25 months, reported Dr. Georgina V. Long and her colleagues from the Melanoma Institute Australia and the University of Sydney in the Journal of Clinical Oncology.
Among 24 patients treated in part B, progression-free survival (PFS) was 18% at 3 years.
“The combination has an acceptable long-term safety profile and is a standard of care for patients with BRAF mutation–positive metastatic melanoma, particularly given the recent publications demonstrating a significant improvement in the PFS and OS in phase III trials of combination versus single-agent BRAF inhibitors,” the investigators wrote (J Clin Oncol. 2016 Jan 25. doi: 10.1200/JCO.2015.62.9345).
The investigators conducted the analysis to determine which patients were the most likely to benefit from combination therapy of the MAP (mitogen-active protein) kinase pathway.
They observed that in the part B, non-randomized cohort, OS was 72% at 1 year, 60% at 2 tears, and 47% at 3 years. In the part C, randomized cohort, OS was 80%, 51%, and 38%, respectively.
Factors associated with better OS in Cox proportional hazards regression models included metastases to fewer than three organs sites, and lower levels of lactate dehydrogenase (LDH) at baseline. Not surprisingly, a complete response to therapy was also associated with better survival. Three-year OS rates were 62% for patients with normal baseline LDH levels, and 63% for patients who had a complete response.
Combination therapy to inhibit the MAP kinase pathway is associated with a median overall survival of more than 2 years for patients with metastatic melanoma positive for the BRAF V600 mutation who have not previously received a BRAF inhibitor.
An analysis of data from a phase I and II trial of the BRAF inhibitor dabrafenib (Tafinlar), 150 mg twice daily, and the MEK inhibitor trametinib (Mekinist), 2 mg once daily, in 78 BRAF inhibitor–naïve patients with metastatic melanoma showed that in a non-randomized cohort (part B) the median overall survival (OS) was 27.4 months. In a cohort randomized to the same doses (part C), the median OS was 25 months, reported Dr. Georgina V. Long and her colleagues from the Melanoma Institute Australia and the University of Sydney in the Journal of Clinical Oncology.
Among 24 patients treated in part B, progression-free survival (PFS) was 18% at 3 years.
“The combination has an acceptable long-term safety profile and is a standard of care for patients with BRAF mutation–positive metastatic melanoma, particularly given the recent publications demonstrating a significant improvement in the PFS and OS in phase III trials of combination versus single-agent BRAF inhibitors,” the investigators wrote (J Clin Oncol. 2016 Jan 25. doi: 10.1200/JCO.2015.62.9345).
The investigators conducted the analysis to determine which patients were the most likely to benefit from combination therapy of the MAP (mitogen-active protein) kinase pathway.
They observed that in the part B, non-randomized cohort, OS was 72% at 1 year, 60% at 2 tears, and 47% at 3 years. In the part C, randomized cohort, OS was 80%, 51%, and 38%, respectively.
Factors associated with better OS in Cox proportional hazards regression models included metastases to fewer than three organs sites, and lower levels of lactate dehydrogenase (LDH) at baseline. Not surprisingly, a complete response to therapy was also associated with better survival. Three-year OS rates were 62% for patients with normal baseline LDH levels, and 63% for patients who had a complete response.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Combination MAP kinase pathway inhibition was associated with prolonged survival of BRAF mutation-positive metastatic melanoma.
Major finding: Median overall survival was 27.4 months in a non-randomized cohort, and 25 months in a randomized cohort.
Data source: Analysis of a subset of patients in a phase I and II trial of dabrafenib and trametinib.
Disclosures: The study was supported by GlaxoSmithKline. Dr. Long and many coauthors disclosed consulting and/or research funding from the company.
Anorectal melanoma rates rose significantly in 20-year analysis
Rates of anorectal melanoma rose substantially in the United States from 1992 to 2011, according to a study published in Dermatologic Surgery.
The increases affected men and women, said Dr. Adrienne Callahan of the department of dermatology, University Hospitals Case Medical Center, Cleveland, and her associates. Anorectal carcinoma was significantly more common among Hispanic whites than non-Hispanic whites (P = .02), “suggesting that this population may be targeted for screening interventions,” they added. (Dermatol Surg. 2016;42[1]:94-9). The highest rates were in Hispanic white elderly women.
