User login
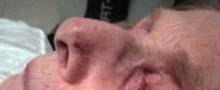
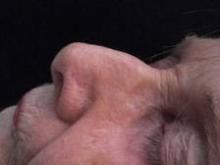
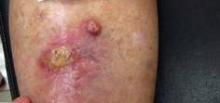
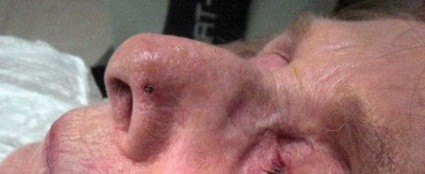
Blood Pressure Meds Linked to Lip Cancer
Commonly used photosensitizing antihypertensive drugs increase the risk for lip cancer by two- to fourfold, according to a study published online Aug. 6 in Archives of Internal Medicine.
"Lip cancer is rare, and an increased risk of its development is generally outweighed by the benefits of drugs that are effective for other conditions. However, physicians prescribing photosensitizing drugs should ascertain whether patients are at high risk of lip cancer because of their fair skin and long-term sun exposure, and discuss lip protection with them," wrote Dr. Gary D. Friedman of the division of research, Kaiser Permanente Medical Care Program, Oakland Calif., and his colleagues. "Likely preventive measures are simple: a hat with a sufficiently wide brim to shade the lips, and lip sunscreens," they added.
The diuretics hydrochlorothiazide and hydrochlorothiazide-triamterene, as well as the calcium channel blocker nifedipine, were associated with at least a doubling in the risk for lip cancer in a study involving 23,616 hypertensive non-Hispanic white adults. The association between lip cancer and the ACE inhibitor lisinopril, which also is photosensitizing, was characterized as "equivocal." And atenolol, a beta-adrenergic blocker that is not photosensitizing, was not linked to a higher risk.
Dr. Friedman and his colleagues used prescription data from the Kaiser Permanente pharmacy database and information from its cancer registry to perform a case-control study examining the relationship between the four classes of antihypertensive medications and lip cancer. They assessed the period from 1994 through 2008. The database includes an ethnically and socioeconomically diverse population of residents in the San Francisco and central valley regions of California.
There were few cases of lip cancer among nonwhite patients, so the analysis was restricted to white patients. Those with human immunodeficiency virus infection and those taking immunosuppressants after receiving organ transplants also were excluded because these factors were likely to be confounders.
The study population comprised 712 patients with lip cancer and 22,904 age-matched control patients. Nearly all malignancies were squamous cell. As expected, cigarette smokers in both study groups were more likely than nonsmokers to develop lip cancer.
The risk of developing lip cancer showed a dose-response relationship with the use of certain antihypertensive drugs, with the risk increasing as the duration of drug use increased. For patients treated with hydrochlorothiazide for 5 years or more, the odds ratio of developing lip cancer was 4.22. The OR for the combination drug hydrochlorothiazide-triamterene was 2.82, and the OR for nifedipine was 2.50. The OR was of borderline significance for lisinopril (1.42).
In contrast, for patients treated with atenolol for 5 years or more, the risk of developing lip cancer was reduced, with an odds ratio of 0.54, the investigators reported (Arch. Intern. Med. 2012 Aug. 6 [doi:10.1001/archinternmed.2012.2754]).
It should not be surprising that previous large clinical trials of these drugs’ efficacy failed to find an association with lip cancer, because of the rarity of the malignancy, they added.
For example, one study of antihypertensive medications involved more than 33,000 patients followed for a mean of 5 years. And after the investigators excluded the nonwhite patients, adjusted for the much lower incidence of lip cancer in women (who comprised half of the study population) than in men, and considered the low background incidence of the malignancy, they found that only seven lip cancers would have been expected to be detected in all the treatment groups combined, reported Dr. Friedman, also of the department of health research and policy, Stanford (Calif.) University, and his associates.
"Although the relatively high odds ratios, the evidence for specificity, and the biological mechanism are consistent with a causal relationship, causality cannot usually be established by a single observational study such as ours. Further investigations are needed to confirm and characterize relationships between photosensitizing antihypertensive agents and lip cancer," the researchers noted.
The study was limited because the researchers were unable to account for patients’ sun exposure, which is the most important factor contributing to lip cancer risk. "However, it does not seem likely that users of the antihypertensive drugs associated with lip cancer experience a great deal more sun exposure than nonusers or than users of atenolol," they wrote.
This study was supported by the National Cancer Institute. Dr. Friedman reported ties to Allergan, and his associates reported ties to Genentech, Merck, Sanofi-Aventis, and Takeda.
In addition to having an increased risk for lip cancer, patients taking photosensitizing antihypertensive agents are probably also at an increased risk for basal and squamous cell cancers of the skin, noted Dr. Mitchell H. Katz.
Physicians should remind their patients of the simple measures available to reduce sun exposure. The findings of Dr. Friedman and his colleagues "are important because simple interventions, such as lip protector, sunscreen, large-brim hats, rash guard swim shirts, and avoiding times of the day when the sun is most intense, are likely to decrease the harmful effects of the sun for everyone, regardless of whether they are receiving a photosensitizing agent," he wrote.
Dr. Katz is a deputy editor of Archives of Internal Medicine and director of the Los Angeles County Department of Health Services. He reported no relevant financial conflicts. His remarks were taken from the Editor’s Note accompanying Dr. Friedman’s report.
In addition to having an increased risk for lip cancer, patients taking photosensitizing antihypertensive agents are probably also at an increased risk for basal and squamous cell cancers of the skin, noted Dr. Mitchell H. Katz.
Physicians should remind their patients of the simple measures available to reduce sun exposure. The findings of Dr. Friedman and his colleagues "are important because simple interventions, such as lip protector, sunscreen, large-brim hats, rash guard swim shirts, and avoiding times of the day when the sun is most intense, are likely to decrease the harmful effects of the sun for everyone, regardless of whether they are receiving a photosensitizing agent," he wrote.
Dr. Katz is a deputy editor of Archives of Internal Medicine and director of the Los Angeles County Department of Health Services. He reported no relevant financial conflicts. His remarks were taken from the Editor’s Note accompanying Dr. Friedman’s report.
In addition to having an increased risk for lip cancer, patients taking photosensitizing antihypertensive agents are probably also at an increased risk for basal and squamous cell cancers of the skin, noted Dr. Mitchell H. Katz.
Physicians should remind their patients of the simple measures available to reduce sun exposure. The findings of Dr. Friedman and his colleagues "are important because simple interventions, such as lip protector, sunscreen, large-brim hats, rash guard swim shirts, and avoiding times of the day when the sun is most intense, are likely to decrease the harmful effects of the sun for everyone, regardless of whether they are receiving a photosensitizing agent," he wrote.
Dr. Katz is a deputy editor of Archives of Internal Medicine and director of the Los Angeles County Department of Health Services. He reported no relevant financial conflicts. His remarks were taken from the Editor’s Note accompanying Dr. Friedman’s report.
Commonly used photosensitizing antihypertensive drugs increase the risk for lip cancer by two- to fourfold, according to a study published online Aug. 6 in Archives of Internal Medicine.
"Lip cancer is rare, and an increased risk of its development is generally outweighed by the benefits of drugs that are effective for other conditions. However, physicians prescribing photosensitizing drugs should ascertain whether patients are at high risk of lip cancer because of their fair skin and long-term sun exposure, and discuss lip protection with them," wrote Dr. Gary D. Friedman of the division of research, Kaiser Permanente Medical Care Program, Oakland Calif., and his colleagues. "Likely preventive measures are simple: a hat with a sufficiently wide brim to shade the lips, and lip sunscreens," they added.
The diuretics hydrochlorothiazide and hydrochlorothiazide-triamterene, as well as the calcium channel blocker nifedipine, were associated with at least a doubling in the risk for lip cancer in a study involving 23,616 hypertensive non-Hispanic white adults. The association between lip cancer and the ACE inhibitor lisinopril, which also is photosensitizing, was characterized as "equivocal." And atenolol, a beta-adrenergic blocker that is not photosensitizing, was not linked to a higher risk.
Dr. Friedman and his colleagues used prescription data from the Kaiser Permanente pharmacy database and information from its cancer registry to perform a case-control study examining the relationship between the four classes of antihypertensive medications and lip cancer. They assessed the period from 1994 through 2008. The database includes an ethnically and socioeconomically diverse population of residents in the San Francisco and central valley regions of California.
There were few cases of lip cancer among nonwhite patients, so the analysis was restricted to white patients. Those with human immunodeficiency virus infection and those taking immunosuppressants after receiving organ transplants also were excluded because these factors were likely to be confounders.
The study population comprised 712 patients with lip cancer and 22,904 age-matched control patients. Nearly all malignancies were squamous cell. As expected, cigarette smokers in both study groups were more likely than nonsmokers to develop lip cancer.
The risk of developing lip cancer showed a dose-response relationship with the use of certain antihypertensive drugs, with the risk increasing as the duration of drug use increased. For patients treated with hydrochlorothiazide for 5 years or more, the odds ratio of developing lip cancer was 4.22. The OR for the combination drug hydrochlorothiazide-triamterene was 2.82, and the OR for nifedipine was 2.50. The OR was of borderline significance for lisinopril (1.42).
In contrast, for patients treated with atenolol for 5 years or more, the risk of developing lip cancer was reduced, with an odds ratio of 0.54, the investigators reported (Arch. Intern. Med. 2012 Aug. 6 [doi:10.1001/archinternmed.2012.2754]).
It should not be surprising that previous large clinical trials of these drugs’ efficacy failed to find an association with lip cancer, because of the rarity of the malignancy, they added.
For example, one study of antihypertensive medications involved more than 33,000 patients followed for a mean of 5 years. And after the investigators excluded the nonwhite patients, adjusted for the much lower incidence of lip cancer in women (who comprised half of the study population) than in men, and considered the low background incidence of the malignancy, they found that only seven lip cancers would have been expected to be detected in all the treatment groups combined, reported Dr. Friedman, also of the department of health research and policy, Stanford (Calif.) University, and his associates.
"Although the relatively high odds ratios, the evidence for specificity, and the biological mechanism are consistent with a causal relationship, causality cannot usually be established by a single observational study such as ours. Further investigations are needed to confirm and characterize relationships between photosensitizing antihypertensive agents and lip cancer," the researchers noted.
The study was limited because the researchers were unable to account for patients’ sun exposure, which is the most important factor contributing to lip cancer risk. "However, it does not seem likely that users of the antihypertensive drugs associated with lip cancer experience a great deal more sun exposure than nonusers or than users of atenolol," they wrote.
This study was supported by the National Cancer Institute. Dr. Friedman reported ties to Allergan, and his associates reported ties to Genentech, Merck, Sanofi-Aventis, and Takeda.
