User login
A return of holism? It never left osteopathic medicine
I enjoyed Dr. Jonas’s article, “A new model of care to return holism to family medicine” (J Fam Pract. 2020;69:493-498).
However, I wanted to point out that for more than 100 years the concept of the patient-centered medical home, and the outgrowth of that, has been part of osteopathic medical education, founded by A.T. Still, MD, in the 1800s.
Congratulations to the allopathic medicine profession for recognizing its significance.
Steven Shapiro, DO
Fenton, MI
I enjoyed Dr. Jonas’s article, “A new model of care to return holism to family medicine” (J Fam Pract. 2020;69:493-498).
However, I wanted to point out that for more than 100 years the concept of the patient-centered medical home, and the outgrowth of that, has been part of osteopathic medical education, founded by A.T. Still, MD, in the 1800s.
Congratulations to the allopathic medicine profession for recognizing its significance.
Steven Shapiro, DO
Fenton, MI
I enjoyed Dr. Jonas’s article, “A new model of care to return holism to family medicine” (J Fam Pract. 2020;69:493-498).
However, I wanted to point out that for more than 100 years the concept of the patient-centered medical home, and the outgrowth of that, has been part of osteopathic medical education, founded by A.T. Still, MD, in the 1800s.
Congratulations to the allopathic medicine profession for recognizing its significance.
Steven Shapiro, DO
Fenton, MI
ERRATUM
In the January 2019 article “Migraine: Expanding our Tx arsenal” (J Fam Pract. 2019;68:10-14,16-24), Table 2: Establishing the differential diagnosis of headache provided information that was incorrectly categorized. The table should not have included “Temporal arteritis” as a trigger for a headache caused by infection. Rather, the table should have listed “Temporal arteritis” among the triggers for a headache caused by an autoimmune disorder. In addition, “Acute and chronic sinusitis” and “Meningitis” should not have been listed as triggers for a headache with an iatrogenic or intoxication cause. Rather, they should have been the only triggers attributed to headaches with an infectious origin. The revised table can be found here.
In the January 2019 article “Migraine: Expanding our Tx arsenal” (J Fam Pract. 2019;68:10-14,16-24), Table 2: Establishing the differential diagnosis of headache provided information that was incorrectly categorized. The table should not have included “Temporal arteritis” as a trigger for a headache caused by infection. Rather, the table should have listed “Temporal arteritis” among the triggers for a headache caused by an autoimmune disorder. In addition, “Acute and chronic sinusitis” and “Meningitis” should not have been listed as triggers for a headache with an iatrogenic or intoxication cause. Rather, they should have been the only triggers attributed to headaches with an infectious origin. The revised table can be found here.
In the January 2019 article “Migraine: Expanding our Tx arsenal” (J Fam Pract. 2019;68:10-14,16-24), Table 2: Establishing the differential diagnosis of headache provided information that was incorrectly categorized. The table should not have included “Temporal arteritis” as a trigger for a headache caused by infection. Rather, the table should have listed “Temporal arteritis” among the triggers for a headache caused by an autoimmune disorder. In addition, “Acute and chronic sinusitis” and “Meningitis” should not have been listed as triggers for a headache with an iatrogenic or intoxication cause. Rather, they should have been the only triggers attributed to headaches with an infectious origin. The revised table can be found here.
Let’s apply the lessons from the AIDS crisis to the COVID-19 pandemic
In 2020, COVID-19 disrupted our medical system, and life in general. In the 1980s, the AIDS epidemic devastated communities and overwhelmed hospitals. There were lessons learned from the AIDS epidemic that can be applied to the current situation.
Patients with HIV-spectrum illness faced stigmatization and societal indifference, including rejection by family members, increased rates of suicide, fears of sexual and/or intrauterine transmission, substance abuse issues, and alterations of body image for those with wasting syndromes and disfiguring Kaposi lesions. AIDS prevention strategies such as the provision of condoms and needle exchange programs were controversial, and many caregivers exposed to contaminated fluids had to endure months of antiretroviral treatment.
Similar to the AIDS epidemic, the COVID-19 pandemic has had significant psychological implications for patients and caregivers. Patients with COVID-19 infections also face feelings of guilt over potentially exposing a family member to the virus; devastating socioeconomic issues; restrictive hospital visitation policies for family members; disease news oversaturation; and feelings of hopelessness. People with AIDS in the 1980s faced the possibility of dying alone, and there was initial skepticism about medications to treat HIV—just as some individuals are now uneasy about recently introduced coronavirus vaccines.
The similarities of both diseases allow us some foresight on how to deal with current COVID-19 issues. Looking back on the AIDS epidemic should teach us to prioritize attending to the mental health of sufferers and caregivers, creating advocacy and support groups for when a patient’s family is unavailable, instilling public confidence in treatment options, maintaining staff morale, addressing substance abuse (due to COVID-related stress), and depoliticizing prevention strategies. Addressing these issues is especially critical for minority populations.
As respected medical care leaders, we can provide and draw extra attention to the needs of patients’ family members and health care personnel during this COVID-19 pandemic. Hopefully, the distribution of vaccines will shorten some of our communal and professional distress.
Robert Frierson, MD
Steven Lippmann, MD
Louisville, KY
In 2020, COVID-19 disrupted our medical system, and life in general. In the 1980s, the AIDS epidemic devastated communities and overwhelmed hospitals. There were lessons learned from the AIDS epidemic that can be applied to the current situation.
Patients with HIV-spectrum illness faced stigmatization and societal indifference, including rejection by family members, increased rates of suicide, fears of sexual and/or intrauterine transmission, substance abuse issues, and alterations of body image for those with wasting syndromes and disfiguring Kaposi lesions. AIDS prevention strategies such as the provision of condoms and needle exchange programs were controversial, and many caregivers exposed to contaminated fluids had to endure months of antiretroviral treatment.
Similar to the AIDS epidemic, the COVID-19 pandemic has had significant psychological implications for patients and caregivers. Patients with COVID-19 infections also face feelings of guilt over potentially exposing a family member to the virus; devastating socioeconomic issues; restrictive hospital visitation policies for family members; disease news oversaturation; and feelings of hopelessness. People with AIDS in the 1980s faced the possibility of dying alone, and there was initial skepticism about medications to treat HIV—just as some individuals are now uneasy about recently introduced coronavirus vaccines.
The similarities of both diseases allow us some foresight on how to deal with current COVID-19 issues. Looking back on the AIDS epidemic should teach us to prioritize attending to the mental health of sufferers and caregivers, creating advocacy and support groups for when a patient’s family is unavailable, instilling public confidence in treatment options, maintaining staff morale, addressing substance abuse (due to COVID-related stress), and depoliticizing prevention strategies. Addressing these issues is especially critical for minority populations.
As respected medical care leaders, we can provide and draw extra attention to the needs of patients’ family members and health care personnel during this COVID-19 pandemic. Hopefully, the distribution of vaccines will shorten some of our communal and professional distress.
Robert Frierson, MD
Steven Lippmann, MD
Louisville, KY
In 2020, COVID-19 disrupted our medical system, and life in general. In the 1980s, the AIDS epidemic devastated communities and overwhelmed hospitals. There were lessons learned from the AIDS epidemic that can be applied to the current situation.
Patients with HIV-spectrum illness faced stigmatization and societal indifference, including rejection by family members, increased rates of suicide, fears of sexual and/or intrauterine transmission, substance abuse issues, and alterations of body image for those with wasting syndromes and disfiguring Kaposi lesions. AIDS prevention strategies such as the provision of condoms and needle exchange programs were controversial, and many caregivers exposed to contaminated fluids had to endure months of antiretroviral treatment.
Similar to the AIDS epidemic, the COVID-19 pandemic has had significant psychological implications for patients and caregivers. Patients with COVID-19 infections also face feelings of guilt over potentially exposing a family member to the virus; devastating socioeconomic issues; restrictive hospital visitation policies for family members; disease news oversaturation; and feelings of hopelessness. People with AIDS in the 1980s faced the possibility of dying alone, and there was initial skepticism about medications to treat HIV—just as some individuals are now uneasy about recently introduced coronavirus vaccines.
