User login
The push is on for universal influenza vaccines
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
The push is on for universal influenza vaccines
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Slap an ‘app’ on these five tough clinical decisions
ESTES PARK, COLO. – These days there’s a smartphone app for everything. But not all apps for clinical decision rules are truly helpful in making better patient care decisions in frontline primary care medicine, Dr. Robert E. Burke cautioned at a conference on internal medicine sponsored by the University of Colorado.
Five clinical decision rules have proven merit, each accessible via app as well as by an online website, he said. All five address medical issues that are common, important, and complex. What these rules have in common is excellent – not merely good – predictive value, thorough validation in multiple populations, a high likelihood that their use will influence patient management in individual cases, and an enthusiastic thumbs-up vote by physician-users.
These top-tier clinical decision rules address the following situations: determining which patients with community-acquired pneumonia can safely be managed at home, assessing whether a patient with chronic liver disease is likely to have advanced fibrosis or cirrhosis without resorting to liver biopsy, identifying who can be sent home safely after an episode of syncope, determining whether to prescribe oral anticoagulation for atrial fibrillation, and stratifying risk after a transient ischemic attack (TIA) or stroke.
These clinical decision rules are not a replacement for clinical judgment, but they do make risk/benefit calculations a lot easier, said Dr. Burke, a hospitalist at the Denver VA Medical Center and the university.
Triage of pneumonia patients. Pneumonia is the third most common reason for hospital admission as well as early readmission in the United States. So lots of different pneumonia severity scores have been developed. But few of them are any good, according to Dr. Burke.
Many of these subpar scores measure the likelihood that a patient with pneumonia will be admitted to the intensive care unit. That’s not what primary care physicians really want to know.
"Unfortunately, ICU admission rates vary widely in the U.S. and have nothing to do with how sick patients are or their comorbid conditions. It’s really not clear why the rates vary so much, but the predictive value of those scores is pretty poor, since ICU admission is a subjective thing," he explained.
The score he recommends is the Pneumonia Severity Index. It has been validated as an admission rule in several randomized trials totaling more than 4,000 patients. The score places patients into one of five risk categories. In the randomized trials, patients in the three lowest-risk categories had no negative outcomes when treated as outpatients. Plus, the use of the Pneumonia Severity Index reduced hospital admissions by up to 20%.
The score is calculated using 20 factors readily obtainable from the patient history, physical exam, and lab work. It’s a laborious calculation that’s perfect for an app. And the app exists: It’s called Simpli PSI.
Assessing fibrosis/cirrhosis risk in patients with chronic liver disease. Liver biopsy is painful, costly, decidedly not risk free, and often misleading due to the small tissue sample obtained. Transient elastography, a noninvasive ultrasound-based alternative, is promising but not ready for prime time. Yet knowing whether a patient has advanced fibrosis or cirrhosis is important in terms of prognosis, treatment, and surveillance strategy.
This is where the FIB-4 Index, a serum score for fibrosis, is of value. The FIB-4 Index is easily calculated from basic laboratory data. Findings from multiple studies have shown excellent discrimination regardless of whether patients had hepatitis C, hepatitis B, or nonalcoholic fatty liver disease.
"It has a c-statistic [equivalent to the area under the receiver operating characteristic curve] of 0.84. That’s about as good as it gets," according to the hospitalist.
The app is called Liver Calc. Like all the other apps Dr. Burke highlighted, it is available to iPhone users at the App Store.
"Or if you were to type in ‘FIB-4 Index’ on Google, you’ll find a website that will actually calculate the score online," he noted.
Whom to hospitalize for syncope. "Syncope is one of the most common things I see as a physician of in-patients," Dr. Burke said. "It accounts for maybe 1%-3% of emergency department visits and 6% of hospitalizations in the United States."
The San Francisco Syncope Rule is of proven value in helping to identify patients at low risk of serious outcomes in the 30 days following a syncopal episode. These are the patients who don’t need to undergo emergency department evaluation. The rule has been prospectively validated in tens of thousands of patients. It had a negative predictive value of 97% in a systematic review. The rule works less well in patients over age 65, where the likelihood of arrhythmia as the cause of syncope increases sharply. And the rule works best when applied only after a thorough history and physical exam aimed at finding a cause for the patient’s syncope.
"For example, the rule assumes you’ve already checked for hypoglycemia, a common cause of syncope," the hospitalist explained.
The San Francisco Syncope Rule is available in app form as Doctor Tools of the Trade.
Atrial fibrillation: To anticoagulate or not? This is a decision that entails balancing the reduction in stroke risk obtained with anticoagulation against the potential harm in the form of bleeding. The best tool for assessing the potential stroke-risk benefit is the CHA2DS2-VASc score, which represents a significant advance over the CHADS2 score, in Dr. Burke’s view.
"What the CHA2DS2-VASc score is really useful for, I think, is in putting people into low- or high-risk buckets. It takes people out of that intermediate-risk category in CHADS2," he explained.
It turns out that when physicians rely upon clinical intuition to estimate the risk of clinically important bleeding in patients on oral anticoagulation, they tend to overestimate the true risk. This observation has led to a proliferation of scales aimed at predicting who is likely to bleed when placed on warfarin. The best performer among them, regardless of whether the endpoint is any clinically relevant bleeding, major bleeding, or all-cause mortality, is the HAS-BLED score.
"I would encourage you to use both the CHA2DS2-VASc and HAS-BLED, and to use them at the same time. In some cases you’ll find the numbers are really discordant; there may be much more benefit than you thought, or much more risk," Dr. Burke said.
The focus-AF calculator is the app that will do the work.
Risk stratification after TIA or stroke. The ABCD2 score provides an estimate of stroke risk within 7 or 90 days after a TIA. This information helps establish the urgency of patient evaluation and risk factor management. A patient with an ABCD2 score of 4 or more should go straight to the emergency department, while a score of 3 or less indicates outpatient evaluation is appropriate. The Neuro Toolkit app will run the numbers.
Dr. Burke is keeping a watchful eye on a number of other clinical decision rules that, while promising, aren’t quite ready for prime time in his view. These include the FRAX score, the TIMI score, and the Marburg Heart Score. The Marburg score, for example, has been extensively validated as a tool to help primary care physicians decide whether chest pain is cardiac or noncardiac. But in the clinical trials, the score wasn’t compared to clinical intuition. That’s a problem.
"I think our clinical intuition here is relatively good. So I’d like to see data showing the rule adds something to clinical intuition before I recommend it," he said.
In a head-to-head comparative trial, the Pittsburgh Knee Rule outperformed the older Ottawa Knee Rule as an aid in figuring out who needs imaging after a knee injury. Impressive, in Dr. Burke’s view, but he’d like to see the results confirmed in a second study.
He reported having no financial conflicts.
ESTES PARK, COLO. – These days there’s a smartphone app for everything. But not all apps for clinical decision rules are truly helpful in making better patient care decisions in frontline primary care medicine, Dr. Robert E. Burke cautioned at a conference on internal medicine sponsored by the University of Colorado.
Five clinical decision rules have proven merit, each accessible via app as well as by an online website, he said. All five address medical issues that are common, important, and complex. What these rules have in common is excellent – not merely good – predictive value, thorough validation in multiple populations, a high likelihood that their use will influence patient management in individual cases, and an enthusiastic thumbs-up vote by physician-users.
These top-tier clinical decision rules address the following situations: determining which patients with community-acquired pneumonia can safely be managed at home, assessing whether a patient with chronic liver disease is likely to have advanced fibrosis or cirrhosis without resorting to liver biopsy, identifying who can be sent home safely after an episode of syncope, determining whether to prescribe oral anticoagulation for atrial fibrillation, and stratifying risk after a transient ischemic attack (TIA) or stroke.
These clinical decision rules are not a replacement for clinical judgment, but they do make risk/benefit calculations a lot easier, said Dr. Burke, a hospitalist at the Denver VA Medical Center and the university.
Triage of pneumonia patients. Pneumonia is the third most common reason for hospital admission as well as early readmission in the United States. So lots of different pneumonia severity scores have been developed. But few of them are any good, according to Dr. Burke.
Many of these subpar scores measure the likelihood that a patient with pneumonia will be admitted to the intensive care unit. That’s not what primary care physicians really want to know.
"Unfortunately, ICU admission rates vary widely in the U.S. and have nothing to do with how sick patients are or their comorbid conditions. It’s really not clear why the rates vary so much, but the predictive value of those scores is pretty poor, since ICU admission is a subjective thing," he explained.
The score he recommends is the Pneumonia Severity Index. It has been validated as an admission rule in several randomized trials totaling more than 4,000 patients. The score places patients into one of five risk categories. In the randomized trials, patients in the three lowest-risk categories had no negative outcomes when treated as outpatients. Plus, the use of the Pneumonia Severity Index reduced hospital admissions by up to 20%.
The score is calculated using 20 factors readily obtainable from the patient history, physical exam, and lab work. It’s a laborious calculation that’s perfect for an app. And the app exists: It’s called Simpli PSI.
Assessing fibrosis/cirrhosis risk in patients with chronic liver disease. Liver biopsy is painful, costly, decidedly not risk free, and often misleading due to the small tissue sample obtained. Transient elastography, a noninvasive ultrasound-based alternative, is promising but not ready for prime time. Yet knowing whether a patient has advanced fibrosis or cirrhosis is important in terms of prognosis, treatment, and surveillance strategy.
This is where the FIB-4 Index, a serum score for fibrosis, is of value. The FIB-4 Index is easily calculated from basic laboratory data. Findings from multiple studies have shown excellent discrimination regardless of whether patients had hepatitis C, hepatitis B, or nonalcoholic fatty liver disease.
"It has a c-statistic [equivalent to the area under the receiver operating characteristic curve] of 0.84. That’s about as good as it gets," according to the hospitalist.
The app is called Liver Calc. Like all the other apps Dr. Burke highlighted, it is available to iPhone users at the App Store.
"Or if you were to type in ‘FIB-4 Index’ on Google, you’ll find a website that will actually calculate the score online," he noted.
Whom to hospitalize for syncope. "Syncope is one of the most common things I see as a physician of in-patients," Dr. Burke said. "It accounts for maybe 1%-3% of emergency department visits and 6% of hospitalizations in the United States."
The San Francisco Syncope Rule is of proven value in helping to identify patients at low risk of serious outcomes in the 30 days following a syncopal episode. These are the patients who don’t need to undergo emergency department evaluation. The rule has been prospectively validated in tens of thousands of patients. It had a negative predictive value of 97% in a systematic review. The rule works less well in patients over age 65, where the likelihood of arrhythmia as the cause of syncope increases sharply. And the rule works best when applied only after a thorough history and physical exam aimed at finding a cause for the patient’s syncope.
"For example, the rule assumes you’ve already checked for hypoglycemia, a common cause of syncope," the hospitalist explained.
The San Francisco Syncope Rule is available in app form as Doctor Tools of the Trade.
Atrial fibrillation: To anticoagulate or not? This is a decision that entails balancing the reduction in stroke risk obtained with anticoagulation against the potential harm in the form of bleeding. The best tool for assessing the potential stroke-risk benefit is the CHA2DS2-VASc score, which represents a significant advance over the CHADS2 score, in Dr. Burke’s view.