Commonly used photosensitizing antihypertensive drugs increase the risk for lip cancer by two- to fourfold, according to a study published online Aug. 6 in Archives of Internal Medicine.
"Lip cancer is rare, and an increased risk of its development is generally outweighed by the benefits of drugs that are effective for other conditions. However, physicians prescribing photosensitizing drugs should ascertain whether patients are at high risk of lip cancer because of their fair skin and long-term sun exposure, and discuss lip protection with them," wrote Dr. Gary D. Friedman of the division of research, Kaiser Permanente Medical Care Program, Oakland Calif., and his colleagues. "Likely preventive measures are simple: a hat with a sufficiently wide brim to shade the lips, and lip sunscreens," they added.
The diuretics hydrochlorothiazide and hydrochlorothiazide-triamterene, as well as the calcium channel blocker nifedipine, were associated with at least a doubling in the risk for lip cancer in a study involving 23,616 hypertensive non-Hispanic white adults. The association between lip cancer and the ACE inhibitor lisinopril, which also is photosensitizing, was characterized as "equivocal." And atenolol, a beta-adrenergic blocker that is not photosensitizing, was not linked to a higher risk.
Dr. Friedman and his colleagues used prescription data from the Kaiser Permanente pharmacy database and information from its cancer registry to perform a case-control study examining the relationship between the four classes of antihypertensive medications and lip cancer. They assessed the period from 1994 through 2008. The database includes an ethnically and socioeconomically diverse population of residents in the San Francisco and central valley regions of California.
There were few cases of lip cancer among nonwhite patients, so the analysis was restricted to white patients. Those with human immunodeficiency virus infection and those taking immunosuppressants after receiving organ transplants also were excluded because these factors were likely to be confounders.
The study population comprised 712 patients with lip cancer and 22,904 age-matched control patients. Nearly all malignancies were squamous cell. As expected, cigarette smokers in both study groups were more likely than nonsmokers to develop lip cancer.
The risk of developing lip cancer showed a dose-response relationship with the use of certain antihypertensive drugs, with the risk increasing as the duration of drug use increased. For patients treated with hydrochlorothiazide for 5 years or more, the odds ratio of developing lip cancer was 4.22. The OR for the combination drug hydrochlorothiazide-triamterene was 2.82, and the OR for nifedipine was 2.50. The OR was of borderline significance for lisinopril (1.42).
In contrast, for patients treated with atenolol for 5 years or more, the risk of developing lip cancer was reduced, with an odds ratio of 0.54, the investigators reported (Arch. Intern. Med. 2012 Aug. 6 [doi:10.1001/archinternmed.2012.2754]).
It should not be surprising that previous large clinical trials of these drugs’ efficacy failed to find an association with lip cancer, because of the rarity of the malignancy, they added.
For example, one study of antihypertensive medications involved more than 33,000 patients followed for a mean of 5 years. And after the investigators excluded the nonwhite patients, adjusted for the much lower incidence of lip cancer in women (who comprised half of the study population) than in men, and considered the low background incidence of the malignancy, they found that only seven lip cancers would have been expected to be detected in all the treatment groups combined, reported Dr. Friedman, also of the department of health research and policy, Stanford (Calif.) University, and his associates.
"Although the relatively high odds ratios, the evidence for specificity, and the biological mechanism are consistent with a causal relationship, causality cannot usually be established by a single observational study such as ours. Further investigations are needed to confirm and characterize relationships between photosensitizing antihypertensive agents and lip cancer," the researchers noted.
The study was limited because the researchers were unable to account for patients’ sun exposure, which is the most important factor contributing to lip cancer risk. "However, it does not seem likely that users of the antihypertensive drugs associated with lip cancer experience a great deal more sun exposure than nonusers or than users of atenolol," they wrote.
This study was supported by the National Cancer Institute. Dr. Friedman reported ties to Allergan, and his associates reported ties to Genentech, Merck, Sanofi-Aventis, and Takeda.
FROM ARCHIVES OF INTERNAL MEDICINE
Major Finding: The odds ratio of patients developing lip cancer was 4.22 with hydrochlorothiazide, 2.82 with combination hydrochlorothiazide-triamterene, and 2.50 with nifedipine.
Data Source: An observational case-control study was conducted involving 712 patients taking antihypertensive medications for at least 5 years who developed lip cancer during a 14-year period and 22,904 control patients who did not.
Disclosures: This study was supported by the National Cancer Institute. Dr. Friedman reported ties to Allergan, and his associates reported ties to Genentech, Merck, Sanofi-Aventis, and Takeda.
Isotretinoin Quells EGFR Inhibitor-Related Rash
ORLANDO – Oral isotretinoin holds potential as a bridge therapy for cancer patients who develop severe rashes during treatment, according to Dr. Milan J. Anadkat.
Dermatologists can play an integral role here. "Most oncologists see these patients first, and most oncologists are not enrolled in the iPledge program," said Dr. Anadkat of the division of dermatology at Washington University in St. Louis.
He noted that all treatment options are off label because there are no Food and Drug Administration–approved agents to treat chemotherapy-related cutaneous toxicities.
Getting patients through rashes that occur within a week or two of beginning targeted chemotherapy is important, as patients who develop the greatest reactions tend to have cancers that respond best to treatment, said Dr. Anadkat at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery. For this reason, the management of these patients is more complicated than simple drug cessation.
"The problem with just taking them off the drug is that they have cancer. Ultimately, the big goal here is treating the cancer, not avoiding the rash," he said.
Tetracycline or doxycycline can also reduce the severity of the rash, but the timing of administration is important. Better outcomes are associated with prophylaxis that is timed with the initiation of EGFR (epidermal growth factor receptor) inhibitors (Cancer 2008;113:847-53). "Waiting for the rash to appear is not the time to give it. It makes a difference if you give at day 0, not in terms of incidence but in the severity of the rash," he said.
Educate patients that the rash typically appears in an estimated 60%-90% of people within 8-10 days of EGFR inhibitor initiation, with a peak presentation at 2-4 weeks. "Bridging them through with something as effective as isotretinoin is useful," said Dr. Anadkat.
The rash generally appears on the face and upper trunk, but be careful not to confuse the presentation with a photo-exposure phenomenon.
Although some oncologists will describe the skin eruptions as "acnelike," histology will show mixed inflammatory infiltrate and follicular rupture. For this reason, "topical acne medications do very little," Dr. Anadkat said. Also, rule out infection, except when patients present with pustules on their arms, legs, or other non–EGFR receptor areas.
EGFR inhibitors can cause inflammatory alopecia, eyelash trichomegaly, and periungual and nail alterations.
About one in six patients will develop periungual or nail abnormalities, typically on their first finger or toe. These effects can be painful, Dr. Anadkat said. Culture is mandatory to rule out superinfection.
Again, the approach is to get patients through the adverse events with petroleum jelly, high-dose topical steroids, or oral tetracyclines. "Tumor markers are going down; [oncologists] are not going to want to stop chemotherapy for a painful thumb or toe," he said. "I recommend antimicrobial soaks with bleach or vinegar to prevent paronychia superinfection."
Dry, itchy skin is another concern with long-term EGFR inhibitor treatment. Histology shows "stark differences" in the stratum corneum. "This is the No. 1 side effect for patients on long-term EGFR – 3 months or longer; [it is] not a grade 3 or higher toxicity, but it is annoying."
Investigators compared oral minocycline and topical tazarotene prophylaxis in a study of 48 patients with cetuximab associated rash (J. Clin. Oncol. 2007:25:5390-6). Although oral minocycline was associated with reduced lesion counts, topical tazarotene yielded no significant benefit. "Again, this is not acne," Dr. Anadkat said.
He disclosed being a consultant or speaker for AstraZeneca, Bristol-Myers Squibb, Eisai, Genentech, and ImClone regarding strategies for managing skin toxicities from chemotherapy. He has never prescribed chemotherapy agents.
ORLANDO – Oral isotretinoin holds potential as a bridge therapy for cancer patients who develop severe rashes during treatment, according to Dr. Milan J. Anadkat.
Dermatologists can play an integral role here. "Most oncologists see these patients first, and most oncologists are not enrolled in the iPledge program," said Dr. Anadkat of the division of dermatology at Washington University in St. Louis.
He noted that all treatment options are off label because there are no Food and Drug Administration–approved agents to treat chemotherapy-related cutaneous toxicities.
Getting patients through rashes that occur within a week or two of beginning targeted chemotherapy is important, as patients who develop the greatest reactions tend to have cancers that respond best to treatment, said Dr. Anadkat at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery. For this reason, the management of these patients is more complicated than simple drug cessation.
"The problem with just taking them off the drug is that they have cancer. Ultimately, the big goal here is treating the cancer, not avoiding the rash," he said.
Tetracycline or doxycycline can also reduce the severity of the rash, but the timing of administration is important. Better outcomes are associated with prophylaxis that is timed with the initiation of EGFR (epidermal growth factor receptor) inhibitors (Cancer 2008;113:847-53). "Waiting for the rash to appear is not the time to give it. It makes a difference if you give at day 0, not in terms of incidence but in the severity of the rash," he said.
Educate patients that the rash typically appears in an estimated 60%-90% of people within 8-10 days of EGFR inhibitor initiation, with a peak presentation at 2-4 weeks. "Bridging them through with something as effective as isotretinoin is useful," said Dr. Anadkat.
The rash generally appears on the face and upper trunk, but be careful not to confuse the presentation with a photo-exposure phenomenon.
Although some oncologists will describe the skin eruptions as "acnelike," histology will show mixed inflammatory infiltrate and follicular rupture. For this reason, "topical acne medications do very little," Dr. Anadkat said. Also, rule out infection, except when patients present with pustules on their arms, legs, or other non–EGFR receptor areas.
EGFR inhibitors can cause inflammatory alopecia, eyelash trichomegaly, and periungual and nail alterations.
About one in six patients will develop periungual or nail abnormalities, typically on their first finger or toe. These effects can be painful, Dr. Anadkat said. Culture is mandatory to rule out superinfection.
Again, the approach is to get patients through the adverse events with petroleum jelly, high-dose topical steroids, or oral tetracyclines. "Tumor markers are going down; [oncologists] are not going to want to stop chemotherapy for a painful thumb or toe," he said. "I recommend antimicrobial soaks with bleach or vinegar to prevent paronychia superinfection."
Dry, itchy skin is another concern with long-term EGFR inhibitor treatment. Histology shows "stark differences" in the stratum corneum. "This is the No. 1 side effect for patients on long-term EGFR – 3 months or longer; [it is] not a grade 3 or higher toxicity, but it is annoying."
