User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
FDA approves ustekinumab (Stelara) for adult Crohn’s disease
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
The drug is the first biologic agent to target interleukin (IL)-12 and IL-23 in Crohn’s disease, and its approval was based on findings in more than 1,300 patients across three Janssen Biotech–sponsored phase III studies (UNITI-1, UNITI-2, IM-UNITI) demonstrating its efficacy with respect to clinical response and remission rates.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
The drug is the first biologic agent to target interleukin (IL)-12 and IL-23 in Crohn’s disease, and its approval was based on findings in more than 1,300 patients across three Janssen Biotech–sponsored phase III studies (UNITI-1, UNITI-2, IM-UNITI) demonstrating its efficacy with respect to clinical response and remission rates.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
The drug is the first biologic agent to target interleukin (IL)-12 and IL-23 in Crohn’s disease, and its approval was based on findings in more than 1,300 patients across three Janssen Biotech–sponsored phase III studies (UNITI-1, UNITI-2, IM-UNITI) demonstrating its efficacy with respect to clinical response and remission rates.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
New-onset pediatric AD phenotype differs from adult AD
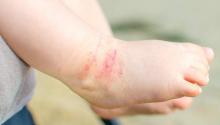
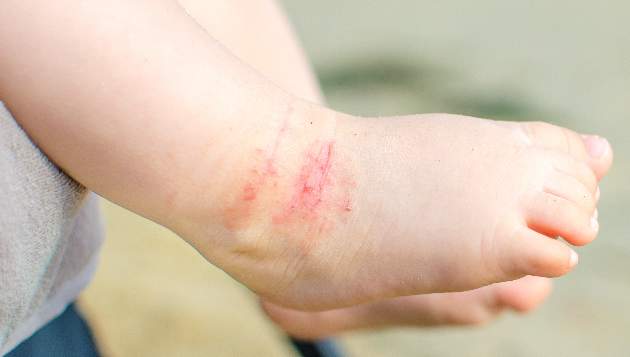
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Key clinical point: The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, which has important therapeutic implications, according to a study of biopsy findings in infants and children.
Major finding: Early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD.
Data source: An analysis of biopsies from 19 children with AD.
Disclosures: This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
No link between vasectomy, prostate cancer in U.S. cohort
Vasectomy does not appear to be associated with prostate cancer mortality or prostate cancer incidence, according to findings from a large U.S. prospective cohort of men aged 40 years or older.
Prostate cancer mortality was examined in the overall Cancer Prevention Study II (CPS-II) cohort of 363,726 men, including 7,451 who died as a result of prostate cancer between 1982 and 2012, and no association was found with vasectomy (hazard ratio, 1.01).
Overall and high-grade prostate cancer incidence rates were examined among 66,542 men from the CPS-II Nutrition cohort subgroup, including 9,133 who were diagnosed with prostate cancer between 1991 and 2011, and again no associations were found with vasectomy (HR, 1.02 and 0.91, respectively), Eric J. Jacobs, PhD, and his colleagues at the American Cancer Society report (J Clin Oncol. 2016 Sep 19. doi: 10.1200/JCO.2015.66.2361).
Results were similar after adjusting for years since vasectomy and in analyses restricted to men aged 50 years and older, the investigators noted.
Findings from prior, smaller studies have been conflicting, but the current study – the largest-known study to date to examine the association between vasectomy and prostate cancer, according to the authors – provides “some reassurance that vasectomy is unlikely to meaningfully increase risk of prostate cancer,” they wrote.
The authors reported having no disclosures.
Vasectomy does not appear to be associated with prostate cancer mortality or prostate cancer incidence, according to findings from a large U.S. prospective cohort of men aged 40 years or older.
Prostate cancer mortality was examined in the overall Cancer Prevention Study II (CPS-II) cohort of 363,726 men, including 7,451 who died as a result of prostate cancer between 1982 and 2012, and no association was found with vasectomy (hazard ratio, 1.01).
Overall and high-grade prostate cancer incidence rates were examined among 66,542 men from the CPS-II Nutrition cohort subgroup, including 9,133 who were diagnosed with prostate cancer between 1991 and 2011, and again no associations were found with vasectomy (HR, 1.02 and 0.91, respectively), Eric J. Jacobs, PhD, and his colleagues at the American Cancer Society report (J Clin Oncol. 2016 Sep 19. doi: 10.1200/JCO.2015.66.2361).
Results were similar after adjusting for years since vasectomy and in analyses restricted to men aged 50 years and older, the investigators noted.
Findings from prior, smaller studies have been conflicting, but the current study – the largest-known study to date to examine the association between vasectomy and prostate cancer, according to the authors – provides “some reassurance that vasectomy is unlikely to meaningfully increase risk of prostate cancer,” they wrote.
The authors reported having no disclosures.
Vasectomy does not appear to be associated with prostate cancer mortality or prostate cancer incidence, according to findings from a large U.S. prospective cohort of men aged 40 years or older.
Prostate cancer mortality was examined in the overall Cancer Prevention Study II (CPS-II) cohort of 363,726 men, including 7,451 who died as a result of prostate cancer between 1982 and 2012, and no association was found with vasectomy (hazard ratio, 1.01).
Overall and high-grade prostate cancer incidence rates were examined among 66,542 men from the CPS-II Nutrition cohort subgroup, including 9,133 who were diagnosed with prostate cancer between 1991 and 2011, and again no associations were found with vasectomy (HR, 1.02 and 0.91, respectively), Eric J. Jacobs, PhD, and his colleagues at the American Cancer Society report (J Clin Oncol. 2016 Sep 19. doi: 10.1200/JCO.2015.66.2361).
Results were similar after adjusting for years since vasectomy and in analyses restricted to men aged 50 years and older, the investigators noted.
Findings from prior, smaller studies have been conflicting, but the current study – the largest-known study to date to examine the association between vasectomy and prostate cancer, according to the authors – provides “some reassurance that vasectomy is unlikely to meaningfully increase risk of prostate cancer,” they wrote.
The authors reported having no disclosures.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Vasectomy does not appear to be associated with prostate cancer mortality or prostate cancer incidence, according to findings from a large U.S. prospective cohort of men aged 40 years or older.
Major finding: No association between vasectomy and prostate cancer mortality (HR, 1.01), or between vasectomy and overall and high-grade prostate cancer incidence (HR, 1.02 and 0.91, respectively).
Data source: The prospective CPS II cohort (363,726 men) and CPS-II Nutrition Cohort (66,542 men).
Disclosures: The authors reported having no disclosures.
Biologic mesh for ventral hernia repair compared for recurrence, cost
The porcine acellular dermal mesh product Strattice was associated with significantly lower odds of hernia recurrence, compared with several other biologic mesh products, in a study of 223 patients who underwent open ventral hernia repair.
Prospective operative outcomes data from a tertiary referral hernia center showed that at a mean follow-up of 18.2 months, the rate of hernia recurrence was 35% in 40 patients who were treated with Alloderm (LifeCell Corporation), 34.5% in 23 patients treated with AlloMax (Bard/Davol), 37.1% in 70 patients treated with FlexHD (Ethicon), and 59.1% in 22 patients treated with Xenmatrix (Bard/Davol), compared with 14.7% in 68 patients treated with Strattice (LifeCell Corporation). Alloderm, AlloMax, and FlexHD are all human acellular dermal mesh products, and Strattice and Xenmatrix are both porcine acellular dermal mesh products, Ciara R. Huntington, MD, and her colleagues at the Carolinas Medical Center in Charlotte, N.C., reported.

