User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
CoreValve holds size advantage for U.S. TAVR
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from CoreValve’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic* is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma* said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.
On Twitter @mitchelzoler
*CORRECTION, 1/30/2014: In an earlier version of this article, the appelant was misidentified.
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from CoreValve’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic* is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma* said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.
On Twitter @mitchelzoler
*CORRECTION, 1/30/2014: In an earlier version of this article, the appelant was misidentified.
When the Food and Drug Administration in January granted marketing approval to a second transcatheter aortic valve replacement system for inoperable patients with aortic stenosis, the CoreValve marketed by Medtronic, the new valve conceded a greater than 2-year head start to the first system on the U.S. market, Sapien marketed by Edwards.
But cardiologists see that 2-year edge in familiarity eclipsed for at least some patients by two major advantages that CoreValve currently holds over Sapien: delivery via a significantly thinner sheath, and the option of larger-diameter valves that allow replacement in patients with a wider aortic annulus.
The CoreValve delivery sheath is 18 French, compared with a 22F or 24F size for the Sapien transcatheter aortic valve replacement (TAVR) with U.S. approval, and the Sapien valves come in diameters of 23 and 26 mm, compared with options of 23, 26, 29, and 31 mm for the CoreValve.
"CoreValve is the device of choice for patients with smaller vessel sizes. Sapien has been a wonderful device to use, and we have so much experience with it, but the smaller CoreValve size will allow many more patients to be done with a transfemoral approach," said Dr. Peter C. Block, a professor of medicine at Emory University in Atlanta and an interventional cardiologist who performs TAVR.
"More patients will qualify for TAVR and will be treated transfemorally with the larger valve diameters and smaller sheath size," agreed Dr. Mauricio G. Cohen, director of cardiac catheterization at the University of Miami and TAVR interventionalist. Another potential positive of having CoreValve on the U.S. market is that competition between the two options will likely drive down the cost of a TAVR system, which until now has run more than $30,000, Dr. Cohen said in an interview.
CoreValve received FDA approval less than 3 months after researchers first reported data from CoreValve’s U.S. Pivotal Trial Extreme Risk Iliofemoral Study last October at the TCT (Transcatheter Cardiovascular Therapeutics) annual meeting. In that study, 471 inoperable aortic stenosis patients had a 26% 1-year rate of death or major stroke, substantially surpassing the 43% rate that the study set up as the target for superiority, reported Dr. Jeffrey J. Popma, lead investigator on the study.
Dr. Popma warned against comparing CoreValve’s efficacy and safety performance in the trial and the Sapien system’s performance in its pivotal trial in inoperable patients, the PARTNER cohort B trial (N. Engl. J. Med. 2010;363:1597-607). "It’s very difficult to make cross-trial comparison," he said in an interview, a limitation also noted by Dr. Cohen and Dr. Block. But Dr. Popma highlighted the 2.4% 30-day stroke rate in the pivotal trial, and a 1.8% 30-day stroke rate seen with CoreValve in inoperable patients in a continued access program at the trial’s study sites. He also highlighted the 11% rate of moderate paravalvular aortic regurgitation after 30 days that dropped to a 4% rate after 1 year.
Perhaps the biggest downside of CoreValve’s performance in the pivotal trial was that 22% of patients required a permanent pacemaker implant within the first 30 days, increasing to 27% of patients with 1-year follow-up. Increased risk for a pacemaker is an inherent downside of CoreValve because of its longer size compared with the Sapien valve and how the CoreValve sits in the aortic annulus. The CoreValve is designed for supravalvular placement and anchoring in the left ventricular outflow tract near the left bundle branch that can result in mechanical irritation and arrhythmia with the need for pacing, explained Dr. Popma, professor of medicine at Harvard University and an interventional cardiologist at Beth Israel Deaconess Medical Center, Boston.
"I think our pacemaker rate was very acceptable. I don’t think it will ever be as low as with Sapien, but it’s a worthwhile trade-off because the CoreValve functions well and results in a low rate of paravalvular regurgitation," he said.
Dr. Popma also stressed that 1-year mortality was no greater among the patients who required a pacemaker implant in the pivotal trial. A subgroup analysis of results from the trial to try to identify which patients had the greatest risk for needing a pacemaker after a CoreValve implant has not yet finished, he said. It’s possible that certain patients with preexisting conduction abnormalities, such as a right bundle branch block coupled with a left anterior fascicular block, have the greatest vulnerability.
Patients for whom the Sapien system remains ideal are those with a narrow sinus of Valsalva, because the longer CoreValve frame crosses the sinus and may compromise coronary blood flow in patients with a narrow sinus, Dr. Popma said.
The choice between CoreValve and Sapien systems will grow even more complicated for U.S. cardiologists and surgeons when the Sapien XT valve system receives FDA marketing approval, likely later this year. The Sapien XT delivery sheath matches the 18F size of CoreValve and will also come in a 29-mm size, blunting two of CoreValves main advantages.
Medtronic also faces charges of patent infringement by its CoreValve in a court case initiated by Edwards. In mid-January, a jury in a U.S. District Court assessed a penalty of $394 million against Medtronic. Edwards is also seeking a court-ordered halt to U.S. marketing of CoreValve. But Medtronic* is appealing the jury verdict and continues to fight the injunction, and a company spokesperson said in an interview that the legal maneuverings will likely take at least another year to fully resolve. In the meantime, Medtronic began U.S. distribution of the CoreValve on Jan. 17.
Dr. Block said that his institution received a research grant to participate in Sapien trials. Dr. Cohen said that he has been a consultant to Medtronic and Edwards. Dr. Popma* said that his institution received research support from Medtronic and that he has been a consultant to and received research support from Boston Scientific.
On Twitter @mitchelzoler
*CORRECTION, 1/30/2014: In an earlier version of this article, the appelant was misidentified.
Former JNC 8 hypertension panel issues minority report
The controversial hypertension-management guidelines released by the former JNC 8 panel in December was not the group’s last word. There was a minority report, too, published almost a month later in mid-January.
Five of the 17 members of the group originally assembled in 2008 by the National Heart, Lung, and Blood Institute (NHLBI) to research and write the eighth edition of the official U.S. hypertension guidelines not only disagreed with their 12 colleagues about resetting the systolic blood pressure target to 150 mm Hg from the prior target of 140 mm Hg for people aged 60-79 without diabetes or chronic kidney disease, but also felt strongly enough to write an article about it.
A published minority report is not how most medical guideline writing panels usually work.
The history of the Eighth Joint National Committee (JNC 8) already featured some unusual twists. After many months of unexpected delays leading up to the report’s release, the NHLBI announced last June that it would hand off the JNC 8 process to an appropriate medical group. A few weeks later, the institute announced that JNC 8 would get published under the auspices of the American Heart Association and American College of Cardiology. Then came word that the arrangement had fallen through, leaving what became the former JNC 8 panel to release its data analysis and recommendations without endorsement from a medical association.
The fact that the 150–mm Hg systolic target for treating 60- to 79-year-olds was controversial among the panel members themselves was no surprise. The former JNC 8 panel’s majority report in December acknowledged their lack of consensus on this issue. What had been previously unknown was exactly how the panel split, and the minority view that led to the disagreement.
The five dissenters were Dr. Jackson T. Wright Jr., Case Medical Center, Cleveland; Dr. Lawrence J. Fine, NHLBI, Bethesda, Md.; Daniel T. Lackland, Ph.D., Medical University of South Carolina, Charleston; Dr. Gbenga Ogedegbe, New York University; and Cheryl R. Dennison Himmelfarb, Ph.D., Johns Hopkins University, Baltimore.
Summarizing their view in their report, the five said they "believed that evidence was insufficient to increase the SBP goal from its current level of less than 140 mm Hg because of concern that increasing the goal may cause harm by increasing the risk for CVD and partially undoing the remarkable progress in reducing cardiovascular mortality in Americans older than 60 years. Because of the overall evidence, including the RCT data reviewed by the panel, and the decrease in CVD mortality, we concluded that the evidence for increasing a blood pressure target in high-risk populations should be at least as strong as the evidence required to decrease the recommended blood pressure target. In addition, one target would simplify implementation for clinicians."
The unusual circumstances that surrounded release of the former JNC 8 panel’s report and the controversial systolic target they set initially raised questions about the impact the recommendations would have on U.S. practice. The minority report puts an asterisk on the majority report and dilutes its influence even more.
On Twitter @mitchelzoler
The controversial hypertension-management guidelines released by the former JNC 8 panel in December was not the group’s last word. There was a minority report, too, published almost a month later in mid-January.
Five of the 17 members of the group originally assembled in 2008 by the National Heart, Lung, and Blood Institute (NHLBI) to research and write the eighth edition of the official U.S. hypertension guidelines not only disagreed with their 12 colleagues about resetting the systolic blood pressure target to 150 mm Hg from the prior target of 140 mm Hg for people aged 60-79 without diabetes or chronic kidney disease, but also felt strongly enough to write an article about it.
A published minority report is not how most medical guideline writing panels usually work.
