User login
Dapagliflozin explored in type 1 diabetes
CHICAGO – Even as dapagliflozin’s resubmitted application for marketing approval for treatment of type 2 diabetes is being scrutinized by the Food and Drug Administration, the drug is under study as a novel potential oral therapy for type 1 diabetes.
"For type 1 diabetes there are no approved oral agents, although some people use metformin off label. But I would predict if the numbers we saw with dapagliflozin in this short-term study persist out to 3 months, it would mean a reduction in hemoglobin A1c of 0.7-1.0 percentage points in type 1 diabetic patients who started at 8.0%," Dr. Robert R. Henry said in an interview at the annual scientific sessions of the American Diabetes Association.
He presented a small, proof-of-concept, multicenter, double-blind, phase IIa study aimed at establishing the safety of dapagliflozin, a sodium glucose cotransporter 2 (SGLT-2) inhibitor, in the management of patients with type 1 diabetes on background insulin. The study also showed early evidence of efficacy.
The five-center trial involved 62 patients who had suboptimally controlled type 1 diabetes despite being on basal bolus insulin or continuous infusion pump therapy. Their mean baseline HbA1c was 8.5%. Since this was the first study of an SGLT-2 inhibitor in patients with type 1 diabetes, it was conducted on an inpatient basis. For the first 3 days, patients were stabilized. Then they were randomized to insulin plus dapagliflozin at 1, 2.5, 5, or 10 mg once daily or to placebo.
The SGLT-2 inhibitor–treated patients demonstrated a dose-dependent increase in 24-hour urine glucose excretion. At the most effective 5- and 10-mg doses, by day 7 the 24-hour urine glucose excretion reached 84 and 100 g, respectively, representing mean 72-g and 89-g increases from baseline, compared to a 22-g decrease from baseline in the placebo group. Yet the dapagliflozin-treated patients showed no significant change in daily urine volume.
Continuous glucose monitoring showed mean 30- and 41-mg/dL reductions in daily average blood glucose levels in the 5- and 10-mg dapagliflozin groups, from a baseline of 174 mg/dL. Daily blood glucose variability decreased by 16%-25% as well, reported Dr. Henry, professor of medicine at the University of California, San Diego, and chief of the section of endocrinology, metabolism, and diabetes at Veterans Affairs Health Care System in San Diego.
Total daily insulin dosing on day 7 was down from baseline by 19% and 16%, respectively, in the 5- and 10-mg dapagliflozin-treated patients.
"This is just what I would have predicted, that dapagliflozin would be very effective in type 1 diabetes," he said.
Hypoglycemia was common in all study arms. The one case of serious hypoglycemia occurred in the dapagliflozin 10-mg group and led to study discontinuation. Two genital infections occurred in the dapagliflozin-treated patients.
Larger, longer-term clinical trials of the SGLT-2 inhibitor in patients with type 1 diabetes are planned.
The FDA initially rejected the New Drug Application that would have made dapagliflozin the first SGLT-2 inhibitor approved for the treatment of patients with type 2 diabetes. The agency requested more data on the risk/benefit ratio. Since then, dapagliflozin received marketing approval in the European Union, and the FDA granted approval to canagliflozin (Invokana) as the first SGLT-2 inhibitor to reach the U.S. market. AstraZeneca and Bristol-Myers Squibb have resubmitted the application for dapagliflozin with additional data. The FDA has indicated a decision will be announced by January.
Dr. Henry reported serving as an adviser to AstraZeneca and Bristol-Myers Squibb, which sponsored the study.
CHICAGO – Even as dapagliflozin’s resubmitted application for marketing approval for treatment of type 2 diabetes is being scrutinized by the Food and Drug Administration, the drug is under study as a novel potential oral therapy for type 1 diabetes.
"For type 1 diabetes there are no approved oral agents, although some people use metformin off label. But I would predict if the numbers we saw with dapagliflozin in this short-term study persist out to 3 months, it would mean a reduction in hemoglobin A1c of 0.7-1.0 percentage points in type 1 diabetic patients who started at 8.0%," Dr. Robert R. Henry said in an interview at the annual scientific sessions of the American Diabetes Association.
He presented a small, proof-of-concept, multicenter, double-blind, phase IIa study aimed at establishing the safety of dapagliflozin, a sodium glucose cotransporter 2 (SGLT-2) inhibitor, in the management of patients with type 1 diabetes on background insulin. The study also showed early evidence of efficacy.
The five-center trial involved 62 patients who had suboptimally controlled type 1 diabetes despite being on basal bolus insulin or continuous infusion pump therapy. Their mean baseline HbA1c was 8.5%. Since this was the first study of an SGLT-2 inhibitor in patients with type 1 diabetes, it was conducted on an inpatient basis. For the first 3 days, patients were stabilized. Then they were randomized to insulin plus dapagliflozin at 1, 2.5, 5, or 10 mg once daily or to placebo.
The SGLT-2 inhibitor–treated patients demonstrated a dose-dependent increase in 24-hour urine glucose excretion. At the most effective 5- and 10-mg doses, by day 7 the 24-hour urine glucose excretion reached 84 and 100 g, respectively, representing mean 72-g and 89-g increases from baseline, compared to a 22-g decrease from baseline in the placebo group. Yet the dapagliflozin-treated patients showed no significant change in daily urine volume.
Continuous glucose monitoring showed mean 30- and 41-mg/dL reductions in daily average blood glucose levels in the 5- and 10-mg dapagliflozin groups, from a baseline of 174 mg/dL. Daily blood glucose variability decreased by 16%-25% as well, reported Dr. Henry, professor of medicine at the University of California, San Diego, and chief of the section of endocrinology, metabolism, and diabetes at Veterans Affairs Health Care System in San Diego.
Total daily insulin dosing on day 7 was down from baseline by 19% and 16%, respectively, in the 5- and 10-mg dapagliflozin-treated patients.
"This is just what I would have predicted, that dapagliflozin would be very effective in type 1 diabetes," he said.
Hypoglycemia was common in all study arms. The one case of serious hypoglycemia occurred in the dapagliflozin 10-mg group and led to study discontinuation. Two genital infections occurred in the dapagliflozin-treated patients.
Larger, longer-term clinical trials of the SGLT-2 inhibitor in patients with type 1 diabetes are planned.
The FDA initially rejected the New Drug Application that would have made dapagliflozin the first SGLT-2 inhibitor approved for the treatment of patients with type 2 diabetes. The agency requested more data on the risk/benefit ratio. Since then, dapagliflozin received marketing approval in the European Union, and the FDA granted approval to canagliflozin (Invokana) as the first SGLT-2 inhibitor to reach the U.S. market. AstraZeneca and Bristol-Myers Squibb have resubmitted the application for dapagliflozin with additional data. The FDA has indicated a decision will be announced by January.
Dr. Henry reported serving as an adviser to AstraZeneca and Bristol-Myers Squibb, which sponsored the study.
CHICAGO – Even as dapagliflozin’s resubmitted application for marketing approval for treatment of type 2 diabetes is being scrutinized by the Food and Drug Administration, the drug is under study as a novel potential oral therapy for type 1 diabetes.
"For type 1 diabetes there are no approved oral agents, although some people use metformin off label. But I would predict if the numbers we saw with dapagliflozin in this short-term study persist out to 3 months, it would mean a reduction in hemoglobin A1c of 0.7-1.0 percentage points in type 1 diabetic patients who started at 8.0%," Dr. Robert R. Henry said in an interview at the annual scientific sessions of the American Diabetes Association.
He presented a small, proof-of-concept, multicenter, double-blind, phase IIa study aimed at establishing the safety of dapagliflozin, a sodium glucose cotransporter 2 (SGLT-2) inhibitor, in the management of patients with type 1 diabetes on background insulin. The study also showed early evidence of efficacy.
The five-center trial involved 62 patients who had suboptimally controlled type 1 diabetes despite being on basal bolus insulin or continuous infusion pump therapy. Their mean baseline HbA1c was 8.5%. Since this was the first study of an SGLT-2 inhibitor in patients with type 1 diabetes, it was conducted on an inpatient basis. For the first 3 days, patients were stabilized. Then they were randomized to insulin plus dapagliflozin at 1, 2.5, 5, or 10 mg once daily or to placebo.
The SGLT-2 inhibitor–treated patients demonstrated a dose-dependent increase in 24-hour urine glucose excretion. At the most effective 5- and 10-mg doses, by day 7 the 24-hour urine glucose excretion reached 84 and 100 g, respectively, representing mean 72-g and 89-g increases from baseline, compared to a 22-g decrease from baseline in the placebo group. Yet the dapagliflozin-treated patients showed no significant change in daily urine volume.
Continuous glucose monitoring showed mean 30- and 41-mg/dL reductions in daily average blood glucose levels in the 5- and 10-mg dapagliflozin groups, from a baseline of 174 mg/dL. Daily blood glucose variability decreased by 16%-25% as well, reported Dr. Henry, professor of medicine at the University of California, San Diego, and chief of the section of endocrinology, metabolism, and diabetes at Veterans Affairs Health Care System in San Diego.
Total daily insulin dosing on day 7 was down from baseline by 19% and 16%, respectively, in the 5- and 10-mg dapagliflozin-treated patients.
"This is just what I would have predicted, that dapagliflozin would be very effective in type 1 diabetes," he said.
Hypoglycemia was common in all study arms. The one case of serious hypoglycemia occurred in the dapagliflozin 10-mg group and led to study discontinuation. Two genital infections occurred in the dapagliflozin-treated patients.
