User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
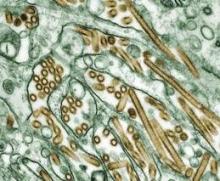
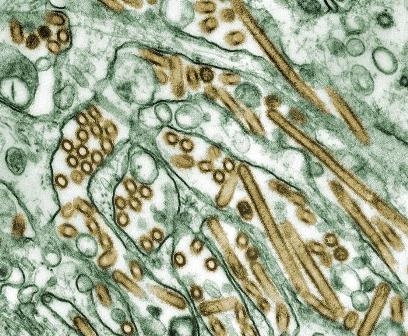
H5N1 flu researchers lift self-imposed moratorium
The 40 researchers who pledged a voluntary moratorium in January 2012 on studies of H5N1 avian influenza and its ability to become airborne transmissible between mammals called their moratorium off a year later, on Jan. 23.
In a letter published simultaneously in the journals Science and Nature, the international panel of 40 influenza experts said "acknowledging that the aims of the voluntary moratorium have been met in some countries and are close to being met in others, we declare an end to the voluntary moratorium on avian flu transmission studies."
But the published letter (Science 2013 [doi: 10.1126/science.1235140]; Nature 2013 [doi: 10.1038/nature11858]) also stressed that the only endorsed settings for resumed H5N1 mammalian studies are countries with established rules for conducting this research, a stipulation that for the time being shuts down this work for U.S.-based investigators.
"Scientists should not restart their work in countries where, as yet, no decision has been reached on the conditions for H5N1 virus transmission research," the letter said, a category that most notably includes the United States. Countries with rules in place and where this research can restart immediately include European Union members, Canada, China, and very soon Japan, said Ron A.M. Fouchier, Ph.D., during a press conference about the moratorium lifting.
That made Dr. Fouchier the sole researcher working on airborne transmission of H5N1 flu in mammals who could announce imminent resumption of his work, something that he said would restart in his Rotterdam lab "in the next few weeks," as soon as he receives "a new order of ferrets," he said at the news conference organized by the publishers of Science and Nature.
A second researcher who has also published results on airborne-transmissible H5N1 flu in ferrets, Yoshihiro Kawaoka, Ph.D., a professor of virology at the University of Wisconsin, said work at his lab remains down until U.S. agencies finish setting conditions for H5N1 research.
"My research on H5N1 transmission is funded by the [U.S. National Institutes of Health], so I will not be able to resume until the U.S. government decides the details of its guidelines," Dr. Kawaoka said at the press conference. The U.S. Department of Health and Human Services released a draft of its new H5N1 rules last October, and while they appear to be nearing completion, U.S. agencies have given no time frame for when the finalized guidelines will be in place, said Dr. Fouchier, a professor of molecular virology at Erasmus Medical Center in Rotterdam.
Dr. Fouchier said that topping his research agenda is further assessment of the several mutations that he previously linked to ferret airborne transmission to try to identify the minimum set of mutations needed for airborne transmission. His second immediate goal is to see whether these mutations are critical for transmission of H5N1 strains isolated from a wider number of world sites, such as from Egypt and China; the strains that he has characterized so far came from Indonesia and Vietnam. A third project will be to assess the prevalence of H5N1 in the wild that carries one or more transmissibility mutations.
The central messages delivered at the press conference by Dr. Fouchier, Dr. Kawaoka, and a third signer of the letter, Richard J. Webby, Ph.D., an influenza expert at St. Jude Children’s Research Hospital in Memphis, were that ongoing research into H5N1 transmission is critical and must now continue, the research can be done safely, and the 1-year hiatus produced by the moratorium (Nature 2012;481:443) accomplished its goals.
"The moratorium was put in place to provide time for discussion about the risks and benefits of this research. That has been achieved. We feel that the need for this moratorium is no longer in place," Dr. Webby said.
"We want to resume research because it is important for pandemic preparedness," said Dr. Kawaoka. "The benefits outweigh the risks. The greater risk is not doing this research. The risk [from air-transmissible H5N1] already exists in nature. It’s not doing this research that really puts us in danger," he said.
The research will protect public health by identifying the mutations that occur in nature that allow airborne transmission, and will allow researchers to better evaluate antiviral drugs and vaccines for H5N1, Dr. Fouchier said.
During the yearlong moratorium, Science and Nature published papers detailing the initial transmission experiments done by Dr. Fouchier’s group (Science 2012;336:1534-41; Science 2012;336:1541-7), and the independent work done in Dr. Kawaoka’s laboratory (Nature 2012;486:420-8). At the time of the moratorium a year ago, some of the controversy over this work centered on whether these papers should be freely available as part of the scientific literature, but regulatory groups subsequently signed off on allowing publication of these reports.
Assessing risk and benefit "is not an exact science," Dr. Fouchier admitted at the press conference, but he stressed that "all 40 people who signed the moratorium letter in 2012 have now signed the letter to restart the work. All 40 feel that the benefits outweigh the risks." The experts clearly spelled this out in the letter’s final line: "Because the risk exists in nature that an H5N1 virus capable of transmission in mammals may emerge, the benefits of this work outweigh the risks," the letter concludes.
An editorial that ran in Nature (2013;493:451-2) on the new letter and the end of the research moratorium said that the risk and benefit issue had not yet been fully resolved, primarily because of the limited scope of the debate over the past year. "There remains a perception ... that the debate has taken place largely behind closed doors, and has been dominated by the scientists and research funders who have vested interests in the outcome," said the editorial. "An irreproachable, independent risk-benefit analysis of such research, perhaps convened by a body such as the World Health Organization (WHO), is still lacking."
The editorial also applauded the H5N1 research guidelines that the U.S. government is readying for approval. "The proposed framework for assessing H5N1 gain-of-function research, outlined by the U.S. National Institutes of Health at an international meeting in Bethesda, Maryland, in December, ... should serve as an important checklist. The criteria include sensible questions."
Dr. Fouchier, Dr. Kawaoka, and Dr. Webby had no disclosures.
On Twitter @mitchelzoler
The 40 researchers who pledged a voluntary moratorium in January 2012 on studies of H5N1 avian influenza and its ability to become airborne transmissible between mammals called their moratorium off a year later, on Jan. 23.
In a letter published simultaneously in the journals Science and Nature, the international panel of 40 influenza experts said "acknowledging that the aims of the voluntary moratorium have been met in some countries and are close to being met in others, we declare an end to the voluntary moratorium on avian flu transmission studies."
But the published letter (Science 2013 [doi: 10.1126/science.1235140]; Nature 2013 [doi: 10.1038/nature11858]) also stressed that the only endorsed settings for resumed H5N1 mammalian studies are countries with established rules for conducting this research, a stipulation that for the time being shuts down this work for U.S.-based investigators.
"Scientists should not restart their work in countries where, as yet, no decision has been reached on the conditions for H5N1 virus transmission research," the letter said, a category that most notably includes the United States. Countries with rules in place and where this research can restart immediately include European Union members, Canada, China, and very soon Japan, said Ron A.M. Fouchier, Ph.D., during a press conference about the moratorium lifting.
That made Dr. Fouchier the sole researcher working on airborne transmission of H5N1 flu in mammals who could announce imminent resumption of his work, something that he said would restart in his Rotterdam lab "in the next few weeks," as soon as he receives "a new order of ferrets," he said at the news conference organized by the publishers of Science and Nature.
A second researcher who has also published results on airborne-transmissible H5N1 flu in ferrets, Yoshihiro Kawaoka, Ph.D., a professor of virology at the University of Wisconsin, said work at his lab remains down until U.S. agencies finish setting conditions for H5N1 research.
"My research on H5N1 transmission is funded by the [U.S. National Institutes of Health], so I will not be able to resume until the U.S. government decides the details of its guidelines," Dr. Kawaoka said at the press conference. The U.S. Department of Health and Human Services released a draft of its new H5N1 rules last October, and while they appear to be nearing completion, U.S. agencies have given no time frame for when the finalized guidelines will be in place, said Dr. Fouchier, a professor of molecular virology at Erasmus Medical Center in Rotterdam.
Dr. Fouchier said that topping his research agenda is further assessment of the several mutations that he previously linked to ferret airborne transmission to try to identify the minimum set of mutations needed for airborne transmission. His second immediate goal is to see whether these mutations are critical for transmission of H5N1 strains isolated from a wider number of world sites, such as from Egypt and China; the strains that he has characterized so far came from Indonesia and Vietnam. A third project will be to assess the prevalence of H5N1 in the wild that carries one or more transmissibility mutations.
The central messages delivered at the press conference by Dr. Fouchier, Dr. Kawaoka, and a third signer of the letter, Richard J. Webby, Ph.D., an influenza expert at St. Jude Children’s Research Hospital in Memphis, were that ongoing research into H5N1 transmission is critical and must now continue, the research can be done safely, and the 1-year hiatus produced by the moratorium (Nature 2012;481:443) accomplished its goals.
"The moratorium was put in place to provide time for discussion about the risks and benefits of this research. That has been achieved. We feel that the need for this moratorium is no longer in place," Dr. Webby said.
"We want to resume research because it is important for pandemic preparedness," said Dr. Kawaoka. "The benefits outweigh the risks. The greater risk is not doing this research. The risk [from air-transmissible H5N1] already exists in nature. It’s not doing this research that really puts us in danger," he said.
The research will protect public health by identifying the mutations that occur in nature that allow airborne transmission, and will allow researchers to better evaluate antiviral drugs and vaccines for H5N1, Dr. Fouchier said.
During the yearlong moratorium, Science and Nature published papers detailing the initial transmission experiments done by Dr. Fouchier’s group (Science 2012;336:1534-41; Science 2012;336:1541-7), and the independent work done in Dr. Kawaoka’s laboratory (Nature 2012;486:420-8). At the time of the moratorium a year ago, some of the controversy over this work centered on whether these papers should be freely available as part of the scientific literature, but regulatory groups subsequently signed off on allowing publication of these reports.
Assessing risk and benefit "is not an exact science," Dr. Fouchier admitted at the press conference, but he stressed that "all 40 people who signed the moratorium letter in 2012 have now signed the letter to restart the work. All 40 feel that the benefits outweigh the risks." The experts clearly spelled this out in the letter’s final line: "Because the risk exists in nature that an H5N1 virus capable of transmission in mammals may emerge, the benefits of this work outweigh the risks," the letter concludes.
An editorial that ran in Nature (2013;493:451-2) on the new letter and the end of the research moratorium said that the risk and benefit issue had not yet been fully resolved, primarily because of the limited scope of the debate over the past year. "There remains a perception ... that the debate has taken place largely behind closed doors, and has been dominated by the scientists and research funders who have vested interests in the outcome," said the editorial. "An irreproachable, independent risk-benefit analysis of such research, perhaps convened by a body such as the World Health Organization (WHO), is still lacking."
The editorial also applauded the H5N1 research guidelines that the U.S. government is readying for approval. "The proposed framework for assessing H5N1 gain-of-function research, outlined by the U.S. National Institutes of Health at an international meeting in Bethesda, Maryland, in December, ... should serve as an important checklist. The criteria include sensible questions."
Dr. Fouchier, Dr. Kawaoka, and Dr. Webby had no disclosures.
On Twitter @mitchelzoler
The 40 researchers who pledged a voluntary moratorium in January 2012 on studies of H5N1 avian influenza and its ability to become airborne transmissible between mammals called their moratorium off a year later, on Jan. 23.
In a letter published simultaneously in the journals Science and Nature, the international panel of 40 influenza experts said "acknowledging that the aims of the voluntary moratorium have been met in some countries and are close to being met in others, we declare an end to the voluntary moratorium on avian flu transmission studies."
But the published letter (Science 2013 [doi: 10.1126/science.1235140]; Nature 2013 [doi: 10.1038/nature11858]) also stressed that the only endorsed settings for resumed H5N1 mammalian studies are countries with established rules for conducting this research, a stipulation that for the time being shuts down this work for U.S.-based investigators.