The history of the Eighth Joint National Committee (JNC 8) already featured some unusual twists. After many months of unexpected delays leading up to the report’s release, the NHLBI announced last June that it would hand off the JNC 8 process to an appropriate medical group. A few weeks later, the institute announced that JNC 8 would get published under the auspices of the American Heart Association and American College of Cardiology. Then came word that the arrangement had fallen through, leaving what became the former JNC 8 panel to release its data analysis and recommendations without endorsement from a medical association.
The fact that the 150–mm Hg systolic target for treating 60- to 79-year-olds was controversial among the panel members themselves was no surprise. The former JNC 8 panel’s majority report in December acknowledged their lack of consensus on this issue. What had been previously unknown was exactly how the panel split, and the minority view that led to the disagreement.
The five dissenters were Dr. Jackson T. Wright Jr., Case Medical Center, Cleveland; Dr. Lawrence J. Fine, NHLBI, Bethesda, Md.; Daniel T. Lackland, Ph.D., Medical University of South Carolina, Charleston; Dr. Gbenga Ogedegbe, New York University; and Cheryl R. Dennison Himmelfarb, Ph.D., Johns Hopkins University, Baltimore.
Summarizing their view in their report, the five said they "believed that evidence was insufficient to increase the SBP goal from its current level of less than 140 mm Hg because of concern that increasing the goal may cause harm by increasing the risk for CVD and partially undoing the remarkable progress in reducing cardiovascular mortality in Americans older than 60 years. Because of the overall evidence, including the RCT data reviewed by the panel, and the decrease in CVD mortality, we concluded that the evidence for increasing a blood pressure target in high-risk populations should be at least as strong as the evidence required to decrease the recommended blood pressure target. In addition, one target would simplify implementation for clinicians."
The unusual circumstances that surrounded release of the former JNC 8 panel’s report and the controversial systolic target they set initially raised questions about the impact the recommendations would have on U.S. practice. The minority report puts an asterisk on the majority report and dilutes its influence even more.
On Twitter @mitchelzoler
The controversial hypertension-management guidelines released by the former JNC 8 panel in December was not the group’s last word. There was a minority report, too, published almost a month later in mid-January.
Five of the 17 members of the group originally assembled in 2008 by the National Heart, Lung, and Blood Institute (NHLBI) to research and write the eighth edition of the official U.S. hypertension guidelines not only disagreed with their 12 colleagues about resetting the systolic blood pressure target to 150 mm Hg from the prior target of 140 mm Hg for people aged 60-79 without diabetes or chronic kidney disease, but also felt strongly enough to write an article about it.
A published minority report is not how most medical guideline writing panels usually work.
The history of the Eighth Joint National Committee (JNC 8) already featured some unusual twists. After many months of unexpected delays leading up to the report’s release, the NHLBI announced last June that it would hand off the JNC 8 process to an appropriate medical group. A few weeks later, the institute announced that JNC 8 would get published under the auspices of the American Heart Association and American College of Cardiology. Then came word that the arrangement had fallen through, leaving what became the former JNC 8 panel to release its data analysis and recommendations without endorsement from a medical association.
The fact that the 150–mm Hg systolic target for treating 60- to 79-year-olds was controversial among the panel members themselves was no surprise. The former JNC 8 panel’s majority report in December acknowledged their lack of consensus on this issue. What had been previously unknown was exactly how the panel split, and the minority view that led to the disagreement.
The five dissenters were Dr. Jackson T. Wright Jr., Case Medical Center, Cleveland; Dr. Lawrence J. Fine, NHLBI, Bethesda, Md.; Daniel T. Lackland, Ph.D., Medical University of South Carolina, Charleston; Dr. Gbenga Ogedegbe, New York University; and Cheryl R. Dennison Himmelfarb, Ph.D., Johns Hopkins University, Baltimore.
Summarizing their view in their report, the five said they "believed that evidence was insufficient to increase the SBP goal from its current level of less than 140 mm Hg because of concern that increasing the goal may cause harm by increasing the risk for CVD and partially undoing the remarkable progress in reducing cardiovascular mortality in Americans older than 60 years. Because of the overall evidence, including the RCT data reviewed by the panel, and the decrease in CVD mortality, we concluded that the evidence for increasing a blood pressure target in high-risk populations should be at least as strong as the evidence required to decrease the recommended blood pressure target. In addition, one target would simplify implementation for clinicians."
The unusual circumstances that surrounded release of the former JNC 8 panel’s report and the controversial systolic target they set initially raised questions about the impact the recommendations would have on U.S. practice. The minority report puts an asterisk on the majority report and dilutes its influence even more.
On Twitter @mitchelzoler
One health system cut diabetes events, with set patient measures and cash for physicians
An incentivized, system-based approach to improve care for patients with diabetes and other chronic diseases appeared to pay off after just 3 years by significantly cutting the incidence of adverse clinical events among 25,000 patients with diabetes managed in a single U.S. health care system.
The first 3 years, starting in 2006, of tracking physician performance in applying a nine-item diabetes-management "bundle" to patients in the Geisinger Health System in rural Pennsylvania resulted in improved patient care that prevented about 300 myocardial infarctions, 140 strokes, and 160 cases of retinopathy, according to Dr. Peter B. Berger.
While the results of this adjusted analysis that used a historic control were "fabulous," they were also a little "tough to believe," admitted Dr. Berger, chairman of cardiology at Geisinger in Danville, Pa. Tough to believe because the benefits appeared quickly for endpoints thought to take years or decades to develop, and because of likely confounding by effects not fully controlled by statistical adjustments.
"There is potential for confounding that is impossible to eliminate entirely," he said in an interview.
Nonetheless, the finding is likely real, said Dr. Frederick J. Bloom Jr., a family medicine physician and chief of the care continuum at Geisinger. "We are looking at the impact of improving multiple risk factors including blood pressure, hemoglobin A1c, and low-density lipoprotein [LDL] cholesterol. The literature supports getting some pretty rapid clinical improvements" when all risk factors significantly improve at the same time, Dr. Bloom said in an interview.
Geisinger employs more than 1,000 physicians, runs several hospitals and more than 40 clinics, and provides care to about 2.6 million patients spread through a large area of north-central Pennsylvania. Almost a decade ago Geisinger began to introduce patient management bundles, including bundles for diabetes, hypertension, coronary artery disease, and adult prevention. A high level of compliance with all four of these bundles by all the physicians in a Geisinger office gains for all physicians working in that office a bonus of $10,000 each per year.
The diabetes bundle contains nine elements (see chart). The hemoglobin A1c, blood pressure, and LDL cholesterol goals have changed over the past 9 years. Currently, the goal for hemoglobin A1c level may be set from less than 7% to less than 8% based on a patient’s age and a shared decision between the patient and physician; the blood pressure goal is less than 140/80 mm Hg; and the LDL cholesterol goal is either less than 100 mg/dL or whatever is achieved with a high-intensity statin regimen.
Geisinger scores its physicians by tallying the percent of patients with diabetes who meet all nine goals of the bundle. Before the program began, in early 2006, this happened for 2.4% of patients. By last August, all the goals had been met by 14.5% of Geisinger patients with diabetes.
"Although 14.5% may seem low, it’s the highest rate in Pennsylvania, and among the highest in the United States. It’s almost uniquely high," Dr. Berger said.
"By using an all-or-none metric, we’re on a different scale; 14.5% is a really good score" for meeting every goal of the diabetes bundle, Dr. Bloom said. Some individual Geisinger sites have had their full compliance rate rise above 20%, showing that more improvement is possible, he added. The biggest challenge is getting patients to meet their blood pressure, hemoglobin A1c, and LDL cholesterol goals.
But the real aim is not good numbers but better clinical outcomes, something that Dr. Berger’s data documented for the first time. The adjusted analysis showed that compared with a propensity score–matched control group drawn from a different health system during the same period, the first 3 years of running the diabetes bundle and offering physician incentives led to one less myocardial infarction for every 82 patients with diabetes managed at Geisinger, one less retinopathy for every 152 patients managed, and one less stroke for every 170 patients managed.
"One difficulty for health systems is to decide how much to invest in these kinds of initiatives and what will be the return on their investment," Dr. Bloom said. "Seeing that these systems of care can produce a quick return will have a profound impact on how people think about investing in these systems," he predicted.
From the provider’s perspective, a financial incentive like the one Geisinger offers "gets people’s attention, but what really sustains the effort is when physicians see patients do better and have better outcomes," he added. While all elements of Geisinger’s approach may not be generalizable to other health care settings, three basic features are: a team-based system of care, data-driven redesign, and incentives, Dr. Bloom said.
Dr. Berger and Dr. Bloom are Geisinger employees.
On Twitter @mitchelzoler
An incentivized, system-based approach to improve care for patients with diabetes and other chronic diseases appeared to pay off after just 3 years by significantly cutting the incidence of adverse clinical events among 25,000 patients with diabetes managed in a single U.S. health care system.
The first 3 years, starting in 2006, of tracking physician performance in applying a nine-item diabetes-management "bundle" to patients in the Geisinger Health System in rural Pennsylvania resulted in improved patient care that prevented about 300 myocardial infarctions, 140 strokes, and 160 cases of retinopathy, according to Dr. Peter B. Berger.
