User login
Generic versions of Suboxone approved for opioid dependence
The Food and Drug Administration has approved the first generic versions of Suboxone (buprenorphine and naloxone) sublingual film for the medication-assisted treatment of opioid dependence.
Medication-assisted treatment (MAT) combines approved medication, such as methadone, buprenorphine, or naltrexone, with counseling and other behavioral therapies to treat opioid use disorders. Suboxone adherence reduces opioid withdrawal symptoms, the desire to use opioid, and the pleasurable effects of opioid use. Patients who receive MAT reduce their risk of death from all causes by about 50%.
“The FDA is taking new steps to advance the development of improved treatments for opioid use disorder and to make sure these medicines are accessible to the patients who need them. That includes promoting the development of better drugs and also facilitating market entry of generic versions of approved drugs to help ensure broader access,” FDA Commissioner Scott Gottlieb, MD, said in the June 14 press release.
.
The agency said data from the Substance Abuse and Mental Health Services Administration show that patients who receive MAT as an intervention for opioid use reduce their risk of death from all causes by 50%.
Common adverse events associated with Suboxone include oral hypoesthesia, glossodynia, oral mucosal erythema, headache, nausea, vomiting, hyperhidrosis, constipation, signs and symptoms of withdrawal, insomnia, pain, and peripheral edema.
Find the full press release on the FDA website.
The Food and Drug Administration has approved the first generic versions of Suboxone (buprenorphine and naloxone) sublingual film for the medication-assisted treatment of opioid dependence.
Medication-assisted treatment (MAT) combines approved medication, such as methadone, buprenorphine, or naltrexone, with counseling and other behavioral therapies to treat opioid use disorders. Suboxone adherence reduces opioid withdrawal symptoms, the desire to use opioid, and the pleasurable effects of opioid use. Patients who receive MAT reduce their risk of death from all causes by about 50%.
“The FDA is taking new steps to advance the development of improved treatments for opioid use disorder and to make sure these medicines are accessible to the patients who need them. That includes promoting the development of better drugs and also facilitating market entry of generic versions of approved drugs to help ensure broader access,” FDA Commissioner Scott Gottlieb, MD, said in the June 14 press release.
.
The agency said data from the Substance Abuse and Mental Health Services Administration show that patients who receive MAT as an intervention for opioid use reduce their risk of death from all causes by 50%.
Common adverse events associated with Suboxone include oral hypoesthesia, glossodynia, oral mucosal erythema, headache, nausea, vomiting, hyperhidrosis, constipation, signs and symptoms of withdrawal, insomnia, pain, and peripheral edema.
Find the full press release on the FDA website.
The Food and Drug Administration has approved the first generic versions of Suboxone (buprenorphine and naloxone) sublingual film for the medication-assisted treatment of opioid dependence.
Medication-assisted treatment (MAT) combines approved medication, such as methadone, buprenorphine, or naltrexone, with counseling and other behavioral therapies to treat opioid use disorders. Suboxone adherence reduces opioid withdrawal symptoms, the desire to use opioid, and the pleasurable effects of opioid use. Patients who receive MAT reduce their risk of death from all causes by about 50%.
“The FDA is taking new steps to advance the development of improved treatments for opioid use disorder and to make sure these medicines are accessible to the patients who need them. That includes promoting the development of better drugs and also facilitating market entry of generic versions of approved drugs to help ensure broader access,” FDA Commissioner Scott Gottlieb, MD, said in the June 14 press release.
.
The agency said data from the Substance Abuse and Mental Health Services Administration show that patients who receive MAT as an intervention for opioid use reduce their risk of death from all causes by 50%.
Common adverse events associated with Suboxone include oral hypoesthesia, glossodynia, oral mucosal erythema, headache, nausea, vomiting, hyperhidrosis, constipation, signs and symptoms of withdrawal, insomnia, pain, and peripheral edema.
Find the full press release on the FDA website.
FDA approves pembrolizumab for relapsed/refractory PMBCL
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
FDA approves bevacizumab for advanced ovarian cancer
The Food and Drug Administration has approved bevacizumab (Avastin) for treating stage III or IV ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, first in combination with chemotherapy (carboplatin and paclitaxel), then as monotherapy.
The approval was based on an improvement in progression-free survival (PFS) in the phase 3, three-arm GOG-0218 trial, evaluating the addition of bevacizumab to carboplatin and paclitaxel for patients with stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, the FDA said in a press statement.
The most serious adverse events of bevacizumab included gastrointestinal perforation, wounds that don’t heal, and serious bleeding. Other possible adverse events included kidney problems, fistula, severe high blood pressure, severe stroke or heart problems, and problems of the nervous system and vision. Less serious events included headache, nosebleeds, rectal bleeding, and dry skin.