Larger, longer-term clinical trials of the SGLT-2 inhibitor in patients with type 1 diabetes are planned.
The FDA initially rejected the New Drug Application that would have made dapagliflozin the first SGLT-2 inhibitor approved for the treatment of patients with type 2 diabetes. The agency requested more data on the risk/benefit ratio. Since then, dapagliflozin received marketing approval in the European Union, and the FDA granted approval to canagliflozin (Invokana) as the first SGLT-2 inhibitor to reach the U.S. market. AstraZeneca and Bristol-Myers Squibb have resubmitted the application for dapagliflozin with additional data. The FDA has indicated a decision will be announced by January.
Dr. Henry reported serving as an adviser to AstraZeneca and Bristol-Myers Squibb, which sponsored the study.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Patients with suboptimally controlled type 1 diabetes on insulin therapy who were randomized to add-on once-daily oral dapagliflozin excreted up to 100 g of glucose in their urine daily, while their average 24-hour blood glucose level fell by 41 mg/dL.
Data source: A randomized, double-blind, placebo-controlled, multicenter, short-term, inpatient, phase IIa study in 62 patients with type 1 diabetes suboptimally controlled with basal bolus insulin or insulin pump therapy.
Disclosures: AstraZeneca and Bristol-Myers Squibb sponsored the study. The presenter is an adviser to those pharmaceutical companies and about a dozen others.
Avoid C. difficile testing in infants
VAIL, COLO. – The worldly private eye in many a film noir has cautioned a potential client, "It’s best not to ask questions you’d rather not know the answer to."
That warning is apt as well when it comes to Clostridium difficile testing in infants with persistent diarrhea.
"We discourage testing of infants because we can’t actually distinguish colonization from C. difficile disease. I think that’s a big, complicated issue. And we get this question not infrequently: ‘What do we do with this positive test in a patient less than 1 year old?’ My answer is, ‘I don’t know. You shouldn’t test them in the first place,’ " Dr. Samuel R. Dominguez said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"Keep in mind that there are a lot of reasons other than C. difficile disease why an infant can have persistent diarrhea. I wouldn’t want anyone to be deluded by a positive C. difficile test. So I would have great caution in testing anyone under 1 year of age. It is true that you can have C. difficile infection under 1 year of age that can cause disease, but it is a rare event, not a common one," stressed Dr. Dominguez, a pediatric infectious diseases specialist at the hospital and the University of Colorado, Denver.
Unlike C. difficile disease, asymptomatic C. difficile colonization is extremely common in infants. For example, a French study involving stool testing of 85 asymptomatic infants in two day-care centers found a C. difficile carriage prevalence of 36% at age 2-6 months, climbing to 67% at 7-9 months, then peaking at 75% at 10-12 months, at which point 19% of the children harbored toxigenic strains. Yet none of the children had clinical disease at any point. After 12 months of age, the carriage rate declined steadily to 6% at 24-36 months (Clin. Infect. Dis. 2012;55:1209-15), which is comparable to the asymptomatic carriage rate in adults.
The significance of the high rate of C. difficile colonization in infants is controversial and poorly understood. It’s known that breastfed infants are less likely to be colonized than formula-fed infants, and that maternal-fetal transmission is rarely the source of infant colonization. Infants have the same rates of colonization and toxin production regardless of whether they have diarrhea, he noted.
Among the hypotheses offered to explain the rarity of C. difficile symptomatic disease in infants despite their high colonization rate is the observation that the gut microbial flora early in life is still in the process of formation. Its composition doesn’t approach that of the adult microbiome until about 1 year of age. Also, the infant immune system is immature. It is possible, although speculative, that the high rate of infant colonization is the result of a lack of protective gut bacteria early in life, while the rarity of symptomatic disease is a consequence of the immature immune system’s inability to recruit neutrophils, or alternatively is perhaps due to an absence of C. difficile receptors in the infant intestine.
Dr. Dominguez’ caution against testing for C. difficile in infants mirrors that of a recent American Academy of Pediatrics policy statement (Pediatrics 2013;131:196-200), which declared "it is prudent" to avoid testing in children under age 1 year. The AAP recommends first looking for other possible causes of diarrhea, especially viral pathogens, before testing for C. difficile in 1- to 3-year-olds. In symptomatic children over age 3, a positive test indicates probable infection.
An intriguing speculation recently raised in the literature is that colonized children might pose a risk to adults, serving as a reservoir for infection in much the same way they act as a reservoir for respiratory pathogens including influenza and pneumococcus (Clin. Infect. Dis. 2013;57:9-12). There is, however, no evidence as yet for this notion, Dr. Dominguez noted.
Current practice guidelines from the American College of Gastroenterology (Am. J. Gastroenterol. 2013;108:478-98) and Infectious Diseases Society of America (Infect. Control Hosp. Epidemiol. 2010;31:431-55) recommend PCR for C. difficile toxin genes as the standard diagnostic test. The guidelines list as an acceptable alternative a two-step method using a glutamate dehydrogenase test as a screen; if positive, it is to be followed by PCR or enzyme immunoassay. Dr. Dominguez indicated he has a problem with this recommendation because the glutamate dehydrogenase test has what he considers unacceptably high false-positive and -negative rates.
Enzyme immunoassay for toxins A and B is not endorsed as a stand-alone test in the latest guidelines because of its lack of sensitivity.
The guidelines further recommend that in light of the substantial asymptomatic colonization rates in both children and adults, only stools from symptomatic patients should be tested. Indeed, the microbiology laboratory at the Children’s Hospital Colorado adheres strictly to this guidance, refusing to test formed stool samples.
Repeat testing because of a suspected initial false-positive result is discouraged in the guidelines, as is a test-for-cure at the end of treatment, since virus, toxins, and genome are shed for weeks after the diarrhea is resolved.
"We see a lot of this test-for-cure at our hospital. However, testing for cure is not recommended in the guidelines and should not be done in most cases," Dr. Dominguez emphasized.
He reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – The worldly private eye in many a film noir has cautioned a potential client, "It’s best not to ask questions you’d rather not know the answer to."
That warning is apt as well when it comes to Clostridium difficile testing in infants with persistent diarrhea.
"We discourage testing of infants because we can’t actually distinguish colonization from C. difficile disease. I think that’s a big, complicated issue. And we get this question not infrequently: ‘What do we do with this positive test in a patient less than 1 year old?’ My answer is, ‘I don’t know. You shouldn’t test them in the first place,’ " Dr. Samuel R. Dominguez said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"Keep in mind that there are a lot of reasons other than C. difficile disease why an infant can have persistent diarrhea. I wouldn’t want anyone to be deluded by a positive C. difficile test. So I would have great caution in testing anyone under 1 year of age. It is true that you can have C. difficile infection under 1 year of age that can cause disease, but it is a rare event, not a common one," stressed Dr. Dominguez, a pediatric infectious diseases specialist at the hospital and the University of Colorado, Denver.
Unlike C. difficile disease, asymptomatic C. difficile colonization is extremely common in infants. For example, a French study involving stool testing of 85 asymptomatic infants in two day-care centers found a C. difficile carriage prevalence of 36% at age 2-6 months, climbing to 67% at 7-9 months, then peaking at 75% at 10-12 months, at which point 19% of the children harbored toxigenic strains. Yet none of the children had clinical disease at any point. After 12 months of age, the carriage rate declined steadily to 6% at 24-36 months (Clin. Infect. Dis. 2012;55:1209-15), which is comparable to the asymptomatic carriage rate in adults.
The significance of the high rate of C. difficile colonization in infants is controversial and poorly understood. It’s known that breastfed infants are less likely to be colonized than formula-fed infants, and that maternal-fetal transmission is rarely the source of infant colonization. Infants have the same rates of colonization and toxin production regardless of whether they have diarrhea, he noted.
Among the hypotheses offered to explain the rarity of C. difficile symptomatic disease in infants despite their high colonization rate is the observation that the gut microbial flora early in life is still in the process of formation. Its composition doesn’t approach that of the adult microbiome until about 1 year of age. Also, the infant immune system is immature. It is possible, although speculative, that the high rate of infant colonization is the result of a lack of protective gut bacteria early in life, while the rarity of symptomatic disease is a consequence of the immature immune system’s inability to recruit neutrophils, or alternatively is perhaps due to an absence of C. difficile receptors in the infant intestine.
Dr. Dominguez’ caution against testing for C. difficile in infants mirrors that of a recent American Academy of Pediatrics policy statement (Pediatrics 2013;131:196-200), which declared "it is prudent" to avoid testing in children under age 1 year. The AAP recommends first looking for other possible causes of diarrhea, especially viral pathogens, before testing for C. difficile in 1- to 3-year-olds. In symptomatic children over age 3, a positive test indicates probable infection.
An intriguing speculation recently raised in the literature is that colonized children might pose a risk to adults, serving as a reservoir for infection in much the same way they act as a reservoir for respiratory pathogens including influenza and pneumococcus (Clin. Infect. Dis. 2013;57:9-12). There is, however, no evidence as yet for this notion, Dr. Dominguez noted.
Current practice guidelines from the American College of Gastroenterology (Am. J. Gastroenterol. 2013;108:478-98) and Infectious Diseases Society of America (Infect. Control Hosp. Epidemiol. 2010;31:431-55) recommend PCR for C. difficile toxin genes as the standard diagnostic test. The guidelines list as an acceptable alternative a two-step method using a glutamate dehydrogenase test as a screen; if positive, it is to be followed by PCR or enzyme immunoassay. Dr. Dominguez indicated he has a problem with this recommendation because the glutamate dehydrogenase test has what he considers unacceptably high false-positive and -negative rates.