The similarities of both diseases allow us some foresight on how to deal with current COVID-19 issues. Looking back on the AIDS epidemic should teach us to prioritize attending to the mental health of sufferers and caregivers, creating advocacy and support groups for when a patient’s family is unavailable, instilling public confidence in treatment options, maintaining staff morale, addressing substance abuse (due to COVID-related stress), and depoliticizing prevention strategies. Addressing these issues is especially critical for minority populations.
As respected medical care leaders, we can provide and draw extra attention to the needs of patients’ family members and health care personnel during this COVID-19 pandemic. Hopefully, the distribution of vaccines will shorten some of our communal and professional distress.
Robert Frierson, MD
Steven Lippmann, MD
Louisville, KY
Meghan Markle disclosures can be used to raise awareness, reduce stigma
Suicidal thoughts require compassion and action
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
Suicidal thoughts require compassion and action
Suicidal thoughts require compassion and action
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
When I sat down to watch Oprah Winfrey’s interview with Meghan Markle on Sunday night, I didn’t know what to expect. As a psychiatrist dedicated to reducing the loss of life through suicide, I homed in on the aspect of the interview in which she discussed the depth of her suffering.
Meghan Markle is one of many celebrities to speak about their experience with suicidal crisis. Those disclosures provide opportunities to increase the public’s understanding of mental health and to deepen compassion for what others may be going through. They challenge our culturally normed assumptions: false beliefs – such as the idea that beauty, wealth, and success protect people from mental health struggles. We would do well to trust that the dichotomy between external appearances and internal experiences is always at play, and we never know what someone is going through. Human beings are both enormously resilient and vulnerable.
Suicide, while complex, is a health issue. Therefore, it is important that we all do our part in approaching mental health and suicide risk in a similar manner to other health issues.
We all have a dynamic and continuous interplay going on between life circumstances and our internal biological, genetic, and psychological makeup. The more honest and the more we learn about our own vulnerabilities and strengths, the more proactive we can be in protecting and optimizing our mental health. All individuals should be able to receive support and access to care to have their mental health needs addressed.
It’s important to note that distress leads many people to instinctually withdraw, just at a time when receiving support is even more important. In addition, cultures that traditionally emphasize self-sufficiency or stoicism may unintentionally create additional barriers to reaching out for help. Therefore, many people who experience suicidal thoughts do not disclose them to anyone. If someone does mention they are struggling, you can thank them for opening up and let them know you want to support them, and that you are there to help them find the help they need.
This is what we should tell our patients: If someone you know is struggling and might be thinking about suicide, assume you are the only one who will reach out. Having an honest conversation with the person will let them know that you care. Take the person seriously and speak up if you’re concerned about them. Ask directly if they are thinking about suicide. Help the person by connecting them with a mental health professional. If you aren’t sure if their safety is imminently at risk, connect with crisis resources and services immediately. Realize that most people who are suicidal are ambivalent about taking their life and really just want relief to their pain: Part of them wants to live and part of them wants to die. by helping them secure firearms, medications, and any other potentially lethal means.
Suicidal thoughts cut across all demographic groups, occupations, and socioeconomic strata. By understanding that life presents intense challenges for all of us and that suicidal thoughts are an indication of suffering warranting professional evaluation and intervention, we can approach the next person who discloses suicidal thoughts with compassion and action. To learn more, go to afsp.org.
Dr. Moutier is chief medical officer of the American Foundation for Suicide Prevention. She reported no disclosures.
Helping parents deal with children’s transition to in-person school
This spring may bring an unusual transition for families: a return to in-person school after nearly a year in a virtual classroom. This will undoubtedly come as a welcome relief to many parents worried about their children’s education and development and struggling with running school from home. But it is important for parents to remember that transitions, even happy ones, are difficult. You can help parents to anticipate what may be challenging about this transition for their children so that they are all prepared and can diminish struggles and support their children’s mastery.
Be curious about their children’s thoughts and feelings
Parents should adopt a truly curious and open-minded approach with their children. Remind parents that, while they are experts on their own children, they should not assume they know what their children are thinking or feeling about the return to school. Some children, especially ones struggling with learning problems or difficulty with peers, will have grown very comfortable being at home with parents or siblings. Some children, especially pre- and early teens, may have changed substantially in the year and might feel uncertain about returning to a prior team or group of friends. Some children may feel concerned about leaving a pet at home alone. Some children may be going to a new school and be anxious about facing such a big transition without the usual planning and supports. Those on a college track may be worried they are “behind” academically or in college preparation.
Parents can show up when and where their children are most likely to talk, perhaps bath time or bedtime for younger children or in the car together with their adolescents. They can ask: “Have you been thinking about what it might be like to go back to school? Have your friends been chatting about it?” They might be curious together about what might have changed in a year. What might be really great about being back in a classroom? What might they miss about home school? And what might be new? Are you worried about the work, any of your friends, or not being home? If children can begin to anticipate both the good and the difficult, they will be better equipped to face and manage the challenges and appreciate the delights.
Children in elementary school are built to master new situations but are also prone to anxiety about new expectations and demands. Parents can be calmly curious about what their thoughts, feelings, and questions are and look for answers together. Often all they need is to see parents being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion. Adolescents may be grieving the things they have missed, or they may have concerns about relationships and practical matters such as the implications for applying to college. Parents can offer compassion and validation and help them to devise their own strategies to face the practical challenges they are concerned about.
Be mindful of their children’s vulnerabilities
While most children will find the transition back to school easier than they may anticipate, there will be some for whom the transition will be very challenging. Children who have been bullied at school may have found themselves able to concentrate and learn free of the fear and stress of a classmate’s taunts or stares. Children with learning disabilities or ADHD have probably struggled with online school, but they have also likely established strategies and supports during the year that have enabled them to get enough individualized help to get their work done. These children are vulnerable to falling behind and getting discouraged when these supports are lost, and possibly not replaced with new ones in the chaos of transition. Parents should reassure their children that they will work with the school to make sure that they can succeed in the classroom as they did at home.
Children with an inhibited or shy temperament might have found that it was easier to focus and listen in the comfortable setting of home than in a busy, stimulating classroom. Children who suffer from anxiety disorders that may make separating from parents or managing the performance and social demands of school extra difficult will find the return to school especially challenging. Some younger children may have experienced the emergence of an anxiety disorder during the past year, and the return to school may mark the challenge that brings heretofore quiet symptoms into full relief.
These children have all enjoyed being able to avoid the discomfort of certain anxiety-provoking situations, and they may be particularly stressed by anticipating a return to school. Younger children may begin to have stomach aches and other physical complaints as the return to school gets close, older children may seem more withdrawn or irritable or begin discussing ways to continue school from home. Parents should help their children try to identify and describe their worries. For anxious children, having a chance to practice may be very helpful. Visiting their school, especially if it is a new school, or having a planned hangout with a friend (with appropriate precautions) is the kind of exposure that can lessen anticipatory anxiety. If this is not enough, parents should not hesitate to bring in other caring, supportive adults, such as school counselors or therapists that may be essential to helping their children face and manage what may be intense anxiety.
Consider routines to support their transition
Just as parents begin to return their children to an earlier bedtime toward the end of summer, it will be helpful to consider how changing certain routines will support their children now. If children will need to get up earlier to be ready for a bus or a team practice, they should start moving bedtime and wake-up time earlier gradually. Uniforms or backpacks that have not been seen for a year should be dug out. Children who are planning a return to a sport may benefit from gradually increasing their exercise or starting training now. This will have the added benefit of improving sleep and energy and fortifying children for the challenges of change. Parents might consider reaching out to other parents in the same class as their children and having a virtual conversation to share their thoughts.
If their family has developed some new “COVID routines” that they have come to enjoy, they should find a way to preserve them. Perhaps they are having dinner together more often or have established a family game night or Netflix night. Help parents consider how to avoid falling back into overscheduling their children and themselves. If they created a time to Zoom with distant or vulnerable loved ones, they might decide to continue this. School may determine some of their routines, but they should also prioritize their family connections and well-being in deciding how to schedule their days.