The recommended bevacizumab dose for stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection is 15 mg/kg every 3 weeks with carboplatin and paclitaxel for up to 6 cycles, followed by 15 mg/kg every 3 weeks as a single agent, for a total of up to 22 cycles, the FDA said.
The Food and Drug Administration has approved bevacizumab (Avastin) for treating stage III or IV ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, first in combination with chemotherapy (carboplatin and paclitaxel), then as monotherapy.
The approval was based on an improvement in progression-free survival (PFS) in the phase 3, three-arm GOG-0218 trial, evaluating the addition of bevacizumab to carboplatin and paclitaxel for patients with stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, the FDA said in a press statement.
The most serious adverse events of bevacizumab included gastrointestinal perforation, wounds that don’t heal, and serious bleeding. Other possible adverse events included kidney problems, fistula, severe high blood pressure, severe stroke or heart problems, and problems of the nervous system and vision. Less serious events included headache, nosebleeds, rectal bleeding, and dry skin.
The recommended bevacizumab dose for stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection is 15 mg/kg every 3 weeks with carboplatin and paclitaxel for up to 6 cycles, followed by 15 mg/kg every 3 weeks as a single agent, for a total of up to 22 cycles, the FDA said.
The Food and Drug Administration has approved bevacizumab (Avastin) for treating stage III or IV ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, first in combination with chemotherapy (carboplatin and paclitaxel), then as monotherapy.
The approval was based on an improvement in progression-free survival (PFS) in the phase 3, three-arm GOG-0218 trial, evaluating the addition of bevacizumab to carboplatin and paclitaxel for patients with stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection, the FDA said in a press statement.
The most serious adverse events of bevacizumab included gastrointestinal perforation, wounds that don’t heal, and serious bleeding. Other possible adverse events included kidney problems, fistula, severe high blood pressure, severe stroke or heart problems, and problems of the nervous system and vision. Less serious events included headache, nosebleeds, rectal bleeding, and dry skin.
The recommended bevacizumab dose for stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer following initial surgical resection is 15 mg/kg every 3 weeks with carboplatin and paclitaxel for up to 6 cycles, followed by 15 mg/kg every 3 weeks as a single agent, for a total of up to 22 cycles, the FDA said.
FDA approves pembrolizumab for cervical cancer
with disease progression on or after chemotherapy whose tumors express programmed cell death ligand 1, as determined by an FDA approved test.
“This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials,” the FDA said in updated labeling.
Two patients (2.6%) had a complete response, and nine (11.7%) had a partial response. Of these 11 patients, 10 had response durations of 6 months or longer. Patients were treated with 200 mg every 3 weeks until unacceptable toxicity or documented disease progression. Over a third had serious adverse reactions, most frequently anemia, fistula, hemorrhage, and infection.
“Keytruda is now the first anti-PD-1 [anti–programmed cell death 1] therapy approved for the treatment of advanced cervical cancer, providing an important new second-line option for certain patients with this disease,” Roy Baynes, MD, a Merck executive, said in a company press release.
with disease progression on or after chemotherapy whose tumors express programmed cell death ligand 1, as determined by an FDA approved test.
“This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials,” the FDA said in updated labeling.
Two patients (2.6%) had a complete response, and nine (11.7%) had a partial response. Of these 11 patients, 10 had response durations of 6 months or longer. Patients were treated with 200 mg every 3 weeks until unacceptable toxicity or documented disease progression. Over a third had serious adverse reactions, most frequently anemia, fistula, hemorrhage, and infection.
“Keytruda is now the first anti-PD-1 [anti–programmed cell death 1] therapy approved for the treatment of advanced cervical cancer, providing an important new second-line option for certain patients with this disease,” Roy Baynes, MD, a Merck executive, said in a company press release.
with disease progression on or after chemotherapy whose tumors express programmed cell death ligand 1, as determined by an FDA approved test.
“This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials,” the FDA said in updated labeling.
Two patients (2.6%) had a complete response, and nine (11.7%) had a partial response. Of these 11 patients, 10 had response durations of 6 months or longer. Patients were treated with 200 mg every 3 weeks until unacceptable toxicity or documented disease progression. Over a third had serious adverse reactions, most frequently anemia, fistula, hemorrhage, and infection.
“Keytruda is now the first anti-PD-1 [anti–programmed cell death 1] therapy approved for the treatment of advanced cervical cancer, providing an important new second-line option for certain patients with this disease,” Roy Baynes, MD, a Merck executive, said in a company press release.