"Scientists should not restart their work in countries where, as yet, no decision has been reached on the conditions for H5N1 virus transmission research," the letter said, a category that most notably includes the United States. Countries with rules in place and where this research can restart immediately include European Union members, Canada, China, and very soon Japan, said Ron A.M. Fouchier, Ph.D., during a press conference about the moratorium lifting.
That made Dr. Fouchier the sole researcher working on airborne transmission of H5N1 flu in mammals who could announce imminent resumption of his work, something that he said would restart in his Rotterdam lab "in the next few weeks," as soon as he receives "a new order of ferrets," he said at the news conference organized by the publishers of Science and Nature.
A second researcher who has also published results on airborne-transmissible H5N1 flu in ferrets, Yoshihiro Kawaoka, Ph.D., a professor of virology at the University of Wisconsin, said work at his lab remains down until U.S. agencies finish setting conditions for H5N1 research.
"My research on H5N1 transmission is funded by the [U.S. National Institutes of Health], so I will not be able to resume until the U.S. government decides the details of its guidelines," Dr. Kawaoka said at the press conference. The U.S. Department of Health and Human Services released a draft of its new H5N1 rules last October, and while they appear to be nearing completion, U.S. agencies have given no time frame for when the finalized guidelines will be in place, said Dr. Fouchier, a professor of molecular virology at Erasmus Medical Center in Rotterdam.
Dr. Fouchier said that topping his research agenda is further assessment of the several mutations that he previously linked to ferret airborne transmission to try to identify the minimum set of mutations needed for airborne transmission. His second immediate goal is to see whether these mutations are critical for transmission of H5N1 strains isolated from a wider number of world sites, such as from Egypt and China; the strains that he has characterized so far came from Indonesia and Vietnam. A third project will be to assess the prevalence of H5N1 in the wild that carries one or more transmissibility mutations.
The central messages delivered at the press conference by Dr. Fouchier, Dr. Kawaoka, and a third signer of the letter, Richard J. Webby, Ph.D., an influenza expert at St. Jude Children’s Research Hospital in Memphis, were that ongoing research into H5N1 transmission is critical and must now continue, the research can be done safely, and the 1-year hiatus produced by the moratorium (Nature 2012;481:443) accomplished its goals.
"The moratorium was put in place to provide time for discussion about the risks and benefits of this research. That has been achieved. We feel that the need for this moratorium is no longer in place," Dr. Webby said.
"We want to resume research because it is important for pandemic preparedness," said Dr. Kawaoka. "The benefits outweigh the risks. The greater risk is not doing this research. The risk [from air-transmissible H5N1] already exists in nature. It’s not doing this research that really puts us in danger," he said.
The research will protect public health by identifying the mutations that occur in nature that allow airborne transmission, and will allow researchers to better evaluate antiviral drugs and vaccines for H5N1, Dr. Fouchier said.
During the yearlong moratorium, Science and Nature published papers detailing the initial transmission experiments done by Dr. Fouchier’s group (Science 2012;336:1534-41; Science 2012;336:1541-7), and the independent work done in Dr. Kawaoka’s laboratory (Nature 2012;486:420-8). At the time of the moratorium a year ago, some of the controversy over this work centered on whether these papers should be freely available as part of the scientific literature, but regulatory groups subsequently signed off on allowing publication of these reports.
Assessing risk and benefit "is not an exact science," Dr. Fouchier admitted at the press conference, but he stressed that "all 40 people who signed the moratorium letter in 2012 have now signed the letter to restart the work. All 40 feel that the benefits outweigh the risks." The experts clearly spelled this out in the letter’s final line: "Because the risk exists in nature that an H5N1 virus capable of transmission in mammals may emerge, the benefits of this work outweigh the risks," the letter concludes.
An editorial that ran in Nature (2013;493:451-2) on the new letter and the end of the research moratorium said that the risk and benefit issue had not yet been fully resolved, primarily because of the limited scope of the debate over the past year. "There remains a perception ... that the debate has taken place largely behind closed doors, and has been dominated by the scientists and research funders who have vested interests in the outcome," said the editorial. "An irreproachable, independent risk-benefit analysis of such research, perhaps convened by a body such as the World Health Organization (WHO), is still lacking."
The editorial also applauded the H5N1 research guidelines that the U.S. government is readying for approval. "The proposed framework for assessing H5N1 gain-of-function research, outlined by the U.S. National Institutes of Health at an international meeting in Bethesda, Maryland, in December, ... should serve as an important checklist. The criteria include sensible questions."
Dr. Fouchier, Dr. Kawaoka, and Dr. Webby had no disclosures.
On Twitter @mitchelzoler
FROM SCIENCE AND NATURE
Locoregional anesthesia boosts amputation success
PALM BEACH, FLA. – Locoregional anesthesia boosts the success rate of lower-extremity amputations, while time-saving shortcuts and relying too heavily on surgical residents to perform the surgery raise the risk that an amputation patient will run into problems following surgery, according to a review of nearly 9,000 U.S. patients.
Based on these findings, "we use locoregional anesthesia when possible," and focus on "careful and meticulous handling of tissue," Dr. P. Joshua O’Brien said at the annual meeting of the Southern Surgical Association. "This is the first paper to suggest that locoregional anesthesia may have a protective effect and improve outcomes."
The study results also made Dr. O’Brien and his colleagues at Duke University in Durham, N.C., more aware that amputations "are an important procedure" even though they are often a "junior-level case frequently overseen by a senior resident." The study results prompted Duke attending surgeons to maintain "careful observation of the residents until they feel comfortable that they [the residents] adequately understand the art of performing an amputation," said Dr. O’Brien, a vascular surgeon at Duke.
The analysis he and his associates performed used data collected during 2005-2010 by the National Surgical Quality Improvement Program of the American College of Surgeons. The study included patients who underwent an above-the-knee amputation (3,415 patients – 38%), a below-the-knee amputation (4,258 patients – 48%), or a transmetatarsal amputation (1,205 patients – 14%), but excluded patients who had another surgical procedure with their amputation, prior surgery within 30 days of the amputation, a preoperative do-not-resuscitate order, or missing data; 63% of all the amputation patients had diabetes.
During 30-day postsurgical follow-up, the overall rate of amputation failure was 13%, death occurred in 7%, wound complications affected 9%, and nonwound complications affected 21%. The patients averaged a 6-day postsurgical hospital length of stay.
Early amputation failure showed a statistically significant link with the type of amputation. Patients with a transmetatarsal amputation had a 26% early failure rate, those who underwent a below-the-knee procedure had a 13% failure rate, while above-the-knee amputations failed 8% of the time.
In a multivariate analysis that controlled for patient- and procedure-related factors, several variables linked with statistically significant increases or decreases in the rate of amputation failure. Notable among the factors that increased failure rates were emergency surgery, which boosted the failure rate 2.2-fold compared with nonemergency surgery, and participation of a surgical trainee, which raised the rate 37% compared with the rate when no trainee participated. Trainee participation was common, occurring in 59% of the 8,878 amputations included in the analysis.
Among the factors significantly linked with a reduced rate of amputation failures were use of locoregional anesthesia, which cut the failure rate by 25% compared with general anesthesia, and operative times of at least 40 minutes, which cut failure rates compared with surgery times of less than 40 minutes. The lowest failure rates occurred when the duration of amputation surgery lasted at least 60 minutes. Among patients included in the study, 20% received locoregional anesthesia.
The results also highlighted the important association of amputation failure with other measures of poor surgical outcomes in these amputation patients. Patients who developed amputation failure within 30 days of their surgery also had a nearly sevenfold increased rate of wound complications, and a twofold increased rate of nonwound complications; the average hospital length of stay was 10 days compared with 5 days among patients without amputation. Amputation failure had no significant impact on postoperative mortality, Dr. O’Brien said.
He said he had no relevant financial disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Locoregional anesthesia boosts the success rate of lower-extremity amputations, while time-saving shortcuts and relying too heavily on surgical residents to perform the surgery raise the risk that an amputation patient will run into problems following surgery, according to a review of nearly 9,000 U.S. patients.
Based on these findings, "we use locoregional anesthesia when possible," and focus on "careful and meticulous handling of tissue," Dr. P. Joshua O’Brien said at the annual meeting of the Southern Surgical Association. "This is the first paper to suggest that locoregional anesthesia may have a protective effect and improve outcomes."
The study results also made Dr. O’Brien and his colleagues at Duke University in Durham, N.C., more aware that amputations "are an important procedure" even though they are often a "junior-level case frequently overseen by a senior resident." The study results prompted Duke attending surgeons to maintain "careful observation of the residents until they feel comfortable that they [the residents] adequately understand the art of performing an amputation," said Dr. O’Brien, a vascular surgeon at Duke.
The analysis he and his associates performed used data collected during 2005-2010 by the National Surgical Quality Improvement Program of the American College of Surgeons. The study included patients who underwent an above-the-knee amputation (3,415 patients – 38%), a below-the-knee amputation (4,258 patients – 48%), or a transmetatarsal amputation (1,205 patients – 14%), but excluded patients who had another surgical procedure with their amputation, prior surgery within 30 days of the amputation, a preoperative do-not-resuscitate order, or missing data; 63% of all the amputation patients had diabetes.
During 30-day postsurgical follow-up, the overall rate of amputation failure was 13%, death occurred in 7%, wound complications affected 9%, and nonwound complications affected 21%. The patients averaged a 6-day postsurgical hospital length of stay.
Early amputation failure showed a statistically significant link with the type of amputation. Patients with a transmetatarsal amputation had a 26% early failure rate, those who underwent a below-the-knee procedure had a 13% failure rate, while above-the-knee amputations failed 8% of the time.
In a multivariate analysis that controlled for patient- and procedure-related factors, several variables linked with statistically significant increases or decreases in the rate of amputation failure. Notable among the factors that increased failure rates were emergency surgery, which boosted the failure rate 2.2-fold compared with nonemergency surgery, and participation of a surgical trainee, which raised the rate 37% compared with the rate when no trainee participated. Trainee participation was common, occurring in 59% of the 8,878 amputations included in the analysis.
Among the factors significantly linked with a reduced rate of amputation failures were use of locoregional anesthesia, which cut the failure rate by 25% compared with general anesthesia, and operative times of at least 40 minutes, which cut failure rates compared with surgery times of less than 40 minutes. The lowest failure rates occurred when the duration of amputation surgery lasted at least 60 minutes. Among patients included in the study, 20% received locoregional anesthesia.
The results also highlighted the important association of amputation failure with other measures of poor surgical outcomes in these amputation patients. Patients who developed amputation failure within 30 days of their surgery also had a nearly sevenfold increased rate of wound complications, and a twofold increased rate of nonwound complications; the average hospital length of stay was 10 days compared with 5 days among patients without amputation. Amputation failure had no significant impact on postoperative mortality, Dr. O’Brien said.
He said he had no relevant financial disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Locoregional anesthesia boosts the success rate of lower-extremity amputations, while time-saving shortcuts and relying too heavily on surgical residents to perform the surgery raise the risk that an amputation patient will run into problems following surgery, according to a review of nearly 9,000 U.S. patients.
Based on these findings, "we use locoregional anesthesia when possible," and focus on "careful and meticulous handling of tissue," Dr. P. Joshua O’Brien said at the annual meeting of the Southern Surgical Association. "This is the first paper to suggest that locoregional anesthesia may have a protective effect and improve outcomes."
The study results also made Dr. O’Brien and his colleagues at Duke University in Durham, N.C., more aware that amputations "are an important procedure" even though they are often a "junior-level case frequently overseen by a senior resident." The study results prompted Duke attending surgeons to maintain "careful observation of the residents until they feel comfortable that they [the residents] adequately understand the art of performing an amputation," said Dr. O’Brien, a vascular surgeon at Duke.