Enzyme immunoassay for toxins A and B is not endorsed as a stand-alone test in the latest guidelines because of its lack of sensitivity.
The guidelines further recommend that in light of the substantial asymptomatic colonization rates in both children and adults, only stools from symptomatic patients should be tested. Indeed, the microbiology laboratory at the Children’s Hospital Colorado adheres strictly to this guidance, refusing to test formed stool samples.
Repeat testing because of a suspected initial false-positive result is discouraged in the guidelines, as is a test-for-cure at the end of treatment, since virus, toxins, and genome are shed for weeks after the diarrhea is resolved.
"We see a lot of this test-for-cure at our hospital. However, testing for cure is not recommended in the guidelines and should not be done in most cases," Dr. Dominguez emphasized.
He reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – The worldly private eye in many a film noir has cautioned a potential client, "It’s best not to ask questions you’d rather not know the answer to."
That warning is apt as well when it comes to Clostridium difficile testing in infants with persistent diarrhea.
"We discourage testing of infants because we can’t actually distinguish colonization from C. difficile disease. I think that’s a big, complicated issue. And we get this question not infrequently: ‘What do we do with this positive test in a patient less than 1 year old?’ My answer is, ‘I don’t know. You shouldn’t test them in the first place,’ " Dr. Samuel R. Dominguez said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"Keep in mind that there are a lot of reasons other than C. difficile disease why an infant can have persistent diarrhea. I wouldn’t want anyone to be deluded by a positive C. difficile test. So I would have great caution in testing anyone under 1 year of age. It is true that you can have C. difficile infection under 1 year of age that can cause disease, but it is a rare event, not a common one," stressed Dr. Dominguez, a pediatric infectious diseases specialist at the hospital and the University of Colorado, Denver.
Unlike C. difficile disease, asymptomatic C. difficile colonization is extremely common in infants. For example, a French study involving stool testing of 85 asymptomatic infants in two day-care centers found a C. difficile carriage prevalence of 36% at age 2-6 months, climbing to 67% at 7-9 months, then peaking at 75% at 10-12 months, at which point 19% of the children harbored toxigenic strains. Yet none of the children had clinical disease at any point. After 12 months of age, the carriage rate declined steadily to 6% at 24-36 months (Clin. Infect. Dis. 2012;55:1209-15), which is comparable to the asymptomatic carriage rate in adults.
The significance of the high rate of C. difficile colonization in infants is controversial and poorly understood. It’s known that breastfed infants are less likely to be colonized than formula-fed infants, and that maternal-fetal transmission is rarely the source of infant colonization. Infants have the same rates of colonization and toxin production regardless of whether they have diarrhea, he noted.
Among the hypotheses offered to explain the rarity of C. difficile symptomatic disease in infants despite their high colonization rate is the observation that the gut microbial flora early in life is still in the process of formation. Its composition doesn’t approach that of the adult microbiome until about 1 year of age. Also, the infant immune system is immature. It is possible, although speculative, that the high rate of infant colonization is the result of a lack of protective gut bacteria early in life, while the rarity of symptomatic disease is a consequence of the immature immune system’s inability to recruit neutrophils, or alternatively is perhaps due to an absence of C. difficile receptors in the infant intestine.
Dr. Dominguez’ caution against testing for C. difficile in infants mirrors that of a recent American Academy of Pediatrics policy statement (Pediatrics 2013;131:196-200), which declared "it is prudent" to avoid testing in children under age 1 year. The AAP recommends first looking for other possible causes of diarrhea, especially viral pathogens, before testing for C. difficile in 1- to 3-year-olds. In symptomatic children over age 3, a positive test indicates probable infection.
An intriguing speculation recently raised in the literature is that colonized children might pose a risk to adults, serving as a reservoir for infection in much the same way they act as a reservoir for respiratory pathogens including influenza and pneumococcus (Clin. Infect. Dis. 2013;57:9-12). There is, however, no evidence as yet for this notion, Dr. Dominguez noted.
Current practice guidelines from the American College of Gastroenterology (Am. J. Gastroenterol. 2013;108:478-98) and Infectious Diseases Society of America (Infect. Control Hosp. Epidemiol. 2010;31:431-55) recommend PCR for C. difficile toxin genes as the standard diagnostic test. The guidelines list as an acceptable alternative a two-step method using a glutamate dehydrogenase test as a screen; if positive, it is to be followed by PCR or enzyme immunoassay. Dr. Dominguez indicated he has a problem with this recommendation because the glutamate dehydrogenase test has what he considers unacceptably high false-positive and -negative rates.
Enzyme immunoassay for toxins A and B is not endorsed as a stand-alone test in the latest guidelines because of its lack of sensitivity.
The guidelines further recommend that in light of the substantial asymptomatic colonization rates in both children and adults, only stools from symptomatic patients should be tested. Indeed, the microbiology laboratory at the Children’s Hospital Colorado adheres strictly to this guidance, refusing to test formed stool samples.
Repeat testing because of a suspected initial false-positive result is discouraged in the guidelines, as is a test-for-cure at the end of treatment, since virus, toxins, and genome are shed for weeks after the diarrhea is resolved.
"We see a lot of this test-for-cure at our hospital. However, testing for cure is not recommended in the guidelines and should not be done in most cases," Dr. Dominguez emphasized.
He reported having no relevant financial relationships with any commercial interests.
EXPERT ANALYSIS FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Risk of stopping dual-antiplatelet therapy after stenting depends upon the reason
AMSTERDAM – Not all coronary stent recipients who halt dual-antiplatelet therapy face an increased risk of stent thrombosis and other cardiac events; it all depends upon the reason they stopped, according to data from the large, prospective observational PARIS registry.
Indeed, patients who discontinue dual-antiplatelet therapy (DAPT) because their physician thinks they no longer need it actually have a significantly lower risk of cardiac events than do those who stay on the medications continuously for 2 full years, PARIS registry chair Dr. Roxana Mehran reported at the annual congress of the European Society of Cardiology.
In contrast, stent recipients who halt DAPT because of bleeding or noncompliance are at significantly increased risk during the next 30 days, after which their risk tails off and becomes statistically nonsignificant. This risk is greatest during the first 7 days after ceasing DAPT, when it is increased sevenfold, compared with remaining on DAPT, according to Dr. Mehran, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York.
She presented key results from the 15-center, multinational observational PARIS registry, in which 5,018 coronary stent recipients were prospectively followed for 2 years after percutaneous coronary intervention (PCI). Three-quarters of them had received second-generation drug-eluting stents. The novel finding in PARIS was that the risk of cardiac events after cessation of DAPT was not uniform; rather, it varied substantially depending upon the reason for the stoppage.
"Our findings show that in a real world setting, when physicians recommend discontinuation of DAPT – presumably because they feel their patients are stable – there is no increased risk of adverse events. But when patients simply don’t comply or are forced off DAPT because they are bleeding, their risk is significantly increased," Dr. Mehran said.
During the first 2 years post PCI, 57% of patients quit taking DAPT, either temporarily or permanently. Investigators classified them into one of three prespecified groups on the basis of their reason for stopping DAPT: "Discontinuation" was defined as physician-recommended cessation of DAPT for patients thought to be at low risk for cardiac events, "interruption" was a temporary halt for up to 14 days for surgery, and "disruption" of DAPT was cessation owing to bleeding or noncompliance.
Over the course of 2 years, 41% of study participants had DAPT discontinuation, 14% had disruption, and 11% had interruption. Discontinuation occurred after a mean of 382 days on DAPT, while disruption occurred at 230 days. Nearly 90% of patients in the discontinuation group stopped thienopyridine only, continuing on aspirin. In contrast, 70% of patients in the interruption group and 50% in the disruption group halted both drugs.
The 2-year overall rate of major adverse cardiac events – cardiac death, definite or probable stent thrombosis, MI, or clinically driven target lesion revascularization – was 11.5%. The adjusted risk among patients who discontinued DAPT under physician guidance was reduced by 37% compared with the risk among patients who remained on DAPT for 2 years (P = .004). Patients who interrupted DAPT for surgical necessity showed a nonsignificant trend for increased risk of cardiac events. And DAPT disruption was associated with a sevenfold increased risk in the first week off treatment and a 2.2-fold increased risk during days 8-30.
Of note, 74% of all cardiac events and 80% of definite or probable cases of stent thrombosis occurred while patients were on DAPT.
Session cochair Dr. Keith A. A. Fox put Dr. Mehran’s feet to the fire by posing a challenging clinical scenario: What will you advise, he inquired, the next time a colleague asks what to do for a patient who had a coronary stent implanted 3 days ago and has just had a major GI bleed while on DAPT?
"This is what we call ‘the DAPT dilemma,’ " she replied. "It’s a critically important question. We all face it. We now know from PARIS that if we disrupt the treatment due to bleeding, the hazard in that first week is going to be increased sevenfold. So I think we should do everything we can to control the bleeding so we can keep the patient on DAPT, but if we must, then I think stopping one drug would be better than stopping two."
In an interview, American Heart Association spokesperson Dr. Elliott Antman said the PARIS registry shows that in general physicians are doing a fairly good job of intuitively deciding who can safely come off DAPT. But what’s really needed is the sort of well-defined practice guidance that can come only from a large, randomized trial.
