User login
Diabetics' stroke risk post MI has plummeted
AMSTERDAM – The risk of ischemic stroke following an acute myocardial infarction in diabetes patients dropped markedly during a recent 10-year period, according to a nationwide Swedish study.
Indeed, the reduction in ischemic stroke risk during the first year after an MI was significantly larger in diabetic than in nondiabetic patients over the course of a decade, Stina Jakobsson said at the annual congress of the European Society of Cardiology.
"We believe that the larger risk reduction seen in the diabetic patients may indicate that they have gained more from the increased use of evidence-based secondary preventive treatment," added Ms. Jakobsson, a medical student at Umea (Sweden) University.
She presented an analysis of all 173,233 patients discharged from Swedish coronary care units after an acute MI during 1998-2008. A total of 19% of them had a previous diagnosis of diabetes.
Among diabetes patients with an MI in 1998-2000, ischemic stroke occurred in 7.1% within 1 year after their coronary event. However, the 1-year ischemic stroke rate in such patients whose MI occurred in 2007-2008 dropped to 4.7%. This was a much more impressive improvement than occurred in the same time span among nondiabetic patients, where the ischemic stroke rate during the first year after an acute MI was 4.2% in 1998-2000, nudging downward to 3.7% in 2007-2008.
Ms. Jakobsson stressed that there is definitely room for improvement in the use of reperfusion therapy and secondary preventive medications among diabetes patients with an MI. Although the use of these key interventions increased over time in both diabetic and nondiabetic MI patients, rates still remained lower in the diabetic group in the most recent study years.
The 4.7% 1-year incidence of ischemic stroke among Swedish diabetes patients with an acute MI in 2007-2008 was significantly greater than the 3.7% rate among nondiabetic patients. Moreover, even among patients on optimized secondary prevention therapies, the ischemic stroke rate was higher in the diabetic group. That’s not surprising because they more often had a history of prior cardiovascular disease at the time of their acute MI.
"They were sicker to start with," Ms. Jakobsson observed.
The most powerful predictors of increased risk of ischemic stroke post MI included older age, atrial fibrillation, an ST-elevation MI, and prior ischemic stroke.
This study was supported by Swedish governmental research funds. Ms. Jakobsson reported having no financial conflicts of interest.
AMSTERDAM – The risk of ischemic stroke following an acute myocardial infarction in diabetes patients dropped markedly during a recent 10-year period, according to a nationwide Swedish study.
Indeed, the reduction in ischemic stroke risk during the first year after an MI was significantly larger in diabetic than in nondiabetic patients over the course of a decade, Stina Jakobsson said at the annual congress of the European Society of Cardiology.
"We believe that the larger risk reduction seen in the diabetic patients may indicate that they have gained more from the increased use of evidence-based secondary preventive treatment," added Ms. Jakobsson, a medical student at Umea (Sweden) University.
She presented an analysis of all 173,233 patients discharged from Swedish coronary care units after an acute MI during 1998-2008. A total of 19% of them had a previous diagnosis of diabetes.
Among diabetes patients with an MI in 1998-2000, ischemic stroke occurred in 7.1% within 1 year after their coronary event. However, the 1-year ischemic stroke rate in such patients whose MI occurred in 2007-2008 dropped to 4.7%. This was a much more impressive improvement than occurred in the same time span among nondiabetic patients, where the ischemic stroke rate during the first year after an acute MI was 4.2% in 1998-2000, nudging downward to 3.7% in 2007-2008.
Ms. Jakobsson stressed that there is definitely room for improvement in the use of reperfusion therapy and secondary preventive medications among diabetes patients with an MI. Although the use of these key interventions increased over time in both diabetic and nondiabetic MI patients, rates still remained lower in the diabetic group in the most recent study years.
The 4.7% 1-year incidence of ischemic stroke among Swedish diabetes patients with an acute MI in 2007-2008 was significantly greater than the 3.7% rate among nondiabetic patients. Moreover, even among patients on optimized secondary prevention therapies, the ischemic stroke rate was higher in the diabetic group. That’s not surprising because they more often had a history of prior cardiovascular disease at the time of their acute MI.
"They were sicker to start with," Ms. Jakobsson observed.
The most powerful predictors of increased risk of ischemic stroke post MI included older age, atrial fibrillation, an ST-elevation MI, and prior ischemic stroke.
This study was supported by Swedish governmental research funds. Ms. Jakobsson reported having no financial conflicts of interest.
AMSTERDAM – The risk of ischemic stroke following an acute myocardial infarction in diabetes patients dropped markedly during a recent 10-year period, according to a nationwide Swedish study.
Indeed, the reduction in ischemic stroke risk during the first year after an MI was significantly larger in diabetic than in nondiabetic patients over the course of a decade, Stina Jakobsson said at the annual congress of the European Society of Cardiology.
"We believe that the larger risk reduction seen in the diabetic patients may indicate that they have gained more from the increased use of evidence-based secondary preventive treatment," added Ms. Jakobsson, a medical student at Umea (Sweden) University.
She presented an analysis of all 173,233 patients discharged from Swedish coronary care units after an acute MI during 1998-2008. A total of 19% of them had a previous diagnosis of diabetes.
Among diabetes patients with an MI in 1998-2000, ischemic stroke occurred in 7.1% within 1 year after their coronary event. However, the 1-year ischemic stroke rate in such patients whose MI occurred in 2007-2008 dropped to 4.7%. This was a much more impressive improvement than occurred in the same time span among nondiabetic patients, where the ischemic stroke rate during the first year after an acute MI was 4.2% in 1998-2000, nudging downward to 3.7% in 2007-2008.
Ms. Jakobsson stressed that there is definitely room for improvement in the use of reperfusion therapy and secondary preventive medications among diabetes patients with an MI. Although the use of these key interventions increased over time in both diabetic and nondiabetic MI patients, rates still remained lower in the diabetic group in the most recent study years.
The 4.7% 1-year incidence of ischemic stroke among Swedish diabetes patients with an acute MI in 2007-2008 was significantly greater than the 3.7% rate among nondiabetic patients. Moreover, even among patients on optimized secondary prevention therapies, the ischemic stroke rate was higher in the diabetic group. That’s not surprising because they more often had a history of prior cardiovascular disease at the time of their acute MI.
"They were sicker to start with," Ms. Jakobsson observed.
The most powerful predictors of increased risk of ischemic stroke post MI included older age, atrial fibrillation, an ST-elevation MI, and prior ischemic stroke.
This study was supported by Swedish governmental research funds. Ms. Jakobsson reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: The rate of ischemic stroke during the first year after an acute MI improved from 7.1% among Swedish diabetic patients whose infarct occurred during 1998-2000 to 4.7% in those with an MI in 2007-2008.
Data source: A retrospective study of 173,233 patients discharged from Swedish coronary care units after an acute MI during 1998-2008. They were followed through national registries in order to learn their incidence of ischemic stroke during the first year post MI.
Disclosures: The presenter reported having no financial conflicts of interest.
How to foil post-CABG aspirin resistance
AMSTERDAM – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AMSTERDAM – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AMSTERDAM – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: The median serum thromboxane level on the morning of post CABG day 4 was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
Data source: A randomized clinical trial in which 110 patients who underwent on-pump CABG surgery were randomized on postoperative day 1 to aspirin at either 81 mg four times daily, 325 mg once daily, or 81 mg once daily.
Disclosures: The study presenter reported having no financial conflicts.
High resting heart rate portends cognitive decline
AMSTERDAM – A high resting heart rate proved to be a strong and independent predictor of cognitive decline within the next 4 years in a study of nearly 28,000 patients at high cardiovascular risk.
The clinical implications of this finding, however, remain unclear, according to Dr. Darryl P. Leong.
"What this study cannot answer, and which must be answered, is whether resting heart rate is just a marker of the risk of cognitive decline or whether it exists in the causal pathway. Further research is needed to determine if resting heart rate represents a therapeutic target to prevent cognitive decline. I think the only way to test this is with some intervention to reduce heart rate – whether using a beta-blocker or a medication such as ivabradine – to see whether or not it influences the incidence of cognitive decline," he said in presenting the study findings at the annual congress of the European Society of Cardiology.
"Cognitive dysfunction and decline, I think, are going to be a major scourge over the next decades," he added in explaining the study rationale. "We have an increasingly aged population, we have increasingly better treatment and survival of cardiovascular disease, and as a result more and more people are going to be living long enough to experience the misfortune of having cognitive impairment," said Dr. Leong, a postdoctoral fellow at the population health research institute at McMaster University, Hamilton, Ont.
He presented a post hoc analysis of two major randomized clinical trials – ONTARGET (the Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial) and TRANSCEND (Telmisartan Randomised Assessment Study in Angiotension-Converting Enzyme Inhibitor–Intolerant Subjects With Cardiovascular Disease). In these two trials, patients with diabetes or vascular disease were assigned to ramipril, telmisartan, both, or placebo. The primary outcomes have already been published. Dr. Leong and his coinvestigators at McMaster used the database for the two trials, which contains information on baseline resting heart rate and subsequent cognitive decline in 27,660 participants.
The quartiles of baseline resting heart rate were less than 60 bpm, 60-66, 67-74, and 75 or more bpm. Cognitive decline was defined as at least a 3-point drop from baseline on the Mini-Mental State Exam at a median 4 years of follow-up.
During the follow-up period, 17% of the 27,660 patients exhibited cognitive decline. The investigators noted a stepwise relationship between baseline resting heart rate and cognitive deterioration: Those in the top two quartiles – that is, patients with a resting heart rate of 67 bpm or more – had a 16% greater risk than did those in the lower two quartiles. This was the case even after extensive adjustment in a multivariate regression analysis controlled for baseline demographic variables, cardiovascular comorbidities, depressive symptoms, blood pressure, dietary habits, renal function, medications, lipid levels, left ventricular hypertrophy, alcohol consumption, and physical activity level.
Even after investigators controlled for baseline use of beta-blockers, calcium channel blockers, and digoxin, resting heart rate remained predictive of subsequent cognitive decline, according to Dr. Leong.
He reported having no relevant financial conflicts.
AMSTERDAM – A high resting heart rate proved to be a strong and independent predictor of cognitive decline within the next 4 years in a study of nearly 28,000 patients at high cardiovascular risk.
The clinical implications of this finding, however, remain unclear, according to Dr. Darryl P. Leong.
"What this study cannot answer, and which must be answered, is whether resting heart rate is just a marker of the risk of cognitive decline or whether it exists in the causal pathway. Further research is needed to determine if resting heart rate represents a therapeutic target to prevent cognitive decline. I think the only way to test this is with some intervention to reduce heart rate – whether using a beta-blocker or a medication such as ivabradine – to see whether or not it influences the incidence of cognitive decline," he said in presenting the study findings at the annual congress of the European Society of Cardiology.
