User login
New agent builds bone bigger, faster
CHICAGO – Abaloparatide, a synthetic analog of human parathyroid hormone–related peptide, displayed jaw-dropping superiority to teriparatide in boosting bone mineral density at multiple anatomic sites in a head-to-head, placebo-controlled phase II study.
"Given the consistency of these increases in BMD [bone mineral density] seen in the phase II studies, abaloparatide may emerge as an important therapeutic agent in the treatment of postmenopausal osteoporosis," Dr. Alan G. Harris observed in presenting the results of two separate phase II abaloparatide studies at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"There is an unmet need for anabolic agents that preferentially increase bone formation as opposed to decreasing bone resorption. There is also a challenge we’re faced with in clinical practice: that is, the lack of early hip BMD increase with teriparatide," added Dr. Harris, chief medical officer at Radius Health of Cambridge, Mass., which is developing the agent.
The phase II data suggest abaloparatide at 80 mcg by once-daily subcutaneous injection meets both needs, he added.
Indeed, based upon the highly positive phase II work, a phase III, placebo- and teriparatide-controlled clinical trial with fracture endpoints is well underway. The 18-month trial involving more than 2,400 patients is due to be completed later this year.
Separately, Gary Hattersley, Ph.D., presented encouraging results from a 231-patient, 24-week, phase-II, dose-ranging study of abaloparatide delivered by transdermal patch.
"We look at this as being a strong proof-of-concept study demonstrating that this simple transdermal patch with only a 5-minute wear time is able to deliver meaningful amounts of abaloparatide through the skin without the need for a subcutaneous injection in order to achieve meaningful increases in BMD," said Dr. Hattersley, chief scientific officer at Radius Health.
"We recognize that there’s really a significant opportunity for an alternative to daily subcutaneous injection. This has the potential to improve both patient convenience as well as patient compliance," he added.
The increases in BMD with the patch – a 2.95% increase from baseline at the spine with the 150-mcg patch and a 1.49% rise in total hip BMD – were not as robust as in controls assigned to once-daily abaloparatide at 80 mcg, which is the optimal injectable dose also being used in the ongoing phase III trial. But Dr. Hattersley said he believes that higher-dose patches now under study will achieve substantially bigger increases in BMD.
Dr. Harris presented data from two phase II studies on a total of 472 postmenopausal women with osteoporosis. In one, subjects were randomized to subcutaneous abaloparatide, teriparatide (Forteo) at its approved dose of 20 mcg by daily subcutaneous injection, or placebo. Although the primary endpoints in this study were assessed at 24 weeks, in an extension out to 48 weeks the increase in lumbar spine BMD over baseline was 12.9% with abaloparatide 80 mcg, 8.6% with teriparatide, and 0.7% with placebo.
In the other study, patients were randomized to abaloparatide or placebo. In this trial, patients on abaloparatide at 80 mcg showed a 5.8% increase over baseline in spine BMD at 24 weeks, along with a 2.74% increase in total hip BMD and a 2.76% increase in femoral neck BMD, as compared with a 0.44% increase in spine BMD with placebo and net BMD losses of less than 1% at each of the other two sites.
In both studies, side effects of abaloparatide were similar in type and incidence to placebo. Of note, the incidence of mild, transient hypercalcemia in abaloparatide-treated patients was half that of the teriparatide group.
Asked why the subcutaneous abaloparatide at 80 mcg is so much more effective at increasing BMD than teriparatide is at its approved dose, Dr. Harris replied, "They’re different peptides." In monkey studies, abaloparatide showed less increase in cortical bone porosity than in studies done using teriparatide. And in the head-to-head phase II study, the increase in bone turnover markers related to resorption was substantially greater with teriparatide. So abaloparatide’s greater BMD-building efficacy is because of greater selectivity for increased bone formation and less bone resorption relative to teriparatide, he suggested.
The abaloparatide patch utilizes proprietary technology developed by 3M. The dime-size patch contains 316 spearlike microprojections, each 500 mcm (micrometers) long. The tip of each microprojection is coated with abaloparatide. When the patch is applied to periumbilical skin, the microprojections penetrate the skin to a depth of about 250 mcm, putting the tip into the upper dermis. Patch application was painless and without side effects in the phase II study, according to Dr. Harris.
These phase II studies were funded by Radius Health.
CHICAGO – Abaloparatide, a synthetic analog of human parathyroid hormone–related peptide, displayed jaw-dropping superiority to teriparatide in boosting bone mineral density at multiple anatomic sites in a head-to-head, placebo-controlled phase II study.
"Given the consistency of these increases in BMD [bone mineral density] seen in the phase II studies, abaloparatide may emerge as an important therapeutic agent in the treatment of postmenopausal osteoporosis," Dr. Alan G. Harris observed in presenting the results of two separate phase II abaloparatide studies at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"There is an unmet need for anabolic agents that preferentially increase bone formation as opposed to decreasing bone resorption. There is also a challenge we’re faced with in clinical practice: that is, the lack of early hip BMD increase with teriparatide," added Dr. Harris, chief medical officer at Radius Health of Cambridge, Mass., which is developing the agent.
The phase II data suggest abaloparatide at 80 mcg by once-daily subcutaneous injection meets both needs, he added.
Indeed, based upon the highly positive phase II work, a phase III, placebo- and teriparatide-controlled clinical trial with fracture endpoints is well underway. The 18-month trial involving more than 2,400 patients is due to be completed later this year.
Separately, Gary Hattersley, Ph.D., presented encouraging results from a 231-patient, 24-week, phase-II, dose-ranging study of abaloparatide delivered by transdermal patch.
"We look at this as being a strong proof-of-concept study demonstrating that this simple transdermal patch with only a 5-minute wear time is able to deliver meaningful amounts of abaloparatide through the skin without the need for a subcutaneous injection in order to achieve meaningful increases in BMD," said Dr. Hattersley, chief scientific officer at Radius Health.
"We recognize that there’s really a significant opportunity for an alternative to daily subcutaneous injection. This has the potential to improve both patient convenience as well as patient compliance," he added.
The increases in BMD with the patch – a 2.95% increase from baseline at the spine with the 150-mcg patch and a 1.49% rise in total hip BMD – were not as robust as in controls assigned to once-daily abaloparatide at 80 mcg, which is the optimal injectable dose also being used in the ongoing phase III trial. But Dr. Hattersley said he believes that higher-dose patches now under study will achieve substantially bigger increases in BMD.
Dr. Harris presented data from two phase II studies on a total of 472 postmenopausal women with osteoporosis. In one, subjects were randomized to subcutaneous abaloparatide, teriparatide (Forteo) at its approved dose of 20 mcg by daily subcutaneous injection, or placebo. Although the primary endpoints in this study were assessed at 24 weeks, in an extension out to 48 weeks the increase in lumbar spine BMD over baseline was 12.9% with abaloparatide 80 mcg, 8.6% with teriparatide, and 0.7% with placebo.
In the other study, patients were randomized to abaloparatide or placebo. In this trial, patients on abaloparatide at 80 mcg showed a 5.8% increase over baseline in spine BMD at 24 weeks, along with a 2.74% increase in total hip BMD and a 2.76% increase in femoral neck BMD, as compared with a 0.44% increase in spine BMD with placebo and net BMD losses of less than 1% at each of the other two sites.
In both studies, side effects of abaloparatide were similar in type and incidence to placebo. Of note, the incidence of mild, transient hypercalcemia in abaloparatide-treated patients was half that of the teriparatide group.
Asked why the subcutaneous abaloparatide at 80 mcg is so much more effective at increasing BMD than teriparatide is at its approved dose, Dr. Harris replied, "They’re different peptides." In monkey studies, abaloparatide showed less increase in cortical bone porosity than in studies done using teriparatide. And in the head-to-head phase II study, the increase in bone turnover markers related to resorption was substantially greater with teriparatide. So abaloparatide’s greater BMD-building efficacy is because of greater selectivity for increased bone formation and less bone resorption relative to teriparatide, he suggested.
The abaloparatide patch utilizes proprietary technology developed by 3M. The dime-size patch contains 316 spearlike microprojections, each 500 mcm (micrometers) long. The tip of each microprojection is coated with abaloparatide. When the patch is applied to periumbilical skin, the microprojections penetrate the skin to a depth of about 250 mcm, putting the tip into the upper dermis. Patch application was painless and without side effects in the phase II study, according to Dr. Harris.
These phase II studies were funded by Radius Health.
CHICAGO – Abaloparatide, a synthetic analog of human parathyroid hormone–related peptide, displayed jaw-dropping superiority to teriparatide in boosting bone mineral density at multiple anatomic sites in a head-to-head, placebo-controlled phase II study.
"Given the consistency of these increases in BMD [bone mineral density] seen in the phase II studies, abaloparatide may emerge as an important therapeutic agent in the treatment of postmenopausal osteoporosis," Dr. Alan G. Harris observed in presenting the results of two separate phase II abaloparatide studies at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"There is an unmet need for anabolic agents that preferentially increase bone formation as opposed to decreasing bone resorption. There is also a challenge we’re faced with in clinical practice: that is, the lack of early hip BMD increase with teriparatide," added Dr. Harris, chief medical officer at Radius Health of Cambridge, Mass., which is developing the agent.
The phase II data suggest abaloparatide at 80 mcg by once-daily subcutaneous injection meets both needs, he added.
Indeed, based upon the highly positive phase II work, a phase III, placebo- and teriparatide-controlled clinical trial with fracture endpoints is well underway. The 18-month trial involving more than 2,400 patients is due to be completed later this year.
Separately, Gary Hattersley, Ph.D., presented encouraging results from a 231-patient, 24-week, phase-II, dose-ranging study of abaloparatide delivered by transdermal patch.
"We look at this as being a strong proof-of-concept study demonstrating that this simple transdermal patch with only a 5-minute wear time is able to deliver meaningful amounts of abaloparatide through the skin without the need for a subcutaneous injection in order to achieve meaningful increases in BMD," said Dr. Hattersley, chief scientific officer at Radius Health.
"We recognize that there’s really a significant opportunity for an alternative to daily subcutaneous injection. This has the potential to improve both patient convenience as well as patient compliance," he added.
The increases in BMD with the patch – a 2.95% increase from baseline at the spine with the 150-mcg patch and a 1.49% rise in total hip BMD – were not as robust as in controls assigned to once-daily abaloparatide at 80 mcg, which is the optimal injectable dose also being used in the ongoing phase III trial. But Dr. Hattersley said he believes that higher-dose patches now under study will achieve substantially bigger increases in BMD.