Anorectal melanoma accounts for 1.3% of all melanomas and 16.5% of mucosal melanomas, and is most common among older women. The prognosis is often poor because the cancer tends to be asymptomatic until its late stages. To study the epidemiology of anorectal melanoma, the researchers analyzed data from the Surveillance, Epidemiology, and End Results 13 (SEER 13) Registries Database for 1992 through 2011.
The SEER database listed 260 cases for the study period. Most involved the rectum, 58% affected adults aged 65 years and older, and almost two-thirds occurred in women – a finding that dovetails with other studies, the researchers said. Notably, the estimated annual change in age-standardized incidence rates increased in both men (5.08%) and women (3.02%), which were statistically significant increases (P less than .05 for both trends).
Anorectal melanoma rates were significantly higher among Hispanic whites, compared with non-Hispanic whites. “Other studies have indicated that Hispanics are underscreened for skin cancer compared to other ethnic groups despite the increasing incidence of melanoma in this population,” said the researchers. “Although this may suggest a role for improved screening among those with Hispanic ethnicity, additional studies must be done to corroborate these results and further elucidate the association of Hispanic ethnicity with anorectal melanoma.”
As far as they know, this is the first study that has analyzed anorectal melanoma incidence in Hispanic whites and non-Hispanic whites, they added.
Ethnicity did not affect survival, which was generally poor. Anorectal melanoma is very rare, so the number of cases was relatively small and rates might have been unstable, the investigators noted. They added that it was not known whether the increases they found were related to improved detection.
The study was funded by the Char and Chuck Fowler Family Foundation, the Dermatology Foundation, and the National Cancer Institute. The researchers had no disclosures.
Rates of anorectal melanoma rose substantially in the United States from 1992 to 2011, according to a study published in Dermatologic Surgery.
The increases affected men and women, said Dr. Adrienne Callahan of the department of dermatology, University Hospitals Case Medical Center, Cleveland, and her associates. Anorectal carcinoma was significantly more common among Hispanic whites than non-Hispanic whites (P = .02), “suggesting that this population may be targeted for screening interventions,” they added. (Dermatol Surg. 2016;42[1]:94-9). The highest rates were in Hispanic white elderly women.
Anorectal melanoma accounts for 1.3% of all melanomas and 16.5% of mucosal melanomas, and is most common among older women. The prognosis is often poor because the cancer tends to be asymptomatic until its late stages. To study the epidemiology of anorectal melanoma, the researchers analyzed data from the Surveillance, Epidemiology, and End Results 13 (SEER 13) Registries Database for 1992 through 2011.
The SEER database listed 260 cases for the study period. Most involved the rectum, 58% affected adults aged 65 years and older, and almost two-thirds occurred in women – a finding that dovetails with other studies, the researchers said. Notably, the estimated annual change in age-standardized incidence rates increased in both men (5.08%) and women (3.02%), which were statistically significant increases (P less than .05 for both trends).
Anorectal melanoma rates were significantly higher among Hispanic whites, compared with non-Hispanic whites. “Other studies have indicated that Hispanics are underscreened for skin cancer compared to other ethnic groups despite the increasing incidence of melanoma in this population,” said the researchers. “Although this may suggest a role for improved screening among those with Hispanic ethnicity, additional studies must be done to corroborate these results and further elucidate the association of Hispanic ethnicity with anorectal melanoma.”
As far as they know, this is the first study that has analyzed anorectal melanoma incidence in Hispanic whites and non-Hispanic whites, they added.
Ethnicity did not affect survival, which was generally poor. Anorectal melanoma is very rare, so the number of cases was relatively small and rates might have been unstable, the investigators noted. They added that it was not known whether the increases they found were related to improved detection.
The study was funded by the Char and Chuck Fowler Family Foundation, the Dermatology Foundation, and the National Cancer Institute. The researchers had no disclosures.