Fortunately, he said, the results of such a study, the ongoing DAPT trial, should be available in about a year. This is a 15,000-patient clinical trial in which all stent recipients were placed on DAPT for 1 year, then randomized to come off of DAPT at that point or continue on, noted Dr. Antman, professor of medicine at Harvard Medical School, Boston.
The PARIS registry is funded by Bristol-Myers Squibb and Sanofi-Aventis. Dr. Mehran reported receiving consulting fees from Sanofi-Aventis and more than half a dozen other pharmaceutical and device companies.
Simultaneous with her presentation of the PARIS registry findings in Amsterdam, the results were published online in the Lancet (Sept. 1, 2013 [doi:10.1016/50140-6736(13)61720-1]).
The study was funded by Bristol-Myers Squibb and Sanofi-Aventis. The presenter is a paid consultant to Sanofi-Aventis and other companies.
AMSTERDAM – Not all coronary stent recipients who halt dual-antiplatelet therapy face an increased risk of stent thrombosis and other cardiac events; it all depends upon the reason they stopped, according to data from the large, prospective observational PARIS registry.
Indeed, patients who discontinue dual-antiplatelet therapy (DAPT) because their physician thinks they no longer need it actually have a significantly lower risk of cardiac events than do those who stay on the medications continuously for 2 full years, PARIS registry chair Dr. Roxana Mehran reported at the annual congress of the European Society of Cardiology.
In contrast, stent recipients who halt DAPT because of bleeding or noncompliance are at significantly increased risk during the next 30 days, after which their risk tails off and becomes statistically nonsignificant. This risk is greatest during the first 7 days after ceasing DAPT, when it is increased sevenfold, compared with remaining on DAPT, according to Dr. Mehran, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York.
She presented key results from the 15-center, multinational observational PARIS registry, in which 5,018 coronary stent recipients were prospectively followed for 2 years after percutaneous coronary intervention (PCI). Three-quarters of them had received second-generation drug-eluting stents. The novel finding in PARIS was that the risk of cardiac events after cessation of DAPT was not uniform; rather, it varied substantially depending upon the reason for the stoppage.
"Our findings show that in a real world setting, when physicians recommend discontinuation of DAPT – presumably because they feel their patients are stable – there is no increased risk of adverse events. But when patients simply don’t comply or are forced off DAPT because they are bleeding, their risk is significantly increased," Dr. Mehran said.
During the first 2 years post PCI, 57% of patients quit taking DAPT, either temporarily or permanently. Investigators classified them into one of three prespecified groups on the basis of their reason for stopping DAPT: "Discontinuation" was defined as physician-recommended cessation of DAPT for patients thought to be at low risk for cardiac events, "interruption" was a temporary halt for up to 14 days for surgery, and "disruption" of DAPT was cessation owing to bleeding or noncompliance.
Over the course of 2 years, 41% of study participants had DAPT discontinuation, 14% had disruption, and 11% had interruption. Discontinuation occurred after a mean of 382 days on DAPT, while disruption occurred at 230 days. Nearly 90% of patients in the discontinuation group stopped thienopyridine only, continuing on aspirin. In contrast, 70% of patients in the interruption group and 50% in the disruption group halted both drugs.
The 2-year overall rate of major adverse cardiac events – cardiac death, definite or probable stent thrombosis, MI, or clinically driven target lesion revascularization – was 11.5%. The adjusted risk among patients who discontinued DAPT under physician guidance was reduced by 37% compared with the risk among patients who remained on DAPT for 2 years (P = .004). Patients who interrupted DAPT for surgical necessity showed a nonsignificant trend for increased risk of cardiac events. And DAPT disruption was associated with a sevenfold increased risk in the first week off treatment and a 2.2-fold increased risk during days 8-30.
Of note, 74% of all cardiac events and 80% of definite or probable cases of stent thrombosis occurred while patients were on DAPT.
Session cochair Dr. Keith A. A. Fox put Dr. Mehran’s feet to the fire by posing a challenging clinical scenario: What will you advise, he inquired, the next time a colleague asks what to do for a patient who had a coronary stent implanted 3 days ago and has just had a major GI bleed while on DAPT?
"This is what we call ‘the DAPT dilemma,’ " she replied. "It’s a critically important question. We all face it. We now know from PARIS that if we disrupt the treatment due to bleeding, the hazard in that first week is going to be increased sevenfold. So I think we should do everything we can to control the bleeding so we can keep the patient on DAPT, but if we must, then I think stopping one drug would be better than stopping two."
In an interview, American Heart Association spokesperson Dr. Elliott Antman said the PARIS registry shows that in general physicians are doing a fairly good job of intuitively deciding who can safely come off DAPT. But what’s really needed is the sort of well-defined practice guidance that can come only from a large, randomized trial.
Fortunately, he said, the results of such a study, the ongoing DAPT trial, should be available in about a year. This is a 15,000-patient clinical trial in which all stent recipients were placed on DAPT for 1 year, then randomized to come off of DAPT at that point or continue on, noted Dr. Antman, professor of medicine at Harvard Medical School, Boston.
The PARIS registry is funded by Bristol-Myers Squibb and Sanofi-Aventis. Dr. Mehran reported receiving consulting fees from Sanofi-Aventis and more than half a dozen other pharmaceutical and device companies.
Simultaneous with her presentation of the PARIS registry findings in Amsterdam, the results were published online in the Lancet (Sept. 1, 2013 [doi:10.1016/50140-6736(13)61720-1]).
The study was funded by Bristol-Myers Squibb and Sanofi-Aventis. The presenter is a paid consultant to Sanofi-Aventis and other companies.
AMSTERDAM – Not all coronary stent recipients who halt dual-antiplatelet therapy face an increased risk of stent thrombosis and other cardiac events; it all depends upon the reason they stopped, according to data from the large, prospective observational PARIS registry.
Indeed, patients who discontinue dual-antiplatelet therapy (DAPT) because their physician thinks they no longer need it actually have a significantly lower risk of cardiac events than do those who stay on the medications continuously for 2 full years, PARIS registry chair Dr. Roxana Mehran reported at the annual congress of the European Society of Cardiology.
In contrast, stent recipients who halt DAPT because of bleeding or noncompliance are at significantly increased risk during the next 30 days, after which their risk tails off and becomes statistically nonsignificant. This risk is greatest during the first 7 days after ceasing DAPT, when it is increased sevenfold, compared with remaining on DAPT, according to Dr. Mehran, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York.
She presented key results from the 15-center, multinational observational PARIS registry, in which 5,018 coronary stent recipients were prospectively followed for 2 years after percutaneous coronary intervention (PCI). Three-quarters of them had received second-generation drug-eluting stents. The novel finding in PARIS was that the risk of cardiac events after cessation of DAPT was not uniform; rather, it varied substantially depending upon the reason for the stoppage.
"Our findings show that in a real world setting, when physicians recommend discontinuation of DAPT – presumably because they feel their patients are stable – there is no increased risk of adverse events. But when patients simply don’t comply or are forced off DAPT because they are bleeding, their risk is significantly increased," Dr. Mehran said.
During the first 2 years post PCI, 57% of patients quit taking DAPT, either temporarily or permanently. Investigators classified them into one of three prespecified groups on the basis of their reason for stopping DAPT: "Discontinuation" was defined as physician-recommended cessation of DAPT for patients thought to be at low risk for cardiac events, "interruption" was a temporary halt for up to 14 days for surgery, and "disruption" of DAPT was cessation owing to bleeding or noncompliance.
Over the course of 2 years, 41% of study participants had DAPT discontinuation, 14% had disruption, and 11% had interruption. Discontinuation occurred after a mean of 382 days on DAPT, while disruption occurred at 230 days. Nearly 90% of patients in the discontinuation group stopped thienopyridine only, continuing on aspirin. In contrast, 70% of patients in the interruption group and 50% in the disruption group halted both drugs.
The 2-year overall rate of major adverse cardiac events – cardiac death, definite or probable stent thrombosis, MI, or clinically driven target lesion revascularization – was 11.5%. The adjusted risk among patients who discontinued DAPT under physician guidance was reduced by 37% compared with the risk among patients who remained on DAPT for 2 years (P = .004). Patients who interrupted DAPT for surgical necessity showed a nonsignificant trend for increased risk of cardiac events. And DAPT disruption was associated with a sevenfold increased risk in the first week off treatment and a 2.2-fold increased risk during days 8-30.
Of note, 74% of all cardiac events and 80% of definite or probable cases of stent thrombosis occurred while patients were on DAPT.
Session cochair Dr. Keith A. A. Fox put Dr. Mehran’s feet to the fire by posing a challenging clinical scenario: What will you advise, he inquired, the next time a colleague asks what to do for a patient who had a coronary stent implanted 3 days ago and has just had a major GI bleed while on DAPT?
"This is what we call ‘the DAPT dilemma,’ " she replied. "It’s a critically important question. We all face it. We now know from PARIS that if we disrupt the treatment due to bleeding, the hazard in that first week is going to be increased sevenfold. So I think we should do everything we can to control the bleeding so we can keep the patient on DAPT, but if we must, then I think stopping one drug would be better than stopping two."
In an interview, American Heart Association spokesperson Dr. Elliott Antman said the PARIS registry shows that in general physicians are doing a fairly good job of intuitively deciding who can safely come off DAPT. But what’s really needed is the sort of well-defined practice guidance that can come only from a large, randomized trial.
Fortunately, he said, the results of such a study, the ongoing DAPT trial, should be available in about a year. This is a 15,000-patient clinical trial in which all stent recipients were placed on DAPT for 1 year, then randomized to come off of DAPT at that point or continue on, noted Dr. Antman, professor of medicine at Harvard Medical School, Boston.