Investigators compared oral minocycline and topical tazarotene prophylaxis in a study of 48 patients with cetuximab associated rash (J. Clin. Oncol. 2007:25:5390-6). Although oral minocycline was associated with reduced lesion counts, topical tazarotene yielded no significant benefit. "Again, this is not acne," Dr. Anadkat said.
He disclosed being a consultant or speaker for AstraZeneca, Bristol-Myers Squibb, Eisai, Genentech, and ImClone regarding strategies for managing skin toxicities from chemotherapy. He has never prescribed chemotherapy agents.
ORLANDO – Oral isotretinoin holds potential as a bridge therapy for cancer patients who develop severe rashes during treatment, according to Dr. Milan J. Anadkat.
Dermatologists can play an integral role here. "Most oncologists see these patients first, and most oncologists are not enrolled in the iPledge program," said Dr. Anadkat of the division of dermatology at Washington University in St. Louis.
He noted that all treatment options are off label because there are no Food and Drug Administration–approved agents to treat chemotherapy-related cutaneous toxicities.
Getting patients through rashes that occur within a week or two of beginning targeted chemotherapy is important, as patients who develop the greatest reactions tend to have cancers that respond best to treatment, said Dr. Anadkat at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery. For this reason, the management of these patients is more complicated than simple drug cessation.
"The problem with just taking them off the drug is that they have cancer. Ultimately, the big goal here is treating the cancer, not avoiding the rash," he said.
Tetracycline or doxycycline can also reduce the severity of the rash, but the timing of administration is important. Better outcomes are associated with prophylaxis that is timed with the initiation of EGFR (epidermal growth factor receptor) inhibitors (Cancer 2008;113:847-53). "Waiting for the rash to appear is not the time to give it. It makes a difference if you give at day 0, not in terms of incidence but in the severity of the rash," he said.
Educate patients that the rash typically appears in an estimated 60%-90% of people within 8-10 days of EGFR inhibitor initiation, with a peak presentation at 2-4 weeks. "Bridging them through with something as effective as isotretinoin is useful," said Dr. Anadkat.
The rash generally appears on the face and upper trunk, but be careful not to confuse the presentation with a photo-exposure phenomenon.
Although some oncologists will describe the skin eruptions as "acnelike," histology will show mixed inflammatory infiltrate and follicular rupture. For this reason, "topical acne medications do very little," Dr. Anadkat said. Also, rule out infection, except when patients present with pustules on their arms, legs, or other non–EGFR receptor areas.
EGFR inhibitors can cause inflammatory alopecia, eyelash trichomegaly, and periungual and nail alterations.
About one in six patients will develop periungual or nail abnormalities, typically on their first finger or toe. These effects can be painful, Dr. Anadkat said. Culture is mandatory to rule out superinfection.
Again, the approach is to get patients through the adverse events with petroleum jelly, high-dose topical steroids, or oral tetracyclines. "Tumor markers are going down; [oncologists] are not going to want to stop chemotherapy for a painful thumb or toe," he said. "I recommend antimicrobial soaks with bleach or vinegar to prevent paronychia superinfection."
Dry, itchy skin is another concern with long-term EGFR inhibitor treatment. Histology shows "stark differences" in the stratum corneum. "This is the No. 1 side effect for patients on long-term EGFR – 3 months or longer; [it is] not a grade 3 or higher toxicity, but it is annoying."
Investigators compared oral minocycline and topical tazarotene prophylaxis in a study of 48 patients with cetuximab associated rash (J. Clin. Oncol. 2007:25:5390-6). Although oral minocycline was associated with reduced lesion counts, topical tazarotene yielded no significant benefit. "Again, this is not acne," Dr. Anadkat said.
He disclosed being a consultant or speaker for AstraZeneca, Bristol-Myers Squibb, Eisai, Genentech, and ImClone regarding strategies for managing skin toxicities from chemotherapy. He has never prescribed chemotherapy agents.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGY AND DERMATOLOGIC SURGERY
Labs Find Evidence of Cancer Stem Cells
In an era of targeted cancer therapies, laboratory scientists working with mice may have found the ultimate target – a reservoir of stem cells that drive cancers to grow and metastasize.
Separate reports in the journals Science and Nature document the presence of cancer stem cells in intestinal adenomas (Science 2012 Aug. 1 [doi:10.1126/science.1224676]), squamous skin cancer, (Nature 2012 Aug. 1 [doi:10.1038/nature11344]), and glioblastoma multiforme (Nature 2012 Aug. 1 [doi:10.1038/nature11287]).
In the last study, mice with these highly lethal brain tumors were given temozolomide (Temodar), an approved treatment in humans, along with ganciclovir, an antiviral. Despite a transient therapeutic response to chemotherapy, the cancers continued to grow, driven by "a relatively quiescent subset of endogenous glioma cells, with properties similar to those proposed for cancer stem cells," the authors wrote.
Whether these reports will resolve controversy over the existence of stem cells or lead to clinically meaningful treatments remains to be seen. There is no doubt, however, that they will lead to further investigation.
In an era of targeted cancer therapies, laboratory scientists working with mice may have found the ultimate target – a reservoir of stem cells that drive cancers to grow and metastasize.
Separate reports in the journals Science and Nature document the presence of cancer stem cells in intestinal adenomas (Science 2012 Aug. 1 [doi:10.1126/science.1224676]), squamous skin cancer, (Nature 2012 Aug. 1 [doi:10.1038/nature11344]), and glioblastoma multiforme (Nature 2012 Aug. 1 [doi:10.1038/nature11287]).
In the last study, mice with these highly lethal brain tumors were given temozolomide (Temodar), an approved treatment in humans, along with ganciclovir, an antiviral. Despite a transient therapeutic response to chemotherapy, the cancers continued to grow, driven by "a relatively quiescent subset of endogenous glioma cells, with properties similar to those proposed for cancer stem cells," the authors wrote.
Whether these reports will resolve controversy over the existence of stem cells or lead to clinically meaningful treatments remains to be seen. There is no doubt, however, that they will lead to further investigation.
In an era of targeted cancer therapies, laboratory scientists working with mice may have found the ultimate target – a reservoir of stem cells that drive cancers to grow and metastasize.
Separate reports in the journals Science and Nature document the presence of cancer stem cells in intestinal adenomas (Science 2012 Aug. 1 [doi:10.1126/science.1224676]), squamous skin cancer, (Nature 2012 Aug. 1 [doi:10.1038/nature11344]), and glioblastoma multiforme (Nature 2012 Aug. 1 [doi:10.1038/nature11287]).
In the last study, mice with these highly lethal brain tumors were given temozolomide (Temodar), an approved treatment in humans, along with ganciclovir, an antiviral. Despite a transient therapeutic response to chemotherapy, the cancers continued to grow, driven by "a relatively quiescent subset of endogenous glioma cells, with properties similar to those proposed for cancer stem cells," the authors wrote.
Whether these reports will resolve controversy over the existence of stem cells or lead to clinically meaningful treatments remains to be seen. There is no doubt, however, that they will lead to further investigation.
Skin Flaps Remedy Defects of the Ear
SAN DIEGO – In the clinical experience of Dr. Michael A. Keefe, 70%-80% of ear defects from auricular cancer treatment can be easily remedied with skin flaps.
The most common locations of auricular cancer are the helix, the posterior auricle skin, and the antihelix, Dr. Keefe said at a meeting on superficial anatomy and cutaneous surgery.
"More than 70% of lesions are smaller than 3 cm in size, and auricular lesions make up an estimated 8% of all skin cancers," said Dr. Keefe, a plastic surgeon with the division of head and neck surgery at Sharp Rees-Stealy Medical Group in San Diego. "The defects are unique, and the underlying cartilage structure makes it all the more interesting."
And challenging – defects may be located on the skin of the ear only, on the lateral side, or on the posterior side, or they may involve a combination of skin and cartilage. Healing by secondary intention is effective for concave defects, but the size of the defect drives the reconstruction options. "If there is no perichondrium, punch holes through cartilage with a 2-3 mm punch to allow granulation tissue to grow through, and then use a skin graft or allow it to heal with secondary intention," he said. "Keep the area moist with antibiotic ointment."
Options for reconstruction of defects in the middle one-third of the ear include primary closure, full-thickness skin grafts (FTSGs), the helical advancement flap, and the retroauricular composite advancement flap, while options for defects in the lower one-third of the ear include primary closure and the preauricular tubed flap. Options for reconstruction of defects in the upper one-third of the ear include primary closure, FTSGs, the helical advancement flap, the retroauricular and preauricular tubed flaps, and constructing an autogenous cartilage framework with FTSGs.
Dr. Keefe said that most small helical rim defects limited to the skin can be closed primarily. "There might be slight rim asymmetry [after closure]," he said at the meeting, which was sponsored by the University of California, San Diego, School of Medicine and the Scripps Clinic. "Some patients might not care [about this], but you have to advise them of that," he added.
A bilobed advancement flap is another option for helical rim defects limited to the skin. This flap "works well for cutaneous defects 2 cm or smaller in the helical rim or the posterior auricle," he said. "The other thing you can do with these bilobed flaps is advance them over the edge to correct helical rim defects."
The banner flap is another effective flap for helical rim defects, especially those located on the superior helix. It does not replace cartilage, but it conceals the incision well. For small composite helix and anterior defects, Dr. Keefe favors the chondrocutaneous advancement flap.
He said that he favors using FTSGs on the anterior surface of the helix for skin defects whenever possible. "You can use a composite skin graft as well, especially to replace cartilage or skin defects that are smaller than 1 cm in size," he said. "A FTSG is easy to harvest and has minimal contraction. Common donor sites include the preauricular, postauricular, supraclavicular, and clavicular regions. Make sure you trim off the fat." For posterior surface defects, the bilobe or advancement flaps work well.
Grafts must be placed on tissue with an adequate blood supply. Effective grafts establish imbibition in the first 24 hours, inosculation within 48-72 hours, and restoration of circulation within 4-7 days.
Dr. Keefe said that he had no relevant financial conflicts to disclose.
SAN DIEGO – In the clinical experience of Dr. Michael A. Keefe, 70%-80% of ear defects from auricular cancer treatment can be easily remedied with skin flaps.
The most common locations of auricular cancer are the helix, the posterior auricle skin, and the antihelix, Dr. Keefe said at a meeting on superficial anatomy and cutaneous surgery.
"More than 70% of lesions are smaller than 3 cm in size, and auricular lesions make up an estimated 8% of all skin cancers," said Dr. Keefe, a plastic surgeon with the division of head and neck surgery at Sharp Rees-Stealy Medical Group in San Diego. "The defects are unique, and the underlying cartilage structure makes it all the more interesting."
