User login
Lung cancer CT scan is chance for ‘opportunistic’ osteoporosis check
Low-dose chest CT for lung cancer screening provides the opportunity to simultaneously screen patients for osteoporosis, detecting notably higher rates of osteoporosis in men than the traditional tool of DXA, research published in the Journal of Bone and Mineral Research shows.
“Our large-scale, multicenter study of bone density measured from routine low-dose CT scans demonstrated the great potential of using low-dose CT for the opportunistic screening of osteoporosis as an alternative to standard DXA scans,” said senior author Wei Tian, MD, of the Chinese Academy of Engineering and Peking University, in a press statement from the journal.
“Our study revealed the unexpectedly high prevalence of osteoporosis in men, which may impact on the management strategy of men in the future,” Dr. Tian added.
Josephine Therkildsen, MD, of Herning Hospital, Denmark, who has conducted similar research using cardiac CT scans, said the findings add important new insights into the issue of opportunistic screening.
“The results are highly interesting, as they show that low-dose CT-based opportunistic screening could identify a substantial number of patients with low lumbar bone mineral density (BMD) with the future potential to diagnose osteoporosis and initiate relevant treatment before a fracture occurs,” she told this news organization.
Perry J. Pickhardt, MD, chief of gastrointestinal imaging at the University of Wisconsin School of Medicine and Public Health in Madison, agrees. He said in an interview that CT scans of the chest and abdomen, commonly performed for a variety of clinical indications and widespread in most developed countries, can in fact be essential for the detection of a multitude of other concerns – yet are underused for those other purposes.
Use of CT in this way “would likely be very cost effective and clinically efficacious,” he said, adding: “We are seeing greatly increased interest in leveraging this extra information that is contained within every CT scan.” And, “Importantly, artificial intelligence advances now allow for automated approaches, which should allow for expanded use.”
Lung cancer CT scans shed light on osteoporosis prevalence
In the study, led by Xiaoguang Cheng, MD, PhD, of the department of radiology, Beijing Jishuitan Hospital, China, researchers examined lung cancer CT screening data from the prospective China Biobank Project to determine the prevalence of osteoporosis in China.
This included the thoracic low-dose CT scans of 69,095 adults, including 40,733 men and 28,362 women, taken between 2018 and 2019.
To screen for osteoporosis, they used quantitative CT software to evaluate lumbar spine (L1-L2) trabecular volume BMD (vBMD) and diagnostic criteria from the American College of Radiology. Using the vBMD measures from the CT imaging, they found the prevalence of osteoporosis among those over 50 years of age in the Chinese population to be 29% for women (49 million) and 13.5% for men (22.8 million).
Interestingly, the osteoporosis prevalence rate among women was comparable to estimates in the population derived from DXA (29.1%); however, the rate in men was twice that estimated from DXA scans (6.5%).
Decreases in trabecular vBMD with age were observed in both genders. However, declines were steeper among women, who had higher peak trabecular vBMD (185.4 mg/cm3), compared with men (176.6 mg/cm3) at age 30-34 years, but significantly lower measures (62.4 mg/cm3) than men (92.1 mg/cm3) at age 80 years.
The prevalence of osteoporosis in women increased from 2.8% at age 50-54 years to 79.8% at age 85 or older, while in men, the prevalence was 3.2% at age 50-54 years and 44.1% at age 85 or older.
“This is the first study to establish Chinese reference data for vBMD using opportunistic screening from low-dose chest CT in a large population cohort,” the authors write.
“The opportunistic screening of osteoporosis using low-dose CT is clinically feasible and requires no additional exposure to ionizing radiation.”
In addition, no additional equipment or patient time was required, suggesting that “this approach has potential for opportunistic screening for osteoporosis.”
They note, however, that further cohort studies are needed to assess clinical utility of this method.
CT ‘likely a more accurate measure’ of volumetric BMD
Dr. Pickhardt said the differences in osteoporosis prevalence observed between DXA and CT-derived measures in men likely reflect the greater accuracy of CT.
“DXA is a planar technique with a number of drawbacks,” he said in an interview. “CT provides a more direct volumetric measure and is likely a more accurate method for BMD assessment.”
He speculated that the greater differences between DXA versus CT seen in men than women “may relate to sex differences in cortical bone of vertebral bodies, which cannot be separated from the underlying trabecular bone with DXA (whereas CT directly measures the inner trabecular bone).”
The authors note that, although areal BMD (aBMD) derived from DXA is required for osteoporosis diagnosis according to World Health Organization criteria, “trabecular vBMD derived from CT can be also used for diagnosis based on thresholds published by the American College of Radiology of 120 mg/cm3 and 80 mg/cm3 to define osteopenia and osteoporosis, respectively, thresholds that were subsequently confirmed for the Chinese population.”
Furthermore, vBMD has been shown in some studies to be more strongly related to fracture risk, compared with DXA aBMD measures.
Importantly, in another recent study involving 9,223 adults, Dr. Pickhardt and colleagues reported that bone and muscle biomarkers derived from CT were comparable to the Fracture Risk Assessment Tool score for the presymptomatic prediction of future osteoporotic fractures.
Dr. Pickhardt is an advisor to Bracco Imaging and Zebra Medical Vision. Dr. Therkildsen has reported no relevant financial relationships.
This article first appeared on Medscape.com.
Low-dose chest CT for lung cancer screening provides the opportunity to simultaneously screen patients for osteoporosis, detecting notably higher rates of osteoporosis in men than the traditional tool of DXA, research published in the Journal of Bone and Mineral Research shows.
“Our large-scale, multicenter study of bone density measured from routine low-dose CT scans demonstrated the great potential of using low-dose CT for the opportunistic screening of osteoporosis as an alternative to standard DXA scans,” said senior author Wei Tian, MD, of the Chinese Academy of Engineering and Peking University, in a press statement from the journal.
“Our study revealed the unexpectedly high prevalence of osteoporosis in men, which may impact on the management strategy of men in the future,” Dr. Tian added.
Josephine Therkildsen, MD, of Herning Hospital, Denmark, who has conducted similar research using cardiac CT scans, said the findings add important new insights into the issue of opportunistic screening.
“The results are highly interesting, as they show that low-dose CT-based opportunistic screening could identify a substantial number of patients with low lumbar bone mineral density (BMD) with the future potential to diagnose osteoporosis and initiate relevant treatment before a fracture occurs,” she told this news organization.
Perry J. Pickhardt, MD, chief of gastrointestinal imaging at the University of Wisconsin School of Medicine and Public Health in Madison, agrees. He said in an interview that CT scans of the chest and abdomen, commonly performed for a variety of clinical indications and widespread in most developed countries, can in fact be essential for the detection of a multitude of other concerns – yet are underused for those other purposes.
Use of CT in this way “would likely be very cost effective and clinically efficacious,” he said, adding: “We are seeing greatly increased interest in leveraging this extra information that is contained within every CT scan.” And, “Importantly, artificial intelligence advances now allow for automated approaches, which should allow for expanded use.”
Lung cancer CT scans shed light on osteoporosis prevalence
In the study, led by Xiaoguang Cheng, MD, PhD, of the department of radiology, Beijing Jishuitan Hospital, China, researchers examined lung cancer CT screening data from the prospective China Biobank Project to determine the prevalence of osteoporosis in China.
This included the thoracic low-dose CT scans of 69,095 adults, including 40,733 men and 28,362 women, taken between 2018 and 2019.
To screen for osteoporosis, they used quantitative CT software to evaluate lumbar spine (L1-L2) trabecular volume BMD (vBMD) and diagnostic criteria from the American College of Radiology. Using the vBMD measures from the CT imaging, they found the prevalence of osteoporosis among those over 50 years of age in the Chinese population to be 29% for women (49 million) and 13.5% for men (22.8 million).
Interestingly, the osteoporosis prevalence rate among women was comparable to estimates in the population derived from DXA (29.1%); however, the rate in men was twice that estimated from DXA scans (6.5%).
Decreases in trabecular vBMD with age were observed in both genders. However, declines were steeper among women, who had higher peak trabecular vBMD (185.4 mg/cm3), compared with men (176.6 mg/cm3) at age 30-34 years, but significantly lower measures (62.4 mg/cm3) than men (92.1 mg/cm3) at age 80 years.
The prevalence of osteoporosis in women increased from 2.8% at age 50-54 years to 79.8% at age 85 or older, while in men, the prevalence was 3.2% at age 50-54 years and 44.1% at age 85 or older.
“This is the first study to establish Chinese reference data for vBMD using opportunistic screening from low-dose chest CT in a large population cohort,” the authors write.
“The opportunistic screening of osteoporosis using low-dose CT is clinically feasible and requires no additional exposure to ionizing radiation.”
In addition, no additional equipment or patient time was required, suggesting that “this approach has potential for opportunistic screening for osteoporosis.”
They note, however, that further cohort studies are needed to assess clinical utility of this method.
CT ‘likely a more accurate measure’ of volumetric BMD
Dr. Pickhardt said the differences in osteoporosis prevalence observed between DXA and CT-derived measures in men likely reflect the greater accuracy of CT.
“DXA is a planar technique with a number of drawbacks,” he said in an interview. “CT provides a more direct volumetric measure and is likely a more accurate method for BMD assessment.”
He speculated that the greater differences between DXA versus CT seen in men than women “may relate to sex differences in cortical bone of vertebral bodies, which cannot be separated from the underlying trabecular bone with DXA (whereas CT directly measures the inner trabecular bone).”
The authors note that, although areal BMD (aBMD) derived from DXA is required for osteoporosis diagnosis according to World Health Organization criteria, “trabecular vBMD derived from CT can be also used for diagnosis based on thresholds published by the American College of Radiology of 120 mg/cm3 and 80 mg/cm3 to define osteopenia and osteoporosis, respectively, thresholds that were subsequently confirmed for the Chinese population.”
Furthermore, vBMD has been shown in some studies to be more strongly related to fracture risk, compared with DXA aBMD measures.
Importantly, in another recent study involving 9,223 adults, Dr. Pickhardt and colleagues reported that bone and muscle biomarkers derived from CT were comparable to the Fracture Risk Assessment Tool score for the presymptomatic prediction of future osteoporotic fractures.
Dr. Pickhardt is an advisor to Bracco Imaging and Zebra Medical Vision. Dr. Therkildsen has reported no relevant financial relationships.
This article first appeared on Medscape.com.
Low-dose chest CT for lung cancer screening provides the opportunity to simultaneously screen patients for osteoporosis, detecting notably higher rates of osteoporosis in men than the traditional tool of DXA, research published in the Journal of Bone and Mineral Research shows.
“Our large-scale, multicenter study of bone density measured from routine low-dose CT scans demonstrated the great potential of using low-dose CT for the opportunistic screening of osteoporosis as an alternative to standard DXA scans,” said senior author Wei Tian, MD, of the Chinese Academy of Engineering and Peking University, in a press statement from the journal.
“Our study revealed the unexpectedly high prevalence of osteoporosis in men, which may impact on the management strategy of men in the future,” Dr. Tian added.
Josephine Therkildsen, MD, of Herning Hospital, Denmark, who has conducted similar research using cardiac CT scans, said the findings add important new insights into the issue of opportunistic screening.
“The results are highly interesting, as they show that low-dose CT-based opportunistic screening could identify a substantial number of patients with low lumbar bone mineral density (BMD) with the future potential to diagnose osteoporosis and initiate relevant treatment before a fracture occurs,” she told this news organization.
Perry J. Pickhardt, MD, chief of gastrointestinal imaging at the University of Wisconsin School of Medicine and Public Health in Madison, agrees. He said in an interview that CT scans of the chest and abdomen, commonly performed for a variety of clinical indications and widespread in most developed countries, can in fact be essential for the detection of a multitude of other concerns – yet are underused for those other purposes.
Use of CT in this way “would likely be very cost effective and clinically efficacious,” he said, adding: “We are seeing greatly increased interest in leveraging this extra information that is contained within every CT scan.” And, “Importantly, artificial intelligence advances now allow for automated approaches, which should allow for expanded use.”
Lung cancer CT scans shed light on osteoporosis prevalence
In the study, led by Xiaoguang Cheng, MD, PhD, of the department of radiology, Beijing Jishuitan Hospital, China, researchers examined lung cancer CT screening data from the prospective China Biobank Project to determine the prevalence of osteoporosis in China.
This included the thoracic low-dose CT scans of 69,095 adults, including 40,733 men and 28,362 women, taken between 2018 and 2019.
To screen for osteoporosis, they used quantitative CT software to evaluate lumbar spine (L1-L2) trabecular volume BMD (vBMD) and diagnostic criteria from the American College of Radiology. Using the vBMD measures from the CT imaging, they found the prevalence of osteoporosis among those over 50 years of age in the Chinese population to be 29% for women (49 million) and 13.5% for men (22.8 million).
Interestingly, the osteoporosis prevalence rate among women was comparable to estimates in the population derived from DXA (29.1%); however, the rate in men was twice that estimated from DXA scans (6.5%).
