User login
Complex Ankle and Hindfoot Arthrodesis Using Circular External Fixation
ABSTRACT
Surgical reconstruction of the ankle and hindfoot in patients with diabetes, Charcot neuroarthropathy, osteomyelitis, deformity, and/or bone loss can be challenging and often results in amputation. In these patients, conventional internal fixation with plates, screws, and intramedullary nails is often not feasible because of ongoing infection or poor bone stock and soft tissue quality. The Ilizarov method of ankle and hindfoot arthrodesis is a well-established technique for limb reconstruction that uses circular external fixation to achieve solid bony fusion, optimal leg length, and eradication of infection in cases of complex pathology. This article discusses indications, contraindications, pearls, and pitfalls of performing ankle and hindfoot arthrodesis using the Ilizarov technique.
Continue to: Patients with complex ankle and hindfoot deformity...
Patients with complex ankle and hindfoot deformity present a unique challenge to both nonoperative management and surgical reconstruction. Nonoperative management focuses on wound care, bracing, and immobilization using ankle-foot orthoses, total contact casts, and Charcot restraint orthotic walker boots for external stabilization. Fusion using the Ilizarov technique with circular fixation is a salvage limb-preservation procedure that has shown good results in select patient populations.1-5 Indications include post-traumatic, degenerative, and rheumatoid arthritis, osteomyelitis, tumors, neuromuscular conditions, and salvage of failed ankle and hindfoot procedures.6-9 Relative contraindications include wet gangrene, severe limb ischemia, and soft tissue compromise requiring urgent amputation. In addition, circular frames are not recommended in patients who are unable to comply with postoperative restrictions, and pin and wire care for the duration of frame placement because of personal, psychological, or socioeconomic reasons.
The Ilizarov technique of ring fixation provides dynamic, modular, and rigid fixation in multiple planes to control shear, bending, and rotational forces, and allows for early weight-bearing and postoperative adjustments as needed.10,11 Percutaneously placed half-pins and wires allow for solid fixation in the setting of both poor bone and soft tissue quality, and fusion can be achieved in the presence of active infection in a 1-stage procedure. The goal of ankle and hindfoot fusion using the Ilizarov technique is to achieve an infection-free, stable, plantigrade foot with neutral ankle alignment to allow for patient ambulation and return to activities of daily living.
Nonunion rates with circular fixation are reported to be as high as 16% to 54%, due to medical comorbidities, such as smoking, peripheral vascular disease, and Charcot neuroarthropathy.1Charcot, in particular, is a risk factor for nonunion as patients lack protective sensation, and have a higher rate of wound dehiscence, noncompliance with weight-bearing precautions, pin site infections, and frame breakage. In these patients, tibiotalocalcaneal (TTC) arthrodesis is preferred over the isolated ankle, or subtalar fusion to both provide a stable platform for ambulation and reduce the incidence of adjacent joint breakdown. Common complications of the Ilizarov technique include pin site infections, wire breakage, talar necrosis, and tibial stress fractures after frame removal.1,2,6,11-13 Circular frames are typically maintained for 3 to 8 months, until solid fusion is achieved radiographically. Frames are removed in the operating room with the concurrent examination of the fusion sites under anesthesia followed by a period of protected weight-bearing in a cast or tall controlled ankle motion (CAM) boot.
This article reviews several technical details, tips, and tricks that can help improve the intraoperative and postoperative outcomes of combined ankle and hindfoot arthrodesis using the Ilizarov technique with circular external fixation.
Continue to: SURGICAL TECHNIQUE...
SURGICAL TECHNIQUE
SETUP AND APPROACH
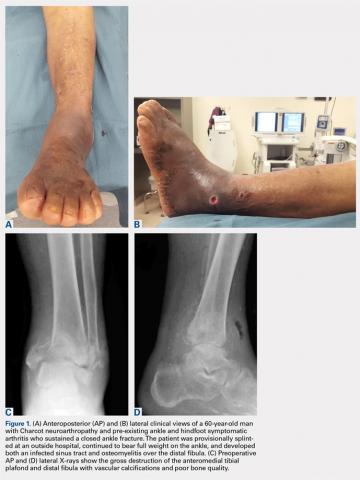
Patients are positioned supine with padding under the operative extremity to achieve neutral leg rotation (Figures 1A-1D). A thigh tourniquet is placed with the foot positioned at the end of the bed and on top of the radiolucent padding to avoid interference of the contralateral leg during lateral X-rays. After sterile prepping and draping, the extremity is exsanguinated above the level of an active infection, and the tourniquet inflated.
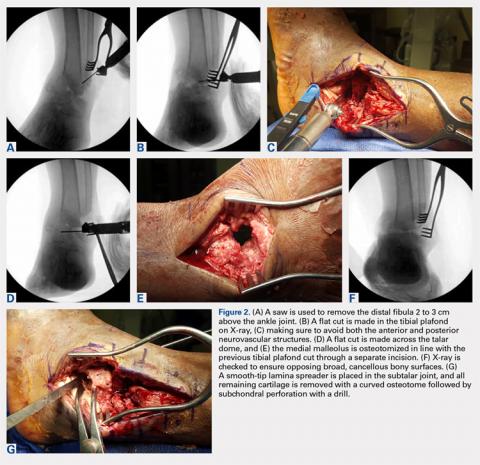
For isolated ankle arthrodesis, an anterior or lateral approach can be used, while for TTC arthrodesis, a lateral approach is required to access both the ankle and subtalar joints. A 10-cm longitudinal incision is made along the distal fibula, curving slightly and anteriorly along the distal extent of the incision. Dissection is continued down to bone using full thickness flaps, and the distal fibula is removed 2 to 3 cm above the ankle joint using a saw and osteotome (Figures 2A-2G). The distal fibula can be used subsequently as bone grafts depending on the quality of bone. The peroneal tendons are retracted posteriorly, and dissection is then continued to the posterior facet of the subtalar joint.
JOINT PREPARATION AND ALIGNMENT
Both the anterior and posterior neurovascular bundles are protected along the distal tibia with Hohmann retractors while a saw is used to create flat cuts across the tibial plafond and talus to allow apposition of flat, broad cancellous bony surfaces. Flat cuts followed by later joint compression will often shorten the limb by 2 to 3 cm. This leg length discrepancy can later be accommodated using a shoe lift, as needed. All retained hardware and/or infected and necrotic tissues in the ankle and hindfoot are removed using a rongeur and a pituitary rongeur.
The medial malleolus is osteotomized vertically using a direct medial incision and approach with full thickness flaps, and in line with the previous tibial plafond, is both cut and removed. The medial malleolus can also be used for bone grafts in fusion sites. A smooth-tip lamina spreader is placed in the subtalar joint for distraction and a curved osteotome, curettes, and a small rongeur are used to remove all remaining cartilage from the subtalar joint. Flat cuts in the subtalar joint can remove excessive bone, particularly from the inferior aspect of the talus. The subchondral bone is perforated using a 2.5- to 3.0-mm drill bit and a curved osteotome.
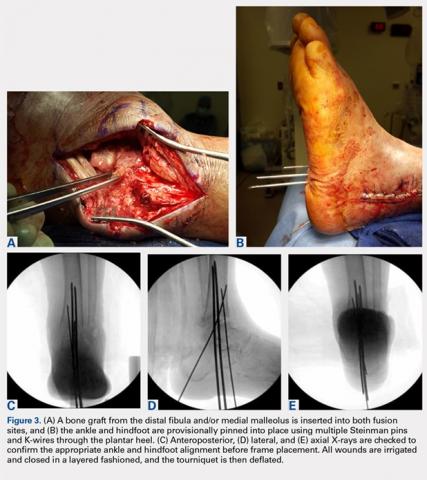
A bone graft from the distal fibula and medial malleolus, with or without the addition of allograft adjuvants, is placed evenly across the ankle and subtalar joints (Figures 3A-3E). At this point, the ankle and subtalar joints can be manipulated in multiple planes to achieve neutral coronal, sagittal, and axial alignment. With both the ankle and hindfoot held in a neutral position, multiple Steinman pins and K-wires in different orientations are inserted through the plantar aspect of the heel to hold the ankle and subtalar joints in place temporarily. Wires are cut short to prevent interference with subsequent foot olive wire placement through the frame.
Continue to: X-rays should be carefully checked...
X-rays should be carefully checked to ensure proper alignment. Wounds are gently irrigated, and vancomycin powder (2 g) can be placed within wounds for local antibiotic delivery. Lateral tissues are sharply debulked to allow for decreased tension on the incision, and small ulcers can be excised in their entirety. Wounds are closed in a layered fashion using 0-polydioxanone (PDS, Ethicon) suture for deep tissue, 2-0 PDS for subcutaneous tissue, and 2-0 nylon for skin closure. The tourniquet is deflated for the remainder of the case to reduce limb ischemia during frame placement.
CIRCULAR FRAME CONCEPTS AND PLACEMENT
The majority of circular frames for both ankle and hindfoot fusion have multiple ring sizes available in aluminum and radiolucent carbon fiber reinforced polymer (Hoffmann LRF, Stryker). Rings are available in full, open, segment, and both short- and long-foot options. Frames can be sterilized in a prebuilt 3 to 4 ring construct with 4 static or dynamic (telescopic) struts (100-277 mm). The most commonly used tibia and foot ring sizes are 155 cm, 180 cm, and 210 cm. Ring size should be able to accommodate posterior soft tissue swelling and avoid circumferential soft tissue abrasion against the rings. Anterior foot arches are used for increased construct stability and can be locked to the distal tibia ring for weight-bearing support. Wire and half-pin bolts, adaptors, and nuts are used to join each ring of the frame to the patient’s bone.
For TTC arthrodesis, 2 rings are typically used in the tibia, and 1 ring is used in the foot. For isolated ankle arthrodesis, an additional ring can be added with olive wires in the talus to permit compression only across the ankle joint. Multiple points of fixation are used in each ring in different planes to achieve both maximal stability and rotational control. If a single wire or half-pin becomes infected and requires removal, there are still multiple other points of fixation in the ring to maintain stability. Fixation within each ring should be off axis compared with the adjacent ring to both avoid stress risers and increase construct rigidity.
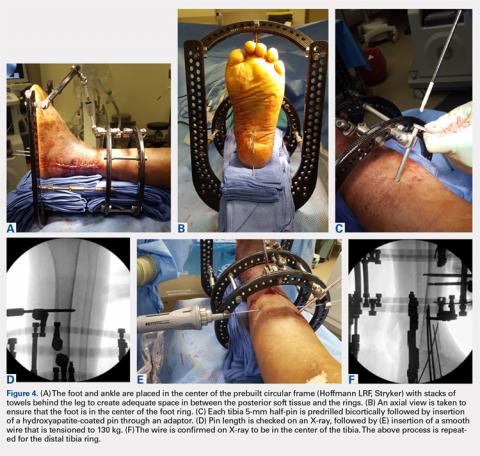
The prebuilt frame is checked on the back table to ensure proper orientation and component alignment. The frame is then placed over both the foot and ankle, and multiple stacks of towels are placed behind the heel, ankle, and calf to center the foot and ankle in the frame (Figures 4A-4F). At least 4 to 6 cm of space is needed in between the posterior soft tissues and each ring to accommodate postoperative swelling. On the lateral view, the foot ring should be in the mid-portion of the calcaneus. If there is a concern, particularly in Charcot patients, regarding early weight-bearing noncompliance, the foot ring can be placed flush with the plantar aspect of the foot, and olive wires can be inserted using longer adaptors. The frame should be checked from multiple viewpoints to ensure that both the foot and ankle are centered and in neutral rotation.
Continue to: TIBIA RING FIXATION...
TIBIA RING FIXATION
Tibia rings can be fixed using 2 to 3 half-pins (4-6 mm) alone or 2 half-pins in combination with a smooth wire. A small incision is made over the area of planned half-pin insertion, and the periosteum is cleared away using a hemostat. An adaptor sleeve is used, and the bone is drilled bicortically, followed by insertion of the half-pin. Hydroxyapatite-coated pins are used to improve the strength of the bone-pin interface and reduce the incidence of pin tract infections. Pins are inserted along both the anterior and medial aspects of the tibia, avoiding the thick lateral musculature. Care is taken to protect the medial neurovascular structures during pin placement following established Ilizarov safe zones.
After each pin is placed in the bone, the pin is secured to the adaptor that is then tightened to the ring. This process is repeated for both the proximal and distal tibia rings. Pins should be placed above and below each ring to avoid creating stress risers. During smooth wire placement, each wire is pushed by hand through the soft tissues and then drilled into the bone while the exposed segment is held with a damp sponge to reduce the incidence of thermal bone necrosis. Once the wire is drilled bicortically, a mallet is used to tap the wire through the remaining soft tissues to avoid wrapping them up in the wire. Each wire should be parallel to the ring to get an even line of compression.
Each wire is secured on 1 end and then tensioned to 130 kg using a hand tensioner. An additional tool can be placed in the wire adaptor to prevent the wire from bending during tensioning. If the wire is passing above or below the ring, longer wire adaptors should be used to build to the wire. The wire should never be bent toward the ring as this can increase the likelihood of improper pin tensioning and breakage. Wire placement should be avoided posteriorly as this can make it difficult to secure and/or tension wires, and also increases the risk of damage to posterior structures.
Ring fixation in the distal tibia near the plafond may require 1 half-pin and 2 wires to avoid damage to the tibialis anterior and posterior tibial tendons. In this case, smooth wires should be placed in a crossing pattern and tensioned simultaneously to avoid pulling the ankle away from the center of the frame. Wires should be bent and curved over each ring and then cut to facilitate subsequent removal.
FOOT RING FIXATION
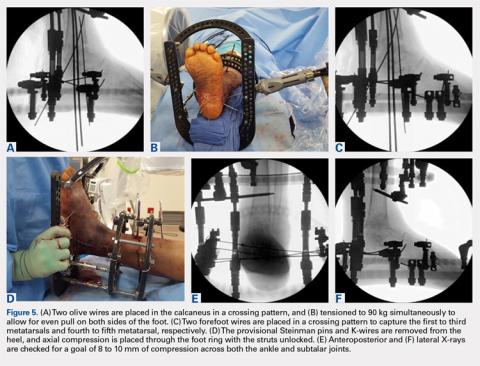
In the foot, olive wires are used to increase fixation against bone. For each olive wire, a small incision is made to accommodate the diameter of the olive through the soft tissue. Similar to the distal tibia, 2 olive wires should be placed above and below the foot ring in a crossing pattern through the calcaneus (Figures 5A-5F). The axial view of the frame should be checked to ensure proper wire orientation. When using olive wires, it is essential to tension both at the same time to 90 kg, as the foot can be pulled medially or laterally in the frame if 1 wire is tensioned before the other.
Forefoot olive wires should also be placed in a crossing pattern, with 1 wire fixed through the first, second, and third metatarsals, and 1 wire through the fourth and fifth metatarsals. Additional forefoot olive wires can be placed if compression is needed across the midfoot or Chopart joints for fusion. Multiple X-rays should be checked to ensure that the calcaneus and forefoot olive wires are firmly fixed both in and against bone.
Continue to: JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS...
JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS
Once all rings are secured to the bone with half-pins and wires, the previously placed Steinman pins, and K-wires through the heel are removed. Both ankle and subtalar joint alignments are rechecked, and then axial compression is placed through the foot ring with the knee extended and the struts unlocked. Static or telescopic struts are used to achieve 8 to 10 mm of bony compression. X-rays are taken before and after to analyze final joint compression and alignment. Struts should be sequentially tightened (1/2 turn of a static strut) 1 at a time as final tightening of 1 strut alone can bind and interfere with both the compression and tightening of the remaining struts.
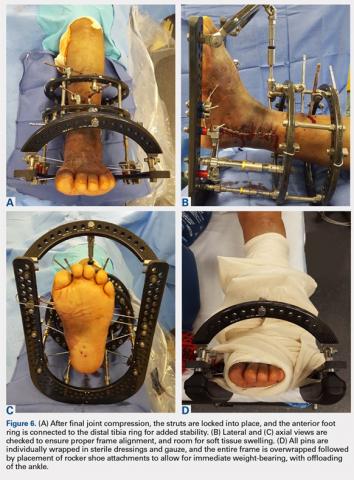
Once final compression is achieved, the struts are locked, and the front foot arch is closed anteriorly and connected to the distal tibia ring for increased stability (Figures 6A-6D). Each pin and wire is covered in a sterile dressing followed by gauze to allow for soft tissue padding. The entire frame is then overwrapped in bias stockinette rolls or ace wraps.
Walking attachments can be added immediately to the frame that allows for early weight-bearing. Rocker shoe attachments with a 15° anterior and posterior slope and rubber soles can help offload the ankle and subtalar joints, decrease pressure on heel strike, and reduce ankle motion during ambulation (Hoffmann LRF, Stryker).
POSTOPERATIVE PROTOCOL
Depending on individual characteristics, patients can be immediately weight-bearing in the circular frame. Patients with Charcot neuroarthropathy are recommended to remain non-weight-bearing for the first 2 months to reduce the likelihood of pin, wire, and frame breakage along with nonunion. Pin and wire site care and maintenance are initiated the day after surgery and continue on a daily basis for the duration of frame placement. Sutures are removed 4 to 5 weeks after surgery to ensure adequate wound healing. Serial X-rays are taken monthly to analyze fusion sites.
If pins or wires become infected, patients are placed on oral antibiotics, and both pins and wires can be removed or exchanged in the operating room. Once fusion is achieved in 3 to 8 months (Figures 7A-7C), the frame is removed in the operating room, and fusion sites are examined under dynamic fluoroscopy. If fusion is confirmed, patients are made weight-bearing as tolerated in a short-leg cast or tall CAM boot for 6 to 8 weeks, and then transitioned to an ankle brace in an accommodative shoe.
Continue to: DISCUSSION...
DISCUSSION
A key aspect of recovery after ankle and hindfoot fusion using the Ilizarov technique is balancing pin care, soft tissue swelling, and weight-bearing status. The average time patients will spend in the frame is approximately 25 to 28 weeks, but can range from 12 to 84 weeks.1,2Given the considerable variability in both soft tissue healing and bony union, patients should be extensively counseled before surgery to set expectations correctly and ensure that they have the necessary help and support to care for the frame during the treatment period. Patients should be followed closely during the first 6 weeks to ensure that pins and wires do not become infected or break, as both of these issues require immediate intervention.
In a review of 11 patients who underwent tibiocalcaneal arthrodesis using an Ilizarov external fixator for infected talar nonunions or extrusions, Rochman and colleagues8 reported an 81% rate of successful fusion with a final mean American Orthopaedic Foot and Ankle Society score of 65 (out of a maximum 86). Similar results were reported by Saltzman9 in a series of 8 patients with diffuse ankle osteomyelitis treated with resection of all infected tissue and hybrid-frame compression arthrodesis. All patients received 6 weeks of intravenous antibiotics, and frames were removed at 3 months, and walking casts were applied for 1 to 2 additional months. Ankle sepsis was eradicated in all patients, and 7/8 (87.5%) ankles successfully fused at an average of 13.5 weeks (range, 10-16 weeks). One limb required below-knee amputation at 5 weeks due to non-reconstructible vascular insufficiency. At an average of 3.4-year follow-up, none of the 7 fused ankles required further surgery.