Rates of anorectal melanoma rose substantially in the United States from 1992 to 2011, according to a study published in Dermatologic Surgery.
The increases affected men and women, said Dr. Adrienne Callahan of the department of dermatology, University Hospitals Case Medical Center, Cleveland, and her associates. Anorectal carcinoma was significantly more common among Hispanic whites than non-Hispanic whites (P = .02), “suggesting that this population may be targeted for screening interventions,” they added. (Dermatol Surg. 2016;42[1]:94-9). The highest rates were in Hispanic white elderly women.
Anorectal melanoma accounts for 1.3% of all melanomas and 16.5% of mucosal melanomas, and is most common among older women. The prognosis is often poor because the cancer tends to be asymptomatic until its late stages. To study the epidemiology of anorectal melanoma, the researchers analyzed data from the Surveillance, Epidemiology, and End Results 13 (SEER 13) Registries Database for 1992 through 2011.
The SEER database listed 260 cases for the study period. Most involved the rectum, 58% affected adults aged 65 years and older, and almost two-thirds occurred in women – a finding that dovetails with other studies, the researchers said. Notably, the estimated annual change in age-standardized incidence rates increased in both men (5.08%) and women (3.02%), which were statistically significant increases (P less than .05 for both trends).
Anorectal melanoma rates were significantly higher among Hispanic whites, compared with non-Hispanic whites. “Other studies have indicated that Hispanics are underscreened for skin cancer compared to other ethnic groups despite the increasing incidence of melanoma in this population,” said the researchers. “Although this may suggest a role for improved screening among those with Hispanic ethnicity, additional studies must be done to corroborate these results and further elucidate the association of Hispanic ethnicity with anorectal melanoma.”
As far as they know, this is the first study that has analyzed anorectal melanoma incidence in Hispanic whites and non-Hispanic whites, they added.
Ethnicity did not affect survival, which was generally poor. Anorectal melanoma is very rare, so the number of cases was relatively small and rates might have been unstable, the investigators noted. They added that it was not known whether the increases they found were related to improved detection.
The study was funded by the Char and Chuck Fowler Family Foundation, the Dermatology Foundation, and the National Cancer Institute. The researchers had no disclosures.
FROM DERMATOLOGIC SURGERY
Key clinical point: Rates of anorectal melanoma rose substantially in the United States over 2 decades.
Major finding: The age-adjusted incidence rate for anorectal melanoma increased annually by an estimated 5.1% for men and 3.0% for women, over a recent 20-year period.
Data source: The study analyzed data from the Surveillance, Epidemiology, and End Results (SEER) 13 Registries Database between 1992 and 2011.
Disclosures: The study was funded by the Char and Chuck Fowler Family Foundation, the Dermatology Foundation, and the National Cancer Institute. The researchers had no disclosures.
Sirolimus reduced posttransplant skin cancer risk
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
FROM JAMA DERMATOLOGY
Key clinical point: Sirolimus protects organ-transplant recipients against skin cancer.
Major finding: The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8% in the untreated group.
Data source: A retrospective cohort study of 329 organ-transplant recipients who had already developed one cancer likely related to their immunosuppressive therapy.
Disclosures: This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
Encouraging results for MMS in digital melanoma cases
Mohs micrographic surgery (MMS) to remove digital melanomas of less than 2 mm Breslow thickness spared patients from amputation without undermining survival, according to a single-center retrospective study published in Dermatologic Surgery.
The overall recurrence rate was 8%, and there were no recurrences with use of the MART-1 (melanoma antigen recognized by T-cells 1 staining) immunostain, said Dr. Vitaly Terushkin of the Zitelli and Brodland Skin Cancer Center in Pittsburgh and his associates. “The use of MART-1 has revolutionized the value of MMS,” the researchers said, while cautioning that the sample size was too small to draw reliable conclusions about thicker tumors.
Digital melanoma historically eluded diagnosis until it was advanced and was treated with amputation. Amputation and wide local excision remain common, and particularly erode function and balance when the tumor involves the thumb or big toe, the researchers noted. In 35 years at their center, 62 patients underwent MMS for digital melanoma, two-thirds of which involved the nail unit. About 92% of tumors were primary, 8% were recurrent, 53% involved the fingers, and 47% involved the toes. About half of patients were women, and the average age at treatment was about 63 years (Dermatol Surg. 2016;42:83-93).