The PARIS registry is funded by Bristol-Myers Squibb and Sanofi-Aventis. Dr. Mehran reported receiving consulting fees from Sanofi-Aventis and more than half a dozen other pharmaceutical and device companies.
Simultaneous with her presentation of the PARIS registry findings in Amsterdam, the results were published online in the Lancet (Sept. 1, 2013 [doi:10.1016/50140-6736(13)61720-1]).
The study was funded by Bristol-Myers Squibb and Sanofi-Aventis. The presenter is a paid consultant to Sanofi-Aventis and other companies.
AT THE ESC CONGRESS 2013
Major finding: Coronary stent recipients who discontinued dual-antiplatelet therapy because their physician felt they no longer needed it, most often after about 380 days, had a 37% lower risk of subsequent cardiac events than those who stayed on the medications for 2 years.
Data source: The PARIS registry, a 15-center, multinational, prospective observational registry in which 5,018 patients were followed for 2 years after stent implantation. Their reasons for stopping dual-antiplatelet therapy were recorded based on prespecified definitions.
Disclosures: The study was funded by Bristol-Myers Squibb and Sanofi-Aventis. The presenter is a paid consultant to Sanofi-Aventis and other companies.
Undetected silent AF is present in 3% of 75-year-olds
AMSTERDAM – A first-of-its-kind mass screening program identified fully 5% of 75- to 76-year-old Swedes as having untreated atrial fibrillation.
Three percent of the aged participants in the ongoing STROKESTOP study had previously unidentified silent atrial fibrillation (AF). Another 2% had been diagnosed with AF but were not on oral anticoagulation despite their substantial stroke risk, Dr. Emma Svennberg reported at the annual congress of the European Society of Cardiology.
The idea behind the screening program was to screen people at an age when the prevalence of AF is very high and yet there is sufficient remaining life expectancy that they could expect to gain substantial benefit from oral anticoagulation therapy for protection against cardioembolic stroke, explained Dr. Svennberg, a cardiologist at the Karolinska Institute, Stockholm.
The STROKESTOP study tested a novel mass screening strategy in which more than 6,000 Swedish 75- and 76-year-olds without known AF were issued a hand-held ECG device. Twice a day for 2 weeks they placed both thumbs on the device for 30 seconds to generate an ECG, which was transmitted to a central database. Compliance in this older age group was extremely high, although 1% of the ECGs were uninterpretable due to tremor, Parkinson’s disease, or other age-related physical infirmities.
Other mass screening programs have relied upon a single screening ECG. That’s a reliable means of detecting individuals with permanent AF, but very much hit-or-miss in those with paroxysmal AF, she noted.
Oral anticoagulant therapy was offered to all STROKESTOP participants found to have untreated AF. More than 90% of them accepted it. They are being prospectively followed for 5 years in the second phase of the STROKESTOP study, which will compare rates of ischemic stroke, major bleeding, mortality, and dementia in that group to a control group of 14,381 Swedish 75- and 76-year-olds – in the same two regions of the country – who were not invited to the screening program.
The prevalence of known AF in subjects invited to the screening program was 8.9%.
Individuals with AF have a fivefold increased risk of ischemic stroke, regardless of whether their atrial arrhythmia is silent or symptomatic. Oral anticoagulation therapy cuts that risk by close to 70%. Among patients with AF discharged from the hospital after a stroke, 70% die or become disabled. Yet undertreatment is common – in Sweden, as elsewhere, fewer than half of AF patients whose risk scores warrant anticoagulation therapy are on it, according to Dr. Svennberg.
Dr. Robert Hatala, session cochair, said one of the important messages of STROKESTOP is that you can’t find silent AF if you don’t look for it.
That’s why current ESC practice guidelines recommend that all patients aged 65 or older have their pulse taken at every physician visit, with an ECG to be ordered when any irregularities are detected, added Dr. Hatala, head of cardiology and director of the arrhythmia and pacing center at Slovak Medical University, Bratislava, Slovakia.
Dr. Svennberg said she and her coinvestigators are in the midst of a randomized trial comparing this low-tech screening method to the approach used in STROKESTOP involving 2 weeks of intermittent ECGs using a hand-held loaner device.
Dr. Hans-Christoph Diener, ESC spokesperson, predicted in an interview that physicians can soon expect to see a huge influx of patients with newly discovered silent AF. The impetus will be the much-anticipated introduction of the Apple iWatch, which he said could happen later this year.
"The detection of silent atrial fibrillation is going to change very quickly. This watch will have a device in it which measures pulse, and it has software which detects atrial fibrillation. More features are available for people with various diseases: It can measure oxygen saturation, pulse, blood pressure. They’ve even developed a chip to measure glucose transdermally. It’s amazing," said Dr. Diener, professor and chair of the department of neurology at the University of Duisburg-Essen, Germany.
The STROKESTOP study is supported by the Heart and Lung Foundation of Sweden and government research funds. Dr. Svennberg reported that she has received lecture fees from Merck Sharp & Dohme.
AMSTERDAM – A first-of-its-kind mass screening program identified fully 5% of 75- to 76-year-old Swedes as having untreated atrial fibrillation.
Three percent of the aged participants in the ongoing STROKESTOP study had previously unidentified silent atrial fibrillation (AF). Another 2% had been diagnosed with AF but were not on oral anticoagulation despite their substantial stroke risk, Dr. Emma Svennberg reported at the annual congress of the European Society of Cardiology.
The idea behind the screening program was to screen people at an age when the prevalence of AF is very high and yet there is sufficient remaining life expectancy that they could expect to gain substantial benefit from oral anticoagulation therapy for protection against cardioembolic stroke, explained Dr. Svennberg, a cardiologist at the Karolinska Institute, Stockholm.
The STROKESTOP study tested a novel mass screening strategy in which more than 6,000 Swedish 75- and 76-year-olds without known AF were issued a hand-held ECG device. Twice a day for 2 weeks they placed both thumbs on the device for 30 seconds to generate an ECG, which was transmitted to a central database. Compliance in this older age group was extremely high, although 1% of the ECGs were uninterpretable due to tremor, Parkinson’s disease, or other age-related physical infirmities.
Other mass screening programs have relied upon a single screening ECG. That’s a reliable means of detecting individuals with permanent AF, but very much hit-or-miss in those with paroxysmal AF, she noted.
Oral anticoagulant therapy was offered to all STROKESTOP participants found to have untreated AF. More than 90% of them accepted it. They are being prospectively followed for 5 years in the second phase of the STROKESTOP study, which will compare rates of ischemic stroke, major bleeding, mortality, and dementia in that group to a control group of 14,381 Swedish 75- and 76-year-olds – in the same two regions of the country – who were not invited to the screening program.
The prevalence of known AF in subjects invited to the screening program was 8.9%.
Individuals with AF have a fivefold increased risk of ischemic stroke, regardless of whether their atrial arrhythmia is silent or symptomatic. Oral anticoagulation therapy cuts that risk by close to 70%. Among patients with AF discharged from the hospital after a stroke, 70% die or become disabled. Yet undertreatment is common – in Sweden, as elsewhere, fewer than half of AF patients whose risk scores warrant anticoagulation therapy are on it, according to Dr. Svennberg.
Dr. Robert Hatala, session cochair, said one of the important messages of STROKESTOP is that you can’t find silent AF if you don’t look for it.
That’s why current ESC practice guidelines recommend that all patients aged 65 or older have their pulse taken at every physician visit, with an ECG to be ordered when any irregularities are detected, added Dr. Hatala, head of cardiology and director of the arrhythmia and pacing center at Slovak Medical University, Bratislava, Slovakia.
Dr. Svennberg said she and her coinvestigators are in the midst of a randomized trial comparing this low-tech screening method to the approach used in STROKESTOP involving 2 weeks of intermittent ECGs using a hand-held loaner device.
Dr. Hans-Christoph Diener, ESC spokesperson, predicted in an interview that physicians can soon expect to see a huge influx of patients with newly discovered silent AF. The impetus will be the much-anticipated introduction of the Apple iWatch, which he said could happen later this year.
"The detection of silent atrial fibrillation is going to change very quickly. This watch will have a device in it which measures pulse, and it has software which detects atrial fibrillation. More features are available for people with various diseases: It can measure oxygen saturation, pulse, blood pressure. They’ve even developed a chip to measure glucose transdermally. It’s amazing," said Dr. Diener, professor and chair of the department of neurology at the University of Duisburg-Essen, Germany.
The STROKESTOP study is supported by the Heart and Lung Foundation of Sweden and government research funds. Dr. Svennberg reported that she has received lecture fees from Merck Sharp & Dohme.
AMSTERDAM – A first-of-its-kind mass screening program identified fully 5% of 75- to 76-year-old Swedes as having untreated atrial fibrillation.
Three percent of the aged participants in the ongoing STROKESTOP study had previously unidentified silent atrial fibrillation (AF). Another 2% had been diagnosed with AF but were not on oral anticoagulation despite their substantial stroke risk, Dr. Emma Svennberg reported at the annual congress of the European Society of Cardiology.
The idea behind the screening program was to screen people at an age when the prevalence of AF is very high and yet there is sufficient remaining life expectancy that they could expect to gain substantial benefit from oral anticoagulation therapy for protection against cardioembolic stroke, explained Dr. Svennberg, a cardiologist at the Karolinska Institute, Stockholm.
