User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
New testing guidelines for infants with possible Zika virus infection
The Centers for Disease Control and Prevention has released interim guidelines for U.S. clinicians caring for infants born to mothers who traveled to or resided in an area with Zika virus transmission during pregnancy.
The guidelines, released Jan. 26, address the evaluation and testing of infants with possible congenital Zika virus infection, and follow the Jan. 19 release of similar guidelines for the care of pregnant women with possible exposure to the mosquito-borne virus. Most importantly, the new guidelines say Zika virus testing should be performed for infants with microcephaly or intracranial calcifications who are born to women with possible Zika virus exposure during pregnancy, and for infants born to women with positive or inconclusive Zika virus test results.
“Pediatric health are providers should work closely with obstetric providers to identify infants whose mothers were potentially infected with Zika virus during pregnancy (based on travel to or residence in an area with Zika virus transmission),” according to the guidelines, which were published in Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 26;65[Early Release:1-5]).
Infants with laboratory evidence of a possible congenital Zika virus infection should undergo additional clinical evaluation, and state or territorial health departments should be contacted to facilitate testing. Zika virus disease is an arboviral disease and thus is a nationally notifiable condition, according to guideline authors Dr. J. Erin Staples and her colleagues at the CDC, Atlanta.
Both molecular and serologic tests are recommended for infants undergoing evaluation for possible congenital Zika virus infection, they noted.
Serum specimens for reverse-transcription-polymerase chain reaction testing should be collected from the umbilical cord or directly from the infant within 2 days of birth, and cerebrospinal fluid collected for other studies, as well as frozen and fixed placenta obtained at delivery, should also be tested by RT-PCR.
IgM ELISA for Zika virus and dengue virus should also be performed on infant serum, infant CSF, and maternal serum, but results from these assays can be falsely positive because of cross-reacting antibodies, the authors noted.
Other tests that can be considered include a plaque reduction neutralization test to measure virus-specific neutralizing antibodies and to discriminate between cross-reacting antibodies and closely related flaviviruses, and immunohistochemical staining to detect the virus antigen on fixed placenta and umbilical cord tissues.
Further clinical evaluation and laboratory testing is recommended for infants with microcephaly or intracranial calcifications detected prenatally or at birth if the mother was potentially infected during pregnancy, they said.
In infants with possible Zika virus exposure during pregnancy, but without microcephaly or intracranial calcification, subsequent evaluation depends on maternal testing results. Routine care is recommended if maternal test results are negative, and testing for a possible congenital infection is recommended if maternal results are positive or inconclusive.
If all of an infant’s tests are negative for Zika virus infection, no further Zika virus testing or evaluation is recommended. In the event of any positive or inconclusive test, further evaluation and follow-up is recommended.
Other considerations
Abnormal eye findings have been reported in infants with possible congenital infection, therefore an opthalmologic evaluation, including retinal examination is advised during the first month of life, as is a repeat hearing screen at age 6 months – even if the initial screen was normal, the authors said.
The infant should be followed to assess for long-term sequelae, and the case should be reported. Follow-up should include a cranial ultrasound to assess for subclinical findings, unless a third trimester ultrasound showed no brain abnormalities, they added.
No specific antiviral treatment or vaccine exists for Zika virus infection; treatment is supportive and should address specific medical and neurodevelopmental issues, and mothers should be encouraged to breastfeed infants regardless of exposure, as available evidence suggests the benefits of breastfeeding outweigh the theoretical risks of transmission through breast milk, they said.
The authors stressed that prevention of maternal infection is the only way to prevent congenital Zika virus infection and is achieved by avoiding areas with ongoing Zika virus transmission or by strictly following steps to avoid mosquito bites by using air-conditioning or window and door screens, wearing protective clothing, and using insect repellents.
Environmental Protection Agency–registered insect repellents are safe for pregnant women when used according to the product label, they noted.
The Centers for Disease Control and Prevention has released interim guidelines for U.S. clinicians caring for infants born to mothers who traveled to or resided in an area with Zika virus transmission during pregnancy.
The guidelines, released Jan. 26, address the evaluation and testing of infants with possible congenital Zika virus infection, and follow the Jan. 19 release of similar guidelines for the care of pregnant women with possible exposure to the mosquito-borne virus. Most importantly, the new guidelines say Zika virus testing should be performed for infants with microcephaly or intracranial calcifications who are born to women with possible Zika virus exposure during pregnancy, and for infants born to women with positive or inconclusive Zika virus test results.
“Pediatric health are providers should work closely with obstetric providers to identify infants whose mothers were potentially infected with Zika virus during pregnancy (based on travel to or residence in an area with Zika virus transmission),” according to the guidelines, which were published in Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 26;65[Early Release:1-5]).
Infants with laboratory evidence of a possible congenital Zika virus infection should undergo additional clinical evaluation, and state or territorial health departments should be contacted to facilitate testing. Zika virus disease is an arboviral disease and thus is a nationally notifiable condition, according to guideline authors Dr. J. Erin Staples and her colleagues at the CDC, Atlanta.
Both molecular and serologic tests are recommended for infants undergoing evaluation for possible congenital Zika virus infection, they noted.
Serum specimens for reverse-transcription-polymerase chain reaction testing should be collected from the umbilical cord or directly from the infant within 2 days of birth, and cerebrospinal fluid collected for other studies, as well as frozen and fixed placenta obtained at delivery, should also be tested by RT-PCR.
IgM ELISA for Zika virus and dengue virus should also be performed on infant serum, infant CSF, and maternal serum, but results from these assays can be falsely positive because of cross-reacting antibodies, the authors noted.
Other tests that can be considered include a plaque reduction neutralization test to measure virus-specific neutralizing antibodies and to discriminate between cross-reacting antibodies and closely related flaviviruses, and immunohistochemical staining to detect the virus antigen on fixed placenta and umbilical cord tissues.
Further clinical evaluation and laboratory testing is recommended for infants with microcephaly or intracranial calcifications detected prenatally or at birth if the mother was potentially infected during pregnancy, they said.
In infants with possible Zika virus exposure during pregnancy, but without microcephaly or intracranial calcification, subsequent evaluation depends on maternal testing results. Routine care is recommended if maternal test results are negative, and testing for a possible congenital infection is recommended if maternal results are positive or inconclusive.
If all of an infant’s tests are negative for Zika virus infection, no further Zika virus testing or evaluation is recommended. In the event of any positive or inconclusive test, further evaluation and follow-up is recommended.
Other considerations
Abnormal eye findings have been reported in infants with possible congenital infection, therefore an opthalmologic evaluation, including retinal examination is advised during the first month of life, as is a repeat hearing screen at age 6 months – even if the initial screen was normal, the authors said.
The infant should be followed to assess for long-term sequelae, and the case should be reported. Follow-up should include a cranial ultrasound to assess for subclinical findings, unless a third trimester ultrasound showed no brain abnormalities, they added.
No specific antiviral treatment or vaccine exists for Zika virus infection; treatment is supportive and should address specific medical and neurodevelopmental issues, and mothers should be encouraged to breastfeed infants regardless of exposure, as available evidence suggests the benefits of breastfeeding outweigh the theoretical risks of transmission through breast milk, they said.
The authors stressed that prevention of maternal infection is the only way to prevent congenital Zika virus infection and is achieved by avoiding areas with ongoing Zika virus transmission or by strictly following steps to avoid mosquito bites by using air-conditioning or window and door screens, wearing protective clothing, and using insect repellents.
Environmental Protection Agency–registered insect repellents are safe for pregnant women when used according to the product label, they noted.
The Centers for Disease Control and Prevention has released interim guidelines for U.S. clinicians caring for infants born to mothers who traveled to or resided in an area with Zika virus transmission during pregnancy.
The guidelines, released Jan. 26, address the evaluation and testing of infants with possible congenital Zika virus infection, and follow the Jan. 19 release of similar guidelines for the care of pregnant women with possible exposure to the mosquito-borne virus. Most importantly, the new guidelines say Zika virus testing should be performed for infants with microcephaly or intracranial calcifications who are born to women with possible Zika virus exposure during pregnancy, and for infants born to women with positive or inconclusive Zika virus test results.
“Pediatric health are providers should work closely with obstetric providers to identify infants whose mothers were potentially infected with Zika virus during pregnancy (based on travel to or residence in an area with Zika virus transmission),” according to the guidelines, which were published in Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 26;65[Early Release:1-5]).
Infants with laboratory evidence of a possible congenital Zika virus infection should undergo additional clinical evaluation, and state or territorial health departments should be contacted to facilitate testing. Zika virus disease is an arboviral disease and thus is a nationally notifiable condition, according to guideline authors Dr. J. Erin Staples and her colleagues at the CDC, Atlanta.
Both molecular and serologic tests are recommended for infants undergoing evaluation for possible congenital Zika virus infection, they noted.
