User login
A Patient's Perspective on Readmissions
Years into the national discourse on reducing readmissions, hospitals and providers are still struggling with how to sustainably reduce 30‐day readmissions.[1] All‐cause hospital readmission rates for Medicare benificiaries averaged 19% from 2007 through 2011 and showed only a modest improvement to 18.4% in 2012.[2] A review of 43 studies in 2011 concluded that no single intervention was reliably associated with reducing readmission rates.[3] However, although no institution has found a magic bullet for reducing readmissions, progress has been made. A 2014 meta‐analysis of randomized trials aimed at preventing 30‐day readmissions found that overall readmission interventions are effective, and that the most successful interventions are more complex in nature and focus on empowering patients to engage in self‐care after discharge.[4] Readmission reduction efforts for patients with specific diagnoses have also made gains. Among patients with heart failure, for instance, higher rates of early outpatient follow‐up and care‐transition interventions for high‐risk patients have been shown to reduce 30‐day readmissions.[5, 6]
An emerging, yet still underexplored, area in readmissions is the importance of evaluating patient perspectives. The patient has intimate knowledge of the circumstances surrounding their readmission and can be a valuable resource. This is particularly true given evidence that patient perspectives do not always align with those of providers.[7, 8] Coleman's Care Transitions Intervention was one of the earliest care‐transition models demonstrating value in engaging patients to become actively involved in their care.[9] Since then, others have begun to analyze transitions of care from the patient perspective, identifying patient‐reported needs in anticipation of discharge and after they are home.[10, 11, 12, 13, 14] However, still only a few studies have endeavored to gain a thorough understanding of the readmitted patient perspective.[7, 15, 16] These studies have already identified important issues such as lack of patient readiness for discharge and the need for additional advanced care planning and caregiver resources. A few smaller studies have interviewed readmitted patients with specific diagnoses and have also shed light on disease‐specific issues.[17, 18, 19, 20] Outside the field of readmissions, improving patient‐centered communication has been shown to reduce expenditures on diagnostic tests,[21, 22] increase adherence to treatment,[23] and improve health outcomes.[24, 25] It is time for us to incorporate the patient voice into all areas of care.
In 2014, our group published the results of a study aimed at understanding the patient perspective surrounding readmissions. In this study, 27% of patients believed their readmission could have been prevented. This opinion was associated with not feeling ready for discharge, not having a follow‐up appointment scheduled, and poor satisfaction with the discharging team.[7] A key observation in these initial interviews was that patients often expressed sentiments of relief rather than frustration when they returned to the hospital. With the results of this previous study in mind, we designed a more comprehensive evaluation to investigate why patients felt unprepared for discharge, explore reasons for and attitudes surrounding readmissions, and identify patient‐centered interventions that could prevent future readmissions.
METHODS
Study Design and Recruitment
We designed the study as an in‐person survey of readmitted patients. Over a 7‐month period (February 11, 2014September 8, 2014), we identified all patients readmitted within 30 days to general medicine and cardiology services through daily queries from the electronic health record. The study took place in a 540‐bed tertiary academic medical center, as well as a 266‐bed affiliated community hospital. We reviewed the discharge summary from the index admission and the history and physical documentation from the readmission for exclusion criteria. Patients were excluded if they were: (1) readmitted to the intensive care unit, (2) had a planned readmission, (3) received an organ transplant in the preceding 3 months, (4) did not speak English, or (5) had a physical or mental incapacity preventing interview and no family member or caregiver was available to interview.
Patient Interviews
Five trained study volunteers approached all eligible patients for an interview starting the day after the patient was readmitted. Prior to the start of the interview, we obtained verbal consent from all patients. Interviews typically lasted 10 to 30 minutes in the patient's hospital room. Caregivers and/or family members were allowed to respond to interview questions if the patient granted them permission or if the patient was unable to participate. The interviewers were not part of the patient's medical team and the patients could refuse the interview at any time. According to the University of California Los Angeles (UCLA) Institutional Review Board, this work met criteria for quality‐improvement activities and was deemed to be exempt.
The survey was comprised of 24 questions addressing causes, preventability, and attitudes toward readmissions, readiness for discharge, quality of the discharge process, outpatient resources, and follow‐up care (see Supporting Information in the online version of this article). These areas of focus were chosen based on a pilot study of 98 patient interviews in which these topics emerged as worthy of further investigation.[7] With regard to patient readiness for discharge, we investigated correlations between patient readiness and symptom resolution, pain control, discharge location, level of support at home, and concerns about independent self‐care after discharge.
Data Analysis
We administered the surveys, collected and managed the data using REDCap (Research Electronic Data Capture) hosted at UCLA.[26] We collected demographic data, including race, ethnicity, and insurance status retrospectively though automated chart abstraction.
We summarized descriptive characteristics by mean and standard deviation (SD) for continuous variables (except for length of stay, which was summarized by median and range) and by proportions for categorical variables. To compare demographic variables between interviewed participants and those not interviewed (not available, not approached, refused, or excluded) we used Pearson 2 tests and Fisher exact tests for categorical variables and Student t tests for the only continuous variable, age. In evaluating patient readiness for discharge, we divided patients into groups of ready and not ready as determined by interview responses, then performed Pearson 2 tests and Fisher exact tests where appropriate.
For comparing the extent of burden and relief patients endorsed upon being readmitted, we subtracted the burden score (110) from the relief score (110) for each patient, resulting in a net relief score. We then performed a 1‐sample t test to determine whether the net relief was significantly different from 0. A P value of<0.05 was considered to be statistically significant. All statistical analyses were performed using R version 3.0.2 (
RESULTS
Patient Characteristics
Eight hundred nineteen patients were readmitted to general medicine and cardiology services over the 7‐month study period at both institutions. Two hundred thirty‐five patients (29%) were excluded based on the predetermined exclusion criteria, and 105 patients (13%) were not approached for interview due to time constraints. Of the 479 eligible patients approached for interview, 164 patients (34%) could not be interviewed because they were unavailable, and 85 patients (18%) refused. We interviewed 230 patients (48%). We conducted 115 interviews at our academic medical center and 115 at our community affiliate. The only significant demographic difference between interviewed and not‐interviewed patients was race (P=0.004).
Interviewed patients had a mean (SD) age of 63 (SD 20) years, and 45% were male. Sixty‐three percent of interviewees were white, 21% black, 8% Asian, and 8% other. The index admission median length of stay was 4 days, and the average time between admission and readmission was 13 days (Table 1). Seventy‐nine percent of the interviews were performed directly with the patient, and 21% were conducted predominantly with the patient's caregiver.
| Characteristic | Value |
|---|---|
| |
| Age, y, mean (SD) | 62.9 (20.2) |
| Female, n (%) | 127 (55.2) |
| Insurance status, n (%) | |
| Commercial | 36 (16.3) |
| Medi‐Cal/Medicaid | 31 (14.0) |
| Medicare | 123 (55.7) |
| Other | 5 (2.3) |
| UCLA managed care | 26 (11.8) |
| Missing | 9 |
| Race, n (%) | |
| Asian | 18 (7.9) |
| Black or African American | 48 (21.1) |
| Other/refused | 19 (8.3) |
| White or Caucasian | 143 (62.7) |
| Missing | 2 |
| Index length of stay, d, median (maximum, minimum) | 4 (1, 49) |
| Time between discharge and readmission, d, mean (SD) | 13 (9) |
| Discharge location following index admission, n (%) | |
| Home | 202 (88.2) |
| Skilled nursing facility | 3 (1.3) |
| Acute rehab facility | 17 (7.4) |
| Assisted living facility | 2 (0.9) |
| Other | 5 (2.2) |
| Missing | 1 |
Patient Readiness
Twenty‐eight percent of patients reported feeling unready for discharge from their index admission. Patients who felt that their readmission was preventable were significantly more likely to report feeling unready at the time of discharge compared to those who did not classify their readmission as preventable (53% vs 17%, P<0.01). Among patients who did not feel ready for discharge, over two‐thirds felt their symptoms were not adequately resolved. Conversely, among patients who did feel ready for discharge, only 8% felt their symptoms were not resolved (P<0.01). Patients who felt they were not ready for discharge were also significantly more likely to endorse poor pain control (43% vs 7%, P<0.01). The location of discharge (ie, home, rehab facility, or skilled nursing facility) and having someone to help take care of them at home did not significantly correlate with patient readiness. Over 80% of patients in both groups reported having someone to help at home, but patients who felt unready for discharge were significantly more likely to have concerns about taking care of themselves at home (54% vs 25%, P<0.001) (Table 2).
| All Participants, n=230 | Ready, n=164 | Not Ready, n=65 | P Value | |
|---|---|---|---|---|
| Symptoms were resolved enough to leave the hospital, n=227 | 170 (74.9%) | 149 (92.0%) | 21 (32.3%) | <0.01 |
| Felt pain was under control when left the hospital, n=229 | 190 (83.0%) | 153 (93.3%) | 37 (56.9%) | <0.01 |
| Discharged to home following index admission, n=229 | 202 (88.2%) | 146 (89.6%) | 56 (86.2%) | 0.62 |
| If discharged home, had someone at home able to help, n=202 | 178 (88.1%) | 132 (90.4%) | 46 (82.1%) | 0.17 |
| If discharged home, had concerns about being able to take of themselves at home or not being strong enough to go home, n=202 | 67 (33.2%) | 37 (25.3%) | 30 (53.6%) | <0.01 |
| Thought something could have been done to prevent them from coming back to the hospital, n=228 | 75 (32.9%) | 35 (21.6%) | 39 (60.0%) | <0.01 |
Discharge Instructions
Twenty‐nine percent of patients did not recall a physician talking to them about their discharge, and 35% did not remember receiving and reviewing the discharge paperwork. Of those who read the discharge paperwork, 23% noted difficulty identifying contact phone numbers, and 22% could not locate warning symptoms indicating when to seek medical attention. Patients were able to identify medications and follow‐up appointments on the discharge paperwork a majority of the time (92% and 85%, respectively).
Ambulatory Resources and Utilization
Patients were asked about their access to outpatient resources as well as their reason(s) for returning to the hospital. Eighty‐five percent of patients reported having a primary care doctor that they would feel comfortable calling if their symptoms worsened at home. Of the patients who indicated that they were given a contact number by their discharging team, only 56% contacted a doctor before returning to the emergency room. One‐third of patients reported knowing where to obtain urgent or same‐day care besides the emergency room. Among those who did report knowledge of same‐day care centers, 89% still chose not to utilize them.
Attitudes About Readmission
To investigate the patient experience with readmissions, patients were asked to rate the extent of the burden they felt upon returning to the hospital on a scale of 1 to 10, where 1 was no burden and 10 was extreme burden. Patients were also asked to evaluate the extent of relief they felt upon readmission using the same scale. On average, patients rated their sense of relief 1.8 points higher than their sense of burden upon readmission to the hospital (7.7 [SD 2.8] vs 5.9 [SD 3.4], P<0.001). The relief of readmission was rated as equal to or greater than the burden of readmission in 79% of cases. Lastly, patients' mean (SD) overall satisfaction with their medical care was 8.5 (SD 2.0) on a scale of 1 to 10, where 1 was the least satisfied and 10 was the most satisfied a patient could imagine.
DISCUSSION
This study performs a comprehensive evaluation of the patient perspective on 30‐day readmissions. Our previous work indicated that patients associate preventable readmissions with lack of preparedness at the time of discharge.[7] This study further evaluates the basis of this association. We found that nearly 1 in 3 readmitted patients did not feel ready to leave the hospital at the time of initial discharge. Feelings of inadequate symptom resolution and poor pain control appear to be major contributors to this sentiment. Furthermore, although 88% of patients endorse having a caretaker at home, patients with concerns about taking care of themselves are more likely to feel unready at discharge. Presumably, when healthcare providers discharge patients, they believe that the patient is ready to be discharged. However, our findings suggest that often patients do not agree, highlighting a gap between the beliefs of patients and those of healthcare providers. Creating patient‐centered education on symptom management and engaging patients in developing skills for independent self‐care may minimize this gap and allow patients to feel more prepared at discharge. Future research investigating provider opinions and the steps providers take when there is a disagreement over discharge readiness would also be useful.
One way to enhance education at the time of discharge is through improvements in printed discharge instructions. Jha et al. previously showed that chart documentation of providing discharge instructions does not correlate with patients reporting receiving discharge instructions.[27] Our study echoes this finding, with only 65% of the patients remembering receiving and reviewing the discharge paperwork. Horwitz et al. have also previously demonstrated poor comprehension of discharge planning and postdischarge care among patients discharged from an academic medical center.[28] Ensuring that all patients understand and retain their discharge instructions is an essential step in improving the patient experience and potentially decreasing readmissions. Our surveys have illuminated potential shortcomings in our own center's discharge instructions. Interventions aimed at clarifying critical pieces of information on the discharge paperwork, such as warning symptoms, contact phone numbers and follow‐up appointments, could be especially helpful.
After discharge, our findings suggest that only about half of patients will call a physician before returning to the hospital. Furthermore, there is limited knowledge and poor utilization of same‐day treatment centers besides the emergency room. In previous studies, Long et al. found that frequently readmitted patients self‐triage to the emergency room because they believe primary care clinics cannot treat acute illness.[11] Another study concluded that low‐income patients prefer hospital care to ambulatory care because of a greater sense of trust in inpatient care.[29]
Our patients' attitudes about readmission may also be different from those of providers. For patients, coming back to the hospital is not a significant burden, and satisfaction with their medical care remains high despite readmission. Additional research is needed to further explore the complex emotions patients have when coming back to the hospital and why patients may not be as upset with returning to the hospital as providers may expect. Ultimately, if patients continue to feel more comfortable being hospitalized, there are few incentives for patients to stay out of the hospital, and readmission rates will remain elevated.
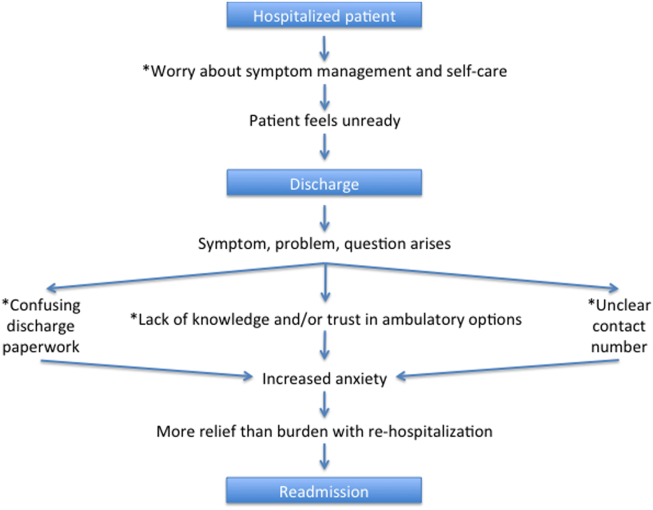
Based on our survey results we have hypothesized a potential framework for studying readmissions from a patient‐centered approach (Figure 1). This figure is not meant to imply causality, but rather to highlight a potential journey from discharge to readmission for a patient who does not feel ready to go home. This schema principally applies to patients who are worried about symptom management and/or self‐care before discharge and may not apply to everyone. Each asterisk in this framework represents an area where an intervention could be designed to improve the patient experience and possibly reduce readmissions. Such interventions should be centered around increasing patient education about symptom management and self‐care at the time of discharge, improving printed discharge instructions, increasing patient awareness of outpatient resources, enhancing communication after discharge, and changing patients' attitudes about readmissions.
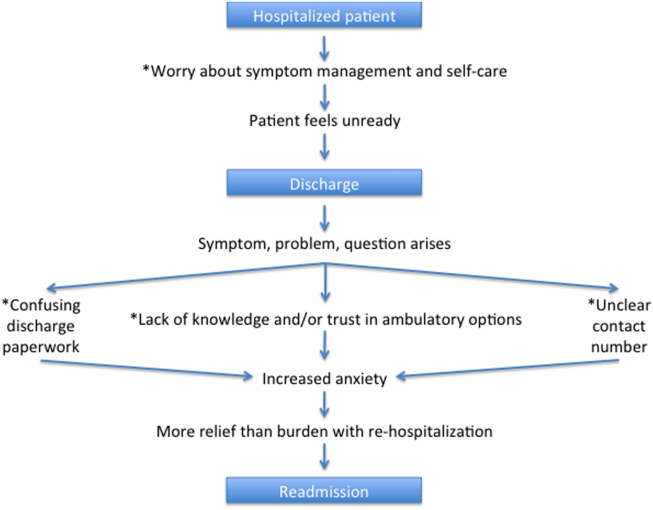
This study's limitations include that it is a single‐institution study focusing on patients admitted to a large academic medical center and its partner community hospital. Only English‐speaking patients were included, and thus our results may not be generalizable to other populations. All patients were interviewed at the time of readmission, potentially introducing recall bias regarding their prior discharge. For example, patients might be more likely to state they were not ready for discharge once they have been readmitted to the hospital. Lastly, because there are only a few prior studies interviewing readmitted patients, our survey instrument was not previously validated. Nevertheless, we believe this study offers a unique view on 30‐day readmissions from the patient perspective, with a focus on identifying areas for quality‐improvement interventions.
In conclusion, this study has enabled us to understand readmissions from a patient‐centered perspective. This perspective helps to challenge provider assumptions and gives much‐needed insight into the patient experience. For example, prior to surveying patients, one might assume that if a patient has a caregiver at home, they are unlikely to have concerns about taking care of themselves. We now know this is not the case. Similarly, we have discovered sections of our discharge paperwork that are confusing. Additionally, this study has revealed that patient attitudes regarding readmission can vary significantly from provider attitudes. By exploring the patient perspective and creating a new transition framework, we have identified specific target areas for interventions that would be meaningful to patients. As the nation continues to strive to identify sustainable solutions to reduce readmissions, the way to redesign care must always start and end with the patient.
Acknowledgements
The authors acknowledge Puneet Rana, James Haggerty‐Skeans, Jae Kim, Rhea Mathew, and Anna Do (UCLA volunteers) for helping to perform the patient interviews. We acknowledge Sandy Berry, MA (Senior Behavioral Scientist at RAND Corporation) for her help in reviewing our patient interview script. Additionally Anna Dermenchyan, RN, BSN (Senior Clinical Quality Specialist in the Department of Medicine at UCLA) provided significant administrative support.
Disclosures: This project was supported by a Patient Experience Grant from the Beryl Institute awarded to Jessica Howard‐Anderson, Sarah Lonowski, Ashley Busuttil, and Nasim Afsar‐manesh. Dr. Howard‐Anderson had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All coauthors have seen and agree with the contents of the article. The article is not under review by any other publication. An earlier version of this work was written as a research report (not peer reviewed) for the Beryl Institute (available at:
- , . What will it take to move the needle on hospital readmissions? Am J Med Qual. 2013;29(4):357–359.
- , , , , , . Medicare readmission rates showed meaningful decline in 2012. Medicare Medicaid Res Rev. 2013;3(2):E1–E12.
- , , , , . Interventions to reduce 30‐day rehospitalization: a systematic review. Ann Intern Med. 2011;155(8):520–528.
- , , , et al. Preventing 30‐day hospital readmissions: a systematic review and meta‐analysis of randomized trials. JAMA Intern Med. 2014;174(7):1095–1107.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , , et al. Allocating scarce resources in real‐time to reduce heart failure readmissions: a prospective, controlled study. BMJ Qual Saf. 2013;22(12):998–1005.
- , , , , , . Readmissions in the era of patient engagement. JAMA Intern Med. 2014;174(11):1870–1872.
- , , , et al. Comparing perspectives of patients, caregivers, and clinicians on heart failure management [published online October 23, 2015]. J Card Fail. doi: 10.1016/j.cardfail.2015.10.011.
- , , , . The care transitions intervention: results of a randomized controlled trial. Arch Intern Med. 2006;166(17):1822–1828.
