User login
Balancing the Challenge of Research with the Joys of Clinical Care
Andrew Ofosu, MD, MPH, loves the variety that GI medicine offers on a day-to-day basis.
Some days are spent in the endoscopy suite, performing endoscopic retrograde cholangiopancreatography in patients with cholangitis, “which is usually a high-stakes situation,” he said. Other days he might be in clinic, helping to manage a patient with chronic pancreatitis.
“The contrast of the immediate impact of a procedure combined with the continuity of long-term relationships, is special to me,” said Dr. Ofosu, an associate professor of medicine at Cincinnati College of Medicine, in Cincinnati, Ohio. He’s also a member of AGA’s Future Leaders program, which provides early career GI physicians with opportunities to network and develop leadership skills.
In an interview, he discussed his research pursuits in the areas of pancreatic cancer and artificial intelligence (AI), and his unique methods for connecting with patients. The art of listening to patient concerns is crucial, he says, especially following a difficult diagnosis.
What’s it like to be part of the AGA Future Leaders Class of 2025-2026? How has the experience enriched your career?
Dr. Ofosu: My time being part of this group has been very transformative. It’s provided mentorship from national leaders. It’s enabled me to collaborate with peers across different institutions and given me opportunities to refine my leadership skills. It’s changed my perspective and created a network that has equipped me to contribute meaningfully to the gastroenterology community and to my institution.
What is the most challenging clinical case you’ve encountered?
Dr. Ofosu: One case that stands out was a young patient with recurrent idiopathic pancreatitis. We went through all the potential differential etiologies that includes genetics, autoimmune disease and structural etiologies. It became a long, diagnostic journey. The challenge wasn’t just the medical aspect of it, but the emotional aspect of it…when you don’t have all the answers available. We were eventually able to figure out what the cause of the pancreatitis was. It was genetic, and the patient is doing great now.
One of your research interests has been developing innovative ways to use AI in endoscopic ultrasound to identify and characterize lesions. Can you discuss some of those innovations?
Dr. Ofosu: It’s definitely an area that I’m looking to explore at this time; to leverage AI to improve diagnostic capability of endoscopic ultrasound. The whole idea is to be able to use AI to analyze images in real time that can help highlight features, which can ultimately help in distinguishing both benign and malignant tumors, and allowing AI to provide real time diagnostic support, improving accuracy of diagnosis and reducing unnecessary treatment.
In 2021, you conducted a study to investigate the demographics, clinical outcomes and survival outcomes of patients diagnosed with early and late onset pancreatic adenocarcinoma. What did your study reveal and what are the next steps?
Dr. Ofosu: Our study looked at over 136,000 patients with pancreatic adenocarcinoma and compared those diagnosed under age 40 to older patients. We found that although pancreatic cancer is rare in the young, both groups are presenting more often with advanced disease, and incidence is rising. Younger patients tend to have tumors in the head of the pancreas, while older patients more often show growth in the body and tail. Survival overall remains very poor—about 6 to 7 months—but slightly better in younger patients.
I think the next step is to better understand the biological drivers of early onset PAC to look at integrating molecular profiling to see if there are distinct genetic patterns that can guide therapy. Ultimately the goal is to improve early detection and tailor management strategy for this subset of patients.
What is your approach to patient communication and education?
Dr. Ofosu: I aim for clarity and empathy. Some GI diagnoses can be intimidating, with all the terminologies, and so I use a lot of analogies and visuals to simplify complex conditions. I also ensure that patients understand what we are discussing because I found that what a patient hears isn’t always what they think I explained.
I believe being honest and compassionate should go hand in hand. I don’t shy away from delivering difficult news, but I always take time to pause, listen, and acknowledge emotions. I found that patients and families appreciate transparency even when the prognosis is tough, as long as they know I’m fully present with them.
Can you share a memorable patient interaction that impacted you?
Dr. Ofosu: There was one patient with chronic pancreatitis due to alcohol who had limited economic and social support. Beyond the medical management, what made a difference was sitting and listening to the patient, helping them connect to resources and social support – a social network. I think this reinforces that medicine isn’t just about lab values. It’s all about restoring dignity and focus with the patient.
What do you think is the biggest misconception about your specialty?
Dr. Ofosu: That gastroenterology is all about procedures, that all we do is scope. In reality, it’s a combination of technical expertise as well as the cognitive aspect of providing long-term management of complex diseases that affect patients, which takes a diverse skillset beyond endoscopy.
Lightning Round
What’s your favorite season of the year?
Fall. I like the colors of changing leaves
What’s your favorite way to spend a weekend?
Watching soccer with family and friends
If you could have dinner with any historical figure, who would it be?
Nelson Mandela
What’s your go-to karaoke song?Don’t Stop Believin’ by Journey
What’s one thing on your bucket list?
Travel to Europe, experience different cultures
What’s your favorite childhood memory?
When I learned how to fly a kite
If you could instantly learn any skill, what would it be?
Playing piano
Are you a planner or more spontaneous?
Planner
What’s your favorite holiday tradition?
Sharing Christmas dinner with family.
Andrew Ofosu, MD, MPH, loves the variety that GI medicine offers on a day-to-day basis.
Some days are spent in the endoscopy suite, performing endoscopic retrograde cholangiopancreatography in patients with cholangitis, “which is usually a high-stakes situation,” he said. Other days he might be in clinic, helping to manage a patient with chronic pancreatitis.
“The contrast of the immediate impact of a procedure combined with the continuity of long-term relationships, is special to me,” said Dr. Ofosu, an associate professor of medicine at Cincinnati College of Medicine, in Cincinnati, Ohio. He’s also a member of AGA’s Future Leaders program, which provides early career GI physicians with opportunities to network and develop leadership skills.
In an interview, he discussed his research pursuits in the areas of pancreatic cancer and artificial intelligence (AI), and his unique methods for connecting with patients. The art of listening to patient concerns is crucial, he says, especially following a difficult diagnosis.
What’s it like to be part of the AGA Future Leaders Class of 2025-2026? How has the experience enriched your career?
Dr. Ofosu: My time being part of this group has been very transformative. It’s provided mentorship from national leaders. It’s enabled me to collaborate with peers across different institutions and given me opportunities to refine my leadership skills. It’s changed my perspective and created a network that has equipped me to contribute meaningfully to the gastroenterology community and to my institution.
What is the most challenging clinical case you’ve encountered?
Dr. Ofosu: One case that stands out was a young patient with recurrent idiopathic pancreatitis. We went through all the potential differential etiologies that includes genetics, autoimmune disease and structural etiologies. It became a long, diagnostic journey. The challenge wasn’t just the medical aspect of it, but the emotional aspect of it…when you don’t have all the answers available. We were eventually able to figure out what the cause of the pancreatitis was. It was genetic, and the patient is doing great now.
One of your research interests has been developing innovative ways to use AI in endoscopic ultrasound to identify and characterize lesions. Can you discuss some of those innovations?
Dr. Ofosu: It’s definitely an area that I’m looking to explore at this time; to leverage AI to improve diagnostic capability of endoscopic ultrasound. The whole idea is to be able to use AI to analyze images in real time that can help highlight features, which can ultimately help in distinguishing both benign and malignant tumors, and allowing AI to provide real time diagnostic support, improving accuracy of diagnosis and reducing unnecessary treatment.
In 2021, you conducted a study to investigate the demographics, clinical outcomes and survival outcomes of patients diagnosed with early and late onset pancreatic adenocarcinoma. What did your study reveal and what are the next steps?
Dr. Ofosu: Our study looked at over 136,000 patients with pancreatic adenocarcinoma and compared those diagnosed under age 40 to older patients. We found that although pancreatic cancer is rare in the young, both groups are presenting more often with advanced disease, and incidence is rising. Younger patients tend to have tumors in the head of the pancreas, while older patients more often show growth in the body and tail. Survival overall remains very poor—about 6 to 7 months—but slightly better in younger patients.
I think the next step is to better understand the biological drivers of early onset PAC to look at integrating molecular profiling to see if there are distinct genetic patterns that can guide therapy. Ultimately the goal is to improve early detection and tailor management strategy for this subset of patients.
What is your approach to patient communication and education?
Dr. Ofosu: I aim for clarity and empathy. Some GI diagnoses can be intimidating, with all the terminologies, and so I use a lot of analogies and visuals to simplify complex conditions. I also ensure that patients understand what we are discussing because I found that what a patient hears isn’t always what they think I explained.
I believe being honest and compassionate should go hand in hand. I don’t shy away from delivering difficult news, but I always take time to pause, listen, and acknowledge emotions. I found that patients and families appreciate transparency even when the prognosis is tough, as long as they know I’m fully present with them.
Can you share a memorable patient interaction that impacted you?
Dr. Ofosu: There was one patient with chronic pancreatitis due to alcohol who had limited economic and social support. Beyond the medical management, what made a difference was sitting and listening to the patient, helping them connect to resources and social support – a social network. I think this reinforces that medicine isn’t just about lab values. It’s all about restoring dignity and focus with the patient.
What do you think is the biggest misconception about your specialty?
Dr. Ofosu: That gastroenterology is all about procedures, that all we do is scope. In reality, it’s a combination of technical expertise as well as the cognitive aspect of providing long-term management of complex diseases that affect patients, which takes a diverse skillset beyond endoscopy.
Lightning Round
What’s your favorite season of the year?
Fall. I like the colors of changing leaves
What’s your favorite way to spend a weekend?
Watching soccer with family and friends
If you could have dinner with any historical figure, who would it be?
Nelson Mandela
What’s your go-to karaoke song?Don’t Stop Believin’ by Journey
What’s one thing on your bucket list?
Travel to Europe, experience different cultures
What’s your favorite childhood memory?
When I learned how to fly a kite
If you could instantly learn any skill, what would it be?
Playing piano
Are you a planner or more spontaneous?
Planner
What’s your favorite holiday tradition?
Sharing Christmas dinner with family.
Andrew Ofosu, MD, MPH, loves the variety that GI medicine offers on a day-to-day basis.
Some days are spent in the endoscopy suite, performing endoscopic retrograde cholangiopancreatography in patients with cholangitis, “which is usually a high-stakes situation,” he said. Other days he might be in clinic, helping to manage a patient with chronic pancreatitis.
“The contrast of the immediate impact of a procedure combined with the continuity of long-term relationships, is special to me,” said Dr. Ofosu, an associate professor of medicine at Cincinnati College of Medicine, in Cincinnati, Ohio. He’s also a member of AGA’s Future Leaders program, which provides early career GI physicians with opportunities to network and develop leadership skills.
In an interview, he discussed his research pursuits in the areas of pancreatic cancer and artificial intelligence (AI), and his unique methods for connecting with patients. The art of listening to patient concerns is crucial, he says, especially following a difficult diagnosis.
What’s it like to be part of the AGA Future Leaders Class of 2025-2026? How has the experience enriched your career?
Dr. Ofosu: My time being part of this group has been very transformative. It’s provided mentorship from national leaders. It’s enabled me to collaborate with peers across different institutions and given me opportunities to refine my leadership skills. It’s changed my perspective and created a network that has equipped me to contribute meaningfully to the gastroenterology community and to my institution.
What is the most challenging clinical case you’ve encountered?
Dr. Ofosu: One case that stands out was a young patient with recurrent idiopathic pancreatitis. We went through all the potential differential etiologies that includes genetics, autoimmune disease and structural etiologies. It became a long, diagnostic journey. The challenge wasn’t just the medical aspect of it, but the emotional aspect of it…when you don’t have all the answers available. We were eventually able to figure out what the cause of the pancreatitis was. It was genetic, and the patient is doing great now.
One of your research interests has been developing innovative ways to use AI in endoscopic ultrasound to identify and characterize lesions. Can you discuss some of those innovations?
Dr. Ofosu: It’s definitely an area that I’m looking to explore at this time; to leverage AI to improve diagnostic capability of endoscopic ultrasound. The whole idea is to be able to use AI to analyze images in real time that can help highlight features, which can ultimately help in distinguishing both benign and malignant tumors, and allowing AI to provide real time diagnostic support, improving accuracy of diagnosis and reducing unnecessary treatment.
In 2021, you conducted a study to investigate the demographics, clinical outcomes and survival outcomes of patients diagnosed with early and late onset pancreatic adenocarcinoma. What did your study reveal and what are the next steps?
Dr. Ofosu: Our study looked at over 136,000 patients with pancreatic adenocarcinoma and compared those diagnosed under age 40 to older patients. We found that although pancreatic cancer is rare in the young, both groups are presenting more often with advanced disease, and incidence is rising. Younger patients tend to have tumors in the head of the pancreas, while older patients more often show growth in the body and tail. Survival overall remains very poor—about 6 to 7 months—but slightly better in younger patients.
I think the next step is to better understand the biological drivers of early onset PAC to look at integrating molecular profiling to see if there are distinct genetic patterns that can guide therapy. Ultimately the goal is to improve early detection and tailor management strategy for this subset of patients.
What is your approach to patient communication and education?
Dr. Ofosu: I aim for clarity and empathy. Some GI diagnoses can be intimidating, with all the terminologies, and so I use a lot of analogies and visuals to simplify complex conditions. I also ensure that patients understand what we are discussing because I found that what a patient hears isn’t always what they think I explained.
I believe being honest and compassionate should go hand in hand. I don’t shy away from delivering difficult news, but I always take time to pause, listen, and acknowledge emotions. I found that patients and families appreciate transparency even when the prognosis is tough, as long as they know I’m fully present with them.
Can you share a memorable patient interaction that impacted you?
Dr. Ofosu: There was one patient with chronic pancreatitis due to alcohol who had limited economic and social support. Beyond the medical management, what made a difference was sitting and listening to the patient, helping them connect to resources and social support – a social network. I think this reinforces that medicine isn’t just about lab values. It’s all about restoring dignity and focus with the patient.
What do you think is the biggest misconception about your specialty?
Dr. Ofosu: That gastroenterology is all about procedures, that all we do is scope. In reality, it’s a combination of technical expertise as well as the cognitive aspect of providing long-term management of complex diseases that affect patients, which takes a diverse skillset beyond endoscopy.
Lightning Round
What’s your favorite season of the year?
Fall. I like the colors of changing leaves
What’s your favorite way to spend a weekend?
Watching soccer with family and friends
If you could have dinner with any historical figure, who would it be?
Nelson Mandela
What’s your go-to karaoke song?Don’t Stop Believin’ by Journey
What’s one thing on your bucket list?
Travel to Europe, experience different cultures
What’s your favorite childhood memory?
When I learned how to fly a kite
If you could instantly learn any skill, what would it be?
Playing piano
Are you a planner or more spontaneous?
Planner
What’s your favorite holiday tradition?
Sharing Christmas dinner with family.

On a Quest To Reduce Stigmas about Anal Cancer
“I think gastroenterologists are uniquely positioned to help with diagnosing anal diseases, in particular anal cancer,” she said. “It is part of the digestive tract, and my mission is to help gastroenterologists remember that.”
Dr. Korman is a gastroenterologist with Capital Digestive Care in Washington D.C., where she serves as chair of its Women’s Committee and as a member of the board of managers. She’s also the medical director of the Endoscopy Center of Washington D.C.
A recipient of the 2025 AGA Distinguished Clinician Award in Private Practice, Dr. Korman has dedicated her career to educating clinicians on anal cancer screening and anal human papillomavirus. On the research front, she participated as an investigator in the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial, which led to international anal cancer screening guidelines.
She also co-directs the International Anal Neoplasia Society (IANS) Standard High Resolution Anoscopy course.
When she’s not serving her patients, Dr. Korman speaks in the community about anal cancer awareness and screening. In the last few years, Dr. Korman has presented grand rounds at various institutions and speaks at major medical conferences. “I just try to advocate and help gastroenterologists understand who is at risk, how to look for anal cancer, how to screen, and who to refer. If anyone invites me to speak, I generally will do it,” said Dr. Korman.
In an interview, she talked about the outcomes of the ANCHOR trial and how it may inform future research, and her work to reduce bias and stigma for LGBTQ+ patients.
You decided to become a physician after studying in Egypt and Israel and volunteering with Physicians for Human Rights. Can you talk about that journey?
Dr. Korman: I majored in Religion and Middle East studies, and I minored in Arabic. I thought I was going to become a professor of religious studies. But during my time studying abroad and volunteering for Physicians for Human Rights, I was deeply moved by how physicians connect with the core of our shared humanity. Becoming a physician allows one to meet the most fundamental of human needs—caring for another’s health—in a direct and meaningful way.
My father is a physician, a gastroenterologist, but I never considered it as a career option growing up. The year after I graduated college, I accompanied my parents to my father’s medical school reunion and I thought, ‘Why did I never think about this?’ I decided to go back to school to take the pre-med requirements. Gastroenterology seemed to combine the ability to work with my hands, do procedures, have long-term relationships with patients, and think about complex problems.
GI medicine often involves detective work. What is the most challenging case you’ve encountered?
Dr. Korman: Sometimes the patients who have very severe disorders of gut-brain interaction can be the most challenging because finding treatments for them or getting them to a place where they accept certain types of treatment can be really difficult. And of course, you have to put your detective hat on and make sure you have ruled out all the “zebras.” It can take years to build the level of trust where patients are willing to accept the diagnosis and then pursue appropriate treatment.
I always try my best, but I don’t like to give up. I will refer a patient to a colleague if they have a problem and I can’t figure out what the diagnosis is or find a treatment that works. I believe in second and third opinions. I recognize that there’s a limit to what my brain can do and that we all have blind spots. Maybe someone will look at the case with fresh eyes and think of something else.
What was the most impactful outcomes of the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial?
Dr. Korman: This was a National Institutes of Health (NIH)-sponsored, randomized controlled trial with 26 clinical sites. We studied people living with human immunodeficiency virus (HIV), as they are the most at-risk group for anal cancer.
We were looking to prove that treating high grade squamous intraepithelial lesions (HSIL) of the anal canal would lead to a significant reduction in the rates of anal cancer. No one in the medical community would accept guidelines or recommendations about what to do with anal pre-cancers until we proved that treatment worked.
We published the findings in 2022. The study concluded when we met our endpoint earlier than expected. We were able to prove that treating high grade anal dysplasia does indeed lead to a very significant reduction in progression to anal cancer. That ultimately led to guidelines. The International Anal Neoplasia Society came out with consensus guidelines on screening for anal cancer in January 2024. In August 2024, NIH, the Centers for Disease Control and Prevention, and the Infectious Diseases Society of America came out with screening guidelines for people living with HIV.
Were there any other outcomes from this research?
Dr. Korman: One of the great things about the study is that we accumulated a bank of tissue and biologic specimens. There were about 4,500 patients randomized into the trial, but about 10,000 patients screened. So, we have a massive collection of biospecimens that we can use to ask questions about the progression of HSIL to anal cancer. We would like to understand more about viral and host molecular mechanisms and hopefully find biomarkers that will identify individuals at particularly high risk of progression. It’s a more precision medicine type of approach.
Education has been a cornerstone of your career. What’s the most rewarding part of teaching the IANS standard high resolution endoscopy course?
Dr. Korman: I first took the course in 2010, and that’s when I started my journey of learning how to perform high resolution endoscopy. Last year I was asked to help co-direct the course. It is now virtual and asynchronous where everything is recorded. But it was exciting to help reorganize the course, update the lectures, and make sure that everything is current. We get to answer questions from participants from all over the world. I think there are participants from 23 countries who have taken the course, which is amazing.
Could you share your work with the LGBTQIA+ population? What specific needs/challenges does this population have with GI care?
Dr. Korman: Many people in the sexual and gender minority community have experienced discrimination in health care settings or know of someone who has. For these reasons, LGBTQIA+ people may approach health care with the expectation of a negative encounter, or they may avoid accessing care altogether. Because anal cancer disproportionately affects sexual and gender minority communities, creating a warm, inclusive environment is key to identifying who is at risk, building trust, and ensuring patients receive the care they need. When you’re talking about anal cancer, there’s a lot of stigma and shame. I think people are afraid to seek care.
Gastroenterology has traditionally been an “old boys club” but that is changing. We’re trying to work on educating people on how to recognize their own biases and move beyond them to provide care that’s affirming and where people feel that they have a safe space to talk about their concerns. Men who have sex with men, in particular living with HIV, are at the highest risk of developing anal cancer. If you don’t know that your patient is a man who has sex with men, or they don’t want to disclose that they’re living with HIV, you don’t know to screen them, and then you’re missing an opportunity to potentially prevent a cancer.
What advice would you give to aspiring medical students interested in GI?
Dr. Korman: GI is the most exciting and interesting field. We take care of so many different organs, and we’re never bored. If medical students want to get into GI, I recommend that they try to be in an office or an endoscopy center and see if it’s really for them and get some hands-on experience if possible. To be truly great at this profession, you really must see it as a calling – jump in with your whole heart and not see it as just a job. If you can do that, you’ll succeed.
How do you handle stress and maintain work-life balance?
Dr. Korman: Exercise. I try to work out at least five days a week. I can’t live without it. That keeps me going. What do I do for fun? I spend time with my family and my friends. I enjoy going to new restaurants and being outdoors, especially near a body of water. I travel, and I love watching movies. I am also guilty of binge-watching TV on a regular basis as well.
Lightning Round
Coffee or tea?
Coffee, 100%
What’s your favorite book?
I can’t say I have just one, but I recently read Tomorrow and Tomorrow and Tomorrow and loved it
Beach vacation or mountain retreat?
Beach
Early bird or night owl?
Early bird
What’s your go-to comfort food?
Anything with bananas
If you could travel anywhere, where would you go?
Vietnam or African safari
What’s your favorite childhood memory?
Swim team when I was a kid
If you could instantly learn any skill, what would it be?
Playing the drums
Are you a planner or more spontaneous?
Planner, although it’s not my strong suit, if I’m being honest.
“I think gastroenterologists are uniquely positioned to help with diagnosing anal diseases, in particular anal cancer,” she said. “It is part of the digestive tract, and my mission is to help gastroenterologists remember that.”
Dr. Korman is a gastroenterologist with Capital Digestive Care in Washington D.C., where she serves as chair of its Women’s Committee and as a member of the board of managers. She’s also the medical director of the Endoscopy Center of Washington D.C.
A recipient of the 2025 AGA Distinguished Clinician Award in Private Practice, Dr. Korman has dedicated her career to educating clinicians on anal cancer screening and anal human papillomavirus. On the research front, she participated as an investigator in the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial, which led to international anal cancer screening guidelines.
