User login
Look for nephrotoxicity in adult survivors of childhood cancer
LAS VEGAS – Adult survivors of childhood cancer treated with high-dose cisplatin or high-dose ifosfamide are at markedly increased risk for chronic renal impairment, according to a large Dutch study with a median 18.3-year follow-up.
Long-term treatment-related nephrotoxicity was also seen in the adult survivors of childhood cancer who underwent unilateral nephrectomy combined with abdominal radiation therapy.
"This study is perhaps a warning to us when we’re seeing these adult survivors of childhood cancer in clinic, particularly if they have a history of ifosfamide or cisplatin use, to pay closer attention to the development of chronic kidney disease so they can be managed better in the future," Dr. Anushree C. Shirali said at a meeting sponsored by the National Kidney Foundation.
Drug-induced acute kidney injury is a common event during cancer therapy. Its mechanisms and treatments are well studied. In contrast, the long-term nephrotoxicity of the powerful therapies used in treating childhood cancers has received much less scrutiny. But it’s an increasingly relevant issue because childhood cancer survival rates have improved substantially. Indeed, as the Dutch investigators observed, today 1 in 570 young adults is a childhood cancer survivor (Clin. J. Am. Soc. Nephrol. 2013;8:922-9).
Dr. Shirali, a nephrologist at Yale University in New Haven, Conn., highlighted the Dutch study of 763 adult survivors of childhood cancer because of its unusually long and complete follow-up. The investigators included nearly 90% of all adult survivors treated at Erasmus University in Rotterdam, the Netherlands, during 1964-2005.
High-dose ifosfamide was associated with a 6.2-fold greater likelihood of an increased urinary beta2-microglobulin/creatinine ratio indicative of persisting tubular dysfunction, compared with cancer survivors not receiving that therapy. High-dose cisplatin was associated with a 5.2-fold increased risk of albuminuria. The estimated glomerular filtration rate in adult survivors who had received high-dose ifosfamide was 88 mL/min per 1.73 m2, significantly lower than the 98 mL/min per 1.73 m2 in others. Similarly, patients who had received high-dose cisplatin had an average estimated glomerular filtration rate of 83 mL/min per 1.73 m2, compared with 101 mL/min per 1.73 m2 in survivors not treated with high-dose cisplatin.
In contrast to ifosfamide, its isomer cyclophosphamide was not associated with long-term nephrotoxicity; neither was carboplatin, a cisplatin analogue, or methotrexate. Although methotrexate is known to cause acute nephrotoxicity, this phenomenon appears to be completely reversible, since methotrexate-treated, long-term cancer survivors didn’t develop tubular or glomerular dysfunction.
This long-term study was supported by the Dutch Kidney Foundation. Dr. Shirali, who was not involved in the study, reported having no financial conflicts.
LAS VEGAS – Adult survivors of childhood cancer treated with high-dose cisplatin or high-dose ifosfamide are at markedly increased risk for chronic renal impairment, according to a large Dutch study with a median 18.3-year follow-up.
Long-term treatment-related nephrotoxicity was also seen in the adult survivors of childhood cancer who underwent unilateral nephrectomy combined with abdominal radiation therapy.
"This study is perhaps a warning to us when we’re seeing these adult survivors of childhood cancer in clinic, particularly if they have a history of ifosfamide or cisplatin use, to pay closer attention to the development of chronic kidney disease so they can be managed better in the future," Dr. Anushree C. Shirali said at a meeting sponsored by the National Kidney Foundation.
Drug-induced acute kidney injury is a common event during cancer therapy. Its mechanisms and treatments are well studied. In contrast, the long-term nephrotoxicity of the powerful therapies used in treating childhood cancers has received much less scrutiny. But it’s an increasingly relevant issue because childhood cancer survival rates have improved substantially. Indeed, as the Dutch investigators observed, today 1 in 570 young adults is a childhood cancer survivor (Clin. J. Am. Soc. Nephrol. 2013;8:922-9).
Dr. Shirali, a nephrologist at Yale University in New Haven, Conn., highlighted the Dutch study of 763 adult survivors of childhood cancer because of its unusually long and complete follow-up. The investigators included nearly 90% of all adult survivors treated at Erasmus University in Rotterdam, the Netherlands, during 1964-2005.
High-dose ifosfamide was associated with a 6.2-fold greater likelihood of an increased urinary beta2-microglobulin/creatinine ratio indicative of persisting tubular dysfunction, compared with cancer survivors not receiving that therapy. High-dose cisplatin was associated with a 5.2-fold increased risk of albuminuria. The estimated glomerular filtration rate in adult survivors who had received high-dose ifosfamide was 88 mL/min per 1.73 m2, significantly lower than the 98 mL/min per 1.73 m2 in others. Similarly, patients who had received high-dose cisplatin had an average estimated glomerular filtration rate of 83 mL/min per 1.73 m2, compared with 101 mL/min per 1.73 m2 in survivors not treated with high-dose cisplatin.
In contrast to ifosfamide, its isomer cyclophosphamide was not associated with long-term nephrotoxicity; neither was carboplatin, a cisplatin analogue, or methotrexate. Although methotrexate is known to cause acute nephrotoxicity, this phenomenon appears to be completely reversible, since methotrexate-treated, long-term cancer survivors didn’t develop tubular or glomerular dysfunction.
This long-term study was supported by the Dutch Kidney Foundation. Dr. Shirali, who was not involved in the study, reported having no financial conflicts.
LAS VEGAS – Adult survivors of childhood cancer treated with high-dose cisplatin or high-dose ifosfamide are at markedly increased risk for chronic renal impairment, according to a large Dutch study with a median 18.3-year follow-up.
Long-term treatment-related nephrotoxicity was also seen in the adult survivors of childhood cancer who underwent unilateral nephrectomy combined with abdominal radiation therapy.
"This study is perhaps a warning to us when we’re seeing these adult survivors of childhood cancer in clinic, particularly if they have a history of ifosfamide or cisplatin use, to pay closer attention to the development of chronic kidney disease so they can be managed better in the future," Dr. Anushree C. Shirali said at a meeting sponsored by the National Kidney Foundation.
Drug-induced acute kidney injury is a common event during cancer therapy. Its mechanisms and treatments are well studied. In contrast, the long-term nephrotoxicity of the powerful therapies used in treating childhood cancers has received much less scrutiny. But it’s an increasingly relevant issue because childhood cancer survival rates have improved substantially. Indeed, as the Dutch investigators observed, today 1 in 570 young adults is a childhood cancer survivor (Clin. J. Am. Soc. Nephrol. 2013;8:922-9).
Dr. Shirali, a nephrologist at Yale University in New Haven, Conn., highlighted the Dutch study of 763 adult survivors of childhood cancer because of its unusually long and complete follow-up. The investigators included nearly 90% of all adult survivors treated at Erasmus University in Rotterdam, the Netherlands, during 1964-2005.
High-dose ifosfamide was associated with a 6.2-fold greater likelihood of an increased urinary beta2-microglobulin/creatinine ratio indicative of persisting tubular dysfunction, compared with cancer survivors not receiving that therapy. High-dose cisplatin was associated with a 5.2-fold increased risk of albuminuria. The estimated glomerular filtration rate in adult survivors who had received high-dose ifosfamide was 88 mL/min per 1.73 m2, significantly lower than the 98 mL/min per 1.73 m2 in others. Similarly, patients who had received high-dose cisplatin had an average estimated glomerular filtration rate of 83 mL/min per 1.73 m2, compared with 101 mL/min per 1.73 m2 in survivors not treated with high-dose cisplatin.
In contrast to ifosfamide, its isomer cyclophosphamide was not associated with long-term nephrotoxicity; neither was carboplatin, a cisplatin analogue, or methotrexate. Although methotrexate is known to cause acute nephrotoxicity, this phenomenon appears to be completely reversible, since methotrexate-treated, long-term cancer survivors didn’t develop tubular or glomerular dysfunction.
This long-term study was supported by the Dutch Kidney Foundation. Dr. Shirali, who was not involved in the study, reported having no financial conflicts.
AT SCM 14
Key clinical point: Today 1 in 570 young adults is a childhood cancer survivor, and many are on the road to chronic kidney disease.
