User login
No OS benefit with gefitinib vs. chemo for EGFR+ NSCLC
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
FROM ASCO 2020
Former smokers using e-cigarettes at risk for cigarette smoking relapse
The use of , results from a large longitudinal cohort study demonstrated.
“For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open (2020 Jun 5. doi: 10.1001/jamanetworkopen.2020.4813).
Dr. Everard, of the National Institute on Drug Abuse, and colleagues based their comments on results from a survey of adult former smokers who participated in Waves 1-4 of the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). They limited the analysis to 2,273 former cigarette smokers who self-reported reported no tobacco product use at Wave 1, and categorized them as recent former smokers (defined as having last smoked within the past 12 previous months) or as long-term former smokers (defined as having last smoked for longer ago than in the previous 12 months). The main outcome of interest was the self-reported current use of cigarettes at follow-up interviews, which was defined as every day or some days. Electronic nicotine delivery systems (ENDS) comprised e-cigarettes, e-cigars, e-pipes, and e-hookahs. Other tobacco products included cigars, pipe tobacco, hookahs, snus tobacco, other smokeless tobacco, and dissolvable tobacco.
Of the 2,273 adult former smokers, 52% were women, 60% were older than age 50, and 80% were non-Hispanic white. Adjusted hazard ratio (AHR) analysis revealed that the use of ENDS was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was associated with significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82).
The authors acknowledged certain limitations of the study, including the fact that it did not assess different ENDS devices, different e-liquid nicotine levels, or frequency of ENDS use and their associations with cigarette smoking relapse. It also did not explore the mechanism by which ENDS use may lead to reestablishing or reinforcing nicotine-seeking behavior among former cigarette users. “Determining pharmacologic, behavioral, or some other explanation for these findings may require laboratory-based research,” they wrote.
The PATH Study is supported with federal funds from the National Institute on Drug Abuse, the National Institutes of Health, and the Food and Drug Administration and Department of Health and Human Services under a contract to Westat. One of the study authors, Wilson M. Compton, MD, reported having long-term stock holdings in General Electric, 3M, and Pfizer. The other authors reported having no financial disclosures.
The use of , results from a large longitudinal cohort study demonstrated.
“For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open (2020 Jun 5. doi: 10.1001/jamanetworkopen.2020.4813).
Dr. Everard, of the National Institute on Drug Abuse, and colleagues based their comments on results from a survey of adult former smokers who participated in Waves 1-4 of the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). They limited the analysis to 2,273 former cigarette smokers who self-reported reported no tobacco product use at Wave 1, and categorized them as recent former smokers (defined as having last smoked within the past 12 previous months) or as long-term former smokers (defined as having last smoked for longer ago than in the previous 12 months). The main outcome of interest was the self-reported current use of cigarettes at follow-up interviews, which was defined as every day or some days. Electronic nicotine delivery systems (ENDS) comprised e-cigarettes, e-cigars, e-pipes, and e-hookahs. Other tobacco products included cigars, pipe tobacco, hookahs, snus tobacco, other smokeless tobacco, and dissolvable tobacco.
Of the 2,273 adult former smokers, 52% were women, 60% were older than age 50, and 80% were non-Hispanic white. Adjusted hazard ratio (AHR) analysis revealed that the use of ENDS was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was associated with significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82).
The authors acknowledged certain limitations of the study, including the fact that it did not assess different ENDS devices, different e-liquid nicotine levels, or frequency of ENDS use and their associations with cigarette smoking relapse. It also did not explore the mechanism by which ENDS use may lead to reestablishing or reinforcing nicotine-seeking behavior among former cigarette users. “Determining pharmacologic, behavioral, or some other explanation for these findings may require laboratory-based research,” they wrote.
The PATH Study is supported with federal funds from the National Institute on Drug Abuse, the National Institutes of Health, and the Food and Drug Administration and Department of Health and Human Services under a contract to Westat. One of the study authors, Wilson M. Compton, MD, reported having long-term stock holdings in General Electric, 3M, and Pfizer. The other authors reported having no financial disclosures.
The use of , results from a large longitudinal cohort study demonstrated.
“For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open (2020 Jun 5. doi: 10.1001/jamanetworkopen.2020.4813).
Dr. Everard, of the National Institute on Drug Abuse, and colleagues based their comments on results from a survey of adult former smokers who participated in Waves 1-4 of the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). They limited the analysis to 2,273 former cigarette smokers who self-reported reported no tobacco product use at Wave 1, and categorized them as recent former smokers (defined as having last smoked within the past 12 previous months) or as long-term former smokers (defined as having last smoked for longer ago than in the previous 12 months). The main outcome of interest was the self-reported current use of cigarettes at follow-up interviews, which was defined as every day or some days. Electronic nicotine delivery systems (ENDS) comprised e-cigarettes, e-cigars, e-pipes, and e-hookahs. Other tobacco products included cigars, pipe tobacco, hookahs, snus tobacco, other smokeless tobacco, and dissolvable tobacco.
Of the 2,273 adult former smokers, 52% were women, 60% were older than age 50, and 80% were non-Hispanic white. Adjusted hazard ratio (AHR) analysis revealed that the use of ENDS was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was associated with significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82).
The authors acknowledged certain limitations of the study, including the fact that it did not assess different ENDS devices, different e-liquid nicotine levels, or frequency of ENDS use and their associations with cigarette smoking relapse. It also did not explore the mechanism by which ENDS use may lead to reestablishing or reinforcing nicotine-seeking behavior among former cigarette users. “Determining pharmacologic, behavioral, or some other explanation for these findings may require laboratory-based research,” they wrote.
The PATH Study is supported with federal funds from the National Institute on Drug Abuse, the National Institutes of Health, and the Food and Drug Administration and Department of Health and Human Services under a contract to Westat. One of the study authors, Wilson M. Compton, MD, reported having long-term stock holdings in General Electric, 3M, and Pfizer. The other authors reported having no financial disclosures.
FROM JAMA NETWORK OPEN
Pulmonary Neuroendocrine Tumor Presenting as a Left Pleural Effusion
Neuroendocrine tumors (NETs) account for about 0.5% of all newly diagnosed malignancies.1 Pulmonary NETs are rare, accounting for 1 to 2% of all invasive lung malignancies and involve about 20 to 25% of primary lung malignancies. 2,3 Their prevalence has increased by an estimated 6% per year over the past 30 years.2 Nonetheless, the time of diagnosis is frequently delayed because of nonspecific symptoms that may imitate other pulmonary conditions.
In the normal pleural space, there is a steady state in which there is a roughly equal rate of fluid formation and absorption. Any disequilibrium may produce a pleural effusion. Pleural fluids can be transudates or exudates. Transudates result from imbalances in hydrostatic and oncotic pressures in the pleural space. Exudates result primarily from pleural and/or lung inflammation or from impaired lymphatic drainage of the pleural space. Clinical manifestations include cough, wheezing, recurrent pneumonia, hemoptysis and pleural effusions. We present a case of a man who developed a large left pleural effusion with a pathology report suggesting a pulmonary NET as the etiology. Being aware of this rare entity may help improve prognosis by making an earlier diagnosis and starting treatment sooner.
Case Presentation
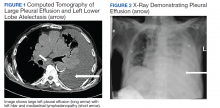
A 90-year-old man with a medical history of arterial hypertension, hyperlipidemia, type 2 diabetes mellitus, coronary artery disease, and vascular dementia presented to the emergency department with hypoactivity, poor appetite, productive cough, and shortness of breath. The patient was a former smoker (unknown pack-years) who quit smoking cigarettes 7 years prior. Vital signs showed sinus tachycardia and peripheral oxygen saturation of 90% at room air. The initial physical examination was remarkable for decreased breath sounds and crackles at the left lung base. Laboratory findings showed leukocytosis with neutrophilia and chronic normocytic anemia. Chest computed tomography (CT) showed a large left-sided pleural effusion occupying most of the left hemithorax with adjacent atelectatic lung, enlarged pretracheal, subcarinal, and left perihilar lymph nodes (Figure 1).
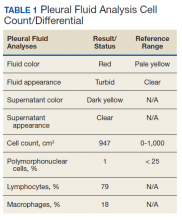
The patient was admitted to the internal medicine ward with the diagnosis of left pneumonic process and started on IV levofloxacin. However, despite 7 days of antibiotic therapy, the patient’s respiratory symptoms worsened. This clinical deterioration prompted pulmonary service consultation. Chest radiography demonstrated an enlarging left pleural effusion (Figure 2). A thoracentesis drained 1.2 L of serosanguineous pleural fluid. Pleural fluid analysis showed a cell count of 947/cm3 with 79% of lymphocytes, total protein 3.8 g/dL, lactic dehydrogenase (LDH) level 607 U/L, and glucose level 109 mg/dL. Serum total protein was 6.62 g/dL, LDH 666 U/L and glucose 92 mg/dL (Tables 1 and 2). Alanine transaminase (ALT) and aspartate aminotransferase (AST) were 11 U/L and 21 U/L, respectively. Using Light criteria, the pleural:serum protein ratio was 0.57, the pleural:serum LDH ratio was 0.91, and the pleural LDH was more than two-thirds of the serum LDH. These calculations were consistent with an exudative effusion. An infectious disease workup, including blood and pleural fluid cultures, was negative.
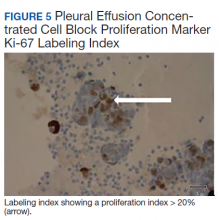
The pleural fluid concentrated cell block hematoxylin and eosin (H&E) staining showed chromatin, prominent nucleoli, and nuclear molding, which was compatible with high-grade lung NET (Figure 3). The cell block immunohistochemistry (IHC) was positive for synaptophysin, chromogranin A, and neuron specific enolase (NSE) also consistent with a high-grade pulmonary NET (Figure 4). The proliferation marker protein Ki-67 labeling index (LI) showed a proliferation index > 20% (Figure 5). The patient did not have decision-making capacity given vascular dementia. Multiple attempts to contact the next of kin or family members were unsuccessful. Risks vs benefits were evaluated, and given the patient’s advanced age and multiple comorbidities, a conservative management approach under palliative care was chosen. For this reason, further genomic studies were not done.
Discussion
NETs are a group of neoplasms that differ in site, amount of cell propagation, and clinical manifestations.4 These tumors are rare with an estimated incidence of 25 to 50 per 100,000.4 The most commonly affected organ systems are the gastroenteropancreatic and the bronchopulmonary tracts, accounting for 60% and 25% of the tumors, respectively.4 The incidence is increasing over the past years in part because of novel diagnostic techniques.
The average age of diagnosis is between the fourth and sixth decades, affecting more women than men.5 Smoking has been identified as a possible culprit for the development of these neoplasms; nonetheless, the association is still not clear.4 For example, poorly differentiated pulmonary NETs have a strong association with smoking but not well-differentiated pulmonary NETs.2
Patients typically present with cough, wheezing, hemoptysis, and recurrent pneumonias, which are in part a consequence of obstruction caused by the mass.2 Sometimes, obstruction may yield persistent pleural effusions. Hemoptysis may be seen secondary to the vascularity of pulmonary NETs.
The diagnosis is often delayed because patients are frequently treated for infection before being diagnosed with the malignancy, such as in our case. Radiologic image findings include round opacities, central masses, and atelectasis. Pulmonary NETs are frequently found incidentally as solitary lung nodules. The CT scan is the most common diagnostic modality and can provide information about the borders of the tumor, the location and surrounding structures, including the presence of atelectasis.5 Pulmonary NETs are usually centrally located in an accessible region for lung biopsy. In cases where the mass is not easily reachable, thoracentesis may provide the only available specimen.
The 2015 World Health Organization classification has identified 4 histologic types of pulmonary NETs, namely, typical carcinoid (TC), atypical carcinoid (AC), large cell neuroendocrine carcinoma (LCNEC) and small cell lung carcinoma (SCLC).6 The low-grade pulmonary NET, the typical carcinoid, is slow growing and has lower rates of metastasis. The intermediate-grade NET, the atypical carcinoid, is more aggressive. The highgrade NETs, the LCNEC and the SCLC, are aggressive and spread quickly to other places.6 Consequently, LCNEC and SCLC have higher mortalities with a 5-year survival, ranging from 13 to 57% and 5%, respectively.7
Tumors may be histomorphologically classified by H&E staining. The main characteristics that differentiate the low- and high-grade NETs are the presence of necrosis and the mitotic rate. Both categories form neuropeptides and have dense granular cores when seen with an electron microscopy.6 The TC and AC have welldefined, organized histologic patterns, no necrosis, and scarce mitosis. On the other hand, the LCNEC and SCLC are poorly differentiated tumors with necrosis, atypia, and mitosis.6 LCNEC can be separated from SCLC and other tumors by IHC staining, whereas SCLC is primarily distinguished by morphology.
If the biopsy sample size is small, then IHC morphology and markers are helpful for subclassification.8 IHC is used to discern between neuroendocrine (NE) vs non-NE. The evaluation of pleural fluid includes preparation of cell blocks. Cell block staining is deemed better for IHC because it mimics a small biopsy that enables superior stains.9 The need for a pleural biopsy in cases where the cytology is negative depends on treatment aims, the kind of tumor, and the presence of metastasis.10 In almost 80% of cases, pleural biopsy and cytology are the only specimens obtained for analysis.Therefore, identification of these markers is practical for diagnosis.10 For this reason, pleural effusion samples are appropriate options to lung biopsy for molecular studies.10
Ki-67 LI in samples has the highest specificity and sensitivity for low-tointermediate- grade vs high-grade tumors. It is being used for guiding clinical and treatment decisions.6 In SCLC, the Ki-67 LI is not necessary for diagnosis but will be about 80%.11 The tumor cells will show epithelial characteristics with positive cytokeratin AE1/AE3 and monoclonal antibody CAM5.2 and neuroendocrine markers, including NCAM/CD56, chromogranin A, and synaptophysin.11
Thyroid transcription factor-1 (TTF- 1) is positive in most cases. In LCNEC, the Ki-67 LI is between 40% and 80%. NCAM/ CD56, chromogranin A, and synaptophysin are present in 92 to 100%, 80 to 85%, and 50 to 60%, respectively.11 TTF-1 is identified in half of the tumors. All these tumors express pancytokeratin (AE1/AE3), cytokeratin 7 or low-molecular-weight cytokeratin. Likewise, the carcinoids will show markers, such as chromogranin A, synaptophysin, CD56, and epithelial markers like pancytokeratin.11 However, the high-molecular-weight cytokeratin and TTF-1 are negative. Furthermore, NSE is considered a good tumor marker in the diagnosis and prognosis of SCLC. NSE also has been reported in NSCLC. The level of NSE correlates with tumor burden, number of metastatic sites, and response to treatment. 12 A potentially useful marker is the insulinoma-associated protein 1, which is a nuclear determinant of NE differentiation that stains all types of pulmonary NETs irrespective of the histology but does not stain adenocarcinoma or squamous cell carcinoma (SCC).6
Recently, genomic studies have identified gene alterations that have become standard of care for diagnosis and targeted therapies.8 For example, epidermal growth factor receptor (EGFR) and echinoderm microtubule- associated proteinlike 4, and anaplastic lymphoma kinase (EML4-ALK) mutations have been found in about 25% of lung adenocarcinomas. 8 Other abnormalities in LKB1/STK11, NF1, CDKN2A, SMARCA4 and KEAP1, KRAS, MET, ROS1, and RET have also been identified.8 On the other hand, SCC rarely have derangements in EGFR and EML4-ALK, but do show changes in RTKs, DDR2M, FGGRs, among others.8 In TC and AC, observed molecular alterations include MEN1 mutations, mTOR, and SSTRs pathway activation, and GC/ CEACAM1 and CD44/OTP expression.13 LCNEC and SCLC have shown TP53 and RB1 mutations and CDX2/VIL1/BAI3 expression. DLL3 expression and MET mutations may be present in SCLC.13 Last, chromatin remodeling gene mutations have been identified in all these lung NET types.13
Furthermore, neuropeptides and neuroamines may be measured in the blood and urine.14 Pulmonary NETs may be functional and secrete these substances, leading to systemic symptoms based on the released molecules.15 However, pulmonary NETs produce less serotonin than gastrointestinal NETs; therefore, carcinoid syndrome is less frequent in pulmonary NETs.16 Liver metastasis is often present when it occurs.5 Other possible clinical features include Cushing syndrome and acromegaly depending on the secreted hormones.5
In a recent metanalysis, serum LDH has been found to have a prognostic role in Ewing sarcoma, urologic cancers, malignant mesothelioma, among others.17 It demonstrated that a higher LDH concentration is associated with worse survival in patients with lung cancer.17 Serum LDH is an enzyme that catalyzes the reaction between lactic acid and pyruvic acid that typically takes place in anaerobic conditions.17 LDH levels are elevated in malignancies because tumors have an anaerobic environment. Elevated LDH levels correlate with the anaerobic metabolism in the tumor. Other studies also have noted that patients with high metastatic score have higher LDH levels.17 Therefore, LDH may reflect tumor extension.
In addition, other techniques, such as somatostatin- receptor imaging are specifically beneficial in tumors that express the somatostatin receptor.16 For this reason, this type of study is typically indicated in patients with known metastasis, not in patients with low-grade tumors. Abdominal CT scans are done because the liver is a common site for metastasis.
Our case report demonstrates how biomarkers help diagnose these potentially aggressive and life-threatening tumors that may present as a common condition such as a pleural effusion. Using a less invasive and quicker approach with thoracentesis rather than with lung biopsies is a diagnostic tool in this entity. IHC in cell blocks is a reasonable diagnostic method especially in patients in whom performing a lung biopsy is difficult.