She also co-directs the International Anal Neoplasia Society (IANS) Standard High Resolution Anoscopy course.
When she’s not serving her patients, Dr. Korman speaks in the community about anal cancer awareness and screening. In the last few years, Dr. Korman has presented grand rounds at various institutions and speaks at major medical conferences. “I just try to advocate and help gastroenterologists understand who is at risk, how to look for anal cancer, how to screen, and who to refer. If anyone invites me to speak, I generally will do it,” said Dr. Korman.
In an interview, she talked about the outcomes of the ANCHOR trial and how it may inform future research, and her work to reduce bias and stigma for LGBTQ+ patients.
You decided to become a physician after studying in Egypt and Israel and volunteering with Physicians for Human Rights. Can you talk about that journey?
Dr. Korman: I majored in Religion and Middle East studies, and I minored in Arabic. I thought I was going to become a professor of religious studies. But during my time studying abroad and volunteering for Physicians for Human Rights, I was deeply moved by how physicians connect with the core of our shared humanity. Becoming a physician allows one to meet the most fundamental of human needs—caring for another’s health—in a direct and meaningful way.
My father is a physician, a gastroenterologist, but I never considered it as a career option growing up. The year after I graduated college, I accompanied my parents to my father’s medical school reunion and I thought, ‘Why did I never think about this?’ I decided to go back to school to take the pre-med requirements. Gastroenterology seemed to combine the ability to work with my hands, do procedures, have long-term relationships with patients, and think about complex problems.
GI medicine often involves detective work. What is the most challenging case you’ve encountered?
Dr. Korman: Sometimes the patients who have very severe disorders of gut-brain interaction can be the most challenging because finding treatments for them or getting them to a place where they accept certain types of treatment can be really difficult. And of course, you have to put your detective hat on and make sure you have ruled out all the “zebras.” It can take years to build the level of trust where patients are willing to accept the diagnosis and then pursue appropriate treatment.
I always try my best, but I don’t like to give up. I will refer a patient to a colleague if they have a problem and I can’t figure out what the diagnosis is or find a treatment that works. I believe in second and third opinions. I recognize that there’s a limit to what my brain can do and that we all have blind spots. Maybe someone will look at the case with fresh eyes and think of something else.
What was the most impactful outcomes of the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial?
Dr. Korman: This was a National Institutes of Health (NIH)-sponsored, randomized controlled trial with 26 clinical sites. We studied people living with human immunodeficiency virus (HIV), as they are the most at-risk group for anal cancer.
We were looking to prove that treating high grade squamous intraepithelial lesions (HSIL) of the anal canal would lead to a significant reduction in the rates of anal cancer. No one in the medical community would accept guidelines or recommendations about what to do with anal pre-cancers until we proved that treatment worked.
We published the findings in 2022. The study concluded when we met our endpoint earlier than expected. We were able to prove that treating high grade anal dysplasia does indeed lead to a very significant reduction in progression to anal cancer. That ultimately led to guidelines. The International Anal Neoplasia Society came out with consensus guidelines on screening for anal cancer in January 2024. In August 2024, NIH, the Centers for Disease Control and Prevention, and the Infectious Diseases Society of America came out with screening guidelines for people living with HIV.
Were there any other outcomes from this research?
Dr. Korman: One of the great things about the study is that we accumulated a bank of tissue and biologic specimens. There were about 4,500 patients randomized into the trial, but about 10,000 patients screened. So, we have a massive collection of biospecimens that we can use to ask questions about the progression of HSIL to anal cancer. We would like to understand more about viral and host molecular mechanisms and hopefully find biomarkers that will identify individuals at particularly high risk of progression. It’s a more precision medicine type of approach.
Education has been a cornerstone of your career. What’s the most rewarding part of teaching the IANS standard high resolution endoscopy course?
Dr. Korman: I first took the course in 2010, and that’s when I started my journey of learning how to perform high resolution endoscopy. Last year I was asked to help co-direct the course. It is now virtual and asynchronous where everything is recorded. But it was exciting to help reorganize the course, update the lectures, and make sure that everything is current. We get to answer questions from participants from all over the world. I think there are participants from 23 countries who have taken the course, which is amazing.
Could you share your work with the LGBTQIA+ population? What specific needs/challenges does this population have with GI care?
Dr. Korman: Many people in the sexual and gender minority community have experienced discrimination in health care settings or know of someone who has. For these reasons, LGBTQIA+ people may approach health care with the expectation of a negative encounter, or they may avoid accessing care altogether. Because anal cancer disproportionately affects sexual and gender minority communities, creating a warm, inclusive environment is key to identifying who is at risk, building trust, and ensuring patients receive the care they need. When you’re talking about anal cancer, there’s a lot of stigma and shame. I think people are afraid to seek care.
Gastroenterology has traditionally been an “old boys club” but that is changing. We’re trying to work on educating people on how to recognize their own biases and move beyond them to provide care that’s affirming and where people feel that they have a safe space to talk about their concerns. Men who have sex with men, in particular living with HIV, are at the highest risk of developing anal cancer. If you don’t know that your patient is a man who has sex with men, or they don’t want to disclose that they’re living with HIV, you don’t know to screen them, and then you’re missing an opportunity to potentially prevent a cancer.
What advice would you give to aspiring medical students interested in GI?
Dr. Korman: GI is the most exciting and interesting field. We take care of so many different organs, and we’re never bored. If medical students want to get into GI, I recommend that they try to be in an office or an endoscopy center and see if it’s really for them and get some hands-on experience if possible. To be truly great at this profession, you really must see it as a calling – jump in with your whole heart and not see it as just a job. If you can do that, you’ll succeed.
How do you handle stress and maintain work-life balance?
Dr. Korman: Exercise. I try to work out at least five days a week. I can’t live without it. That keeps me going. What do I do for fun? I spend time with my family and my friends. I enjoy going to new restaurants and being outdoors, especially near a body of water. I travel, and I love watching movies. I am also guilty of binge-watching TV on a regular basis as well.
Lightning Round
Coffee or tea?
Coffee, 100%
What’s your favorite book?
I can’t say I have just one, but I recently read Tomorrow and Tomorrow and Tomorrow and loved it
Beach vacation or mountain retreat?
Beach
Early bird or night owl?
Early bird
What’s your go-to comfort food?
Anything with bananas
If you could travel anywhere, where would you go?
Vietnam or African safari
What’s your favorite childhood memory?
Swim team when I was a kid
If you could instantly learn any skill, what would it be?
Playing the drums
Are you a planner or more spontaneous?
Planner, although it’s not my strong suit, if I’m being honest.
“I think gastroenterologists are uniquely positioned to help with diagnosing anal diseases, in particular anal cancer,” she said. “It is part of the digestive tract, and my mission is to help gastroenterologists remember that.”
Dr. Korman is a gastroenterologist with Capital Digestive Care in Washington D.C., where she serves as chair of its Women’s Committee and as a member of the board of managers. She’s also the medical director of the Endoscopy Center of Washington D.C.
A recipient of the 2025 AGA Distinguished Clinician Award in Private Practice, Dr. Korman has dedicated her career to educating clinicians on anal cancer screening and anal human papillomavirus. On the research front, she participated as an investigator in the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial, which led to international anal cancer screening guidelines.
She also co-directs the International Anal Neoplasia Society (IANS) Standard High Resolution Anoscopy course.
When she’s not serving her patients, Dr. Korman speaks in the community about anal cancer awareness and screening. In the last few years, Dr. Korman has presented grand rounds at various institutions and speaks at major medical conferences. “I just try to advocate and help gastroenterologists understand who is at risk, how to look for anal cancer, how to screen, and who to refer. If anyone invites me to speak, I generally will do it,” said Dr. Korman.
In an interview, she talked about the outcomes of the ANCHOR trial and how it may inform future research, and her work to reduce bias and stigma for LGBTQ+ patients.
You decided to become a physician after studying in Egypt and Israel and volunteering with Physicians for Human Rights. Can you talk about that journey?
Dr. Korman: I majored in Religion and Middle East studies, and I minored in Arabic. I thought I was going to become a professor of religious studies. But during my time studying abroad and volunteering for Physicians for Human Rights, I was deeply moved by how physicians connect with the core of our shared humanity. Becoming a physician allows one to meet the most fundamental of human needs—caring for another’s health—in a direct and meaningful way.
My father is a physician, a gastroenterologist, but I never considered it as a career option growing up. The year after I graduated college, I accompanied my parents to my father’s medical school reunion and I thought, ‘Why did I never think about this?’ I decided to go back to school to take the pre-med requirements. Gastroenterology seemed to combine the ability to work with my hands, do procedures, have long-term relationships with patients, and think about complex problems.
GI medicine often involves detective work. What is the most challenging case you’ve encountered?
Dr. Korman: Sometimes the patients who have very severe disorders of gut-brain interaction can be the most challenging because finding treatments for them or getting them to a place where they accept certain types of treatment can be really difficult. And of course, you have to put your detective hat on and make sure you have ruled out all the “zebras.” It can take years to build the level of trust where patients are willing to accept the diagnosis and then pursue appropriate treatment.
I always try my best, but I don’t like to give up. I will refer a patient to a colleague if they have a problem and I can’t figure out what the diagnosis is or find a treatment that works. I believe in second and third opinions. I recognize that there’s a limit to what my brain can do and that we all have blind spots. Maybe someone will look at the case with fresh eyes and think of something else.
What was the most impactful outcomes of the ANAL Cancer-HSIL Outcomes Research (ANCHOR) trial?
Dr. Korman: This was a National Institutes of Health (NIH)-sponsored, randomized controlled trial with 26 clinical sites. We studied people living with human immunodeficiency virus (HIV), as they are the most at-risk group for anal cancer.
We were looking to prove that treating high grade squamous intraepithelial lesions (HSIL) of the anal canal would lead to a significant reduction in the rates of anal cancer. No one in the medical community would accept guidelines or recommendations about what to do with anal pre-cancers until we proved that treatment worked.
We published the findings in 2022. The study concluded when we met our endpoint earlier than expected. We were able to prove that treating high grade anal dysplasia does indeed lead to a very significant reduction in progression to anal cancer. That ultimately led to guidelines. The International Anal Neoplasia Society came out with consensus guidelines on screening for anal cancer in January 2024. In August 2024, NIH, the Centers for Disease Control and Prevention, and the Infectious Diseases Society of America came out with screening guidelines for people living with HIV.
Were there any other outcomes from this research?
Dr. Korman: One of the great things about the study is that we accumulated a bank of tissue and biologic specimens. There were about 4,500 patients randomized into the trial, but about 10,000 patients screened. So, we have a massive collection of biospecimens that we can use to ask questions about the progression of HSIL to anal cancer. We would like to understand more about viral and host molecular mechanisms and hopefully find biomarkers that will identify individuals at particularly high risk of progression. It’s a more precision medicine type of approach.
Education has been a cornerstone of your career. What’s the most rewarding part of teaching the IANS standard high resolution endoscopy course?
Dr. Korman: I first took the course in 2010, and that’s when I started my journey of learning how to perform high resolution endoscopy. Last year I was asked to help co-direct the course. It is now virtual and asynchronous where everything is recorded. But it was exciting to help reorganize the course, update the lectures, and make sure that everything is current. We get to answer questions from participants from all over the world. I think there are participants from 23 countries who have taken the course, which is amazing.
Could you share your work with the LGBTQIA+ population? What specific needs/challenges does this population have with GI care?
Dr. Korman: Many people in the sexual and gender minority community have experienced discrimination in health care settings or know of someone who has. For these reasons, LGBTQIA+ people may approach health care with the expectation of a negative encounter, or they may avoid accessing care altogether. Because anal cancer disproportionately affects sexual and gender minority communities, creating a warm, inclusive environment is key to identifying who is at risk, building trust, and ensuring patients receive the care they need. When you’re talking about anal cancer, there’s a lot of stigma and shame. I think people are afraid to seek care.
Gastroenterology has traditionally been an “old boys club” but that is changing. We’re trying to work on educating people on how to recognize their own biases and move beyond them to provide care that’s affirming and where people feel that they have a safe space to talk about their concerns. Men who have sex with men, in particular living with HIV, are at the highest risk of developing anal cancer. If you don’t know that your patient is a man who has sex with men, or they don’t want to disclose that they’re living with HIV, you don’t know to screen them, and then you’re missing an opportunity to potentially prevent a cancer.
What advice would you give to aspiring medical students interested in GI?
Dr. Korman: GI is the most exciting and interesting field. We take care of so many different organs, and we’re never bored. If medical students want to get into GI, I recommend that they try to be in an office or an endoscopy center and see if it’s really for them and get some hands-on experience if possible. To be truly great at this profession, you really must see it as a calling – jump in with your whole heart and not see it as just a job. If you can do that, you’ll succeed.
How do you handle stress and maintain work-life balance?
Dr. Korman: Exercise. I try to work out at least five days a week. I can’t live without it. That keeps me going. What do I do for fun? I spend time with my family and my friends. I enjoy going to new restaurants and being outdoors, especially near a body of water. I travel, and I love watching movies. I am also guilty of binge-watching TV on a regular basis as well.
Lightning Round
Coffee or tea?
Coffee, 100%
What’s your favorite book?
I can’t say I have just one, but I recently read Tomorrow and Tomorrow and Tomorrow and loved it
Beach vacation or mountain retreat?
Beach
Early bird or night owl?
Early bird
What’s your go-to comfort food?
Anything with bananas
If you could travel anywhere, where would you go?
Vietnam or African safari
What’s your favorite childhood memory?
Swim team when I was a kid
If you could instantly learn any skill, what would it be?
Playing the drums
Are you a planner or more spontaneous?
Planner, although it’s not my strong suit, if I’m being honest.

AGA President Brings Forth “Message Of Inclusivity”
“I was always interested in medicine. From a relatively early age I thought that’s what I would be doing,” said Dr. Kim. When his father became disillusioned with his own career as a pathologist, he encouraged his son to look in other directions.
“In college I had the opportunity to study and learn broadly and I became interested in public policy and eventually majored in that discipline,” he said.
The mentorship of the late Uwe Reinhardt, a well-respected health economist at Princeton University, had a major impact on Dr. Kim during his senior year of college. Reinhardt told him that physicians are afforded a special position in society. “They have a moral responsibility to take the lead in terms of guiding and shaping healthcare. His message made a big impression upon me,” said Dr. Kim.
Ultimately, he decided to go into clinical medicine, but maintained his interest in healthcare policy. Experiences outside of the standard approach to medicine “helped me stay in the big picture of healthcare, to make a difference beyond just my individual patients. And that’s played a big part in keeping me involved in organized medicine,” said Dr. Kim, who began his term as AGA president in May 2025.
Dr. Kim is also a partner at South Denver Gastroenterology, a 33-provider, independent gastroenterology practice in Colorado. As the first physician in Colorado with fellowship training in endoscopic ultrasound, he introduced this service line into South Denver’s advanced endoscopy practice.
Dr. Kim has served in numerous roles with AGA, among them the co-director of the AGA Clinical Congress, the Partners in Quality program, and the Nurse Practitioner and Physician Assistant Course. He is a Digestive Disease Week® abstract reviewer, has served as AGA representative to the Accreditation Association for Ambulatory Health Care and to the Alliance of Specialty Medicine. He has also served on the AGA Governing Board as clinical private practice councilor and secretary treasurer.
He discussed the high points of his career in an interview, revealing his plans as AGA president for unifying the sectors of GI medicine and fostering GI innovation and technology.
As the new AGA president, what are your goals for the society?
Dr. Kim: I want to put out a message of inclusivity. I think what’s special about AGA is that we’re the society for all gastroenterologists. Among all the other GI organizations, I think we really have the biggest tent and we work to unite clinicians, educators, and researchers – all gastroenterologists, regardless of their individual practice situation. These days, there is a tendency toward tribalism. People are starting to gravitate toward limiting their interactions to others that are from the same backgrounds. But as gastroenterologists we have more that unites us than divides us. It’s only by working together that we can make things better for everyone.
I think the second point is that we’re on the cusp of some important transformations in gastroenterology. The screening colonoscopy model that has sustained our specialty for decades is rapidly evolving. In addition, there is an increasing ability for patients as consumers to direct their own care through advances in technology, such as virtual health platforms. We’re seeing this as patients increasingly adopt things like complementary and alternative medicine outside of the standard model of physician-directed healthcare. These are two important trends that gastroenterologists need to be aware of and learn how to manage and to adapt to. I think AGA’s role is to help guide that evolution and to give physicians the tools to be able to respond.
We want to focus on innovation and we want to focus on practical solutions.
In terms of fostering innovation in gastroenterology, we’re the first medical professional society to create an incubator for new technologies. Not only do we provide that resource to our members, but we’re also putting our money where our mouth is. Through venture capital initiatives such as our GI Opportunity Fund, we directly invest in companies that we’re helping to develop.
On the practice side, we have been engaging directly with payers to foster improved communication and address pain points on both sides. I think we’re the only medical society that’s taking this type of approach and moving away from the traditional adversarial approach to dealing with payers. Recently, we had a very productive discussion with UnitedHealthcare around some of their upcoming formulary changes for inflammatory bowel disease. We used that opportunity to highlight how nonmedical switching between existing therapies can adversely impact patients, as well as increasing burden of red tape for practices.
Your practice was one of the original groups that formed the Digestive Health Physicians Association (DPHA). What accomplishments of the association are you most proud of?
Dr. Kim: DHPA formed about 10 years ago as an advocacy organization to combat a specific perceived threat, which was the in-office ancillary exception. This is the legislative pathway that allows gastroenterologists to provide ancillary services within their practice. An example of this is pathology for endoscopic procedures, which is an incredible value to patients and improves quality of care. This was under a significant legislative threat at that time. As independent physicians, DHPA took the lead in advocating against eliminating that exception.
I think the larger accomplishment was it demonstrated that gastroenterologists, specifically independent community practice gastroenterologists, could come together successfully and advocate for issues that were of importance to our specialty. AGA and DHPA have worked very well together, collaborating on shared policy interests and have worked closely on both legislative as well as regulatory issues. We’ve sponsored joint meetings that we’ve programmed together and we’re looking forward to continuing a robust partnership.
You have introduced several new clinical practice and practice management models. Can you discuss the part-time partnership model and what it has achieved?
Dr. Kim: Like many practices, South Denver Gastroenterology historically required physician partners to work full time. This conflicted with our desire and our need to attract more women gastroenterologists into our practice. The process involved careful analysis of our direct and indirect expenses, but more importantly it required a negotiation and a meeting of the minds among our partners. A lot of this ultimately came down to trust. It helped a great deal that our practice has always had strong cohesiveness. That helped us to build that trust that partners would stay engaged in the practice even if they worked part time.
Our practice has also always prioritized work-life balance. We were able to come up with a formula that allows partners to work three days per week, retaining their partnership interest and their participation in practice decisions. They stay involved but are also financially sustainable for the practice. It’s been very successful. It’s been a big draw, not just for women, but it has allowed us to create a situation where women are fully one third of our partnership. It’s something we’re all extremely proud of.
How did you get involved in AGA?
Dr. Kim: One of the first projects I participated in was the Roadmap to the Future of GI Practice. This was an initiative to help prepare GI practices for value-based care. We did things like develop quality measure sets for GI conditions such as inflammatory bowel disease and hepatitis C. We published a bundled payment model for screening colonoscopies. We also created a model for obesity management by gastroenterologists. This was 15 years ago, and I think it was about 15 years ahead of its time! It’s interesting to see how many of these changes in GI practice that we envisioned are slowly coming to pass.
I saw that AGA was interested in me as a community-based clinician. They focused on trying to develop those practical tools to help me succeed. It’s one of the reasons I’ve stayed engaged.
What is your approach to patient communication and education?
Dr. Kim: There are two things that I always tell both my staff as well as young people who come to me asking for advice. I think the first and most important is that you should always strive to treat your patients the way that you would want your family treated. Of course, we’re not perfect, but when that doesn’t happen, look at your behavior, the way that you’re interacting, but also the way the system is treating your patients and try to improve things within your own practice. And then the other thing that I tell folks is try to spend more time listening to your patients than talking or speaking at them.
What do you think is the biggest misconception about GI?
Dr. Kim: We’re not just about colonoscopies! I went into GI not just because I enjoy performing procedures, but because our specialty covers such a broad spectrum of physiology and diseases. We also have the ability as gastroenterologists to develop long-term relationships with our patients. I’ve been in practice now more than 25 years, and the greatest satisfaction in my career doesn’t come from the endoscopy center, although I still enjoy performing procedures. It comes from the clinic; it comes from the patients whom I’ve known for decades, and the interaction and conversations that I can have with them, the ability to see their families, their parents, and now in some cases their kids or even their grandkids. It’s incredibly satisfying. It makes my job fun.
What advice would you give to aspiring medical students?
Dr. Kim: One of the things I would say is stay involved in organized medicine. As physicians, we are endowed with great trust. We also have a great responsibility to help shape our healthcare care system. If we work together, we really can make a difference, not just for our profession, but also for society at large and for the patients whom we serve.
I really hope that young people don’t lose their optimism. We hear a lot these days about how much negativity and pessimism there is about the future, especially among young people in our society. But I think it’s a great time to be in medicine. Advances in medical science have made huge strides in our ability to make real differences for our patients. And the pace of technology progress is only going to continue to accelerate. Sure, there are lots of shortcomings in the practice of medicine, but honestly, that’s always been the case. I have faith that as a profession, we are smart people, we’re committed people, and we will be successful in overcoming those challenges. That’s the message that I have for young folks.
Lightning Round
Coffee or tea?
Coffee, black
What’s one hobby you’d like to pick up?
Anything except pickleball
What’s your favorite season of the year?
Winter, I’m a skier
What’s your favorite way to spend a weekend?
Doing anything outside
If you could have dinner with any historical figure, who would it be?
Ben Franklin
What’s your go-to karaoke song?
You don’t want to hear me sing
What’s one thing on your bucket list?
Skiing in South America
What’s the best piece of advice you’ve ever received?
Follow your heart
“I was always interested in medicine. From a relatively early age I thought that’s what I would be doing,” said Dr. Kim. When his father became disillusioned with his own career as a pathologist, he encouraged his son to look in other directions.
“In college I had the opportunity to study and learn broadly and I became interested in public policy and eventually majored in that discipline,” he said.
