User login
Polymorphic light eruption: A diagnosis of exclusion
MAUI, HAWAII – Polymorphic light eruption is the most common of all the photodermatoses, yet few physicians will ever actually see the rash in their office, according to Dr. Vincent A. DeLeo.
That’s because polymorphic light eruption (PMLE) typically occurs when someone goes on vacation someplace sunny. By the time the patient returns home, the itchy rash is gone. But the history tells the tale, Dr. DeLeo said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
"PMLE is a clinical diagnosis you make when you’ve ruled out photoallergic contact dermatitis and lupus. It’s a diagnosis of exclusion," explained Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center, both in New York.
The salient features of PMLE are that it’s a recurrent rash, which typically erupts several days after – not within minutes of – sun exposure; it lasts for days; and it spares the face.
The face is spared because patients with PMLE gradually become "hardened" to sunlight. The more sun they get on a body part over time, the less likely they are to experience an eruption at that location. The face gets more sunlight than other body parts during everyday outdoor exposure. The rash of PMLE occurs on skin sites that are usually covered in winter, but exposed to the sun in the spring or while vacationing, such as the upper chest and arms.
A study conducted several decades ago among Harvard Medical School students suggested that PMLE affects about 10% of Americans. European studies indicate 21% of Swedes and 15% of U.K. residents are affected. Women outnumber men with PMLE by at least 2:1. The onset of the condition is typically in the 20s among women and somewhat later in men.
The itchy rash is variable in appearance. It can take the form of papules, papulovesicular lesions, or plaques. In the unlikely event the physician actually sees the rash and takes a biopsy, the histology shows a dense perivascular dermal lymphocytic infiltrate.
The differential diagnosis involves photoallergic contact dermatitis and lupus, both of which, like PMLE, feature recurrent, persistent rashes that erupt after a delay in response to sun exposure. Photoallergic contact dermatitis usually affects the face and almost always is due to sunscreens, with oxybenzone the No. 1 culprit. Photopatch testing will confirm the diagnosis. Titanium- or zinc-based physical sun blockers are protective.
It’s important to recognize that oxybenzone and other offenders are also present in shampoos, rinses, and other personal care products in addition to sunscreens, the dermatologist noted.
The plaque form of PMLE looks morphologically like lupus erythematosus (LE). But LE can be ruled out on the basis of negative results for antinuclear antibody, anti-Ro, and anti-La testing.
Because episodes of PMLE are recurrent and predictable, prophylaxis is the best approach. Dr. DeLeo recommended the use of sunscreens containing avobenzone and ecamsule; the combination works better than either agent alone.
If sunscreens don’t work, his top choice is narrow-band UVB phototherapy. "We use it like you would for psoriasis. We give most patients three treatments per week for 3-4 weeks before they go away on their trip. That usually suppresses the eruption. Essentially we’re hardening them," he said.
A product that physicians can expect to be hearing a lot more about is Polypodium leucotomos, an oral fern extract marketed as a sunscreen in pill form. In one recent open-label study involving 57 patients with PMLE, three-quarters of them benefited (G. Ital. Dermatol. Venereol. 2011;146:85-7). In another open-label study by other investigators, this one involving 35 PMLE patients, daily P. leucotomos also resulted in a significant reduction in sensitivity to sunlight (J. Am. Acad. Dermatol. 2012;66:58-62). The usual dosage is 240 mg twice daily.
P. leucotomos has been a commercially available natural product in Europe for 3 decades, with well-established safety. It has been acquired by Ferndale Pharma, which plans to raise its market profile in the United States.
Dr. DeLeo reported serving as a consultant to numerous pharmaceutical and cosmetics companies. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Polymorphic light eruption is the most common of all the photodermatoses, yet few physicians will ever actually see the rash in their office, according to Dr. Vincent A. DeLeo.
That’s because polymorphic light eruption (PMLE) typically occurs when someone goes on vacation someplace sunny. By the time the patient returns home, the itchy rash is gone. But the history tells the tale, Dr. DeLeo said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
"PMLE is a clinical diagnosis you make when you’ve ruled out photoallergic contact dermatitis and lupus. It’s a diagnosis of exclusion," explained Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center, both in New York.
The salient features of PMLE are that it’s a recurrent rash, which typically erupts several days after – not within minutes of – sun exposure; it lasts for days; and it spares the face.
The face is spared because patients with PMLE gradually become "hardened" to sunlight. The more sun they get on a body part over time, the less likely they are to experience an eruption at that location. The face gets more sunlight than other body parts during everyday outdoor exposure. The rash of PMLE occurs on skin sites that are usually covered in winter, but exposed to the sun in the spring or while vacationing, such as the upper chest and arms.
A study conducted several decades ago among Harvard Medical School students suggested that PMLE affects about 10% of Americans. European studies indicate 21% of Swedes and 15% of U.K. residents are affected. Women outnumber men with PMLE by at least 2:1. The onset of the condition is typically in the 20s among women and somewhat later in men.
The itchy rash is variable in appearance. It can take the form of papules, papulovesicular lesions, or plaques. In the unlikely event the physician actually sees the rash and takes a biopsy, the histology shows a dense perivascular dermal lymphocytic infiltrate.
The differential diagnosis involves photoallergic contact dermatitis and lupus, both of which, like PMLE, feature recurrent, persistent rashes that erupt after a delay in response to sun exposure. Photoallergic contact dermatitis usually affects the face and almost always is due to sunscreens, with oxybenzone the No. 1 culprit. Photopatch testing will confirm the diagnosis. Titanium- or zinc-based physical sun blockers are protective.
It’s important to recognize that oxybenzone and other offenders are also present in shampoos, rinses, and other personal care products in addition to sunscreens, the dermatologist noted.
The plaque form of PMLE looks morphologically like lupus erythematosus (LE). But LE can be ruled out on the basis of negative results for antinuclear antibody, anti-Ro, and anti-La testing.
Because episodes of PMLE are recurrent and predictable, prophylaxis is the best approach. Dr. DeLeo recommended the use of sunscreens containing avobenzone and ecamsule; the combination works better than either agent alone.
If sunscreens don’t work, his top choice is narrow-band UVB phototherapy. "We use it like you would for psoriasis. We give most patients three treatments per week for 3-4 weeks before they go away on their trip. That usually suppresses the eruption. Essentially we’re hardening them," he said.
A product that physicians can expect to be hearing a lot more about is Polypodium leucotomos, an oral fern extract marketed as a sunscreen in pill form. In one recent open-label study involving 57 patients with PMLE, three-quarters of them benefited (G. Ital. Dermatol. Venereol. 2011;146:85-7). In another open-label study by other investigators, this one involving 35 PMLE patients, daily P. leucotomos also resulted in a significant reduction in sensitivity to sunlight (J. Am. Acad. Dermatol. 2012;66:58-62). The usual dosage is 240 mg twice daily.
P. leucotomos has been a commercially available natural product in Europe for 3 decades, with well-established safety. It has been acquired by Ferndale Pharma, which plans to raise its market profile in the United States.
Dr. DeLeo reported serving as a consultant to numerous pharmaceutical and cosmetics companies. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Polymorphic light eruption is the most common of all the photodermatoses, yet few physicians will ever actually see the rash in their office, according to Dr. Vincent A. DeLeo.
That’s because polymorphic light eruption (PMLE) typically occurs when someone goes on vacation someplace sunny. By the time the patient returns home, the itchy rash is gone. But the history tells the tale, Dr. DeLeo said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
"PMLE is a clinical diagnosis you make when you’ve ruled out photoallergic contact dermatitis and lupus. It’s a diagnosis of exclusion," explained Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center, both in New York.
The salient features of PMLE are that it’s a recurrent rash, which typically erupts several days after – not within minutes of – sun exposure; it lasts for days; and it spares the face.
The face is spared because patients with PMLE gradually become "hardened" to sunlight. The more sun they get on a body part over time, the less likely they are to experience an eruption at that location. The face gets more sunlight than other body parts during everyday outdoor exposure. The rash of PMLE occurs on skin sites that are usually covered in winter, but exposed to the sun in the spring or while vacationing, such as the upper chest and arms.
A study conducted several decades ago among Harvard Medical School students suggested that PMLE affects about 10% of Americans. European studies indicate 21% of Swedes and 15% of U.K. residents are affected. Women outnumber men with PMLE by at least 2:1. The onset of the condition is typically in the 20s among women and somewhat later in men.
The itchy rash is variable in appearance. It can take the form of papules, papulovesicular lesions, or plaques. In the unlikely event the physician actually sees the rash and takes a biopsy, the histology shows a dense perivascular dermal lymphocytic infiltrate.
The differential diagnosis involves photoallergic contact dermatitis and lupus, both of which, like PMLE, feature recurrent, persistent rashes that erupt after a delay in response to sun exposure. Photoallergic contact dermatitis usually affects the face and almost always is due to sunscreens, with oxybenzone the No. 1 culprit. Photopatch testing will confirm the diagnosis. Titanium- or zinc-based physical sun blockers are protective.
It’s important to recognize that oxybenzone and other offenders are also present in shampoos, rinses, and other personal care products in addition to sunscreens, the dermatologist noted.
The plaque form of PMLE looks morphologically like lupus erythematosus (LE). But LE can be ruled out on the basis of negative results for antinuclear antibody, anti-Ro, and anti-La testing.
Because episodes of PMLE are recurrent and predictable, prophylaxis is the best approach. Dr. DeLeo recommended the use of sunscreens containing avobenzone and ecamsule; the combination works better than either agent alone.
If sunscreens don’t work, his top choice is narrow-band UVB phototherapy. "We use it like you would for psoriasis. We give most patients three treatments per week for 3-4 weeks before they go away on their trip. That usually suppresses the eruption. Essentially we’re hardening them," he said.
A product that physicians can expect to be hearing a lot more about is Polypodium leucotomos, an oral fern extract marketed as a sunscreen in pill form. In one recent open-label study involving 57 patients with PMLE, three-quarters of them benefited (G. Ital. Dermatol. Venereol. 2011;146:85-7). In another open-label study by other investigators, this one involving 35 PMLE patients, daily P. leucotomos also resulted in a significant reduction in sensitivity to sunlight (J. Am. Acad. Dermatol. 2012;66:58-62). The usual dosage is 240 mg twice daily.
P. leucotomos has been a commercially available natural product in Europe for 3 decades, with well-established safety. It has been acquired by Ferndale Pharma, which plans to raise its market profile in the United States.
Dr. DeLeo reported serving as a consultant to numerous pharmaceutical and cosmetics companies. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Six questions flag risk for cardiovascular hospitalization
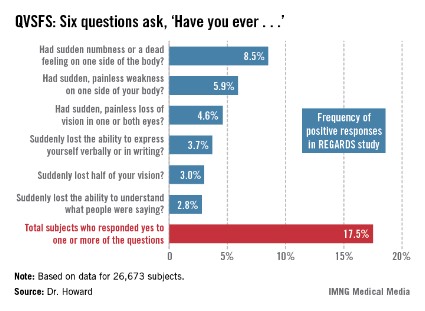
HONOLULU – The answers to six questions can identify 18% of the stroke-free general population as having a greater than 40% chance of hospitalization or an emergency department visit for cardiovascular disease within the next 5 years, according to a large national study.
New evidence from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) study indicates that the predictive power of the six questions compares to that of traditional cardiovascular risk factors, Virginia J. Howard, Ph.D., said at the International Stroke Conference sponsored by the American Heart Association.

These six questions – called the Questionnaire for Verifying Stroke-Free Status, or QVSFS – can easily be asked by office staff during a routine patient evaluation. Although the questions relate to strokelike symptoms, they also were predictive of other cardiovascular events.
REGARDS is a prospective, population-based, longitudinal study of 30,239 African Americans and whites over age 45. The national study oversampled African Americans, who made up 40% of the study population.
Dr. Howard’s analysis was restricted to the 26,673 subjects, mean age 64 years, with no baseline history of stroke or transient ischemic attack (TIA). Of these participants, 49% characterized their general health as excellent or very good, and another 35% described it as good. Yet 57% of subjects had hypertension, 32% were dyslipidemic, and 21% had diabetes.
During a mean 5.6 years of follow-up, 30% of subjects were hospitalized or had an emergency department visit for heart disease, stroke (9%), or TIA.
Fully 17.5% of subjects answered one or more of the questions affirmatively at baseline (see chart). Their rate of hospitalization or an ED visit for cardiovascular disease during the follow-up period was nearly twice that of patients who answered ‘no’ to all six questions.
In a multivariate analysis fully adjusted for demographics, socioeconomic status, conventional cardiovascular risk factors, and self-reported general health, subjects with any positive answer on the QVSFS at baseline had a 62% greater incidence of hospitalization or an ED visit for cardiovascular disease and a 47% greater incidence of hospitalization or a trip to the ED for stroke than did those who answered ‘no’ to all six questions, reported Dr. Howard of the University of Alabama, Birmingham.
A positive response to a single QVSFS question was provided by 10.5% of subjects. Another 4.6% gave affirmative answers to two questions, 1.4% did so for three, and 0.9% gave four or more positive answers.
In the fully adjusted multivariate analysis, each additional positive response was associated with an additional 29% increase in the risk of a cardiovascular hospitalization or ED visit and a 22% increase in the risk of a hospitalization or ED visit for stroke.
Looking at traditional cardiovascular risk factors, subjects with hypertension had a 12% greater risk of cardiovascular hospitalization or an ED visit and a 17% greater risk of stroke hospitalization or an ED visit. Diabetes conveyed a 29% greater risk of cardiovascular hospitalization or an ED visit and a 43% increase in stroke risk.
The REGARDS study is funded by the National Institute of Neurological Disorders and Stroke. Dr. Howard reported having no financial conflicts.
HONOLULU – The answers to six questions can identify 18% of the stroke-free general population as having a greater than 40% chance of hospitalization or an emergency department visit for cardiovascular disease within the next 5 years, according to a large national study.
New evidence from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) study indicates that the predictive power of the six questions compares to that of traditional cardiovascular risk factors, Virginia J. Howard, Ph.D., said at the International Stroke Conference sponsored by the American Heart Association.

These six questions – called the Questionnaire for Verifying Stroke-Free Status, or QVSFS – can easily be asked by office staff during a routine patient evaluation. Although the questions relate to strokelike symptoms, they also were predictive of other cardiovascular events.
REGARDS is a prospective, population-based, longitudinal study of 30,239 African Americans and whites over age 45. The national study oversampled African Americans, who made up 40% of the study population.
Dr. Howard’s analysis was restricted to the 26,673 subjects, mean age 64 years, with no baseline history of stroke or transient ischemic attack (TIA). Of these participants, 49% characterized their general health as excellent or very good, and another 35% described it as good. Yet 57% of subjects had hypertension, 32% were dyslipidemic, and 21% had diabetes.
During a mean 5.6 years of follow-up, 30% of subjects were hospitalized or had an emergency department visit for heart disease, stroke (9%), or TIA.
Fully 17.5% of subjects answered one or more of the questions affirmatively at baseline (see chart). Their rate of hospitalization or an ED visit for cardiovascular disease during the follow-up period was nearly twice that of patients who answered ‘no’ to all six questions.