Serum specimens for reverse-transcription-polymerase chain reaction testing should be collected from the umbilical cord or directly from the infant within 2 days of birth, and cerebrospinal fluid collected for other studies, as well as frozen and fixed placenta obtained at delivery, should also be tested by RT-PCR.
IgM ELISA for Zika virus and dengue virus should also be performed on infant serum, infant CSF, and maternal serum, but results from these assays can be falsely positive because of cross-reacting antibodies, the authors noted.
Other tests that can be considered include a plaque reduction neutralization test to measure virus-specific neutralizing antibodies and to discriminate between cross-reacting antibodies and closely related flaviviruses, and immunohistochemical staining to detect the virus antigen on fixed placenta and umbilical cord tissues.
Further clinical evaluation and laboratory testing is recommended for infants with microcephaly or intracranial calcifications detected prenatally or at birth if the mother was potentially infected during pregnancy, they said.
In infants with possible Zika virus exposure during pregnancy, but without microcephaly or intracranial calcification, subsequent evaluation depends on maternal testing results. Routine care is recommended if maternal test results are negative, and testing for a possible congenital infection is recommended if maternal results are positive or inconclusive.
If all of an infant’s tests are negative for Zika virus infection, no further Zika virus testing or evaluation is recommended. In the event of any positive or inconclusive test, further evaluation and follow-up is recommended.
Other considerations
Abnormal eye findings have been reported in infants with possible congenital infection, therefore an opthalmologic evaluation, including retinal examination is advised during the first month of life, as is a repeat hearing screen at age 6 months – even if the initial screen was normal, the authors said.
The infant should be followed to assess for long-term sequelae, and the case should be reported. Follow-up should include a cranial ultrasound to assess for subclinical findings, unless a third trimester ultrasound showed no brain abnormalities, they added.
No specific antiviral treatment or vaccine exists for Zika virus infection; treatment is supportive and should address specific medical and neurodevelopmental issues, and mothers should be encouraged to breastfeed infants regardless of exposure, as available evidence suggests the benefits of breastfeeding outweigh the theoretical risks of transmission through breast milk, they said.
The authors stressed that prevention of maternal infection is the only way to prevent congenital Zika virus infection and is achieved by avoiding areas with ongoing Zika virus transmission or by strictly following steps to avoid mosquito bites by using air-conditioning or window and door screens, wearing protective clothing, and using insect repellents.
Environmental Protection Agency–registered insect repellents are safe for pregnant women when used according to the product label, they noted.
FROM MMWR
Gene signatures aid diagnosis of acute respiratory infection etiology
Pathogen-specific host gene expression patterns accurately discriminated most noninfectious from infectious illnesses, and bacterial from viral causes of acute respiratory infection (ARI) in an observational study conducted in acute care settings.
The findings could have important implications for combating inappropriate antibiotic use and emerging antibiotic resistance, Dr. Ephraim L. Tsalik of the department of medicine at Duke University, Durham, N.C., and his colleagues reported online Jan. 20 in Science Translational Medicine.
The investigators analyzed peripheral whole-blood gene expression from 273 subjects with community-onset viral ARI (115 subjects), bacterial ARI (70 subjects), or noninfectious illness (88 subjects) who were seen in an emergency department, and from 44 healthy control subjects. Classifiers for bacterial ARI, viral ARI, and noninfectious causes of illness were developed, and were 87% accurate overall (Sci Transl Med. 2016;8[322]:322ra11. doi/ 10.1126/scitranslmed.aad6873).
“Bacterial ARI was identified in 83% of patients and excluded in 94% without bacterial infection. Viral ARI was identified in 90% and excluded in 92% of cases. Using the noninfectious illness classifier, infection was excluded in 86% of cases,” they wrote.
The classifiers were more accurate than procalcitonin – a widely used biomarker with some specificity for bacterial infection (86% vs. 78% accuracy in 238 available samples), and three published classifiers of bacterial vs. viral infection, and were validated in five publicly available data sets, they noted.
The gene signature patterns identified in the course of this study mark an important step toward development of a rapid blood test that could be used in clinics to guide appropriate treatment for ARIs, the investigators said.
Precision treatment of viruses
More precise ways to distinguish infections could reduce unnecessary antibiotic use and lead to more precise treatment of viruses, senior author Dr. Geoffrey S. Ginsburg, director of Duke’s Center for Applied Genomics & Precision Medicine, said in a press statement.
“Right now, we can give patients [oseltamivir] Tamiflu to help them recover from an influenza infection, but for most viral infections, the treatment is fluids and rest until it resolves. In the next 5-10 years, we will likely see new antiviral medications for common bugs like respiratory syncytial virus and even rhinovirus, and guiding treatment choices will be even more important,” he added.
Senior author Dr. Christopher W. Woods, also of Duke University, further explained in an interview that the findings are particularly exciting because “there isn’t much out there that accomplishes what we’ve done. So just about any level of accuracy is an improvement.” Further, he said the test is “a tool to aid in diagnosis, used in conjunction with the patient’s symptoms, examination, and other testing. So an imperfect test is okay, because it does not stand alone.”
Next steps include putting the assay on a testing platform that can be used at the point of care, and validating the findings in all populations, including infants, the elderly, and across ethnic groups, he said.
“The work is ongoing, and we expect to have results available within the course of an outpatient visit in the near future,” Dr. Woods, also a professor of medicine and global health, added, noting that efforts also are underway to “expand the repertoire of this approach to many different types of viral and bacterial infections and also to fungal infections, and to address the challenges of critically ill patients in intensive care units.”
This study was supported by the U.S. Defense Advanced Research Projects Agency, the National Institutes of Health, the Agency for Healthcare Research and Quality, the U.S. Department of Veterans Affairs Office of Research and Development, and an in-kind contribution from bioMérieux. The authors reported having no relevant competing interests.
Pathogen-specific host gene expression patterns accurately discriminated most noninfectious from infectious illnesses, and bacterial from viral causes of acute respiratory infection (ARI) in an observational study conducted in acute care settings.
The findings could have important implications for combating inappropriate antibiotic use and emerging antibiotic resistance, Dr. Ephraim L. Tsalik of the department of medicine at Duke University, Durham, N.C., and his colleagues reported online Jan. 20 in Science Translational Medicine.
The investigators analyzed peripheral whole-blood gene expression from 273 subjects with community-onset viral ARI (115 subjects), bacterial ARI (70 subjects), or noninfectious illness (88 subjects) who were seen in an emergency department, and from 44 healthy control subjects. Classifiers for bacterial ARI, viral ARI, and noninfectious causes of illness were developed, and were 87% accurate overall (Sci Transl Med. 2016;8[322]:322ra11. doi/ 10.1126/scitranslmed.aad6873).
“Bacterial ARI was identified in 83% of patients and excluded in 94% without bacterial infection. Viral ARI was identified in 90% and excluded in 92% of cases. Using the noninfectious illness classifier, infection was excluded in 86% of cases,” they wrote.
The classifiers were more accurate than procalcitonin – a widely used biomarker with some specificity for bacterial infection (86% vs. 78% accuracy in 238 available samples), and three published classifiers of bacterial vs. viral infection, and were validated in five publicly available data sets, they noted.
The gene signature patterns identified in the course of this study mark an important step toward development of a rapid blood test that could be used in clinics to guide appropriate treatment for ARIs, the investigators said.
Precision treatment of viruses
More precise ways to distinguish infections could reduce unnecessary antibiotic use and lead to more precise treatment of viruses, senior author Dr. Geoffrey S. Ginsburg, director of Duke’s Center for Applied Genomics & Precision Medicine, said in a press statement.
“Right now, we can give patients [oseltamivir] Tamiflu to help them recover from an influenza infection, but for most viral infections, the treatment is fluids and rest until it resolves. In the next 5-10 years, we will likely see new antiviral medications for common bugs like respiratory syncytial virus and even rhinovirus, and guiding treatment choices will be even more important,” he added.
Senior author Dr. Christopher W. Woods, also of Duke University, further explained in an interview that the findings are particularly exciting because “there isn’t much out there that accomplishes what we’ve done. So just about any level of accuracy is an improvement.” Further, he said the test is “a tool to aid in diagnosis, used in conjunction with the patient’s symptoms, examination, and other testing. So an imperfect test is okay, because it does not stand alone.”
Next steps include putting the assay on a testing platform that can be used at the point of care, and validating the findings in all populations, including infants, the elderly, and across ethnic groups, he said.
“The work is ongoing, and we expect to have results available within the course of an outpatient visit in the near future,” Dr. Woods, also a professor of medicine and global health, added, noting that efforts also are underway to “expand the repertoire of this approach to many different types of viral and bacterial infections and also to fungal infections, and to address the challenges of critically ill patients in intensive care units.”
This study was supported by the U.S. Defense Advanced Research Projects Agency, the National Institutes of Health, the Agency for Healthcare Research and Quality, the U.S. Department of Veterans Affairs Office of Research and Development, and an in-kind contribution from bioMérieux. The authors reported having no relevant competing interests.