|
| Photo courtesy Acelity. STRATTICETM Reconstructive Tissue Matrix |
After multivariate analysis to adjust for factors such as comorbidities, hernia size, and intraoperative techniques, the odds ratios for recurrence with each product as compared with Strattice were 2.4 with Alloderm, 2.9 with FlexHD, 3.4 with AlloMax, and 7.8 with Xenmatrix. The odds for recurrence were significantly greater with all except Alloderm, the investigators said (Surgery. 2016. doi: 10.1016/j.surg.2016.07.008).
The significant differences between the two porcine acellular dermal meshes (Xenmatrix and Strattice) may reflect variation in tissue processing and design in biomesh engineering, they noted.
Study subjects were adults with a mean age of 57.7 years and mean body mass index of 34.8 kg/m2. Overall, 9.8% had an American Society of Anesthesiology classification of 4, 54.6% had a classification of 3, and 35.6% had a classification of 1 or 2. Average operative time was 241 minutes with estimated blood loss of 202 mL.
Average hernia defect size was 257 cm2, with average mesh size of 384 cm2.
“Component separation was performed in 47.5% of cases, and abdomen was left open prior to definitive closure in 10.7%. Biologic mesh was used to bridge fascial defects in 19.6% of cases. The mesh was placed in the preperitoneal space in 38.2% of cases,” the investigators wrote, noting that a concomitant procedure was performed in 82% of cases.
Sepsis developed in 6.7% of patients, 36.3% had a wound infection, and 24.3% required a negative pressure dressing for healing. The inpatient mortality rate was 1.4%.
However, mesh infections requiring explantation occurred in less than 1% of cases.
On adjusted analysis, Xenmatrix was the most expensive mesh and AlloMax was the least expensive (mean of $59,122 and $22,304, respectively). Strattice costs averaged $40,490.
Ventral hernia repair (VHR) is a common operation, with about 350,000 performed each year. Rates of postoperative wound infection and hernia recurrence vary widely, but may be improved with appropriate mesh selection. However, prospective data to guide selection are lacking, the investigators said.
“The great number of meshes available for use complicates the debate surrounding the best timing and use of biologic mesh in VHR, and the search for the better mesh for use in the abdominal wall reconstruction continues. Biologic mesh usually is reserved for the patients at the highest risk for developing a postoperative wound complication, and although there is a current dearth of high-level evidence supporting its use, this report confirms that complications are low despite obvious surgical complexity presented herein,” they wrote.
The findings of this study – the largest report of outcomes with biologic mesh in ventral hernia repair to date, according to the authors – support the safety of using biologic mesh in high-risk patients, they said.
They noted, however, that the study may still be underpowered to make final clinical decisions.
“Although our study provides useful information to the practicing surgeon, there is much work to be done regarding the selection of biologic mesh,” they wrote, adding that while “a well-performing biologic mesh should be in the toolkit of every general surgeon who may face complex abdominal walls requiring reconstruction in patients that are at high risk for a postoperative wound complication,” additional research is necessary to further clarify the role of biologic mesh in these operations.
Dr. Huntington reported having no disclosures. Other authors reported having been awarded honoraria, speaking fees, surgical research funding, and education grants from W.L. Gore and Associates, Ethicon, Novadaq, Bard/Davol, and LifeCell Corporation.
The porcine acellular dermal mesh product Strattice was associated with significantly lower odds of hernia recurrence, compared with several other biologic mesh products, in a study of 223 patients who underwent open ventral hernia repair.
Prospective operative outcomes data from a tertiary referral hernia center showed that at a mean follow-up of 18.2 months, the rate of hernia recurrence was 35% in 40 patients who were treated with Alloderm (LifeCell Corporation), 34.5% in 23 patients treated with AlloMax (Bard/Davol), 37.1% in 70 patients treated with FlexHD (Ethicon), and 59.1% in 22 patients treated with Xenmatrix (Bard/Davol), compared with 14.7% in 68 patients treated with Strattice (LifeCell Corporation). Alloderm, AlloMax, and FlexHD are all human acellular dermal mesh products, and Strattice and Xenmatrix are both porcine acellular dermal mesh products, Ciara R. Huntington, MD, and her colleagues at the Carolinas Medical Center in Charlotte, N.C., reported.

|
| Photo courtesy Acelity. STRATTICETM Reconstructive Tissue Matrix |
After multivariate analysis to adjust for factors such as comorbidities, hernia size, and intraoperative techniques, the odds ratios for recurrence with each product as compared with Strattice were 2.4 with Alloderm, 2.9 with FlexHD, 3.4 with AlloMax, and 7.8 with Xenmatrix. The odds for recurrence were significantly greater with all except Alloderm, the investigators said (Surgery. 2016. doi: 10.1016/j.surg.2016.07.008).
The significant differences between the two porcine acellular dermal meshes (Xenmatrix and Strattice) may reflect variation in tissue processing and design in biomesh engineering, they noted.
Study subjects were adults with a mean age of 57.7 years and mean body mass index of 34.8 kg/m2. Overall, 9.8% had an American Society of Anesthesiology classification of 4, 54.6% had a classification of 3, and 35.6% had a classification of 1 or 2. Average operative time was 241 minutes with estimated blood loss of 202 mL.
Average hernia defect size was 257 cm2, with average mesh size of 384 cm2.
“Component separation was performed in 47.5% of cases, and abdomen was left open prior to definitive closure in 10.7%. Biologic mesh was used to bridge fascial defects in 19.6% of cases. The mesh was placed in the preperitoneal space in 38.2% of cases,” the investigators wrote, noting that a concomitant procedure was performed in 82% of cases.
Sepsis developed in 6.7% of patients, 36.3% had a wound infection, and 24.3% required a negative pressure dressing for healing. The inpatient mortality rate was 1.4%.
However, mesh infections requiring explantation occurred in less than 1% of cases.
On adjusted analysis, Xenmatrix was the most expensive mesh and AlloMax was the least expensive (mean of $59,122 and $22,304, respectively). Strattice costs averaged $40,490.
Ventral hernia repair (VHR) is a common operation, with about 350,000 performed each year. Rates of postoperative wound infection and hernia recurrence vary widely, but may be improved with appropriate mesh selection. However, prospective data to guide selection are lacking, the investigators said.
“The great number of meshes available for use complicates the debate surrounding the best timing and use of biologic mesh in VHR, and the search for the better mesh for use in the abdominal wall reconstruction continues. Biologic mesh usually is reserved for the patients at the highest risk for developing a postoperative wound complication, and although there is a current dearth of high-level evidence supporting its use, this report confirms that complications are low despite obvious surgical complexity presented herein,” they wrote.
The findings of this study – the largest report of outcomes with biologic mesh in ventral hernia repair to date, according to the authors – support the safety of using biologic mesh in high-risk patients, they said.
They noted, however, that the study may still be underpowered to make final clinical decisions.
“Although our study provides useful information to the practicing surgeon, there is much work to be done regarding the selection of biologic mesh,” they wrote, adding that while “a well-performing biologic mesh should be in the toolkit of every general surgeon who may face complex abdominal walls requiring reconstruction in patients that are at high risk for a postoperative wound complication,” additional research is necessary to further clarify the role of biologic mesh in these operations.
Dr. Huntington reported having no disclosures. Other authors reported having been awarded honoraria, speaking fees, surgical research funding, and education grants from W.L. Gore and Associates, Ethicon, Novadaq, Bard/Davol, and LifeCell Corporation.
The porcine acellular dermal mesh product Strattice was associated with significantly lower odds of hernia recurrence, compared with several other biologic mesh products, in a study of 223 patients who underwent open ventral hernia repair.