"What the CHA2DS2-VASc score is really useful for, I think, is in putting people into low- or high-risk buckets. It takes people out of that intermediate-risk category in CHADS2," he explained.
It turns out that when physicians rely upon clinical intuition to estimate the risk of clinically important bleeding in patients on oral anticoagulation, they tend to overestimate the true risk. This observation has led to a proliferation of scales aimed at predicting who is likely to bleed when placed on warfarin. The best performer among them, regardless of whether the endpoint is any clinically relevant bleeding, major bleeding, or all-cause mortality, is the HAS-BLED score.
"I would encourage you to use both the CHA2DS2-VASc and HAS-BLED, and to use them at the same time. In some cases you’ll find the numbers are really discordant; there may be much more benefit than you thought, or much more risk," Dr. Burke said.
The focus-AF calculator is the app that will do the work.
Risk stratification after TIA or stroke. The ABCD2 score provides an estimate of stroke risk within 7 or 90 days after a TIA. This information helps establish the urgency of patient evaluation and risk factor management. A patient with an ABCD2 score of 4 or more should go straight to the emergency department, while a score of 3 or less indicates outpatient evaluation is appropriate. The Neuro Toolkit app will run the numbers.
Dr. Burke is keeping a watchful eye on a number of other clinical decision rules that, while promising, aren’t quite ready for prime time in his view. These include the FRAX score, the TIMI score, and the Marburg Heart Score. The Marburg score, for example, has been extensively validated as a tool to help primary care physicians decide whether chest pain is cardiac or noncardiac. But in the clinical trials, the score wasn’t compared to clinical intuition. That’s a problem.
"I think our clinical intuition here is relatively good. So I’d like to see data showing the rule adds something to clinical intuition before I recommend it," he said.
In a head-to-head comparative trial, the Pittsburgh Knee Rule outperformed the older Ottawa Knee Rule as an aid in figuring out who needs imaging after a knee injury. Impressive, in Dr. Burke’s view, but he’d like to see the results confirmed in a second study.
He reported having no financial conflicts.
ESTES PARK, COLO. – These days there’s a smartphone app for everything. But not all apps for clinical decision rules are truly helpful in making better patient care decisions in frontline primary care medicine, Dr. Robert E. Burke cautioned at a conference on internal medicine sponsored by the University of Colorado.
Five clinical decision rules have proven merit, each accessible via app as well as by an online website, he said. All five address medical issues that are common, important, and complex. What these rules have in common is excellent – not merely good – predictive value, thorough validation in multiple populations, a high likelihood that their use will influence patient management in individual cases, and an enthusiastic thumbs-up vote by physician-users.
These top-tier clinical decision rules address the following situations: determining which patients with community-acquired pneumonia can safely be managed at home, assessing whether a patient with chronic liver disease is likely to have advanced fibrosis or cirrhosis without resorting to liver biopsy, identifying who can be sent home safely after an episode of syncope, determining whether to prescribe oral anticoagulation for atrial fibrillation, and stratifying risk after a transient ischemic attack (TIA) or stroke.
These clinical decision rules are not a replacement for clinical judgment, but they do make risk/benefit calculations a lot easier, said Dr. Burke, a hospitalist at the Denver VA Medical Center and the university.
Triage of pneumonia patients. Pneumonia is the third most common reason for hospital admission as well as early readmission in the United States. So lots of different pneumonia severity scores have been developed. But few of them are any good, according to Dr. Burke.
Many of these subpar scores measure the likelihood that a patient with pneumonia will be admitted to the intensive care unit. That’s not what primary care physicians really want to know.
"Unfortunately, ICU admission rates vary widely in the U.S. and have nothing to do with how sick patients are or their comorbid conditions. It’s really not clear why the rates vary so much, but the predictive value of those scores is pretty poor, since ICU admission is a subjective thing," he explained.
The score he recommends is the Pneumonia Severity Index. It has been validated as an admission rule in several randomized trials totaling more than 4,000 patients. The score places patients into one of five risk categories. In the randomized trials, patients in the three lowest-risk categories had no negative outcomes when treated as outpatients. Plus, the use of the Pneumonia Severity Index reduced hospital admissions by up to 20%.
The score is calculated using 20 factors readily obtainable from the patient history, physical exam, and lab work. It’s a laborious calculation that’s perfect for an app. And the app exists: It’s called Simpli PSI.
Assessing fibrosis/cirrhosis risk in patients with chronic liver disease. Liver biopsy is painful, costly, decidedly not risk free, and often misleading due to the small tissue sample obtained. Transient elastography, a noninvasive ultrasound-based alternative, is promising but not ready for prime time. Yet knowing whether a patient has advanced fibrosis or cirrhosis is important in terms of prognosis, treatment, and surveillance strategy.
This is where the FIB-4 Index, a serum score for fibrosis, is of value. The FIB-4 Index is easily calculated from basic laboratory data. Findings from multiple studies have shown excellent discrimination regardless of whether patients had hepatitis C, hepatitis B, or nonalcoholic fatty liver disease.
"It has a c-statistic [equivalent to the area under the receiver operating characteristic curve] of 0.84. That’s about as good as it gets," according to the hospitalist.
The app is called Liver Calc. Like all the other apps Dr. Burke highlighted, it is available to iPhone users at the App Store.
"Or if you were to type in ‘FIB-4 Index’ on Google, you’ll find a website that will actually calculate the score online," he noted.
Whom to hospitalize for syncope. "Syncope is one of the most common things I see as a physician of in-patients," Dr. Burke said. "It accounts for maybe 1%-3% of emergency department visits and 6% of hospitalizations in the United States."
The San Francisco Syncope Rule is of proven value in helping to identify patients at low risk of serious outcomes in the 30 days following a syncopal episode. These are the patients who don’t need to undergo emergency department evaluation. The rule has been prospectively validated in tens of thousands of patients. It had a negative predictive value of 97% in a systematic review. The rule works less well in patients over age 65, where the likelihood of arrhythmia as the cause of syncope increases sharply. And the rule works best when applied only after a thorough history and physical exam aimed at finding a cause for the patient’s syncope.
"For example, the rule assumes you’ve already checked for hypoglycemia, a common cause of syncope," the hospitalist explained.
The San Francisco Syncope Rule is available in app form as Doctor Tools of the Trade.
Atrial fibrillation: To anticoagulate or not? This is a decision that entails balancing the reduction in stroke risk obtained with anticoagulation against the potential harm in the form of bleeding. The best tool for assessing the potential stroke-risk benefit is the CHA2DS2-VASc score, which represents a significant advance over the CHADS2 score, in Dr. Burke’s view.
"What the CHA2DS2-VASc score is really useful for, I think, is in putting people into low- or high-risk buckets. It takes people out of that intermediate-risk category in CHADS2," he explained.
It turns out that when physicians rely upon clinical intuition to estimate the risk of clinically important bleeding in patients on oral anticoagulation, they tend to overestimate the true risk. This observation has led to a proliferation of scales aimed at predicting who is likely to bleed when placed on warfarin. The best performer among them, regardless of whether the endpoint is any clinically relevant bleeding, major bleeding, or all-cause mortality, is the HAS-BLED score.
"I would encourage you to use both the CHA2DS2-VASc and HAS-BLED, and to use them at the same time. In some cases you’ll find the numbers are really discordant; there may be much more benefit than you thought, or much more risk," Dr. Burke said.
The focus-AF calculator is the app that will do the work.
Risk stratification after TIA or stroke. The ABCD2 score provides an estimate of stroke risk within 7 or 90 days after a TIA. This information helps establish the urgency of patient evaluation and risk factor management. A patient with an ABCD2 score of 4 or more should go straight to the emergency department, while a score of 3 or less indicates outpatient evaluation is appropriate. The Neuro Toolkit app will run the numbers.
Dr. Burke is keeping a watchful eye on a number of other clinical decision rules that, while promising, aren’t quite ready for prime time in his view. These include the FRAX score, the TIMI score, and the Marburg Heart Score. The Marburg score, for example, has been extensively validated as a tool to help primary care physicians decide whether chest pain is cardiac or noncardiac. But in the clinical trials, the score wasn’t compared to clinical intuition. That’s a problem.
"I think our clinical intuition here is relatively good. So I’d like to see data showing the rule adds something to clinical intuition before I recommend it," he said.
In a head-to-head comparative trial, the Pittsburgh Knee Rule outperformed the older Ottawa Knee Rule as an aid in figuring out who needs imaging after a knee injury. Impressive, in Dr. Burke’s view, but he’d like to see the results confirmed in a second study.
He reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
Current diagnostic criteria for early rheumatoid arthritis must be used judiciously
ESTES PARK, COLO. – The most important thing to know about the current American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis is that they are primarily a research tool and should only be applied diagnostically in selected circumstances.
The current criteria, known as the 2010 criteria, are markedly more effective at detecting RA early on – when it is far more treatment-responsive – than were the former 1987 criteria. That’s the upside. The downside of the 2010 criteria is unless they are employed judiciously, many patients will be inappropriately labeled as having RA and subjected to treatments they don’t actually need, Dr. Jason Kolfenbach explained at a conference on internal medicine sponsored by the University of Colorado.
Two essential preconditions must be met before the 2010 criteria can appropriately be brought to bear in diagnosing RA in the clinic. First, the patient has to have at least one swollen joint; joint pain without swelling isn’t sufficient.
Second, any alternative diagnoses that might better explain an individual’s synovitis must first be ruled out. It has been demonstrated that if the 2010 RA criteria are applied without first ruling out conditions including gout, lupus, and sarcoid, the false-positive rate, even in rheumatologists’ hands, is roughly 20%. When rheumatologists took the time to first remove the cases they thought likely to be something other than RA, however, the false-positive rate using the 2010 criteria fell to 9%, noted Dr. Kolfenbach, a rheumatologist at the university.
And then there’s the whole squirrelly matter of transient joint swelling.
Among patients with one or more swollen joints and no obvious etiology for their arthritis, the spontaneous remission rate approaches 50%. But among the subset of patients with at least one swollen joint who fulfill the 2010 criteria, the spontaneous remission rate appears to be much lower, on the order of 10% (Arthritis Rheum. 2012;64:389-93).
The 1987 criteria required radiographic evidence of erosions as well as the presence of subcutaneous rheumatoid nodules. Those are highly specific features of long-standing RA, but they’re not helpful in identifying patients with early disease. The impetus for developing the 2010 criteria was a persuasive body of evidence that in order to maximize outcomes physicians need to intervene earlier in the disease process than was possible using the 1987 criteria.
"The window for intervention is actually quite small. If you delay more than 3 or 4 months after symptom onset, your response to disease-modifying antirheumatic therapy is worse," he said.
