User login
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
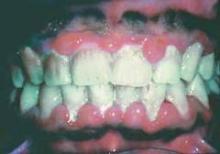
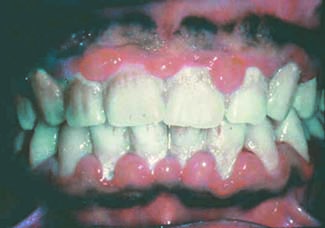
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler
The autoimmunity that causes rheumatoid arthritis needs to start somewhere in the body, and prime candidates include mucosal surfaces, like the mouth, gut, and lungs. The idea is that various microbes that colonize or infect these surfaces and form local microbiomes may trigger pathophysiologic processes that end up causing an autoimmune reaction, which results in rheumatoid arthritis.
Perhaps the most advanced research along these lines has looked at the links between rheumatoid arthritis (RA) and periodontitis, and particularly links between RA and infection with Porphyromonas gingivalis, a periodontitis pathogen. Evidence accumulated from several different research groups worldwide points to ways by which periodontal infection or P. gingivalis could produce autoimmune reactions, and the case has become compelling enough to launch a clinical intervention study that tries to interrupt this etiology.
"There is a good deal of evidence that the pattern of microbial colonization on mucosal surfaces contributes to autoimmunity, which probably also depends on underlying genetic predispositions," said Dr. Jerry A. Molitor, a rheumatologist at the University of Minnesota in Minneapolis. "I’m convinced there is a risk from periodontal infection for subsequent development of RA. Our data and multiple other data sets suggest that."
Dr. Molitor referred to epidemiologic data that he and his associates have analyzed from the nearly 16,000 Americans enrolled in Atherosclerosis Risk in Communities (ARIC), a cardiovascular-disease study that also collected data on RA and periodontitis as well as serum specimens. Their studies showed associations between periodontitis, development of antibodies to cyclic citrullinated peptides (CCPs), and later development of RA.
P. gingivalis is unique among oral bacteria in producing an enzyme capable of citrullinating human peptides and proteins.
"We see a very interesting association of immune responses to P. gingivalis and autoantibody expression" both in patients with RA and in those at high risk for developing RA, said Dr. Ted R. Mikuls, a rheumatologist at the University of Nebraska in Omaha. "We know that citrullinated proteins serve as targets for the autoimmunity that characterizes RA, so it’s very strong circumstantial evidence" for an etiologic relationship between P. gingivalis and RA, he said in an interview.
Results from a case-control study with 332 people that he published in 2012 showed that in those at high risk for developing RA, immunity to P. gingivalis was significantly associated with the presence of RA-related autoantibodies (Arthritis Rheum. 2012;64:3522-30).
A study of 31 new-onset RA patients, 34 chronic RA patients, and 18 healthy controls by a team of researchers at New York University showed that 62% of new-onset patients and 53% with chronic RA had severe periodontal disease, compared with 22% of the healthy controls (Arthritis Rheum. 2012;64:3083-94). Although indicators of exposure to P. gingivalis were similar in the new-onset RA patients and the other two subgroups, the new-onset patients showed unique exposures to Prevotella and Leptotrichia genuses, evidence implicating these bacteria as potential RA triggers.
A study of 50 patients with recently-diagnosed RA showed that 34% had immunoglobulin G antibody to P. gingivalis, often as anti-CCP antibody (Arthritis Res. Ther. 2013;15:R109). This subgroup also had significantly elevated levels of rheumatoid factor, a higher erythrocyte sedimentation rate, and a trend toward greater disease activity, reported Dr. Sheila L. Arvikar and her associates in a 2013 paper.
"Our work and those of others demonstrate that P. gingivalis seems to play a role in RA pathogenesis," said Dr. Arvikar, a rheumatologist at Massachusetts General Hospital in Boston. "I emphasize that it may only be important in a subset of patients. P. gingivalis antibodies and periodontal disease are not universal findings among RA patients. In other patients, different microbial pathogens and other sites may play more of a role, such as gut pathogens like Prevotella copri, or pathogens in the lungs and airways," she said in an interview.
Others agree that while the most study so far has been done on periodontitis and P. gingivalis, the range of other microbes that might be involved at mucosal sites in the mouth or elsewhere remains mostly unexamined.