The mentorship of the late Uwe Reinhardt, a well-respected health economist at Princeton University, had a major impact on Dr. Kim during his senior year of college. Reinhardt told him that physicians are afforded a special position in society. “They have a moral responsibility to take the lead in terms of guiding and shaping healthcare. His message made a big impression upon me,” said Dr. Kim.
Ultimately, he decided to go into clinical medicine, but maintained his interest in healthcare policy. Experiences outside of the standard approach to medicine “helped me stay in the big picture of healthcare, to make a difference beyond just my individual patients. And that’s played a big part in keeping me involved in organized medicine,” said Dr. Kim, who began his term as AGA president in May 2025.
Dr. Kim is also a partner at South Denver Gastroenterology, a 33-provider, independent gastroenterology practice in Colorado. As the first physician in Colorado with fellowship training in endoscopic ultrasound, he introduced this service line into South Denver’s advanced endoscopy practice.
Dr. Kim has served in numerous roles with AGA, among them the co-director of the AGA Clinical Congress, the Partners in Quality program, and the Nurse Practitioner and Physician Assistant Course. He is a Digestive Disease Week® abstract reviewer, has served as AGA representative to the Accreditation Association for Ambulatory Health Care and to the Alliance of Specialty Medicine. He has also served on the AGA Governing Board as clinical private practice councilor and secretary treasurer.
He discussed the high points of his career in an interview, revealing his plans as AGA president for unifying the sectors of GI medicine and fostering GI innovation and technology.
As the new AGA president, what are your goals for the society?
Dr. Kim: I want to put out a message of inclusivity. I think what’s special about AGA is that we’re the society for all gastroenterologists. Among all the other GI organizations, I think we really have the biggest tent and we work to unite clinicians, educators, and researchers – all gastroenterologists, regardless of their individual practice situation. These days, there is a tendency toward tribalism. People are starting to gravitate toward limiting their interactions to others that are from the same backgrounds. But as gastroenterologists we have more that unites us than divides us. It’s only by working together that we can make things better for everyone.
I think the second point is that we’re on the cusp of some important transformations in gastroenterology. The screening colonoscopy model that has sustained our specialty for decades is rapidly evolving. In addition, there is an increasing ability for patients as consumers to direct their own care through advances in technology, such as virtual health platforms. We’re seeing this as patients increasingly adopt things like complementary and alternative medicine outside of the standard model of physician-directed healthcare. These are two important trends that gastroenterologists need to be aware of and learn how to manage and to adapt to. I think AGA’s role is to help guide that evolution and to give physicians the tools to be able to respond.
We want to focus on innovation and we want to focus on practical solutions.
In terms of fostering innovation in gastroenterology, we’re the first medical professional society to create an incubator for new technologies. Not only do we provide that resource to our members, but we’re also putting our money where our mouth is. Through venture capital initiatives such as our GI Opportunity Fund, we directly invest in companies that we’re helping to develop.
On the practice side, we have been engaging directly with payers to foster improved communication and address pain points on both sides. I think we’re the only medical society that’s taking this type of approach and moving away from the traditional adversarial approach to dealing with payers. Recently, we had a very productive discussion with UnitedHealthcare around some of their upcoming formulary changes for inflammatory bowel disease. We used that opportunity to highlight how nonmedical switching between existing therapies can adversely impact patients, as well as increasing burden of red tape for practices.
Your practice was one of the original groups that formed the Digestive Health Physicians Association (DPHA). What accomplishments of the association are you most proud of?
Dr. Kim: DHPA formed about 10 years ago as an advocacy organization to combat a specific perceived threat, which was the in-office ancillary exception. This is the legislative pathway that allows gastroenterologists to provide ancillary services within their practice. An example of this is pathology for endoscopic procedures, which is an incredible value to patients and improves quality of care. This was under a significant legislative threat at that time. As independent physicians, DHPA took the lead in advocating against eliminating that exception.
I think the larger accomplishment was it demonstrated that gastroenterologists, specifically independent community practice gastroenterologists, could come together successfully and advocate for issues that were of importance to our specialty. AGA and DHPA have worked very well together, collaborating on shared policy interests and have worked closely on both legislative as well as regulatory issues. We’ve sponsored joint meetings that we’ve programmed together and we’re looking forward to continuing a robust partnership.
You have introduced several new clinical practice and practice management models. Can you discuss the part-time partnership model and what it has achieved?
Dr. Kim: Like many practices, South Denver Gastroenterology historically required physician partners to work full time. This conflicted with our desire and our need to attract more women gastroenterologists into our practice. The process involved careful analysis of our direct and indirect expenses, but more importantly it required a negotiation and a meeting of the minds among our partners. A lot of this ultimately came down to trust. It helped a great deal that our practice has always had strong cohesiveness. That helped us to build that trust that partners would stay engaged in the practice even if they worked part time.
Our practice has also always prioritized work-life balance. We were able to come up with a formula that allows partners to work three days per week, retaining their partnership interest and their participation in practice decisions. They stay involved but are also financially sustainable for the practice. It’s been very successful. It’s been a big draw, not just for women, but it has allowed us to create a situation where women are fully one third of our partnership. It’s something we’re all extremely proud of.
How did you get involved in AGA?
Dr. Kim: One of the first projects I participated in was the Roadmap to the Future of GI Practice. This was an initiative to help prepare GI practices for value-based care. We did things like develop quality measure sets for GI conditions such as inflammatory bowel disease and hepatitis C. We published a bundled payment model for screening colonoscopies. We also created a model for obesity management by gastroenterologists. This was 15 years ago, and I think it was about 15 years ahead of its time! It’s interesting to see how many of these changes in GI practice that we envisioned are slowly coming to pass.
I saw that AGA was interested in me as a community-based clinician. They focused on trying to develop those practical tools to help me succeed. It’s one of the reasons I’ve stayed engaged.
What is your approach to patient communication and education?
Dr. Kim: There are two things that I always tell both my staff as well as young people who come to me asking for advice. I think the first and most important is that you should always strive to treat your patients the way that you would want your family treated. Of course, we’re not perfect, but when that doesn’t happen, look at your behavior, the way that you’re interacting, but also the way the system is treating your patients and try to improve things within your own practice. And then the other thing that I tell folks is try to spend more time listening to your patients than talking or speaking at them.
What do you think is the biggest misconception about GI?
Dr. Kim: We’re not just about colonoscopies! I went into GI not just because I enjoy performing procedures, but because our specialty covers such a broad spectrum of physiology and diseases. We also have the ability as gastroenterologists to develop long-term relationships with our patients. I’ve been in practice now more than 25 years, and the greatest satisfaction in my career doesn’t come from the endoscopy center, although I still enjoy performing procedures. It comes from the clinic; it comes from the patients whom I’ve known for decades, and the interaction and conversations that I can have with them, the ability to see their families, their parents, and now in some cases their kids or even their grandkids. It’s incredibly satisfying. It makes my job fun.
What advice would you give to aspiring medical students?
Dr. Kim: One of the things I would say is stay involved in organized medicine. As physicians, we are endowed with great trust. We also have a great responsibility to help shape our healthcare care system. If we work together, we really can make a difference, not just for our profession, but also for society at large and for the patients whom we serve.
I really hope that young people don’t lose their optimism. We hear a lot these days about how much negativity and pessimism there is about the future, especially among young people in our society. But I think it’s a great time to be in medicine. Advances in medical science have made huge strides in our ability to make real differences for our patients. And the pace of technology progress is only going to continue to accelerate. Sure, there are lots of shortcomings in the practice of medicine, but honestly, that’s always been the case. I have faith that as a profession, we are smart people, we’re committed people, and we will be successful in overcoming those challenges. That’s the message that I have for young folks.
Lightning Round
Coffee or tea?
Coffee, black
What’s one hobby you’d like to pick up?
Anything except pickleball
What’s your favorite season of the year?
Winter, I’m a skier
What’s your favorite way to spend a weekend?
Doing anything outside
If you could have dinner with any historical figure, who would it be?
Ben Franklin
What’s your go-to karaoke song?
You don’t want to hear me sing
What’s one thing on your bucket list?
Skiing in South America
What’s the best piece of advice you’ve ever received?
Follow your heart
“I was always interested in medicine. From a relatively early age I thought that’s what I would be doing,” said Dr. Kim. When his father became disillusioned with his own career as a pathologist, he encouraged his son to look in other directions.
“In college I had the opportunity to study and learn broadly and I became interested in public policy and eventually majored in that discipline,” he said.
The mentorship of the late Uwe Reinhardt, a well-respected health economist at Princeton University, had a major impact on Dr. Kim during his senior year of college. Reinhardt told him that physicians are afforded a special position in society. “They have a moral responsibility to take the lead in terms of guiding and shaping healthcare. His message made a big impression upon me,” said Dr. Kim.
Ultimately, he decided to go into clinical medicine, but maintained his interest in healthcare policy. Experiences outside of the standard approach to medicine “helped me stay in the big picture of healthcare, to make a difference beyond just my individual patients. And that’s played a big part in keeping me involved in organized medicine,” said Dr. Kim, who began his term as AGA president in May 2025.
Dr. Kim is also a partner at South Denver Gastroenterology, a 33-provider, independent gastroenterology practice in Colorado. As the first physician in Colorado with fellowship training in endoscopic ultrasound, he introduced this service line into South Denver’s advanced endoscopy practice.
Dr. Kim has served in numerous roles with AGA, among them the co-director of the AGA Clinical Congress, the Partners in Quality program, and the Nurse Practitioner and Physician Assistant Course. He is a Digestive Disease Week® abstract reviewer, has served as AGA representative to the Accreditation Association for Ambulatory Health Care and to the Alliance of Specialty Medicine. He has also served on the AGA Governing Board as clinical private practice councilor and secretary treasurer.
He discussed the high points of his career in an interview, revealing his plans as AGA president for unifying the sectors of GI medicine and fostering GI innovation and technology.
As the new AGA president, what are your goals for the society?
Dr. Kim: I want to put out a message of inclusivity. I think what’s special about AGA is that we’re the society for all gastroenterologists. Among all the other GI organizations, I think we really have the biggest tent and we work to unite clinicians, educators, and researchers – all gastroenterologists, regardless of their individual practice situation. These days, there is a tendency toward tribalism. People are starting to gravitate toward limiting their interactions to others that are from the same backgrounds. But as gastroenterologists we have more that unites us than divides us. It’s only by working together that we can make things better for everyone.
I think the second point is that we’re on the cusp of some important transformations in gastroenterology. The screening colonoscopy model that has sustained our specialty for decades is rapidly evolving. In addition, there is an increasing ability for patients as consumers to direct their own care through advances in technology, such as virtual health platforms. We’re seeing this as patients increasingly adopt things like complementary and alternative medicine outside of the standard model of physician-directed healthcare. These are two important trends that gastroenterologists need to be aware of and learn how to manage and to adapt to. I think AGA’s role is to help guide that evolution and to give physicians the tools to be able to respond.
We want to focus on innovation and we want to focus on practical solutions.
In terms of fostering innovation in gastroenterology, we’re the first medical professional society to create an incubator for new technologies. Not only do we provide that resource to our members, but we’re also putting our money where our mouth is. Through venture capital initiatives such as our GI Opportunity Fund, we directly invest in companies that we’re helping to develop.
On the practice side, we have been engaging directly with payers to foster improved communication and address pain points on both sides. I think we’re the only medical society that’s taking this type of approach and moving away from the traditional adversarial approach to dealing with payers. Recently, we had a very productive discussion with UnitedHealthcare around some of their upcoming formulary changes for inflammatory bowel disease. We used that opportunity to highlight how nonmedical switching between existing therapies can adversely impact patients, as well as increasing burden of red tape for practices.
Your practice was one of the original groups that formed the Digestive Health Physicians Association (DPHA). What accomplishments of the association are you most proud of?
Dr. Kim: DHPA formed about 10 years ago as an advocacy organization to combat a specific perceived threat, which was the in-office ancillary exception. This is the legislative pathway that allows gastroenterologists to provide ancillary services within their practice. An example of this is pathology for endoscopic procedures, which is an incredible value to patients and improves quality of care. This was under a significant legislative threat at that time. As independent physicians, DHPA took the lead in advocating against eliminating that exception.
I think the larger accomplishment was it demonstrated that gastroenterologists, specifically independent community practice gastroenterologists, could come together successfully and advocate for issues that were of importance to our specialty. AGA and DHPA have worked very well together, collaborating on shared policy interests and have worked closely on both legislative as well as regulatory issues. We’ve sponsored joint meetings that we’ve programmed together and we’re looking forward to continuing a robust partnership.
You have introduced several new clinical practice and practice management models. Can you discuss the part-time partnership model and what it has achieved?
Dr. Kim: Like many practices, South Denver Gastroenterology historically required physician partners to work full time. This conflicted with our desire and our need to attract more women gastroenterologists into our practice. The process involved careful analysis of our direct and indirect expenses, but more importantly it required a negotiation and a meeting of the minds among our partners. A lot of this ultimately came down to trust. It helped a great deal that our practice has always had strong cohesiveness. That helped us to build that trust that partners would stay engaged in the practice even if they worked part time.
Our practice has also always prioritized work-life balance. We were able to come up with a formula that allows partners to work three days per week, retaining their partnership interest and their participation in practice decisions. They stay involved but are also financially sustainable for the practice. It’s been very successful. It’s been a big draw, not just for women, but it has allowed us to create a situation where women are fully one third of our partnership. It’s something we’re all extremely proud of.
How did you get involved in AGA?
Dr. Kim: One of the first projects I participated in was the Roadmap to the Future of GI Practice. This was an initiative to help prepare GI practices for value-based care. We did things like develop quality measure sets for GI conditions such as inflammatory bowel disease and hepatitis C. We published a bundled payment model for screening colonoscopies. We also created a model for obesity management by gastroenterologists. This was 15 years ago, and I think it was about 15 years ahead of its time! It’s interesting to see how many of these changes in GI practice that we envisioned are slowly coming to pass.
I saw that AGA was interested in me as a community-based clinician. They focused on trying to develop those practical tools to help me succeed. It’s one of the reasons I’ve stayed engaged.
What is your approach to patient communication and education?
Dr. Kim: There are two things that I always tell both my staff as well as young people who come to me asking for advice. I think the first and most important is that you should always strive to treat your patients the way that you would want your family treated. Of course, we’re not perfect, but when that doesn’t happen, look at your behavior, the way that you’re interacting, but also the way the system is treating your patients and try to improve things within your own practice. And then the other thing that I tell folks is try to spend more time listening to your patients than talking or speaking at them.
What do you think is the biggest misconception about GI?
Dr. Kim: We’re not just about colonoscopies! I went into GI not just because I enjoy performing procedures, but because our specialty covers such a broad spectrum of physiology and diseases. We also have the ability as gastroenterologists to develop long-term relationships with our patients. I’ve been in practice now more than 25 years, and the greatest satisfaction in my career doesn’t come from the endoscopy center, although I still enjoy performing procedures. It comes from the clinic; it comes from the patients whom I’ve known for decades, and the interaction and conversations that I can have with them, the ability to see their families, their parents, and now in some cases their kids or even their grandkids. It’s incredibly satisfying. It makes my job fun.
What advice would you give to aspiring medical students?
Dr. Kim: One of the things I would say is stay involved in organized medicine. As physicians, we are endowed with great trust. We also have a great responsibility to help shape our healthcare care system. If we work together, we really can make a difference, not just for our profession, but also for society at large and for the patients whom we serve.
I really hope that young people don’t lose their optimism. We hear a lot these days about how much negativity and pessimism there is about the future, especially among young people in our society. But I think it’s a great time to be in medicine. Advances in medical science have made huge strides in our ability to make real differences for our patients. And the pace of technology progress is only going to continue to accelerate. Sure, there are lots of shortcomings in the practice of medicine, but honestly, that’s always been the case. I have faith that as a profession, we are smart people, we’re committed people, and we will be successful in overcoming those challenges. That’s the message that I have for young folks.
Lightning Round
Coffee or tea?
Coffee, black
What’s one hobby you’d like to pick up?
Anything except pickleball
What’s your favorite season of the year?
Winter, I’m a skier
What’s your favorite way to spend a weekend?
Doing anything outside
If you could have dinner with any historical figure, who would it be?
Ben Franklin
What’s your go-to karaoke song?
You don’t want to hear me sing
What’s one thing on your bucket list?
Skiing in South America
What’s the best piece of advice you’ve ever received?
Follow your heart

Michigan GI Designs a Simple Tool For a Common Problem
Patients sometimes drive hundreds of miles to see their GI physicians for problems that never seem to resolve. Constipation is one of those ailments that can affect quality of life.
The advice is, “Try this diet or laxative. Get a colonoscopy. Often, that’s not getting at the root problem,” said Eric Dinesh Shah, MD, MBA, a gastroenterologist at the University of Michigan, Ann Arbor.
Such methods aren’t equipped to test the pelvic floor, said Dr. Shah, who worked with clinical experts to develop a simple point-of-care device called RED (rectal expulsion device) that makes it easier to diagnose and predict treatment options for constipation.
The device uses a foam-filled balloon to evaluate pelvic floor problems related to constipation, after a digital rectal exam during an office visit. Because the procedure can be performed during a patient’s initial office visit, it can eliminate the need for referrals to far-away specialists for many patients.
In 2019, Dr. Shah received the AGA-Shire Research Scholar Award in Functional GI and Motility Disorders from the AGA Research Foundation for developing RED, and the device was recently cleared by the Food and Drug Administration.
GI doctors don’t always have the answers, he acknowledged in an interview, but this creates the opportunity for new advancements such as RED. utilizing local and regional workshops as well as national conferences to meet like-minded people at similar career stages, and to look for funding opportunities to explore those ideas.
What is the most challenging case you’ve encountered?
Dr. Shah: The most challenging cases to me have been the ones where I wish we could have helped people years ago. It’s not that anyone did anything wrong or was poorly intentioned. It’s quite the opposite: There sometimes is no real avenue to offer testing locally with current technology, even though the local clinical teams completely understand what should be done in a perfect world. That creates challenges where patients go hours out of their way to see specialists, just to find an answer that might have been 1 mile down the road all along.
What has been your solution to help these patients?
Dr. Shah: My work has been about helping patients who drive a hundred miles or routinely go hours out of their way for their care. Usually that’s a sign that things just aren’t working locally. Patients have lost trust in their ability to get care with the teams they have. Or the teams themselves just need help. I think a major part of the job is to reinforce the bond between the patient and their local team by giving them the tools and expertise so that the patients can get that care locally.
There’s been this trend toward this ‘hub and spoke’ model in care where all the patients are filtering into these large hospital-owned mega practices. I wonder about the sustainability of that model because it takes away the ability of patients to see doctors who are invested in their local community. What we need to be doing is trying to flip that.
I’d love to discuss the RED device and how was this device conceived?
Dr. Shah: I partnered with experts, including William Chey, MD, AGAF, at the University of Michigan, who dedicate their entire careers toward creating robust science in large academic medical centers. In understanding the best ways to care for patients today, I could focus my own career on how to translate that level of care for the patients of tomorrow. I would encourage GI trainees to find senior and peer mentors who share perspective on this approach as an anchor to shared success.
For the RED device, the problem in constipation is that patients see their gastroenterologist over and over and over. It’s ‘try this diet, try this laxative, try this drug, try this other treatment,’ and we’re not getting at the root problem. Patients might go through a series of colonoscopies to reassure them but also to reassure their doctor that they’re not missing something. What we haven’t had is a way to test and evaluate the pelvic floor locally because those technologies are high tech and live in these big academic medical centers.
What are plans for its distribution and use in the consumer space?
Dr. Shah: The device is now available in the United States (https://www.red4constipation.com).
As an AGA Research Scholar Award winner, how might AGA play a role in supporting GI doctors?
Dr. Shah: The AGA Research Scholar Award enabled me to learn how RED predicted outcomes for patients seeing general gastroenterologists who then see pelvic floor physical therapy in the community to treat constipation. The availability of pelvic floor physical therapy and the field at large, has exploded in recent years across the country (https://www.pelvicrehab.com), making it easier for patients to get the local care they need.
In looking at what this award did for my own career and those of others in my cohort, I think the AGA Research Scholar Award mechanism serves as an example of what other GI trainees can do across the many areas of GI that are ripe for transformation.
What other AGA workshops are useful to GI doctors?
Dr. Shah: The AGA Tech Summit and Innovation Fellows programs give access to a positive learning environment to network with people across career stages who are seeking to advance the field in this way. These programs are particularly successful because they focus on helping GI trainees find peer success and professional satisfaction in the shared journey, rather than focusing on the accolades. I would strongly encourage GI trainees who have an interest but don’t know where to start to apply for these programs.
What do you think is the biggest misconception about your specialty?
Dr. Shah: That gastroenterologists have all the answers with current technology. There’s a lot we still don’t know. What gives me reassurance is the momentum around new ways of thinking that GI trainees and early-stage gastroenterologists continually bring forward to improve how we care for patients.
Lightning Round
Do you prefer coffee or tea?
Coffee
Are you an early bird or night owl?
Early bird
What’s your go-to comfort food?
Tex Mex
If you could travel anywhere, where would you go?
Antarctica
What’s your favorite TV show?
Below Deck
What’s one hobby you’d like to pick up?
Painting
What’s your favorite way to spend a weekend?
A lazy weekend
If you could have dinner with any historical figure, who would it be?
Winston Churchill
What’s your go-to karaoke song?
Our endoscopy nurses give no choice other than Taylor Swift, Green Day, and the Backstreet Boys
Patients sometimes drive hundreds of miles to see their GI physicians for problems that never seem to resolve. Constipation is one of those ailments that can affect quality of life.
The advice is, “Try this diet or laxative. Get a colonoscopy. Often, that’s not getting at the root problem,” said Eric Dinesh Shah, MD, MBA, a gastroenterologist at the University of Michigan, Ann Arbor.
Such methods aren’t equipped to test the pelvic floor, said Dr. Shah, who worked with clinical experts to develop a simple point-of-care device called RED (rectal expulsion device) that makes it easier to diagnose and predict treatment options for constipation.
The device uses a foam-filled balloon to evaluate pelvic floor problems related to constipation, after a digital rectal exam during an office visit. Because the procedure can be performed during a patient’s initial office visit, it can eliminate the need for referrals to far-away specialists for many patients.
In 2019, Dr. Shah received the AGA-Shire Research Scholar Award in Functional GI and Motility Disorders from the AGA Research Foundation for developing RED, and the device was recently cleared by the Food and Drug Administration.
