User login
Surgeons tackle readmission risk reduction
INDIANAPOLIS – Average 30-day readmission rates in a large national study varied widely by surgical specialty, ranging from 5% for general surgery patients to 12% for vascular surgery patients and 16% after hepato-pancreatic-biliary surgery.
This retrospective study was based upon American College of Surgeons National Surgical Quality Improvement Program (NSQIP) 2011 data on 240,125 patients discharged from 316 hospitals after these types of surgery. The results enabled investigators to identify the major risk factors for readmission. They used this information to generate a predictive model to identify patients at greatest risk with an eye toward introducing interventions to keep them out of the hospital, Dr. Timothy M. Pawlik said at the annual meeting of the American Surgical Association.
How effective such interventions will be in a surgical population is as yet unclear, he added.
The importance of this work lies in the growing emphasis health care payers are placing upon 30-day readmission as a quality-of-care indicator. In 2012, Medicare began cutting reimbursement by 1% to hospitals with above-average 30-day readmissions. Next year, this penalty is scheduled to increase to 3% under the Medicare Hospital Readmission Reduction Program. Prior research efforts to identify risk factors for readmission have focused chiefly on medical rather than surgical conditions, noted Dr. Pawlik, professor of surgery and chief of the division of surgical oncology at Johns Hopkins University, Baltimore.
The readmission risk predictive formula he and his coinvestigators developed is simple: American Society of Anesthesiologists physical status class + (length of stay/2). This number is rounded up to yield a risk score of 1-10. A patient with a readmission score of 1 has a 1% risk of readmission within 30 days of discharge. The risk climbs to 12% with a score of 5, 20% with a score of 8, and 40% with a score of 10.
"A score of 4 had an 8% readmission rate, with 77% sensitivity, 52% specificity, a low positive predictive value of 12%, but a high negative predictive value of 95%," the surgeon observed. "In essence, a score of 4 or higher was able to identify 80% of all readmissions, but it also included about half of all patients."
Because of a quirk in the NSQIP database – it records readmissions within 30 days of surgery instead of starting from discharge – the investigators had to limit the study population to surgery patients with a length of stay of 10 days or less and then apply statistical modeling. However, limiting the data set to patients with a maximum 10-day hospital stay only restricted the study cohort by 6%. Thus, the study results remain highly generalizable to U.S. surgery patients.
The area under the curve (AUC) of the receiver operating characteristic for the readmission risk formula was 0.70. Statisticians consider a test having an AUC of 0.50 to be worthless. An AUC of 1.0 would define a "perfect" test, while a test with an AUC in the 0.70-0.80 range is deemed of only "fair" accuracy.
The AUC for the readmission score varied considerably when applied to the various surgical subspecialties. For example, the AUC was 0.69 for general surgery patients but only 0.51 for thoracic surgery patients, 0.64 for vascular surgery patients, and 0.59 for colorectal surgery patients.
The fact that the predictive formula doesn’t perform any better than barely "fair" is testimony to the difficulty in identifying who will require readmission. That being said, the AUC for the readmission score after surgery compares favorably to published hospital readmission risk formulas developed for medical patients, which have similar and in many cases lower AUCs (JAMA 2011;306:1688-98), Dr. Pawlik observed.
At Johns Hopkins, the plan is to target surgery patients who have high readmission scores with interventions including more frequent follow-up phone calls and earlier scheduled postoperative clinic visits in an effort to keep them out of the hospital, he continued.
Discussant Dr. Keith D. Lillemoe was skeptical that this will result in reduced readmission rates. Indeed, he questioned whether the 30-day readmission rate is a legitimate quality measure for surgeons.
"I can look across the room and predict if a patient is going to come back in to the hospital. But what can we really do to keep such patients from being readmitted other than keeping them indeterminately long until we’ve passed that window? It seems like all the phone calls and early clinic visits in the world can’t stop the progression sometimes," reflected Dr. Lillemoe, professor and chairman of the department of surgery at Massachusetts General Hospital, Boston.
He noted with frustration that he had just stepped out of the lecture hall to arrange for readmission of a patient 1 week after discharge post pancreaticoduodenectomy.
"I made three phone calls myself to that guy trying to nurse him through his nausea and vomiting and other symptoms," the surgeon recalled.
Dr. Pawlik was sympathetic.
"It’s very hard to prevent readmissions. It’s a very complicated metric, and many would argue that it’s an inappropriate quality metric," he said. "Many things are outside our control before we even meet the patient, as are some things that occur in the hospital, and many things are beyond our control after discharge as far as where they live, their family structure, and their financial resources. In my opinion it’s a very problematic quality measure and I am not sure how we are going to tackle it."
He reported having no conflicts of interest.
INDIANAPOLIS – Average 30-day readmission rates in a large national study varied widely by surgical specialty, ranging from 5% for general surgery patients to 12% for vascular surgery patients and 16% after hepato-pancreatic-biliary surgery.
This retrospective study was based upon American College of Surgeons National Surgical Quality Improvement Program (NSQIP) 2011 data on 240,125 patients discharged from 316 hospitals after these types of surgery. The results enabled investigators to identify the major risk factors for readmission. They used this information to generate a predictive model to identify patients at greatest risk with an eye toward introducing interventions to keep them out of the hospital, Dr. Timothy M. Pawlik said at the annual meeting of the American Surgical Association.
How effective such interventions will be in a surgical population is as yet unclear, he added.
The importance of this work lies in the growing emphasis health care payers are placing upon 30-day readmission as a quality-of-care indicator. In 2012, Medicare began cutting reimbursement by 1% to hospitals with above-average 30-day readmissions. Next year, this penalty is scheduled to increase to 3% under the Medicare Hospital Readmission Reduction Program. Prior research efforts to identify risk factors for readmission have focused chiefly on medical rather than surgical conditions, noted Dr. Pawlik, professor of surgery and chief of the division of surgical oncology at Johns Hopkins University, Baltimore.
The readmission risk predictive formula he and his coinvestigators developed is simple: American Society of Anesthesiologists physical status class + (length of stay/2). This number is rounded up to yield a risk score of 1-10. A patient with a readmission score of 1 has a 1% risk of readmission within 30 days of discharge. The risk climbs to 12% with a score of 5, 20% with a score of 8, and 40% with a score of 10.
"A score of 4 had an 8% readmission rate, with 77% sensitivity, 52% specificity, a low positive predictive value of 12%, but a high negative predictive value of 95%," the surgeon observed. "In essence, a score of 4 or higher was able to identify 80% of all readmissions, but it also included about half of all patients."
Because of a quirk in the NSQIP database – it records readmissions within 30 days of surgery instead of starting from discharge – the investigators had to limit the study population to surgery patients with a length of stay of 10 days or less and then apply statistical modeling. However, limiting the data set to patients with a maximum 10-day hospital stay only restricted the study cohort by 6%. Thus, the study results remain highly generalizable to U.S. surgery patients.
The area under the curve (AUC) of the receiver operating characteristic for the readmission risk formula was 0.70. Statisticians consider a test having an AUC of 0.50 to be worthless. An AUC of 1.0 would define a "perfect" test, while a test with an AUC in the 0.70-0.80 range is deemed of only "fair" accuracy.
The AUC for the readmission score varied considerably when applied to the various surgical subspecialties. For example, the AUC was 0.69 for general surgery patients but only 0.51 for thoracic surgery patients, 0.64 for vascular surgery patients, and 0.59 for colorectal surgery patients.
The fact that the predictive formula doesn’t perform any better than barely "fair" is testimony to the difficulty in identifying who will require readmission. That being said, the AUC for the readmission score after surgery compares favorably to published hospital readmission risk formulas developed for medical patients, which have similar and in many cases lower AUCs (JAMA 2011;306:1688-98), Dr. Pawlik observed.
At Johns Hopkins, the plan is to target surgery patients who have high readmission scores with interventions including more frequent follow-up phone calls and earlier scheduled postoperative clinic visits in an effort to keep them out of the hospital, he continued.
Discussant Dr. Keith D. Lillemoe was skeptical that this will result in reduced readmission rates. Indeed, he questioned whether the 30-day readmission rate is a legitimate quality measure for surgeons.
"I can look across the room and predict if a patient is going to come back in to the hospital. But what can we really do to keep such patients from being readmitted other than keeping them indeterminately long until we’ve passed that window? It seems like all the phone calls and early clinic visits in the world can’t stop the progression sometimes," reflected Dr. Lillemoe, professor and chairman of the department of surgery at Massachusetts General Hospital, Boston.
He noted with frustration that he had just stepped out of the lecture hall to arrange for readmission of a patient 1 week after discharge post pancreaticoduodenectomy.
"I made three phone calls myself to that guy trying to nurse him through his nausea and vomiting and other symptoms," the surgeon recalled.
Dr. Pawlik was sympathetic.
"It’s very hard to prevent readmissions. It’s a very complicated metric, and many would argue that it’s an inappropriate quality metric," he said. "Many things are outside our control before we even meet the patient, as are some things that occur in the hospital, and many things are beyond our control after discharge as far as where they live, their family structure, and their financial resources. In my opinion it’s a very problematic quality measure and I am not sure how we are going to tackle it."
He reported having no conflicts of interest.
INDIANAPOLIS – Average 30-day readmission rates in a large national study varied widely by surgical specialty, ranging from 5% for general surgery patients to 12% for vascular surgery patients and 16% after hepato-pancreatic-biliary surgery.
This retrospective study was based upon American College of Surgeons National Surgical Quality Improvement Program (NSQIP) 2011 data on 240,125 patients discharged from 316 hospitals after these types of surgery. The results enabled investigators to identify the major risk factors for readmission. They used this information to generate a predictive model to identify patients at greatest risk with an eye toward introducing interventions to keep them out of the hospital, Dr. Timothy M. Pawlik said at the annual meeting of the American Surgical Association.
How effective such interventions will be in a surgical population is as yet unclear, he added.
The importance of this work lies in the growing emphasis health care payers are placing upon 30-day readmission as a quality-of-care indicator. In 2012, Medicare began cutting reimbursement by 1% to hospitals with above-average 30-day readmissions. Next year, this penalty is scheduled to increase to 3% under the Medicare Hospital Readmission Reduction Program. Prior research efforts to identify risk factors for readmission have focused chiefly on medical rather than surgical conditions, noted Dr. Pawlik, professor of surgery and chief of the division of surgical oncology at Johns Hopkins University, Baltimore.
The readmission risk predictive formula he and his coinvestigators developed is simple: American Society of Anesthesiologists physical status class + (length of stay/2). This number is rounded up to yield a risk score of 1-10. A patient with a readmission score of 1 has a 1% risk of readmission within 30 days of discharge. The risk climbs to 12% with a score of 5, 20% with a score of 8, and 40% with a score of 10.
"A score of 4 had an 8% readmission rate, with 77% sensitivity, 52% specificity, a low positive predictive value of 12%, but a high negative predictive value of 95%," the surgeon observed. "In essence, a score of 4 or higher was able to identify 80% of all readmissions, but it also included about half of all patients."
Because of a quirk in the NSQIP database – it records readmissions within 30 days of surgery instead of starting from discharge – the investigators had to limit the study population to surgery patients with a length of stay of 10 days or less and then apply statistical modeling. However, limiting the data set to patients with a maximum 10-day hospital stay only restricted the study cohort by 6%. Thus, the study results remain highly generalizable to U.S. surgery patients.
The area under the curve (AUC) of the receiver operating characteristic for the readmission risk formula was 0.70. Statisticians consider a test having an AUC of 0.50 to be worthless. An AUC of 1.0 would define a "perfect" test, while a test with an AUC in the 0.70-0.80 range is deemed of only "fair" accuracy.
The AUC for the readmission score varied considerably when applied to the various surgical subspecialties. For example, the AUC was 0.69 for general surgery patients but only 0.51 for thoracic surgery patients, 0.64 for vascular surgery patients, and 0.59 for colorectal surgery patients.
The fact that the predictive formula doesn’t perform any better than barely "fair" is testimony to the difficulty in identifying who will require readmission. That being said, the AUC for the readmission score after surgery compares favorably to published hospital readmission risk formulas developed for medical patients, which have similar and in many cases lower AUCs (JAMA 2011;306:1688-98), Dr. Pawlik observed.
At Johns Hopkins, the plan is to target surgery patients who have high readmission scores with interventions including more frequent follow-up phone calls and earlier scheduled postoperative clinic visits in an effort to keep them out of the hospital, he continued.
Discussant Dr. Keith D. Lillemoe was skeptical that this will result in reduced readmission rates. Indeed, he questioned whether the 30-day readmission rate is a legitimate quality measure for surgeons.
"I can look across the room and predict if a patient is going to come back in to the hospital. But what can we really do to keep such patients from being readmitted other than keeping them indeterminately long until we’ve passed that window? It seems like all the phone calls and early clinic visits in the world can’t stop the progression sometimes," reflected Dr. Lillemoe, professor and chairman of the department of surgery at Massachusetts General Hospital, Boston.
He noted with frustration that he had just stepped out of the lecture hall to arrange for readmission of a patient 1 week after discharge post pancreaticoduodenectomy.
"I made three phone calls myself to that guy trying to nurse him through his nausea and vomiting and other symptoms," the surgeon recalled.
Dr. Pawlik was sympathetic.
"It’s very hard to prevent readmissions. It’s a very complicated metric, and many would argue that it’s an inappropriate quality metric," he said. "Many things are outside our control before we even meet the patient, as are some things that occur in the hospital, and many things are beyond our control after discharge as far as where they live, their family structure, and their financial resources. In my opinion it’s a very problematic quality measure and I am not sure how we are going to tackle it."
He reported having no conflicts of interest.
AT THE ASA ANNUAL MEETING
Major Finding: The 30-day readmission rate following general, thoracic, and vascular surgery was 8%. It ranged from 5% to 16% depending upon the surgical subspecialty. The study led to generation of a simple readmission risk scoring system.
Data Source: A retrospective study of 240,125 patients in the American College of Surgeons National Surgical Quality Improvement Program who were discharged in 2011 following these types of surgery.
Disclosures: The presenter reported having no conflicts of interest.
Preop walking speed predicts postop morbidity
INDIANAPOLIS – Slower walking speed on the timed-up-and-go test in elderly patients scheduled for surgery is a significantly better forecaster of postoperative complications and 1-year mortality than are the considerably more complex patient risk calculators currently considered standard of care, Dr. Thomas N. Robinson reported at the annual meeting of the American Surgical Association.
"I think what walking speed reflects is global reduced physiologic reserve. It’s frailty. And by definition, an individual who’s frail will have adverse health care outcomes," explained Dr. Robinson, a general surgeon at the University of Colorado, Denver.
Use of preoperative walking speed to assess postoperative risk is a paradigm shift, he noted. Current surgical risk assessment strategies rely upon math-heavy patient risk calculators which evaluate single end-organ dysfunction, in some cases summing up the individual scores for heart, lung, liver, and other organ dysfunction in an attempt to define chronic disease burden. But in older patients, this approach is less effective than a simple frailty assessment based upon mobility: that is, walking speed, he continued.
Surgeons in the Veterans Affairs health care system use a risk calculator that involves input of 24 variables. Hitting the "compute risk" button then produces the patient’s estimated 30-day morbidity and mortality risks.
Dr. Robinson presented a prospective study in which the VA tool was compared to the timed-up-and-go (TUG) test in 272 patients aged older than 65 years who were followed for a minimum of 1 year after elective surgery. To see how the two tests performed across surgical specialties, the investigators included 174 patients with a cardiac operation and 98 who had colorectal surgery.
In the TUG test, a clinician starts a stopwatch as the patient rises from a chair, walks 10 feet, returns, and sits back down. The patient is instructed to walk at his or her normal pace and is free to use a walking aid. Dr. Robinson chose to study the TUG rather than a simple 5-meter gait speed test because he considers TUG more relevant to surgical patients.
"TUG combines lower extremity strength to stand up in addition to walking speed. And if you think about somebody who needs to make the transition from hospital to home, lower extremity strength is important," he said.
The investigators categorized a TUG time of 10 seconds or less as fast, 11-14 seconds as intermediate, and 15 seconds or longer as slow. Of note, the subjects’ TUG times were unrelated to common comorbid conditions in the elderly, including stroke, diabetes, heart failure, and hypertension.
In contrast, walking speed was strongly associated with classic indicators of frailty. For example, impaired cognition was present in 3% of subjects with a fast TUG time, 41% of those with an intermediate time, and 92% of slow performers. Another frailty indicator – a history of falling within the past 6 months – was present in 7% of the fast group, 21% of intermediate TUG walkers, and 85% of those with a TUG speed of 15 seconds or more.
In the cardiac surgery group, one or more postsurgical complications occurred in 11% of the 53 patients in the fast group, 25% of 88 patients with an intermediate TUG time, and 52% of 33 individuals in the slow group. The 1-year mortality rates were 2%, 3%, and 12%, respectively.
Similarly, in the colorectal surgery group, the complication rate was 12% among 30 fast walkers, 29% of 42 patients in the intermediate group, and 77% of 26 patients in the slow group. The 1-year mortality rates were 3%, 10%, and 31%, respectively.
The investigators judged comparative test performance in predicting postoperative morbidity and mortality on the basis of the receiver operating characteristic area under the curve, which was 77% with the TUG test compared to 55% with the VA risk calculator in the colorectal surgery patients. In the cardiac surgery group, the figures were 68% for TUG and 55% with the risk calculator.
Geriatricians typically measure TUG in seconds as a continuous variable. Dr. Robinson and coworkers decided the test would be more useful for surgeons if they created the three discrete categories of fast, intermediate, and slow.
Discussant Dr. Michael E. Zenilman praised the investigators for what he called "an outstanding study," and one that’s particularly welcome right now, as the wave of aging baby boomers swells.