Pathogen-specific host gene expression patterns accurately discriminated most noninfectious from infectious illnesses, and bacterial from viral causes of acute respiratory infection (ARI) in an observational study conducted in acute care settings.
The findings could have important implications for combating inappropriate antibiotic use and emerging antibiotic resistance, Dr. Ephraim L. Tsalik of the department of medicine at Duke University, Durham, N.C., and his colleagues reported online Jan. 20 in Science Translational Medicine.
The investigators analyzed peripheral whole-blood gene expression from 273 subjects with community-onset viral ARI (115 subjects), bacterial ARI (70 subjects), or noninfectious illness (88 subjects) who were seen in an emergency department, and from 44 healthy control subjects. Classifiers for bacterial ARI, viral ARI, and noninfectious causes of illness were developed, and were 87% accurate overall (Sci Transl Med. 2016;8[322]:322ra11. doi/ 10.1126/scitranslmed.aad6873).
“Bacterial ARI was identified in 83% of patients and excluded in 94% without bacterial infection. Viral ARI was identified in 90% and excluded in 92% of cases. Using the noninfectious illness classifier, infection was excluded in 86% of cases,” they wrote.
The classifiers were more accurate than procalcitonin – a widely used biomarker with some specificity for bacterial infection (86% vs. 78% accuracy in 238 available samples), and three published classifiers of bacterial vs. viral infection, and were validated in five publicly available data sets, they noted.
The gene signature patterns identified in the course of this study mark an important step toward development of a rapid blood test that could be used in clinics to guide appropriate treatment for ARIs, the investigators said.
Precision treatment of viruses
More precise ways to distinguish infections could reduce unnecessary antibiotic use and lead to more precise treatment of viruses, senior author Dr. Geoffrey S. Ginsburg, director of Duke’s Center for Applied Genomics & Precision Medicine, said in a press statement.
“Right now, we can give patients [oseltamivir] Tamiflu to help them recover from an influenza infection, but for most viral infections, the treatment is fluids and rest until it resolves. In the next 5-10 years, we will likely see new antiviral medications for common bugs like respiratory syncytial virus and even rhinovirus, and guiding treatment choices will be even more important,” he added.
Senior author Dr. Christopher W. Woods, also of Duke University, further explained in an interview that the findings are particularly exciting because “there isn’t much out there that accomplishes what we’ve done. So just about any level of accuracy is an improvement.” Further, he said the test is “a tool to aid in diagnosis, used in conjunction with the patient’s symptoms, examination, and other testing. So an imperfect test is okay, because it does not stand alone.”
Next steps include putting the assay on a testing platform that can be used at the point of care, and validating the findings in all populations, including infants, the elderly, and across ethnic groups, he said.
“The work is ongoing, and we expect to have results available within the course of an outpatient visit in the near future,” Dr. Woods, also a professor of medicine and global health, added, noting that efforts also are underway to “expand the repertoire of this approach to many different types of viral and bacterial infections and also to fungal infections, and to address the challenges of critically ill patients in intensive care units.”
This study was supported by the U.S. Defense Advanced Research Projects Agency, the National Institutes of Health, the Agency for Healthcare Research and Quality, the U.S. Department of Veterans Affairs Office of Research and Development, and an in-kind contribution from bioMérieux. The authors reported having no relevant competing interests.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Pathogen-specific host gene expression patterns accurately discriminated most noninfectious from infectious illnesses, and bacterial from viral causes of acute respiratory infection (ARI) in an observational study.
Major finding: Classifiers for bacterial ARI, viral ARI, and noninfectious causes of illness were 87% accurate overall.
Data source: An observational cohort study involving 273 patients and 44 controls.
Disclosures: This study was supported by the U.S. Defense Advanced Research Projects Agency, the National Institutes of Health, the Agency for Healthcare Research and Quality, the U.S. Department of Veterans Affairs Office of Research and Development, and an in-kind contribution from bioMérieux. The authors reported having no relevant competing interests..
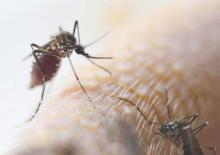
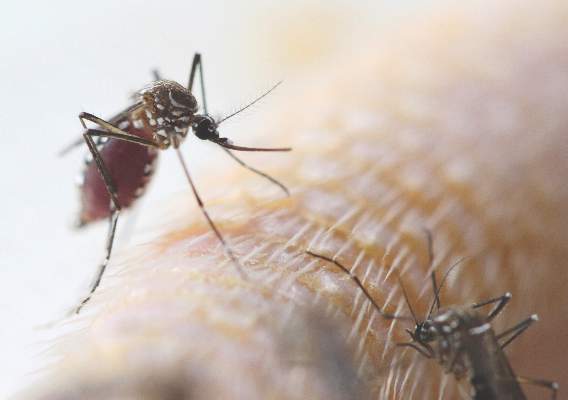
CDC: Ask Pregnant Women About Zika Virus Exposure
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
FROM MMWR
CDC: Ask pregnant women about Zika virus exposure
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
The Centers for Disease Control and Prevention has released interim guidelines for the care of pregnant women during a Zika virus outbreak.
The guidelines, developed in consultation with the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine, come on the heels of a Jan. 15 advisory warning pregnant women to avoid traveling to tropical countries and territories with outbreaks of the mosquito-borne virus. The virus is typically associated with only mild symptoms, but has been linked with cases of microcephaly and other poor outcomes in pregnancy.
“Health care providers should ask all pregnant women about recent travel. Pregnant women with a history of travel to an area with Zika virus transmission who report two or more symptoms consistent with Zika virus disease (acute onset of fever, maculopapular rash, arthralgia, or conjunctivitis) during or within 2 weeks of travel, or who have ultrasound findings of fetal microcephaly or intracranial calcifications, should be tested for Zika virus infection in consultation with their state or local health department,” according to the guidelines, which were published Jan. 19 in an early release of the Morbidity and Mortality Weekly Report (MMWR. 2016 Jan 19;65[Early Release]:1-4).
Testing is not indicated for women who have not traveled to areas with Zika virus transmission, according to CDC.
For women who test positive for the virus, serial ultrasounds to monitor fetal growth and anatomy should be considered, as well as referral to a maternal-fetal medicine specialist or infectious disease specialist with expertise in pregnancy management, according to the guidelines.
While a positive reverse-transcription polymerase chain reaction (RT-PCR) result on amniotic fluid would suggest intrauterine infection and could potentially be useful to pregnant women and their health care providers, it is currently not known how sensitive or specific the test is for congenital infection or whether a positive result is predictive of a subsequent fetal abnormality.
“Health care providers should discuss the risks and benefits of amniocentesis with their patients,” CDC officials wrote in the interim guidance.
The following tests are advised for live births with evidence of infection: histopathological examination of the placenta and umbilical cord, testing of frozen placental tissue and cord tissue for Zika virus RNA, and testing of cord serum for Zika and dengue virus IgM and neutralizing antibodies. Guidelines for infected infants are currently being developed.
No specific treatment exists for Zika virus infection; supportive care, including rest, fluids, use of analgesics and antipyretics, and acetaminophen for fever is advised.
The CDC continues to recommend that pregnant women avoid travel to areas where Zika virus transmission is ongoing. Strict steps to avoid mosquito bites are advised for those who do travel to such areas. This includes use of protective clothing and U.S. Environmental Protection Agency–registered insect repellents, as well as staying and sleeping in screened-in or air-conditioned rooms. Insect repellents containing DEET, picaridin, and IR3535 are safe for pregnant women when used as directed on the label, according to the CDC.
Updates on areas with ongoing Zika virus transmission are available on the CDC website.
FROM MMWR
Concerning number of HIV-associated Hodgkin lymphoma patients go untreated
Lower 5-year survival among HIV-positive vs. HIV-negative Hodgkin lymphoma patients is largely attributable to a high rate of non-treatment among those who are HIV positive, a review of cases from the National Cancer Data Base suggests.
Of 2,090 HIV-associated Hodgkin lymphoma patients diagnosed between 2004 and 2012 and included in the database, 81% received chemotherapy, and 13% received radiation therapy, but 16% received no treatment. Survival was 66% compared with 80% survival among HIV-negative Hodgkin lymphoma patients, reported Dr. Adam J. Olszewski of Brown University, Providence, R.I. and Dr. Jorge J. Castillo of Harvard Medical School, Boston (AIDS. 2016 Jan 4. doi: 10.1097/QAD.0000000000000986).
Another factor that contributed to the disparity was poor prognosis in patients with undetermined histologic subtype.
Among patients who received chemotherapy, HIV-positive status was not significantly associated with increased mortality in those with classical histologic subtypes, the authors said.