Dr. Harris presented data from two phase II studies on a total of 472 postmenopausal women with osteoporosis. In one, subjects were randomized to subcutaneous abaloparatide, teriparatide (Forteo) at its approved dose of 20 mcg by daily subcutaneous injection, or placebo. Although the primary endpoints in this study were assessed at 24 weeks, in an extension out to 48 weeks the increase in lumbar spine BMD over baseline was 12.9% with abaloparatide 80 mcg, 8.6% with teriparatide, and 0.7% with placebo.
In the other study, patients were randomized to abaloparatide or placebo. In this trial, patients on abaloparatide at 80 mcg showed a 5.8% increase over baseline in spine BMD at 24 weeks, along with a 2.74% increase in total hip BMD and a 2.76% increase in femoral neck BMD, as compared with a 0.44% increase in spine BMD with placebo and net BMD losses of less than 1% at each of the other two sites.
In both studies, side effects of abaloparatide were similar in type and incidence to placebo. Of note, the incidence of mild, transient hypercalcemia in abaloparatide-treated patients was half that of the teriparatide group.
Asked why the subcutaneous abaloparatide at 80 mcg is so much more effective at increasing BMD than teriparatide is at its approved dose, Dr. Harris replied, "They’re different peptides." In monkey studies, abaloparatide showed less increase in cortical bone porosity than in studies done using teriparatide. And in the head-to-head phase II study, the increase in bone turnover markers related to resorption was substantially greater with teriparatide. So abaloparatide’s greater BMD-building efficacy is because of greater selectivity for increased bone formation and less bone resorption relative to teriparatide, he suggested.
The abaloparatide patch utilizes proprietary technology developed by 3M. The dime-size patch contains 316 spearlike microprojections, each 500 mcm (micrometers) long. The tip of each microprojection is coated with abaloparatide. When the patch is applied to periumbilical skin, the microprojections penetrate the skin to a depth of about 250 mcm, putting the tip into the upper dermis. Patch application was painless and without side effects in the phase II study, according to Dr. Harris.
These phase II studies were funded by Radius Health.
AT ICE/ENDO 2014
Key clinical point: A novel anabolic agent being developed for the treatment of postmenopausal osteoporosis increased BMD faster and to a greater extent than did teriparatide.
Major finding: Total hip bone mineral density increased by 2.6% over baseline after 24 weeks of abaloparatide at 80 mcg daily, compared with 0.45% with teriparatide at 20 mcg daily and 0.65% with placebo.
Data source: This phase II randomized trial included 222 postmenopausal women with osteoporosis.
Disclosures: The study was sponsored by Radius Health. The presenter is the company’s chief medical officer.
Three simple questions can screen for suicide risk in ED patients
DALLAS – Using a brief three-question screen, it’s feasible to increase dramatically the detection of suicide risk during routine emergency department care.
That’s the key message from phase II of ED-SAFE (Emergency Department Safety Assessment and Follow-up Evaluation), a National Institute of Mental Health–sponsored multicenter study of the impact of implementing universal suicide risk screening in the nation’s EDs.
The third and final phase of ED-SAFE, now underway, will determine whether universal ED suicide risk screening actually becomes policy. In phase III, patients detected as at-risk through screening will receive post-ED telephone counseling from a centralized center. They are being followed for 12 months to see if detection and intervention affects the incidence of attempted or completed suicide, Edwin D. Boudreaux, Ph.D., explained at the annual meeting of the Society for Academic Emergency Medicine.
ED-SAFE is taking place at eight EDs around the country. It has entailed review of more than 236,000 charts of patients presenting to participating EDs with a nonpsychiatric chief complaint.
During the 12-month baseline treatment-as-usual phase of the study, 26% of patients presenting to the ED with a nonpsychiatric chief complaint were screened for suicide risk as documented in the medical record. During phase II, when triage nurses underwent formal training in the effective use of the three-item ED-SAFE Patient Safety Screen, this rate jumped to 73%. And in phase III – the intervention phase – the proportion of ED patients documented as having been screened for suicide risk climbed further to 85%, reported Dr. Boudreaux, chair of the ED-SAFE steering committee and professor of emergency medicine, psychiatry, and quantitative health sciences at the University of Massachusetts, Worcester.
In the baseline phase, 2.9% of all patients presenting to the ED with a nonpsychiatric chief complaint were detected as having some level of suicide risk/self-harm, as recorded in their chart. In phase II, this figure rose to 5.2%, and in phase III, 5.7% of ED patients were identified through screening as having some level of self-harm.
While this virtual doubling of the rate of suicide risk detection through the use of the three-question screen is impressive, it nonetheless falls well short of the true prevalence of current or past suicidal ideation or behavior among ED patients based upon prospective studies, the psychologist noted.
"Those studies would suggest 15% of ED patients should screen positive using our definition," Dr. Boudreaux said.
He conceded the existence of substantial barriers to implementation of universal ED screening for suicide risk. EDs are already very busy places. Nationally there are more than 120 million ED visits per year, and they occur around the clock.
"You get lots of resistance from clinical staff for doing this kind of screening routinely," he explained. "It’s because of the Pandora’s Box phenomenon. People are afraid that if they start to screen for suicide they’ll actually detect suicide risk, and that means they’ll have to act on it. Many clinicians have told us that they just prefer not to ask because they really don’t want to have to take the next step because it’s complicated, it’s perceived to delay care, and there’s not a ready solution to the problem once it’s identified. There are limited aftercare and referral options to specialty services even if you want to try to do something."
Nevertheless, the Joint Commission has called for organizations to identify patients at risk for suicide. And the ED is a logical place to conduct universal screening for occult suicidality because there is abundant evidence that a much larger proportion of individuals who come to the ED for a nonpsychiatric chief complaint have some degree of suicidal ideation or have made a past attempt than is the case among the general population.
Moreover, the annual number of ED visits for attempted suicide and self-inflicted injury more than doubled in a recent 16-year period, as shown in a study by Dr. Boudreaux and coworkers (Gen. Hosp. Psychiatry 2012;34:557-65).
The three-question ED-SAFE Patient Safety Screener borrows from well-validated screening instruments. It begins with a brief introductory script designed to foster a nonthreatening atmosphere. Then comes question one, which screens for depressed mood: Over the past 2 weeks, have you felt down, depressed, or hopeless? Question two: Over the past 2 weeks, have you had thoughts of killing yourself? Then question three: Have you ever attempted to kill yourself? If ‘yes,’ then when did this happen? A recent attempt is defined as one within the past 6 months.
When the triage nurse gets a positive result on the three-question screen, an ED physician is then brought in to ask additional questions aimed at gauging the severity and acuity of the patient’s suicidality.
"That’s done to help decide whether to consult psychiatry. That secondary screening is particularly important whenever you’re implementing universal screening because your primary screening identifies many low- to moderate-risk individuals," the psychologist explained.
ED-SAFE is funded by the National Institute of Mental Health. Dr. Boudreaux reported having no financial conflicts.
DALLAS – Using a brief three-question screen, it’s feasible to increase dramatically the detection of suicide risk during routine emergency department care.
That’s the key message from phase II of ED-SAFE (Emergency Department Safety Assessment and Follow-up Evaluation), a National Institute of Mental Health–sponsored multicenter study of the impact of implementing universal suicide risk screening in the nation’s EDs.
The third and final phase of ED-SAFE, now underway, will determine whether universal ED suicide risk screening actually becomes policy. In phase III, patients detected as at-risk through screening will receive post-ED telephone counseling from a centralized center. They are being followed for 12 months to see if detection and intervention affects the incidence of attempted or completed suicide, Edwin D. Boudreaux, Ph.D., explained at the annual meeting of the Society for Academic Emergency Medicine.
ED-SAFE is taking place at eight EDs around the country. It has entailed review of more than 236,000 charts of patients presenting to participating EDs with a nonpsychiatric chief complaint.
During the 12-month baseline treatment-as-usual phase of the study, 26% of patients presenting to the ED with a nonpsychiatric chief complaint were screened for suicide risk as documented in the medical record. During phase II, when triage nurses underwent formal training in the effective use of the three-item ED-SAFE Patient Safety Screen, this rate jumped to 73%. And in phase III – the intervention phase – the proportion of ED patients documented as having been screened for suicide risk climbed further to 85%, reported Dr. Boudreaux, chair of the ED-SAFE steering committee and professor of emergency medicine, psychiatry, and quantitative health sciences at the University of Massachusetts, Worcester.
In the baseline phase, 2.9% of all patients presenting to the ED with a nonpsychiatric chief complaint were detected as having some level of suicide risk/self-harm, as recorded in their chart. In phase II, this figure rose to 5.2%, and in phase III, 5.7% of ED patients were identified through screening as having some level of self-harm.
While this virtual doubling of the rate of suicide risk detection through the use of the three-question screen is impressive, it nonetheless falls well short of the true prevalence of current or past suicidal ideation or behavior among ED patients based upon prospective studies, the psychologist noted.
"Those studies would suggest 15% of ED patients should screen positive using our definition," Dr. Boudreaux said.
He conceded the existence of substantial barriers to implementation of universal ED screening for suicide risk. EDs are already very busy places. Nationally there are more than 120 million ED visits per year, and they occur around the clock.
"You get lots of resistance from clinical staff for doing this kind of screening routinely," he explained. "It’s because of the Pandora’s Box phenomenon. People are afraid that if they start to screen for suicide they’ll actually detect suicide risk, and that means they’ll have to act on it. Many clinicians have told us that they just prefer not to ask because they really don’t want to have to take the next step because it’s complicated, it’s perceived to delay care, and there’s not a ready solution to the problem once it’s identified. There are limited aftercare and referral options to specialty services even if you want to try to do something."
Nevertheless, the Joint Commission has called for organizations to identify patients at risk for suicide. And the ED is a logical place to conduct universal screening for occult suicidality because there is abundant evidence that a much larger proportion of individuals who come to the ED for a nonpsychiatric chief complaint have some degree of suicidal ideation or have made a past attempt than is the case among the general population.
Moreover, the annual number of ED visits for attempted suicide and self-inflicted injury more than doubled in a recent 16-year period, as shown in a study by Dr. Boudreaux and coworkers (Gen. Hosp. Psychiatry 2012;34:557-65).
The three-question ED-SAFE Patient Safety Screener borrows from well-validated screening instruments. It begins with a brief introductory script designed to foster a nonthreatening atmosphere. Then comes question one, which screens for depressed mood: Over the past 2 weeks, have you felt down, depressed, or hopeless? Question two: Over the past 2 weeks, have you had thoughts of killing yourself? Then question three: Have you ever attempted to kill yourself? If ‘yes,’ then when did this happen? A recent attempt is defined as one within the past 6 months.
When the triage nurse gets a positive result on the three-question screen, an ED physician is then brought in to ask additional questions aimed at gauging the severity and acuity of the patient’s suicidality.