In a multivariate analysis fully adjusted for demographics, socioeconomic status, conventional cardiovascular risk factors, and self-reported general health, subjects with any positive answer on the QVSFS at baseline had a 62% greater incidence of hospitalization or an ED visit for cardiovascular disease and a 47% greater incidence of hospitalization or a trip to the ED for stroke than did those who answered ‘no’ to all six questions, reported Dr. Howard of the University of Alabama, Birmingham.
A positive response to a single QVSFS question was provided by 10.5% of subjects. Another 4.6% gave affirmative answers to two questions, 1.4% did so for three, and 0.9% gave four or more positive answers.
In the fully adjusted multivariate analysis, each additional positive response was associated with an additional 29% increase in the risk of a cardiovascular hospitalization or ED visit and a 22% increase in the risk of a hospitalization or ED visit for stroke.
Looking at traditional cardiovascular risk factors, subjects with hypertension had a 12% greater risk of cardiovascular hospitalization or an ED visit and a 17% greater risk of stroke hospitalization or an ED visit. Diabetes conveyed a 29% greater risk of cardiovascular hospitalization or an ED visit and a 43% increase in stroke risk.
The REGARDS study is funded by the National Institute of Neurological Disorders and Stroke. Dr. Howard reported having no financial conflicts.
HONOLULU – The answers to six questions can identify 18% of the stroke-free general population as having a greater than 40% chance of hospitalization or an emergency department visit for cardiovascular disease within the next 5 years, according to a large national study.
New evidence from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) study indicates that the predictive power of the six questions compares to that of traditional cardiovascular risk factors, Virginia J. Howard, Ph.D., said at the International Stroke Conference sponsored by the American Heart Association.

These six questions – called the Questionnaire for Verifying Stroke-Free Status, or QVSFS – can easily be asked by office staff during a routine patient evaluation. Although the questions relate to strokelike symptoms, they also were predictive of other cardiovascular events.
REGARDS is a prospective, population-based, longitudinal study of 30,239 African Americans and whites over age 45. The national study oversampled African Americans, who made up 40% of the study population.
Dr. Howard’s analysis was restricted to the 26,673 subjects, mean age 64 years, with no baseline history of stroke or transient ischemic attack (TIA). Of these participants, 49% characterized their general health as excellent or very good, and another 35% described it as good. Yet 57% of subjects had hypertension, 32% were dyslipidemic, and 21% had diabetes.
During a mean 5.6 years of follow-up, 30% of subjects were hospitalized or had an emergency department visit for heart disease, stroke (9%), or TIA.
Fully 17.5% of subjects answered one or more of the questions affirmatively at baseline (see chart). Their rate of hospitalization or an ED visit for cardiovascular disease during the follow-up period was nearly twice that of patients who answered ‘no’ to all six questions.
In a multivariate analysis fully adjusted for demographics, socioeconomic status, conventional cardiovascular risk factors, and self-reported general health, subjects with any positive answer on the QVSFS at baseline had a 62% greater incidence of hospitalization or an ED visit for cardiovascular disease and a 47% greater incidence of hospitalization or a trip to the ED for stroke than did those who answered ‘no’ to all six questions, reported Dr. Howard of the University of Alabama, Birmingham.
A positive response to a single QVSFS question was provided by 10.5% of subjects. Another 4.6% gave affirmative answers to two questions, 1.4% did so for three, and 0.9% gave four or more positive answers.
In the fully adjusted multivariate analysis, each additional positive response was associated with an additional 29% increase in the risk of a cardiovascular hospitalization or ED visit and a 22% increase in the risk of a hospitalization or ED visit for stroke.
Looking at traditional cardiovascular risk factors, subjects with hypertension had a 12% greater risk of cardiovascular hospitalization or an ED visit and a 17% greater risk of stroke hospitalization or an ED visit. Diabetes conveyed a 29% greater risk of cardiovascular hospitalization or an ED visit and a 43% increase in stroke risk.
The REGARDS study is funded by the National Institute of Neurological Disorders and Stroke. Dr. Howard reported having no financial conflicts.
AT THE INTERNATIONAL STROKE CONFERENCE
Major finding: Any positive answer on the QVSFS at baseline was associated with a 62% greater incidence of hospitalization or an ED visit for cardiovascular disease.
Data source: REGARDS, a national, prospective, population-based, longitudinal study of more than 32,000 African-American and white adults.
Disclosures: The REGARDS study is sponsored by the National Institute of Neurological Disorders and Stroke. The presenter reported having no conflicts of interest.
Therapeutic combos make inroads in advanced melanoma
WAILEA, HAWAII – "The past 2 years have been a really exciting time for those of us who have spent the last several decades" in the field of melanoma, said Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York.
"We are in a whole new place with a very promising future for turning stage IV melanoma into maybe a chronic disease for many patients, instead of a death sentence. For some patients, we’re already seeing what may be cures," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The greatest enthusiasm in the field now involves combining a pathway-targeted agent, such as vemurafenib, with an immunologic checkpoint blocker, such as ipilimumab. The vemurafenib knocks down 60%-70% of metastatic melanomas temporarily and the ipilimumab promotes durable responses.
But there’s a formidable economic obstacle to this approach: The strongest drug combinations often put big pharmaceutical companies in the uncomfortable position of having to cooperate with their competitors in expensive research projects. "A lot of the drug companies, to their credit, are finding ways to make it work," Dr. Halpern said.
Dr. Halpern detailed the therapeutic history that has revolutionized the treatment of metastatic melanoma.
Prior to 2011 there were only two Food and Drug Administration–approved therapies for metastatic melanoma, dacarbazine and high-dose interleukin II. Both were unimpressive. The therapeutic dry spell has ended, he said. "There are for the first time in melanoma, instead of no hopeful drugs, a slew of hopeful drugs."
Targeted therapeutic approaches, the result of laboratory insights into the molecular pathways to melanoma and the key genetic mutations involved, led to the development of vemurafenib, a selective, first-in-class BRAF inhibitor approved in 2011.
"Vemurafenib is an astounding drug. When you give it to a BRAF-mutated cell, it essentially turns off the cell’s metabolic activity." When given to patients whose tumors test positive for the BRAF mutation, "it’s dramatically effective in 60%-70%." But the response does not persist. "After about 6-18 months, the tumor develops resistance to the drug. It’s like somebody hit a switch to turn the tumor back on. The tumor comes roaring back, in the same places for the most part," Dr. Halpern said.
As a result of this limited success, ongoing clinical trials are aimed at determining whether dual pathway blockade using combination therapy will provide more durable responses. Trials are underway with the oral BRAF inhibitor dabrafenib plus the oral MEK 1/2 pathway inhibitor trametinib. Other dual pathway combinations are also under study in melanoma.
The prospects are even more promising, according to Dr. Halpern, for immunologic checkpoint blockade, which is based upon the concept that some cancers progress because the immune system turns off prematurely and stops battling the malignancy. Ipilimumab is one such agent. An anti-CTLA-4 antibody, ipilimumab enhances T-cell activation and proliferation and has earned FDA approval as single-agent therapy in advanced melanoma.
Tumors often don’t begin to shrink until after 3-4 months, but the response is impressively durable in the roughly 30% of patients who respond to immunologic checkpoint blockade.
"These people look like they might be cured," said Dr. Halpern.
Another important immunologic checkpoint molecule is PD-1. The anti-PD-1 agent known as MDX-1106 appears to be nearly as effective as ipilimumab, but with less toxicity. The early impression from ongoing clinical trials is that dual immunologic checkpoint blockade using anti-CTLA-4 therapy along with an anti-PD-1 drug provides synergistic anti-tumor activity.
Dr. Halpern reported serving as a consultant to Canfield Scientific, DermTech, SciBase, Quintiles, and Lucid.
SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – "The past 2 years have been a really exciting time for those of us who have spent the last several decades" in the field of melanoma, said Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York.
"We are in a whole new place with a very promising future for turning stage IV melanoma into maybe a chronic disease for many patients, instead of a death sentence. For some patients, we’re already seeing what may be cures," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The greatest enthusiasm in the field now involves combining a pathway-targeted agent, such as vemurafenib, with an immunologic checkpoint blocker, such as ipilimumab. The vemurafenib knocks down 60%-70% of metastatic melanomas temporarily and the ipilimumab promotes durable responses.
But there’s a formidable economic obstacle to this approach: The strongest drug combinations often put big pharmaceutical companies in the uncomfortable position of having to cooperate with their competitors in expensive research projects. "A lot of the drug companies, to their credit, are finding ways to make it work," Dr. Halpern said.
Dr. Halpern detailed the therapeutic history that has revolutionized the treatment of metastatic melanoma.
Prior to 2011 there were only two Food and Drug Administration–approved therapies for metastatic melanoma, dacarbazine and high-dose interleukin II. Both were unimpressive. The therapeutic dry spell has ended, he said. "There are for the first time in melanoma, instead of no hopeful drugs, a slew of hopeful drugs."
Targeted therapeutic approaches, the result of laboratory insights into the molecular pathways to melanoma and the key genetic mutations involved, led to the development of vemurafenib, a selective, first-in-class BRAF inhibitor approved in 2011.
"Vemurafenib is an astounding drug. When you give it to a BRAF-mutated cell, it essentially turns off the cell’s metabolic activity." When given to patients whose tumors test positive for the BRAF mutation, "it’s dramatically effective in 60%-70%." But the response does not persist. "After about 6-18 months, the tumor develops resistance to the drug. It’s like somebody hit a switch to turn the tumor back on. The tumor comes roaring back, in the same places for the most part," Dr. Halpern said.
As a result of this limited success, ongoing clinical trials are aimed at determining whether dual pathway blockade using combination therapy will provide more durable responses. Trials are underway with the oral BRAF inhibitor dabrafenib plus the oral MEK 1/2 pathway inhibitor trametinib. Other dual pathway combinations are also under study in melanoma.
The prospects are even more promising, according to Dr. Halpern, for immunologic checkpoint blockade, which is based upon the concept that some cancers progress because the immune system turns off prematurely and stops battling the malignancy. Ipilimumab is one such agent. An anti-CTLA-4 antibody, ipilimumab enhances T-cell activation and proliferation and has earned FDA approval as single-agent therapy in advanced melanoma.
Tumors often don’t begin to shrink until after 3-4 months, but the response is impressively durable in the roughly 30% of patients who respond to immunologic checkpoint blockade.
"These people look like they might be cured," said Dr. Halpern.
Another important immunologic checkpoint molecule is PD-1. The anti-PD-1 agent known as MDX-1106 appears to be nearly as effective as ipilimumab, but with less toxicity. The early impression from ongoing clinical trials is that dual immunologic checkpoint blockade using anti-CTLA-4 therapy along with an anti-PD-1 drug provides synergistic anti-tumor activity.
Dr. Halpern reported serving as a consultant to Canfield Scientific, DermTech, SciBase, Quintiles, and Lucid.
SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – "The past 2 years have been a really exciting time for those of us who have spent the last several decades" in the field of melanoma, said Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan Kettering Cancer Center, New York.
"We are in a whole new place with a very promising future for turning stage IV melanoma into maybe a chronic disease for many patients, instead of a death sentence. For some patients, we’re already seeing what may be cures," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The greatest enthusiasm in the field now involves combining a pathway-targeted agent, such as vemurafenib, with an immunologic checkpoint blocker, such as ipilimumab. The vemurafenib knocks down 60%-70% of metastatic melanomas temporarily and the ipilimumab promotes durable responses.
But there’s a formidable economic obstacle to this approach: The strongest drug combinations often put big pharmaceutical companies in the uncomfortable position of having to cooperate with their competitors in expensive research projects. "A lot of the drug companies, to their credit, are finding ways to make it work," Dr. Halpern said.
Dr. Halpern detailed the therapeutic history that has revolutionized the treatment of metastatic melanoma.
Prior to 2011 there were only two Food and Drug Administration–approved therapies for metastatic melanoma, dacarbazine and high-dose interleukin II. Both were unimpressive. The therapeutic dry spell has ended, he said. "There are for the first time in melanoma, instead of no hopeful drugs, a slew of hopeful drugs."
Targeted therapeutic approaches, the result of laboratory insights into the molecular pathways to melanoma and the key genetic mutations involved, led to the development of vemurafenib, a selective, first-in-class BRAF inhibitor approved in 2011.
"Vemurafenib is an astounding drug. When you give it to a BRAF-mutated cell, it essentially turns off the cell’s metabolic activity." When given to patients whose tumors test positive for the BRAF mutation, "it’s dramatically effective in 60%-70%." But the response does not persist. "After about 6-18 months, the tumor develops resistance to the drug. It’s like somebody hit a switch to turn the tumor back on. The tumor comes roaring back, in the same places for the most part," Dr. Halpern said.
As a result of this limited success, ongoing clinical trials are aimed at determining whether dual pathway blockade using combination therapy will provide more durable responses. Trials are underway with the oral BRAF inhibitor dabrafenib plus the oral MEK 1/2 pathway inhibitor trametinib. Other dual pathway combinations are also under study in melanoma.
The prospects are even more promising, according to Dr. Halpern, for immunologic checkpoint blockade, which is based upon the concept that some cancers progress because the immune system turns off prematurely and stops battling the malignancy. Ipilimumab is one such agent. An anti-CTLA-4 antibody, ipilimumab enhances T-cell activation and proliferation and has earned FDA approval as single-agent therapy in advanced melanoma.
Tumors often don’t begin to shrink until after 3-4 months, but the response is impressively durable in the roughly 30% of patients who respond to immunologic checkpoint blockade.
"These people look like they might be cured," said Dr. Halpern.
Another important immunologic checkpoint molecule is PD-1. The anti-PD-1 agent known as MDX-1106 appears to be nearly as effective as ipilimumab, but with less toxicity. The early impression from ongoing clinical trials is that dual immunologic checkpoint blockade using anti-CTLA-4 therapy along with an anti-PD-1 drug provides synergistic anti-tumor activity.
Dr. Halpern reported serving as a consultant to Canfield Scientific, DermTech, SciBase, Quintiles, and Lucid.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Inclacumab reduces troponin in NSTEMI
SAN FRANCISCO – Inclacumab, an inhibitor of the P-selectin pathway, may be a novel treatment for reducing myocardial damage after percutaneous coronary intervention for non–ST-elevation myocardial infarction, based on results from a phase-II study.
The trial, SELECT-ACS, is best viewed as a proof-of-concept study utilizing a reduction in biomarkers of myocardial damage as the endpoint, Dr. Jean-Claude Tardif said at the annual meeting of the American College of Cardiology.
Periprocedural myocardial damage, albeit often mild in nature, is common in PCI patients and is due in part to platelet activation and inflammation. The clinical relevance of post-PCI changes in biomarkers of myocardial damage is uncertain. What is clear from SELECT-ACS is that inclacumab is biologically active in NSTEMI patients. Future phase III studies with hard clinical endpoints conducted in patients with or without PCI will be critical to determining the drug’s relevance, he said.