For example, the hazard ratios for mortality were 1.08 for nodular sclerosis and 1.06 for mixed cellularity. Those with undetermined histology had a significantly worse prognosis (hazard ratio, 1.56), and particular attention should be paid to these patients given their worse prognosis and high risk of nontreatment, the authors said.
Factors found on logistic regression analysis to be associated with higher risk of nontreatment among all patients were advanced age, male sex, nonwhite race, poor socioeconomic status, and undetermined histologic subtype. In 2012, the majority of HIV-positive patients were of nonwhite race; nearly half (49%) were black – an increase from 31% in 2004, and 15% were Hispanic, which raises additional concerns about the disparity in treatment delivery between HIV-positive and HIV-negative patients with Hodgkin lymphoma, they noted.
Dr. Olszewski is supported by the American Society of Hematology Scholar Award and the American Cancer Society Research Scholar Award. Both authors reported having no relevant conflicts of interest.
Lower 5-year survival among HIV-positive vs. HIV-negative Hodgkin lymphoma patients is largely attributable to a high rate of non-treatment among those who are HIV positive, a review of cases from the National Cancer Data Base suggests.
Of 2,090 HIV-associated Hodgkin lymphoma patients diagnosed between 2004 and 2012 and included in the database, 81% received chemotherapy, and 13% received radiation therapy, but 16% received no treatment. Survival was 66% compared with 80% survival among HIV-negative Hodgkin lymphoma patients, reported Dr. Adam J. Olszewski of Brown University, Providence, R.I. and Dr. Jorge J. Castillo of Harvard Medical School, Boston (AIDS. 2016 Jan 4. doi: 10.1097/QAD.0000000000000986).
Another factor that contributed to the disparity was poor prognosis in patients with undetermined histologic subtype.
Among patients who received chemotherapy, HIV-positive status was not significantly associated with increased mortality in those with classical histologic subtypes, the authors said.
For example, the hazard ratios for mortality were 1.08 for nodular sclerosis and 1.06 for mixed cellularity. Those with undetermined histology had a significantly worse prognosis (hazard ratio, 1.56), and particular attention should be paid to these patients given their worse prognosis and high risk of nontreatment, the authors said.
Factors found on logistic regression analysis to be associated with higher risk of nontreatment among all patients were advanced age, male sex, nonwhite race, poor socioeconomic status, and undetermined histologic subtype. In 2012, the majority of HIV-positive patients were of nonwhite race; nearly half (49%) were black – an increase from 31% in 2004, and 15% were Hispanic, which raises additional concerns about the disparity in treatment delivery between HIV-positive and HIV-negative patients with Hodgkin lymphoma, they noted.
Dr. Olszewski is supported by the American Society of Hematology Scholar Award and the American Cancer Society Research Scholar Award. Both authors reported having no relevant conflicts of interest.
Lower 5-year survival among HIV-positive vs. HIV-negative Hodgkin lymphoma patients is largely attributable to a high rate of non-treatment among those who are HIV positive, a review of cases from the National Cancer Data Base suggests.
Of 2,090 HIV-associated Hodgkin lymphoma patients diagnosed between 2004 and 2012 and included in the database, 81% received chemotherapy, and 13% received radiation therapy, but 16% received no treatment. Survival was 66% compared with 80% survival among HIV-negative Hodgkin lymphoma patients, reported Dr. Adam J. Olszewski of Brown University, Providence, R.I. and Dr. Jorge J. Castillo of Harvard Medical School, Boston (AIDS. 2016 Jan 4. doi: 10.1097/QAD.0000000000000986).
Another factor that contributed to the disparity was poor prognosis in patients with undetermined histologic subtype.
Among patients who received chemotherapy, HIV-positive status was not significantly associated with increased mortality in those with classical histologic subtypes, the authors said.
For example, the hazard ratios for mortality were 1.08 for nodular sclerosis and 1.06 for mixed cellularity. Those with undetermined histology had a significantly worse prognosis (hazard ratio, 1.56), and particular attention should be paid to these patients given their worse prognosis and high risk of nontreatment, the authors said.
Factors found on logistic regression analysis to be associated with higher risk of nontreatment among all patients were advanced age, male sex, nonwhite race, poor socioeconomic status, and undetermined histologic subtype. In 2012, the majority of HIV-positive patients were of nonwhite race; nearly half (49%) were black – an increase from 31% in 2004, and 15% were Hispanic, which raises additional concerns about the disparity in treatment delivery between HIV-positive and HIV-negative patients with Hodgkin lymphoma, they noted.
Dr. Olszewski is supported by the American Society of Hematology Scholar Award and the American Cancer Society Research Scholar Award. Both authors reported having no relevant conflicts of interest.
FROM AIDS
Key clinical point: Lower 5-year survival among HIV-positive vs. HIV-negative Hodgkin lymphoma patients is largely attributable to a high rate of nontreatment among those who are HIV positive, a review of cases from the National Cancer Data Base suggests.
Major finding: Sixteen percent of HIV-associated Hodgkin lymphoma patients were untreated.
Data source: A review of 2,090 cases from the National Cancer Data Base.
Disclosures: Dr. Olszewski is supported by the American Society of Hematology Scholar Award and the American Cancer Society Research Scholar Award. Both authors reported having no conflicts of interest.
Microbead ban could impede nanomaterial development
The new law that will ban the sale or distribution of plastic microbeads in over-the-counter and personal care products beginning in July 2017 could have an unintended effect: a negative impact on the development and acceptance of micro- and nanotechnology–based medical and diagnostic products.
Environmentalists and others cheered the new law for its potentially protective effects on the environment and ultimately on public health. And the unavailability of such products is not expected to adversely affect consumers, as there are alternatives to the scrubs and other products that contain these microbeads – and manufacturers have started to reformulate their products that contain plastic microbeads.
However, the ban could potentially do more harm than good if the message consumers hear is that microtechnology – and, by extension, nanotechnology – is bad, according to Dr. Adam Friedman, director of translational research in the department of dermatology at George Washington University, Washington, who is a Dermatology News Board Member.
“What I’m most concerned about is the impact on public perception, that this ban infers that micro- and nanotechnology is inherently bad, and therefore, how it might impact approval of both over-the-counter and prescription medications that incorporate microscopic carriers,” said Dr. Friedman, who has a particular interest in nanotechnology. He explained that nanomaterials have enormous potential for helping to deliver drugs that are unstable, difficult to administer, or even toxic in their bulk form.
How such materials can be evaluated from a safety and efficacy standpoint in order to facilitate approval is currently under investigation, and negativity toward this field of research could hinder the progress of related research, he added, noting that “not all nano- and microtechnology is created equal.”
“This [ban] is about microplastics specifically,” not microspheres, -particles, or -beads, overall, he pointed out.
The basis of the Microbead-Free Waters Act of 2015 – which was signed into law by President Barack Obama on Dec. 28 after it sailed through Congress with an unusual level of bipartisan consensus among lawmakers regarding its importance – is that the plastic microbeads used in products such as facial scrubs and toothpaste pose a threat to marine life and ultimately to humans via the food chain.
Researchers have found that tiny microbeads – an estimated 11 billion daily – slip through wastewater treatment systems into the environment, where they appear to attract harmful chemicals that could make them toxic to marine life and ultimately to humans.
In fact, the biological dangers associated with microbeads upon which the ban was based are hypothetical, Dr. Friedman countered, noting that much of the argument against microbeads is based on an oft-cited publication that is actually a non-peer-reviewed editorial in support of a microbead ban (Environ Sci Technol. 2015;49:10759-61). The authors cited studies demonstrating the inability of current sanitation measures to effectively remove the microbeads from the water supply. They also observed that “the argument has been raised that there is not yet enough scientific evidence to support banning microbeads,” but added, “though there are gaps in our understanding of the precise impact of microbeads on aquatic ecosystems, this should not delay action.”
Indeed, there is reason to believe that microplastics could contain endocrine-disrupting chemicals linked with a host of intractable human diseases, and that they may otherwise pose a threat to human health – particularly by attracting and collecting pesticides, bisphenol A (BPA), and phthalates and binding to them, then entering the food chain, noted Deborah M. Kurrasch, Ph.D., of the department of medical genetics at the University of Calgary, Alberta, in an interview. However, there is no direct evidence of this, she agreed.
“Common sense says that might be the case, but there are no data to support that,” said Dr. Kurrasch, who is currently studying how environmental insults, including those from plastics and herbicides, affect the brain.
“There is lots of interest in plastics, and this ban is more of an environmental toxicology ban ... the leap to a human problem is very vague,” she commented.
In fact, the plastics in microbeads are generally considered “safe plastics,” as opposed to those containing BPA, for example, Dr. Kurrasch said. However, she added that she doesn’t think there is such a thing as a safe plastic.
“Ninety-one percent of plastics have been shown to leach compounds that affect estrogen signaling, and the fact that microbeads are made of plastic means they are likely to have some sort of effect down the road,” she said. “But the route of exposure? We just don’t know.”