"That’s done to help decide whether to consult psychiatry. That secondary screening is particularly important whenever you’re implementing universal screening because your primary screening identifies many low- to moderate-risk individuals," the psychologist explained.
ED-SAFE is funded by the National Institute of Mental Health. Dr. Boudreaux reported having no financial conflicts.
DALLAS – Using a brief three-question screen, it’s feasible to increase dramatically the detection of suicide risk during routine emergency department care.
That’s the key message from phase II of ED-SAFE (Emergency Department Safety Assessment and Follow-up Evaluation), a National Institute of Mental Health–sponsored multicenter study of the impact of implementing universal suicide risk screening in the nation’s EDs.
The third and final phase of ED-SAFE, now underway, will determine whether universal ED suicide risk screening actually becomes policy. In phase III, patients detected as at-risk through screening will receive post-ED telephone counseling from a centralized center. They are being followed for 12 months to see if detection and intervention affects the incidence of attempted or completed suicide, Edwin D. Boudreaux, Ph.D., explained at the annual meeting of the Society for Academic Emergency Medicine.
ED-SAFE is taking place at eight EDs around the country. It has entailed review of more than 236,000 charts of patients presenting to participating EDs with a nonpsychiatric chief complaint.
During the 12-month baseline treatment-as-usual phase of the study, 26% of patients presenting to the ED with a nonpsychiatric chief complaint were screened for suicide risk as documented in the medical record. During phase II, when triage nurses underwent formal training in the effective use of the three-item ED-SAFE Patient Safety Screen, this rate jumped to 73%. And in phase III – the intervention phase – the proportion of ED patients documented as having been screened for suicide risk climbed further to 85%, reported Dr. Boudreaux, chair of the ED-SAFE steering committee and professor of emergency medicine, psychiatry, and quantitative health sciences at the University of Massachusetts, Worcester.
In the baseline phase, 2.9% of all patients presenting to the ED with a nonpsychiatric chief complaint were detected as having some level of suicide risk/self-harm, as recorded in their chart. In phase II, this figure rose to 5.2%, and in phase III, 5.7% of ED patients were identified through screening as having some level of self-harm.
While this virtual doubling of the rate of suicide risk detection through the use of the three-question screen is impressive, it nonetheless falls well short of the true prevalence of current or past suicidal ideation or behavior among ED patients based upon prospective studies, the psychologist noted.
"Those studies would suggest 15% of ED patients should screen positive using our definition," Dr. Boudreaux said.
He conceded the existence of substantial barriers to implementation of universal ED screening for suicide risk. EDs are already very busy places. Nationally there are more than 120 million ED visits per year, and they occur around the clock.
"You get lots of resistance from clinical staff for doing this kind of screening routinely," he explained. "It’s because of the Pandora’s Box phenomenon. People are afraid that if they start to screen for suicide they’ll actually detect suicide risk, and that means they’ll have to act on it. Many clinicians have told us that they just prefer not to ask because they really don’t want to have to take the next step because it’s complicated, it’s perceived to delay care, and there’s not a ready solution to the problem once it’s identified. There are limited aftercare and referral options to specialty services even if you want to try to do something."
Nevertheless, the Joint Commission has called for organizations to identify patients at risk for suicide. And the ED is a logical place to conduct universal screening for occult suicidality because there is abundant evidence that a much larger proportion of individuals who come to the ED for a nonpsychiatric chief complaint have some degree of suicidal ideation or have made a past attempt than is the case among the general population.
Moreover, the annual number of ED visits for attempted suicide and self-inflicted injury more than doubled in a recent 16-year period, as shown in a study by Dr. Boudreaux and coworkers (Gen. Hosp. Psychiatry 2012;34:557-65).
The three-question ED-SAFE Patient Safety Screener borrows from well-validated screening instruments. It begins with a brief introductory script designed to foster a nonthreatening atmosphere. Then comes question one, which screens for depressed mood: Over the past 2 weeks, have you felt down, depressed, or hopeless? Question two: Over the past 2 weeks, have you had thoughts of killing yourself? Then question three: Have you ever attempted to kill yourself? If ‘yes,’ then when did this happen? A recent attempt is defined as one within the past 6 months.
When the triage nurse gets a positive result on the three-question screen, an ED physician is then brought in to ask additional questions aimed at gauging the severity and acuity of the patient’s suicidality.
"That’s done to help decide whether to consult psychiatry. That secondary screening is particularly important whenever you’re implementing universal screening because your primary screening identifies many low- to moderate-risk individuals," the psychologist explained.
ED-SAFE is funded by the National Institute of Mental Health. Dr. Boudreaux reported having no financial conflicts.
AT SAEM 2014
Key clinical point: Universal screening for suicide risk in patients who present to emergency departments with a nonpsychiatric chief complaint is now a step closer to reality.
Major finding: Implementation of a brief, structured three-question screen for suicide risk administered by triage nurses resulted in detection of increased risk of self-harm in 5.7% of all ED patients presenting with a nonpsychiatric chief complaint, compared with a 2.9% rate during treatment as usual.
Data source: The ED-SAFE study is taking place at eight U.S. emergency departments. It uses an interrupted time series design with three sequential phases in order to determine the impact of implementing universal ED suicide risk screening.
Disclosures: The study is funded by the National Institute of Mental Health. The presenter reported having no financial conflicts.
Aspirin’s benefits may be blunted in African American women
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) actually shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the Ohio State University Center for Women’s Health. She reported having no financial conflicts.

|
|
There has been a significant amount of research focused on understanding the relationship between inflammation and cardiovascular disease, but how this relationship is impacted by gender and race has been less studied. Although the sample size here was small, the key point is that more work is needed in the critical area of ethnic differences. Also, the correlation between anti-inflammatory levels and outcomes in this study needs more clarity, and although the results are intriguing, we should not change current practice until more research is undertaken in a larger population and linked to clear cardiovascular outcomes. Finally, there may be additional confounders such as body weight or other modifiable cardiovascular risk factors that may have led to the corresponding differences in these markers.
In summary, this study does hint at the notion that when designing interventions to reduce cardiovascular risks for elderly women, clearly "one size may not fit all."
Dr. Hiren Shah is with Northwestern University, Chicago, and medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital.

|
|
There has been a significant amount of research focused on understanding the relationship between inflammation and cardiovascular disease, but how this relationship is impacted by gender and race has been less studied. Although the sample size here was small, the key point is that more work is needed in the critical area of ethnic differences. Also, the correlation between anti-inflammatory levels and outcomes in this study needs more clarity, and although the results are intriguing, we should not change current practice until more research is undertaken in a larger population and linked to clear cardiovascular outcomes. Finally, there may be additional confounders such as body weight or other modifiable cardiovascular risk factors that may have led to the corresponding differences in these markers.
In summary, this study does hint at the notion that when designing interventions to reduce cardiovascular risks for elderly women, clearly "one size may not fit all."
Dr. Hiren Shah is with Northwestern University, Chicago, and medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital.

|
|
There has been a significant amount of research focused on understanding the relationship between inflammation and cardiovascular disease, but how this relationship is impacted by gender and race has been less studied. Although the sample size here was small, the key point is that more work is needed in the critical area of ethnic differences. Also, the correlation between anti-inflammatory levels and outcomes in this study needs more clarity, and although the results are intriguing, we should not change current practice until more research is undertaken in a larger population and linked to clear cardiovascular outcomes. Finally, there may be additional confounders such as body weight or other modifiable cardiovascular risk factors that may have led to the corresponding differences in these markers.
In summary, this study does hint at the notion that when designing interventions to reduce cardiovascular risks for elderly women, clearly "one size may not fit all."
Dr. Hiren Shah is with Northwestern University, Chicago, and medical director of the Medicine and Cardiac Telemetry Hospitalist Unit at Northwestern Memorial Hospital.
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) actually shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the Ohio State University Center for Women’s Health. She reported having no financial conflicts.
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) actually shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the Ohio State University Center for Women’s Health. She reported having no financial conflicts.
AT ICE/ENDO 2014
Key clinical point: Postmenopausal African American women may have a blunted response to the anti-inflammatory effects of aspirin at 325 mg/day.
Major finding: Mean levels of hsCRP dropped by 25% and IL-6 decreased by 48% in white women over the course of 6 months of aspirin at 325 mg/day. In contrast, CRP remained essentially unchanged despite daily aspirin in African American women, and their IL-6 levels rose.
Data source: This randomized, double-blind, placebo-controlled pilot study included 42 postmenopausal, nondiabetic women; half were African American, half were white. All had documented subclinical atherosclerosis. Subjects received aspirin at 325 mg/day and were studied over the course of 6 months.
Disclosures: The study was supported by the Ohio State University Center for Women’s Health. The presenter reported having no financial conflicts.
J-Tip syringe cuts venipuncture pain in young kids
DALLAS – Jet-injected lidocaine is superior to vapocoolant spray in reducing venipuncture pain in children under 7 years of age, according to a randomized, double-blind clinical trial.
This form of needle-free local anesthesia, administered through what is popularly known as the J-Tip syringe, has been shown previously to decrease venipuncture pain in adults and older children. But data regarding the effectiveness of the J-Tip in young children has been scanty until now, Dr. Maren M. Lunoe noted at the annual meeting of the Society for Academic Emergency Medicine.
She presented the findings of a randomized, sham-controlled, double-blind clinical trial involving 205 children aged 1-6 years who presented for venipuncture. Ninety percent had undergone the procedure before.
The J-Tip device utilizes a cartridge of compressed carbon dioxide to drive buffered lidocaine into the skin. However, it does so with a loud "pop," which is why the study included a control arm featuring a sham J-Tip syringe with the compressed gas but no lidocaine, explained Dr. Lunoe of the Medical College of Wisconsin, Milwaukee.
Participants were randomized 2:1:1 to the J-Tip syringe deployed roughly 30 seconds prior to venipuncture; to a refrigerated vapocoolant spray, also applied immediately prior to venipuncture, which has been usual care for a blood draw at Children’s Hospital of Wisconsin; or to a second control group who received vapocoolant spray and a sham J-Tip.
The primary endpoint was the FLACC score at venipuncture as assessed by two blinded physicians viewing videotapes of every procedure. The FLACC (Face, Legs, Activity, Cry, and Consolability) scale is a validated pain assessment tool scored 0-10.
The median FLACC score while the young children were waiting for the nurse was 2.0. The score jumped by about 2 points in all three groups when the child saw the device. It climbed by another 5.5 points from that point to the actual venipuncture in controls who got vapocoolant spray only, and by 2.5 points in those who got vapocoolant spray plus a sham J-Tip. These increases in FLACC pain score at venipuncture were statistically significant and clinically meaningful. In contrast, patients who received needle-free lidocaine through a loaded J-Tip syringe did not experience a significant increase in FLACC scores at venipuncture.