The analysis he and his associates performed used data collected during 2005-2010 by the National Surgical Quality Improvement Program of the American College of Surgeons. The study included patients who underwent an above-the-knee amputation (3,415 patients – 38%), a below-the-knee amputation (4,258 patients – 48%), or a transmetatarsal amputation (1,205 patients – 14%), but excluded patients who had another surgical procedure with their amputation, prior surgery within 30 days of the amputation, a preoperative do-not-resuscitate order, or missing data; 63% of all the amputation patients had diabetes.
During 30-day postsurgical follow-up, the overall rate of amputation failure was 13%, death occurred in 7%, wound complications affected 9%, and nonwound complications affected 21%. The patients averaged a 6-day postsurgical hospital length of stay.
Early amputation failure showed a statistically significant link with the type of amputation. Patients with a transmetatarsal amputation had a 26% early failure rate, those who underwent a below-the-knee procedure had a 13% failure rate, while above-the-knee amputations failed 8% of the time.
In a multivariate analysis that controlled for patient- and procedure-related factors, several variables linked with statistically significant increases or decreases in the rate of amputation failure. Notable among the factors that increased failure rates were emergency surgery, which boosted the failure rate 2.2-fold compared with nonemergency surgery, and participation of a surgical trainee, which raised the rate 37% compared with the rate when no trainee participated. Trainee participation was common, occurring in 59% of the 8,878 amputations included in the analysis.
Among the factors significantly linked with a reduced rate of amputation failures were use of locoregional anesthesia, which cut the failure rate by 25% compared with general anesthesia, and operative times of at least 40 minutes, which cut failure rates compared with surgery times of less than 40 minutes. The lowest failure rates occurred when the duration of amputation surgery lasted at least 60 minutes. Among patients included in the study, 20% received locoregional anesthesia.
The results also highlighted the important association of amputation failure with other measures of poor surgical outcomes in these amputation patients. Patients who developed amputation failure within 30 days of their surgery also had a nearly sevenfold increased rate of wound complications, and a twofold increased rate of nonwound complications; the average hospital length of stay was 10 days compared with 5 days among patients without amputation. Amputation failure had no significant impact on postoperative mortality, Dr. O’Brien said.
He said he had no relevant financial disclosures.
On Twitter @mitchelzoler
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: Use of locoregional anesthesia cut the rate of amputation failure within 30 days after surgery 25% compared with general anesthesia.
Data Source: Data came from a review of 8,878 U.S. patients who underwent a lower-extremity amputation during 2005-2010.
Disclosures: Dr. O’Brien said he had no relevant financial disclosures.
New approaches expand kidney transplant pool
PALM BEACH, FLA. – Renal transplant surgeons are using novel methods to expand the pool of donor organs: Using kidneys from donors with acute kidney injury, and vetting and improving the function of kidneys by applying pulsatile machine perfusion to stored kidneys pending transplant.
These approaches can overlap, as machine perfusion has become an important tool for improving the function of kidneys from donors with acute kidney injury (AKI) as well as other marginal kidneys such as those from extended-criteria donors and donation after cardiac death.
Surgeons at Wake Forest University, Winston-Salem, N.C., began transplanting kidneys from AKI donors in 2007, and by mid-2012 they had placed 84 of these organs, resulting in actuarial 5-year patient-survival and graft-survival rates that matched transplants during the same period with kidneys from non-AKI donors, Dr. Alan C. Farney said at the annual meeting of the Southern Surgical Association.
Seventy-four of these kidneys (88%) underwent machine perfusion, for a minimum of 6 hours and more often overnight, said Dr. Farney’s colleague, Dr. Robert J. Stratta, professor of surgery at Wake Forest. "We try to pump whenever possible, and in a perfect world we’d like to see all kidneys pumped" before they are transplanted, Dr. Stratta said. In addition to improving function, mechanical perfusion allows surgeons to assess kidney function. If resistance in the kidney is more than 0.4 or 0.5 mm Hg/mL per minute, "we tend to discard it," he noted.
A second report at the meeting further documented the ability of mechanical perfusion to boost kidney function. In a review of more than 50,000 adult, isolated kidney transplants done on American patients during January 2005–March 2011, machine perfusion prior to transplant led to an average 8-percentage-point cut in the rate of delayed kidney function in a pair of analyses that accounted for baseline patient differences. This means that every 13 kidneys treated before transplant with mechanical perfusion prevented a case of delayed graft function (DGF) following transplantation, resulting in fewer patients requiring hemodialysis, Dr. Glen A. Franklin reported at the meeting.
Prevention of DGF mitigates edema, reduces the need for wound drainage, and decreases the risk for infection, factors that – along with the need for dialysis – drive up costs. Preventing these complications and their associated costs potentially offsets the extra expense of routinely perfusing each kidney before transplantation, Dr. Stratta said.
Dr. Stratta and his associates reviewed the outcomes of 84 transplants of kidneys from donors with AKI done at Wake Forest since 2007 and compared this against the outcomes of 283 concurrent kidney transplants performed during the same 2007-2012 period using organs from donors without AKI. A major, statistically significant difference in protocol for the two types of organs was that 88% of the AKI-derived kidneys underwent machine perfusion before transplant, compared with 51% of the kidneys that came from non-AKI donors, reported Dr. Farney, professor of surgery at Wake Forest.
A major difference in outcomes was that the incidence of DGF following transplantation occurred in 41% of patients who received a kidney from an AKI donor, compared with a 27% DGF rate among patients whose kidneys came from non-AKI donors, a statistically-significant difference.
Despite this, actuarial 5-year patient survival was 98% among the AKI kidney recipients and 90% among the non-AKI kidney recipients. Five-year graft survival was 78% in the AKI-kidney recipients and 71% in patients who received a non-AKI organ. The between-group differences were not statistically significant, Dr. Farney said.
The data also showed an unexpected difference in the way that DGF appeared to affect graft survival. Among patients whose kidneys came from non-AKI donors, the 5-year graft survival rate was 90% among the 206 patients who did not have DGF, but fell to 68% among the 77 patients in this group who had DGF, a statistically-significant difference. In contrast, among patients who received kidneys from AKI donors, the incidence of DGF had no significant impact on long-term graft survival.
The data also showed that the severity of AKI had no significant impact on graft survival. Donors with terminal estimated glomerular filtration rates of 10-20 mL/min per 1.73 m2 had graft survival rates similar to those of patients who received kidneys from AKI donors with terminal estimated glomerular filtration rates above 30 mL/min per 1.732.
Overall, the results suggest that "using kidneys from AKI donors is a safe method to significantly expand the donor pool for kidney transplantation," Dr. Farney concluded.
"I’m impressed with how, for several years, the Wake Forest group has used kidneys that others may have discarded. The outcomes they report are quite encouraging," commented Dr. Douglas P. Slakey, professor and chairman of surgery at Tulane Medical Center, New Orleans.
The impact that machine perfusion of kidneys can have on DGF before transplantation was examined in a much larger data set by Dr. Franklin and his associates at the University of Louisville (Ky.). They reviewed data collected by the United Network for Organ Sharing on 52,052 isolated, adult U.S. kidney transplants during January 2005–March 2011. The series included 19,372 kidneys (37%) that underwent machine perfusion before transplant.
A propensity-score analysis that compared 13,293 organ recipients who received perfused kidneys with 13,293 recipients who received nonperfused kidneys and matched by a variety of donor and recipient characteristics showed that the incidence of DGF during the first week following transplantation was 21% among the perfused organ recipients. The incidence among patients who received a nonperfused kidney was 29%, a statistically significant difference, said Dr. Franklin, a professor of surgery at the University of Louisville.
A second analysis focused on pairs of kidneys that came from 2,290 donors where one kidney underwent perfusion and the other did not. After adjustment for recipient differences, the incidence of DGF was 20% in recipients who received a perfused kidney and 28% in those who received a nonperfused kidney, again an 8% difference that was statistically significant.
The cost of machine perfusion of a kidney varies from region to region, but at Louisville runs about $1,000 to $2,000 per organ, said Dr. Robert M. Cannon, a surgeon at the University of Louisville and a collaborator on the study. "I think you can justify this cost based on the decrease in DGF," and because it allows scheduling of transplants during usual operating-room hours, eliminating the need for emergency transplant surgery in the middle of the night, Dr. Cannon said in an interview.
Several hours of machine perfusion also likely improves kidney function, he noted. "With cold storage, kidneys undergo intense vasospasm that damages the organ. Machine perfusion alleviates that to an extent, and gets more preservation fluid into the organ."
Dr. Farney, Dr. Stratta, Dr. Franklin, and Dr. Cannon, and Dr. Slakey had no disclosures to report.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Renal transplant surgeons are using novel methods to expand the pool of donor organs: Using kidneys from donors with acute kidney injury, and vetting and improving the function of kidneys by applying pulsatile machine perfusion to stored kidneys pending transplant.
These approaches can overlap, as machine perfusion has become an important tool for improving the function of kidneys from donors with acute kidney injury (AKI) as well as other marginal kidneys such as those from extended-criteria donors and donation after cardiac death.
Surgeons at Wake Forest University, Winston-Salem, N.C., began transplanting kidneys from AKI donors in 2007, and by mid-2012 they had placed 84 of these organs, resulting in actuarial 5-year patient-survival and graft-survival rates that matched transplants during the same period with kidneys from non-AKI donors, Dr. Alan C. Farney said at the annual meeting of the Southern Surgical Association.
Seventy-four of these kidneys (88%) underwent machine perfusion, for a minimum of 6 hours and more often overnight, said Dr. Farney’s colleague, Dr. Robert J. Stratta, professor of surgery at Wake Forest. "We try to pump whenever possible, and in a perfect world we’d like to see all kidneys pumped" before they are transplanted, Dr. Stratta said. In addition to improving function, mechanical perfusion allows surgeons to assess kidney function. If resistance in the kidney is more than 0.4 or 0.5 mm Hg/mL per minute, "we tend to discard it," he noted.
A second report at the meeting further documented the ability of mechanical perfusion to boost kidney function. In a review of more than 50,000 adult, isolated kidney transplants done on American patients during January 2005–March 2011, machine perfusion prior to transplant led to an average 8-percentage-point cut in the rate of delayed kidney function in a pair of analyses that accounted for baseline patient differences. This means that every 13 kidneys treated before transplant with mechanical perfusion prevented a case of delayed graft function (DGF) following transplantation, resulting in fewer patients requiring hemodialysis, Dr. Glen A. Franklin reported at the meeting.
Prevention of DGF mitigates edema, reduces the need for wound drainage, and decreases the risk for infection, factors that – along with the need for dialysis – drive up costs. Preventing these complications and their associated costs potentially offsets the extra expense of routinely perfusing each kidney before transplantation, Dr. Stratta said.
Dr. Stratta and his associates reviewed the outcomes of 84 transplants of kidneys from donors with AKI done at Wake Forest since 2007 and compared this against the outcomes of 283 concurrent kidney transplants performed during the same 2007-2012 period using organs from donors without AKI. A major, statistically significant difference in protocol for the two types of organs was that 88% of the AKI-derived kidneys underwent machine perfusion before transplant, compared with 51% of the kidneys that came from non-AKI donors, reported Dr. Farney, professor of surgery at Wake Forest.
A major difference in outcomes was that the incidence of DGF following transplantation occurred in 41% of patients who received a kidney from an AKI donor, compared with a 27% DGF rate among patients whose kidneys came from non-AKI donors, a statistically-significant difference.
Despite this, actuarial 5-year patient survival was 98% among the AKI kidney recipients and 90% among the non-AKI kidney recipients. Five-year graft survival was 78% in the AKI-kidney recipients and 71% in patients who received a non-AKI organ. The between-group differences were not statistically significant, Dr. Farney said.
The data also showed an unexpected difference in the way that DGF appeared to affect graft survival. Among patients whose kidneys came from non-AKI donors, the 5-year graft survival rate was 90% among the 206 patients who did not have DGF, but fell to 68% among the 77 patients in this group who had DGF, a statistically-significant difference. In contrast, among patients who received kidneys from AKI donors, the incidence of DGF had no significant impact on long-term graft survival.