CDC warns of hepatitis A outbreaks in injection drug users
From January 2017 to April 2018, more than 2,500 cases of hepatitis A infection associated with person-to-person transmission were reported to the CDC; of the 1,900 cases where risk factors are known, 68% were related to drug use, homelessness, or both. Various state responses caused a shortage in hepatitis A vaccine during this time, however, because of improvements in controlling outbreaks and an increased vaccine supply, the vaccine has become more available.
Usage of contaminated needles or other injection paraphernalia increase risk of hepatitis A infection, and transience, economic instability, limited health care access, distrust of public officials and public messages, and frequent lack of follow-up contact information in the population who regularly inject drugs make them difficult to reach with preventative services, such as vaccination, use of sterile injection equipment, and case management and contact tracing.
“These challenges make outbreaks among these groups difficult to control,” the CDC said in a statement.
The CDC recommends health departments ensure people who report drug use are vaccinated for hepatitis A, and consider programs to educate at-risk populations, as well as to provide vaccinations in places where at-risk populations may seek treatment. Health care providers should encourage patients who report drug use to be vaccinated for the disease.
For health care professionals, the CDC recommends considering a diagnosis of hepatitis A in any patient with jaundice and clinically compatible symptoms. The agency also recommends one dose of single-antigen hepatitis A vaccine or immune globulin within 2 weeks of exposure for unvaccinated patients who have been exposed to hepatitis A virus.
Find the full Health Advisory on the CDC website.
From January 2017 to April 2018, more than 2,500 cases of hepatitis A infection associated with person-to-person transmission were reported to the CDC; of the 1,900 cases where risk factors are known, 68% were related to drug use, homelessness, or both. Various state responses caused a shortage in hepatitis A vaccine during this time, however, because of improvements in controlling outbreaks and an increased vaccine supply, the vaccine has become more available.
Usage of contaminated needles or other injection paraphernalia increase risk of hepatitis A infection, and transience, economic instability, limited health care access, distrust of public officials and public messages, and frequent lack of follow-up contact information in the population who regularly inject drugs make them difficult to reach with preventative services, such as vaccination, use of sterile injection equipment, and case management and contact tracing.
“These challenges make outbreaks among these groups difficult to control,” the CDC said in a statement.
The CDC recommends health departments ensure people who report drug use are vaccinated for hepatitis A, and consider programs to educate at-risk populations, as well as to provide vaccinations in places where at-risk populations may seek treatment. Health care providers should encourage patients who report drug use to be vaccinated for the disease.
For health care professionals, the CDC recommends considering a diagnosis of hepatitis A in any patient with jaundice and clinically compatible symptoms. The agency also recommends one dose of single-antigen hepatitis A vaccine or immune globulin within 2 weeks of exposure for unvaccinated patients who have been exposed to hepatitis A virus.
Find the full Health Advisory on the CDC website.
From January 2017 to April 2018, more than 2,500 cases of hepatitis A infection associated with person-to-person transmission were reported to the CDC; of the 1,900 cases where risk factors are known, 68% were related to drug use, homelessness, or both. Various state responses caused a shortage in hepatitis A vaccine during this time, however, because of improvements in controlling outbreaks and an increased vaccine supply, the vaccine has become more available.
Usage of contaminated needles or other injection paraphernalia increase risk of hepatitis A infection, and transience, economic instability, limited health care access, distrust of public officials and public messages, and frequent lack of follow-up contact information in the population who regularly inject drugs make them difficult to reach with preventative services, such as vaccination, use of sterile injection equipment, and case management and contact tracing.
“These challenges make outbreaks among these groups difficult to control,” the CDC said in a statement.
The CDC recommends health departments ensure people who report drug use are vaccinated for hepatitis A, and consider programs to educate at-risk populations, as well as to provide vaccinations in places where at-risk populations may seek treatment. Health care providers should encourage patients who report drug use to be vaccinated for the disease.
For health care professionals, the CDC recommends considering a diagnosis of hepatitis A in any patient with jaundice and clinically compatible symptoms. The agency also recommends one dose of single-antigen hepatitis A vaccine or immune globulin within 2 weeks of exposure for unvaccinated patients who have been exposed to hepatitis A virus.
Find the full Health Advisory on the CDC website.
FDA issues recommendations to avoid surgical fires
The Food and Drug Administration on May 29 issued a set of recommendations to medical professionals and health care facility staff to reduce the occurrence of surgical fires on or near a patient.
Surgical fires most often occur when there is an oxygen-enriched environment (a concentration of greater than 30%). In addition to an oxygen source, the other two necessary elements of the “fire triangle” are an ignition source and a fuel source.
The recommendations discuss the safe use of devices or items that may serve as a source of any one of those three elements.