Find opportunity for mastery and meaning
As parents are listening, validating, and planning with their children, they might use this time to reflect on valuable lessons. They might point out the value of patience: Adjusting to change takes time and happens in fits and starts. It has been 12 months since many of the pandemic changes started and it will take more than a few days to adjust as schools reopen. They might point out how proud they are of what their children have been able to learn, build, or do during this year, what they admire about them. It may be a time to consider what their family may have lost and gained during the past year, what they are eager to leave behind, and what they might like to keep. And it is also a chance for parents to observe that change is an inevitable part of life (especially when growing up). It is always challenging, and often brings loss and sadness. But if we pay attention, there are also the green shoots of what is new and possible.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at pdnews@mdedge.com.
This spring may bring an unusual transition for families: a return to in-person school after nearly a year in a virtual classroom. This will undoubtedly come as a welcome relief to many parents worried about their children’s education and development and struggling with running school from home. But it is important for parents to remember that transitions, even happy ones, are difficult. You can help parents to anticipate what may be challenging about this transition for their children so that they are all prepared and can diminish struggles and support their children’s mastery.
Be curious about their children’s thoughts and feelings
Parents should adopt a truly curious and open-minded approach with their children. Remind parents that, while they are experts on their own children, they should not assume they know what their children are thinking or feeling about the return to school. Some children, especially ones struggling with learning problems or difficulty with peers, will have grown very comfortable being at home with parents or siblings. Some children, especially pre- and early teens, may have changed substantially in the year and might feel uncertain about returning to a prior team or group of friends. Some children may feel concerned about leaving a pet at home alone. Some children may be going to a new school and be anxious about facing such a big transition without the usual planning and supports. Those on a college track may be worried they are “behind” academically or in college preparation.
Parents can show up when and where their children are most likely to talk, perhaps bath time or bedtime for younger children or in the car together with their adolescents. They can ask: “Have you been thinking about what it might be like to go back to school? Have your friends been chatting about it?” They might be curious together about what might have changed in a year. What might be really great about being back in a classroom? What might they miss about home school? And what might be new? Are you worried about the work, any of your friends, or not being home? If children can begin to anticipate both the good and the difficult, they will be better equipped to face and manage the challenges and appreciate the delights.
Children in elementary school are built to master new situations but are also prone to anxiety about new expectations and demands. Parents can be calmly curious about what their thoughts, feelings, and questions are and look for answers together. Often all they need is to see parents being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion. Adolescents may be grieving the things they have missed, or they may have concerns about relationships and practical matters such as the implications for applying to college. Parents can offer compassion and validation and help them to devise their own strategies to face the practical challenges they are concerned about.
Be mindful of their children’s vulnerabilities
While most children will find the transition back to school easier than they may anticipate, there will be some for whom the transition will be very challenging. Children who have been bullied at school may have found themselves able to concentrate and learn free of the fear and stress of a classmate’s taunts or stares. Children with learning disabilities or ADHD have probably struggled with online school, but they have also likely established strategies and supports during the year that have enabled them to get enough individualized help to get their work done. These children are vulnerable to falling behind and getting discouraged when these supports are lost, and possibly not replaced with new ones in the chaos of transition. Parents should reassure their children that they will work with the school to make sure that they can succeed in the classroom as they did at home.
Children with an inhibited or shy temperament might have found that it was easier to focus and listen in the comfortable setting of home than in a busy, stimulating classroom. Children who suffer from anxiety disorders that may make separating from parents or managing the performance and social demands of school extra difficult will find the return to school especially challenging. Some younger children may have experienced the emergence of an anxiety disorder during the past year, and the return to school may mark the challenge that brings heretofore quiet symptoms into full relief.
These children have all enjoyed being able to avoid the discomfort of certain anxiety-provoking situations, and they may be particularly stressed by anticipating a return to school. Younger children may begin to have stomach aches and other physical complaints as the return to school gets close, older children may seem more withdrawn or irritable or begin discussing ways to continue school from home. Parents should help their children try to identify and describe their worries. For anxious children, having a chance to practice may be very helpful. Visiting their school, especially if it is a new school, or having a planned hangout with a friend (with appropriate precautions) is the kind of exposure that can lessen anticipatory anxiety. If this is not enough, parents should not hesitate to bring in other caring, supportive adults, such as school counselors or therapists that may be essential to helping their children face and manage what may be intense anxiety.
Consider routines to support their transition
Just as parents begin to return their children to an earlier bedtime toward the end of summer, it will be helpful to consider how changing certain routines will support their children now. If children will need to get up earlier to be ready for a bus or a team practice, they should start moving bedtime and wake-up time earlier gradually. Uniforms or backpacks that have not been seen for a year should be dug out. Children who are planning a return to a sport may benefit from gradually increasing their exercise or starting training now. This will have the added benefit of improving sleep and energy and fortifying children for the challenges of change. Parents might consider reaching out to other parents in the same class as their children and having a virtual conversation to share their thoughts.
If their family has developed some new “COVID routines” that they have come to enjoy, they should find a way to preserve them. Perhaps they are having dinner together more often or have established a family game night or Netflix night. Help parents consider how to avoid falling back into overscheduling their children and themselves. If they created a time to Zoom with distant or vulnerable loved ones, they might decide to continue this. School may determine some of their routines, but they should also prioritize their family connections and well-being in deciding how to schedule their days.
Find opportunity for mastery and meaning
As parents are listening, validating, and planning with their children, they might use this time to reflect on valuable lessons. They might point out the value of patience: Adjusting to change takes time and happens in fits and starts. It has been 12 months since many of the pandemic changes started and it will take more than a few days to adjust as schools reopen. They might point out how proud they are of what their children have been able to learn, build, or do during this year, what they admire about them. It may be a time to consider what their family may have lost and gained during the past year, what they are eager to leave behind, and what they might like to keep. And it is also a chance for parents to observe that change is an inevitable part of life (especially when growing up). It is always challenging, and often brings loss and sadness. But if we pay attention, there are also the green shoots of what is new and possible.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at pdnews@mdedge.com.
This spring may bring an unusual transition for families: a return to in-person school after nearly a year in a virtual classroom. This will undoubtedly come as a welcome relief to many parents worried about their children’s education and development and struggling with running school from home. But it is important for parents to remember that transitions, even happy ones, are difficult. You can help parents to anticipate what may be challenging about this transition for their children so that they are all prepared and can diminish struggles and support their children’s mastery.
Be curious about their children’s thoughts and feelings
Parents should adopt a truly curious and open-minded approach with their children. Remind parents that, while they are experts on their own children, they should not assume they know what their children are thinking or feeling about the return to school. Some children, especially ones struggling with learning problems or difficulty with peers, will have grown very comfortable being at home with parents or siblings. Some children, especially pre- and early teens, may have changed substantially in the year and might feel uncertain about returning to a prior team or group of friends. Some children may feel concerned about leaving a pet at home alone. Some children may be going to a new school and be anxious about facing such a big transition without the usual planning and supports. Those on a college track may be worried they are “behind” academically or in college preparation.
Parents can show up when and where their children are most likely to talk, perhaps bath time or bedtime for younger children or in the car together with their adolescents. They can ask: “Have you been thinking about what it might be like to go back to school? Have your friends been chatting about it?” They might be curious together about what might have changed in a year. What might be really great about being back in a classroom? What might they miss about home school? And what might be new? Are you worried about the work, any of your friends, or not being home? If children can begin to anticipate both the good and the difficult, they will be better equipped to face and manage the challenges and appreciate the delights.
Children in elementary school are built to master new situations but are also prone to anxiety about new expectations and demands. Parents can be calmly curious about what their thoughts, feelings, and questions are and look for answers together. Often all they need is to see parents being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion. Adolescents may be grieving the things they have missed, or they may have concerns about relationships and practical matters such as the implications for applying to college. Parents can offer compassion and validation and help them to devise their own strategies to face the practical challenges they are concerned about.
Be mindful of their children’s vulnerabilities
While most children will find the transition back to school easier than they may anticipate, there will be some for whom the transition will be very challenging. Children who have been bullied at school may have found themselves able to concentrate and learn free of the fear and stress of a classmate’s taunts or stares. Children with learning disabilities or ADHD have probably struggled with online school, but they have also likely established strategies and supports during the year that have enabled them to get enough individualized help to get their work done. These children are vulnerable to falling behind and getting discouraged when these supports are lost, and possibly not replaced with new ones in the chaos of transition. Parents should reassure their children that they will work with the school to make sure that they can succeed in the classroom as they did at home.