Conclusions
The presence of a symptomatic and recurrent unilateral pleural effusion must urge physicians to consider thoracentesis with mindful use of biomarkers not only for therapeutic purposes, but also for diagnosis of a variety of etiologies, both benign and malignant.
1. Oronsky B, Ma PC, Morgensztern D, Carter CA. Nothing but NET: a review of neuroendocrine tumors and carcinomas. Neoplasia. 2017;19(12):991-1002. doi: 10.1016/j.neo.2017.09.002
2. Hendifar AE, Marchevsky AM, Tuli R. Neuroendocrine tumors of the lung: current challenges and advances in the diagnosis and management of well-differentiated disease. J Thorac Oncol. 2017;12(3):425-436. doi: 10.1016/j.jtho.2016.11.2222
3. Fisseler-Eckhoff A, Demes M. Neuroendocrine tumors of the lung. Cancers (Basel). 2012;4(3):777-798. doi: 10.3390/cancers4030777
4. Mandegaran R, David S, Screaton N. Cardiothoracic manifestations of neuroendocrine tumours. Br J Radiol. 2016;89(1060). doi: 10.1259/bjr.20150787
5. Caplin ME, Baudin E, Ferolla P, et al; ENETS consensus conference participants. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620. doi: 10.1093/annonc/mdv041
6. Pelosi G, Sonzogni A, Harari S, et al. Classification of pulmonary neuroendocrine tumors: new insights. Transl Lung Cancer Res. 2017;6(5):513-529. doi: 10.21037/tlcr.2017.09.04
7. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394.
8. Osmani L, Askin F, Gabrielson E, Li QK. Current WHO guidelines and the critical role of immunohistochemical markers in the subclassification of non-small cell lung carcinoma (NSCLC): moving from targeted therapy to immunotherapy. Semin Cancer Biol. 2018;52(pt 1):103-109. doi: 10.1016/j.semcancer.2017.11.019
9. Kaur G, Nijhawan R, Gupta N, Singh N, Rajwanshi A. Pleural fluid cytology samples in cases of suspected lung cancer: an experience from a tertiary care centre. Diagn Cytopathol. 2017;45(3):195-201.
10. Porcel JM. Biomarkers in the diagnosis of pleural diseases: a 2018 update. Ther Adv Respir Dis. 2018;12. doi: 10.1177/1753466618808660
11. Kim JY, Hong SM, Ro JY. Recent updates on grading and classification of neuroendocrine tumors. Ann Diagn Pathol. 2017;29:11-16. doi: 10.1016/j.anndiagpath.2017.04.005
12. Isgrò MA, Bottoni P, Scatena R. Neuron-specific enolase as a biomarker: biochemical and clinical aspects. Adv Exp Med Biol. 2015;867:125-143. doi: 10.1007/978-94-017-7215-0_9
13. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394
14. Eriksson B, Oberg K, Stridsberg M. Tumor markers in neuroendocrine tumors. Digestion. 2000;62(suppl 1):33-38.
15. Melosky B. Low grade neuroendocrine tumors of the lung. Front Oncol. 2017;7:119. doi: 10.3389/fonc.2017.00119
16. Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2001;113(1):5-21. https://doi.org/10.1002/cncr.23542
17. Deng T, Zhang J, Meng Y, Zhou Y, Li W. Higher pretreatment lactate dehydrogenase concentration predicts worse overall survival in patients with lung cancer. Medicine (Baltimore). 2018;97(38):e12524
Neuroendocrine tumors (NETs) account for about 0.5% of all newly diagnosed malignancies.1 Pulmonary NETs are rare, accounting for 1 to 2% of all invasive lung malignancies and involve about 20 to 25% of primary lung malignancies. 2,3 Their prevalence has increased by an estimated 6% per year over the past 30 years.2 Nonetheless, the time of diagnosis is frequently delayed because of nonspecific symptoms that may imitate other pulmonary conditions.
In the normal pleural space, there is a steady state in which there is a roughly equal rate of fluid formation and absorption. Any disequilibrium may produce a pleural effusion. Pleural fluids can be transudates or exudates. Transudates result from imbalances in hydrostatic and oncotic pressures in the pleural space. Exudates result primarily from pleural and/or lung inflammation or from impaired lymphatic drainage of the pleural space. Clinical manifestations include cough, wheezing, recurrent pneumonia, hemoptysis and pleural effusions. We present a case of a man who developed a large left pleural effusion with a pathology report suggesting a pulmonary NET as the etiology. Being aware of this rare entity may help improve prognosis by making an earlier diagnosis and starting treatment sooner.
Case Presentation
A 90-year-old man with a medical history of arterial hypertension, hyperlipidemia, type 2 diabetes mellitus, coronary artery disease, and vascular dementia presented to the emergency department with hypoactivity, poor appetite, productive cough, and shortness of breath. The patient was a former smoker (unknown pack-years) who quit smoking cigarettes 7 years prior. Vital signs showed sinus tachycardia and peripheral oxygen saturation of 90% at room air. The initial physical examination was remarkable for decreased breath sounds and crackles at the left lung base. Laboratory findings showed leukocytosis with neutrophilia and chronic normocytic anemia. Chest computed tomography (CT) showed a large left-sided pleural effusion occupying most of the left hemithorax with adjacent atelectatic lung, enlarged pretracheal, subcarinal, and left perihilar lymph nodes (Figure 1).
The patient was admitted to the internal medicine ward with the diagnosis of left pneumonic process and started on IV levofloxacin. However, despite 7 days of antibiotic therapy, the patient’s respiratory symptoms worsened. This clinical deterioration prompted pulmonary service consultation. Chest radiography demonstrated an enlarging left pleural effusion (Figure 2). A thoracentesis drained 1.2 L of serosanguineous pleural fluid. Pleural fluid analysis showed a cell count of 947/cm3 with 79% of lymphocytes, total protein 3.8 g/dL, lactic dehydrogenase (LDH) level 607 U/L, and glucose level 109 mg/dL. Serum total protein was 6.62 g/dL, LDH 666 U/L and glucose 92 mg/dL (Tables 1 and 2). Alanine transaminase (ALT) and aspartate aminotransferase (AST) were 11 U/L and 21 U/L, respectively. Using Light criteria, the pleural:serum protein ratio was 0.57, the pleural:serum LDH ratio was 0.91, and the pleural LDH was more than two-thirds of the serum LDH. These calculations were consistent with an exudative effusion. An infectious disease workup, including blood and pleural fluid cultures, was negative.
The pleural fluid concentrated cell block hematoxylin and eosin (H&E) staining showed chromatin, prominent nucleoli, and nuclear molding, which was compatible with high-grade lung NET (Figure 3). The cell block immunohistochemistry (IHC) was positive for synaptophysin, chromogranin A, and neuron specific enolase (NSE) also consistent with a high-grade pulmonary NET (Figure 4). The proliferation marker protein Ki-67 labeling index (LI) showed a proliferation index > 20% (Figure 5). The patient did not have decision-making capacity given vascular dementia. Multiple attempts to contact the next of kin or family members were unsuccessful. Risks vs benefits were evaluated, and given the patient’s advanced age and multiple comorbidities, a conservative management approach under palliative care was chosen. For this reason, further genomic studies were not done.
Discussion
NETs are a group of neoplasms that differ in site, amount of cell propagation, and clinical manifestations.4 These tumors are rare with an estimated incidence of 25 to 50 per 100,000.4 The most commonly affected organ systems are the gastroenteropancreatic and the bronchopulmonary tracts, accounting for 60% and 25% of the tumors, respectively.4 The incidence is increasing over the past years in part because of novel diagnostic techniques.
The average age of diagnosis is between the fourth and sixth decades, affecting more women than men.5 Smoking has been identified as a possible culprit for the development of these neoplasms; nonetheless, the association is still not clear.4 For example, poorly differentiated pulmonary NETs have a strong association with smoking but not well-differentiated pulmonary NETs.2
Patients typically present with cough, wheezing, hemoptysis, and recurrent pneumonias, which are in part a consequence of obstruction caused by the mass.2 Sometimes, obstruction may yield persistent pleural effusions. Hemoptysis may be seen secondary to the vascularity of pulmonary NETs.
The diagnosis is often delayed because patients are frequently treated for infection before being diagnosed with the malignancy, such as in our case. Radiologic image findings include round opacities, central masses, and atelectasis. Pulmonary NETs are frequently found incidentally as solitary lung nodules. The CT scan is the most common diagnostic modality and can provide information about the borders of the tumor, the location and surrounding structures, including the presence of atelectasis.5 Pulmonary NETs are usually centrally located in an accessible region for lung biopsy. In cases where the mass is not easily reachable, thoracentesis may provide the only available specimen.
The 2015 World Health Organization classification has identified 4 histologic types of pulmonary NETs, namely, typical carcinoid (TC), atypical carcinoid (AC), large cell neuroendocrine carcinoma (LCNEC) and small cell lung carcinoma (SCLC).6 The low-grade pulmonary NET, the typical carcinoid, is slow growing and has lower rates of metastasis. The intermediate-grade NET, the atypical carcinoid, is more aggressive. The highgrade NETs, the LCNEC and the SCLC, are aggressive and spread quickly to other places.6 Consequently, LCNEC and SCLC have higher mortalities with a 5-year survival, ranging from 13 to 57% and 5%, respectively.7
Tumors may be histomorphologically classified by H&E staining. The main characteristics that differentiate the low- and high-grade NETs are the presence of necrosis and the mitotic rate. Both categories form neuropeptides and have dense granular cores when seen with an electron microscopy.6 The TC and AC have welldefined, organized histologic patterns, no necrosis, and scarce mitosis. On the other hand, the LCNEC and SCLC are poorly differentiated tumors with necrosis, atypia, and mitosis.6 LCNEC can be separated from SCLC and other tumors by IHC staining, whereas SCLC is primarily distinguished by morphology.
If the biopsy sample size is small, then IHC morphology and markers are helpful for subclassification.8 IHC is used to discern between neuroendocrine (NE) vs non-NE. The evaluation of pleural fluid includes preparation of cell blocks. Cell block staining is deemed better for IHC because it mimics a small biopsy that enables superior stains.9 The need for a pleural biopsy in cases where the cytology is negative depends on treatment aims, the kind of tumor, and the presence of metastasis.10 In almost 80% of cases, pleural biopsy and cytology are the only specimens obtained for analysis.Therefore, identification of these markers is practical for diagnosis.10 For this reason, pleural effusion samples are appropriate options to lung biopsy for molecular studies.10
Ki-67 LI in samples has the highest specificity and sensitivity for low-tointermediate- grade vs high-grade tumors. It is being used for guiding clinical and treatment decisions.6 In SCLC, the Ki-67 LI is not necessary for diagnosis but will be about 80%.11 The tumor cells will show epithelial characteristics with positive cytokeratin AE1/AE3 and monoclonal antibody CAM5.2 and neuroendocrine markers, including NCAM/CD56, chromogranin A, and synaptophysin.11
Thyroid transcription factor-1 (TTF- 1) is positive in most cases. In LCNEC, the Ki-67 LI is between 40% and 80%. NCAM/ CD56, chromogranin A, and synaptophysin are present in 92 to 100%, 80 to 85%, and 50 to 60%, respectively.11 TTF-1 is identified in half of the tumors. All these tumors express pancytokeratin (AE1/AE3), cytokeratin 7 or low-molecular-weight cytokeratin. Likewise, the carcinoids will show markers, such as chromogranin A, synaptophysin, CD56, and epithelial markers like pancytokeratin.11 However, the high-molecular-weight cytokeratin and TTF-1 are negative. Furthermore, NSE is considered a good tumor marker in the diagnosis and prognosis of SCLC. NSE also has been reported in NSCLC. The level of NSE correlates with tumor burden, number of metastatic sites, and response to treatment. 12 A potentially useful marker is the insulinoma-associated protein 1, which is a nuclear determinant of NE differentiation that stains all types of pulmonary NETs irrespective of the histology but does not stain adenocarcinoma or squamous cell carcinoma (SCC).6
Recently, genomic studies have identified gene alterations that have become standard of care for diagnosis and targeted therapies.8 For example, epidermal growth factor receptor (EGFR) and echinoderm microtubule- associated proteinlike 4, and anaplastic lymphoma kinase (EML4-ALK) mutations have been found in about 25% of lung adenocarcinomas. 8 Other abnormalities in LKB1/STK11, NF1, CDKN2A, SMARCA4 and KEAP1, KRAS, MET, ROS1, and RET have also been identified.8 On the other hand, SCC rarely have derangements in EGFR and EML4-ALK, but do show changes in RTKs, DDR2M, FGGRs, among others.8 In TC and AC, observed molecular alterations include MEN1 mutations, mTOR, and SSTRs pathway activation, and GC/ CEACAM1 and CD44/OTP expression.13 LCNEC and SCLC have shown TP53 and RB1 mutations and CDX2/VIL1/BAI3 expression. DLL3 expression and MET mutations may be present in SCLC.13 Last, chromatin remodeling gene mutations have been identified in all these lung NET types.13
Furthermore, neuropeptides and neuroamines may be measured in the blood and urine.14 Pulmonary NETs may be functional and secrete these substances, leading to systemic symptoms based on the released molecules.15 However, pulmonary NETs produce less serotonin than gastrointestinal NETs; therefore, carcinoid syndrome is less frequent in pulmonary NETs.16 Liver metastasis is often present when it occurs.5 Other possible clinical features include Cushing syndrome and acromegaly depending on the secreted hormones.5
In a recent metanalysis, serum LDH has been found to have a prognostic role in Ewing sarcoma, urologic cancers, malignant mesothelioma, among others.17 It demonstrated that a higher LDH concentration is associated with worse survival in patients with lung cancer.17 Serum LDH is an enzyme that catalyzes the reaction between lactic acid and pyruvic acid that typically takes place in anaerobic conditions.17 LDH levels are elevated in malignancies because tumors have an anaerobic environment. Elevated LDH levels correlate with the anaerobic metabolism in the tumor. Other studies also have noted that patients with high metastatic score have higher LDH levels.17 Therefore, LDH may reflect tumor extension.
In addition, other techniques, such as somatostatin- receptor imaging are specifically beneficial in tumors that express the somatostatin receptor.16 For this reason, this type of study is typically indicated in patients with known metastasis, not in patients with low-grade tumors. Abdominal CT scans are done because the liver is a common site for metastasis.
Our case report demonstrates how biomarkers help diagnose these potentially aggressive and life-threatening tumors that may present as a common condition such as a pleural effusion. Using a less invasive and quicker approach with thoracentesis rather than with lung biopsies is a diagnostic tool in this entity. IHC in cell blocks is a reasonable diagnostic method especially in patients in whom performing a lung biopsy is difficult.
Conclusions
The presence of a symptomatic and recurrent unilateral pleural effusion must urge physicians to consider thoracentesis with mindful use of biomarkers not only for therapeutic purposes, but also for diagnosis of a variety of etiologies, both benign and malignant.
Neuroendocrine tumors (NETs) account for about 0.5% of all newly diagnosed malignancies.1 Pulmonary NETs are rare, accounting for 1 to 2% of all invasive lung malignancies and involve about 20 to 25% of primary lung malignancies. 2,3 Their prevalence has increased by an estimated 6% per year over the past 30 years.2 Nonetheless, the time of diagnosis is frequently delayed because of nonspecific symptoms that may imitate other pulmonary conditions.
In the normal pleural space, there is a steady state in which there is a roughly equal rate of fluid formation and absorption. Any disequilibrium may produce a pleural effusion. Pleural fluids can be transudates or exudates. Transudates result from imbalances in hydrostatic and oncotic pressures in the pleural space. Exudates result primarily from pleural and/or lung inflammation or from impaired lymphatic drainage of the pleural space. Clinical manifestations include cough, wheezing, recurrent pneumonia, hemoptysis and pleural effusions. We present a case of a man who developed a large left pleural effusion with a pathology report suggesting a pulmonary NET as the etiology. Being aware of this rare entity may help improve prognosis by making an earlier diagnosis and starting treatment sooner.
Case Presentation
A 90-year-old man with a medical history of arterial hypertension, hyperlipidemia, type 2 diabetes mellitus, coronary artery disease, and vascular dementia presented to the emergency department with hypoactivity, poor appetite, productive cough, and shortness of breath. The patient was a former smoker (unknown pack-years) who quit smoking cigarettes 7 years prior. Vital signs showed sinus tachycardia and peripheral oxygen saturation of 90% at room air. The initial physical examination was remarkable for decreased breath sounds and crackles at the left lung base. Laboratory findings showed leukocytosis with neutrophilia and chronic normocytic anemia. Chest computed tomography (CT) showed a large left-sided pleural effusion occupying most of the left hemithorax with adjacent atelectatic lung, enlarged pretracheal, subcarinal, and left perihilar lymph nodes (Figure 1).
The patient was admitted to the internal medicine ward with the diagnosis of left pneumonic process and started on IV levofloxacin. However, despite 7 days of antibiotic therapy, the patient’s respiratory symptoms worsened. This clinical deterioration prompted pulmonary service consultation. Chest radiography demonstrated an enlarging left pleural effusion (Figure 2). A thoracentesis drained 1.2 L of serosanguineous pleural fluid. Pleural fluid analysis showed a cell count of 947/cm3 with 79% of lymphocytes, total protein 3.8 g/dL, lactic dehydrogenase (LDH) level 607 U/L, and glucose level 109 mg/dL. Serum total protein was 6.62 g/dL, LDH 666 U/L and glucose 92 mg/dL (Tables 1 and 2). Alanine transaminase (ALT) and aspartate aminotransferase (AST) were 11 U/L and 21 U/L, respectively. Using Light criteria, the pleural:serum protein ratio was 0.57, the pleural:serum LDH ratio was 0.91, and the pleural LDH was more than two-thirds of the serum LDH. These calculations were consistent with an exudative effusion. An infectious disease workup, including blood and pleural fluid cultures, was negative.