Major finding: Adult survivors of childhood cancer treated with high-dose cisplatin or high-dose ifosfamide had a significantly lower estimated glomerular filtration rate than those who were not.
Data source: This was a retrospective, single-center study involving 763 adult survivors of childhood cancer with a median 18.3 years of follow-up.
Disclosures: This long-term study was supported by the Dutch Kidney Foundation. Dr. Shirali, who was not involved in the study, reported having no financial conflicts.
An insider’s look at the 2014 atopic dermatitis guidelines
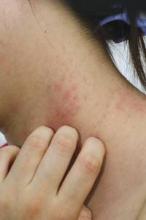
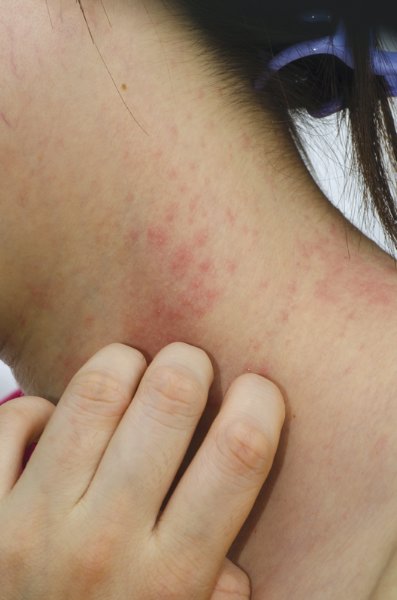
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
EXPERT OPINION FROM THE SPD ANNUAL MEETING
New insights emerge from pediatric chronic cutaneous lupus
COEUR D’ALENE, ID. – Pediatric chronic cutaneous lupus erythematosus transforms into systemic disease in one-quarter of cases, and if it’s going to do so, it’ll typically be within the first year after diagnosis, according to a retrospective study.
"Importantly, no one developed end-organ damage through the end of follow-up, perhaps suggesting that SLE arising in pediatric patients with CCLE follows a more indolent, benign course, just as we’ve seen in previous studies in adults with CCLE with transformation to SLE," Dr. Lisa Arkin observed at the annual meeting of the Society for Pediatric Dermatology.
If that is indeed true, it’s good news for pediatric CCLE patients and their families because it sets them apart from the others who develop SLE prior to adulthood. Twenty percent of all cases of SLE present before age 20, and in general they tend to follow a more aggressive course than adult-onset SLE, with a higher frequency of end-organ involvement necessitating sustained immunosuppression, as well as higher mortality, noted Dr. Arkin, recently appointed as head of pediatric dermatology at Rush University Medical Center, Chicago.
Pediatric CCLE is a rare and little-studied disease. Its most common form is discoid LE, characterized by scaling, telangiectasias, atrophy, follicular plugging, and scarring.
Much more is known about adult discoid LE, a far more common condition. Less than 5%-10% of adults with discoid LE develop SLE, and when they do, the systemic manifestations are more likely to be benign than is typical in adult SLE patients whose systemic disease was not preceded by discoid LE, the pediatric dermatologist explained.
Because so little is known about pediatric CCLE, she conducted a retrospective study including 45 affected patients seen at Lurie Children’s Hospital of Chicago. Seven of the 45 presented initially with concurrent CCLE and SLE. The remaining 38 were followed for a median of 4.4 years, during which 9 patients (24%) transitioned to SLE. Of note, that 1-in-4 risk of transformation from skin to systemic disease is spot-on with the results of the three previous small retrospective studies of pediatric CCLE, the most recent of which was reported 6 years ago from Brazil (Pediatr. Dermatol. 2008;25:163-7).
Of the 7 patients in Dr. Arkin’s study with concurrent CCLE and SLE at presentation, 6 had discoid LE, as did 34 of the 38 with only CCLE at presentation who were followed for development of SLE.
Of the 38 patients with only CCLE at presentation, 12 (32%) had disease limited to the skin throughout follow-up.
Another 17 (45%) with only CCLE at presentation developed laboratory abnormalities during follow-up without transitioning to SLE, including 14 with antinuclear antibodies. Nine of these patients developed more than two laboratory abnormalities. Notably, all had been on hydroxychloroquine ever since the onset of their CCLE. This suggests the intriguing possibility that hydroxychloroquine – an immunomodulatory agent rather than an immunosuppressant – may favorably alter the natural history of disease progression in this young population, according to Dr. Arkin. That’s been shown to be the case in studies conducted in adult military personnel, where long-term follow-up of troops randomized to hydroxychloroquine for antimalarial prophylaxis delayed the diagnosis of SLE, lowered the rate of autoantibody accumulation, retarded renal damage, reduced the risk of infection, and improved overall survival (Lupus 2007;16:401-9).
Patients with concurrent CCLE and SLE presented at a considerably younger age: a mean of 8.7 years old, compared with 13.3 years of age for those with only CCLE.
No formal guidelines exist for monitoring patients with CCLE. Based upon her study findings, Dr. Arkin recommended that "it seems reasonable" to perform a meticulous physical exam and review of systems and obtain laboratory tests on a quarterly basis, particularly in the first year following diagnosis, when the risk of transformation to SLE is highest. The quarterly lab workup should include a complete blood count and complete metabolic panel, urinalysis, complement, antiphospholipid antibodies, and antinuclear antibodies with titer.
"I think one of the big questions which has not been answered is, should all kids with CCLE get oral hydroxychloroquine? There are no studies in children. The only FDA-approved indication is for antimalarial prophylaxis, although it’s certainly used commonly off-label for pediatric CCLE and also for SLE," she said.
The major side effects of oral hydroxychloroquine in children include retinal toxicity, which is dose-dependent, along with GI upset, headaches, and vivid dreams.
Dr. Arkin observed that while a randomized trial of hydroxychloroquine to delay or prevent SLE in high-risk individuals is warranted, she was unable to identify any predictors of transformation from CCLE to SLE in her study, despite an extensive search.
"I love this study," commented SPD President Dr. Beth A. Drolet, professor of dermatology at the Medical College of Wisconsin, Milwaukee. "Your point that if transformation to SLE happens, it’s going to happen quickly – that’s enormously helpful to me clinically."
The study was funded by the SPD. Dr. Arkin reported having no financial conflicts.
COEUR D’ALENE, ID. – Pediatric chronic cutaneous lupus erythematosus transforms into systemic disease in one-quarter of cases, and if it’s going to do so, it’ll typically be within the first year after diagnosis, according to a retrospective study.
"Importantly, no one developed end-organ damage through the end of follow-up, perhaps suggesting that SLE arising in pediatric patients with CCLE follows a more indolent, benign course, just as we’ve seen in previous studies in adults with CCLE with transformation to SLE," Dr. Lisa Arkin observed at the annual meeting of the Society for Pediatric Dermatology.
If that is indeed true, it’s good news for pediatric CCLE patients and their families because it sets them apart from the others who develop SLE prior to adulthood. Twenty percent of all cases of SLE present before age 20, and in general they tend to follow a more aggressive course than adult-onset SLE, with a higher frequency of end-organ involvement necessitating sustained immunosuppression, as well as higher mortality, noted Dr. Arkin, recently appointed as head of pediatric dermatology at Rush University Medical Center, Chicago.
Pediatric CCLE is a rare and little-studied disease. Its most common form is discoid LE, characterized by scaling, telangiectasias, atrophy, follicular plugging, and scarring.
Much more is known about adult discoid LE, a far more common condition. Less than 5%-10% of adults with discoid LE develop SLE, and when they do, the systemic manifestations are more likely to be benign than is typical in adult SLE patients whose systemic disease was not preceded by discoid LE, the pediatric dermatologist explained.
Because so little is known about pediatric CCLE, she conducted a retrospective study including 45 affected patients seen at Lurie Children’s Hospital of Chicago. Seven of the 45 presented initially with concurrent CCLE and SLE. The remaining 38 were followed for a median of 4.4 years, during which 9 patients (24%) transitioned to SLE. Of note, that 1-in-4 risk of transformation from skin to systemic disease is spot-on with the results of the three previous small retrospective studies of pediatric CCLE, the most recent of which was reported 6 years ago from Brazil (Pediatr. Dermatol. 2008;25:163-7).