Prospective operative outcomes data from a tertiary referral hernia center showed that at a mean follow-up of 18.2 months, the rate of hernia recurrence was 35% in 40 patients who were treated with Alloderm (LifeCell Corporation), 34.5% in 23 patients treated with AlloMax (Bard/Davol), 37.1% in 70 patients treated with FlexHD (Ethicon), and 59.1% in 22 patients treated with Xenmatrix (Bard/Davol), compared with 14.7% in 68 patients treated with Strattice (LifeCell Corporation). Alloderm, AlloMax, and FlexHD are all human acellular dermal mesh products, and Strattice and Xenmatrix are both porcine acellular dermal mesh products, Ciara R. Huntington, MD, and her colleagues at the Carolinas Medical Center in Charlotte, N.C., reported.

|
| Photo courtesy Acelity. STRATTICETM Reconstructive Tissue Matrix |
After multivariate analysis to adjust for factors such as comorbidities, hernia size, and intraoperative techniques, the odds ratios for recurrence with each product as compared with Strattice were 2.4 with Alloderm, 2.9 with FlexHD, 3.4 with AlloMax, and 7.8 with Xenmatrix. The odds for recurrence were significantly greater with all except Alloderm, the investigators said (Surgery. 2016. doi: 10.1016/j.surg.2016.07.008).
The significant differences between the two porcine acellular dermal meshes (Xenmatrix and Strattice) may reflect variation in tissue processing and design in biomesh engineering, they noted.
Study subjects were adults with a mean age of 57.7 years and mean body mass index of 34.8 kg/m2. Overall, 9.8% had an American Society of Anesthesiology classification of 4, 54.6% had a classification of 3, and 35.6% had a classification of 1 or 2. Average operative time was 241 minutes with estimated blood loss of 202 mL.
Average hernia defect size was 257 cm2, with average mesh size of 384 cm2.
“Component separation was performed in 47.5% of cases, and abdomen was left open prior to definitive closure in 10.7%. Biologic mesh was used to bridge fascial defects in 19.6% of cases. The mesh was placed in the preperitoneal space in 38.2% of cases,” the investigators wrote, noting that a concomitant procedure was performed in 82% of cases.
Sepsis developed in 6.7% of patients, 36.3% had a wound infection, and 24.3% required a negative pressure dressing for healing. The inpatient mortality rate was 1.4%.
However, mesh infections requiring explantation occurred in less than 1% of cases.
On adjusted analysis, Xenmatrix was the most expensive mesh and AlloMax was the least expensive (mean of $59,122 and $22,304, respectively). Strattice costs averaged $40,490.
Ventral hernia repair (VHR) is a common operation, with about 350,000 performed each year. Rates of postoperative wound infection and hernia recurrence vary widely, but may be improved with appropriate mesh selection. However, prospective data to guide selection are lacking, the investigators said.
“The great number of meshes available for use complicates the debate surrounding the best timing and use of biologic mesh in VHR, and the search for the better mesh for use in the abdominal wall reconstruction continues. Biologic mesh usually is reserved for the patients at the highest risk for developing a postoperative wound complication, and although there is a current dearth of high-level evidence supporting its use, this report confirms that complications are low despite obvious surgical complexity presented herein,” they wrote.
The findings of this study – the largest report of outcomes with biologic mesh in ventral hernia repair to date, according to the authors – support the safety of using biologic mesh in high-risk patients, they said.
They noted, however, that the study may still be underpowered to make final clinical decisions.
“Although our study provides useful information to the practicing surgeon, there is much work to be done regarding the selection of biologic mesh,” they wrote, adding that while “a well-performing biologic mesh should be in the toolkit of every general surgeon who may face complex abdominal walls requiring reconstruction in patients that are at high risk for a postoperative wound complication,” additional research is necessary to further clarify the role of biologic mesh in these operations.
Dr. Huntington reported having no disclosures. Other authors reported having been awarded honoraria, speaking fees, surgical research funding, and education grants from W.L. Gore and Associates, Ethicon, Novadaq, Bard/Davol, and LifeCell Corporation.
FROM SURGERY
Key clinical point: The porcine acellular dermal mesh product Strattice was associated with significantly lower odds of hernia recurrence, compared with several other biologic mesh products, in a study of 223 patients who underwent open ventral hernia repair.
Major finding: The adjusted odds ratios for recurrence, compared with Strattice, were 2.4 with Alloderm, 2.9 with FlexHD, 3.4 with AlloMax, and 7.8 with Xenmatrix.
Data source: 223 cases from a prospective operative outcomes database.
Disclosures: Dr. Huntington reported having no disclosures. Other authors reported having been awarded honoraria, speaking fees, surgical research funding, and education grants from W.L. Gore and Associates, Ethicon, Novadaq, Bard/Davol, and LifeCell Corporation.
Aim for modulation with neurotoxin injections
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Minimal residual disease negativity predicts better survival in MM
Minimal residual disease negativity following treatment for newly diagnosed multiple myeloma is associated with long-term survival, according to findings from a meta-analysis of relevant data.
The findings suggest that assessment for minimal residual disease (MRD) should be included as an end point in clinical trials of multiple myeloma, Nikhil C. Munshi, MD, of Harvard Medical School, Boston, and his colleagues reported online Sept. 15 in JAMA Oncology.
“This large cohort meta-analysis confirms that MRD status has prognostic value and is a valid surrogate marker for both PFS [progression-free survival] and OS [overall survival] in patients with multiple myeloma, including those who had achieved a CR [complete response],” the researchers wrote, noting that all of the studies confirmed the impact of MRD status on outcome, indicating that the predictive value of MRD status was independent of the type of treatment used.
Of 1,273 patients from 14 studies that looked at the impact of MRD status on PFS, 660 were MRD-negative and 613 were MRD-positive. Of 1,100 patients from 12 studies that looked at the impact of MRD on OS, 599 were MRD-negative, and 501 were MRD-positive. MRD-negative vs. -positive status was associated with better PFS and OS (hazard ratio, 0.41 and 0.57, respectively).
“Median PFS was 54 months for MRD-negative patients and 26 months for MRD-positive patients; median OS was 98 and 82 months, respectively,” the researchers wrote (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3160).
Further, to evaluate the impact of MRD status on PFS and OS in patients who achieved conventional CR, they analyzed data from five studies looking at PFS in 396 MRD-negative and 178 MRD-positive patients, and 6 studies looking at OS in 430 MRD-negative and 186 MRD-positive patients. In patients achieving CR, the presence of MRD predicted shorter PFS (HR, 0.44) and shorter OS (HR, 0.47), they said.
“Median PFS was 56 months for MRD-negative patients and 34 months for MRD-positive patients, and median OS was 112 and 82 months, respectively. The OS rate was higher for MRD-negative patients, compared with MRD-positive patients at 3 years (94% vs. 80%), 5 years (80% vs. 61%), and 7 years (67% vs. 47%),” they wrote.
Although none of the studies included in the analysis compared the effect of two different treatment approaches on MRD status, five did evaluate MRD status before and after autologous stem cell transplantation, and all indicated that the treatment increased the proportion of MRD-negative patients.
Maintenance therapy also appeared, based on some of the studies, to have a beneficial effect on MRD status. In one study, MRD-negative status was maintained in 96% of patients receiving thalidomide maintenance therapy vs. 69% of MRD-negative patients receiving no maintenance therapy.