The STROKESTOP study tested a novel mass screening strategy in which more than 6,000 Swedish 75- and 76-year-olds without known AF were issued a hand-held ECG device. Twice a day for 2 weeks they placed both thumbs on the device for 30 seconds to generate an ECG, which was transmitted to a central database. Compliance in this older age group was extremely high, although 1% of the ECGs were uninterpretable due to tremor, Parkinson’s disease, or other age-related physical infirmities.
Other mass screening programs have relied upon a single screening ECG. That’s a reliable means of detecting individuals with permanent AF, but very much hit-or-miss in those with paroxysmal AF, she noted.
Oral anticoagulant therapy was offered to all STROKESTOP participants found to have untreated AF. More than 90% of them accepted it. They are being prospectively followed for 5 years in the second phase of the STROKESTOP study, which will compare rates of ischemic stroke, major bleeding, mortality, and dementia in that group to a control group of 14,381 Swedish 75- and 76-year-olds – in the same two regions of the country – who were not invited to the screening program.
The prevalence of known AF in subjects invited to the screening program was 8.9%.
Individuals with AF have a fivefold increased risk of ischemic stroke, regardless of whether their atrial arrhythmia is silent or symptomatic. Oral anticoagulation therapy cuts that risk by close to 70%. Among patients with AF discharged from the hospital after a stroke, 70% die or become disabled. Yet undertreatment is common – in Sweden, as elsewhere, fewer than half of AF patients whose risk scores warrant anticoagulation therapy are on it, according to Dr. Svennberg.
Dr. Robert Hatala, session cochair, said one of the important messages of STROKESTOP is that you can’t find silent AF if you don’t look for it.
That’s why current ESC practice guidelines recommend that all patients aged 65 or older have their pulse taken at every physician visit, with an ECG to be ordered when any irregularities are detected, added Dr. Hatala, head of cardiology and director of the arrhythmia and pacing center at Slovak Medical University, Bratislava, Slovakia.
Dr. Svennberg said she and her coinvestigators are in the midst of a randomized trial comparing this low-tech screening method to the approach used in STROKESTOP involving 2 weeks of intermittent ECGs using a hand-held loaner device.
Dr. Hans-Christoph Diener, ESC spokesperson, predicted in an interview that physicians can soon expect to see a huge influx of patients with newly discovered silent AF. The impetus will be the much-anticipated introduction of the Apple iWatch, which he said could happen later this year.
"The detection of silent atrial fibrillation is going to change very quickly. This watch will have a device in it which measures pulse, and it has software which detects atrial fibrillation. More features are available for people with various diseases: It can measure oxygen saturation, pulse, blood pressure. They’ve even developed a chip to measure glucose transdermally. It’s amazing," said Dr. Diener, professor and chair of the department of neurology at the University of Duisburg-Essen, Germany.
The STROKESTOP study is supported by the Heart and Lung Foundation of Sweden and government research funds. Dr. Svennberg reported that she has received lecture fees from Merck Sharp & Dohme.
AT THE ESC CONGRESS 2013
Major finding: Five percent of 75- and 76-year-olds in Sweden have untreated atrial fibrillation. Three percent have silent AF and another 2% have been diagnosed with AF but are not on anticoagulant therapy.
Data source: The STROKESTOP study of nearly 29,000 75- and 76-year-old residents of two Swedish counties, one urban, one rural.
Disclosures: The ongoing study is supported by the Heart and Lung Foundation of Sweden and government research funds. Dr. Svennberg reported that she has received lecture fees from Merck Sharp & Dohme.
Pearls in clinical diagnosis of pertussis
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
Pearls in clinical diagnosis of pertussis
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
EXPERT ANALYSIS FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
A call for new pertussis vaccines
VAIL, COLO. – Development of a better pertussis vaccine has emerged as a pressing need in light of persuasive evidence of waning immunity to current vaccines.
But can it be done? Can a new vaccine be created that provides enhanced efficacy and longer-lasting immunity without increasing reactogenicity?
"I think it can be done," Dr. Myron J. Levin said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"The question you might ask is, can we do a better job than nature? After all, pertussis infection doesn’t cause lasting immunity, so could we do a better job than nature? I think so. And here’s an example: We know that antibody is protective against HPV [human papillomavirus], and that the HPV vaccine gives a much stronger antibody response, often by 100-fold, than does natural infection with HPV," observed Dr. Levin, who led development of the shingles vaccine.
Convincing evidence of waning immunity to the current five-dose diphtheria, tetanus, and acellular pertussis (DTaP) vaccine series completed before age 7 years in the United States comes from research conducted at the Kaiser Permanente Vaccine Study Center in Oakland, Calif. The investigators analyzed the large California pertussis outbreak of 2010. Their case-control study showed that the protection provided by the DTaP vaccine was short-lived, such that each year after completing the fifth dose the likelihood of developing PCR-proven pertussis climbed by an average of 42% (N. Engl. J. Med. 2012;367:1012-9).
Thus, they calculated that if the initial effectiveness of DTaP was 90%, it would drop to a mere 42% 5 years after the fifth and final dose. In addition, there is a strong argument that the original estimate of the vaccine’s efficacy of about 85% was inflated due to the case definition used. The efficacy rate after 5 years becomes disturbingly low if the actual efficacy rate was roughly 7%, as many now believe, noted Dr. Levin, professor of pediatrics and medicine at the University of Colorado, Denver.
The whole-cell pertussis vaccines were clearly more effective than the DTaP, which began replacing them in the early 1990s. In another Kaiser Permanente study that zeroed in on the transition period between the use of whole-cell pertussis vaccine and DTaP, patients who contracted PCR-confirmed pertussis did so an average of 14.7 years after receiving their last dose in the series if they received one or more doses of whole-cell vaccine compared with a mere 5.6 years after the last dose if they got the all-DTaP series.
The national switch to DTaP was carried out because the vaccine has fewer adverse effects than does the whole-cell pertussis vaccine. Components of whole-cell vaccines, including lipopolysaccharides and internal proteins, were important in signaling the innate immune system to shape a more robust response. Unfortunately, these whole-cell vaccine components also trigger unwanted reactogenic local and systemic effects.
"Whenever a vaccine is cleaned up to decrease side effects, there is a great likelihood that the efficacy will be affected. This is almost certainly the case with acellular pertussis, and probably with the split-product flu vaccines as well. I would say we need to learn why the whole-cell pertussis vaccine was so effective – which component was most important in directing the immune response – and see if we can separate that from the side effects. This is a research issue," said Dr. Levin.
Possible ways to create a better DTaP vaccine might involve incorporating a detoxified lipopolysaccharide or adding additional adjuvants to increase the innate immune response to the pertussis antigen.
Vaccine development takes time. In the interim, the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has taken steps to deal with the nation’s worsening pertussis epidemic outbreak. Because the herd protection effect is probably low, a "cocooning" strategy has been adopted. This involves targeted immunization of pregnant women and all close contacts of their babies during the first year of life, when pertussis has its highest morbidity and mortality. In addition, an adolescent dose of pertussis vaccine has been introduced. And consideration is being given to the possibility of universal once-per-decade booster doses in adults, but this has thus far been rejected as not being cost-effective.
Dr. Levin reported receiving honoraria from Novartis and GlaxoSmithKline and royalties as well as honoraria from Merck, Sharpe, and Dohme.
VAIL, COLO. – Development of a better pertussis vaccine has emerged as a pressing need in light of persuasive evidence of waning immunity to current vaccines.
But can it be done? Can a new vaccine be created that provides enhanced efficacy and longer-lasting immunity without increasing reactogenicity?
"I think it can be done," Dr. Myron J. Levin said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"The question you might ask is, can we do a better job than nature? After all, pertussis infection doesn’t cause lasting immunity, so could we do a better job than nature? I think so. And here’s an example: We know that antibody is protective against HPV [human papillomavirus], and that the HPV vaccine gives a much stronger antibody response, often by 100-fold, than does natural infection with HPV," observed Dr. Levin, who led development of the shingles vaccine.
Convincing evidence of waning immunity to the current five-dose diphtheria, tetanus, and acellular pertussis (DTaP) vaccine series completed before age 7 years in the United States comes from research conducted at the Kaiser Permanente Vaccine Study Center in Oakland, Calif. The investigators analyzed the large California pertussis outbreak of 2010. Their case-control study showed that the protection provided by the DTaP vaccine was short-lived, such that each year after completing the fifth dose the likelihood of developing PCR-proven pertussis climbed by an average of 42% (N. Engl. J. Med. 2012;367:1012-9).
Thus, they calculated that if the initial effectiveness of DTaP was 90%, it would drop to a mere 42% 5 years after the fifth and final dose. In addition, there is a strong argument that the original estimate of the vaccine’s efficacy of about 85% was inflated due to the case definition used. The efficacy rate after 5 years becomes disturbingly low if the actual efficacy rate was roughly 7%, as many now believe, noted Dr. Levin, professor of pediatrics and medicine at the University of Colorado, Denver.
The whole-cell pertussis vaccines were clearly more effective than the DTaP, which began replacing them in the early 1990s. In another Kaiser Permanente study that zeroed in on the transition period between the use of whole-cell pertussis vaccine and DTaP, patients who contracted PCR-confirmed pertussis did so an average of 14.7 years after receiving their last dose in the series if they received one or more doses of whole-cell vaccine compared with a mere 5.6 years after the last dose if they got the all-DTaP series.
The national switch to DTaP was carried out because the vaccine has fewer adverse effects than does the whole-cell pertussis vaccine. Components of whole-cell vaccines, including lipopolysaccharides and internal proteins, were important in signaling the innate immune system to shape a more robust response. Unfortunately, these whole-cell vaccine components also trigger unwanted reactogenic local and systemic effects.