- , , . Understanding rehospitalization risk: can hospital discharge be modified to reduce recurrent hospitalization? J Hosp Med. 2007;2(5):297–304.
- , , . Reasons for readmission in an underserved high‐risk population: a qualitative analysis of a series of inpatient interviews. BMJ Open. 2013;3(9):e003212.
- , , , , , . Improving care transitions: the patient perspective. J Health Commun. 2012;17(suppl 3):312–324.
- , , , et al. Challenges faced by patients with low socioeconomic status during the post‐hospital transition. J Gen Intern Med. 2014;29(2):283–289.
- , , , et al. “Missing pieces”—functional, social, and environmental barriers to recovery for vulnerable older adults transitioning from hospital to home. J Am Geriatr Soc. 2014;62(8):1556–1561.
- , , , , , . Perceptions of readmitted patients on the transition from hospital to home. J Hosp Med. 2012;7(9):709–712.
- , , , et al. Factors contributing to all‐cause 30‐day readmissions: a structured case series across 18 hospitals. Med Care. 2012;50(7):599–605.
- , , . Reasons for readmission in heart failure: perspectives of patients, caregivers, cardiologists, and heart failure nurses. Heart Lung. 2009;38(5):427–434.
- , , , et al. Patient‐identified factors related to heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2013;6(2):171–177.
- , , , , . Early readmission among patients with diabetes: a qualitative assessment of contributing factors. J Diabetes Complications. 2014;28(6):869–873.
- , , , , . “Because I was sick”: seriously ill veterans' perspectives on reason for 30‐day readmissions. J Am Geriatr Soc. 2015;63(3):537–542.
- , , , et al. The impact of patient‐centered care on outcomes. J Fam Pract. 2000;49(9):796–804.
- , , , et al. Patient‐centered communication and diagnostic testing. Ann Fam Med. 2005;3(5):415–421.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , , , et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163(1):83–90.
- , , , , . Patients' participation in medical care: effects on blood sugar control and quality of life in diabetes. J Gen Intern Med. 1988;3(5):448–457.
- , , , , , . Research electronic data capture (REDCap)—a metadata‐driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.
- , , . Public reporting of discharge planning and rates of readmissions. N Engl J Med. 2009;361(27):2637–2645.
- , , , et al. Quality of discharge practices and patient understanding at an academic medical center. JAMA Intern Med. 2013;173(18):1715–1722
- , , , , , . Understanding why patients of low socioeconomic status prefer hospitals over ambulatory care. Health Aff (Millwood). 2013;32(7):1196–1203.
Years into the national discourse on reducing readmissions, hospitals and providers are still struggling with how to sustainably reduce 30‐day readmissions.[1] All‐cause hospital readmission rates for Medicare benificiaries averaged 19% from 2007 through 2011 and showed only a modest improvement to 18.4% in 2012.[2] A review of 43 studies in 2011 concluded that no single intervention was reliably associated with reducing readmission rates.[3] However, although no institution has found a magic bullet for reducing readmissions, progress has been made. A 2014 meta‐analysis of randomized trials aimed at preventing 30‐day readmissions found that overall readmission interventions are effective, and that the most successful interventions are more complex in nature and focus on empowering patients to engage in self‐care after discharge.[4] Readmission reduction efforts for patients with specific diagnoses have also made gains. Among patients with heart failure, for instance, higher rates of early outpatient follow‐up and care‐transition interventions for high‐risk patients have been shown to reduce 30‐day readmissions.[5, 6]
An emerging, yet still underexplored, area in readmissions is the importance of evaluating patient perspectives. The patient has intimate knowledge of the circumstances surrounding their readmission and can be a valuable resource. This is particularly true given evidence that patient perspectives do not always align with those of providers.[7, 8] Coleman's Care Transitions Intervention was one of the earliest care‐transition models demonstrating value in engaging patients to become actively involved in their care.[9] Since then, others have begun to analyze transitions of care from the patient perspective, identifying patient‐reported needs in anticipation of discharge and after they are home.[10, 11, 12, 13, 14] However, still only a few studies have endeavored to gain a thorough understanding of the readmitted patient perspective.[7, 15, 16] These studies have already identified important issues such as lack of patient readiness for discharge and the need for additional advanced care planning and caregiver resources. A few smaller studies have interviewed readmitted patients with specific diagnoses and have also shed light on disease‐specific issues.[17, 18, 19, 20] Outside the field of readmissions, improving patient‐centered communication has been shown to reduce expenditures on diagnostic tests,[21, 22] increase adherence to treatment,[23] and improve health outcomes.[24, 25] It is time for us to incorporate the patient voice into all areas of care.
In 2014, our group published the results of a study aimed at understanding the patient perspective surrounding readmissions. In this study, 27% of patients believed their readmission could have been prevented. This opinion was associated with not feeling ready for discharge, not having a follow‐up appointment scheduled, and poor satisfaction with the discharging team.[7] A key observation in these initial interviews was that patients often expressed sentiments of relief rather than frustration when they returned to the hospital. With the results of this previous study in mind, we designed a more comprehensive evaluation to investigate why patients felt unprepared for discharge, explore reasons for and attitudes surrounding readmissions, and identify patient‐centered interventions that could prevent future readmissions.
METHODS
Study Design and Recruitment
We designed the study as an in‐person survey of readmitted patients. Over a 7‐month period (February 11, 2014September 8, 2014), we identified all patients readmitted within 30 days to general medicine and cardiology services through daily queries from the electronic health record. The study took place in a 540‐bed tertiary academic medical center, as well as a 266‐bed affiliated community hospital. We reviewed the discharge summary from the index admission and the history and physical documentation from the readmission for exclusion criteria. Patients were excluded if they were: (1) readmitted to the intensive care unit, (2) had a planned readmission, (3) received an organ transplant in the preceding 3 months, (4) did not speak English, or (5) had a physical or mental incapacity preventing interview and no family member or caregiver was available to interview.
Patient Interviews
Five trained study volunteers approached all eligible patients for an interview starting the day after the patient was readmitted. Prior to the start of the interview, we obtained verbal consent from all patients. Interviews typically lasted 10 to 30 minutes in the patient's hospital room. Caregivers and/or family members were allowed to respond to interview questions if the patient granted them permission or if the patient was unable to participate. The interviewers were not part of the patient's medical team and the patients could refuse the interview at any time. According to the University of California Los Angeles (UCLA) Institutional Review Board, this work met criteria for quality‐improvement activities and was deemed to be exempt.
The survey was comprised of 24 questions addressing causes, preventability, and attitudes toward readmissions, readiness for discharge, quality of the discharge process, outpatient resources, and follow‐up care (see Supporting Information in the online version of this article). These areas of focus were chosen based on a pilot study of 98 patient interviews in which these topics emerged as worthy of further investigation.[7] With regard to patient readiness for discharge, we investigated correlations between patient readiness and symptom resolution, pain control, discharge location, level of support at home, and concerns about independent self‐care after discharge.
Data Analysis
We administered the surveys, collected and managed the data using REDCap (Research Electronic Data Capture) hosted at UCLA.[26] We collected demographic data, including race, ethnicity, and insurance status retrospectively though automated chart abstraction.
We summarized descriptive characteristics by mean and standard deviation (SD) for continuous variables (except for length of stay, which was summarized by median and range) and by proportions for categorical variables. To compare demographic variables between interviewed participants and those not interviewed (not available, not approached, refused, or excluded) we used Pearson 2 tests and Fisher exact tests for categorical variables and Student t tests for the only continuous variable, age. In evaluating patient readiness for discharge, we divided patients into groups of ready and not ready as determined by interview responses, then performed Pearson 2 tests and Fisher exact tests where appropriate.
For comparing the extent of burden and relief patients endorsed upon being readmitted, we subtracted the burden score (110) from the relief score (110) for each patient, resulting in a net relief score. We then performed a 1‐sample t test to determine whether the net relief was significantly different from 0. A P value of<0.05 was considered to be statistically significant. All statistical analyses were performed using R version 3.0.2 (
RESULTS
Patient Characteristics
Eight hundred nineteen patients were readmitted to general medicine and cardiology services over the 7‐month study period at both institutions. Two hundred thirty‐five patients (29%) were excluded based on the predetermined exclusion criteria, and 105 patients (13%) were not approached for interview due to time constraints. Of the 479 eligible patients approached for interview, 164 patients (34%) could not be interviewed because they were unavailable, and 85 patients (18%) refused. We interviewed 230 patients (48%). We conducted 115 interviews at our academic medical center and 115 at our community affiliate. The only significant demographic difference between interviewed and not‐interviewed patients was race (P=0.004).
Interviewed patients had a mean (SD) age of 63 (SD 20) years, and 45% were male. Sixty‐three percent of interviewees were white, 21% black, 8% Asian, and 8% other. The index admission median length of stay was 4 days, and the average time between admission and readmission was 13 days (Table 1). Seventy‐nine percent of the interviews were performed directly with the patient, and 21% were conducted predominantly with the patient's caregiver.
| Characteristic | Value |
|---|---|
| |
| Age, y, mean (SD) | 62.9 (20.2) |
| Female, n (%) | 127 (55.2) |
| Insurance status, n (%) | |
| Commercial | 36 (16.3) |
| Medi‐Cal/Medicaid | 31 (14.0) |
| Medicare | 123 (55.7) |
| Other | 5 (2.3) |
| UCLA managed care | 26 (11.8) |
| Missing | 9 |
| Race, n (%) | |
| Asian | 18 (7.9) |
| Black or African American | 48 (21.1) |
| Other/refused | 19 (8.3) |
| White or Caucasian | 143 (62.7) |
| Missing | 2 |
| Index length of stay, d, median (maximum, minimum) | 4 (1, 49) |
| Time between discharge and readmission, d, mean (SD) | 13 (9) |
| Discharge location following index admission, n (%) | |
| Home | 202 (88.2) |
| Skilled nursing facility | 3 (1.3) |
| Acute rehab facility | 17 (7.4) |
| Assisted living facility | 2 (0.9) |
| Other | 5 (2.2) |
| Missing | 1 |
Patient Readiness
Twenty‐eight percent of patients reported feeling unready for discharge from their index admission. Patients who felt that their readmission was preventable were significantly more likely to report feeling unready at the time of discharge compared to those who did not classify their readmission as preventable (53% vs 17%, P<0.01). Among patients who did not feel ready for discharge, over two‐thirds felt their symptoms were not adequately resolved. Conversely, among patients who did feel ready for discharge, only 8% felt their symptoms were not resolved (P<0.01). Patients who felt they were not ready for discharge were also significantly more likely to endorse poor pain control (43% vs 7%, P<0.01). The location of discharge (ie, home, rehab facility, or skilled nursing facility) and having someone to help take care of them at home did not significantly correlate with patient readiness. Over 80% of patients in both groups reported having someone to help at home, but patients who felt unready for discharge were significantly more likely to have concerns about taking care of themselves at home (54% vs 25%, P<0.001) (Table 2).
| All Participants, n=230 | Ready, n=164 | Not Ready, n=65 | P Value | |
|---|---|---|---|---|
| Symptoms were resolved enough to leave the hospital, n=227 | 170 (74.9%) | 149 (92.0%) | 21 (32.3%) | <0.01 |
| Felt pain was under control when left the hospital, n=229 | 190 (83.0%) | 153 (93.3%) | 37 (56.9%) | <0.01 |
| Discharged to home following index admission, n=229 | 202 (88.2%) | 146 (89.6%) | 56 (86.2%) | 0.62 |
| If discharged home, had someone at home able to help, n=202 | 178 (88.1%) | 132 (90.4%) | 46 (82.1%) | 0.17 |
| If discharged home, had concerns about being able to take of themselves at home or not being strong enough to go home, n=202 | 67 (33.2%) | 37 (25.3%) | 30 (53.6%) | <0.01 |
| Thought something could have been done to prevent them from coming back to the hospital, n=228 | 75 (32.9%) | 35 (21.6%) | 39 (60.0%) | <0.01 |
Discharge Instructions
Twenty‐nine percent of patients did not recall a physician talking to them about their discharge, and 35% did not remember receiving and reviewing the discharge paperwork. Of those who read the discharge paperwork, 23% noted difficulty identifying contact phone numbers, and 22% could not locate warning symptoms indicating when to seek medical attention. Patients were able to identify medications and follow‐up appointments on the discharge paperwork a majority of the time (92% and 85%, respectively).
Ambulatory Resources and Utilization
Patients were asked about their access to outpatient resources as well as their reason(s) for returning to the hospital. Eighty‐five percent of patients reported having a primary care doctor that they would feel comfortable calling if their symptoms worsened at home. Of the patients who indicated that they were given a contact number by their discharging team, only 56% contacted a doctor before returning to the emergency room. One‐third of patients reported knowing where to obtain urgent or same‐day care besides the emergency room. Among those who did report knowledge of same‐day care centers, 89% still chose not to utilize them.
Attitudes About Readmission
To investigate the patient experience with readmissions, patients were asked to rate the extent of the burden they felt upon returning to the hospital on a scale of 1 to 10, where 1 was no burden and 10 was extreme burden. Patients were also asked to evaluate the extent of relief they felt upon readmission using the same scale. On average, patients rated their sense of relief 1.8 points higher than their sense of burden upon readmission to the hospital (7.7 [SD 2.8] vs 5.9 [SD 3.4], P<0.001). The relief of readmission was rated as equal to or greater than the burden of readmission in 79% of cases. Lastly, patients' mean (SD) overall satisfaction with their medical care was 8.5 (SD 2.0) on a scale of 1 to 10, where 1 was the least satisfied and 10 was the most satisfied a patient could imagine.
DISCUSSION
This study performs a comprehensive evaluation of the patient perspective on 30‐day readmissions. Our previous work indicated that patients associate preventable readmissions with lack of preparedness at the time of discharge.[7] This study further evaluates the basis of this association. We found that nearly 1 in 3 readmitted patients did not feel ready to leave the hospital at the time of initial discharge. Feelings of inadequate symptom resolution and poor pain control appear to be major contributors to this sentiment. Furthermore, although 88% of patients endorse having a caretaker at home, patients with concerns about taking care of themselves are more likely to feel unready at discharge. Presumably, when healthcare providers discharge patients, they believe that the patient is ready to be discharged. However, our findings suggest that often patients do not agree, highlighting a gap between the beliefs of patients and those of healthcare providers. Creating patient‐centered education on symptom management and engaging patients in developing skills for independent self‐care may minimize this gap and allow patients to feel more prepared at discharge. Future research investigating provider opinions and the steps providers take when there is a disagreement over discharge readiness would also be useful.
One way to enhance education at the time of discharge is through improvements in printed discharge instructions. Jha et al. previously showed that chart documentation of providing discharge instructions does not correlate with patients reporting receiving discharge instructions.[27] Our study echoes this finding, with only 65% of the patients remembering receiving and reviewing the discharge paperwork. Horwitz et al. have also previously demonstrated poor comprehension of discharge planning and postdischarge care among patients discharged from an academic medical center.[28] Ensuring that all patients understand and retain their discharge instructions is an essential step in improving the patient experience and potentially decreasing readmissions. Our surveys have illuminated potential shortcomings in our own center's discharge instructions. Interventions aimed at clarifying critical pieces of information on the discharge paperwork, such as warning symptoms, contact phone numbers and follow‐up appointments, could be especially helpful.
After discharge, our findings suggest that only about half of patients will call a physician before returning to the hospital. Furthermore, there is limited knowledge and poor utilization of same‐day treatment centers besides the emergency room. In previous studies, Long et al. found that frequently readmitted patients self‐triage to the emergency room because they believe primary care clinics cannot treat acute illness.[11] Another study concluded that low‐income patients prefer hospital care to ambulatory care because of a greater sense of trust in inpatient care.[29]
Our patients' attitudes about readmission may also be different from those of providers. For patients, coming back to the hospital is not a significant burden, and satisfaction with their medical care remains high despite readmission. Additional research is needed to further explore the complex emotions patients have when coming back to the hospital and why patients may not be as upset with returning to the hospital as providers may expect. Ultimately, if patients continue to feel more comfortable being hospitalized, there are few incentives for patients to stay out of the hospital, and readmission rates will remain elevated.
Based on our survey results we have hypothesized a potential framework for studying readmissions from a patient‐centered approach (Figure 1). This figure is not meant to imply causality, but rather to highlight a potential journey from discharge to readmission for a patient who does not feel ready to go home. This schema principally applies to patients who are worried about symptom management and/or self‐care before discharge and may not apply to everyone. Each asterisk in this framework represents an area where an intervention could be designed to improve the patient experience and possibly reduce readmissions. Such interventions should be centered around increasing patient education about symptom management and self‐care at the time of discharge, improving printed discharge instructions, increasing patient awareness of outpatient resources, enhancing communication after discharge, and changing patients' attitudes about readmissions.

This study's limitations include that it is a single‐institution study focusing on patients admitted to a large academic medical center and its partner community hospital. Only English‐speaking patients were included, and thus our results may not be generalizable to other populations. All patients were interviewed at the time of readmission, potentially introducing recall bias regarding their prior discharge. For example, patients might be more likely to state they were not ready for discharge once they have been readmitted to the hospital. Lastly, because there are only a few prior studies interviewing readmitted patients, our survey instrument was not previously validated. Nevertheless, we believe this study offers a unique view on 30‐day readmissions from the patient perspective, with a focus on identifying areas for quality‐improvement interventions.
In conclusion, this study has enabled us to understand readmissions from a patient‐centered perspective. This perspective helps to challenge provider assumptions and gives much‐needed insight into the patient experience. For example, prior to surveying patients, one might assume that if a patient has a caregiver at home, they are unlikely to have concerns about taking care of themselves. We now know this is not the case. Similarly, we have discovered sections of our discharge paperwork that are confusing. Additionally, this study has revealed that patient attitudes regarding readmission can vary significantly from provider attitudes. By exploring the patient perspective and creating a new transition framework, we have identified specific target areas for interventions that would be meaningful to patients. As the nation continues to strive to identify sustainable solutions to reduce readmissions, the way to redesign care must always start and end with the patient.
Acknowledgements
The authors acknowledge Puneet Rana, James Haggerty‐Skeans, Jae Kim, Rhea Mathew, and Anna Do (UCLA volunteers) for helping to perform the patient interviews. We acknowledge Sandy Berry, MA (Senior Behavioral Scientist at RAND Corporation) for her help in reviewing our patient interview script. Additionally Anna Dermenchyan, RN, BSN (Senior Clinical Quality Specialist in the Department of Medicine at UCLA) provided significant administrative support.
Disclosures: This project was supported by a Patient Experience Grant from the Beryl Institute awarded to Jessica Howard‐Anderson, Sarah Lonowski, Ashley Busuttil, and Nasim Afsar‐manesh. Dr. Howard‐Anderson had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All coauthors have seen and agree with the contents of the article. The article is not under review by any other publication. An earlier version of this work was written as a research report (not peer reviewed) for the Beryl Institute (available at:
Years into the national discourse on reducing readmissions, hospitals and providers are still struggling with how to sustainably reduce 30‐day readmissions.[1] All‐cause hospital readmission rates for Medicare benificiaries averaged 19% from 2007 through 2011 and showed only a modest improvement to 18.4% in 2012.[2] A review of 43 studies in 2011 concluded that no single intervention was reliably associated with reducing readmission rates.[3] However, although no institution has found a magic bullet for reducing readmissions, progress has been made. A 2014 meta‐analysis of randomized trials aimed at preventing 30‐day readmissions found that overall readmission interventions are effective, and that the most successful interventions are more complex in nature and focus on empowering patients to engage in self‐care after discharge.[4] Readmission reduction efforts for patients with specific diagnoses have also made gains. Among patients with heart failure, for instance, higher rates of early outpatient follow‐up and care‐transition interventions for high‐risk patients have been shown to reduce 30‐day readmissions.[5, 6]
An emerging, yet still underexplored, area in readmissions is the importance of evaluating patient perspectives. The patient has intimate knowledge of the circumstances surrounding their readmission and can be a valuable resource. This is particularly true given evidence that patient perspectives do not always align with those of providers.[7, 8] Coleman's Care Transitions Intervention was one of the earliest care‐transition models demonstrating value in engaging patients to become actively involved in their care.[9] Since then, others have begun to analyze transitions of care from the patient perspective, identifying patient‐reported needs in anticipation of discharge and after they are home.[10, 11, 12, 13, 14] However, still only a few studies have endeavored to gain a thorough understanding of the readmitted patient perspective.[7, 15, 16] These studies have already identified important issues such as lack of patient readiness for discharge and the need for additional advanced care planning and caregiver resources. A few smaller studies have interviewed readmitted patients with specific diagnoses and have also shed light on disease‐specific issues.[17, 18, 19, 20] Outside the field of readmissions, improving patient‐centered communication has been shown to reduce expenditures on diagnostic tests,[21, 22] increase adherence to treatment,[23] and improve health outcomes.[24, 25] It is time for us to incorporate the patient voice into all areas of care.