Of the 7 patients in Dr. Arkin’s study with concurrent CCLE and SLE at presentation, 6 had discoid LE, as did 34 of the 38 with only CCLE at presentation who were followed for development of SLE.
Of the 38 patients with only CCLE at presentation, 12 (32%) had disease limited to the skin throughout follow-up.
Another 17 (45%) with only CCLE at presentation developed laboratory abnormalities during follow-up without transitioning to SLE, including 14 with antinuclear antibodies. Nine of these patients developed more than two laboratory abnormalities. Notably, all had been on hydroxychloroquine ever since the onset of their CCLE. This suggests the intriguing possibility that hydroxychloroquine – an immunomodulatory agent rather than an immunosuppressant – may favorably alter the natural history of disease progression in this young population, according to Dr. Arkin. That’s been shown to be the case in studies conducted in adult military personnel, where long-term follow-up of troops randomized to hydroxychloroquine for antimalarial prophylaxis delayed the diagnosis of SLE, lowered the rate of autoantibody accumulation, retarded renal damage, reduced the risk of infection, and improved overall survival (Lupus 2007;16:401-9).
Patients with concurrent CCLE and SLE presented at a considerably younger age: a mean of 8.7 years old, compared with 13.3 years of age for those with only CCLE.
No formal guidelines exist for monitoring patients with CCLE. Based upon her study findings, Dr. Arkin recommended that "it seems reasonable" to perform a meticulous physical exam and review of systems and obtain laboratory tests on a quarterly basis, particularly in the first year following diagnosis, when the risk of transformation to SLE is highest. The quarterly lab workup should include a complete blood count and complete metabolic panel, urinalysis, complement, antiphospholipid antibodies, and antinuclear antibodies with titer.
"I think one of the big questions which has not been answered is, should all kids with CCLE get oral hydroxychloroquine? There are no studies in children. The only FDA-approved indication is for antimalarial prophylaxis, although it’s certainly used commonly off-label for pediatric CCLE and also for SLE," she said.
The major side effects of oral hydroxychloroquine in children include retinal toxicity, which is dose-dependent, along with GI upset, headaches, and vivid dreams.
Dr. Arkin observed that while a randomized trial of hydroxychloroquine to delay or prevent SLE in high-risk individuals is warranted, she was unable to identify any predictors of transformation from CCLE to SLE in her study, despite an extensive search.
"I love this study," commented SPD President Dr. Beth A. Drolet, professor of dermatology at the Medical College of Wisconsin, Milwaukee. "Your point that if transformation to SLE happens, it’s going to happen quickly – that’s enormously helpful to me clinically."
The study was funded by the SPD. Dr. Arkin reported having no financial conflicts.
COEUR D’ALENE, ID. – Pediatric chronic cutaneous lupus erythematosus transforms into systemic disease in one-quarter of cases, and if it’s going to do so, it’ll typically be within the first year after diagnosis, according to a retrospective study.
"Importantly, no one developed end-organ damage through the end of follow-up, perhaps suggesting that SLE arising in pediatric patients with CCLE follows a more indolent, benign course, just as we’ve seen in previous studies in adults with CCLE with transformation to SLE," Dr. Lisa Arkin observed at the annual meeting of the Society for Pediatric Dermatology.
If that is indeed true, it’s good news for pediatric CCLE patients and their families because it sets them apart from the others who develop SLE prior to adulthood. Twenty percent of all cases of SLE present before age 20, and in general they tend to follow a more aggressive course than adult-onset SLE, with a higher frequency of end-organ involvement necessitating sustained immunosuppression, as well as higher mortality, noted Dr. Arkin, recently appointed as head of pediatric dermatology at Rush University Medical Center, Chicago.
Pediatric CCLE is a rare and little-studied disease. Its most common form is discoid LE, characterized by scaling, telangiectasias, atrophy, follicular plugging, and scarring.
Much more is known about adult discoid LE, a far more common condition. Less than 5%-10% of adults with discoid LE develop SLE, and when they do, the systemic manifestations are more likely to be benign than is typical in adult SLE patients whose systemic disease was not preceded by discoid LE, the pediatric dermatologist explained.
Because so little is known about pediatric CCLE, she conducted a retrospective study including 45 affected patients seen at Lurie Children’s Hospital of Chicago. Seven of the 45 presented initially with concurrent CCLE and SLE. The remaining 38 were followed for a median of 4.4 years, during which 9 patients (24%) transitioned to SLE. Of note, that 1-in-4 risk of transformation from skin to systemic disease is spot-on with the results of the three previous small retrospective studies of pediatric CCLE, the most recent of which was reported 6 years ago from Brazil (Pediatr. Dermatol. 2008;25:163-7).
Of the 7 patients in Dr. Arkin’s study with concurrent CCLE and SLE at presentation, 6 had discoid LE, as did 34 of the 38 with only CCLE at presentation who were followed for development of SLE.
Of the 38 patients with only CCLE at presentation, 12 (32%) had disease limited to the skin throughout follow-up.
Another 17 (45%) with only CCLE at presentation developed laboratory abnormalities during follow-up without transitioning to SLE, including 14 with antinuclear antibodies. Nine of these patients developed more than two laboratory abnormalities. Notably, all had been on hydroxychloroquine ever since the onset of their CCLE. This suggests the intriguing possibility that hydroxychloroquine – an immunomodulatory agent rather than an immunosuppressant – may favorably alter the natural history of disease progression in this young population, according to Dr. Arkin. That’s been shown to be the case in studies conducted in adult military personnel, where long-term follow-up of troops randomized to hydroxychloroquine for antimalarial prophylaxis delayed the diagnosis of SLE, lowered the rate of autoantibody accumulation, retarded renal damage, reduced the risk of infection, and improved overall survival (Lupus 2007;16:401-9).
Patients with concurrent CCLE and SLE presented at a considerably younger age: a mean of 8.7 years old, compared with 13.3 years of age for those with only CCLE.
No formal guidelines exist for monitoring patients with CCLE. Based upon her study findings, Dr. Arkin recommended that "it seems reasonable" to perform a meticulous physical exam and review of systems and obtain laboratory tests on a quarterly basis, particularly in the first year following diagnosis, when the risk of transformation to SLE is highest. The quarterly lab workup should include a complete blood count and complete metabolic panel, urinalysis, complement, antiphospholipid antibodies, and antinuclear antibodies with titer.
"I think one of the big questions which has not been answered is, should all kids with CCLE get oral hydroxychloroquine? There are no studies in children. The only FDA-approved indication is for antimalarial prophylaxis, although it’s certainly used commonly off-label for pediatric CCLE and also for SLE," she said.
The major side effects of oral hydroxychloroquine in children include retinal toxicity, which is dose-dependent, along with GI upset, headaches, and vivid dreams.
Dr. Arkin observed that while a randomized trial of hydroxychloroquine to delay or prevent SLE in high-risk individuals is warranted, she was unable to identify any predictors of transformation from CCLE to SLE in her study, despite an extensive search.
"I love this study," commented SPD President Dr. Beth A. Drolet, professor of dermatology at the Medical College of Wisconsin, Milwaukee. "Your point that if transformation to SLE happens, it’s going to happen quickly – that’s enormously helpful to me clinically."
The study was funded by the SPD. Dr. Arkin reported having no financial conflicts.
AT THE SPD ANNUAL MEETING
Key clinical point: If pediatric chronic cutaneous lupus erythematosus is going to transform into SLE, it generally does so within the first year following diagnosis.
Major finding: During a median 4.4 years of follow-up, 9 of 38 (24%) children and adolescents with chronic cutaneous lupus erythematosus transitioned to SLE.
Data source: This single-center, retrospective study included 45 children and adolescents with chronic cutaneous lupus erythematosus.
Disclosures: This study was funded by the Society for Pediatric Dermatology. The presenter reported having no financial conflicts.
What should PCPs know about pediatric dermatology?
COEUR D’ALENE, IDAHO – Thought leaders in pediatrics, family practice medicine, and pediatric dermatology all agree: Primary care providers need to know more – a lot more – about pediatric skin conditions.
