User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Antipsychotic agents key for treating delusional infestation
Boston – Patients with delusional infestation often resist referral to a psychiatrist and insist on treatment by a dermatologist, according to Mark D. P. Davis, MD.
Dermatologists, conversely, often don’t want to treat these challenging patients because they aren’t really sure how to manage them, Dr. Davis said during a “practice gaps” session at the American Academy of Dermatology summer meeting.
An important practice gap is the lack of a standard approach to the investigation and management of these patients, he said.
An audience poll showed that respondents were divided as to whether patients presenting with delusional infestation, also known as delusional parasitosis, should be confronted, referred to a psychiatrist, treated with an antipsychotic, or approached in some other way.
The condition, which involves the delusional belief that one’s skin is “infested” with parasites, fibers, or some other materials, can result from another cause, said Dr. Davis of the Mayo Clinic, Rochester, Minn, noting that anxiety, depression, and medications such as opioids or treatments for attention deficit/hyperactivity disorder can contribute to the delusions.
Skin diseases themselves can also cause the sensation of infestation.
A good history should assess medical comorbidities, electrolyte abnormalities, or medication use that might be contributing to the problem.
For dermatologists who are not well-versed in “picking up on psychiatric comorbidities,” validated measures can be useful. Tools such as the Personal Health Questionnaire (PHQ)-9 for depression and Generalized Anxiety Disorder (GAD)-7 can be useful, Dr. Davis said.
However, delusions, by definition, are “fixed false beliefs.” Challenging a patient regarding their delusions is likely pointless, as the patients’ minds cannot be changed.
For cases of primary delusional infestation, the goal generally is to decrease the patient’s preoccupation with the delusion and to improve social and occupational functions. Treatment with antipsychotic drugs is usually helpful–if patients will comply with treatment. “I’ve been very unsuccessful” at getting patients to use antipsychotics, he said.
A good treatment option is risperidone, which has low cost and good efficacy and tolerability. Start with a daily dose of 1 mg/day and gradually increase up to 6 mg daily if necessary, he said.
Establishing a rapport over multiple visits may help with compliance. For example, take a history on the first visit, order laboratory tests at a second visit, perform a biopsy at a third, and then broach the subject of treatment at a subsequent visit, he suggested.
Getting the patient on board with taking risperidone or another antipsychotic may be easier if you ask the patient early on whether they would like treatment for their symptoms in the event a definitive cause for their symptoms can’t be identified, he said.
Dr. Davis reported having no disclosures.
Boston – Patients with delusional infestation often resist referral to a psychiatrist and insist on treatment by a dermatologist, according to Mark D. P. Davis, MD.
Dermatologists, conversely, often don’t want to treat these challenging patients because they aren’t really sure how to manage them, Dr. Davis said during a “practice gaps” session at the American Academy of Dermatology summer meeting.
An important practice gap is the lack of a standard approach to the investigation and management of these patients, he said.
An audience poll showed that respondents were divided as to whether patients presenting with delusional infestation, also known as delusional parasitosis, should be confronted, referred to a psychiatrist, treated with an antipsychotic, or approached in some other way.
The condition, which involves the delusional belief that one’s skin is “infested” with parasites, fibers, or some other materials, can result from another cause, said Dr. Davis of the Mayo Clinic, Rochester, Minn, noting that anxiety, depression, and medications such as opioids or treatments for attention deficit/hyperactivity disorder can contribute to the delusions.
Skin diseases themselves can also cause the sensation of infestation.
A good history should assess medical comorbidities, electrolyte abnormalities, or medication use that might be contributing to the problem.
For dermatologists who are not well-versed in “picking up on psychiatric comorbidities,” validated measures can be useful. Tools such as the Personal Health Questionnaire (PHQ)-9 for depression and Generalized Anxiety Disorder (GAD)-7 can be useful, Dr. Davis said.
However, delusions, by definition, are “fixed false beliefs.” Challenging a patient regarding their delusions is likely pointless, as the patients’ minds cannot be changed.
For cases of primary delusional infestation, the goal generally is to decrease the patient’s preoccupation with the delusion and to improve social and occupational functions. Treatment with antipsychotic drugs is usually helpful–if patients will comply with treatment. “I’ve been very unsuccessful” at getting patients to use antipsychotics, he said.
A good treatment option is risperidone, which has low cost and good efficacy and tolerability. Start with a daily dose of 1 mg/day and gradually increase up to 6 mg daily if necessary, he said.
Establishing a rapport over multiple visits may help with compliance. For example, take a history on the first visit, order laboratory tests at a second visit, perform a biopsy at a third, and then broach the subject of treatment at a subsequent visit, he suggested.
Getting the patient on board with taking risperidone or another antipsychotic may be easier if you ask the patient early on whether they would like treatment for their symptoms in the event a definitive cause for their symptoms can’t be identified, he said.
Dr. Davis reported having no disclosures.
Boston – Patients with delusional infestation often resist referral to a psychiatrist and insist on treatment by a dermatologist, according to Mark D. P. Davis, MD.
Dermatologists, conversely, often don’t want to treat these challenging patients because they aren’t really sure how to manage them, Dr. Davis said during a “practice gaps” session at the American Academy of Dermatology summer meeting.
An important practice gap is the lack of a standard approach to the investigation and management of these patients, he said.
An audience poll showed that respondents were divided as to whether patients presenting with delusional infestation, also known as delusional parasitosis, should be confronted, referred to a psychiatrist, treated with an antipsychotic, or approached in some other way.
The condition, which involves the delusional belief that one’s skin is “infested” with parasites, fibers, or some other materials, can result from another cause, said Dr. Davis of the Mayo Clinic, Rochester, Minn, noting that anxiety, depression, and medications such as opioids or treatments for attention deficit/hyperactivity disorder can contribute to the delusions.
Skin diseases themselves can also cause the sensation of infestation.
A good history should assess medical comorbidities, electrolyte abnormalities, or medication use that might be contributing to the problem.
For dermatologists who are not well-versed in “picking up on psychiatric comorbidities,” validated measures can be useful. Tools such as the Personal Health Questionnaire (PHQ)-9 for depression and Generalized Anxiety Disorder (GAD)-7 can be useful, Dr. Davis said.
However, delusions, by definition, are “fixed false beliefs.” Challenging a patient regarding their delusions is likely pointless, as the patients’ minds cannot be changed.
For cases of primary delusional infestation, the goal generally is to decrease the patient’s preoccupation with the delusion and to improve social and occupational functions. Treatment with antipsychotic drugs is usually helpful–if patients will comply with treatment. “I’ve been very unsuccessful” at getting patients to use antipsychotics, he said.
A good treatment option is risperidone, which has low cost and good efficacy and tolerability. Start with a daily dose of 1 mg/day and gradually increase up to 6 mg daily if necessary, he said.
Establishing a rapport over multiple visits may help with compliance. For example, take a history on the first visit, order laboratory tests at a second visit, perform a biopsy at a third, and then broach the subject of treatment at a subsequent visit, he suggested.
Getting the patient on board with taking risperidone or another antipsychotic may be easier if you ask the patient early on whether they would like treatment for their symptoms in the event a definitive cause for their symptoms can’t be identified, he said.
Dr. Davis reported having no disclosures.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
CANDLE syndrome case highlights key features of this type 1 interferonopathy
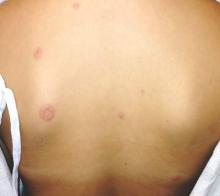
BOSTON – Annular erythematous plaques in a child with fever and dense, atypical, mixed mononuclear and neutrophilic dermal infiltrate on biopsy could signal the recently described autoinflammatory disorder known as CANDLE Syndrome, according to Raegan Hunt, MD.
CANDLE, which stands for chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature, is a proteasome-associated autoinflammatory syndrome characterized by dysregulation of type 1 interferon signaling, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital said during a presentation at the American Academy of Dermatology summer meeting.
She described a case involving a 12-year-old girl with erythematous, warm, tender, nonpruritic papules and plaques on the superior chest, neck, and upper back. Most had an annular configuration with central clearing and occasionally central duskiness and ulceration.
The child also sometimes had violaceous swelling of the eyelid, nodules on the ear, and figurate erythematous plaques on the upper arm.
“She’s been having these since she was an infant in periodic bursts, and carried a diagnosis of annular erythema of infancy,” Dr. Hunt said.
Other symptoms included recurrent fevers, myalgias, transient and migratory arthritis, elevated C-reactive protein and erythrocyte sedimentation rate, and aseptic meningitis requiring hospitalization.
She had no family history of autoimmune disease, immune deficiency, or other genetic diseases, Dr. Hunt said.
“She was treated with methotrexate and prednisone, as well as IVIG [intravenous immunoglobulin] every 4 weeks. Her prednisone was never weaned successfully below 0.8 mg/kg. She had complications of chronic corticosteroid disease, including many fractures,” she said.
A biopsy showed a dense infiltrate of atypical, mixed mononuclear and neutrophilic dermal infiltrate.
“These taken together are suggestive of ... CANDLE,” she said.
Genetic testing confirmed the presence of a compound heterozygous mutation in the proteasome subunit beta type 9, or PSMB8 gene.
Key features of CANDLE syndrome, first described in a 2010 article and further described by Liu, et al in 2012, include early disease onset, recurrent febrile episodes, skin lesions – including recurrent attacks of erythematous annular plaques and violaceous swelling of the eyelids, delayed physical development, progressive lipodystrophy, arthralgias, systemic inflammation, and aseptic meningitis episodes.
“Our patient did have very, very subtle lipodystrophy, but overall not as quick onset as some other [cases in children] that have been described,” Dr. Hunt said.
The type 1 interferonopathy is of autosomal recessive inheritance, and involves either homozygous or compound heterozygous mutations in PSMB8, as found in this patient. PSMB8 is an immunoproteasome unit involved in proteolysis and maintenance of cell homeostasis, she explained.
Treatment of the syndrome involves high-dose corticosteroids (usually 1-2 mg/kg day), which helps improve cutaneous eruptions, joint pain, and fever, but it is important to remember that disease flares can occur with tapering, Dr. Hunt noted.
Response is typically poor in patients treated with steroid-sparing agents, such as methotrexate, cyclosporine, azathioprine, or IVIG, as well as with tumor necrosis factor–alpha inhibitors and interleukin-1 receptor antagonists.
“But there is an interesting molecule on the horizon for possibly treating this more specifically and that’s JAK inhibitors,” she said, noting that a National Institutes of Health compassionate use clinical research trial is evaluating the JAK 1/2 inhibitor, baricitinib.
Dr. Hunt reported having no relevant disclosures.
BOSTON – Annular erythematous plaques in a child with fever and dense, atypical, mixed mononuclear and neutrophilic dermal infiltrate on biopsy could signal the recently described autoinflammatory disorder known as CANDLE Syndrome, according to Raegan Hunt, MD.