Fragomen and colleagues1 retrospectively reviewed 101 patients who underwent complex ankle fusion using the Ilizarov technique and found that 76/91 (83.5%) patients achieved fusion at an average of 25 weeks (range, 10-65 weeks). Smoking was associated with a 54% rate of nonunion and 15/19 (79%) patients with Charcot neuroarthropathy achieved ankle fusion, but had a subsequent subtalar joint failure, thus highlighting the need for TTC arthrodesis in Charcot patients. Salem and colleagues2 reviewed 21 Ilizarov ankle fusions and reported that all patients achieved fusion at an average of 28 weeks (range, 12-84 weeks). Complications occurred in 11 patients, including 2 nonunions that healed after revision frame application and 4 pin tract infections.
CONCLUSION
Overall, the Ilizarov technique using circular external fixation is a powerful tool that can be used to treat a variety of disorders including complex foot and ankle deformity and infection. While case series generally show favorable outcomes, patients must be informed that this technique is a salvage procedure for limb preservation that requires meticulous operative technique, diligent postoperative care, and tight control of medical comorbidities, such as blood sugar levels in individuals with diabetes to achieve a successful outcome.
1. Fragomen AT, Borst E, Schachter L, Lyman S, Rozbruch SR. Complex ankle arthrodesis using the Ilizarov method yields high rate of fusion. Clin Orthop Relat Res. 2012;470(10):2864-2873. doi:10.1007/s11999-012-2470-9.
2. Salem KH, Kinzl L, Schmelz A. Ankle arthrodesis using Ilizarov ring fixators: a review of 22 cases. Foot Ankle Int. 2006;27(10):764-770. doi:10.1177/107110070602701002.
3. Cierny G 3rd, Cook WG, Mader JT. Ankle arthrodesis in the presence of ongoing sepsis. Indications, methods, and results. Orthop Clin North Am. 1989;20(4):709-721.
4. Dalla Paola L, Brocco E, Ceccacci T, et al. Limb salvage in Charcot foot and ankle osteomyelitis: combined use single stage/double stage of arthrodesis and external fixation. Foot Ankle Int. 2009;30(11):1065-1070. doi:10.3113/FAI.2009.1065.
5. Eylon S, Porat S, Bor N, Leibner ED. Outcome of Ilizarov ankle arthrodesis. Foot Ankle Int. 2007;28(8):873-879. doi:10.3113/FAI.2007.0873.
6. Kalish S, Fleming J, Weinstein R. External fixators for elective rearfoot and ankle arthrodesis. Techniques and indications. Clin Podiatr Med Surg. 2003;20(1):65-96, vi.
7. Kollig E, Esenwein SA, Muhr G, Kutscha-Lissberg F. Fusion of the septic ankle: experience with 15 cases using hybrid external fixation. J Trauma. 2003;55(4):685-691. doi:10.1097/01.TA.0000051933.83342.E4.
8. Rochman R, Jackson Hutson J, Alade O. Tibiocalcaneal arthrodesis using the Ilizarov technique in the presence of bone loss and infection of the talus. Foot Ankle Int. 2008;29(10):1001-1008. doi:10.3113/FAI.2008.1001.
9. Saltzman CL. Salvage of diffuse ankle osteomyelitis by single-stage resection and circumferential frame compression arthrodesis. Iowa Orthop J. 2005;2547-52.
10. Fragomen AT, Rozbruch SR. The mechanics of external fixation. HSS J. 2007;3(1):13-29. doi:10.1007/s11420-006-9025-0.
11. Hawkins BJ, Langerman RJ, Anger DM, Calhoun JH. The Ilizarov technique in ankle fusion. Clin Orthop Relat Res. 1994;(303):217-225.
12. Jones CP, Youngblood CS, Waldrop N, Davis WH, Pinzur MS. Tibial Stress Fracture Secondary to Half-Pins in Circular Ring External Fixation for Charcot Foot. Foot Ankle Int. 2014;35(6):572-577. doi:10.1177/1071100714531229.
13. Kazmers NH, Fragomen AT, Rozbruch SR. Prevention of pin site infection in external fixation: a review of the literature. Strategies Trauma Limb Reconstr. 2016;11(2):75-85. doi:10.1007/s11751-016-0256-4.
ABSTRACT
Surgical reconstruction of the ankle and hindfoot in patients with diabetes, Charcot neuroarthropathy, osteomyelitis, deformity, and/or bone loss can be challenging and often results in amputation. In these patients, conventional internal fixation with plates, screws, and intramedullary nails is often not feasible because of ongoing infection or poor bone stock and soft tissue quality. The Ilizarov method of ankle and hindfoot arthrodesis is a well-established technique for limb reconstruction that uses circular external fixation to achieve solid bony fusion, optimal leg length, and eradication of infection in cases of complex pathology. This article discusses indications, contraindications, pearls, and pitfalls of performing ankle and hindfoot arthrodesis using the Ilizarov technique.
Continue to: Patients with complex ankle and hindfoot deformity...
Patients with complex ankle and hindfoot deformity present a unique challenge to both nonoperative management and surgical reconstruction. Nonoperative management focuses on wound care, bracing, and immobilization using ankle-foot orthoses, total contact casts, and Charcot restraint orthotic walker boots for external stabilization. Fusion using the Ilizarov technique with circular fixation is a salvage limb-preservation procedure that has shown good results in select patient populations.1-5 Indications include post-traumatic, degenerative, and rheumatoid arthritis, osteomyelitis, tumors, neuromuscular conditions, and salvage of failed ankle and hindfoot procedures.6-9 Relative contraindications include wet gangrene, severe limb ischemia, and soft tissue compromise requiring urgent amputation. In addition, circular frames are not recommended in patients who are unable to comply with postoperative restrictions, and pin and wire care for the duration of frame placement because of personal, psychological, or socioeconomic reasons.
The Ilizarov technique of ring fixation provides dynamic, modular, and rigid fixation in multiple planes to control shear, bending, and rotational forces, and allows for early weight-bearing and postoperative adjustments as needed.10,11 Percutaneously placed half-pins and wires allow for solid fixation in the setting of both poor bone and soft tissue quality, and fusion can be achieved in the presence of active infection in a 1-stage procedure. The goal of ankle and hindfoot fusion using the Ilizarov technique is to achieve an infection-free, stable, plantigrade foot with neutral ankle alignment to allow for patient ambulation and return to activities of daily living.
Nonunion rates with circular fixation are reported to be as high as 16% to 54%, due to medical comorbidities, such as smoking, peripheral vascular disease, and Charcot neuroarthropathy.1Charcot, in particular, is a risk factor for nonunion as patients lack protective sensation, and have a higher rate of wound dehiscence, noncompliance with weight-bearing precautions, pin site infections, and frame breakage. In these patients, tibiotalocalcaneal (TTC) arthrodesis is preferred over the isolated ankle, or subtalar fusion to both provide a stable platform for ambulation and reduce the incidence of adjacent joint breakdown. Common complications of the Ilizarov technique include pin site infections, wire breakage, talar necrosis, and tibial stress fractures after frame removal.1,2,6,11-13 Circular frames are typically maintained for 3 to 8 months, until solid fusion is achieved radiographically. Frames are removed in the operating room with the concurrent examination of the fusion sites under anesthesia followed by a period of protected weight-bearing in a cast or tall controlled ankle motion (CAM) boot.
This article reviews several technical details, tips, and tricks that can help improve the intraoperative and postoperative outcomes of combined ankle and hindfoot arthrodesis using the Ilizarov technique with circular external fixation.
Continue to: SURGICAL TECHNIQUE...
SURGICAL TECHNIQUE
SETUP AND APPROACH
Patients are positioned supine with padding under the operative extremity to achieve neutral leg rotation (Figures 1A-1D). A thigh tourniquet is placed with the foot positioned at the end of the bed and on top of the radiolucent padding to avoid interference of the contralateral leg during lateral X-rays. After sterile prepping and draping, the extremity is exsanguinated above the level of an active infection, and the tourniquet inflated.
For isolated ankle arthrodesis, an anterior or lateral approach can be used, while for TTC arthrodesis, a lateral approach is required to access both the ankle and subtalar joints. A 10-cm longitudinal incision is made along the distal fibula, curving slightly and anteriorly along the distal extent of the incision. Dissection is continued down to bone using full thickness flaps, and the distal fibula is removed 2 to 3 cm above the ankle joint using a saw and osteotome (Figures 2A-2G). The distal fibula can be used subsequently as bone grafts depending on the quality of bone. The peroneal tendons are retracted posteriorly, and dissection is then continued to the posterior facet of the subtalar joint.
JOINT PREPARATION AND ALIGNMENT
Both the anterior and posterior neurovascular bundles are protected along the distal tibia with Hohmann retractors while a saw is used to create flat cuts across the tibial plafond and talus to allow apposition of flat, broad cancellous bony surfaces. Flat cuts followed by later joint compression will often shorten the limb by 2 to 3 cm. This leg length discrepancy can later be accommodated using a shoe lift, as needed. All retained hardware and/or infected and necrotic tissues in the ankle and hindfoot are removed using a rongeur and a pituitary rongeur.
The medial malleolus is osteotomized vertically using a direct medial incision and approach with full thickness flaps, and in line with the previous tibial plafond, is both cut and removed. The medial malleolus can also be used for bone grafts in fusion sites. A smooth-tip lamina spreader is placed in the subtalar joint for distraction and a curved osteotome, curettes, and a small rongeur are used to remove all remaining cartilage from the subtalar joint. Flat cuts in the subtalar joint can remove excessive bone, particularly from the inferior aspect of the talus. The subchondral bone is perforated using a 2.5- to 3.0-mm drill bit and a curved osteotome.
A bone graft from the distal fibula and medial malleolus, with or without the addition of allograft adjuvants, is placed evenly across the ankle and subtalar joints (Figures 3A-3E). At this point, the ankle and subtalar joints can be manipulated in multiple planes to achieve neutral coronal, sagittal, and axial alignment. With both the ankle and hindfoot held in a neutral position, multiple Steinman pins and K-wires in different orientations are inserted through the plantar aspect of the heel to hold the ankle and subtalar joints in place temporarily. Wires are cut short to prevent interference with subsequent foot olive wire placement through the frame.
Continue to: X-rays should be carefully checked...
X-rays should be carefully checked to ensure proper alignment. Wounds are gently irrigated, and vancomycin powder (2 g) can be placed within wounds for local antibiotic delivery. Lateral tissues are sharply debulked to allow for decreased tension on the incision, and small ulcers can be excised in their entirety. Wounds are closed in a layered fashion using 0-polydioxanone (PDS, Ethicon) suture for deep tissue, 2-0 PDS for subcutaneous tissue, and 2-0 nylon for skin closure. The tourniquet is deflated for the remainder of the case to reduce limb ischemia during frame placement.
CIRCULAR FRAME CONCEPTS AND PLACEMENT
The majority of circular frames for both ankle and hindfoot fusion have multiple ring sizes available in aluminum and radiolucent carbon fiber reinforced polymer (Hoffmann LRF, Stryker). Rings are available in full, open, segment, and both short- and long-foot options. Frames can be sterilized in a prebuilt 3 to 4 ring construct with 4 static or dynamic (telescopic) struts (100-277 mm). The most commonly used tibia and foot ring sizes are 155 cm, 180 cm, and 210 cm. Ring size should be able to accommodate posterior soft tissue swelling and avoid circumferential soft tissue abrasion against the rings. Anterior foot arches are used for increased construct stability and can be locked to the distal tibia ring for weight-bearing support. Wire and half-pin bolts, adaptors, and nuts are used to join each ring of the frame to the patient’s bone.
For TTC arthrodesis, 2 rings are typically used in the tibia, and 1 ring is used in the foot. For isolated ankle arthrodesis, an additional ring can be added with olive wires in the talus to permit compression only across the ankle joint. Multiple points of fixation are used in each ring in different planes to achieve both maximal stability and rotational control. If a single wire or half-pin becomes infected and requires removal, there are still multiple other points of fixation in the ring to maintain stability. Fixation within each ring should be off axis compared with the adjacent ring to both avoid stress risers and increase construct rigidity.
The prebuilt frame is checked on the back table to ensure proper orientation and component alignment. The frame is then placed over both the foot and ankle, and multiple stacks of towels are placed behind the heel, ankle, and calf to center the foot and ankle in the frame (Figures 4A-4F). At least 4 to 6 cm of space is needed in between the posterior soft tissues and each ring to accommodate postoperative swelling. On the lateral view, the foot ring should be in the mid-portion of the calcaneus. If there is a concern, particularly in Charcot patients, regarding early weight-bearing noncompliance, the foot ring can be placed flush with the plantar aspect of the foot, and olive wires can be inserted using longer adaptors. The frame should be checked from multiple viewpoints to ensure that both the foot and ankle are centered and in neutral rotation.
Continue to: TIBIA RING FIXATION...
TIBIA RING FIXATION
Tibia rings can be fixed using 2 to 3 half-pins (4-6 mm) alone or 2 half-pins in combination with a smooth wire. A small incision is made over the area of planned half-pin insertion, and the periosteum is cleared away using a hemostat. An adaptor sleeve is used, and the bone is drilled bicortically, followed by insertion of the half-pin. Hydroxyapatite-coated pins are used to improve the strength of the bone-pin interface and reduce the incidence of pin tract infections. Pins are inserted along both the anterior and medial aspects of the tibia, avoiding the thick lateral musculature. Care is taken to protect the medial neurovascular structures during pin placement following established Ilizarov safe zones.
After each pin is placed in the bone, the pin is secured to the adaptor that is then tightened to the ring. This process is repeated for both the proximal and distal tibia rings. Pins should be placed above and below each ring to avoid creating stress risers. During smooth wire placement, each wire is pushed by hand through the soft tissues and then drilled into the bone while the exposed segment is held with a damp sponge to reduce the incidence of thermal bone necrosis. Once the wire is drilled bicortically, a mallet is used to tap the wire through the remaining soft tissues to avoid wrapping them up in the wire. Each wire should be parallel to the ring to get an even line of compression.
Each wire is secured on 1 end and then tensioned to 130 kg using a hand tensioner. An additional tool can be placed in the wire adaptor to prevent the wire from bending during tensioning. If the wire is passing above or below the ring, longer wire adaptors should be used to build to the wire. The wire should never be bent toward the ring as this can increase the likelihood of improper pin tensioning and breakage. Wire placement should be avoided posteriorly as this can make it difficult to secure and/or tension wires, and also increases the risk of damage to posterior structures.
Ring fixation in the distal tibia near the plafond may require 1 half-pin and 2 wires to avoid damage to the tibialis anterior and posterior tibial tendons. In this case, smooth wires should be placed in a crossing pattern and tensioned simultaneously to avoid pulling the ankle away from the center of the frame. Wires should be bent and curved over each ring and then cut to facilitate subsequent removal.
FOOT RING FIXATION
In the foot, olive wires are used to increase fixation against bone. For each olive wire, a small incision is made to accommodate the diameter of the olive through the soft tissue. Similar to the distal tibia, 2 olive wires should be placed above and below the foot ring in a crossing pattern through the calcaneus (Figures 5A-5F). The axial view of the frame should be checked to ensure proper wire orientation. When using olive wires, it is essential to tension both at the same time to 90 kg, as the foot can be pulled medially or laterally in the frame if 1 wire is tensioned before the other.
Forefoot olive wires should also be placed in a crossing pattern, with 1 wire fixed through the first, second, and third metatarsals, and 1 wire through the fourth and fifth metatarsals. Additional forefoot olive wires can be placed if compression is needed across the midfoot or Chopart joints for fusion. Multiple X-rays should be checked to ensure that the calcaneus and forefoot olive wires are firmly fixed both in and against bone.
Continue to: JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS...
JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS
Once all rings are secured to the bone with half-pins and wires, the previously placed Steinman pins, and K-wires through the heel are removed. Both ankle and subtalar joint alignments are rechecked, and then axial compression is placed through the foot ring with the knee extended and the struts unlocked. Static or telescopic struts are used to achieve 8 to 10 mm of bony compression. X-rays are taken before and after to analyze final joint compression and alignment. Struts should be sequentially tightened (1/2 turn of a static strut) 1 at a time as final tightening of 1 strut alone can bind and interfere with both the compression and tightening of the remaining struts.
Once final compression is achieved, the struts are locked, and the front foot arch is closed anteriorly and connected to the distal tibia ring for increased stability (Figures 6A-6D). Each pin and wire is covered in a sterile dressing followed by gauze to allow for soft tissue padding. The entire frame is then overwrapped in bias stockinette rolls or ace wraps.
Walking attachments can be added immediately to the frame that allows for early weight-bearing. Rocker shoe attachments with a 15° anterior and posterior slope and rubber soles can help offload the ankle and subtalar joints, decrease pressure on heel strike, and reduce ankle motion during ambulation (Hoffmann LRF, Stryker).
POSTOPERATIVE PROTOCOL
Depending on individual characteristics, patients can be immediately weight-bearing in the circular frame. Patients with Charcot neuroarthropathy are recommended to remain non-weight-bearing for the first 2 months to reduce the likelihood of pin, wire, and frame breakage along with nonunion. Pin and wire site care and maintenance are initiated the day after surgery and continue on a daily basis for the duration of frame placement. Sutures are removed 4 to 5 weeks after surgery to ensure adequate wound healing. Serial X-rays are taken monthly to analyze fusion sites.
If pins or wires become infected, patients are placed on oral antibiotics, and both pins and wires can be removed or exchanged in the operating room. Once fusion is achieved in 3 to 8 months (Figures 7A-7C), the frame is removed in the operating room, and fusion sites are examined under dynamic fluoroscopy. If fusion is confirmed, patients are made weight-bearing as tolerated in a short-leg cast or tall CAM boot for 6 to 8 weeks, and then transitioned to an ankle brace in an accommodative shoe.
Continue to: DISCUSSION...
DISCUSSION
A key aspect of recovery after ankle and hindfoot fusion using the Ilizarov technique is balancing pin care, soft tissue swelling, and weight-bearing status. The average time patients will spend in the frame is approximately 25 to 28 weeks, but can range from 12 to 84 weeks.1,2Given the considerable variability in both soft tissue healing and bony union, patients should be extensively counseled before surgery to set expectations correctly and ensure that they have the necessary help and support to care for the frame during the treatment period. Patients should be followed closely during the first 6 weeks to ensure that pins and wires do not become infected or break, as both of these issues require immediate intervention.