In addition, the results of the ongoing SELECT-CABG study, in which the effects of inclacumab are being studied in patients undergoing surgical revascularization, will be presented later this year, according to Dr. Tardif, professor of medicine and director of research at the Montreal Heart Institute.
Inclacumab is a human recombinant monoclonal antibody that is a highly specific P-selectin antagonist. P-selectin is a cell adhesion molecule known to play a critical role in communication between activated platelets, WBCs, and the arterial wall. In animal studies, P-selectin inhibition reduces platelet stickiness, macrophage accumulation, and neointimal formation after injury.
"The P-selectin pathway is really at the crossroads of thrombosis and inflammation," Dr. Tardif explained. "It’s probably important to see this drug, inclacumab, not only as an antithrombotic but as an anti-inflammatory agent."
In SELECT-ACS, 544 patients with NSTEMI scheduled for coronary angiography and possible PCI were randomized to receive a single 1-hour-long infusion of inclacumab at 5 or 20 mg/kg or a placebo infusion up to 24 hours before their procedure. Most patients received the infusion just a few hours before angiography.
The primary study endpoint in SELECT-ACS was change in troponin I level from baseline at 16 and 24 hours post-PCI, compared with placebo. Thus, the analysis was restricted to the 322 study participants who underwent PCI. The medically managed SELECT-ACS participants treated with inclacumab will be the subject of a future report.
In the inclacumab 20 mg/kg group, the drop in troponin I was 22% greater than seen in placebo-treated controls at 16 hours and 24% greater at 24 hours. Peak troponin I level was reduced by 24% relative to placebo, and the area under the curve over a 24-hour span was reduced by 34%.
In addition, the decrease in creatine kinase MB (CK-MB) fraction was 16% greater with inclacumab 20 mg/kg than with placebo at 16 hours, and it also showed a 17% greater decrease at 24 hours. The incidence of a CK-MB rise greater than three times the upper limit of normal within the first 24 hours following PCI was 8.9% with inclacumab 20 mg/kg, compared with 18% with placebo. Moreover, soluble P-selectin levels were 22% lower in the inclacumab 20 mg/kg group than in controls.
Inclacumab at 5 mg/kg had no effect.
The pattern and intensity of adverse events were similar in the inclacumab and placebo groups. Given the dual antithrombotic and anti-inflammatory effects of P-selectin inhibition, it’s encouraging to note that the inclacumab-treated patients had no increase in bleeding or infections, Dr. Tardif said.
The study was funded by F. Hoffmann-La Roche. Dr. Tardif reported having no relevant financial interests.
Simultaneous with Dr. Tardif’s presentation of the SELECT-ACS findings in San Francisco, the study was published online (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jaac.2013.03.003]).
The SELECT-ACS study is a great phase II trial – and I emphasize "phase II." It’s an early look at a very new and exciting thing. We haven’t been down this road before.

|
|
I’m struck by the disconnect between the inclacumab-induced reduction in troponin release and clinical events. There were no deaths in the placebo group, but four in the low-dose and two in the high-dose inclacumab groups. Also, nonfatal MI occurred in two placebo-treated patients, compared with four on low-dose and seven on high-dose inclacumab.
Whether this is a real signal, is due to chance, or is simply the way the investigators reported periprocedural MIs in this study is not entirely certain. More definitive understanding is likely to come from the phase-III trials.
Dr. Erik Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University in Durham, N.C. Dr. Ohman was the study discussant at ACC 13.
The SELECT-ACS study is a great phase II trial – and I emphasize "phase II." It’s an early look at a very new and exciting thing. We haven’t been down this road before.

|
|
I’m struck by the disconnect between the inclacumab-induced reduction in troponin release and clinical events. There were no deaths in the placebo group, but four in the low-dose and two in the high-dose inclacumab groups. Also, nonfatal MI occurred in two placebo-treated patients, compared with four on low-dose and seven on high-dose inclacumab.
Whether this is a real signal, is due to chance, or is simply the way the investigators reported periprocedural MIs in this study is not entirely certain. More definitive understanding is likely to come from the phase-III trials.
Dr. Erik Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University in Durham, N.C. Dr. Ohman was the study discussant at ACC 13.
The SELECT-ACS study is a great phase II trial – and I emphasize "phase II." It’s an early look at a very new and exciting thing. We haven’t been down this road before.

|
|
I’m struck by the disconnect between the inclacumab-induced reduction in troponin release and clinical events. There were no deaths in the placebo group, but four in the low-dose and two in the high-dose inclacumab groups. Also, nonfatal MI occurred in two placebo-treated patients, compared with four on low-dose and seven on high-dose inclacumab.
Whether this is a real signal, is due to chance, or is simply the way the investigators reported periprocedural MIs in this study is not entirely certain. More definitive understanding is likely to come from the phase-III trials.
Dr. Erik Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University in Durham, N.C. Dr. Ohman was the study discussant at ACC 13.
SAN FRANCISCO – Inclacumab, an inhibitor of the P-selectin pathway, may be a novel treatment for reducing myocardial damage after percutaneous coronary intervention for non–ST-elevation myocardial infarction, based on results from a phase-II study.
The trial, SELECT-ACS, is best viewed as a proof-of-concept study utilizing a reduction in biomarkers of myocardial damage as the endpoint, Dr. Jean-Claude Tardif said at the annual meeting of the American College of Cardiology.
Periprocedural myocardial damage, albeit often mild in nature, is common in PCI patients and is due in part to platelet activation and inflammation. The clinical relevance of post-PCI changes in biomarkers of myocardial damage is uncertain. What is clear from SELECT-ACS is that inclacumab is biologically active in NSTEMI patients. Future phase III studies with hard clinical endpoints conducted in patients with or without PCI will be critical to determining the drug’s relevance, he said.
In addition, the results of the ongoing SELECT-CABG study, in which the effects of inclacumab are being studied in patients undergoing surgical revascularization, will be presented later this year, according to Dr. Tardif, professor of medicine and director of research at the Montreal Heart Institute.
Inclacumab is a human recombinant monoclonal antibody that is a highly specific P-selectin antagonist. P-selectin is a cell adhesion molecule known to play a critical role in communication between activated platelets, WBCs, and the arterial wall. In animal studies, P-selectin inhibition reduces platelet stickiness, macrophage accumulation, and neointimal formation after injury.
"The P-selectin pathway is really at the crossroads of thrombosis and inflammation," Dr. Tardif explained. "It’s probably important to see this drug, inclacumab, not only as an antithrombotic but as an anti-inflammatory agent."
In SELECT-ACS, 544 patients with NSTEMI scheduled for coronary angiography and possible PCI were randomized to receive a single 1-hour-long infusion of inclacumab at 5 or 20 mg/kg or a placebo infusion up to 24 hours before their procedure. Most patients received the infusion just a few hours before angiography.
The primary study endpoint in SELECT-ACS was change in troponin I level from baseline at 16 and 24 hours post-PCI, compared with placebo. Thus, the analysis was restricted to the 322 study participants who underwent PCI. The medically managed SELECT-ACS participants treated with inclacumab will be the subject of a future report.
In the inclacumab 20 mg/kg group, the drop in troponin I was 22% greater than seen in placebo-treated controls at 16 hours and 24% greater at 24 hours. Peak troponin I level was reduced by 24% relative to placebo, and the area under the curve over a 24-hour span was reduced by 34%.
In addition, the decrease in creatine kinase MB (CK-MB) fraction was 16% greater with inclacumab 20 mg/kg than with placebo at 16 hours, and it also showed a 17% greater decrease at 24 hours. The incidence of a CK-MB rise greater than three times the upper limit of normal within the first 24 hours following PCI was 8.9% with inclacumab 20 mg/kg, compared with 18% with placebo. Moreover, soluble P-selectin levels were 22% lower in the inclacumab 20 mg/kg group than in controls.
Inclacumab at 5 mg/kg had no effect.
The pattern and intensity of adverse events were similar in the inclacumab and placebo groups. Given the dual antithrombotic and anti-inflammatory effects of P-selectin inhibition, it’s encouraging to note that the inclacumab-treated patients had no increase in bleeding or infections, Dr. Tardif said.
The study was funded by F. Hoffmann-La Roche. Dr. Tardif reported having no relevant financial interests.
Simultaneous with Dr. Tardif’s presentation of the SELECT-ACS findings in San Francisco, the study was published online (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jaac.2013.03.003]).
SAN FRANCISCO – Inclacumab, an inhibitor of the P-selectin pathway, may be a novel treatment for reducing myocardial damage after percutaneous coronary intervention for non–ST-elevation myocardial infarction, based on results from a phase-II study.
The trial, SELECT-ACS, is best viewed as a proof-of-concept study utilizing a reduction in biomarkers of myocardial damage as the endpoint, Dr. Jean-Claude Tardif said at the annual meeting of the American College of Cardiology.
Periprocedural myocardial damage, albeit often mild in nature, is common in PCI patients and is due in part to platelet activation and inflammation. The clinical relevance of post-PCI changes in biomarkers of myocardial damage is uncertain. What is clear from SELECT-ACS is that inclacumab is biologically active in NSTEMI patients. Future phase III studies with hard clinical endpoints conducted in patients with or without PCI will be critical to determining the drug’s relevance, he said.
In addition, the results of the ongoing SELECT-CABG study, in which the effects of inclacumab are being studied in patients undergoing surgical revascularization, will be presented later this year, according to Dr. Tardif, professor of medicine and director of research at the Montreal Heart Institute.
Inclacumab is a human recombinant monoclonal antibody that is a highly specific P-selectin antagonist. P-selectin is a cell adhesion molecule known to play a critical role in communication between activated platelets, WBCs, and the arterial wall. In animal studies, P-selectin inhibition reduces platelet stickiness, macrophage accumulation, and neointimal formation after injury.
"The P-selectin pathway is really at the crossroads of thrombosis and inflammation," Dr. Tardif explained. "It’s probably important to see this drug, inclacumab, not only as an antithrombotic but as an anti-inflammatory agent."
In SELECT-ACS, 544 patients with NSTEMI scheduled for coronary angiography and possible PCI were randomized to receive a single 1-hour-long infusion of inclacumab at 5 or 20 mg/kg or a placebo infusion up to 24 hours before their procedure. Most patients received the infusion just a few hours before angiography.
The primary study endpoint in SELECT-ACS was change in troponin I level from baseline at 16 and 24 hours post-PCI, compared with placebo. Thus, the analysis was restricted to the 322 study participants who underwent PCI. The medically managed SELECT-ACS participants treated with inclacumab will be the subject of a future report.
In the inclacumab 20 mg/kg group, the drop in troponin I was 22% greater than seen in placebo-treated controls at 16 hours and 24% greater at 24 hours. Peak troponin I level was reduced by 24% relative to placebo, and the area under the curve over a 24-hour span was reduced by 34%.
In addition, the decrease in creatine kinase MB (CK-MB) fraction was 16% greater with inclacumab 20 mg/kg than with placebo at 16 hours, and it also showed a 17% greater decrease at 24 hours. The incidence of a CK-MB rise greater than three times the upper limit of normal within the first 24 hours following PCI was 8.9% with inclacumab 20 mg/kg, compared with 18% with placebo. Moreover, soluble P-selectin levels were 22% lower in the inclacumab 20 mg/kg group than in controls.
Inclacumab at 5 mg/kg had no effect.
The pattern and intensity of adverse events were similar in the inclacumab and placebo groups. Given the dual antithrombotic and anti-inflammatory effects of P-selectin inhibition, it’s encouraging to note that the inclacumab-treated patients had no increase in bleeding or infections, Dr. Tardif said.
The study was funded by F. Hoffmann-La Roche. Dr. Tardif reported having no relevant financial interests.
Simultaneous with Dr. Tardif’s presentation of the SELECT-ACS findings in San Francisco, the study was published online (J. Am. Coll. Cardiol. 2013 [doi:10.1016/j.jaac.2013.03.003]).
AT ACC 13
Major finding: In the inclacumab 20 mg/kg group, the drop in troponin I was 22% greater than seen in placebo-treated controls at 16 hours and 24% greater at 24 hours. Peak troponin I level was reduced by 24%, relative to placebo, and the area under the curve over a 24-hour span was reduced by 34%.
Data source: SELECT-ACS was an international, prospective, placebo-controlled, randomized, double-blind trial involving 544 NSTEMI patients assigned to low- or high-dose inclacumab or placebo up to 24 hours prior to coronary angiography.
Disclosures: The study was funded by F. Hoffmann-La Roche. The presenter reported having no financial conflicts.
High-sensitivity troponin T assay shows prognostic superiority
SAN FRANCISCO – An investigational high-sensitivity troponin T assay provides prognostically meaningful information beyond that yielded by its commercially available sibling test.
The Roche high-sensitivity troponin T assay (hsTnT) excelled over the commercially available Roche Elecys fourth-generation cardiac troponin T test (cTnT) in patients presenting with high-risk non–ST-elevation acute coronary syndrome, Dr. Jonathan Grinstein reported at the annual meeting of the American College of Cardiology.
The investigational test results prove strikingly useful for the subset of patients who test hsTnT-positive and cTnT-negative. In this group, the adjusted odds ratio of 30-day cardiovascular death or new MI is 6.7-fold greater than that of patients who were hsTnT negative and cTnT positive.
"Saying it another way, among this patient subgroup, who represent about 3.5% of our total study population, if one were to analyze the troponins using only the fourth-generation assay, we’d be falsely reassured regarding the likelihood of recurrent cardiovascular events in the next 30 days. In reality, this is a high-risk group," Dr. Grinstein observed.
Both assays were utilized for comparative purposes in 4,160 patients with suspected high-risk non–ST-elevation acute coronary syndrome who participated in two completed and previously reported randomized clinical trials. The studies, known as SEPIA-ACS1 TIMI 42 and EARLY ACS, had as their primary purpose the investigation of novel anticoagulants and optimal timing of administration. All subjects included in this secondary analysis had cardiac ischemia at rest for at least 10 minutes and one or more other high-risk features and underwent dual troponin T testing within 24 hours following symptom onset, explained Dr. Grinstein, a cardiology fellow and TIMI study investigator at Brigham and Women’s Hospital, Boston.
The 99th percentile standard for the investigational hsTnT assay is 14 ng/L. The 3,697 patients in the two clinical trials who met or exceeded this standard had a 30-day incidence of cardiovascular death or new MI of 9.1%, compared with a much more modest rate of 1.9% in the 463 subjects with an hsTnT level below 14 ng/L.
The relationship between hsTnT and 30-day adverse outcome was continuous: The adverse event rate was 1.9% in the 463 patients with an hsTnT below 14 ng/L, 6.4% in 532 subjects with an hsTnT of 14-50 ng/L, 8.6% in 383 patients with a level of 50-100 ng/L, 9.5% in 2,417 patients with an hsTnT of 100-1,500 ng/L, and 11% in the 365 patients with a level in excess of 1,500 ng/L, according to Dr. Grinstein.
Fifteen percent of the subjects with an hsTnT below 14 ng/L had a high TIMI risk score, compared with 36% of those with an hsTnT of 14 ng/L or more.