Dr. Kurrasch believes the ban is a step in the right direction, and that it’s good that attention is being paid to protecting wildlife and humans from the potential harms of plastics.
Dr. Naissan O. Wesley, a dermatologist in private practice in Beverly Hills, noted that she, too is pleased about the concern regarding potential detrimental effects of microbeads.
“This is a huge step in skin care for products that are healthy for us and our environment,” she said, adding that while the quality of some products may be affected by the microbead ban, there are plenty of alternative scrubs, including sugar- or salt-based products.
“There are also other ways to achieve exfoliation, such as with things like the Clarisonic brush once a day, glycolic acid washes, Retin-A [tretinoin], chemical peels, microdermabrasion, or hydrofacials, and lasers,” Dr. Wesley, who is also a Dermatology News Board Member, noted. “Some women also do dermaplaning.”
Dr. Friedman also agreed with the importance of protecting the environment and the minimal impact the ban will have on products.
“I’m all for putting appropriate measures in place to prevent inappropriate exposures and damage to the natural setting. That said, I’m also for preserving the scientific method.”
Taking microbead-containing products off the shelf is not a life-altering process, Dr. Friedman said, noting that he doesn’t usually even recommend such products for patients. However, it will be expensive for the industry as it adapts to the microbead ban – and the money that will be spent is money that could have been applied to initiatives and efforts that may have had a bigger impact, he added.
“What frustrates me is that no opportunity was given to improve the technology,” he said, explaining that nano- and microparticles could be augmented to prevent them from entering the water system. For example, surface modifications applied to the microbeads could be used to increase aggregation under specific environmental conditions, and thereby prevent filtration failure. “There was a missed opportunity to engage industry and scientists alike to better understand how materials at this scale behave and how they can be manipulated for good,” he maintained.
“The ban opens the door to adversely impact more important technology that could be life altering. Now, we have an uphill battle.”
Dr. Friedman, Dr. Kurrasch, and Dr. Wesley reported having no relevant disclosures.
The new law that will ban the sale or distribution of plastic microbeads in over-the-counter and personal care products beginning in July 2017 could have an unintended effect: a negative impact on the development and acceptance of micro- and nanotechnology–based medical and diagnostic products.
Environmentalists and others cheered the new law for its potentially protective effects on the environment and ultimately on public health. And the unavailability of such products is not expected to adversely affect consumers, as there are alternatives to the scrubs and other products that contain these microbeads – and manufacturers have started to reformulate their products that contain plastic microbeads.
However, the ban could potentially do more harm than good if the message consumers hear is that microtechnology – and, by extension, nanotechnology – is bad, according to Dr. Adam Friedman, director of translational research in the department of dermatology at George Washington University, Washington, who is a Dermatology News Board Member.
“What I’m most concerned about is the impact on public perception, that this ban infers that micro- and nanotechnology is inherently bad, and therefore, how it might impact approval of both over-the-counter and prescription medications that incorporate microscopic carriers,” said Dr. Friedman, who has a particular interest in nanotechnology. He explained that nanomaterials have enormous potential for helping to deliver drugs that are unstable, difficult to administer, or even toxic in their bulk form.
How such materials can be evaluated from a safety and efficacy standpoint in order to facilitate approval is currently under investigation, and negativity toward this field of research could hinder the progress of related research, he added, noting that “not all nano- and microtechnology is created equal.”
“This [ban] is about microplastics specifically,” not microspheres, -particles, or -beads, overall, he pointed out.
The basis of the Microbead-Free Waters Act of 2015 – which was signed into law by President Barack Obama on Dec. 28 after it sailed through Congress with an unusual level of bipartisan consensus among lawmakers regarding its importance – is that the plastic microbeads used in products such as facial scrubs and toothpaste pose a threat to marine life and ultimately to humans via the food chain.
Researchers have found that tiny microbeads – an estimated 11 billion daily – slip through wastewater treatment systems into the environment, where they appear to attract harmful chemicals that could make them toxic to marine life and ultimately to humans.
In fact, the biological dangers associated with microbeads upon which the ban was based are hypothetical, Dr. Friedman countered, noting that much of the argument against microbeads is based on an oft-cited publication that is actually a non-peer-reviewed editorial in support of a microbead ban (Environ Sci Technol. 2015;49:10759-61). The authors cited studies demonstrating the inability of current sanitation measures to effectively remove the microbeads from the water supply. They also observed that “the argument has been raised that there is not yet enough scientific evidence to support banning microbeads,” but added, “though there are gaps in our understanding of the precise impact of microbeads on aquatic ecosystems, this should not delay action.”
Indeed, there is reason to believe that microplastics could contain endocrine-disrupting chemicals linked with a host of intractable human diseases, and that they may otherwise pose a threat to human health – particularly by attracting and collecting pesticides, bisphenol A (BPA), and phthalates and binding to them, then entering the food chain, noted Deborah M. Kurrasch, Ph.D., of the department of medical genetics at the University of Calgary, Alberta, in an interview. However, there is no direct evidence of this, she agreed.
“Common sense says that might be the case, but there are no data to support that,” said Dr. Kurrasch, who is currently studying how environmental insults, including those from plastics and herbicides, affect the brain.
“There is lots of interest in plastics, and this ban is more of an environmental toxicology ban ... the leap to a human problem is very vague,” she commented.
In fact, the plastics in microbeads are generally considered “safe plastics,” as opposed to those containing BPA, for example, Dr. Kurrasch said. However, she added that she doesn’t think there is such a thing as a safe plastic.
“Ninety-one percent of plastics have been shown to leach compounds that affect estrogen signaling, and the fact that microbeads are made of plastic means they are likely to have some sort of effect down the road,” she said. “But the route of exposure? We just don’t know.”
Dr. Kurrasch believes the ban is a step in the right direction, and that it’s good that attention is being paid to protecting wildlife and humans from the potential harms of plastics.
Dr. Naissan O. Wesley, a dermatologist in private practice in Beverly Hills, noted that she, too is pleased about the concern regarding potential detrimental effects of microbeads.
“This is a huge step in skin care for products that are healthy for us and our environment,” she said, adding that while the quality of some products may be affected by the microbead ban, there are plenty of alternative scrubs, including sugar- or salt-based products.
“There are also other ways to achieve exfoliation, such as with things like the Clarisonic brush once a day, glycolic acid washes, Retin-A [tretinoin], chemical peels, microdermabrasion, or hydrofacials, and lasers,” Dr. Wesley, who is also a Dermatology News Board Member, noted. “Some women also do dermaplaning.”
Dr. Friedman also agreed with the importance of protecting the environment and the minimal impact the ban will have on products.
“I’m all for putting appropriate measures in place to prevent inappropriate exposures and damage to the natural setting. That said, I’m also for preserving the scientific method.”
Taking microbead-containing products off the shelf is not a life-altering process, Dr. Friedman said, noting that he doesn’t usually even recommend such products for patients. However, it will be expensive for the industry as it adapts to the microbead ban – and the money that will be spent is money that could have been applied to initiatives and efforts that may have had a bigger impact, he added.
“What frustrates me is that no opportunity was given to improve the technology,” he said, explaining that nano- and microparticles could be augmented to prevent them from entering the water system. For example, surface modifications applied to the microbeads could be used to increase aggregation under specific environmental conditions, and thereby prevent filtration failure. “There was a missed opportunity to engage industry and scientists alike to better understand how materials at this scale behave and how they can be manipulated for good,” he maintained.
“The ban opens the door to adversely impact more important technology that could be life altering. Now, we have an uphill battle.”
Dr. Friedman, Dr. Kurrasch, and Dr. Wesley reported having no relevant disclosures.
The new law that will ban the sale or distribution of plastic microbeads in over-the-counter and personal care products beginning in July 2017 could have an unintended effect: a negative impact on the development and acceptance of micro- and nanotechnology–based medical and diagnostic products.
Environmentalists and others cheered the new law for its potentially protective effects on the environment and ultimately on public health. And the unavailability of such products is not expected to adversely affect consumers, as there are alternatives to the scrubs and other products that contain these microbeads – and manufacturers have started to reformulate their products that contain plastic microbeads.
However, the ban could potentially do more harm than good if the message consumers hear is that microtechnology – and, by extension, nanotechnology – is bad, according to Dr. Adam Friedman, director of translational research in the department of dermatology at George Washington University, Washington, who is a Dermatology News Board Member.
“What I’m most concerned about is the impact on public perception, that this ban infers that micro- and nanotechnology is inherently bad, and therefore, how it might impact approval of both over-the-counter and prescription medications that incorporate microscopic carriers,” said Dr. Friedman, who has a particular interest in nanotechnology. He explained that nanomaterials have enormous potential for helping to deliver drugs that are unstable, difficult to administer, or even toxic in their bulk form.
How such materials can be evaluated from a safety and efficacy standpoint in order to facilitate approval is currently under investigation, and negativity toward this field of research could hinder the progress of related research, he added, noting that “not all nano- and microtechnology is created equal.”