While the results of this adjusted analysis that used a historic control were "fabulous," they were also a little "tough to believe," admitted Dr. Berger, chairman of cardiology at Geisinger in Danville, Pa. Tough to believe because the benefits appeared quickly for endpoints thought to take years or decades to develop, and because of likely confounding by effects not fully controlled by statistical adjustments.
"There is potential for confounding that is impossible to eliminate entirely," he said in an interview.
Nonetheless, the finding is likely real, said Dr. Frederick J. Bloom Jr., a family medicine physician and chief of the care continuum at Geisinger. "We are looking at the impact of improving multiple risk factors including blood pressure, hemoglobin A1c, and low-density lipoprotein [LDL] cholesterol. The literature supports getting some pretty rapid clinical improvements" when all risk factors significantly improve at the same time, Dr. Bloom said in an interview.
Geisinger employs more than 1,000 physicians, runs several hospitals and more than 40 clinics, and provides care to about 2.6 million patients spread through a large area of north-central Pennsylvania. Almost a decade ago Geisinger began to introduce patient management bundles, including bundles for diabetes, hypertension, coronary artery disease, and adult prevention. A high level of compliance with all four of these bundles by all the physicians in a Geisinger office gains for all physicians working in that office a bonus of $10,000 each per year.
The diabetes bundle contains nine elements (see chart). The hemoglobin A1c, blood pressure, and LDL cholesterol goals have changed over the past 9 years. Currently, the goal for hemoglobin A1c level may be set from less than 7% to less than 8% based on a patient’s age and a shared decision between the patient and physician; the blood pressure goal is less than 140/80 mm Hg; and the LDL cholesterol goal is either less than 100 mg/dL or whatever is achieved with a high-intensity statin regimen.
Geisinger scores its physicians by tallying the percent of patients with diabetes who meet all nine goals of the bundle. Before the program began, in early 2006, this happened for 2.4% of patients. By last August, all the goals had been met by 14.5% of Geisinger patients with diabetes.
"Although 14.5% may seem low, it’s the highest rate in Pennsylvania, and among the highest in the United States. It’s almost uniquely high," Dr. Berger said.
"By using an all-or-none metric, we’re on a different scale; 14.5% is a really good score" for meeting every goal of the diabetes bundle, Dr. Bloom said. Some individual Geisinger sites have had their full compliance rate rise above 20%, showing that more improvement is possible, he added. The biggest challenge is getting patients to meet their blood pressure, hemoglobin A1c, and LDL cholesterol goals.
But the real aim is not good numbers but better clinical outcomes, something that Dr. Berger’s data documented for the first time. The adjusted analysis showed that compared with a propensity score–matched control group drawn from a different health system during the same period, the first 3 years of running the diabetes bundle and offering physician incentives led to one less myocardial infarction for every 82 patients with diabetes managed at Geisinger, one less retinopathy for every 152 patients managed, and one less stroke for every 170 patients managed.
"One difficulty for health systems is to decide how much to invest in these kinds of initiatives and what will be the return on their investment," Dr. Bloom said. "Seeing that these systems of care can produce a quick return will have a profound impact on how people think about investing in these systems," he predicted.
From the provider’s perspective, a financial incentive like the one Geisinger offers "gets people’s attention, but what really sustains the effort is when physicians see patients do better and have better outcomes," he added. While all elements of Geisinger’s approach may not be generalizable to other health care settings, three basic features are: a team-based system of care, data-driven redesign, and incentives, Dr. Bloom said.
Dr. Berger and Dr. Bloom are Geisinger employees.
On Twitter @mitchelzoler
An incentivized, system-based approach to improve care for patients with diabetes and other chronic diseases appeared to pay off after just 3 years by significantly cutting the incidence of adverse clinical events among 25,000 patients with diabetes managed in a single U.S. health care system.
The first 3 years, starting in 2006, of tracking physician performance in applying a nine-item diabetes-management "bundle" to patients in the Geisinger Health System in rural Pennsylvania resulted in improved patient care that prevented about 300 myocardial infarctions, 140 strokes, and 160 cases of retinopathy, according to Dr. Peter B. Berger.
While the results of this adjusted analysis that used a historic control were "fabulous," they were also a little "tough to believe," admitted Dr. Berger, chairman of cardiology at Geisinger in Danville, Pa. Tough to believe because the benefits appeared quickly for endpoints thought to take years or decades to develop, and because of likely confounding by effects not fully controlled by statistical adjustments.
"There is potential for confounding that is impossible to eliminate entirely," he said in an interview.
Nonetheless, the finding is likely real, said Dr. Frederick J. Bloom Jr., a family medicine physician and chief of the care continuum at Geisinger. "We are looking at the impact of improving multiple risk factors including blood pressure, hemoglobin A1c, and low-density lipoprotein [LDL] cholesterol. The literature supports getting some pretty rapid clinical improvements" when all risk factors significantly improve at the same time, Dr. Bloom said in an interview.
Geisinger employs more than 1,000 physicians, runs several hospitals and more than 40 clinics, and provides care to about 2.6 million patients spread through a large area of north-central Pennsylvania. Almost a decade ago Geisinger began to introduce patient management bundles, including bundles for diabetes, hypertension, coronary artery disease, and adult prevention. A high level of compliance with all four of these bundles by all the physicians in a Geisinger office gains for all physicians working in that office a bonus of $10,000 each per year.
The diabetes bundle contains nine elements (see chart). The hemoglobin A1c, blood pressure, and LDL cholesterol goals have changed over the past 9 years. Currently, the goal for hemoglobin A1c level may be set from less than 7% to less than 8% based on a patient’s age and a shared decision between the patient and physician; the blood pressure goal is less than 140/80 mm Hg; and the LDL cholesterol goal is either less than 100 mg/dL or whatever is achieved with a high-intensity statin regimen.
Geisinger scores its physicians by tallying the percent of patients with diabetes who meet all nine goals of the bundle. Before the program began, in early 2006, this happened for 2.4% of patients. By last August, all the goals had been met by 14.5% of Geisinger patients with diabetes.
"Although 14.5% may seem low, it’s the highest rate in Pennsylvania, and among the highest in the United States. It’s almost uniquely high," Dr. Berger said.
"By using an all-or-none metric, we’re on a different scale; 14.5% is a really good score" for meeting every goal of the diabetes bundle, Dr. Bloom said. Some individual Geisinger sites have had their full compliance rate rise above 20%, showing that more improvement is possible, he added. The biggest challenge is getting patients to meet their blood pressure, hemoglobin A1c, and LDL cholesterol goals.
But the real aim is not good numbers but better clinical outcomes, something that Dr. Berger’s data documented for the first time. The adjusted analysis showed that compared with a propensity score–matched control group drawn from a different health system during the same period, the first 3 years of running the diabetes bundle and offering physician incentives led to one less myocardial infarction for every 82 patients with diabetes managed at Geisinger, one less retinopathy for every 152 patients managed, and one less stroke for every 170 patients managed.
"One difficulty for health systems is to decide how much to invest in these kinds of initiatives and what will be the return on their investment," Dr. Bloom said. "Seeing that these systems of care can produce a quick return will have a profound impact on how people think about investing in these systems," he predicted.
From the provider’s perspective, a financial incentive like the one Geisinger offers "gets people’s attention, but what really sustains the effort is when physicians see patients do better and have better outcomes," he added. While all elements of Geisinger’s approach may not be generalizable to other health care settings, three basic features are: a team-based system of care, data-driven redesign, and incentives, Dr. Bloom said.
Dr. Berger and Dr. Bloom are Geisinger employees.
On Twitter @mitchelzoler
Major finding: Systematic management of patients with diabetes prevented about 500 episodes of myocardial infarction, stroke, or retinopathy during the first 3 years.
Data source: A review of 25,000 patients with diabetes managed at Geisinger Health System over 3 years compared with a propensity score–matched control group from another health system.
Disclosures: Dr. Berger and Dr. Bloom are Geisinger employees.
Microbiomes assessed as autoimmune triggers for RA
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
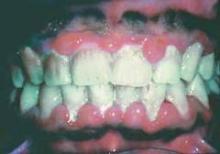
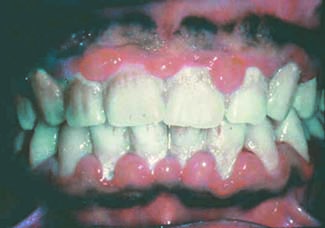
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler
PCSK9 antibody shows safety, efficacy to 1 year
DALLAS – Monthly treatment with a human antibody directed against the PCSK9 enzyme was safe and effective for substantially lowering low-density-lipoprotein cholesterol levels in 736 patients treated for 1 year in a controlled phase II study, the longest and largest series of patients treated with a PCSK9 antibody yet reported.
An injection of evolocumab (formerly known as Amgen 145) a human antibody to the proprotein convertase subtilisin/kexin type 9 (PCSK9) enzyme once every 4 weeks led to an average 52% reduction in blood levels of LDL cholesterol after 1 year on top of background treatment with standard-of-care medications, which in many cases also included maximal statin treatment, Dr. Michael J. Koren said at the American Heart Association scientific sessions.
