User login
New clinical practice guidelines on pheochromocytomas
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
AT ICE/ENDO 2014
Soy supplements boost low testosterone in diabetic men
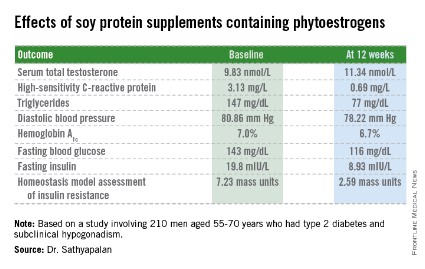
CHICAGO – Soy protein bars containing phytoestrogens raised borderline low serum total testosterone levels while improving cardiovascular risk factors and glycemic control in older type 2 diabetic men with compensated hypogonadism.
The randomized trial was conducted because of theoretic safety concerns surrounding consumption of soy supplements by this population. Soy contains phytoestrogens, natural estrogens that might have a testosterone-lowering effect that would be particularly unwelcome in older men with type 2 diabetes, because low testosterone is associated with increased cardiovascular risk.
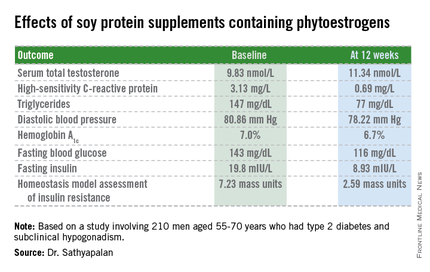
Thus, there was the possibility that the established cardiovascular and type 2 diabetic preventive benefits of frequent soy intake might be negated by a concomitant testosterone-lowering effect in men already at increased cardiovascular risk with low testosterone, Dr. Thozhukat Sathyapalan explained at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Those concerns were shown to be groundless in the 12-week, double-blind, randomized clinical trial, reported Dr. Sathyapalan, an endocrinologist at the University of Hull (U.K.).
The study included 210 men with type 2 diabetes aged 55-70 years with subclinical hypogonadism as defined by a serum total testosterone of 12 nmol/L or less but normal gonadotrophin levels. They were randomized to 3 months of daily consumption of two cereal bars, each containing 30 g of soy protein, with the bars eaten by one group containing 66 mg each of soy phytoestrogens, while the other group’s soy bars were phytoestrogen free. That level of soy phytoestrogen consumption – 132 mg/day – is equivalent to intake in a typical Asian diet.
The primary outcome was change in serum total testosterone over the course of 3 months. It rose in both groups. Diastolic blood pressure fell to a similar extent in both study arms, while systolic pressure was unchanged over time. Endothelial function as assessed by the reactive hyperemia index improved in the soy-plus-phytoestrogens group and worsened in the soy-only group. Improvements in multiple other markers of cardiovascular risk and glycemic control were restricted to the soy-with-phytoestrogens group. Body weight remained steady in both groups throughout the study period.
The study was funded by the U.K. Food Standards Agency, which regulates food safety. Dr. Sathyapalan reported having no financial conflicts.
CHICAGO – Soy protein bars containing phytoestrogens raised borderline low serum total testosterone levels while improving cardiovascular risk factors and glycemic control in older type 2 diabetic men with compensated hypogonadism.
The randomized trial was conducted because of theoretic safety concerns surrounding consumption of soy supplements by this population. Soy contains phytoestrogens, natural estrogens that might have a testosterone-lowering effect that would be particularly unwelcome in older men with type 2 diabetes, because low testosterone is associated with increased cardiovascular risk.

Thus, there was the possibility that the established cardiovascular and type 2 diabetic preventive benefits of frequent soy intake might be negated by a concomitant testosterone-lowering effect in men already at increased cardiovascular risk with low testosterone, Dr. Thozhukat Sathyapalan explained at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Those concerns were shown to be groundless in the 12-week, double-blind, randomized clinical trial, reported Dr. Sathyapalan, an endocrinologist at the University of Hull (U.K.).
The study included 210 men with type 2 diabetes aged 55-70 years with subclinical hypogonadism as defined by a serum total testosterone of 12 nmol/L or less but normal gonadotrophin levels. They were randomized to 3 months of daily consumption of two cereal bars, each containing 30 g of soy protein, with the bars eaten by one group containing 66 mg each of soy phytoestrogens, while the other group’s soy bars were phytoestrogen free. That level of soy phytoestrogen consumption – 132 mg/day – is equivalent to intake in a typical Asian diet.
The primary outcome was change in serum total testosterone over the course of 3 months. It rose in both groups. Diastolic blood pressure fell to a similar extent in both study arms, while systolic pressure was unchanged over time. Endothelial function as assessed by the reactive hyperemia index improved in the soy-plus-phytoestrogens group and worsened in the soy-only group. Improvements in multiple other markers of cardiovascular risk and glycemic control were restricted to the soy-with-phytoestrogens group. Body weight remained steady in both groups throughout the study period.
The study was funded by the U.K. Food Standards Agency, which regulates food safety. Dr. Sathyapalan reported having no financial conflicts.
CHICAGO – Soy protein bars containing phytoestrogens raised borderline low serum total testosterone levels while improving cardiovascular risk factors and glycemic control in older type 2 diabetic men with compensated hypogonadism.
The randomized trial was conducted because of theoretic safety concerns surrounding consumption of soy supplements by this population. Soy contains phytoestrogens, natural estrogens that might have a testosterone-lowering effect that would be particularly unwelcome in older men with type 2 diabetes, because low testosterone is associated with increased cardiovascular risk.

Thus, there was the possibility that the established cardiovascular and type 2 diabetic preventive benefits of frequent soy intake might be negated by a concomitant testosterone-lowering effect in men already at increased cardiovascular risk with low testosterone, Dr. Thozhukat Sathyapalan explained at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Those concerns were shown to be groundless in the 12-week, double-blind, randomized clinical trial, reported Dr. Sathyapalan, an endocrinologist at the University of Hull (U.K.).
The study included 210 men with type 2 diabetes aged 55-70 years with subclinical hypogonadism as defined by a serum total testosterone of 12 nmol/L or less but normal gonadotrophin levels. They were randomized to 3 months of daily consumption of two cereal bars, each containing 30 g of soy protein, with the bars eaten by one group containing 66 mg each of soy phytoestrogens, while the other group’s soy bars were phytoestrogen free. That level of soy phytoestrogen consumption – 132 mg/day – is equivalent to intake in a typical Asian diet.
The primary outcome was change in serum total testosterone over the course of 3 months. It rose in both groups. Diastolic blood pressure fell to a similar extent in both study arms, while systolic pressure was unchanged over time. Endothelial function as assessed by the reactive hyperemia index improved in the soy-plus-phytoestrogens group and worsened in the soy-only group. Improvements in multiple other markers of cardiovascular risk and glycemic control were restricted to the soy-with-phytoestrogens group. Body weight remained steady in both groups throughout the study period.