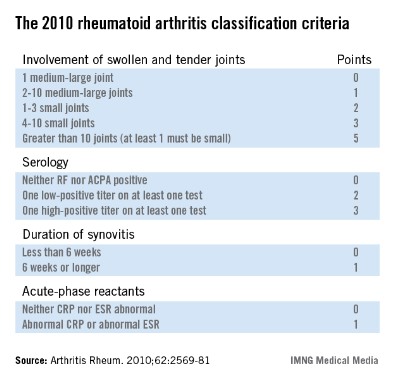
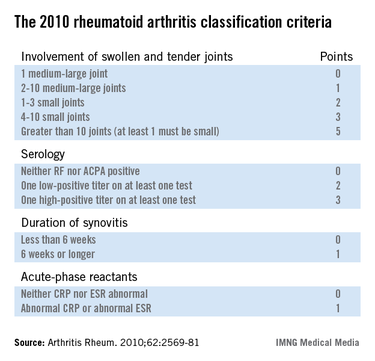
The 2010 criteria (Arthritis Rheum. 2010;62:2569-81) were developed through longitudinal follow-up of a cohort of patients who presented with an inflammatory arthritis that didn’t meet the 1987 criteria for RA. Investigators looked for features present initially that helped predict later definite RA. They found four: inflammatory joint involvement; antibody status; duration of synovitis; and the presence of inflammatory mediators, also known as acute phase reactants. Under the 2010 criteria, a score of 6 or more out of a possible 10 based upon these four elements is deemed definite RA. Of note, erosions and rheumatoid nodules aren’t part of the current diagnostic criteria.
Dr. Kolfenbach noted that in a large Dutch study, the sensitivity of the 2010 criteria for the diagnosis of RA as defined by the use of methotrexate or any other disease-modifying antirheumatic therapy within the first year of follow-up was 84%, an impressive absolute 23% improvement over the 61% sensitivity using the outmoded 1987 criteria (Arthritis Rheum. 2011;63:37-42).
"The problem, like with any test, is if we improve sensitivity it reduces specificity – and that increases the probability of inappropriately labeling someone as having rheumatoid arthritis," he observed.
Indeed, the Dutch study also showed that the diagnostic specificity of the 2010 criteria was only 60%, down from 74% when the 1987 criteria were applied to the same population.
So what’s the smart way to apply the 2010 classification criteria for RA?
"Clinical history and the physical exam are still king. If a patient comes through your office door with polyarticular swelling, mostly in the fingers and wrists, and it’s symmetrical, with a lack of systemic organ disease, that’s a person at much higher risk of having rheumatoid arthritis. It’s really reasonable then to apply the 2010 criteria and test for serologic antibodies and inflammatory markers. And if those are positive, then you’ve really cemented your feeling that this person very likely has rheumatoid arthritis," according to Dr. Kolfenbach.
Under the 2010 criteria, antibody testing remains "hugely important" in establishing the diagnosis of RA, he continued.
"If you don’t have a positive rheumatoid factor or anti-CCP [anti–cyclic citrullinated peptide] antibodies, you have to have more than 10 clinically involved joints to get to a score of 6. So we make the diagnosis of rheumatoid arthritis clinically and by listening to the patients and examining them, but antibodies are the icing on the cake. They’re a very, very important piece. And higher titers are specific for rheumatoid arthritis. A high-titer anti-CCP antibody level makes it very unlikely that it’s a false-positive test," the rheumatologist said.
He reported having no financial conflicts.
ESTES PARK, COLO. – The most important thing to know about the current American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis is that they are primarily a research tool and should only be applied diagnostically in selected circumstances.
The current criteria, known as the 2010 criteria, are markedly more effective at detecting RA early on – when it is far more treatment-responsive – than were the former 1987 criteria. That’s the upside. The downside of the 2010 criteria is unless they are employed judiciously, many patients will be inappropriately labeled as having RA and subjected to treatments they don’t actually need, Dr. Jason Kolfenbach explained at a conference on internal medicine sponsored by the University of Colorado.

Two essential preconditions must be met before the 2010 criteria can appropriately be brought to bear in diagnosing RA in the clinic. First, the patient has to have at least one swollen joint; joint pain without swelling isn’t sufficient.
Second, any alternative diagnoses that might better explain an individual’s synovitis must first be ruled out. It has been demonstrated that if the 2010 RA criteria are applied without first ruling out conditions including gout, lupus, and sarcoid, the false-positive rate, even in rheumatologists’ hands, is roughly 20%. When rheumatologists took the time to first remove the cases they thought likely to be something other than RA, however, the false-positive rate using the 2010 criteria fell to 9%, noted Dr. Kolfenbach, a rheumatologist at the university.
And then there’s the whole squirrelly matter of transient joint swelling.
Among patients with one or more swollen joints and no obvious etiology for their arthritis, the spontaneous remission rate approaches 50%. But among the subset of patients with at least one swollen joint who fulfill the 2010 criteria, the spontaneous remission rate appears to be much lower, on the order of 10% (Arthritis Rheum. 2012;64:389-93).
The 1987 criteria required radiographic evidence of erosions as well as the presence of subcutaneous rheumatoid nodules. Those are highly specific features of long-standing RA, but they’re not helpful in identifying patients with early disease. The impetus for developing the 2010 criteria was a persuasive body of evidence that in order to maximize outcomes physicians need to intervene earlier in the disease process than was possible using the 1987 criteria.
"The window for intervention is actually quite small. If you delay more than 3 or 4 months after symptom onset, your response to disease-modifying antirheumatic therapy is worse," he said.
The 2010 criteria (Arthritis Rheum. 2010;62:2569-81) were developed through longitudinal follow-up of a cohort of patients who presented with an inflammatory arthritis that didn’t meet the 1987 criteria for RA. Investigators looked for features present initially that helped predict later definite RA. They found four: inflammatory joint involvement; antibody status; duration of synovitis; and the presence of inflammatory mediators, also known as acute phase reactants. Under the 2010 criteria, a score of 6 or more out of a possible 10 based upon these four elements is deemed definite RA. Of note, erosions and rheumatoid nodules aren’t part of the current diagnostic criteria.
Dr. Kolfenbach noted that in a large Dutch study, the sensitivity of the 2010 criteria for the diagnosis of RA as defined by the use of methotrexate or any other disease-modifying antirheumatic therapy within the first year of follow-up was 84%, an impressive absolute 23% improvement over the 61% sensitivity using the outmoded 1987 criteria (Arthritis Rheum. 2011;63:37-42).
"The problem, like with any test, is if we improve sensitivity it reduces specificity – and that increases the probability of inappropriately labeling someone as having rheumatoid arthritis," he observed.
Indeed, the Dutch study also showed that the diagnostic specificity of the 2010 criteria was only 60%, down from 74% when the 1987 criteria were applied to the same population.
So what’s the smart way to apply the 2010 classification criteria for RA?
"Clinical history and the physical exam are still king. If a patient comes through your office door with polyarticular swelling, mostly in the fingers and wrists, and it’s symmetrical, with a lack of systemic organ disease, that’s a person at much higher risk of having rheumatoid arthritis. It’s really reasonable then to apply the 2010 criteria and test for serologic antibodies and inflammatory markers. And if those are positive, then you’ve really cemented your feeling that this person very likely has rheumatoid arthritis," according to Dr. Kolfenbach.
Under the 2010 criteria, antibody testing remains "hugely important" in establishing the diagnosis of RA, he continued.
"If you don’t have a positive rheumatoid factor or anti-CCP [anti–cyclic citrullinated peptide] antibodies, you have to have more than 10 clinically involved joints to get to a score of 6. So we make the diagnosis of rheumatoid arthritis clinically and by listening to the patients and examining them, but antibodies are the icing on the cake. They’re a very, very important piece. And higher titers are specific for rheumatoid arthritis. A high-titer anti-CCP antibody level makes it very unlikely that it’s a false-positive test," the rheumatologist said.
He reported having no financial conflicts.
ESTES PARK, COLO. – The most important thing to know about the current American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis is that they are primarily a research tool and should only be applied diagnostically in selected circumstances.
The current criteria, known as the 2010 criteria, are markedly more effective at detecting RA early on – when it is far more treatment-responsive – than were the former 1987 criteria. That’s the upside. The downside of the 2010 criteria is unless they are employed judiciously, many patients will be inappropriately labeled as having RA and subjected to treatments they don’t actually need, Dr. Jason Kolfenbach explained at a conference on internal medicine sponsored by the University of Colorado.

Two essential preconditions must be met before the 2010 criteria can appropriately be brought to bear in diagnosing RA in the clinic. First, the patient has to have at least one swollen joint; joint pain without swelling isn’t sufficient.
Second, any alternative diagnoses that might better explain an individual’s synovitis must first be ruled out. It has been demonstrated that if the 2010 RA criteria are applied without first ruling out conditions including gout, lupus, and sarcoid, the false-positive rate, even in rheumatologists’ hands, is roughly 20%. When rheumatologists took the time to first remove the cases they thought likely to be something other than RA, however, the false-positive rate using the 2010 criteria fell to 9%, noted Dr. Kolfenbach, a rheumatologist at the university.
And then there’s the whole squirrelly matter of transient joint swelling.
Among patients with one or more swollen joints and no obvious etiology for their arthritis, the spontaneous remission rate approaches 50%. But among the subset of patients with at least one swollen joint who fulfill the 2010 criteria, the spontaneous remission rate appears to be much lower, on the order of 10% (Arthritis Rheum. 2012;64:389-93).
The 1987 criteria required radiographic evidence of erosions as well as the presence of subcutaneous rheumatoid nodules. Those are highly specific features of long-standing RA, but they’re not helpful in identifying patients with early disease. The impetus for developing the 2010 criteria was a persuasive body of evidence that in order to maximize outcomes physicians need to intervene earlier in the disease process than was possible using the 1987 criteria.
"The window for intervention is actually quite small. If you delay more than 3 or 4 months after symptom onset, your response to disease-modifying antirheumatic therapy is worse," he said.
The 2010 criteria (Arthritis Rheum. 2010;62:2569-81) were developed through longitudinal follow-up of a cohort of patients who presented with an inflammatory arthritis that didn’t meet the 1987 criteria for RA. Investigators looked for features present initially that helped predict later definite RA. They found four: inflammatory joint involvement; antibody status; duration of synovitis; and the presence of inflammatory mediators, also known as acute phase reactants. Under the 2010 criteria, a score of 6 or more out of a possible 10 based upon these four elements is deemed definite RA. Of note, erosions and rheumatoid nodules aren’t part of the current diagnostic criteria.
Dr. Kolfenbach noted that in a large Dutch study, the sensitivity of the 2010 criteria for the diagnosis of RA as defined by the use of methotrexate or any other disease-modifying antirheumatic therapy within the first year of follow-up was 84%, an impressive absolute 23% improvement over the 61% sensitivity using the outmoded 1987 criteria (Arthritis Rheum. 2011;63:37-42).
"The problem, like with any test, is if we improve sensitivity it reduces specificity – and that increases the probability of inappropriately labeling someone as having rheumatoid arthritis," he observed.
Indeed, the Dutch study also showed that the diagnostic specificity of the 2010 criteria was only 60%, down from 74% when the 1987 criteria were applied to the same population.
So what’s the smart way to apply the 2010 classification criteria for RA?