In addition, the safety results for evolocumab "were very clean," he said, with no evidence that patients developed neutralizing antibody against the monoclonal antibody drug, and with injection-site reactions occurring in 5% of treated patients with one patient withdrawing because of the injection-site effect.
The Food and Drug Administration should "certainly vote to approve these drugs for familial hypercholesterolemia," said Dr. Koren, director of noninvasive cardiology at Jacksonville (Fla.) Memorial Hospital. "These are patients in whom we see a lot of complication, and we need lots of ways to lower their cholesterol."
PCSK9-inhibitor treatment of other patient populations, such as patients who are statin intolerant, will "probably need outcome studies for us to feel comfortable with widespread adoption," he added. Dr. Eckel estimated that about 4 million American patients who have been prescribed a statin can’t take the drug because of intolerance.
The Open-Label Study of Long-Term Evaluation Against LDL-C (OSLER) enrolled patients who had been enrolled in any of four prior phase II studies of evolocumab that each ran for 12 weeks. OSLER randomized 736 patients at 155 centers in 17 countries to receive 420 mg of evolocumab as a subcutaneous injection every 4 weeks in addition to standard-of-care treatment; 368 patients were assigned to standard of care only as controls. Patients averaged 56 years of age, their average LDL cholesterol level at baseline was about 140 mg/dL, and just under two-thirds of patients also received treatment with a statin.
After 1 year, 86% of the evolocumab-treated patients had an LDL cholesterol level below 100 mg/dL and 63% had their level below 70 mg/dL. In contrast, 16% and 1% respectively of patients on standard care reached these levels.
"We’re super pleased with the safety and tolerability data we’ve seen so far," Dr. Koren said. Even among the 98 patients treated with evolocumab who had their LDL cholesterol level below 25 mg/dL at 1year, the data showed no signal of increased serious adverse reactions or kidney or liver abnormalities. The only suggestion of a possible problem was a reported 9% incidence of headaches, compared with a 3% rate among patients on standard care and a 6% rate among patients on evolocumab for 1 year with LDL cholesterol levels of less than 50 mg/dL.
Concurrent with Dr. Koren’s report, the results also were published online (Circulation 2013;128 [doi:10.1161/CIRCULATIONAHA.113.007012]).
OSLER was sponsored by Amgen. Dr. Koren said that he has received research support from Amgen and from Regeneron, Sanofi, Roche, and Pfizer.
On Twitter @mitchelzoler
The OSLER results represent some progress for the PCSK9 antibodies. The results come from the longest PCSK9-antibody study so far reported. The adverse effects were not very noteworthy, and the 50% reduction in LDL cholesterol was sustained over 1 year. The adverse effects seen in patients whose LDL cholesterol level fell below 25 mg/dL did not look alarming. The results also showed an intriguing reduction in lipoprotein(a) of about 20% beyond what was achieved by standard care.
These results still leave many questions, such as the safety and efficacy of longer-term treatment. Having 1-year treatment results is nice, but it’s not like having 5-year or 20-year results. Another important issue is which patients are potential candidates for such treatment. Some people have estimated that as many as 4 million U.S. patients who need cholesterol-lowering treatment are statin intolerant, defined as patients who developed myopathy from monotherapy with two or more different statins. We also anxiously await the results from ongoing trials that are assessing the impact of PCSK9-antibody treatment on cardiovascular disease outcomes.
A trickier issue is whether a new agent can benefit patients on standard LDL-reducing regimens who have not reached their "goal" level. Now that the new cholesterol guidelines (Circulation 2013;128 [doi:10.1161/01.cir.0000437738.63853.7a]) eliminate target LDL levels for treatment, do patients still have goals? I believe the answer is yes.
Even though the guidelines concluded that no evidence exists for setting LDL-cholesterol goals, this does not means that goal-setting must be eliminated. Physicians practice medicine in the clinic, and setting a target goal for a patient’s LDL cholesterol level is a useful approach for treating patients with lipid disorders. Target goals may not be evidence based, but they can be part of effective practice.
Dr. Robert H. Eckel is professor of medicine at the University of Colorado, Aurora. He said that he had no disclosures. He made these comments as designated discussant for the report.
The OSLER results represent some progress for the PCSK9 antibodies. The results come from the longest PCSK9-antibody study so far reported. The adverse effects were not very noteworthy, and the 50% reduction in LDL cholesterol was sustained over 1 year. The adverse effects seen in patients whose LDL cholesterol level fell below 25 mg/dL did not look alarming. The results also showed an intriguing reduction in lipoprotein(a) of about 20% beyond what was achieved by standard care.
These results still leave many questions, such as the safety and efficacy of longer-term treatment. Having 1-year treatment results is nice, but it’s not like having 5-year or 20-year results. Another important issue is which patients are potential candidates for such treatment. Some people have estimated that as many as 4 million U.S. patients who need cholesterol-lowering treatment are statin intolerant, defined as patients who developed myopathy from monotherapy with two or more different statins. We also anxiously await the results from ongoing trials that are assessing the impact of PCSK9-antibody treatment on cardiovascular disease outcomes.
A trickier issue is whether a new agent can benefit patients on standard LDL-reducing regimens who have not reached their "goal" level. Now that the new cholesterol guidelines (Circulation 2013;128 [doi:10.1161/01.cir.0000437738.63853.7a]) eliminate target LDL levels for treatment, do patients still have goals? I believe the answer is yes.
Even though the guidelines concluded that no evidence exists for setting LDL-cholesterol goals, this does not means that goal-setting must be eliminated. Physicians practice medicine in the clinic, and setting a target goal for a patient’s LDL cholesterol level is a useful approach for treating patients with lipid disorders. Target goals may not be evidence based, but they can be part of effective practice.
Dr. Robert H. Eckel is professor of medicine at the University of Colorado, Aurora. He said that he had no disclosures. He made these comments as designated discussant for the report.
The OSLER results represent some progress for the PCSK9 antibodies. The results come from the longest PCSK9-antibody study so far reported. The adverse effects were not very noteworthy, and the 50% reduction in LDL cholesterol was sustained over 1 year. The adverse effects seen in patients whose LDL cholesterol level fell below 25 mg/dL did not look alarming. The results also showed an intriguing reduction in lipoprotein(a) of about 20% beyond what was achieved by standard care.
These results still leave many questions, such as the safety and efficacy of longer-term treatment. Having 1-year treatment results is nice, but it’s not like having 5-year or 20-year results. Another important issue is which patients are potential candidates for such treatment. Some people have estimated that as many as 4 million U.S. patients who need cholesterol-lowering treatment are statin intolerant, defined as patients who developed myopathy from monotherapy with two or more different statins. We also anxiously await the results from ongoing trials that are assessing the impact of PCSK9-antibody treatment on cardiovascular disease outcomes.
A trickier issue is whether a new agent can benefit patients on standard LDL-reducing regimens who have not reached their "goal" level. Now that the new cholesterol guidelines (Circulation 2013;128 [doi:10.1161/01.cir.0000437738.63853.7a]) eliminate target LDL levels for treatment, do patients still have goals? I believe the answer is yes.
Even though the guidelines concluded that no evidence exists for setting LDL-cholesterol goals, this does not means that goal-setting must be eliminated. Physicians practice medicine in the clinic, and setting a target goal for a patient’s LDL cholesterol level is a useful approach for treating patients with lipid disorders. Target goals may not be evidence based, but they can be part of effective practice.
Dr. Robert H. Eckel is professor of medicine at the University of Colorado, Aurora. He said that he had no disclosures. He made these comments as designated discussant for the report.
DALLAS – Monthly treatment with a human antibody directed against the PCSK9 enzyme was safe and effective for substantially lowering low-density-lipoprotein cholesterol levels in 736 patients treated for 1 year in a controlled phase II study, the longest and largest series of patients treated with a PCSK9 antibody yet reported.
An injection of evolocumab (formerly known as Amgen 145) a human antibody to the proprotein convertase subtilisin/kexin type 9 (PCSK9) enzyme once every 4 weeks led to an average 52% reduction in blood levels of LDL cholesterol after 1 year on top of background treatment with standard-of-care medications, which in many cases also included maximal statin treatment, Dr. Michael J. Koren said at the American Heart Association scientific sessions.
In addition, the safety results for evolocumab "were very clean," he said, with no evidence that patients developed neutralizing antibody against the monoclonal antibody drug, and with injection-site reactions occurring in 5% of treated patients with one patient withdrawing because of the injection-site effect.
The Food and Drug Administration should "certainly vote to approve these drugs for familial hypercholesterolemia," said Dr. Koren, director of noninvasive cardiology at Jacksonville (Fla.) Memorial Hospital. "These are patients in whom we see a lot of complication, and we need lots of ways to lower their cholesterol."
PCSK9-inhibitor treatment of other patient populations, such as patients who are statin intolerant, will "probably need outcome studies for us to feel comfortable with widespread adoption," he added. Dr. Eckel estimated that about 4 million American patients who have been prescribed a statin can’t take the drug because of intolerance.