Overall, two-thirds of the 30-day adverse events were centrally adjudicated acute MIs and one-third were cardiovascular deaths, but the proportion of cardiovascular deaths rose in patients with higher initial hsTnT values.
In the side-by-side analysis including both the hsTnT and fourth-generation cTnT assays, 463 patients had sufficiently low values on both tests that they were categorized as dual negative; their 30-day event rate was 1.9%. Another 378 patients were hsTnT negative but cTnT positive, with an associated 1.3% event rate.
More interesting were the 146 patients who were hsTnT positive and cTnT negative; their event rate was 8.2%. The adjusted odds ratio of 30-day cardiovascular death or new MI in such patients was 6.7-fold greater than in those who were hsTnT negative and fourth-generation positive.
Among the 3,551 subjects with a high-risk score on both assays, the 30-day cardiovascular event rate was 9.1%, he added.
The hsTnT assay, now under Food and Drug Administration review, is widely available in other countries. Compared with commercially available assays in the United States, it provides greatly increased sensitivity in the detection of myocardial necrosis. There have been concerns, however, that this enhanced sensitivity might come at the expense of reduced specificity, leaving the clinical significance of low-level elevations in hsTnT debatable. This study provides reassurance on that score, Dr. Grinstein said.
The SEPIA-ACS1 TIMI 42 trial was sponsored by Sanofi-Aventis. EARLY ACS was sponsored by Schering-Plough. The troponin T assays are manufactured by Roche. Dr. Grinstein reported having no financial conflicts.
SAN FRANCISCO – An investigational high-sensitivity troponin T assay provides prognostically meaningful information beyond that yielded by its commercially available sibling test.
The Roche high-sensitivity troponin T assay (hsTnT) excelled over the commercially available Roche Elecys fourth-generation cardiac troponin T test (cTnT) in patients presenting with high-risk non–ST-elevation acute coronary syndrome, Dr. Jonathan Grinstein reported at the annual meeting of the American College of Cardiology.
The investigational test results prove strikingly useful for the subset of patients who test hsTnT-positive and cTnT-negative. In this group, the adjusted odds ratio of 30-day cardiovascular death or new MI is 6.7-fold greater than that of patients who were hsTnT negative and cTnT positive.
"Saying it another way, among this patient subgroup, who represent about 3.5% of our total study population, if one were to analyze the troponins using only the fourth-generation assay, we’d be falsely reassured regarding the likelihood of recurrent cardiovascular events in the next 30 days. In reality, this is a high-risk group," Dr. Grinstein observed.
Both assays were utilized for comparative purposes in 4,160 patients with suspected high-risk non–ST-elevation acute coronary syndrome who participated in two completed and previously reported randomized clinical trials. The studies, known as SEPIA-ACS1 TIMI 42 and EARLY ACS, had as their primary purpose the investigation of novel anticoagulants and optimal timing of administration. All subjects included in this secondary analysis had cardiac ischemia at rest for at least 10 minutes and one or more other high-risk features and underwent dual troponin T testing within 24 hours following symptom onset, explained Dr. Grinstein, a cardiology fellow and TIMI study investigator at Brigham and Women’s Hospital, Boston.
The 99th percentile standard for the investigational hsTnT assay is 14 ng/L. The 3,697 patients in the two clinical trials who met or exceeded this standard had a 30-day incidence of cardiovascular death or new MI of 9.1%, compared with a much more modest rate of 1.9% in the 463 subjects with an hsTnT level below 14 ng/L.
The relationship between hsTnT and 30-day adverse outcome was continuous: The adverse event rate was 1.9% in the 463 patients with an hsTnT below 14 ng/L, 6.4% in 532 subjects with an hsTnT of 14-50 ng/L, 8.6% in 383 patients with a level of 50-100 ng/L, 9.5% in 2,417 patients with an hsTnT of 100-1,500 ng/L, and 11% in the 365 patients with a level in excess of 1,500 ng/L, according to Dr. Grinstein.
Fifteen percent of the subjects with an hsTnT below 14 ng/L had a high TIMI risk score, compared with 36% of those with an hsTnT of 14 ng/L or more.
Overall, two-thirds of the 30-day adverse events were centrally adjudicated acute MIs and one-third were cardiovascular deaths, but the proportion of cardiovascular deaths rose in patients with higher initial hsTnT values.
In the side-by-side analysis including both the hsTnT and fourth-generation cTnT assays, 463 patients had sufficiently low values on both tests that they were categorized as dual negative; their 30-day event rate was 1.9%. Another 378 patients were hsTnT negative but cTnT positive, with an associated 1.3% event rate.
More interesting were the 146 patients who were hsTnT positive and cTnT negative; their event rate was 8.2%. The adjusted odds ratio of 30-day cardiovascular death or new MI in such patients was 6.7-fold greater than in those who were hsTnT negative and fourth-generation positive.
Among the 3,551 subjects with a high-risk score on both assays, the 30-day cardiovascular event rate was 9.1%, he added.
The hsTnT assay, now under Food and Drug Administration review, is widely available in other countries. Compared with commercially available assays in the United States, it provides greatly increased sensitivity in the detection of myocardial necrosis. There have been concerns, however, that this enhanced sensitivity might come at the expense of reduced specificity, leaving the clinical significance of low-level elevations in hsTnT debatable. This study provides reassurance on that score, Dr. Grinstein said.
The SEPIA-ACS1 TIMI 42 trial was sponsored by Sanofi-Aventis. EARLY ACS was sponsored by Schering-Plough. The troponin T assays are manufactured by Roche. Dr. Grinstein reported having no financial conflicts.
SAN FRANCISCO – An investigational high-sensitivity troponin T assay provides prognostically meaningful information beyond that yielded by its commercially available sibling test.
The Roche high-sensitivity troponin T assay (hsTnT) excelled over the commercially available Roche Elecys fourth-generation cardiac troponin T test (cTnT) in patients presenting with high-risk non–ST-elevation acute coronary syndrome, Dr. Jonathan Grinstein reported at the annual meeting of the American College of Cardiology.
The investigational test results prove strikingly useful for the subset of patients who test hsTnT-positive and cTnT-negative. In this group, the adjusted odds ratio of 30-day cardiovascular death or new MI is 6.7-fold greater than that of patients who were hsTnT negative and cTnT positive.
"Saying it another way, among this patient subgroup, who represent about 3.5% of our total study population, if one were to analyze the troponins using only the fourth-generation assay, we’d be falsely reassured regarding the likelihood of recurrent cardiovascular events in the next 30 days. In reality, this is a high-risk group," Dr. Grinstein observed.
Both assays were utilized for comparative purposes in 4,160 patients with suspected high-risk non–ST-elevation acute coronary syndrome who participated in two completed and previously reported randomized clinical trials. The studies, known as SEPIA-ACS1 TIMI 42 and EARLY ACS, had as their primary purpose the investigation of novel anticoagulants and optimal timing of administration. All subjects included in this secondary analysis had cardiac ischemia at rest for at least 10 minutes and one or more other high-risk features and underwent dual troponin T testing within 24 hours following symptom onset, explained Dr. Grinstein, a cardiology fellow and TIMI study investigator at Brigham and Women’s Hospital, Boston.
The 99th percentile standard for the investigational hsTnT assay is 14 ng/L. The 3,697 patients in the two clinical trials who met or exceeded this standard had a 30-day incidence of cardiovascular death or new MI of 9.1%, compared with a much more modest rate of 1.9% in the 463 subjects with an hsTnT level below 14 ng/L.
The relationship between hsTnT and 30-day adverse outcome was continuous: The adverse event rate was 1.9% in the 463 patients with an hsTnT below 14 ng/L, 6.4% in 532 subjects with an hsTnT of 14-50 ng/L, 8.6% in 383 patients with a level of 50-100 ng/L, 9.5% in 2,417 patients with an hsTnT of 100-1,500 ng/L, and 11% in the 365 patients with a level in excess of 1,500 ng/L, according to Dr. Grinstein.
Fifteen percent of the subjects with an hsTnT below 14 ng/L had a high TIMI risk score, compared with 36% of those with an hsTnT of 14 ng/L or more.
Overall, two-thirds of the 30-day adverse events were centrally adjudicated acute MIs and one-third were cardiovascular deaths, but the proportion of cardiovascular deaths rose in patients with higher initial hsTnT values.
In the side-by-side analysis including both the hsTnT and fourth-generation cTnT assays, 463 patients had sufficiently low values on both tests that they were categorized as dual negative; their 30-day event rate was 1.9%. Another 378 patients were hsTnT negative but cTnT positive, with an associated 1.3% event rate.
More interesting were the 146 patients who were hsTnT positive and cTnT negative; their event rate was 8.2%. The adjusted odds ratio of 30-day cardiovascular death or new MI in such patients was 6.7-fold greater than in those who were hsTnT negative and fourth-generation positive.
Among the 3,551 subjects with a high-risk score on both assays, the 30-day cardiovascular event rate was 9.1%, he added.
The hsTnT assay, now under Food and Drug Administration review, is widely available in other countries. Compared with commercially available assays in the United States, it provides greatly increased sensitivity in the detection of myocardial necrosis. There have been concerns, however, that this enhanced sensitivity might come at the expense of reduced specificity, leaving the clinical significance of low-level elevations in hsTnT debatable. This study provides reassurance on that score, Dr. Grinstein said.
The SEPIA-ACS1 TIMI 42 trial was sponsored by Sanofi-Aventis. EARLY ACS was sponsored by Schering-Plough. The troponin T assays are manufactured by Roche. Dr. Grinstein reported having no financial conflicts.
AT ACC 13
Major Finding: Patients with suspected high-risk non–ST-elevation acute coronary syndrome had a 30-day incidence of cardiovascular death or new MI of 8.2% if they had a positive result on an investigational high-sensitivity troponin T assay and a negative result on a commercially available fourth-generation cardiac troponin T test.
Data Source: A secondary analysis of data on 4,160 participants in EARLY ACS and SEPIA-ACS1 TIMI 42.
Disclosures: The SEPIA-ACS1 TIMI 42 trial was sponsored by Sanofi-Aventis. EARLY ACS was sponsored by Schering-Plough. The troponin T assays are manufactured by Roche. The presenter reported having no financial conflicts.
Dermoscopy characterized as patient trust builder
MAUI, HAWAII – Dermatologists in the know view dermoscopy as a powerful tool to increase diagnostic accuracy in evaluating pigmented lesions; less well appreciated is dermoscopy’s value in building patient trust in the physician, according to Dr. Steven Q. Wang.
"With the dermoscope, people feel like you’re providing a much more detailed exam," observed Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan-Kettering Cancer Center’s Basking Ridge, N.J., campus.
"We are a tertiary referral center. We have lots of patients come in who are high risk, with a personal or family history of melanoma and numerous nevi. I always ask why they have transferred their care. The common answer I hear is they feel their dermatologist was not giving them a detailed examination," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wang said he makes a point of giving every patient a full-body clinical examination, including looking between the toes. And he views every single lesion with his dermoscope, up close and personal, scope to skin.
"The dermoscope makes it easier to spot the outlier lesions, the ugly ducklings. And we’re all so busy in the office, running from room to room – dermoscopy helps me slow down my mind and really look," he explained.
It doesn’t take all that long, either. In a classic multicenter study, 1,328 patients with one or more melanocytic or nonmelanocytic skin lesions were randomized to receive a complete skin examination with or without dermoscopy. The median time for a complete skin examination alone was 70 seconds; with dermoscopy it rose to 142 seconds (Arch. Dermatol. 2008;144:509-13).
"You double the time required, but it’s still only a little over 2 minutes. Yet you’ve changed the patient’s perception," Dr. Wang said.
A digital dermoscope is basically a handheld microscope that permits detailed visualization of structures in the deep epidermis and superficial dermis not visible to the naked eye. There’s a learning curve involved. Dermatologists who pick up a dermoscope and try to use it without formal training have worse diagnostic accuracy than with clinical examination, while experienced dermoscopists have significantly greater diagnostic accuracy than can be achieved with clinical exam alone (Lancet Oncol. 2002;3:159-65).
Dr. Wang said that because patients have more trust in their dermatologist when they feel they are receiving a thorough skin examination including dermoscopy, they are more likely to be adherent to scheduled follow-up evaluations. And that, in turn, spells improved long-term outcomes in patients at elevated risk for melanoma.
This point was already brought home forcefully for him nearly a decade ago, he said, when he and his coinvestigators reported their experience with long-term follow-up of 258 patients at high risk for melanoma. The monitoring strategy consisted of annual total body photography, total skin examination, and dermoscopy. The cumulative 10-year incidence of melanoma was 14% in the 160 patients with classic atypical mole syndrome and 10% in the other 98 high-risk patients. Impressively, all of the melanomas were either in situ or less than 1 mm thick. There were no metastases and no melanoma-related deaths (J. Am. Acad. Dermatol. 2004;50:15-20).
Although dermoscopy is used primarily in examining pigmented skin lesions, it has other applications. For example, in performing Mohs surgery for basal cell carcinomas, Dr. Wang has found dermoscopy to be of assistance in a couple of ways: In patients with ill-defined tumor borders on clinical examination, dermoscopy can define the tumor borders presurgically, thereby reducing the number of Mohs surgical stages required; and when a patient returns for Mohs surgery 6 weeks after skin biopsy and the biopsy site has healed so completely it can’t be found with the naked eye, the dermoscope can identify the site by visualizing subtle scars and telangiectasias.
In general dermatology, Dr. Wang said he turns to dermoscopy as an aid in diagnosing connective tissue diseases, including dermatomyositis, scleroderma, and lupus. He applies ultrasound gel to the proximal nail fold and examines the site using the dermoscope. A finding of dilated blood vessels stands out as a helpful diagnostic clue.
In addition, Dr. Wang said he has utilized the dermoscope in diagnosing scabies by spotting the mites and their trails, in detecting the telltale Wickham striae of lichen planus, and in diagnosing other dermatologic disorders.
Dr. Wang reported having no relevant financial conflicts.
SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Dermatologists in the know view dermoscopy as a powerful tool to increase diagnostic accuracy in evaluating pigmented lesions; less well appreciated is dermoscopy’s value in building patient trust in the physician, according to Dr. Steven Q. Wang.
"With the dermoscope, people feel like you’re providing a much more detailed exam," observed Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan-Kettering Cancer Center’s Basking Ridge, N.J., campus.
"We are a tertiary referral center. We have lots of patients come in who are high risk, with a personal or family history of melanoma and numerous nevi. I always ask why they have transferred their care. The common answer I hear is they feel their dermatologist was not giving them a detailed examination," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wang said he makes a point of giving every patient a full-body clinical examination, including looking between the toes. And he views every single lesion with his dermoscope, up close and personal, scope to skin.
"The dermoscope makes it easier to spot the outlier lesions, the ugly ducklings. And we’re all so busy in the office, running from room to room – dermoscopy helps me slow down my mind and really look," he explained.
It doesn’t take all that long, either. In a classic multicenter study, 1,328 patients with one or more melanocytic or nonmelanocytic skin lesions were randomized to receive a complete skin examination with or without dermoscopy. The median time for a complete skin examination alone was 70 seconds; with dermoscopy it rose to 142 seconds (Arch. Dermatol. 2008;144:509-13).