Forty-five percent of patients in the J-Tip group had no or only mild pain at venipuncture as defined by a FLACC score of 0-3. This was the case in only 23% of the vapocoolant spray–only group and in 30% of controls who got vapocoolant spray and a sham J-Tip.
There were no between-group differences in adverse events, all of which were minor, consisting mostly of mild bruising.
Asked why the study didn’t include a comparison arm pretreated with EMLA cream, Dr. Lunoe said that topical agent takes 30 minutes to take effect, making it unattractive for use in an emergency department or busy clinic.
National Medical Products and Gebauer, which market the J-Tip syringe and the vapocoolant spray, respectively, provided those supplies for the study but had no further involvement. Dr. Lunoe reported no financial conflicts with regard to the investigation.
DALLAS – Jet-injected lidocaine is superior to vapocoolant spray in reducing venipuncture pain in children under 7 years of age, according to a randomized, double-blind clinical trial.
This form of needle-free local anesthesia, administered through what is popularly known as the J-Tip syringe, has been shown previously to decrease venipuncture pain in adults and older children. But data regarding the effectiveness of the J-Tip in young children has been scanty until now, Dr. Maren M. Lunoe noted at the annual meeting of the Society for Academic Emergency Medicine.
She presented the findings of a randomized, sham-controlled, double-blind clinical trial involving 205 children aged 1-6 years who presented for venipuncture. Ninety percent had undergone the procedure before.
The J-Tip device utilizes a cartridge of compressed carbon dioxide to drive buffered lidocaine into the skin. However, it does so with a loud "pop," which is why the study included a control arm featuring a sham J-Tip syringe with the compressed gas but no lidocaine, explained Dr. Lunoe of the Medical College of Wisconsin, Milwaukee.
Participants were randomized 2:1:1 to the J-Tip syringe deployed roughly 30 seconds prior to venipuncture; to a refrigerated vapocoolant spray, also applied immediately prior to venipuncture, which has been usual care for a blood draw at Children’s Hospital of Wisconsin; or to a second control group who received vapocoolant spray and a sham J-Tip.
The primary endpoint was the FLACC score at venipuncture as assessed by two blinded physicians viewing videotapes of every procedure. The FLACC (Face, Legs, Activity, Cry, and Consolability) scale is a validated pain assessment tool scored 0-10.
The median FLACC score while the young children were waiting for the nurse was 2.0. The score jumped by about 2 points in all three groups when the child saw the device. It climbed by another 5.5 points from that point to the actual venipuncture in controls who got vapocoolant spray only, and by 2.5 points in those who got vapocoolant spray plus a sham J-Tip. These increases in FLACC pain score at venipuncture were statistically significant and clinically meaningful. In contrast, patients who received needle-free lidocaine through a loaded J-Tip syringe did not experience a significant increase in FLACC scores at venipuncture.
Forty-five percent of patients in the J-Tip group had no or only mild pain at venipuncture as defined by a FLACC score of 0-3. This was the case in only 23% of the vapocoolant spray–only group and in 30% of controls who got vapocoolant spray and a sham J-Tip.
There were no between-group differences in adverse events, all of which were minor, consisting mostly of mild bruising.
Asked why the study didn’t include a comparison arm pretreated with EMLA cream, Dr. Lunoe said that topical agent takes 30 minutes to take effect, making it unattractive for use in an emergency department or busy clinic.
National Medical Products and Gebauer, which market the J-Tip syringe and the vapocoolant spray, respectively, provided those supplies for the study but had no further involvement. Dr. Lunoe reported no financial conflicts with regard to the investigation.
DALLAS – Jet-injected lidocaine is superior to vapocoolant spray in reducing venipuncture pain in children under 7 years of age, according to a randomized, double-blind clinical trial.
This form of needle-free local anesthesia, administered through what is popularly known as the J-Tip syringe, has been shown previously to decrease venipuncture pain in adults and older children. But data regarding the effectiveness of the J-Tip in young children has been scanty until now, Dr. Maren M. Lunoe noted at the annual meeting of the Society for Academic Emergency Medicine.
She presented the findings of a randomized, sham-controlled, double-blind clinical trial involving 205 children aged 1-6 years who presented for venipuncture. Ninety percent had undergone the procedure before.
The J-Tip device utilizes a cartridge of compressed carbon dioxide to drive buffered lidocaine into the skin. However, it does so with a loud "pop," which is why the study included a control arm featuring a sham J-Tip syringe with the compressed gas but no lidocaine, explained Dr. Lunoe of the Medical College of Wisconsin, Milwaukee.
Participants were randomized 2:1:1 to the J-Tip syringe deployed roughly 30 seconds prior to venipuncture; to a refrigerated vapocoolant spray, also applied immediately prior to venipuncture, which has been usual care for a blood draw at Children’s Hospital of Wisconsin; or to a second control group who received vapocoolant spray and a sham J-Tip.
The primary endpoint was the FLACC score at venipuncture as assessed by two blinded physicians viewing videotapes of every procedure. The FLACC (Face, Legs, Activity, Cry, and Consolability) scale is a validated pain assessment tool scored 0-10.
The median FLACC score while the young children were waiting for the nurse was 2.0. The score jumped by about 2 points in all three groups when the child saw the device. It climbed by another 5.5 points from that point to the actual venipuncture in controls who got vapocoolant spray only, and by 2.5 points in those who got vapocoolant spray plus a sham J-Tip. These increases in FLACC pain score at venipuncture were statistically significant and clinically meaningful. In contrast, patients who received needle-free lidocaine through a loaded J-Tip syringe did not experience a significant increase in FLACC scores at venipuncture.
Forty-five percent of patients in the J-Tip group had no or only mild pain at venipuncture as defined by a FLACC score of 0-3. This was the case in only 23% of the vapocoolant spray–only group and in 30% of controls who got vapocoolant spray and a sham J-Tip.
There were no between-group differences in adverse events, all of which were minor, consisting mostly of mild bruising.
Asked why the study didn’t include a comparison arm pretreated with EMLA cream, Dr. Lunoe said that topical agent takes 30 minutes to take effect, making it unattractive for use in an emergency department or busy clinic.
National Medical Products and Gebauer, which market the J-Tip syringe and the vapocoolant spray, respectively, provided those supplies for the study but had no further involvement. Dr. Lunoe reported no financial conflicts with regard to the investigation.
AT SAEM 2014
Key clinical point: Needle-free local anesthesia via jet-injected lidocaine is a fast and effective means of reducing venipuncture pain for young children.
Major finding: Forty-five percent of young children had no or mild pain at venipuncture if they received jet-injected lidocaine immediately beforehand, a rate twice that seen in controls pretreated with a vapocoolant spray.
Data source: A randomized, prospective, double-blind, sham procedure-controlled study involving 205 children aged 1-6 years undergoing venipuncture.
Disclosures: The presenter reported having no financial conflicts regarding this study, which was carried out with institutional funds.
Variation in admission rates from EDs raises eyebrows
DALLAS – Emergency departments across the United States vary widely in their admission rates for the 15 most common medical and surgical conditions resulting in hospitalization.
The variability is important from a cost perspective because ED admission is increasingly the dominant route by which patients enter the hospital, Dr. Keith E. Kocher observed at the annual meeting of the Society for Academic Emergency Medicine. "We’re talking about potentially billions of dollars that may be in play if we narrow these differences." The 15 conditions collectively account for more than $266 billion/year in hospital charges to payers.
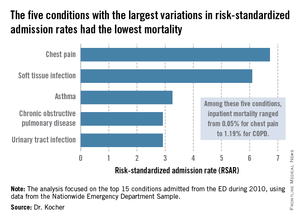
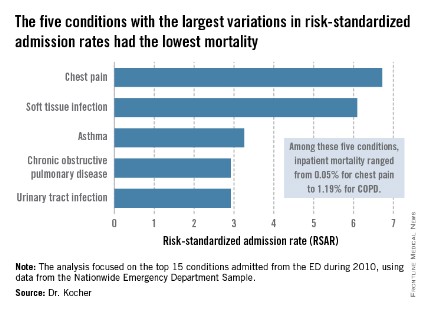
Dr. Kocher, an emergency medicine physician at the University of Michigan, Ann Arbor, and his colleagues conducted a retrospective analysis of the Nationwide Emergency Department Sample for 2010. This database, maintained by the Agency for Healthcare Research and Quality (AHRQ), contains extensive records on the millions of ED visits at nearly 1,000 hospitals in 28 states.
Their analysis adjusted for the severity of case mix by incorporating demographics, comorbid conditions, primary payer, median income, and patient zip code. The researchers then determined risk-standardized admission rates – the number of predicted admissions for each ED given the institutional case mix, divided by the number of expected admissions had those patients been treated at the average ED, multiplied by the mean admission rate for the sample.

The five disorders with the least variation in admission rates among the 15 most commonly admitted conditions were heart failure, stroke, acute renal failure, acute MI, and sepsis. All are characterized by relatively high inpatient mortality.
The five with the greatest variation in admission rates were chest pain, soft tissue infection, asthma, chronic obstructive pulmonary disease, and urinary tract infection. Importantly, these conditions were among those with the lowest inpatient mortality, ranging from 0.05% for patients admitted from the ED for chest pain to a high of 1.19% for those admitted for COPD.
"High-mortality/low-variation diagnoses like sepsis and MI provide little opportunity to realize meaningful spending reductions. Instead, the Big-5 high-variation/low-mortality conditions represent the greatest source of potential savings," Dr. Kocher said.
The ED could become "a workshop for developing innovative strategies for care coordination and alternatives to acute hospitalization, particularly around a select group of high-variation/low-mortality conditions" with the goal of reducing health costs, he said.
If EDs with high risk-standardized admission rates above the median reduced admissions for the five high-variation/low-mortality conditions to the median rate, it would save an estimated $16.9 billion in charges and $5.1 billion in costs per year.Another option might be to set incentives to induce EDs with high-risk–standardized admission rates in the top quartile to reduce admissions to the 75th percentile; the resulting saving would be $7 billion less in charges and $2.1 billion less in costs per year.
Incentivizing top quartile and bottom quartile EDs to meet the median rate would yield an estimated $2.8 billion reduction in charges and a $0.8 billion decrease in costs per year.
The in-hospital mortality implications of moving admission rates toward the median are not known, Dr. Kocher acknowledged. "We’re not implying that we know the optimal rate of admission. In fact, it probably varies from condition to condition."
Further, a formal economic analysis of net expenditures would need to incorporate the increased outpatient expenditures of shifting care to ambulatory settings, he said.
The study was supported by AHRQ. Dr. Kocher reported having no financial conflicts.