Oxygen: Evaluate if supplemental oxygen is needed. If it is, titrate to the minimum concentration needed for adequate saturation. Closed oxygen delivery systems (such as a laryngeal mask or endotracheal tube) are safer than open oxygen delivery systems (such as a nasal cannula or mask). If you must use an open system, take additional precautions to exclude oxygen and flammable/combustible gases from the operative field, such as draping techniques that avoid accumulation of oxygen.
Ignition sources: Consider alternatives to using an ignition source for surgery of the head, neck, and upper chest if high concentrations of supplemental oxygen are being delivered. Check for insulation failure before use, and keep devices clean of char and tissue. When not in use, place the devices safely away from the patient and drapes. Devices are safer to use if you can allow time for the oxygen concentration in the room to decrease.
Fuel sources: Ensure dry conditions prior to draping, avoiding pooling of alcohol-based antiseptics during skin preparation. Use the appropriate-sized applicator for the surgical site. Be aware of products that may serve as a fuel source, such as oxygen-trapping gauze, plastic laryngeal masks, and aware of potential patient sources such as hair or gastrointestinal gases.
Training should include how to manage fires that do occur – stop the ignition source, then extinguish the fire – and evacuation procedures.
Read the full recommendations here.
The Food and Drug Administration on May 29 issued a set of recommendations to medical professionals and health care facility staff to reduce the occurrence of surgical fires on or near a patient.
Surgical fires most often occur when there is an oxygen-enriched environment (a concentration of greater than 30%). In addition to an oxygen source, the other two necessary elements of the “fire triangle” are an ignition source and a fuel source.
The recommendations discuss the safe use of devices or items that may serve as a source of any one of those three elements.
Oxygen: Evaluate if supplemental oxygen is needed. If it is, titrate to the minimum concentration needed for adequate saturation. Closed oxygen delivery systems (such as a laryngeal mask or endotracheal tube) are safer than open oxygen delivery systems (such as a nasal cannula or mask). If you must use an open system, take additional precautions to exclude oxygen and flammable/combustible gases from the operative field, such as draping techniques that avoid accumulation of oxygen.
Ignition sources: Consider alternatives to using an ignition source for surgery of the head, neck, and upper chest if high concentrations of supplemental oxygen are being delivered. Check for insulation failure before use, and keep devices clean of char and tissue. When not in use, place the devices safely away from the patient and drapes. Devices are safer to use if you can allow time for the oxygen concentration in the room to decrease.
Fuel sources: Ensure dry conditions prior to draping, avoiding pooling of alcohol-based antiseptics during skin preparation. Use the appropriate-sized applicator for the surgical site. Be aware of products that may serve as a fuel source, such as oxygen-trapping gauze, plastic laryngeal masks, and aware of potential patient sources such as hair or gastrointestinal gases.
Training should include how to manage fires that do occur – stop the ignition source, then extinguish the fire – and evacuation procedures.
Read the full recommendations here.
The Food and Drug Administration on May 29 issued a set of recommendations to medical professionals and health care facility staff to reduce the occurrence of surgical fires on or near a patient.
Surgical fires most often occur when there is an oxygen-enriched environment (a concentration of greater than 30%). In addition to an oxygen source, the other two necessary elements of the “fire triangle” are an ignition source and a fuel source.
The recommendations discuss the safe use of devices or items that may serve as a source of any one of those three elements.
Oxygen: Evaluate if supplemental oxygen is needed. If it is, titrate to the minimum concentration needed for adequate saturation. Closed oxygen delivery systems (such as a laryngeal mask or endotracheal tube) are safer than open oxygen delivery systems (such as a nasal cannula or mask). If you must use an open system, take additional precautions to exclude oxygen and flammable/combustible gases from the operative field, such as draping techniques that avoid accumulation of oxygen.
Ignition sources: Consider alternatives to using an ignition source for surgery of the head, neck, and upper chest if high concentrations of supplemental oxygen are being delivered. Check for insulation failure before use, and keep devices clean of char and tissue. When not in use, place the devices safely away from the patient and drapes. Devices are safer to use if you can allow time for the oxygen concentration in the room to decrease.
Fuel sources: Ensure dry conditions prior to draping, avoiding pooling of alcohol-based antiseptics during skin preparation. Use the appropriate-sized applicator for the surgical site. Be aware of products that may serve as a fuel source, such as oxygen-trapping gauze, plastic laryngeal masks, and aware of potential patient sources such as hair or gastrointestinal gases.
Training should include how to manage fires that do occur – stop the ignition source, then extinguish the fire – and evacuation procedures.
Read the full recommendations here.