Children with an inhibited or shy temperament might have found that it was easier to focus and listen in the comfortable setting of home than in a busy, stimulating classroom. Children who suffer from anxiety disorders that may make separating from parents or managing the performance and social demands of school extra difficult will find the return to school especially challenging. Some younger children may have experienced the emergence of an anxiety disorder during the past year, and the return to school may mark the challenge that brings heretofore quiet symptoms into full relief.
These children have all enjoyed being able to avoid the discomfort of certain anxiety-provoking situations, and they may be particularly stressed by anticipating a return to school. Younger children may begin to have stomach aches and other physical complaints as the return to school gets close, older children may seem more withdrawn or irritable or begin discussing ways to continue school from home. Parents should help their children try to identify and describe their worries. For anxious children, having a chance to practice may be very helpful. Visiting their school, especially if it is a new school, or having a planned hangout with a friend (with appropriate precautions) is the kind of exposure that can lessen anticipatory anxiety. If this is not enough, parents should not hesitate to bring in other caring, supportive adults, such as school counselors or therapists that may be essential to helping their children face and manage what may be intense anxiety.
Consider routines to support their transition
Just as parents begin to return their children to an earlier bedtime toward the end of summer, it will be helpful to consider how changing certain routines will support their children now. If children will need to get up earlier to be ready for a bus or a team practice, they should start moving bedtime and wake-up time earlier gradually. Uniforms or backpacks that have not been seen for a year should be dug out. Children who are planning a return to a sport may benefit from gradually increasing their exercise or starting training now. This will have the added benefit of improving sleep and energy and fortifying children for the challenges of change. Parents might consider reaching out to other parents in the same class as their children and having a virtual conversation to share their thoughts.
If their family has developed some new “COVID routines” that they have come to enjoy, they should find a way to preserve them. Perhaps they are having dinner together more often or have established a family game night or Netflix night. Help parents consider how to avoid falling back into overscheduling their children and themselves. If they created a time to Zoom with distant or vulnerable loved ones, they might decide to continue this. School may determine some of their routines, but they should also prioritize their family connections and well-being in deciding how to schedule their days.
Find opportunity for mastery and meaning
As parents are listening, validating, and planning with their children, they might use this time to reflect on valuable lessons. They might point out the value of patience: Adjusting to change takes time and happens in fits and starts. It has been 12 months since many of the pandemic changes started and it will take more than a few days to adjust as schools reopen. They might point out how proud they are of what their children have been able to learn, build, or do during this year, what they admire about them. It may be a time to consider what their family may have lost and gained during the past year, what they are eager to leave behind, and what they might like to keep. And it is also a chance for parents to observe that change is an inevitable part of life (especially when growing up). It is always challenging, and often brings loss and sadness. But if we pay attention, there are also the green shoots of what is new and possible.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at pdnews@mdedge.com.
The vanguard of HIV care: Don’t forget this screening
In response, clinical care is continually adapting to the dramatically altered natural history of disease.
Today, the cutting edge of clinical care overlaps with primary care. The clinical vanguard addresses the medical vulnerabilities of patients with HIV, seeking to eliminate preventable morbidity and premature death. Among this clinical vanguard is the screening for and prevention of anal cancer. With the increased longevity of people living with HIV and the nearly universal exposure to human papillomavirus (HPV), there is now potential for progression to mucosal cellular dysplasia and eventual malignancy.
We know that prevention is possible because of the example of cervical cancer, the etiology of which is exposure to oncogenic serotypes of HPV (16 and 18 are most common). Screenings for cervical cancer (regular clinical examinations and Pap smears) and treatments to eliminate high-grade dysplasia have decreased the incidence rate by over 50% since the 1970s. Vaccination against HPV has been available since 2006 and offers the prospect of preventing HPV-associated malignancies, including head and neck cancer, in future decades.
However, rates of anal cancer are increasing. The CDC estimates that about 4,700 new cases of HPV-associated anal cancers are diagnosed in women and about 2,300 are diagnosed in men each year in the United States. Anal cancer rates in individuals with HIV have increased in the era of effective antiretrovirals and greater longevity. The highest rates, at 95 per 100,000, are in HIV-positive men who have sex with men. Very similar rates were noted in a more recent study that found increased risk with advancing age and in those with an AIDS diagnosis.
All patients with HIV should be screened
The New York State AIDS Institute Clinical Guidelines Program recommends screening for anal dysplasia in all patients with HIV. A proactive approach similar to cervical cancer screening is appropriate and includes measures easily implemented by all clinicians.
- History: Assess for rectal symptoms, anal pain, discharge, and lumps.
- Physical exam: Assess for presence of perianal lesions; perform a thorough digital rectal exam.
- Anal Pap test for anal cytology: Insert a Dacron swab moistened with tap water about 3 inches into the anal canal, applying pressure to lateral anal walls and rotating the swab. Then remove and place the swab into liquid cytology solution, shake vigorously for a full 30 seconds, and assess for any dysplasia (high-grade squamous intraepithelial lesion, low-grade intraepithelial lesion, atypical squamous cells of undetermined significance), which would warrant further evaluation by high-resolution anoscopy (HRA).
High-resolution anoscopy
HRA for anal dysplasia corresponds to colposcopy for cervical dysplasia. The ability to treat and eliminate high-risk precursor lesions interrupts the progression to malignancy. The efficacy of this strategy is being evaluated in a National Institutes of Health prospective trial called the Anchor Study. The epidemiology of HPV; the clinical horror of witnessing the painful, preventable deaths of young patients with well-controlled HIV caused by anal cancer; and the example of controlling cervical cancer have motivated my practice to assure comprehensive care for our patients.
Unfortunately, establishing HRA in one’s practice is challenging. Barriers to practice include the expense of required equipment and the absence of consensus on specific products. In addition, hands-on precepting to ease newcomers to competence is not generally available. Considerable skill is required for complete visualization of the anal transformative zone in the folds of the anal canal, and recognizing high-risk lesions requires study and accumulated experience. The International Anal Neoplasia Society is a useful resource that also offers a training course. We are invited to train ourselves, and to rely on the eventual feedback of biopsy results and the forbearance of our early patients.
The expanding scope of our medical practices must shift to meet the evolving needs of the growing population of virologically suppressed patients who are living longer. HIV care involves curing life-threatening opportunistic infections, encouraging antiretroviral adherence, and providing comprehensive care – which now includes preventing anal cancer.
A version of this article first appeared on Medscape.com.
In response, clinical care is continually adapting to the dramatically altered natural history of disease.
Today, the cutting edge of clinical care overlaps with primary care. The clinical vanguard addresses the medical vulnerabilities of patients with HIV, seeking to eliminate preventable morbidity and premature death. Among this clinical vanguard is the screening for and prevention of anal cancer. With the increased longevity of people living with HIV and the nearly universal exposure to human papillomavirus (HPV), there is now potential for progression to mucosal cellular dysplasia and eventual malignancy.
We know that prevention is possible because of the example of cervical cancer, the etiology of which is exposure to oncogenic serotypes of HPV (16 and 18 are most common). Screenings for cervical cancer (regular clinical examinations and Pap smears) and treatments to eliminate high-grade dysplasia have decreased the incidence rate by over 50% since the 1970s. Vaccination against HPV has been available since 2006 and offers the prospect of preventing HPV-associated malignancies, including head and neck cancer, in future decades.
However, rates of anal cancer are increasing. The CDC estimates that about 4,700 new cases of HPV-associated anal cancers are diagnosed in women and about 2,300 are diagnosed in men each year in the United States. Anal cancer rates in individuals with HIV have increased in the era of effective antiretrovirals and greater longevity. The highest rates, at 95 per 100,000, are in HIV-positive men who have sex with men. Very similar rates were noted in a more recent study that found increased risk with advancing age and in those with an AIDS diagnosis.
All patients with HIV should be screened
The New York State AIDS Institute Clinical Guidelines Program recommends screening for anal dysplasia in all patients with HIV. A proactive approach similar to cervical cancer screening is appropriate and includes measures easily implemented by all clinicians.