In a review of 11 patients who underwent tibiocalcaneal arthrodesis using an Ilizarov external fixator for infected talar nonunions or extrusions, Rochman and colleagues8 reported an 81% rate of successful fusion with a final mean American Orthopaedic Foot and Ankle Society score of 65 (out of a maximum 86). Similar results were reported by Saltzman9 in a series of 8 patients with diffuse ankle osteomyelitis treated with resection of all infected tissue and hybrid-frame compression arthrodesis. All patients received 6 weeks of intravenous antibiotics, and frames were removed at 3 months, and walking casts were applied for 1 to 2 additional months. Ankle sepsis was eradicated in all patients, and 7/8 (87.5%) ankles successfully fused at an average of 13.5 weeks (range, 10-16 weeks). One limb required below-knee amputation at 5 weeks due to non-reconstructible vascular insufficiency. At an average of 3.4-year follow-up, none of the 7 fused ankles required further surgery.
Fragomen and colleagues1 retrospectively reviewed 101 patients who underwent complex ankle fusion using the Ilizarov technique and found that 76/91 (83.5%) patients achieved fusion at an average of 25 weeks (range, 10-65 weeks). Smoking was associated with a 54% rate of nonunion and 15/19 (79%) patients with Charcot neuroarthropathy achieved ankle fusion, but had a subsequent subtalar joint failure, thus highlighting the need for TTC arthrodesis in Charcot patients. Salem and colleagues2 reviewed 21 Ilizarov ankle fusions and reported that all patients achieved fusion at an average of 28 weeks (range, 12-84 weeks). Complications occurred in 11 patients, including 2 nonunions that healed after revision frame application and 4 pin tract infections.
CONCLUSION
Overall, the Ilizarov technique using circular external fixation is a powerful tool that can be used to treat a variety of disorders including complex foot and ankle deformity and infection. While case series generally show favorable outcomes, patients must be informed that this technique is a salvage procedure for limb preservation that requires meticulous operative technique, diligent postoperative care, and tight control of medical comorbidities, such as blood sugar levels in individuals with diabetes to achieve a successful outcome.
ABSTRACT
Surgical reconstruction of the ankle and hindfoot in patients with diabetes, Charcot neuroarthropathy, osteomyelitis, deformity, and/or bone loss can be challenging and often results in amputation. In these patients, conventional internal fixation with plates, screws, and intramedullary nails is often not feasible because of ongoing infection or poor bone stock and soft tissue quality. The Ilizarov method of ankle and hindfoot arthrodesis is a well-established technique for limb reconstruction that uses circular external fixation to achieve solid bony fusion, optimal leg length, and eradication of infection in cases of complex pathology. This article discusses indications, contraindications, pearls, and pitfalls of performing ankle and hindfoot arthrodesis using the Ilizarov technique.
Continue to: Patients with complex ankle and hindfoot deformity...
Patients with complex ankle and hindfoot deformity present a unique challenge to both nonoperative management and surgical reconstruction. Nonoperative management focuses on wound care, bracing, and immobilization using ankle-foot orthoses, total contact casts, and Charcot restraint orthotic walker boots for external stabilization. Fusion using the Ilizarov technique with circular fixation is a salvage limb-preservation procedure that has shown good results in select patient populations.1-5 Indications include post-traumatic, degenerative, and rheumatoid arthritis, osteomyelitis, tumors, neuromuscular conditions, and salvage of failed ankle and hindfoot procedures.6-9 Relative contraindications include wet gangrene, severe limb ischemia, and soft tissue compromise requiring urgent amputation. In addition, circular frames are not recommended in patients who are unable to comply with postoperative restrictions, and pin and wire care for the duration of frame placement because of personal, psychological, or socioeconomic reasons.
The Ilizarov technique of ring fixation provides dynamic, modular, and rigid fixation in multiple planes to control shear, bending, and rotational forces, and allows for early weight-bearing and postoperative adjustments as needed.10,11 Percutaneously placed half-pins and wires allow for solid fixation in the setting of both poor bone and soft tissue quality, and fusion can be achieved in the presence of active infection in a 1-stage procedure. The goal of ankle and hindfoot fusion using the Ilizarov technique is to achieve an infection-free, stable, plantigrade foot with neutral ankle alignment to allow for patient ambulation and return to activities of daily living.
Nonunion rates with circular fixation are reported to be as high as 16% to 54%, due to medical comorbidities, such as smoking, peripheral vascular disease, and Charcot neuroarthropathy.1Charcot, in particular, is a risk factor for nonunion as patients lack protective sensation, and have a higher rate of wound dehiscence, noncompliance with weight-bearing precautions, pin site infections, and frame breakage. In these patients, tibiotalocalcaneal (TTC) arthrodesis is preferred over the isolated ankle, or subtalar fusion to both provide a stable platform for ambulation and reduce the incidence of adjacent joint breakdown. Common complications of the Ilizarov technique include pin site infections, wire breakage, talar necrosis, and tibial stress fractures after frame removal.1,2,6,11-13 Circular frames are typically maintained for 3 to 8 months, until solid fusion is achieved radiographically. Frames are removed in the operating room with the concurrent examination of the fusion sites under anesthesia followed by a period of protected weight-bearing in a cast or tall controlled ankle motion (CAM) boot.
This article reviews several technical details, tips, and tricks that can help improve the intraoperative and postoperative outcomes of combined ankle and hindfoot arthrodesis using the Ilizarov technique with circular external fixation.
Continue to: SURGICAL TECHNIQUE...
SURGICAL TECHNIQUE
SETUP AND APPROACH
Patients are positioned supine with padding under the operative extremity to achieve neutral leg rotation (Figures 1A-1D). A thigh tourniquet is placed with the foot positioned at the end of the bed and on top of the radiolucent padding to avoid interference of the contralateral leg during lateral X-rays. After sterile prepping and draping, the extremity is exsanguinated above the level of an active infection, and the tourniquet inflated.
For isolated ankle arthrodesis, an anterior or lateral approach can be used, while for TTC arthrodesis, a lateral approach is required to access both the ankle and subtalar joints. A 10-cm longitudinal incision is made along the distal fibula, curving slightly and anteriorly along the distal extent of the incision. Dissection is continued down to bone using full thickness flaps, and the distal fibula is removed 2 to 3 cm above the ankle joint using a saw and osteotome (Figures 2A-2G). The distal fibula can be used subsequently as bone grafts depending on the quality of bone. The peroneal tendons are retracted posteriorly, and dissection is then continued to the posterior facet of the subtalar joint.
JOINT PREPARATION AND ALIGNMENT
Both the anterior and posterior neurovascular bundles are protected along the distal tibia with Hohmann retractors while a saw is used to create flat cuts across the tibial plafond and talus to allow apposition of flat, broad cancellous bony surfaces. Flat cuts followed by later joint compression will often shorten the limb by 2 to 3 cm. This leg length discrepancy can later be accommodated using a shoe lift, as needed. All retained hardware and/or infected and necrotic tissues in the ankle and hindfoot are removed using a rongeur and a pituitary rongeur.
The medial malleolus is osteotomized vertically using a direct medial incision and approach with full thickness flaps, and in line with the previous tibial plafond, is both cut and removed. The medial malleolus can also be used for bone grafts in fusion sites. A smooth-tip lamina spreader is placed in the subtalar joint for distraction and a curved osteotome, curettes, and a small rongeur are used to remove all remaining cartilage from the subtalar joint. Flat cuts in the subtalar joint can remove excessive bone, particularly from the inferior aspect of the talus. The subchondral bone is perforated using a 2.5- to 3.0-mm drill bit and a curved osteotome.
A bone graft from the distal fibula and medial malleolus, with or without the addition of allograft adjuvants, is placed evenly across the ankle and subtalar joints (Figures 3A-3E). At this point, the ankle and subtalar joints can be manipulated in multiple planes to achieve neutral coronal, sagittal, and axial alignment. With both the ankle and hindfoot held in a neutral position, multiple Steinman pins and K-wires in different orientations are inserted through the plantar aspect of the heel to hold the ankle and subtalar joints in place temporarily. Wires are cut short to prevent interference with subsequent foot olive wire placement through the frame.
Continue to: X-rays should be carefully checked...
X-rays should be carefully checked to ensure proper alignment. Wounds are gently irrigated, and vancomycin powder (2 g) can be placed within wounds for local antibiotic delivery. Lateral tissues are sharply debulked to allow for decreased tension on the incision, and small ulcers can be excised in their entirety. Wounds are closed in a layered fashion using 0-polydioxanone (PDS, Ethicon) suture for deep tissue, 2-0 PDS for subcutaneous tissue, and 2-0 nylon for skin closure. The tourniquet is deflated for the remainder of the case to reduce limb ischemia during frame placement.
CIRCULAR FRAME CONCEPTS AND PLACEMENT
The majority of circular frames for both ankle and hindfoot fusion have multiple ring sizes available in aluminum and radiolucent carbon fiber reinforced polymer (Hoffmann LRF, Stryker). Rings are available in full, open, segment, and both short- and long-foot options. Frames can be sterilized in a prebuilt 3 to 4 ring construct with 4 static or dynamic (telescopic) struts (100-277 mm). The most commonly used tibia and foot ring sizes are 155 cm, 180 cm, and 210 cm. Ring size should be able to accommodate posterior soft tissue swelling and avoid circumferential soft tissue abrasion against the rings. Anterior foot arches are used for increased construct stability and can be locked to the distal tibia ring for weight-bearing support. Wire and half-pin bolts, adaptors, and nuts are used to join each ring of the frame to the patient’s bone.
For TTC arthrodesis, 2 rings are typically used in the tibia, and 1 ring is used in the foot. For isolated ankle arthrodesis, an additional ring can be added with olive wires in the talus to permit compression only across the ankle joint. Multiple points of fixation are used in each ring in different planes to achieve both maximal stability and rotational control. If a single wire or half-pin becomes infected and requires removal, there are still multiple other points of fixation in the ring to maintain stability. Fixation within each ring should be off axis compared with the adjacent ring to both avoid stress risers and increase construct rigidity.
The prebuilt frame is checked on the back table to ensure proper orientation and component alignment. The frame is then placed over both the foot and ankle, and multiple stacks of towels are placed behind the heel, ankle, and calf to center the foot and ankle in the frame (Figures 4A-4F). At least 4 to 6 cm of space is needed in between the posterior soft tissues and each ring to accommodate postoperative swelling. On the lateral view, the foot ring should be in the mid-portion of the calcaneus. If there is a concern, particularly in Charcot patients, regarding early weight-bearing noncompliance, the foot ring can be placed flush with the plantar aspect of the foot, and olive wires can be inserted using longer adaptors. The frame should be checked from multiple viewpoints to ensure that both the foot and ankle are centered and in neutral rotation.
Continue to: TIBIA RING FIXATION...
TIBIA RING FIXATION
Tibia rings can be fixed using 2 to 3 half-pins (4-6 mm) alone or 2 half-pins in combination with a smooth wire. A small incision is made over the area of planned half-pin insertion, and the periosteum is cleared away using a hemostat. An adaptor sleeve is used, and the bone is drilled bicortically, followed by insertion of the half-pin. Hydroxyapatite-coated pins are used to improve the strength of the bone-pin interface and reduce the incidence of pin tract infections. Pins are inserted along both the anterior and medial aspects of the tibia, avoiding the thick lateral musculature. Care is taken to protect the medial neurovascular structures during pin placement following established Ilizarov safe zones.
After each pin is placed in the bone, the pin is secured to the adaptor that is then tightened to the ring. This process is repeated for both the proximal and distal tibia rings. Pins should be placed above and below each ring to avoid creating stress risers. During smooth wire placement, each wire is pushed by hand through the soft tissues and then drilled into the bone while the exposed segment is held with a damp sponge to reduce the incidence of thermal bone necrosis. Once the wire is drilled bicortically, a mallet is used to tap the wire through the remaining soft tissues to avoid wrapping them up in the wire. Each wire should be parallel to the ring to get an even line of compression.
Each wire is secured on 1 end and then tensioned to 130 kg using a hand tensioner. An additional tool can be placed in the wire adaptor to prevent the wire from bending during tensioning. If the wire is passing above or below the ring, longer wire adaptors should be used to build to the wire. The wire should never be bent toward the ring as this can increase the likelihood of improper pin tensioning and breakage. Wire placement should be avoided posteriorly as this can make it difficult to secure and/or tension wires, and also increases the risk of damage to posterior structures.
Ring fixation in the distal tibia near the plafond may require 1 half-pin and 2 wires to avoid damage to the tibialis anterior and posterior tibial tendons. In this case, smooth wires should be placed in a crossing pattern and tensioned simultaneously to avoid pulling the ankle away from the center of the frame. Wires should be bent and curved over each ring and then cut to facilitate subsequent removal.
FOOT RING FIXATION
In the foot, olive wires are used to increase fixation against bone. For each olive wire, a small incision is made to accommodate the diameter of the olive through the soft tissue. Similar to the distal tibia, 2 olive wires should be placed above and below the foot ring in a crossing pattern through the calcaneus (Figures 5A-5F). The axial view of the frame should be checked to ensure proper wire orientation. When using olive wires, it is essential to tension both at the same time to 90 kg, as the foot can be pulled medially or laterally in the frame if 1 wire is tensioned before the other.
Forefoot olive wires should also be placed in a crossing pattern, with 1 wire fixed through the first, second, and third metatarsals, and 1 wire through the fourth and fifth metatarsals. Additional forefoot olive wires can be placed if compression is needed across the midfoot or Chopart joints for fusion. Multiple X-rays should be checked to ensure that the calcaneus and forefoot olive wires are firmly fixed both in and against bone.
Continue to: JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS...
JOINT COMPRESSION AND FINAL FRAME ADJUSTMENTS
Once all rings are secured to the bone with half-pins and wires, the previously placed Steinman pins, and K-wires through the heel are removed. Both ankle and subtalar joint alignments are rechecked, and then axial compression is placed through the foot ring with the knee extended and the struts unlocked. Static or telescopic struts are used to achieve 8 to 10 mm of bony compression. X-rays are taken before and after to analyze final joint compression and alignment. Struts should be sequentially tightened (1/2 turn of a static strut) 1 at a time as final tightening of 1 strut alone can bind and interfere with both the compression and tightening of the remaining struts.
Once final compression is achieved, the struts are locked, and the front foot arch is closed anteriorly and connected to the distal tibia ring for increased stability (Figures 6A-6D). Each pin and wire is covered in a sterile dressing followed by gauze to allow for soft tissue padding. The entire frame is then overwrapped in bias stockinette rolls or ace wraps.
Walking attachments can be added immediately to the frame that allows for early weight-bearing. Rocker shoe attachments with a 15° anterior and posterior slope and rubber soles can help offload the ankle and subtalar joints, decrease pressure on heel strike, and reduce ankle motion during ambulation (Hoffmann LRF, Stryker).
POSTOPERATIVE PROTOCOL
Depending on individual characteristics, patients can be immediately weight-bearing in the circular frame. Patients with Charcot neuroarthropathy are recommended to remain non-weight-bearing for the first 2 months to reduce the likelihood of pin, wire, and frame breakage along with nonunion. Pin and wire site care and maintenance are initiated the day after surgery and continue on a daily basis for the duration of frame placement. Sutures are removed 4 to 5 weeks after surgery to ensure adequate wound healing. Serial X-rays are taken monthly to analyze fusion sites.
If pins or wires become infected, patients are placed on oral antibiotics, and both pins and wires can be removed or exchanged in the operating room. Once fusion is achieved in 3 to 8 months (Figures 7A-7C), the frame is removed in the operating room, and fusion sites are examined under dynamic fluoroscopy. If fusion is confirmed, patients are made weight-bearing as tolerated in a short-leg cast or tall CAM boot for 6 to 8 weeks, and then transitioned to an ankle brace in an accommodative shoe.
Continue to: DISCUSSION...
DISCUSSION
A key aspect of recovery after ankle and hindfoot fusion using the Ilizarov technique is balancing pin care, soft tissue swelling, and weight-bearing status. The average time patients will spend in the frame is approximately 25 to 28 weeks, but can range from 12 to 84 weeks.1,2Given the considerable variability in both soft tissue healing and bony union, patients should be extensively counseled before surgery to set expectations correctly and ensure that they have the necessary help and support to care for the frame during the treatment period. Patients should be followed closely during the first 6 weeks to ensure that pins and wires do not become infected or break, as both of these issues require immediate intervention.
In a review of 11 patients who underwent tibiocalcaneal arthrodesis using an Ilizarov external fixator for infected talar nonunions or extrusions, Rochman and colleagues8 reported an 81% rate of successful fusion with a final mean American Orthopaedic Foot and Ankle Society score of 65 (out of a maximum 86). Similar results were reported by Saltzman9 in a series of 8 patients with diffuse ankle osteomyelitis treated with resection of all infected tissue and hybrid-frame compression arthrodesis. All patients received 6 weeks of intravenous antibiotics, and frames were removed at 3 months, and walking casts were applied for 1 to 2 additional months. Ankle sepsis was eradicated in all patients, and 7/8 (87.5%) ankles successfully fused at an average of 13.5 weeks (range, 10-16 weeks). One limb required below-knee amputation at 5 weeks due to non-reconstructible vascular insufficiency. At an average of 3.4-year follow-up, none of the 7 fused ankles required further surgery.
Fragomen and colleagues1 retrospectively reviewed 101 patients who underwent complex ankle fusion using the Ilizarov technique and found that 76/91 (83.5%) patients achieved fusion at an average of 25 weeks (range, 10-65 weeks). Smoking was associated with a 54% rate of nonunion and 15/19 (79%) patients with Charcot neuroarthropathy achieved ankle fusion, but had a subsequent subtalar joint failure, thus highlighting the need for TTC arthrodesis in Charcot patients. Salem and colleagues2 reviewed 21 Ilizarov ankle fusions and reported that all patients achieved fusion at an average of 28 weeks (range, 12-84 weeks). Complications occurred in 11 patients, including 2 nonunions that healed after revision frame application and 4 pin tract infections.
CONCLUSION
Overall, the Ilizarov technique using circular external fixation is a powerful tool that can be used to treat a variety of disorders including complex foot and ankle deformity and infection. While case series generally show favorable outcomes, patients must be informed that this technique is a salvage procedure for limb preservation that requires meticulous operative technique, diligent postoperative care, and tight control of medical comorbidities, such as blood sugar levels in individuals with diabetes to achieve a successful outcome.
1. Fragomen AT, Borst E, Schachter L, Lyman S, Rozbruch SR. Complex ankle arthrodesis using the Ilizarov method yields high rate of fusion. Clin Orthop Relat Res. 2012;470(10):2864-2873. doi:10.1007/s11999-012-2470-9.
2. Salem KH, Kinzl L, Schmelz A. Ankle arthrodesis using Ilizarov ring fixators: a review of 22 cases. Foot Ankle Int. 2006;27(10):764-770. doi:10.1177/107110070602701002.