The Open-Label Study of Long-Term Evaluation Against LDL-C (OSLER) enrolled patients who had been enrolled in any of four prior phase II studies of evolocumab that each ran for 12 weeks. OSLER randomized 736 patients at 155 centers in 17 countries to receive 420 mg of evolocumab as a subcutaneous injection every 4 weeks in addition to standard-of-care treatment; 368 patients were assigned to standard of care only as controls. Patients averaged 56 years of age, their average LDL cholesterol level at baseline was about 140 mg/dL, and just under two-thirds of patients also received treatment with a statin.
After 1 year, 86% of the evolocumab-treated patients had an LDL cholesterol level below 100 mg/dL and 63% had their level below 70 mg/dL. In contrast, 16% and 1% respectively of patients on standard care reached these levels.
"We’re super pleased with the safety and tolerability data we’ve seen so far," Dr. Koren said. Even among the 98 patients treated with evolocumab who had their LDL cholesterol level below 25 mg/dL at 1year, the data showed no signal of increased serious adverse reactions or kidney or liver abnormalities. The only suggestion of a possible problem was a reported 9% incidence of headaches, compared with a 3% rate among patients on standard care and a 6% rate among patients on evolocumab for 1 year with LDL cholesterol levels of less than 50 mg/dL.
Concurrent with Dr. Koren’s report, the results also were published online (Circulation 2013;128 [doi:10.1161/CIRCULATIONAHA.113.007012]).
OSLER was sponsored by Amgen. Dr. Koren said that he has received research support from Amgen and from Regeneron, Sanofi, Roche, and Pfizer.
On Twitter @mitchelzoler
DALLAS – Monthly treatment with a human antibody directed against the PCSK9 enzyme was safe and effective for substantially lowering low-density-lipoprotein cholesterol levels in 736 patients treated for 1 year in a controlled phase II study, the longest and largest series of patients treated with a PCSK9 antibody yet reported.
An injection of evolocumab (formerly known as Amgen 145) a human antibody to the proprotein convertase subtilisin/kexin type 9 (PCSK9) enzyme once every 4 weeks led to an average 52% reduction in blood levels of LDL cholesterol after 1 year on top of background treatment with standard-of-care medications, which in many cases also included maximal statin treatment, Dr. Michael J. Koren said at the American Heart Association scientific sessions.
In addition, the safety results for evolocumab "were very clean," he said, with no evidence that patients developed neutralizing antibody against the monoclonal antibody drug, and with injection-site reactions occurring in 5% of treated patients with one patient withdrawing because of the injection-site effect.
The Food and Drug Administration should "certainly vote to approve these drugs for familial hypercholesterolemia," said Dr. Koren, director of noninvasive cardiology at Jacksonville (Fla.) Memorial Hospital. "These are patients in whom we see a lot of complication, and we need lots of ways to lower their cholesterol."
PCSK9-inhibitor treatment of other patient populations, such as patients who are statin intolerant, will "probably need outcome studies for us to feel comfortable with widespread adoption," he added. Dr. Eckel estimated that about 4 million American patients who have been prescribed a statin can’t take the drug because of intolerance.
The Open-Label Study of Long-Term Evaluation Against LDL-C (OSLER) enrolled patients who had been enrolled in any of four prior phase II studies of evolocumab that each ran for 12 weeks. OSLER randomized 736 patients at 155 centers in 17 countries to receive 420 mg of evolocumab as a subcutaneous injection every 4 weeks in addition to standard-of-care treatment; 368 patients were assigned to standard of care only as controls. Patients averaged 56 years of age, their average LDL cholesterol level at baseline was about 140 mg/dL, and just under two-thirds of patients also received treatment with a statin.
After 1 year, 86% of the evolocumab-treated patients had an LDL cholesterol level below 100 mg/dL and 63% had their level below 70 mg/dL. In contrast, 16% and 1% respectively of patients on standard care reached these levels.
"We’re super pleased with the safety and tolerability data we’ve seen so far," Dr. Koren said. Even among the 98 patients treated with evolocumab who had their LDL cholesterol level below 25 mg/dL at 1year, the data showed no signal of increased serious adverse reactions or kidney or liver abnormalities. The only suggestion of a possible problem was a reported 9% incidence of headaches, compared with a 3% rate among patients on standard care and a 6% rate among patients on evolocumab for 1 year with LDL cholesterol levels of less than 50 mg/dL.
Concurrent with Dr. Koren’s report, the results also were published online (Circulation 2013;128 [doi:10.1161/CIRCULATIONAHA.113.007012]).
OSLER was sponsored by Amgen. Dr. Koren said that he has received research support from Amgen and from Regeneron, Sanofi, Roche, and Pfizer.
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Treatment with evolocumab every 4 weeks for 1 year safely cut average LDL cholesterol levels by 50%, compared with controls.
Data source: OSLER, an open-label study that randomized 368 patients to standard care and 736 to standard care plus evolocumab treatment.
Disclosures: OSLER was sponsored by Amgen. Dr. Koren said that he has received research support from Amgen and from Regeneron, Sanofi, Roche, and Pfizer.
Gene panel identifies residual neuroblastoma metastases
BRUSSELS – In advanced-stage neuroblastoma patients with residual metastases in their bone marrow following two cycles of anti-GD2 immunotherapy, another cycle of this treatment is futile and only causes adverse events, based on a review of 343 stage IV patients treated at one U.S. center.
"Bone marrow minimal residual disease [MRD] measured after two cycles of immunotherapy was the strongest predictor of outcome, irrespective of disease status at the start of immunotherapy," Dr. Nai-Kong V. Cheung said at the Markers in Cancer meeting. "If a patient is positive for MRD after two cycles, don’t continue the treatment."
In the series of 343 patients aged 18 months or older with metastatic, stage IV neuroblastoma that he reviewed, all patients with detectable MRD after two cycles of immunotherapy with GD2 antibody eventually relapsed or died within the next 5 years. In contrast, roughly half of the patients who lacked MRD after the first two cycles of anti-GD2 therapy remained progression free and alive during up to 20 years of follow-up.
The full course of anti-GD2 treatment takes 2 years, has the potential to cause adverse effects, and is painful and expensive. "Why continue and subject patients to a treatment that won’t be beneficial?" Dr. Cheung asked during an interview. "We can use [MRD] as a marker to take patients off of a protocol that will not be useful to them and try a different treatment."
Immunotherapy with anti-GD2 is part of standard treatment for patients with advanced neuroblastoma.
Dr. Cheung, a pediatric oncologist and head of the neuroblastoma program at Memorial Sloan-Kettering Cancer Center in New York, and his associates used a four-marker genetic analysis to find evidence of residual, metastatic neuroblastoma cells in patients’ bone marrow. The four markers they used were:
• GD2 synthase, the gene for an enzyme that helps produce a ganglioside-abundant in neuroblastoma cells;
• PHOX2B, the gene for a transcription factor that promotes nerve cell growth and maturation;
• CCND1, the gene for cyclin D1 protein, an oncogene; and
• ISL 1, the gene for islet 1, a transcription factor involved in cell growth.
The database included 169 patients treated during first remission, 69 treated during second or later remission, and 105 with primary refractory disease. The researchers used the four-test genetic panel to screen for MRD in bone marrow specimens taken from these patients after the first two rounds of anti-GD2 treatment with or without granulocyte-macrophage colony-stimulating factor in a series of four treatment protocols. A patient was considered positive for MRD if at least one of the genetic markers was positive for the presence of neuroblastoma cells in the bone marrow.
In a multivariate analysis, patients negative for MRD had about a fourfold increased rate of progression-free survival and about a threefold increased rate of overall survival, compared with patients positive for MRD; both differences were statistically significant.
Dr. Cheung said that the four-gene panel his group used was developed through a project begun 15 years ago to look for the most discriminating gene signatures of metastatic neuroblastoma cells against the background of normal bone marrow cells, the metastatic destination for at least 90% of advanced neuroblastoma tumors. A similar approach could identify genetic tests for treatment response of other metastatic tumor types, he said.
The meeting was sponsored by the American Society of Clinical Oncology, the European Organisation for Research and Treatment of Cancer, and the National Cancer Institute. Dr. Cheung said that he is a coinventor on patents held by Memorial Sloan-Kettering Cancer Center.
On Twitter @mitchelzoler
BRUSSELS – In advanced-stage neuroblastoma patients with residual metastases in their bone marrow following two cycles of anti-GD2 immunotherapy, another cycle of this treatment is futile and only causes adverse events, based on a review of 343 stage IV patients treated at one U.S. center.
"Bone marrow minimal residual disease [MRD] measured after two cycles of immunotherapy was the strongest predictor of outcome, irrespective of disease status at the start of immunotherapy," Dr. Nai-Kong V. Cheung said at the Markers in Cancer meeting. "If a patient is positive for MRD after two cycles, don’t continue the treatment."
In the series of 343 patients aged 18 months or older with metastatic, stage IV neuroblastoma that he reviewed, all patients with detectable MRD after two cycles of immunotherapy with GD2 antibody eventually relapsed or died within the next 5 years. In contrast, roughly half of the patients who lacked MRD after the first two cycles of anti-GD2 therapy remained progression free and alive during up to 20 years of follow-up.