The study was funded by the U.K. Food Standards Agency, which regulates food safety. Dr. Sathyapalan reported having no financial conflicts.
AT ICE/ENDO 2014
Key clinical point: Soy supplements containing phytoestrogens in amounts equivalent to a typical Asian diet don’t reduce testosterone levels in older type 2 diabetic men with subclinical hypogonadism; instead, they increase testosterone.
Major finding: Eating cereal bars containing soy protein with soy phytoestrogens resulted in increased serum total testosterone, lowered cardiovascular risk factors, and improved metabolic control in older type 2 diabetic men with borderline low testosterone.
Data source: A 12-week, randomized, double-blind clinical trial of 210 men aged 55-70.
Disclosures: The U.K. Food Standards Agency funded the study. The presenter reported having no conflicts of interest.
Reoperative Bariatric Surgery Yields Low Complication Rates
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.
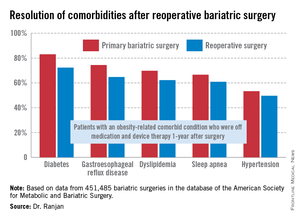
"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
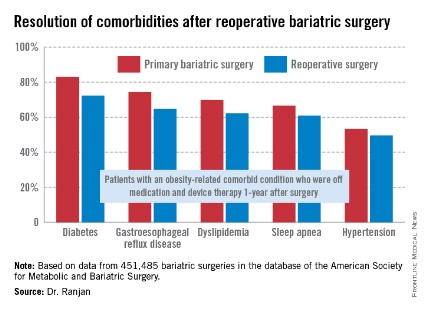
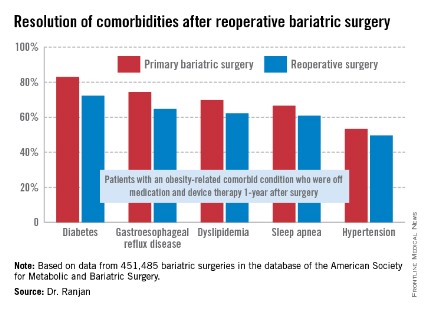
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.

"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.

"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
AT DDW 2014
Reoperative bariatric surgery yields low complication rates, substantial weight loss at 1 year
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.
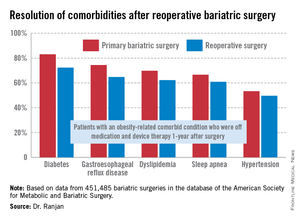
"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.

"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
CHICAGO – Reoperative bariatric surgery has impressively low major morbidity and mortality, substantial 1-year weight loss, and a high rate of resolution of the common obesity-linked comorbid conditions, according to an analysis of a large national database.
These data demonstrate that outcomes after contemporary reoperative bariatric surgery are better than believed by insurance carriers, who often deny coverage for these procedures because of a misperception that complication rates are high and benefits uncertain, Dr. Ranjan Sudan said at the annual Digestive Disease Week.

"I think that these data need to get out there to the stakeholders," added Dr. Sudan, a bariatric surgeon and a digestive disorders specialist who is vice chair of education in the department of surgery at Duke University, Durham, N.C.
He presented an analysis of outcomes after reoperative surgery that was carried out by a task force of the American Society for Metabolic and Bariatric Surgery. He and his coinvestigators reviewed all 451,485 bariatric surgery operations entered into the society’s prospective database during a 5-year period ending in spring 2012. The procedures were performed by 1,029 participating surgeons at 709 U.S. hospitals.
The focus of this analysis was on the 6.3% of operations that were reoperations. A total of 70% of the reoperations were corrective operations, essentially redos of the same type of procedure performed initially. The other 30% were conversion procedures, as when a patient who had a gastric band procedure on the first go-round subsequently was converted to a Roux-en-Y gastric bypass or sleeve procedure.
The mean length of stay for primary bariatric operations was 1.78 days, compared with 2.04 days for reoperative corrective procedures and 2.86 days for conversion operations.
The 30-day incidence of serious adverse events, such as leaks, bleeding, or pulmonary embolism, was 1.61% for primary procedures, nearly identical at 1.66% for corrective reoperations, and 3.26% for conversion procedures. The 1-year rate of serious adverse events was 1.87% for primary bariatric operations, 1.9% for corrective procedures, and 3.61% for conversions.
The mortality rate in patients undergoing a primary operation was 0.10% at 30 days and 0.17% at 1 year. Among patients who underwent reoperative bariatric surgery, the 30-day and 1-year mortality rates were 0.14% and 0.26%, respectively.
"Most bariatric surgery patients do not need reoperations. It’s gratifying to see that among those who do, the severe complication rates were low and acceptable and comorbidities often resolved [see chart]," Dr. Sudan declared.
The 1-year rate of excess weight loss following reoperative surgery averaged 36%.
Discussant Dr. Alfons Pomp liked what he saw from the registry.
"Your data show just how good we as bariatric surgeons are to operate on these surgically difficult, very obese, and seriously ill patients, mostly laparoscopically, and get pretty amazing results," commented Dr. Pomp, professor of surgery and chief of GI metabolic and bariatric surgery at Cornell University in New York.
The registry study was funded by Covidien. Dr. Sudan reported having no financial conflicts of interest.
AT DDW 2014
Key clinical point: Reoperative bariatric surgery is considerably safer than previously recognized outside the specialist surgical community. The 1-year excess weight loss is substantial, and common obesity-related comorbidities often resolve.
Major finding: Mortality rates at 30 days and 1-year following reoperative bariatric surgery were just 0.14% and 0. 26%, with an average excess weight loss of 36%at 1 year.
Data source: This was an analysis of more than 450,000 consecutive bariatric surgery operations entered into a prospective national database. Reoperations accounted for 6.3% of the procedures.
Disclosures: The study was funded by Covidien. The presenter reported having no financial conflicts.
Primary prevention of diabetic kidney disease: Thumbs up/down
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
EXPERT ANALYSIS FROM SCM 14
Hypothyroid patients may need to surrender the car keys
CHICAGO – Hypothyroid patients exhibit objective cognitive deficits and motor slowing rendering them unsafe to operate a motor vehicle.
That’s the key take-home message from a longitudinal study in which 32 patients with thyroid cancer completed an extensive battery of neurocognitive and psychological tests as well as measured performance on a driving simulator at three time points: while euthyroid, again while temporarily hypothyroid as part of their cancer therapy and assessment, and finally while once again euthyroid after restoration of thyroid hormone therapy.
"These findings provide objective evidence warranting admonitions against operating motor vehicles for hypothyroid patients and confidence in removing such stipulations upon restoration of a euthyroid state," Dr. Kenneth B. Ain said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
In his own clinical practice he has long included a boxed warning against driving while hypothyroid on all of his written instructions to patients. But most physicians don’t warn their hypothyroid patients that they are driving impaired, nor do the joint practice guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists address the issue. That’s largely because there hasn’t been objective, quantitative evidence to provide firm support for such cautionary admonitions – until now, observed Dr. Ain, professor of medicine and director of the thyroid oncology program at the University of Kentucky, Lexington.