Minimal residual disease status is already considered an important prognostic factor in patients with multiple myeloma, and testing could be used to monitor response to therapy and guide subsequent treatment decisions, the investigators wrote, noting that “recent development of multiparameter flow cytometry– and next-generation sequencing–based methods has allowed for MRD assessment in larger studies.”
The findings provide quantitative evidence to support the conceptual basis for integrating MRD assessment after initial treatment in clinical trials as a surrogate end point for PFS and/or OS and in clinical practice to aid in prognostication and to guide treatment. However, “integration of MRD testing into standard practice requires optimization and standardization of MRD assessment and standardization of its timing,” they said.
This study was supported by the National Institutes of Health, the Medical Research Council, and Celgene. Dr. Munshi reported consultancy positions and advisory committee memberships with Celgene, Takeda, Janssen, and Merck.
The meta-analysis by Munshi et al. begins to answer questions surrounding MRD, and highlights areas that require further investigation.
For example, the threshold used to define an MRD level that correlates best with clinical benefit remains to be determined.
While some data suggest that “deeper is better,” this remains unclear as deeper levels may be unnecessary – and harmful if achieved with greater toxic effects – in some patients. Alternatively, if deeper is better, perhaps MRD should be considered a continuous variable rather than a dichotomous one with a set cutoff.
Among other factors that require more examination are the role of MRD in patients not receiving autologous stem cell transplantation, whether MRD should only be assessed in those with complete response or those who attain a very good partial response, and whether – given the impact of cytogenetics on MRD – MRD negativity in patients with high-risk cytogenetics should be considered the same as MRD negativity in those with standard-risk cytogenetics.
What trial design or analysis features should be implemented to address this issue? Is stratification based on cytogenetic risk sufficient? What is the appropriate timing of MRD assessment? How will novel agents, such as monoclonal antibodies, affect the MRD assessment? What is the role of imaging? While the analysis is a considerable first step toward addressing these questions regarding MRD and its potential as a surrogate end point, many unanswered questions remain to be resolved.
Nicole J. Gormley, MD, and her colleagues made these comments in an editorial (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3112) that accompanied the study. Dr. Gormley is with the Division of Hematology Products, U.S. Food and Drug Administration, Silver Spring, Md. She reported having no disclosures.
The meta-analysis by Munshi et al. begins to answer questions surrounding MRD, and highlights areas that require further investigation.
For example, the threshold used to define an MRD level that correlates best with clinical benefit remains to be determined.
While some data suggest that “deeper is better,” this remains unclear as deeper levels may be unnecessary – and harmful if achieved with greater toxic effects – in some patients. Alternatively, if deeper is better, perhaps MRD should be considered a continuous variable rather than a dichotomous one with a set cutoff.
Among other factors that require more examination are the role of MRD in patients not receiving autologous stem cell transplantation, whether MRD should only be assessed in those with complete response or those who attain a very good partial response, and whether – given the impact of cytogenetics on MRD – MRD negativity in patients with high-risk cytogenetics should be considered the same as MRD negativity in those with standard-risk cytogenetics.
What trial design or analysis features should be implemented to address this issue? Is stratification based on cytogenetic risk sufficient? What is the appropriate timing of MRD assessment? How will novel agents, such as monoclonal antibodies, affect the MRD assessment? What is the role of imaging? While the analysis is a considerable first step toward addressing these questions regarding MRD and its potential as a surrogate end point, many unanswered questions remain to be resolved.
Nicole J. Gormley, MD, and her colleagues made these comments in an editorial (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3112) that accompanied the study. Dr. Gormley is with the Division of Hematology Products, U.S. Food and Drug Administration, Silver Spring, Md. She reported having no disclosures.
The meta-analysis by Munshi et al. begins to answer questions surrounding MRD, and highlights areas that require further investigation.
For example, the threshold used to define an MRD level that correlates best with clinical benefit remains to be determined.
While some data suggest that “deeper is better,” this remains unclear as deeper levels may be unnecessary – and harmful if achieved with greater toxic effects – in some patients. Alternatively, if deeper is better, perhaps MRD should be considered a continuous variable rather than a dichotomous one with a set cutoff.
Among other factors that require more examination are the role of MRD in patients not receiving autologous stem cell transplantation, whether MRD should only be assessed in those with complete response or those who attain a very good partial response, and whether – given the impact of cytogenetics on MRD – MRD negativity in patients with high-risk cytogenetics should be considered the same as MRD negativity in those with standard-risk cytogenetics.
What trial design or analysis features should be implemented to address this issue? Is stratification based on cytogenetic risk sufficient? What is the appropriate timing of MRD assessment? How will novel agents, such as monoclonal antibodies, affect the MRD assessment? What is the role of imaging? While the analysis is a considerable first step toward addressing these questions regarding MRD and its potential as a surrogate end point, many unanswered questions remain to be resolved.
Nicole J. Gormley, MD, and her colleagues made these comments in an editorial (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3112) that accompanied the study. Dr. Gormley is with the Division of Hematology Products, U.S. Food and Drug Administration, Silver Spring, Md. She reported having no disclosures.
Minimal residual disease negativity following treatment for newly diagnosed multiple myeloma is associated with long-term survival, according to findings from a meta-analysis of relevant data.
The findings suggest that assessment for minimal residual disease (MRD) should be included as an end point in clinical trials of multiple myeloma, Nikhil C. Munshi, MD, of Harvard Medical School, Boston, and his colleagues reported online Sept. 15 in JAMA Oncology.
“This large cohort meta-analysis confirms that MRD status has prognostic value and is a valid surrogate marker for both PFS [progression-free survival] and OS [overall survival] in patients with multiple myeloma, including those who had achieved a CR [complete response],” the researchers wrote, noting that all of the studies confirmed the impact of MRD status on outcome, indicating that the predictive value of MRD status was independent of the type of treatment used.
Of 1,273 patients from 14 studies that looked at the impact of MRD status on PFS, 660 were MRD-negative and 613 were MRD-positive. Of 1,100 patients from 12 studies that looked at the impact of MRD on OS, 599 were MRD-negative, and 501 were MRD-positive. MRD-negative vs. -positive status was associated with better PFS and OS (hazard ratio, 0.41 and 0.57, respectively).
“Median PFS was 54 months for MRD-negative patients and 26 months for MRD-positive patients; median OS was 98 and 82 months, respectively,” the researchers wrote (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3160).
Further, to evaluate the impact of MRD status on PFS and OS in patients who achieved conventional CR, they analyzed data from five studies looking at PFS in 396 MRD-negative and 178 MRD-positive patients, and 6 studies looking at OS in 430 MRD-negative and 186 MRD-positive patients. In patients achieving CR, the presence of MRD predicted shorter PFS (HR, 0.44) and shorter OS (HR, 0.47), they said.
“Median PFS was 56 months for MRD-negative patients and 34 months for MRD-positive patients, and median OS was 112 and 82 months, respectively. The OS rate was higher for MRD-negative patients, compared with MRD-positive patients at 3 years (94% vs. 80%), 5 years (80% vs. 61%), and 7 years (67% vs. 47%),” they wrote.
Although none of the studies included in the analysis compared the effect of two different treatment approaches on MRD status, five did evaluate MRD status before and after autologous stem cell transplantation, and all indicated that the treatment increased the proportion of MRD-negative patients.
Maintenance therapy also appeared, based on some of the studies, to have a beneficial effect on MRD status. In one study, MRD-negative status was maintained in 96% of patients receiving thalidomide maintenance therapy vs. 69% of MRD-negative patients receiving no maintenance therapy.