"You double the time required, but it’s still only a little over 2 minutes. Yet you’ve changed the patient’s perception," Dr. Wang said.
A digital dermoscope is basically a handheld microscope that permits detailed visualization of structures in the deep epidermis and superficial dermis not visible to the naked eye. There’s a learning curve involved. Dermatologists who pick up a dermoscope and try to use it without formal training have worse diagnostic accuracy than with clinical examination, while experienced dermoscopists have significantly greater diagnostic accuracy than can be achieved with clinical exam alone (Lancet Oncol. 2002;3:159-65).
Dr. Wang said that because patients have more trust in their dermatologist when they feel they are receiving a thorough skin examination including dermoscopy, they are more likely to be adherent to scheduled follow-up evaluations. And that, in turn, spells improved long-term outcomes in patients at elevated risk for melanoma.
This point was already brought home forcefully for him nearly a decade ago, he said, when he and his coinvestigators reported their experience with long-term follow-up of 258 patients at high risk for melanoma. The monitoring strategy consisted of annual total body photography, total skin examination, and dermoscopy. The cumulative 10-year incidence of melanoma was 14% in the 160 patients with classic atypical mole syndrome and 10% in the other 98 high-risk patients. Impressively, all of the melanomas were either in situ or less than 1 mm thick. There were no metastases and no melanoma-related deaths (J. Am. Acad. Dermatol. 2004;50:15-20).
Although dermoscopy is used primarily in examining pigmented skin lesions, it has other applications. For example, in performing Mohs surgery for basal cell carcinomas, Dr. Wang has found dermoscopy to be of assistance in a couple of ways: In patients with ill-defined tumor borders on clinical examination, dermoscopy can define the tumor borders presurgically, thereby reducing the number of Mohs surgical stages required; and when a patient returns for Mohs surgery 6 weeks after skin biopsy and the biopsy site has healed so completely it can’t be found with the naked eye, the dermoscope can identify the site by visualizing subtle scars and telangiectasias.
In general dermatology, Dr. Wang said he turns to dermoscopy as an aid in diagnosing connective tissue diseases, including dermatomyositis, scleroderma, and lupus. He applies ultrasound gel to the proximal nail fold and examines the site using the dermoscope. A finding of dilated blood vessels stands out as a helpful diagnostic clue.
In addition, Dr. Wang said he has utilized the dermoscope in diagnosing scabies by spotting the mites and their trails, in detecting the telltale Wickham striae of lichen planus, and in diagnosing other dermatologic disorders.
Dr. Wang reported having no relevant financial conflicts.
SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Dermatologists in the know view dermoscopy as a powerful tool to increase diagnostic accuracy in evaluating pigmented lesions; less well appreciated is dermoscopy’s value in building patient trust in the physician, according to Dr. Steven Q. Wang.
"With the dermoscope, people feel like you’re providing a much more detailed exam," observed Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan-Kettering Cancer Center’s Basking Ridge, N.J., campus.
"We are a tertiary referral center. We have lots of patients come in who are high risk, with a personal or family history of melanoma and numerous nevi. I always ask why they have transferred their care. The common answer I hear is they feel their dermatologist was not giving them a detailed examination," he said at the Hawaii Dermatology Seminar sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wang said he makes a point of giving every patient a full-body clinical examination, including looking between the toes. And he views every single lesion with his dermoscope, up close and personal, scope to skin.
"The dermoscope makes it easier to spot the outlier lesions, the ugly ducklings. And we’re all so busy in the office, running from room to room – dermoscopy helps me slow down my mind and really look," he explained.
It doesn’t take all that long, either. In a classic multicenter study, 1,328 patients with one or more melanocytic or nonmelanocytic skin lesions were randomized to receive a complete skin examination with or without dermoscopy. The median time for a complete skin examination alone was 70 seconds; with dermoscopy it rose to 142 seconds (Arch. Dermatol. 2008;144:509-13).
"You double the time required, but it’s still only a little over 2 minutes. Yet you’ve changed the patient’s perception," Dr. Wang said.
A digital dermoscope is basically a handheld microscope that permits detailed visualization of structures in the deep epidermis and superficial dermis not visible to the naked eye. There’s a learning curve involved. Dermatologists who pick up a dermoscope and try to use it without formal training have worse diagnostic accuracy than with clinical examination, while experienced dermoscopists have significantly greater diagnostic accuracy than can be achieved with clinical exam alone (Lancet Oncol. 2002;3:159-65).
Dr. Wang said that because patients have more trust in their dermatologist when they feel they are receiving a thorough skin examination including dermoscopy, they are more likely to be adherent to scheduled follow-up evaluations. And that, in turn, spells improved long-term outcomes in patients at elevated risk for melanoma.
This point was already brought home forcefully for him nearly a decade ago, he said, when he and his coinvestigators reported their experience with long-term follow-up of 258 patients at high risk for melanoma. The monitoring strategy consisted of annual total body photography, total skin examination, and dermoscopy. The cumulative 10-year incidence of melanoma was 14% in the 160 patients with classic atypical mole syndrome and 10% in the other 98 high-risk patients. Impressively, all of the melanomas were either in situ or less than 1 mm thick. There were no metastases and no melanoma-related deaths (J. Am. Acad. Dermatol. 2004;50:15-20).
Although dermoscopy is used primarily in examining pigmented skin lesions, it has other applications. For example, in performing Mohs surgery for basal cell carcinomas, Dr. Wang has found dermoscopy to be of assistance in a couple of ways: In patients with ill-defined tumor borders on clinical examination, dermoscopy can define the tumor borders presurgically, thereby reducing the number of Mohs surgical stages required; and when a patient returns for Mohs surgery 6 weeks after skin biopsy and the biopsy site has healed so completely it can’t be found with the naked eye, the dermoscope can identify the site by visualizing subtle scars and telangiectasias.
In general dermatology, Dr. Wang said he turns to dermoscopy as an aid in diagnosing connective tissue diseases, including dermatomyositis, scleroderma, and lupus. He applies ultrasound gel to the proximal nail fold and examines the site using the dermoscope. A finding of dilated blood vessels stands out as a helpful diagnostic clue.
In addition, Dr. Wang said he has utilized the dermoscope in diagnosing scabies by spotting the mites and their trails, in detecting the telltale Wickham striae of lichen planus, and in diagnosing other dermatologic disorders.
Dr. Wang reported having no relevant financial conflicts.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
No benefit of endovascular therapy added to TPA for stroke
Functional outcomes in patients treated with intravenous tissue plasminogen activator with or without endovascular therapy after a moderate to severe acute ischemic stroke were not significantly different, and safety outcomes were similar, in a study that was stopped early because of these results.
In the IMS (Interventional Management of Stroke) III study, 40.8% of patients randomized to receive endovascular therapy plus intravenous TPA met the primary endpoint, a measure of functional independence -- a modified Rankin score of 2 or less at 90 days -- compared with 38.7% among those who had intravenous TPA alone, a difference that was not statistically significant, reported Dr. Joseph Broderick of the University of Cincinnati Neuroscience Institute, and the other IMS III investigators.
Mortality and other safety outcomes were also not significantly different between the two groups of patients in the study, which was stopped early because of futility after 656 of the planned 900 patients had been randomized.
The study was published online to coincide with the presentation of the results at the International Stroke Conference (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1214300]).
Referring to the lack of randomized clinical trial data, the authors pointed out that it is uncertain whether endovascular therapy (which includes endovascular pharmacologic thrombolysis and, more recently, the use of stent retrievers) alone or combined with intravenous TPA is a more effective treatment of acute stroke than intravenous TPA alone, "the only proven reperfusion therapy for acute ischemic stroke."
In the study, conducted at 58 centers in the United States, Canada, Australia, and Europe, 434 patients were randomized to endovascular therapy plus intravenous TPA and 222 were randomized to standard treatment with intravenous TPA alone (started within 3 hours of stroke onset). The median age of those enrolled was 68-69 years (range, 23-89 years), a little over half were men, about 14% were black or Hispanic, and the median the National Institute of Health Stroke Scale (NIHSS) score was 16-17 (8-19 is a moderately severe stroke and 20 or greater is a severe stroke At the beginning of the study, only one thrombectomy device had been cleared by the Food and Drug Administration and, as the trial continued, other devices were used as they became cleared for use in the different countries.
In addition to the main finding, there were no differences in the primary outcome among those patients with an NIHSS score of 20 or more, and those with a score of 19 or lower, said the authors, who had hypothesized that endovascular therapy would have greater efficacy in patients with more-severe strokes since they "have the highest likelihood of occlusion in a major intracranial artery and the greatest volume of ischemic brain at risk."
They had also hypothesized that receiving endovascular therapy earlier would be associated with a greater benefit, but this was also not a significant factor in outcomes.
Mortality at 90 days was 19.1% in the endovascular therapy group and 21.6% in the intravenous TPA–alone group. Within 30 hours of TPA initiation, 6.2% of those on endovascular therapy and 5.9% of those on TPA alone had a symptomatic intracerebral hemorrhage. The differences in mortality at 7 days and in parenchymal hematoma rates were also not significantly different between the two groups. The rate of asymptomatic intracerebral hemorrhage, however, was significantly higher in the endovascular group.
Outcomes consistently trended better with combined therapy in patients with strokes involving larger artery occlusions and those with the shortest times from stroke onset to initiation of treatment, although because of small patient numbers the differences didn’t achieve statistical significance. These will be the subgroups that ought to be the focus of future clinical trials, Dr. Broderick said in a press briefing at the conference.
The underlying rationale for combined therapy is that intravenous TPA can quickly be started in the emergency department while the endovascular device therapy team is assembling, often at another hospital, which entails time-consuming patient transfer.
Intravenous TPA is the only proven therapy for acute ischemic stroke, but endovascular therapy is more effective at achieving recanalization. The study results bore this out: for example, the rate of partial or complete recanalization at 24 hours for an occlusion in the internal carotid artery was 81% with combined therapy compared to 35% with intravenous TPA alone. Yet this higher recanalization rate bore no clinical benefit, possibly because recanalization occurred too late, after ischemia had turned into infarction, Dr. Broderick explained.
"IMS III is going to be disappointing for a lot of people who are proponents of endovascular therapy. However, there is a light at the end of the tunnel in that there are these subgroups who may benefit," Dr. Brian Silver, who was not involved in the trial, said in an interview.
"The most critical feature is to treat the patients as soon as possible when they arrive in the emergency department, perhaps within 90 minutes. I think that’s the best chance for recovery. We are nowhere near what’s being done in cardiology, where there are door-to-balloon times of an hour. We need to do that in stroke. Since we're dealing with an organ that's more sensitive than the heart to ischemia, we probably need to be even faster than what's being done in cardiology. There is definitely room for improvement in our systems, perhaps by having the endovascular team stay in the hospital. Expense will be the limitation," according to Dr. Silver, director of the stroke center at Brown University, Providence, R.I.
IMS III investigator and interventional neuroradiologist Dr. Thomas A. Tomsick said in an interview that the study results won't change his own clinical practice.
"IMS III is by no means the final word on combined therapy. In Cincinnati tomorrow, if a patient with a large NIH Stroke Severity score shows up and we're treating him with IV TPA at 2 hours from stroke onset, we're not going to do a CT angiogram to evaluate that patient. He's going to the cath lab for angiography to see if there's a clot suitable for endovascular therapy," said Dr. Tomsick, professor of radiology at the University of Cincinnati.
Five different endovascular device therapies were utilized in IMS III. As new devices reached clinical practice, their use was allowed by investigators in order to keep the randomized trial clinically relevant. But recruitment for the study was slow because so many clinicians were already convinced by anecdotal experience that combined therapy is better. So the endovascular therapies used most frequently in IMS III aren't the ones widely used in clinical practice today. Major new randomized trials are now getting underway comparing combined therapy using state-of-the-art, more effective stent clot retriever devices to intravenous TPA alone, he added.
In the New England Journal of Medicine report, the authors noted that "the use of randomization in ongoing and future stroke trials, rather than the treatment of eligible patients with endovascular therapy outside any trial, and minimization of the time to treatment will be essential for assessing the potential benefit of endovascular therapy for acute ischemic stroke."
No matter how future trials of combined therapy turn out, endovascular therapy is not going away, Dr. Broderick observed.
"It's a very good tool. The reason why is there are patients who can't get TPA. For example, roughly 5% of patients who undergo coronary artery bypass surgery have a stroke. If you have somebody with a big stroke 2 days after having their chest cracked, you can't use TPA. In that case, those endovascular devices are the way we can get up in there and get rid of the clot," he explained.
In an editorial accompanying the report in the New England Journal of Medicine (doi: 10.1056/NEJMe1215730), Dr. Marc I. Chimowitz declared that the clinical implication of IMS III is that endovascular therapy remains unproven and intravenous TPA should continue to be the first-line treatment for patients with acute ischemic stroke within 4.5 hours after stroke onset.
While new clinical trials featuring more effective IV clot busters, such as tenecteplase, and next-generation endovascular devices are urgently needed in an effort to improve stroke outcomes, patient recruitment is likely to continue to be a challenge in the current environment. This could be overcome if Medicare were to place a moratorium on reimbursement for endovascular therapy of acute ischemic stroke except as part of a randomized trial, according to Dr. Chimowitz, professor of neurology at the Medical University of South Carolina, Charleston.
The study was supported with grants from NIH and the National Institute of Neurological Disorders and Stroke; and by Genentech (which supplied the TPA); and EKOS, Concentric Medical, and Cordis Neurovascular (which supplied catheters); and Actilyse (alteplase) manufacturer Boehringer Ingelheim (which, along with Genentech and EKOS, provided support for investigator meetings). Dr. Broderick disclosed consulting fees from PhotoThera. Of the 28 other authors, disclosures for 14 were listed and included having received consulting fees, grant support, and/or lecture fees from a variety of device and pharmaceutical companies that include Genentech. Dr. Chimowitz, Dr. Silver, and Dr. Tomsick reported having no financial conflicts.
The key to understanding the results of this study is the difference in the recanalization rates between the two groups and their lack of relationship to outcome. In the IV TPA plus catheter directed TPA group, the recanalization rate was higher than in the IV alone group, but the clinical outcomes were not better. Undoubtedly this is due to the fact that the recanalization was accomplished after brain tissue death had already occurred.
Hence the next and critical question is....what would the result be if we could administer the catheter directed TPA in a timely fashion, as is done with acute myocardial infarction? If the study had been performed in centers where this therapy was available without delay, would the results have been different? I think almost certainly. While it is true that this therapy is not available as widely as coronary interventions, it is critical for us to know whether patients who are fortunate enough to be treated in an institution where the therapy is available should receive it. Another disappointing aspect of the study design was the inability to perform subgroup analysis. While it appeared that patients with larger stroke distribution might benefit, the study was apparently not powered to detect this difference. Lastly, it is puzzling that the investigators themselves do not appear to believe the results of their own study, with two of them indicating the results would not change their own practice. If that's the case, why spend all this time and money designing a trial that doesn't answer the questions?