"Cognitive dysfunction and decline, I think, are going to be a major scourge over the next decades," he added in explaining the study rationale. "We have an increasingly aged population, we have increasingly better treatment and survival of cardiovascular disease, and as a result more and more people are going to be living long enough to experience the misfortune of having cognitive impairment," said Dr. Leong, a postdoctoral fellow at the population health research institute at McMaster University, Hamilton, Ont.
He presented a post hoc analysis of two major randomized clinical trials – ONTARGET (the Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial) and TRANSCEND (Telmisartan Randomised Assessment Study in Angiotension-Converting Enzyme Inhibitor–Intolerant Subjects With Cardiovascular Disease). In these two trials, patients with diabetes or vascular disease were assigned to ramipril, telmisartan, both, or placebo. The primary outcomes have already been published. Dr. Leong and his coinvestigators at McMaster used the database for the two trials, which contains information on baseline resting heart rate and subsequent cognitive decline in 27,660 participants.
The quartiles of baseline resting heart rate were less than 60 bpm, 60-66, 67-74, and 75 or more bpm. Cognitive decline was defined as at least a 3-point drop from baseline on the Mini-Mental State Exam at a median 4 years of follow-up.
During the follow-up period, 17% of the 27,660 patients exhibited cognitive decline. The investigators noted a stepwise relationship between baseline resting heart rate and cognitive deterioration: Those in the top two quartiles – that is, patients with a resting heart rate of 67 bpm or more – had a 16% greater risk than did those in the lower two quartiles. This was the case even after extensive adjustment in a multivariate regression analysis controlled for baseline demographic variables, cardiovascular comorbidities, depressive symptoms, blood pressure, dietary habits, renal function, medications, lipid levels, left ventricular hypertrophy, alcohol consumption, and physical activity level.
Even after investigators controlled for baseline use of beta-blockers, calcium channel blockers, and digoxin, resting heart rate remained predictive of subsequent cognitive decline, according to Dr. Leong.
He reported having no relevant financial conflicts.
AMSTERDAM – A high resting heart rate proved to be a strong and independent predictor of cognitive decline within the next 4 years in a study of nearly 28,000 patients at high cardiovascular risk.
The clinical implications of this finding, however, remain unclear, according to Dr. Darryl P. Leong.
"What this study cannot answer, and which must be answered, is whether resting heart rate is just a marker of the risk of cognitive decline or whether it exists in the causal pathway. Further research is needed to determine if resting heart rate represents a therapeutic target to prevent cognitive decline. I think the only way to test this is with some intervention to reduce heart rate – whether using a beta-blocker or a medication such as ivabradine – to see whether or not it influences the incidence of cognitive decline," he said in presenting the study findings at the annual congress of the European Society of Cardiology.
"Cognitive dysfunction and decline, I think, are going to be a major scourge over the next decades," he added in explaining the study rationale. "We have an increasingly aged population, we have increasingly better treatment and survival of cardiovascular disease, and as a result more and more people are going to be living long enough to experience the misfortune of having cognitive impairment," said Dr. Leong, a postdoctoral fellow at the population health research institute at McMaster University, Hamilton, Ont.
He presented a post hoc analysis of two major randomized clinical trials – ONTARGET (the Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial) and TRANSCEND (Telmisartan Randomised Assessment Study in Angiotension-Converting Enzyme Inhibitor–Intolerant Subjects With Cardiovascular Disease). In these two trials, patients with diabetes or vascular disease were assigned to ramipril, telmisartan, both, or placebo. The primary outcomes have already been published. Dr. Leong and his coinvestigators at McMaster used the database for the two trials, which contains information on baseline resting heart rate and subsequent cognitive decline in 27,660 participants.
The quartiles of baseline resting heart rate were less than 60 bpm, 60-66, 67-74, and 75 or more bpm. Cognitive decline was defined as at least a 3-point drop from baseline on the Mini-Mental State Exam at a median 4 years of follow-up.
During the follow-up period, 17% of the 27,660 patients exhibited cognitive decline. The investigators noted a stepwise relationship between baseline resting heart rate and cognitive deterioration: Those in the top two quartiles – that is, patients with a resting heart rate of 67 bpm or more – had a 16% greater risk than did those in the lower two quartiles. This was the case even after extensive adjustment in a multivariate regression analysis controlled for baseline demographic variables, cardiovascular comorbidities, depressive symptoms, blood pressure, dietary habits, renal function, medications, lipid levels, left ventricular hypertrophy, alcohol consumption, and physical activity level.
Even after investigators controlled for baseline use of beta-blockers, calcium channel blockers, and digoxin, resting heart rate remained predictive of subsequent cognitive decline, according to Dr. Leong.
He reported having no relevant financial conflicts.
AT THE ESC CONGRESS 2013
Major finding: Individuals at high cardiovascular risk who had a baseline resting heart rate of 67 bpm or more had a 16% greater risk of clinically meaningful cognitive decline during the next 4 years than did those with a resting heart rate below that threshold.
Data source: A post hoc secondary analysis of data on the nearly 28,000 participants in the randomized, double-blind ONTARGET and TRANSCEND clinical trials.
Disclosures: The post hoc analysis was funded by the population health research institute at McMaster University. The presenter reported having no relevant financial conflicts.
Prophylactic beta-blockers and noncardiac surgery: It's complicated!
AMSTERDAM – Results of a new Danish national study suggest the effects of prophylactic beta-blocker therapy in patients with ischemic heart disease undergoing noncardiac surgery are considerably more heterogeneous than portrayed in current pro-prophylaxis practice guidelines or, at the opposite extreme, in a recent highly critical meta-analysis.
"This is an extraordinarily confusing area at the moment," Dr. Charlotte Andersson observed in presenting the Danish national registry findings at the annual congress of the European Society of Cardiology.
She reported on 28,263 adults with ischemic heart disease who underwent noncardiac surgery during 2004-2009. Hip or knee replacements were the most common operations, accounting for roughly one-third of the total. Patients were followed for 30 days postoperatively for the composite endpoint of acute myocardial infarction, ischemic stroke, or cardiovascular death, as well as for 30-day all-cause mortality.
In short, the effects of prophylactic beta-blocker therapy depended upon the type of background ischemic heart disease a surgical patient had.
"Our data suggest a beneficial effect of beta-blockers among patients with heart failure, perhaps a beneficial effect as well among patients with an MI within the previous 2 years, but no beneficial effect among patients with a more distant MI, and perhaps even harm associated with beta-blocker therapy among patients with neither heart failure nor a history of MI," according to Dr. Andersson of the University of Copenhagen.
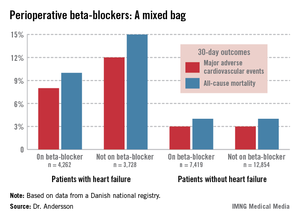
The study population included 7,990 patients with heart failure, 53% of whom were on beta-blockers when they underwent noncardiac surgery. Those on beta-blockers fared significantly better in terms of the study endpoints (see graphic).
In contrast, 30-day outcomes in the 37% of patients without heart failure were identical regardless of whether or not they were on beta-blockers at surgery.
In a multivariate analysis, the use of beta-blockers in noncardiac surgery patients with heart failure was associated with a 22% reduction in major adverse cardiovascular events (MACEs) and an 18% reduction in all-cause mortality compared with no use of beta-blockers, both of which were statistically significant advantages. The analysis was adjusted for patient demographics, acute versus elective surgery, chronic obstructive pulmonary disease, diabetes, atrial fibrillation, peripheral artery disease, cancer, anemia, smoking, alcohol consumption, cerebrovascular disease, and American Society of Anesthesiologists score.
Among the 1,664 patients with an MI within the past 2 years, being on a beta-blocker at the time of surgery was associated with an adjusted highly significant 46% reduction in MACE and a 20% decrease in all-cause mortality, compared with no use of beta-blockers.
For the 1,679 patients with an MI 2-5 years prior to surgery, being on a beta-blocker was associated with a 29% reduction in the risk of MACE and a 26% reduction in 30-day all-cause mortality.
Among the 5,018 patients with an MI more than 5 years earlier, the use of beta-blockers at surgery was associated with a 35% greater risk of MACE than in nonusers of beta-blockers as well as a 33% increase in all-cause mortality. These differences in adverse outcomes rates barely missed achieving statistical significance.
Perhaps the most striking study finding was that patients with no prior MI or heart failure who were on a beta-blocker at the time of noncardiac surgery had a 44% increased risk of 30-day MACE and a 30% higher all-cause mortality, compared with those not on a beta-blocker, with both differences being significant.
Session cochair Dr. Elmir Omerovic thanked Dr. Andersson for a presentation that "really adds important new information" and asked whether she had been surprised by the findings.
"Yes, I have to say I was surprised by the increased risk in patients without prior MI or heart failure, because the ESC [European Society of Cardiology] guidelines state as a class I recommendation that all patients with ischemic heart disease undergoing noncardiac surgery should be on a beta-blocker. Perhaps we should reevaluate beta-blockers in noncardiac surgery," Dr. Andersson replied.
Current American College of Cardiology/American Heart Association guidelines also endorse perioperative beta-blockade in patients with coronary artery disease undergoing vascular or intermediate-risk noncardiac surgery.
She noted that in drawing up the current guidelines, the ESC and ACC/AHA committees relied heavily on strongly positive randomized clinical trials whose validity has recently been called into question in a major research scandal. Indeed, the lead investigator in those studies, Dr. Don Poldermans – who also happened to be chairperson of the ESC guidelines-writing task force – has been dismissed from the faculty at Erasmus University in Rotterdam.
Dr. Omerovic, of Sahlgrenska University, Gothenburg, Sweden, asked for Dr. Andersson’s thoughts regarding a new meta-analysis by investigators at Imperial College London which excluded the suspect Dutch clinical trials. The investigators concluded that initiation of perioperative beta-blocker therapy was associated with a 27% increase in 30-day all-cause mortality, a 27% reduction in nonfatal MI, a 73% increase in stroke, and a 51% increase in hypotension.
"Patient safety being paramount, guidelines for perioperative beta-blocker initiation should be retracted without further delay," the meta-analysts argued (Heart 2013 July 31 [doi: 10.1136/heartjnl-2013-304262]).
"I read that meta-analysis with great interest," Dr. Andersson said. "I think there definitely is a heterogeneity in the effects of perioperative beta-blockers, and it depends on your baseline risk. Most of the studies in the meta-analysis included many patients at lower risk."
Dr. Andersson’s study was funded by the Danish Medical Research Foundation. She reported having no financial conflicts of interest.
Dr. Jun Chiong, FCCP, comments: There are similar studies published regarding preoperative beta-blockers. Most studies are from large databases and not randomized. The results are varied. Important factors such as severity of comorbid conditions prior to surgery (eg, uncontrolled diabetic, noncompliance), amount of blood loss, and surgical technique (not just the organ involved) are often not factored in. I applaud the authors for reporting a significant finding from such a large database. I hope this will encourage investigators to initiate a large, well-controlled, randomized study.