CANDLE, which stands for chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature, is a proteasome-associated autoinflammatory syndrome characterized by dysregulation of type 1 interferon signaling, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital said during a presentation at the American Academy of Dermatology summer meeting.
She described a case involving a 12-year-old girl with erythematous, warm, tender, nonpruritic papules and plaques on the superior chest, neck, and upper back. Most had an annular configuration with central clearing and occasionally central duskiness and ulceration.
The child also sometimes had violaceous swelling of the eyelid, nodules on the ear, and figurate erythematous plaques on the upper arm.
“She’s been having these since she was an infant in periodic bursts, and carried a diagnosis of annular erythema of infancy,” Dr. Hunt said.
Other symptoms included recurrent fevers, myalgias, transient and migratory arthritis, elevated C-reactive protein and erythrocyte sedimentation rate, and aseptic meningitis requiring hospitalization.
She had no family history of autoimmune disease, immune deficiency, or other genetic diseases, Dr. Hunt said.
“She was treated with methotrexate and prednisone, as well as IVIG [intravenous immunoglobulin] every 4 weeks. Her prednisone was never weaned successfully below 0.8 mg/kg. She had complications of chronic corticosteroid disease, including many fractures,” she said.
A biopsy showed a dense infiltrate of atypical, mixed mononuclear and neutrophilic dermal infiltrate.
“These taken together are suggestive of ... CANDLE,” she said.
Genetic testing confirmed the presence of a compound heterozygous mutation in the proteasome subunit beta type 9, or PSMB8 gene.
Key features of CANDLE syndrome, first described in a 2010 article and further described by Liu, et al in 2012, include early disease onset, recurrent febrile episodes, skin lesions – including recurrent attacks of erythematous annular plaques and violaceous swelling of the eyelids, delayed physical development, progressive lipodystrophy, arthralgias, systemic inflammation, and aseptic meningitis episodes.
“Our patient did have very, very subtle lipodystrophy, but overall not as quick onset as some other [cases in children] that have been described,” Dr. Hunt said.
The type 1 interferonopathy is of autosomal recessive inheritance, and involves either homozygous or compound heterozygous mutations in PSMB8, as found in this patient. PSMB8 is an immunoproteasome unit involved in proteolysis and maintenance of cell homeostasis, she explained.
Treatment of the syndrome involves high-dose corticosteroids (usually 1-2 mg/kg day), which helps improve cutaneous eruptions, joint pain, and fever, but it is important to remember that disease flares can occur with tapering, Dr. Hunt noted.
Response is typically poor in patients treated with steroid-sparing agents, such as methotrexate, cyclosporine, azathioprine, or IVIG, as well as with tumor necrosis factor–alpha inhibitors and interleukin-1 receptor antagonists.
“But there is an interesting molecule on the horizon for possibly treating this more specifically and that’s JAK inhibitors,” she said, noting that a National Institutes of Health compassionate use clinical research trial is evaluating the JAK 1/2 inhibitor, baricitinib.
Dr. Hunt reported having no relevant disclosures.
BOSTON – Annular erythematous plaques in a child with fever and dense, atypical, mixed mononuclear and neutrophilic dermal infiltrate on biopsy could signal the recently described autoinflammatory disorder known as CANDLE Syndrome, according to Raegan Hunt, MD.
CANDLE, which stands for chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature, is a proteasome-associated autoinflammatory syndrome characterized by dysregulation of type 1 interferon signaling, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital said during a presentation at the American Academy of Dermatology summer meeting.
She described a case involving a 12-year-old girl with erythematous, warm, tender, nonpruritic papules and plaques on the superior chest, neck, and upper back. Most had an annular configuration with central clearing and occasionally central duskiness and ulceration.
The child also sometimes had violaceous swelling of the eyelid, nodules on the ear, and figurate erythematous plaques on the upper arm.
“She’s been having these since she was an infant in periodic bursts, and carried a diagnosis of annular erythema of infancy,” Dr. Hunt said.
Other symptoms included recurrent fevers, myalgias, transient and migratory arthritis, elevated C-reactive protein and erythrocyte sedimentation rate, and aseptic meningitis requiring hospitalization.
She had no family history of autoimmune disease, immune deficiency, or other genetic diseases, Dr. Hunt said.
“She was treated with methotrexate and prednisone, as well as IVIG [intravenous immunoglobulin] every 4 weeks. Her prednisone was never weaned successfully below 0.8 mg/kg. She had complications of chronic corticosteroid disease, including many fractures,” she said.
A biopsy showed a dense infiltrate of atypical, mixed mononuclear and neutrophilic dermal infiltrate.
“These taken together are suggestive of ... CANDLE,” she said.
Genetic testing confirmed the presence of a compound heterozygous mutation in the proteasome subunit beta type 9, or PSMB8 gene.
Key features of CANDLE syndrome, first described in a 2010 article and further described by Liu, et al in 2012, include early disease onset, recurrent febrile episodes, skin lesions – including recurrent attacks of erythematous annular plaques and violaceous swelling of the eyelids, delayed physical development, progressive lipodystrophy, arthralgias, systemic inflammation, and aseptic meningitis episodes.
“Our patient did have very, very subtle lipodystrophy, but overall not as quick onset as some other [cases in children] that have been described,” Dr. Hunt said.
The type 1 interferonopathy is of autosomal recessive inheritance, and involves either homozygous or compound heterozygous mutations in PSMB8, as found in this patient. PSMB8 is an immunoproteasome unit involved in proteolysis and maintenance of cell homeostasis, she explained.
Treatment of the syndrome involves high-dose corticosteroids (usually 1-2 mg/kg day), which helps improve cutaneous eruptions, joint pain, and fever, but it is important to remember that disease flares can occur with tapering, Dr. Hunt noted.
Response is typically poor in patients treated with steroid-sparing agents, such as methotrexate, cyclosporine, azathioprine, or IVIG, as well as with tumor necrosis factor–alpha inhibitors and interleukin-1 receptor antagonists.
“But there is an interesting molecule on the horizon for possibly treating this more specifically and that’s JAK inhibitors,” she said, noting that a National Institutes of Health compassionate use clinical research trial is evaluating the JAK 1/2 inhibitor, baricitinib.
Dr. Hunt reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
Key clinical point: Annular erythematous plaques in a child with fever and dense, atypical, mixed mononuclear and neutrophilic dermal infiltrate on biopsy could signal the recently described autoinflammatory disorder known as CANDLE syndrome.
Major finding: Genetic testing confirmed the presence of a compound heterozygous mutation in the proteasome subunit beta type 9, or PSMB8 gene.
Data source: Expert analysis – case description.
Disclosures: Dr. Hunt reported having no disclosures.
SHARE initiative releases consensus-based JDM management recommendations
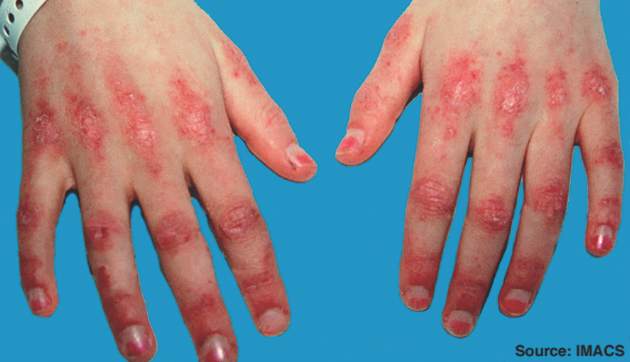
Early and aggressive therapy may prevent or stabilize organ damage and disease-related complications in patients with juvenile dermatomyositis, according to new consensus-based recommendations for the management of the disorder.
Overall, the recommendations – a project of the Single Hub and Access Point for Pediatric Rheumatology in Europe (SHARE) initiative – include 7 overarching principles, 33 recommendations on diagnosis, and 19 recommendations on therapy that were accepted with greater than 80% agreement among experts. The recommendations, which address the assessment of skin, muscle, and major organ involvement and treatment suggestions at disease onset and in refractory cases, fill a void in the area of evidence-based guidelines for this rare disease within the group of pediatric rheumatic diseases, according to Felicitas Bellutti Enders, MD, of University Medical Center Utrecht (the Netherlands) and her colleagues.
“Clear recommendations can help clinicians in the care of patients with JDM [juvenile dermatomyositis] as no international consensus regarding diagnosis and treatment is currently available and management therefore varies,” they wrote (Ann Rheum Dis. 2016 Aug 11. doi: 10.1136/annrheumdis-2016-209247).
The consensus committee, including 19 experienced pediatric rheumatologists and 2 experts in pediatric exercise physiology and physical therapy, developed recommendations based on a validated systematic literature review. The recommendations were evaluated by an online survey and then discussed at two subsequent consensus meetings.
The overarching principles accepted by more than 80% of respondents (100% in all but number 4 below), hold that:
1) All children with suspected idiopathic inflammatory myopathies should be referred to a specialized center.
2) High-risk patients need immediate/urgent referral to a specialized center.
3) Patient-/parent-reported outcomes measures are helpful when assessing disease activity and should be used at diagnosis and during disease monitoring.
4) Validated tools such as the Childhood Health Assessment Questionnaire, patient/parent visual analog scale and Juvenile Dermatomyositis Multi-dimensional Assessment Report should be used to measure health status.
5) All children with JDM should have disease activity (muscle, skin, major organ) assessed regularly in a standardized way, using tools such as the Disease Activity Score.
6) All children with JDM should have disease damage assessed at least yearly using a standardized disease damage measure, such as the Myositis Damage index.
7) All patients with JDM should have the opportunity to be registered within a research registry/repository such as the Euromyositis registry.
The consensus process also yielded both general and specific recommendations for diagnosis and management – also with 100% agreement in almost all cases – addressing investigations to consider in all those with a JDM diagnosis (muscle enzymes, full blood count, renal function and liver function tests, and infection screen – to name a few), ways that MRI can be used (both for diagnosis and disease activity monitoring), the use of muscle biopsy (use in all cases involving atypical presentation), and assessment of calcinosis and skin, lung, and cardiac involvement.
Treatment recommendations address sun protection (encourage routine use of sunblock), exercise (should be safe and appropriate and monitored by a physiotherapist), corticosteroid use (administer systematically either orally or intravenously in moderate to severe JDM and wean as the patient shows clinical improvement), use of intravenous immunoglobulin (a useful adjunct for resistant disease, especially when skin features are prominent), and the use of anti–tumor necrosis factor therapies (consider in refractory disease; infliximab or adalimumab are favored over etanercept), as well as other treatments.
The recommendations state that while there is no high-level evidence of when to stop therapy, consideration may be given to withdrawing treatment if a patient has been off steroids and in remission on methotrexate or an alternative disease-modifying antirheumatic drug [DMARD] for at least 1 year.