And challenging – defects may be located on the skin of the ear only, on the lateral side, or on the posterior side, or they may involve a combination of skin and cartilage. Healing by secondary intention is effective for concave defects, but the size of the defect drives the reconstruction options. "If there is no perichondrium, punch holes through cartilage with a 2-3 mm punch to allow granulation tissue to grow through, and then use a skin graft or allow it to heal with secondary intention," he said. "Keep the area moist with antibiotic ointment."
Options for reconstruction of defects in the middle one-third of the ear include primary closure, full-thickness skin grafts (FTSGs), the helical advancement flap, and the retroauricular composite advancement flap, while options for defects in the lower one-third of the ear include primary closure and the preauricular tubed flap. Options for reconstruction of defects in the upper one-third of the ear include primary closure, FTSGs, the helical advancement flap, the retroauricular and preauricular tubed flaps, and constructing an autogenous cartilage framework with FTSGs.
Dr. Keefe said that most small helical rim defects limited to the skin can be closed primarily. "There might be slight rim asymmetry [after closure]," he said at the meeting, which was sponsored by the University of California, San Diego, School of Medicine and the Scripps Clinic. "Some patients might not care [about this], but you have to advise them of that," he added.
A bilobed advancement flap is another option for helical rim defects limited to the skin. This flap "works well for cutaneous defects 2 cm or smaller in the helical rim or the posterior auricle," he said. "The other thing you can do with these bilobed flaps is advance them over the edge to correct helical rim defects."
The banner flap is another effective flap for helical rim defects, especially those located on the superior helix. It does not replace cartilage, but it conceals the incision well. For small composite helix and anterior defects, Dr. Keefe favors the chondrocutaneous advancement flap.
He said that he favors using FTSGs on the anterior surface of the helix for skin defects whenever possible. "You can use a composite skin graft as well, especially to replace cartilage or skin defects that are smaller than 1 cm in size," he said. "A FTSG is easy to harvest and has minimal contraction. Common donor sites include the preauricular, postauricular, supraclavicular, and clavicular regions. Make sure you trim off the fat." For posterior surface defects, the bilobe or advancement flaps work well.
Grafts must be placed on tissue with an adequate blood supply. Effective grafts establish imbibition in the first 24 hours, inosculation within 48-72 hours, and restoration of circulation within 4-7 days.
Dr. Keefe said that he had no relevant financial conflicts to disclose.
SAN DIEGO – In the clinical experience of Dr. Michael A. Keefe, 70%-80% of ear defects from auricular cancer treatment can be easily remedied with skin flaps.
The most common locations of auricular cancer are the helix, the posterior auricle skin, and the antihelix, Dr. Keefe said at a meeting on superficial anatomy and cutaneous surgery.
"More than 70% of lesions are smaller than 3 cm in size, and auricular lesions make up an estimated 8% of all skin cancers," said Dr. Keefe, a plastic surgeon with the division of head and neck surgery at Sharp Rees-Stealy Medical Group in San Diego. "The defects are unique, and the underlying cartilage structure makes it all the more interesting."
And challenging – defects may be located on the skin of the ear only, on the lateral side, or on the posterior side, or they may involve a combination of skin and cartilage. Healing by secondary intention is effective for concave defects, but the size of the defect drives the reconstruction options. "If there is no perichondrium, punch holes through cartilage with a 2-3 mm punch to allow granulation tissue to grow through, and then use a skin graft or allow it to heal with secondary intention," he said. "Keep the area moist with antibiotic ointment."
Options for reconstruction of defects in the middle one-third of the ear include primary closure, full-thickness skin grafts (FTSGs), the helical advancement flap, and the retroauricular composite advancement flap, while options for defects in the lower one-third of the ear include primary closure and the preauricular tubed flap. Options for reconstruction of defects in the upper one-third of the ear include primary closure, FTSGs, the helical advancement flap, the retroauricular and preauricular tubed flaps, and constructing an autogenous cartilage framework with FTSGs.
Dr. Keefe said that most small helical rim defects limited to the skin can be closed primarily. "There might be slight rim asymmetry [after closure]," he said at the meeting, which was sponsored by the University of California, San Diego, School of Medicine and the Scripps Clinic. "Some patients might not care [about this], but you have to advise them of that," he added.
A bilobed advancement flap is another option for helical rim defects limited to the skin. This flap "works well for cutaneous defects 2 cm or smaller in the helical rim or the posterior auricle," he said. "The other thing you can do with these bilobed flaps is advance them over the edge to correct helical rim defects."
The banner flap is another effective flap for helical rim defects, especially those located on the superior helix. It does not replace cartilage, but it conceals the incision well. For small composite helix and anterior defects, Dr. Keefe favors the chondrocutaneous advancement flap.
He said that he favors using FTSGs on the anterior surface of the helix for skin defects whenever possible. "You can use a composite skin graft as well, especially to replace cartilage or skin defects that are smaller than 1 cm in size," he said. "A FTSG is easy to harvest and has minimal contraction. Common donor sites include the preauricular, postauricular, supraclavicular, and clavicular regions. Make sure you trim off the fat." For posterior surface defects, the bilobe or advancement flaps work well.
Grafts must be placed on tissue with an adequate blood supply. Effective grafts establish imbibition in the first 24 hours, inosculation within 48-72 hours, and restoration of circulation within 4-7 days.
Dr. Keefe said that he had no relevant financial conflicts to disclose.
EXPERT ANALYSIS FROM A MEETING ON SUPERFICIAL ANATOMY AND CUTANEOUS SURGERY
Expert: Reclaim Radiation for Skin Cancer
ORLANDO – Careful patient selection is key to treating nonmelanoma skin cancer patients with superficial radiation therapy, according to Dr. William I. Roth.
"If you pick the right patients for radiation therapy, you can get great results," Dr. Roth said. An elderly patient with a basal cell or squamous cell carcinoma on the face or leg can be a good candidate, for example, especially if there are other comorbidities that make that person less than ideal as a candidate for surgery.
The typical skin cancer patient at Dr. Roth’s practice in Boynton Beach, Fla., is between 75 and 80 years old, and many are very active and play golf or tennis several times a week. They prefer radiation therapy over surgery because it is less likely to limit their activities or impinge on their lifestyle, he said.
Reduced risk of bleeding and infection are other advantages of radiation therapy compared with surgery, Dr. Roth said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"Radiation therapy is not going to replace Mohs. [But] I think skin cancer practices need both," said Dr. Roth, a volunteer clinical professor in the department of dermatology at the University of Miami.
Cure rates of up to 95% or more are possible with superficial radiation therapy. "Radiation oncologists in my area are advertising 98% cure rates," Dr. Roth said. "I do still send a lot of patients to radiation oncologists, but I think you get excellent results [within dermatology] as long as you pick the right patient. It’s a great therapy."
Referral is appropriate when a lesion is deep and significantly large, and its depth and width cannot be accurately determined, Dr. Roth said. Patients also should be referred to a specialist if there is any evidence of perineural involvement.
Patients should be advised that some immediate posttreatment hypopigmentation is possible and that it generally improves quickly. The treated area usually looks its worst at about 2 weeks post treatment, Dr. Roth said. Some patients experience late effects, even up to a decade post treatment.
"I always scallop my port in the lead shielding so that 10 years later when the person gets some hypopigmentation it’s not going to be sharply defined," he said.
Four weeks post therapy skin can still appear thick, said Dr. Roth. "I would not rush to rebiopsy these patients, especially on the legs where there is a low turnover."
Although some physicians avoid radiation treatment of basal cell or squamous cell carcinoma on the legs, Dr. Roth has found value to this approach. "The legs are not absolutely a piece of cake, but as you move [treatment] up from the middle of the leg it becomes easier and easier." He added that grafting can be a challenge on the legs and surgical sutures can sometime pull through the skin. "I’m happy to do surgery. But to be honest with you, radiation therapy on the legs generally is less of a hassle for the patient."
However, use caution when treating lesions directly over the anterior tibial surface, he advised. "You have to be more careful ... right over the bone." He encountered two patients treated in this area whose skin healed nicely but they still experienced some sensitivity in their tibia. Dr. Roth suggested less aggressive treatment near the anterior tibia because "bone absorbs radiation to a much greater degree than the skin."
Dr. Roth gave an example of a patient with two well differentiated SCC lesions on her leg that he treated with radiation therapy. Twelve fractions of radiation were delivered with the SRT-100 (Sensus Healthcare, Boca Raton, Fla.). The patient ulcerated and had a fairly brisk response but did not report any pain. Follow-up was conducted at months 1,2 and 5. "From my standpoint, it was a remarkably good response."
Results with the SRT-100 are very reproducible, Dr. Roth said. Energy imparted across the port or opening in the metal shield is consistent. The device delivers a flat field of radiation with a fairly fast drop-off deeper into the skin. The depth of the nonmelanoma skin cancer lesion generally determines the setting, Dr. Roth said.
The control unit is outside the radiation room, thus avoiding the need to work behind a leaded glass window. The control unit counts down duration of treatment and features an emergency stop. "I use a baby monitor to speak with patients during treatment," Dr. Roth said. "I did have one patient who yelled that her eye shields fell off. I hit the emergency button, entered the room, and put the shields back on." The unit holds the reading (pauses therapy) and then allows physicians to continue when ready.
"Radiation therapy has been a part of dermatology basically forever. We have been treating skin cancers since before I was born," Dr. Roth said. "Now that we have some more choices, this should be a part of dermatology again. We should take this back."
Dr. Roth is a consultant and researcher for Sensus, but purchased his SRT-100 at full price.
ORLANDO – Careful patient selection is key to treating nonmelanoma skin cancer patients with superficial radiation therapy, according to Dr. William I. Roth.
"If you pick the right patients for radiation therapy, you can get great results," Dr. Roth said. An elderly patient with a basal cell or squamous cell carcinoma on the face or leg can be a good candidate, for example, especially if there are other comorbidities that make that person less than ideal as a candidate for surgery.
The typical skin cancer patient at Dr. Roth’s practice in Boynton Beach, Fla., is between 75 and 80 years old, and many are very active and play golf or tennis several times a week. They prefer radiation therapy over surgery because it is less likely to limit their activities or impinge on their lifestyle, he said.
Reduced risk of bleeding and infection are other advantages of radiation therapy compared with surgery, Dr. Roth said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"Radiation therapy is not going to replace Mohs. [But] I think skin cancer practices need both," said Dr. Roth, a volunteer clinical professor in the department of dermatology at the University of Miami.
Cure rates of up to 95% or more are possible with superficial radiation therapy. "Radiation oncologists in my area are advertising 98% cure rates," Dr. Roth said. "I do still send a lot of patients to radiation oncologists, but I think you get excellent results [within dermatology] as long as you pick the right patient. It’s a great therapy."
Referral is appropriate when a lesion is deep and significantly large, and its depth and width cannot be accurately determined, Dr. Roth said. Patients also should be referred to a specialist if there is any evidence of perineural involvement.