- History: Assess for rectal symptoms, anal pain, discharge, and lumps.
- Physical exam: Assess for presence of perianal lesions; perform a thorough digital rectal exam.
- Anal Pap test for anal cytology: Insert a Dacron swab moistened with tap water about 3 inches into the anal canal, applying pressure to lateral anal walls and rotating the swab. Then remove and place the swab into liquid cytology solution, shake vigorously for a full 30 seconds, and assess for any dysplasia (high-grade squamous intraepithelial lesion, low-grade intraepithelial lesion, atypical squamous cells of undetermined significance), which would warrant further evaluation by high-resolution anoscopy (HRA).
High-resolution anoscopy
HRA for anal dysplasia corresponds to colposcopy for cervical dysplasia. The ability to treat and eliminate high-risk precursor lesions interrupts the progression to malignancy. The efficacy of this strategy is being evaluated in a National Institutes of Health prospective trial called the Anchor Study. The epidemiology of HPV; the clinical horror of witnessing the painful, preventable deaths of young patients with well-controlled HIV caused by anal cancer; and the example of controlling cervical cancer have motivated my practice to assure comprehensive care for our patients.
Unfortunately, establishing HRA in one’s practice is challenging. Barriers to practice include the expense of required equipment and the absence of consensus on specific products. In addition, hands-on precepting to ease newcomers to competence is not generally available. Considerable skill is required for complete visualization of the anal transformative zone in the folds of the anal canal, and recognizing high-risk lesions requires study and accumulated experience. The International Anal Neoplasia Society is a useful resource that also offers a training course. We are invited to train ourselves, and to rely on the eventual feedback of biopsy results and the forbearance of our early patients.
The expanding scope of our medical practices must shift to meet the evolving needs of the growing population of virologically suppressed patients who are living longer. HIV care involves curing life-threatening opportunistic infections, encouraging antiretroviral adherence, and providing comprehensive care – which now includes preventing anal cancer.
A version of this article first appeared on Medscape.com.
In response, clinical care is continually adapting to the dramatically altered natural history of disease.
Today, the cutting edge of clinical care overlaps with primary care. The clinical vanguard addresses the medical vulnerabilities of patients with HIV, seeking to eliminate preventable morbidity and premature death. Among this clinical vanguard is the screening for and prevention of anal cancer. With the increased longevity of people living with HIV and the nearly universal exposure to human papillomavirus (HPV), there is now potential for progression to mucosal cellular dysplasia and eventual malignancy.
We know that prevention is possible because of the example of cervical cancer, the etiology of which is exposure to oncogenic serotypes of HPV (16 and 18 are most common). Screenings for cervical cancer (regular clinical examinations and Pap smears) and treatments to eliminate high-grade dysplasia have decreased the incidence rate by over 50% since the 1970s. Vaccination against HPV has been available since 2006 and offers the prospect of preventing HPV-associated malignancies, including head and neck cancer, in future decades.
However, rates of anal cancer are increasing. The CDC estimates that about 4,700 new cases of HPV-associated anal cancers are diagnosed in women and about 2,300 are diagnosed in men each year in the United States. Anal cancer rates in individuals with HIV have increased in the era of effective antiretrovirals and greater longevity. The highest rates, at 95 per 100,000, are in HIV-positive men who have sex with men. Very similar rates were noted in a more recent study that found increased risk with advancing age and in those with an AIDS diagnosis.
All patients with HIV should be screened
The New York State AIDS Institute Clinical Guidelines Program recommends screening for anal dysplasia in all patients with HIV. A proactive approach similar to cervical cancer screening is appropriate and includes measures easily implemented by all clinicians.
- History: Assess for rectal symptoms, anal pain, discharge, and lumps.
- Physical exam: Assess for presence of perianal lesions; perform a thorough digital rectal exam.
- Anal Pap test for anal cytology: Insert a Dacron swab moistened with tap water about 3 inches into the anal canal, applying pressure to lateral anal walls and rotating the swab. Then remove and place the swab into liquid cytology solution, shake vigorously for a full 30 seconds, and assess for any dysplasia (high-grade squamous intraepithelial lesion, low-grade intraepithelial lesion, atypical squamous cells of undetermined significance), which would warrant further evaluation by high-resolution anoscopy (HRA).
High-resolution anoscopy
HRA for anal dysplasia corresponds to colposcopy for cervical dysplasia. The ability to treat and eliminate high-risk precursor lesions interrupts the progression to malignancy. The efficacy of this strategy is being evaluated in a National Institutes of Health prospective trial called the Anchor Study. The epidemiology of HPV; the clinical horror of witnessing the painful, preventable deaths of young patients with well-controlled HIV caused by anal cancer; and the example of controlling cervical cancer have motivated my practice to assure comprehensive care for our patients.
Unfortunately, establishing HRA in one’s practice is challenging. Barriers to practice include the expense of required equipment and the absence of consensus on specific products. In addition, hands-on precepting to ease newcomers to competence is not generally available. Considerable skill is required for complete visualization of the anal transformative zone in the folds of the anal canal, and recognizing high-risk lesions requires study and accumulated experience. The International Anal Neoplasia Society is a useful resource that also offers a training course. We are invited to train ourselves, and to rely on the eventual feedback of biopsy results and the forbearance of our early patients.
The expanding scope of our medical practices must shift to meet the evolving needs of the growing population of virologically suppressed patients who are living longer. HIV care involves curing life-threatening opportunistic infections, encouraging antiretroviral adherence, and providing comprehensive care – which now includes preventing anal cancer.
A version of this article first appeared on Medscape.com.
I am the best. Sometimes.
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at dermnews@mdedge.com.
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at dermnews@mdedge.com.
The fifth and last time I was listed as Best of Boston was in 2019, when I shared honors with obstetrics, ice cream, interior design, and kitchenware.
My first time on that list was 10 years earlier, and came as a surprise. Though the magazine that runs the feature said that selections are “peer-generated,” I was never asked to evaluate any colleagues, so I don’t know who my admiring peers were or what they admired.
Three years later I was dropped from the list, for equally mysterious reasons. Maybe my acne patients did worse that year. Be that as it may, I was reinstated several years later. Perhaps my eczema outcomes surged.
How do you know when a doctor is good? I don’t need to remind you how many different ways we are evaluated. Hospitals and insurance companies monitor our prescribing practices and therapeutic outcomes. Many websites rate our performance. Read your own reviews, if you dare, penned by people who range from the totally disgruntled to the charmingly gruntled.
Often, their reasons are either beside the point or just wrong.
An example: (1 star out of 5): “Dr. Rockoff was terrible. He prescribed a very powerful regimen, and when I told him it was drying me out, he just insisted I keep using it.”
In fact – I was able to figure out who the patient was – my “powerful treatment” was over-the-counter benzoyl peroxide along with topical clindamycin. As for my insistence that she continue, she never came back for another visit. But she had called for refills.
You can surely come up with your own review tales.
But if patients don’t really understand how well we do, doctors are not necessarily much better at assessing colleagues. This came to mind recently when a close friend, increasingly hobbled by arthritis (you get more such friends as the years roll by) was looking into getting his knee replaced. He asked friends and family and got several names of orthopedists at respectable institutions. (I don’t know how many of them were Best of Boston, or even Best of Nashua, New Hampshire.)
The patients made these referrals because either they or people they knew had Dr. So-and-So replace their knee and had been pleased. That is nice to hear, but what does it prove? Even backup shortstops get on base sometimes.
So my friend called his rheumatologist, who recommended a knee specialist. My friend consulted that doctor, found her pleasant and personable, and liked what she had to say about the surgery and its expected aftermath.
My friend called back his rheumatologist to report his decision to go with his recommended doctor.
“I’m glad to hear that,” said the rheumatologist. “Three of my friends went to her and were very pleased.”
I am not in any way criticizing the rheumatologist. When people ask me for referrals – to internists, to plastic surgeons – I give them names of people I know or have sent patients to who had good experiences, or whom I just heard good things about. What can I really know about their diagnostic acumen or surgical dexterity?