Dr. Jun Chiong, FCCP, comments: There are similar studies published regarding preoperative beta-blockers. Most studies are from large databases and not randomized. The results are varied. Important factors such as severity of comorbid conditions prior to surgery (eg, uncontrolled diabetic, noncompliance), amount of blood loss, and surgical technique (not just the organ involved) are often not factored in. I applaud the authors for reporting a significant finding from such a large database. I hope this will encourage investigators to initiate a large, well-controlled, randomized study.
Dr. Jun Chiong, FCCP, comments: There are similar studies published regarding preoperative beta-blockers. Most studies are from large databases and not randomized. The results are varied. Important factors such as severity of comorbid conditions prior to surgery (eg, uncontrolled diabetic, noncompliance), amount of blood loss, and surgical technique (not just the organ involved) are often not factored in. I applaud the authors for reporting a significant finding from such a large database. I hope this will encourage investigators to initiate a large, well-controlled, randomized study.
AMSTERDAM – Results of a new Danish national study suggest the effects of prophylactic beta-blocker therapy in patients with ischemic heart disease undergoing noncardiac surgery are considerably more heterogeneous than portrayed in current pro-prophylaxis practice guidelines or, at the opposite extreme, in a recent highly critical meta-analysis.
"This is an extraordinarily confusing area at the moment," Dr. Charlotte Andersson observed in presenting the Danish national registry findings at the annual congress of the European Society of Cardiology.

She reported on 28,263 adults with ischemic heart disease who underwent noncardiac surgery during 2004-2009. Hip or knee replacements were the most common operations, accounting for roughly one-third of the total. Patients were followed for 30 days postoperatively for the composite endpoint of acute myocardial infarction, ischemic stroke, or cardiovascular death, as well as for 30-day all-cause mortality.
In short, the effects of prophylactic beta-blocker therapy depended upon the type of background ischemic heart disease a surgical patient had.
"Our data suggest a beneficial effect of beta-blockers among patients with heart failure, perhaps a beneficial effect as well among patients with an MI within the previous 2 years, but no beneficial effect among patients with a more distant MI, and perhaps even harm associated with beta-blocker therapy among patients with neither heart failure nor a history of MI," according to Dr. Andersson of the University of Copenhagen.
The study population included 7,990 patients with heart failure, 53% of whom were on beta-blockers when they underwent noncardiac surgery. Those on beta-blockers fared significantly better in terms of the study endpoints (see graphic).
In contrast, 30-day outcomes in the 37% of patients without heart failure were identical regardless of whether or not they were on beta-blockers at surgery.
In a multivariate analysis, the use of beta-blockers in noncardiac surgery patients with heart failure was associated with a 22% reduction in major adverse cardiovascular events (MACEs) and an 18% reduction in all-cause mortality compared with no use of beta-blockers, both of which were statistically significant advantages. The analysis was adjusted for patient demographics, acute versus elective surgery, chronic obstructive pulmonary disease, diabetes, atrial fibrillation, peripheral artery disease, cancer, anemia, smoking, alcohol consumption, cerebrovascular disease, and American Society of Anesthesiologists score.
Among the 1,664 patients with an MI within the past 2 years, being on a beta-blocker at the time of surgery was associated with an adjusted highly significant 46% reduction in MACE and a 20% decrease in all-cause mortality, compared with no use of beta-blockers.
For the 1,679 patients with an MI 2-5 years prior to surgery, being on a beta-blocker was associated with a 29% reduction in the risk of MACE and a 26% reduction in 30-day all-cause mortality.
Among the 5,018 patients with an MI more than 5 years earlier, the use of beta-blockers at surgery was associated with a 35% greater risk of MACE than in nonusers of beta-blockers as well as a 33% increase in all-cause mortality. These differences in adverse outcomes rates barely missed achieving statistical significance.
Perhaps the most striking study finding was that patients with no prior MI or heart failure who were on a beta-blocker at the time of noncardiac surgery had a 44% increased risk of 30-day MACE and a 30% higher all-cause mortality, compared with those not on a beta-blocker, with both differences being significant.
Session cochair Dr. Elmir Omerovic thanked Dr. Andersson for a presentation that "really adds important new information" and asked whether she had been surprised by the findings.
"Yes, I have to say I was surprised by the increased risk in patients without prior MI or heart failure, because the ESC [European Society of Cardiology] guidelines state as a class I recommendation that all patients with ischemic heart disease undergoing noncardiac surgery should be on a beta-blocker. Perhaps we should reevaluate beta-blockers in noncardiac surgery," Dr. Andersson replied.
Current American College of Cardiology/American Heart Association guidelines also endorse perioperative beta-blockade in patients with coronary artery disease undergoing vascular or intermediate-risk noncardiac surgery.
She noted that in drawing up the current guidelines, the ESC and ACC/AHA committees relied heavily on strongly positive randomized clinical trials whose validity has recently been called into question in a major research scandal. Indeed, the lead investigator in those studies, Dr. Don Poldermans – who also happened to be chairperson of the ESC guidelines-writing task force – has been dismissed from the faculty at Erasmus University in Rotterdam.
Dr. Omerovic, of Sahlgrenska University, Gothenburg, Sweden, asked for Dr. Andersson’s thoughts regarding a new meta-analysis by investigators at Imperial College London which excluded the suspect Dutch clinical trials. The investigators concluded that initiation of perioperative beta-blocker therapy was associated with a 27% increase in 30-day all-cause mortality, a 27% reduction in nonfatal MI, a 73% increase in stroke, and a 51% increase in hypotension.
"Patient safety being paramount, guidelines for perioperative beta-blocker initiation should be retracted without further delay," the meta-analysts argued (Heart 2013 July 31 [doi: 10.1136/heartjnl-2013-304262]).
"I read that meta-analysis with great interest," Dr. Andersson said. "I think there definitely is a heterogeneity in the effects of perioperative beta-blockers, and it depends on your baseline risk. Most of the studies in the meta-analysis included many patients at lower risk."
Dr. Andersson’s study was funded by the Danish Medical Research Foundation. She reported having no financial conflicts of interest.
AMSTERDAM – Results of a new Danish national study suggest the effects of prophylactic beta-blocker therapy in patients with ischemic heart disease undergoing noncardiac surgery are considerably more heterogeneous than portrayed in current pro-prophylaxis practice guidelines or, at the opposite extreme, in a recent highly critical meta-analysis.
"This is an extraordinarily confusing area at the moment," Dr. Charlotte Andersson observed in presenting the Danish national registry findings at the annual congress of the European Society of Cardiology.

She reported on 28,263 adults with ischemic heart disease who underwent noncardiac surgery during 2004-2009. Hip or knee replacements were the most common operations, accounting for roughly one-third of the total. Patients were followed for 30 days postoperatively for the composite endpoint of acute myocardial infarction, ischemic stroke, or cardiovascular death, as well as for 30-day all-cause mortality.
In short, the effects of prophylactic beta-blocker therapy depended upon the type of background ischemic heart disease a surgical patient had.
"Our data suggest a beneficial effect of beta-blockers among patients with heart failure, perhaps a beneficial effect as well among patients with an MI within the previous 2 years, but no beneficial effect among patients with a more distant MI, and perhaps even harm associated with beta-blocker therapy among patients with neither heart failure nor a history of MI," according to Dr. Andersson of the University of Copenhagen.
The study population included 7,990 patients with heart failure, 53% of whom were on beta-blockers when they underwent noncardiac surgery. Those on beta-blockers fared significantly better in terms of the study endpoints (see graphic).
In contrast, 30-day outcomes in the 37% of patients without heart failure were identical regardless of whether or not they were on beta-blockers at surgery.
In a multivariate analysis, the use of beta-blockers in noncardiac surgery patients with heart failure was associated with a 22% reduction in major adverse cardiovascular events (MACEs) and an 18% reduction in all-cause mortality compared with no use of beta-blockers, both of which were statistically significant advantages. The analysis was adjusted for patient demographics, acute versus elective surgery, chronic obstructive pulmonary disease, diabetes, atrial fibrillation, peripheral artery disease, cancer, anemia, smoking, alcohol consumption, cerebrovascular disease, and American Society of Anesthesiologists score.
Among the 1,664 patients with an MI within the past 2 years, being on a beta-blocker at the time of surgery was associated with an adjusted highly significant 46% reduction in MACE and a 20% decrease in all-cause mortality, compared with no use of beta-blockers.
For the 1,679 patients with an MI 2-5 years prior to surgery, being on a beta-blocker was associated with a 29% reduction in the risk of MACE and a 26% reduction in 30-day all-cause mortality.
Among the 5,018 patients with an MI more than 5 years earlier, the use of beta-blockers at surgery was associated with a 35% greater risk of MACE than in nonusers of beta-blockers as well as a 33% increase in all-cause mortality. These differences in adverse outcomes rates barely missed achieving statistical significance.
Perhaps the most striking study finding was that patients with no prior MI or heart failure who were on a beta-blocker at the time of noncardiac surgery had a 44% increased risk of 30-day MACE and a 30% higher all-cause mortality, compared with those not on a beta-blocker, with both differences being significant.
Session cochair Dr. Elmir Omerovic thanked Dr. Andersson for a presentation that "really adds important new information" and asked whether she had been surprised by the findings.
"Yes, I have to say I was surprised by the increased risk in patients without prior MI or heart failure, because the ESC [European Society of Cardiology] guidelines state as a class I recommendation that all patients with ischemic heart disease undergoing noncardiac surgery should be on a beta-blocker. Perhaps we should reevaluate beta-blockers in noncardiac surgery," Dr. Andersson replied.
Current American College of Cardiology/American Heart Association guidelines also endorse perioperative beta-blockade in patients with coronary artery disease undergoing vascular or intermediate-risk noncardiac surgery.
She noted that in drawing up the current guidelines, the ESC and ACC/AHA committees relied heavily on strongly positive randomized clinical trials whose validity has recently been called into question in a major research scandal. Indeed, the lead investigator in those studies, Dr. Don Poldermans – who also happened to be chairperson of the ESC guidelines-writing task force – has been dismissed from the faculty at Erasmus University in Rotterdam.
Dr. Omerovic, of Sahlgrenska University, Gothenburg, Sweden, asked for Dr. Andersson’s thoughts regarding a new meta-analysis by investigators at Imperial College London which excluded the suspect Dutch clinical trials. The investigators concluded that initiation of perioperative beta-blocker therapy was associated with a 27% increase in 30-day all-cause mortality, a 27% reduction in nonfatal MI, a 73% increase in stroke, and a 51% increase in hypotension.
"Patient safety being paramount, guidelines for perioperative beta-blocker initiation should be retracted without further delay," the meta-analysts argued (Heart 2013 July 31 [doi: 10.1136/heartjnl-2013-304262]).
"I read that meta-analysis with great interest," Dr. Andersson said. "I think there definitely is a heterogeneity in the effects of perioperative beta-blockers, and it depends on your baseline risk. Most of the studies in the meta-analysis included many patients at lower risk."