In 2014, our group published the results of a study aimed at understanding the patient perspective surrounding readmissions. In this study, 27% of patients believed their readmission could have been prevented. This opinion was associated with not feeling ready for discharge, not having a follow‐up appointment scheduled, and poor satisfaction with the discharging team.[7] A key observation in these initial interviews was that patients often expressed sentiments of relief rather than frustration when they returned to the hospital. With the results of this previous study in mind, we designed a more comprehensive evaluation to investigate why patients felt unprepared for discharge, explore reasons for and attitudes surrounding readmissions, and identify patient‐centered interventions that could prevent future readmissions.
METHODS
Study Design and Recruitment
We designed the study as an in‐person survey of readmitted patients. Over a 7‐month period (February 11, 2014September 8, 2014), we identified all patients readmitted within 30 days to general medicine and cardiology services through daily queries from the electronic health record. The study took place in a 540‐bed tertiary academic medical center, as well as a 266‐bed affiliated community hospital. We reviewed the discharge summary from the index admission and the history and physical documentation from the readmission for exclusion criteria. Patients were excluded if they were: (1) readmitted to the intensive care unit, (2) had a planned readmission, (3) received an organ transplant in the preceding 3 months, (4) did not speak English, or (5) had a physical or mental incapacity preventing interview and no family member or caregiver was available to interview.
Patient Interviews
Five trained study volunteers approached all eligible patients for an interview starting the day after the patient was readmitted. Prior to the start of the interview, we obtained verbal consent from all patients. Interviews typically lasted 10 to 30 minutes in the patient's hospital room. Caregivers and/or family members were allowed to respond to interview questions if the patient granted them permission or if the patient was unable to participate. The interviewers were not part of the patient's medical team and the patients could refuse the interview at any time. According to the University of California Los Angeles (UCLA) Institutional Review Board, this work met criteria for quality‐improvement activities and was deemed to be exempt.
The survey was comprised of 24 questions addressing causes, preventability, and attitudes toward readmissions, readiness for discharge, quality of the discharge process, outpatient resources, and follow‐up care (see Supporting Information in the online version of this article). These areas of focus were chosen based on a pilot study of 98 patient interviews in which these topics emerged as worthy of further investigation.[7] With regard to patient readiness for discharge, we investigated correlations between patient readiness and symptom resolution, pain control, discharge location, level of support at home, and concerns about independent self‐care after discharge.
Data Analysis
We administered the surveys, collected and managed the data using REDCap (Research Electronic Data Capture) hosted at UCLA.[26] We collected demographic data, including race, ethnicity, and insurance status retrospectively though automated chart abstraction.
We summarized descriptive characteristics by mean and standard deviation (SD) for continuous variables (except for length of stay, which was summarized by median and range) and by proportions for categorical variables. To compare demographic variables between interviewed participants and those not interviewed (not available, not approached, refused, or excluded) we used Pearson 2 tests and Fisher exact tests for categorical variables and Student t tests for the only continuous variable, age. In evaluating patient readiness for discharge, we divided patients into groups of ready and not ready as determined by interview responses, then performed Pearson 2 tests and Fisher exact tests where appropriate.
For comparing the extent of burden and relief patients endorsed upon being readmitted, we subtracted the burden score (110) from the relief score (110) for each patient, resulting in a net relief score. We then performed a 1‐sample t test to determine whether the net relief was significantly different from 0. A P value of<0.05 was considered to be statistically significant. All statistical analyses were performed using R version 3.0.2 (
RESULTS
Patient Characteristics
Eight hundred nineteen patients were readmitted to general medicine and cardiology services over the 7‐month study period at both institutions. Two hundred thirty‐five patients (29%) were excluded based on the predetermined exclusion criteria, and 105 patients (13%) were not approached for interview due to time constraints. Of the 479 eligible patients approached for interview, 164 patients (34%) could not be interviewed because they were unavailable, and 85 patients (18%) refused. We interviewed 230 patients (48%). We conducted 115 interviews at our academic medical center and 115 at our community affiliate. The only significant demographic difference between interviewed and not‐interviewed patients was race (P=0.004).
Interviewed patients had a mean (SD) age of 63 (SD 20) years, and 45% were male. Sixty‐three percent of interviewees were white, 21% black, 8% Asian, and 8% other. The index admission median length of stay was 4 days, and the average time between admission and readmission was 13 days (Table 1). Seventy‐nine percent of the interviews were performed directly with the patient, and 21% were conducted predominantly with the patient's caregiver.
| Characteristic | Value |
|---|---|
| |
| Age, y, mean (SD) | 62.9 (20.2) |
| Female, n (%) | 127 (55.2) |
| Insurance status, n (%) | |
| Commercial | 36 (16.3) |
| Medi‐Cal/Medicaid | 31 (14.0) |
| Medicare | 123 (55.7) |
| Other | 5 (2.3) |
| UCLA managed care | 26 (11.8) |
| Missing | 9 |
| Race, n (%) | |
| Asian | 18 (7.9) |
| Black or African American | 48 (21.1) |
| Other/refused | 19 (8.3) |
| White or Caucasian | 143 (62.7) |
| Missing | 2 |
| Index length of stay, d, median (maximum, minimum) | 4 (1, 49) |
| Time between discharge and readmission, d, mean (SD) | 13 (9) |
| Discharge location following index admission, n (%) | |
| Home | 202 (88.2) |
| Skilled nursing facility | 3 (1.3) |
| Acute rehab facility | 17 (7.4) |
| Assisted living facility | 2 (0.9) |
| Other | 5 (2.2) |
| Missing | 1 |
Patient Readiness
Twenty‐eight percent of patients reported feeling unready for discharge from their index admission. Patients who felt that their readmission was preventable were significantly more likely to report feeling unready at the time of discharge compared to those who did not classify their readmission as preventable (53% vs 17%, P<0.01). Among patients who did not feel ready for discharge, over two‐thirds felt their symptoms were not adequately resolved. Conversely, among patients who did feel ready for discharge, only 8% felt their symptoms were not resolved (P<0.01). Patients who felt they were not ready for discharge were also significantly more likely to endorse poor pain control (43% vs 7%, P<0.01). The location of discharge (ie, home, rehab facility, or skilled nursing facility) and having someone to help take care of them at home did not significantly correlate with patient readiness. Over 80% of patients in both groups reported having someone to help at home, but patients who felt unready for discharge were significantly more likely to have concerns about taking care of themselves at home (54% vs 25%, P<0.001) (Table 2).
| All Participants, n=230 | Ready, n=164 | Not Ready, n=65 | P Value | |
|---|---|---|---|---|
| Symptoms were resolved enough to leave the hospital, n=227 | 170 (74.9%) | 149 (92.0%) | 21 (32.3%) | <0.01 |
| Felt pain was under control when left the hospital, n=229 | 190 (83.0%) | 153 (93.3%) | 37 (56.9%) | <0.01 |
| Discharged to home following index admission, n=229 | 202 (88.2%) | 146 (89.6%) | 56 (86.2%) | 0.62 |
| If discharged home, had someone at home able to help, n=202 | 178 (88.1%) | 132 (90.4%) | 46 (82.1%) | 0.17 |
| If discharged home, had concerns about being able to take of themselves at home or not being strong enough to go home, n=202 | 67 (33.2%) | 37 (25.3%) | 30 (53.6%) | <0.01 |
| Thought something could have been done to prevent them from coming back to the hospital, n=228 | 75 (32.9%) | 35 (21.6%) | 39 (60.0%) | <0.01 |
Discharge Instructions
Twenty‐nine percent of patients did not recall a physician talking to them about their discharge, and 35% did not remember receiving and reviewing the discharge paperwork. Of those who read the discharge paperwork, 23% noted difficulty identifying contact phone numbers, and 22% could not locate warning symptoms indicating when to seek medical attention. Patients were able to identify medications and follow‐up appointments on the discharge paperwork a majority of the time (92% and 85%, respectively).
Ambulatory Resources and Utilization
Patients were asked about their access to outpatient resources as well as their reason(s) for returning to the hospital. Eighty‐five percent of patients reported having a primary care doctor that they would feel comfortable calling if their symptoms worsened at home. Of the patients who indicated that they were given a contact number by their discharging team, only 56% contacted a doctor before returning to the emergency room. One‐third of patients reported knowing where to obtain urgent or same‐day care besides the emergency room. Among those who did report knowledge of same‐day care centers, 89% still chose not to utilize them.
Attitudes About Readmission
To investigate the patient experience with readmissions, patients were asked to rate the extent of the burden they felt upon returning to the hospital on a scale of 1 to 10, where 1 was no burden and 10 was extreme burden. Patients were also asked to evaluate the extent of relief they felt upon readmission using the same scale. On average, patients rated their sense of relief 1.8 points higher than their sense of burden upon readmission to the hospital (7.7 [SD 2.8] vs 5.9 [SD 3.4], P<0.001). The relief of readmission was rated as equal to or greater than the burden of readmission in 79% of cases. Lastly, patients' mean (SD) overall satisfaction with their medical care was 8.5 (SD 2.0) on a scale of 1 to 10, where 1 was the least satisfied and 10 was the most satisfied a patient could imagine.
DISCUSSION
This study performs a comprehensive evaluation of the patient perspective on 30‐day readmissions. Our previous work indicated that patients associate preventable readmissions with lack of preparedness at the time of discharge.[7] This study further evaluates the basis of this association. We found that nearly 1 in 3 readmitted patients did not feel ready to leave the hospital at the time of initial discharge. Feelings of inadequate symptom resolution and poor pain control appear to be major contributors to this sentiment. Furthermore, although 88% of patients endorse having a caretaker at home, patients with concerns about taking care of themselves are more likely to feel unready at discharge. Presumably, when healthcare providers discharge patients, they believe that the patient is ready to be discharged. However, our findings suggest that often patients do not agree, highlighting a gap between the beliefs of patients and those of healthcare providers. Creating patient‐centered education on symptom management and engaging patients in developing skills for independent self‐care may minimize this gap and allow patients to feel more prepared at discharge. Future research investigating provider opinions and the steps providers take when there is a disagreement over discharge readiness would also be useful.
One way to enhance education at the time of discharge is through improvements in printed discharge instructions. Jha et al. previously showed that chart documentation of providing discharge instructions does not correlate with patients reporting receiving discharge instructions.[27] Our study echoes this finding, with only 65% of the patients remembering receiving and reviewing the discharge paperwork. Horwitz et al. have also previously demonstrated poor comprehension of discharge planning and postdischarge care among patients discharged from an academic medical center.[28] Ensuring that all patients understand and retain their discharge instructions is an essential step in improving the patient experience and potentially decreasing readmissions. Our surveys have illuminated potential shortcomings in our own center's discharge instructions. Interventions aimed at clarifying critical pieces of information on the discharge paperwork, such as warning symptoms, contact phone numbers and follow‐up appointments, could be especially helpful.
After discharge, our findings suggest that only about half of patients will call a physician before returning to the hospital. Furthermore, there is limited knowledge and poor utilization of same‐day treatment centers besides the emergency room. In previous studies, Long et al. found that frequently readmitted patients self‐triage to the emergency room because they believe primary care clinics cannot treat acute illness.[11] Another study concluded that low‐income patients prefer hospital care to ambulatory care because of a greater sense of trust in inpatient care.[29]
Our patients' attitudes about readmission may also be different from those of providers. For patients, coming back to the hospital is not a significant burden, and satisfaction with their medical care remains high despite readmission. Additional research is needed to further explore the complex emotions patients have when coming back to the hospital and why patients may not be as upset with returning to the hospital as providers may expect. Ultimately, if patients continue to feel more comfortable being hospitalized, there are few incentives for patients to stay out of the hospital, and readmission rates will remain elevated.
Based on our survey results we have hypothesized a potential framework for studying readmissions from a patient‐centered approach (Figure 1). This figure is not meant to imply causality, but rather to highlight a potential journey from discharge to readmission for a patient who does not feel ready to go home. This schema principally applies to patients who are worried about symptom management and/or self‐care before discharge and may not apply to everyone. Each asterisk in this framework represents an area where an intervention could be designed to improve the patient experience and possibly reduce readmissions. Such interventions should be centered around increasing patient education about symptom management and self‐care at the time of discharge, improving printed discharge instructions, increasing patient awareness of outpatient resources, enhancing communication after discharge, and changing patients' attitudes about readmissions.

This study's limitations include that it is a single‐institution study focusing on patients admitted to a large academic medical center and its partner community hospital. Only English‐speaking patients were included, and thus our results may not be generalizable to other populations. All patients were interviewed at the time of readmission, potentially introducing recall bias regarding their prior discharge. For example, patients might be more likely to state they were not ready for discharge once they have been readmitted to the hospital. Lastly, because there are only a few prior studies interviewing readmitted patients, our survey instrument was not previously validated. Nevertheless, we believe this study offers a unique view on 30‐day readmissions from the patient perspective, with a focus on identifying areas for quality‐improvement interventions.
In conclusion, this study has enabled us to understand readmissions from a patient‐centered perspective. This perspective helps to challenge provider assumptions and gives much‐needed insight into the patient experience. For example, prior to surveying patients, one might assume that if a patient has a caregiver at home, they are unlikely to have concerns about taking care of themselves. We now know this is not the case. Similarly, we have discovered sections of our discharge paperwork that are confusing. Additionally, this study has revealed that patient attitudes regarding readmission can vary significantly from provider attitudes. By exploring the patient perspective and creating a new transition framework, we have identified specific target areas for interventions that would be meaningful to patients. As the nation continues to strive to identify sustainable solutions to reduce readmissions, the way to redesign care must always start and end with the patient.
Acknowledgements
The authors acknowledge Puneet Rana, James Haggerty‐Skeans, Jae Kim, Rhea Mathew, and Anna Do (UCLA volunteers) for helping to perform the patient interviews. We acknowledge Sandy Berry, MA (Senior Behavioral Scientist at RAND Corporation) for her help in reviewing our patient interview script. Additionally Anna Dermenchyan, RN, BSN (Senior Clinical Quality Specialist in the Department of Medicine at UCLA) provided significant administrative support.
Disclosures: This project was supported by a Patient Experience Grant from the Beryl Institute awarded to Jessica Howard‐Anderson, Sarah Lonowski, Ashley Busuttil, and Nasim Afsar‐manesh. Dr. Howard‐Anderson had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All coauthors have seen and agree with the contents of the article. The article is not under review by any other publication. An earlier version of this work was written as a research report (not peer reviewed) for the Beryl Institute (available at:
- , . What will it take to move the needle on hospital readmissions? Am J Med Qual. 2013;29(4):357–359.
- , , , , , . Medicare readmission rates showed meaningful decline in 2012. Medicare Medicaid Res Rev. 2013;3(2):E1–E12.
- , , , , . Interventions to reduce 30‐day rehospitalization: a systematic review. Ann Intern Med. 2011;155(8):520–528.
- , , , et al. Preventing 30‐day hospital readmissions: a systematic review and meta‐analysis of randomized trials. JAMA Intern Med. 2014;174(7):1095–1107.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , , et al. Allocating scarce resources in real‐time to reduce heart failure readmissions: a prospective, controlled study. BMJ Qual Saf. 2013;22(12):998–1005.
- , , , , , . Readmissions in the era of patient engagement. JAMA Intern Med. 2014;174(11):1870–1872.
- , , , et al. Comparing perspectives of patients, caregivers, and clinicians on heart failure management [published online October 23, 2015]. J Card Fail. doi: 10.1016/j.cardfail.2015.10.011.
- , , , . The care transitions intervention: results of a randomized controlled trial. Arch Intern Med. 2006;166(17):1822–1828.
- , , . Understanding rehospitalization risk: can hospital discharge be modified to reduce recurrent hospitalization? J Hosp Med. 2007;2(5):297–304.
- , , . Reasons for readmission in an underserved high‐risk population: a qualitative analysis of a series of inpatient interviews. BMJ Open. 2013;3(9):e003212.
- , , , , , . Improving care transitions: the patient perspective. J Health Commun. 2012;17(suppl 3):312–324.
- , , , et al. Challenges faced by patients with low socioeconomic status during the post‐hospital transition. J Gen Intern Med. 2014;29(2):283–289.
- , , , et al. “Missing pieces”—functional, social, and environmental barriers to recovery for vulnerable older adults transitioning from hospital to home. J Am Geriatr Soc. 2014;62(8):1556–1561.
- , , , , , . Perceptions of readmitted patients on the transition from hospital to home. J Hosp Med. 2012;7(9):709–712.
- , , , et al. Factors contributing to all‐cause 30‐day readmissions: a structured case series across 18 hospitals. Med Care. 2012;50(7):599–605.
- , , . Reasons for readmission in heart failure: perspectives of patients, caregivers, cardiologists, and heart failure nurses. Heart Lung. 2009;38(5):427–434.
- , , , et al. Patient‐identified factors related to heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2013;6(2):171–177.
- , , , , . Early readmission among patients with diabetes: a qualitative assessment of contributing factors. J Diabetes Complications. 2014;28(6):869–873.
- , , , , . “Because I was sick”: seriously ill veterans' perspectives on reason for 30‐day readmissions. J Am Geriatr Soc. 2015;63(3):537–542.
- , , , et al. The impact of patient‐centered care on outcomes. J Fam Pract. 2000;49(9):796–804.
- , , , et al. Patient‐centered communication and diagnostic testing. Ann Fam Med. 2005;3(5):415–421.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , , , et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163(1):83–90.
- , , , , . Patients' participation in medical care: effects on blood sugar control and quality of life in diabetes. J Gen Intern Med. 1988;3(5):448–457.
- , , , , , . Research electronic data capture (REDCap)—a metadata‐driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.
- , , . Public reporting of discharge planning and rates of readmissions. N Engl J Med. 2009;361(27):2637–2645.
- , , , et al. Quality of discharge practices and patient understanding at an academic medical center. JAMA Intern Med. 2013;173(18):1715–1722
- , , , , , . Understanding why patients of low socioeconomic status prefer hospitals over ambulatory care. Health Aff (Millwood). 2013;32(7):1196–1203.
- , . What will it take to move the needle on hospital readmissions? Am J Med Qual. 2013;29(4):357–359.
- , , , , , . Medicare readmission rates showed meaningful decline in 2012. Medicare Medicaid Res Rev. 2013;3(2):E1–E12.
- , , , , . Interventions to reduce 30‐day rehospitalization: a systematic review. Ann Intern Med. 2011;155(8):520–528.
- , , , et al. Preventing 30‐day hospital readmissions: a systematic review and meta‐analysis of randomized trials. JAMA Intern Med. 2014;174(7):1095–1107.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , , et al. Allocating scarce resources in real‐time to reduce heart failure readmissions: a prospective, controlled study. BMJ Qual Saf. 2013;22(12):998–1005.
- , , , , , . Readmissions in the era of patient engagement. JAMA Intern Med. 2014;174(11):1870–1872.
