User login
The medicolegal considerations of interacting with your patients online
CASE: Patient discloses personal information in electronic communication. How to respond and what’s at stake?
Your nurse comes to you with a dilemma. Last Friday she received an email from a patient, sent to the nurse’s personal email account (G-mail) that conveyed information regarding the patient’s recent treatment for a herpetic vulvar lesion. The text details presumed exposure, date and time, number of sexual partners, concernfor “spread of disease,” and the patient’s desire to have a comprehensive sexually transmitted infection screening as soon as possible.
Your nurse has years of professional experience, but she is perhaps not the most savvy with regard to current information technology and social media. Nonetheless, she knows it is best not to immediately respond to the patient’s email without checking with you. She tracks you down on Monday morning to review the email and the dilemma she feels she has been placed in. What’s the best next step?
While discussing the general question with the staff, another nurse notes that there have been some reviews of the office on social media. It seems that this second nurse tweets and texts with patients all the time. The office manager strongly suggests that the office “join the 21st Century” by setting up a Facebook page and using their webpage to attract new patients and communicate with current patients.
How do you prepare for this? Is your staff knowledgeable about the dos and don’ts of social media?
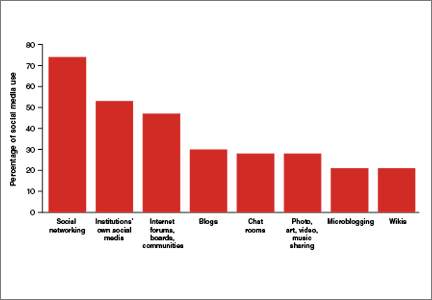
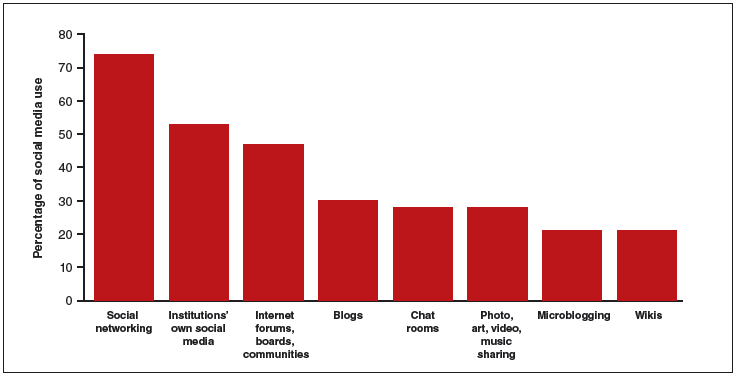
The use of social media by health care providers has been growing for several years. Back in 2011 a large survey by QuantiaMD revealed that 87% of physicians used social media for personal reasons, and 67% of them used it professionally.1 How they used it for professional purposes also was explored in 2011, with almost 3 of 4 physicians using it for social networking and more than half engaging with their own institution’s social media (FIGURE).2 In 2013, 53% of physicians indicated that their practice had a Facebook platform, 28% had a presence on LinkedIn, and 21% were on Twitter.3 Not surprisingly, social media use is higher among younger physicians4; the 2016 equivalents to these percentages most likely are higher.
| Health providers’ use of social media for professional reasons2 |
|
In 2011, a survey found that most health providers used social networking, their institutions’ own social media, and Internet forums, boards, and communities for professional reasons. |
Patients’ outreach through social media regarding health care information continues to grow, with 33.8% asking for health advice using social media.5 While email and other social media open the possibility of improved communication with patients, they also present a number of important professional and legal issues that deserve special consideration.6 Each medium presents its own challenges, but there are 4 categories of concern related to basic values and rights that we consider important to review:
- confidentiality
- dual relationships and conflicts of interest
- quality of care and advice
- general professionalism (including advertising).
Confidentiality
Few values of the medical profession are of longer standing than the commitment to maintain patient privacy. Fifth Century BC obligations continue to apply to the technology of the 21st Century AD. And the challenges are significant.
Email is not secure
In the opening case, the choice to email her clinician was apparently the patient’s. She probably does not realize that email is not very confidential, although it is undoubtedly in the Terms of Service Agreement she clicked through. Her email was likely scanned by her email service provider—Google, in this case—as well as the nurse. If, however, the physician’s office responds by email, it may well compound the confidentiality problem by further distributing the information through yet another email provider.
If, as a physician, you encourage email communication by your patients, a smart approach is to emphasize that such communications are not very confidential. At a minimum, until a secure email system can be established, it is best not to transmit medical information via email and to inform patients of the risk of such communication. In the case above, the nurse who received the email should respond to the patient by telephone (much more secure). Or she can respond to the patient by email (not including the patient’s message in the return), writing that, because email communications are inherently not confidential, she suggests a phone call or personal visit.
This case also notes that the patient sent the email to the nurse’s personal account, not to an office email account. Sending medical emails to an employee’s personal account raises additional problems of confidentiality and appropriate controls. It should be made clear that employees should not be discussing private medical matters via their own email accounts.
Other forms of social media are also not secure
Similar concerns arise about texting and using Twitter by the second nurse. These activities apparently had been unknown to the physician, but the practice still may be responsible for her actions. These are insecure forms of communication and raise serious ethical and legal concerns.
Other social media pose confidentiality risks as well. For example, a physician was dismissed from a position and reprimanded by the medical board for posting patient information on Facebook,7 and an ObGyn caused problems by posting a nasty note about a patient who showed up late for an appointment.8 Too many patients may not understand that posting on social media is the equivalent of standing on a street corner yelling private information. Social media sites that invite the discussion of personal matters are an invitation to trouble.
Physicians are ethically obliged to protect confidentiality
Professional standards place significant ethical obligations on physicians to protect patient confidentiality. The American Medical Association (AMA) has an ethics opinion on professionalism with social media,9 as does the American College of Obstetricians and Gynecologists (ACOG).10 Another excellent discussion of ethical and practical issues is a joint position paper by the American College of Physicians and the Federation of State Medical Boards.11 Both documents focus attention on issues of confidentiality.
Physicians are legally obliged to protect confidentiality
There are many legal protections for confidentiality that can be implicated by electronic communications and social media. All states provide protection for unwarranted disclosure of private patient information. Such disclosures made electronically are included.12 Indeed, because electronic disclosures may be broadcast more widely, they may be especially dangerous. The misuse of social media may result in license discipline by the state board, regulatory sanctions, or civil liability (rare, but criminal sanctions are a possibility in extreme circumstances).
In addition to state laws regarding confidentiality, there are a number of federal laws that cover confidential medical information. None is more important than the Health Insurance Portability and Accountability Act (HIPAA) and the more recent HITECH amendments (Health Information Technology for Economic and Clinical Health).13 These laws have both privacy provisions and security (including “encryption”) requirements. These are complicated laws but at their core are the notions that health care providers and some others:
- are responsible for maintaining the security and privacy of health information
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.14
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.
A good source of step-by-step information about these laws is “Health information privacy: Covered entities and business associates,” on the US Health and Human Services website.14
HITECH also provides for notice to patients when health information is inappropriately transmitted. Thus, a missing USB flash drive with patient information may require notification to thousands of patients.15 Any consideration of the use of email or social media in medical practice must take into account the HIPAA/HITECH obligations to protect the security of patient health information. There can be serious professional consequences for failing to follow the HIPAA requirements.16
Dual relationships and conflicts of interest
In our hypothetical case, the office manager’s suggestion that the office use Facebook and their website to attract new patients also may raise confidentiality problems. The Facebook suggestion especially needs to be considered carefully. Facebook use is estimated to be 63% to 96% among students and 13% to 47% among health care professionals.17 Facebook is most often seen as an interactive social site; it risks blurring the lines between personal and professional relationships.9 There is a consensus that a physician should not “friend” patients on Facebook. The AMA ethics opinion notes that “physicians must maintain appropriate boundaries of the patient-physician relationship in accordance with professional ethical guidelines, just as they would in any other context.”9
Separate personal and professional contacts
Difficulties with interactive social media are not limited to the physicians in a practice. The problems increase with the number of staff members who post or respond on social media. Control of social media is essential. The practice must ensure that staff members do not slip into inappropriate personal comments and relationships. Staff should understand (and be reminded of) the necessity of separating personal and professional contacts.
Avoid misunderstandings
In addition, whatever the intent of the physician and staff may be, it is essentially impossible to know how patients will interpret interactions on these social media. The very informal, off-the-cuff, chatty way in which Facebook and similar sites are used invites misunderstandings, and maintaining professional boundaries is necessary.
Ground rules
All of this is not to say that professionals should never use Facebook or similar sites. Rather, if used, ground rules need to be established.
Social media communications must:
- be professional and not related to personal matters
- not be used to give medical advice
- be controlled by high level staff
- be reviewed periodically.
Staff training
Particularly for interactive social media (email, texts, Twitter, Facebook, etc), it is essential that there be both clear policies and good staff training (TABLE).9–11,18 There really should be no “making it up as we go along.” Staff on a social media lark of their own can be disastrous for the practice. Policies need to be updated frequently, and staff training reinforced and repeated periodically.
Quality of care and advice
Start with your website
Institutions’ websites are major sources of health care information: Nearly 32% of US adults would be very likely to prefer a hospital based on its website.5 Your website can be an important face of your practice to the community—for good or for bad. On one hand, the practice can control what is on a website and, unlike some social media, it will not be directed to individual patients. Done well, it “provides golden opportunities for marketing physician services, as well as for contributing to public health by providing high-quality online content that is both accurate and understandable to laypeople.”19 Done badly, it can convey incorrect and harmful information and discredit the medical practice that established it.
Your website introduces the practice and settings, but it will serve another purpose to thousands of people who likely will see it over time as a source of credible health information. The importance of ensuring that your website is carefully constructed to provide, or link to, good medical advice that contributes to quality of care cannot be overstated.
A good website begins with a clear statement of the reasons and goals for having the site. Professional design assistance generally is used to create the site, but that design process needs to be overseen by a medical professional to ensure that it conveys the sense of the practice and provides completely accurate information. A homepage of dancing clowns with stethoscopes may seem good to a 20-something-year-old designer, but it is not appropriate for a physician. It will be the practice, not the designer, who is held accountable for the site content. Links to other sites need to be vetted and used with care. Patients and other members of the public may well take the links as carrying the endorsement of the practice and its physicians.
Perhaps the greatest risk of a website is that it will not be kept current. Unfortunately, they do not update themselves. Some knowledgeable staff member must frequently review it to update everything from office hours and personnel to links to other sites. In addition, the physicians periodically must review it to ensure that all medical information is up to date and accurate. Old, outdated information about the office can put off potential patients. Outdated medical information may be harmful to patients who rely on it.
Any professional website should include disclaimers informing users that the site is not intended to establish a professional relationship or to give professional advice. The nature and extent of the disclaimer will depend on the type of information on the site. An example of a particularly thorough disclaimer is the Mayo Clinic disclaimer and terms of use (http://www.mayoclinic.org/about-this-site/terms-conditions-use-policy).
General professionalism
At the end of the day, social media are an outreach from a medical practice and from the profession to the public.20 Failure to treat these platforms with appropriate professional standards may result in professional discipline, damages, or civil penalties. Almost all of the reviews of social media use in health care practice note that the risks of inappropriate use are not only to the individual physician but also to the general medical profession, which may be undermined. Consider posting policies of the relevent state medical boards, the AMA, and ACOG in your office after you have had a discussion with your staff about them.21
The AMA statement includes a provision that a physician seeing unprofessional social media conduct by a colleague has the responsibility to bring that to the attention of the colleague. If the colleague does not correct a significant problem, “the physician should report the matter to appropriate authorities.”9
Bottom line
Any practitioner considering the use of social media must view it as a major step that requires caution, expert assistance, and constant attention to potential privacy, quality, and professionalism issues. If you are considering it, ensure that all staff associated with the practice understand and agree to the established limits on social media use.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- Modahl M, Tompsett L, Moorhead T. Doctors, patients, & social media. Quantia MD website. http://www.quantiamd.com/q-qcp/DoctorsPatientSocialMedia.pdf. Published September 2011. Accessed February 18, 2016.
- Kuberacka A, Wengrojj J, Fabozzi N. Social media use in U.S. healthcare provider institutions: Insights from Frost & Sullivan and iHT2 survey. Frost and Sullivan website. http://ihealthtran.com/pdf/frostiht2survey.pdf. Published August 30, 2011. Accessed February 18, 2016.
- O’Connor ME. How do tech savvy physicians use health technology and social media? Health Care Social Media website. http://hcsmmonitor.com/2014/01/08/how-do-tech-savvy-physicians-use-health-technology-and-social-media/. Published January 8, 2014. Accessed February 18, 2016.
- American Medical Association (AMA) Insurance. 2014 work/life profiles of today’s U.S. physician. AMA Insurance website. https://www.amainsure.com/work-life-profiles-of-todays-us-physician.html. Published April 2014. Accessed February 18, 2016.
- National Research Corporation. 2013 National Market Insights Survey: Health care social media website. https://healthcaresocialmedia.files.wordpress.com/2014/04/nrc-infographiclong.jpg. Accessed February 18, 2016.
- Suby C. Social media in health care: benefits, concerns and guidelines for use. Creat Nurs. 2013;19(3):140–147.
- Conaboy C. For doctors, social media a tricky case. Boston Globe. http://www.boston.com/lifestyle/health/articles/2011/04/20/for_doctors_social_media_a_tricky_case/?page=full. Published April 20, 2011. Accessed February 18, 2016.
- Matyszczyk C. Outcry as ob-gyn uses Facebook to complain about patient. CNET. http://www.cnet.com/news/outcry-as-ob-gyn-uses-facebook-to-complain-about-patient/Minion Pro. Published February 9, 2013. Accessed February 18, 2016.
- American Medical Association (AMA). Opinion 9.124: Professionalism in the use of social media. AMA website. http://www.ama-assn.org/ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion9124.page? Published June 2011. Accessed February 18, 2016.
- American College of Obstetricians and Gynecologists (ACOG) Committee on Professional Liability. ACOG Committee Opinion No. 622: professional use of digital and social media. Obstet Gynecol. 2015;125(2):516-520.
- Farnan JM, Sulmasy LS, Worster BK, et al. Online medical professionalism: patient and public relationships: Policy Statement From the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158(8):620–627.
- Hader A, Drown E. Patient privacy and social media. AANA J. 2010;78(4):270–274.
- Kavoussi SC, Huang JJ, Tsai JC, Kempton JE. HIPAA for physicians in the information age. Conn Med. 2014;78(7):425–427.
- U.S. Department of Health & Human Services (HHS). Health information privacy: Covered entities and business associates. HHS website. http://www.hhs.gov/ocr/privacy/hipaa/understanding/coveredentities/. Published March 14, 2012. Accessed February 18, 2016.
- Perna G. Breach report: lost flash drive at Kaiser Permanente affects 49,000 patients. Healthcare Informatics website. http://www.healthcare-informatics.com/news-item/breach-report-lost-flash-drive-kaiser-permanente-affects-49000-patients. Published December 11, 2013. Accessed February 18, 2016.