The management of JDM is complex and warrants a multidisciplinary approach, involving physiotherapists, specialist nurses, pediatric rheumatologists, and other specialists as needed, the authors wrote, noting that the mainstay of therapy is high-dose corticosteroids initially in combination with DMARDS.
However, the evidence base for treatment is limited and “often confined to small case-controlled studies, with the exception of two randomized controlled trials,” they said.
Similarly, there is no high-level evidence regarding when to stop immunosuppressive therapy, but the expert group suggested considering the withdrawal of methotrexate or alternative DMARD once the patient is in remission and off steroids for a minimum of 1 year.
The authors concluded that “this SHARE initiative is based on expert opinion informed by the best available evidence and provides recommendations ... with a view to improving the outcome for patients with JDM in Europe.
“It will now be important to broaden discussion and test acceptability of these to the wider community,” they wrote.
The SHARE initiative is funded by a grant from the European Agency for Health and Consumers. Dr. Enders disclosed a relationship with the Valeria e Ettore Bossi Foundation. Her coauthors reported financial or other relationships with Roche/Chugai, AbbVie, Pfizer, Novartis, Bristol-Myers Squibb, SOBI, Medac, The Myositis Association, Neopharm, GlaxoSmithKline, and/or Genzyme.
The consensus-based recommendations for the management of juvenile dermatomyositis published in Annals of Rheumatic Diseases again highlight the ability of the European and Canadian investigators to work together and pool the information from a large number of centers. Similar efforts to pool data and encourage the development of protocols to optimize care are occurring in the United States under the auspices of CARRA (the Childhood Arthritis and Rheumatology Research Alliance).
These working groups represent an important first step toward standardizing optimal care for children with rheumatic diseases. However, the protocols put forth by both groups suffer from failure to address the diversity of presentations within their diseases and a resultant lack of specificity in their recommendations. The authors of these guidelines make specific recommendations regarding diagnosis, but these fail to encompass the range of weakness that may be present initially. Is it truly appropriate to bolus every child diagnosed with dermatomyositis with high-dose corticosteroids and begin methotrexate? No mention is made of the hypertension, pancreatitis, or systemic infection that might result.
The guidelines and protocols being promulgated in pediatric rheumatology continue to suffer from the grouping of children with diverse disease presentations and probably diverse diseases under a single diagnosis. Charles Spencer and colleagues described the highly variable course of juvenile dermatomyositis and the presence of distinct subsets of patients more than 30 years ago (J Pediatr. 1984 Sep;105[3]:399-408), but this diversity is not reflected in the current protocol. Methods for better characterizing our patients based on gene activation and cytokine profiles have been developed. More effort should be placed on accurate characterization of our patients with many diseases before we attempt to treat them all the same way.
Thomas J.A. Lehman, MD, is chief of the division of pediatric rheumatology at the Hospital for Special Surgery, and professor of clinical pediatrics at Weill Medical Center, Cornell University, New York. He has no relevant disclosures.
The consensus-based recommendations for the management of juvenile dermatomyositis published in Annals of Rheumatic Diseases again highlight the ability of the European and Canadian investigators to work together and pool the information from a large number of centers. Similar efforts to pool data and encourage the development of protocols to optimize care are occurring in the United States under the auspices of CARRA (the Childhood Arthritis and Rheumatology Research Alliance).
These working groups represent an important first step toward standardizing optimal care for children with rheumatic diseases. However, the protocols put forth by both groups suffer from failure to address the diversity of presentations within their diseases and a resultant lack of specificity in their recommendations. The authors of these guidelines make specific recommendations regarding diagnosis, but these fail to encompass the range of weakness that may be present initially. Is it truly appropriate to bolus every child diagnosed with dermatomyositis with high-dose corticosteroids and begin methotrexate? No mention is made of the hypertension, pancreatitis, or systemic infection that might result.
The guidelines and protocols being promulgated in pediatric rheumatology continue to suffer from the grouping of children with diverse disease presentations and probably diverse diseases under a single diagnosis. Charles Spencer and colleagues described the highly variable course of juvenile dermatomyositis and the presence of distinct subsets of patients more than 30 years ago (J Pediatr. 1984 Sep;105[3]:399-408), but this diversity is not reflected in the current protocol. Methods for better characterizing our patients based on gene activation and cytokine profiles have been developed. More effort should be placed on accurate characterization of our patients with many diseases before we attempt to treat them all the same way.
Thomas J.A. Lehman, MD, is chief of the division of pediatric rheumatology at the Hospital for Special Surgery, and professor of clinical pediatrics at Weill Medical Center, Cornell University, New York. He has no relevant disclosures.
The consensus-based recommendations for the management of juvenile dermatomyositis published in Annals of Rheumatic Diseases again highlight the ability of the European and Canadian investigators to work together and pool the information from a large number of centers. Similar efforts to pool data and encourage the development of protocols to optimize care are occurring in the United States under the auspices of CARRA (the Childhood Arthritis and Rheumatology Research Alliance).
These working groups represent an important first step toward standardizing optimal care for children with rheumatic diseases. However, the protocols put forth by both groups suffer from failure to address the diversity of presentations within their diseases and a resultant lack of specificity in their recommendations. The authors of these guidelines make specific recommendations regarding diagnosis, but these fail to encompass the range of weakness that may be present initially. Is it truly appropriate to bolus every child diagnosed with dermatomyositis with high-dose corticosteroids and begin methotrexate? No mention is made of the hypertension, pancreatitis, or systemic infection that might result.
The guidelines and protocols being promulgated in pediatric rheumatology continue to suffer from the grouping of children with diverse disease presentations and probably diverse diseases under a single diagnosis. Charles Spencer and colleagues described the highly variable course of juvenile dermatomyositis and the presence of distinct subsets of patients more than 30 years ago (J Pediatr. 1984 Sep;105[3]:399-408), but this diversity is not reflected in the current protocol. Methods for better characterizing our patients based on gene activation and cytokine profiles have been developed. More effort should be placed on accurate characterization of our patients with many diseases before we attempt to treat them all the same way.
Thomas J.A. Lehman, MD, is chief of the division of pediatric rheumatology at the Hospital for Special Surgery, and professor of clinical pediatrics at Weill Medical Center, Cornell University, New York. He has no relevant disclosures.
Early and aggressive therapy may prevent or stabilize organ damage and disease-related complications in patients with juvenile dermatomyositis, according to new consensus-based recommendations for the management of the disorder.
Overall, the recommendations – a project of the Single Hub and Access Point for Pediatric Rheumatology in Europe (SHARE) initiative – include 7 overarching principles, 33 recommendations on diagnosis, and 19 recommendations on therapy that were accepted with greater than 80% agreement among experts. The recommendations, which address the assessment of skin, muscle, and major organ involvement and treatment suggestions at disease onset and in refractory cases, fill a void in the area of evidence-based guidelines for this rare disease within the group of pediatric rheumatic diseases, according to Felicitas Bellutti Enders, MD, of University Medical Center Utrecht (the Netherlands) and her colleagues.
“Clear recommendations can help clinicians in the care of patients with JDM [juvenile dermatomyositis] as no international consensus regarding diagnosis and treatment is currently available and management therefore varies,” they wrote (Ann Rheum Dis. 2016 Aug 11. doi: 10.1136/annrheumdis-2016-209247).
The consensus committee, including 19 experienced pediatric rheumatologists and 2 experts in pediatric exercise physiology and physical therapy, developed recommendations based on a validated systematic literature review. The recommendations were evaluated by an online survey and then discussed at two subsequent consensus meetings.
The overarching principles accepted by more than 80% of respondents (100% in all but number 4 below), hold that:
1) All children with suspected idiopathic inflammatory myopathies should be referred to a specialized center.
2) High-risk patients need immediate/urgent referral to a specialized center.
3) Patient-/parent-reported outcomes measures are helpful when assessing disease activity and should be used at diagnosis and during disease monitoring.
4) Validated tools such as the Childhood Health Assessment Questionnaire, patient/parent visual analog scale and Juvenile Dermatomyositis Multi-dimensional Assessment Report should be used to measure health status.
5) All children with JDM should have disease activity (muscle, skin, major organ) assessed regularly in a standardized way, using tools such as the Disease Activity Score.
6) All children with JDM should have disease damage assessed at least yearly using a standardized disease damage measure, such as the Myositis Damage index.
7) All patients with JDM should have the opportunity to be registered within a research registry/repository such as the Euromyositis registry.
The consensus process also yielded both general and specific recommendations for diagnosis and management – also with 100% agreement in almost all cases – addressing investigations to consider in all those with a JDM diagnosis (muscle enzymes, full blood count, renal function and liver function tests, and infection screen – to name a few), ways that MRI can be used (both for diagnosis and disease activity monitoring), the use of muscle biopsy (use in all cases involving atypical presentation), and assessment of calcinosis and skin, lung, and cardiac involvement.
Treatment recommendations address sun protection (encourage routine use of sunblock), exercise (should be safe and appropriate and monitored by a physiotherapist), corticosteroid use (administer systematically either orally or intravenously in moderate to severe JDM and wean as the patient shows clinical improvement), use of intravenous immunoglobulin (a useful adjunct for resistant disease, especially when skin features are prominent), and the use of anti–tumor necrosis factor therapies (consider in refractory disease; infliximab or adalimumab are favored over etanercept), as well as other treatments.
The recommendations state that while there is no high-level evidence of when to stop therapy, consideration may be given to withdrawing treatment if a patient has been off steroids and in remission on methotrexate or an alternative disease-modifying antirheumatic drug [DMARD] for at least 1 year.
The management of JDM is complex and warrants a multidisciplinary approach, involving physiotherapists, specialist nurses, pediatric rheumatologists, and other specialists as needed, the authors wrote, noting that the mainstay of therapy is high-dose corticosteroids initially in combination with DMARDS.
However, the evidence base for treatment is limited and “often confined to small case-controlled studies, with the exception of two randomized controlled trials,” they said.
Similarly, there is no high-level evidence regarding when to stop immunosuppressive therapy, but the expert group suggested considering the withdrawal of methotrexate or alternative DMARD once the patient is in remission and off steroids for a minimum of 1 year.
The authors concluded that “this SHARE initiative is based on expert opinion informed by the best available evidence and provides recommendations ... with a view to improving the outcome for patients with JDM in Europe.
“It will now be important to broaden discussion and test acceptability of these to the wider community,” they wrote.
The SHARE initiative is funded by a grant from the European Agency for Health and Consumers. Dr. Enders disclosed a relationship with the Valeria e Ettore Bossi Foundation. Her coauthors reported financial or other relationships with Roche/Chugai, AbbVie, Pfizer, Novartis, Bristol-Myers Squibb, SOBI, Medac, The Myositis Association, Neopharm, GlaxoSmithKline, and/or Genzyme.