Dr. Andersson’s study was funded by the Danish Medical Research Foundation. She reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: Patients with heart failure who were on beta-blocker therapy at the time of noncardiac surgery had a 22% reduction in 30-day major adverse cardiovascular events, compared with those not on a beta-blocker perioperatively. In stark contrast, patients with ischemic heart disease but no history of heart failure or MI had a 44% greater risk of such events if they were on a perioperative beta-blocker.
Data source: This was a Danish national registry study that included more than 28,000 patients with ischemic heart disease who underwent noncardiac surgery.
Disclosures: Dr. Andersson’s study was funded by the Danish Medical Research Foundation. She reported having no financial conflicts of interest.
Psoriasis linked to increased heart failure risk
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AMSTERDAM – Psoriasis proved to be independently associated with an increased risk for new-onset heart failure in the first nationwide study to look at a possible relationship.
As word of the newly identified psoriasis/heart failure link spreads, it’s likely cardiologists will receive a growing number of referrals from dermatologists and primary care physicians for evaluation of possible heart failure in psoriasis patients who develop shortness of breath or other symptoms suggestive of ventricular dysfunction, Dr. Usman Khalid said at the annual congress of the European Society of Cardiology.
"Our results underlie the importance of considering the psoriatic population as a high-risk patient group in terms of cardiovascular risk. We encourage early screening for cardiovascular risk factors in psoriasis patients," he said.
Psoriasis is known to be associated with an increased cardiovascular event rate, presumably due to shared systemic inflammatory pathways, but the association between the dermatologic disease and heart failure, specifically, hadn’t been looked at in depth prior to Dr. Khalid’s presentation of a Danish study involving all 5.85 million Danish adults, who were followed from 1997 through 2009.
Among the Danish population without known heart failure at baseline were 57,049 individuals with mild psoriasis – identified by their use of prescription topical agents – and 11,638 others with severe psoriasis. The incidence of new-onset heart failure during follow-up was 2.27 cases per 1,000 person-years in subjects without psoriasis and a significantly greater 4.02 per 1,000 person-years in those with mild psoriasis and 4.50 per 1,000 person-years in individuals with severe psoriasis, reported Dr. Khalid of the University of Copenhagen.
In a multivariate regression analysis adjusted for demographic and socioeconomic factors, comorbid conditions, and use of cardiovascular medications, the likelihood of developing new-onset heart failure during follow-up was 64% greater among individuals with mild psoriasis and 85% greater in those with severe psoriasis than in psoriasis-free subjects.
Because of limitations in the registry database, it is not possible to determine the proportion of new-onset heart failure among psoriasis patients that involved systolic as opposed to diastolic dysfunction, or ischemic versus nonischemic etiology, Dr. Khalid said in response to audience questions.
This study was supported by a research grant from Leo Pharma. Dr. Khalid reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: Danish patients with mild psoriasis developed new-onset heart failure at the rate of 4.02 cases per 1,000 person-years of follow-up, while those with severe psoriasis did so at a pace of 4.50 cases per 1,000 person-years, both significantly higher rates than the 2.27 per 1,000 person-years in the general psoriasis-free population.
Data source: This was a Danish nationwide registry study that included all 5.85 million Danish adults. They were followed during 1997-2009 for development of heart failure.
Disclosures: The study was supported by a research grant from Leo Pharma. The presenter reported having no financial conflicts of interest.
Stroke risk climbs sharply with more risk factors, even without AF
AMSTERDAM – The presence of three or more ischemic stroke risk factors in patients with no history of atrial fibrillation or stroke spells high stroke risk, comparable with that seen in patients who do have atrial fibrillation, according to a Danish national study.
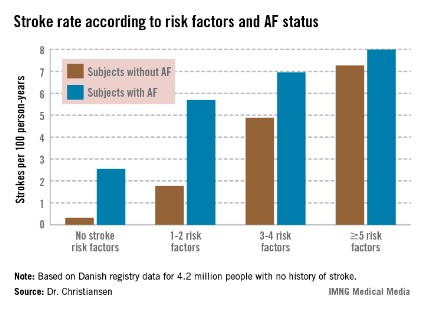
Moreover, individuals with five or more stroke risk factors have essentially the same high stroke risk – roughly 8% per year – regardless of whether they have a history of atrial fibrillation (AF) or not, Dr. Christine Benn Christiansen reported at the annual congress of the European Society of Cardiology.
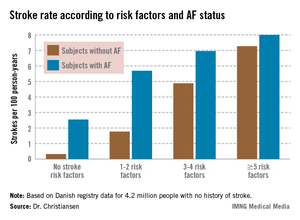
"The stroke risk increases with the number of risk factors. This has not been shown before in patients without prior stroke or atrial fibrillation," said Dr. Christiansen of Gentofte Hospital, Hellerup, Denmark.
"The main message here is there’s a great potential for reducing stroke risk in patients without atrial fibrillation," she added.
The impetus for the Danish national study lies in the fact that 80% of ischemic strokes occur in individuals without AF. The majority of these are first-time strokes. Yet the well-known guideline-recommended stroke-risk scores CHADS2 and CHA2DS2-VASc apply only to patients with AF.
"In patients without prior stroke, without AF, there is no systematic risk approach to deciding whether such people should receive any kind of antithrombotic medication to prevent ischemic strokes," Dr. Christiansen observed.
She and her coinvestigators analyzed comprehensive Danish national health care registries and identified 4.2 million adult Danes with no history of stroke. The investigators then followed these subjects, including 31,716 with a baseline diagnosis of AF, through the registries for 11 years, during which 130,336 patients were diagnosed with new-onset AF. The main goal was to compare 11-year stroke rates between patients with and without baseline AF based upon the number of stroke risk factors present.
The investigators cast a wide net in terms of stroke risk factors. They included epilepsy, heart failure, chronic systemic inflammatory conditions, hypertension, chronic renal disease, prior MI, diabetes, and peripheral artery disease. They also took into account venous thromboembolism, migraine, arterial embolism, carotid stenosis, and retinal vascular occlusion.
The stroke rate in subjects without baseline AF maxed out at 7.27 cases per 100 patient-years, or 7.27% annually, with five or more stroke risk factors present. Notably, the stroke rate was similar at 8.0% per year in patients with five or more baseline stroke risk factors plus AF (see graph).
ESC spokesperson Dr. Güenter Breithardt, emeritus professor of medicine at the University of Münster (Germany), said that he suspects the mechanism underlying the high stroke rate seen in patients with large numbers of stroke risk factors but no AF is that many of these individuals actually had silent AF that went unrecognized. Many of these stroke risk factors are also risk factors for development of AF, he observed.
Arguing against that hypothesis, Dr. Christiansen replied, is that each of the stroke risk factors as well as the development of AF were analyzed as time-dependent variables adjusted for age, gender, and the remaining stroke risk factors. Thus, patients with silent and unrecognized AF at baseline whose arrhythmia became symptomatic and got diagnosed during 11 years of follow-up were removed from the no-baseline-AF group for the data analysis, she said.
Dr. Harry Crijns, session cochair, summarized the central study findings as follows: "For patients who do not have a lot of accumulated risk factors, the additional presence of atrial fibrillation has a significant impact. On the other hand, in patients who have a lot of risk factors, atrial fibrillation doesn’t seem to have an impact. However, I would say searching for atrial fibrillation in such patients might differentiate a subgroup with even higher risk, although that wasn’t looked at in this study. That would be a good topic for future research."
In an interview, he said he doesn’t consider the Danish results practice changing. Patients with a large accumulation of the stroke risk factors used in the study, especially patients with established vascular disease, should already be on daily aspirin, yet at this point the Danish investigators aren’t able to say how many such patients were. However, he granted that in everyday practice today many such patients aren’t on aspirin, which is how the organizers of the ongoing, 5-year ARRIVE (Aspirin to Reduce Risk of Initial Vascular Events) trial were able to enroll roughly 12,000 high-risk patients who weren’t on aspirin to randomize them to daily aspirin or placebo.
No expert would recommend oral anticoagulant therapy for stroke prevention in patients without AF or prior stroke at this time, no matter how many stroke risk factors they have, because there are as yet no randomized trial data to support such a step. There will need to be evidence that the preventive benefit outstrips the increased bleeding risk.
"It would be marvelous if we knew how to fine-tune stroke risk, but the data are not there," added Dr. Crijns, professor and head of the department of cardiology at Maastricht (the Netherlands) University.
The study was funded by the University of Copenhagen and national research grants. None of the physicians had any financial conflicts of interest to disclose.
AMSTERDAM – The presence of three or more ischemic stroke risk factors in patients with no history of atrial fibrillation or stroke spells high stroke risk, comparable with that seen in patients who do have atrial fibrillation, according to a Danish national study.
Moreover, individuals with five or more stroke risk factors have essentially the same high stroke risk – roughly 8% per year – regardless of whether they have a history of atrial fibrillation (AF) or not, Dr. Christine Benn Christiansen reported at the annual congress of the European Society of Cardiology.

"The stroke risk increases with the number of risk factors. This has not been shown before in patients without prior stroke or atrial fibrillation," said Dr. Christiansen of Gentofte Hospital, Hellerup, Denmark.
"The main message here is there’s a great potential for reducing stroke risk in patients without atrial fibrillation," she added.
The impetus for the Danish national study lies in the fact that 80% of ischemic strokes occur in individuals without AF. The majority of these are first-time strokes. Yet the well-known guideline-recommended stroke-risk scores CHADS2 and CHA2DS2-VASc apply only to patients with AF.
"In patients without prior stroke, without AF, there is no systematic risk approach to deciding whether such people should receive any kind of antithrombotic medication to prevent ischemic strokes," Dr. Christiansen observed.
She and her coinvestigators analyzed comprehensive Danish national health care registries and identified 4.2 million adult Danes with no history of stroke. The investigators then followed these subjects, including 31,716 with a baseline diagnosis of AF, through the registries for 11 years, during which 130,336 patients were diagnosed with new-onset AF. The main goal was to compare 11-year stroke rates between patients with and without baseline AF based upon the number of stroke risk factors present.
The investigators cast a wide net in terms of stroke risk factors. They included epilepsy, heart failure, chronic systemic inflammatory conditions, hypertension, chronic renal disease, prior MI, diabetes, and peripheral artery disease. They also took into account venous thromboembolism, migraine, arterial embolism, carotid stenosis, and retinal vascular occlusion.
The stroke rate in subjects without baseline AF maxed out at 7.27 cases per 100 patient-years, or 7.27% annually, with five or more stroke risk factors present. Notably, the stroke rate was similar at 8.0% per year in patients with five or more baseline stroke risk factors plus AF (see graph).
ESC spokesperson Dr. Güenter Breithardt, emeritus professor of medicine at the University of Münster (Germany), said that he suspects the mechanism underlying the high stroke rate seen in patients with large numbers of stroke risk factors but no AF is that many of these individuals actually had silent AF that went unrecognized. Many of these stroke risk factors are also risk factors for development of AF, he observed.
