User login
Aspirin’s benefits may be blunted in black women
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the university’s Center for Women’s Health. She reported no financial conflicts.
Dr. Jennifer Cox, FCCP, comments: Although most results were not statistically significant, Dr. Alghothani presents an interesting pilot study that revealed the blunted response in anti-inflammatory markers to aspirin between African American women and white women. More studies are needed before changing prescribing practices, but it is clear that genetics, ethnicity, and drug metabolism should be on the forefront of drug research for more that just antineoplastic therapies.
Dr. Jennifer Cox, FCCP, comments: Although most results were not statistically significant, Dr. Alghothani presents an interesting pilot study that revealed the blunted response in anti-inflammatory markers to aspirin between African American women and white women. More studies are needed before changing prescribing practices, but it is clear that genetics, ethnicity, and drug metabolism should be on the forefront of drug research for more that just antineoplastic therapies.
Dr. Jennifer Cox, FCCP, comments: Although most results were not statistically significant, Dr. Alghothani presents an interesting pilot study that revealed the blunted response in anti-inflammatory markers to aspirin between African American women and white women. More studies are needed before changing prescribing practices, but it is clear that genetics, ethnicity, and drug metabolism should be on the forefront of drug research for more that just antineoplastic therapies.
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the university’s Center for Women’s Health. She reported no financial conflicts.
CHICAGO – Postmenopausal African American women with subclinical atherosclerosis appear to be more resistant to the anti-inflammatory effects of daily aspirin than their white counterparts.
In a 6-month, double-blind, placebo-controlled pilot study, daily aspirin at 325 mg showed essentially no impact on high-sensitivity C-reactive protein (hsCRP ) levels in the African American women. Moreover, their levels of interleukin-6 (IL-6) shot up while on aspirin. In contrast, levels of both proinflammatory markers declined markedly with aspirin therapy in the white women, Dr. Nora Alghothani reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"Given apparent ethnic differences in response to aspirin-mediated anti-inflammatory benefits, perhaps a higher dose of aspirin may be required in African American women already at higher risk of inflammatory disease processes in order to reduce cardiovascular disease outcomes and lessen disparities," concluded Dr. Alghothani, of the department of endocrinology at the Ohio State University in Columbus.
This remark lit a four-alarm fire among audience members. They were quick to emphasize that aspirin at doses greater than 325 mg/day is associated with a sharply increased risk of bleeding and should thus not be considered as part of an individualized cardioprevention strategy for African American women unless and until there is solid evidence that the benefits outweigh the risks.
Dr. Alghothani concurred that a large-scale dose-response study is needed. In the meantime, though, the take home message of her pilot study is that physicians should not necessarily expect the same robust cardiovascular benefits with daily aspirin in their postmenopausal African American patients as in other populations, she added.
The pilot study included 42 postmenopausal, nondiabetic women with evidence of subclinical atherosclerosis based upon carotid intimal medial thickness measurements. Half were African American; half were white. Participants in each group were randomized in double-blind fashion to 6 months of aspirin at 325 mg/day or placebo, with fasting blood samples and anthropomorphic measurements obtained at baseline and 6 months. Consistent with findings from much larger studies, the African American women were heavier, with a mean body mass index of 32.8 kg/m2, compared with 27.8 kg/m2 for the white women. The African Americans also had significantly lower triglycerides and higher apolopoprotein A-I levels; however, the two groups didn’t differ in terms of fasting insulin or glucose, high-density lipoprotein, low-density lipoprotein, or blood pressure.
In the aspirin-treated African American women, levels of hsCRP remained static over time, going from a mean of 4.53 mg/L at baseline to 4.62 mg/L at 6 months. In placebo-treated African American women, however, hsCRP jumped from 3.34 mg/L at baseline to 8.36 mg/L at follow-up.
The mean hsCRP in white women on aspirin dropped from 2.13 to 1.6 mg/L over the course of 6 months, while with placebo it went from 2.19 to 2.69 mg/L.
Levels of IL-6 in aspirin-treated African American women climbed from 0.93 pg/mL at baseline to 2.56 pg/mL at 6 months. In contrast, mean IL-6 levels in white women on daily aspirin fell from 2.69 to 1.39 pg/mL. White women on placebo experienced a rise in IL-6 from 0.58 to 2.97 pg/mL.
Most of these differences didn’t achieve statistical significance because of the small sample size, but the consistent trends suggest an overall blunted response to the anti-inflammatory effects of aspirin among African Americans, according to Dr. Alghothani. She added that these findings might help explain the well-documented ethnic disparities in cardiovascular outcomes, whereby African American women have a significantly higher cardiovascular mortality rate than white women despite on average having higher HDL and lower triglycerides.
Her study was funded by the university’s Center for Women’s Health. She reported no financial conflicts.
Key clinical point: Postmenopausal African American women may have a blunted response to the anti-inflammatory effects of aspirin at 325 mg/day.
Major finding: Mean levels of hsCRP dropped by 25% and IL-6 decreased by 48% in white women over the course of 6 months of aspirin at 325 mg/day. In contrast, CRP remained essentially unchanged despite daily aspirin in African American women, and their IL-6 levels rose.
Data source: This randomized, double-blind, placebo-controlled pilot study included 42 postmenopausal, nondiabetic women; half were African American, half were white. All had documented subclinical atherosclerosis. Subjects received aspirin at 325 mg/day and were studied over the course of 6 months.
Disclosures: The study was supported by the Ohio State University Center for Women’s Health. The presenter reported having no financial conflicts.
Mobile Teledermatology Predicted to Be a Game Changer
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Mobile teledermatology predicted to be a game changer
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
COEUR D’ALENE, IDAHO – Teledermatology is gathering steam – and supportive data – as a potentially invaluable way to address the overlapping problems of a dermatology workforce shortage, gruelingly long wait times for appointments, limited dermatologic skills on the part of busy primary care physicians, and the anticipated sharply increased demand for dermatologic services as the Affordable Care Act brings many millions of new patients into the ranks of the insured.
But, in the main, it won’t be telemedicine in the familiar form that physicians have come to think of: that is, as remote, face-to-face, real-time video conferencing. The future lies in mobile teledermatology, also known as store and forward, Dr. Carrie L. Kovarik asserted at the annual meeting of the Society for Pediatric Dermatology.
Store-and-forward teledermatology takes advantage of the ubiquity of smart phones and the dazzlingly high quality of their photographic capability. Here’s how it works under AccessDerm, a mobile store-and-forward teledermatology platform developed by the American Academy of Dermatology. Dr. Kovarik, a University of Pennsylvania, Philadelphia, dermatologist and a pioneer of teledermatology in Africa, heads the AAD AccessDerm initiative.
Mobile teledermatology is conducted between a primary care provider and a remote consulting dermatologist. Using the AccessDerm app, the primary care physician submits a case, providing clinical images, and filling out a standardized template to provide mandatory information regarding clinical history.
At present, AccessDerm is connected in 19 states and the District of Columbia. Any AAD member in those locales can serve as a voluntary teledermatology consultant. At the University of Pennsylvania, the dermatology department faculty and residents serve as a pool as the cases come in, picking them up as their workflow permits.
The program operates in 14 Philadelphia health clinics, including all 10 city public health clinics. It has been a hit with patients, many of whom are immigrants who are reluctant to go to the university dermatology clinic and who are far more comfortable in their local clinic, where translators are available. The city council loves the program as well.
In an as-yet unpublished prospective study involving 202 AccessDerm consultations in which the primary care provider was asked in submitting the case to provide their thoughts regarding diagnosis and management, the Penn dermatology consultants overturned the primary care providers’ opinions in the majority of instances. Indeed, the diagnostic concordance rate was just 22%, and the concordance rate regarding management was 23%.
A total of 77% of the cases were managed via teledermatology alone. In the other 23%, an in-person dermatology consultation was recommended; this rate was higher than when primary care providers are taught to do careful skin biopsies and submit them as part of the teledermatology consult.
"Typically, if you teach primary care providers to do biopsies, you need to see patients only 5%-10% of the time as a safety net for those complex cases in a teledermatology program," according to Dr. Kovarik.
The primary care providers stated that, in the absence of teledermatology, 61% of the patients would have not had any access to dermatologic care.
"We’re providing a lot of access where there was no access," Dr. Kovarik observed.
An added benefit of the Philadelphia mobile teledermatology experience is that the primary care clinicians are getting better and better at dermatology.
"They’re asking us harder questions now," she said. "They’re not asking us about eczema anymore."