While hypothyroid, study participants experienced an 8.5% increase in braking time on a driving simulator. That’s equivalent to the degree of impairment other investigators have shown to be associated with a blood alcohol level of 82 mg/dL, which is above the legal driving limit in the United States.
"Once our study is published, a patient who is involved in an auto accident [in which] there is death or significant harm could be considered an impaired patient. And if physicians do not warn the patient of this risk, they would be considered an agent of harm. They could be liable for the consequences, the same as a neurologist who doesn’t warn a patient with a grand mal seizure disorder not to drive," Dr. Ain said.
In an interview, he noted that thyroid cancer patients undergoing thyroid hormone depletion temporarily as part of their treatment are merely a small fraction of the total impaired hypothyroid driver population. Investigators with the Framingham Heart Study have reported that 4.4% of individuals above age 60 are hypothyroid. Many of these individuals remain undiagnosed or undertreated. Plus, the noncompliance rate with levothyroxine therapy has been estimated at 17%-32%.
Moreover, once a patient is newly diagnosed as being profoundly hypothyroid and receives a prescription for thyroid hormone replacement, there is a lag time involved in achieving a euthyroid state. The half-life of levothyroxine is 1 week. It takes 6-8 weeks to reach a steady state. Probably at least 2 weeks of therapy are required before there is any improvement in the neurologic impairments documented in this study, Dr. Ain speculated.
"We’re really talking here about a public health problem, one that requires a public health response and acknowledgment that this is a danger," according to the endocrinologist.
Testing during the hypothyroid phase of the study showed significant declines in measures of executive function and information-processing speed. Fine motor performance of the hands was slowed by 13%. Mean scores on the Beck Depression Inventory deteriorated from 7.9 while euthyroid to 18.9 while hypothyroid, consistent with mild bordering on moderate depression; this depression was characterized by vegetative symptoms and altered mood, but without the impaired self-esteem and sense of guilt often characteristic of other forms of depression.
Dr. Ain reported receiving a research grant from Genzyme, which funded this study.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
CHICAGO – Hypothyroid patients exhibit objective cognitive deficits and motor slowing rendering them unsafe to operate a motor vehicle.
That’s the key take-home message from a longitudinal study in which 32 patients with thyroid cancer completed an extensive battery of neurocognitive and psychological tests as well as measured performance on a driving simulator at three time points: while euthyroid, again while temporarily hypothyroid as part of their cancer therapy and assessment, and finally while once again euthyroid after restoration of thyroid hormone therapy.
"These findings provide objective evidence warranting admonitions against operating motor vehicles for hypothyroid patients and confidence in removing such stipulations upon restoration of a euthyroid state," Dr. Kenneth B. Ain said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
In his own clinical practice he has long included a boxed warning against driving while hypothyroid on all of his written instructions to patients. But most physicians don’t warn their hypothyroid patients that they are driving impaired, nor do the joint practice guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists address the issue. That’s largely because there hasn’t been objective, quantitative evidence to provide firm support for such cautionary admonitions – until now, observed Dr. Ain, professor of medicine and director of the thyroid oncology program at the University of Kentucky, Lexington.
While hypothyroid, study participants experienced an 8.5% increase in braking time on a driving simulator. That’s equivalent to the degree of impairment other investigators have shown to be associated with a blood alcohol level of 82 mg/dL, which is above the legal driving limit in the United States.
"Once our study is published, a patient who is involved in an auto accident [in which] there is death or significant harm could be considered an impaired patient. And if physicians do not warn the patient of this risk, they would be considered an agent of harm. They could be liable for the consequences, the same as a neurologist who doesn’t warn a patient with a grand mal seizure disorder not to drive," Dr. Ain said.
In an interview, he noted that thyroid cancer patients undergoing thyroid hormone depletion temporarily as part of their treatment are merely a small fraction of the total impaired hypothyroid driver population. Investigators with the Framingham Heart Study have reported that 4.4% of individuals above age 60 are hypothyroid. Many of these individuals remain undiagnosed or undertreated. Plus, the noncompliance rate with levothyroxine therapy has been estimated at 17%-32%.
Moreover, once a patient is newly diagnosed as being profoundly hypothyroid and receives a prescription for thyroid hormone replacement, there is a lag time involved in achieving a euthyroid state. The half-life of levothyroxine is 1 week. It takes 6-8 weeks to reach a steady state. Probably at least 2 weeks of therapy are required before there is any improvement in the neurologic impairments documented in this study, Dr. Ain speculated.
"We’re really talking here about a public health problem, one that requires a public health response and acknowledgment that this is a danger," according to the endocrinologist.
Testing during the hypothyroid phase of the study showed significant declines in measures of executive function and information-processing speed. Fine motor performance of the hands was slowed by 13%. Mean scores on the Beck Depression Inventory deteriorated from 7.9 while euthyroid to 18.9 while hypothyroid, consistent with mild bordering on moderate depression; this depression was characterized by vegetative symptoms and altered mood, but without the impaired self-esteem and sense of guilt often characteristic of other forms of depression.
Dr. Ain reported receiving a research grant from Genzyme, which funded this study.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
CHICAGO – Hypothyroid patients exhibit objective cognitive deficits and motor slowing rendering them unsafe to operate a motor vehicle.
That’s the key take-home message from a longitudinal study in which 32 patients with thyroid cancer completed an extensive battery of neurocognitive and psychological tests as well as measured performance on a driving simulator at three time points: while euthyroid, again while temporarily hypothyroid as part of their cancer therapy and assessment, and finally while once again euthyroid after restoration of thyroid hormone therapy.
"These findings provide objective evidence warranting admonitions against operating motor vehicles for hypothyroid patients and confidence in removing such stipulations upon restoration of a euthyroid state," Dr. Kenneth B. Ain said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
In his own clinical practice he has long included a boxed warning against driving while hypothyroid on all of his written instructions to patients. But most physicians don’t warn their hypothyroid patients that they are driving impaired, nor do the joint practice guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists address the issue. That’s largely because there hasn’t been objective, quantitative evidence to provide firm support for such cautionary admonitions – until now, observed Dr. Ain, professor of medicine and director of the thyroid oncology program at the University of Kentucky, Lexington.
While hypothyroid, study participants experienced an 8.5% increase in braking time on a driving simulator. That’s equivalent to the degree of impairment other investigators have shown to be associated with a blood alcohol level of 82 mg/dL, which is above the legal driving limit in the United States.
"Once our study is published, a patient who is involved in an auto accident [in which] there is death or significant harm could be considered an impaired patient. And if physicians do not warn the patient of this risk, they would be considered an agent of harm. They could be liable for the consequences, the same as a neurologist who doesn’t warn a patient with a grand mal seizure disorder not to drive," Dr. Ain said.