"As we take care of more elderly patients, it’s important that we develop tools like this to quickly and objectively assess risk. The tools that we have now, such as the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) models and the VA risk calculator, are for me just too complicated," said Dr. Zenilman of Johns Hopkins University, Baltimore. Dr. Zenilman is the university’s vice chair and regional director of surgery for the Washington area. Noting that TUG, Mini-Mental Status scores, history of falling, and serum albumin levels all have been shown to serve as proxies for frailty, he asked Dr. Robinson to predict which one he thinks will win out as a postoperative risk predictor.
Dr. Robinson replied that TUG is a good frailty assessment tool for now, but he and others are trying to develop something better. The American College of Surgeons geriatric task force is collaborating with the NSQIP to identify variables present in patients’ charts that correlate with global frailty and can serve as reliable predictors of postoperative risk.
He reported having no financial conflicts.
INDIANAPOLIS – Slower walking speed on the timed-up-and-go test in elderly patients scheduled for surgery is a significantly better forecaster of postoperative complications and 1-year mortality than are the considerably more complex patient risk calculators currently considered standard of care, Dr. Thomas N. Robinson reported at the annual meeting of the American Surgical Association.
"I think what walking speed reflects is global reduced physiologic reserve. It’s frailty. And by definition, an individual who’s frail will have adverse health care outcomes," explained Dr. Robinson, a general surgeon at the University of Colorado, Denver.
Use of preoperative walking speed to assess postoperative risk is a paradigm shift, he noted. Current surgical risk assessment strategies rely upon math-heavy patient risk calculators which evaluate single end-organ dysfunction, in some cases summing up the individual scores for heart, lung, liver, and other organ dysfunction in an attempt to define chronic disease burden. But in older patients, this approach is less effective than a simple frailty assessment based upon mobility: that is, walking speed, he continued.
Surgeons in the Veterans Affairs health care system use a risk calculator that involves input of 24 variables. Hitting the "compute risk" button then produces the patient’s estimated 30-day morbidity and mortality risks.
Dr. Robinson presented a prospective study in which the VA tool was compared to the timed-up-and-go (TUG) test in 272 patients aged older than 65 years who were followed for a minimum of 1 year after elective surgery. To see how the two tests performed across surgical specialties, the investigators included 174 patients with a cardiac operation and 98 who had colorectal surgery.
In the TUG test, a clinician starts a stopwatch as the patient rises from a chair, walks 10 feet, returns, and sits back down. The patient is instructed to walk at his or her normal pace and is free to use a walking aid. Dr. Robinson chose to study the TUG rather than a simple 5-meter gait speed test because he considers TUG more relevant to surgical patients.
"TUG combines lower extremity strength to stand up in addition to walking speed. And if you think about somebody who needs to make the transition from hospital to home, lower extremity strength is important," he said.
The investigators categorized a TUG time of 10 seconds or less as fast, 11-14 seconds as intermediate, and 15 seconds or longer as slow. Of note, the subjects’ TUG times were unrelated to common comorbid conditions in the elderly, including stroke, diabetes, heart failure, and hypertension.
In contrast, walking speed was strongly associated with classic indicators of frailty. For example, impaired cognition was present in 3% of subjects with a fast TUG time, 41% of those with an intermediate time, and 92% of slow performers. Another frailty indicator – a history of falling within the past 6 months – was present in 7% of the fast group, 21% of intermediate TUG walkers, and 85% of those with a TUG speed of 15 seconds or more.
In the cardiac surgery group, one or more postsurgical complications occurred in 11% of the 53 patients in the fast group, 25% of 88 patients with an intermediate TUG time, and 52% of 33 individuals in the slow group. The 1-year mortality rates were 2%, 3%, and 12%, respectively.
Similarly, in the colorectal surgery group, the complication rate was 12% among 30 fast walkers, 29% of 42 patients in the intermediate group, and 77% of 26 patients in the slow group. The 1-year mortality rates were 3%, 10%, and 31%, respectively.
The investigators judged comparative test performance in predicting postoperative morbidity and mortality on the basis of the receiver operating characteristic area under the curve, which was 77% with the TUG test compared to 55% with the VA risk calculator in the colorectal surgery patients. In the cardiac surgery group, the figures were 68% for TUG and 55% with the risk calculator.
Geriatricians typically measure TUG in seconds as a continuous variable. Dr. Robinson and coworkers decided the test would be more useful for surgeons if they created the three discrete categories of fast, intermediate, and slow.
Discussant Dr. Michael E. Zenilman praised the investigators for what he called "an outstanding study," and one that’s particularly welcome right now, as the wave of aging baby boomers swells.
"As we take care of more elderly patients, it’s important that we develop tools like this to quickly and objectively assess risk. The tools that we have now, such as the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) models and the VA risk calculator, are for me just too complicated," said Dr. Zenilman of Johns Hopkins University, Baltimore. Dr. Zenilman is the university’s vice chair and regional director of surgery for the Washington area. Noting that TUG, Mini-Mental Status scores, history of falling, and serum albumin levels all have been shown to serve as proxies for frailty, he asked Dr. Robinson to predict which one he thinks will win out as a postoperative risk predictor.
Dr. Robinson replied that TUG is a good frailty assessment tool for now, but he and others are trying to develop something better. The American College of Surgeons geriatric task force is collaborating with the NSQIP to identify variables present in patients’ charts that correlate with global frailty and can serve as reliable predictors of postoperative risk.
He reported having no financial conflicts.
INDIANAPOLIS – Slower walking speed on the timed-up-and-go test in elderly patients scheduled for surgery is a significantly better forecaster of postoperative complications and 1-year mortality than are the considerably more complex patient risk calculators currently considered standard of care, Dr. Thomas N. Robinson reported at the annual meeting of the American Surgical Association.
"I think what walking speed reflects is global reduced physiologic reserve. It’s frailty. And by definition, an individual who’s frail will have adverse health care outcomes," explained Dr. Robinson, a general surgeon at the University of Colorado, Denver.
Use of preoperative walking speed to assess postoperative risk is a paradigm shift, he noted. Current surgical risk assessment strategies rely upon math-heavy patient risk calculators which evaluate single end-organ dysfunction, in some cases summing up the individual scores for heart, lung, liver, and other organ dysfunction in an attempt to define chronic disease burden. But in older patients, this approach is less effective than a simple frailty assessment based upon mobility: that is, walking speed, he continued.
Surgeons in the Veterans Affairs health care system use a risk calculator that involves input of 24 variables. Hitting the "compute risk" button then produces the patient’s estimated 30-day morbidity and mortality risks.
Dr. Robinson presented a prospective study in which the VA tool was compared to the timed-up-and-go (TUG) test in 272 patients aged older than 65 years who were followed for a minimum of 1 year after elective surgery. To see how the two tests performed across surgical specialties, the investigators included 174 patients with a cardiac operation and 98 who had colorectal surgery.
In the TUG test, a clinician starts a stopwatch as the patient rises from a chair, walks 10 feet, returns, and sits back down. The patient is instructed to walk at his or her normal pace and is free to use a walking aid. Dr. Robinson chose to study the TUG rather than a simple 5-meter gait speed test because he considers TUG more relevant to surgical patients.
"TUG combines lower extremity strength to stand up in addition to walking speed. And if you think about somebody who needs to make the transition from hospital to home, lower extremity strength is important," he said.
The investigators categorized a TUG time of 10 seconds or less as fast, 11-14 seconds as intermediate, and 15 seconds or longer as slow. Of note, the subjects’ TUG times were unrelated to common comorbid conditions in the elderly, including stroke, diabetes, heart failure, and hypertension.
In contrast, walking speed was strongly associated with classic indicators of frailty. For example, impaired cognition was present in 3% of subjects with a fast TUG time, 41% of those with an intermediate time, and 92% of slow performers. Another frailty indicator – a history of falling within the past 6 months – was present in 7% of the fast group, 21% of intermediate TUG walkers, and 85% of those with a TUG speed of 15 seconds or more.
In the cardiac surgery group, one or more postsurgical complications occurred in 11% of the 53 patients in the fast group, 25% of 88 patients with an intermediate TUG time, and 52% of 33 individuals in the slow group. The 1-year mortality rates were 2%, 3%, and 12%, respectively.
Similarly, in the colorectal surgery group, the complication rate was 12% among 30 fast walkers, 29% of 42 patients in the intermediate group, and 77% of 26 patients in the slow group. The 1-year mortality rates were 3%, 10%, and 31%, respectively.
The investigators judged comparative test performance in predicting postoperative morbidity and mortality on the basis of the receiver operating characteristic area under the curve, which was 77% with the TUG test compared to 55% with the VA risk calculator in the colorectal surgery patients. In the cardiac surgery group, the figures were 68% for TUG and 55% with the risk calculator.
Geriatricians typically measure TUG in seconds as a continuous variable. Dr. Robinson and coworkers decided the test would be more useful for surgeons if they created the three discrete categories of fast, intermediate, and slow.
Discussant Dr. Michael E. Zenilman praised the investigators for what he called "an outstanding study," and one that’s particularly welcome right now, as the wave of aging baby boomers swells.
"As we take care of more elderly patients, it’s important that we develop tools like this to quickly and objectively assess risk. The tools that we have now, such as the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) models and the VA risk calculator, are for me just too complicated," said Dr. Zenilman of Johns Hopkins University, Baltimore. Dr. Zenilman is the university’s vice chair and regional director of surgery for the Washington area. Noting that TUG, Mini-Mental Status scores, history of falling, and serum albumin levels all have been shown to serve as proxies for frailty, he asked Dr. Robinson to predict which one he thinks will win out as a postoperative risk predictor.
Dr. Robinson replied that TUG is a good frailty assessment tool for now, but he and others are trying to develop something better. The American College of Surgeons geriatric task force is collaborating with the NSQIP to identify variables present in patients’ charts that correlate with global frailty and can serve as reliable predictors of postoperative risk.
He reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding. One-year mortality rates for fast, intermediate, and slow cardiac patients were 2%, 3%, and 12%, respectively. In the colorectal surgery group, the 1-year mortality rates were 3%, 10%, and 31%, respectively.
Data Source: A prospective cohort study of postsurgical complications and 1-year mortality in 98 elderly patients undergoing elective colorectal surgery and 174 with elective cardiac surgery. All underwent a preoperative timed-up-and-go test as well as assessment via the Veterans Affairs surgical risk calculator.
Disclosures: The study presenter reported having no conflicts of interest.
Early surgery for adhesive bowel obstruction can save lives
INDIANAPOLIS – Patients requiring surgery for adhesive small bowel obstruction have markedly lower major morbidity and mortality rates if they’re operated on within 24 hours of hospital admission, according to an analysis of a large national database.
This finding is at odds with the conventional wisdom.
Both the World Society of Emergency Surgery and the Eastern Association for the Surgery of Trauma recommend in published guidelines an initial 3-5 days of nonoperative management to give the obstruction a chance to resolve on its own, Dr. Pedro G. Teixeira noted in presenting the study findings at the annual meeting of the American Surgical Association.
He and his coinvestigators identified 4,163 patients in the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database for 2005-2010 who underwent emergency laparotomy for adhesive bowel obstruction. Thirty-day mortality was 3% in those operated upon within 24 hours of hospital admission. It rose in stepwise fashion thereafter: 4% mortality with surgery at 24-48 hours, 7% with surgery at 48-72 hours, and 9% a threefold increase – when surgery was delayed beyond 72 hours, according to Dr. Teixeira of the University of Southern California, Los Angeles.
Similarly, the incidence of systemic infectious complications, including pneumonia, urinary tract infections, and sepsis, climbed from 12% with early operation to 17% when surgery occurred at 24-48 hours, 21% at 48-72 hours, and 24% thereafter.
In a multivariate analysis adjusted for baseline comorbidities and other potential confounding variables, surgery delayed for 24 hours or more after admission was associated with a highly significant 58% increased risk of mortality, a 33% increase in surgical site infections, a 36% greater risk of pneumonia, and a 47% increased risk of septic shock, he continued.
Discussant Gregory J. Jurkovich commented that this study challenges current dogma and harkens back to a century-old adage that has since been cast aside, namely, "Never let the sun set on a bowel obstruction."
The trouble is, however, that having a low threshold for surgery within 24 hours would subject a massive number of patients to an unnecessary operation.
An analysis of Nationwide Inpatient Sample data for 2009 by other investigators concluded that bowel obstruction resolved on its own within 3 days in 60% of patients and within 5 days in 80%. Fewer than 20% of the patients who presented with adhesive small bowel obstruction without evidence of ischemia underwent surgery, noted Dr. Jurkovich, director of surgery at Denver Health Medical Center and professor of trauma surgery and vice chairman of the department of surgery at the University of Colorado at Denver.
Dr. Teixeira concurred that bowel obstruction will resolve on its own in most patients. The challenge for surgeons in light of his study findings, he stressed, is to expedite the identification of those patients who will fail the period of nonoperative management. The best tool for that, in his view, is a CT scan of the abdomen and pelvis with water-soluble contrast.
At the University of Southern California, he explained, a patient who presents with adhesive bowel obstruction without evidence of ischemia undergoes the CT scan and is admitted to the surgical observation unit for close monitoring.
"At our institution, failure to demonstrate contrast progression through the colon within 24 hours would be a very strong indication for surgical exploration," according to Dr. Teixeira.
He reported having no financial conflicts.
The study by Dr. Teixeira is intriguing in
that it suggests a return to practice patterns from a prior era.

|
| Dr. Chad Whelan |
The study does report increased risk in
complications including mortality with delays in surgery for small bowel
obstructions, even with risk adjustment. However, this is not a controlled
trial which limits our ability to reach definitive conclusions from it. Still,
hospitalists often are the primary physicians for patients admitted for small
bowel obstructions and should be aware of these findings so that they can
ensure that they have early surgical involvement.
Chad Whelan, M.D., is associate chief medical officer for
performance improvement and innovation and an associate professor of medicine
at the University
of Chicago Medical Center.
The study by Dr. Teixeira is intriguing in
that it suggests a return to practice patterns from a prior era.

|
| Dr. Chad Whelan |
The study does report increased risk in
complications including mortality with delays in surgery for small bowel
obstructions, even with risk adjustment. However, this is not a controlled
trial which limits our ability to reach definitive conclusions from it. Still,
hospitalists often are the primary physicians for patients admitted for small
bowel obstructions and should be aware of these findings so that they can
ensure that they have early surgical involvement.
Chad Whelan, M.D., is associate chief medical officer for
performance improvement and innovation and an associate professor of medicine
at the University
of Chicago Medical Center.
The study by Dr. Teixeira is intriguing in
that it suggests a return to practice patterns from a prior era.

|
| Dr. Chad Whelan |
The study does report increased risk in
complications including mortality with delays in surgery for small bowel
obstructions, even with risk adjustment. However, this is not a controlled
trial which limits our ability to reach definitive conclusions from it. Still,
hospitalists often are the primary physicians for patients admitted for small
bowel obstructions and should be aware of these findings so that they can
ensure that they have early surgical involvement.
Chad Whelan, M.D., is associate chief medical officer for
performance improvement and innovation and an associate professor of medicine
at the University
of Chicago Medical Center.
INDIANAPOLIS – Patients requiring surgery for adhesive small bowel obstruction have markedly lower major morbidity and mortality rates if they’re operated on within 24 hours of hospital admission, according to an analysis of a large national database.
This finding is at odds with the conventional wisdom.
Both the World Society of Emergency Surgery and the Eastern Association for the Surgery of Trauma recommend in published guidelines an initial 3-5 days of nonoperative management to give the obstruction a chance to resolve on its own, Dr. Pedro G. Teixeira noted in presenting the study findings at the annual meeting of the American Surgical Association.
He and his coinvestigators identified 4,163 patients in the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database for 2005-2010 who underwent emergency laparotomy for adhesive bowel obstruction. Thirty-day mortality was 3% in those operated upon within 24 hours of hospital admission. It rose in stepwise fashion thereafter: 4% mortality with surgery at 24-48 hours, 7% with surgery at 48-72 hours, and 9% a threefold increase – when surgery was delayed beyond 72 hours, according to Dr. Teixeira of the University of Southern California, Los Angeles.
Similarly, the incidence of systemic infectious complications, including pneumonia, urinary tract infections, and sepsis, climbed from 12% with early operation to 17% when surgery occurred at 24-48 hours, 21% at 48-72 hours, and 24% thereafter.
In a multivariate analysis adjusted for baseline comorbidities and other potential confounding variables, surgery delayed for 24 hours or more after admission was associated with a highly significant 58% increased risk of mortality, a 33% increase in surgical site infections, a 36% greater risk of pneumonia, and a 47% increased risk of septic shock, he continued.
Discussant Gregory J. Jurkovich commented that this study challenges current dogma and harkens back to a century-old adage that has since been cast aside, namely, "Never let the sun set on a bowel obstruction."
The trouble is, however, that having a low threshold for surgery within 24 hours would subject a massive number of patients to an unnecessary operation.
An analysis of Nationwide Inpatient Sample data for 2009 by other investigators concluded that bowel obstruction resolved on its own within 3 days in 60% of patients and within 5 days in 80%. Fewer than 20% of the patients who presented with adhesive small bowel obstruction without evidence of ischemia underwent surgery, noted Dr. Jurkovich, director of surgery at Denver Health Medical Center and professor of trauma surgery and vice chairman of the department of surgery at the University of Colorado at Denver.
Dr. Teixeira concurred that bowel obstruction will resolve on its own in most patients. The challenge for surgeons in light of his study findings, he stressed, is to expedite the identification of those patients who will fail the period of nonoperative management. The best tool for that, in his view, is a CT scan of the abdomen and pelvis with water-soluble contrast.
At the University of Southern California, he explained, a patient who presents with adhesive bowel obstruction without evidence of ischemia undergoes the CT scan and is admitted to the surgical observation unit for close monitoring.