In another recent study, Dr. Kovarik and her Penn coworkers demonstrated that mobile teledermatology is a reliable and efficient way to triage inpatient dermatology consultations. This is an issue of practical importance to office-based dermatologists. Most of the time, if they get asked to consult on a hospital inpatient, they feel obligated to go to the hospital that day because they can’t tell from the phone conversation how urgent the situation really is.
In this prospective study of 50 adult inpatients for whom a dermatology consultation was requested, all participants were evaluated separately by an in-person dermatologist and two independent teledermatologists. At least 90% of the time, the on-site dermatologist recommended the patient be seen that day, so did the teledermatologists. When the in-person dermatologist recommended a biopsy, 95% of the time the teledermatologists did, too. The teledermatologists were able to triage 60% of the consultations to be seen the next day or later, enabling office-based teledermatologists to bunch some of their inpatient consultations (JAMA Dermatology 2014;150:419-24).
"At Penn, we’re big advocates of teletriage. We’re setting up a system where we have all the consults come to us as teledermatology. We’ll look at the case and say we can help the primary care provider treat that problem, we can triage it to a certain clinic, or the patient could go see the next available dermatologist. But everyone is going to be seen by teledermatology first so we can put [the patients] in the right place to get the care they need. Otherwise, the consults come in with a note saying ‘rash on the right leg’ and who knows how bad it is," Dr. Kovarik said.
Right now only 9 states provide reimbursement for telemedicine services, although another 30 states are considering it. Proposed federal legislation would provide national coverage; Dr. Kovarik sees passage of such a law as an essential major step forward in gaining widespread acceptance of mobile teledermatology.
Asked about billing, she replied that she is aware of one major health insurer interested in providing coverage for teledermatology, in part so they can promote it in consumer advertising as an example of their high-quality care.
"I don’t see teledermatology as part of direct fee for service in the next decade. I see it more as part of a value-based health care plan. I think patients like it, and it adds value. So I think it will be part of the new health care landscape before too long," Dr. Kovarik predicted.
She reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
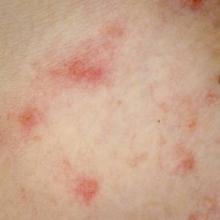
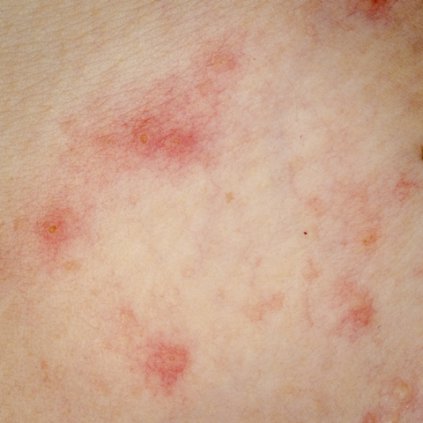
Psoriasiform lesions in Kawasaki disease may spell trouble
COEUR D’ALENE, IDAHO – The psoriasiform eruptions that occur in a subgroup of patients with Kawasaki disease during the acute or subacute phase may be a red flag for more severe coronary artery involvement, according to a retrospective, case-control study.
Another striking feature of these psoriasiform lesions is that they go into remission. No recurrences were seen in the study population during up to 13 years of follow-up, in marked contrast to classic psoriasis, a chronic disease, Dr. Wynnis L. Tom observed at the annual meeting of the Society for Pediatric Dermatology.
She presented what she believes to be the first study to formally compare the psoriasiform lesions arising in a minority of Kawasaki disease (KD) patients with the lesions of classic psoriasis. The study population consisted of 11 KD patients with psoriasiform eruptions whose median age was 1.9 years, 22 matched controls with KD and no psoriasiform lesions, and another 22 matched controls with psoriasis but not KD.
Kawasaki disease patients who developed psoriasiform eruptions had significantly more dilated coronary arteries than did those who did not, as reflected in their median maximal echocardiographic z-score of 2.7, compared with 1.8 in controls. A z-score greater than 2.5 is deemed to indicate clinically significant coronary artery dilation, noted Dr. Tom, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego.
The psoriasiform eruptions resolved within 13 months in all 11 affected KD patients, with no recurrences. In contrast, only 5 of 22 controls with classic psoriasis experienced remission during follow-up of up to 6 years.
The cutaneous distribution of the psoriasiform lesions in patients with KD was also distinctive. The eruptions were significantly less common in the head, neck, and diaper area than is the case in classic psoriasis. In addition, patients with KD and psoriasiform eruptions were significantly less likely to be overweight or obese than were controls with classic psoriasis.
Skin biopsies read by a blinded dermatopathologist showed that the psoriasiform lesions that arose during KD demonstrated suprabasilar staining of keratin 16 and increased expression of Ki-67 antigen. They differed from classic psoriasis lesions in that they displayed more crusting, serum, and an increased prevalence of bacteria at the epidermis.
There was no significant difference between KD patients with and without psoriasiform eruptions in terms of maximum levels of C-reactive protein, erythrocyte sedimentation rate, and other inflammatory markers. However, patients with psoriasiform lesions required significantly more time for their gamma-glutamyl transferase level and platelet count to return to normal.
One possible explanation for the distinct phenotype of these psoriasiform eruptions is that KD brings forth the skin lesions in patients who are predisposed to psoriasis, but without causing typical chronic psoriasis because the underlying KD is self-limited. But further study with larger numbers of patients is required for clarification, according to Dr. Tom.
Her work is supported by a career development award from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. She reported having no financial conflicts regarding this study.
COEUR D’ALENE, IDAHO – The psoriasiform eruptions that occur in a subgroup of patients with Kawasaki disease during the acute or subacute phase may be a red flag for more severe coronary artery involvement, according to a retrospective, case-control study.
Another striking feature of these psoriasiform lesions is that they go into remission. No recurrences were seen in the study population during up to 13 years of follow-up, in marked contrast to classic psoriasis, a chronic disease, Dr. Wynnis L. Tom observed at the annual meeting of the Society for Pediatric Dermatology.
She presented what she believes to be the first study to formally compare the psoriasiform lesions arising in a minority of Kawasaki disease (KD) patients with the lesions of classic psoriasis. The study population consisted of 11 KD patients with psoriasiform eruptions whose median age was 1.9 years, 22 matched controls with KD and no psoriasiform lesions, and another 22 matched controls with psoriasis but not KD.
Kawasaki disease patients who developed psoriasiform eruptions had significantly more dilated coronary arteries than did those who did not, as reflected in their median maximal echocardiographic z-score of 2.7, compared with 1.8 in controls. A z-score greater than 2.5 is deemed to indicate clinically significant coronary artery dilation, noted Dr. Tom, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego.
The psoriasiform eruptions resolved within 13 months in all 11 affected KD patients, with no recurrences. In contrast, only 5 of 22 controls with classic psoriasis experienced remission during follow-up of up to 6 years.
The cutaneous distribution of the psoriasiform lesions in patients with KD was also distinctive. The eruptions were significantly less common in the head, neck, and diaper area than is the case in classic psoriasis. In addition, patients with KD and psoriasiform eruptions were significantly less likely to be overweight or obese than were controls with classic psoriasis.
Skin biopsies read by a blinded dermatopathologist showed that the psoriasiform lesions that arose during KD demonstrated suprabasilar staining of keratin 16 and increased expression of Ki-67 antigen. They differed from classic psoriasis lesions in that they displayed more crusting, serum, and an increased prevalence of bacteria at the epidermis.
There was no significant difference between KD patients with and without psoriasiform eruptions in terms of maximum levels of C-reactive protein, erythrocyte sedimentation rate, and other inflammatory markers. However, patients with psoriasiform lesions required significantly more time for their gamma-glutamyl transferase level and platelet count to return to normal.
One possible explanation for the distinct phenotype of these psoriasiform eruptions is that KD brings forth the skin lesions in patients who are predisposed to psoriasis, but without causing typical chronic psoriasis because the underlying KD is self-limited. But further study with larger numbers of patients is required for clarification, according to Dr. Tom.
Her work is supported by a career development award from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. She reported having no financial conflicts regarding this study.
COEUR D’ALENE, IDAHO – The psoriasiform eruptions that occur in a subgroup of patients with Kawasaki disease during the acute or subacute phase may be a red flag for more severe coronary artery involvement, according to a retrospective, case-control study.
Another striking feature of these psoriasiform lesions is that they go into remission. No recurrences were seen in the study population during up to 13 years of follow-up, in marked contrast to classic psoriasis, a chronic disease, Dr. Wynnis L. Tom observed at the annual meeting of the Society for Pediatric Dermatology.