In an interview, he noted that thyroid cancer patients undergoing thyroid hormone depletion temporarily as part of their treatment are merely a small fraction of the total impaired hypothyroid driver population. Investigators with the Framingham Heart Study have reported that 4.4% of individuals above age 60 are hypothyroid. Many of these individuals remain undiagnosed or undertreated. Plus, the noncompliance rate with levothyroxine therapy has been estimated at 17%-32%.
Moreover, once a patient is newly diagnosed as being profoundly hypothyroid and receives a prescription for thyroid hormone replacement, there is a lag time involved in achieving a euthyroid state. The half-life of levothyroxine is 1 week. It takes 6-8 weeks to reach a steady state. Probably at least 2 weeks of therapy are required before there is any improvement in the neurologic impairments documented in this study, Dr. Ain speculated.
"We’re really talking here about a public health problem, one that requires a public health response and acknowledgment that this is a danger," according to the endocrinologist.
Testing during the hypothyroid phase of the study showed significant declines in measures of executive function and information-processing speed. Fine motor performance of the hands was slowed by 13%. Mean scores on the Beck Depression Inventory deteriorated from 7.9 while euthyroid to 18.9 while hypothyroid, consistent with mild bordering on moderate depression; this depression was characterized by vegetative symptoms and altered mood, but without the impaired self-esteem and sense of guilt often characteristic of other forms of depression.
Dr. Ain reported receiving a research grant from Genzyme, which funded this study.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
AT ICE/ENDO 2014
Key clinical point: Hypothyroid patients are driving impaired. Physicians need to document, firmly cautioning them to that effect, or face possible liability concerns in the event of a serious motor vehicle accident.
Major finding: Hypothyroid patients showed an increased automobile braking time on a driving simulator that was equivalent to having a blood alcohol level above the U.S. legal driving limit.
Data source: A longitudinal study in which 32 thyroid cancer patients served as their own controls. They completed an extensive neurocognitive test battery and driving simulator performance test while euthyroid, then while temporarily hypothyroid as part of their cancer-treatment regimen, and once again after restoration to the euthyroid state.
Disclosures: Dr. Ain reported receiving a research grant from Genzyme, which funded this study.
Weight loss: Most obese adults aren’t even trying
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society*.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
*Correction, 7/1/2014: An earlier version of this article misstated the name of the International Congress of Endocrinology.
AT ICE/ENDO 2014
Key clinical point: Most obese adults aren’t making any effort to lose weight.
Major finding: Among obese adults who are taking action to lose weight, just 1 in 20 is on prescription weight loss medication or has undergone bariatric surgery. Of that group, 39% indicated they are extremely or very satisfied with their weight loss approach, compared with just 20% of those using other methods, including diet, exercise, OTC drugs, and weight management programs.
Data source: This analysis included nearly 23,000 adults with a body mass index of 30 kg/m2 or more who participated in the 2012 National Health and Wellness Survey.
Disclosures: The survey was conducted by Kantar Health. The analysis was supported by Eisai. The presenter is an Eisai employee.
Adjunctive ketamine boosts acute pain control
DALLAS – Low-dose ketamine as an adjunct to intravenous morphine for moderate to severe pain in the emergency department provides superior relief, compared with morphine alone, according to a randomized, double-blind clinical trial.
Ketamine at subanesthetic doses was an effective analgesic with an opioid-sparing effect. However, this utility must be balanced against an increase in potentially problematic side effects, including dizziness, dysphoria, and sinus tachycardia in some cases, at the higher of the two doses tested, Dr. Francesca L. Beaudoin said at the annual meeting of the Society for Academic Emergency Medicine.
The study was conducted at a single, large, urban, academic ED, where 60 adults presented with pain of less than 7 days’ duration and an average baseline pain score of 9 out of a possible 10 on a numeric rating scale. Study participants were placed on standard of care IV morphine at 0.1 mg/kg and randomized to one of three treatment arms featuring adjunctive placebo, IV ketamine at 0.1 mg/kg, or ketamine at 0.3 mg/kg. Patients’ pain status was assessed at 30, 60, and 120 minutes after administration of their study medication. They received rescue analgesia as needed in order to achieve a 50% reduction in pain, explained Dr. Beaudoin, an emergency medicine physician at Rhode Island Hospital and Brown University, Providence, R.I.
The primary endpoint was pain intensity reduction, as calculated by adding together the differences in pain scores from baseline to each of the three assessment points. Results on this endpoint and related secondary outcome measures were significantly better in the two ketamine groups than in the morphine-only control arm. A clinically meaningful reduction in acute pain scores was achieved in 25% of those treated with 0.1 mg/kg of IV morphine plus placebo, 50% given morphine plus 0.1 mg/kg of IV ketamine, and 70% given morphine plus IV ketamine at 0.3 mg/kg.
Although there were consistent trends favoring greater efficacy for the higher vs. the lower dose of ketamine, these differences didn’t achieve statistical significance, probably because of the small study size, she said.
There was a trend for ketamine at 0.3 mg/kg to provide more durable pain relief. Both ketamine groups had a median 4-point decrease in numeric pain rating scores at 30 and 60 minutes. At 120 minutes, however, the group given lower-dose ketamine had backslid to a median 2.5-point reduction, compared with baseline; the higher-dose group still averaged a 4-point improvement in pain scores. In contrast, patients in the control group had a median 2-point decrease in pain intensity at all three time points.
No clinically important differences in sedation scores were noted between the three treatment groups. Nor were there any differences in time to discharge from the ED, nor in hospital admission rates.
Nine patients in the morphine-alone arm received rescue analgesia, as did four in each of the two adjunctive ketamine groups.
The adverse event profile was noteworthy, she said, and "it’s probably going to be important when we use this study information moving forward." Nine of the 20 patients in the higher-dose ketamine group had dizziness, and five of them had dizziness that lasted up to an hour. Three in the higher-dose ketamine group and two in the lower-dose group had dysphoria, which they regarded as unpleasant. "Two patients in the higher-dose ketamine group actually called the experience ‘scary’ and said they wouldn’t want to do it again," according to Dr. Beaudoin.
Also, three patients on higher-dose ketamine developed sinus tachycardia, with a heart rate of 150 bpm in one case. These events were transient, didn’t require intervention, and resolved spontaneously within 30 minutes. Nevertheless, this must be a consideration in introducing adjunctive low-dose ketamine more broadly into clinical practice, particularly in patients who may have cardiac disease.
The surgical literature provides evidence that low-dose ketamine improves postoperative pain and confers an opioid-sparing effect. The reason the practice hasn’t been adopted into clinical practice in EDs is probably because of the lack of randomized, controlled clinical trials addressing this question – until now, she noted.
"In terms of future directions, I think what’s really interesting is to try to identify what subgroups this is useful for and who it is safe in. That’s where I think this should move," Dr. Beaudoin said.
She reported having no relevant financial conflicts. The study was funded by the University Emergency Medicine Foundation.