- McBride M. How to ensure your social media efforts are HIPAA-compliant. Med Econ. 2012;89:70–74.
- Von Muhlen M, Ohno-Machado L. Reviewing social media use by clinicians. J Am Med Inform Assoc. 2012;19(5):777–781.
- Omurtag K, Turek P. Incorporating social media into practice: a blueprint for reproductive health providers. Clin Obstet Gynecol. 2013;56(3):463–470.
- Radmanesh A, Duszak R, Fitzgerald R. Social media and public outreach: a physician primer. Am J Neuroradiol. 2015;36(7):1223–1224.
- Grajales FJ 3rd, Sheps S, Ho K, Novak-Lauscher H, Eysenbach G. Social media: a review and tutorial of applications in medicine and health care. J Med Internet Res. 2014;16(2):e13.
- ACOG Today. Social media guide: how to comment with patients and spread women’s health messages. American Congress of Obstetricians and Gynecologists website. http://www.acog.org/-/media/ACOG-Today/acogToday201211.pdf. Published November 2012. Accessed February 18, 2016.
CASE: Patient discloses personal information in electronic communication. How to respond and what’s at stake?
Your nurse comes to you with a dilemma. Last Friday she received an email from a patient, sent to the nurse’s personal email account (G-mail) that conveyed information regarding the patient’s recent treatment for a herpetic vulvar lesion. The text details presumed exposure, date and time, number of sexual partners, concernfor “spread of disease,” and the patient’s desire to have a comprehensive sexually transmitted infection screening as soon as possible.
Your nurse has years of professional experience, but she is perhaps not the most savvy with regard to current information technology and social media. Nonetheless, she knows it is best not to immediately respond to the patient’s email without checking with you. She tracks you down on Monday morning to review the email and the dilemma she feels she has been placed in. What’s the best next step?
While discussing the general question with the staff, another nurse notes that there have been some reviews of the office on social media. It seems that this second nurse tweets and texts with patients all the time. The office manager strongly suggests that the office “join the 21st Century” by setting up a Facebook page and using their webpage to attract new patients and communicate with current patients.
How do you prepare for this? Is your staff knowledgeable about the dos and don’ts of social media?
The use of social media by health care providers has been growing for several years. Back in 2011 a large survey by QuantiaMD revealed that 87% of physicians used social media for personal reasons, and 67% of them used it professionally.1 How they used it for professional purposes also was explored in 2011, with almost 3 of 4 physicians using it for social networking and more than half engaging with their own institution’s social media (FIGURE).2 In 2013, 53% of physicians indicated that their practice had a Facebook platform, 28% had a presence on LinkedIn, and 21% were on Twitter.3 Not surprisingly, social media use is higher among younger physicians4; the 2016 equivalents to these percentages most likely are higher.
| Health providers’ use of social media for professional reasons2 |

|
In 2011, a survey found that most health providers used social networking, their institutions’ own social media, and Internet forums, boards, and communities for professional reasons. |
Patients’ outreach through social media regarding health care information continues to grow, with 33.8% asking for health advice using social media.5 While email and other social media open the possibility of improved communication with patients, they also present a number of important professional and legal issues that deserve special consideration.6 Each medium presents its own challenges, but there are 4 categories of concern related to basic values and rights that we consider important to review:
- confidentiality
- dual relationships and conflicts of interest
- quality of care and advice
- general professionalism (including advertising).
Confidentiality
Few values of the medical profession are of longer standing than the commitment to maintain patient privacy. Fifth Century BC obligations continue to apply to the technology of the 21st Century AD. And the challenges are significant.
Email is not secure
In the opening case, the choice to email her clinician was apparently the patient’s. She probably does not realize that email is not very confidential, although it is undoubtedly in the Terms of Service Agreement she clicked through. Her email was likely scanned by her email service provider—Google, in this case—as well as the nurse. If, however, the physician’s office responds by email, it may well compound the confidentiality problem by further distributing the information through yet another email provider.
If, as a physician, you encourage email communication by your patients, a smart approach is to emphasize that such communications are not very confidential. At a minimum, until a secure email system can be established, it is best not to transmit medical information via email and to inform patients of the risk of such communication. In the case above, the nurse who received the email should respond to the patient by telephone (much more secure). Or she can respond to the patient by email (not including the patient’s message in the return), writing that, because email communications are inherently not confidential, she suggests a phone call or personal visit.
This case also notes that the patient sent the email to the nurse’s personal account, not to an office email account. Sending medical emails to an employee’s personal account raises additional problems of confidentiality and appropriate controls. It should be made clear that employees should not be discussing private medical matters via their own email accounts.
Other forms of social media are also not secure
Similar concerns arise about texting and using Twitter by the second nurse. These activities apparently had been unknown to the physician, but the practice still may be responsible for her actions. These are insecure forms of communication and raise serious ethical and legal concerns.
Other social media pose confidentiality risks as well. For example, a physician was dismissed from a position and reprimanded by the medical board for posting patient information on Facebook,7 and an ObGyn caused problems by posting a nasty note about a patient who showed up late for an appointment.8 Too many patients may not understand that posting on social media is the equivalent of standing on a street corner yelling private information. Social media sites that invite the discussion of personal matters are an invitation to trouble.
Physicians are ethically obliged to protect confidentiality
Professional standards place significant ethical obligations on physicians to protect patient confidentiality. The American Medical Association (AMA) has an ethics opinion on professionalism with social media,9 as does the American College of Obstetricians and Gynecologists (ACOG).10 Another excellent discussion of ethical and practical issues is a joint position paper by the American College of Physicians and the Federation of State Medical Boards.11 Both documents focus attention on issues of confidentiality.
Physicians are legally obliged to protect confidentiality
There are many legal protections for confidentiality that can be implicated by electronic communications and social media. All states provide protection for unwarranted disclosure of private patient information. Such disclosures made electronically are included.12 Indeed, because electronic disclosures may be broadcast more widely, they may be especially dangerous. The misuse of social media may result in license discipline by the state board, regulatory sanctions, or civil liability (rare, but criminal sanctions are a possibility in extreme circumstances).
In addition to state laws regarding confidentiality, there are a number of federal laws that cover confidential medical information. None is more important than the Health Insurance Portability and Accountability Act (HIPAA) and the more recent HITECH amendments (Health Information Technology for Economic and Clinical Health).13 These laws have both privacy provisions and security (including “encryption”) requirements. These are complicated laws but at their core are the notions that health care providers and some others:
- are responsible for maintaining the security and privacy of health information
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.14
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.
A good source of step-by-step information about these laws is “Health information privacy: Covered entities and business associates,” on the US Health and Human Services website.14
HITECH also provides for notice to patients when health information is inappropriately transmitted. Thus, a missing USB flash drive with patient information may require notification to thousands of patients.15 Any consideration of the use of email or social media in medical practice must take into account the HIPAA/HITECH obligations to protect the security of patient health information. There can be serious professional consequences for failing to follow the HIPAA requirements.16
Dual relationships and conflicts of interest
In our hypothetical case, the office manager’s suggestion that the office use Facebook and their website to attract new patients also may raise confidentiality problems. The Facebook suggestion especially needs to be considered carefully. Facebook use is estimated to be 63% to 96% among students and 13% to 47% among health care professionals.17 Facebook is most often seen as an interactive social site; it risks blurring the lines between personal and professional relationships.9 There is a consensus that a physician should not “friend” patients on Facebook. The AMA ethics opinion notes that “physicians must maintain appropriate boundaries of the patient-physician relationship in accordance with professional ethical guidelines, just as they would in any other context.”9
Separate personal and professional contacts
Difficulties with interactive social media are not limited to the physicians in a practice. The problems increase with the number of staff members who post or respond on social media. Control of social media is essential. The practice must ensure that staff members do not slip into inappropriate personal comments and relationships. Staff should understand (and be reminded of) the necessity of separating personal and professional contacts.
Avoid misunderstandings
In addition, whatever the intent of the physician and staff may be, it is essentially impossible to know how patients will interpret interactions on these social media. The very informal, off-the-cuff, chatty way in which Facebook and similar sites are used invites misunderstandings, and maintaining professional boundaries is necessary.
Ground rules
All of this is not to say that professionals should never use Facebook or similar sites. Rather, if used, ground rules need to be established.
Social media communications must:
- be professional and not related to personal matters
- not be used to give medical advice
- be controlled by high level staff
- be reviewed periodically.
Staff training
Particularly for interactive social media (email, texts, Twitter, Facebook, etc), it is essential that there be both clear policies and good staff training (TABLE).9–11,18 There really should be no “making it up as we go along.” Staff on a social media lark of their own can be disastrous for the practice. Policies need to be updated frequently, and staff training reinforced and repeated periodically.
Quality of care and advice
Start with your website
Institutions’ websites are major sources of health care information: Nearly 32% of US adults would be very likely to prefer a hospital based on its website.5 Your website can be an important face of your practice to the community—for good or for bad. On one hand, the practice can control what is on a website and, unlike some social media, it will not be directed to individual patients. Done well, it “provides golden opportunities for marketing physician services, as well as for contributing to public health by providing high-quality online content that is both accurate and understandable to laypeople.”19 Done badly, it can convey incorrect and harmful information and discredit the medical practice that established it.
Your website introduces the practice and settings, but it will serve another purpose to thousands of people who likely will see it over time as a source of credible health information. The importance of ensuring that your website is carefully constructed to provide, or link to, good medical advice that contributes to quality of care cannot be overstated.
A good website begins with a clear statement of the reasons and goals for having the site. Professional design assistance generally is used to create the site, but that design process needs to be overseen by a medical professional to ensure that it conveys the sense of the practice and provides completely accurate information. A homepage of dancing clowns with stethoscopes may seem good to a 20-something-year-old designer, but it is not appropriate for a physician. It will be the practice, not the designer, who is held accountable for the site content. Links to other sites need to be vetted and used with care. Patients and other members of the public may well take the links as carrying the endorsement of the practice and its physicians.
Perhaps the greatest risk of a website is that it will not be kept current. Unfortunately, they do not update themselves. Some knowledgeable staff member must frequently review it to update everything from office hours and personnel to links to other sites. In addition, the physicians periodically must review it to ensure that all medical information is up to date and accurate. Old, outdated information about the office can put off potential patients. Outdated medical information may be harmful to patients who rely on it.
Any professional website should include disclaimers informing users that the site is not intended to establish a professional relationship or to give professional advice. The nature and extent of the disclaimer will depend on the type of information on the site. An example of a particularly thorough disclaimer is the Mayo Clinic disclaimer and terms of use (http://www.mayoclinic.org/about-this-site/terms-conditions-use-policy).
General professionalism
At the end of the day, social media are an outreach from a medical practice and from the profession to the public.20 Failure to treat these platforms with appropriate professional standards may result in professional discipline, damages, or civil penalties. Almost all of the reviews of social media use in health care practice note that the risks of inappropriate use are not only to the individual physician but also to the general medical profession, which may be undermined. Consider posting policies of the relevent state medical boards, the AMA, and ACOG in your office after you have had a discussion with your staff about them.21
The AMA statement includes a provision that a physician seeing unprofessional social media conduct by a colleague has the responsibility to bring that to the attention of the colleague. If the colleague does not correct a significant problem, “the physician should report the matter to appropriate authorities.”9
Bottom line
Any practitioner considering the use of social media must view it as a major step that requires caution, expert assistance, and constant attention to potential privacy, quality, and professionalism issues. If you are considering it, ensure that all staff associated with the practice understand and agree to the established limits on social media use.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
CASE: Patient discloses personal information in electronic communication. How to respond and what’s at stake?
Your nurse comes to you with a dilemma. Last Friday she received an email from a patient, sent to the nurse’s personal email account (G-mail) that conveyed information regarding the patient’s recent treatment for a herpetic vulvar lesion. The text details presumed exposure, date and time, number of sexual partners, concernfor “spread of disease,” and the patient’s desire to have a comprehensive sexually transmitted infection screening as soon as possible.
Your nurse has years of professional experience, but she is perhaps not the most savvy with regard to current information technology and social media. Nonetheless, she knows it is best not to immediately respond to the patient’s email without checking with you. She tracks you down on Monday morning to review the email and the dilemma she feels she has been placed in. What’s the best next step?
While discussing the general question with the staff, another nurse notes that there have been some reviews of the office on social media. It seems that this second nurse tweets and texts with patients all the time. The office manager strongly suggests that the office “join the 21st Century” by setting up a Facebook page and using their webpage to attract new patients and communicate with current patients.
How do you prepare for this? Is your staff knowledgeable about the dos and don’ts of social media?
The use of social media by health care providers has been growing for several years. Back in 2011 a large survey by QuantiaMD revealed that 87% of physicians used social media for personal reasons, and 67% of them used it professionally.1 How they used it for professional purposes also was explored in 2011, with almost 3 of 4 physicians using it for social networking and more than half engaging with their own institution’s social media (FIGURE).2 In 2013, 53% of physicians indicated that their practice had a Facebook platform, 28% had a presence on LinkedIn, and 21% were on Twitter.3 Not surprisingly, social media use is higher among younger physicians4; the 2016 equivalents to these percentages most likely are higher.
| Health providers’ use of social media for professional reasons2 |

|
In 2011, a survey found that most health providers used social networking, their institutions’ own social media, and Internet forums, boards, and communities for professional reasons. |
Patients’ outreach through social media regarding health care information continues to grow, with 33.8% asking for health advice using social media.5 While email and other social media open the possibility of improved communication with patients, they also present a number of important professional and legal issues that deserve special consideration.6 Each medium presents its own challenges, but there are 4 categories of concern related to basic values and rights that we consider important to review:
- confidentiality
- dual relationships and conflicts of interest
- quality of care and advice
- general professionalism (including advertising).
Confidentiality
Few values of the medical profession are of longer standing than the commitment to maintain patient privacy. Fifth Century BC obligations continue to apply to the technology of the 21st Century AD. And the challenges are significant.
Email is not secure
In the opening case, the choice to email her clinician was apparently the patient’s. She probably does not realize that email is not very confidential, although it is undoubtedly in the Terms of Service Agreement she clicked through. Her email was likely scanned by her email service provider—Google, in this case—as well as the nurse. If, however, the physician’s office responds by email, it may well compound the confidentiality problem by further distributing the information through yet another email provider.
If, as a physician, you encourage email communication by your patients, a smart approach is to emphasize that such communications are not very confidential. At a minimum, until a secure email system can be established, it is best not to transmit medical information via email and to inform patients of the risk of such communication. In the case above, the nurse who received the email should respond to the patient by telephone (much more secure). Or she can respond to the patient by email (not including the patient’s message in the return), writing that, because email communications are inherently not confidential, she suggests a phone call or personal visit.
This case also notes that the patient sent the email to the nurse’s personal account, not to an office email account. Sending medical emails to an employee’s personal account raises additional problems of confidentiality and appropriate controls. It should be made clear that employees should not be discussing private medical matters via their own email accounts.
Other forms of social media are also not secure
Similar concerns arise about texting and using Twitter by the second nurse. These activities apparently had been unknown to the physician, but the practice still may be responsible for her actions. These are insecure forms of communication and raise serious ethical and legal concerns.