She presented what she believes to be the first study to formally compare the psoriasiform lesions arising in a minority of Kawasaki disease (KD) patients with the lesions of classic psoriasis. The study population consisted of 11 KD patients with psoriasiform eruptions whose median age was 1.9 years, 22 matched controls with KD and no psoriasiform lesions, and another 22 matched controls with psoriasis but not KD.
Kawasaki disease patients who developed psoriasiform eruptions had significantly more dilated coronary arteries than did those who did not, as reflected in their median maximal echocardiographic z-score of 2.7, compared with 1.8 in controls. A z-score greater than 2.5 is deemed to indicate clinically significant coronary artery dilation, noted Dr. Tom, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego.
The psoriasiform eruptions resolved within 13 months in all 11 affected KD patients, with no recurrences. In contrast, only 5 of 22 controls with classic psoriasis experienced remission during follow-up of up to 6 years.
The cutaneous distribution of the psoriasiform lesions in patients with KD was also distinctive. The eruptions were significantly less common in the head, neck, and diaper area than is the case in classic psoriasis. In addition, patients with KD and psoriasiform eruptions were significantly less likely to be overweight or obese than were controls with classic psoriasis.
Skin biopsies read by a blinded dermatopathologist showed that the psoriasiform lesions that arose during KD demonstrated suprabasilar staining of keratin 16 and increased expression of Ki-67 antigen. They differed from classic psoriasis lesions in that they displayed more crusting, serum, and an increased prevalence of bacteria at the epidermis.
There was no significant difference between KD patients with and without psoriasiform eruptions in terms of maximum levels of C-reactive protein, erythrocyte sedimentation rate, and other inflammatory markers. However, patients with psoriasiform lesions required significantly more time for their gamma-glutamyl transferase level and platelet count to return to normal.
One possible explanation for the distinct phenotype of these psoriasiform eruptions is that KD brings forth the skin lesions in patients who are predisposed to psoriasis, but without causing typical chronic psoriasis because the underlying KD is self-limited. But further study with larger numbers of patients is required for clarification, according to Dr. Tom.
Her work is supported by a career development award from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. She reported having no financial conflicts regarding this study.
AT THE SPD ANNUAL MEETING
Key clinical point: Further study will need to determine
if treatment, time to diagnosis, or other factors influence the
association between psoriasis-like skin lesions during the acute or subacute phase of Kawasaki disease and greater coronary artery dilatation.
Major finding: The median maximal echocardiographic z-score in Kawasaki disease patients who developed psoriasiform eruptions was 2.7, compared with 1.8 in those who did not.
Data source: A retrospective, case-control study involving 11 children with psoriasiform eruptions during Kawasaki disease, 22 matched controls with Kawasaki disease but not the psoriasis-like skin lesions, and another 22 matched controls with typical psoriasis but not Kawasaki disease.
Disclosures: The study presenter reported having no financial conflicts.
Weight Loss: Most Obese Adults Aren’t Even Trying
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
CHICAGO – Nearly 60% of obese adults aren’t currently taking any steps to lose weight, according to a large national survey.
"That’s a surprisingly high figure. It suggests a dire need to better educate the public about the health consequences of obesity and the importance of addressing the problem with their doctors," Z. Jason Wang, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Moreover, of the minority of obese U.S. adults who report they actually are trying to lose weight, only 1 in 20 is taking prescription weight loss medication or has resorted to bariatric surgery. The rest are using what Dr. Wang categorized as self-modification methods: diet, exercise, OTC weight loss agents, structured weight management programs, and/or nutritional supplements.
Patient satisfaction was much higher among those using surgery or prescription medications. Thirty-nine percent of them reported being extremely or very satisfied with their weight loss method, compared with just 20% using only self-modification methods.
"This finding may mean that diet and exercise alone just don’t work for a lot of people," said Dr. Wang, director of health economics and outcomes research at Eisai in Woodcliff Lake, N.J.
He presented an analysis of data obtained from 22,927 obese adult participants in the 2012 National Health and Wellness Survey, an annual Internet-based survey which samples a demographically representative slice of the adult U.S. population.
Fully 59% of the obese respondents indicated they aren’t taking any action in an effort to lose weight. A mere 2% reported taking prescription weight loss medication or having bariatric surgery. Another 39% were relying on self-modification methods.
The National Health and Wellness Survey is conducted by Kantar Health, a health care industry consulting company. The analysis was supported by Eisai. The presenter is an Eisai employee.
Pull the hair for pediatric alopecia diagnosis
COEUR D’ALENE, IDAHO – The most important test in determining the cause of diffuse hair loss in children and adolescents is a gentle hair pull, Dr. Elise A. Olsen said at the annual meeting of the Society for Pediatric Dermatology.
"This is something you should do in all patients with alopecia, accompanied by looking under the microscope at the hair you’ve pulled. Grasp a small clump of hair close to the scalp and gently pull through to the ends. It’s important that you do it not just in one area but all over, in various places on the scalp. Coming away with three or four hairs per pull is abnormal. Be especially gentle in young children because you can actually induce what looks like a loose anagen syndrome, confusing the picture," explained Dr. Olsen, professor of dermatology and medicine and director of the hair disorders research and treatment center at Duke University in Durham, N.C.
She described how to use the hair pull and other tools to differentiate between telogen effluvium, alopecia areata, androgenetic alopecia, loose anagen syndrome, and short anagen syndrome.
Telogen effluvium: This condition is characterized by a global decrease in hair density. This global reduction can be confirmed by performing a midline part on the back and top of the scalp, which should show a similarly widened, thinned part.
Microscopic examination of the proximal end of all hairs that come off with hair pulls in an adolescent with telogen effluvium should show them to be telogen hairs.
"If you see any anagen hairs, that’s abnormal, and I would urge you to think about another condition, like loose anagen syndrome or alopecia areata," Dr. Olsen said.
Potential etiologies of telogen effluvium include stress, thyroid disease, medication side effects, vitamin A intake in excess of 15,000 IU/day, and numerous diet or nutritional deficits.
"Diet is incredibly important in figuring out the cause of telogen effluvium, particularly in children and adolescents, where you might be dealing with bulimia, anorexia nervosa, or another abnormal diet," according to the dermatologist.
The relationship between hair loss and iron deficiency is a matter of long-standing controversy. Low iron levels have often been linked to hair loss. As yet, however, there is no well-controlled clinical trial showing that iron replacement improves telogen effluvium in an iron-deficient patient.
Isotretinoin tops the list of medications that can cause telogen effluvium in pediatric patients. Other drugs that need to be considered include sodium valproate, antidepressants, lithium, and medications for attention-deficit/hyperactivity disorder.
Vitamin supplements should be stopped for at least 24 hours before conducting screening laboratory testing in a patient with telogen effluvium. Dr. Olsen recommended ordering a CBC with differential; thyroid-stimulating hormone and free thyroxine; serum ferritin; total iron binding capacity; and an erythrocyte sedimentation rate to screen for occult inflammatory conditions, which would skew the ferritin results. While a serum ferritin less than 40 ng/mL ordinarily has 98% sensitivity and specificity for iron deficiency, the bar rises to less than 70 ng/mL in patients with any kind of underlying systemic inflammation.
Alopecia areata: This form of diffuse hair loss can look clinically just like telogen effluvium. The key distinguishing feature is a positive hair pull showing not only telogen hairs but dystrophic, "exclamation point" anagen hairs as well.
"These anagen hairs are broken off or bayonet-like in appearance, and there’s usually a distortion of the hair shaft diameter as well," she explained.
Scalp dermoscopy will show yellow dots of keratinaceous debris in the empty follicles of patients with alopecia areata, an uncommon condition in children.
Androgenetic alopecia: The most useful clue in differentiating this condition from telogen effluvium is that a midline part will be widened on the central scalp but never over the occiput. The gentle hair pull typically doesn’t yield any hairs except in affected areas on the top portion of the scalp. Any hairs produced via the hair pull will be telogen hairs, and they will typically vary in diameter. Dermoscopy may show perifollicular pigmentation in areas of hair loss.
"If you diagnose alopecia areata in an adolescent, there are some key things you need to discuss with the parents," Dr. Olsen stressed.
For example, their child is likely to have a more rapidly progressive course of hair loss. And affected girls are at increased risk for underlying hyperandrogenemia-related symptoms, including hirsutism, insulin resistance, polycystic ovarian syndrome, and metabolic syndrome. A finding of acanthosis nigricans in a nonobese teen is a very good indicator that they may have underlying insulin resistance.