3. Cierny G 3rd, Cook WG, Mader JT. Ankle arthrodesis in the presence of ongoing sepsis. Indications, methods, and results. Orthop Clin North Am. 1989;20(4):709-721.
4. Dalla Paola L, Brocco E, Ceccacci T, et al. Limb salvage in Charcot foot and ankle osteomyelitis: combined use single stage/double stage of arthrodesis and external fixation. Foot Ankle Int. 2009;30(11):1065-1070. doi:10.3113/FAI.2009.1065.
5. Eylon S, Porat S, Bor N, Leibner ED. Outcome of Ilizarov ankle arthrodesis. Foot Ankle Int. 2007;28(8):873-879. doi:10.3113/FAI.2007.0873.
6. Kalish S, Fleming J, Weinstein R. External fixators for elective rearfoot and ankle arthrodesis. Techniques and indications. Clin Podiatr Med Surg. 2003;20(1):65-96, vi.
7. Kollig E, Esenwein SA, Muhr G, Kutscha-Lissberg F. Fusion of the septic ankle: experience with 15 cases using hybrid external fixation. J Trauma. 2003;55(4):685-691. doi:10.1097/01.TA.0000051933.83342.E4.
8. Rochman R, Jackson Hutson J, Alade O. Tibiocalcaneal arthrodesis using the Ilizarov technique in the presence of bone loss and infection of the talus. Foot Ankle Int. 2008;29(10):1001-1008. doi:10.3113/FAI.2008.1001.
9. Saltzman CL. Salvage of diffuse ankle osteomyelitis by single-stage resection and circumferential frame compression arthrodesis. Iowa Orthop J. 2005;2547-52.
10. Fragomen AT, Rozbruch SR. The mechanics of external fixation. HSS J. 2007;3(1):13-29. doi:10.1007/s11420-006-9025-0.
11. Hawkins BJ, Langerman RJ, Anger DM, Calhoun JH. The Ilizarov technique in ankle fusion. Clin Orthop Relat Res. 1994;(303):217-225.
12. Jones CP, Youngblood CS, Waldrop N, Davis WH, Pinzur MS. Tibial Stress Fracture Secondary to Half-Pins in Circular Ring External Fixation for Charcot Foot. Foot Ankle Int. 2014;35(6):572-577. doi:10.1177/1071100714531229.
13. Kazmers NH, Fragomen AT, Rozbruch SR. Prevention of pin site infection in external fixation: a review of the literature. Strategies Trauma Limb Reconstr. 2016;11(2):75-85. doi:10.1007/s11751-016-0256-4.
1. Fragomen AT, Borst E, Schachter L, Lyman S, Rozbruch SR. Complex ankle arthrodesis using the Ilizarov method yields high rate of fusion. Clin Orthop Relat Res. 2012;470(10):2864-2873. doi:10.1007/s11999-012-2470-9.
2. Salem KH, Kinzl L, Schmelz A. Ankle arthrodesis using Ilizarov ring fixators: a review of 22 cases. Foot Ankle Int. 2006;27(10):764-770. doi:10.1177/107110070602701002.
3. Cierny G 3rd, Cook WG, Mader JT. Ankle arthrodesis in the presence of ongoing sepsis. Indications, methods, and results. Orthop Clin North Am. 1989;20(4):709-721.
4. Dalla Paola L, Brocco E, Ceccacci T, et al. Limb salvage in Charcot foot and ankle osteomyelitis: combined use single stage/double stage of arthrodesis and external fixation. Foot Ankle Int. 2009;30(11):1065-1070. doi:10.3113/FAI.2009.1065.
5. Eylon S, Porat S, Bor N, Leibner ED. Outcome of Ilizarov ankle arthrodesis. Foot Ankle Int. 2007;28(8):873-879. doi:10.3113/FAI.2007.0873.
6. Kalish S, Fleming J, Weinstein R. External fixators for elective rearfoot and ankle arthrodesis. Techniques and indications. Clin Podiatr Med Surg. 2003;20(1):65-96, vi.
7. Kollig E, Esenwein SA, Muhr G, Kutscha-Lissberg F. Fusion of the septic ankle: experience with 15 cases using hybrid external fixation. J Trauma. 2003;55(4):685-691. doi:10.1097/01.TA.0000051933.83342.E4.
8. Rochman R, Jackson Hutson J, Alade O. Tibiocalcaneal arthrodesis using the Ilizarov technique in the presence of bone loss and infection of the talus. Foot Ankle Int. 2008;29(10):1001-1008. doi:10.3113/FAI.2008.1001.
9. Saltzman CL. Salvage of diffuse ankle osteomyelitis by single-stage resection and circumferential frame compression arthrodesis. Iowa Orthop J. 2005;2547-52.
10. Fragomen AT, Rozbruch SR. The mechanics of external fixation. HSS J. 2007;3(1):13-29. doi:10.1007/s11420-006-9025-0.
11. Hawkins BJ, Langerman RJ, Anger DM, Calhoun JH. The Ilizarov technique in ankle fusion. Clin Orthop Relat Res. 1994;(303):217-225.
12. Jones CP, Youngblood CS, Waldrop N, Davis WH, Pinzur MS. Tibial Stress Fracture Secondary to Half-Pins in Circular Ring External Fixation for Charcot Foot. Foot Ankle Int. 2014;35(6):572-577. doi:10.1177/1071100714531229.
13. Kazmers NH, Fragomen AT, Rozbruch SR. Prevention of pin site infection in external fixation: a review of the literature. Strategies Trauma Limb Reconstr. 2016;11(2):75-85. doi:10.1007/s11751-016-0256-4.
TAKE-HOME POINTS
- Ankle and hindfoot fusion using circular external fixation is a useful surgical technique in patients with diabetes, Charcot, osteomyelitis, deformity, and/or bone and soft tissue compromise in order to obtain solid bony fusion, stable limb alignment, and eradication of infection in cases of complex pathology.
- Deformity correction with osteotomies and meticulous joint preparation is required in order to obtain broad, cancellous bony surfaces for fusion with neutral alignment. Autograft from the distal fibula and/or medial malleolus can be combined with bone allograft to assist with joint fusion.
- The ankle and hindfoot are provisionally pinned into neutral coronal and sagittal alignment through the plantar surface of the foot using large K-wires prior to placement of the lower leg in the center of a circular 3-ring compression frame. Typically, 2 to 3 points of fixation are used per ring with a combination of half-pins and smooth wires.
- Ring attachments are built up or down to the level of the half-pins and wires in order to prevent pins and wires from bending, breaking, or causing iatrogenic deformity during tensioning. Crossing olive wires are used in the midfoot and calcaneus with dual tensioning devices to ensure an even pull on both sides of the foot.
- Dynamic or static compression struts are used to obtain 8 to 10 mm of compression across the ankle and hindfoot, followed by addition of an anterior foot ring to increase construct rigidity. Daily pin care is started 3 to 4 days after surgery and patients are kept non-weight-bearing for approximately 2 months in the frame with a total frame period of 3 to 8 months depending on bony healing on X-ray.
Neoadjuvant combo yields high pathologic response in NSCLC
TORONTO – Neoadjuvant combined chemotherapy and immunotherapy (CT-IO) yielded high pathologic response rates for locally advanced, potentially resectable non-small cell lung cancer in the phase 2 multicenter study Nadim study.
All 30 adults with stage IIIA N2 non-small cell lung cancer (NSCLC) who enrolled in the ongoing single-arm, open-label study and have undergone surgery to date, had resectable tumors following neoadjuvant treatment, Mariano Provencio, MD, of Hospital Puerta de Hierro, Madrid, Spain, reported at the World Conference on Lung Cancer.
The complete and partial response rates following the treatment were 10% and 60%, respectively, and the remaining patients had stable disease, Dr. Provencio said, noting that an additional 10 enrolled patients have completed treatment and are awaiting resection, and another 3 are still completing treatment.
The overall major pathologic response rate was 80%, including 75% with complete remission, he said at the conference, which was sponsored by the International Association for the Study of Lung Cancer.
“With a median follow-up of 4 months, no patients suffered any recurrence,” he said. “We have been unable to identify any significant factor related to the response, but we still do not have information about biomarkers.”
The study participants had a median age of 64 years, most (73%) were men, and all were current or former smokers. All received the planned neoadjuvant doses, which included 3 cycles of intravenous nivolumab at a dose of 360 mg every 3 weeks, plus 200 mg/m2 of paclitaxel and carboplatin at AUC 6 every 3 weeks.
“The most important toxicity was hematological toxicity related to chemotherapy; there were very few episodes of non-hematological toxicity,” he said.
Tumors were then assessed, and surgery–lobectomy in 90% of cases–was performed in the 3rd or 4th week after day 21 of the third neoadjuvant treatment cycle.
“There were no intraoperative complications, and 7 patients experienced some postoperative complications, but there was no postoperative mortality,” he said.
Adjuvant treatment included 240 mg of intravenous nivolumab every 2 weeks for 4 months and then 480 mg every 4 weeks for 8 months after resection.
The primary end-point of the study is 24-month progression-free survival.
CT-IO has a high response rate and longer survival in unselected patients with metastatic NSCLC, but there were previously no data about this combination in the neoadjuvant setting, Dr. Provencio said.
The findings of this ongoing study are limited by their preliminary nature and the related lack of information about overall survival and biomarkers, Dr. Provencio said.
“But the higher complete remission and major pathological response rates make this a promising future treatment in stage 3 [NSCLC,] he said.
Indeed, the Nadim study thus far has shown “an amazing result that needs to be verified ... [there is] no prospective proof yet in this context that major pathologic response is parlayed with improved [progression-free survival] or [overall survival],” said discussant Corey Langer, MD, professor of medicine and director of thoracic oncology at the University of Pennsylvania, Philadelphia. “We need to track the late sequelae and [central nervous system] recurrences.”
Dr. Provencio reported relationships with AstraZeneca, Bristol-Meyers Squibb, Fabre, Merck, Pierre, Roche, and Bristol-Meyers Squibb. Dr. Langer reported relationships with numerous companies and organizations.
SOURCE: Provencio, Mario et al., WCLC 2018 Abstract OA01.05.
TORONTO – Neoadjuvant combined chemotherapy and immunotherapy (CT-IO) yielded high pathologic response rates for locally advanced, potentially resectable non-small cell lung cancer in the phase 2 multicenter study Nadim study.
All 30 adults with stage IIIA N2 non-small cell lung cancer (NSCLC) who enrolled in the ongoing single-arm, open-label study and have undergone surgery to date, had resectable tumors following neoadjuvant treatment, Mariano Provencio, MD, of Hospital Puerta de Hierro, Madrid, Spain, reported at the World Conference on Lung Cancer.
The complete and partial response rates following the treatment were 10% and 60%, respectively, and the remaining patients had stable disease, Dr. Provencio said, noting that an additional 10 enrolled patients have completed treatment and are awaiting resection, and another 3 are still completing treatment.
The overall major pathologic response rate was 80%, including 75% with complete remission, he said at the conference, which was sponsored by the International Association for the Study of Lung Cancer.
“With a median follow-up of 4 months, no patients suffered any recurrence,” he said. “We have been unable to identify any significant factor related to the response, but we still do not have information about biomarkers.”
The study participants had a median age of 64 years, most (73%) were men, and all were current or former smokers. All received the planned neoadjuvant doses, which included 3 cycles of intravenous nivolumab at a dose of 360 mg every 3 weeks, plus 200 mg/m2 of paclitaxel and carboplatin at AUC 6 every 3 weeks.
“The most important toxicity was hematological toxicity related to chemotherapy; there were very few episodes of non-hematological toxicity,” he said.
Tumors were then assessed, and surgery–lobectomy in 90% of cases–was performed in the 3rd or 4th week after day 21 of the third neoadjuvant treatment cycle.
“There were no intraoperative complications, and 7 patients experienced some postoperative complications, but there was no postoperative mortality,” he said.
Adjuvant treatment included 240 mg of intravenous nivolumab every 2 weeks for 4 months and then 480 mg every 4 weeks for 8 months after resection.
The primary end-point of the study is 24-month progression-free survival.
CT-IO has a high response rate and longer survival in unselected patients with metastatic NSCLC, but there were previously no data about this combination in the neoadjuvant setting, Dr. Provencio said.
The findings of this ongoing study are limited by their preliminary nature and the related lack of information about overall survival and biomarkers, Dr. Provencio said.
“But the higher complete remission and major pathological response rates make this a promising future treatment in stage 3 [NSCLC,] he said.
Indeed, the Nadim study thus far has shown “an amazing result that needs to be verified ... [there is] no prospective proof yet in this context that major pathologic response is parlayed with improved [progression-free survival] or [overall survival],” said discussant Corey Langer, MD, professor of medicine and director of thoracic oncology at the University of Pennsylvania, Philadelphia. “We need to track the late sequelae and [central nervous system] recurrences.”
Dr. Provencio reported relationships with AstraZeneca, Bristol-Meyers Squibb, Fabre, Merck, Pierre, Roche, and Bristol-Meyers Squibb. Dr. Langer reported relationships with numerous companies and organizations.
SOURCE: Provencio, Mario et al., WCLC 2018 Abstract OA01.05.
TORONTO – Neoadjuvant combined chemotherapy and immunotherapy (CT-IO) yielded high pathologic response rates for locally advanced, potentially resectable non-small cell lung cancer in the phase 2 multicenter study Nadim study.
All 30 adults with stage IIIA N2 non-small cell lung cancer (NSCLC) who enrolled in the ongoing single-arm, open-label study and have undergone surgery to date, had resectable tumors following neoadjuvant treatment, Mariano Provencio, MD, of Hospital Puerta de Hierro, Madrid, Spain, reported at the World Conference on Lung Cancer.
The complete and partial response rates following the treatment were 10% and 60%, respectively, and the remaining patients had stable disease, Dr. Provencio said, noting that an additional 10 enrolled patients have completed treatment and are awaiting resection, and another 3 are still completing treatment.
The overall major pathologic response rate was 80%, including 75% with complete remission, he said at the conference, which was sponsored by the International Association for the Study of Lung Cancer.
“With a median follow-up of 4 months, no patients suffered any recurrence,” he said. “We have been unable to identify any significant factor related to the response, but we still do not have information about biomarkers.”
The study participants had a median age of 64 years, most (73%) were men, and all were current or former smokers. All received the planned neoadjuvant doses, which included 3 cycles of intravenous nivolumab at a dose of 360 mg every 3 weeks, plus 200 mg/m2 of paclitaxel and carboplatin at AUC 6 every 3 weeks.
“The most important toxicity was hematological toxicity related to chemotherapy; there were very few episodes of non-hematological toxicity,” he said.
Tumors were then assessed, and surgery–lobectomy in 90% of cases–was performed in the 3rd or 4th week after day 21 of the third neoadjuvant treatment cycle.
“There were no intraoperative complications, and 7 patients experienced some postoperative complications, but there was no postoperative mortality,” he said.
Adjuvant treatment included 240 mg of intravenous nivolumab every 2 weeks for 4 months and then 480 mg every 4 weeks for 8 months after resection.
The primary end-point of the study is 24-month progression-free survival.
CT-IO has a high response rate and longer survival in unselected patients with metastatic NSCLC, but there were previously no data about this combination in the neoadjuvant setting, Dr. Provencio said.
The findings of this ongoing study are limited by their preliminary nature and the related lack of information about overall survival and biomarkers, Dr. Provencio said.
“But the higher complete remission and major pathological response rates make this a promising future treatment in stage 3 [NSCLC,] he said.
Indeed, the Nadim study thus far has shown “an amazing result that needs to be verified ... [there is] no prospective proof yet in this context that major pathologic response is parlayed with improved [progression-free survival] or [overall survival],” said discussant Corey Langer, MD, professor of medicine and director of thoracic oncology at the University of Pennsylvania, Philadelphia. “We need to track the late sequelae and [central nervous system] recurrences.”
Dr. Provencio reported relationships with AstraZeneca, Bristol-Meyers Squibb, Fabre, Merck, Pierre, Roche, and Bristol-Meyers Squibb. Dr. Langer reported relationships with numerous companies and organizations.
SOURCE: Provencio, Mario et al., WCLC 2018 Abstract OA01.05.
REPORTING FROM WCLC 2018
Key clinical point: Neoadjuvant CT-IO yielded high pathologic response rates for stage IIIA N2 NSCLC.
Major finding: The major pathologic response rate was 80%, including 10% with a complete response.
Study details: A phase 2, single-arm, open-label study of 30 patients.
Disclosures: Dr. Provencio reported relationships with AstraZeneca, Bristol-Meyers Squibb, Fabre, Merck, Pierre, Roche, and Bristol-Meyers Squibb. Dr. Langer reported relationships with numerous companies and organizations.
Source: Provencio, Mario et al., WCLC 2018 Abstract OA01.05.
Transition to noninjected drug use lowers risk of HCV infection
A protective effect was seen with regard to hepatitis C virus (HCV) transmission among individuals who “reversed transitioned” to noninjected drug use and in persons who used noninjected drugs in addition to injecting, according to an interview study of 846 drug users.
Data were collected between January 2007 and December 2017 as part of a long-running study of persons entering Mount Sinai Beth Israel drug detoxification and methadone maintenance programs, reported Don C. Des Jarlais, MD, of the Icahn School of Medicine at Mount Sinai, New York, and his colleagues.
Among the 846 individuals studied, men comprised 79%; 41% were white, 15% African American, and 40% Hispanic, with a mean age of 35 years. They were all currently persons who injected drugs (PWID), according to the report published in Drug and Alcohol Dependence.
A total of 97 persons (11%) “reverse transitioned” back to noninjected drug use, according to the researchers; this reverse transitioning was strongly associated with lower HCV positivity, compared with those who continued injecting (30% vs. 47%, respectively; P less than .005).
There was a substantial difference in the HCV prevalence between PWID who reported noninjected use of heroin (43%) versus PWID who did not report noninjected use of heroin (53%) (P = .005).
Distributive needle sharing was common in HCV seropositives who were injecting cocaine in the 6 months prior to the interview: 28% of the HCV seropositives injecting cocaine reported distributive sharing versus 14% of the HCV seropositives not injecting cocaine (P = .001).
“Harm reduction for PWID has been largely defined in terms of ‘safer injection,’ using sterile injection equipment, proper preparation of the injecting site, etc. We would like to suggest that harm reduction for PWID be broadened to include reverse transitions back to exclusive noninjecting use and frequent substitution of noninjecting for injecting use,” the investigators suggested.
The study was funded by the U.S. National Institute on Drug Abuse. The authors reported that they had no conflicts of interest.
SOURCE: Des Jarlais DC et al. Drug Alcohol Depend. 2018 Sep 12. doi: 10.1016/j.drugalcdep.2018.07.034.
A protective effect was seen with regard to hepatitis C virus (HCV) transmission among individuals who “reversed transitioned” to noninjected drug use and in persons who used noninjected drugs in addition to injecting, according to an interview study of 846 drug users.