DALLAS – Low-dose ketamine as an adjunct to intravenous morphine for moderate to severe pain in the emergency department provides superior relief, compared with morphine alone, according to a randomized, double-blind clinical trial.
Ketamine at subanesthetic doses was an effective analgesic with an opioid-sparing effect. However, this utility must be balanced against an increase in potentially problematic side effects, including dizziness, dysphoria, and sinus tachycardia in some cases, at the higher of the two doses tested, Dr. Francesca L. Beaudoin said at the annual meeting of the Society for Academic Emergency Medicine.

The study was conducted at a single, large, urban, academic ED, where 60 adults presented with pain of less than 7 days’ duration and an average baseline pain score of 9 out of a possible 10 on a numeric rating scale. Study participants were placed on standard of care IV morphine at 0.1 mg/kg and randomized to one of three treatment arms featuring adjunctive placebo, IV ketamine at 0.1 mg/kg, or ketamine at 0.3 mg/kg. Patients’ pain status was assessed at 30, 60, and 120 minutes after administration of their study medication. They received rescue analgesia as needed in order to achieve a 50% reduction in pain, explained Dr. Beaudoin, an emergency medicine physician at Rhode Island Hospital and Brown University, Providence, R.I.
The primary endpoint was pain intensity reduction, as calculated by adding together the differences in pain scores from baseline to each of the three assessment points. Results on this endpoint and related secondary outcome measures were significantly better in the two ketamine groups than in the morphine-only control arm. A clinically meaningful reduction in acute pain scores was achieved in 25% of those treated with 0.1 mg/kg of IV morphine plus placebo, 50% given morphine plus 0.1 mg/kg of IV ketamine, and 70% given morphine plus IV ketamine at 0.3 mg/kg.
Although there were consistent trends favoring greater efficacy for the higher vs. the lower dose of ketamine, these differences didn’t achieve statistical significance, probably because of the small study size, she said.
There was a trend for ketamine at 0.3 mg/kg to provide more durable pain relief. Both ketamine groups had a median 4-point decrease in numeric pain rating scores at 30 and 60 minutes. At 120 minutes, however, the group given lower-dose ketamine had backslid to a median 2.5-point reduction, compared with baseline; the higher-dose group still averaged a 4-point improvement in pain scores. In contrast, patients in the control group had a median 2-point decrease in pain intensity at all three time points.
No clinically important differences in sedation scores were noted between the three treatment groups. Nor were there any differences in time to discharge from the ED, nor in hospital admission rates.
Nine patients in the morphine-alone arm received rescue analgesia, as did four in each of the two adjunctive ketamine groups.
The adverse event profile was noteworthy, she said, and "it’s probably going to be important when we use this study information moving forward." Nine of the 20 patients in the higher-dose ketamine group had dizziness, and five of them had dizziness that lasted up to an hour. Three in the higher-dose ketamine group and two in the lower-dose group had dysphoria, which they regarded as unpleasant. "Two patients in the higher-dose ketamine group actually called the experience ‘scary’ and said they wouldn’t want to do it again," according to Dr. Beaudoin.
Also, three patients on higher-dose ketamine developed sinus tachycardia, with a heart rate of 150 bpm in one case. These events were transient, didn’t require intervention, and resolved spontaneously within 30 minutes. Nevertheless, this must be a consideration in introducing adjunctive low-dose ketamine more broadly into clinical practice, particularly in patients who may have cardiac disease.
The surgical literature provides evidence that low-dose ketamine improves postoperative pain and confers an opioid-sparing effect. The reason the practice hasn’t been adopted into clinical practice in EDs is probably because of the lack of randomized, controlled clinical trials addressing this question – until now, she noted.
"In terms of future directions, I think what’s really interesting is to try to identify what subgroups this is useful for and who it is safe in. That’s where I think this should move," Dr. Beaudoin said.
She reported having no relevant financial conflicts. The study was funded by the University Emergency Medicine Foundation.
DALLAS – Low-dose ketamine as an adjunct to intravenous morphine for moderate to severe pain in the emergency department provides superior relief, compared with morphine alone, according to a randomized, double-blind clinical trial.
Ketamine at subanesthetic doses was an effective analgesic with an opioid-sparing effect. However, this utility must be balanced against an increase in potentially problematic side effects, including dizziness, dysphoria, and sinus tachycardia in some cases, at the higher of the two doses tested, Dr. Francesca L. Beaudoin said at the annual meeting of the Society for Academic Emergency Medicine.

The study was conducted at a single, large, urban, academic ED, where 60 adults presented with pain of less than 7 days’ duration and an average baseline pain score of 9 out of a possible 10 on a numeric rating scale. Study participants were placed on standard of care IV morphine at 0.1 mg/kg and randomized to one of three treatment arms featuring adjunctive placebo, IV ketamine at 0.1 mg/kg, or ketamine at 0.3 mg/kg. Patients’ pain status was assessed at 30, 60, and 120 minutes after administration of their study medication. They received rescue analgesia as needed in order to achieve a 50% reduction in pain, explained Dr. Beaudoin, an emergency medicine physician at Rhode Island Hospital and Brown University, Providence, R.I.
The primary endpoint was pain intensity reduction, as calculated by adding together the differences in pain scores from baseline to each of the three assessment points. Results on this endpoint and related secondary outcome measures were significantly better in the two ketamine groups than in the morphine-only control arm. A clinically meaningful reduction in acute pain scores was achieved in 25% of those treated with 0.1 mg/kg of IV morphine plus placebo, 50% given morphine plus 0.1 mg/kg of IV ketamine, and 70% given morphine plus IV ketamine at 0.3 mg/kg.
Although there were consistent trends favoring greater efficacy for the higher vs. the lower dose of ketamine, these differences didn’t achieve statistical significance, probably because of the small study size, she said.
There was a trend for ketamine at 0.3 mg/kg to provide more durable pain relief. Both ketamine groups had a median 4-point decrease in numeric pain rating scores at 30 and 60 minutes. At 120 minutes, however, the group given lower-dose ketamine had backslid to a median 2.5-point reduction, compared with baseline; the higher-dose group still averaged a 4-point improvement in pain scores. In contrast, patients in the control group had a median 2-point decrease in pain intensity at all three time points.
No clinically important differences in sedation scores were noted between the three treatment groups. Nor were there any differences in time to discharge from the ED, nor in hospital admission rates.
Nine patients in the morphine-alone arm received rescue analgesia, as did four in each of the two adjunctive ketamine groups.
The adverse event profile was noteworthy, she said, and "it’s probably going to be important when we use this study information moving forward." Nine of the 20 patients in the higher-dose ketamine group had dizziness, and five of them had dizziness that lasted up to an hour. Three in the higher-dose ketamine group and two in the lower-dose group had dysphoria, which they regarded as unpleasant. "Two patients in the higher-dose ketamine group actually called the experience ‘scary’ and said they wouldn’t want to do it again," according to Dr. Beaudoin.