Decreases in trabecular vBMD with age were observed in both genders. However, declines were steeper among women, who had higher peak trabecular vBMD (185.4 mg/cm3), compared with men (176.6 mg/cm3) at age 30-34 years, but significantly lower measures (62.4 mg/cm3) than men (92.1 mg/cm3) at age 80 years.
The prevalence of osteoporosis in women increased from 2.8% at age 50-54 years to 79.8% at age 85 or older, while in men, the prevalence was 3.2% at age 50-54 years and 44.1% at age 85 or older.
“This is the first study to establish Chinese reference data for vBMD using opportunistic screening from low-dose chest CT in a large population cohort,” the authors write.
“The opportunistic screening of osteoporosis using low-dose CT is clinically feasible and requires no additional exposure to ionizing radiation.”
In addition, no additional equipment or patient time was required, suggesting that “this approach has potential for opportunistic screening for osteoporosis.”
They note, however, that further cohort studies are needed to assess clinical utility of this method.
CT ‘likely a more accurate measure’ of volumetric BMD
Dr. Pickhardt said the differences in osteoporosis prevalence observed between DXA and CT-derived measures in men likely reflect the greater accuracy of CT.
“DXA is a planar technique with a number of drawbacks,” he said in an interview. “CT provides a more direct volumetric measure and is likely a more accurate method for BMD assessment.”
He speculated that the greater differences between DXA versus CT seen in men than women “may relate to sex differences in cortical bone of vertebral bodies, which cannot be separated from the underlying trabecular bone with DXA (whereas CT directly measures the inner trabecular bone).”
The authors note that, although areal BMD (aBMD) derived from DXA is required for osteoporosis diagnosis according to World Health Organization criteria, “trabecular vBMD derived from CT can be also used for diagnosis based on thresholds published by the American College of Radiology of 120 mg/cm3 and 80 mg/cm3 to define osteopenia and osteoporosis, respectively, thresholds that were subsequently confirmed for the Chinese population.”
Furthermore, vBMD has been shown in some studies to be more strongly related to fracture risk, compared with DXA aBMD measures.
Importantly, in another recent study involving 9,223 adults, Dr. Pickhardt and colleagues reported that bone and muscle biomarkers derived from CT were comparable to the Fracture Risk Assessment Tool score for the presymptomatic prediction of future osteoporotic fractures.
Dr. Pickhardt is an advisor to Bracco Imaging and Zebra Medical Vision. Dr. Therkildsen has reported no relevant financial relationships.
This article first appeared on Medscape.com.
Black patients with ES-SCLC get less chemo but have better survival
Black patients with extensive-stage small cell lung cancer (ES-SCLC) are less likely to receive chemotherapy but have better survival, compared with White patients, according to a study published in JTO Clinical Research and Reports.
This study provides a large-scale analysis of real-world data identifying racial and socioeconomic factors impacting systemic therapy delivery and survival in ES-SCLC.
“The most important finding was the significant disparity in receipt of chemotherapy,” said study author Umit Tapan, MD, of Boston Medical Center.
“Black individuals with ES-SCLC were less likely to receive chemotherapy compared to Whites and other racial groups. Similarly, elderly, uninsured patients, patients with nonprivate health insurance, and those with lower education levels were less likely to be treated with chemotherapy,” Dr. Tapan said.
Using the National Cancer Data Base (NCDB), Dr. Tapan and colleagues identified 148,961 patients who were diagnosed with stage IV ES-SCLC during 2004-2016. In all, 82,592 patients were included in the study.
Results: Treatment and survival
Compared with White patients, Black patients (adjusted odds ratio, 0.85; P = .0004) and patients from other racial groups (aOR, 0.87; P = .126) had lower odds of receiving chemotherapy on multivariate analysis.
However, survival was superior in Black patients (adjusted hazard ratio, 0.92; P < .0001) and other non-White patients (aHR 0.86; P < .0001).
“We speculate that additional factors, such as performance status, which is not captured by NCDB, might have accounted for better survival for Black patients,” Dr. Tapan said, noting that the analysis was adjusted for known possible confounding factors, such as age, gender, and comorbidity status.
Black patients had higher odds of receiving chemotherapy between 2010 and 2016 compared with 2004 and 2009. “This suggests a positive impact of the Patient Protection and Affordable Care Act (ACA) in 2010,” Dr. Tapan said.
Another surprising finding pertained to patients with nonprivate insurance. These patients had even lower odds of getting chemotherapy after the implementation of ACA, Dr. Tapan said. Patients who had private insurance had higher survival compared with those who were uninsured.
Higher level of education, measured by percentage of residents with a high school degree, increased the odds of receiving chemotherapy.
Age also had a significant impact on receipt of chemotherapy. About 83% of patients over age 80 years received chemotherapy, compared with 94% of patients aged 40-64 years.
Real-world data
Minorities are underrepresented in cancer clinical trials in the United States, with only 2% of National Cancer Institute trials having sufficient minority participants, Dr. Tapan said. A study published in Academic Medicine in 2018 showed that only 13% of 782 National Institute of Health–sponsored clinical trials reported outcomes by race and ethnicity.
As a result, we are missing data on patient care in minority populations, Dr. Tapan said. “Collecting and analyzing real-world data becomes critical to study treatment patterns and outcomes,” he added.
The current real-world study had a somewhat diverse patient population, but 90.6% of patients were White, 7.8% were Black, and 1.7% were other races.
“We would have expected a higher percentage of Black patients considering the most recent U.S. Census Bureau estimates that 76.3% of the U.S. population is White and 13.4% is Black,” Dr. Tapan said. “There are conflicting results in the literature regarding racial disparities in SCLC and survival. Many of these studies were performed via state-based cancer registries instead of on a national level, making prior reports less generalizable.”
‘More work to do’
While the new study showed patients with nonprivate insurance or those with no insurance were less likely to receive chemotherapy, studies have shown that chemotherapy administration was not impacted by insurance status in limited-stage SCLC.
This is in contrast to radiotherapy delivery. Studies have revealed a lower likelihood of radiotherapy delivery in limited-stage SCLC for patients with government health insurance such as Medicare/Medicaid, Dr. Tapan said.
“Access to cancer care has been shown to be one of the most important barriers in racial disparity. Studies analyzing outcomes in the equal access health systems, such as the Veteran Administration, have revealed less racial disparities,” Dr. Tapan said.
Even when Black patients have equal access to care, they might receive suboptimal treatment, Dr. Tapan noted.
“Studies have shown that Black patients are not only more likely to refuse surgery, but also are more likely to be given a negative recommendation by a surgeon as compared to Whites, suggesting potential involvement of miscommunication or bias during patient-physician encounters,” Dr. Tapan said. “In the same vein, physicians would need to acknowledge their patients’ beliefs. Not doing so may lead to unsatisfactory physician-patient interactions and suboptimal decision-making.”
“Measures to reduce physician bias are an important step to reduce disparities,” Dr. Tapan continued. “Studies have shown that Black patients are perceived to be less intelligent and educated, less likely to have social support, and more likely to be at risk of noncompliance. For some patients and oncologists, extra effort is needed so that every patient can access the best possible treatments and outcomes. It is the oncologist’s responsibility to advocate for patients, but, ultimately, further legislative actions are needed to mitigate the disparities around cancer care.”
Dr. Tapan noted that, in 1966, Martin Luther King Jr., PhD, stated that “of all the forms of inequality, injustice in health care is the most shocking and inhumane.”
Dr. Tapan said: “We have overcome some barriers since 1966, but we have more work to do.” He and colleagues had no disclosures related to this study.
SOURCE: Tapan U et al. JTO Clin Res Rep. 2020. doi: 10.1016/j.jtocrr.2020.100109.
Black patients with extensive-stage small cell lung cancer (ES-SCLC) are less likely to receive chemotherapy but have better survival, compared with White patients, according to a study published in JTO Clinical Research and Reports.
This study provides a large-scale analysis of real-world data identifying racial and socioeconomic factors impacting systemic therapy delivery and survival in ES-SCLC.
“The most important finding was the significant disparity in receipt of chemotherapy,” said study author Umit Tapan, MD, of Boston Medical Center.
“Black individuals with ES-SCLC were less likely to receive chemotherapy compared to Whites and other racial groups. Similarly, elderly, uninsured patients, patients with nonprivate health insurance, and those with lower education levels were less likely to be treated with chemotherapy,” Dr. Tapan said.
Using the National Cancer Data Base (NCDB), Dr. Tapan and colleagues identified 148,961 patients who were diagnosed with stage IV ES-SCLC during 2004-2016. In all, 82,592 patients were included in the study.
Results: Treatment and survival
Compared with White patients, Black patients (adjusted odds ratio, 0.85; P = .0004) and patients from other racial groups (aOR, 0.87; P = .126) had lower odds of receiving chemotherapy on multivariate analysis.
However, survival was superior in Black patients (adjusted hazard ratio, 0.92; P < .0001) and other non-White patients (aHR 0.86; P < .0001).
“We speculate that additional factors, such as performance status, which is not captured by NCDB, might have accounted for better survival for Black patients,” Dr. Tapan said, noting that the analysis was adjusted for known possible confounding factors, such as age, gender, and comorbidity status.
Black patients had higher odds of receiving chemotherapy between 2010 and 2016 compared with 2004 and 2009. “This suggests a positive impact of the Patient Protection and Affordable Care Act (ACA) in 2010,” Dr. Tapan said.
Another surprising finding pertained to patients with nonprivate insurance. These patients had even lower odds of getting chemotherapy after the implementation of ACA, Dr. Tapan said. Patients who had private insurance had higher survival compared with those who were uninsured.
Higher level of education, measured by percentage of residents with a high school degree, increased the odds of receiving chemotherapy.
Age also had a significant impact on receipt of chemotherapy. About 83% of patients over age 80 years received chemotherapy, compared with 94% of patients aged 40-64 years.
Real-world data
Minorities are underrepresented in cancer clinical trials in the United States, with only 2% of National Cancer Institute trials having sufficient minority participants, Dr. Tapan said. A study published in Academic Medicine in 2018 showed that only 13% of 782 National Institute of Health–sponsored clinical trials reported outcomes by race and ethnicity.
As a result, we are missing data on patient care in minority populations, Dr. Tapan said. “Collecting and analyzing real-world data becomes critical to study treatment patterns and outcomes,” he added.
The current real-world study had a somewhat diverse patient population, but 90.6% of patients were White, 7.8% were Black, and 1.7% were other races.
“We would have expected a higher percentage of Black patients considering the most recent U.S. Census Bureau estimates that 76.3% of the U.S. population is White and 13.4% is Black,” Dr. Tapan said. “There are conflicting results in the literature regarding racial disparities in SCLC and survival. Many of these studies were performed via state-based cancer registries instead of on a national level, making prior reports less generalizable.”
‘More work to do’
While the new study showed patients with nonprivate insurance or those with no insurance were less likely to receive chemotherapy, studies have shown that chemotherapy administration was not impacted by insurance status in limited-stage SCLC.
This is in contrast to radiotherapy delivery. Studies have revealed a lower likelihood of radiotherapy delivery in limited-stage SCLC for patients with government health insurance such as Medicare/Medicaid, Dr. Tapan said.
“Access to cancer care has been shown to be one of the most important barriers in racial disparity. Studies analyzing outcomes in the equal access health systems, such as the Veteran Administration, have revealed less racial disparities,” Dr. Tapan said.
Even when Black patients have equal access to care, they might receive suboptimal treatment, Dr. Tapan noted.
“Studies have shown that Black patients are not only more likely to refuse surgery, but also are more likely to be given a negative recommendation by a surgeon as compared to Whites, suggesting potential involvement of miscommunication or bias during patient-physician encounters,” Dr. Tapan said. “In the same vein, physicians would need to acknowledge their patients’ beliefs. Not doing so may lead to unsatisfactory physician-patient interactions and suboptimal decision-making.”
“Measures to reduce physician bias are an important step to reduce disparities,” Dr. Tapan continued. “Studies have shown that Black patients are perceived to be less intelligent and educated, less likely to have social support, and more likely to be at risk of noncompliance. For some patients and oncologists, extra effort is needed so that every patient can access the best possible treatments and outcomes. It is the oncologist’s responsibility to advocate for patients, but, ultimately, further legislative actions are needed to mitigate the disparities around cancer care.”
Dr. Tapan noted that, in 1966, Martin Luther King Jr., PhD, stated that “of all the forms of inequality, injustice in health care is the most shocking and inhumane.”
Dr. Tapan said: “We have overcome some barriers since 1966, but we have more work to do.” He and colleagues had no disclosures related to this study.
SOURCE: Tapan U et al. JTO Clin Res Rep. 2020. doi: 10.1016/j.jtocrr.2020.100109.
Black patients with extensive-stage small cell lung cancer (ES-SCLC) are less likely to receive chemotherapy but have better survival, compared with White patients, according to a study published in JTO Clinical Research and Reports.
This study provides a large-scale analysis of real-world data identifying racial and socioeconomic factors impacting systemic therapy delivery and survival in ES-SCLC.