"Clinical history and the physical exam are still king. If a patient comes through your office door with polyarticular swelling, mostly in the fingers and wrists, and it’s symmetrical, with a lack of systemic organ disease, that’s a person at much higher risk of having rheumatoid arthritis. It’s really reasonable then to apply the 2010 criteria and test for serologic antibodies and inflammatory markers. And if those are positive, then you’ve really cemented your feeling that this person very likely has rheumatoid arthritis," according to Dr. Kolfenbach.
Under the 2010 criteria, antibody testing remains "hugely important" in establishing the diagnosis of RA, he continued.
"If you don’t have a positive rheumatoid factor or anti-CCP [anti–cyclic citrullinated peptide] antibodies, you have to have more than 10 clinically involved joints to get to a score of 6. So we make the diagnosis of rheumatoid arthritis clinically and by listening to the patients and examining them, but antibodies are the icing on the cake. They’re a very, very important piece. And higher titers are specific for rheumatoid arthritis. A high-titer anti-CCP antibody level makes it very unlikely that it’s a false-positive test," the rheumatologist said.
He reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
Novel GLP-1 agonist dulaglutide wins AWARD for type 2 diabetes
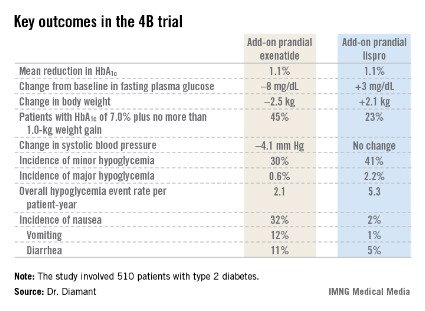
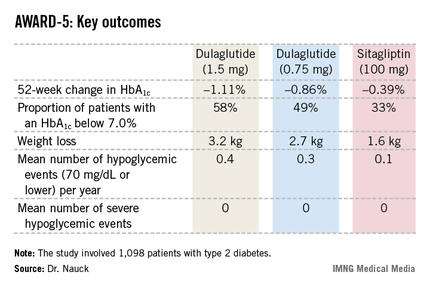
CHICAGO – Dulaglutide, an investigational once-weekly long-acting glucagonlike peptide–1 receptor agonist, provided superior glycemic control compared with sitagliptin in patients with type 2 diabetes in the phase III AWARD-5 trial.
In this 52-week, 1,098-patient randomized trial, both the 0.75- and 1.5-mg doses of once-weekly dulaglutide proved superior to oral sitagliptin (Januvia) at the standard dose of 100 mg/day in key efficacy endpoints. These outcomes included magnitude of hemoglobin A1c reduction, achievement of guideline-recommended HbA1c targets, and weight loss, Dr. Michael A. Nauck reported at the annual scientific sessions of the American Diabetes Association.
Dulaglutide is a recombinant glucagonlike peptide–1 (GLP-1) fusion protein linking a human GLP-1 peptide analogue to a large protein that gives the medication a lengthy half-life of 5 days. Metabolism of dulaglutide is not renally dependent, making the drug a logical candidate for future study in type 2 diabetic patients with renal impairment, observed Dr. Nauck, head of the Bad Lauterberg (Germany) Diabetes Center.
AWARD-5: Dulaglutide vs. sitagliptin
The fifth Assessment of Weekly Administration of LY2189265 in Diabetes (AWARD-5) was one of three phase III clinical trials in the AWARD program presented at the meeting. In AWARD-1, the once-weekly injectable GLP-1 agonist demonstrated superior glycemic control compared with twice-daily exenatide (Byetta) in type 2 diabetic patients not adequately controlled with full-dose metformin and pioglitazone. AWARD-3 showed dulaglutide to be superior to metformin in patients with early type 2 diabetes.
The 1,098 participants in AWARD-5 had a mean baseline HbA1c of 8.1% despite being on metformin and, in one-quarter of cases, an additional oral antidiabetic agent. They stayed on their previous oral medication during the trial. Their mean baseline body mass index was 31 kg/m2, and they averaged a 7-year history of diabetes.
The primary objective in AWARD-5 was to establish that dulaglutide was noninferior to sitagliptin in its ability to reduce HbA1c from baseline through 52 weeks. But in fact, Dr. Nauck reported, both doses of dulaglutide exceeded that standard and actually achieved statistically significant and clinically meaningful superiority to the dipeptidyl peptidase-4 inhibitor (see chart).
With regard to safety issues, the endocrinologist noted that hypoglycemia rates didn’t vary significantly between the study arms. No cases of pancreatitis or pancreatic cancer occurred in dulaglutide-treated patients. There were three cases of pancreatitis in the sitagliptin group.
Gastrointestinal side effects were common among recipients of the once-weekly GLP-1 agonist. The incidence of nausea was 17% in patients who received dulaglutide at 1.5 mg, 14% with 0.75 mg, and 5% with sitagliptin. Diarrhea occurred in 15% and 10% of patients on the high- and low-dose dulaglutide regimens, respectively, compared with 3% on sitagliptin. The incidence of vomiting was 13% with dulaglutide at 1.5 mg, 8% at 0.75 mg, and 2% with sitagliptin. However, the frequency of dulaglutide-related GI side effects dropped off markedly after the first 4-8 weeks, the side effects were mostly mild to moderate in nature, and very few patients dropped out of the study due to GI symptoms, according to Dr. Nauck.
AWARD-1: Dulaglutide vs. exenatide
In a separate presentation, Dr. Carol Wysham reported on 978 patients with type 2 diabetes in the 52-week, phase III AWARD-1 trial who were randomized to dulaglutide at 1.5 or 0.75 mg, the injectable GLP-1 agonist exenatide at 10 mcg twice daily, or placebo. Their baseline HbA1c was 8.07% despite being on metformin and pioglitazone.
The primary endpoint was change in HbA1c at 26 weeks. The reduction was significantly bigger in the two dulaglutide study arms than with exenatide: –1.51% compared with baseline in the dulaglutide 1.5-mg arm, –1.3% with dulaglutide 0.75 mg, –0.99% with exenatide, and –0.46% with placebo. The proportion of patients with an HbA1c of 6.5% or less at 26 weeks was 64% with dulaglutide 1.5 mg, 54% with the long-acting agent at 0.75 mg, 39% with sitagliptin, and 26% in placebo-treated controls.
These results held up at the 52-week mark as well. At that time, 71% of the dulaglutide 1.5-mg group had an HbA1c below 7.0%, as did 59% of patients on dulaglutide 0.75 mg and 49% on exenatide.
As in the other AWARD trials, most of the reduction in body weight and fasting plasma glucose in dulaglutide-treated patients occurred in the first 2 weeks of treatment. The early benefits were then maintained through 52 weeks, observed Dr. Wysham, an endocrinologist in Spokane, Wash.
AWARD-3: Dulaglutide vs. metformin
Dr. Guillermo E. Umpierrez presented the results of AWARD-3, which involved 807 patients with early type 2 diabetes as evidenced by their mean 2.6-year disease history. Their baseline HbA1c was 7.6% with diet and exercise alone or in combination with a single, low-dose, oral antidiabetic medication. Participants were randomized to dulaglutide at 1.5 or 0.75 mg or to metformin at 1,000 mg twice daily.
The primary endpoint was change in HbA1c over 26 weeks. Both doses of dulaglutide proved superior to metformin, with reductions of 0.78% with dulaglutide at 1.5 mg and 0.71% at 0.75 mg, compared with 0.56% with metformin, reported Dr. Umpierrez, professor of medicine at Emory University and chief of diabetes and endocrinology at Grady Memorial Hospital, Atlanta.
Session chair Dr. Julio Rosenstock didn’t buy Dr. Umpierrez’ conclusion that AWARD-3 proved dulaglutide to be superior to metformin. He argued that even if the differences in HbA1c reduction between the dulaglutide- and metformin-treated patients were statistically significant, they were too small to be clinically meaningful. "I don’t think this is clinical superiority. You’ve presented evidence that dulaglutide is comparable in efficacy to metformin in early type 2 diabetes. I see it as an option for patients who can’t tolerate metformin," commented Dr. Rosenstock, director of the Dallas Diabetes and Endocrine Center.
The AWARD trials were supported by Eli Lilly, which anticipates applying to the Food and Drug Administration for marketing approval of dulaglutide before the end of the year. All three presenters reported receiving research grants from Eli Lilly and other pharmaceutical companies. Drs. Wysham and Nauck are also on the Eli Lilly advisory board.
CHICAGO – Dulaglutide, an investigational once-weekly long-acting glucagonlike peptide–1 receptor agonist, provided superior glycemic control compared with sitagliptin in patients with type 2 diabetes in the phase III AWARD-5 trial.
In this 52-week, 1,098-patient randomized trial, both the 0.75- and 1.5-mg doses of once-weekly dulaglutide proved superior to oral sitagliptin (Januvia) at the standard dose of 100 mg/day in key efficacy endpoints. These outcomes included magnitude of hemoglobin A1c reduction, achievement of guideline-recommended HbA1c targets, and weight loss, Dr. Michael A. Nauck reported at the annual scientific sessions of the American Diabetes Association.

Dulaglutide is a recombinant glucagonlike peptide–1 (GLP-1) fusion protein linking a human GLP-1 peptide analogue to a large protein that gives the medication a lengthy half-life of 5 days. Metabolism of dulaglutide is not renally dependent, making the drug a logical candidate for future study in type 2 diabetic patients with renal impairment, observed Dr. Nauck, head of the Bad Lauterberg (Germany) Diabetes Center.
AWARD-5: Dulaglutide vs. sitagliptin
The fifth Assessment of Weekly Administration of LY2189265 in Diabetes (AWARD-5) was one of three phase III clinical trials in the AWARD program presented at the meeting. In AWARD-1, the once-weekly injectable GLP-1 agonist demonstrated superior glycemic control compared with twice-daily exenatide (Byetta) in type 2 diabetic patients not adequately controlled with full-dose metformin and pioglitazone. AWARD-3 showed dulaglutide to be superior to metformin in patients with early type 2 diabetes.
The 1,098 participants in AWARD-5 had a mean baseline HbA1c of 8.1% despite being on metformin and, in one-quarter of cases, an additional oral antidiabetic agent. They stayed on their previous oral medication during the trial. Their mean baseline body mass index was 31 kg/m2, and they averaged a 7-year history of diabetes.
The primary objective in AWARD-5 was to establish that dulaglutide was noninferior to sitagliptin in its ability to reduce HbA1c from baseline through 52 weeks. But in fact, Dr. Nauck reported, both doses of dulaglutide exceeded that standard and actually achieved statistically significant and clinically meaningful superiority to the dipeptidyl peptidase-4 inhibitor (see chart).
With regard to safety issues, the endocrinologist noted that hypoglycemia rates didn’t vary significantly between the study arms. No cases of pancreatitis or pancreatic cancer occurred in dulaglutide-treated patients. There were three cases of pancreatitis in the sitagliptin group.