"At our institution, failure to demonstrate contrast progression through the colon within 24 hours would be a very strong indication for surgical exploration," according to Dr. Teixeira.
He reported having no financial conflicts.
INDIANAPOLIS – Patients requiring surgery for adhesive small bowel obstruction have markedly lower major morbidity and mortality rates if they’re operated on within 24 hours of hospital admission, according to an analysis of a large national database.
This finding is at odds with the conventional wisdom.
Both the World Society of Emergency Surgery and the Eastern Association for the Surgery of Trauma recommend in published guidelines an initial 3-5 days of nonoperative management to give the obstruction a chance to resolve on its own, Dr. Pedro G. Teixeira noted in presenting the study findings at the annual meeting of the American Surgical Association.
He and his coinvestigators identified 4,163 patients in the American College of Surgeons National Surgical Quality Improvement Program (NSQIP) database for 2005-2010 who underwent emergency laparotomy for adhesive bowel obstruction. Thirty-day mortality was 3% in those operated upon within 24 hours of hospital admission. It rose in stepwise fashion thereafter: 4% mortality with surgery at 24-48 hours, 7% with surgery at 48-72 hours, and 9% a threefold increase – when surgery was delayed beyond 72 hours, according to Dr. Teixeira of the University of Southern California, Los Angeles.
Similarly, the incidence of systemic infectious complications, including pneumonia, urinary tract infections, and sepsis, climbed from 12% with early operation to 17% when surgery occurred at 24-48 hours, 21% at 48-72 hours, and 24% thereafter.
In a multivariate analysis adjusted for baseline comorbidities and other potential confounding variables, surgery delayed for 24 hours or more after admission was associated with a highly significant 58% increased risk of mortality, a 33% increase in surgical site infections, a 36% greater risk of pneumonia, and a 47% increased risk of septic shock, he continued.
Discussant Gregory J. Jurkovich commented that this study challenges current dogma and harkens back to a century-old adage that has since been cast aside, namely, "Never let the sun set on a bowel obstruction."
The trouble is, however, that having a low threshold for surgery within 24 hours would subject a massive number of patients to an unnecessary operation.
An analysis of Nationwide Inpatient Sample data for 2009 by other investigators concluded that bowel obstruction resolved on its own within 3 days in 60% of patients and within 5 days in 80%. Fewer than 20% of the patients who presented with adhesive small bowel obstruction without evidence of ischemia underwent surgery, noted Dr. Jurkovich, director of surgery at Denver Health Medical Center and professor of trauma surgery and vice chairman of the department of surgery at the University of Colorado at Denver.
Dr. Teixeira concurred that bowel obstruction will resolve on its own in most patients. The challenge for surgeons in light of his study findings, he stressed, is to expedite the identification of those patients who will fail the period of nonoperative management. The best tool for that, in his view, is a CT scan of the abdomen and pelvis with water-soluble contrast.
At the University of Southern California, he explained, a patient who presents with adhesive bowel obstruction without evidence of ischemia undergoes the CT scan and is admitted to the surgical observation unit for close monitoring.
"At our institution, failure to demonstrate contrast progression through the colon within 24 hours would be a very strong indication for surgical exploration," according to Dr. Teixeira.
He reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Surgery for adhesive small bowel obstruction had a 30-day mortality rate of 3% if performed within 24 hours of hospital admission, rising stepwise to 9% when the operation was delayed beyond 72 hours.
Data Source: This was a retrospective analysis of 4,163 patients in the American College of Surgeons National Quality Improvement Program database for 2005-2010 who underwent emergency laparotomy for adhesive bowel obstruction.
Disclosures: The presenter reported having no conflicts of interest.
Esophagectomy cases rising steadily
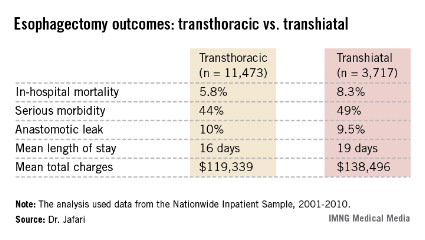
INDIANAPOLIS – Transthoracic esophagectomy for esophageal cancer provides significantly lower in-hospital mortality and major morbidity rates than does transhiatal esophagectomy, according to an analysis of a large multiyear national database.
Further, in-hospital outcomes of esophagectomy didn’t differ significantly between high-volume centers – in this study, defined as those doing 10 or more cases per year – and low-volume centers, Dr. Mehraneh D. Jafari reported at the annual meeting of the American Surgical Association.

That finding was met with skepticism, and discussants were quick to argue that study limitations make it difficult to draw any meaningful conclusions from the data. For one thing, speakers contended that defining a high-volume center based upon an institutional threshold of 10 or more cases per year sets the bar far too low given that a single dedicated esophageal surgery specialist might easily perform 50 or more esophagectomies annually.
Dr. Jafari presented an analysis of 11,473 transthoracic and 3,717 transhiatal esophagectomies performed for esophageal cancer. The data came from the Nationwide Inpatient Sample (NIS) during 2001-2010. The NIS records data on in-hospital outcomes for a nationally representative sample composed of roughly 20% of the country’s hospital discharges each year.
The number of esophagectomies rose steadily by an average of 4% annually during the study years, reflecting the substantial national increase in cases of esophageal cancer. The growing case count, expected to reach an estimated 18,000 cases of esophageal cancer nationwide in 2013, has been attributed to rising rates of gastroesophageal reflux disease, Barrett’s esophagus, and obesity. Transthoracic esophagectomy, used in 76% of cases, remained the preferred operative strategy throughout the study years.
In-hospital outcomes were markedly better in patients who had transthoracic esophagectomy. After adjustment for potential confounding variables in a multivariate analysis, transhiatal esophagectomy recipients had a 67% increased risk of in-hospital mortality and a 39% greater risk of serious complications, including a 37% increased risk of pulmonary complications. However, anastomotic leak rates were similar with both operations, according to Dr. Jafari of the University of California, Irvine.
Of note, the referral rate to high-volume esophagectomy centers climbed steadily over time, rising from 22% of all cases in 2001 to 58% in 2010.
The 35 high-volume centers performed an average of 16 cases per year. In contrast, the 484 low-volume centers averaged 2 cases per year. In-hospital mortality among the 9,386 patients treated in low-volume centers averaged 7.6% compared with 4.3% for patients in high-volume centers. Overall in-hospital serious morbidity rates were greater in the low-volume centers as well: 47% versus 41%. While these raw differences were statistically significant, a risk-adjusted multivariate analysis found no significant outcome differences between low- and high-volume centers.
Discussant Dr. Michael J. Zinner noted that in an earlier study he and his coworkers showed that an institutional threshold of roughly 30 esophagectomies per year is required to discriminate between low- and high-volume centers in terms of in-hospital mortality. So why define high-volume centers as those doing a mere 10 cases per year? asked Dr. Zinner, chairman of the department of surgery at Brigham and Women’s Hospital and professor of surgery at Harvard Medical School, Boston.
"The problem here is if you establish 30 cases per year as the threshold for a high-volume center, I can tell you there are probably less than 20 centers in the whole U.S. capable of doing that volume. That’s a real issue, because then how are patients who live in a remote region going to get care at one of those centers?" replied Dr. Jafari’s senior coauthor Dr. Ninh T. Nguyen, professor and vice-chair of surgery at UC Irvine.
"I think instead we should try to lift all boats: develop a national esophageal center network to identify the qualities reflective of better outcomes in the high-volume centers and introduce those factors at low-volume centers. This way we’re not impeding access to care for our patients," he continued.
Dr. Nguyen said that in-hospital surgical morbidity rates in the NIS need to be taken with a grain of salt, as the accuracy of coding for complications is "rather low." The development of minimally invasive techniques for intrathoracic anastomosis has transformed transthoracic esophagectomy into a procedure with an improved complication profile.
"I switched to transthoracic esophagectomy 5 years ago. One reason was development of the minimally invasive approach. As a result, we’re not scared of a chest anastomosis like we used to be. When patients undergoing transthoracic esophagectomy with thoracotomy had a leak in the chest they had a very high risk for mortality. That’s not the case anymore. We have not observed any mortality associated with a leak in the chest for many, many years now," he said.
The investigators reported having no conflicts of interest.
Dr. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
I have major problems with this study, stemming from inherent limitations in the Nationwide Inpatient Sample. It’s an administrative database set up chiefly to track costs, utilization, and length of stay. It contains no information at all on key clinical outcomes such as 30- and 90-day mortality, discharge disposition, or 30-day readmission rates.

|
|
In addition, the accuracy of the quoted in-hospital morbidity rates is suspect, probably because data entry isn’t performed by trained researchers. For example, the 8% incidence of renal failure in esophagectomy patients cited in this study sounds too high to be right.
And there’s another major problem with this database: The superior outcomes reported for transthoracic esophagectomy recipients in this study fly in the face of earlier, well-conducted meta-analyses that reached the opposite conclusion. The most likely explanation for the discordant findings lies in the fact that the NIS doesn’t show whether a transthoracic esophagectomy was performed via open thoracotomy in the old-school manner or with an intrathoracic anastomosis created using contemporary minimally invasive techniques which, while complex, have been associated with better outcomes.
Dr. James D. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
I have major problems with this study, stemming from inherent limitations in the Nationwide Inpatient Sample. It’s an administrative database set up chiefly to track costs, utilization, and length of stay. It contains no information at all on key clinical outcomes such as 30- and 90-day mortality, discharge disposition, or 30-day readmission rates.

|
|
In addition, the accuracy of the quoted in-hospital morbidity rates is suspect, probably because data entry isn’t performed by trained researchers. For example, the 8% incidence of renal failure in esophagectomy patients cited in this study sounds too high to be right.
And there’s another major problem with this database: The superior outcomes reported for transthoracic esophagectomy recipients in this study fly in the face of earlier, well-conducted meta-analyses that reached the opposite conclusion. The most likely explanation for the discordant findings lies in the fact that the NIS doesn’t show whether a transthoracic esophagectomy was performed via open thoracotomy in the old-school manner or with an intrathoracic anastomosis created using contemporary minimally invasive techniques which, while complex, have been associated with better outcomes.
Dr. James D. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
I have major problems with this study, stemming from inherent limitations in the Nationwide Inpatient Sample. It’s an administrative database set up chiefly to track costs, utilization, and length of stay. It contains no information at all on key clinical outcomes such as 30- and 90-day mortality, discharge disposition, or 30-day readmission rates.

|
|
In addition, the accuracy of the quoted in-hospital morbidity rates is suspect, probably because data entry isn’t performed by trained researchers. For example, the 8% incidence of renal failure in esophagectomy patients cited in this study sounds too high to be right.
And there’s another major problem with this database: The superior outcomes reported for transthoracic esophagectomy recipients in this study fly in the face of earlier, well-conducted meta-analyses that reached the opposite conclusion. The most likely explanation for the discordant findings lies in the fact that the NIS doesn’t show whether a transthoracic esophagectomy was performed via open thoracotomy in the old-school manner or with an intrathoracic anastomosis created using contemporary minimally invasive techniques which, while complex, have been associated with better outcomes.
Dr. James D. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
INDIANAPOLIS – Transthoracic esophagectomy for esophageal cancer provides significantly lower in-hospital mortality and major morbidity rates than does transhiatal esophagectomy, according to an analysis of a large multiyear national database.
Further, in-hospital outcomes of esophagectomy didn’t differ significantly between high-volume centers – in this study, defined as those doing 10 or more cases per year – and low-volume centers, Dr. Mehraneh D. Jafari reported at the annual meeting of the American Surgical Association.

That finding was met with skepticism, and discussants were quick to argue that study limitations make it difficult to draw any meaningful conclusions from the data. For one thing, speakers contended that defining a high-volume center based upon an institutional threshold of 10 or more cases per year sets the bar far too low given that a single dedicated esophageal surgery specialist might easily perform 50 or more esophagectomies annually.
Dr. Jafari presented an analysis of 11,473 transthoracic and 3,717 transhiatal esophagectomies performed for esophageal cancer. The data came from the Nationwide Inpatient Sample (NIS) during 2001-2010. The NIS records data on in-hospital outcomes for a nationally representative sample composed of roughly 20% of the country’s hospital discharges each year.
The number of esophagectomies rose steadily by an average of 4% annually during the study years, reflecting the substantial national increase in cases of esophageal cancer. The growing case count, expected to reach an estimated 18,000 cases of esophageal cancer nationwide in 2013, has been attributed to rising rates of gastroesophageal reflux disease, Barrett’s esophagus, and obesity. Transthoracic esophagectomy, used in 76% of cases, remained the preferred operative strategy throughout the study years.
In-hospital outcomes were markedly better in patients who had transthoracic esophagectomy. After adjustment for potential confounding variables in a multivariate analysis, transhiatal esophagectomy recipients had a 67% increased risk of in-hospital mortality and a 39% greater risk of serious complications, including a 37% increased risk of pulmonary complications. However, anastomotic leak rates were similar with both operations, according to Dr. Jafari of the University of California, Irvine.
Of note, the referral rate to high-volume esophagectomy centers climbed steadily over time, rising from 22% of all cases in 2001 to 58% in 2010.
The 35 high-volume centers performed an average of 16 cases per year. In contrast, the 484 low-volume centers averaged 2 cases per year. In-hospital mortality among the 9,386 patients treated in low-volume centers averaged 7.6% compared with 4.3% for patients in high-volume centers. Overall in-hospital serious morbidity rates were greater in the low-volume centers as well: 47% versus 41%. While these raw differences were statistically significant, a risk-adjusted multivariate analysis found no significant outcome differences between low- and high-volume centers.
Discussant Dr. Michael J. Zinner noted that in an earlier study he and his coworkers showed that an institutional threshold of roughly 30 esophagectomies per year is required to discriminate between low- and high-volume centers in terms of in-hospital mortality. So why define high-volume centers as those doing a mere 10 cases per year? asked Dr. Zinner, chairman of the department of surgery at Brigham and Women’s Hospital and professor of surgery at Harvard Medical School, Boston.
"The problem here is if you establish 30 cases per year as the threshold for a high-volume center, I can tell you there are probably less than 20 centers in the whole U.S. capable of doing that volume. That’s a real issue, because then how are patients who live in a remote region going to get care at one of those centers?" replied Dr. Jafari’s senior coauthor Dr. Ninh T. Nguyen, professor and vice-chair of surgery at UC Irvine.
"I think instead we should try to lift all boats: develop a national esophageal center network to identify the qualities reflective of better outcomes in the high-volume centers and introduce those factors at low-volume centers. This way we’re not impeding access to care for our patients," he continued.
Dr. Nguyen said that in-hospital surgical morbidity rates in the NIS need to be taken with a grain of salt, as the accuracy of coding for complications is "rather low." The development of minimally invasive techniques for intrathoracic anastomosis has transformed transthoracic esophagectomy into a procedure with an improved complication profile.
"I switched to transthoracic esophagectomy 5 years ago. One reason was development of the minimally invasive approach. As a result, we’re not scared of a chest anastomosis like we used to be. When patients undergoing transthoracic esophagectomy with thoracotomy had a leak in the chest they had a very high risk for mortality. That’s not the case anymore. We have not observed any mortality associated with a leak in the chest for many, many years now," he said.
The investigators reported having no conflicts of interest.
Dr. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
INDIANAPOLIS – Transthoracic esophagectomy for esophageal cancer provides significantly lower in-hospital mortality and major morbidity rates than does transhiatal esophagectomy, according to an analysis of a large multiyear national database.
Further, in-hospital outcomes of esophagectomy didn’t differ significantly between high-volume centers – in this study, defined as those doing 10 or more cases per year – and low-volume centers, Dr. Mehraneh D. Jafari reported at the annual meeting of the American Surgical Association.

That finding was met with skepticism, and discussants were quick to argue that study limitations make it difficult to draw any meaningful conclusions from the data. For one thing, speakers contended that defining a high-volume center based upon an institutional threshold of 10 or more cases per year sets the bar far too low given that a single dedicated esophageal surgery specialist might easily perform 50 or more esophagectomies annually.
Dr. Jafari presented an analysis of 11,473 transthoracic and 3,717 transhiatal esophagectomies performed for esophageal cancer. The data came from the Nationwide Inpatient Sample (NIS) during 2001-2010. The NIS records data on in-hospital outcomes for a nationally representative sample composed of roughly 20% of the country’s hospital discharges each year.
The number of esophagectomies rose steadily by an average of 4% annually during the study years, reflecting the substantial national increase in cases of esophageal cancer. The growing case count, expected to reach an estimated 18,000 cases of esophageal cancer nationwide in 2013, has been attributed to rising rates of gastroesophageal reflux disease, Barrett’s esophagus, and obesity. Transthoracic esophagectomy, used in 76% of cases, remained the preferred operative strategy throughout the study years.
In-hospital outcomes were markedly better in patients who had transthoracic esophagectomy. After adjustment for potential confounding variables in a multivariate analysis, transhiatal esophagectomy recipients had a 67% increased risk of in-hospital mortality and a 39% greater risk of serious complications, including a 37% increased risk of pulmonary complications. However, anastomotic leak rates were similar with both operations, according to Dr. Jafari of the University of California, Irvine.
Of note, the referral rate to high-volume esophagectomy centers climbed steadily over time, rising from 22% of all cases in 2001 to 58% in 2010.
The 35 high-volume centers performed an average of 16 cases per year. In contrast, the 484 low-volume centers averaged 2 cases per year. In-hospital mortality among the 9,386 patients treated in low-volume centers averaged 7.6% compared with 4.3% for patients in high-volume centers. Overall in-hospital serious morbidity rates were greater in the low-volume centers as well: 47% versus 41%. While these raw differences were statistically significant, a risk-adjusted multivariate analysis found no significant outcome differences between low- and high-volume centers.