Other social media pose confidentiality risks as well. For example, a physician was dismissed from a position and reprimanded by the medical board for posting patient information on Facebook,7 and an ObGyn caused problems by posting a nasty note about a patient who showed up late for an appointment.8 Too many patients may not understand that posting on social media is the equivalent of standing on a street corner yelling private information. Social media sites that invite the discussion of personal matters are an invitation to trouble.
Physicians are ethically obliged to protect confidentiality
Professional standards place significant ethical obligations on physicians to protect patient confidentiality. The American Medical Association (AMA) has an ethics opinion on professionalism with social media,9 as does the American College of Obstetricians and Gynecologists (ACOG).10 Another excellent discussion of ethical and practical issues is a joint position paper by the American College of Physicians and the Federation of State Medical Boards.11 Both documents focus attention on issues of confidentiality.
Physicians are legally obliged to protect confidentiality
There are many legal protections for confidentiality that can be implicated by electronic communications and social media. All states provide protection for unwarranted disclosure of private patient information. Such disclosures made electronically are included.12 Indeed, because electronic disclosures may be broadcast more widely, they may be especially dangerous. The misuse of social media may result in license discipline by the state board, regulatory sanctions, or civil liability (rare, but criminal sanctions are a possibility in extreme circumstances).
In addition to state laws regarding confidentiality, there are a number of federal laws that cover confidential medical information. None is more important than the Health Insurance Portability and Accountability Act (HIPAA) and the more recent HITECH amendments (Health Information Technology for Economic and Clinical Health).13 These laws have both privacy provisions and security (including “encryption”) requirements. These are complicated laws but at their core are the notions that health care providers and some others:
- are responsible for maintaining the security and privacy of health information
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.14
- may not transmit (even unintentionally) such information to others without patient permission or legal authority.
A good source of step-by-step information about these laws is “Health information privacy: Covered entities and business associates,” on the US Health and Human Services website.14
HITECH also provides for notice to patients when health information is inappropriately transmitted. Thus, a missing USB flash drive with patient information may require notification to thousands of patients.15 Any consideration of the use of email or social media in medical practice must take into account the HIPAA/HITECH obligations to protect the security of patient health information. There can be serious professional consequences for failing to follow the HIPAA requirements.16
Dual relationships and conflicts of interest
In our hypothetical case, the office manager’s suggestion that the office use Facebook and their website to attract new patients also may raise confidentiality problems. The Facebook suggestion especially needs to be considered carefully. Facebook use is estimated to be 63% to 96% among students and 13% to 47% among health care professionals.17 Facebook is most often seen as an interactive social site; it risks blurring the lines between personal and professional relationships.9 There is a consensus that a physician should not “friend” patients on Facebook. The AMA ethics opinion notes that “physicians must maintain appropriate boundaries of the patient-physician relationship in accordance with professional ethical guidelines, just as they would in any other context.”9
Separate personal and professional contacts
Difficulties with interactive social media are not limited to the physicians in a practice. The problems increase with the number of staff members who post or respond on social media. Control of social media is essential. The practice must ensure that staff members do not slip into inappropriate personal comments and relationships. Staff should understand (and be reminded of) the necessity of separating personal and professional contacts.
Avoid misunderstandings
In addition, whatever the intent of the physician and staff may be, it is essentially impossible to know how patients will interpret interactions on these social media. The very informal, off-the-cuff, chatty way in which Facebook and similar sites are used invites misunderstandings, and maintaining professional boundaries is necessary.
Ground rules
All of this is not to say that professionals should never use Facebook or similar sites. Rather, if used, ground rules need to be established.
Social media communications must:
- be professional and not related to personal matters
- not be used to give medical advice
- be controlled by high level staff
- be reviewed periodically.
Staff training
Particularly for interactive social media (email, texts, Twitter, Facebook, etc), it is essential that there be both clear policies and good staff training (TABLE).9–11,18 There really should be no “making it up as we go along.” Staff on a social media lark of their own can be disastrous for the practice. Policies need to be updated frequently, and staff training reinforced and repeated periodically.
Quality of care and advice
Start with your website
Institutions’ websites are major sources of health care information: Nearly 32% of US adults would be very likely to prefer a hospital based on its website.5 Your website can be an important face of your practice to the community—for good or for bad. On one hand, the practice can control what is on a website and, unlike some social media, it will not be directed to individual patients. Done well, it “provides golden opportunities for marketing physician services, as well as for contributing to public health by providing high-quality online content that is both accurate and understandable to laypeople.”19 Done badly, it can convey incorrect and harmful information and discredit the medical practice that established it.
Your website introduces the practice and settings, but it will serve another purpose to thousands of people who likely will see it over time as a source of credible health information. The importance of ensuring that your website is carefully constructed to provide, or link to, good medical advice that contributes to quality of care cannot be overstated.
A good website begins with a clear statement of the reasons and goals for having the site. Professional design assistance generally is used to create the site, but that design process needs to be overseen by a medical professional to ensure that it conveys the sense of the practice and provides completely accurate information. A homepage of dancing clowns with stethoscopes may seem good to a 20-something-year-old designer, but it is not appropriate for a physician. It will be the practice, not the designer, who is held accountable for the site content. Links to other sites need to be vetted and used with care. Patients and other members of the public may well take the links as carrying the endorsement of the practice and its physicians.
Perhaps the greatest risk of a website is that it will not be kept current. Unfortunately, they do not update themselves. Some knowledgeable staff member must frequently review it to update everything from office hours and personnel to links to other sites. In addition, the physicians periodically must review it to ensure that all medical information is up to date and accurate. Old, outdated information about the office can put off potential patients. Outdated medical information may be harmful to patients who rely on it.
Any professional website should include disclaimers informing users that the site is not intended to establish a professional relationship or to give professional advice. The nature and extent of the disclaimer will depend on the type of information on the site. An example of a particularly thorough disclaimer is the Mayo Clinic disclaimer and terms of use (http://www.mayoclinic.org/about-this-site/terms-conditions-use-policy).
General professionalism
At the end of the day, social media are an outreach from a medical practice and from the profession to the public.20 Failure to treat these platforms with appropriate professional standards may result in professional discipline, damages, or civil penalties. Almost all of the reviews of social media use in health care practice note that the risks of inappropriate use are not only to the individual physician but also to the general medical profession, which may be undermined. Consider posting policies of the relevent state medical boards, the AMA, and ACOG in your office after you have had a discussion with your staff about them.21
The AMA statement includes a provision that a physician seeing unprofessional social media conduct by a colleague has the responsibility to bring that to the attention of the colleague. If the colleague does not correct a significant problem, “the physician should report the matter to appropriate authorities.”9
Bottom line
Any practitioner considering the use of social media must view it as a major step that requires caution, expert assistance, and constant attention to potential privacy, quality, and professionalism issues. If you are considering it, ensure that all staff associated with the practice understand and agree to the established limits on social media use.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- Modahl M, Tompsett L, Moorhead T. Doctors, patients, & social media. Quantia MD website. http://www.quantiamd.com/q-qcp/DoctorsPatientSocialMedia.pdf. Published September 2011. Accessed February 18, 2016.
- Kuberacka A, Wengrojj J, Fabozzi N. Social media use in U.S. healthcare provider institutions: Insights from Frost & Sullivan and iHT2 survey. Frost and Sullivan website. http://ihealthtran.com/pdf/frostiht2survey.pdf. Published August 30, 2011. Accessed February 18, 2016.
- O’Connor ME. How do tech savvy physicians use health technology and social media? Health Care Social Media website. http://hcsmmonitor.com/2014/01/08/how-do-tech-savvy-physicians-use-health-technology-and-social-media/. Published January 8, 2014. Accessed February 18, 2016.
- American Medical Association (AMA) Insurance. 2014 work/life profiles of today’s U.S. physician. AMA Insurance website. https://www.amainsure.com/work-life-profiles-of-todays-us-physician.html. Published April 2014. Accessed February 18, 2016.
- National Research Corporation. 2013 National Market Insights Survey: Health care social media website. https://healthcaresocialmedia.files.wordpress.com/2014/04/nrc-infographiclong.jpg. Accessed February 18, 2016.
- Suby C. Social media in health care: benefits, concerns and guidelines for use. Creat Nurs. 2013;19(3):140–147.
- Conaboy C. For doctors, social media a tricky case. Boston Globe. http://www.boston.com/lifestyle/health/articles/2011/04/20/for_doctors_social_media_a_tricky_case/?page=full. Published April 20, 2011. Accessed February 18, 2016.
- Matyszczyk C. Outcry as ob-gyn uses Facebook to complain about patient. CNET. http://www.cnet.com/news/outcry-as-ob-gyn-uses-facebook-to-complain-about-patient/Minion Pro. Published February 9, 2013. Accessed February 18, 2016.
- American Medical Association (AMA). Opinion 9.124: Professionalism in the use of social media. AMA website. http://www.ama-assn.org/ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion9124.page? Published June 2011. Accessed February 18, 2016.
- American College of Obstetricians and Gynecologists (ACOG) Committee on Professional Liability. ACOG Committee Opinion No. 622: professional use of digital and social media. Obstet Gynecol. 2015;125(2):516-520.
- Farnan JM, Sulmasy LS, Worster BK, et al. Online medical professionalism: patient and public relationships: Policy Statement From the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158(8):620–627.
- Hader A, Drown E. Patient privacy and social media. AANA J. 2010;78(4):270–274.
- Kavoussi SC, Huang JJ, Tsai JC, Kempton JE. HIPAA for physicians in the information age. Conn Med. 2014;78(7):425–427.
- U.S. Department of Health & Human Services (HHS). Health information privacy: Covered entities and business associates. HHS website. http://www.hhs.gov/ocr/privacy/hipaa/understanding/coveredentities/. Published March 14, 2012. Accessed February 18, 2016.
- Perna G. Breach report: lost flash drive at Kaiser Permanente affects 49,000 patients. Healthcare Informatics website. http://www.healthcare-informatics.com/news-item/breach-report-lost-flash-drive-kaiser-permanente-affects-49000-patients. Published December 11, 2013. Accessed February 18, 2016.
- McBride M. How to ensure your social media efforts are HIPAA-compliant. Med Econ. 2012;89:70–74.
- Von Muhlen M, Ohno-Machado L. Reviewing social media use by clinicians. J Am Med Inform Assoc. 2012;19(5):777–781.
- Omurtag K, Turek P. Incorporating social media into practice: a blueprint for reproductive health providers. Clin Obstet Gynecol. 2013;56(3):463–470.
- Radmanesh A, Duszak R, Fitzgerald R. Social media and public outreach: a physician primer. Am J Neuroradiol. 2015;36(7):1223–1224.
- Grajales FJ 3rd, Sheps S, Ho K, Novak-Lauscher H, Eysenbach G. Social media: a review and tutorial of applications in medicine and health care. J Med Internet Res. 2014;16(2):e13.
- ACOG Today. Social media guide: how to comment with patients and spread women’s health messages. American Congress of Obstetricians and Gynecologists website. http://www.acog.org/-/media/ACOG-Today/acogToday201211.pdf. Published November 2012. Accessed February 18, 2016.
- Modahl M, Tompsett L, Moorhead T. Doctors, patients, & social media. Quantia MD website. http://www.quantiamd.com/q-qcp/DoctorsPatientSocialMedia.pdf. Published September 2011. Accessed February 18, 2016.
- Kuberacka A, Wengrojj J, Fabozzi N. Social media use in U.S. healthcare provider institutions: Insights from Frost & Sullivan and iHT2 survey. Frost and Sullivan website. http://ihealthtran.com/pdf/frostiht2survey.pdf. Published August 30, 2011. Accessed February 18, 2016.
- O’Connor ME. How do tech savvy physicians use health technology and social media? Health Care Social Media website. http://hcsmmonitor.com/2014/01/08/how-do-tech-savvy-physicians-use-health-technology-and-social-media/. Published January 8, 2014. Accessed February 18, 2016.
- American Medical Association (AMA) Insurance. 2014 work/life profiles of today’s U.S. physician. AMA Insurance website. https://www.amainsure.com/work-life-profiles-of-todays-us-physician.html. Published April 2014. Accessed February 18, 2016.
- National Research Corporation. 2013 National Market Insights Survey: Health care social media website. https://healthcaresocialmedia.files.wordpress.com/2014/04/nrc-infographiclong.jpg. Accessed February 18, 2016.
- Suby C. Social media in health care: benefits, concerns and guidelines for use. Creat Nurs. 2013;19(3):140–147.
- Conaboy C. For doctors, social media a tricky case. Boston Globe. http://www.boston.com/lifestyle/health/articles/2011/04/20/for_doctors_social_media_a_tricky_case/?page=full. Published April 20, 2011. Accessed February 18, 2016.
- Matyszczyk C. Outcry as ob-gyn uses Facebook to complain about patient. CNET. http://www.cnet.com/news/outcry-as-ob-gyn-uses-facebook-to-complain-about-patient/Minion Pro. Published February 9, 2013. Accessed February 18, 2016.
- American Medical Association (AMA). Opinion 9.124: Professionalism in the use of social media. AMA website. http://www.ama-assn.org/ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion9124.page? Published June 2011. Accessed February 18, 2016.
- American College of Obstetricians and Gynecologists (ACOG) Committee on Professional Liability. ACOG Committee Opinion No. 622: professional use of digital and social media. Obstet Gynecol. 2015;125(2):516-520.
- Farnan JM, Sulmasy LS, Worster BK, et al. Online medical professionalism: patient and public relationships: Policy Statement From the American College of Physicians and the Federation of State Medical Boards. Ann Intern Med. 2013;158(8):620–627.
- Hader A, Drown E. Patient privacy and social media. AANA J. 2010;78(4):270–274.
- Kavoussi SC, Huang JJ, Tsai JC, Kempton JE. HIPAA for physicians in the information age. Conn Med. 2014;78(7):425–427.
- U.S. Department of Health & Human Services (HHS). Health information privacy: Covered entities and business associates. HHS website. http://www.hhs.gov/ocr/privacy/hipaa/understanding/coveredentities/. Published March 14, 2012. Accessed February 18, 2016.
- Perna G. Breach report: lost flash drive at Kaiser Permanente affects 49,000 patients. Healthcare Informatics website. http://www.healthcare-informatics.com/news-item/breach-report-lost-flash-drive-kaiser-permanente-affects-49000-patients. Published December 11, 2013. Accessed February 18, 2016.
- McBride M. How to ensure your social media efforts are HIPAA-compliant. Med Econ. 2012;89:70–74.
- Von Muhlen M, Ohno-Machado L. Reviewing social media use by clinicians. J Am Med Inform Assoc. 2012;19(5):777–781.
- Omurtag K, Turek P. Incorporating social media into practice: a blueprint for reproductive health providers. Clin Obstet Gynecol. 2013;56(3):463–470.
- Radmanesh A, Duszak R, Fitzgerald R. Social media and public outreach: a physician primer. Am J Neuroradiol. 2015;36(7):1223–1224.