Loose anagen syndrome: This condition, whose onset is usually before 8 years of age, is characterized by short, very slow-growing hair. The hair pull yields anagen hairs, which under the microscope have a distinctive appearance marked by a rolled-up proximal tip.
Short anagen syndrome: The hallmarks here are decreased hair density, increased shedding, and an inability to grow hair long. On the hair pull, affected patients have an increased number of telogen hairs. The telogen hairs, which are newly regrowing in response to the truncated anagen cycle, will have a tapered tip at the distal end.
Scalp biopsy is of variable utility in diagnosing the cause of diffuse hair loss in young patients. It can be used to make the diagnosis of certain conditions, including acute alopecia areata, trichotillomania, connective tissue disease, infection, tumor, and scarring disorders. However, scalp biopsy can’t be used to make a definitive diagnosis of telogen effluvium, androgenetic alopecia, or long-standing alopecia areata – it can only be suggestive.
Pathologic standards dictate that the scalp biopsy should always be 4 mm. Normal white adults have roughly 36 follicles per 4-mm biopsy. African American adults have about 22. While no standard numbers have been established for children, the number of follicles per biopsy is higher than in adults because of the child’s smaller head size. After all, the number of follicles present at birth is what an individual will carry throughout life, Dr. Olsen explained.
It’s important that patients and parents understand the need for patience regarding hair regrowth, which takes 6-12 months after the cause of alopecia has been identified and eliminated. That means, for example, that in a patient with thyroid disease the clock for regrowth starts ticking not at the time treatment begins, but when the patient becomes euthyroid.
Dr. Olsen reported serving as a consultant to Canfield Scientific and Allergan.
COEUR D’ALENE, IDAHO – The most important test in determining the cause of diffuse hair loss in children and adolescents is a gentle hair pull, Dr. Elise A. Olsen said at the annual meeting of the Society for Pediatric Dermatology.
"This is something you should do in all patients with alopecia, accompanied by looking under the microscope at the hair you’ve pulled. Grasp a small clump of hair close to the scalp and gently pull through to the ends. It’s important that you do it not just in one area but all over, in various places on the scalp. Coming away with three or four hairs per pull is abnormal. Be especially gentle in young children because you can actually induce what looks like a loose anagen syndrome, confusing the picture," explained Dr. Olsen, professor of dermatology and medicine and director of the hair disorders research and treatment center at Duke University in Durham, N.C.
She described how to use the hair pull and other tools to differentiate between telogen effluvium, alopecia areata, androgenetic alopecia, loose anagen syndrome, and short anagen syndrome.
Telogen effluvium: This condition is characterized by a global decrease in hair density. This global reduction can be confirmed by performing a midline part on the back and top of the scalp, which should show a similarly widened, thinned part.
Microscopic examination of the proximal end of all hairs that come off with hair pulls in an adolescent with telogen effluvium should show them to be telogen hairs.
"If you see any anagen hairs, that’s abnormal, and I would urge you to think about another condition, like loose anagen syndrome or alopecia areata," Dr. Olsen said.
Potential etiologies of telogen effluvium include stress, thyroid disease, medication side effects, vitamin A intake in excess of 15,000 IU/day, and numerous diet or nutritional deficits.
"Diet is incredibly important in figuring out the cause of telogen effluvium, particularly in children and adolescents, where you might be dealing with bulimia, anorexia nervosa, or another abnormal diet," according to the dermatologist.
The relationship between hair loss and iron deficiency is a matter of long-standing controversy. Low iron levels have often been linked to hair loss. As yet, however, there is no well-controlled clinical trial showing that iron replacement improves telogen effluvium in an iron-deficient patient.
Isotretinoin tops the list of medications that can cause telogen effluvium in pediatric patients. Other drugs that need to be considered include sodium valproate, antidepressants, lithium, and medications for attention-deficit/hyperactivity disorder.
Vitamin supplements should be stopped for at least 24 hours before conducting screening laboratory testing in a patient with telogen effluvium. Dr. Olsen recommended ordering a CBC with differential; thyroid-stimulating hormone and free thyroxine; serum ferritin; total iron binding capacity; and an erythrocyte sedimentation rate to screen for occult inflammatory conditions, which would skew the ferritin results. While a serum ferritin less than 40 ng/mL ordinarily has 98% sensitivity and specificity for iron deficiency, the bar rises to less than 70 ng/mL in patients with any kind of underlying systemic inflammation.
Alopecia areata: This form of diffuse hair loss can look clinically just like telogen effluvium. The key distinguishing feature is a positive hair pull showing not only telogen hairs but dystrophic, "exclamation point" anagen hairs as well.
"These anagen hairs are broken off or bayonet-like in appearance, and there’s usually a distortion of the hair shaft diameter as well," she explained.
Scalp dermoscopy will show yellow dots of keratinaceous debris in the empty follicles of patients with alopecia areata, an uncommon condition in children.
Androgenetic alopecia: The most useful clue in differentiating this condition from telogen effluvium is that a midline part will be widened on the central scalp but never over the occiput. The gentle hair pull typically doesn’t yield any hairs except in affected areas on the top portion of the scalp. Any hairs produced via the hair pull will be telogen hairs, and they will typically vary in diameter. Dermoscopy may show perifollicular pigmentation in areas of hair loss.
"If you diagnose alopecia areata in an adolescent, there are some key things you need to discuss with the parents," Dr. Olsen stressed.
For example, their child is likely to have a more rapidly progressive course of hair loss. And affected girls are at increased risk for underlying hyperandrogenemia-related symptoms, including hirsutism, insulin resistance, polycystic ovarian syndrome, and metabolic syndrome. A finding of acanthosis nigricans in a nonobese teen is a very good indicator that they may have underlying insulin resistance.
Loose anagen syndrome: This condition, whose onset is usually before 8 years of age, is characterized by short, very slow-growing hair. The hair pull yields anagen hairs, which under the microscope have a distinctive appearance marked by a rolled-up proximal tip.
Short anagen syndrome: The hallmarks here are decreased hair density, increased shedding, and an inability to grow hair long. On the hair pull, affected patients have an increased number of telogen hairs. The telogen hairs, which are newly regrowing in response to the truncated anagen cycle, will have a tapered tip at the distal end.
Scalp biopsy is of variable utility in diagnosing the cause of diffuse hair loss in young patients. It can be used to make the diagnosis of certain conditions, including acute alopecia areata, trichotillomania, connective tissue disease, infection, tumor, and scarring disorders. However, scalp biopsy can’t be used to make a definitive diagnosis of telogen effluvium, androgenetic alopecia, or long-standing alopecia areata – it can only be suggestive.
Pathologic standards dictate that the scalp biopsy should always be 4 mm. Normal white adults have roughly 36 follicles per 4-mm biopsy. African American adults have about 22. While no standard numbers have been established for children, the number of follicles per biopsy is higher than in adults because of the child’s smaller head size. After all, the number of follicles present at birth is what an individual will carry throughout life, Dr. Olsen explained.
It’s important that patients and parents understand the need for patience regarding hair regrowth, which takes 6-12 months after the cause of alopecia has been identified and eliminated. That means, for example, that in a patient with thyroid disease the clock for regrowth starts ticking not at the time treatment begins, but when the patient becomes euthyroid.
Dr. Olsen reported serving as a consultant to Canfield Scientific and Allergan.
COEUR D’ALENE, IDAHO – The most important test in determining the cause of diffuse hair loss in children and adolescents is a gentle hair pull, Dr. Elise A. Olsen said at the annual meeting of the Society for Pediatric Dermatology.
"This is something you should do in all patients with alopecia, accompanied by looking under the microscope at the hair you’ve pulled. Grasp a small clump of hair close to the scalp and gently pull through to the ends. It’s important that you do it not just in one area but all over, in various places on the scalp. Coming away with three or four hairs per pull is abnormal. Be especially gentle in young children because you can actually induce what looks like a loose anagen syndrome, confusing the picture," explained Dr. Olsen, professor of dermatology and medicine and director of the hair disorders research and treatment center at Duke University in Durham, N.C.
She described how to use the hair pull and other tools to differentiate between telogen effluvium, alopecia areata, androgenetic alopecia, loose anagen syndrome, and short anagen syndrome.
Telogen effluvium: This condition is characterized by a global decrease in hair density. This global reduction can be confirmed by performing a midline part on the back and top of the scalp, which should show a similarly widened, thinned part.
Microscopic examination of the proximal end of all hairs that come off with hair pulls in an adolescent with telogen effluvium should show them to be telogen hairs.
"If you see any anagen hairs, that’s abnormal, and I would urge you to think about another condition, like loose anagen syndrome or alopecia areata," Dr. Olsen said.