The pleural fluid concentrated cell block hematoxylin and eosin (H&E) staining showed chromatin, prominent nucleoli, and nuclear molding, which was compatible with high-grade lung NET (Figure 3). The cell block immunohistochemistry (IHC) was positive for synaptophysin, chromogranin A, and neuron specific enolase (NSE) also consistent with a high-grade pulmonary NET (Figure 4). The proliferation marker protein Ki-67 labeling index (LI) showed a proliferation index > 20% (Figure 5). The patient did not have decision-making capacity given vascular dementia. Multiple attempts to contact the next of kin or family members were unsuccessful. Risks vs benefits were evaluated, and given the patient’s advanced age and multiple comorbidities, a conservative management approach under palliative care was chosen. For this reason, further genomic studies were not done.
Discussion
NETs are a group of neoplasms that differ in site, amount of cell propagation, and clinical manifestations.4 These tumors are rare with an estimated incidence of 25 to 50 per 100,000.4 The most commonly affected organ systems are the gastroenteropancreatic and the bronchopulmonary tracts, accounting for 60% and 25% of the tumors, respectively.4 The incidence is increasing over the past years in part because of novel diagnostic techniques.
The average age of diagnosis is between the fourth and sixth decades, affecting more women than men.5 Smoking has been identified as a possible culprit for the development of these neoplasms; nonetheless, the association is still not clear.4 For example, poorly differentiated pulmonary NETs have a strong association with smoking but not well-differentiated pulmonary NETs.2
Patients typically present with cough, wheezing, hemoptysis, and recurrent pneumonias, which are in part a consequence of obstruction caused by the mass.2 Sometimes, obstruction may yield persistent pleural effusions. Hemoptysis may be seen secondary to the vascularity of pulmonary NETs.
The diagnosis is often delayed because patients are frequently treated for infection before being diagnosed with the malignancy, such as in our case. Radiologic image findings include round opacities, central masses, and atelectasis. Pulmonary NETs are frequently found incidentally as solitary lung nodules. The CT scan is the most common diagnostic modality and can provide information about the borders of the tumor, the location and surrounding structures, including the presence of atelectasis.5 Pulmonary NETs are usually centrally located in an accessible region for lung biopsy. In cases where the mass is not easily reachable, thoracentesis may provide the only available specimen.
The 2015 World Health Organization classification has identified 4 histologic types of pulmonary NETs, namely, typical carcinoid (TC), atypical carcinoid (AC), large cell neuroendocrine carcinoma (LCNEC) and small cell lung carcinoma (SCLC).6 The low-grade pulmonary NET, the typical carcinoid, is slow growing and has lower rates of metastasis. The intermediate-grade NET, the atypical carcinoid, is more aggressive. The highgrade NETs, the LCNEC and the SCLC, are aggressive and spread quickly to other places.6 Consequently, LCNEC and SCLC have higher mortalities with a 5-year survival, ranging from 13 to 57% and 5%, respectively.7
Tumors may be histomorphologically classified by H&E staining. The main characteristics that differentiate the low- and high-grade NETs are the presence of necrosis and the mitotic rate. Both categories form neuropeptides and have dense granular cores when seen with an electron microscopy.6 The TC and AC have welldefined, organized histologic patterns, no necrosis, and scarce mitosis. On the other hand, the LCNEC and SCLC are poorly differentiated tumors with necrosis, atypia, and mitosis.6 LCNEC can be separated from SCLC and other tumors by IHC staining, whereas SCLC is primarily distinguished by morphology.
If the biopsy sample size is small, then IHC morphology and markers are helpful for subclassification.8 IHC is used to discern between neuroendocrine (NE) vs non-NE. The evaluation of pleural fluid includes preparation of cell blocks. Cell block staining is deemed better for IHC because it mimics a small biopsy that enables superior stains.9 The need for a pleural biopsy in cases where the cytology is negative depends on treatment aims, the kind of tumor, and the presence of metastasis.10 In almost 80% of cases, pleural biopsy and cytology are the only specimens obtained for analysis.Therefore, identification of these markers is practical for diagnosis.10 For this reason, pleural effusion samples are appropriate options to lung biopsy for molecular studies.10
Ki-67 LI in samples has the highest specificity and sensitivity for low-tointermediate- grade vs high-grade tumors. It is being used for guiding clinical and treatment decisions.6 In SCLC, the Ki-67 LI is not necessary for diagnosis but will be about 80%.11 The tumor cells will show epithelial characteristics with positive cytokeratin AE1/AE3 and monoclonal antibody CAM5.2 and neuroendocrine markers, including NCAM/CD56, chromogranin A, and synaptophysin.11
Thyroid transcription factor-1 (TTF- 1) is positive in most cases. In LCNEC, the Ki-67 LI is between 40% and 80%. NCAM/ CD56, chromogranin A, and synaptophysin are present in 92 to 100%, 80 to 85%, and 50 to 60%, respectively.11 TTF-1 is identified in half of the tumors. All these tumors express pancytokeratin (AE1/AE3), cytokeratin 7 or low-molecular-weight cytokeratin. Likewise, the carcinoids will show markers, such as chromogranin A, synaptophysin, CD56, and epithelial markers like pancytokeratin.11 However, the high-molecular-weight cytokeratin and TTF-1 are negative. Furthermore, NSE is considered a good tumor marker in the diagnosis and prognosis of SCLC. NSE also has been reported in NSCLC. The level of NSE correlates with tumor burden, number of metastatic sites, and response to treatment. 12 A potentially useful marker is the insulinoma-associated protein 1, which is a nuclear determinant of NE differentiation that stains all types of pulmonary NETs irrespective of the histology but does not stain adenocarcinoma or squamous cell carcinoma (SCC).6
Recently, genomic studies have identified gene alterations that have become standard of care for diagnosis and targeted therapies.8 For example, epidermal growth factor receptor (EGFR) and echinoderm microtubule- associated proteinlike 4, and anaplastic lymphoma kinase (EML4-ALK) mutations have been found in about 25% of lung adenocarcinomas. 8 Other abnormalities in LKB1/STK11, NF1, CDKN2A, SMARCA4 and KEAP1, KRAS, MET, ROS1, and RET have also been identified.8 On the other hand, SCC rarely have derangements in EGFR and EML4-ALK, but do show changes in RTKs, DDR2M, FGGRs, among others.8 In TC and AC, observed molecular alterations include MEN1 mutations, mTOR, and SSTRs pathway activation, and GC/ CEACAM1 and CD44/OTP expression.13 LCNEC and SCLC have shown TP53 and RB1 mutations and CDX2/VIL1/BAI3 expression. DLL3 expression and MET mutations may be present in SCLC.13 Last, chromatin remodeling gene mutations have been identified in all these lung NET types.13
Furthermore, neuropeptides and neuroamines may be measured in the blood and urine.14 Pulmonary NETs may be functional and secrete these substances, leading to systemic symptoms based on the released molecules.15 However, pulmonary NETs produce less serotonin than gastrointestinal NETs; therefore, carcinoid syndrome is less frequent in pulmonary NETs.16 Liver metastasis is often present when it occurs.5 Other possible clinical features include Cushing syndrome and acromegaly depending on the secreted hormones.5
In a recent metanalysis, serum LDH has been found to have a prognostic role in Ewing sarcoma, urologic cancers, malignant mesothelioma, among others.17 It demonstrated that a higher LDH concentration is associated with worse survival in patients with lung cancer.17 Serum LDH is an enzyme that catalyzes the reaction between lactic acid and pyruvic acid that typically takes place in anaerobic conditions.17 LDH levels are elevated in malignancies because tumors have an anaerobic environment. Elevated LDH levels correlate with the anaerobic metabolism in the tumor. Other studies also have noted that patients with high metastatic score have higher LDH levels.17 Therefore, LDH may reflect tumor extension.
In addition, other techniques, such as somatostatin- receptor imaging are specifically beneficial in tumors that express the somatostatin receptor.16 For this reason, this type of study is typically indicated in patients with known metastasis, not in patients with low-grade tumors. Abdominal CT scans are done because the liver is a common site for metastasis.
Our case report demonstrates how biomarkers help diagnose these potentially aggressive and life-threatening tumors that may present as a common condition such as a pleural effusion. Using a less invasive and quicker approach with thoracentesis rather than with lung biopsies is a diagnostic tool in this entity. IHC in cell blocks is a reasonable diagnostic method especially in patients in whom performing a lung biopsy is difficult.
Conclusions
The presence of a symptomatic and recurrent unilateral pleural effusion must urge physicians to consider thoracentesis with mindful use of biomarkers not only for therapeutic purposes, but also for diagnosis of a variety of etiologies, both benign and malignant.
1. Oronsky B, Ma PC, Morgensztern D, Carter CA. Nothing but NET: a review of neuroendocrine tumors and carcinomas. Neoplasia. 2017;19(12):991-1002. doi: 10.1016/j.neo.2017.09.002
2. Hendifar AE, Marchevsky AM, Tuli R. Neuroendocrine tumors of the lung: current challenges and advances in the diagnosis and management of well-differentiated disease. J Thorac Oncol. 2017;12(3):425-436. doi: 10.1016/j.jtho.2016.11.2222
3. Fisseler-Eckhoff A, Demes M. Neuroendocrine tumors of the lung. Cancers (Basel). 2012;4(3):777-798. doi: 10.3390/cancers4030777
4. Mandegaran R, David S, Screaton N. Cardiothoracic manifestations of neuroendocrine tumours. Br J Radiol. 2016;89(1060). doi: 10.1259/bjr.20150787
5. Caplin ME, Baudin E, Ferolla P, et al; ENETS consensus conference participants. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620. doi: 10.1093/annonc/mdv041
6. Pelosi G, Sonzogni A, Harari S, et al. Classification of pulmonary neuroendocrine tumors: new insights. Transl Lung Cancer Res. 2017;6(5):513-529. doi: 10.21037/tlcr.2017.09.04
7. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394.
8. Osmani L, Askin F, Gabrielson E, Li QK. Current WHO guidelines and the critical role of immunohistochemical markers in the subclassification of non-small cell lung carcinoma (NSCLC): moving from targeted therapy to immunotherapy. Semin Cancer Biol. 2018;52(pt 1):103-109. doi: 10.1016/j.semcancer.2017.11.019
9. Kaur G, Nijhawan R, Gupta N, Singh N, Rajwanshi A. Pleural fluid cytology samples in cases of suspected lung cancer: an experience from a tertiary care centre. Diagn Cytopathol. 2017;45(3):195-201.
10. Porcel JM. Biomarkers in the diagnosis of pleural diseases: a 2018 update. Ther Adv Respir Dis. 2018;12. doi: 10.1177/1753466618808660
11. Kim JY, Hong SM, Ro JY. Recent updates on grading and classification of neuroendocrine tumors. Ann Diagn Pathol. 2017;29:11-16. doi: 10.1016/j.anndiagpath.2017.04.005
12. Isgrò MA, Bottoni P, Scatena R. Neuron-specific enolase as a biomarker: biochemical and clinical aspects. Adv Exp Med Biol. 2015;867:125-143. doi: 10.1007/978-94-017-7215-0_9
13. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394
14. Eriksson B, Oberg K, Stridsberg M. Tumor markers in neuroendocrine tumors. Digestion. 2000;62(suppl 1):33-38.
15. Melosky B. Low grade neuroendocrine tumors of the lung. Front Oncol. 2017;7:119. doi: 10.3389/fonc.2017.00119
16. Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2001;113(1):5-21. https://doi.org/10.1002/cncr.23542
17. Deng T, Zhang J, Meng Y, Zhou Y, Li W. Higher pretreatment lactate dehydrogenase concentration predicts worse overall survival in patients with lung cancer. Medicine (Baltimore). 2018;97(38):e12524
1. Oronsky B, Ma PC, Morgensztern D, Carter CA. Nothing but NET: a review of neuroendocrine tumors and carcinomas. Neoplasia. 2017;19(12):991-1002. doi: 10.1016/j.neo.2017.09.002
2. Hendifar AE, Marchevsky AM, Tuli R. Neuroendocrine tumors of the lung: current challenges and advances in the diagnosis and management of well-differentiated disease. J Thorac Oncol. 2017;12(3):425-436. doi: 10.1016/j.jtho.2016.11.2222
3. Fisseler-Eckhoff A, Demes M. Neuroendocrine tumors of the lung. Cancers (Basel). 2012;4(3):777-798. doi: 10.3390/cancers4030777
4. Mandegaran R, David S, Screaton N. Cardiothoracic manifestations of neuroendocrine tumours. Br J Radiol. 2016;89(1060). doi: 10.1259/bjr.20150787
5. Caplin ME, Baudin E, Ferolla P, et al; ENETS consensus conference participants. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620. doi: 10.1093/annonc/mdv041
6. Pelosi G, Sonzogni A, Harari S, et al. Classification of pulmonary neuroendocrine tumors: new insights. Transl Lung Cancer Res. 2017;6(5):513-529. doi: 10.21037/tlcr.2017.09.04
7. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394.
8. Osmani L, Askin F, Gabrielson E, Li QK. Current WHO guidelines and the critical role of immunohistochemical markers in the subclassification of non-small cell lung carcinoma (NSCLC): moving from targeted therapy to immunotherapy. Semin Cancer Biol. 2018;52(pt 1):103-109. doi: 10.1016/j.semcancer.2017.11.019
9. Kaur G, Nijhawan R, Gupta N, Singh N, Rajwanshi A. Pleural fluid cytology samples in cases of suspected lung cancer: an experience from a tertiary care centre. Diagn Cytopathol. 2017;45(3):195-201.
10. Porcel JM. Biomarkers in the diagnosis of pleural diseases: a 2018 update. Ther Adv Respir Dis. 2018;12. doi: 10.1177/1753466618808660
11. Kim JY, Hong SM, Ro JY. Recent updates on grading and classification of neuroendocrine tumors. Ann Diagn Pathol. 2017;29:11-16. doi: 10.1016/j.anndiagpath.2017.04.005
12. Isgrò MA, Bottoni P, Scatena R. Neuron-specific enolase as a biomarker: biochemical and clinical aspects. Adv Exp Med Biol. 2015;867:125-143. doi: 10.1007/978-94-017-7215-0_9
13. Rossi G, Bertero L, Marchiò C, Papotti M. Molecular alterations of neuroendocrine tumours of the lung. Histopathology. 2018;72(1):142-152. doi: 10.1111/his.13394
14. Eriksson B, Oberg K, Stridsberg M. Tumor markers in neuroendocrine tumors. Digestion. 2000;62(suppl 1):33-38.
15. Melosky B. Low grade neuroendocrine tumors of the lung. Front Oncol. 2017;7:119. doi: 10.3389/fonc.2017.00119
16. Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2001;113(1):5-21. https://doi.org/10.1002/cncr.23542
17. Deng T, Zhang J, Meng Y, Zhou Y, Li W. Higher pretreatment lactate dehydrogenase concentration predicts worse overall survival in patients with lung cancer. Medicine (Baltimore). 2018;97(38):e12524
Tepotinib elicits responses in METex14 NSCLC
The objective response rate was 46.5% among the 99 patients followed for 9 or more months, as assessed by independent reviewers. There were no complete responders, according to the reviewers.
However, the response rate according to investigator assessment was 55.6%, including two responses that were judged to be complete.
The median duration of response was 11.1 months according to reviewers and 14 months according to investigators.
“The success of this trial, alongside other studies on the same class of drugs, establishes MET exon 14 as an actionable target for non–small cell lung cancer,” said senior author Xiuning Le, MD, PhD, assistant professor in the department of thoracic/head and neck medical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Le presented results from this trial as part of the American Society of Clinical Oncology virtual scientific program. Results were published simultaneously in the New England Journal of Medicine.
The trial, dubbed VISION, already won tepotinib approval in Japan to treat NSCLC patients with MET exon 14–skipping mutations. The Food and Drug Administration has granted tepotinib breakthrough status, and Merck, the drug’s manufacturer, plans to submit tepotinib for review this year.
The VISION trial enrolled 152 patients with NSCLC – 99 with at least 9 months of follow-up and 53 with shorter follow-up. The patients’ MET exon 14 mutations were confirmed by liquid or tissue biopsy.
The patients’ median age at baseline was 74 years, 54% were men, and almost half had no smoking history. Patients received tepotinib at 500 mg daily until disease progression or intolerable toxicity.
Overall, the VISION results “compare favorably” with those from studies of other MET inhibitors, the investigators wrote.
The 46.5% objective response rate (per independent reviewers) included the 99 patients with follow-up of at least 9 months who were liquid- or tissue-biopsy positive (combined group). The response rate was 48.5% among the 66 patients with positive liquid biopsies and 50% among the 60 patients with positive tissue biopsies.
The median progression-free survival was 8.5 months in the combined group, 8.5 months in the liquid-biopsy group, and 11 months in the tissue-biopsy group. The median overall survival was 17.1 months, 15.8 months, and 22.3 months, respectively.
The 11 patients with brain metastases at baseline had results that were in line with the other patients’ results. Patients with brain metastases had an objective response rate of 54.5%, a median response duration of 9.5 months, and a median progression-free survival of 10.9 months.
Overall, 88.8% of patients reported adverse events related to treatment, including peripheral edema in 63.2%, nausea in 25.7%, and diarrhea in 21.7%.
Grade 3-4 adverse events occurred in 27% of patients. Peripheral edema was the most common of these events, reported in 7.2% of patients.