- Grajales FJ 3rd, Sheps S, Ho K, Novak-Lauscher H, Eysenbach G. Social media: a review and tutorial of applications in medicine and health care. J Med Internet Res. 2014;16(2):e13.
- ACOG Today. Social media guide: how to comment with patients and spread women’s health messages. American Congress of Obstetricians and Gynecologists website. http://www.acog.org/-/media/ACOG-Today/acogToday201211.pdf. Published November 2012. Accessed February 18, 2016.
In this Article
- Health providers’ use of social media
- Protecting confidentiality
- Creating a social media policy
Postpartum life-threatening strep infection
Postpartum life-threatening strep infection
A pregnant woman received prenatal care from a midwifery practice. A week before her scheduled delivery, the patient became ill with fever and vomiting and visited her midwife. While tests were still pending, the midwife decided to admit the mother to the hospital for induction of labor. The baby was born by vaginal delivery under the midwife’s care. The mother remained in the hospital for observation.
Two days after delivery, the mother began to have nausea, vomiting, and a low-grade fever. The nurse called the midwife, who ordered acetaminophen (Tylenol) but did not come to examine the patient. Two hours later, the nurse notified the midwife that the patient’s condition had worsened and that she was experiencing abdominal pain; the midwife ordered oxycodone. Over the next few hours, the midwife was apprised of the patient’s condition several times by telephone, but she never came to examine the patient nor did she ask her supervising ObGyn to examine the patient.
The next morning, a second midwife noted that the patient was experiencing an itchy rash on her extremities and abdomen. A complete blood count (CBC) showed a “critical lab value” of 44% band neutrophils (normal, 0% to 10% for the hospital laboratory). The second midwife and nurse told the supervising ObGyn that the patient otherwise looked well; he discharged the patient.
At home, the patient’s condition worsened. Her husband called the ObGyn several times and took her to the emergency department (ED) that evening. Her condition deteriorated and she was transferred to another facility where she was diagnosed with a life-threatening Group A Streptococcus (GAS) infection. After weeks of treatment for sepsis, the patient’s foot was amputated.
Patient's claim: The first midwife was negligent in her postpartum treatment of the patient; she should have come to the hospital to examine the patient or have requested that the supervising ObGyn examine the patient. The rash and CBC test results should have initiated further treatment and investigation; the patient should not have been discharged. GAS was not found or treated in a timely manner, resulting in sepsis and amputation.
Defendants' defense: The case was settled during the trial.
Verdict: A $2,500,000 Massachusetts settlement was reached with the midwife, her practice, and the ObGyn.
Failure to follow-up on abnormal Pap
A woman in her 50s reported abnormal bleeding to her gynecologist. Results of an endometrial biopsy were negative for cancer; the gynecologist prescribed hormone therapy. The patient continued to bleed until she entered menopause.
Ten years later, the bleeding returned. Results of a Pap test indicated atypical endometrial cells; an ultrasound showed a markedly abnormal endometrium. The gynecologist recommended a hysteroscopic dilation and curettage (D&C). When he attempted the procedure it ended prematurely because he was unable to enter the patient’s endometrium. The patient’s discharge instructions indicated that she should call the physician for follow up. In a letter to the patient written a month later, the physician discussed the abnormal Pap test results and indicated that the patient had 2 options: another D&C under ultrasound guidance or hysterectomy. He also noted that he would contact the patient’s primary care physician (PCP) for input.
Two years later, the patient returned to the gynecologist because the bleeding, which had never stopped, had increased in intensity. Endometrial cancer was diagnosed.
Patient's claim: The gynecologist never followed up with the patient or her PCP after the incomplete D&C. There is no record that communication ever occurred between the gynecologist and PCP. Lack of follow-up and treatment resulted in progression of the cancer from stage 1 to stage 3C, with a 5-year survivability of 47% (stage 1 survivability is 83%).
Physician's defense: The gynecologist was surprised that no one had ever followed up with the patient. The patient was comparatively negligent for failing to seek medical care for the 2-year period.
Verdict: A $430,000 Minnesota settlement was reached at mediation.
LIVER DISEASE LED TO STILLBIRTH
A 37-year-old woman reported nausea, vomiting, headaches, heartburn, and upper abdominal pain to her ObGyn several times during her third trimester. She had been pregnant before and knew that this pregnancy “felt” different. She went to the ED 1 week before the birth of her child, but she was discharged. The child was stillborn.
Parent's claim: Neither the ObGyn who provided prenatal care nor the on-call ED ObGyn ordered laboratory testing, which would have revealed a rare disease: acute fatty liver of pregnancy. Action could have saved the life of her child.
The patient’s ObGyn disregarded the patient’s reported symptoms; no blood work or liver testing was done. The ObGyn should have recognized the symptoms of liver disease that presented during the third trimester. A diagnosis of liver disease would have initiated induction of labor.
The patient’s expert witness noted that the severity of the third trimester symptoms warranted follow-up testing; the patient should not have had all of those symptoms so late in pregnancy. Testing would have revealed that, by not functioning properly, the liver was creating a toxic environment for the fetus. Labor should have been induced at 36 weeks when the fetal heart testing was still normal.
The ED nurses contacted the on-call ObGyn by telephone to discuss the patient’s symptoms; the ObGyn did not come to the ED to examine the patient or order testing.
The patient suffered emotional distress as a result of the loss of her child.
Defendants' defense: The medical center and the on-call ObGyn settled prior to trial.
The ObGyn claimed that the patient’s symptoms were common for pregnancy and that the disease could not be diagnosed based on the presented symptoms. It was not a violation of the standard of care for the extremely rare liver disease to not be diagnosed. The defense’s expert claimed that the symptoms reported by the patient did not warrant follow-up blood work. There was no way to determine whether or not the fetus died as a result of the mother’s liver disease or nuchal cord involvement.
A placental pathologist noted that the placenta was injured by thrombosis; the fetus’ death was most likely idiopathic. He later acknowledged that thrombosis can be related to liver disease.
Verdict: Jurors were instructed to consider this a personal injury case for the mother due to an unborn fetus’ lacks standing for injury or death under California law. A $160,090 California verdict was returned against the ObGyn who provided prenatal care.
Postpartum life-threatening strep infection
A pregnant woman received prenatal care from a midwifery practice. A week before her scheduled delivery, the patient became ill with fever and vomiting and visited her midwife. While tests were still pending, the midwife decided to admit the mother to the hospital for induction of labor. The baby was born by vaginal delivery under the midwife’s care. The mother remained in the hospital for observation.
Two days after delivery, the mother began to have nausea, vomiting, and a low-grade fever. The nurse called the midwife, who ordered acetaminophen (Tylenol) but did not come to examine the patient. Two hours later, the nurse notified the midwife that the patient’s condition had worsened and that she was experiencing abdominal pain; the midwife ordered oxycodone. Over the next few hours, the midwife was apprised of the patient’s condition several times by telephone, but she never came to examine the patient nor did she ask her supervising ObGyn to examine the patient.
The next morning, a second midwife noted that the patient was experiencing an itchy rash on her extremities and abdomen. A complete blood count (CBC) showed a “critical lab value” of 44% band neutrophils (normal, 0% to 10% for the hospital laboratory). The second midwife and nurse told the supervising ObGyn that the patient otherwise looked well; he discharged the patient.
At home, the patient’s condition worsened. Her husband called the ObGyn several times and took her to the emergency department (ED) that evening. Her condition deteriorated and she was transferred to another facility where she was diagnosed with a life-threatening Group A Streptococcus (GAS) infection. After weeks of treatment for sepsis, the patient’s foot was amputated.
Patient's claim: The first midwife was negligent in her postpartum treatment of the patient; she should have come to the hospital to examine the patient or have requested that the supervising ObGyn examine the patient. The rash and CBC test results should have initiated further treatment and investigation; the patient should not have been discharged. GAS was not found or treated in a timely manner, resulting in sepsis and amputation.
Defendants' defense: The case was settled during the trial.
Verdict: A $2,500,000 Massachusetts settlement was reached with the midwife, her practice, and the ObGyn.
Failure to follow-up on abnormal Pap
A woman in her 50s reported abnormal bleeding to her gynecologist. Results of an endometrial biopsy were negative for cancer; the gynecologist prescribed hormone therapy. The patient continued to bleed until she entered menopause.
Ten years later, the bleeding returned. Results of a Pap test indicated atypical endometrial cells; an ultrasound showed a markedly abnormal endometrium. The gynecologist recommended a hysteroscopic dilation and curettage (D&C). When he attempted the procedure it ended prematurely because he was unable to enter the patient’s endometrium. The patient’s discharge instructions indicated that she should call the physician for follow up. In a letter to the patient written a month later, the physician discussed the abnormal Pap test results and indicated that the patient had 2 options: another D&C under ultrasound guidance or hysterectomy. He also noted that he would contact the patient’s primary care physician (PCP) for input.
Two years later, the patient returned to the gynecologist because the bleeding, which had never stopped, had increased in intensity. Endometrial cancer was diagnosed.
Patient's claim: The gynecologist never followed up with the patient or her PCP after the incomplete D&C. There is no record that communication ever occurred between the gynecologist and PCP. Lack of follow-up and treatment resulted in progression of the cancer from stage 1 to stage 3C, with a 5-year survivability of 47% (stage 1 survivability is 83%).
Physician's defense: The gynecologist was surprised that no one had ever followed up with the patient. The patient was comparatively negligent for failing to seek medical care for the 2-year period.
Verdict: A $430,000 Minnesota settlement was reached at mediation.
LIVER DISEASE LED TO STILLBIRTH
A 37-year-old woman reported nausea, vomiting, headaches, heartburn, and upper abdominal pain to her ObGyn several times during her third trimester. She had been pregnant before and knew that this pregnancy “felt” different. She went to the ED 1 week before the birth of her child, but she was discharged. The child was stillborn.
Parent's claim: Neither the ObGyn who provided prenatal care nor the on-call ED ObGyn ordered laboratory testing, which would have revealed a rare disease: acute fatty liver of pregnancy. Action could have saved the life of her child.
The patient’s ObGyn disregarded the patient’s reported symptoms; no blood work or liver testing was done. The ObGyn should have recognized the symptoms of liver disease that presented during the third trimester. A diagnosis of liver disease would have initiated induction of labor.
The patient’s expert witness noted that the severity of the third trimester symptoms warranted follow-up testing; the patient should not have had all of those symptoms so late in pregnancy. Testing would have revealed that, by not functioning properly, the liver was creating a toxic environment for the fetus. Labor should have been induced at 36 weeks when the fetal heart testing was still normal.
The ED nurses contacted the on-call ObGyn by telephone to discuss the patient’s symptoms; the ObGyn did not come to the ED to examine the patient or order testing.
The patient suffered emotional distress as a result of the loss of her child.
Defendants' defense: The medical center and the on-call ObGyn settled prior to trial.
The ObGyn claimed that the patient’s symptoms were common for pregnancy and that the disease could not be diagnosed based on the presented symptoms. It was not a violation of the standard of care for the extremely rare liver disease to not be diagnosed. The defense’s expert claimed that the symptoms reported by the patient did not warrant follow-up blood work. There was no way to determine whether or not the fetus died as a result of the mother’s liver disease or nuchal cord involvement.
A placental pathologist noted that the placenta was injured by thrombosis; the fetus’ death was most likely idiopathic. He later acknowledged that thrombosis can be related to liver disease.
Verdict: Jurors were instructed to consider this a personal injury case for the mother due to an unborn fetus’ lacks standing for injury or death under California law. A $160,090 California verdict was returned against the ObGyn who provided prenatal care.
Postpartum life-threatening strep infection
A pregnant woman received prenatal care from a midwifery practice. A week before her scheduled delivery, the patient became ill with fever and vomiting and visited her midwife. While tests were still pending, the midwife decided to admit the mother to the hospital for induction of labor. The baby was born by vaginal delivery under the midwife’s care. The mother remained in the hospital for observation.
Two days after delivery, the mother began to have nausea, vomiting, and a low-grade fever. The nurse called the midwife, who ordered acetaminophen (Tylenol) but did not come to examine the patient. Two hours later, the nurse notified the midwife that the patient’s condition had worsened and that she was experiencing abdominal pain; the midwife ordered oxycodone. Over the next few hours, the midwife was apprised of the patient’s condition several times by telephone, but she never came to examine the patient nor did she ask her supervising ObGyn to examine the patient.
The next morning, a second midwife noted that the patient was experiencing an itchy rash on her extremities and abdomen. A complete blood count (CBC) showed a “critical lab value” of 44% band neutrophils (normal, 0% to 10% for the hospital laboratory). The second midwife and nurse told the supervising ObGyn that the patient otherwise looked well; he discharged the patient.
At home, the patient’s condition worsened. Her husband called the ObGyn several times and took her to the emergency department (ED) that evening. Her condition deteriorated and she was transferred to another facility where she was diagnosed with a life-threatening Group A Streptococcus (GAS) infection. After weeks of treatment for sepsis, the patient’s foot was amputated.
Patient's claim: The first midwife was negligent in her postpartum treatment of the patient; she should have come to the hospital to examine the patient or have requested that the supervising ObGyn examine the patient. The rash and CBC test results should have initiated further treatment and investigation; the patient should not have been discharged. GAS was not found or treated in a timely manner, resulting in sepsis and amputation.
Defendants' defense: The case was settled during the trial.
Verdict: A $2,500,000 Massachusetts settlement was reached with the midwife, her practice, and the ObGyn.
Failure to follow-up on abnormal Pap
A woman in her 50s reported abnormal bleeding to her gynecologist. Results of an endometrial biopsy were negative for cancer; the gynecologist prescribed hormone therapy. The patient continued to bleed until she entered menopause.
Ten years later, the bleeding returned. Results of a Pap test indicated atypical endometrial cells; an ultrasound showed a markedly abnormal endometrium. The gynecologist recommended a hysteroscopic dilation and curettage (D&C). When he attempted the procedure it ended prematurely because he was unable to enter the patient’s endometrium. The patient’s discharge instructions indicated that she should call the physician for follow up. In a letter to the patient written a month later, the physician discussed the abnormal Pap test results and indicated that the patient had 2 options: another D&C under ultrasound guidance or hysterectomy. He also noted that he would contact the patient’s primary care physician (PCP) for input.
Two years later, the patient returned to the gynecologist because the bleeding, which had never stopped, had increased in intensity. Endometrial cancer was diagnosed.
Patient's claim: The gynecologist never followed up with the patient or her PCP after the incomplete D&C. There is no record that communication ever occurred between the gynecologist and PCP. Lack of follow-up and treatment resulted in progression of the cancer from stage 1 to stage 3C, with a 5-year survivability of 47% (stage 1 survivability is 83%).
Physician's defense: The gynecologist was surprised that no one had ever followed up with the patient. The patient was comparatively negligent for failing to seek medical care for the 2-year period.
Verdict: A $430,000 Minnesota settlement was reached at mediation.