Discussant Dr. Michael J. Zinner noted that in an earlier study he and his coworkers showed that an institutional threshold of roughly 30 esophagectomies per year is required to discriminate between low- and high-volume centers in terms of in-hospital mortality. So why define high-volume centers as those doing a mere 10 cases per year? asked Dr. Zinner, chairman of the department of surgery at Brigham and Women’s Hospital and professor of surgery at Harvard Medical School, Boston.
"The problem here is if you establish 30 cases per year as the threshold for a high-volume center, I can tell you there are probably less than 20 centers in the whole U.S. capable of doing that volume. That’s a real issue, because then how are patients who live in a remote region going to get care at one of those centers?" replied Dr. Jafari’s senior coauthor Dr. Ninh T. Nguyen, professor and vice-chair of surgery at UC Irvine.
"I think instead we should try to lift all boats: develop a national esophageal center network to identify the qualities reflective of better outcomes in the high-volume centers and introduce those factors at low-volume centers. This way we’re not impeding access to care for our patients," he continued.
Dr. Nguyen said that in-hospital surgical morbidity rates in the NIS need to be taken with a grain of salt, as the accuracy of coding for complications is "rather low." The development of minimally invasive techniques for intrathoracic anastomosis has transformed transthoracic esophagectomy into a procedure with an improved complication profile.
"I switched to transthoracic esophagectomy 5 years ago. One reason was development of the minimally invasive approach. As a result, we’re not scared of a chest anastomosis like we used to be. When patients undergoing transthoracic esophagectomy with thoracotomy had a leak in the chest they had a very high risk for mortality. That’s not the case anymore. We have not observed any mortality associated with a leak in the chest for many, many years now," he said.
The investigators reported having no conflicts of interest.
Dr. Luketich is professor of surgery and chief of the Heart, Lung, and Esophageal Surgery Institute at the University of Pittsburgh. He was the designated discussant of the study at the meeting.
AT THE ASA ANNUAL MEETING
Major finding: In-hospital mortality occurred nationally in 5.8% of esophageal cancer patients who underwent transthoracic esophagectomy compared with 8.3% of transhiatal esophagectomy recipients.
Data source: A retrospective study of more than 15,000 patients who underwent esophagectomy for esophageal cancer during 2001-2010 and were Included in the Nationwide Inpatient Sample, a database sponsored by the Agency for Healthcare Research and Quality.
Disclosures: The study presenters reported having no financial conflicts.
Microwave therapy zaps hyperhidrosis long-term
MAUI, HAWAII – The future of hyperhidrosis therapy may be microwave ablation, a Food and Drug Administration–cleared method of destroying sweat glands noninvasively.
"We’ve seen that it’s effective in about 90% of patients, the patients are really satisfied, and the side effects are transient," Dr. Nazanin Saedi said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Larger patient numbers and more long-term data are needed before the therapy becomes common in clinical practice, but it shouldn’t take long to accumulate such data, because the proprietary miraDry microwave treatment system (marketed by Miramar Labs) is much in demand by patients, noted Dr. Saedi of Thomas Jefferson University, Philadelphia.
An estimated 4.3 million Americans have axillary hyperhidrosis, Dr. Saedi said. Numerous treatment options are available, but most have significant drawbacks, she noted. First-line therapy is topical aluminum chloride, with or without salicylic acid; irritant dermatitis is a common complaint.
Second-line options include iontophoresis, which requires treatments two or three times per week and can be quite painful, Dr. Saedi said. Botulinum toxin A injections are highly effective, but they must be repeated every 4-6 months, and can cause weakness of the underlying muscles. Oral medications include anticholinergic agents and alpha2-adrenergic drugs, all with side effects many patients find problematic.
Before microwave therapy, surgical procedures were the only means of destroying sweat glands, and surgery is considered third-line therapy, Dr. Saedi said.
"Now that we have the newer technologies, I think surgery will become even more rare," she predicted.
The study that earned the miraDry device its Food and Drug Administration marketing clearance was a blinded trial in which 120 patients were randomized 2:1 to microwave therapy or a sham procedure. At baseline, all participants were rated 3 or 4 on the 1-4 Hyperhidrosis Disease Severity Scale (HDSS). At a follow-up point of 30 days, 89% of patients in the treatment group met the primary endpoint – an HDSS score of 1 or 2 – compared with 54% of controls. Roughly 70% of patients in the microwave treatment group still had an HDSS score of 1-2 at 12 months (Dermatol. Surg. 2012;38:185-91).
However, that study involved an earlier-generation, less efficient device, Dr. Saedi noted. In an open-label study of 31 patients treated with the current-generation, commercially available miraDry device, all subjects had a baseline HDSS score of 3 or 4. Twelve months after patients received 1-3 treatment sessions, 90% had an HDSS score of 1 or 2. In addition, 90% of patients had at least a 50% reduction in gravimetrically measured axillary sweat, and 85% had at least a 5-point improvement on the Dermatologic Life Quality Index (Dermatol. Surg. 2012;38:728-35).
In the microwave treatment procedure, an applicator is placed on the skin surface to deliver focused energy to the dermal-fat interface, where sweat glands are located. Microwaves are preferentially absorbed by tissues with high water content, such as sweat glands, and poorly absorbed by fat. A cooling system keeps the heat at the lower skin layer. A heat dome at about 60° C is created along the dermal-fat junction, resulting in sweat gland thermolysis, Dr. Saedi explained.
Two hours prior to treatment, the patient takes 800 mg of ibuprofen. A grid of temporary markings is placed over the area to be treated, and the dermis is anesthetized with injections of a 1:1 mix of 1% plain lidocaine and 1% lidocaine plus epinephrine at 1-cm intervals.
Microwave therapy entails two treatment sessions roughly 3 months apart. The first typically takes 60-90 minutes, the second 30-60 minutes.
Dr. Saedi said swelling, bruising, discomfort, and altered sensation in and around the treatment area are all common side effects of microwave therapy, but all are short-term. Less common side effects include swelling extending beyond the treatment area, a tight banding sensation in the axilla, and arm and hand numbness lasting less than 24 hours, she said.
One audience member who has treated 130 patients via microwave therapy reported that one of them developed a significant brachial nerve palsy that has lasted for 5 months. As a consequence, the commenter said he now routinely employs tumescent anesthesia in an effort to steer clear of the axillary nerve bundles. But Dr. Suzanne L. Kilmer said she doubts that will prevent the occasional case of brachial nerve palsy, which she believes is the result of variant anatomy.
"We’ve done the procedure on many thin people, and not had brachial nerve palsy be an issue, so I’m not sure that tumescent anesthesia will make a difference," said Dr. Kilmer of the University of California, Davis.
Pulsed ultrasound is currently under investigation as a means of treating hyperhidrosis through destruction of sweat glands, session moderator Dr. Christopher B. Zachary said in response to an audience question. "This would be an off-label indication right now, but you can absolutely hit that region with an ultrasound device and cause heating. It’s quite likely that ultrasound will be used extensively in exactly the same way as microwave, and probably with the same efficacy," predicted Dr. Zachary of the University of California, Irvine.
Dr. Zachary is a consultant to and has received research funding from numerous laser and other medical device companies, as has Dr. Kilmer. Dr. Saedi had no financial conflicts to disclose. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – The future of hyperhidrosis therapy may be microwave ablation, a Food and Drug Administration–cleared method of destroying sweat glands noninvasively.
"We’ve seen that it’s effective in about 90% of patients, the patients are really satisfied, and the side effects are transient," Dr. Nazanin Saedi said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Larger patient numbers and more long-term data are needed before the therapy becomes common in clinical practice, but it shouldn’t take long to accumulate such data, because the proprietary miraDry microwave treatment system (marketed by Miramar Labs) is much in demand by patients, noted Dr. Saedi of Thomas Jefferson University, Philadelphia.
An estimated 4.3 million Americans have axillary hyperhidrosis, Dr. Saedi said. Numerous treatment options are available, but most have significant drawbacks, she noted. First-line therapy is topical aluminum chloride, with or without salicylic acid; irritant dermatitis is a common complaint.
Second-line options include iontophoresis, which requires treatments two or three times per week and can be quite painful, Dr. Saedi said. Botulinum toxin A injections are highly effective, but they must be repeated every 4-6 months, and can cause weakness of the underlying muscles. Oral medications include anticholinergic agents and alpha2-adrenergic drugs, all with side effects many patients find problematic.
Before microwave therapy, surgical procedures were the only means of destroying sweat glands, and surgery is considered third-line therapy, Dr. Saedi said.
"Now that we have the newer technologies, I think surgery will become even more rare," she predicted.
The study that earned the miraDry device its Food and Drug Administration marketing clearance was a blinded trial in which 120 patients were randomized 2:1 to microwave therapy or a sham procedure. At baseline, all participants were rated 3 or 4 on the 1-4 Hyperhidrosis Disease Severity Scale (HDSS). At a follow-up point of 30 days, 89% of patients in the treatment group met the primary endpoint – an HDSS score of 1 or 2 – compared with 54% of controls. Roughly 70% of patients in the microwave treatment group still had an HDSS score of 1-2 at 12 months (Dermatol. Surg. 2012;38:185-91).
However, that study involved an earlier-generation, less efficient device, Dr. Saedi noted. In an open-label study of 31 patients treated with the current-generation, commercially available miraDry device, all subjects had a baseline HDSS score of 3 or 4. Twelve months after patients received 1-3 treatment sessions, 90% had an HDSS score of 1 or 2. In addition, 90% of patients had at least a 50% reduction in gravimetrically measured axillary sweat, and 85% had at least a 5-point improvement on the Dermatologic Life Quality Index (Dermatol. Surg. 2012;38:728-35).
In the microwave treatment procedure, an applicator is placed on the skin surface to deliver focused energy to the dermal-fat interface, where sweat glands are located. Microwaves are preferentially absorbed by tissues with high water content, such as sweat glands, and poorly absorbed by fat. A cooling system keeps the heat at the lower skin layer. A heat dome at about 60° C is created along the dermal-fat junction, resulting in sweat gland thermolysis, Dr. Saedi explained.
Two hours prior to treatment, the patient takes 800 mg of ibuprofen. A grid of temporary markings is placed over the area to be treated, and the dermis is anesthetized with injections of a 1:1 mix of 1% plain lidocaine and 1% lidocaine plus epinephrine at 1-cm intervals.
Microwave therapy entails two treatment sessions roughly 3 months apart. The first typically takes 60-90 minutes, the second 30-60 minutes.
Dr. Saedi said swelling, bruising, discomfort, and altered sensation in and around the treatment area are all common side effects of microwave therapy, but all are short-term. Less common side effects include swelling extending beyond the treatment area, a tight banding sensation in the axilla, and arm and hand numbness lasting less than 24 hours, she said.
One audience member who has treated 130 patients via microwave therapy reported that one of them developed a significant brachial nerve palsy that has lasted for 5 months. As a consequence, the commenter said he now routinely employs tumescent anesthesia in an effort to steer clear of the axillary nerve bundles. But Dr. Suzanne L. Kilmer said she doubts that will prevent the occasional case of brachial nerve palsy, which she believes is the result of variant anatomy.
"We’ve done the procedure on many thin people, and not had brachial nerve palsy be an issue, so I’m not sure that tumescent anesthesia will make a difference," said Dr. Kilmer of the University of California, Davis.
Pulsed ultrasound is currently under investigation as a means of treating hyperhidrosis through destruction of sweat glands, session moderator Dr. Christopher B. Zachary said in response to an audience question. "This would be an off-label indication right now, but you can absolutely hit that region with an ultrasound device and cause heating. It’s quite likely that ultrasound will be used extensively in exactly the same way as microwave, and probably with the same efficacy," predicted Dr. Zachary of the University of California, Irvine.
Dr. Zachary is a consultant to and has received research funding from numerous laser and other medical device companies, as has Dr. Kilmer. Dr. Saedi had no financial conflicts to disclose. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – The future of hyperhidrosis therapy may be microwave ablation, a Food and Drug Administration–cleared method of destroying sweat glands noninvasively.
"We’ve seen that it’s effective in about 90% of patients, the patients are really satisfied, and the side effects are transient," Dr. Nazanin Saedi said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Larger patient numbers and more long-term data are needed before the therapy becomes common in clinical practice, but it shouldn’t take long to accumulate such data, because the proprietary miraDry microwave treatment system (marketed by Miramar Labs) is much in demand by patients, noted Dr. Saedi of Thomas Jefferson University, Philadelphia.
An estimated 4.3 million Americans have axillary hyperhidrosis, Dr. Saedi said. Numerous treatment options are available, but most have significant drawbacks, she noted. First-line therapy is topical aluminum chloride, with or without salicylic acid; irritant dermatitis is a common complaint.
Second-line options include iontophoresis, which requires treatments two or three times per week and can be quite painful, Dr. Saedi said. Botulinum toxin A injections are highly effective, but they must be repeated every 4-6 months, and can cause weakness of the underlying muscles. Oral medications include anticholinergic agents and alpha2-adrenergic drugs, all with side effects many patients find problematic.
Before microwave therapy, surgical procedures were the only means of destroying sweat glands, and surgery is considered third-line therapy, Dr. Saedi said.
"Now that we have the newer technologies, I think surgery will become even more rare," she predicted.
The study that earned the miraDry device its Food and Drug Administration marketing clearance was a blinded trial in which 120 patients were randomized 2:1 to microwave therapy or a sham procedure. At baseline, all participants were rated 3 or 4 on the 1-4 Hyperhidrosis Disease Severity Scale (HDSS). At a follow-up point of 30 days, 89% of patients in the treatment group met the primary endpoint – an HDSS score of 1 or 2 – compared with 54% of controls. Roughly 70% of patients in the microwave treatment group still had an HDSS score of 1-2 at 12 months (Dermatol. Surg. 2012;38:185-91).
However, that study involved an earlier-generation, less efficient device, Dr. Saedi noted. In an open-label study of 31 patients treated with the current-generation, commercially available miraDry device, all subjects had a baseline HDSS score of 3 or 4. Twelve months after patients received 1-3 treatment sessions, 90% had an HDSS score of 1 or 2. In addition, 90% of patients had at least a 50% reduction in gravimetrically measured axillary sweat, and 85% had at least a 5-point improvement on the Dermatologic Life Quality Index (Dermatol. Surg. 2012;38:728-35).
In the microwave treatment procedure, an applicator is placed on the skin surface to deliver focused energy to the dermal-fat interface, where sweat glands are located. Microwaves are preferentially absorbed by tissues with high water content, such as sweat glands, and poorly absorbed by fat. A cooling system keeps the heat at the lower skin layer. A heat dome at about 60° C is created along the dermal-fat junction, resulting in sweat gland thermolysis, Dr. Saedi explained.
Two hours prior to treatment, the patient takes 800 mg of ibuprofen. A grid of temporary markings is placed over the area to be treated, and the dermis is anesthetized with injections of a 1:1 mix of 1% plain lidocaine and 1% lidocaine plus epinephrine at 1-cm intervals.
Microwave therapy entails two treatment sessions roughly 3 months apart. The first typically takes 60-90 minutes, the second 30-60 minutes.
Dr. Saedi said swelling, bruising, discomfort, and altered sensation in and around the treatment area are all common side effects of microwave therapy, but all are short-term. Less common side effects include swelling extending beyond the treatment area, a tight banding sensation in the axilla, and arm and hand numbness lasting less than 24 hours, she said.
One audience member who has treated 130 patients via microwave therapy reported that one of them developed a significant brachial nerve palsy that has lasted for 5 months. As a consequence, the commenter said he now routinely employs tumescent anesthesia in an effort to steer clear of the axillary nerve bundles. But Dr. Suzanne L. Kilmer said she doubts that will prevent the occasional case of brachial nerve palsy, which she believes is the result of variant anatomy.
"We’ve done the procedure on many thin people, and not had brachial nerve palsy be an issue, so I’m not sure that tumescent anesthesia will make a difference," said Dr. Kilmer of the University of California, Davis.
Pulsed ultrasound is currently under investigation as a means of treating hyperhidrosis through destruction of sweat glands, session moderator Dr. Christopher B. Zachary said in response to an audience question. "This would be an off-label indication right now, but you can absolutely hit that region with an ultrasound device and cause heating. It’s quite likely that ultrasound will be used extensively in exactly the same way as microwave, and probably with the same efficacy," predicted Dr. Zachary of the University of California, Irvine.
Dr. Zachary is a consultant to and has received research funding from numerous laser and other medical device companies, as has Dr. Kilmer. Dr. Saedi had no financial conflicts to disclose. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
'Slow Mohs' advised for lentigo maligna
MAUI, HAWAII – "Slow Mohs" has gained near-universal acceptance among skin cancer specialists as a definitive surgical technique for complete removal of lentigo maligna melanoma while simultaneously sparing normal tissue, according to Dr. Ellen Marmur of Mount Sinai School of Medicine, New York.
The big advantage that slow Mohs has over standard wide local excision with 0.5- to 1-cm margins is a 5-year cure rate approaching 100%. In contrast, standard excision has a recurrence rate of up to 20%, she said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Slow Mohs is a modified form of Mohs micrographic surgery. The surgery compares with conventional Mohs: It is staged, margin-controlled excision. But in slow Mohs, rush permanent sections are sent off to the pathologist rather than the frozen sections integral to conventional Mohs.
Dr. Marmur relies upon slow Mohs, with "bread-loafing" of the central tumor by the pathologist, because the permanent sections better preserve the tumor’s microscopic features. Interpreting atypical melanocytes in frozen sections can be quite a challenge. However, she added, some Mohs surgeons have found that using rapid immunostains also markedly improves the sensitivity and specificity of frozen sections in lentigo maligna surgery.