Dr. Daniel Ouellette, FCCP, comments: Health care professionals are beginning to appreciate the maxim that those in technology and industry sectors have known for years: Reduction in variance leads to improvement in quality. When this maxim is applied to the subject of hospital admission after an encounter in the ED, we find that a "good news/bad news" scenario exists.
A recent study examining admission rates after an ED visit for the 15 most common diagnoses finds a low variance in admission rates across the country for those diagnoses with a high mortality. On the other hand, those diagnoses with a low mortality had a high variance in admission rates. Can we improve quality and cost by reducing variance in admission rates for these disorders? The authors of this study suggest that costs will be improved substantially by reducing variance. I suspect that this will only happen (and should only happen!) if the optimal admission rates for quality care are low ones.
Dr. Daniel Ouellette, FCCP, comments: Health care professionals are beginning to appreciate the maxim that those in technology and industry sectors have known for years: Reduction in variance leads to improvement in quality. When this maxim is applied to the subject of hospital admission after an encounter in the ED, we find that a "good news/bad news" scenario exists.
A recent study examining admission rates after an ED visit for the 15 most common diagnoses finds a low variance in admission rates across the country for those diagnoses with a high mortality. On the other hand, those diagnoses with a low mortality had a high variance in admission rates. Can we improve quality and cost by reducing variance in admission rates for these disorders? The authors of this study suggest that costs will be improved substantially by reducing variance. I suspect that this will only happen (and should only happen!) if the optimal admission rates for quality care are low ones.
Dr. Daniel Ouellette, FCCP, comments: Health care professionals are beginning to appreciate the maxim that those in technology and industry sectors have known for years: Reduction in variance leads to improvement in quality. When this maxim is applied to the subject of hospital admission after an encounter in the ED, we find that a "good news/bad news" scenario exists.
A recent study examining admission rates after an ED visit for the 15 most common diagnoses finds a low variance in admission rates across the country for those diagnoses with a high mortality. On the other hand, those diagnoses with a low mortality had a high variance in admission rates. Can we improve quality and cost by reducing variance in admission rates for these disorders? The authors of this study suggest that costs will be improved substantially by reducing variance. I suspect that this will only happen (and should only happen!) if the optimal admission rates for quality care are low ones.
DALLAS – Emergency departments across the United States vary widely in their admission rates for the 15 most common medical and surgical conditions resulting in hospitalization.
The variability is important from a cost perspective because ED admission is increasingly the dominant route by which patients enter the hospital, Dr. Keith E. Kocher observed at the annual meeting of the Society for Academic Emergency Medicine. "We’re talking about potentially billions of dollars that may be in play if we narrow these differences." The 15 conditions collectively account for more than $266 billion/year in hospital charges to payers.

Dr. Kocher, an emergency medicine physician at the University of Michigan, Ann Arbor, and his colleagues conducted a retrospective analysis of the Nationwide Emergency Department Sample for 2010. This database, maintained by the Agency for Healthcare Research and Quality (AHRQ), contains extensive records on the millions of ED visits at nearly 1,000 hospitals in 28 states.
Their analysis adjusted for the severity of case mix by incorporating demographics, comorbid conditions, primary payer, median income, and patient zip code. The researchers then determined risk-standardized admission rates – the number of predicted admissions for each ED given the institutional case mix, divided by the number of expected admissions had those patients been treated at the average ED, multiplied by the mean admission rate for the sample.

The five disorders with the least variation in admission rates among the 15 most commonly admitted conditions were heart failure, stroke, acute renal failure, acute MI, and sepsis. All are characterized by relatively high inpatient mortality.
The five with the greatest variation in admission rates were chest pain, soft tissue infection, asthma, chronic obstructive pulmonary disease, and urinary tract infection. Importantly, these conditions were among those with the lowest inpatient mortality, ranging from 0.05% for patients admitted from the ED for chest pain to a high of 1.19% for those admitted for COPD.
"High-mortality/low-variation diagnoses like sepsis and MI provide little opportunity to realize meaningful spending reductions. Instead, the Big-5 high-variation/low-mortality conditions represent the greatest source of potential savings," Dr. Kocher said.
The ED could become "a workshop for developing innovative strategies for care coordination and alternatives to acute hospitalization, particularly around a select group of high-variation/low-mortality conditions" with the goal of reducing health costs, he said.
If EDs with high risk-standardized admission rates above the median reduced admissions for the five high-variation/low-mortality conditions to the median rate, it would save an estimated $16.9 billion in charges and $5.1 billion in costs per year.Another option might be to set incentives to induce EDs with high-risk–standardized admission rates in the top quartile to reduce admissions to the 75th percentile; the resulting saving would be $7 billion less in charges and $2.1 billion less in costs per year.
Incentivizing top quartile and bottom quartile EDs to meet the median rate would yield an estimated $2.8 billion reduction in charges and a $0.8 billion decrease in costs per year.
The in-hospital mortality implications of moving admission rates toward the median are not known, Dr. Kocher acknowledged. "We’re not implying that we know the optimal rate of admission. In fact, it probably varies from condition to condition."
Further, a formal economic analysis of net expenditures would need to incorporate the increased outpatient expenditures of shifting care to ambulatory settings, he said.
The study was supported by AHRQ. Dr. Kocher reported having no financial conflicts.
DALLAS – Emergency departments across the United States vary widely in their admission rates for the 15 most common medical and surgical conditions resulting in hospitalization.
The variability is important from a cost perspective because ED admission is increasingly the dominant route by which patients enter the hospital, Dr. Keith E. Kocher observed at the annual meeting of the Society for Academic Emergency Medicine. "We’re talking about potentially billions of dollars that may be in play if we narrow these differences." The 15 conditions collectively account for more than $266 billion/year in hospital charges to payers.

Dr. Kocher, an emergency medicine physician at the University of Michigan, Ann Arbor, and his colleagues conducted a retrospective analysis of the Nationwide Emergency Department Sample for 2010. This database, maintained by the Agency for Healthcare Research and Quality (AHRQ), contains extensive records on the millions of ED visits at nearly 1,000 hospitals in 28 states.
Their analysis adjusted for the severity of case mix by incorporating demographics, comorbid conditions, primary payer, median income, and patient zip code. The researchers then determined risk-standardized admission rates – the number of predicted admissions for each ED given the institutional case mix, divided by the number of expected admissions had those patients been treated at the average ED, multiplied by the mean admission rate for the sample.

The five disorders with the least variation in admission rates among the 15 most commonly admitted conditions were heart failure, stroke, acute renal failure, acute MI, and sepsis. All are characterized by relatively high inpatient mortality.
The five with the greatest variation in admission rates were chest pain, soft tissue infection, asthma, chronic obstructive pulmonary disease, and urinary tract infection. Importantly, these conditions were among those with the lowest inpatient mortality, ranging from 0.05% for patients admitted from the ED for chest pain to a high of 1.19% for those admitted for COPD.
"High-mortality/low-variation diagnoses like sepsis and MI provide little opportunity to realize meaningful spending reductions. Instead, the Big-5 high-variation/low-mortality conditions represent the greatest source of potential savings," Dr. Kocher said.
The ED could become "a workshop for developing innovative strategies for care coordination and alternatives to acute hospitalization, particularly around a select group of high-variation/low-mortality conditions" with the goal of reducing health costs, he said.
If EDs with high risk-standardized admission rates above the median reduced admissions for the five high-variation/low-mortality conditions to the median rate, it would save an estimated $16.9 billion in charges and $5.1 billion in costs per year.Another option might be to set incentives to induce EDs with high-risk–standardized admission rates in the top quartile to reduce admissions to the 75th percentile; the resulting saving would be $7 billion less in charges and $2.1 billion less in costs per year.
Incentivizing top quartile and bottom quartile EDs to meet the median rate would yield an estimated $2.8 billion reduction in charges and a $0.8 billion decrease in costs per year.
The in-hospital mortality implications of moving admission rates toward the median are not known, Dr. Kocher acknowledged. "We’re not implying that we know the optimal rate of admission. In fact, it probably varies from condition to condition."
Further, a formal economic analysis of net expenditures would need to incorporate the increased outpatient expenditures of shifting care to ambulatory settings, he said.
The study was supported by AHRQ. Dr. Kocher reported having no financial conflicts.
Key clinical point: Reducing variation in admission rates from EDs for selected common conditions with low inpatient mortality rates could save billions of dollars in health care expenditures annually.
Major finding: If EDs with hospital admission rates above the national median for five target conditions were to reduce those rates to the median, payers would save an estimated $16.9 billion in charges annually.
Data source: This was a retrospective analysis of the 2010 Nationwide Emergency Department Sample, which contains detailed records on millions of ED visits at nearly 1,000 hospitals in 28 states.
Disclosures: This study was supported by the AHRQ. The presenter reported having no financial conflicts.
Program prevented antipsychotic-induced weight gain in youth
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
AT ICE/ENDO 2014
Key clinical point: Prescribing antipsychotics for children and youth is associated with a high risk of clinically significant weight gain and cardiometabolic deterioration, which can be prevented through an early lifestyle intervention.
Major finding: Twelve percent of young patients on antipsychotic agents for first-episode psychosis experienced a greater than 7% weight gain after 12 weeks in a novel lifestyle intervention program, compared with 75% of nonparticipating controls.
Data source: This pilot study compared outcomes in 16 youth who participated in a 12-week multidisciplinary lifestyle intervention and 12 controls who received usual care. All had gone on antipsychotic medications less than 4 weeks earlier for first-episode psychosis.
Disclosures: The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
Gene variant detects increased cardiometabolic risk among nonobese
CHICAGO – Normal-weight individuals who carry a common variant of the caveolin-1 gene are at nearly fourfold increased risk of the metabolic syndrome compared with those who are homozygous for the major allele, according to a cross-sectional case-control study.
"This particular genotype may improve individual risk profiling and help detect nonobese subjects who are metabolically unhealthy and at increased cardiovascular risk before they experience severe clinical events," Dr. Rene Baudrand observed at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
These findings take on added clinical import in light of mounting evidence that the long-term risks of cardiovascular events and type 2 diabetes in patients with the metabolic syndrome are of similar magnitude regardless of whether an individual is lean, overweight, or obese.
"There is no linear relationship between cardiovascular risk and BMI [body mass index]. So assessment of individual cardiovascular risk should involve more in-depth consideration than BMI alone," said Dr. Baudrand of Brigham and Women’s Hospital, Boston, and the Pontifical Catholic University of Chile, Santiago.
He and others have previously shown that caveolin-1 (cav-1) plays a critical role in cholesterol trafficking and signal transduction. It interacts with insulin receptors, steroid receptors, and ion channels.