“Proactive monitoring for peripheral edema is recommended and can be managed with temporary discontinuation of tepotinib or dose reduction,” the investigators wrote.
The death of a 79-year-old patient with respiratory failure and dyspnea, secondary to interstitial lung disease, was the only death considered to be treatment related.
The study was funded by Merck. Dr. Le and other investigators disclosed relationships, including employment, with the company.
SOURCE: Le X et al. ASCO 2020, Abstract 9556.
The objective response rate was 46.5% among the 99 patients followed for 9 or more months, as assessed by independent reviewers. There were no complete responders, according to the reviewers.
However, the response rate according to investigator assessment was 55.6%, including two responses that were judged to be complete.
The median duration of response was 11.1 months according to reviewers and 14 months according to investigators.
“The success of this trial, alongside other studies on the same class of drugs, establishes MET exon 14 as an actionable target for non–small cell lung cancer,” said senior author Xiuning Le, MD, PhD, assistant professor in the department of thoracic/head and neck medical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Le presented results from this trial as part of the American Society of Clinical Oncology virtual scientific program. Results were published simultaneously in the New England Journal of Medicine.
The trial, dubbed VISION, already won tepotinib approval in Japan to treat NSCLC patients with MET exon 14–skipping mutations. The Food and Drug Administration has granted tepotinib breakthrough status, and Merck, the drug’s manufacturer, plans to submit tepotinib for review this year.
The VISION trial enrolled 152 patients with NSCLC – 99 with at least 9 months of follow-up and 53 with shorter follow-up. The patients’ MET exon 14 mutations were confirmed by liquid or tissue biopsy.
The patients’ median age at baseline was 74 years, 54% were men, and almost half had no smoking history. Patients received tepotinib at 500 mg daily until disease progression or intolerable toxicity.
Overall, the VISION results “compare favorably” with those from studies of other MET inhibitors, the investigators wrote.
The 46.5% objective response rate (per independent reviewers) included the 99 patients with follow-up of at least 9 months who were liquid- or tissue-biopsy positive (combined group). The response rate was 48.5% among the 66 patients with positive liquid biopsies and 50% among the 60 patients with positive tissue biopsies.
The median progression-free survival was 8.5 months in the combined group, 8.5 months in the liquid-biopsy group, and 11 months in the tissue-biopsy group. The median overall survival was 17.1 months, 15.8 months, and 22.3 months, respectively.
The 11 patients with brain metastases at baseline had results that were in line with the other patients’ results. Patients with brain metastases had an objective response rate of 54.5%, a median response duration of 9.5 months, and a median progression-free survival of 10.9 months.
Overall, 88.8% of patients reported adverse events related to treatment, including peripheral edema in 63.2%, nausea in 25.7%, and diarrhea in 21.7%.
Grade 3-4 adverse events occurred in 27% of patients. Peripheral edema was the most common of these events, reported in 7.2% of patients.
“Proactive monitoring for peripheral edema is recommended and can be managed with temporary discontinuation of tepotinib or dose reduction,” the investigators wrote.
The death of a 79-year-old patient with respiratory failure and dyspnea, secondary to interstitial lung disease, was the only death considered to be treatment related.
The study was funded by Merck. Dr. Le and other investigators disclosed relationships, including employment, with the company.
SOURCE: Le X et al. ASCO 2020, Abstract 9556.
The objective response rate was 46.5% among the 99 patients followed for 9 or more months, as assessed by independent reviewers. There were no complete responders, according to the reviewers.
However, the response rate according to investigator assessment was 55.6%, including two responses that were judged to be complete.
The median duration of response was 11.1 months according to reviewers and 14 months according to investigators.
“The success of this trial, alongside other studies on the same class of drugs, establishes MET exon 14 as an actionable target for non–small cell lung cancer,” said senior author Xiuning Le, MD, PhD, assistant professor in the department of thoracic/head and neck medical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Le presented results from this trial as part of the American Society of Clinical Oncology virtual scientific program. Results were published simultaneously in the New England Journal of Medicine.
The trial, dubbed VISION, already won tepotinib approval in Japan to treat NSCLC patients with MET exon 14–skipping mutations. The Food and Drug Administration has granted tepotinib breakthrough status, and Merck, the drug’s manufacturer, plans to submit tepotinib for review this year.
The VISION trial enrolled 152 patients with NSCLC – 99 with at least 9 months of follow-up and 53 with shorter follow-up. The patients’ MET exon 14 mutations were confirmed by liquid or tissue biopsy.
The patients’ median age at baseline was 74 years, 54% were men, and almost half had no smoking history. Patients received tepotinib at 500 mg daily until disease progression or intolerable toxicity.
Overall, the VISION results “compare favorably” with those from studies of other MET inhibitors, the investigators wrote.
The 46.5% objective response rate (per independent reviewers) included the 99 patients with follow-up of at least 9 months who were liquid- or tissue-biopsy positive (combined group). The response rate was 48.5% among the 66 patients with positive liquid biopsies and 50% among the 60 patients with positive tissue biopsies.
The median progression-free survival was 8.5 months in the combined group, 8.5 months in the liquid-biopsy group, and 11 months in the tissue-biopsy group. The median overall survival was 17.1 months, 15.8 months, and 22.3 months, respectively.
The 11 patients with brain metastases at baseline had results that were in line with the other patients’ results. Patients with brain metastases had an objective response rate of 54.5%, a median response duration of 9.5 months, and a median progression-free survival of 10.9 months.
Overall, 88.8% of patients reported adverse events related to treatment, including peripheral edema in 63.2%, nausea in 25.7%, and diarrhea in 21.7%.
Grade 3-4 adverse events occurred in 27% of patients. Peripheral edema was the most common of these events, reported in 7.2% of patients.
“Proactive monitoring for peripheral edema is recommended and can be managed with temporary discontinuation of tepotinib or dose reduction,” the investigators wrote.
The death of a 79-year-old patient with respiratory failure and dyspnea, secondary to interstitial lung disease, was the only death considered to be treatment related.
The study was funded by Merck. Dr. Le and other investigators disclosed relationships, including employment, with the company.
SOURCE: Le X et al. ASCO 2020, Abstract 9556.
FROM ASCO 2020
American Cancer Society update: ‘It is best not to drink alcohol’
In its updated cancer prevention guidelines, the American Cancer Society now recommends that “it is best not to drink alcohol.”
Previously, ACS suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive.
The revised guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity.
But importantly, there is also a call for action from public, private, and community organizations to work to together to increase access to affordable, nutritious foods and physical activity.
“Making healthy choices can be challenging for many, and there are strategies included in the guidelines that communities can undertake to help reduce barriers to eating well and physical activity,” said Laura Makaroff, DO, American Cancer Society senior vice president. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support.”
The guidelines were published in CA: A Cancer Journal for Clinicians.
The link between cancer and lifestyle factors has long been established, and for the past 4 decades, both government and leading nonprofit health organizations, including the ACS and the World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR), have released cancer prevention guidelines and recommendations that focus on managing weight, diet, physical activity, and alcohol consumption.
In 2012, the ACS issued guidelines on diet and physical activity, and their current guideline is largely based on the WCRF/AICR systematic reviews and Continuous Update Project reports, which were last updated in 2018. The ACS guidelines also incorporated systematic reviews conducted by the International Agency on Cancer Research (IARC) and the U.S. Department of Agriculture and the Department of Health and Human Services (USDA/HHS) and other analyses that were published since the WCRF/AICR recommendations were released.
Emphasis on three areas
The differences between the old guidelines and the update do not differ dramatically, but Makaroff highlighted a few areas that have increased emphasis.
Time spent being physically active is critical. The recommendation has changed to encourage adults to engage in 150-300 minutes (2.5-5 hours) of moderate-intensity physical activity, or 75-150 minutes (1.25-2.5 hours) of vigorous-intensity physical activity, or an equivalent combination, per week. Achieving or exceeding the upper limit of 300 minutes is optimal.
“That is more than what we have recommended in the past, along with the continued message that children and adolescents engage in at least 1 hour of moderate- or vigorous-intensity activity each day,” she told Medscape Medical News.
The ACS has also increased emphasis on reducing the consumption of processed and red meat. “This is part of a healthy eating pattern and making sure that people are eating food that is high in nutrients that help achieve and maintain a healthy body weight,” said Makaroff.
A healthy diet should include a variety of dark green, red, and orange vegetables; fiber-rich legumes; and fruits with a variety of colors and whole grains, according to the guidelines. Sugar-sweetened beverages, highly processed foods, and refined grain products should be limited or avoided.
The revised dietary recommendations reflect a shift from a “reductionist or nutrient-centric” approach to one that is more “holistic” and that focuses on dietary patterns. In contrast to a focus on individual nutrients and bioactive compounds, the new approach is more consistent with what and how people actually eat, ACS points out.
The third area that Makaroff highlighted is alcohol, where the recommendation is to avoid or limit consumption. “The current update says not to drink alcohol, which is in line with the scientific evidence, but for those people who choose to drink alcohol, to limit it to one drink per day for women and two drinks per day for men.”
Thus, the change here is that the previous guideline only recommended limiting alcohol consumption, while the update suggests that, optimally, it should be avoided completely.
The ACS has also called for community involvement to help implement these goals: “Public, private, and community organizations should work collaboratively at national, state, and local levels to develop, advocate for, and implement policy and environmental changes that increase access to affordable, nutritious foods; provide safe, enjoyable, and accessible opportunities for physical activity; and limit alcohol for all individuals.”
No smoking guns
Commenting on the guidelines, Steven K. Clinton, MD, PhD, associate director of the Center for Advanced Functional Foods Research and Entrepreneurship at the Ohio State University, Columbus, explained that he didn’t view the change in alcohol as that much of an evolution. “It’s been 8 years since they revised their overall guidelines, and during that time frame, there has been an enormous growth in the evidence that has been used by many organizations,” he said.
Clinton noted that the guidelines are consistent with the whole body of current scientific literature. “It’s very easy to go to the document and look for the ‘smoking gun’ – but the smoking gun is really not one thing,” he said. “It’s a pattern, and what dietitians and nutritionists are telling people is that you need to orchestrate a healthy lifestyle and diet, with a diet that has a foundation of fruits, vegetables, whole grains, and modest intake of refined grains and meat. You are orchestrating an entire pattern to get the maximum benefit.”
Makaroff is an employee of the ACS. Clinton has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
In its updated cancer prevention guidelines, the American Cancer Society now recommends that “it is best not to drink alcohol.”
Previously, ACS suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive.
The revised guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity.
But importantly, there is also a call for action from public, private, and community organizations to work to together to increase access to affordable, nutritious foods and physical activity.
“Making healthy choices can be challenging for many, and there are strategies included in the guidelines that communities can undertake to help reduce barriers to eating well and physical activity,” said Laura Makaroff, DO, American Cancer Society senior vice president. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support.”
The guidelines were published in CA: A Cancer Journal for Clinicians.
The link between cancer and lifestyle factors has long been established, and for the past 4 decades, both government and leading nonprofit health organizations, including the ACS and the World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR), have released cancer prevention guidelines and recommendations that focus on managing weight, diet, physical activity, and alcohol consumption.
In 2012, the ACS issued guidelines on diet and physical activity, and their current guideline is largely based on the WCRF/AICR systematic reviews and Continuous Update Project reports, which were last updated in 2018. The ACS guidelines also incorporated systematic reviews conducted by the International Agency on Cancer Research (IARC) and the U.S. Department of Agriculture and the Department of Health and Human Services (USDA/HHS) and other analyses that were published since the WCRF/AICR recommendations were released.
Emphasis on three areas
The differences between the old guidelines and the update do not differ dramatically, but Makaroff highlighted a few areas that have increased emphasis.
Time spent being physically active is critical. The recommendation has changed to encourage adults to engage in 150-300 minutes (2.5-5 hours) of moderate-intensity physical activity, or 75-150 minutes (1.25-2.5 hours) of vigorous-intensity physical activity, or an equivalent combination, per week. Achieving or exceeding the upper limit of 300 minutes is optimal.
“That is more than what we have recommended in the past, along with the continued message that children and adolescents engage in at least 1 hour of moderate- or vigorous-intensity activity each day,” she told Medscape Medical News.
The ACS has also increased emphasis on reducing the consumption of processed and red meat. “This is part of a healthy eating pattern and making sure that people are eating food that is high in nutrients that help achieve and maintain a healthy body weight,” said Makaroff.
A healthy diet should include a variety of dark green, red, and orange vegetables; fiber-rich legumes; and fruits with a variety of colors and whole grains, according to the guidelines. Sugar-sweetened beverages, highly processed foods, and refined grain products should be limited or avoided.
The revised dietary recommendations reflect a shift from a “reductionist or nutrient-centric” approach to one that is more “holistic” and that focuses on dietary patterns. In contrast to a focus on individual nutrients and bioactive compounds, the new approach is more consistent with what and how people actually eat, ACS points out.
The third area that Makaroff highlighted is alcohol, where the recommendation is to avoid or limit consumption. “The current update says not to drink alcohol, which is in line with the scientific evidence, but for those people who choose to drink alcohol, to limit it to one drink per day for women and two drinks per day for men.”
Thus, the change here is that the previous guideline only recommended limiting alcohol consumption, while the update suggests that, optimally, it should be avoided completely.
The ACS has also called for community involvement to help implement these goals: “Public, private, and community organizations should work collaboratively at national, state, and local levels to develop, advocate for, and implement policy and environmental changes that increase access to affordable, nutritious foods; provide safe, enjoyable, and accessible opportunities for physical activity; and limit alcohol for all individuals.”
No smoking guns
Commenting on the guidelines, Steven K. Clinton, MD, PhD, associate director of the Center for Advanced Functional Foods Research and Entrepreneurship at the Ohio State University, Columbus, explained that he didn’t view the change in alcohol as that much of an evolution. “It’s been 8 years since they revised their overall guidelines, and during that time frame, there has been an enormous growth in the evidence that has been used by many organizations,” he said.
Clinton noted that the guidelines are consistent with the whole body of current scientific literature. “It’s very easy to go to the document and look for the ‘smoking gun’ – but the smoking gun is really not one thing,” he said. “It’s a pattern, and what dietitians and nutritionists are telling people is that you need to orchestrate a healthy lifestyle and diet, with a diet that has a foundation of fruits, vegetables, whole grains, and modest intake of refined grains and meat. You are orchestrating an entire pattern to get the maximum benefit.”
Makaroff is an employee of the ACS. Clinton has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
In its updated cancer prevention guidelines, the American Cancer Society now recommends that “it is best not to drink alcohol.”
Previously, ACS suggested that, for those who consume alcoholic beverages, intake should be no more than one drink per day for women or two per day for men. That recommendation is still in place, but is now accompanied by this new, stronger directive.
The revised guidelines also place more emphasis on reducing the consumption of processed and red meat and highly processed foods, and on increasing physical activity.
But importantly, there is also a call for action from public, private, and community organizations to work to together to increase access to affordable, nutritious foods and physical activity.
“Making healthy choices can be challenging for many, and there are strategies included in the guidelines that communities can undertake to help reduce barriers to eating well and physical activity,” said Laura Makaroff, DO, American Cancer Society senior vice president. “Individual choice is an important part of a healthy lifestyle, but having the right policies and environmental factors to break down these barriers is also important, and that is something that clinicians can support.”
The guidelines were published in CA: A Cancer Journal for Clinicians.
The link between cancer and lifestyle factors has long been established, and for the past 4 decades, both government and leading nonprofit health organizations, including the ACS and the World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR), have released cancer prevention guidelines and recommendations that focus on managing weight, diet, physical activity, and alcohol consumption.
In 2012, the ACS issued guidelines on diet and physical activity, and their current guideline is largely based on the WCRF/AICR systematic reviews and Continuous Update Project reports, which were last updated in 2018. The ACS guidelines also incorporated systematic reviews conducted by the International Agency on Cancer Research (IARC) and the U.S. Department of Agriculture and the Department of Health and Human Services (USDA/HHS) and other analyses that were published since the WCRF/AICR recommendations were released.
Emphasis on three areas
The differences between the old guidelines and the update do not differ dramatically, but Makaroff highlighted a few areas that have increased emphasis.
Time spent being physically active is critical. The recommendation has changed to encourage adults to engage in 150-300 minutes (2.5-5 hours) of moderate-intensity physical activity, or 75-150 minutes (1.25-2.5 hours) of vigorous-intensity physical activity, or an equivalent combination, per week. Achieving or exceeding the upper limit of 300 minutes is optimal.
“That is more than what we have recommended in the past, along with the continued message that children and adolescents engage in at least 1 hour of moderate- or vigorous-intensity activity each day,” she told Medscape Medical News.
The ACS has also increased emphasis on reducing the consumption of processed and red meat. “This is part of a healthy eating pattern and making sure that people are eating food that is high in nutrients that help achieve and maintain a healthy body weight,” said Makaroff.
A healthy diet should include a variety of dark green, red, and orange vegetables; fiber-rich legumes; and fruits with a variety of colors and whole grains, according to the guidelines. Sugar-sweetened beverages, highly processed foods, and refined grain products should be limited or avoided.
The revised dietary recommendations reflect a shift from a “reductionist or nutrient-centric” approach to one that is more “holistic” and that focuses on dietary patterns. In contrast to a focus on individual nutrients and bioactive compounds, the new approach is more consistent with what and how people actually eat, ACS points out.
The third area that Makaroff highlighted is alcohol, where the recommendation is to avoid or limit consumption. “The current update says not to drink alcohol, which is in line with the scientific evidence, but for those people who choose to drink alcohol, to limit it to one drink per day for women and two drinks per day for men.”