“The most important finding was the significant disparity in receipt of chemotherapy,” said study author Umit Tapan, MD, of Boston Medical Center.
“Black individuals with ES-SCLC were less likely to receive chemotherapy compared to Whites and other racial groups. Similarly, elderly, uninsured patients, patients with nonprivate health insurance, and those with lower education levels were less likely to be treated with chemotherapy,” Dr. Tapan said.
Using the National Cancer Data Base (NCDB), Dr. Tapan and colleagues identified 148,961 patients who were diagnosed with stage IV ES-SCLC during 2004-2016. In all, 82,592 patients were included in the study.
Results: Treatment and survival
Compared with White patients, Black patients (adjusted odds ratio, 0.85; P = .0004) and patients from other racial groups (aOR, 0.87; P = .126) had lower odds of receiving chemotherapy on multivariate analysis.
However, survival was superior in Black patients (adjusted hazard ratio, 0.92; P < .0001) and other non-White patients (aHR 0.86; P < .0001).
“We speculate that additional factors, such as performance status, which is not captured by NCDB, might have accounted for better survival for Black patients,” Dr. Tapan said, noting that the analysis was adjusted for known possible confounding factors, such as age, gender, and comorbidity status.
Black patients had higher odds of receiving chemotherapy between 2010 and 2016 compared with 2004 and 2009. “This suggests a positive impact of the Patient Protection and Affordable Care Act (ACA) in 2010,” Dr. Tapan said.
Another surprising finding pertained to patients with nonprivate insurance. These patients had even lower odds of getting chemotherapy after the implementation of ACA, Dr. Tapan said. Patients who had private insurance had higher survival compared with those who were uninsured.
Higher level of education, measured by percentage of residents with a high school degree, increased the odds of receiving chemotherapy.
Age also had a significant impact on receipt of chemotherapy. About 83% of patients over age 80 years received chemotherapy, compared with 94% of patients aged 40-64 years.
Real-world data
Minorities are underrepresented in cancer clinical trials in the United States, with only 2% of National Cancer Institute trials having sufficient minority participants, Dr. Tapan said. A study published in Academic Medicine in 2018 showed that only 13% of 782 National Institute of Health–sponsored clinical trials reported outcomes by race and ethnicity.
As a result, we are missing data on patient care in minority populations, Dr. Tapan said. “Collecting and analyzing real-world data becomes critical to study treatment patterns and outcomes,” he added.
The current real-world study had a somewhat diverse patient population, but 90.6% of patients were White, 7.8% were Black, and 1.7% were other races.
“We would have expected a higher percentage of Black patients considering the most recent U.S. Census Bureau estimates that 76.3% of the U.S. population is White and 13.4% is Black,” Dr. Tapan said. “There are conflicting results in the literature regarding racial disparities in SCLC and survival. Many of these studies were performed via state-based cancer registries instead of on a national level, making prior reports less generalizable.”
‘More work to do’
While the new study showed patients with nonprivate insurance or those with no insurance were less likely to receive chemotherapy, studies have shown that chemotherapy administration was not impacted by insurance status in limited-stage SCLC.
This is in contrast to radiotherapy delivery. Studies have revealed a lower likelihood of radiotherapy delivery in limited-stage SCLC for patients with government health insurance such as Medicare/Medicaid, Dr. Tapan said.
“Access to cancer care has been shown to be one of the most important barriers in racial disparity. Studies analyzing outcomes in the equal access health systems, such as the Veteran Administration, have revealed less racial disparities,” Dr. Tapan said.
Even when Black patients have equal access to care, they might receive suboptimal treatment, Dr. Tapan noted.
“Studies have shown that Black patients are not only more likely to refuse surgery, but also are more likely to be given a negative recommendation by a surgeon as compared to Whites, suggesting potential involvement of miscommunication or bias during patient-physician encounters,” Dr. Tapan said. “In the same vein, physicians would need to acknowledge their patients’ beliefs. Not doing so may lead to unsatisfactory physician-patient interactions and suboptimal decision-making.”
“Measures to reduce physician bias are an important step to reduce disparities,” Dr. Tapan continued. “Studies have shown that Black patients are perceived to be less intelligent and educated, less likely to have social support, and more likely to be at risk of noncompliance. For some patients and oncologists, extra effort is needed so that every patient can access the best possible treatments and outcomes. It is the oncologist’s responsibility to advocate for patients, but, ultimately, further legislative actions are needed to mitigate the disparities around cancer care.”
Dr. Tapan noted that, in 1966, Martin Luther King Jr., PhD, stated that “of all the forms of inequality, injustice in health care is the most shocking and inhumane.”
Dr. Tapan said: “We have overcome some barriers since 1966, but we have more work to do.” He and colleagues had no disclosures related to this study.
SOURCE: Tapan U et al. JTO Clin Res Rep. 2020. doi: 10.1016/j.jtocrr.2020.100109.
FROM JTO CLINICAL AND RESEARCH REPORTS
Immune checkpoint inhibitors don’t increase COVID-19 incidence or mortality, studies suggest
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
FROM SITC 2020
Reduced cancer mortality with Medicaid expansion
Researchers reviewed data on 523,802 patients in the National Cancer Database who were diagnosed with cancer from 2012 through 2015. Slightly more than half of patients (55.2%) lived in Medicaid expansion states.
After expansion, mortality significantly decreased in expansion states (hazard ratio, 0.98; P = .008) but not in nonexpansion states (HR, 1.01; P = .43). The difference was significant in a difference-in-difference analysis (HR, 1.03; P = .01).
Across 69,000 patients with newly diagnosed cancer in Medicaid expansion states, the 2% decrease in the hazard of death would translate to 1,384 lives saved annually.
The benefit was primarily observed in patients with nonmetastatic cancer. For patients with stage I-III cancer, the risk of death was increased in nonexpansion states (HR, 1.05; P < .001) and unchanged in expansion states (HR, 0.99; P = .64). Mortality significantly improved in expansion states vs. nonexpansion states (HR, 1.05; P = .003).
For patients with stage IV cancer, both expansion and nonexpansion states had improvements in mortality, but the differences were not significant.
“Earlier stage at diagnosis appears to explain the mortality improvement,” wrote study author Miranda Lam, MD, of Harvard Medical School, Boston, and colleagues.
Clinical benefits, ‘no economic downside’
Under the Affordable Care Act, passed in 2010, states have the option of expanding Medicaid eligibility to adults with incomes at or below 138% of the federal poverty level. As of March 2020, 36 states and the District of Columbia had expanded Medicaid, with more than 20 million residents obtaining coverage.
Previous studies have associated Medicaid expansion with fewer patients being uninsured, increased cancer screening, and earlier stage of diagnosis, as well as reduced racial disparities in access to high-volume hospitals for cancer surgery and increased rates of cancer surgery among low-income patients.
“This study adds to an increasingly large body of research finding that Medicaid expansion has improved our ability to fight cancer,” said Coleman Drake, PhD, of the University of Pittsburgh, who was not involved in this study.
“Obtaining health insurance through Medicaid allows patients to receive recommended preventive cancer screenings, which explains the increase in early-stage diagnosis rates. Detecting cancer early is critical for successful cancer treatment,” Dr. Drake noted.
“It is hard to overstate the positive effects of Medicaid expansion on health outcomes. At the same time, concerns that Medicaid expansion would be costly to state governments’ budgets have not been realized. In short, Medicaid expansion yields many benefits and has no economic downside for state policymakers. Clinical and economic evidence make an overwhelming case for states to expand Medicaid,” Dr. Drake said.
Significant difference for lung cancer
Most patients in this study were women (73.6%), and the patients’ mean age was 54.8 years (range, 40-64 years). Patients had newly diagnosed breast cancer (52.2%), colorectal cancer (21.3%), and lung cancer (26.5%).
The benefits of Medicaid expansion persisted after adjustment for education, income, insurance, and race.
The lower mortality in expansion states compared with nonexpansion states was similar across all three cancer types. However, in stratified analyses, the difference was significant only for lung cancer (P = .03).
“Lung cancer has a higher mortality rate than breast and colorectal cancer, and with longer follow-up, it is possible that the lower mortality rates seen for breast and colorectal cancer may also become significant,” the authors wrote.
This research was funded by Harvard Catalyst, the Harvard Clinical and Translational Science Center, and the National Center for Advancing Translational Sciences at the National Institutes of Health. The investigators and Dr. Drake had no relevant disclosures.
SOURCE: Lam MB et al. JAMA Netw Open. 2020 Nov 2;3(11):e2024366.
Researchers reviewed data on 523,802 patients in the National Cancer Database who were diagnosed with cancer from 2012 through 2015. Slightly more than half of patients (55.2%) lived in Medicaid expansion states.
After expansion, mortality significantly decreased in expansion states (hazard ratio, 0.98; P = .008) but not in nonexpansion states (HR, 1.01; P = .43). The difference was significant in a difference-in-difference analysis (HR, 1.03; P = .01).
Across 69,000 patients with newly diagnosed cancer in Medicaid expansion states, the 2% decrease in the hazard of death would translate to 1,384 lives saved annually.
The benefit was primarily observed in patients with nonmetastatic cancer. For patients with stage I-III cancer, the risk of death was increased in nonexpansion states (HR, 1.05; P < .001) and unchanged in expansion states (HR, 0.99; P = .64). Mortality significantly improved in expansion states vs. nonexpansion states (HR, 1.05; P = .003).
For patients with stage IV cancer, both expansion and nonexpansion states had improvements in mortality, but the differences were not significant.
“Earlier stage at diagnosis appears to explain the mortality improvement,” wrote study author Miranda Lam, MD, of Harvard Medical School, Boston, and colleagues.
Clinical benefits, ‘no economic downside’
Under the Affordable Care Act, passed in 2010, states have the option of expanding Medicaid eligibility to adults with incomes at or below 138% of the federal poverty level. As of March 2020, 36 states and the District of Columbia had expanded Medicaid, with more than 20 million residents obtaining coverage.
Previous studies have associated Medicaid expansion with fewer patients being uninsured, increased cancer screening, and earlier stage of diagnosis, as well as reduced racial disparities in access to high-volume hospitals for cancer surgery and increased rates of cancer surgery among low-income patients.
“This study adds to an increasingly large body of research finding that Medicaid expansion has improved our ability to fight cancer,” said Coleman Drake, PhD, of the University of Pittsburgh, who was not involved in this study.
“Obtaining health insurance through Medicaid allows patients to receive recommended preventive cancer screenings, which explains the increase in early-stage diagnosis rates. Detecting cancer early is critical for successful cancer treatment,” Dr. Drake noted.
“It is hard to overstate the positive effects of Medicaid expansion on health outcomes. At the same time, concerns that Medicaid expansion would be costly to state governments’ budgets have not been realized. In short, Medicaid expansion yields many benefits and has no economic downside for state policymakers. Clinical and economic evidence make an overwhelming case for states to expand Medicaid,” Dr. Drake said.
Significant difference for lung cancer
Most patients in this study were women (73.6%), and the patients’ mean age was 54.8 years (range, 40-64 years). Patients had newly diagnosed breast cancer (52.2%), colorectal cancer (21.3%), and lung cancer (26.5%).
The benefits of Medicaid expansion persisted after adjustment for education, income, insurance, and race.
The lower mortality in expansion states compared with nonexpansion states was similar across all three cancer types. However, in stratified analyses, the difference was significant only for lung cancer (P = .03).
“Lung cancer has a higher mortality rate than breast and colorectal cancer, and with longer follow-up, it is possible that the lower mortality rates seen for breast and colorectal cancer may also become significant,” the authors wrote.
This research was funded by Harvard Catalyst, the Harvard Clinical and Translational Science Center, and the National Center for Advancing Translational Sciences at the National Institutes of Health. The investigators and Dr. Drake had no relevant disclosures.
SOURCE: Lam MB et al. JAMA Netw Open. 2020 Nov 2;3(11):e2024366.
Researchers reviewed data on 523,802 patients in the National Cancer Database who were diagnosed with cancer from 2012 through 2015. Slightly more than half of patients (55.2%) lived in Medicaid expansion states.
After expansion, mortality significantly decreased in expansion states (hazard ratio, 0.98; P = .008) but not in nonexpansion states (HR, 1.01; P = .43). The difference was significant in a difference-in-difference analysis (HR, 1.03; P = .01).
Across 69,000 patients with newly diagnosed cancer in Medicaid expansion states, the 2% decrease in the hazard of death would translate to 1,384 lives saved annually.
The benefit was primarily observed in patients with nonmetastatic cancer. For patients with stage I-III cancer, the risk of death was increased in nonexpansion states (HR, 1.05; P < .001) and unchanged in expansion states (HR, 0.99; P = .64). Mortality significantly improved in expansion states vs. nonexpansion states (HR, 1.05; P = .003).
For patients with stage IV cancer, both expansion and nonexpansion states had improvements in mortality, but the differences were not significant.