Gastrointestinal side effects were common among recipients of the once-weekly GLP-1 agonist. The incidence of nausea was 17% in patients who received dulaglutide at 1.5 mg, 14% with 0.75 mg, and 5% with sitagliptin. Diarrhea occurred in 15% and 10% of patients on the high- and low-dose dulaglutide regimens, respectively, compared with 3% on sitagliptin. The incidence of vomiting was 13% with dulaglutide at 1.5 mg, 8% at 0.75 mg, and 2% with sitagliptin. However, the frequency of dulaglutide-related GI side effects dropped off markedly after the first 4-8 weeks, the side effects were mostly mild to moderate in nature, and very few patients dropped out of the study due to GI symptoms, according to Dr. Nauck.
AWARD-1: Dulaglutide vs. exenatide
In a separate presentation, Dr. Carol Wysham reported on 978 patients with type 2 diabetes in the 52-week, phase III AWARD-1 trial who were randomized to dulaglutide at 1.5 or 0.75 mg, the injectable GLP-1 agonist exenatide at 10 mcg twice daily, or placebo. Their baseline HbA1c was 8.07% despite being on metformin and pioglitazone.
The primary endpoint was change in HbA1c at 26 weeks. The reduction was significantly bigger in the two dulaglutide study arms than with exenatide: –1.51% compared with baseline in the dulaglutide 1.5-mg arm, –1.3% with dulaglutide 0.75 mg, –0.99% with exenatide, and –0.46% with placebo. The proportion of patients with an HbA1c of 6.5% or less at 26 weeks was 64% with dulaglutide 1.5 mg, 54% with the long-acting agent at 0.75 mg, 39% with sitagliptin, and 26% in placebo-treated controls.
These results held up at the 52-week mark as well. At that time, 71% of the dulaglutide 1.5-mg group had an HbA1c below 7.0%, as did 59% of patients on dulaglutide 0.75 mg and 49% on exenatide.
As in the other AWARD trials, most of the reduction in body weight and fasting plasma glucose in dulaglutide-treated patients occurred in the first 2 weeks of treatment. The early benefits were then maintained through 52 weeks, observed Dr. Wysham, an endocrinologist in Spokane, Wash.
AWARD-3: Dulaglutide vs. metformin
Dr. Guillermo E. Umpierrez presented the results of AWARD-3, which involved 807 patients with early type 2 diabetes as evidenced by their mean 2.6-year disease history. Their baseline HbA1c was 7.6% with diet and exercise alone or in combination with a single, low-dose, oral antidiabetic medication. Participants were randomized to dulaglutide at 1.5 or 0.75 mg or to metformin at 1,000 mg twice daily.
The primary endpoint was change in HbA1c over 26 weeks. Both doses of dulaglutide proved superior to metformin, with reductions of 0.78% with dulaglutide at 1.5 mg and 0.71% at 0.75 mg, compared with 0.56% with metformin, reported Dr. Umpierrez, professor of medicine at Emory University and chief of diabetes and endocrinology at Grady Memorial Hospital, Atlanta.
Session chair Dr. Julio Rosenstock didn’t buy Dr. Umpierrez’ conclusion that AWARD-3 proved dulaglutide to be superior to metformin. He argued that even if the differences in HbA1c reduction between the dulaglutide- and metformin-treated patients were statistically significant, they were too small to be clinically meaningful. "I don’t think this is clinical superiority. You’ve presented evidence that dulaglutide is comparable in efficacy to metformin in early type 2 diabetes. I see it as an option for patients who can’t tolerate metformin," commented Dr. Rosenstock, director of the Dallas Diabetes and Endocrine Center.
The AWARD trials were supported by Eli Lilly, which anticipates applying to the Food and Drug Administration for marketing approval of dulaglutide before the end of the year. All three presenters reported receiving research grants from Eli Lilly and other pharmaceutical companies. Drs. Wysham and Nauck are also on the Eli Lilly advisory board.
CHICAGO – Dulaglutide, an investigational once-weekly long-acting glucagonlike peptide–1 receptor agonist, provided superior glycemic control compared with sitagliptin in patients with type 2 diabetes in the phase III AWARD-5 trial.
In this 52-week, 1,098-patient randomized trial, both the 0.75- and 1.5-mg doses of once-weekly dulaglutide proved superior to oral sitagliptin (Januvia) at the standard dose of 100 mg/day in key efficacy endpoints. These outcomes included magnitude of hemoglobin A1c reduction, achievement of guideline-recommended HbA1c targets, and weight loss, Dr. Michael A. Nauck reported at the annual scientific sessions of the American Diabetes Association.

Dulaglutide is a recombinant glucagonlike peptide–1 (GLP-1) fusion protein linking a human GLP-1 peptide analogue to a large protein that gives the medication a lengthy half-life of 5 days. Metabolism of dulaglutide is not renally dependent, making the drug a logical candidate for future study in type 2 diabetic patients with renal impairment, observed Dr. Nauck, head of the Bad Lauterberg (Germany) Diabetes Center.
AWARD-5: Dulaglutide vs. sitagliptin
The fifth Assessment of Weekly Administration of LY2189265 in Diabetes (AWARD-5) was one of three phase III clinical trials in the AWARD program presented at the meeting. In AWARD-1, the once-weekly injectable GLP-1 agonist demonstrated superior glycemic control compared with twice-daily exenatide (Byetta) in type 2 diabetic patients not adequately controlled with full-dose metformin and pioglitazone. AWARD-3 showed dulaglutide to be superior to metformin in patients with early type 2 diabetes.
The 1,098 participants in AWARD-5 had a mean baseline HbA1c of 8.1% despite being on metformin and, in one-quarter of cases, an additional oral antidiabetic agent. They stayed on their previous oral medication during the trial. Their mean baseline body mass index was 31 kg/m2, and they averaged a 7-year history of diabetes.
The primary objective in AWARD-5 was to establish that dulaglutide was noninferior to sitagliptin in its ability to reduce HbA1c from baseline through 52 weeks. But in fact, Dr. Nauck reported, both doses of dulaglutide exceeded that standard and actually achieved statistically significant and clinically meaningful superiority to the dipeptidyl peptidase-4 inhibitor (see chart).
With regard to safety issues, the endocrinologist noted that hypoglycemia rates didn’t vary significantly between the study arms. No cases of pancreatitis or pancreatic cancer occurred in dulaglutide-treated patients. There were three cases of pancreatitis in the sitagliptin group.
Gastrointestinal side effects were common among recipients of the once-weekly GLP-1 agonist. The incidence of nausea was 17% in patients who received dulaglutide at 1.5 mg, 14% with 0.75 mg, and 5% with sitagliptin. Diarrhea occurred in 15% and 10% of patients on the high- and low-dose dulaglutide regimens, respectively, compared with 3% on sitagliptin. The incidence of vomiting was 13% with dulaglutide at 1.5 mg, 8% at 0.75 mg, and 2% with sitagliptin. However, the frequency of dulaglutide-related GI side effects dropped off markedly after the first 4-8 weeks, the side effects were mostly mild to moderate in nature, and very few patients dropped out of the study due to GI symptoms, according to Dr. Nauck.
AWARD-1: Dulaglutide vs. exenatide
In a separate presentation, Dr. Carol Wysham reported on 978 patients with type 2 diabetes in the 52-week, phase III AWARD-1 trial who were randomized to dulaglutide at 1.5 or 0.75 mg, the injectable GLP-1 agonist exenatide at 10 mcg twice daily, or placebo. Their baseline HbA1c was 8.07% despite being on metformin and pioglitazone.
The primary endpoint was change in HbA1c at 26 weeks. The reduction was significantly bigger in the two dulaglutide study arms than with exenatide: –1.51% compared with baseline in the dulaglutide 1.5-mg arm, –1.3% with dulaglutide 0.75 mg, –0.99% with exenatide, and –0.46% with placebo. The proportion of patients with an HbA1c of 6.5% or less at 26 weeks was 64% with dulaglutide 1.5 mg, 54% with the long-acting agent at 0.75 mg, 39% with sitagliptin, and 26% in placebo-treated controls.
These results held up at the 52-week mark as well. At that time, 71% of the dulaglutide 1.5-mg group had an HbA1c below 7.0%, as did 59% of patients on dulaglutide 0.75 mg and 49% on exenatide.
As in the other AWARD trials, most of the reduction in body weight and fasting plasma glucose in dulaglutide-treated patients occurred in the first 2 weeks of treatment. The early benefits were then maintained through 52 weeks, observed Dr. Wysham, an endocrinologist in Spokane, Wash.
AWARD-3: Dulaglutide vs. metformin
Dr. Guillermo E. Umpierrez presented the results of AWARD-3, which involved 807 patients with early type 2 diabetes as evidenced by their mean 2.6-year disease history. Their baseline HbA1c was 7.6% with diet and exercise alone or in combination with a single, low-dose, oral antidiabetic medication. Participants were randomized to dulaglutide at 1.5 or 0.75 mg or to metformin at 1,000 mg twice daily.
The primary endpoint was change in HbA1c over 26 weeks. Both doses of dulaglutide proved superior to metformin, with reductions of 0.78% with dulaglutide at 1.5 mg and 0.71% at 0.75 mg, compared with 0.56% with metformin, reported Dr. Umpierrez, professor of medicine at Emory University and chief of diabetes and endocrinology at Grady Memorial Hospital, Atlanta.
Session chair Dr. Julio Rosenstock didn’t buy Dr. Umpierrez’ conclusion that AWARD-3 proved dulaglutide to be superior to metformin. He argued that even if the differences in HbA1c reduction between the dulaglutide- and metformin-treated patients were statistically significant, they were too small to be clinically meaningful. "I don’t think this is clinical superiority. You’ve presented evidence that dulaglutide is comparable in efficacy to metformin in early type 2 diabetes. I see it as an option for patients who can’t tolerate metformin," commented Dr. Rosenstock, director of the Dallas Diabetes and Endocrine Center.
The AWARD trials were supported by Eli Lilly, which anticipates applying to the Food and Drug Administration for marketing approval of dulaglutide before the end of the year. All three presenters reported receiving research grants from Eli Lilly and other pharmaceutical companies. Drs. Wysham and Nauck are also on the Eli Lilly advisory board.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Anserine bursitis: Often-overlooked cause of knee pain
ESTES PARK, COLO. – Anserine bursitis is a common overuse injury frequently misinterpreted as osteoarthritis, according to Dr. Robert W. Janson.
The pain is focused at the medial aspect of the knee about 2 inches below the tibial plateau, or joint line. The bursa is located at the anatomic site known as the pes anserinus, or goose foot, where the conjoined tendons of the semitendinosus, gracilis, and sartorius muscles insert at the tibia, Dr. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
"How do you find this bursa? It’s very easy. Take two fingers, grab the hamstring, and just run the two fingers down until they meet and you’re right there," said Dr. Janson, a rheumatologist at the university.
An affected patient will report the pain is worse with stair climbing or lying on one side with the knees opposed.
"Obese women with valgus knees tend to get this injury a lot," according to Dr. Janson.
Other risk factors for anserine bursitis are gait abnormalities or running and jogging.
Management is via the general measures utilized in treating overuse syndromes: rest, ice or heat, full-dose NSAIDs, and hamstring stretching.