Also, three patients on higher-dose ketamine developed sinus tachycardia, with a heart rate of 150 bpm in one case. These events were transient, didn’t require intervention, and resolved spontaneously within 30 minutes. Nevertheless, this must be a consideration in introducing adjunctive low-dose ketamine more broadly into clinical practice, particularly in patients who may have cardiac disease.
The surgical literature provides evidence that low-dose ketamine improves postoperative pain and confers an opioid-sparing effect. The reason the practice hasn’t been adopted into clinical practice in EDs is probably because of the lack of randomized, controlled clinical trials addressing this question – until now, she noted.
"In terms of future directions, I think what’s really interesting is to try to identify what subgroups this is useful for and who it is safe in. That’s where I think this should move," Dr. Beaudoin said.
She reported having no relevant financial conflicts. The study was funded by the University Emergency Medicine Foundation.
AT SAEM 2014
Major finding: A clinically meaningful reduction in acute pain scores was achieved in 25% of those treated with 0.1 mg/kg of IV morphine plus placebo, 50% given morphine plus 0.1 mg/kg of IV ketamine, and 70% given morphine plus IV ketamine at 0.3 mg/kg.
Data source: A single-center, randomized, double-blind clinical trial that involved 60 patients who presented to the ED with moderate to severe acute pain.
Disclosures: The study was supported by the University Emergency Medicine Foundation. The presenter reported having no financial conflicts.
Polymyxin use generates flood of acute kidney injuries
LAS VEGAS – Polymyxins are back on the scene due to the worrisome increase in multidrug-resistant gram-negative bacterial infections.
"These drugs were taken off the shelf many years ago because of their severe nephrotoxicity, but they’ve entered the arena again because they’re very effective – and their toxicity has reemerged as well," Dr. Mark A. Perazella warned at a meeting sponsored by the National Kidney Foundation.
Drug-induced renal injury accounts for 18%-27% of all acute kidney injury in hospital cases. Antimicrobial agents are among the top causes of drug-related nephropathy, particularly in the hospitalized population, he added.
The incidence of acute kidney injury associated with polymyxin B and polymyxin E (colistin) in contemporary practice is a whopping 30%-60%, depending upon the cumulative dose and duration of therapy, comorbid conditions, and whether other nephrotoxic medications are being used concomitantly. Often an individual’s risk of acute kidney injury is at the high end of this range because the polymyxins are viewed as agents of last resort, and vancomycin – a nephrotoxin in its own right, albeit a less potent one – is commonly on board.
Plus, patients with a serious multidrug-resistant gram-negative infection often have multiple comorbidities and are in a weakened state, further predisposing them to acute kidney injury upon exposure to a nephrotoxin, added Dr. Perazella, professor of medicine and director of the acute dialysis unit at Yale University in New Haven, Conn.
There are no randomized trial data to define polymyxin dosing parameters that maximize efficacy and minimize renal risk, which is clearly dose and duration dependent. For example, in a retrospective study of 173 adults on polymyxin B, the incidence of acute kidney injury was 60%; 10% required dialysis, and 14% had to discontinue treatment because of nephrotoxicity. The median cumulative dose of polymyxin B in patients with acute kidney injury was 1,578 mg, as compared with 800 mg in those without acute kidney injury. Concomitant vancomycin was used by 82% of patients with and by 55% without acute kidney injury (J. Infect. 2012;65:80-7).
The reported incidence of acute kidney injury in patients on vancomycin is most often in the 10%-20% range. Risk is higher in patients with multiple comorbidities and increases with higher cumulative doses and longer-duration therapy. In an effort to improve cure rates, the target vancomycin trough level has increase from 10-20 mg/dL to 15-20 mg/dL. That higher trough level means more cases of acute kidney injury, according to Dr. Perazella.
The randomized prospective ZEPHYR study highlighted fixed-dose linezolid as an attractive alternative to dose-optimized vancomycin for treatment of hospital-acquired methicillin-resistant Staphylococcus aureus pneumonia. In a randomized study of 165 participants, the clinical cure rate was significantly better with linezolid by a margin of 58%-47%. The incidence of nephrotoxicity was 8% in the linezolid group, compared with 18% with vancomycin.
Dr. Perazella reported having no financial conflicts.
LAS VEGAS – Polymyxins are back on the scene due to the worrisome increase in multidrug-resistant gram-negative bacterial infections.
"These drugs were taken off the shelf many years ago because of their severe nephrotoxicity, but they’ve entered the arena again because they’re very effective – and their toxicity has reemerged as well," Dr. Mark A. Perazella warned at a meeting sponsored by the National Kidney Foundation.
Drug-induced renal injury accounts for 18%-27% of all acute kidney injury in hospital cases. Antimicrobial agents are among the top causes of drug-related nephropathy, particularly in the hospitalized population, he added.
The incidence of acute kidney injury associated with polymyxin B and polymyxin E (colistin) in contemporary practice is a whopping 30%-60%, depending upon the cumulative dose and duration of therapy, comorbid conditions, and whether other nephrotoxic medications are being used concomitantly. Often an individual’s risk of acute kidney injury is at the high end of this range because the polymyxins are viewed as agents of last resort, and vancomycin – a nephrotoxin in its own right, albeit a less potent one – is commonly on board.
Plus, patients with a serious multidrug-resistant gram-negative infection often have multiple comorbidities and are in a weakened state, further predisposing them to acute kidney injury upon exposure to a nephrotoxin, added Dr. Perazella, professor of medicine and director of the acute dialysis unit at Yale University in New Haven, Conn.
There are no randomized trial data to define polymyxin dosing parameters that maximize efficacy and minimize renal risk, which is clearly dose and duration dependent. For example, in a retrospective study of 173 adults on polymyxin B, the incidence of acute kidney injury was 60%; 10% required dialysis, and 14% had to discontinue treatment because of nephrotoxicity. The median cumulative dose of polymyxin B in patients with acute kidney injury was 1,578 mg, as compared with 800 mg in those without acute kidney injury. Concomitant vancomycin was used by 82% of patients with and by 55% without acute kidney injury (J. Infect. 2012;65:80-7).
The reported incidence of acute kidney injury in patients on vancomycin is most often in the 10%-20% range. Risk is higher in patients with multiple comorbidities and increases with higher cumulative doses and longer-duration therapy. In an effort to improve cure rates, the target vancomycin trough level has increase from 10-20 mg/dL to 15-20 mg/dL. That higher trough level means more cases of acute kidney injury, according to Dr. Perazella.
The randomized prospective ZEPHYR study highlighted fixed-dose linezolid as an attractive alternative to dose-optimized vancomycin for treatment of hospital-acquired methicillin-resistant Staphylococcus aureus pneumonia. In a randomized study of 165 participants, the clinical cure rate was significantly better with linezolid by a margin of 58%-47%. The incidence of nephrotoxicity was 8% in the linezolid group, compared with 18% with vancomycin.
Dr. Perazella reported having no financial conflicts.
LAS VEGAS – Polymyxins are back on the scene due to the worrisome increase in multidrug-resistant gram-negative bacterial infections.