“Earlier stage at diagnosis appears to explain the mortality improvement,” wrote study author Miranda Lam, MD, of Harvard Medical School, Boston, and colleagues.
Clinical benefits, ‘no economic downside’
Under the Affordable Care Act, passed in 2010, states have the option of expanding Medicaid eligibility to adults with incomes at or below 138% of the federal poverty level. As of March 2020, 36 states and the District of Columbia had expanded Medicaid, with more than 20 million residents obtaining coverage.
Previous studies have associated Medicaid expansion with fewer patients being uninsured, increased cancer screening, and earlier stage of diagnosis, as well as reduced racial disparities in access to high-volume hospitals for cancer surgery and increased rates of cancer surgery among low-income patients.
“This study adds to an increasingly large body of research finding that Medicaid expansion has improved our ability to fight cancer,” said Coleman Drake, PhD, of the University of Pittsburgh, who was not involved in this study.
“Obtaining health insurance through Medicaid allows patients to receive recommended preventive cancer screenings, which explains the increase in early-stage diagnosis rates. Detecting cancer early is critical for successful cancer treatment,” Dr. Drake noted.
“It is hard to overstate the positive effects of Medicaid expansion on health outcomes. At the same time, concerns that Medicaid expansion would be costly to state governments’ budgets have not been realized. In short, Medicaid expansion yields many benefits and has no economic downside for state policymakers. Clinical and economic evidence make an overwhelming case for states to expand Medicaid,” Dr. Drake said.
Significant difference for lung cancer
Most patients in this study were women (73.6%), and the patients’ mean age was 54.8 years (range, 40-64 years). Patients had newly diagnosed breast cancer (52.2%), colorectal cancer (21.3%), and lung cancer (26.5%).
The benefits of Medicaid expansion persisted after adjustment for education, income, insurance, and race.
The lower mortality in expansion states compared with nonexpansion states was similar across all three cancer types. However, in stratified analyses, the difference was significant only for lung cancer (P = .03).
“Lung cancer has a higher mortality rate than breast and colorectal cancer, and with longer follow-up, it is possible that the lower mortality rates seen for breast and colorectal cancer may also become significant,” the authors wrote.
This research was funded by Harvard Catalyst, the Harvard Clinical and Translational Science Center, and the National Center for Advancing Translational Sciences at the National Institutes of Health. The investigators and Dr. Drake had no relevant disclosures.
SOURCE: Lam MB et al. JAMA Netw Open. 2020 Nov 2;3(11):e2024366.
FROM JAMA OPEN NETWORK
50.6 million tobacco users are not a homogeneous group
Cigarettes are still the product of choice among U.S. adults who use tobacco, but the youngest adults are more likely to use e-cigarettes than any other product, according to data from the 2019 National Health Interview Survey.
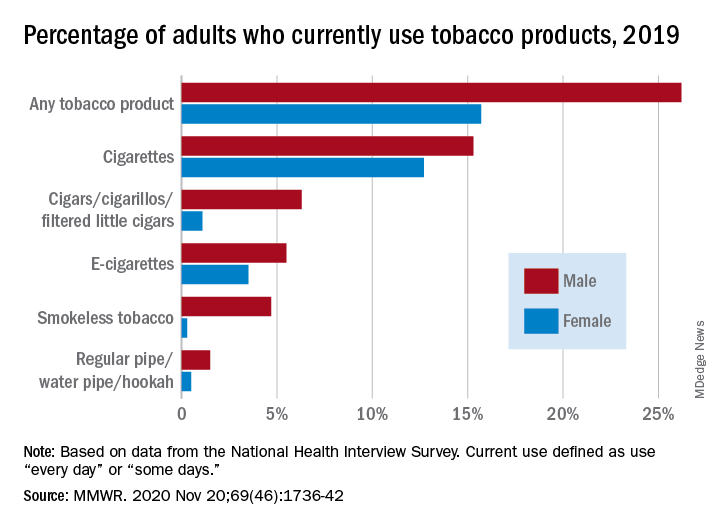
with cigarette use reported by the largest share of respondents (14.0%) and e-cigarettes next at 4.5%, Monica E. Cornelius, PhD, and associates said in the Morbidity and Mortality Weekly Report.
Among adults aged 18-24 years, however, e-cigarettes were used by 9.3% of respondents in 2019, compared with 8.0% who used cigarettes every day or some days. Current e-cigarette use was 6.4% in 25- to 44-year-olds and continued to diminish with increasing age, said Dr. Cornelius and associates at the Centers for Disease Control and Prevention’s National Center for Chronic Disease Prevention and Health Promotion.
Men were more likely than women to use e-cigarettes (5.5% vs. 3.5%), and to use any tobacco product (26.2% vs. 15.7%). Use of other products, including cigarettes (15.3% for men vs. 12.7% for women), followed the same pattern to varying degrees, the national survey data show.
“Differences in prevalence of tobacco use also were also seen across population groups, with higher prevalence among those with a [high school equivalency degree], American Indian/Alaska Natives, uninsured adults and adults with Medicaid, and [lesbian, gay, or bisexual] adults,” the investigators said.
Among those groups, overall tobacco use and cigarette use were highest in those with an equivalency degree (43.8%, 37.1%), while lesbian/gay/bisexual individuals had the highest prevalence of e-cigarette use at 11.5%, they reported.
“As part of a comprehensive approach” to reduce tobacco-related disease and death, Dr. Cornelius and associates suggested, “targeted interventions are also warranted to reach subpopulations with the highest prevalence of use, which might vary by tobacco product type.”
SOURCE: Cornelius ME et al. MMWR. 2020 Nov 20;69(46);1736-42.
Cigarettes are still the product of choice among U.S. adults who use tobacco, but the youngest adults are more likely to use e-cigarettes than any other product, according to data from the 2019 National Health Interview Survey.

with cigarette use reported by the largest share of respondents (14.0%) and e-cigarettes next at 4.5%, Monica E. Cornelius, PhD, and associates said in the Morbidity and Mortality Weekly Report.
Among adults aged 18-24 years, however, e-cigarettes were used by 9.3% of respondents in 2019, compared with 8.0% who used cigarettes every day or some days. Current e-cigarette use was 6.4% in 25- to 44-year-olds and continued to diminish with increasing age, said Dr. Cornelius and associates at the Centers for Disease Control and Prevention’s National Center for Chronic Disease Prevention and Health Promotion.
Men were more likely than women to use e-cigarettes (5.5% vs. 3.5%), and to use any tobacco product (26.2% vs. 15.7%). Use of other products, including cigarettes (15.3% for men vs. 12.7% for women), followed the same pattern to varying degrees, the national survey data show.
“Differences in prevalence of tobacco use also were also seen across population groups, with higher prevalence among those with a [high school equivalency degree], American Indian/Alaska Natives, uninsured adults and adults with Medicaid, and [lesbian, gay, or bisexual] adults,” the investigators said.
Among those groups, overall tobacco use and cigarette use were highest in those with an equivalency degree (43.8%, 37.1%), while lesbian/gay/bisexual individuals had the highest prevalence of e-cigarette use at 11.5%, they reported.
“As part of a comprehensive approach” to reduce tobacco-related disease and death, Dr. Cornelius and associates suggested, “targeted interventions are also warranted to reach subpopulations with the highest prevalence of use, which might vary by tobacco product type.”
SOURCE: Cornelius ME et al. MMWR. 2020 Nov 20;69(46);1736-42.
Cigarettes are still the product of choice among U.S. adults who use tobacco, but the youngest adults are more likely to use e-cigarettes than any other product, according to data from the 2019 National Health Interview Survey.

with cigarette use reported by the largest share of respondents (14.0%) and e-cigarettes next at 4.5%, Monica E. Cornelius, PhD, and associates said in the Morbidity and Mortality Weekly Report.
Among adults aged 18-24 years, however, e-cigarettes were used by 9.3% of respondents in 2019, compared with 8.0% who used cigarettes every day or some days. Current e-cigarette use was 6.4% in 25- to 44-year-olds and continued to diminish with increasing age, said Dr. Cornelius and associates at the Centers for Disease Control and Prevention’s National Center for Chronic Disease Prevention and Health Promotion.
Men were more likely than women to use e-cigarettes (5.5% vs. 3.5%), and to use any tobacco product (26.2% vs. 15.7%). Use of other products, including cigarettes (15.3% for men vs. 12.7% for women), followed the same pattern to varying degrees, the national survey data show.
“Differences in prevalence of tobacco use also were also seen across population groups, with higher prevalence among those with a [high school equivalency degree], American Indian/Alaska Natives, uninsured adults and adults with Medicaid, and [lesbian, gay, or bisexual] adults,” the investigators said.
Among those groups, overall tobacco use and cigarette use were highest in those with an equivalency degree (43.8%, 37.1%), while lesbian/gay/bisexual individuals had the highest prevalence of e-cigarette use at 11.5%, they reported.
“As part of a comprehensive approach” to reduce tobacco-related disease and death, Dr. Cornelius and associates suggested, “targeted interventions are also warranted to reach subpopulations with the highest prevalence of use, which might vary by tobacco product type.”
SOURCE: Cornelius ME et al. MMWR. 2020 Nov 20;69(46);1736-42.
FROM MMWR
New findings on ‘exceptional responders’ to cancer therapies
An ongoing research project is studying why some patients have exceptional responses. The researchers have found particular molecular features in the tumors of about a quarter of these patients. In some cases, there are multiple rare genetic changes in the tumor genome. In other cases, the tumors are infiltrated with certain types of immune cells.
The findings were published online November 19 in Cancer Cell. They come from a genomic analysis of tumor biopsy specimens from 111 patients who were identified by the National Cancer Institute’s (NCI’s) Exceptional Responders Initiative, a national project launched in 2014.
An exceptional responder is defined as an individual who achieves a partial or complete response to a treatment that would be effective in fewer than 10% of similar patients. For exceptional response, the duration of response is at least three times longer than the usual median response time.
In this study of 111 such patients, about one quarter (24%, n = 26 patients) were found to have tumors in which there were molecular features that could potentially explain exceptional responses to treatment.
“We won’t be able to identify, in every patient, which particular drugs will be beneficial,” said Louis Staudt, MD, PhD, director of the NCI’s Center for Cancer Genomics, who co-led the study. “We are nowhere near that. But what it does say is that we have identified particular mutations, some of which we knew about in some types of cancer but can also occur less commonly in other cancer types.”
Staudt noted that these mutations can “illuminate” the path that the cancer will take — and potentially can be used to predict whether the cancer will be aggressive and will require treatment or could be managed with surveillance. This is why this research can be useful in the short term, he said.
“In the longer term, this is the kind of research that inspires future work,” he told Medscape Medical News. “That would encompass clinical trials involving drugs that target some of the pathways we found to be genetically inactivated in some of these responders.”
These results support the use of genetic testing in routine clinical care, he said.
Earlier this year, the NCI team published the results of a pilot study that affirmed the feasibility of this approach. Of the more than 100 cases that were analyzed, six were identified as involving potentially clinically actionable germline mutations.
‘Curiosity drove the research’
“We had these wonderful and gratifying experiences with our patients, so we were immediately curious how that happened, so it was pretty much that curiosity that drove a lot of this work,” said Staudt.
In the current study, Staudt and colleagues used multiple genomic methodologies to detect mutations, copy number changes, aberrant methylation, outlier gene expression, and the cellular makeup of the tumor microenvironment.
The hypothesized mechanisms for exceptional responses were broadly divided into the following four categories: DNA damage response (n = 15), intracellular signaling pathway (n = 9), prognostic genetics (n = 9), and immunologic engagement (n = 16). For many patients, two or more of these mechanisms were involved.
The authors note that the “predominance of plausible DNA damage response mechanisms parallels the frequent use of cytotoxic chemotherapy in routine cancer treatment reflected in this cohort.”
Twenty-six patients were identified as exceptional responders. Among these patients, a variety of cancer types was represented: brain (8); gastrointestinal tract (6); breast (4); cholangiocarcinoma (2); lung (2); pancreas, endometrium, ovarian, and bladder (1 each). Many of these patients (65%, n = 17) were treated with chemotherapy that included DNA-damaging agents. For more than half (54%, n = 14), targeted therapies were used, and some patients received both.
The authors highlight several patients as examples of exceptional responders:
- One patient with glioblastoma multiforme (GBM) was treated sequentially with surgery, localized carmustine, and radiotherapy. When the cancer recurred, temozolomide was administered. This induced a complete response that has lasted for more than a decade.
- A patient with metastatic colon adenocarcinoma has had an ongoing and nearly complete response that has lasted 45 months (last follow-up) after receiving temozolomide in combination with the investigational drug TRC102 (methoxyamine, under development by Tracon) in a phase 1 clinical trial. TRC102 is an inhibitor of the DNA base excision repair pathway, which is a pathway that causes resistance to alkylating and antimetabolite chemotherapeutics.
- A patient with metastatic, estrogen receptor–positive breast cancer received trastuzumab because of a high-level ERBB2 amplification, together with anastrozole. This resulted in an ongoing 2.4-year partial response.