Nearly 97% of patients avoided amputation over an average of 6.5 years of follow-up, the investigators reported. Five-year and 10-year local recurrence-free survival rates for the primary melanomas were about 92% and 83%, respectively. Of the five local recurrences, three were salvaged with additional MMS and two were treated with an amputation; all five recurrences involved the nail unit. Failing to remove the entire nail unit apparatus probably led to false-negative MMS layers and subsequent recurrence in at least two cases, they pointed out.
This is only the fifth published study to describe the treatment of digital melanoma with MMS, according to Dr. Terushkin and his associates. Although the study lacked a comparison group, it suggested that MART-1 improved outcomes and that most recurrences could be retreated with MMS without amputation or negative effects on survival. These findings will hopefully help inform future melanoma guidelines, said the researchers.
The investigators reported no funding sources and had no disclosures.
Mohs micrographic surgery (MMS) to remove digital melanomas of less than 2 mm Breslow thickness spared patients from amputation without undermining survival, according to a single-center retrospective study published in Dermatologic Surgery.
The overall recurrence rate was 8%, and there were no recurrences with use of the MART-1 (melanoma antigen recognized by T-cells 1 staining) immunostain, said Dr. Vitaly Terushkin of the Zitelli and Brodland Skin Cancer Center in Pittsburgh and his associates. “The use of MART-1 has revolutionized the value of MMS,” the researchers said, while cautioning that the sample size was too small to draw reliable conclusions about thicker tumors.
Digital melanoma historically eluded diagnosis until it was advanced and was treated with amputation. Amputation and wide local excision remain common, and particularly erode function and balance when the tumor involves the thumb or big toe, the researchers noted. In 35 years at their center, 62 patients underwent MMS for digital melanoma, two-thirds of which involved the nail unit. About 92% of tumors were primary, 8% were recurrent, 53% involved the fingers, and 47% involved the toes. About half of patients were women, and the average age at treatment was about 63 years (Dermatol Surg. 2016;42:83-93).
Nearly 97% of patients avoided amputation over an average of 6.5 years of follow-up, the investigators reported. Five-year and 10-year local recurrence-free survival rates for the primary melanomas were about 92% and 83%, respectively. Of the five local recurrences, three were salvaged with additional MMS and two were treated with an amputation; all five recurrences involved the nail unit. Failing to remove the entire nail unit apparatus probably led to false-negative MMS layers and subsequent recurrence in at least two cases, they pointed out.
This is only the fifth published study to describe the treatment of digital melanoma with MMS, according to Dr. Terushkin and his associates. Although the study lacked a comparison group, it suggested that MART-1 improved outcomes and that most recurrences could be retreated with MMS without amputation or negative effects on survival. These findings will hopefully help inform future melanoma guidelines, said the researchers.
The investigators reported no funding sources and had no disclosures.
Mohs micrographic surgery (MMS) to remove digital melanomas of less than 2 mm Breslow thickness spared patients from amputation without undermining survival, according to a single-center retrospective study published in Dermatologic Surgery.
The overall recurrence rate was 8%, and there were no recurrences with use of the MART-1 (melanoma antigen recognized by T-cells 1 staining) immunostain, said Dr. Vitaly Terushkin of the Zitelli and Brodland Skin Cancer Center in Pittsburgh and his associates. “The use of MART-1 has revolutionized the value of MMS,” the researchers said, while cautioning that the sample size was too small to draw reliable conclusions about thicker tumors.
Digital melanoma historically eluded diagnosis until it was advanced and was treated with amputation. Amputation and wide local excision remain common, and particularly erode function and balance when the tumor involves the thumb or big toe, the researchers noted. In 35 years at their center, 62 patients underwent MMS for digital melanoma, two-thirds of which involved the nail unit. About 92% of tumors were primary, 8% were recurrent, 53% involved the fingers, and 47% involved the toes. About half of patients were women, and the average age at treatment was about 63 years (Dermatol Surg. 2016;42:83-93).