If the pain persists for 6-8 weeks, a local injection of 10-20 mg of methylprednisolone with 1% lidocaine is enormously helpful. Find the tender area, mark it, direct the needle down to the bone, then back off about 1 mm and simply infiltrate that area, the rheumatologist advised.
He reported having no relevant financial conflicts.
ESTES PARK, COLO. – Anserine bursitis is a common overuse injury frequently misinterpreted as osteoarthritis, according to Dr. Robert W. Janson.
The pain is focused at the medial aspect of the knee about 2 inches below the tibial plateau, or joint line. The bursa is located at the anatomic site known as the pes anserinus, or goose foot, where the conjoined tendons of the semitendinosus, gracilis, and sartorius muscles insert at the tibia, Dr. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
"How do you find this bursa? It’s very easy. Take two fingers, grab the hamstring, and just run the two fingers down until they meet and you’re right there," said Dr. Janson, a rheumatologist at the university.
An affected patient will report the pain is worse with stair climbing or lying on one side with the knees opposed.
"Obese women with valgus knees tend to get this injury a lot," according to Dr. Janson.
Other risk factors for anserine bursitis are gait abnormalities or running and jogging.
Management is via the general measures utilized in treating overuse syndromes: rest, ice or heat, full-dose NSAIDs, and hamstring stretching.
If the pain persists for 6-8 weeks, a local injection of 10-20 mg of methylprednisolone with 1% lidocaine is enormously helpful. Find the tender area, mark it, direct the needle down to the bone, then back off about 1 mm and simply infiltrate that area, the rheumatologist advised.
He reported having no relevant financial conflicts.
ESTES PARK, COLO. – Anserine bursitis is a common overuse injury frequently misinterpreted as osteoarthritis, according to Dr. Robert W. Janson.
The pain is focused at the medial aspect of the knee about 2 inches below the tibial plateau, or joint line. The bursa is located at the anatomic site known as the pes anserinus, or goose foot, where the conjoined tendons of the semitendinosus, gracilis, and sartorius muscles insert at the tibia, Dr. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
"How do you find this bursa? It’s very easy. Take two fingers, grab the hamstring, and just run the two fingers down until they meet and you’re right there," said Dr. Janson, a rheumatologist at the university.
An affected patient will report the pain is worse with stair climbing or lying on one side with the knees opposed.
"Obese women with valgus knees tend to get this injury a lot," according to Dr. Janson.
Other risk factors for anserine bursitis are gait abnormalities or running and jogging.
Management is via the general measures utilized in treating overuse syndromes: rest, ice or heat, full-dose NSAIDs, and hamstring stretching.
If the pain persists for 6-8 weeks, a local injection of 10-20 mg of methylprednisolone with 1% lidocaine is enormously helpful. Find the tender area, mark it, direct the needle down to the bone, then back off about 1 mm and simply infiltrate that area, the rheumatologist advised.
He reported having no relevant financial conflicts.
EXPERT ANALYSIS from the annual internal medicine program
4B trial showcases novel strategy in uncontrolled type 2 diabetes
CHICAGO – Adding a premeal short-acting glucagonlike peptide–1 agonist is an attractive alternative to add-on prandial insulin when intensification of basal insulin no longer controls type 2 diabetes, according to the findings of the 4B trial.
"Addition of prandial insulin has been the gold standard. But we know that that is often associated with a high risk of hypoglycemia, weight gain, and low patient acceptance, so we need alternative treatment approaches," principal investigator Dr. Michaela Diamant said, in explaining the study rationale at the annual scientific sessions of the American Diabetes Association.
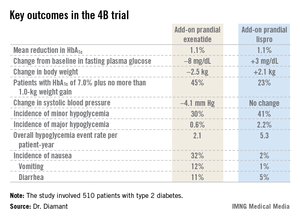
The 4B (basal insulin glargine and exenatide b.i.d. treatment or basal insulin glargine and mealtime bolus insulin lispro treatment) trial was the first study to evaluate a prandial glucagonlike peptide–1 agonist as such an approach. The open-label, prospective, randomized phase IIIb study involved 510 type 2 diabetic patients in 17 countries. All had a hemoglobin A1c level above 7.0% despite metformin plus 12 weeks of basal insulin glargine (Lantus) intensification using the standardized titration algorithm known as INITIATE. Indeed, their mean HbA1c level at randomization was 8.3%.
Participants were randomized to 30 weeks of either add-on, twice-daily premeal exenatide (Byetta) or mealtime bolus insulin lispro (Humalog) three times daily. The mean daily insulin lispro dose was 42.1 U. Eighty percent of patients in the exenatide group ended up on 10 mcg twice daily, with the rest on half that dose, explained Dr. Diamant of the Free University of Amsterdam.
The key study findings: The two treatment strategies resulted in similar HbA1c reductions, but the exenatide group had a lower final mean fasting plasma glucose level, a significant advantage in terms of change in body weight, less daytime hypoglycemia, lower systolic blood pressure, better patient treatment-satisfaction scores, and much more nausea.
The 4B trial was supported by Eli Lilly. Dr. Diamant reported receiving research grants from as well as serving as a consultant to and an advisory board member for Eli Lilly and other pharmaceutical companies.
CHICAGO – Adding a premeal short-acting glucagonlike peptide–1 agonist is an attractive alternative to add-on prandial insulin when intensification of basal insulin no longer controls type 2 diabetes, according to the findings of the 4B trial.
"Addition of prandial insulin has been the gold standard. But we know that that is often associated with a high risk of hypoglycemia, weight gain, and low patient acceptance, so we need alternative treatment approaches," principal investigator Dr. Michaela Diamant said, in explaining the study rationale at the annual scientific sessions of the American Diabetes Association.

The 4B (basal insulin glargine and exenatide b.i.d. treatment or basal insulin glargine and mealtime bolus insulin lispro treatment) trial was the first study to evaluate a prandial glucagonlike peptide–1 agonist as such an approach. The open-label, prospective, randomized phase IIIb study involved 510 type 2 diabetic patients in 17 countries. All had a hemoglobin A1c level above 7.0% despite metformin plus 12 weeks of basal insulin glargine (Lantus) intensification using the standardized titration algorithm known as INITIATE. Indeed, their mean HbA1c level at randomization was 8.3%.
Participants were randomized to 30 weeks of either add-on, twice-daily premeal exenatide (Byetta) or mealtime bolus insulin lispro (Humalog) three times daily. The mean daily insulin lispro dose was 42.1 U. Eighty percent of patients in the exenatide group ended up on 10 mcg twice daily, with the rest on half that dose, explained Dr. Diamant of the Free University of Amsterdam.
The key study findings: The two treatment strategies resulted in similar HbA1c reductions, but the exenatide group had a lower final mean fasting plasma glucose level, a significant advantage in terms of change in body weight, less daytime hypoglycemia, lower systolic blood pressure, better patient treatment-satisfaction scores, and much more nausea.
The 4B trial was supported by Eli Lilly. Dr. Diamant reported receiving research grants from as well as serving as a consultant to and an advisory board member for Eli Lilly and other pharmaceutical companies.
CHICAGO – Adding a premeal short-acting glucagonlike peptide–1 agonist is an attractive alternative to add-on prandial insulin when intensification of basal insulin no longer controls type 2 diabetes, according to the findings of the 4B trial.
"Addition of prandial insulin has been the gold standard. But we know that that is often associated with a high risk of hypoglycemia, weight gain, and low patient acceptance, so we need alternative treatment approaches," principal investigator Dr. Michaela Diamant said, in explaining the study rationale at the annual scientific sessions of the American Diabetes Association.

The 4B (basal insulin glargine and exenatide b.i.d. treatment or basal insulin glargine and mealtime bolus insulin lispro treatment) trial was the first study to evaluate a prandial glucagonlike peptide–1 agonist as such an approach. The open-label, prospective, randomized phase IIIb study involved 510 type 2 diabetic patients in 17 countries. All had a hemoglobin A1c level above 7.0% despite metformin plus 12 weeks of basal insulin glargine (Lantus) intensification using the standardized titration algorithm known as INITIATE. Indeed, their mean HbA1c level at randomization was 8.3%.
Participants were randomized to 30 weeks of either add-on, twice-daily premeal exenatide (Byetta) or mealtime bolus insulin lispro (Humalog) three times daily. The mean daily insulin lispro dose was 42.1 U. Eighty percent of patients in the exenatide group ended up on 10 mcg twice daily, with the rest on half that dose, explained Dr. Diamant of the Free University of Amsterdam.
The key study findings: The two treatment strategies resulted in similar HbA1c reductions, but the exenatide group had a lower final mean fasting plasma glucose level, a significant advantage in terms of change in body weight, less daytime hypoglycemia, lower systolic blood pressure, better patient treatment-satisfaction scores, and much more nausea.
The 4B trial was supported by Eli Lilly. Dr. Diamant reported receiving research grants from as well as serving as a consultant to and an advisory board member for Eli Lilly and other pharmaceutical companies.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major Finding: Patients with inadequately controlled type 2 diabetes, despite their being on metformin and optimized basal insulin, had better outcomes in multiple domains if they received add-on premeal exenatide twice daily than with add-on prandial insulin lispro three times daily.
Data Source: A phase IIIb, open-label, prospective randomized trial involving 510 type 2 diabetic patients with inadequate glycemic control.
Disclosures: The trial was supported by Eli Lilly. Dr. Diamant reported receiving research grants from as well as serving as a consultant to and an advisory board member for Eli Lilly and other pharmaceutical companies.
Identifying pediatric emergencies involving fever and rash
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
EXPERT ANALYSIS FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Identifying pediatric emergencies involving fever and rash
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
VAIL, COLO. – The differential diagnosis of pediatric infectious disease emergencies marked by fever and rash can quickly be narrowed by paying close attention to the type of rash.
Infectious diseases featuring fever and rash that require emergency diagnosis and treatment include fever with rashes that are purpuric, petechial, or erythrodermic, Dr. Marsha S. Anderson said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
Fever with purpura
Heading the list of diagnostic possibilities here is meningococcemia, which she called "every pediatrician’s nightmare" because of a case-fatality rate of 10%-20% even with appropriate treatment.
Complicating the situation is the fact that in a minority of cases the rash is petechial rather than purpuric.
"This is a disease that deserves your respect. Just remember: With fever and purpura, or fever and petechiae, meningococcemia needs to be at the top of your differential," stressed Dr. Anderson, a pediatric infectious diseases specialist at the University of Colorado.
The attack rate is highest in infants and young children because they haven’t yet experienced the transient nasopharyngeal colonization by Neisseria species that permits formation of protective strain-specific antibodies, she explained.
Empiric therapy until the diagnostic lab work and drug susceptibilities become available is vancomycin plus ceftriaxone. This combination also covers the other serious infectious causes of pediatric fever and purpura: Haemophilus influenzae, Streptococcus pneumoniae, and Staphylococcus aureus.
Once Neisseria meningitidis has been isolated, the antibiotic therapy can be narrowed to penicillin, cefotaxime, or ceftriaxone. The recommended treatment duration is 7 days for uncomplicated meningococcemia or meningococcal meningitis.