- Although the patient was clinically HER2 positive, her tumor had exceedingly low expression of ERBB2 mRNA. Molecular profiling had classified the tumor as of the basal-like subtype rather than the HER2-enriched subtype. This meant that it was unlikely that trastuzumab contributed to the exceptional response, the authors note. Because the patient was estrogen-receptor positive, she received anastrozole, an inhibitor of aromatase (CYP19A), which converts testosterone into estradiol.
- A patient who had a gastrointestinal stromal tumor with a deletion of KIT exon 11 experienced relapse after an initial response to imatinib, which targets KIT and other tyrosine kinases, but then achieved a complete response with sunitinib. Gene expression profiling revealed high expression not only of KIT but also of genes encoding several tyrosine kinases that are targeted by sunitinib (KDR, FLT1, and FLT3). This may have accounted for the patient’s response.
Favorable genomic characteristics
The authors defined a “prognostic genetics” category of tumors, characterized by genetic lesions that are now known to be associated with a favorable prognosis but that were not addressed through routine care that these patients received when they were first diagnosed. Although the patients experienced relapse after first-line treatment, their exceptional survival after salvage therapy could be linked to favorable genomic characteristics.
For example, several of the patients with high-grade GBMs and astrocytomas had genetic lesions that are generally more common in low-grade glioma and that have been associated with an indolent clinical course following standard therapy.
The authors also assessed immune response. Examining immune cell infiltration in responder tumors in comparison with control cases, the team found that signatures of B cells and activated (CD56dim) natural killer cells were higher in exceptional-responder tumors.
In one patient with metastatic urothelial cancer who experienced disease progression after chemotherapy, radiotherapy, and surgery, treatment with nivolumab produced a complete response that lasted 7 months. Such an outcome occurs in only about 3% of bladder cancer patients. The tumor expressed high mRNA levels of PDCD1, which encodes the nivolumab target PD-1, and CD274, which encodes the PD-1 ligand PD-L1. There was also a high level of amplification of IFNG, which encodes interferon-gamma, a cytokine that has been linked to favorable response to immune checkpoint blockade.
Moving to precision medicine
“It is very valuable to be tested up front and again when the disease progresses, because there may have been some genetic changes, and this may change the treatment,” said co–lead author S. Percy Ivy, MD, of the NCI’s Division of Cancer Treatment and Diagnosis.
“The goal of this study was to understand what was unique about these patients and their genetic makeup that led them to be classified as exceptional responders, and hopefully we will be able to tease that out,” she added.
“As researchers, we have a lot to learn from these patients, and they have a lot to teach us,” she added. “In the future, they will help us as we move closer to the goal of delivering precision oncology to all of our patients. We’re not there yet, but every time we study more deeply and learn more, we are able to provide better care.”
To encourage participation in this effort by investigators around the world, the NCI team and their colleagues have made their molecular profiling results and clinical information publicly available in the NCI Genomic Data Commons.
The study was supported by the NCI’s Intramural Research Program, the National Institutes of Health, the Center for Cancer Research, and the NCI’s Center for Cancer Genomics. Staudt and Ivy have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
An ongoing research project is studying why some patients have exceptional responses. The researchers have found particular molecular features in the tumors of about a quarter of these patients. In some cases, there are multiple rare genetic changes in the tumor genome. In other cases, the tumors are infiltrated with certain types of immune cells.
The findings were published online November 19 in Cancer Cell. They come from a genomic analysis of tumor biopsy specimens from 111 patients who were identified by the National Cancer Institute’s (NCI’s) Exceptional Responders Initiative, a national project launched in 2014.
An exceptional responder is defined as an individual who achieves a partial or complete response to a treatment that would be effective in fewer than 10% of similar patients. For exceptional response, the duration of response is at least three times longer than the usual median response time.
In this study of 111 such patients, about one quarter (24%, n = 26 patients) were found to have tumors in which there were molecular features that could potentially explain exceptional responses to treatment.
“We won’t be able to identify, in every patient, which particular drugs will be beneficial,” said Louis Staudt, MD, PhD, director of the NCI’s Center for Cancer Genomics, who co-led the study. “We are nowhere near that. But what it does say is that we have identified particular mutations, some of which we knew about in some types of cancer but can also occur less commonly in other cancer types.”
Staudt noted that these mutations can “illuminate” the path that the cancer will take — and potentially can be used to predict whether the cancer will be aggressive and will require treatment or could be managed with surveillance. This is why this research can be useful in the short term, he said.
“In the longer term, this is the kind of research that inspires future work,” he told Medscape Medical News. “That would encompass clinical trials involving drugs that target some of the pathways we found to be genetically inactivated in some of these responders.”
These results support the use of genetic testing in routine clinical care, he said.
Earlier this year, the NCI team published the results of a pilot study that affirmed the feasibility of this approach. Of the more than 100 cases that were analyzed, six were identified as involving potentially clinically actionable germline mutations.
‘Curiosity drove the research’
“We had these wonderful and gratifying experiences with our patients, so we were immediately curious how that happened, so it was pretty much that curiosity that drove a lot of this work,” said Staudt.
In the current study, Staudt and colleagues used multiple genomic methodologies to detect mutations, copy number changes, aberrant methylation, outlier gene expression, and the cellular makeup of the tumor microenvironment.
The hypothesized mechanisms for exceptional responses were broadly divided into the following four categories: DNA damage response (n = 15), intracellular signaling pathway (n = 9), prognostic genetics (n = 9), and immunologic engagement (n = 16). For many patients, two or more of these mechanisms were involved.
The authors note that the “predominance of plausible DNA damage response mechanisms parallels the frequent use of cytotoxic chemotherapy in routine cancer treatment reflected in this cohort.”
Twenty-six patients were identified as exceptional responders. Among these patients, a variety of cancer types was represented: brain (8); gastrointestinal tract (6); breast (4); cholangiocarcinoma (2); lung (2); pancreas, endometrium, ovarian, and bladder (1 each). Many of these patients (65%, n = 17) were treated with chemotherapy that included DNA-damaging agents. For more than half (54%, n = 14), targeted therapies were used, and some patients received both.
The authors highlight several patients as examples of exceptional responders:
- One patient with glioblastoma multiforme (GBM) was treated sequentially with surgery, localized carmustine, and radiotherapy. When the cancer recurred, temozolomide was administered. This induced a complete response that has lasted for more than a decade.
- A patient with metastatic colon adenocarcinoma has had an ongoing and nearly complete response that has lasted 45 months (last follow-up) after receiving temozolomide in combination with the investigational drug TRC102 (methoxyamine, under development by Tracon) in a phase 1 clinical trial. TRC102 is an inhibitor of the DNA base excision repair pathway, which is a pathway that causes resistance to alkylating and antimetabolite chemotherapeutics.
- A patient with metastatic, estrogen receptor–positive breast cancer received trastuzumab because of a high-level ERBB2 amplification, together with anastrozole. This resulted in an ongoing 2.4-year partial response.
- Although the patient was clinically HER2 positive, her tumor had exceedingly low expression of ERBB2 mRNA. Molecular profiling had classified the tumor as of the basal-like subtype rather than the HER2-enriched subtype. This meant that it was unlikely that trastuzumab contributed to the exceptional response, the authors note. Because the patient was estrogen-receptor positive, she received anastrozole, an inhibitor of aromatase (CYP19A), which converts testosterone into estradiol.
- A patient who had a gastrointestinal stromal tumor with a deletion of KIT exon 11 experienced relapse after an initial response to imatinib, which targets KIT and other tyrosine kinases, but then achieved a complete response with sunitinib. Gene expression profiling revealed high expression not only of KIT but also of genes encoding several tyrosine kinases that are targeted by sunitinib (KDR, FLT1, and FLT3). This may have accounted for the patient’s response.
Favorable genomic characteristics
The authors defined a “prognostic genetics” category of tumors, characterized by genetic lesions that are now known to be associated with a favorable prognosis but that were not addressed through routine care that these patients received when they were first diagnosed. Although the patients experienced relapse after first-line treatment, their exceptional survival after salvage therapy could be linked to favorable genomic characteristics.
For example, several of the patients with high-grade GBMs and astrocytomas had genetic lesions that are generally more common in low-grade glioma and that have been associated with an indolent clinical course following standard therapy.
The authors also assessed immune response. Examining immune cell infiltration in responder tumors in comparison with control cases, the team found that signatures of B cells and activated (CD56dim) natural killer cells were higher in exceptional-responder tumors.
In one patient with metastatic urothelial cancer who experienced disease progression after chemotherapy, radiotherapy, and surgery, treatment with nivolumab produced a complete response that lasted 7 months. Such an outcome occurs in only about 3% of bladder cancer patients. The tumor expressed high mRNA levels of PDCD1, which encodes the nivolumab target PD-1, and CD274, which encodes the PD-1 ligand PD-L1. There was also a high level of amplification of IFNG, which encodes interferon-gamma, a cytokine that has been linked to favorable response to immune checkpoint blockade.
Moving to precision medicine
“It is very valuable to be tested up front and again when the disease progresses, because there may have been some genetic changes, and this may change the treatment,” said co–lead author S. Percy Ivy, MD, of the NCI’s Division of Cancer Treatment and Diagnosis.
“The goal of this study was to understand what was unique about these patients and their genetic makeup that led them to be classified as exceptional responders, and hopefully we will be able to tease that out,” she added.
“As researchers, we have a lot to learn from these patients, and they have a lot to teach us,” she added. “In the future, they will help us as we move closer to the goal of delivering precision oncology to all of our patients. We’re not there yet, but every time we study more deeply and learn more, we are able to provide better care.”
To encourage participation in this effort by investigators around the world, the NCI team and their colleagues have made their molecular profiling results and clinical information publicly available in the NCI Genomic Data Commons.
The study was supported by the NCI’s Intramural Research Program, the National Institutes of Health, the Center for Cancer Research, and the NCI’s Center for Cancer Genomics. Staudt and Ivy have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
An ongoing research project is studying why some patients have exceptional responses. The researchers have found particular molecular features in the tumors of about a quarter of these patients. In some cases, there are multiple rare genetic changes in the tumor genome. In other cases, the tumors are infiltrated with certain types of immune cells.
The findings were published online November 19 in Cancer Cell. They come from a genomic analysis of tumor biopsy specimens from 111 patients who were identified by the National Cancer Institute’s (NCI’s) Exceptional Responders Initiative, a national project launched in 2014.
An exceptional responder is defined as an individual who achieves a partial or complete response to a treatment that would be effective in fewer than 10% of similar patients. For exceptional response, the duration of response is at least three times longer than the usual median response time.
In this study of 111 such patients, about one quarter (24%, n = 26 patients) were found to have tumors in which there were molecular features that could potentially explain exceptional responses to treatment.
“We won’t be able to identify, in every patient, which particular drugs will be beneficial,” said Louis Staudt, MD, PhD, director of the NCI’s Center for Cancer Genomics, who co-led the study. “We are nowhere near that. But what it does say is that we have identified particular mutations, some of which we knew about in some types of cancer but can also occur less commonly in other cancer types.”
Staudt noted that these mutations can “illuminate” the path that the cancer will take — and potentially can be used to predict whether the cancer will be aggressive and will require treatment or could be managed with surveillance. This is why this research can be useful in the short term, he said.
“In the longer term, this is the kind of research that inspires future work,” he told Medscape Medical News. “That would encompass clinical trials involving drugs that target some of the pathways we found to be genetically inactivated in some of these responders.”
These results support the use of genetic testing in routine clinical care, he said.
Earlier this year, the NCI team published the results of a pilot study that affirmed the feasibility of this approach. Of the more than 100 cases that were analyzed, six were identified as involving potentially clinically actionable germline mutations.
‘Curiosity drove the research’
“We had these wonderful and gratifying experiences with our patients, so we were immediately curious how that happened, so it was pretty much that curiosity that drove a lot of this work,” said Staudt.
In the current study, Staudt and colleagues used multiple genomic methodologies to detect mutations, copy number changes, aberrant methylation, outlier gene expression, and the cellular makeup of the tumor microenvironment.
The hypothesized mechanisms for exceptional responses were broadly divided into the following four categories: DNA damage response (n = 15), intracellular signaling pathway (n = 9), prognostic genetics (n = 9), and immunologic engagement (n = 16). For many patients, two or more of these mechanisms were involved.
The authors note that the “predominance of plausible DNA damage response mechanisms parallels the frequent use of cytotoxic chemotherapy in routine cancer treatment reflected in this cohort.”
Twenty-six patients were identified as exceptional responders. Among these patients, a variety of cancer types was represented: brain (8); gastrointestinal tract (6); breast (4); cholangiocarcinoma (2); lung (2); pancreas, endometrium, ovarian, and bladder (1 each). Many of these patients (65%, n = 17) were treated with chemotherapy that included DNA-damaging agents. For more than half (54%, n = 14), targeted therapies were used, and some patients received both.