Data were collected between January 2007 and December 2017 as part of a long-running study of persons entering Mount Sinai Beth Israel drug detoxification and methadone maintenance programs, reported Don C. Des Jarlais, MD, of the Icahn School of Medicine at Mount Sinai, New York, and his colleagues.
Among the 846 individuals studied, men comprised 79%; 41% were white, 15% African American, and 40% Hispanic, with a mean age of 35 years. They were all currently persons who injected drugs (PWID), according to the report published in Drug and Alcohol Dependence.
A total of 97 persons (11%) “reverse transitioned” back to noninjected drug use, according to the researchers; this reverse transitioning was strongly associated with lower HCV positivity, compared with those who continued injecting (30% vs. 47%, respectively; P less than .005).
There was a substantial difference in the HCV prevalence between PWID who reported noninjected use of heroin (43%) versus PWID who did not report noninjected use of heroin (53%) (P = .005).
Distributive needle sharing was common in HCV seropositives who were injecting cocaine in the 6 months prior to the interview: 28% of the HCV seropositives injecting cocaine reported distributive sharing versus 14% of the HCV seropositives not injecting cocaine (P = .001).
“Harm reduction for PWID has been largely defined in terms of ‘safer injection,’ using sterile injection equipment, proper preparation of the injecting site, etc. We would like to suggest that harm reduction for PWID be broadened to include reverse transitions back to exclusive noninjecting use and frequent substitution of noninjecting for injecting use,” the investigators suggested.
The study was funded by the U.S. National Institute on Drug Abuse. The authors reported that they had no conflicts of interest.
SOURCE: Des Jarlais DC et al. Drug Alcohol Depend. 2018 Sep 12. doi: 10.1016/j.drugalcdep.2018.07.034.
A protective effect was seen with regard to hepatitis C virus (HCV) transmission among individuals who “reversed transitioned” to noninjected drug use and in persons who used noninjected drugs in addition to injecting, according to an interview study of 846 drug users.
Data were collected between January 2007 and December 2017 as part of a long-running study of persons entering Mount Sinai Beth Israel drug detoxification and methadone maintenance programs, reported Don C. Des Jarlais, MD, of the Icahn School of Medicine at Mount Sinai, New York, and his colleagues.
Among the 846 individuals studied, men comprised 79%; 41% were white, 15% African American, and 40% Hispanic, with a mean age of 35 years. They were all currently persons who injected drugs (PWID), according to the report published in Drug and Alcohol Dependence.
A total of 97 persons (11%) “reverse transitioned” back to noninjected drug use, according to the researchers; this reverse transitioning was strongly associated with lower HCV positivity, compared with those who continued injecting (30% vs. 47%, respectively; P less than .005).
There was a substantial difference in the HCV prevalence between PWID who reported noninjected use of heroin (43%) versus PWID who did not report noninjected use of heroin (53%) (P = .005).
Distributive needle sharing was common in HCV seropositives who were injecting cocaine in the 6 months prior to the interview: 28% of the HCV seropositives injecting cocaine reported distributive sharing versus 14% of the HCV seropositives not injecting cocaine (P = .001).
“Harm reduction for PWID has been largely defined in terms of ‘safer injection,’ using sterile injection equipment, proper preparation of the injecting site, etc. We would like to suggest that harm reduction for PWID be broadened to include reverse transitions back to exclusive noninjecting use and frequent substitution of noninjecting for injecting use,” the investigators suggested.
The study was funded by the U.S. National Institute on Drug Abuse. The authors reported that they had no conflicts of interest.
SOURCE: Des Jarlais DC et al. Drug Alcohol Depend. 2018 Sep 12. doi: 10.1016/j.drugalcdep.2018.07.034.
FROM DRUG AND ALCOHOL DEPENDENCE
Key clinical point: Encouraging reverse transitioning to noninjected drug use could help persons who injected drugs lower their risk of hepatitis C virus infection.
Major finding: Reverse transitioning was strongly associated with lower hepatitis C virus positivity, compared with continued drug injection (P less than .005).
Study details: An interview study of 846 drug users.
Disclosures: The study was funded by the U.S. National Institute on Drug Abuse. The authors reported that they had no conflicts of interest.
Source: Des Jarlais DC et al. Drug Alcohol Depend. 2018 Sep 12. doi: 10.1016/j.drugalcdep.2018.07.034.
5 things to know about Trump’s new ‘public charge’ immigration proposal
A proposed rule from the White House would make it harder for legal immigrants to get green cards if they have received certain kinds of public assistance – including Medicaid, food stamps, and housing subsidies. Green cards allow them to live and work permanently in the United States.
“Those seeking to immigrate to the United States must show they can support themselves financially,” Homeland Security Secretary Kirstjen Nielsen said in a statement.
The proposal, announced Sept. 22, marks a new frontier in the administration’s long-term effort to curb immigration, both legal and illegal. It already has spurred intense criticism from Democrats, anti-poverty activists, health care organizations, and immigrants’ rights advocates, who call its restrictions unprecedented.
“We are operating in an overall climate of tremendous fear and anxiety as a result of the administration’s overall approach to immigration enforcement and immigration policy,” said Mark Greenberg, a senior fellow at the Migration Policy Institute, which studies migration and refugee policies at local, national, and international levels. He is also a former Obama administration official.
But what effect would this proposal have?
It’s a complicated question, touching upon vast government programs, with billions of dollars at stake. While the implications aren’t all immediately clear, Kaiser Health News breaks down some of the key elements.
1. First thing first: What is the White House proposing?
The Trump administration wants to redefine a status known as “public charge” – a category used to determine whether someone seeking permanent resident status is “likely to become primarily dependent on the government for subsistence.”
In the past, people have been at risk of being defined a “public charge” if they took cash welfare – known as Temporary Assistance for Needy Families, or Supplemental Security Income – or federal help paying for long-term care. (Immigrants must be in the country legally for 5 years before being eligible for TANF or SSI.)
And that “public charge” designation could undermine their applications for permanent residence.
The new rule would expand the list to include some health insurance, food, and housing programs. Specifically, it would penalize green-card applicants for using Medicaid, a federal-state health plan for low-income people. (Penalties would not apply for using Medicaid in certain emergencies or for some Medicaid services provided through schools and disability programs.)
Using food stamps, Section 8 rental assistance, and federal housing vouchers also would count against applicants. Enrollment in a Medicare Part D program subsidy to help low-income people buy prescription drugs would work against them, too.
The proposal “is definitely a dramatic change from how public charge works today,” said Kelly Whitener, an associate professor at Georgetown University’s Center for Children and Families who specializes in pediatric health benefits and managed-care systems.
A leaked version of the rule from March suggested officials then were also considering penalizing those who receive subsidies to buy health insurance on the Affordable Care Act marketplaces. But that idea was not in the proposal. The marketplace subsidies are aimed at people at a generally higher income bracket than the beneficiaries targeted in the Trump plan, Whitener noted.
“They’re really homing in on low-income immigrants,” she added.
Nielsen said the proposed rule is “intended to promote immigrant self-sufficiency and protect finite resources.”
2. Is this as unprecedented as critics say?
Yes.
Public charge is an old idea. In the 1990s, lawmakers expanded it to consider explicitly whether people had received cash-based welfare.
But including programs like Medicaid and food stamps, which are much wider in scope, is a significant change. It would more likely hit working people – the majority of people on Medicaid are themselves employed, and almost 80% live in families with at least one working member, according to data compiled by the Kaiser Family Foundation. (Kaiser Health News is an editorially independent program of the foundation.)
Children who are American citizens but whose parents are immigrants could be more likely to suffer repercussions, said some experts. When parents opt out of public assistance for fear of their own legal status, their kids are less likely to be enrolled in programs such as the Children’s Health Insurance Program, or CHIP, for which they would qualify.
To be clear, receiving public aid wouldn’t necessarily stop people from getting a green card. But it would tilt the odds against them.
“Another piece is the enormous discretion the administration will have under its proposal in making judgments about who gets admitted to the country and who gets a green card,” said the Migration Policy Institute’s Greenberg.
3. When will the policy shift take effect?
This is an early step in the complex federal rule-making process. And a lot could still change.
Once the proposed rule appears in the Federal Register, a 60-day countdown starts, during which anyone can weigh in with comments.
A final rule likely wouldn’t take effect until 2019.
And DHS is still seeking input on some details. For instance, it hasn’t decided whether CHIP would be counted as one of the “public charge” eligible programs.
In the interim, people who had received public benefits before the rule took effect would not be penalized for doing so.
4. Already, though, the proposal is having effects
DHS estimates that 2.5% of eligible immigrants would drop out of public benefits programs because of this change – which would tally about $1.5 billion worth of federal money per year. But others expect a much larger impact.
“The chilling effects will be vastly greater than the individuals directly affected,” Greenberg said. “There’s considerable reason to believe that [the White House estimate] may be a significant understatement.”
In the proposed rule, DHS notes that the changes could result in “worse health outcomes,” “increased use of emergency rooms,” “increased prevalence of communicable diseases,” “increased rates of poverty,” and other concerns.
Given the complexity of these programs and the proposed rule – and the high stakes at play – low-income immigrants would be much more likely to avoid public benefits altogether, immigration experts said. Millions of immigrants are likely to be affected directly or indirectly, according to the Center for Law and Social Policy, a D.C.-based nonprofit organization.
That could have stark health implications.
Take free vaccines, for which children are often eligible and which would not be subject to the public charge rule. Families afraid of jeopardizing a green card could still be more likely to opt out of that service, Whitener said.
Already, she added, there are reports of people declining federal assistance – even though nothing has yet happened.
“The fear factor cannot be underestimated,” she said.
5. Will people sue?
Legal action is likely.
Officials such as California Attorney General Xavier Becerra, who has frequently clashed with the White House, are weighing challenges to the rule.
“The Trump Administration’s proposal punishes hard-working immigrant families – even targeting children who are citizens – for utilizing programs that provide basic nutrition and healthcare. This is an assault on our families and our communities,” Becerra said in a statement.
But these actions depend on the final shape of the regulation, which could change through the rule-making process.
“They are likely to receive a very large number of sharply critical comments, and there is no way to know what changes they might make as a result,” Greenberg said.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente. KHN’s coverage of children’s health care issues is supported in part by the Heising-Simons Foundation.
A proposed rule from the White House would make it harder for legal immigrants to get green cards if they have received certain kinds of public assistance – including Medicaid, food stamps, and housing subsidies. Green cards allow them to live and work permanently in the United States.
“Those seeking to immigrate to the United States must show they can support themselves financially,” Homeland Security Secretary Kirstjen Nielsen said in a statement.
The proposal, announced Sept. 22, marks a new frontier in the administration’s long-term effort to curb immigration, both legal and illegal. It already has spurred intense criticism from Democrats, anti-poverty activists, health care organizations, and immigrants’ rights advocates, who call its restrictions unprecedented.
“We are operating in an overall climate of tremendous fear and anxiety as a result of the administration’s overall approach to immigration enforcement and immigration policy,” said Mark Greenberg, a senior fellow at the Migration Policy Institute, which studies migration and refugee policies at local, national, and international levels. He is also a former Obama administration official.
But what effect would this proposal have?
It’s a complicated question, touching upon vast government programs, with billions of dollars at stake. While the implications aren’t all immediately clear, Kaiser Health News breaks down some of the key elements.
1. First thing first: What is the White House proposing?
The Trump administration wants to redefine a status known as “public charge” – a category used to determine whether someone seeking permanent resident status is “likely to become primarily dependent on the government for subsistence.”
In the past, people have been at risk of being defined a “public charge” if they took cash welfare – known as Temporary Assistance for Needy Families, or Supplemental Security Income – or federal help paying for long-term care. (Immigrants must be in the country legally for 5 years before being eligible for TANF or SSI.)
And that “public charge” designation could undermine their applications for permanent residence.
The new rule would expand the list to include some health insurance, food, and housing programs. Specifically, it would penalize green-card applicants for using Medicaid, a federal-state health plan for low-income people. (Penalties would not apply for using Medicaid in certain emergencies or for some Medicaid services provided through schools and disability programs.)
Using food stamps, Section 8 rental assistance, and federal housing vouchers also would count against applicants. Enrollment in a Medicare Part D program subsidy to help low-income people buy prescription drugs would work against them, too.
The proposal “is definitely a dramatic change from how public charge works today,” said Kelly Whitener, an associate professor at Georgetown University’s Center for Children and Families who specializes in pediatric health benefits and managed-care systems.
A leaked version of the rule from March suggested officials then were also considering penalizing those who receive subsidies to buy health insurance on the Affordable Care Act marketplaces. But that idea was not in the proposal. The marketplace subsidies are aimed at people at a generally higher income bracket than the beneficiaries targeted in the Trump plan, Whitener noted.
“They’re really homing in on low-income immigrants,” she added.
Nielsen said the proposed rule is “intended to promote immigrant self-sufficiency and protect finite resources.”
2. Is this as unprecedented as critics say?
Yes.
Public charge is an old idea. In the 1990s, lawmakers expanded it to consider explicitly whether people had received cash-based welfare.
But including programs like Medicaid and food stamps, which are much wider in scope, is a significant change. It would more likely hit working people – the majority of people on Medicaid are themselves employed, and almost 80% live in families with at least one working member, according to data compiled by the Kaiser Family Foundation. (Kaiser Health News is an editorially independent program of the foundation.)
Children who are American citizens but whose parents are immigrants could be more likely to suffer repercussions, said some experts. When parents opt out of public assistance for fear of their own legal status, their kids are less likely to be enrolled in programs such as the Children’s Health Insurance Program, or CHIP, for which they would qualify.
To be clear, receiving public aid wouldn’t necessarily stop people from getting a green card. But it would tilt the odds against them.
“Another piece is the enormous discretion the administration will have under its proposal in making judgments about who gets admitted to the country and who gets a green card,” said the Migration Policy Institute’s Greenberg.
3. When will the policy shift take effect?
This is an early step in the complex federal rule-making process. And a lot could still change.
Once the proposed rule appears in the Federal Register, a 60-day countdown starts, during which anyone can weigh in with comments.
A final rule likely wouldn’t take effect until 2019.
And DHS is still seeking input on some details. For instance, it hasn’t decided whether CHIP would be counted as one of the “public charge” eligible programs.
In the interim, people who had received public benefits before the rule took effect would not be penalized for doing so.
4. Already, though, the proposal is having effects
DHS estimates that 2.5% of eligible immigrants would drop out of public benefits programs because of this change – which would tally about $1.5 billion worth of federal money per year. But others expect a much larger impact.
“The chilling effects will be vastly greater than the individuals directly affected,” Greenberg said. “There’s considerable reason to believe that [the White House estimate] may be a significant understatement.”
In the proposed rule, DHS notes that the changes could result in “worse health outcomes,” “increased use of emergency rooms,” “increased prevalence of communicable diseases,” “increased rates of poverty,” and other concerns.
Given the complexity of these programs and the proposed rule – and the high stakes at play – low-income immigrants would be much more likely to avoid public benefits altogether, immigration experts said. Millions of immigrants are likely to be affected directly or indirectly, according to the Center for Law and Social Policy, a D.C.-based nonprofit organization.
That could have stark health implications.
Take free vaccines, for which children are often eligible and which would not be subject to the public charge rule. Families afraid of jeopardizing a green card could still be more likely to opt out of that service, Whitener said.
Already, she added, there are reports of people declining federal assistance – even though nothing has yet happened.
“The fear factor cannot be underestimated,” she said.
5. Will people sue?
Legal action is likely.
Officials such as California Attorney General Xavier Becerra, who has frequently clashed with the White House, are weighing challenges to the rule.
“The Trump Administration’s proposal punishes hard-working immigrant families – even targeting children who are citizens – for utilizing programs that provide basic nutrition and healthcare. This is an assault on our families and our communities,” Becerra said in a statement.
But these actions depend on the final shape of the regulation, which could change through the rule-making process.
“They are likely to receive a very large number of sharply critical comments, and there is no way to know what changes they might make as a result,” Greenberg said.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente. KHN’s coverage of children’s health care issues is supported in part by the Heising-Simons Foundation.
A proposed rule from the White House would make it harder for legal immigrants to get green cards if they have received certain kinds of public assistance – including Medicaid, food stamps, and housing subsidies. Green cards allow them to live and work permanently in the United States.
“Those seeking to immigrate to the United States must show they can support themselves financially,” Homeland Security Secretary Kirstjen Nielsen said in a statement.
The proposal, announced Sept. 22, marks a new frontier in the administration’s long-term effort to curb immigration, both legal and illegal. It already has spurred intense criticism from Democrats, anti-poverty activists, health care organizations, and immigrants’ rights advocates, who call its restrictions unprecedented.
“We are operating in an overall climate of tremendous fear and anxiety as a result of the administration’s overall approach to immigration enforcement and immigration policy,” said Mark Greenberg, a senior fellow at the Migration Policy Institute, which studies migration and refugee policies at local, national, and international levels. He is also a former Obama administration official.
But what effect would this proposal have?
It’s a complicated question, touching upon vast government programs, with billions of dollars at stake. While the implications aren’t all immediately clear, Kaiser Health News breaks down some of the key elements.
1. First thing first: What is the White House proposing?
The Trump administration wants to redefine a status known as “public charge” – a category used to determine whether someone seeking permanent resident status is “likely to become primarily dependent on the government for subsistence.”
In the past, people have been at risk of being defined a “public charge” if they took cash welfare – known as Temporary Assistance for Needy Families, or Supplemental Security Income – or federal help paying for long-term care. (Immigrants must be in the country legally for 5 years before being eligible for TANF or SSI.)
And that “public charge” designation could undermine their applications for permanent residence.
The new rule would expand the list to include some health insurance, food, and housing programs. Specifically, it would penalize green-card applicants for using Medicaid, a federal-state health plan for low-income people. (Penalties would not apply for using Medicaid in certain emergencies or for some Medicaid services provided through schools and disability programs.)
Using food stamps, Section 8 rental assistance, and federal housing vouchers also would count against applicants. Enrollment in a Medicare Part D program subsidy to help low-income people buy prescription drugs would work against them, too.
The proposal “is definitely a dramatic change from how public charge works today,” said Kelly Whitener, an associate professor at Georgetown University’s Center for Children and Families who specializes in pediatric health benefits and managed-care systems.
A leaked version of the rule from March suggested officials then were also considering penalizing those who receive subsidies to buy health insurance on the Affordable Care Act marketplaces. But that idea was not in the proposal. The marketplace subsidies are aimed at people at a generally higher income bracket than the beneficiaries targeted in the Trump plan, Whitener noted.
“They’re really homing in on low-income immigrants,” she added.
Nielsen said the proposed rule is “intended to promote immigrant self-sufficiency and protect finite resources.”
2. Is this as unprecedented as critics say?
Yes.