GI doctors don’t always have the answers, he acknowledged in an interview, but this creates the opportunity for new advancements such as RED. utilizing local and regional workshops as well as national conferences to meet like-minded people at similar career stages, and to look for funding opportunities to explore those ideas.
What is the most challenging case you’ve encountered?
Dr. Shah: The most challenging cases to me have been the ones where I wish we could have helped people years ago. It’s not that anyone did anything wrong or was poorly intentioned. It’s quite the opposite: There sometimes is no real avenue to offer testing locally with current technology, even though the local clinical teams completely understand what should be done in a perfect world. That creates challenges where patients go hours out of their way to see specialists, just to find an answer that might have been 1 mile down the road all along.
What has been your solution to help these patients?
Dr. Shah: My work has been about helping patients who drive a hundred miles or routinely go hours out of their way for their care. Usually that’s a sign that things just aren’t working locally. Patients have lost trust in their ability to get care with the teams they have. Or the teams themselves just need help. I think a major part of the job is to reinforce the bond between the patient and their local team by giving them the tools and expertise so that the patients can get that care locally.
There’s been this trend toward this ‘hub and spoke’ model in care where all the patients are filtering into these large hospital-owned mega practices. I wonder about the sustainability of that model because it takes away the ability of patients to see doctors who are invested in their local community. What we need to be doing is trying to flip that.
I’d love to discuss the RED device and how was this device conceived?
Dr. Shah: I partnered with experts, including William Chey, MD, AGAF, at the University of Michigan, who dedicate their entire careers toward creating robust science in large academic medical centers. In understanding the best ways to care for patients today, I could focus my own career on how to translate that level of care for the patients of tomorrow. I would encourage GI trainees to find senior and peer mentors who share perspective on this approach as an anchor to shared success.
For the RED device, the problem in constipation is that patients see their gastroenterologist over and over and over. It’s ‘try this diet, try this laxative, try this drug, try this other treatment,’ and we’re not getting at the root problem. Patients might go through a series of colonoscopies to reassure them but also to reassure their doctor that they’re not missing something. What we haven’t had is a way to test and evaluate the pelvic floor locally because those technologies are high tech and live in these big academic medical centers.
What are plans for its distribution and use in the consumer space?
Dr. Shah: The device is now available in the United States (https://www.red4constipation.com).
As an AGA Research Scholar Award winner, how might AGA play a role in supporting GI doctors?
Dr. Shah: The AGA Research Scholar Award enabled me to learn how RED predicted outcomes for patients seeing general gastroenterologists who then see pelvic floor physical therapy in the community to treat constipation. The availability of pelvic floor physical therapy and the field at large, has exploded in recent years across the country (https://www.pelvicrehab.com), making it easier for patients to get the local care they need.
In looking at what this award did for my own career and those of others in my cohort, I think the AGA Research Scholar Award mechanism serves as an example of what other GI trainees can do across the many areas of GI that are ripe for transformation.
What other AGA workshops are useful to GI doctors?
Dr. Shah: The AGA Tech Summit and Innovation Fellows programs give access to a positive learning environment to network with people across career stages who are seeking to advance the field in this way. These programs are particularly successful because they focus on helping GI trainees find peer success and professional satisfaction in the shared journey, rather than focusing on the accolades. I would strongly encourage GI trainees who have an interest but don’t know where to start to apply for these programs.
What do you think is the biggest misconception about your specialty?
Dr. Shah: That gastroenterologists have all the answers with current technology. There’s a lot we still don’t know. What gives me reassurance is the momentum around new ways of thinking that GI trainees and early-stage gastroenterologists continually bring forward to improve how we care for patients.
Lightning Round
Do you prefer coffee or tea?
Coffee
Are you an early bird or night owl?
Early bird
What’s your go-to comfort food?
Tex Mex
If you could travel anywhere, where would you go?
Antarctica
What’s your favorite TV show?
Below Deck
What’s one hobby you’d like to pick up?
Painting
What’s your favorite way to spend a weekend?
A lazy weekend
If you could have dinner with any historical figure, who would it be?
Winston Churchill
What’s your go-to karaoke song?
Our endoscopy nurses give no choice other than Taylor Swift, Green Day, and the Backstreet Boys
Patients sometimes drive hundreds of miles to see their GI physicians for problems that never seem to resolve. Constipation is one of those ailments that can affect quality of life.
The advice is, “Try this diet or laxative. Get a colonoscopy. Often, that’s not getting at the root problem,” said Eric Dinesh Shah, MD, MBA, a gastroenterologist at the University of Michigan, Ann Arbor.
Such methods aren’t equipped to test the pelvic floor, said Dr. Shah, who worked with clinical experts to develop a simple point-of-care device called RED (rectal expulsion device) that makes it easier to diagnose and predict treatment options for constipation.
The device uses a foam-filled balloon to evaluate pelvic floor problems related to constipation, after a digital rectal exam during an office visit. Because the procedure can be performed during a patient’s initial office visit, it can eliminate the need for referrals to far-away specialists for many patients.
In 2019, Dr. Shah received the AGA-Shire Research Scholar Award in Functional GI and Motility Disorders from the AGA Research Foundation for developing RED, and the device was recently cleared by the Food and Drug Administration.
GI doctors don’t always have the answers, he acknowledged in an interview, but this creates the opportunity for new advancements such as RED. utilizing local and regional workshops as well as national conferences to meet like-minded people at similar career stages, and to look for funding opportunities to explore those ideas.
What is the most challenging case you’ve encountered?
Dr. Shah: The most challenging cases to me have been the ones where I wish we could have helped people years ago. It’s not that anyone did anything wrong or was poorly intentioned. It’s quite the opposite: There sometimes is no real avenue to offer testing locally with current technology, even though the local clinical teams completely understand what should be done in a perfect world. That creates challenges where patients go hours out of their way to see specialists, just to find an answer that might have been 1 mile down the road all along.
What has been your solution to help these patients?
Dr. Shah: My work has been about helping patients who drive a hundred miles or routinely go hours out of their way for their care. Usually that’s a sign that things just aren’t working locally. Patients have lost trust in their ability to get care with the teams they have. Or the teams themselves just need help. I think a major part of the job is to reinforce the bond between the patient and their local team by giving them the tools and expertise so that the patients can get that care locally.
There’s been this trend toward this ‘hub and spoke’ model in care where all the patients are filtering into these large hospital-owned mega practices. I wonder about the sustainability of that model because it takes away the ability of patients to see doctors who are invested in their local community. What we need to be doing is trying to flip that.
I’d love to discuss the RED device and how was this device conceived?
Dr. Shah: I partnered with experts, including William Chey, MD, AGAF, at the University of Michigan, who dedicate their entire careers toward creating robust science in large academic medical centers. In understanding the best ways to care for patients today, I could focus my own career on how to translate that level of care for the patients of tomorrow. I would encourage GI trainees to find senior and peer mentors who share perspective on this approach as an anchor to shared success.
For the RED device, the problem in constipation is that patients see their gastroenterologist over and over and over. It’s ‘try this diet, try this laxative, try this drug, try this other treatment,’ and we’re not getting at the root problem. Patients might go through a series of colonoscopies to reassure them but also to reassure their doctor that they’re not missing something. What we haven’t had is a way to test and evaluate the pelvic floor locally because those technologies are high tech and live in these big academic medical centers.
What are plans for its distribution and use in the consumer space?
Dr. Shah: The device is now available in the United States (https://www.red4constipation.com).
As an AGA Research Scholar Award winner, how might AGA play a role in supporting GI doctors?
Dr. Shah: The AGA Research Scholar Award enabled me to learn how RED predicted outcomes for patients seeing general gastroenterologists who then see pelvic floor physical therapy in the community to treat constipation. The availability of pelvic floor physical therapy and the field at large, has exploded in recent years across the country (https://www.pelvicrehab.com), making it easier for patients to get the local care they need.
In looking at what this award did for my own career and those of others in my cohort, I think the AGA Research Scholar Award mechanism serves as an example of what other GI trainees can do across the many areas of GI that are ripe for transformation.
What other AGA workshops are useful to GI doctors?
Dr. Shah: The AGA Tech Summit and Innovation Fellows programs give access to a positive learning environment to network with people across career stages who are seeking to advance the field in this way. These programs are particularly successful because they focus on helping GI trainees find peer success and professional satisfaction in the shared journey, rather than focusing on the accolades. I would strongly encourage GI trainees who have an interest but don’t know where to start to apply for these programs.
What do you think is the biggest misconception about your specialty?
Dr. Shah: That gastroenterologists have all the answers with current technology. There’s a lot we still don’t know. What gives me reassurance is the momentum around new ways of thinking that GI trainees and early-stage gastroenterologists continually bring forward to improve how we care for patients.
Lightning Round
Do you prefer coffee or tea?
Coffee
Are you an early bird or night owl?
Early bird
What’s your go-to comfort food?
Tex Mex
If you could travel anywhere, where would you go?
Antarctica
What’s your favorite TV show?
Below Deck
What’s one hobby you’d like to pick up?
Painting
What’s your favorite way to spend a weekend?
A lazy weekend
If you could have dinner with any historical figure, who would it be?
Winston Churchill
What’s your go-to karaoke song?
Our endoscopy nurses give no choice other than Taylor Swift, Green Day, and the Backstreet Boys

Ergonomic ‘Timeouts’ Make Endoscopy Easier For GIs
Amandeep Shergill, MD, MS, AGAF, always thought she had good hand-eye coordination until she entered her gastroenterology fellowship.
“You’re learning how to scope and the endoscope just feels so awkward in the hands. It can be such a difficult instrument to both learn and to use,” said Dr. Shergill, professor of clinical medicine at University of California, San Francisco.
Her attendings and mentors couldn’t give her the feedback she needed.
“I was told that I wasn’t holding it right. But every time I tried to do something that someone was trying to tell me, it seemed like my hands were too small. I couldn’t hold it the way that they were teaching me to hold it.” She began to wonder: Was this about her or the tool itself?
A deep dive into hand tool interactions and medical device designs led her to human factors and ergonomics. Her fellowship mentor, Ken McQuaid, MD, AGAF, had gone to medical school with David Rempel, MD, MPH who was one of the top-funded ergonomists in the country. “He emailed David and wrote: I have a fellow who’s interested in learning more about ergonomics and applying it to endoscopy,” said Dr. Shergill.
Through her work with Dr. Rempel, she was able to uncover the mechanisms that lead to musculoskeletal disorders in endoscopists.
Over time, she has become a trailblazer in this field, helming the UC Berkeley Center for Ergonomic Endoscopy with Carisa Harris-Adamson PhD, CPE, her ergonomics collaborator. In an interview, she described the unique “timeout” algorithm she created to ease the process of endoscopy for GI physicians.
What is your favorite aspect of being a GI physician?
I really love the diversity of patients and cases. You’re always learning something new. It’s an internal medicine subspecialty and a cognitive field, so we must think about differential diagnoses, risks and benefits of procedures for patients. But as a procedural field, we get to diagnose and immediately treat certain disorders. What’s exciting about GI right now is there’s still so much to learn. I think that we’re still discovering more about how the brain-gut interaction works every day. There’s been additional research about the microbiome and the immense influence it has on both health and disease. The field is continuing to evolve rapidly. There’s always something new to learn, and I think it keeps us fresh.
Tell me about your work in ergonomics and endoscopy.
Ken McQuaid connected me with David Rempel. I worked with David to approach this problem of endoscopy ergonomics from a very rigorous ergonomics perspective. Early in my fellowship, endoscopy ergonomics wasn’t well known. There were few survey-based studies, including one from the American Society for Gastrointestinal Endoscopy (ASGE) that documented a high prevalence of endoscopist injury. But not a lot was known about what was causing injury in endoscopists.
What were the risk factors for endoscopist injury? Instead of just doing another survey, I wanted to show that there was this potential for causation given the design of the endoscopes. I worked with David to do a pilot study where we collected some pinch forces and forearm muscle loads. I was able to collect some pilot data that I used to apply for the ASGE Endoscopic Research Award. And luckily, ASGE supported that work.
Another award I received, the ASGE Career Development Award, was instrumental in allowing me to become more proficient in the science of ergonomics. I was able to leverage that career development award to go back to school. I went to UC Berkeley and got a master’s in environmental health sciences with a focus on ergonomics. It really helped me to lay the foundation and understanding for ergonomics and then apply that to endoscopy to generate a more rigorous scientific background for endoscopy ergonomics and start that conversation within the field of GI.
What leads to musculoskeletal disorders in endoscopists and how can it be prevented?
Musculoskeletal disorders are associated with the repetitive procedures that we’re performing, often utilizing high forces and in non-neutral postures. This is because of how we’re interacting with our tools and how we’re interacting with our environments. The studies I have done with Carisa Harris-Adamson have been able to demonstrate and document the high forces that are required to interact with the endoscope. To turn the control section dials and to torque and manipulate the insertion tube, there are really high distal upper extremity muscle loads that are being applied.
We were able to compare the loads and the forces we were seeing to established risk thresholds from the ergonomics literature and demonstrate that performing endoscopy was associated with moderate to high risk of development of distal upper extremity disorders.
What research are you doing now?
We’re trying to focus more on interventions. We’ve done some studies on engineering controls we can utilize to decrease the loads of holding the scope. First, it was an anti-gravity support arm. More recently we’re hoping to publish data on whether a scope stand can alleviate some of those left distal upper extremity loads because the stand is holding the scope instead of the hand holding the scope. Can we decrease injury risk by decreasing static loading?
Neck and back injuries, which have a high prevalence in endoscopists, are usually associated with how the room is set up. One of the things that I’ve tried to help promote is a pre-procedure ergonomic “timeout.” Before an endoscopist does a procedure, we’re supposed to perform a timeout focused on the patient’s safety. We should also try to advocate for physician safety and an ergonomic timeout. I developed a mnemonic device utilizing the word “MYSELF” to help endoscopists remember the ergonomic timeout checklist: M = monitor, Y = upside-down Y stance, S = scope, E = elbow/ bed position, L = lower extremities, F = free movement of endoscope/ processor placement.
First, thinking about the monitor, “M”, and fixing the monitor height so that the neck is in neutral position. Then, thinking of an upside down “Y” standing straight with the feet either hip width or shoulder width apart, so that the physician has a stable, neutral standing posture. Then “S” is for checking the scope to ensure you have a scope with optimal angulation that’s working properly.
“E” is for elbows — adjusting the bed to an optimal position so that elbows and shoulders are in neutral position. “L” is for lower extremities — are the foot pedals within an easy reach? Do you have comfortable shoes on, an anti-fatigue floor mat if you need it? And then the “F” in “MYSELF” is for the processor placement, to ensure “free movement” of the scope. By placing the processor directly behind you and lining up the processor with the orifice to be scoped, you can ensure free movement of the scope so that you can leverage large movements of the control section to result in tip deflection.
We studied the MYSELF mnemonic device for a pre-procedure ergonomic timeout in a simulated setting and presented our results at Digestive Disease Week (DDW) 2024, where we showed a reduction in ergonomic risk scores based on the Rapid Entire Body Assessment tool.
We presented the results of the scope stand study at DDW 2025 in San Diego this May.
What has been the feedback from physicians who use these supportive tools?
While physicians are very grateful for bringing attention to this issue, and many have found utility in some of the tools that I proposed, I think we still have so much work to do. We’re just all hoping to continue to move this field forward for better tools that are designed more with the breadth of endoscopists in mind.
How do you handle stress and maintain work-life balance?
A few years ago, during DDW I gave a talk entitled “Achieving Work-Life Harmony.” I disclosed at the beginning of the talk that I had not achieved work-life harmony. It’s definitely a difficult thing to do, especially in our field as GI proceduralists, where we’re frequently on call and there are potentially on-call emergencies.
One of the key things that I’ve tried to do is create boundaries to prioritize both things in my personal life and my professional life and really try to stay true to the things that are important to me. For instance, things like family time and mealtimes, I think that’s so critical. Trying to be home on evenings for dinnertime is so important.
One of my GI colleagues, Raj Keswani, MD, MS gave a talk about burnout and described imagining life as juggling balls; trying to figure out which balls are glass balls and need to be handled with care, and which balls are rubber balls.
More often, work is the rubber ball. If you drop it, it’ll bounce back and the work that you have will still be there the next day. Family, friends, our health, those are the glass balls that if they fall, they can get scuffed or shatter sometimes. That image helps me think in the moment. If I need to decide between two competing priorities, which one will still be here tomorrow? Which is the one that’s going to be more resilient, and which is the one that I need to focus on? That’s been a helpful image for me.
I also want to give a shout out to my amazing colleagues. We all pitch in with the ‘juggling’ and help to keep everyone’s ‘balls’ in the air, and cover for each other. Whether it’s a sick patient or whatever’s going on in our personal lives, we always take care of each other.
What advice would you give to aspiring GI fellows or graduating fellows?
GI is such an amazing field and many people end up focusing on the procedural aspect of it. What I think defines an exceptional gastroenterologist and physician in general is adopting both a “growth mindset” and a “mastery mindset.” And really, it starts out with when you’re exploring an area of focus, listening to what consistently draws your attention, what you’re excited about learning more about.
Finding mentors, getting involved in projects, doing deep learning, and really trying to develop an expertise in that area through additional training, coursework, and education. I think that idea of a mastery mindset will really help set you up for becoming deeply knowledgeable about a field.
Lightning Round
Coffee or tea?
Coffee
What’s your favorite book?
Project Hail Mary (audiobook)
Beach vacation or mountain retreat?
Mountain retreat
Early bird or night owl?
Night owl
What’s your go-to comfort food?
Chaat (Indian street food)
Do you prefer dogs or cats?
Dogs
What’s one hobby you’d like to pick up?
Sewing
If you could have dinner with any historical figure, who would it be?
Ruth Bader Ginsburg
What’s your go-to karaoke song?
I Wanna Dance with Somebody
What’s one thing on your bucket list?
To see the Northern Lights
Amandeep Shergill, MD, MS, AGAF, always thought she had good hand-eye coordination until she entered her gastroenterology fellowship.
“You’re learning how to scope and the endoscope just feels so awkward in the hands. It can be such a difficult instrument to both learn and to use,” said Dr. Shergill, professor of clinical medicine at University of California, San Francisco.
Her attendings and mentors couldn’t give her the feedback she needed.
“I was told that I wasn’t holding it right. But every time I tried to do something that someone was trying to tell me, it seemed like my hands were too small. I couldn’t hold it the way that they were teaching me to hold it.” She began to wonder: Was this about her or the tool itself?
A deep dive into hand tool interactions and medical device designs led her to human factors and ergonomics. Her fellowship mentor, Ken McQuaid, MD, AGAF, had gone to medical school with David Rempel, MD, MPH who was one of the top-funded ergonomists in the country. “He emailed David and wrote: I have a fellow who’s interested in learning more about ergonomics and applying it to endoscopy,” said Dr. Shergill.
Through her work with Dr. Rempel, she was able to uncover the mechanisms that lead to musculoskeletal disorders in endoscopists.
Over time, she has become a trailblazer in this field, helming the UC Berkeley Center for Ergonomic Endoscopy with Carisa Harris-Adamson PhD, CPE, her ergonomics collaborator. In an interview, she described the unique “timeout” algorithm she created to ease the process of endoscopy for GI physicians.
What is your favorite aspect of being a GI physician?
I really love the diversity of patients and cases. You’re always learning something new. It’s an internal medicine subspecialty and a cognitive field, so we must think about differential diagnoses, risks and benefits of procedures for patients. But as a procedural field, we get to diagnose and immediately treat certain disorders. What’s exciting about GI right now is there’s still so much to learn. I think that we’re still discovering more about how the brain-gut interaction works every day. There’s been additional research about the microbiome and the immense influence it has on both health and disease. The field is continuing to evolve rapidly. There’s always something new to learn, and I think it keeps us fresh.
Tell me about your work in ergonomics and endoscopy.
Ken McQuaid connected me with David Rempel. I worked with David to approach this problem of endoscopy ergonomics from a very rigorous ergonomics perspective. Early in my fellowship, endoscopy ergonomics wasn’t well known. There were few survey-based studies, including one from the American Society for Gastrointestinal Endoscopy (ASGE) that documented a high prevalence of endoscopist injury. But not a lot was known about what was causing injury in endoscopists.
What were the risk factors for endoscopist injury? Instead of just doing another survey, I wanted to show that there was this potential for causation given the design of the endoscopes. I worked with David to do a pilot study where we collected some pinch forces and forearm muscle loads. I was able to collect some pilot data that I used to apply for the ASGE Endoscopic Research Award. And luckily, ASGE supported that work.
Another award I received, the ASGE Career Development Award, was instrumental in allowing me to become more proficient in the science of ergonomics. I was able to leverage that career development award to go back to school. I went to UC Berkeley and got a master’s in environmental health sciences with a focus on ergonomics. It really helped me to lay the foundation and understanding for ergonomics and then apply that to endoscopy to generate a more rigorous scientific background for endoscopy ergonomics and start that conversation within the field of GI.
What leads to musculoskeletal disorders in endoscopists and how can it be prevented?
Musculoskeletal disorders are associated with the repetitive procedures that we’re performing, often utilizing high forces and in non-neutral postures. This is because of how we’re interacting with our tools and how we’re interacting with our environments. The studies I have done with Carisa Harris-Adamson have been able to demonstrate and document the high forces that are required to interact with the endoscope. To turn the control section dials and to torque and manipulate the insertion tube, there are really high distal upper extremity muscle loads that are being applied.
We were able to compare the loads and the forces we were seeing to established risk thresholds from the ergonomics literature and demonstrate that performing endoscopy was associated with moderate to high risk of development of distal upper extremity disorders.
What research are you doing now?
We’re trying to focus more on interventions. We’ve done some studies on engineering controls we can utilize to decrease the loads of holding the scope. First, it was an anti-gravity support arm. More recently we’re hoping to publish data on whether a scope stand can alleviate some of those left distal upper extremity loads because the stand is holding the scope instead of the hand holding the scope. Can we decrease injury risk by decreasing static loading?
Neck and back injuries, which have a high prevalence in endoscopists, are usually associated with how the room is set up. One of the things that I’ve tried to help promote is a pre-procedure ergonomic “timeout.” Before an endoscopist does a procedure, we’re supposed to perform a timeout focused on the patient’s safety. We should also try to advocate for physician safety and an ergonomic timeout. I developed a mnemonic device utilizing the word “MYSELF” to help endoscopists remember the ergonomic timeout checklist: M = monitor, Y = upside-down Y stance, S = scope, E = elbow/ bed position, L = lower extremities, F = free movement of endoscope/ processor placement.