The authors highlight several patients as examples of exceptional responders:
- One patient with glioblastoma multiforme (GBM) was treated sequentially with surgery, localized carmustine, and radiotherapy. When the cancer recurred, temozolomide was administered. This induced a complete response that has lasted for more than a decade.
- A patient with metastatic colon adenocarcinoma has had an ongoing and nearly complete response that has lasted 45 months (last follow-up) after receiving temozolomide in combination with the investigational drug TRC102 (methoxyamine, under development by Tracon) in a phase 1 clinical trial. TRC102 is an inhibitor of the DNA base excision repair pathway, which is a pathway that causes resistance to alkylating and antimetabolite chemotherapeutics.
- A patient with metastatic, estrogen receptor–positive breast cancer received trastuzumab because of a high-level ERBB2 amplification, together with anastrozole. This resulted in an ongoing 2.4-year partial response.
- Although the patient was clinically HER2 positive, her tumor had exceedingly low expression of ERBB2 mRNA. Molecular profiling had classified the tumor as of the basal-like subtype rather than the HER2-enriched subtype. This meant that it was unlikely that trastuzumab contributed to the exceptional response, the authors note. Because the patient was estrogen-receptor positive, she received anastrozole, an inhibitor of aromatase (CYP19A), which converts testosterone into estradiol.
- A patient who had a gastrointestinal stromal tumor with a deletion of KIT exon 11 experienced relapse after an initial response to imatinib, which targets KIT and other tyrosine kinases, but then achieved a complete response with sunitinib. Gene expression profiling revealed high expression not only of KIT but also of genes encoding several tyrosine kinases that are targeted by sunitinib (KDR, FLT1, and FLT3). This may have accounted for the patient’s response.
Favorable genomic characteristics
The authors defined a “prognostic genetics” category of tumors, characterized by genetic lesions that are now known to be associated with a favorable prognosis but that were not addressed through routine care that these patients received when they were first diagnosed. Although the patients experienced relapse after first-line treatment, their exceptional survival after salvage therapy could be linked to favorable genomic characteristics.
For example, several of the patients with high-grade GBMs and astrocytomas had genetic lesions that are generally more common in low-grade glioma and that have been associated with an indolent clinical course following standard therapy.
The authors also assessed immune response. Examining immune cell infiltration in responder tumors in comparison with control cases, the team found that signatures of B cells and activated (CD56dim) natural killer cells were higher in exceptional-responder tumors.
In one patient with metastatic urothelial cancer who experienced disease progression after chemotherapy, radiotherapy, and surgery, treatment with nivolumab produced a complete response that lasted 7 months. Such an outcome occurs in only about 3% of bladder cancer patients. The tumor expressed high mRNA levels of PDCD1, which encodes the nivolumab target PD-1, and CD274, which encodes the PD-1 ligand PD-L1. There was also a high level of amplification of IFNG, which encodes interferon-gamma, a cytokine that has been linked to favorable response to immune checkpoint blockade.
Moving to precision medicine
“It is very valuable to be tested up front and again when the disease progresses, because there may have been some genetic changes, and this may change the treatment,” said co–lead author S. Percy Ivy, MD, of the NCI’s Division of Cancer Treatment and Diagnosis.
“The goal of this study was to understand what was unique about these patients and their genetic makeup that led them to be classified as exceptional responders, and hopefully we will be able to tease that out,” she added.
“As researchers, we have a lot to learn from these patients, and they have a lot to teach us,” she added. “In the future, they will help us as we move closer to the goal of delivering precision oncology to all of our patients. We’re not there yet, but every time we study more deeply and learn more, we are able to provide better care.”
To encourage participation in this effort by investigators around the world, the NCI team and their colleagues have made their molecular profiling results and clinical information publicly available in the NCI Genomic Data Commons.
The study was supported by the NCI’s Intramural Research Program, the National Institutes of Health, the Center for Cancer Research, and the NCI’s Center for Cancer Genomics. Staudt and Ivy have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Immunotherapy could fill unmet need in leptomeningeal metastases
Results from the trial were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Unfortunately, when patients present with leptomeningeal disease, they usually have a poor prognosis. Their median survival is measured at 6-24 weeks,” commented lead study author Jarushka Naidoo, MBBCh, an adjunct assistant professor of oncology at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore, and a consultant medical oncologist at Beaumont Hospital in Dublin.
“While there may be some standard approaches for how we treat leptomeningeal disease, there are no universal standard therapies that are efficacious across solid tumor types,” Dr. Naidoo added.
With this in mind, Dr. Naidoo and colleagues tested systemic pembrolizumab in a trial of patients with leptomeningeal metastases from solid tumors.
The trial closed early because of poor accrual, after enrolling 13 patients: 5 with breast carcinoma, 3 with high-grade glioma, 3 with non–small cell lung cancer, 1 with squamous cell carcinoma of the skin, and 1 with head and neck squamous carcinoma. Nine patients (69%) had received at least two prior lines of systemic therapy.
Response, safety, and biomarkers
Overall, five patients (38%) had a central nervous system response, as ascertained from radiologic response on MRI, cytologic response in cerebrospinal fluid (CSF), and/or clinical response in neurologic symptoms, Dr. Naidoo reported.
Two patients had a complete CNS response: a patient with squamous cell carcinoma of the skin, who was still alive at 3 years, and a patient with non–small cell lung cancer, who survived 9 months but succumbed to metastases elsewhere.
For the entire cohort, median CNS progression-free survival was 2.9 months, and median overall survival was 4.9 months.
“This is consistent with published prospective studies of systemic agents for leptomeningeal disease,” Dr. Naidoo pointed out. “Notably, even though numbers are small, we do see the tail-on-the-curve phenomenon in both of these survival curves, which is consistent with immune checkpoint blockade prospective studies.”
The rate of grade 3 or higher treatment-related adverse events was 15.4%, and there were no grade 3 or higher immune-related adverse events.
The number of patients was too small for formal correlational testing, but both patients who achieved a complete response developed immune-related adverse events.
The trial’s biomarker analyses showed that an aneuploidy assay using CSF tumor-derived DNA performed well at detecting leptomeningeal metastases, with sensitivity of 84.6%, compared with just 53.8% for CSF cytopathology (the current preferred method).
A multiplex assay of CSF cytokines identified similar baseline profiles for patients who went on to have responses and showed similar changes in profile (notably a reduction in proinflammatory cytokines) for the two patients who had complete responses.
Given the trial’s 38% CNS response rate, pembrolizumab “needs to be studied in larger populations of patients to confirm this result, but it could be used as a potential treatment option for patients with leptomeningeal disease from solid tumors,” Dr. Naidoo concluded. “Reassuringly, pembrolizumab was well tolerated, and this is extremely important in a patient population that is traditionally quite frail and in which other standard therapies that are used, such as high-dose methotrexate or intrathecal chemotherapy, are associated with far higher rates of toxicity.”
An unmet need
“Leptomeningeal metastasis is a strong unmet need, although its occurrence is fortunately quite rare,” commented Kim Margolin, MD, a clinical professor and medical oncologist at City of Hope National Medical Center in Duarte, Calif., who was not involved in this study.
The trial is noteworthy for showing activity of programmed death–1 (PD-1) blockade given only systemically and not with additional intrathecal therapy (as has been done in a concurrent study at MD Anderson Cancer Center) and for providing insight into various biomarkers, Dr. Margolin said in an interview.
“I cannot take a stand on author conclusions other than to agree it warrants further evaluation in carefully selected patients, and it would be great to compare something like peripheral PD-1 blockade alone versus in combination with intrathecal therapy versus a combination such as CTLA4 blockade plus PD-1 blockade such as our group and others have shown to have increased activity in CNS metastases over PD-1 block alone,” Dr. Margolin said.
“The drugs in this class are already approved, so there is no reason not to try them,” she noted.
However, patients with leptomeningeal metastases of melanoma, for example, are likely to have already received anti-PD-1 immunotherapy.
“So the settings in which off-the-shelf PD-1 blockade would be useful are extremely limited,” she concluded.
The current trial was funded by Merck, the National Institutes of Health, the Lung Cancer Foundation of America, the International Association for the Study of Lung Cancer, and Johns Hopkins University Seed Grants. Dr. Naidoo disclosed relationships with AstraZeneca, Merck, Bristol Myers Squibb, and Roche/Genentech. Dr. Margolin disclosed no relevant conflicts of interest.
SOURCE: Naidoo J et al. SITC 2020, Abstract 788.
Results from the trial were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Unfortunately, when patients present with leptomeningeal disease, they usually have a poor prognosis. Their median survival is measured at 6-24 weeks,” commented lead study author Jarushka Naidoo, MBBCh, an adjunct assistant professor of oncology at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore, and a consultant medical oncologist at Beaumont Hospital in Dublin.
“While there may be some standard approaches for how we treat leptomeningeal disease, there are no universal standard therapies that are efficacious across solid tumor types,” Dr. Naidoo added.
With this in mind, Dr. Naidoo and colleagues tested systemic pembrolizumab in a trial of patients with leptomeningeal metastases from solid tumors.
The trial closed early because of poor accrual, after enrolling 13 patients: 5 with breast carcinoma, 3 with high-grade glioma, 3 with non–small cell lung cancer, 1 with squamous cell carcinoma of the skin, and 1 with head and neck squamous carcinoma. Nine patients (69%) had received at least two prior lines of systemic therapy.
Response, safety, and biomarkers
Overall, five patients (38%) had a central nervous system response, as ascertained from radiologic response on MRI, cytologic response in cerebrospinal fluid (CSF), and/or clinical response in neurologic symptoms, Dr. Naidoo reported.
Two patients had a complete CNS response: a patient with squamous cell carcinoma of the skin, who was still alive at 3 years, and a patient with non–small cell lung cancer, who survived 9 months but succumbed to metastases elsewhere.
For the entire cohort, median CNS progression-free survival was 2.9 months, and median overall survival was 4.9 months.
“This is consistent with published prospective studies of systemic agents for leptomeningeal disease,” Dr. Naidoo pointed out. “Notably, even though numbers are small, we do see the tail-on-the-curve phenomenon in both of these survival curves, which is consistent with immune checkpoint blockade prospective studies.”
The rate of grade 3 or higher treatment-related adverse events was 15.4%, and there were no grade 3 or higher immune-related adverse events.
The number of patients was too small for formal correlational testing, but both patients who achieved a complete response developed immune-related adverse events.
The trial’s biomarker analyses showed that an aneuploidy assay using CSF tumor-derived DNA performed well at detecting leptomeningeal metastases, with sensitivity of 84.6%, compared with just 53.8% for CSF cytopathology (the current preferred method).
A multiplex assay of CSF cytokines identified similar baseline profiles for patients who went on to have responses and showed similar changes in profile (notably a reduction in proinflammatory cytokines) for the two patients who had complete responses.
Given the trial’s 38% CNS response rate, pembrolizumab “needs to be studied in larger populations of patients to confirm this result, but it could be used as a potential treatment option for patients with leptomeningeal disease from solid tumors,” Dr. Naidoo concluded. “Reassuringly, pembrolizumab was well tolerated, and this is extremely important in a patient population that is traditionally quite frail and in which other standard therapies that are used, such as high-dose methotrexate or intrathecal chemotherapy, are associated with far higher rates of toxicity.”
An unmet need
“Leptomeningeal metastasis is a strong unmet need, although its occurrence is fortunately quite rare,” commented Kim Margolin, MD, a clinical professor and medical oncologist at City of Hope National Medical Center in Duarte, Calif., who was not involved in this study.
The trial is noteworthy for showing activity of programmed death–1 (PD-1) blockade given only systemically and not with additional intrathecal therapy (as has been done in a concurrent study at MD Anderson Cancer Center) and for providing insight into various biomarkers, Dr. Margolin said in an interview.
“I cannot take a stand on author conclusions other than to agree it warrants further evaluation in carefully selected patients, and it would be great to compare something like peripheral PD-1 blockade alone versus in combination with intrathecal therapy versus a combination such as CTLA4 blockade plus PD-1 blockade such as our group and others have shown to have increased activity in CNS metastases over PD-1 block alone,” Dr. Margolin said.
“The drugs in this class are already approved, so there is no reason not to try them,” she noted.
However, patients with leptomeningeal metastases of melanoma, for example, are likely to have already received anti-PD-1 immunotherapy.
“So the settings in which off-the-shelf PD-1 blockade would be useful are extremely limited,” she concluded.
The current trial was funded by Merck, the National Institutes of Health, the Lung Cancer Foundation of America, the International Association for the Study of Lung Cancer, and Johns Hopkins University Seed Grants. Dr. Naidoo disclosed relationships with AstraZeneca, Merck, Bristol Myers Squibb, and Roche/Genentech. Dr. Margolin disclosed no relevant conflicts of interest.
SOURCE: Naidoo J et al. SITC 2020, Abstract 788.
Results from the trial were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Unfortunately, when patients present with leptomeningeal disease, they usually have a poor prognosis. Their median survival is measured at 6-24 weeks,” commented lead study author Jarushka Naidoo, MBBCh, an adjunct assistant professor of oncology at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore, and a consultant medical oncologist at Beaumont Hospital in Dublin.