FDA grants regular approval to venetoclax for CLL/SLL
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
FDA approves rituximab for treating pemphigus vulgaris
Rituximab (Rituxan) has been approved for the treatment of adults with moderate to severe pemphigus vulgaris, the manufacturer announced on June 7.
Rituximab is the first biologic approved for treating pemphigus vulgaris, Genentech, a member of the Roche group, stated in a press release announcing the approval.
The prospective, multicenter, open-label, randomized trial, conducted in France, compared the rituximab product approved in the European Union, plus short-term corticosteroid therapy (1,000 mg rituximab administered intravenously at baseline and day 14, then 500 mg at 12 and 18 months, plus 0.5 mg/kg or 1.0 mg/kg per day of prednisone tapered over 3 or 6 months) to corticosteroid therapy alone (oral prednisone 1.0 or 1.5 mg/kg per day tapered over 12 or 18 months), in 90 patients newly diagnosed with moderate to severe pemphigus.
At 24 months, 89% of those in the rituximab group met the primary endpoint, complete remission off therapy at 24 months, compared with 34% of those on corticosteroids (P less than .0001), the investigators reported (Lancet. 2017 May 20;389[10083]:2031-40). Severe adverse events were more commonly reported in the prednisone-only group.
First approved in 1997, rituximab, an anti-CD20 monoclonal antibody, is approved for non-Hodgkin lymphoma, rheumatoid arthritis, chronic lymphocytic leukemia, and granulomatosis with polyangiitis. The prescribing information includes a boxed warning about the risks of fatal infusion reactions, severe mucocutaneous reactions, hepatitis B virus reactivation, and progressive multifocal leukoencephalopathy.
The study published in the Lancet was funded by Roche, the French Ministry of Health, and the French Society of Dermatology.
Rituximab (Rituxan) has been approved for the treatment of adults with moderate to severe pemphigus vulgaris, the manufacturer announced on June 7.
Rituximab is the first biologic approved for treating pemphigus vulgaris, Genentech, a member of the Roche group, stated in a press release announcing the approval.
The prospective, multicenter, open-label, randomized trial, conducted in France, compared the rituximab product approved in the European Union, plus short-term corticosteroid therapy (1,000 mg rituximab administered intravenously at baseline and day 14, then 500 mg at 12 and 18 months, plus 0.5 mg/kg or 1.0 mg/kg per day of prednisone tapered over 3 or 6 months) to corticosteroid therapy alone (oral prednisone 1.0 or 1.5 mg/kg per day tapered over 12 or 18 months), in 90 patients newly diagnosed with moderate to severe pemphigus.
At 24 months, 89% of those in the rituximab group met the primary endpoint, complete remission off therapy at 24 months, compared with 34% of those on corticosteroids (P less than .0001), the investigators reported (Lancet. 2017 May 20;389[10083]:2031-40). Severe adverse events were more commonly reported in the prednisone-only group.
First approved in 1997, rituximab, an anti-CD20 monoclonal antibody, is approved for non-Hodgkin lymphoma, rheumatoid arthritis, chronic lymphocytic leukemia, and granulomatosis with polyangiitis. The prescribing information includes a boxed warning about the risks of fatal infusion reactions, severe mucocutaneous reactions, hepatitis B virus reactivation, and progressive multifocal leukoencephalopathy.
The study published in the Lancet was funded by Roche, the French Ministry of Health, and the French Society of Dermatology.
Rituximab (Rituxan) has been approved for the treatment of adults with moderate to severe pemphigus vulgaris, the manufacturer announced on June 7.
Rituximab is the first biologic approved for treating pemphigus vulgaris, Genentech, a member of the Roche group, stated in a press release announcing the approval.
The prospective, multicenter, open-label, randomized trial, conducted in France, compared the rituximab product approved in the European Union, plus short-term corticosteroid therapy (1,000 mg rituximab administered intravenously at baseline and day 14, then 500 mg at 12 and 18 months, plus 0.5 mg/kg or 1.0 mg/kg per day of prednisone tapered over 3 or 6 months) to corticosteroid therapy alone (oral prednisone 1.0 or 1.5 mg/kg per day tapered over 12 or 18 months), in 90 patients newly diagnosed with moderate to severe pemphigus.
At 24 months, 89% of those in the rituximab group met the primary endpoint, complete remission off therapy at 24 months, compared with 34% of those on corticosteroids (P less than .0001), the investigators reported (Lancet. 2017 May 20;389[10083]:2031-40). Severe adverse events were more commonly reported in the prednisone-only group.