The data also showed that the severity of AKI had no significant impact on graft survival. Donors with terminal estimated glomerular filtration rates of 10-20 mL/min per 1.73 m2 had graft survival rates similar to those of patients who received kidneys from AKI donors with terminal estimated glomerular filtration rates above 30 mL/min per 1.732.
Overall, the results suggest that "using kidneys from AKI donors is a safe method to significantly expand the donor pool for kidney transplantation," Dr. Farney concluded.
"I’m impressed with how, for several years, the Wake Forest group has used kidneys that others may have discarded. The outcomes they report are quite encouraging," commented Dr. Douglas P. Slakey, professor and chairman of surgery at Tulane Medical Center, New Orleans.
The impact that machine perfusion of kidneys can have on DGF before transplantation was examined in a much larger data set by Dr. Franklin and his associates at the University of Louisville (Ky.). They reviewed data collected by the United Network for Organ Sharing on 52,052 isolated, adult U.S. kidney transplants during January 2005–March 2011. The series included 19,372 kidneys (37%) that underwent machine perfusion before transplant.
A propensity-score analysis that compared 13,293 organ recipients who received perfused kidneys with 13,293 recipients who received nonperfused kidneys and matched by a variety of donor and recipient characteristics showed that the incidence of DGF during the first week following transplantation was 21% among the perfused organ recipients. The incidence among patients who received a nonperfused kidney was 29%, a statistically significant difference, said Dr. Franklin, a professor of surgery at the University of Louisville.
A second analysis focused on pairs of kidneys that came from 2,290 donors where one kidney underwent perfusion and the other did not. After adjustment for recipient differences, the incidence of DGF was 20% in recipients who received a perfused kidney and 28% in those who received a nonperfused kidney, again an 8% difference that was statistically significant.
The cost of machine perfusion of a kidney varies from region to region, but at Louisville runs about $1,000 to $2,000 per organ, said Dr. Robert M. Cannon, a surgeon at the University of Louisville and a collaborator on the study. "I think you can justify this cost based on the decrease in DGF," and because it allows scheduling of transplants during usual operating-room hours, eliminating the need for emergency transplant surgery in the middle of the night, Dr. Cannon said in an interview.
Several hours of machine perfusion also likely improves kidney function, he noted. "With cold storage, kidneys undergo intense vasospasm that damages the organ. Machine perfusion alleviates that to an extent, and gets more preservation fluid into the organ."
Dr. Farney, Dr. Stratta, Dr. Franklin, and Dr. Cannon, and Dr. Slakey had no disclosures to report.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Renal transplant surgeons are using novel methods to expand the pool of donor organs: Using kidneys from donors with acute kidney injury, and vetting and improving the function of kidneys by applying pulsatile machine perfusion to stored kidneys pending transplant.
These approaches can overlap, as machine perfusion has become an important tool for improving the function of kidneys from donors with acute kidney injury (AKI) as well as other marginal kidneys such as those from extended-criteria donors and donation after cardiac death.
Surgeons at Wake Forest University, Winston-Salem, N.C., began transplanting kidneys from AKI donors in 2007, and by mid-2012 they had placed 84 of these organs, resulting in actuarial 5-year patient-survival and graft-survival rates that matched transplants during the same period with kidneys from non-AKI donors, Dr. Alan C. Farney said at the annual meeting of the Southern Surgical Association.
Seventy-four of these kidneys (88%) underwent machine perfusion, for a minimum of 6 hours and more often overnight, said Dr. Farney’s colleague, Dr. Robert J. Stratta, professor of surgery at Wake Forest. "We try to pump whenever possible, and in a perfect world we’d like to see all kidneys pumped" before they are transplanted, Dr. Stratta said. In addition to improving function, mechanical perfusion allows surgeons to assess kidney function. If resistance in the kidney is more than 0.4 or 0.5 mm Hg/mL per minute, "we tend to discard it," he noted.
A second report at the meeting further documented the ability of mechanical perfusion to boost kidney function. In a review of more than 50,000 adult, isolated kidney transplants done on American patients during January 2005–March 2011, machine perfusion prior to transplant led to an average 8-percentage-point cut in the rate of delayed kidney function in a pair of analyses that accounted for baseline patient differences. This means that every 13 kidneys treated before transplant with mechanical perfusion prevented a case of delayed graft function (DGF) following transplantation, resulting in fewer patients requiring hemodialysis, Dr. Glen A. Franklin reported at the meeting.
Prevention of DGF mitigates edema, reduces the need for wound drainage, and decreases the risk for infection, factors that – along with the need for dialysis – drive up costs. Preventing these complications and their associated costs potentially offsets the extra expense of routinely perfusing each kidney before transplantation, Dr. Stratta said.
Dr. Stratta and his associates reviewed the outcomes of 84 transplants of kidneys from donors with AKI done at Wake Forest since 2007 and compared this against the outcomes of 283 concurrent kidney transplants performed during the same 2007-2012 period using organs from donors without AKI. A major, statistically significant difference in protocol for the two types of organs was that 88% of the AKI-derived kidneys underwent machine perfusion before transplant, compared with 51% of the kidneys that came from non-AKI donors, reported Dr. Farney, professor of surgery at Wake Forest.
A major difference in outcomes was that the incidence of DGF following transplantation occurred in 41% of patients who received a kidney from an AKI donor, compared with a 27% DGF rate among patients whose kidneys came from non-AKI donors, a statistically-significant difference.
Despite this, actuarial 5-year patient survival was 98% among the AKI kidney recipients and 90% among the non-AKI kidney recipients. Five-year graft survival was 78% in the AKI-kidney recipients and 71% in patients who received a non-AKI organ. The between-group differences were not statistically significant, Dr. Farney said.
The data also showed an unexpected difference in the way that DGF appeared to affect graft survival. Among patients whose kidneys came from non-AKI donors, the 5-year graft survival rate was 90% among the 206 patients who did not have DGF, but fell to 68% among the 77 patients in this group who had DGF, a statistically-significant difference. In contrast, among patients who received kidneys from AKI donors, the incidence of DGF had no significant impact on long-term graft survival.
The data also showed that the severity of AKI had no significant impact on graft survival. Donors with terminal estimated glomerular filtration rates of 10-20 mL/min per 1.73 m2 had graft survival rates similar to those of patients who received kidneys from AKI donors with terminal estimated glomerular filtration rates above 30 mL/min per 1.732.
Overall, the results suggest that "using kidneys from AKI donors is a safe method to significantly expand the donor pool for kidney transplantation," Dr. Farney concluded.
"I’m impressed with how, for several years, the Wake Forest group has used kidneys that others may have discarded. The outcomes they report are quite encouraging," commented Dr. Douglas P. Slakey, professor and chairman of surgery at Tulane Medical Center, New Orleans.
The impact that machine perfusion of kidneys can have on DGF before transplantation was examined in a much larger data set by Dr. Franklin and his associates at the University of Louisville (Ky.). They reviewed data collected by the United Network for Organ Sharing on 52,052 isolated, adult U.S. kidney transplants during January 2005–March 2011. The series included 19,372 kidneys (37%) that underwent machine perfusion before transplant.
A propensity-score analysis that compared 13,293 organ recipients who received perfused kidneys with 13,293 recipients who received nonperfused kidneys and matched by a variety of donor and recipient characteristics showed that the incidence of DGF during the first week following transplantation was 21% among the perfused organ recipients. The incidence among patients who received a nonperfused kidney was 29%, a statistically significant difference, said Dr. Franklin, a professor of surgery at the University of Louisville.
A second analysis focused on pairs of kidneys that came from 2,290 donors where one kidney underwent perfusion and the other did not. After adjustment for recipient differences, the incidence of DGF was 20% in recipients who received a perfused kidney and 28% in those who received a nonperfused kidney, again an 8% difference that was statistically significant.
The cost of machine perfusion of a kidney varies from region to region, but at Louisville runs about $1,000 to $2,000 per organ, said Dr. Robert M. Cannon, a surgeon at the University of Louisville and a collaborator on the study. "I think you can justify this cost based on the decrease in DGF," and because it allows scheduling of transplants during usual operating-room hours, eliminating the need for emergency transplant surgery in the middle of the night, Dr. Cannon said in an interview.
Several hours of machine perfusion also likely improves kidney function, he noted. "With cold storage, kidneys undergo intense vasospasm that damages the organ. Machine perfusion alleviates that to an extent, and gets more preservation fluid into the organ."
Dr. Farney, Dr. Stratta, Dr. Franklin, and Dr. Cannon, and Dr. Slakey had no disclosures to report.
On Twitter @mitchelzoler
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: Machine perfusion of donor kidneys before transplantation cut delayed-graft function rate by 8%.
Data Source: A review of 52,052 U.S. adults who received an isolated kidney transplant during 2007-2012.
Disclosures:. Dr. Farney, Dr. Stratta, Dr. Franklin, and Dr. Cannon, and Dr. Slakey had no disclosures to report.
Novel OR scheduling and staffing boosts efficiency
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: During the first year, surgical cases rose 4%, overtime fell 27%, staff turnover fell 8%, and hospital margin rose 5%.
Data Source: The first-year experience of new operating room staffing and scheduling procedures at one U.S. hospital.
Disclosures: Dr. Smith said that he had no disclosures.
Comprehensive mechanical circulatory support guidelines issued
As a further sign of how much mechanical circulatory support for advanced heart failure has matured, the International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment" said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"When MSC started, the role of the devices was as a bridge to heart transplantation, but the field has evolved over the past decade and now MCS for destination therapy has opened a new array of patients who could benefit from these devices," said Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham. "We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," she said in an interview.
"As pumps improve and the number of patients with advanced heart failure increases more and more patients will receive a ventricular assist device [VAD], and heart transplant will grow less relevant. These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD, and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and another cochair of the guidelines committee.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-87).
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients. Even a busy center may put in 50 or 60 VADs a year. Hopefully, a result of the guidelines is that they will help centers get together and produce the critical mass of patients needed to conduct meaningful trials. It was time to get something on paper; the new guidelines are what we will now work off of." But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The most limited number of recommendations came from the third task force of the panel, which handled intraoperative procedures and immediate postoperative care. Though this section runs 17 pages and deals with topics such as anesthesia, implantation techniques, establishing hemostasis, performing concomitant procedures, methods for explantation, and management of postoperative hemodynamics and bleeding, it contains just three specific recommendations, all dealing with anesthesia. "There are essentially no studies that have looked at how to make things better in the surgical suite," explained Dr. Feldman.
"It’s very challenging to standardize a surgical procedure," added Dr. Pamboukian. "We tried to summarize useful practices, but consensus-based recommendations are difficult to do."
Another topic the guidelines finesse is patient selection. The field is currently trying to sort out the best stage of advanced heart failure for patients to receive mechanical circulatory support. "You’d be amazed at the disparity of who gets these devices now," Dr. Feldman said. In addition, the guideline writing committee decided to defer definitive choices until results are available from a large study starting later this year. The study, Evaluation of VAD Intervention Before Inotropic Therapy (REVIVE-IT), will examine the outcomes of patients with advanced New York Heart Association stage III heart failure who receive a VAD. "It didn’t seem appropriate to address this because of the trial," he added.
Both Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
As a further sign of how much mechanical circulatory support for advanced heart failure has matured, the International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment" said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"When MSC started, the role of the devices was as a bridge to heart transplantation, but the field has evolved over the past decade and now MCS for destination therapy has opened a new array of patients who could benefit from these devices," said Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham. "We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," she said in an interview.
"As pumps improve and the number of patients with advanced heart failure increases more and more patients will receive a ventricular assist device [VAD], and heart transplant will grow less relevant. These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD, and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and another cochair of the guidelines committee.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-87).