First, thinking about the monitor, “M”, and fixing the monitor height so that the neck is in neutral position. Then, thinking of an upside down “Y” standing straight with the feet either hip width or shoulder width apart, so that the physician has a stable, neutral standing posture. Then “S” is for checking the scope to ensure you have a scope with optimal angulation that’s working properly.
“E” is for elbows — adjusting the bed to an optimal position so that elbows and shoulders are in neutral position. “L” is for lower extremities — are the foot pedals within an easy reach? Do you have comfortable shoes on, an anti-fatigue floor mat if you need it? And then the “F” in “MYSELF” is for the processor placement, to ensure “free movement” of the scope. By placing the processor directly behind you and lining up the processor with the orifice to be scoped, you can ensure free movement of the scope so that you can leverage large movements of the control section to result in tip deflection.
We studied the MYSELF mnemonic device for a pre-procedure ergonomic timeout in a simulated setting and presented our results at Digestive Disease Week (DDW) 2024, where we showed a reduction in ergonomic risk scores based on the Rapid Entire Body Assessment tool.
We presented the results of the scope stand study at DDW 2025 in San Diego this May.
What has been the feedback from physicians who use these supportive tools?
While physicians are very grateful for bringing attention to this issue, and many have found utility in some of the tools that I proposed, I think we still have so much work to do. We’re just all hoping to continue to move this field forward for better tools that are designed more with the breadth of endoscopists in mind.
How do you handle stress and maintain work-life balance?
A few years ago, during DDW I gave a talk entitled “Achieving Work-Life Harmony.” I disclosed at the beginning of the talk that I had not achieved work-life harmony. It’s definitely a difficult thing to do, especially in our field as GI proceduralists, where we’re frequently on call and there are potentially on-call emergencies.
One of the key things that I’ve tried to do is create boundaries to prioritize both things in my personal life and my professional life and really try to stay true to the things that are important to me. For instance, things like family time and mealtimes, I think that’s so critical. Trying to be home on evenings for dinnertime is so important.
One of my GI colleagues, Raj Keswani, MD, MS gave a talk about burnout and described imagining life as juggling balls; trying to figure out which balls are glass balls and need to be handled with care, and which balls are rubber balls.
More often, work is the rubber ball. If you drop it, it’ll bounce back and the work that you have will still be there the next day. Family, friends, our health, those are the glass balls that if they fall, they can get scuffed or shatter sometimes. That image helps me think in the moment. If I need to decide between two competing priorities, which one will still be here tomorrow? Which is the one that’s going to be more resilient, and which is the one that I need to focus on? That’s been a helpful image for me.
I also want to give a shout out to my amazing colleagues. We all pitch in with the ‘juggling’ and help to keep everyone’s ‘balls’ in the air, and cover for each other. Whether it’s a sick patient or whatever’s going on in our personal lives, we always take care of each other.
What advice would you give to aspiring GI fellows or graduating fellows?
GI is such an amazing field and many people end up focusing on the procedural aspect of it. What I think defines an exceptional gastroenterologist and physician in general is adopting both a “growth mindset” and a “mastery mindset.” And really, it starts out with when you’re exploring an area of focus, listening to what consistently draws your attention, what you’re excited about learning more about.
Finding mentors, getting involved in projects, doing deep learning, and really trying to develop an expertise in that area through additional training, coursework, and education. I think that idea of a mastery mindset will really help set you up for becoming deeply knowledgeable about a field.
Lightning Round
Coffee or tea?
Coffee
What’s your favorite book?
Project Hail Mary (audiobook)
Beach vacation or mountain retreat?
Mountain retreat
Early bird or night owl?
Night owl
What’s your go-to comfort food?
Chaat (Indian street food)
Do you prefer dogs or cats?
Dogs
What’s one hobby you’d like to pick up?
Sewing
If you could have dinner with any historical figure, who would it be?
Ruth Bader Ginsburg
What’s your go-to karaoke song?
I Wanna Dance with Somebody
What’s one thing on your bucket list?
To see the Northern Lights
Amandeep Shergill, MD, MS, AGAF, always thought she had good hand-eye coordination until she entered her gastroenterology fellowship.
“You’re learning how to scope and the endoscope just feels so awkward in the hands. It can be such a difficult instrument to both learn and to use,” said Dr. Shergill, professor of clinical medicine at University of California, San Francisco.
Her attendings and mentors couldn’t give her the feedback she needed.
“I was told that I wasn’t holding it right. But every time I tried to do something that someone was trying to tell me, it seemed like my hands were too small. I couldn’t hold it the way that they were teaching me to hold it.” She began to wonder: Was this about her or the tool itself?
A deep dive into hand tool interactions and medical device designs led her to human factors and ergonomics. Her fellowship mentor, Ken McQuaid, MD, AGAF, had gone to medical school with David Rempel, MD, MPH who was one of the top-funded ergonomists in the country. “He emailed David and wrote: I have a fellow who’s interested in learning more about ergonomics and applying it to endoscopy,” said Dr. Shergill.
Through her work with Dr. Rempel, she was able to uncover the mechanisms that lead to musculoskeletal disorders in endoscopists.
Over time, she has become a trailblazer in this field, helming the UC Berkeley Center for Ergonomic Endoscopy with Carisa Harris-Adamson PhD, CPE, her ergonomics collaborator. In an interview, she described the unique “timeout” algorithm she created to ease the process of endoscopy for GI physicians.
What is your favorite aspect of being a GI physician?
I really love the diversity of patients and cases. You’re always learning something new. It’s an internal medicine subspecialty and a cognitive field, so we must think about differential diagnoses, risks and benefits of procedures for patients. But as a procedural field, we get to diagnose and immediately treat certain disorders. What’s exciting about GI right now is there’s still so much to learn. I think that we’re still discovering more about how the brain-gut interaction works every day. There’s been additional research about the microbiome and the immense influence it has on both health and disease. The field is continuing to evolve rapidly. There’s always something new to learn, and I think it keeps us fresh.
Tell me about your work in ergonomics and endoscopy.
Ken McQuaid connected me with David Rempel. I worked with David to approach this problem of endoscopy ergonomics from a very rigorous ergonomics perspective. Early in my fellowship, endoscopy ergonomics wasn’t well known. There were few survey-based studies, including one from the American Society for Gastrointestinal Endoscopy (ASGE) that documented a high prevalence of endoscopist injury. But not a lot was known about what was causing injury in endoscopists.
What were the risk factors for endoscopist injury? Instead of just doing another survey, I wanted to show that there was this potential for causation given the design of the endoscopes. I worked with David to do a pilot study where we collected some pinch forces and forearm muscle loads. I was able to collect some pilot data that I used to apply for the ASGE Endoscopic Research Award. And luckily, ASGE supported that work.
Another award I received, the ASGE Career Development Award, was instrumental in allowing me to become more proficient in the science of ergonomics. I was able to leverage that career development award to go back to school. I went to UC Berkeley and got a master’s in environmental health sciences with a focus on ergonomics. It really helped me to lay the foundation and understanding for ergonomics and then apply that to endoscopy to generate a more rigorous scientific background for endoscopy ergonomics and start that conversation within the field of GI.
What leads to musculoskeletal disorders in endoscopists and how can it be prevented?
Musculoskeletal disorders are associated with the repetitive procedures that we’re performing, often utilizing high forces and in non-neutral postures. This is because of how we’re interacting with our tools and how we’re interacting with our environments. The studies I have done with Carisa Harris-Adamson have been able to demonstrate and document the high forces that are required to interact with the endoscope. To turn the control section dials and to torque and manipulate the insertion tube, there are really high distal upper extremity muscle loads that are being applied.
We were able to compare the loads and the forces we were seeing to established risk thresholds from the ergonomics literature and demonstrate that performing endoscopy was associated with moderate to high risk of development of distal upper extremity disorders.
What research are you doing now?
We’re trying to focus more on interventions. We’ve done some studies on engineering controls we can utilize to decrease the loads of holding the scope. First, it was an anti-gravity support arm. More recently we’re hoping to publish data on whether a scope stand can alleviate some of those left distal upper extremity loads because the stand is holding the scope instead of the hand holding the scope. Can we decrease injury risk by decreasing static loading?
Neck and back injuries, which have a high prevalence in endoscopists, are usually associated with how the room is set up. One of the things that I’ve tried to help promote is a pre-procedure ergonomic “timeout.” Before an endoscopist does a procedure, we’re supposed to perform a timeout focused on the patient’s safety. We should also try to advocate for physician safety and an ergonomic timeout. I developed a mnemonic device utilizing the word “MYSELF” to help endoscopists remember the ergonomic timeout checklist: M = monitor, Y = upside-down Y stance, S = scope, E = elbow/ bed position, L = lower extremities, F = free movement of endoscope/ processor placement.
First, thinking about the monitor, “M”, and fixing the monitor height so that the neck is in neutral position. Then, thinking of an upside down “Y” standing straight with the feet either hip width or shoulder width apart, so that the physician has a stable, neutral standing posture. Then “S” is for checking the scope to ensure you have a scope with optimal angulation that’s working properly.
“E” is for elbows — adjusting the bed to an optimal position so that elbows and shoulders are in neutral position. “L” is for lower extremities — are the foot pedals within an easy reach? Do you have comfortable shoes on, an anti-fatigue floor mat if you need it? And then the “F” in “MYSELF” is for the processor placement, to ensure “free movement” of the scope. By placing the processor directly behind you and lining up the processor with the orifice to be scoped, you can ensure free movement of the scope so that you can leverage large movements of the control section to result in tip deflection.
We studied the MYSELF mnemonic device for a pre-procedure ergonomic timeout in a simulated setting and presented our results at Digestive Disease Week (DDW) 2024, where we showed a reduction in ergonomic risk scores based on the Rapid Entire Body Assessment tool.
We presented the results of the scope stand study at DDW 2025 in San Diego this May.
What has been the feedback from physicians who use these supportive tools?
While physicians are very grateful for bringing attention to this issue, and many have found utility in some of the tools that I proposed, I think we still have so much work to do. We’re just all hoping to continue to move this field forward for better tools that are designed more with the breadth of endoscopists in mind.
How do you handle stress and maintain work-life balance?
A few years ago, during DDW I gave a talk entitled “Achieving Work-Life Harmony.” I disclosed at the beginning of the talk that I had not achieved work-life harmony. It’s definitely a difficult thing to do, especially in our field as GI proceduralists, where we’re frequently on call and there are potentially on-call emergencies.
One of the key things that I’ve tried to do is create boundaries to prioritize both things in my personal life and my professional life and really try to stay true to the things that are important to me. For instance, things like family time and mealtimes, I think that’s so critical. Trying to be home on evenings for dinnertime is so important.
One of my GI colleagues, Raj Keswani, MD, MS gave a talk about burnout and described imagining life as juggling balls; trying to figure out which balls are glass balls and need to be handled with care, and which balls are rubber balls.
More often, work is the rubber ball. If you drop it, it’ll bounce back and the work that you have will still be there the next day. Family, friends, our health, those are the glass balls that if they fall, they can get scuffed or shatter sometimes. That image helps me think in the moment. If I need to decide between two competing priorities, which one will still be here tomorrow? Which is the one that’s going to be more resilient, and which is the one that I need to focus on? That’s been a helpful image for me.
I also want to give a shout out to my amazing colleagues. We all pitch in with the ‘juggling’ and help to keep everyone’s ‘balls’ in the air, and cover for each other. Whether it’s a sick patient or whatever’s going on in our personal lives, we always take care of each other.
What advice would you give to aspiring GI fellows or graduating fellows?
GI is such an amazing field and many people end up focusing on the procedural aspect of it. What I think defines an exceptional gastroenterologist and physician in general is adopting both a “growth mindset” and a “mastery mindset.” And really, it starts out with when you’re exploring an area of focus, listening to what consistently draws your attention, what you’re excited about learning more about.
Finding mentors, getting involved in projects, doing deep learning, and really trying to develop an expertise in that area through additional training, coursework, and education. I think that idea of a mastery mindset will really help set you up for becoming deeply knowledgeable about a field.
Lightning Round
Coffee or tea?
Coffee
What’s your favorite book?
Project Hail Mary (audiobook)
Beach vacation or mountain retreat?
Mountain retreat
Early bird or night owl?
Night owl
What’s your go-to comfort food?
Chaat (Indian street food)
Do you prefer dogs or cats?
Dogs
What’s one hobby you’d like to pick up?
Sewing
If you could have dinner with any historical figure, who would it be?
Ruth Bader Ginsburg
What’s your go-to karaoke song?
I Wanna Dance with Somebody
What’s one thing on your bucket list?
To see the Northern Lights

Bringing HCC Patients Hope Through Trials, Advanced Treatments
For Reena Salgia, MD, the most rewarding part about working with patients with hepatocellular carcinoma is being there for their entire journey, thanks to advancements in treatment. “It brings a smile to my face just to think about it,” says Dr. Salgia, medical director of Henry Ford Health’s Liver Cancer Clinic in Detroit.
Hepatocellular carcinoma accounts for 80% of all liver cancer. When she first entered the field, Dr. Salgia often heard that survival rates 5 years after diagnosis were less than 10%. Over the last decade however, “I’ve seen an expansion in the procedural options that we offer these patients. We have an array of options both surgically as well as procedurally,” she said.
Especially over the last three to four years, “we’ve seen meaningful responses for patients with medications that we previously didn’t have in our toolbox. That’s really been exciting, along with continued involvement in clinical trials and being able to offer patients a number of different approaches to their care of liver cancer,” said Dr. Salgia.
A regular attendee and presenter at national GI meetings, Dr. Salgia participated in AGA’s Women’s Executive Leadership Conference in 2023. Her academic resume includes a long list of clinical trials to assess treatments for patients at different stages of hepatocellular carcinoma.
In an interview, she discussed the highlights of her career as a researcher and mentor of fellows, and how she guides and supports her transplant patients.
What drove you to pursue the field of hepatology and transplant hepatology?
I came across this field during my fourth year of medical school. I didn’t know anything about hepatology when I reached that stage and had the opportunity to do an elective. I just fell in love with the specialty. I liked the complex pathophysiology of liver disease, the long-term follow-up and care of patients. It appealed to the type of science that I had enjoyed back in college.
As I went into my GI fellowship training, I got to learn more about the field of transplant medicine. For instance, how you can take these patients who are incredibly ill, really at a very vulnerable point of their illness, and then offer them great hope and see their lives turn around afterwards. When I had the opportunity to see patients go from end stage liver disease to such significant improvement in their quality of life, and restoring their physical functioning beyond what we would’ve ever imagined when they were ill, it reaffirmed my interest in both hepatology as well as in transplant medicine.
How do you help those patients waiting on transplant lists for a liver?
We are intimately involved in their care all the way through their journey with liver disease, up until the time of physically getting the liver transplant, which is performed by our colleagues in transplant surgery. From the time they are transplanted, we are involved in their inpatient and outpatient post-transplant care. We’ve helped to get them on the transplant list with the work of the multidisciplinary team. If there are opportunities to help them understand their position on the list or obtaining exceptions—though that is done in a very objective fashion through the regulatory system—we help to guide them through that journey.
You’ve worked on many studies that involve treatments for hepatocellular carcinoma. Can you highlight a paper that yielded clinically significant benefits?
What really stands out the most to me was our site’s involvement in the IMbrave150 trial, which was published in 2020. This multicenter study made a big difference in the outcomes and treatments for patients, as it brought the adoption of first-line immunotherapy (atezolizumab plus bevacizumab) for patients with advanced hepatocellular carcinoma. I remember vividly the patients we had the opportunity to enroll in that trial – some who we continue to care for today. This stands out as one of the trials that I was involved in that had a lasting impact.
What were the clinical endpoints and key results of that trial?
The endpoint was to see an improvement in overall survival utilizing immunotherapy, compared with the prior standard of care then available, oral therapy. The results led to the adoption and FDA approval of immunotherapy in the first line setting for advanced unresectable hepatocellular carcinoma patients.
What are some of the highlights of serving as director of Henry Ford’s fellowship program?
Education is my passion. I went into medical training feeling that at some point I would love to blend in teaching in a formal role. Becoming program director of the gastroenterology and hepatology fellowship at Henry Ford in 2018 was one of the most meaningful things that I’ve had the opportunity to do in my career. I get to see trainees who are at a very impressionable point of their journey go on to become gastroenterologists and then launch into their first job and really develop in this field. Seeing them come in day one, not knowing how to hold a scope or do a procedure on a patient of this nature, then quickly evolve over the first year and grow over three years to achieve this specialty training [is rewarding]. I’ve learned a lot from the fellows along the way. I think of them as an extension of my family. We have 15 fellows currently in our program and we’ll be growing this summer. So that’s really been a highlight of my career thus far.
What fears did you have to push past to get to where you are in your career?
I think that there have been a few. One is certainly the fear of making the wrong choice with your first career opportunity. I did choose to leave my comfort zone from where I had done my training. I met that with some fear, but also excitement for new opportunities of personal and professional growth.
Another fear is: Am I going to be able to be ambitious in this field? Can I pursue research, become a program director, and do things that my role models and mentors were able to achieve? There’s also the fear of being able to balance a busy work life with a busy home life and figuring out how to do both well and minimize the guilt on both sides. I have a family with two girls. They are definitely a top priority.
What teacher or mentor had the greatest impact on you?
Helen Te, MD, a hepatologist at the University of Chicago. When I was a medical student there, I had the opportunity to work with her and saw her passion for this field. She really had so much enthusiasm for teaching and was a big part of why I started to fall in love with liver disease.
Karen Kim, MD, now the dean of Penn State College of Medicine, was one of my assigned mentors as a medical student. She helped me explore the fields where there were opportunities for residency and helped me make the decision to go into internal medicine, which often is a key deciding point for medical students. She was also a very influential teacher. The other individual who stands out is my fellowship program director, Hari Sree Conjeevaram, MD, MSc, at University of Michigan Health. He exhibited the qualities as an educator and program director that helped me recognize that education was something that I wanted to pursue in a formal fashion once I moved on in my career.
Describe how you would spend a free Saturday afternoon.
Likely taking a hike or go to a park with my family, enjoying the outdoors and spending time with them.
Lightning Round
If you weren’t a gastroenterologist, what would you be?
Philanthropist
Favorite city in U.S. besides the one you live in?
Chicago
Place you most want to travel?
New Zealand
Favorite breakfast?
Avocado toast
Favorite ice cream flavor?
Cookies and cream
How many cups of coffee do you drink per day?
Two…or more
Cat person or dog person?
Dog
Texting or talking?
Talk
Favorite season?
Autumn
Favorite type of music?
Pop
Favorite movie genre?
Action
For Reena Salgia, MD, the most rewarding part about working with patients with hepatocellular carcinoma is being there for their entire journey, thanks to advancements in treatment. “It brings a smile to my face just to think about it,” says Dr. Salgia, medical director of Henry Ford Health’s Liver Cancer Clinic in Detroit.
Hepatocellular carcinoma accounts for 80% of all liver cancer. When she first entered the field, Dr. Salgia often heard that survival rates 5 years after diagnosis were less than 10%. Over the last decade however, “I’ve seen an expansion in the procedural options that we offer these patients. We have an array of options both surgically as well as procedurally,” she said.
Especially over the last three to four years, “we’ve seen meaningful responses for patients with medications that we previously didn’t have in our toolbox. That’s really been exciting, along with continued involvement in clinical trials and being able to offer patients a number of different approaches to their care of liver cancer,” said Dr. Salgia.
A regular attendee and presenter at national GI meetings, Dr. Salgia participated in AGA’s Women’s Executive Leadership Conference in 2023. Her academic resume includes a long list of clinical trials to assess treatments for patients at different stages of hepatocellular carcinoma.
In an interview, she discussed the highlights of her career as a researcher and mentor of fellows, and how she guides and supports her transplant patients.
What drove you to pursue the field of hepatology and transplant hepatology?
I came across this field during my fourth year of medical school. I didn’t know anything about hepatology when I reached that stage and had the opportunity to do an elective. I just fell in love with the specialty. I liked the complex pathophysiology of liver disease, the long-term follow-up and care of patients. It appealed to the type of science that I had enjoyed back in college.
As I went into my GI fellowship training, I got to learn more about the field of transplant medicine. For instance, how you can take these patients who are incredibly ill, really at a very vulnerable point of their illness, and then offer them great hope and see their lives turn around afterwards. When I had the opportunity to see patients go from end stage liver disease to such significant improvement in their quality of life, and restoring their physical functioning beyond what we would’ve ever imagined when they were ill, it reaffirmed my interest in both hepatology as well as in transplant medicine.
How do you help those patients waiting on transplant lists for a liver?
We are intimately involved in their care all the way through their journey with liver disease, up until the time of physically getting the liver transplant, which is performed by our colleagues in transplant surgery. From the time they are transplanted, we are involved in their inpatient and outpatient post-transplant care. We’ve helped to get them on the transplant list with the work of the multidisciplinary team. If there are opportunities to help them understand their position on the list or obtaining exceptions—though that is done in a very objective fashion through the regulatory system—we help to guide them through that journey.
You’ve worked on many studies that involve treatments for hepatocellular carcinoma. Can you highlight a paper that yielded clinically significant benefits?
What really stands out the most to me was our site’s involvement in the IMbrave150 trial, which was published in 2020. This multicenter study made a big difference in the outcomes and treatments for patients, as it brought the adoption of first-line immunotherapy (atezolizumab plus bevacizumab) for patients with advanced hepatocellular carcinoma. I remember vividly the patients we had the opportunity to enroll in that trial – some who we continue to care for today. This stands out as one of the trials that I was involved in that had a lasting impact.
What were the clinical endpoints and key results of that trial?
The endpoint was to see an improvement in overall survival utilizing immunotherapy, compared with the prior standard of care then available, oral therapy. The results led to the adoption and FDA approval of immunotherapy in the first line setting for advanced unresectable hepatocellular carcinoma patients.
What are some of the highlights of serving as director of Henry Ford’s fellowship program?