A useful counterexample is what happened with my cousin who underwent back surgery a while back. He was considering several specialists when he had a discussion with a younger acquaintance who was chief resident in neurosurgery at a local medical center, and had actually operated with several of the surgeons under consideration. “Don’t go to Dr A,” said the young man. “It takes him 7 hours to do that procedure. Better go to Dr. B, who gets it done in under 3. The shorter operative time makes a big difference in speed of recovery.”
That is the kind of specialized and relevant knowledge that actually matters. How many referrals can you think of that you made or heard of about which the same can be said?
In the meantime, I will return to my own Bestness, which has been frequent, though intermittent. I like to think of myself as a vintage Chardonnay. Some years I am the best. Other years, not so much. Your best bet is to consult me in one of the former.
Preferably chilled.
Dr. Rockoff, who wrote the Dermatology News column “Under My Skin,” is now semiretired, after 40 years of practice in Brookline, Mass. He served on the clinical faculty at Tufts University, Boston, and taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available online. Write to him at dermnews@mdedge.com.
Office-based ambulatory cervical ripening prior to inpatient induction of labor
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
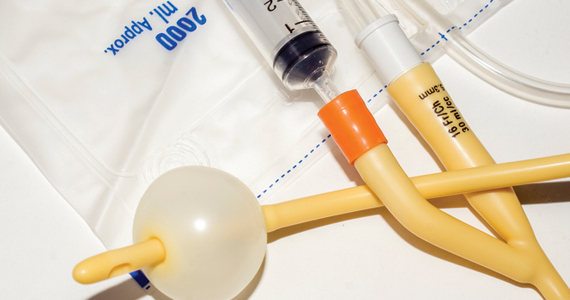
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
Assessing Psychological Interventions for Hidradenitis Suppurativa as a First Step Toward Patient-Centered Practice
Hidradenitis suppurativa (HS)(also known as acne inversa) is a chronic, recurrent, and debilitating inflammatory dermatologic disease of the hair follicle. It usually presents after puberty, with painful, deep-seated, inflamed lesions in apocrine gland–bearing areas of the body, most commonly the axillae and inguinal and anogenital regions.1
Hidradenitis suppurativa patients have a high rate of psychologic and psychiatric comorbidities that often are interrelated and multidirectional. Approximately 1 in 4 adults with HS also experience depression (prevalence among all HS patients, 16.9%), and 1 in 5 experience anxiety (prevalence, 4.9%).2,3 Hidradenitis suppurativa has been associated with bipolar disorder, schizophrenia, and suicidality.2,4
These comorbidity factors have a remarkable impact on HS patients’ quality of life (QOL). Compared to other diseases, including psoriasis, stroke, and conditions that create candidacy for heart transplantation, HS was identified as the most impairing condition.5,6 It is estimated that more than 50% of HS patients experience a very or extremely large effect on their QOL, as measured by the dermatology life quality index.6
Pain, a major component of low QOL in HS patients, has an adverse impact on emotional health. Hidradenitis suppurativa causes body image dissatisfaction, leading to shame, embarrassment, lack of self-confidence, stigmatization, and social isolation.7-9 Furthermore, patients with HS have an increased risk for antidepressant drug use, completed suicide, and suicidal behavior compared to the general population.10
Focusing therapy on physical manifestations of HS only while ignoring the psychologic aspect could lead to a vicious cycle in which stress triggers flares, leading to worsening HS, leading to more stress, and so on.11 Therefore, psychological support for HS patients is critical, and we believe it should be an integral part of managing the disease.
There is no evidence to support effective therapeutic intervention for psychological aspects of HS. We conducted a PubMed search of articles indexed for MEDLINE using the term hidradenitis in combination with psychology, psychological, mindfulness, and cognitive behavioral therapy. No relevant articles were found. Most articles on HS focused on the low QOL associated with the disease and patient coping mechanisms. However, there are a number of psychological therapies to consider and evaluate for the management of HS.
Psychological Therapies to Consider in HS
Cognitive Behavioral Treatment
Cognitive behavioral treatment has been successfully used to manage skin diseases other than HS.12 Patients’ shame and stigmatization due to body dissatisfaction often cause social isolation, which might appear as social anxiety.9,13 Cognitive behavioral treatment, or compassion-focused therapy, could increase patients’ self-acceptance and reduce shameful feelings.13
Group Therapy
Alternatively, group therapy might be beneficial for HS patients. Research has shown that most HS patients know others affected by the same disease or attend an HS support group, and patients value the support of peers with the disease.13 Therefore, group therapy meetings with HS patients that are directed by a health care professional might reduce feelings of shame and stigmatization and increase feelings of social acceptance.
Mindfulness
Another approach for managing psychological aspects of skin diseases that might be useful in HS is mindfulness-based stress reduction (MBSR), developed by Kabat-Zinn and colleagues,14 which helps patients develop mindfulness through training in meditation. It is an intensive, structured, patient-centered approach that has been successfully used in a variety of settings.14,15
Current evidence supports the use of MBSR in the adjunct treatment of chronic pain, anxiety, and depression—symptoms that have a great impact on HS patients’ QOL.16 Furthermore, MBSR is offered in a group setting, which is potentially an opportunity for peer support and understanding; social support has been reported to be highly beneficial for HS patients.17
Can the Placebo Effect Aid in Managing HS?
A recent review that assessed the placebo effect in randomized clinical trials (RCTs) of treatments for cutaneous disease demonstrated that the placebo effect in HS therapy trials is higher than in RCTs of therapies for psoriasis and eczema. This finding highlights the importance of the physician-patient relationship when managing HS, which can result in greater treatment adherence and more patient education, empowerment, and encouragement toward beneficial lifestyle changes.18
Complementary psychological interventions for managing HS might maximize the placebo effect in clinical practice.18 The placebo effect in RCTs is higher for HS treatments than for psoriasis treatments, and if patients with psoriasis improved with psychological interventions,12 it would be reasonable to expect an improvement in QOL with psychological interventions for HS.
Final Thoughts
Although a number of studies have been published in the medical literature regarding psychological intervention in psoriasis management,12 we found no clinical studies assessing the psychological management of HS. We conclude that more research is necessary to develop psychological interventions targeting HS patients because a multidisciplinary and patient-centered approach is essential for the management of HS.
- Zouboulis CC, Desai N, Emtestam L, et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J Eur Acad Dermatol Venereol. 2015;29:619-644.
- Patel KR, Lee HH, Rastogi S, et al. Association between hidradenitis suppurativa, depression, anxiety, and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;83:737-744.
- Machado MO, Stergiopoulos V, Maes M, et al. Depression and anxiety in adults with hidradenitis suppurativa: a systematic review and meta-analysis. JAMA Dermatol. 2019;155:939-945.
- Huilaja L, Tiri H, Jokelainen J, et al. Patients with hidradenitis suppurativa have a high psychiatric disease burden: a Finnish nationwide registry study. J Invest Dermatol. 2018;138:46-51.
- Sampogna F, Fania L, Mazzanti C, et al. The broad-spectrum impact of hidradenitis suppurativa on quality of life: a comparison with psoriasis. Dermatology. 2019;235:308-314.
- von der Werth JM, Jemec GB. Morbidity in patients with hidradenitis suppurativa. Br J Dermatol. 2001;144:809-813.
- Esmann S, Jemec GBE. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332.
- Schneider-Burrus S, Jost A, Peters EMJ, et al. Association of hidradenitis suppurativa with body image. JAMA Dermatol. 2018;154:447-451.
- Koumaki D, Efthymiou O, Bozi E, et al. Perspectives on perceived stigma and self-stigma in patients with hidradenitis suppurativa. Clin Cosmet Investig Dermatol. 2019;12:785-790.
- Thorlacius L, Cohen AD, Gislason GH, et al. Increased suicide risk in patients with hidradenitis suppurativa. J Invest Dermatol. 2018;138:52-57.
- Gill L, Williams M, Hamzavi I. Update on hidradenitis suppurativa: connecting the tracts. F1000Prime Rep. 2014;6:112.
- Qureshi AA, Awosika O, Baruffi F, et al. Psychological therapies in management of psoriatic skin disease: a systematic review. Am J Clin Dermatol. 2019;20:607-624.
- Keary E, Hevey D, Tobin AM. A qualitative analysis of psychological distress in hidradenitis suppurativa. Br J Dermatol. 2020;182:342-347.