Early and aggressive therapy may prevent or stabilize organ damage and disease-related complications in patients with juvenile dermatomyositis, according to new consensus-based recommendations for the management of the disorder.
Overall, the recommendations – a project of the Single Hub and Access Point for Pediatric Rheumatology in Europe (SHARE) initiative – include 7 overarching principles, 33 recommendations on diagnosis, and 19 recommendations on therapy that were accepted with greater than 80% agreement among experts. The recommendations, which address the assessment of skin, muscle, and major organ involvement and treatment suggestions at disease onset and in refractory cases, fill a void in the area of evidence-based guidelines for this rare disease within the group of pediatric rheumatic diseases, according to Felicitas Bellutti Enders, MD, of University Medical Center Utrecht (the Netherlands) and her colleagues.
“Clear recommendations can help clinicians in the care of patients with JDM [juvenile dermatomyositis] as no international consensus regarding diagnosis and treatment is currently available and management therefore varies,” they wrote (Ann Rheum Dis. 2016 Aug 11. doi: 10.1136/annrheumdis-2016-209247).
The consensus committee, including 19 experienced pediatric rheumatologists and 2 experts in pediatric exercise physiology and physical therapy, developed recommendations based on a validated systematic literature review. The recommendations were evaluated by an online survey and then discussed at two subsequent consensus meetings.
The overarching principles accepted by more than 80% of respondents (100% in all but number 4 below), hold that:
1) All children with suspected idiopathic inflammatory myopathies should be referred to a specialized center.
2) High-risk patients need immediate/urgent referral to a specialized center.
3) Patient-/parent-reported outcomes measures are helpful when assessing disease activity and should be used at diagnosis and during disease monitoring.
4) Validated tools such as the Childhood Health Assessment Questionnaire, patient/parent visual analog scale and Juvenile Dermatomyositis Multi-dimensional Assessment Report should be used to measure health status.
5) All children with JDM should have disease activity (muscle, skin, major organ) assessed regularly in a standardized way, using tools such as the Disease Activity Score.
6) All children with JDM should have disease damage assessed at least yearly using a standardized disease damage measure, such as the Myositis Damage index.
7) All patients with JDM should have the opportunity to be registered within a research registry/repository such as the Euromyositis registry.
The consensus process also yielded both general and specific recommendations for diagnosis and management – also with 100% agreement in almost all cases – addressing investigations to consider in all those with a JDM diagnosis (muscle enzymes, full blood count, renal function and liver function tests, and infection screen – to name a few), ways that MRI can be used (both for diagnosis and disease activity monitoring), the use of muscle biopsy (use in all cases involving atypical presentation), and assessment of calcinosis and skin, lung, and cardiac involvement.
Treatment recommendations address sun protection (encourage routine use of sunblock), exercise (should be safe and appropriate and monitored by a physiotherapist), corticosteroid use (administer systematically either orally or intravenously in moderate to severe JDM and wean as the patient shows clinical improvement), use of intravenous immunoglobulin (a useful adjunct for resistant disease, especially when skin features are prominent), and the use of anti–tumor necrosis factor therapies (consider in refractory disease; infliximab or adalimumab are favored over etanercept), as well as other treatments.
The recommendations state that while there is no high-level evidence of when to stop therapy, consideration may be given to withdrawing treatment if a patient has been off steroids and in remission on methotrexate or an alternative disease-modifying antirheumatic drug [DMARD] for at least 1 year.
The management of JDM is complex and warrants a multidisciplinary approach, involving physiotherapists, specialist nurses, pediatric rheumatologists, and other specialists as needed, the authors wrote, noting that the mainstay of therapy is high-dose corticosteroids initially in combination with DMARDS.
However, the evidence base for treatment is limited and “often confined to small case-controlled studies, with the exception of two randomized controlled trials,” they said.
Similarly, there is no high-level evidence regarding when to stop immunosuppressive therapy, but the expert group suggested considering the withdrawal of methotrexate or alternative DMARD once the patient is in remission and off steroids for a minimum of 1 year.
The authors concluded that “this SHARE initiative is based on expert opinion informed by the best available evidence and provides recommendations ... with a view to improving the outcome for patients with JDM in Europe.
“It will now be important to broaden discussion and test acceptability of these to the wider community,” they wrote.
The SHARE initiative is funded by a grant from the European Agency for Health and Consumers. Dr. Enders disclosed a relationship with the Valeria e Ettore Bossi Foundation. Her coauthors reported financial or other relationships with Roche/Chugai, AbbVie, Pfizer, Novartis, Bristol-Myers Squibb, SOBI, Medac, The Myositis Association, Neopharm, GlaxoSmithKline, and/or Genzyme.
FROM ANNALS OF THE RHEUMATIC DISEASES
Nonpharmacologic AD therapy: Strongest evidence supports moisturizers
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
Food allergy testing only rarely needed for AD patients
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
DEET and picaridin safely protect against insect bites
BOSTON – Insect repellents containing DEET or picaridin are safe when used properly, and are important for bite protection in children, according to Mercedes E. Gonzalez, MD.
Insect bite reactions are common in children aged 2-10 years, and the emergence of Zika virus raises new concerns about the dangers of mosquito bites, in particular; the World Health Organization has declared Zika-related effects – namely microcephaly and Guillain-Barré syndrome – to be a “public health emergency of international concern.”
In children, illness associated with Zika virus is generally mild, but can include fever, rash, conjunctivitis, and/or arthralgia, Dr. Gonzalez said at the American Academy of Dermatology summer meeting.
DEET, used since 1957, is effective against mosquitoes, black flies, ticks, mites, and land leeches. It works by forming a vapor barrier that deters insects from coming into contact with the skin. The barrier extends about 4 cm from the skin. DEET can also be used on clothing but may cause damage to spandex, rayon, acetate, and leather, and can dissolve plastic and vinyl.
Although it is available in concentrations of 5%-100%, concentrations of 10%-35% provide adequate protection in most situations, said Dr. Gonzalez of the University of Miami.
Animal studies using large doses have shown that DEET is not a specific neurotoxin, and while there have been case reports of central nervous system toxicity in humans, there is no link to DEET dose or mechanistic pathway. Reported deaths have involved intentional ingestion and overuse or incorrect use of products, she said.
In fact, safety concerns are so minimal that the Environmental Protection Agency removed labels indicating caution in children, and the American Academy of Pediatrics recommends the use of DEET for preventing insect bites in children older than age 2 months, and in pregnant and lactating women.
One DEET safety concern, however, is flammability. Both DEET and the aerosol vehicle used in some DEET-containing products, are flammable, so caution is warranted, she said. Occlusion following use of DEET should also be avoided as it can increase absorption, and the product should be washed off after use.
Picaridin is another insect repellent that, like DEET, forms a vapor barrier to deter insects from getting close to the skin and biting, and can be used on both the skin and clothing, but it does not damage plastics or fabrics.
It has similar efficacy as DEET, and has a number of advantages over DEET in that it is odorless and does not feel sticky or greasy when applied. It has not been reported to cause any serious toxicity or mutagenesis.
Picaridin – which is effective against mosquitoes, dog and deer ticks, chiggers, and flies – has been used in Australia since 1998, and in the United States since 2005. That year, the Centers for Disease Control and Prevention recommended that it be used to protect against West Nile virus, and the World Health Organization said it was the best agent for preventing malaria, Dr. Gonzalez noted.
“So when [patients] ask about the best insect repellent, for most situations I do recommend DEET or picaridin, at 10%-25% for DEET, or 7%-15% for picaridin,” she said. She encourages people to read labels, noting that the EPA is encouraging the use of “repellency awareness” labels on insect repellents to inform the consumer whether it prevents against mosquitoes and/or ticks, and for how long.
It helps to provide specific recommendations, providing pictures and circling those that are recommended. Selling the products in the office is also a good idea to make sure patients “leave with the right product,” she said.
Also, advise patients about what to avoid, such as products that contain blends of natural plant oils, which have been shown to be ineffective, providing less than an hour of protection, she said.
Dr. Gonzalez also advises against the use of combination insect repellent/sunscreen products. One reason is that sunscreen needs frequent reapplication, while insect repellent does not. Further, studies have demonstrated that using sunscreen over insect repellent dramatically increases the percutaneous absorption of DEET, and reduces the SPF of the sunscreen. If both are needed, sunscreen should be applied first to reduce transdermal penetration of the active insect repellent ingredient, and should be reapplied every 2 hours, she said.
“Proper insect repellent use is just one part of protection,” she added.
Other measures that should be encouraged include the use of protective clothing, such as light cotton long sleeves and pants; avoidance of clothing with bright colors or flowery prints; avoidance of scented soaps, perfumes, or hair spray; removal of mosquito habitats by eliminating any standing water, covering gaps in doors, using screens and nets; and, if possible, staying indoors at sunrise, sunset, and early evening when mosquitoes are most active.
Dr. Gonzalez noted that many free resources are available online, including a tool at the epa.gov site that helps in selection of an appropriate product for one’s specific needs.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Insect repellents containing DEET or picaridin are safe when used properly, and are important for bite protection in children, according to Mercedes E. Gonzalez, MD.
Insect bite reactions are common in children aged 2-10 years, and the emergence of Zika virus raises new concerns about the dangers of mosquito bites, in particular; the World Health Organization has declared Zika-related effects – namely microcephaly and Guillain-Barré syndrome – to be a “public health emergency of international concern.”
In children, illness associated with Zika virus is generally mild, but can include fever, rash, conjunctivitis, and/or arthralgia, Dr. Gonzalez said at the American Academy of Dermatology summer meeting.
DEET, used since 1957, is effective against mosquitoes, black flies, ticks, mites, and land leeches. It works by forming a vapor barrier that deters insects from coming into contact with the skin. The barrier extends about 4 cm from the skin. DEET can also be used on clothing but may cause damage to spandex, rayon, acetate, and leather, and can dissolve plastic and vinyl.
Although it is available in concentrations of 5%-100%, concentrations of 10%-35% provide adequate protection in most situations, said Dr. Gonzalez of the University of Miami.
Animal studies using large doses have shown that DEET is not a specific neurotoxin, and while there have been case reports of central nervous system toxicity in humans, there is no link to DEET dose or mechanistic pathway. Reported deaths have involved intentional ingestion and overuse or incorrect use of products, she said.
In fact, safety concerns are so minimal that the Environmental Protection Agency removed labels indicating caution in children, and the American Academy of Pediatrics recommends the use of DEET for preventing insect bites in children older than age 2 months, and in pregnant and lactating women.
One DEET safety concern, however, is flammability. Both DEET and the aerosol vehicle used in some DEET-containing products, are flammable, so caution is warranted, she said. Occlusion following use of DEET should also be avoided as it can increase absorption, and the product should be washed off after use.