LIVER DISEASE LED TO STILLBIRTH
A 37-year-old woman reported nausea, vomiting, headaches, heartburn, and upper abdominal pain to her ObGyn several times during her third trimester. She had been pregnant before and knew that this pregnancy “felt” different. She went to the ED 1 week before the birth of her child, but she was discharged. The child was stillborn.
Parent's claim: Neither the ObGyn who provided prenatal care nor the on-call ED ObGyn ordered laboratory testing, which would have revealed a rare disease: acute fatty liver of pregnancy. Action could have saved the life of her child.
The patient’s ObGyn disregarded the patient’s reported symptoms; no blood work or liver testing was done. The ObGyn should have recognized the symptoms of liver disease that presented during the third trimester. A diagnosis of liver disease would have initiated induction of labor.
The patient’s expert witness noted that the severity of the third trimester symptoms warranted follow-up testing; the patient should not have had all of those symptoms so late in pregnancy. Testing would have revealed that, by not functioning properly, the liver was creating a toxic environment for the fetus. Labor should have been induced at 36 weeks when the fetal heart testing was still normal.
The ED nurses contacted the on-call ObGyn by telephone to discuss the patient’s symptoms; the ObGyn did not come to the ED to examine the patient or order testing.
The patient suffered emotional distress as a result of the loss of her child.
Defendants' defense: The medical center and the on-call ObGyn settled prior to trial.
The ObGyn claimed that the patient’s symptoms were common for pregnancy and that the disease could not be diagnosed based on the presented symptoms. It was not a violation of the standard of care for the extremely rare liver disease to not be diagnosed. The defense’s expert claimed that the symptoms reported by the patient did not warrant follow-up blood work. There was no way to determine whether or not the fetus died as a result of the mother’s liver disease or nuchal cord involvement.
A placental pathologist noted that the placenta was injured by thrombosis; the fetus’ death was most likely idiopathic. He later acknowledged that thrombosis can be related to liver disease.
Verdict: Jurors were instructed to consider this a personal injury case for the mother due to an unborn fetus’ lacks standing for injury or death under California law. A $160,090 California verdict was returned against the ObGyn who provided prenatal care.
Additional Medical Verdicts
• Failure to follow-up on abnormal Pap
• Liver disease led to stillbirth
HDAC inhibition may reverse anthracycline resistance in patients with sarcoma
In patients with advanced solid tumors, including sarcoma, the combination of panobinostat, a histone deacetylase (HDAC) inhibitor, and the anthracycline epirubicin demonstrated a correlation between neutropenia, peripheral blood mononucleocyte (PBMC) histone acetylation, and clinical benefit. Acquired topoisomerase resistance was reversed in 8 of 14 patients, suggesting HDAC inhibition reverses resistance.
In 37 evaluable patients, 4 (11%) had partial responses and 17 (46%) had stable disease. The median time to progression and median overall survival were 3.1 (95% CI, 1.8 to 4.6) months and 7.3 (5.9 to 10.3) months, respectively. All four patients with objective partial response had progressed on previous topoisomerase II inhibitors.
“The potential for prolonged treatment with an anthracycline in combination with an HDAC inhibitor speaks to the tolerability of this regimen. This study suggests that further investigation of HDAC inhibition in combination with DNA-damaging agents in defined advanced sarcoma subtypes to validate these preliminary findings is warranted,” wrote Dr. Scott Thomas of the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and colleagues (Ann Oncol. 2016 Feb 21. doi: 10.1093/annonc/mdw044).
HDACs regulate protein acetylation, thereby modulating protein activity and gene expression. Preclinical studies showed that HDAC inhibitors potentiate DNA damaging activity of anthracyclines in various cancer types, including sarcoma.
The phase I trial enrolled patients with metastatic solid tumors in dose escalation cohorts, and 20 patients with advanced sarcoma in the dose expansion cohort at the maximum tolerated dose of 50 mg/day of panobinostat on days 1, 3, and 5, and 75 mg/m2 of epirubicin on day 5.
In total, 24 patients (60%) had at least one grade 3 or 4 adverse event, including neutropenia (45%), leukopenia (35%), lymphopenia (22.5%), thrombocytopenia (17.5%), anemia (15%), and febrile neutropenia (7.5%). Major nonhematologic toxicities of panobinostat were myelotoxicity, nausea/vomiting, and fatigue, which required dose modification in 26% of patients.
In patients with advanced solid tumors, including sarcoma, the combination of panobinostat, a histone deacetylase (HDAC) inhibitor, and the anthracycline epirubicin demonstrated a correlation between neutropenia, peripheral blood mononucleocyte (PBMC) histone acetylation, and clinical benefit. Acquired topoisomerase resistance was reversed in 8 of 14 patients, suggesting HDAC inhibition reverses resistance.
In 37 evaluable patients, 4 (11%) had partial responses and 17 (46%) had stable disease. The median time to progression and median overall survival were 3.1 (95% CI, 1.8 to 4.6) months and 7.3 (5.9 to 10.3) months, respectively. All four patients with objective partial response had progressed on previous topoisomerase II inhibitors.
“The potential for prolonged treatment with an anthracycline in combination with an HDAC inhibitor speaks to the tolerability of this regimen. This study suggests that further investigation of HDAC inhibition in combination with DNA-damaging agents in defined advanced sarcoma subtypes to validate these preliminary findings is warranted,” wrote Dr. Scott Thomas of the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and colleagues (Ann Oncol. 2016 Feb 21. doi: 10.1093/annonc/mdw044).
HDACs regulate protein acetylation, thereby modulating protein activity and gene expression. Preclinical studies showed that HDAC inhibitors potentiate DNA damaging activity of anthracyclines in various cancer types, including sarcoma.
The phase I trial enrolled patients with metastatic solid tumors in dose escalation cohorts, and 20 patients with advanced sarcoma in the dose expansion cohort at the maximum tolerated dose of 50 mg/day of panobinostat on days 1, 3, and 5, and 75 mg/m2 of epirubicin on day 5.
In total, 24 patients (60%) had at least one grade 3 or 4 adverse event, including neutropenia (45%), leukopenia (35%), lymphopenia (22.5%), thrombocytopenia (17.5%), anemia (15%), and febrile neutropenia (7.5%). Major nonhematologic toxicities of panobinostat were myelotoxicity, nausea/vomiting, and fatigue, which required dose modification in 26% of patients.
In patients with advanced solid tumors, including sarcoma, the combination of panobinostat, a histone deacetylase (HDAC) inhibitor, and the anthracycline epirubicin demonstrated a correlation between neutropenia, peripheral blood mononucleocyte (PBMC) histone acetylation, and clinical benefit. Acquired topoisomerase resistance was reversed in 8 of 14 patients, suggesting HDAC inhibition reverses resistance.
In 37 evaluable patients, 4 (11%) had partial responses and 17 (46%) had stable disease. The median time to progression and median overall survival were 3.1 (95% CI, 1.8 to 4.6) months and 7.3 (5.9 to 10.3) months, respectively. All four patients with objective partial response had progressed on previous topoisomerase II inhibitors.
“The potential for prolonged treatment with an anthracycline in combination with an HDAC inhibitor speaks to the tolerability of this regimen. This study suggests that further investigation of HDAC inhibition in combination with DNA-damaging agents in defined advanced sarcoma subtypes to validate these preliminary findings is warranted,” wrote Dr. Scott Thomas of the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and colleagues (Ann Oncol. 2016 Feb 21. doi: 10.1093/annonc/mdw044).
HDACs regulate protein acetylation, thereby modulating protein activity and gene expression. Preclinical studies showed that HDAC inhibitors potentiate DNA damaging activity of anthracyclines in various cancer types, including sarcoma.
The phase I trial enrolled patients with metastatic solid tumors in dose escalation cohorts, and 20 patients with advanced sarcoma in the dose expansion cohort at the maximum tolerated dose of 50 mg/day of panobinostat on days 1, 3, and 5, and 75 mg/m2 of epirubicin on day 5.
In total, 24 patients (60%) had at least one grade 3 or 4 adverse event, including neutropenia (45%), leukopenia (35%), lymphopenia (22.5%), thrombocytopenia (17.5%), anemia (15%), and febrile neutropenia (7.5%). Major nonhematologic toxicities of panobinostat were myelotoxicity, nausea/vomiting, and fatigue, which required dose modification in 26% of patients.
FROM ANNALS OF ONCOLOGY
Key clinical point: Despite prior exposure to multiple regimens, clinical benefit was observed in several patients with advanced solid tumors, including some with sarcoma, who received panobinostat and epirubicin.
Major finding: In 37 evaluable patients, 4 (11%) had partial responses and 17 (46%) had stable disease; acquired topoisomerase resistance was reversed in 8 of 14 patients.
Data source: Phase I trial of 20 patients with metastatic solid tumors in dose escalation cohorts, and 20 patients with advanced sarcoma in the dose expansion cohort.
Disclosures: Research was supported in part by Novartis International AG. Dr. Thomas reported having no disclosures. Dr. Munster received research support from Novartis for this and other clinical trials.
Tender, red thigh
The FP diagnosed an abscess with some surrounding cellulitis. An abscess is a collection of pus in infected tissue. The abscess represents a walled-off infection in which there is a pocket of purulence. In abscesses of the skin, the offending organism is almost always Staphylococcus aureus. In 2004, methicillin-resistant S aureus (MRSA) was the most common identifiable cause of skin and soft-tissue infections among patients presenting to emergency departments in 11 US cities. S aureus was isolated from 76% of these infections and 59% of the infections were community-acquired MRSA.
A clinical cure is often obtained with incision and drainage alone. It is reasonable to obtain wound cultures in high-risk patients, those with signs of systemic infection, and in patients with a history of high recurrence rates.
While the patient’s abscess was beginning to drain spontaneously, it needed to drain some more. The FP performed an incision and drained more than 10 mL of pus. The large abscess cavity was packed with non-iodinated gauze (there is no benefit to iodinated gauze and iodine can be toxic to open tissues). The patient was placed on trimethoprim-sulfamethoxazole DS (double strength) twice daily to cover the surrounding cellulitis. His diabetes medications were renewed and he was reminded of the importance of taking (and not skipping) these important medicines.
When the patient returned 2 days later, the erythema was gone and the packing was removed. The patient was feeling much better and his blood sugar was down to 150 mg/dL. The FP inserted a small amount of packing and told the patient that he could remove it himself in 2 days while in the shower. The patient’s culture grew out MRSA sensitive to trimethoprim-sulfamethoxazole. Two weeks later the patient was fully healed, with only a small scar from the incision remaining.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Abscess. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill;2013:698-701.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com
The FP diagnosed an abscess with some surrounding cellulitis. An abscess is a collection of pus in infected tissue. The abscess represents a walled-off infection in which there is a pocket of purulence. In abscesses of the skin, the offending organism is almost always Staphylococcus aureus. In 2004, methicillin-resistant S aureus (MRSA) was the most common identifiable cause of skin and soft-tissue infections among patients presenting to emergency departments in 11 US cities. S aureus was isolated from 76% of these infections and 59% of the infections were community-acquired MRSA.
A clinical cure is often obtained with incision and drainage alone. It is reasonable to obtain wound cultures in high-risk patients, those with signs of systemic infection, and in patients with a history of high recurrence rates.
While the patient’s abscess was beginning to drain spontaneously, it needed to drain some more. The FP performed an incision and drained more than 10 mL of pus. The large abscess cavity was packed with non-iodinated gauze (there is no benefit to iodinated gauze and iodine can be toxic to open tissues). The patient was placed on trimethoprim-sulfamethoxazole DS (double strength) twice daily to cover the surrounding cellulitis. His diabetes medications were renewed and he was reminded of the importance of taking (and not skipping) these important medicines.
When the patient returned 2 days later, the erythema was gone and the packing was removed. The patient was feeling much better and his blood sugar was down to 150 mg/dL. The FP inserted a small amount of packing and told the patient that he could remove it himself in 2 days while in the shower. The patient’s culture grew out MRSA sensitive to trimethoprim-sulfamethoxazole. Two weeks later the patient was fully healed, with only a small scar from the incision remaining.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Abscess. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill;2013:698-701.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com
The FP diagnosed an abscess with some surrounding cellulitis. An abscess is a collection of pus in infected tissue. The abscess represents a walled-off infection in which there is a pocket of purulence. In abscesses of the skin, the offending organism is almost always Staphylococcus aureus. In 2004, methicillin-resistant S aureus (MRSA) was the most common identifiable cause of skin and soft-tissue infections among patients presenting to emergency departments in 11 US cities. S aureus was isolated from 76% of these infections and 59% of the infections were community-acquired MRSA.
A clinical cure is often obtained with incision and drainage alone. It is reasonable to obtain wound cultures in high-risk patients, those with signs of systemic infection, and in patients with a history of high recurrence rates.
While the patient’s abscess was beginning to drain spontaneously, it needed to drain some more. The FP performed an incision and drained more than 10 mL of pus. The large abscess cavity was packed with non-iodinated gauze (there is no benefit to iodinated gauze and iodine can be toxic to open tissues). The patient was placed on trimethoprim-sulfamethoxazole DS (double strength) twice daily to cover the surrounding cellulitis. His diabetes medications were renewed and he was reminded of the importance of taking (and not skipping) these important medicines.
When the patient returned 2 days later, the erythema was gone and the packing was removed. The patient was feeling much better and his blood sugar was down to 150 mg/dL. The FP inserted a small amount of packing and told the patient that he could remove it himself in 2 days while in the shower. The patient’s culture grew out MRSA sensitive to trimethoprim-sulfamethoxazole. Two weeks later the patient was fully healed, with only a small scar from the incision remaining.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Abscess. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill;2013:698-701.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com
New drug comparable to voriconazole for aspergillosis
The broad-spectrum triazole isavuconazole was as effective as voriconazole in patients with suspected invasive mold disease and caused significantly fewer drug-related adverse events, particularly those of the skin, eyes, and hepatobiliary system, a randomized double-blind study of 516 adults has shown.
The findings suggest that the newer agent “could allow safer therapy” for the primary treatment of invasive aspergillosis and other mold disease than standard therapy with voriconazole, researchers for the phase III, industry-sponsored SECURE trial say in a report published in the Lancet.
The researchers assessed the safety and efficacy of isavuconazole versus voriconazole in patients with invasive mold infection. Patients were recruited from 102 centers across 26 countries over a 7-year period and were randomized to receive either drug.
In the study group of 516 adults with suspected invasive mold infection who received at least one dose of either antifungal drug, isavuconazole proved to be noninferior to voriconazole, by the primary endpoint of all-cause mortality at 6 weeks.
All-cause mortality at 6 weeks in this intention-to-treat group, of whom more than 80% had hematologic malignant disease, was 19% in the isavuconazole group (48 of 258) and 20% (52 of 258) in the voriconazole group.
This primary endpoint was chosen because “it provides the most objective and reproducible effect of therapy, and approximates best the attributable mortality, because deaths due to competing causes occur increasingly after 6 weeks,” Dr. Johan A. Maertensof the UZ Leuven (Belgium), and his associates wrote.
Secondary endpoints included overall response at the end of treatment among patients who were determined by an independent review committee to have proven or probable invasive mold disease – the study’s modified intention-to-treat population – as well as all-cause mortality at day 42 and day 84.