A 20-member expert committee drawn from these disciplines has reached consensus on a lengthy wish list of educational objectives. The committee members scrutinized 235 proposed objectives in 16 content areas of pediatric dermatology. Their task was to rate the importance of each of these objectives for resident physicians who plan to see children in their general practice of primary ambulatory care or urgent care.
Ultimately 72% of the items were approved by the panel, which used the Delphi method of achieving consensus, Dr. Erin Mathes reported at the annual meeting of the Society for Pediatric Dermatology.
It’s a successful initial step in the long-term goal of creating an online pediatric dermatology curriculum for primary care providers. Such a tool is badly needed because primary care physicians, not dermatologists, see most children with skin disease. By some estimates, skin complaints account for up to 30% of all primary care and emergency department visits. Moreover, primary care physicians rate their access to pediatric dermatologists as the third worst of all pediatric subspecialties, behind only child psychiatry and developmental and behavioral pediatrics.
Education in dermatology in medical school is quite limited. At the University of California, San Francisco, for example, where Dr. Mathes serves on the pediatric dermatology faculty, medical students receive a grand total of 7 hours of dermatologic education.
To help with this unmet need, the American Academy of Dermatology has created its online basic dermatology curriculum for self-directed learning. It has been a big hit with primary care providers and trainees. In 2013, the website received 317,000 page views, and 18% of the visitors to the site were international.
"But the AAD site lacks important pediatric dermatology content and is not particularly sophisticated in certain areas. There is a lot of room for improvement, and we can help out," Dr. Mathes explained by way of background to the SPD-supported curriculum development project.
Items that made the panel’s final cut generally fell into two broad categories: diagnosis and management of common conditions such as acne, warts, atopic dermatitis, reactive erythemas, and viral and bacterial skin disease; and recognition, triage, and appropriate referral of more rare or dangerous conditions, including Stevens-Johnson syndrome, vasculitis, and drug reactions.
What did not make the list were benign conditions such as lichen striatus, cysts, juvenile xanthogranuloma, and nail disorders. "A lot of the lumps and bumps weren’t deemed important," said Dr. Mathes.
Also shot down was education regarding inherited conditions, with two notable exceptions: neurofibromatosis and tuberous sclerosis. Ichthyosis and epidermolysis bullosa were not considered to be important.
A specialty-based split emerged regarding the perceived importance of learning to perform fungal cultures and other office-based diagnostic tests. The pediatricians on the expert panel felt for the most part that they shouldn’t ask pediatric residents to know how to do them, while the family physicians and pediatric dermatologists rated that as clinically important information.
The next step will be to create educational modules to address the approved educational objectives. The modules will then be evaluated in test runs involving medical residents at collaborating institutions. Partnerships are being pursued with major medical societies, including the American Academy of Pediatrics, the American Academy of Dermatology, the American Academy of Family Physicians, and the physician assistant organizations.
Several audience members at the SPD meeting rose to complain that some of the panel’s recommendations just don’t seem to make sense.
"It seems like, for example, primary care providers need to understand at least the basics of the pathophysiology of atopic dermatitis or they’ll never stop referring patients to us, they’ll never understand why we treat it the way we do, and they’ll never be able to take ownership of atopic dermatitis in some small way. It seems glaringly obvious," one pediatric dermatologist asserted.
Dr. Mathes replied: "You have a very expert opinion on this. It’s what many pediatric dermatologists would think," she said. "The pediatricians and family physicians feel differently. They would counter, ‘I do not have time to know the pathophysiology of all these things.’ They have a lot of other stuff they need to know. They have to know about the heart, the lungs, about normal development – all sorts of stuff. They have just too much to know."
The educational objectives consensus project was funded by the Society for Pediatric Dermatology. Dr. Mathes reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Thought leaders in pediatrics, family practice medicine, and pediatric dermatology all agree: Primary care providers need to know more – a lot more – about pediatric skin conditions.
A 20-member expert committee drawn from these disciplines has reached consensus on a lengthy wish list of educational objectives. The committee members scrutinized 235 proposed objectives in 16 content areas of pediatric dermatology. Their task was to rate the importance of each of these objectives for resident physicians who plan to see children in their general practice of primary ambulatory care or urgent care.
Ultimately 72% of the items were approved by the panel, which used the Delphi method of achieving consensus, Dr. Erin Mathes reported at the annual meeting of the Society for Pediatric Dermatology.
It’s a successful initial step in the long-term goal of creating an online pediatric dermatology curriculum for primary care providers. Such a tool is badly needed because primary care physicians, not dermatologists, see most children with skin disease. By some estimates, skin complaints account for up to 30% of all primary care and emergency department visits. Moreover, primary care physicians rate their access to pediatric dermatologists as the third worst of all pediatric subspecialties, behind only child psychiatry and developmental and behavioral pediatrics.
Education in dermatology in medical school is quite limited. At the University of California, San Francisco, for example, where Dr. Mathes serves on the pediatric dermatology faculty, medical students receive a grand total of 7 hours of dermatologic education.
To help with this unmet need, the American Academy of Dermatology has created its online basic dermatology curriculum for self-directed learning. It has been a big hit with primary care providers and trainees. In 2013, the website received 317,000 page views, and 18% of the visitors to the site were international.
"But the AAD site lacks important pediatric dermatology content and is not particularly sophisticated in certain areas. There is a lot of room for improvement, and we can help out," Dr. Mathes explained by way of background to the SPD-supported curriculum development project.
Items that made the panel’s final cut generally fell into two broad categories: diagnosis and management of common conditions such as acne, warts, atopic dermatitis, reactive erythemas, and viral and bacterial skin disease; and recognition, triage, and appropriate referral of more rare or dangerous conditions, including Stevens-Johnson syndrome, vasculitis, and drug reactions.
What did not make the list were benign conditions such as lichen striatus, cysts, juvenile xanthogranuloma, and nail disorders. "A lot of the lumps and bumps weren’t deemed important," said Dr. Mathes.
Also shot down was education regarding inherited conditions, with two notable exceptions: neurofibromatosis and tuberous sclerosis. Ichthyosis and epidermolysis bullosa were not considered to be important.
A specialty-based split emerged regarding the perceived importance of learning to perform fungal cultures and other office-based diagnostic tests. The pediatricians on the expert panel felt for the most part that they shouldn’t ask pediatric residents to know how to do them, while the family physicians and pediatric dermatologists rated that as clinically important information.
The next step will be to create educational modules to address the approved educational objectives. The modules will then be evaluated in test runs involving medical residents at collaborating institutions. Partnerships are being pursued with major medical societies, including the American Academy of Pediatrics, the American Academy of Dermatology, the American Academy of Family Physicians, and the physician assistant organizations.
Several audience members at the SPD meeting rose to complain that some of the panel’s recommendations just don’t seem to make sense.
"It seems like, for example, primary care providers need to understand at least the basics of the pathophysiology of atopic dermatitis or they’ll never stop referring patients to us, they’ll never understand why we treat it the way we do, and they’ll never be able to take ownership of atopic dermatitis in some small way. It seems glaringly obvious," one pediatric dermatologist asserted.
Dr. Mathes replied: "You have a very expert opinion on this. It’s what many pediatric dermatologists would think," she said. "The pediatricians and family physicians feel differently. They would counter, ‘I do not have time to know the pathophysiology of all these things.’ They have a lot of other stuff they need to know. They have to know about the heart, the lungs, about normal development – all sorts of stuff. They have just too much to know."
The educational objectives consensus project was funded by the Society for Pediatric Dermatology. Dr. Mathes reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Thought leaders in pediatrics, family practice medicine, and pediatric dermatology all agree: Primary care providers need to know more – a lot more – about pediatric skin conditions.
A 20-member expert committee drawn from these disciplines has reached consensus on a lengthy wish list of educational objectives. The committee members scrutinized 235 proposed objectives in 16 content areas of pediatric dermatology. Their task was to rate the importance of each of these objectives for resident physicians who plan to see children in their general practice of primary ambulatory care or urgent care.
Ultimately 72% of the items were approved by the panel, which used the Delphi method of achieving consensus, Dr. Erin Mathes reported at the annual meeting of the Society for Pediatric Dermatology.