Picaridin is another insect repellent that, like DEET, forms a vapor barrier to deter insects from getting close to the skin and biting, and can be used on both the skin and clothing, but it does not damage plastics or fabrics.
It has similar efficacy as DEET, and has a number of advantages over DEET in that it is odorless and does not feel sticky or greasy when applied. It has not been reported to cause any serious toxicity or mutagenesis.
Picaridin – which is effective against mosquitoes, dog and deer ticks, chiggers, and flies – has been used in Australia since 1998, and in the United States since 2005. That year, the Centers for Disease Control and Prevention recommended that it be used to protect against West Nile virus, and the World Health Organization said it was the best agent for preventing malaria, Dr. Gonzalez noted.
“So when [patients] ask about the best insect repellent, for most situations I do recommend DEET or picaridin, at 10%-25% for DEET, or 7%-15% for picaridin,” she said. She encourages people to read labels, noting that the EPA is encouraging the use of “repellency awareness” labels on insect repellents to inform the consumer whether it prevents against mosquitoes and/or ticks, and for how long.
It helps to provide specific recommendations, providing pictures and circling those that are recommended. Selling the products in the office is also a good idea to make sure patients “leave with the right product,” she said.
Also, advise patients about what to avoid, such as products that contain blends of natural plant oils, which have been shown to be ineffective, providing less than an hour of protection, she said.
Dr. Gonzalez also advises against the use of combination insect repellent/sunscreen products. One reason is that sunscreen needs frequent reapplication, while insect repellent does not. Further, studies have demonstrated that using sunscreen over insect repellent dramatically increases the percutaneous absorption of DEET, and reduces the SPF of the sunscreen. If both are needed, sunscreen should be applied first to reduce transdermal penetration of the active insect repellent ingredient, and should be reapplied every 2 hours, she said.
“Proper insect repellent use is just one part of protection,” she added.
Other measures that should be encouraged include the use of protective clothing, such as light cotton long sleeves and pants; avoidance of clothing with bright colors or flowery prints; avoidance of scented soaps, perfumes, or hair spray; removal of mosquito habitats by eliminating any standing water, covering gaps in doors, using screens and nets; and, if possible, staying indoors at sunrise, sunset, and early evening when mosquitoes are most active.
Dr. Gonzalez noted that many free resources are available online, including a tool at the epa.gov site that helps in selection of an appropriate product for one’s specific needs.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Insect repellents containing DEET or picaridin are safe when used properly, and are important for bite protection in children, according to Mercedes E. Gonzalez, MD.
Insect bite reactions are common in children aged 2-10 years, and the emergence of Zika virus raises new concerns about the dangers of mosquito bites, in particular; the World Health Organization has declared Zika-related effects – namely microcephaly and Guillain-Barré syndrome – to be a “public health emergency of international concern.”
In children, illness associated with Zika virus is generally mild, but can include fever, rash, conjunctivitis, and/or arthralgia, Dr. Gonzalez said at the American Academy of Dermatology summer meeting.
DEET, used since 1957, is effective against mosquitoes, black flies, ticks, mites, and land leeches. It works by forming a vapor barrier that deters insects from coming into contact with the skin. The barrier extends about 4 cm from the skin. DEET can also be used on clothing but may cause damage to spandex, rayon, acetate, and leather, and can dissolve plastic and vinyl.
Although it is available in concentrations of 5%-100%, concentrations of 10%-35% provide adequate protection in most situations, said Dr. Gonzalez of the University of Miami.
Animal studies using large doses have shown that DEET is not a specific neurotoxin, and while there have been case reports of central nervous system toxicity in humans, there is no link to DEET dose or mechanistic pathway. Reported deaths have involved intentional ingestion and overuse or incorrect use of products, she said.
In fact, safety concerns are so minimal that the Environmental Protection Agency removed labels indicating caution in children, and the American Academy of Pediatrics recommends the use of DEET for preventing insect bites in children older than age 2 months, and in pregnant and lactating women.
One DEET safety concern, however, is flammability. Both DEET and the aerosol vehicle used in some DEET-containing products, are flammable, so caution is warranted, she said. Occlusion following use of DEET should also be avoided as it can increase absorption, and the product should be washed off after use.
Picaridin is another insect repellent that, like DEET, forms a vapor barrier to deter insects from getting close to the skin and biting, and can be used on both the skin and clothing, but it does not damage plastics or fabrics.
It has similar efficacy as DEET, and has a number of advantages over DEET in that it is odorless and does not feel sticky or greasy when applied. It has not been reported to cause any serious toxicity or mutagenesis.
Picaridin – which is effective against mosquitoes, dog and deer ticks, chiggers, and flies – has been used in Australia since 1998, and in the United States since 2005. That year, the Centers for Disease Control and Prevention recommended that it be used to protect against West Nile virus, and the World Health Organization said it was the best agent for preventing malaria, Dr. Gonzalez noted.
“So when [patients] ask about the best insect repellent, for most situations I do recommend DEET or picaridin, at 10%-25% for DEET, or 7%-15% for picaridin,” she said. She encourages people to read labels, noting that the EPA is encouraging the use of “repellency awareness” labels on insect repellents to inform the consumer whether it prevents against mosquitoes and/or ticks, and for how long.
It helps to provide specific recommendations, providing pictures and circling those that are recommended. Selling the products in the office is also a good idea to make sure patients “leave with the right product,” she said.
Also, advise patients about what to avoid, such as products that contain blends of natural plant oils, which have been shown to be ineffective, providing less than an hour of protection, she said.
Dr. Gonzalez also advises against the use of combination insect repellent/sunscreen products. One reason is that sunscreen needs frequent reapplication, while insect repellent does not. Further, studies have demonstrated that using sunscreen over insect repellent dramatically increases the percutaneous absorption of DEET, and reduces the SPF of the sunscreen. If both are needed, sunscreen should be applied first to reduce transdermal penetration of the active insect repellent ingredient, and should be reapplied every 2 hours, she said.
“Proper insect repellent use is just one part of protection,” she added.
Other measures that should be encouraged include the use of protective clothing, such as light cotton long sleeves and pants; avoidance of clothing with bright colors or flowery prints; avoidance of scented soaps, perfumes, or hair spray; removal of mosquito habitats by eliminating any standing water, covering gaps in doors, using screens and nets; and, if possible, staying indoors at sunrise, sunset, and early evening when mosquitoes are most active.
Dr. Gonzalez noted that many free resources are available online, including a tool at the epa.gov site that helps in selection of an appropriate product for one’s specific needs.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Antipsychotics in pregnancy pose no meaningful malformation risk
Antipsychotic use early in pregnancy does not appear to meaningfully increase the risk of congenital malformations, according to findings from a large Medicaid pregnancy cohort.
A possible exception is with the use of risperidone; a small increase in the risk for malformations seen with the drug should be viewed as “an initial safety signal that will require confirmation in other studies,” Krista F. Huybrechts, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues reported online Aug. 17 in JAMA Psychiatry.
In a nationwide sample of more than 1.3 million pregnant women with a live-born infant between January 1, 2000 and December 31, 2010, the rate of congenital malformations identified within 90 days after delivery was 32.7 per 1,000 births among 1,331,724 women with no antipsychotic (AP) exposure, compared with a rate of 44.5 per 1,000 births among 9,258 women who filled at least one prescription for an atypical AP during their first trimester, and 38.2 per 1,000 in 733 women who filled at least one prescription for a typical AP during their first trimester, the researchers found (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1520).
On unadjusted analyses, an overall risk was seen with atypical AP exposure (relative risk, 1.36), but not with typical AP exposure (RR, 1.17). After adjusting for confounding variables, the risk was not statistically significant (RR, 1.05 and 0.90, respectively). The findings were similar for cardiac malformations, the researchers noted.
When agents were analyzed individually, a small increased risk in overall malformations and cardiac malformations was found with risperidone (RR, 1.26 for each).
“The findings for risperidone should be viewed as an initial safety signal that will require confirmation in other studies,” the researchers wrote.
The study was supported by grants from the National Institutes of Health and the Swiss National Science Foundation. Dr. Huybrechts reported having no financial disclosures. Other authors disclosed receiving consulting fees and/or grant support from AstraZeneca, UCB, Alkermes, Bristol-Myers Squibb/Otsuka, Sunovion, Bayer, Ortho-McNeil Janssen, Pfizer, Forest Laboratories, Cephalon, GlaxoSmithKline, Takeda/Lundbeck, the National Institutes of Health, JDS Therapeutics, Noven Pharmaceuticals, and PamLab.
This “landmark report” by Huybrechts et al involves the largest population of women exposed to APs published to date, and addresses a “major source of concern for women and prescribers,” Katherine L. Wisner, MD, and her colleagues wrote in an editorial.
With the exception of risperidone, which was found to be associated with a 26% increase in the risk of overall and cardiac malformations, and possibly even greater risk among those who filled at least two AP prescriptions – and thus merits further exploration – AP use was found in the study to be safe.
“This study increases the field’s appetite for additional high-quality data on other maternal and offspring outcomes to improve the sophistication of risk-benefit decision making,” the authors wrote.
Nowhere in medicine is there a greater need for personalization of care, particularly when it comes to the treatment of serious mental illness with consideration of “pregnancy physiology and the mother’s capacity to provide sustenance for the growing fetus and newborn,” they wrote.
Dr. Wisner is with Northwestern University, Chicago. She reported that her department at Northwestern received contractual fees for her consultation to Quinn Emanuel Urquhart & Sullivan, LLP, who represented Pfizer Pharmaceutical Company in 2015. A coauthor, Christina Chambers, PhD, reported receiving research funding from AbbVie, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Janssen, Pfizer, Sandoz, and Teva. No other disclosures were reported. Their comments are excerpted from an accompanying editorial (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1538).
This “landmark report” by Huybrechts et al involves the largest population of women exposed to APs published to date, and addresses a “major source of concern for women and prescribers,” Katherine L. Wisner, MD, and her colleagues wrote in an editorial.
With the exception of risperidone, which was found to be associated with a 26% increase in the risk of overall and cardiac malformations, and possibly even greater risk among those who filled at least two AP prescriptions – and thus merits further exploration – AP use was found in the study to be safe.
“This study increases the field’s appetite for additional high-quality data on other maternal and offspring outcomes to improve the sophistication of risk-benefit decision making,” the authors wrote.
Nowhere in medicine is there a greater need for personalization of care, particularly when it comes to the treatment of serious mental illness with consideration of “pregnancy physiology and the mother’s capacity to provide sustenance for the growing fetus and newborn,” they wrote.