Dr. Cynthia K. Shortell is Professor and Chief, Division of Vascular Surgery, Duke University Medical Center, and an associate medical editor for Vascular Specialist.
The key to understanding the results of this study is the difference in the recanalization rates between the two groups and their lack of relationship to outcome. In the IV TPA plus catheter directed TPA group, the recanalization rate was higher than in the IV alone group, but the clinical outcomes were not better. Undoubtedly this is due to the fact that the recanalization was accomplished after brain tissue death had already occurred.
Hence the next and critical question is....what would the result be if we could administer the catheter directed TPA in a timely fashion, as is done with acute myocardial infarction? If the study had been performed in centers where this therapy was available without delay, would the results have been different? I think almost certainly. While it is true that this therapy is not available as widely as coronary interventions, it is critical for us to know whether patients who are fortunate enough to be treated in an institution where the therapy is available should receive it. Another disappointing aspect of the study design was the inability to perform subgroup analysis. While it appeared that patients with larger stroke distribution might benefit, the study was apparently not powered to detect this difference. Lastly, it is puzzling that the investigators themselves do not appear to believe the results of their own study, with two of them indicating the results would not change their own practice. If that's the case, why spend all this time and money designing a trial that doesn't answer the questions?
Dr. Cynthia K. Shortell is Professor and Chief, Division of Vascular Surgery, Duke University Medical Center, and an associate medical editor for Vascular Specialist.
The key to understanding the results of this study is the difference in the recanalization rates between the two groups and their lack of relationship to outcome. In the IV TPA plus catheter directed TPA group, the recanalization rate was higher than in the IV alone group, but the clinical outcomes were not better. Undoubtedly this is due to the fact that the recanalization was accomplished after brain tissue death had already occurred.
Hence the next and critical question is....what would the result be if we could administer the catheter directed TPA in a timely fashion, as is done with acute myocardial infarction? If the study had been performed in centers where this therapy was available without delay, would the results have been different? I think almost certainly. While it is true that this therapy is not available as widely as coronary interventions, it is critical for us to know whether patients who are fortunate enough to be treated in an institution where the therapy is available should receive it. Another disappointing aspect of the study design was the inability to perform subgroup analysis. While it appeared that patients with larger stroke distribution might benefit, the study was apparently not powered to detect this difference. Lastly, it is puzzling that the investigators themselves do not appear to believe the results of their own study, with two of them indicating the results would not change their own practice. If that's the case, why spend all this time and money designing a trial that doesn't answer the questions?
Dr. Cynthia K. Shortell is Professor and Chief, Division of Vascular Surgery, Duke University Medical Center, and an associate medical editor for Vascular Specialist.
Functional outcomes in patients treated with intravenous tissue plasminogen activator with or without endovascular therapy after a moderate to severe acute ischemic stroke were not significantly different, and safety outcomes were similar, in a study that was stopped early because of these results.
In the IMS (Interventional Management of Stroke) III study, 40.8% of patients randomized to receive endovascular therapy plus intravenous TPA met the primary endpoint, a measure of functional independence -- a modified Rankin score of 2 or less at 90 days -- compared with 38.7% among those who had intravenous TPA alone, a difference that was not statistically significant, reported Dr. Joseph Broderick of the University of Cincinnati Neuroscience Institute, and the other IMS III investigators.
Mortality and other safety outcomes were also not significantly different between the two groups of patients in the study, which was stopped early because of futility after 656 of the planned 900 patients had been randomized.
The study was published online to coincide with the presentation of the results at the International Stroke Conference (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1214300]).
Referring to the lack of randomized clinical trial data, the authors pointed out that it is uncertain whether endovascular therapy (which includes endovascular pharmacologic thrombolysis and, more recently, the use of stent retrievers) alone or combined with intravenous TPA is a more effective treatment of acute stroke than intravenous TPA alone, "the only proven reperfusion therapy for acute ischemic stroke."
In the study, conducted at 58 centers in the United States, Canada, Australia, and Europe, 434 patients were randomized to endovascular therapy plus intravenous TPA and 222 were randomized to standard treatment with intravenous TPA alone (started within 3 hours of stroke onset). The median age of those enrolled was 68-69 years (range, 23-89 years), a little over half were men, about 14% were black or Hispanic, and the median the National Institute of Health Stroke Scale (NIHSS) score was 16-17 (8-19 is a moderately severe stroke and 20 or greater is a severe stroke At the beginning of the study, only one thrombectomy device had been cleared by the Food and Drug Administration and, as the trial continued, other devices were used as they became cleared for use in the different countries.
In addition to the main finding, there were no differences in the primary outcome among those patients with an NIHSS score of 20 or more, and those with a score of 19 or lower, said the authors, who had hypothesized that endovascular therapy would have greater efficacy in patients with more-severe strokes since they "have the highest likelihood of occlusion in a major intracranial artery and the greatest volume of ischemic brain at risk."
They had also hypothesized that receiving endovascular therapy earlier would be associated with a greater benefit, but this was also not a significant factor in outcomes.
Mortality at 90 days was 19.1% in the endovascular therapy group and 21.6% in the intravenous TPA–alone group. Within 30 hours of TPA initiation, 6.2% of those on endovascular therapy and 5.9% of those on TPA alone had a symptomatic intracerebral hemorrhage. The differences in mortality at 7 days and in parenchymal hematoma rates were also not significantly different between the two groups. The rate of asymptomatic intracerebral hemorrhage, however, was significantly higher in the endovascular group.
Outcomes consistently trended better with combined therapy in patients with strokes involving larger artery occlusions and those with the shortest times from stroke onset to initiation of treatment, although because of small patient numbers the differences didn’t achieve statistical significance. These will be the subgroups that ought to be the focus of future clinical trials, Dr. Broderick said in a press briefing at the conference.
The underlying rationale for combined therapy is that intravenous TPA can quickly be started in the emergency department while the endovascular device therapy team is assembling, often at another hospital, which entails time-consuming patient transfer.
Intravenous TPA is the only proven therapy for acute ischemic stroke, but endovascular therapy is more effective at achieving recanalization. The study results bore this out: for example, the rate of partial or complete recanalization at 24 hours for an occlusion in the internal carotid artery was 81% with combined therapy compared to 35% with intravenous TPA alone. Yet this higher recanalization rate bore no clinical benefit, possibly because recanalization occurred too late, after ischemia had turned into infarction, Dr. Broderick explained.
"IMS III is going to be disappointing for a lot of people who are proponents of endovascular therapy. However, there is a light at the end of the tunnel in that there are these subgroups who may benefit," Dr. Brian Silver, who was not involved in the trial, said in an interview.
"The most critical feature is to treat the patients as soon as possible when they arrive in the emergency department, perhaps within 90 minutes. I think that’s the best chance for recovery. We are nowhere near what’s being done in cardiology, where there are door-to-balloon times of an hour. We need to do that in stroke. Since we're dealing with an organ that's more sensitive than the heart to ischemia, we probably need to be even faster than what's being done in cardiology. There is definitely room for improvement in our systems, perhaps by having the endovascular team stay in the hospital. Expense will be the limitation," according to Dr. Silver, director of the stroke center at Brown University, Providence, R.I.
IMS III investigator and interventional neuroradiologist Dr. Thomas A. Tomsick said in an interview that the study results won't change his own clinical practice.
"IMS III is by no means the final word on combined therapy. In Cincinnati tomorrow, if a patient with a large NIH Stroke Severity score shows up and we're treating him with IV TPA at 2 hours from stroke onset, we're not going to do a CT angiogram to evaluate that patient. He's going to the cath lab for angiography to see if there's a clot suitable for endovascular therapy," said Dr. Tomsick, professor of radiology at the University of Cincinnati.
Five different endovascular device therapies were utilized in IMS III. As new devices reached clinical practice, their use was allowed by investigators in order to keep the randomized trial clinically relevant. But recruitment for the study was slow because so many clinicians were already convinced by anecdotal experience that combined therapy is better. So the endovascular therapies used most frequently in IMS III aren't the ones widely used in clinical practice today. Major new randomized trials are now getting underway comparing combined therapy using state-of-the-art, more effective stent clot retriever devices to intravenous TPA alone, he added.
In the New England Journal of Medicine report, the authors noted that "the use of randomization in ongoing and future stroke trials, rather than the treatment of eligible patients with endovascular therapy outside any trial, and minimization of the time to treatment will be essential for assessing the potential benefit of endovascular therapy for acute ischemic stroke."
No matter how future trials of combined therapy turn out, endovascular therapy is not going away, Dr. Broderick observed.
"It's a very good tool. The reason why is there are patients who can't get TPA. For example, roughly 5% of patients who undergo coronary artery bypass surgery have a stroke. If you have somebody with a big stroke 2 days after having their chest cracked, you can't use TPA. In that case, those endovascular devices are the way we can get up in there and get rid of the clot," he explained.
In an editorial accompanying the report in the New England Journal of Medicine (doi: 10.1056/NEJMe1215730), Dr. Marc I. Chimowitz declared that the clinical implication of IMS III is that endovascular therapy remains unproven and intravenous TPA should continue to be the first-line treatment for patients with acute ischemic stroke within 4.5 hours after stroke onset.
While new clinical trials featuring more effective IV clot busters, such as tenecteplase, and next-generation endovascular devices are urgently needed in an effort to improve stroke outcomes, patient recruitment is likely to continue to be a challenge in the current environment. This could be overcome if Medicare were to place a moratorium on reimbursement for endovascular therapy of acute ischemic stroke except as part of a randomized trial, according to Dr. Chimowitz, professor of neurology at the Medical University of South Carolina, Charleston.
The study was supported with grants from NIH and the National Institute of Neurological Disorders and Stroke; and by Genentech (which supplied the TPA); and EKOS, Concentric Medical, and Cordis Neurovascular (which supplied catheters); and Actilyse (alteplase) manufacturer Boehringer Ingelheim (which, along with Genentech and EKOS, provided support for investigator meetings). Dr. Broderick disclosed consulting fees from PhotoThera. Of the 28 other authors, disclosures for 14 were listed and included having received consulting fees, grant support, and/or lecture fees from a variety of device and pharmaceutical companies that include Genentech. Dr. Chimowitz, Dr. Silver, and Dr. Tomsick reported having no financial conflicts.
Functional outcomes in patients treated with intravenous tissue plasminogen activator with or without endovascular therapy after a moderate to severe acute ischemic stroke were not significantly different, and safety outcomes were similar, in a study that was stopped early because of these results.
In the IMS (Interventional Management of Stroke) III study, 40.8% of patients randomized to receive endovascular therapy plus intravenous TPA met the primary endpoint, a measure of functional independence -- a modified Rankin score of 2 or less at 90 days -- compared with 38.7% among those who had intravenous TPA alone, a difference that was not statistically significant, reported Dr. Joseph Broderick of the University of Cincinnati Neuroscience Institute, and the other IMS III investigators.
Mortality and other safety outcomes were also not significantly different between the two groups of patients in the study, which was stopped early because of futility after 656 of the planned 900 patients had been randomized.
The study was published online to coincide with the presentation of the results at the International Stroke Conference (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1214300]).
Referring to the lack of randomized clinical trial data, the authors pointed out that it is uncertain whether endovascular therapy (which includes endovascular pharmacologic thrombolysis and, more recently, the use of stent retrievers) alone or combined with intravenous TPA is a more effective treatment of acute stroke than intravenous TPA alone, "the only proven reperfusion therapy for acute ischemic stroke."
In the study, conducted at 58 centers in the United States, Canada, Australia, and Europe, 434 patients were randomized to endovascular therapy plus intravenous TPA and 222 were randomized to standard treatment with intravenous TPA alone (started within 3 hours of stroke onset). The median age of those enrolled was 68-69 years (range, 23-89 years), a little over half were men, about 14% were black or Hispanic, and the median the National Institute of Health Stroke Scale (NIHSS) score was 16-17 (8-19 is a moderately severe stroke and 20 or greater is a severe stroke At the beginning of the study, only one thrombectomy device had been cleared by the Food and Drug Administration and, as the trial continued, other devices were used as they became cleared for use in the different countries.
In addition to the main finding, there were no differences in the primary outcome among those patients with an NIHSS score of 20 or more, and those with a score of 19 or lower, said the authors, who had hypothesized that endovascular therapy would have greater efficacy in patients with more-severe strokes since they "have the highest likelihood of occlusion in a major intracranial artery and the greatest volume of ischemic brain at risk."
They had also hypothesized that receiving endovascular therapy earlier would be associated with a greater benefit, but this was also not a significant factor in outcomes.
Mortality at 90 days was 19.1% in the endovascular therapy group and 21.6% in the intravenous TPA–alone group. Within 30 hours of TPA initiation, 6.2% of those on endovascular therapy and 5.9% of those on TPA alone had a symptomatic intracerebral hemorrhage. The differences in mortality at 7 days and in parenchymal hematoma rates were also not significantly different between the two groups. The rate of asymptomatic intracerebral hemorrhage, however, was significantly higher in the endovascular group.
Outcomes consistently trended better with combined therapy in patients with strokes involving larger artery occlusions and those with the shortest times from stroke onset to initiation of treatment, although because of small patient numbers the differences didn’t achieve statistical significance. These will be the subgroups that ought to be the focus of future clinical trials, Dr. Broderick said in a press briefing at the conference.
The underlying rationale for combined therapy is that intravenous TPA can quickly be started in the emergency department while the endovascular device therapy team is assembling, often at another hospital, which entails time-consuming patient transfer.
Intravenous TPA is the only proven therapy for acute ischemic stroke, but endovascular therapy is more effective at achieving recanalization. The study results bore this out: for example, the rate of partial or complete recanalization at 24 hours for an occlusion in the internal carotid artery was 81% with combined therapy compared to 35% with intravenous TPA alone. Yet this higher recanalization rate bore no clinical benefit, possibly because recanalization occurred too late, after ischemia had turned into infarction, Dr. Broderick explained.
"IMS III is going to be disappointing for a lot of people who are proponents of endovascular therapy. However, there is a light at the end of the tunnel in that there are these subgroups who may benefit," Dr. Brian Silver, who was not involved in the trial, said in an interview.
"The most critical feature is to treat the patients as soon as possible when they arrive in the emergency department, perhaps within 90 minutes. I think that’s the best chance for recovery. We are nowhere near what’s being done in cardiology, where there are door-to-balloon times of an hour. We need to do that in stroke. Since we're dealing with an organ that's more sensitive than the heart to ischemia, we probably need to be even faster than what's being done in cardiology. There is definitely room for improvement in our systems, perhaps by having the endovascular team stay in the hospital. Expense will be the limitation," according to Dr. Silver, director of the stroke center at Brown University, Providence, R.I.
IMS III investigator and interventional neuroradiologist Dr. Thomas A. Tomsick said in an interview that the study results won't change his own clinical practice.
"IMS III is by no means the final word on combined therapy. In Cincinnati tomorrow, if a patient with a large NIH Stroke Severity score shows up and we're treating him with IV TPA at 2 hours from stroke onset, we're not going to do a CT angiogram to evaluate that patient. He's going to the cath lab for angiography to see if there's a clot suitable for endovascular therapy," said Dr. Tomsick, professor of radiology at the University of Cincinnati.