It’s a successful initial step in the long-term goal of creating an online pediatric dermatology curriculum for primary care providers. Such a tool is badly needed because primary care physicians, not dermatologists, see most children with skin disease. By some estimates, skin complaints account for up to 30% of all primary care and emergency department visits. Moreover, primary care physicians rate their access to pediatric dermatologists as the third worst of all pediatric subspecialties, behind only child psychiatry and developmental and behavioral pediatrics.
Education in dermatology in medical school is quite limited. At the University of California, San Francisco, for example, where Dr. Mathes serves on the pediatric dermatology faculty, medical students receive a grand total of 7 hours of dermatologic education.
To help with this unmet need, the American Academy of Dermatology has created its online basic dermatology curriculum for self-directed learning. It has been a big hit with primary care providers and trainees. In 2013, the website received 317,000 page views, and 18% of the visitors to the site were international.
"But the AAD site lacks important pediatric dermatology content and is not particularly sophisticated in certain areas. There is a lot of room for improvement, and we can help out," Dr. Mathes explained by way of background to the SPD-supported curriculum development project.
Items that made the panel’s final cut generally fell into two broad categories: diagnosis and management of common conditions such as acne, warts, atopic dermatitis, reactive erythemas, and viral and bacterial skin disease; and recognition, triage, and appropriate referral of more rare or dangerous conditions, including Stevens-Johnson syndrome, vasculitis, and drug reactions.
What did not make the list were benign conditions such as lichen striatus, cysts, juvenile xanthogranuloma, and nail disorders. "A lot of the lumps and bumps weren’t deemed important," said Dr. Mathes.
Also shot down was education regarding inherited conditions, with two notable exceptions: neurofibromatosis and tuberous sclerosis. Ichthyosis and epidermolysis bullosa were not considered to be important.
A specialty-based split emerged regarding the perceived importance of learning to perform fungal cultures and other office-based diagnostic tests. The pediatricians on the expert panel felt for the most part that they shouldn’t ask pediatric residents to know how to do them, while the family physicians and pediatric dermatologists rated that as clinically important information.
The next step will be to create educational modules to address the approved educational objectives. The modules will then be evaluated in test runs involving medical residents at collaborating institutions. Partnerships are being pursued with major medical societies, including the American Academy of Pediatrics, the American Academy of Dermatology, the American Academy of Family Physicians, and the physician assistant organizations.
Several audience members at the SPD meeting rose to complain that some of the panel’s recommendations just don’t seem to make sense.
"It seems like, for example, primary care providers need to understand at least the basics of the pathophysiology of atopic dermatitis or they’ll never stop referring patients to us, they’ll never understand why we treat it the way we do, and they’ll never be able to take ownership of atopic dermatitis in some small way. It seems glaringly obvious," one pediatric dermatologist asserted.
Dr. Mathes replied: "You have a very expert opinion on this. It’s what many pediatric dermatologists would think," she said. "The pediatricians and family physicians feel differently. They would counter, ‘I do not have time to know the pathophysiology of all these things.’ They have a lot of other stuff they need to know. They have to know about the heart, the lungs, about normal development – all sorts of stuff. They have just too much to know."
The educational objectives consensus project was funded by the Society for Pediatric Dermatology. Dr. Mathes reported having no financial conflicts.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Emergency physicians may be overprescribing PPIs
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
AT SAEM 2014
Key clinical point: ED physicians may soon be getting an earful from advocates of the ABIM’s Choosing Wisely Program in light of new evidence of ED overprescribing of proton pump inhibitors.
Major finding: In 2001, 3% of adult visits to U.S. EDs included a prescription for a PPI. By 2010, this rate had more than doubled to 7.2%.
Data source: This was a retrospective analysis of data from the annual National Hospital Ambulatory Care Survey for 2001-2010, which provides a detailed snapshot of ED visits across the United States.
Disclosures: The presenter reported having no financial conflicts regarding his study, which was supported by institutional funds.
More Vitamin D Didn’t Beat Placebo for Kids’ Atopic Dermatitis
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
AT THE SPD ANNUAL MEETING
More vitamin D didn’t beat placebo for kids’ atopic dermatitis
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
AT THE SPD ANNUAL MEETING
Key clinical point: There may be more behind atopic dermatitis severity than just vitamin D deficiency.
Major finding: Correcting the atopic dermatitis patients’ low serum vitamin D with 2,000 IU of cholecalciferol for 3 months improved their atopic dermatitis severity scores, but only to the same extent as in placebo-treated controls.
Data source: This was a randomized, double-blind, single-center trial in which 40 atopic dermatitis patients aged 1-18 years with vitamin D insufficiency or deficiency were placed on vitamin D supplementation or placebo for 3 months.
Disclosures: The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
Body wash offers alternative to bleach baths for atopic dermatitis
COEUR D’ALENE, IDAHO – Daily use of an over-the-counter 2-minute body wash by children and adolescents with moderate to severe atopic dermatitis resulted in marked improvement in symptoms and quality of life measures, as well as impressive changes in the skin microbiome in an open-label study.
"CLn Body Wash represents a simple and effective alternative to bleach bath use for patients who have colonization by Staphylococcus aureus and who prefer showers over baths," Dr. Adelaide A. Hebert said in presenting the study results at the annual meeting of the Society for Pediatric Dermatology.
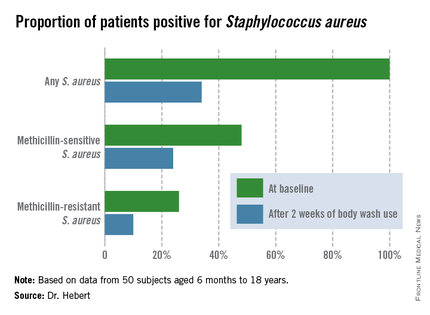
The body wash, marketed by TopMD Skin Care, is a skin-friendly gel cleanser containing sodium hypochlorite at a concentration of 0.006%.
S. aureus colonization is common on both lesional and nonlesional skin of atopic dermatitis patients. It triggers inflammation and increases severity of the skin disease.
"I view colonization by Staph as the bully of atopic dermatitis. It really pushes and annoys the disease, and that’s the way I explain it to parents," said Dr. Hebert, professor and director of pediatric dermatology at the University of Texas, Houston.
Dilute bleach baths have been shown to reduce S. aureus colonization and diminish atopic dermatitis disease severity. The typical formula is one-quarter cup of household bleach added to half a bathtub of warm water, yielding a 0.005% sodium hypochlorite solution. But many people don’t like taking baths. They prefer showers. In addition, bathtubs are uncommon in many areas outside of the United States, Dr. Hebert noted.
Ease and convenience was the impetus for the development of CLn Body Wash. The product is packaged in a 2.5-oz bottle. Patients lather a few squirts of the wash onto the skin, then rinse it off after 1-2 minutes.
To evaluate the over-the-counter product’s effectiveness, Dr. Hebert and her coinvestigators conducted a 6-week, open-label, two-center study involving 50 subjects aged 6 months to 18 years, all with moderate to severe atopic dermatitis. Their mean age was 8 years. All participants were colonized by S. aureus, but none had an active infection. They were assessed at baseline, and again after 2 weeks and 6 weeks of once-daily use of the body wash.
Improvement was marked and consistent across numerous measures of disease severity and quality of life. In addition, there was a sharp reduction after 2 weeks of use in the proportion of patients who were skin-positive for S. aureus by PCR and a comparable decrease in those who were culture-positive. The prevalence of Staphylococcus aureus colonization was cut by two-thirds.
"All along the way we were able to evoke all the parameters of improvement simply by changing the colonization status of these patients. And that was very exciting," Dr. Hebert said.
But audience member Dr. Alfred T. Lane, recipient of the annual Alvin Jacobs Award at this year’s SPD meeting, noted that he found the study less than convincing.
"I’ve seen too many open-label studies that don’t work out. I really think that to have success, you want to have a placebo group. These data look fantastic. It looks extremely exciting, but without a placebo control, I’m a little bit skeptical, to be honest," declared Dr. Lane, emeritus professor of dermatology and of pediatrics at Stanford (Calif.) University.
The study was funded by TopMD Skin Care. Dr. Hebert reported having no relevant financial conflicts.
COEUR D’ALENE, IDAHO – Daily use of an over-the-counter 2-minute body wash by children and adolescents with moderate to severe atopic dermatitis resulted in marked improvement in symptoms and quality of life measures, as well as impressive changes in the skin microbiome in an open-label study.