Dr. Wisner is with Northwestern University, Chicago. She reported that her department at Northwestern received contractual fees for her consultation to Quinn Emanuel Urquhart & Sullivan, LLP, who represented Pfizer Pharmaceutical Company in 2015. A coauthor, Christina Chambers, PhD, reported receiving research funding from AbbVie, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Janssen, Pfizer, Sandoz, and Teva. No other disclosures were reported. Their comments are excerpted from an accompanying editorial (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1538).
This “landmark report” by Huybrechts et al involves the largest population of women exposed to APs published to date, and addresses a “major source of concern for women and prescribers,” Katherine L. Wisner, MD, and her colleagues wrote in an editorial.
With the exception of risperidone, which was found to be associated with a 26% increase in the risk of overall and cardiac malformations, and possibly even greater risk among those who filled at least two AP prescriptions – and thus merits further exploration – AP use was found in the study to be safe.
“This study increases the field’s appetite for additional high-quality data on other maternal and offspring outcomes to improve the sophistication of risk-benefit decision making,” the authors wrote.
Nowhere in medicine is there a greater need for personalization of care, particularly when it comes to the treatment of serious mental illness with consideration of “pregnancy physiology and the mother’s capacity to provide sustenance for the growing fetus and newborn,” they wrote.
Dr. Wisner is with Northwestern University, Chicago. She reported that her department at Northwestern received contractual fees for her consultation to Quinn Emanuel Urquhart & Sullivan, LLP, who represented Pfizer Pharmaceutical Company in 2015. A coauthor, Christina Chambers, PhD, reported receiving research funding from AbbVie, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Janssen, Pfizer, Sandoz, and Teva. No other disclosures were reported. Their comments are excerpted from an accompanying editorial (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1538).
Antipsychotic use early in pregnancy does not appear to meaningfully increase the risk of congenital malformations, according to findings from a large Medicaid pregnancy cohort.
A possible exception is with the use of risperidone; a small increase in the risk for malformations seen with the drug should be viewed as “an initial safety signal that will require confirmation in other studies,” Krista F. Huybrechts, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues reported online Aug. 17 in JAMA Psychiatry.
In a nationwide sample of more than 1.3 million pregnant women with a live-born infant between January 1, 2000 and December 31, 2010, the rate of congenital malformations identified within 90 days after delivery was 32.7 per 1,000 births among 1,331,724 women with no antipsychotic (AP) exposure, compared with a rate of 44.5 per 1,000 births among 9,258 women who filled at least one prescription for an atypical AP during their first trimester, and 38.2 per 1,000 in 733 women who filled at least one prescription for a typical AP during their first trimester, the researchers found (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1520).
On unadjusted analyses, an overall risk was seen with atypical AP exposure (relative risk, 1.36), but not with typical AP exposure (RR, 1.17). After adjusting for confounding variables, the risk was not statistically significant (RR, 1.05 and 0.90, respectively). The findings were similar for cardiac malformations, the researchers noted.
When agents were analyzed individually, a small increased risk in overall malformations and cardiac malformations was found with risperidone (RR, 1.26 for each).
“The findings for risperidone should be viewed as an initial safety signal that will require confirmation in other studies,” the researchers wrote.
The study was supported by grants from the National Institutes of Health and the Swiss National Science Foundation. Dr. Huybrechts reported having no financial disclosures. Other authors disclosed receiving consulting fees and/or grant support from AstraZeneca, UCB, Alkermes, Bristol-Myers Squibb/Otsuka, Sunovion, Bayer, Ortho-McNeil Janssen, Pfizer, Forest Laboratories, Cephalon, GlaxoSmithKline, Takeda/Lundbeck, the National Institutes of Health, JDS Therapeutics, Noven Pharmaceuticals, and PamLab.
Antipsychotic use early in pregnancy does not appear to meaningfully increase the risk of congenital malformations, according to findings from a large Medicaid pregnancy cohort.
A possible exception is with the use of risperidone; a small increase in the risk for malformations seen with the drug should be viewed as “an initial safety signal that will require confirmation in other studies,” Krista F. Huybrechts, PhD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues reported online Aug. 17 in JAMA Psychiatry.
In a nationwide sample of more than 1.3 million pregnant women with a live-born infant between January 1, 2000 and December 31, 2010, the rate of congenital malformations identified within 90 days after delivery was 32.7 per 1,000 births among 1,331,724 women with no antipsychotic (AP) exposure, compared with a rate of 44.5 per 1,000 births among 9,258 women who filled at least one prescription for an atypical AP during their first trimester, and 38.2 per 1,000 in 733 women who filled at least one prescription for a typical AP during their first trimester, the researchers found (JAMA Psychiatry. 2016 Aug 17. doi: 10.1001/jamapsychiatry.2016.1520).
On unadjusted analyses, an overall risk was seen with atypical AP exposure (relative risk, 1.36), but not with typical AP exposure (RR, 1.17). After adjusting for confounding variables, the risk was not statistically significant (RR, 1.05 and 0.90, respectively). The findings were similar for cardiac malformations, the researchers noted.
When agents were analyzed individually, a small increased risk in overall malformations and cardiac malformations was found with risperidone (RR, 1.26 for each).
“The findings for risperidone should be viewed as an initial safety signal that will require confirmation in other studies,” the researchers wrote.
The study was supported by grants from the National Institutes of Health and the Swiss National Science Foundation. Dr. Huybrechts reported having no financial disclosures. Other authors disclosed receiving consulting fees and/or grant support from AstraZeneca, UCB, Alkermes, Bristol-Myers Squibb/Otsuka, Sunovion, Bayer, Ortho-McNeil Janssen, Pfizer, Forest Laboratories, Cephalon, GlaxoSmithKline, Takeda/Lundbeck, the National Institutes of Health, JDS Therapeutics, Noven Pharmaceuticals, and PamLab.
FROM JAMA PSYCHIATRY
Key clinical point: Antipsychotic use in the first trimester does not appear to meaningfully increase the risk of congenital malformations.
Major finding: The adjusted relative risk for malformations is 1.05 and 0.90 with atypical and typical antipsychotic drug exposure, respectively.
Data source: A total of 1,341,715 pregnancies from the Medicaid Analytic Extract database pregnancy cohort.
Disclosures: The study was supported by grants from the National Institutes of Health and the Swiss National Science Foundation. Dr. Huybrechts reported having no financial disclosures. Other authors disclosed receiving consulting fees and/or grant support from AstraZeneca, UCB, Alkermes, Bristol-Myers Squibb/Otsuka, Sunovion, Bayer, Ortho-McNeil Janssen, Pfizer, Forest Laboratories, Cephalon, GlaxoSmithKline, Takeda/Lundbeck, the National Institutes of Health, JDS Therapeutics, Noven Pharmaceuticals, and PamLab.
Distinguish between autoinflammatory, autoimmune disorders in children
Boston – Autoinflammatory and autoimmune disorders in children have many overlapping features, but the former represents a malfunction of the innate immune system, and the latter a malfunction of the adaptive immune system. Fevers, skin eruptions, joint pain and swelling, fatigue, and growth failure can occur in both, but it is important to identify the underlying problem, according to Raegan Hunt, MD.
Autoinflammatory disorders can be difficult to detect, so it is important to maintain a high index of suspicion for them, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital, Houston, said at the American Academy of Dermatology annual summer meeting.
She discussed five signs of possible autoinflammatory disease in children, as described in a recent report by Hal M. Hoffman, MD, and Lori Broderick, MD, of Children’s Hospital of San Diego, La Jolla, Calif. (J Allergy Clin Immunol. July 2016; 138:3-14)*:
1. More than three episodes of fever over 101 degrees Farenheit with no explained infectious etiology.
2. Having predictable patterns of characteristic course of each episode.
3. Having specific symptoms during episodes, including nonpruritic skin eruptions, joint or bone pain, severe abdominal pain, and conjunctivitis, with no upper respiratory infection symptoms.
4. Episodes may be triggered by specific stimuli, such as cold exposure or vaccines.
5. A family history of autoinflammatory disease or amyloidosis.
The authors “propose that two or more of these might suggest that an autoinflammatory disorder is possible and should be investigated,” Dr. Hunt said.
Dr. Hunt reported having no relevant disclosures.
sworcester@frontlinemedcom.com
*Correction, 8/16/16: An earlier version of this article failed to note that Dr. Lori Broderick was a co-investigator in the cited report.
Boston – Autoinflammatory and autoimmune disorders in children have many overlapping features, but the former represents a malfunction of the innate immune system, and the latter a malfunction of the adaptive immune system. Fevers, skin eruptions, joint pain and swelling, fatigue, and growth failure can occur in both, but it is important to identify the underlying problem, according to Raegan Hunt, MD.
Autoinflammatory disorders can be difficult to detect, so it is important to maintain a high index of suspicion for them, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital, Houston, said at the American Academy of Dermatology annual summer meeting.
She discussed five signs of possible autoinflammatory disease in children, as described in a recent report by Hal M. Hoffman, MD, and Lori Broderick, MD, of Children’s Hospital of San Diego, La Jolla, Calif. (J Allergy Clin Immunol. July 2016; 138:3-14)*:
1. More than three episodes of fever over 101 degrees Farenheit with no explained infectious etiology.
2. Having predictable patterns of characteristic course of each episode.
3. Having specific symptoms during episodes, including nonpruritic skin eruptions, joint or bone pain, severe abdominal pain, and conjunctivitis, with no upper respiratory infection symptoms.
4. Episodes may be triggered by specific stimuli, such as cold exposure or vaccines.
5. A family history of autoinflammatory disease or amyloidosis.
The authors “propose that two or more of these might suggest that an autoinflammatory disorder is possible and should be investigated,” Dr. Hunt said.
Dr. Hunt reported having no relevant disclosures.
sworcester@frontlinemedcom.com
*Correction, 8/16/16: An earlier version of this article failed to note that Dr. Lori Broderick was a co-investigator in the cited report.
Boston – Autoinflammatory and autoimmune disorders in children have many overlapping features, but the former represents a malfunction of the innate immune system, and the latter a malfunction of the adaptive immune system. Fevers, skin eruptions, joint pain and swelling, fatigue, and growth failure can occur in both, but it is important to identify the underlying problem, according to Raegan Hunt, MD.
Autoinflammatory disorders can be difficult to detect, so it is important to maintain a high index of suspicion for them, Dr. Hunt of Baylor College of Medicine and Texas Children’s Hospital, Houston, said at the American Academy of Dermatology annual summer meeting.
She discussed five signs of possible autoinflammatory disease in children, as described in a recent report by Hal M. Hoffman, MD, and Lori Broderick, MD, of Children’s Hospital of San Diego, La Jolla, Calif. (J Allergy Clin Immunol. July 2016; 138:3-14)*:
1. More than three episodes of fever over 101 degrees Farenheit with no explained infectious etiology.
2. Having predictable patterns of characteristic course of each episode.
3. Having specific symptoms during episodes, including nonpruritic skin eruptions, joint or bone pain, severe abdominal pain, and conjunctivitis, with no upper respiratory infection symptoms.