"These drugs were taken off the shelf many years ago because of their severe nephrotoxicity, but they’ve entered the arena again because they’re very effective – and their toxicity has reemerged as well," Dr. Mark A. Perazella warned at a meeting sponsored by the National Kidney Foundation.
Drug-induced renal injury accounts for 18%-27% of all acute kidney injury in hospital cases. Antimicrobial agents are among the top causes of drug-related nephropathy, particularly in the hospitalized population, he added.
The incidence of acute kidney injury associated with polymyxin B and polymyxin E (colistin) in contemporary practice is a whopping 30%-60%, depending upon the cumulative dose and duration of therapy, comorbid conditions, and whether other nephrotoxic medications are being used concomitantly. Often an individual’s risk of acute kidney injury is at the high end of this range because the polymyxins are viewed as agents of last resort, and vancomycin – a nephrotoxin in its own right, albeit a less potent one – is commonly on board.
Plus, patients with a serious multidrug-resistant gram-negative infection often have multiple comorbidities and are in a weakened state, further predisposing them to acute kidney injury upon exposure to a nephrotoxin, added Dr. Perazella, professor of medicine and director of the acute dialysis unit at Yale University in New Haven, Conn.
There are no randomized trial data to define polymyxin dosing parameters that maximize efficacy and minimize renal risk, which is clearly dose and duration dependent. For example, in a retrospective study of 173 adults on polymyxin B, the incidence of acute kidney injury was 60%; 10% required dialysis, and 14% had to discontinue treatment because of nephrotoxicity. The median cumulative dose of polymyxin B in patients with acute kidney injury was 1,578 mg, as compared with 800 mg in those without acute kidney injury. Concomitant vancomycin was used by 82% of patients with and by 55% without acute kidney injury (J. Infect. 2012;65:80-7).
The reported incidence of acute kidney injury in patients on vancomycin is most often in the 10%-20% range. Risk is higher in patients with multiple comorbidities and increases with higher cumulative doses and longer-duration therapy. In an effort to improve cure rates, the target vancomycin trough level has increase from 10-20 mg/dL to 15-20 mg/dL. That higher trough level means more cases of acute kidney injury, according to Dr. Perazella.
The randomized prospective ZEPHYR study highlighted fixed-dose linezolid as an attractive alternative to dose-optimized vancomycin for treatment of hospital-acquired methicillin-resistant Staphylococcus aureus pneumonia. In a randomized study of 165 participants, the clinical cure rate was significantly better with linezolid by a margin of 58%-47%. The incidence of nephrotoxicity was 8% in the linezolid group, compared with 18% with vancomycin.
Dr. Perazella reported having no financial conflicts.
EXPERT ANALYSIS FROM SCM 14
Study: Excedrin ‘good choice’ as first-line therapy for migraine in ED
DALLAS – Acetaminophen, aspirin, and caffeine in combination – the tablet familiarly known as Excedrin – proved an attractive alternative to intravenous prochlorperazine as first-line therapy for uncomplicated acute migraine in the emergency department, in a randomized, prospective, double-blind clinical trial.
"Excedrin worked as well as prochlorperazine in this study and doesn’t require an IV. I think it’s a good choice for acute cephalgia without significant nausea and vomiting," Dr. Kenneth R. Deitch said at the annual meeting of the Society for Academic Emergency Medicine.
In this 71-patient study, both therapies provided significant and comparable pain relief at 60 minutes. Restlessness and/or muscle spasms occurred within the first 120 minutes in 10 patients in the prochlorperazine group and in 3 on acetaminophen, aspirin, and caffeine (AAC). At follow-up 24 hours later, roughly 90% of patients in each study arm said they would use their study medication again, according to Dr. Deitch, an emergency medicine physician at Einstein Medical Center, Philadelphia.
All study participants met International Headache Society criteria for migraine or probable migraine. They were randomized to receive 10 mg of prochlorperazine IV in a 2-minute push plus placebo, or two generic AAC tablets, each containing 250 mg of acetaminophen, 250 mg of aspirin, and 65 mg of caffeine, along with a placebo IV.
The primary endpoint was reduction in pain on a 0-100 visual analog scale at 60 minutes post treatment. The mean reduction was 47 points in the prochlorperazine group and 37 points with AAC; the difference was not statistically significant.
If patients reported inadequate pain relief after 60 minutes, the study code was broken and physicians prescribed a rescue medication of their choosing – most often an opiate. Ten patients in each study arm required the use of rescue medication after 60 minutes.
The study initially included an additional 10 patients – 6 in the prochlorperazine arm and 4 in the AAC arm – who demanded opiates or other drugs before the 60-minute mark. Those patients were excluded from the analysis.
Dr. Deitch noted that migraine or presumed migraine annually accounts for several million ED visits. Guidelines vary for treatment of migraine in the ED and don’t rely on a strong evidence base. Oral narcotics are the most frequently prescribed agents, but the International Headache Society does not recommend them as first-line therapy because of their major drawbacks, which include nausea, constipation, and the potential for abuse and overuse. Other guidelines advocate triptans, but they’re not popular with emergency medicine physicians because of the cardiovascular risk profile.
Prochlorperazine – the most commonly prescribed dopamine agonist for acute migraine in the ED – does not have a Food and Drug Administration indication for migraine. The drug’s off-label usage in this setting is well established, however. Randomized trials have demonstrated that prochlorperazine is as effective as are narcotics and may be more effective than sumatriptan. Akathisia and dystonia are common side effects with prochlorperazine, and those adverse effects are often sufficiently severe to require rescue diphenhydramine, which prolongs the ED stay.
AAC (Excedrin) is the only OTC medication with FDA approval for the treatment of migraine. Its side effect profile is better than that of prochlorperazine, yet AAC is rarely used as first-line therapy for acute migraine in the ED. In part, the evidence was lacking. But even with demonstrated efficacy in a randomized head-to-head comparison with prochlorperazine, "Excedrin is an oral medication. Physicians and patients have an expectation that if a headache is bad enough to bring someone into the ED, the patient expects an IV, and we expect to do that for them."
With fairly good evidence of efficacy even in patients with mild to moderate migraine with nausea, the situation may begin to change as the majority of patients who receive Excedrin have pain relief, he said.
Dr. Deitch reported having no financial conflicts of interest with regard to this study, conducted free of commercial support.
DALLAS – Acetaminophen, aspirin, and caffeine in combination – the tablet familiarly known as Excedrin – proved an attractive alternative to intravenous prochlorperazine as first-line therapy for uncomplicated acute migraine in the emergency department, in a randomized, prospective, double-blind clinical trial.
"Excedrin worked as well as prochlorperazine in this study and doesn’t require an IV. I think it’s a good choice for acute cephalgia without significant nausea and vomiting," Dr. Kenneth R. Deitch said at the annual meeting of the Society for Academic Emergency Medicine.