Patients should be advised that some immediate posttreatment hypopigmentation is possible and that it generally improves quickly. The treated area usually looks its worst at about 2 weeks post treatment, Dr. Roth said. Some patients experience late effects, even up to a decade post treatment.
"I always scallop my port in the lead shielding so that 10 years later when the person gets some hypopigmentation it’s not going to be sharply defined," he said.
Four weeks post therapy skin can still appear thick, said Dr. Roth. "I would not rush to rebiopsy these patients, especially on the legs where there is a low turnover."
Although some physicians avoid radiation treatment of basal cell or squamous cell carcinoma on the legs, Dr. Roth has found value to this approach. "The legs are not absolutely a piece of cake, but as you move [treatment] up from the middle of the leg it becomes easier and easier." He added that grafting can be a challenge on the legs and surgical sutures can sometime pull through the skin. "I’m happy to do surgery. But to be honest with you, radiation therapy on the legs generally is less of a hassle for the patient."
However, use caution when treating lesions directly over the anterior tibial surface, he advised. "You have to be more careful ... right over the bone." He encountered two patients treated in this area whose skin healed nicely but they still experienced some sensitivity in their tibia. Dr. Roth suggested less aggressive treatment near the anterior tibia because "bone absorbs radiation to a much greater degree than the skin."
Dr. Roth gave an example of a patient with two well differentiated SCC lesions on her leg that he treated with radiation therapy. Twelve fractions of radiation were delivered with the SRT-100 (Sensus Healthcare, Boca Raton, Fla.). The patient ulcerated and had a fairly brisk response but did not report any pain. Follow-up was conducted at months 1,2 and 5. "From my standpoint, it was a remarkably good response."
Results with the SRT-100 are very reproducible, Dr. Roth said. Energy imparted across the port or opening in the metal shield is consistent. The device delivers a flat field of radiation with a fairly fast drop-off deeper into the skin. The depth of the nonmelanoma skin cancer lesion generally determines the setting, Dr. Roth said.
The control unit is outside the radiation room, thus avoiding the need to work behind a leaded glass window. The control unit counts down duration of treatment and features an emergency stop. "I use a baby monitor to speak with patients during treatment," Dr. Roth said. "I did have one patient who yelled that her eye shields fell off. I hit the emergency button, entered the room, and put the shields back on." The unit holds the reading (pauses therapy) and then allows physicians to continue when ready.
"Radiation therapy has been a part of dermatology basically forever. We have been treating skin cancers since before I was born," Dr. Roth said. "Now that we have some more choices, this should be a part of dermatology again. We should take this back."
Dr. Roth is a consultant and researcher for Sensus, but purchased his SRT-100 at full price.
ORLANDO – Careful patient selection is key to treating nonmelanoma skin cancer patients with superficial radiation therapy, according to Dr. William I. Roth.
"If you pick the right patients for radiation therapy, you can get great results," Dr. Roth said. An elderly patient with a basal cell or squamous cell carcinoma on the face or leg can be a good candidate, for example, especially if there are other comorbidities that make that person less than ideal as a candidate for surgery.
The typical skin cancer patient at Dr. Roth’s practice in Boynton Beach, Fla., is between 75 and 80 years old, and many are very active and play golf or tennis several times a week. They prefer radiation therapy over surgery because it is less likely to limit their activities or impinge on their lifestyle, he said.
Reduced risk of bleeding and infection are other advantages of radiation therapy compared with surgery, Dr. Roth said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"Radiation therapy is not going to replace Mohs. [But] I think skin cancer practices need both," said Dr. Roth, a volunteer clinical professor in the department of dermatology at the University of Miami.
Cure rates of up to 95% or more are possible with superficial radiation therapy. "Radiation oncologists in my area are advertising 98% cure rates," Dr. Roth said. "I do still send a lot of patients to radiation oncologists, but I think you get excellent results [within dermatology] as long as you pick the right patient. It’s a great therapy."
Referral is appropriate when a lesion is deep and significantly large, and its depth and width cannot be accurately determined, Dr. Roth said. Patients also should be referred to a specialist if there is any evidence of perineural involvement.
Patients should be advised that some immediate posttreatment hypopigmentation is possible and that it generally improves quickly. The treated area usually looks its worst at about 2 weeks post treatment, Dr. Roth said. Some patients experience late effects, even up to a decade post treatment.
"I always scallop my port in the lead shielding so that 10 years later when the person gets some hypopigmentation it’s not going to be sharply defined," he said.
Four weeks post therapy skin can still appear thick, said Dr. Roth. "I would not rush to rebiopsy these patients, especially on the legs where there is a low turnover."
Although some physicians avoid radiation treatment of basal cell or squamous cell carcinoma on the legs, Dr. Roth has found value to this approach. "The legs are not absolutely a piece of cake, but as you move [treatment] up from the middle of the leg it becomes easier and easier." He added that grafting can be a challenge on the legs and surgical sutures can sometime pull through the skin. "I’m happy to do surgery. But to be honest with you, radiation therapy on the legs generally is less of a hassle for the patient."
However, use caution when treating lesions directly over the anterior tibial surface, he advised. "You have to be more careful ... right over the bone." He encountered two patients treated in this area whose skin healed nicely but they still experienced some sensitivity in their tibia. Dr. Roth suggested less aggressive treatment near the anterior tibia because "bone absorbs radiation to a much greater degree than the skin."
Dr. Roth gave an example of a patient with two well differentiated SCC lesions on her leg that he treated with radiation therapy. Twelve fractions of radiation were delivered with the SRT-100 (Sensus Healthcare, Boca Raton, Fla.). The patient ulcerated and had a fairly brisk response but did not report any pain. Follow-up was conducted at months 1,2 and 5. "From my standpoint, it was a remarkably good response."
Results with the SRT-100 are very reproducible, Dr. Roth said. Energy imparted across the port or opening in the metal shield is consistent. The device delivers a flat field of radiation with a fairly fast drop-off deeper into the skin. The depth of the nonmelanoma skin cancer lesion generally determines the setting, Dr. Roth said.
The control unit is outside the radiation room, thus avoiding the need to work behind a leaded glass window. The control unit counts down duration of treatment and features an emergency stop. "I use a baby monitor to speak with patients during treatment," Dr. Roth said. "I did have one patient who yelled that her eye shields fell off. I hit the emergency button, entered the room, and put the shields back on." The unit holds the reading (pauses therapy) and then allows physicians to continue when ready.
"Radiation therapy has been a part of dermatology basically forever. We have been treating skin cancers since before I was born," Dr. Roth said. "Now that we have some more choices, this should be a part of dermatology again. We should take this back."
Dr. Roth is a consultant and researcher for Sensus, but purchased his SRT-100 at full price.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGY AND DERMATOLOGIC SURGERY
Washington Post Blasts Proliferation of ESAs for Anemia
Anemia drugs sold under the brand names of Procrit, Aranesp, and Epogen come under new and scathing scrutiny in an exclusive report published July 20 in the Washington Post.
The investigative article by Peter Whoriskey alleges that pharmaceutical giants Amgen and Johnson & Johnson "wildly overstated" benefits while understating potentially lethal side effects of these erythropoiesis-stimulating agents (ESAs).
While safety trials required by the Food and Drug Administration lagged for more than a decade, the companies successfully lobbied for a payment system that rewarded physicians for giving large doses of their high-priced drugs, according to the report.
Use of the drugs declined in recent years after studies showed higher mortality rates in patients given ESAs. Epoetin-alfa (Procrit and Epogen) and darbepoetin alfa (Aranesp) are used to treat anemia in patients undergoing cancer chemotherapy or dialysis for chronic kidney disease.
Anemia drugs sold under the brand names of Procrit, Aranesp, and Epogen come under new and scathing scrutiny in an exclusive report published July 20 in the Washington Post.
The investigative article by Peter Whoriskey alleges that pharmaceutical giants Amgen and Johnson & Johnson "wildly overstated" benefits while understating potentially lethal side effects of these erythropoiesis-stimulating agents (ESAs).
While safety trials required by the Food and Drug Administration lagged for more than a decade, the companies successfully lobbied for a payment system that rewarded physicians for giving large doses of their high-priced drugs, according to the report.
Use of the drugs declined in recent years after studies showed higher mortality rates in patients given ESAs. Epoetin-alfa (Procrit and Epogen) and darbepoetin alfa (Aranesp) are used to treat anemia in patients undergoing cancer chemotherapy or dialysis for chronic kidney disease.
Anemia drugs sold under the brand names of Procrit, Aranesp, and Epogen come under new and scathing scrutiny in an exclusive report published July 20 in the Washington Post.
The investigative article by Peter Whoriskey alleges that pharmaceutical giants Amgen and Johnson & Johnson "wildly overstated" benefits while understating potentially lethal side effects of these erythropoiesis-stimulating agents (ESAs).
While safety trials required by the Food and Drug Administration lagged for more than a decade, the companies successfully lobbied for a payment system that rewarded physicians for giving large doses of their high-priced drugs, according to the report.
Use of the drugs declined in recent years after studies showed higher mortality rates in patients given ESAs. Epoetin-alfa (Procrit and Epogen) and darbepoetin alfa (Aranesp) are used to treat anemia in patients undergoing cancer chemotherapy or dialysis for chronic kidney disease.
Frozen Fat Plumps Up Aging Face: The Skinny Vodcast
Skin & Allergy News Managing Editor Amy Pfeiffer and Senior Editor Terry Rudd review hot news in dermatology with the experts in this month's Skinny Vodcast.
Highlights include an interview with Dr. Axel Hauschild about two new promising treatments for BRAF-mutant melanoma. Doug Brunk talks to Dr. Christopher Zachary about a new light source that may someday replace lasers.
And, Dr. Lisa M. Donofrio discusses the advantages of fat transfer for volumizing the face.
Lastly, Dr. Lily Talakoub recommends a classification system for defining under eye circles.
Skin & Allergy News Managing Editor Amy Pfeiffer and Senior Editor Terry Rudd review hot news in dermatology with the experts in this month's Skinny Vodcast.
Highlights include an interview with Dr. Axel Hauschild about two new promising treatments for BRAF-mutant melanoma. Doug Brunk talks to Dr. Christopher Zachary about a new light source that may someday replace lasers.
And, Dr. Lisa M. Donofrio discusses the advantages of fat transfer for volumizing the face.
Lastly, Dr. Lily Talakoub recommends a classification system for defining under eye circles.
Skin & Allergy News Managing Editor Amy Pfeiffer and Senior Editor Terry Rudd review hot news in dermatology with the experts in this month's Skinny Vodcast.