Potential etiologies of telogen effluvium include stress, thyroid disease, medication side effects, vitamin A intake in excess of 15,000 IU/day, and numerous diet or nutritional deficits.
"Diet is incredibly important in figuring out the cause of telogen effluvium, particularly in children and adolescents, where you might be dealing with bulimia, anorexia nervosa, or another abnormal diet," according to the dermatologist.
The relationship between hair loss and iron deficiency is a matter of long-standing controversy. Low iron levels have often been linked to hair loss. As yet, however, there is no well-controlled clinical trial showing that iron replacement improves telogen effluvium in an iron-deficient patient.
Isotretinoin tops the list of medications that can cause telogen effluvium in pediatric patients. Other drugs that need to be considered include sodium valproate, antidepressants, lithium, and medications for attention-deficit/hyperactivity disorder.
Vitamin supplements should be stopped for at least 24 hours before conducting screening laboratory testing in a patient with telogen effluvium. Dr. Olsen recommended ordering a CBC with differential; thyroid-stimulating hormone and free thyroxine; serum ferritin; total iron binding capacity; and an erythrocyte sedimentation rate to screen for occult inflammatory conditions, which would skew the ferritin results. While a serum ferritin less than 40 ng/mL ordinarily has 98% sensitivity and specificity for iron deficiency, the bar rises to less than 70 ng/mL in patients with any kind of underlying systemic inflammation.
Alopecia areata: This form of diffuse hair loss can look clinically just like telogen effluvium. The key distinguishing feature is a positive hair pull showing not only telogen hairs but dystrophic, "exclamation point" anagen hairs as well.
"These anagen hairs are broken off or bayonet-like in appearance, and there’s usually a distortion of the hair shaft diameter as well," she explained.
Scalp dermoscopy will show yellow dots of keratinaceous debris in the empty follicles of patients with alopecia areata, an uncommon condition in children.
Androgenetic alopecia: The most useful clue in differentiating this condition from telogen effluvium is that a midline part will be widened on the central scalp but never over the occiput. The gentle hair pull typically doesn’t yield any hairs except in affected areas on the top portion of the scalp. Any hairs produced via the hair pull will be telogen hairs, and they will typically vary in diameter. Dermoscopy may show perifollicular pigmentation in areas of hair loss.
"If you diagnose alopecia areata in an adolescent, there are some key things you need to discuss with the parents," Dr. Olsen stressed.
For example, their child is likely to have a more rapidly progressive course of hair loss. And affected girls are at increased risk for underlying hyperandrogenemia-related symptoms, including hirsutism, insulin resistance, polycystic ovarian syndrome, and metabolic syndrome. A finding of acanthosis nigricans in a nonobese teen is a very good indicator that they may have underlying insulin resistance.
Loose anagen syndrome: This condition, whose onset is usually before 8 years of age, is characterized by short, very slow-growing hair. The hair pull yields anagen hairs, which under the microscope have a distinctive appearance marked by a rolled-up proximal tip.
Short anagen syndrome: The hallmarks here are decreased hair density, increased shedding, and an inability to grow hair long. On the hair pull, affected patients have an increased number of telogen hairs. The telogen hairs, which are newly regrowing in response to the truncated anagen cycle, will have a tapered tip at the distal end.
Scalp biopsy is of variable utility in diagnosing the cause of diffuse hair loss in young patients. It can be used to make the diagnosis of certain conditions, including acute alopecia areata, trichotillomania, connective tissue disease, infection, tumor, and scarring disorders. However, scalp biopsy can’t be used to make a definitive diagnosis of telogen effluvium, androgenetic alopecia, or long-standing alopecia areata – it can only be suggestive.
Pathologic standards dictate that the scalp biopsy should always be 4 mm. Normal white adults have roughly 36 follicles per 4-mm biopsy. African American adults have about 22. While no standard numbers have been established for children, the number of follicles per biopsy is higher than in adults because of the child’s smaller head size. After all, the number of follicles present at birth is what an individual will carry throughout life, Dr. Olsen explained.
It’s important that patients and parents understand the need for patience regarding hair regrowth, which takes 6-12 months after the cause of alopecia has been identified and eliminated. That means, for example, that in a patient with thyroid disease the clock for regrowth starts ticking not at the time treatment begins, but when the patient becomes euthyroid.
Dr. Olsen reported serving as a consultant to Canfield Scientific and Allergan.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Childhood eczema takes financial, emotional toll on families
COEUR D’ALENE, ID. – A new study puts a price on the financial and emotional costs of childhood atopic dermatitis – and concludes both are much steeper than generally recognized.
Moreover, among low-income families, a significant correlation was documented between the monthly financial cost of atopic dermatitis care and the emotional burden imposed by the disease as reflected in higher CADIS (Childhood Atopic Dermatitis Impact Scale) scores, Ms. Michelle G. Filanovsky reported at the annual meeting of the Society for Pediatric Dermatology.
"Our study is the first to correlate financial burden with emotional impact of atopic dermatitis for patients of lower socioeconomic status. This has great implications for how practitioners can help lessen the burden of the disease: Perhaps by helping families lower the cost of disease, we can help lower the emotional burden," observed Ms. Filanovsky, a medical student at Case Western Reserve University, Cleveland.
She and her coinvestigators surveyed parents or other caretakers of 79 children aged 6 months to 12 years with moderate to severe atopic dermatitis who presented to Cleveland dermatology clinics, typically because of a disease flare. Forty-five of the children were covered by Medicaid.
Subjects were queried regarding their total direct and indirect costs for atopic dermatitis care during the past 4 weeks. Direct costs include physician office and emergency department visits, prescription medications, complementary and alternative medicine, and – most importantly – over-the-counter (OTC) products, which are used extensively in atopic dermatitis care. Indirect costs included time missed from work or school and additional child-care expenses.
The disease’s emotional impact was assessed by CADIS on a 1-10 scale. CADIS addresses issues including caregiver sleep disruption and other caregiver concerns, as well as any difficulties the child is experiencing with sleep, school, socialization and conduct, self-esteem, and activity limitations.
The mean personal cost of atopic dermatitis in the month prior to the office visit was $273.78, representing on average more than one-third of a family’s available monthly money, Ms. Filanovsky said. The breakdown was $75.12 in direct costs and $198.66 for indirect costs. Families with commercial health care insurance averaged $130.58 in direct and $436.16 in indirect costs, with much lower costs in the Medicaid group.
OTC products made up the largest portion of direct costs. An average of $15.28 was spent on moisturizers, $9.42 on OTC topical steroids, $8.46 on bath products, and $4.66 on antihistamines. Time missed from work accounted for the bulk of the indirect costs.
Medicaid patients showed a significant linear correlation between CADIS scores and both total monthly costs of atopic dermatitis care and monthly costs adjusted by family size and income. However, commercially insured families did not.
Ms. Filanovsky proposed several measures as worthy of further study to help reduce the financial and emotional burden of atopic dermatitis, including insurance coverage of moisturizers, physician guidance regarding the most cost-effective OTC products, and implementation of afterwork office hours or nurse on-call visits during flares to minimize the indirect cost of care.
Survey respondents received $25 for their participation, with the funds provided by Nestle. Ms. Filanovsky reported no financial conflicts regarding this study.
COEUR D’ALENE, ID. – A new study puts a price on the financial and emotional costs of childhood atopic dermatitis – and concludes both are much steeper than generally recognized.
Moreover, among low-income families, a significant correlation was documented between the monthly financial cost of atopic dermatitis care and the emotional burden imposed by the disease as reflected in higher CADIS (Childhood Atopic Dermatitis Impact Scale) scores, Ms. Michelle G. Filanovsky reported at the annual meeting of the Society for Pediatric Dermatology.
"Our study is the first to correlate financial burden with emotional impact of atopic dermatitis for patients of lower socioeconomic status. This has great implications for how practitioners can help lessen the burden of the disease: Perhaps by helping families lower the cost of disease, we can help lower the emotional burden," observed Ms. Filanovsky, a medical student at Case Western Reserve University, Cleveland.
She and her coinvestigators surveyed parents or other caretakers of 79 children aged 6 months to 12 years with moderate to severe atopic dermatitis who presented to Cleveland dermatology clinics, typically because of a disease flare. Forty-five of the children were covered by Medicaid.
Subjects were queried regarding their total direct and indirect costs for atopic dermatitis care during the past 4 weeks. Direct costs include physician office and emergency department visits, prescription medications, complementary and alternative medicine, and – most importantly – over-the-counter (OTC) products, which are used extensively in atopic dermatitis care. Indirect costs included time missed from work or school and additional child-care expenses.