Slow Mohs takes place over the course of days, she said. "Basically, you do your Wood’s lamp to define the lesion diameter, you measure out your margins, you excise the tumor, pack the area with a bandage, and send the patient home. You rush your pathology, and you don’t do any reconstruction until you get the margins clear."
A pathology report that comes back stating narrow margins are present is "a heart stopper," she added.
"You have the option of observing the area if the margin is clear but the tumor was close to the margin. That’s a good approach for an elderly patient or when the lentigo maligna was in a cosmetically important area."
Lentigo maligna melanoma accounts for 4% of all cases of melanoma. It typically arises on sun-damaged skin in individuals in their 70s or older. Common sites include the malar area, forehead, nose, and temple. The differential diagnosis includes seborrheic keratosis, pigmented actinic keratosis, and pigmented nevus.
Lentigo maligna becomes lentigo maligna melanoma when malignant melanoma cells invade the dermis and deeper appendages. Roughly 5% of lentigo malignas eventually progress to invasive melanoma, according to Dr. Marmur. Typically, a lentigo maligna undergoes extended gradual horizontal growth before beginning a vertical growth phase.
"It spreads like an oil slick for many years," Dr. Marmur said at the seminar.
Established treatment modalities for patients who aren’t surgical candidates include cryotherapy, radiotherapy, and topical imiquimod 5%. All have disadvantages, including high 5-year recurrence rates.
Dr. Marmur noted that a newer nonsurgical therapy drawing considerable interest involves off-label use of topical combination therapy with imiquimod and tazarotene gel. The concept is to use the topical retinoid to disrupt the stratum corneum in order to enhance imiquimod penetration, thereby achieving a greater inflammatory response than possible with imiquimod alone.
Initial data have been published by researchers at the University of Utah, Salt Lake City. They randomized 90 patients with 91 lentigo malignas to imiquimod 5% cream applied 5 days per week for 3 months or to the imiquimod regimen plus tazarotene 0.1% gel on the other 2 days per week. After 3 months of topical therapy, patients underwent conservative staged excision with frozen section analysis with Melan A immunostaining to confirm negative margins.
Of those treated with dual topical therapy for 3 months, 29 of 37 lesions (78%) had complete responses with no residual lentigo maligna at the time of staged excision. So did 27 of 42 (64%) treated with imiquimod alone (Arch. Dermatol. 2012;148:592-6).
The modest difference in outcome was not significant (P = .17). Nevertheless, the Utah investigators wrote that topical pretreatment appears to reduce surgical defect sizes, an important consideration in lentigo maligna because the lesions are often large and located on cosmetically sensitive facial sites. At the patient’s first visit, the researchers saucerize the entire tumor to remove all visible evidence of lentigo maligna and nip in the bud any invasive element that might be present. One month later, after the wound has healed by secondary intention, the patient begins topical imiquimod therapy 5 days per week. If no inflammation is observed, tazarotene gel is added on the other 2 days per week. After 3 months of topical therapy, the patient goes off treatment for 2 months so the inflammatory response can subside. Then a staged excision is performed with 2-mm margins around the perimeter of the original tumor outline.
"This is an interesting thought, but it’s still not considered standard of care," Dr. Marmur commented in describing the Utah research.
Session chair Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan-Kettering Cancer Center in New York, said he and his colleagues, like the Utah group, are using a lot of topical imiquimod as adjunctive therapy for lentigo maligna.
"Because we’re a referral center, we see all those patients whose melanomas unfortunately do recur at the edge of large surgeries. We see a lot of patients whose local surgeons have chased around their faces for a certain amount of time. So I think the general concept of the cure rates of surgery is a bit overstated. I’m not trying to sell an off-label use of a drug, but I think sometimes imiquimod can be very helpful," Dr. Halpern said.
He offered a clinical pearl in diagnosing lentigo maligna: Consider a broad shave biopsy of a suspicious lesion rather than a deeper, smaller-diameter biopsy.
"Sampling errors are a real problem with lentigo maligna. We learned early on that if you take a large lentigo maligna and do multiple biopsies of the same lesion and send them to what we think are our very expert pathologists, on average, we get two or three back that were melanoma and two or three back that were pigmented actinic keratoses. The sampling error tends to be more in the horizontal than in the depth for these very early lesions," according to Dr. Halpern.
Dr. Marmur and Dr. Halpern reported having no relevant financial interests. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – "Slow Mohs" has gained near-universal acceptance among skin cancer specialists as a definitive surgical technique for complete removal of lentigo maligna melanoma while simultaneously sparing normal tissue, according to Dr. Ellen Marmur of Mount Sinai School of Medicine, New York.
The big advantage that slow Mohs has over standard wide local excision with 0.5- to 1-cm margins is a 5-year cure rate approaching 100%. In contrast, standard excision has a recurrence rate of up to 20%, she said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Slow Mohs is a modified form of Mohs micrographic surgery. The surgery compares with conventional Mohs: It is staged, margin-controlled excision. But in slow Mohs, rush permanent sections are sent off to the pathologist rather than the frozen sections integral to conventional Mohs.
Dr. Marmur relies upon slow Mohs, with "bread-loafing" of the central tumor by the pathologist, because the permanent sections better preserve the tumor’s microscopic features. Interpreting atypical melanocytes in frozen sections can be quite a challenge. However, she added, some Mohs surgeons have found that using rapid immunostains also markedly improves the sensitivity and specificity of frozen sections in lentigo maligna surgery.
Slow Mohs takes place over the course of days, she said. "Basically, you do your Wood’s lamp to define the lesion diameter, you measure out your margins, you excise the tumor, pack the area with a bandage, and send the patient home. You rush your pathology, and you don’t do any reconstruction until you get the margins clear."
A pathology report that comes back stating narrow margins are present is "a heart stopper," she added.
"You have the option of observing the area if the margin is clear but the tumor was close to the margin. That’s a good approach for an elderly patient or when the lentigo maligna was in a cosmetically important area."
Lentigo maligna melanoma accounts for 4% of all cases of melanoma. It typically arises on sun-damaged skin in individuals in their 70s or older. Common sites include the malar area, forehead, nose, and temple. The differential diagnosis includes seborrheic keratosis, pigmented actinic keratosis, and pigmented nevus.
Lentigo maligna becomes lentigo maligna melanoma when malignant melanoma cells invade the dermis and deeper appendages. Roughly 5% of lentigo malignas eventually progress to invasive melanoma, according to Dr. Marmur. Typically, a lentigo maligna undergoes extended gradual horizontal growth before beginning a vertical growth phase.
"It spreads like an oil slick for many years," Dr. Marmur said at the seminar.
Established treatment modalities for patients who aren’t surgical candidates include cryotherapy, radiotherapy, and topical imiquimod 5%. All have disadvantages, including high 5-year recurrence rates.
Dr. Marmur noted that a newer nonsurgical therapy drawing considerable interest involves off-label use of topical combination therapy with imiquimod and tazarotene gel. The concept is to use the topical retinoid to disrupt the stratum corneum in order to enhance imiquimod penetration, thereby achieving a greater inflammatory response than possible with imiquimod alone.
Initial data have been published by researchers at the University of Utah, Salt Lake City. They randomized 90 patients with 91 lentigo malignas to imiquimod 5% cream applied 5 days per week for 3 months or to the imiquimod regimen plus tazarotene 0.1% gel on the other 2 days per week. After 3 months of topical therapy, patients underwent conservative staged excision with frozen section analysis with Melan A immunostaining to confirm negative margins.
Of those treated with dual topical therapy for 3 months, 29 of 37 lesions (78%) had complete responses with no residual lentigo maligna at the time of staged excision. So did 27 of 42 (64%) treated with imiquimod alone (Arch. Dermatol. 2012;148:592-6).
The modest difference in outcome was not significant (P = .17). Nevertheless, the Utah investigators wrote that topical pretreatment appears to reduce surgical defect sizes, an important consideration in lentigo maligna because the lesions are often large and located on cosmetically sensitive facial sites. At the patient’s first visit, the researchers saucerize the entire tumor to remove all visible evidence of lentigo maligna and nip in the bud any invasive element that might be present. One month later, after the wound has healed by secondary intention, the patient begins topical imiquimod therapy 5 days per week. If no inflammation is observed, tazarotene gel is added on the other 2 days per week. After 3 months of topical therapy, the patient goes off treatment for 2 months so the inflammatory response can subside. Then a staged excision is performed with 2-mm margins around the perimeter of the original tumor outline.
"This is an interesting thought, but it’s still not considered standard of care," Dr. Marmur commented in describing the Utah research.
Session chair Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan-Kettering Cancer Center in New York, said he and his colleagues, like the Utah group, are using a lot of topical imiquimod as adjunctive therapy for lentigo maligna.
"Because we’re a referral center, we see all those patients whose melanomas unfortunately do recur at the edge of large surgeries. We see a lot of patients whose local surgeons have chased around their faces for a certain amount of time. So I think the general concept of the cure rates of surgery is a bit overstated. I’m not trying to sell an off-label use of a drug, but I think sometimes imiquimod can be very helpful," Dr. Halpern said.
He offered a clinical pearl in diagnosing lentigo maligna: Consider a broad shave biopsy of a suspicious lesion rather than a deeper, smaller-diameter biopsy.
"Sampling errors are a real problem with lentigo maligna. We learned early on that if you take a large lentigo maligna and do multiple biopsies of the same lesion and send them to what we think are our very expert pathologists, on average, we get two or three back that were melanoma and two or three back that were pigmented actinic keratoses. The sampling error tends to be more in the horizontal than in the depth for these very early lesions," according to Dr. Halpern.
Dr. Marmur and Dr. Halpern reported having no relevant financial interests. SDEF and this news organization are owned by the same parent company.
MAUI, HAWAII – "Slow Mohs" has gained near-universal acceptance among skin cancer specialists as a definitive surgical technique for complete removal of lentigo maligna melanoma while simultaneously sparing normal tissue, according to Dr. Ellen Marmur of Mount Sinai School of Medicine, New York.
The big advantage that slow Mohs has over standard wide local excision with 0.5- to 1-cm margins is a 5-year cure rate approaching 100%. In contrast, standard excision has a recurrence rate of up to 20%, she said at the Hawaii Dermatology Seminar, sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
Slow Mohs is a modified form of Mohs micrographic surgery. The surgery compares with conventional Mohs: It is staged, margin-controlled excision. But in slow Mohs, rush permanent sections are sent off to the pathologist rather than the frozen sections integral to conventional Mohs.
Dr. Marmur relies upon slow Mohs, with "bread-loafing" of the central tumor by the pathologist, because the permanent sections better preserve the tumor’s microscopic features. Interpreting atypical melanocytes in frozen sections can be quite a challenge. However, she added, some Mohs surgeons have found that using rapid immunostains also markedly improves the sensitivity and specificity of frozen sections in lentigo maligna surgery.
Slow Mohs takes place over the course of days, she said. "Basically, you do your Wood’s lamp to define the lesion diameter, you measure out your margins, you excise the tumor, pack the area with a bandage, and send the patient home. You rush your pathology, and you don’t do any reconstruction until you get the margins clear."
A pathology report that comes back stating narrow margins are present is "a heart stopper," she added.
"You have the option of observing the area if the margin is clear but the tumor was close to the margin. That’s a good approach for an elderly patient or when the lentigo maligna was in a cosmetically important area."
Lentigo maligna melanoma accounts for 4% of all cases of melanoma. It typically arises on sun-damaged skin in individuals in their 70s or older. Common sites include the malar area, forehead, nose, and temple. The differential diagnosis includes seborrheic keratosis, pigmented actinic keratosis, and pigmented nevus.
Lentigo maligna becomes lentigo maligna melanoma when malignant melanoma cells invade the dermis and deeper appendages. Roughly 5% of lentigo malignas eventually progress to invasive melanoma, according to Dr. Marmur. Typically, a lentigo maligna undergoes extended gradual horizontal growth before beginning a vertical growth phase.
"It spreads like an oil slick for many years," Dr. Marmur said at the seminar.
Established treatment modalities for patients who aren’t surgical candidates include cryotherapy, radiotherapy, and topical imiquimod 5%. All have disadvantages, including high 5-year recurrence rates.
Dr. Marmur noted that a newer nonsurgical therapy drawing considerable interest involves off-label use of topical combination therapy with imiquimod and tazarotene gel. The concept is to use the topical retinoid to disrupt the stratum corneum in order to enhance imiquimod penetration, thereby achieving a greater inflammatory response than possible with imiquimod alone.
Initial data have been published by researchers at the University of Utah, Salt Lake City. They randomized 90 patients with 91 lentigo malignas to imiquimod 5% cream applied 5 days per week for 3 months or to the imiquimod regimen plus tazarotene 0.1% gel on the other 2 days per week. After 3 months of topical therapy, patients underwent conservative staged excision with frozen section analysis with Melan A immunostaining to confirm negative margins.
Of those treated with dual topical therapy for 3 months, 29 of 37 lesions (78%) had complete responses with no residual lentigo maligna at the time of staged excision. So did 27 of 42 (64%) treated with imiquimod alone (Arch. Dermatol. 2012;148:592-6).
The modest difference in outcome was not significant (P = .17). Nevertheless, the Utah investigators wrote that topical pretreatment appears to reduce surgical defect sizes, an important consideration in lentigo maligna because the lesions are often large and located on cosmetically sensitive facial sites. At the patient’s first visit, the researchers saucerize the entire tumor to remove all visible evidence of lentigo maligna and nip in the bud any invasive element that might be present. One month later, after the wound has healed by secondary intention, the patient begins topical imiquimod therapy 5 days per week. If no inflammation is observed, tazarotene gel is added on the other 2 days per week. After 3 months of topical therapy, the patient goes off treatment for 2 months so the inflammatory response can subside. Then a staged excision is performed with 2-mm margins around the perimeter of the original tumor outline.
"This is an interesting thought, but it’s still not considered standard of care," Dr. Marmur commented in describing the Utah research.
Session chair Dr. Allan C. Halpern, chief of the dermatology service at Memorial Sloan-Kettering Cancer Center in New York, said he and his colleagues, like the Utah group, are using a lot of topical imiquimod as adjunctive therapy for lentigo maligna.
"Because we’re a referral center, we see all those patients whose melanomas unfortunately do recur at the edge of large surgeries. We see a lot of patients whose local surgeons have chased around their faces for a certain amount of time. So I think the general concept of the cure rates of surgery is a bit overstated. I’m not trying to sell an off-label use of a drug, but I think sometimes imiquimod can be very helpful," Dr. Halpern said.
He offered a clinical pearl in diagnosing lentigo maligna: Consider a broad shave biopsy of a suspicious lesion rather than a deeper, smaller-diameter biopsy.
"Sampling errors are a real problem with lentigo maligna. We learned early on that if you take a large lentigo maligna and do multiple biopsies of the same lesion and send them to what we think are our very expert pathologists, on average, we get two or three back that were melanoma and two or three back that were pigmented actinic keratoses. The sampling error tends to be more in the horizontal than in the depth for these very early lesions," according to Dr. Halpern.
Dr. Marmur and Dr. Halpern reported having no relevant financial interests. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Obesity epidemic's hidden cost: Hospital staff injuries
INDIANAPOLIS – A major hidden cost of the obesity epidemic is the physical toll it takes on hospital nurses and other employees in helping to move heavy patients. The cost of these workplace injuries is skyrocketing, not only in terms of direct medical care bills, but also in terms of work absenteeism, activity restrictions, retraining, and employee dissatisfaction and fear.
The solution at one large tertiary academic medical center in eastern North Carolina has been to create specially trained two-person lift teams available 24/7. The results of a pilot study have been impressive, particularly in light of the fact that nothing else hospital officials tried earlier – including spending more than $1.5 million for motorized patient ceiling lifts – had any significant impact, Dr. Walter J. Pories said at the annual meeting of the American Surgical Association.
Hospital employee injuries incurred in handling obese patients are an issue that has until now been largely beneath physicians’ radar.
"I don’t think anybody here would ask two nurses to pick up a washing machine, or a 400-lb calf, or an 800-lb lathe, and yet whether we like it or not we ask our staff to move patients with such weights daily," observed Dr. Pories, professor of surgery at East Carolina University in Greenville, N.C.
He practices at Vidant Medical Center, a 909-bed hospital where administrators noticed back in 2005 that employee workplace compensation claims for injuries during patient transport were rapidly escalating. In that year, 98 staff experienced patient-handling injuries, resulting in 670 lost workdays and 3,022 restricted workdays.
Hospital officials tried several interventions. First came an intensive education program on safe lifting and patient mobilization conducted by an ergonomist.
"It didn’t make a bit of difference," Dr. Pories recalled.
Indeed, during 2008 there were 2,141 lost workdays due to these types of injuries, more than triple the number in 2005.
Next came a big investment in ceiling-mounted motorized lifts. The impact was minimal: roughly a 10% reduction in staff injuries. Problems with the lifts abounded. They broke. Some staff had difficulty operating them. But the biggest issue was that the rooms with the lifts weren’t always available because the hospital is always fully occupied.
Dr. Pories credited the nursing staff with providing the leadership in developing what he calls the Vidant Medical Center lift team model. Coverage is available 24/7. There are three teams of two individuals on duty during the day and two teams of two at night. They work 12-hour shifts. There is one supervisor for 23 lift team technicians, including three women. They are paid an average of $10 per hour, which the surgeon called "a pretty good wage in eastern North Carolina."
Individuals must pass a rigorous physical assessment before they can join the lift team. Members take a 3-week orientation program in which they learn safe lift techniques, skin and wound care, and infection control. They have their own uniforms to foster esprit de corps.
Many other organizations had tried using lift teams, with disappointing results. That’s because they limited the teams to the day shift or scheduled transfers, according to Dr. Pories.
"The real problems happen late at night," he said.