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients. Even a busy center may put in 50 or 60 VADs a year. Hopefully, a result of the guidelines is that they will help centers get together and produce the critical mass of patients needed to conduct meaningful trials. It was time to get something on paper; the new guidelines are what we will now work off of." But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The most limited number of recommendations came from the third task force of the panel, which handled intraoperative procedures and immediate postoperative care. Though this section runs 17 pages and deals with topics such as anesthesia, implantation techniques, establishing hemostasis, performing concomitant procedures, methods for explantation, and management of postoperative hemodynamics and bleeding, it contains just three specific recommendations, all dealing with anesthesia. "There are essentially no studies that have looked at how to make things better in the surgical suite," explained Dr. Feldman.
"It’s very challenging to standardize a surgical procedure," added Dr. Pamboukian. "We tried to summarize useful practices, but consensus-based recommendations are difficult to do."
Another topic the guidelines finesse is patient selection. The field is currently trying to sort out the best stage of advanced heart failure for patients to receive mechanical circulatory support. "You’d be amazed at the disparity of who gets these devices now," Dr. Feldman said. In addition, the guideline writing committee decided to defer definitive choices until results are available from a large study starting later this year. The study, Evaluation of VAD Intervention Before Inotropic Therapy (REVIVE-IT), will examine the outcomes of patients with advanced New York Heart Association stage III heart failure who receive a VAD. "It didn’t seem appropriate to address this because of the trial," he added.
Both Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
As a further sign of how much mechanical circulatory support for advanced heart failure has matured, the International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment" said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"When MSC started, the role of the devices was as a bridge to heart transplantation, but the field has evolved over the past decade and now MCS for destination therapy has opened a new array of patients who could benefit from these devices," said Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham. "We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," she said in an interview.
"As pumps improve and the number of patients with advanced heart failure increases more and more patients will receive a ventricular assist device [VAD], and heart transplant will grow less relevant. These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD, and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and another cochair of the guidelines committee.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-87).
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients. Even a busy center may put in 50 or 60 VADs a year. Hopefully, a result of the guidelines is that they will help centers get together and produce the critical mass of patients needed to conduct meaningful trials. It was time to get something on paper; the new guidelines are what we will now work off of." But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The most limited number of recommendations came from the third task force of the panel, which handled intraoperative procedures and immediate postoperative care. Though this section runs 17 pages and deals with topics such as anesthesia, implantation techniques, establishing hemostasis, performing concomitant procedures, methods for explantation, and management of postoperative hemodynamics and bleeding, it contains just three specific recommendations, all dealing with anesthesia. "There are essentially no studies that have looked at how to make things better in the surgical suite," explained Dr. Feldman.
"It’s very challenging to standardize a surgical procedure," added Dr. Pamboukian. "We tried to summarize useful practices, but consensus-based recommendations are difficult to do."
Another topic the guidelines finesse is patient selection. The field is currently trying to sort out the best stage of advanced heart failure for patients to receive mechanical circulatory support. "You’d be amazed at the disparity of who gets these devices now," Dr. Feldman said. In addition, the guideline writing committee decided to defer definitive choices until results are available from a large study starting later this year. The study, Evaluation of VAD Intervention Before Inotropic Therapy (REVIVE-IT), will examine the outcomes of patients with advanced New York Heart Association stage III heart failure who receive a VAD. "It didn’t seem appropriate to address this because of the trial," he added.
Both Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
FROM THE JOURNAL OF HEART AND LUNG TRANSPLANTATION
Night on call has no effect on next-day operations
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: General surgeons who performed 132 elective operations after a night on call had no increased complications, readmissions, or deaths, compared with surgeons not on call the previous night (737 surgeries).
Data Source: A review of 869 surgeries at one U.S. center during 2006-2009.
Disclosures: Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
Mechanical circulatory support guidelines issued
The International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally, management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment," said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham, said in an interview.
"These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD (ventricular assist device), and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and a cochair of the guidelines committee.
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients."
But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-187).
The fourth of the five task forces addressed inpatient management of patients with MCSDs and was cochaired by Dr. Pamboukian and Dr. Stephanie A. Moore. Select recommendations may be of interest to hospitalists.
According to this task force, a multidisciplinary team led cooperatively by cardiac surgeons and cardiologists and composed of subspecialists (palliative care, psychiatry, and others as needed), MCS coordinators, and other ancillary specialties (social worker, psychologist, pharmacist, dietitian, physical therapist, occupational therapist, and rehabilitation services) is indicated for the in-hospital management of MCS patients.
For patients who develop right heart dysfunction in the non-ICU postoperative period, the task force said, inotropic support may need to be continued beyond 2 weeks if there is elevated jugular venous pressure, signs of venous congestion, decreased VAD flows (or low pulsatility in continuous flow MCSD), or end-organ dysfunction. Once euvolemic, inotropes should be weaned cautiously, with ongoing examination for recurrent signs and symptoms of RV dysfunction.
Diuretics and renal replacement therapy, such as continuous venovenous hemofiltration, should be used early and continued as needed to maintain optimal volume status.
Cardiac glycosides may be used to support RV function. For patients with persistent pulmonary hypertension who exhibit signs of RV dysfunction, pulmonary hypertension-specific therapies, such as phosphodiesterase-5 inhibitors, should be considered.
Recommendations for neurohormonal blockade and the treatment of hypertension post-MCS implant include pharmacotherapy with heart failure medications for blood pressure management.
Echocardiography is an integral part of determining the revolutions per minute of continuous-flow pumps. Common goals include adequate LV unloading while maintaining the LV septum in the midline and minimizing mitral regurgitation. Postoperatively, the revolutions per minute of continuous-flow pumps should be set low enough to allow for intermittent aortic valve opening which may reduce the risk of aortic valve fusion and the risk of late aortic valve insufficiency.
Anticoagulation and antiplatelet therapy initiated post operatively in the ICU setting should be continued with the aim of achieving device-specific recommended INR for warfarin and desired antiplatelet effects, according to the task force.
Bleeding in the early postoperative period during the index hospitalization should be urgently evaluated with lowering, discontinuation, and/or reversal of anticoagulation and antiplatelet medications.
To reduce risk for infection after MCS placement, the driveline should be stabilized immediately after the device is placed and throughout the duration of support. A dressing change protocol should be immediately initiated post operatively. Secondary antibiotic prophylaxis for prevention of endocarditis has not been studied in the MCS population but would be considered reasonable due to the risk of bacteremia in this group.
Recommendations for optimal nutritional status include consultation with nutritional services at the time of implantation with ongoing follow-up post operatively to ensure nutrition goals are being met. For those unable to meet nutritional goals orally, feeding should be started early and preferably through an enteral feeding tube. Parenteral nutrition should only be started if enteral nutrition is not possible and under the guidance of nutritional consultation.
Prealbumin and C-reactive protein levels can be monitored weekly to track the nutritional status of the postoperative patient. As nutrition improves, prealbumin should rise and C-reactive protein should decrease.
Patient and caregiver education should be initiated shortly after surgery and reinforced by the nursing staff. Educational strategies should use written, verbal, and practical methods.
MCS parameters should be written in the medical record at regular intervals with established criteria for parameters that require physician notification.
The patient and family members should be taught to track their device parameters and alert staff when changes are observed. Changes in parameters outside of normal ranges should be thoroughly evaluated and treated appropriately.
Routine support should be available from social workers, psychologists, or psychiatrists as patients and families adjust to life changes after MCS. Routine surveillance for psychiatric symptoms should be performed. If symptoms develop, consultation with specialists (including social work, psychology, and/or psychiatry) for diagnosis, treatment, and follow-up is recommended.
Assessment of specific problems that are related to domains of health-related quality of life (depression, anxiety, or pain) based on symptoms should help guide an action plan for these patients.
Successful discharge should include patient, caregiver and community provider education with written discharge instructions and preemptive home preparation regarding the safe management of the device and the MCS patient.
Recommendations for management of anticoagulation and antiplatelet therapy for patients who present with gastrointestinal bleeding include holding back anticoagulation and antiplatelet therapy in the setting of clinically significant bleeding and reversing anticoagulation in the setting of an elevated INR and clinically significant bleeding.
Anticoagulation and antiplatelet therapy should continue to be held until clinically significant bleeding resolves in the absence of evidence of pump dysfunction. The patient, device parameters, and the pump housing (if applicable) should be carefully monitored while anticoagulation and antiplatelet therapy is being withheld or the dose reduced.
Patients who present with a first episode of gastrointestinal bleeding should be managed in consultation with gastroenterology. Patients should at least have a colonoscopy and/or upper endoscopic evaluation. If the results are negative, evaluation of the small bowel, particularly in those with continuous-flow devices, should be considered.
In patients with persistent bleeding and a negative endoscopic evaluation, a tagged red blood scan or angiography should be considered.
Once the gastrointestinal bleeding has resolved, anticoagulation and antiplatelet therapy can be reintroduced with careful monitoring.
For patients with recurrent episodes of gastrointestinal bleeding, repeated endoscopic evaluation should take place in conjunction with gastroenterology consultation. When no source of bleeding can be detected or the source is not amenable to therapy, the type and intensity or even the use of antiplatelet therapy should be reevaluated in the context of the bleeding severity and pump type. Further, the goal INR or even the continued use of warfarin should be reevaluated in the context of the bleeding severity and pump type.
The patient and device parameters should be carefully monitored when anticoagulation and antiplatelet therapy have been reduced or discontinued because of recurrent gastrointestinal bleeding.
Reducing the pump speed for continuous-flow pumps in the setting of recurrent gastrointestinal bleeding due to arteriovenous malformations may be considered.
When patients present with a new neurologic deficit, assess the current INR and review the recent INR. Prompt consultation with neurology is recommended, as is CT and angiography of the head and neck.
Review the pump parameters for signs of device thrombosis or malfunction, and inspect the pump housing for clots in extracorporeal pumps. Discontinuation or reversal of anticoagulation in the setting of hemorrhagic stroke is recommended.
Assessing for the source of the thrombus should be considered in embolic stroke patients. Selective use of an interventional radiologic approach to thrombotic strokes or selective use of thrombolytic agents in the setting of thrombotic stroke without CT scan evidence of hemorrhage may be considered.
Chronic management of patients after presentation with a new neurologic deficit may include formal stroke rehabilitation in consultation with neurology, close monitoring of anticoagulation in the setting of an embolic event to ensure adequate levels of anticoagulation, long-term control of blood pressure, administration of the National Institutes of Health (NIH) stroke scale at 30 and 60 days, and resumption of anticoagulation in consultation with neurology or neurosurgery in the setting of hemorrhagic stroke.
Recommendations for assessment of neurocognitive deficits include routine neurocognitive assessment at 3, 6, 12, and 18 months after implant.
Recommendations for evaluation of MCS patients with a suspected infection include a complete blood count, chest radiographic imaging, and blood cultures.
At least three sets of blood cultures over 24 hours should be drawn, with at least one culture from any indwelling central venous catheters.
For those with a suspected cannula or driveline infection, obtaining a sample for Gram stain, KOH, and routine bacterial and fungal cultures is recommended.
When clinically indicated, aspirate from other potential sources, as dictated by presenting symptoms and examination.
Directed radiographic studies based on presenting symptoms and examination are recommended.
MCS patients with incessant ventricular arrhythmias require prompt admission for further management because hemodynamic compromise may occur. Patients with ongoing VT refractory to medical therapy may require catheter ablation, which should be performed by an electrophysiologist with expertise in treating patients with MCS.
When an MCS patient is undergoing a noncardiac procedure, the MCS and surgical teams need to collaborate. For nonemergency procedures, warfarin and antiplatelet therapy may be continued if the risk of bleeding associated with the procedure is low. If therapy needs to be stopped, warfarin and antiplatelet therapy should be held for an appropriate period of time as determined by the type of procedure being undertaken and risk of bleeding. Bridging with heparin or a heparin alternative while a patient is off warfarin may be considered.
For emergency procedures, warfarin may need to be rapidly reversed with fresh frozen plasma or prothrombin protein concentrate. Vitamin K can be administered with caution, but has slower onset of action. Post procedure, warfarin and antiplatelet therapy may be resumed when risk of surgical bleeding is deemed acceptable. Patients may be bridged with heparin or a heparin alternative while waiting for the INR to reach the target range.
Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
The International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally, management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment," said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham, said in an interview.
"These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD (ventricular assist device), and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and a cochair of the guidelines committee.
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients."
But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-187).
The fourth of the five task forces addressed inpatient management of patients with MCSDs and was cochaired by Dr. Pamboukian and Dr. Stephanie A. Moore. Select recommendations may be of interest to hospitalists.
According to this task force, a multidisciplinary team led cooperatively by cardiac surgeons and cardiologists and composed of subspecialists (palliative care, psychiatry, and others as needed), MCS coordinators, and other ancillary specialties (social worker, psychologist, pharmacist, dietitian, physical therapist, occupational therapist, and rehabilitation services) is indicated for the in-hospital management of MCS patients.
For patients who develop right heart dysfunction in the non-ICU postoperative period, the task force said, inotropic support may need to be continued beyond 2 weeks if there is elevated jugular venous pressure, signs of venous congestion, decreased VAD flows (or low pulsatility in continuous flow MCSD), or end-organ dysfunction. Once euvolemic, inotropes should be weaned cautiously, with ongoing examination for recurrent signs and symptoms of RV dysfunction.
Diuretics and renal replacement therapy, such as continuous venovenous hemofiltration, should be used early and continued as needed to maintain optimal volume status.
Cardiac glycosides may be used to support RV function. For patients with persistent pulmonary hypertension who exhibit signs of RV dysfunction, pulmonary hypertension-specific therapies, such as phosphodiesterase-5 inhibitors, should be considered.
Recommendations for neurohormonal blockade and the treatment of hypertension post-MCS implant include pharmacotherapy with heart failure medications for blood pressure management.
Echocardiography is an integral part of determining the revolutions per minute of continuous-flow pumps. Common goals include adequate LV unloading while maintaining the LV septum in the midline and minimizing mitral regurgitation. Postoperatively, the revolutions per minute of continuous-flow pumps should be set low enough to allow for intermittent aortic valve opening which may reduce the risk of aortic valve fusion and the risk of late aortic valve insufficiency.
Anticoagulation and antiplatelet therapy initiated post operatively in the ICU setting should be continued with the aim of achieving device-specific recommended INR for warfarin and desired antiplatelet effects, according to the task force.
Bleeding in the early postoperative period during the index hospitalization should be urgently evaluated with lowering, discontinuation, and/or reversal of anticoagulation and antiplatelet medications.
To reduce risk for infection after MCS placement, the driveline should be stabilized immediately after the device is placed and throughout the duration of support. A dressing change protocol should be immediately initiated post operatively. Secondary antibiotic prophylaxis for prevention of endocarditis has not been studied in the MCS population but would be considered reasonable due to the risk of bacteremia in this group.
Recommendations for optimal nutritional status include consultation with nutritional services at the time of implantation with ongoing follow-up post operatively to ensure nutrition goals are being met. For those unable to meet nutritional goals orally, feeding should be started early and preferably through an enteral feeding tube. Parenteral nutrition should only be started if enteral nutrition is not possible and under the guidance of nutritional consultation.
Prealbumin and C-reactive protein levels can be monitored weekly to track the nutritional status of the postoperative patient. As nutrition improves, prealbumin should rise and C-reactive protein should decrease.
Patient and caregiver education should be initiated shortly after surgery and reinforced by the nursing staff. Educational strategies should use written, verbal, and practical methods.
MCS parameters should be written in the medical record at regular intervals with established criteria for parameters that require physician notification.
The patient and family members should be taught to track their device parameters and alert staff when changes are observed. Changes in parameters outside of normal ranges should be thoroughly evaluated and treated appropriately.
Routine support should be available from social workers, psychologists, or psychiatrists as patients and families adjust to life changes after MCS. Routine surveillance for psychiatric symptoms should be performed. If symptoms develop, consultation with specialists (including social work, psychology, and/or psychiatry) for diagnosis, treatment, and follow-up is recommended.
Assessment of specific problems that are related to domains of health-related quality of life (depression, anxiety, or pain) based on symptoms should help guide an action plan for these patients.
Successful discharge should include patient, caregiver and community provider education with written discharge instructions and preemptive home preparation regarding the safe management of the device and the MCS patient.
Recommendations for management of anticoagulation and antiplatelet therapy for patients who present with gastrointestinal bleeding include holding back anticoagulation and antiplatelet therapy in the setting of clinically significant bleeding and reversing anticoagulation in the setting of an elevated INR and clinically significant bleeding.
Anticoagulation and antiplatelet therapy should continue to be held until clinically significant bleeding resolves in the absence of evidence of pump dysfunction. The patient, device parameters, and the pump housing (if applicable) should be carefully monitored while anticoagulation and antiplatelet therapy is being withheld or the dose reduced.
Patients who present with a first episode of gastrointestinal bleeding should be managed in consultation with gastroenterology. Patients should at least have a colonoscopy and/or upper endoscopic evaluation. If the results are negative, evaluation of the small bowel, particularly in those with continuous-flow devices, should be considered.
In patients with persistent bleeding and a negative endoscopic evaluation, a tagged red blood scan or angiography should be considered.
Once the gastrointestinal bleeding has resolved, anticoagulation and antiplatelet therapy can be reintroduced with careful monitoring.
For patients with recurrent episodes of gastrointestinal bleeding, repeated endoscopic evaluation should take place in conjunction with gastroenterology consultation. When no source of bleeding can be detected or the source is not amenable to therapy, the type and intensity or even the use of antiplatelet therapy should be reevaluated in the context of the bleeding severity and pump type. Further, the goal INR or even the continued use of warfarin should be reevaluated in the context of the bleeding severity and pump type.
The patient and device parameters should be carefully monitored when anticoagulation and antiplatelet therapy have been reduced or discontinued because of recurrent gastrointestinal bleeding.
Reducing the pump speed for continuous-flow pumps in the setting of recurrent gastrointestinal bleeding due to arteriovenous malformations may be considered.
When patients present with a new neurologic deficit, assess the current INR and review the recent INR. Prompt consultation with neurology is recommended, as is CT and angiography of the head and neck.
Review the pump parameters for signs of device thrombosis or malfunction, and inspect the pump housing for clots in extracorporeal pumps. Discontinuation or reversal of anticoagulation in the setting of hemorrhagic stroke is recommended.
Assessing for the source of the thrombus should be considered in embolic stroke patients. Selective use of an interventional radiologic approach to thrombotic strokes or selective use of thrombolytic agents in the setting of thrombotic stroke without CT scan evidence of hemorrhage may be considered.
Chronic management of patients after presentation with a new neurologic deficit may include formal stroke rehabilitation in consultation with neurology, close monitoring of anticoagulation in the setting of an embolic event to ensure adequate levels of anticoagulation, long-term control of blood pressure, administration of the National Institutes of Health (NIH) stroke scale at 30 and 60 days, and resumption of anticoagulation in consultation with neurology or neurosurgery in the setting of hemorrhagic stroke.
Recommendations for assessment of neurocognitive deficits include routine neurocognitive assessment at 3, 6, 12, and 18 months after implant.
Recommendations for evaluation of MCS patients with a suspected infection include a complete blood count, chest radiographic imaging, and blood cultures.
At least three sets of blood cultures over 24 hours should be drawn, with at least one culture from any indwelling central venous catheters.
For those with a suspected cannula or driveline infection, obtaining a sample for Gram stain, KOH, and routine bacterial and fungal cultures is recommended.
When clinically indicated, aspirate from other potential sources, as dictated by presenting symptoms and examination.
Directed radiographic studies based on presenting symptoms and examination are recommended.
MCS patients with incessant ventricular arrhythmias require prompt admission for further management because hemodynamic compromise may occur. Patients with ongoing VT refractory to medical therapy may require catheter ablation, which should be performed by an electrophysiologist with expertise in treating patients with MCS.
When an MCS patient is undergoing a noncardiac procedure, the MCS and surgical teams need to collaborate. For nonemergency procedures, warfarin and antiplatelet therapy may be continued if the risk of bleeding associated with the procedure is low. If therapy needs to be stopped, warfarin and antiplatelet therapy should be held for an appropriate period of time as determined by the type of procedure being undertaken and risk of bleeding. Bridging with heparin or a heparin alternative while a patient is off warfarin may be considered.
For emergency procedures, warfarin may need to be rapidly reversed with fresh frozen plasma or prothrombin protein concentrate. Vitamin K can be administered with caution, but has slower onset of action. Post procedure, warfarin and antiplatelet therapy may be resumed when risk of surgical bleeding is deemed acceptable. Patients may be bridged with heparin or a heparin alternative while waiting for the INR to reach the target range.
Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
The International Society of Heart and Lung Transplantation issued on Jan. 10 the first comprehensive guidelines for all phases of evaluating, implanting, and managing patients who receive left ventricular assist devices or related equipment.
"Traditionally, management of patients with mechanical circulatory support [MCS] was very center specific, but because the number of treated patients has increased, and because patients now live with these devices for years, we reached a point where we needed best practices guidelines, an expert consensus on what is the best way to approach this treatment," said Dr. Salpy V. Pamboukian, a cardiologist and one of three cochairs of the guidelines-writing project.
"We hope these guidelines will serve as a springboard for further research into the long-term management of these patients," Dr. Pamboukian, medical director of the MCS device program at the University of Alabama, Birmingham, said in an interview.
"These guidelines are much more comprehensive [than anything previously published] and they represent the opinions of the physicians, surgeons, nurses, and other providers who care for these patients," said Dr. David S. Feldman, a cardiologist who is director of the heart failure, VAD (ventricular assist device), and cardiac transplantation program at the Minneapolis Heart Institute at Abbott Northwestern Hospital, and a cochair of the guidelines committee.
The writing committee admitted up front in the paper that most of the recommendations are consensus opinions with no clear evidence base. "It’s a limitation," admitted Dr. Pamboukian, "but you need a common approach to patients."
But despite an absence of evidence on which to base many recommendations, "I was pleasantly surprised that there was more consensus than controversy. There was more commonality in our approaches than differences," she added.
The guidelines, which took about 3 years to produce, came from a committee of 35 health care providers, with initial review by three independent experts followed by additional peer review and then a period of open comment from the society’s membership. The 146-page document consists of more than 250 individual recommendations presented in five sections: patient selection; risk management prior to surgery; intraoperative procedures and immediate postoperative management; in-patient management during the immediate postoperative period; and long-term outpatient management (J. Heart Lung Transplant. 2013;32:157-187).
The fourth of the five task forces addressed inpatient management of patients with MCSDs and was cochaired by Dr. Pamboukian and Dr. Stephanie A. Moore. Select recommendations may be of interest to hospitalists.
According to this task force, a multidisciplinary team led cooperatively by cardiac surgeons and cardiologists and composed of subspecialists (palliative care, psychiatry, and others as needed), MCS coordinators, and other ancillary specialties (social worker, psychologist, pharmacist, dietitian, physical therapist, occupational therapist, and rehabilitation services) is indicated for the in-hospital management of MCS patients.
For patients who develop right heart dysfunction in the non-ICU postoperative period, the task force said, inotropic support may need to be continued beyond 2 weeks if there is elevated jugular venous pressure, signs of venous congestion, decreased VAD flows (or low pulsatility in continuous flow MCSD), or end-organ dysfunction. Once euvolemic, inotropes should be weaned cautiously, with ongoing examination for recurrent signs and symptoms of RV dysfunction.
Diuretics and renal replacement therapy, such as continuous venovenous hemofiltration, should be used early and continued as needed to maintain optimal volume status.
Cardiac glycosides may be used to support RV function. For patients with persistent pulmonary hypertension who exhibit signs of RV dysfunction, pulmonary hypertension-specific therapies, such as phosphodiesterase-5 inhibitors, should be considered.
Recommendations for neurohormonal blockade and the treatment of hypertension post-MCS implant include pharmacotherapy with heart failure medications for blood pressure management.