Five different endovascular device therapies were utilized in IMS III. As new devices reached clinical practice, their use was allowed by investigators in order to keep the randomized trial clinically relevant. But recruitment for the study was slow because so many clinicians were already convinced by anecdotal experience that combined therapy is better. So the endovascular therapies used most frequently in IMS III aren't the ones widely used in clinical practice today. Major new randomized trials are now getting underway comparing combined therapy using state-of-the-art, more effective stent clot retriever devices to intravenous TPA alone, he added.
In the New England Journal of Medicine report, the authors noted that "the use of randomization in ongoing and future stroke trials, rather than the treatment of eligible patients with endovascular therapy outside any trial, and minimization of the time to treatment will be essential for assessing the potential benefit of endovascular therapy for acute ischemic stroke."
No matter how future trials of combined therapy turn out, endovascular therapy is not going away, Dr. Broderick observed.
"It's a very good tool. The reason why is there are patients who can't get TPA. For example, roughly 5% of patients who undergo coronary artery bypass surgery have a stroke. If you have somebody with a big stroke 2 days after having their chest cracked, you can't use TPA. In that case, those endovascular devices are the way we can get up in there and get rid of the clot," he explained.
In an editorial accompanying the report in the New England Journal of Medicine (doi: 10.1056/NEJMe1215730), Dr. Marc I. Chimowitz declared that the clinical implication of IMS III is that endovascular therapy remains unproven and intravenous TPA should continue to be the first-line treatment for patients with acute ischemic stroke within 4.5 hours after stroke onset.
While new clinical trials featuring more effective IV clot busters, such as tenecteplase, and next-generation endovascular devices are urgently needed in an effort to improve stroke outcomes, patient recruitment is likely to continue to be a challenge in the current environment. This could be overcome if Medicare were to place a moratorium on reimbursement for endovascular therapy of acute ischemic stroke except as part of a randomized trial, according to Dr. Chimowitz, professor of neurology at the Medical University of South Carolina, Charleston.
The study was supported with grants from NIH and the National Institute of Neurological Disorders and Stroke; and by Genentech (which supplied the TPA); and EKOS, Concentric Medical, and Cordis Neurovascular (which supplied catheters); and Actilyse (alteplase) manufacturer Boehringer Ingelheim (which, along with Genentech and EKOS, provided support for investigator meetings). Dr. Broderick disclosed consulting fees from PhotoThera. Of the 28 other authors, disclosures for 14 were listed and included having received consulting fees, grant support, and/or lecture fees from a variety of device and pharmaceutical companies that include Genentech. Dr. Chimowitz, Dr. Silver, and Dr. Tomsick reported having no financial conflicts.
FROM THE INTERNATIONAL STROKE CONFERENCE
Major Finding: Endovascular therapy plus intravenous TPA showed no added outcome benefits, compared with TPA alone, with 40.8% and 38.7% of patients, respectively, reaching functional independence at 90 days.
Data Source: An international, phase III study of 656 patients with an acute moderate to severe ischemic stroke randomized 2:1 to IV TPA with endovascular therapy or IV TPA alone.
Disclosures: The study was supported with grants from NIH and the National Institute of Neurological Disorders and Stroke, and by Genentech (supplier of TPA), and EKOS, Concentric Medical, and Cordis Neurovascular (which supplied catheters). Dr. Broderick disclosed consulting fees from PhotoThera. Of the 28 other authors, disclosures for 14 were listed and included having received consulting fees, grant support, and/or lecture fees from a variety of device and pharmaceutical companies that included Genentech. Dr. Chimowitz, Dr. Silver, and Dr. Tomsick reported having no financial conflicts.
Elevated glucose after colorectal surgery spells trouble
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.
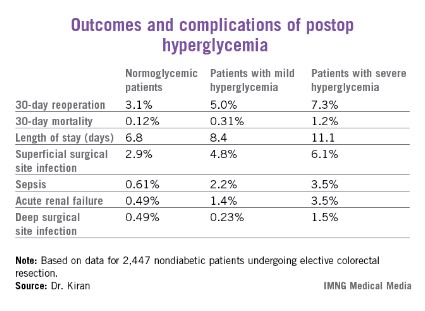
He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.

He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.

He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
AT THE ASA ANNUAL MEETING
Major finding: Mild hyperglycemia occurring within 48 hours after major colorectal surgery in nondiabetic patients was independently associated with a doubled risk of reoperation within 30 days and a 7.9-fold increase in sepsis, compared with patients who remained normoglycemic. Severe hyperglycemia – a blood glucose measurement in excess of 200 mg/dL – was associated with even worse outcomes.
Data source: A single-center study of more than 16,000 postoperative blood glucose measurements in 2,628 consecutive patients who underwent colorectal resection.
Disclosures: The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
Where Belotero Balance excels in cosmetic dermatology
MAUI, HAWAII – Belotero Balance, the most recent filler to receive Food and Drug Administration approval, offers unique advantages over other hyaluronic acid fillers in treatment of superficial fine lines and wrinkles in the perioral area, according to Dr. Mark G. Rubin, a dermatologist in Beverly Hills, Calif.
"This product has become my collagen replacement. It’s a filler you want to inject into the dermis. It enables dermatologists to go after very superficial thin lines without causing lumpiness, ridges, or the Tyndall effect," he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Be aware, though, that this is not a product to use straight out of the box. Unlike the older, widely used hyaluronic acid fillers Restylane and Juvederm, it doesn’t come premixed with anesthetic, so patients will find that Belotero injected without first being diluted with lidocaine hurts more than other fillers.
The other reason Dr. Rubin dilutes Belotero is that even though it is thinner and softer than other hyaluronic acid fillers, it can still result in unsightly ridges lasting for several weeks if injected intradermally in too strong a concentration.
For treatment of thin perioral lines, he typically dilutes 1 cc of Belotero with 0.5 cc of 1% lidocaine with epinephrine. He favors a serial puncture technique, although some other cosmetic dermatologists prefer a linear threading strategy.
"This behaves a lot like Zyderm did. It can be injected superficially and still give people significant improvement in very fine lines," said Dr. Rubin, also of the University of California, San Diego.
Zyderm and other collagen implants were taken off the U.S. market by FDA request. Cosmetic dermatologists have been looking for a suitable replacement thin filler for superficial use ever since – and Belotero is it, he said.
To treat deeper folds and contour irregularities, Dr. Rubin will dilute 1 cc of Belotero with 0.3 cc of 1% lidocaine with epinephrine, which provides more lift. Sometimes he’ll partially fill in a deeper perioral line with the less dilute product before topping it off with the more dilute version in the same treatment session.
Belotero Balance is a non-animal–derived hyaluronic acid–based cohesive gel that is crosslinked twice. Its clinical behavior is significantly different from the other hyaluronic acid products; it’s stretchy, like a rubber band, while the earlier-generation hyaluronic acid fillers tend to break when stretched, the dermatologist explained.
"It’s like an amoeba, working its way between cells and fibers. It really does smooth out better than the hyaluronic acids we’re used to," he continued.
On the other hand, Belotero provides less lift when injected deep than the other hyaluronic acid fillers.
"It’s not a go-to product for volume replacement. It requires more product to achieve a similar result. I still use a lot of Restylane and Juvederm, particularly where I’m looking for volume," Dr. Rubin said.
The FDA approval of Belotero Balance was based upon a phase III study of 118 patients. In contrast, Dr. Rubin has injected more than 1,300 syringes of Belotero into his patients. He offered several tips based upon his experience:
– Don’t overcorrect. "Correct to 100%. Because of the dilution, a week or two later the patient will no longer be filled all the way and may need a second treatment," Dr. Rubin said. "I tell patients who come in for superficial wrinkle therapy that they’re going to be treated twice. They’ll have a good result the first time and they’ll have a great result the second time when they come in a couple weeks later."
– Massage the treated area vigorously right after the injection to help smooth out the product. "I’ll stick my finger inside their cheek and squash them hard for 5 seconds," he said.
– Expect the results to last at least 6 months before beginning to fade.
– Anticipate a day or two of beadiness or ridging until the Belotero settles into the skin. Occasionally this can take up to 5 or 6 days.
– Consider prescribing an antihistamine if the ridging and edema lasts more than about 5 days. "There is some type of a histamine-mediated response in those patients. It almost looks urticarial. It’s a small group, probably no greater than 5%-10% of the people I inject, who have this persistent ridginess and puffiness," Dr. Rubin said.
Dr. Rubin reported serving as a paid researcher and consultant for Medicis, but has no financial relationship with Merz, which markets Belotero Balance. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Belotero Balance, the most recent filler to receive Food and Drug Administration approval, offers unique advantages over other hyaluronic acid fillers in treatment of superficial fine lines and wrinkles in the perioral area, according to Dr. Mark G. Rubin, a dermatologist in Beverly Hills, Calif.
"This product has become my collagen replacement. It’s a filler you want to inject into the dermis. It enables dermatologists to go after very superficial thin lines without causing lumpiness, ridges, or the Tyndall effect," he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Be aware, though, that this is not a product to use straight out of the box. Unlike the older, widely used hyaluronic acid fillers Restylane and Juvederm, it doesn’t come premixed with anesthetic, so patients will find that Belotero injected without first being diluted with lidocaine hurts more than other fillers.
The other reason Dr. Rubin dilutes Belotero is that even though it is thinner and softer than other hyaluronic acid fillers, it can still result in unsightly ridges lasting for several weeks if injected intradermally in too strong a concentration.
For treatment of thin perioral lines, he typically dilutes 1 cc of Belotero with 0.5 cc of 1% lidocaine with epinephrine. He favors a serial puncture technique, although some other cosmetic dermatologists prefer a linear threading strategy.
"This behaves a lot like Zyderm did. It can be injected superficially and still give people significant improvement in very fine lines," said Dr. Rubin, also of the University of California, San Diego.
Zyderm and other collagen implants were taken off the U.S. market by FDA request. Cosmetic dermatologists have been looking for a suitable replacement thin filler for superficial use ever since – and Belotero is it, he said.
To treat deeper folds and contour irregularities, Dr. Rubin will dilute 1 cc of Belotero with 0.3 cc of 1% lidocaine with epinephrine, which provides more lift. Sometimes he’ll partially fill in a deeper perioral line with the less dilute product before topping it off with the more dilute version in the same treatment session.
Belotero Balance is a non-animal–derived hyaluronic acid–based cohesive gel that is crosslinked twice. Its clinical behavior is significantly different from the other hyaluronic acid products; it’s stretchy, like a rubber band, while the earlier-generation hyaluronic acid fillers tend to break when stretched, the dermatologist explained.
"It’s like an amoeba, working its way between cells and fibers. It really does smooth out better than the hyaluronic acids we’re used to," he continued.
On the other hand, Belotero provides less lift when injected deep than the other hyaluronic acid fillers.
"It’s not a go-to product for volume replacement. It requires more product to achieve a similar result. I still use a lot of Restylane and Juvederm, particularly where I’m looking for volume," Dr. Rubin said.
The FDA approval of Belotero Balance was based upon a phase III study of 118 patients. In contrast, Dr. Rubin has injected more than 1,300 syringes of Belotero into his patients. He offered several tips based upon his experience:
– Don’t overcorrect. "Correct to 100%. Because of the dilution, a week or two later the patient will no longer be filled all the way and may need a second treatment," Dr. Rubin said. "I tell patients who come in for superficial wrinkle therapy that they’re going to be treated twice. They’ll have a good result the first time and they’ll have a great result the second time when they come in a couple weeks later."
– Massage the treated area vigorously right after the injection to help smooth out the product. "I’ll stick my finger inside their cheek and squash them hard for 5 seconds," he said.
– Expect the results to last at least 6 months before beginning to fade.
– Anticipate a day or two of beadiness or ridging until the Belotero settles into the skin. Occasionally this can take up to 5 or 6 days.
– Consider prescribing an antihistamine if the ridging and edema lasts more than about 5 days. "There is some type of a histamine-mediated response in those patients. It almost looks urticarial. It’s a small group, probably no greater than 5%-10% of the people I inject, who have this persistent ridginess and puffiness," Dr. Rubin said.
Dr. Rubin reported serving as a paid researcher and consultant for Medicis, but has no financial relationship with Merz, which markets Belotero Balance. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – Belotero Balance, the most recent filler to receive Food and Drug Administration approval, offers unique advantages over other hyaluronic acid fillers in treatment of superficial fine lines and wrinkles in the perioral area, according to Dr. Mark G. Rubin, a dermatologist in Beverly Hills, Calif.
"This product has become my collagen replacement. It’s a filler you want to inject into the dermis. It enables dermatologists to go after very superficial thin lines without causing lumpiness, ridges, or the Tyndall effect," he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Be aware, though, that this is not a product to use straight out of the box. Unlike the older, widely used hyaluronic acid fillers Restylane and Juvederm, it doesn’t come premixed with anesthetic, so patients will find that Belotero injected without first being diluted with lidocaine hurts more than other fillers.
The other reason Dr. Rubin dilutes Belotero is that even though it is thinner and softer than other hyaluronic acid fillers, it can still result in unsightly ridges lasting for several weeks if injected intradermally in too strong a concentration.
For treatment of thin perioral lines, he typically dilutes 1 cc of Belotero with 0.5 cc of 1% lidocaine with epinephrine. He favors a serial puncture technique, although some other cosmetic dermatologists prefer a linear threading strategy.
"This behaves a lot like Zyderm did. It can be injected superficially and still give people significant improvement in very fine lines," said Dr. Rubin, also of the University of California, San Diego.
Zyderm and other collagen implants were taken off the U.S. market by FDA request. Cosmetic dermatologists have been looking for a suitable replacement thin filler for superficial use ever since – and Belotero is it, he said.
To treat deeper folds and contour irregularities, Dr. Rubin will dilute 1 cc of Belotero with 0.3 cc of 1% lidocaine with epinephrine, which provides more lift. Sometimes he’ll partially fill in a deeper perioral line with the less dilute product before topping it off with the more dilute version in the same treatment session.
Belotero Balance is a non-animal–derived hyaluronic acid–based cohesive gel that is crosslinked twice. Its clinical behavior is significantly different from the other hyaluronic acid products; it’s stretchy, like a rubber band, while the earlier-generation hyaluronic acid fillers tend to break when stretched, the dermatologist explained.
"It’s like an amoeba, working its way between cells and fibers. It really does smooth out better than the hyaluronic acids we’re used to," he continued.
On the other hand, Belotero provides less lift when injected deep than the other hyaluronic acid fillers.
"It’s not a go-to product for volume replacement. It requires more product to achieve a similar result. I still use a lot of Restylane and Juvederm, particularly where I’m looking for volume," Dr. Rubin said.