Arguing against that hypothesis, Dr. Christiansen replied, is that each of the stroke risk factors as well as the development of AF were analyzed as time-dependent variables adjusted for age, gender, and the remaining stroke risk factors. Thus, patients with silent and unrecognized AF at baseline whose arrhythmia became symptomatic and got diagnosed during 11 years of follow-up were removed from the no-baseline-AF group for the data analysis, she said.
Dr. Harry Crijns, session cochair, summarized the central study findings as follows: "For patients who do not have a lot of accumulated risk factors, the additional presence of atrial fibrillation has a significant impact. On the other hand, in patients who have a lot of risk factors, atrial fibrillation doesn’t seem to have an impact. However, I would say searching for atrial fibrillation in such patients might differentiate a subgroup with even higher risk, although that wasn’t looked at in this study. That would be a good topic for future research."
In an interview, he said he doesn’t consider the Danish results practice changing. Patients with a large accumulation of the stroke risk factors used in the study, especially patients with established vascular disease, should already be on daily aspirin, yet at this point the Danish investigators aren’t able to say how many such patients were. However, he granted that in everyday practice today many such patients aren’t on aspirin, which is how the organizers of the ongoing, 5-year ARRIVE (Aspirin to Reduce Risk of Initial Vascular Events) trial were able to enroll roughly 12,000 high-risk patients who weren’t on aspirin to randomize them to daily aspirin or placebo.
No expert would recommend oral anticoagulant therapy for stroke prevention in patients without AF or prior stroke at this time, no matter how many stroke risk factors they have, because there are as yet no randomized trial data to support such a step. There will need to be evidence that the preventive benefit outstrips the increased bleeding risk.
"It would be marvelous if we knew how to fine-tune stroke risk, but the data are not there," added Dr. Crijns, professor and head of the department of cardiology at Maastricht (the Netherlands) University.
The study was funded by the University of Copenhagen and national research grants. None of the physicians had any financial conflicts of interest to disclose.
AMSTERDAM – The presence of three or more ischemic stroke risk factors in patients with no history of atrial fibrillation or stroke spells high stroke risk, comparable with that seen in patients who do have atrial fibrillation, according to a Danish national study.
Moreover, individuals with five or more stroke risk factors have essentially the same high stroke risk – roughly 8% per year – regardless of whether they have a history of atrial fibrillation (AF) or not, Dr. Christine Benn Christiansen reported at the annual congress of the European Society of Cardiology.

"The stroke risk increases with the number of risk factors. This has not been shown before in patients without prior stroke or atrial fibrillation," said Dr. Christiansen of Gentofte Hospital, Hellerup, Denmark.
"The main message here is there’s a great potential for reducing stroke risk in patients without atrial fibrillation," she added.
The impetus for the Danish national study lies in the fact that 80% of ischemic strokes occur in individuals without AF. The majority of these are first-time strokes. Yet the well-known guideline-recommended stroke-risk scores CHADS2 and CHA2DS2-VASc apply only to patients with AF.
"In patients without prior stroke, without AF, there is no systematic risk approach to deciding whether such people should receive any kind of antithrombotic medication to prevent ischemic strokes," Dr. Christiansen observed.
She and her coinvestigators analyzed comprehensive Danish national health care registries and identified 4.2 million adult Danes with no history of stroke. The investigators then followed these subjects, including 31,716 with a baseline diagnosis of AF, through the registries for 11 years, during which 130,336 patients were diagnosed with new-onset AF. The main goal was to compare 11-year stroke rates between patients with and without baseline AF based upon the number of stroke risk factors present.
The investigators cast a wide net in terms of stroke risk factors. They included epilepsy, heart failure, chronic systemic inflammatory conditions, hypertension, chronic renal disease, prior MI, diabetes, and peripheral artery disease. They also took into account venous thromboembolism, migraine, arterial embolism, carotid stenosis, and retinal vascular occlusion.
The stroke rate in subjects without baseline AF maxed out at 7.27 cases per 100 patient-years, or 7.27% annually, with five or more stroke risk factors present. Notably, the stroke rate was similar at 8.0% per year in patients with five or more baseline stroke risk factors plus AF (see graph).
ESC spokesperson Dr. Güenter Breithardt, emeritus professor of medicine at the University of Münster (Germany), said that he suspects the mechanism underlying the high stroke rate seen in patients with large numbers of stroke risk factors but no AF is that many of these individuals actually had silent AF that went unrecognized. Many of these stroke risk factors are also risk factors for development of AF, he observed.
Arguing against that hypothesis, Dr. Christiansen replied, is that each of the stroke risk factors as well as the development of AF were analyzed as time-dependent variables adjusted for age, gender, and the remaining stroke risk factors. Thus, patients with silent and unrecognized AF at baseline whose arrhythmia became symptomatic and got diagnosed during 11 years of follow-up were removed from the no-baseline-AF group for the data analysis, she said.
Dr. Harry Crijns, session cochair, summarized the central study findings as follows: "For patients who do not have a lot of accumulated risk factors, the additional presence of atrial fibrillation has a significant impact. On the other hand, in patients who have a lot of risk factors, atrial fibrillation doesn’t seem to have an impact. However, I would say searching for atrial fibrillation in such patients might differentiate a subgroup with even higher risk, although that wasn’t looked at in this study. That would be a good topic for future research."
In an interview, he said he doesn’t consider the Danish results practice changing. Patients with a large accumulation of the stroke risk factors used in the study, especially patients with established vascular disease, should already be on daily aspirin, yet at this point the Danish investigators aren’t able to say how many such patients were. However, he granted that in everyday practice today many such patients aren’t on aspirin, which is how the organizers of the ongoing, 5-year ARRIVE (Aspirin to Reduce Risk of Initial Vascular Events) trial were able to enroll roughly 12,000 high-risk patients who weren’t on aspirin to randomize them to daily aspirin or placebo.
No expert would recommend oral anticoagulant therapy for stroke prevention in patients without AF or prior stroke at this time, no matter how many stroke risk factors they have, because there are as yet no randomized trial data to support such a step. There will need to be evidence that the preventive benefit outstrips the increased bleeding risk.
"It would be marvelous if we knew how to fine-tune stroke risk, but the data are not there," added Dr. Crijns, professor and head of the department of cardiology at Maastricht (the Netherlands) University.
The study was funded by the University of Copenhagen and national research grants. None of the physicians had any financial conflicts of interest to disclose.
AT THE ESC CONGRESS 2013
Major finding: The ischemic stroke rate in patients with no history of stroke or atrial fibrillation who had five or more stroke risk factors at baseline was 7.27% annually, similar to the 8.0% rate in patients with five or more risk factors as well as atrial fibrillation.
Data source: A Danish national registry study of 4.2 million adults with no baseline history of stroke who were followed for 11 years.*
Disclosures: The study was funded by the University of Copenhagen and national research grants. The presenter reported having no financial conflicts.
* Correction, 9/24/2013: An earlier version of this story misstated the baseline history of people in the study.
Tour de France riders live longer
AMSTERDAM – French bicyclists who have competed in the Tour de France have a 41% lower mortality rate, compared with the age-matched male French general population, according to a first-of-its-kind study carried out on the 100th anniversary of the world’s most venerable professional bike race.
Causes of death were consistently lower across the board among the ex-racers. They had a 33% reduction in mortality owing to cardiovascular diseases, a 44% decrease in fatal cancers, a 72% reduction in deaths from respiratory disease, and a 78% decrease in deaths from digestive diseases, compared with controls. The sole cause-of-death category that was not significantly less common among the former Tour de France athletes was trauma, mostly from bike or auto crashes, Dr. Xavier Jouven reported at the annual congress of the European Society of Cardiology.
The study included all 786 French professional bike racers who competed in the Tour de France during 1947-2012. They participated in a mean 2.5 tours. By 2012, there had been 208 deaths.
The mortality advantage favoring the ex-racers was consistent across all 5-year age groups. It’s estimated that the Tour de France participants are averaging a 6- to 7-year greater longevity than do other Frenchmen, according to Dr. Jouven of Georges Pompidou Hospital, Paris.
He confessed to being surprised by the study results. He had anticipated finding an above-average mortality rate in the ex-racers because of the deleterious long-term effects of the various performance-enhancing drugs widely used over the decades, coupled with the sheer physical grind of participating in an event described as similar to racing a marathon per day for nearly 3 weeks and the crashes that are part of the job description.
"If there was really a danger in doing high-level exercise, then we should have observed an increased mortality rate. That’s actually what we’d expected to find," the cardiologist said.
The cyclists’ mortality reduction was consistent across all eras of the race: the 1950s and 1960s, when the use of amphetamines was common; the 1970s and 1980s, when anabolic steroids were the performance-enhancing drugs of choice; and most recently, the era of erythropoietin and human growth hormone, he noted.
The study was restricted to French cyclists because the investigators knew they could capture every single death through French registries.
Discussant Sanjay Sharma took issue with Dr. Jouven’s interpretation of the Tour de France data, which he said do not really address one of the hottest controversies in all of sports medicine: Is too much exercise cardiotoxic?
"I would like to have some data on comorbidities. I would like to have known how many of the surviving individuals have atrial fibrillation, compared with the general population. How many have implanted pacemakers? I would urge the investigators to go back and look at the survivors and produce [these] very important data," said Dr. Sharma of St. George’s Hospital, London.
He added that he was unimpressed by the investigators’ choice of a comparison group.
"They have compared possibly the fittest human beings in the world, people who are probably genetically, physically, physiologically, and psychologically superior to the general population, in which there is of course a much higher burden of comorbidities, a higher burden of risk factors for cardiovascular disease, a higher burden of adverse lifestyle, as well as a lower socioeconomic class. So I don’t believe that the Tour de France per se increases longevity of life; what I believe they have shown is, if you are capable of doing the Tour de France, then you may live 6-7 years longer than the average individual in the community," the cardiologist continued.
The benefits of physical exercise in the range of 40 metabolic equivalents (METs) per week spread over five or six workouts are well established. This level of exercise confers a 40%-50% reduction in cardiovascular mortality. But elite endurance athletes exercise at intensities 10- to 20-fold greater than that.
"There is emerging evidence that too much exercise may be deleterious to the heart," according to Dr. Sharma. "We know that marathon runners have high circulating markers of cardiac damage. Animal studies show increased inflammation and fibrosis within the myocardium. Recent cardiac magnetic resonance imaging studies have shown fibrosis in veteran athletes, and [electrophysiologists] continue to talk about the high prevalence of atrial fibrillation amongst veteran athletes."
Dr. Jouven’s presentation was big news in daily newspapers across cycling-crazy Europe. Simultaneous with his report on the Tour de France study in Amsterdam, the findings were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht347]) with an accompanying editorial coauthored by Dr. Sharma (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht373]).
Neither cardiologist had any relevant financial interests. The study was funded by a research grant from the Sudden Death Expertise Center in Paris.
AMSTERDAM – French bicyclists who have competed in the Tour de France have a 41% lower mortality rate, compared with the age-matched male French general population, according to a first-of-its-kind study carried out on the 100th anniversary of the world’s most venerable professional bike race.