The full course of anti-GD2 treatment takes 2 years, has the potential to cause adverse effects, and is painful and expensive. "Why continue and subject patients to a treatment that won’t be beneficial?" Dr. Cheung asked during an interview. "We can use [MRD] as a marker to take patients off of a protocol that will not be useful to them and try a different treatment."
Immunotherapy with anti-GD2 is part of standard treatment for patients with advanced neuroblastoma.
Dr. Cheung, a pediatric oncologist and head of the neuroblastoma program at Memorial Sloan-Kettering Cancer Center in New York, and his associates used a four-marker genetic analysis to find evidence of residual, metastatic neuroblastoma cells in patients’ bone marrow. The four markers they used were:
• GD2 synthase, the gene for an enzyme that helps produce a ganglioside-abundant in neuroblastoma cells;
• PHOX2B, the gene for a transcription factor that promotes nerve cell growth and maturation;
• CCND1, the gene for cyclin D1 protein, an oncogene; and
• ISL 1, the gene for islet 1, a transcription factor involved in cell growth.
The database included 169 patients treated during first remission, 69 treated during second or later remission, and 105 with primary refractory disease. The researchers used the four-test genetic panel to screen for MRD in bone marrow specimens taken from these patients after the first two rounds of anti-GD2 treatment with or without granulocyte-macrophage colony-stimulating factor in a series of four treatment protocols. A patient was considered positive for MRD if at least one of the genetic markers was positive for the presence of neuroblastoma cells in the bone marrow.
In a multivariate analysis, patients negative for MRD had about a fourfold increased rate of progression-free survival and about a threefold increased rate of overall survival, compared with patients positive for MRD; both differences were statistically significant.
Dr. Cheung said that the four-gene panel his group used was developed through a project begun 15 years ago to look for the most discriminating gene signatures of metastatic neuroblastoma cells against the background of normal bone marrow cells, the metastatic destination for at least 90% of advanced neuroblastoma tumors. A similar approach could identify genetic tests for treatment response of other metastatic tumor types, he said.
The meeting was sponsored by the American Society of Clinical Oncology, the European Organisation for Research and Treatment of Cancer, and the National Cancer Institute. Dr. Cheung said that he is a coinventor on patents held by Memorial Sloan-Kettering Cancer Center.
On Twitter @mitchelzoler
BRUSSELS – In advanced-stage neuroblastoma patients with residual metastases in their bone marrow following two cycles of anti-GD2 immunotherapy, another cycle of this treatment is futile and only causes adverse events, based on a review of 343 stage IV patients treated at one U.S. center.
"Bone marrow minimal residual disease [MRD] measured after two cycles of immunotherapy was the strongest predictor of outcome, irrespective of disease status at the start of immunotherapy," Dr. Nai-Kong V. Cheung said at the Markers in Cancer meeting. "If a patient is positive for MRD after two cycles, don’t continue the treatment."
In the series of 343 patients aged 18 months or older with metastatic, stage IV neuroblastoma that he reviewed, all patients with detectable MRD after two cycles of immunotherapy with GD2 antibody eventually relapsed or died within the next 5 years. In contrast, roughly half of the patients who lacked MRD after the first two cycles of anti-GD2 therapy remained progression free and alive during up to 20 years of follow-up.
The full course of anti-GD2 treatment takes 2 years, has the potential to cause adverse effects, and is painful and expensive. "Why continue and subject patients to a treatment that won’t be beneficial?" Dr. Cheung asked during an interview. "We can use [MRD] as a marker to take patients off of a protocol that will not be useful to them and try a different treatment."
Immunotherapy with anti-GD2 is part of standard treatment for patients with advanced neuroblastoma.
Dr. Cheung, a pediatric oncologist and head of the neuroblastoma program at Memorial Sloan-Kettering Cancer Center in New York, and his associates used a four-marker genetic analysis to find evidence of residual, metastatic neuroblastoma cells in patients’ bone marrow. The four markers they used were:
• GD2 synthase, the gene for an enzyme that helps produce a ganglioside-abundant in neuroblastoma cells;
• PHOX2B, the gene for a transcription factor that promotes nerve cell growth and maturation;
• CCND1, the gene for cyclin D1 protein, an oncogene; and
• ISL 1, the gene for islet 1, a transcription factor involved in cell growth.
The database included 169 patients treated during first remission, 69 treated during second or later remission, and 105 with primary refractory disease. The researchers used the four-test genetic panel to screen for MRD in bone marrow specimens taken from these patients after the first two rounds of anti-GD2 treatment with or without granulocyte-macrophage colony-stimulating factor in a series of four treatment protocols. A patient was considered positive for MRD if at least one of the genetic markers was positive for the presence of neuroblastoma cells in the bone marrow.
In a multivariate analysis, patients negative for MRD had about a fourfold increased rate of progression-free survival and about a threefold increased rate of overall survival, compared with patients positive for MRD; both differences were statistically significant.
Dr. Cheung said that the four-gene panel his group used was developed through a project begun 15 years ago to look for the most discriminating gene signatures of metastatic neuroblastoma cells against the background of normal bone marrow cells, the metastatic destination for at least 90% of advanced neuroblastoma tumors. A similar approach could identify genetic tests for treatment response of other metastatic tumor types, he said.
The meeting was sponsored by the American Society of Clinical Oncology, the European Organisation for Research and Treatment of Cancer, and the National Cancer Institute. Dr. Cheung said that he is a coinventor on patents held by Memorial Sloan-Kettering Cancer Center.
On Twitter @mitchelzoler
AT THE MARKERS IN CANCER MEETING
Major finding: In a multivariate analysis, patients negative for minimal residual disease had about a fourfold increased rate of progression-free survival and about a threefold increased rate of overall survival, compared with patients positive for MRD; both differences were statistically significant.
Data source: A review of 343 patients with stage IV neuroblastoma treated with immunotherapy at one U.S. center.
Disclosures: Dr. Cheung said that he is a coinventor on patents held by Memorial Sloan-Kettering Cancer Center.
'JNC 8' Guideline Follows Convoluted Endgame
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
'JNC 8' guideline follows convoluted endgame
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
The federally funded program to produce a set of U.S. guidelines for hypertension management, a process more than 5 years in the making, came to an unusual end on December 18 when the members of what had already become the officially-disbanded JNC 8 panel published their conclusions and guideline.
No longer recognized or supported by the National Heart Lung and Blood Institute (NHLBI), the Federal agency that had organized the Eighth Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 8) panel in 2008, and unwilling to work with potential collaborating groups like the American Heart Association (AHA), the American College of Cardiology (ACC), or the American Society of Hypertension (ASH), the 17-person group that wound up identifying themselves as the “panel members appointed to the Eighth Joint National Committee (JNC 8).”
Call them JNC Ain’t.
The U.S. hypertension guidelines began veering off on an unexpected course last June, when Dr. Gary H. Gibbons, NHLBI director, announced that the agency was withdrawing from issuing guidelines itself and would instead collaborate with “partner organizations.”
In August, Dr. Gibbons, said that the AHA and ACC had reached an agreement with the agency to “spearhead” development of three sets of practice guidelines, for hypertension, cholesterol, and obesity. This agreement led to the release in November of the cholesterol and obesity guidelines under the auspices of the AHA and ACC, but instead of also releasing hypertension guidelines, the AHA and ACC as well as the NHLBI said that the process had fallen through and failed to produce guidelines.
According to Dr. Paul A. James, co-chair of the former JNC 8 panel and professor of family medicine at the University of Iowa in Iowa City, that’s because the panel members decided they weren’t comfortable with “the idea of shopping our guideline around prior to publication and getting an endorsement.” Now that the panel’s conclusions have been published “we hope to get active public review of our work; we invite people to analyze our process, and hopefully organizations will endorse our findings,” he said in an interview. “Our belief is that the approach we took, the transparent nature of our guideline development, and our release of it through JAMA will increase the credibility of our work.”
But others said that the panel’s break with the NHLBI and its inability to partner with any organization will inevitably affect how people view these recommendations, especially because parts are also clinically controversial.
“There was clear controversy when this guideline was circulated” while under review, said Dr. John M. Flack, professor and chief of medicine at Wayne State University in Detroit. “The biggest problem this committee has is that many experts with a very significant stake in the recommendations were excluded from the process of generating the guideline. That limits buy-in from key opinion leaders, which will be needed for the uptake of this guideline into clinical practice,” he said in an interview.
“Unlike the previous JNC reports, this one will be seen as interesting, but not as persuasive,” said Dr. Michael A. Weber, professor of medicine at the State University of New York Downstate Medical Center in Brooklyn. “I believe the AHA, ACC, and ASH had hoped to endorse these guidelines, but that wasn’t possible.”
ASH leaders had discussions with the JNC 8 panel last summer, but the two groups could not reach an agreement on how to use the panel’s work for management recommendations, said Dr. William B. White, ASH president and chief of hypertension and clinical pharmacology at the University of Connecticut in Farmington.