All-cause mortality in this modified intention-to-treat group, as well as in the group of patients found to have proven or probable invasive aspergillosis, specifically, supported the study’s primary findings (Lancet 2016 Feb:387:760-9).
Nearly all patients in the study had at least one treatment-emergent adverse event, and the proportion with serious treatment-emergent adverse events was similar between the treatment groups. However, patients treated with isavuconazole had a significantly lower frequency of hepatobiliary disorders, eye disorders, and skin or subcutaneous disorders.
And overall, significantly fewer patients reported drug-related adverse events with isavuconazole (42% of patients) than with voriconazole (60% of patients). Discontinuation from adverse events, moreover, was significantly less common among isavuconazole-treated patients.
Of the 516 patients in the intention-to-treat group, approximately 53% were confirmed to have proven or probable invasive mold disease, and more than 80% of the mycologically documented cases were Aspergillus infections. Enrollment of patients with possible invasive mold disease at the start “reflects the real-life strategy of early initiation of antifungal treatment,” the investigators say.
Isavuconazonium sulfate was approved in 2015 by the FDA for the treatment of invasive aspergillosis and invasive mucormycosis.
Voriconazole is the current gold standard for the primary treatment of invasive aspergillosis and is recommended for some other mold infections as well, but it is not active against mucormycosis and has “highly variable nonlinear pharmacokinetics in adults,” which has triggered recommendations for drug monitoring, Dr. Maertens and his associates say.
Therapeutic monitoring aimed at individualizing dosage regimes in order to improve response and prevent adverse events became the standard of care in some institutions during the study period (2007-2013). The study used the labeled dose of voriconazole, however, and did not address the efficacy of either drug with therapeutic drug monitoring.
The study also excluded patients with AIDS, abnormal liver or renal function, and those receiving antifungal prophylaxis with a mold-active triazole – factors that may limit generalizability of the findings, the investigators note.
Funding for the study was provided by Astellas Pharma Global Development and Basilea Pharmaceutica International.
Dr. Maertens disclosed receiving grants and fees from Bio-Rad, personal fees and nonfinancial support from Astellas and Basilea, and grants, fees and support from Gilead Sciences, Merck Sharp and Dohme, and Pfizer during the study.
The advantages of isavuconazole over voriconazole include its broader spectrum of activity, linear pharmacokinetics, once-daily dosing after the loading dose, and fewer CYP enzyme-mediated drug-drug interactions. This trial represents important progress in widening the therapeutic options for mold infections.
Numerous issues require further evaluation, including the effectiveness of isavuconazole after mold-active triazole prophylaxis, which is a common practice in patients at risk for mold infection, and the agent’s effectiveness against molds other than Aspergillus.
In addition, experience in a more varied patient population will be required to be certain that therapeutic drug monitoring is unnecessary.
Cost-effectiveness must also be explored. Isavuconazole will probably achieve an equivalent recommendation as voriconazole for initial treatment of aspergillosis in clinical guidelines, but voriconazole will soon come off patent in many countries and new formulations of posaconazole are now available.
That the finding that 42-day mortality in both treatment groups (isavuconazole and voriconazole) was no different than the mortality seen in research done 15 years ago on voriconazole treatment of aspergillosis is disappointing and suggests that we need to do better with the prevention and early detection of mold infection in vulnerable patients.
Dr. Monica A. Slavin and Dr. Karin A. Thursky are affiliated with the Peter MacCallum Cancer Centre, East Melbourne, Australia. Their comments are excerpted from an accompanying editorial in the Lancet. Dr. Slavin reported receiving grants from Merck, Gilead, and Pfizer. Dr. Thursky reported no disclosures.
The advantages of isavuconazole over voriconazole include its broader spectrum of activity, linear pharmacokinetics, once-daily dosing after the loading dose, and fewer CYP enzyme-mediated drug-drug interactions. This trial represents important progress in widening the therapeutic options for mold infections.
Numerous issues require further evaluation, including the effectiveness of isavuconazole after mold-active triazole prophylaxis, which is a common practice in patients at risk for mold infection, and the agent’s effectiveness against molds other than Aspergillus.
In addition, experience in a more varied patient population will be required to be certain that therapeutic drug monitoring is unnecessary.
Cost-effectiveness must also be explored. Isavuconazole will probably achieve an equivalent recommendation as voriconazole for initial treatment of aspergillosis in clinical guidelines, but voriconazole will soon come off patent in many countries and new formulations of posaconazole are now available.
That the finding that 42-day mortality in both treatment groups (isavuconazole and voriconazole) was no different than the mortality seen in research done 15 years ago on voriconazole treatment of aspergillosis is disappointing and suggests that we need to do better with the prevention and early detection of mold infection in vulnerable patients.
Dr. Monica A. Slavin and Dr. Karin A. Thursky are affiliated with the Peter MacCallum Cancer Centre, East Melbourne, Australia. Their comments are excerpted from an accompanying editorial in the Lancet. Dr. Slavin reported receiving grants from Merck, Gilead, and Pfizer. Dr. Thursky reported no disclosures.
The advantages of isavuconazole over voriconazole include its broader spectrum of activity, linear pharmacokinetics, once-daily dosing after the loading dose, and fewer CYP enzyme-mediated drug-drug interactions. This trial represents important progress in widening the therapeutic options for mold infections.
Numerous issues require further evaluation, including the effectiveness of isavuconazole after mold-active triazole prophylaxis, which is a common practice in patients at risk for mold infection, and the agent’s effectiveness against molds other than Aspergillus.
In addition, experience in a more varied patient population will be required to be certain that therapeutic drug monitoring is unnecessary.
Cost-effectiveness must also be explored. Isavuconazole will probably achieve an equivalent recommendation as voriconazole for initial treatment of aspergillosis in clinical guidelines, but voriconazole will soon come off patent in many countries and new formulations of posaconazole are now available.
That the finding that 42-day mortality in both treatment groups (isavuconazole and voriconazole) was no different than the mortality seen in research done 15 years ago on voriconazole treatment of aspergillosis is disappointing and suggests that we need to do better with the prevention and early detection of mold infection in vulnerable patients.
Dr. Monica A. Slavin and Dr. Karin A. Thursky are affiliated with the Peter MacCallum Cancer Centre, East Melbourne, Australia. Their comments are excerpted from an accompanying editorial in the Lancet. Dr. Slavin reported receiving grants from Merck, Gilead, and Pfizer. Dr. Thursky reported no disclosures.
The broad-spectrum triazole isavuconazole was as effective as voriconazole in patients with suspected invasive mold disease and caused significantly fewer drug-related adverse events, particularly those of the skin, eyes, and hepatobiliary system, a randomized double-blind study of 516 adults has shown.
The findings suggest that the newer agent “could allow safer therapy” for the primary treatment of invasive aspergillosis and other mold disease than standard therapy with voriconazole, researchers for the phase III, industry-sponsored SECURE trial say in a report published in the Lancet.
The researchers assessed the safety and efficacy of isavuconazole versus voriconazole in patients with invasive mold infection. Patients were recruited from 102 centers across 26 countries over a 7-year period and were randomized to receive either drug.
In the study group of 516 adults with suspected invasive mold infection who received at least one dose of either antifungal drug, isavuconazole proved to be noninferior to voriconazole, by the primary endpoint of all-cause mortality at 6 weeks.
All-cause mortality at 6 weeks in this intention-to-treat group, of whom more than 80% had hematologic malignant disease, was 19% in the isavuconazole group (48 of 258) and 20% (52 of 258) in the voriconazole group.
This primary endpoint was chosen because “it provides the most objective and reproducible effect of therapy, and approximates best the attributable mortality, because deaths due to competing causes occur increasingly after 6 weeks,” Dr. Johan A. Maertensof the UZ Leuven (Belgium), and his associates wrote.
Secondary endpoints included overall response at the end of treatment among patients who were determined by an independent review committee to have proven or probable invasive mold disease – the study’s modified intention-to-treat population – as well as all-cause mortality at day 42 and day 84.
All-cause mortality in this modified intention-to-treat group, as well as in the group of patients found to have proven or probable invasive aspergillosis, specifically, supported the study’s primary findings (Lancet 2016 Feb:387:760-9).
Nearly all patients in the study had at least one treatment-emergent adverse event, and the proportion with serious treatment-emergent adverse events was similar between the treatment groups. However, patients treated with isavuconazole had a significantly lower frequency of hepatobiliary disorders, eye disorders, and skin or subcutaneous disorders.
And overall, significantly fewer patients reported drug-related adverse events with isavuconazole (42% of patients) than with voriconazole (60% of patients). Discontinuation from adverse events, moreover, was significantly less common among isavuconazole-treated patients.
Of the 516 patients in the intention-to-treat group, approximately 53% were confirmed to have proven or probable invasive mold disease, and more than 80% of the mycologically documented cases were Aspergillus infections. Enrollment of patients with possible invasive mold disease at the start “reflects the real-life strategy of early initiation of antifungal treatment,” the investigators say.
Isavuconazonium sulfate was approved in 2015 by the FDA for the treatment of invasive aspergillosis and invasive mucormycosis.
Voriconazole is the current gold standard for the primary treatment of invasive aspergillosis and is recommended for some other mold infections as well, but it is not active against mucormycosis and has “highly variable nonlinear pharmacokinetics in adults,” which has triggered recommendations for drug monitoring, Dr. Maertens and his associates say.
Therapeutic monitoring aimed at individualizing dosage regimes in order to improve response and prevent adverse events became the standard of care in some institutions during the study period (2007-2013). The study used the labeled dose of voriconazole, however, and did not address the efficacy of either drug with therapeutic drug monitoring.
The study also excluded patients with AIDS, abnormal liver or renal function, and those receiving antifungal prophylaxis with a mold-active triazole – factors that may limit generalizability of the findings, the investigators note.
Funding for the study was provided by Astellas Pharma Global Development and Basilea Pharmaceutica International.
Dr. Maertens disclosed receiving grants and fees from Bio-Rad, personal fees and nonfinancial support from Astellas and Basilea, and grants, fees and support from Gilead Sciences, Merck Sharp and Dohme, and Pfizer during the study.
The broad-spectrum triazole isavuconazole was as effective as voriconazole in patients with suspected invasive mold disease and caused significantly fewer drug-related adverse events, particularly those of the skin, eyes, and hepatobiliary system, a randomized double-blind study of 516 adults has shown.
The findings suggest that the newer agent “could allow safer therapy” for the primary treatment of invasive aspergillosis and other mold disease than standard therapy with voriconazole, researchers for the phase III, industry-sponsored SECURE trial say in a report published in the Lancet.
The researchers assessed the safety and efficacy of isavuconazole versus voriconazole in patients with invasive mold infection. Patients were recruited from 102 centers across 26 countries over a 7-year period and were randomized to receive either drug.
In the study group of 516 adults with suspected invasive mold infection who received at least one dose of either antifungal drug, isavuconazole proved to be noninferior to voriconazole, by the primary endpoint of all-cause mortality at 6 weeks.
All-cause mortality at 6 weeks in this intention-to-treat group, of whom more than 80% had hematologic malignant disease, was 19% in the isavuconazole group (48 of 258) and 20% (52 of 258) in the voriconazole group.
This primary endpoint was chosen because “it provides the most objective and reproducible effect of therapy, and approximates best the attributable mortality, because deaths due to competing causes occur increasingly after 6 weeks,” Dr. Johan A. Maertensof the UZ Leuven (Belgium), and his associates wrote.
Secondary endpoints included overall response at the end of treatment among patients who were determined by an independent review committee to have proven or probable invasive mold disease – the study’s modified intention-to-treat population – as well as all-cause mortality at day 42 and day 84.
All-cause mortality in this modified intention-to-treat group, as well as in the group of patients found to have proven or probable invasive aspergillosis, specifically, supported the study’s primary findings (Lancet 2016 Feb:387:760-9).
Nearly all patients in the study had at least one treatment-emergent adverse event, and the proportion with serious treatment-emergent adverse events was similar between the treatment groups. However, patients treated with isavuconazole had a significantly lower frequency of hepatobiliary disorders, eye disorders, and skin or subcutaneous disorders.
And overall, significantly fewer patients reported drug-related adverse events with isavuconazole (42% of patients) than with voriconazole (60% of patients). Discontinuation from adverse events, moreover, was significantly less common among isavuconazole-treated patients.
Of the 516 patients in the intention-to-treat group, approximately 53% were confirmed to have proven or probable invasive mold disease, and more than 80% of the mycologically documented cases were Aspergillus infections. Enrollment of patients with possible invasive mold disease at the start “reflects the real-life strategy of early initiation of antifungal treatment,” the investigators say.
Isavuconazonium sulfate was approved in 2015 by the FDA for the treatment of invasive aspergillosis and invasive mucormycosis.
Voriconazole is the current gold standard for the primary treatment of invasive aspergillosis and is recommended for some other mold infections as well, but it is not active against mucormycosis and has “highly variable nonlinear pharmacokinetics in adults,” which has triggered recommendations for drug monitoring, Dr. Maertens and his associates say.
Therapeutic monitoring aimed at individualizing dosage regimes in order to improve response and prevent adverse events became the standard of care in some institutions during the study period (2007-2013). The study used the labeled dose of voriconazole, however, and did not address the efficacy of either drug with therapeutic drug monitoring.
The study also excluded patients with AIDS, abnormal liver or renal function, and those receiving antifungal prophylaxis with a mold-active triazole – factors that may limit generalizability of the findings, the investigators note.
Funding for the study was provided by Astellas Pharma Global Development and Basilea Pharmaceutica International.
Dr. Maertens disclosed receiving grants and fees from Bio-Rad, personal fees and nonfinancial support from Astellas and Basilea, and grants, fees and support from Gilead Sciences, Merck Sharp and Dohme, and Pfizer during the study.
FROM THE LANCET
Key clinical point: Isavuconazole is an appropriate alternative for primary treatment of suspected invasive aspergillosis.
Major finding: All-cause mortality at 6 weeks in the intention-to-treat group of 516 patients was 19% with isavuconazole and 20% with voriconazole. Fewer drug-related adverse events were reported with isavuconazole, however (42% vs. 60% of patients).
Data source: A phase III randomized, double-blind noninferiority trial – the SECURE trial – comparing the safety and efficacy of intravenous and oral formulations of isavuconazole and voriconazole for the primary treatment of invasive aspergillosis and disease caused by other molds.
Disclosures: Funding for the study was provided by Astellas Pharma Global Development and Basilea Pharmaceutica International. Dr. Maertens disclosed receiving grants and fees from Bio-Rad, personal fees and nonfinancial support from Astellas and Basilea, and grants, fees, and support from Gilead Sciences, Merck Sharp and Dohme, and Pfizer, during the study.
Uveitis in juvenile idiopathic arthritis may be preventable
MAUI, HAWAII – Uveitis is a common, highly destructive manifestation of juvenile idiopathic arthritis that is readily treatable when caught early, Dr. Anne M. Stevens said at the 2016 Rheumatology Winter Clinical Symposium.