Causes of death were consistently lower across the board among the ex-racers. They had a 33% reduction in mortality owing to cardiovascular diseases, a 44% decrease in fatal cancers, a 72% reduction in deaths from respiratory disease, and a 78% decrease in deaths from digestive diseases, compared with controls. The sole cause-of-death category that was not significantly less common among the former Tour de France athletes was trauma, mostly from bike or auto crashes, Dr. Xavier Jouven reported at the annual congress of the European Society of Cardiology.
The study included all 786 French professional bike racers who competed in the Tour de France during 1947-2012. They participated in a mean 2.5 tours. By 2012, there had been 208 deaths.
The mortality advantage favoring the ex-racers was consistent across all 5-year age groups. It’s estimated that the Tour de France participants are averaging a 6- to 7-year greater longevity than do other Frenchmen, according to Dr. Jouven of Georges Pompidou Hospital, Paris.
He confessed to being surprised by the study results. He had anticipated finding an above-average mortality rate in the ex-racers because of the deleterious long-term effects of the various performance-enhancing drugs widely used over the decades, coupled with the sheer physical grind of participating in an event described as similar to racing a marathon per day for nearly 3 weeks and the crashes that are part of the job description.
"If there was really a danger in doing high-level exercise, then we should have observed an increased mortality rate. That’s actually what we’d expected to find," the cardiologist said.
The cyclists’ mortality reduction was consistent across all eras of the race: the 1950s and 1960s, when the use of amphetamines was common; the 1970s and 1980s, when anabolic steroids were the performance-enhancing drugs of choice; and most recently, the era of erythropoietin and human growth hormone, he noted.
The study was restricted to French cyclists because the investigators knew they could capture every single death through French registries.
Discussant Sanjay Sharma took issue with Dr. Jouven’s interpretation of the Tour de France data, which he said do not really address one of the hottest controversies in all of sports medicine: Is too much exercise cardiotoxic?
"I would like to have some data on comorbidities. I would like to have known how many of the surviving individuals have atrial fibrillation, compared with the general population. How many have implanted pacemakers? I would urge the investigators to go back and look at the survivors and produce [these] very important data," said Dr. Sharma of St. George’s Hospital, London.
He added that he was unimpressed by the investigators’ choice of a comparison group.
"They have compared possibly the fittest human beings in the world, people who are probably genetically, physically, physiologically, and psychologically superior to the general population, in which there is of course a much higher burden of comorbidities, a higher burden of risk factors for cardiovascular disease, a higher burden of adverse lifestyle, as well as a lower socioeconomic class. So I don’t believe that the Tour de France per se increases longevity of life; what I believe they have shown is, if you are capable of doing the Tour de France, then you may live 6-7 years longer than the average individual in the community," the cardiologist continued.
The benefits of physical exercise in the range of 40 metabolic equivalents (METs) per week spread over five or six workouts are well established. This level of exercise confers a 40%-50% reduction in cardiovascular mortality. But elite endurance athletes exercise at intensities 10- to 20-fold greater than that.
"There is emerging evidence that too much exercise may be deleterious to the heart," according to Dr. Sharma. "We know that marathon runners have high circulating markers of cardiac damage. Animal studies show increased inflammation and fibrosis within the myocardium. Recent cardiac magnetic resonance imaging studies have shown fibrosis in veteran athletes, and [electrophysiologists] continue to talk about the high prevalence of atrial fibrillation amongst veteran athletes."
Dr. Jouven’s presentation was big news in daily newspapers across cycling-crazy Europe. Simultaneous with his report on the Tour de France study in Amsterdam, the findings were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht347]) with an accompanying editorial coauthored by Dr. Sharma (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht373]).
Neither cardiologist had any relevant financial interests. The study was funded by a research grant from the Sudden Death Expertise Center in Paris.
AMSTERDAM – French bicyclists who have competed in the Tour de France have a 41% lower mortality rate, compared with the age-matched male French general population, according to a first-of-its-kind study carried out on the 100th anniversary of the world’s most venerable professional bike race.
Causes of death were consistently lower across the board among the ex-racers. They had a 33% reduction in mortality owing to cardiovascular diseases, a 44% decrease in fatal cancers, a 72% reduction in deaths from respiratory disease, and a 78% decrease in deaths from digestive diseases, compared with controls. The sole cause-of-death category that was not significantly less common among the former Tour de France athletes was trauma, mostly from bike or auto crashes, Dr. Xavier Jouven reported at the annual congress of the European Society of Cardiology.
The study included all 786 French professional bike racers who competed in the Tour de France during 1947-2012. They participated in a mean 2.5 tours. By 2012, there had been 208 deaths.
The mortality advantage favoring the ex-racers was consistent across all 5-year age groups. It’s estimated that the Tour de France participants are averaging a 6- to 7-year greater longevity than do other Frenchmen, according to Dr. Jouven of Georges Pompidou Hospital, Paris.
He confessed to being surprised by the study results. He had anticipated finding an above-average mortality rate in the ex-racers because of the deleterious long-term effects of the various performance-enhancing drugs widely used over the decades, coupled with the sheer physical grind of participating in an event described as similar to racing a marathon per day for nearly 3 weeks and the crashes that are part of the job description.
"If there was really a danger in doing high-level exercise, then we should have observed an increased mortality rate. That’s actually what we’d expected to find," the cardiologist said.
The cyclists’ mortality reduction was consistent across all eras of the race: the 1950s and 1960s, when the use of amphetamines was common; the 1970s and 1980s, when anabolic steroids were the performance-enhancing drugs of choice; and most recently, the era of erythropoietin and human growth hormone, he noted.
The study was restricted to French cyclists because the investigators knew they could capture every single death through French registries.
Discussant Sanjay Sharma took issue with Dr. Jouven’s interpretation of the Tour de France data, which he said do not really address one of the hottest controversies in all of sports medicine: Is too much exercise cardiotoxic?
"I would like to have some data on comorbidities. I would like to have known how many of the surviving individuals have atrial fibrillation, compared with the general population. How many have implanted pacemakers? I would urge the investigators to go back and look at the survivors and produce [these] very important data," said Dr. Sharma of St. George’s Hospital, London.
He added that he was unimpressed by the investigators’ choice of a comparison group.
"They have compared possibly the fittest human beings in the world, people who are probably genetically, physically, physiologically, and psychologically superior to the general population, in which there is of course a much higher burden of comorbidities, a higher burden of risk factors for cardiovascular disease, a higher burden of adverse lifestyle, as well as a lower socioeconomic class. So I don’t believe that the Tour de France per se increases longevity of life; what I believe they have shown is, if you are capable of doing the Tour de France, then you may live 6-7 years longer than the average individual in the community," the cardiologist continued.
The benefits of physical exercise in the range of 40 metabolic equivalents (METs) per week spread over five or six workouts are well established. This level of exercise confers a 40%-50% reduction in cardiovascular mortality. But elite endurance athletes exercise at intensities 10- to 20-fold greater than that.
"There is emerging evidence that too much exercise may be deleterious to the heart," according to Dr. Sharma. "We know that marathon runners have high circulating markers of cardiac damage. Animal studies show increased inflammation and fibrosis within the myocardium. Recent cardiac magnetic resonance imaging studies have shown fibrosis in veteran athletes, and [electrophysiologists] continue to talk about the high prevalence of atrial fibrillation amongst veteran athletes."
Dr. Jouven’s presentation was big news in daily newspapers across cycling-crazy Europe. Simultaneous with his report on the Tour de France study in Amsterdam, the findings were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht347]) with an accompanying editorial coauthored by Dr. Sharma (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht373]).
Neither cardiologist had any relevant financial interests. The study was funded by a research grant from the Sudden Death Expertise Center in Paris.
AT THE ESC CONGRESS 2013
Major finding: French professional cyclists who participated in the Tour de France during 1947-2012 had a 41% lower mortality rate than did the age-matched general French male population.
Data source: This was a retrospective study that captured all 208 deaths that have occurred among the 786 French cyclists.
Disclosures: The study was funded by a research grant from the Sudden Death Expertise Center in Paris. The presenter had no financial conflicts.
RE-ALIGN reaction: Nix novel anticoagulants in mechanical valve patients
AMSTERDAM – The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (the Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug’s indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterwards, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf’s report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615].
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn’t address either.
The novel oral factor Xa inhibitors don’t act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf’s interpretation that the negative results most likely stemmed from dabigatran’s absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don’t have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf’s blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That’s quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It’s a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I’m not saying you necessarily should do that. If the patient is doing well on warfarin, I don’t see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers’ fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM – The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (the Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug’s indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterwards, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf’s report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615].
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn’t address either.
The novel oral factor Xa inhibitors don’t act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf’s interpretation that the negative results most likely stemmed from dabigatran’s absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don’t have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf’s blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That’s quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It’s a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I’m not saying you necessarily should do that. If the patient is doing well on warfarin, I don’t see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers’ fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AMSTERDAM – The complete failure of the oral direct thrombin inhibitor dabigatran in the RE-ALIGN trial spells big trouble for efforts to use the novel oral factor Xa inhibitors for anticoagulation in patients with a mechanical heart valve, RE-ALIGN steering committee cochair Dr. Frans Van de Werf cautioned at the annual congress of the European Society of Cardiology.
"It would not be wise for a cardiologist to prescribe these agents for a patient with a mechanical heart valve," declared Dr. Van de Werf, professor and chairman of the department of cardiovascular medicine at the Catholic University at Leuven (Belgium).
Dabigatran (Pradaxa) is approved as a more effective and convenient alternative to warfarin for prevention of stroke and other systemic embolism in patients with nonvalvular atrial fibrillation. RE-ALIGN (the Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) was an effort to expand the drug’s indications to include prevention of thromboembolic events in patients with mechanical heart valves.
The study was halted prematurely late last year when an interim analysis showed that dabigatran was both less safe and less protective than standard warfarin therapy. Shortly afterwards, the Food and Drug Administration and the European regulatory agency warned that dabigatran is contraindicated in patients with mechanical heart valves. Dr. Van de Werf’s report in Amsterdam, however, was the first presentation of the actual data.
RE-ALIGN was stopped after 252 patients with mechanical heart valves had been randomized 2:1 to dabigatran or warfarin. Dabigatran dosing was adjusted on the basis of creatinine clearance to achieve a trough plasma level of at least 50 ng/mL.
The study halt came in response to an interim analysis showing a 5% stroke rate in the dabigatran group, compared with 0% with warfarin. This was accompanied by a 4% rate of major bleeding with dabigatran, compared with 2% with warfarin, and a 27% rate of any bleeding with dabigatran versus 12% with warfarin. All dabigatran-treated patients with major bleeding had pericardial bleeding and were in the subgroup of participants who started on the direct thrombin inhibitor within 7 days post surgery.
Other unwelcome outcomes in the interim analysis included a 2% MI rate and 3% rate of valve thrombosis without symptoms in the dabigatran group, versus no such events in the warfarin arm.