“This [ban] is about microplastics specifically,” not microspheres, -particles, or -beads, overall, he pointed out.
The basis of the Microbead-Free Waters Act of 2015 – which was signed into law by President Barack Obama on Dec. 28 after it sailed through Congress with an unusual level of bipartisan consensus among lawmakers regarding its importance – is that the plastic microbeads used in products such as facial scrubs and toothpaste pose a threat to marine life and ultimately to humans via the food chain.
Researchers have found that tiny microbeads – an estimated 11 billion daily – slip through wastewater treatment systems into the environment, where they appear to attract harmful chemicals that could make them toxic to marine life and ultimately to humans.
In fact, the biological dangers associated with microbeads upon which the ban was based are hypothetical, Dr. Friedman countered, noting that much of the argument against microbeads is based on an oft-cited publication that is actually a non-peer-reviewed editorial in support of a microbead ban (Environ Sci Technol. 2015;49:10759-61). The authors cited studies demonstrating the inability of current sanitation measures to effectively remove the microbeads from the water supply. They also observed that “the argument has been raised that there is not yet enough scientific evidence to support banning microbeads,” but added, “though there are gaps in our understanding of the precise impact of microbeads on aquatic ecosystems, this should not delay action.”
Indeed, there is reason to believe that microplastics could contain endocrine-disrupting chemicals linked with a host of intractable human diseases, and that they may otherwise pose a threat to human health – particularly by attracting and collecting pesticides, bisphenol A (BPA), and phthalates and binding to them, then entering the food chain, noted Deborah M. Kurrasch, Ph.D., of the department of medical genetics at the University of Calgary, Alberta, in an interview. However, there is no direct evidence of this, she agreed.
“Common sense says that might be the case, but there are no data to support that,” said Dr. Kurrasch, who is currently studying how environmental insults, including those from plastics and herbicides, affect the brain.
“There is lots of interest in plastics, and this ban is more of an environmental toxicology ban ... the leap to a human problem is very vague,” she commented.
In fact, the plastics in microbeads are generally considered “safe plastics,” as opposed to those containing BPA, for example, Dr. Kurrasch said. However, she added that she doesn’t think there is such a thing as a safe plastic.
“Ninety-one percent of plastics have been shown to leach compounds that affect estrogen signaling, and the fact that microbeads are made of plastic means they are likely to have some sort of effect down the road,” she said. “But the route of exposure? We just don’t know.”
Dr. Kurrasch believes the ban is a step in the right direction, and that it’s good that attention is being paid to protecting wildlife and humans from the potential harms of plastics.
Dr. Naissan O. Wesley, a dermatologist in private practice in Beverly Hills, noted that she, too is pleased about the concern regarding potential detrimental effects of microbeads.
“This is a huge step in skin care for products that are healthy for us and our environment,” she said, adding that while the quality of some products may be affected by the microbead ban, there are plenty of alternative scrubs, including sugar- or salt-based products.
“There are also other ways to achieve exfoliation, such as with things like the Clarisonic brush once a day, glycolic acid washes, Retin-A [tretinoin], chemical peels, microdermabrasion, or hydrofacials, and lasers,” Dr. Wesley, who is also a Dermatology News Board Member, noted. “Some women also do dermaplaning.”
Dr. Friedman also agreed with the importance of protecting the environment and the minimal impact the ban will have on products.
“I’m all for putting appropriate measures in place to prevent inappropriate exposures and damage to the natural setting. That said, I’m also for preserving the scientific method.”
Taking microbead-containing products off the shelf is not a life-altering process, Dr. Friedman said, noting that he doesn’t usually even recommend such products for patients. However, it will be expensive for the industry as it adapts to the microbead ban – and the money that will be spent is money that could have been applied to initiatives and efforts that may have had a bigger impact, he added.
“What frustrates me is that no opportunity was given to improve the technology,” he said, explaining that nano- and microparticles could be augmented to prevent them from entering the water system. For example, surface modifications applied to the microbeads could be used to increase aggregation under specific environmental conditions, and thereby prevent filtration failure. “There was a missed opportunity to engage industry and scientists alike to better understand how materials at this scale behave and how they can be manipulated for good,” he maintained.
“The ban opens the door to adversely impact more important technology that could be life altering. Now, we have an uphill battle.”
Dr. Friedman, Dr. Kurrasch, and Dr. Wesley reported having no relevant disclosures.
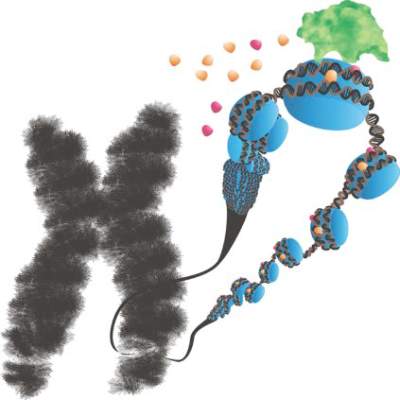
Blocking two targets boosted fetal hemoglobin expression
Two distinct proteins appear to control the switch from fetal to adult globin, based on studies performed in a humanized mouse model and human cells. The findings suggest therapies that target both proteins might induce a fetal-type globin state, which could prove therapeutically useful in individuals with human hemoglobinopathies such as sickle cell disease and thalassemia.
The leukemia/lymphoma-related factor (LRF) and B-cell lymphoma/leukemia 11A (BCL11A) are independently involved in the switch, and blocking their production may turn on fetal globin expression, Takeshi Masuda, Ph.D., of Brigham and Women’s Hospital and Harvard Medical School, Boston, and his colleagues report (SCIENCE. 2015 Jan 15;351[6270]:285-9)
Using a humanized mouse model, the researchers knocked out the ZBTB7A gene, which is responsible for producing LRF. This action boosted the expression of genes that control fetal but not adult-type hemoglobin. Knocking out the gene in human cells also resulted in an increase in fetal hemoglobin proteins.
The researchers then examined BCL11A, which is involved with fetal hemoglobin but does not suppress it. When genes were knocked out for both ZBTB7A and BCL11A in the mice, fetal hemoglobin represented a 91%-94% greater percentage of total hemoglobin than when either gene alone was knocked out.
The research was supported by awards and/or grants from the National Institute of Diabetes and Digestive and Kidney Disease, the Doris Duke Charitable Foundation, the National Institutes of Health, and the American Society of Hematology. Dr. Masuda, along with two other study authors, is a contributor to a patent application filed on behalf of Brigham and Women’s Hospital related to therapeutic targeting of the pathways.
Click here to read the study at Science.
Two distinct proteins appear to control the switch from fetal to adult globin, based on studies performed in a humanized mouse model and human cells. The findings suggest therapies that target both proteins might induce a fetal-type globin state, which could prove therapeutically useful in individuals with human hemoglobinopathies such as sickle cell disease and thalassemia.
The leukemia/lymphoma-related factor (LRF) and B-cell lymphoma/leukemia 11A (BCL11A) are independently involved in the switch, and blocking their production may turn on fetal globin expression, Takeshi Masuda, Ph.D., of Brigham and Women’s Hospital and Harvard Medical School, Boston, and his colleagues report (SCIENCE. 2015 Jan 15;351[6270]:285-9)
Using a humanized mouse model, the researchers knocked out the ZBTB7A gene, which is responsible for producing LRF. This action boosted the expression of genes that control fetal but not adult-type hemoglobin. Knocking out the gene in human cells also resulted in an increase in fetal hemoglobin proteins.
The researchers then examined BCL11A, which is involved with fetal hemoglobin but does not suppress it. When genes were knocked out for both ZBTB7A and BCL11A in the mice, fetal hemoglobin represented a 91%-94% greater percentage of total hemoglobin than when either gene alone was knocked out.
The research was supported by awards and/or grants from the National Institute of Diabetes and Digestive and Kidney Disease, the Doris Duke Charitable Foundation, the National Institutes of Health, and the American Society of Hematology. Dr. Masuda, along with two other study authors, is a contributor to a patent application filed on behalf of Brigham and Women’s Hospital related to therapeutic targeting of the pathways.
Click here to read the study at Science.
Two distinct proteins appear to control the switch from fetal to adult globin, based on studies performed in a humanized mouse model and human cells. The findings suggest therapies that target both proteins might induce a fetal-type globin state, which could prove therapeutically useful in individuals with human hemoglobinopathies such as sickle cell disease and thalassemia.
The leukemia/lymphoma-related factor (LRF) and B-cell lymphoma/leukemia 11A (BCL11A) are independently involved in the switch, and blocking their production may turn on fetal globin expression, Takeshi Masuda, Ph.D., of Brigham and Women’s Hospital and Harvard Medical School, Boston, and his colleagues report (SCIENCE. 2015 Jan 15;351[6270]:285-9)
Using a humanized mouse model, the researchers knocked out the ZBTB7A gene, which is responsible for producing LRF. This action boosted the expression of genes that control fetal but not adult-type hemoglobin. Knocking out the gene in human cells also resulted in an increase in fetal hemoglobin proteins.