Thus, the change here is that the previous guideline only recommended limiting alcohol consumption, while the update suggests that, optimally, it should be avoided completely.
The ACS has also called for community involvement to help implement these goals: “Public, private, and community organizations should work collaboratively at national, state, and local levels to develop, advocate for, and implement policy and environmental changes that increase access to affordable, nutritious foods; provide safe, enjoyable, and accessible opportunities for physical activity; and limit alcohol for all individuals.”
No smoking guns
Commenting on the guidelines, Steven K. Clinton, MD, PhD, associate director of the Center for Advanced Functional Foods Research and Entrepreneurship at the Ohio State University, Columbus, explained that he didn’t view the change in alcohol as that much of an evolution. “It’s been 8 years since they revised their overall guidelines, and during that time frame, there has been an enormous growth in the evidence that has been used by many organizations,” he said.
Clinton noted that the guidelines are consistent with the whole body of current scientific literature. “It’s very easy to go to the document and look for the ‘smoking gun’ – but the smoking gun is really not one thing,” he said. “It’s a pattern, and what dietitians and nutritionists are telling people is that you need to orchestrate a healthy lifestyle and diet, with a diet that has a foundation of fruits, vegetables, whole grains, and modest intake of refined grains and meat. You are orchestrating an entire pattern to get the maximum benefit.”
Makaroff is an employee of the ACS. Clinton has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Can an app guide cancer treatment decisions during the pandemic?
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic.
In early April, as the COVID-19 surge was bearing down on New York City, those treatment decisions were “a juggling act every single day,” Jonathan Yang, MD, PhD, a radiation oncologist from New York’s Memorial Sloan Kettering Cancer Center, told Medscape Medical News.
Eventually, a glut of guidelines, recommendations, and expert opinions aimed at helping oncologists emerged. The tools help navigate the complicated risk-benefit analysis of their patient’s risk of infection by SARS-CoV-2 and delaying therapy.
Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help?
Three-Tier Systems Are Not Very Sophisticated
OncCOVID, a free tool that was launched May 26 by the University of Michigan, allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care.
Combining these personal details with data from the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registry and the National Cancer Database, the Michigan app then estimates a patient’s 5- or 10-year survival with immediate vs delayed treatment and weighs that against their risk for COVID-19 using data from the Johns Hopkins Coronavirus Resource Center.
“We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine.
Spratt explained that almost every organization, professional society, and government has created something like a three-tier system. Tier 1 represents urgent cases and patients who need immediate treatment. For tier 2, treatment can be delayed weeks or a month, and with tier 3, it can be delayed until the pandemic is over or it’s deemed safe.
“[This system] sounds good at first glance, but in cancer, we’re always talking about personalized medicine, and it’s mind-blowing that these tier systems are only based on urgency and prognosis,” he told Medscape Medical News.
Spratt offered an example. Consider a patient with a very aggressive brain tumor ― that patient is in tier 1 and should undergo treatment immediately. But will the treatment actually help? And how helpful would the procedure be if, say, the patient is 80 years old and, if infected, would have a 30% to 50% chance of dying from the coronavirus?
“If the model says this guy has a 5% harm and this one has 30% harm, you can use that to help prioritize,” summarized Spratt.
The app can generate risk estimates for patients living anywhere in the world and has already been accessed by people from 37 countries. However, Spratt cautions that it is primarily “designed and calibrated for the US.
“The estimates are based on very large US registries, and though it’s probably somewhat similar across much of the world, there’s probably certain cancer types that are more region specific ― especially something like stomach cancer or certain types of head and neck cancer in parts of Asia, for example,” he said.
Although the app’s COVID-19 data are specific to the county level in the United States, elsewhere in the world, it is only country specific.
“We’re using the best data we have for coronavirus, but everyone knows we still have large data gaps,” he acknowledged.
How Accurate?
Asked to comment on the app, Richard Bleicher, MD, leader of the Breast Cancer Program at Fox Chase Cancer Center, Philadelphia, praised the effort and the goal but had some concerns.
“Several questions arise, most important of which is, How accurate is this, and how has this been validated, if at all ― especially as it is too soon to see the outcomes of patients affected in this pandemic?” he told Medscape Medical News.
“We are imposing delays on a broad scale because of the coronavirus, and we are getting continuously changing data as we test more patients. But both situations are novel and may not be accurately represented by the data being pulled, because the datasets use patients from a few years ago, and confounders in these datasets may not apply to this situation,” Bleicher continued.
Although acknowledging the “value in delineating the risk of dying from cancer vs the risk of dying from the SARS-CoV-2 pandemic,” Bleicher urged caution in using the tool to make individual patient decisions.
“We need to remember that the best of modeling ... can be wildly inaccurate and needs to be validated using patients having the circumstances in question. ... This won’t be possible until long after the pandemic is completed, and so the model’s accuracy remains unknown.”
That sentiment was echoed by Giampaolo Bianchini, MD, head of the Breast Cancer Group, Department of Medical Oncology, Ospedale San Raffaele, in Milan, Italy.
“Arbitrarily postponing and modifying treatment strategies including surgery, radiation therapy, and medical therapy without properly balancing the risk/benefit ratio may lead to significantly worse cancer-related outcomes, which largely exceed the actual risks for COVID,” he wrote in an email.
“The OncCOVID app is a remarkable attempt to fill the gap between perception and estimation,” he said. The app provides side by side the COVID-19 risk estimation and the consequences of arbitrary deviation from the standard of care, observed Bianchini.
However, he pointed out weaknesses, including the fact that the “data generated in literature are not always of high quality and do not take into consideration relevant characteristics of the disease and treatment benefit. It should for sure be used, but then also interpreted with caution.”
Another Italian group responded more positively.
“In our opinion, it could be a useful tool for clinicians,” wrote colleagues Alessio Cortelinni and Giampiero Porzio, both medical oncologists at San Salvatore Hospital and the University of L’Aquila, in Italy. “This Web app might assist clinicians in balancing the risk/benefit ratio of being treated and/or access to the outpatient cancer center for each kind of patient (both early and advanced stages), in order to make a more tailored counseling,” they wrote in an email. “Importantly, the Web app might help those clinicians who work ‘alone,’ in peripheral centers, without resources, colleagues, and multidisciplinary tumor boards on whom they can rely.”
Bleicher, who was involved in the COVID-19 Breast Cancer Consortium’s recommendations for prioritizing breast cancer treatment, summarized that the app “may end up being close or accurate, but we won’t know except in hindsight.”
This article first appeared on Medscape.com.
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic.
In early April, as the COVID-19 surge was bearing down on New York City, those treatment decisions were “a juggling act every single day,” Jonathan Yang, MD, PhD, a radiation oncologist from New York’s Memorial Sloan Kettering Cancer Center, told Medscape Medical News.
Eventually, a glut of guidelines, recommendations, and expert opinions aimed at helping oncologists emerged. The tools help navigate the complicated risk-benefit analysis of their patient’s risk of infection by SARS-CoV-2 and delaying therapy.
Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help?
Three-Tier Systems Are Not Very Sophisticated
OncCOVID, a free tool that was launched May 26 by the University of Michigan, allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care.
Combining these personal details with data from the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registry and the National Cancer Database, the Michigan app then estimates a patient’s 5- or 10-year survival with immediate vs delayed treatment and weighs that against their risk for COVID-19 using data from the Johns Hopkins Coronavirus Resource Center.
“We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine.
Spratt explained that almost every organization, professional society, and government has created something like a three-tier system. Tier 1 represents urgent cases and patients who need immediate treatment. For tier 2, treatment can be delayed weeks or a month, and with tier 3, it can be delayed until the pandemic is over or it’s deemed safe.
“[This system] sounds good at first glance, but in cancer, we’re always talking about personalized medicine, and it’s mind-blowing that these tier systems are only based on urgency and prognosis,” he told Medscape Medical News.
Spratt offered an example. Consider a patient with a very aggressive brain tumor ― that patient is in tier 1 and should undergo treatment immediately. But will the treatment actually help? And how helpful would the procedure be if, say, the patient is 80 years old and, if infected, would have a 30% to 50% chance of dying from the coronavirus?
“If the model says this guy has a 5% harm and this one has 30% harm, you can use that to help prioritize,” summarized Spratt.
The app can generate risk estimates for patients living anywhere in the world and has already been accessed by people from 37 countries. However, Spratt cautions that it is primarily “designed and calibrated for the US.
“The estimates are based on very large US registries, and though it’s probably somewhat similar across much of the world, there’s probably certain cancer types that are more region specific ― especially something like stomach cancer or certain types of head and neck cancer in parts of Asia, for example,” he said.
Although the app’s COVID-19 data are specific to the county level in the United States, elsewhere in the world, it is only country specific.
“We’re using the best data we have for coronavirus, but everyone knows we still have large data gaps,” he acknowledged.
How Accurate?
Asked to comment on the app, Richard Bleicher, MD, leader of the Breast Cancer Program at Fox Chase Cancer Center, Philadelphia, praised the effort and the goal but had some concerns.
“Several questions arise, most important of which is, How accurate is this, and how has this been validated, if at all ― especially as it is too soon to see the outcomes of patients affected in this pandemic?” he told Medscape Medical News.
“We are imposing delays on a broad scale because of the coronavirus, and we are getting continuously changing data as we test more patients. But both situations are novel and may not be accurately represented by the data being pulled, because the datasets use patients from a few years ago, and confounders in these datasets may not apply to this situation,” Bleicher continued.
Although acknowledging the “value in delineating the risk of dying from cancer vs the risk of dying from the SARS-CoV-2 pandemic,” Bleicher urged caution in using the tool to make individual patient decisions.
“We need to remember that the best of modeling ... can be wildly inaccurate and needs to be validated using patients having the circumstances in question. ... This won’t be possible until long after the pandemic is completed, and so the model’s accuracy remains unknown.”
That sentiment was echoed by Giampaolo Bianchini, MD, head of the Breast Cancer Group, Department of Medical Oncology, Ospedale San Raffaele, in Milan, Italy.
“Arbitrarily postponing and modifying treatment strategies including surgery, radiation therapy, and medical therapy without properly balancing the risk/benefit ratio may lead to significantly worse cancer-related outcomes, which largely exceed the actual risks for COVID,” he wrote in an email.
“The OncCOVID app is a remarkable attempt to fill the gap between perception and estimation,” he said. The app provides side by side the COVID-19 risk estimation and the consequences of arbitrary deviation from the standard of care, observed Bianchini.
However, he pointed out weaknesses, including the fact that the “data generated in literature are not always of high quality and do not take into consideration relevant characteristics of the disease and treatment benefit. It should for sure be used, but then also interpreted with caution.”
Another Italian group responded more positively.
“In our opinion, it could be a useful tool for clinicians,” wrote colleagues Alessio Cortelinni and Giampiero Porzio, both medical oncologists at San Salvatore Hospital and the University of L’Aquila, in Italy. “This Web app might assist clinicians in balancing the risk/benefit ratio of being treated and/or access to the outpatient cancer center for each kind of patient (both early and advanced stages), in order to make a more tailored counseling,” they wrote in an email. “Importantly, the Web app might help those clinicians who work ‘alone,’ in peripheral centers, without resources, colleagues, and multidisciplinary tumor boards on whom they can rely.”
Bleicher, who was involved in the COVID-19 Breast Cancer Consortium’s recommendations for prioritizing breast cancer treatment, summarized that the app “may end up being close or accurate, but we won’t know except in hindsight.”
This article first appeared on Medscape.com.
Deciding which cancer patients need immediate treatment and who can safely wait is an uncomfortable assessment for cancer clinicians during the COVID-19 pandemic.
In early April, as the COVID-19 surge was bearing down on New York City, those treatment decisions were “a juggling act every single day,” Jonathan Yang, MD, PhD, a radiation oncologist from New York’s Memorial Sloan Kettering Cancer Center, told Medscape Medical News.
Eventually, a glut of guidelines, recommendations, and expert opinions aimed at helping oncologists emerged. The tools help navigate the complicated risk-benefit analysis of their patient’s risk of infection by SARS-CoV-2 and delaying therapy.
Now, a new tool, which appears to be the first of its kind, quantifies that risk-benefit analysis. But its presence immediately raises the question: can it help?
Three-Tier Systems Are Not Very Sophisticated
OncCOVID, a free tool that was launched May 26 by the University of Michigan, allows physicians to individualize risk estimates for delaying treatment of up to 25 early- to late-stage cancers. It includes more than 45 patient characteristics, such as age, location, cancer type, cancer stage, treatment plan, underlying medical conditions, and proposed length of delay in care.
Combining these personal details with data from the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registry and the National Cancer Database, the Michigan app then estimates a patient’s 5- or 10-year survival with immediate vs delayed treatment and weighs that against their risk for COVID-19 using data from the Johns Hopkins Coronavirus Resource Center.
“We thought, isn’t it better to at least provide some evidence-based quantification, rather than a back-of-the-envelope three-tier system that is just sort of ‘made up’?“ explained one of the developers, Daniel Spratt, MD, associate professor of radiation oncology at Michigan Medicine.
Spratt explained that almost every organization, professional society, and government has created something like a three-tier system. Tier 1 represents urgent cases and patients who need immediate treatment. For tier 2, treatment can be delayed weeks or a month, and with tier 3, it can be delayed until the pandemic is over or it’s deemed safe.
“[This system] sounds good at first glance, but in cancer, we’re always talking about personalized medicine, and it’s mind-blowing that these tier systems are only based on urgency and prognosis,” he told Medscape Medical News.
Spratt offered an example. Consider a patient with a very aggressive brain tumor ― that patient is in tier 1 and should undergo treatment immediately. But will the treatment actually help? And how helpful would the procedure be if, say, the patient is 80 years old and, if infected, would have a 30% to 50% chance of dying from the coronavirus?
“If the model says this guy has a 5% harm and this one has 30% harm, you can use that to help prioritize,” summarized Spratt.
The app can generate risk estimates for patients living anywhere in the world and has already been accessed by people from 37 countries. However, Spratt cautions that it is primarily “designed and calibrated for the US.
“The estimates are based on very large US registries, and though it’s probably somewhat similar across much of the world, there’s probably certain cancer types that are more region specific ― especially something like stomach cancer or certain types of head and neck cancer in parts of Asia, for example,” he said.
Although the app’s COVID-19 data are specific to the county level in the United States, elsewhere in the world, it is only country specific.
“We’re using the best data we have for coronavirus, but everyone knows we still have large data gaps,” he acknowledged.
How Accurate?
Asked to comment on the app, Richard Bleicher, MD, leader of the Breast Cancer Program at Fox Chase Cancer Center, Philadelphia, praised the effort and the goal but had some concerns.
“Several questions arise, most important of which is, How accurate is this, and how has this been validated, if at all ― especially as it is too soon to see the outcomes of patients affected in this pandemic?” he told Medscape Medical News.
“We are imposing delays on a broad scale because of the coronavirus, and we are getting continuously changing data as we test more patients. But both situations are novel and may not be accurately represented by the data being pulled, because the datasets use patients from a few years ago, and confounders in these datasets may not apply to this situation,” Bleicher continued.
Although acknowledging the “value in delineating the risk of dying from cancer vs the risk of dying from the SARS-CoV-2 pandemic,” Bleicher urged caution in using the tool to make individual patient decisions.
“We need to remember that the best of modeling ... can be wildly inaccurate and needs to be validated using patients having the circumstances in question. ... This won’t be possible until long after the pandemic is completed, and so the model’s accuracy remains unknown.”
That sentiment was echoed by Giampaolo Bianchini, MD, head of the Breast Cancer Group, Department of Medical Oncology, Ospedale San Raffaele, in Milan, Italy.
“Arbitrarily postponing and modifying treatment strategies including surgery, radiation therapy, and medical therapy without properly balancing the risk/benefit ratio may lead to significantly worse cancer-related outcomes, which largely exceed the actual risks for COVID,” he wrote in an email.
“The OncCOVID app is a remarkable attempt to fill the gap between perception and estimation,” he said. The app provides side by side the COVID-19 risk estimation and the consequences of arbitrary deviation from the standard of care, observed Bianchini.
However, he pointed out weaknesses, including the fact that the “data generated in literature are not always of high quality and do not take into consideration relevant characteristics of the disease and treatment benefit. It should for sure be used, but then also interpreted with caution.”
Another Italian group responded more positively.
“In our opinion, it could be a useful tool for clinicians,” wrote colleagues Alessio Cortelinni and Giampiero Porzio, both medical oncologists at San Salvatore Hospital and the University of L’Aquila, in Italy. “This Web app might assist clinicians in balancing the risk/benefit ratio of being treated and/or access to the outpatient cancer center for each kind of patient (both early and advanced stages), in order to make a more tailored counseling,” they wrote in an email. “Importantly, the Web app might help those clinicians who work ‘alone,’ in peripheral centers, without resources, colleagues, and multidisciplinary tumor boards on whom they can rely.”
Bleicher, who was involved in the COVID-19 Breast Cancer Consortium’s recommendations for prioritizing breast cancer treatment, summarized that the app “may end up being close or accurate, but we won’t know except in hindsight.”
This article first appeared on Medscape.com.
TKI plus SBRT tops TKI alone for oligometastatic EGFRm NSCLC
Adding aggressive local radiotherapy to treatment with tyrosine kinase inhibitor (TKI) significantly improved progression-free and overall survival in patients with previously untreated, EGFR-mutated, oligometastatic non–small cell lung cancer (NSCLC) in a phase 3 trial presented as part of the American Society of Clinical Oncology virtual scientific program.
Sixty-eight patients were randomized at diagnosis to receive a first-generation TKI plus stereotactic body radiation therapy (SBRT) to all disease sites. The other 68 patients were randomized to receive a TKI alone, but 3 patients were lost follow-up and not included in the analysis. The TKIs used were gefitinib, erlotinib, and icotinib.