Dr. Pories and his coinvestigators conducted a pilot study in which they utilized the lift teams in the five hospital units having the highest staff injury rates due to patient-handling mishaps. The lift team was called to those units for moving patients who weighed more than 200 lb, had pressure ulcers, or who were at risk for pressure ulcers by virtue of a Braden scale score of 18 or less.
During the pilot study, 8 employee injuries resulted from patient handling in the five test units, for a rate of 0.134 injuries per 1,000 patient-days, compared with 71 injuries and a rate of 0.319 injuries per 1,000 patient-days in control units.
Once use of the lift teams expanded hospital-wide, the result was a 39% drop in employee injuries due to patient handling, as well as a 43% reduction in hospital-acquired pressure ulcers. Multiple staff surveys have shown 90%-99% satisfaction rates with the lift teams as having improved the workplace while demonstrating that the hospital cares about employee safety.
The total direct and indirect cost of staff injuries due to patient handling was approximately $2.76 million per year prior to introduction of the lift teams. The lift teams provided an estimated $423,152 in savings due to fewer staff injuries and hospital-acquired pressure ulcers, even after factoring in team salaries and equipment, Dr. Pories reported.
The next step in this project will be to document whether the presence of the lift teams also has resulted in fewer patient injuries, he added.
Discussant Dr. Philip R. Schauer commented that if the favorable Vidant experience with dedicated lift teams can be confirmed elsewhere, this is the type of program that should be widely instituted all across the country.
"It’s quite extraordinary to improve patient care and employee health while at the same time reducing overall cost," noted Dr. Schauer, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic.
Discussant Dr. William B. Inabnet III said many malpractice insurance carriers are now offering hospitals incentives for reduced premiums for implementation of best-practices clinical pathways for management of obese patients.
"I think your work will support that type of mission. It’s a win-win for all parties involved," said Dr. Inabnet, professor of surgery at Mt. Sinai Hospital in New York.
Dr. Pories reported having no conflicts of interest.
INDIANAPOLIS – A major hidden cost of the obesity epidemic is the physical toll it takes on hospital nurses and other employees in helping to move heavy patients. The cost of these workplace injuries is skyrocketing, not only in terms of direct medical care bills, but also in terms of work absenteeism, activity restrictions, retraining, and employee dissatisfaction and fear.
The solution at one large tertiary academic medical center in eastern North Carolina has been to create specially trained two-person lift teams available 24/7. The results of a pilot study have been impressive, particularly in light of the fact that nothing else hospital officials tried earlier – including spending more than $1.5 million for motorized patient ceiling lifts – had any significant impact, Dr. Walter J. Pories said at the annual meeting of the American Surgical Association.
Hospital employee injuries incurred in handling obese patients are an issue that has until now been largely beneath physicians’ radar.
"I don’t think anybody here would ask two nurses to pick up a washing machine, or a 400-lb calf, or an 800-lb lathe, and yet whether we like it or not we ask our staff to move patients with such weights daily," observed Dr. Pories, professor of surgery at East Carolina University in Greenville, N.C.
He practices at Vidant Medical Center, a 909-bed hospital where administrators noticed back in 2005 that employee workplace compensation claims for injuries during patient transport were rapidly escalating. In that year, 98 staff experienced patient-handling injuries, resulting in 670 lost workdays and 3,022 restricted workdays.
Hospital officials tried several interventions. First came an intensive education program on safe lifting and patient mobilization conducted by an ergonomist.
"It didn’t make a bit of difference," Dr. Pories recalled.
Indeed, during 2008 there were 2,141 lost workdays due to these types of injuries, more than triple the number in 2005.
Next came a big investment in ceiling-mounted motorized lifts. The impact was minimal: roughly a 10% reduction in staff injuries. Problems with the lifts abounded. They broke. Some staff had difficulty operating them. But the biggest issue was that the rooms with the lifts weren’t always available because the hospital is always fully occupied.
Dr. Pories credited the nursing staff with providing the leadership in developing what he calls the Vidant Medical Center lift team model. Coverage is available 24/7. There are three teams of two individuals on duty during the day and two teams of two at night. They work 12-hour shifts. There is one supervisor for 23 lift team technicians, including three women. They are paid an average of $10 per hour, which the surgeon called "a pretty good wage in eastern North Carolina."
Individuals must pass a rigorous physical assessment before they can join the lift team. Members take a 3-week orientation program in which they learn safe lift techniques, skin and wound care, and infection control. They have their own uniforms to foster esprit de corps.
Many other organizations had tried using lift teams, with disappointing results. That’s because they limited the teams to the day shift or scheduled transfers, according to Dr. Pories.
"The real problems happen late at night," he said.
Dr. Pories and his coinvestigators conducted a pilot study in which they utilized the lift teams in the five hospital units having the highest staff injury rates due to patient-handling mishaps. The lift team was called to those units for moving patients who weighed more than 200 lb, had pressure ulcers, or who were at risk for pressure ulcers by virtue of a Braden scale score of 18 or less.
During the pilot study, 8 employee injuries resulted from patient handling in the five test units, for a rate of 0.134 injuries per 1,000 patient-days, compared with 71 injuries and a rate of 0.319 injuries per 1,000 patient-days in control units.
Once use of the lift teams expanded hospital-wide, the result was a 39% drop in employee injuries due to patient handling, as well as a 43% reduction in hospital-acquired pressure ulcers. Multiple staff surveys have shown 90%-99% satisfaction rates with the lift teams as having improved the workplace while demonstrating that the hospital cares about employee safety.
The total direct and indirect cost of staff injuries due to patient handling was approximately $2.76 million per year prior to introduction of the lift teams. The lift teams provided an estimated $423,152 in savings due to fewer staff injuries and hospital-acquired pressure ulcers, even after factoring in team salaries and equipment, Dr. Pories reported.
The next step in this project will be to document whether the presence of the lift teams also has resulted in fewer patient injuries, he added.
Discussant Dr. Philip R. Schauer commented that if the favorable Vidant experience with dedicated lift teams can be confirmed elsewhere, this is the type of program that should be widely instituted all across the country.
"It’s quite extraordinary to improve patient care and employee health while at the same time reducing overall cost," noted Dr. Schauer, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic.
Discussant Dr. William B. Inabnet III said many malpractice insurance carriers are now offering hospitals incentives for reduced premiums for implementation of best-practices clinical pathways for management of obese patients.
"I think your work will support that type of mission. It’s a win-win for all parties involved," said Dr. Inabnet, professor of surgery at Mt. Sinai Hospital in New York.
Dr. Pories reported having no conflicts of interest.
INDIANAPOLIS – A major hidden cost of the obesity epidemic is the physical toll it takes on hospital nurses and other employees in helping to move heavy patients. The cost of these workplace injuries is skyrocketing, not only in terms of direct medical care bills, but also in terms of work absenteeism, activity restrictions, retraining, and employee dissatisfaction and fear.
The solution at one large tertiary academic medical center in eastern North Carolina has been to create specially trained two-person lift teams available 24/7. The results of a pilot study have been impressive, particularly in light of the fact that nothing else hospital officials tried earlier – including spending more than $1.5 million for motorized patient ceiling lifts – had any significant impact, Dr. Walter J. Pories said at the annual meeting of the American Surgical Association.
Hospital employee injuries incurred in handling obese patients are an issue that has until now been largely beneath physicians’ radar.
"I don’t think anybody here would ask two nurses to pick up a washing machine, or a 400-lb calf, or an 800-lb lathe, and yet whether we like it or not we ask our staff to move patients with such weights daily," observed Dr. Pories, professor of surgery at East Carolina University in Greenville, N.C.
He practices at Vidant Medical Center, a 909-bed hospital where administrators noticed back in 2005 that employee workplace compensation claims for injuries during patient transport were rapidly escalating. In that year, 98 staff experienced patient-handling injuries, resulting in 670 lost workdays and 3,022 restricted workdays.
Hospital officials tried several interventions. First came an intensive education program on safe lifting and patient mobilization conducted by an ergonomist.
"It didn’t make a bit of difference," Dr. Pories recalled.
Indeed, during 2008 there were 2,141 lost workdays due to these types of injuries, more than triple the number in 2005.
Next came a big investment in ceiling-mounted motorized lifts. The impact was minimal: roughly a 10% reduction in staff injuries. Problems with the lifts abounded. They broke. Some staff had difficulty operating them. But the biggest issue was that the rooms with the lifts weren’t always available because the hospital is always fully occupied.
Dr. Pories credited the nursing staff with providing the leadership in developing what he calls the Vidant Medical Center lift team model. Coverage is available 24/7. There are three teams of two individuals on duty during the day and two teams of two at night. They work 12-hour shifts. There is one supervisor for 23 lift team technicians, including three women. They are paid an average of $10 per hour, which the surgeon called "a pretty good wage in eastern North Carolina."
Individuals must pass a rigorous physical assessment before they can join the lift team. Members take a 3-week orientation program in which they learn safe lift techniques, skin and wound care, and infection control. They have their own uniforms to foster esprit de corps.
Many other organizations had tried using lift teams, with disappointing results. That’s because they limited the teams to the day shift or scheduled transfers, according to Dr. Pories.
"The real problems happen late at night," he said.
Dr. Pories and his coinvestigators conducted a pilot study in which they utilized the lift teams in the five hospital units having the highest staff injury rates due to patient-handling mishaps. The lift team was called to those units for moving patients who weighed more than 200 lb, had pressure ulcers, or who were at risk for pressure ulcers by virtue of a Braden scale score of 18 or less.
During the pilot study, 8 employee injuries resulted from patient handling in the five test units, for a rate of 0.134 injuries per 1,000 patient-days, compared with 71 injuries and a rate of 0.319 injuries per 1,000 patient-days in control units.
Once use of the lift teams expanded hospital-wide, the result was a 39% drop in employee injuries due to patient handling, as well as a 43% reduction in hospital-acquired pressure ulcers. Multiple staff surveys have shown 90%-99% satisfaction rates with the lift teams as having improved the workplace while demonstrating that the hospital cares about employee safety.
The total direct and indirect cost of staff injuries due to patient handling was approximately $2.76 million per year prior to introduction of the lift teams. The lift teams provided an estimated $423,152 in savings due to fewer staff injuries and hospital-acquired pressure ulcers, even after factoring in team salaries and equipment, Dr. Pories reported.
The next step in this project will be to document whether the presence of the lift teams also has resulted in fewer patient injuries, he added.
Discussant Dr. Philip R. Schauer commented that if the favorable Vidant experience with dedicated lift teams can be confirmed elsewhere, this is the type of program that should be widely instituted all across the country.
"It’s quite extraordinary to improve patient care and employee health while at the same time reducing overall cost," noted Dr. Schauer, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic.
Discussant Dr. William B. Inabnet III said many malpractice insurance carriers are now offering hospitals incentives for reduced premiums for implementation of best-practices clinical pathways for management of obese patients.
"I think your work will support that type of mission. It’s a win-win for all parties involved," said Dr. Inabnet, professor of surgery at Mt. Sinai Hospital in New York.
Dr. Pories reported having no conflicts of interest.
AT THE ASA ANNUAL MEETING
Diabetes 'cure' holds up 6 years after bariatric surgery
INDIANAPOLIS – At 6 years of follow-up data, bariatric surgical cures of type 2 diabetes are holding steady in a single-center series of 217 patients.
"We see sustained weight loss, particularly in Roux-en-Y gastric bypass patients, 5-9 years after surgery." Diabetes remitted in 50% of patients, according to Dr. Stacy A. Brethauer of the Cleveland Clinic. "Bariatric surgery also achieved excellent long-term control of other cardiovascular risk factors; and diabetic nephropathy improved or stabilized."
Of 217 obese type 2 diabetic patients followed for a median of 6 years after bariatric surgery, 24% have been "cured." They have maintained a hemoglobin A1c level below 6% and fasting blood glucose values below 100 mg/dL for more than 5 years while off all antidiabetic medications, Dr. Brethauer reported at the annual meeting of the American Surgical Association.
Another 26% of patients have had partial remissions, meaning they maintained target HbA1c and fasting blood glucose levels off antidiabetic medications for more than 1 year but less than 5 years. In addition, 34% of patients were classified as "improved" based upon an absolute 1% or more reduction in HbA1c, a drop in fasting blood glucose in excess of 25 mg/dL, and a halving in the dose of antidiabetic medication for at least 1 year.
Diabetes recurred in 19% of subjects. Recurrence was defined as a return to an HbA1c of 6.5% or more or a fasting blood glucose level of at least 126 mg/dL.
The overall study population improved from a mean body mass index of 49 kg/m2 before surgery to 37 kg/m2 at 6 years after surgery. The mean HbA1c was 7.5% at baseline and 6.5% at 6 years. Fasting blood glucose at 6 years was a mean of 41.6 mg/dL lower than it was before surgery. LDL cholesterol levels were down by a mean of 10.1 mg/dL and HDL levels were up by 9.8 mg/dL. Systolic blood pressure was reduced by a mean of 10.9 mm Hg, with diastolic blood pressure was down by 3.2 mm Hg at 6 years after surgery.
Preoperatively, all but 5% of patients were on any antidiabetes medications; at 6 years after bariatric surgery, 54% were not on any antidiabetes drugs.
At baseline, 3% of subjects met all three key metabolic goals for diabetic patients as defined by the American Diabetes Association: an HbA1c below 7%, blood pressure below 130/80 mm Hg, and an LDL level below 100 mg/dL. At 6 years after surgery, 28% of patients met these goals. That result is particularly impressive in light of other studies that indicate 13% of the U.S. diabetic population as a whole meet all three goals.
The bariatric surgery group had a mean Framingham 10-year cardiovascular risk score of 28% preoperatively and of 22% at long-term followup.
The incidence of diabetic nephropathy among patients with type 2 diabetes is typically 2%-4% per year. Yet of 40 gastric bypass recipients known to have a normal urinary albumin-to-creatinine ratio preoperatively, only 1 developed macroalbuminuria and 1 microalbuminuria during a mean of 6 years postsurgical follow-up.
Also impressive, albuminuria regressed in 10 of the 19 gastric bypass recipients known to have the disorder at baseline. Albuminuria remained stable in the other 9 patients over the course of 6 years.
Mean excess weight loss at 1-2 years of follow-up was 61% in the 162 patients who underwent gastric bypass, 50% in the 23 with sleeve gastrectomy, and 30% in patients who had a gastric banding procedure.
In a multivariate analysis adjusted for baseline clinical characteristics, the significant predictors of diabetes remission following bariatric surgery were greater excess weight loss, preoperative diabetes duration of less than 5 years, and having a gastric bypass operation rather than sleeve gastrectomy or a gastric banding procedure.
Dr. Brethauer said this study, taken together with the findings of an earlier randomized clinical trial by the same investigators (N. Engl. J. Med. 2012;366:1567-76), conveys a clear message : "Bariatric surgery can induce a significant and sustainable remission of type 2 diabetes and other metabolic risk factors in obese patients and should be considered early in the course of the disease."
Discussant Dr. Walter J. Pories called Dr. Brethauer’s study "a really important contribution." He added that it’s high time for nonsurgeons to get on board.
"One in four adults over age 65 in this country has diabetes. One would have thought our medical colleagues would be ecstatic at the news that an operation on the gut – a safe procedure that can be done in about an hour – could produce full and durable remission of diabetes with complete prevention of amputations, blindness, and kidney failure. But that hasn’t been the case. All we’ve heard are cries for more and more evidence," said Dr. Pories, professor of surgery at East Carolina University, Greenville, N.C.
He added that in his view, Dr. Brethauer and his colleagues set the bar too high in requiring a sustained HbA1c below 6% as the definition of disease cure when the American Diabetes Association uses a figure of 7%. He noted that if the investigators had accepted the ADA metric, their combined cure/partial remission rate would have been considerably greater than the 50% figure they reported.
Dr. Brethauer replied that he and his coinvestigators chose a cutoff of 6% in order to make a point.
"When we raise this issue of ‘cure,’ which is still quite controversial and somewhat provocative, particularly with our endocrinology colleagues, I think we have to find the strictest and most conservative criteria that we can," he explained. "We continue to provide data in support of the concept that this is a surgically treated disease. It’s a major paradigm shift for our endocrinology colleagues to accept. I think it’s going to take a generation of endocrinologists before it’s embraced."
He reported that he serves as a consultant to Ethicon Endosurgery and Apollo Endosurgery.
INDIANAPOLIS – At 6 years of follow-up data, bariatric surgical cures of type 2 diabetes are holding steady in a single-center series of 217 patients.
"We see sustained weight loss, particularly in Roux-en-Y gastric bypass patients, 5-9 years after surgery." Diabetes remitted in 50% of patients, according to Dr. Stacy A. Brethauer of the Cleveland Clinic. "Bariatric surgery also achieved excellent long-term control of other cardiovascular risk factors; and diabetic nephropathy improved or stabilized."
Of 217 obese type 2 diabetic patients followed for a median of 6 years after bariatric surgery, 24% have been "cured." They have maintained a hemoglobin A1c level below 6% and fasting blood glucose values below 100 mg/dL for more than 5 years while off all antidiabetic medications, Dr. Brethauer reported at the annual meeting of the American Surgical Association.
Another 26% of patients have had partial remissions, meaning they maintained target HbA1c and fasting blood glucose levels off antidiabetic medications for more than 1 year but less than 5 years. In addition, 34% of patients were classified as "improved" based upon an absolute 1% or more reduction in HbA1c, a drop in fasting blood glucose in excess of 25 mg/dL, and a halving in the dose of antidiabetic medication for at least 1 year.
Diabetes recurred in 19% of subjects. Recurrence was defined as a return to an HbA1c of 6.5% or more or a fasting blood glucose level of at least 126 mg/dL.