- , , , et al. Comparing perspectives of patients, caregivers, and clinicians on heart failure management [published online October 23, 2015]. J Card Fail. doi: 10.1016/j.cardfail.2015.10.011.
- , , , . The care transitions intervention: results of a randomized controlled trial. Arch Intern Med. 2006;166(17):1822–1828.
- , , . Understanding rehospitalization risk: can hospital discharge be modified to reduce recurrent hospitalization? J Hosp Med. 2007;2(5):297–304.
- , , . Reasons for readmission in an underserved high‐risk population: a qualitative analysis of a series of inpatient interviews. BMJ Open. 2013;3(9):e003212.
- , , , , , . Improving care transitions: the patient perspective. J Health Commun. 2012;17(suppl 3):312–324.
- , , , et al. Challenges faced by patients with low socioeconomic status during the post‐hospital transition. J Gen Intern Med. 2014;29(2):283–289.
- , , , et al. “Missing pieces”—functional, social, and environmental barriers to recovery for vulnerable older adults transitioning from hospital to home. J Am Geriatr Soc. 2014;62(8):1556–1561.
- , , , , , . Perceptions of readmitted patients on the transition from hospital to home. J Hosp Med. 2012;7(9):709–712.
- , , , et al. Factors contributing to all‐cause 30‐day readmissions: a structured case series across 18 hospitals. Med Care. 2012;50(7):599–605.
- , , . Reasons for readmission in heart failure: perspectives of patients, caregivers, cardiologists, and heart failure nurses. Heart Lung. 2009;38(5):427–434.
- , , , et al. Patient‐identified factors related to heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2013;6(2):171–177.
- , , , , . Early readmission among patients with diabetes: a qualitative assessment of contributing factors. J Diabetes Complications. 2014;28(6):869–873.
- , , , , . “Because I was sick”: seriously ill veterans' perspectives on reason for 30‐day readmissions. J Am Geriatr Soc. 2015;63(3):537–542.
- , , , et al. The impact of patient‐centered care on outcomes. J Fam Pract. 2000;49(9):796–804.
- , , , et al. Patient‐centered communication and diagnostic testing. Ann Fam Med. 2005;3(5):415–421.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , , , et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163(1):83–90.
- , , , , . Patients' participation in medical care: effects on blood sugar control and quality of life in diabetes. J Gen Intern Med. 1988;3(5):448–457.
- , , , , , . Research electronic data capture (REDCap)—a metadata‐driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.
- , , . Public reporting of discharge planning and rates of readmissions. N Engl J Med. 2009;361(27):2637–2645.
- , , , et al. Quality of discharge practices and patient understanding at an academic medical center. JAMA Intern Med. 2013;173(18):1715–1722
- , , , , , . Understanding why patients of low socioeconomic status prefer hospitals over ambulatory care. Health Aff (Millwood). 2013;32(7):1196–1203.
© 2016 Society of Hospital Medicine
Emergency hernia surgery risk predicted by access, age, and race
JACKSONVILLE, FLA. – Age and access to medical care may be key drivers of emergency surgery for ventral hernia repair, a large retrospective study has found.
Patients who do not have health insurance, are advanced in age, are black or Hispanic, or have unrelated health problems are at significantly higher risk than other patients with hernias of having emergency surgery for ventral hernia repair, facing a higher risk of death, a higher cost, and a longer hospital stay, Dr. Lindsey Wolf said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress. “This study demonstrates persistent disparities in access to elective surgery care that must be understood and mitigated,” she said. “The strongest predictor was being uninsured. The self-pay group had an odds ratio of 3.5 for undergoing emergency surgery, compared with those who were primarily insured.”
The goal of the study was to identify patient and hospital factors associated with emergency ventral hernia surgery in the U.S. population, said Dr. Wolf of Brigham and Women’s Hospital in Boston. “Prior studies that have been done on predictors of emergency repair are from universally insured populations,” she said. One was a national cohort study in Denmark, and another involved the Veterans Affairs population, she said. “Both of these identified several demographic and clinical risk factors for emergency hernia repair,” she said.
The current Brigham and Women’s study involved a retrospective cross-sectional data analysis of approximately 453,000 elective and emergency ventral hernia repairs performed from 2003 to 2011 in the Nationwide Inpatient Sample. Any cases that involved a trauma diagnosis were excluded. Forty percent of the cases in the sample were emergency admissions.
When considering the effect of age, the investigators found that the aged 65-75 group had the lowest risk of emergency hernia surgery of all age groups with an odds ratio of 0.77, compared with those under 45 years. Those aged 85 and older, however, had the highest risk of all age groups with an odds ratio of 2.23. “The proportion of the cohort undergoing emergency surgery really increases drastically with age after 75 years,” Dr. Wolf said.
Other factors that had an impact on emergency hernia repair were Medicaid coverage (OR, 1.29, compared with private insurance), black race (OR, 1.64, compared with white race), Hispanic ethnicity (OR, 1.44, compared with non-Hispanic white race/ethnicity), and comorbidities, ranging from 1.13 for one comorbidity to 1.68 for three or more, compared with none.
The study also elucidated a few consequences of emergency ventral hernia repair: 2.58 times higher odds of death, a 15% greater cost per hospital stay, and 26% longer hospital stays.
“Looking forward there are both patient and provider areas to target,” Dr. Wolf said. “For patients, interventions must be designed to populations that may have poor access to elective surgical services.” She acknowledged that race was a strong predictor, “but race is a social construct that may be a proxy to many barriers to access and care.”
The study findings may also help inform surgeons on when to operate on ventral hernias. “In the absence of any clinical guidelines for when a hernia should be repaired, our results with regard to age and multiple comorbidities may assist surgeons in risk stratifying patients when considering [whether] to perform an elective repair,” she said.
Dr. Wolf and her coauthors had no relevant financial conflicts to disclose.
JACKSONVILLE, FLA. – Age and access to medical care may be key drivers of emergency surgery for ventral hernia repair, a large retrospective study has found.
Patients who do not have health insurance, are advanced in age, are black or Hispanic, or have unrelated health problems are at significantly higher risk than other patients with hernias of having emergency surgery for ventral hernia repair, facing a higher risk of death, a higher cost, and a longer hospital stay, Dr. Lindsey Wolf said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress. “This study demonstrates persistent disparities in access to elective surgery care that must be understood and mitigated,” she said. “The strongest predictor was being uninsured. The self-pay group had an odds ratio of 3.5 for undergoing emergency surgery, compared with those who were primarily insured.”
The goal of the study was to identify patient and hospital factors associated with emergency ventral hernia surgery in the U.S. population, said Dr. Wolf of Brigham and Women’s Hospital in Boston. “Prior studies that have been done on predictors of emergency repair are from universally insured populations,” she said. One was a national cohort study in Denmark, and another involved the Veterans Affairs population, she said. “Both of these identified several demographic and clinical risk factors for emergency hernia repair,” she said.
The current Brigham and Women’s study involved a retrospective cross-sectional data analysis of approximately 453,000 elective and emergency ventral hernia repairs performed from 2003 to 2011 in the Nationwide Inpatient Sample. Any cases that involved a trauma diagnosis were excluded. Forty percent of the cases in the sample were emergency admissions.
When considering the effect of age, the investigators found that the aged 65-75 group had the lowest risk of emergency hernia surgery of all age groups with an odds ratio of 0.77, compared with those under 45 years. Those aged 85 and older, however, had the highest risk of all age groups with an odds ratio of 2.23. “The proportion of the cohort undergoing emergency surgery really increases drastically with age after 75 years,” Dr. Wolf said.
Other factors that had an impact on emergency hernia repair were Medicaid coverage (OR, 1.29, compared with private insurance), black race (OR, 1.64, compared with white race), Hispanic ethnicity (OR, 1.44, compared with non-Hispanic white race/ethnicity), and comorbidities, ranging from 1.13 for one comorbidity to 1.68 for three or more, compared with none.
The study also elucidated a few consequences of emergency ventral hernia repair: 2.58 times higher odds of death, a 15% greater cost per hospital stay, and 26% longer hospital stays.
“Looking forward there are both patient and provider areas to target,” Dr. Wolf said. “For patients, interventions must be designed to populations that may have poor access to elective surgical services.” She acknowledged that race was a strong predictor, “but race is a social construct that may be a proxy to many barriers to access and care.”
The study findings may also help inform surgeons on when to operate on ventral hernias. “In the absence of any clinical guidelines for when a hernia should be repaired, our results with regard to age and multiple comorbidities may assist surgeons in risk stratifying patients when considering [whether] to perform an elective repair,” she said.
Dr. Wolf and her coauthors had no relevant financial conflicts to disclose.
JACKSONVILLE, FLA. – Age and access to medical care may be key drivers of emergency surgery for ventral hernia repair, a large retrospective study has found.
Patients who do not have health insurance, are advanced in age, are black or Hispanic, or have unrelated health problems are at significantly higher risk than other patients with hernias of having emergency surgery for ventral hernia repair, facing a higher risk of death, a higher cost, and a longer hospital stay, Dr. Lindsey Wolf said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress. “This study demonstrates persistent disparities in access to elective surgery care that must be understood and mitigated,” she said. “The strongest predictor was being uninsured. The self-pay group had an odds ratio of 3.5 for undergoing emergency surgery, compared with those who were primarily insured.”
The goal of the study was to identify patient and hospital factors associated with emergency ventral hernia surgery in the U.S. population, said Dr. Wolf of Brigham and Women’s Hospital in Boston. “Prior studies that have been done on predictors of emergency repair are from universally insured populations,” she said. One was a national cohort study in Denmark, and another involved the Veterans Affairs population, she said. “Both of these identified several demographic and clinical risk factors for emergency hernia repair,” she said.
The current Brigham and Women’s study involved a retrospective cross-sectional data analysis of approximately 453,000 elective and emergency ventral hernia repairs performed from 2003 to 2011 in the Nationwide Inpatient Sample. Any cases that involved a trauma diagnosis were excluded. Forty percent of the cases in the sample were emergency admissions.
When considering the effect of age, the investigators found that the aged 65-75 group had the lowest risk of emergency hernia surgery of all age groups with an odds ratio of 0.77, compared with those under 45 years. Those aged 85 and older, however, had the highest risk of all age groups with an odds ratio of 2.23. “The proportion of the cohort undergoing emergency surgery really increases drastically with age after 75 years,” Dr. Wolf said.
Other factors that had an impact on emergency hernia repair were Medicaid coverage (OR, 1.29, compared with private insurance), black race (OR, 1.64, compared with white race), Hispanic ethnicity (OR, 1.44, compared with non-Hispanic white race/ethnicity), and comorbidities, ranging from 1.13 for one comorbidity to 1.68 for three or more, compared with none.
The study also elucidated a few consequences of emergency ventral hernia repair: 2.58 times higher odds of death, a 15% greater cost per hospital stay, and 26% longer hospital stays.
“Looking forward there are both patient and provider areas to target,” Dr. Wolf said. “For patients, interventions must be designed to populations that may have poor access to elective surgical services.” She acknowledged that race was a strong predictor, “but race is a social construct that may be a proxy to many barriers to access and care.”
The study findings may also help inform surgeons on when to operate on ventral hernias. “In the absence of any clinical guidelines for when a hernia should be repaired, our results with regard to age and multiple comorbidities may assist surgeons in risk stratifying patients when considering [whether] to perform an elective repair,” she said.
Dr. Wolf and her coauthors had no relevant financial conflicts to disclose.
AT THE ACADEMIC SURGICAL CONGRESS
Key clinical point: Disparities among patients more likely to get emergency rather than elective ventral hernia repair include race, insurance status, and advanced age.
Major finding: Among demographic groups with a significantly higher likelihood of undergoing emergency ventral hernia repair were blacks (odds ratio, 1.64), Hispanics (OR, 1.44), and people over age 85 (OR, 2.23).
Data source: Nationwide Inpatient Sample of 453,000 adults who had inpatient ventral hernia repair from 2003 to 2011.
Disclosures: The study authors reported having no relevant financial disclosures.
ACOG pushes for contraception measures in Core Quality Measures Collaborative
Disagreement over quality measures regarding contraception has led the American College of Obstetricians and Gynecologists to withhold its imprimatur from the Core Quality Measures Collaborative.
“Although ACOG representatives did participate in the process to select measures to be included in the Core Quality Measures Collaborative process, ACOG did not choose to be recognized for participation until further agreement can be made regarding quality measures related to effective contraceptives and immediate postpartum contraception,” Dr. Barbara Levy, ACOG vice president of health policy, said in an interview. “ACOG believes that measures can help create accountability among health systems regarding contraceptive access, providing an opportunity to prevent unintended pregnancies.”
The Core Quality Measures Collaborative is lead by the Centers for Medicare & Medicaid Services and America’s Health Insurance Plans, with input from the National Quality Forum, medical societies, employer groups, and consumer groups, with the goal of building a uniform set of quality measures to be used by both public and private payers in value-based payment structures.
The first seven sets of measures under the collaborative were announced Feb. 16; ob.gyn. measures were included in this limited release.
For ob.gyn., the measures are blocked into two sets. Metrics in the ambulatory care setting look at frequency of ongoing prenatal care, cervical and breast cancer screening, chlamydia screening and follow-up, and appropriate work-up prior to endometrial ablation. Measures in the hospital/acute care setting include incidence of episiotomy, elective delivery, cesarean sections, antenatal steroids, and exclusive breastfeeding.
Four areas identified for future development include physician-level urinary incontinence screening, more on cesarean sections, Tdap/influenza administration in pregnancy, and HIV screening of STI patients.
ACOG said it will continue to push for the inclusion of contraception measures as part of the measures, particularly as access to them continues to be an issue.
“Although the Affordable Care Act requires insurance plans to cover the full range of Food and Drug Administration–approved contraceptive methods, we know that implementation of this provision has not been universal, and some women’s needs are currently unmet,” Dr. Levy said. “Because of this, ACOG continues to advocate for quality measures that will lead to meaningful improvements in access to effective contraception and immediate postpartum contraception. It is our hope that we can advance the commitment of commercial health insurance plans to promoting the most effective contraceptive methods in a way that meets the needs of more American women.”
Disagreement over quality measures regarding contraception has led the American College of Obstetricians and Gynecologists to withhold its imprimatur from the Core Quality Measures Collaborative.
“Although ACOG representatives did participate in the process to select measures to be included in the Core Quality Measures Collaborative process, ACOG did not choose to be recognized for participation until further agreement can be made regarding quality measures related to effective contraceptives and immediate postpartum contraception,” Dr. Barbara Levy, ACOG vice president of health policy, said in an interview. “ACOG believes that measures can help create accountability among health systems regarding contraceptive access, providing an opportunity to prevent unintended pregnancies.”
The Core Quality Measures Collaborative is lead by the Centers for Medicare & Medicaid Services and America’s Health Insurance Plans, with input from the National Quality Forum, medical societies, employer groups, and consumer groups, with the goal of building a uniform set of quality measures to be used by both public and private payers in value-based payment structures.
The first seven sets of measures under the collaborative were announced Feb. 16; ob.gyn. measures were included in this limited release.
For ob.gyn., the measures are blocked into two sets. Metrics in the ambulatory care setting look at frequency of ongoing prenatal care, cervical and breast cancer screening, chlamydia screening and follow-up, and appropriate work-up prior to endometrial ablation. Measures in the hospital/acute care setting include incidence of episiotomy, elective delivery, cesarean sections, antenatal steroids, and exclusive breastfeeding.
Four areas identified for future development include physician-level urinary incontinence screening, more on cesarean sections, Tdap/influenza administration in pregnancy, and HIV screening of STI patients.
ACOG said it will continue to push for the inclusion of contraception measures as part of the measures, particularly as access to them continues to be an issue.
“Although the Affordable Care Act requires insurance plans to cover the full range of Food and Drug Administration–approved contraceptive methods, we know that implementation of this provision has not been universal, and some women’s needs are currently unmet,” Dr. Levy said. “Because of this, ACOG continues to advocate for quality measures that will lead to meaningful improvements in access to effective contraception and immediate postpartum contraception. It is our hope that we can advance the commitment of commercial health insurance plans to promoting the most effective contraceptive methods in a way that meets the needs of more American women.”
Disagreement over quality measures regarding contraception has led the American College of Obstetricians and Gynecologists to withhold its imprimatur from the Core Quality Measures Collaborative.
“Although ACOG representatives did participate in the process to select measures to be included in the Core Quality Measures Collaborative process, ACOG did not choose to be recognized for participation until further agreement can be made regarding quality measures related to effective contraceptives and immediate postpartum contraception,” Dr. Barbara Levy, ACOG vice president of health policy, said in an interview. “ACOG believes that measures can help create accountability among health systems regarding contraceptive access, providing an opportunity to prevent unintended pregnancies.”
The Core Quality Measures Collaborative is lead by the Centers for Medicare & Medicaid Services and America’s Health Insurance Plans, with input from the National Quality Forum, medical societies, employer groups, and consumer groups, with the goal of building a uniform set of quality measures to be used by both public and private payers in value-based payment structures.
The first seven sets of measures under the collaborative were announced Feb. 16; ob.gyn. measures were included in this limited release.
For ob.gyn., the measures are blocked into two sets. Metrics in the ambulatory care setting look at frequency of ongoing prenatal care, cervical and breast cancer screening, chlamydia screening and follow-up, and appropriate work-up prior to endometrial ablation. Measures in the hospital/acute care setting include incidence of episiotomy, elective delivery, cesarean sections, antenatal steroids, and exclusive breastfeeding.
Four areas identified for future development include physician-level urinary incontinence screening, more on cesarean sections, Tdap/influenza administration in pregnancy, and HIV screening of STI patients.
ACOG said it will continue to push for the inclusion of contraception measures as part of the measures, particularly as access to them continues to be an issue.
“Although the Affordable Care Act requires insurance plans to cover the full range of Food and Drug Administration–approved contraceptive methods, we know that implementation of this provision has not been universal, and some women’s needs are currently unmet,” Dr. Levy said. “Because of this, ACOG continues to advocate for quality measures that will lead to meaningful improvements in access to effective contraception and immediate postpartum contraception. It is our hope that we can advance the commitment of commercial health insurance plans to promoting the most effective contraceptive methods in a way that meets the needs of more American women.”
STS: Minimizing LVAD pump thrombosis poses new challenges
PHOENIX – Cardiothoracic surgeons who implant left ventricular assist devices in patients with failing hearts remain at a loss to fully explain why they started seeing a sharp increase in thrombus clogging in these devices in 2012, but nevertheless they are gaining a better sense of how to minimize the risk.
Three key principles for minimizing thrombosis risk are selecting the right patients to receive left ventricular assist devices (LVAD), applying optimal management strategies once patients receive a LVAD, and maintaining adequate flow of blood through the pump, Dr. Francis D. Pagani said in a talk at a session devoted to pump thrombosis at the annual meeting of the Society of Thoracic Surgeons.
Other critical aspects include optimal implantation technique, quick work-up of patients to rule out reversible LVAD inflow or outflow problems once pump thrombosis is suspected, and ceasing medical therapy of the thrombosis if it proves ineffective and instead progress to surgical pump exchange, pump explantation, or heart transplant when necessary, said Dr. Ahmet Kilic, a cardiothoracic surgeon at the Ohio State University, Columbus.
Another key issue is that, now that the pump thrombosis incidence is averaging about 10% of LVAD recipients, with an incidence rate during 2-year follow-up as high as 24% reported from one series, surgeons and physicians who care for LVAD patients must have a high index of suspicion and routinely screen LVAD recipients for early signs of pump thrombosis. The best way to catch pump thrombosis early seems to be by regularly measuring patients’ serum level of lactate dehydrogenase (LDH), said Dr. Robert L. Kormos, professor of surgery and director of the artificial heart program at the University of Pittsburgh.
“We measure LDH on most clinic visits, whether or not the patient has an indication of pump thrombosis. We need to screen [LDH levels] much more routinely than we used to,” he said during the session. “Elevated LDH is probably the first and most reliable early sign, but you need to also assess LDH isoenzymes because we’ve had patients with an elevation but no sign of pump thrombosis, and their isoenzymes showed that the increased LDH was coming from their liver,” Dr. Kormos said in an interview.