- Kabat-Zinn J, Massion AO, Kristeller J, et al. Effectiveness of a meditation-based stress reduction program in the treatment of anxiety disorders. Am J Psychiatry. 1992;149:936-943.
- Evans S, Ferrando S, Findler M, et al. Mindfulness-based cognitive therapy for generalized anxiety disorder. J Anxiety Disord. 2008;22:716-721.
- Gotink RA, Chu P, Busschbach JJV, et al. Standardised mindfulness-based interventions in healthcare: an overview of systematic reviews and meta-analyses of RCTs. PLoS One. 2015;10:e0124344.
- Golbari NM, Porter ML, Kimball AM. Online communications among hidradenitis suppurativa patients reflect community needs. J Am Acad Dermatol. 2019;80:1760-1762.
- Ali AA, Seng EK, Alavi A, et al. Exploring changes in placebo treatment arms in hidradenitis suppurativa randomized clinical trials: a systematic review. J Am Acad Dermatol. 2020;82:45-53.
Hidradenitis suppurativa (HS)(also known as acne inversa) is a chronic, recurrent, and debilitating inflammatory dermatologic disease of the hair follicle. It usually presents after puberty, with painful, deep-seated, inflamed lesions in apocrine gland–bearing areas of the body, most commonly the axillae and inguinal and anogenital regions.1
Hidradenitis suppurativa patients have a high rate of psychologic and psychiatric comorbidities that often are interrelated and multidirectional. Approximately 1 in 4 adults with HS also experience depression (prevalence among all HS patients, 16.9%), and 1 in 5 experience anxiety (prevalence, 4.9%).2,3 Hidradenitis suppurativa has been associated with bipolar disorder, schizophrenia, and suicidality.2,4
These comorbidity factors have a remarkable impact on HS patients’ quality of life (QOL). Compared to other diseases, including psoriasis, stroke, and conditions that create candidacy for heart transplantation, HS was identified as the most impairing condition.5,6 It is estimated that more than 50% of HS patients experience a very or extremely large effect on their QOL, as measured by the dermatology life quality index.6
Pain, a major component of low QOL in HS patients, has an adverse impact on emotional health. Hidradenitis suppurativa causes body image dissatisfaction, leading to shame, embarrassment, lack of self-confidence, stigmatization, and social isolation.7-9 Furthermore, patients with HS have an increased risk for antidepressant drug use, completed suicide, and suicidal behavior compared to the general population.10
Focusing therapy on physical manifestations of HS only while ignoring the psychologic aspect could lead to a vicious cycle in which stress triggers flares, leading to worsening HS, leading to more stress, and so on.11 Therefore, psychological support for HS patients is critical, and we believe it should be an integral part of managing the disease.
There is no evidence to support effective therapeutic intervention for psychological aspects of HS. We conducted a PubMed search of articles indexed for MEDLINE using the term hidradenitis in combination with psychology, psychological, mindfulness, and cognitive behavioral therapy. No relevant articles were found. Most articles on HS focused on the low QOL associated with the disease and patient coping mechanisms. However, there are a number of psychological therapies to consider and evaluate for the management of HS.
Psychological Therapies to Consider in HS
Cognitive Behavioral Treatment
Cognitive behavioral treatment has been successfully used to manage skin diseases other than HS.12 Patients’ shame and stigmatization due to body dissatisfaction often cause social isolation, which might appear as social anxiety.9,13 Cognitive behavioral treatment, or compassion-focused therapy, could increase patients’ self-acceptance and reduce shameful feelings.13
Group Therapy
Alternatively, group therapy might be beneficial for HS patients. Research has shown that most HS patients know others affected by the same disease or attend an HS support group, and patients value the support of peers with the disease.13 Therefore, group therapy meetings with HS patients that are directed by a health care professional might reduce feelings of shame and stigmatization and increase feelings of social acceptance.
Mindfulness
Another approach for managing psychological aspects of skin diseases that might be useful in HS is mindfulness-based stress reduction (MBSR), developed by Kabat-Zinn and colleagues,14 which helps patients develop mindfulness through training in meditation. It is an intensive, structured, patient-centered approach that has been successfully used in a variety of settings.14,15
Current evidence supports the use of MBSR in the adjunct treatment of chronic pain, anxiety, and depression—symptoms that have a great impact on HS patients’ QOL.16 Furthermore, MBSR is offered in a group setting, which is potentially an opportunity for peer support and understanding; social support has been reported to be highly beneficial for HS patients.17
Can the Placebo Effect Aid in Managing HS?
A recent review that assessed the placebo effect in randomized clinical trials (RCTs) of treatments for cutaneous disease demonstrated that the placebo effect in HS therapy trials is higher than in RCTs of therapies for psoriasis and eczema. This finding highlights the importance of the physician-patient relationship when managing HS, which can result in greater treatment adherence and more patient education, empowerment, and encouragement toward beneficial lifestyle changes.18
Complementary psychological interventions for managing HS might maximize the placebo effect in clinical practice.18 The placebo effect in RCTs is higher for HS treatments than for psoriasis treatments, and if patients with psoriasis improved with psychological interventions,12 it would be reasonable to expect an improvement in QOL with psychological interventions for HS.
Final Thoughts
Although a number of studies have been published in the medical literature regarding psychological intervention in psoriasis management,12 we found no clinical studies assessing the psychological management of HS. We conclude that more research is necessary to develop psychological interventions targeting HS patients because a multidisciplinary and patient-centered approach is essential for the management of HS.
Hidradenitis suppurativa (HS)(also known as acne inversa) is a chronic, recurrent, and debilitating inflammatory dermatologic disease of the hair follicle. It usually presents after puberty, with painful, deep-seated, inflamed lesions in apocrine gland–bearing areas of the body, most commonly the axillae and inguinal and anogenital regions.1
Hidradenitis suppurativa patients have a high rate of psychologic and psychiatric comorbidities that often are interrelated and multidirectional. Approximately 1 in 4 adults with HS also experience depression (prevalence among all HS patients, 16.9%), and 1 in 5 experience anxiety (prevalence, 4.9%).2,3 Hidradenitis suppurativa has been associated with bipolar disorder, schizophrenia, and suicidality.2,4
These comorbidity factors have a remarkable impact on HS patients’ quality of life (QOL). Compared to other diseases, including psoriasis, stroke, and conditions that create candidacy for heart transplantation, HS was identified as the most impairing condition.5,6 It is estimated that more than 50% of HS patients experience a very or extremely large effect on their QOL, as measured by the dermatology life quality index.6
Pain, a major component of low QOL in HS patients, has an adverse impact on emotional health. Hidradenitis suppurativa causes body image dissatisfaction, leading to shame, embarrassment, lack of self-confidence, stigmatization, and social isolation.7-9 Furthermore, patients with HS have an increased risk for antidepressant drug use, completed suicide, and suicidal behavior compared to the general population.10
Focusing therapy on physical manifestations of HS only while ignoring the psychologic aspect could lead to a vicious cycle in which stress triggers flares, leading to worsening HS, leading to more stress, and so on.11 Therefore, psychological support for HS patients is critical, and we believe it should be an integral part of managing the disease.
There is no evidence to support effective therapeutic intervention for psychological aspects of HS. We conducted a PubMed search of articles indexed for MEDLINE using the term hidradenitis in combination with psychology, psychological, mindfulness, and cognitive behavioral therapy. No relevant articles were found. Most articles on HS focused on the low QOL associated with the disease and patient coping mechanisms. However, there are a number of psychological therapies to consider and evaluate for the management of HS.
Psychological Therapies to Consider in HS
Cognitive Behavioral Treatment
Cognitive behavioral treatment has been successfully used to manage skin diseases other than HS.12 Patients’ shame and stigmatization due to body dissatisfaction often cause social isolation, which might appear as social anxiety.9,13 Cognitive behavioral treatment, or compassion-focused therapy, could increase patients’ self-acceptance and reduce shameful feelings.13
Group Therapy
Alternatively, group therapy might be beneficial for HS patients. Research has shown that most HS patients know others affected by the same disease or attend an HS support group, and patients value the support of peers with the disease.13 Therefore, group therapy meetings with HS patients that are directed by a health care professional might reduce feelings of shame and stigmatization and increase feelings of social acceptance.