S. pneumoniae causes fever and purpura fulminans mainly in children who are immunocompromised.
Fever with petechiae
"Rocky Mountain spotted fever is another diagnosis you really don’t want to miss. You want to make this diagnosis and institute therapy promptly. Many studies have shown decreased mortality if doxycycline is initiated in the first 5 days of illness," Dr. Anderson said.
That can be a challenge. The distinctive petechial rash, which begins on the hands, wrists, feet, and ankles, and then moves centrally, typically doesn’t show up until day 5 or 6. In one study of 92 patients admitted with an ultimate diagnosis of Rocky Mountain spotted fever, the rash on admission was maculopapular rather than petechial in about one-third of cases (J. Pediatr. 2007;150:180-4).
Fever, myalgias, and headache are common at the onset of the illness, which is typically 2-14 days after a tick bite involving transmission of Rickettsia rickettsii. The diagnosis must be made clinically as there is no rapid testing commercially available. Demonstration of a fourfold antibody titer rise between acute and convalescent serology can confirm the diagnosis. Only about one-half of confirmed cases feature patient recall of a tick bite, so absence of such a history shouldn’t allay suspicion that this disease is present.
Doxycycline is the first-line agent used for treatment of Rocky Mountain spotted fever in children of all ages. The usual length of doxycycline therapy is 7-14 days, or for at least several days after the patient is afebrile and clinically improved. In a patient with a life-threatening allergy to doxycycline, chloramphenicol is an alternative. Availability of chloramphenicol, especially in the oral formulation, may be limited in the United States.
Other serious infections involving fever and petechiae include meningococcemia, group A streptococcal infection, rat bite fever, ehrlichiosis, and anaplasmosis. Unlike Rocky Mountain spotted fever, where a petechial or maculopapular rash occurs in 90% of cases, only about 10% of patients with ehrlichiosis or anaplasmosis develop a rash.
Rat bite fever is an acute febrile illness caused by Streptobacillus moniliformis, which colonizes the nasopharynx of a substantial percentage of domestic and wild rats. Children can develop the illness by kissing their pet rat or letting it eat food from their mouth.
Most cases of rat bite fever resolve spontaneously within 2 weeks; however, the case-fatality rate is 13% without treatment, which is penicillin or doxycycline.
Fever with erythroderma, a sunburnlike rash
The differential diagnosis here consists of toxic shock syndrome, staphylococcal scalded skin, and Kawasaki disease. Drug reactions, including Stevens-Johnson syndrome, are an important noninfectious cause. Sulfa drugs, anticonvulsants, and NSAIDs are the drugs most commonly associated with Stevens-Johnson syndrome.
Suspect toxic shock syndrome when fever and erythroderma are accompanied by hypotension, red eyes and lips, and a strawberry tongue. These are patients who warrant transfer to an intensive care unit. A key treatment principle is to search out the source of infection and drain it, whether it is a paronychia, sinusitis, perirectal abscess, or some other focus. If tampons are present they should be removed. Many experts use vancomycin and clindamycin as empiric therapy to treat toxic shock syndrome. Toxic shock syndrome results in vascular leakage, so fluid resuscitation and support of vascular volume are vital.
Dr. Anderson reported having no financial conflicts of interest.
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
Infectious disease, purpuric, petechial, erythrodermic, Dr. Marsha S. Anderson, Children’s Hospital Colorado, meningococcemia, petechial, purpuric, nasopharyngeal colonization, Neisseria species, vancomycin plus ceftriaxone, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, Neisseria meningitidis, Rocky Mountain spotted fever,
EXPERT ANALYSIS FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
New drug for hepatitis C may put treatment in the hands of primary care
ESTES PARK, COLO. – Treatment of hepatitis C infection is likely to shift from gastroenterology and hepatology clinics to primary care physicians’ offices, perhaps as early as next year.
That’s the prediction of Dr. Gregory T. Everson, who cited rapid progress in developing interferon-free treatment regimens as the driving force behind what is anticipated to be a huge change in clinical practice, given that there are an estimated 5 million or more patients with hepatitis C virus (HCV) infection in the United States
Five major pharmaceutical companies are developing oral, interferon-free HCV treatment regimens based upon still-investigational direct-acting antiviral agents (DAAs) that target HCV proteins and their functions. The clinical trials experience to date indicates these DAA-based regimens have substantially higher cure rates, far better tolerability and safety, and a much lower pill burden. Also, they are vastly simpler to administer than are current standard therapies. And all this is being achieved with a 12-week treatment duration instead of the 24 weeks required with standard therapy in 2013, Dr. Everson said at a conference on internal medicine sponsored by the University of Colorado.
"In the past I could say we really had pretty [intolerable] therapy, and it was pretty difficult for patients to take it. Interferon-free therapy is evolving rapidly, and I hope that it will be in primary care physicians’ backyards in the near future. I think this is probably going to be a treatment that you all provide," added Dr. Everson, professor of medicine and director of hepatology at the university.
Indeed, at hepatology clinics around the country, patients with early-stage HCV and their physicians are taking a "wait until next year" attitude toward starting treatment in anticipation that the Food and Drug Administration could approve the first of these new DAAs, sofosbuvir, before the year’s end.
The number of prescriptions for HCV therapy for treatment-naive patients at the University of Colorado clinic has plummeted in 2013 compared with 2012. The only patients starting treatment now are those with advanced HCV liver disease, to halt further disease progression and reduce the risk of developing hepatocellular carcinoma, according to Dr. Everson.
The FDA has granted Gilead Sciences priority review status for sofosbuvir, an oral inhibitor of nucleotide N55b polymerase, with a target decision date in early December. The application for marketing approval is for two indications. One involves sofosbuvir as part of a 12-week, triple-therapy regimen including pegylated interferon and ribavirin in treatment-naive patients with HCV genotypes 1, 4, 5, and 6, based in large part upon the highly favorable results of the phase III NEUTRINO trial (N. Engl. J. Med. 2013;368:1878-87).
The other proposed indication is sofosbuvir in combination with ribavirin as an interferon-free, 12-week regimen in patients with HCV genotypes 2 or 3, based upon the results of the FISSION and POSITRON trials (N. Engl. J. Med. 2013;368:1867-77).
In the NEUTRINO trial, 12 weeks of triple-therapy with sofosbuvir/interferon/ribavirin resulted in a 90% cure rate in patients with HCV genotype 1, which accounts for three-quarters of all HCV infections in the United States. The dropout rate due to side effects was a mere 2%. In contrast, today’s standard therapy, consisting of either of the protease inhibitors telaprevir (Incivek) or boceprevir (Victrelis) combined with pegylated interferon and ribavirin, has a 70%-75% cure rate. And many patients can’t tolerate or are ineligible for interferon.
"When I started treating hepatitis C patients 25 years ago, I was happy because the ALT would normalize in half the patients with genotype 1, but I wasn’t curing anybody. And now triple therapy with sofosbuvir, with a 90% cure rate, could be approved by the end of the year," Dr. Everson marveled.
In the FISSION trial, treatment-naive patients with HCV genotype 2 had a 97% cure rate with 12 weeks of sofosbuvir/ribavirin compared with 78% in those assigned to today’s standard regimen of 24 weeks of pegylated interferon/ribavirin. And sofosbuvir is just one pill per day, coupled with ribavirin at two or three pills twice daily.
In POSITRON, conducted in patients who had relapsed or were nonresponders to the standard 24 weeks of pegylated interferon/ribavirin, 12 weeks of sofosbuvir/ribavirin had an 86% response rate in patients with HCV genotype 2. With an additional 4 weeks of the interferon-free regimen, the cure rate climbed to 94%. Cure rates were lower in genotype 3 patients, but of note, the cure rate in treatment-experienced genotype 3 patients with cirrhosis more than tripled from 19% with 12 weeks of sofosbuvir/ribavirin to 61% with 16 weeks.
Other oral DAAs in the developmental pipeline include simeprevir, daclatasvir, and asunaprevir. The clinical trials experience to date demonstrate that combination therapy with more than one DAA boosts the cure rate even higher than with sofosbuvir/ribavirin. For example, in the phase II, open-label AVIATOR study, 12 weeks of a cocktail comprising three DAAs plus ribavirin brought a 96% cure rate in treatment-naive patients with HCV genotype 1 and a 93% cure rate in those who had previously failed on standard interferon-containing therapy. The AVIATOR cocktail is being formulated as a two-pills-per-day regimen.
Further, Dr. Everson was principal investigator in a Bristol-Myers Squibb–sponsored study of a totally interferon- and ribavirin-free regimen consisting of triple-DAA therapy. Each of the DAAs has a different mechanism of action: Daclatasvir is an inhibitor of the HCV NS5a protein; asunaprevir is an NS3 protease inhibitor; and the agent known for now as BMS-791325 is a nonnucleoside polymerase NS5b inhibitor. The cure rate with 12 weeks of triple-DAA therapy in treatment-naive, noncirrhotic patients with HCV genotype 1 was 94%.
Hepatologists define cure of HCV as an SVR12, or a sustained virologic response featuring no detectable HCV RNA in the blood for 12 weeks after the conclusion of therapy. The likelihood that a patient who achieves an SVR12 will remain HCV free through 10 years is 99%-100% (Gastroenterology 2010;139:1593-1601).
"The future looks pretty good for hepatitis C patients," Dr. Everson observed.
Moreover, curing HCV is going to have major downstream benefits for the health care system, he added. Today, 36% of all patients on the liver transplantation waiting list have HCV; that proportion will drop substantially. There will be fewer cases of hepatocellular carcinoma, B-cell lymphoma, and adult-onset diabetes, a drop in HCV-related autoimmune disorders, and reduced costs of care for patients with chronic HCV.
Dr. Everson reported that he receives research grants from and/or serves as a consultant to or advisory board member for roughly two dozen pharmaceutical companies.
ESTES PARK, COLO. – Treatment of hepatitis C infection is likely to shift from gastroenterology and hepatology clinics to primary care physicians’ offices, perhaps as early as next year.
That’s the prediction of Dr. Gregory T. Everson, who cited rapid progress in developing interferon-free treatment regimens as the driving force behind what is anticipated to be a huge change in clinical practice, given that there are an estimated 5 million or more patients with hepatitis C virus (HCV) infection in the United States
Five major pharmaceutical companies are developing oral, interferon-free HCV treatment regimens based upon still-investigational direct-acting antiviral agents (DAAs) that target HCV proteins and their functions. The clinical trials experience to date indicates these DAA-based regimens have substantially higher cure rates, far better tolerability and safety, and a much lower pill burden. Also, they are vastly simpler to administer than are current standard therapies. And all this is being achieved with a 12-week treatment duration instead of the 24 weeks required with standard therapy in 2013, Dr. Everson said at a conference on internal medicine sponsored by the University of Colorado.
"In the past I could say we really had pretty [intolerable] therapy, and it was pretty difficult for patients to take it. Interferon-free therapy is evolving rapidly, and I hope that it will be in primary care physicians’ backyards in the near future. I think this is probably going to be a treatment that you all provide," added Dr. Everson, professor of medicine and director of hepatology at the university.
Indeed, at hepatology clinics around the country, patients with early-stage HCV and their physicians are taking a "wait until next year" attitude toward starting treatment in anticipation that the Food and Drug Administration could approve the first of these new DAAs, sofosbuvir, before the year’s end.