Nearly 97% of patients avoided amputation over an average of 6.5 years of follow-up, the investigators reported. Five-year and 10-year local recurrence-free survival rates for the primary melanomas were about 92% and 83%, respectively. Of the five local recurrences, three were salvaged with additional MMS and two were treated with an amputation; all five recurrences involved the nail unit. Failing to remove the entire nail unit apparatus probably led to false-negative MMS layers and subsequent recurrence in at least two cases, they pointed out.
This is only the fifth published study to describe the treatment of digital melanoma with MMS, according to Dr. Terushkin and his associates. Although the study lacked a comparison group, it suggested that MART-1 improved outcomes and that most recurrences could be retreated with MMS without amputation or negative effects on survival. These findings will hopefully help inform future melanoma guidelines, said the researchers.
The investigators reported no funding sources and had no disclosures.
FROM DERMATOLOGIC SURGERY
EADV: Vismodegib treatment breaks don’t hurt efficacy
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
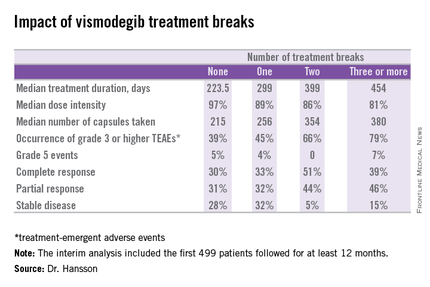
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

AT THE EADV CONGRESS
Key clinical point: Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t compromise efficacy.
Major finding: The complete response rate to vismodegib in patients with advanced BCC was intriguingly higher in those with more treatment breaks due to adverse events.
Data source: A prespecified interim analysis of the first 499 patients with advanced BCC enrolled in STEVIE, a large ongoing phase II, long-term, open-label international safety study of vismodegib.
Disclosures: The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. The presenter reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
Cancer prevention field riding high into the new year
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
FROM CANCER PREVENTION RESEARCH
T-VEC: Advancing the Fight Against Melanoma

Following a phase III, open-label trial conducted by Andtbacka et al (J Clin Oncol. 2015;33:2780-2788), the US Food and Drug Administration recently approved the first oncolytic immunotherapy talimogene laherparepvec (T-VEC) for the treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with advanced melanoma (stage IIIB/C–stage IV) following initial surgery.
A group of 436 patients with injectable melanomas (melanomas that are accessible via a percutaneous injection) that were not surgically resectable were randomly assigned (2:1) to treatment with intralesional T-VEC or subcutaneous granulocyte macrophage colony-stimulating factor (GM-CSF). The primary endpoint of the study was durable response rate (DRR), defined as objective response lasting continuously for 6 months or longer. Secondary endpoints included overall survival (OS) and overall response rate.
Talimogene laherparepvec was shown to extend DRRs compared to GM-CSF. Durable response rates were significantly higher with T-VEC (16.3%; 95% confidence interval [CI], 12.1%–20.5%) versus GM-CSF (2.1%; 95% CI, 0%–4.5%)(odds ratio, 8.9; P<.001).
In the OS analysis, a 4.4-month extension with T-VEC was observed; however, this was not deemed to be statistically significant (P=.051). The median OS was 23.3 months (95% CI, 19.5–29.6 months) with T-VEC and 18.9 months (95% CI, 16.0–23.7 months) with GM-CSF (hazard ratio, 0.79; 95% CI, 0.62–1.00; P=.051). Overall response rate also was higher in the T-VEC arm (26.4%; 95% CI, 21.4%–31.5%) versus GM-CSF (5.7%; 95% CI, 1.9%–9.5%).
Talimogene laherparepvec is a herpes simplex virus type 1–derived oncolytic immunotherapy designed to replicate within tumors and produce GM-CSF, which enhances systemic antitumor immune responses and induces tumor lysis.