At baseline, patients had a maximum of two lesions in any one organ and no more than five metastases overall. Patients with brain metastases were excluded.
The median progression-free survival was 20.2 months in the SBRT arm and 12.5 months in the TKI-only arm (hazard ratio, 0.618; P < .001). The median overall survival was 25.5 months and 17.4 months, respectively (HR, 0.682; P < .001).
There were no grade 4/5 adverse events nor any statistically significant between-group differences in grade 3 events.
‘Compelling’ data with caveats
The study results suggest that “aggressive local therapy to sites at diagnosis should be explored further in large cohort phase 3 trials as a standard treatment option in this clinical scenario,” said investigator Xiao-shan Wang, MD, PhD, of the University of Electronic Science and Technology of China and Sichuan Provincial People’s Hospital in Chengdu.
“The data are compelling,” Dr. Wang added. “As we attempt to maximize the benefits of EGFR-directed targeted therapies, we are likely going to be moving away from a sequentially administered approach to treatment and considering combinations.”
The new findings, combined with prior phase 2 results, support “incorporation of upfront SBRT with TKI into practice for selected patients with oligometastatic disease, with the open question remaining of how many metastases are too many,” said study discussant Rachel Sanborn, MD, of Providence Cancer Institute Franz Clinic in Portland, Ore.
However, “it’s important to make note of the baseline characteristics of the patients enrolled,” she said.
Twelve percent of patients in the control arm and 4% of those in the SBRT group had EGFR exon 20 insertions. This “imbalance could have negatively impacted the outcomes in the TKI-alone arm,” Dr. Sanborn said.
Also, a higher proportion of patients in the TKI-alone arm received gefitinib, and “there was no information offered on second-line therapies in the study, which might have also affected outcomes,” Dr. Sanborn added.
Additional details
The study (NCT02893332) enrolled NSCLC patients with a life expectancy of at least 6 months and an Eastern Cooperative Oncology Group performance status score of 0-2. NSCLC was confirmed by pathology and EGFR mutations by gene sequencing.
The radiation dose was 25-40 Gy in five fractions. Gefitinib was used in 47% of patients in the SBRT arm and 58% of the control group. Erlotinib was used in 44% of the SBRT arm and 35% of controls. Icotinib was used by less than 10% of patients in each group.
Grade 3 skin rash occurred in 50% of patients in the SBRT arm and 62% of those in the TKI-alone arm. Grade 3 pneumonitis occurred in 30% and 15%, respectively. Grade 3 esophagitis occurred in 15% of patients in both arms.
One patient in the TKI arm had severe liver injury. One patient in the SBRT arm fractured a rib, which was considered probably related to the radiation.
Multivariate analysis revealed that, in addition to SBRT, lower baseline performance status score (0 vs. 1-2) and fewer metastases (<2 vs. ≥3) were protective for progression-free survival. Lower performance scores, fewer metastases, lower T stage (T1-2 vs. T3-4), and exon 19 versus exon 20 and 21 mutations were protective for overall survival.
The study arms were well balanced at baseline. The mean patient age was 66.9 years in the SBRT arm and 63.32 years in the TKI-only arm. In both arms, most patients were women (63% and 60%, respectively).
The study was sponsored by Sichuan Provincial People’s Hospital. The investigators and Dr. Sanborn have no relevant disclosures.
aotto@mdedge.com
SOURCE: Wang X et al. ASCO 2020, Abstract 9508.
Adding aggressive local radiotherapy to treatment with tyrosine kinase inhibitor (TKI) significantly improved progression-free and overall survival in patients with previously untreated, EGFR-mutated, oligometastatic non–small cell lung cancer (NSCLC) in a phase 3 trial presented as part of the American Society of Clinical Oncology virtual scientific program.
Sixty-eight patients were randomized at diagnosis to receive a first-generation TKI plus stereotactic body radiation therapy (SBRT) to all disease sites. The other 68 patients were randomized to receive a TKI alone, but 3 patients were lost follow-up and not included in the analysis. The TKIs used were gefitinib, erlotinib, and icotinib.
At baseline, patients had a maximum of two lesions in any one organ and no more than five metastases overall. Patients with brain metastases were excluded.
The median progression-free survival was 20.2 months in the SBRT arm and 12.5 months in the TKI-only arm (hazard ratio, 0.618; P < .001). The median overall survival was 25.5 months and 17.4 months, respectively (HR, 0.682; P < .001).
There were no grade 4/5 adverse events nor any statistically significant between-group differences in grade 3 events.
‘Compelling’ data with caveats
The study results suggest that “aggressive local therapy to sites at diagnosis should be explored further in large cohort phase 3 trials as a standard treatment option in this clinical scenario,” said investigator Xiao-shan Wang, MD, PhD, of the University of Electronic Science and Technology of China and Sichuan Provincial People’s Hospital in Chengdu.
“The data are compelling,” Dr. Wang added. “As we attempt to maximize the benefits of EGFR-directed targeted therapies, we are likely going to be moving away from a sequentially administered approach to treatment and considering combinations.”
The new findings, combined with prior phase 2 results, support “incorporation of upfront SBRT with TKI into practice for selected patients with oligometastatic disease, with the open question remaining of how many metastases are too many,” said study discussant Rachel Sanborn, MD, of Providence Cancer Institute Franz Clinic in Portland, Ore.
However, “it’s important to make note of the baseline characteristics of the patients enrolled,” she said.
Twelve percent of patients in the control arm and 4% of those in the SBRT group had EGFR exon 20 insertions. This “imbalance could have negatively impacted the outcomes in the TKI-alone arm,” Dr. Sanborn said.
Also, a higher proportion of patients in the TKI-alone arm received gefitinib, and “there was no information offered on second-line therapies in the study, which might have also affected outcomes,” Dr. Sanborn added.
Additional details
The study (NCT02893332) enrolled NSCLC patients with a life expectancy of at least 6 months and an Eastern Cooperative Oncology Group performance status score of 0-2. NSCLC was confirmed by pathology and EGFR mutations by gene sequencing.
The radiation dose was 25-40 Gy in five fractions. Gefitinib was used in 47% of patients in the SBRT arm and 58% of the control group. Erlotinib was used in 44% of the SBRT arm and 35% of controls. Icotinib was used by less than 10% of patients in each group.
Grade 3 skin rash occurred in 50% of patients in the SBRT arm and 62% of those in the TKI-alone arm. Grade 3 pneumonitis occurred in 30% and 15%, respectively. Grade 3 esophagitis occurred in 15% of patients in both arms.
One patient in the TKI arm had severe liver injury. One patient in the SBRT arm fractured a rib, which was considered probably related to the radiation.
Multivariate analysis revealed that, in addition to SBRT, lower baseline performance status score (0 vs. 1-2) and fewer metastases (<2 vs. ≥3) were protective for progression-free survival. Lower performance scores, fewer metastases, lower T stage (T1-2 vs. T3-4), and exon 19 versus exon 20 and 21 mutations were protective for overall survival.
The study arms were well balanced at baseline. The mean patient age was 66.9 years in the SBRT arm and 63.32 years in the TKI-only arm. In both arms, most patients were women (63% and 60%, respectively).
The study was sponsored by Sichuan Provincial People’s Hospital. The investigators and Dr. Sanborn have no relevant disclosures.
aotto@mdedge.com
SOURCE: Wang X et al. ASCO 2020, Abstract 9508.
Adding aggressive local radiotherapy to treatment with tyrosine kinase inhibitor (TKI) significantly improved progression-free and overall survival in patients with previously untreated, EGFR-mutated, oligometastatic non–small cell lung cancer (NSCLC) in a phase 3 trial presented as part of the American Society of Clinical Oncology virtual scientific program.
Sixty-eight patients were randomized at diagnosis to receive a first-generation TKI plus stereotactic body radiation therapy (SBRT) to all disease sites. The other 68 patients were randomized to receive a TKI alone, but 3 patients were lost follow-up and not included in the analysis. The TKIs used were gefitinib, erlotinib, and icotinib.
At baseline, patients had a maximum of two lesions in any one organ and no more than five metastases overall. Patients with brain metastases were excluded.
The median progression-free survival was 20.2 months in the SBRT arm and 12.5 months in the TKI-only arm (hazard ratio, 0.618; P < .001). The median overall survival was 25.5 months and 17.4 months, respectively (HR, 0.682; P < .001).
There were no grade 4/5 adverse events nor any statistically significant between-group differences in grade 3 events.
‘Compelling’ data with caveats
The study results suggest that “aggressive local therapy to sites at diagnosis should be explored further in large cohort phase 3 trials as a standard treatment option in this clinical scenario,” said investigator Xiao-shan Wang, MD, PhD, of the University of Electronic Science and Technology of China and Sichuan Provincial People’s Hospital in Chengdu.
“The data are compelling,” Dr. Wang added. “As we attempt to maximize the benefits of EGFR-directed targeted therapies, we are likely going to be moving away from a sequentially administered approach to treatment and considering combinations.”
The new findings, combined with prior phase 2 results, support “incorporation of upfront SBRT with TKI into practice for selected patients with oligometastatic disease, with the open question remaining of how many metastases are too many,” said study discussant Rachel Sanborn, MD, of Providence Cancer Institute Franz Clinic in Portland, Ore.
However, “it’s important to make note of the baseline characteristics of the patients enrolled,” she said.
Twelve percent of patients in the control arm and 4% of those in the SBRT group had EGFR exon 20 insertions. This “imbalance could have negatively impacted the outcomes in the TKI-alone arm,” Dr. Sanborn said.
Also, a higher proportion of patients in the TKI-alone arm received gefitinib, and “there was no information offered on second-line therapies in the study, which might have also affected outcomes,” Dr. Sanborn added.
Additional details
The study (NCT02893332) enrolled NSCLC patients with a life expectancy of at least 6 months and an Eastern Cooperative Oncology Group performance status score of 0-2. NSCLC was confirmed by pathology and EGFR mutations by gene sequencing.
The radiation dose was 25-40 Gy in five fractions. Gefitinib was used in 47% of patients in the SBRT arm and 58% of the control group. Erlotinib was used in 44% of the SBRT arm and 35% of controls. Icotinib was used by less than 10% of patients in each group.
Grade 3 skin rash occurred in 50% of patients in the SBRT arm and 62% of those in the TKI-alone arm. Grade 3 pneumonitis occurred in 30% and 15%, respectively. Grade 3 esophagitis occurred in 15% of patients in both arms.
One patient in the TKI arm had severe liver injury. One patient in the SBRT arm fractured a rib, which was considered probably related to the radiation.
Multivariate analysis revealed that, in addition to SBRT, lower baseline performance status score (0 vs. 1-2) and fewer metastases (<2 vs. ≥3) were protective for progression-free survival. Lower performance scores, fewer metastases, lower T stage (T1-2 vs. T3-4), and exon 19 versus exon 20 and 21 mutations were protective for overall survival.
The study arms were well balanced at baseline. The mean patient age was 66.9 years in the SBRT arm and 63.32 years in the TKI-only arm. In both arms, most patients were women (63% and 60%, respectively).
The study was sponsored by Sichuan Provincial People’s Hospital. The investigators and Dr. Sanborn have no relevant disclosures.
aotto@mdedge.com
SOURCE: Wang X et al. ASCO 2020, Abstract 9508.
FROM ASCO 2020
‘A good and peaceful death’: Cancer hospice during the pandemic
Lillie Shockney, RN, MAS, a two-time breast cancer survivor and adjunct professor at Johns Hopkins School of Nursing in Baltimore, Maryland, mourns the many losses that her patients with advanced cancer now face in the midst of the COVID-19 pandemic. But in the void of the usual support networks and treatment plans, she sees the resurgence of something that has recently been crowded out: hospice.
The pandemic has forced patients and their physicians to reassess the risk/benefit balance of continuing or embarking on yet another cancer treatment.
“It’s one of the pearls that we will get out of this nightmare,” said Ms. Shockney, who recently retired as administrative director of the cancer survivorship programs at the Sidney Kimmel Comprehensive Cancer Center.
“Physicians have been taught to treat the disease – so as long as there’s a treatment they give another treatment,” she told Medscape Medical News during a Zoom call from her home. “But for some patients with advanced disease, those treatments were making them very sick, so they were trading longevity over quality of life.”
Of course, longevity has never been a guarantee with cancer treatment, and even less so now, with the risk of COVID-19.
“This is going to bring them to some hard discussions,” says Brenda Nevidjon, RN, MSN, chief executive officer at the Oncology Nursing Society.
“We’ve known for a long time that there are patients who are on third- and fourth-round treatment options that have very little evidence of prolonging life or quality of life,” she told Medscape Medical News. “Do we bring these people out of their home to a setting where there could be a fair number of COVID-positive patients? Do we expose them to that?”
Across the world, these dilemmas are pushing cancer specialists to initiate discussions of hospice sooner with patients who have advanced disease, and with more clarity than before.
One of the reasons such conversations have often been avoided is that the concept of hospice is generally misunderstood, said Ms. Shockney.
“Patients think ‘you’re giving up on me, you’ve abandoned me’, but hospice is all about preserving the remainder of their quality of life and letting them have time with family and time to fulfill those elements of experiencing a good and peaceful death,” she said.
Indeed, hospice is “a benefit meant for somebody with at least a 6-month horizon,” agrees Ms. Nevidjon. Yet the average length of hospice in the United States is just 5 days. “It’s at the very, very end, and yet for some of these patients the 6 months they could get in hospice might be a better quality of life than the 4 months on another whole plan of chemotherapy. I can’t imagine that on the backside of this pandemic we will not have learned and we won’t start to change practices around initiating more of these conversations.”
Silver lining of this pandemic?
It’s too early into the pandemic to have hard data on whether hospice uptake has increased, but “it’s encouraging to hear that hospice is being discussed and offered sooner as an alternative to that third- or fourth-round chemo,” said Lori Bishop, MHA, RN, vice president of palliative and advanced care at the National Hospice and Palliative Care Organization.
“I agree that improving informed-decision discussions and timely access to hospice is a silver lining of the pandemic,” she told Medscape Medical News.
But she points out that today’s hospice looks quite different than it did before the pandemic, with the immediate and very obvious difference being telehealth, which was not widely utilized previously.
In March, the Centers for Medicare & Medicaid Services expanded telehealth options for hospice providers, something that Ms. Bishop and other hospice providers hope will remain in place after the pandemic passes.
“Telehealth visits are offered to replace some in-home visits both to minimize risk of exposure to COVID-19 and reduce the drain on personal protective equipment,” Bishop explained.
“In-patient hospice programs are also finding unique ways to provide support and connect patients to their loved ones: visitors are allowed but limited to one or two. Music and pet therapy are being provided through the window or virtually and devices such as iPads are being used to help patients connect with loved ones,” she said.
Telehealth links patients out of loneliness, but the one thing it cannot do is provide the comfort of touch – an important part of any hospice program.
“Hand-holding ... I miss that a lot,” says Ms. Shockney, her eyes filling with tears. “When you take somebody’s hand, you don’t even have to speak; that connection, and eye contact, is all you need to help that person emotionally heal.”
This article first appeared on Medscape.com.
Lillie Shockney, RN, MAS, a two-time breast cancer survivor and adjunct professor at Johns Hopkins School of Nursing in Baltimore, Maryland, mourns the many losses that her patients with advanced cancer now face in the midst of the COVID-19 pandemic. But in the void of the usual support networks and treatment plans, she sees the resurgence of something that has recently been crowded out: hospice.
The pandemic has forced patients and their physicians to reassess the risk/benefit balance of continuing or embarking on yet another cancer treatment.
“It’s one of the pearls that we will get out of this nightmare,” said Ms. Shockney, who recently retired as administrative director of the cancer survivorship programs at the Sidney Kimmel Comprehensive Cancer Center.
“Physicians have been taught to treat the disease – so as long as there’s a treatment they give another treatment,” she told Medscape Medical News during a Zoom call from her home. “But for some patients with advanced disease, those treatments were making them very sick, so they were trading longevity over quality of life.”
Of course, longevity has never been a guarantee with cancer treatment, and even less so now, with the risk of COVID-19.
“This is going to bring them to some hard discussions,” says Brenda Nevidjon, RN, MSN, chief executive officer at the Oncology Nursing Society.
“We’ve known for a long time that there are patients who are on third- and fourth-round treatment options that have very little evidence of prolonging life or quality of life,” she told Medscape Medical News. “Do we bring these people out of their home to a setting where there could be a fair number of COVID-positive patients? Do we expose them to that?”
Across the world, these dilemmas are pushing cancer specialists to initiate discussions of hospice sooner with patients who have advanced disease, and with more clarity than before.
One of the reasons such conversations have often been avoided is that the concept of hospice is generally misunderstood, said Ms. Shockney.
“Patients think ‘you’re giving up on me, you’ve abandoned me’, but hospice is all about preserving the remainder of their quality of life and letting them have time with family and time to fulfill those elements of experiencing a good and peaceful death,” she said.
Indeed, hospice is “a benefit meant for somebody with at least a 6-month horizon,” agrees Ms. Nevidjon. Yet the average length of hospice in the United States is just 5 days. “It’s at the very, very end, and yet for some of these patients the 6 months they could get in hospice might be a better quality of life than the 4 months on another whole plan of chemotherapy. I can’t imagine that on the backside of this pandemic we will not have learned and we won’t start to change practices around initiating more of these conversations.”
Silver lining of this pandemic?
It’s too early into the pandemic to have hard data on whether hospice uptake has increased, but “it’s encouraging to hear that hospice is being discussed and offered sooner as an alternative to that third- or fourth-round chemo,” said Lori Bishop, MHA, RN, vice president of palliative and advanced care at the National Hospice and Palliative Care Organization.