Moreover, recent evidence from Germany suggests that uveitis may actually be preventable through early aggressive anti-inflammatory therapy, noted Dr. Stevens, a pediatric rheumatologist at Seattle Children’s Hospital and the University of Washington.
She cited a report from investigators participating in the National Pediatric Rheumatological Database in Germany. This large study included 3,512 juvenile idiopathic arthritis (JIA) patients with a mean age at arthritis onset of 7.8 years, a disease duration of less than 12 months at enrollment, and a mean follow-up of 3.6 years.
Uveitis occurred in 5.1% of patients within the first year after onset of JIA, and in another 7.1% following the first year. The key finding in the German study was that aggressive disease-modifying antirheumatic drug (DMARD) therapy during the year prior to uveitis significantly reduced the likelihood of developing this complication. Children on methotrexate during that time period were 37% less likely to develop uveitis than were those not on a DMARD. Moreover, patients placed on methotrexate within the first year after being diagnosed with JIA had a 71% relative risk reduction.
Patients on a tumor necrosis factor inhibitor during the year prior to uveitis had a 44% reduction in the risk of developing the eye complication. Most impressive of all, children on both methotrexate and a TNF inhibitor during the year prior to uveitis had a whopping 90% reduction in the risk of developing uveitis, compared with those not on a DMARD (Arthritis Care Res [Hoboken]. 2016 Jan;68[1]:46-54).
“I was fascinated by this study showing that treatment with two types of therapy may be preventive,” Dr. Stevens commented. “If this is substantiated in another large population-based cohort, it will be interesting to see if practice moves to treating the ANA [antinuclear antibody]–positive oligo JIA patients very early with TNF [tumor necrosis factor] inhibitors and methotrexate to prevent uveitis.”
The logic behind this aggressive preventive therapy lies in the fact that while uveitis occurs in about 20% of patients with oligoarticular JIA overall, roughly 90% of cases involve ANA-positive patients. For this reason, guidelines recommend slit lamp examinations every 3 months for a year in patients with young-onset, ANA-positive JIA.
“Imagine trying to do a slit lamp exam on a 2-year-old. It really helps to send these kids to a pediatric ophthalmologist who’s done a lot of them,” she advised.
The uveitis of JIA is typically anterior, asymptomatic, and low grade. It’s also a leading cause of blindness in childhood. Complications include band keratopathy, glaucomatous optic neuropathy, cataracts, and maculopathy.
“If we catch uveitis early, we can treat this disease really well now,” according to Dr. Stevens.
The initial therapy is short-term topical steroids. It’s important to keep in touch with the ophthalmologist regarding the slit lamp findings, however, because ophthalmologists generally tend to favor longer-term topical steroid therapy, while rheumatologists are appropriately primed to push on to more aggressive systemic therapy very quickly.
The first-line systemic agent for treatment of uveitis in patients with JIA is methotrexate at 1 mg/kg/week. If that doesn’t achieve satisfactory results, pediatric rheumatologists are quick to move on to second-line therapy with cyclosporine, azathioprine, or mycophenolate (CellCept).
“We move on fairly quickly if need be to TNF inhibitors, and we go with very high doses. The literature for infliximab [Remicade] is supportive of 20 mg/kg every 4 weeks. That’s what I use,” she continued.
High-dose adalimumab (Humira) is another option (J Rheumatol. 2013 Jan;40[1]:74-9). However, etanercept (Enbrel) is not effective for this condition. The use of abatacept (Orencia) or rituximab (Rituxan) in refractory patients is supported by favorable case reports.
Dr. Stevens reported having no financial interests relevant to her presentation.
MAUI, HAWAII – Uveitis is a common, highly destructive manifestation of juvenile idiopathic arthritis that is readily treatable when caught early, Dr. Anne M. Stevens said at the 2016 Rheumatology Winter Clinical Symposium.
Moreover, recent evidence from Germany suggests that uveitis may actually be preventable through early aggressive anti-inflammatory therapy, noted Dr. Stevens, a pediatric rheumatologist at Seattle Children’s Hospital and the University of Washington.
She cited a report from investigators participating in the National Pediatric Rheumatological Database in Germany. This large study included 3,512 juvenile idiopathic arthritis (JIA) patients with a mean age at arthritis onset of 7.8 years, a disease duration of less than 12 months at enrollment, and a mean follow-up of 3.6 years.
Uveitis occurred in 5.1% of patients within the first year after onset of JIA, and in another 7.1% following the first year. The key finding in the German study was that aggressive disease-modifying antirheumatic drug (DMARD) therapy during the year prior to uveitis significantly reduced the likelihood of developing this complication. Children on methotrexate during that time period were 37% less likely to develop uveitis than were those not on a DMARD. Moreover, patients placed on methotrexate within the first year after being diagnosed with JIA had a 71% relative risk reduction.
Patients on a tumor necrosis factor inhibitor during the year prior to uveitis had a 44% reduction in the risk of developing the eye complication. Most impressive of all, children on both methotrexate and a TNF inhibitor during the year prior to uveitis had a whopping 90% reduction in the risk of developing uveitis, compared with those not on a DMARD (Arthritis Care Res [Hoboken]. 2016 Jan;68[1]:46-54).
“I was fascinated by this study showing that treatment with two types of therapy may be preventive,” Dr. Stevens commented. “If this is substantiated in another large population-based cohort, it will be interesting to see if practice moves to treating the ANA [antinuclear antibody]–positive oligo JIA patients very early with TNF [tumor necrosis factor] inhibitors and methotrexate to prevent uveitis.”
The logic behind this aggressive preventive therapy lies in the fact that while uveitis occurs in about 20% of patients with oligoarticular JIA overall, roughly 90% of cases involve ANA-positive patients. For this reason, guidelines recommend slit lamp examinations every 3 months for a year in patients with young-onset, ANA-positive JIA.
“Imagine trying to do a slit lamp exam on a 2-year-old. It really helps to send these kids to a pediatric ophthalmologist who’s done a lot of them,” she advised.
The uveitis of JIA is typically anterior, asymptomatic, and low grade. It’s also a leading cause of blindness in childhood. Complications include band keratopathy, glaucomatous optic neuropathy, cataracts, and maculopathy.
“If we catch uveitis early, we can treat this disease really well now,” according to Dr. Stevens.
The initial therapy is short-term topical steroids. It’s important to keep in touch with the ophthalmologist regarding the slit lamp findings, however, because ophthalmologists generally tend to favor longer-term topical steroid therapy, while rheumatologists are appropriately primed to push on to more aggressive systemic therapy very quickly.
The first-line systemic agent for treatment of uveitis in patients with JIA is methotrexate at 1 mg/kg/week. If that doesn’t achieve satisfactory results, pediatric rheumatologists are quick to move on to second-line therapy with cyclosporine, azathioprine, or mycophenolate (CellCept).
“We move on fairly quickly if need be to TNF inhibitors, and we go with very high doses. The literature for infliximab [Remicade] is supportive of 20 mg/kg every 4 weeks. That’s what I use,” she continued.
High-dose adalimumab (Humira) is another option (J Rheumatol. 2013 Jan;40[1]:74-9). However, etanercept (Enbrel) is not effective for this condition. The use of abatacept (Orencia) or rituximab (Rituxan) in refractory patients is supported by favorable case reports.
Dr. Stevens reported having no financial interests relevant to her presentation.
MAUI, HAWAII – Uveitis is a common, highly destructive manifestation of juvenile idiopathic arthritis that is readily treatable when caught early, Dr. Anne M. Stevens said at the 2016 Rheumatology Winter Clinical Symposium.
Moreover, recent evidence from Germany suggests that uveitis may actually be preventable through early aggressive anti-inflammatory therapy, noted Dr. Stevens, a pediatric rheumatologist at Seattle Children’s Hospital and the University of Washington.
She cited a report from investigators participating in the National Pediatric Rheumatological Database in Germany. This large study included 3,512 juvenile idiopathic arthritis (JIA) patients with a mean age at arthritis onset of 7.8 years, a disease duration of less than 12 months at enrollment, and a mean follow-up of 3.6 years.
Uveitis occurred in 5.1% of patients within the first year after onset of JIA, and in another 7.1% following the first year. The key finding in the German study was that aggressive disease-modifying antirheumatic drug (DMARD) therapy during the year prior to uveitis significantly reduced the likelihood of developing this complication. Children on methotrexate during that time period were 37% less likely to develop uveitis than were those not on a DMARD. Moreover, patients placed on methotrexate within the first year after being diagnosed with JIA had a 71% relative risk reduction.
Patients on a tumor necrosis factor inhibitor during the year prior to uveitis had a 44% reduction in the risk of developing the eye complication. Most impressive of all, children on both methotrexate and a TNF inhibitor during the year prior to uveitis had a whopping 90% reduction in the risk of developing uveitis, compared with those not on a DMARD (Arthritis Care Res [Hoboken]. 2016 Jan;68[1]:46-54).
“I was fascinated by this study showing that treatment with two types of therapy may be preventive,” Dr. Stevens commented. “If this is substantiated in another large population-based cohort, it will be interesting to see if practice moves to treating the ANA [antinuclear antibody]–positive oligo JIA patients very early with TNF [tumor necrosis factor] inhibitors and methotrexate to prevent uveitis.”
The logic behind this aggressive preventive therapy lies in the fact that while uveitis occurs in about 20% of patients with oligoarticular JIA overall, roughly 90% of cases involve ANA-positive patients. For this reason, guidelines recommend slit lamp examinations every 3 months for a year in patients with young-onset, ANA-positive JIA.
“Imagine trying to do a slit lamp exam on a 2-year-old. It really helps to send these kids to a pediatric ophthalmologist who’s done a lot of them,” she advised.
The uveitis of JIA is typically anterior, asymptomatic, and low grade. It’s also a leading cause of blindness in childhood. Complications include band keratopathy, glaucomatous optic neuropathy, cataracts, and maculopathy.
“If we catch uveitis early, we can treat this disease really well now,” according to Dr. Stevens.
The initial therapy is short-term topical steroids. It’s important to keep in touch with the ophthalmologist regarding the slit lamp findings, however, because ophthalmologists generally tend to favor longer-term topical steroid therapy, while rheumatologists are appropriately primed to push on to more aggressive systemic therapy very quickly.
The first-line systemic agent for treatment of uveitis in patients with JIA is methotrexate at 1 mg/kg/week. If that doesn’t achieve satisfactory results, pediatric rheumatologists are quick to move on to second-line therapy with cyclosporine, azathioprine, or mycophenolate (CellCept).
“We move on fairly quickly if need be to TNF inhibitors, and we go with very high doses. The literature for infliximab [Remicade] is supportive of 20 mg/kg every 4 weeks. That’s what I use,” she continued.
High-dose adalimumab (Humira) is another option (J Rheumatol. 2013 Jan;40[1]:74-9). However, etanercept (Enbrel) is not effective for this condition. The use of abatacept (Orencia) or rituximab (Rituxan) in refractory patients is supported by favorable case reports.
Dr. Stevens reported having no financial interests relevant to her presentation.
EXPERT ANALYSIS FROM RWCS 2016
Utah Health Sciences Center Realizes Significant Savings from Cost-Analysis Tool
Most healthcare systems don’t know or understand their cost of care at the unit that matters, i.e., the actual cost of care for an individual patient. Knowing this, University of Utah Health Sciences Center (UHSC) sought to develop methodologies using clinical data that would enable it to determine cost of care. Ultimately, a team led by systems, data, and analytics personnel, and including hospitalists, created an analytics framework known as value driven outcomes (VDO) using an agile methodology. Evaluation consisted of measurement against project objectives, including implementation timeliness, system performance, completeness, accuracy, extensibility, adoption, satisfaction, and the ability to support value improvement.1
“The initial results of employing the cost-savings tool have been exciting. For example, a 30% reduction in the cost of an orthopedic joint replacement—an inpatient hospital procedure—was realized,” says Robert C. Pendleton, MD, chief medical quality officer and professor of medicine at the University of Utah in Salt Lake City.
In another instance, hospitalists at UHSC led an initiative to reduce the use of unnecessary laboratory tests. That resulted in $400,000 in cost savings in the first year.
Another UHSC hospitalist used data to reduce unnecessary telemetry utilization by 60% on the hospitalist service.
“The accumulative impact of these initiatives, among others, will be a savings of millions of dollars annually by making process changes,” says Dr. Pendleton, a hospitalist who served on the executive steering committee that designed the web-based tool. “Our early experience shows that there is a lot of waste in the healthcare system.”
Along with the costing tool, quality and outcome measures were integrated so that the actual value of care (best outcomes at the lowest cost) could be assessed and incrementally improved.
Widespread Use?
Dr. Pendleton believes the tool could become popular among hospitalists, explaining that the field is known as the “leading system thinkers in healthcare.”
“By their nature, if you give them meaningful data and they can compare themselves to their peers, they are highly engaged to determine where waste in their hospitals exists and to lead improvement efforts,” he says.
UHSC is currently in the process of rolling out the VDO tool systemwide to its 1,200 faculty members. The rollout will take six to 12 months. In the next year, it’s also working to define and integrate outcome measures for the top 50 medical conditions it treats, which will allow it to robustly assess the value of care delivery.
“Success will equally be to increase quality without an increase in cost,” he says.
What’s more, Dr. Pendleton says efforts are in the works to make the self-intuitive system available to institutions nationwide. For example, the center is working to address technical complexities resulting from the use of multiple electronic health record vendors.
“The goal is to make it usable in every environment so we can support other organizations in the most effective ways,” he says.
Although the tool is very easy to use, stakeholders are providing feedback that allows for continuous improvements in how data are presented.
“Anyone with basic computer skills and the ability to interpret bar charts and basic graphs can use it,” Dr. Pendleton says. The center also offers a 15-minute, confidential, training module and a one-hour, one-on-one training session with an analyst. TH
Karen Appold is a freelance author in Pennsylvania.
Reference
- Kawamoto K, Martin CJ, Williams K, et al. Value driven outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015;22(1):223-235.
Most healthcare systems don’t know or understand their cost of care at the unit that matters, i.e., the actual cost of care for an individual patient. Knowing this, University of Utah Health Sciences Center (UHSC) sought to develop methodologies using clinical data that would enable it to determine cost of care. Ultimately, a team led by systems, data, and analytics personnel, and including hospitalists, created an analytics framework known as value driven outcomes (VDO) using an agile methodology. Evaluation consisted of measurement against project objectives, including implementation timeliness, system performance, completeness, accuracy, extensibility, adoption, satisfaction, and the ability to support value improvement.1
“The initial results of employing the cost-savings tool have been exciting. For example, a 30% reduction in the cost of an orthopedic joint replacement—an inpatient hospital procedure—was realized,” says Robert C. Pendleton, MD, chief medical quality officer and professor of medicine at the University of Utah in Salt Lake City.
In another instance, hospitalists at UHSC led an initiative to reduce the use of unnecessary laboratory tests. That resulted in $400,000 in cost savings in the first year.
Another UHSC hospitalist used data to reduce unnecessary telemetry utilization by 60% on the hospitalist service.