The researchers then examined BCL11A, which is involved with fetal hemoglobin but does not suppress it. When genes were knocked out for both ZBTB7A and BCL11A in the mice, fetal hemoglobin represented a 91%-94% greater percentage of total hemoglobin than when either gene alone was knocked out.
The research was supported by awards and/or grants from the National Institute of Diabetes and Digestive and Kidney Disease, the Doris Duke Charitable Foundation, the National Institutes of Health, and the American Society of Hematology. Dr. Masuda, along with two other study authors, is a contributor to a patent application filed on behalf of Brigham and Women’s Hospital related to therapeutic targeting of the pathways.
Click here to read the study at Science.
FROM SCIENCE
Mavoglurant disappoints in fragile X trials
The selective metabotropic glutamate receptor 5 (mGluR5) antagonist mavoglurant failed to improve behavioral symptoms in patients with fragile X syndrome in two phase IIb, multicenter, randomized, placebo-controlled studies.
A post hoc analysis of a prior phase II trial suggested that mavoglurant improved such symptoms in fragile X syndrome patients with completely methylated FMR1 genes, and the current trials were designed to confirm the findings in 175 adults and 139 adolescents, respectively.
But no change in maladaptive behavior, as measured using the Aberrant Behavior Checklist–Community Edition and fragile X syndrome-specific algorithm (ABC-Cfx), was seen 12 weeks after treatment with either 25, 50, or 100 mg of mavoglurant or placebo twice daily for 12 weeks, Dr. Elizabeth Berry-Kravis of Rush University Medical Center, Chicago, and her colleagues reported online Jan. 13 in Science Translational Medicine.
In completely methylated adults, the change in ABC-Cfx score at 12 weeks was similar with placebo and 25 mg mavoglurant (–11.4 and –14.3), and nearly unchanged with 50 and 100 mg of mavoglurant (1.8, -1.8). In adolescents, the corresponding changes were –9.4, –11.8, –3.4, and 8.6 (a statistically significant deterioration vs. placebo). Outcomes were similar in partially methylated adults and adolescents.
Since the primary efficacy endpoint of improvement in behavioral symptoms was not met, the mGluR theory of fragile X syndrome as suggested by animal models – namely that the absence of fragile X mental retardation protein can cause overactivation of mGluR signaling, contributing to the features of the fragile X syndrome phenotype – could not be confirmed, nor could the ability of the methylation state of the FMR1 promoter to predict mavoglurant efficacy, the investigators wrote (Sci Transl Med. 2016 Jan. 13. doi:10.1126/scitranslmed.aab4109).
“One of the most plausible explanations for the negative results is that the mGluR theory on which the studies were based may not be valid or may manifest itself differently in humans, compared to rodents,” they wrote, noting that a combination of targeted treatments may be needed for optimal treatment of fragile X syndrome. Further, preclinical trials suggested that treatment at a younger age and for longer durations might have a greater effect; this approach warrants evaluation in a future trial, they wrote.
This study was supported by Novartis Pharma AG. Dr. Berry-Kravis has served on the Novartis Fragile X Advisory Board and has consulted for Novartis. Other authors also served on the board and have consulted for Novartis and/or Roche, or are employed by Novartis.
The findings of Dr. Berry-Kravis et al. yield important lessons for clinical trial design. Rather than concluding that the negative findings prove that a treatment is ineffective or that presumed underlying pathophysiological mechanisms are invalid, trials like this one provide an opportunity to reflect on clinical trial design and implementation, as well as the underlying mechanistic motivations, and provide lessons for treatment studies of neurodevelopmental disorders in general. This trial highlights key themes to consider in clinical trials, including choice of treatment group, target engagement, selection of outcome measures, and the impact of the placebo effect.
We stand at a therapeutic frontier, with much more to learn. While the result of negative trials are disappointing, they also can be crucial for refining hypotheses and encouraging dialogue to accelerate the process of bringing effective treatments to our patients.
Dr. Shafali S. Jeste and Dr. Daniel H. Geschwind are with the University of California at Los Angeles. These comments are adapted from an accompanying editorial (Sci Transl Med. 2016 Jan 13. doi:10.1126/scitranslmed.aab4109.). The authors did not report financial disclosures.
The findings of Dr. Berry-Kravis et al. yield important lessons for clinical trial design. Rather than concluding that the negative findings prove that a treatment is ineffective or that presumed underlying pathophysiological mechanisms are invalid, trials like this one provide an opportunity to reflect on clinical trial design and implementation, as well as the underlying mechanistic motivations, and provide lessons for treatment studies of neurodevelopmental disorders in general. This trial highlights key themes to consider in clinical trials, including choice of treatment group, target engagement, selection of outcome measures, and the impact of the placebo effect.
We stand at a therapeutic frontier, with much more to learn. While the result of negative trials are disappointing, they also can be crucial for refining hypotheses and encouraging dialogue to accelerate the process of bringing effective treatments to our patients.
Dr. Shafali S. Jeste and Dr. Daniel H. Geschwind are with the University of California at Los Angeles. These comments are adapted from an accompanying editorial (Sci Transl Med. 2016 Jan 13. doi:10.1126/scitranslmed.aab4109.). The authors did not report financial disclosures.
The findings of Dr. Berry-Kravis et al. yield important lessons for clinical trial design. Rather than concluding that the negative findings prove that a treatment is ineffective or that presumed underlying pathophysiological mechanisms are invalid, trials like this one provide an opportunity to reflect on clinical trial design and implementation, as well as the underlying mechanistic motivations, and provide lessons for treatment studies of neurodevelopmental disorders in general. This trial highlights key themes to consider in clinical trials, including choice of treatment group, target engagement, selection of outcome measures, and the impact of the placebo effect.
We stand at a therapeutic frontier, with much more to learn. While the result of negative trials are disappointing, they also can be crucial for refining hypotheses and encouraging dialogue to accelerate the process of bringing effective treatments to our patients.
Dr. Shafali S. Jeste and Dr. Daniel H. Geschwind are with the University of California at Los Angeles. These comments are adapted from an accompanying editorial (Sci Transl Med. 2016 Jan 13. doi:10.1126/scitranslmed.aab4109.). The authors did not report financial disclosures.
The selective metabotropic glutamate receptor 5 (mGluR5) antagonist mavoglurant failed to improve behavioral symptoms in patients with fragile X syndrome in two phase IIb, multicenter, randomized, placebo-controlled studies.
A post hoc analysis of a prior phase II trial suggested that mavoglurant improved such symptoms in fragile X syndrome patients with completely methylated FMR1 genes, and the current trials were designed to confirm the findings in 175 adults and 139 adolescents, respectively.
But no change in maladaptive behavior, as measured using the Aberrant Behavior Checklist–Community Edition and fragile X syndrome-specific algorithm (ABC-Cfx), was seen 12 weeks after treatment with either 25, 50, or 100 mg of mavoglurant or placebo twice daily for 12 weeks, Dr. Elizabeth Berry-Kravis of Rush University Medical Center, Chicago, and her colleagues reported online Jan. 13 in Science Translational Medicine.
In completely methylated adults, the change in ABC-Cfx score at 12 weeks was similar with placebo and 25 mg mavoglurant (–11.4 and –14.3), and nearly unchanged with 50 and 100 mg of mavoglurant (1.8, -1.8). In adolescents, the corresponding changes were –9.4, –11.8, –3.4, and 8.6 (a statistically significant deterioration vs. placebo). Outcomes were similar in partially methylated adults and adolescents.
Since the primary efficacy endpoint of improvement in behavioral symptoms was not met, the mGluR theory of fragile X syndrome as suggested by animal models – namely that the absence of fragile X mental retardation protein can cause overactivation of mGluR signaling, contributing to the features of the fragile X syndrome phenotype – could not be confirmed, nor could the ability of the methylation state of the FMR1 promoter to predict mavoglurant efficacy, the investigators wrote (Sci Transl Med. 2016 Jan. 13. doi:10.1126/scitranslmed.aab4109).
“One of the most plausible explanations for the negative results is that the mGluR theory on which the studies were based may not be valid or may manifest itself differently in humans, compared to rodents,” they wrote, noting that a combination of targeted treatments may be needed for optimal treatment of fragile X syndrome. Further, preclinical trials suggested that treatment at a younger age and for longer durations might have a greater effect; this approach warrants evaluation in a future trial, they wrote.
This study was supported by Novartis Pharma AG. Dr. Berry-Kravis has served on the Novartis Fragile X Advisory Board and has consulted for Novartis. Other authors also served on the board and have consulted for Novartis and/or Roche, or are employed by Novartis.