Public charge is an old idea. In the 1990s, lawmakers expanded it to consider explicitly whether people had received cash-based welfare.
But including programs like Medicaid and food stamps, which are much wider in scope, is a significant change. It would more likely hit working people – the majority of people on Medicaid are themselves employed, and almost 80% live in families with at least one working member, according to data compiled by the Kaiser Family Foundation. (Kaiser Health News is an editorially independent program of the foundation.)
Children who are American citizens but whose parents are immigrants could be more likely to suffer repercussions, said some experts. When parents opt out of public assistance for fear of their own legal status, their kids are less likely to be enrolled in programs such as the Children’s Health Insurance Program, or CHIP, for which they would qualify.
To be clear, receiving public aid wouldn’t necessarily stop people from getting a green card. But it would tilt the odds against them.
“Another piece is the enormous discretion the administration will have under its proposal in making judgments about who gets admitted to the country and who gets a green card,” said the Migration Policy Institute’s Greenberg.
3. When will the policy shift take effect?
This is an early step in the complex federal rule-making process. And a lot could still change.
Once the proposed rule appears in the Federal Register, a 60-day countdown starts, during which anyone can weigh in with comments.
A final rule likely wouldn’t take effect until 2019.
And DHS is still seeking input on some details. For instance, it hasn’t decided whether CHIP would be counted as one of the “public charge” eligible programs.
In the interim, people who had received public benefits before the rule took effect would not be penalized for doing so.
4. Already, though, the proposal is having effects
DHS estimates that 2.5% of eligible immigrants would drop out of public benefits programs because of this change – which would tally about $1.5 billion worth of federal money per year. But others expect a much larger impact.
“The chilling effects will be vastly greater than the individuals directly affected,” Greenberg said. “There’s considerable reason to believe that [the White House estimate] may be a significant understatement.”
In the proposed rule, DHS notes that the changes could result in “worse health outcomes,” “increased use of emergency rooms,” “increased prevalence of communicable diseases,” “increased rates of poverty,” and other concerns.
Given the complexity of these programs and the proposed rule – and the high stakes at play – low-income immigrants would be much more likely to avoid public benefits altogether, immigration experts said. Millions of immigrants are likely to be affected directly or indirectly, according to the Center for Law and Social Policy, a D.C.-based nonprofit organization.
That could have stark health implications.
Take free vaccines, for which children are often eligible and which would not be subject to the public charge rule. Families afraid of jeopardizing a green card could still be more likely to opt out of that service, Whitener said.
Already, she added, there are reports of people declining federal assistance – even though nothing has yet happened.
“The fear factor cannot be underestimated,” she said.
5. Will people sue?
Legal action is likely.
Officials such as California Attorney General Xavier Becerra, who has frequently clashed with the White House, are weighing challenges to the rule.
“The Trump Administration’s proposal punishes hard-working immigrant families – even targeting children who are citizens – for utilizing programs that provide basic nutrition and healthcare. This is an assault on our families and our communities,” Becerra said in a statement.
But these actions depend on the final shape of the regulation, which could change through the rule-making process.
“They are likely to receive a very large number of sharply critical comments, and there is no way to know what changes they might make as a result,” Greenberg said.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente. KHN’s coverage of children’s health care issues is supported in part by the Heising-Simons Foundation.
Mutation in patients with chronic HCV raises risk of liver cancer
An adenine mutation in the MUTYH base repair excision gene increased the risk of developing hepatocellular carcinoma (HCC) in patients with chronic hepatitis C virus (HCV). The mutation was discovered in an analysis of 19 tagging single-nucleotide polymorphism (SNP) variants examined for multiple base repair excision genes (MUTYH, OGG1, and MTH1), according to the results of a retrospective analysis of patient genotypes reported in Free Radical Biology and Medicine (2018;129:88-96).
In addition, the researchers examined MUTYH-null mice to assess the involvement of oxidative stress and DNA repair enzymes in hepatocarcinogenesis.
One significant SNP was found and confirmed from 93 Japanese patients with chronic HCV (38 with HCC and 55 controls), according to Akira Sakurada, MD, of Sapporo (Japan) Medical University, and his colleagues.
Patients diagnosed as having chronic HCV between 2007 and 2015 were enrolled. All patients were 45 years of age or older with detectable anti-HCV antibodies and HCV-RNA, as well as histopathological evidence of chronic hepatitis on liver biopsy. An exploratory cohort of 40 patients was first used before the 93 patient confirmatory analysis.
Genomic DNA was extracted from peripheral mononuclear cells of all of the patients studied.
Dr. Sakurada and his colleagues found significant associations for the single intron SNP (rs3219487) in the MUTYH gene. Human MutY homolog (MUTYH) protein is responsible for recognition and removal of an inappropriately inserted adenine (A).
The risk of developing HCC in patients with A/A or G/A genotypes was higher than in those with the G/G genotype (odds ratio = 9.27, 95% confidence interval = 2.39-32.1, P = .0005). In addition, they found that MUTYH mRNA levels were significantly lower in G/A or A/A genotyped subjects as compared with the wild type (G/G) (P = .0157 and P = .0108, respectively).
In their mouse model, the investigators found that liver tumors developed in MUTYH-null mice. As a physiological confirmation of their results, the researchers investigated a MUTYH-null mouse model. They found that decreased MUTYH activity in the null mice resulted in the development of HCC in these animals after 12 months of a high iron diet, but no tumors were observed when a dietary antioxidant (N-Acetyl-L-cysteine) was also provided.
“HCC may develop even in patients with an SVR [sustained virologic response]. This may be a problem especially for the numerous elderly patients. Our study suggests that SNP genotyping may predict the risk of developing HCC in such patients. We propose that in the setting of appropriate screening intervals, stratification of patients for whom antioxidant treatment would be appropriate could be achieved in this manner,” the researchers concluded.
The study was supported by grants from the Japan Society for Scientific Research. The authors reported they had nothing to disclose.
SOURCE: Sakurada A et al. Free Radic Biol Med. 2018;129:88-96.
An adenine mutation in the MUTYH base repair excision gene increased the risk of developing hepatocellular carcinoma (HCC) in patients with chronic hepatitis C virus (HCV). The mutation was discovered in an analysis of 19 tagging single-nucleotide polymorphism (SNP) variants examined for multiple base repair excision genes (MUTYH, OGG1, and MTH1), according to the results of a retrospective analysis of patient genotypes reported in Free Radical Biology and Medicine (2018;129:88-96).
In addition, the researchers examined MUTYH-null mice to assess the involvement of oxidative stress and DNA repair enzymes in hepatocarcinogenesis.
One significant SNP was found and confirmed from 93 Japanese patients with chronic HCV (38 with HCC and 55 controls), according to Akira Sakurada, MD, of Sapporo (Japan) Medical University, and his colleagues.
Patients diagnosed as having chronic HCV between 2007 and 2015 were enrolled. All patients were 45 years of age or older with detectable anti-HCV antibodies and HCV-RNA, as well as histopathological evidence of chronic hepatitis on liver biopsy. An exploratory cohort of 40 patients was first used before the 93 patient confirmatory analysis.
Genomic DNA was extracted from peripheral mononuclear cells of all of the patients studied.
Dr. Sakurada and his colleagues found significant associations for the single intron SNP (rs3219487) in the MUTYH gene. Human MutY homolog (MUTYH) protein is responsible for recognition and removal of an inappropriately inserted adenine (A).
The risk of developing HCC in patients with A/A or G/A genotypes was higher than in those with the G/G genotype (odds ratio = 9.27, 95% confidence interval = 2.39-32.1, P = .0005). In addition, they found that MUTYH mRNA levels were significantly lower in G/A or A/A genotyped subjects as compared with the wild type (G/G) (P = .0157 and P = .0108, respectively).
In their mouse model, the investigators found that liver tumors developed in MUTYH-null mice. As a physiological confirmation of their results, the researchers investigated a MUTYH-null mouse model. They found that decreased MUTYH activity in the null mice resulted in the development of HCC in these animals after 12 months of a high iron diet, but no tumors were observed when a dietary antioxidant (N-Acetyl-L-cysteine) was also provided.
“HCC may develop even in patients with an SVR [sustained virologic response]. This may be a problem especially for the numerous elderly patients. Our study suggests that SNP genotyping may predict the risk of developing HCC in such patients. We propose that in the setting of appropriate screening intervals, stratification of patients for whom antioxidant treatment would be appropriate could be achieved in this manner,” the researchers concluded.
The study was supported by grants from the Japan Society for Scientific Research. The authors reported they had nothing to disclose.
SOURCE: Sakurada A et al. Free Radic Biol Med. 2018;129:88-96.
An adenine mutation in the MUTYH base repair excision gene increased the risk of developing hepatocellular carcinoma (HCC) in patients with chronic hepatitis C virus (HCV). The mutation was discovered in an analysis of 19 tagging single-nucleotide polymorphism (SNP) variants examined for multiple base repair excision genes (MUTYH, OGG1, and MTH1), according to the results of a retrospective analysis of patient genotypes reported in Free Radical Biology and Medicine (2018;129:88-96).
In addition, the researchers examined MUTYH-null mice to assess the involvement of oxidative stress and DNA repair enzymes in hepatocarcinogenesis.
One significant SNP was found and confirmed from 93 Japanese patients with chronic HCV (38 with HCC and 55 controls), according to Akira Sakurada, MD, of Sapporo (Japan) Medical University, and his colleagues.
Patients diagnosed as having chronic HCV between 2007 and 2015 were enrolled. All patients were 45 years of age or older with detectable anti-HCV antibodies and HCV-RNA, as well as histopathological evidence of chronic hepatitis on liver biopsy. An exploratory cohort of 40 patients was first used before the 93 patient confirmatory analysis.
Genomic DNA was extracted from peripheral mononuclear cells of all of the patients studied.
Dr. Sakurada and his colleagues found significant associations for the single intron SNP (rs3219487) in the MUTYH gene. Human MutY homolog (MUTYH) protein is responsible for recognition and removal of an inappropriately inserted adenine (A).
The risk of developing HCC in patients with A/A or G/A genotypes was higher than in those with the G/G genotype (odds ratio = 9.27, 95% confidence interval = 2.39-32.1, P = .0005). In addition, they found that MUTYH mRNA levels were significantly lower in G/A or A/A genotyped subjects as compared with the wild type (G/G) (P = .0157 and P = .0108, respectively).
In their mouse model, the investigators found that liver tumors developed in MUTYH-null mice. As a physiological confirmation of their results, the researchers investigated a MUTYH-null mouse model. They found that decreased MUTYH activity in the null mice resulted in the development of HCC in these animals after 12 months of a high iron diet, but no tumors were observed when a dietary antioxidant (N-Acetyl-L-cysteine) was also provided.
“HCC may develop even in patients with an SVR [sustained virologic response]. This may be a problem especially for the numerous elderly patients. Our study suggests that SNP genotyping may predict the risk of developing HCC in such patients. We propose that in the setting of appropriate screening intervals, stratification of patients for whom antioxidant treatment would be appropriate could be achieved in this manner,” the researchers concluded.
The study was supported by grants from the Japan Society for Scientific Research. The authors reported they had nothing to disclose.
SOURCE: Sakurada A et al. Free Radic Biol Med. 2018;129:88-96.
FROM FREE RADICAL BIOLOGY AND MEDICINE
Key clinical point: An adenine mutation in the MUTYH gene increased the risk of developing liver cancer in patients with chronic HCV.
Major finding: Hepatocellular carcinoma risk with the mutation was significantly higher than in patients with the standard genotype (OR = 9.27, P = .0005).
Study details: Nineteen SNPs in base excision repair genes were examined in 93 patients with chronic hepatitis C.
Disclosures: The study was supported by grants from the Japan Society for Scientific Research. The authors reported they had nothing to disclose.
Source: Sakurada A et al. Free Radic Biol Med. 2018;129:88-96.
FDA approves duvelisib for CLL/SLL and FL
The U.S. Food and Drug Administration (FDA) has approved duvelisib (Copiktra™), a dual PI3K delta/gamma inhibitor, for two indications.
Duvelisib has full FDA approval to treat adults with relapsed or refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) who have received at least two prior therapies.
Duvelisib also has accelerated approval to treat adults with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.
Accelerated approval is based on a surrogate or intermediate endpoint—in this case, overall response rate—that is reasonably likely to predict clinical benefit. Continued approval of duvelisib in FL may be contingent upon results of confirmatory trials verifying that the drug provides a clinical benefit.
Duvelisib will be available in the U.S. immediately, according to Verastem Inc., the company marketing the drug.
Verastem said it will help patients access duvelisib through the Verastem Cares™ program, which is designed to provide information and assistance to patients who are prescribed duvelisib.
The prescribing information for duvelisib includes a boxed warning detailing four fatal and/or serious toxicities associated with the drug—infections, diarrhea or colitis, cutaneous reactions, and pneumonitis.
Verastem said it is implementing an informational risk evaluation and mitigation strategy to provide appropriate dosing and safety information for duvelisib.
The recommended dose of duvelisib is 25 mg orally twice daily, taken continuously in 28-day treatment cycles.
The FDA assessed the new drug application for duvelisib under priority review. The FDA also granted duvelisib fast track designation in CLL and FL as well as orphan drug designation for CLL/SLL and FL.
The FDA’s approval of duvelisib is supported by data from the phase 3 DUO trial and the phase 2 DYNAMO trial. Updated results from both studies are available in the prescribing information for duvelisib.
DUO trial
DUO included 319 patients with CLL (n=312) or SLL (n=7) who had received at least one prior therapy. They were randomized to receive either duvelisib (25 mg orally twice daily) or ofatumumab (initial infusion of 300 mg followed by 7 weekly infusions and 4 monthly infusions of 2000 mg).
The following efficacy results were observed in patients who had received at least two prior therapies, which includes 95 patients in the duvelisib arm and 101 in the ofatumumab arm.
The overall response rate was 78% in the duvelisib arm and 39% in the ofatumumab arm. All responses in both arms were partial responses.
The median progression-free survival was 16.4 months with duvelisib and 9.1 months with ofatumumab (hazard ratio=0.40).
The safety results include all patients treated with duvelisib or ofatumumab.
Twelve percent of patients in the duvelisib arm had fatal adverse events (AEs) within 30 days of the last dose. The same was true of 4% of patients treated with ofatumumab.
Serious AEs occurred in 73% of patients treated with duvelisib. The most common were infection (38%) and diarrhea/colitis (23%).
Thirty-six percent of patients discontinued duvelisib. Most discontinuations were due to diarrhea/ colitis, infection, and rash. Twenty-nine percent of patients in the duvelisib arm required dose reductions, most often due to diarrhea/colitis and rash.
DYNAMO trial
DYNAMO enrolled patients with indolent non-Hodgkin lymphoma whose disease was refractory to both rituximab and chemotherapy or radioimmunotherapy.
There were 83 patients with FL. They had a median of 3 prior anticancer regimens (range, 1-10).
The patients received duvelisib at 25 mg orally twice daily until disease progression or unacceptable toxicity.
The overall response rate was 42%. One patient achieved a complete response, and 34 had a partial response.
Forty-three percent of responders maintained their response at 6 months, and 17% maintained their response at 12 months.
Serious AEs occurred in 58% of FL patients. The most common were diarrhea/colitis, pneumonia, renal insufficiency, rash, and sepsis.
AEs occurring in at least 20% of FL patients included diarrhea/colitis, nausea, fatigue, musculoskeletal pain, rash, neutropenia, cough, anemia, pyrexia, headache, mucositis, abdominal pain, vomiting, transaminase elevation, and thrombocytopenia.
Twenty-nine percent of FL patients discontinued duvelisib, and 23% had dose reductions. Most discontinuations were due to diarrhea/colitis and rash, and most dose reductions were due to transaminase elevation, diarrhea/colitis, lipase increase, and infection.
The U.S. Food and Drug Administration (FDA) has approved duvelisib (Copiktra™), a dual PI3K delta/gamma inhibitor, for two indications.
Duvelisib has full FDA approval to treat adults with relapsed or refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) who have received at least two prior therapies.
Duvelisib also has accelerated approval to treat adults with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.
Accelerated approval is based on a surrogate or intermediate endpoint—in this case, overall response rate—that is reasonably likely to predict clinical benefit. Continued approval of duvelisib in FL may be contingent upon results of confirmatory trials verifying that the drug provides a clinical benefit.
Duvelisib will be available in the U.S. immediately, according to Verastem Inc., the company marketing the drug.
Verastem said it will help patients access duvelisib through the Verastem Cares™ program, which is designed to provide information and assistance to patients who are prescribed duvelisib.
The prescribing information for duvelisib includes a boxed warning detailing four fatal and/or serious toxicities associated with the drug—infections, diarrhea or colitis, cutaneous reactions, and pneumonitis.
Verastem said it is implementing an informational risk evaluation and mitigation strategy to provide appropriate dosing and safety information for duvelisib.
The recommended dose of duvelisib is 25 mg orally twice daily, taken continuously in 28-day treatment cycles.
The FDA assessed the new drug application for duvelisib under priority review. The FDA also granted duvelisib fast track designation in CLL and FL as well as orphan drug designation for CLL/SLL and FL.
The FDA’s approval of duvelisib is supported by data from the phase 3 DUO trial and the phase 2 DYNAMO trial. Updated results from both studies are available in the prescribing information for duvelisib.
DUO trial
DUO included 319 patients with CLL (n=312) or SLL (n=7) who had received at least one prior therapy. They were randomized to receive either duvelisib (25 mg orally twice daily) or ofatumumab (initial infusion of 300 mg followed by 7 weekly infusions and 4 monthly infusions of 2000 mg).
The following efficacy results were observed in patients who had received at least two prior therapies, which includes 95 patients in the duvelisib arm and 101 in the ofatumumab arm.
The overall response rate was 78% in the duvelisib arm and 39% in the ofatumumab arm. All responses in both arms were partial responses.
The median progression-free survival was 16.4 months with duvelisib and 9.1 months with ofatumumab (hazard ratio=0.40).
The safety results include all patients treated with duvelisib or ofatumumab.
Twelve percent of patients in the duvelisib arm had fatal adverse events (AEs) within 30 days of the last dose. The same was true of 4% of patients treated with ofatumumab.
Serious AEs occurred in 73% of patients treated with duvelisib. The most common were infection (38%) and diarrhea/colitis (23%).
Thirty-six percent of patients discontinued duvelisib. Most discontinuations were due to diarrhea/ colitis, infection, and rash. Twenty-nine percent of patients in the duvelisib arm required dose reductions, most often due to diarrhea/colitis and rash.
DYNAMO trial
DYNAMO enrolled patients with indolent non-Hodgkin lymphoma whose disease was refractory to both rituximab and chemotherapy or radioimmunotherapy.
There were 83 patients with FL. They had a median of 3 prior anticancer regimens (range, 1-10).
The patients received duvelisib at 25 mg orally twice daily until disease progression or unacceptable toxicity.