Minimal residual disease status is already considered an important prognostic factor in patients with multiple myeloma, and testing could be used to monitor response to therapy and guide subsequent treatment decisions, the investigators wrote, noting that “recent development of multiparameter flow cytometry– and next-generation sequencing–based methods has allowed for MRD assessment in larger studies.”
The findings provide quantitative evidence to support the conceptual basis for integrating MRD assessment after initial treatment in clinical trials as a surrogate end point for PFS and/or OS and in clinical practice to aid in prognostication and to guide treatment. However, “integration of MRD testing into standard practice requires optimization and standardization of MRD assessment and standardization of its timing,” they said.
This study was supported by the National Institutes of Health, the Medical Research Council, and Celgene. Dr. Munshi reported consultancy positions and advisory committee memberships with Celgene, Takeda, Janssen, and Merck.
Minimal residual disease negativity following treatment for newly diagnosed multiple myeloma is associated with long-term survival, according to findings from a meta-analysis of relevant data.
The findings suggest that assessment for minimal residual disease (MRD) should be included as an end point in clinical trials of multiple myeloma, Nikhil C. Munshi, MD, of Harvard Medical School, Boston, and his colleagues reported online Sept. 15 in JAMA Oncology.
“This large cohort meta-analysis confirms that MRD status has prognostic value and is a valid surrogate marker for both PFS [progression-free survival] and OS [overall survival] in patients with multiple myeloma, including those who had achieved a CR [complete response],” the researchers wrote, noting that all of the studies confirmed the impact of MRD status on outcome, indicating that the predictive value of MRD status was independent of the type of treatment used.
Of 1,273 patients from 14 studies that looked at the impact of MRD status on PFS, 660 were MRD-negative and 613 were MRD-positive. Of 1,100 patients from 12 studies that looked at the impact of MRD on OS, 599 were MRD-negative, and 501 were MRD-positive. MRD-negative vs. -positive status was associated with better PFS and OS (hazard ratio, 0.41 and 0.57, respectively).
“Median PFS was 54 months for MRD-negative patients and 26 months for MRD-positive patients; median OS was 98 and 82 months, respectively,” the researchers wrote (JAMA Oncol. 2016 Sep 15. doi: 10.1001/jamaoncol.2016.3160).
Further, to evaluate the impact of MRD status on PFS and OS in patients who achieved conventional CR, they analyzed data from five studies looking at PFS in 396 MRD-negative and 178 MRD-positive patients, and 6 studies looking at OS in 430 MRD-negative and 186 MRD-positive patients. In patients achieving CR, the presence of MRD predicted shorter PFS (HR, 0.44) and shorter OS (HR, 0.47), they said.
“Median PFS was 56 months for MRD-negative patients and 34 months for MRD-positive patients, and median OS was 112 and 82 months, respectively. The OS rate was higher for MRD-negative patients, compared with MRD-positive patients at 3 years (94% vs. 80%), 5 years (80% vs. 61%), and 7 years (67% vs. 47%),” they wrote.
Although none of the studies included in the analysis compared the effect of two different treatment approaches on MRD status, five did evaluate MRD status before and after autologous stem cell transplantation, and all indicated that the treatment increased the proportion of MRD-negative patients.
Maintenance therapy also appeared, based on some of the studies, to have a beneficial effect on MRD status. In one study, MRD-negative status was maintained in 96% of patients receiving thalidomide maintenance therapy vs. 69% of MRD-negative patients receiving no maintenance therapy.
Minimal residual disease status is already considered an important prognostic factor in patients with multiple myeloma, and testing could be used to monitor response to therapy and guide subsequent treatment decisions, the investigators wrote, noting that “recent development of multiparameter flow cytometry– and next-generation sequencing–based methods has allowed for MRD assessment in larger studies.”
The findings provide quantitative evidence to support the conceptual basis for integrating MRD assessment after initial treatment in clinical trials as a surrogate end point for PFS and/or OS and in clinical practice to aid in prognostication and to guide treatment. However, “integration of MRD testing into standard practice requires optimization and standardization of MRD assessment and standardization of its timing,” they said.
This study was supported by the National Institutes of Health, the Medical Research Council, and Celgene. Dr. Munshi reported consultancy positions and advisory committee memberships with Celgene, Takeda, Janssen, and Merck.
FROM JAMA ONCOLOGY
Key clinical point: Minimal residual disease negativity following treatment for newly diagnosed multiple myeloma is associated with long-term survival, according to findings from a meta-analysis of relevant data.
Major finding: MRD-negative vs. -positive status was associated with better PFS and OS (HR, 0.41 and 0.57, respectively).
Data source: A meta-analysis of data from 21 studies.
Disclosures: This study was supported by the National Institutes of Health, the Medical Research Council, and Celgene. Dr. Munshi reported consultancy positions and advisory committee memberships with Celgene, Takeda, Janssen, and Merck.
Sequential protocol benefits patients with field cancerization
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Treat bed bug bites with topical steroids
BOSTON – Preventing repeated occurrences of bed bug bites means eliminating infestations or – if the bites occur during travel – avoiding bringing the bugs home, according to Theodore Rosen, MD.
Bed bugs are “very, very, very hardy,” and can live months or maybe even years between blood meals, Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston, said at the American Academy of Dermatology summer meeting.
“They lay lots of eggs and have lots of offspring,” he said, adding that bed bugs have evolved and developed mutations that make them resistant to insecticides, and can survive up to a week at a continuous temperature of 10 degrees Fahrenheit.
Advise patients who will be traveling to check whether the area they are traveling to has known bed bug infestations. Recommend that they always check areas within 3 feet or so of hotel beds for evidence of bed bugs, he said.
Bed bugs like right angles, so they may be found on headboards, bed frames, mattress piping, drawers, and windows. They can also be found hiding under peeling paint, and or in the piping or under surfaces of furniture in hotel rooms.
Dr. Rosen is often asked by patients if he really looks for bed bugs when he travels. “I do look – and I’ve found them,” he said, noting that they can be found in the nicest of hotels. He found some lurking beneath the drawer of a nightstand in one luxury hotel.
Bugs can be seen, but droppings are another telltale sign of infestation; bed bug excrement stains fabrics, so if round brown splotches are seen on mattress or other fabric, bed bugs have been there and probably still are there, Dr. Rosen said.
“I pull up the sheets, look at the piping on the mattress, pull up the mattress and look at the frame, look at the coils, look at the night stand,” he said.
A pricey monitoring and trapping device called the NightWatch can be purchased to help in the event of an infestation. The device emits heated carbon dioxide and pheromones that attract and trap bed bugs, but the $300-$400 price tag can be prohibitive, he noted.
The best bet is to avoid bringing home bugs by keeping all bags and clothes off the floor. Bags should be kept on a dresser or stand, clothes should be hung.
If infestation occurs, let patients know that the bites aren’t dangerous. There is no convincing evidence that bed bugs transmit any diseases. Anemia with heavy infestation and repeated bites could theoretically be a concern, but other than itching and looking bad, bites are rarely an issue.
Bed bug bites usually respond well to topical steroid treatment, and only in rare cases are oral steroids or antihistamines needed. Home bites will recur if there is infestation, and the problem can be a source of depression and anxiety, which in some patients can be severe and lead to suicidal ideation.
Infestation should be addressed by heavy vacuuming and steaming, which kills bed bugs if he temperatures are high enough. Mattress encasement can also help, but insecticides are rarely effective. Freezing smaller objects can be tried, but bed bugs can withstand freezing temperatures for long periods of time, he said.
“If you bring bed bugs home, you have to treat the whole environment,” he said.