“I agree that improving informed-decision discussions and timely access to hospice is a silver lining of the pandemic,” she told Medscape Medical News.
But she points out that today’s hospice looks quite different than it did before the pandemic, with the immediate and very obvious difference being telehealth, which was not widely utilized previously.
In March, the Centers for Medicare & Medicaid Services expanded telehealth options for hospice providers, something that Ms. Bishop and other hospice providers hope will remain in place after the pandemic passes.
“Telehealth visits are offered to replace some in-home visits both to minimize risk of exposure to COVID-19 and reduce the drain on personal protective equipment,” Bishop explained.
“In-patient hospice programs are also finding unique ways to provide support and connect patients to their loved ones: visitors are allowed but limited to one or two. Music and pet therapy are being provided through the window or virtually and devices such as iPads are being used to help patients connect with loved ones,” she said.
Telehealth links patients out of loneliness, but the one thing it cannot do is provide the comfort of touch – an important part of any hospice program.
“Hand-holding ... I miss that a lot,” says Ms. Shockney, her eyes filling with tears. “When you take somebody’s hand, you don’t even have to speak; that connection, and eye contact, is all you need to help that person emotionally heal.”
This article first appeared on Medscape.com.
Lillie Shockney, RN, MAS, a two-time breast cancer survivor and adjunct professor at Johns Hopkins School of Nursing in Baltimore, Maryland, mourns the many losses that her patients with advanced cancer now face in the midst of the COVID-19 pandemic. But in the void of the usual support networks and treatment plans, she sees the resurgence of something that has recently been crowded out: hospice.
The pandemic has forced patients and their physicians to reassess the risk/benefit balance of continuing or embarking on yet another cancer treatment.
“It’s one of the pearls that we will get out of this nightmare,” said Ms. Shockney, who recently retired as administrative director of the cancer survivorship programs at the Sidney Kimmel Comprehensive Cancer Center.
“Physicians have been taught to treat the disease – so as long as there’s a treatment they give another treatment,” she told Medscape Medical News during a Zoom call from her home. “But for some patients with advanced disease, those treatments were making them very sick, so they were trading longevity over quality of life.”
Of course, longevity has never been a guarantee with cancer treatment, and even less so now, with the risk of COVID-19.
“This is going to bring them to some hard discussions,” says Brenda Nevidjon, RN, MSN, chief executive officer at the Oncology Nursing Society.
“We’ve known for a long time that there are patients who are on third- and fourth-round treatment options that have very little evidence of prolonging life or quality of life,” she told Medscape Medical News. “Do we bring these people out of their home to a setting where there could be a fair number of COVID-positive patients? Do we expose them to that?”
Across the world, these dilemmas are pushing cancer specialists to initiate discussions of hospice sooner with patients who have advanced disease, and with more clarity than before.
One of the reasons such conversations have often been avoided is that the concept of hospice is generally misunderstood, said Ms. Shockney.
“Patients think ‘you’re giving up on me, you’ve abandoned me’, but hospice is all about preserving the remainder of their quality of life and letting them have time with family and time to fulfill those elements of experiencing a good and peaceful death,” she said.
Indeed, hospice is “a benefit meant for somebody with at least a 6-month horizon,” agrees Ms. Nevidjon. Yet the average length of hospice in the United States is just 5 days. “It’s at the very, very end, and yet for some of these patients the 6 months they could get in hospice might be a better quality of life than the 4 months on another whole plan of chemotherapy. I can’t imagine that on the backside of this pandemic we will not have learned and we won’t start to change practices around initiating more of these conversations.”
Silver lining of this pandemic?
It’s too early into the pandemic to have hard data on whether hospice uptake has increased, but “it’s encouraging to hear that hospice is being discussed and offered sooner as an alternative to that third- or fourth-round chemo,” said Lori Bishop, MHA, RN, vice president of palliative and advanced care at the National Hospice and Palliative Care Organization.
“I agree that improving informed-decision discussions and timely access to hospice is a silver lining of the pandemic,” she told Medscape Medical News.
But she points out that today’s hospice looks quite different than it did before the pandemic, with the immediate and very obvious difference being telehealth, which was not widely utilized previously.
In March, the Centers for Medicare & Medicaid Services expanded telehealth options for hospice providers, something that Ms. Bishop and other hospice providers hope will remain in place after the pandemic passes.
“Telehealth visits are offered to replace some in-home visits both to minimize risk of exposure to COVID-19 and reduce the drain on personal protective equipment,” Bishop explained.
“In-patient hospice programs are also finding unique ways to provide support and connect patients to their loved ones: visitors are allowed but limited to one or two. Music and pet therapy are being provided through the window or virtually and devices such as iPads are being used to help patients connect with loved ones,” she said.
Telehealth links patients out of loneliness, but the one thing it cannot do is provide the comfort of touch – an important part of any hospice program.
“Hand-holding ... I miss that a lot,” says Ms. Shockney, her eyes filling with tears. “When you take somebody’s hand, you don’t even have to speak; that connection, and eye contact, is all you need to help that person emotionally heal.”
This article first appeared on Medscape.com.
Pembrolizumab plus EP gives slight PFS edge in ES-SCLC
Adding the immune checkpoint inhibitor pembrolizumab to chemotherapy resulted in a modest improvement in progression-free survival (PFS) but no overall survival (OS) benefit as first-line therapy for patients with extensive-stage small cell lung cancer (ES-SCLC), results of the KEYNOTE-604 study showed.
Among 453 patients with ES-SCLC randomized to receive pembrolizumab plus etoposide and a platinum agent (EP) or placebo, the median PFS was 4.5 months with pembrolizumab and with 4.3 months with placebo.
This difference, although small, met the prespecified definition for significance, with a hazard ratio favoring pembrolizumab of 0.75 (P = .0023), reported Charles M. Rudin, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
Median OS, the other primary endpoint, was 10.8 months for patients who received pembrolizumab and 9.7 months for those who received placebo. Although this translated to a hazard ratio of 0.80 for pembrolizumab, the P value of .0164 missed the prespecified threshold of .0128 and was therefore not statistically significant.
Dr. Rudin presented these results as part of the American Society of Clinical Oncology virtual scientific program. The study was also published online to coincide with the abstract’s release in the Journal of Clinical Oncology.
Beneficial but not practice-changing (yet)
“The addition of pembrolizumab results in durable responses in a subset of patients,” Dr. Rudin said. “I believe additional correlative analyses may help to identify those patients who derive long-term benefit from pembrolizumab.
“The safety profile was manageable with no new or unexpected toxicities. Taken together, these data support the benefit of pembrolizumab in patients with small cell lung cancer and add to the growing body of evidence supporting the value of immune checkpoint inhibitors in a historically difficult-to-treat cancer.”
The results suggest combination pembrolizumab and chemotherapy offers a “viable platform for a novel treatment strategy,” said invited discussant Taofeek K. Owonikoko, MD, PhD, director of thoracic oncology at the Winship Cancer Institute of Emory University in Atlanta.
However, because the trial did not meet the predefined threshold for success, “the immediate impact on practice of this trial is limited at present, and any future impact will have to be supported by regulatory decision,” Dr. Owonikoko said.
“The outcome of this trial also highlights the need for an uncomplicated study design and straightforward analytical plan to ensure accurate results,” he added.
Study details
KEYNOTE-604 investigators enrolled 453 patients with ES-SCLC who had no prior systemic therapy, good performance status, and a life expectancy of at least 3 months. Patients were stratified by type of platinum agent (cisplatin vs. carboplatin), Eastern Cooperative Oncology Group performance status 0 or 1, and lactate dehydrogenase levels below or above the upper limit of normal.
Patients were randomized to receive pembrolizumab at 200 mg or normal saline placebo on day 1. Both arms also received etoposide at 100 mg/m2 on days 1-3 and investigator’s choice of carboplatin to an area under the curve of 5 on day 1 or cisplatin at 75 mg/m2 on day 1 for four cycles every 3 weeks.
The assigned agent (pembrolizumab or placebo) could then be continued as maintenance therapy for up to 31 cycles every 21 days.
Patients with a complete or partial response after cycle 4 could receive up to 25 Gy of prophylactic cranial irradiation delivered over 10 fractions at the investigator’s discretion.
Survival and response
As noted, the median PFS was modestly but significantly longer with pembrolizumab plus EP at the second interim analysis, the protocol-specified time for final PFS analysis. The estimated 12-months PFS rates were 13.6% with pembrolizumab plus EP and 3.1% with placebo plus EP.
The final analysis was planned to occur about 31 months after the start of the study or when 284 deaths had occurred, whichever was later. At the final analysis, the median PFS was 4.8 months in the pembrolizumab arm and 4.3 months in the placebo arm. The hazard ratio was 0.73 (95% confidence interval 0.60-0.88).
The 12-month OS rate was 45.1% in the pembrolizumab arm and 39.6% in the placebo arm. Respective 24-month OS rates were 22.5% and 11.2%.
Overall responses rates were 70.6% in the pembrolizumab arm and 61.8% in the placebo arm. There were four and two complete responses per arm, respectively.
Safety
Approximately 75% of patients in both arms experienced grade 3 or 4 adverse events.
Fatal adverse events occurred in 6.3% of patients in the pembrolizumab arm and 5.4% in the control arm. The rates of death attributed to study treatment were identical, at 2.7% in each arm.
Events leading to discontinuation occurred in 14.8% of patients who received pembrolizumab and 6.3% of patients who received placebo. Adverse events leading to all treatment discontinuation were similar, at 4% and 3.6%, respectively.
The most common adverse events were hematologic, which are common with EP chemotherapy and did not appear to be exacerbated by the addition of pembrolizumab. Aside from hematologic toxicities, most events were of grade 1 or 2 severity.
Immune-mediated adverse events of any kind occurred in 24.7% of patients in the pembrolizumab arm and 10.3% of those in the placebo arm. Grade 3 or 4 immune-mediated events occurred in 7.2% and 1.3%, respectively.
There were no deaths from immune-mediated reactions in the pembrolizumab arm, but one patient on placebo died from pneumonia.
Merck Sharp & Dohme supported the study. Dr. Rudin disclosed institutional research funding from Merck and a consulting or advisory role for other companies. Dr. Owonikoko disclosed a consulting/advisory role and institutional research funding from Merck and others, and he is a cofounder and stock owner in Cambium Oncology.
SOURCE: Rudin CM et al. ASCO 2020. Abstract 9001.
Adding the immune checkpoint inhibitor pembrolizumab to chemotherapy resulted in a modest improvement in progression-free survival (PFS) but no overall survival (OS) benefit as first-line therapy for patients with extensive-stage small cell lung cancer (ES-SCLC), results of the KEYNOTE-604 study showed.
Among 453 patients with ES-SCLC randomized to receive pembrolizumab plus etoposide and a platinum agent (EP) or placebo, the median PFS was 4.5 months with pembrolizumab and with 4.3 months with placebo.
This difference, although small, met the prespecified definition for significance, with a hazard ratio favoring pembrolizumab of 0.75 (P = .0023), reported Charles M. Rudin, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
Median OS, the other primary endpoint, was 10.8 months for patients who received pembrolizumab and 9.7 months for those who received placebo. Although this translated to a hazard ratio of 0.80 for pembrolizumab, the P value of .0164 missed the prespecified threshold of .0128 and was therefore not statistically significant.
Dr. Rudin presented these results as part of the American Society of Clinical Oncology virtual scientific program. The study was also published online to coincide with the abstract’s release in the Journal of Clinical Oncology.
Beneficial but not practice-changing (yet)
“The addition of pembrolizumab results in durable responses in a subset of patients,” Dr. Rudin said. “I believe additional correlative analyses may help to identify those patients who derive long-term benefit from pembrolizumab.
“The safety profile was manageable with no new or unexpected toxicities. Taken together, these data support the benefit of pembrolizumab in patients with small cell lung cancer and add to the growing body of evidence supporting the value of immune checkpoint inhibitors in a historically difficult-to-treat cancer.”
The results suggest combination pembrolizumab and chemotherapy offers a “viable platform for a novel treatment strategy,” said invited discussant Taofeek K. Owonikoko, MD, PhD, director of thoracic oncology at the Winship Cancer Institute of Emory University in Atlanta.
However, because the trial did not meet the predefined threshold for success, “the immediate impact on practice of this trial is limited at present, and any future impact will have to be supported by regulatory decision,” Dr. Owonikoko said.
“The outcome of this trial also highlights the need for an uncomplicated study design and straightforward analytical plan to ensure accurate results,” he added.
Study details
KEYNOTE-604 investigators enrolled 453 patients with ES-SCLC who had no prior systemic therapy, good performance status, and a life expectancy of at least 3 months. Patients were stratified by type of platinum agent (cisplatin vs. carboplatin), Eastern Cooperative Oncology Group performance status 0 or 1, and lactate dehydrogenase levels below or above the upper limit of normal.
Patients were randomized to receive pembrolizumab at 200 mg or normal saline placebo on day 1. Both arms also received etoposide at 100 mg/m2 on days 1-3 and investigator’s choice of carboplatin to an area under the curve of 5 on day 1 or cisplatin at 75 mg/m2 on day 1 for four cycles every 3 weeks.
The assigned agent (pembrolizumab or placebo) could then be continued as maintenance therapy for up to 31 cycles every 21 days.
Patients with a complete or partial response after cycle 4 could receive up to 25 Gy of prophylactic cranial irradiation delivered over 10 fractions at the investigator’s discretion.
Survival and response
As noted, the median PFS was modestly but significantly longer with pembrolizumab plus EP at the second interim analysis, the protocol-specified time for final PFS analysis. The estimated 12-months PFS rates were 13.6% with pembrolizumab plus EP and 3.1% with placebo plus EP.
The final analysis was planned to occur about 31 months after the start of the study or when 284 deaths had occurred, whichever was later. At the final analysis, the median PFS was 4.8 months in the pembrolizumab arm and 4.3 months in the placebo arm. The hazard ratio was 0.73 (95% confidence interval 0.60-0.88).
The 12-month OS rate was 45.1% in the pembrolizumab arm and 39.6% in the placebo arm. Respective 24-month OS rates were 22.5% and 11.2%.
Overall responses rates were 70.6% in the pembrolizumab arm and 61.8% in the placebo arm. There were four and two complete responses per arm, respectively.
Safety
Approximately 75% of patients in both arms experienced grade 3 or 4 adverse events.
Fatal adverse events occurred in 6.3% of patients in the pembrolizumab arm and 5.4% in the control arm. The rates of death attributed to study treatment were identical, at 2.7% in each arm.
Events leading to discontinuation occurred in 14.8% of patients who received pembrolizumab and 6.3% of patients who received placebo. Adverse events leading to all treatment discontinuation were similar, at 4% and 3.6%, respectively.
The most common adverse events were hematologic, which are common with EP chemotherapy and did not appear to be exacerbated by the addition of pembrolizumab. Aside from hematologic toxicities, most events were of grade 1 or 2 severity.
Immune-mediated adverse events of any kind occurred in 24.7% of patients in the pembrolizumab arm and 10.3% of those in the placebo arm. Grade 3 or 4 immune-mediated events occurred in 7.2% and 1.3%, respectively.
There were no deaths from immune-mediated reactions in the pembrolizumab arm, but one patient on placebo died from pneumonia.
Merck Sharp & Dohme supported the study. Dr. Rudin disclosed institutional research funding from Merck and a consulting or advisory role for other companies. Dr. Owonikoko disclosed a consulting/advisory role and institutional research funding from Merck and others, and he is a cofounder and stock owner in Cambium Oncology.
SOURCE: Rudin CM et al. ASCO 2020. Abstract 9001.
Adding the immune checkpoint inhibitor pembrolizumab to chemotherapy resulted in a modest improvement in progression-free survival (PFS) but no overall survival (OS) benefit as first-line therapy for patients with extensive-stage small cell lung cancer (ES-SCLC), results of the KEYNOTE-604 study showed.
Among 453 patients with ES-SCLC randomized to receive pembrolizumab plus etoposide and a platinum agent (EP) or placebo, the median PFS was 4.5 months with pembrolizumab and with 4.3 months with placebo.
This difference, although small, met the prespecified definition for significance, with a hazard ratio favoring pembrolizumab of 0.75 (P = .0023), reported Charles M. Rudin, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
Median OS, the other primary endpoint, was 10.8 months for patients who received pembrolizumab and 9.7 months for those who received placebo. Although this translated to a hazard ratio of 0.80 for pembrolizumab, the P value of .0164 missed the prespecified threshold of .0128 and was therefore not statistically significant.
Dr. Rudin presented these results as part of the American Society of Clinical Oncology virtual scientific program. The study was also published online to coincide with the abstract’s release in the Journal of Clinical Oncology.
Beneficial but not practice-changing (yet)
“The addition of pembrolizumab results in durable responses in a subset of patients,” Dr. Rudin said. “I believe additional correlative analyses may help to identify those patients who derive long-term benefit from pembrolizumab.
“The safety profile was manageable with no new or unexpected toxicities. Taken together, these data support the benefit of pembrolizumab in patients with small cell lung cancer and add to the growing body of evidence supporting the value of immune checkpoint inhibitors in a historically difficult-to-treat cancer.”
The results suggest combination pembrolizumab and chemotherapy offers a “viable platform for a novel treatment strategy,” said invited discussant Taofeek K. Owonikoko, MD, PhD, director of thoracic oncology at the Winship Cancer Institute of Emory University in Atlanta.
However, because the trial did not meet the predefined threshold for success, “the immediate impact on practice of this trial is limited at present, and any future impact will have to be supported by regulatory decision,” Dr. Owonikoko said.