The overall study population improved from a mean body mass index of 49 kg/m2 before surgery to 37 kg/m2 at 6 years after surgery. The mean HbA1c was 7.5% at baseline and 6.5% at 6 years. Fasting blood glucose at 6 years was a mean of 41.6 mg/dL lower than it was before surgery. LDL cholesterol levels were down by a mean of 10.1 mg/dL and HDL levels were up by 9.8 mg/dL. Systolic blood pressure was reduced by a mean of 10.9 mm Hg, with diastolic blood pressure was down by 3.2 mm Hg at 6 years after surgery.
Preoperatively, all but 5% of patients were on any antidiabetes medications; at 6 years after bariatric surgery, 54% were not on any antidiabetes drugs.
At baseline, 3% of subjects met all three key metabolic goals for diabetic patients as defined by the American Diabetes Association: an HbA1c below 7%, blood pressure below 130/80 mm Hg, and an LDL level below 100 mg/dL. At 6 years after surgery, 28% of patients met these goals. That result is particularly impressive in light of other studies that indicate 13% of the U.S. diabetic population as a whole meet all three goals.
The bariatric surgery group had a mean Framingham 10-year cardiovascular risk score of 28% preoperatively and of 22% at long-term followup.
The incidence of diabetic nephropathy among patients with type 2 diabetes is typically 2%-4% per year. Yet of 40 gastric bypass recipients known to have a normal urinary albumin-to-creatinine ratio preoperatively, only 1 developed macroalbuminuria and 1 microalbuminuria during a mean of 6 years postsurgical follow-up.
Also impressive, albuminuria regressed in 10 of the 19 gastric bypass recipients known to have the disorder at baseline. Albuminuria remained stable in the other 9 patients over the course of 6 years.
Mean excess weight loss at 1-2 years of follow-up was 61% in the 162 patients who underwent gastric bypass, 50% in the 23 with sleeve gastrectomy, and 30% in patients who had a gastric banding procedure.
In a multivariate analysis adjusted for baseline clinical characteristics, the significant predictors of diabetes remission following bariatric surgery were greater excess weight loss, preoperative diabetes duration of less than 5 years, and having a gastric bypass operation rather than sleeve gastrectomy or a gastric banding procedure.
Dr. Brethauer said this study, taken together with the findings of an earlier randomized clinical trial by the same investigators (N. Engl. J. Med. 2012;366:1567-76), conveys a clear message : "Bariatric surgery can induce a significant and sustainable remission of type 2 diabetes and other metabolic risk factors in obese patients and should be considered early in the course of the disease."
Discussant Dr. Walter J. Pories called Dr. Brethauer’s study "a really important contribution." He added that it’s high time for nonsurgeons to get on board.
"One in four adults over age 65 in this country has diabetes. One would have thought our medical colleagues would be ecstatic at the news that an operation on the gut – a safe procedure that can be done in about an hour – could produce full and durable remission of diabetes with complete prevention of amputations, blindness, and kidney failure. But that hasn’t been the case. All we’ve heard are cries for more and more evidence," said Dr. Pories, professor of surgery at East Carolina University, Greenville, N.C.
He added that in his view, Dr. Brethauer and his colleagues set the bar too high in requiring a sustained HbA1c below 6% as the definition of disease cure when the American Diabetes Association uses a figure of 7%. He noted that if the investigators had accepted the ADA metric, their combined cure/partial remission rate would have been considerably greater than the 50% figure they reported.
Dr. Brethauer replied that he and his coinvestigators chose a cutoff of 6% in order to make a point.
"When we raise this issue of ‘cure,’ which is still quite controversial and somewhat provocative, particularly with our endocrinology colleagues, I think we have to find the strictest and most conservative criteria that we can," he explained. "We continue to provide data in support of the concept that this is a surgically treated disease. It’s a major paradigm shift for our endocrinology colleagues to accept. I think it’s going to take a generation of endocrinologists before it’s embraced."
He reported that he serves as a consultant to Ethicon Endosurgery and Apollo Endosurgery.
INDIANAPOLIS – At 6 years of follow-up data, bariatric surgical cures of type 2 diabetes are holding steady in a single-center series of 217 patients.
"We see sustained weight loss, particularly in Roux-en-Y gastric bypass patients, 5-9 years after surgery." Diabetes remitted in 50% of patients, according to Dr. Stacy A. Brethauer of the Cleveland Clinic. "Bariatric surgery also achieved excellent long-term control of other cardiovascular risk factors; and diabetic nephropathy improved or stabilized."
Of 217 obese type 2 diabetic patients followed for a median of 6 years after bariatric surgery, 24% have been "cured." They have maintained a hemoglobin A1c level below 6% and fasting blood glucose values below 100 mg/dL for more than 5 years while off all antidiabetic medications, Dr. Brethauer reported at the annual meeting of the American Surgical Association.
Another 26% of patients have had partial remissions, meaning they maintained target HbA1c and fasting blood glucose levels off antidiabetic medications for more than 1 year but less than 5 years. In addition, 34% of patients were classified as "improved" based upon an absolute 1% or more reduction in HbA1c, a drop in fasting blood glucose in excess of 25 mg/dL, and a halving in the dose of antidiabetic medication for at least 1 year.
Diabetes recurred in 19% of subjects. Recurrence was defined as a return to an HbA1c of 6.5% or more or a fasting blood glucose level of at least 126 mg/dL.
The overall study population improved from a mean body mass index of 49 kg/m2 before surgery to 37 kg/m2 at 6 years after surgery. The mean HbA1c was 7.5% at baseline and 6.5% at 6 years. Fasting blood glucose at 6 years was a mean of 41.6 mg/dL lower than it was before surgery. LDL cholesterol levels were down by a mean of 10.1 mg/dL and HDL levels were up by 9.8 mg/dL. Systolic blood pressure was reduced by a mean of 10.9 mm Hg, with diastolic blood pressure was down by 3.2 mm Hg at 6 years after surgery.
Preoperatively, all but 5% of patients were on any antidiabetes medications; at 6 years after bariatric surgery, 54% were not on any antidiabetes drugs.
At baseline, 3% of subjects met all three key metabolic goals for diabetic patients as defined by the American Diabetes Association: an HbA1c below 7%, blood pressure below 130/80 mm Hg, and an LDL level below 100 mg/dL. At 6 years after surgery, 28% of patients met these goals. That result is particularly impressive in light of other studies that indicate 13% of the U.S. diabetic population as a whole meet all three goals.
The bariatric surgery group had a mean Framingham 10-year cardiovascular risk score of 28% preoperatively and of 22% at long-term followup.
The incidence of diabetic nephropathy among patients with type 2 diabetes is typically 2%-4% per year. Yet of 40 gastric bypass recipients known to have a normal urinary albumin-to-creatinine ratio preoperatively, only 1 developed macroalbuminuria and 1 microalbuminuria during a mean of 6 years postsurgical follow-up.
Also impressive, albuminuria regressed in 10 of the 19 gastric bypass recipients known to have the disorder at baseline. Albuminuria remained stable in the other 9 patients over the course of 6 years.
Mean excess weight loss at 1-2 years of follow-up was 61% in the 162 patients who underwent gastric bypass, 50% in the 23 with sleeve gastrectomy, and 30% in patients who had a gastric banding procedure.
In a multivariate analysis adjusted for baseline clinical characteristics, the significant predictors of diabetes remission following bariatric surgery were greater excess weight loss, preoperative diabetes duration of less than 5 years, and having a gastric bypass operation rather than sleeve gastrectomy or a gastric banding procedure.
Dr. Brethauer said this study, taken together with the findings of an earlier randomized clinical trial by the same investigators (N. Engl. J. Med. 2012;366:1567-76), conveys a clear message : "Bariatric surgery can induce a significant and sustainable remission of type 2 diabetes and other metabolic risk factors in obese patients and should be considered early in the course of the disease."
Discussant Dr. Walter J. Pories called Dr. Brethauer’s study "a really important contribution." He added that it’s high time for nonsurgeons to get on board.
"One in four adults over age 65 in this country has diabetes. One would have thought our medical colleagues would be ecstatic at the news that an operation on the gut – a safe procedure that can be done in about an hour – could produce full and durable remission of diabetes with complete prevention of amputations, blindness, and kidney failure. But that hasn’t been the case. All we’ve heard are cries for more and more evidence," said Dr. Pories, professor of surgery at East Carolina University, Greenville, N.C.
He added that in his view, Dr. Brethauer and his colleagues set the bar too high in requiring a sustained HbA1c below 6% as the definition of disease cure when the American Diabetes Association uses a figure of 7%. He noted that if the investigators had accepted the ADA metric, their combined cure/partial remission rate would have been considerably greater than the 50% figure they reported.
Dr. Brethauer replied that he and his coinvestigators chose a cutoff of 6% in order to make a point.
"When we raise this issue of ‘cure,’ which is still quite controversial and somewhat provocative, particularly with our endocrinology colleagues, I think we have to find the strictest and most conservative criteria that we can," he explained. "We continue to provide data in support of the concept that this is a surgically treated disease. It’s a major paradigm shift for our endocrinology colleagues to accept. I think it’s going to take a generation of endocrinologists before it’s embraced."
He reported that he serves as a consultant to Ethicon Endosurgery and Apollo Endosurgery.
AT THE ASA ANNUAL MEETING
Major finding: At a median of 6 years after bariatric surgery, 24% of patients have maintained an HbA1c below 6% and fasting blood glucose values below 100 mg/dL for more than 5 years while off all antidiabetic medications.
Data source: This is an ongoing retrospective single-center study in which 217 obese patients with type 2 diabetes have been followed for 5-9 years after bariatric surgery.
Disclosures: This study is sponsored by the Cleveland Clinic. The presenter reported serving as a consultant to Ethicon Endosurgery and Apollo Endosurgery.
REMINDER: Early eplerenone reduces BNP in STEMI
SAN FRANCISCO – Early administration of the aldosterone blocker eplerenone improved cardiovascular outcomes in patients presenting with ST-elevation myocardial infarction without concomitant heart failure.
In the randomized, double-blind REMINDER trial, starting eplerenone within 24 hours after symptom onset without first checking serum potassium, as per the study protocol, proved reassuringly safe. There was no significant increase in hyperkalemia compared with placebo. Hypokalemia, defined as a serum potassium level less than 3.5 mEq/L, occurred in 1.4% of the eplerenone group and in 5.5% of controls (P = .0002). Rates of other adverse events were similar in the two study arms, Dr. Gilles Montalescot reported at the annual meeting of the American College of Cardiology.
REMINDER was an international clinical trial involving 1,012 patients presenting with acute STEMI with no history or current signs or symptoms of heart failure and with a left ventricular ejection fraction of at least 40%. A series of studies have shown that the more elevated the early aldosterone in acute STEMI, the worse the prognosis in terms of mortality, new or worsening heart failure, and ventricular fibrillation.
This was a low-risk STEMI population. Patients with renal failure were ineligible. Rates of guideline-recommended medications were strikingly high. Primary percutaneous coronary intervention was the means of myocardial reperfusion in 85% of participants.
The primary study endpoint during a mean 10.5 months of follow-up was a composite of cardiovascular death, hospitalization due to diagnosed heart failure, sustained ventricular tachycardia or fibrillation, an LVEF of 40% after more than 1 month, and an elevated level of brain natriuretic peptide (BNP) or N-terminal prohormone of BNP (NT-proBNP) occurring after 1 month of follow-up.
The rate of the primary endpoint was 18.4% in the eplerenone group, compared with 29.6% with placebo, for a significant 43% relative risk reduction, said Dr. Montalescot, chair of the REMINDER steering committee and professor of cardiology at Pitié-Salpêtrière Hospital, Paris.
The difference in the composite outcome was driven by a sharply lower incidence of elevated BNP/NT-proBNP in the eplerenone group: 16% as compared to 25.9% in controls. The other elements of the composite endpoint didn’t differ significantly between the two groups, although they tended to be lower in the eplerenone group.
Aldosterone has a multitude of cardiotoxic effects. It increases sodium retention, arrhythmogenic depletion of potassium and calcium, cardiac myocyte necrosis, myocardial remodeling, and endothelial dysfunction.
Earlier, in the landmark, randomized EPHESUS trial, investigators showed that prescribing eplerenone in patients with left ventricular systolic dysfunction and heart failure present 3-14 days after an acute MI resulted in a 17% reduction in cardiovascular mortality relative to placebo (N. Engl. J. Med. 2003;348:1309-21). Subsequently, in the EMPHASIS-HF trial eplerenone achieved a 24% reduction in cardiovascular mortality when prescribed for patients with left ventricular systolic dysfunction and mild, NYHA class II heart failure post-MI (N. Engl. J. Med. 2011;364:11-21). The goal in REMINDER was to preempt that systolic dysfunction from arising.
Dr. Montalescot noted that the ongoing 1,600-patient ALBATROSS (Aldosterone Blockade Early After Acute MI) trial, sponsored by the Public Assistance Hospital of Paris, is looking at the use of spironolactone in moderate-risk acute STEMI and has 6-month hard clinical endpoints.
The REMINDER trial was sponsored by Pfizer. Dr. Montalescot reported having no financial conflicts.
The long-term clinical relevance of an elevated BNP is questionable and constitutes "the fly in the ointment" in the REMINDER trial. The biomarker is not a rock-solid predictor of later major adverse clinical events.

|
|
Eplerenone is already recommended in the European Society of Cardiology and American heart failure guidelines based upon firm evidence of clinical benefit. Yet this recommendation is the least followed, and hardly ever used in practice. In light of that situation, it’s unlikely that reliance on a biomarker endpoint will persuade many physicians to extend use of the aldosterone blocking agent to acute STEMI patients who don’t actually have heart failure.
Dr. Montalescot has said that to demonstrate a 20% reduction in the dual endpoint of mortality or onset of heart failure in low-risk STEMI patients would require 10,000 patients in each study arm, a preclusively costly endeavor.
The combined rate of the ‘hard’ clinical endpoints of cardiovascular mortality, heart failure, or severe ventricular arrhythmia in REMINDER was 3.2% in the control group and 1.8% with eplerenone therapy. Given eplerenone’s cost, prescribing it early on in all acute STEMI patients is unlikely when 97% of them do not appear to benefit.
Dr. E. Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University, Durham, N.C. Dr. Ohman was the discussant of the study at ACC 13.
The long-term clinical relevance of an elevated BNP is questionable and constitutes "the fly in the ointment" in the REMINDER trial. The biomarker is not a rock-solid predictor of later major adverse clinical events.

|
|
Eplerenone is already recommended in the European Society of Cardiology and American heart failure guidelines based upon firm evidence of clinical benefit. Yet this recommendation is the least followed, and hardly ever used in practice. In light of that situation, it’s unlikely that reliance on a biomarker endpoint will persuade many physicians to extend use of the aldosterone blocking agent to acute STEMI patients who don’t actually have heart failure.
Dr. Montalescot has said that to demonstrate a 20% reduction in the dual endpoint of mortality or onset of heart failure in low-risk STEMI patients would require 10,000 patients in each study arm, a preclusively costly endeavor.
The combined rate of the ‘hard’ clinical endpoints of cardiovascular mortality, heart failure, or severe ventricular arrhythmia in REMINDER was 3.2% in the control group and 1.8% with eplerenone therapy. Given eplerenone’s cost, prescribing it early on in all acute STEMI patients is unlikely when 97% of them do not appear to benefit.
Dr. E. Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University, Durham, N.C. Dr. Ohman was the discussant of the study at ACC 13.
The long-term clinical relevance of an elevated BNP is questionable and constitutes "the fly in the ointment" in the REMINDER trial. The biomarker is not a rock-solid predictor of later major adverse clinical events.

|
|
Eplerenone is already recommended in the European Society of Cardiology and American heart failure guidelines based upon firm evidence of clinical benefit. Yet this recommendation is the least followed, and hardly ever used in practice. In light of that situation, it’s unlikely that reliance on a biomarker endpoint will persuade many physicians to extend use of the aldosterone blocking agent to acute STEMI patients who don’t actually have heart failure.
Dr. Montalescot has said that to demonstrate a 20% reduction in the dual endpoint of mortality or onset of heart failure in low-risk STEMI patients would require 10,000 patients in each study arm, a preclusively costly endeavor.
The combined rate of the ‘hard’ clinical endpoints of cardiovascular mortality, heart failure, or severe ventricular arrhythmia in REMINDER was 3.2% in the control group and 1.8% with eplerenone therapy. Given eplerenone’s cost, prescribing it early on in all acute STEMI patients is unlikely when 97% of them do not appear to benefit.
Dr. E. Magnus Ohman is professor of medicine and director of the program for advanced coronary disease at Duke University, Durham, N.C. Dr. Ohman was the discussant of the study at ACC 13.
SAN FRANCISCO – Early administration of the aldosterone blocker eplerenone improved cardiovascular outcomes in patients presenting with ST-elevation myocardial infarction without concomitant heart failure.
In the randomized, double-blind REMINDER trial, starting eplerenone within 24 hours after symptom onset without first checking serum potassium, as per the study protocol, proved reassuringly safe. There was no significant increase in hyperkalemia compared with placebo. Hypokalemia, defined as a serum potassium level less than 3.5 mEq/L, occurred in 1.4% of the eplerenone group and in 5.5% of controls (P = .0002). Rates of other adverse events were similar in the two study arms, Dr. Gilles Montalescot reported at the annual meeting of the American College of Cardiology.
REMINDER was an international clinical trial involving 1,012 patients presenting with acute STEMI with no history or current signs or symptoms of heart failure and with a left ventricular ejection fraction of at least 40%. A series of studies have shown that the more elevated the early aldosterone in acute STEMI, the worse the prognosis in terms of mortality, new or worsening heart failure, and ventricular fibrillation.
This was a low-risk STEMI population. Patients with renal failure were ineligible. Rates of guideline-recommended medications were strikingly high. Primary percutaneous coronary intervention was the means of myocardial reperfusion in 85% of participants.