In this 71-patient study, both therapies provided significant and comparable pain relief at 60 minutes. Restlessness and/or muscle spasms occurred within the first 120 minutes in 10 patients in the prochlorperazine group and in 3 on acetaminophen, aspirin, and caffeine (AAC). At follow-up 24 hours later, roughly 90% of patients in each study arm said they would use their study medication again, according to Dr. Deitch, an emergency medicine physician at Einstein Medical Center, Philadelphia.
All study participants met International Headache Society criteria for migraine or probable migraine. They were randomized to receive 10 mg of prochlorperazine IV in a 2-minute push plus placebo, or two generic AAC tablets, each containing 250 mg of acetaminophen, 250 mg of aspirin, and 65 mg of caffeine, along with a placebo IV.
The primary endpoint was reduction in pain on a 0-100 visual analog scale at 60 minutes post treatment. The mean reduction was 47 points in the prochlorperazine group and 37 points with AAC; the difference was not statistically significant.
If patients reported inadequate pain relief after 60 minutes, the study code was broken and physicians prescribed a rescue medication of their choosing – most often an opiate. Ten patients in each study arm required the use of rescue medication after 60 minutes.
The study initially included an additional 10 patients – 6 in the prochlorperazine arm and 4 in the AAC arm – who demanded opiates or other drugs before the 60-minute mark. Those patients were excluded from the analysis.
Dr. Deitch noted that migraine or presumed migraine annually accounts for several million ED visits. Guidelines vary for treatment of migraine in the ED and don’t rely on a strong evidence base. Oral narcotics are the most frequently prescribed agents, but the International Headache Society does not recommend them as first-line therapy because of their major drawbacks, which include nausea, constipation, and the potential for abuse and overuse. Other guidelines advocate triptans, but they’re not popular with emergency medicine physicians because of the cardiovascular risk profile.
Prochlorperazine – the most commonly prescribed dopamine agonist for acute migraine in the ED – does not have a Food and Drug Administration indication for migraine. The drug’s off-label usage in this setting is well established, however. Randomized trials have demonstrated that prochlorperazine is as effective as are narcotics and may be more effective than sumatriptan. Akathisia and dystonia are common side effects with prochlorperazine, and those adverse effects are often sufficiently severe to require rescue diphenhydramine, which prolongs the ED stay.
AAC (Excedrin) is the only OTC medication with FDA approval for the treatment of migraine. Its side effect profile is better than that of prochlorperazine, yet AAC is rarely used as first-line therapy for acute migraine in the ED. In part, the evidence was lacking. But even with demonstrated efficacy in a randomized head-to-head comparison with prochlorperazine, "Excedrin is an oral medication. Physicians and patients have an expectation that if a headache is bad enough to bring someone into the ED, the patient expects an IV, and we expect to do that for them."
With fairly good evidence of efficacy even in patients with mild to moderate migraine with nausea, the situation may begin to change as the majority of patients who receive Excedrin have pain relief, he said.
Dr. Deitch reported having no financial conflicts of interest with regard to this study, conducted free of commercial support.
DALLAS – Acetaminophen, aspirin, and caffeine in combination – the tablet familiarly known as Excedrin – proved an attractive alternative to intravenous prochlorperazine as first-line therapy for uncomplicated acute migraine in the emergency department, in a randomized, prospective, double-blind clinical trial.
"Excedrin worked as well as prochlorperazine in this study and doesn’t require an IV. I think it’s a good choice for acute cephalgia without significant nausea and vomiting," Dr. Kenneth R. Deitch said at the annual meeting of the Society for Academic Emergency Medicine.
In this 71-patient study, both therapies provided significant and comparable pain relief at 60 minutes. Restlessness and/or muscle spasms occurred within the first 120 minutes in 10 patients in the prochlorperazine group and in 3 on acetaminophen, aspirin, and caffeine (AAC). At follow-up 24 hours later, roughly 90% of patients in each study arm said they would use their study medication again, according to Dr. Deitch, an emergency medicine physician at Einstein Medical Center, Philadelphia.
All study participants met International Headache Society criteria for migraine or probable migraine. They were randomized to receive 10 mg of prochlorperazine IV in a 2-minute push plus placebo, or two generic AAC tablets, each containing 250 mg of acetaminophen, 250 mg of aspirin, and 65 mg of caffeine, along with a placebo IV.
The primary endpoint was reduction in pain on a 0-100 visual analog scale at 60 minutes post treatment. The mean reduction was 47 points in the prochlorperazine group and 37 points with AAC; the difference was not statistically significant.
If patients reported inadequate pain relief after 60 minutes, the study code was broken and physicians prescribed a rescue medication of their choosing – most often an opiate. Ten patients in each study arm required the use of rescue medication after 60 minutes.
The study initially included an additional 10 patients – 6 in the prochlorperazine arm and 4 in the AAC arm – who demanded opiates or other drugs before the 60-minute mark. Those patients were excluded from the analysis.
Dr. Deitch noted that migraine or presumed migraine annually accounts for several million ED visits. Guidelines vary for treatment of migraine in the ED and don’t rely on a strong evidence base. Oral narcotics are the most frequently prescribed agents, but the International Headache Society does not recommend them as first-line therapy because of their major drawbacks, which include nausea, constipation, and the potential for abuse and overuse. Other guidelines advocate triptans, but they’re not popular with emergency medicine physicians because of the cardiovascular risk profile.
Prochlorperazine – the most commonly prescribed dopamine agonist for acute migraine in the ED – does not have a Food and Drug Administration indication for migraine. The drug’s off-label usage in this setting is well established, however. Randomized trials have demonstrated that prochlorperazine is as effective as are narcotics and may be more effective than sumatriptan. Akathisia and dystonia are common side effects with prochlorperazine, and those adverse effects are often sufficiently severe to require rescue diphenhydramine, which prolongs the ED stay.
AAC (Excedrin) is the only OTC medication with FDA approval for the treatment of migraine. Its side effect profile is better than that of prochlorperazine, yet AAC is rarely used as first-line therapy for acute migraine in the ED. In part, the evidence was lacking. But even with demonstrated efficacy in a randomized head-to-head comparison with prochlorperazine, "Excedrin is an oral medication. Physicians and patients have an expectation that if a headache is bad enough to bring someone into the ED, the patient expects an IV, and we expect to do that for them."
With fairly good evidence of efficacy even in patients with mild to moderate migraine with nausea, the situation may begin to change as the majority of patients who receive Excedrin have pain relief, he said.
Dr. Deitch reported having no financial conflicts of interest with regard to this study, conducted free of commercial support.
AT SAEM 2014
Major finding: Patients who present to the ED with acute migraine experience comparable pain relief at 60 minutes regardless of whether their treatment consists of Excedrin or the more cumbersome intravenous prochlorperazine.
Data source: A single-center, prospective, double-blind randomized trial of 71 ED patients with acute migraine or presumed migraine.
Disclosures: The presenter reported having no financial conflicts regarding this study, which was free of commercial support.