What ASH has since agreed to do is join with the AHA and ACC to produce evidence-based hypertension guidelines using NHLBI materials, Dr. White said in an interview. The planned guidelines will “use some of the evidence derived by the NHLBI’s methodology, but adding to it other clinical issues,” including blood pressure measurement. These groups also hope the guidelines development will receive participation from a primary-care society such as the American College of Physicians, he said.
“The ACC/AHA Task Force on Practice Guidelines has begun the process of developing the collaborative model to update the national hypertensive guidelines in partnership with the NHLBI, which will provide an updated systematic review informed by the relevant critical clinical questions. We are seeking appropriate partners to begin this work in early 2014. The writing group will draft recommendations, followed by a peer and stakeholder review process. Once the review process is complete the ACC/AHA and partnering organizations will publish the guidelines in 2015 for clinicians to follow as the national standard for hypertension prevention and treatment,” said a spokeswoman for the American College of Cardiology in a statement released on December 18.
Leaders from the AHA and ACC said that once it became clear several weeks ago that they would not be able to collaborate with the JNC 8 panel, they felt compelled to immediately develop some form of updated guidance on hypertensive management. That led to an AHA-ACC Science Advisory (J. Am. Coll. Card. 2013;doi:10.1016/j.jacc.2013.11.007) released on Nov. 15 in collaboration with the Centers for Disease Control and Prevention that endorsed the use of treatment algorithms when managing patients with hypertension.
“Because the JNC 8 panel chose not to be part of the AHA-ACC structure, we felt we needed to go forward to make sure that we had guidance that reflected the evidence,” said Dr. Kim A. Williams Sr., professor and head of cardiology at Rush University in Chicago, vice president of the ACC, and a member of the group that wrote the advisory. “We felt the need to have risk covered as best we could, and have some hypertension guidance out there, even if it is not a guideline,” he said in an interview.
“We felt that after the enormous progress forward with the other four guidelines” released on Nov. 12 by the AHA and ACC (Circulation 2013 [doi: 10.1161/01.cir.0000437738.63853.7a; doi: 10.1161/01.cir.0000437739.71477.ee; doi: 10.1161/01.cir.0000437740.48606.d1; doi: 10.1161/01.cir.0000437741.48606.98]) “there was some urgency” to provide guidance for hypertension too, said Dr. Mariell Jessup, professor and medical director of the Penn Heart and Vascular Center at the University of Pennsylvania in Philadelphia and president of the AHA, during a session on the new guidelines at the AHA Scientific Sessions in Dallas in November.
The potential this now presents for the AHA and ACC to produce unified U.S. guidelines for all aspects of cardiovascular disease risk, integrating the assessment and treatment of hypertension, cholesterol, and obesity, is a positive development, said Dr. Eric D. Peterson, professor of medicine at Duke University in Durham, N.C. He was also hopeful that this new collaboration will draw in groups like ASH and the American College of Physicians to represent the interests of subspecialists and primary-care physicians. “Ideally you want consensus on where you’re trying to get blood pressure” that cuts across all strata of U.S. medicine, he said.
Dr. James, Dr. White, Dr. Jessup, Dr. Williams and Dr. Peterson said that they had no disclosures. Dr. Flack said that he has been a consultant to Novartis, Medtronic, and Back Beat Hypertension and received funding from Novartis and Medtronic. Dr. Weber said that he has been a consultant to Novartis, Takeda, and Forest.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
**UPDATED Jan. 4, 2014
Genotyping adds little to optimized warfarin dosing
DALLAS – Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we’ve seen, I don’t believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don’t think we’ll see another genetics trial in warfarin treatment. I think this is it."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients’ international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it’s more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment. Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient’s genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients’ average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial’s primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non–African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non–African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden. The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment. In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study’s primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting. The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1311386]).
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed and Dr. Ellinor had no disclosures.
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market – dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) – are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it’s what you know versus what you don’t know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you’d see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don’t encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn’t change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
DALLAS – Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we’ve seen, I don’t believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don’t think we’ll see another genetics trial in warfarin treatment. I think this is it."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients’ international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it’s more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment. Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient’s genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients’ average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial’s primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non–African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non–African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden. The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment. In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study’s primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting. The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1311386]).
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed and Dr. Ellinor had no disclosures.
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market – dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) – are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it’s what you know versus what you don’t know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you’d see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don’t encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn’t change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
DALLAS – Genotyping to guide the starting dosage of warfarin treatment showed no added value above tailoring treatment with a panel of clinical features in a randomized, controlled U.S. trial of more than 1,000 patients.
The resounding null result from adding genotype information should spell the end of this practice, said Dr. Stephen E. Kimmel, lead investigator for the study, and professor and director of cardiovascular epidemiology at the University of Pennsylvania in Philadelphia.
"Based on what we’ve seen, I don’t believe there is sufficient evidence to add genetic information on top of the available clinical algorithms," Dr. Kimmel said at the American Heart Association scientific sessions. "I don’t think we’ll see another genetics trial in warfarin treatment. I think this is it."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Good outcomes in the comparison group largely accounted for the failure of genotyping to significantly improve the percentage of time that patients’ international normalized ratios (INRs) were in the target range of 2.0-3.0. The amount of time in the therapeutic INR range (PTTR) averaged 45% in patients in the comparison arm during the first 4 weeks of warfarin treatment.
"When the comparison group does so well, it’s more difficult for genotyping to have an effect," said Dr. Elaine M. Hylek, professor of medicine and an anticoagulant specialist at Boston University.
That limitation, coupled with the shifting anticoagulant landscape, knock genotyping out of the picture, she agreed. "It will be difficult funding [another study of genotyping] with all the anticoagulant alternatives that are now out there" for preventing thrombosis in patients with atrial fibrillation or a recent venous thromboembolism.
The Clarification of Optimal Anticoagulant through Genetics (COAG) trial enrolled 1,015 patients initiating warfarin therapy at 18 U.S. centers during September 2009 to April 2013. The study randomized patients to two different ways to calculate their warfarin dosage during the first 5 days of treatment. Half had their dosage calculated by a formula that took into account seven clinical and demographic factors, including age, race, smoking status, and body surface area. The others had their dosage calculated with the same formula and factors plus added information on the patient’s genotype for two genes that affect warfarin activity, CYP2C9 and VKORC1. Patients’ average age was 58 years; just over a quarter were African American.
During the first 4 weeks on treatment, the average PTTR was 45% in both arms of the study, the trial’s primary endpoint. Adding genotyping information led to a bigger improvement in the PTTR among the non–African American patients compared with those who were African American, but did not produce a significantly increased PTTR in the non–African American subgroup.
Dr. Munir Pirmohamed reported results from a similar study that enrolled 455 patients starting warfarin therapy at any of five centers in the United Kingdom and Sweden. The EU-Pharmacogenetics of Anticoagulant Therapy (EU-PACT) Warfarin study mainly differed from the COAG study by the background method used to calculate a starting warfarin dosage over the first 3 days of treatment. In EU-PACT, the warfarin dosage of all patients was adjusted for age but not for other factors. In the intervention arm, the starting dosages were adjusted for the status of the same two genes, CYP2C9 and VKORC1.
During the first 12 weeks after starting warfarin, the cumulative average PTTR was 67% in the patients whose dose was adjusted by genotype and 60% in patients who were not genotyped, a statistically significant difference for this study’s primary endpoint, reported Dr. Pirmohamed, professor and head of molecular and clinical pharmacology at the University of Liverpool, England.
Taken together, the findings of the two studies highlight that patients should not start warfarin treatment on a fixed dosage, said Dr. Patrick T. Ellinor, a cardiologist and arrhythmia specialist at Massachusetts General Hospital, Boston, and designated discussant for both reports at the meeting. The findings support use of a clinical algorithm that takes into account several clinical factors, he added.
Concurrently with the meeting, the reports were published online for COAG (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1310669]) and for EU-PACT (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1311386]).
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed and Dr. Ellinor had no disclosures.
Warfarin on top despite competition
Warfarin remains the most widely used anticoagulant in the United States. Among patients with atrial fibrillation taking anticoagulation treatment, 72% used warfarin during the third quarter of 2013, data from a large registry show.
The new anticoagulants on the U.S. market – dabigatran (Pradaxa), apixaban (Eliquis), and rivaroxaban (Xarelto) – are gaining ground and widely acknowledged to be better, safer, and easier to manage. But warfarin clings to the market largely because of familiarity and low price, according to several experts.
"There is no doubt that the new anticoagulants look better compared with warfarin, but the clinical fact is that it’s what you know versus what you don’t know, and warfarin has been used for 60 years," Dr. Kimmel said at the press conference.
"The new drugs perform better than warfarin does; they get patients to where they need to be more quickly, but warfarin has been around a long time. We know its interactions and risks, there is the ability to reverse its effect, and the cost to patients is a real issue. If the new anticoagulants were the same price as warfarin then I think you’d see a lot more patients get a new drug," Dr. Ellinor said in an interview.