"Overall, this study was clearly negative: more thrombotic events and more bleeding complications in spite of adjusting the dose of dabigatran based upon renal function," Dr. Van de Werf observed.
Simultaneous with his presentation of the RE-ALIGN findings at the ESC congress in Amsterdam, the results were published online (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMoa1300615].
In an accompanying editorial, Dr. Elaine M. Hylek of Boston University said the most likely explanation for the disappointing results in RE-ALIGN was that blood levels of dabigatran were inadequate (N. Engl. J. Med. 2013 Sept. 1 [doi:10.1056/NEJMe1310399]).
Dr. Van de Werf, however, rejected that as unlikely. Given that there were excesses of both thromboembolic and bleeding events, other dosing regimens would surely have sent rates of one type of complication or the other even higher, he argued.
Instead, he and his coinvestigators believe that the most likely explanation for the negative results lies in the differences in the mechanisms of action of dabigatran and warfarin. Dabigatran acts near the tail end of the coagulation cascade, on thrombin. In patients with a mechanical heart valve, thrombin is generated by exposure of blood to the artificial surface of the valve leaflets, activating what is known as the contact pathway of coagulation. Thrombin also is generated by the release of tissue factor from tissues injured during the valve surgery. Warfarin acts at both the contact pathway and tissue injury levels; dabigatran doesn’t address either.
The novel oral factor Xa inhibitors don’t act at the contact pathway and tissue injury levels, either, which is why Dr. Van de Werf cautioned against their off-label use in patients with a mechanical valve.
Discussant Dr. Alec Vahanian said that he "fully agrees" with Dr. Van de Werf’s interpretation that the negative results most likely stemmed from dabigatran’s absence of efficacy on the contact pathway.
"It sounds from this study that there is no future for dabigatran with this indication. We should not give this agent to a patient with a mechanical prosthesis, and personally I would extend that further and say we should not give this agent to patients with a bioprosthesis or other high-risk patients with severe valve disease. We don’t have the evidence for that yet," said Dr. Vahanian, head of cardiology at Bichat Hospital, Paris.
Patients with a mechanical heart valve constitute a challenging population at extreme thromboembolic risk, but Dr. Vahanian offered a comforting prediction: "We are nearing the end of the era of mechanical prosthetic valves," he declared. "Patients are getting older, bioprostheses are getting better, and valve repair techniques are improving."
Dr. Harry R. Buller took issue with Dr. Van de Werf’s blanket prediction that the factor Xa inhibitors will prove similarly unsafe and ineffective in patients with mechanical valves, as was dabigatran.
"We still need to find that out. One molecule of factor Xa generates about 1,000 thrombin molecules, so inhibiting coagulation at the level of Xa might actually be effective. What you see with dabigatran is inhibition at the very last stages of the whole coagulation cascade, when there are millions and millions of molecules of thrombin. That’s quite difficult. If you do it higher up in the cascade it might work," argued Dr. Buller, chairman of the department of vascular medicine at the Academic Medical Center, Amsterdam.
"Would you take that risk?" challenged Dr. Van de Werf.
"I know there are people taking that risk. I love studies; I would study it," Dr. Buller replied.
Session cochair Dr. Keith A.A. Fox of the University of Edinburgh posed a provocative question to Dr. Van de Werf: "Would you extrapolate these RE-ALIGN data to a patient in atrial fibrillation with a bioprosthetic valve?"
"It’s a difficult question," Dr. Van de Werf replied. "In principle, you could give one of the novel anticoagulants approved for atrial fibrillation, including dabigatran, because it is assumed there is no indication for oral anticoagulation for the bioprosthetic valve. I’m not saying you necessarily should do that. If the patient is doing well on warfarin, I don’t see the need to switch to one of the new anticoagulants."
RE-ALIGN was supported by Boehringer Ingelheim. Dr. Van de Werf reported receiving research grants and speakers’ fees from the company. Dr. Buller, Dr. Fox, and Dr. Vahanian reported no relevant financial interests.
AT THE ESC CONGRESS 2013
Major finding: Patients with a mechanical heart valve had significantly higher rates of both major bleeding (4% vs. 2%) and stroke (5% vs. 0%) on dabigatran, compared with warfarin.
Data source: The randomized, multicenter, international RE-ALIGN study was terminated early due to safety concerns after enrollment of 252 patients.
Disclosures: RE-ALIGN was supported by Boehringer Ingelheim. The presenter has received research funds and speaker’s fees from the company.
Fibroblast growth factor 21 promising in treating metabolic disorders
CHICAGO – Fibroblast growth factor 21 shows early promise as a novel therapy for a variety of metabolic disorders closely associated with type 2 diabetes.
A first-in-humans, proof-of-concept randomized trial involving 38 patients with type 2 diabetes showed that a pharmaceutically engineered variant of endogenous fibroblast growth factor 21 (FGF21) achieved "rapid and robust" improvements in lipid levels along with weight loss and reduced fasting insulin levels, Dr. Gregory A. Gaich reported at the annual scientific sessions of the American Diabetes Association.
"These results suggest that FGF21-based therapeutics may be useful for select metabolic disorders, including dyslipidemia and obesity. However, higher doses and longer periods of exposure may be required to really fully assess the potential of FGF21-based interventions in patients with type 2 diabetes," said Dr. Gaich of Eli Lilly, Indianapolis.
FGF21, discovered by Eli Lilly scientists, is a nonmitogenic member of the fibroblast growth factor family. It is involved in energy expenditure, induction of brown fat, free fatty acid oxidation, thermogenesis, and other physiologic processes. In primate and other animal studies, administration of exogenous FGF21 showed beneficial effects on lipids, body weight, and serum glucose, observations that paved the way for this randomized trial.
Participants in the 28-day, double-blind study were randomized to once-daily subcutaneous injection of the FGF21 variant, known for now as LY2405319, at 3, 10, or 20 mg or to placebo.
Subjects who received either of the two highest doses of the study drug showed 40%-50% reductions from baseline in serum triglycerides within 7 days, with maintenance of that benefit throughout the remaining 3 weeks of the trial. In contrast, reductions in LDL cholesterol occurred more gradually. By day 28, patients on the FGF21 compound at 10 or 20 mg/day showed 20%-30% reductions in LDL; however, their levels were still falling at that point, so the maximum LDL-lowering effect must await further study.
The levels of HDL cholesterol rose significantly by 15%-20% at all 3 doses of FGF21.
Body weight in the FGF21-treated patients fell by a mean of 1.7 kg. "We were delighted to see what we thought was a pretty encouraging weight loss for such a short study," Dr. Gaich commented.
Reduction in fasting plasma glucose showed a favorable dose-dependent trend that didn’t achieve statistical difference. Patients assigned to FGF21 at 20 mg/day had a 13-mg/dL greater drop over the course of the study than placebo-treated controls.
The reduction in fasting insulin levels with FGF21 was more encouraging. The effect was dose-dependent, with subjects in the 20 mg/day group exhibiting a highly significant 40% decrease from baseline.
Levels of two biomarkers followed serially in the study provide a hint as to FGF21’s mechanism of action. Levels of adiponectin, which is associated with increased insulin sensitivity, increased in dose-dependent fashion across all three groups of FGF21-treated patients and were nearly doubled at 20 mg/day compared to baseline. And levels of beta-hydroxybutyrate, a biomarker for lipid oxidation, showed across-the-board increases of 50%-90%.
No dose-dependent increase in the incidence of hypoglycemia was noted in the study. The chief safety concern was the occurrence of significant hypersensitivity reactions in two FGF21-treated patients, both in the 20 mg/day group. One patient developed a diffuse rash roughly halfway through the treatment period. The other experienced a more severe hypersensitivity reaction, including hypotension, on the second-to-last day of treatment.
Anti-LY2405319 antibodies arose in 80%-87% of patients on the drug at 10 and 20 mg/day. The clinical significance, if any, of these antibodies awaits further study. In this small initial study, there was no association between antibody production and any of the efficacy or safety parameters. Only one of the two patients with hypersensitivity reactions was antibody-positive, according to Dr. Gaich.
Dr. Gaich is an employee of Eli Lilly, which sponsored the study.
CHICAGO – Fibroblast growth factor 21 shows early promise as a novel therapy for a variety of metabolic disorders closely associated with type 2 diabetes.
A first-in-humans, proof-of-concept randomized trial involving 38 patients with type 2 diabetes showed that a pharmaceutically engineered variant of endogenous fibroblast growth factor 21 (FGF21) achieved "rapid and robust" improvements in lipid levels along with weight loss and reduced fasting insulin levels, Dr. Gregory A. Gaich reported at the annual scientific sessions of the American Diabetes Association.
"These results suggest that FGF21-based therapeutics may be useful for select metabolic disorders, including dyslipidemia and obesity. However, higher doses and longer periods of exposure may be required to really fully assess the potential of FGF21-based interventions in patients with type 2 diabetes," said Dr. Gaich of Eli Lilly, Indianapolis.
FGF21, discovered by Eli Lilly scientists, is a nonmitogenic member of the fibroblast growth factor family. It is involved in energy expenditure, induction of brown fat, free fatty acid oxidation, thermogenesis, and other physiologic processes. In primate and other animal studies, administration of exogenous FGF21 showed beneficial effects on lipids, body weight, and serum glucose, observations that paved the way for this randomized trial.
Participants in the 28-day, double-blind study were randomized to once-daily subcutaneous injection of the FGF21 variant, known for now as LY2405319, at 3, 10, or 20 mg or to placebo.
Subjects who received either of the two highest doses of the study drug showed 40%-50% reductions from baseline in serum triglycerides within 7 days, with maintenance of that benefit throughout the remaining 3 weeks of the trial. In contrast, reductions in LDL cholesterol occurred more gradually. By day 28, patients on the FGF21 compound at 10 or 20 mg/day showed 20%-30% reductions in LDL; however, their levels were still falling at that point, so the maximum LDL-lowering effect must await further study.
The levels of HDL cholesterol rose significantly by 15%-20% at all 3 doses of FGF21.
Body weight in the FGF21-treated patients fell by a mean of 1.7 kg. "We were delighted to see what we thought was a pretty encouraging weight loss for such a short study," Dr. Gaich commented.
Reduction in fasting plasma glucose showed a favorable dose-dependent trend that didn’t achieve statistical difference. Patients assigned to FGF21 at 20 mg/day had a 13-mg/dL greater drop over the course of the study than placebo-treated controls.
The reduction in fasting insulin levels with FGF21 was more encouraging. The effect was dose-dependent, with subjects in the 20 mg/day group exhibiting a highly significant 40% decrease from baseline.
Levels of two biomarkers followed serially in the study provide a hint as to FGF21’s mechanism of action. Levels of adiponectin, which is associated with increased insulin sensitivity, increased in dose-dependent fashion across all three groups of FGF21-treated patients and were nearly doubled at 20 mg/day compared to baseline. And levels of beta-hydroxybutyrate, a biomarker for lipid oxidation, showed across-the-board increases of 50%-90%.