"CLn Body Wash represents a simple and effective alternative to bleach bath use for patients who have colonization by Staphylococcus aureus and who prefer showers over baths," Dr. Adelaide A. Hebert said in presenting the study results at the annual meeting of the Society for Pediatric Dermatology.

The body wash, marketed by TopMD Skin Care, is a skin-friendly gel cleanser containing sodium hypochlorite at a concentration of 0.006%.
S. aureus colonization is common on both lesional and nonlesional skin of atopic dermatitis patients. It triggers inflammation and increases severity of the skin disease.
"I view colonization by Staph as the bully of atopic dermatitis. It really pushes and annoys the disease, and that’s the way I explain it to parents," said Dr. Hebert, professor and director of pediatric dermatology at the University of Texas, Houston.
Dilute bleach baths have been shown to reduce S. aureus colonization and diminish atopic dermatitis disease severity. The typical formula is one-quarter cup of household bleach added to half a bathtub of warm water, yielding a 0.005% sodium hypochlorite solution. But many people don’t like taking baths. They prefer showers. In addition, bathtubs are uncommon in many areas outside of the United States, Dr. Hebert noted.
Ease and convenience was the impetus for the development of CLn Body Wash. The product is packaged in a 2.5-oz bottle. Patients lather a few squirts of the wash onto the skin, then rinse it off after 1-2 minutes.
To evaluate the over-the-counter product’s effectiveness, Dr. Hebert and her coinvestigators conducted a 6-week, open-label, two-center study involving 50 subjects aged 6 months to 18 years, all with moderate to severe atopic dermatitis. Their mean age was 8 years. All participants were colonized by S. aureus, but none had an active infection. They were assessed at baseline, and again after 2 weeks and 6 weeks of once-daily use of the body wash.

Improvement was marked and consistent across numerous measures of disease severity and quality of life. In addition, there was a sharp reduction after 2 weeks of use in the proportion of patients who were skin-positive for S. aureus by PCR and a comparable decrease in those who were culture-positive. The prevalence of Staphylococcus aureus colonization was cut by two-thirds.
"All along the way we were able to evoke all the parameters of improvement simply by changing the colonization status of these patients. And that was very exciting," Dr. Hebert said.
But audience member Dr. Alfred T. Lane, recipient of the annual Alvin Jacobs Award at this year’s SPD meeting, noted that he found the study less than convincing.
"I’ve seen too many open-label studies that don’t work out. I really think that to have success, you want to have a placebo group. These data look fantastic. It looks extremely exciting, but without a placebo control, I’m a little bit skeptical, to be honest," declared Dr. Lane, emeritus professor of dermatology and of pediatrics at Stanford (Calif.) University.
The study was funded by TopMD Skin Care. Dr. Hebert reported having no relevant financial conflicts.
COEUR D’ALENE, IDAHO – Daily use of an over-the-counter 2-minute body wash by children and adolescents with moderate to severe atopic dermatitis resulted in marked improvement in symptoms and quality of life measures, as well as impressive changes in the skin microbiome in an open-label study.
"CLn Body Wash represents a simple and effective alternative to bleach bath use for patients who have colonization by Staphylococcus aureus and who prefer showers over baths," Dr. Adelaide A. Hebert said in presenting the study results at the annual meeting of the Society for Pediatric Dermatology.

The body wash, marketed by TopMD Skin Care, is a skin-friendly gel cleanser containing sodium hypochlorite at a concentration of 0.006%.
S. aureus colonization is common on both lesional and nonlesional skin of atopic dermatitis patients. It triggers inflammation and increases severity of the skin disease.
"I view colonization by Staph as the bully of atopic dermatitis. It really pushes and annoys the disease, and that’s the way I explain it to parents," said Dr. Hebert, professor and director of pediatric dermatology at the University of Texas, Houston.
Dilute bleach baths have been shown to reduce S. aureus colonization and diminish atopic dermatitis disease severity. The typical formula is one-quarter cup of household bleach added to half a bathtub of warm water, yielding a 0.005% sodium hypochlorite solution. But many people don’t like taking baths. They prefer showers. In addition, bathtubs are uncommon in many areas outside of the United States, Dr. Hebert noted.
Ease and convenience was the impetus for the development of CLn Body Wash. The product is packaged in a 2.5-oz bottle. Patients lather a few squirts of the wash onto the skin, then rinse it off after 1-2 minutes.
To evaluate the over-the-counter product’s effectiveness, Dr. Hebert and her coinvestigators conducted a 6-week, open-label, two-center study involving 50 subjects aged 6 months to 18 years, all with moderate to severe atopic dermatitis. Their mean age was 8 years. All participants were colonized by S. aureus, but none had an active infection. They were assessed at baseline, and again after 2 weeks and 6 weeks of once-daily use of the body wash.

Improvement was marked and consistent across numerous measures of disease severity and quality of life. In addition, there was a sharp reduction after 2 weeks of use in the proportion of patients who were skin-positive for S. aureus by PCR and a comparable decrease in those who were culture-positive. The prevalence of Staphylococcus aureus colonization was cut by two-thirds.
"All along the way we were able to evoke all the parameters of improvement simply by changing the colonization status of these patients. And that was very exciting," Dr. Hebert said.
But audience member Dr. Alfred T. Lane, recipient of the annual Alvin Jacobs Award at this year’s SPD meeting, noted that he found the study less than convincing.
"I’ve seen too many open-label studies that don’t work out. I really think that to have success, you want to have a placebo group. These data look fantastic. It looks extremely exciting, but without a placebo control, I’m a little bit skeptical, to be honest," declared Dr. Lane, emeritus professor of dermatology and of pediatrics at Stanford (Calif.) University.
The study was funded by TopMD Skin Care. Dr. Hebert reported having no relevant financial conflicts.
AT THE SPD ANNUAL MEETING
Key clinical point: An effective and convenient alternative is available for atopic dermatitis patients who find bleach baths a hassle or not possible.
Major finding: Mean scores on the Eczema Area and Severity Index dropped by 46% after patients with moderate to severe atopic dermatitis used an OTC daily body wash for 6 weeks. Scores on the Children’s Dermatologic Life Quality Index improved by 38%.
Data source: A 6-week, open-label, two-center study including 50 pediatric patients with moderate to severe atopic dermatitis and colonization by S. aureus.
Disclosures: The study was funded by TopMD Skin Care, which markets the OTC body wash. Dr. Hebert reported having no relevant financial conflicts.
Ped derm patients embrace cosmetic camouflage
COEUR D’ALENE, IDAHO – The impaired quality of life so common among children and teens with visible skin disorders is significantly improved through instruction in the use of cosmetic camouflage, according to data from a 6-month prospective study.
"We believe cosmetic camouflage may allow patients a protective window of time in order to adapt to or accept a new visible skin condition," Dr. Michele Ramien said at the annual meeting of the Society for Pediatric Dermatology.
"Visible skin conditions are a source of emotional and psychologic stress," she added. "Amongst children, differences are quickly identified and questioned by peers. The pediatric burn literature shows that visible scarring results in an array of negative responses: Stares, avoidance, teasing, bullying."
Dr. Ramien reported data from 38 patients aged 5-18 years with vascular or pigmentary disorders visible while wearing a T-shirt and pants. Two-thirds of the individuals were light skinned, and two-thirds of the skin disorders were facial. All but three of the patients were female. They were taught to use cosmetic camouflage by hospital-based cosmeticians, and they received a free 6-month supply of the products.
Mean scores on the Children's Dermatology Life Quality Index improved from 5.1 (out of a theoretically possible 30) at baseline to 2.1 at 6 months. The scores of children with pigmentary conditions improved from a mean of 6.2 to 3.2, while the scores of patients with vascular anomalies improved from 4.1 at baseline to 1.0 at 6 months.
The quality-of-life improvement was comparable in dark- and light-skinned patients, according to Dr. Ramien of the University of Montreal.
Patient surveys at 1 and 6 months showed cosmetic camouflage was well tolerated and used by the majority of patients. Indeed, all but one tried the prescribed regimen during the first month. The majority of subjects used cosmetic camouflage on a daily to once-weekly basis. At the 6-month mark, all but seven patients continued to use cosmetic camouflage.