Although serial measurements and isoenzyme analysis can establish a sharp rise in heart-specific LDH in an individual patient, a report at the meeting documented that in a series of 53 patients with pump thrombosis treated at either of two U.S. centers, an LDH level of at least 1,155 IU/L flagged pump thrombosis with a fairly high sensitivity and specificity. This LDH level is roughly five times the upper limit of normal, noted Dr. Pagani, professor of surgery and surgical director of adult heart transplantation at the University of Michigan, Ann Arbor, and a senior author on this report.
But prior to this report Dr. Kormos said that he regarded a LDH level of 600-800 IU/L as enough of an elevation above normal to prompt concern and investigation. And he criticized some LVAD programs that allow LDH levels to rise much higher.
“I know of clinicians who see a LDH of 1,500-2,000 IU/L but the patient seems okay and they wonder if they should change out the pump. For me, it’s a no brainer. Others try to list a patient like this for a heart transplant so they can avoid doing a pump exchange. I think that’s dangerous; it risks liver failure or renal failure. I would not sit on any LVAD that is starting to produce signs of hemolysis syndrome, but some places do this,” Dr. Kormos said in an interview.
“Pump thrombosis probably did not get addressed in as timely a fashion as it should have been” when it was first seen on the rise in 2012, noted Dr. James K. Kirklin, professor of surgery and director of cardiothoracic surgery at the University of Alabama, Birmingham. “It is now being addressed, and we realize that this is not just a pump problem but also involves patient factors and management factors that we need to learn more about. We are quite ignorant of the patient factors and understanding their contributions to bleeding and thrombosis,” said Dr. Kirklin. He also acknowledged that whatever role the current generation of LVAD pumps play in causing thrombosis will not quickly resolve.
“I’m looking forward to a new generation of pumps, but the pumps we have today will probably remain for another 3-5 years.”
The issue of LVAD pump thrombosis first came into clear focus with publication at the start of 2014 of a report that tracked its incidence from 2004 to mid-2013 at three U.S. centers that had placed a total of 895 LVADs in 837 patients. The annual rate of new episodes of pump thrombosis jumped from about 1%-2% of LVAD recipients throughout the first part of the study period through the end of 2011, to an annual rate of about 10% by mid 2013 (N Engl J Med. 2014 Jan 2;370[1]:33-40).
“The inflection occurred in about 2012,” noted Dr. Nicholas G. Smedira, a cardiothoracic surgeon at the Cleveland Clinic. “No one has figured out why” the incidence suddenly spiked starting in 2012 and intensified in 2013, he said. This epidemic of pump thrombosis has produced “devastating complications” that have led to multiple readmissions and reduced cost-effectiveness of LVADs and has affected how the heart transplant community allocates hearts, Dr. Smedira said during his talk at the session. He noted that once the surge in pump thrombosis started, the timing of the appearance of significant thrombus shifted earlier, often occurring within 2-3 months after LVAD placement. There now is “increasing device-related pessimism” and increasing demoralization among clinicians because of this recurring complication, he said.
More recent data show the trend toward increasingly higher rates of pump thrombosis continuing through the end of 2013, with the situation during 2014 a bit less clear. Late last year, data from 9,808 U.S. patients who received an LVAD and entered the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed that the incidence of pump thrombosis during the first 6 months following an implant rose from 1% in 2008 to 2% in 2009 and in 2010, 4% in 2011, 7% in 2012, 8% in 2013, and then eased back to 5% in the first half of 2014 (J Heart Lung Transplant. 2015 Dec;34[12]:1515-26). The annual rate rose from 2% in 2008 to a peak of 11% in 2013, with 12-month data from 2014 not yet available at the time of this report.
“The modest reduction of observed pump thrombosis at 6 months during 2014 has occurred in a milieu of heightened intensity of anti-coagulation management, greater surgical awareness of optimal pump implantation and positioning and pump speed management. Thus, one may speculate that current thrombosis risk-mitigation strategies have contributed to reducing but not eliminating the increased thrombosis risk observed since 2011,” concluded the authors of the report.
Surgeons and cardiologists must now have a high index of suspicion for pump thrombosis in LVAD recipients, and be especially on the lookout for four key flags of a problem, said Dr. Kormos. The first is a rising LDH level, but additional flags include an isolated power elevation that doesn’t correlate with anything else, evidence of hemolysis, and new-onset heart failure symptoms. These can occur individually or in some combination. He recommended following a diagnostic algorithm first presented in 2013 that remains very valid today (J Heart Lung Transplant. 2013 July;32[7]:667-70).
Dr. Kormos also highlighted that the presentation of pump thrombosis can differ between the two LVADs most commonly used in U.S. practice, the HeartMate II and the HeartWare devices. A LDH elevation is primarily an indicator for HeartMate II, while both that model and the HeartWare device show sustained, isolated power elevations when thrombosis occurs.
Dr. Pagani, Dr. Kirklin, and Dr. Smedira had no disclosures. Dr. Kormos has received travel support from HeartWare. Dr. Kilic has been a consultant to Thoratec and a speaker on behalf of Baxter International.
On Twitter @mitchelzoler
Dr. Hossein Almassi, FCCP, comments: With improvements in technology and development of rotary pumps, there has been a significant growth in the use of mechanical circulatory support (MCS) for treatment of end stage heart failure with a parallel improvement in patients’ survival and the quality of life.

|
| Dr. Hossein Almassi |
The authors of this report presented at the 2016 annual meeting of the STS, are authorities in the field of MCS outlining the observed increase in pump thrombosis noted in 2012. The sharp increase in the thrombosis rate is different from the lower incidence seen in the preapproval stage of the pump trial.
It should be noted that the report is related mainly to the HeatMate II left ventricular assist device (LVAD) and not the more recently implanted HeartWare device.
The diagnostic algorithm outlined in the accompanying reference (J Heart Lung Transplant. 2013 July;32[7]:667-70) regarding the diagnosis and management of suspected pump thrombosis is worth reading with the main criteria heralding a potential pump thrombosis being 1)sustained pump power elevation, 2) elevation of cardiac LDH or plasma-free hemoglobin, 3) hemolysis, and 4) symptoms of heart failure.
With further refinements in technology, the field of MCS is awaiting the development of newer LVAD devices that would mitigate the serious problem of pump thrombosis.
Dr. Hossein Almassi, FCCP, comments: With improvements in technology and development of rotary pumps, there has been a significant growth in the use of mechanical circulatory support (MCS) for treatment of end stage heart failure with a parallel improvement in patients’ survival and the quality of life.

|
| Dr. Hossein Almassi |
The authors of this report presented at the 2016 annual meeting of the STS, are authorities in the field of MCS outlining the observed increase in pump thrombosis noted in 2012. The sharp increase in the thrombosis rate is different from the lower incidence seen in the preapproval stage of the pump trial.
It should be noted that the report is related mainly to the HeatMate II left ventricular assist device (LVAD) and not the more recently implanted HeartWare device.
The diagnostic algorithm outlined in the accompanying reference (J Heart Lung Transplant. 2013 July;32[7]:667-70) regarding the diagnosis and management of suspected pump thrombosis is worth reading with the main criteria heralding a potential pump thrombosis being 1)sustained pump power elevation, 2) elevation of cardiac LDH or plasma-free hemoglobin, 3) hemolysis, and 4) symptoms of heart failure.
With further refinements in technology, the field of MCS is awaiting the development of newer LVAD devices that would mitigate the serious problem of pump thrombosis.
Dr. Hossein Almassi, FCCP, comments: With improvements in technology and development of rotary pumps, there has been a significant growth in the use of mechanical circulatory support (MCS) for treatment of end stage heart failure with a parallel improvement in patients’ survival and the quality of life.

|
| Dr. Hossein Almassi |
The authors of this report presented at the 2016 annual meeting of the STS, are authorities in the field of MCS outlining the observed increase in pump thrombosis noted in 2012. The sharp increase in the thrombosis rate is different from the lower incidence seen in the preapproval stage of the pump trial.
It should be noted that the report is related mainly to the HeatMate II left ventricular assist device (LVAD) and not the more recently implanted HeartWare device.
The diagnostic algorithm outlined in the accompanying reference (J Heart Lung Transplant. 2013 July;32[7]:667-70) regarding the diagnosis and management of suspected pump thrombosis is worth reading with the main criteria heralding a potential pump thrombosis being 1)sustained pump power elevation, 2) elevation of cardiac LDH or plasma-free hemoglobin, 3) hemolysis, and 4) symptoms of heart failure.
With further refinements in technology, the field of MCS is awaiting the development of newer LVAD devices that would mitigate the serious problem of pump thrombosis.
PHOENIX – Cardiothoracic surgeons who implant left ventricular assist devices in patients with failing hearts remain at a loss to fully explain why they started seeing a sharp increase in thrombus clogging in these devices in 2012, but nevertheless they are gaining a better sense of how to minimize the risk.
Three key principles for minimizing thrombosis risk are selecting the right patients to receive left ventricular assist devices (LVAD), applying optimal management strategies once patients receive a LVAD, and maintaining adequate flow of blood through the pump, Dr. Francis D. Pagani said in a talk at a session devoted to pump thrombosis at the annual meeting of the Society of Thoracic Surgeons.
Other critical aspects include optimal implantation technique, quick work-up of patients to rule out reversible LVAD inflow or outflow problems once pump thrombosis is suspected, and ceasing medical therapy of the thrombosis if it proves ineffective and instead progress to surgical pump exchange, pump explantation, or heart transplant when necessary, said Dr. Ahmet Kilic, a cardiothoracic surgeon at the Ohio State University, Columbus.
Another key issue is that, now that the pump thrombosis incidence is averaging about 10% of LVAD recipients, with an incidence rate during 2-year follow-up as high as 24% reported from one series, surgeons and physicians who care for LVAD patients must have a high index of suspicion and routinely screen LVAD recipients for early signs of pump thrombosis. The best way to catch pump thrombosis early seems to be by regularly measuring patients’ serum level of lactate dehydrogenase (LDH), said Dr. Robert L. Kormos, professor of surgery and director of the artificial heart program at the University of Pittsburgh.
“We measure LDH on most clinic visits, whether or not the patient has an indication of pump thrombosis. We need to screen [LDH levels] much more routinely than we used to,” he said during the session. “Elevated LDH is probably the first and most reliable early sign, but you need to also assess LDH isoenzymes because we’ve had patients with an elevation but no sign of pump thrombosis, and their isoenzymes showed that the increased LDH was coming from their liver,” Dr. Kormos said in an interview.
Although serial measurements and isoenzyme analysis can establish a sharp rise in heart-specific LDH in an individual patient, a report at the meeting documented that in a series of 53 patients with pump thrombosis treated at either of two U.S. centers, an LDH level of at least 1,155 IU/L flagged pump thrombosis with a fairly high sensitivity and specificity. This LDH level is roughly five times the upper limit of normal, noted Dr. Pagani, professor of surgery and surgical director of adult heart transplantation at the University of Michigan, Ann Arbor, and a senior author on this report.
But prior to this report Dr. Kormos said that he regarded a LDH level of 600-800 IU/L as enough of an elevation above normal to prompt concern and investigation. And he criticized some LVAD programs that allow LDH levels to rise much higher.
“I know of clinicians who see a LDH of 1,500-2,000 IU/L but the patient seems okay and they wonder if they should change out the pump. For me, it’s a no brainer. Others try to list a patient like this for a heart transplant so they can avoid doing a pump exchange. I think that’s dangerous; it risks liver failure or renal failure. I would not sit on any LVAD that is starting to produce signs of hemolysis syndrome, but some places do this,” Dr. Kormos said in an interview.
“Pump thrombosis probably did not get addressed in as timely a fashion as it should have been” when it was first seen on the rise in 2012, noted Dr. James K. Kirklin, professor of surgery and director of cardiothoracic surgery at the University of Alabama, Birmingham. “It is now being addressed, and we realize that this is not just a pump problem but also involves patient factors and management factors that we need to learn more about. We are quite ignorant of the patient factors and understanding their contributions to bleeding and thrombosis,” said Dr. Kirklin. He also acknowledged that whatever role the current generation of LVAD pumps play in causing thrombosis will not quickly resolve.
“I’m looking forward to a new generation of pumps, but the pumps we have today will probably remain for another 3-5 years.”
The issue of LVAD pump thrombosis first came into clear focus with publication at the start of 2014 of a report that tracked its incidence from 2004 to mid-2013 at three U.S. centers that had placed a total of 895 LVADs in 837 patients. The annual rate of new episodes of pump thrombosis jumped from about 1%-2% of LVAD recipients throughout the first part of the study period through the end of 2011, to an annual rate of about 10% by mid 2013 (N Engl J Med. 2014 Jan 2;370[1]:33-40).
“The inflection occurred in about 2012,” noted Dr. Nicholas G. Smedira, a cardiothoracic surgeon at the Cleveland Clinic. “No one has figured out why” the incidence suddenly spiked starting in 2012 and intensified in 2013, he said. This epidemic of pump thrombosis has produced “devastating complications” that have led to multiple readmissions and reduced cost-effectiveness of LVADs and has affected how the heart transplant community allocates hearts, Dr. Smedira said during his talk at the session. He noted that once the surge in pump thrombosis started, the timing of the appearance of significant thrombus shifted earlier, often occurring within 2-3 months after LVAD placement. There now is “increasing device-related pessimism” and increasing demoralization among clinicians because of this recurring complication, he said.
More recent data show the trend toward increasingly higher rates of pump thrombosis continuing through the end of 2013, with the situation during 2014 a bit less clear. Late last year, data from 9,808 U.S. patients who received an LVAD and entered the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed that the incidence of pump thrombosis during the first 6 months following an implant rose from 1% in 2008 to 2% in 2009 and in 2010, 4% in 2011, 7% in 2012, 8% in 2013, and then eased back to 5% in the first half of 2014 (J Heart Lung Transplant. 2015 Dec;34[12]:1515-26). The annual rate rose from 2% in 2008 to a peak of 11% in 2013, with 12-month data from 2014 not yet available at the time of this report.
“The modest reduction of observed pump thrombosis at 6 months during 2014 has occurred in a milieu of heightened intensity of anti-coagulation management, greater surgical awareness of optimal pump implantation and positioning and pump speed management. Thus, one may speculate that current thrombosis risk-mitigation strategies have contributed to reducing but not eliminating the increased thrombosis risk observed since 2011,” concluded the authors of the report.
Surgeons and cardiologists must now have a high index of suspicion for pump thrombosis in LVAD recipients, and be especially on the lookout for four key flags of a problem, said Dr. Kormos. The first is a rising LDH level, but additional flags include an isolated power elevation that doesn’t correlate with anything else, evidence of hemolysis, and new-onset heart failure symptoms. These can occur individually or in some combination. He recommended following a diagnostic algorithm first presented in 2013 that remains very valid today (J Heart Lung Transplant. 2013 July;32[7]:667-70).
Dr. Kormos also highlighted that the presentation of pump thrombosis can differ between the two LVADs most commonly used in U.S. practice, the HeartMate II and the HeartWare devices. A LDH elevation is primarily an indicator for HeartMate II, while both that model and the HeartWare device show sustained, isolated power elevations when thrombosis occurs.
Dr. Pagani, Dr. Kirklin, and Dr. Smedira had no disclosures. Dr. Kormos has received travel support from HeartWare. Dr. Kilic has been a consultant to Thoratec and a speaker on behalf of Baxter International.
On Twitter @mitchelzoler
PHOENIX – Cardiothoracic surgeons who implant left ventricular assist devices in patients with failing hearts remain at a loss to fully explain why they started seeing a sharp increase in thrombus clogging in these devices in 2012, but nevertheless they are gaining a better sense of how to minimize the risk.
Three key principles for minimizing thrombosis risk are selecting the right patients to receive left ventricular assist devices (LVAD), applying optimal management strategies once patients receive a LVAD, and maintaining adequate flow of blood through the pump, Dr. Francis D. Pagani said in a talk at a session devoted to pump thrombosis at the annual meeting of the Society of Thoracic Surgeons.
Other critical aspects include optimal implantation technique, quick work-up of patients to rule out reversible LVAD inflow or outflow problems once pump thrombosis is suspected, and ceasing medical therapy of the thrombosis if it proves ineffective and instead progress to surgical pump exchange, pump explantation, or heart transplant when necessary, said Dr. Ahmet Kilic, a cardiothoracic surgeon at the Ohio State University, Columbus.
Another key issue is that, now that the pump thrombosis incidence is averaging about 10% of LVAD recipients, with an incidence rate during 2-year follow-up as high as 24% reported from one series, surgeons and physicians who care for LVAD patients must have a high index of suspicion and routinely screen LVAD recipients for early signs of pump thrombosis. The best way to catch pump thrombosis early seems to be by regularly measuring patients’ serum level of lactate dehydrogenase (LDH), said Dr. Robert L. Kormos, professor of surgery and director of the artificial heart program at the University of Pittsburgh.
“We measure LDH on most clinic visits, whether or not the patient has an indication of pump thrombosis. We need to screen [LDH levels] much more routinely than we used to,” he said during the session. “Elevated LDH is probably the first and most reliable early sign, but you need to also assess LDH isoenzymes because we’ve had patients with an elevation but no sign of pump thrombosis, and their isoenzymes showed that the increased LDH was coming from their liver,” Dr. Kormos said in an interview.
Although serial measurements and isoenzyme analysis can establish a sharp rise in heart-specific LDH in an individual patient, a report at the meeting documented that in a series of 53 patients with pump thrombosis treated at either of two U.S. centers, an LDH level of at least 1,155 IU/L flagged pump thrombosis with a fairly high sensitivity and specificity. This LDH level is roughly five times the upper limit of normal, noted Dr. Pagani, professor of surgery and surgical director of adult heart transplantation at the University of Michigan, Ann Arbor, and a senior author on this report.
But prior to this report Dr. Kormos said that he regarded a LDH level of 600-800 IU/L as enough of an elevation above normal to prompt concern and investigation. And he criticized some LVAD programs that allow LDH levels to rise much higher.
“I know of clinicians who see a LDH of 1,500-2,000 IU/L but the patient seems okay and they wonder if they should change out the pump. For me, it’s a no brainer. Others try to list a patient like this for a heart transplant so they can avoid doing a pump exchange. I think that’s dangerous; it risks liver failure or renal failure. I would not sit on any LVAD that is starting to produce signs of hemolysis syndrome, but some places do this,” Dr. Kormos said in an interview.
“Pump thrombosis probably did not get addressed in as timely a fashion as it should have been” when it was first seen on the rise in 2012, noted Dr. James K. Kirklin, professor of surgery and director of cardiothoracic surgery at the University of Alabama, Birmingham. “It is now being addressed, and we realize that this is not just a pump problem but also involves patient factors and management factors that we need to learn more about. We are quite ignorant of the patient factors and understanding their contributions to bleeding and thrombosis,” said Dr. Kirklin. He also acknowledged that whatever role the current generation of LVAD pumps play in causing thrombosis will not quickly resolve.
“I’m looking forward to a new generation of pumps, but the pumps we have today will probably remain for another 3-5 years.”
The issue of LVAD pump thrombosis first came into clear focus with publication at the start of 2014 of a report that tracked its incidence from 2004 to mid-2013 at three U.S. centers that had placed a total of 895 LVADs in 837 patients. The annual rate of new episodes of pump thrombosis jumped from about 1%-2% of LVAD recipients throughout the first part of the study period through the end of 2011, to an annual rate of about 10% by mid 2013 (N Engl J Med. 2014 Jan 2;370[1]:33-40).