4. Episodes may be triggered by specific stimuli, such as cold exposure or vaccines.
5. A family history of autoinflammatory disease or amyloidosis.
The authors “propose that two or more of these might suggest that an autoinflammatory disorder is possible and should be investigated,” Dr. Hunt said.
Dr. Hunt reported having no relevant disclosures.
sworcester@frontlinemedcom.com
*Correction, 8/16/16: An earlier version of this article failed to note that Dr. Lori Broderick was a co-investigator in the cited report.
Expert Analysis from the AAD Summer Academy 2016
Sunscreens safe in babies, children
BOSTON – Despite what some popular online media outlets report, sunscreens are safe in children and can even be used on infants under 6 months of age when sun avoidance – the best approach to protecting babies from the damaging effects of the sun – is not possible, according to Mercedes E. Gonzalez, MD.
A quick Google search reveals numerous, widely-shared articles about the dangers lurking in one’s beach bag, and while many product labels recommend asking a doctor about whether the product is safe for babies under age 6 months, that’s only because most product safety studies didn’t include that age group, Dr. Gonzalez of the University of Miami said at the American Academy of Dermatology summer meeting.
In fact, there is “nothing magical that happens” in infant skin after 6 months that makes sunscreen use safer, she said, explaining that infant skin is structurally and functionally different from adult skin, and that while gradual maturation takes place over time, thereby reducing susceptibility to percutaneous absorption of topically applied products, the risk is minimal even in babies younger than age 6 months.
The skin characteristics that make younger skin more susceptible to percutaneous absorption also make babies and children unusually susceptible to ultraviolet radiation and ultraviolet radiation–induced immunosuppression, for which the consequences are not fully understood, she said.
Among the more commonly cited sunscreen ingredients of concern are oxybenzone, or benzonephenone-3, and nanoparticles, she noted.
However, the overall consensus based on studies of oxybenzone is that aside from causing some cases of allergic and irritant contact dermatitis, the compound is safe; no harmful cause and effect relationship with oxybenzone and systemic side effects in humans have been reported, and periodic reviews by European, Australian and U.S. safety panels all conclude that it is safe.
Numerous studies of nanoparticles – such as nanosized zinc oxide and titanium dioxide – have shown that absorption is confined to the level of stratum corneum – even when skin barrier function has been altered, she said, noting that most are coated with aluminum oxide and SiO2 to minimize contact.
However, the safety of sunscreen shouldn’t be seen as license to ignore sun-exposure recommendations; sunscreen in infants should be considered “the last layer of protection,” used only on exposed areas when adequate clothing and shade are not available, according to a 2011 American Academy of Pediatrics statement (Pediatrics. 2011 Feb. doi: 10.1542/peds.2010-3501).
Efforts should be made to keep babies in the shade when outdoors whenever possible, especially during peak sun hours. Use sun-protective clothing, including hats, sunglasses, and long-sleeved shirts, Dr. Gonzalez advised.
When sunscreen is required, a broad-spectrum water-resistant product with an SPF of more than 30 is preferable.
“But the best sunscreen is the one you and your child will use,” she said.
Mineral-based products are less irritating and thus may be a preferred option for children with atopic dermatitis, she added.
Advise parents to apply sunscreen to all areas not protected by their child’s clothing, paying particular attention to vulnerable areas, including the back of the neck, ears, and dorsal feet. Reapply before going outdoors, and then again every 2 hours, she advised.
“So the overall answer to the parents’ question, ‘Are sunscreens safe?’ ... the overwhelming answer here is yes, and the weight of the evidence shows there is no proven harm from sunscreen use especially when used properly,” she said.
Provide specific guidance for pediatric sunscreen use
In the face of conflicting information about sunscreen safety and efficacy, parents with questions about sunscreen are looking for specific direction, Dr. Gonzalez said.
She said she finds it helpful to teach them about the importance of reading labels. That is, looking at the ingredients, and looking for SPF above 30, broad-spectrum coverage, and water resistance. She also recommends providing a list or images of good options, and circling the specific preferred products.
For babies, she finds stick sunscreens most useful for application.
“I generally don’t recommend sprays, but if they’re going to use a spray – and parents love sprays because they are easy to apply – I recommend the ones that have some zinc oxide in them, so that when they apply them they can see where they’re going on the skin,” Dr. Gonzalez said.
Tell patients to apply sunscreen before leaving the house, she advised, adding that making sunscreen application part of a daily routine helps encourage healthy behaviors, as does allowing children, at the right age, to participate in sunscreen application.
For adolescents, avoid scare tactics such as warning about skin cancer. Rather, focus on benefits of avoiding the sun, help them find a product they like by finding out why they don’t like a particular product and recommending an alternative, then following up on that when they come back in, she suggested.
“I really try to address it at every visit,” Dr. Gonzalez said.
“Finally, the most important message is that sunscreen is really just one part of complete sun protection,” she said, noting that specific information about where to buy sun-protective clothing and hats is also important.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Despite what some popular online media outlets report, sunscreens are safe in children and can even be used on infants under 6 months of age when sun avoidance – the best approach to protecting babies from the damaging effects of the sun – is not possible, according to Mercedes E. Gonzalez, MD.
A quick Google search reveals numerous, widely-shared articles about the dangers lurking in one’s beach bag, and while many product labels recommend asking a doctor about whether the product is safe for babies under age 6 months, that’s only because most product safety studies didn’t include that age group, Dr. Gonzalez of the University of Miami said at the American Academy of Dermatology summer meeting.
In fact, there is “nothing magical that happens” in infant skin after 6 months that makes sunscreen use safer, she said, explaining that infant skin is structurally and functionally different from adult skin, and that while gradual maturation takes place over time, thereby reducing susceptibility to percutaneous absorption of topically applied products, the risk is minimal even in babies younger than age 6 months.
The skin characteristics that make younger skin more susceptible to percutaneous absorption also make babies and children unusually susceptible to ultraviolet radiation and ultraviolet radiation–induced immunosuppression, for which the consequences are not fully understood, she said.
Among the more commonly cited sunscreen ingredients of concern are oxybenzone, or benzonephenone-3, and nanoparticles, she noted.
However, the overall consensus based on studies of oxybenzone is that aside from causing some cases of allergic and irritant contact dermatitis, the compound is safe; no harmful cause and effect relationship with oxybenzone and systemic side effects in humans have been reported, and periodic reviews by European, Australian and U.S. safety panels all conclude that it is safe.
Numerous studies of nanoparticles – such as nanosized zinc oxide and titanium dioxide – have shown that absorption is confined to the level of stratum corneum – even when skin barrier function has been altered, she said, noting that most are coated with aluminum oxide and SiO2 to minimize contact.
However, the safety of sunscreen shouldn’t be seen as license to ignore sun-exposure recommendations; sunscreen in infants should be considered “the last layer of protection,” used only on exposed areas when adequate clothing and shade are not available, according to a 2011 American Academy of Pediatrics statement (Pediatrics. 2011 Feb. doi: 10.1542/peds.2010-3501).
Efforts should be made to keep babies in the shade when outdoors whenever possible, especially during peak sun hours. Use sun-protective clothing, including hats, sunglasses, and long-sleeved shirts, Dr. Gonzalez advised.
When sunscreen is required, a broad-spectrum water-resistant product with an SPF of more than 30 is preferable.
“But the best sunscreen is the one you and your child will use,” she said.
Mineral-based products are less irritating and thus may be a preferred option for children with atopic dermatitis, she added.
Advise parents to apply sunscreen to all areas not protected by their child’s clothing, paying particular attention to vulnerable areas, including the back of the neck, ears, and dorsal feet. Reapply before going outdoors, and then again every 2 hours, she advised.
“So the overall answer to the parents’ question, ‘Are sunscreens safe?’ ... the overwhelming answer here is yes, and the weight of the evidence shows there is no proven harm from sunscreen use especially when used properly,” she said.
Provide specific guidance for pediatric sunscreen use
In the face of conflicting information about sunscreen safety and efficacy, parents with questions about sunscreen are looking for specific direction, Dr. Gonzalez said.
She said she finds it helpful to teach them about the importance of reading labels. That is, looking at the ingredients, and looking for SPF above 30, broad-spectrum coverage, and water resistance. She also recommends providing a list or images of good options, and circling the specific preferred products.
For babies, she finds stick sunscreens most useful for application.
“I generally don’t recommend sprays, but if they’re going to use a spray – and parents love sprays because they are easy to apply – I recommend the ones that have some zinc oxide in them, so that when they apply them they can see where they’re going on the skin,” Dr. Gonzalez said.
Tell patients to apply sunscreen before leaving the house, she advised, adding that making sunscreen application part of a daily routine helps encourage healthy behaviors, as does allowing children, at the right age, to participate in sunscreen application.
For adolescents, avoid scare tactics such as warning about skin cancer. Rather, focus on benefits of avoiding the sun, help them find a product they like by finding out why they don’t like a particular product and recommending an alternative, then following up on that when they come back in, she suggested.
“I really try to address it at every visit,” Dr. Gonzalez said.
“Finally, the most important message is that sunscreen is really just one part of complete sun protection,” she said, noting that specific information about where to buy sun-protective clothing and hats is also important.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Despite what some popular online media outlets report, sunscreens are safe in children and can even be used on infants under 6 months of age when sun avoidance – the best approach to protecting babies from the damaging effects of the sun – is not possible, according to Mercedes E. Gonzalez, MD.
A quick Google search reveals numerous, widely-shared articles about the dangers lurking in one’s beach bag, and while many product labels recommend asking a doctor about whether the product is safe for babies under age 6 months, that’s only because most product safety studies didn’t include that age group, Dr. Gonzalez of the University of Miami said at the American Academy of Dermatology summer meeting.
In fact, there is “nothing magical that happens” in infant skin after 6 months that makes sunscreen use safer, she said, explaining that infant skin is structurally and functionally different from adult skin, and that while gradual maturation takes place over time, thereby reducing susceptibility to percutaneous absorption of topically applied products, the risk is minimal even in babies younger than age 6 months.
The skin characteristics that make younger skin more susceptible to percutaneous absorption also make babies and children unusually susceptible to ultraviolet radiation and ultraviolet radiation–induced immunosuppression, for which the consequences are not fully understood, she said.
Among the more commonly cited sunscreen ingredients of concern are oxybenzone, or benzonephenone-3, and nanoparticles, she noted.
However, the overall consensus based on studies of oxybenzone is that aside from causing some cases of allergic and irritant contact dermatitis, the compound is safe; no harmful cause and effect relationship with oxybenzone and systemic side effects in humans have been reported, and periodic reviews by European, Australian and U.S. safety panels all conclude that it is safe.