Highlights include an interview with Dr. Axel Hauschild about two new promising treatments for BRAF-mutant melanoma. Doug Brunk talks to Dr. Christopher Zachary about a new light source that may someday replace lasers.
And, Dr. Lisa M. Donofrio discusses the advantages of fat transfer for volumizing the face.
Lastly, Dr. Lily Talakoub recommends a classification system for defining under eye circles.
Invasive SCC Rates Doubled in Last 20 Years
RALEIGH, N.C. – The incidence of invasive squamous cell carcinoma has more than doubled among U.S. nonphysician health professionals in the last 20 years, with marked sex differences evident in body-site distribution.
Dr. Khang Nguyen said his meta-analysis of data drawn from the Nurses' Health Study, the Nurses' Health Study II, and the Health Professionals Follow-Up Study was spurred by the fact that the National Cancer Institute’s SEER (Surveillance, Epidemiology and End Results) database and other national databases that record cancer statistics don’t track squamous cell carcinoma (SCC).
"Much of what we know about the epidemiology of squamous cell carcinoma comes from studies from the 1990s and earlier," noted Dr. Nguyen of Brigham and Women’s Hospital, Boston.
To help rectify this situation, he and his coinvestigators analyzed cases of pathologist-confirmed invasive SCC in the roughly 121,000 female participants in the Nurses’ Health Study, the 117,000 in the Nurses’ Health Study II, and 51,000 men in the Health Professionals Follow-Up Study. There were 1,580 cases in the Nurses’ Health Study, 468 in the Nurses’ Health Study II, and 1,194 in the Health Professionals Follow-Up Study.
Among participants in the Nurses’ Health Study, the incidence of invasive SCC climbed from 40 per 100,000 person-years in 1987 to a peak of 120 per 100,000 person-years in 2005 before declining to 80 per 100,000 in 2007. Rates were lower in the Nurses’ Health Study II, a younger cohort. Among men in the Health Professionals Follow-Up Study, the incidence was 80 per 100,000 person-years in 1987, peaking at more than 160 per 100,000 person-years in 2002, and then falling back slightly to 140 per 100,000 person-years in 2007.
Invasive SCC occurred more often in the head and neck region among men, and in the thigh, buttock, legs, and feet region in women.
The significant risk factors for the malignancy, which emerged from a multivariate logistic regression analysis, included a history of painful or blistering sunburns (with an associated 2.2-fold increased risk) and a history of six or more sunburns in childhood. Other independent risk factors for invasive SCC were a family history of melanoma and lighter hair color.
The Nurses’ Health Study, the Nurses’ Health Study II, and the Health Professionals Follow-Up Study were funded by the National Institutes of Health. Dr. Nguyen’s analysis was funded in part by a grant from the Doris Duke Charitable Foundation.
RALEIGH, N.C. – The incidence of invasive squamous cell carcinoma has more than doubled among U.S. nonphysician health professionals in the last 20 years, with marked sex differences evident in body-site distribution.
Dr. Khang Nguyen said his meta-analysis of data drawn from the Nurses' Health Study, the Nurses' Health Study II, and the Health Professionals Follow-Up Study was spurred by the fact that the National Cancer Institute’s SEER (Surveillance, Epidemiology and End Results) database and other national databases that record cancer statistics don’t track squamous cell carcinoma (SCC).
"Much of what we know about the epidemiology of squamous cell carcinoma comes from studies from the 1990s and earlier," noted Dr. Nguyen of Brigham and Women’s Hospital, Boston.
To help rectify this situation, he and his coinvestigators analyzed cases of pathologist-confirmed invasive SCC in the roughly 121,000 female participants in the Nurses’ Health Study, the 117,000 in the Nurses’ Health Study II, and 51,000 men in the Health Professionals Follow-Up Study. There were 1,580 cases in the Nurses’ Health Study, 468 in the Nurses’ Health Study II, and 1,194 in the Health Professionals Follow-Up Study.
Among participants in the Nurses’ Health Study, the incidence of invasive SCC climbed from 40 per 100,000 person-years in 1987 to a peak of 120 per 100,000 person-years in 2005 before declining to 80 per 100,000 in 2007. Rates were lower in the Nurses’ Health Study II, a younger cohort. Among men in the Health Professionals Follow-Up Study, the incidence was 80 per 100,000 person-years in 1987, peaking at more than 160 per 100,000 person-years in 2002, and then falling back slightly to 140 per 100,000 person-years in 2007.
Invasive SCC occurred more often in the head and neck region among men, and in the thigh, buttock, legs, and feet region in women.
The significant risk factors for the malignancy, which emerged from a multivariate logistic regression analysis, included a history of painful or blistering sunburns (with an associated 2.2-fold increased risk) and a history of six or more sunburns in childhood. Other independent risk factors for invasive SCC were a family history of melanoma and lighter hair color.
The Nurses’ Health Study, the Nurses’ Health Study II, and the Health Professionals Follow-Up Study were funded by the National Institutes of Health. Dr. Nguyen’s analysis was funded in part by a grant from the Doris Duke Charitable Foundation.
RALEIGH, N.C. – The incidence of invasive squamous cell carcinoma has more than doubled among U.S. nonphysician health professionals in the last 20 years, with marked sex differences evident in body-site distribution.
Dr. Khang Nguyen said his meta-analysis of data drawn from the Nurses' Health Study, the Nurses' Health Study II, and the Health Professionals Follow-Up Study was spurred by the fact that the National Cancer Institute’s SEER (Surveillance, Epidemiology and End Results) database and other national databases that record cancer statistics don’t track squamous cell carcinoma (SCC).
"Much of what we know about the epidemiology of squamous cell carcinoma comes from studies from the 1990s and earlier," noted Dr. Nguyen of Brigham and Women’s Hospital, Boston.
To help rectify this situation, he and his coinvestigators analyzed cases of pathologist-confirmed invasive SCC in the roughly 121,000 female participants in the Nurses’ Health Study, the 117,000 in the Nurses’ Health Study II, and 51,000 men in the Health Professionals Follow-Up Study. There were 1,580 cases in the Nurses’ Health Study, 468 in the Nurses’ Health Study II, and 1,194 in the Health Professionals Follow-Up Study.
Among participants in the Nurses’ Health Study, the incidence of invasive SCC climbed from 40 per 100,000 person-years in 1987 to a peak of 120 per 100,000 person-years in 2005 before declining to 80 per 100,000 in 2007. Rates were lower in the Nurses’ Health Study II, a younger cohort. Among men in the Health Professionals Follow-Up Study, the incidence was 80 per 100,000 person-years in 1987, peaking at more than 160 per 100,000 person-years in 2002, and then falling back slightly to 140 per 100,000 person-years in 2007.
Invasive SCC occurred more often in the head and neck region among men, and in the thigh, buttock, legs, and feet region in women.
The significant risk factors for the malignancy, which emerged from a multivariate logistic regression analysis, included a history of painful or blistering sunburns (with an associated 2.2-fold increased risk) and a history of six or more sunburns in childhood. Other independent risk factors for invasive SCC were a family history of melanoma and lighter hair color.
The Nurses’ Health Study, the Nurses’ Health Study II, and the Health Professionals Follow-Up Study were funded by the National Institutes of Health. Dr. Nguyen’s analysis was funded in part by a grant from the Doris Duke Charitable Foundation.
AT THE ANNUAL MEETING OF SOCIETY FOR INVESTIGATIVE DERMATOLOGY
Major Finding: Rates of invasive squamous cell carcinoma jumped two- to threefold between 1987 and 2007 in American nonphysician health professionals.
Data Source: This finding came from a meta-analysis of three large, prospective epidemiologic studies: the Nurses’ Health Study, the Nurses’ Health Study II, and the Health Professionals Follow-Up Study.
Disclosures: Dr. Nguyen’s analysis was funded in part by a grant from the Doris Duke Charitable Foundation.
Biochemotherapy Gets Mixed Results in High-Risk Melanoma
CHICAGO – Biochemotherapy is efficacious when used as adjuvant treatment for high-risk melanoma, but not enough to displace the current standard of care, suggests a randomized phase III trial of the Southwest Oncology Group.
The trial, known as S0008, pitted biochemotherapy (chemotherapy plus interleukin-2 and interferon) against high-dose interferon in 402 patients who had undergone wide local excision and lymphadenectomy for stage III melanoma that had features conferring a high risk of recurrence.
Biochemotherapy was associated with a 24% lower risk of relapse, compared with interferon, but there was no significant difference in overall survival, according to trial results reported at the annual meeting of the American Society of Clinical Oncology. Adverse event profiles differed somewhat.
"Biochemotherapy is the first and only therapy to demonstrate a significant improvement in relapse-free survival compared to high-dose interferon in stage III melanoma patients. Biochemotherapy, however, was not associated with an improvement in overall survival," said lead investigator Dr. Lawrence E. Flaherty of the Karmanos Cancer Institute in Detroit.
"Further follow-up will be done on the trial to provide additional insights on longer-term outcomes, but they are not expected to be statistically significant."
"Without an overall survival benefit, biochemotherapy does not replace interferon as the standard of care. However, this may be an attractive alternative to interferon for high-risk melanoma patients," he added, noting biochemotherapy’s advantages of a much shorter treatment duration (2 vs. 12 months) and higher rate of treatment completion.
"Probably more important for our research community is the ability to modify all of the elements that we see here – all of the chemotherapy and biotherapy – as newer elements enter into our field," Dr. Flaherty commented. "This is a template that we can move forward with much more easily and effectively, and efforts of that nature are already ongoing in the stage IV setting."
A session attendee asked, "Would you care to speculate why there was no impact on survival? The disease-free survival impact is very reasonable ... [but] the [overall survival] curves never separated. Any thoughts?"
Dr. Flaherty proposed that it may be related to salvage therapies received after progression, which have improved since the trial. "Certainly, [with] these patients’ having progressed on biochemotherapy, the question would be, what other agents are available then to treat them at progression? I think that that would be less of an issue today with newer agents out, [such as] vemurafenib [and] ipilimumab. It might look differently today than it did with data in the 2000s," he said.
Another attendee questioned whether the short duration of biochemotherapy in the study compromised its efficacy. "Maybe one of the issues here is, by doing 2 months of treatment, you are not really fully exploiting the value of biochemotherapy," he said. "But I know that in community settings, it’s very hard to go beyond this, so it’s a tricky situation."
"We agree," Dr. Flaherty replied. "We had to look for a regimen that could be done throughout the United States with the potential of safety as well as efficacy. And that may be different than what can be done in specialty comprehensive cancer centers."
Discussant Dr. Lynn Schuchter of the Abramson Cancer Center at the University of Pennsylvania, Philadelphia, said, "It’s reasonable to consider biochemotherapy in the adjuvant setting in highly selected patients with melanoma. When I’ve used it, it’s been in young patients with multiple positive lymph nodes."