Education is my passion. I went into medical training feeling that at some point I would love to blend in teaching in a formal role. Becoming program director of the gastroenterology and hepatology fellowship at Henry Ford in 2018 was one of the most meaningful things that I’ve had the opportunity to do in my career. I get to see trainees who are at a very impressionable point of their journey go on to become gastroenterologists and then launch into their first job and really develop in this field. Seeing them come in day one, not knowing how to hold a scope or do a procedure on a patient of this nature, then quickly evolve over the first year and grow over three years to achieve this specialty training [is rewarding]. I’ve learned a lot from the fellows along the way. I think of them as an extension of my family. We have 15 fellows currently in our program and we’ll be growing this summer. So that’s really been a highlight of my career thus far.
What fears did you have to push past to get to where you are in your career?
I think that there have been a few. One is certainly the fear of making the wrong choice with your first career opportunity. I did choose to leave my comfort zone from where I had done my training. I met that with some fear, but also excitement for new opportunities of personal and professional growth.
Another fear is: Am I going to be able to be ambitious in this field? Can I pursue research, become a program director, and do things that my role models and mentors were able to achieve? There’s also the fear of being able to balance a busy work life with a busy home life and figuring out how to do both well and minimize the guilt on both sides. I have a family with two girls. They are definitely a top priority.
What teacher or mentor had the greatest impact on you?
Helen Te, MD, a hepatologist at the University of Chicago. When I was a medical student there, I had the opportunity to work with her and saw her passion for this field. She really had so much enthusiasm for teaching and was a big part of why I started to fall in love with liver disease.
Karen Kim, MD, now the dean of Penn State College of Medicine, was one of my assigned mentors as a medical student. She helped me explore the fields where there were opportunities for residency and helped me make the decision to go into internal medicine, which often is a key deciding point for medical students. She was also a very influential teacher. The other individual who stands out is my fellowship program director, Hari Sree Conjeevaram, MD, MSc, at University of Michigan Health. He exhibited the qualities as an educator and program director that helped me recognize that education was something that I wanted to pursue in a formal fashion once I moved on in my career.
Describe how you would spend a free Saturday afternoon.
Likely taking a hike or go to a park with my family, enjoying the outdoors and spending time with them.
Lightning Round
If you weren’t a gastroenterologist, what would you be?
Philanthropist
Favorite city in U.S. besides the one you live in?
Chicago
Place you most want to travel?
New Zealand
Favorite breakfast?
Avocado toast
Favorite ice cream flavor?
Cookies and cream
How many cups of coffee do you drink per day?
Two…or more
Cat person or dog person?
Dog
Texting or talking?
Talk
Favorite season?
Autumn
Favorite type of music?
Pop
Favorite movie genre?
Action
For Reena Salgia, MD, the most rewarding part about working with patients with hepatocellular carcinoma is being there for their entire journey, thanks to advancements in treatment. “It brings a smile to my face just to think about it,” says Dr. Salgia, medical director of Henry Ford Health’s Liver Cancer Clinic in Detroit.
Hepatocellular carcinoma accounts for 80% of all liver cancer. When she first entered the field, Dr. Salgia often heard that survival rates 5 years after diagnosis were less than 10%. Over the last decade however, “I’ve seen an expansion in the procedural options that we offer these patients. We have an array of options both surgically as well as procedurally,” she said.
Especially over the last three to four years, “we’ve seen meaningful responses for patients with medications that we previously didn’t have in our toolbox. That’s really been exciting, along with continued involvement in clinical trials and being able to offer patients a number of different approaches to their care of liver cancer,” said Dr. Salgia.
A regular attendee and presenter at national GI meetings, Dr. Salgia participated in AGA’s Women’s Executive Leadership Conference in 2023. Her academic resume includes a long list of clinical trials to assess treatments for patients at different stages of hepatocellular carcinoma.
In an interview, she discussed the highlights of her career as a researcher and mentor of fellows, and how she guides and supports her transplant patients.
What drove you to pursue the field of hepatology and transplant hepatology?
I came across this field during my fourth year of medical school. I didn’t know anything about hepatology when I reached that stage and had the opportunity to do an elective. I just fell in love with the specialty. I liked the complex pathophysiology of liver disease, the long-term follow-up and care of patients. It appealed to the type of science that I had enjoyed back in college.
As I went into my GI fellowship training, I got to learn more about the field of transplant medicine. For instance, how you can take these patients who are incredibly ill, really at a very vulnerable point of their illness, and then offer them great hope and see their lives turn around afterwards. When I had the opportunity to see patients go from end stage liver disease to such significant improvement in their quality of life, and restoring their physical functioning beyond what we would’ve ever imagined when they were ill, it reaffirmed my interest in both hepatology as well as in transplant medicine.
How do you help those patients waiting on transplant lists for a liver?
We are intimately involved in their care all the way through their journey with liver disease, up until the time of physically getting the liver transplant, which is performed by our colleagues in transplant surgery. From the time they are transplanted, we are involved in their inpatient and outpatient post-transplant care. We’ve helped to get them on the transplant list with the work of the multidisciplinary team. If there are opportunities to help them understand their position on the list or obtaining exceptions—though that is done in a very objective fashion through the regulatory system—we help to guide them through that journey.
You’ve worked on many studies that involve treatments for hepatocellular carcinoma. Can you highlight a paper that yielded clinically significant benefits?
What really stands out the most to me was our site’s involvement in the IMbrave150 trial, which was published in 2020. This multicenter study made a big difference in the outcomes and treatments for patients, as it brought the adoption of first-line immunotherapy (atezolizumab plus bevacizumab) for patients with advanced hepatocellular carcinoma. I remember vividly the patients we had the opportunity to enroll in that trial – some who we continue to care for today. This stands out as one of the trials that I was involved in that had a lasting impact.
What were the clinical endpoints and key results of that trial?
The endpoint was to see an improvement in overall survival utilizing immunotherapy, compared with the prior standard of care then available, oral therapy. The results led to the adoption and FDA approval of immunotherapy in the first line setting for advanced unresectable hepatocellular carcinoma patients.
What are some of the highlights of serving as director of Henry Ford’s fellowship program?
Education is my passion. I went into medical training feeling that at some point I would love to blend in teaching in a formal role. Becoming program director of the gastroenterology and hepatology fellowship at Henry Ford in 2018 was one of the most meaningful things that I’ve had the opportunity to do in my career. I get to see trainees who are at a very impressionable point of their journey go on to become gastroenterologists and then launch into their first job and really develop in this field. Seeing them come in day one, not knowing how to hold a scope or do a procedure on a patient of this nature, then quickly evolve over the first year and grow over three years to achieve this specialty training [is rewarding]. I’ve learned a lot from the fellows along the way. I think of them as an extension of my family. We have 15 fellows currently in our program and we’ll be growing this summer. So that’s really been a highlight of my career thus far.
What fears did you have to push past to get to where you are in your career?
I think that there have been a few. One is certainly the fear of making the wrong choice with your first career opportunity. I did choose to leave my comfort zone from where I had done my training. I met that with some fear, but also excitement for new opportunities of personal and professional growth.
Another fear is: Am I going to be able to be ambitious in this field? Can I pursue research, become a program director, and do things that my role models and mentors were able to achieve? There’s also the fear of being able to balance a busy work life with a busy home life and figuring out how to do both well and minimize the guilt on both sides. I have a family with two girls. They are definitely a top priority.
What teacher or mentor had the greatest impact on you?
Helen Te, MD, a hepatologist at the University of Chicago. When I was a medical student there, I had the opportunity to work with her and saw her passion for this field. She really had so much enthusiasm for teaching and was a big part of why I started to fall in love with liver disease.
Karen Kim, MD, now the dean of Penn State College of Medicine, was one of my assigned mentors as a medical student. She helped me explore the fields where there were opportunities for residency and helped me make the decision to go into internal medicine, which often is a key deciding point for medical students. She was also a very influential teacher. The other individual who stands out is my fellowship program director, Hari Sree Conjeevaram, MD, MSc, at University of Michigan Health. He exhibited the qualities as an educator and program director that helped me recognize that education was something that I wanted to pursue in a formal fashion once I moved on in my career.
Describe how you would spend a free Saturday afternoon.
Likely taking a hike or go to a park with my family, enjoying the outdoors and spending time with them.
Lightning Round
If you weren’t a gastroenterologist, what would you be?
Philanthropist
Favorite city in U.S. besides the one you live in?
Chicago
Place you most want to travel?
New Zealand
Favorite breakfast?
Avocado toast
Favorite ice cream flavor?
Cookies and cream
How many cups of coffee do you drink per day?
Two…or more
Cat person or dog person?
Dog
Texting or talking?
Talk
Favorite season?
Autumn
Favorite type of music?
Pop
Favorite movie genre?
Action

Giving the Smallest GI Transplant Patients a New Lease On Life
The best part about working with kids is that “I get to laugh every day,” said Ke-You (Yoyo) Zhang, MD, clinical assistant professor for pediatrics–gastroenterology and hepatology at Stanford Medicine in California.
Everyday life for them is a challenge.
Dealing with sick children is difficult. “But I think the difference between pediatrics and adults is despite how hard things get, children are the single most resilient people you’re ever going to meet,” she said.
Kids don’t always know they’re sick and they don’t act sick, even when they are. “Every day, I literally get on the floor, I get to play, I get to run around. And truly, I have fun every single day. I get excited to go to work. And I think that’s what makes work not feel like work,” said Dr. Zhang.
In an interview, she discussed the satisfaction of following patients throughout their care continuum and her research to reduce the likelihood of transplant rejection.
She also shared an inspirational story of one young patient who spent his life tied to an IV, and how a transplant exposed him to the normal joys of life, like swimming, going to camp and getting on a plane for the first time.
Q: Why did you choose this subspecialty of pediatric GI?
I think it’s the best subspecialty because I think it combines a lot of the things that I enjoy, which is long-term continuity of care. It’s about growing up with your patients and seeing them through all the various stages of their life, often meeting patients when they’re babies. I get pictures of high school graduations and life milestones and even see some of my patients have families of their own. Becoming a part of their family is very meaningful to me. I also like complexity and acuity, and gastroenterology and hepatology provide those things.
And then lastly, it’s great to be able to exercise procedural skills and constantly learn new procedural skills.
Q: How did you become interested in the field of pediatric intestinal and liver transplantation?
I did all my training here at Stanford. We have one of the largest pediatric transplant centers and we also have a very large intestinal rehabilitation population.
Coming through residency and fellowship, I had a lot of exposure to transplant and intestinal failure, intestinal rehabilitation. I really liked the longitudinal relationship I got to form with my patients. Sometimes they’re in the neonatal ICU, where you’re meeting them in their very first days of life. You follow them through their chronic illness, through transplant and after transplant for many years. You become not just their GI, but the center of their care.
Q: What challenges are unique to this type of transplant work?
Pediatric intestinal failure and intestinal transplant represents an incredibly small subset of children. Oftentimes, they do not get the resources and recognition on a national policy level or even at the hospital level that other gastrointestinal diseases receive. What’s difficult is they are such a small subset but their complexity and their needs are probably in the highest percentile. So that’s a really challenging combination to start with. And there’s only a few centers that specialize in doing intestinal rehabilitation and intestinal transplantation for children in the country.
Developing expertise has been slow. But I think in the last decade or so, our understanding and success with intestinal rehabilitation and intestinal transplantation has really improved, especially at large centers like Stanford. We’ve had a lot of success stories and have not had any graft loss since 2014.
Q: Are these transplants hard to acquire?
Yes, especially when you’re transplanting not just the intestines but the liver as well. You’re waiting for two organs, not just one organ. And on top of that, you’re waiting for an appropriately sized donor; usually a child who’s around the same size or same age who’s passed away. Those organs would have to be a good match. Children can wait multiple years for a transplant.
Q: Is there a success story you’d like to share?
One patient I met in the neonatal ICU had congenital short bowel syndrome. He was born with hardly any intestines. He developed complications of being on long-term intravenous nutrition, which included recurrent central line infections and liver disease. He was never able to eat because he really didn’t have a digestive system that could adequately absorb anything. He had a central line in one of his large veins, so he couldn’t go swimming.
He had to have special adaptive wear to even shower or bathe and couldn’t travel. It’s these types of patients that benefit so much from transplant. Putting any kid through transplant is a massive undertaking and it certainly has risks. But he underwent a successful transplant at the age of 8—not just an intestinal transplant, but a multi-visceral transplant of the liver, intestine, and pancreas. He’s 9 years old now, and no longer needs intravenous nutrition. He ate by mouth for the very first time after transplant. He’s trying all sorts of new foods and he was able to go to a special transplant camp for children. Getting on a plane to Los Angeles, which is where our transplant camp is, was a huge deal.
He was able to swim in the lake. He’s never been able to do that. And he wants to start doing sports this fall. This was really a life-changing story for him.
Q: What advancements lie ahead for this field of work? Have you work on any notable research?
I think our understanding of transplant immunology has really progressed, especially recently. That’s what part of my research is about—using novel therapies to modulate the immune system of pediatric transplant recipients. The No. 1 complication that occurs after intestinal transplant is rejection because obviously you’re implanting somebody else’s organs into a patient.
I am involved in a clinical trial that’s looking at the use of extracellular vesicles that are isolated from hematopoietic stem cells. These vesicles contain various growth factors, anti-inflammatory proteins and tissue repair factors that we are infusing into intestinal transplant patients with the aim to repair the intestinal tissue patients are rejecting.
Q: When you’re not being a GI, how do you spend your free weekend afternoons?
My husband and I have an almost 2-year-old little girl. She keeps us busy and I spend my afternoons chasing after a crazy toddler.
Lightning Round
Texting or talking?
Huge texter
Favorite junk food?
French fries
Cat or dog person?
Dog
Favorite ice cream?
Strawberry
If you weren’t a gastroenterologist, what would you be?Florist
Best place you’ve traveled to?
Thailand
Number of cups of coffee you drink per day?
Too many
Favorite city in the US besides the one you live in?
New York City
Favorite sport?
Tennis
Optimist or pessimist?
Optimist
The best part about working with kids is that “I get to laugh every day,” said Ke-You (Yoyo) Zhang, MD, clinical assistant professor for pediatrics–gastroenterology and hepatology at Stanford Medicine in California.
Everyday life for them is a challenge.
Dealing with sick children is difficult. “But I think the difference between pediatrics and adults is despite how hard things get, children are the single most resilient people you’re ever going to meet,” she said.
Kids don’t always know they’re sick and they don’t act sick, even when they are. “Every day, I literally get on the floor, I get to play, I get to run around. And truly, I have fun every single day. I get excited to go to work. And I think that’s what makes work not feel like work,” said Dr. Zhang.
In an interview, she discussed the satisfaction of following patients throughout their care continuum and her research to reduce the likelihood of transplant rejection.
She also shared an inspirational story of one young patient who spent his life tied to an IV, and how a transplant exposed him to the normal joys of life, like swimming, going to camp and getting on a plane for the first time.
Q: Why did you choose this subspecialty of pediatric GI?
I think it’s the best subspecialty because I think it combines a lot of the things that I enjoy, which is long-term continuity of care. It’s about growing up with your patients and seeing them through all the various stages of their life, often meeting patients when they’re babies. I get pictures of high school graduations and life milestones and even see some of my patients have families of their own. Becoming a part of their family is very meaningful to me. I also like complexity and acuity, and gastroenterology and hepatology provide those things.
And then lastly, it’s great to be able to exercise procedural skills and constantly learn new procedural skills.
Q: How did you become interested in the field of pediatric intestinal and liver transplantation?
I did all my training here at Stanford. We have one of the largest pediatric transplant centers and we also have a very large intestinal rehabilitation population.
Coming through residency and fellowship, I had a lot of exposure to transplant and intestinal failure, intestinal rehabilitation. I really liked the longitudinal relationship I got to form with my patients. Sometimes they’re in the neonatal ICU, where you’re meeting them in their very first days of life. You follow them through their chronic illness, through transplant and after transplant for many years. You become not just their GI, but the center of their care.
Q: What challenges are unique to this type of transplant work?
Pediatric intestinal failure and intestinal transplant represents an incredibly small subset of children. Oftentimes, they do not get the resources and recognition on a national policy level or even at the hospital level that other gastrointestinal diseases receive. What’s difficult is they are such a small subset but their complexity and their needs are probably in the highest percentile. So that’s a really challenging combination to start with. And there’s only a few centers that specialize in doing intestinal rehabilitation and intestinal transplantation for children in the country.
Developing expertise has been slow. But I think in the last decade or so, our understanding and success with intestinal rehabilitation and intestinal transplantation has really improved, especially at large centers like Stanford. We’ve had a lot of success stories and have not had any graft loss since 2014.
Q: Are these transplants hard to acquire?
Yes, especially when you’re transplanting not just the intestines but the liver as well. You’re waiting for two organs, not just one organ. And on top of that, you’re waiting for an appropriately sized donor; usually a child who’s around the same size or same age who’s passed away. Those organs would have to be a good match. Children can wait multiple years for a transplant.
Q: Is there a success story you’d like to share?
One patient I met in the neonatal ICU had congenital short bowel syndrome. He was born with hardly any intestines. He developed complications of being on long-term intravenous nutrition, which included recurrent central line infections and liver disease. He was never able to eat because he really didn’t have a digestive system that could adequately absorb anything. He had a central line in one of his large veins, so he couldn’t go swimming.
He had to have special adaptive wear to even shower or bathe and couldn’t travel. It’s these types of patients that benefit so much from transplant. Putting any kid through transplant is a massive undertaking and it certainly has risks. But he underwent a successful transplant at the age of 8—not just an intestinal transplant, but a multi-visceral transplant of the liver, intestine, and pancreas. He’s 9 years old now, and no longer needs intravenous nutrition. He ate by mouth for the very first time after transplant. He’s trying all sorts of new foods and he was able to go to a special transplant camp for children. Getting on a plane to Los Angeles, which is where our transplant camp is, was a huge deal.
He was able to swim in the lake. He’s never been able to do that. And he wants to start doing sports this fall. This was really a life-changing story for him.
Q: What advancements lie ahead for this field of work? Have you work on any notable research?
I think our understanding of transplant immunology has really progressed, especially recently. That’s what part of my research is about—using novel therapies to modulate the immune system of pediatric transplant recipients. The No. 1 complication that occurs after intestinal transplant is rejection because obviously you’re implanting somebody else’s organs into a patient.
I am involved in a clinical trial that’s looking at the use of extracellular vesicles that are isolated from hematopoietic stem cells. These vesicles contain various growth factors, anti-inflammatory proteins and tissue repair factors that we are infusing into intestinal transplant patients with the aim to repair the intestinal tissue patients are rejecting.
Q: When you’re not being a GI, how do you spend your free weekend afternoons?
My husband and I have an almost 2-year-old little girl. She keeps us busy and I spend my afternoons chasing after a crazy toddler.
Lightning Round
Texting or talking?
Huge texter
Favorite junk food?
French fries
Cat or dog person?
Dog
Favorite ice cream?
Strawberry
If you weren’t a gastroenterologist, what would you be?Florist
Best place you’ve traveled to?
Thailand
Number of cups of coffee you drink per day?
Too many
Favorite city in the US besides the one you live in?
New York City
Favorite sport?
Tennis
Optimist or pessimist?
Optimist
The best part about working with kids is that “I get to laugh every day,” said Ke-You (Yoyo) Zhang, MD, clinical assistant professor for pediatrics–gastroenterology and hepatology at Stanford Medicine in California.
Everyday life for them is a challenge.
Dealing with sick children is difficult. “But I think the difference between pediatrics and adults is despite how hard things get, children are the single most resilient people you’re ever going to meet,” she said.
Kids don’t always know they’re sick and they don’t act sick, even when they are. “Every day, I literally get on the floor, I get to play, I get to run around. And truly, I have fun every single day. I get excited to go to work. And I think that’s what makes work not feel like work,” said Dr. Zhang.
In an interview, she discussed the satisfaction of following patients throughout their care continuum and her research to reduce the likelihood of transplant rejection.
She also shared an inspirational story of one young patient who spent his life tied to an IV, and how a transplant exposed him to the normal joys of life, like swimming, going to camp and getting on a plane for the first time.
Q: Why did you choose this subspecialty of pediatric GI?
I think it’s the best subspecialty because I think it combines a lot of the things that I enjoy, which is long-term continuity of care. It’s about growing up with your patients and seeing them through all the various stages of their life, often meeting patients when they’re babies. I get pictures of high school graduations and life milestones and even see some of my patients have families of their own. Becoming a part of their family is very meaningful to me. I also like complexity and acuity, and gastroenterology and hepatology provide those things.
And then lastly, it’s great to be able to exercise procedural skills and constantly learn new procedural skills.
Q: How did you become interested in the field of pediatric intestinal and liver transplantation?
I did all my training here at Stanford. We have one of the largest pediatric transplant centers and we also have a very large intestinal rehabilitation population.
Coming through residency and fellowship, I had a lot of exposure to transplant and intestinal failure, intestinal rehabilitation. I really liked the longitudinal relationship I got to form with my patients. Sometimes they’re in the neonatal ICU, where you’re meeting them in their very first days of life. You follow them through their chronic illness, through transplant and after transplant for many years. You become not just their GI, but the center of their care.
Q: What challenges are unique to this type of transplant work?
Pediatric intestinal failure and intestinal transplant represents an incredibly small subset of children. Oftentimes, they do not get the resources and recognition on a national policy level or even at the hospital level that other gastrointestinal diseases receive. What’s difficult is they are such a small subset but their complexity and their needs are probably in the highest percentile. So that’s a really challenging combination to start with. And there’s only a few centers that specialize in doing intestinal rehabilitation and intestinal transplantation for children in the country.
Developing expertise has been slow. But I think in the last decade or so, our understanding and success with intestinal rehabilitation and intestinal transplantation has really improved, especially at large centers like Stanford. We’ve had a lot of success stories and have not had any graft loss since 2014.
Q: Are these transplants hard to acquire?
Yes, especially when you’re transplanting not just the intestines but the liver as well. You’re waiting for two organs, not just one organ. And on top of that, you’re waiting for an appropriately sized donor; usually a child who’s around the same size or same age who’s passed away. Those organs would have to be a good match. Children can wait multiple years for a transplant.
Q: Is there a success story you’d like to share?
One patient I met in the neonatal ICU had congenital short bowel syndrome. He was born with hardly any intestines. He developed complications of being on long-term intravenous nutrition, which included recurrent central line infections and liver disease. He was never able to eat because he really didn’t have a digestive system that could adequately absorb anything. He had a central line in one of his large veins, so he couldn’t go swimming.