Cosmetic camouflage entails the skillful application of specialized, commercially available products that are waterproof, are opaque, adhere to damaged skin, and will last for 8-16 hours. There is ample published evidence of the quality-of-life benefits of cosmetic camouflage in adults with disfiguring skin conditions, but little evidence of effectiveness in children until recently, Dr. Ramien observed.
"The proposed drawbacks of cosmetic camouflage that you may come across in the older literature – poor coverage of textural differences and shame related to concealing one’s true identity – those are not supported in the newer literature," she said.
Physicians without ready access to a hospital-based cosmetician have several other options.
"We realized that community dermatologists are really going to be the introduction point for the majority of pediatric patients with visible skin anomalies who might benefit from cosmetic camouflage," Dr. Ramien said. "Anyone who is interested in becoming further informed can contact representatives of the various companies that market the products, or networks of cosmeticians and aestheticians in your area who are interested in developing these services for your patients. Even the online websites of many of the cosmetic camouflage producers can be very useful in learning more about what’s available," she emphasized.
Dr. Ramien reported having no financial conflicts regarding this study, which was supported by institutional funds.
COEUR D’ALENE, IDAHO – The impaired quality of life so common among children and teens with visible skin disorders is significantly improved through instruction in the use of cosmetic camouflage, according to data from a 6-month prospective study.
"We believe cosmetic camouflage may allow patients a protective window of time in order to adapt to or accept a new visible skin condition," Dr. Michele Ramien said at the annual meeting of the Society for Pediatric Dermatology.
"Visible skin conditions are a source of emotional and psychologic stress," she added. "Amongst children, differences are quickly identified and questioned by peers. The pediatric burn literature shows that visible scarring results in an array of negative responses: Stares, avoidance, teasing, bullying."
Dr. Ramien reported data from 38 patients aged 5-18 years with vascular or pigmentary disorders visible while wearing a T-shirt and pants. Two-thirds of the individuals were light skinned, and two-thirds of the skin disorders were facial. All but three of the patients were female. They were taught to use cosmetic camouflage by hospital-based cosmeticians, and they received a free 6-month supply of the products.
Mean scores on the Children's Dermatology Life Quality Index improved from 5.1 (out of a theoretically possible 30) at baseline to 2.1 at 6 months. The scores of children with pigmentary conditions improved from a mean of 6.2 to 3.2, while the scores of patients with vascular anomalies improved from 4.1 at baseline to 1.0 at 6 months.
The quality-of-life improvement was comparable in dark- and light-skinned patients, according to Dr. Ramien of the University of Montreal.
Patient surveys at 1 and 6 months showed cosmetic camouflage was well tolerated and used by the majority of patients. Indeed, all but one tried the prescribed regimen during the first month. The majority of subjects used cosmetic camouflage on a daily to once-weekly basis. At the 6-month mark, all but seven patients continued to use cosmetic camouflage.
Cosmetic camouflage entails the skillful application of specialized, commercially available products that are waterproof, are opaque, adhere to damaged skin, and will last for 8-16 hours. There is ample published evidence of the quality-of-life benefits of cosmetic camouflage in adults with disfiguring skin conditions, but little evidence of effectiveness in children until recently, Dr. Ramien observed.
"The proposed drawbacks of cosmetic camouflage that you may come across in the older literature – poor coverage of textural differences and shame related to concealing one’s true identity – those are not supported in the newer literature," she said.
Physicians without ready access to a hospital-based cosmetician have several other options.
"We realized that community dermatologists are really going to be the introduction point for the majority of pediatric patients with visible skin anomalies who might benefit from cosmetic camouflage," Dr. Ramien said. "Anyone who is interested in becoming further informed can contact representatives of the various companies that market the products, or networks of cosmeticians and aestheticians in your area who are interested in developing these services for your patients. Even the online websites of many of the cosmetic camouflage producers can be very useful in learning more about what’s available," she emphasized.
Dr. Ramien reported having no financial conflicts regarding this study, which was supported by institutional funds.
COEUR D’ALENE, IDAHO – The impaired quality of life so common among children and teens with visible skin disorders is significantly improved through instruction in the use of cosmetic camouflage, according to data from a 6-month prospective study.
"We believe cosmetic camouflage may allow patients a protective window of time in order to adapt to or accept a new visible skin condition," Dr. Michele Ramien said at the annual meeting of the Society for Pediatric Dermatology.
"Visible skin conditions are a source of emotional and psychologic stress," she added. "Amongst children, differences are quickly identified and questioned by peers. The pediatric burn literature shows that visible scarring results in an array of negative responses: Stares, avoidance, teasing, bullying."
Dr. Ramien reported data from 38 patients aged 5-18 years with vascular or pigmentary disorders visible while wearing a T-shirt and pants. Two-thirds of the individuals were light skinned, and two-thirds of the skin disorders were facial. All but three of the patients were female. They were taught to use cosmetic camouflage by hospital-based cosmeticians, and they received a free 6-month supply of the products.
Mean scores on the Children's Dermatology Life Quality Index improved from 5.1 (out of a theoretically possible 30) at baseline to 2.1 at 6 months. The scores of children with pigmentary conditions improved from a mean of 6.2 to 3.2, while the scores of patients with vascular anomalies improved from 4.1 at baseline to 1.0 at 6 months.
The quality-of-life improvement was comparable in dark- and light-skinned patients, according to Dr. Ramien of the University of Montreal.
Patient surveys at 1 and 6 months showed cosmetic camouflage was well tolerated and used by the majority of patients. Indeed, all but one tried the prescribed regimen during the first month. The majority of subjects used cosmetic camouflage on a daily to once-weekly basis. At the 6-month mark, all but seven patients continued to use cosmetic camouflage.
Cosmetic camouflage entails the skillful application of specialized, commercially available products that are waterproof, are opaque, adhere to damaged skin, and will last for 8-16 hours. There is ample published evidence of the quality-of-life benefits of cosmetic camouflage in adults with disfiguring skin conditions, but little evidence of effectiveness in children until recently, Dr. Ramien observed.
"The proposed drawbacks of cosmetic camouflage that you may come across in the older literature – poor coverage of textural differences and shame related to concealing one’s true identity – those are not supported in the newer literature," she said.
Physicians without ready access to a hospital-based cosmetician have several other options.
"We realized that community dermatologists are really going to be the introduction point for the majority of pediatric patients with visible skin anomalies who might benefit from cosmetic camouflage," Dr. Ramien said. "Anyone who is interested in becoming further informed can contact representatives of the various companies that market the products, or networks of cosmeticians and aestheticians in your area who are interested in developing these services for your patients. Even the online websites of many of the cosmetic camouflage producers can be very useful in learning more about what’s available," she emphasized.
Dr. Ramien reported having no financial conflicts regarding this study, which was supported by institutional funds.
AT THE SPD ANNUAL MEETING
Key clinical point: Cosmetic camouflage can diminish impairments in self-esteem and quality of life in children and adolescents with visible skin conditions.
Major finding: Mean scores on the Children’s Dermatology Life Quality Index improved from 5.1 at baseline to 2.1 6 months after instruction in the use of cosmetic camouflage.
Data source: A prospective study of 38 pediatric patients with visible skin conditions who were taught how to use cosmetic camouflage and then followed for 6 months.
Disclosures: The study was supported by institutional funds. The presenter reported having no financial conflicts.
Novel oral testosterone undecanoate under FDA review
CHICAGO – A unique investigational oral testosterone replacement product for hypogonadal men showed efficacy and safety comparable to a commercially available testosterone gel in two phase III clinical trials, which drew considerable interest when presented at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
This formulation of oral testosterone undecanoate, known as Rextoro, uses proprietary self-emulsifying technology to provide more consistent absorption and a longer half-life than oral testosterone undecanoate products long available outside the United States.
The new product fulfills an unmet need for an effective and convenient treatment for male hypogonadism that avoids the risk of unintended testosterone transference to women and children inherent in currently marketed topical testosterone products, said lead investigator Dr. Ronald S. Swerdloff, professor of medicine at the University of California, Los Angeles, and chief of the division of endocrinology at Harbor-UCLA Medical Center.