“While there may be some standard approaches for how we treat leptomeningeal disease, there are no universal standard therapies that are efficacious across solid tumor types,” Dr. Naidoo added.
With this in mind, Dr. Naidoo and colleagues tested systemic pembrolizumab in a trial of patients with leptomeningeal metastases from solid tumors.
The trial closed early because of poor accrual, after enrolling 13 patients: 5 with breast carcinoma, 3 with high-grade glioma, 3 with non–small cell lung cancer, 1 with squamous cell carcinoma of the skin, and 1 with head and neck squamous carcinoma. Nine patients (69%) had received at least two prior lines of systemic therapy.
Response, safety, and biomarkers
Overall, five patients (38%) had a central nervous system response, as ascertained from radiologic response on MRI, cytologic response in cerebrospinal fluid (CSF), and/or clinical response in neurologic symptoms, Dr. Naidoo reported.
Two patients had a complete CNS response: a patient with squamous cell carcinoma of the skin, who was still alive at 3 years, and a patient with non–small cell lung cancer, who survived 9 months but succumbed to metastases elsewhere.
For the entire cohort, median CNS progression-free survival was 2.9 months, and median overall survival was 4.9 months.
“This is consistent with published prospective studies of systemic agents for leptomeningeal disease,” Dr. Naidoo pointed out. “Notably, even though numbers are small, we do see the tail-on-the-curve phenomenon in both of these survival curves, which is consistent with immune checkpoint blockade prospective studies.”
The rate of grade 3 or higher treatment-related adverse events was 15.4%, and there were no grade 3 or higher immune-related adverse events.
The number of patients was too small for formal correlational testing, but both patients who achieved a complete response developed immune-related adverse events.
The trial’s biomarker analyses showed that an aneuploidy assay using CSF tumor-derived DNA performed well at detecting leptomeningeal metastases, with sensitivity of 84.6%, compared with just 53.8% for CSF cytopathology (the current preferred method).
A multiplex assay of CSF cytokines identified similar baseline profiles for patients who went on to have responses and showed similar changes in profile (notably a reduction in proinflammatory cytokines) for the two patients who had complete responses.
Given the trial’s 38% CNS response rate, pembrolizumab “needs to be studied in larger populations of patients to confirm this result, but it could be used as a potential treatment option for patients with leptomeningeal disease from solid tumors,” Dr. Naidoo concluded. “Reassuringly, pembrolizumab was well tolerated, and this is extremely important in a patient population that is traditionally quite frail and in which other standard therapies that are used, such as high-dose methotrexate or intrathecal chemotherapy, are associated with far higher rates of toxicity.”
An unmet need
“Leptomeningeal metastasis is a strong unmet need, although its occurrence is fortunately quite rare,” commented Kim Margolin, MD, a clinical professor and medical oncologist at City of Hope National Medical Center in Duarte, Calif., who was not involved in this study.
The trial is noteworthy for showing activity of programmed death–1 (PD-1) blockade given only systemically and not with additional intrathecal therapy (as has been done in a concurrent study at MD Anderson Cancer Center) and for providing insight into various biomarkers, Dr. Margolin said in an interview.
“I cannot take a stand on author conclusions other than to agree it warrants further evaluation in carefully selected patients, and it would be great to compare something like peripheral PD-1 blockade alone versus in combination with intrathecal therapy versus a combination such as CTLA4 blockade plus PD-1 blockade such as our group and others have shown to have increased activity in CNS metastases over PD-1 block alone,” Dr. Margolin said.
“The drugs in this class are already approved, so there is no reason not to try them,” she noted.
However, patients with leptomeningeal metastases of melanoma, for example, are likely to have already received anti-PD-1 immunotherapy.
“So the settings in which off-the-shelf PD-1 blockade would be useful are extremely limited,” she concluded.
The current trial was funded by Merck, the National Institutes of Health, the Lung Cancer Foundation of America, the International Association for the Study of Lung Cancer, and Johns Hopkins University Seed Grants. Dr. Naidoo disclosed relationships with AstraZeneca, Merck, Bristol Myers Squibb, and Roche/Genentech. Dr. Margolin disclosed no relevant conflicts of interest.
SOURCE: Naidoo J et al. SITC 2020, Abstract 788.
FROM SITC 2020
Risk factors for severe immune-related AEs identified
The first nationwide study of severe immune-related adverse events among cancer patients treated with immune checkpoint inhibitors helps identify those at elevated risk. The findings were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Immune-related adverse events are a very serious side effect of immune checkpoint inhibitor therapy, and as this therapy has become more common for treating advanced cancers, the incidence of immune-related adverse events has increased as well,” said presenting author William Murphy, a dual MD and MBA student at Harvard Medical School and Harvard Business School, both in Boston.
“However, because there is no ICD code for immune-related adverse events, it’s very difficult to study them at a population level. Most of the current literature around the incidence of immune-related adverse events and factors that are predictive of incidence are based on clinical trials and small studies,” Mr. Murphy noted.
He and his colleagues analyzed claims data from a U.S. nationwide health insurance plan for 14,378 patients who had a primary cancer and received at least one administration of an immune checkpoint inhibitor – an inhibitor of PD-1, PD-L1, or CTLA4 – during 2011-2019.
Over 19,117 patient-years of follow-up, 504 patients (3.5%) developed a severe immune-related adverse event (irAE), defined as one occurring within 2 years of their treatment and requiring inpatient hospitalization and new immunosuppression.
The incidence of severe irAEs per patient treatment year was 2.6% overall, rising from 0% in 2011 to 3.7% in 2016.
In multivariate analysis, patients had an elevated risk of severe irAEs if they received combination immunotherapy as compared with monotherapy (odds ratio, 2.44; P < .001).
On the other hand, risk fell with advancing age (OR, 0.98 per additional year; P < .001). And risk was lower for patients with melanoma (OR, 0.70; P = .01), renal cell carcinoma (OR, 0.71; P = .03), and other cancers (OR, 0.50; P < .001), compared with lung cancer.
Sex, geographic region, income, employment status, and comorbidity were not significantly associated with the risk of severe irAEs.
“We hope that patients and providers can use this evidence from a nationwide study of severe irAEs to guide treatment and management decisions,” Mr. Murphy concluded.
Real-world evidence
“As the use of immune checkpoint inhibitors increases for patients with a variety of different tumor types, there is increasing need for population-level evidence for patients treated outside of clinical trials,” said Allison Betof Warner, MD, PhD, an assistant attending physician with the melanoma service at Memorial Sloan Kettering Cancer Center in New York.
“This is a well-conducted study with an innovative approach to using real-world evidence to examine immune-related adverse events,” she added.
To her knowledge, it is the first study to look at multiple cancers for which immunotherapy is approved, Dr. Betof Warner said. This approach resulted in a large patient sample, giving power to detect differences between groups.
“The authors’ finding that combination immunotherapy is associated with more severe irAEs is in line with our clinical experience and other data sets, and the data regarding increased odds of severe irAEs in younger patients and those with lung cancer raise interesting biological questions about the etiology of irAEs,” Dr. Betof Warner noted.
However, certain factors complicate interpretation of the study’s findings, she cautioned. One such factor is requiring hospitalization to define an irAE.
“Practice patterns regarding hospitalization vary quite widely from center to center. For example, in some centers, all patients with immune-mediated colitis are hospitalized, whereas in others, these patients are managed predominantly in the outpatient setting, even in cases of high-grade toxicity,” she explained. “Practice patterns have also changed drastically over time as oncologists have grown more comfortable managing immune-related adverse events.”
Another factor is potential confounding. For example, patients with melanoma are more likely to receive combination immunotherapy given its longstanding approval for this cancer, whereas it is comparatively new for other cancers. Also, age may differ across cancers.
“The data the authors have provided are a great starting point, but I think further analysis is needed before these observations can be validated and integrated into practice,” Dr. Betof Warner concluded.
This study did not receive any specific funding. Mr. Murphy and Dr. Betof Warner disclosed no relevant conflicts of interest.
SOURCE: Murphy W et al. SITC 2020, Abstract 854.
The first nationwide study of severe immune-related adverse events among cancer patients treated with immune checkpoint inhibitors helps identify those at elevated risk. The findings were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Immune-related adverse events are a very serious side effect of immune checkpoint inhibitor therapy, and as this therapy has become more common for treating advanced cancers, the incidence of immune-related adverse events has increased as well,” said presenting author William Murphy, a dual MD and MBA student at Harvard Medical School and Harvard Business School, both in Boston.
“However, because there is no ICD code for immune-related adverse events, it’s very difficult to study them at a population level. Most of the current literature around the incidence of immune-related adverse events and factors that are predictive of incidence are based on clinical trials and small studies,” Mr. Murphy noted.
He and his colleagues analyzed claims data from a U.S. nationwide health insurance plan for 14,378 patients who had a primary cancer and received at least one administration of an immune checkpoint inhibitor – an inhibitor of PD-1, PD-L1, or CTLA4 – during 2011-2019.
Over 19,117 patient-years of follow-up, 504 patients (3.5%) developed a severe immune-related adverse event (irAE), defined as one occurring within 2 years of their treatment and requiring inpatient hospitalization and new immunosuppression.
The incidence of severe irAEs per patient treatment year was 2.6% overall, rising from 0% in 2011 to 3.7% in 2016.
In multivariate analysis, patients had an elevated risk of severe irAEs if they received combination immunotherapy as compared with monotherapy (odds ratio, 2.44; P < .001).
On the other hand, risk fell with advancing age (OR, 0.98 per additional year; P < .001). And risk was lower for patients with melanoma (OR, 0.70; P = .01), renal cell carcinoma (OR, 0.71; P = .03), and other cancers (OR, 0.50; P < .001), compared with lung cancer.
Sex, geographic region, income, employment status, and comorbidity were not significantly associated with the risk of severe irAEs.
“We hope that patients and providers can use this evidence from a nationwide study of severe irAEs to guide treatment and management decisions,” Mr. Murphy concluded.
Real-world evidence
“As the use of immune checkpoint inhibitors increases for patients with a variety of different tumor types, there is increasing need for population-level evidence for patients treated outside of clinical trials,” said Allison Betof Warner, MD, PhD, an assistant attending physician with the melanoma service at Memorial Sloan Kettering Cancer Center in New York.
“This is a well-conducted study with an innovative approach to using real-world evidence to examine immune-related adverse events,” she added.
To her knowledge, it is the first study to look at multiple cancers for which immunotherapy is approved, Dr. Betof Warner said. This approach resulted in a large patient sample, giving power to detect differences between groups.
“The authors’ finding that combination immunotherapy is associated with more severe irAEs is in line with our clinical experience and other data sets, and the data regarding increased odds of severe irAEs in younger patients and those with lung cancer raise interesting biological questions about the etiology of irAEs,” Dr. Betof Warner noted.
However, certain factors complicate interpretation of the study’s findings, she cautioned. One such factor is requiring hospitalization to define an irAE.
“Practice patterns regarding hospitalization vary quite widely from center to center. For example, in some centers, all patients with immune-mediated colitis are hospitalized, whereas in others, these patients are managed predominantly in the outpatient setting, even in cases of high-grade toxicity,” she explained. “Practice patterns have also changed drastically over time as oncologists have grown more comfortable managing immune-related adverse events.”
Another factor is potential confounding. For example, patients with melanoma are more likely to receive combination immunotherapy given its longstanding approval for this cancer, whereas it is comparatively new for other cancers. Also, age may differ across cancers.
“The data the authors have provided are a great starting point, but I think further analysis is needed before these observations can be validated and integrated into practice,” Dr. Betof Warner concluded.
This study did not receive any specific funding. Mr. Murphy and Dr. Betof Warner disclosed no relevant conflicts of interest.
SOURCE: Murphy W et al. SITC 2020, Abstract 854.
The first nationwide study of severe immune-related adverse events among cancer patients treated with immune checkpoint inhibitors helps identify those at elevated risk. The findings were reported at the Society for Immunotherapy of Cancer’s 35th Anniversary Annual Meeting.
“Immune-related adverse events are a very serious side effect of immune checkpoint inhibitor therapy, and as this therapy has become more common for treating advanced cancers, the incidence of immune-related adverse events has increased as well,” said presenting author William Murphy, a dual MD and MBA student at Harvard Medical School and Harvard Business School, both in Boston.
“However, because there is no ICD code for immune-related adverse events, it’s very difficult to study them at a population level. Most of the current literature around the incidence of immune-related adverse events and factors that are predictive of incidence are based on clinical trials and small studies,” Mr. Murphy noted.
He and his colleagues analyzed claims data from a U.S. nationwide health insurance plan for 14,378 patients who had a primary cancer and received at least one administration of an immune checkpoint inhibitor – an inhibitor of PD-1, PD-L1, or CTLA4 – during 2011-2019.