One treatment that has shown promise is ivermectin in the host. Studies suggest this approach kills some bugs, but it is not 100% effective and is probably best reserved for widespread infestation, such as in a nursing home, Dr. Rosen said.
He disclosed financial or other relationships with Anacor Pharmaceuticals; Innocutis; Sandoz, a Novartis company; and Valeant Pharmaceuticals International.
BOSTON – Preventing repeated occurrences of bed bug bites means eliminating infestations or – if the bites occur during travel – avoiding bringing the bugs home, according to Theodore Rosen, MD.
Bed bugs are “very, very, very hardy,” and can live months or maybe even years between blood meals, Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston, said at the American Academy of Dermatology summer meeting.
“They lay lots of eggs and have lots of offspring,” he said, adding that bed bugs have evolved and developed mutations that make them resistant to insecticides, and can survive up to a week at a continuous temperature of 10 degrees Fahrenheit.
Advise patients who will be traveling to check whether the area they are traveling to has known bed bug infestations. Recommend that they always check areas within 3 feet or so of hotel beds for evidence of bed bugs, he said.
Bed bugs like right angles, so they may be found on headboards, bed frames, mattress piping, drawers, and windows. They can also be found hiding under peeling paint, and or in the piping or under surfaces of furniture in hotel rooms.
Dr. Rosen is often asked by patients if he really looks for bed bugs when he travels. “I do look – and I’ve found them,” he said, noting that they can be found in the nicest of hotels. He found some lurking beneath the drawer of a nightstand in one luxury hotel.
Bugs can be seen, but droppings are another telltale sign of infestation; bed bug excrement stains fabrics, so if round brown splotches are seen on mattress or other fabric, bed bugs have been there and probably still are there, Dr. Rosen said.
“I pull up the sheets, look at the piping on the mattress, pull up the mattress and look at the frame, look at the coils, look at the night stand,” he said.
A pricey monitoring and trapping device called the NightWatch can be purchased to help in the event of an infestation. The device emits heated carbon dioxide and pheromones that attract and trap bed bugs, but the $300-$400 price tag can be prohibitive, he noted.
The best bet is to avoid bringing home bugs by keeping all bags and clothes off the floor. Bags should be kept on a dresser or stand, clothes should be hung.
If infestation occurs, let patients know that the bites aren’t dangerous. There is no convincing evidence that bed bugs transmit any diseases. Anemia with heavy infestation and repeated bites could theoretically be a concern, but other than itching and looking bad, bites are rarely an issue.
Bed bug bites usually respond well to topical steroid treatment, and only in rare cases are oral steroids or antihistamines needed. Home bites will recur if there is infestation, and the problem can be a source of depression and anxiety, which in some patients can be severe and lead to suicidal ideation.
Infestation should be addressed by heavy vacuuming and steaming, which kills bed bugs if he temperatures are high enough. Mattress encasement can also help, but insecticides are rarely effective. Freezing smaller objects can be tried, but bed bugs can withstand freezing temperatures for long periods of time, he said.
“If you bring bed bugs home, you have to treat the whole environment,” he said.
One treatment that has shown promise is ivermectin in the host. Studies suggest this approach kills some bugs, but it is not 100% effective and is probably best reserved for widespread infestation, such as in a nursing home, Dr. Rosen said.
He disclosed financial or other relationships with Anacor Pharmaceuticals; Innocutis; Sandoz, a Novartis company; and Valeant Pharmaceuticals International.
BOSTON – Preventing repeated occurrences of bed bug bites means eliminating infestations or – if the bites occur during travel – avoiding bringing the bugs home, according to Theodore Rosen, MD.
Bed bugs are “very, very, very hardy,” and can live months or maybe even years between blood meals, Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston, said at the American Academy of Dermatology summer meeting.
“They lay lots of eggs and have lots of offspring,” he said, adding that bed bugs have evolved and developed mutations that make them resistant to insecticides, and can survive up to a week at a continuous temperature of 10 degrees Fahrenheit.
Advise patients who will be traveling to check whether the area they are traveling to has known bed bug infestations. Recommend that they always check areas within 3 feet or so of hotel beds for evidence of bed bugs, he said.
Bed bugs like right angles, so they may be found on headboards, bed frames, mattress piping, drawers, and windows. They can also be found hiding under peeling paint, and or in the piping or under surfaces of furniture in hotel rooms.
Dr. Rosen is often asked by patients if he really looks for bed bugs when he travels. “I do look – and I’ve found them,” he said, noting that they can be found in the nicest of hotels. He found some lurking beneath the drawer of a nightstand in one luxury hotel.
Bugs can be seen, but droppings are another telltale sign of infestation; bed bug excrement stains fabrics, so if round brown splotches are seen on mattress or other fabric, bed bugs have been there and probably still are there, Dr. Rosen said.
“I pull up the sheets, look at the piping on the mattress, pull up the mattress and look at the frame, look at the coils, look at the night stand,” he said.
A pricey monitoring and trapping device called the NightWatch can be purchased to help in the event of an infestation. The device emits heated carbon dioxide and pheromones that attract and trap bed bugs, but the $300-$400 price tag can be prohibitive, he noted.
The best bet is to avoid bringing home bugs by keeping all bags and clothes off the floor. Bags should be kept on a dresser or stand, clothes should be hung.
If infestation occurs, let patients know that the bites aren’t dangerous. There is no convincing evidence that bed bugs transmit any diseases. Anemia with heavy infestation and repeated bites could theoretically be a concern, but other than itching and looking bad, bites are rarely an issue.
Bed bug bites usually respond well to topical steroid treatment, and only in rare cases are oral steroids or antihistamines needed. Home bites will recur if there is infestation, and the problem can be a source of depression and anxiety, which in some patients can be severe and lead to suicidal ideation.
Infestation should be addressed by heavy vacuuming and steaming, which kills bed bugs if he temperatures are high enough. Mattress encasement can also help, but insecticides are rarely effective. Freezing smaller objects can be tried, but bed bugs can withstand freezing temperatures for long periods of time, he said.
“If you bring bed bugs home, you have to treat the whole environment,” he said.
One treatment that has shown promise is ivermectin in the host. Studies suggest this approach kills some bugs, but it is not 100% effective and is probably best reserved for widespread infestation, such as in a nursing home, Dr. Rosen said.
He disclosed financial or other relationships with Anacor Pharmaceuticals; Innocutis; Sandoz, a Novartis company; and Valeant Pharmaceuticals International.
EXPERT ANALYSIS FROM AAD Summer Academy 2016
FDA: New labeling warns against combining opioids, benzodiazepines
Labeling for prescription opioid pain or cough medicines and benzodiazepines will now carry the strongest available warning regarding serious side effects and death associated with their combined use, according to the Food and Drug Administration.
The new boxed warnings urge health care professionals to limit prescribing opioid pain medicines with benzodiazepines or other central nervous system depressants only to patients for whom alternative treatment options are inadequate, and to limit dosages and treatment duration to the minimum possible while achieving the desired clinical effect.
“First, the FDA is requiring companies to update their product labeling for ... benzodiazepines and opioids to include possible harms when they are used together. Second, we are requiring new or updated medication guides for these drugs reflecting those same warnings,” said Doug Throckmorton, MD, deputy director of the FDA’s Center for Drug Evaluation and Research, during a telebriefing.
Opioids will include a warning regarding prescribing with benzodiazepines and other central nervous system depressants, including alcohol. Benzodiazepines will include a warning regarding prescribing with opioids.
In addition, the FDA has issued a safety communication to “warn the public about the serious risk of taking these products together to help make doctors more cautious and patients better informed,” Dr. Throckmorton said.