First approved in 1997, rituximab, an anti-CD20 monoclonal antibody, is approved for non-Hodgkin lymphoma, rheumatoid arthritis, chronic lymphocytic leukemia, and granulomatosis with polyangiitis. The prescribing information includes a boxed warning about the risks of fatal infusion reactions, severe mucocutaneous reactions, hepatitis B virus reactivation, and progressive multifocal leukoencephalopathy.
The study published in the Lancet was funded by Roche, the French Ministry of Health, and the French Society of Dermatology.
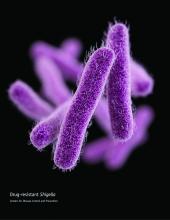
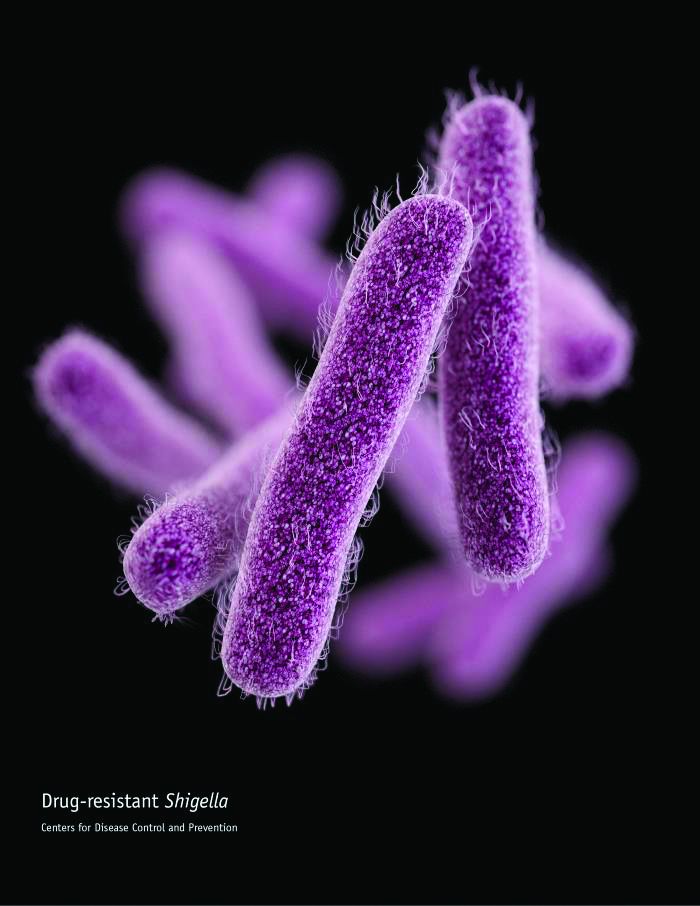
CDC concerned about multidrug-resistant Shigella
The Centers for Disease Control and Prevention have issued follow-up recommendations for managing and reporting Shigella infections because of concerns about increasing antibiotic resistance and the possibility of treatment failures.
Isolates with no resistance to quinolone antibiotics have ciprofloxacin minimum inhibitory concentration (MIC) values of less than 0.015 mcg/mL. However, the CDC has continued to identify isolates of Shigella that, while still within the susceptible range for the fluoroquinolone antibiotic ciprofloxacin (that is, having MIC values less than 1 mcg/mL), have MIC values for ciprofloxacin of 0.12-1.0 mcg/mL, thus appearing to harbor one or more resistance mechanisms. Furthermore, the CDC has identified an increasing number of isolates that have MIC values for azithromycin exceeding the epidemiologic cutoff value, which suggests some form of acquired resistance.
“CDC is particularly concerned about people who are at high risk for multidrug-resistant Shigella infections and are more likely to require antibiotic treatment, such as men who have sex with men, patients who are homeless, and immunocompromised patients. These patients often have more severe disease, prolonged shedding, and recurrent infections,” the recommendations stated.
More information can be found in the CDC’s Health Alert Network release.
The Centers for Disease Control and Prevention have issued follow-up recommendations for managing and reporting Shigella infections because of concerns about increasing antibiotic resistance and the possibility of treatment failures.
Isolates with no resistance to quinolone antibiotics have ciprofloxacin minimum inhibitory concentration (MIC) values of less than 0.015 mcg/mL. However, the CDC has continued to identify isolates of Shigella that, while still within the susceptible range for the fluoroquinolone antibiotic ciprofloxacin (that is, having MIC values less than 1 mcg/mL), have MIC values for ciprofloxacin of 0.12-1.0 mcg/mL, thus appearing to harbor one or more resistance mechanisms. Furthermore, the CDC has identified an increasing number of isolates that have MIC values for azithromycin exceeding the epidemiologic cutoff value, which suggests some form of acquired resistance.