"I would say that the study results that we heard today do not change the standard of care for our patients with stage III melanoma," she agreed. "And importantly, I would say that further tweaking of the interferon dose, schedule, or route, or type of interferon is not going to move the field forward. ... It’s time that we really look at the new phase III adjuvant trials," such as those involving ipilimumab, vemurafenib, and dabrafenib, and vaccines.
Patients in the S0008 trial were randomized in balanced fashion to biochemotherapy (a 2-month course involving cisplatin, vinblastine, dacarbazine, IL-2, and interferon) or high-dose interferon (4-week induction followed by 48-week maintenance).
Results showed that patients in the biochemotherapy arm were significantly more likely to complete treatment (80% vs. 43%; P less than .001).
With a median 6.25-year follow-up, "the relapse-free survival is strikingly in favor of the biochemotherapy arm" over the high-dose interferon arm, Dr. Flaherty reported (median, 4.3 vs. 1.9 years; hazard ratio, 0.76; P = .03). The 5-year rate of relapse-free survival was 47% and 39%, respectively.
Findings were generally similar across subgroups, although "the more favorable groups appear to be younger individuals, females, those with one to three positive nodes, macroscopic nodes, and those without ulceration," he noted.
There was no significant difference between groups in overall survival. The median duration was 8.4 years with high-dose interferon and was not reached with biochemotherapy. The 5-year rate was 56% in both groups. And findings again were generally similar across patient subgroups.
The two treatments had differing grade 3/4 adverse event profiles: Biochemotherapy was associated with higher rates of gastrointestinal, hematologic, and metabolic toxicities, but lower rates of hepatic and neuropsychiatric toxicities.
Dr. Flaherty disclosed that he is a consultant to and receives honoraria and research funding from Merck and Novartis. Dr. Schuchter disclosed that she receives research funding from Genentech, GlaxoSmithKline, Merck, and Roche.
CHICAGO – Biochemotherapy is efficacious when used as adjuvant treatment for high-risk melanoma, but not enough to displace the current standard of care, suggests a randomized phase III trial of the Southwest Oncology Group.
The trial, known as S0008, pitted biochemotherapy (chemotherapy plus interleukin-2 and interferon) against high-dose interferon in 402 patients who had undergone wide local excision and lymphadenectomy for stage III melanoma that had features conferring a high risk of recurrence.
Biochemotherapy was associated with a 24% lower risk of relapse, compared with interferon, but there was no significant difference in overall survival, according to trial results reported at the annual meeting of the American Society of Clinical Oncology. Adverse event profiles differed somewhat.
"Biochemotherapy is the first and only therapy to demonstrate a significant improvement in relapse-free survival compared to high-dose interferon in stage III melanoma patients. Biochemotherapy, however, was not associated with an improvement in overall survival," said lead investigator Dr. Lawrence E. Flaherty of the Karmanos Cancer Institute in Detroit.
"Further follow-up will be done on the trial to provide additional insights on longer-term outcomes, but they are not expected to be statistically significant."
"Without an overall survival benefit, biochemotherapy does not replace interferon as the standard of care. However, this may be an attractive alternative to interferon for high-risk melanoma patients," he added, noting biochemotherapy’s advantages of a much shorter treatment duration (2 vs. 12 months) and higher rate of treatment completion.
"Probably more important for our research community is the ability to modify all of the elements that we see here – all of the chemotherapy and biotherapy – as newer elements enter into our field," Dr. Flaherty commented. "This is a template that we can move forward with much more easily and effectively, and efforts of that nature are already ongoing in the stage IV setting."
A session attendee asked, "Would you care to speculate why there was no impact on survival? The disease-free survival impact is very reasonable ... [but] the [overall survival] curves never separated. Any thoughts?"
Dr. Flaherty proposed that it may be related to salvage therapies received after progression, which have improved since the trial. "Certainly, [with] these patients’ having progressed on biochemotherapy, the question would be, what other agents are available then to treat them at progression? I think that that would be less of an issue today with newer agents out, [such as] vemurafenib [and] ipilimumab. It might look differently today than it did with data in the 2000s," he said.
Another attendee questioned whether the short duration of biochemotherapy in the study compromised its efficacy. "Maybe one of the issues here is, by doing 2 months of treatment, you are not really fully exploiting the value of biochemotherapy," he said. "But I know that in community settings, it’s very hard to go beyond this, so it’s a tricky situation."
"We agree," Dr. Flaherty replied. "We had to look for a regimen that could be done throughout the United States with the potential of safety as well as efficacy. And that may be different than what can be done in specialty comprehensive cancer centers."
Discussant Dr. Lynn Schuchter of the Abramson Cancer Center at the University of Pennsylvania, Philadelphia, said, "It’s reasonable to consider biochemotherapy in the adjuvant setting in highly selected patients with melanoma. When I’ve used it, it’s been in young patients with multiple positive lymph nodes."
"I would say that the study results that we heard today do not change the standard of care for our patients with stage III melanoma," she agreed. "And importantly, I would say that further tweaking of the interferon dose, schedule, or route, or type of interferon is not going to move the field forward. ... It’s time that we really look at the new phase III adjuvant trials," such as those involving ipilimumab, vemurafenib, and dabrafenib, and vaccines.
Patients in the S0008 trial were randomized in balanced fashion to biochemotherapy (a 2-month course involving cisplatin, vinblastine, dacarbazine, IL-2, and interferon) or high-dose interferon (4-week induction followed by 48-week maintenance).
Results showed that patients in the biochemotherapy arm were significantly more likely to complete treatment (80% vs. 43%; P less than .001).
With a median 6.25-year follow-up, "the relapse-free survival is strikingly in favor of the biochemotherapy arm" over the high-dose interferon arm, Dr. Flaherty reported (median, 4.3 vs. 1.9 years; hazard ratio, 0.76; P = .03). The 5-year rate of relapse-free survival was 47% and 39%, respectively.
Findings were generally similar across subgroups, although "the more favorable groups appear to be younger individuals, females, those with one to three positive nodes, macroscopic nodes, and those without ulceration," he noted.
There was no significant difference between groups in overall survival. The median duration was 8.4 years with high-dose interferon and was not reached with biochemotherapy. The 5-year rate was 56% in both groups. And findings again were generally similar across patient subgroups.
The two treatments had differing grade 3/4 adverse event profiles: Biochemotherapy was associated with higher rates of gastrointestinal, hematologic, and metabolic toxicities, but lower rates of hepatic and neuropsychiatric toxicities.
Dr. Flaherty disclosed that he is a consultant to and receives honoraria and research funding from Merck and Novartis. Dr. Schuchter disclosed that she receives research funding from Genentech, GlaxoSmithKline, Merck, and Roche.
CHICAGO – Biochemotherapy is efficacious when used as adjuvant treatment for high-risk melanoma, but not enough to displace the current standard of care, suggests a randomized phase III trial of the Southwest Oncology Group.
The trial, known as S0008, pitted biochemotherapy (chemotherapy plus interleukin-2 and interferon) against high-dose interferon in 402 patients who had undergone wide local excision and lymphadenectomy for stage III melanoma that had features conferring a high risk of recurrence.
Biochemotherapy was associated with a 24% lower risk of relapse, compared with interferon, but there was no significant difference in overall survival, according to trial results reported at the annual meeting of the American Society of Clinical Oncology. Adverse event profiles differed somewhat.
"Biochemotherapy is the first and only therapy to demonstrate a significant improvement in relapse-free survival compared to high-dose interferon in stage III melanoma patients. Biochemotherapy, however, was not associated with an improvement in overall survival," said lead investigator Dr. Lawrence E. Flaherty of the Karmanos Cancer Institute in Detroit.
"Further follow-up will be done on the trial to provide additional insights on longer-term outcomes, but they are not expected to be statistically significant."
"Without an overall survival benefit, biochemotherapy does not replace interferon as the standard of care. However, this may be an attractive alternative to interferon for high-risk melanoma patients," he added, noting biochemotherapy’s advantages of a much shorter treatment duration (2 vs. 12 months) and higher rate of treatment completion.
"Probably more important for our research community is the ability to modify all of the elements that we see here – all of the chemotherapy and biotherapy – as newer elements enter into our field," Dr. Flaherty commented. "This is a template that we can move forward with much more easily and effectively, and efforts of that nature are already ongoing in the stage IV setting."
A session attendee asked, "Would you care to speculate why there was no impact on survival? The disease-free survival impact is very reasonable ... [but] the [overall survival] curves never separated. Any thoughts?"
Dr. Flaherty proposed that it may be related to salvage therapies received after progression, which have improved since the trial. "Certainly, [with] these patients’ having progressed on biochemotherapy, the question would be, what other agents are available then to treat them at progression? I think that that would be less of an issue today with newer agents out, [such as] vemurafenib [and] ipilimumab. It might look differently today than it did with data in the 2000s," he said.
Another attendee questioned whether the short duration of biochemotherapy in the study compromised its efficacy. "Maybe one of the issues here is, by doing 2 months of treatment, you are not really fully exploiting the value of biochemotherapy," he said. "But I know that in community settings, it’s very hard to go beyond this, so it’s a tricky situation."
"We agree," Dr. Flaherty replied. "We had to look for a regimen that could be done throughout the United States with the potential of safety as well as efficacy. And that may be different than what can be done in specialty comprehensive cancer centers."
Discussant Dr. Lynn Schuchter of the Abramson Cancer Center at the University of Pennsylvania, Philadelphia, said, "It’s reasonable to consider biochemotherapy in the adjuvant setting in highly selected patients with melanoma. When I’ve used it, it’s been in young patients with multiple positive lymph nodes."
"I would say that the study results that we heard today do not change the standard of care for our patients with stage III melanoma," she agreed. "And importantly, I would say that further tweaking of the interferon dose, schedule, or route, or type of interferon is not going to move the field forward. ... It’s time that we really look at the new phase III adjuvant trials," such as those involving ipilimumab, vemurafenib, and dabrafenib, and vaccines.
Patients in the S0008 trial were randomized in balanced fashion to biochemotherapy (a 2-month course involving cisplatin, vinblastine, dacarbazine, IL-2, and interferon) or high-dose interferon (4-week induction followed by 48-week maintenance).
Results showed that patients in the biochemotherapy arm were significantly more likely to complete treatment (80% vs. 43%; P less than .001).
With a median 6.25-year follow-up, "the relapse-free survival is strikingly in favor of the biochemotherapy arm" over the high-dose interferon arm, Dr. Flaherty reported (median, 4.3 vs. 1.9 years; hazard ratio, 0.76; P = .03). The 5-year rate of relapse-free survival was 47% and 39%, respectively.