The primary study endpoint during a mean 10.5 months of follow-up was a composite of cardiovascular death, hospitalization due to diagnosed heart failure, sustained ventricular tachycardia or fibrillation, an LVEF of 40% after more than 1 month, and an elevated level of brain natriuretic peptide (BNP) or N-terminal prohormone of BNP (NT-proBNP) occurring after 1 month of follow-up.
The rate of the primary endpoint was 18.4% in the eplerenone group, compared with 29.6% with placebo, for a significant 43% relative risk reduction, said Dr. Montalescot, chair of the REMINDER steering committee and professor of cardiology at Pitié-Salpêtrière Hospital, Paris.
The difference in the composite outcome was driven by a sharply lower incidence of elevated BNP/NT-proBNP in the eplerenone group: 16% as compared to 25.9% in controls. The other elements of the composite endpoint didn’t differ significantly between the two groups, although they tended to be lower in the eplerenone group.
Aldosterone has a multitude of cardiotoxic effects. It increases sodium retention, arrhythmogenic depletion of potassium and calcium, cardiac myocyte necrosis, myocardial remodeling, and endothelial dysfunction.
Earlier, in the landmark, randomized EPHESUS trial, investigators showed that prescribing eplerenone in patients with left ventricular systolic dysfunction and heart failure present 3-14 days after an acute MI resulted in a 17% reduction in cardiovascular mortality relative to placebo (N. Engl. J. Med. 2003;348:1309-21). Subsequently, in the EMPHASIS-HF trial eplerenone achieved a 24% reduction in cardiovascular mortality when prescribed for patients with left ventricular systolic dysfunction and mild, NYHA class II heart failure post-MI (N. Engl. J. Med. 2011;364:11-21). The goal in REMINDER was to preempt that systolic dysfunction from arising.
Dr. Montalescot noted that the ongoing 1,600-patient ALBATROSS (Aldosterone Blockade Early After Acute MI) trial, sponsored by the Public Assistance Hospital of Paris, is looking at the use of spironolactone in moderate-risk acute STEMI and has 6-month hard clinical endpoints.
The REMINDER trial was sponsored by Pfizer. Dr. Montalescot reported having no financial conflicts.
SAN FRANCISCO – Early administration of the aldosterone blocker eplerenone improved cardiovascular outcomes in patients presenting with ST-elevation myocardial infarction without concomitant heart failure.
In the randomized, double-blind REMINDER trial, starting eplerenone within 24 hours after symptom onset without first checking serum potassium, as per the study protocol, proved reassuringly safe. There was no significant increase in hyperkalemia compared with placebo. Hypokalemia, defined as a serum potassium level less than 3.5 mEq/L, occurred in 1.4% of the eplerenone group and in 5.5% of controls (P = .0002). Rates of other adverse events were similar in the two study arms, Dr. Gilles Montalescot reported at the annual meeting of the American College of Cardiology.
REMINDER was an international clinical trial involving 1,012 patients presenting with acute STEMI with no history or current signs or symptoms of heart failure and with a left ventricular ejection fraction of at least 40%. A series of studies have shown that the more elevated the early aldosterone in acute STEMI, the worse the prognosis in terms of mortality, new or worsening heart failure, and ventricular fibrillation.
This was a low-risk STEMI population. Patients with renal failure were ineligible. Rates of guideline-recommended medications were strikingly high. Primary percutaneous coronary intervention was the means of myocardial reperfusion in 85% of participants.
The primary study endpoint during a mean 10.5 months of follow-up was a composite of cardiovascular death, hospitalization due to diagnosed heart failure, sustained ventricular tachycardia or fibrillation, an LVEF of 40% after more than 1 month, and an elevated level of brain natriuretic peptide (BNP) or N-terminal prohormone of BNP (NT-proBNP) occurring after 1 month of follow-up.
The rate of the primary endpoint was 18.4% in the eplerenone group, compared with 29.6% with placebo, for a significant 43% relative risk reduction, said Dr. Montalescot, chair of the REMINDER steering committee and professor of cardiology at Pitié-Salpêtrière Hospital, Paris.
The difference in the composite outcome was driven by a sharply lower incidence of elevated BNP/NT-proBNP in the eplerenone group: 16% as compared to 25.9% in controls. The other elements of the composite endpoint didn’t differ significantly between the two groups, although they tended to be lower in the eplerenone group.
Aldosterone has a multitude of cardiotoxic effects. It increases sodium retention, arrhythmogenic depletion of potassium and calcium, cardiac myocyte necrosis, myocardial remodeling, and endothelial dysfunction.
Earlier, in the landmark, randomized EPHESUS trial, investigators showed that prescribing eplerenone in patients with left ventricular systolic dysfunction and heart failure present 3-14 days after an acute MI resulted in a 17% reduction in cardiovascular mortality relative to placebo (N. Engl. J. Med. 2003;348:1309-21). Subsequently, in the EMPHASIS-HF trial eplerenone achieved a 24% reduction in cardiovascular mortality when prescribed for patients with left ventricular systolic dysfunction and mild, NYHA class II heart failure post-MI (N. Engl. J. Med. 2011;364:11-21). The goal in REMINDER was to preempt that systolic dysfunction from arising.
Dr. Montalescot noted that the ongoing 1,600-patient ALBATROSS (Aldosterone Blockade Early After Acute MI) trial, sponsored by the Public Assistance Hospital of Paris, is looking at the use of spironolactone in moderate-risk acute STEMI and has 6-month hard clinical endpoints.
The REMINDER trial was sponsored by Pfizer. Dr. Montalescot reported having no financial conflicts.
AT ACC 13
Major finding: The rate of the primary endpoint was 18.4% in the eplerenone group, compared with 29.6% with placebo, for a significant 43% relative risk reduction based on more than 10 months of follow-up.
Data source: REMINDER is an international, randomized, double-blind, placebo-controlled phase III trial involving 1,012 patients presenting with acute STEMI and no heart failure.
Disclosures: The REMINDER trial was funded by Pfizer. Dr. Montalescot reported having no financial conflicts.
Surgical fellowship directors: General surgery trainees arrive ill prepared
INDIANAPOLIS – The nation’s elite surgical educators are up in arms over reported widespread deficiencies in the skill set and judgment of recent graduates of 5-year general surgery residencies.
Many trainees arrive at surgical subspecialty fellowships unprepared in their basic skills, according to a detailed new survey of the nation’s subspecialty fellowship program directors. The underlying theme of the responses is that many fellows are pursuing fellowship positions to make up for inadequacies in their general surgery residency rather than to push their skills to the next level, according to Dr. Samer Mattar.
About 80% of graduating general surgery residents now seek fellowships to obtain advanced training in bariatric, colorectal, thoracic, hepatobiliary, or other surgical specialty areas.
"Many new fellows must gain basic and fundamental skills at the beginning of their fellowship before they can commence to benefit from the advanced skills that they originally came to obtain. The current high demand for fellowship training and the lack of readiness upon completion of general surgery residencies should be a call to action for all stakeholders in surgical training," Dr. Mattar said in presenting the survey results at the annual meeting of the American Surgical Association.
Constructive changes are afoot, according to Dr. Mattar of Indiana University, Indianapolis. Plans are well underway to change the fourth year of medical school so that students interested in a career in surgery can begin preparing for it. And there are also efforts to custom tailor the final year of general surgery residency so residents can focus on their planned fellowship year. Toward that end, the Fellowship Council has moved the fellowship match date up to June so residents who know they are fellowship-bound can put their fifth year to the best use.
The survey was conducted by the Fellowship Council, an umbrella organization in charge of standardizing curricula, accrediting programs, and matching residents to fellowships. The group distributed the surveys to all 145 subspecialty fellowship program directors and drew a 63% response rate. That’s considered high for such a lengthy survey, said Dr. Mattar, and is an indication of the importance educators place on the subject.
The survey assessed five key educational domains: professionalism, independent practice, psychomotor skills, expertise in their chosen disease state, and scholarly focus.
"Incoming fellows exhibited high levels of professionalism, but there were deficiencies in autonomy and independence, psychomotor abilities, and – most profoundly – in academics and scholarship," Dr. Mattar said.
Among the key survey findings:
• Incoming fellows were unable to independently perform half an hour of a major procedure, according to 43% of program directors.
• Of incoming fellows, 30% couldn’t independently perform basic operations such as laparoscopic cholecystectomy.
• Of incoming fellows, 56% were able to laparoscopically suture and tie knots properly; 26% couldn’t recognize anatomic planes through the laparoscope.
• One-quarter was deemed unable to recognize early signs of complications.
In other findings, nearly 40% of program directors said new fellows display a lack of "patient ownership." "We promote patient ownership in our programs. We are somewhat disappointed and dismayed that the fellows feel that the patient is part of a service and not their own," Dr. Mattar commented. Half of the program directors indicated their incoming fellows demonstrated independence in the operating room and on call, although they reported the fellows showed marked improvement in these areas as the year went on.
A large majority of program directors felt their fellows were disinterested in research and in advancing the field, even though, as Dr. Mattar noted, "This is a mandate in our curriculum."
He reported having no financial conflicts.
No one should be surprised by the Fellowship Council survey results. During the past decade, the failure rate on the American Board of Surgery’s oral exam has climbed steadily from 16% to 28%. At present, the percentage of examinees who fail either the oral or written American Board of Surgery exam the first time around is in the mid-30s. That’s arguably an absurd failure rate for a 5-year training program in a group of people who should have mastered the subject.
The 80-hour work limit has effectively subtracted 6-12 months from the general surgery residency, yet I do not believe this emotional and contentious issue is the explanation.
At present, the average number of operations done by a first-year resident is less than two per week, while second-year residents average two to three per week. Our residents are spending 80 hours a week while doing two or three operations per week, which arguably could be done in half a day. It would be hard to imagine a less efficient educational process.
Dr. Frank R. Lewis is executive director of the American Board of Surgery. He was an invited discussant of the presentation.
No one should be surprised by the Fellowship Council survey results. During the past decade, the failure rate on the American Board of Surgery’s oral exam has climbed steadily from 16% to 28%. At present, the percentage of examinees who fail either the oral or written American Board of Surgery exam the first time around is in the mid-30s. That’s arguably an absurd failure rate for a 5-year training program in a group of people who should have mastered the subject.
The 80-hour work limit has effectively subtracted 6-12 months from the general surgery residency, yet I do not believe this emotional and contentious issue is the explanation.
At present, the average number of operations done by a first-year resident is less than two per week, while second-year residents average two to three per week. Our residents are spending 80 hours a week while doing two or three operations per week, which arguably could be done in half a day. It would be hard to imagine a less efficient educational process.
Dr. Frank R. Lewis is executive director of the American Board of Surgery. He was an invited discussant of the presentation.
No one should be surprised by the Fellowship Council survey results. During the past decade, the failure rate on the American Board of Surgery’s oral exam has climbed steadily from 16% to 28%. At present, the percentage of examinees who fail either the oral or written American Board of Surgery exam the first time around is in the mid-30s. That’s arguably an absurd failure rate for a 5-year training program in a group of people who should have mastered the subject.
The 80-hour work limit has effectively subtracted 6-12 months from the general surgery residency, yet I do not believe this emotional and contentious issue is the explanation.
At present, the average number of operations done by a first-year resident is less than two per week, while second-year residents average two to three per week. Our residents are spending 80 hours a week while doing two or three operations per week, which arguably could be done in half a day. It would be hard to imagine a less efficient educational process.
Dr. Frank R. Lewis is executive director of the American Board of Surgery. He was an invited discussant of the presentation.
INDIANAPOLIS – The nation’s elite surgical educators are up in arms over reported widespread deficiencies in the skill set and judgment of recent graduates of 5-year general surgery residencies.
Many trainees arrive at surgical subspecialty fellowships unprepared in their basic skills, according to a detailed new survey of the nation’s subspecialty fellowship program directors. The underlying theme of the responses is that many fellows are pursuing fellowship positions to make up for inadequacies in their general surgery residency rather than to push their skills to the next level, according to Dr. Samer Mattar.
About 80% of graduating general surgery residents now seek fellowships to obtain advanced training in bariatric, colorectal, thoracic, hepatobiliary, or other surgical specialty areas.
"Many new fellows must gain basic and fundamental skills at the beginning of their fellowship before they can commence to benefit from the advanced skills that they originally came to obtain. The current high demand for fellowship training and the lack of readiness upon completion of general surgery residencies should be a call to action for all stakeholders in surgical training," Dr. Mattar said in presenting the survey results at the annual meeting of the American Surgical Association.
Constructive changes are afoot, according to Dr. Mattar of Indiana University, Indianapolis. Plans are well underway to change the fourth year of medical school so that students interested in a career in surgery can begin preparing for it. And there are also efforts to custom tailor the final year of general surgery residency so residents can focus on their planned fellowship year. Toward that end, the Fellowship Council has moved the fellowship match date up to June so residents who know they are fellowship-bound can put their fifth year to the best use.
The survey was conducted by the Fellowship Council, an umbrella organization in charge of standardizing curricula, accrediting programs, and matching residents to fellowships. The group distributed the surveys to all 145 subspecialty fellowship program directors and drew a 63% response rate. That’s considered high for such a lengthy survey, said Dr. Mattar, and is an indication of the importance educators place on the subject.
The survey assessed five key educational domains: professionalism, independent practice, psychomotor skills, expertise in their chosen disease state, and scholarly focus.
"Incoming fellows exhibited high levels of professionalism, but there were deficiencies in autonomy and independence, psychomotor abilities, and – most profoundly – in academics and scholarship," Dr. Mattar said.
Among the key survey findings:
• Incoming fellows were unable to independently perform half an hour of a major procedure, according to 43% of program directors.
• Of incoming fellows, 30% couldn’t independently perform basic operations such as laparoscopic cholecystectomy.
• Of incoming fellows, 56% were able to laparoscopically suture and tie knots properly; 26% couldn’t recognize anatomic planes through the laparoscope.
• One-quarter was deemed unable to recognize early signs of complications.
In other findings, nearly 40% of program directors said new fellows display a lack of "patient ownership." "We promote patient ownership in our programs. We are somewhat disappointed and dismayed that the fellows feel that the patient is part of a service and not their own," Dr. Mattar commented. Half of the program directors indicated their incoming fellows demonstrated independence in the operating room and on call, although they reported the fellows showed marked improvement in these areas as the year went on.
A large majority of program directors felt their fellows were disinterested in research and in advancing the field, even though, as Dr. Mattar noted, "This is a mandate in our curriculum."
He reported having no financial conflicts.
INDIANAPOLIS – The nation’s elite surgical educators are up in arms over reported widespread deficiencies in the skill set and judgment of recent graduates of 5-year general surgery residencies.
Many trainees arrive at surgical subspecialty fellowships unprepared in their basic skills, according to a detailed new survey of the nation’s subspecialty fellowship program directors. The underlying theme of the responses is that many fellows are pursuing fellowship positions to make up for inadequacies in their general surgery residency rather than to push their skills to the next level, according to Dr. Samer Mattar.
About 80% of graduating general surgery residents now seek fellowships to obtain advanced training in bariatric, colorectal, thoracic, hepatobiliary, or other surgical specialty areas.
"Many new fellows must gain basic and fundamental skills at the beginning of their fellowship before they can commence to benefit from the advanced skills that they originally came to obtain. The current high demand for fellowship training and the lack of readiness upon completion of general surgery residencies should be a call to action for all stakeholders in surgical training," Dr. Mattar said in presenting the survey results at the annual meeting of the American Surgical Association.
Constructive changes are afoot, according to Dr. Mattar of Indiana University, Indianapolis. Plans are well underway to change the fourth year of medical school so that students interested in a career in surgery can begin preparing for it. And there are also efforts to custom tailor the final year of general surgery residency so residents can focus on their planned fellowship year. Toward that end, the Fellowship Council has moved the fellowship match date up to June so residents who know they are fellowship-bound can put their fifth year to the best use.
The survey was conducted by the Fellowship Council, an umbrella organization in charge of standardizing curricula, accrediting programs, and matching residents to fellowships. The group distributed the surveys to all 145 subspecialty fellowship program directors and drew a 63% response rate. That’s considered high for such a lengthy survey, said Dr. Mattar, and is an indication of the importance educators place on the subject.
The survey assessed five key educational domains: professionalism, independent practice, psychomotor skills, expertise in their chosen disease state, and scholarly focus.
"Incoming fellows exhibited high levels of professionalism, but there were deficiencies in autonomy and independence, psychomotor abilities, and – most profoundly – in academics and scholarship," Dr. Mattar said.
Among the key survey findings:
• Incoming fellows were unable to independently perform half an hour of a major procedure, according to 43% of program directors.
• Of incoming fellows, 30% couldn’t independently perform basic operations such as laparoscopic cholecystectomy.
• Of incoming fellows, 56% were able to laparoscopically suture and tie knots properly; 26% couldn’t recognize anatomic planes through the laparoscope.
• One-quarter was deemed unable to recognize early signs of complications.
In other findings, nearly 40% of program directors said new fellows display a lack of "patient ownership." "We promote patient ownership in our programs. We are somewhat disappointed and dismayed that the fellows feel that the patient is part of a service and not their own," Dr. Mattar commented. Half of the program directors indicated their incoming fellows demonstrated independence in the operating room and on call, although they reported the fellows showed marked improvement in these areas as the year went on.
A large majority of program directors felt their fellows were disinterested in research and in advancing the field, even though, as Dr. Mattar noted, "This is a mandate in our curriculum."
He reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major finding: Surgical subspecialty program directors said that 43% of incoming fellows were unable to independently perform half an hour of a major procedure.
Data source: Survey responses from 91 of the nation’s 145 surgical subspecialty program directors.
Disclosures: The survey was conducted by the Fellowship Council, an umbrella organization with oversight over surgical subspecialty fellowships. The presenter reported having no financial conflicts.