The number of prescriptions for HCV therapy for treatment-naive patients at the University of Colorado clinic has plummeted in 2013 compared with 2012. The only patients starting treatment now are those with advanced HCV liver disease, to halt further disease progression and reduce the risk of developing hepatocellular carcinoma, according to Dr. Everson.
The FDA has granted Gilead Sciences priority review status for sofosbuvir, an oral inhibitor of nucleotide N55b polymerase, with a target decision date in early December. The application for marketing approval is for two indications. One involves sofosbuvir as part of a 12-week, triple-therapy regimen including pegylated interferon and ribavirin in treatment-naive patients with HCV genotypes 1, 4, 5, and 6, based in large part upon the highly favorable results of the phase III NEUTRINO trial (N. Engl. J. Med. 2013;368:1878-87).
The other proposed indication is sofosbuvir in combination with ribavirin as an interferon-free, 12-week regimen in patients with HCV genotypes 2 or 3, based upon the results of the FISSION and POSITRON trials (N. Engl. J. Med. 2013;368:1867-77).
In the NEUTRINO trial, 12 weeks of triple-therapy with sofosbuvir/interferon/ribavirin resulted in a 90% cure rate in patients with HCV genotype 1, which accounts for three-quarters of all HCV infections in the United States. The dropout rate due to side effects was a mere 2%. In contrast, today’s standard therapy, consisting of either of the protease inhibitors telaprevir (Incivek) or boceprevir (Victrelis) combined with pegylated interferon and ribavirin, has a 70%-75% cure rate. And many patients can’t tolerate or are ineligible for interferon.
"When I started treating hepatitis C patients 25 years ago, I was happy because the ALT would normalize in half the patients with genotype 1, but I wasn’t curing anybody. And now triple therapy with sofosbuvir, with a 90% cure rate, could be approved by the end of the year," Dr. Everson marveled.
In the FISSION trial, treatment-naive patients with HCV genotype 2 had a 97% cure rate with 12 weeks of sofosbuvir/ribavirin compared with 78% in those assigned to today’s standard regimen of 24 weeks of pegylated interferon/ribavirin. And sofosbuvir is just one pill per day, coupled with ribavirin at two or three pills twice daily.
In POSITRON, conducted in patients who had relapsed or were nonresponders to the standard 24 weeks of pegylated interferon/ribavirin, 12 weeks of sofosbuvir/ribavirin had an 86% response rate in patients with HCV genotype 2. With an additional 4 weeks of the interferon-free regimen, the cure rate climbed to 94%. Cure rates were lower in genotype 3 patients, but of note, the cure rate in treatment-experienced genotype 3 patients with cirrhosis more than tripled from 19% with 12 weeks of sofosbuvir/ribavirin to 61% with 16 weeks.
Other oral DAAs in the developmental pipeline include simeprevir, daclatasvir, and asunaprevir. The clinical trials experience to date demonstrate that combination therapy with more than one DAA boosts the cure rate even higher than with sofosbuvir/ribavirin. For example, in the phase II, open-label AVIATOR study, 12 weeks of a cocktail comprising three DAAs plus ribavirin brought a 96% cure rate in treatment-naive patients with HCV genotype 1 and a 93% cure rate in those who had previously failed on standard interferon-containing therapy. The AVIATOR cocktail is being formulated as a two-pills-per-day regimen.
Further, Dr. Everson was principal investigator in a Bristol-Myers Squibb–sponsored study of a totally interferon- and ribavirin-free regimen consisting of triple-DAA therapy. Each of the DAAs has a different mechanism of action: Daclatasvir is an inhibitor of the HCV NS5a protein; asunaprevir is an NS3 protease inhibitor; and the agent known for now as BMS-791325 is a nonnucleoside polymerase NS5b inhibitor. The cure rate with 12 weeks of triple-DAA therapy in treatment-naive, noncirrhotic patients with HCV genotype 1 was 94%.
Hepatologists define cure of HCV as an SVR12, or a sustained virologic response featuring no detectable HCV RNA in the blood for 12 weeks after the conclusion of therapy. The likelihood that a patient who achieves an SVR12 will remain HCV free through 10 years is 99%-100% (Gastroenterology 2010;139:1593-1601).
"The future looks pretty good for hepatitis C patients," Dr. Everson observed.
Moreover, curing HCV is going to have major downstream benefits for the health care system, he added. Today, 36% of all patients on the liver transplantation waiting list have HCV; that proportion will drop substantially. There will be fewer cases of hepatocellular carcinoma, B-cell lymphoma, and adult-onset diabetes, a drop in HCV-related autoimmune disorders, and reduced costs of care for patients with chronic HCV.
Dr. Everson reported that he receives research grants from and/or serves as a consultant to or advisory board member for roughly two dozen pharmaceutical companies.
ESTES PARK, COLO. – Treatment of hepatitis C infection is likely to shift from gastroenterology and hepatology clinics to primary care physicians’ offices, perhaps as early as next year.
That’s the prediction of Dr. Gregory T. Everson, who cited rapid progress in developing interferon-free treatment regimens as the driving force behind what is anticipated to be a huge change in clinical practice, given that there are an estimated 5 million or more patients with hepatitis C virus (HCV) infection in the United States
Five major pharmaceutical companies are developing oral, interferon-free HCV treatment regimens based upon still-investigational direct-acting antiviral agents (DAAs) that target HCV proteins and their functions. The clinical trials experience to date indicates these DAA-based regimens have substantially higher cure rates, far better tolerability and safety, and a much lower pill burden. Also, they are vastly simpler to administer than are current standard therapies. And all this is being achieved with a 12-week treatment duration instead of the 24 weeks required with standard therapy in 2013, Dr. Everson said at a conference on internal medicine sponsored by the University of Colorado.
"In the past I could say we really had pretty [intolerable] therapy, and it was pretty difficult for patients to take it. Interferon-free therapy is evolving rapidly, and I hope that it will be in primary care physicians’ backyards in the near future. I think this is probably going to be a treatment that you all provide," added Dr. Everson, professor of medicine and director of hepatology at the university.
Indeed, at hepatology clinics around the country, patients with early-stage HCV and their physicians are taking a "wait until next year" attitude toward starting treatment in anticipation that the Food and Drug Administration could approve the first of these new DAAs, sofosbuvir, before the year’s end.
The number of prescriptions for HCV therapy for treatment-naive patients at the University of Colorado clinic has plummeted in 2013 compared with 2012. The only patients starting treatment now are those with advanced HCV liver disease, to halt further disease progression and reduce the risk of developing hepatocellular carcinoma, according to Dr. Everson.
The FDA has granted Gilead Sciences priority review status for sofosbuvir, an oral inhibitor of nucleotide N55b polymerase, with a target decision date in early December. The application for marketing approval is for two indications. One involves sofosbuvir as part of a 12-week, triple-therapy regimen including pegylated interferon and ribavirin in treatment-naive patients with HCV genotypes 1, 4, 5, and 6, based in large part upon the highly favorable results of the phase III NEUTRINO trial (N. Engl. J. Med. 2013;368:1878-87).
The other proposed indication is sofosbuvir in combination with ribavirin as an interferon-free, 12-week regimen in patients with HCV genotypes 2 or 3, based upon the results of the FISSION and POSITRON trials (N. Engl. J. Med. 2013;368:1867-77).
In the NEUTRINO trial, 12 weeks of triple-therapy with sofosbuvir/interferon/ribavirin resulted in a 90% cure rate in patients with HCV genotype 1, which accounts for three-quarters of all HCV infections in the United States. The dropout rate due to side effects was a mere 2%. In contrast, today’s standard therapy, consisting of either of the protease inhibitors telaprevir (Incivek) or boceprevir (Victrelis) combined with pegylated interferon and ribavirin, has a 70%-75% cure rate. And many patients can’t tolerate or are ineligible for interferon.
"When I started treating hepatitis C patients 25 years ago, I was happy because the ALT would normalize in half the patients with genotype 1, but I wasn’t curing anybody. And now triple therapy with sofosbuvir, with a 90% cure rate, could be approved by the end of the year," Dr. Everson marveled.
In the FISSION trial, treatment-naive patients with HCV genotype 2 had a 97% cure rate with 12 weeks of sofosbuvir/ribavirin compared with 78% in those assigned to today’s standard regimen of 24 weeks of pegylated interferon/ribavirin. And sofosbuvir is just one pill per day, coupled with ribavirin at two or three pills twice daily.
In POSITRON, conducted in patients who had relapsed or were nonresponders to the standard 24 weeks of pegylated interferon/ribavirin, 12 weeks of sofosbuvir/ribavirin had an 86% response rate in patients with HCV genotype 2. With an additional 4 weeks of the interferon-free regimen, the cure rate climbed to 94%. Cure rates were lower in genotype 3 patients, but of note, the cure rate in treatment-experienced genotype 3 patients with cirrhosis more than tripled from 19% with 12 weeks of sofosbuvir/ribavirin to 61% with 16 weeks.
Other oral DAAs in the developmental pipeline include simeprevir, daclatasvir, and asunaprevir. The clinical trials experience to date demonstrate that combination therapy with more than one DAA boosts the cure rate even higher than with sofosbuvir/ribavirin. For example, in the phase II, open-label AVIATOR study, 12 weeks of a cocktail comprising three DAAs plus ribavirin brought a 96% cure rate in treatment-naive patients with HCV genotype 1 and a 93% cure rate in those who had previously failed on standard interferon-containing therapy. The AVIATOR cocktail is being formulated as a two-pills-per-day regimen.
Further, Dr. Everson was principal investigator in a Bristol-Myers Squibb–sponsored study of a totally interferon- and ribavirin-free regimen consisting of triple-DAA therapy. Each of the DAAs has a different mechanism of action: Daclatasvir is an inhibitor of the HCV NS5a protein; asunaprevir is an NS3 protease inhibitor; and the agent known for now as BMS-791325 is a nonnucleoside polymerase NS5b inhibitor. The cure rate with 12 weeks of triple-DAA therapy in treatment-naive, noncirrhotic patients with HCV genotype 1 was 94%.
Hepatologists define cure of HCV as an SVR12, or a sustained virologic response featuring no detectable HCV RNA in the blood for 12 weeks after the conclusion of therapy. The likelihood that a patient who achieves an SVR12 will remain HCV free through 10 years is 99%-100% (Gastroenterology 2010;139:1593-1601).
"The future looks pretty good for hepatitis C patients," Dr. Everson observed.
Moreover, curing HCV is going to have major downstream benefits for the health care system, he added. Today, 36% of all patients on the liver transplantation waiting list have HCV; that proportion will drop substantially. There will be fewer cases of hepatocellular carcinoma, B-cell lymphoma, and adult-onset diabetes, a drop in HCV-related autoimmune disorders, and reduced costs of care for patients with chronic HCV.
Dr. Everson reported that he receives research grants from and/or serves as a consultant to or advisory board member for roughly two dozen pharmaceutical companies.
AT THE ANNUAL INTERNAL MEDICINE PROGRAM