The objective of the new study was to investigate the role of a suspected culprit cav-1 gene variant known as cav-1 rs926198 in terms of cardiometabolic risk. Expression of cav-1 has been shown to be significantly diminished in carriers of the variant allele.
The study included 735 white and Hispanic subjects on a strictly controlled dietary regimen. They averaged 45 years of age, with a mean BMI of 27.2 kg/m2. Two-thirds of participants were hypertensive, 9% had type 2 diabetes, and 30% had metabolic syndrome. Among the 200 obese subjects, the prevalence of metabolic syndrome was 62%, compared with 18% among the 535 nonobese subjects.
Fully 57% of subjects were found to carry the cav-1 gene variant. The other 43% were homozygous for what is known as the major allele. Carriers of the cav-1 gene variant were found to have significantly higher rates of type 2 diabetes, prediabetes, metabolic syndrome, and other abnormalities than those with the major allele (see chart). These differences were not explainable by differences in BMI; indeed, the average BMI in the two groups was nearly identical, according to Dr. Baudrand.
He and his coworkers also sought to learn whether study participants with metabolic syndrome would be more likely to have a sibling with metabolic syndrome if they were concordant for the cav-1 gene variant. This proved to be the case. In evaluating 348 subjects belonging to 142 sibships, the likelihood of concordance for metabolic syndrome was threefold greater if the siblings shared the cav-1 risk allele than if they were cav-1 discordant.
Upon closer scrutiny of the data, it became apparent that the seriously unfavorable cardiometabolic profile associated with the cav-1 gene variant was confined to the nonobese subjects. In a multivariate analysis adjusted for age, gender, BMI, and study site, the likelihood of metabolic syndrome among nonobese subjects with the variant allele was 3.86-fold higher than in nonobese individuals carrying the major allele. In addition, the adjusted likelihood of type 2 diabetes was 2.26-fold greater and that of low HDL was 1.78-fold greater, with all differences highly statistically significant. In contrast, the risk of these cardiometabolic problems in obese subjects didn’t vary regardless of the form of the cav-1 gene they possessed.
On the basis of these encouraging findings, the next step will be to conduct prospective studies to see if cav-1 genotyping is useful as a screening tool for cardiometabolic risk in nonobese patients. Further investigation is also warranted to identify the specific mechanisms by which the cav-1 risk allele results in cardiometabolic impairment. Among the leading possibilities are defects in aldosterone signaling, the insulin receptor, or adipocyte function, Dr. Baudrand said.
This study was supported by the National Institutes of Health and the Chilean National Science and Technology Research Fund. Dr. Baudrand reported having no financial conflicts.
CHICAGO – Normal-weight individuals who carry a common variant of the caveolin-1 gene are at nearly fourfold increased risk of the metabolic syndrome compared with those who are homozygous for the major allele, according to a cross-sectional case-control study.
"This particular genotype may improve individual risk profiling and help detect nonobese subjects who are metabolically unhealthy and at increased cardiovascular risk before they experience severe clinical events," Dr. Rene Baudrand observed at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

These findings take on added clinical import in light of mounting evidence that the long-term risks of cardiovascular events and type 2 diabetes in patients with the metabolic syndrome are of similar magnitude regardless of whether an individual is lean, overweight, or obese.
"There is no linear relationship between cardiovascular risk and BMI [body mass index]. So assessment of individual cardiovascular risk should involve more in-depth consideration than BMI alone," said Dr. Baudrand of Brigham and Women’s Hospital, Boston, and the Pontifical Catholic University of Chile, Santiago.
He and others have previously shown that caveolin-1 (cav-1) plays a critical role in cholesterol trafficking and signal transduction. It interacts with insulin receptors, steroid receptors, and ion channels.
The objective of the new study was to investigate the role of a suspected culprit cav-1 gene variant known as cav-1 rs926198 in terms of cardiometabolic risk. Expression of cav-1 has been shown to be significantly diminished in carriers of the variant allele.
The study included 735 white and Hispanic subjects on a strictly controlled dietary regimen. They averaged 45 years of age, with a mean BMI of 27.2 kg/m2. Two-thirds of participants were hypertensive, 9% had type 2 diabetes, and 30% had metabolic syndrome. Among the 200 obese subjects, the prevalence of metabolic syndrome was 62%, compared with 18% among the 535 nonobese subjects.
Fully 57% of subjects were found to carry the cav-1 gene variant. The other 43% were homozygous for what is known as the major allele. Carriers of the cav-1 gene variant were found to have significantly higher rates of type 2 diabetes, prediabetes, metabolic syndrome, and other abnormalities than those with the major allele (see chart). These differences were not explainable by differences in BMI; indeed, the average BMI in the two groups was nearly identical, according to Dr. Baudrand.
He and his coworkers also sought to learn whether study participants with metabolic syndrome would be more likely to have a sibling with metabolic syndrome if they were concordant for the cav-1 gene variant. This proved to be the case. In evaluating 348 subjects belonging to 142 sibships, the likelihood of concordance for metabolic syndrome was threefold greater if the siblings shared the cav-1 risk allele than if they were cav-1 discordant.
Upon closer scrutiny of the data, it became apparent that the seriously unfavorable cardiometabolic profile associated with the cav-1 gene variant was confined to the nonobese subjects. In a multivariate analysis adjusted for age, gender, BMI, and study site, the likelihood of metabolic syndrome among nonobese subjects with the variant allele was 3.86-fold higher than in nonobese individuals carrying the major allele. In addition, the adjusted likelihood of type 2 diabetes was 2.26-fold greater and that of low HDL was 1.78-fold greater, with all differences highly statistically significant. In contrast, the risk of these cardiometabolic problems in obese subjects didn’t vary regardless of the form of the cav-1 gene they possessed.
On the basis of these encouraging findings, the next step will be to conduct prospective studies to see if cav-1 genotyping is useful as a screening tool for cardiometabolic risk in nonobese patients. Further investigation is also warranted to identify the specific mechanisms by which the cav-1 risk allele results in cardiometabolic impairment. Among the leading possibilities are defects in aldosterone signaling, the insulin receptor, or adipocyte function, Dr. Baudrand said.
This study was supported by the National Institutes of Health and the Chilean National Science and Technology Research Fund. Dr. Baudrand reported having no financial conflicts.
CHICAGO – Normal-weight individuals who carry a common variant of the caveolin-1 gene are at nearly fourfold increased risk of the metabolic syndrome compared with those who are homozygous for the major allele, according to a cross-sectional case-control study.
"This particular genotype may improve individual risk profiling and help detect nonobese subjects who are metabolically unhealthy and at increased cardiovascular risk before they experience severe clinical events," Dr. Rene Baudrand observed at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

These findings take on added clinical import in light of mounting evidence that the long-term risks of cardiovascular events and type 2 diabetes in patients with the metabolic syndrome are of similar magnitude regardless of whether an individual is lean, overweight, or obese.
"There is no linear relationship between cardiovascular risk and BMI [body mass index]. So assessment of individual cardiovascular risk should involve more in-depth consideration than BMI alone," said Dr. Baudrand of Brigham and Women’s Hospital, Boston, and the Pontifical Catholic University of Chile, Santiago.
He and others have previously shown that caveolin-1 (cav-1) plays a critical role in cholesterol trafficking and signal transduction. It interacts with insulin receptors, steroid receptors, and ion channels.
The objective of the new study was to investigate the role of a suspected culprit cav-1 gene variant known as cav-1 rs926198 in terms of cardiometabolic risk. Expression of cav-1 has been shown to be significantly diminished in carriers of the variant allele.
The study included 735 white and Hispanic subjects on a strictly controlled dietary regimen. They averaged 45 years of age, with a mean BMI of 27.2 kg/m2. Two-thirds of participants were hypertensive, 9% had type 2 diabetes, and 30% had metabolic syndrome. Among the 200 obese subjects, the prevalence of metabolic syndrome was 62%, compared with 18% among the 535 nonobese subjects.
Fully 57% of subjects were found to carry the cav-1 gene variant. The other 43% were homozygous for what is known as the major allele. Carriers of the cav-1 gene variant were found to have significantly higher rates of type 2 diabetes, prediabetes, metabolic syndrome, and other abnormalities than those with the major allele (see chart). These differences were not explainable by differences in BMI; indeed, the average BMI in the two groups was nearly identical, according to Dr. Baudrand.
He and his coworkers also sought to learn whether study participants with metabolic syndrome would be more likely to have a sibling with metabolic syndrome if they were concordant for the cav-1 gene variant. This proved to be the case. In evaluating 348 subjects belonging to 142 sibships, the likelihood of concordance for metabolic syndrome was threefold greater if the siblings shared the cav-1 risk allele than if they were cav-1 discordant.
Upon closer scrutiny of the data, it became apparent that the seriously unfavorable cardiometabolic profile associated with the cav-1 gene variant was confined to the nonobese subjects. In a multivariate analysis adjusted for age, gender, BMI, and study site, the likelihood of metabolic syndrome among nonobese subjects with the variant allele was 3.86-fold higher than in nonobese individuals carrying the major allele. In addition, the adjusted likelihood of type 2 diabetes was 2.26-fold greater and that of low HDL was 1.78-fold greater, with all differences highly statistically significant. In contrast, the risk of these cardiometabolic problems in obese subjects didn’t vary regardless of the form of the cav-1 gene they possessed.
On the basis of these encouraging findings, the next step will be to conduct prospective studies to see if cav-1 genotyping is useful as a screening tool for cardiometabolic risk in nonobese patients. Further investigation is also warranted to identify the specific mechanisms by which the cav-1 risk allele results in cardiometabolic impairment. Among the leading possibilities are defects in aldosterone signaling, the insulin receptor, or adipocyte function, Dr. Baudrand said.
This study was supported by the National Institutes of Health and the Chilean National Science and Technology Research Fund. Dr. Baudrand reported having no financial conflicts.
AT ICE/ENDO 2014
Key clinical point: A genetic screening test shows promise as a means of identifying lean individuals at elevated risk for metabolic syndrome and type 2 diabetes.
Major finding: Nonobese individuals carrying a common variant of the caveolin-1 gene were at nearly fourfold increased risk for prevalent metabolic syndrome as well as 2.26-fold increased risk for type 2 diabetes, compared with those who were homozygous for the major allele.
Data source: This cross-sectional study entailed caveolin-1 genotyping of 735 subjects.
Disclosures: The study was supported by the National Institutes of Health and the Chilean National Science and Technology Research Fund. The presenter reported having no financial conflicts.
Off-label use of novel oral anticoagulants accelerates
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available.
By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves.
Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available.
By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves.
Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available.
By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves.
Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
Major finding: By the fourth quarter of 2012, 14.9% of patients with nonvalvular AF were on a novel anticoagulant. So were 13.8% of those with valvular AF, even though this is an off-label use of these drugs.