Echocardiography is an integral part of determining the revolutions per minute of continuous-flow pumps. Common goals include adequate LV unloading while maintaining the LV septum in the midline and minimizing mitral regurgitation. Postoperatively, the revolutions per minute of continuous-flow pumps should be set low enough to allow for intermittent aortic valve opening which may reduce the risk of aortic valve fusion and the risk of late aortic valve insufficiency.
Anticoagulation and antiplatelet therapy initiated post operatively in the ICU setting should be continued with the aim of achieving device-specific recommended INR for warfarin and desired antiplatelet effects, according to the task force.
Bleeding in the early postoperative period during the index hospitalization should be urgently evaluated with lowering, discontinuation, and/or reversal of anticoagulation and antiplatelet medications.
To reduce risk for infection after MCS placement, the driveline should be stabilized immediately after the device is placed and throughout the duration of support. A dressing change protocol should be immediately initiated post operatively. Secondary antibiotic prophylaxis for prevention of endocarditis has not been studied in the MCS population but would be considered reasonable due to the risk of bacteremia in this group.
Recommendations for optimal nutritional status include consultation with nutritional services at the time of implantation with ongoing follow-up post operatively to ensure nutrition goals are being met. For those unable to meet nutritional goals orally, feeding should be started early and preferably through an enteral feeding tube. Parenteral nutrition should only be started if enteral nutrition is not possible and under the guidance of nutritional consultation.
Prealbumin and C-reactive protein levels can be monitored weekly to track the nutritional status of the postoperative patient. As nutrition improves, prealbumin should rise and C-reactive protein should decrease.
Patient and caregiver education should be initiated shortly after surgery and reinforced by the nursing staff. Educational strategies should use written, verbal, and practical methods.
MCS parameters should be written in the medical record at regular intervals with established criteria for parameters that require physician notification.
The patient and family members should be taught to track their device parameters and alert staff when changes are observed. Changes in parameters outside of normal ranges should be thoroughly evaluated and treated appropriately.
Routine support should be available from social workers, psychologists, or psychiatrists as patients and families adjust to life changes after MCS. Routine surveillance for psychiatric symptoms should be performed. If symptoms develop, consultation with specialists (including social work, psychology, and/or psychiatry) for diagnosis, treatment, and follow-up is recommended.
Assessment of specific problems that are related to domains of health-related quality of life (depression, anxiety, or pain) based on symptoms should help guide an action plan for these patients.
Successful discharge should include patient, caregiver and community provider education with written discharge instructions and preemptive home preparation regarding the safe management of the device and the MCS patient.
Recommendations for management of anticoagulation and antiplatelet therapy for patients who present with gastrointestinal bleeding include holding back anticoagulation and antiplatelet therapy in the setting of clinically significant bleeding and reversing anticoagulation in the setting of an elevated INR and clinically significant bleeding.
Anticoagulation and antiplatelet therapy should continue to be held until clinically significant bleeding resolves in the absence of evidence of pump dysfunction. The patient, device parameters, and the pump housing (if applicable) should be carefully monitored while anticoagulation and antiplatelet therapy is being withheld or the dose reduced.
Patients who present with a first episode of gastrointestinal bleeding should be managed in consultation with gastroenterology. Patients should at least have a colonoscopy and/or upper endoscopic evaluation. If the results are negative, evaluation of the small bowel, particularly in those with continuous-flow devices, should be considered.
In patients with persistent bleeding and a negative endoscopic evaluation, a tagged red blood scan or angiography should be considered.
Once the gastrointestinal bleeding has resolved, anticoagulation and antiplatelet therapy can be reintroduced with careful monitoring.
For patients with recurrent episodes of gastrointestinal bleeding, repeated endoscopic evaluation should take place in conjunction with gastroenterology consultation. When no source of bleeding can be detected or the source is not amenable to therapy, the type and intensity or even the use of antiplatelet therapy should be reevaluated in the context of the bleeding severity and pump type. Further, the goal INR or even the continued use of warfarin should be reevaluated in the context of the bleeding severity and pump type.
The patient and device parameters should be carefully monitored when anticoagulation and antiplatelet therapy have been reduced or discontinued because of recurrent gastrointestinal bleeding.
Reducing the pump speed for continuous-flow pumps in the setting of recurrent gastrointestinal bleeding due to arteriovenous malformations may be considered.
When patients present with a new neurologic deficit, assess the current INR and review the recent INR. Prompt consultation with neurology is recommended, as is CT and angiography of the head and neck.
Review the pump parameters for signs of device thrombosis or malfunction, and inspect the pump housing for clots in extracorporeal pumps. Discontinuation or reversal of anticoagulation in the setting of hemorrhagic stroke is recommended.
Assessing for the source of the thrombus should be considered in embolic stroke patients. Selective use of an interventional radiologic approach to thrombotic strokes or selective use of thrombolytic agents in the setting of thrombotic stroke without CT scan evidence of hemorrhage may be considered.
Chronic management of patients after presentation with a new neurologic deficit may include formal stroke rehabilitation in consultation with neurology, close monitoring of anticoagulation in the setting of an embolic event to ensure adequate levels of anticoagulation, long-term control of blood pressure, administration of the National Institutes of Health (NIH) stroke scale at 30 and 60 days, and resumption of anticoagulation in consultation with neurology or neurosurgery in the setting of hemorrhagic stroke.
Recommendations for assessment of neurocognitive deficits include routine neurocognitive assessment at 3, 6, 12, and 18 months after implant.
Recommendations for evaluation of MCS patients with a suspected infection include a complete blood count, chest radiographic imaging, and blood cultures.
At least three sets of blood cultures over 24 hours should be drawn, with at least one culture from any indwelling central venous catheters.
For those with a suspected cannula or driveline infection, obtaining a sample for Gram stain, KOH, and routine bacterial and fungal cultures is recommended.
When clinically indicated, aspirate from other potential sources, as dictated by presenting symptoms and examination.
Directed radiographic studies based on presenting symptoms and examination are recommended.
MCS patients with incessant ventricular arrhythmias require prompt admission for further management because hemodynamic compromise may occur. Patients with ongoing VT refractory to medical therapy may require catheter ablation, which should be performed by an electrophysiologist with expertise in treating patients with MCS.
When an MCS patient is undergoing a noncardiac procedure, the MCS and surgical teams need to collaborate. For nonemergency procedures, warfarin and antiplatelet therapy may be continued if the risk of bleeding associated with the procedure is low. If therapy needs to be stopped, warfarin and antiplatelet therapy should be held for an appropriate period of time as determined by the type of procedure being undertaken and risk of bleeding. Bridging with heparin or a heparin alternative while a patient is off warfarin may be considered.
For emergency procedures, warfarin may need to be rapidly reversed with fresh frozen plasma or prothrombin protein concentrate. Vitamin K can be administered with caution, but has slower onset of action. Post procedure, warfarin and antiplatelet therapy may be resumed when risk of surgical bleeding is deemed acceptable. Patients may be bridged with heparin or a heparin alternative while waiting for the INR to reach the target range.
Dr. Pamboukian and Dr. Feldman agreed that the newly released guidelines will likely be in place for only a couple of years before a revision comes out, testament to the rapid changes in this field. Dr. Feldman cited new VADs from at least two manufacturers expected to enter first-in-man studies this year, and the continued snowballing of VAD implantation rates. The most recent 2012 numbers (through Sept. 30, 2012) from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) showed nearly 2,000 VADS getting implanted into U.S. patients last year, the highest annual rate ever.
"Because the field is growing, a lot of new centers want to establish programs. We want this treatment to reach as many appropriate patients as possible, but we want it to grow responsibly. These guidelines help establish the best practices, and help ensure that patients get the best care wherever they go," Dr. Pamboukian said.
Dr. Pamboukian said that she had no disclosures. Dr. Feldman said that he has received research support from Terumo.
FROM THE JOURNAL OF HEART AND LUNG TRANSPLANTATION
PFOs raise stroke risk from ICDs, pacemakers
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be perfect candidates for targeted closure of their heart defect, based on a review of more than 6,000 patients who received a device at one U.S. center.
During an average follow-up of nearly 5 years, patients with a patent foramen ovale (PFO) who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop a stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure.
"If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-99). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective and potentially subject to unadjusted confounding.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said.
In contrast to the strong, consistent link between PFOs and stroke or TIA, the researchers saw no significant link between the presence of a PFO and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves then continued to diverge more and more over time. He hypothesized that micro-emboli that originate on the device leads pass through the PFO and into the pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
Also, the stroke risk in this analysis may have underestimated the true risk posed by the combination of a PFO and a heart device because the methods used to detect PFOs and stroke may not have been optimal in many cases.
Dr. Desimone said that he had no disclosures.
On Twitter @mitchelzoler
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be perfect candidates for targeted closure of their heart defect, based on a review of more than 6,000 patients who received a device at one U.S. center.
During an average follow-up of nearly 5 years, patients with a patent foramen ovale (PFO) who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop a stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure.
"If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-99). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective and potentially subject to unadjusted confounding.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said.
In contrast to the strong, consistent link between PFOs and stroke or TIA, the researchers saw no significant link between the presence of a PFO and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves then continued to diverge more and more over time. He hypothesized that micro-emboli that originate on the device leads pass through the PFO and into the pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
Also, the stroke risk in this analysis may have underestimated the true risk posed by the combination of a PFO and a heart device because the methods used to detect PFOs and stroke may not have been optimal in many cases.
Dr. Desimone said that he had no disclosures.
On Twitter @mitchelzoler
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be perfect candidates for targeted closure of their heart defect, based on a review of more than 6,000 patients who received a device at one U.S. center.
During an average follow-up of nearly 5 years, patients with a patent foramen ovale (PFO) who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop a stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure.
"If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-99). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective and potentially subject to unadjusted confounding.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said.
In contrast to the strong, consistent link between PFOs and stroke or TIA, the researchers saw no significant link between the presence of a PFO and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves then continued to diverge more and more over time. He hypothesized that micro-emboli that originate on the device leads pass through the PFO and into the pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
Also, the stroke risk in this analysis may have underestimated the true risk posed by the combination of a PFO and a heart device because the methods used to detect PFOs and stroke may not have been optimal in many cases.
Dr. Desimone said that he had no disclosures.
On Twitter @mitchelzoler
AT THE ANNUAL SCIENTIFIC SESSIONS OF THE AMERICAN HEART ASSOCIATION
Major Finding: ICD or pacemaker recipients with a PFO had a 4.6-fold increased risk of stroke or TIA.
Data Source: A review of 6,086 patients at the Mayo Clinic.
Disclosures: Dr. DeSimone said that he had no disclosures.
Staged surgery found unnecessary for colorectal tumors
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: Removal of a colorectal tumor and synchronous liver metastases in a single operation or in two staged surgeries had equal outcomes.
Data Source: A review of 1,004 patients who underwent surgery to excise a primary colorectal tumor and synchronous liver metastases at four major hepatobiliary centers.
Disclosures: Dr. Pawlik said he had no relevant financial disclosures.
Computer program refines postsurgical insulin treatment
PALM BEACH, FLA. – A commercially available computer program designed to calculate optimal insulin dosages for postsurgical ICU patients halved the rate of hyperglycemic episodes that patients experienced while dropping the rate of hypoglycemic events by 95% in a single-center, U.S. experience with a total of more than 2,000 patients.
The computer program also resulted in a dramatic reduction in the number of blood glucose measures the nursing staff performed, cutting testing down from 7,495 blood glucose measures/month before the system began to 4,072 blood measures/month once it was in place, Dr. Christopher C. Baker, FACS, said at the annual meeting of the Southern Surgical Association.
An additional associated benefit was a substantial reduction in hospital-acquired infections in the postsurgery patients once computer-guided insulin dosing came online, but this was only a temporal association that may have also been driven by other improvements in patient management that happened at about the same time, said Dr. Baker, chairman of surgery at the Carilion Clinic in Roanoke, Va.