He had to have special adaptive wear to even shower or bathe and couldn’t travel. It’s these types of patients that benefit so much from transplant. Putting any kid through transplant is a massive undertaking and it certainly has risks. But he underwent a successful transplant at the age of 8—not just an intestinal transplant, but a multi-visceral transplant of the liver, intestine, and pancreas. He’s 9 years old now, and no longer needs intravenous nutrition. He ate by mouth for the very first time after transplant. He’s trying all sorts of new foods and he was able to go to a special transplant camp for children. Getting on a plane to Los Angeles, which is where our transplant camp is, was a huge deal.
He was able to swim in the lake. He’s never been able to do that. And he wants to start doing sports this fall. This was really a life-changing story for him.
Q: What advancements lie ahead for this field of work? Have you work on any notable research?
I think our understanding of transplant immunology has really progressed, especially recently. That’s what part of my research is about—using novel therapies to modulate the immune system of pediatric transplant recipients. The No. 1 complication that occurs after intestinal transplant is rejection because obviously you’re implanting somebody else’s organs into a patient.
I am involved in a clinical trial that’s looking at the use of extracellular vesicles that are isolated from hematopoietic stem cells. These vesicles contain various growth factors, anti-inflammatory proteins and tissue repair factors that we are infusing into intestinal transplant patients with the aim to repair the intestinal tissue patients are rejecting.
Q: When you’re not being a GI, how do you spend your free weekend afternoons?
My husband and I have an almost 2-year-old little girl. She keeps us busy and I spend my afternoons chasing after a crazy toddler.
Lightning Round
Texting or talking?
Huge texter
Favorite junk food?
French fries
Cat or dog person?
Dog
Favorite ice cream?
Strawberry
If you weren’t a gastroenterologist, what would you be?Florist
Best place you’ve traveled to?
Thailand
Number of cups of coffee you drink per day?
Too many
Favorite city in the US besides the one you live in?
New York City
Favorite sport?
Tennis
Optimist or pessimist?
Optimist

Patient Navigators for Serious Illnesses Can Now Bill Under New Medicare Codes
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.
In a move that acknowledges the gauntlet the US health system poses for people facing serious and fatal illnesses, Medicare will pay for a new class of workers to help patients manage treatments for conditions like cancer and heart failure.
The 2024 Medicare physician fee schedule includes new billing codes, including G0023, to pay for 60 minutes a month of care coordination by certified or trained auxiliary personnel working under the direction of a clinician.
A diagnosis of cancer or another serious illness takes a toll beyond the physical effects of the disease. Patients often scramble to make adjustments in family and work schedules to manage treatment, said Samyukta Mullangi, MD, MBA, medical director of oncology at Thyme Care, a Nashville, Tennessee–based firm that provides navigation and coordination services to oncology practices and insurers.
“It just really does create a bit of a pressure cooker for patients,” Dr. Mullangi told this news organization.
Medicare has for many years paid for medical professionals to help patients cope with the complexities of disease, such as chronic care management (CCM) provided by physicians, nurses, and physician assistants.
The new principal illness navigation (PIN) payments are intended to pay for work that to date typically has been done by people without medical degrees, including those involved in peer support networks and community health programs. The US Centers for Medicare and Medicaid Services(CMS) expects these navigators will undergo training and work under the supervision of clinicians.
The new navigators may coordinate care transitions between medical settings, follow up with patients after emergency department (ED) visits, or communicate with skilled nursing facilities regarding the psychosocial needs and functional deficits of a patient, among other functions.
CMS expects the new navigators may:
- Conduct assessments to understand a patient’s life story, strengths, needs, goals, preferences, and desired outcomes, including understanding cultural and linguistic factors.
- Provide support to accomplish the clinician’s treatment plan.
- Coordinate the receipt of needed services from healthcare facilities, home- and community-based service providers, and caregivers.
Peers as Navigators
The new navigators can be former patients who have undergone similar treatments for serious diseases, CMS said. This approach sets the new program apart from other care management services Medicare already covers, program officials wrote in the 2024 physician fee schedule.
“For some conditions, patients are best able to engage with the healthcare system and access care if they have assistance from a single, dedicated individual who has ‘lived experience,’ ” according to the rule.
The agency has taken a broad initial approach in defining what kinds of illnesses a patient may have to qualify for services. Patients must have a serious condition that is expected to last at least 3 months, such as cancer, heart failure, or substance use disorder.
But those without a definitive diagnosis may also qualify to receive navigator services.
In the rule, CMS cited a case in which a CT scan identified a suspicious mass in a patient’s colon. A clinician might decide this person would benefit from navigation services due to the potential risks for an undiagnosed illness.
“Regardless of the definitive diagnosis of the mass, presence of a colonic mass for that patient may be a serious high-risk condition that could, for example, cause obstruction and lead the patient to present to the emergency department, as well as be potentially indicative of an underlying life-threatening illness such as colon cancer,” CMS wrote in the rule.
Navigators often start their work when cancer patients are screened and guide them through initial diagnosis, potential surgery, radiation, or chemotherapy, said Sharon Gentry, MSN, RN, a former nurse navigator who is now the editor in chief of the Journal of the Academy of Oncology Nurse & Patient Navigators.
The navigators are meant to be a trusted and continual presence for patients, who otherwise might be left to start anew in finding help at each phase of care.
The navigators “see the whole picture. They see the whole journey the patient takes, from pre-diagnosis all the way through diagnosis care out through survival,” Ms. Gentry said.
Gaining a special Medicare payment for these kinds of services will elevate this work, she said.
Many newer drugs can target specific mechanisms and proteins of cancer. Often, oncology treatment involves testing to find out if mutations are allowing the cancer cells to evade a patient’s immune system.
Checking these biomarkers takes time, however. Patients sometimes become frustrated because they are anxious to begin treatment. Patients may receive inaccurate information from friends or family who went through treatment previously. Navigators can provide knowledge on the current state of care for a patient’s disease, helping them better manage anxieties.
“You have to explain to them that things have changed since the guy you drink coffee with was diagnosed with cancer, and there may be a drug that could target that,” Ms. Gentry said.
Potential Challenges
Initial uptake of the new PIN codes may be slow going, however, as clinicians and health systems may already use well-established codes. These include CCM and principal care management services, which may pay higher rates, Mullangi said.
“There might be sensitivity around not wanting to cannibalize existing programs with a new program,” Dr. Mullangi said.
In addition, many patients will have a copay for the services of principal illness navigators, Dr. Mullangi said.
While many patients have additional insurance that would cover the service, not all do. People with traditional Medicare coverage can sometimes pay 20% of the cost of some medical services.
“I think that may give patients pause, particularly if they’re already feeling the financial burden of a cancer treatment journey,” Dr. Mullangi said.
Pay rates for PIN services involve calculations of regional price differences, which are posted publicly by CMS, and potential added fees for services provided by hospital-affiliated organizations.
Consider payments for code G0023, covering 60 minutes of principal navigation services provided in a single month.
A set reimbursement for patients cared for in independent medical practices exists, with variation for local costs. Medicare’s non-facility price for G0023 would be $102.41 in some parts of Silicon Valley in California, including San Jose. In Arkansas, where costs are lower, reimbursement would be $73.14 for this same service.
Patients who get services covered by code G0023 in independent medical practices would have monthly copays of about $15-$20, depending on where they live.
The tab for patients tends to be higher for these same services if delivered through a medical practice owned by a hospital, as this would trigger the addition of facility fees to the payments made to cover the services. Facility fees are difficult for the public to ascertain before getting a treatment or service.
Dr. Mullangi and Ms. Gentry reported no relevant financial disclosures outside of their employers.
A version of this article first appeared on Medscape.com.

Ready for post-acute care?
The definition of “hospitalist,” according to the SHM website, is a clinician “dedicated to delivering comprehensive medical care to hospitalized patients.” For years, the hospital setting was the specialties’ identifier. But as hospitalists’ scope has expanded, and post-acute care (PAC) in the United States has grown, more hospitalists are extending their roles into this space.
PAC today is more than the traditional nursing home, according to Manoj K. Mathew, MD, SFHM, national medical director of Agilon Health in Los Angeles.
Many of those expanded settings Dr. Mathew describes emerged as a result of the Affordable Care Act. Since its enactment in 2010, the ACA has heightened providers’ focus on the “Triple Aim” of improving the patient experience (including quality and satisfaction), improving the health of populations, and reducing the per capita cost of healthcare.1 Vishal Kuchaculla, MD, New England regional post-acute medical director of Knoxville,Tenn.-based TeamHealth, says new service lines also developed as Medicare clamped down on long-term inpatient hospital stays by giving financial impetus to discharge patients as soon as possible.
“Over the last few years, there’s been a major shift from fee-for-service to risk-based payment models,” Dr. Kuchaculla says. “The government’s financial incentives are driving outcomes to improve performance initiatives.”
“Today, LTACHs can be used as substitutes for short-term acute care,” says Sean R. Muldoon, MD, MPH, FCCP, chief medical officer of Kindred Healthcare in Louisville, Ky., and former chair of SHM’s Post-Acute Care Committee. “This means that a patient can be directly admitted from their home to an LTACH. In fact, many hospice and home-care patients are referred from physicians’ offices without a preceding hospitalization.”
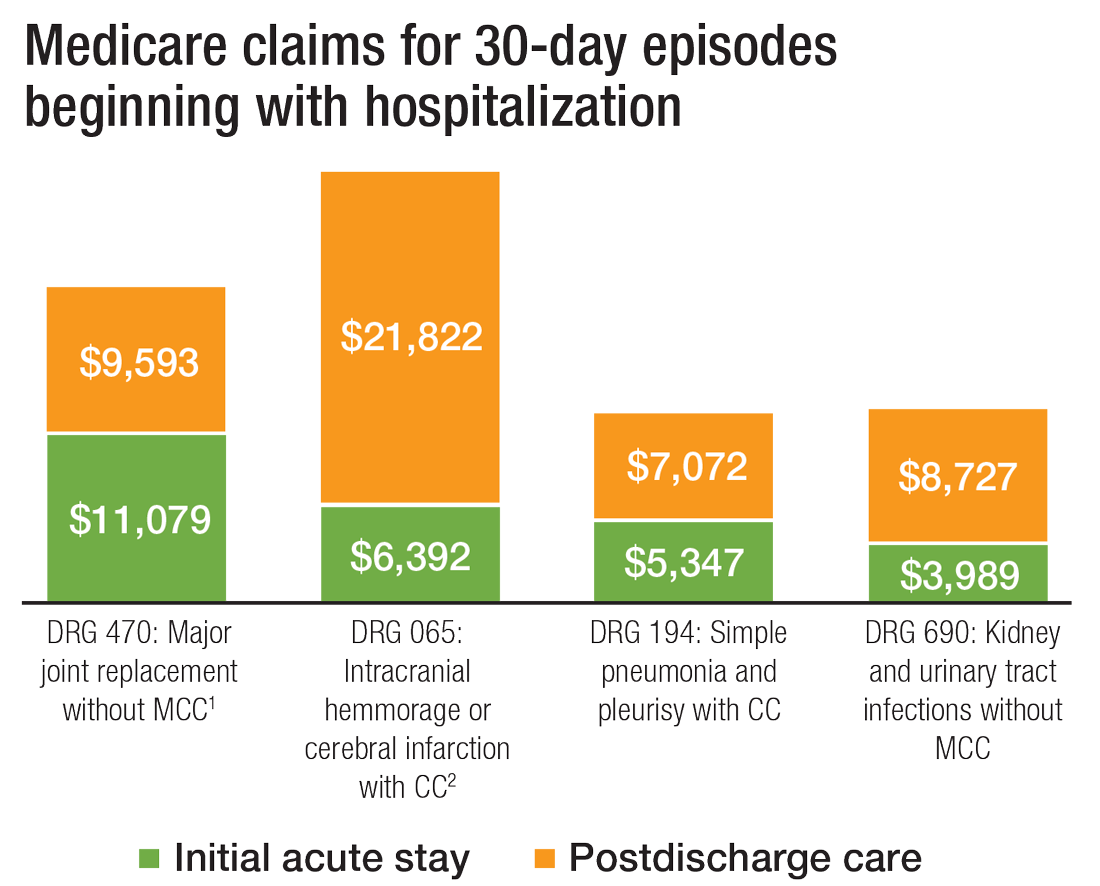
Hospitalists can fill a need

More hospitalists are working in PACs for a number of reasons. Dr. Mathew says PAC facilities and services have “typically lacked the clinical structure and processes to obtain the results that patients and payors expect.
“These deficits needed to be quickly remedied as patients discharged from hospitals have increased acuity and higher disease burdens,” he adds. “Hospitalists were the natural choice to fill roles requiring their expertise and experience.”
Dr. Muldoon considers the expanded scope of practice into PACs an additional layer to hospital medicine’s value proposition to the healthcare system.
“As experts in the management of inpatient populations, it’s natural for hospitalists to expand to other facilities with inpatient-like populations,” he says, noting SNFs are the most popular choice, with IRFs and LTACHs also being common places to work. Few hospitalists work in home care or hospice.
PAC settings are designed to help patients who are transitioning from an inpatient setting back to their home or other setting.
“Many patients go home after a SNF stay, while others will move to a nursing home or other longer-term care setting for the first time,” says Tiffany Radcliff, PhD, a health economist in the department of health policy and management at Texas A&M University School of Public Health in College Station. “With this in mind, hospitalists working in PAC have the opportunity to address each patient’s ongoing care needs and prepare them for their next setting. Hospitalists can manage medication or other care regimen changes that resulted from an inpatient stay, reinforce discharge instructions to the patient and their caregivers, and identify any other issues with continuing care that need to be addressed before discharge to the next care setting.”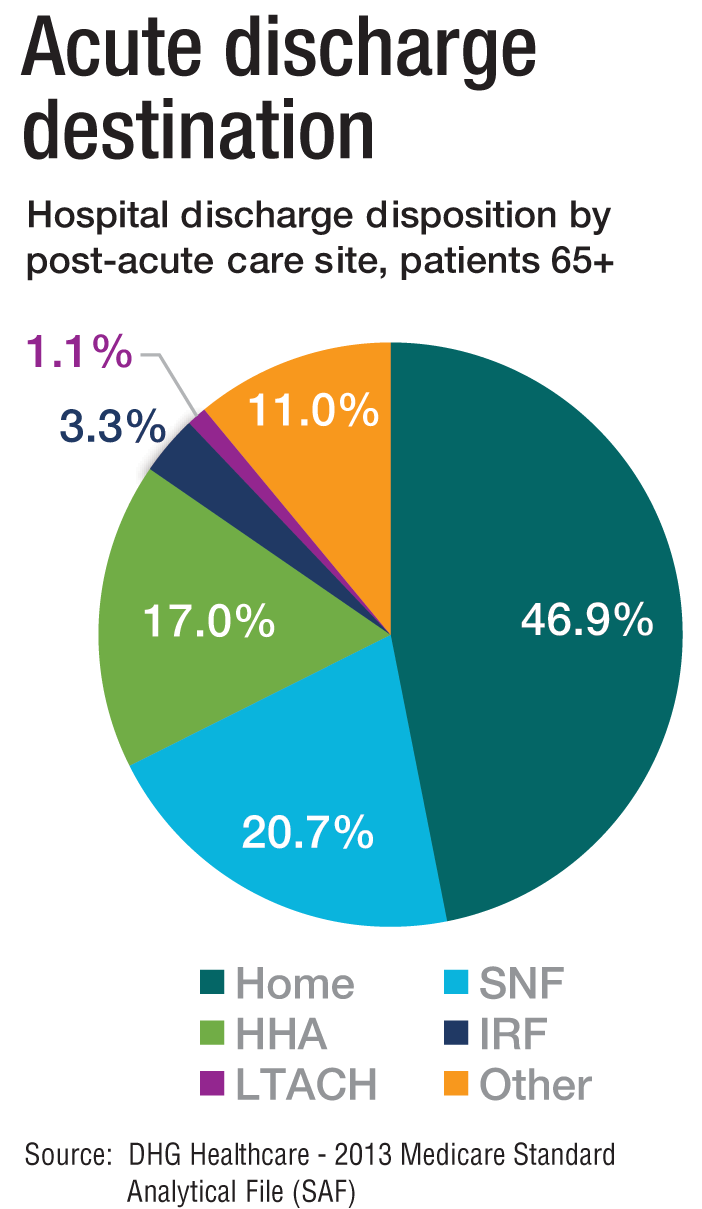
Transitioning Care
Even if a hospitalist is not employed at a PAC, it’s important that they know something about them.
“As patients are moved downstream earlier, hospitalists are being asked to help make a judgment regarding when and where an inpatient is transitioned,” Dr. Muldoon says. As organizations move toward becoming fully risk capable, it is necessary to develop referral networks of high-quality PAC providers to achieve the best clinical outcomes, reduce readmissions, and lower costs.2“Therefore, hospitalists should have a working knowledge of the different sites of service as well as some opinion on the suitability of available options in their community,” Dr. Muldoon says. “The hospitalist can also help to educate the hospitalized patient on what to expect at a PAC.”
If a patient is inappropriately prepared for the PAC setting, it could lead to incomplete management of their condition, which ultimately could lead to readmission.
“When hospitalists know how care is provided in a PAC setting, they are better able to ensure a smoother transition of care between settings,” says Tochi Iroku-Malize, MD, MPH, MBA, FAAFP, SFHM, chair of family medicine at Northwell Health in Long Island, N.Y. “This will ultimately prevent unnecessary readmissions.”
Further, the quality metrics that hospitals and thereby hospitalists are judged by no longer end at the hospital’s exit.
“The ownership of acute-care outcomes requires extending the accountability to outside of the institution’s four walls,” Dr. Mathew says. “The inpatient team needs to place great importance on the transition of care and the subsequent quality of that care when the patient is discharged.”
Robert W. Harrington Jr., MD, SFHM, chief medical officer of Plano, Texas–based Reliant Post-Acute Care Solutions and former SHM president, says the health system landscapes are pushing HM beyond the hospitals’ walls.
How PAC settings differ from hospitals
Practicing in PAC has some important nuances that hospitalists from short-term acute care need to get accustomed to, Dr. Muldoon says. Primarily, the diagnostic capabilities are much more limited, as is the presence of high-level staffing. Further, patients are less resilient to medication changes and interventions, so changes need to be done gradually.
“Hospitalists who try to practice acute-care medicine in a PAC setting may become frustrated by the length of time it takes to do a work-up, get a consultation, and respond to a patient’s change of condition,” Dr. Muldoon says. “Nonetheless, hospitalists can overcome this once recognizing this mind shift.”
According to Dr. Harrington, another challenge hospitalists may face is the inability of the hospital’s and PAC facility’s IT platforms to exchange electronic information.
“The major vendors on both sides need to figure out an interoperability strategy,” he says. “Currently, it often takes 1-3 days to receive a new patient’s discharge summary. The summary may consist of a stack of paper that takes significant time to sort through and requires the PAC facility to perform duplicate data entry. It’s a very highly inefficient process that opens up the doors to mistakes and errors of omission and commission that can result in bad patient outcomes.”
Arif Nazir, MD, CMD, FACP, AGSF, chief medical officer of Signature HealthCARE and president of SHC Medical Partners, both in Louisville, Ky., cites additional reasons the lack of seamless communication between a hospital and PAC facility is problematic. “I see physicians order laboratory tests and investigations that were already done in the hospital because they didn’t know they were already performed or never received the results,” he says. “Similarly, I see patients continue to take medications prescribed in the hospital long term even though they were only supposed to take them short term. I’ve also seen patients come to a PAC setting from a hospital without any formal understanding of their rehabilitative period and expectations for recovery.”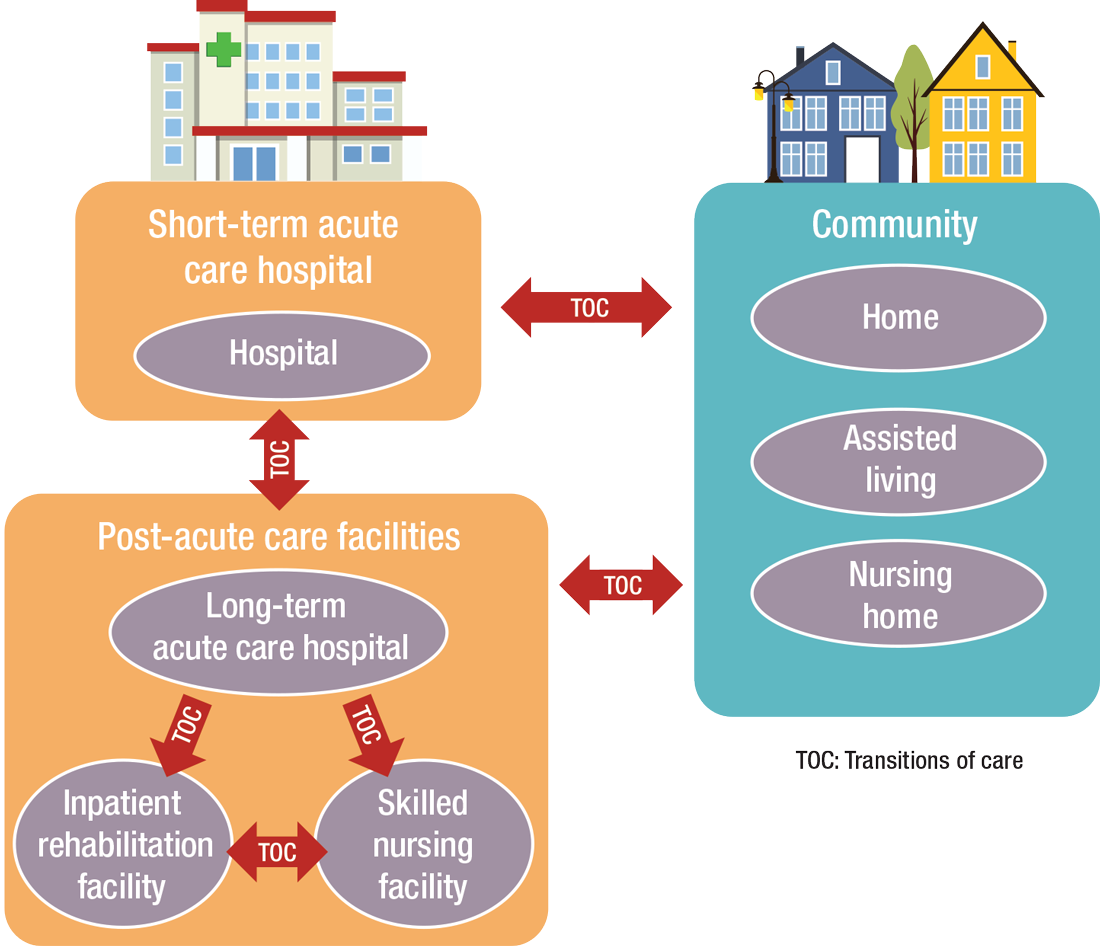
What’s ahead?