The overall response rate was 42%. One patient achieved a complete response, and 34 had a partial response.
Forty-three percent of responders maintained their response at 6 months, and 17% maintained their response at 12 months.
Serious AEs occurred in 58% of FL patients. The most common were diarrhea/colitis, pneumonia, renal insufficiency, rash, and sepsis.
AEs occurring in at least 20% of FL patients included diarrhea/colitis, nausea, fatigue, musculoskeletal pain, rash, neutropenia, cough, anemia, pyrexia, headache, mucositis, abdominal pain, vomiting, transaminase elevation, and thrombocytopenia.
Twenty-nine percent of FL patients discontinued duvelisib, and 23% had dose reductions. Most discontinuations were due to diarrhea/colitis and rash, and most dose reductions were due to transaminase elevation, diarrhea/colitis, lipase increase, and infection.
The U.S. Food and Drug Administration (FDA) has approved duvelisib (Copiktra™), a dual PI3K delta/gamma inhibitor, for two indications.
Duvelisib has full FDA approval to treat adults with relapsed or refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) who have received at least two prior therapies.
Duvelisib also has accelerated approval to treat adults with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.
Accelerated approval is based on a surrogate or intermediate endpoint—in this case, overall response rate—that is reasonably likely to predict clinical benefit. Continued approval of duvelisib in FL may be contingent upon results of confirmatory trials verifying that the drug provides a clinical benefit.
Duvelisib will be available in the U.S. immediately, according to Verastem Inc., the company marketing the drug.
Verastem said it will help patients access duvelisib through the Verastem Cares™ program, which is designed to provide information and assistance to patients who are prescribed duvelisib.
The prescribing information for duvelisib includes a boxed warning detailing four fatal and/or serious toxicities associated with the drug—infections, diarrhea or colitis, cutaneous reactions, and pneumonitis.
Verastem said it is implementing an informational risk evaluation and mitigation strategy to provide appropriate dosing and safety information for duvelisib.
The recommended dose of duvelisib is 25 mg orally twice daily, taken continuously in 28-day treatment cycles.
The FDA assessed the new drug application for duvelisib under priority review. The FDA also granted duvelisib fast track designation in CLL and FL as well as orphan drug designation for CLL/SLL and FL.
The FDA’s approval of duvelisib is supported by data from the phase 3 DUO trial and the phase 2 DYNAMO trial. Updated results from both studies are available in the prescribing information for duvelisib.
DUO trial
DUO included 319 patients with CLL (n=312) or SLL (n=7) who had received at least one prior therapy. They were randomized to receive either duvelisib (25 mg orally twice daily) or ofatumumab (initial infusion of 300 mg followed by 7 weekly infusions and 4 monthly infusions of 2000 mg).
The following efficacy results were observed in patients who had received at least two prior therapies, which includes 95 patients in the duvelisib arm and 101 in the ofatumumab arm.
The overall response rate was 78% in the duvelisib arm and 39% in the ofatumumab arm. All responses in both arms were partial responses.
The median progression-free survival was 16.4 months with duvelisib and 9.1 months with ofatumumab (hazard ratio=0.40).
The safety results include all patients treated with duvelisib or ofatumumab.
Twelve percent of patients in the duvelisib arm had fatal adverse events (AEs) within 30 days of the last dose. The same was true of 4% of patients treated with ofatumumab.
Serious AEs occurred in 73% of patients treated with duvelisib. The most common were infection (38%) and diarrhea/colitis (23%).
Thirty-six percent of patients discontinued duvelisib. Most discontinuations were due to diarrhea/ colitis, infection, and rash. Twenty-nine percent of patients in the duvelisib arm required dose reductions, most often due to diarrhea/colitis and rash.
DYNAMO trial
DYNAMO enrolled patients with indolent non-Hodgkin lymphoma whose disease was refractory to both rituximab and chemotherapy or radioimmunotherapy.
There were 83 patients with FL. They had a median of 3 prior anticancer regimens (range, 1-10).
The patients received duvelisib at 25 mg orally twice daily until disease progression or unacceptable toxicity.
The overall response rate was 42%. One patient achieved a complete response, and 34 had a partial response.
Forty-three percent of responders maintained their response at 6 months, and 17% maintained their response at 12 months.
Serious AEs occurred in 58% of FL patients. The most common were diarrhea/colitis, pneumonia, renal insufficiency, rash, and sepsis.
AEs occurring in at least 20% of FL patients included diarrhea/colitis, nausea, fatigue, musculoskeletal pain, rash, neutropenia, cough, anemia, pyrexia, headache, mucositis, abdominal pain, vomiting, transaminase elevation, and thrombocytopenia.
Twenty-nine percent of FL patients discontinued duvelisib, and 23% had dose reductions. Most discontinuations were due to diarrhea/colitis and rash, and most dose reductions were due to transaminase elevation, diarrhea/colitis, lipase increase, and infection.
Five-year follow-up confirms safety of antibiotics for uncomplicated appendicitis
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
“When the earlier results of the APPAC trial were published, showing that 73% of patients with uncomplicated acute appendicitis did not require surgery at 1 year of follow-up, critics raised concerns that many more of these patients would eventually require surgery,” wrote Edward H. Livingston, MD, deputy editor of JAMA in an accompanying editorial (JAMA 2018;320:1245-46).Surgeons have since been waiting until longer-term outcomes were known. These 5-year results are supportive of the antibiotics approach. “They show no increase in major complications in patients who experienced a recurrence and underwent appendectomy after initially being randomized to antibiotic therapy. They challenge the notion that uncomplicated acute appendicitis is a surgical emergency and show that nonsurgical treatment is a reasonable option,” Dr Livingston wrote.
He declared no conflicts of interest.
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
Longer-term outcomes of treating uncomplicated acute appendicitis with antibiotics suggest it is a feasible alternative to appendectomy.
Researchers have presented the 5-year follow-up data from the Appendicitis Acuta (APPAC) multicenter randomized clinical trial comparing appendectomy with antibiotic therapy in 530 patients.
They found that 39.1% (100) of the 257 patients randomized to antibiotic therapy – 3 days of intravenous ertapenem followed by 7 days of oral levofloxacin and metronidazole – experienced a recurrence of appendicitis within 5 years and subsequently had surgery.
However the authors noted that seven of these patients were later found not to have appendicitis, so the true success rate for antibiotic treatment was actually 63.7%.
Seventy of the patients who experienced a recurrence underwent surgery in the first year after randomization, 17 in the second year, 3 in the third year, 5 in the fourth year, and the remaining 5 patients in the fifth year, the authors wrote in an article published in JAMA.
However the overall complication rate was similar in patients who were randomized to undergo appendectomy and in those who were initially randomized to the antibiotic group but later experienced a recurrence and underwent surgery.
“No patient initially treated with antibiotics, who ultimately developed recurrent appendicitis, had any complications related to the delay in surgery,” wrote Paulina Salminen, MD, from Turku (Finland) University Hospital and coauthors. “Nearly two-thirds of all patients who initially presented with uncomplicated appendicitis were successfully treated with antibiotics alone, and those who ultimately developed recurrent disease did not experience any adverse outcomes related to the delay in appendectomy.”
Of the 100 patients randomized to antibiotics who underwent appendectomy after a recurrence, 15 were operated on when they were first hospitalized at study admission.
The authors commented that the study design allowed for surgeons to exercise their clinical judgment in choosing when to perform an appendectomy on patients in the antibiotic group, because antibiotics alone was not considered acceptable treatment for appendicitis.
“This led to some patients undergoing appendectomy who did not have appendicitis or who might have been successfully treated with antibiotics or an another course of antibiotics,” they wrote. “Future studies should investigate protocols for further imaging or antibiotic treatment for patients who develop recurrent appendicitis after they were initially treated with antibiotics.”
In the recurrence group, the majority were found to have uncomplicated appendicitis, but complicated appendicitis was seen in two patients between 2 and 5 years after the index admission.
There was a significant 17.9% higher complication rate in the appendectomy group, compared with the antibiotic group – 24.4% versus 6.5% – at 5 years and two patients in the appendectomy group had severe complications requiring reoperation.
They suggested that the higher complication rate with surgery, which was mostly attributable to infections, could be reduced by the use of laparoscopic appendectomy, which is also associated with faster recovery.
The median length of hospital stay was 3 days for both the appendectomy group and the antibiotics-only group, but patients randomized to appendectomy took a median of 22 days of sick leave, compared with 11 days for those randomized to antibiotics (P less than .001).
In the absence of standard protocol on treating appendicitis with antibiotics, the authors noted that they took a conservative approach, using broad-spectrum antibiotics and keeping patients in hospital for 3 days for observation.
“The success of antibiotic treatment for appendicitis calls into question prior beliefs that appendicitis inevitably results in serious intra-abdominal infection if appendectomy is not performed.”
The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
SOURCE: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
FROM JAMA
Key clinical point: Antibiotics could be used as an alternative to surgery for uncomplicated acute appendicitis.
Major finding: Around two-thirds of patients with acute appendicitis treated with antibiotics only did not experience a recurrence in 5 years.
Study details: Randomized controlled study in 530 patients with uncomplicated acute appendicitis.
Disclosures: The study was supported by the Mary and Georg C. Ehrnrooth Foundation, the EVO Foundation, and Turku University. One author declared lecture fees from three pharmaceutical companies but no other conflicts of interest were declared.
Source: Salminen P et al. JAMA 2018;320:1259-65. doi: 10.1001/jama.2018.13201.
Digital CBT improves health, well-being, quality of life in randomized trial
Digitally delivered cognitive-behavioral therapy not only improved insomnia, but also provided “around-the-clock” health, psychological, and quality-of-life improvements, according to investigators.
Compared with control subjects, patients who used a cognitive-behavioral therapy (CBT) program and associated iPhone app had small improvements in functional health and psychological well-being and large improvements in sleep-related quality of life, the investigators reported.
Those changes were mediated by a large improvement in insomnia, according to Colin A. Espie, PhD, professor of sleep medicine at the University of Oxford (England), and his colleagues.
“These findings indicate that digital CBT improves both daytime and nighttime aspects of insomnia, lending further weight to the clinical guideline recommendation of CBT as the treatment of choice for insomnia,” Dr. Espie and his colleagues reported in JAMA Psychiatry.
Their study included 1,711 adults with symptoms of insomnia that were self-reported and a score of 16 or less on the Sleep Condition Indicator (SCI), which has a range of 0-32. A total of 853 were randomized to receive digital CBT, of whom 413 completed six scheduled 20-minute sessions; an additional 276 adults completed at least one session. The control arm included 858 individuals randomized to sleep hygiene education, of whom 759 went on to receive that intervention.
Small but significant improvements were seen in self-reported measures of functional health, Dr. Espie and his colleagues reported. The adjusted differences in mean scores on the Patient-Reported Outcomes Measurement Information System: Global Health Scale were 0.90 in a midtreatment evaluation at 4 weeks after baseline, 1.76 in a posttreatment evaluation at 8 weeks, and 1.76 at a 24-week follow-up assessment (P less than 0.001 for all comparisons).
Likewise, adjusted differences in the Warwick-Edinburgh Mental Wellbeing Scale were 1.04, 2.68, and 2.95 at 4, 8, and 24 weeks, respectively.
Adjusted differences in the Glasgow Sleep Impact Index, which measures sleep-related quality of life, were –8.76, –17.60, and –18.72 at 4, 8, and 24 weeks, respectively, indicating a benefit of the digital CBT intervention over the control intervention, the investigators said.
About 45%-84% of these effects were attributable to changes in insomnia symptoms, results of a mediation analysis showed.
Insomnia scores as measured by the eight-item SCI scale were 6.5 and 6.6 at baseline for the digital CBT and control groups, respectively. By week 4, SCI scores were 9.96 and 13.00 for the two groups (P less than 0.001), with significant differences also reported at the 8- and 24-week evaluations.
The investigators noted that their findings might not be generalizable because participants were not drawn from patient populations. In addition, although 58% of the participants completed 4 weeks or more of the digital CBT sessions, the dropout rate was “substantial.”
Nevertheless, they wrote, these findings, together with results of a recently reported parallel study looking at the effects of sleep on mental health, affirm CBT as the treatment of choice for insomnia. “The mediation analyses in these two studies, with a total of 5,466 participants, provide novel and convincing evidence that insomnia may be a legitimate and important target for mental health and well-being.”
Dr. Espie reported that he is cofounder and chief medical officer of Big Health. He is a shareholder and receives a salary from the company, which was involved in the design and conduct of the study, interpretation of data, and development of the manuscript appearing in JAMA Psychiatry. The study was funded by Big Health and other sources, including the National Institute for Health Research Oxford Biomedical Research Centre.
SOURCE: Espie CA et al. JAMA Psychiatry. 2018 Sep 25. doi: 10.1001/jamapsychiatry.2018.2745.
Digitally delivered cognitive-behavioral therapy not only improved insomnia, but also provided “around-the-clock” health, psychological, and quality-of-life improvements, according to investigators.
Compared with control subjects, patients who used a cognitive-behavioral therapy (CBT) program and associated iPhone app had small improvements in functional health and psychological well-being and large improvements in sleep-related quality of life, the investigators reported.
Those changes were mediated by a large improvement in insomnia, according to Colin A. Espie, PhD, professor of sleep medicine at the University of Oxford (England), and his colleagues.
“These findings indicate that digital CBT improves both daytime and nighttime aspects of insomnia, lending further weight to the clinical guideline recommendation of CBT as the treatment of choice for insomnia,” Dr. Espie and his colleagues reported in JAMA Psychiatry.
Their study included 1,711 adults with symptoms of insomnia that were self-reported and a score of 16 or less on the Sleep Condition Indicator (SCI), which has a range of 0-32. A total of 853 were randomized to receive digital CBT, of whom 413 completed six scheduled 20-minute sessions; an additional 276 adults completed at least one session. The control arm included 858 individuals randomized to sleep hygiene education, of whom 759 went on to receive that intervention.
Small but significant improvements were seen in self-reported measures of functional health, Dr. Espie and his colleagues reported. The adjusted differences in mean scores on the Patient-Reported Outcomes Measurement Information System: Global Health Scale were 0.90 in a midtreatment evaluation at 4 weeks after baseline, 1.76 in a posttreatment evaluation at 8 weeks, and 1.76 at a 24-week follow-up assessment (P less than 0.001 for all comparisons).
Likewise, adjusted differences in the Warwick-Edinburgh Mental Wellbeing Scale were 1.04, 2.68, and 2.95 at 4, 8, and 24 weeks, respectively.
Adjusted differences in the Glasgow Sleep Impact Index, which measures sleep-related quality of life, were –8.76, –17.60, and –18.72 at 4, 8, and 24 weeks, respectively, indicating a benefit of the digital CBT intervention over the control intervention, the investigators said.
About 45%-84% of these effects were attributable to changes in insomnia symptoms, results of a mediation analysis showed.
Insomnia scores as measured by the eight-item SCI scale were 6.5 and 6.6 at baseline for the digital CBT and control groups, respectively. By week 4, SCI scores were 9.96 and 13.00 for the two groups (P less than 0.001), with significant differences also reported at the 8- and 24-week evaluations.
The investigators noted that their findings might not be generalizable because participants were not drawn from patient populations. In addition, although 58% of the participants completed 4 weeks or more of the digital CBT sessions, the dropout rate was “substantial.”
Nevertheless, they wrote, these findings, together with results of a recently reported parallel study looking at the effects of sleep on mental health, affirm CBT as the treatment of choice for insomnia. “The mediation analyses in these two studies, with a total of 5,466 participants, provide novel and convincing evidence that insomnia may be a legitimate and important target for mental health and well-being.”
Dr. Espie reported that he is cofounder and chief medical officer of Big Health. He is a shareholder and receives a salary from the company, which was involved in the design and conduct of the study, interpretation of data, and development of the manuscript appearing in JAMA Psychiatry. The study was funded by Big Health and other sources, including the National Institute for Health Research Oxford Biomedical Research Centre.
SOURCE: Espie CA et al. JAMA Psychiatry. 2018 Sep 25. doi: 10.1001/jamapsychiatry.2018.2745.
Digitally delivered cognitive-behavioral therapy not only improved insomnia, but also provided “around-the-clock” health, psychological, and quality-of-life improvements, according to investigators.
Compared with control subjects, patients who used a cognitive-behavioral therapy (CBT) program and associated iPhone app had small improvements in functional health and psychological well-being and large improvements in sleep-related quality of life, the investigators reported.
Those changes were mediated by a large improvement in insomnia, according to Colin A. Espie, PhD, professor of sleep medicine at the University of Oxford (England), and his colleagues.
“These findings indicate that digital CBT improves both daytime and nighttime aspects of insomnia, lending further weight to the clinical guideline recommendation of CBT as the treatment of choice for insomnia,” Dr. Espie and his colleagues reported in JAMA Psychiatry.
Their study included 1,711 adults with symptoms of insomnia that were self-reported and a score of 16 or less on the Sleep Condition Indicator (SCI), which has a range of 0-32. A total of 853 were randomized to receive digital CBT, of whom 413 completed six scheduled 20-minute sessions; an additional 276 adults completed at least one session. The control arm included 858 individuals randomized to sleep hygiene education, of whom 759 went on to receive that intervention.
Small but significant improvements were seen in self-reported measures of functional health, Dr. Espie and his colleagues reported. The adjusted differences in mean scores on the Patient-Reported Outcomes Measurement Information System: Global Health Scale were 0.90 in a midtreatment evaluation at 4 weeks after baseline, 1.76 in a posttreatment evaluation at 8 weeks, and 1.76 at a 24-week follow-up assessment (P less than 0.001 for all comparisons).
Likewise, adjusted differences in the Warwick-Edinburgh Mental Wellbeing Scale were 1.04, 2.68, and 2.95 at 4, 8, and 24 weeks, respectively.
Adjusted differences in the Glasgow Sleep Impact Index, which measures sleep-related quality of life, were –8.76, –17.60, and –18.72 at 4, 8, and 24 weeks, respectively, indicating a benefit of the digital CBT intervention over the control intervention, the investigators said.
About 45%-84% of these effects were attributable to changes in insomnia symptoms, results of a mediation analysis showed.
Insomnia scores as measured by the eight-item SCI scale were 6.5 and 6.6 at baseline for the digital CBT and control groups, respectively. By week 4, SCI scores were 9.96 and 13.00 for the two groups (P less than 0.001), with significant differences also reported at the 8- and 24-week evaluations.
The investigators noted that their findings might not be generalizable because participants were not drawn from patient populations. In addition, although 58% of the participants completed 4 weeks or more of the digital CBT sessions, the dropout rate was “substantial.”
Nevertheless, they wrote, these findings, together with results of a recently reported parallel study looking at the effects of sleep on mental health, affirm CBT as the treatment of choice for insomnia. “The mediation analyses in these two studies, with a total of 5,466 participants, provide novel and convincing evidence that insomnia may be a legitimate and important target for mental health and well-being.”