"People have shied away from glucose controls systems [like the one introduced at the Carilion Clinic] because the results from the NICE-SUGAR [Normoglycemia in Intensive Care Evaluation–Survival Using Glucose Algorithm Regulation] trial (N. Engl. J. Med. 2009;360:1283-97) showed that hypoglycemia episodes increase when you do this," commented Dr. David Herndon, FACS, professor of surgery at the University of Texas Medical branch in Galveston. "What is important in the Carilion results is that the computerized support system reduced hypoglycemia. If that can be done across the board, they might have a winner. We need to better control sugar in the ICU."
The next step is to prove the efficacy of this computer program in a prospective, randomized controlled trial, said Dr. Herndon, a suggestion that Dr. Baker also endorsed.
"The reduced incidence of hypoglycemia was incredibly impressive," commented Dr. Michael Rotondo, FACS, professor and chairman of surgery at East Carolina University in Greenville, N.C., who also called for results from a prospective, controlled study.
Surgeons at Carilion introduced the computerized support program for insulin dosing in early 2010 into five surgical intensive care departments: surgical ICU, trauma ICU, neurotrauma ICU, cardiac surgery ICU, and the cardiac surgery progressive care unit. Dr. Baker and his associates compared data collected on patients from all five units with the computerized system in place during July 2010-December 2011, a total of 1,682 patients treated using the computer program, with 449 patients treated in the five units during July 2009-December 2009, before use of the computer program started.
When using the program, nurses take an initial blood specimen from a patient newly arrived at the unit and enter the blood glucose level into the program along with the patient’s weight and blood creatinine level. The program then immediately calculates the appropriate bolus insulin dose, the insulin infusion rate, and the time to the next blood draw, said Dr. Sandy L. Fogel, FACS, a surgeon at Carilion who collaborated on the study. At first, the next blood draw is specified for about 15-30 minutes following the first, but subsequently the blood draws are directed to occur at longer and longer intervals as the patient’s blood glucose comes under control, within the target range of 70-150 mg/dL. Eventually, draws occur at 6-8 hour intervals, Dr. Fogel said.
During the 6-month historical control, nurses drew 44,972 blood specimens for glucose measurement, an average of 7,495/month, compared with 73,290 blood draws during the 18-month period with the program in place, an average of 4,072 blood specimens drawn/month.
During the 18 months with the program in use, hyperglycemic episodes, defined as a blood glucose level greater than 150 mg/dL, dropped by 45%-57% across the five units using the system compared with each unit’s historical control. Overall, hyperglycemic events fell by 50%, a statistically significant difference.
The incidence of hypoglycemic episodes, defined as a blood glucose level below 40 mg/dL, fell from a 1% rate during the historical control period, to rates that ranged from zero to 0.12% with the program in place, with an overall rate across all five units of 0.05%, a 95% relative decrease that was statistically significant.
The substantial decline in hypoglycemic episodes "was a surprise for us," Dr. Fogel said. In addition, out of all the blood measures performed using the computer program 17% had glucose levels of 60-69 mg/dL, and 3% were below 60 mg/dL. In short, the computer program "did not increase hypoglycemic episodes by any way you measure it," Dr. Fogel said.
The insulin-dosing program used at Carilion is the EndoTool, marketed by Hospira, Inc.
Dr. Baker, Dr. Herndon, Dr. Rotondo, and Dr. Fogel had no disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – A commercially available computer program designed to calculate optimal insulin dosages for postsurgical ICU patients halved the rate of hyperglycemic episodes that patients experienced while dropping the rate of hypoglycemic events by 95% in a single-center, U.S. experience with a total of more than 2,000 patients.
The computer program also resulted in a dramatic reduction in the number of blood glucose measures the nursing staff performed, cutting testing down from 7,495 blood glucose measures/month before the system began to 4,072 blood measures/month once it was in place, Dr. Christopher C. Baker, FACS, said at the annual meeting of the Southern Surgical Association.
An additional associated benefit was a substantial reduction in hospital-acquired infections in the postsurgery patients once computer-guided insulin dosing came online, but this was only a temporal association that may have also been driven by other improvements in patient management that happened at about the same time, said Dr. Baker, chairman of surgery at the Carilion Clinic in Roanoke, Va.
"People have shied away from glucose controls systems [like the one introduced at the Carilion Clinic] because the results from the NICE-SUGAR [Normoglycemia in Intensive Care Evaluation–Survival Using Glucose Algorithm Regulation] trial (N. Engl. J. Med. 2009;360:1283-97) showed that hypoglycemia episodes increase when you do this," commented Dr. David Herndon, FACS, professor of surgery at the University of Texas Medical branch in Galveston. "What is important in the Carilion results is that the computerized support system reduced hypoglycemia. If that can be done across the board, they might have a winner. We need to better control sugar in the ICU."
The next step is to prove the efficacy of this computer program in a prospective, randomized controlled trial, said Dr. Herndon, a suggestion that Dr. Baker also endorsed.
"The reduced incidence of hypoglycemia was incredibly impressive," commented Dr. Michael Rotondo, FACS, professor and chairman of surgery at East Carolina University in Greenville, N.C., who also called for results from a prospective, controlled study.
Surgeons at Carilion introduced the computerized support program for insulin dosing in early 2010 into five surgical intensive care departments: surgical ICU, trauma ICU, neurotrauma ICU, cardiac surgery ICU, and the cardiac surgery progressive care unit. Dr. Baker and his associates compared data collected on patients from all five units with the computerized system in place during July 2010-December 2011, a total of 1,682 patients treated using the computer program, with 449 patients treated in the five units during July 2009-December 2009, before use of the computer program started.
When using the program, nurses take an initial blood specimen from a patient newly arrived at the unit and enter the blood glucose level into the program along with the patient’s weight and blood creatinine level. The program then immediately calculates the appropriate bolus insulin dose, the insulin infusion rate, and the time to the next blood draw, said Dr. Sandy L. Fogel, FACS, a surgeon at Carilion who collaborated on the study. At first, the next blood draw is specified for about 15-30 minutes following the first, but subsequently the blood draws are directed to occur at longer and longer intervals as the patient’s blood glucose comes under control, within the target range of 70-150 mg/dL. Eventually, draws occur at 6-8 hour intervals, Dr. Fogel said.
During the 6-month historical control, nurses drew 44,972 blood specimens for glucose measurement, an average of 7,495/month, compared with 73,290 blood draws during the 18-month period with the program in place, an average of 4,072 blood specimens drawn/month.
During the 18 months with the program in use, hyperglycemic episodes, defined as a blood glucose level greater than 150 mg/dL, dropped by 45%-57% across the five units using the system compared with each unit’s historical control. Overall, hyperglycemic events fell by 50%, a statistically significant difference.
The incidence of hypoglycemic episodes, defined as a blood glucose level below 40 mg/dL, fell from a 1% rate during the historical control period, to rates that ranged from zero to 0.12% with the program in place, with an overall rate across all five units of 0.05%, a 95% relative decrease that was statistically significant.
The substantial decline in hypoglycemic episodes "was a surprise for us," Dr. Fogel said. In addition, out of all the blood measures performed using the computer program 17% had glucose levels of 60-69 mg/dL, and 3% were below 60 mg/dL. In short, the computer program "did not increase hypoglycemic episodes by any way you measure it," Dr. Fogel said.
The insulin-dosing program used at Carilion is the EndoTool, marketed by Hospira, Inc.
Dr. Baker, Dr. Herndon, Dr. Rotondo, and Dr. Fogel had no disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – A commercially available computer program designed to calculate optimal insulin dosages for postsurgical ICU patients halved the rate of hyperglycemic episodes that patients experienced while dropping the rate of hypoglycemic events by 95% in a single-center, U.S. experience with a total of more than 2,000 patients.
The computer program also resulted in a dramatic reduction in the number of blood glucose measures the nursing staff performed, cutting testing down from 7,495 blood glucose measures/month before the system began to 4,072 blood measures/month once it was in place, Dr. Christopher C. Baker, FACS, said at the annual meeting of the Southern Surgical Association.
An additional associated benefit was a substantial reduction in hospital-acquired infections in the postsurgery patients once computer-guided insulin dosing came online, but this was only a temporal association that may have also been driven by other improvements in patient management that happened at about the same time, said Dr. Baker, chairman of surgery at the Carilion Clinic in Roanoke, Va.
"People have shied away from glucose controls systems [like the one introduced at the Carilion Clinic] because the results from the NICE-SUGAR [Normoglycemia in Intensive Care Evaluation–Survival Using Glucose Algorithm Regulation] trial (N. Engl. J. Med. 2009;360:1283-97) showed that hypoglycemia episodes increase when you do this," commented Dr. David Herndon, FACS, professor of surgery at the University of Texas Medical branch in Galveston. "What is important in the Carilion results is that the computerized support system reduced hypoglycemia. If that can be done across the board, they might have a winner. We need to better control sugar in the ICU."
The next step is to prove the efficacy of this computer program in a prospective, randomized controlled trial, said Dr. Herndon, a suggestion that Dr. Baker also endorsed.
"The reduced incidence of hypoglycemia was incredibly impressive," commented Dr. Michael Rotondo, FACS, professor and chairman of surgery at East Carolina University in Greenville, N.C., who also called for results from a prospective, controlled study.
Surgeons at Carilion introduced the computerized support program for insulin dosing in early 2010 into five surgical intensive care departments: surgical ICU, trauma ICU, neurotrauma ICU, cardiac surgery ICU, and the cardiac surgery progressive care unit. Dr. Baker and his associates compared data collected on patients from all five units with the computerized system in place during July 2010-December 2011, a total of 1,682 patients treated using the computer program, with 449 patients treated in the five units during July 2009-December 2009, before use of the computer program started.
When using the program, nurses take an initial blood specimen from a patient newly arrived at the unit and enter the blood glucose level into the program along with the patient’s weight and blood creatinine level. The program then immediately calculates the appropriate bolus insulin dose, the insulin infusion rate, and the time to the next blood draw, said Dr. Sandy L. Fogel, FACS, a surgeon at Carilion who collaborated on the study. At first, the next blood draw is specified for about 15-30 minutes following the first, but subsequently the blood draws are directed to occur at longer and longer intervals as the patient’s blood glucose comes under control, within the target range of 70-150 mg/dL. Eventually, draws occur at 6-8 hour intervals, Dr. Fogel said.
During the 6-month historical control, nurses drew 44,972 blood specimens for glucose measurement, an average of 7,495/month, compared with 73,290 blood draws during the 18-month period with the program in place, an average of 4,072 blood specimens drawn/month.
During the 18 months with the program in use, hyperglycemic episodes, defined as a blood glucose level greater than 150 mg/dL, dropped by 45%-57% across the five units using the system compared with each unit’s historical control. Overall, hyperglycemic events fell by 50%, a statistically significant difference.
The incidence of hypoglycemic episodes, defined as a blood glucose level below 40 mg/dL, fell from a 1% rate during the historical control period, to rates that ranged from zero to 0.12% with the program in place, with an overall rate across all five units of 0.05%, a 95% relative decrease that was statistically significant.
The substantial decline in hypoglycemic episodes "was a surprise for us," Dr. Fogel said. In addition, out of all the blood measures performed using the computer program 17% had glucose levels of 60-69 mg/dL, and 3% were below 60 mg/dL. In short, the computer program "did not increase hypoglycemic episodes by any way you measure it," Dr. Fogel said.
The insulin-dosing program used at Carilion is the EndoTool, marketed by Hospira, Inc.
Dr. Baker, Dr. Herndon, Dr. Rotondo, and Dr. Fogel had no disclosures.
On Twitter @mitchelzoler
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: A computer program for calculating insulin doses for postsurgical patients cut hyperglycemic episodes by 50%, and hypoglycemic episodes by 95%, compared with historic controls.
Data Source: A study of 1,682 postsurgical patients treated in five intensive care units at one U.S. center and 449 historical controls from the same units.
Disclosures: Dr. Baker, Dr. Herndon, Dr. Rotondo, and Dr. Fogel had no disclosures.