“CDC is particularly concerned about people who are at high risk for multidrug-resistant Shigella infections and are more likely to require antibiotic treatment, such as men who have sex with men, patients who are homeless, and immunocompromised patients. These patients often have more severe disease, prolonged shedding, and recurrent infections,” the recommendations stated.
More information can be found in the CDC’s Health Alert Network release.
The Centers for Disease Control and Prevention have issued follow-up recommendations for managing and reporting Shigella infections because of concerns about increasing antibiotic resistance and the possibility of treatment failures.
Isolates with no resistance to quinolone antibiotics have ciprofloxacin minimum inhibitory concentration (MIC) values of less than 0.015 mcg/mL. However, the CDC has continued to identify isolates of Shigella that, while still within the susceptible range for the fluoroquinolone antibiotic ciprofloxacin (that is, having MIC values less than 1 mcg/mL), have MIC values for ciprofloxacin of 0.12-1.0 mcg/mL, thus appearing to harbor one or more resistance mechanisms. Furthermore, the CDC has identified an increasing number of isolates that have MIC values for azithromycin exceeding the epidemiologic cutoff value, which suggests some form of acquired resistance.
“CDC is particularly concerned about people who are at high risk for multidrug-resistant Shigella infections and are more likely to require antibiotic treatment, such as men who have sex with men, patients who are homeless, and immunocompromised patients. These patients often have more severe disease, prolonged shedding, and recurrent infections,” the recommendations stated.
More information can be found in the CDC’s Health Alert Network release.
Suicides up 30%; risk factors go beyond diagnosed disorders
About 45,000 individuals in the United States took their own lives in 2016, and about half of them had no known mental health diagnosis at the time of death, based on data from the Centers for Disease Control and Prevention. Suicide rates rose by approximately 30% across all age groups up to age 75 years.
“Suicide is preventable; that’s why it is important to understand all the factors,” Anne Schuchat, MD, principal deputy director of the Centers for Disease Control and Prevention, said in a June 7 teleconference announcing the findings. Although mental health conditions often are seen as the cause of suicide, the results highlight the need to address other factors, including relationship problems, substance abuse, trouble with life transitions, and financial difficulties.
In a Vital Signs report published June 7, a team of CDC researchers led by Deborah M. Stone, ScD, reviewed data from suicide rates by state from 1999-2016. To examine the circumstances of suicide among individuals with and without mental health conditions, the researchers also reviewed data from the CDC’s National Violent Death Reporting System for 2015, which included 27 states.
Although rates increased among all age groups,.
Overall, 54% of the suicides in 2016 had no mental health diagnosis. Compared with those with a mental health diagnosis, those without a diagnosis were more likely to be male, part of an ethnic minority, and to have a history of homicide. In addition, those without known mental health conditions were more likely to have served in the military.
The most common causes of suicide were firearms, hanging/suffocation/strangulation, and poisoning.
Individuals without known mental health conditions were significantly more likely than those with mental health con-ditions to have used firearms (55% vs. 41%) and significantly less likely to die from hanging/suffocation/strangulation (27% vs. 31%) or poisoning (10% vs. 20%) in adjusted models, the researchers noted.
“If we only look at this as a mental health issue, we won’t make the progress that we need,” Dr. Schuchat said. She urged health professionals, community organizations, government organizations, and the public at large to learn to rec-ognize warning signs and factors that can lead to suicide.
To help achieve the national goal of a 20% reduction in the annual suicide rate by 2025, the CDC has developed a technical package of recommendations for policies, prevention strategies, and resources aimed at communities and states.
In addition, “Health care providers have an important role to play” to prevent those at risk for suicide from falling through the cracks, Dr. Schuchat said. She noted the importance of protocols for patient safety and support, and she stressed that health providers should be especially vigilant during times of life transition such as changes in relationship stages, leaving for college, retirement, financial insecurity, or the loss of a loved one.
“We don’t think we can just leave this to the mental health discipline,” Dr. Schuchat noted. “Preventing suicide takes everyone; everyone in the community can help by learning the warning signs,” she said.
cpnews@mdedge.com
SOURCE: Stone D et al. MMWR. 2018 Jun 7; 67(22):617-24
About 45,000 individuals in the United States took their own lives in 2016, and about half of them had no known mental health diagnosis at the time of death, based on data from the Centers for Disease Control and Prevention. Suicide rates rose by approximately 30% across all age groups up to age 75 years.
“Suicide is preventable; that’s why it is important to understand all the factors,” Anne Schuchat, MD, principal deputy director of the Centers for Disease Control and Prevention, said in a June 7 teleconference announcing the findings. Although mental health conditions often are seen as the cause of suicide, the results highlight the need to address other factors, including relationship problems, substance abuse, trouble with life transitions, and financial difficulties.