Findings were generally similar across subgroups, although "the more favorable groups appear to be younger individuals, females, those with one to three positive nodes, macroscopic nodes, and those without ulceration," he noted.
There was no significant difference between groups in overall survival. The median duration was 8.4 years with high-dose interferon and was not reached with biochemotherapy. The 5-year rate was 56% in both groups. And findings again were generally similar across patient subgroups.
The two treatments had differing grade 3/4 adverse event profiles: Biochemotherapy was associated with higher rates of gastrointestinal, hematologic, and metabolic toxicities, but lower rates of hepatic and neuropsychiatric toxicities.
Dr. Flaherty disclosed that he is a consultant to and receives honoraria and research funding from Merck and Novartis. Dr. Schuchter disclosed that she receives research funding from Genentech, GlaxoSmithKline, Merck, and Roche.
AT THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: Compared with high-dose interferon, biochemotherapy significantly improved relapse-free survival (HR, 0.76), but not overall survival.
Data Source: The randomized, phase III SWOG S0008 trial involved 402 patients with high-risk, stage III melanoma.
Disclosures: Dr. Flaherty disclosed that he is a consultant to and receives honoraria and research funding from Merck and Novartis. Dr. Schuchter disclosed that she receives research funding from Genentech, GlaxoSmithKline, Merck, and Roche.
Melanoma In Situ Diagnoses Ramping Up
RALEIGH, N.C. – The ratio of melanoma in situ to invasive melanoma jumped 12-fold among American health professionals during a recent 3-decade period.
Just how much of this steep rise in melanoma in situ diagnoses represents a true increase in the development of in situ lesions as opposed to the product of improved screening techniques or overdiagnosis due to medicolegal pressure remains a matter for conjecture. The new analysis of melanoma trends in the Nurses' Health Study and the Health Professionals Follow-Up Study provides evidence suggesting that all three factors may be involved, according to Erin X. Wei of Massachusetts General Hospital, Boston.
Her analysis included roughly 121,000 female nurses followed prospectively in the Nurses’ Health Study since 1976 and 51,000 men followed in the Health Professionals Follow-Up Study since 1986. During the follow-up period, new cases of invasive melanoma increased dramatically, but they were far outpaced by the rise in cases of melanoma in situ, she said at the annual meeting of the Society for Investigative Dermatology.
For example, while the ratio of in situ to invasive melanoma among women was 0.09:1 in 1977, by 2002 it had reached 1:1.
Support for the notion that the rise in melanoma in situ is due in part to overdiagnosis in response to medicolegal concerns comes from the finding that the bulk of the increase involves lesions reported in this study as having Not-Otherwise-Specified pathology. Lentigo maligna cases have remained relatively flat over time, she noted.
Also, in this large patient population, as well as in studies from elsewhere around the world, the increase in melanoma in situ has not been accompanied by a shift in the pattern of invasive melanoma thickness or by a reduction in melanoma mortality, as would be expected in response to a major increase in detection of invasive melanoma precursor lesions.
But perhaps in situ melanomas are often not precursors to invasive melanoma. If they were precursors, one would expect them to be diagnosed at a younger average age than invasive melanoma. Such was not the case in this analysis. Indeed, while roughly 40% of invasive melanomas were diagnosed prior to age 60, that was true for only 20% of melanoma in situ. Nurses diagnosed with melanoma in situ were an average of 6 years older than those diagnosed with invasive melanoma; among men, the difference was 2 years.
Melanoma in situ and invasive melanomas tended to differ in their anatomic distribution. Invasive melanomas were far less likely than in situ lesions to occur on the head and neck, and much more likely to occur on the lower extremities. This suggests some in situ melanomas are indolent and do not become malignant over time, Ms. Wei continued.
In support of the existence of a true biologic increase in the incidence of melanoma in situ, Ms. Wei and her coworkers found that older individuals were two- to threefold more likely to develop their lesions on chronically sun-exposed areas than on intermittently sun-exposed areas.
An intriguing gender difference in the anatomic distribution of melanoma in situ was evident. Among men, more than 98% of all melanoma in situ were found on the upper half of the body. In women, the lesions were more evenly distributed anatomically, with 27% of in situ lesions being found on the lower extremities.
The Nurses’ Health Study and Health Professionals Follow-up Study are funded by the National Institutes of Health. Ms. Wei reported having no financial conflicts.
RALEIGH, N.C. – The ratio of melanoma in situ to invasive melanoma jumped 12-fold among American health professionals during a recent 3-decade period.
Just how much of this steep rise in melanoma in situ diagnoses represents a true increase in the development of in situ lesions as opposed to the product of improved screening techniques or overdiagnosis due to medicolegal pressure remains a matter for conjecture. The new analysis of melanoma trends in the Nurses' Health Study and the Health Professionals Follow-Up Study provides evidence suggesting that all three factors may be involved, according to Erin X. Wei of Massachusetts General Hospital, Boston.
Her analysis included roughly 121,000 female nurses followed prospectively in the Nurses’ Health Study since 1976 and 51,000 men followed in the Health Professionals Follow-Up Study since 1986. During the follow-up period, new cases of invasive melanoma increased dramatically, but they were far outpaced by the rise in cases of melanoma in situ, she said at the annual meeting of the Society for Investigative Dermatology.
For example, while the ratio of in situ to invasive melanoma among women was 0.09:1 in 1977, by 2002 it had reached 1:1.
Support for the notion that the rise in melanoma in situ is due in part to overdiagnosis in response to medicolegal concerns comes from the finding that the bulk of the increase involves lesions reported in this study as having Not-Otherwise-Specified pathology. Lentigo maligna cases have remained relatively flat over time, she noted.
Also, in this large patient population, as well as in studies from elsewhere around the world, the increase in melanoma in situ has not been accompanied by a shift in the pattern of invasive melanoma thickness or by a reduction in melanoma mortality, as would be expected in response to a major increase in detection of invasive melanoma precursor lesions.
But perhaps in situ melanomas are often not precursors to invasive melanoma. If they were precursors, one would expect them to be diagnosed at a younger average age than invasive melanoma. Such was not the case in this analysis. Indeed, while roughly 40% of invasive melanomas were diagnosed prior to age 60, that was true for only 20% of melanoma in situ. Nurses diagnosed with melanoma in situ were an average of 6 years older than those diagnosed with invasive melanoma; among men, the difference was 2 years.
Melanoma in situ and invasive melanomas tended to differ in their anatomic distribution. Invasive melanomas were far less likely than in situ lesions to occur on the head and neck, and much more likely to occur on the lower extremities. This suggests some in situ melanomas are indolent and do not become malignant over time, Ms. Wei continued.
In support of the existence of a true biologic increase in the incidence of melanoma in situ, Ms. Wei and her coworkers found that older individuals were two- to threefold more likely to develop their lesions on chronically sun-exposed areas than on intermittently sun-exposed areas.
An intriguing gender difference in the anatomic distribution of melanoma in situ was evident. Among men, more than 98% of all melanoma in situ were found on the upper half of the body. In women, the lesions were more evenly distributed anatomically, with 27% of in situ lesions being found on the lower extremities.
The Nurses’ Health Study and Health Professionals Follow-up Study are funded by the National Institutes of Health. Ms. Wei reported having no financial conflicts.
RALEIGH, N.C. – The ratio of melanoma in situ to invasive melanoma jumped 12-fold among American health professionals during a recent 3-decade period.
Just how much of this steep rise in melanoma in situ diagnoses represents a true increase in the development of in situ lesions as opposed to the product of improved screening techniques or overdiagnosis due to medicolegal pressure remains a matter for conjecture. The new analysis of melanoma trends in the Nurses' Health Study and the Health Professionals Follow-Up Study provides evidence suggesting that all three factors may be involved, according to Erin X. Wei of Massachusetts General Hospital, Boston.
Her analysis included roughly 121,000 female nurses followed prospectively in the Nurses’ Health Study since 1976 and 51,000 men followed in the Health Professionals Follow-Up Study since 1986. During the follow-up period, new cases of invasive melanoma increased dramatically, but they were far outpaced by the rise in cases of melanoma in situ, she said at the annual meeting of the Society for Investigative Dermatology.
For example, while the ratio of in situ to invasive melanoma among women was 0.09:1 in 1977, by 2002 it had reached 1:1.
Support for the notion that the rise in melanoma in situ is due in part to overdiagnosis in response to medicolegal concerns comes from the finding that the bulk of the increase involves lesions reported in this study as having Not-Otherwise-Specified pathology. Lentigo maligna cases have remained relatively flat over time, she noted.
Also, in this large patient population, as well as in studies from elsewhere around the world, the increase in melanoma in situ has not been accompanied by a shift in the pattern of invasive melanoma thickness or by a reduction in melanoma mortality, as would be expected in response to a major increase in detection of invasive melanoma precursor lesions.
But perhaps in situ melanomas are often not precursors to invasive melanoma. If they were precursors, one would expect them to be diagnosed at a younger average age than invasive melanoma. Such was not the case in this analysis. Indeed, while roughly 40% of invasive melanomas were diagnosed prior to age 60, that was true for only 20% of melanoma in situ. Nurses diagnosed with melanoma in situ were an average of 6 years older than those diagnosed with invasive melanoma; among men, the difference was 2 years.
Melanoma in situ and invasive melanomas tended to differ in their anatomic distribution. Invasive melanomas were far less likely than in situ lesions to occur on the head and neck, and much more likely to occur on the lower extremities. This suggests some in situ melanomas are indolent and do not become malignant over time, Ms. Wei continued.
In support of the existence of a true biologic increase in the incidence of melanoma in situ, Ms. Wei and her coworkers found that older individuals were two- to threefold more likely to develop their lesions on chronically sun-exposed areas than on intermittently sun-exposed areas.
An intriguing gender difference in the anatomic distribution of melanoma in situ was evident. Among men, more than 98% of all melanoma in situ were found on the upper half of the body. In women, the lesions were more evenly distributed anatomically, with 27% of in situ lesions being found on the lower extremities.
The Nurses’ Health Study and Health Professionals Follow-up Study are funded by the National Institutes of Health. Ms. Wei reported having no financial conflicts.
FROM THE ANNUAL MEETING OF THE SOCIETY FOR INVESTIGATIVE DERMATOLOGY
Major Finding: Roughly 40% of invasive melanomas in a large prospectively followed population were diagnosed prior to age 60, compared with 20% of in situ melanomas. This casts doubt on the notion that melanoma in situ is necessarily a precursor lesion for invasive melanoma.
Data Source: This was an analysis of data from the prospective 121,000-subject Nurses’ Health Study and 51,000-man Health Professionals Follow-Up Study.
Disclosures: The Nurses’ Health Study and Health Professionals Follow-Up Study are funded by the National Institutes of Health. Ms. Wei reported having no financial conflicts.