In this study, T-VEC efficacy was greatest in patients with stage IIIB, IIIC, or IVM1a melanomas and in patients with treatment-naive disease. Differences in DRRs in patients with stage IIIB/C melanomas were 33% in the T-VEC group versus 0% for patients treated with GM-CSF alone. In the stage IVM1a group, DRR was 16% with T-VEC versus 2% with GM-CSF. The difference between both treatments was smaller in more advanced melanomas (IVM1b group, 3% vs 4%; IVM1c, 7% vs 3%). In the first-line treatment, the DRR with T-VEC was 24% versus 0% with GM-CSF. In the second-line and beyond, the DRR with T-VEC was 10% compared to 4% for GM-CSF.
The main adverse events seen in this study were fatigue, chills, and pyrexia. Serious adverse events occurred in 25.7% and 13.4% of participants in the T-VEC and GM-CSF arms, respectively, with disease progression (3.1% vs 1.6%) and cellulitis (2.4% vs 0.8%) being the most common. Six immune-mediated events occurred in the T-VEC group compared to 3 in the GM-CSF group.
Twelve patient deaths occurred within 30 days of the last dose of T-VEC; 9 were associated with progressive disease and the other 3 were associated with myocardial infarction, cardiac arrest, and sepsis, respectively. Four patient deaths were reported in the GM-CSF arm within the same 30 days.
What’s the Issue?
Immunotherapy represents a promising treatment option for metastatic melanoma. These promising results along with the US Food and Drug Administration’s approval of T-VEC will lead to further studies of the uses of T-VEC in combination with other therapies, including a phase I/II study to assess T-VEC in combination with ipilimumab for unresected melanomas (NCT01740297) and a phase III study of T-VEC in combination with pembrolizumab for unresected melanomas (NCT02263508). It is important for dermatologists to be familiar with the new frontier of melanoma treatments. How will these new immunotherapies affect your treatment of melanoma?

Following a phase III, open-label trial conducted by Andtbacka et al (J Clin Oncol. 2015;33:2780-2788), the US Food and Drug Administration recently approved the first oncolytic immunotherapy talimogene laherparepvec (T-VEC) for the treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with advanced melanoma (stage IIIB/C–stage IV) following initial surgery.
A group of 436 patients with injectable melanomas (melanomas that are accessible via a percutaneous injection) that were not surgically resectable were randomly assigned (2:1) to treatment with intralesional T-VEC or subcutaneous granulocyte macrophage colony-stimulating factor (GM-CSF). The primary endpoint of the study was durable response rate (DRR), defined as objective response lasting continuously for 6 months or longer. Secondary endpoints included overall survival (OS) and overall response rate.
Talimogene laherparepvec was shown to extend DRRs compared to GM-CSF. Durable response rates were significantly higher with T-VEC (16.3%; 95% confidence interval [CI], 12.1%–20.5%) versus GM-CSF (2.1%; 95% CI, 0%–4.5%)(odds ratio, 8.9; P<.001).
In the OS analysis, a 4.4-month extension with T-VEC was observed; however, this was not deemed to be statistically significant (P=.051). The median OS was 23.3 months (95% CI, 19.5–29.6 months) with T-VEC and 18.9 months (95% CI, 16.0–23.7 months) with GM-CSF (hazard ratio, 0.79; 95% CI, 0.62–1.00; P=.051). Overall response rate also was higher in the T-VEC arm (26.4%; 95% CI, 21.4%–31.5%) versus GM-CSF (5.7%; 95% CI, 1.9%–9.5%).
Talimogene laherparepvec is a herpes simplex virus type 1–derived oncolytic immunotherapy designed to replicate within tumors and produce GM-CSF, which enhances systemic antitumor immune responses and induces tumor lysis.
In this study, T-VEC efficacy was greatest in patients with stage IIIB, IIIC, or IVM1a melanomas and in patients with treatment-naive disease. Differences in DRRs in patients with stage IIIB/C melanomas were 33% in the T-VEC group versus 0% for patients treated with GM-CSF alone. In the stage IVM1a group, DRR was 16% with T-VEC versus 2% with GM-CSF. The difference between both treatments was smaller in more advanced melanomas (IVM1b group, 3% vs 4%; IVM1c, 7% vs 3%). In the first-line treatment, the DRR with T-VEC was 24% versus 0% with GM-CSF. In the second-line and beyond, the DRR with T-VEC was 10% compared to 4% for GM-CSF.