In a Vital Signs report published June 7, a team of CDC researchers led by Deborah M. Stone, ScD, reviewed data from suicide rates by state from 1999-2016. To examine the circumstances of suicide among individuals with and without mental health conditions, the researchers also reviewed data from the CDC’s National Violent Death Reporting System for 2015, which included 27 states.
Although rates increased among all age groups,.
Overall, 54% of the suicides in 2016 had no mental health diagnosis. Compared with those with a mental health diagnosis, those without a diagnosis were more likely to be male, part of an ethnic minority, and to have a history of homicide. In addition, those without known mental health conditions were more likely to have served in the military.
The most common causes of suicide were firearms, hanging/suffocation/strangulation, and poisoning.
Individuals without known mental health conditions were significantly more likely than those with mental health con-ditions to have used firearms (55% vs. 41%) and significantly less likely to die from hanging/suffocation/strangulation (27% vs. 31%) or poisoning (10% vs. 20%) in adjusted models, the researchers noted.
“If we only look at this as a mental health issue, we won’t make the progress that we need,” Dr. Schuchat said. She urged health professionals, community organizations, government organizations, and the public at large to learn to rec-ognize warning signs and factors that can lead to suicide.
To help achieve the national goal of a 20% reduction in the annual suicide rate by 2025, the CDC has developed a technical package of recommendations for policies, prevention strategies, and resources aimed at communities and states.
In addition, “Health care providers have an important role to play” to prevent those at risk for suicide from falling through the cracks, Dr. Schuchat said. She noted the importance of protocols for patient safety and support, and she stressed that health providers should be especially vigilant during times of life transition such as changes in relationship stages, leaving for college, retirement, financial insecurity, or the loss of a loved one.
“We don’t think we can just leave this to the mental health discipline,” Dr. Schuchat noted. “Preventing suicide takes everyone; everyone in the community can help by learning the warning signs,” she said.
cpnews@mdedge.com
SOURCE: Stone D et al. MMWR. 2018 Jun 7; 67(22):617-24
About 45,000 individuals in the United States took their own lives in 2016, and about half of them had no known mental health diagnosis at the time of death, based on data from the Centers for Disease Control and Prevention. Suicide rates rose by approximately 30% across all age groups up to age 75 years.
“Suicide is preventable; that’s why it is important to understand all the factors,” Anne Schuchat, MD, principal deputy director of the Centers for Disease Control and Prevention, said in a June 7 teleconference announcing the findings. Although mental health conditions often are seen as the cause of suicide, the results highlight the need to address other factors, including relationship problems, substance abuse, trouble with life transitions, and financial difficulties.
In a Vital Signs report published June 7, a team of CDC researchers led by Deborah M. Stone, ScD, reviewed data from suicide rates by state from 1999-2016. To examine the circumstances of suicide among individuals with and without mental health conditions, the researchers also reviewed data from the CDC’s National Violent Death Reporting System for 2015, which included 27 states.
Although rates increased among all age groups,.
Overall, 54% of the suicides in 2016 had no mental health diagnosis. Compared with those with a mental health diagnosis, those without a diagnosis were more likely to be male, part of an ethnic minority, and to have a history of homicide. In addition, those without known mental health conditions were more likely to have served in the military.
The most common causes of suicide were firearms, hanging/suffocation/strangulation, and poisoning.
Individuals without known mental health conditions were significantly more likely than those with mental health con-ditions to have used firearms (55% vs. 41%) and significantly less likely to die from hanging/suffocation/strangulation (27% vs. 31%) or poisoning (10% vs. 20%) in adjusted models, the researchers noted.
“If we only look at this as a mental health issue, we won’t make the progress that we need,” Dr. Schuchat said. She urged health professionals, community organizations, government organizations, and the public at large to learn to rec-ognize warning signs and factors that can lead to suicide.
To help achieve the national goal of a 20% reduction in the annual suicide rate by 2025, the CDC has developed a technical package of recommendations for policies, prevention strategies, and resources aimed at communities and states.
In addition, “Health care providers have an important role to play” to prevent those at risk for suicide from falling through the cracks, Dr. Schuchat said. She noted the importance of protocols for patient safety and support, and she stressed that health providers should be especially vigilant during times of life transition such as changes in relationship stages, leaving for college, retirement, financial insecurity, or the loss of a loved one.
“We don’t think we can just leave this to the mental health discipline,” Dr. Schuchat noted. “Preventing suicide takes everyone; everyone in the community can help by learning the warning signs,” she said.
cpnews@mdedge.com
SOURCE: Stone D et al. MMWR. 2018 Jun 7; 67(22):617-24