Data source: This study involved more than 190,000 patients with nonvalvular AF and 2,142 with valvular AF in 95 practices participating in the PINNACLE Registry.
Disclosures The PINNACLE Registry is funded by the ACC’s National Cardiovascular Data Registry. The presenter reported having no relevant financial conflicts.
Denosumab's benefits persist through 8 years
CHICAGO – Postmenopausal women on denosumab for 8 years straight showed a continued near-linear increase in bone mineral density at the lumbar spine as well as persistent reduction of bone turnover markers and a sustained low incidence of new vertebral and nonvertebral fractures in the ongoing FREEDOM open-label extension study.
Moreover, no new safety signals have emerged during 5 years of additional denosumab (Prolia) on top of an initial 3 years occurring in the pivotal, phase III, randomized, double-blind, placebo-controlled FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months) trial.
Importantly, the overall risk of adverse events has not increased over time, and rates of malignancies, serious infections, eczema, hypocalcemia, and other adverse events in denosumab-treated patients remain comparable to rates seen in placebo-treated controls in the earlier double-blind phase, Dr. E. Michael Lewiecki reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"The benefit/risk profile for denosumab remains favorable," declared Dr. Lewiecki, director of the New Mexico Clinical Research and Osteoporosis Center, Albuquerque.
The FREEDOM open-label extension is designed to assess the efficacy and safety of up to 10 years of denosumab therapy. Dr. Lewiecki presented the latest update, based on 8 years of treatment, or 5 years for patients in the original placebo arm who later elected to crossover to denosumab upon completing the 3-year double-blind phase. His report included 2,243 women on denosumab at 60 mg by subcutaneous injection every 6 months continuously for 8 years and another 2,207 on the drug for 5 years.
Five cases of osteonecrosis of the jaw have occurred in the long-term therapy group, as well as three cases among those who crossed over to the drug. In addition, there has been one atypical femoral fracture during 8 consecutive years on denosumab and one case in the crossover group.
Impressively, lumbar spine bone mineral density (BMD) has increased by 18.4% over baseline with 8 years of denosumab and by 13.7% with 5 years of active therapy.
"The near-linear slope of increase is unchanged over the course of the study. A similar slope is seen in the crossover group," the endocrinologist observed.
Total hip BMD increased by 8.3% with 8 years of denosumab and 4.9% with 5 years of therapy.
In the pivotal phase III FREEDOM trial, 3 years of denosumab reduced the risk of vertebral fractures by 68% compared to placebo, hip fractures by 40%, and nonvertebral fractures by 20% (N. Engl. J. Med. 2009;361:756-65).
The yearly incidence of new vertebral fractures was 0.7%-1.1% in denosumab-treated patients during years 1-3 and 1.1%-1.3% annually in open-label years 4-8. The annual incidence of new nonvertebral fractures in the denosumab group was 2.1%-2.6% in the first 3 years of double-blind therapy and has trended lower since then: 1.5% in year 4, 1.2% in year 5, 1.8% in year 6, 1.6% in year 7, and 0.7% in year 8.
"There’s a suggestion of a further reduction in the risk of nonvertebral fractures out to the 8-year time point. It’ll be fascinating to look at the data for years 9 and 10 to see if that’s just a statistical aberrance or that very low risk of nonvertebral fractures persists," Dr. Lewiecki commented.
Levels of the bone turnover markers serum C-terminal telopeptide of type 1 collagen and procollagen type 1 N-terminal propeptide quickly dropped upon initiation of denosumab and have remained low through 8 years.
Asked about the mechanism underlying the continued linear increase in lumbar spine BMD seen through 8 years of denosumab, in sharp contrast to the bisphosphonates, where BMD plateaus, Dr. Lewiecki confessed that "Many of us are scratching our heads about this and trying to come up with an explanation."
"I don’t know the reason why, but there are several hypotheses worth considering," he continued. "One is that the effects on cortical bone appear to be different with denosumab than with bisphosphonates. We see a reduction in cortical porosity and a larger improvement in bone mineral density at cortical skeletal sites. Secondly, there’s a possible parathyroid hormone effect that may play a role here: There is a larger and longer-lasting increase in parathyroid hormone with denosumab as compared with bisphosphonates. And finally, there’s animal data in monkeys showing ongoing bone modeling taking place with denosumab; it’s possible that may also play a role."
Denosumab is a fully human monoclonal antibody directed against the receptor activator of nuclear factor-kappa B (RANK) ligand.
The FREEDOM study is supported by Amgen. Dr. Lewiecki reported serving as a consultant to that company as well as to AgNovos Healthcare, Lilly, Merck, and Radius Health.
CHICAGO – Postmenopausal women on denosumab for 8 years straight showed a continued near-linear increase in bone mineral density at the lumbar spine as well as persistent reduction of bone turnover markers and a sustained low incidence of new vertebral and nonvertebral fractures in the ongoing FREEDOM open-label extension study.
Moreover, no new safety signals have emerged during 5 years of additional denosumab (Prolia) on top of an initial 3 years occurring in the pivotal, phase III, randomized, double-blind, placebo-controlled FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months) trial.
Importantly, the overall risk of adverse events has not increased over time, and rates of malignancies, serious infections, eczema, hypocalcemia, and other adverse events in denosumab-treated patients remain comparable to rates seen in placebo-treated controls in the earlier double-blind phase, Dr. E. Michael Lewiecki reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"The benefit/risk profile for denosumab remains favorable," declared Dr. Lewiecki, director of the New Mexico Clinical Research and Osteoporosis Center, Albuquerque.
The FREEDOM open-label extension is designed to assess the efficacy and safety of up to 10 years of denosumab therapy. Dr. Lewiecki presented the latest update, based on 8 years of treatment, or 5 years for patients in the original placebo arm who later elected to crossover to denosumab upon completing the 3-year double-blind phase. His report included 2,243 women on denosumab at 60 mg by subcutaneous injection every 6 months continuously for 8 years and another 2,207 on the drug for 5 years.
Five cases of osteonecrosis of the jaw have occurred in the long-term therapy group, as well as three cases among those who crossed over to the drug. In addition, there has been one atypical femoral fracture during 8 consecutive years on denosumab and one case in the crossover group.
Impressively, lumbar spine bone mineral density (BMD) has increased by 18.4% over baseline with 8 years of denosumab and by 13.7% with 5 years of active therapy.
"The near-linear slope of increase is unchanged over the course of the study. A similar slope is seen in the crossover group," the endocrinologist observed.
Total hip BMD increased by 8.3% with 8 years of denosumab and 4.9% with 5 years of therapy.
In the pivotal phase III FREEDOM trial, 3 years of denosumab reduced the risk of vertebral fractures by 68% compared to placebo, hip fractures by 40%, and nonvertebral fractures by 20% (N. Engl. J. Med. 2009;361:756-65).
The yearly incidence of new vertebral fractures was 0.7%-1.1% in denosumab-treated patients during years 1-3 and 1.1%-1.3% annually in open-label years 4-8. The annual incidence of new nonvertebral fractures in the denosumab group was 2.1%-2.6% in the first 3 years of double-blind therapy and has trended lower since then: 1.5% in year 4, 1.2% in year 5, 1.8% in year 6, 1.6% in year 7, and 0.7% in year 8.
"There’s a suggestion of a further reduction in the risk of nonvertebral fractures out to the 8-year time point. It’ll be fascinating to look at the data for years 9 and 10 to see if that’s just a statistical aberrance or that very low risk of nonvertebral fractures persists," Dr. Lewiecki commented.
Levels of the bone turnover markers serum C-terminal telopeptide of type 1 collagen and procollagen type 1 N-terminal propeptide quickly dropped upon initiation of denosumab and have remained low through 8 years.
Asked about the mechanism underlying the continued linear increase in lumbar spine BMD seen through 8 years of denosumab, in sharp contrast to the bisphosphonates, where BMD plateaus, Dr. Lewiecki confessed that "Many of us are scratching our heads about this and trying to come up with an explanation."
"I don’t know the reason why, but there are several hypotheses worth considering," he continued. "One is that the effects on cortical bone appear to be different with denosumab than with bisphosphonates. We see a reduction in cortical porosity and a larger improvement in bone mineral density at cortical skeletal sites. Secondly, there’s a possible parathyroid hormone effect that may play a role here: There is a larger and longer-lasting increase in parathyroid hormone with denosumab as compared with bisphosphonates. And finally, there’s animal data in monkeys showing ongoing bone modeling taking place with denosumab; it’s possible that may also play a role."
Denosumab is a fully human monoclonal antibody directed against the receptor activator of nuclear factor-kappa B (RANK) ligand.
The FREEDOM study is supported by Amgen. Dr. Lewiecki reported serving as a consultant to that company as well as to AgNovos Healthcare, Lilly, Merck, and Radius Health.
CHICAGO – Postmenopausal women on denosumab for 8 years straight showed a continued near-linear increase in bone mineral density at the lumbar spine as well as persistent reduction of bone turnover markers and a sustained low incidence of new vertebral and nonvertebral fractures in the ongoing FREEDOM open-label extension study.
Moreover, no new safety signals have emerged during 5 years of additional denosumab (Prolia) on top of an initial 3 years occurring in the pivotal, phase III, randomized, double-blind, placebo-controlled FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months) trial.
Importantly, the overall risk of adverse events has not increased over time, and rates of malignancies, serious infections, eczema, hypocalcemia, and other adverse events in denosumab-treated patients remain comparable to rates seen in placebo-treated controls in the earlier double-blind phase, Dr. E. Michael Lewiecki reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"The benefit/risk profile for denosumab remains favorable," declared Dr. Lewiecki, director of the New Mexico Clinical Research and Osteoporosis Center, Albuquerque.
The FREEDOM open-label extension is designed to assess the efficacy and safety of up to 10 years of denosumab therapy. Dr. Lewiecki presented the latest update, based on 8 years of treatment, or 5 years for patients in the original placebo arm who later elected to crossover to denosumab upon completing the 3-year double-blind phase. His report included 2,243 women on denosumab at 60 mg by subcutaneous injection every 6 months continuously for 8 years and another 2,207 on the drug for 5 years.
Five cases of osteonecrosis of the jaw have occurred in the long-term therapy group, as well as three cases among those who crossed over to the drug. In addition, there has been one atypical femoral fracture during 8 consecutive years on denosumab and one case in the crossover group.
Impressively, lumbar spine bone mineral density (BMD) has increased by 18.4% over baseline with 8 years of denosumab and by 13.7% with 5 years of active therapy.
"The near-linear slope of increase is unchanged over the course of the study. A similar slope is seen in the crossover group," the endocrinologist observed.
Total hip BMD increased by 8.3% with 8 years of denosumab and 4.9% with 5 years of therapy.