The main adverse events seen in this study were fatigue, chills, and pyrexia. Serious adverse events occurred in 25.7% and 13.4% of participants in the T-VEC and GM-CSF arms, respectively, with disease progression (3.1% vs 1.6%) and cellulitis (2.4% vs 0.8%) being the most common. Six immune-mediated events occurred in the T-VEC group compared to 3 in the GM-CSF group.
Twelve patient deaths occurred within 30 days of the last dose of T-VEC; 9 were associated with progressive disease and the other 3 were associated with myocardial infarction, cardiac arrest, and sepsis, respectively. Four patient deaths were reported in the GM-CSF arm within the same 30 days.
What’s the Issue?
Immunotherapy represents a promising treatment option for metastatic melanoma. These promising results along with the US Food and Drug Administration’s approval of T-VEC will lead to further studies of the uses of T-VEC in combination with other therapies, including a phase I/II study to assess T-VEC in combination with ipilimumab for unresected melanomas (NCT01740297) and a phase III study of T-VEC in combination with pembrolizumab for unresected melanomas (NCT02263508). It is important for dermatologists to be familiar with the new frontier of melanoma treatments. How will these new immunotherapies affect your treatment of melanoma?

Following a phase III, open-label trial conducted by Andtbacka et al (J Clin Oncol. 2015;33:2780-2788), the US Food and Drug Administration recently approved the first oncolytic immunotherapy talimogene laherparepvec (T-VEC) for the treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with advanced melanoma (stage IIIB/C–stage IV) following initial surgery.
A group of 436 patients with injectable melanomas (melanomas that are accessible via a percutaneous injection) that were not surgically resectable were randomly assigned (2:1) to treatment with intralesional T-VEC or subcutaneous granulocyte macrophage colony-stimulating factor (GM-CSF). The primary endpoint of the study was durable response rate (DRR), defined as objective response lasting continuously for 6 months or longer. Secondary endpoints included overall survival (OS) and overall response rate.
Talimogene laherparepvec was shown to extend DRRs compared to GM-CSF. Durable response rates were significantly higher with T-VEC (16.3%; 95% confidence interval [CI], 12.1%–20.5%) versus GM-CSF (2.1%; 95% CI, 0%–4.5%)(odds ratio, 8.9; P<.001).
In the OS analysis, a 4.4-month extension with T-VEC was observed; however, this was not deemed to be statistically significant (P=.051). The median OS was 23.3 months (95% CI, 19.5–29.6 months) with T-VEC and 18.9 months (95% CI, 16.0–23.7 months) with GM-CSF (hazard ratio, 0.79; 95% CI, 0.62–1.00; P=.051). Overall response rate also was higher in the T-VEC arm (26.4%; 95% CI, 21.4%–31.5%) versus GM-CSF (5.7%; 95% CI, 1.9%–9.5%).
Talimogene laherparepvec is a herpes simplex virus type 1–derived oncolytic immunotherapy designed to replicate within tumors and produce GM-CSF, which enhances systemic antitumor immune responses and induces tumor lysis.
In this study, T-VEC efficacy was greatest in patients with stage IIIB, IIIC, or IVM1a melanomas and in patients with treatment-naive disease. Differences in DRRs in patients with stage IIIB/C melanomas were 33% in the T-VEC group versus 0% for patients treated with GM-CSF alone. In the stage IVM1a group, DRR was 16% with T-VEC versus 2% with GM-CSF. The difference between both treatments was smaller in more advanced melanomas (IVM1b group, 3% vs 4%; IVM1c, 7% vs 3%). In the first-line treatment, the DRR with T-VEC was 24% versus 0% with GM-CSF. In the second-line and beyond, the DRR with T-VEC was 10% compared to 4% for GM-CSF.