Numerous studies of nanoparticles – such as nanosized zinc oxide and titanium dioxide – have shown that absorption is confined to the level of stratum corneum – even when skin barrier function has been altered, she said, noting that most are coated with aluminum oxide and SiO2 to minimize contact.
However, the safety of sunscreen shouldn’t be seen as license to ignore sun-exposure recommendations; sunscreen in infants should be considered “the last layer of protection,” used only on exposed areas when adequate clothing and shade are not available, according to a 2011 American Academy of Pediatrics statement (Pediatrics. 2011 Feb. doi: 10.1542/peds.2010-3501).
Efforts should be made to keep babies in the shade when outdoors whenever possible, especially during peak sun hours. Use sun-protective clothing, including hats, sunglasses, and long-sleeved shirts, Dr. Gonzalez advised.
When sunscreen is required, a broad-spectrum water-resistant product with an SPF of more than 30 is preferable.
“But the best sunscreen is the one you and your child will use,” she said.
Mineral-based products are less irritating and thus may be a preferred option for children with atopic dermatitis, she added.
Advise parents to apply sunscreen to all areas not protected by their child’s clothing, paying particular attention to vulnerable areas, including the back of the neck, ears, and dorsal feet. Reapply before going outdoors, and then again every 2 hours, she advised.
“So the overall answer to the parents’ question, ‘Are sunscreens safe?’ ... the overwhelming answer here is yes, and the weight of the evidence shows there is no proven harm from sunscreen use especially when used properly,” she said.
Provide specific guidance for pediatric sunscreen use
In the face of conflicting information about sunscreen safety and efficacy, parents with questions about sunscreen are looking for specific direction, Dr. Gonzalez said.
She said she finds it helpful to teach them about the importance of reading labels. That is, looking at the ingredients, and looking for SPF above 30, broad-spectrum coverage, and water resistance. She also recommends providing a list or images of good options, and circling the specific preferred products.
For babies, she finds stick sunscreens most useful for application.
“I generally don’t recommend sprays, but if they’re going to use a spray – and parents love sprays because they are easy to apply – I recommend the ones that have some zinc oxide in them, so that when they apply them they can see where they’re going on the skin,” Dr. Gonzalez said.
Tell patients to apply sunscreen before leaving the house, she advised, adding that making sunscreen application part of a daily routine helps encourage healthy behaviors, as does allowing children, at the right age, to participate in sunscreen application.
For adolescents, avoid scare tactics such as warning about skin cancer. Rather, focus on benefits of avoiding the sun, help them find a product they like by finding out why they don’t like a particular product and recommending an alternative, then following up on that when they come back in, she suggested.
“I really try to address it at every visit,” Dr. Gonzalez said.
“Finally, the most important message is that sunscreen is really just one part of complete sun protection,” she said, noting that specific information about where to buy sun-protective clothing and hats is also important.
Dr. Gonzalez reported serving as a speaker and/or advisory board member and receiving honoraria from Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
COMFORT mnemonic can help guide malignant wound care
BOSTON – Malignancy in a chronic leg wound can be very aggressive, and multidisciplinary care is a must – particularly in those who oppose amputation, according to Tania J. Phillips, MD.
“When you get a more advanced, malignant, ulcerated wound, this is really a complex situation that requires multidisciplinary care,” she said at the American Academy of Dermatology summer meeting.
In addition to dermatologic care, patients may need medical oncology, surgery, radiation, and good nursing care, she explained.
For patients with malignancy who oppose amputation, a palliative care team may also be a necessity, said Dr. Phillips, professor of dermatology at Boston University. She described one patient she cared for who had an ulcerated wound with squamous cell carcinoma. The patient was treated daily 5 days per week for 2 months. The wound did not heal.
“In these kinds of patients you may not heal the wound; you may just have to try to keep the wound clean, free of pain, and free of infection, she said.
COMFORT is a valuable mnemonic device for caring for such patients, she said: Care for pain and itching, use Odor control, Manage exudate and bleeding, Fight infection, Optimize peri-wound skin integrity, use Reparative and aesthetic wound dressings, Treat the cancer.
Pain is a major issue in patients with such wounds. It is important assess the wound etiology and treat the underlying disease, but it is also important to assess the pain and try to manage it, Dr. Phillips said. Often, addressing local wound management will help with the pain. Appropriate dressing selection is particularly important, as dressing changes can be very painful, she said, recommending the use of non-stick dressings that keep the wound moist. Local and systemic treatment may also be necessary to control pain and itching, she said.
Odor control is another concern. Necrotic tissue has a lot of odor, so debridement can help.
“Use a 19-guage needle to cleanse the wound,” she suggested, noting that this provides higher pressure that can be effective for cleansing.
Dressings that contain nanocrystalline silver or activated charcoal can also help.
As for managing exudate and bleeding, dressing choice is again an important consideration. More absorbent dressings like alginates are useful and have hemostatic properties that can also help with bleeding. Frequent dressing changes are also important.
“And obviously, you want to treat infection,” Dr. Phillips added. Metronidazole gel or powder can help with both infection and odor, she noted.
Peri-wound skin can be very fragile, so it is important to provide protection. In some cases, this can be addressed simply with zinc oxide or petrolatum. Various skin sealants are also available, and can be used to form a barrier to dressings that can be damaging to the skin.
Silicone dressings are very powerful because they don’t stick to the skin, and are not painful or damaging when removed, she said.
The use of a window with hydrocolloid dressing, which allows for changing only the inside of the dressing, is another useful approach, and using mesh rather than tape to hold the dressing in place can help to protect peri-wound skin.
Dr. Phillips reported a financial relationship with Hygeia.
BOSTON – Malignancy in a chronic leg wound can be very aggressive, and multidisciplinary care is a must – particularly in those who oppose amputation, according to Tania J. Phillips, MD.
“When you get a more advanced, malignant, ulcerated wound, this is really a complex situation that requires multidisciplinary care,” she said at the American Academy of Dermatology summer meeting.
In addition to dermatologic care, patients may need medical oncology, surgery, radiation, and good nursing care, she explained.
For patients with malignancy who oppose amputation, a palliative care team may also be a necessity, said Dr. Phillips, professor of dermatology at Boston University. She described one patient she cared for who had an ulcerated wound with squamous cell carcinoma. The patient was treated daily 5 days per week for 2 months. The wound did not heal.
“In these kinds of patients you may not heal the wound; you may just have to try to keep the wound clean, free of pain, and free of infection, she said.
COMFORT is a valuable mnemonic device for caring for such patients, she said: Care for pain and itching, use Odor control, Manage exudate and bleeding, Fight infection, Optimize peri-wound skin integrity, use Reparative and aesthetic wound dressings, Treat the cancer.
Pain is a major issue in patients with such wounds. It is important assess the wound etiology and treat the underlying disease, but it is also important to assess the pain and try to manage it, Dr. Phillips said. Often, addressing local wound management will help with the pain. Appropriate dressing selection is particularly important, as dressing changes can be very painful, she said, recommending the use of non-stick dressings that keep the wound moist. Local and systemic treatment may also be necessary to control pain and itching, she said.
Odor control is another concern. Necrotic tissue has a lot of odor, so debridement can help.
“Use a 19-guage needle to cleanse the wound,” she suggested, noting that this provides higher pressure that can be effective for cleansing.
Dressings that contain nanocrystalline silver or activated charcoal can also help.
As for managing exudate and bleeding, dressing choice is again an important consideration. More absorbent dressings like alginates are useful and have hemostatic properties that can also help with bleeding. Frequent dressing changes are also important.
“And obviously, you want to treat infection,” Dr. Phillips added. Metronidazole gel or powder can help with both infection and odor, she noted.
Peri-wound skin can be very fragile, so it is important to provide protection. In some cases, this can be addressed simply with zinc oxide or petrolatum. Various skin sealants are also available, and can be used to form a barrier to dressings that can be damaging to the skin.
Silicone dressings are very powerful because they don’t stick to the skin, and are not painful or damaging when removed, she said.
The use of a window with hydrocolloid dressing, which allows for changing only the inside of the dressing, is another useful approach, and using mesh rather than tape to hold the dressing in place can help to protect peri-wound skin.
Dr. Phillips reported a financial relationship with Hygeia.
BOSTON – Malignancy in a chronic leg wound can be very aggressive, and multidisciplinary care is a must – particularly in those who oppose amputation, according to Tania J. Phillips, MD.
“When you get a more advanced, malignant, ulcerated wound, this is really a complex situation that requires multidisciplinary care,” she said at the American Academy of Dermatology summer meeting.
In addition to dermatologic care, patients may need medical oncology, surgery, radiation, and good nursing care, she explained.
For patients with malignancy who oppose amputation, a palliative care team may also be a necessity, said Dr. Phillips, professor of dermatology at Boston University. She described one patient she cared for who had an ulcerated wound with squamous cell carcinoma. The patient was treated daily 5 days per week for 2 months. The wound did not heal.
“In these kinds of patients you may not heal the wound; you may just have to try to keep the wound clean, free of pain, and free of infection, she said.
COMFORT is a valuable mnemonic device for caring for such patients, she said: Care for pain and itching, use Odor control, Manage exudate and bleeding, Fight infection, Optimize peri-wound skin integrity, use Reparative and aesthetic wound dressings, Treat the cancer.
Pain is a major issue in patients with such wounds. It is important assess the wound etiology and treat the underlying disease, but it is also important to assess the pain and try to manage it, Dr. Phillips said. Often, addressing local wound management will help with the pain. Appropriate dressing selection is particularly important, as dressing changes can be very painful, she said, recommending the use of non-stick dressings that keep the wound moist. Local and systemic treatment may also be necessary to control pain and itching, she said.
Odor control is another concern. Necrotic tissue has a lot of odor, so debridement can help.
“Use a 19-guage needle to cleanse the wound,” she suggested, noting that this provides higher pressure that can be effective for cleansing.
Dressings that contain nanocrystalline silver or activated charcoal can also help.
As for managing exudate and bleeding, dressing choice is again an important consideration. More absorbent dressings like alginates are useful and have hemostatic properties that can also help with bleeding. Frequent dressing changes are also important.
“And obviously, you want to treat infection,” Dr. Phillips added. Metronidazole gel or powder can help with both infection and odor, she noted.
Peri-wound skin can be very fragile, so it is important to provide protection. In some cases, this can be addressed simply with zinc oxide or petrolatum. Various skin sealants are also available, and can be used to form a barrier to dressings that can be damaging to the skin.
Silicone dressings are very powerful because they don’t stick to the skin, and are not painful or damaging when removed, she said.
The use of a window with hydrocolloid dressing, which allows for changing only the inside of the dressing, is another useful approach, and using mesh rather than tape to hold the dressing in place can help to protect peri-wound skin.
Dr. Phillips reported a financial relationship with Hygeia.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016