The disease’s emotional impact was assessed by CADIS on a 1-10 scale. CADIS addresses issues including caregiver sleep disruption and other caregiver concerns, as well as any difficulties the child is experiencing with sleep, school, socialization and conduct, self-esteem, and activity limitations.
The mean personal cost of atopic dermatitis in the month prior to the office visit was $273.78, representing on average more than one-third of a family’s available monthly money, Ms. Filanovsky said. The breakdown was $75.12 in direct costs and $198.66 for indirect costs. Families with commercial health care insurance averaged $130.58 in direct and $436.16 in indirect costs, with much lower costs in the Medicaid group.
OTC products made up the largest portion of direct costs. An average of $15.28 was spent on moisturizers, $9.42 on OTC topical steroids, $8.46 on bath products, and $4.66 on antihistamines. Time missed from work accounted for the bulk of the indirect costs.
Medicaid patients showed a significant linear correlation between CADIS scores and both total monthly costs of atopic dermatitis care and monthly costs adjusted by family size and income. However, commercially insured families did not.
Ms. Filanovsky proposed several measures as worthy of further study to help reduce the financial and emotional burden of atopic dermatitis, including insurance coverage of moisturizers, physician guidance regarding the most cost-effective OTC products, and implementation of afterwork office hours or nurse on-call visits during flares to minimize the indirect cost of care.
Survey respondents received $25 for their participation, with the funds provided by Nestle. Ms. Filanovsky reported no financial conflicts regarding this study.
COEUR D’ALENE, ID. – A new study puts a price on the financial and emotional costs of childhood atopic dermatitis – and concludes both are much steeper than generally recognized.
Moreover, among low-income families, a significant correlation was documented between the monthly financial cost of atopic dermatitis care and the emotional burden imposed by the disease as reflected in higher CADIS (Childhood Atopic Dermatitis Impact Scale) scores, Ms. Michelle G. Filanovsky reported at the annual meeting of the Society for Pediatric Dermatology.
"Our study is the first to correlate financial burden with emotional impact of atopic dermatitis for patients of lower socioeconomic status. This has great implications for how practitioners can help lessen the burden of the disease: Perhaps by helping families lower the cost of disease, we can help lower the emotional burden," observed Ms. Filanovsky, a medical student at Case Western Reserve University, Cleveland.
She and her coinvestigators surveyed parents or other caretakers of 79 children aged 6 months to 12 years with moderate to severe atopic dermatitis who presented to Cleveland dermatology clinics, typically because of a disease flare. Forty-five of the children were covered by Medicaid.
Subjects were queried regarding their total direct and indirect costs for atopic dermatitis care during the past 4 weeks. Direct costs include physician office and emergency department visits, prescription medications, complementary and alternative medicine, and – most importantly – over-the-counter (OTC) products, which are used extensively in atopic dermatitis care. Indirect costs included time missed from work or school and additional child-care expenses.
The disease’s emotional impact was assessed by CADIS on a 1-10 scale. CADIS addresses issues including caregiver sleep disruption and other caregiver concerns, as well as any difficulties the child is experiencing with sleep, school, socialization and conduct, self-esteem, and activity limitations.
The mean personal cost of atopic dermatitis in the month prior to the office visit was $273.78, representing on average more than one-third of a family’s available monthly money, Ms. Filanovsky said. The breakdown was $75.12 in direct costs and $198.66 for indirect costs. Families with commercial health care insurance averaged $130.58 in direct and $436.16 in indirect costs, with much lower costs in the Medicaid group.
OTC products made up the largest portion of direct costs. An average of $15.28 was spent on moisturizers, $9.42 on OTC topical steroids, $8.46 on bath products, and $4.66 on antihistamines. Time missed from work accounted for the bulk of the indirect costs.
Medicaid patients showed a significant linear correlation between CADIS scores and both total monthly costs of atopic dermatitis care and monthly costs adjusted by family size and income. However, commercially insured families did not.
Ms. Filanovsky proposed several measures as worthy of further study to help reduce the financial and emotional burden of atopic dermatitis, including insurance coverage of moisturizers, physician guidance regarding the most cost-effective OTC products, and implementation of afterwork office hours or nurse on-call visits during flares to minimize the indirect cost of care.
Survey respondents received $25 for their participation, with the funds provided by Nestle. Ms. Filanovsky reported no financial conflicts regarding this study.
AT THE SPD ANNUAL MEETING
Key clinical point: Managing the financial cost of a child’s atopic dermatitis care may mitigate the emotional burden on these children and their families.
Major finding: Families spent an average of $273.78 in direct and indirect costs, including moisturizers and other OTC products, on the care of their child with moderate to severe atopic dermatitis during the 4 weeks prior to a dermatologic office visit.
Data source: A survey of the parents or caregivers of 79 children with moderate to severe atopic dermatitis seen at Cleveland dermatology clinics.
Disclosures: Survey respondents received $25 for their participation, with the funds provided by Nestle. Ms. Filanovsky reported no financial conflicts regarding this study.
‘Soak and smear’ not superior for kids’ atopic dermatitis
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
AT THE SPD ANNUAL MEETING
Key clinical point: Physicians can anticipate equally profound improvement in the severity of pediatric atopic dermatitis after 2 weeks of twice-daily topical corticosteroid therapy, regardless of whether the medication is applied to prehydrated or dry skin.
Major finding: After 2 weeks of twice-daily topical corticosteroid therapy, pediatric atopic dermatitis patients showed roughly an 85% improvement in disease severity, regardless of whether the medication was applied to dry skin or using the soak and smear method.
Data source: A 2-week, randomized, investigator-blinded clinical trial of twice-daily topical corticosteroid therapy in 47 children and teens with atopic dermatitis.
Disclosures: The presenter reported having no financial conflicts regarding this study, conducted free of commercial support.
Reassuring results on cardiovascular safety of DPP-4 inhibitors
CHICAGO – Treatment with a dipeptidyl peptidase-4 inhibitor in real-world community practice doesn’t increase the risk of cardiovascular events in type 2 diabetic patients; in fact, the opposite was true in a very large population-based cohort study.
The study, hailed by audience members as "a tour de force" and "a really excellent study," involved 39,769 type 2 diabetes patients who initiated treatment with a dipeptidyl peptidase-4 inhibitor (DPP-4i), either as monotherapy or, far more commonly, on top of metformin, and another 39,769 patients with type 2 diabetes on metformin who initiated combination therapy with a non–DPP-4i hypoglycemic agent. The two groups were closely matched using propensity scores that incorporated 50 variables, including comorbid conditions, medications, hemoglobin A1c, and health care utilization.
The primary endpoint was a composite cardiovascular outcome comprising acute MI, stroke, coronary revascularization, and hospitalization for heart failure. The rate was 30.3 per 1,000 person-years in patients after initiating DPP-4i therapy and 34.76 per 1,000 in controls who initiated treatment with a non–DPP-4i, for a statistically significant 13% relative risk reduction, Dr. Allison B. Goldfine reported at a joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Also noteworthy was the finding that patients who initiated treatment with a DPP-4i had a 19% lower subsequent risk of hospitalization for heart failure than did those who initiated therapy with another glucose-lowering agent (see chart). This statistically significant reduction in heart failure hospitalization was particularly reassuring. Earlier, the roughly 16,500-subject, randomized, SAVOR-TIMI 53 trial showed a significant 27% increased risk of hospitalization for heart failure in patients assigned to saxagliptin, compared with placebo-treated controls (N. Engl. J. Med. 2013;369:1317-26).
Moreover, the EXAMINE study, another randomized trial, albeit smaller, reported a nonsignificant 19% increased relative risk of hospitalization for heart failure, compared with placebo (N. Engl. J. Med. 2013;369:1327-35). These findings raised a red flag at the Food and Drug Administration, which sought additional information.
Both SAVOR-TIMI 53 and EXAMINE involved type 2 diabetes patients with known cardiovascular disease at enrollment. In the new population-based cohort study, 7,293 of the 39,769 patient pairs had baseline cardiovascular disease. Reassuringly, those with baseline cardiovascular disease had a 12% relative risk reduction for the composite cardiovascular endpoint and a 16% lower risk of hospitalization for heart failure after going on a DPP-4i, compared with controls who initiated treatment with a non–DPP-4i. Both risk reductions barely missed achieving statistical significance because of insufficient patient numbers, said Dr. Goldfine, head of the section of clinical, behavioral, and outcomes research at the Joslin Diabetes Center, Boston, and an endocrinologist at Harvard University.