In the pivotal phase III FREEDOM trial, 3 years of denosumab reduced the risk of vertebral fractures by 68% compared to placebo, hip fractures by 40%, and nonvertebral fractures by 20% (N. Engl. J. Med. 2009;361:756-65).
The yearly incidence of new vertebral fractures was 0.7%-1.1% in denosumab-treated patients during years 1-3 and 1.1%-1.3% annually in open-label years 4-8. The annual incidence of new nonvertebral fractures in the denosumab group was 2.1%-2.6% in the first 3 years of double-blind therapy and has trended lower since then: 1.5% in year 4, 1.2% in year 5, 1.8% in year 6, 1.6% in year 7, and 0.7% in year 8.
"There’s a suggestion of a further reduction in the risk of nonvertebral fractures out to the 8-year time point. It’ll be fascinating to look at the data for years 9 and 10 to see if that’s just a statistical aberrance or that very low risk of nonvertebral fractures persists," Dr. Lewiecki commented.
Levels of the bone turnover markers serum C-terminal telopeptide of type 1 collagen and procollagen type 1 N-terminal propeptide quickly dropped upon initiation of denosumab and have remained low through 8 years.
Asked about the mechanism underlying the continued linear increase in lumbar spine BMD seen through 8 years of denosumab, in sharp contrast to the bisphosphonates, where BMD plateaus, Dr. Lewiecki confessed that "Many of us are scratching our heads about this and trying to come up with an explanation."
"I don’t know the reason why, but there are several hypotheses worth considering," he continued. "One is that the effects on cortical bone appear to be different with denosumab than with bisphosphonates. We see a reduction in cortical porosity and a larger improvement in bone mineral density at cortical skeletal sites. Secondly, there’s a possible parathyroid hormone effect that may play a role here: There is a larger and longer-lasting increase in parathyroid hormone with denosumab as compared with bisphosphonates. And finally, there’s animal data in monkeys showing ongoing bone modeling taking place with denosumab; it’s possible that may also play a role."
Denosumab is a fully human monoclonal antibody directed against the receptor activator of nuclear factor-kappa B (RANK) ligand.
The FREEDOM study is supported by Amgen. Dr. Lewiecki reported serving as a consultant to that company as well as to AgNovos Healthcare, Lilly, Merck, and Radius Health.
AT ICE/ENDO 2014
Key clinical point: An ongoing major study provides reassurance regarding the long-term safety and effectiveness of denosumab for postmenopausal osteoporosis.
Major finding: Eight years of denosumab was associated with sustained low fracture rates, steadily increasing bone mineral density, persistent reduction of bone turnover, and no increase in adverse events over time.
Data source: The FREEDOM trial open-label extension includes 4,450 women on denosumab for either 5 or 8 years and counting.
Disclosures: The study was sponsored by Amgen. The presenter serves as a consultant to the company.
Acromegaly: Look closely for sleep-disordered breathing
CHICAGO – Roughly 80% of patients with newly diagnosed and as-yet untreated acromegaly already have obstructive sleep apnea, according to the findings of what’s believed to be the largest-ever polysomnography study in such subjects.
Importantly, this study showed that the common practice of screening for sleep-disordered breathing with the Epworth Sleepiness Scale (ESS) and overnight oximetry to measure the desaturation index will greatly underdiagnose this important condition in patients with acromegaly. These are poor screening tools in this setting. They need to be supplemented by polysomnography, with the diagnosis of obstructive sleep apnea (OSA) hinging upon the finding of an abnormal apnea-hypopnea index, Dr. Andrew S. Powlson asserted at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Dr. Powlson of the University of Cambridge (U.K.), reported on 40 consecutive patients with newly diagnosed, treatment-naive acromegaly who were evaluated by the ESS, desaturation index, and polysomnography. The study’s purpose was to shed light on a controversy: Is sleep-disordered breathing an inherent feature of acromegaly or a secondary side effect of its treatment? The question takes on added clinical relevance in light of OSA’s known predisposition to premature cardiovascular and metabolic disease, motor vehicle accidents, and impaired quality of life.
Of the 40 patients, 31 (78%) met diagnostic criteria for OSA by polysomnography. The OSA was defined on the basis of the apnea-hypopnea index as mild in 12, moderate in 5, and severe in 14.
In contrast, the ESS performed dismally as a screening instrument: Only 12 of the 31 patients with OSA had an ESS score greater than 11, which is the standard threshold for further investigation. Moreover, measurement of the diffusion index during overnight oximetry identified only 21 patients as having OSA: 11 rated mild, 7 moderate, and 3 severe, as compared to 14 patients classified as having severe OSA by polysomnography.
Patients with acromegaly displayed an increased sleep arousal index and more periodic limb movements during sleep than in reference norms, which translates into marked disruption of the normal sleep cycle. Of note, however, polysomnography showed that sleep latency – that is, the time it takes to fall asleep – and total sleep time were normal in the acromegaly patients with OSA.
Instead, the predominant pattern was one of disrupted sleep architecture. Twenty-seven acromegaly patients spent longer than expected in stage-1 sleep, while the deeper sleep stages were dramatically diminished. Indeed, 26 patients had reduced stage-2 sleep, 31 had reduced REM sleep, and 26 had shortened slow wave sleep.
Dr. Powlson reported having no financial disclosures in connection with this study, conducted with institutional funds.
CHICAGO – Roughly 80% of patients with newly diagnosed and as-yet untreated acromegaly already have obstructive sleep apnea, according to the findings of what’s believed to be the largest-ever polysomnography study in such subjects.
Importantly, this study showed that the common practice of screening for sleep-disordered breathing with the Epworth Sleepiness Scale (ESS) and overnight oximetry to measure the desaturation index will greatly underdiagnose this important condition in patients with acromegaly. These are poor screening tools in this setting. They need to be supplemented by polysomnography, with the diagnosis of obstructive sleep apnea (OSA) hinging upon the finding of an abnormal apnea-hypopnea index, Dr. Andrew S. Powlson asserted at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Dr. Powlson of the University of Cambridge (U.K.), reported on 40 consecutive patients with newly diagnosed, treatment-naive acromegaly who were evaluated by the ESS, desaturation index, and polysomnography. The study’s purpose was to shed light on a controversy: Is sleep-disordered breathing an inherent feature of acromegaly or a secondary side effect of its treatment? The question takes on added clinical relevance in light of OSA’s known predisposition to premature cardiovascular and metabolic disease, motor vehicle accidents, and impaired quality of life.
Of the 40 patients, 31 (78%) met diagnostic criteria for OSA by polysomnography. The OSA was defined on the basis of the apnea-hypopnea index as mild in 12, moderate in 5, and severe in 14.
In contrast, the ESS performed dismally as a screening instrument: Only 12 of the 31 patients with OSA had an ESS score greater than 11, which is the standard threshold for further investigation. Moreover, measurement of the diffusion index during overnight oximetry identified only 21 patients as having OSA: 11 rated mild, 7 moderate, and 3 severe, as compared to 14 patients classified as having severe OSA by polysomnography.
Patients with acromegaly displayed an increased sleep arousal index and more periodic limb movements during sleep than in reference norms, which translates into marked disruption of the normal sleep cycle. Of note, however, polysomnography showed that sleep latency – that is, the time it takes to fall asleep – and total sleep time were normal in the acromegaly patients with OSA.
Instead, the predominant pattern was one of disrupted sleep architecture. Twenty-seven acromegaly patients spent longer than expected in stage-1 sleep, while the deeper sleep stages were dramatically diminished. Indeed, 26 patients had reduced stage-2 sleep, 31 had reduced REM sleep, and 26 had shortened slow wave sleep.
Dr. Powlson reported having no financial disclosures in connection with this study, conducted with institutional funds.
CHICAGO – Roughly 80% of patients with newly diagnosed and as-yet untreated acromegaly already have obstructive sleep apnea, according to the findings of what’s believed to be the largest-ever polysomnography study in such subjects.
Importantly, this study showed that the common practice of screening for sleep-disordered breathing with the Epworth Sleepiness Scale (ESS) and overnight oximetry to measure the desaturation index will greatly underdiagnose this important condition in patients with acromegaly. These are poor screening tools in this setting. They need to be supplemented by polysomnography, with the diagnosis of obstructive sleep apnea (OSA) hinging upon the finding of an abnormal apnea-hypopnea index, Dr. Andrew S. Powlson asserted at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Dr. Powlson of the University of Cambridge (U.K.), reported on 40 consecutive patients with newly diagnosed, treatment-naive acromegaly who were evaluated by the ESS, desaturation index, and polysomnography. The study’s purpose was to shed light on a controversy: Is sleep-disordered breathing an inherent feature of acromegaly or a secondary side effect of its treatment? The question takes on added clinical relevance in light of OSA’s known predisposition to premature cardiovascular and metabolic disease, motor vehicle accidents, and impaired quality of life.
Of the 40 patients, 31 (78%) met diagnostic criteria for OSA by polysomnography. The OSA was defined on the basis of the apnea-hypopnea index as mild in 12, moderate in 5, and severe in 14.
In contrast, the ESS performed dismally as a screening instrument: Only 12 of the 31 patients with OSA had an ESS score greater than 11, which is the standard threshold for further investigation. Moreover, measurement of the diffusion index during overnight oximetry identified only 21 patients as having OSA: 11 rated mild, 7 moderate, and 3 severe, as compared to 14 patients classified as having severe OSA by polysomnography.
Patients with acromegaly displayed an increased sleep arousal index and more periodic limb movements during sleep than in reference norms, which translates into marked disruption of the normal sleep cycle. Of note, however, polysomnography showed that sleep latency – that is, the time it takes to fall asleep – and total sleep time were normal in the acromegaly patients with OSA.
Instead, the predominant pattern was one of disrupted sleep architecture. Twenty-seven acromegaly patients spent longer than expected in stage-1 sleep, while the deeper sleep stages were dramatically diminished. Indeed, 26 patients had reduced stage-2 sleep, 31 had reduced REM sleep, and 26 had shortened slow wave sleep.
Dr. Powlson reported having no financial disclosures in connection with this study, conducted with institutional funds.
AT ICE/ENDO 2014
Key clinical point: Reliance on the Epworth Sleepiness Scale and desaturation index on overnight oximetry to screen for OSA in patients with newly diagnosed acromegaly results in an unacceptable underdiagnosis rate.
Major finding: Thirty-one of 40 consecutive patients with de novo acromegaly had OSA on polysomnography.
Data source: Forty consecutive patients with newly diagnosed acromegaly underwent evaluation via the Epworth Sleepiness Scale, desaturation index, and polysomnography.
Disclosures: The presenter reported having no financial conflicts regarding this study, carried out with institutional funds.