Looking to the future, Surafel Tsega, MD, clinical instructor at Mount Sinai Hospital in New York, says he thinks there will be a move toward greater collaboration among inpatient and PAC facilities, particularly in the discharge process, given that hospitals have an added incentive to ensure safe transitions because reimbursement from the Centers for Medicare & Medicaid Services is tied to readmissions and there are penalties for readmission. This involves more comprehensive planning regarding “warm handoffs” (e.g., real-time discussions with PAC providers about a patient’s hospital course and plan of care upon discharge), transferring of information, and so forth.
And while it can still be challenging to identify high-risk patients or determine the intensity and duration of their care, Dr. Mathew says risk-stratification tools and care pathways are continually being refined to maximize value with the limited resources available. In addition, with an increased emphasis on employing a team approach to care, there will be better integration of non-medical services to address the social determinants of health, which play significant roles in overall health and healing.
“Working with community-based organizations for this purpose will be a valuable tool for any of the population health–based initiatives,” he says.
Dr. Muldoon says he believes healthcare reform will increasingly view an inpatient admission as something to be avoided.
“If hospitalization can’t be avoided, then it should be shortened as much as possible,” he says. “This will shift inpatient care into LTACHs, SNFs, and IRFs. Hospitalists would be wise to follow patients into those settings as traditional inpatient census is reduced. This will take a few years, so hospitalists should start now in preparing for that downstream transition of individuals who were previously inpatients.”
The cost of care, and other PAC facts and figures
The amount of money that Medicare spends on post-acute care (PAC) has been increasing. In 2012, 12.6% of Medicare beneficiaries used some form of PAC, costing $62 billion.2 That amounts to the Centers for Medicare & Medicaid Services spending close to 25% of Medicare beneficiary expenses on PAC, a 133% increase from 2001 to 2012. Among the different types, $30.4 billion was spent on skilled nursing facilities (SNFs), $18.6 billion on home health, and $13.1 billion on long-term acute care (LTAC) and acute-care rehabilitation.2
It’s also been reported that after short-term acute-care hospitalization, about one in five Medicare beneficiaries requires continued specialized treatment in one of the three typical Medicare PAC settings: inpatient rehabilitation facilities (IRFs), LTAC hospitals, and SNFs.3
What’s more, hospital readmission nearly doubles the cost of an episode, so the financial implications for organizations operating in risk-bearing arrangements are significant. In 2013, 2,213 hospitals were charged $280 million in readmission penalties.2
References
1. The role of post-acute care in new care delivery models. American Hospital Association website. Available at: http://www.aha.org/research/reports/tw/15dec-tw-postacute.pdf. Accessed Nov. 7, 2016.
2. Post-acute care integration: Today and in the future. DHG Healthcare website. Available at: http://www2.dhgllp.com/res_pubs/HCG-Post-Acute-Care-Integration.pdf. Accessed Nov. 7, 2016.
3. Overview: Post-acute care transitions toolkit. Society for Hospital Medicine website. Available at: http://www.hospitalmedicine.org/Web/Quality___Innovation/Implementation_Toolkit/pact/Overview_PACT.aspx?hkey=dea3da3c-8620-46db-a00f-89f07f021958. Accessed Nov. 10, 2016.
The definition of “hospitalist,” according to the SHM website, is a clinician “dedicated to delivering comprehensive medical care to hospitalized patients.” For years, the hospital setting was the specialties’ identifier. But as hospitalists’ scope has expanded, and post-acute care (PAC) in the United States has grown, more hospitalists are extending their roles into this space.
PAC today is more than the traditional nursing home, according to Manoj K. Mathew, MD, SFHM, national medical director of Agilon Health in Los Angeles.
Many of those expanded settings Dr. Mathew describes emerged as a result of the Affordable Care Act. Since its enactment in 2010, the ACA has heightened providers’ focus on the “Triple Aim” of improving the patient experience (including quality and satisfaction), improving the health of populations, and reducing the per capita cost of healthcare.1 Vishal Kuchaculla, MD, New England regional post-acute medical director of Knoxville,Tenn.-based TeamHealth, says new service lines also developed as Medicare clamped down on long-term inpatient hospital stays by giving financial impetus to discharge patients as soon as possible.
“Over the last few years, there’s been a major shift from fee-for-service to risk-based payment models,” Dr. Kuchaculla says. “The government’s financial incentives are driving outcomes to improve performance initiatives.”
“Today, LTACHs can be used as substitutes for short-term acute care,” says Sean R. Muldoon, MD, MPH, FCCP, chief medical officer of Kindred Healthcare in Louisville, Ky., and former chair of SHM’s Post-Acute Care Committee. “This means that a patient can be directly admitted from their home to an LTACH. In fact, many hospice and home-care patients are referred from physicians’ offices without a preceding hospitalization.”

Hospitalists can fill a need

More hospitalists are working in PACs for a number of reasons. Dr. Mathew says PAC facilities and services have “typically lacked the clinical structure and processes to obtain the results that patients and payors expect.
“These deficits needed to be quickly remedied as patients discharged from hospitals have increased acuity and higher disease burdens,” he adds. “Hospitalists were the natural choice to fill roles requiring their expertise and experience.”
Dr. Muldoon considers the expanded scope of practice into PACs an additional layer to hospital medicine’s value proposition to the healthcare system.
“As experts in the management of inpatient populations, it’s natural for hospitalists to expand to other facilities with inpatient-like populations,” he says, noting SNFs are the most popular choice, with IRFs and LTACHs also being common places to work. Few hospitalists work in home care or hospice.
PAC settings are designed to help patients who are transitioning from an inpatient setting back to their home or other setting.
“Many patients go home after a SNF stay, while others will move to a nursing home or other longer-term care setting for the first time,” says Tiffany Radcliff, PhD, a health economist in the department of health policy and management at Texas A&M University School of Public Health in College Station. “With this in mind, hospitalists working in PAC have the opportunity to address each patient’s ongoing care needs and prepare them for their next setting. Hospitalists can manage medication or other care regimen changes that resulted from an inpatient stay, reinforce discharge instructions to the patient and their caregivers, and identify any other issues with continuing care that need to be addressed before discharge to the next care setting.”
Transitioning Care
Even if a hospitalist is not employed at a PAC, it’s important that they know something about them.
“As patients are moved downstream earlier, hospitalists are being asked to help make a judgment regarding when and where an inpatient is transitioned,” Dr. Muldoon says. As organizations move toward becoming fully risk capable, it is necessary to develop referral networks of high-quality PAC providers to achieve the best clinical outcomes, reduce readmissions, and lower costs.2“Therefore, hospitalists should have a working knowledge of the different sites of service as well as some opinion on the suitability of available options in their community,” Dr. Muldoon says. “The hospitalist can also help to educate the hospitalized patient on what to expect at a PAC.”
If a patient is inappropriately prepared for the PAC setting, it could lead to incomplete management of their condition, which ultimately could lead to readmission.
“When hospitalists know how care is provided in a PAC setting, they are better able to ensure a smoother transition of care between settings,” says Tochi Iroku-Malize, MD, MPH, MBA, FAAFP, SFHM, chair of family medicine at Northwell Health in Long Island, N.Y. “This will ultimately prevent unnecessary readmissions.”
Further, the quality metrics that hospitals and thereby hospitalists are judged by no longer end at the hospital’s exit.
“The ownership of acute-care outcomes requires extending the accountability to outside of the institution’s four walls,” Dr. Mathew says. “The inpatient team needs to place great importance on the transition of care and the subsequent quality of that care when the patient is discharged.”
Robert W. Harrington Jr., MD, SFHM, chief medical officer of Plano, Texas–based Reliant Post-Acute Care Solutions and former SHM president, says the health system landscapes are pushing HM beyond the hospitals’ walls.
How PAC settings differ from hospitals
Practicing in PAC has some important nuances that hospitalists from short-term acute care need to get accustomed to, Dr. Muldoon says. Primarily, the diagnostic capabilities are much more limited, as is the presence of high-level staffing. Further, patients are less resilient to medication changes and interventions, so changes need to be done gradually.
“Hospitalists who try to practice acute-care medicine in a PAC setting may become frustrated by the length of time it takes to do a work-up, get a consultation, and respond to a patient’s change of condition,” Dr. Muldoon says. “Nonetheless, hospitalists can overcome this once recognizing this mind shift.”
According to Dr. Harrington, another challenge hospitalists may face is the inability of the hospital’s and PAC facility’s IT platforms to exchange electronic information.
“The major vendors on both sides need to figure out an interoperability strategy,” he says. “Currently, it often takes 1-3 days to receive a new patient’s discharge summary. The summary may consist of a stack of paper that takes significant time to sort through and requires the PAC facility to perform duplicate data entry. It’s a very highly inefficient process that opens up the doors to mistakes and errors of omission and commission that can result in bad patient outcomes.”
Arif Nazir, MD, CMD, FACP, AGSF, chief medical officer of Signature HealthCARE and president of SHC Medical Partners, both in Louisville, Ky., cites additional reasons the lack of seamless communication between a hospital and PAC facility is problematic. “I see physicians order laboratory tests and investigations that were already done in the hospital because they didn’t know they were already performed or never received the results,” he says. “Similarly, I see patients continue to take medications prescribed in the hospital long term even though they were only supposed to take them short term. I’ve also seen patients come to a PAC setting from a hospital without any formal understanding of their rehabilitative period and expectations for recovery.”
What’s ahead?
Looking to the future, Surafel Tsega, MD, clinical instructor at Mount Sinai Hospital in New York, says he thinks there will be a move toward greater collaboration among inpatient and PAC facilities, particularly in the discharge process, given that hospitals have an added incentive to ensure safe transitions because reimbursement from the Centers for Medicare & Medicaid Services is tied to readmissions and there are penalties for readmission. This involves more comprehensive planning regarding “warm handoffs” (e.g., real-time discussions with PAC providers about a patient’s hospital course and plan of care upon discharge), transferring of information, and so forth.
And while it can still be challenging to identify high-risk patients or determine the intensity and duration of their care, Dr. Mathew says risk-stratification tools and care pathways are continually being refined to maximize value with the limited resources available. In addition, with an increased emphasis on employing a team approach to care, there will be better integration of non-medical services to address the social determinants of health, which play significant roles in overall health and healing.
“Working with community-based organizations for this purpose will be a valuable tool for any of the population health–based initiatives,” he says.
Dr. Muldoon says he believes healthcare reform will increasingly view an inpatient admission as something to be avoided.
“If hospitalization can’t be avoided, then it should be shortened as much as possible,” he says. “This will shift inpatient care into LTACHs, SNFs, and IRFs. Hospitalists would be wise to follow patients into those settings as traditional inpatient census is reduced. This will take a few years, so hospitalists should start now in preparing for that downstream transition of individuals who were previously inpatients.”
The cost of care, and other PAC facts and figures
The amount of money that Medicare spends on post-acute care (PAC) has been increasing. In 2012, 12.6% of Medicare beneficiaries used some form of PAC, costing $62 billion.2 That amounts to the Centers for Medicare & Medicaid Services spending close to 25% of Medicare beneficiary expenses on PAC, a 133% increase from 2001 to 2012. Among the different types, $30.4 billion was spent on skilled nursing facilities (SNFs), $18.6 billion on home health, and $13.1 billion on long-term acute care (LTAC) and acute-care rehabilitation.2
It’s also been reported that after short-term acute-care hospitalization, about one in five Medicare beneficiaries requires continued specialized treatment in one of the three typical Medicare PAC settings: inpatient rehabilitation facilities (IRFs), LTAC hospitals, and SNFs.3
What’s more, hospital readmission nearly doubles the cost of an episode, so the financial implications for organizations operating in risk-bearing arrangements are significant. In 2013, 2,213 hospitals were charged $280 million in readmission penalties.2
References
1. The role of post-acute care in new care delivery models. American Hospital Association website. Available at: http://www.aha.org/research/reports/tw/15dec-tw-postacute.pdf. Accessed Nov. 7, 2016.
2. Post-acute care integration: Today and in the future. DHG Healthcare website. Available at: http://www2.dhgllp.com/res_pubs/HCG-Post-Acute-Care-Integration.pdf. Accessed Nov. 7, 2016.
3. Overview: Post-acute care transitions toolkit. Society for Hospital Medicine website. Available at: http://www.hospitalmedicine.org/Web/Quality___Innovation/Implementation_Toolkit/pact/Overview_PACT.aspx?hkey=dea3da3c-8620-46db-a00f-89f07f021958. Accessed Nov. 10, 2016.
The definition of “hospitalist,” according to the SHM website, is a clinician “dedicated to delivering comprehensive medical care to hospitalized patients.” For years, the hospital setting was the specialties’ identifier. But as hospitalists’ scope has expanded, and post-acute care (PAC) in the United States has grown, more hospitalists are extending their roles into this space.
PAC today is more than the traditional nursing home, according to Manoj K. Mathew, MD, SFHM, national medical director of Agilon Health in Los Angeles.
Many of those expanded settings Dr. Mathew describes emerged as a result of the Affordable Care Act. Since its enactment in 2010, the ACA has heightened providers’ focus on the “Triple Aim” of improving the patient experience (including quality and satisfaction), improving the health of populations, and reducing the per capita cost of healthcare.1 Vishal Kuchaculla, MD, New England regional post-acute medical director of Knoxville,Tenn.-based TeamHealth, says new service lines also developed as Medicare clamped down on long-term inpatient hospital stays by giving financial impetus to discharge patients as soon as possible.
“Over the last few years, there’s been a major shift from fee-for-service to risk-based payment models,” Dr. Kuchaculla says. “The government’s financial incentives are driving outcomes to improve performance initiatives.”
“Today, LTACHs can be used as substitutes for short-term acute care,” says Sean R. Muldoon, MD, MPH, FCCP, chief medical officer of Kindred Healthcare in Louisville, Ky., and former chair of SHM’s Post-Acute Care Committee. “This means that a patient can be directly admitted from their home to an LTACH. In fact, many hospice and home-care patients are referred from physicians’ offices without a preceding hospitalization.”

Hospitalists can fill a need

More hospitalists are working in PACs for a number of reasons. Dr. Mathew says PAC facilities and services have “typically lacked the clinical structure and processes to obtain the results that patients and payors expect.
“These deficits needed to be quickly remedied as patients discharged from hospitals have increased acuity and higher disease burdens,” he adds. “Hospitalists were the natural choice to fill roles requiring their expertise and experience.”
Dr. Muldoon considers the expanded scope of practice into PACs an additional layer to hospital medicine’s value proposition to the healthcare system.
“As experts in the management of inpatient populations, it’s natural for hospitalists to expand to other facilities with inpatient-like populations,” he says, noting SNFs are the most popular choice, with IRFs and LTACHs also being common places to work. Few hospitalists work in home care or hospice.
PAC settings are designed to help patients who are transitioning from an inpatient setting back to their home or other setting.
“Many patients go home after a SNF stay, while others will move to a nursing home or other longer-term care setting for the first time,” says Tiffany Radcliff, PhD, a health economist in the department of health policy and management at Texas A&M University School of Public Health in College Station. “With this in mind, hospitalists working in PAC have the opportunity to address each patient’s ongoing care needs and prepare them for their next setting. Hospitalists can manage medication or other care regimen changes that resulted from an inpatient stay, reinforce discharge instructions to the patient and their caregivers, and identify any other issues with continuing care that need to be addressed before discharge to the next care setting.”
Transitioning Care
Even if a hospitalist is not employed at a PAC, it’s important that they know something about them.
“As patients are moved downstream earlier, hospitalists are being asked to help make a judgment regarding when and where an inpatient is transitioned,” Dr. Muldoon says. As organizations move toward becoming fully risk capable, it is necessary to develop referral networks of high-quality PAC providers to achieve the best clinical outcomes, reduce readmissions, and lower costs.2“Therefore, hospitalists should have a working knowledge of the different sites of service as well as some opinion on the suitability of available options in their community,” Dr. Muldoon says. “The hospitalist can also help to educate the hospitalized patient on what to expect at a PAC.”
If a patient is inappropriately prepared for the PAC setting, it could lead to incomplete management of their condition, which ultimately could lead to readmission.
“When hospitalists know how care is provided in a PAC setting, they are better able to ensure a smoother transition of care between settings,” says Tochi Iroku-Malize, MD, MPH, MBA, FAAFP, SFHM, chair of family medicine at Northwell Health in Long Island, N.Y. “This will ultimately prevent unnecessary readmissions.”
Further, the quality metrics that hospitals and thereby hospitalists are judged by no longer end at the hospital’s exit.
“The ownership of acute-care outcomes requires extending the accountability to outside of the institution’s four walls,” Dr. Mathew says. “The inpatient team needs to place great importance on the transition of care and the subsequent quality of that care when the patient is discharged.”
Robert W. Harrington Jr., MD, SFHM, chief medical officer of Plano, Texas–based Reliant Post-Acute Care Solutions and former SHM president, says the health system landscapes are pushing HM beyond the hospitals’ walls.
How PAC settings differ from hospitals
Practicing in PAC has some important nuances that hospitalists from short-term acute care need to get accustomed to, Dr. Muldoon says. Primarily, the diagnostic capabilities are much more limited, as is the presence of high-level staffing. Further, patients are less resilient to medication changes and interventions, so changes need to be done gradually.
“Hospitalists who try to practice acute-care medicine in a PAC setting may become frustrated by the length of time it takes to do a work-up, get a consultation, and respond to a patient’s change of condition,” Dr. Muldoon says. “Nonetheless, hospitalists can overcome this once recognizing this mind shift.”
According to Dr. Harrington, another challenge hospitalists may face is the inability of the hospital’s and PAC facility’s IT platforms to exchange electronic information.
“The major vendors on both sides need to figure out an interoperability strategy,” he says. “Currently, it often takes 1-3 days to receive a new patient’s discharge summary. The summary may consist of a stack of paper that takes significant time to sort through and requires the PAC facility to perform duplicate data entry. It’s a very highly inefficient process that opens up the doors to mistakes and errors of omission and commission that can result in bad patient outcomes.”
Arif Nazir, MD, CMD, FACP, AGSF, chief medical officer of Signature HealthCARE and president of SHC Medical Partners, both in Louisville, Ky., cites additional reasons the lack of seamless communication between a hospital and PAC facility is problematic. “I see physicians order laboratory tests and investigations that were already done in the hospital because they didn’t know they were already performed or never received the results,” he says. “Similarly, I see patients continue to take medications prescribed in the hospital long term even though they were only supposed to take them short term. I’ve also seen patients come to a PAC setting from a hospital without any formal understanding of their rehabilitative period and expectations for recovery.”
What’s ahead?
Looking to the future, Surafel Tsega, MD, clinical instructor at Mount Sinai Hospital in New York, says he thinks there will be a move toward greater collaboration among inpatient and PAC facilities, particularly in the discharge process, given that hospitals have an added incentive to ensure safe transitions because reimbursement from the Centers for Medicare & Medicaid Services is tied to readmissions and there are penalties for readmission. This involves more comprehensive planning regarding “warm handoffs” (e.g., real-time discussions with PAC providers about a patient’s hospital course and plan of care upon discharge), transferring of information, and so forth.
And while it can still be challenging to identify high-risk patients or determine the intensity and duration of their care, Dr. Mathew says risk-stratification tools and care pathways are continually being refined to maximize value with the limited resources available. In addition, with an increased emphasis on employing a team approach to care, there will be better integration of non-medical services to address the social determinants of health, which play significant roles in overall health and healing.
“Working with community-based organizations for this purpose will be a valuable tool for any of the population health–based initiatives,” he says.
Dr. Muldoon says he believes healthcare reform will increasingly view an inpatient admission as something to be avoided.
“If hospitalization can’t be avoided, then it should be shortened as much as possible,” he says. “This will shift inpatient care into LTACHs, SNFs, and IRFs. Hospitalists would be wise to follow patients into those settings as traditional inpatient census is reduced. This will take a few years, so hospitalists should start now in preparing for that downstream transition of individuals who were previously inpatients.”
The cost of care, and other PAC facts and figures
The amount of money that Medicare spends on post-acute care (PAC) has been increasing. In 2012, 12.6% of Medicare beneficiaries used some form of PAC, costing $62 billion.2 That amounts to the Centers for Medicare & Medicaid Services spending close to 25% of Medicare beneficiary expenses on PAC, a 133% increase from 2001 to 2012. Among the different types, $30.4 billion was spent on skilled nursing facilities (SNFs), $18.6 billion on home health, and $13.1 billion on long-term acute care (LTAC) and acute-care rehabilitation.2
It’s also been reported that after short-term acute-care hospitalization, about one in five Medicare beneficiaries requires continued specialized treatment in one of the three typical Medicare PAC settings: inpatient rehabilitation facilities (IRFs), LTAC hospitals, and SNFs.3
What’s more, hospital readmission nearly doubles the cost of an episode, so the financial implications for organizations operating in risk-bearing arrangements are significant. In 2013, 2,213 hospitals were charged $280 million in readmission penalties.2
References
1. The role of post-acute care in new care delivery models. American Hospital Association website. Available at: http://www.aha.org/research/reports/tw/15dec-tw-postacute.pdf. Accessed Nov. 7, 2016.
2. Post-acute care integration: Today and in the future. DHG Healthcare website. Available at: http://www2.dhgllp.com/res_pubs/HCG-Post-Acute-Care-Integration.pdf. Accessed Nov. 7, 2016.
3. Overview: Post-acute care transitions toolkit. Society for Hospital Medicine website. Available at: http://www.hospitalmedicine.org/Web/Quality___Innovation/Implementation_Toolkit/pact/Overview_PACT.aspx?hkey=dea3da3c-8620-46db-a00f-89f07f021958. Accessed Nov. 10, 2016.
Transplantation palliative care: The time is ripe
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.