The FDA approval of Belotero Balance was based upon a phase III study of 118 patients. In contrast, Dr. Rubin has injected more than 1,300 syringes of Belotero into his patients. He offered several tips based upon his experience:
– Don’t overcorrect. "Correct to 100%. Because of the dilution, a week or two later the patient will no longer be filled all the way and may need a second treatment," Dr. Rubin said. "I tell patients who come in for superficial wrinkle therapy that they’re going to be treated twice. They’ll have a good result the first time and they’ll have a great result the second time when they come in a couple weeks later."
– Massage the treated area vigorously right after the injection to help smooth out the product. "I’ll stick my finger inside their cheek and squash them hard for 5 seconds," he said.
– Expect the results to last at least 6 months before beginning to fade.
– Anticipate a day or two of beadiness or ridging until the Belotero settles into the skin. Occasionally this can take up to 5 or 6 days.
– Consider prescribing an antihistamine if the ridging and edema lasts more than about 5 days. "There is some type of a histamine-mediated response in those patients. It almost looks urticarial. It’s a small group, probably no greater than 5%-10% of the people I inject, who have this persistent ridginess and puffiness," Dr. Rubin said.
Dr. Rubin reported serving as a paid researcher and consultant for Medicis, but has no financial relationship with Merz, which markets Belotero Balance. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Be mindful of psoriasis/cutaneous T-cell lymphoma link
MAUI, HAWAII – The strong association between psoriasis and cutaneous T-cell lymphoma warrants a low threshold for skin biopsy in psoriasis patients, particularly since the two diseases can be tough to differentiate, experts emphasized at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Indeed, skin biopsy is appropriate in any psoriasis patient with atypical features or psoriasis that isn’t responding to treatment, according to Dr. Joel M. Gelfand of the University of Pennsylvania, Philadelphia.
"I’ve found that my psoriasis patients actually like to be biopsied. They’ve been living with their disease for an average of 20 years, and they’re curious," he said. "And they’re also suspicious. They wonder, ‘How could I have this for 20 years and it’s still here?’ They’re worried that it’s really cancer or something else. So my patients never decline a biopsy of psoriasis," he said.
The literature on psoriasis and solid malignancies is "a mess," with some observational studies showing a positive association and others not, Dr. Gelfand noted.
In contrast, the studies looking at psoriasis and cutaneous T-cell lymphoma (CTCL) have been consistently positive. Case in point: when Dr. Gelfand and colleagues turned to the U.K. General Practice Research Database to study more than 153,000 psoriasis patients and nearly 800,000 controls, they found participants with mild psoriasis had a 4.1-fold increased relative risk of developing CTCL, while those with severe psoriasis had a 10.75-fold increased risk (J. Invest. Dermatol. 2006;126:2194-201).
One limitation of large, observational studies is that it’s hard to know whether some psoriasis patients who developed CTCL were initially misdiagnosed and actually had CTCL all along, Dr. Gelfand said.
"We have a large lymphoma study group at Penn, and we see all kinds: we see some patients who clearly had psoriasis for many years and then developed erythrodermic CTCL, and we have people who clearly were misdiagnosed as having psoriasis," he said.
Whether the increased lymphoma risk is caused by psoriasis itself, as opposed to the chronic use of immunosuppressive drugs employed in treating the skin disease, remains uncertain. To date, the studies are mostly reassuring that the psoriasis medications don’t significantly increase the risk. One thing that is clear, however, is that CTCL can progress very rapidly in a patient who is on potent immunosuppressive therapy for what is mistakenly thought to be psoriasis.
"The patient with tumor-stage mycosis fungoides is not a person you want to hit with a TNF inhibitor," Dr. Gelfand observed.
The distinction between severe psoriasis and CTCL can be tricky, according to veteran psoriasis researcher Dr. Craig L. Leonardi of Saint Louis University.
"In the last 13 years, while we’ve been developing the biologic agents, every now and then we’ll get a patient in a research trial who actually has CTCL. This is in an environment where patients are being examined closely, and yet the clinician is totally fooled by it," Dr. Leonardi said. "These drugs have evolved to the point that we get PASI-75 responses in the 70%-85% range. Most patients will have a rocking experience on these medications. So it’s a good general rule that if you find somebody who doesn’t respond, it’s time to start thinking about other diagnoses."
When Dr. Gelfand’s suspicions are raised, he typically biopsies one or two representative lesions after the patient has been off of topical steroids for at least a week, since the medication can reduce the epidermal infiltrate.
Be cautious if the histologic specimen is going to be read by a general pathologist. The situation is fairly straightforward if the report comes back as unequivocally psoriasis or CTCL. But if the report says, ‘psoriasiform dermatitis NOS’ it’s appropriate to insist that a dermatopathologist takes a look at the slides, Dr. Gelfand said.
"A lot of malpractice cases come across my desk, and ‘failure to diagnose’ is always a big one," said Dr. Guy F. Webster of Thomas Jefferson University, Philadelphia.
"It’s not always melanoma, either," he continued. "The story is always that somebody had a rash for a long time. It looked like eczema, psoriasis, whatever. And the physician treated it without ever questioning the diagnosis when the patient didn’t get better."
Dr. Alan Menter noted convincing evidence that, at pretreatment baseline, psoriasis patients, like those with rheumatoid arthritis, have a slightly increased risk of lymphoma.
"It’s kind of sensitive to talk to patients about the cancer issue. They all ask, ‘Is there a chance you’ll cause cancer by giving me this biologic drug?’ You have to gently tell them they are already at slightly increased risk," Dr. Menter said.
"Is the use of an anti-TNF agent going to increase that risk? The answer is that in all of the more than 2 million patients who’ve been treated with TNF inhibitors, there does not appear to be a statistical increase above the baseline risk," said Dr. Menter of Baylor University Medical Center, Dallas, and chair of the American Academy of Dermatology psoriasis guidelines committee.
SDEF and this news organization are owned by the same parent company. Dr. Menter, Dr. Gelfand, Dr. Webster, and Dr. Leonardi have received research funds and/or served as consultants to numerous pharmaceutical companies.
MAUI, HAWAII – The strong association between psoriasis and cutaneous T-cell lymphoma warrants a low threshold for skin biopsy in psoriasis patients, particularly since the two diseases can be tough to differentiate, experts emphasized at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Indeed, skin biopsy is appropriate in any psoriasis patient with atypical features or psoriasis that isn’t responding to treatment, according to Dr. Joel M. Gelfand of the University of Pennsylvania, Philadelphia.
"I’ve found that my psoriasis patients actually like to be biopsied. They’ve been living with their disease for an average of 20 years, and they’re curious," he said. "And they’re also suspicious. They wonder, ‘How could I have this for 20 years and it’s still here?’ They’re worried that it’s really cancer or something else. So my patients never decline a biopsy of psoriasis," he said.
The literature on psoriasis and solid malignancies is "a mess," with some observational studies showing a positive association and others not, Dr. Gelfand noted.
In contrast, the studies looking at psoriasis and cutaneous T-cell lymphoma (CTCL) have been consistently positive. Case in point: when Dr. Gelfand and colleagues turned to the U.K. General Practice Research Database to study more than 153,000 psoriasis patients and nearly 800,000 controls, they found participants with mild psoriasis had a 4.1-fold increased relative risk of developing CTCL, while those with severe psoriasis had a 10.75-fold increased risk (J. Invest. Dermatol. 2006;126:2194-201).
One limitation of large, observational studies is that it’s hard to know whether some psoriasis patients who developed CTCL were initially misdiagnosed and actually had CTCL all along, Dr. Gelfand said.
"We have a large lymphoma study group at Penn, and we see all kinds: we see some patients who clearly had psoriasis for many years and then developed erythrodermic CTCL, and we have people who clearly were misdiagnosed as having psoriasis," he said.
Whether the increased lymphoma risk is caused by psoriasis itself, as opposed to the chronic use of immunosuppressive drugs employed in treating the skin disease, remains uncertain. To date, the studies are mostly reassuring that the psoriasis medications don’t significantly increase the risk. One thing that is clear, however, is that CTCL can progress very rapidly in a patient who is on potent immunosuppressive therapy for what is mistakenly thought to be psoriasis.
"The patient with tumor-stage mycosis fungoides is not a person you want to hit with a TNF inhibitor," Dr. Gelfand observed.
The distinction between severe psoriasis and CTCL can be tricky, according to veteran psoriasis researcher Dr. Craig L. Leonardi of Saint Louis University.
"In the last 13 years, while we’ve been developing the biologic agents, every now and then we’ll get a patient in a research trial who actually has CTCL. This is in an environment where patients are being examined closely, and yet the clinician is totally fooled by it," Dr. Leonardi said. "These drugs have evolved to the point that we get PASI-75 responses in the 70%-85% range. Most patients will have a rocking experience on these medications. So it’s a good general rule that if you find somebody who doesn’t respond, it’s time to start thinking about other diagnoses."
When Dr. Gelfand’s suspicions are raised, he typically biopsies one or two representative lesions after the patient has been off of topical steroids for at least a week, since the medication can reduce the epidermal infiltrate.
Be cautious if the histologic specimen is going to be read by a general pathologist. The situation is fairly straightforward if the report comes back as unequivocally psoriasis or CTCL. But if the report says, ‘psoriasiform dermatitis NOS’ it’s appropriate to insist that a dermatopathologist takes a look at the slides, Dr. Gelfand said.
"A lot of malpractice cases come across my desk, and ‘failure to diagnose’ is always a big one," said Dr. Guy F. Webster of Thomas Jefferson University, Philadelphia.
"It’s not always melanoma, either," he continued. "The story is always that somebody had a rash for a long time. It looked like eczema, psoriasis, whatever. And the physician treated it without ever questioning the diagnosis when the patient didn’t get better."
Dr. Alan Menter noted convincing evidence that, at pretreatment baseline, psoriasis patients, like those with rheumatoid arthritis, have a slightly increased risk of lymphoma.
"It’s kind of sensitive to talk to patients about the cancer issue. They all ask, ‘Is there a chance you’ll cause cancer by giving me this biologic drug?’ You have to gently tell them they are already at slightly increased risk," Dr. Menter said.
"Is the use of an anti-TNF agent going to increase that risk? The answer is that in all of the more than 2 million patients who’ve been treated with TNF inhibitors, there does not appear to be a statistical increase above the baseline risk," said Dr. Menter of Baylor University Medical Center, Dallas, and chair of the American Academy of Dermatology psoriasis guidelines committee.
SDEF and this news organization are owned by the same parent company. Dr. Menter, Dr. Gelfand, Dr. Webster, and Dr. Leonardi have received research funds and/or served as consultants to numerous pharmaceutical companies.
MAUI, HAWAII – The strong association between psoriasis and cutaneous T-cell lymphoma warrants a low threshold for skin biopsy in psoriasis patients, particularly since the two diseases can be tough to differentiate, experts emphasized at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Indeed, skin biopsy is appropriate in any psoriasis patient with atypical features or psoriasis that isn’t responding to treatment, according to Dr. Joel M. Gelfand of the University of Pennsylvania, Philadelphia.
"I’ve found that my psoriasis patients actually like to be biopsied. They’ve been living with their disease for an average of 20 years, and they’re curious," he said. "And they’re also suspicious. They wonder, ‘How could I have this for 20 years and it’s still here?’ They’re worried that it’s really cancer or something else. So my patients never decline a biopsy of psoriasis," he said.
The literature on psoriasis and solid malignancies is "a mess," with some observational studies showing a positive association and others not, Dr. Gelfand noted.
In contrast, the studies looking at psoriasis and cutaneous T-cell lymphoma (CTCL) have been consistently positive. Case in point: when Dr. Gelfand and colleagues turned to the U.K. General Practice Research Database to study more than 153,000 psoriasis patients and nearly 800,000 controls, they found participants with mild psoriasis had a 4.1-fold increased relative risk of developing CTCL, while those with severe psoriasis had a 10.75-fold increased risk (J. Invest. Dermatol. 2006;126:2194-201).
One limitation of large, observational studies is that it’s hard to know whether some psoriasis patients who developed CTCL were initially misdiagnosed and actually had CTCL all along, Dr. Gelfand said.
"We have a large lymphoma study group at Penn, and we see all kinds: we see some patients who clearly had psoriasis for many years and then developed erythrodermic CTCL, and we have people who clearly were misdiagnosed as having psoriasis," he said.
Whether the increased lymphoma risk is caused by psoriasis itself, as opposed to the chronic use of immunosuppressive drugs employed in treating the skin disease, remains uncertain. To date, the studies are mostly reassuring that the psoriasis medications don’t significantly increase the risk. One thing that is clear, however, is that CTCL can progress very rapidly in a patient who is on potent immunosuppressive therapy for what is mistakenly thought to be psoriasis.
"The patient with tumor-stage mycosis fungoides is not a person you want to hit with a TNF inhibitor," Dr. Gelfand observed.
The distinction between severe psoriasis and CTCL can be tricky, according to veteran psoriasis researcher Dr. Craig L. Leonardi of Saint Louis University.
"In the last 13 years, while we’ve been developing the biologic agents, every now and then we’ll get a patient in a research trial who actually has CTCL. This is in an environment where patients are being examined closely, and yet the clinician is totally fooled by it," Dr. Leonardi said. "These drugs have evolved to the point that we get PASI-75 responses in the 70%-85% range. Most patients will have a rocking experience on these medications. So it’s a good general rule that if you find somebody who doesn’t respond, it’s time to start thinking about other diagnoses."
When Dr. Gelfand’s suspicions are raised, he typically biopsies one or two representative lesions after the patient has been off of topical steroids for at least a week, since the medication can reduce the epidermal infiltrate.
Be cautious if the histologic specimen is going to be read by a general pathologist. The situation is fairly straightforward if the report comes back as unequivocally psoriasis or CTCL. But if the report says, ‘psoriasiform dermatitis NOS’ it’s appropriate to insist that a dermatopathologist takes a look at the slides, Dr. Gelfand said.
"A lot of malpractice cases come across my desk, and ‘failure to diagnose’ is always a big one," said Dr. Guy F. Webster of Thomas Jefferson University, Philadelphia.
"It’s not always melanoma, either," he continued. "The story is always that somebody had a rash for a long time. It looked like eczema, psoriasis, whatever. And the physician treated it without ever questioning the diagnosis when the patient didn’t get better."
Dr. Alan Menter noted convincing evidence that, at pretreatment baseline, psoriasis patients, like those with rheumatoid arthritis, have a slightly increased risk of lymphoma.
"It’s kind of sensitive to talk to patients about the cancer issue. They all ask, ‘Is there a chance you’ll cause cancer by giving me this biologic drug?’ You have to gently tell them they are already at slightly increased risk," Dr. Menter said.
"Is the use of an anti-TNF agent going to increase that risk? The answer is that in all of the more than 2 million patients who’ve been treated with TNF inhibitors, there does not appear to be a statistical increase above the baseline risk," said Dr. Menter of Baylor University Medical Center, Dallas, and chair of the American Academy of Dermatology psoriasis guidelines committee.
SDEF and this news organization are owned by the same parent company. Dr. Menter, Dr. Gelfand, Dr. Webster, and Dr. Leonardi have received research funds and/or served as consultants to numerous pharmaceutical companies.
EXPERT OPINION FROM SDEF HAWAII DERMATOLOGY SEMINAR