The action comes amid ongoing efforts to address an epidemic of opioid addiction across the United States, and in response to a first-of-its-kind “citizen petition” calling for the boxed warnings.
A coalition of health officials from multiple cities, states, and U.S. territories initiated that petition in February, and thousands of concerned community members started an additional online petition. Those petitions were in response to both the increasing combined use of opioids and benzodiazepines and a concomitant increase in the risk of serious side effects and deaths associated with their combined use, according to Baltimore City Health Commissioner Leana Wen, MD.
As an emergency physician, Dr. Wen said that she has seen firsthand the alarming trends; one in three unintentional overdose deaths from prescribed opioids also involve benzodiazepines, she noted.
“In my state of Maryland in 2014, benzodiazepines were associated with 19% of prescription opioid deaths, and 59% of benzodiazepine-associated deaths involved prescription opioids. We also noted the growing biological evidence that combining these medications caused sleepiness and slowed breathing, increasing the likelihood of a fatal overdose,” she said.
Dr. Throckmorton further noted that emergency department visits and deaths involving patients prescribed both opioids and benzodiazepines have increased significantly over time. From 2004 to 2011, the rate of nonmedical use–related emergency department visits increased significantly each year, and overdose deaths involving both drug classes during that period nearly tripled on an annual basis.
“Communities have been seeing this trend for some time, but ultimately we needed data in order to act today,” FDA Commissioner Robert Califf, MD, said during the telebriefing.
The current action is just “one part of a larger effort to address this epidemic.
“We remain focused and deeply committed to contributing to the comprehensive effort to address the opioid epidemic,” Dr. Califf said. The FDA “will continue to monitor these products carefully and take additional actions as needed, and will share updates with the public as necessary as we work to address this public health crisis.”
Dr. Califf noted that the current action is part of the FDA’s Opioids Action Plan, which is “importantly not meant just to cover illicit or abusive use of opioids.”
“So, you’ll be hearing a lot more from us, because this is a national crisis that is not going away. We’re making progress on the prescribing, and we’re seeing a reduction in the use of opioids now,” he noted. “But we’re still seeing many overdoses.
“This is a continuum, and we’ll continue to try to do everything we can to address the epidemic,” Dr. Califf concluded.
Labeling for prescription opioid pain or cough medicines and benzodiazepines will now carry the strongest available warning regarding serious side effects and death associated with their combined use, according to the Food and Drug Administration.
The new boxed warnings urge health care professionals to limit prescribing opioid pain medicines with benzodiazepines or other central nervous system depressants only to patients for whom alternative treatment options are inadequate, and to limit dosages and treatment duration to the minimum possible while achieving the desired clinical effect.
“First, the FDA is requiring companies to update their product labeling for ... benzodiazepines and opioids to include possible harms when they are used together. Second, we are requiring new or updated medication guides for these drugs reflecting those same warnings,” said Doug Throckmorton, MD, deputy director of the FDA’s Center for Drug Evaluation and Research, during a telebriefing.
Opioids will include a warning regarding prescribing with benzodiazepines and other central nervous system depressants, including alcohol. Benzodiazepines will include a warning regarding prescribing with opioids.
In addition, the FDA has issued a safety communication to “warn the public about the serious risk of taking these products together to help make doctors more cautious and patients better informed,” Dr. Throckmorton said.
The action comes amid ongoing efforts to address an epidemic of opioid addiction across the United States, and in response to a first-of-its-kind “citizen petition” calling for the boxed warnings.
A coalition of health officials from multiple cities, states, and U.S. territories initiated that petition in February, and thousands of concerned community members started an additional online petition. Those petitions were in response to both the increasing combined use of opioids and benzodiazepines and a concomitant increase in the risk of serious side effects and deaths associated with their combined use, according to Baltimore City Health Commissioner Leana Wen, MD.
As an emergency physician, Dr. Wen said that she has seen firsthand the alarming trends; one in three unintentional overdose deaths from prescribed opioids also involve benzodiazepines, she noted.
“In my state of Maryland in 2014, benzodiazepines were associated with 19% of prescription opioid deaths, and 59% of benzodiazepine-associated deaths involved prescription opioids. We also noted the growing biological evidence that combining these medications caused sleepiness and slowed breathing, increasing the likelihood of a fatal overdose,” she said.
Dr. Throckmorton further noted that emergency department visits and deaths involving patients prescribed both opioids and benzodiazepines have increased significantly over time. From 2004 to 2011, the rate of nonmedical use–related emergency department visits increased significantly each year, and overdose deaths involving both drug classes during that period nearly tripled on an annual basis.
“Communities have been seeing this trend for some time, but ultimately we needed data in order to act today,” FDA Commissioner Robert Califf, MD, said during the telebriefing.
The current action is just “one part of a larger effort to address this epidemic.
“We remain focused and deeply committed to contributing to the comprehensive effort to address the opioid epidemic,” Dr. Califf said. The FDA “will continue to monitor these products carefully and take additional actions as needed, and will share updates with the public as necessary as we work to address this public health crisis.”
Dr. Califf noted that the current action is part of the FDA’s Opioids Action Plan, which is “importantly not meant just to cover illicit or abusive use of opioids.”
“So, you’ll be hearing a lot more from us, because this is a national crisis that is not going away. We’re making progress on the prescribing, and we’re seeing a reduction in the use of opioids now,” he noted. “But we’re still seeing many overdoses.
“This is a continuum, and we’ll continue to try to do everything we can to address the epidemic,” Dr. Califf concluded.
Labeling for prescription opioid pain or cough medicines and benzodiazepines will now carry the strongest available warning regarding serious side effects and death associated with their combined use, according to the Food and Drug Administration.
The new boxed warnings urge health care professionals to limit prescribing opioid pain medicines with benzodiazepines or other central nervous system depressants only to patients for whom alternative treatment options are inadequate, and to limit dosages and treatment duration to the minimum possible while achieving the desired clinical effect.
“First, the FDA is requiring companies to update their product labeling for ... benzodiazepines and opioids to include possible harms when they are used together. Second, we are requiring new or updated medication guides for these drugs reflecting those same warnings,” said Doug Throckmorton, MD, deputy director of the FDA’s Center for Drug Evaluation and Research, during a telebriefing.
Opioids will include a warning regarding prescribing with benzodiazepines and other central nervous system depressants, including alcohol. Benzodiazepines will include a warning regarding prescribing with opioids.
In addition, the FDA has issued a safety communication to “warn the public about the serious risk of taking these products together to help make doctors more cautious and patients better informed,” Dr. Throckmorton said.
The action comes amid ongoing efforts to address an epidemic of opioid addiction across the United States, and in response to a first-of-its-kind “citizen petition” calling for the boxed warnings.
A coalition of health officials from multiple cities, states, and U.S. territories initiated that petition in February, and thousands of concerned community members started an additional online petition. Those petitions were in response to both the increasing combined use of opioids and benzodiazepines and a concomitant increase in the risk of serious side effects and deaths associated with their combined use, according to Baltimore City Health Commissioner Leana Wen, MD.
As an emergency physician, Dr. Wen said that she has seen firsthand the alarming trends; one in three unintentional overdose deaths from prescribed opioids also involve benzodiazepines, she noted.
“In my state of Maryland in 2014, benzodiazepines were associated with 19% of prescription opioid deaths, and 59% of benzodiazepine-associated deaths involved prescription opioids. We also noted the growing biological evidence that combining these medications caused sleepiness and slowed breathing, increasing the likelihood of a fatal overdose,” she said.