“The inflection occurred in about 2012,” noted Dr. Nicholas G. Smedira, a cardiothoracic surgeon at the Cleveland Clinic. “No one has figured out why” the incidence suddenly spiked starting in 2012 and intensified in 2013, he said. This epidemic of pump thrombosis has produced “devastating complications” that have led to multiple readmissions and reduced cost-effectiveness of LVADs and has affected how the heart transplant community allocates hearts, Dr. Smedira said during his talk at the session. He noted that once the surge in pump thrombosis started, the timing of the appearance of significant thrombus shifted earlier, often occurring within 2-3 months after LVAD placement. There now is “increasing device-related pessimism” and increasing demoralization among clinicians because of this recurring complication, he said.
More recent data show the trend toward increasingly higher rates of pump thrombosis continuing through the end of 2013, with the situation during 2014 a bit less clear. Late last year, data from 9,808 U.S. patients who received an LVAD and entered the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed that the incidence of pump thrombosis during the first 6 months following an implant rose from 1% in 2008 to 2% in 2009 and in 2010, 4% in 2011, 7% in 2012, 8% in 2013, and then eased back to 5% in the first half of 2014 (J Heart Lung Transplant. 2015 Dec;34[12]:1515-26). The annual rate rose from 2% in 2008 to a peak of 11% in 2013, with 12-month data from 2014 not yet available at the time of this report.
“The modest reduction of observed pump thrombosis at 6 months during 2014 has occurred in a milieu of heightened intensity of anti-coagulation management, greater surgical awareness of optimal pump implantation and positioning and pump speed management. Thus, one may speculate that current thrombosis risk-mitigation strategies have contributed to reducing but not eliminating the increased thrombosis risk observed since 2011,” concluded the authors of the report.
Surgeons and cardiologists must now have a high index of suspicion for pump thrombosis in LVAD recipients, and be especially on the lookout for four key flags of a problem, said Dr. Kormos. The first is a rising LDH level, but additional flags include an isolated power elevation that doesn’t correlate with anything else, evidence of hemolysis, and new-onset heart failure symptoms. These can occur individually or in some combination. He recommended following a diagnostic algorithm first presented in 2013 that remains very valid today (J Heart Lung Transplant. 2013 July;32[7]:667-70).
Dr. Kormos also highlighted that the presentation of pump thrombosis can differ between the two LVADs most commonly used in U.S. practice, the HeartMate II and the HeartWare devices. A LDH elevation is primarily an indicator for HeartMate II, while both that model and the HeartWare device show sustained, isolated power elevations when thrombosis occurs.
Dr. Pagani, Dr. Kirklin, and Dr. Smedira had no disclosures. Dr. Kormos has received travel support from HeartWare. Dr. Kilic has been a consultant to Thoratec and a speaker on behalf of Baxter International.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE STS ANNUAL MEETING
Exciting Program Set for AATS Week 2016, Register Today!
Be a part of an extraordinary week of science, sharing the latest advances in cardiothoracic surgery with colleagues from around the world -- AATS Week 2016. The week starts off with the two-day Aortic Symposium (May 12–13/New York City) followed by the 96th AATS Annual Meeting (May 14-18/Baltimore, MD). Register now!
Be a part of an extraordinary week of science, sharing the latest advances in cardiothoracic surgery with colleagues from around the world -- AATS Week 2016. The week starts off with the two-day Aortic Symposium (May 12–13/New York City) followed by the 96th AATS Annual Meeting (May 14-18/Baltimore, MD). Register now!
Be a part of an extraordinary week of science, sharing the latest advances in cardiothoracic surgery with colleagues from around the world -- AATS Week 2016. The week starts off with the two-day Aortic Symposium (May 12–13/New York City) followed by the 96th AATS Annual Meeting (May 14-18/Baltimore, MD). Register now!
End of life care
As the busy days of a long, gray winter drag on and an early season of Lent has begun, this column flows from reflections on life, death, and the promise of a new season of spring and green trees, hopefully coming soon – no matter what that groundhog saw.
Clinical ethics consultation often involves end of life (EOL) care. When I recall patients from my career, many memories are of patients who died. I trained with an intensivist who said he felt he was under less stress than the general pediatricians. While his patients were sicker, if one of his patients died, it was not unexpected. The presumption was that in the ICU everything possible had been done. General pediatricians rarely have a patient who dies. When they do, everyone, including the pediatrician, will ponder over whether something might have been caught earlier or something could have been done differently. But the most difficult problems in pediatric EOL care involve deciding when to stop aggressive care and let nature take its course.
I think the natural tendency for scholars is to approach difficult decisions by seeking more information. The delusion (which scholars can usually elucidate in great postmodern detail as long as it doesn’t apply to our own behavior) is that extensive information and deep reflection will lead to the best, most accurate, immutable, and nonambivalent declaration of goals, desires, values, and intents. To even make this delusion plausible, a sci-fi writer had to create a nonhuman species of Vulcans. But medical ethicists routinely apply this paradigm to existentially frightening EOL matters in a medical world full of prognostic uncertainty. Ethicists have recommended that this method of decision making be adopted by a population with low health literacy and a predilection to using Tinder.
For the internists, EOL care is mostly about specifying which modalities to use and which to limit. Over time, it became evident that a single check box, labeled Do Not Resuscitate yes/no, was insufficient to capture patient preferences. I recall once seeing a draft document with 49 boxes. It was clearly unwieldy. Most states that have adopted a POLST (Physician Orders for Life-Sustaining Treatment) paradigm have settled for forms with five to seven options. I think that if it were possible to capture advance directives better than that, eharmony.com, with its plethora of questions, would have already created an app for it. In adult medicine, we always can lament that the conversation about EOL care was too brief, but in my experience raising expectations of a more comprehensive discussion usually lowers the likelihood of people ever undertaking even an abbreviated conversation. In ethics, if you raise the bar high enough, people seem to walk under it.
Pediatrics at the EOL tends to focus discussion more on the goals of care, the likely outcomes, and the quality of life. These perspectives are then sometimes mapped, with poor reliability, onto the POLST paradigm designed for adults. Parents are usually at the bedside often enough to recognize their child’s suffering during aggressive care. Surrogate decision makers for adult EOL care occasionally lack that insight.
While advance directive documents are helpful, I don’t think there is a substitute for a physician motivated by empathy and a caring, involved surrogate decision maker at bedside. Providing information is not the key focus. The process should emphasize building trusting relationships, clear understanding, and reasonable expectations. I am reminded in those situations of an ancient quote: Medical care is “to cure sometimes, to relieve often, and to comfort always.”
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Dr. Powell said he had no relevant financial disclosures or conflicts of interest. E-mail him at pdnews@frontlinemedcom.com.
As the busy days of a long, gray winter drag on and an early season of Lent has begun, this column flows from reflections on life, death, and the promise of a new season of spring and green trees, hopefully coming soon – no matter what that groundhog saw.
Clinical ethics consultation often involves end of life (EOL) care. When I recall patients from my career, many memories are of patients who died. I trained with an intensivist who said he felt he was under less stress than the general pediatricians. While his patients were sicker, if one of his patients died, it was not unexpected. The presumption was that in the ICU everything possible had been done. General pediatricians rarely have a patient who dies. When they do, everyone, including the pediatrician, will ponder over whether something might have been caught earlier or something could have been done differently. But the most difficult problems in pediatric EOL care involve deciding when to stop aggressive care and let nature take its course.
I think the natural tendency for scholars is to approach difficult decisions by seeking more information. The delusion (which scholars can usually elucidate in great postmodern detail as long as it doesn’t apply to our own behavior) is that extensive information and deep reflection will lead to the best, most accurate, immutable, and nonambivalent declaration of goals, desires, values, and intents. To even make this delusion plausible, a sci-fi writer had to create a nonhuman species of Vulcans. But medical ethicists routinely apply this paradigm to existentially frightening EOL matters in a medical world full of prognostic uncertainty. Ethicists have recommended that this method of decision making be adopted by a population with low health literacy and a predilection to using Tinder.
For the internists, EOL care is mostly about specifying which modalities to use and which to limit. Over time, it became evident that a single check box, labeled Do Not Resuscitate yes/no, was insufficient to capture patient preferences. I recall once seeing a draft document with 49 boxes. It was clearly unwieldy. Most states that have adopted a POLST (Physician Orders for Life-Sustaining Treatment) paradigm have settled for forms with five to seven options. I think that if it were possible to capture advance directives better than that, eharmony.com, with its plethora of questions, would have already created an app for it. In adult medicine, we always can lament that the conversation about EOL care was too brief, but in my experience raising expectations of a more comprehensive discussion usually lowers the likelihood of people ever undertaking even an abbreviated conversation. In ethics, if you raise the bar high enough, people seem to walk under it.
Pediatrics at the EOL tends to focus discussion more on the goals of care, the likely outcomes, and the quality of life. These perspectives are then sometimes mapped, with poor reliability, onto the POLST paradigm designed for adults. Parents are usually at the bedside often enough to recognize their child’s suffering during aggressive care. Surrogate decision makers for adult EOL care occasionally lack that insight.
While advance directive documents are helpful, I don’t think there is a substitute for a physician motivated by empathy and a caring, involved surrogate decision maker at bedside. Providing information is not the key focus. The process should emphasize building trusting relationships, clear understanding, and reasonable expectations. I am reminded in those situations of an ancient quote: Medical care is “to cure sometimes, to relieve often, and to comfort always.”
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Dr. Powell said he had no relevant financial disclosures or conflicts of interest. E-mail him at pdnews@frontlinemedcom.com.
As the busy days of a long, gray winter drag on and an early season of Lent has begun, this column flows from reflections on life, death, and the promise of a new season of spring and green trees, hopefully coming soon – no matter what that groundhog saw.
Clinical ethics consultation often involves end of life (EOL) care. When I recall patients from my career, many memories are of patients who died. I trained with an intensivist who said he felt he was under less stress than the general pediatricians. While his patients were sicker, if one of his patients died, it was not unexpected. The presumption was that in the ICU everything possible had been done. General pediatricians rarely have a patient who dies. When they do, everyone, including the pediatrician, will ponder over whether something might have been caught earlier or something could have been done differently. But the most difficult problems in pediatric EOL care involve deciding when to stop aggressive care and let nature take its course.
I think the natural tendency for scholars is to approach difficult decisions by seeking more information. The delusion (which scholars can usually elucidate in great postmodern detail as long as it doesn’t apply to our own behavior) is that extensive information and deep reflection will lead to the best, most accurate, immutable, and nonambivalent declaration of goals, desires, values, and intents. To even make this delusion plausible, a sci-fi writer had to create a nonhuman species of Vulcans. But medical ethicists routinely apply this paradigm to existentially frightening EOL matters in a medical world full of prognostic uncertainty. Ethicists have recommended that this method of decision making be adopted by a population with low health literacy and a predilection to using Tinder.
For the internists, EOL care is mostly about specifying which modalities to use and which to limit. Over time, it became evident that a single check box, labeled Do Not Resuscitate yes/no, was insufficient to capture patient preferences. I recall once seeing a draft document with 49 boxes. It was clearly unwieldy. Most states that have adopted a POLST (Physician Orders for Life-Sustaining Treatment) paradigm have settled for forms with five to seven options. I think that if it were possible to capture advance directives better than that, eharmony.com, with its plethora of questions, would have already created an app for it. In adult medicine, we always can lament that the conversation about EOL care was too brief, but in my experience raising expectations of a more comprehensive discussion usually lowers the likelihood of people ever undertaking even an abbreviated conversation. In ethics, if you raise the bar high enough, people seem to walk under it.
Pediatrics at the EOL tends to focus discussion more on the goals of care, the likely outcomes, and the quality of life. These perspectives are then sometimes mapped, with poor reliability, onto the POLST paradigm designed for adults. Parents are usually at the bedside often enough to recognize their child’s suffering during aggressive care. Surrogate decision makers for adult EOL care occasionally lack that insight.
While advance directive documents are helpful, I don’t think there is a substitute for a physician motivated by empathy and a caring, involved surrogate decision maker at bedside. Providing information is not the key focus. The process should emphasize building trusting relationships, clear understanding, and reasonable expectations. I am reminded in those situations of an ancient quote: Medical care is “to cure sometimes, to relieve often, and to comfort always.”
Dr. Powell is a pediatric hospitalist and clinical ethics consultant living in St. Louis. Dr. Powell said he had no relevant financial disclosures or conflicts of interest. E-mail him at pdnews@frontlinemedcom.com.
Cryo-Compression Therapy
CoolSystems, Inc. (www.gameready.com)
The Game Ready Injury Treatment System
Peter Millett, MD, The Steadman Clinic, Vail, CO; Consultant, Major League Baseball Players’ Association
At the Steadman Clinic, we have developed best-practice techniques and protocols to accelerate our patients’ recoveries. Game Ready helps my patients recover faster. The Game Ready device has the most advanced level of rehab technology with the cost-effective cryotherapy delivery system, intermittent compression, and ergonomically designed wraps tailored for specific areas of the body. It just works better than ice alone or other cryotherapy devices. Game Ready reduces swelling and gets patients back faster.
I prescribe Game Ready after surgical procedures because it decreases pain, reduces the need for pain medication, and results in a faster recovery. For my overhead athletes, I routinely use the shoulder and elbow wraps for labral tears, shoulder instability, biceps tendon disorders, and rotator cuff problems.
J.W. Thomas Byrd, MD, Nashville Sports Medicine and Orthopaedics, Orthopaedic Surgical Consultant, various Major League Baseball Clubs
Performing hip arthroscopy procedures for Major League Baseball pitchers over the last 3 decades, I have come to realize the importance of choosing the most effective recovery therapy device. We have trialed numerous products and found the Game Ready cold-intermittent-compression device to be an incredible asset in the recovery and pain management strategy.
During the rehab process, pain control is essential to the athlete’s ability to participate and achieve optimal recovery. Hip procedures can be painful because they usually revolve around restoring the acetabular labrum, which is richly innervated with nociceptive fibers. In order to control discomfort following surgery, regional anesthetic nerve blocks are sometimes necessary. However, these blocks can hinder an athlete’s ability to participate in, and benefit from, the early postoperative rehabilitation process. Applying the Game Ready led to a noticeable drop in postoperative pain, obviating the need for a block.
Kenneth Akizuki, MD, SOAR, San Francisco, CA, Team Physician, San Francisco Giants
Among pro players, Tommy John surgery is a common procedure. The day after surgery, we start the player on the Game Ready system to relieve pain and quickly control swelling. We typically start with cold therapy, then add compression about a week in, and use it throughout recovery.
The players love the comfort of the ergonomic wrap designs and I really like the flexed elbow wrap. The cold is adjustable so we don’t get overcooling, and the wrap design keeps the surgery site dry, which cuts the risk of infection. The pre-set treatment programs are another big advantage. They take the hassle out of application. Whether a professional athlete or not, all our patients want convenience, and we want to see progress. Progress is motivating, it encourages compliance—and that improves outcomes.
CoolSystems, Inc. (www.gameready.com)
The Game Ready Injury Treatment System
Peter Millett, MD, The Steadman Clinic, Vail, CO; Consultant, Major League Baseball Players’ Association
At the Steadman Clinic, we have developed best-practice techniques and protocols to accelerate our patients’ recoveries. Game Ready helps my patients recover faster. The Game Ready device has the most advanced level of rehab technology with the cost-effective cryotherapy delivery system, intermittent compression, and ergonomically designed wraps tailored for specific areas of the body. It just works better than ice alone or other cryotherapy devices. Game Ready reduces swelling and gets patients back faster.
I prescribe Game Ready after surgical procedures because it decreases pain, reduces the need for pain medication, and results in a faster recovery. For my overhead athletes, I routinely use the shoulder and elbow wraps for labral tears, shoulder instability, biceps tendon disorders, and rotator cuff problems.
J.W. Thomas Byrd, MD, Nashville Sports Medicine and Orthopaedics, Orthopaedic Surgical Consultant, various Major League Baseball Clubs
Performing hip arthroscopy procedures for Major League Baseball pitchers over the last 3 decades, I have come to realize the importance of choosing the most effective recovery therapy device. We have trialed numerous products and found the Game Ready cold-intermittent-compression device to be an incredible asset in the recovery and pain management strategy.
During the rehab process, pain control is essential to the athlete’s ability to participate and achieve optimal recovery. Hip procedures can be painful because they usually revolve around restoring the acetabular labrum, which is richly innervated with nociceptive fibers. In order to control discomfort following surgery, regional anesthetic nerve blocks are sometimes necessary. However, these blocks can hinder an athlete’s ability to participate in, and benefit from, the early postoperative rehabilitation process. Applying the Game Ready led to a noticeable drop in postoperative pain, obviating the need for a block.
Kenneth Akizuki, MD, SOAR, San Francisco, CA, Team Physician, San Francisco Giants
Among pro players, Tommy John surgery is a common procedure. The day after surgery, we start the player on the Game Ready system to relieve pain and quickly control swelling. We typically start with cold therapy, then add compression about a week in, and use it throughout recovery.
The players love the comfort of the ergonomic wrap designs and I really like the flexed elbow wrap. The cold is adjustable so we don’t get overcooling, and the wrap design keeps the surgery site dry, which cuts the risk of infection. The pre-set treatment programs are another big advantage. They take the hassle out of application. Whether a professional athlete or not, all our patients want convenience, and we want to see progress. Progress is motivating, it encourages compliance—and that improves outcomes.
CoolSystems, Inc. (www.gameready.com)
The Game Ready Injury Treatment System
Peter Millett, MD, The Steadman Clinic, Vail, CO; Consultant, Major League Baseball Players’ Association
At the Steadman Clinic, we have developed best-practice techniques and protocols to accelerate our patients’ recoveries. Game Ready helps my patients recover faster. The Game Ready device has the most advanced level of rehab technology with the cost-effective cryotherapy delivery system, intermittent compression, and ergonomically designed wraps tailored for specific areas of the body. It just works better than ice alone or other cryotherapy devices. Game Ready reduces swelling and gets patients back faster.
I prescribe Game Ready after surgical procedures because it decreases pain, reduces the need for pain medication, and results in a faster recovery. For my overhead athletes, I routinely use the shoulder and elbow wraps for labral tears, shoulder instability, biceps tendon disorders, and rotator cuff problems.
J.W. Thomas Byrd, MD, Nashville Sports Medicine and Orthopaedics, Orthopaedic Surgical Consultant, various Major League Baseball Clubs
Performing hip arthroscopy procedures for Major League Baseball pitchers over the last 3 decades, I have come to realize the importance of choosing the most effective recovery therapy device. We have trialed numerous products and found the Game Ready cold-intermittent-compression device to be an incredible asset in the recovery and pain management strategy.
During the rehab process, pain control is essential to the athlete’s ability to participate and achieve optimal recovery. Hip procedures can be painful because they usually revolve around restoring the acetabular labrum, which is richly innervated with nociceptive fibers. In order to control discomfort following surgery, regional anesthetic nerve blocks are sometimes necessary. However, these blocks can hinder an athlete’s ability to participate in, and benefit from, the early postoperative rehabilitation process. Applying the Game Ready led to a noticeable drop in postoperative pain, obviating the need for a block.
Kenneth Akizuki, MD, SOAR, San Francisco, CA, Team Physician, San Francisco Giants
Among pro players, Tommy John surgery is a common procedure. The day after surgery, we start the player on the Game Ready system to relieve pain and quickly control swelling. We typically start with cold therapy, then add compression about a week in, and use it throughout recovery.
The players love the comfort of the ergonomic wrap designs and I really like the flexed elbow wrap. The cold is adjustable so we don’t get overcooling, and the wrap design keeps the surgery site dry, which cuts the risk of infection. The pre-set treatment programs are another big advantage. They take the hassle out of application. Whether a professional athlete or not, all our patients want convenience, and we want to see progress. Progress is motivating, it encourages compliance—and that improves outcomes.
All-Inside Meniscal Repair Devices
Tools of the Trade features reviews of the hottest new products, with surgical pearls written by surgeons who know these products best.