Dr. Espie reported that he is cofounder and chief medical officer of Big Health. He is a shareholder and receives a salary from the company, which was involved in the design and conduct of the study, interpretation of data, and development of the manuscript appearing in JAMA Psychiatry. The study was funded by Big Health and other sources, including the National Institute for Health Research Oxford Biomedical Research Centre.
SOURCE: Espie CA et al. JAMA Psychiatry. 2018 Sep 25. doi: 10.1001/jamapsychiatry.2018.2745.
FROM JAMA PSYCHIATRY
Key clinical point: Individuals who used a cognitive-behavioral therapy program and app had significant health, psychological, and quality-of-life improvements, compared with controls.
Major finding: Significant improvements were seen in self-reported measures of functional health, psychological well-being, and sleep-related quality of life were reported. About 45%-84% of the effects were mediated by improvements in insomnia symptoms.
Study details: An online, randomized, parallel-group trial including 1,711 adults with self-reported insomnia symptoms who received digital cognitive-behavioral therapy or sleep hygiene education.
Disclosures: The senior study author is the cofounder and chief medical officer of Big Health, which was involved in the design and conduct of the study, interpretation of data, and development of the manuscript. The study was funded by Big Health and other sources, including the National Institute for Health Research Oxford Biomedical Research Centre.
Source: Espie CA et al. JAMA Psychiatry. 2018 Sep 25. doi: 10.1001/jamapsychiatry.2018.2745.
Perianal Ulceration and Verrucous Papules
The Diagnosis: Herpes Simplex Virus Infection
Viral culture of the ulcer was positive for herpes simplex virus type 2 (HHV-2). Bacterial culture grew enteric flora. The patient was started on intravenous acyclovir 5 mg/kg every 8 hours for 7 days and then transitioned to oral acyclovir for chronic suppressive therapy. One month later, there was near-complete reepithelialization with 2 remaining 1-cm shallow ulcers. The verrucous lesions had dried up and were flaking off (Figure). At 6-month follow-up, the ulcers and verrucous lesions had completely resolved.
Herpes simplex virus type 2 is the most common cause of genital and perianal ulcers in immunocompromised individuals.1 Patients classically present with painful grouped vesicles followed by painful superficial ulcers that may rapidly progress to extensive confluent ulceration. A hypertrophic variant of genital herpes characterized by anogenital verrucous lesions, similar to condyloma acuminata, also can be seen in immunocompromised individuals.2 This form has almost exclusively been observed in patients with human immunodeficiency virus and may occur in isolation or together with the ulcerative form.1-5 A case of vegetative HHV infection of the genital area in a patient with common variable immunodeficiency has been reported.6 Verrucous lesions of the mouth secondary to HHV have been observed in Hodgkin lymphoma, acute myelogenous leukemia, and individuals on immunosuppressive medications.7-10
Perianal involvement of Crohn disease typically presents with fistulas, ulcers, abscesses, strictures, and skin tags in some cases. Invasive squamous cell carcinoma may arise within a chronic ulcer of the anogenital area or may itself manifest as an ulcer or anal fissure. Perianal ulcerative skin tuberculosis has been reported in the literature as a rare manifestation of extrapulmonary tuberculosis and should be considered in a patient with an appropriate clinical history. Pyoderma gangrenosum classically pre-sents as a large ulcer with irregular rolled borders, though a rare variant of vegetative pyoderma gangrenosum may manifest as a nodular or verrucous plaque.
Studies to diagnose HHV include viral cell culture, HHV polymerase chain reaction testing, HHV serology, and direct fluorescent antibody testing. Skin biopsy may be necessary to rule out underlying malignancy. Treatment of perianal HHV infection includes acyclovir, valacyclovir, or famciclovir.1,5,6 Hypertrophic lesions often are refractory to first-line antiviral therapy and may require surgical resection or treatment with alternative medications such as imiquimod, a topical immunomodulator.3,5,6,11
- Ranu H, Lee J, Chio M, et al. Tumour-like presentations of anogenital herpes simplex in HIV-positive patients. Int J STD AIDS. 2011;22:181-186.
- Tong P, Mutasim DF. Herpes simplex virus infection masquerading as condylomata accuminata in a patient with HIV disease. Br J Dermatol. 1996;134:797-800.
- Mosunjac M, Park J, Wang W, et al. Genital and perianal herpes simplex simulating neoplasia in patients with AIDS. AIDS Patient Care STDS. 2009;23:153-158.
- Gubinelli E, Cocuroccia B, Lazzarotto T, et al. Nodular perianal herpes simplex with prominent plasma cell infiltration. Sexually Transm Dis. 2003;30:157-159.
- Nadal SR, Calore EE, Manzione CR, et al. Hypertrophic herpes simplex simulating anal neoplasia in AIDS patients: report of five cases. Dis Colon Rectum. 2005;48:2289-2293.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Burke EM, Karp DL, Wu TC, et al. Atypical oral presentation of herpes simplex virus infection in a patient after orthotopic liver transplantation. Eur Arch Otorhinolaryngol. 1994;251:301-303.
- Tabaee A, Saltman B, Shutter J, et al. Recurrent oral herpes simplex virus infection presenting as a tongue mass. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:376-380.
- Leming PD, Martin SE, Zwelling LA. Atypical herpes simplex (HSV) infection in a patient with Hodgkin's disease. Cancer. 1984;54:3043-3047.
- Burgoyne M, Burke W. Atypical herpes simplex infection in patients with acute myelogenous leukemia recovering from chemotherapy. J Am Acad Dermatol. 1989;20:1125-1126.
- Deza G, Martin-Ezquerra G, Curto-Barredo L, et al. Successful treatment of hypertrophic herpes simplex genitalis in HIV-infected patient with topical imiquimod. J Dermatol. 2015;42:1176-1178.
The Diagnosis: Herpes Simplex Virus Infection
Viral culture of the ulcer was positive for herpes simplex virus type 2 (HHV-2). Bacterial culture grew enteric flora. The patient was started on intravenous acyclovir 5 mg/kg every 8 hours for 7 days and then transitioned to oral acyclovir for chronic suppressive therapy. One month later, there was near-complete reepithelialization with 2 remaining 1-cm shallow ulcers. The verrucous lesions had dried up and were flaking off (Figure). At 6-month follow-up, the ulcers and verrucous lesions had completely resolved.

Herpes simplex virus type 2 is the most common cause of genital and perianal ulcers in immunocompromised individuals.1 Patients classically present with painful grouped vesicles followed by painful superficial ulcers that may rapidly progress to extensive confluent ulceration. A hypertrophic variant of genital herpes characterized by anogenital verrucous lesions, similar to condyloma acuminata, also can be seen in immunocompromised individuals.2 This form has almost exclusively been observed in patients with human immunodeficiency virus and may occur in isolation or together with the ulcerative form.1-5 A case of vegetative HHV infection of the genital area in a patient with common variable immunodeficiency has been reported.6 Verrucous lesions of the mouth secondary to HHV have been observed in Hodgkin lymphoma, acute myelogenous leukemia, and individuals on immunosuppressive medications.7-10
Perianal involvement of Crohn disease typically presents with fistulas, ulcers, abscesses, strictures, and skin tags in some cases. Invasive squamous cell carcinoma may arise within a chronic ulcer of the anogenital area or may itself manifest as an ulcer or anal fissure. Perianal ulcerative skin tuberculosis has been reported in the literature as a rare manifestation of extrapulmonary tuberculosis and should be considered in a patient with an appropriate clinical history. Pyoderma gangrenosum classically pre-sents as a large ulcer with irregular rolled borders, though a rare variant of vegetative pyoderma gangrenosum may manifest as a nodular or verrucous plaque.
Studies to diagnose HHV include viral cell culture, HHV polymerase chain reaction testing, HHV serology, and direct fluorescent antibody testing. Skin biopsy may be necessary to rule out underlying malignancy. Treatment of perianal HHV infection includes acyclovir, valacyclovir, or famciclovir.1,5,6 Hypertrophic lesions often are refractory to first-line antiviral therapy and may require surgical resection or treatment with alternative medications such as imiquimod, a topical immunomodulator.3,5,6,11
The Diagnosis: Herpes Simplex Virus Infection
Viral culture of the ulcer was positive for herpes simplex virus type 2 (HHV-2). Bacterial culture grew enteric flora. The patient was started on intravenous acyclovir 5 mg/kg every 8 hours for 7 days and then transitioned to oral acyclovir for chronic suppressive therapy. One month later, there was near-complete reepithelialization with 2 remaining 1-cm shallow ulcers. The verrucous lesions had dried up and were flaking off (Figure). At 6-month follow-up, the ulcers and verrucous lesions had completely resolved.

Herpes simplex virus type 2 is the most common cause of genital and perianal ulcers in immunocompromised individuals.1 Patients classically present with painful grouped vesicles followed by painful superficial ulcers that may rapidly progress to extensive confluent ulceration. A hypertrophic variant of genital herpes characterized by anogenital verrucous lesions, similar to condyloma acuminata, also can be seen in immunocompromised individuals.2 This form has almost exclusively been observed in patients with human immunodeficiency virus and may occur in isolation or together with the ulcerative form.1-5 A case of vegetative HHV infection of the genital area in a patient with common variable immunodeficiency has been reported.6 Verrucous lesions of the mouth secondary to HHV have been observed in Hodgkin lymphoma, acute myelogenous leukemia, and individuals on immunosuppressive medications.7-10
Perianal involvement of Crohn disease typically presents with fistulas, ulcers, abscesses, strictures, and skin tags in some cases. Invasive squamous cell carcinoma may arise within a chronic ulcer of the anogenital area or may itself manifest as an ulcer or anal fissure. Perianal ulcerative skin tuberculosis has been reported in the literature as a rare manifestation of extrapulmonary tuberculosis and should be considered in a patient with an appropriate clinical history. Pyoderma gangrenosum classically pre-sents as a large ulcer with irregular rolled borders, though a rare variant of vegetative pyoderma gangrenosum may manifest as a nodular or verrucous plaque.
Studies to diagnose HHV include viral cell culture, HHV polymerase chain reaction testing, HHV serology, and direct fluorescent antibody testing. Skin biopsy may be necessary to rule out underlying malignancy. Treatment of perianal HHV infection includes acyclovir, valacyclovir, or famciclovir.1,5,6 Hypertrophic lesions often are refractory to first-line antiviral therapy and may require surgical resection or treatment with alternative medications such as imiquimod, a topical immunomodulator.3,5,6,11
- Ranu H, Lee J, Chio M, et al. Tumour-like presentations of anogenital herpes simplex in HIV-positive patients. Int J STD AIDS. 2011;22:181-186.
- Tong P, Mutasim DF. Herpes simplex virus infection masquerading as condylomata accuminata in a patient with HIV disease. Br J Dermatol. 1996;134:797-800.
- Mosunjac M, Park J, Wang W, et al. Genital and perianal herpes simplex simulating neoplasia in patients with AIDS. AIDS Patient Care STDS. 2009;23:153-158.
- Gubinelli E, Cocuroccia B, Lazzarotto T, et al. Nodular perianal herpes simplex with prominent plasma cell infiltration. Sexually Transm Dis. 2003;30:157-159.
- Nadal SR, Calore EE, Manzione CR, et al. Hypertrophic herpes simplex simulating anal neoplasia in AIDS patients: report of five cases. Dis Colon Rectum. 2005;48:2289-2293.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Burke EM, Karp DL, Wu TC, et al. Atypical oral presentation of herpes simplex virus infection in a patient after orthotopic liver transplantation. Eur Arch Otorhinolaryngol. 1994;251:301-303.
- Tabaee A, Saltman B, Shutter J, et al. Recurrent oral herpes simplex virus infection presenting as a tongue mass. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:376-380.
- Leming PD, Martin SE, Zwelling LA. Atypical herpes simplex (HSV) infection in a patient with Hodgkin's disease. Cancer. 1984;54:3043-3047.
- Burgoyne M, Burke W. Atypical herpes simplex infection in patients with acute myelogenous leukemia recovering from chemotherapy. J Am Acad Dermatol. 1989;20:1125-1126.
- Deza G, Martin-Ezquerra G, Curto-Barredo L, et al. Successful treatment of hypertrophic herpes simplex genitalis in HIV-infected patient with topical imiquimod. J Dermatol. 2015;42:1176-1178.
- Ranu H, Lee J, Chio M, et al. Tumour-like presentations of anogenital herpes simplex in HIV-positive patients. Int J STD AIDS. 2011;22:181-186.
- Tong P, Mutasim DF. Herpes simplex virus infection masquerading as condylomata accuminata in a patient with HIV disease. Br J Dermatol. 1996;134:797-800.
- Mosunjac M, Park J, Wang W, et al. Genital and perianal herpes simplex simulating neoplasia in patients with AIDS. AIDS Patient Care STDS. 2009;23:153-158.
- Gubinelli E, Cocuroccia B, Lazzarotto T, et al. Nodular perianal herpes simplex with prominent plasma cell infiltration. Sexually Transm Dis. 2003;30:157-159.
- Nadal SR, Calore EE, Manzione CR, et al. Hypertrophic herpes simplex simulating anal neoplasia in AIDS patients: report of five cases. Dis Colon Rectum. 2005;48:2289-2293.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Burke EM, Karp DL, Wu TC, et al. Atypical oral presentation of herpes simplex virus infection in a patient after orthotopic liver transplantation. Eur Arch Otorhinolaryngol. 1994;251:301-303.
- Tabaee A, Saltman B, Shutter J, et al. Recurrent oral herpes simplex virus infection presenting as a tongue mass. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:376-380.
- Leming PD, Martin SE, Zwelling LA. Atypical herpes simplex (HSV) infection in a patient with Hodgkin's disease. Cancer. 1984;54:3043-3047.
- Burgoyne M, Burke W. Atypical herpes simplex infection in patients with acute myelogenous leukemia recovering from chemotherapy. J Am Acad Dermatol. 1989;20:1125-1126.
- Deza G, Martin-Ezquerra G, Curto-Barredo L, et al. Successful treatment of hypertrophic herpes simplex genitalis in HIV-infected patient with topical imiquimod. J Dermatol. 2015;42:1176-1178.
A 75-year-old woman with chronic lymphocytic leukemia undergoing ibrutinib targeted therapy presented to the emergency department with fever and perianal pain of 4 months' duration. The patient denied history of genital or perianal ulcers, warts, masses, bedsores, prolonged immobilization, anal surgeries, or recent travel. She had not been previously treated for the perianal pain. On physical examination there was an 18×15-cm shallow ulceration with rolled borders involving the intergluteal cleft and perianal area. There were numerous hyperpigmented verrucous papules clustered in the center of the ulceration. No vesicles or bullae were present. Laboratory results were pertinent for a white blood cell count of 3600/µL (reference range, 4500-11,000/µL) and absolute neutrophil count of 1300/µL (reference range, 1900-8000/µL). Human immunodeficiency virus testing was negative.
Signs point to growing abuse of gabapentinoids in the U.S.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
LAS VEGAS – Up to 22% of opioid abusers also abuse gabapentin, taking high doses of the antiepileptic for its psychoactive effects, or to potentiate the effect of opioids.
The drug – the 10th most commonly prescribed in the United States – is increasingly implicated in overdose deaths. In response, several states have recently reclassified the antiepileptic as a Schedule V controlled substance. Other states have declined to go that far, but have added gabapentin (Neurontin and others) to the drugs that must be reported to state prescription monitoring programs.
“Gabapentin and pregabalin [Lyrica] are versatile and important drugs that are widely prescribed and used off label for a number of conditions from seizure disorder to fibromyalgia,” said Joseph Pergolizzi Jr., MD, who discussed the issue during the annual PAINWeek. “Abuse patterns differ somewhat from abuse patterns with prescription opioids. People who misuse gabapentinoids tend to already use other drugs inappropriately. It’s rare to find a person who only takes them recreationally, but it is increasingly common to find polydrug abusers who take gabapentinoids.”
Gabapentin abuse appears to be more common in Europe than in the United States, where it’s just beginning to emerge, Dr. Pergolizzi said. And the picture is more nuanced than it might first appear: Many of those who are misusing the drug are using it as a “do-it-yourself” opioid withdrawal aid, he said in an interview.
At the meeting, he presented a literature review comprising 46 papers on pregabalin abuse and 263 on gabapentin abuse. Several important themes arose from these papers, said Dr. Pergolizzi, cofounder and chief operating officer of NEMA Research Inc., Naples, Fla.:
- Gabapentin and pregabalin are being prescribed off label for numerous conditions, including bipolar disorder, neuropathic pain, diabetic neuropathy, complex regional pain syndrome, attention deficit disorder, restless leg syndrome, trigeminal neuralgia, periodic limb movement disorder of sleep, migraine, drug and alcohol withdrawal seizures, chronic low back pain, and even menopausal symptoms.
- About a one-third of users experience withdrawal symptoms with sudden discontinuation. Symptoms include disorientation, anxiety, insomnia, heart palpitations, diaphoresis, and abdominal cramps.
- Risk factors for abuse are emerging. These include opioid use disorders, mental illnesses, and a history of taking supratherapeutic doses of the drugs. Age and sex don’t seem to be a consistent risk factor for abuse.
- Abusers can obtain nonprescription gabapentinoids more easily each year, including street sales and online orders, Dr. Pergolizzi said. “A Google search conducted in July for ‘buy gabapentin without a prescription’ yielded 4.48 million results and ‘buy pregabalin without a prescription,’ 622,000. A similar search conducted in September 2017 yielded 1.19 million and 352,000 results, respectively.”
- Few urine drug assays screen for gabapentinoids, which makes them easy to conceal in random drug testing.
Reports of gabapentin-involved drug overdoses and even deaths have recently emerged in the United States, particularly in the opioid abuse-plagued Appalachian states. An article in May in Drug and Alcohol Dependence examined the prevalence of gabapentin in postmortem toxicology in drug overdose deaths in four Appalachian states in 2015 (Drug Alcohol Depend. 2018;186:80-5). Rates were 4% in eastern Tennessee, 15% in West Virginia, 20% in North Carolina, and 41% in Kentucky.
Three states have now added gabapentin to their list of Schedule V controlled substances: Kentucky in 2017, West Virginia this May, and Tennessee in July.
Ohio, Minnesota, Virginia, and Massachusetts have taken a different tack to controlling dispensing. In those states, all pharmacies, prescribers, and wholesalers must report all dispensing and sales of gabapentin to their prescription monitoring databases.
Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
REPORTING FROM PAINWEEK 2018
Key clinical point:
Major finding: Up to 22% of opioid users are also using gabapentin.
Study details: A literature review of 46 papers on pregabalin abuse and 263 on gabapentin abuse.
Disclosures: Dr. Pergolizzi disclosed financial relationships with numerous pharmaceutical companies.
Source: Pergolizzi J et al. PAINWeek 2018, Poster 55.