Over 19,117 patient-years of follow-up, 504 patients (3.5%) developed a severe immune-related adverse event (irAE), defined as one occurring within 2 years of their treatment and requiring inpatient hospitalization and new immunosuppression.
The incidence of severe irAEs per patient treatment year was 2.6% overall, rising from 0% in 2011 to 3.7% in 2016.
In multivariate analysis, patients had an elevated risk of severe irAEs if they received combination immunotherapy as compared with monotherapy (odds ratio, 2.44; P < .001).
On the other hand, risk fell with advancing age (OR, 0.98 per additional year; P < .001). And risk was lower for patients with melanoma (OR, 0.70; P = .01), renal cell carcinoma (OR, 0.71; P = .03), and other cancers (OR, 0.50; P < .001), compared with lung cancer.
Sex, geographic region, income, employment status, and comorbidity were not significantly associated with the risk of severe irAEs.
“We hope that patients and providers can use this evidence from a nationwide study of severe irAEs to guide treatment and management decisions,” Mr. Murphy concluded.
Real-world evidence
“As the use of immune checkpoint inhibitors increases for patients with a variety of different tumor types, there is increasing need for population-level evidence for patients treated outside of clinical trials,” said Allison Betof Warner, MD, PhD, an assistant attending physician with the melanoma service at Memorial Sloan Kettering Cancer Center in New York.
“This is a well-conducted study with an innovative approach to using real-world evidence to examine immune-related adverse events,” she added.
To her knowledge, it is the first study to look at multiple cancers for which immunotherapy is approved, Dr. Betof Warner said. This approach resulted in a large patient sample, giving power to detect differences between groups.
“The authors’ finding that combination immunotherapy is associated with more severe irAEs is in line with our clinical experience and other data sets, and the data regarding increased odds of severe irAEs in younger patients and those with lung cancer raise interesting biological questions about the etiology of irAEs,” Dr. Betof Warner noted.
However, certain factors complicate interpretation of the study’s findings, she cautioned. One such factor is requiring hospitalization to define an irAE.
“Practice patterns regarding hospitalization vary quite widely from center to center. For example, in some centers, all patients with immune-mediated colitis are hospitalized, whereas in others, these patients are managed predominantly in the outpatient setting, even in cases of high-grade toxicity,” she explained. “Practice patterns have also changed drastically over time as oncologists have grown more comfortable managing immune-related adverse events.”
Another factor is potential confounding. For example, patients with melanoma are more likely to receive combination immunotherapy given its longstanding approval for this cancer, whereas it is comparatively new for other cancers. Also, age may differ across cancers.
“The data the authors have provided are a great starting point, but I think further analysis is needed before these observations can be validated and integrated into practice,” Dr. Betof Warner concluded.
This study did not receive any specific funding. Mr. Murphy and Dr. Betof Warner disclosed no relevant conflicts of interest.
SOURCE: Murphy W et al. SITC 2020, Abstract 854.
FROM SITC 2020
Using telehealth to deliver palliative care to cancer patients
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
FROM ASCO QUALITY CARE SYMPOSIUM 2020
Lung cancer: Proton beam radiotherapy likely reduces cardiovascular events
The findings were reported at the American Society for Radiation Oncology Annual Meeting 2020.
Patients with lung cancer often have underlying cardiac risk factors, noted lead investigator Timothy Kegelman, MD, PhD, of University of Pennsylvania in Philadelphia.
“The dose to the heart correlates with adverse cardiovascular events following radiation therapy. One strategy to minimize dose to the heart is proton beam radiation,” Dr. Kegelman said.
He and his colleagues retrospectively studied consecutive patients with locally advanced non–small cell lung cancer (NSCLC) treated with chemotherapy plus either proton beam radiotherapy or conventional photon radiotherapy.
The team used electronic health records to ascertain incidence of six cardiovascular events: MI, atrial fibrillation, coronary artery disease, heart failure, stroke, and transient ischemic attack. Patients who had previously experienced an event were not considered as part of the at-risk population for that specific event after radiotherapy.
Analyses were based on 98 patients who received proton beam radiotherapy and 104 patients who received conventional photon radiotherapy.
At baseline, the proton cohort was older, had a heavier smoking history, and had a higher prevalence of previous cardiovascular events (46.9% vs. 31.7%; P = .03).
The total median radiation dose was identical for the proton and photon groups (66.6 Gy), but the former group had significantly lower measures of cardiac radiation dose, including roughly half the mean dose to the heart (6.9 vs. 13.3 Gy).
Outcomes and next steps
At a median follow-up of 29 months, the proton beam radiotherapy group had a significantly lower incidence of transient ischemic attack, compared with the photon radiotherapy group (1.1% vs. 8.2%; P = .04).
The proton group also had numerically lower incidences of MI (2.3% vs. 9.0%; P = .06) and stroke (3.2% vs. 6.1%; P = .50).
The proton and photon groups were similar as far as the incidence of total cardiovascular events (53.1% vs. 47.1%; P = .48) and the 3-year overall survival rate (38.8% vs. 42.1%; P = .99).
“Our future studies aim to examine the potential relationships between grade of cardiac event and type of radiotherapy and dose to cardiac substructures,” Dr. Kegelman commented.
In addition, his institution is participating in RTOG 1308, a phase 3 trial comparing photon and proton beam radiotherapy in patients with inoperable lung cancer that will better assess cardiac-related morbidity and mortality. The trial is expected to be completed by the end of 2025.
Accumulating evidence
“This study adds to a growing body of evidence about the potential importance of heart dose in any radiation modality,” said Daniel Gomez, MD, MBA, of Memorial Sloan Kettering Cancer Center in New York, who was not involved in the study.
The RTOG 0617 trial and the Lung ART trial previously showed correlations between lower radiation dose to the heart and better survival in patients with lung cancer, Dr. Gomez noted.
“It’s been well established that protons can improve heart dose, and therefore it’s been inferred that they may improve outcomes, but the exact mechanisms remain unclear,” Dr. Gomez said.
Proton beam radiotherapy performed well in a single-arm, phase 2 trial among patients with unresectable NSCLC.
“The ongoing phase 3 trial is using a more modern proton technique and has a larger population, with a randomized study design. It will be much more informative,” Dr. Gomez predicted.
The current study did not receive specific funding. Dr. Kegelman disclosed no relevant conflicts of interest. Dr. Gomez disclosed honoraria from Varian.
SOURCE: Kegelman TP et al. ASTRO 2020, Abstract 1046.
The findings were reported at the American Society for Radiation Oncology Annual Meeting 2020.
Patients with lung cancer often have underlying cardiac risk factors, noted lead investigator Timothy Kegelman, MD, PhD, of University of Pennsylvania in Philadelphia.
“The dose to the heart correlates with adverse cardiovascular events following radiation therapy. One strategy to minimize dose to the heart is proton beam radiation,” Dr. Kegelman said.
He and his colleagues retrospectively studied consecutive patients with locally advanced non–small cell lung cancer (NSCLC) treated with chemotherapy plus either proton beam radiotherapy or conventional photon radiotherapy.
The team used electronic health records to ascertain incidence of six cardiovascular events: MI, atrial fibrillation, coronary artery disease, heart failure, stroke, and transient ischemic attack. Patients who had previously experienced an event were not considered as part of the at-risk population for that specific event after radiotherapy.
Analyses were based on 98 patients who received proton beam radiotherapy and 104 patients who received conventional photon radiotherapy.
At baseline, the proton cohort was older, had a heavier smoking history, and had a higher prevalence of previous cardiovascular events (46.9% vs. 31.7%; P = .03).
The total median radiation dose was identical for the proton and photon groups (66.6 Gy), but the former group had significantly lower measures of cardiac radiation dose, including roughly half the mean dose to the heart (6.9 vs. 13.3 Gy).
Outcomes and next steps
At a median follow-up of 29 months, the proton beam radiotherapy group had a significantly lower incidence of transient ischemic attack, compared with the photon radiotherapy group (1.1% vs. 8.2%; P = .04).
The proton group also had numerically lower incidences of MI (2.3% vs. 9.0%; P = .06) and stroke (3.2% vs. 6.1%; P = .50).
The proton and photon groups were similar as far as the incidence of total cardiovascular events (53.1% vs. 47.1%; P = .48) and the 3-year overall survival rate (38.8% vs. 42.1%; P = .99).
“Our future studies aim to examine the potential relationships between grade of cardiac event and type of radiotherapy and dose to cardiac substructures,” Dr. Kegelman commented.
In addition, his institution is participating in RTOG 1308, a phase 3 trial comparing photon and proton beam radiotherapy in patients with inoperable lung cancer that will better assess cardiac-related morbidity and mortality. The trial is expected to be completed by the end of 2025.
Accumulating evidence
“This study adds to a growing body of evidence about the potential importance of heart dose in any radiation modality,” said Daniel Gomez, MD, MBA, of Memorial Sloan Kettering Cancer Center in New York, who was not involved in the study.
The RTOG 0617 trial and the Lung ART trial previously showed correlations between lower radiation dose to the heart and better survival in patients with lung cancer, Dr. Gomez noted.
“It’s been well established that protons can improve heart dose, and therefore it’s been inferred that they may improve outcomes, but the exact mechanisms remain unclear,” Dr. Gomez said.
Proton beam radiotherapy performed well in a single-arm, phase 2 trial among patients with unresectable NSCLC.
“The ongoing phase 3 trial is using a more modern proton technique and has a larger population, with a randomized study design. It will be much more informative,” Dr. Gomez predicted.
The current study did not receive specific funding. Dr. Kegelman disclosed no relevant conflicts of interest. Dr. Gomez disclosed honoraria from Varian.
SOURCE: Kegelman TP et al. ASTRO 2020, Abstract 1046.
The findings were reported at the American Society for Radiation Oncology Annual Meeting 2020.
Patients with lung cancer often have underlying cardiac risk factors, noted lead investigator Timothy Kegelman, MD, PhD, of University of Pennsylvania in Philadelphia.
“The dose to the heart correlates with adverse cardiovascular events following radiation therapy. One strategy to minimize dose to the heart is proton beam radiation,” Dr. Kegelman said.
He and his colleagues retrospectively studied consecutive patients with locally advanced non–small cell lung cancer (NSCLC) treated with chemotherapy plus either proton beam radiotherapy or conventional photon radiotherapy.
The team used electronic health records to ascertain incidence of six cardiovascular events: MI, atrial fibrillation, coronary artery disease, heart failure, stroke, and transient ischemic attack. Patients who had previously experienced an event were not considered as part of the at-risk population for that specific event after radiotherapy.
Analyses were based on 98 patients who received proton beam radiotherapy and 104 patients who received conventional photon radiotherapy.
At baseline, the proton cohort was older, had a heavier smoking history, and had a higher prevalence of previous cardiovascular events (46.9% vs. 31.7%; P = .03).
The total median radiation dose was identical for the proton and photon groups (66.6 Gy), but the former group had significantly lower measures of cardiac radiation dose, including roughly half the mean dose to the heart (6.9 vs. 13.3 Gy).
Outcomes and next steps
At a median follow-up of 29 months, the proton beam radiotherapy group had a significantly lower incidence of transient ischemic attack, compared with the photon radiotherapy group (1.1% vs. 8.2%; P = .04).
The proton group also had numerically lower incidences of MI (2.3% vs. 9.0%; P = .06) and stroke (3.2% vs. 6.1%; P = .50).
The proton and photon groups were similar as far as the incidence of total cardiovascular events (53.1% vs. 47.1%; P = .48) and the 3-year overall survival rate (38.8% vs. 42.1%; P = .99).
“Our future studies aim to examine the potential relationships between grade of cardiac event and type of radiotherapy and dose to cardiac substructures,” Dr. Kegelman commented.
In addition, his institution is participating in RTOG 1308, a phase 3 trial comparing photon and proton beam radiotherapy in patients with inoperable lung cancer that will better assess cardiac-related morbidity and mortality. The trial is expected to be completed by the end of 2025.
Accumulating evidence
“This study adds to a growing body of evidence about the potential importance of heart dose in any radiation modality,” said Daniel Gomez, MD, MBA, of Memorial Sloan Kettering Cancer Center in New York, who was not involved in the study.
The RTOG 0617 trial and the Lung ART trial previously showed correlations between lower radiation dose to the heart and better survival in patients with lung cancer, Dr. Gomez noted.
“It’s been well established that protons can improve heart dose, and therefore it’s been inferred that they may improve outcomes, but the exact mechanisms remain unclear,” Dr. Gomez said.
Proton beam radiotherapy performed well in a single-arm, phase 2 trial among patients with unresectable NSCLC.
“The ongoing phase 3 trial is using a more modern proton technique and has a larger population, with a randomized study design. It will be much more informative,” Dr. Gomez predicted.
The current study did not receive specific funding. Dr. Kegelman disclosed no relevant conflicts of interest. Dr. Gomez disclosed honoraria from Varian.
SOURCE: Kegelman TP et al. ASTRO 2020, Abstract 1046.
FROM ASTRO 2020