Arthrex, Inc. (http://www.arthrex.com)
Knee Scorpion
The Arthrex Knee Scorpion allows for simple passage of suture at the root to repair the tissue. The mechanism of the Knee Scorpion self-retrieving the suture after passage eliminates the need for another step in the procedure, which saves time. There are various types of suture configurations that can be incorporated into meniscus repairs with the Knee Scorpion. Depending on tissue quality and location, I will either pass the center of the suture to create a cinch or luggage tag type of stitch, or sometimes a simple stitch.
A challenging meniscal tear pattern that repair is particularly made easier by use of the Knee Scorpion is the radial tear of the lateral meniscus. Here it is easy to place a side-to-side “spanning” circumferential suture pattern, which is the strongest suture configuration, and it reduces the tissue very well. This approach also is ideal for variant root type tears, those that are 3 to 4 mm from the root commonly seen with the lateral meniscus associated with anterior cruciate ligament tears.
Benefits: The Knee Scorpion has a very low profile to facilitate placement in the joint with a 5º upcurve to avoid injury to the femoral condyle. The needle captures the suture after passage, which saves time and an additional surgical step. It can be used with both 0 FiberWire and 2-0 FiberWire.
Surgical pearl: Using a cannula through the working portal prevents any tissue bridge when passing and tying sutures. I prefer a Passport button cannula since its flexibility allows more access in different planes compared to a rigid type cannula.
Patrick A. Smith, MD, Columbia Orthopaedic Group, Head Section of Sports Medicine University of Missouri, Head Team Physician University of Missouri
Arthrex, Inc. (http://www.arthrex.com)
SpeedCinch
The SpeedCinch’s pistol grip design permits ergonomic, one-handed all-inside meniscal repair. It is best suited for meniscal tears of the posterior horn and body. The posterior horn of the meniscus can be repaired with the SpeedCinch inserted through either the ipsilateral or contralateral portal. Meniscal body tears, however, are best approached via a contralateral portal. The end of the device contains a 15g needle that should be advanced across the meniscus. After passage of the needle, the first implant is pushed through the needle when the trigger is fully deployed.
Next, the SpeedCinch needle should be moved at least 1 cm for placement of the second implant in either a horizontal or vertical mattress fashion. After the needle is brought to the second insertion site, the implant selector button is moved to 2 and the trigger is depressed until the first click is felt and heard. Next, the trigger is held in place after the first click and the needle is advanced across the meniscus before fully depressing the trigger, which will advance the second implant. The device is then removed and the pre-tied knot is secured with a knot pusher and then cut.
Vic Goradia, MD, G2 Orthopedics and Sports Medicine, Glen Allen, VA
Cayenne Medical, Inc. (www.cayennemedical.com)
The CrossFix II System
The CrossFix II System offers a unique, all-arthroscopic, suture-only meniscal repair that reduces the risk of chondral injury. Its “all-inside” technique uses 2 parallel suture delivery needles, available in both curved and straight designs, inserted through a single incision to provide an all-inside meniscus repair. This simple procedure can be performed in minutes via the device’s pre-tied, sliding knot, creating a “suture only” mattress stitch that replicates the repair of standard open suturing techniques. The speed and reproducibility of the repair dramatically improves operating room efficiency. Even complex tears requiring multiple sutures can be repaired in minutes.
Benefits: All-suture, no implants; single insertion; pre-tied, sliding knot. Parallel needles may be difficult to use in very tight knees.
Surgical pearl: Avoid torqueing the needles upon insertion into the meniscus.
Kenneth Montgomery, MD, Tri-County Orthopedics, Cedar Knolls, NJ.
Ceterix Orthopaedics, Inc. (www.ceterix.com)
NovoStitch Plus
The Ceterix NovoStitch Plus enables surgeons to place circumferential compression stitches around meniscus tears. These stitch patterns are designed to provide anatomical reduction and uniform compression of the tear edges, and repair the femoral surface (top) and tibial surface (bottom) of the tear with each stitch. It is also designed to eliminate neurovascular risk and avoid excessive entrapment or extrusion of the meniscus to capsule. The NovoStitch Plus passes circumferential stitches via an all-inside arthroscopic technique. In addition to anatomically repairing vertical tears, circumferential stitches also enable repair of tear types that were previously considered difficult to adequately sew.
Benefits: Passes both sides of the stitch with 1 insertion (avoids tissue bridges and girth hitches). Retractable lower jaw (allows access to tight knees, and allows removal from knees with mobile menisci). Needle extends into the posterior gutter through the tip of the lower jaw (does not touch chondral surfaces). Smaller lower jaw tooth (easier insertion of the lower jaw under the meniscus). Upper jaw curved to follow the shape contour of the femoral side of the meniscus and femoral condyle. Compared to hybrid all-inside devices, inside-out and outside-in repairs. Enables anatomical reduction and uniform compression. Each stitch repairs the top and bottom of the tear. Designed to eliminate neurovascular risk. Avoids excessive capsular entrapment. Repairs effectively in front of the popliteal hiatus. Enables side-to-side radial tear repair, and top-to-bottom horizontal tear repair. Significantly smaller needle than used by hybrid all-inside devices.
Surgical pearl: A spinal needle should be used to establish the skin incision location of the working portal. The optimal skin incision location is one where a spinal needle can be inserted such that the distal end of the needle is parallel to the region of the tibial plateau under the meniscus.
Justin Saliman, MD, Cedars-Sinai Orthopaedic Center, Los Angeles CA
Mitek Sports Medicine (www.depuysynthes.com)
Mitek Omnispan
The Mitek Omnispan Meniscal Repair System is an all-inside arthroscopic meniscal repair device. It consists of a low profile needle, pre-loaded with 2 PEEK backstops and No. 2/0 Orthocord High Strength Orthopaedic Suture, which is delivered using the Omnispan System Applier. The needles come in 3 angles (0°, 12°, and 27°) and the Applier is a single-patient, multi-use design, meaning the same Applier can be used for multiple implants with a single patient. The needles also have a safety sleeve over each set of implants to prevent delivery failure. Once the needle is attached to the Applier, the device is inserted into the knee. Using the gray trigger, the first implant is delivered into the meniscocapsular region. The red trigger is then pulled, advancing the second implant. Once appropriate positioning of the second implant is determined, the gray trigger is fired again. The surgeon pulls on the sliding knot advancing the suture through the implants creating a suture bridge over the tear. The final repair consists of Orthocord Suture spanning the meniscus, while the PEEK backstops are embedded in the meniscocapsular area only.
Surgical pearls:
An adequate portal opening is paramount for successful implementation. I widen my portal with an instrument clamp to allow easy passage of the Applier. It is usually necessary to switch the camera back and forth between both portals to gain access to the angles needed to create the repair. I also always use a skid when inserting the Applier to prevent soft tissue impingement.
The surgeon should always check to make sure the implant is properly loaded on the back table, ensuring it is fully inserted into the locking mechanism and loaded in the correct direction.
The surgeon should hold the Applier firmly when firing the gray trigger, as some kick-back does occur.
The Applier should remain in the joint throughout the advancing and deployment of implants, so the surgeon can maintain visualization with the scope. Once the implants are in position, I then remove the Applier from the joint space.
Use a probe to place counter-pressure on the meniscus when tightening the suture. The sliding suture system is a double-loop design. In order to smoothly advance the suture, I recommend that a probe be placed on the smaller loop closest to the meniscus, so that when the suture is tightened, the probe is under the smaller loop. A gentle back-and-forth pulling maneuver between the free suture and the probe creates a smooth transition during tightening.
Scott A. Sigman, MD, Chief of Orthopedics, Lowell General Hospital; Team Physician, UMass Lowell
Smith & Nephew Inc. (www.smith-nephew.com)
FAST-FIX 360
The FAST-FIX360 design enables you to deploy implants in any hand position—vertically, horizontally on either side of the meniscus—with a fast, smooth advancing motion. This spring-action design facilitates the advancement of each implant into the capsule. Smaller implants and pre-tied, self-sliding knots made of ULTRABRAID 2-0 Suture create smaller needle insertions, reducing disruption to the meniscus. Low-profile, stiffer needle shaft improves control while enabling access and visibility to hard-to-reach areas of the meniscus. Set needle depth penetration from 10 mm to 18 mm with the push of a button. The FAST-FIX 360 System has biomechanical properties that best reproduce the vertical mattress suture technique.
Surgical pearl: Assess tear pattern and reparability, then reduce and template repair construct and suture points. Precisely select portal insertion sites, ensuring perpendicular vector needle delivery at the repair site. Measure the meniscal fragment size and rim width, setting the depth penetration limiter (usually set to 15 to 18 mm). Insert first anchor posteriorly and superiorly or away from the insertion portal and then after deployment, insert the second anchor anteriorly and inferiorly or into the “near” tear fragment. Tension the suture using the knot pusher/cutter as a “suture stent” to manually “pull and push” the suture, compressing the repair construct and coapting the tear. Avoid “over-repairing” the tear by spacing out sutures at 3 mm to 5 mm and alternating femoral and tibial undersurface placement.
Nicholas A. Sgaglione, MD (pictured), and Ryan A. Harrell, DO
Tools of the Trade features reviews of the hottest new products, with surgical pearls written by surgeons who know these products best.
Arthrex, Inc. (http://www.arthrex.com)
Knee Scorpion
The Arthrex Knee Scorpion allows for simple passage of suture at the root to repair the tissue. The mechanism of the Knee Scorpion self-retrieving the suture after passage eliminates the need for another step in the procedure, which saves time. There are various types of suture configurations that can be incorporated into meniscus repairs with the Knee Scorpion. Depending on tissue quality and location, I will either pass the center of the suture to create a cinch or luggage tag type of stitch, or sometimes a simple stitch.
A challenging meniscal tear pattern that repair is particularly made easier by use of the Knee Scorpion is the radial tear of the lateral meniscus. Here it is easy to place a side-to-side “spanning” circumferential suture pattern, which is the strongest suture configuration, and it reduces the tissue very well. This approach also is ideal for variant root type tears, those that are 3 to 4 mm from the root commonly seen with the lateral meniscus associated with anterior cruciate ligament tears.
Benefits: The Knee Scorpion has a very low profile to facilitate placement in the joint with a 5º upcurve to avoid injury to the femoral condyle. The needle captures the suture after passage, which saves time and an additional surgical step. It can be used with both 0 FiberWire and 2-0 FiberWire.
Surgical pearl: Using a cannula through the working portal prevents any tissue bridge when passing and tying sutures. I prefer a Passport button cannula since its flexibility allows more access in different planes compared to a rigid type cannula.
Patrick A. Smith, MD, Columbia Orthopaedic Group, Head Section of Sports Medicine University of Missouri, Head Team Physician University of Missouri
Arthrex, Inc. (http://www.arthrex.com)
SpeedCinch
The SpeedCinch’s pistol grip design permits ergonomic, one-handed all-inside meniscal repair. It is best suited for meniscal tears of the posterior horn and body. The posterior horn of the meniscus can be repaired with the SpeedCinch inserted through either the ipsilateral or contralateral portal. Meniscal body tears, however, are best approached via a contralateral portal. The end of the device contains a 15g needle that should be advanced across the meniscus. After passage of the needle, the first implant is pushed through the needle when the trigger is fully deployed.
Next, the SpeedCinch needle should be moved at least 1 cm for placement of the second implant in either a horizontal or vertical mattress fashion. After the needle is brought to the second insertion site, the implant selector button is moved to 2 and the trigger is depressed until the first click is felt and heard. Next, the trigger is held in place after the first click and the needle is advanced across the meniscus before fully depressing the trigger, which will advance the second implant. The device is then removed and the pre-tied knot is secured with a knot pusher and then cut.
Vic Goradia, MD, G2 Orthopedics and Sports Medicine, Glen Allen, VA
Cayenne Medical, Inc. (www.cayennemedical.com)
The CrossFix II System
The CrossFix II System offers a unique, all-arthroscopic, suture-only meniscal repair that reduces the risk of chondral injury. Its “all-inside” technique uses 2 parallel suture delivery needles, available in both curved and straight designs, inserted through a single incision to provide an all-inside meniscus repair. This simple procedure can be performed in minutes via the device’s pre-tied, sliding knot, creating a “suture only” mattress stitch that replicates the repair of standard open suturing techniques. The speed and reproducibility of the repair dramatically improves operating room efficiency. Even complex tears requiring multiple sutures can be repaired in minutes.
Benefits: All-suture, no implants; single insertion; pre-tied, sliding knot. Parallel needles may be difficult to use in very tight knees.
Surgical pearl: Avoid torqueing the needles upon insertion into the meniscus.
Kenneth Montgomery, MD, Tri-County Orthopedics, Cedar Knolls, NJ.
Ceterix Orthopaedics, Inc. (www.ceterix.com)
NovoStitch Plus
The Ceterix NovoStitch Plus enables surgeons to place circumferential compression stitches around meniscus tears. These stitch patterns are designed to provide anatomical reduction and uniform compression of the tear edges, and repair the femoral surface (top) and tibial surface (bottom) of the tear with each stitch. It is also designed to eliminate neurovascular risk and avoid excessive entrapment or extrusion of the meniscus to capsule. The NovoStitch Plus passes circumferential stitches via an all-inside arthroscopic technique. In addition to anatomically repairing vertical tears, circumferential stitches also enable repair of tear types that were previously considered difficult to adequately sew.
Benefits: Passes both sides of the stitch with 1 insertion (avoids tissue bridges and girth hitches). Retractable lower jaw (allows access to tight knees, and allows removal from knees with mobile menisci). Needle extends into the posterior gutter through the tip of the lower jaw (does not touch chondral surfaces). Smaller lower jaw tooth (easier insertion of the lower jaw under the meniscus). Upper jaw curved to follow the shape contour of the femoral side of the meniscus and femoral condyle. Compared to hybrid all-inside devices, inside-out and outside-in repairs. Enables anatomical reduction and uniform compression. Each stitch repairs the top and bottom of the tear. Designed to eliminate neurovascular risk. Avoids excessive capsular entrapment. Repairs effectively in front of the popliteal hiatus. Enables side-to-side radial tear repair, and top-to-bottom horizontal tear repair. Significantly smaller needle than used by hybrid all-inside devices.
Surgical pearl: A spinal needle should be used to establish the skin incision location of the working portal. The optimal skin incision location is one where a spinal needle can be inserted such that the distal end of the needle is parallel to the region of the tibial plateau under the meniscus.
Justin Saliman, MD, Cedars-Sinai Orthopaedic Center, Los Angeles CA
Mitek Sports Medicine (www.depuysynthes.com)
Mitek Omnispan
The Mitek Omnispan Meniscal Repair System is an all-inside arthroscopic meniscal repair device. It consists of a low profile needle, pre-loaded with 2 PEEK backstops and No. 2/0 Orthocord High Strength Orthopaedic Suture, which is delivered using the Omnispan System Applier. The needles come in 3 angles (0°, 12°, and 27°) and the Applier is a single-patient, multi-use design, meaning the same Applier can be used for multiple implants with a single patient. The needles also have a safety sleeve over each set of implants to prevent delivery failure. Once the needle is attached to the Applier, the device is inserted into the knee. Using the gray trigger, the first implant is delivered into the meniscocapsular region. The red trigger is then pulled, advancing the second implant. Once appropriate positioning of the second implant is determined, the gray trigger is fired again. The surgeon pulls on the sliding knot advancing the suture through the implants creating a suture bridge over the tear. The final repair consists of Orthocord Suture spanning the meniscus, while the PEEK backstops are embedded in the meniscocapsular area only.
Surgical pearls:
An adequate portal opening is paramount for successful implementation. I widen my portal with an instrument clamp to allow easy passage of the Applier. It is usually necessary to switch the camera back and forth between both portals to gain access to the angles needed to create the repair. I also always use a skid when inserting the Applier to prevent soft tissue impingement.
The surgeon should always check to make sure the implant is properly loaded on the back table, ensuring it is fully inserted into the locking mechanism and loaded in the correct direction.
The surgeon should hold the Applier firmly when firing the gray trigger, as some kick-back does occur.
The Applier should remain in the joint throughout the advancing and deployment of implants, so the surgeon can maintain visualization with the scope. Once the implants are in position, I then remove the Applier from the joint space.
Use a probe to place counter-pressure on the meniscus when tightening the suture. The sliding suture system is a double-loop design. In order to smoothly advance the suture, I recommend that a probe be placed on the smaller loop closest to the meniscus, so that when the suture is tightened, the probe is under the smaller loop. A gentle back-and-forth pulling maneuver between the free suture and the probe creates a smooth transition during tightening.
Scott A. Sigman, MD, Chief of Orthopedics, Lowell General Hospital; Team Physician, UMass Lowell
Smith & Nephew Inc. (www.smith-nephew.com)
FAST-FIX 360
The FAST-FIX360 design enables you to deploy implants in any hand position—vertically, horizontally on either side of the meniscus—with a fast, smooth advancing motion. This spring-action design facilitates the advancement of each implant into the capsule. Smaller implants and pre-tied, self-sliding knots made of ULTRABRAID 2-0 Suture create smaller needle insertions, reducing disruption to the meniscus. Low-profile, stiffer needle shaft improves control while enabling access and visibility to hard-to-reach areas of the meniscus. Set needle depth penetration from 10 mm to 18 mm with the push of a button. The FAST-FIX 360 System has biomechanical properties that best reproduce the vertical mattress suture technique.
Surgical pearl: Assess tear pattern and reparability, then reduce and template repair construct and suture points. Precisely select portal insertion sites, ensuring perpendicular vector needle delivery at the repair site. Measure the meniscal fragment size and rim width, setting the depth penetration limiter (usually set to 15 to 18 mm). Insert first anchor posteriorly and superiorly or away from the insertion portal and then after deployment, insert the second anchor anteriorly and inferiorly or into the “near” tear fragment. Tension the suture using the knot pusher/cutter as a “suture stent” to manually “pull and push” the suture, compressing the repair construct and coapting the tear. Avoid “over-repairing” the tear by spacing out sutures at 3 mm to 5 mm and alternating femoral and tibial undersurface placement.
Nicholas A. Sgaglione, MD (pictured), and Ryan A. Harrell, DO
Tools of the Trade features reviews of the hottest new products, with surgical pearls written by surgeons who know these products best.
Arthrex, Inc. (http://www.arthrex.com)
Knee Scorpion
The Arthrex Knee Scorpion allows for simple passage of suture at the root to repair the tissue. The mechanism of the Knee Scorpion self-retrieving the suture after passage eliminates the need for another step in the procedure, which saves time. There are various types of suture configurations that can be incorporated into meniscus repairs with the Knee Scorpion. Depending on tissue quality and location, I will either pass the center of the suture to create a cinch or luggage tag type of stitch, or sometimes a simple stitch.
A challenging meniscal tear pattern that repair is particularly made easier by use of the Knee Scorpion is the radial tear of the lateral meniscus. Here it is easy to place a side-to-side “spanning” circumferential suture pattern, which is the strongest suture configuration, and it reduces the tissue very well. This approach also is ideal for variant root type tears, those that are 3 to 4 mm from the root commonly seen with the lateral meniscus associated with anterior cruciate ligament tears.
Benefits: The Knee Scorpion has a very low profile to facilitate placement in the joint with a 5º upcurve to avoid injury to the femoral condyle. The needle captures the suture after passage, which saves time and an additional surgical step. It can be used with both 0 FiberWire and 2-0 FiberWire.
Surgical pearl: Using a cannula through the working portal prevents any tissue bridge when passing and tying sutures. I prefer a Passport button cannula since its flexibility allows more access in different planes compared to a rigid type cannula.
Patrick A. Smith, MD, Columbia Orthopaedic Group, Head Section of Sports Medicine University of Missouri, Head Team Physician University of Missouri
Arthrex, Inc. (http://www.arthrex.com)
SpeedCinch
The SpeedCinch’s pistol grip design permits ergonomic, one-handed all-inside meniscal repair. It is best suited for meniscal tears of the posterior horn and body. The posterior horn of the meniscus can be repaired with the SpeedCinch inserted through either the ipsilateral or contralateral portal. Meniscal body tears, however, are best approached via a contralateral portal. The end of the device contains a 15g needle that should be advanced across the meniscus. After passage of the needle, the first implant is pushed through the needle when the trigger is fully deployed.