Mindfulness
Another approach for managing psychological aspects of skin diseases that might be useful in HS is mindfulness-based stress reduction (MBSR), developed by Kabat-Zinn and colleagues,14 which helps patients develop mindfulness through training in meditation. It is an intensive, structured, patient-centered approach that has been successfully used in a variety of settings.14,15
Current evidence supports the use of MBSR in the adjunct treatment of chronic pain, anxiety, and depression—symptoms that have a great impact on HS patients’ QOL.16 Furthermore, MBSR is offered in a group setting, which is potentially an opportunity for peer support and understanding; social support has been reported to be highly beneficial for HS patients.17
Can the Placebo Effect Aid in Managing HS?
A recent review that assessed the placebo effect in randomized clinical trials (RCTs) of treatments for cutaneous disease demonstrated that the placebo effect in HS therapy trials is higher than in RCTs of therapies for psoriasis and eczema. This finding highlights the importance of the physician-patient relationship when managing HS, which can result in greater treatment adherence and more patient education, empowerment, and encouragement toward beneficial lifestyle changes.18
Complementary psychological interventions for managing HS might maximize the placebo effect in clinical practice.18 The placebo effect in RCTs is higher for HS treatments than for psoriasis treatments, and if patients with psoriasis improved with psychological interventions,12 it would be reasonable to expect an improvement in QOL with psychological interventions for HS.
Final Thoughts
Although a number of studies have been published in the medical literature regarding psychological intervention in psoriasis management,12 we found no clinical studies assessing the psychological management of HS. We conclude that more research is necessary to develop psychological interventions targeting HS patients because a multidisciplinary and patient-centered approach is essential for the management of HS.
- Zouboulis CC, Desai N, Emtestam L, et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J Eur Acad Dermatol Venereol. 2015;29:619-644.
- Patel KR, Lee HH, Rastogi S, et al. Association between hidradenitis suppurativa, depression, anxiety, and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;83:737-744.
- Machado MO, Stergiopoulos V, Maes M, et al. Depression and anxiety in adults with hidradenitis suppurativa: a systematic review and meta-analysis. JAMA Dermatol. 2019;155:939-945.
- Huilaja L, Tiri H, Jokelainen J, et al. Patients with hidradenitis suppurativa have a high psychiatric disease burden: a Finnish nationwide registry study. J Invest Dermatol. 2018;138:46-51.
- Sampogna F, Fania L, Mazzanti C, et al. The broad-spectrum impact of hidradenitis suppurativa on quality of life: a comparison with psoriasis. Dermatology. 2019;235:308-314.
- von der Werth JM, Jemec GB. Morbidity in patients with hidradenitis suppurativa. Br J Dermatol. 2001;144:809-813.
- Esmann S, Jemec GBE. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332.
- Schneider-Burrus S, Jost A, Peters EMJ, et al. Association of hidradenitis suppurativa with body image. JAMA Dermatol. 2018;154:447-451.
- Koumaki D, Efthymiou O, Bozi E, et al. Perspectives on perceived stigma and self-stigma in patients with hidradenitis suppurativa. Clin Cosmet Investig Dermatol. 2019;12:785-790.
- Thorlacius L, Cohen AD, Gislason GH, et al. Increased suicide risk in patients with hidradenitis suppurativa. J Invest Dermatol. 2018;138:52-57.
- Gill L, Williams M, Hamzavi I. Update on hidradenitis suppurativa: connecting the tracts. F1000Prime Rep. 2014;6:112.
- Qureshi AA, Awosika O, Baruffi F, et al. Psychological therapies in management of psoriatic skin disease: a systematic review. Am J Clin Dermatol. 2019;20:607-624.
- Keary E, Hevey D, Tobin AM. A qualitative analysis of psychological distress in hidradenitis suppurativa. Br J Dermatol. 2020;182:342-347.
- Kabat-Zinn J, Massion AO, Kristeller J, et al. Effectiveness of a meditation-based stress reduction program in the treatment of anxiety disorders. Am J Psychiatry. 1992;149:936-943.
- Evans S, Ferrando S, Findler M, et al. Mindfulness-based cognitive therapy for generalized anxiety disorder. J Anxiety Disord. 2008;22:716-721.
- Gotink RA, Chu P, Busschbach JJV, et al. Standardised mindfulness-based interventions in healthcare: an overview of systematic reviews and meta-analyses of RCTs. PLoS One. 2015;10:e0124344.
- Golbari NM, Porter ML, Kimball AM. Online communications among hidradenitis suppurativa patients reflect community needs. J Am Acad Dermatol. 2019;80:1760-1762.
- Ali AA, Seng EK, Alavi A, et al. Exploring changes in placebo treatment arms in hidradenitis suppurativa randomized clinical trials: a systematic review. J Am Acad Dermatol. 2020;82:45-53.
- Zouboulis CC, Desai N, Emtestam L, et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J Eur Acad Dermatol Venereol. 2015;29:619-644.
- Patel KR, Lee HH, Rastogi S, et al. Association between hidradenitis suppurativa, depression, anxiety, and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;83:737-744.
- Machado MO, Stergiopoulos V, Maes M, et al. Depression and anxiety in adults with hidradenitis suppurativa: a systematic review and meta-analysis. JAMA Dermatol. 2019;155:939-945.
- Huilaja L, Tiri H, Jokelainen J, et al. Patients with hidradenitis suppurativa have a high psychiatric disease burden: a Finnish nationwide registry study. J Invest Dermatol. 2018;138:46-51.
- Sampogna F, Fania L, Mazzanti C, et al. The broad-spectrum impact of hidradenitis suppurativa on quality of life: a comparison with psoriasis. Dermatology. 2019;235:308-314.
- von der Werth JM, Jemec GB. Morbidity in patients with hidradenitis suppurativa. Br J Dermatol. 2001;144:809-813.
- Esmann S, Jemec GBE. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332.
- Schneider-Burrus S, Jost A, Peters EMJ, et al. Association of hidradenitis suppurativa with body image. JAMA Dermatol. 2018;154:447-451.
- Koumaki D, Efthymiou O, Bozi E, et al. Perspectives on perceived stigma and self-stigma in patients with hidradenitis suppurativa. Clin Cosmet Investig Dermatol. 2019;12:785-790.
- Thorlacius L, Cohen AD, Gislason GH, et al. Increased suicide risk in patients with hidradenitis suppurativa. J Invest Dermatol. 2018;138:52-57.
- Gill L, Williams M, Hamzavi I. Update on hidradenitis suppurativa: connecting the tracts. F1000Prime Rep. 2014;6:112.
- Qureshi AA, Awosika O, Baruffi F, et al. Psychological therapies in management of psoriatic skin disease: a systematic review. Am J Clin Dermatol. 2019;20:607-624.
- Keary E, Hevey D, Tobin AM. A qualitative analysis of psychological distress in hidradenitis suppurativa. Br J Dermatol. 2020;182:342-347.
- Kabat-Zinn J, Massion AO, Kristeller J, et al. Effectiveness of a meditation-based stress reduction program in the treatment of anxiety disorders. Am J Psychiatry. 1992;149:936-943.
- Evans S, Ferrando S, Findler M, et al. Mindfulness-based cognitive therapy for generalized anxiety disorder. J Anxiety Disord. 2008;22:716-721.
- Gotink RA, Chu P, Busschbach JJV, et al. Standardised mindfulness-based interventions in healthcare: an overview of systematic reviews and meta-analyses of RCTs. PLoS One. 2015;10:e0124344.
- Golbari NM, Porter ML, Kimball AM. Online communications among hidradenitis suppurativa patients reflect community needs. J Am Acad Dermatol. 2019;80:1760-1762.
- Ali AA, Seng EK, Alavi A, et al. Exploring changes in placebo treatment arms in hidradenitis suppurativa randomized clinical trials: a systematic review. J Am Acad Dermatol. 2020;82:45-53.
PRACTICE POINTS
- Although hidradenitis suppurativa (HS) has high rates of psychological comorbidities, management of the psychological aspects of the disease has not been studied extensively.
- Complementary psychological interventions should be evaluated for the management of HS.