“The outcome of this trial also highlights the need for an uncomplicated study design and straightforward analytical plan to ensure accurate results,” he added.
Study details
KEYNOTE-604 investigators enrolled 453 patients with ES-SCLC who had no prior systemic therapy, good performance status, and a life expectancy of at least 3 months. Patients were stratified by type of platinum agent (cisplatin vs. carboplatin), Eastern Cooperative Oncology Group performance status 0 or 1, and lactate dehydrogenase levels below or above the upper limit of normal.
Patients were randomized to receive pembrolizumab at 200 mg or normal saline placebo on day 1. Both arms also received etoposide at 100 mg/m2 on days 1-3 and investigator’s choice of carboplatin to an area under the curve of 5 on day 1 or cisplatin at 75 mg/m2 on day 1 for four cycles every 3 weeks.
The assigned agent (pembrolizumab or placebo) could then be continued as maintenance therapy for up to 31 cycles every 21 days.
Patients with a complete or partial response after cycle 4 could receive up to 25 Gy of prophylactic cranial irradiation delivered over 10 fractions at the investigator’s discretion.
Survival and response
As noted, the median PFS was modestly but significantly longer with pembrolizumab plus EP at the second interim analysis, the protocol-specified time for final PFS analysis. The estimated 12-months PFS rates were 13.6% with pembrolizumab plus EP and 3.1% with placebo plus EP.
The final analysis was planned to occur about 31 months after the start of the study or when 284 deaths had occurred, whichever was later. At the final analysis, the median PFS was 4.8 months in the pembrolizumab arm and 4.3 months in the placebo arm. The hazard ratio was 0.73 (95% confidence interval 0.60-0.88).
The 12-month OS rate was 45.1% in the pembrolizumab arm and 39.6% in the placebo arm. Respective 24-month OS rates were 22.5% and 11.2%.
Overall responses rates were 70.6% in the pembrolizumab arm and 61.8% in the placebo arm. There were four and two complete responses per arm, respectively.
Safety
Approximately 75% of patients in both arms experienced grade 3 or 4 adverse events.
Fatal adverse events occurred in 6.3% of patients in the pembrolizumab arm and 5.4% in the control arm. The rates of death attributed to study treatment were identical, at 2.7% in each arm.
Events leading to discontinuation occurred in 14.8% of patients who received pembrolizumab and 6.3% of patients who received placebo. Adverse events leading to all treatment discontinuation were similar, at 4% and 3.6%, respectively.
The most common adverse events were hematologic, which are common with EP chemotherapy and did not appear to be exacerbated by the addition of pembrolizumab. Aside from hematologic toxicities, most events were of grade 1 or 2 severity.
Immune-mediated adverse events of any kind occurred in 24.7% of patients in the pembrolizumab arm and 10.3% of those in the placebo arm. Grade 3 or 4 immune-mediated events occurred in 7.2% and 1.3%, respectively.
There were no deaths from immune-mediated reactions in the pembrolizumab arm, but one patient on placebo died from pneumonia.
Merck Sharp & Dohme supported the study. Dr. Rudin disclosed institutional research funding from Merck and a consulting or advisory role for other companies. Dr. Owonikoko disclosed a consulting/advisory role and institutional research funding from Merck and others, and he is a cofounder and stock owner in Cambium Oncology.
SOURCE: Rudin CM et al. ASCO 2020. Abstract 9001.
FROM ASCO 2020
‘Promising’ durvalumab results spark phase 3 trial in mesothelioma
Adding durvalumab to first-line pemetrexed and cisplatin improved survival in patients with unresectable malignant pleural mesothelioma (MPM) in a phase 2 trial, compared with historical controls who received only pemetrexed and cisplatin.
The median overall survival was 20.4 months in patients who received durvalumab plus pemetrexed-cisplatin. This is significantly longer than the median overall survival of 12.1 months (P = .0014) observed with pemetrexed-cisplatin in a prior phase 3 study (J Clin Oncol. 2003 Jul 15;21[14]:2636-44).
The new phase 2 results are “promising,” said lead investigator Patrick Forde, MBBCh, director of the thoracic cancer clinical research program at Johns Hopkins University in Baltimore.
He presented the results as part of the American Society of Clinical Oncology virtual scientific program.
Dr. Forde noted that a phase 3 trial directly comparing pemetrexed-cisplatin plus durvalumab to pemetrexed-cisplatin will begin recruiting this year. The trial is a collaboration between U.S. investigators and Australian researchers who reported their own phase 2 results with durvalumab plus pemetrexed-cisplatin in 2018 (J Thorac Oncol. 2018 Oct;13[10]:S338-339).
Study details
Dr. Forde’s phase 2 study enrolled 55 patients with treatment-naive, unresectable MPM. Their median age was 68 years (range, 35-83 years), and 45 (82%) were men. All had an Eastern Cooperative Oncology Group performance status of 0-1.
Epithelioid mesothelioma was the histologic subtype in three-quarters of patients. “It was a fairly typical mesothelioma population,” Dr. Forde said.
The patients received durvalumab at 1,120 mg plus pemetrexed at 500 mg/m2 and cisplatin at 75 mg/m2 every 3 weeks for up to six cycles. Carboplatin was substituted when cisplatin was contraindicated or patients developed toxicities.
All but one patient had stable or responding disease on radiography and went on to durvalumab maintenance, also given at 1,120 mg every 3 weeks, for up to 1 year from study entry.
Results
Dr. Forde said this study had 90% power to detect a 58% improvement in median overall survival, from the 12.1 months seen in historical controls to 19 months, which was the goal of this study.
It was a positive study, he said, as the median overall survival was 20.4 months (P = .0014).
The overall survival rate was 87.2% at 6 months, 70.4% at 12 months, and 44.2% at 24 months. The progression-free survival rate was 69.1% at 6 months, 16.4% at 12 months, and 10.9% at 24 months.
The overall response rate was 56.4%, which comprised 31 partial responses. Forty percent of patients (n = 22) had stable disease. One patient had progressive disease, and one was not evaluable (1.8% each).
To help with future patient selection, the researchers looked for baseline biomarkers that predicted response. Tumor PD-L1 expression, tumor mutation burden, and other potential candidates haven’t worked out so far, but the work continues, Dr. Forde said.
He noted that many of the adverse events in this trial are those typically seen with platinum-based chemotherapy.
Grade 3/4 treatment-emergent adverse events included anemia (n = 14), fatigue (n = 4), decreased appetite (n = 1), and hypomagnesemia (n = 1).
The most common grade 1/2 adverse events of special interest were hypothyroidism (n = 7), rash (n = 5), pruritus (n = 3), AST elevation (n = 3), and hyperthyroidism (n = 3).
Putting the results in context
Given the role of inflammation in MPM, durvalumab is among several immunotherapies under investigation for the disease.
A phase 3 French trial showed MPM patients had a median overall survival of 18.8 months with pemetrexed-cisplatin plus bevacizumab versus 16.1 months with pemetrexed-cisplatin only (Lancet. 2016 Apr 2;387[10026]:1405-1414).
The higher overall survival in the French study’s pemetrexed-cisplatin arm, compared with the 2003 trial results, is likely due to the use of modern second-line options, said Marjorie Zauderer, MD, codirector of the mesothelioma program at Memorial Sloan Kettering Cancer Center in New York, who was the discussant for Dr. Forde’s presentation.
“I think the improvement in overall survival presented by Dr. Forde is potentially clinically meaningful,” she said, but it was “well within the 95% confidence interval” of the bevacizumab trial. Even so, “I look forward” to the phase 3 results, she said.
Dr. Zauderer also pointed out an April press release from Bristol Myers Squibb that reported improved survival over pemetrexed-cisplatin with two of the company’s immunotherapies, nivolumab and ipilimumab, not as additions but as replacement first-line therapy. However, the randomized trial data haven’t been released yet. “We are all eager to evaluate this option further,” she said.
AstraZeneca, maker of durvalumab, funded the current study. Dr. Forde is an adviser for the company and reported research funding. Dr. Zauderer reported a relationship with Roche, which markets bevacizumab through its subsidiary, Genentech. She also disclosed research funding from Bristol Myers Squibb.
SOURCE: Forde PM et al. ASCO 2020, Abstract 9003.
Adding durvalumab to first-line pemetrexed and cisplatin improved survival in patients with unresectable malignant pleural mesothelioma (MPM) in a phase 2 trial, compared with historical controls who received only pemetrexed and cisplatin.
The median overall survival was 20.4 months in patients who received durvalumab plus pemetrexed-cisplatin. This is significantly longer than the median overall survival of 12.1 months (P = .0014) observed with pemetrexed-cisplatin in a prior phase 3 study (J Clin Oncol. 2003 Jul 15;21[14]:2636-44).
The new phase 2 results are “promising,” said lead investigator Patrick Forde, MBBCh, director of the thoracic cancer clinical research program at Johns Hopkins University in Baltimore.
He presented the results as part of the American Society of Clinical Oncology virtual scientific program.
Dr. Forde noted that a phase 3 trial directly comparing pemetrexed-cisplatin plus durvalumab to pemetrexed-cisplatin will begin recruiting this year. The trial is a collaboration between U.S. investigators and Australian researchers who reported their own phase 2 results with durvalumab plus pemetrexed-cisplatin in 2018 (J Thorac Oncol. 2018 Oct;13[10]:S338-339).
Study details
Dr. Forde’s phase 2 study enrolled 55 patients with treatment-naive, unresectable MPM. Their median age was 68 years (range, 35-83 years), and 45 (82%) were men. All had an Eastern Cooperative Oncology Group performance status of 0-1.
Epithelioid mesothelioma was the histologic subtype in three-quarters of patients. “It was a fairly typical mesothelioma population,” Dr. Forde said.
The patients received durvalumab at 1,120 mg plus pemetrexed at 500 mg/m2 and cisplatin at 75 mg/m2 every 3 weeks for up to six cycles. Carboplatin was substituted when cisplatin was contraindicated or patients developed toxicities.
All but one patient had stable or responding disease on radiography and went on to durvalumab maintenance, also given at 1,120 mg every 3 weeks, for up to 1 year from study entry.
Results
Dr. Forde said this study had 90% power to detect a 58% improvement in median overall survival, from the 12.1 months seen in historical controls to 19 months, which was the goal of this study.
It was a positive study, he said, as the median overall survival was 20.4 months (P = .0014).
The overall survival rate was 87.2% at 6 months, 70.4% at 12 months, and 44.2% at 24 months. The progression-free survival rate was 69.1% at 6 months, 16.4% at 12 months, and 10.9% at 24 months.
The overall response rate was 56.4%, which comprised 31 partial responses. Forty percent of patients (n = 22) had stable disease. One patient had progressive disease, and one was not evaluable (1.8% each).
To help with future patient selection, the researchers looked for baseline biomarkers that predicted response. Tumor PD-L1 expression, tumor mutation burden, and other potential candidates haven’t worked out so far, but the work continues, Dr. Forde said.
He noted that many of the adverse events in this trial are those typically seen with platinum-based chemotherapy.
Grade 3/4 treatment-emergent adverse events included anemia (n = 14), fatigue (n = 4), decreased appetite (n = 1), and hypomagnesemia (n = 1).
The most common grade 1/2 adverse events of special interest were hypothyroidism (n = 7), rash (n = 5), pruritus (n = 3), AST elevation (n = 3), and hyperthyroidism (n = 3).
Putting the results in context
Given the role of inflammation in MPM, durvalumab is among several immunotherapies under investigation for the disease.
A phase 3 French trial showed MPM patients had a median overall survival of 18.8 months with pemetrexed-cisplatin plus bevacizumab versus 16.1 months with pemetrexed-cisplatin only (Lancet. 2016 Apr 2;387[10026]:1405-1414).
The higher overall survival in the French study’s pemetrexed-cisplatin arm, compared with the 2003 trial results, is likely due to the use of modern second-line options, said Marjorie Zauderer, MD, codirector of the mesothelioma program at Memorial Sloan Kettering Cancer Center in New York, who was the discussant for Dr. Forde’s presentation.
“I think the improvement in overall survival presented by Dr. Forde is potentially clinically meaningful,” she said, but it was “well within the 95% confidence interval” of the bevacizumab trial. Even so, “I look forward” to the phase 3 results, she said.
Dr. Zauderer also pointed out an April press release from Bristol Myers Squibb that reported improved survival over pemetrexed-cisplatin with two of the company’s immunotherapies, nivolumab and ipilimumab, not as additions but as replacement first-line therapy. However, the randomized trial data haven’t been released yet. “We are all eager to evaluate this option further,” she said.
AstraZeneca, maker of durvalumab, funded the current study. Dr. Forde is an adviser for the company and reported research funding. Dr. Zauderer reported a relationship with Roche, which markets bevacizumab through its subsidiary, Genentech. She also disclosed research funding from Bristol Myers Squibb.
SOURCE: Forde PM et al. ASCO 2020, Abstract 9003.
Adding durvalumab to first-line pemetrexed and cisplatin improved survival in patients with unresectable malignant pleural mesothelioma (MPM) in a phase 2 trial, compared with historical controls who received only pemetrexed and cisplatin.
The median overall survival was 20.4 months in patients who received durvalumab plus pemetrexed-cisplatin. This is significantly longer than the median overall survival of 12.1 months (P = .0014) observed with pemetrexed-cisplatin in a prior phase 3 study (J Clin Oncol. 2003 Jul 15;21[14]:2636-44).
The new phase 2 results are “promising,” said lead investigator Patrick Forde, MBBCh, director of the thoracic cancer clinical research program at Johns Hopkins University in Baltimore.
He presented the results as part of the American Society of Clinical Oncology virtual scientific program.
Dr. Forde noted that a phase 3 trial directly comparing pemetrexed-cisplatin plus durvalumab to pemetrexed-cisplatin will begin recruiting this year. The trial is a collaboration between U.S. investigators and Australian researchers who reported their own phase 2 results with durvalumab plus pemetrexed-cisplatin in 2018 (J Thorac Oncol. 2018 Oct;13[10]:S338-339).
Study details
Dr. Forde’s phase 2 study enrolled 55 patients with treatment-naive, unresectable MPM. Their median age was 68 years (range, 35-83 years), and 45 (82%) were men. All had an Eastern Cooperative Oncology Group performance status of 0-1.
Epithelioid mesothelioma was the histologic subtype in three-quarters of patients. “It was a fairly typical mesothelioma population,” Dr. Forde said.
The patients received durvalumab at 1,120 mg plus pemetrexed at 500 mg/m2 and cisplatin at 75 mg/m2 every 3 weeks for up to six cycles. Carboplatin was substituted when cisplatin was contraindicated or patients developed toxicities.
All but one patient had stable or responding disease on radiography and went on to durvalumab maintenance, also given at 1,120 mg every 3 weeks, for up to 1 year from study entry.
Results
Dr. Forde said this study had 90% power to detect a 58% improvement in median overall survival, from the 12.1 months seen in historical controls to 19 months, which was the goal of this study.
It was a positive study, he said, as the median overall survival was 20.4 months (P = .0014).
The overall survival rate was 87.2% at 6 months, 70.4% at 12 months, and 44.2% at 24 months. The progression-free survival rate was 69.1% at 6 months, 16.4% at 12 months, and 10.9% at 24 months.
The overall response rate was 56.4%, which comprised 31 partial responses. Forty percent of patients (n = 22) had stable disease. One patient had progressive disease, and one was not evaluable (1.8% each).
To help with future patient selection, the researchers looked for baseline biomarkers that predicted response. Tumor PD-L1 expression, tumor mutation burden, and other potential candidates haven’t worked out so far, but the work continues, Dr. Forde said.
He noted that many of the adverse events in this trial are those typically seen with platinum-based chemotherapy.
Grade 3/4 treatment-emergent adverse events included anemia (n = 14), fatigue (n = 4), decreased appetite (n = 1), and hypomagnesemia (n = 1).
The most common grade 1/2 adverse events of special interest were hypothyroidism (n = 7), rash (n = 5), pruritus (n = 3), AST elevation (n = 3), and hyperthyroidism (n = 3).
Putting the results in context
Given the role of inflammation in MPM, durvalumab is among several immunotherapies under investigation for the disease.
A phase 3 French trial showed MPM patients had a median overall survival of 18.8 months with pemetrexed-cisplatin plus bevacizumab versus 16.1 months with pemetrexed-cisplatin only (Lancet. 2016 Apr 2;387[10026]:1405-1414).
The higher overall survival in the French study’s pemetrexed-cisplatin arm, compared with the 2003 trial results, is likely due to the use of modern second-line options, said Marjorie Zauderer, MD, codirector of the mesothelioma program at Memorial Sloan Kettering Cancer Center in New York, who was the discussant for Dr. Forde’s presentation.
“I think the improvement in overall survival presented by Dr. Forde is potentially clinically meaningful,” she said, but it was “well within the 95% confidence interval” of the bevacizumab trial. Even so, “I look forward” to the phase 3 results, she said.
Dr. Zauderer also pointed out an April press release from Bristol Myers Squibb that reported improved survival over pemetrexed-cisplatin with two of the company’s immunotherapies, nivolumab and ipilimumab, not as additions but as replacement first-line therapy. However, the randomized trial data haven’t been released yet. “We are all eager to evaluate this option further,” she said.
AstraZeneca, maker of durvalumab, funded the current study. Dr. Forde is an adviser for the company and reported research funding. Dr. Zauderer reported a relationship with Roche, which markets bevacizumab through its subsidiary, Genentech. She also disclosed research funding from Bristol Myers Squibb.
SOURCE: Forde PM et al. ASCO 2020, Abstract 9003.
FROM ASCO 2020