Data on current U.S. uptake of the new oral anticoagulants came from the 148,320 unique patients with atrial fibrillation included during June to September of 2013 in the PINNACLE Registry database run by the American College of Cardiology. Among these patients, about 83,000 (56%) were on some anticoagulant treatment, and within this subgroup, 72% were on warfarin, 27% on a new anticoagulant, and the remainder on different treatment, said Dr. John Gordon Harold, ACC president and a cardiologist at Cedars-Sinai Heart Institute in Los Angeles.
"In my own practice the new anticoagulants are being used with increasing frequency, mainly driven by direct-to-consumer advertising," Dr. Harold said in an interview. "We have patients who are completely stable on warfarin. (They) come in because of a consumer ad and they ask if they should switch drugs. When patients are stable I don’t encourage them to switch, but we have a shared decision making conversation and go over the pros and cons, the cost, and the outcomes data. A lot of patients prefer to pay the difference" and switch to a new anticoagulant.
Dr. Harold said he also recommends that patients switch off warfarin if they have problems with compliance and variability in their international normalized ratio (INR).
"If you can keep a patient on warfarin in their INR target range 80% or more of the time then I wouldn’t change, but most patients on warfarin have a very hard time maintaining an INR of 2-3," said Dr. Mark S. Link, professor and codirector of the cardiac arrhythmia center at Tufts Medical Center, Boston. But he said cost is a major factor keeping many patients on warfarin.
The new anticoagulants "are better than warfarin, but we are often forced by insurers to start with the cheaper drug," Dr. Link said during the news conference.
The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed, Dr. Ellinor, Dr. Harold, and Dr. Link had no relevant disclosures.
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Patients starting warfarin with dosing based on a formula that took into account seven clinical and demographic factors averaged 45% of the time in therapeutic range regardless of whether the starting dosage was adjusted based on genotype results.
Data source: COAG, a randomized trial with 1,015 patients starting warfarin therapy at 18 U.S. centers.
Disclosures: The COAG and EU-PACT studies did not receive any direct commercial sponsorship. Dr. Kimmel has been a consultant to Pfizer and Janssen. Dr. Hylek has been a consultant or adviser to Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Johnson & Johnson, and Pfizer. Dr. Pirmohamed and Dr. Ellinor had no disclosures.
Cardiac stress-imaging in stable angina patients
No one knows whether patients with stable ischemic heart disease and moderate or severe inducible ischemia benefit from revascularization when added to optimal medical therapy. A major, federally-funded trial named ISCHEMIA is underway to answer this question.
Main results from ISCHEMIA are still several years off, but the study has already produced an interesting finding about current cardiology practice and the way that cardiac stress-imaging studies are ordered.
Based on first-year enrollment data from the ISCHEMIA trial, the vast majority of both U.S. and European patients currently referred for stress-imaging assessment of ischemia have little or no inducible ischemia, Dr. Judith S. Hochman, head of the study, said during a talk at the American Heart Association’s Scientific Sessions in November.
Stable angina patients enrolled into ISCHEMIA need to have at least moderate inducible ischemia, defined as involving at least 10% of the left ventricle, in a cardiac imaging study read by a core laboratory. Since the trial began in July 2012, fewer than 700 patients had been enrolled based on imaging studies from about 15,000 patients. In the United States, about 3% of imaged patients had moderate or severe induced ischemia; the other 97% of patients referred for assessment had mild or no induced ischemia. In Europe, the rate with moderate or severe induced ischemia was slightly higher at 5%.
This observation made Dr. Hochman, a New York University cardiologist, ask why the prevalence of moderate or severe ischemia is so low in patients referred for stress imaging. She also wondered which patients with stable angina are undergoing revascularization today if so few qualify with moderate or severe inducible ischemia.
Another surprising fact she highlighted is how cardiologists manage patients found to have moderate or severe inducible ischemia. "Most of us think that all these patients with moderate to severe inducible ischemia are referred for catheterization," but that’s not what study results showed. She cited U.S. data from the mid- and late 2000s documenting that about one-third to two-thirds of these patients undergo cardiac catheterization.
This finding shows that there is "clinical equipoise" on how to manage these patients, and the ISCHEMIA trial will address that issue.
But until the results arrive in 2020, will as many stress-imaging studies continue for patients with stable angina when so many referred patients turn out to be negative for more advanced coronary disease? And, as Dr. Tracy Y. Wang, a cardiologist at Duke University, Durham, N.C., asked after hearing about the equivocal use of catheterization for patients with worse inducible ischemia: Why do physicians order these tests if they don’t intend to catheterize patients found to have moderate-to-severe inducible ischemia?
On Twitter @mitchelzoler
No one knows whether patients with stable ischemic heart disease and moderate or severe inducible ischemia benefit from revascularization when added to optimal medical therapy. A major, federally-funded trial named ISCHEMIA is underway to answer this question.
Main results from ISCHEMIA are still several years off, but the study has already produced an interesting finding about current cardiology practice and the way that cardiac stress-imaging studies are ordered.
Based on first-year enrollment data from the ISCHEMIA trial, the vast majority of both U.S. and European patients currently referred for stress-imaging assessment of ischemia have little or no inducible ischemia, Dr. Judith S. Hochman, head of the study, said during a talk at the American Heart Association’s Scientific Sessions in November.
Stable angina patients enrolled into ISCHEMIA need to have at least moderate inducible ischemia, defined as involving at least 10% of the left ventricle, in a cardiac imaging study read by a core laboratory. Since the trial began in July 2012, fewer than 700 patients had been enrolled based on imaging studies from about 15,000 patients. In the United States, about 3% of imaged patients had moderate or severe induced ischemia; the other 97% of patients referred for assessment had mild or no induced ischemia. In Europe, the rate with moderate or severe induced ischemia was slightly higher at 5%.
This observation made Dr. Hochman, a New York University cardiologist, ask why the prevalence of moderate or severe ischemia is so low in patients referred for stress imaging. She also wondered which patients with stable angina are undergoing revascularization today if so few qualify with moderate or severe inducible ischemia.
Another surprising fact she highlighted is how cardiologists manage patients found to have moderate or severe inducible ischemia. "Most of us think that all these patients with moderate to severe inducible ischemia are referred for catheterization," but that’s not what study results showed. She cited U.S. data from the mid- and late 2000s documenting that about one-third to two-thirds of these patients undergo cardiac catheterization.
This finding shows that there is "clinical equipoise" on how to manage these patients, and the ISCHEMIA trial will address that issue.
But until the results arrive in 2020, will as many stress-imaging studies continue for patients with stable angina when so many referred patients turn out to be negative for more advanced coronary disease? And, as Dr. Tracy Y. Wang, a cardiologist at Duke University, Durham, N.C., asked after hearing about the equivocal use of catheterization for patients with worse inducible ischemia: Why do physicians order these tests if they don’t intend to catheterize patients found to have moderate-to-severe inducible ischemia?
On Twitter @mitchelzoler
No one knows whether patients with stable ischemic heart disease and moderate or severe inducible ischemia benefit from revascularization when added to optimal medical therapy. A major, federally-funded trial named ISCHEMIA is underway to answer this question.
Main results from ISCHEMIA are still several years off, but the study has already produced an interesting finding about current cardiology practice and the way that cardiac stress-imaging studies are ordered.
Based on first-year enrollment data from the ISCHEMIA trial, the vast majority of both U.S. and European patients currently referred for stress-imaging assessment of ischemia have little or no inducible ischemia, Dr. Judith S. Hochman, head of the study, said during a talk at the American Heart Association’s Scientific Sessions in November.
Stable angina patients enrolled into ISCHEMIA need to have at least moderate inducible ischemia, defined as involving at least 10% of the left ventricle, in a cardiac imaging study read by a core laboratory. Since the trial began in July 2012, fewer than 700 patients had been enrolled based on imaging studies from about 15,000 patients. In the United States, about 3% of imaged patients had moderate or severe induced ischemia; the other 97% of patients referred for assessment had mild or no induced ischemia. In Europe, the rate with moderate or severe induced ischemia was slightly higher at 5%.
This observation made Dr. Hochman, a New York University cardiologist, ask why the prevalence of moderate or severe ischemia is so low in patients referred for stress imaging. She also wondered which patients with stable angina are undergoing revascularization today if so few qualify with moderate or severe inducible ischemia.
Another surprising fact she highlighted is how cardiologists manage patients found to have moderate or severe inducible ischemia. "Most of us think that all these patients with moderate to severe inducible ischemia are referred for catheterization," but that’s not what study results showed. She cited U.S. data from the mid- and late 2000s documenting that about one-third to two-thirds of these patients undergo cardiac catheterization.
This finding shows that there is "clinical equipoise" on how to manage these patients, and the ISCHEMIA trial will address that issue.
But until the results arrive in 2020, will as many stress-imaging studies continue for patients with stable angina when so many referred patients turn out to be negative for more advanced coronary disease? And, as Dr. Tracy Y. Wang, a cardiologist at Duke University, Durham, N.C., asked after hearing about the equivocal use of catheterization for patients with worse inducible ischemia: Why do physicians order these tests if they don’t intend to catheterize patients found to have moderate-to-severe inducible ischemia?
On Twitter @mitchelzoler