The selective metabotropic glutamate receptor 5 (mGluR5) antagonist mavoglurant failed to improve behavioral symptoms in patients with fragile X syndrome in two phase IIb, multicenter, randomized, placebo-controlled studies.
A post hoc analysis of a prior phase II trial suggested that mavoglurant improved such symptoms in fragile X syndrome patients with completely methylated FMR1 genes, and the current trials were designed to confirm the findings in 175 adults and 139 adolescents, respectively.
But no change in maladaptive behavior, as measured using the Aberrant Behavior Checklist–Community Edition and fragile X syndrome-specific algorithm (ABC-Cfx), was seen 12 weeks after treatment with either 25, 50, or 100 mg of mavoglurant or placebo twice daily for 12 weeks, Dr. Elizabeth Berry-Kravis of Rush University Medical Center, Chicago, and her colleagues reported online Jan. 13 in Science Translational Medicine.
In completely methylated adults, the change in ABC-Cfx score at 12 weeks was similar with placebo and 25 mg mavoglurant (–11.4 and –14.3), and nearly unchanged with 50 and 100 mg of mavoglurant (1.8, -1.8). In adolescents, the corresponding changes were –9.4, –11.8, –3.4, and 8.6 (a statistically significant deterioration vs. placebo). Outcomes were similar in partially methylated adults and adolescents.
Since the primary efficacy endpoint of improvement in behavioral symptoms was not met, the mGluR theory of fragile X syndrome as suggested by animal models – namely that the absence of fragile X mental retardation protein can cause overactivation of mGluR signaling, contributing to the features of the fragile X syndrome phenotype – could not be confirmed, nor could the ability of the methylation state of the FMR1 promoter to predict mavoglurant efficacy, the investigators wrote (Sci Transl Med. 2016 Jan. 13. doi:10.1126/scitranslmed.aab4109).
“One of the most plausible explanations for the negative results is that the mGluR theory on which the studies were based may not be valid or may manifest itself differently in humans, compared to rodents,” they wrote, noting that a combination of targeted treatments may be needed for optimal treatment of fragile X syndrome. Further, preclinical trials suggested that treatment at a younger age and for longer durations might have a greater effect; this approach warrants evaluation in a future trial, they wrote.
This study was supported by Novartis Pharma AG. Dr. Berry-Kravis has served on the Novartis Fragile X Advisory Board and has consulted for Novartis. Other authors also served on the board and have consulted for Novartis and/or Roche, or are employed by Novartis.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: The selective metabotropic glutamate receptor 5 (mGluR5) antagonist mavoglurant failed to improve behavioral symptoms in patients with fragile X syndrome in two phase 2b, multicenter, randomized, placebo-controlled studies.
Major finding: Change in ABC-Cfx score in completely methylated adults was –11.4, –14.3, 1.8 and –1.8 with placebo, 25, 50 and 100 mg of mavoglurant, respectively. In adolescents, the corresponding changes were –9.4, –11.8, –3.4, and 8.6.
Data source: A phase 2b randomized placebo-controlled trial involving 175 adults and 139 adolescents.
Disclosures: This study was supported by Novartis Pharma AG. Dr. Berry-Kravis has served on the Novartis Fragile X Advisory Board and has consulted for Novartis. Other authors also served on the board and have consulted for Novartis and/or Roche, or are employed by Novartis.
USPSTF Supports Mammography Start at Age 50
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
FROM ANNALS OF INTERNAL MEDICINE
USPSTF supports mammography start at age 50
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
Women aged 50-74 years should undergo biennial screening mammography, and the decision to screen before age 50 should be individualized, according to a final recommendation from the U.S. Preventive Services Task Force.
The recommendation statement, published Jan. 11 in Annals of Internal Medicine, is based on a comprehensive review of data since 2009, when the USPSTF last released breast cancer screening recommendations, and follows a public comment period in early 2015.
“The task force continues to find that the benefit of mammography increases with age, and recommends biennial screening in women ages 50 to 74. Mammography can also be effective for women in their 40s, but the benefits are less and the harms potentially greater. The decision by women to start screening in their 40s should be an individual one, made in partnership with a doctor,” according to a statement from the USPSTF.
The new recommendations will not affect private insurance coverage of mammography without cost sharing as outlined in the Affordable Care Act. As part of a spending bill enacted in December 2015, Congress passed a provision placing a 2-year moratorium on the use of any new USPSTF recommendations related to breast cancer screening to determine coverage, effectively preserving mammography coverage without cost sharing for women aged 40 years and older.
Recommendations by age
The latest USPSTF recommendations by age state that women aged 40-49 years should base their screening decision on personal values, preferences, and health history; women with a family history of breast cancer may benefit more than average-risk women by beginning screening before age 50. This is a C recommendation, indicating “moderate certainty that net benefit is small.”
The recommendation for biennial screening of those aged 50-74 is a B recommendation indicating “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial,” Dr. Albert L. Siu, task force chair, reported on behalf of the USPSTF (Ann Intern Med. 2016;164:279-96. doi: 10.7326/M15-2886).
The task force found inadequate evidence to recommend for or against screening those aged 75 and older.
Final evidence documents, including a systematic review of data on the harms associated with breast cancer screening and a modeling study of the benefits and harms associated with different screening strategies, are published along with the recommendation statement.
The USPSTF did not make a recommendations about the use of digital breast tomosynthesis as a primary screening method for breast cancer, noting that the current evidence is insufficient. Evidence also was insufficient to make a recommendation on the benefits and harms of adjunctive screening for breast cancer using breast ultrasonography, magnetic resonance imaging, digital breast tomosynthesis, or other methods in women with dense breasts who had a negative screening mammogram.
Dousing the ‘firestorm’
In an editorial penned by Annals of Internal Medicine Editor-in-Chief and Senior Vice President of the American College of Physicians Christine Laine and her colleagues, they urged a dousing of the “firestorm around breast cancer screening.”
That firestorm was ignited with the 2009 USPSTF breast cancer screening recommendation and was rekindled when the current recommendation was presented in draft form in 2015.
However, “the USPSTF did a difficult job well” and based its recommendations on an important understanding of the updated evidence, as well as potential harms and tradeoffs of different screening strategies, the authors wrote.
“When the USPSTF posted its draft recommendations for comment, it noted, ‘Women deserve to be aware of what the science says so they can make the best choice for themselves, together with their doctor.’ We could not agree more. Let’s douse the flames and clear the smoke so that we can clearly see what the evidence shows and where we need to focus efforts to fill gaps in our knowledge so that women, along with their health care providers, can make the best decision to reduce their risk for breast cancer–related morbidity and mortality,” they wrote (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-2978).
ACOG supports screening at 40
The American College of Obstetricians and Gynecologists is standing by its recommendation of annual mammograms beginning at age 40 and continues to support use of clinical breast examinations. In a Jan. 11 statement, Dr. Mark S. DeFrancesco, ACOG president, said that “evidence and experience have shown that early detection can lead to improved outcomes in women diagnosed with breast cancer.”
The organization similarly stood by its recommendation in October 2015, when the American Cancer Society released recommendations for annual screening mammography for asymptomatic women at average risk for breast cancer beginning at age 45 years, with a transition to biennial screening mammography beginning at age 55 (JAMA. 2015;314[15]:1599-1614. doi: 10.1001/jama.2015.12783).
ACOG also supports the omnibus legislation passed by Congress in December that provides 2 years of no-copay coverage of breast cancer screening after age 40 via a moratorium on new breast cancer screening recommendations to allow time for additional research, an ACOG spokesperson said in an interview.
“ACOG strongly supports shared decision-making between doctor and patient, and in the case of screening for breast cancer, it is essential,” Dr. DeFrancesco said. “Given the differences among current organizational recommendations on breast cancer screening, we recognize that there may be confusion among women about when they should begin screening for breast cancer. ACOG encourages women to discuss this with their doctor, including concerns such as family history of cancer, risk factors such as overweight, and their own personal experiences with breast cancer. Moreover, it is essential that physicians counsel women about the potential consequences of mammography, including false positives.”
ACOG will convene a consensus conference later in January “with the intent to develop a consistent set of uniform guidelines for breast cancer screening that can be implemented nationwide” in an effort to “avoid the confusion that currently exists among the women we treat,” according to the statement.
The issue of divergence – and convergence – among various guidelines was the topic of another editorial published in conjunction with the USPSTF recommendations. In that article, task force chair Dr. Siu and his colleagues acknowledged that disagreements exist but stressed that “it would be a disservice to women and their clinicians if these disagreements obscured a strong emerging convergence among groups who have recently issued evidence-based guidelines” (Ann Intern Med. 2016 Jan 11. doi:10.7326/M15-3065).
The operations of the USPSTF are supported by the Agency for Healthcare Research and Quality. One of the members of the USPSTF reported receiving past grants and contracts from the National Cancer Institute and the Centers for Disease Control and Prevention.
FROM ANNALS OF INTERNAL MEDICINE