To investigate the additional possibility that going on DPP-4i therapy might somehow predispose to mild heart failure not severe enough to result in hospitalization, she and her coinvestigators looked at new use of loop diuretics among matched patient pairs not on that medication class at baseline. The risk of getting a new prescription for a loop diuretic during follow-up turned out to be lower by a statistically significant 26% in DPP-4i users, compared with nonusers.
The study utilized the UnitedHealthcare insurance claims database for 2005-2012. From an initial population of 4.35 million patients with type 2 diabetes, Dr. Goldfine and her coworkers utilized propensity scores to whittle down to a study population of just less than 40,000 very closely matched patient pairs.
The great majority of DPP-4i users in the study were on sitagliptin (Januvia), the first-in-class agent to reach the market.
The study was funded by the National Institutes of Health. Dr. Goldfine reported serving as an investigator on studies funded by Daiichi Sankyo, NovoNordisk, and other companies.
CHICAGO – Treatment with a dipeptidyl peptidase-4 inhibitor in real-world community practice doesn’t increase the risk of cardiovascular events in type 2 diabetic patients; in fact, the opposite was true in a very large population-based cohort study.
The study, hailed by audience members as "a tour de force" and "a really excellent study," involved 39,769 type 2 diabetes patients who initiated treatment with a dipeptidyl peptidase-4 inhibitor (DPP-4i), either as monotherapy or, far more commonly, on top of metformin, and another 39,769 patients with type 2 diabetes on metformin who initiated combination therapy with a non–DPP-4i hypoglycemic agent. The two groups were closely matched using propensity scores that incorporated 50 variables, including comorbid conditions, medications, hemoglobin A1c, and health care utilization.

The primary endpoint was a composite cardiovascular outcome comprising acute MI, stroke, coronary revascularization, and hospitalization for heart failure. The rate was 30.3 per 1,000 person-years in patients after initiating DPP-4i therapy and 34.76 per 1,000 in controls who initiated treatment with a non–DPP-4i, for a statistically significant 13% relative risk reduction, Dr. Allison B. Goldfine reported at a joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Also noteworthy was the finding that patients who initiated treatment with a DPP-4i had a 19% lower subsequent risk of hospitalization for heart failure than did those who initiated therapy with another glucose-lowering agent (see chart). This statistically significant reduction in heart failure hospitalization was particularly reassuring. Earlier, the roughly 16,500-subject, randomized, SAVOR-TIMI 53 trial showed a significant 27% increased risk of hospitalization for heart failure in patients assigned to saxagliptin, compared with placebo-treated controls (N. Engl. J. Med. 2013;369:1317-26).
Moreover, the EXAMINE study, another randomized trial, albeit smaller, reported a nonsignificant 19% increased relative risk of hospitalization for heart failure, compared with placebo (N. Engl. J. Med. 2013;369:1327-35). These findings raised a red flag at the Food and Drug Administration, which sought additional information.
Both SAVOR-TIMI 53 and EXAMINE involved type 2 diabetes patients with known cardiovascular disease at enrollment. In the new population-based cohort study, 7,293 of the 39,769 patient pairs had baseline cardiovascular disease. Reassuringly, those with baseline cardiovascular disease had a 12% relative risk reduction for the composite cardiovascular endpoint and a 16% lower risk of hospitalization for heart failure after going on a DPP-4i, compared with controls who initiated treatment with a non–DPP-4i. Both risk reductions barely missed achieving statistical significance because of insufficient patient numbers, said Dr. Goldfine, head of the section of clinical, behavioral, and outcomes research at the Joslin Diabetes Center, Boston, and an endocrinologist at Harvard University.
To investigate the additional possibility that going on DPP-4i therapy might somehow predispose to mild heart failure not severe enough to result in hospitalization, she and her coinvestigators looked at new use of loop diuretics among matched patient pairs not on that medication class at baseline. The risk of getting a new prescription for a loop diuretic during follow-up turned out to be lower by a statistically significant 26% in DPP-4i users, compared with nonusers.
The study utilized the UnitedHealthcare insurance claims database for 2005-2012. From an initial population of 4.35 million patients with type 2 diabetes, Dr. Goldfine and her coworkers utilized propensity scores to whittle down to a study population of just less than 40,000 very closely matched patient pairs.
The great majority of DPP-4i users in the study were on sitagliptin (Januvia), the first-in-class agent to reach the market.
The study was funded by the National Institutes of Health. Dr. Goldfine reported serving as an investigator on studies funded by Daiichi Sankyo, NovoNordisk, and other companies.
CHICAGO – Treatment with a dipeptidyl peptidase-4 inhibitor in real-world community practice doesn’t increase the risk of cardiovascular events in type 2 diabetic patients; in fact, the opposite was true in a very large population-based cohort study.
The study, hailed by audience members as "a tour de force" and "a really excellent study," involved 39,769 type 2 diabetes patients who initiated treatment with a dipeptidyl peptidase-4 inhibitor (DPP-4i), either as monotherapy or, far more commonly, on top of metformin, and another 39,769 patients with type 2 diabetes on metformin who initiated combination therapy with a non–DPP-4i hypoglycemic agent. The two groups were closely matched using propensity scores that incorporated 50 variables, including comorbid conditions, medications, hemoglobin A1c, and health care utilization.

The primary endpoint was a composite cardiovascular outcome comprising acute MI, stroke, coronary revascularization, and hospitalization for heart failure. The rate was 30.3 per 1,000 person-years in patients after initiating DPP-4i therapy and 34.76 per 1,000 in controls who initiated treatment with a non–DPP-4i, for a statistically significant 13% relative risk reduction, Dr. Allison B. Goldfine reported at a joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Also noteworthy was the finding that patients who initiated treatment with a DPP-4i had a 19% lower subsequent risk of hospitalization for heart failure than did those who initiated therapy with another glucose-lowering agent (see chart). This statistically significant reduction in heart failure hospitalization was particularly reassuring. Earlier, the roughly 16,500-subject, randomized, SAVOR-TIMI 53 trial showed a significant 27% increased risk of hospitalization for heart failure in patients assigned to saxagliptin, compared with placebo-treated controls (N. Engl. J. Med. 2013;369:1317-26).
Moreover, the EXAMINE study, another randomized trial, albeit smaller, reported a nonsignificant 19% increased relative risk of hospitalization for heart failure, compared with placebo (N. Engl. J. Med. 2013;369:1327-35). These findings raised a red flag at the Food and Drug Administration, which sought additional information.
Both SAVOR-TIMI 53 and EXAMINE involved type 2 diabetes patients with known cardiovascular disease at enrollment. In the new population-based cohort study, 7,293 of the 39,769 patient pairs had baseline cardiovascular disease. Reassuringly, those with baseline cardiovascular disease had a 12% relative risk reduction for the composite cardiovascular endpoint and a 16% lower risk of hospitalization for heart failure after going on a DPP-4i, compared with controls who initiated treatment with a non–DPP-4i. Both risk reductions barely missed achieving statistical significance because of insufficient patient numbers, said Dr. Goldfine, head of the section of clinical, behavioral, and outcomes research at the Joslin Diabetes Center, Boston, and an endocrinologist at Harvard University.
To investigate the additional possibility that going on DPP-4i therapy might somehow predispose to mild heart failure not severe enough to result in hospitalization, she and her coinvestigators looked at new use of loop diuretics among matched patient pairs not on that medication class at baseline. The risk of getting a new prescription for a loop diuretic during follow-up turned out to be lower by a statistically significant 26% in DPP-4i users, compared with nonusers.
The study utilized the UnitedHealthcare insurance claims database for 2005-2012. From an initial population of 4.35 million patients with type 2 diabetes, Dr. Goldfine and her coworkers utilized propensity scores to whittle down to a study population of just less than 40,000 very closely matched patient pairs.
The great majority of DPP-4i users in the study were on sitagliptin (Januvia), the first-in-class agent to reach the market.
The study was funded by the National Institutes of Health. Dr. Goldfine reported serving as an investigator on studies funded by Daiichi Sankyo, NovoNordisk, and other companies.
AT ICE/ENDO 2014
Key clinical point: Prescribing a DPP-4 inhibitor in type 2 diabetes appears safe from a cardiovascular risk standpoint in community practice, contrary to earlier concerns.
Major finding: Patients with type 2 diabetes who initiated therapy with a DPP-4 inhibitor had a 13% reduction in the subsequent risk of a composite cardiovascular endpoint and a 19% reduction in hospitalization for heart failure, compared with closely-matched patients who initiated treatment with hypoglycemic agents from other classes.