Dr. Throckmorton further noted that emergency department visits and deaths involving patients prescribed both opioids and benzodiazepines have increased significantly over time. From 2004 to 2011, the rate of nonmedical use–related emergency department visits increased significantly each year, and overdose deaths involving both drug classes during that period nearly tripled on an annual basis.
“Communities have been seeing this trend for some time, but ultimately we needed data in order to act today,” FDA Commissioner Robert Califf, MD, said during the telebriefing.
The current action is just “one part of a larger effort to address this epidemic.
“We remain focused and deeply committed to contributing to the comprehensive effort to address the opioid epidemic,” Dr. Califf said. The FDA “will continue to monitor these products carefully and take additional actions as needed, and will share updates with the public as necessary as we work to address this public health crisis.”
Dr. Califf noted that the current action is part of the FDA’s Opioids Action Plan, which is “importantly not meant just to cover illicit or abusive use of opioids.”
“So, you’ll be hearing a lot more from us, because this is a national crisis that is not going away. We’re making progress on the prescribing, and we’re seeing a reduction in the use of opioids now,” he noted. “But we’re still seeing many overdoses.
“This is a continuum, and we’ll continue to try to do everything we can to address the epidemic,” Dr. Califf concluded.
Dasatinib plus venetoclax shows promise in mouse model of Ph+ALL
The combination of dasatinib and venetoclax had a synergistic effect that was associated with lower toxicity than single-agent therapy, based on responses of primary Philadelphia chromosome–positive acute lymphoblastic leukemia (PH+ALL) samples in xenografted immunodeficient mice, according to Jessica T. Leonard, MD.
Dr. Leonard and her colleagues at Oregon Health and Science University in Portland demonstrated that the combination of venetoclax – a selective inhibitor of B cell lymphoma 2 – was highly synergistic with tyrosine kinase inhibitors in vitro. In the preclinical model of PH+ALL, a stepwise reduction in median inhibitory concentration of dasatinib was observed with increasing doses of venetoclax, as was decreased cell viability and induced apoptosis. Dasatinib – a breakpoint cluster region–Abelson kinase inhibitor – has an additional advantage of potentially overcoming venetoclax resistance by blocking a common mechanism of resistance to the agent, the investigators reported Aug. 31 in Science Translational Medicine (2016; 8[354]:354ra114).
The combination boosted antitumor activity against Ph+ALL cells grown in culture. In the mouse model of Ph+ALL, all of the mice in the combination dosing group remained alive during the 4-week treatment period. The combination therapy was well tolerated, and superior to either agent alone with respect to antileukemic efficacy.
The investigators focused on combining venetoclax with dasatinib because it is “the current backbone for the treatment of adult Ph+ALL ... These results lay the foundation for the testing of this combination in patients with Ph+ALL with the goal of improving treatment,” they concluded.
This study was supported in part by the Leukemia & Lymphoma Society and the Newman’s Own Foundation. Dr. Leonard reported having no other disclosures, but various coauthors reported receiving research support from, and/or serving as a consultant or scientific advisory board member to Genentech, the maker of venetoclax, and Bristol-Myers Squibb, the maker of dasatinib, as well as numerous other drug companies.
The combination of dasatinib and venetoclax had a synergistic effect that was associated with lower toxicity than single-agent therapy, based on responses of primary Philadelphia chromosome–positive acute lymphoblastic leukemia (PH+ALL) samples in xenografted immunodeficient mice, according to Jessica T. Leonard, MD.
Dr. Leonard and her colleagues at Oregon Health and Science University in Portland demonstrated that the combination of venetoclax – a selective inhibitor of B cell lymphoma 2 – was highly synergistic with tyrosine kinase inhibitors in vitro. In the preclinical model of PH+ALL, a stepwise reduction in median inhibitory concentration of dasatinib was observed with increasing doses of venetoclax, as was decreased cell viability and induced apoptosis. Dasatinib – a breakpoint cluster region–Abelson kinase inhibitor – has an additional advantage of potentially overcoming venetoclax resistance by blocking a common mechanism of resistance to the agent, the investigators reported Aug. 31 in Science Translational Medicine (2016; 8[354]:354ra114).
The combination boosted antitumor activity against Ph+ALL cells grown in culture. In the mouse model of Ph+ALL, all of the mice in the combination dosing group remained alive during the 4-week treatment period. The combination therapy was well tolerated, and superior to either agent alone with respect to antileukemic efficacy.
The investigators focused on combining venetoclax with dasatinib because it is “the current backbone for the treatment of adult Ph+ALL ... These results lay the foundation for the testing of this combination in patients with Ph+ALL with the goal of improving treatment,” they concluded.
This study was supported in part by the Leukemia & Lymphoma Society and the Newman’s Own Foundation. Dr. Leonard reported having no other disclosures, but various coauthors reported receiving research support from, and/or serving as a consultant or scientific advisory board member to Genentech, the maker of venetoclax, and Bristol-Myers Squibb, the maker of dasatinib, as well as numerous other drug companies.
The combination of dasatinib and venetoclax had a synergistic effect that was associated with lower toxicity than single-agent therapy, based on responses of primary Philadelphia chromosome–positive acute lymphoblastic leukemia (PH+ALL) samples in xenografted immunodeficient mice, according to Jessica T. Leonard, MD.
Dr. Leonard and her colleagues at Oregon Health and Science University in Portland demonstrated that the combination of venetoclax – a selective inhibitor of B cell lymphoma 2 – was highly synergistic with tyrosine kinase inhibitors in vitro. In the preclinical model of PH+ALL, a stepwise reduction in median inhibitory concentration of dasatinib was observed with increasing doses of venetoclax, as was decreased cell viability and induced apoptosis. Dasatinib – a breakpoint cluster region–Abelson kinase inhibitor – has an additional advantage of potentially overcoming venetoclax resistance by blocking a common mechanism of resistance to the agent, the investigators reported Aug. 31 in Science Translational Medicine (2016; 8[354]:354ra114).
The combination boosted antitumor activity against Ph+ALL cells grown in culture. In the mouse model of Ph+ALL, all of the mice in the combination dosing group remained alive during the 4-week treatment period. The combination therapy was well tolerated, and superior to either agent alone with respect to antileukemic efficacy.
The investigators focused on combining venetoclax with dasatinib because it is “the current backbone for the treatment of adult Ph+ALL ... These results lay the foundation for the testing of this combination in patients with Ph+ALL with the goal of improving treatment,” they concluded.
This study was supported in part by the Leukemia & Lymphoma Society and the Newman’s Own Foundation. Dr. Leonard reported having no other disclosures, but various coauthors reported receiving research support from, and/or serving as a consultant or scientific advisory board member to Genentech, the maker of venetoclax, and Bristol-Myers Squibb, the maker of dasatinib, as well as numerous other drug companies.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: The combination of dasatinib and venetoclax shows promise for the treatment of primary Philadelphia chromosome–positive acute lymphoblastic leukemia (Ph+ALL) samples in xenografted immunodeficient mice and should be further evaluated for patient care.
Major finding: A stepwise reduction in median inhibitory concentration of dasatinib was seen with increasing doses of venetoclax.
Data source: In vitro and in vivo evaluation of BCL-2 inhibition in combination with kinase inhibition in a murine model of Ph+ALL.
Disclosures: This study was supported in part by the Leukemia & Lymphoma Society and the Newman’s Own Foundation. Dr. Leonard reported having no other disclosures, but various coauthors reported receiving research support from, and/or serving as a consultant or scientific advisory board member to Genentech, the maker of venetoclax, and Bristol-Myers Squibb, the maker of dasatinib, as well as numerous other drug companies.