"Whenever a vaccine is cleaned up to decrease side effects, there is a great likelihood that the efficacy will be affected. This is almost certainly the case with acellular pertussis, and probably with the split-product flu vaccines as well. I would say we need to learn why the whole-cell pertussis vaccine was so effective – which component was most important in directing the immune response – and see if we can separate that from the side effects. This is a research issue," said Dr. Levin.
Possible ways to create a better DTaP vaccine might involve incorporating a detoxified lipopolysaccharide or adding additional adjuvants to increase the innate immune response to the pertussis antigen.
Vaccine development takes time. In the interim, the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has taken steps to deal with the nation’s worsening pertussis epidemic outbreak. Because the herd protection effect is probably low, a "cocooning" strategy has been adopted. This involves targeted immunization of pregnant women and all close contacts of their babies during the first year of life, when pertussis has its highest morbidity and mortality. In addition, an adolescent dose of pertussis vaccine has been introduced. And consideration is being given to the possibility of universal once-per-decade booster doses in adults, but this has thus far been rejected as not being cost-effective.
Dr. Levin reported receiving honoraria from Novartis and GlaxoSmithKline and royalties as well as honoraria from Merck, Sharpe, and Dohme.
VAIL, COLO. – Development of a better pertussis vaccine has emerged as a pressing need in light of persuasive evidence of waning immunity to current vaccines.
But can it be done? Can a new vaccine be created that provides enhanced efficacy and longer-lasting immunity without increasing reactogenicity?
"I think it can be done," Dr. Myron J. Levin said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
"The question you might ask is, can we do a better job than nature? After all, pertussis infection doesn’t cause lasting immunity, so could we do a better job than nature? I think so. And here’s an example: We know that antibody is protective against HPV [human papillomavirus], and that the HPV vaccine gives a much stronger antibody response, often by 100-fold, than does natural infection with HPV," observed Dr. Levin, who led development of the shingles vaccine.
Convincing evidence of waning immunity to the current five-dose diphtheria, tetanus, and acellular pertussis (DTaP) vaccine series completed before age 7 years in the United States comes from research conducted at the Kaiser Permanente Vaccine Study Center in Oakland, Calif. The investigators analyzed the large California pertussis outbreak of 2010. Their case-control study showed that the protection provided by the DTaP vaccine was short-lived, such that each year after completing the fifth dose the likelihood of developing PCR-proven pertussis climbed by an average of 42% (N. Engl. J. Med. 2012;367:1012-9).
Thus, they calculated that if the initial effectiveness of DTaP was 90%, it would drop to a mere 42% 5 years after the fifth and final dose. In addition, there is a strong argument that the original estimate of the vaccine’s efficacy of about 85% was inflated due to the case definition used. The efficacy rate after 5 years becomes disturbingly low if the actual efficacy rate was roughly 7%, as many now believe, noted Dr. Levin, professor of pediatrics and medicine at the University of Colorado, Denver.
The whole-cell pertussis vaccines were clearly more effective than the DTaP, which began replacing them in the early 1990s. In another Kaiser Permanente study that zeroed in on the transition period between the use of whole-cell pertussis vaccine and DTaP, patients who contracted PCR-confirmed pertussis did so an average of 14.7 years after receiving their last dose in the series if they received one or more doses of whole-cell vaccine compared with a mere 5.6 years after the last dose if they got the all-DTaP series.
The national switch to DTaP was carried out because the vaccine has fewer adverse effects than does the whole-cell pertussis vaccine. Components of whole-cell vaccines, including lipopolysaccharides and internal proteins, were important in signaling the innate immune system to shape a more robust response. Unfortunately, these whole-cell vaccine components also trigger unwanted reactogenic local and systemic effects.
"Whenever a vaccine is cleaned up to decrease side effects, there is a great likelihood that the efficacy will be affected. This is almost certainly the case with acellular pertussis, and probably with the split-product flu vaccines as well. I would say we need to learn why the whole-cell pertussis vaccine was so effective – which component was most important in directing the immune response – and see if we can separate that from the side effects. This is a research issue," said Dr. Levin.
Possible ways to create a better DTaP vaccine might involve incorporating a detoxified lipopolysaccharide or adding additional adjuvants to increase the innate immune response to the pertussis antigen.
Vaccine development takes time. In the interim, the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention has taken steps to deal with the nation’s worsening pertussis epidemic outbreak. Because the herd protection effect is probably low, a "cocooning" strategy has been adopted. This involves targeted immunization of pregnant women and all close contacts of their babies during the first year of life, when pertussis has its highest morbidity and mortality. In addition, an adolescent dose of pertussis vaccine has been introduced. And consideration is being given to the possibility of universal once-per-decade booster doses in adults, but this has thus far been rejected as not being cost-effective.
Dr. Levin reported receiving honoraria from Novartis and GlaxoSmithKline and royalties as well as honoraria from Merck, Sharpe, and Dohme.
EXPERT ANALYSIS FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Why chemoprophylaxis is needed after pertussis exposure
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
'But I'm vaccinated!': Why chemoprophylaxis is needed after pertussis exposure
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
VAIL, COLO. – It’s a question hospital infection control officers field from physicians and other health care personnel every time a pertussis exposure occurs: "I’ve been vaccinated, so why do I have to get a course of azithromycin for postexposure prophylaxis?"
The Advisory Committee on Immunization Practices (ACIP) based its updated recommendation for antibiotic postexposure prophylaxis on the findings of a randomized trial known as the Vanderbilt Study. The results, while nondefinitive, suggested that a policy of watchful waiting with daily symptom monitoring for 21 days post exposure may not be as effective as azithromycin postexposure prophylaxis in preventing pertussis infection, Dr. Ann-Christine Nyquist explained at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The ACIP recommendation is for antibiotic prophylaxis for all pertussis-exposed health care workers likely to secondarily expose high-risk patients, such as neonates and pregnant women. Other vaccinated health care workers could receive either postexposure prophylaxis or 21 days of symptom monitoring with prompt antimicrobial therapy to be started should pertussis symptoms arise.
For Dr. Nyquist, the issue is a no-brainer. The vaccine is not 100% protective, the duration of protection is uncertain, and the adverse impact of a pertussis outbreak in a health care facility is enormous.
"When I put on my hospital epidemiologist hat and I think about pertussis in my hospital, it scares me to death. I would give everyone I was concerned about 5 days of azithromycin at $60 a pop," said Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
The children’s hospital affiliated with Vanderbilt University, Nashville, Tenn., has a universal tetanus-diphtheria-acellular pertussis (Tdap) vaccine immunization policy for all health care personnel. So it was an ideal location for a randomized comparison of two strategies to prevent infection following pertussis exposure in vaccinated physicians, nurses, and other health care personnel.
Following a pertussis exposure, health care personnel were randomized to 5 days of azithromycin or 21 days of watchful waiting. A bona fide exposure typically involved face-to-face exposure within a few feet when the health care provider wasn’t wearing a mask, or ungloved contact with a patient’s secretions.
Although 1,091 health care personnel enrolled in the trial, during a 30-month period only 86 subjects were randomized, limiting the statistical power of the findings. The key result: Only 1 of 42 patients who received postexposure prophylaxis met the prespecified definition of pertussis, compared with 6 of 44 in the watchful waiting group.
However, pertussis infection was defined quite strictly as a positive culture or PCR, a twofold increase in anti–pertussis toxin titer, or a single anti–pertussis toxin titer of at least 94 enzyme-linked immunosorbent assay units per milliliter. In fact, not a single study participant developed symptomatic pertussis, and the investigators concluded that "it is likely that none of the health care personnel who met the predefined serologic or PCR for infection were truly infected with pertussis" (Clin. Infect. Dis. 2012;54:938-45).
Dr. Nyquist noted that the Centers for Disease Control and Prevention (CDC) has identified health care workers as being at the epicenter of numerous pertussis outbreaks in hospitals. Health care personnel have regular contact with infected patients, and as adults they have waning immunity. The cost per hospital outbreak was calculated by the CDC at $44,000-$75,000.
"But that figure doesn’t include the human pain and suffering, which I would multiply maybe times five," she added.
ACIP recommends that all health care personnel with direct patient contact in hospitals or ambulatory settings receive a single dose of Tdap. In addition, at its June meeting ACIP directed the Pertussis Vaccines Work Group to explore the possibility of giving a booster dose of Tdap to health care workers in order to beef up their protection.
Dr. Nyquist reported having no relevant financial relationships with any commercial interests.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Novel risk factors for serious RSV identified
VAIL, COLO. – Children with congenital malformations or a variety of chronic diseases are at a previously undescribed, sharply increased risk for serious respiratory syncytial virus infection, according to a landmark Danish study.
Among the newly identified risk factors for hospitalization for RSV infection are neuromuscular disease, interstitial lung disease, liver disease, congenital malformations, liver disease, and congenital immunodeficiencies, Dr. Eric A.F. Simões said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
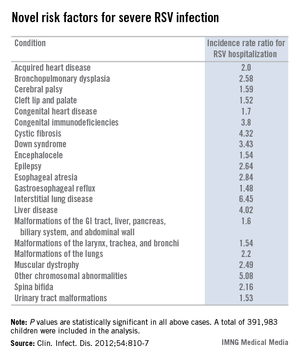
"When you are fighting with an insurance company over whether treatment with palivizumab is appropriate, you can show them this study. It’s the only one of its kind," said Dr. Simões, a coauthor of the Danish study and professor of pediatrics at the University of Colorado, Denver.