No dose-dependent increase in the incidence of hypoglycemia was noted in the study. The chief safety concern was the occurrence of significant hypersensitivity reactions in two FGF21-treated patients, both in the 20 mg/day group. One patient developed a diffuse rash roughly halfway through the treatment period. The other experienced a more severe hypersensitivity reaction, including hypotension, on the second-to-last day of treatment.
Anti-LY2405319 antibodies arose in 80%-87% of patients on the drug at 10 and 20 mg/day. The clinical significance, if any, of these antibodies awaits further study. In this small initial study, there was no association between antibody production and any of the efficacy or safety parameters. Only one of the two patients with hypersensitivity reactions was antibody-positive, according to Dr. Gaich.
Dr. Gaich is an employee of Eli Lilly, which sponsored the study.
CHICAGO – Fibroblast growth factor 21 shows early promise as a novel therapy for a variety of metabolic disorders closely associated with type 2 diabetes.
A first-in-humans, proof-of-concept randomized trial involving 38 patients with type 2 diabetes showed that a pharmaceutically engineered variant of endogenous fibroblast growth factor 21 (FGF21) achieved "rapid and robust" improvements in lipid levels along with weight loss and reduced fasting insulin levels, Dr. Gregory A. Gaich reported at the annual scientific sessions of the American Diabetes Association.
"These results suggest that FGF21-based therapeutics may be useful for select metabolic disorders, including dyslipidemia and obesity. However, higher doses and longer periods of exposure may be required to really fully assess the potential of FGF21-based interventions in patients with type 2 diabetes," said Dr. Gaich of Eli Lilly, Indianapolis.
FGF21, discovered by Eli Lilly scientists, is a nonmitogenic member of the fibroblast growth factor family. It is involved in energy expenditure, induction of brown fat, free fatty acid oxidation, thermogenesis, and other physiologic processes. In primate and other animal studies, administration of exogenous FGF21 showed beneficial effects on lipids, body weight, and serum glucose, observations that paved the way for this randomized trial.
Participants in the 28-day, double-blind study were randomized to once-daily subcutaneous injection of the FGF21 variant, known for now as LY2405319, at 3, 10, or 20 mg or to placebo.
Subjects who received either of the two highest doses of the study drug showed 40%-50% reductions from baseline in serum triglycerides within 7 days, with maintenance of that benefit throughout the remaining 3 weeks of the trial. In contrast, reductions in LDL cholesterol occurred more gradually. By day 28, patients on the FGF21 compound at 10 or 20 mg/day showed 20%-30% reductions in LDL; however, their levels were still falling at that point, so the maximum LDL-lowering effect must await further study.
The levels of HDL cholesterol rose significantly by 15%-20% at all 3 doses of FGF21.
Body weight in the FGF21-treated patients fell by a mean of 1.7 kg. "We were delighted to see what we thought was a pretty encouraging weight loss for such a short study," Dr. Gaich commented.
Reduction in fasting plasma glucose showed a favorable dose-dependent trend that didn’t achieve statistical difference. Patients assigned to FGF21 at 20 mg/day had a 13-mg/dL greater drop over the course of the study than placebo-treated controls.
The reduction in fasting insulin levels with FGF21 was more encouraging. The effect was dose-dependent, with subjects in the 20 mg/day group exhibiting a highly significant 40% decrease from baseline.
Levels of two biomarkers followed serially in the study provide a hint as to FGF21’s mechanism of action. Levels of adiponectin, which is associated with increased insulin sensitivity, increased in dose-dependent fashion across all three groups of FGF21-treated patients and were nearly doubled at 20 mg/day compared to baseline. And levels of beta-hydroxybutyrate, a biomarker for lipid oxidation, showed across-the-board increases of 50%-90%.
No dose-dependent increase in the incidence of hypoglycemia was noted in the study. The chief safety concern was the occurrence of significant hypersensitivity reactions in two FGF21-treated patients, both in the 20 mg/day group. One patient developed a diffuse rash roughly halfway through the treatment period. The other experienced a more severe hypersensitivity reaction, including hypotension, on the second-to-last day of treatment.
Anti-LY2405319 antibodies arose in 80%-87% of patients on the drug at 10 and 20 mg/day. The clinical significance, if any, of these antibodies awaits further study. In this small initial study, there was no association between antibody production and any of the efficacy or safety parameters. Only one of the two patients with hypersensitivity reactions was antibody-positive, according to Dr. Gaich.
Dr. Gaich is an employee of Eli Lilly, which sponsored the study.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Patients with type 2 diabetes who received fibroblast growth factor 21 responded with 40%-50% reductions in serum triglycerides within 7 days, and these reduced levels were maintained throughout the remaining 3 weeks of the study.
Data source: A randomized, prospective, double-blind, first-in-humans pilot study in 38 patients with type 2 diabetes assigned to 28 days of once-daily subcutaneous injection of a synthetic fibroblast growth factor 21 variant or to placebo.
Disclosures: The study was sponsored by Eli Lilly. The presenter is a company employee.
New heart failure inotrope could be ‘Holy Grail’
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AMSTERDAM – The novel intravenous inotropic agent omecamtiv mecarbil achieved greater reduction in dyspnea scores and less likelihood of worsening heart failure at the highest dose studied, compared with placebo, in patients with acute decompensated heart failure in a phase II, dose-ranging study.
The drug also demonstrated a strong dose-dependent increase in systolic ejection time, no evidence of a proarrhythmic effect, and a greater reduction in heart rate with a larger increase in blood pressure than in placebo-treated controls, Dr. John R. Teerlink reported at the annual congress of the European Society of Cardiology.
In the overall population on omecamtiv mecarbil, however, which included the patients in the low- and intermediate-dose arms of this dose-ranging study, the dyspnea response rate wasn’t better than with placebo. Hence, the drug’s future is uncertain.
In a press release following Dr. Teerlink’s presentation, Amgen, cosponsor of the phase II ATOMIC-AHF (Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure) trial, said that no decision has been made as to whether to move omecamtiv mecarbil into phase III clinical trials. Dr. Teerlink thinks the drug has earned a shot.
"I personally believe this drug has continued to demonstrate its promise all along the way. It could replace milrinone and dobutamine in the hospital to improve cardiac performance," predicted Dr. Teerlink, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the San Francisco Veterans Affairs Medical Center.
Omecamtiv mecarbil is a novel selective cardiac myosin activator. It increases the entry rate of myosin into the force-producing state with actin. It boosts cardiac output through prolongation of the duration of systole and increased stroke volume, with no change in myocardial contractility and no increases in myocardial oxygen demand or myocyte calcium.
"It’s essentially producing more hands pulling on the rope," the cardiologist explained.
Discussant Theresa A. McDonagh said omecamtiv mecarbil has drawn great interest within the heart failure field because current inotropes are recognized as "very inadequate."
"It is truly a new inotrope, a drug that acts on the sarcomere itself. It does seem to be free of the nemesis of other inotropes: increased heart rate, greater myocardial oxygen consumption, arrhythmia production, and increased mortality in heart failure. It appears to be the Holy Grail, and may be capable of replacing dobutamine and milrinone," said Dr. McDonagh, professor of heart failure at King’s College London.
"I think this [ATOMIC-AHF trial] is a very promising start for omecamtiv mecarbil in acute heart failure," she added.
ATOMIC-AHF included 613 patients with a left ventricular ejection fraction of 40% or less who were hospitalized for acute heart failure with dyspnea at rest or with minimal exertion despite intravenous diuretic therapy. The study was conducted at 106 sites on three continents. Participants were randomized to 48 hours of intravenous placebo or omecamtiv mecarbil in three different ascending dose regimens.
The primary efficacy endpoint, dyspnea at 6, 24, and 48 hours across the three doses, compared with the pooled placebo group, was not met.
However, patients in the highest-dose omecamtiv mecarbil study arm had a 51% rate of significant improvement in dyspnea on a self-rated 7-point Likert scale at 48 hours, a significantly better response than the 37% rate in matched placebo-treated controls (P = .03). This comparison was a prespecified secondary endpoint in the study.
In addition, patients in the middle- and high-dose omecamtiv mecarbil arms had roughly a 45% reduction in the 7-day incidence of worsening heart failure, compared with the 17% rate in controls.
Not only was there no evidence of a proarrhythmic effect with omecamtiv mecarbil, the drug was associated with a 50% reduction in the incidence of supraventricular tachyarrhythmias, compared with placebo, he continued.
A modest increase in troponin I levels was noted in the omecamtiv mecarbil–treated group, but it was unrelated to plasma drug concentrations, which Dr. Teerlink found reassuring. Nevertheless, Dr. McDonagh said the small increases in troponin "should herald some caution amid the enthusiasm here."
The drug’s mechanism of action, which results in greater systolic ejection time, always raises the concern that it might also shorten diastole and compromise coronary perfusion. However, the observed reduction in heart rate makes that seem less likely, she added.
The ATOMIC-AHF study population was narrowly defined and of a profile commonly seen in clinical practice. They had an estimated glomerular filtration rate of about 50 mL/min, and they presented with dyspnea but didn’t have an acute coronary syndrome.
"This study population is an area of great unmet need in heart failure, with mortality rates of about 10% during their hospitalization and 30% at 1 year following discharge," according to Dr. McDonagh.
Nevertheless, it will be important in phase III testing to see how the drug performs in a broader group of heart failure patients, including those with lower estimated glomerular filtration rates, she added.
In an interview, Dr. Teerlink said that before a decision is reached regarding advancement of omecamtiv mecarbil to phase III studies, investigators and the study sponsors first want to see the data from an ongoing phase II dose-finding study of an oral formulation of the drug in chronic heart failure patients. This study, COSMIC-HF (Chronic Oral Study of Myosin Activation to Increase Contractility in Heart Failure), is still enrolling patients.
"We’ll get the combined data from ATOMIC-AHF and COSMIC-HF and then decide what phase III should look like. There are a lot of options here: IV [intravenous] alone, IV transitioning to chronic oral therapy, or even oral alone," he explained.
"One of the greatest challenges of the ATOMIC-AHF trial has been managing expectations," according to Dr. Teerlink. "Omecamtiv mecarbil is probably one of the most interesting new chemical entities in cardiovascular medicine now, and consequently expectations are very high in the scientific and lay communities."
The studies are sponsored by Amgen and Cytokinetics. Dr. Teerlink has received research grants and/or consulting fees from those companies as well as a handful of others. Dr. McDonagh reported no financial conflicts.
AT THE ESC CONGRESS 2013
Major finding: Fifty-one percent of patients with acute heart failure assigned to the high-dose arm in a study of intravenous omecamtiv mecarbil showed significant improvement in dyspnea scores at 48 hours, compared with 37% of placebo-treated controls.
Data source: The ATOMIC-AHF trial was a 613-patient, phase II, randomized, placebo-controlled, double-blind, dose-finding study.
Disclosures: The study was sponsored by Amgen and Cytokinetics. The presenter has received research grants and consultant fees from the companies.