Next, the SpeedCinch needle should be moved at least 1 cm for placement of the second implant in either a horizontal or vertical mattress fashion. After the needle is brought to the second insertion site, the implant selector button is moved to 2 and the trigger is depressed until the first click is felt and heard. Next, the trigger is held in place after the first click and the needle is advanced across the meniscus before fully depressing the trigger, which will advance the second implant. The device is then removed and the pre-tied knot is secured with a knot pusher and then cut.
Vic Goradia, MD, G2 Orthopedics and Sports Medicine, Glen Allen, VA
Cayenne Medical, Inc. (www.cayennemedical.com)
The CrossFix II System
The CrossFix II System offers a unique, all-arthroscopic, suture-only meniscal repair that reduces the risk of chondral injury. Its “all-inside” technique uses 2 parallel suture delivery needles, available in both curved and straight designs, inserted through a single incision to provide an all-inside meniscus repair. This simple procedure can be performed in minutes via the device’s pre-tied, sliding knot, creating a “suture only” mattress stitch that replicates the repair of standard open suturing techniques. The speed and reproducibility of the repair dramatically improves operating room efficiency. Even complex tears requiring multiple sutures can be repaired in minutes.
Benefits: All-suture, no implants; single insertion; pre-tied, sliding knot. Parallel needles may be difficult to use in very tight knees.
Surgical pearl: Avoid torqueing the needles upon insertion into the meniscus.
Kenneth Montgomery, MD, Tri-County Orthopedics, Cedar Knolls, NJ.
Ceterix Orthopaedics, Inc. (www.ceterix.com)
NovoStitch Plus
The Ceterix NovoStitch Plus enables surgeons to place circumferential compression stitches around meniscus tears. These stitch patterns are designed to provide anatomical reduction and uniform compression of the tear edges, and repair the femoral surface (top) and tibial surface (bottom) of the tear with each stitch. It is also designed to eliminate neurovascular risk and avoid excessive entrapment or extrusion of the meniscus to capsule. The NovoStitch Plus passes circumferential stitches via an all-inside arthroscopic technique. In addition to anatomically repairing vertical tears, circumferential stitches also enable repair of tear types that were previously considered difficult to adequately sew.
Benefits: Passes both sides of the stitch with 1 insertion (avoids tissue bridges and girth hitches). Retractable lower jaw (allows access to tight knees, and allows removal from knees with mobile menisci). Needle extends into the posterior gutter through the tip of the lower jaw (does not touch chondral surfaces). Smaller lower jaw tooth (easier insertion of the lower jaw under the meniscus). Upper jaw curved to follow the shape contour of the femoral side of the meniscus and femoral condyle. Compared to hybrid all-inside devices, inside-out and outside-in repairs. Enables anatomical reduction and uniform compression. Each stitch repairs the top and bottom of the tear. Designed to eliminate neurovascular risk. Avoids excessive capsular entrapment. Repairs effectively in front of the popliteal hiatus. Enables side-to-side radial tear repair, and top-to-bottom horizontal tear repair. Significantly smaller needle than used by hybrid all-inside devices.
Surgical pearl: A spinal needle should be used to establish the skin incision location of the working portal. The optimal skin incision location is one where a spinal needle can be inserted such that the distal end of the needle is parallel to the region of the tibial plateau under the meniscus.
Justin Saliman, MD, Cedars-Sinai Orthopaedic Center, Los Angeles CA
Mitek Sports Medicine (www.depuysynthes.com)
Mitek Omnispan
The Mitek Omnispan Meniscal Repair System is an all-inside arthroscopic meniscal repair device. It consists of a low profile needle, pre-loaded with 2 PEEK backstops and No. 2/0 Orthocord High Strength Orthopaedic Suture, which is delivered using the Omnispan System Applier. The needles come in 3 angles (0°, 12°, and 27°) and the Applier is a single-patient, multi-use design, meaning the same Applier can be used for multiple implants with a single patient. The needles also have a safety sleeve over each set of implants to prevent delivery failure. Once the needle is attached to the Applier, the device is inserted into the knee. Using the gray trigger, the first implant is delivered into the meniscocapsular region. The red trigger is then pulled, advancing the second implant. Once appropriate positioning of the second implant is determined, the gray trigger is fired again. The surgeon pulls on the sliding knot advancing the suture through the implants creating a suture bridge over the tear. The final repair consists of Orthocord Suture spanning the meniscus, while the PEEK backstops are embedded in the meniscocapsular area only.
Surgical pearls:
An adequate portal opening is paramount for successful implementation. I widen my portal with an instrument clamp to allow easy passage of the Applier. It is usually necessary to switch the camera back and forth between both portals to gain access to the angles needed to create the repair. I also always use a skid when inserting the Applier to prevent soft tissue impingement.
The surgeon should always check to make sure the implant is properly loaded on the back table, ensuring it is fully inserted into the locking mechanism and loaded in the correct direction.
The surgeon should hold the Applier firmly when firing the gray trigger, as some kick-back does occur.
The Applier should remain in the joint throughout the advancing and deployment of implants, so the surgeon can maintain visualization with the scope. Once the implants are in position, I then remove the Applier from the joint space.
Use a probe to place counter-pressure on the meniscus when tightening the suture. The sliding suture system is a double-loop design. In order to smoothly advance the suture, I recommend that a probe be placed on the smaller loop closest to the meniscus, so that when the suture is tightened, the probe is under the smaller loop. A gentle back-and-forth pulling maneuver between the free suture and the probe creates a smooth transition during tightening.
Scott A. Sigman, MD, Chief of Orthopedics, Lowell General Hospital; Team Physician, UMass Lowell
Smith & Nephew Inc. (www.smith-nephew.com)
FAST-FIX 360
The FAST-FIX360 design enables you to deploy implants in any hand position—vertically, horizontally on either side of the meniscus—with a fast, smooth advancing motion. This spring-action design facilitates the advancement of each implant into the capsule. Smaller implants and pre-tied, self-sliding knots made of ULTRABRAID 2-0 Suture create smaller needle insertions, reducing disruption to the meniscus. Low-profile, stiffer needle shaft improves control while enabling access and visibility to hard-to-reach areas of the meniscus. Set needle depth penetration from 10 mm to 18 mm with the push of a button. The FAST-FIX 360 System has biomechanical properties that best reproduce the vertical mattress suture technique.
Surgical pearl: Assess tear pattern and reparability, then reduce and template repair construct and suture points. Precisely select portal insertion sites, ensuring perpendicular vector needle delivery at the repair site. Measure the meniscal fragment size and rim width, setting the depth penetration limiter (usually set to 15 to 18 mm). Insert first anchor posteriorly and superiorly or away from the insertion portal and then after deployment, insert the second anchor anteriorly and inferiorly or into the “near” tear fragment. Tension the suture using the knot pusher/cutter as a “suture stent” to manually “pull and push” the suture, compressing the repair construct and coapting the tear. Avoid “over-repairing” the tear by spacing out sutures at 3 mm to 5 mm and alternating femoral and tibial undersurface placement.
Nicholas A. Sgaglione, MD (pictured), and Ryan A. Harrell, DO
VIDEO: Migraine plus aura doubles ischemic stroke rate
LOS ANGELES – Adults who have migraine with aura had twice the rate of ischemic strokes, compared with people with migraine without aura in a review of 11,663 older Americans followed for an average of 18 years.
Migraine with aura appeared to increase the rate of cardioembolic strokes particularly, and the findings highlighted that middle-aged and older adults diagnosed with migraine with aura require special attention to interventions that could reduce their cardiovascular disease risks, Dr. Souvik Sen said in a video interview during the International Stroke Conference.
While results from prior studies have implicated migraine with aura as a risk factor for ischemic stroke, the new analysis was notable for involving an especially large number of community-dwelling older Americans, suggesting that the finding is broadly applicable to the general population of people with an average age of 58-60 years, said Dr. Sen, professor of clinical neurology and chairman of the department of neurology at the University of South Carolina in Columbia.
“It’s been known that migraine can increase the risk of stroke and that certain subgroups of migraine patients are especially important, such as patients with aura,” commented Dr. Ralph Sacco, professor and chairman of neurology at the University of Miami. “When I see patients with migraine I think about treating their migraine effectively and getting it under control, and putting even greater focus on controlling stroke risk factors. I also warn women with migraine about their increased stroke risk from using estrogen-containing oral contraceptives or hormone replacement,” Dr. Sacco noted.
Dr. Sen noted that the new analysis included people enrolled in the Atherosclerosis Risk in Communities (ARIC) study, which enrolled a random sample of more than 15,000 participants from four U.S. communities in 1987. The analysis focused on 11,663 people with migraine information collected during the third ARIC examination during 1993-1995, which identified 458 people (4%) who had migraine with aura, 1,158 (10%) who had migraine without aura, and 10,047 (86%) who were migraine free. During 18 years of follow-up the incidence of ischemic stroke among those with migraine with aura was twofold higher than among those with migraine without aura after adjustment for baseline rate of stroke risk factors, a statistically significant difference, he reported at the meeting sponsored by the American Heart Association. This difference seemed largely driven by a significant increase in the rate of cardioembolic strokes among the people with migraine with aura.
One hypothesis explaining the increased stroke rate in patients with aura is that, in patients with this presentation of migraine, the neurologic activity of migraine appears to spread to cerebral blood vessels and may affect blood flow within those vessels, Dr. Sen explained. It is also possible that migraine with aura may be a risk marker for or may predispose people to more extensive cardiovascular disease including atrial fibrillation.
The primary clinical implication of the finding is that people with migraine with aura should especially undergo a thorough assessment for cardiovascular disease risk factors, and should receive especially aggressive risk factor management, including controlling hypertension and smoking cessation, assessing ECG and atrial fibrillation, and avoiding estrogen treatment, Dr. Sen said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
LOS ANGELES – Adults who have migraine with aura had twice the rate of ischemic strokes, compared with people with migraine without aura in a review of 11,663 older Americans followed for an average of 18 years.
Migraine with aura appeared to increase the rate of cardioembolic strokes particularly, and the findings highlighted that middle-aged and older adults diagnosed with migraine with aura require special attention to interventions that could reduce their cardiovascular disease risks, Dr. Souvik Sen said in a video interview during the International Stroke Conference.
While results from prior studies have implicated migraine with aura as a risk factor for ischemic stroke, the new analysis was notable for involving an especially large number of community-dwelling older Americans, suggesting that the finding is broadly applicable to the general population of people with an average age of 58-60 years, said Dr. Sen, professor of clinical neurology and chairman of the department of neurology at the University of South Carolina in Columbia.
“It’s been known that migraine can increase the risk of stroke and that certain subgroups of migraine patients are especially important, such as patients with aura,” commented Dr. Ralph Sacco, professor and chairman of neurology at the University of Miami. “When I see patients with migraine I think about treating their migraine effectively and getting it under control, and putting even greater focus on controlling stroke risk factors. I also warn women with migraine about their increased stroke risk from using estrogen-containing oral contraceptives or hormone replacement,” Dr. Sacco noted.
Dr. Sen noted that the new analysis included people enrolled in the Atherosclerosis Risk in Communities (ARIC) study, which enrolled a random sample of more than 15,000 participants from four U.S. communities in 1987. The analysis focused on 11,663 people with migraine information collected during the third ARIC examination during 1993-1995, which identified 458 people (4%) who had migraine with aura, 1,158 (10%) who had migraine without aura, and 10,047 (86%) who were migraine free. During 18 years of follow-up the incidence of ischemic stroke among those with migraine with aura was twofold higher than among those with migraine without aura after adjustment for baseline rate of stroke risk factors, a statistically significant difference, he reported at the meeting sponsored by the American Heart Association. This difference seemed largely driven by a significant increase in the rate of cardioembolic strokes among the people with migraine with aura.
One hypothesis explaining the increased stroke rate in patients with aura is that, in patients with this presentation of migraine, the neurologic activity of migraine appears to spread to cerebral blood vessels and may affect blood flow within those vessels, Dr. Sen explained. It is also possible that migraine with aura may be a risk marker for or may predispose people to more extensive cardiovascular disease including atrial fibrillation.
The primary clinical implication of the finding is that people with migraine with aura should especially undergo a thorough assessment for cardiovascular disease risk factors, and should receive especially aggressive risk factor management, including controlling hypertension and smoking cessation, assessing ECG and atrial fibrillation, and avoiding estrogen treatment, Dr. Sen said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
LOS ANGELES – Adults who have migraine with aura had twice the rate of ischemic strokes, compared with people with migraine without aura in a review of 11,663 older Americans followed for an average of 18 years.
Migraine with aura appeared to increase the rate of cardioembolic strokes particularly, and the findings highlighted that middle-aged and older adults diagnosed with migraine with aura require special attention to interventions that could reduce their cardiovascular disease risks, Dr. Souvik Sen said in a video interview during the International Stroke Conference.
While results from prior studies have implicated migraine with aura as a risk factor for ischemic stroke, the new analysis was notable for involving an especially large number of community-dwelling older Americans, suggesting that the finding is broadly applicable to the general population of people with an average age of 58-60 years, said Dr. Sen, professor of clinical neurology and chairman of the department of neurology at the University of South Carolina in Columbia.
“It’s been known that migraine can increase the risk of stroke and that certain subgroups of migraine patients are especially important, such as patients with aura,” commented Dr. Ralph Sacco, professor and chairman of neurology at the University of Miami. “When I see patients with migraine I think about treating their migraine effectively and getting it under control, and putting even greater focus on controlling stroke risk factors. I also warn women with migraine about their increased stroke risk from using estrogen-containing oral contraceptives or hormone replacement,” Dr. Sacco noted.
Dr. Sen noted that the new analysis included people enrolled in the Atherosclerosis Risk in Communities (ARIC) study, which enrolled a random sample of more than 15,000 participants from four U.S. communities in 1987. The analysis focused on 11,663 people with migraine information collected during the third ARIC examination during 1993-1995, which identified 458 people (4%) who had migraine with aura, 1,158 (10%) who had migraine without aura, and 10,047 (86%) who were migraine free. During 18 years of follow-up the incidence of ischemic stroke among those with migraine with aura was twofold higher than among those with migraine without aura after adjustment for baseline rate of stroke risk factors, a statistically significant difference, he reported at the meeting sponsored by the American Heart Association. This difference seemed largely driven by a significant increase in the rate of cardioembolic strokes among the people with migraine with aura.
One hypothesis explaining the increased stroke rate in patients with aura is that, in patients with this presentation of migraine, the neurologic activity of migraine appears to spread to cerebral blood vessels and may affect blood flow within those vessels, Dr. Sen explained. It is also possible that migraine with aura may be a risk marker for or may predispose people to more extensive cardiovascular disease including atrial fibrillation.
The primary clinical implication of the finding is that people with migraine with aura should especially undergo a thorough assessment for cardiovascular disease risk factors, and should receive especially aggressive risk factor management, including controlling hypertension and smoking cessation, assessing ECG and atrial fibrillation, and avoiding estrogen treatment, Dr. Sen said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point: Older adults with migraine and aura had twice the ischemic strokes, compared with patients with aura-free migraines.
Major finding: Strokes occurred twofold more often in patients with migraine with aura, compared with patients with migraine alone after adjustments.
Data source: Review of 11,663 older adult Americans enrolled in the Atherosclerosis Risk in Communities study.
Disclosures: Dr. Sen had no disclosures.
Using a Multiplex of Biomarkers to Detect Prostate Cancer
Because prostate cancer (PCa) is a heterogeneous disease, researchers from Universitat de Barcelona in Spain say that a combination of biomarkers would outperform single markers in detection. In a previous study, they identified new putative mRNA markers for PCa diagnosis, tested some of those biomarkers, and validated the commercially available test based on urine prostate cancer antigen 3 (PCA3 ) expression along with the best-performing mRNA panels of biomarkers reported.
Related:Early-Stage Prostate Cancer Incidence Declines Following USPSTF Guidelines Update
Urine samples collected from 224 men and 10 patients with prostate specific antigen (PSA) levels > 4 were included as controls. Seven of 42 genes that the researchers evaluated were independent predictors for PCa. From these data, the researchers developed a 4-gene expression signature, HIST1H2BG, SPP1, ELF3, and PCA3, with higher diagnostic accuracy than PCA3, the only noninvasive urinary biomarker commercially available. Their 4-gene signature had a sensitivity of 77% and specificity of 67% for discriminating between tumor and control urines and a positive predictive value of 83%.
Related: Prostate Cancer in Seniors, Part 1: Epidemiology, Pathology, and Screening
The researchers say their findings suggest that a urinary biomarker panel could improve detection of PCa. However, they add that the accuracy of the available urinary panels—including their signature—is not yet high enough to warrant using them routinely.
Related: Prostate Cancer Survivorship Care
Mengual L, Lozano JJ, Ingelmo-Torres M, et al. BMC Cancer. 2016;16(1):76.
Because prostate cancer (PCa) is a heterogeneous disease, researchers from Universitat de Barcelona in Spain say that a combination of biomarkers would outperform single markers in detection. In a previous study, they identified new putative mRNA markers for PCa diagnosis, tested some of those biomarkers, and validated the commercially available test based on urine prostate cancer antigen 3 (PCA3 ) expression along with the best-performing mRNA panels of biomarkers reported.
Related:Early-Stage Prostate Cancer Incidence Declines Following USPSTF Guidelines Update
Urine samples collected from 224 men and 10 patients with prostate specific antigen (PSA) levels > 4 were included as controls. Seven of 42 genes that the researchers evaluated were independent predictors for PCa. From these data, the researchers developed a 4-gene expression signature, HIST1H2BG, SPP1, ELF3, and PCA3, with higher diagnostic accuracy than PCA3, the only noninvasive urinary biomarker commercially available. Their 4-gene signature had a sensitivity of 77% and specificity of 67% for discriminating between tumor and control urines and a positive predictive value of 83%.
Related: Prostate Cancer in Seniors, Part 1: Epidemiology, Pathology, and Screening
The researchers say their findings suggest that a urinary biomarker panel could improve detection of PCa. However, they add that the accuracy of the available urinary panels—including their signature—is not yet high enough to warrant using them routinely.
Related: Prostate Cancer Survivorship Care
Mengual L, Lozano JJ, Ingelmo-Torres M, et al. BMC Cancer. 2016;16(1):76.
Because prostate cancer (PCa) is a heterogeneous disease, researchers from Universitat de Barcelona in Spain say that a combination of biomarkers would outperform single markers in detection. In a previous study, they identified new putative mRNA markers for PCa diagnosis, tested some of those biomarkers, and validated the commercially available test based on urine prostate cancer antigen 3 (PCA3 ) expression along with the best-performing mRNA panels of biomarkers reported.
Related:Early-Stage Prostate Cancer Incidence Declines Following USPSTF Guidelines Update
Urine samples collected from 224 men and 10 patients with prostate specific antigen (PSA) levels > 4 were included as controls. Seven of 42 genes that the researchers evaluated were independent predictors for PCa. From these data, the researchers developed a 4-gene expression signature, HIST1H2BG, SPP1, ELF3, and PCA3, with higher diagnostic accuracy than PCA3, the only noninvasive urinary biomarker commercially available. Their 4-gene signature had a sensitivity of 77% and specificity of 67% for discriminating between tumor and control urines and a positive predictive value of 83%.
Related: Prostate Cancer in Seniors, Part 1: Epidemiology, Pathology, and Screening
The researchers say their findings suggest that a urinary biomarker panel could improve detection of PCa. However, they add that the accuracy of the available urinary panels—including their signature—is not yet high enough to warrant using them routinely.
Related: Prostate Cancer Survivorship Care
Mengual L, Lozano JJ, Ingelmo-Torres M, et al. BMC Cancer. 2016;16(1):76.