This was a population-based cohort study including 391,983 children born in Denmark during 1997-2003. Dr. Simões and his coinvestigators utilized the comprehensive Danish National Patient Registry to determine that 2.7% of the children carried a diagnosis for one or more chronic diseases, a broad heading which included congenital malformations, chromosomal abnormalities, and acquired chronic disorders.
During their first 23 months of life, 2.8% of the study population was hospitalized for an RSV infection. Of those hospitalized children, 8.8% had at least one diagnosis of chronic disease. In a multivariate analysis adjusted for maternal smoking, prematurity, hospitalization within the last 30 days, and other potential confounding factors, the incidence rate ratio for RSV hospitalization in children with any congenital chronic condition was 2.18. For children with any acquired chronic condition, it was 2.25. (See chart.)
Dr. Simoes and his colleagues put forth biologically plausible mechanistic explanations for the increased risks of severe RSV in many of the newly identified at-risk subgroups. For example, they argued that children with cleft lip and palate are known to have a high incidence of middle ear disease, and it’s possible that difficulties in swallowing could result in aspiration of infected nasal secretions, with resultant lower respiratory tract RSV infection.
Children with various neuromuscular diseases were found to be at increased risk for RSV hospitalization. These included children with epilepsy, cerebral palsy, muscular dystrophy, and spina bifida or other congenital malformations of the nervous system. Moreover, children with neuromuscular disease also had significantly longer-than-average RSV hospitalizations. It’s possible that the increased RSV morbidity seen in these patients was due at least in part to diminished vital capacity secondary to muscular dysfunction, coupled with disrupted clearance of respiratory secretions, the investigators speculated.
The finding of an elevated risk of RSV hospitalization among children with malformations of the urinary system might be explained as follows: Lower urinary tract obstruction results in oligohydramnios, in turn resulting in pulmonary hypoplasia.
The 4.32-fold increased risk of severe RSV infection documented in this study in children with cystic fibrosis may be related to the reduced levels of surfactant proteins A and D, which characterize this disease. Those proteins are key components of the pulmonary innate immune system, Dr. Simões and his coworkers noted (Clin. Infect. Dis. 2012;54:810-7).
In contrast, the finding of a fourfold increased risk of RSV hospitalization in children with liver disease was unexpected, and the investigators had no explanation for it.
The Danish study was supported by Abbott Laboratories. Dr. Simões has received research grants from and served as a consultant to Abbott and half a dozen other companies, as well as UNICEF, the Bill & Melinda Gates Foundation, and the World Health Organization.
VAIL, COLO. – Children with congenital malformations or a variety of chronic diseases are at a previously undescribed, sharply increased risk for serious respiratory syncytial virus infection, according to a landmark Danish study.
Among the newly identified risk factors for hospitalization for RSV infection are neuromuscular disease, interstitial lung disease, liver disease, congenital malformations, liver disease, and congenital immunodeficiencies, Dr. Eric A.F. Simões said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.

"When you are fighting with an insurance company over whether treatment with palivizumab is appropriate, you can show them this study. It’s the only one of its kind," said Dr. Simões, a coauthor of the Danish study and professor of pediatrics at the University of Colorado, Denver.
This was a population-based cohort study including 391,983 children born in Denmark during 1997-2003. Dr. Simões and his coinvestigators utilized the comprehensive Danish National Patient Registry to determine that 2.7% of the children carried a diagnosis for one or more chronic diseases, a broad heading which included congenital malformations, chromosomal abnormalities, and acquired chronic disorders.
During their first 23 months of life, 2.8% of the study population was hospitalized for an RSV infection. Of those hospitalized children, 8.8% had at least one diagnosis of chronic disease. In a multivariate analysis adjusted for maternal smoking, prematurity, hospitalization within the last 30 days, and other potential confounding factors, the incidence rate ratio for RSV hospitalization in children with any congenital chronic condition was 2.18. For children with any acquired chronic condition, it was 2.25. (See chart.)
Dr. Simoes and his colleagues put forth biologically plausible mechanistic explanations for the increased risks of severe RSV in many of the newly identified at-risk subgroups. For example, they argued that children with cleft lip and palate are known to have a high incidence of middle ear disease, and it’s possible that difficulties in swallowing could result in aspiration of infected nasal secretions, with resultant lower respiratory tract RSV infection.
Children with various neuromuscular diseases were found to be at increased risk for RSV hospitalization. These included children with epilepsy, cerebral palsy, muscular dystrophy, and spina bifida or other congenital malformations of the nervous system. Moreover, children with neuromuscular disease also had significantly longer-than-average RSV hospitalizations. It’s possible that the increased RSV morbidity seen in these patients was due at least in part to diminished vital capacity secondary to muscular dysfunction, coupled with disrupted clearance of respiratory secretions, the investigators speculated.
The finding of an elevated risk of RSV hospitalization among children with malformations of the urinary system might be explained as follows: Lower urinary tract obstruction results in oligohydramnios, in turn resulting in pulmonary hypoplasia.
The 4.32-fold increased risk of severe RSV infection documented in this study in children with cystic fibrosis may be related to the reduced levels of surfactant proteins A and D, which characterize this disease. Those proteins are key components of the pulmonary innate immune system, Dr. Simões and his coworkers noted (Clin. Infect. Dis. 2012;54:810-7).
In contrast, the finding of a fourfold increased risk of RSV hospitalization in children with liver disease was unexpected, and the investigators had no explanation for it.
The Danish study was supported by Abbott Laboratories. Dr. Simões has received research grants from and served as a consultant to Abbott and half a dozen other companies, as well as UNICEF, the Bill & Melinda Gates Foundation, and the World Health Organization.
VAIL, COLO. – Children with congenital malformations or a variety of chronic diseases are at a previously undescribed, sharply increased risk for serious respiratory syncytial virus infection, according to a landmark Danish study.
Among the newly identified risk factors for hospitalization for RSV infection are neuromuscular disease, interstitial lung disease, liver disease, congenital malformations, liver disease, and congenital immunodeficiencies, Dr. Eric A.F. Simões said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.

"When you are fighting with an insurance company over whether treatment with palivizumab is appropriate, you can show them this study. It’s the only one of its kind," said Dr. Simões, a coauthor of the Danish study and professor of pediatrics at the University of Colorado, Denver.
This was a population-based cohort study including 391,983 children born in Denmark during 1997-2003. Dr. Simões and his coinvestigators utilized the comprehensive Danish National Patient Registry to determine that 2.7% of the children carried a diagnosis for one or more chronic diseases, a broad heading which included congenital malformations, chromosomal abnormalities, and acquired chronic disorders.
During their first 23 months of life, 2.8% of the study population was hospitalized for an RSV infection. Of those hospitalized children, 8.8% had at least one diagnosis of chronic disease. In a multivariate analysis adjusted for maternal smoking, prematurity, hospitalization within the last 30 days, and other potential confounding factors, the incidence rate ratio for RSV hospitalization in children with any congenital chronic condition was 2.18. For children with any acquired chronic condition, it was 2.25. (See chart.)
Dr. Simoes and his colleagues put forth biologically plausible mechanistic explanations for the increased risks of severe RSV in many of the newly identified at-risk subgroups. For example, they argued that children with cleft lip and palate are known to have a high incidence of middle ear disease, and it’s possible that difficulties in swallowing could result in aspiration of infected nasal secretions, with resultant lower respiratory tract RSV infection.
Children with various neuromuscular diseases were found to be at increased risk for RSV hospitalization. These included children with epilepsy, cerebral palsy, muscular dystrophy, and spina bifida or other congenital malformations of the nervous system. Moreover, children with neuromuscular disease also had significantly longer-than-average RSV hospitalizations. It’s possible that the increased RSV morbidity seen in these patients was due at least in part to diminished vital capacity secondary to muscular dysfunction, coupled with disrupted clearance of respiratory secretions, the investigators speculated.
The finding of an elevated risk of RSV hospitalization among children with malformations of the urinary system might be explained as follows: Lower urinary tract obstruction results in oligohydramnios, in turn resulting in pulmonary hypoplasia.
The 4.32-fold increased risk of severe RSV infection documented in this study in children with cystic fibrosis may be related to the reduced levels of surfactant proteins A and D, which characterize this disease. Those proteins are key components of the pulmonary innate immune system, Dr. Simões and his coworkers noted (Clin. Infect. Dis. 2012;54:810-7).
In contrast, the finding of a fourfold increased risk of RSV hospitalization in children with liver disease was unexpected, and the investigators had no explanation for it.
The Danish study was supported by Abbott Laboratories. Dr. Simões has received research grants from and served as a consultant to Abbott and half a dozen other companies, as well as UNICEF, the Bill & Melinda Gates Foundation, and the World Health Organization.
AT THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Major finding: Children with congenital malformations, neuromuscular disease, interstitial lung disease, a variety of chromosomal abnormalities, acquired heart disease, and other chronic conditions are newly recognized as being at significantly increased risk for severe respiratory syncytial virus infection.
Data source: This was a Danish national population-based cohort study involving nearly 400,000 children born in Denmark during 1997-2003, of whom 2.8% were hospitalized for RSV before 24 months of age.
Disclosures: The Danish study was supported by Abbott Laboratories. Dr. Simões has received research grants from and served as a consultant to Abbott and half a dozen other companies, as well as UNICEF, the Bill & Melinda Gates Foundation, and the World Health Organization.