“The accumulative impact of these initiatives, among others, will be a savings of millions of dollars annually by making process changes,” says Dr. Pendleton, a hospitalist who served on the executive steering committee that designed the web-based tool. “Our early experience shows that there is a lot of waste in the healthcare system.”
Along with the costing tool, quality and outcome measures were integrated so that the actual value of care (best outcomes at the lowest cost) could be assessed and incrementally improved.
Widespread Use?
Dr. Pendleton believes the tool could become popular among hospitalists, explaining that the field is known as the “leading system thinkers in healthcare.”
“By their nature, if you give them meaningful data and they can compare themselves to their peers, they are highly engaged to determine where waste in their hospitals exists and to lead improvement efforts,” he says.
UHSC is currently in the process of rolling out the VDO tool systemwide to its 1,200 faculty members. The rollout will take six to 12 months. In the next year, it’s also working to define and integrate outcome measures for the top 50 medical conditions it treats, which will allow it to robustly assess the value of care delivery.
“Success will equally be to increase quality without an increase in cost,” he says.
What’s more, Dr. Pendleton says efforts are in the works to make the self-intuitive system available to institutions nationwide. For example, the center is working to address technical complexities resulting from the use of multiple electronic health record vendors.
“The goal is to make it usable in every environment so we can support other organizations in the most effective ways,” he says.
Although the tool is very easy to use, stakeholders are providing feedback that allows for continuous improvements in how data are presented.
“Anyone with basic computer skills and the ability to interpret bar charts and basic graphs can use it,” Dr. Pendleton says. The center also offers a 15-minute, confidential, training module and a one-hour, one-on-one training session with an analyst. TH
Karen Appold is a freelance author in Pennsylvania.
Reference
- Kawamoto K, Martin CJ, Williams K, et al. Value driven outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015;22(1):223-235.
Most healthcare systems don’t know or understand their cost of care at the unit that matters, i.e., the actual cost of care for an individual patient. Knowing this, University of Utah Health Sciences Center (UHSC) sought to develop methodologies using clinical data that would enable it to determine cost of care. Ultimately, a team led by systems, data, and analytics personnel, and including hospitalists, created an analytics framework known as value driven outcomes (VDO) using an agile methodology. Evaluation consisted of measurement against project objectives, including implementation timeliness, system performance, completeness, accuracy, extensibility, adoption, satisfaction, and the ability to support value improvement.1
“The initial results of employing the cost-savings tool have been exciting. For example, a 30% reduction in the cost of an orthopedic joint replacement—an inpatient hospital procedure—was realized,” says Robert C. Pendleton, MD, chief medical quality officer and professor of medicine at the University of Utah in Salt Lake City.
In another instance, hospitalists at UHSC led an initiative to reduce the use of unnecessary laboratory tests. That resulted in $400,000 in cost savings in the first year.
Another UHSC hospitalist used data to reduce unnecessary telemetry utilization by 60% on the hospitalist service.
“The accumulative impact of these initiatives, among others, will be a savings of millions of dollars annually by making process changes,” says Dr. Pendleton, a hospitalist who served on the executive steering committee that designed the web-based tool. “Our early experience shows that there is a lot of waste in the healthcare system.”
Along with the costing tool, quality and outcome measures were integrated so that the actual value of care (best outcomes at the lowest cost) could be assessed and incrementally improved.
Widespread Use?
Dr. Pendleton believes the tool could become popular among hospitalists, explaining that the field is known as the “leading system thinkers in healthcare.”
“By their nature, if you give them meaningful data and they can compare themselves to their peers, they are highly engaged to determine where waste in their hospitals exists and to lead improvement efforts,” he says.
UHSC is currently in the process of rolling out the VDO tool systemwide to its 1,200 faculty members. The rollout will take six to 12 months. In the next year, it’s also working to define and integrate outcome measures for the top 50 medical conditions it treats, which will allow it to robustly assess the value of care delivery.
“Success will equally be to increase quality without an increase in cost,” he says.
What’s more, Dr. Pendleton says efforts are in the works to make the self-intuitive system available to institutions nationwide. For example, the center is working to address technical complexities resulting from the use of multiple electronic health record vendors.
“The goal is to make it usable in every environment so we can support other organizations in the most effective ways,” he says.
Although the tool is very easy to use, stakeholders are providing feedback that allows for continuous improvements in how data are presented.
“Anyone with basic computer skills and the ability to interpret bar charts and basic graphs can use it,” Dr. Pendleton says. The center also offers a 15-minute, confidential, training module and a one-hour, one-on-one training session with an analyst. TH
Karen Appold is a freelance author in Pennsylvania.
Reference
- Kawamoto K, Martin CJ, Williams K, et al. Value driven outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015;22(1):223-235.
Risk of Chronic Kidney Disease is Increased If You Have Sleep Apnea
(Reuters Health) - Having sleep apnea may increase the risk of chronic kidney disease, according to a report from Taiwan.
Researchers analyzed data from 2000 through 2010 on 8,600 adults diagnosed with sleep apnea and four times as many adults of similar age, sex and monthly income without sleep apnea, using Taiwan's National Health Insurance Research Database.
They found 157 new cases of chronic kidney disease among people with sleep apnea and 298 cases in the comparison group, according to Yung-Tai Chen of Taipei City Hospital Heping Fuyou Branch in Taiwan and coauthors.
After taking other health factors into account, sleep apnea increased the risk of kidney disease by 58%. By comparison, hypertension (a known risk factor for kidney disease) increased the risk by 17%. Diabetes was a stronger predictor than both other factors, more than doubling the risk of kidney disease, the research team reported online February 1 in Respirology.
Intermittent low oxygen levels during the night and fragmented sleep patterns may activate higher blood pressure, which would damage the kidneys and could make individuals more susceptible to chronic kidney disease, said Tetyana Kendzerska of the University of Toronto Institute for Clinical Evaluative Sciences, who was not part of the study in Taiwan.
But, "the findings from this study are limited by lack of information on sleep apnea and chronic kidney disease severity given that these conditions were defined through the health administrative data," Kendzerska said.
Factors like obesity and smoking status are also important for kidney risk but were not included in the assessment, she told Reuters Health by email.
"So, instead of concluding that sleep apnea has the same impact as high blood pressure on the kidney, I would rather conclude that this study suggests that the association between sleep apnea and chronic kidney disease may exist," and should be validated with more powerful studies, she said.
Moderate to severe obstructive sleep apnea can be treated with continuous positive airway pressure (CPAP) at night which may decrease high blood pressure and mitigate kidney risk, Kendzerska said.
"These findings raise the issue of whether the relationship between sleep apnea and chronic kidney disease is unidirectional or bidirectional," she said. "If the importance of sleep apnea and preventive effect of treatment will be confirmed in further studies, sleep apnea should be added to the list of modifiable risk factors considered in (chronic kidney disease) risk assessment."
The authors of the study did not respond to a request for comment.
(Reuters Health) - Having sleep apnea may increase the risk of chronic kidney disease, according to a report from Taiwan.
Researchers analyzed data from 2000 through 2010 on 8,600 adults diagnosed with sleep apnea and four times as many adults of similar age, sex and monthly income without sleep apnea, using Taiwan's National Health Insurance Research Database.
They found 157 new cases of chronic kidney disease among people with sleep apnea and 298 cases in the comparison group, according to Yung-Tai Chen of Taipei City Hospital Heping Fuyou Branch in Taiwan and coauthors.
After taking other health factors into account, sleep apnea increased the risk of kidney disease by 58%. By comparison, hypertension (a known risk factor for kidney disease) increased the risk by 17%. Diabetes was a stronger predictor than both other factors, more than doubling the risk of kidney disease, the research team reported online February 1 in Respirology.
Intermittent low oxygen levels during the night and fragmented sleep patterns may activate higher blood pressure, which would damage the kidneys and could make individuals more susceptible to chronic kidney disease, said Tetyana Kendzerska of the University of Toronto Institute for Clinical Evaluative Sciences, who was not part of the study in Taiwan.
But, "the findings from this study are limited by lack of information on sleep apnea and chronic kidney disease severity given that these conditions were defined through the health administrative data," Kendzerska said.
Factors like obesity and smoking status are also important for kidney risk but were not included in the assessment, she told Reuters Health by email.
"So, instead of concluding that sleep apnea has the same impact as high blood pressure on the kidney, I would rather conclude that this study suggests that the association between sleep apnea and chronic kidney disease may exist," and should be validated with more powerful studies, she said.
Moderate to severe obstructive sleep apnea can be treated with continuous positive airway pressure (CPAP) at night which may decrease high blood pressure and mitigate kidney risk, Kendzerska said.
"These findings raise the issue of whether the relationship between sleep apnea and chronic kidney disease is unidirectional or bidirectional," she said. "If the importance of sleep apnea and preventive effect of treatment will be confirmed in further studies, sleep apnea should be added to the list of modifiable risk factors considered in (chronic kidney disease) risk assessment."
The authors of the study did not respond to a request for comment.
(Reuters Health) - Having sleep apnea may increase the risk of chronic kidney disease, according to a report from Taiwan.
Researchers analyzed data from 2000 through 2010 on 8,600 adults diagnosed with sleep apnea and four times as many adults of similar age, sex and monthly income without sleep apnea, using Taiwan's National Health Insurance Research Database.
They found 157 new cases of chronic kidney disease among people with sleep apnea and 298 cases in the comparison group, according to Yung-Tai Chen of Taipei City Hospital Heping Fuyou Branch in Taiwan and coauthors.
After taking other health factors into account, sleep apnea increased the risk of kidney disease by 58%. By comparison, hypertension (a known risk factor for kidney disease) increased the risk by 17%. Diabetes was a stronger predictor than both other factors, more than doubling the risk of kidney disease, the research team reported online February 1 in Respirology.
Intermittent low oxygen levels during the night and fragmented sleep patterns may activate higher blood pressure, which would damage the kidneys and could make individuals more susceptible to chronic kidney disease, said Tetyana Kendzerska of the University of Toronto Institute for Clinical Evaluative Sciences, who was not part of the study in Taiwan.
But, "the findings from this study are limited by lack of information on sleep apnea and chronic kidney disease severity given that these conditions were defined through the health administrative data," Kendzerska said.
Factors like obesity and smoking status are also important for kidney risk but were not included in the assessment, she told Reuters Health by email.
"So, instead of concluding that sleep apnea has the same impact as high blood pressure on the kidney, I would rather conclude that this study suggests that the association between sleep apnea and chronic kidney disease may exist," and should be validated with more powerful studies, she said.
Moderate to severe obstructive sleep apnea can be treated with continuous positive airway pressure (CPAP) at night which may decrease high blood pressure and mitigate kidney risk, Kendzerska said.
"These findings raise the issue of whether the relationship between sleep apnea and chronic kidney disease is unidirectional or bidirectional," she said. "If the importance of sleep apnea and preventive effect of treatment will be confirmed in further studies, sleep apnea should be added to the list of modifiable risk factors considered in (chronic kidney disease) risk assessment."
The authors of the study did not respond to a request for comment.
FDA issues new guidance on Zika virus transmission

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus. ![]()

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus. ![]()

Photo courtesy of NHS
The US Food and Drug Administration (FDA) has issued a guidance document intended to help reduce the risk of Zika virus transmission via human cells, tissues, and cellular and tissue-based products (HCT/Ps).
The guidance addresses donation of HCT/Ps from both living and deceased donors, including donors of umbilical cord blood and hematopoietic stem/progenitor cells.
In this document, the FDA recommends a 6-month deferral period for most HCT/P donors who may have been exposed to the Zika virus.
The exception is donors of gestational tissues, who should be considered ineligible if they were at risk of contracting the virus at any time during their pregnancy.
The new guidance is a part of the FDA’s ongoing efforts to protect HCT/Ps and blood products from Zika virus transmission. Last month, the agency issued recommendations for reducing the risk of Zika virus transmission via blood transfusion.
There is a potential risk that Zika virus can be transmitted by HCT/Ps used as part of a medical, surgical, or reproductive procedure. HCT/Ps include products such as corneas, bone, skin, heart valves, hematopoietic stem/progenitor cells, gestational tissues such as amniotic membrane, and reproductive tissues such as semen and oocytes.
According to the Centers for Disease Control and Prevention, Zika virus can be spread by a man to his sexual partners. To date, there have been several cases of sexual transmission in the US.
Current information about Zika virus detection in semen suggests the period of deferral for HCT/P donors should be longer than the period recommended for donors of whole blood and blood components.
Therefore, the FDA has recommended that living donors of HCT/Ps be considered ineligible if they were diagnosed with Zika virus infection, were in an area with active Zika virus transmission, or had sex with a male with either of those risk factors within the past 6 months.
Donors of umbilical cord blood, placenta, or other gestational tissues should be considered ineligible if they have had any of the above risk factors at any point during their pregnancy.
Deceased donors should be considered ineligible if they were diagnosed with Zika virus infection in the past 6 months.
A deferral period of 6 months was chosen because of the limited data available on the length of time the virus can persist in all tissues. Zika virus has been detected in tissues and body fluids after the virus is no longer detectable in the blood stream and has been detected in semen possibly up to 10 weeks after the onset of symptoms.
Less evidence exists regarding the potential for transmission of Zika virus by HCT/Ps typically recovered from deceased donors. As more information becomes available, the understanding of the risks to recipients of HCT/Ps, including HCT/Ps recovered from deceased donors, may evolve.
The FDA said it will continue to monitor the situation and evaluate new information regarding the associated risks as it becomes available.
In addition, the FDA said it is prioritizing the development of blood donor screening and diagnostic tests that may be useful for identifying the presence of or recent infection with the Zika virus. The agency is prepared to evaluate investigational vaccines and therapeutics that might be developed and review technology that may help suppress populations of the mosquitoes that can spread the virus. ![]()
Factors driving blood cell development
Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/. ![]()

Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/. ![]()

Researchers believe they have identified key factors that drive hematopoietic specification and differentiation.
The team performed detailed analyses on cells at 6 consecutive stages of development—starting with embryonic stem cells and ending with macrophages.
They said this revealed the complete set of regulatory elements driving differential gene expression during these stages of development.
The researchers described this work in Developmental Cell.
The team studied hematopoietic specification and differentiation by looking at 6 stages of cell development—embryonic stem cells, mesoderm cells, hemangioblasts, hemogenic endothelium, hematopoietic precursors, and macrophages.
“We examined how embryonic cells develop towards blood cells by collecting multi-omics data from measuring gene activity, changes in chromosome structure, and the interaction of regulatory factors with the genes themselves,” explained study author Constanze Bonifer, PhD, of the University of Birmingham in the UK.
“Our research shows, in unprecedented detail, how a vast network of interacting genes control blood cell development. It also shows how we can use such data to enhance our knowledge of this process.”
The researchers said their findings help explain how regulatory elements in the DNA work together, driving gene expression and the switch from one developmental stage to another.
The team believes the work also revealed the minimum requirements for generating blood cells from an unrelated, cultured cell type.
The researchers have made their findings available to the public on the following website: http://www.haemopoiesis.leeds.ac.uk/data_analysis/. ![]()