Data source: A population-based cohort study of 39,769 pairs of adults with type 2 diabetes. Half initiated treatment with a dipeptidyl peptidase-4 inhibitor, the other half with a drug from a different class of antihyperglycemic agents.
Disclosures: The study was sponsored by the National Institutes of Health. Dr. Goldfine reported serving as an investigator on studies funded by Daiichi Sankyo, NovoNordisk, and other companies.
Etomidate drip quickly curbs severe hypercortisolism
CHICAGO – Continuous intravenous infusion of etomidate safely and swiftly gains control of severe hypercortisolism in patients with adrenocorticotropic hormone–dependent Cushing’s syndrome when conventional presurgical oral treatment is problematic.
"From our cumulative experience, we have now developed a standardized titrated etomidate infusion protocol, which should provide clinicians with a simple, safe, and effective means to lower serum cortisol in patients with severe clinical, metabolic, and neuropsychiatric consequences of prodigious hypercortisolism as a bridge to definitive medical or surgical therapy," explained Dr. Katarzyna G. Zarnecki at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Etomidate (Amidate) is a sedative hypnotic agent with an excellent cardiovascular safety profile. It is widely used in emergency settings, such as reduction of dislocated joints and cardioversion. It suppresses adrenal steroidogenesis by potently inhibiting 11-beta hydroxylase. Fortunately for endocrinologic purposes, etomidate suppresses cortisol synthesis even at subhypnotic doses. In using it off label for management of severe hypercortisolism, it’s essential to keep the drug at subhypnotic doses, meaning not more than 0.3 mg/kg per hour, emphasized Dr. Zarnecki of the University of Wisconsin, Milwaukee.
Dr. Zarnecki and her coworkers utilize as their standard etomidate infusion protocol an initial 5-mg bolus followed by an infusion at 0.02 mg/kg per hour, with dose titration in increments of 0.01-0.02 mg/kg per hour every 4-6 hours based on changes in serum cortisol level. The goal is to bring the cortisol down to a target range of 10-20 mcg/dL.
She presented an illustrative six-patient series in which she and her colleagues turned to continuous infusion of etomidate because conventional oral therapy would have taken too long to rein in the severe hypercortisolism or because medication side effects were intolerable.
Mean baseline pretreatment serum cortisol was 138 mcg/dL, with an adrenocorticotropic hormone level of 419 pg/mL. Five of the six patients reached the goal of 10-20 mcg/dL in an average time of 64 hours. The mean rate of serum cortisol reduction was 1.93 mcg/dL per hour. The average etomidate infusion rate at the time the target level was reached was 0.07 mg/kg per hour, with a maximum rate of 0.1 mg/kg per hour. Monitoring via the Richmond Agitation Sedation Scale confirmed that none of the patients experienced sedative effects.
In the sole patient who didn’t reach goal, etomidate therapy was suspended because the patient entered palliative care because of extensive tumor progression.
Dr. Zarnecki reported having no financial conflicts of interest.
CHICAGO – Continuous intravenous infusion of etomidate safely and swiftly gains control of severe hypercortisolism in patients with adrenocorticotropic hormone–dependent Cushing’s syndrome when conventional presurgical oral treatment is problematic.
"From our cumulative experience, we have now developed a standardized titrated etomidate infusion protocol, which should provide clinicians with a simple, safe, and effective means to lower serum cortisol in patients with severe clinical, metabolic, and neuropsychiatric consequences of prodigious hypercortisolism as a bridge to definitive medical or surgical therapy," explained Dr. Katarzyna G. Zarnecki at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Etomidate (Amidate) is a sedative hypnotic agent with an excellent cardiovascular safety profile. It is widely used in emergency settings, such as reduction of dislocated joints and cardioversion. It suppresses adrenal steroidogenesis by potently inhibiting 11-beta hydroxylase. Fortunately for endocrinologic purposes, etomidate suppresses cortisol synthesis even at subhypnotic doses. In using it off label for management of severe hypercortisolism, it’s essential to keep the drug at subhypnotic doses, meaning not more than 0.3 mg/kg per hour, emphasized Dr. Zarnecki of the University of Wisconsin, Milwaukee.
Dr. Zarnecki and her coworkers utilize as their standard etomidate infusion protocol an initial 5-mg bolus followed by an infusion at 0.02 mg/kg per hour, with dose titration in increments of 0.01-0.02 mg/kg per hour every 4-6 hours based on changes in serum cortisol level. The goal is to bring the cortisol down to a target range of 10-20 mcg/dL.
She presented an illustrative six-patient series in which she and her colleagues turned to continuous infusion of etomidate because conventional oral therapy would have taken too long to rein in the severe hypercortisolism or because medication side effects were intolerable.
Mean baseline pretreatment serum cortisol was 138 mcg/dL, with an adrenocorticotropic hormone level of 419 pg/mL. Five of the six patients reached the goal of 10-20 mcg/dL in an average time of 64 hours. The mean rate of serum cortisol reduction was 1.93 mcg/dL per hour. The average etomidate infusion rate at the time the target level was reached was 0.07 mg/kg per hour, with a maximum rate of 0.1 mg/kg per hour. Monitoring via the Richmond Agitation Sedation Scale confirmed that none of the patients experienced sedative effects.
In the sole patient who didn’t reach goal, etomidate therapy was suspended because the patient entered palliative care because of extensive tumor progression.
Dr. Zarnecki reported having no financial conflicts of interest.
CHICAGO – Continuous intravenous infusion of etomidate safely and swiftly gains control of severe hypercortisolism in patients with adrenocorticotropic hormone–dependent Cushing’s syndrome when conventional presurgical oral treatment is problematic.
"From our cumulative experience, we have now developed a standardized titrated etomidate infusion protocol, which should provide clinicians with a simple, safe, and effective means to lower serum cortisol in patients with severe clinical, metabolic, and neuropsychiatric consequences of prodigious hypercortisolism as a bridge to definitive medical or surgical therapy," explained Dr. Katarzyna G. Zarnecki at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Etomidate (Amidate) is a sedative hypnotic agent with an excellent cardiovascular safety profile. It is widely used in emergency settings, such as reduction of dislocated joints and cardioversion. It suppresses adrenal steroidogenesis by potently inhibiting 11-beta hydroxylase. Fortunately for endocrinologic purposes, etomidate suppresses cortisol synthesis even at subhypnotic doses. In using it off label for management of severe hypercortisolism, it’s essential to keep the drug at subhypnotic doses, meaning not more than 0.3 mg/kg per hour, emphasized Dr. Zarnecki of the University of Wisconsin, Milwaukee.
Dr. Zarnecki and her coworkers utilize as their standard etomidate infusion protocol an initial 5-mg bolus followed by an infusion at 0.02 mg/kg per hour, with dose titration in increments of 0.01-0.02 mg/kg per hour every 4-6 hours based on changes in serum cortisol level. The goal is to bring the cortisol down to a target range of 10-20 mcg/dL.
She presented an illustrative six-patient series in which she and her colleagues turned to continuous infusion of etomidate because conventional oral therapy would have taken too long to rein in the severe hypercortisolism or because medication side effects were intolerable.
Mean baseline pretreatment serum cortisol was 138 mcg/dL, with an adrenocorticotropic hormone level of 419 pg/mL. Five of the six patients reached the goal of 10-20 mcg/dL in an average time of 64 hours. The mean rate of serum cortisol reduction was 1.93 mcg/dL per hour. The average etomidate infusion rate at the time the target level was reached was 0.07 mg/kg per hour, with a maximum rate of 0.1 mg/kg per hour. Monitoring via the Richmond Agitation Sedation Scale confirmed that none of the patients experienced sedative effects.
In the sole patient who didn’t reach goal, etomidate therapy was suspended because the patient entered palliative care because of extensive tumor progression.
Dr. Zarnecki reported having no financial conflicts of interest.
AT ICE/ENDO 2014
Key clinical point: The anesthetic induction agent etomidate is a potent suppressor of cortisol synthesis in the adrenal cortex at subhypnotic doses, making it a safe and effective agent for management of severe hypercortisolism in Cushing’s syndrome.
Major finding: Continuous infusion of etomidate using a standardized protocol resulted in a reduction in serum cortisol from a mean of 138 mcg/dL to a goal range of 10-20 mcg/dL in an average of 64 hours.
Data source: This was a retrospective case series involving six patients with severe hypercortisolism caused by adrenocorticotropic hormone–dependent Cushing’s syndrome.
Disclosures: The study was carried out with institutional funds. The presenter reported having no financial conflicts.