"An oral testosterone replacement therapy would be the preferred choice for many hypogonal men," he said.
Clarus Therapeutics filed for marketing approval for Rextoro with the Food and Drug Administration earlier this year on the strength of the two phase III studies. In addition, the company is sponsoring a supplemental phase IV, open-label, 12-month safety study to be reported later this summer.
The phase IV study is being conducted to resolve safety questions raised in one of the two phase III studies presented at ICE/ENDO 2014. In this 325-patient, open-label, 12-month multicenter trial in which patients were randomized to the investigational oral testosterone or to 1% AndroGel, the oral therapy group experienced a 22% reduction from baseline in HDL, compared with a 12% decrease with the testosterone gel. In addition, two patients in the oral testosterone group had an acute MI; none did in the topical therapy group. Hematocrit levels rose by 3% in oral testosterone–treated patients, although mean values remained within the normal range.
LDL and total cholesterol, triglyceride, C-reactive protein, lipoprotein-associated phospholipase A2, and prostate-specific antigen levels did not differ between the two treatment groups.
Moreover, in the second and pivotal phase III trial, which utilized a revised Rextoro dose-titration algorithm, the reduction in HDL over the course of 114 days of treatment was a more modest 9%. This second trial was a single-arm study involving 144 Rextoro-treated patients.
The revised dose-adjustment algorithm involved starting treatment at 200 mg of oral Rextoro twice daily, then measuring serum testosterone 3-5 hours after the morning dose on days 30 and 72. A value outside the range of 200-750 ng/dL triggered a 50-mg increase or decrease. When this algorithm was used, 75% of treated patients were maintained within the target eugonadal range, compared with 84% of the oral testosterone group and 79% of the testosterone gel–treated patients in the first phase III trial. Under the revised algorithm, only 3% of patients experienced a transient serum peak testosterone excursion greater than 2,500 ng/dL, Dr. Swedloff reported.
The phase III trials were funded by Clarus. Dr. Swerdloff is a consultant to Clarus and Endo Pharmaceuticals.
CHICAGO – A unique investigational oral testosterone replacement product for hypogonadal men showed efficacy and safety comparable to a commercially available testosterone gel in two phase III clinical trials, which drew considerable interest when presented at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
This formulation of oral testosterone undecanoate, known as Rextoro, uses proprietary self-emulsifying technology to provide more consistent absorption and a longer half-life than oral testosterone undecanoate products long available outside the United States.
The new product fulfills an unmet need for an effective and convenient treatment for male hypogonadism that avoids the risk of unintended testosterone transference to women and children inherent in currently marketed topical testosterone products, said lead investigator Dr. Ronald S. Swerdloff, professor of medicine at the University of California, Los Angeles, and chief of the division of endocrinology at Harbor-UCLA Medical Center.
"An oral testosterone replacement therapy would be the preferred choice for many hypogonal men," he said.
Clarus Therapeutics filed for marketing approval for Rextoro with the Food and Drug Administration earlier this year on the strength of the two phase III studies. In addition, the company is sponsoring a supplemental phase IV, open-label, 12-month safety study to be reported later this summer.
The phase IV study is being conducted to resolve safety questions raised in one of the two phase III studies presented at ICE/ENDO 2014. In this 325-patient, open-label, 12-month multicenter trial in which patients were randomized to the investigational oral testosterone or to 1% AndroGel, the oral therapy group experienced a 22% reduction from baseline in HDL, compared with a 12% decrease with the testosterone gel. In addition, two patients in the oral testosterone group had an acute MI; none did in the topical therapy group. Hematocrit levels rose by 3% in oral testosterone–treated patients, although mean values remained within the normal range.
LDL and total cholesterol, triglyceride, C-reactive protein, lipoprotein-associated phospholipase A2, and prostate-specific antigen levels did not differ between the two treatment groups.
Moreover, in the second and pivotal phase III trial, which utilized a revised Rextoro dose-titration algorithm, the reduction in HDL over the course of 114 days of treatment was a more modest 9%. This second trial was a single-arm study involving 144 Rextoro-treated patients.
The revised dose-adjustment algorithm involved starting treatment at 200 mg of oral Rextoro twice daily, then measuring serum testosterone 3-5 hours after the morning dose on days 30 and 72. A value outside the range of 200-750 ng/dL triggered a 50-mg increase or decrease. When this algorithm was used, 75% of treated patients were maintained within the target eugonadal range, compared with 84% of the oral testosterone group and 79% of the testosterone gel–treated patients in the first phase III trial. Under the revised algorithm, only 3% of patients experienced a transient serum peak testosterone excursion greater than 2,500 ng/dL, Dr. Swedloff reported.
The phase III trials were funded by Clarus. Dr. Swerdloff is a consultant to Clarus and Endo Pharmaceuticals.
CHICAGO – A unique investigational oral testosterone replacement product for hypogonadal men showed efficacy and safety comparable to a commercially available testosterone gel in two phase III clinical trials, which drew considerable interest when presented at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
This formulation of oral testosterone undecanoate, known as Rextoro, uses proprietary self-emulsifying technology to provide more consistent absorption and a longer half-life than oral testosterone undecanoate products long available outside the United States.
The new product fulfills an unmet need for an effective and convenient treatment for male hypogonadism that avoids the risk of unintended testosterone transference to women and children inherent in currently marketed topical testosterone products, said lead investigator Dr. Ronald S. Swerdloff, professor of medicine at the University of California, Los Angeles, and chief of the division of endocrinology at Harbor-UCLA Medical Center.
"An oral testosterone replacement therapy would be the preferred choice for many hypogonal men," he said.
Clarus Therapeutics filed for marketing approval for Rextoro with the Food and Drug Administration earlier this year on the strength of the two phase III studies. In addition, the company is sponsoring a supplemental phase IV, open-label, 12-month safety study to be reported later this summer.
The phase IV study is being conducted to resolve safety questions raised in one of the two phase III studies presented at ICE/ENDO 2014. In this 325-patient, open-label, 12-month multicenter trial in which patients were randomized to the investigational oral testosterone or to 1% AndroGel, the oral therapy group experienced a 22% reduction from baseline in HDL, compared with a 12% decrease with the testosterone gel. In addition, two patients in the oral testosterone group had an acute MI; none did in the topical therapy group. Hematocrit levels rose by 3% in oral testosterone–treated patients, although mean values remained within the normal range.
LDL and total cholesterol, triglyceride, C-reactive protein, lipoprotein-associated phospholipase A2, and prostate-specific antigen levels did not differ between the two treatment groups.
Moreover, in the second and pivotal phase III trial, which utilized a revised Rextoro dose-titration algorithm, the reduction in HDL over the course of 114 days of treatment was a more modest 9%. This second trial was a single-arm study involving 144 Rextoro-treated patients.
The revised dose-adjustment algorithm involved starting treatment at 200 mg of oral Rextoro twice daily, then measuring serum testosterone 3-5 hours after the morning dose on days 30 and 72. A value outside the range of 200-750 ng/dL triggered a 50-mg increase or decrease. When this algorithm was used, 75% of treated patients were maintained within the target eugonadal range, compared with 84% of the oral testosterone group and 79% of the testosterone gel–treated patients in the first phase III trial. Under the revised algorithm, only 3% of patients experienced a transient serum peak testosterone excursion greater than 2,500 ng/dL, Dr. Swedloff reported.
The phase III trials were funded by Clarus. Dr. Swerdloff is a consultant to Clarus and Endo Pharmaceuticals.
AT ICE/ENDO 2014
Key clinical point: Hypogonadal men may gain access to a convenient oral form of testosterone replacement therapy that avoids the risk of medication transference to women and children posed by current topical testosterone products.
Major finding: An investigational oral testosterone undecanoate showed safety and efficacy comparable to a commercially available testosterone gel in two phase III clinical trials.
Data source: One open-label, multicenter phase III study in 325 hypogonadal men randomized to 12 months of daily investigational oral testosterone replacement or commercially available 1% testosterone gel, and a second phase III single-arm trial in which 144 subjects with male hypogonadism were treated with the oral agent for 114 days using a revised dose-adjustment scheme.
Disclosures: The studies were sponsored by Clarus Therapeutics. The presenter is a consultant to the company.