"Is periodontitis the only risk factor? No. It’s probably a major player for a subset of patients, but RA is very heterogeneous," said Dr. Mikuls. "There are multiple pathways to the disease. Periodontitis is along at least one of those paths, but there are others."
Evidence implicating P. copri came from a study with 114 people: 44 patients with new-onset and untreated RA, 26 with chronic RA, 16 with psoriatic arthritis, and 28 healthy controls (eLife 2013;2:e01202). Researchers used stool specimens to assess each person’s intestinal microbiome and found a high prevalence of Prevotella species in general and P. copri specifically in the patients with new-onset RA. Presence of P. copri linked with reduced levels of other bacterial types, including beneficial microbes, and results from a mouse study suggested that P. copri exerts proinflammatory effects.
The intestinal microbiome "is vast and diverse. It contains 100 times more protein-coding genes than the human genome, and harbors 100 trillion cells, 10-fold the amount of total host human cells," wrote New York University researchers Dr. Jose U. Scher and Dr. Steven B. Abramson recently (Arthritis Res. Ther. 2013;15:122). "Virtually all studies consistently report only a small fraction of RA patients as being exposed to P. gingivalis." It is "plausible" that changes in the microbial composition at several mucosal sites is required for progression to RA, they wrote.
"The largest amount of recent evidence related to a specific species of bacteria and the etiology of RA has been for periodontal disease and P. gingivalis, but there have been many other investigations that focused on other factors and other mucosal sites, including the lung, gastrointestinal, and genitourinary mucosal surfaces," said Dr. M. Kristen Demoruelle, a rheumatologist at the University of Colorado in Aurora. "There are some very interesting data for P. gingivalis, especially the finding that it has an enzyme capable of citrullinating human proteins. However, other findings showed P. gingivalis was not specifically associated with RA, and studies of other mucosa sites such as the GI tract found other bacteria – not P. gingivalis – associated with RA. Overall, P. gingivalis and the gingival mucosa may play an important role in the etiology of RA in some people, but in many other people, other factors and other musocal sites are likely involved," she said in an interview.
The field "is still in its infancy and struggling to figure out the best way forward," cautioned Dr. Molitor. The range of possible mucosal sites and their microbiome diversity produce findings that are "complex and confusing," he said. "I am almost certain that no single study will provide a giant breakthrough, that this is the one bacterial species that is the major risk for RA and all we need to do is use one antibiotic to prevent it." Despite that, "the driving force behind this research remains the idea of preventing RA."
"I’d like to have a better understanding of the complex environment [of the oral mucosa] and the mechanisms involved in RA association, but early studies of intervention are probably warranted," said Dr. Mikuls.
"I think there is sufficient evidence to support" an intervention trial, agreed Dr. Arvikar. "Treatment of periodontal disease does not carry much risk, and it arguably is the standard of care."
She cited a periodontal disease intervention trial launched in France that enrolled 40 patients with RA and periodontal disease that was designed to assess the impact of periodontal treatment on RA. Another intervention run by Dr. Scher and Dr. Abramson from NYU randomized about 180 people with RA, psoriatic arthritis, or controls without arthritis to treatment with doxycycline, vancomycin, or placebo. Both studies were scheduled for completion during 2013.
"A key question is whether bacteria such as P. gingivalis are a cause or effect of RA. The main clinical application will be if a specific bacterium is identified to be causal. Then it could be directly targeted in treatment. And if a specific bacterium is associated with development of preclinical RA-related autoimmunity it may be a biomarker to identify future RA risk," Dr. Demoruelle said. "We still need to know the best time to target these bacteria. If bacteria trigger disease prior to the first symptoms, we’ll need to have ways to find these people."
With a deeper understanding of mucosal-surface microbiomes as well as contributing genetic factors "we may be able to design a reasonable, pre-disease intervention trial for people at very high risk, see if we can step in early and stop progression before it gets to where it results in full-blown RA," said Dr. Molitor.
Dr. Molitor, Dr. Mikuls, Dr. Arvikar, and Dr. Demoruelle said that they had no disclosures.
On Twitter @mitchelzoler