The main adverse events seen in this study were fatigue, chills, and pyrexia. Serious adverse events occurred in 25.7% and 13.4% of participants in the T-VEC and GM-CSF arms, respectively, with disease progression (3.1% vs 1.6%) and cellulitis (2.4% vs 0.8%) being the most common. Six immune-mediated events occurred in the T-VEC group compared to 3 in the GM-CSF group.
Twelve patient deaths occurred within 30 days of the last dose of T-VEC; 9 were associated with progressive disease and the other 3 were associated with myocardial infarction, cardiac arrest, and sepsis, respectively. Four patient deaths were reported in the GM-CSF arm within the same 30 days.
What’s the Issue?
Immunotherapy represents a promising treatment option for metastatic melanoma. These promising results along with the US Food and Drug Administration’s approval of T-VEC will lead to further studies of the uses of T-VEC in combination with other therapies, including a phase I/II study to assess T-VEC in combination with ipilimumab for unresected melanomas (NCT01740297) and a phase III study of T-VEC in combination with pembrolizumab for unresected melanomas (NCT02263508). It is important for dermatologists to be familiar with the new frontier of melanoma treatments. How will these new immunotherapies affect your treatment of melanoma?
FDA approves pembrolizumab as first-line advanced melanoma therapy
Pembrolizumab, the programmed death receptor-1 (PD-1)–blocking antibody, is now approved as a first-line treatment of unresectable or metastatic melanoma, based on the results of the KEYNOTE-006 study.
This is the second approval for an advanced melanoma indication for pembrolizumab (Keytruda), according to a statement issued by Merck, the manufacturer. Pembrolizumab was first approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab and, if BRAF V600 mutation positive, a BRAF inhibitor.
The FDA-approved dose is 2 mg/kg every 3 weeks.
In KEYNOTE-006, a phase III study that compared pembrolizumab to ipilimumab (Yervoy), also a PD-1–blocking antibody, in 834 patients with unresectable or metastatic melanoma with progression of disease, overall survival and progression-free survival were significantly increased among those treated with pembrolizumab, compared with ipilimumab.
The results were reported at the annual meeting of the American Association for Cancer Research in April, and published simultaneously in the New England Journal of Medicine (2015; 372:2521-32).
Pembrolizumab, the programmed death receptor-1 (PD-1)–blocking antibody, is now approved as a first-line treatment of unresectable or metastatic melanoma, based on the results of the KEYNOTE-006 study.
This is the second approval for an advanced melanoma indication for pembrolizumab (Keytruda), according to a statement issued by Merck, the manufacturer. Pembrolizumab was first approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab and, if BRAF V600 mutation positive, a BRAF inhibitor.
The FDA-approved dose is 2 mg/kg every 3 weeks.
In KEYNOTE-006, a phase III study that compared pembrolizumab to ipilimumab (Yervoy), also a PD-1–blocking antibody, in 834 patients with unresectable or metastatic melanoma with progression of disease, overall survival and progression-free survival were significantly increased among those treated with pembrolizumab, compared with ipilimumab.
The results were reported at the annual meeting of the American Association for Cancer Research in April, and published simultaneously in the New England Journal of Medicine (2015; 372:2521-32).
Pembrolizumab, the programmed death receptor-1 (PD-1)–blocking antibody, is now approved as a first-line treatment of unresectable or metastatic melanoma, based on the results of the KEYNOTE-006 study.
This is the second approval for an advanced melanoma indication for pembrolizumab (Keytruda), according to a statement issued by Merck, the manufacturer. Pembrolizumab was first approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab and, if BRAF V600 mutation positive, a BRAF inhibitor.
The FDA-approved dose is 2 mg/kg every 3 weeks.
In KEYNOTE-006, a phase III study that compared pembrolizumab to ipilimumab (Yervoy), also a PD-1–blocking antibody, in 834 patients with unresectable or metastatic melanoma with progression of disease, overall survival and progression-free survival were significantly increased among those treated with pembrolizumab, compared with ipilimumab.
The results were reported at the annual meeting of the American Association for Cancer Research in April, and published simultaneously in the New England Journal of Medicine (2015; 372:2521-32).







